Diagnosis, management and new treatments for Spinal Muscular Atrophy Special Focus: SMA Type 1
|
|
|
- Mary Lane
- 5 years ago
- Views:
Transcription
1 Diagnosis, management and new treatments for Spinal Muscular Atrophy Special Focus: SMA Type 1 17 th April 2018 Adnan Manzur Consultant Paediatric Neurologist Dubowitz Neuromuscular Centre, GOSH & ICH, London
2 Conflict on interests NHS clinician On UK advisory board for Biogen (Spinraza manufacturer) Slides with green background in this presentation were provided as a free teaching resource by Biogen
3 Telephone consultation: 8 month old Male infant Feeding difficulties: 2-3 months old 4 months: unable to hold head 5 months: ref to SLT: unsafe swallow 7 months: Severe pneumonia Intubation - Ventilated EMG myopathic unusual breathing diaphragmatic Local hospital wanted to do muscle biopsy
4 8 month old Male infant SMN gene study homozygous SMN1 deletion of exon 7 SMN2 copy number - 2 Extubated to NIV BiPAP dependent (enabled extubation) 8 months: Night and day NIV dependent, with coming off the NIV for 3 breaks or half an hour each. Nasogastric feeding (history of failure to thrive & dysphagia) Gastro-oesophageal reflux Constipation
5 Focus Clinical presentation, severity classification Diagnosis Differential diagnosis Standards of care Changing natural history Nusinersen Implications
6 SMA I. 5q linked AR motor neuron disorder Onset by 6 months, never sit Life limiting to 2 yrs Spectrum of severity: Type IA: (SMA0) presentation at birth, joint contractures, respiratory compromise Type IB: onset of symptoms < age 3 months Type IC: onset after 3 months of age. SMN1 (telomeric) mutations Reduced full length SMN protein SMN2, nearly identical, has exonic splicing enhancer mutation that limits inclusion of exon 7 SMN2 copy number is inversely related to clinical severity
7 SMA I. 5q linked Autosomal Recessive motor neuron disorder SMN1 gene deletions, SMN protein reduction Onset by 6 months, never sit Life limiting
8
9
10 3 newborn siblings generalized weakness respiratory insufficiency, facial diplegia, extraocular muscle involvement. axonal neuropathy. homozygous deletions SMN gene. remained dependent on artificial ventilation died at the ages of 3 weeks, 4 weeks, and 2 days
11
12
13 Outcome measures and treatment of spinal muscular atrophy, 134th ENMC International Workshop. Bertini E, et al. Neuromuscul Disord 2005;15: Type 1a: the severe neonatal variant with joint contractures and a paucity of movement present at birth has a poor prognosis, often needing ventilatory support as a neonate. Type 1b: the typical SMA-1 patient having poor head control and difficulty handling oral secretions upon or shortly after presentation and has an intermediate prognosis. Type 1c: the minority of type 1 patients who achieve head control or who can sit with support and have the best prognosis.
14 Observational study of spinal muscular atrophy type I and implications for clinical trials. Finkel et al. Neurology Aug 26;83(9):810-7 Type IA - (also described in literature as SMA 0) with presentation at birth with joint contractures and respiratory compromise Type IB - onset of symptoms before age 3 months Type IC - onset after 3 months of age.
15 Spinal muscular atrophy. SMN1telomeric & SMN2Centromeric Spinal muscular atrophy. Lunn &Wong Lancet 2008; 371:
16 SMN2 copy number. (centromeric, SMNc) Survival (less than/greater than 24 months)
17 SMN protein Wertz & Sahin 2015
18 Clinical ECG baseline tremor (CK normal to mild elevation) (EMG) (muscle histology) SMN1 gene testing Diagnosis
19 EMG Abnormal spontaneous activity, e.g. Fibrillations, positive sharp waves and fasciculations by EMG. Increased mean duration and amplitude of motor unit action potentials by EMG. Comments: MNCVs may be markedly reduced in SMA type I. There is a rare congenital-onset SMA with death within the first weeks of life in which MNCVs are very long and SNAPs are absent.
20 EMG in SMA diagnosis clinical perspective SMA 1. go straight to SMN gene test (ICU ventilated infant useful to initiate tests, but caution about relying on EMG as the sole diagnostic / prognostic test) Confirmation of neurogenic EMG in affected individuals with one SMN1 copy deleted, before proceeding with point mutation study in the other allele
21 Histopathology of muscle Groups of atrophic Fibers of both types. Hypertrophic fibers of type I. Type grouping (chronic cases). Comment: In early-onset cases of SMA type I these characteristic features may not be present. Instead there are small fibers of both types. In SMA III there may be a concomitant myopathic pattern.
22 Differential Diagnosis of SMA I In the typical case, clinical diagnosis is easy Pompes (severe alpha glucosidase deficiency) Olivopontocerebellar hypolpasia (type 1) with SMA Mitochondrial SMARD 1 key clinical differences Other neurogenic conditions
23 SMA1 Management QOL Survival Customizing care MDT approach Standards of care (Anticipatory care ) Research trials Novel drugs Evolving care Beyond standards of care
24 SMA 1 Survival - changing Natural History Mannaa et al 2009 Thomas et al 1994 Improvement in survival based on General care / nutritional / respiratory support Country / cultural context Implications for treatment trials
25 SMA. Standards of care. SMA1. Anticipatory care. NIV Cough machine
26 SMA1. Genetic therapeutic approaches Wertz and Sahin 2015
27 Anderton & Mastaglia 2015
28 Anderton & Mastaglia 2015
29
30 Multicentre randomized, double-blind, shamcontrolled, phase 3 Inclusion criteria: Homozygous deletion or mutation SMN1 2 copies of the SMN2 gene onset of symptoms that were consistent with spinal muscular atrophy before t 6 months age 7 months or younger at screening, did not have low peripheral oxygen saturation
31
32
33 Panel A - probability of event-free survival (the proportion of infants who were alive without the use of permanent assisted ventilation) Panel B -probability of overall survival (proportion of infants who were alive)
34
35 Can nusinersen prevent onset of symptoms in genetically diagnosed pre-symptomatic SMA1 infants? in context of positive family history or future newborn screening
36
37 Translational timelines. Nusinersen Spinraza TM 1 st Human use 2011, Orphan drug status Apr ISIS (ISIS) Endear Study pre-specified primary endpoint met Aug 16 Biogen announces Extended Access Programme (EAP) Aug 16 FDA approval Dec 2016 NHS commissioners leave EAP uptake to individual trusts UK EAP Sporadic uptake starts Belfast - Apr 2017 CHMP (Committee for Medicinal Products ) +ve recommendation Apr 17 EMA (European Medicine Agency) Marketing authorization June 2017 NICE evaluation pending ( STA? HST?) (NICE Technology Appraisal (TA) or Highly Specialised Technologies Evaluation (HST) NHS England approves EAP supportive funding for SMA1 (2 copies of SMN2 ) Aug 17
38
39 15 patients with SMA1 single dose of intravenous adeno-associated virus serotype 9 carrying SMN complementary DNA encoding the missing SMN protein. 3 patients received a low dose ( vg per kilogram of body weight) 12 received a high dose ( vg per kilogram). The primary outcome was safety. The secondary outcome was the time until death or the need for permanent ventilator assistance.
40 Data cutoff on August 7, 2017: all 15 patients were alive and event-free at 20 months of age, as compared with a rate of survival of 8% in a historical cohort. In the high-dose cohort: CHOP INTEND scale increase of: 9.8 points at 1 month 15.4 points at 3 months 11 sat unassisted, 9 rolled over, 11 fed orally and could speak, and 2 walked independently. Elevated serum ALT levels occurred in 4 patients and were attenuated by prednisolone.
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: nusinersen_spinraza 03/2017 10/2018 10/2019 10/2018 Description of Procedure or Service Spinal muscular atrophy
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: nusinersen_spinraza 03/2017 10/2018 10/2019 10/2018 Description of Procedure or Service Spinal muscular atrophy
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: nusinersen_spinraza 03/2017 10/2017 10/2018 10/2017 Description of Procedure or Service Spinal muscular atrophy
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: nusinersen_spinraza 03/2017 10/2017 10/2018 10/2017 Description of Procedure or Service Spinal muscular atrophy
Spinraza (nusinersen)
 Spinraza (nusinersen) Policy Number: 5.02.540 Last Review: 04/2018 Origination: 03/2017 Next Review: 04/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage for Spinraza
Spinraza (nusinersen) Policy Number: 5.02.540 Last Review: 04/2018 Origination: 03/2017 Next Review: 04/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage for Spinraza
Cigna Drug and Biologic Coverage Policy
 Cigna Drug and Biologic Coverage Policy Subject Nusinersen Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information... 5 References... 5 Effective Date... 10/15/2017 Next
Cigna Drug and Biologic Coverage Policy Subject Nusinersen Table of Contents Coverage Policy... 1 General Background... 2 Coding/Billing Information... 5 References... 5 Effective Date... 10/15/2017 Next
Spinal Muscular Atrophy Newborn Screening
 10.2018 Spinal Muscular Atrophy Newborn Screening Melissa Gibbons, MS, CGC Erica Wright, MS, CGC Clinical Genetics and Metabolism Department of Pediatrics Children s Hospital Colorado/ University of Colorado
10.2018 Spinal Muscular Atrophy Newborn Screening Melissa Gibbons, MS, CGC Erica Wright, MS, CGC Clinical Genetics and Metabolism Department of Pediatrics Children s Hospital Colorado/ University of Colorado
Spinal Muscular Atrophy in 2017
 Spinal Muscular Atrophy in 2017 Leigh Maria Ramos-Platt, MD Children s Hospital of Los Angeles University of Southern California Keck School of Medicine Disclosures MDA Care Center Grant In this presentation,
Spinal Muscular Atrophy in 2017 Leigh Maria Ramos-Platt, MD Children s Hospital of Los Angeles University of Southern California Keck School of Medicine Disclosures MDA Care Center Grant In this presentation,
Nusinersen Use in Spinal Muscular Atrophy
 Nusinersen Use in Spinal Muscular Atrophy Report by: Guideline Development, Dissemination, and Implementation Subcommittee of the American Academy of Neurology Slide 1 Evidence in Focus Endorsement and
Nusinersen Use in Spinal Muscular Atrophy Report by: Guideline Development, Dissemination, and Implementation Subcommittee of the American Academy of Neurology Slide 1 Evidence in Focus Endorsement and
Clinical Policy Bulletin: Nusinersen (Spinraza)
 Clinical Policy Bulletin: Nusinersen (Spinraza) Number: 0915 Policy *Pleasesee amendment forpennsylvaniamedicaidattheendofthiscpb. Note: REQUIRES PRECERTIFICATION.Footnotes for Precertification of nusinersen
Clinical Policy Bulletin: Nusinersen (Spinraza) Number: 0915 Policy *Pleasesee amendment forpennsylvaniamedicaidattheendofthiscpb. Note: REQUIRES PRECERTIFICATION.Footnotes for Precertification of nusinersen
Spinraza (Nusinersen) Drug Prior Authorization Protocol (Medical Benefit & Part B Benefit)
 Line of Business: All Lines of Business Effective Date: August 16, 2017 Spinraza (Nusinersen) Drug Prior Authorization Protocol (Medical Benefit & Part B Benefit) This policy has been developed through
Line of Business: All Lines of Business Effective Date: August 16, 2017 Spinraza (Nusinersen) Drug Prior Authorization Protocol (Medical Benefit & Part B Benefit) This policy has been developed through
SPINRAZA (NUSINERSEN)
 SPINRAZA (NUSINERSEN) UnitedHealthcare Commercial Medical Benefit Drug Policy Policy Number: 2018D0059D Effective Date: April 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 BENEFIT CONSIDERATIONS...
SPINRAZA (NUSINERSEN) UnitedHealthcare Commercial Medical Benefit Drug Policy Policy Number: 2018D0059D Effective Date: April 1, 2018 Table of Contents Page INSTRUCTIONS FOR USE... 1 BENEFIT CONSIDERATIONS...
Letter of Medical Necessity The Use of SPINRAZA (nusinersen) for Spinal Muscular Atrophy
 TEMPLATE Letter of Medical Necessity The Use of SPINRAZA (nusinersen) for Spinal Muscular Atrophy Date: [Insert Name of Medical Director] RE: Patient Name [ ] [Insurance Company] Policy Number [ ] [Address]
TEMPLATE Letter of Medical Necessity The Use of SPINRAZA (nusinersen) for Spinal Muscular Atrophy Date: [Insert Name of Medical Director] RE: Patient Name [ ] [Insurance Company] Policy Number [ ] [Address]
Clinical Policy: Nusinersen (Spinraza) Reference Number: CP.PHAR.327
 Clinical Policy: (Spinraza) Reference Number: CP.PHAR.327 Effective Date: 03/17 Last Review Date: 03/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Spinraza) Reference Number: CP.PHAR.327 Effective Date: 03/17 Last Review Date: 03/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Spinraza) Reference Number: CP.PHAR.327 Effective Date: 11.28.17 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder
Clinical Policy: (Spinraza) Reference Number: CP.PHAR.327 Effective Date: 11.28.17 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important Reminder
The 2017 Update of the Standard of Care Recommendations for Spinal Muscular Atrophy
 Richard Finkel, MD Nemours Children s Hospital Orlando, FL, USA Thomas Crawford, MD Johns Hopkins Hospital Baltimore, MD, USA The 2017 Update of the Standard of Care Recommendations for Spinal Muscular
Richard Finkel, MD Nemours Children s Hospital Orlando, FL, USA Thomas Crawford, MD Johns Hopkins Hospital Baltimore, MD, USA The 2017 Update of the Standard of Care Recommendations for Spinal Muscular
SMA Treatments and Clinical Trials. Kenneth Hobby, President Mary Schroth, MD, Chief Medical Officer Jill Jarecki, PhD, Chief Scientific Officer
 SMA Treatments and Clinical Trials Kenneth Hobby, President Mary Schroth, MD, Chief Medical Officer Jill Jarecki, PhD, Chief Scientific Officer Agenda Introduction Kenneth Hobby, President Spinraza Update
SMA Treatments and Clinical Trials Kenneth Hobby, President Mary Schroth, MD, Chief Medical Officer Jill Jarecki, PhD, Chief Scientific Officer Agenda Introduction Kenneth Hobby, President Spinraza Update
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: Nusinersen (Spinraza) Reference Number: CP.PHAR.327 Effective Date: 10.01.18 Last Review Date: 07.13.18 Line of Business: Oregon Health Plan Coding Implications Revision Log See Important
Clinical Policy: Nusinersen (Spinraza) Reference Number: CP.PHAR.327 Effective Date: 10.01.18 Last Review Date: 07.13.18 Line of Business: Oregon Health Plan Coding Implications Revision Log See Important
Association of motor milestones and SMN2 copy and outcome in spinal muscular. atrophy types 0 4
 jnnp-2016-314292 1 - SUPPLEMENTARY FILE - Methods and additional data on clinical characteristics and motor development Association of motor milestones and SMN2 copy and outcome in spinal muscular atrophy
jnnp-2016-314292 1 - SUPPLEMENTARY FILE - Methods and additional data on clinical characteristics and motor development Association of motor milestones and SMN2 copy and outcome in spinal muscular atrophy
Nusinersen in Pre-symptomatic Infants With Spinal Muscular Atrophy (SMA): Interim Efficacy and Safety Results From the Phase 2 NURTURE Study
 21 st International Congress of the World Muscle Society -8 October, 2016 Granada, Spain Nusinersen in Pre-symptomatic Infants With Spinal Muscular Atrophy (SMA): Interim Efficacy and Safety Results From
21 st International Congress of the World Muscle Society -8 October, 2016 Granada, Spain Nusinersen in Pre-symptomatic Infants With Spinal Muscular Atrophy (SMA): Interim Efficacy and Safety Results From
Interim Efficacy and Safety Results from the Phase 2 NURTURE Study Evaluating Nusinersen in Presymptomatic Infants With Spinal Muscular Atrophy
 American Academy of Neurology 2017 69th Annual Meeting April 22 28, 2017 Boston, MA Interim Efficacy and Safety Results from the Phase 2 NURTURE Study Evaluating Nusinersen in Presymptomatic Infants With
American Academy of Neurology 2017 69th Annual Meeting April 22 28, 2017 Boston, MA Interim Efficacy and Safety Results from the Phase 2 NURTURE Study Evaluating Nusinersen in Presymptomatic Infants With
Thomas O. Crawford, MD July 1, Annual Spinal Muscular Atrophy Conference June 29 July 2, 2017 Orlando, FL
 2017 Annual Spinal Muscular Atrophy Conference June 29 July 2, 2017 Orlando, FL Efficacy and Safety of Nusinersen in Genetically Diagnosed Infants With Presymptomatic Spinal Muscular Atrophy (SMA): Results
2017 Annual Spinal Muscular Atrophy Conference June 29 July 2, 2017 Orlando, FL Efficacy and Safety of Nusinersen in Genetically Diagnosed Infants With Presymptomatic Spinal Muscular Atrophy (SMA): Results
When a Threshold Crossing approach may and may not be appropriate: A Case Study in SMA
 When a Threshold Crossing approach may and may not be appropriate: A Case Study in SMA EFSPI Regulatory Statistics Workshop, 24-25 th September 2018 Carol Reid and Uli Burger picture placeholder Outline
When a Threshold Crossing approach may and may not be appropriate: A Case Study in SMA EFSPI Regulatory Statistics Workshop, 24-25 th September 2018 Carol Reid and Uli Burger picture placeholder Outline
Prior Authorization Update: Nusinersen
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Clinical Review Report
 CADTH COMMON DRUG REVIEW Clinical Review Report Nusinersen (Spinraza) (Biogen Canada Inc.) Indication: Treatment of patients with 5q SMA Service Line: CADTH Common Drug Review Version: Version 1.0 Publication
CADTH COMMON DRUG REVIEW Clinical Review Report Nusinersen (Spinraza) (Biogen Canada Inc.) Indication: Treatment of patients with 5q SMA Service Line: CADTH Common Drug Review Version: Version 1.0 Publication
Update on Pulmonary Management in Spinal Muscular Atrophy type 1
 Update on Pulmonary Management in Spinal Muscular Atrophy type 1 David Zielinski, MD FRCPC, FCCP Associate Professor McGill University Montreal Children s Hospital None Disclosures Objectives Review genetic
Update on Pulmonary Management in Spinal Muscular Atrophy type 1 David Zielinski, MD FRCPC, FCCP Associate Professor McGill University Montreal Children s Hospital None Disclosures Objectives Review genetic
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Amyotrophic Lateral Sclerosis 10 (ALS10) and Amyotrophic Lateral Sclerosis 6 (ALS6)
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Amyotrophic Lateral Sclerosis 10 (ALS10) and Amyotrophic Lateral Sclerosis 6 (ALS6)
Roche leads the clinical development of risdiplam as part of a collaboration with the SMA Foundation and PTC Therapeutics.
 Media Release PRIME designation granted by European Medicines Agency for Roche s risdiplam for treatment of spinal muscular atrophy (SMA) Risdiplam has the potential to be the first oral medicine for the
Media Release PRIME designation granted by European Medicines Agency for Roche s risdiplam for treatment of spinal muscular atrophy (SMA) Risdiplam has the potential to be the first oral medicine for the
COMPANY OVERVIEW. June 2016
 COMPANY OVERVIEW June 2016 Disclaimers Forward-Looking Statements This presentation contains forward-looking statements, including: statements about: the timing, progress and results of preclinical studies
COMPANY OVERVIEW June 2016 Disclaimers Forward-Looking Statements This presentation contains forward-looking statements, including: statements about: the timing, progress and results of preclinical studies
Panel II: SMA Drugs in Development
 Panel II: SMA Drugs in Development Jinsy Andrews MD, Director of Clinical Research and Development, Cytokinetics Thomas Blaettler MD, Global Clinical Development Team Leader, F. Hoffmann-La Roche Jerry
Panel II: SMA Drugs in Development Jinsy Andrews MD, Director of Clinical Research and Development, Cytokinetics Thomas Blaettler MD, Global Clinical Development Team Leader, F. Hoffmann-La Roche Jerry
New Drug Evaluation: Nusinersen Injection, Intrathecal
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Ambulant & non ambulant (Types 2 & 3) Spinal Muscular Atrophy
 Ambulant & non ambulant (Types 2 & 3) Spinal Muscular Atrophy Mário Miguel Rosa, MD, PhD SAWP, CNSWP (EMA) FMUL, INFARMED Lisbon - Portugal SMA EMA workshop An agency of the European Union Content Relevant
Ambulant & non ambulant (Types 2 & 3) Spinal Muscular Atrophy Mário Miguel Rosa, MD, PhD SAWP, CNSWP (EMA) FMUL, INFARMED Lisbon - Portugal SMA EMA workshop An agency of the European Union Content Relevant
Muscular Dystrophies. Pinki Munot Consultant Paediatric Neurologist Great Ormond Street Hospital Practical Neurology Study days April 2018
 Muscular Dystrophies Pinki Munot Consultant Paediatric Neurologist Great Ormond Street Hospital Practical Neurology Study days April 2018 Definition and classification Clinical guide to recognize muscular
Muscular Dystrophies Pinki Munot Consultant Paediatric Neurologist Great Ormond Street Hospital Practical Neurology Study days April 2018 Definition and classification Clinical guide to recognize muscular
Screening for spinal muscular atrophy External review against programme appraisal criteria for the UK National Screening Committee (UK NSC)
 Screening for spinal muscular atrophy External review against programme appraisal criteria for the UK National Screening Committee (UK NSC) Version: Third Draft Consultation Author: Costello Medical Date:
Screening for spinal muscular atrophy External review against programme appraisal criteria for the UK National Screening Committee (UK NSC) Version: Third Draft Consultation Author: Costello Medical Date:
Related Policies None
 Medical Policy MP 5.01.28 BCBSA Ref. Policy: 5.01.28 Last Review: 04/30/2018 Effective Date: 07/30/2018 Section: Prescription Drug Related Policies None DISCLAIMER Our medical policies are designed for
Medical Policy MP 5.01.28 BCBSA Ref. Policy: 5.01.28 Last Review: 04/30/2018 Effective Date: 07/30/2018 Section: Prescription Drug Related Policies None DISCLAIMER Our medical policies are designed for
Final Results of the Phase 3 ENDEAR Study Assessing the Efficacy and Safety of Nusinersen in Infants With Spinal Muscular Atrophy (SMA)
 69th Annual Meeting of the American Academy of Neurology April 22-28, 2017 Boston, MA, USA Final Results of the Phase 3 ENDEAR Study Assessing the Efficacy and Safety of Nusinersen in Infants With Spinal
69th Annual Meeting of the American Academy of Neurology April 22-28, 2017 Boston, MA, USA Final Results of the Phase 3 ENDEAR Study Assessing the Efficacy and Safety of Nusinersen in Infants With Spinal
INDUSTRY PERSPECTIVE ON DRUG DEVELOPMENT FOR TYPE 1 SMA
 INDUSTRY PERSPECTIVE ON DRUG DEVELOPMENT FOR TYPE 1 SMA Kathie M. Bishop, PhD Disclosures: Currently, CSO of Tioga Pharmaceuticals, no activities in SMA; 2009-2015, full-time employee of Ionis Pharmaceuticals,
INDUSTRY PERSPECTIVE ON DRUG DEVELOPMENT FOR TYPE 1 SMA Kathie M. Bishop, PhD Disclosures: Currently, CSO of Tioga Pharmaceuticals, no activities in SMA; 2009-2015, full-time employee of Ionis Pharmaceuticals,
Update in Neuromuscular Disorders Tuesday 5- Friday 8 May Clinical Neuroscience Lecture Theatre at 33 Queen Square, London WC1N 3BG PROGRAMME
 Update in Neuromuscular Disorders Tuesday 5- Friday 8 May 2015 Clinical Neuroscience Lecture Theatre at 33 Queen Square, London WC1N 3BG Day 1 Tuesday 5 May PROGRAMME 10:00 10:30 Registration* and coffee
Update in Neuromuscular Disorders Tuesday 5- Friday 8 May 2015 Clinical Neuroscience Lecture Theatre at 33 Queen Square, London WC1N 3BG Day 1 Tuesday 5 May PROGRAMME 10:00 10:30 Registration* and coffee
SALSA MLPA KIT P060-B2 SMA
 SALSA MLPA KIT P6-B2 SMA Lot 111, 511: As compared to the previous version B1 (lot 11), the 88 and 96 nt DNA Denaturation control fragments have been replaced (QDX2). Please note that, in contrast to the
SALSA MLPA KIT P6-B2 SMA Lot 111, 511: As compared to the previous version B1 (lot 11), the 88 and 96 nt DNA Denaturation control fragments have been replaced (QDX2). Please note that, in contrast to the
SMA IS A SEVERE NEUROLOGICAL DISORDER [1]
![SMA IS A SEVERE NEUROLOGICAL DISORDER [1] SMA IS A SEVERE NEUROLOGICAL DISORDER [1]](/thumbs/72/67086153.jpg) SMA OVERVIEW SMA IS A SEVERE NEUROLOGICAL DISORDER [1] Autosomal recessive genetic inheritance 1 in 50 people (approximately 6 million Americans) are carriers [2] 1 in 6,000 to 1 in 10,000 children born
SMA OVERVIEW SMA IS A SEVERE NEUROLOGICAL DISORDER [1] Autosomal recessive genetic inheritance 1 in 50 people (approximately 6 million Americans) are carriers [2] 1 in 6,000 to 1 in 10,000 children born
Family Health, Nottingham Children s Hospital Date of submission February 2018
 Nusinersen Title of Guideline Guideline for the prescribing and administration of Nusinersen to patients with Spinal Muscular Atrophy Type 1 Contact Name and Job Title (author) Dr Gabriel Chow- Consultant
Nusinersen Title of Guideline Guideline for the prescribing and administration of Nusinersen to patients with Spinal Muscular Atrophy Type 1 Contact Name and Job Title (author) Dr Gabriel Chow- Consultant
NEW HORIZONS IN TREATING SMA. Dr. Huda Mussaffi Schneider Children s Medical Center of Israel
 NEW HORIZONS IN TREATING SMA Dr. Huda Mussaffi Schneider Children s Medical Center of Israel WHAT IS SMA? Rare and debilitating autosomal recessive neuromuscular disease characterized by degeneration of
NEW HORIZONS IN TREATING SMA Dr. Huda Mussaffi Schneider Children s Medical Center of Israel WHAT IS SMA? Rare and debilitating autosomal recessive neuromuscular disease characterized by degeneration of
AVXS-101 and Nusinersen for Spinal Muscular Atrophy: Effectiveness and Value
 AVXS-101 and Nusinersen for Spinal Muscular Atrophy: Effectiveness and Value Background Draft Background and Scope August 22, 2018 Spinal muscular atrophy (SMA) is a rare, genetic neuromuscular disease
AVXS-101 and Nusinersen for Spinal Muscular Atrophy: Effectiveness and Value Background Draft Background and Scope August 22, 2018 Spinal muscular atrophy (SMA) is a rare, genetic neuromuscular disease
SMA Treatment Access and Clinical Trials Webinar. February 15, 2018
 SMA Treatment Access and Clinical Trials Webinar February 15, 2018 Treatment Access Update Current Dosing Metrics December 2017: ~1,900 patients been dosed in US Including those in extension trials and
SMA Treatment Access and Clinical Trials Webinar February 15, 2018 Treatment Access Update Current Dosing Metrics December 2017: ~1,900 patients been dosed in US Including those in extension trials and
Nusinersen Demonstrates Efficacy in Infants With and Without Permanent Ventilation: Final Results From the ENDEAR Study
 2017 Annual Spinal Muscular Atrophy Conference June 29 July 2, 2017 Orlando, FL Nusinersen Demonstrates Efficacy in Infants With and Without Permanent Ventilation: Final Results From the ENDEAR Study Richard
2017 Annual Spinal Muscular Atrophy Conference June 29 July 2, 2017 Orlando, FL Nusinersen Demonstrates Efficacy in Infants With and Without Permanent Ventilation: Final Results From the ENDEAR Study Richard
Update in Neuromuscular Disorders Tuesday 5- Friday 8 May Clinical Neuroscience Lecture Theatre at 33 Queen Square, London WC1N 3BG PROGRAMME
 Update in Neuromuscular Disorders Tuesday 5- Friday 8 May 2015 Clinical Neuroscience Lecture Theatre at 33 Queen Square, London WC1N 3BG Day 1 Tuesday 5 May PROGRAMME 10:00 10:30 Registration* and coffee
Update in Neuromuscular Disorders Tuesday 5- Friday 8 May 2015 Clinical Neuroscience Lecture Theatre at 33 Queen Square, London WC1N 3BG Day 1 Tuesday 5 May PROGRAMME 10:00 10:30 Registration* and coffee
Evolving therapeutic landscape for inherited neurologic disorders. Kathryn Swoboda, MD HMS Child Neurology Course September
 Evolving therapeutic landscape for inherited neurologic disorders Kathryn Swoboda, MD HMS Child Neurology Course September 5 2017 Disclosures I ve received funding as a consultant for Biogen for clinical
Evolving therapeutic landscape for inherited neurologic disorders Kathryn Swoboda, MD HMS Child Neurology Course September 5 2017 Disclosures I ve received funding as a consultant for Biogen for clinical
Pompe. Lysosomal Disorders. Introduction
 Introduction disease is an inherited, genetic disorder which results in the lack of an enzyme 'acid alpha-glucosidase. disease is also known as acid maltase deficiency or glycogen storage disease type
Introduction disease is an inherited, genetic disorder which results in the lack of an enzyme 'acid alpha-glucosidase. disease is also known as acid maltase deficiency or glycogen storage disease type
MRC-Holland MLPA. Description version 19;
 SALSA MLPA probemix P6-B2 SMA Lot B2-712, B2-312, B2-111, B2-511: As compared to the previous version B1 (lot B1-11), the 88 and 96 nt DNA Denaturation control fragments have been replaced (QDX2). SPINAL
SALSA MLPA probemix P6-B2 SMA Lot B2-712, B2-312, B2-111, B2-511: As compared to the previous version B1 (lot B1-11), the 88 and 96 nt DNA Denaturation control fragments have been replaced (QDX2). SPINAL
Nusinersen versus Sham Control in Infantile-Onset Spinal Muscular Atrophy
 The new england journal of medicine Original Article versus Sham in Infantile-Onset Spinal Muscular Atrophy R.S. Finkel, E. Mercuri, B.T. Darras, A.M. Connolly, N.L. Kuntz, J. Kirschner, C.A. Chiriboga,
The new england journal of medicine Original Article versus Sham in Infantile-Onset Spinal Muscular Atrophy R.S. Finkel, E. Mercuri, B.T. Darras, A.M. Connolly, N.L. Kuntz, J. Kirschner, C.A. Chiriboga,
Final Phase 3 Study Data Show SPINRAZA (nusinersen) Significantly Improved Motor Function in Children with Later-Onset Spinal Muscular Atrophy
 MEDIA CONTACT: Biogen Ligia Del Bianco Ph: +1 781 464 3260 public.affairs@biogen.com INVESTOR CONTACT: Biogen Ben Strain Ph: +1 781 464 2442 IR@biogen.com Final Phase 3 Study Data Show SPINRAZA (nusinersen)
MEDIA CONTACT: Biogen Ligia Del Bianco Ph: +1 781 464 3260 public.affairs@biogen.com INVESTOR CONTACT: Biogen Ben Strain Ph: +1 781 464 2442 IR@biogen.com Final Phase 3 Study Data Show SPINRAZA (nusinersen)
Objectives. What is SMA? Pathophysiologic and genetic mechanisms How to identify a case of SMA
 Objectives What is SMA? Pathophysiologic and genetic mechanisms How to identify a case of SMA What can be done? Review of advances in standards of care and treatment Detailed review of treatment available
Objectives What is SMA? Pathophysiologic and genetic mechanisms How to identify a case of SMA What can be done? Review of advances in standards of care and treatment Detailed review of treatment available
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE. Proposed Highly Specialised Technology Evaluation
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Proposed Highly Specialised Technology Evaluation Drisapersen for treating Duchenne muscular Draft scope (pre-referral) Draft remit/evaluation objective
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Proposed Highly Specialised Technology Evaluation Drisapersen for treating Duchenne muscular Draft scope (pre-referral) Draft remit/evaluation objective
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name HEMOCHROMATOSIS, TYPE 4; HFE4 OMIM number for disease #606069 Disease alternative
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name HEMOCHROMATOSIS, TYPE 4; HFE4 OMIM number for disease #606069 Disease alternative
Patient L.L. KRISTEN ARREDONDO, MD CHILD NEUROLOGY PGY5, UT SOUTHWESTERN
 Patient L.L. KRISTEN ARREDONDO, MD CHILD NEUROLOGY PGY5, UT SOUTHWESTERN Birth History LL was born to a healthy first time mother with an uncomplicated pregnancy Delivered at 38 weeks via C-section due
Patient L.L. KRISTEN ARREDONDO, MD CHILD NEUROLOGY PGY5, UT SOUTHWESTERN Birth History LL was born to a healthy first time mother with an uncomplicated pregnancy Delivered at 38 weeks via C-section due
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Leber congenital amaurosis OMIM number for disease 204000 Disease alternative
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Leber congenital amaurosis OMIM number for disease 204000 Disease alternative
TEST INFORMATION Test: CarrierMap GEN (Genotyping) Panel: CarrierMap Expanded Diseases Tested: 311 Genes Tested: 299 Mutations Tested: 2647
 Ordering Practice Jane Smith John Smith Practice Code: 675 Miller MD 374 Broadway New York, NY 10000 Physician: Dr. Frank Miller Report Generated: 2016-02-03 DOB: 1973-02-19 Gender: Female Ethnicity: European
Ordering Practice Jane Smith John Smith Practice Code: 675 Miller MD 374 Broadway New York, NY 10000 Physician: Dr. Frank Miller Report Generated: 2016-02-03 DOB: 1973-02-19 Gender: Female Ethnicity: European
Faculty. W4: Identifying Patients with Rare Disorders Using Administrative Data. O Dan Malone, RPh, PhD University of Arizona
 W4: Identifying Patients with Rare Disorders Using Administrative Data Faculty O Dan Malone, RPh, PhD University of Arizona O Danny Yeh, PhD Biogen O Ed Armstrong Strategic Therapeutics, LLC O Eric Bell,
W4: Identifying Patients with Rare Disorders Using Administrative Data Faculty O Dan Malone, RPh, PhD University of Arizona O Danny Yeh, PhD Biogen O Ed Armstrong Strategic Therapeutics, LLC O Eric Bell,
Pharmacoeconomic Review Report
 CADTH COMMON DRUG REVIEW Pharmacoeconomic Review Report NUSINERSEN (SPINRAZA) (Biogen Canada Inc.) Indication: Treatment of patients with 5q SMA Service Line: CADTH Common Drug Review Version: Final Publication
CADTH COMMON DRUG REVIEW Pharmacoeconomic Review Report NUSINERSEN (SPINRAZA) (Biogen Canada Inc.) Indication: Treatment of patients with 5q SMA Service Line: CADTH Common Drug Review Version: Final Publication
Neonatal Hypotonia. Encephalopathy acute No encephalopathy. Neurology Chapter of IAP
 The floppy infant assumes a frog legged position. On ventral suspension, the baby can not maintain limb posture against gravity and assumes the position of a rag doll. Encephalopathy acute No encephalopathy
The floppy infant assumes a frog legged position. On ventral suspension, the baby can not maintain limb posture against gravity and assumes the position of a rag doll. Encephalopathy acute No encephalopathy
Amyotrophic Lateral Sclerosis: Developing Drugs for Treatment Guidance for Industry
 Amyotrophic Lateral Sclerosis: Developing Drugs for Treatment Guidance for Industry DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding
Amyotrophic Lateral Sclerosis: Developing Drugs for Treatment Guidance for Industry DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding
Title: Developmental milestones in type I spinal muscular atrophy
 Accepted Manuscript Title: Developmental milestones in type I spinal muscular atrophy Author: Roberto De Sanctis, Giorgia Coratti, Amy Pasternak, Jacqueline Montes, Marika Pane, Elena S. Mazzone, Sally
Accepted Manuscript Title: Developmental milestones in type I spinal muscular atrophy Author: Roberto De Sanctis, Giorgia Coratti, Amy Pasternak, Jacqueline Montes, Marika Pane, Elena S. Mazzone, Sally
Assessing the Needs of the SMA Population: Survey Results of Health Care Providers and Families
 559018SGOXXX10.1177/2158244014559018SAGE OpenHalanski et al. research-article2014 Article Assessing the Needs of the SMA Population: Survey Results of Health Care Providers and Families SAGE Open October-December
559018SGOXXX10.1177/2158244014559018SAGE OpenHalanski et al. research-article2014 Article Assessing the Needs of the SMA Population: Survey Results of Health Care Providers and Families SAGE Open October-December
Spinal Muscular Atrophy: Case Study. Spinal muscular atrophy (SMA) is a fairly common genetic disorder, affecting
 Spinal Muscular Atrophy: Case Study Spinal muscular atrophy (SMA) is a fairly common genetic disorder, affecting approximately one in 6,000 babies. It is estimated that one in every 40 Americans carries
Spinal Muscular Atrophy: Case Study Spinal muscular atrophy (SMA) is a fairly common genetic disorder, affecting approximately one in 6,000 babies. It is estimated that one in every 40 Americans carries
SMA Therapeutics: A Comparative Overview of Drugs Approved and in Development. Sponsored By:
 SMA Therapeutics: A Comparative Overview of Drugs Approved and in Development Sponsored By: August 8, 2017 Targets for Therapeutic Intervention in SMA Decrease in SMN protein due to SMN1 gene deletion
SMA Therapeutics: A Comparative Overview of Drugs Approved and in Development Sponsored By: August 8, 2017 Targets for Therapeutic Intervention in SMA Decrease in SMN protein due to SMN1 gene deletion
1/28/2019. OSF HealthCare INI Care Center Team. Neuromuscular Disease: Muscular Dystrophy. OSF HealthCare INI Care Center Team: Who are we?
 Neuromuscular Disease: Muscular Dystrophy Muscular Dystrophy Association (MDA) and OSF HealthCare Illinois Neurological Institute (INI) Care Center Team The Neuromuscular clinic is a designated MDA Care
Neuromuscular Disease: Muscular Dystrophy Muscular Dystrophy Association (MDA) and OSF HealthCare Illinois Neurological Institute (INI) Care Center Team The Neuromuscular clinic is a designated MDA Care
Genetic diagnosis of limb girdle muscular dystrophy type 2A, A Case Report
 Genetic diagnosis of limb girdle muscular dystrophy type 2A, A Case Report Roshanak Jazayeri, MD, PhD Assistant Professor of Medical Genetics Faculty of Medicine, Alborz University of Medical Sciences
Genetic diagnosis of limb girdle muscular dystrophy type 2A, A Case Report Roshanak Jazayeri, MD, PhD Assistant Professor of Medical Genetics Faculty of Medicine, Alborz University of Medical Sciences
ABSTRACT ORIGINAL RESEARCH
 https://doi.org/10.1007/s12325-019-00923-8 ORIGINAL RESEARCH Survival, Motor Function, and Motor Milestones: Comparison of AVXS-101 Relative to Nusinersen for the Treatment of Infants with Spinal Muscular
https://doi.org/10.1007/s12325-019-00923-8 ORIGINAL RESEARCH Survival, Motor Function, and Motor Milestones: Comparison of AVXS-101 Relative to Nusinersen for the Treatment of Infants with Spinal Muscular
Myotubular (centronuclear) myopathy
 Myotubular (centronuclear) myopathy Myotubular, or centronuclear, myopathy falls under the umbrella of congenital myopathies. It is characterised by a specific pattern in the muscle tissue when viewed
Myotubular (centronuclear) myopathy Myotubular, or centronuclear, myopathy falls under the umbrella of congenital myopathies. It is characterised by a specific pattern in the muscle tissue when viewed
Pediatric Patients. Neuromuscular Disease. Teera Kijmassuwan, MD Phetcharat Netmuy, B.N.S., MA Oranee Sanmaneechai, MD : Preceptor
 Patient Management Pediatric Patients with Neuromuscular Disease Teera Kijmassuwan, MD Phetcharat Netmuy, B.N.S., MA Oranee Sanmaneechai, MD : Preceptor Case Thai boy 1 year old Present with Respiratory
Patient Management Pediatric Patients with Neuromuscular Disease Teera Kijmassuwan, MD Phetcharat Netmuy, B.N.S., MA Oranee Sanmaneechai, MD : Preceptor Case Thai boy 1 year old Present with Respiratory
The Floppy Baby. Clare Betteridge
 The Floppy Baby Clare Betteridge The floppy baby Identification Evaluation Investigation Diagnosis Examples What is a floppy baby? Elbows and knees loosely extended. Head control is usually poor or absent.
The Floppy Baby Clare Betteridge The floppy baby Identification Evaluation Investigation Diagnosis Examples What is a floppy baby? Elbows and knees loosely extended. Head control is usually poor or absent.
Clinical Policy Title: Spinraza
 Clinical Policy Title: Spinraza Clinical Policy Number: 02.02.03 Effective Date: July 1, 2018 Initial Review Date: May 19, 2017 Most Recent Review Date: June 5, 2018 Next Review Date: June 2019 Related
Clinical Policy Title: Spinraza Clinical Policy Number: 02.02.03 Effective Date: July 1, 2018 Initial Review Date: May 19, 2017 Most Recent Review Date: June 5, 2018 Next Review Date: June 2019 Related
CADTH Canadian Drug Expert Committee Recommendation
 CADTH COMMON DRUG REVIEW CADTH Canadian Drug Expert Committee Recommendation (FINAL) Nusinersen (Spinraza Biogen Canada Inc.) Indication: Treatment of 5q Spinal Muscular Atrophy RECOMMENDATION: The CADTH
CADTH COMMON DRUG REVIEW CADTH Canadian Drug Expert Committee Recommendation (FINAL) Nusinersen (Spinraza Biogen Canada Inc.) Indication: Treatment of 5q Spinal Muscular Atrophy RECOMMENDATION: The CADTH
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names you wish listed) (A)-Testing
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names you wish listed) (A)-Testing
Three siblings with progressive respiratory distress as infants
 Case reports Three siblings with progressive respiratory distress as infants 1459 63 The assessment of rare congenital neuromuscular disorders can be difficult. Although muscle biopsy neurophysiological
Case reports Three siblings with progressive respiratory distress as infants 1459 63 The assessment of rare congenital neuromuscular disorders can be difficult. Although muscle biopsy neurophysiological
Genotype phenotype studies in infantile spinal muscular atrophy (SMA) type I in Germany: implications for clinical trials and genetic counselling
 Clin Genet 2009: 76: 168 178 Printed in Singapore. All rights reserved Short Report 2009 John Wiley & Sons A/S CLINICAL GENETICS doi: 10.1111/j.1399-0004.2009.01200.x Genotype phenotype studies in infantile
Clin Genet 2009: 76: 168 178 Printed in Singapore. All rights reserved Short Report 2009 John Wiley & Sons A/S CLINICAL GENETICS doi: 10.1111/j.1399-0004.2009.01200.x Genotype phenotype studies in infantile
What s New in Newborn Screening?
 What s New in Newborn Screening? Funded by: Illinois Department of Public Health Information on Newborn Screening Newborn screening in Illinois is administered by the Illinois Department of Public Health.
What s New in Newborn Screening? Funded by: Illinois Department of Public Health Information on Newborn Screening Newborn screening in Illinois is administered by the Illinois Department of Public Health.
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Choroideremia OMIM number for disease 303100 Disease alternative names please
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Choroideremia OMIM number for disease 303100 Disease alternative names please
Spinal muscular atrophy 5Q Treatment with nusinersen
 GUIDELINES IN FOCUS Spinal muscular atrophy 5Q Treatment with nusinersen Author: Brazilian Medical Association Participants: Antonio Silvinato; Wanderley M Bernardo Final version: May 5, 2018 1. Brazilian
GUIDELINES IN FOCUS Spinal muscular atrophy 5Q Treatment with nusinersen Author: Brazilian Medical Association Participants: Antonio Silvinato; Wanderley M Bernardo Final version: May 5, 2018 1. Brazilian
Nemaline (rod) myopathies
 Nemaline (rod) myopathies Nemaline, or rod, myopathies are a group of conditions which fall under the umbrella of congenital myopathies. They are characterised by rod-like structures in the muscle cells,
Nemaline (rod) myopathies Nemaline, or rod, myopathies are a group of conditions which fall under the umbrella of congenital myopathies. They are characterised by rod-like structures in the muscle cells,
Guidance on format of the risk management plan (RMP) in the EU part II: Module SIV - Populations not studied in clinical trials
 25 July 2013 EMA/465927/2013 Rev.1 Patient Health Protection Guidance on format of the risk management plan (RMP) in the EU part II: Module SIV - Populations not studied in clinical trials Active substance
25 July 2013 EMA/465927/2013 Rev.1 Patient Health Protection Guidance on format of the risk management plan (RMP) in the EU part II: Module SIV - Populations not studied in clinical trials Active substance
Neuromuscular in the Pediatric Clinic: Recognition and Referral
 Neuromuscular in the Pediatric Clinic: Recognition and Referral Matthew Harmelink, MD Assistant Professor, Pediatric Neurology Medical College of Wisconsin Objectives: 1. Understand common presentations
Neuromuscular in the Pediatric Clinic: Recognition and Referral Matthew Harmelink, MD Assistant Professor, Pediatric Neurology Medical College of Wisconsin Objectives: 1. Understand common presentations
Orofacial function of persons having. Report from questionnaires. Spinal muscular atrophy
 27-2-8 Orofacial function of persons having Spinal muscular atrophy Report from questionnaires The survey comprises questionnaires. Synonyms: SMA I (Werdnig-Hoffmann disease, SMA II, SMA III (Kugelberg-Welander
27-2-8 Orofacial function of persons having Spinal muscular atrophy Report from questionnaires The survey comprises questionnaires. Synonyms: SMA I (Werdnig-Hoffmann disease, SMA II, SMA III (Kugelberg-Welander
Evaluation of the Hypotonic Infant and Child
 Evaluation of the Hypotonic Infant and Child Basil T. Darras, M.D. Neuromuscular Program Boston Children s Hospital Harvard Medical School Boston, MA, USA Classification and General Clinical Evaluation
Evaluation of the Hypotonic Infant and Child Basil T. Darras, M.D. Neuromuscular Program Boston Children s Hospital Harvard Medical School Boston, MA, USA Classification and General Clinical Evaluation
Learn the steps to identify pediatric muscle weakness and signs of neuromuscular disease.
 Learn the steps to identify pediatric muscle weakness and signs of neuromuscular disease. Listen Observe Evaluate Test Refer Guide for primary care providers includes: Surveillance Aid: Assessing Weakness
Learn the steps to identify pediatric muscle weakness and signs of neuromuscular disease. Listen Observe Evaluate Test Refer Guide for primary care providers includes: Surveillance Aid: Assessing Weakness
ASPIRO Phase 1/2 Gene Therapy Trial in X-Linked Myotubular Myopathy (XLMTM): Preliminary Safety and Efficacy Findings
 ASPIRO Phase 1/2 Gene Therapy Trial in X-Linked Myotubular Myopathy (XLMTM): Preliminary Safety and Efficacy Findings Safe Harbor Except for statements of historical fact, any information contained in
ASPIRO Phase 1/2 Gene Therapy Trial in X-Linked Myotubular Myopathy (XLMTM): Preliminary Safety and Efficacy Findings Safe Harbor Except for statements of historical fact, any information contained in
SPINRAZA (nusinersen) CLINICAL OVERVIEW
 SPINRAZA (nusinersen) CLINICAL OVERVIEW IN BOTH CONTROLLED AND OPEN-LABEL STUDIES, SOME PATIENTS WHO HAD OR WERE LIKELY TO DEVELOP SPINAL MUSCULAR ATROPHY (SMA) TYPE 1, 2, OR 3 TREATED WITH SPINRAZA 1
SPINRAZA (nusinersen) CLINICAL OVERVIEW IN BOTH CONTROLLED AND OPEN-LABEL STUDIES, SOME PATIENTS WHO HAD OR WERE LIKELY TO DEVELOP SPINAL MUSCULAR ATROPHY (SMA) TYPE 1, 2, OR 3 TREATED WITH SPINRAZA 1
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name OMIM number for disease 231300 Disease alternative names Please provide any alternative
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name OMIM number for disease 231300 Disease alternative names Please provide any alternative
CARE CONSIDERATIONS FOR DUCHENNE MUSCULAR DYSTROPHY
 IMPORTANT NEW UPDATE A Summary of the Report of the DMD Care Considerations Working Group Intended for US healthcare professionals only. CARE CONSIDERATIONS FOR DUCHENNE MUSCULAR DYSTROPHY Full article
IMPORTANT NEW UPDATE A Summary of the Report of the DMD Care Considerations Working Group Intended for US healthcare professionals only. CARE CONSIDERATIONS FOR DUCHENNE MUSCULAR DYSTROPHY Full article
Evaluation of Nusinersen (SPINRAZA TM ) in Medicinrådet
 Evaluation of Nusinersen (SPINRAZA TM ) in Medicinrådet Contents 1. Executive summary... 3 2. Natural history of the disease... 4 3. Medicine Council Questions... 6 4. Clinical added value of nusinersen
Evaluation of Nusinersen (SPINRAZA TM ) in Medicinrådet Contents 1. Executive summary... 3 2. Natural history of the disease... 4 3. Medicine Council Questions... 6 4. Clinical added value of nusinersen
Should Universal Carrier Screening be Universal?
 Should Universal Carrier Screening be Universal? Disclosures Research funding from Natera Mary E Norton MD University of California, San Francisco Antepartum and Intrapartum Management June 15, 2017 Burden
Should Universal Carrier Screening be Universal? Disclosures Research funding from Natera Mary E Norton MD University of California, San Francisco Antepartum and Intrapartum Management June 15, 2017 Burden
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Genetic Causes of Hypothyroidism 1. Loss of function mutations in TSHR cause thyroid
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Genetic Causes of Hypothyroidism 1. Loss of function mutations in TSHR cause thyroid
Lactic Acidosis and Ascending Neuromuscular Syndrome. Mina Hosseinipour, M.D, MPH. University of North Carolina Project, Lilongwe Malawi
 Lactic Acidosis and Ascending Neuromuscular Syndrome Mina Hosseinipour, M.D, MPH. University of North Carolina Project, Lilongwe Malawi Introduction ART is rapidly scaling up in resource poor countries
Lactic Acidosis and Ascending Neuromuscular Syndrome Mina Hosseinipour, M.D, MPH. University of North Carolina Project, Lilongwe Malawi Introduction ART is rapidly scaling up in resource poor countries
DOSAGE FORMS AND STRENGTHS Injection: 12 mg/5 ml (2.4 mg/ml) in a single-dose vial (3)
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use SPINRAZA safely and effectively. See full prescribing information for SPINRAZA. SPINRAZA (nusinersen)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use SPINRAZA safely and effectively. See full prescribing information for SPINRAZA. SPINRAZA (nusinersen)
DSS-1. No financial disclosures
 DSS-1 No financial disclosures Clinical History 9 year old boy with past medical history significant for cerebral palsy, in-turning right foot, left clubfoot that was surgically corrected at 3 years of
DSS-1 No financial disclosures Clinical History 9 year old boy with past medical history significant for cerebral palsy, in-turning right foot, left clubfoot that was surgically corrected at 3 years of
LIMP BABIES - WHAT MAY THAT MEAN?
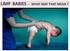 LIMP BABIES - WHAT MAY THAT MEAN? ARE WE LOOKING WHERE WE SHOULD? FLOPPY = LIMP = FLACCID = WEAK Poor head control when pulled C-posture sitting, or in ventral susp. Frog-leg lying Dropping arms Casey
LIMP BABIES - WHAT MAY THAT MEAN? ARE WE LOOKING WHERE WE SHOULD? FLOPPY = LIMP = FLACCID = WEAK Poor head control when pulled C-posture sitting, or in ventral susp. Frog-leg lying Dropping arms Casey
The first and only treatment for spinal muscular atrophy (SMA)
 Discover SPINRAZA The first and only treatment for spinal muscular atrophy (SMA) INDICATION SPINRAZA is a prescription medicine used to treat spinal muscular atrophy (SMA) in pediatric and adult patients.
Discover SPINRAZA The first and only treatment for spinal muscular atrophy (SMA) INDICATION SPINRAZA is a prescription medicine used to treat spinal muscular atrophy (SMA) in pediatric and adult patients.
9/30/2017. SMA: Spinal Muscular Atrophy Learning Outcomes: SMA. Prevalence. Review of the diagnosis. Here are the odds
 SMA: Spinal Muscular Atrophy Learning Outcomes: 1 in 6,000 to 1 in 10,000 but 100% to CRT Kay Koch, OTR/L, ATP The van Halem Group, A Division of VGM kkotrchoa@yahoo.com The participant will be able to
SMA: Spinal Muscular Atrophy Learning Outcomes: 1 in 6,000 to 1 in 10,000 but 100% to CRT Kay Koch, OTR/L, ATP The van Halem Group, A Division of VGM kkotrchoa@yahoo.com The participant will be able to
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Epileptic encephalopathy, early infantile 4. OMIM number for disease 612164 Disease
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Epileptic encephalopathy, early infantile 4. OMIM number for disease 612164 Disease
An Overview of Bronchopulmonary Dysplasia and Chronic Lung Disease in Infancy
 An Overview of Bronchopulmonary Dysplasia and Chronic Lung Disease in Infancy Housekeeping: I have no financial disclosures Learning objectives: Develop an understanding of bronchopulmonary dysplasia (BPD)
An Overview of Bronchopulmonary Dysplasia and Chronic Lung Disease in Infancy Housekeeping: I have no financial disclosures Learning objectives: Develop an understanding of bronchopulmonary dysplasia (BPD)
An Introduction to mitochondrial disease.
 9 th September 2017 An Introduction to mitochondrial disease. Dr Andy Schaefer Consultant Neurologist and Clinical Lead NHS Highly Specialised Rare Mitochondrial Disease Service and Wellcome Trust Centre
9 th September 2017 An Introduction to mitochondrial disease. Dr Andy Schaefer Consultant Neurologist and Clinical Lead NHS Highly Specialised Rare Mitochondrial Disease Service and Wellcome Trust Centre
Motor Neurone Disease NICE to manage Management of ineffective cough. Alex Long Specialist NIV/Respiratory physiotherapist June 2016
 Motor Neurone Disease NICE to manage Management of ineffective cough Alex Long Specialist NIV/Respiratory physiotherapist June 2016 Content NICE guideline recommendations Respiratory involvement in MND
Motor Neurone Disease NICE to manage Management of ineffective cough Alex Long Specialist NIV/Respiratory physiotherapist June 2016 Content NICE guideline recommendations Respiratory involvement in MND
