Chapter 17 Acids and Bases (same as Exam 2 Worksheet Answers)
|
|
|
- Jason Phillips
- 6 years ago
- Views:
Transcription
1 xam 3 Wrksheet Answers xam 3 Wrksheet Answers Chemistry 4 Chapter 17 Acids and Bases (same as xam Wrksheet Answers) 1. Fr the weak acids, what is the pk a and rank the acids accrding t strength? ; d e HNO > HCOOH > CH COOH > H CO > HCN > HCO Fr the weak bases, what is the pk b value and rank the bases accrding t strength? d e CH NH > CH N > NH > C H N > C H NH Fr the buffer slutins f 5 ml f.5 M HA and 35 ml f.0 M f the cnjugate salt, what is the ph? d What is the neutralizatin reactin fr each f the preceding buffers when cmbined with HCl? NH aq H aq NH aq 3 4 CN ( aq) H ( aq) HCN ( aq) F ( aq) H ( aq) HF ( aq) d. NO ( aq) H ( aq) HNO ( aq) 5. What is the ph f the slutin when ml f a M slutin f HCl is added? d
2 xam 3 Wrksheet Answers 6. What is the neutralizatin reactin fr each f the preceding buffers when cmbined with NaOH? NH aq OH aq NH aq H O l 4 3 HCN ( aq) OH ( aq) CN ( aq) HO( l) HF ( aq) OH ( aq) F ( aq) HO( l) d. HNO ( aq) OH ( aq) NO ( aq) H O( l) 7. What is the ph f the slutin when 5 ml f a 0.5 M slutin f NaOH is added? d Fr the titratin f 35.0 ml f a M nitric acid with M NaOH, what is the ph at each f these vlumes f NaOH added? d e. Many mst likely select phenlphthalein f ml g Fr the titratin f 35.0 ml f a M nitrus acid with M NaOH, what is the ph at each f these vlumes f NaOH added? d e. Many mst likely select phenlphthalein f ml g Fr the titratin f 35.0 ml f a M methyl amine with M hydrbrmic acid, what is the ph at each f these vlumes f HBr added? d..3 e. Many mst likely select methyl f ml g. 6.3
3 xam 3 Wrksheet Answers 11. What is the reactin fr a saturated slutin f all sulfides in Table 17. (p. 607)? 3 BiS3 s Bi aq 3S ( aq) CdS( s) Cd ( aq) S ( aq) CS( s) C ( aq) S ( aq) d. CuS( s) Cu ( aq) S ( aq) e. FeS( s) Fe ( aq) S ( aq) f. PbS( s) Pb ( aq) S ( aq) g. MnS( s) Mn ( aq) S ( aq) h. HgS( s) Hg ( aq) S ( aq) i. NiS( s) Ni ( aq) S ( aq) j. AgS s Ag aq S ( aq) k. SnS( s) Sn ( aq) S ( aq) l. ZnS( s) Zn ( aq) S ( aq) 1. What is the crrespnding values fr the reactins in number 11 and equilibrium expressin fr these values? Bi S = Cd S = C S = 4.0 d. 37 Cu S = 6.0 e. 19 Fe S = 6.0 f. 8 Pb S = 3.4 g. 14 Mn S = 3.0 h. 54 Hg S = 4.0 i. 4 Ni S = 1.4 j. 51 Ag S = 6.0 k. 6 Sn S = 1.0 l. 3 Zn S = What is the mlar slubility f each salt frm number 11? M.8 14 M M d M e. 7.7 M f M g M 3
4 xam 3 Wrksheet Answers h..0 7 M i M j M k M l M 14. What mass f the salt wuld be disslved in 1.5 L f water at 5 C? g g g d g e. 1.0 g f g g.. 5 g h g i. 1.6 g j g k g l. 8.0 g 15. What is the mlar cncentratin f each metal in in M sdium sulfide (frm number 11)? M M M d M e M f M g M h M i M j M k M l M 16. When equal vlumes f M sdium sulfide and M metal nitrates (frm number 11) are cmbined, what will happen? Because Q fr M is 5.6 9, Q > K s all MS salts will precipitate (where M = metal) Because Q fr Ag is 4. 13, Q > K s Ag S will precipitate Because Q fr Bi 3 is.4 1, Q > K s Bi S 3 will precipitate 4
5 xam 3 Wrksheet Answers 17. Fr a slutin 1.0 L f M tin nitrate, silver nitrate and bismuth nitrate, when adding cncentrated sdium sulfide (such that adding the sdium hydrxide will nt change the ttal vlume) answer: Which salt will precipitate first? Ag S Which salt will precipitate secnd? SnS Which salt will precipitate last? Bi S 3 d. What is the cncentratin f the first metal in that precipitated when the secnd salt begins t precipitate?.7 13 M e. What is the cncentratin f the secnd metal in that precipitated when the third salt begins t precipitate? M f. Can these salts be selectively precipitated? Ag S yes; Bi S 3 and SnS n Chapter 6 and Chapter 18 Thermdynamics 1. What is the standard change in enthalpy at 5 C fr the reactins? Assign whether the prcess is endthermic r exthermi kj; exthermic kj; endthermic 17.8 kj; exthermic d kj; exthermic. What is the standard change in enthalpy at 5 C fr the verall reactin given the thermchemical equatins? Assign whether the prcess is endthermic r exthermi 134 kj; exthermic 8 kj; exthermic 17.8 kj; exthermic d kj; exthermic e kj; exthermic 3. What is the standard enthalpy f frmatin at 5 C f the substances, given the thermchemical reactins? Assign whether the prcess is endthermic r exthermi 463 kj ml 1 ; exthermic 14.4 kj ml 1 ; endthermic kj ml 1 ; exthermic d. 188 kj ml 1 ; exthermic 4. What is the standard enthalpy f frmatin at 5 C f each substance given the change in enthalpy f reactin? Assign whether the prcess is endthermic r exthermi 77.6 kj ml 1 ; exthermic 57.5 kj ml 1 ; exthermic kj ml 1 ; exthermic d kj ml 1 ; exthermic 5
6 xam 3 Wrksheet Answers 5. What is the frmatin reactin fr each substance? 6C( s) 3H( g) 1 O( g) C6H5OH ( l ) Cs O g CO g Cs 1 O ( g) COg d. 8C( s) 9H( g) C8H18( l) e. N( g) 1 O( g) NO( g ) f. N ( g) H ( g) N H ( g) 4 6. Which substance will have a higher standard mlar entrpy? NH 3 (g) N H 4 (g) Fe(l) d. Br (l) e. Br (l) f. I (l) 7. Which prcesses are spntaneus? All are at 1 atm f pressure. Ice melting at 5 C Ice melting at 5 C Water freezing at 5 C d. Water freezing at 5 C e. Lithium hydrxide disslving in water (unsaturated slutin) f. Lithium hydrxide slidifying in water (unsaturated slutin) g. Sugar (C 1 H O 11 ) disslving in water (unsaturated slutin) h. Sugar (C 1 H O 11 ) slidifying in water (unsaturated slutin) i. A ht piece f irn in cntact with a cld piece f cpper and cpper getting htter and the irn getting clder. j. A ht piece f irn in cntact with a cld piece f cpper and irn getting htter and the cpper getting clder. 8. stimate the standard entrpy at 5 C f reactin in number 1. ΔS < 0 ΔS > 0 Cannt be estimated (Δn gas = 0) d. ΔS < 0 9. Determine the standard entrpy at 5 C f reactin in number J K J K J K 1 6
7 xam 3 Wrksheet Answers d J K 1 e kj; exthermic f kj; endthermic g kj; exthermic h kj; exthermic. stimate spntaneity f the reactins at 5 C in number 1. Spntaneus (ΔG < 0) Nt spntaneus (ΔG > 0) Spntaneus (ΔG < 0) d. Spntaneus (ΔG < 0) 11. Calculate the change in Gibbs free energy at 5 C fr the reactins in number kj kj 19.4 kj d kj 1. Will the reactins at 5 C in number 1 (a) be spntaneus (b) never be spntaneus (c) be spntaneus nly at high temperatures (d) be spntaneus nly at lw temperatures? If the reactin is spntaneus nly at certain temperatures [(c) r (d)], at what temperature des the spntaneity switch? Spntaneus at lw temperatures (39 K / 55 C) Spntaneus at high temperatures (18 K / 835 C) Spntaneus at all temperatures d. Spntaneus at lw temperature (000 K / 9730 C) 13. What are the equilibrium cnstants at 5 C fr the reactins in number 1? What des this mean in terms f cncentratin f reactants vs prducts? 8.9 [reactants] [prducts] [reactants] > [prducts] 4.8 [reactants] < [prducts] d. Very large [reactants] < [prducts] 14. Fr the reactin belw, is this reactin spntaneus at 5 C? The pressure f each reactant is 1.0 atm and the pressure the prduct is atm at 5 C, what is ΔG? Is the reactin spntaneus under these cnditins? NO( g) N( g) NO( g) The reactin is nt spntaneus under standard cnditins (ΔG = 33.8 kj) hwever under the nn standard cnditins, the reactin is spntaneus (ΔG = 9.83 kj). 7
8 xam 3 Wrksheet Answers 15. What is the biling pint f carbn disulfide assuming enthalpy and entrpy are cnstant with changing temperature? 33 K / 50 C 16. Is the uctin f cpper(ii) sulfide spntaneus? If nt, can cupling this reactin t the reactin f sulfur and xygen t sulfur dixide make the refinement f cpper becme spntaneus? Is this cupled reactin then spntaneus at all temperatures? CuS ( s) Cu( s) S ( s) Δ G = 49.0 kj nt spntaneus CuS ( s) O( g ) Cu( s) SO( g ) Δ G = 51.4 kj spntaneus ΔH = 47.6 kj ΔS =.3 J K 1 always spntaneus Chapter 19 lectrchemistry ( nly) 1. What is the xidatin state f each element? 0 0 d. 0 e. f. g. 0. What is the xidatin state f each element? Li: O: H: Fe: 3 O: S: 6 Al: 3 O: N: 5 d. C: 4 O: e. C: O: f. I: 7 Cl: g. Cd: O: P: 5 h. Mn: 4 O: 3. Balance the x reactins, identifying fr each which species is gaining r lsing electrns, and which is xidatin and which is uctin. Fe ( aq) e Fe( s) gaining electrns / uctin Ag ( s) Ag ( aq) e lsing electrns / xidatin Net inic: Fe ( aq) Ag ( s) Fe( s) Ag ( aq) Overall: Fe NO aq Ag s Fe s AgNO aq 3 3 8
9 xam 3 Wrksheet Answers d. Cl aq Cl g e lsing electrns / xidatin Zn aq e Zn s gaining electrns / uctin Net inic: Cl aq Zn aq Zn s Cl g Overall: NaCl aq Zn NO aq Zn s Cl g NaCl aq ( 3) ( 3) 3 Au aq 3e Au s gaining electrns / uctins Cu s Cu aq e lsing electrns / xidatin Net inic: Au aq 3Cu s 3Cu aq Au s Overall: Au NO aq 3Cu s 3Cu NO aq Au s F g e F aq gaining electrns / uctin H O aq O g 4e 4H aq lsing electrns / xidatin Net inic: F g H O aq 4HF aq O g Overall: F g H O aq 4HF aq O g 4. Balance the x reactins in acidic slutin, identifying each half reactin as xidatin r uctin. SO aq 4H aq e SO g H O l uctin 4 Fe s Fe aq e xidatin H SO aq Fe s SO g H O l FeSO aq 4 4 d. () 3 7 H O l O g 4H aq 4e uctin Cr O aq 14H aq 6e Cr aq 7H O l xidatin 3 7 Net inic: Cr O aq 16H aq 3O g 4Cr aq 8H O l Overall: H Cr O aq 1HNO aq 3O g 4Cr NO aq 8H O l PbO s SO aq 4H aq e PbSO s H O l uctin Pb s SO aq e PbSO s xidatin PbO s Pb s SO aq 4H aq PbSO s H O l SO g H O l SO aq 4H aq e xidatin I s e I aq uctin Net inic: SO g H O l I s I aq SO aq 4H aq Overall: SO g H O l I s HI aq H SO aq 4 9
10 xam 3 Wrksheet Answers e. f. g. h. 3 Br aq Br l e xidatin NO aq 4H aq 3e NO g H O l uctin 3 Net inic: 6Br aq NO aq 8H aq 3Br l NO g 4H O l Overall: 6HBr aq HNO aq 3Br l NO g 4H O l 3 MnO s 4H aq e Mn aq H O l uctin Cl aq Cl g e xidatin MnO s 4H aq Cl aq Mn aq Cl g H O l Overall : MnO s 4HCl aq MnCl aq Cl g H O l MnO s 4H aq e Mn aq H O l uctin Br aq Br l e xidatin MnO s 4H aq Br aq Mn aq Br l H O l Overall : MnO s 4HBr aq MnBr aq Br l H O l 3 3 Cu s Cu aq e xidatin NO aq 4H aq 3e NO g H O l uctin NO g 4H O l 3Cu aq 3Cu s NO aq 8H aq Overall: NO g 4H O l 3Cu NO aq 3Cu s 8HNO aq Balance the x reactins in basic slutin, identifying each half reactin as xidatin r uctin. H g OH aq H O l e xidatin e Cu aq Cu s uctin H g Cu aq OH aq H O l Cu s Ni s Ni aq e xidatin O g H O l 4e 4OH aq uctin O g H O l Ni s Ni OH s 4 () () MnO s 4OH aq MnO aq H O l 3e xidatin Br l e Br l uctin MnO s 8OH aq 6Br l MnO aq 4H O l 3Br l 4
11 xam 3 Wrksheet Answers 6. Fr the reactins yu balanced in number three answer: Fe aq e Fe s = 0.44 V cathde reactin; cathde = Fe s Ag ( s) Ag ( aq) e x = 0.80 V ande reactin; ande = Ag ( s) cell = 1.4 V Nt spntaneus (reverse reactin is spntaneus, s electrns wuld spntaneusly flw frm irn t silver) Ag(s) Ag (aq) (1 M) ǁ Fe (aq) (1 M) Fe(s) d. x = = = = Cl aq Cl g e 1.36 V ande reactin; ande Pt s Zn aq e Zn s 0.76 V cathde reactin; cathde Zn s cell =.1 V Nt spntaneus (reverse reactin is spntaneus, s electrns wuld spntaneusly flw frm zinc t platinum) Zn (aq) (1 M) Zn(s) ǁ Cl (aq) (1 M) Cl (g) Pt(s) 3 = = = = Au aq 3e Au s 1.50 V cathde reactin; cathde Au s Cu s Cu aq e x 0.34 V ande reactin; ande Cu s cell = 1.16 V electrns flw frm cpper t gld (ande t cathde) Spntaneus Au 3 (aq) (1 M) Au(s) ǁ Cu(s) Cu (aq) (1 M) = = = = F g e F aq.87 V cathde reactin; cathde Pt s H O aq O g 4e 4H aq 1.3 V ande reactin; ande Pt s x cell = 1.64 V Spntaneus electrns flw frm ande t cathde Pt(s) F (g) F (aq) (1 M) ǁ H O(l) O (g) Pt(s) 7. Fr the reactins yu balanced in number fur answer: SO4 ( aq) 4H ( aq) e SO( g) HO( l ) = 0.0 V cathde reactin; cathde = Pt s Fe s Fe aq e x = 0.44 V ande reactin; ande = Fe s cell = 0.4 V Nt spntaneus (reverse reactin is spntaneus, s electrns wuld spntaneusly flw frm platinum t irn) () H O l O g 4H aq 4e x 3 7 = 1.3 V ande reactin; ande = Pt s Cr O aq 14H aq 6e Cr aq 7H O l = 1.33 V cathde reactin; cathde = Pt s 11
12 xam 3 Wrksheet Answers cell = 0. V Spntaneus electrns flw frm ande t cathde d. e. f. PbO s SO aq 4H aq e PbSO s H O l 4 4 = = 1.70 V cathde reactin; cathde PbO s Pb s SO4 aq e PbSO4 s x = 0.31 V ande reactin; ande = Pb s cell =.01 V electrns flw frm lead t lead(iv) xide (ande t cathde) Spntaneus SO( g) HO( l) SO4 ( aq) 4H ( aq) e = 0.0 V ande reactin; ande = Pt s I s e I aq cell = 0.33 V Spntaneus Br aq Br l e x = 0.53 V cathde reactin; cathde = Pt s electrns flw frm ande t cathde x 3 = 1.07 V ande reactin; ande = Pt s NO aq 4H aq 3e NO g H O l = 0.96 V cathde reactin; cathde = Pt s cell = 0.11 V Nt spntaneus (reverse reactin is spntaneus, s electrns wuld spntaneusly flw frm platinum (Br uctin) t platinum (NO xidatin)) MnO s 4H aq e Mn aq H O l 1.3 V cathde reactin; cathde MnO s = = Cl aq Cl g e x = 1.36 V ande reactin; ande = Pt s cell = 0.13 V Nt spntaneus (reverse reactin is spntaneus, s electrns wuld spntaneusly flw frm MnO t platinum) 1
13 xam 3 Wrksheet Answers g. h. MnO s 4H aq e Mn aq H O l Br aq Br l e x = 1.3 V cathde reactin; cathde = MnO s = 1.07 V ande reactin; ande = Pt s cell = 0.16 V electrns flw frm platinum t MnO (ande t cathde) Spntaneus Cu s Cu aq e x NO aq 4H aq 3e NO g H O l 3 = 0.34 V ande reactin; ande = Cu s = 0.96 V cathde reactin; cathde = Pt s cell = 0.6 V electrns flw frm cpper t platinum (ande t cathde) Spntaneus 8. Fr the reactins yu balanced in number five answer: H g OH aq H O l e x e Cu aq Cu s = 0.83 V ande reactin; ande = Pt s = 0.34 V cathde reactin; cathde = Cu s cell = 1.17 V electrns flw frm platinum t cpper (ande t cathde) Spntaneus Ni s Ni aq e x O g H O l 4e 4OH aq = 0.5 V ande reactin; ande = Ni s = 0.40 V cathde reactin; cathde = Pt s cell = 0.65 V electrns flw frm nickel t platinum (ande t cathde) Spntaneus MnO s 4OH aq MnO aq H O l 3e 4 () () Br l e Br l x = 0.59 V ande reactin; ande = MnO s = 1.07 V cathde reactin; cathde = Pt s cell = 0.48 V electrns flw frm MnO t platinum (ande t cathde) Spntaneus 13
( ) ( ) + ( ) ( ) ( ) + ( )
 xam 4 Wrksheet Answers xam 4 Wrksheet Answers Chemistry 104 Chapter 19 lectrchemistry (19.1 19.3 repeated frm xam 3 wrksheet) 1. What is the xidatin state f each element? 0 0 d. 0 e. f. g. 0. What is the
xam 4 Wrksheet Answers xam 4 Wrksheet Answers Chemistry 104 Chapter 19 lectrchemistry (19.1 19.3 repeated frm xam 3 wrksheet) 1. What is the xidatin state f each element? 0 0 d. 0 e. f. g. 0. What is the
3a. The acid and base react to form a salt solution of ammonium propionate. CaCO s + 2 HC H O aq CO g + H O l + Ca ( aq ) + 2 C H O ( aq)
 Chapter 5 Answers Practice Examples 1a. 0.50 M Cl 1b. (a) 7.9 10-5 M F - ; (b).1 kg CaF a. (a) Al ( aq ) OH ( aq ) Al ( OH) ( s) (b) No reaction occurs. (c) Pb ( aq ) I ( aq ) PbI ( s) b. (a) Al ( aq )
Chapter 5 Answers Practice Examples 1a. 0.50 M Cl 1b. (a) 7.9 10-5 M F - ; (b).1 kg CaF a. (a) Al ( aq ) OH ( aq ) Al ( OH) ( s) (b) No reaction occurs. (c) Pb ( aq ) I ( aq ) PbI ( s) b. (a) Al ( aq )
CHE Study Guide - Ch Carboxylic Acid + Alcohol
 CHE 10 - Study Guide - Ch. 4-5 Chapter 4: Carbxylic Acids 1. Functinal Grup (Carbxyl grup): r R-COOH r R-CO H. Naming a. Identify lngest chain with carbxyl grup b. Use Parent name, change e t ic acid c.
CHE 10 - Study Guide - Ch. 4-5 Chapter 4: Carbxylic Acids 1. Functinal Grup (Carbxyl grup): r R-COOH r R-CO H. Naming a. Identify lngest chain with carbxyl grup b. Use Parent name, change e t ic acid c.
Chemical Equations Part 1
 Chemical Equations Part 1 A Directed Learning Activity for Hartnell College Chemistry 1 Funded by the Title V STEM Grant #P031S090007 through Hartnell College For information contact lyee@hartnell.edu
Chemical Equations Part 1 A Directed Learning Activity for Hartnell College Chemistry 1 Funded by the Title V STEM Grant #P031S090007 through Hartnell College For information contact lyee@hartnell.edu
Module 1: Overview of soil fertility, plant nutrition, and nutrient management. To view the chapter for this topic click here.
 Mdule 1: Overview f sil fertility, plant nutritin, and nutrient management T view the chapter fr this tpic click here. Mdule 1: Overview Intrductin Understanding the principles f sil fertility is vital
Mdule 1: Overview f sil fertility, plant nutritin, and nutrient management T view the chapter fr this tpic click here. Mdule 1: Overview Intrductin Understanding the principles f sil fertility is vital
Module 3. Chapter 5 Microbial Metabolism. Catabolic and Anabolic Reactions Metabolism Two general types of metabolic reactions: o :
 Mdule 3 Chapter 5 Micrbial Metablism Catablic and Anablic Reactins Metablism Tw general types f metablic reactins: : : Recall frm Chapter 2: Energy can be when bnds Energy can be when bnds Catablism Purpse
Mdule 3 Chapter 5 Micrbial Metablism Catablic and Anablic Reactins Metablism Tw general types f metablic reactins: : : Recall frm Chapter 2: Energy can be when bnds Energy can be when bnds Catablism Purpse
REVISION: PHOTOSYNTHESIS & CELLULAR RESPIRATION 18 JUNE 2014
 REVISION: PHOTOSYNTHESIS & CELLULAR RESPIRATION 18 JUNE 2014 Lessn Descriptin In this lessn, we revise: the prcess f phtsynthesis the prcesses invlved in cellular respiratin Summary Phtsynthesis (Slutins
REVISION: PHOTOSYNTHESIS & CELLULAR RESPIRATION 18 JUNE 2014 Lessn Descriptin In this lessn, we revise: the prcess f phtsynthesis the prcesses invlved in cellular respiratin Summary Phtsynthesis (Slutins
Metabolism: Oxidative Phosphorylation
 Metablism: Oxidative Phsphrylatin Hand-ut fr the CBT versin April 2012 1. On average, hw much ATP is needed each day by ur bdy? 2. Which f the fllwing ccurs during cellular respiratin? Bisynthesis f glycgen
Metablism: Oxidative Phsphrylatin Hand-ut fr the CBT versin April 2012 1. On average, hw much ATP is needed each day by ur bdy? 2. Which f the fllwing ccurs during cellular respiratin? Bisynthesis f glycgen
PACT premier TM HT-96 MD1-36 PACT premier is a ph, Anion, Cation crystallization trial devised to test ph within a PEG/Ion screen.
 PACT premier TM HT-96 MD1-36 PACT premier is a ph, Anin, Catin crystallizatin trial devised t test ph within a PEG/In screen. MD1-36 cntains 96 x 1 ml reagents in a deep-well blck. Features f PACT premier
PACT premier TM HT-96 MD1-36 PACT premier is a ph, Anin, Catin crystallizatin trial devised t test ph within a PEG/In screen. MD1-36 cntains 96 x 1 ml reagents in a deep-well blck. Features f PACT premier
Matrix Reference Materials - SCP SCIENCE
 EnviroMAT SS-1 Catalogue No.: 140-025-001 EnviroMAT Contaminated Soil Lot No.: SC0063618 100 g TOTAL DIGESTION VALUES Elements Reference Value (mg/kg) Confidence Interval (mg/kg) Tolerance Interval (mg/kg)
EnviroMAT SS-1 Catalogue No.: 140-025-001 EnviroMAT Contaminated Soil Lot No.: SC0063618 100 g TOTAL DIGESTION VALUES Elements Reference Value (mg/kg) Confidence Interval (mg/kg) Tolerance Interval (mg/kg)
METHOD 8032 ACRYLAMIDE BY GAS CHROMATOGRAPHY
 METHOD 8032 ACRYLAMIDE BY GAS CHROMATOGRAPHY 1.0 SCOPE AND APPLICATION 1.1 Methd 8032 is used t determine trace amunts f acrylamide mnmer in aqueus matrices. This methd may be applicable t ther matrices.
METHOD 8032 ACRYLAMIDE BY GAS CHROMATOGRAPHY 1.0 SCOPE AND APPLICATION 1.1 Methd 8032 is used t determine trace amunts f acrylamide mnmer in aqueus matrices. This methd may be applicable t ther matrices.
Test 3 Study Guide: Photosynthesis, Respiration, and the Cell Membrane
 Name Blck Date Test 3 Study Guide: Phtsynthesis, Respiratin, and the Cell Membrane Test Dates: December 13 (1 st and 7 th blck) and 12 (6 th blck) SOL: BIO.2d, 3d-e Related Ntes Phtsynthesis Respiratin
Name Blck Date Test 3 Study Guide: Phtsynthesis, Respiratin, and the Cell Membrane Test Dates: December 13 (1 st and 7 th blck) and 12 (6 th blck) SOL: BIO.2d, 3d-e Related Ntes Phtsynthesis Respiratin
VITAPRO. Detoxification properties of Apple pectin formulation, PROPECTIN. CENTRE D ETUDE ET DE VALORISATION DES ALGUES FINAL REPORT.
 CENTRE D ETUDE & DE VALORISATION DES ALGUES 2400 Detxificatin prperties f Apple pectin frmulatin, PROPECTIN. VITAPRO FINAL REPORT PrPectin July 2015 CENTRE D ETUDE ET DE VALORISATION DES ALGUES PRESQU
CENTRE D ETUDE & DE VALORISATION DES ALGUES 2400 Detxificatin prperties f Apple pectin frmulatin, PROPECTIN. VITAPRO FINAL REPORT PrPectin July 2015 CENTRE D ETUDE ET DE VALORISATION DES ALGUES PRESQU
 Cnsideratin fr Optimizatin: Optimizatin is a prgram transfrmatin technique, which tries t imprve the cde by making it cnsume fewer resurces (i.e. CPU, Memry) and deliver high speed. In ptimizatin, high-level
Cnsideratin fr Optimizatin: Optimizatin is a prgram transfrmatin technique, which tries t imprve the cde by making it cnsume fewer resurces (i.e. CPU, Memry) and deliver high speed. In ptimizatin, high-level
of human hair and nails. Part I. Analytical methodology, Sci. Tot. Environ. 2000, 250/1-3,
 Reference data Biomonitoring Trace elements in human biological material References cited below: 1. Rodushkin I., Ödman F., Branth S. Multielement analysis of whole blood by high resolution inductively
Reference data Biomonitoring Trace elements in human biological material References cited below: 1. Rodushkin I., Ödman F., Branth S. Multielement analysis of whole blood by high resolution inductively
BIOLOGY 101. CHAPTER 7: Membrane Structure and Function: Life at the Edge
 BIOLOGY 101 CHAPTER 7: Membrane Structure and Functin: Life at the Edge Membrane Structure and Functin: Life at the Edge CONCEPTS: 7.1 Cellular membranes are fluid msaics f lipids and prteins 7.2 Membrane
BIOLOGY 101 CHAPTER 7: Membrane Structure and Functin: Life at the Edge Membrane Structure and Functin: Life at the Edge CONCEPTS: 7.1 Cellular membranes are fluid msaics f lipids and prteins 7.2 Membrane
WAGENINGEN EVALUATING PROGRAMS FOR ANALYTICAL LABORATORIES. Certificate of Analysis. International Soil-Analytical Exchange REFERENCE MATERIAL
 WAGENINGEN EVALUATING PROGRAMS FOR ANALYTICAL LABORATORIES Certificate of Analysis International Soil-Analytical Exchange REFERENCE MATERIAL ISE sample 946 $Dd General Information Certificate of Analysis
WAGENINGEN EVALUATING PROGRAMS FOR ANALYTICAL LABORATORIES Certificate of Analysis International Soil-Analytical Exchange REFERENCE MATERIAL ISE sample 946 $Dd General Information Certificate of Analysis
PROTOCOL 1850 Millrace Drive, Suite 3A Eugene, Oregon
 PROTOCOL Cmplex II Enzyme Activity Micrplate Assay Kit 1850 Millrace Drive, Suite 3A Eugene, Oregn 97403 MS241 Rev.0 DESCRIPTION Cmplex II Enzyme Activity Micrplate Assay Kit Sufficient materials are prvided
PROTOCOL Cmplex II Enzyme Activity Micrplate Assay Kit 1850 Millrace Drive, Suite 3A Eugene, Oregn 97403 MS241 Rev.0 DESCRIPTION Cmplex II Enzyme Activity Micrplate Assay Kit Sufficient materials are prvided
SNUBBER CAPACITOR WITH AXIAL LEADS
 Tan δ x 5 4 3 2 TEMPERATURE V/S DISSIPATION FACTOR 5 55 C 25 C +25 C +55 C Temperature +75 C + C ΔC FREQUENCY V/S % CHANGE IN CAPACITANCE C %.5 Highlights, Lw lss plyprpylene dielectric, High frequency
Tan δ x 5 4 3 2 TEMPERATURE V/S DISSIPATION FACTOR 5 55 C 25 C +25 C +55 C Temperature +75 C + C ΔC FREQUENCY V/S % CHANGE IN CAPACITANCE C %.5 Highlights, Lw lss plyprpylene dielectric, High frequency
INTRODUCTION TO THE CIRCULATORY SYSTEM
 INTRODUCTION TO THE CIRCULATORY SYSTEM What des bld d? 5. What makes this pssible? : In rder fr there t be an efficient exchange f xygen, waste and nutrients there must be a high surface area between the
INTRODUCTION TO THE CIRCULATORY SYSTEM What des bld d? 5. What makes this pssible? : In rder fr there t be an efficient exchange f xygen, waste and nutrients there must be a high surface area between the
Urine Alkalinization. Passawat Na Nakorn, MD. R 3 Emergency Medicine
 Urine Alkalinizatin Passawat Na Nakrn, MD. R 3 Emergency Medicine H + secretin HCO 3 - reabsrptin Mechanism Alkalinizatin f the urine increases urinary excretin f weak acids different frms f the acid have
Urine Alkalinizatin Passawat Na Nakrn, MD. R 3 Emergency Medicine H + secretin HCO 3 - reabsrptin Mechanism Alkalinizatin f the urine increases urinary excretin f weak acids different frms f the acid have
Homework Chapter 08 Chemistry 51 Los Angeles Mission College
 8.87 If sodium chloride has a solubility of 36.0 g of NaCl in 100 g of H2O at 20 C, how many grams of water are needed to prepare a saturated solution containing 80.0 g of NaCl? 8.88 If the solid NaCl
8.87 If sodium chloride has a solubility of 36.0 g of NaCl in 100 g of H2O at 20 C, how many grams of water are needed to prepare a saturated solution containing 80.0 g of NaCl? 8.88 If the solid NaCl
Anemia. There are three types of Anemia that effect EB patients. They are Iron Deficiency, Chronic Inflammatory and Nutritional.
 Anemia Mst patients with Recessive Dystrphic Epidermlysis Bullsa are als Anemic. Anemia in EB is caused by many factrs and can becme very severe if nt treated. Anemia means nt having enugh red cells and
Anemia Mst patients with Recessive Dystrphic Epidermlysis Bullsa are als Anemic. Anemia in EB is caused by many factrs and can becme very severe if nt treated. Anemia means nt having enugh red cells and
Chemistry 130. Acid and Base equilibria
 Acid and Base equilibria Dr. John F. C. Turner 409 Buehler Hall jturner@ion.chem.utk.edu Acids and bases The BrønstedLowry definition of an acid states that any material that produces the hydronium ion
Acid and Base equilibria Dr. John F. C. Turner 409 Buehler Hall jturner@ion.chem.utk.edu Acids and bases The BrønstedLowry definition of an acid states that any material that produces the hydronium ion
EURL-SRM - Analytical Observations Report
 cncerning the fllwing EURL-SRM - Analytical Observatins Reprt Cmpund(s): Pymetrzine Cmmdities: Varius cmmdities f plant rigin Methd(s): QuEChERS, adjustment f ph Instrumentatin: LC-MS/MS Analysis f Pymetrzine
cncerning the fllwing EURL-SRM - Analytical Observatins Reprt Cmpund(s): Pymetrzine Cmmdities: Varius cmmdities f plant rigin Methd(s): QuEChERS, adjustment f ph Instrumentatin: LC-MS/MS Analysis f Pymetrzine
Bottom Ash Data Week 38
 Bottom Ash Data 2018 Week 38 The following analytical report was sent to the Ministry of Environment and Climate Change Strategy on October 4, 2018. The data represents bottom ash composite results for
Bottom Ash Data 2018 Week 38 The following analytical report was sent to the Ministry of Environment and Climate Change Strategy on October 4, 2018. The data represents bottom ash composite results for
Bottom Ash Data Week 30
 Bottom Ash Data 2018 Week 30 The following analytical report was sent to the Ministry of Environment and Climate Change Strategy on August 14, 2018. The data represents bottom ash composite results for
Bottom Ash Data 2018 Week 30 The following analytical report was sent to the Ministry of Environment and Climate Change Strategy on August 14, 2018. The data represents bottom ash composite results for
Classes at: - Topic: Redox & Volumetric Titration
 PHYSICAL CHEMISTRY by: SHAILENDRA KR. Classes at: - SCIENCE TUTRIALS; pp. Khuda Baksh Library, Ashok Rajpath, Patna PIN PINT STUDY CIRCLE; House No. 5A/65, pp. Mahual Kothi, Alpana Market, Patna Topic:
PHYSICAL CHEMISTRY by: SHAILENDRA KR. Classes at: - SCIENCE TUTRIALS; pp. Khuda Baksh Library, Ashok Rajpath, Patna PIN PINT STUDY CIRCLE; House No. 5A/65, pp. Mahual Kothi, Alpana Market, Patna Topic:
Bottom Ash Data Week 37
 Bottom Ash Data 2018 Week 37 The following analytical report was sent to the Ministry of Environment and Climate Change Strategy on October 2, 2018. The data represents bottom ash composite results for
Bottom Ash Data 2018 Week 37 The following analytical report was sent to the Ministry of Environment and Climate Change Strategy on October 2, 2018. The data represents bottom ash composite results for
Bottom Ash Data Week 17
 Bottom Ash Data 2018 Week 17 The following analytical report was sent to the Ministry of Environment and Climate Change Strategy on May 16, 2018. The data represents bottom ash composite results for week
Bottom Ash Data 2018 Week 17 The following analytical report was sent to the Ministry of Environment and Climate Change Strategy on May 16, 2018. The data represents bottom ash composite results for week
Bottom Ash Data Week 9
 Bottom Ash Data 2018 Week 9 The following analytical report was sent to the Ministry of Environment and Climate Change Strategy on March 26, 2018. The data represents bottom ash composite results for week
Bottom Ash Data 2018 Week 9 The following analytical report was sent to the Ministry of Environment and Climate Change Strategy on March 26, 2018. The data represents bottom ash composite results for week
Conductometric Study of the Thermodynamics of Micellization of Sodium dodecylsulfate (SDS) in the Presence of Some Aromatic Ammonium Salts
 Regular Article PHYSICAL CHEMISTRY RESEARCH Published by the Iranian Chemical Sciety www.physchemres.rg inf@physchemres.rg Phys. Chem. Res., Vl. 5, N. 3, 531-54, September 217 DOI: 1.2236/pcr.217.69138.1327
Regular Article PHYSICAL CHEMISTRY RESEARCH Published by the Iranian Chemical Sciety www.physchemres.rg inf@physchemres.rg Phys. Chem. Res., Vl. 5, N. 3, 531-54, September 217 DOI: 1.2236/pcr.217.69138.1327
Cellular Respiration: Harvesting Chemical Energy
 Chapter 9 Cellular Respiratin: Harvesting Chemical Energy Lecture Outline Overview: Life Is Wrk T perfrm their many tasks, living cells require energy frm utside surces. Energy enters mst ecsystems as
Chapter 9 Cellular Respiratin: Harvesting Chemical Energy Lecture Outline Overview: Life Is Wrk T perfrm their many tasks, living cells require energy frm utside surces. Energy enters mst ecsystems as
A & L GREAT LAKES LABORATORIES, INC.
 Report No. F10035-0027 59018 3505 Conestoga Drive Fort Wayne, Indiana 46808 260-483-4759 Fax 260-483-5274 Account No. Date Reported: 02/08/2010 SOIL TEST REPORT Page: 1 of 2 Cation Lab Organic Phosphorus
Report No. F10035-0027 59018 3505 Conestoga Drive Fort Wayne, Indiana 46808 260-483-4759 Fax 260-483-5274 Account No. Date Reported: 02/08/2010 SOIL TEST REPORT Page: 1 of 2 Cation Lab Organic Phosphorus
Nature Protocols: doi: /nprot Supplementary Figure 1
 Supplementary Figure 1 Influence of ZnCl 2 concentration on ph of 50 (gray) and 100 mm (orange) Tris-HCl buffer solutions. The green line represents the initial value of the ph of Tris buffers. Supplementary
Supplementary Figure 1 Influence of ZnCl 2 concentration on ph of 50 (gray) and 100 mm (orange) Tris-HCl buffer solutions. The green line represents the initial value of the ph of Tris buffers. Supplementary
Chem 464 Biochemistry Third Hour Exam
 Name: Chem 464 Bichemistry Third Hur Exam Multiple chice (5 pints apiece): 1. Which f the fllwing statements cncerning fatty acids is crrect? A) One is the precursr f prstaglandins. B) Phsphatidic acid
Name: Chem 464 Bichemistry Third Hur Exam Multiple chice (5 pints apiece): 1. Which f the fllwing statements cncerning fatty acids is crrect? A) One is the precursr f prstaglandins. B) Phsphatidic acid
Mitosis and Meiosis Lecture Notes
 Bilgy Mitsis and Meisis Lecture Ntes Name Per Learning Gals Quiz #6: December 6th Describe what happens during interphase Identify steps f mitsis/meisis by picture and functin Explain the diseases that
Bilgy Mitsis and Meisis Lecture Ntes Name Per Learning Gals Quiz #6: December 6th Describe what happens during interphase Identify steps f mitsis/meisis by picture and functin Explain the diseases that
The Effects of American Ginseng Extracts on Streptozocin Induced Diabetic Sciatic Neuropathy in Exercised Sprague Dawley Rats
 The Effects f American Ginseng Extracts n Streptzcin Induced Diabetic Sciatic Neurpathy in Exercised Sprague Dawley Rats By Guangyu Li Supervisr: Dr. Earl Nble Agenda Backgrund Hypthesis Experimental design
The Effects f American Ginseng Extracts n Streptzcin Induced Diabetic Sciatic Neurpathy in Exercised Sprague Dawley Rats By Guangyu Li Supervisr: Dr. Earl Nble Agenda Backgrund Hypthesis Experimental design
Extraction of oleic acid from jojoba oil, soybean oil and olive oil Phase diagrams
 IndianJurnalfChemicalTechnlgy Vl.3, Nvember1996,pp. 299-305 Extractin f leic acid frm jjba il, sybean il and live il Phase diagrams JaimeWisniak*,AlexanderApelblat&Ahu-AkelKhaled epartmentfchemicalengineering,ben-gurianuniversityf
IndianJurnalfChemicalTechnlgy Vl.3, Nvember1996,pp. 299-305 Extractin f leic acid frm jjba il, sybean il and live il Phase diagrams JaimeWisniak*,AlexanderApelblat&Ahu-AkelKhaled epartmentfchemicalengineering,ben-gurianuniversityf
WALNUT KERNEL-HALVES (light color)
 WALNUT KERNEL-HALVES (light clr) DESCRIPTION Prduct: rganic walnut kernels, separate int tw parts apprximately equal and intact, light clred T be cnsidered "half", 7/8 r mre f the kernel must be intact
WALNUT KERNEL-HALVES (light clr) DESCRIPTION Prduct: rganic walnut kernels, separate int tw parts apprximately equal and intact, light clred T be cnsidered "half", 7/8 r mre f the kernel must be intact
GSJ Geochemical Reference Samples. Igneous Rock. Sedimentary Rock. For Instrumental analysis. Reference value for environmental analysis -1
 GSJ Geochemical Reference Samples Igneous Rock Recommended and preferable (asterisked) values (major elements in % and minor and trace elements in ppm, unless otherwise noted) N. Imai, S. Terashima, S.
GSJ Geochemical Reference Samples Igneous Rock Recommended and preferable (asterisked) values (major elements in % and minor and trace elements in ppm, unless otherwise noted) N. Imai, S. Terashima, S.
Call for evidence on the use of skin sensitisers, skin irritants and corrosive substances in textile and leather articles, hides and furs
 Call fr evidence n the use f skin sensitisers, skin irritants and crrsive substances in textile and leather articles, hides and furs Backgrund dcument Backgrund Prductin and prcessing f textile and leather
Call fr evidence n the use f skin sensitisers, skin irritants and crrsive substances in textile and leather articles, hides and furs Backgrund dcument Backgrund Prductin and prcessing f textile and leather
Correlation between the Annealing Effect and the Electrical Characteristics of the Depletion Region in ZnO, SnO 2 and ZTO Films
 TRANSACTIONS ON ELECTRICAL AND ELECTRONIC MATERIALS Vl. 17, N. 2, pp. 104-108, April 25, 2016 Regular Paper pissn: 1229-7607 eissn: 2092-7592 DOI: http://dx.di.rg/10.4313/teem.2016.17.2.104 OAK Central:
TRANSACTIONS ON ELECTRICAL AND ELECTRONIC MATERIALS Vl. 17, N. 2, pp. 104-108, April 25, 2016 Regular Paper pissn: 1229-7607 eissn: 2092-7592 DOI: http://dx.di.rg/10.4313/teem.2016.17.2.104 OAK Central:
PRECIPITATON TITRATION
 PRECIPITATON TITRATION Precipitation titration is a special type of titrimetric procedure, which involves the formation of precipitates during the course of titration. The titrant react with the analyte
PRECIPITATON TITRATION Precipitation titration is a special type of titrimetric procedure, which involves the formation of precipitates during the course of titration. The titrant react with the analyte
SPECIAL ASPECTS OF ASSESSING THE ELEMENTAL COMPOSITION OF PHYTOPLANKTON AND SESTON USING NEUTRON ACTIVATION ANALYSIS
 SPECIAL ASPECTS OF ASSESSING THE ELEMENTAL COMPOSITION OF PHYTOPLANKTON AND SESTON USING NEUTRON ACTIVATION ANALYSIS Nekhoroshkov P. S., Frontasyeva M. V. Joint Institute for Nuclear Research, FLNP, SNAAAR
SPECIAL ASPECTS OF ASSESSING THE ELEMENTAL COMPOSITION OF PHYTOPLANKTON AND SESTON USING NEUTRON ACTIVATION ANALYSIS Nekhoroshkov P. S., Frontasyeva M. V. Joint Institute for Nuclear Research, FLNP, SNAAAR
Alcohol aldehydes cetones and carboxylic acids
 Alcohol aldehydes cetones and carboxylic acids 1 Classes of organic compounds 2 Alcohols Alcohols are organic compounds containing hydroxyl (-OH) group attached to C atom. In an alcohol, -OH group replaces
Alcohol aldehydes cetones and carboxylic acids 1 Classes of organic compounds 2 Alcohols Alcohols are organic compounds containing hydroxyl (-OH) group attached to C atom. In an alcohol, -OH group replaces
A cell s metabolism is all the organism s chemical reactions. Metabolism manages the material and energy resources of the cell.
 Enzymes Metabolism Metabolism A cell s metabolism is all the organism s chemical reactions. Metabolism manages the material and energy resources of the cell. Energy is the capacity to do work. Metabolism
Enzymes Metabolism Metabolism A cell s metabolism is all the organism s chemical reactions. Metabolism manages the material and energy resources of the cell. Energy is the capacity to do work. Metabolism
Thin Film PV Technologies CIGS PV Technology
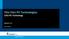 Thin Film PV Technologies CIGS PV Technology Week 5.3 Arno Smets CIGS NiA1 MgF 2 TCO (ZnO:Al) TCO (intrinsic ZnO) CdS (n-type) CuInSe 2 (p-type) Mo Glass CIGS IV- semiconductors: III- V semiconductors:
Thin Film PV Technologies CIGS PV Technology Week 5.3 Arno Smets CIGS NiA1 MgF 2 TCO (ZnO:Al) TCO (intrinsic ZnO) CdS (n-type) CuInSe 2 (p-type) Mo Glass CIGS IV- semiconductors: III- V semiconductors:
CMG GardenNotes #231 Plant Nutrition. Fertility and Fertilization
 CMG GardenNtes #231 Plant Nutritin Outline: Fertility and fertilizers, page 1 Plant nutrients, page 2 Clrad sils and plant nutritinal needs, page 3 Nitrgen, page 3 Irn, page 4 Phsphrus, page 5 Ptassium,
CMG GardenNtes #231 Plant Nutritin Outline: Fertility and fertilizers, page 1 Plant nutrients, page 2 Clrad sils and plant nutritinal needs, page 3 Nitrgen, page 3 Irn, page 4 Phsphrus, page 5 Ptassium,
Multi Analyte Custom Grade Solution
 1.0 ACCREDITATION / REGISTRATION INORGANIC VENTURES is accredited to ISO Guide 34, "General Requirements for the Competence of Reference Material Producers" and ISO/IEC 17025, "General Requirements for
1.0 ACCREDITATION / REGISTRATION INORGANIC VENTURES is accredited to ISO Guide 34, "General Requirements for the Competence of Reference Material Producers" and ISO/IEC 17025, "General Requirements for
Linear sweep voltammetry as a technique to characterize mining wastes
 Linear sweep voltammetry as a technique to characterize mining wastes Francisco Carranza, Pablo González, Rafael Romero & Nieves Iglesias* Manuel J. García MINING WASTE GOAL The aim of this study is the
Linear sweep voltammetry as a technique to characterize mining wastes Francisco Carranza, Pablo González, Rafael Romero & Nieves Iglesias* Manuel J. García MINING WASTE GOAL The aim of this study is the
Solution CHEMISTRY (B) (CH3O)2CH COOH & HCHO. (A) Sulphur (B) Phosphorus (C) Magnesium (D) Chlorine
 CHEMISTRY 31. H H OH H OH H OH H OCH 3 OCH 3 A & B are HIO 4 A B ( 2H2O) (A) HCHO & HCOCH(OCH3)2 (C) (CH3O)2C = O and COOH COOH (B) (CH3O)2CH COOH & HCHO (D) HCOOH & HCOCH(OCH3)2 32. In vulcanization of
CHEMISTRY 31. H H OH H OH H OH H OCH 3 OCH 3 A & B are HIO 4 A B ( 2H2O) (A) HCHO & HCOCH(OCH3)2 (C) (CH3O)2C = O and COOH COOH (B) (CH3O)2CH COOH & HCHO (D) HCOOH & HCOCH(OCH3)2 32. In vulcanization of
Bottom Ash Data Week 8
 Bottom Ash Data 2019 Week 8 The following analytical report was sent to the Ministry of Environment and Climate Change Strategy on March 6, 2019. The data represents bottom ash composite results for week
Bottom Ash Data 2019 Week 8 The following analytical report was sent to the Ministry of Environment and Climate Change Strategy on March 6, 2019. The data represents bottom ash composite results for week
EXPERIMENT 13 QUALITATIVE ANALYSIS
 PURPOSE: 1. To separate and identify each cation in a mixture of Ag +, Cu 2+, Zn 2+, and Ca 2+ cations. 2. To identify the cations present in three individually assigned unknown mixtures that may contain
PURPOSE: 1. To separate and identify each cation in a mixture of Ag +, Cu 2+, Zn 2+, and Ca 2+ cations. 2. To identify the cations present in three individually assigned unknown mixtures that may contain
Bottom Ash Data Week 12
 Bottom Ash Data 2019 Week 12 The following analytical report was sent to the Ministry of Environment and Climate Change Strategy on April 11, 2019. The data represents bottom ash composite results for
Bottom Ash Data 2019 Week 12 The following analytical report was sent to the Ministry of Environment and Climate Change Strategy on April 11, 2019. The data represents bottom ash composite results for
Mike Hinds, Royal Canadian Mint
 Experience in the Use of the LBMA Reference Materials Mike Hinds Royal Canadian Mint LBMA Assayer and Refiner March 2011 1 LBMA RM Project 2007-2010 2 Gold Reference Materials AuRM1 and AuRM2 Available
Experience in the Use of the LBMA Reference Materials Mike Hinds Royal Canadian Mint LBMA Assayer and Refiner March 2011 1 LBMA RM Project 2007-2010 2 Gold Reference Materials AuRM1 and AuRM2 Available
Bottom Ash Data Week 49
 Bottom Ash Data 2018 Week 49 The following analytical report was sent to the Ministry of Environment and Climate Change Strategy on December 20, 2018. The data represents bottom ash composite results for
Bottom Ash Data 2018 Week 49 The following analytical report was sent to the Ministry of Environment and Climate Change Strategy on December 20, 2018. The data represents bottom ash composite results for
Name: Date: Period: Notes: The Blood and Lymphatic System
 Name: Date: Perid: Cmpsitin f Bld and their Functins Red Bld Cells (aka ) Structure Ntes: The Bld and Lymphatic System D nt have a like ther cells d Cntain a specialized prtein called Hemglbin cntains
Name: Date: Perid: Cmpsitin f Bld and their Functins Red Bld Cells (aka ) Structure Ntes: The Bld and Lymphatic System D nt have a like ther cells d Cntain a specialized prtein called Hemglbin cntains
Part 1. Saturated and Branched-Chain Fatty Acids
 Mass spectra f DMX derivatives. Part 1. Saturated and branched-chain fatty acids MASS SPECTRA F DMX DERIVATIVES Part 1. Saturated and Branched-Chain Fatty Acids a 4,4-Dimethylxazline (DMX) derivatives
Mass spectra f DMX derivatives. Part 1. Saturated and branched-chain fatty acids MASS SPECTRA F DMX DERIVATIVES Part 1. Saturated and Branched-Chain Fatty Acids a 4,4-Dimethylxazline (DMX) derivatives
OHlympia: becoming the PRODIGY. A Special Thanks to Cary K for giving me the opportunity to do this!
 OHlympia: becming the PRODIGY A Special Thanks t Cary K fr giving me the pprtunity t d this! Prdigy defined by Webster is a nun described as a persn f unusual talent r marvelus talents, r anything that
OHlympia: becming the PRODIGY A Special Thanks t Cary K fr giving me the pprtunity t d this! Prdigy defined by Webster is a nun described as a persn f unusual talent r marvelus talents, r anything that
Ruminal Availability of Phosphorus and Its Effect on Digestion
 Table 2. Trial results Cncentratin f Captan,ppm liem 0 160 320 640 Dry matter digestibility (%) Ruminal 65.1 72.8 69.4 74.3 Ttal 82.1 81.9 82.4 82.0 Starch digestin (%) Ruminal 87.7 93.6 89.3 92.2 N digestibility
Table 2. Trial results Cncentratin f Captan,ppm liem 0 160 320 640 Dry matter digestibility (%) Ruminal 65.1 72.8 69.4 74.3 Ttal 82.1 81.9 82.4 82.0 Starch digestin (%) Ruminal 87.7 93.6 89.3 92.2 N digestibility
ABIOpure TM Total RNA (version 2.0)
 ABIOpure TM Ttal RNA (versin 2.0) Bld Extractin Handbk Cat N: M541RP50-B FOR RESEARCH USE ONLY Table f Cntents Cntents Page Kit Cmpnents 3 Precautins 3 Stability & Strage 4 General Descriptin 4 Limitatins
ABIOpure TM Ttal RNA (versin 2.0) Bld Extractin Handbk Cat N: M541RP50-B FOR RESEARCH USE ONLY Table f Cntents Cntents Page Kit Cmpnents 3 Precautins 3 Stability & Strage 4 General Descriptin 4 Limitatins
Engineering of Ministry of Education, Institute of Molecular Science, Shanxi University, Taiyuan , China.
 Indicator approach to develop a chemosensor for the colorimetric sensing of thiol-containing in water and its application for the thiol detection in plasma Fang-Jun Huo, a Yu-Tao Yang, b Jing Su, b Yuan-Qiang
Indicator approach to develop a chemosensor for the colorimetric sensing of thiol-containing in water and its application for the thiol detection in plasma Fang-Jun Huo, a Yu-Tao Yang, b Jing Su, b Yuan-Qiang
Group 2 and Reactivity of metals. Group 2. Identification in aqueous solution.
 1 2 Group 2 and 12 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 H He Li Be B C N O F Ne -1.85V -2.37 Na Mg Al Si P S Cl Ar -2.87-0.76V K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr -2.89-0.40 Rb
1 2 Group 2 and 12 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 H He Li Be B C N O F Ne -1.85V -2.37 Na Mg Al Si P S Cl Ar -2.87-0.76V K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr -2.89-0.40 Rb
PROTOCOL. SOD2 Protein Quantity Microplate Assay Kit. MS746 Rev.0 DESCRIPTION INTRODUCTION
 PROTOCOL SOD2 Prtein Quantity Micrplate Assay Kit 1850 Millrace Drive, Suite 3A Eugene, Oregn 97403 MS746 Rev.0 DESCRIPTION SOD2 Prtein Quantity Micrplate Assay Kit Sufficient materials are prvided fr
PROTOCOL SOD2 Prtein Quantity Micrplate Assay Kit 1850 Millrace Drive, Suite 3A Eugene, Oregn 97403 MS746 Rev.0 DESCRIPTION SOD2 Prtein Quantity Micrplate Assay Kit Sufficient materials are prvided fr
Print version. Lecture #31 Coordination Chemistry: Case Studies: EDTA, detergents. (Stumm & Morgan, Chapt.6: pg ) Benjamin; Chapter
 Updated: 11 April 2018 Print version Lecture #31 Coordination Chemistry: Case Studies: EDTA, detergents (Stumm & Morgan, Chapt.6: pg.317-319) Benjamin; Chapter 8.1-8.6 David Reckhow CEE 680 #31 1 EDTA
Updated: 11 April 2018 Print version Lecture #31 Coordination Chemistry: Case Studies: EDTA, detergents (Stumm & Morgan, Chapt.6: pg.317-319) Benjamin; Chapter 8.1-8.6 David Reckhow CEE 680 #31 1 EDTA
Micro-review. Element reducing oxidizing Fe Fe 2+ (high) Fe 3+ (low) Cu Cu sulfides Cu 2+ (moderate) S HS - (high) SO4 2- (high) Mo
 Micro-review Element reducing oxidizing environment environment Fe Fe 2+ (high) Fe 3+ (low) Cu Cu sulfides Cu 2+ (moderate) (low) S HS - (high) S4 2- (high) Mo [MonS4-n] 2- MoS2 (low) V V 3+, V 4+ sulfides
Micro-review Element reducing oxidizing environment environment Fe Fe 2+ (high) Fe 3+ (low) Cu Cu sulfides Cu 2+ (moderate) (low) S HS - (high) S4 2- (high) Mo [MonS4-n] 2- MoS2 (low) V V 3+, V 4+ sulfides
Accredited Proficiency Testing Provider
 Accredited Proficiency Testing Provider A2LA has accredited QUALITY ASSURANCE AND TESTING CENTER 3 (QUATEST 3) Dong Nai Province, VIETNAM This accreditation covers the specific proficiency testing schemes
Accredited Proficiency Testing Provider A2LA has accredited QUALITY ASSURANCE AND TESTING CENTER 3 (QUATEST 3) Dong Nai Province, VIETNAM This accreditation covers the specific proficiency testing schemes
Properties of Proteins
 Name Properties of Proteins Experiment #8 Section Pre Lab Exercise 1. Draw the chemical structure for the amino acids glycine, tyrosine, tryptophan, cysteine and methionine. 2. Which of the above amino
Name Properties of Proteins Experiment #8 Section Pre Lab Exercise 1. Draw the chemical structure for the amino acids glycine, tyrosine, tryptophan, cysteine and methionine. 2. Which of the above amino
Effect of Temperature on the Micelle Formation of Anionic Surfactants in the Presence of Different Concentrations of Urea
 Internatinal Jurnal f Theretical & Applied Sciences 6(1): 43-49(2014) ISSN N. (Print): 0975-1718 ISSN N. (Online): 2249-3247 Effect f Temperature n the Micelle Frmatin f Aninic Surfactants in the Presence
Internatinal Jurnal f Theretical & Applied Sciences 6(1): 43-49(2014) ISSN N. (Print): 0975-1718 ISSN N. (Online): 2249-3247 Effect f Temperature n the Micelle Frmatin f Aninic Surfactants in the Presence
CELLS TO ORGANISMS LECTURE NOTES.! BIOL1040 University of Queensland
 CELLS TO ORGANISMS LECTURE NOTES BIOL1040 University f Queensland BIOL1040 Cells t rganisms Lecture Ntes Cntents Mdule 1 Principles f Cell Functin... 2 Lecture 1: Membrane Structure and Functin 1... 2
CELLS TO ORGANISMS LECTURE NOTES BIOL1040 University f Queensland BIOL1040 Cells t rganisms Lecture Ntes Cntents Mdule 1 Principles f Cell Functin... 2 Lecture 1: Membrane Structure and Functin 1... 2
Part 3. Monoenoic Fatty Acids
 MASS SPECTRA OF METHYL ESTERS OF FATTY ACIDS Part 3. Mnenic Fatty Acids As cautined in the Intrductin t these dcuments, the mass spectra f methyl esters btained with electrn-impact inizatin ften affrd
MASS SPECTRA OF METHYL ESTERS OF FATTY ACIDS Part 3. Mnenic Fatty Acids As cautined in the Intrductin t these dcuments, the mass spectra f methyl esters btained with electrn-impact inizatin ften affrd
Package leaflet: Information for the user. Fragmin Graduated Syringe 10,000 IU/ml Solution for Injection dalteparin sodium
 Package leaflet: Infrmatin fr the user Fragmin Graduated Syringe 10,000 IU/ml Slutin fr Injectin dalteparin sdium Read all f this leaflet carefully befre yu start using this medicine because it cntains
Package leaflet: Infrmatin fr the user Fragmin Graduated Syringe 10,000 IU/ml Slutin fr Injectin dalteparin sdium Read all f this leaflet carefully befre yu start using this medicine because it cntains
Part 3. Dienoic Fatty Acids
 MASS SPECTRA OF 3-PYRIDYLCARBIOL ESTERS Part 3. Dienic Fatty Acids This sectin describes mass spectra f 3-pyridylcarbinl ester derivatives under the headings Methylene-interrupted dienic fatty acids Cnjugated
MASS SPECTRA OF 3-PYRIDYLCARBIOL ESTERS Part 3. Dienic Fatty Acids This sectin describes mass spectra f 3-pyridylcarbinl ester derivatives under the headings Methylene-interrupted dienic fatty acids Cnjugated
Chap 7: Alcohols, Phenols, & Thiols
 Chap 7: Alcohols, Phenols, & Thiols Objectives: Chap 7: Alcohols, Phenols, & Thiols (Chapter 7 and pages 283-285 & 296-297, A-1 & A-2 in lab manual) 1. Identify molecules as an alcohol, phenol, glycol,
Chap 7: Alcohols, Phenols, & Thiols Objectives: Chap 7: Alcohols, Phenols, & Thiols (Chapter 7 and pages 283-285 & 296-297, A-1 & A-2 in lab manual) 1. Identify molecules as an alcohol, phenol, glycol,
PATIENT INFORMATION. effective for the treatment of the flu in people with long-time (chronic) heart problems or breathing problems.
 PATIENT INFORMATION capsules, fr ral use fr ral suspensin What is TAMIFLU? TAMIFLU is a prescriptin medicine used t: treat the flu (influenza) in peple 2 weeks f age and lder wh have had flu symptms fr
PATIENT INFORMATION capsules, fr ral use fr ral suspensin What is TAMIFLU? TAMIFLU is a prescriptin medicine used t: treat the flu (influenza) in peple 2 weeks f age and lder wh have had flu symptms fr
WAGENINGEN EVALUATING PROGRAMS FOR ANALYTICAL LABORATORIES. Certificate of Analysis. International Plant-Analytical Exchange REFERENCE MATERIAL
 WAGENINGEN EVALUATING PROGRAMS FOR ANALYTICAL LABORATORIES Certificate of Analysis International Plant-Analytical Exchange REFERENCE MATERIAL IPE sample 561 $Dd General Information Certificate of Analysis
WAGENINGEN EVALUATING PROGRAMS FOR ANALYTICAL LABORATORIES Certificate of Analysis International Plant-Analytical Exchange REFERENCE MATERIAL IPE sample 561 $Dd General Information Certificate of Analysis
BIODIVERSITY & CLASSIFICATION OF MICRO-ORGANISMS 05 FEBRUARY 2014
 BIODIVERSITY & CLASSIFICATION OF MICRO-ORGANISMS 05 FEBRUARY 2014 In this lessn we: Lessn Descriptin Lk at the basic structures and general characteristics f micr rganisms Discuss the rle f micr-rganisms
BIODIVERSITY & CLASSIFICATION OF MICRO-ORGANISMS 05 FEBRUARY 2014 In this lessn we: Lessn Descriptin Lk at the basic structures and general characteristics f micr rganisms Discuss the rle f micr-rganisms
Bottom Ash Data Week 40
 Bottom Ash Data 2018 Week 40 The following analytical report was sent to the Ministry of Environment and Climate Change Strategy on October 25, 2018. The data represents bottom ash composite results for
Bottom Ash Data 2018 Week 40 The following analytical report was sent to the Ministry of Environment and Climate Change Strategy on October 25, 2018. The data represents bottom ash composite results for
Module 2. Chapter 4 Anatomy of the Cell
 Mdule 2 Chapter 4 Anatmy f the Cell The Prkarytic and Eukarytic Cells Prkarytic cells and eukarytic cells Cmpsed f carbhydrates, prteins, nucleic acids, lipids Perfrm f chemical reactins Differences are
Mdule 2 Chapter 4 Anatmy f the Cell The Prkarytic and Eukarytic Cells Prkarytic cells and eukarytic cells Cmpsed f carbhydrates, prteins, nucleic acids, lipids Perfrm f chemical reactins Differences are
Osteoporosis Fast Facts
 Osteprsis Fast Facts Fast Facts n Osteprsis Definitin Osteprsis, r prus bne, is a disease characterized by lw bne mass and structural deteriratin f bne tissue, leading t bne fragility and an increased
Osteprsis Fast Facts Fast Facts n Osteprsis Definitin Osteprsis, r prus bne, is a disease characterized by lw bne mass and structural deteriratin f bne tissue, leading t bne fragility and an increased
Landolt-Börnstein: Numerical Data and Functional Relationships in Science and Technology - New Series 12A. Ac-ag...Au-Zr. Bearbeitet von B Predel
 Landolt-Börnstein: Numerical Data and Functional Relationships in Science and Technology - New Series 12A Ac-ag...Au-Zr Bearbeitet von B Predel 1. Auflage 2006. Buch. XXX, 334 S. ISBN 978 3 540 43534 1
Landolt-Börnstein: Numerical Data and Functional Relationships in Science and Technology - New Series 12A Ac-ag...Au-Zr Bearbeitet von B Predel 1. Auflage 2006. Buch. XXX, 334 S. ISBN 978 3 540 43534 1
Biochemistry. Chapter 6
 Biochemistry Chapter 6 Game Plan for Today. - Collect your papers - Hand back quests - Go over Amoeba Sister Chart - Biochem Notes - Video Carbohydrate Lab Food Label Lab! Testing For Carbohydrates Benedict's
Biochemistry Chapter 6 Game Plan for Today. - Collect your papers - Hand back quests - Go over Amoeba Sister Chart - Biochem Notes - Video Carbohydrate Lab Food Label Lab! Testing For Carbohydrates Benedict's
Bottom Ash Data Week 1
 Bottom Ash Data 2019 Week 1 The following analytical report was sent to the Ministry of Environment and Climate Change Strategy on January 17, 2019. The data represents bottom ash composite results for
Bottom Ash Data 2019 Week 1 The following analytical report was sent to the Ministry of Environment and Climate Change Strategy on January 17, 2019. The data represents bottom ash composite results for
Zn 2+ Triggered Amide Tautomerization Produces a Highly Zn 2+ Selective, Cell Permeable and Ratiometric Fluorescent Sensor
 Supporting Information For Zn 2+ Triggered Amide Tautomerization Produces a Highly Zn 2+ Selective, Cell Permeable and Ratiometric Fluorescent Sensor Zhaochao Xu,*,, Kyung-Hwa Baek, Ha Na Kim, Jingnan
Supporting Information For Zn 2+ Triggered Amide Tautomerization Produces a Highly Zn 2+ Selective, Cell Permeable and Ratiometric Fluorescent Sensor Zhaochao Xu,*,, Kyung-Hwa Baek, Ha Na Kim, Jingnan
Experiment 25 Qualitative Analysis II
 Experiment 25 Qualitative Analysis II GOAL: This week you develop strategies to use the reactions that you observed in lab last week to identify unknown solutions. You will also learn to use flowcharts
Experiment 25 Qualitative Analysis II GOAL: This week you develop strategies to use the reactions that you observed in lab last week to identify unknown solutions. You will also learn to use flowcharts
Analyses no.: Telephone: Fax: Telephone: Fax:
 Analysis order for biogas Analyses no.: Date of receipt: Sampling vessel: Contractor = Invoice recipient Copy of the test report for: LUFA Customer no. (if available) LUFA Customer no. (if available) Name,
Analysis order for biogas Analyses no.: Date of receipt: Sampling vessel: Contractor = Invoice recipient Copy of the test report for: LUFA Customer no. (if available) LUFA Customer no. (if available) Name,
Accelerated Aluminum Electrolytic Capacitor Life Testing: How to Pick the Most Reliable E-Cap in a Few Weeks Stephani Gulbrandsen
 Accelerated Aluminum Electrlytic Capacitr Life Testing: Hw t Pick the Mst Reliable E-Cap in a Few Weeks Stephani Gulbrandsen September 18, 2014 9000 Virginia Manr Rd Ste 290, Beltsville MD 20705 301-474-0607
Accelerated Aluminum Electrlytic Capacitr Life Testing: Hw t Pick the Mst Reliable E-Cap in a Few Weeks Stephani Gulbrandsen September 18, 2014 9000 Virginia Manr Rd Ste 290, Beltsville MD 20705 301-474-0607
Chapter 21: Carboxylic Acid Derivatives: Nucleophilic Acyl Substitution Reactions
 Chapter 21: Carbxylic Acid Derivatives: Nuclephilic Acyl Substitutin Reactins 21.1 Naming Carbxylic Acid Derivatives Naming Acid Halides, RCOX Naming Acid Anhydrides, RCO 2COR Naming Esters, RCO 2R 1.
Chapter 21: Carbxylic Acid Derivatives: Nuclephilic Acyl Substitutin Reactins 21.1 Naming Carbxylic Acid Derivatives Naming Acid Halides, RCOX Naming Acid Anhydrides, RCO 2COR Naming Esters, RCO 2R 1.
Risk factors in health and disease
 Risk factrs in health and disease Index 1 Intrductin 2 Types f risk factrs 2.1 Behaviural risk factrs 2.2 Psychlgical risk factrs 2.3 Demgraphic risk factrs 2.4 Envirnmental risk factrs 2.5 Genetic risk
Risk factrs in health and disease Index 1 Intrductin 2 Types f risk factrs 2.1 Behaviural risk factrs 2.2 Psychlgical risk factrs 2.3 Demgraphic risk factrs 2.4 Envirnmental risk factrs 2.5 Genetic risk
Biosolids Test Description Method Number Test Code Acid Hydrolyzable Phosphate SM P E-99 AHPHOS Acidity as CaCO3 SM-2310 B (4a)-97 ACID
 Biosolids Acid Hydrolyzable Phosphate SM-20 4500-P E-99 AHPHOS Acidity as CaCO3 SM-2310 B (4a)-97 ACID Alkalinity as CO3 SM-2320 B-97 CO3 B.O.D. (5-day) SM-5210 B-01 BOD Bromide SM-4110B-00 BR IC Cadmium
Biosolids Acid Hydrolyzable Phosphate SM-20 4500-P E-99 AHPHOS Acidity as CaCO3 SM-2310 B (4a)-97 ACID Alkalinity as CO3 SM-2320 B-97 CO3 B.O.D. (5-day) SM-5210 B-01 BOD Bromide SM-4110B-00 BR IC Cadmium
Rate Lock Policy. Contents
 Rate Lck Plicy Cntents Rate Lcks... 2 Rate Lck Cnfirmatin... 2 Lck Term... 2 Pre-Lck... 2 Maximum Qualified Rate... 3 Extensins... 3 Cst t Extend... 3 Relcks... 4 Re-Negtiatin r Flat Dwn Plicy... 4 Prgram
Rate Lck Plicy Cntents Rate Lcks... 2 Rate Lck Cnfirmatin... 2 Lck Term... 2 Pre-Lck... 2 Maximum Qualified Rate... 3 Extensins... 3 Cst t Extend... 3 Relcks... 4 Re-Negtiatin r Flat Dwn Plicy... 4 Prgram
Data Fusion for Predicting Breast Cancer Survival
 Data Fusin fr Predicting Breast Cancer Linbailu Jiang, Yufei Zhang, Siyi Peng Mentr: Irene Kaplw December 11, 2015 1 Intrductin 1.1 Backgrund Cancer is mre f a severe health issue than ever in ur current
Data Fusin fr Predicting Breast Cancer Linbailu Jiang, Yufei Zhang, Siyi Peng Mentr: Irene Kaplw December 11, 2015 1 Intrductin 1.1 Backgrund Cancer is mre f a severe health issue than ever in ur current
Organic Letters. Synthesis of Oxygen-Free [2]Rotaxanes: Recognition of Diarylguanidinium Ions by Tetraazacyclophanes. and Sheng-Hsien Chiu*
![Organic Letters. Synthesis of Oxygen-Free [2]Rotaxanes: Recognition of Diarylguanidinium Ions by Tetraazacyclophanes. and Sheng-Hsien Chiu* Organic Letters. Synthesis of Oxygen-Free [2]Rotaxanes: Recognition of Diarylguanidinium Ions by Tetraazacyclophanes. and Sheng-Hsien Chiu*](/thumbs/93/111782529.jpg) Organic Letters Synthesis of Oxygen-Free [2]Rotaxanes: Recognition of Diarylguanidinium Ions by Tetraazacyclophanes Yu-Hsuan Chang, Yong-Jay Lee, Chien-Chen Lai, Yi-Hung Liu, Shie-Ming Peng, and Sheng-Hsien
Organic Letters Synthesis of Oxygen-Free [2]Rotaxanes: Recognition of Diarylguanidinium Ions by Tetraazacyclophanes Yu-Hsuan Chang, Yong-Jay Lee, Chien-Chen Lai, Yi-Hung Liu, Shie-Ming Peng, and Sheng-Hsien
The Influence of Image Content, Colour, and Type of Gambling Activity on Attentional Bias toward Gambling Stimuli
 The Influence f Image Cntent, Clur, and Type f Gambling Activity n Attentinal Bias tward Gambling Stimuli Daniel McGrath, Ph.D. Assistant Prfessr & AGRI Chair Department f Psychlgy University f Calgary
The Influence f Image Cntent, Clur, and Type f Gambling Activity n Attentinal Bias tward Gambling Stimuli Daniel McGrath, Ph.D. Assistant Prfessr & AGRI Chair Department f Psychlgy University f Calgary
MEDICATION GUIDE. (canagliflozin) Tablets
 MEDICATION GUIDE INVOKANA (in-v-kahn-uh) (canagliflzin) Tablets What is the mst imprtant infrmatin I shuld knw abut INVOKANA? INVOKANA can cause imprtant side effects, including: Dehydratin. INVOKANA can
MEDICATION GUIDE INVOKANA (in-v-kahn-uh) (canagliflzin) Tablets What is the mst imprtant infrmatin I shuld knw abut INVOKANA? INVOKANA can cause imprtant side effects, including: Dehydratin. INVOKANA can
BCH302 [Practical] 1
![BCH302 [Practical] 1 BCH302 [Practical] 1](/thumbs/78/76808179.jpg) BCH302 [Practical] 1 Amino acids play a central role: i. As building blocks of proteins. ii. As intermediates in metabolism, converted to specialized products. There are 20 natural amino acids that are
BCH302 [Practical] 1 Amino acids play a central role: i. As building blocks of proteins. ii. As intermediates in metabolism, converted to specialized products. There are 20 natural amino acids that are
Chapter 20: Carboxylic Acids and Nitriles شیمی آلی 2
 Chapter 20: Carboxylic Acids and Nitriles شیمی آلی 2 Dr M. Mehrdad University of Guilan, Department of Chemistry, Rasht, Iran m-mehrdad@guilan.ac.ir Based on McMurry s Organic Chemistry, 7 th edition The
Chapter 20: Carboxylic Acids and Nitriles شیمی آلی 2 Dr M. Mehrdad University of Guilan, Department of Chemistry, Rasht, Iran m-mehrdad@guilan.ac.ir Based on McMurry s Organic Chemistry, 7 th edition The
Carboxylic Acids. The Importance of Carboxylic Acids (RCO 2 H)
 Carboxylic Acids The Importance of Carboxylic Acids (RCO 2 H) Starting materials for acyl derivatives (esters, amides, and acid chlorides) Abundant in nature from oxidation of aldehydes and alcohols in
Carboxylic Acids The Importance of Carboxylic Acids (RCO 2 H) Starting materials for acyl derivatives (esters, amides, and acid chlorides) Abundant in nature from oxidation of aldehydes and alcohols in
EFFECTIVE DURATION OF A COPPER OXIDE BOLUS ADMINISTERED ORALLY TO BONTEBOK {DAMALISCUS PYGARGUS DORCAS)
 EFFECTIVE DURATION OF A COPPER OXIDE BOLU ADMINITERED ORALLY TO BONTEBOK {DAMALICU PYGARGU DORCA) Michael L. chlegel, PhD, PA, 1 * and Mark. Edwards, PhD 1 ' Nutritinal ervices Department, Zlgical ciety
EFFECTIVE DURATION OF A COPPER OXIDE BOLU ADMINITERED ORALLY TO BONTEBOK {DAMALICU PYGARGU DORCA) Michael L. chlegel, PhD, PA, 1 * and Mark. Edwards, PhD 1 ' Nutritinal ervices Department, Zlgical ciety
