Comprehensive Research Plan: Inhaled corticosteroids + long-acting beta agonists (ICS+LABA) for the treatment of asthma
|
|
|
- Darrell Wilkinson
- 5 years ago
- Views:
Transcription
1 Comprehensive Research Plan: Inhaled corticosteroids + long-acting beta agonists (ICS+LABA) for the treatment of asthma Pharmacoepidemiology Unit July 10, Bond Street, Toronto ON, M5B 1W8 info@odprn.ca
2 2 Objectives: 1. To examine national and provincial trends in use of asthma drug therapies across Canada 2. To examine trends in use of asthma drug therapies for asthma in Ontario 3. To describe characteristics of asthma patients prescribed ICS/LABA combination products 4. To investigate course of therapy and adherence with asthma drugs in Ontario 5. To summarize any observational studies evaluating the comparative effectiveness of ICS/LABA combination products Objective 1: National and Provincial Trends in Asthma Therapies Study Population: Design: Time series analysis with quarterly time intervals Study period: National and provincial trends (IMS Compuscript): October 2009 to September 2013 Ontario trends among publically funded prescriptions (ODB): January 2000 to March 2013 Population: All provinces Data Sources: IMS Compuscript: aggregated data for all prescriptions dispensed at retail pharmacies across Canada Ontario Drug Benefit Database (ODB): individual level data for all publically funded prescriptions dispensed in Ontario to individuals aged 12 and older. Inclusion Criteria: All privately and publically-funded prescriptions dispensed in Canada for treatment of asthma, including: o ICS/LABA combination products Fluticasone+salmeterol (Advair, Advair Diskus) Budesonide+formoterol (Symbicort) Mometasone+formoterol (Zenhale) o Other therapies (inhaled corticosteroids, long-acting beta agonists, long acting muscarinic antagonist, short-acting beta agonists, short acting muscarinic antagonist, Leukotriene receptor antagonists)
3 3 Outcome(s) of Interest: Measured over entire study period (quarterly): Number of prescriptions dispensed Total cost of prescriptions Number of users (Ontario public drug plan only) Report: Overall rates of use by province National rates of use by drug Distribution of prescriptions by payer (public, private, cash, NIHB) Distribution of prescriptions by age (12-17, 18-64, 65+; Ontario public drug plan only) Limitations: The IMS data is only available at the prescription and unit level. Therefore, national and provincial trends in prescribing cannot differentiate by indication. Objective 2: Trends in Use of Asthma Drug Therapies for Asthma in Ontario Design: Time series analysis with annual time intervals Study period: April 2000 to March 2013 Data Source: Ontario Drug Benefit Database (ODB) Study Population: Inclusion Criteria: All publically-funded prescriptions for ICS/LABA combination products dispensed in Ontario All publically-funded prescriptions for other drug therapies (inhaled corticosteroids, long-acting beta agonists, long acting muscarinic antagonist, short-acting beta agonists, short acting muscarinic antagonist, Leukotriene receptor antagonists) Individuals aged 12+ at time of drug dispensing Individuals with a diagnosis of asthma using the ICES disease cohort
4 4 Outcome(s) of Interest: Measured over entire study period (annually) Number and rate of users of ICS/LABA products among asthma patients Number of ICS/LABA prescriptions dispensed to asthma patients Drug costs Stratify all analyses by: ICS/LABA combination product (Advair, Symbicort, Zenhale) Age (12-17, 18-64, 65+) Limitations: Asthma diagnosis defined via validated dataset developed at ICES. Although this dataset has 84% sensitivity and 76% specificity, there may be some misclassification. Objective 3: Characteristics of Asthma Patients treated with ICS/LABA Combination Products in Ontario Study Population: Design: Cross-sectional analysis Study period: April 2012 to March 2013 Data Sources: Ontario Drug Benefit Database (ODB) Canadian Institute for Health Information-Discharge Abstract Database (CIHI-DAD) National Ambulatory Care Reporting System Database (NACRS) Inclusion Criteria: All publically-funded beneficiaries of Ontario with asthma who are prescribed an ICS/LABA combination product Cohort #1: Individuals aged at time of ICS/LABA dispensing Cohort #2: Individuals aged at time of ICS/LABA dispensing Cohort #3: Individuals aged 66+ at time of ICS/LABA dispensing Past diagnosis with asthma (prior to cohort entry date) Cohort Entry Date: defined as date of first prescription for an ICS/LABA combination product, following 12 th, 18 th or 66 th birthday, over the study period. Index drug: Defined as the specific ICS/LABA product that was prescribed on cohort entry date (e.g. Advair, Symbicort or Zenhale)
5 5 Variables of Interest: For each cohort, measure: Number of asthma patients treated Number and rate of new ICS/LABA users o New users aged 66 and older defined as having no past ICS/LABA use in prior 365 days o New users <66 years of age defined as having a prescription for any drug in the past days and who didn t have a prescription for an ICS/LABA in the past 180 days Age at cohort entry date Proportion of patients who were male Proportion of patients residing in LTC at cohort entry Proportion of urban residents at cohort entry Socioeconomic status (measured using income quintiles at cohort entry date) Asthma severity o Defined using the prescribed treatment steps outlined in the Global Initiative for Asthma (GINA) 2014 report. Average cost of ICS/LABA prescriptions per person Number of puffs dispensed, per user Number of puffers dispensed, per user Past asthma maintenance therapy (past 1 year): o ICS o LABA o LAMA o LTRA o SABA o SAMA o Theophylline o Oral corticosteroids Past hospitalization or ED visit for asthma exacerbations (past 1 year) Stratify analyses by: Type of ICS/LABA combination product o Advair Diskus, Advair HFA, Symbicort, Zenhale Limitations: Individuals aged 65 are grouped in with those aged because although they have universal drug coverage, we have incomplete medical records for these patients in the prior year (e.g. when aged 64) since they were not eligible for public drug coverage at this time. Therefore, it is inappropriate to group these in with those aged 66+.
6 6 Objective 4: a. Adherence to therapy between those treated with combination ICS/LABA products and those treated with dual therapy of individual ICS and LABA components b. Typical course of therapy of asthma drugs in Ontario Design: Cohort Study Study period: April 2008 to March 2013 Accrual period: April 2008 to March 2012 Maximum follow-up date: March 2013 (1 year minimum follow-up) Data Sources: Ontario Drug Benefit Database (ODB) Canadian Institute for Health Information-Discharge Abstract Database (CIHI-DAD) National Ambulatory Care Reporting System Database (NACRS) Study Population: Inclusion Criteria: All publically-funded beneficiaries of Ontario with asthma who initiate ICS/LABA therapy. Combination therapy defined in 2 ways: o o Receipt of a combination product (e.g. Advair, Symbicort, Zenhale) Dual therapy of individual ICS and LABA components (e.g. receipt of separate ICS and LABA products for use at the same time). Include any combination of ICS and LABA products (not just those available in combination products) Cohort #1: Individuals aged at time of ICS/LABA dispensing Cohort #2: Individuals aged at time of ICS/LABA dispensing Cohort #3: Individuals aged 66+ at time of ICS/LABA dispensing Adults aged 12+ at time of ICS/LABA dispensing Restrict to those with asthma diagnosis prior to drug initiation New users aged 66 and older, defined as having no past ICS/LABA use in prior 365 days New users <66 years of age, defined as having a prescription for any drug in the past days and who didn t have a prescription for an ICS/LABA in the past 180 days
7 7 Outcomes of interest: Duration of ICS/LABA Therapy: DEFINITION 1: RELAXED DEFINITION OF ADHERENCE Define ongoing use of ICS/LABA therapy according to receipt of a subsequent prescription within 180 days of the prior prescription. DEFINITION 2: STRICT CLINICAL ADHERENCE Define ongoing use of ICS/LABA therapy according to receipt of a subsequent prescription within 1.5x days supply of the prior prescription. For each definition: Discontinuation of combination therapy defined as: o Discontinuation of combination product (set discontinuation date as date of last prescription) o Reducing to single agent therapy from dual therapy (set discontinuation date as the end of the period of continuous use for the first drug to be discontinued) Censor on: o End of study period o Death o For analysis stratified by ICS/LABA combination, censor on switch between products Report the following: o Total number of new ICS/LABA users o Number of ICS/LABA users with only 1 prescription before discontinuing Among patients with more than 1 prescription dispensed over period of continuous use Report the following: o Age o Gender o Residence in LTC o Urban vs rural residents o Socioeconomic status o Asthma severity Defined using the prescribed treatment steps outlined in the Global Initiative for Asthma (GINA) 2014 report. o Cost of therapy o Median duration of therapy o Percent adherent after: 1 year, 2 years o Number who discontinued due to death
8 8 Outcomes of interest (continued): o Past hospitalization or ED visit for asthma exacerbations (past 1 year) o In the 1 year prior to start of ICS/LABA therapy, report any prior asthma medications: No prior use asthma therapy ICS LABA LAMA LTRA SABA SAMA Theophylline Oral corticosteroids o Over period of ongoing use: Number of different ICS/LABA combinations prescribed (among those on combination therapy only) Concomitant use of other asthma treatment options ICS LABA LAMA LTRA SABA SABA Theophylline Oral corticosteroids o Kaplan Meier curves constructed and log-rank test used to test for differences o Cox Proportional Hazards models to calculate unadjusted and adjusted hazard ratio (with 95% CI) Adjust for age, sex, asthma severity Stratify above analysis by: Combination Products vs. Dual Therapy Type of ICS/LABA combination initiated (for those on combination therapy only) Limitations Individuals aged 65 are grouped in with those aged because although they have universal drug coverage, we have incomplete medical records for these patients in the prior year (e.g. when aged 64) since they were not eligible for public drug coverage at this time. Therefore, it is inappropriate to group these in with those aged 66+.
9 9 Objective 5: To summarize any observational studies evaluating the comparative effectiveness of ICS/LABA combination products Objective: Review of population-based studies investigating comparative effectiveness and/or safety of ICS/LABA combination products among patients with asthma Study Population Children and adults with asthma Observational studies Comparative effectiveness studies Safety studies Study Inclusion Criteria 1. English Language 2. Published in last 10 years Interventions ICS/LABA combination products Comparators ICS/LABA combination products (either as a single product or dual therapy) Outcomes Any reported outcomes
LAMA Products for the Treatment of COPD
 FINAL REPORT LAMA Products for the Treatment of COPD Pharmacoepidemiology Unit: Censored Final Report Tara Gomes, Andrea Gershon, Matthew Stanbrook, Ximena Camacho, Diana Martins, Samantha Singh, Zhan
FINAL REPORT LAMA Products for the Treatment of COPD Pharmacoepidemiology Unit: Censored Final Report Tara Gomes, Andrea Gershon, Matthew Stanbrook, Ximena Camacho, Diana Martins, Samantha Singh, Zhan
Mina Tadrous, Diana Martins, Zhan Yao, Kimberly Fernandes, Samantha Singh, Nikita Arora, David Juurlink, Muhammad Mamdani and Tara Gomes
 Cognitive Enhancers Pharmacoepidemiology Report: FINAL CENSORED Report Mina Tadrous, Diana Martins, Zhan Yao, Kimberly Fernandes, Samantha Singh, Nikita Arora, David Juurlink, Muhammad Mamdani and Tara
Cognitive Enhancers Pharmacoepidemiology Report: FINAL CENSORED Report Mina Tadrous, Diana Martins, Zhan Yao, Kimberly Fernandes, Samantha Singh, Nikita Arora, David Juurlink, Muhammad Mamdani and Tara
Drug Class Review: Long-acting muscarinic antagonists (LAMAs) for treatment of chronic obstructive pulmonary disease (COPD)
 Drug Class Review: Lng-acting muscarinic antagnists (LAMAs) fr treatment f chrnic bstructive pulmnary disease (COPD) Cmprehensive Research Plan: Pharmacepidemilgy Unit April 10 th, 2014 ODPRN Drug Class
Drug Class Review: Lng-acting muscarinic antagnists (LAMAs) fr treatment f chrnic bstructive pulmnary disease (COPD) Cmprehensive Research Plan: Pharmacepidemilgy Unit April 10 th, 2014 ODPRN Drug Class
April 10 th, Bond Street, Toronto ON, M5B 1W8
 Comprehensive Research Plan: Inhaled long-acting muscarinic antagonists (LAMAs; long-acting anticholinergics) for the treatment of chronic obstructive pulmonary disease (COPD) April 10 th, 2014 30 Bond
Comprehensive Research Plan: Inhaled long-acting muscarinic antagonists (LAMAs; long-acting anticholinergics) for the treatment of chronic obstructive pulmonary disease (COPD) April 10 th, 2014 30 Bond
Optum Research Database Proportion of asthma patients with prescription fills for rescue and controller medications by year,
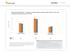 ASTHMA CHART 1 Optum Research Database Proportion of asthma patients with prescription fills for rescue and controller medications by year, 215-216 7 6 5 4 3 2 1 1 Controller medication 1 Rescue medication
ASTHMA CHART 1 Optum Research Database Proportion of asthma patients with prescription fills for rescue and controller medications by year, 215-216 7 6 5 4 3 2 1 1 Controller medication 1 Rescue medication
Cognitive enhancers for the treatment of Alzheimer s disease
 Cmprehensive Research Plan: Cgnitive enhancers fr the treatment f Alzheimer s disease Pharmacepidemilgy Unit February 13 th, 2015 30 Bnd Street, Trnt ON, M5B 1W8 www.dprn.ca inf@dprn.ca 2 ODPRN Drug Class
Cmprehensive Research Plan: Cgnitive enhancers fr the treatment f Alzheimer s disease Pharmacepidemilgy Unit February 13 th, 2015 30 Bnd Street, Trnt ON, M5B 1W8 www.dprn.ca inf@dprn.ca 2 ODPRN Drug Class
COPD Medications Coverage Summary Non-Insured Health Benefits Coverage SABA Bricanyl turbuhaler Yes Yes
 COPD Medications Coverage Summary Drug Non-Insured Health Benefits Coverage SABA Bricanyl turbuhaler Yes Yes Ventolin MDI + generics Yes Yes Ventolin Diskus NO NO Yukon Pharmacare/Chronic Disease Program
COPD Medications Coverage Summary Drug Non-Insured Health Benefits Coverage SABA Bricanyl turbuhaler Yes Yes Ventolin MDI + generics Yes Yes Ventolin Diskus NO NO Yukon Pharmacare/Chronic Disease Program
Drug Class Monograph
 Drug Class Monograph Class: Inhaled Corticosteroids Drugs: Aerospan (flunisolide), Advair Diskus, Advair HFA (fluticasone/salmeterol), Alvesco (ciclesonide), Arnuity Ellipta (fluticasone furoate), Asmanex
Drug Class Monograph Class: Inhaled Corticosteroids Drugs: Aerospan (flunisolide), Advair Diskus, Advair HFA (fluticasone/salmeterol), Alvesco (ciclesonide), Arnuity Ellipta (fluticasone furoate), Asmanex
Long Term Care Formulary RS -29
 RESTRICTED USE Asthma/COPD Management 1 of 6 PROTOCOL: Asthma Glossary of Medication Acronyms: SABA: short-acting beta agonist (e.g. salbutamol) SABD: short-acting bronchodilator (e.g. ipratropium or SABA)
RESTRICTED USE Asthma/COPD Management 1 of 6 PROTOCOL: Asthma Glossary of Medication Acronyms: SABA: short-acting beta agonist (e.g. salbutamol) SABD: short-acting bronchodilator (e.g. ipratropium or SABA)
Asthma Population Management: Identifying Persistent Asthma, Defining High Risk Asthma, and Measuring Quality of Asthma Care
 Asthma Population Management: Identifying Persistent Asthma, Defining High Risk Asthma, and Measuring Quality of Asthma Care Michael Schatz, MD, MS Allergy Department Kaiser Permanente, San Diego, CA Constructs
Asthma Population Management: Identifying Persistent Asthma, Defining High Risk Asthma, and Measuring Quality of Asthma Care Michael Schatz, MD, MS Allergy Department Kaiser Permanente, San Diego, CA Constructs
TRELEGY ELLIPTA (fluticasone-umeclidinium-vilanterol) aerosol powder
 TRELEGY ELLIPTA (fluticasone-umeclidinium-vilanterol) aerosol powder Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific
TRELEGY ELLIPTA (fluticasone-umeclidinium-vilanterol) aerosol powder Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific
Exposure to potentially inappropriate medications among long-term care residents with cognitive impairment in Ontario:
 Exposure to potentially inappropriate medications among long-term care residents with cognitive impairment in Ontario: Is there an association with frailty? Laura Maclagan, Jun Guan, Sima Gandhi, Colleen
Exposure to potentially inappropriate medications among long-term care residents with cognitive impairment in Ontario: Is there an association with frailty? Laura Maclagan, Jun Guan, Sima Gandhi, Colleen
Outcomes: Initially, our primary definitions of pneumonia was severe pneumonia, where the subject was hospitalized
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Adjustment of Inhaled Controller Therapy of Asthma in the Yellow Zone, Based on the Inhaler Product Used in the Green Zone Age 16 Years and Older
 Adjustment of Inhaled Controller Therapy of Asthma in the Yellow Zone, Based on the Inhaler Product Used in the Green Zone Age 16 Years and Older The Canadian Thoracic Society and other international asthma
Adjustment of Inhaled Controller Therapy of Asthma in the Yellow Zone, Based on the Inhaler Product Used in the Green Zone Age 16 Years and Older The Canadian Thoracic Society and other international asthma
SYNOPSIS A two-stage randomized, open-label, parallel group, phase III, multicenter, 7-month study to assess the efficacy and safety of SYMBICORT
 Drug product: Drug substance(s): Edition No.: Study code: SYMBICORT pmdi 160/4.5 g Budesonide/formoterol D5896C00005 Date: 8 May 2006 SYNOPSIS A two-stage randomized, open-label, parallel group, phase
Drug product: Drug substance(s): Edition No.: Study code: SYMBICORT pmdi 160/4.5 g Budesonide/formoterol D5896C00005 Date: 8 May 2006 SYNOPSIS A two-stage randomized, open-label, parallel group, phase
MEDICAL COVERAGE GUIDELINES ORIGINAL EFFECTIVE DATE: 07/05/18 SECTION: DRUGS LAST REVIEW DATE: LAST CRITERIA REVISION DATE: ARCHIVE DATE:
 CINQAIR (reslizumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
CINQAIR (reslizumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
Global Initiative for Asthma (GINA) What s new in GINA 2015?
 Global Initiative for Asthma (GINA) What s new in GINA 2015? GINA Global Strategy for Asthma Management and Prevention What s new in GINA 2015 (1) Add-on tiotropium by soft-mist inhaler is a new other
Global Initiative for Asthma (GINA) What s new in GINA 2015? GINA Global Strategy for Asthma Management and Prevention What s new in GINA 2015 (1) Add-on tiotropium by soft-mist inhaler is a new other
Models and Methods in Health Economics and the Decision-Making Process. Monday, October 17, 2016 MaRS Discovery District, Toronto
 Models and Methods in Health Economics and the Decision-Making Process Monday, October 17, 2016 MaRS Discovery District, Toronto Meet the Panel Moderator: Murray Krahn (University of Toronto) Speakers:
Models and Methods in Health Economics and the Decision-Making Process Monday, October 17, 2016 MaRS Discovery District, Toronto Meet the Panel Moderator: Murray Krahn (University of Toronto) Speakers:
THIS TALK STATINS REDUCE: Immortal time bias Immeasurable time bias Time-window bias TIME-RELATED BIASES IN PHARMACOEPIDEMIOLOGY
 TIME-RELATED BIASES IN PHARMACOEPIDEMIOLOGY Samy Suissa McGill University and Jewish General Hospital Montreal, Canada ISPE mid-year meeting, Miami April 22, 2012 STATINS REDUCE: JAMA 2000: Fracture rate
TIME-RELATED BIASES IN PHARMACOEPIDEMIOLOGY Samy Suissa McGill University and Jewish General Hospital Montreal, Canada ISPE mid-year meeting, Miami April 22, 2012 STATINS REDUCE: JAMA 2000: Fracture rate
Inhaled Corticosteroids Drug Class Prior Authorization Protocol
 Inhaled Corticosteroids Drug Class Prior Authorization Protocol Line of Business: Medi-Cal P&T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has been developed through review
Inhaled Corticosteroids Drug Class Prior Authorization Protocol Line of Business: Medi-Cal P&T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has been developed through review
SYNOPSIS. Study centre(s) A total of 91 centres across Canada participated in this study.
 Drug product: Symbicort Turbuhaler Drug substance(s): Budesonide/formoterol Edition No.: 1 Date: August 22, 2007 SYNOPSIS A comparison of Symbicort SMART a (Symbicort 200 Turbuhaler 1 inhalation b.i.d.
Drug product: Symbicort Turbuhaler Drug substance(s): Budesonide/formoterol Edition No.: 1 Date: August 22, 2007 SYNOPSIS A comparison of Symbicort SMART a (Symbicort 200 Turbuhaler 1 inhalation b.i.d.
Inhaled Corticosteroids Drug Class Prior Authorization Protocol
 Inhaled Corticosteroids Drug Class Prior Authorization Protocol Line of Business: Medicaid P&T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has been developed through review
Inhaled Corticosteroids Drug Class Prior Authorization Protocol Line of Business: Medicaid P&T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has been developed through review
Canadian Expert Drug Advisory Committee Final Recommendation Plain Language Version
 Canadian Expert Drug Advisory Committee Final Recommendation Plain Language Version MOMETASONE FUROATE/FORMOTEROL FUMARATE DIHYDRATE INHALATION AEROSOL (Zenhale Merck Canada Inc.) Indication: Asthma Maintenance
Canadian Expert Drug Advisory Committee Final Recommendation Plain Language Version MOMETASONE FUROATE/FORMOTEROL FUMARATE DIHYDRATE INHALATION AEROSOL (Zenhale Merck Canada Inc.) Indication: Asthma Maintenance
Consolidated Comprehensive Research Plan: Pharmacologic Treatment of Attention Deficit Hyperactivity Disorder in Adults
 FINAL Consolidated Comprehensive Research Plan: Pharmacologic Treatment of Attention Deficit Hyperactivity Disorder in Adults April 29 th, 2015 30 Bond Street, Toronto ON, M5B 1W8 www.odprn.ca info@odprn.ca
FINAL Consolidated Comprehensive Research Plan: Pharmacologic Treatment of Attention Deficit Hyperactivity Disorder in Adults April 29 th, 2015 30 Bond Street, Toronto ON, M5B 1W8 www.odprn.ca info@odprn.ca
GINA. At-A-Glance Asthma Management Reference. for adults, adolescents and children 6 11 years. Updated 2017
 GINA At-A-Glance Asthma Management Reference for adults, adolescents and children 6 11 years Updated 2017 This resource should be used in conjunction with the Global Strategy for Asthma Management and
GINA At-A-Glance Asthma Management Reference for adults, adolescents and children 6 11 years Updated 2017 This resource should be used in conjunction with the Global Strategy for Asthma Management and
Estimates and predictors of health care costs of esophageal adenocarcinoma: a population-based cohort study
 Thein et al. BMC Cancer (2018) 18:694 https://doi.org/10.1186/s12885-018-4620-2 RESEARCH ARTICLE Estimates and predictors of health care costs of esophageal adenocarcinoma: a population-based cohort study
Thein et al. BMC Cancer (2018) 18:694 https://doi.org/10.1186/s12885-018-4620-2 RESEARCH ARTICLE Estimates and predictors of health care costs of esophageal adenocarcinoma: a population-based cohort study
Evaluation of Asthma Management in Middle EAst North Africa Adult population
 STUDY REPORT SUMMARY Evaluation of Asthma Management in Middle EAst North Africa Adult population Descriptive study on the management of asthma in an asthmatic Middle East Africa adult population Background/Rationale:
STUDY REPORT SUMMARY Evaluation of Asthma Management in Middle EAst North Africa Adult population Descriptive study on the management of asthma in an asthmatic Middle East Africa adult population Background/Rationale:
Trends in Glucocorticoid-Induced Osteoporosis Management Among Seniors in Ontario,
 Trends in Glucocorticoid-Induced Osteoporosis Management Among Seniors in Ontario, 1996-2012 Jordan M Albaum 1, Linda E Lévesque 2, Andrea S Gershon 3, Yan Yun Liu 1, Suzanne M Cadarette 1 1 Leslie Dan
Trends in Glucocorticoid-Induced Osteoporosis Management Among Seniors in Ontario, 1996-2012 Jordan M Albaum 1, Linda E Lévesque 2, Andrea S Gershon 3, Yan Yun Liu 1, Suzanne M Cadarette 1 1 Leslie Dan
APPENDIX 1 Printable point-of-care tables Asthma Action Plan Yellow Zone Formulation Table Region: Europe
 APPENDIX 1 Printable point-of-care tables Asthma Action Plan Yellow Zone Formulation Table Region: Europe Instructions: Print on 8.5 x14 (216 x 279 mm) paper (Legal size) Medication in Green Zone Change
APPENDIX 1 Printable point-of-care tables Asthma Action Plan Yellow Zone Formulation Table Region: Europe Instructions: Print on 8.5 x14 (216 x 279 mm) paper (Legal size) Medication in Green Zone Change
Atypical Antipsychotic Use for the Behavioural and Psychological Symptoms of Dementia in the Elderly
 Overall Comprehensive Research Plan: Atypical Antipsychotic Use for the Behavioural and Psychological Symptoms of Dementia in the Elderly October 9, 2014 30 Bond Street, Toronto ON, M5B 1W8 www.odprn.ca
Overall Comprehensive Research Plan: Atypical Antipsychotic Use for the Behavioural and Psychological Symptoms of Dementia in the Elderly October 9, 2014 30 Bond Street, Toronto ON, M5B 1W8 www.odprn.ca
Disclosure Statement. Epidemiological Data
 EVALUATION OF THE MEDICATION UTILIZATION OF COPD PATIENTS AT THE MIAMI VA HEALTHCARE SYSTEM Simone Edgerton, PharmD. PGY 1 Pharmacy Resident Miami VA Healthcare System Miami, Florida Simone.edgerton2@va.gov
EVALUATION OF THE MEDICATION UTILIZATION OF COPD PATIENTS AT THE MIAMI VA HEALTHCARE SYSTEM Simone Edgerton, PharmD. PGY 1 Pharmacy Resident Miami VA Healthcare System Miami, Florida Simone.edgerton2@va.gov
Identifying and evaluating patterns of prescription opioid use and associated risks in Ontario, Canada Gomes, T.
 UvA-DARE (Digital Academic Repository) Identifying and evaluating patterns of prescription opioid use and associated risks in Ontario, Canada Gomes, T. Link to publication Citation for published version
UvA-DARE (Digital Academic Repository) Identifying and evaluating patterns of prescription opioid use and associated risks in Ontario, Canada Gomes, T. Link to publication Citation for published version
Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing
 Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing Diana M. Sobieraj, PharmD, BCPS Assistant Professor University of Connecticut School
Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing Diana M. Sobieraj, PharmD, BCPS Assistant Professor University of Connecticut School
Interventions to improve adherence to inhaled steroids for asthma. Respiratory department
 Interventions to improve adherence to inhaled steroids for asthma Respiratory department Content Overview Research References Overview Asthma is a chronic breathing condition that affects more than 300
Interventions to improve adherence to inhaled steroids for asthma Respiratory department Content Overview Research References Overview Asthma is a chronic breathing condition that affects more than 300
Disclosure. Case. Objectives. Case Continued. Inhalers. Asthma: A GINA Update to the NAEPP 2007 Guidelines 1/20/2015
 Disclosure Asthma: A GINA Update to the NAEPP 2007 Guidelines Robert (RC) Hellinga, Pharm.D. PGY 1 Pharmacy Resident Wolfson Children s Hospital/Baptist Health I do not have a vested interest in or affiliation
Disclosure Asthma: A GINA Update to the NAEPP 2007 Guidelines Robert (RC) Hellinga, Pharm.D. PGY 1 Pharmacy Resident Wolfson Children s Hospital/Baptist Health I do not have a vested interest in or affiliation
Clinical Policy: Dupilumab (Dupixent) Reference Number: ERX.SPA.49 Effective Date:
 Clinical Policy: (Dupixent) Reference Number: ERX.SPA.49 Effective Date: 06.01.17 Last Review Date: 02.19 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Dupixent) Reference Number: ERX.SPA.49 Effective Date: 06.01.17 Last Review Date: 02.19 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Alberta Childhood Asthma Pathway for Primary Care
 Asthma Diagnosis Box 1 Diagnosis: Based on symptom pattern, careful and thorough history of symptoms (wheeze, cough, night waking and activity limitations), and assessment of family history of asthma and
Asthma Diagnosis Box 1 Diagnosis: Based on symptom pattern, careful and thorough history of symptoms (wheeze, cough, night waking and activity limitations), and assessment of family history of asthma and
Lead team presentation: Roflumilast for treating chronic obstructive pulmonary disease [ID984]
![Lead team presentation: Roflumilast for treating chronic obstructive pulmonary disease [ID984] Lead team presentation: Roflumilast for treating chronic obstructive pulmonary disease [ID984]](/thumbs/85/91889043.jpg) Lead team presentation: Roflumilast for treating chronic obstructive pulmonary disease [ID984] 1 st Appraisal Committee meeting Background & Clinical Effectiveness John McMurray 11 th January 2016 For
Lead team presentation: Roflumilast for treating chronic obstructive pulmonary disease [ID984] 1 st Appraisal Committee meeting Background & Clinical Effectiveness John McMurray 11 th January 2016 For
Up in FLAMES: Stable Chronic Obstructive Pulmonary Disease (COPD) Management. Colleen Sakon, PharmD BCPS September 27, 2018
 Up in FLAMES: Stable Chronic Obstructive Pulmonary Disease (COPD) Management Colleen Sakon, PharmD BCPS September 27, 2018 Disclosures I have no actual or potential conflicts of interest 2 Objectives Summarize
Up in FLAMES: Stable Chronic Obstructive Pulmonary Disease (COPD) Management Colleen Sakon, PharmD BCPS September 27, 2018 Disclosures I have no actual or potential conflicts of interest 2 Objectives Summarize
Policy Evaluation: Step Therapy Prior Authorization of Combination Inhaled Corticosteroid / Long-Acting Beta-Agonists
 Drug Use Research & Management Program OHA Division of Medical Assistance Programs 500 Summer Street NE, E35; Salem, OR 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Policy Evaluation: Step Therapy Prior
Drug Use Research & Management Program OHA Division of Medical Assistance Programs 500 Summer Street NE, E35; Salem, OR 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Policy Evaluation: Step Therapy Prior
Global Initiative for Asthma (GINA) What s new in GINA 2016?
 Global Initiative for Asthma (GINA) What s new in GINA 2016? GINA Global Strategy for Asthma Management and Prevention GINA: A Brief History Established in 1993 Collaboration between NHLBI and WHO Multiple
Global Initiative for Asthma (GINA) What s new in GINA 2016? GINA Global Strategy for Asthma Management and Prevention GINA: A Brief History Established in 1993 Collaboration between NHLBI and WHO Multiple
Stakeholder Comments and Ontario Drug Policy Research Network (ODPRN) Response:
 30 Bond Street, Toronto ON, M5B 1W8 www.odprn.ca info@odprn.ca Stakeholder Comments and Ontario Drug Policy Research Network (ODPRN) Response: Reimbursement Options Report April 7 th, 2014 2 COMMENT We
30 Bond Street, Toronto ON, M5B 1W8 www.odprn.ca info@odprn.ca Stakeholder Comments and Ontario Drug Policy Research Network (ODPRN) Response: Reimbursement Options Report April 7 th, 2014 2 COMMENT We
FASENRA (benralizumab)
 FASENRA (benralizumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
FASENRA (benralizumab) Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
Drugs for Overactive Bladder (OAB)
 ril 2014 Drugs for Overactive Bladder (OAB) FINAL CONSOLIDATED COMPREHENSIVE RESEARCH PLAN July 2015 FINAL COMPREHENSIVE RESEARCH PLAN 2 A. Introduction The objective of the drug class review on drugs
ril 2014 Drugs for Overactive Bladder (OAB) FINAL CONSOLIDATED COMPREHENSIVE RESEARCH PLAN July 2015 FINAL COMPREHENSIVE RESEARCH PLAN 2 A. Introduction The objective of the drug class review on drugs
The effect of surgeon volume on procedure selection in non-small cell lung cancer surgeries. Dr. Christian Finley MD MPH FRCSC McMaster University
 The effect of surgeon volume on procedure selection in non-small cell lung cancer surgeries Dr. Christian Finley MD MPH FRCSC McMaster University Disclosures I have no conflict of interest disclosures
The effect of surgeon volume on procedure selection in non-small cell lung cancer surgeries Dr. Christian Finley MD MPH FRCSC McMaster University Disclosures I have no conflict of interest disclosures
FINAL REPORT Inhaled corticosteroids in combination with long-acting beta2-agonist (ICS+LABA) for chronic obstructive pulmonary disease (COPD)
 FINAL REPORT Inhaled corticosteroids in combination with long-acting beta2-agonist (ICS+LABA) for chronic obstructive pulmonary disease (COPD) Pharmacoeconomic Unit December 29, 2014 Note Some details
FINAL REPORT Inhaled corticosteroids in combination with long-acting beta2-agonist (ICS+LABA) for chronic obstructive pulmonary disease (COPD) Pharmacoeconomic Unit December 29, 2014 Note Some details
Global Initiative for Asthma (GINA) What s new in GINA 2017?
 Global Initiative for Asthma (GINA) GINA Global Strategy for Asthma Management and Prevention Asthma-COPD overlap The word syndrome has been removed from the previous term asthma-copd overlap syndrome
Global Initiative for Asthma (GINA) GINA Global Strategy for Asthma Management and Prevention Asthma-COPD overlap The word syndrome has been removed from the previous term asthma-copd overlap syndrome
Stakeholder Comments and Responses for
 Stakeholder Comments and Responses for Comprehensive Research Plan: Treatment of ADHD in Adults Consolidated Report April 29 th, 2015 30 Bond Street, Toronto ON, M5B 1W8 www.odprn.ca info@odprn.ca 2 OVERALL
Stakeholder Comments and Responses for Comprehensive Research Plan: Treatment of ADHD in Adults Consolidated Report April 29 th, 2015 30 Bond Street, Toronto ON, M5B 1W8 www.odprn.ca info@odprn.ca 2 OVERALL
CHARM ASTHMA TREATMENT GUIDELINE
 NHS City and Hackney Prescribing Guidelines Adults ( 12 years of age) CHARM ASTHMA TREATMENT GUIDELINE Written by: Hetal Dhruve (Specialist Respiratory Pharmacist, City and Hackney CCG) Checked by: Prof
NHS City and Hackney Prescribing Guidelines Adults ( 12 years of age) CHARM ASTHMA TREATMENT GUIDELINE Written by: Hetal Dhruve (Specialist Respiratory Pharmacist, City and Hackney CCG) Checked by: Prof
GSK Medicine: salmeterol, Salmeterol+Fluticasone proprionate, fluticasone propionate, beclomethasone
 GSK Medicine: salmeterol, Salmeterol+Fluticasone proprionate, fluticasone propionate, beclomethasone Study No.: WWE111984/WEUSRTP2640/EPI40528 Title: The Asthma Death Case Control Study (ADCCS): Association
GSK Medicine: salmeterol, Salmeterol+Fluticasone proprionate, fluticasone propionate, beclomethasone Study No.: WWE111984/WEUSRTP2640/EPI40528 Title: The Asthma Death Case Control Study (ADCCS): Association
Asthma Treatment Guideline for Adults (aged 17 and over)
 Asthma Treatment Guideline for Adults (aged 17 and over) Sharon Andrew MLCSU January 2019 (Review date 0 January 2022) VERSION CONTROL. Please access via the LMMG website to ensure that the correct version
Asthma Treatment Guideline for Adults (aged 17 and over) Sharon Andrew MLCSU January 2019 (Review date 0 January 2022) VERSION CONTROL. Please access via the LMMG website to ensure that the correct version
The methodology behind GINA and EPR-3 medication recommendations: Stepwise treatment in asthma
 The methodology behind GINA and EPR-3 medication recommendations: Stepwise treatment in asthma Maureen George PhD RN AE-C FAAN Columbia University mg3656@cumc.columbia.edu Faculty Disclosures Maureen George
The methodology behind GINA and EPR-3 medication recommendations: Stepwise treatment in asthma Maureen George PhD RN AE-C FAAN Columbia University mg3656@cumc.columbia.edu Faculty Disclosures Maureen George
Stepping down asthma treatment guidelines
 Stepping down asthma treatment guidelines The potential for inhaled corticosteroids (ICS) to cause dose-related side-effects has led to asthma management guidelines recommending a dose reduction once asthma
Stepping down asthma treatment guidelines The potential for inhaled corticosteroids (ICS) to cause dose-related side-effects has led to asthma management guidelines recommending a dose reduction once asthma
Changing patterns of sedative use over time in older adults in Ontario
 Changing patterns of sedative use over time in older adults in Ontario Andrea Iaboni, MD, DPhil, FRCPC Toronto Rehabilitation Institute University Health Network Faculty/Presenter Disclosure Faculty: Andrea
Changing patterns of sedative use over time in older adults in Ontario Andrea Iaboni, MD, DPhil, FRCPC Toronto Rehabilitation Institute University Health Network Faculty/Presenter Disclosure Faculty: Andrea
Pharmacy Medical Policy Asthma and Chronic Obstructive Pulmonary Disease Medication Management
 Pharmacy Medical Policy Asthma and Chronic Obstructive Pulmonary Disease Medication Management Table of Contents Policy: Commercial Information Pertaining to All Policies Endnotes Policy: Medicare References
Pharmacy Medical Policy Asthma and Chronic Obstructive Pulmonary Disease Medication Management Table of Contents Policy: Commercial Information Pertaining to All Policies Endnotes Policy: Medicare References
Key features and changes to these four components of asthma care include:
 Guidelines for the Diagnosis and Management of Asthma in Adults Clinical Practice Guideline MedStar Health These guidelines are provided to assist physicians and other clinicians in making decisions regarding
Guidelines for the Diagnosis and Management of Asthma in Adults Clinical Practice Guideline MedStar Health These guidelines are provided to assist physicians and other clinicians in making decisions regarding
Three s Company - The role of triple therapy in chronic obstructive pulmonary
 Three s Company - The role of triple therapy in chronic obstructive pulmonary disease (COPD) October 26 th, 2018 Zahava Picado, PharmD PGY1 Pharmacy Resident Central Texas Veterans Healthcare System Zahava.Picado@va.gov
Three s Company - The role of triple therapy in chronic obstructive pulmonary disease (COPD) October 26 th, 2018 Zahava Picado, PharmD PGY1 Pharmacy Resident Central Texas Veterans Healthcare System Zahava.Picado@va.gov
Project Cohort EXPOSED GROUP
 Project Cohort Study Design Matched cohort study EXPOSED GROUP Index Event / Inclusion Criteria FOR EXPOSED GROUP Index Event: First eligible prescription for a study medication (S_fin, S_dut). Note: the
Project Cohort Study Design Matched cohort study EXPOSED GROUP Index Event / Inclusion Criteria FOR EXPOSED GROUP Index Event: First eligible prescription for a study medication (S_fin, S_dut). Note: the
Real-life effectiveness evaluation of budesonide/formoterol (BF) Spiromax for the management of asthma and COPD
 Study protocol Study protocol Real-life effectiveness evaluation of budesonide/formoterol (BF) Spiromax for the management of asthma and COPD Four complimentary post-marketing retrospective, observational
Study protocol Study protocol Real-life effectiveness evaluation of budesonide/formoterol (BF) Spiromax for the management of asthma and COPD Four complimentary post-marketing retrospective, observational
Clinical Policy: Omalizumab (Xolair) Reference Number: ERX.SPA.141 Effective Date: Last Review Date: 08.17
 Clinical Policy: (Xolair) Reference Number: ERX.SPA.141 Effective Date: 03.01.14 Last Review Date: 08.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Xolair) Reference Number: ERX.SPA.141 Effective Date: 03.01.14 Last Review Date: 08.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
12/18/2017. Disclosures. Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing
 Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing Diana M. Sobieraj, PharmD, BCPS Assistant Professor University of Connecticut School
Asthma Management Updates: A Focus on Long-acting Muscarinic Antagonists and Intermittent Inhaled Corticosteroid Dosing Diana M. Sobieraj, PharmD, BCPS Assistant Professor University of Connecticut School
Using Inhaled Corticosteroids as Needed for Asthma: giving patients relief or leaving them breathless?
 Using Inhaled Corticosteroids as Needed for Asthma: giving patients relief or leaving them breathless? Lindsay Thomas, Pharm.D. PGY2 Ambulatory Care Resident Department of Pharmacotherapy and Pharmacy
Using Inhaled Corticosteroids as Needed for Asthma: giving patients relief or leaving them breathless? Lindsay Thomas, Pharm.D. PGY2 Ambulatory Care Resident Department of Pharmacotherapy and Pharmacy
benralizumab (Fasenra )
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
The University of Mississippi School of Pharmacy
 LONG TERM PERSISTENCE WITH ACEI/ARB THERAPY AFTER ACUTE MYOCARDIAL INFARCTION: AN ANALYSIS OF THE 2006-2007 MEDICARE 5% NATIONAL SAMPLE DATA Lokhandwala T. MS, Yang Y. PhD, Thumula V. MS, Bentley J.P.
LONG TERM PERSISTENCE WITH ACEI/ARB THERAPY AFTER ACUTE MYOCARDIAL INFARCTION: AN ANALYSIS OF THE 2006-2007 MEDICARE 5% NATIONAL SAMPLE DATA Lokhandwala T. MS, Yang Y. PhD, Thumula V. MS, Bentley J.P.
Surveillance report Published: 6 April 2016 nice.org.uk. NICE All rights reserved.
 Surveillance report 2016 Chronic obstructive pulmonary disease in over 16s: diagnosis and management (2010) NICE guideline CG101 Surveillance report Published: 6 April 2016 nice.org.uk NICE 2016. All rights
Surveillance report 2016 Chronic obstructive pulmonary disease in over 16s: diagnosis and management (2010) NICE guideline CG101 Surveillance report Published: 6 April 2016 nice.org.uk NICE 2016. All rights
AHA Clinical Science Special Report: November 10, 2015
 www.canheart.ca High-density lipoprotein cholesterol and cause-specific mortality: A population-based study of more than 630,000 individuals without prior cardiovascular conditions Dennis T. Ko, MD, MSc;
www.canheart.ca High-density lipoprotein cholesterol and cause-specific mortality: A population-based study of more than 630,000 individuals without prior cardiovascular conditions Dennis T. Ko, MD, MSc;
Greater Manchester Asthma Management Plan 2018 Inhaler therapy options for adult patients (18 and over) with asthma
 Greater Manchester Asthma Management Plan 2018 Inhaler therapy options for adult patients (18 and over) with asthma Non-pharmacological options for ALL patients, consider at ALL stages Make sure diagnosis
Greater Manchester Asthma Management Plan 2018 Inhaler therapy options for adult patients (18 and over) with asthma Non-pharmacological options for ALL patients, consider at ALL stages Make sure diagnosis
Site Training. The 39 th Multicenter Airway Research Collaboration (MARC-39) Study
 Site Training The 39 th Multicenter Airway Research Collaboration (MARC-39) Study 1 Study Leadership Kohei Hasegawa, MD, MPH (PI) Carlos Camargo, MD, DrPH (Co-I) Project Coordination: Catalina Gimenez-Zapiola
Site Training The 39 th Multicenter Airway Research Collaboration (MARC-39) Study 1 Study Leadership Kohei Hasegawa, MD, MPH (PI) Carlos Camargo, MD, DrPH (Co-I) Project Coordination: Catalina Gimenez-Zapiola
David M Kern 1*, Jill Davis 2, Setareh A Williams 3, Ozgur Tunceli 1, Bingcao Wu 1, Sally Hollis 4, Charlie Strange 5 and Frank Trudo 2
 Kern et al. Respiratory Research (2015) 16:52 DOI 10.1186/s12931-015-0210-x RESEARCH Open Access Comparative effectiveness of budesonide/ formoterol combination and fluticasone/ salmeterol combination
Kern et al. Respiratory Research (2015) 16:52 DOI 10.1186/s12931-015-0210-x RESEARCH Open Access Comparative effectiveness of budesonide/ formoterol combination and fluticasone/ salmeterol combination
Position within the Organisation
 ASTHMA TREATMENT GUIDELINES Document Description Document Type Service Application Guidelines All healthcare professionals(hcps) caring for patients with asthma Version 4.0 Ratification date September
ASTHMA TREATMENT GUIDELINES Document Description Document Type Service Application Guidelines All healthcare professionals(hcps) caring for patients with asthma Version 4.0 Ratification date September
Decline In lung-function Among Patients with chronic obstructive Lung disease On maintenance therapy (DIAPLO)
 Version V1.4 Decline In lung-function Among Patients with chronic obstructive Lung disease On maintenance therapy (DIAPLO) An observational study evaluating the benefits of early intervention with maintenance
Version V1.4 Decline In lung-function Among Patients with chronic obstructive Lung disease On maintenance therapy (DIAPLO) An observational study evaluating the benefits of early intervention with maintenance
Who can get most benefit
 Who can get most benefit from tiotropium in asthma? Y-M. Oh Asan Medical Center Univ. of Ulsan College of Medicine Seoul, Korea Tiotripium for Asthma 1 New in GINA 2015 Add-on tiotropium by soft-mist inhaler
Who can get most benefit from tiotropium in asthma? Y-M. Oh Asan Medical Center Univ. of Ulsan College of Medicine Seoul, Korea Tiotripium for Asthma 1 New in GINA 2015 Add-on tiotropium by soft-mist inhaler
Methods to Diagnose Adherence Status
 Methods to Diagnose Adherence Status March 4, 2014 Andrew G Weinstein MD Associate Clinical Professor Pediatrics Jefferson Medical College President, Adherence Management Systems Disclosures President,
Methods to Diagnose Adherence Status March 4, 2014 Andrew G Weinstein MD Associate Clinical Professor Pediatrics Jefferson Medical College President, Adherence Management Systems Disclosures President,
Scottish Medicines Consortium
 Scottish Medicines Consortium budesonide/formoterol 100/6, 200/6 turbohaler (Symbicort SMART ) No. (362/07) Astra Zeneca UK Limited 9 March 2007 (Issued May 2007) The Scottish Medicines Consortium (SMC)
Scottish Medicines Consortium budesonide/formoterol 100/6, 200/6 turbohaler (Symbicort SMART ) No. (362/07) Astra Zeneca UK Limited 9 March 2007 (Issued May 2007) The Scottish Medicines Consortium (SMC)
Statistical Analysis Plan
 Drug utilisation study of new users of fluticasone furoate / vilanterol (FF/VI) in the primary care setting: UK Clinical Practice Research Datalink (CPRD) study Statistical Analysis Plan Subject: 205052:
Drug utilisation study of new users of fluticasone furoate / vilanterol (FF/VI) in the primary care setting: UK Clinical Practice Research Datalink (CPRD) study Statistical Analysis Plan Subject: 205052:
Beta-Blocker Use and Morbidity from Chronic Lung Disease in Patients Undertaking Pulmonary Rehabilitation
 Beta-Blocker Use and Morbidity from Chronic Lung Disease in Patients Undertaking Pulmonary Rehabilitation by Robert Gabriel Varadi A thesis submitted in conformity with the requirements for the degree
Beta-Blocker Use and Morbidity from Chronic Lung Disease in Patients Undertaking Pulmonary Rehabilitation by Robert Gabriel Varadi A thesis submitted in conformity with the requirements for the degree
DOD PHARMACY AND THERAPEUTICS COMMITTEE RECOMMENDATIONS INFORMATION FOR THE UNIFORM FORMULARY BENEFICIARY ADVISORY PANEL
 DOD PHARMACY AND THERAPEUTICS COMMITTEE RECOMMENDATIONS INFORMATION FOR THE UNIFORM FORMULARY BENEFICIARY ADVISORY PANEL I. Uniform Formulary Review Process Under 10 U.S.C. 1074g, as implemented by 32
DOD PHARMACY AND THERAPEUTICS COMMITTEE RECOMMENDATIONS INFORMATION FOR THE UNIFORM FORMULARY BENEFICIARY ADVISORY PANEL I. Uniform Formulary Review Process Under 10 U.S.C. 1074g, as implemented by 32
Estimated date of first patient enrolled Q III/IV Estimated date of last patient completed Q3 2015
 PROTOCOL SYNOPSIS A 26 week, randomized, double-blind, parallel-group, active controlled, multicenter, multinational safety study evaluating the risk of serious asthma-related events during treatment with
PROTOCOL SYNOPSIS A 26 week, randomized, double-blind, parallel-group, active controlled, multicenter, multinational safety study evaluating the risk of serious asthma-related events during treatment with
CHRONIC OBSTRUCTIVE PULMONARY DISEASE (COPD) TREATMENT GUIDELINES
 CHRONIC OBSTRUCTIVE PULMONARY DISEASE (COPD) TREATMENT GUIDELINES Document Description Document Type Service Application Version Guidelines All healthcare professionals(hcps) caring for patients with asthma
CHRONIC OBSTRUCTIVE PULMONARY DISEASE (COPD) TREATMENT GUIDELINES Document Description Document Type Service Application Version Guidelines All healthcare professionals(hcps) caring for patients with asthma
reslizumab (Cinqair )
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Zhao Y Y et al. Ann Intern Med 2012;156:
 Zhao Y Y et al. Ann Intern Med 2012;156:560-569 Introduction Fibrates are commonly prescribed to treat dyslipidemia An increase in serum creatinine level after use has been observed in randomized, placebocontrolled
Zhao Y Y et al. Ann Intern Med 2012;156:560-569 Introduction Fibrates are commonly prescribed to treat dyslipidemia An increase in serum creatinine level after use has been observed in randomized, placebocontrolled
Rayzel Shulman MD, PhD CAHSPR May 10, 2016
 + Low Socioeconomic Status is Associated with Adverse Events in Children and Teens on Insulin Pumps Under a Universal Access Program: A population-based Cohort Study Rayzel Shulman MD, PhD CAHSPR May 10,
+ Low Socioeconomic Status is Associated with Adverse Events in Children and Teens on Insulin Pumps Under a Universal Access Program: A population-based Cohort Study Rayzel Shulman MD, PhD CAHSPR May 10,
THE COPD PRESCRIBING TOOL
 THE COPD PRESCRIBING TOOL Revised edition, 2017 www.bpac.org.nz/copd CLASSIFICATION The COPD prescribing tool This tool provides pharmacological treatment options for patients with COPD based on their
THE COPD PRESCRIBING TOOL Revised edition, 2017 www.bpac.org.nz/copd CLASSIFICATION The COPD prescribing tool This tool provides pharmacological treatment options for patients with COPD based on their
Supplementary Online Content
 Supplementary Online Content Pham B, Stern A, Chen W, et al. Preventing pressure ulcers in long-term care: a costeffectiveness analysis. Arch Intern Med. Published online September 26, 2011. doi:10.1001/archinternmed.2011.473
Supplementary Online Content Pham B, Stern A, Chen W, et al. Preventing pressure ulcers in long-term care: a costeffectiveness analysis. Arch Intern Med. Published online September 26, 2011. doi:10.1001/archinternmed.2011.473
Patterns and Cost of Health Care Transitions to Adult Care among Youth with Chronic Conditions in Ontario: A Population Based Cohort Study
 Patterns and Cost of Health Care Transitions to Adult Care among Youth with Chronic Conditions in Ontario: A Population Based Cohort Study Eyal Cohen, MD, MSc; Sima Gandhi, MSc; Charlotte Moore, MD; Alene
Patterns and Cost of Health Care Transitions to Adult Care among Youth with Chronic Conditions in Ontario: A Population Based Cohort Study Eyal Cohen, MD, MSc; Sima Gandhi, MSc; Charlotte Moore, MD; Alene
Why Asthma Still Kills The National Review of Asthma Deaths (NRAD)
 APPROVED FINAL VERSION NHS Protect Why Asthma Still Kills The National Review of Asthma Deaths (NRAD) Summary of Recommendations for GP Practices and Community Pharmacies Author: Anne Henry Contact: anne.henry@nhs.net
APPROVED FINAL VERSION NHS Protect Why Asthma Still Kills The National Review of Asthma Deaths (NRAD) Summary of Recommendations for GP Practices and Community Pharmacies Author: Anne Henry Contact: anne.henry@nhs.net
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Fasenra) Reference Number: CP.PHAR.## Effective Date: 01.16.18 Last Review Date: 05.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
Clinical Policy: (Fasenra) Reference Number: CP.PHAR.## Effective Date: 01.16.18 Last Review Date: 05.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this policy
«Impact of a pharmaceutical intervention to improve adherence of inhaled medication in asthma and COPD patients»
 «Impact of a pharmaceutical intervention to improve adherence of inhaled medication in asthma and COPD patients» Baseline data from a randomized controlled trial 5 th Swiss Health Services Research Symposium,
«Impact of a pharmaceutical intervention to improve adherence of inhaled medication in asthma and COPD patients» Baseline data from a randomized controlled trial 5 th Swiss Health Services Research Symposium,
CHRONIC OBSTRUCTIVE PULMONARY DISEASE
 A F O C A l p O I N T l E A r N I N g p r O g r A m m E CHRONIC OBSTRUCTIVE PULMONARY DISEASE S E C O N D E D I T I O N BOOK 1 September 2013 FP120/1 CENTRE FOR PHARMACY POSTGRADUATE EDUCATION About CPPE
A F O C A l p O I N T l E A r N I N g p r O g r A m m E CHRONIC OBSTRUCTIVE PULMONARY DISEASE S E C O N D E D I T I O N BOOK 1 September 2013 FP120/1 CENTRE FOR PHARMACY POSTGRADUATE EDUCATION About CPPE
Optimising asthma management in high risk patients
 C L I N I C A L AU D I T Optimising asthma management in high risk patients Valid to June 2022 bpac nz better medicine Audit focus This audit helps primary care health professionals identify patients with
C L I N I C A L AU D I T Optimising asthma management in high risk patients Valid to June 2022 bpac nz better medicine Audit focus This audit helps primary care health professionals identify patients with
Asthma Update A/Prof. John Abisheganaden. Senior Consultant, Dept Of Respiratory & Crit Care Medicine Tan Tock Seng Hospital
 Asthma Update - 2013 A/Prof. John Abisheganaden Senior Consultant, Dept Of Respiratory & Crit Care Medicine Tan Tock Seng Hospital Asthma A complex syndrome Multifaceted disease Heterogeneous Genetic and
Asthma Update - 2013 A/Prof. John Abisheganaden Senior Consultant, Dept Of Respiratory & Crit Care Medicine Tan Tock Seng Hospital Asthma A complex syndrome Multifaceted disease Heterogeneous Genetic and
AEROSOL. Indication: /formoterol to. older with pressurized. two inhalations
 CEDAC FINAL RECOMMENDATION MOMETASONE FUROATE/ FORMOTEROL FUMARATE DIHYDRATE INHALATION AEROSOL (Zenhale Merck Canada Inc.) Indication: Asthma Maintenanc ce (Adults, Children 12 Years or Older) Recommendation:
CEDAC FINAL RECOMMENDATION MOMETASONE FUROATE/ FORMOTEROL FUMARATE DIHYDRATE INHALATION AEROSOL (Zenhale Merck Canada Inc.) Indication: Asthma Maintenanc ce (Adults, Children 12 Years or Older) Recommendation:
aclidinium 322 micrograms inhalation powder (Eklira Genuair ) SMC No. (810/12) Almirall S.A.
 aclidinium 322 micrograms inhalation powder (Eklira Genuair ) SMC No. (810/12) Almirall S.A. 05 October 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and
aclidinium 322 micrograms inhalation powder (Eklira Genuair ) SMC No. (810/12) Almirall S.A. 05 October 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and
Adherence to asthma controller medication regimens
 Respiratory Medicine (2005) 99, 1263 1267 Adherence to asthma controller medication regimens D.A. Stempel a,, S.W. Stoloff b, J.R. Carranza Rosenzweig c, R.H. Stanford c, K.L. Ryskina d, A.P. Legorreta
Respiratory Medicine (2005) 99, 1263 1267 Adherence to asthma controller medication regimens D.A. Stempel a,, S.W. Stoloff b, J.R. Carranza Rosenzweig c, R.H. Stanford c, K.L. Ryskina d, A.P. Legorreta
Common Drug Review Pharmacoeconomic Review Report
 Common Drug Review Pharmacoeconomic Review Report January 2018 Drug umeclidinium bromide (Incruse Ellipta) Indication Listing request Dosage form(s) Manufacturer Indicated for long-term, once daily maintenance
Common Drug Review Pharmacoeconomic Review Report January 2018 Drug umeclidinium bromide (Incruse Ellipta) Indication Listing request Dosage form(s) Manufacturer Indicated for long-term, once daily maintenance
Include patients: with a confirmed diagnosis of asthma who have been free of asthma symptoms for 3 months or more.
 Corby Clinical Commissioning Group Kettering General Hospital NHS Trust Nene Clinical Commissioning Group rthampton General Hospital NHS Trust rthamptonshire Healthcare Foundation Trust Stepping down asthma
Corby Clinical Commissioning Group Kettering General Hospital NHS Trust Nene Clinical Commissioning Group rthampton General Hospital NHS Trust rthamptonshire Healthcare Foundation Trust Stepping down asthma
Improving Outcomes in the Management & Treatment of Asthma. April 21, Spring Managed Care Forum
 Improving Outcomes in the Management & Treatment of Asthma April 21, 2016 2016 Spring Managed Care Forum David M. Mannino, M.D. Professor Department of Preventive Medicine and Environmental Health University
Improving Outcomes in the Management & Treatment of Asthma April 21, 2016 2016 Spring Managed Care Forum David M. Mannino, M.D. Professor Department of Preventive Medicine and Environmental Health University
AIRDUO RESPICLICK (fluticasone-salmeterol) aerosol DULERA (mometasone furoate and formoterol fumarate dihydrate) aerosol
 DULERA (mometasone furoate and formoterol fumarate dihydrate) aerosol Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific
DULERA (mometasone furoate and formoterol fumarate dihydrate) aerosol Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific
BC Methadone Maintenance System
 BC Methadone Maintenance System Performance Measures 211/212 Office of the Provincial Health Officer With contributions by: Pharmaceutical Services Division & Population and Public Health Division British
BC Methadone Maintenance System Performance Measures 211/212 Office of the Provincial Health Officer With contributions by: Pharmaceutical Services Division & Population and Public Health Division British
