Asthma and COPD Drug Discoveries:
|
|
|
- Tracey Moody
- 6 years ago
- Views:
Transcription
1 Asthma and COPD Drug Discoveries: What the future holds World Leaders in Health Industry Analysis
2 Asthma and COPD Drug Discoveries: what the future holds ISBN: Copyright 2006 Espicom Business Intelligence All rights reserved. No part of this publication may be reproduced or used in any form or by any means graphic, electronic or mechanical, including photocopying, recording, taping or storage in information retrieval systems without the express permission of the publisher. Every care has been taken to ensure that the information contained in this report is correct. The publisher accepts no liability for decisions made on the basis of information contained herein. British Library Cataloguing in Publication Data. A catalogue record for this report is available from the British Library.
3 Asthma and COPD Drug Discoveries: what the future holds Published by Espicom Business Intelligence, August
4 This page left intentionally blank
5 Contents Foreword... 1 Executive Summary... 3 What are Asthma and COPD?... 5 Morbidity and Mortality...5 Diagnosis and Treatment... 7 Asthma Management...8 COPD Management...9 Pharmacotherapy Current Market Leaders Drugs in the Pipeline Competitor Ratio Analysis Short-Acting Beta-2 Agonists Xopenex (levalbuterol) Sales Forecast...20 Patent Challenges...21 Clinical Trials...21 Xopenex versus racemic albuterol in COPD...21 Xopenex HFA study...21 Xopenex in the emergency room...22 Inhaled Corticosteroids Flixotide/Flovent (fluticasone propionate) Sales Forecasts...24 Clinical Trials...25 Pulmicort (budesonide) Sales Forecast...28 Patent Challenge...29 Clinical Trials...29 START budesonide during pregnancy...29 START budesonide in children with mild persistent asthma...30 START safety outcome data...30 EUROSCOP...31 Long-term data from early intervention study in Finland...31 Asmanex (mometasone furoate) Sales Forecasts and Assumptions...34 Competitor Ratio Analysis...34 Novelty/rationale for mechanism of action...34 Proof of concept/clinical data...35 August 2006 i
6 Management/clinical expertise Competition within marketplace Risks associated with drug development in therapeutic class Clinical Trials Patients previously maintained on bronchodilators alone Patients previously maintained on inhaled corticosteroids Patients previously maintained on oral corticosteroids Alvesco (ciclesonide) Sales Forecast and Assumptions Competitor Ratio Analysis Novelty/rationale for mechanism of action Proof of concept/clinical data Management/clinical expertise Competition within marketplace Risks associated with drug development in therapeutic class Clinical trials Ciclesonide versus fluticasone propionate Low incidence of oropharyngeal side effects Ciclesonide versus budesonide and fluticasone Intranasal formulation Ciclesonide versus budesonide Muscarinic Antagonists Spiriva (tiotropium) Sales Forecast and Assumptions Competitor Ratio Analysis Novelty/rationale for mechanism of action Proof of concept/clinical data Management/clinical expertise Competition within marketplace Risks associated with drug development in therapeutic class Clinical Trials Pooled Studies Study on COPD patients in France Spiriva+formoterol versus salmeterol+fluticasone propionate UPLIFT TIPHON Combination Therapy Seretide/Advair (salmeterol+fluticasone propionate) Sales Forecasts Patent Challenge ii August 2006
7 Safety Issues...52 Clinical Trials: COPD...53 TORCH...53 TRISTAN...53 Clinical Trials: Asthma...55 Biopsy Study in Moderate Asthma...55 EXCEL...55 GOAL...56 Advair versus montelukast...56 Symbicort (budesonide+formoterol) Sales Forecast...59 European patent challenge...60 Clinical Trials: COPD...60 Calverly et al Clinical Trials: Asthma...61 SUND...61 STAY...62 Head to head study: Symbicort Turbuhaler versus Seretide (salmeterol+fluticasone)...62 Clinical Trials: Asthma+COPD...63 ASSURE...63 Leukotriene Receptor Antagonists Singulair (montelukast) Sales Forecast...65 Clinical Trials...66 PREVIA...66 Anti-Immunoglobulin E Therapy Xolair (omalizumab) Sales Forecast and Assumptions...68 Competitor Ratio Analysis...69 Novelty/rationale for mechanism of action...69 Proof of concept/clinical data...69 Management/clinical expertise...69 Competition within marketplace...70 Risks associated with drug development in therapeutic class...70 Clinical Trials...70 Long-term data...70 Pooled study data...71 Drugs in Development PDE-IV Inhibitors Ariflo (cilomilast) August 2006 iii
8 Sales Forecasts and Assumptions Competitor Ratio Analysis Novelty/rationale for mechanism of action Proof of concept/clinical data Management/clinical expertise Competition within marketplace Risks associated with drug development in therapeutic class Clinical trials Daxas (roflumilast) Sales Forecasts and Assumptions Competitor Ratio Analysis Novelty/rationale for mechanism of action Proof of concept/clinical data Management/clinical expertise Competition within marketplace Risks associated with drug development in therapeutic class Clinical Trials Efficacy & safety study in COPD RATIO/M RECORD Other studies Long-Acting Beta Agonists Arformoterol Sales Forecasts and Assumptions Competitor Ratio Analysis Novelty/rationale for mechanism of action Proof of concept/clinical data Management/clinical expertise Competition within marketplace Risks associated with drug development in therapeutic class Clinical Trials Indacaterol Sales Forecast and Assumptions Competitor Ratio Analysis Novelty/rationale for mechanism of action Proof of concept/clinical data Management/clinical expertise Competition within marketplace Risks associated with drug development in therapeutic class Clinical Trials iv August 2006
9 GSK Sales Forecasts and Assumptions...89 Competitor Ratio Analysis...90 Novelty/rationale for mechanism of action...90 Proof of concept/clinical data...90 Management/clinical expertise...90 Competition within marketplace...91 Risks associated with drug development in therapeutic class...91 Clinical Trials...91 Long-Acting Muscarinic Antagonists NVA Sales Forecast and Assumptions...92 Competitor Ratio Analysis...93 Novelty/rationale for mechanism of action...93 Proof of concept/clinical data...93 Management/clinical expertise...93 Competition within marketplace...94 Risks associated with drug development in therapeutic class...94 Clinical Trials...94 SOURCES Espicom Sources Others August 2006 v
10 List of Tables Deaths from COPD and Asthma in Major Markets, Deaths from COPD and Asthma in the USA, GINA Stepwise Approach to Asthma Medication...8 GOLD Therapy Recommendations for COPD Leading Asthma and COPD Therapies by Sales Value, (US$ Million) US Patent Expiry for Major Asthma and COPD Therapies Launched Products in Development Novel Drugs in Development Competitor Ratio Analysis Summary Xopenex Sales, (US$ Million) Flixotide/Flovent Sales, ( & US$ Million) Pulmicort Sales, (US$ Million) Spiriva Sales, (Euros & US$ Million) Advair/Seretide Sales, ( & US$ Million) Symbicort Sales, (US$ Million) Singulair Sales, (US$ Million) Xolair Sales, (US$ Million) Xolair Projected Sales, (US$ Million) List of Figures Novel Drugs in Clinical Development by Activity Xopenex Projected Sales, (US$ Million) Flixotide/Flovent Projected Sales, (US$ Million) Pulmicort Projected Sales, (US$ Million) Asmanex Twisthaler Projected Sales, (US$ Million) Alvesco Projected Sales, (US$ Million) Spiriva Projected Sales, (US$ Million) Seretide/Advair Projected Sales, (US$ Million) Symbicort Projected Sales, (US$ Million) Singulair Projected Sales, (US$ Million) Ariflo Projected Sales, (US$ Million) Daxas Projected Sales, (US$ Million) Arformoterol Projected Sales, (US$ Million) vi August 2006
11 Indacaterol Projected Sales, (US$ Million) GSK Projected Sales, (US$ Million) NVA237 Projected Sales, (US$ Million) August 2006 vii
II: Moderate Worsening airflow limitations Dyspnea on exertion, cough, and sputum production; patient usually seeks medical
 Table 3.1. Classification of COPD Severity Stage Pulmonary Function Test Findings Symptoms I: Mild Mild airflow limitations +/ Chronic cough and sputum production; patient unaware of abnormal FEV 1 80%
Table 3.1. Classification of COPD Severity Stage Pulmonary Function Test Findings Symptoms I: Mild Mild airflow limitations +/ Chronic cough and sputum production; patient unaware of abnormal FEV 1 80%
A Visual Approach to Simplifying Respiratory Drug Regimens
 A Visual Approach to Simplifying Respiratory Drug Regimens Stephanie Cheng, PharmD, MPH, BCGP 3 Main Categories Inhaled Respiratory Drugs Binds to beta-2 receptors Relaxation of smooth muscles in the lung
A Visual Approach to Simplifying Respiratory Drug Regimens Stephanie Cheng, PharmD, MPH, BCGP 3 Main Categories Inhaled Respiratory Drugs Binds to beta-2 receptors Relaxation of smooth muscles in the lung
Inhaled bronchodilators relax constricted airways and treat the noisy part of asthma: coughing, wheezing, choking and shortness of breath.
 Inhaled bronchodilators relax constricted airways and treat the noisy part of asthma: coughing, wheezing, choking and shortness of breath. AccuNeb inhalation 0.021% solution: 0.63mg/3mL 3-4 times solution
Inhaled bronchodilators relax constricted airways and treat the noisy part of asthma: coughing, wheezing, choking and shortness of breath. AccuNeb inhalation 0.021% solution: 0.63mg/3mL 3-4 times solution
Diagnosis and Management of Asthma
 Supporting Evidence: Diagnosis and Management of Asthma The subdivision of this section is: Appendix B Tables Copyright 2016 by 1 Eleventh Edition/December 2016 Appendix B Asthma Summary Tables Class:
Supporting Evidence: Diagnosis and Management of Asthma The subdivision of this section is: Appendix B Tables Copyright 2016 by 1 Eleventh Edition/December 2016 Appendix B Asthma Summary Tables Class:
Drug Class Monograph
 Drug Class Monograph Class: Inhaled Corticosteroids Drugs: Aerospan (flunisolide), Advair Diskus, Advair HFA (fluticasone/salmeterol), Alvesco (ciclesonide), Arnuity Ellipta (fluticasone furoate), Asmanex
Drug Class Monograph Class: Inhaled Corticosteroids Drugs: Aerospan (flunisolide), Advair Diskus, Advair HFA (fluticasone/salmeterol), Alvesco (ciclesonide), Arnuity Ellipta (fluticasone furoate), Asmanex
Select Inhaled Respiratory Agents
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Fasenra) Reference Number: CP.PHAR.## Effective Date: 01.16.18 Last Review Date: 05.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
Clinical Policy: (Fasenra) Reference Number: CP.PHAR.## Effective Date: 01.16.18 Last Review Date: 05.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
COPD Medications Coverage Summary Non-Insured Health Benefits Coverage SABA Bricanyl turbuhaler Yes Yes
 COPD Medications Coverage Summary Drug Non-Insured Health Benefits Coverage SABA Bricanyl turbuhaler Yes Yes Ventolin MDI + generics Yes Yes Ventolin Diskus NO NO Yukon Pharmacare/Chronic Disease Program
COPD Medications Coverage Summary Drug Non-Insured Health Benefits Coverage SABA Bricanyl turbuhaler Yes Yes Ventolin MDI + generics Yes Yes Ventolin Diskus NO NO Yukon Pharmacare/Chronic Disease Program
reslizumab (Cinqair )
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
benralizumab (Fasenra )
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
APPENDIX 1 Printable point-of-care tables Asthma Action Plan Yellow Zone Formulation Table Region: Europe
 APPENDIX 1 Printable point-of-care tables Asthma Action Plan Yellow Zone Formulation Table Region: Europe Instructions: Print on 8.5 x14 (216 x 279 mm) paper (Legal size) Medication in Green Zone Change
APPENDIX 1 Printable point-of-care tables Asthma Action Plan Yellow Zone Formulation Table Region: Europe Instructions: Print on 8.5 x14 (216 x 279 mm) paper (Legal size) Medication in Green Zone Change
Asthma medications: Know your options - MayoClinic.com. Asthma medications: Know your options
 MayoClinic.com reprints This single copy is for your personal, noncommercial use only. For permission to reprint multiple copies or to order presentation-ready copies for distribution, use the reprints
MayoClinic.com reprints This single copy is for your personal, noncommercial use only. For permission to reprint multiple copies or to order presentation-ready copies for distribution, use the reprints
12/18/2017. Disclosures. Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing
 Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing Diana M. Sobieraj, PharmD, BCPS Assistant Professor University of Connecticut School
Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing Diana M. Sobieraj, PharmD, BCPS Assistant Professor University of Connecticut School
Allergies and Asthma 5/21/2013. Objectives. Allergic Rhinitis (AR): Risk Factor for ASTHMA. Rhinitis and Asthma
 Allergies and Asthma Presented By: Dr. Fadwa Gillanders, Pharm.D Clinical Pharmacy Specialist May 2013 Objectives Understand the relationship between asthma and allergic rhinitis Understand what is going
Allergies and Asthma Presented By: Dr. Fadwa Gillanders, Pharm.D Clinical Pharmacy Specialist May 2013 Objectives Understand the relationship between asthma and allergic rhinitis Understand what is going
A Visual Approach to Simplifying Respiratory Drug Regimens
 Adverse Effects of Inhaled Medications A Visual Approach to Simplifying Respiratory Drug Regimens Stephanie Cheng, PharmD, MPH, BCGP June 28, 2017 Drug Category Beta 2 agonists antagonists Adverse Effects
Adverse Effects of Inhaled Medications A Visual Approach to Simplifying Respiratory Drug Regimens Stephanie Cheng, PharmD, MPH, BCGP June 28, 2017 Drug Category Beta 2 agonists antagonists Adverse Effects
A Visual Approach to Simplifying Respiratory Drug Regimens
 A Visual Approach to Simplifying Respiratory Drug Regimens Stephanie Cheng, PharmD, MPH, BCGP October 23, 2017 Learning Objectives Be able to list at least 3 major adverse effects of inhaled medications
A Visual Approach to Simplifying Respiratory Drug Regimens Stephanie Cheng, PharmD, MPH, BCGP October 23, 2017 Learning Objectives Be able to list at least 3 major adverse effects of inhaled medications
ANTINEOPLASTIC DRUGS CHAPTER 21. Antineoplastic drugs - designed to treat malignancies, now also used to treat diseases with inflammatory component
 ANTINEOPLASTIC DRUGS CHAPTER 21 Antineoplastic drugs - designed to treat malignancies, now also used to treat diseases with inflammatory component Tx of malignancies Antineoplastic drugs: methotrexate
ANTINEOPLASTIC DRUGS CHAPTER 21 Antineoplastic drugs - designed to treat malignancies, now also used to treat diseases with inflammatory component Tx of malignancies Antineoplastic drugs: methotrexate
Nucala (mepolizumab injection for subcutaneous use)
 Nucala (mepolizumab injection for subcutaneous use) Policy Number: 5.01.612 Last Review: 01/2018 Origination: 02/2016 Next Review: 02/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will
Nucala (mepolizumab injection for subcutaneous use) Policy Number: 5.01.612 Last Review: 01/2018 Origination: 02/2016 Next Review: 02/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will
Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing
 Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing Diana M. Sobieraj, PharmD, BCPS Assistant Professor University of Connecticut School
Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing Diana M. Sobieraj, PharmD, BCPS Assistant Professor University of Connecticut School
TRELEGY ELLIPTA (fluticasone-umeclidinium-vilanterol) aerosol powder
 TRELEGY ELLIPTA (fluticasone-umeclidinium-vilanterol) aerosol powder Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific
TRELEGY ELLIPTA (fluticasone-umeclidinium-vilanterol) aerosol powder Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific
MEDICAL COVERAGE GUIDELINES ORIGINAL EFFECTIVE DATE: 07/05/18 SECTION: DRUGS LAST REVIEW DATE: LAST CRITERIA REVISION DATE: ARCHIVE DATE:
 CINQAIR (reslizumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
CINQAIR (reslizumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
Using Inhaled Corticosteroids as Needed for Asthma: giving patients relief or leaving them breathless?
 Using Inhaled Corticosteroids as Needed for Asthma: giving patients relief or leaving them breathless? Lindsay Thomas, Pharm.D. PGY2 Ambulatory Care Resident Department of Pharmacotherapy and Pharmacy
Using Inhaled Corticosteroids as Needed for Asthma: giving patients relief or leaving them breathless? Lindsay Thomas, Pharm.D. PGY2 Ambulatory Care Resident Department of Pharmacotherapy and Pharmacy
Adjustment of Inhaled Controller Therapy of Asthma in the Yellow Zone, Based on the Inhaler Product Used in the Green Zone Age 16 Years and Older
 Adjustment of Inhaled Controller Therapy of Asthma in the Yellow Zone, Based on the Inhaler Product Used in the Green Zone Age 16 Years and Older The Canadian Thoracic Society and other international asthma
Adjustment of Inhaled Controller Therapy of Asthma in the Yellow Zone, Based on the Inhaler Product Used in the Green Zone Age 16 Years and Older The Canadian Thoracic Society and other international asthma
End Stage COPD Guidance Document
 End Stage COPD Guidance Document Suggested Guidelines for the Determination of Hospice Eligibility A patient with severe chronic pulmonary disease that meets the following criteria may be eligible for
End Stage COPD Guidance Document Suggested Guidelines for the Determination of Hospice Eligibility A patient with severe chronic pulmonary disease that meets the following criteria may be eligible for
Asthma/COPD Update with Inhaler Workshop
 Asthma/COPD Update with Inhaler Workshop October 8, 2017 Nathan Samsa, DO, Pharm D, RPh, FACOI None Disclosures Agenda Asthma Updates COPD Updates Inhaler Workshop Medication Acronyms SABA: Short acting
Asthma/COPD Update with Inhaler Workshop October 8, 2017 Nathan Samsa, DO, Pharm D, RPh, FACOI None Disclosures Agenda Asthma Updates COPD Updates Inhaler Workshop Medication Acronyms SABA: Short acting
First to Market or 505 (b)2 CMC Considerations IPAC-RS/UF Orlando Inhalation Conference Orlando, Florida
 First to Market or 505 (b)2 CMC Considerations IPAC-RS/UF Orlando Inhalation Conference Orlando, Florida Prasad Peri, Ph.D., Branch Chief, ONDQA, FDA March 19, 2014 1 Topics for discussion Introduction
First to Market or 505 (b)2 CMC Considerations IPAC-RS/UF Orlando Inhalation Conference Orlando, Florida Prasad Peri, Ph.D., Branch Chief, ONDQA, FDA March 19, 2014 1 Topics for discussion Introduction
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Nucala) Reference Number: CP.PHAR.200 Effective Date: 04.01.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at
Clinical Policy: (Nucala) Reference Number: CP.PHAR.200 Effective Date: 04.01.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder at
CLINICAL MEDICAL POLICY
 CLINICAL MEDICAL POLICY Policy Name: Xolair (omalizumab) Policy Number: MP-051-MD-DE Responsible Department(s): Medical Management; Clinical Pharmacy Provider Notice Date: 10/01/2017 Original Effective
CLINICAL MEDICAL POLICY Policy Name: Xolair (omalizumab) Policy Number: MP-051-MD-DE Responsible Department(s): Medical Management; Clinical Pharmacy Provider Notice Date: 10/01/2017 Original Effective
Drug Effectiveness Review Project Summary Report
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
FASENRA (benralizumab)
 FASENRA (benralizumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
FASENRA (benralizumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
Key features and changes to these four components of asthma care include:
 Guidelines for the Diagnosis and Management of Asthma in Adults Clinical Practice Guideline MedStar Health These guidelines are provided to assist physicians and other clinicians in making decisions regarding
Guidelines for the Diagnosis and Management of Asthma in Adults Clinical Practice Guideline MedStar Health These guidelines are provided to assist physicians and other clinicians in making decisions regarding
Cinqair (reslizumab injection for intravenous use)
 Cinqair (reslizumab injection for intravenous use) Policy Number: 5.02.522 Last Review: 04/2018 Origination: 04/2016 Next Review: 04/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will
Cinqair (reslizumab injection for intravenous use) Policy Number: 5.02.522 Last Review: 04/2018 Origination: 04/2016 Next Review: 04/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will
Orally Inhaled Corticosteroids to 2022
 Greystone Research Associates 1+603-595-4340 April 2015 Orally Inhaled Corticosteroids to 2022 Drugs, Devices, Markets and Forecasts Contents A Comprehensive Market Analysis Report Scope & Overview 2 Table
Greystone Research Associates 1+603-595-4340 April 2015 Orally Inhaled Corticosteroids to 2022 Drugs, Devices, Markets and Forecasts Contents A Comprehensive Market Analysis Report Scope & Overview 2 Table
Inhaled Corticosteroids Drug Class Prior Authorization Protocol
 Inhaled Corticosteroids Drug Class Prior Authorization Protocol Line of Business: Medi-Cal P&T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has been developed through review
Inhaled Corticosteroids Drug Class Prior Authorization Protocol Line of Business: Medi-Cal P&T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has been developed through review
Foundations of Pharmacology
 Pharmacologic Management of Asthma Objectives: 1. Review the physiological basis for asthma therapy 2. Discuss the differences between SABA and LABA 3. Discuss the role of inhaled and oral systemic corticosteroids
Pharmacologic Management of Asthma Objectives: 1. Review the physiological basis for asthma therapy 2. Discuss the differences between SABA and LABA 3. Discuss the role of inhaled and oral systemic corticosteroids
Inhaled Corticosteroids Drug Class Prior Authorization Protocol
 Inhaled Corticosteroids Drug Class Prior Authorization Protocol Line of Business: Medicaid P&T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has been developed through review
Inhaled Corticosteroids Drug Class Prior Authorization Protocol Line of Business: Medicaid P&T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has been developed through review
Asthma. Definition. Symptoms
 Asthma Definition Asthma is a condition in which your airways narrow and swell and produce extra mucus. This can make breathing difficult and trigger coughing, wheezing and shortness of breath. For some
Asthma Definition Asthma is a condition in which your airways narrow and swell and produce extra mucus. This can make breathing difficult and trigger coughing, wheezing and shortness of breath. For some
Ferris State University College of Pharmacy MPA CE Symposium 2016 Paul Thill, PharmD, BCPS
 Ferris State University College of Pharmacy MPA CE Symposium 2016 Paul Thill, PharmD, BCPS Objectives Categorize the new asthma and COPD inhalers in to existing or newly created categories Discuss the
Ferris State University College of Pharmacy MPA CE Symposium 2016 Paul Thill, PharmD, BCPS Objectives Categorize the new asthma and COPD inhalers in to existing or newly created categories Discuss the
MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY: Asthma/COPD P&T DATE 12/14/2017 CLASS:
 MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY: Asthma/COPD P&T DATE 12/14/2017 CLASS: LOB: Respiratory Disorders Medi-Cal REVIEW HISTORY (MONTH/YEAR) 12/17,12/16, 5/15,
MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY: Asthma/COPD P&T DATE 12/14/2017 CLASS: LOB: Respiratory Disorders Medi-Cal REVIEW HISTORY (MONTH/YEAR) 12/17,12/16, 5/15,
Impact of a Comprehensive COPD Therapeutic Interchange Program on 30-Day Readmission Rates in Hospitalized Patients
 Impact of a Comprehensive COPD Therapeutic Interchange Program on 30-Day Readmission Rates in Hospitalized Patients Maren A. McGurran, PharmD, BCPS; Lisa M. Richter, PharmD, BCPS, BCCCP; Nathan D. Leedahl,
Impact of a Comprehensive COPD Therapeutic Interchange Program on 30-Day Readmission Rates in Hospitalized Patients Maren A. McGurran, PharmD, BCPS; Lisa M. Richter, PharmD, BCPS, BCCCP; Nathan D. Leedahl,
AIRDUO RESPICLICK (fluticasone-salmeterol) aerosol DULERA (mometasone furoate and formoterol fumarate dihydrate) aerosol
 DULERA (mometasone furoate and formoterol fumarate dihydrate) aerosol Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific
DULERA (mometasone furoate and formoterol fumarate dihydrate) aerosol Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific
Stepping down asthma treatment guidelines
 Stepping down asthma treatment guidelines The potential for inhaled corticosteroids (ICS) to cause dose-related side-effects has led to asthma management guidelines recommending a dose reduction once asthma
Stepping down asthma treatment guidelines The potential for inhaled corticosteroids (ICS) to cause dose-related side-effects has led to asthma management guidelines recommending a dose reduction once asthma
COPD and Asthma Devices Market by Inhalers Type (Drug powder, Metered Dose, Soft Mist), Nebulizers (Compressor, Ultrasonic, Mesh) - Global
 COPD and Asthma Devices Market by Inhalers Type (Drug powder, Metered Dose, Soft Mist), Nebulizers (Compressor, Ultrasonic, Mesh) - Global Opportunity Analysis and Industry Forecasts, 2015-2022 COPD and
COPD and Asthma Devices Market by Inhalers Type (Drug powder, Metered Dose, Soft Mist), Nebulizers (Compressor, Ultrasonic, Mesh) - Global Opportunity Analysis and Industry Forecasts, 2015-2022 COPD and
Asthma COPD Update 2018
 Asthma COPD Update 2018 Roger Hefflinger, Pharm.D. Clinical Associate Professor ISU COP Clinical Teaching Pharmacist Family Medicine Residency of Idaho In support of improving patient care, Idaho State
Asthma COPD Update 2018 Roger Hefflinger, Pharm.D. Clinical Associate Professor ISU COP Clinical Teaching Pharmacist Family Medicine Residency of Idaho In support of improving patient care, Idaho State
Asthma/COPD Update with Inhaler Workshop
 Asthma/COPD Update with Inhaler Workshop October 8, 2017 Nathan Samsa, DO, Pharm D, RPh, FACOI None Disclosures Agenda Asthma Updates COPD Updates Inhaler Workshop Asthma Updates Asthma Updates SMART Trial
Asthma/COPD Update with Inhaler Workshop October 8, 2017 Nathan Samsa, DO, Pharm D, RPh, FACOI None Disclosures Agenda Asthma Updates COPD Updates Inhaler Workshop Asthma Updates Asthma Updates SMART Trial
Three s Company - The role of triple therapy in chronic obstructive pulmonary
 Three s Company - The role of triple therapy in chronic obstructive pulmonary disease (COPD) October 26 th, 2018 Zahava Picado, PharmD PGY1 Pharmacy Resident Central Texas Veterans Healthcare System Zahava.Picado@va.gov
Three s Company - The role of triple therapy in chronic obstructive pulmonary disease (COPD) October 26 th, 2018 Zahava Picado, PharmD PGY1 Pharmacy Resident Central Texas Veterans Healthcare System Zahava.Picado@va.gov
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: Fluticasone/Salmeterol (Advair Diskus, Advair HFA) Reference Number: CP.PMN.31 Effective Date: 10.01.18 Last Review Date: 07.13.18 Line of Business: Oregon Health Plan See Important Reminder
Clinical Policy: Fluticasone/Salmeterol (Advair Diskus, Advair HFA) Reference Number: CP.PMN.31 Effective Date: 10.01.18 Last Review Date: 07.13.18 Line of Business: Oregon Health Plan See Important Reminder
Clinical Policy: Omalizumab (Xolair) Reference Number: ERX.SPA.141 Effective Date: Last Review Date: 08.17
 Clinical Policy: (Xolair) Reference Number: ERX.SPA.141 Effective Date: 03.01.14 Last Review Date: 08.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Xolair) Reference Number: ERX.SPA.141 Effective Date: 03.01.14 Last Review Date: 08.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Up in FLAMES: Stable Chronic Obstructive Pulmonary Disease (COPD) Management. Colleen Sakon, PharmD BCPS September 27, 2018
 Up in FLAMES: Stable Chronic Obstructive Pulmonary Disease (COPD) Management Colleen Sakon, PharmD BCPS September 27, 2018 Disclosures I have no actual or potential conflicts of interest 2 Objectives Summarize
Up in FLAMES: Stable Chronic Obstructive Pulmonary Disease (COPD) Management Colleen Sakon, PharmD BCPS September 27, 2018 Disclosures I have no actual or potential conflicts of interest 2 Objectives Summarize
Clinical Policy: Dupilumab (Dupixent) Reference Number: ERX.SPA.49 Effective Date:
 Clinical Policy: (Dupixent) Reference Number: ERX.SPA.49 Effective Date: 06.01.17 Last Review Date: 02.19 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Dupixent) Reference Number: ERX.SPA.49 Effective Date: 06.01.17 Last Review Date: 02.19 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
continuing education for pharmacists
 continuing education for pharmacists Volume XXXIV, No. 12 Asthma: Approaches to Treatment and New Therapies Cortney M. Mospan, PharmD, BCACP, CGP, Assistant Professor of Pharmacy, Wingate University School
continuing education for pharmacists Volume XXXIV, No. 12 Asthma: Approaches to Treatment and New Therapies Cortney M. Mospan, PharmD, BCACP, CGP, Assistant Professor of Pharmacy, Wingate University School
Global Strategy for the Diagnosis, Management and Prevention of COPD 2016 Clinical Practice Guideline. MedStar Health
 Global Strategy for the Diagnosis, Management and Prevention of COPD 2016 Clinical Practice Guideline MedStar Health These guidelines are provided to assist physicians and other clinicians in making decisions
Global Strategy for the Diagnosis, Management and Prevention of COPD 2016 Clinical Practice Guideline MedStar Health These guidelines are provided to assist physicians and other clinicians in making decisions
UPMC HEALTH PLAN COPD CLINICAL PRACTICE GUIDELINE
 Relevance to Population: COPD affects 12 million people in the United States, making it the 4 th leading cause of mortality and the 2 nd leading cause of disability. It is predicted that these statistics
Relevance to Population: COPD affects 12 million people in the United States, making it the 4 th leading cause of mortality and the 2 nd leading cause of disability. It is predicted that these statistics
Adjustment of Inhaled Controller Therapy of Asthma in the Yellow Zone, Based on the Inhaler Product Used in the Green Zone Age 12 Years and Older
 Adjustment of Inhaled Controller Therapy of Asthma in the Yellow Zone, Based on the Inhaler Product Used in the Green Zone Age 12 Years and Older The Canadian Thacic Society recommends a tempary, greater
Adjustment of Inhaled Controller Therapy of Asthma in the Yellow Zone, Based on the Inhaler Product Used in the Green Zone Age 12 Years and Older The Canadian Thacic Society recommends a tempary, greater
FDCs for CO PD: From Famine to a Feast of Therapeutic Choices
 FDCs for CO PD: From Famine to a Feast of Therapeutic Choices LAURA RUNKEL, PhD Associate Director, CNS, AutoImmune/Inflammation, Ophthalmology Historically, there have been few therapies developed for
FDCs for CO PD: From Famine to a Feast of Therapeutic Choices LAURA RUNKEL, PhD Associate Director, CNS, AutoImmune/Inflammation, Ophthalmology Historically, there have been few therapies developed for
Respiratory Medications and Devices Update 2/15
 Respiratory Medications and Devices Update 2/15 Dewey Hahlbohm, PA-C, AE-C Wendy Brown, Pharm.D., MPAS, PA-C, AE-C Objectives! Review mechanism of action for asthma pharmacologic agents! Describe key patient
Respiratory Medications and Devices Update 2/15 Dewey Hahlbohm, PA-C, AE-C Wendy Brown, Pharm.D., MPAS, PA-C, AE-C Objectives! Review mechanism of action for asthma pharmacologic agents! Describe key patient
The Medical Letter. on Drugs and Therapeutics
 The Medical Letter publications are protected by US and international copyright laws. Forwarding, copying or any other distribution of this material is strictly prohibited. For further information call:
The Medical Letter publications are protected by US and international copyright laws. Forwarding, copying or any other distribution of this material is strictly prohibited. For further information call:
Pharmacy Medical Policy Asthma and Chronic Obstructive Pulmonary Disease Medication Management
 Pharmacy Medical Policy Asthma and Chronic Obstructive Pulmonary Disease Medication Management Table of Contents Policy: Commercial Information Pertaining to All Policies Endnotes Policy: Medicare References
Pharmacy Medical Policy Asthma and Chronic Obstructive Pulmonary Disease Medication Management Table of Contents Policy: Commercial Information Pertaining to All Policies Endnotes Policy: Medicare References
REVISED RESPIRATORY MEDICATION USE QUESTIONNAIRE
 REVISED RESPIRATORY MEDICATION USE QUESTIONNAIRE ID NUMBER: 0a) Date of Collection / / 0b) Staff Code Instructions: This form should be completed during the participant s clinic visit. 1) Are you regularly
REVISED RESPIRATORY MEDICATION USE QUESTIONNAIRE ID NUMBER: 0a) Date of Collection / / 0b) Staff Code Instructions: This form should be completed during the participant s clinic visit. 1) Are you regularly
GINA. At-A-Glance Asthma Management Reference. for adults, adolescents and children 6 11 years. Updated 2017
 GINA At-A-Glance Asthma Management Reference for adults, adolescents and children 6 11 years Updated 2017 This resource should be used in conjunction with the Global Strategy for Asthma Management and
GINA At-A-Glance Asthma Management Reference for adults, adolescents and children 6 11 years Updated 2017 This resource should be used in conjunction with the Global Strategy for Asthma Management and
Reference Guide for Caring for Pediatric Patients with Asthma
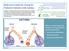 Reference Guide for Caring for Pediatric Patients with Asthma Co-Chair: Nancy Cantey Banasiak, DNP, PPCNP-BC, APRN Co-Chair: Deborah Hickman, DNP, APRN-CNP, CPNP-PC, NNP-BC Asthma and Allergy SIG Members
Reference Guide for Caring for Pediatric Patients with Asthma Co-Chair: Nancy Cantey Banasiak, DNP, PPCNP-BC, APRN Co-Chair: Deborah Hickman, DNP, APRN-CNP, CPNP-PC, NNP-BC Asthma and Allergy SIG Members
Inhaled Corticosteroid Dose Comparison in Asthma
 This Clinical Resource gives subscribers additional insight related to the Recommendations published in April 2017 ~ Resource #330402 Inhaled Corticosteroid Dose Comparison in Asthma The chart below provides
This Clinical Resource gives subscribers additional insight related to the Recommendations published in April 2017 ~ Resource #330402 Inhaled Corticosteroid Dose Comparison in Asthma The chart below provides
Clinical Policy: Roflumilast (Daliresp) Reference Number: CP.PMN.46 Effective Date: Last Review Date: 08.18
 Clinical Policy: (Daliresp) Reference Number: CP.PMN.46 Effective Date: 11.01.11 Last Review Date: 08.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Daliresp) Reference Number: CP.PMN.46 Effective Date: 11.01.11 Last Review Date: 08.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
STRIVERDI RESPIMAT (olodaterol hcl) aerosol
 STRIVERDI RESPIMAT (olodaterol hcl) aerosol Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy
STRIVERDI RESPIMAT (olodaterol hcl) aerosol Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy
REFERENCE CODE GDHC1170DFR PUBLICATION DATE M AY 2013
 REFERENCE CODE GDHC1170DFR PUBLICATION DATE M AY 2013 BRODALUMAB (PSORIASIS) - Executive Summary Table below presents key metrics for brodalumab in seven of the nine major pharmaceutical markets covered
REFERENCE CODE GDHC1170DFR PUBLICATION DATE M AY 2013 BRODALUMAB (PSORIASIS) - Executive Summary Table below presents key metrics for brodalumab in seven of the nine major pharmaceutical markets covered
REFERENCE CODE GDHC1174DFR PUBLICATION DATE M AY 2013
 REFERENCE CODE GDHC1174DFR PUBLICATION DATE M AY 2013 XELJANZ (PSORIASIS) - Executive Summary Table below presents key metrics for Xeljanz (tofacitinib) in seven of the nine major pharmaceutical markets
REFERENCE CODE GDHC1174DFR PUBLICATION DATE M AY 2013 XELJANZ (PSORIASIS) - Executive Summary Table below presents key metrics for Xeljanz (tofacitinib) in seven of the nine major pharmaceutical markets
Keppra (Epilepsy) Forecast and Market Analysis to Reference Code: GDHC1061DFR Publication Date: February 2013
 Keppra (Epilepsy) Forecast and Market Analysis to 2022 Reference Code: GDHC1061DFR Publication Date: February 2013 Executive Summary Keppra (levetiracetam) Key Metrics in the Nine Major Pharmaceutical
Keppra (Epilepsy) Forecast and Market Analysis to 2022 Reference Code: GDHC1061DFR Publication Date: February 2013 Executive Summary Keppra (levetiracetam) Key Metrics in the Nine Major Pharmaceutical
Interventions to improve adherence to inhaled steroids for asthma. Respiratory department
 Interventions to improve adherence to inhaled steroids for asthma Respiratory department Content Overview Research References Overview Asthma is a chronic breathing condition that affects more than 300
Interventions to improve adherence to inhaled steroids for asthma Respiratory department Content Overview Research References Overview Asthma is a chronic breathing condition that affects more than 300
Report generated from BNF provided by FormularyComplete (www.pharmpress.com). Accessed Formulary Status. TA Number. Section.
 Report generated from BNF provided by FormularyComplete (www.pharmpress.com). Accessed 16 02 2017 Title Formulary Status Section TA Number TA Link Annotation ACLIDINIUM BROMIDE bronchodilators ADRENALINE/EPINEPHRINE
Report generated from BNF provided by FormularyComplete (www.pharmpress.com). Accessed 16 02 2017 Title Formulary Status Section TA Number TA Link Annotation ACLIDINIUM BROMIDE bronchodilators ADRENALINE/EPINEPHRINE
Asthma Pharmacotherapy 2017: Stepwise Approach to Managing Asthma
 Asthma Pharmacotherapy 2017: Stepwise Approach to Managing Asthma Webinar for Michigan Center for Clinical Systems Improvement (Mi-CCSI) Karen Meyerson, MSN, APRN, NP-C, AE-C Director, Commercial Care
Asthma Pharmacotherapy 2017: Stepwise Approach to Managing Asthma Webinar for Michigan Center for Clinical Systems Improvement (Mi-CCSI) Karen Meyerson, MSN, APRN, NP-C, AE-C Director, Commercial Care
Joseph E. Rowane, D.O. Home Address: 6073 Volkman Road Erie, PA 16506
 Joseph E. Rowane, D.O. Home Address: 6073 Volkman Road Erie, PA 16506 Professional Address: Chest Diseases of Northwestern PA 3580 Peach Street--Suite 103 Erie, Pennsylvania 16508 Phone: (814) 864-4755
Joseph E. Rowane, D.O. Home Address: 6073 Volkman Road Erie, PA 16506 Professional Address: Chest Diseases of Northwestern PA 3580 Peach Street--Suite 103 Erie, Pennsylvania 16508 Phone: (814) 864-4755
REFERENCE CODE GDHC294DFR PUBLICATION DATE NOVEMBER 2013 PROTOPIC (ATOPIC DERMATITIS) FORECAST AND MARKET ANALYSIS TO 2022
 REFERENCE CODE GDHC294DFR PUBLICATION DATE NOVEMBER 2013 PROTOPIC (ATOPIC DERMATITIS) Executive Summary The below mentioned table presents key metrics for Protopic in the nine major pharmaceutical markets
REFERENCE CODE GDHC294DFR PUBLICATION DATE NOVEMBER 2013 PROTOPIC (ATOPIC DERMATITIS) Executive Summary The below mentioned table presents key metrics for Protopic in the nine major pharmaceutical markets
THE COPD PRESCRIBING TOOL
 THE COPD PRESCRIBING TOOL Revised edition, 2017 www.bpac.org.nz/copd CLASSIFICATION The COPD prescribing tool This tool provides pharmacological treatment options for patients with COPD based on their
THE COPD PRESCRIBING TOOL Revised edition, 2017 www.bpac.org.nz/copd CLASSIFICATION The COPD prescribing tool This tool provides pharmacological treatment options for patients with COPD based on their
About The Report. imarc. Key Questions Answered in this Report:
 About IMARC For more than 5 years, the International Market Analysis Research and Consulting Group has been a leading advisor on management strategy and market research worldwide. We partner with clients
About IMARC For more than 5 years, the International Market Analysis Research and Consulting Group has been a leading advisor on management strategy and market research worldwide. We partner with clients
What You Need to Know about Metered-Dose Inhalers and the HFA Propellant
 What You Need to Know about Metered- Inhalers and the HFA Propellant There are a number ways to deliver inhaled medication. They include: Metered-dose inhaler () Metered-dose inhaler with spacer/holding
What You Need to Know about Metered- Inhalers and the HFA Propellant There are a number ways to deliver inhaled medication. They include: Metered-dose inhaler () Metered-dose inhaler with spacer/holding
COPD Update: Focus on Intensifying LABA, LAMA and ICS Therapy
 Update: Focus on Intensifying LABA, LAMA and ICS Therapy B.C. Provincial Academic Detailing Service February 2017 Background In Canada, approximately 20 inhaled medications are approved to treat Chronic
Update: Focus on Intensifying LABA, LAMA and ICS Therapy B.C. Provincial Academic Detailing Service February 2017 Background In Canada, approximately 20 inhaled medications are approved to treat Chronic
REFERENCE CODE GDHC295DFR PUBLICATION DATE NOVEMBER 2013 ELIDEL (ATOPIC DERMATITIS) FORECAST AND MARKET ANALYSIS TO 2022
 REFERENCE CODE GDHC295DFR PUBLICATION DATE NOVEMBER 2013 ELIDEL (ATOPIC DERMATITIS) Executive Summary The below mentioned table presents key metrics for Elidel in the major pharmaceutical markets: the
REFERENCE CODE GDHC295DFR PUBLICATION DATE NOVEMBER 2013 ELIDEL (ATOPIC DERMATITIS) Executive Summary The below mentioned table presents key metrics for Elidel in the major pharmaceutical markets: the
Asthma Management in Pregnancy HEATHER HOWE, MD UNIVERSITY OF UTAH PULMONARY DIVISION
 Asthma Management in Pregnancy HEATHER HOWE, MD UNIVERSITY OF UTAH PULMONARY DIVISION Asthma Management in Pregnancy Effects of asthma on pregnancy outcomes Effects of pregnancy on asthma control Management
Asthma Management in Pregnancy HEATHER HOWE, MD UNIVERSITY OF UTAH PULMONARY DIVISION Asthma Management in Pregnancy Effects of asthma on pregnancy outcomes Effects of pregnancy on asthma control Management
Global Initiative for Asthma (GINA) What s new in GINA 2016?
 Global Initiative for Asthma (GINA) What s new in GINA 2016? GINA Global Strategy for Asthma Management and Prevention GINA: A Brief History Established in 1993 Collaboration between NHLBI and WHO Multiple
Global Initiative for Asthma (GINA) What s new in GINA 2016? GINA Global Strategy for Asthma Management and Prevention GINA: A Brief History Established in 1993 Collaboration between NHLBI and WHO Multiple
Choosing an inhaler for COPD made simple. Dr Simon Hart Castle Hill Hospital
 Choosing an inhaler for COPD made simple Dr Simon Hart Castle Hill Hospital 1 Declaration of interests I have received speaker fees, sponsorship to attend conferences, and funding for research from companies
Choosing an inhaler for COPD made simple Dr Simon Hart Castle Hill Hospital 1 Declaration of interests I have received speaker fees, sponsorship to attend conferences, and funding for research from companies
CURRICULUM VITAE KENNETH D. CHINSKY, M.D.
 KENNETH D. CHINSKY, M.D. Chest Diseases of Northwestern PA 3580 Peach Street, Suite 103A Erie, PA 16508 www.chestdiseases.yourmd.com Phone: (814) 864-4755 FAX: (814) 864-5430 Email: Sigsbee@aol.com Home
KENNETH D. CHINSKY, M.D. Chest Diseases of Northwestern PA 3580 Peach Street, Suite 103A Erie, PA 16508 www.chestdiseases.yourmd.com Phone: (814) 864-4755 FAX: (814) 864-5430 Email: Sigsbee@aol.com Home
COPD Update. Plus New and Improved Products for Inhaled Therapy. Catherine Bourg Rebitch, PharmD, BCACP Clinical Associate Professor
 COPD Update Plus New and Improved Products for Inhaled Therapy Catherine Bourg Rebitch, PharmD, BCACP Clinical Associate Professor Disclosure The presenter has nothing to disclose concerning possible financial
COPD Update Plus New and Improved Products for Inhaled Therapy Catherine Bourg Rebitch, PharmD, BCACP Clinical Associate Professor Disclosure The presenter has nothing to disclose concerning possible financial
COPD: Treatment Update Property of Presenter. Not for Reproduction. Barry Make, MD Professor of Medicine National Jewish Health
 COPD: Treatment Update Barry Make, MD Professor of Medicine National Jewish Health Disclosures Advisory board, consultant, multi-center trial, research funding, Data Safety Monitoring Board (DSMB), or
COPD: Treatment Update Barry Make, MD Professor of Medicine National Jewish Health Disclosures Advisory board, consultant, multi-center trial, research funding, Data Safety Monitoring Board (DSMB), or
Lamictal (Epilepsy) Forecast and Market Analysis to Reference Code: GDHC1062DFR Publication Date: February 2013
 Lamictal (Epilepsy) Forecast and Market Analysis to 2022 Reference Code: GDHC1062DFR Publication Date: February 2013 Executive Summary Lamictal (lamotrigine) Key Metrics in the Nine Major Pharmaceutical
Lamictal (Epilepsy) Forecast and Market Analysis to 2022 Reference Code: GDHC1062DFR Publication Date: February 2013 Executive Summary Lamictal (lamotrigine) Key Metrics in the Nine Major Pharmaceutical
3/24/2017. Learning Objectives. Current Issues In Airway Pharmacology. Beta-Agonists. Bronchodilating Agents. Adrenergic Autonomic Control
 Learning Objectives Current Issues In Airway Pharmacology Arthur Jones, EdD, RRT This Presentation is Approved for 1.5 CRCE Credit Hours Explain the actions, effects, indications, & contraindications for
Learning Objectives Current Issues In Airway Pharmacology Arthur Jones, EdD, RRT This Presentation is Approved for 1.5 CRCE Credit Hours Explain the actions, effects, indications, & contraindications for
Clinical Policy: Mepolizumab (Nucala) Reference Number: ERX.SPA.214 Effective Date:
 Clinical Policy: (Nucala) Reference Number: ERX.SPA.214 Effective Date: 07.01.16 Last Review Date: 02.19 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Nucala) Reference Number: ERX.SPA.214 Effective Date: 07.01.16 Last Review Date: 02.19 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
4/3/2018 BRANDY BURGESS, APRN-CNS, MSN-RN APRIL 12-14, 2018
 BRANDY BURGESS, APRN-CNS, MSN-RN APRIL 12-14, 2018 Approach to treatment List of medication Abbreviations When to use medications Pharmokinetics Pharmacodynamics Step wise therapy both for acute and chronic
BRANDY BURGESS, APRN-CNS, MSN-RN APRIL 12-14, 2018 Approach to treatment List of medication Abbreviations When to use medications Pharmokinetics Pharmacodynamics Step wise therapy both for acute and chronic
Long Term Care Formulary RS -29
 RESTRICTED USE Asthma/COPD Management 1 of 6 PROTOCOL: Asthma Glossary of Medication Acronyms: SABA: short-acting beta agonist (e.g. salbutamol) SABD: short-acting bronchodilator (e.g. ipratropium or SABA)
RESTRICTED USE Asthma/COPD Management 1 of 6 PROTOCOL: Asthma Glossary of Medication Acronyms: SABA: short-acting beta agonist (e.g. salbutamol) SABD: short-acting bronchodilator (e.g. ipratropium or SABA)
REFERENCE CODE GDHC1114CFR PUBLICATION DATE M AY 2013
 REFERENCE CODE GDHC1114CFR PUBLICATION DATE M AY 2013 PSORIASIS - Psoriasis - India Drug Forecast and Market Analysis to Executive Summary Sales for Psoriasis in India 2012 We estimate the 2012 PsO drug
REFERENCE CODE GDHC1114CFR PUBLICATION DATE M AY 2013 PSORIASIS - Psoriasis - India Drug Forecast and Market Analysis to Executive Summary Sales for Psoriasis in India 2012 We estimate the 2012 PsO drug
Abbreviated Class Review: Chronic Obstructive Pulmonary Disease (COPD)
 Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Executive summary: Month/Year of Review: February
Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Executive summary: Month/Year of Review: February
Proposed Preferred Drug List. Clinical Criteria
 Proposed Preferred Drug List with Clinical Criteria Proposal for TennCare February 7, 2008 Page 1 of 87 PDL Decision Process The primary clinical decision that needs to be made is determining if the drugs
Proposed Preferred Drug List with Clinical Criteria Proposal for TennCare February 7, 2008 Page 1 of 87 PDL Decision Process The primary clinical decision that needs to be made is determining if the drugs
The clinical effectiveness and costeffectiveness. treatment of chronic asthma in children under the age of 12 years
 The clinical effectiveness and costeffectiveness of corticosteroids for the treatment of chronic asthma in children under the age of 12 years Submission of evidence from AstraZeneca UK Ltd regarding the
The clinical effectiveness and costeffectiveness of corticosteroids for the treatment of chronic asthma in children under the age of 12 years Submission of evidence from AstraZeneca UK Ltd regarding the
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Seebri Neohaler) Reference Number: CP.CPA.150 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy
Clinical Policy: (Seebri Neohaler) Reference Number: CP.CPA.150 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy
Alberta Childhood Asthma Pathway for Primary Care
 Asthma Diagnosis Box 1 Diagnosis: Based on symptom pattern, careful and thorough history of symptoms (wheeze, cough, night waking and activity limitations), and assessment of family history of asthma and
Asthma Diagnosis Box 1 Diagnosis: Based on symptom pattern, careful and thorough history of symptoms (wheeze, cough, night waking and activity limitations), and assessment of family history of asthma and
He is still Short of Breath Is there any new puffer? Saidul Ansary
 He is still Short of Breath Is there any new puffer? Saidul Ansary Mr S C 66 yr. old retired Engineer for pre op elective assessment I am fine but Anaesthetist said I need to see you. I am little bit SOB,
He is still Short of Breath Is there any new puffer? Saidul Ansary Mr S C 66 yr. old retired Engineer for pre op elective assessment I am fine but Anaesthetist said I need to see you. I am little bit SOB,
Objectives. Speaker has no relationship to disclose. Sneezes, Wheezes, and Respiratory Diseases: An update on Asthma, Allergic Rhinitis,
 Sneezes, Wheezes, and Respiratory Diseases: An update on Asthma, Allergic Rhinitis, and COPD Amelie Hollier, DNP, FNP-BC, FAANP Advanced Practice Education Associates Objectives Evaluation of medications,
Sneezes, Wheezes, and Respiratory Diseases: An update on Asthma, Allergic Rhinitis, and COPD Amelie Hollier, DNP, FNP-BC, FAANP Advanced Practice Education Associates Objectives Evaluation of medications,
DOD PHARMACY AND THERAPEUTICS COMMITTEE RECOMMENDATIONS INFORMATION FOR THE UNIFORM FORMULARY BENEFICIARY ADVISORY PANEL
 DOD PHARMACY AND THERAPEUTICS COMMITTEE RECOMMENDATIONS INFORMATION FOR THE UNIFORM FORMULARY BENEFICIARY ADVISORY PANEL I. Uniform Formulary Review Process Under 10 U.S.C. 1074g, as implemented by 32
DOD PHARMACY AND THERAPEUTICS COMMITTEE RECOMMENDATIONS INFORMATION FOR THE UNIFORM FORMULARY BENEFICIARY ADVISORY PANEL I. Uniform Formulary Review Process Under 10 U.S.C. 1074g, as implemented by 32
MDI Bonanza. Dwayne Griffin, DO
 MDI Bonanza Dwayne Griffin, DO Bonanza 3. A MDI costing $200 - $500 per month SISYPHUS MDI Griffin Mountain Evolution of Deliver Systems for COPD in the US 2003 2009 2011 2013 2004 2012 2014 Prescribing
MDI Bonanza Dwayne Griffin, DO Bonanza 3. A MDI costing $200 - $500 per month SISYPHUS MDI Griffin Mountain Evolution of Deliver Systems for COPD in the US 2003 2009 2011 2013 2004 2012 2014 Prescribing
Clinical Policy: Omalizumab (Xolair) Reference Number: ERX.SPA.141 Effective Date:
 Clinical Policy: (Xolair) Reference Number: ERX.SPA.141 Effective Date: 03.01.14 Last Review Date: 02.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Xolair) Reference Number: ERX.SPA.141 Effective Date: 03.01.14 Last Review Date: 02.18 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Oregon Health Plan prescription benefit updates
 Oregon Health Plan prescription benefit updates EOCCO s prescription program is a pharmacy benefit that offers members a choice of safe and effective medication treatments. The program also helps you save
Oregon Health Plan prescription benefit updates EOCCO s prescription program is a pharmacy benefit that offers members a choice of safe and effective medication treatments. The program also helps you save
