Policy Evaluation: Step Therapy Prior Authorization of Combination Inhaled Corticosteroid / Long-Acting Beta-Agonists
|
|
|
- Julia Louisa Hood
- 6 years ago
- Views:
Transcription
1 Drug Use Research & Management Program OHA Division of Medical Assistance Programs 500 Summer Street NE, E35; Salem, OR Phone Fax Policy Evaluation: Step Therapy Prior Authorization of Combination Inhaled Corticosteroid / Long-Acting Beta-Agonists This review evaluates the effect of enforcing either the trial of an inhaled corticosteroid or evidence of severe disease for asthma or a trial of an anti-cholinergic and long-acting beta-agonist for Chronic Obstructive Pulmonary Disease (COPD) prior to approval of a combination inhaler. 1 This policy was initiated on January 1, 2011 and grandfathered all current patients. Automatic electronic approvals for COPD with prior use of an anti-cholinergic and long-acting beta-agonist were implemented on September 1, Background The Oregon Drug Use Review (DUR) Board reviewed combination inhaled corticosteroid / long-acting beta-agonist (ICS/LABA) use and literature at the March 2010 meeting. 2 Major literature findings from the review include: - Overall, systematic reviews and guidelines consistently demonstrate that adding LABA to ICS in adults and children with persistent asthma will improve airway function, asthma symptoms, quality of life and reduce short-acting rescue inhaler use compared to ICS dose escalation. - The benefit of adding LABA to ICS for preventing severe exacerbations and mortality for asthma patients is not supported or is unclear. - Combination ICS appears to reduce the risk of exacerbation, improve lung function, and health status in patients with COPD. - The addition of ICS to LABA does not appear to have meaningful impact on mortality and increases the risk for pneumonia. - Several systematic reviews have evaluated the association between LABA use and life threatening adverse events and the data is mixed for use of the combination of ICS/LABA. Based on FDA meta-analyses of over 60,000 patients the excess risk for asthma-related death, death or intubation, asthma hospitalization, or a composite safety measure were all significantly
2 elevated for patients using LABA. When trials were stratified by ICS at randomization, the risk appeared to be less and became non-significant, however due to lower sample sizes in these sub-analyses, the same risk excess cannot be ruled out. - On February 18, 2010, the FDA issued a safety announcement based on these data and the past advisory group deliberations. 3 The FDA recommends the following to ensure the safe use of LABA for asthma (does not extend to the use of LABA for COPD): o The use of LABAs is contraindicated without the use of an asthma controller medication such as an inhaled corticosteroid. Single-ingredient LABAs should only be used in combination with an asthma controller medication; they should not be used alone. o LABAs should only be used long-term in patients whose asthma cannot be adequately controlled on asthma controller medications. o LABAs should be used for the shortest duration of time required to achieve control of asthma symptoms and discontinued, if possible, once asthma control is achieved. Patients should then be maintained on an asthma controller medication. o Pediatric and adolescent patients who require the addition of a LABA to an inhaled corticosteroid should use a combination product containing both an inhaled corticosteroid and a LABA, to ensure compliance with both medications. Medicaid claims data analyses suggested that utilization patterns are not consistent with practice recommendations. This was consistent with a previous analysis in this population which found less than 10% of patients started on ICS/LABA had a history of ICS monotherapy use. 4 A Retrospective educational letter intervention was implemented in 2006 but was largely without effect on prescribing. - Depending on the denominator, between 6% and 14% of patients were using ICS prior to starting ICS/LABA. Using a less conservative definition of controller increased the proportion of users to 22% to 50%. - While asthma appears to represent the predominate condition treated, individuals with COPD may represent a significant proportion (13%) of individuals. - Individuals with a history of higher severity of disease were more likely to have previous controller use prior to starting ICS/LABA treatment. - General practitioners represent the largest prescribers of this drug class. Author: Ketchum May 1, 2012 Page 2
3 Previous DUR Board Recommendations included: - Prior authorize new starts of ICS/LABA inhalers. Electronically exempt individuals with any evidence of increased asthma severity of disease such as HEDIS asthma definition, >1 oral steroid claim, specialist prescriber, any previous controller prescription or evidence of COPD. Current users would be grandfathered in. Pros: Prospectively identifies patients for reduced exposure to LABA. Cons: Risks delay of care for moderate-severe asthma patients in crisis. Risks are mitigated by electronically excluding clients with descriptors of more severe disease. - Prior authorize LABA monotherapy. Electronically exempt individuals with evidence of COPD or evidence of concurrent asthma controller agent. Pros: Prospectively identifies patients at highest risk for reduced exposure to unnecessary LABA. Cons: Limited risk (currently very low utilization) of delay of care for moderate-severe COPD clients. Methods: The goals of the current analysis were to quantify any change in prescribing patterns of study groups temporal to the prior authorization policy implemented January 1, In addition, any change in use of the emergency department (ED) or hospitalizations for asthma or COPD were evaluated. Patients were identified for inclusion in the analysis if they had a fee-for-service (FFS) drug claim for any drug in Appendix 1 from January 1, 2010 through December 31, Patients also eligible for Medicare Part D as identified by benefit package codes BMM or BMD were excluded. All FFS drug and medical claims from January 1, 2009 March 31, 2010 for the identified patients were retained. These patients were further classified into four study groups; inhaled corticosteroid monotherapy (ICS), long-acting betaagonist monotherapy (LABA), combination ICS/LABA inhaler (ICSLABA) and ICS + LABA if filled within 7 days of each other. The gross trend in utilization was reported as a count of unique patients on the study group drug each month divided by the number of patients enrolled in FFS each month (PMPM). The cost trend was reported using the amount reimbursed to pharmacies PMPM. The estimated pharmacy reimbursed costs avoided each month was the difference between the predicted costs PMPM based upon the trend for the 6 months prior to the policy and the actual costs incurred PMPM, multiplied by the FFS enrollment for the month. The primary analysis focused on new users of the drugs of interest. Patients were included in the analysis if they had an index fills of a study drug in 2010 and An index fill is new fill for a drug in any of the study groups and no previous fills for any drug in Author: Ketchum May 1, 2012 Page 3
4 the study groups above for the same patient. Patients with valid demographic data between the ages of 5-64 on the index fill date were included. The age restriction was placed because the guidelines are most clear for those ages. Patients must have a minimum of 6 months FFS enrollment in the year prior to the index fill. This could be up to two discrete spans with no more than 31 days between spans. Patients were identified as patients with an asthma diagnosis if they had any medical claim data with an asthma ICD-9 code during the year prior to the index fill (Appendix 2). Patients were identified as patients with a COPD diagnosis if they had any medical claim data with a COPD ICD-9 code during the years prior to the index fill (Appendix 2). Patients were then put into the following groups and can only be in 1 group: 1) Asthma (no COPD): Asthma flag and no COPD flag 2) Asthma + COPD: Asthma flag and COPD flag 3) COPD (no Asthma): COPD flag and no Asthma flag 4) No Asthma & no COPD: neither flag Patients were further grouped as follows: All Users = Any Asthma = COPD (no Asthma) = 3 Prescriber specialties were determined from the National Provider Identifier information used upon Oregon Medicaid provider enrollment. Hospital discharges were identified with claims where International Classification of Disease, 9th edition (ICD-9) codes had a diagnosis as defined in Appendix 2. The ICD-9 could reside as primary through sixth place on the claim. Discharge dates were used rather than service date for hospital encounters. ED visits and outpatient visits were identified with claims with ICD-9 codes associated with diagnoses in Appendix 2. These were used to establish patient disease severity at baseline using claims up to 1 year prior to the index fill. Hospital and ED visits were also measured for 90 days post index fill to detect differences in drug groups. Disease severity was estimated using several measures. The number of ED, hospital encounters and fills for oral corticosteroids were constructed as possible metrics of asthma or COPD exacerbation. The Healthcare Effectiveness Data and Information Set (HEDIS) description of persistent asthma was used. A ratio of controller medication fills to total asthma medication fills was used Author: Ketchum May 1, 2012 Page 4
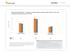
5 Jan-10 Feb-10 Mar-10 Apr-10 May-10 Jun-10 Jul-10 Aug-10 Sep-10 Oct-10 Nov-10 Dec-10 Jan-11 Feb-11 Mar-11 Apr-11 May-11 Jun-11 Jul-11 Aug-11 Sep-11 Oct-11 Nov-11 Dec-11 Policy Evaluation: Step Therapy PA of Combination ICS/LABA Inhalers to identify individuals with ratios <50% for patients who had a minimum of 4 total asthma drug prescriptions in the previous year. 2 A combined disease severity variable was created to provide the greatest sensitivity for any history of possible asthma exacerbation. The asthma severity variable was defined by any of the following: a controller to total asthma drug ratio <50%, or >1 oral steroid prescription in the previous year, or ED visit or hospitalization related for asthma or COPD in the previous year or HEDIS persistent asthma. Results The gross number of patients using any ICS/LABA combination product decreased dramatically after the policy was implemented (Figure 1). While the use of ICS does trend upward, the slope is not as dramatic. There continues to be low use of LABA as monotherapy. Figure 1 - Unique Patient Count Trend (all comers) PMPM x ICS monotherapy LABA/ICS Comb LABA Author: Ketchum May 1, 2012 Page 5
6 A summary of the drug group and diagnostic group proportions is represented in Table 1. A dramatic shift (~21%) from the ICS/LABA combination to ICS monotherapy can be observed across all groups. There is also an increase in the use of LABA monotherapy for COPD patients. General practice physicians remain the predominate prescribers of initial therapy, with pulmonologists identified as prescribers less than 7% of the time. Table 1 Study Group Proportions and Prescribers All users Any Asthma COPD (no Asthma) n= 1, count % count % count % count % count % count % Index Drug Characteristics ICS monotherapy % % % % % % LABA/ICS Comb % % % % % % LABA + ICS concurrently % % % % % % LABA % % % % % % Prescriber Specialty Pulmonary % % % % % % Pediatrics % % % % % % General Practice % % % % % % Other % % % % % % The comparative demographics for the study groups are presented in Table 2. Both the Asthma and COPD populations have more ED visits and oral steroid use than the All User populations which indicates poorer control or more disease severity or both. In addition the COPD population has more hospitalizations than the All User population. In general, there is a significant reduction in all drug use in this category from 2010 to 2011 (~45% overall). There was no asthma or COPD diagnosis on record for 43% of patients in 2010 and 46% of patients in Asthma is the predominant diagnosis of record (50% in 2010, 46% in 2011) with roughly 6-7% co-morbid with COPD. Patients with COPD and no asthma diagnosis comprise 7% of all users in 2010 and 9% in Disease severity indicators generally decreased from 2010 to 2011, notably ED and Hospital visits for asthma patients. However, COPD ED and hospital visits increased slightly Author: Ketchum May 1, 2012 Page 6
7 Table 2 Baseline Demographics and Morbidity at Time of Index Fill. All users Any Asthma COPD (no Asthma) n= 1, mean SD mean SD mean SD mean SD mean SD mean SD Asthma/ COPD ED visits (mean visits per patient in year prior) Asthma/ COPD Hospitalizations (mean hospitalization per patient in year prior) Oral Steroid Rx in year prior Age count % count % count % count % count % count % Sex (female) % % % % % % Race (non-white) % % % % % % Diagnostic Characteristics Asthma (no COPD) % % % % % % Asthma + COPD % % % % % % COPD (no Asthma) % % % % % % No Asthma & no COPD % % % % % % Disease Severity Indicators Persistent Asthma (HEDIS) % % % % % % Controller Ratio < 50% % % % % % % > 1 oral steroid % % % % % % Patients with Asthma/COPD ED or Hospital Encounter % % % % % % Any Severity Indicator % % % % % % Author: Ketchum May 1, 2012 Page 7
8 Table 3 counts patients in each study group that have any of the severity indicators. More patients using ICS monotherapy had severe disease as indicated by any of the indicators. There was an increase in the rate of ICS patients with severe disease in 2011 and is most dramatic for the COPD patients. Table 3 - Any Severity Indicator by Index Drug All users Any Asthma COPD (no Asthma) n= 1, Count % Count % Count % Count % Count % Count % ICS monotherapy % % % % % % LABA/ICS Comb % % % % % % LABA + ICS concurrently % % % % % % LABA % % % % % % Table 4 presents the number of patients started on monotherapy who were switched to a combination inhaler within 90 days. This indicator was used as a proxy of inadequate control with the monotherapy. There is no appreciable difference before or after the step therapy was implemented. Table 4 - Presence of ICSLABA Pharmacotherapy in 3 Months Following Initiation All users Any Asthma COPD (no Asthma) n= 1, count % count % count % count % count % count % ICS monotherapy % % % % % % LABA + ICS concurrently % % % % % % LABA % % % % % % Author: Ketchum May 1, 2012 Page 8
9 Table 5 represents the patients that were admitted to ED or hospital within 3 months of initiating therapy. The combination inhaler group having a diagnosis of asthma or COPD increased the number of ED or hospital visits in There was also an increase for those patients with COPD initiated on ICS or LABA monotherapy. All other groups remained similar or decreased. Table 5 - Asthma/COPD ED/Hospital Admits Within 3 Months Following Therapy Initiation All users Any Asthma COPD (no Asthma) n= 1, mean SD mean SD mean SD mean SD mean SD mean SD Mean per Patient Asthma-related ED/Hospital Visits in the 3 months after initiating: ICS monotherapy LABA/ICS Comb LABA + ICS concurrently LABA Mean per Patient COPD-related ED/Hospital Visits in the 3 months after initiating: ICS monotherapy LABA/ICS Comb LABA + ICS concurrently LABA Author: Ketchum May 1, 2012 Page 9
10 2010_ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _ _12 Patient Count Policy Evaluation: Step Therapy PA of Combination ICS/LABA Inhalers Figure 2 depicts a trend of all patients with a hospital and ED encounter with either an asthma or COPD diagnosis, regardless of drug therapy. It trends slightly downward overall in 2011 though the most prominent feature is a seasonal trend. Figure 2 Count of unique patients per month with hospital or ED encounter with asthma or COPD (no asthma) diagnosis on claim Asthma COPD Month Author: Ketchum May 1, 2012 Page 10
11 Discussion There was a temporal and significant decrease (47%) in the use of ICS/LABA products and an increase (6%) of ICS monotherapy in There was an overall 45% decrease in use of all drugs in this class. This is concerning since this group of drugs is recommended treatment for both asthma and COPD control. However, most indicators of disease severity decreased in 2011 as well. There were no increases in the use of ED or hospitals within 90 days of index fill of ICS monotherapy for asthma and patients did not switch to the ICS/LABA within 90 days of the index fill. However, there was an increase in ED and hospital admits for ICS/LABA asthma patients, despite fewer indicators of severe disease in this group. ED and hospital admits for COPD patients increased. Overall, no increase in hospital or ED visits with asthma or COPD diagnoses was detected as a result of this policy. This analysis is limited because it is retrospective and uses administrative data which is known to have the potential for missing data and miscoding. In particular, there is a known potential for loss of follow-up for patients with index fills in the latter part of 2011, where hospital and ED data may not be submitted yet by March 31, Limiting the follow-up period to 90-days was an attempt to limit this effect, but it may still account from some of the decrease in ED and hospital admits in Recommendations: 1) Continue the policy to prior authorize ICS/LABA combinations for step therapy 2) Consider loosening the electronic criteria to require only a diagnosis of COPD OR prior anti-cholinergic inhaler use 3) Implement RetroDUR education lettering on LABA monotherapy in the absence of COPD indicators 4) Further study to evaluate patient outcomes after encountering a PA for ICS/LABA 5) Further study of patients without a diagnosis Author: Ketchum May 1, 2012 Page 11
12 References: 1. OHA Medicaid FFS Prior Authorization Approval Criteria. Oregon Health Authority - OHP Policies,rules and Guidelines. Available at: Accessed April 19, Oregon Drug Use Reviiew Board. Combination Long-Acting Beta-Agonist Inhaled Corticosteroid: Summary of Clinical Evidence and Drug Utilization Evaluation. Oregon State University Drug Use Research and Management Available at: Accessed April 19, Postmarket Drug Safety Information for Patients and Providers > FDA Drug Safety Communication: New safety requirements for long-acting inhaled asthma medications called Long-Acting Beta-Agonists (LABAs). United States Food and Drug Administration Available at: Accessed April 19, Oregon Drug Use Review Board. Drug Use Evaluation: LongActing Beta Agonist (LABA). Oregon State University Drug Use Research and Management Available at: Accessed April 19, Author: Ketchum May 1, 2012 Page 12
13 Appendix 1 Drugs of Interest and Study Group Classification GSN Generic Name Route Study Group BECLOMETHASONE DIPROPIONATE IH ICS BECLOMETHASONE DIPROPIONATE IH ICS BUDESONIDE IH ICS BUDESONIDE IH ICS BUDESONIDE IH ICS BUDESONIDE IH ICS BUDESONIDE IH ICS CICLESONIDE IH ICS CICLESONIDE IH ICS 213 FLUNISOLIDE IH ICS FLUNISOLIDE/MENTHOL IH ICS FLUTICASONE PROPIONATE IH ICS FLUTICASONE PROPIONATE IH ICS FLUTICASONE PROPIONATE IH ICS FLUTICASONE PROPIONATE IH ICS FLUTICASONE PROPIONATE IH ICS FLUTICASONE PROPIONATE IH ICS MOMETASONE FUROATE IH ICS MOMETASONE FUROATE IH ICS MOMETASONE FUROATE IH ICS MOMETASONE FUROATE IH ICS MOMETASONE FUROATE IH ICS MOMETASONE FUROATE IH ICS 212 TRIAMCINOLONE ACETONIDE IH ICS BUDESONIDE/FORMOTEROL FUMARATE IH ICSLABA BUDESONIDE/FORMOTEROL FUMARATE IH ICSLABA FLUTICASONE/SALMETEROL IH ICSLABA FLUTICASONE/SALMETEROL IH ICSLABA Author: Ketchum May 1, 2012 Page 13
14 43368 FLUTICASONE/SALMETEROL IH ICSLABA FLUTICASONE/SALMETEROL IH ICSLABA FLUTICASONE/SALMETEROL IH ICSLABA FLUTICASONE/SALMETEROL IH ICSLABA MOMETASONE/FORMOTEROL IH ICSLABA MOMETASONE/FORMOTEROL IH ICSLABA ARFORMOTEROL TARTRATE IH LABA FORMOTEROL FUMARATE IH LABA FORMOTEROL FUMARATE IH LABA INDACATEROL MALEATE IH LABA SALMETEROL XINAFOATE IH LABA SALMETEROL XINAFOATE IH LABA SALMETEROL XINAFOATE IH LABA Author: Ketchum May 1, 2012 Page 14
15 Appendix 2 Diagnostic Criteria Author: Ketchum May 1, 2012 Page 15
Copyright 2012 Oregon State University. All Rights Reserved
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Drug Class Monograph
 Drug Class Monograph Class: Inhaled Corticosteroids Drugs: Aerospan (flunisolide), Advair Diskus, Advair HFA (fluticasone/salmeterol), Alvesco (ciclesonide), Arnuity Ellipta (fluticasone furoate), Asmanex
Drug Class Monograph Class: Inhaled Corticosteroids Drugs: Aerospan (flunisolide), Advair Diskus, Advair HFA (fluticasone/salmeterol), Alvesco (ciclesonide), Arnuity Ellipta (fluticasone furoate), Asmanex
Author: K. Ketchum Date: July 2015
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Inhaled Corticosteroids Drug Class Prior Authorization Protocol
 Inhaled Corticosteroids Drug Class Prior Authorization Protocol Line of Business: Medicaid P&T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has been developed through review
Inhaled Corticosteroids Drug Class Prior Authorization Protocol Line of Business: Medicaid P&T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has been developed through review
Reducing COPD Exacerbation Readmissions in a Community-Based Teaching Hospital
 Reducing COPD Exacerbation Readmissions in a Community-Based Teaching Hospital Dawn Waddell, PharmD, BCPS Clinical Pharmacy Manager Lisa Kingdon, PharmD, BCPS Clinical Pharmacy Specialist Dawn Waddell
Reducing COPD Exacerbation Readmissions in a Community-Based Teaching Hospital Dawn Waddell, PharmD, BCPS Clinical Pharmacy Manager Lisa Kingdon, PharmD, BCPS Clinical Pharmacy Specialist Dawn Waddell
Emergency Department Boarding of Psychiatric Patients in Oregon
 College of Public Health and Human Sciences Emergency Department Boarding of Psychiatric Patients in Oregon Jangho Yoon, PhD, Jeff Luck, PhD April 25, 2017 Scope Quantify the extent of psychiatric emergency
College of Public Health and Human Sciences Emergency Department Boarding of Psychiatric Patients in Oregon Jangho Yoon, PhD, Jeff Luck, PhD April 25, 2017 Scope Quantify the extent of psychiatric emergency
Inhaled Corticosteroids Drug Class Prior Authorization Protocol
 Inhaled Corticosteroids Drug Class Prior Authorization Protocol Line of Business: Medi-Cal P&T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has been developed through review
Inhaled Corticosteroids Drug Class Prior Authorization Protocol Line of Business: Medi-Cal P&T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has been developed through review
Drug Use Evaluation: Low Dose Quetiapine
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
TRELEGY ELLIPTA (fluticasone-umeclidinium-vilanterol) aerosol powder
 TRELEGY ELLIPTA (fluticasone-umeclidinium-vilanterol) aerosol powder Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific
TRELEGY ELLIPTA (fluticasone-umeclidinium-vilanterol) aerosol powder Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific
Drug Effectiveness Review Project Summary Report
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Proposal on subsidy changes for some respiratory inhalation products and access restrictions to combination inhalers.
 30 August 2011 Proposal on changes for some respiratory products and access restrictions to combination inhalers. As notified on 16 June 2011, PHARMAC has been considering options for managing the funding
30 August 2011 Proposal on changes for some respiratory products and access restrictions to combination inhalers. As notified on 16 June 2011, PHARMAC has been considering options for managing the funding
April 10 th, Bond Street, Toronto ON, M5B 1W8
 Comprehensive Research Plan: Inhaled long-acting muscarinic antagonists (LAMAs; long-acting anticholinergics) for the treatment of chronic obstructive pulmonary disease (COPD) April 10 th, 2014 30 Bond
Comprehensive Research Plan: Inhaled long-acting muscarinic antagonists (LAMAs; long-acting anticholinergics) for the treatment of chronic obstructive pulmonary disease (COPD) April 10 th, 2014 30 Bond
Outcomes: Initially, our primary definitions of pneumonia was severe pneumonia, where the subject was hospitalized
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
1.* Dosage. A. Adults
 Aerosolized Agents (Metered-Dose Inhalers) - Inhaled Anti-Inflammatory Drugs: Corticosteroids [Developed, January 1995; Revised, February, 1997; August 1997; March 1998; February 1999; January 2000; March
Aerosolized Agents (Metered-Dose Inhalers) - Inhaled Anti-Inflammatory Drugs: Corticosteroids [Developed, January 1995; Revised, February, 1997; August 1997; March 1998; February 1999; January 2000; March
GSK Clinical Study Register
 In February 2013, GlaxoSmithKline (GSK) announced a commitment to further clinical transparency through the public disclosure of GSK Clinical Study Reports (CSRs) on the GSK Clinical Study Register. The
In February 2013, GlaxoSmithKline (GSK) announced a commitment to further clinical transparency through the public disclosure of GSK Clinical Study Reports (CSRs) on the GSK Clinical Study Register. The
Louisiana Medicaid. Provider Update. Volume 27, Issue 3 May/June UNISYS Health Information Management Acquired. by Molina Healthcare
 Louisiana Medicaid Provider Update Volume 27, Issue 3 May/June 2010 UNISYS Health Information Management Acquired by Molina Healthcare Effective May 1, 2010, Molina Healthcare purchased the Health Information
Louisiana Medicaid Provider Update Volume 27, Issue 3 May/June 2010 UNISYS Health Information Management Acquired by Molina Healthcare Effective May 1, 2010, Molina Healthcare purchased the Health Information
Figure 1: Quantity Dispensed/100 Members (Ambien and Sonata on left axis)
 Sedative Hypnotic Quantity Limit Policy Impact Analysis Effective 12/6/2 OMAP implemented a 15 tablet per 3 day quantity limit on non-barbiturate sedative hypnotics for FFS patients. This policy was made
Sedative Hypnotic Quantity Limit Policy Impact Analysis Effective 12/6/2 OMAP implemented a 15 tablet per 3 day quantity limit on non-barbiturate sedative hypnotics for FFS patients. This policy was made
Quality ID #444 (NQF 1799): Medication Management for People with Asthma National Quality Strategy Domain: Efficiency and Cost Reduction
 Quality ID #444 (NQF 1799): Medication Management for People with Asthma National Quality Strategy Domain: Efficiency and Cost Reduction 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE:
Quality ID #444 (NQF 1799): Medication Management for People with Asthma National Quality Strategy Domain: Efficiency and Cost Reduction 2018 OPTIONS FOR INDIVIDUAL MEASURES: REGISTRY ONLY MEASURE TYPE:
The National Asthma Education and Prevention Program s
 Long-Acting b-agonist Among Children and Adults With Asthma Elizabeth A. Wasilevich, PhD, MPH; Sarah J. Clark, MPH; Lisa M. Cohn, MS; and Kevin J. Dombkowski, DrPH Managed Care & Healthcare Communications,
Long-Acting b-agonist Among Children and Adults With Asthma Elizabeth A. Wasilevich, PhD, MPH; Sarah J. Clark, MPH; Lisa M. Cohn, MS; and Kevin J. Dombkowski, DrPH Managed Care & Healthcare Communications,
Kirthi Gunasekera MD Respiratory Physician National Hospital of Sri Lanka Colombo,
 Kirthi Gunasekera MD Respiratory Physician National Hospital of Sri Lanka Colombo, BRONCHODILATORS: Beta Adrenoreceptor Agonists Actions Adrenoreceptor agonists have many of the same actions as epinephrine/adrenaline,
Kirthi Gunasekera MD Respiratory Physician National Hospital of Sri Lanka Colombo, BRONCHODILATORS: Beta Adrenoreceptor Agonists Actions Adrenoreceptor agonists have many of the same actions as epinephrine/adrenaline,
Clinical Policy: Roflumilast (Daliresp) Reference Number: CP.PMN.46 Effective Date: Last Review Date: 08.18
 Clinical Policy: (Daliresp) Reference Number: CP.PMN.46 Effective Date: 11.01.11 Last Review Date: 08.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Daliresp) Reference Number: CP.PMN.46 Effective Date: 11.01.11 Last Review Date: 08.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Global Initiative for Asthma (GINA) What s new in GINA 2016?
 Global Initiative for Asthma (GINA) What s new in GINA 2016? GINA Global Strategy for Asthma Management and Prevention GINA: A Brief History Established in 1993 Collaboration between NHLBI and WHO Multiple
Global Initiative for Asthma (GINA) What s new in GINA 2016? GINA Global Strategy for Asthma Management and Prevention GINA: A Brief History Established in 1993 Collaboration between NHLBI and WHO Multiple
DOD PHARMACY AND THERAPEUTICS COMMITTEE RECOMMENDATIONS INFORMATION FOR THE UNIFORM FORMULARY BENEFICIARY ADVISORY PANEL
 DOD PHARMACY AND THERAPEUTICS COMMITTEE RECOMMENDATIONS INFORMATION FOR THE UNIFORM FORMULARY BENEFICIARY ADVISORY PANEL I. Uniform Formulary Review Process Under 10 U.S.C. 1074g, as implemented by 32
DOD PHARMACY AND THERAPEUTICS COMMITTEE RECOMMENDATIONS INFORMATION FOR THE UNIFORM FORMULARY BENEFICIARY ADVISORY PANEL I. Uniform Formulary Review Process Under 10 U.S.C. 1074g, as implemented by 32
Long Term Care Formulary RS -29
 RESTRICTED USE Asthma/COPD Management 1 of 6 PROTOCOL: Asthma Glossary of Medication Acronyms: SABA: short-acting beta agonist (e.g. salbutamol) SABD: short-acting bronchodilator (e.g. ipratropium or SABA)
RESTRICTED USE Asthma/COPD Management 1 of 6 PROTOCOL: Asthma Glossary of Medication Acronyms: SABA: short-acting beta agonist (e.g. salbutamol) SABD: short-acting bronchodilator (e.g. ipratropium or SABA)
Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing
 Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing Diana M. Sobieraj, PharmD, BCPS Assistant Professor University of Connecticut School
Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing Diana M. Sobieraj, PharmD, BCPS Assistant Professor University of Connecticut School
Carisoprodolol Quantity Limit Policy Impact Analysis
 Carisoprodolol Quantity Limit Policy Impact Analysis Carisoprodolol (Soma ) is a skeletal muscle relaxant that has deemed by many to have limited effectiveness for the treatment of acute musculoskeletal
Carisoprodolol Quantity Limit Policy Impact Analysis Carisoprodolol (Soma ) is a skeletal muscle relaxant that has deemed by many to have limited effectiveness for the treatment of acute musculoskeletal
Interventions to improve adherence to inhaled steroids for asthma. Respiratory department
 Interventions to improve adherence to inhaled steroids for asthma Respiratory department Content Overview Research References Overview Asthma is a chronic breathing condition that affects more than 300
Interventions to improve adherence to inhaled steroids for asthma Respiratory department Content Overview Research References Overview Asthma is a chronic breathing condition that affects more than 300
Jan Feb Mar Apr May Jun Jul Aug Sep X X X X X X X. Jan Feb Mar Apr May Jun Jul Aug Sep Oct Nov X X X X X X X X X X X X X
 Primary Prevention Breast Cancer Prevention Member: Mammography reminder letters to female members ages 51.5-74 who are overdue to get a mammogram Jan Feb Mar Apr May Jun Jul Aug Sep Oct Nov Providers:
Primary Prevention Breast Cancer Prevention Member: Mammography reminder letters to female members ages 51.5-74 who are overdue to get a mammogram Jan Feb Mar Apr May Jun Jul Aug Sep Oct Nov Providers:
Drug Effectiveness Review Project Literature Scan Summary. Month/Year of Review: January 2015 Date of Last Review: January 2013
 Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301 1079 Phone 503 947 5220 Fax 503 947 1119 Copyright 2012 Oregon State University. All Rights
Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301 1079 Phone 503 947 5220 Fax 503 947 1119 Copyright 2012 Oregon State University. All Rights
Select Inhaled Respiratory Agents
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Adherence to asthma controller medication regimens
 Respiratory Medicine (2005) 99, 1263 1267 Adherence to asthma controller medication regimens D.A. Stempel a,, S.W. Stoloff b, J.R. Carranza Rosenzweig c, R.H. Stanford c, K.L. Ryskina d, A.P. Legorreta
Respiratory Medicine (2005) 99, 1263 1267 Adherence to asthma controller medication regimens D.A. Stempel a,, S.W. Stoloff b, J.R. Carranza Rosenzweig c, R.H. Stanford c, K.L. Ryskina d, A.P. Legorreta
ABCBS PCMH Specifications. ARKANSAS BLUE CROSS and BLUE SHIELD An Independent Licensee of the Blue Cross and Blue Shield Association
 ABCBS PCMH 2016 Specifications An Independent Licensee of the Blue Cross and Blue Shield Association Table of Contents I. Terminology 3 II. Transformation Activities 2016 List of Activities 4 3 month 5-9
ABCBS PCMH 2016 Specifications An Independent Licensee of the Blue Cross and Blue Shield Association Table of Contents I. Terminology 3 II. Transformation Activities 2016 List of Activities 4 3 month 5-9
GSK Medicine: salmeterol, Salmeterol+Fluticasone proprionate, fluticasone propionate, beclomethasone
 GSK Medicine: salmeterol, Salmeterol+Fluticasone proprionate, fluticasone propionate, beclomethasone Study No.: WWE111984/WEUSRTP2640/EPI40528 Title: The Asthma Death Case Control Study (ADCCS): Association
GSK Medicine: salmeterol, Salmeterol+Fluticasone proprionate, fluticasone propionate, beclomethasone Study No.: WWE111984/WEUSRTP2640/EPI40528 Title: The Asthma Death Case Control Study (ADCCS): Association
II: Moderate Worsening airflow limitations Dyspnea on exertion, cough, and sputum production; patient usually seeks medical
 Table 3.1. Classification of COPD Severity Stage Pulmonary Function Test Findings Symptoms I: Mild Mild airflow limitations +/ Chronic cough and sputum production; patient unaware of abnormal FEV 1 80%
Table 3.1. Classification of COPD Severity Stage Pulmonary Function Test Findings Symptoms I: Mild Mild airflow limitations +/ Chronic cough and sputum production; patient unaware of abnormal FEV 1 80%
Peak Expiratory Flow Rate (PEFR) for ED Management of Acute Asthma Exacerbation
 Peak Expiratory Flow Rate (PEFR) for ED Management of Acute Asthma Exacerbation PI: Brian Driver, MD Checklist Reviewed Inclusion and Exclusion Criteria Confirm pertinent exclusion criteria with PMP Engage
Peak Expiratory Flow Rate (PEFR) for ED Management of Acute Asthma Exacerbation PI: Brian Driver, MD Checklist Reviewed Inclusion and Exclusion Criteria Confirm pertinent exclusion criteria with PMP Engage
The clinical effectiveness and costeffectiveness. treatment of chronic asthma in children under the age of 12 years
 The clinical effectiveness and costeffectiveness of corticosteroids for the treatment of chronic asthma in children under the age of 12 years Submission of evidence from AstraZeneca UK Ltd regarding the
The clinical effectiveness and costeffectiveness of corticosteroids for the treatment of chronic asthma in children under the age of 12 years Submission of evidence from AstraZeneca UK Ltd regarding the
Online supplementary material
 Online supplementary material Add-on long-acting β2-agonist (LABA) in a separate inhaler as asthma step-up therapy versus increased dose of inhaled corticosteroid (ICS) or ICS/LABA combination inhaler
Online supplementary material Add-on long-acting β2-agonist (LABA) in a separate inhaler as asthma step-up therapy versus increased dose of inhaled corticosteroid (ICS) or ICS/LABA combination inhaler
The clinical quality measures as selected by the Clinical Management subcommittee for 2016 for the adult population are:
 For 2016 the Clinical Integration Program is moving its clinical quality measures from the Verisk Healthcare Quality and Risk Measures to the National Committee for Quality Assurance HEDIS based measures.
For 2016 the Clinical Integration Program is moving its clinical quality measures from the Verisk Healthcare Quality and Risk Measures to the National Committee for Quality Assurance HEDIS based measures.
Not available 100/6mcg 2 BD formoterol (Fostair MDI) 100/6mcg 33
 COMMISSIONING POLICY RECOMMENDATION TREATMENT ADVISORY GROUP FLUTICASONE FUROATE/VILANTEROL COMBINATION INHALER - ASTHMA Policy agreed by Vale of York CCG (date) Drug, Treatment, Device name Fluticasone
COMMISSIONING POLICY RECOMMENDATION TREATMENT ADVISORY GROUP FLUTICASONE FUROATE/VILANTEROL COMBINATION INHALER - ASTHMA Policy agreed by Vale of York CCG (date) Drug, Treatment, Device name Fluticasone
AEROSOL. Indication: /formoterol to. older with pressurized. two inhalations
 CEDAC FINAL RECOMMENDATION MOMETASONE FUROATE/ FORMOTEROL FUMARATE DIHYDRATE INHALATION AEROSOL (Zenhale Merck Canada Inc.) Indication: Asthma Maintenanc ce (Adults, Children 12 Years or Older) Recommendation:
CEDAC FINAL RECOMMENDATION MOMETASONE FUROATE/ FORMOTEROL FUMARATE DIHYDRATE INHALATION AEROSOL (Zenhale Merck Canada Inc.) Indication: Asthma Maintenanc ce (Adults, Children 12 Years or Older) Recommendation:
Three s Company - The role of triple therapy in chronic obstructive pulmonary disease (COPD)
 Three s Company - The role of triple therapy in chronic obstructive pulmonary disease (COPD) Zahava Picado, PharmD PGY1 Pharmacy Practice Resident Central Texas Veterans Healthcare System Temple, TX October
Three s Company - The role of triple therapy in chronic obstructive pulmonary disease (COPD) Zahava Picado, PharmD PGY1 Pharmacy Practice Resident Central Texas Veterans Healthcare System Temple, TX October
Surveillance report Published: 6 April 2016 nice.org.uk. NICE All rights reserved.
 Surveillance report 2016 Chronic obstructive pulmonary disease in over 16s: diagnosis and management (2010) NICE guideline CG101 Surveillance report Published: 6 April 2016 nice.org.uk NICE 2016. All rights
Surveillance report 2016 Chronic obstructive pulmonary disease in over 16s: diagnosis and management (2010) NICE guideline CG101 Surveillance report Published: 6 April 2016 nice.org.uk NICE 2016. All rights
Adjustment of Inhaled Controller Therapy of Asthma in the Yellow Zone, Based on the Inhaler Product Used in the Green Zone Age 16 Years and Older
 Adjustment of Inhaled Controller Therapy of Asthma in the Yellow Zone, Based on the Inhaler Product Used in the Green Zone Age 16 Years and Older The Canadian Thoracic Society and other international asthma
Adjustment of Inhaled Controller Therapy of Asthma in the Yellow Zone, Based on the Inhaler Product Used in the Green Zone Age 16 Years and Older The Canadian Thoracic Society and other international asthma
12/18/2017. Disclosures. Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing
 Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing Diana M. Sobieraj, PharmD, BCPS Assistant Professor University of Connecticut School
Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing Diana M. Sobieraj, PharmD, BCPS Assistant Professor University of Connecticut School
Estimated date of first patient enrolled Q III/IV Estimated date of last patient completed Q3 2015
 PROTOCOL SYNOPSIS A 26 week, randomized, double-blind, parallel-group, active controlled, multicenter, multinational safety study evaluating the risk of serious asthma-related events during treatment with
PROTOCOL SYNOPSIS A 26 week, randomized, double-blind, parallel-group, active controlled, multicenter, multinational safety study evaluating the risk of serious asthma-related events during treatment with
PROTOCOL FOR THE ASSESSMENT OF FDA REGULATORY POLICIES FOR LONG ACTING BETA 2 AGONISTS
 PROTOCOL FOR THE ASSESSMENT OF FDA REGULATORY POLICIES FOR LONG ACTING BETA 2 AGONISTS Prepared by: Melissa G. Butler, PharmD, MPH, PhD 1, Esther H. Zhou, MD, PhD 2, Fang Zhang, PhD 3, Mark Levenson, PhD
PROTOCOL FOR THE ASSESSMENT OF FDA REGULATORY POLICIES FOR LONG ACTING BETA 2 AGONISTS Prepared by: Melissa G. Butler, PharmD, MPH, PhD 1, Esther H. Zhou, MD, PhD 2, Fang Zhang, PhD 3, Mark Levenson, PhD
Comprehensive Research Plan: Inhaled corticosteroids + long-acting beta agonists (ICS+LABA) for the treatment of asthma
 Comprehensive Research Plan: Inhaled corticosteroids + long-acting beta agonists (ICS+LABA) for the treatment of asthma Pharmacoepidemiology Unit July 10, 2014 30 Bond Street, Toronto ON, M5B 1W8 www.odprn.ca
Comprehensive Research Plan: Inhaled corticosteroids + long-acting beta agonists (ICS+LABA) for the treatment of asthma Pharmacoepidemiology Unit July 10, 2014 30 Bond Street, Toronto ON, M5B 1W8 www.odprn.ca
Policy Evaluation: Low Dose Quetiapine Safety Edit
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University 500 Summer Street NE, E35, Salem, Oregon 97301 1079 Phone 503 947 5220 Fax 503
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University 500 Summer Street NE, E35, Salem, Oregon 97301 1079 Phone 503 947 5220 Fax 503
NHS Dumfries & Galloway Triple therapy in COPD patients over 16 years
 Title of Project: NHS Dumfries & Galloway Triple therapy in COPD patients over 16 years 1 Reason for the review Respiratory prescribing is long term and can be costly. Appropriate choice and use of inhaled
Title of Project: NHS Dumfries & Galloway Triple therapy in COPD patients over 16 years 1 Reason for the review Respiratory prescribing is long term and can be costly. Appropriate choice and use of inhaled
Abbreviated Class Review: Chronic Obstructive Pulmonary Disease (COPD)
 Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Executive summary: Month/Year of Review: February
Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Executive summary: Month/Year of Review: February
Tri-County Opioid Safety Coalition Data Brief December 2017 Clackamas, Multnomah, and Washington Counties
 Medicaid-Funded Alternative Treatment for Back Pain in the Tri-County Region Key Findings The percentage of members with a back pain diagnosis who received an alternative treatment increased from 29% in
Medicaid-Funded Alternative Treatment for Back Pain in the Tri-County Region Key Findings The percentage of members with a back pain diagnosis who received an alternative treatment increased from 29% in
QUANTITY LIMIT CRITERIA. BROVANA (arformoterol tartrate) SEREVENT DISKUS (salmeterol) STRIVERDI RESPIMAT (olodaterol)
 Carelirst. +.V Family of health care plans DRUG CLASS COMBINATIONS QUANTITY LIMIT CRITERIA LONG ACTING BETA2-ADRENERGIC AGONIST, ORAL INHALATION BRAND NAME (generic) LONG-ACTING BETA2-ADRENERGIC AGONISTS:
Carelirst. +.V Family of health care plans DRUG CLASS COMBINATIONS QUANTITY LIMIT CRITERIA LONG ACTING BETA2-ADRENERGIC AGONIST, ORAL INHALATION BRAND NAME (generic) LONG-ACTING BETA2-ADRENERGIC AGONISTS:
Up in FLAMES: Stable Chronic Obstructive Pulmonary Disease (COPD) Management. Colleen Sakon, PharmD BCPS September 27, 2018
 Up in FLAMES: Stable Chronic Obstructive Pulmonary Disease (COPD) Management Colleen Sakon, PharmD BCPS September 27, 2018 Disclosures I have no actual or potential conflicts of interest 2 Objectives Summarize
Up in FLAMES: Stable Chronic Obstructive Pulmonary Disease (COPD) Management Colleen Sakon, PharmD BCPS September 27, 2018 Disclosures I have no actual or potential conflicts of interest 2 Objectives Summarize
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: Fluticasone/Salmeterol (Advair Diskus, Advair HFA) Reference Number: CP.PMN.31 Effective Date: 10.01.18 Last Review Date: 07.13.18 Line of Business: Oregon Health Plan See Important Reminder
Clinical Policy: Fluticasone/Salmeterol (Advair Diskus, Advair HFA) Reference Number: CP.PMN.31 Effective Date: 10.01.18 Last Review Date: 07.13.18 Line of Business: Oregon Health Plan See Important Reminder
THE USE OF LONG-ACTING β 2 -AGONISTS AS MONOTHERAPY IN CHILDREN AND ADULTS
 THE USE OF LONG-ACTING β 2 -AGONISTS AS MONOTHERAPY IN CHILDREN AND ADULTS Pat G Camp 1,2, Tingting Zhang 3, M Anne Smith 3, Bruce C Carleton 3 1 UBC James Hogg Research Centre and the Institute for Heart
THE USE OF LONG-ACTING β 2 -AGONISTS AS MONOTHERAPY IN CHILDREN AND ADULTS Pat G Camp 1,2, Tingting Zhang 3, M Anne Smith 3, Bruce C Carleton 3 1 UBC James Hogg Research Centre and the Institute for Heart
Three s Company - The role of triple therapy in chronic obstructive pulmonary
 Three s Company - The role of triple therapy in chronic obstructive pulmonary disease (COPD) October 26 th, 2018 Zahava Picado, PharmD PGY1 Pharmacy Resident Central Texas Veterans Healthcare System Zahava.Picado@va.gov
Three s Company - The role of triple therapy in chronic obstructive pulmonary disease (COPD) October 26 th, 2018 Zahava Picado, PharmD PGY1 Pharmacy Resident Central Texas Veterans Healthcare System Zahava.Picado@va.gov
Orally Inhaled Corticosteroids to 2022
 Greystone Research Associates 1+603-595-4340 April 2015 Orally Inhaled Corticosteroids to 2022 Drugs, Devices, Markets and Forecasts Contents A Comprehensive Market Analysis Report Scope & Overview 2 Table
Greystone Research Associates 1+603-595-4340 April 2015 Orally Inhaled Corticosteroids to 2022 Drugs, Devices, Markets and Forecasts Contents A Comprehensive Market Analysis Report Scope & Overview 2 Table
Supplementary materials
 Supplementary materials Table S1 Patient comorbidities by diagnosis Total Asthma n (%) 5348 (148.4) COPD n (%) 4563 (143.4) ACOS n (%) 469 (197.9) No other comorbidities 95 (26.4) 614 (19.3) 27 (11.4)
Supplementary materials Table S1 Patient comorbidities by diagnosis Total Asthma n (%) 5348 (148.4) COPD n (%) 4563 (143.4) ACOS n (%) 469 (197.9) No other comorbidities 95 (26.4) 614 (19.3) 27 (11.4)
New Therapies for Asthma
 New Therapies for Asthma Tracy Bridges, MD Speaker Disclosure: Dr. Bridges participates in speaker bureaus for Teva, Genetech & Astra Zeneca. Objectives: Discuss the use of LAMA s for Asthma Detail the
New Therapies for Asthma Tracy Bridges, MD Speaker Disclosure: Dr. Bridges participates in speaker bureaus for Teva, Genetech & Astra Zeneca. Objectives: Discuss the use of LAMA s for Asthma Detail the
Optum Research Database Proportion of asthma patients with prescription fills for rescue and controller medications by year,
 ASTHMA CHART 1 Optum Research Database Proportion of asthma patients with prescription fills for rescue and controller medications by year, 215-216 7 6 5 4 3 2 1 1 Controller medication 1 Rescue medication
ASTHMA CHART 1 Optum Research Database Proportion of asthma patients with prescription fills for rescue and controller medications by year, 215-216 7 6 5 4 3 2 1 1 Controller medication 1 Rescue medication
October 2015 news bulletin
 October 2015 news bulletin Claims tip of the month billing medical injectables For single dose vials, providers should bill Amerigroup Washington, Inc. for the total amount of the drug contained in the
October 2015 news bulletin Claims tip of the month billing medical injectables For single dose vials, providers should bill Amerigroup Washington, Inc. for the total amount of the drug contained in the
Utilization of LABAs versus SABAs in Albanian Insured Outpatients with COPD during
 Utilization of LABAs versus SABAs in Albanian Insured Outpatients with COPD during 2008-2012 Doi:10.5901/mjss.2013.v4n9p573 Abstract Alida Sina PhD Student, Health Insurance Institute, Tirana, Albania
Utilization of LABAs versus SABAs in Albanian Insured Outpatients with COPD during 2008-2012 Doi:10.5901/mjss.2013.v4n9p573 Abstract Alida Sina PhD Student, Health Insurance Institute, Tirana, Albania
COPD: Treatment Update Property of Presenter. Not for Reproduction. Barry Make, MD Professor of Medicine National Jewish Health
 COPD: Treatment Update Barry Make, MD Professor of Medicine National Jewish Health Disclosures Advisory board, consultant, multi-center trial, research funding, Data Safety Monitoring Board (DSMB), or
COPD: Treatment Update Barry Make, MD Professor of Medicine National Jewish Health Disclosures Advisory board, consultant, multi-center trial, research funding, Data Safety Monitoring Board (DSMB), or
Reference ID:
 1 MEDICATION GUIDE 2 ADVAIR [ad vair] DISKUS 100/50 3 (fluticasone propionate 100 mcg and salmeterol 50 mcg inhalation powder) 4 ADVAIR DISKUS 250/50 5 (fluticasone propionate 250 mcg and salmeterol 50
1 MEDICATION GUIDE 2 ADVAIR [ad vair] DISKUS 100/50 3 (fluticasone propionate 100 mcg and salmeterol 50 mcg inhalation powder) 4 ADVAIR DISKUS 250/50 5 (fluticasone propionate 250 mcg and salmeterol 50
Overview of inhaled and nasal corticosteroids and haematoma
 Overview of inhaled and nasal corticosteroids and haematoma Introduction Inhaled glucocorticoids (ICS) are widely used to treat asthma and chronic obstructive pulmonary disease (COPD). Nasal glucocorticoids
Overview of inhaled and nasal corticosteroids and haematoma Introduction Inhaled glucocorticoids (ICS) are widely used to treat asthma and chronic obstructive pulmonary disease (COPD). Nasal glucocorticoids
Class Update: Asthma / COPD Medications
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
CLINICAL MEDICAL POLICY
 CLINICAL MEDICAL POLICY Policy Name: Xolair (omalizumab) Policy Number: MP-051-MD-DE Responsible Department(s): Medical Management; Clinical Pharmacy Provider Notice Date: 10/01/2017 Original Effective
CLINICAL MEDICAL POLICY Policy Name: Xolair (omalizumab) Policy Number: MP-051-MD-DE Responsible Department(s): Medical Management; Clinical Pharmacy Provider Notice Date: 10/01/2017 Original Effective
SHORT COMMUNICATION. Abstract. Kevin R. Murphy, 1 Tom Uryniak, 2 Ubaldo J. Martin 2 and James Zangrilli 2
 SHORT COMMUNICATION Drugs R D 212; 12 (1): 9-14 1179-691/12/1-9 ª 212 Murphy et al., publisher and licensee Adis Data Information BV. This is an open access article published under the terms of the Creative
SHORT COMMUNICATION Drugs R D 212; 12 (1): 9-14 1179-691/12/1-9 ª 212 Murphy et al., publisher and licensee Adis Data Information BV. This is an open access article published under the terms of the Creative
Combination Beta2-Agonist/Corticosteroid Inhalers
 Combination Beta2-Agonist/Corticosteroid Inhalers Policy Number: 5.01.572 Last Review: 7/2017 Origination: 6/2014 Next Review: 7/2018 LoB: ACA Policy Blue Cross and Blue Shield of Kansas City (Blue KC)
Combination Beta2-Agonist/Corticosteroid Inhalers Policy Number: 5.01.572 Last Review: 7/2017 Origination: 6/2014 Next Review: 7/2018 LoB: ACA Policy Blue Cross and Blue Shield of Kansas City (Blue KC)
Changes in Long-acting β-agonist Utilization After the FDA s 2010 Drug Safety Communication
 Changes in Long-acting β-agonist Utilization After the FDA s 2010 Drug Safety Communication Hartung, D. M., Middleton, L., Markwardt, S., Williamson, K., & Ketchum, K. (2015). Changes in Long-acting β-agonist
Changes in Long-acting β-agonist Utilization After the FDA s 2010 Drug Safety Communication Hartung, D. M., Middleton, L., Markwardt, S., Williamson, K., & Ketchum, K. (2015). Changes in Long-acting β-agonist
Drug prescriptions (Pharm) Exposure (36/48 months)
 ANNEX SECTION PART A - Study design: Figure 1 overview of the study design Drug prescriptions (Pharm) 2006-2010 Exposure (36/48 months) flexible time windows (e.g. 90 days) time index date (hospital discharge)
ANNEX SECTION PART A - Study design: Figure 1 overview of the study design Drug prescriptions (Pharm) 2006-2010 Exposure (36/48 months) flexible time windows (e.g. 90 days) time index date (hospital discharge)
XOLAIR (omalizumab) Prior Authorization
 MP9309 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes as shown below May only be prescribed by Allergy, Pulmonary, Immunology or Dermatology specialists
MP9309 Covered Service: Prior Authorization Required: Additional Information: Yes when meets criteria below Yes as shown below May only be prescribed by Allergy, Pulmonary, Immunology or Dermatology specialists
fluticasone furoate / vilanterol 92/22, 184/22 micrograms inhalation powder (Relvar Ellipta ) SMC No. (966/14) GlaxoSmithKline UK
 fluticasone furoate / vilanterol 92/22, 184/22 micrograms inhalation powder (Relvar Ellipta ) SMC No. (966/14) GlaxoSmithKline UK 09 May 2014 The Scottish Medicines Consortium (SMC) has completed its assessment
fluticasone furoate / vilanterol 92/22, 184/22 micrograms inhalation powder (Relvar Ellipta ) SMC No. (966/14) GlaxoSmithKline UK 09 May 2014 The Scottish Medicines Consortium (SMC) has completed its assessment
Dual-Controller Asthma Therapy: Rationale and Clinical Benefits
 B/1 Dual-Controller Asthma Therapy: Rationale and Clinical Benefits MODULE B The 1997 National Heart, Lung, and Blood Institute (NHLBI) Expert Panel guidelines on asthma management recommend a 4-step approach
B/1 Dual-Controller Asthma Therapy: Rationale and Clinical Benefits MODULE B The 1997 National Heart, Lung, and Blood Institute (NHLBI) Expert Panel guidelines on asthma management recommend a 4-step approach
Chronic obstructive pulmonary disease
 0 Chronic obstructive pulmonary disease Implementing NICE guidance June 2010 NICE clinical guideline 101 What this presentation covers Background Scope Key priorities for implementation Discussion Find
0 Chronic obstructive pulmonary disease Implementing NICE guidance June 2010 NICE clinical guideline 101 What this presentation covers Background Scope Key priorities for implementation Discussion Find
Dr Christopher Worsnop
 Dr Christopher Worsnop Respiratory & Sleep Physician Austin Hospital, Melbourne Supported by: Top Tips in Modern Asthma Management Dr Christopher Worsnop Rotorua GPCME Meeting June 2013 Speaker declaration
Dr Christopher Worsnop Respiratory & Sleep Physician Austin Hospital, Melbourne Supported by: Top Tips in Modern Asthma Management Dr Christopher Worsnop Rotorua GPCME Meeting June 2013 Speaker declaration
Pharmacy Medical Policy Asthma and Chronic Obstructive Pulmonary Disease Medication Management
 Pharmacy Medical Policy Asthma and Chronic Obstructive Pulmonary Disease Medication Management Table of Contents Policy: Commercial Information Pertaining to All Policies Endnotes Policy: Medicare References
Pharmacy Medical Policy Asthma and Chronic Obstructive Pulmonary Disease Medication Management Table of Contents Policy: Commercial Information Pertaining to All Policies Endnotes Policy: Medicare References
Combination Beta2-Agonist/Corticosteroid Inhalers Policy Number: Last Review: Origination: Next Review: Policy When Policy Topic is covered:
 Combina ation Beta2-Agonist/Corticosteroid Inhalers Policy Number: 5.01.572 Origination: 06/2014 Last Review: 07/2014 Next Review: 07/2015 Policy BCBSKC will provide coverage for the combination beta2-agonist/corticosteroid
Combina ation Beta2-Agonist/Corticosteroid Inhalers Policy Number: 5.01.572 Origination: 06/2014 Last Review: 07/2014 Next Review: 07/2015 Policy BCBSKC will provide coverage for the combination beta2-agonist/corticosteroid
Key features and changes to these four components of asthma care include:
 Guidelines for the Diagnosis and Management of Asthma in Adults Clinical Practice Guideline MedStar Health These guidelines are provided to assist physicians and other clinicians in making decisions regarding
Guidelines for the Diagnosis and Management of Asthma in Adults Clinical Practice Guideline MedStar Health These guidelines are provided to assist physicians and other clinicians in making decisions regarding
Pharmacotherapy for COPD
 10/3/2017 Topics to be covered Pharmacotherapy for chronic treatment Pharmacotherapy for COPD Dr. W C Yu 3rd September 2017 Commonly used drugs Guidelines for their use Inhaled corticosteroids (ICS) in
10/3/2017 Topics to be covered Pharmacotherapy for chronic treatment Pharmacotherapy for COPD Dr. W C Yu 3rd September 2017 Commonly used drugs Guidelines for their use Inhaled corticosteroids (ICS) in
Disclosure. Case. Objectives. Case Continued. Inhalers. Asthma: A GINA Update to the NAEPP 2007 Guidelines 1/20/2015
 Disclosure Asthma: A GINA Update to the NAEPP 2007 Guidelines Robert (RC) Hellinga, Pharm.D. PGY 1 Pharmacy Resident Wolfson Children s Hospital/Baptist Health I do not have a vested interest in or affiliation
Disclosure Asthma: A GINA Update to the NAEPP 2007 Guidelines Robert (RC) Hellinga, Pharm.D. PGY 1 Pharmacy Resident Wolfson Children s Hospital/Baptist Health I do not have a vested interest in or affiliation
A Partnership in Quality Reporting. Effectiveness of Care: Prevention and Screening
 A Partnership in Quality Reporting Effectiveness of Care: Prevention and Screening Codes to Identify Well-Child Visits Completed during First 15 Months of Life ICD-9-CM Diagnosis Codes 99381, 99382, 99391,
A Partnership in Quality Reporting Effectiveness of Care: Prevention and Screening Codes to Identify Well-Child Visits Completed during First 15 Months of Life ICD-9-CM Diagnosis Codes 99381, 99382, 99391,
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Fasenra) Reference Number: CP.PHAR.## Effective Date: 01.16.18 Last Review Date: 05.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
Clinical Policy: (Fasenra) Reference Number: CP.PHAR.## Effective Date: 01.16.18 Last Review Date: 05.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
September 2009 Based on Data collected for the DERP report of 11/2008
 Controller Medications for Asthma September 2009 Based on Data collected for the DERP report of 11/2008 Produced by: The Health Resources Commission Office for Oregon Health Policy & Research 1225 Ferry
Controller Medications for Asthma September 2009 Based on Data collected for the DERP report of 11/2008 Produced by: The Health Resources Commission Office for Oregon Health Policy & Research 1225 Ferry
Impact of a Comprehensive COPD Therapeutic Interchange Program on 30-Day Readmission Rates in Hospitalized Patients
 Impact of a Comprehensive COPD Therapeutic Interchange Program on 30-Day Readmission Rates in Hospitalized Patients Maren A. McGurran, PharmD, BCPS; Lisa M. Richter, PharmD, BCPS, BCCCP; Nathan D. Leedahl,
Impact of a Comprehensive COPD Therapeutic Interchange Program on 30-Day Readmission Rates in Hospitalized Patients Maren A. McGurran, PharmD, BCPS; Lisa M. Richter, PharmD, BCPS, BCCCP; Nathan D. Leedahl,
Clinical Policy: Mepolizumab (Nucala) Reference Number: ERX.SPA.214 Effective Date:
 Clinical Policy: (Nucala) Reference Number: ERX.SPA.214 Effective Date: 07.01.16 Last Review Date: 02.19 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Nucala) Reference Number: ERX.SPA.214 Effective Date: 07.01.16 Last Review Date: 02.19 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Drug Use Research & Management Program Phone Fax Month/Year of Review: July 2013
 Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Copyright 2012 Oregon State University. All Rights
Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Copyright 2012 Oregon State University. All Rights
Canadian Expert Drug Advisory Committee Final Recommendation Plain Language Version
 Canadian Expert Drug Advisory Committee Final Recommendation Plain Language Version MOMETASONE FUROATE/FORMOTEROL FUMARATE DIHYDRATE INHALATION AEROSOL (Zenhale Merck Canada Inc.) Indication: Asthma Maintenance
Canadian Expert Drug Advisory Committee Final Recommendation Plain Language Version MOMETASONE FUROATE/FORMOTEROL FUMARATE DIHYDRATE INHALATION AEROSOL (Zenhale Merck Canada Inc.) Indication: Asthma Maintenance
AIRDUO RESPICLICK (fluticasone-salmeterol) aerosol DULERA (mometasone furoate and formoterol fumarate dihydrate) aerosol
 DULERA (mometasone furoate and formoterol fumarate dihydrate) aerosol Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific
DULERA (mometasone furoate and formoterol fumarate dihydrate) aerosol Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific
UPDATE IN CHRONIC OBSTRUCTIVE PULMONARY DISEASE
 UPDATE IN CHRONIC OBSTRUCTIVE PULMONARY DISEASE Radhika Shah, MD Erlanger Health System University of Tennessee College of Medicine Chattanooga Respiratory, Critical Care, and Sleep medicine No disclosures
UPDATE IN CHRONIC OBSTRUCTIVE PULMONARY DISEASE Radhika Shah, MD Erlanger Health System University of Tennessee College of Medicine Chattanooga Respiratory, Critical Care, and Sleep medicine No disclosures
Diagnosis and Management of Asthma
 Supporting Evidence: Diagnosis and Management of Asthma The subdivision of this section is: Appendix B Tables Copyright 2016 by 1 Eleventh Edition/December 2016 Appendix B Asthma Summary Tables Class:
Supporting Evidence: Diagnosis and Management of Asthma The subdivision of this section is: Appendix B Tables Copyright 2016 by 1 Eleventh Edition/December 2016 Appendix B Asthma Summary Tables Class:
Emergency Department Visits for Behavioral Health Conditions in Harris County, Texas,
 Emergency Department Visits for Behavioral Health Conditions in Harris County, Texas, 2007-2008 Prepared by School of Public Health UT Health Patrick Courtney, MA August 2010 1 Table of Contents Executive
Emergency Department Visits for Behavioral Health Conditions in Harris County, Texas, 2007-2008 Prepared by School of Public Health UT Health Patrick Courtney, MA August 2010 1 Table of Contents Executive
Scottish Medicines Consortium
 Scottish Medicines Consortium montelukast 10mg tablets (Singulair ) No. (185/05) Merck, Sharp & Dohme Ltd (MSD) New indication: for asthmatic patients in whom montelukast is indicated in asthma, montelukast
Scottish Medicines Consortium montelukast 10mg tablets (Singulair ) No. (185/05) Merck, Sharp & Dohme Ltd (MSD) New indication: for asthmatic patients in whom montelukast is indicated in asthma, montelukast
Optimising asthma management in high risk patients
 C L I N I C A L AU D I T Optimising asthma management in high risk patients Valid to June 2022 bpac nz better medicine Audit focus This audit helps primary care health professionals identify patients with
C L I N I C A L AU D I T Optimising asthma management in high risk patients Valid to June 2022 bpac nz better medicine Audit focus This audit helps primary care health professionals identify patients with
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Nucala) Reference Number: CP.PHAR.200 Effective Date: 04.01.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at
Clinical Policy: (Nucala) Reference Number: CP.PHAR.200 Effective Date: 04.01.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at
Sponsor. Generic drug name. Trial indication(s) Protocol number. Protocol title. Clinical trial phase. Study Start/End Dates.
 Sponsor Novartis Generic drug name Fluticasone propionate Trial indication(s) Moderate-severe bronchial asthma Protocol number CQAE397A2202 Protocol title A randomized open label study to assess the utility
Sponsor Novartis Generic drug name Fluticasone propionate Trial indication(s) Moderate-severe bronchial asthma Protocol number CQAE397A2202 Protocol title A randomized open label study to assess the utility
Cost-Motivated Treatment Changes in Commercial Claims:
 Cost-Motivated Treatment Changes in Commercial Claims: Implications for Non- Medical Switching August 2017 THE MORAN COMPANY 1 Cost-Motivated Treatment Changes in Commercial Claims: Implications for Non-Medical
Cost-Motivated Treatment Changes in Commercial Claims: Implications for Non- Medical Switching August 2017 THE MORAN COMPANY 1 Cost-Motivated Treatment Changes in Commercial Claims: Implications for Non-Medical
GINA. At-A-Glance Asthma Management Reference. for adults, adolescents and children 6 11 years. Updated 2017
 GINA At-A-Glance Asthma Management Reference for adults, adolescents and children 6 11 years Updated 2017 This resource should be used in conjunction with the Global Strategy for Asthma Management and
GINA At-A-Glance Asthma Management Reference for adults, adolescents and children 6 11 years Updated 2017 This resource should be used in conjunction with the Global Strategy for Asthma Management and
Executive Summary. Classes Under Review: Analysis:
 Outcome Report on MHQAC Edits Classes Under Review: Analysis: Atypical Antipsychotics, SSRIs, SNRIs, Antianxietals, Hypnotics, and Drugs to Treat ADD/ADHD Utilization and Cost Trends: Pre and Post Implementation
Outcome Report on MHQAC Edits Classes Under Review: Analysis: Atypical Antipsychotics, SSRIs, SNRIs, Antianxietals, Hypnotics, and Drugs to Treat ADD/ADHD Utilization and Cost Trends: Pre and Post Implementation
