MILLIPRED H 27 O 8. Na 2. P; the molecular weight is Its chemical structure is:
|
|
|
- Daisy Palmer
- 6 years ago
- Views:
Transcription
1 l510c00 R 06/13 MILLIPRED (Prednisolone Sodium Phosphate Oral Solution, 10 mg Prednisolone Base per 5 ml) MILLIPRED (Prednisolone Sodium Phosphate Oral Solution,10 mg Prednisolone Base per 5 ml) Zylera Pharmaceuticals Rx only DESCRIPTION Prednisolone per 5 ml) is a dye free, pale to light yellow solution. Each 5 ml (teaspoonful) of Millipred Oral Solution contains 13.4 mg prednisolone sodium phosphate (10 mg prednisolone base) in a palatable, aqueous vehicle. Inactive Ingredients: Prednisolone per 5 ml) contains the following inactive ingredients: anti-bitter mask, corn syrup, edetate disodium, glycerin, grape flavor, hydroxyethylcellulose, methylparaben, potassium phosphate dibasic, potassium phosphate monobasic, purified water, and sodium saccharin. Prednisolone sodium phosphate occurs as white or slightly yellow, friable granules or powder. It is freely soluble in water; soluble in methanol; slightly soluble in alcohol and in chloroform; and very slightly soluble in acetone and in dioxane.the chemical name of prednisolone sodium phosphate is pregna-1,4-diene-3,20-dione,11,1 7-dihydroxy-21- (phosphonooxy)- disodium salt, (11β)-. The empirical formula is C 21 H 27 Na 2 O 8 P; the molecular weight is Its chemical structure is: Pharmacological Category: Glucocorticoid CLINICAL PHARMACOLOGY Naturally occurring glucocorticoids (hydrocortisone), which also have salt-retaining properties, are used as replacement therapy in adrenocortical deficiency states. Their synthetic analogs are primarily used for their potent anti-inflammatory effects in disorders of many organ systems. Prednisolone is a synthetic adrenocortical steroid drug with predominantly glucocorticoid properties. Some of these properties reproduce the physiologi-cal actions of endogenous glucocorticosteroids, but others do not necessarily reflect any of the adrenal hormones' normal functions; they are seen only after administration of large therapeutic doses of the drug. The pharmacological effects of prednisolone which are due to its glucocorticoid properties include: promotion of gluconeogenesis; increased deposition of glycogen in the liver; inhibition of the utilization of glucose; anti-insulin activity; increased catabolism of protein; increased lipolysis; stimulation of fat synthesis and storage; increased glomerular filtration rate and resulting increase in urinary excretion of urate (creatinine excretion remains unchanged); and increased calcium excretion. Depressed production of eosinophils and lymphocytes occurs, but erythropoiesis and production of polymorphonuclear leukocytes are stimulated. Inflammatory processes (edema, fibrin deposition, capillary dilatation, migration of leukocytes and phagocytosis) and the later stages of wound healing (capillary proliferation, deposition of collagen, cicatrization) are inhibited. Prednisolone can stimulate secretion of various components of gastric juice. Suppression of the production of corticotropin may lead to suppression of endogenous corticosteroids. Prednisolone has slight mineralocorticoid activity, whereby entry of sodium into cells and loss of intracellular potassium is stimulated. This is particularly evident in the kidney, where rapid ion exchange leads to sodium retention and hypertension. Prednisolone is rapidly and well absorbed from the gastrointestinal tract following oral administration. Prednisolone per 5 ml) produces a 14% higher peak plasma level of prednisolone which occurs 20% faster than that seen with tablets. Prednisolone is 70-90% protein bound in the plasma, and it is eliminated from the plasma with a half-life of 2 to 4 hours. It is metabolized mainly in the liver and excreted in the urine as sulfate and glucuronide conjugates. The systemic availability, metabolism and elimination of prednisolone after administration of single weight-based doses (0.8 mg/ kg) of intravenous (IV) prednisolone and oral prednisone were reported in a small study of 19 young (23 to 34 years) and 12 elderly (65 to 89 years) subjects. Results showed that the systemic availability of total and unbound prednisolone, as well as interconversion between prednisolone and prednisone were independent of age. The mean unbound fraction of prednisolone was higher, and steady- state volume of distribution (Vss) of unbound prednisolone was reduced in elderly patients. Plasma prednisolone concentrations were higher in elderly subjects, and the higher AUCs of total and unbound prednisolone were most likely reflective of an impairedmetabolic clearance, evidenced by reduced fractional urinary clearance of 6 β-hydroxyprednisolone. Despite these findings of higher total and unbound prednisolone concentra-tions, elderly subjects had higher AUCs of cortisol, suggesting that the elderly population is less sensitive to suppression of endogenous cortisol or their capacity for hepatic inactivation of cortisol is diminished. INDICATIONS AND USAGE Prednisolone per 5 ml) is indicated in the following conditions: 1. Allergic States Control of severe or incapacitating allergic conditions intractable to adequate trials of conventional treatment in adult and pediatric populations with: seasonal or perennial allergic rhinitis; asthma; contact dermatitis; atopic dermatitis; serum sickness; drug hypersensitivity reactions. 2. Dermatologic Diseases Pemphigus; bullous dermatitis herpetiformis; severe erythema multiforme (Stevens-Johnson syndrome); exfoliative erythroderma; mycosis fungoides. 3. Edematous States To induce diuresis or remission of proteinuria in nephrotic
2 syndrome in adults with lupus erythematosus and in adults and pediatric populations, with idiopathic nephrotic syndrome, without uremia. 4. Endocrine Disorders Primary or secondary adrenocortical insufficiency (hydrocortisone or cortisone is the first choice; synthetic analogs may be used in conjunction with mineralocorticoids where applicable; in infancy mineralocorticoid supplementation is of particular importance); congenital adrenal hyperplasia; 5. Gastrointestinal Diseases To tide the patient over a critical period of the disease in: ulcerative colitis; regional enteritis. 6. Hematologic Disorders Idiopathic thrombocytopenic purpura in adults; selected cases of secondary thrombocytopenia; acquired (autoimmune) hemolytic anemia; pure red cell aplasia; Diamond-Blackfan anemia. 7. Neoplastic Diseases For the treatment of acute leukemia and aggressive lymphomas in adults and children. 8. Nervous System Acute exacerbations of multiple sclerosis. 9. Ophthalmic Diseases Uveitis and ocular inflammatory conditions unresponsive to topical corticosteroids; temporal arteritis; sympathetic ophthalmia. 10. Respiratory Diseases Symptomatic sarcoidosis; idiopathic eosinophilic pneumonias; fulminating or disseminated pulmonary tuberculosis when used concurrently with appropriate antituberculous chemotherapy; asthma (as distinct from allergic asthma listed above under "Allergic States"), hypersensitivity pneumonitis, idiopathic pulmonary fibrosis, acute exacerbations of chronic obstructive pulmonary disease (COPD), and Pneumocystis carinii pneumonia (PCP) associated with hypoxemia occurring in an HIV (+) individual who is also under treatment with appropriate anti-pcp antibiotics. Studies support the efficacy of systemic corticosteroids for the treatment of these conditions: allergic bronchopulmonary aspergillosis, idiopathic bronchiolitis obliterans with organizing pneumonia. 11. Rheumatic Disorders As adjunctive therapy for short term administration (to tide the patient over an acute episode or exacerbation) in: psoriatic arthritis; rheumatoid arthritis, including juvenile rheumatoid arthritis (selected cases may require low dose maintenance therapy); ankylosing spondylitis; acute and subacute bursitis; acute nonspecific tenosynovitis; acute gouty arthritis; epicondylitis. For the treatmentof systemic lupus erythematosus, dermatomyositis (polymyositis), polymyalgia rheumatica, Sjogren's syndrome, relapsing polychondritis, and certain cases of vasculitis. 12. Miscellaneous Tuberculous meningitis with subarachnoid block or impending block, tuberculosis with enlarged mediastinal lymph nodes causing respiratory difficulty, and tuberculosis with pleural or pericardial effusion (appropriate antituberculous chemotherapy must be used concurrently when treating any tuberculosis complications); trichinosis with neurologic or myocardial involvement; acute or chronic solid organ rejection (with or without other agents). CONTRAINDICATIONS Systemic fungal infections. Hypersensitivity to the drug or any of its components. WARNINGS General in patients on corticosteroid therapy subjected to unusual stress, increased dosage of rapidly acting corticosteroids before, during and after the stressful situation is indicated. Cardio-renal Average and large doses of hydrocortisone or cortisone can cause elevation of blood pressure, salt and water retention, and increased excretion of potassium. These effects are less likely to occur with the synthetic derivatives except when used in large doses. Dietary salt restriction and potassium supplementation may be necessary. All corticoste-roids increase calcium excretion. Endocrine Corticosteroids can produce reversible hypothalamic-pituitary adrenal (HPA) axis suppression with the potential for glucocorticoste-roid insufficiency after withdrawal of treatment.metabolic clearance of corticosteroids is decreased in hypothyroid patients and increased in hyperthyroid patients. Changes in thyroid status of the patient may necessitate adjustment in dosage. Infections (General) Persons who are on drugs which suppress the immune system are more susceptible to infections than healthy individuals. There may be decreased resistance and inability to localize infection when corticosteroids are used. Infection with any pathogen including viral, bacterial, fungal, protozoan or helminthic infection, in any location of the body, may be associated with the use of corticosteroids alone or in combination with other immunosuppressive agents that affect humoral or cellular immunity, or neutrophil function. These infections may be mild to severe, and, with increasing doses of corticosteroids, the rate of occurrence of infectious complications increases. Corticosteroids may also mask some signs of infection after it has already started. Infections (Viral) Chicken pox and measles, for example, can have a more serious or even fatal course in non-immune children or adults on corticosteroids. In such children or adults who have not had these diseases, particular care should be taken to avoid exposure. How the dose, route and duration of corticosteroid administration affect the risk of developing a disseminated infection is not known. The contribution of the underlying disease and/or prior corticosteroid treatment to the risk is also not known. If exposed to chicken pox,prophylaxis with varicella zoster immune globulin (VZIG) may be indicated. If exposed to measles, prophylaxis with immunoglobulin (IG) may be indicated. (See the respective package inserts for complete VZIG and IG prescribing information). If chicken pox develops, treatment with antiviral agents should be considered. Ophthalmic Use of corticosteroids may produce posterior subcapsular cataracts, glaucoma with possible damage to the optic nerves, and may enhance the establishment of secondary ocular infections due to bacteria, fungi or viruses. The use of oral corticosteroids is not recommended in the treatment of optic neuritis and may lead to an increase in the risk of new episodes. Corticoste-roids should not be used in active ocular herpes simplex. Special pathogens Latent disease may be activated or there may be an exacerbation of intercurrent infections due to pathogens, including those caused by Candida, Mycobacterium, Ameba, Toxoplasma, Pneumocys-tis, Cryptococcus, Nocardia, etc. Corticosteroids may activate latent amebiasis. Therefore, it is recommended that latent or active amebiasis be ruled out before initiating corticosteroid therapy in any patient who has spent time in the tropics or in any patient with unexplained diarrhea. Similarly, corticosteroids should be used with great care in patients with known or suspected Strongyloides (threadworm) infestation. In such patients, corticosteroid-induced immunosuppression may lead to Strongyloides hyperinfection and dissemination with widespread larval migration, often accompanied by severe enterocolitis and potentially fatal gram-negative septicemia. Corticosteroids should not be used in cerebral malaria. TuberculosisThe use of prednisolone in active tuberculosis should be restricted to those cases of fulminating or disseminated tuberculosis in which the corticosteroid is used for the management of the disease in conjunction with an appropriate antituberculous regimen.if corticosteroids are indicated in
3 patients with latent tuberculosis or tuberculin reactivity, close observation is necessary as reactivation of the disease may occur. During prolonged corticosteroid therapy, these patients should receive chemopro-phylaxis. Vaccination Administration of live or live, attenuated vaccines is contraindicated in patients receiving immunosup-pressive doses of corticosteroids. Killed or inactivated vaccines may be administered, however, the response to such vaccines cannot be predicted. Immunization procedures may be undertaken in patients who are receiving corticosteroids as replacement therapy, e.g., for Addison's disease. PRECAUTIONS GeneralThe lowest possible dose of corticosteroid should be used to control the condition under treatment, and when reduction in dosage is possible, the reduction should be gradual.since complications of treatment with glucocorti-coids are dependent on the size of the dose and the duration of treatment, a risk/ benefit decision must be made in each individual case as to dose and duration of treatment and as to whether daily or intermittent therapy should be used.there is an enhanced effect of corticosteroids in patients with hypothyroidism and in those with cirrhosis.kaposi's sarcoma has been reported to occur in patients receiving corticosteroid therapy, most often for chronic conditions. Discontinuation of corticosteroids may result in clinical improvement. Cardio-renal As sodium retention with resultant edema and potassium loss may occur in patients receiving corticosteroids, these agents should be used with caution in patients with hypertension, congestive heart failure, or renal insufficiency. Endocrine Drug-induced secondary adrenocortical insufficiency may be minimized by gradual reduction of dosage. This type of relative insufficiency may persist for months after discontinuation of therapy; therefore, in any situation of stress occurring during that period, hormone therapy should be reinstituted. Since mineralocorticoid secretion may be impaired, salt and/or a mineralocorticoid should be administered concurrently. Gastrointestinal Steroids should be used with caution in nonspecific ulcerative colitis, if there is a probability of impending perforation, abscess or other pyogenic infection; diverticulitis; fresh intestinal anastomo-ses; active or latent peptic ulcer.signs of peritoneal irritation following gastrointes-tinal perforation in patients receiving corticoste-roids may be minimal or absent. Musculoskeletal Corticosteroids decrease bone formation and increase bone resorption both through their effect on calcium regulation (i.e., decreasing absorption and increasing excretion) and inhibition of osteoblast function. This, together with a decrease in the protein matrix of the bone secondary to an increase in protein catabolism, and reduced sex hormone production, may lead to inhibition of bone growth in children and adolescents and the development of osteoporosis at any age. Special consideration should be given to patients at increased risk of osteoporosis (i.e., post-menopausal women) before initiating corticosteroid therapy. Neuro-psychiatric Although controlled clinical trials have shown corticosteroids to be effective in speeding the resolution of acute exacerbations of multiple sclerosis, they do not show that they affect the ultimate outcome or natural history of the disease. The studies do show that relatively high doses of corticosteroids are necessary to demonstrate a significant effect. (See DOSAGE AND ADMINISTRATION.) An acute myopathy has been observed with the use of high doses of corticoste-roids, most often occurring in patients with disorders of neuromuscular transmission (e.g., myasthenia gravis), or in patients receiving concomitant therapy with neuromuscular blocking drugs (e.g., pancuronium). This acute myopathy is generalized, may involve ocular and respiratory muscles, and may result in quadriparesis. Elevation of creatinine kinase may occur. Clinical improvement or recovery after stopping corticoste-roids may require weeks to years. Psychic derangements may appear when corticosteroids are used, ranging from euphoria, insomnia, mood swings, personality changes, and severe depression, to frank psychotic manifestations. Also, existing emotional instability or psychotic tendencies may be aggravated by corticosteroids. Ophthalmic Intraocular pressure may become elevated in some individuals. If steroid therapy is continued for more than 6 weeks, intraocular pressure should be monitored. Information for Patients Patients should be warned not to discontinue the use of Millipred Oral Solution (10 mg Prednisolone per 5 ml) abruptly or without medical supervision, to advise any medical attendants that they are taking it, and to seek medical advice at once should they develop fever or other signs of infection.persons who are on immunosuppressant doses of corticosteroids should be warned to avoid exposure to chicken pox or measles. Patients should also be advised that if they are exposed, medical advice should be sought without delay. Drug Interactions Drugs such as barbiturates, phenytoin, ephedrine, and rifampin, which induce hepatic microsomal drug metabolizing enzyme activity may enhance metabolism of prednisolone and require that the dosage of Millipred Oral Solution (10 mg Prednisolone per 5 ml) be increased. Increased activity of both cyclosporin and corticosteroids may occur when the two are used concurrently. Convulsions have been reported with this concurrent use. Estrogens may decrease the hepatic metabolism of certain corticosteroids thereby increasing their effect. Ketoconazole has been reported to decrease the metabolism of certain corticosteroids by up to 60% leading to an increased risk of corticosteroid side effects. Coadministration of corticosteroids and warfarin usually results in inhibition of response to warfarin, although there have been some conflicting reports. Therefore, coagulation indices should be monitored frequently to maintain the desired anticoagulant effect. Concomitant use of aspirin (or other nonsteroidal anti-inflammatory agents) and corticosteroids increases the risk of gastrointesti-nal side effects. Aspirin should be used cautiously in conjunction with corticosteroids in hypoprothrombinemia. The clearance of salicylates may be increased with concurrent use of corticosteroids. When corticosteroids are administered concomitantly with potassium-depleting agents (i.e., diuretics, amphotericin-b), patients should be observed closely for development of hypokalemia. Patients on digitalis glycosides may be at increased risk of arrhythmias due to hypokalemia. Concomitant use of anticholinesterase agents and corticosteroids may produce severe weakness in patients with myasthenia gravis. If possible, anticholinesterase agents should be withdrawn at least 24 hours before initiating corticosteroid therapy. Due to inhibition of antibody response, patients on prolonged corticosteroid therapy may exhibit a diminished response to toxoids and live or inactivated vaccines. Corticosteroids may also potentiate the replication of some organisms contained in live attenuated vaccines. If possible, routine administration of vaccines or toxoids should be deferred until corticosteroid therapy is discontinued. Because corticosteroids may increase blood glucose concentrations, dosage adjustments of antidiabetic agents may be required. Corticosteroids may suppress reactions to skin tests. Pregnancy Teratogenic Effects Pregnancy Category CPrednisolone has been shown to be teratogenic in many species when given in doses equivalent to the human dose. Animal studies in which prednisolone has been given to pregnant mice, rats, and
4 rabbits have yielded an increased incidence of cleft palate in the offspring. There are no adequate and well-controlled studies in pregnant women. Millipred Oral Solution (10 mg Prednisolone per 5 ml) should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. Infants born to mothers who have received corticosteroids during pregnancy should be carefully observed for signs of hypoadrenalism. Nursing Mothers Systemically administered corticosteroids appear in human milk and could suppress growth, interfere with endogenous corticosteroid production, or cause other untoward effects. Caution should be exercised when Millipred Oral Solution (10 mg Prednisolone per 5 ml) is administered to a nursing woman. Pediatric Use The efficacy and safety of prednisolone in the pediatric population are based on the well-established course of effect of corticosteroids which is similar in pediatric and adult populations. Published studies provide evidence of efficacy and safety in pediatric patients for the treatment of nephrotic syndrome (>2 years of age), and aggressive lymphomas and leukemias (>1 month of age). However, some of these conclusions and other indications for pediatric use of corticosteroid, e.g., severe asthma and wheezing, are based on adequate and well-controlled trials conducted in adults, on the premises that the course of the diseases and their pathophysiology are considered to be substantially similar in both populations. The adverse effects of prednisolone in pediatric patients are similar to those in adults (see ADVERSE REACTIONS). Like adults, pediatric patients should be carefully observed with frequent measurements of blood pressure, weight, height, intraocular pressure, and clinical evaluation for the presence of infection, psychosocial disturbances, thromboembolism, peptic ulcers, cataracts, and osteoporosis. Children who are treated with corticosteroids by any route, including systemically administered corticosteroids,may experience a decrease in their growth velocity. This negative impact of corticosteroids on growth has been observed at low systemic doses and in the absence of laboratory evidence of HPA axis suppression (i.e., cosyntropin stimulation and basal cortisol plasma levels). Growth velocity may therefore be a more sensitive indicator of systemic corticosteroid exposure in children than some commonly used tests of HPA axis function. The linear growth of children treated with corticoste-roids by any route should be monitored, and the potential growth effects of prolonged treatment should be weighed against clinical benefits obtained and the availability of other treatment alternatives. In order to minimize the potential growth effects of corticosteroids, children should be titrated to the lowest effective dose. Geriatric Use Clinical studies of prednisolone sodium phosphate oral solution did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience with prednisolone sodium phosphate has not identified differences in responses between the elderly and younger patients. However, the incidence of corticosteroid-induced side effects may be increased in geriatric patients and appear to be dose-related. Osteoporosis is the most frequently encountered complication, which occurs at a higher incidence rate in corticosteroid-treated geriatric patients as compared to younger populations and in age-matched controls. Losses of bone mineral density appear to be greatest early on in the course of treatment and may recover over time after steroid withdrawal or use of lower doses (i.e., 5 mg/day). Prednisolone doses of 7.5 mg/ day or higher have been associated with an increased relative risk of both vertebral and nonvertebral fractures, even in the presence of higher bone density compared to patients with involutional osteoporosis. Routine screening of geriatric patients, including regular assessments of bone mineral density and institution of fracture preventionstrategies along with regular review of prednisolone sodium phosphate indication should be undertaken to minimize complications and keep the dose at the lowest acceptable level. Co-administration of bisphosphonates has been shown to retard the rate of bone loss in corticosteroid-treated males and post-menopausal females, and these agents are recommended in the prevention and treatment of corticosteroid-induced osteoporosis. It has been reported that equivalent weight-based doses yield higher total and unbound prednisolone plasma concentrations and reduced renal and non-renal clearance in elderly patients compared to younger populations. However, it is not clear whether dosing reductions would be necessary in elderly patients, since these pharmacokinetic alterations may be offset by age-related differences in responsiveness of target organs and/or less pronounced suppression of adrenal release of cortisol. Dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy. This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function (see CLINICAL PHARMACOLOGY). ADVERSE REACTIONS (LISTED ALPHABETI-CALLY UNDER EACH SUBSECTION) Cardiovascular: Hypertrophic cardiomyopathy in premature infants. Dermatologic: Facial erythema; increased sweating; impaired wound healing; may suppress reactions to skin tests; petechiae and ecchymoses; thin fragile skin; urticaria; edema. Endocrine: Decreased carbohydrate tolerance; development of cushingoid state; hirsutism; increased requirements for insulin or oral hypoglycemic agents in diabetic patients; manifestations of latent diabetes mellitus; menstrual irregularities; secondary adrenocortical and pituitary unresponsiveness, particularly in times of stress, as in trauma, surgery or illness; suppression of growth in children. Fluid and Electrolyte Disturbances:Congestive heart failure in susceptible patients; fluid retention; hypertension; hypokalemic alkalosis; potassium loss; sodium retention. Gastrointestinal: Abdominal distention; elevation in serum liver enzyme levels (usually reversible upon discontinuation); pancreatitis; peptic ulcer with possible perforation and hemorrhage; ulcerative esophagitis. Metabolic: Negative nitrogen balance due to protein catabolism. Musculoskeletal: Aseptic necrosis of femoral and humeral heads; loss of muscle mass; muscle weakness; osteoporosis; pathologic fracture of long bones; steroid myopathy; tendon rupture; vertebral compression fractures. Neurological: Convulsions; headache; increased intracranial pressure with papilledema (pseudotumor cerebri) usually following discontinuation of treatment; psychic disorders; vertigo. Ophthalmic: Exophthalmos; glaucoma; increased intraocular pressure; posterior subcapsular cataracts. Other: Increased appetite; malaise; nausea; weight gain. OVERDOSAGE The effects of accidental ingestion of large quantities of prednisolone over a very short period of time have not been reported, but prolonged use of the drug can produce mental symptoms, moon face, abnormal fat deposits, fluid retention, excessive appetite, weight gain, hypertrichosis, acne, striae, ecchymosis,
5 increased sweating, pigmentation, dry scaly skin, thinning scalp hair, increased blood pressure, tachycardia, Ithrombophlebitis, decreased resistance to infection, negative nitrogen balance with delayed bone and wound healing, headache, weakness, menstrual disorders, accentuated menopausal symptoms, neuropathy, fractures, osteoporosis, peptic ulcer, decreased glucose tolerance, hypokalemia, and adrenal insufficiency. Hepatomegaly and abdominal distention have been observed in children.treatment of acute overdosage is by immediate gastric lavage or emesis followed by supportive and symptomatic therapy. For chronic overdosage in the face of severe disease requiring continuous steroid therapy, the dosage of prednisolone may be reduced only temporarily, or alternate day treatment may be introduced. DOSAGE AND ADMINISTRATION The initial dosage of Millipred Oral Solution (10 mg Prednisolone per 5 ml) may vary from 2.5 ml to 30 ml (5 to 60 mg prednisolone base) per day depending on the specific disease entity being treated. In situations of less severity, lower doses will generally suffice while in selected patients higher initial doses may be required. The initial dosage should be maintained or adjusted until a satisfactory response is noted. If after a reasonable period of time, there is a lack of satisfactory clinical response, Millipred Oral Solution (10 mg Prednisolone per 5 ml) should be discontinued and the patient placed on other appropriate therapy. IT SHOULD BE EMPHASIZED THAT DOSAGE REQUIREMENTS ARE VARIABLE AND MUST BE INDIVIDUALIZED ON THE BASIS OF THE DISEASE UNDER TREATMENT AND THE RESPONSE OF THE PATIENT. After a favorable response is noted, the proper maintenance dosage should be determined by decreasing the initial drug dosage in small decrements at appropriate time intervals until the lowest dosage which will maintain an adequate clinical response is reached. It should be kept in mind that constant monitoring is needed in regard to drug dosage. Included in the situations which may make dosage adjustments necessary are changes in clinical status secondary to remissions or exacerbations in the disease process, the patient's individual drug responsiveness, and the effect of patient exposure to stressful situations not directly related to the disease entity under treatment; in this latter situation it may be necessary to increase the dosage of Millipred Oral Solution (10 mg Prednisolone per 5 ml) for a period of time consistent with the patient's condition. If after long term therapy the drug is to be stopped, it is recommended that it be withdrawn gradually rather than abruptly. In the treatment of acute exacerbations of multiple sclerosis, daily doses of 200 mg of prednisolone for a week followed by 80 mg every other day or 4 to 8 mg dexamethasone every other day for one month have been shown to be effective. In pediatric patients, the initial dose of Millipred Oral Solution (10 mg Prednisolone per 5 ml) may vary depending on the specific disease entity being treated. The range of initial doses is 0.14 to 2 mg/kg/day in three or four divided doses (4 to 60 mg/m2bsa/day). The standard regimen used to treat nephrotic syndrome in pediatric patients is 60 mg/m2/day given in three divided doses for 4 weeks, followed by 4 weeks of single dose alternate-day therapy at 40 mg/m2/day.the National Heart, Lung, and Blood Institute (NHLBI) recommended dosing for systemic prednisone, prednisolone or methylprednisolone in children whose asthma is uncontrolled by inhaled corticosteroids and long-acting bronchodilators is 1-2 mg/ kg/day in single or divided doses. It is further recommended that short course, or "burst" therapy, be continued until a child achieves a peak expiratory flow rate of 80% of his or her personal best or symptoms resolve. This usually requires 3 to 10 days of treatment, although it can take longer. There is no evidence that tapering the dose after improvement will prevent a relapse. For the purpose of comparison, 5 ml of Millipred Oral Solution (13.4 mg Prednisolone sodium phosphate) is equivalent to the following milligram dosage of the various glucocorticoids: Cortisone, 50 Triamcinolone, 8 Hydrocortisone, 40 Paramethasone, 4 Prednisolone, 10 Betamethasone, 1.5 Prednisone, 10 Dexamethasone, 1.5 Methylprednisolone, 8 These dose relationships apply only to oral or intravenous administration of these compounds. When these substances or their derivatives are injected intramuscularly or into joint spaces, their relative properties may be greatly altered. HOW SUPPLIED Each 5 ml (teaspoonful) of Millipred Oral Solution contains 13.4 mg Prednisolone sodium phosphate (10 mg Prednisolone base) in a grape flavored solution.ndc fl oz (237 ml) bottledispense in tight, light-resistant glass or PET plastic containers as defined in the USP. Store at C (68-77 F). [See USP Controlled Room Temperature]. Keep tightly closed and out of the reach of children. Manufactured for: Zylera Pharmaceuticals Research Triangle Park, NC Manufactured by: Pharmaceutical Associates, Inc. Greenville, SC Rev 06/2013 PRINCIPAL DISPLAY PANEL ML BOTTLE LABEL NDC Millipred (Prednisolone Sodium Phosphate Oral Solution) Equivalent to prednisolone 10 mg/5 mlalcohol FREE / DYE FREE Rx Only 8 fl oz (237 ml)zylera MILL PI
Rayos Prior Authorization Program Summary
 Rayos Prior Authorization Program Summary FDA APPROVED INDICATIONS AND DOSAGE FDA-Approved Indications: 1 Agent Indication Dosage Rayos (prednisone delayedrelease tablet) as an anti-inflammatory or immunosuppressive
Rayos Prior Authorization Program Summary FDA APPROVED INDICATIONS AND DOSAGE FDA-Approved Indications: 1 Agent Indication Dosage Rayos (prednisone delayedrelease tablet) as an anti-inflammatory or immunosuppressive
PRESCRIBING INFORMATION
 PRESCRIBING INFORMATION Pr FLORINEF (fludrocortisone acetate) 0.1 mg Tablets Mineralocorticoid for adrenal insufficiency Paladin Labs Inc. Date of Preparation: 6111 Royalmount Avenue, Suite 102 May 1,
PRESCRIBING INFORMATION Pr FLORINEF (fludrocortisone acetate) 0.1 mg Tablets Mineralocorticoid for adrenal insufficiency Paladin Labs Inc. Date of Preparation: 6111 Royalmount Avenue, Suite 102 May 1,
HYDROCORTISONE 10 MG. Composition Each tablet contains 10 mg hydrocortisone.
 HYDROCORTISONE 10 MG Composition Each tablet contains 10 mg hydrocortisone. Tablets Action Hydrocortisone is the main glucocorticoid secreted by the adrenal cortex. It exhibits antiinflammatory and immunosuppressant
HYDROCORTISONE 10 MG Composition Each tablet contains 10 mg hydrocortisone. Tablets Action Hydrocortisone is the main glucocorticoid secreted by the adrenal cortex. It exhibits antiinflammatory and immunosuppressant
PRESCRIBING INFORMATION. (betamethasone sodium phoshate) Enema 5 mg/100 ml. Topical Corticosteroid
 PRESCRIBING INFORMATION Pr BETNESOL (betamethasone sodium phoshate) Enema 5 mg/100 ml Topical Corticosteroid Paladin Labs Inc. Date of revision: 100 Alexis Nihon Blvd, Suite 600 May 1, 2009 St Laurent,
PRESCRIBING INFORMATION Pr BETNESOL (betamethasone sodium phoshate) Enema 5 mg/100 ml Topical Corticosteroid Paladin Labs Inc. Date of revision: 100 Alexis Nihon Blvd, Suite 600 May 1, 2009 St Laurent,
Pharmacokinetics Absorption Prednisolone is readily and almost completely absorbed from the GI tract after oral administration.
 PREDNITAB Composition Prednitab 5 Tablets Each tablet contains Prednisolone 5 mg. Tablets Prednitab 20 Tablets Each tablet contains Prednisolone 20 mg. Action Prednisolone is a synthetic glucocorticoid
PREDNITAB Composition Prednitab 5 Tablets Each tablet contains Prednisolone 5 mg. Tablets Prednitab 20 Tablets Each tablet contains Prednisolone 20 mg. Action Prednisolone is a synthetic glucocorticoid
Action Dexamethasone is a synthetic corticosteroid (glucocorticoid). As such, its main actions may be grouped as follows:
 DECORT Composition Decort 0.5 mg Tablets Each tablet contains Dexamethasone 0.5 mg. Tablets Decort 0.75 mg Tablets Each tablet contains Dexamethasone 0.75 mg. Decort 2 mg Tablets Each tablet contains Dexamethasone
DECORT Composition Decort 0.5 mg Tablets Each tablet contains Dexamethasone 0.5 mg. Tablets Decort 0.75 mg Tablets Each tablet contains Dexamethasone 0.75 mg. Decort 2 mg Tablets Each tablet contains Dexamethasone
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Rayos) Reference Number: CP.CPA.273 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Rayos) Reference Number: CP.CPA.273 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy for important
Glucocorticoids cause profound and varied metabolic effects. In addition, they modify the body s immune responses to diverse stimuli.
 Cortef hydrocortisone tablets, USP DESCRIPTION CORTEF Tablets contain hydrocortisone which is a glucocorticoid. Glucocorticoids are adrenocortical steroids, both naturally occurring and synthetic, which
Cortef hydrocortisone tablets, USP DESCRIPTION CORTEF Tablets contain hydrocortisone which is a glucocorticoid. Glucocorticoids are adrenocortical steroids, both naturally occurring and synthetic, which
PRESCRIBING INFORMATION. TEVA-PREDNISONE (Prednisone) 5 mg, 50 mg Tablets USP. Corticosteroid
 PRESCRIBING INFORMATION TEVA-PREDNISONE (Prednisone) 5 mg, 50 mg Tablets USP Corticosteroid Teva Canada Ltd. 30 Novopharm Court Toronto, Ontario M1B 2K9 Date of Revision: March 20, 2018 Submission Control
PRESCRIBING INFORMATION TEVA-PREDNISONE (Prednisone) 5 mg, 50 mg Tablets USP Corticosteroid Teva Canada Ltd. 30 Novopharm Court Toronto, Ontario M1B 2K9 Date of Revision: March 20, 2018 Submission Control
METHYLPREDNISOLONE SODIUM SUCCINATE
 451013B/Revised: February 2008 METHYLPREDNISOLONE SODIUM SUCCINATE FOR INJECTION, USP For Intravenous or Intramuscular Administration Rx only DESCRIPTION: Methylprednisolone Sodium Succinate for Injection,
451013B/Revised: February 2008 METHYLPREDNISOLONE SODIUM SUCCINATE FOR INJECTION, USP For Intravenous or Intramuscular Administration Rx only DESCRIPTION: Methylprednisolone Sodium Succinate for Injection,
ENTOFOAM Rectal Foam (Hydrocortisone acetate)
 Published on: 10 Jul 2014 ENTOFOAM Rectal Foam (Hydrocortisone acetate) Composition Each canister contains: Hydrocortisone Acetate, IP. 10% w/w Dosage Form Aerosol rectal foam Pharmacology Pharmacodynamics
Published on: 10 Jul 2014 ENTOFOAM Rectal Foam (Hydrocortisone acetate) Composition Each canister contains: Hydrocortisone Acetate, IP. 10% w/w Dosage Form Aerosol rectal foam Pharmacology Pharmacodynamics
Pharmacology of Corticosteroids
 Pharmacology of Corticosteroids Dr. Aliah Alshanwani Dept. of Pharmacology College of Medicine, KSU Feb 2018 1 The Corticosteroids are steroid hormones produced by the adrenal cortex. They consist of two
Pharmacology of Corticosteroids Dr. Aliah Alshanwani Dept. of Pharmacology College of Medicine, KSU Feb 2018 1 The Corticosteroids are steroid hormones produced by the adrenal cortex. They consist of two
RAYOS (prednisone tablet delayed release) oral tablet
 RAYOS (prednisone tablet delayed release) oral tablet Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan.
RAYOS (prednisone tablet delayed release) oral tablet Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan.
Each MEDROL Tablet for oral administration contains 2 mg, 4 mg, 8 mg, 16 mg or 32 mg of methylprednisolone. Inactive ingredients:
 Medrol methylprednisolone tablets, USP DESCRIPTION MEDROL Tablets contain methylprednisolone which is a glucocorticoid. Glucocorticoids are adrenocortical steroids, both naturally occurring and synthetic,
Medrol methylprednisolone tablets, USP DESCRIPTION MEDROL Tablets contain methylprednisolone which is a glucocorticoid. Glucocorticoids are adrenocortical steroids, both naturally occurring and synthetic,
Composition Decort 4 mg Injection Each ampoule of 1 ml contains Dexamethasone phosphate (as sodium) 4 mg.
 DECORT Composition Decort 4 mg Injection Each ampoule of 1 ml contains Dexamethasone phosphate (as sodium) 4 mg. Ampoule Decort 20 mg Injection Each ampoule of 1 ml contains Dexamethasone phosphate (as
DECORT Composition Decort 4 mg Injection Each ampoule of 1 ml contains Dexamethasone phosphate (as sodium) 4 mg. Ampoule Decort 20 mg Injection Each ampoule of 1 ml contains Dexamethasone phosphate (as
FURAMIST Nasal Spray (Fluticasone furoate )
 Published on: 21 Jan 2016 FURAMIST Nasal Spray (Fluticasone furoate ) Composition Each spray contains: Fluticasone furoate 27.5 mcg Dosage Form Aqueous intranasal spray Pharmacology Pharmacodynamics Fluticasone
Published on: 21 Jan 2016 FURAMIST Nasal Spray (Fluticasone furoate ) Composition Each spray contains: Fluticasone furoate 27.5 mcg Dosage Form Aqueous intranasal spray Pharmacology Pharmacodynamics Fluticasone
SOLU-MEDROL is available in several strengths and packages for intravenous or intramuscular administration.
 Solu-Medrol methylprednisolone sodium succinate for injection, USP For Intravenous or Intramuscular Administration DESCRIPTION SOLU-MEDROL Sterile Powder contains methylprednisolone sodium succinate as
Solu-Medrol methylprednisolone sodium succinate for injection, USP For Intravenous or Intramuscular Administration DESCRIPTION SOLU-MEDROL Sterile Powder contains methylprednisolone sodium succinate as
For topical use only. Not for oral, ophthalmic, or intravaginal use.
 DESOXIMETASONE Ointment USP, 0.25% For topical use only. Not for oral, ophthalmic, or intravaginal use. Rx only DESCRIPTION Desoximetasone ointment USP, 0.25% contains the active synthetic corticosteroid
DESOXIMETASONE Ointment USP, 0.25% For topical use only. Not for oral, ophthalmic, or intravaginal use. Rx only DESCRIPTION Desoximetasone ointment USP, 0.25% contains the active synthetic corticosteroid
Emflaza. (deflazacort) New Product Slideshow
 Emflaza (deflazacort) New Product Slideshow Introduction Brand name: Emflaza Generic name: Deflazacort Pharmacological class: Corticosteroid Strength and Formulation: 6mg, 18mg, 30mg, 36mg tablets; 22.75mg/mL
Emflaza (deflazacort) New Product Slideshow Introduction Brand name: Emflaza Generic name: Deflazacort Pharmacological class: Corticosteroid Strength and Formulation: 6mg, 18mg, 30mg, 36mg tablets; 22.75mg/mL
Prednisolone Sodium Phosphate Ophthalmic Solution USP, 1% (Sterile) Rx only
 Prednisolone Sodium Phosphate Ophthalmic Solution USP, 1% (Sterile) Rx only DESCRIPTION Prednisolone Sodium Phosphate Ophthalmic Solution, 1%, is a sterile solution for ophthalmic administration having
Prednisolone Sodium Phosphate Ophthalmic Solution USP, 1% (Sterile) Rx only DESCRIPTION Prednisolone Sodium Phosphate Ophthalmic Solution, 1%, is a sterile solution for ophthalmic administration having
ROXANE LABORATORIES, INC.
 4070100//02 ROXANE LABORATORIES, INC. PredniSONE TABLETS USP 1, 2.5, 5, 10, 20, or 50 mg, PredniSONE ORAL SOLUTION 5 mg per 5 ml and PredniSONE INTENSOL 5 mg per ml Each tablet contains: Prednisone...
4070100//02 ROXANE LABORATORIES, INC. PredniSONE TABLETS USP 1, 2.5, 5, 10, 20, or 50 mg, PredniSONE ORAL SOLUTION 5 mg per 5 ml and PredniSONE INTENSOL 5 mg per ml Each tablet contains: Prednisone...
FLOMIST Aqueous Nasal Spray (Fluticasone propionate)
 Published on: 10 Jul 2014 FLOMIST Aqueous Nasal Spray (Fluticasone propionate) Composition FLOMIST Aqueous Nasal Spray Each spray delivers: Fluticasone Propionate BP...50 mcg Fluticasone Propionate BP...
Published on: 10 Jul 2014 FLOMIST Aqueous Nasal Spray (Fluticasone propionate) Composition FLOMIST Aqueous Nasal Spray Each spray delivers: Fluticasone Propionate BP...50 mcg Fluticasone Propionate BP...
100 mg ACT-O-VIAL Each 2 ml contains: (when mixed) equiv. to 100 mg. 250 mg ACT-O-VIAL Each 2 ml contains: (when mixed) equiv.
 SOLU-CORTEF (hydrocortisone sodium succinate for injection, USP) For Intravenous or Intramuscular Administration DESCRIPTION SOLU-CORTEF Sterile Powder is an anti-inflammatory glucocorticoid, which contains
SOLU-CORTEF (hydrocortisone sodium succinate for injection, USP) For Intravenous or Intramuscular Administration DESCRIPTION SOLU-CORTEF Sterile Powder is an anti-inflammatory glucocorticoid, which contains
Reference ID:
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use RAYOS safely and effectively. See full prescribing information for RAYOS. RAYOS (prednisone) delayed-release
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use RAYOS safely and effectively. See full prescribing information for RAYOS. RAYOS (prednisone) delayed-release
The formulations containing benzyl alcohol should not be used in neonates.
 SOLU-MEDROL (methylprednisolone sodium succinate for injection, USP) The formulations containing benzyl alcohol should not be used in neonates. For Intravenous or Intramuscular Administration DESCRIPTION
SOLU-MEDROL (methylprednisolone sodium succinate for injection, USP) The formulations containing benzyl alcohol should not be used in neonates. For Intravenous or Intramuscular Administration DESCRIPTION
50 microgram/g Calcipotriol and 500 microgram/g betamethasone (as dipropionate).
 DUPISOR Composition Gel 50 microgram/g Calcipotriol and 500 microgram/g betamethasone (as dipropionate). Action Calcipotriol is a non-steroidal antipsoriatic agent, derived from vitamin D. Calcipotriol
DUPISOR Composition Gel 50 microgram/g Calcipotriol and 500 microgram/g betamethasone (as dipropionate). Action Calcipotriol is a non-steroidal antipsoriatic agent, derived from vitamin D. Calcipotriol
Gastrointestinal diseases To tide the patient over a critical period of the disease in regional enteritis (systemic therapy) and ulcerative colitis.
 SOLU-MEDROL (methylprednisolone sodium succinate for injection, USP) The formulations containing benzyl alcohol should not be used in neonates. For Intravenous or Intramuscular Administration DESCRIPTION
SOLU-MEDROL (methylprednisolone sodium succinate for injection, USP) The formulations containing benzyl alcohol should not be used in neonates. For Intravenous or Intramuscular Administration DESCRIPTION
H.P. Acthar Gel (repository corticotropin) Prior Authorization Criteria Program Summary
 OBJECTIVE The intent of the H.P. Acthar Gel (repository corticotropin) Prior Authorization (PA) Criteria is to appropriately select patients for therapy according to product labeling and/or clinical studies
OBJECTIVE The intent of the H.P. Acthar Gel (repository corticotropin) Prior Authorization (PA) Criteria is to appropriately select patients for therapy according to product labeling and/or clinical studies
Corticosteroids. Abdulmoein Al-Agha, FRCPCH Professor of Pediatric Endocrinology, King Abdulaziz University Hospital,
 Corticosteroids Abdulmoein Al-Agha, FRCPCH Professor of Pediatric Endocrinology, King Abdulaziz University Hospital, http://aagha.kau.edu.sa History 1855 Addison's disease 1856 Adrenal glands essential
Corticosteroids Abdulmoein Al-Agha, FRCPCH Professor of Pediatric Endocrinology, King Abdulaziz University Hospital, http://aagha.kau.edu.sa History 1855 Addison's disease 1856 Adrenal glands essential
WARNING LETTER. ( (ORA05050) for OrapredCI (prednisolone sodium phosphate
 +t-tlserviçès-,& v,ù',' : -::~.,'" ( -ldepartment OF HEALTH & HUMAN SERVICES 'P"~~\- Public Health Service Food and Drug Administration Rockville, MD 20857 TRANSMITTED BY FACSIMILE October 11, 2006 Jean-Jacques
+t-tlserviçès-,& v,ù',' : -::~.,'" ( -ldepartment OF HEALTH & HUMAN SERVICES 'P"~~\- Public Health Service Food and Drug Administration Rockville, MD 20857 TRANSMITTED BY FACSIMILE October 11, 2006 Jean-Jacques
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Prednicare Tablets 1 mg 2. QUALITATIVE AND QUANTITATIVE COMPOSITION mg per tablet Active: Prednisolone 1.0 Other ingredients:
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE VETERINARY MEDICINAL PRODUCT Prednicare Tablets 1 mg 2. QUALITATIVE AND QUANTITATIVE COMPOSITION mg per tablet Active: Prednisolone 1.0 Other ingredients:
KENALOG -10 INJECTION triamcinolone acetonide injectable suspension, USP
 KENALOG -10 INJECTION triamcinolone acetonide injectable suspension, USP NOT FOR USE IN NEONATES CONTAINS BENZYL ALCOHOL For Intra-articular or Intralesional Use Only NOT FOR INTRAVENOUS, INTRAMUSCULAR,
KENALOG -10 INJECTION triamcinolone acetonide injectable suspension, USP NOT FOR USE IN NEONATES CONTAINS BENZYL ALCOHOL For Intra-articular or Intralesional Use Only NOT FOR INTRAVENOUS, INTRAMUSCULAR,
HYDROCORTISONE OINTMENT USP,
 HYDROCORTISONE- hydrocortisone ointment E. Fougera & Co. a division of Fougera Pharmaceuticals Inc. ---------- HYDROCORTISONE OINTMENT USP, 1% Rx only For External Us e Only Not For Ophthalmic Us e DESCRIPTION:
HYDROCORTISONE- hydrocortisone ointment E. Fougera & Co. a division of Fougera Pharmaceuticals Inc. ---------- HYDROCORTISONE OINTMENT USP, 1% Rx only For External Us e Only Not For Ophthalmic Us e DESCRIPTION:
Distribution The in vitro protein binding for Mometasone furoate was reported to be 98% to 99% in concentra on range of 5 to 500 ng/ml.
 NOSATREX Composition Mometasone Furoate 0.05% w/w Spray Action Mechanism of Action Mometasone Nasal Spray 50 mcg is a cor costeroid demonstra ng potent an -inflammatory properties. The precise mechanism
NOSATREX Composition Mometasone Furoate 0.05% w/w Spray Action Mechanism of Action Mometasone Nasal Spray 50 mcg is a cor costeroid demonstra ng potent an -inflammatory properties. The precise mechanism
PRODUCT NAME: SOLONE
 NAME OF THE MEDICINE Prednisolone Structural formula: PRODUCT INFORMATION PRODUCT NAME: SOLONE Chemical name: 11, 17,21-Trihydroxypregna 1,4-diene-3,20-dione Cas No. 50-24-8 (Anhydrous) DESCRIPTION Prednisolone
NAME OF THE MEDICINE Prednisolone Structural formula: PRODUCT INFORMATION PRODUCT NAME: SOLONE Chemical name: 11, 17,21-Trihydroxypregna 1,4-diene-3,20-dione Cas No. 50-24-8 (Anhydrous) DESCRIPTION Prednisolone
Adrenal Pharmacology
 Adrenal Pharmacology Pharmacology Team Naim Kittana, Suhaib Hattab, Ansam Sawalha, Adham Abu Taha, Waleed Sweileh, Ramzi Shawahneh Faculty of Medicine & Health Sciences An-Najah National University 1 Steroidal
Adrenal Pharmacology Pharmacology Team Naim Kittana, Suhaib Hattab, Ansam Sawalha, Adham Abu Taha, Waleed Sweileh, Ramzi Shawahneh Faculty of Medicine & Health Sciences An-Najah National University 1 Steroidal
PRESCRIBING INFORMATION DEXAMETHASONE OMEGA UNIDOSE. (Dexamethasone Sodium Phosphate Injection USP) (10 mg/ml) Sterile.
 PRESCRIBING INFORMATION Pr DEXAMETHASONE OMEGA UNIDOSE (10 mg/ml) Sterile Corticosteroid Omega Laboratories Limited 11,177 Hamon Montreal, Quebec, Canada H3M 3E4 Date of preparation: June 12, 2012 Control
PRESCRIBING INFORMATION Pr DEXAMETHASONE OMEGA UNIDOSE (10 mg/ml) Sterile Corticosteroid Omega Laboratories Limited 11,177 Hamon Montreal, Quebec, Canada H3M 3E4 Date of preparation: June 12, 2012 Control
Prescribing Information
 Prescribing Information Pr DERMOVATE Cream (clobetasol propionate cream, USP) Pr DERMOVATE Ointment (clobetasol propionate ointment, USP) Topical corticosteroid TaroPharma Preparation Date: A Division
Prescribing Information Pr DERMOVATE Cream (clobetasol propionate cream, USP) Pr DERMOVATE Ointment (clobetasol propionate ointment, USP) Topical corticosteroid TaroPharma Preparation Date: A Division
KENALOG -40 INJECTION (triamcinolone acetonide injectable suspension, USP)
 KENALOG -40 INJECTION (triamcinolone acetonide injectable suspension, USP) NOT FOR USE IN NEONATES CONTAINS BENZYL ALCOHOL For Intramuscular or Intra-articular Use Only NOT FOR INTRAVENOUS, INTRADERMAL,
KENALOG -40 INJECTION (triamcinolone acetonide injectable suspension, USP) NOT FOR USE IN NEONATES CONTAINS BENZYL ALCOHOL For Intramuscular or Intra-articular Use Only NOT FOR INTRAVENOUS, INTRADERMAL,
Summary of Product Characteristics
 Summary of Product Characteristics 1 NAME OF THE MEDICINAL PRODUCT Prednesol 5mg Tablets. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 5 mg of prednisolone as Prednisolone Sodium Phosphate.
Summary of Product Characteristics 1 NAME OF THE MEDICINAL PRODUCT Prednesol 5mg Tablets. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 5 mg of prednisolone as Prednisolone Sodium Phosphate.
Package Insert. Pr Hydrocortisone Sodium Succinate for Injection BP
 page 1 of 7 Sterile Powder Pr Hydrocortisone Sodium Succinate for Injection BP Glucocorticoid ACTION AND CLINICAL PHARMACOLOGY Hydrocortisone sodium succinate is a corticosteroid, and a highly water soluble
page 1 of 7 Sterile Powder Pr Hydrocortisone Sodium Succinate for Injection BP Glucocorticoid ACTION AND CLINICAL PHARMACOLOGY Hydrocortisone sodium succinate is a corticosteroid, and a highly water soluble
Cordran Cream and Cordran Ointment Flurandrenolide, USP
 Cordran Cream and Cordran Ointment Flurandrenolide, USP DESCRIPTION Cordran (flurandrenolide, USP) is a potent corticosteroid intended for topical use. Flurandrenolide occurs as white to off-white, fluffy,
Cordran Cream and Cordran Ointment Flurandrenolide, USP DESCRIPTION Cordran (flurandrenolide, USP) is a potent corticosteroid intended for topical use. Flurandrenolide occurs as white to off-white, fluffy,
Prescribing Information. Taro-Clobetasol. Taro-Clobetasol
 Prescribing Information Pr Taro-Clobetasol Clobetasol Propionate Cream USP, 0.05% w/w Pr Taro-Clobetasol Clobetasol Propionate Ointment USP, 0.05% w/w Therapeutic Classification Topical corticosteroid
Prescribing Information Pr Taro-Clobetasol Clobetasol Propionate Cream USP, 0.05% w/w Pr Taro-Clobetasol Clobetasol Propionate Ointment USP, 0.05% w/w Therapeutic Classification Topical corticosteroid
MEDROLONE HIKMA PHARMACEUTICALS
 09-15 MEDROLONE HIKMA PHARMACEUTICALS Sterile powder 40 mg Methylprednisolone PROPERTIES This product is an I.M. and I.V. injectable form of methylprednisolone, a synthetic glucocorticosteroid. This highly
09-15 MEDROLONE HIKMA PHARMACEUTICALS Sterile powder 40 mg Methylprednisolone PROPERTIES This product is an I.M. and I.V. injectable form of methylprednisolone, a synthetic glucocorticosteroid. This highly
Each gram of the ointment contains 0.25 mg Fluocinolone Acetonide in a base containing White Petrolatum.
 FLUOCINOLONE ACETONIDE- fluocinolone acetonide ointment E. Fougera & Co. a division of Fougera Pharmaceuticals Inc. ---------- FLUOCINOLONE ACETONIDE OINTMENT, USP, 0.025% For Topical Us e Only. Not for
FLUOCINOLONE ACETONIDE- fluocinolone acetonide ointment E. Fougera & Co. a division of Fougera Pharmaceuticals Inc. ---------- FLUOCINOLONE ACETONIDE OINTMENT, USP, 0.025% For Topical Us e Only. Not for
Salapin: Salbutamol BP 2mg as sulphate in each 5mL of a raspberry cola flavoured, sugar free syrup.
 Salapin Salbutamol Syrup 2mg/5mL Qualitative and quantitative composition Salapin: Salbutamol BP 2mg as sulphate in each 5mL of a raspberry cola flavoured, sugar free syrup. Clinical particulars Therapeutic
Salapin Salbutamol Syrup 2mg/5mL Qualitative and quantitative composition Salapin: Salbutamol BP 2mg as sulphate in each 5mL of a raspberry cola flavoured, sugar free syrup. Clinical particulars Therapeutic
DOSING GUIDE. Indications. Important Safety Information. Enable the immune system. RECOGNIZE. RESPOND.
 DOSING GUIDE For patients with unresectable Stage III NSCLC following concurrent CRT For patients with locally advanced or metastatic UC previously treated with platinum-based therapy Enable the immune
DOSING GUIDE For patients with unresectable Stage III NSCLC following concurrent CRT For patients with locally advanced or metastatic UC previously treated with platinum-based therapy Enable the immune
SYNACTHEN DEPOT i.m. tetracosactide hexaacetate
 SYNACTHEN DEPOT i.m. tetracosactide hexaacetate 1 mg/ml suspension for injection New Zealand Consumer Medicine Information What is in this leaflet Read all of this leaflet carefully before you are given
SYNACTHEN DEPOT i.m. tetracosactide hexaacetate 1 mg/ml suspension for injection New Zealand Consumer Medicine Information What is in this leaflet Read all of this leaflet carefully before you are given
PRESCRIBING INFORMATION
 PRESCRIBING INFORMATION ratio-dexamethasone Dexamethasone, USP 0.5 and 4 mg tablets Corticosteroid Teva Canada Limited 30 Novopharm Court Toronto, Canada M1B 2K9 DATE OF PREPARATION: February 26, 2013
PRESCRIBING INFORMATION ratio-dexamethasone Dexamethasone, USP 0.5 and 4 mg tablets Corticosteroid Teva Canada Limited 30 Novopharm Court Toronto, Canada M1B 2K9 DATE OF PREPARATION: February 26, 2013
The CAS number is and the molecular weight is
 PRODUCT INFORMATION SOLU-CORTEF (hydrocortisone sodium succinate) NAME OF THE MEDICINE Non-proprietary name: hydrocortisone sodium succinate Chemical structure: The CAS number is 125-04-2 and the molecular
PRODUCT INFORMATION SOLU-CORTEF (hydrocortisone sodium succinate) NAME OF THE MEDICINE Non-proprietary name: hydrocortisone sodium succinate Chemical structure: The CAS number is 125-04-2 and the molecular
Summary of Product Characteristics
 1 NAME OF THE MEDICINAL PRODUCT Synacthen 250 micrograms/ml solution for injection. Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Active Substance 250 micrograms of Tetracosactide.
1 NAME OF THE MEDICINAL PRODUCT Synacthen 250 micrograms/ml solution for injection. Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Active Substance 250 micrograms of Tetracosactide.
Corticosteroids. Hawler Medical University College of Medicine Department of Pharmacology and Biophysics Dr.Susan Abdulkadir Farhadi MSc Pharmacology
 Corticosteroids Hawler Medical University College of Medicine Department of Pharmacology and Biophysics Dr.Susan Abdulkadir Farhadi MSc Pharmacology Objectives By the end of this lecture you should be
Corticosteroids Hawler Medical University College of Medicine Department of Pharmacology and Biophysics Dr.Susan Abdulkadir Farhadi MSc Pharmacology Objectives By the end of this lecture you should be
PROSTIGMIN (neostigmine bromide)
 PROSTIGMIN (neostigmine bromide) TABLETS DESCRIPTION: Prostigmin (neostigmine bromide), an anticholinesterase agent, is available for oral administration in 15 mg tablets. Each tablet also contains gelatin,
PROSTIGMIN (neostigmine bromide) TABLETS DESCRIPTION: Prostigmin (neostigmine bromide), an anticholinesterase agent, is available for oral administration in 15 mg tablets. Each tablet also contains gelatin,
Patients recovered more quickly and completely with ACTH gel compared to placebo during the 30-day treatment period* 1
 In one study of MS patients treated for optic neuritis Patients recovered more quickly and completely with ACTH gel compared to placebo during the 30-day treatment period* 1 PERCENTAGE OF PATIENTS WITH
In one study of MS patients treated for optic neuritis Patients recovered more quickly and completely with ACTH gel compared to placebo during the 30-day treatment period* 1 PERCENTAGE OF PATIENTS WITH
New Zealand Data Sheet
 New Zealand Data Sheet Prednisolone-AFT 1% Prednisolone acetate (Ph Eur) 1% w/v ophthalmic suspension Presentation Prednisolone-AFT 1% is a milky white suspension in an eyedropper bottle for ophthalmic
New Zealand Data Sheet Prednisolone-AFT 1% Prednisolone acetate (Ph Eur) 1% w/v ophthalmic suspension Presentation Prednisolone-AFT 1% is a milky white suspension in an eyedropper bottle for ophthalmic
Chemically, clobetasol propionate is 21-chloro-9-fluoro,11β,17-dihydroxy-16βmethylpregna-1,4-diene-3,20-dione
 Clobetasol Propionate Cream USP, 0.05% FOR TOPICAL DERMATOLOGIC USE ONLY NOT FOR OPHTHALMIC, ORAL, OR INTRAVAGINAL USE. DESCRIPTION Clobetasol propionate cream USP, 0.05% contain the active compound clobetasol
Clobetasol Propionate Cream USP, 0.05% FOR TOPICAL DERMATOLOGIC USE ONLY NOT FOR OPHTHALMIC, ORAL, OR INTRAVAGINAL USE. DESCRIPTION Clobetasol propionate cream USP, 0.05% contain the active compound clobetasol
Core Safety Profile. Pharmaceutical form(s)/strength: Dry Powder Inhaler, Nebuliser Suspension, pressurised Metered Dose Inhaler (pmdi)
 Core Safety Profile Active substance: Budesonide Pharmaceutical form(s)/strength: Dry Powder Inhaler, Nebuliser Suspension, pressurised Metered Dose Inhaler (pmdi) P - RMS: DK/H/PSUR/0041/001 Date of FAR:
Core Safety Profile Active substance: Budesonide Pharmaceutical form(s)/strength: Dry Powder Inhaler, Nebuliser Suspension, pressurised Metered Dose Inhaler (pmdi) P - RMS: DK/H/PSUR/0041/001 Date of FAR:
PRESCRIBING INFORMATION. Triamcinolone Acetonide Injectable Suspension, (U.S.P); 40 mg/ml
 1 PRESCRIBING INFORMATION Pr KENALOG -40 INJECTION Triamcinolone Acetonide Injectable Suspension, (U.S.P); 40 mg/ml THERAPEUTIC CLASSIFICATION Corticosteroid NOTE: KENALOG-40 INJECTION (triamcinolone acetonide)
1 PRESCRIBING INFORMATION Pr KENALOG -40 INJECTION Triamcinolone Acetonide Injectable Suspension, (U.S.P); 40 mg/ml THERAPEUTIC CLASSIFICATION Corticosteroid NOTE: KENALOG-40 INJECTION (triamcinolone acetonide)
WARNING: TENDON EFFECTS and EXACERBATION OF MYASTHENIA GRAVIS
 DECLESAU (dergrafloxacin) tablets, for oral use DECLESAU (dergrafloxacin) injection, solution for intravenous use WARNING: TENDON EFFECTS and EXACERBATION OF MYASTHENIA GRAVIS Fluoroquinolones, including
DECLESAU (dergrafloxacin) tablets, for oral use DECLESAU (dergrafloxacin) injection, solution for intravenous use WARNING: TENDON EFFECTS and EXACERBATION OF MYASTHENIA GRAVIS Fluoroquinolones, including
Revised: 03/2018. *Sections or subsections omitted from the full prescribing information are not listed.
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DIPROLENE Ointment safely and effectively. See full prescribing information for DIPROLENE Ointment.
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DIPROLENE Ointment safely and effectively. See full prescribing information for DIPROLENE Ointment.
When necessary, ph was adjusted with sodium hydroxide and/or hydrochloric acid.
 DEPO-MEDROL (methylprednisolone acetate injectable suspension, USP) Single-Dose Vial Not For Intravenous Use DESCRIPTION DEPO-MEDROL is an anti-inflammatory glucocorticoid for intramuscular, intra-articular,
DEPO-MEDROL (methylprednisolone acetate injectable suspension, USP) Single-Dose Vial Not For Intravenous Use DESCRIPTION DEPO-MEDROL is an anti-inflammatory glucocorticoid for intramuscular, intra-articular,
When necessary, ph was adjusted with sodium hydroxide and/or hydrochloric acid.
 DEPO-MEDROL (methylprednisolone acetate injectable suspension, USP) Single-Dose Vial Not For Intravenous Use DESCRIPTION DEPO-MEDROL is an anti-inflammatory glucocorticoid for intramuscular, intra-articular,
DEPO-MEDROL (methylprednisolone acetate injectable suspension, USP) Single-Dose Vial Not For Intravenous Use DESCRIPTION DEPO-MEDROL is an anti-inflammatory glucocorticoid for intramuscular, intra-articular,
CORTISPORIN Cream (neomycin and polymyxin B sulfates and hydrocortisone acetate cream, USP)
 CORTISPORIN Cream (neomycin and polymyxin B sulfates and hydrocortisone acetate cream, USP) DESCRIPTION CORTISPORIN Cream (neomycin and polymyxin B sulfates and hydrocortisone acetate cream, USP) is a
CORTISPORIN Cream (neomycin and polymyxin B sulfates and hydrocortisone acetate cream, USP) DESCRIPTION CORTISPORIN Cream (neomycin and polymyxin B sulfates and hydrocortisone acetate cream, USP) is a
PRESCRIBING INFORMATION. (Triamcinolone Acetonide Injectable Suspension, (U.S.P); 10 mg/ml
 1 PRESCRIBING INFORMATION Pr KENALOG -10 INJECTION (Triamcinolone Acetonide Injectable Suspension, (U.S.P); 10 mg/ml THERAPEUTIC CLASSIFICATION Corticosteroid NOTE: KENALOG-10 INJECTION is triamcinolone
1 PRESCRIBING INFORMATION Pr KENALOG -10 INJECTION (Triamcinolone Acetonide Injectable Suspension, (U.S.P); 10 mg/ml THERAPEUTIC CLASSIFICATION Corticosteroid NOTE: KENALOG-10 INJECTION is triamcinolone
CLINICAL GUIDELINES. Summary of Literature and Recommendations Concerning Immunization and Steroid Injections Thomas J. Gilbert M.D., M.P.P.
 CLINICAL GUIDELINES Summary of Literature and Recommendations Concerning Immunization and Steroid Injections Thomas J. Gilbert M.D., M.P.P. 11/2/15 Several practices routinely delay steroid injections
CLINICAL GUIDELINES Summary of Literature and Recommendations Concerning Immunization and Steroid Injections Thomas J. Gilbert M.D., M.P.P. 11/2/15 Several practices routinely delay steroid injections
Action Naproxen sodium, the active principle of Naproxan, has been developed as an analgesic because it is more rapidly absorbed than Naproxen.
 NAPROXAN Composition Each tablet contains Naproxen sodium 275 mg. Tablets Action Naproxen sodium, the active principle of Naproxan, has been developed as an analgesic because it is more rapidly absorbed
NAPROXAN Composition Each tablet contains Naproxen sodium 275 mg. Tablets Action Naproxen sodium, the active principle of Naproxan, has been developed as an analgesic because it is more rapidly absorbed
When necessary, ph was adjusted with sodium hydroxide and/or hydrochloric acid.
 DEPO-MEDROL (methylprednisolone acetate injectable suspension, USP) NOT FOR USE IN NEONATES CONTAINS BENZYL ALCOHOL Not For Intravenous Use DESCRIPTION DEPO-MEDROL is an anti-inflammatory glucocorticoid
DEPO-MEDROL (methylprednisolone acetate injectable suspension, USP) NOT FOR USE IN NEONATES CONTAINS BENZYL ALCOHOL Not For Intravenous Use DESCRIPTION DEPO-MEDROL is an anti-inflammatory glucocorticoid
Shock unresponsive to conventional therapy if adrenocortical insufficiency exists or is suspected
 DEXAMETHASONE SODIUM PHOSPHATE - dexamethasone sodium phosphate injection Baxter Healthcare Corporation DESCRIPTION Dexamethasone sodium phosphate, a synthetic adrenocortical steroid, is a white or slightly
DEXAMETHASONE SODIUM PHOSPHATE - dexamethasone sodium phosphate injection Baxter Healthcare Corporation DESCRIPTION Dexamethasone sodium phosphate, a synthetic adrenocortical steroid, is a white or slightly
PRESCRIBING INFORMATION
 PRESCRIBING INFORMATION pdp-pyrazinamide Pyrazinamide Tablets, USP 500 mg Antimycobacterial / Antituberculosis Agent PENDOPHARM, Division of Pharmascience Inc.. 6111, Royalmount Ave, Suite 100 Montréal,
PRESCRIBING INFORMATION pdp-pyrazinamide Pyrazinamide Tablets, USP 500 mg Antimycobacterial / Antituberculosis Agent PENDOPHARM, Division of Pharmascience Inc.. 6111, Royalmount Ave, Suite 100 Montréal,
PROFESSIONAL INFORMATION
 SCHEDULING STATUS: S1 PROPRIETARY NAME AND DOSAGE FORM: Aleve Tablets COMPOSITION: Each tablet contains naproxen sodium 220 mg (equivalent to 200 mg naproxen) CATEGORY AND CLASS: A / 2.7 Antipyretic or
SCHEDULING STATUS: S1 PROPRIETARY NAME AND DOSAGE FORM: Aleve Tablets COMPOSITION: Each tablet contains naproxen sodium 220 mg (equivalent to 200 mg naproxen) CATEGORY AND CLASS: A / 2.7 Antipyretic or
Children Enteric coated tablet : 1-3 mg/kg per day in divided doses.
 Ultrafen Tablet/SR Tablet/Suppository/Gel Description Ultrafen is a preparation of Diclofenac is a non-steroidal antiinflammatory agent with marked analgesic, anti-inflammatory and antipyretic properties.
Ultrafen Tablet/SR Tablet/Suppository/Gel Description Ultrafen is a preparation of Diclofenac is a non-steroidal antiinflammatory agent with marked analgesic, anti-inflammatory and antipyretic properties.
Leucovorin Calcium Tablets USP, 5 mg, 10 mg, 15 mg, and 25 mg
 Leucovorin Calcium Tablets USP, 5 mg, 10 mg, 15 mg, and 25 mg Rx only DESCRIPTION Leucovorin Calcium Tablets USP contain either 5 mg, 10 mg, 15 mg or 25 mg leucovorin as the calcium salt of N-[4-[[(2-amino-5-formyl-1,4,5,6,7,8-hexahydro-4-oxo-6-pteridinyl)methyl]
Leucovorin Calcium Tablets USP, 5 mg, 10 mg, 15 mg, and 25 mg Rx only DESCRIPTION Leucovorin Calcium Tablets USP contain either 5 mg, 10 mg, 15 mg or 25 mg leucovorin as the calcium salt of N-[4-[[(2-amino-5-formyl-1,4,5,6,7,8-hexahydro-4-oxo-6-pteridinyl)methyl]
AUSTRALIAN PRODUCT INFORMATION FLAREX (FLUOROMETHOLONE ACETATE) EYE DROPS SUSPENSION
 AUSTRALIAN PRODUCT INFORMATION FLAREX (FLUOROMETHOLONE ACETATE) EYE DROPS SUSPENSION 1 NAME OF THE MEDICINE Fluorometholone acetate. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION The active ingredient in
AUSTRALIAN PRODUCT INFORMATION FLAREX (FLUOROMETHOLONE ACETATE) EYE DROPS SUSPENSION 1 NAME OF THE MEDICINE Fluorometholone acetate. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION The active ingredient in
INVELTYS (loteprednol etabonate ophthalmic suspension) 1%, for topical ophthalmic use Initial U.S. Approval: 1998
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use INVELTYS safely and effectively. See full prescribing information for INVELTYS. INVELTYS (loteprednol
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use INVELTYS safely and effectively. See full prescribing information for INVELTYS. INVELTYS (loteprednol
Spironolactone has not been demonstrated to elevate serum uric acid, to precipitate gout or to alter carbohydrate metabolism.
 SPIRONE Composition Each tablet contains Spironolactone 100 mg. Tablets Action Spironolactone is a specific pharmacologic antagonist of aldosterone, acting primarily through competitive binding of receptors
SPIRONE Composition Each tablet contains Spironolactone 100 mg. Tablets Action Spironolactone is a specific pharmacologic antagonist of aldosterone, acting primarily through competitive binding of receptors
INDICATIONS For steroid responsive inflammation of the palpebral and bulbar conjunctiva, cornea, and anterior segment of the eye globe.
 Page 1 of 5 SCHEDULING STATUS Schedule 4 PROPRIETARY NAME AND DOSAGE FORM PRED FORTE Sterile Eye Suspension COMPOSITION PRED FORTE Sterile Eye Suspension contains: Prednisolone acetate 10 mg/ml Preservative:
Page 1 of 5 SCHEDULING STATUS Schedule 4 PROPRIETARY NAME AND DOSAGE FORM PRED FORTE Sterile Eye Suspension COMPOSITION PRED FORTE Sterile Eye Suspension contains: Prednisolone acetate 10 mg/ml Preservative:
PREDNISOLONE SODIUM PHOSPHATE- prednis olone s odium phos phate tablet, orally
 PREDNISOLONE SODIUM PHOSPHATE ODT- prednis olone s odium phos phate tablet, orally disintegrating PREDNISOLONE SODIUM PHOSPHATE- prednis olone s odium phos phate tablet, orally disintegrating Pras co Laboratories
PREDNISOLONE SODIUM PHOSPHATE ODT- prednis olone s odium phos phate tablet, orally disintegrating PREDNISOLONE SODIUM PHOSPHATE- prednis olone s odium phos phate tablet, orally disintegrating Pras co Laboratories
See 17 for PATIENT COUNSELING INFORMATION. Revised: 03/2018
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DIPROLENE Lotion safely and effectively. See full prescribing information for DIPROLENE Lotion. DIPROLENE
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DIPROLENE Lotion safely and effectively. See full prescribing information for DIPROLENE Lotion. DIPROLENE
TOPCORT Cream/Ointment (Mometasone furoate 0.1%)
 Published on: 10 Jul 2014 TOPCORT Cream/Ointment (Mometasone furoate 0.1%) Composition TOPCORT Cream Mometasone Furoate, IP... 0.1% w/w In a cream base... q.s. TOPCORT Ointment Mometasone Furoate, IP...
Published on: 10 Jul 2014 TOPCORT Cream/Ointment (Mometasone furoate 0.1%) Composition TOPCORT Cream Mometasone Furoate, IP... 0.1% w/w In a cream base... q.s. TOPCORT Ointment Mometasone Furoate, IP...
SCHEDULING STATUS Schedule 4 PROPRIETARY NAME AND DOSAGE FORM
 Page 1 of 5 SCHEDULING STATUS Schedule 4 PROPRIETARY NAME AND DOSAGE FORM FML Liquifilm Sterile Eye Suspension COMPOSITION FML Liquifilm Sterile Eye Suspension contains: Fluorometholone 1,0 mg/ml Liquifilm
Page 1 of 5 SCHEDULING STATUS Schedule 4 PROPRIETARY NAME AND DOSAGE FORM FML Liquifilm Sterile Eye Suspension COMPOSITION FML Liquifilm Sterile Eye Suspension contains: Fluorometholone 1,0 mg/ml Liquifilm
Learn how to identify multiple sclerosis (MS) relapses, when to report them, and about a treatment option to discuss with your doctor.
 Learn how to identify multiple sclerosis (MS) relapses, when to report them, and about a treatment option to discuss with your doctor. Actor portrayal. Actor portrayal. Ask your doctor about Acthar, A
Learn how to identify multiple sclerosis (MS) relapses, when to report them, and about a treatment option to discuss with your doctor. Actor portrayal. Actor portrayal. Ask your doctor about Acthar, A
DIPROLENE AF (augmented betamethasone dipropionate) Cream, 0.05% for topical use Initial U.S. Approval: 1983
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DIPROLENE AF Cream safely and effectively. See full prescribing information for DIPROLENE AF Cream.
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use DIPROLENE AF Cream safely and effectively. See full prescribing information for DIPROLENE AF Cream.
Chapter 39. Adrenocorticosteroids
 Chapter 39 Adrenocorticosteroids - The adrenal glands are located on the top of both kidneys. - Each adrenal gland consists of medulla and cortex. Adrenal cortex corticoid Glucocorticoid, GC Mineralocorticoid
Chapter 39 Adrenocorticosteroids - The adrenal glands are located on the top of both kidneys. - Each adrenal gland consists of medulla and cortex. Adrenal cortex corticoid Glucocorticoid, GC Mineralocorticoid
What prescribers need to know
 HUMIRA Citrate-free presentations in an Electronic Medical Record (EMR) What prescribers need to know 2 / This is your guide to identifying HUMIRA Citrate-free presentations in your Electronic Medical
HUMIRA Citrate-free presentations in an Electronic Medical Record (EMR) What prescribers need to know 2 / This is your guide to identifying HUMIRA Citrate-free presentations in your Electronic Medical
D90 (27/10/2005) Final SmPC NL/H/653/01
 1/6 1. NAME OF THE MEDICINAL PRODUCT MONOFREE DEXAMETHASON 1 mg/ml, eye drops, solution 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml solution contains 1 mg of dexamethasone phosphate as dexamethasone
1/6 1. NAME OF THE MEDICINAL PRODUCT MONOFREE DEXAMETHASON 1 mg/ml, eye drops, solution 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 ml solution contains 1 mg of dexamethasone phosphate as dexamethasone
PRESCRIBING INFORMATION. ratio-topisone. Betamethasone dipropionate USP. 0.5 mg Cream, Ointment and Lotion. Topical corticosteroid
 PRESCRIBING INFORMATION ratio-topisone Betamethasone dipropionate USP 0.5 mg Cream, Ointment and Lotion Topical corticosteroid Teva Canada Limited Date of Revision 30 Novopharm Court January 21, 2013 Toronto,
PRESCRIBING INFORMATION ratio-topisone Betamethasone dipropionate USP 0.5 mg Cream, Ointment and Lotion Topical corticosteroid Teva Canada Limited Date of Revision 30 Novopharm Court January 21, 2013 Toronto,
NOTOPAIN CAPLETS. Diclofenac Sodium + Paracetamol. Composition. Each tablet contains: Diclofenac Sodium BP 50mg Paracetamol BP 500mg.
 NOTOPAIN CAPLETS Diclofenac Sodium + Paracetamol Composition Each tablet contains: Diclofenac Sodium BP 50mg Paracetamol BP 500mg Pharmacology Phamacodynamics Diclofenac relieves pain and inflammation
NOTOPAIN CAPLETS Diclofenac Sodium + Paracetamol Composition Each tablet contains: Diclofenac Sodium BP 50mg Paracetamol BP 500mg Pharmacology Phamacodynamics Diclofenac relieves pain and inflammation
CORTISPORIN Ointment (neomycin and polymyxin B sulfates, bacitracin zinc, and. hydrocortisone ointment, USP)
 CORTISPORIN Ointment (neomycin and polymyxin B sulfates, bacitracin zinc, and hydrocortisone ointment, USP) DESCRIPTION CORTISPORIN Ointment (neomycin and polymyxin B sulfates, bacitracin zinc, and hydrocortisone
CORTISPORIN Ointment (neomycin and polymyxin B sulfates, bacitracin zinc, and hydrocortisone ointment, USP) DESCRIPTION CORTISPORIN Ointment (neomycin and polymyxin B sulfates, bacitracin zinc, and hydrocortisone
Abiraterone Acetate is an antiandrogen used in the treatment of Castration-Resistant Prostate Cancer(CRPC).
 For the use only of an Oncologist or a Hospital or a Laboratory ABIRATERONE ACETATE TABLETS Zabiteron-250 COMPOSITION Abiraterone Acetate Tablets 250mg Each uncoated tablets contains: Abiraterone Acetate
For the use only of an Oncologist or a Hospital or a Laboratory ABIRATERONE ACETATE TABLETS Zabiteron-250 COMPOSITION Abiraterone Acetate Tablets 250mg Each uncoated tablets contains: Abiraterone Acetate
Adrenocorticosteroids
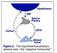 Adrenocorticosteroids Divided into those having: 1. Important effects on intermediary metabolism and immune function = glucocorticoids (cortisol). 2. Salt-retaining activity = mineralocorticoids (aldosterone).
Adrenocorticosteroids Divided into those having: 1. Important effects on intermediary metabolism and immune function = glucocorticoids (cortisol). 2. Salt-retaining activity = mineralocorticoids (aldosterone).
January 2008 IMPORTANT DRUG WARNING
 January 2008 IMPORTANT DRUG WARNING Dear Healthcare Professional: Roche would like to advise you of a recent update to the PEGASYS (Peginterferon alfa-2a) and COPEGUS (Ribavirin, USP) package inserts.
January 2008 IMPORTANT DRUG WARNING Dear Healthcare Professional: Roche would like to advise you of a recent update to the PEGASYS (Peginterferon alfa-2a) and COPEGUS (Ribavirin, USP) package inserts.
3 DOSAGE FORMS AND STRENGTHS
 PATADAY- olopatadine hydrochloride solution/ drops Alcon Laboratories, Inc. ---------- HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PATADAY safely
PATADAY- olopatadine hydrochloride solution/ drops Alcon Laboratories, Inc. ---------- HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use PATADAY safely
PACKAGE INSERT TEMPLATE FOR SALBUTAMOL TABLET & SALBUTAMOL SYRUP
 PACKAGE INSERT TEMPLATE FOR SALBUTAMOL TABLET & SALBUTAMOL SYRUP Brand or Product Name [Product name] Tablet 2mg [Product name] Tablet 4mg [Product name] Syrup 2mg/5ml Name and Strength of Active Substance(s)
PACKAGE INSERT TEMPLATE FOR SALBUTAMOL TABLET & SALBUTAMOL SYRUP Brand or Product Name [Product name] Tablet 2mg [Product name] Tablet 4mg [Product name] Syrup 2mg/5ml Name and Strength of Active Substance(s)
PRODUCT LEAFLET: USER INFORMATION HYDROCORTISONE ERFA 20 MG TABLETS. Hydrocortisone
 PRODUCT LEAFLET: USER INFORMATION HYDROCORTISONE ERFA 20 MG TABLETS Hydrocortisone Read this leaflet carefully before you start taking this medicine; it contains important information. - Keep this leaflet.
PRODUCT LEAFLET: USER INFORMATION HYDROCORTISONE ERFA 20 MG TABLETS Hydrocortisone Read this leaflet carefully before you start taking this medicine; it contains important information. - Keep this leaflet.
Chemical Names: Prednisolone acetate: 11ß,17,21-Trihydroxypregna-1,4-diene-3,20-dione 21-acetate.
 PRED-G (gentamicin and prednisolone acetate ophthalmic suspension, USP) 0.3%/1% sterile DESCRIPTION PRED-G sterile ophthalmic suspension is a topical anti-inflammatory/anti-infective combination product
PRED-G (gentamicin and prednisolone acetate ophthalmic suspension, USP) 0.3%/1% sterile DESCRIPTION PRED-G sterile ophthalmic suspension is a topical anti-inflammatory/anti-infective combination product
OVERALL CONDITION IMPROVEMENT compared to
 DEMONSTRATED EFFICACY Actor portrayal. IN A RANDOMIZED, DOUBLE-BLIND, PLACEBO-CONTROLLED CLINICAL STUDY, ACTHAR DELIVERED MORE RAPID RESOLUTION OF RELAPSE VS PLACEBO 70% ACTHAR- TREATED OVERALL CONDITION
DEMONSTRATED EFFICACY Actor portrayal. IN A RANDOMIZED, DOUBLE-BLIND, PLACEBO-CONTROLLED CLINICAL STUDY, ACTHAR DELIVERED MORE RAPID RESOLUTION OF RELAPSE VS PLACEBO 70% ACTHAR- TREATED OVERALL CONDITION
Levocetirizine dihydrochloride
 INSERT TEXT UAP Levocetirizine dihydrochloride Allerzet 5 mg Tablet Antihistamine FORMULATION Each film-coated tablet contains: Levocetirizine dihydrochloride.. 5 mg PRODUCT DESCRIPTION Levocetirine 5
INSERT TEXT UAP Levocetirizine dihydrochloride Allerzet 5 mg Tablet Antihistamine FORMULATION Each film-coated tablet contains: Levocetirizine dihydrochloride.. 5 mg PRODUCT DESCRIPTION Levocetirine 5
Monocast Description Indications
 Monocast Tablet Description The active ingredient of Monocast tablet is Montelukast Sodium INN. Montelukast is a selective and orally active leukotriene receptor antagonist that inhibits the cysteinyl
Monocast Tablet Description The active ingredient of Monocast tablet is Montelukast Sodium INN. Montelukast is a selective and orally active leukotriene receptor antagonist that inhibits the cysteinyl
