Pharmacy and Therapeutics Committee
|
|
|
- Christopher Gilmore
- 5 years ago
- Views:
Transcription
1 Michigan Department of Health and Human Services Pharmacy and Therapeutics Committee December 13, 2016 Minutes Attendees: Dr. Tutag Lehr, Andrew Mac, James Miller, Brian Peltz, Dr. Anthony Ognjan, Dr. Steve Hadesman (by phone), Dr. Brad Uren, Dr. Jayne Courts, Dr. Venkat Rao, Dr. Eggleston. A quorum was met. The MDHHS P & T committee was called to order at 6:05 pm by the chair, Dr. Vicki Tutag Lehr. The agenda was reviewed and accepted. Introductions were made and quorum established. Conflict of interest statement was reviewed. Annual attestations were signed. None of the members had COI to report. Review of the June 14, 2016 and September 13, 2016 meeting minutes were reviewed and accepted. P & T Business: The Committee was provided Policy Updates, Legislative Updates, Pipeline Drugs and New Indications, an update on the revamping of MAPS, the DUR update and general clinical updates. New Drug Review: The committee reviewed the following new drugs and approved that the following new agents reviewed be added to the formulary as non-preferred as follows:
2 1. Bevespi Aerosphere (glycopyrrolate and formoterol) [PDL class: COPD: Beta Adrenergic/Anticholinergic Combinations; nonpreferred] 2. Migranow (sumatriptan tablets with a camphor/menthol gel) [PDL class: Serotonin Receptor Agonists: Triptans; non-preferred] 3. Xiidra (lifitegrast) 5% ophthalmic solution - [Not a PDL class, add to the MPPL--?PA] 4. Xtampza ER (oxycodone extended-release) - [PDL class: Narcotics, Longacting; non-preferred] 5. Zinbryta (daclizumab subcutaneous injection) - [PDL class: Multiple Sclerosis Agents: non-preferred] The review of the following new agent was tabled pending further evaluation of the topical NSAID subclass. 1. Vopac MDS (diclofenac) [PDL class: NSAIDs; tabled] Preferred Drug List Classes: A Review of the following Preferred Drug Classes was performed and the following actions were taken. Analgesics: 1. Narcotics: Long-acting 2. Narcotics: Short- and Intermediate-acting
3 a. Move oxycodone oral syringes to non-preferred. 3. Narcotics: Transdermal 4. NSAIDs: COX-II Inhibitors 5. NSAIDs a. Move fenoprofen to non-preferred b. Move flurbiprofen to non-preferred Central Nervous System Agents 1. Alzheimer s Dementia 2. Anti-Anxiety - General 3. Drugs for ADHD Stimulants a. Move Ritalin LA (all strengths except 10mg) to non-preferred. b. Move methylphenidate LA (all strengths except 10mg) to preferred. 4. Drugs for ADHD Non-stimulants 5. Fibromyalgia Agents 6. Multiple Sclerosis Agents: a. Move Extavia (interferon beta-1b) kit and vial to non-preferred 7. Non-Ergot Dopamine Receptor Agonsists (Anti-Parkinson s Agents) 8. Sedative Hyponotics: Nona. No change to the current classification of drug products
4 9. Serotonin Receptor Agonists (Antimigraine Agents, Triptans) a. Move Maxalt MLT (rizatriptan) to non-preferred b. Move Relpax (eletriptan) to preferred. Dermatological Agents: 1. Topical Immunomodulators 2. Topical Steroids Low Potency 3. Topical Steroids Medium Potency 4. Topical Steroids High Potency a. Move fluocinonide cream, ointment and gel to non-preferred b. Move betamethasone valerate cream, ointment and lotion to preferred. 5. Topical Steroids Very High Potency Public Comments: 1. Birgit Amann, MD, Psychiatrist & Marcus DeGraw, MD, Pediatrician: Rhodes Pharmaceuticals: Aptensio XR 2. Rick Detloff, PharmD, Pfizer: Eliquis 3. Ashley Polce, PharmD, Abbvie: Zinbryta 4. Ndidi Yaucher, PharmD, MBA, Novartis: Entresto 5. Paul Miner, PharmD, Gilead: Hepatitis C - passed 6. Mary Pat Petrillo, PharmD, Johnson & Johnson: Invokamet XR and Stelara 7. Jeff Martin, PharmD, MPA, Amgen: Enbrel
5 8. Sharad Rastogi, MD, MBA, Shire Medical Affairs Group: Xiidra The meeting was adjourned at 8:32 PM Next Meeting: Tuesday, March 14, 2016 Location: Kellogg Center
Michigan Pharmacy and Therapeutics Committee September 11, Minutes
 Michigan Pharmacy and Therapeutics Committee September 11, 2018 Minutes Attendees: Vickie Tutag Lehr, Brian Peltz, Jayne Courts, Rony Foumia, Andrew Mac, Andrew Adair, Margo Farber, David Neff, Brad Uren
Michigan Pharmacy and Therapeutics Committee September 11, 2018 Minutes Attendees: Vickie Tutag Lehr, Brian Peltz, Jayne Courts, Rony Foumia, Andrew Mac, Andrew Adair, Margo Farber, David Neff, Brad Uren
Michigan Department of Health and Human Services Pharmacy and Therapeutics Committee
 Michigan Department of Health and Human Services Pharmacy and Therapeutics Committee June 14, 2016 Minutes Final Attendee: Dr. Tutag Lehr, Andrew Mac, James Miller, Brian Peltz, Dr. Anthony Ognjan, Dr.
Michigan Department of Health and Human Services Pharmacy and Therapeutics Committee June 14, 2016 Minutes Final Attendee: Dr. Tutag Lehr, Andrew Mac, James Miller, Brian Peltz, Dr. Anthony Ognjan, Dr.
Pharmacy and Therapeutics Committee
 Michigan Department of Health and Human Services Pharmacy and Therapeutics Committee September 12, 2017 Minutes - Final Attendees: Vickie Tutag Lehr, Andrew Mac, James Miller, Brian Peltz, Anthony Ognjan,
Michigan Department of Health and Human Services Pharmacy and Therapeutics Committee September 12, 2017 Minutes - Final Attendees: Vickie Tutag Lehr, Andrew Mac, James Miller, Brian Peltz, Anthony Ognjan,
Michigan Pharmacy and Therapeutics Committee
 Michigan Pharmacy and Therapeutics Committee June 14, 2016 at 6:00 PM Kellogg Center, East Lansing, Michigan Agenda: I. Introductions II. Conflict of Interest Statement III. Approval of the Agenda IV.
Michigan Pharmacy and Therapeutics Committee June 14, 2016 at 6:00 PM Kellogg Center, East Lansing, Michigan Agenda: I. Introductions II. Conflict of Interest Statement III. Approval of the Agenda IV.
Michigan Pharmacy and Therapeutics Committee September 9, 2014 at 6:00 PM Kellogg Center, East Lansing, Michigan
 Michigan Pharmacy and Therapeutics Committee September 9, 2014 at 6:00 PM Kellogg Center, East Lansing, Michigan Agenda: Introductions Approval of Minutes of July 8, 2014 Meeting P & T Business Review
Michigan Pharmacy and Therapeutics Committee September 9, 2014 at 6:00 PM Kellogg Center, East Lansing, Michigan Agenda: Introductions Approval of Minutes of July 8, 2014 Meeting P & T Business Review
Drug Utilization Review Board
 DUR Members present DUR members absent MDCH/MMA/MPA present Steven Bernstein, M.D., Chair Carrie Germain, R. Ph, Vice Chair Bob DeYoung, PharmD James Forshee, M.D. Debera Eggleston, M.D. Sahar Swiden,
DUR Members present DUR members absent MDCH/MMA/MPA present Steven Bernstein, M.D., Chair Carrie Germain, R. Ph, Vice Chair Bob DeYoung, PharmD James Forshee, M.D. Debera Eggleston, M.D. Sahar Swiden,
Alameda Alliance for Health Pharmacy & Therapeutics (P&T) Committee Decisions
 Alameda Alliance for Health FORMULARY UPDATE Effective: October 27, 2017. Drugs notated with an * have an undetermined implementation date Alameda Alliance for Health Pharmacy & Therapeutics (P&T) Committee
Alameda Alliance for Health FORMULARY UPDATE Effective: October 27, 2017. Drugs notated with an * have an undetermined implementation date Alameda Alliance for Health Pharmacy & Therapeutics (P&T) Committee
Step Therapy Approval Criteria
 Effective Date: 01/01/2018 This document contains Step Therapy Approval Criteria for the following medications: 1. Colcrys (colchicine) 2. Dovonex (calcipotriene) 3. Enbrel (etanercept) 4. Humira (adalimumab)
Effective Date: 01/01/2018 This document contains Step Therapy Approval Criteria for the following medications: 1. Colcrys (colchicine) 2. Dovonex (calcipotriene) 3. Enbrel (etanercept) 4. Humira (adalimumab)
Pharmacy Updates Summary
 All of the following changes were reviewed and approved by the SFHP Pharmacy & Therapeutics (P&T) Committee on 4/16/2014 Effective date: 5/15/2014 Therapeutic Classes reviewed: ADHD Ophthalmic antihistamines
All of the following changes were reviewed and approved by the SFHP Pharmacy & Therapeutics (P&T) Committee on 4/16/2014 Effective date: 5/15/2014 Therapeutic Classes reviewed: ADHD Ophthalmic antihistamines
Central Nervous System Stimulants Drug Class Prior Authorization Protocol
 Line of Business: Medi-Cal Effective Date: August 16, 2017 Revision Date: August 16, 2017 Central Nervous System Stimulants Drug Class Prior Authorization Protocol This policy has been developed through
Line of Business: Medi-Cal Effective Date: August 16, 2017 Revision Date: August 16, 2017 Central Nervous System Stimulants Drug Class Prior Authorization Protocol This policy has been developed through
Quarterly pharmacy formulary change notice
 Quarterly pharmacy formulary change notice Provider update Summary: The formulary changes listed in the table below were reviewed and approved at our second quarter 2018, Pharmacy and Therapeutics Committee
Quarterly pharmacy formulary change notice Provider update Summary: The formulary changes listed in the table below were reviewed and approved at our second quarter 2018, Pharmacy and Therapeutics Committee
FORMULARY UPDATES TO DENVER HEALTH MEDICAID CHOICE (DHMC) AND CHILD HEALTH PLAN PLUS (CHP+) PLANS
 FORMULARY UPDATES TO DENVER HEALTH MEDICAID CHOICE (DHMC) AND CHILD HEALTH PLAN PLUS (CHP+) PLANS DHMC/CHP+ may add or remove drugs from the formulary or make changes to restrictions on formulary drugs
FORMULARY UPDATES TO DENVER HEALTH MEDICAID CHOICE (DHMC) AND CHILD HEALTH PLAN PLUS (CHP+) PLANS DHMC/CHP+ may add or remove drugs from the formulary or make changes to restrictions on formulary drugs
MEDICAL ASSISTANCE BULLETIN
 ISSUE DATE February 18, 2015 SUBJECT EFFECTIVE DATE January 21, 2015 MEDICAL ASSISTANCE BULLETIN NUMBER *See below BY Drug List (PDL) Update January 21, 2015 Pharmacy Services Vincent D. Gordon, Deputy
ISSUE DATE February 18, 2015 SUBJECT EFFECTIVE DATE January 21, 2015 MEDICAL ASSISTANCE BULLETIN NUMBER *See below BY Drug List (PDL) Update January 21, 2015 Pharmacy Services Vincent D. Gordon, Deputy
Step Therapy Approval Criteria
 Effective Date: 01/01/2019 This document contains for the following medications: 1. Colcrys (colchicine) 2. Dovonex (calcipotriene) 3. Enbrel (etanercept) 4. Humira (adalimumab) 5. Imitrex Injection vial
Effective Date: 01/01/2019 This document contains for the following medications: 1. Colcrys (colchicine) 2. Dovonex (calcipotriene) 3. Enbrel (etanercept) 4. Humira (adalimumab) 5. Imitrex Injection vial
Quarterly pharmacy formulary change notice
 Provider Bulletin October 2018 Quarterly pharmacy formulary change notice The formulary changes listed in the table below apply to all Anthem HealthKeepers Plus patients. The changes listed in the table
Provider Bulletin October 2018 Quarterly pharmacy formulary change notice The formulary changes listed in the table below apply to all Anthem HealthKeepers Plus patients. The changes listed in the table
FORMULARY UPDATES TO DENVER HEALTH MEDICAID CHOICE (DHMC) AND CHILD HEALTH PLAN PLUS (CHP+) PLANS
 FORMULARY UPDATES TO DENVER HEALTH MEDICAID CHOICE (DHMC) AND CHILD HEALTH PLAN PLUS (CHP+) PLANS DHMC/CHP+ may add or remove drugs from the formulary or make changes to restrictions on formulary drugs
FORMULARY UPDATES TO DENVER HEALTH MEDICAID CHOICE (DHMC) AND CHILD HEALTH PLAN PLUS (CHP+) PLANS DHMC/CHP+ may add or remove drugs from the formulary or make changes to restrictions on formulary drugs
Methylphenidate also has an off-label indication for depression, although published trials are limited in size and duration (14).
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.60.25 Yeah Subject: Methylphenidates Page: 1 of 5 Last Review Date: December 2, 2016 Methylphenidate
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.60.25 Yeah Subject: Methylphenidates Page: 1 of 5 Last Review Date: December 2, 2016 Methylphenidate
Step Therapy Approval Criteria
 Effective Date: 07/01/2015 This document contains for the following medications: 1. Ambien CR (zolpidem ER) 2. Chantix Continuing Month (varenicline) 3. Chantix Starting Month (varenicline) 4. Cymbalta
Effective Date: 07/01/2015 This document contains for the following medications: 1. Ambien CR (zolpidem ER) 2. Chantix Continuing Month (varenicline) 3. Chantix Starting Month (varenicline) 4. Cymbalta
South Carolina Department of Health and Human Services Post Office Box 8206 Columbia, South Carolina
 South Carolina Department of Health and Human Services Post Office Box 8206 Columbia, South Carolina 29202-8206 Pharmacy and Therapeutics (P&T) Committee Meeting MINUTES 1. Call To Order A meeting of the
South Carolina Department of Health and Human Services Post Office Box 8206 Columbia, South Carolina 29202-8206 Pharmacy and Therapeutics (P&T) Committee Meeting MINUTES 1. Call To Order A meeting of the
Triptan Quantity Limit
 *- Florida Healthy Kids Triptan Quantity Limit Override(s) Quantity Limit Approval Duration 1 year Oral Tablets Axert (almotriptan) tablets Relpax (eletriptan) tablets 6 tablets (6.25 mg) 12 tablets (12.5
*- Florida Healthy Kids Triptan Quantity Limit Override(s) Quantity Limit Approval Duration 1 year Oral Tablets Axert (almotriptan) tablets Relpax (eletriptan) tablets 6 tablets (6.25 mg) 12 tablets (12.5
Pharmacy Medical Necessity Guidelines: ADHD CNS Stimulant Medications
 Pharmacy Medical Necessity Guidelines: ADHD CNS Stimulant Medications Effective: November 13, 2018 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review
Pharmacy Medical Necessity Guidelines: ADHD CNS Stimulant Medications Effective: November 13, 2018 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review
Quarterly pharmacy formulary change notice
 Quarterly pharmacy formulary change notice The formulary changes listed in the table below were reviewed and approved at our second quarter 2018 Pharmacy and Therapeutics Committee meeting. Effective October
Quarterly pharmacy formulary change notice The formulary changes listed in the table below were reviewed and approved at our second quarter 2018 Pharmacy and Therapeutics Committee meeting. Effective October
Alzheimer Disease Agents Drug Class Prior Authorization Protocol
 Line of Business: Medi-Cal Effective Date: August 16, 2017 Revision Date: August 16, 2017 Alzheimer Disease Agents Drug Class Prior Authorization Protocol This policy has been developed through review
Line of Business: Medi-Cal Effective Date: August 16, 2017 Revision Date: August 16, 2017 Alzheimer Disease Agents Drug Class Prior Authorization Protocol This policy has been developed through review
Pharmacy Prior Authorization Clinical Guideline for Attention Deficit Disorder/Attention Deficit Hyperactivity CNS Stimulants
 AETNA BETTER HEALTH Pharmacy Prior Authorization Clinical Guideline for Attention Deficit Disorder/Attention Deficit Hyperactivity CNS Stimulants Formulary amphetamine/dextroamphetamine IR, ER (generic
AETNA BETTER HEALTH Pharmacy Prior Authorization Clinical Guideline for Attention Deficit Disorder/Attention Deficit Hyperactivity CNS Stimulants Formulary amphetamine/dextroamphetamine IR, ER (generic
Step Therapy Approval Criteria
 Effective Date: 10/01/2016 This document contains Step Therapy Approval Criteria for the following medications: 1. Colcrys (colchicine) 2. Cymbalta (duloxetine) 3. Dovonex (calcipotriene) 4. Enbrel (etanercept)
Effective Date: 10/01/2016 This document contains Step Therapy Approval Criteria for the following medications: 1. Colcrys (colchicine) 2. Cymbalta (duloxetine) 3. Dovonex (calcipotriene) 4. Enbrel (etanercept)
MEETING MINUTES. Members Present: Cathy Zehrung, RPh; Bill Origer, MD; Rich Clark, MD, MPH; James Slater, PharmD; Walter Hardin, D.O.
 Drug Use Research & Management Program OHA Division of Medical Assistance Programs 500 Summer Street NE, E35; Salem, OR 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Oregon Drug Use Review / Pharmacy
Drug Use Research & Management Program OHA Division of Medical Assistance Programs 500 Summer Street NE, E35; Salem, OR 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Oregon Drug Use Review / Pharmacy
Quarterly pharmacy formulary change notice
 MEDICAID PROVIDER BULLETIN October 2018 The formulary changes listed in the table below were reviewed and approved at the second-quarter 2018 Pharmacy and Therapeutics Committee meeting. Effective October
MEDICAID PROVIDER BULLETIN October 2018 The formulary changes listed in the table below were reviewed and approved at the second-quarter 2018 Pharmacy and Therapeutics Committee meeting. Effective October
Methylphenidate Dexmethylphenidate
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.60.25 Yeah Subject: Methylphenidates Page: 1 of 6 Last Review Date: September 15, 2017 Methylphenidate
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.60.25 Yeah Subject: Methylphenidates Page: 1 of 6 Last Review Date: September 15, 2017 Methylphenidate
STEP THERAPY CRITERIA
 STEP THERAPY This is a complete list of drugs that have written coverage determination policies. Drugs on this list do not indicate that this particular drug will be covered under your medical or prescription
STEP THERAPY This is a complete list of drugs that have written coverage determination policies. Drugs on this list do not indicate that this particular drug will be covered under your medical or prescription
Page: 1 of 6. Aimovig (erenumab-aooe) injection, Ajovy (fremanezumab-vfrm) injection, Emgality (galcanezumab-gnim)
 Page: 1 of 6 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Last Review Date: November 30, 2018 Description Aimovig (erenumab-aooe) injection, Ajovy (fremanezumab-vfrm)
Page: 1 of 6 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Last Review Date: November 30, 2018 Description Aimovig (erenumab-aooe) injection, Ajovy (fremanezumab-vfrm)
OVERVIEW FOOD AND DRUG ADMINISTRATION-APPROVED INDICATIONS
 Pharmacy Medical Necessity Guidelines: Effective: September 18, 2017 Prior Authorization Required Type of Review Care Management Not Type of Review Clinical Review Pharmacy (RX) or Medical (MED) Benefit
Pharmacy Medical Necessity Guidelines: Effective: September 18, 2017 Prior Authorization Required Type of Review Care Management Not Type of Review Clinical Review Pharmacy (RX) or Medical (MED) Benefit
2016 Step Therapy (ST) Criteria
 2016 Step Therapy (ST) Some drugs require step therapy pre-approval. This means that your doctor must have you first try a different drug to treat your medical condition before we will cover a drug that
2016 Step Therapy (ST) Some drugs require step therapy pre-approval. This means that your doctor must have you first try a different drug to treat your medical condition before we will cover a drug that
Pharmacy Medical Necessity Guidelines: Migraine Medications
 Pharmacy Medical Necessity Guidelines: Effective: September 18, 2017 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review Pharmacy (RX) or Medical (MED)
Pharmacy Medical Necessity Guidelines: Effective: September 18, 2017 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review Pharmacy (RX) or Medical (MED)
Methylphenidate Dexmethylphenidate
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Methylphenidates Page: 1 of 7 Last Review Date: November 30, 2018 Methylphenidate Dexmethylphenidate
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Methylphenidates Page: 1 of 7 Last Review Date: November 30, 2018 Methylphenidate Dexmethylphenidate
Calgary Long Term Care Formulary
 Page 1 of 10 Calgary Long Term Care Formulary Pharmacy & Therapeutics November 2018 Highlights https://www.albertahealthservices.ca/info/page4071.aspx Page 2 of 10 Contents November 2018... 3 Formulary
Page 1 of 10 Calgary Long Term Care Formulary Pharmacy & Therapeutics November 2018 Highlights https://www.albertahealthservices.ca/info/page4071.aspx Page 2 of 10 Contents November 2018... 3 Formulary
Texas Vendor Drug Program. Formulary Delimited File Layout. April 26, 2017
 Texas Vendor Drug Program Formulary Delimited File Layout April 26, 2017 The Vendor Drug Program provides a weekly update of resource data available for download from txvendordrug.com/resources/downloads.
Texas Vendor Drug Program Formulary Delimited File Layout April 26, 2017 The Vendor Drug Program provides a weekly update of resource data available for download from txvendordrug.com/resources/downloads.
Connecticut Medical Assistance Pharmacy Program Drug Utilization Review (DUR) Program DUR Board Meeting
 September 2008 Minutes ATTENDEES Board Members Present: Kenneth Fisher, R.Ph. (Chair); Dennis Chapron, M.S.; Richard Gannon, Pharm.D.; Keith Lyke R.Ph., Mike Moore, R.Ph., MPH; Bhupesh Mangla, M.D., Ram
September 2008 Minutes ATTENDEES Board Members Present: Kenneth Fisher, R.Ph. (Chair); Dennis Chapron, M.S.; Richard Gannon, Pharm.D.; Keith Lyke R.Ph., Mike Moore, R.Ph., MPH; Bhupesh Mangla, M.D., Ram
2018 Step Therapy (ST) Criteria
 2018 Step Therapy (ST) Criteria Some drugs require step therapy pre-approval. This means that your doctor must have you first try a different drug to treat your medical condition before we will cover a
2018 Step Therapy (ST) Criteria Some drugs require step therapy pre-approval. This means that your doctor must have you first try a different drug to treat your medical condition before we will cover a
South Carolina Department of Health and Human Services Post Office Box 8206 Columbia, South Carolina
 South Carolina Department of Health and Human Services Post Office Box 8206 Columbia, South Carolina 29202-8206 Pharmacy and Therapeutics (P&T) Committee Meeting MINUTES 1. Call To Order A meeting of the
South Carolina Department of Health and Human Services Post Office Box 8206 Columbia, South Carolina 29202-8206 Pharmacy and Therapeutics (P&T) Committee Meeting MINUTES 1. Call To Order A meeting of the
Medication For Migraine Chart: Table 1: Acute Treatment when the attack begins
 Medication For Migraine Chart: Table 1: Acute Treatment when the attack begins Page a Analgesics (painkillers) Non-steroidal antiinflammatory drugs (NSAIDs) Prescription required Brand Name Formulation
Medication For Migraine Chart: Table 1: Acute Treatment when the attack begins Page a Analgesics (painkillers) Non-steroidal antiinflammatory drugs (NSAIDs) Prescription required Brand Name Formulation
Acyclovir Ointment. Aetna Better Health Pennsylvania. Products Affected. acyclovir ointment 5 % external Details. Criteria
 Medications that require Step Therapy (ST) require trial and failure of preferred formulary agents prior to their authorization. If the prerequisite medications have been filled within the specified time
Medications that require Step Therapy (ST) require trial and failure of preferred formulary agents prior to their authorization. If the prerequisite medications have been filled within the specified time
Texas Vendor Drug Program. Formulary Drug Index File Layout. Layout effective: Jul. 2, 2018 Document update: Oct. 1, 2018
 Texas Vendor Drug Program Formulary Drug Index File Layout Layout effective: Jul. 2, 2018 Document update: Oct. 1, 2018 The Vendor Drug Program provides a weekly update of resource data available for download
Texas Vendor Drug Program Formulary Drug Index File Layout Layout effective: Jul. 2, 2018 Document update: Oct. 1, 2018 The Vendor Drug Program provides a weekly update of resource data available for download
UPMC for You Pharmacy and Therapeutics Committee Meeting April 7, 2014 meeting
 UPMC for You Pharmacy and Therapeutics Committee Meeting April 7, 2014 meeting 1. Call to order: The meeting was called to order at 7:05 a.m. 2. Review of the minutes: The minutes of the January meeting
UPMC for You Pharmacy and Therapeutics Committee Meeting April 7, 2014 meeting 1. Call to order: The meeting was called to order at 7:05 a.m. 2. Review of the minutes: The minutes of the January meeting
Idaho DUR Board Meeting Minutes. Committee Member Present: Matthew Hyde, Pharm.D., Perry Brown M.D., Paul Cady, Pharm.D., Ryan Heyborne M.D.
 Date: February 16, 2017 Time: 9am-12:15pm Idaho DUR Board Meeting Minutes Location: Idaho Medicaid, 3232 Elder Street, Boise, Idaho, Conference Room D-West Moderator: Perry Brown, M.D. Committee Member
Date: February 16, 2017 Time: 9am-12:15pm Idaho DUR Board Meeting Minutes Location: Idaho Medicaid, 3232 Elder Street, Boise, Idaho, Conference Room D-West Moderator: Perry Brown, M.D. Committee Member
IHCP bulletin INDIANA HEALTH COVERAGE PROGRAMS BT MAY 29, 2012
 IHCP bulletin INDIANA HEALTH COVERAGE PROGRAMS BT201218 MAY 29, 2012 Changes to the Preferred Drug List Changes to the Preferred Drug List (PDL) were made at the May 18, 2012, Drug Utilization Review (DUR)
IHCP bulletin INDIANA HEALTH COVERAGE PROGRAMS BT201218 MAY 29, 2012 Changes to the Preferred Drug List Changes to the Preferred Drug List (PDL) were made at the May 18, 2012, Drug Utilization Review (DUR)
Idaho DUR Board Meeting Minutes
 Idaho DUR Board Meeting Minutes Date: January 21, 2016 Time: 9am-1:30pm Location: Idaho Medicaid, 3232 Elder Street, Boise, Idaho, Conference Room D-West Moderator: Mark Turner, M.D. Committee Members
Idaho DUR Board Meeting Minutes Date: January 21, 2016 Time: 9am-1:30pm Location: Idaho Medicaid, 3232 Elder Street, Boise, Idaho, Conference Room D-West Moderator: Mark Turner, M.D. Committee Members
Maxalt. Maxalt / Maxalt-MLT (rizatriptan) Description. Section: Prescription Drugs Effective Date: April 1, 2016
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Maxalt Page: 1 of 5 Last Review Date: March 18, 2016 Maxalt Description Maxalt / Maxalt-MLT (rizatriptan)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Maxalt Page: 1 of 5 Last Review Date: March 18, 2016 Maxalt Description Maxalt / Maxalt-MLT (rizatriptan)
Iowa Medicaid Drug Utilization Review (DUR) Commission Meeting March 4, 2009
 CHESTER J. CULVER, GOVERNOR PATTY JUDGE, LT. GOVERNOR DEPARTMENT OF HUMAN SERVICES EUGENE I. GESSOW, DIRECTOR Iowa Medicaid Drug Utilization Review (DUR) Commission Meeting March 4, 2009 Location: Learning
CHESTER J. CULVER, GOVERNOR PATTY JUDGE, LT. GOVERNOR DEPARTMENT OF HUMAN SERVICES EUGENE I. GESSOW, DIRECTOR Iowa Medicaid Drug Utilization Review (DUR) Commission Meeting March 4, 2009 Location: Learning
Subject: HPN/SHL COMMERCIAL PDL UPDATES EFFECTIVE JANUARY 1, 2018
 Date: November 7, 2017 To: From: HPN/SHL Contracted Pharmacies and Providers Ryan Bitton, PharmD, MBA Senior Director, Pharmacy Subject: HPN/SHL COMMERCIAL PDL UPDATES EFFECTIVE JANUARY 1, 2018 Effective
Date: November 7, 2017 To: From: HPN/SHL Contracted Pharmacies and Providers Ryan Bitton, PharmD, MBA Senior Director, Pharmacy Subject: HPN/SHL COMMERCIAL PDL UPDATES EFFECTIVE JANUARY 1, 2018 Effective
STEP THERAPY CRITERIA
 STEP THERAPY This is a complete list of drugs that have written coverage determination policies. Drugs on this list do not indicate that this particular drug will be covered under your medical or prescription
STEP THERAPY This is a complete list of drugs that have written coverage determination policies. Drugs on this list do not indicate that this particular drug will be covered under your medical or prescription
UnitedHealthcare Pharmacy Clinical Pharmacy Programs. Program Number 2017 P Prior Authorization/Notification CNS Stimulants
 UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2017 P 1020-6 Program Prior Authorization/Notification CNS Stimulants Medication Adderall* (amphetamine-dextroamphetamine mixed salts),
UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2017 P 1020-6 Program Prior Authorization/Notification CNS Stimulants Medication Adderall* (amphetamine-dextroamphetamine mixed salts),
Step Therapy Medications
 Step Therapy Medications Step Therapy (ST PA ) is an automated form of prior authorization. It encourages the use of therapies that should be tried first, before other treatments are covered, based on
Step Therapy Medications Step Therapy (ST PA ) is an automated form of prior authorization. It encourages the use of therapies that should be tried first, before other treatments are covered, based on
WELLCARE/ OHANA HEALTH PLAN 2015 STEP THERAPY CRITERIA (No Changes Made Since: 08/2015)
 WELLCARE/ OHANA HEALTH PLAN 2015 STEP THERAPY CRITERIA (No Changes Made Since: 08/2015) **To get updated information about the drugs covered by WellCare/ Ohana, please visit our website (https://www.wellcare.com
WELLCARE/ OHANA HEALTH PLAN 2015 STEP THERAPY CRITERIA (No Changes Made Since: 08/2015) **To get updated information about the drugs covered by WellCare/ Ohana, please visit our website (https://www.wellcare.com
STEP THERAPY CRITERIA
 STEP THERAPY This is a complete list of drugs that have written coverage determination policies. Drugs on this list do not indicate that this particular drug will be covered under your medical or prescription
STEP THERAPY This is a complete list of drugs that have written coverage determination policies. Drugs on this list do not indicate that this particular drug will be covered under your medical or prescription
SUMAVEL DOSEPRO (sumatriptan succinate) solution for injection
 SUMAVEL DOSEPRO (sumatriptan succinate) solution for injection Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit
SUMAVEL DOSEPRO (sumatriptan succinate) solution for injection Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit
Updates to your prescription benefits
 Updates to your prescription benefits Effective July 1, 2018 Update Summary Within the Prescription Drug List (PDL), medications are grouped by tier. The tier indicates the amount you pay when you fill
Updates to your prescription benefits Effective July 1, 2018 Update Summary Within the Prescription Drug List (PDL), medications are grouped by tier. The tier indicates the amount you pay when you fill
IHCP bulletin INDIANA HEALTH COVERAGE PROGRAMS BT NOVEMBER 30, 2010
 IHCP bulletin INDIANA HEALTH COVERAGE PROGRAMS BT201056 NOVEMBER 30, 2010 Changes to the Preferred Drug List Changes to the Preferred Drug List (PDL) were made at the November 19, 2010, Drug Utilization
IHCP bulletin INDIANA HEALTH COVERAGE PROGRAMS BT201056 NOVEMBER 30, 2010 Changes to the Preferred Drug List Changes to the Preferred Drug List (PDL) were made at the November 19, 2010, Drug Utilization
Celecoxib Powder, Diclofenac Powder, Flurbiprofen Powder, Ibuprofen Powder, Ketoprofen Powder, Meloxicam Powder, Tramadol Powder
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.02.26 Subject: Anti-Inflammatory Pain Powders Page: 1 of 5 Last Review Date: December 3, 2015 Anti-Inflammatory
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.02.26 Subject: Anti-Inflammatory Pain Powders Page: 1 of 5 Last Review Date: December 3, 2015 Anti-Inflammatory
ALOGLIPTIN STEP. Details. Step Therapy Requirements Effective November 1, 2017
 ALOGLIPTIN STEP alogliptin 12.5 mg tablet alogliptin 12.5 mg-metformin 1,000 mg tablet alogliptin 12.5 mg-metformin 500 mg tablet alogliptin 12.5 mg-pioglitazone 15 mg tablet alogliptin 12.5 mg-pioglitazone
ALOGLIPTIN STEP alogliptin 12.5 mg tablet alogliptin 12.5 mg-metformin 1,000 mg tablet alogliptin 12.5 mg-metformin 500 mg tablet alogliptin 12.5 mg-pioglitazone 15 mg tablet alogliptin 12.5 mg-pioglitazone
Updates to your prescription benefits
 Updates to your prescription benefits Effective July 1, 2018 Update Summary Within the Prescription Drug List (PDL), medications are grouped by tier. The tier indicates the amount you pay when you fill
Updates to your prescription benefits Effective July 1, 2018 Update Summary Within the Prescription Drug List (PDL), medications are grouped by tier. The tier indicates the amount you pay when you fill
New Products to Market
 Fee-for-Service Pharmacy Provider Notice #222 **Preferred Drug Status Changes** December 01, 2017 Dear Kentucky Medicaid Provider: As we bring 2017 to a close, below is a recap of the new products to market
Fee-for-Service Pharmacy Provider Notice #222 **Preferred Drug Status Changes** December 01, 2017 Dear Kentucky Medicaid Provider: As we bring 2017 to a close, below is a recap of the new products to market
Pharmacy Medical Necessity Guidelines: ADHD CNS Stimulant Medications
 Pharmacy Medical Necessity Guidelines: ADHD CNS Stimulant Medications Effective: January 1, 2018 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review Pharmacy
Pharmacy Medical Necessity Guidelines: ADHD CNS Stimulant Medications Effective: January 1, 2018 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review Pharmacy
2017 Formulary Changes Year to Date
 2017 Formulary Changes Year to Date Health Choice Arizona may add or remove drugs from our formulary during the year. If we remove drugs from our formulary, add prior authorization, quantity limits and/or
2017 Formulary Changes Year to Date Health Choice Arizona may add or remove drugs from our formulary during the year. If we remove drugs from our formulary, add prior authorization, quantity limits and/or
Clinical Policy: CNS Stimulants Reference Number: CP.PMN.XX Effective Date: Last Review Date: Line of Business: Commercial, Medicaid
 Clinical Policy: Reference Number: CP.PMN.XX Effective Date: 03.01.18 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: Reference Number: CP.PMN.XX Effective Date: 03.01.18 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy for important
Page: 1 of 5. Methylphenidate also has an off-label indication for depression, although published trials are limited in size and duration (12).
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.06.25 Page: 1 of 5 Last Review Date: June 19, 2015 Description Concerta / Daytrana / Metadate CD / Metadate
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.06.25 Page: 1 of 5 Last Review Date: June 19, 2015 Description Concerta / Daytrana / Metadate CD / Metadate
Sumatriptan Tablets, Nasal Spray (Imitrex), Nasal Powder (Onzetra Xsail), sumatriptan and naproxen sodium (Treximet tablets)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 05.70.10 Subject: Sumatriptan Page: 1 of 5 Last Review Date: December 2, 2016 Sumatriptan Description Sumatriptan
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 05.70.10 Subject: Sumatriptan Page: 1 of 5 Last Review Date: December 2, 2016 Sumatriptan Description Sumatriptan
Commercial Formulary and Utilization Management Program Updates for January 2017 Producer Communication #782 Issued November 10, 2016
 Commercial and Utilization Management Program Updates for January 2017 Producer Communication #782 Issued November 10, 2016 Message The Capital BlueCross Pharmacy & Therapeutics (P&T) Committee, consisting
Commercial and Utilization Management Program Updates for January 2017 Producer Communication #782 Issued November 10, 2016 Message The Capital BlueCross Pharmacy & Therapeutics (P&T) Committee, consisting
PHARMACY COVERAGE GUIDELINES ORIGINAL EFFECTIVE DATE: 3/15/18 SECTION: DRUGS LAST REVIEW DATE: 3/15/18 LAST CRITERIA REVISION DATE: ARCHIVE DATE:
 DYANAVEL XR (amphetamine) extended-release oral suspension Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit
DYANAVEL XR (amphetamine) extended-release oral suspension Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit
Pharmacy Medical Necessity Guidelines: CNS Stimulant Medications
 Pharmacy Medical Necessity Guidelines: CNS Stimulant Medications Effective: June 1, 2017 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review Pharmacy
Pharmacy Medical Necessity Guidelines: CNS Stimulant Medications Effective: June 1, 2017 Prior Authorization Required Type of Review Care Management Not Covered Type of Review Clinical Review Pharmacy
UTILIZATION MANAGEMENT CRITERIA
 SUMATRIPTAN (ALSUMA, IMITREX, SUMAVEL DOSEPRO, ZECUITY ) UTILIZATION MANAGEMENT CRITERIA DRUG CLASS: BRAND (generic) NAME: Serotonin 5-HT1 receptor agonists Imitrex (sumatriptan), Alsuma (sumatriptan),
SUMATRIPTAN (ALSUMA, IMITREX, SUMAVEL DOSEPRO, ZECUITY ) UTILIZATION MANAGEMENT CRITERIA DRUG CLASS: BRAND (generic) NAME: Serotonin 5-HT1 receptor agonists Imitrex (sumatriptan), Alsuma (sumatriptan),
WY P&T Committee Meeting Minutes Thursday, November 8, 2018 Cheyenne, WY 10 a.m 1 p.m.
 WY P&T Committee Meeting Minutes Thursday, November 8, 2018 Cheyenne, WY 10 a.m 1 p.m. Members present: Hoo Fang Choo, Joseph Horam, Paul Johnson, Scott Johnston, Rhonda McLaughlin, Robert Monger, Chris
WY P&T Committee Meeting Minutes Thursday, November 8, 2018 Cheyenne, WY 10 a.m 1 p.m. Members present: Hoo Fang Choo, Joseph Horam, Paul Johnson, Scott Johnston, Rhonda McLaughlin, Robert Monger, Chris
South Carolina Department of Health and Human Services Post Office Box 8206 Columbia, South Carolina
 South Carolina Department of Health and Human Services Post Office Box 8206 Columbia, South Carolina 29202-8206 Pharmacy and Therapeutics (P&T) Committee Meeting MINUTES 1. Call to Order A meeting of the
South Carolina Department of Health and Human Services Post Office Box 8206 Columbia, South Carolina 29202-8206 Pharmacy and Therapeutics (P&T) Committee Meeting MINUTES 1. Call to Order A meeting of the
Drug Therapy Guidelines
 Drug Therapy Guidelines Applicable Medical Benefit Effective: 5/1/18 Pharmacy- Formulary 1 x Next Review: 3/19 Pharmacy- Formulary 2 x Date of Origin: 8/29/06 Triptans: almotriptan, Amerge, Axert, Frova,
Drug Therapy Guidelines Applicable Medical Benefit Effective: 5/1/18 Pharmacy- Formulary 1 x Next Review: 3/19 Pharmacy- Formulary 2 x Date of Origin: 8/29/06 Triptans: almotriptan, Amerge, Axert, Frova,
Medical and Pharmacy Cost and Utilization Outcomes of a Quantity Limit on the 5-HT1 Agonists (Triptans) by a Managed Care Organization
 REVIEW Medical and Pharmacy Cost and Utilization Outcomes of a Quantity Limit on the 5-HT1 Agonists (Triptans) by a Managed Care Organization by Eric J. Culley and Robert T. Wanovich OBJECTIVE: To determine
REVIEW Medical and Pharmacy Cost and Utilization Outcomes of a Quantity Limit on the 5-HT1 Agonists (Triptans) by a Managed Care Organization by Eric J. Culley and Robert T. Wanovich OBJECTIVE: To determine
ANTIEMETICS STEP. Step Therapy Requirements Effective April 1, 2019
 Step Therapy Requirements Effective April 1, 2019 ANTIEMETICS STEP Sancuso 3.1 mg/24 hour transdermal patch Zuplenz 4 mg oral soluble film Zuplenz 8 mg oral soluble film COVERAGE OF CERTAIN BRAND NAME
Step Therapy Requirements Effective April 1, 2019 ANTIEMETICS STEP Sancuso 3.1 mg/24 hour transdermal patch Zuplenz 4 mg oral soluble film Zuplenz 8 mg oral soluble film COVERAGE OF CERTAIN BRAND NAME
Triptans Quantity Limit Program Summary
 Triptans Quantity Limit Program Summary FDA APPROVED INDICATIONS AND DOSAGE 1-13,14,23,24 Agents Amerge (naratriptan) 1, 2.5 tablets Axert (almotriptan) 6.25, 12.5 tablets migraine attacks with/without
Triptans Quantity Limit Program Summary FDA APPROVED INDICATIONS AND DOSAGE 1-13,14,23,24 Agents Amerge (naratriptan) 1, 2.5 tablets Axert (almotriptan) 6.25, 12.5 tablets migraine attacks with/without
QUANTITY LIMIT CRITERIA
 DRUG CLASS (ADHD) AGENTS BRAND NAME (generic) QUANTITY LIMIT CRITERIA ATTENTION DEFICIT HYPERACTIVITY DISORDER ADDERALL (amphetamine mixture) ADDERALL XR (amphetamine extended-release mixture) ADZENYS
DRUG CLASS (ADHD) AGENTS BRAND NAME (generic) QUANTITY LIMIT CRITERIA ATTENTION DEFICIT HYPERACTIVITY DISORDER ADDERALL (amphetamine mixture) ADDERALL XR (amphetamine extended-release mixture) ADZENYS
NB Drug Plans Formulary Update
 Bulletin #980 August 23, 2018 NB Drug Plans Formulary Update This update to the New Brunswick Drug Plans Formulary is effective August 23, 2018. Included in this bulletin: Regular Benefit Additions Special
Bulletin #980 August 23, 2018 NB Drug Plans Formulary Update This update to the New Brunswick Drug Plans Formulary is effective August 23, 2018. Included in this bulletin: Regular Benefit Additions Special
Faculty Disclosures. Learning Objectives. Acute Treatment Strategies
 WWW.AMERICANHEADACHESOCIETY.ORG Acute Treatment Strategies Content developed by: Lawrence C. Newman, MD, FAHS Donna Gutterman, PharmD Faculty Disclosures LAWRENCE C. NEWMAN, MD, FAHS Dr. Newman has received
WWW.AMERICANHEADACHESOCIETY.ORG Acute Treatment Strategies Content developed by: Lawrence C. Newman, MD, FAHS Donna Gutterman, PharmD Faculty Disclosures LAWRENCE C. NEWMAN, MD, FAHS Dr. Newman has received
Clinical Policy: CNS Stimulants Reference Number: CP.PMN.92 Effective Date: Last Review Date: Line of Business: Commercial, Medicaid
 Clinical Policy: Reference Number: CP.PMN.92 Effective Date: 03.01.18 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: Reference Number: CP.PMN.92 Effective Date: 03.01.18 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy for important
Updates to your prescription benefits
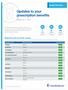 Updates to your prescription benefits Effective Jan. 1, 2018 Update Summary Within the Prescription Drug List (PDL), medications are grouped by tier. The tier indicates the amount you pay when you fill
Updates to your prescription benefits Effective Jan. 1, 2018 Update Summary Within the Prescription Drug List (PDL), medications are grouped by tier. The tier indicates the amount you pay when you fill
New Product to Market: Lonhala Magnair
 Drug Review and The following tables list the Agenda items as well as the that are scheduled to be presented and reviewed at the May 17, 2018 meeting of the Pharmacy and Therapeutics Advisory Committee.
Drug Review and The following tables list the Agenda items as well as the that are scheduled to be presented and reviewed at the May 17, 2018 meeting of the Pharmacy and Therapeutics Advisory Committee.
STATE OF TENNESSEE DEPARTMENT OF FINANCE AND ADMINISTRATION DIVISION OF HEALTH CARE FINANCE AND ADMINISTRATION BUREAU OF TENNCARE
 STATE OF TENNESSEE DEPARTMENT OF FINANCE AND ADMINISTRATION DIVISION OF HEALTH CARE FINANCE AND ADMINISTRATION BUREAU OF TENNCARE 310 Great Circle Road NASHVILLE, TENNESSEE 37243 This notice is to advise
STATE OF TENNESSEE DEPARTMENT OF FINANCE AND ADMINISTRATION DIVISION OF HEALTH CARE FINANCE AND ADMINISTRATION BUREAU OF TENNCARE 310 Great Circle Road NASHVILLE, TENNESSEE 37243 This notice is to advise
Kentucky Department for Medicaid Services Pharmacy and Therapeutics Advisory Committee Recommendations
 Kentucky Department for Medicaid Services Pharmacy and May 17, 2018 The following chart provides a summary of the recommendations that were made by the Pharmacy and Therapeutics (P&T) Advisory Committee
Kentucky Department for Medicaid Services Pharmacy and May 17, 2018 The following chart provides a summary of the recommendations that were made by the Pharmacy and Therapeutics (P&T) Advisory Committee
Regulatory Status FDA approved indication: Migranal Nasal Spray is indicated for the acute treatment of migraine headaches with or without aura (1).
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.60 Subject: Migranal Nasal Spray Page: 1 of 5 Last Review Date: November 30, 2018 Migranal Nasal Spray
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.60 Subject: Migranal Nasal Spray Page: 1 of 5 Last Review Date: November 30, 2018 Migranal Nasal Spray
New Generics: Specialty Network: Retail Pharmacies Dispensing Specialty Products
 SUPPORTING OUR PROVIDER PARTNERS THROUGH COMMUNICATION AND COLLABORATION. DATE JANUARY 2016 ISSUE 1 HELPFUL NUMBERS FOR PROVIDERS Magellan: 1-800-846-7971 Bin: 016523 Processor control: 747 HELPFUL NUMBERS
SUPPORTING OUR PROVIDER PARTNERS THROUGH COMMUNICATION AND COLLABORATION. DATE JANUARY 2016 ISSUE 1 HELPFUL NUMBERS FOR PROVIDERS Magellan: 1-800-846-7971 Bin: 016523 Processor control: 747 HELPFUL NUMBERS
ONZETRA XSAIL (sumatriptan) nasal powder
 ONZETRA XSAIL (sumatriptan) nasal powder Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy
ONZETRA XSAIL (sumatriptan) nasal powder Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy
South Carolina Department of Health and Human Services Post Office Box 8206 Columbia, South Carolina
 South Carolina Department of Health and Human Services Post Office Box 8206 Columbia, South Carolina 29202-8206 Pharmacy and Therapeutics (P&T) Committee Meeting MINUTES 1. Call To Order A meeting of the
South Carolina Department of Health and Human Services Post Office Box 8206 Columbia, South Carolina 29202-8206 Pharmacy and Therapeutics (P&T) Committee Meeting MINUTES 1. Call To Order A meeting of the
Sumatriptan Tablets, Nasal Spray (Imitrex), Nasal Powder (Onzetra Xsail), sumatriptan and naproxen sodium (Treximet tablets)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 0 Subject: Sumatriptan Page: 1 of 6 Last Review Date: November 30, 2018 Sumatriptan Description Sumatriptan
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 0 Subject: Sumatriptan Page: 1 of 6 Last Review Date: November 30, 2018 Sumatriptan Description Sumatriptan
ADVANCES IN MIGRAINE MANAGEMENT
 ADVANCES IN MIGRAINE MANAGEMENT Joanna Girard Katzman, M.D.MSPH Assistant Professor, Dept. of Neurology Project ECHO, Chronic Pain Program University of New Mexico Outline Migraine throughout the decades
ADVANCES IN MIGRAINE MANAGEMENT Joanna Girard Katzman, M.D.MSPH Assistant Professor, Dept. of Neurology Project ECHO, Chronic Pain Program University of New Mexico Outline Migraine throughout the decades
Your prescription benefit updates Formulary Updates - Effective January 1, 2019
 Your prescription benefit updates Formulary Updates - Effective January 1, 2019 Medications are grouped by the conditions they treat. Each medication is placed in a tier that shows the amount you will
Your prescription benefit updates Formulary Updates - Effective January 1, 2019 Medications are grouped by the conditions they treat. Each medication is placed in a tier that shows the amount you will
Extended release adderall
 Extended release adderall The Borg System is 100 % Extended release adderall Use Adderall XR (dextroamphetamine and amphetamine extended-release capsules (adderall XR)) as you were told by your doctor.
Extended release adderall The Borg System is 100 % Extended release adderall Use Adderall XR (dextroamphetamine and amphetamine extended-release capsules (adderall XR)) as you were told by your doctor.
UnitedHealthcare Pharmacy Clinical Pharmacy Programs
 UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2017 P 3053-7 Program Step Therapy Long Acting Opioids Medication Includes both brand and generic versions of the listed products unless
UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2017 P 3053-7 Program Step Therapy Long Acting Opioids Medication Includes both brand and generic versions of the listed products unless
I. UNIFORM FORMULARY REVIEW PROCESS
 DOD PHARMACY AND THERAPEUTICS COMMITTEE RECOMMENDATIONS INFORMATION FOR THE UNIFORM FORMULARY BENEFICIARY ADVISORY PANEL I. UNIFORM FORMULARY REVIEW PROCESS Under 10 United States Code 1074g, as implemented
DOD PHARMACY AND THERAPEUTICS COMMITTEE RECOMMENDATIONS INFORMATION FOR THE UNIFORM FORMULARY BENEFICIARY ADVISORY PANEL I. UNIFORM FORMULARY REVIEW PROCESS Under 10 United States Code 1074g, as implemented
MEETING MINUTES. Members Present: Tracy Klein, PhD, FNP; Cathy Zehrung, RPh; Phillip Levine, PhD; Zahia Esber, MD;
 Drug Use Research & Management Program OHA Division of Medical Assistance Programs 500 Summer Street NE, E35; Salem, OR 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Oregon Drug Use Review / Pharmacy
Drug Use Research & Management Program OHA Division of Medical Assistance Programs 500 Summer Street NE, E35; Salem, OR 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Oregon Drug Use Review / Pharmacy
FirstCarolinaCare Insurance Company Step Therapy Requirements
 ANALGESICS, NARCOTICS KADIAN MORPHINE SULFATE ER PRIOR CLAIM FOR MORPHINE SULFATE SUSTAINED ACTION TABLET (MS CONTIN) WITHIN THE PAST 120 DAYS. ANTIBACTERIALS (EENT) BESIVANCE PRIOR CLAIM FOR CIPROFLOXACIN
ANALGESICS, NARCOTICS KADIAN MORPHINE SULFATE ER PRIOR CLAIM FOR MORPHINE SULFATE SUSTAINED ACTION TABLET (MS CONTIN) WITHIN THE PAST 120 DAYS. ANTIBACTERIALS (EENT) BESIVANCE PRIOR CLAIM FOR CIPROFLOXACIN
MEDICAL ASSISTANCE BULLETIN
 ISSUE DATE January 6, 2016 SUBJECT EFFECTIVE DATE January 20, 2016 MEDICAL ASSISTANCE BULLETIN NUMBER *See below BY Prior Authorization of COPD Agents Pharmacy Service Leesa M. Allen, Deputy Secretary
ISSUE DATE January 6, 2016 SUBJECT EFFECTIVE DATE January 20, 2016 MEDICAL ASSISTANCE BULLETIN NUMBER *See below BY Prior Authorization of COPD Agents Pharmacy Service Leesa M. Allen, Deputy Secretary
ALOGLIPTIN STEP. Step Therapy Requirements Effective April 1, 2018
 Step Therapy Requirements Effective April 1, 2018 ALOGLIPTIN STEP alogliptin 12.5 mg tablet alogliptin 12.5 mg-metformin 1,000 mg tablet alogliptin 12.5 mg-metformin 500 mg tablet alogliptin 12.5 mg-pioglitazone
Step Therapy Requirements Effective April 1, 2018 ALOGLIPTIN STEP alogliptin 12.5 mg tablet alogliptin 12.5 mg-metformin 1,000 mg tablet alogliptin 12.5 mg-metformin 500 mg tablet alogliptin 12.5 mg-pioglitazone
