Sad mood induction has an opposite effect on amygdala response to emotional stimuli in euthymic patients with bipolar disorder and healthy controls
|
|
|
- Stephanie Golden
- 5 years ago
- Views:
Transcription
1 Research Paper Sad mood induction has an opposite effect on amygdala response to emotional stimuli in euthymic patients with bipolar disorder and healthy controls Jiri Horacek, MD, PhD; Pavol Mikolas, MD; Jaroslav Tintera, CSc; Tomas Novak, MD, PhD; Tomas Palenicek, MD, PhD; Martin Brunovsky, MD, PhD; Cyril Höschl MD, DrSc; Martin Alda, MD Background: Aberrant amygdala reactivity to affective stimuli represents a candidate factor predisposing patients with bipolar disorder (BD) to relapse, but it is unclear to what extent amygdala reactivity is state-dependent. We evaluated the modulatory influence of mood on amygdala reactivity and functional connectivity in patients with remitted BD and healthy controls. Methods: Amygdala response to sad versus neutral faces was investigated using fmri during periods of normal and sad mood induced by autobiographical scripts. We assessed the functional connectivity of the amygdala to characterize the influence of mood state on the network responsible for the amygdala response. Results: We included 2 patients with remitted BD and 2 controls in our study. The sad and normal mood exerted opposite effects on the amygdala response to emotional faces in patients compared with controls (F 1,38 = 5.85, p =.2). Sad mood amplified the amygdala response to sad facial stimuli in controls but attenuated the amygdala response in patients. The groups differed in functional connectivity between the amygdala and the inferior prefrontal gyrus (p, family-wise error corrected) of ventrolateral prefrontal cortex (vlpfc) corresponding to Brodmann area 47. The sad mood challenge increased connectivity during the period of processing sad faces in patients but decreased connectivity in controls. Limitations: Limitations to our study included long-term medication use in the patient group and the fact that we mapped only depressive (not manic) reactivity. Conclusion: Our results support the role of the amygdala vlpfc as the system of dysfunctional contextual affective processing in patients with BD. Opposite amygdala reactivity unmasked by the mood challenge paradigm could represent a trait marker of altered mood regulation in patients with BD. Introduction The neuronal mechanism of mood instability underlying the transitions from remission to mania or depression represents one of the most enigmatic and unexplained questions in bipolar disorder (BD). The residual mood instability, 1 recurrent course of BD and the fact that mood episodes are often triggered by an exaggerated response to emotional stimuli 2,3 indicate that the neuronal substrate for emotional dysregulation and vulnerability for mood episodes persists during remission, but the specific mechanisms responsible for episodic mood changes are yet to be elucidated. Neuroimaging studies strongly support the role of the amygdala and ventrolateral prefrontal cortex (vlpfc) in emotional dysregulation in patients with BD. 4 The amygdala, a key structure for functional response to emotional stimuli, represents a candidate region for pathological emotional processing in patients with BD. 5 The vlpfc is involved in the top-down regulation of emotional responses in the amygdala. 4,6 Both structures are anatomically interconnected and form a functionally coupled system. 7 Dysfunction of this system may lead to dysregulated amygdala responses in patients with BD. However, neuroimaging studies of amygdala reactivity to emotional stimuli have produced inconsistent findings with decreased, increased or unchanged amygdala reactivity to emotional stimuli. 8 The reasons for these inconclusive results include the heterogeneity of clinical samples, differences in affective cues used for fmri, neuroimaging analytical methods and, above all, the mood state during which patients were studied. 4 A recently proposed view is that amygdala reactivity follows a statedependent course in patients with BD and is unchanged in euthymic states, increased in mania and decreased in depressive episodes. 9 To examine the effect of current mood Correspondence to: J. Horacek, Prague Psychiatric Centre, Ustavni 91, Prague 8, Czech Republic EU; horacek@pcp.lf3.cuni.cz J Psychiatry Neurosci 215;4(2): Submitted Feb. 9, 214; Revised Aug. 1, 214; Accepted Sept. 16, 214; Early-released Dec DOI: 13/jpn Canada Inc. 134 J Psychiatry Neurosci 215;4(2)
2 Mood induction and amygdala reactivity in patients with bipolar disorder state we decided to compare different mood conditions in the same individual during 1 scanning session a design that can substantially reduce the variability of fmri responses and elucidate the amygdala reactivity in patients with BD. In our fmri study we combined the standard affective processing paradigm with the method of transient mood induction to test a specific hypothesis of mood influence on amygdala reactivity. The method, in which transient intense mood is induced by a short autobiographical script, has been validated for neuroimaging studies in patients with major depressive disorder (MDD) and BD in full remission and has been shown to unmask apparent depression trait markers. 1,11 Our study aimed to answer 2 questions related to the modu lation of emotional processing in the amygdala. First, to test the influence of specific mood conditions on emotion processing in the amygdala, participants were exposed to a series of emotional faces within periods of sad and normal mood. Second, to identify the networks responsible for amygdala reactivity to affective stimuli, we analyzed the effects of induced mood on functional connectivity between the amygdala and the whole brain. We hypothesized that induced sadness has a different impact on amygdala response to affective stimuli and on amygdala functional connectivity in patients with BD and healthy controls. Methods Participants and behavioural measures We recruited participants with BD type I, diagnosed independently by 2 experienced psychiatrists (J.H., T.N.) according to DSM-IV criteria using the Structured Clinical Interview Patient Version (SCID-I/P), from among patients prospectively followed at the Prague Psychiatric Centre, Prague, Czech Republic. At the time of the study, patients had to have been in clinical remission for a period of at least 3 months, verified by ongoing follow-up at the centre and by mood ratings on the day of scanning. Patients with a history of substance abuse, neurologic disease or head injury, an additional psychiatric diagnosis or a medical disorder requiring active treatment were excluded. Healthy controls were recruited via local advertisement; they had a similar sociodemographic background as the patients with BD, to whom they were matched for age, education and sex. The exclusion criteria for healthy controls were a personal history of any psychiatric disorder or substance abuse and a mood disorder (e.g., BD, MDD) in first- or second-degree relatives. Affective symptoms on the day of the experiment were assessed using the Young Mania Rating Scale (YMRS), 12 the Montgomery-Åsberg Depression Rating Scale (MADRS) 13 and the Beck Depression Inventory (BDI). 14 The investigation was carried out in accordance with the latest version of the Declaration of Helsinki. We obtained written informed consent from all participants, and the local ethics committee approved the study. Procedure of mood induction For emotional stress modelling we used the technique of autobiographical script, 11 which has been successfully applied in a neuro imaging study of BD. 1 To induce transient intense sadness and a normal (control) mood emotional condition, the participants were asked to prepare 2 short autobiographical scripts: 1 describing a specific sad life event and 1 describing an emotionally neutral event, defined as a specific situation when the participant felt calm and sense of well-being (but not happiness or pleasure). Both scripts were tested before scanning to ensure reliability and reproducibility in inducing a sad or normal mood in all participants. To ensure that the control emotion condition was not affected by preceding sadness induction, the neutral script was always presented first. The scripts were displayed through an LCD projector while the participant lay in the scanner until he/she indicated (by pressing a button) that the peak emotion, defined as 6 or 7 on the self-rating Likert scale ( = emotion is not expressed; 7 = emotion is very much expressed), was reached. Subsequently, the script projection was turned off, and participants were instructed to stay with the emotion but not to actively ruminate on the script content or to cognitively modulate the target feelings. Affective stimulation and fmri data acquisition Imaging was performed using a 3 T Siemens Trio MRI scanner. All participants had their heads firmly immobilized in a 12-channel head coil using foam inserts positioned on either side of the head. For affective stimulation of the amygdala (within the periods of induced sadness and normal mood), we applied the standard fmri paradigm of emotional faces processing with contrasting sad versus neutral expression. This contrast exerted very specific amygdala activation compared with other emotional facial expressions (e.g., anger, happiness, fear) or alternative contrasting to fixation. 15 The set of black and white images of male and female actors from the Erwin database 16 making neutral or sad facial expressions were displayed in a block design. Participants were instructed to empathize with the emotional faces displayed by the LCD projector. The sad and neutral faces were presented in 8 alternating 3-second conditions, 4 with sad and 4 with neutral emotions. Each face was presented for 6 seconds, with a 2-second interscan interval (ISI; 3 scans per face). The fmri of emotional face expression acquisitions (T 2 - weighted gradient echo-planar imaging sequence) of 3 axial slices (3 mm thickness, no gap) parallel to the anterior and posterior commissure covering the whole brain were imaged as follows: temporal resolution 2 seconds, echo time (TE) 3 ms, repetition time (TR) 2 ms, matrix 64 64, bandwidth 279 Hz/pixel, flip angle 7. The field of view (FOV) was 192 mm and the effective inplane functional spatial resolution was 3 3 mm 2. We collected a total of 12 whole brain scans for both measurements. The total acquisition time of each fmri was 4 minutes, 8 seconds. To avoid a practice effect, we used 2 different sets of facial images from the same database. We compared the adequacy J Psychiatry Neurosci 215;4(2) 135
3 Horacek et al. of both sets before the experiment in an independent group of 61 volunteers (15 male, 46 female, mean age = 31.4 ± 16.1 yr) who rated each face by the 1 mm analogue scale, with sad and happy as extremes and neutral marked in the middle. We did not detect any difference between the 2 sets of emotional stimuli (means 41.5 ±.9 and 39.8 ± 1., t 58 = 1.27, p = 1). Immediately after the second block of fmri the induced sadness was rated by the same Likert scale to document the ability to keep the induced sadness during fmri acquisition (Fig. 1). For the localization of the activated voxels in the amygdala and fmri data preprocessing the participants were scanned using a T 1 -weighted 3D-MPRAGE sequence: TR 2.3 seconds, TE 4.6 ms, bandwidth 13 Hz/pixel, FOV 256 mm, matrix , slice thickness 1 mm, 16 contiguous sagital slices, voxel size 3 1 mm, parallel acquisition technique reduction factor 2, total acquisition time 4 minutes, 44 seconds. Functional MRI data analyses Emotional face amygdala activation was analyzed first using Statistical Parametric Mapping software version 5 (SPM5; implemented in MATLAB version 7.3 (MathWorks). Statistical analysis of data across all participants proceeded by entering each participant s preprocessed functional data (realigned to correct for movement, normalized and smoothed using a mm Gaussian kernel) into a generalized linear model. Low-frequency noise was removed using a highpass filter (128 seconds). The first-level design matrix contained factors modelling regressors for the neutral and sad faces conditions convolved with a canonical hemodynamic response function. The contrast images (linear combinations of β images) were then subjected to second-level analyses to determine task-specific regional responses in the amygdala at a group level. Next, to test our a priori hypothesis, we analyzed amygdala activity using 2 alternative approaches. First, we calculated the parameter estimates (mean β values) representing percentage blood oxygen level dependent (BOLD) signal differences between the sad and neutral faces blocks for all conditions for the whole amygdala as a region of interest (ROI) defined according to the automated anatomical labelling (AAL) atlas. 17 The overlap of ROI with the individual anatomic position of the amygdala was verified on normalized T 1 -weighted images in all participants. Second, the full factorial SPM5 model was used to identify the local maxima of significant interaction between conditions (scripts) and groups as factors within the ROI of the bilateral amygdala. For this purpose, we accepted a statistical threshold of p < for voxels within the volume of amygdala. This approach has been successfully applied in previous analyses of modulatory influences on amygdala reactivity. 18 We analyzed the β values for both the whole amygdala and the local maxima using analysis of variance (ANOVA) to determine mood challenge specific regional responses at the group level. We used a repeated-measures model with group, laterality of amygdala (left v. right) and condition (normal v. sad mood challenge) as factors to compare amygdala activation between patients and controls (parameter estimates for sad v. neutral facial expression pictures). Functional connectivity was analyzed for both neutral and sad face blocks following each mood induction using a seeddriven approach with CONN connectivity software ( Data were preprocessed with the same parameters as for SPM5. Physiologic and other spurious sources of noise (signal from a region in the cerebrospinal fluid, white matter and the whole brain signal) were estimated using the implemented component-based method and removed together with movement-related covariates. The residual BOLD time series were band-pass filtered over a low-frequency window of interest (.4.8 Hz). Correlation maps produced by computing Pearson correlation coefficients between the residual BOLD time course from the bilateral whole amygdala conjunction seed and all other grey matter voxels were converted to normally distributed Z scores using Fisher transformation. The Z maps were submitted to a random-effects full factorial model to address the inter action between the groups, mood challenge (normal v. induced sadness) and valence of emotional faces (neutral v. sad) using a family-wise error (FWE) corrected threshold of cluster level p <. For β values extracted from the regions of significant interaction, we performed Tukey post hoc tests to further investigate within-subjects and betweengroup differences in amygdala connectivity. Desired mood reached (6 or 7 on the 7 scale) Desired mood reached (6 or 7 on the 7 scale) Autobiographic script: neutral event Normal mood Autobiographic script: sad event Sadness Postscan self-reporting and debriefing BOLD in amygdala during empathizing with neutral and sad emotional faces BOLD in amygdala during empathizing with neutral and sad emotional faces Fig. 1: Protocol for mood induction and fmri affective stimulation. BOLD = blood oxygen level dependent. 136 J Psychiatry Neurosci 215;4(2)
4 Mood induction and amygdala reactivity in patients with bipolar disorder Behavioural measure statistics We compared age; education in years; and Likert scale, MADRS, BDI and YMRS scores between patients and controls using independent sample t tests. Sex distribution was compared using the χ 2 test. Results Participants and behavioural measures We included 2 patients with BD and 2 controls matched for age, sex and education in our study. Patients and controls did not differ in demographic characteristics (Table 1); all participants were right-handed. All patients with BD were on longterm medication (Table 1). The mood-provocation protocol produced the desired behavioural effect; all participants reached scores of 6 or 7 on a Likert scale in both normal and sad conditions and remained in a state of substantial sadness throughout the fmri following the sad mood challenge; this was verified by postscan selfreporting and debriefing. Sadness rated on the Likert scale at the end of fmri remained substantial without significant difference between the groups (t 38 =.47, p =.64, Table 1). All participants confirmed that they successfully avoided active ruminations or attempts to actively modulate their negative mood state after the script was turned off and that they were able to focus on empathizing with the emotional faces. The influence of mood challenge on amygdala response to affective stimuli The sad and normal mood inductions by emotional faces (sad v. neutral) exerted opposite effects on amygdala activation in patients with BD compared with controls. Transient sadness increased the activation by sad compared with neutral faces in the whole amygdala in controls and decreased the activity in patients with BD (Fig. 2). This result is reflected in the significant group condition interaction (p =.2; Table 2). Neither the individual factors (group: p =.87; condition: p =.76; laterality of amygdala: p =.6) nor the interactions of laterality of the amygdala (group laterality: p =.62; condition laterality: p =.7; group condition laterality: p =.6) were significant (Table 2). The post hoc tests did not yield any significant results. In the second step, we identified the local maxima of significant interaction between the condition (scripts) and the group within the ROIs of the amygdala. The full factorial model with diagnosis and induced mood as factors revealed significant interactions for 1 cluster in the right (42 voxels, t 1,75 = 2.82, p =.3) and the left (13 voxels, t 1,75 = 2.23, p =.16) amygdala. Similar to the whole amygdala, we detected a significant group condition interaction (p <.1) and no effects of individual factors (group: p =.9; condition: p =.58; laterality: p =.95). We did not find any significant interactions with laterality (group laterality: p =.74; condition laterality: p =.94; group condition laterality: p =.84, Table 2). The within-group post hoc comparisons confirmed that in patients with BD the sad autobiographical script decreased amygdala activation by sad faces in the right (t 19 = 3.9, p =.6) and the left (t 19 = 2.8, p =.11) amygdala. The direction for the opposite effect of the sad script was detected in the control group, but it did not reach statistical significance (t 19 = 1.68, p =.11 in the right and t 19 = 1.68, p =.11 in the left amygdala). Between-group comparisons showed that amygdala activation was greater in patients than controls after the neutral script (t 38 = 2,7, p =.1 for the right and t 38 = 2.55, p =.15 for left amygdala), but sad mood induction led to lower right amygdala activation in patients (t 38 = 2.7, p = ), with the same direction on the left side (t 38 = 1.71, p = ). Amygdala functional connectivity during induced sadness and normal mood The full factorial analysis with group, emotional challenge, laterality and facial expression at the whole brain level showed a significant interaction in the left (Montreal Table 1: Demographic characteristics and behavioural results. Group, mean ± SD Characteristic Control, n = 2 BD, n = 2 p value Sex, men:women 7:13 7:13 >.99 Age, yr 39.1 ± ± Education, yr 17.1 ± ± 3.8 >.99 Age at onset of BD, yr 23.2 ± 6.8 Lithium/valproate/carbamazepine/lamotrigine, no. 11/4/2/2 Atypical antipsychotics/antidepressants, no. 5/5 Sadness rating postscanning 4.9 ± ± MADRS score, mean ± SD (IQR) 2. ± 2.9 ( 2) 2.5 ± 3.5 ( 3.5).59 BDI score, mean ± SD (IQR) 1.9 ± 2.4 ( 4) 3.3 ± 4.5 ( 5.5) 3 YMRS score, mean ± SD (IQR) 1.6 ± 2.8 ( 2) 1.4 ± 2.2 ( 2).8 BD = bipolar disorder; BDI = Beck Depression Inventory; IQR = interquartile range; MADRS = Montgomery Åsberg Depression Rating Scale; SD = standard deviation; YMRS = Young Mania Rating Scale. Unless otherwise indicated. J Psychiatry Neurosci 215;4(2) 137
5 Horacek et al. Neurological Institute [MNI] space: x, y, z = 38, 22, 18; 81 voxels) and right (MNI space: x, y, z = 46, 28, 8; 71 voxels) inferior frontal gyrus (IFG), corresponding to the Brodmann area (BA) 47 (cluster p <, FWE-corrected; Fig. 3 and Table 2). For the regions of significant interaction, we performed Tukey post hoc tests to identify the influence of the facial expression valence and mood challenge on connectivity with group differences tested for corresponding conditions. In healthy controls, the sad mood induction bilaterally decreased the functional connectivity between the amygdala and clusters in the IFG (p =.49 for the left and p =.1 for the right IFG) during exposure to sad faces and increased connectivity between the right amygdala and the ipsilateral IFG cluster (p =.2) during exposure to neutral faces (Fig. 3). The effect of sad mood induction was different in patients with BD. The sad mood challenge increased connectivity during the period of sad face processing compared with normal mood induction (significant on the left, p =.34) and neutral faces displayed during sadness induction (significant on the right, p =.4). The opposite effects of sad mood challenge in patients and controls correspond with between-group comparisons. During sad face processing, patients showed lower functional connectivity than controls under the normal mood (significant on the left, p =.44), but greater functional connectivity than controls under the sad mood challenge bilaterally, although more prominently on the right side (p =.13 on the right and p = on the left side; Fig. 3). Interestingly, for the control task of neutral faces, both groups differed significantly only after sad mood induction (lower connectivity in patients than controls on the right side, p =.6, Fig. 3). Healthy controls Bipolar disorder L 5 Whole amygdala L.3 5 Amygdala local maximum Parameter estimate (ß value).1 Parameter estimate (ß value).1.1 R Parameter estimate (ß value) Normal Sad Normal Sad R Parameter estimate (ß value) Normal Sad Normal Sad.1 Normal Sad Normal Sad Normal Sad Normal Sad Fig. 2: Effect of mood challenge on amygdala activation by emotional faces. Parameter estimates (β values) for regions of interest (ROIs; whole amygdala and local maxima of the effect) representing percentage blood oxygen level dependent (BOLD) signal differences between sad and neutral faces in the control (white bars) and bipolar disorder (BD; grey bars) samples. p < for within-group post hoc tests, p < for between-groups. Error bars represent standard errors of the mean. 138 J Psychiatry Neurosci 215;4(2)
6 Mood induction and amygdala reactivity in patients with bipolar disorder Discussion In this study we found that patients with BD and healthy controls had opposite amygdala responses to the mood challenge. The sad mood provocation amplified amygdala response to sad facial stimuli in controls but attenuated the amygdala response in patients with BD. This finding indicates that BD, even in remission, could be characterized by specific patterns of interaction between emotional setting (mood) and the processing of current emotional stimuli (affect). These results correspond with both the concept of trait dysregulation of the corticolimbic network in patients with BD 2 and with the recently formulated assumption that amygdala reactivity in patients with BD follows a statedependent course with decreased response to negative affective stimuli in depression. 9 It has been shown repeatedly that depression in individuals with BD type I 21 and in those with BD type II 22 is characterized by amygdala hypoactivation during negative affect face processing. Similarly, induced sad mood in our experiment attenuated the amygdala response in patients with fully remitted BD and mirrored bipolar depression. Therefore, blunted amygdala activation may represent a state marker of a substantial change of amygdala reactivity in both bipolar depression and experimentally induced sadness. This finding could also clarify the inconsistencies among previous fmri studies focused on amygdala response to facial emotion activation. 8 To elucidate the network regulation responsible for the opposite reactivity of the amygdala in patients with BD, we ana lyzed the functional connectivity of the amygdala. We identified a significant group condition interaction in the vlpfc (IFG), which is considered essential for top down emotional regulation. 6,9 Interestingly, a meta-analysis of 5 primary fmri studies has documented that patients with BD, regardless of current mood state, exhibit underactivation in the right IFG during both emotional and cognitive tasks. 8 The role of the IFG in BD is also supported by morphometric studies that replicated reduced right IFG volumes in patients with BD ,32,38 The vlpfc connections to the amygdala are critical for regulation of amygdala activation, and decoupling of these regions has been demonstrated in relation to emotion dysregulation. 28 The recent model assumes that BD arises from disruption in early brain development leading to decreased functional and/or structural connectivity between the vlpfc and amygdala. 5,28 However, the functional connectivity between both regions depends also on the valence of emotional stimuli. 29,3 We showed that coupling of the amygdala and IFG in patients with BD was differently influenced by both emotional valence of facial stimuli and mood. In healthy controls, sad mood induction reduced the functional connectivity between the amygdala and IFG (bilaterally) during exposure to sad faces. The opposite was detected in patients with BD, in whom the sad mood challenge increased connectivity during the period of sad face processing with a subsequent blunted amygdala response. We need to stress, however, that our data alone cannot resolve the direction of altered functional connectivity; for instance, the decreased coupling of vlpfc and the amygdala in controls during the sad mood challenge could be responsible for the exaggerated amygdala response to sad stimuli. Taken together, our data are congruent with the assumption that the amygdala vlpfc connectivity in patients with BD is state-dependent. During periods of sad face processing, patients with BD had lower connectivity after the neutral script but increased connectivity during sad mood induction (Fig. 3). Our observation that sad mood induction in patients with BD increases amygdala vlpfc connectivity in response to sad stimuli is in line with the finding that depressed but not euthymic patients with BD have greater amygdala vlpfc connectivity than controls, specifically during exposure to sad faces. 29 Several studies of BD have documented increased connectivity in a resting state. 2,31 However, the fact that amygdala vlpfc Table 2: Repeated-measures ANOVA results of amygdala activation (parameter estimates) at the level of the whole amygdala, the local maxima of the effect, and the effect of mood challenge on amygdala IFG functional connectivity identified at the whole brain level. Activation, source of variation Statistic p value Whole amygdala Between-subjects Group F 1,38 =.3.87 Within subjects Laterality F 1,38 = Condition F 1,38 =.9.76 Laterality group F 1,38 = 5.62 Laterality condition F 1,38 =.7 Condition group F 1,38 = Laterality condition group F 1,38 = 8.6 Local maxima of amygdala activation Between-subjects Group F 1,38 =.1.9 Within-subjects Laterality F 1,38 =..95 Condition F 1,38 = Laterality group F 1,38 = Laterality condition F 1,38 =.1.94 Condition group F 1,38 = 14.4 <.1 Laterality condition group F 1,38 =.4.85 Functional connectivity between amygdala and IFG (vlpfc) Between-subjects Group F 1,114 =.47.5 Within-subjects Laterality F 1,114 = Condition F 3,114 = Laterality group F 1,114 = Laterality condition F 3,114 = Condition group F 3,114 = 7.41 <.1 Laterality condition group F 3,114 = ANOVA = analysis of variance; IFG = inferior frontal gyrus; vlpfc = ventrolateral prefrontal cortex. J Psychiatry Neurosci 215;4(2) 139
7 Horacek et al. 5 ß (correlation coefficient).1 Normal Normal Sad Sad Normal Normal Sad Sad HC BD x,y,z k (vx) p (FWE) -38, 22, , 28, ß (correlation coefficient).1 Normal Normal Sad Sad Normal Normal Sad Sad HC BD Fig. 3: Effect of mood challenge on functional connectivity with the amygdala as a seed during emotional face processing. Middle panel: The whole brain F test results with significant regions of interaction (group emotional challenge laterality emotional faces) in left and right inferior frontal gyrus (IFG; Brodmann area 47). The graphs show the Z scores of functional connectivity between left (upper panel) and right (lower panel) amygdala and corresponding IFG clusters during exposure to neutral and sad faces. Coordinates of the regions of interaction are in Montreal Neurological Institute space. BD = bipolar disorder; HC = healthy controls; k (vx) = number of voxels. p < for within-group post hoc tests, p < for between-groups. p values are family-wise error (FWE) corrected at the cluster level. Error bars represent standard errors of the mean. 14 J Psychiatry Neurosci 215;4(2)
8 Mood induction and amygdala reactivity in patients with bipolar disorder coupling in our sample depended on mood induction does not support the explanation that the increased connectivity in patients with BD results from the sensitization of these connections. 2 If hyperconnectivity was a result of simple sensitization of anatomic connections, it would be detectable either in all conditions tested or specifically during exposure to sad emotional faces during both mood inductions. Considering the moodaffective stimuli interaction in our sample, it is also unlikely that aberrant white matter tracts from the vlpfc are responsible for amygdala dysregulation in patients with BD. Together our findings point to the vlpfc (specifically IFG) as the primary region of dysfunctional contextual affective processing in patients with BD leading to aberrant amygdala response. The crucial role of the vlpfc in these patients is supported by voxel-based morphometry studies that revealed the greatest effect size of grey matter reduction in the right IFG at the whole brain level. 27 The accelerated volume reduction of the right IFG correlating with disease burden 27,32 indicates that the IFG could be a candidate region for sensitization by mood episodes. We can speculate that greater amygdala vlpfc (BA 47) coupling in patients with BD within the periods of sad stimu li presented during a sad mood might be associated with abnormal overappraisal of such stimuli at the level of the vlpfc with subsequent blunted amygdala response in this condition (Fig. 2). An alternative explanation is that patients with BD exposed to a sad mood challenge process sad and neutral stimu li in a similar way and that the vlpfc amygdala system does not discriminate the emotional valence of stimuli. In other words, patients with BD lack the typical mood-congruent processing bias where sad mood facilitates the response to sad stimuli. In this regard, BD appears different from MDD, in which the mood-congruent processing bias in the amygdala (depression amplifies response to sad faces) has been consistently replicated Future research should clarify if the effect of mood on emotional stimuli activation observed in our sample is specific to sad faces and not to other negative facial emotions (e.g., anger, fear). This specificity for sad faces (v. fearful faces) in mood-congruent processing bias was identified in patients with MDD. 36 The differences between patients with BD and healthy controls in vlpfc amygdala coupling could be mediated also by a third region: the anterior cingulate (ACC). The ACC has been implicated in mood disorders, and it has been documented that this region could mediate the hyperconnectivity between the vlpfc and amygdala in patients with BD type I. 2 However, our amygdala connectivity analysis revealed only the IFG as significant. In a previous study, the mediatory role of the ACC in amygdala vlpfc coupling was found in resting fmri, 2 whereas in our study we used an affective activation fmri paradigm. Further investigation is necessary to elucidate the role of the ACC (and other regions, such as the hippocampus) in the regulation of the vlpfc amygdala complex. We did not find any substantial amygdala lateralization effect in our sample in terms of both activation and vlpfc connectivity, and the patterns of mood challenge emotional stimuli interaction were similar on both sides. The lack of lateralized effect observed in our study could be related to the applied combination of sad mood challenge, which has exerted effect almost exclusively in the right hemisphere, 1 with the activation by sad faces (v. other negative faces), which is lateralized to the left amygdala. 15 Limitations There are several limitations to the present study. First, all patients with BD were stabilized on long-term medication. However, a recent systematic review documented that the impact of medications on imaging data in patients with BD is limited and that this impact, if any, is normalizing (i.e., medicated patients with BD are more similar to unmedicated controls, with a lower probability of type-i error). 37 Second, our study maps only 1 emotional pole of BD. We did not challenge the manic reactivity because happy mood induction is more problematic, has not been validated for mood disorders and might be difficult to achieve along with sad mood provocation during a single scanning session. The baseline neutral script was always presented before the sad mood challenge, and we cannot rule out the effect of order and carry-over effect. This is a tradeoff for being able to study the same participants in 2 different mood states during a single scanning session. However, the design was the same for all participants, and the very pronounced subjective emotional differences between both conditions were confirmed by postscan debriefing. We did not control for IQ, but its effect on our findings is unlikely because the groups did not differ in education level. Finally, we used an AAL atlas to define ROI for the amygdala and, despite the visual control, certain errors might occur. However, both local maxima of group condition interactions within these ROIs were signifi cant, even in the whole brain SPM analysis (p < ). On the other hand, the less clearly anatomically bordered vlpfc regions were functionally defined strictly as clusters of significant group condition interactions of functional connectivity with the amygdala at the whole brain level. Conclusion Our results of inverse amygdala reactivity to the mood challenge are in accordance with those of prior studies of bipolar depression. That the sad mood challenge attenuates amygdala response to sad stimuli in euthymic patients with BD suggests that a mood challenge can unmask the vulnerability to abnormal mood regulation in these patients and that they differ from patients with remitted MDD, which is characterized by mood-congruent processing bias. The changes in amygdala reactivity are paralleled by differences between patients with BD and healthy controls in the amygdala vlpfc connectivity. The amygdala vlpfc hyperconnectivity in response to negative (sad) stimuli during the mood challenge is congruent with previous findings in studies of bipolar depression and may be responsible for the blunted amygdala response to sad stimuli. Acknowledgments: J. Horacek is supported by a 29 NARSAD Independent Investigator Award and grants from the Ministry of Health of the Czech Republic (DRO PCP, and IGA NT1224). The authors thank H. Fridrichova, N. Gornerova, M. Holec and I. Ibrahim for technical assistance and participant recruitment, and J. Garnham and C. Hampson for language editing. J Psychiatry Neurosci 215;4(2) 141
9 Horacek et al. Affiliations: Prague Psychiatric Centre, Prague, Czech Republic (Horacek, Mikolas, Novak, Palenicek, Brunovsky, Höschl); Third Faculty of Medicine, Charles University, Prague, Czech Republic (Horacek, Novak, Palenicek, Brunovsky, Höschl, Alda); Department of Psychiatry, Dalhousie University, Halifax, Canada (Horacek, Alda); Institute for Clinical and Experimental Medicine, Prague, Czech Republic (Tintera). Competing interests: J. Horacek has received a speaking honorarium from Janssen Cilag, Zentiva group, Eli Lilly, Lundbeck, Novartis, Pfizer and MEDA Pharma. He has also received grant support from NARSAD and the Ministry of Health, Czech Republic. C. Höschl has served as a study coordinator with Servier, a faculty member of the Lundbeck International Neuroscience Foundation and as a speaker for Eli Lilly, Janssen Cilag and Krka. M. Brunovsky has served as an adviser and speaker for Hoffmann-La Roche, Krka, MEDA Pharma, Novartis and Zentiva group. M. Alda has received grants from Canad ian Institutes of Health Research, Genome Quebec, Nova Scotia Health Research Foundation, Stanley Foundation, NARSAD, and Killam Trust. No other competing interests declared. Contributors: J. Horacek and M. Alda designed the study. J. Horacek, J. Tintera, T. Palenicek and M. Brunovsky acquired the data, which J. Horacek, M. Alda, C. Höschl, T. Novak and P. Mikolas analyzed. J. Horacek and M. Alda wrote the article, which all authors reviewed and approved for publication. References 1. Judd LL, Akiskal HS, Schettler PJ, et al. The long-term natural history of the weekly symptomatic status of bipolar I disorder. Arch Gen Psychiatry 22;59: Fukuda K, Etoh T, Iwadate T, et al. The course and prognosis of manic-depressive psychosis: a quantitative analysis of episodes and intervals. Tohoku J Exp Med 1983;139: Keller MB. The course of manic-depressive illness. J Clin Psychiatry 1988;49(suppl): Townsend J, Altshuler LL. Emotion processing and regulation in bipolar disorder: a review. Bipolar Disord 212;14: Strakowski SM, Adler CM, Almeida J, et al. The functional neuroanatomy of bipolar disorder: a consensus model. Bipolar Disord 212;14: Foland LC, Altshuler LL, Bookheimer SY, et al. Evidence for deficient modulation of amygdala response by prefrontal cortex in bipolar mania. Psychiatry Res 28;162: Price JL, Drevets WC. Neural circuits underlying the pathophysiology of mood disorders. Trends Cogn Sci 212;16: Chen CH, Suckling J, Lennox BR, et al. A quantitative meta-analysis of fmri studies in bipolar disorder. Bipolar Disord 211;13: Hariri AR. The highs and lows of amygdala reactivity in bipolar disorders. Am J Psychiatry 212;169: Krüger S, Alda M, Young LT, et al. Risk and resilience markers in bipolar disorder: brain responses to emotional challenge in bipolar patients and their healthy siblings. Am J Psychiatry 26;163: Liotti M, Mayberg HS, McGinnis S, et al. Unmasking disease- specific cerebral blood flow abnormalities: mood challenge in patients with remitted unipolar depression. Am J Psychiatry 22;159: Young RC, Biggs JT, Ziegler VE, et al. A rating scale for mania: reliability, validity and sensitivity. Br J Psychiatry 1978;133: Montgomery SA, Asberg M. A new depression scale designed to be sensitive to change. Br J Psychiatry 1979;134: Beck AT, Ward CH, Mendelson M, et al. An inventory for measuring depression. Arch Gen Psychiatry 1961;4: Fusar-Poli P, Placentino A, Carletti F, et al. Functional atlas of emotional faces processing: a voxel-based meta-analysis of 15 functional magnetic resonance imaging studies. J Psychiatry Neurosci 29;34: Erwin RJ, Gur RC, Gur RE, et al. Facial emotion discrimination: I. Task construction and behavioral findings in normal subjects. Psychiatry Res 1992;42: Tzourio-Mazoyer N, Landeau B, Papathanassiou D, et al. Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. Neuroimage 22;15: Hariri AR, Mattay VS, Tessitore A, et al. Serotonin transporter genetic variation and the response of the human amygdala. Science 22;297: Phillips ML, Ladouceur CD, Drevets WC. A neural model of voluntary and automatic emotion regulation: implications for understanding the pathophysiology and neurodevelopment of bipolar disorder. Mol Psychiatry 28;13: Torrisi S, Moody TD, Vizueta N, et al. Differences in resting corticolimbic functional connectivity in bipolar I euthymia. Bipolar Disord 213;15: Altshuler L, Bookheimer S, Townsend J, et al. Regional brain changes in bipolar I depression: a functional magnetic resonance imaging study. Bipolar Disord 28;1: Vizueta N, Rudie JD, Townsend JD, et al. Regional fmri hypoactivation and altered functional connectivity during emotion processing in nonmedicated depressed patients with bipolar II disorder. Am J Psychiatry 212;169: Haldane M, Cunningham G, Androutsos C, et al. Structural brain correlates of response inhibition in Bipolar Disorder I. J Psychopharmacol 28;22: López-Larson MP, DelBello MP, Zimmerman ME, et al. Regional prefrontal gray and white matter abnormalities in bipolar disorder. Biol Psychiatry 22;52: Matsuo K, Kopecek M, Nicoletti MA, et al. New structural brain imaging endophenotype in bipolar disorder. Mol Psychiatry 212;17: Stanfield AC, Moorhead TW, Job DE, et al. Structural abnormalities of ventrolateral and orbitofrontal cortex in patients with familial bipolar disorder. Bipolar Disord 29;11: Bora E, Fornito A, Yucel M, et al. Voxelwise meta-analysis of gray matter abnormalities in bipolar disorder. Biol Psychiatry 21;67: Phillips ML, Drevets WC, Rauch SL, et al. Neurobiology of emotion perception II: implications for major psychiatric disorders. Biol Psychiatry 23;54: Versace A, Thompson WK, Zhou D, et al. Abnormal left and right amygdala-orbitofrontal cortical functional connectivity to emotional faces: state versus trait vulnerability markers of depression in bipolar disorder. Biol Psychiatry 21;67: Banks SJ, Eddy KT, Angstadt M, et al. Amygdala-frontal connectivity during emotion regulation. Soc Cogn Affect Neurosci 27;2: Chepenik LG, Raffo M, Hampson M, et al. Functional connectivity between ventral prefrontal cortex and amygdala at low frequency in the resting state in bipolar disorder. Psychiatry Res 21;182: Hajek T, Cullis J, Novak T, et al. Brain structural signature of familial predisposition for bipolar disorder: replicable evidence for involvement of the right inferior frontal gyrus. Biol Psychiatry 213;73: Suslow T, Konrad C, Kugel H, et al. Automatic mood-congruent amygdala responses to masked facial expressions in major depression. Biol Psychiatry 21;67: Victor TA, Furey ML, Fromm SJ, et al. Relationship between amygdala responses to masked faces and mood state and treatment in major depressive disorder. Arch Gen Psychiatry 21;67: Victor TA, Furey ML, Fromm SJ, et al. The extended functional neuroanatomy of emotional processing biases for masked faces in major depressive disorder. PLoS ONE 212;7:e Arnone D, McKie S, Elliott R, et al. Increased amygdala responses to sad but not fearful faces in major depression: relation to mood state and pharmacological treatment. Am J Psychiatry 212;169: Hafeman DM, Chang KD, Garrett AS, et al. Effects of medication on neuroimaging findings in bipolar disorder: an updated review. Bipolar Disord 212;14: Selvaraj S, Arnone D, Job D, et al. Grey matter differences in bipolar disorder: a meta-analysis of voxel-based morphometry studies. Bipolar Disord 212;14: J Psychiatry Neurosci 215;4(2)
SUPPLEMENT: DYNAMIC FUNCTIONAL CONNECTIVITY IN DEPRESSION. Supplemental Information. Dynamic Resting-State Functional Connectivity in Major Depression
 Supplemental Information Dynamic Resting-State Functional Connectivity in Major Depression Roselinde H. Kaiser, Ph.D., Susan Whitfield-Gabrieli, Ph.D., Daniel G. Dillon, Ph.D., Franziska Goer, B.S., Miranda
Supplemental Information Dynamic Resting-State Functional Connectivity in Major Depression Roselinde H. Kaiser, Ph.D., Susan Whitfield-Gabrieli, Ph.D., Daniel G. Dillon, Ph.D., Franziska Goer, B.S., Miranda
Supplementary Information Methods Subjects The study was comprised of 84 chronic pain patients with either chronic back pain (CBP) or osteoarthritis
 Supplementary Information Methods Subjects The study was comprised of 84 chronic pain patients with either chronic back pain (CBP) or osteoarthritis (OA). All subjects provided informed consent to procedures
Supplementary Information Methods Subjects The study was comprised of 84 chronic pain patients with either chronic back pain (CBP) or osteoarthritis (OA). All subjects provided informed consent to procedures
WHAT DOES THE BRAIN TELL US ABOUT TRUST AND DISTRUST? EVIDENCE FROM A FUNCTIONAL NEUROIMAGING STUDY 1
 SPECIAL ISSUE WHAT DOES THE BRAIN TE US ABOUT AND DIS? EVIDENCE FROM A FUNCTIONAL NEUROIMAGING STUDY 1 By: Angelika Dimoka Fox School of Business Temple University 1801 Liacouras Walk Philadelphia, PA
SPECIAL ISSUE WHAT DOES THE BRAIN TE US ABOUT AND DIS? EVIDENCE FROM A FUNCTIONAL NEUROIMAGING STUDY 1 By: Angelika Dimoka Fox School of Business Temple University 1801 Liacouras Walk Philadelphia, PA
Supplementary Online Content
 Supplementary Online Content Green SA, Hernandez L, Tottenham N, Krasileva K, Bookheimer SY, Dapretto M. The neurobiology of sensory overresponsivity in youth with autism spectrum disorders. Published
Supplementary Online Content Green SA, Hernandez L, Tottenham N, Krasileva K, Bookheimer SY, Dapretto M. The neurobiology of sensory overresponsivity in youth with autism spectrum disorders. Published
Investigations in Resting State Connectivity. Overview
 Investigations in Resting State Connectivity Scott FMRI Laboratory Overview Introduction Functional connectivity explorations Dynamic change (motor fatigue) Neurological change (Asperger s Disorder, depression)
Investigations in Resting State Connectivity Scott FMRI Laboratory Overview Introduction Functional connectivity explorations Dynamic change (motor fatigue) Neurological change (Asperger s Disorder, depression)
smokers) aged 37.3 ± 7.4 yrs (mean ± sd) and a group of twelve, age matched, healthy
 Methods Participants We examined a group of twelve male pathological gamblers (ten strictly right handed, all smokers) aged 37.3 ± 7.4 yrs (mean ± sd) and a group of twelve, age matched, healthy males,
Methods Participants We examined a group of twelve male pathological gamblers (ten strictly right handed, all smokers) aged 37.3 ± 7.4 yrs (mean ± sd) and a group of twelve, age matched, healthy males,
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/324/5927/646/dc1 Supporting Online Material for Self-Control in Decision-Making Involves Modulation of the vmpfc Valuation System Todd A. Hare,* Colin F. Camerer, Antonio
www.sciencemag.org/cgi/content/full/324/5927/646/dc1 Supporting Online Material for Self-Control in Decision-Making Involves Modulation of the vmpfc Valuation System Todd A. Hare,* Colin F. Camerer, Antonio
Twelve right-handed subjects between the ages of 22 and 30 were recruited from the
 Supplementary Methods Materials & Methods Subjects Twelve right-handed subjects between the ages of 22 and 30 were recruited from the Dartmouth community. All subjects were native speakers of English,
Supplementary Methods Materials & Methods Subjects Twelve right-handed subjects between the ages of 22 and 30 were recruited from the Dartmouth community. All subjects were native speakers of English,
Supplementary Online Content
 1 Supplementary Online Content Miller CH, Hamilton JP, Sacchet MD, Gotlib IH. Meta-analysis of functional neuroimaging of major depressive disorder in youth. JAMA Psychiatry. Published online September
1 Supplementary Online Content Miller CH, Hamilton JP, Sacchet MD, Gotlib IH. Meta-analysis of functional neuroimaging of major depressive disorder in youth. JAMA Psychiatry. Published online September
Supplementary Online Content
 Supplementary Online Content Redlich R, Opel N, Grotegerd D, et al. Prediction of individual response to electroconvulsive therapy via machine learning on structural magnetic resonance imaging data. JAMA
Supplementary Online Content Redlich R, Opel N, Grotegerd D, et al. Prediction of individual response to electroconvulsive therapy via machine learning on structural magnetic resonance imaging data. JAMA
Differential Patterns of Abnormal Activity and Connectivity in the Amygdala Prefrontal Circuitry in Bipolar-I and Bipolar-NOS Youth
 NEW RESEARCH Differential Patterns of Abnormal Activity and Connectivity in the Amygdala Prefrontal Circuitry in Bipolar-I and Bipolar-NOS Youth Cecile D. adouceur, Ph.D., Tiffany Farchione, M.D., Vaibhav
NEW RESEARCH Differential Patterns of Abnormal Activity and Connectivity in the Amygdala Prefrontal Circuitry in Bipolar-I and Bipolar-NOS Youth Cecile D. adouceur, Ph.D., Tiffany Farchione, M.D., Vaibhav
The Neural Correlates of Moral Decision-Making in Psychopathy
 University of Pennsylvania ScholarlyCommons Neuroethics Publications Center for Neuroscience & Society 1-1-2009 The Neural Correlates of Moral Decision-Making in Psychopathy Andrea L. Glenn University
University of Pennsylvania ScholarlyCommons Neuroethics Publications Center for Neuroscience & Society 1-1-2009 The Neural Correlates of Moral Decision-Making in Psychopathy Andrea L. Glenn University
Supplemental Data. Inclusion/exclusion criteria for major depressive disorder group and healthy control group
 1 Supplemental Data Inclusion/exclusion criteria for major depressive disorder group and healthy control group Additional inclusion criteria for the major depressive disorder group were: age of onset of
1 Supplemental Data Inclusion/exclusion criteria for major depressive disorder group and healthy control group Additional inclusion criteria for the major depressive disorder group were: age of onset of
Classification and Statistical Analysis of Auditory FMRI Data Using Linear Discriminative Analysis and Quadratic Discriminative Analysis
 International Journal of Innovative Research in Computer Science & Technology (IJIRCST) ISSN: 2347-5552, Volume-2, Issue-6, November-2014 Classification and Statistical Analysis of Auditory FMRI Data Using
International Journal of Innovative Research in Computer Science & Technology (IJIRCST) ISSN: 2347-5552, Volume-2, Issue-6, November-2014 Classification and Statistical Analysis of Auditory FMRI Data Using
A possible mechanism for impaired joint attention in autism
 A possible mechanism for impaired joint attention in autism Justin H G Williams Morven McWhirr Gordon D Waiter Cambridge Sept 10 th 2010 Joint attention in autism Declarative and receptive aspects initiating
A possible mechanism for impaired joint attention in autism Justin H G Williams Morven McWhirr Gordon D Waiter Cambridge Sept 10 th 2010 Joint attention in autism Declarative and receptive aspects initiating
Neural activity to positive expressions predicts daily experience of schizophrenia-spectrum symptoms in adults with high social anhedonia
 1 Neural activity to positive expressions predicts daily experience of schizophrenia-spectrum symptoms in adults with high social anhedonia Christine I. Hooker, Taylor L. Benson, Anett Gyurak, Hong Yin,
1 Neural activity to positive expressions predicts daily experience of schizophrenia-spectrum symptoms in adults with high social anhedonia Christine I. Hooker, Taylor L. Benson, Anett Gyurak, Hong Yin,
Functional MRI Mapping Cognition
 Outline Functional MRI Mapping Cognition Michael A. Yassa, B.A. Division of Psychiatric Neuro-imaging Psychiatry and Behavioral Sciences Johns Hopkins School of Medicine Why fmri? fmri - How it works Research
Outline Functional MRI Mapping Cognition Michael A. Yassa, B.A. Division of Psychiatric Neuro-imaging Psychiatry and Behavioral Sciences Johns Hopkins School of Medicine Why fmri? fmri - How it works Research
Procedia - Social and Behavioral Sciences 159 ( 2014 ) WCPCG 2014
 Available online at www.sciencedirect.com ScienceDirect Procedia - Social and Behavioral Sciences 159 ( 2014 ) 743 748 WCPCG 2014 Differences in Visuospatial Cognition Performance and Regional Brain Activation
Available online at www.sciencedirect.com ScienceDirect Procedia - Social and Behavioral Sciences 159 ( 2014 ) 743 748 WCPCG 2014 Differences in Visuospatial Cognition Performance and Regional Brain Activation
Personal Space Regulation by the Human Amygdala. California Institute of Technology
 Personal Space Regulation by the Human Amygdala Daniel P. Kennedy 1, Jan Gläscher 1, J. Michael Tyszka 2 & Ralph Adolphs 1,2 1 Division of Humanities and Social Sciences 2 Division of Biology California
Personal Space Regulation by the Human Amygdala Daniel P. Kennedy 1, Jan Gläscher 1, J. Michael Tyszka 2 & Ralph Adolphs 1,2 1 Division of Humanities and Social Sciences 2 Division of Biology California
Supplementary materials. Appendix A;
 Supplementary materials Appendix A; To determine ADHD diagnoses, a combination of Conners' ADHD questionnaires and a semi-structured diagnostic interview was used(1-4). Each participant was assessed with
Supplementary materials Appendix A; To determine ADHD diagnoses, a combination of Conners' ADHD questionnaires and a semi-structured diagnostic interview was used(1-4). Each participant was assessed with
Identification of Neuroimaging Biomarkers
 Identification of Neuroimaging Biomarkers Dan Goodwin, Tom Bleymaier, Shipra Bhal Advisor: Dr. Amit Etkin M.D./PhD, Stanford Psychiatry Department Abstract We present a supervised learning approach to
Identification of Neuroimaging Biomarkers Dan Goodwin, Tom Bleymaier, Shipra Bhal Advisor: Dr. Amit Etkin M.D./PhD, Stanford Psychiatry Department Abstract We present a supervised learning approach to
Methods to examine brain activity associated with emotional states and traits
 Methods to examine brain activity associated with emotional states and traits Brain electrical activity methods description and explanation of method state effects trait effects Positron emission tomography
Methods to examine brain activity associated with emotional states and traits Brain electrical activity methods description and explanation of method state effects trait effects Positron emission tomography
Supplementary Information
 Supplementary Information The neural correlates of subjective value during intertemporal choice Joseph W. Kable and Paul W. Glimcher a 10 0 b 10 0 10 1 10 1 Discount rate k 10 2 Discount rate k 10 2 10
Supplementary Information The neural correlates of subjective value during intertemporal choice Joseph W. Kable and Paul W. Glimcher a 10 0 b 10 0 10 1 10 1 Discount rate k 10 2 Discount rate k 10 2 10
The Impact of Anxiety-Inducing Distraction on Cognitive Performance: A Combined Brain Imaging and Personality Investigation
 The Impact of Anxiety-Inducing Distraction on Cognitive Performance: A Combined Brain Imaging and Personality Investigation Ekaterina Denkova 1, Gloria Wong 2, Sanda Dolcos 3, Keen Sung 1, Lihong Wang
The Impact of Anxiety-Inducing Distraction on Cognitive Performance: A Combined Brain Imaging and Personality Investigation Ekaterina Denkova 1, Gloria Wong 2, Sanda Dolcos 3, Keen Sung 1, Lihong Wang
Supplementary Materials for
 Supplementary Materials for Folk Explanations of Behavior: A Specialized Use of a Domain-General Mechanism Robert P. Spunt & Ralph Adolphs California Institute of Technology Correspondence may be addressed
Supplementary Materials for Folk Explanations of Behavior: A Specialized Use of a Domain-General Mechanism Robert P. Spunt & Ralph Adolphs California Institute of Technology Correspondence may be addressed
Systematic review of the neural basis of social cognition in patients with mood disorders
 Review Paper Systematic review of the neural basis of social cognition in patients with mood disorders Andrée M. Cusi, BSc; Anthony Nazarov, BSc; Katherine Holshausen, BSc; Glenda M. MacQueen, MD, PhD;
Review Paper Systematic review of the neural basis of social cognition in patients with mood disorders Andrée M. Cusi, BSc; Anthony Nazarov, BSc; Katherine Holshausen, BSc; Glenda M. MacQueen, MD, PhD;
Supporting online material. Materials and Methods. We scanned participants in two groups of 12 each. Group 1 was composed largely of
 Placebo effects in fmri Supporting online material 1 Supporting online material Materials and Methods Study 1 Procedure and behavioral data We scanned participants in two groups of 12 each. Group 1 was
Placebo effects in fmri Supporting online material 1 Supporting online material Materials and Methods Study 1 Procedure and behavioral data We scanned participants in two groups of 12 each. Group 1 was
Comparing event-related and epoch analysis in blocked design fmri
 Available online at www.sciencedirect.com R NeuroImage 18 (2003) 806 810 www.elsevier.com/locate/ynimg Technical Note Comparing event-related and epoch analysis in blocked design fmri Andrea Mechelli,
Available online at www.sciencedirect.com R NeuroImage 18 (2003) 806 810 www.elsevier.com/locate/ynimg Technical Note Comparing event-related and epoch analysis in blocked design fmri Andrea Mechelli,
Resistance to forgetting associated with hippocampus-mediated. reactivation during new learning
 Resistance to Forgetting 1 Resistance to forgetting associated with hippocampus-mediated reactivation during new learning Brice A. Kuhl, Arpeet T. Shah, Sarah DuBrow, & Anthony D. Wagner Resistance to
Resistance to Forgetting 1 Resistance to forgetting associated with hippocampus-mediated reactivation during new learning Brice A. Kuhl, Arpeet T. Shah, Sarah DuBrow, & Anthony D. Wagner Resistance to
Functional Magnetic Resonance Imaging with Arterial Spin Labeling: Techniques and Potential Clinical and Research Applications
 pissn 2384-1095 eissn 2384-1109 imri 2017;21:91-96 https://doi.org/10.13104/imri.2017.21.2.91 Functional Magnetic Resonance Imaging with Arterial Spin Labeling: Techniques and Potential Clinical and Research
pissn 2384-1095 eissn 2384-1109 imri 2017;21:91-96 https://doi.org/10.13104/imri.2017.21.2.91 Functional Magnetic Resonance Imaging with Arterial Spin Labeling: Techniques and Potential Clinical and Research
Citation for published version (APA): Mocking, R. J. T. (2017). Fatty acids in context: Neurometabolic perspectives on depression vulnerability.
 UvA-DARE (Digital Academic Repository) Fatty acids in context Mocking, R.J.T. Link to publication Citation for published version (APA): Mocking, R. J. T. (2017). Fatty acids in context: Neurometabolic
UvA-DARE (Digital Academic Repository) Fatty acids in context Mocking, R.J.T. Link to publication Citation for published version (APA): Mocking, R. J. T. (2017). Fatty acids in context: Neurometabolic
Brain Imaging Investigation of the Impairing Effect of Emotion on Cognition
 Brain Imaging Investigation of the Impairing Effect of Emotion on Cognition Gloria Wong 1,2, Sanda Dolcos 1,3, Ekaterina Denkova 1, Rajendra A. Morey 4,5, Lihong Wang 4,5, Nicholas Coupland 1, Gregory
Brain Imaging Investigation of the Impairing Effect of Emotion on Cognition Gloria Wong 1,2, Sanda Dolcos 1,3, Ekaterina Denkova 1, Rajendra A. Morey 4,5, Lihong Wang 4,5, Nicholas Coupland 1, Gregory
HST.583 Functional Magnetic Resonance Imaging: Data Acquisition and Analysis Fall 2008
 MIT OpenCourseWare http://ocw.mit.edu HST.583 Functional Magnetic Resonance Imaging: Data Acquisition and Analysis Fall 2008 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms.
MIT OpenCourseWare http://ocw.mit.edu HST.583 Functional Magnetic Resonance Imaging: Data Acquisition and Analysis Fall 2008 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms.
Supplementary information Detailed Materials and Methods
 Supplementary information Detailed Materials and Methods Subjects The experiment included twelve subjects: ten sighted subjects and two blind. Five of the ten sighted subjects were expert users of a visual-to-auditory
Supplementary information Detailed Materials and Methods Subjects The experiment included twelve subjects: ten sighted subjects and two blind. Five of the ten sighted subjects were expert users of a visual-to-auditory
Cover Page. The handle holds various files of this Leiden University dissertation
 Cover Page The handle http://hdl.handle.net/1887/32078 holds various files of this Leiden University dissertation Author: Pannekoek, Nienke Title: Using novel imaging approaches in affective disorders
Cover Page The handle http://hdl.handle.net/1887/32078 holds various files of this Leiden University dissertation Author: Pannekoek, Nienke Title: Using novel imaging approaches in affective disorders
SUPPLEMENTARY METHODS. Subjects and Confederates. We investigated a total of 32 healthy adult volunteers, 16
 SUPPLEMENTARY METHODS Subjects and Confederates. We investigated a total of 32 healthy adult volunteers, 16 women and 16 men. One female had to be excluded from brain data analyses because of strong movement
SUPPLEMENTARY METHODS Subjects and Confederates. We investigated a total of 32 healthy adult volunteers, 16 women and 16 men. One female had to be excluded from brain data analyses because of strong movement
QUANTIFYING CEREBRAL CONTRIBUTIONS TO PAIN 1
 QUANTIFYING CEREBRAL CONTRIBUTIONS TO PAIN 1 Supplementary Figure 1. Overview of the SIIPS1 development. The development of the SIIPS1 consisted of individual- and group-level analysis steps. 1) Individual-person
QUANTIFYING CEREBRAL CONTRIBUTIONS TO PAIN 1 Supplementary Figure 1. Overview of the SIIPS1 development. The development of the SIIPS1 consisted of individual- and group-level analysis steps. 1) Individual-person
Experimental Design. Outline. Outline. A very simple experiment. Activation for movement versus rest
 Experimental Design Kate Watkins Department of Experimental Psychology University of Oxford With thanks to: Heidi Johansen-Berg Joe Devlin Outline Choices for experimental paradigm Subtraction / hierarchical
Experimental Design Kate Watkins Department of Experimental Psychology University of Oxford With thanks to: Heidi Johansen-Berg Joe Devlin Outline Choices for experimental paradigm Subtraction / hierarchical
Using structural MRI to identify individuals at genetic risk for bipolar disorders: a 2-cohort, machine learning study
 Research Paper Using structural MRI to identify individuals at genetic risk for bipolar disorders: a 2-cohort, machine learning study Tomas Hajek, MD, PhD; Christopher Cooke, MSc; Miloslav Kopecek, MD,
Research Paper Using structural MRI to identify individuals at genetic risk for bipolar disorders: a 2-cohort, machine learning study Tomas Hajek, MD, PhD; Christopher Cooke, MSc; Miloslav Kopecek, MD,
Disrupted cortico- limbic connectivity during reward processing in remitted bipolar I disorder
 Received: 6 March 2017 Accepted: 8 August 2017 DOI: 10.1111/bdi.12560 ORIGINAL ARTICLE Disrupted cortico- limbic connectivity during reward processing in remitted bipolar I disorder Sunny J Dutra 1,2 Vincent
Received: 6 March 2017 Accepted: 8 August 2017 DOI: 10.1111/bdi.12560 ORIGINAL ARTICLE Disrupted cortico- limbic connectivity during reward processing in remitted bipolar I disorder Sunny J Dutra 1,2 Vincent
Supplementary Online Content
 Supplementary Online Content Gregg NM, Kim AE, Gurol ME, et al. Incidental cerebral microbleeds and cerebral blood flow in elderly individuals. JAMA Neurol. Published online July 13, 2015. doi:10.1001/jamaneurol.2015.1359.
Supplementary Online Content Gregg NM, Kim AE, Gurol ME, et al. Incidental cerebral microbleeds and cerebral blood flow in elderly individuals. JAMA Neurol. Published online July 13, 2015. doi:10.1001/jamaneurol.2015.1359.
Supporting Information. Demonstration of effort-discounting in dlpfc
 Supporting Information Demonstration of effort-discounting in dlpfc In the fmri study on effort discounting by Botvinick, Huffstettler, and McGuire [1], described in detail in the original publication,
Supporting Information Demonstration of effort-discounting in dlpfc In the fmri study on effort discounting by Botvinick, Huffstettler, and McGuire [1], described in detail in the original publication,
Altered effective connectivity network of the thalamus in post traumatic stress disorder: a resting state FMRI study with granger causality method
 DOI 10.1186/s40535-016-0025-y RESEARCH Open Access Altered effective connectivity network of the thalamus in post traumatic stress disorder: a resting state FMRI study with granger causality method Youxue
DOI 10.1186/s40535-016-0025-y RESEARCH Open Access Altered effective connectivity network of the thalamus in post traumatic stress disorder: a resting state FMRI study with granger causality method Youxue
Amy Garrett, Ph.D., Victor Carrion, M.D., and Allan Reiss, M.D. Stanford University School of Medicine Department of Psychiatry
 Amy Garrett, Ph.D., Victor Carrion, M.D., and Allan Reiss, M.D. Stanford University School of Medicine Department of Psychiatry Center for Interdisciplinary Brain Sciences Research and Stanford Early Life
Amy Garrett, Ph.D., Victor Carrion, M.D., and Allan Reiss, M.D. Stanford University School of Medicine Department of Psychiatry Center for Interdisciplinary Brain Sciences Research and Stanford Early Life
The rewarding nature of provocationfocused rumination in women with borderline personality disorder: a preliminary fmri investigation
 Peters et al. Borderline Personality Disorder and Emotion Dysregulation (2018) 5:1 DOI 10.1186/s40479-018-0079-7 RESEARCH ARTICLE Open Access The rewarding nature of provocationfocused rumination in women
Peters et al. Borderline Personality Disorder and Emotion Dysregulation (2018) 5:1 DOI 10.1186/s40479-018-0079-7 RESEARCH ARTICLE Open Access The rewarding nature of provocationfocused rumination in women
Neuroanatomy of Emotion, Fear, and Anxiety
 Neuroanatomy of Emotion, Fear, and Anxiety Outline Neuroanatomy of emotion Critical conceptual, experimental design, and interpretation issues in neuroimaging research Fear and anxiety Neuroimaging research
Neuroanatomy of Emotion, Fear, and Anxiety Outline Neuroanatomy of emotion Critical conceptual, experimental design, and interpretation issues in neuroimaging research Fear and anxiety Neuroimaging research
Talk 2. Neurocognitive differences in children with or without CU- traits 05/12/2013. Psychological. Behavioural
 Neurocognitive differences in children with or without CU- traits Prof Essi Viding Developmental Risk and Resilience Unit, Psychology and Language Sciences, UCL e.viding@ucl.ac.uk Talk 2. Environment Psychological
Neurocognitive differences in children with or without CU- traits Prof Essi Viding Developmental Risk and Resilience Unit, Psychology and Language Sciences, UCL e.viding@ucl.ac.uk Talk 2. Environment Psychological
Final Report 2017 Authors: Affiliations: Title of Project: Background:
 Final Report 2017 Authors: Dr Gershon Spitz, Ms Abbie Taing, Professor Jennie Ponsford, Dr Matthew Mundy, Affiliations: Epworth Research Foundation and Monash University Title of Project: The return of
Final Report 2017 Authors: Dr Gershon Spitz, Ms Abbie Taing, Professor Jennie Ponsford, Dr Matthew Mundy, Affiliations: Epworth Research Foundation and Monash University Title of Project: The return of
Neuroimaging and Psychiatry
 Neuroimaging and Psychiatry Chris Gale Otago Regional Psychiatry Training Programme Feb 12, 2011 In psychosis. Decrease volume frontal, parietal, hippocampus, corpus collosum. Increase CSF volume Functional
Neuroimaging and Psychiatry Chris Gale Otago Regional Psychiatry Training Programme Feb 12, 2011 In psychosis. Decrease volume frontal, parietal, hippocampus, corpus collosum. Increase CSF volume Functional
Behavioural Brain Research
 Behavioural Brain Research 197 (2009) 186 197 Contents lists available at ScienceDirect Behavioural Brain Research j o u r n a l h o m e p a g e : www.elsevier.com/locate/bbr Research report Top-down attentional
Behavioural Brain Research 197 (2009) 186 197 Contents lists available at ScienceDirect Behavioural Brain Research j o u r n a l h o m e p a g e : www.elsevier.com/locate/bbr Research report Top-down attentional
Neuroimaging in Clinical Practice
 Neuroimaging in Clinical Practice John Gabrieli Department of Brain and Cognitive Sciences & Martinos Imaging Center at the McGovern Institute for Brain Research, MIT Disclosures Neither I nor my spouse/partner
Neuroimaging in Clinical Practice John Gabrieli Department of Brain and Cognitive Sciences & Martinos Imaging Center at the McGovern Institute for Brain Research, MIT Disclosures Neither I nor my spouse/partner
JPBS. Unipolar Depression and Bipolar Depression Manifest Different Brain Abnormalities: A Voxel-based Morphometry Study ABSTRACT INTRODUCTION
 Unipolar Depression and Bipolar Depression Manifest Different Brain Abnormalities: A Voxel-based Morphometry Study Aimin Hu 1,2, Zhimin Xue 1, Zhening Liu 1, Peng Wang 3, Zhong He 4 1 Mental Health Institute
Unipolar Depression and Bipolar Depression Manifest Different Brain Abnormalities: A Voxel-based Morphometry Study Aimin Hu 1,2, Zhimin Xue 1, Zhening Liu 1, Peng Wang 3, Zhong He 4 1 Mental Health Institute
Chronic irritability in youth that may be misdiagnosed as bipolar disorder. Ellen Leibenluft, M.D.
 Chronic irritability in youth that may be misdiagnosed as bipolar disorder Ellen Leibenluft, M.D. Chief, Section on Bipolar Spectrum Disorders Emotion and Development Branch National Institute of Mental
Chronic irritability in youth that may be misdiagnosed as bipolar disorder Ellen Leibenluft, M.D. Chief, Section on Bipolar Spectrum Disorders Emotion and Development Branch National Institute of Mental
Reporting Checklist for Nature Neuroscience
 Corresponding Author: Manuscript Number: NNA49200 Manuscript Type: Matthew Walker & Bryce Mander Article Reporting Checklist for Nature Neuroscience # Main Figures: 4 # Supplementary Figures: 3 # Supplementary
Corresponding Author: Manuscript Number: NNA49200 Manuscript Type: Matthew Walker & Bryce Mander Article Reporting Checklist for Nature Neuroscience # Main Figures: 4 # Supplementary Figures: 3 # Supplementary
Selective attention to emotional stimuli in a verbal go/no-go task: an fmri study
 BRAIN IMAGING NEUROREPORT Selective attention to emotional stimuli in a verbal go/no-go task: an fmri study Rebecca Elliott, CA Judy S. Rubinsztein, 1 Barbara J. Sahakian 1 and Raymond J. Dolan 2 Neuroscience
BRAIN IMAGING NEUROREPORT Selective attention to emotional stimuli in a verbal go/no-go task: an fmri study Rebecca Elliott, CA Judy S. Rubinsztein, 1 Barbara J. Sahakian 1 and Raymond J. Dolan 2 Neuroscience
Original Article. School of Medicine, Pittsburgh, PA, USA, b Department of Psychiatry, University of São Paulo
 Bipolar Disorders 2008: 10: 916 927 ª 2008 The Authors Journal compilation ª 2008 Blackwell Munksgaard BIPOLAR DISORDERS Original Article Elevated striatal and decreased dorsolateral prefrontal cortical
Bipolar Disorders 2008: 10: 916 927 ª 2008 The Authors Journal compilation ª 2008 Blackwell Munksgaard BIPOLAR DISORDERS Original Article Elevated striatal and decreased dorsolateral prefrontal cortical
Estimation of Statistical Power in a Multicentre MRI study.
 Estimation of Statistical Power in a Multicentre MRI study. PhD Student,Centre for Neurosciences Ninewells Hospital and Medical School University of Dundee. IPEM Conference Experiences and Optimisation
Estimation of Statistical Power in a Multicentre MRI study. PhD Student,Centre for Neurosciences Ninewells Hospital and Medical School University of Dundee. IPEM Conference Experiences and Optimisation
Neuroanatomy of Emotion, Fear, and Anxiety
 Neuroanatomy of Emotion, Fear, and Anxiety Outline Neuroanatomy of emotion Fear and anxiety Brain imaging research on anxiety Brain functional activation fmri Brain functional connectivity fmri Brain structural
Neuroanatomy of Emotion, Fear, and Anxiety Outline Neuroanatomy of emotion Fear and anxiety Brain imaging research on anxiety Brain functional activation fmri Brain functional connectivity fmri Brain structural
doi: /brain/aws024 Brain 2012: 135; Altered brain mechanisms of emotion processing in pre-manifest Huntington s disease
 doi:10.1093/brain/aws024 Brain 2012: 135; 1165 1179 1165 BRAIN A JOURNAL OF NEUROLOGY Altered brain mechanisms of emotion processing in pre-manifest Huntington s disease Marianne J. U. Novak, 1 Jason D.
doi:10.1093/brain/aws024 Brain 2012: 135; 1165 1179 1165 BRAIN A JOURNAL OF NEUROLOGY Altered brain mechanisms of emotion processing in pre-manifest Huntington s disease Marianne J. U. Novak, 1 Jason D.
Distinct neural activity associated with focused-attention meditation and loving-kindness meditation
 Title Distinct neural activity associated with focused-attention meditation and loving-kindness meditation Author(s) Lee, TMC; Leung, MK; Hou, WK; Tang, JCY; Jing, Y; So, KF; Lee, CF; Chan, CCH Citation
Title Distinct neural activity associated with focused-attention meditation and loving-kindness meditation Author(s) Lee, TMC; Leung, MK; Hou, WK; Tang, JCY; Jing, Y; So, KF; Lee, CF; Chan, CCH Citation
For better or for worse: neural systems supporting the cognitive down- and up-regulation of negative emotion
 For better or for worse: neural systems supporting the cognitive down- and up-regulation of negative emotion Kevin N. Ochsner, a, * Rebecca D. Ray, b Jeffrey C. Cooper, b Elaine R. Robertson, b Sita Chopra,
For better or for worse: neural systems supporting the cognitive down- and up-regulation of negative emotion Kevin N. Ochsner, a, * Rebecca D. Ray, b Jeffrey C. Cooper, b Elaine R. Robertson, b Sita Chopra,
Role of the ventral striatum in developing anorexia nervosa
 Role of the ventral striatum in developing anorexia nervosa Anne-Katharina Fladung 1 PhD, Ulrike M. E.Schulze 2 MD, Friederike Schöll 1, Kathrin Bauer 1, Georg Grön 1 PhD 1 University of Ulm, Department
Role of the ventral striatum in developing anorexia nervosa Anne-Katharina Fladung 1 PhD, Ulrike M. E.Schulze 2 MD, Friederike Schöll 1, Kathrin Bauer 1, Georg Grön 1 PhD 1 University of Ulm, Department
Voxel-based morphometry in clinical neurosciences
 Voxel-based morphometry in clinical neurosciences Ph.D. Thesis Ádám Feldmann Department of Behavioural Sciences Leader of Doctoral School: Prof. Dr.Sámuel Komoly, D.Sc. Program leader: Prof. Dr.Sámuel
Voxel-based morphometry in clinical neurosciences Ph.D. Thesis Ádám Feldmann Department of Behavioural Sciences Leader of Doctoral School: Prof. Dr.Sámuel Komoly, D.Sc. Program leader: Prof. Dr.Sámuel
Functional topography of a distributed neural system for spatial and nonspatial information maintenance in working memory
 Neuropsychologia 41 (2003) 341 356 Functional topography of a distributed neural system for spatial and nonspatial information maintenance in working memory Joseph B. Sala a,, Pia Rämä a,c,d, Susan M.
Neuropsychologia 41 (2003) 341 356 Functional topography of a distributed neural system for spatial and nonspatial information maintenance in working memory Joseph B. Sala a,, Pia Rämä a,c,d, Susan M.
Reporting Checklist for Nature Neuroscience
 Corresponding Author: Manuscript Number: Manuscript Type: Alex Pouget NN-A46249B Article Reporting Checklist for Nature Neuroscience # Main Figures: 7 # Supplementary Figures: 3 # Supplementary Tables:
Corresponding Author: Manuscript Number: Manuscript Type: Alex Pouget NN-A46249B Article Reporting Checklist for Nature Neuroscience # Main Figures: 7 # Supplementary Figures: 3 # Supplementary Tables:
Define functional MRI. Briefly describe fmri image acquisition. Discuss relative functional neuroanatomy. Review clinical applications.
 Dr. Peter J. Fiester November 14, 2012 Define functional MRI. Briefly describe fmri image acquisition. Discuss relative functional neuroanatomy. Review clinical applications. Briefly discuss a few examples
Dr. Peter J. Fiester November 14, 2012 Define functional MRI. Briefly describe fmri image acquisition. Discuss relative functional neuroanatomy. Review clinical applications. Briefly discuss a few examples
VIII. 10. Right Temporal-Lobe Contribution to the Retrieval of Family Relationships in Person Identification
 CYRIC Annual Report 2009 VIII. 10. Right Temporal-Lobe Contribution to the Retrieval of Family Relationships in Person Identification Abe N. 1, Fujii T. 1, Ueno A. 1, Shigemune Y. 1, Suzuki M. 2, Tashiro
CYRIC Annual Report 2009 VIII. 10. Right Temporal-Lobe Contribution to the Retrieval of Family Relationships in Person Identification Abe N. 1, Fujii T. 1, Ueno A. 1, Shigemune Y. 1, Suzuki M. 2, Tashiro
SUPPLEMENTARY MATERIAL. Table. Neuroimaging studies on the premonitory urge and sensory function in patients with Tourette syndrome.
 SUPPLEMENTARY MATERIAL Table. Neuroimaging studies on the premonitory urge and sensory function in patients with Tourette syndrome. Authors Year Patients Male gender (%) Mean age (range) Adults/ Children
SUPPLEMENTARY MATERIAL Table. Neuroimaging studies on the premonitory urge and sensory function in patients with Tourette syndrome. Authors Year Patients Male gender (%) Mean age (range) Adults/ Children
Pro-cognitive strategies in Bipolar Disorder: Challenges and New Directions. Sophia Frangou, MD, PhD, FRCPsych Professor of Psychiatry
 Pro-cognitive strategies in Bipolar Disorder: Challenges and New Directions Sophia Frangou, MD, PhD, FRCPsych Professor of Psychiatry Challenges 1. Conceptual challenges Relationship between cognitive
Pro-cognitive strategies in Bipolar Disorder: Challenges and New Directions Sophia Frangou, MD, PhD, FRCPsych Professor of Psychiatry Challenges 1. Conceptual challenges Relationship between cognitive
fmri and Voxel-based Morphometry in Detection of Early Stages of Alzheimer's Disease
 fmri and Voxel-based Morphometry in Detection of Early Stages of Alzheimer's Disease Andrey V. Sokolov 1,3, Sergey V. Vorobyev 2, Aleksandr Yu. Efimtcev 1,3, Viacheslav S. Dekan 1,3, Gennadiy E. Trufanov
fmri and Voxel-based Morphometry in Detection of Early Stages of Alzheimer's Disease Andrey V. Sokolov 1,3, Sergey V. Vorobyev 2, Aleksandr Yu. Efimtcev 1,3, Viacheslav S. Dekan 1,3, Gennadiy E. Trufanov
BINGES, BLUNTS AND BRAIN DEVELOPMENT
 BINGES, BLUNTS AND BRAIN DEVELOPMENT Why delaying the onset of alcohol and other drug use during adolescence is so important Aaron White, PhD Division of Epidemiology and Prevention Research National Institute
BINGES, BLUNTS AND BRAIN DEVELOPMENT Why delaying the onset of alcohol and other drug use during adolescence is so important Aaron White, PhD Division of Epidemiology and Prevention Research National Institute
Nature Neuroscience: doi: /nn Supplementary Figure 1. Task timeline for Solo and Info trials.
 Supplementary Figure 1 Task timeline for Solo and Info trials. Each trial started with a New Round screen. Participants made a series of choices between two gambles, one of which was objectively riskier
Supplementary Figure 1 Task timeline for Solo and Info trials. Each trial started with a New Round screen. Participants made a series of choices between two gambles, one of which was objectively riskier
HST.583 Functional Magnetic Resonance Imaging: Data Acquisition and Analysis Fall 2006
 MIT OpenCourseWare http://ocw.mit.edu HST.583 Functional Magnetic Resonance Imaging: Data Acquisition and Analysis Fall 2006 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms.
MIT OpenCourseWare http://ocw.mit.edu HST.583 Functional Magnetic Resonance Imaging: Data Acquisition and Analysis Fall 2006 For information about citing these materials or our Terms of Use, visit: http://ocw.mit.edu/terms.
doi: /brain/awq006 Brain 2010: 133; Imaging memory in temporal lobe epilepsy: predicting the effects of temporal lobe resection
 doi:10.1093/brain/awq006 Brain 2010: 133; 1186 1199 1186 BRAIN A JOURNAL OF NEUROLOGY Imaging memory in temporal lobe epilepsy: predicting the effects of temporal lobe resection Silvia B. Bonelli, 1,2
doi:10.1093/brain/awq006 Brain 2010: 133; 1186 1199 1186 BRAIN A JOURNAL OF NEUROLOGY Imaging memory in temporal lobe epilepsy: predicting the effects of temporal lobe resection Silvia B. Bonelli, 1,2
Self-related neural response to tailored smoking-cessation messages. predicts quitting
 Neural response to tailored messages and quitting Page 1 Supporting Online Material Self-related neural response to tailored smoking-cessation messages predicts quitting Hannah Faye Chua 1, S. Shaun Ho
Neural response to tailored messages and quitting Page 1 Supporting Online Material Self-related neural response to tailored smoking-cessation messages predicts quitting Hannah Faye Chua 1, S. Shaun Ho
Distinct neural bases of disruptive behavior and autism symptom severity in boys with autism spectrum disorder.
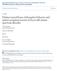 Himmelfarb Health Sciences Library, The George Washington University Health Sciences Research Commons Pediatrics Faculty Publications Pediatrics 1-1-2017 Distinct neural bases of disruptive behavior and
Himmelfarb Health Sciences Library, The George Washington University Health Sciences Research Commons Pediatrics Faculty Publications Pediatrics 1-1-2017 Distinct neural bases of disruptive behavior and
SUPPLEMENTARY MATERIALS: Appetitive and aversive goal values are encoded in the medial orbitofrontal cortex at the time of decision-making
 SUPPLEMENTARY MATERIALS: Appetitive and aversive goal values are encoded in the medial orbitofrontal cortex at the time of decision-making Hilke Plassmann 1,2, John P. O'Doherty 3,4, Antonio Rangel 3,5*
SUPPLEMENTARY MATERIALS: Appetitive and aversive goal values are encoded in the medial orbitofrontal cortex at the time of decision-making Hilke Plassmann 1,2, John P. O'Doherty 3,4, Antonio Rangel 3,5*
Prefrontal mediation of age differences in cognitive reappraisal
 Neurobiology of Aging 33 (2012) 645 655 www.elsevier.com/locate/neuaging Prefrontal mediation of age differences in cognitive reappraisal Philipp C. Opitz, Lindsay C. Rauch, Douglas P. Terry, Heather L.
Neurobiology of Aging 33 (2012) 645 655 www.elsevier.com/locate/neuaging Prefrontal mediation of age differences in cognitive reappraisal Philipp C. Opitz, Lindsay C. Rauch, Douglas P. Terry, Heather L.
Network-based pattern recognition models for neuroimaging
 Network-based pattern recognition models for neuroimaging Maria J. Rosa Centre for Neuroimaging Sciences, Institute of Psychiatry King s College London, UK Outline Introduction Pattern recognition Network-based
Network-based pattern recognition models for neuroimaging Maria J. Rosa Centre for Neuroimaging Sciences, Institute of Psychiatry King s College London, UK Outline Introduction Pattern recognition Network-based
Reviews and Overviews. Mechanisms of Psychiatric Illness
 Reviews and Overviews Mechanisms of Psychiatric Illness A Critical Appraisal of Neuroimaging Studies of Bipolar Disorder: Toward a New Conceptualization of Underlying Neural Circuitry and a Road Map for
Reviews and Overviews Mechanisms of Psychiatric Illness A Critical Appraisal of Neuroimaging Studies of Bipolar Disorder: Toward a New Conceptualization of Underlying Neural Circuitry and a Road Map for
SUPPLEMENTARY INFORMATION
 doi:10.1038/nature11239 Introduction The first Supplementary Figure shows additional regions of fmri activation evoked by the task. The second, sixth, and eighth shows an alternative way of analyzing reaction
doi:10.1038/nature11239 Introduction The first Supplementary Figure shows additional regions of fmri activation evoked by the task. The second, sixth, and eighth shows an alternative way of analyzing reaction
Real-Time fmri Neurofeedback Training of Amygdala Activity in Patients with Major Depressive Disorder
 Real-Time fmri Neurofeedback Training of Amygdala Activity in Patients with Major Depressive Disorder Kymberly D. Young 1, Vadim Zotev 1, Raquel Phillips 1, Masaya Misaki 1, Han Yuan 1, Wayne C. Drevets
Real-Time fmri Neurofeedback Training of Amygdala Activity in Patients with Major Depressive Disorder Kymberly D. Young 1, Vadim Zotev 1, Raquel Phillips 1, Masaya Misaki 1, Han Yuan 1, Wayne C. Drevets
Title of file for HTML: Supplementary Information Description: Supplementary Figures, Supplementary Tables and Supplementary References
 Title of file for HTML: Supplementary Information Description: Supplementary Figures, Supplementary Tables and Supplementary References Supplementary Information Supplementary Figure 1. The mean parameter
Title of file for HTML: Supplementary Information Description: Supplementary Figures, Supplementary Tables and Supplementary References Supplementary Information Supplementary Figure 1. The mean parameter
Reporting Checklist for Nature Neuroscience
 Corresponding Author: Manuscript Number: Manuscript Type: Simon Musall NNA47695 Article Reporting Checklist for Nature Neuroscience # Main Figures: 6 # Supplementary Figures: 14 # Supplementary Tables:
Corresponding Author: Manuscript Number: Manuscript Type: Simon Musall NNA47695 Article Reporting Checklist for Nature Neuroscience # Main Figures: 6 # Supplementary Figures: 14 # Supplementary Tables:
Self-Regulation of Amygdala Activation Using Real-Time fmri Neurofeedback
 Self-Regulation of Amygdala Activation Using Real-Time fmri Neurofeedback Vadim Zotev 1., Frank Krueger 2,3., Raquel Phillips 1, Ruben P. Alvarez 1, W. Kyle Simmons 1, Patrick Bellgowan 1, Wayne C. Drevets
Self-Regulation of Amygdala Activation Using Real-Time fmri Neurofeedback Vadim Zotev 1., Frank Krueger 2,3., Raquel Phillips 1, Ruben P. Alvarez 1, W. Kyle Simmons 1, Patrick Bellgowan 1, Wayne C. Drevets
Supporting Information
 Supporting Information Braver et al. 10.1073/pnas.0808187106 SI Methods Participants. Participants were neurologically normal, righthanded younger or older adults. The groups did not differ in gender breakdown
Supporting Information Braver et al. 10.1073/pnas.0808187106 SI Methods Participants. Participants were neurologically normal, righthanded younger or older adults. The groups did not differ in gender breakdown
Biology of Mood & Anxiety Disorders 2012, 2:11
 Biology of Mood & Anxiety Disorders This Provisional PDF corresponds to the article as it appeared upon acceptance. Fully formatted PDF and full text (HTML) versions will be made available soon. Neural
Biology of Mood & Anxiety Disorders This Provisional PDF corresponds to the article as it appeared upon acceptance. Fully formatted PDF and full text (HTML) versions will be made available soon. Neural
Supplementary Information Appendix: Default Mode Contributions to Automated Information Processing
 Supplementary Information Appendix: Default Mode Contributions to Automated Information Processing Authors: Deniz Vatansever a,b,c,1, David K. Menon a,d, Emmanuel A. Stamatakis a,b,d Affiliations: a Division
Supplementary Information Appendix: Default Mode Contributions to Automated Information Processing Authors: Deniz Vatansever a,b,c,1, David K. Menon a,d, Emmanuel A. Stamatakis a,b,d Affiliations: a Division
Supporting Information
 Supporting Information Carhart-Harris et al. 10.1073/pnas.1119598109 Fig. S1. Slices for arterial spin labeling (ASL) result. Lightbox display of slices showing regions where there were significant decreases
Supporting Information Carhart-Harris et al. 10.1073/pnas.1119598109 Fig. S1. Slices for arterial spin labeling (ASL) result. Lightbox display of slices showing regions where there were significant decreases
Relationship between ambient light and glucose metabolism in healthy subjects
 https://doi.org/10.1186/s12868-018-0444-x BMC Neuroscience RESEARCH ARTICLE Open Access Relationship between ambient light and glucose metabolism in healthy subjects Hirofumi Hirakawa *, Takeshi Terao,
https://doi.org/10.1186/s12868-018-0444-x BMC Neuroscience RESEARCH ARTICLE Open Access Relationship between ambient light and glucose metabolism in healthy subjects Hirofumi Hirakawa *, Takeshi Terao,
Differences in resting corticolimbic functional connectivity in bipolar I euthymia
 Bipolar Disorders 2013 Original Article Ó 2013 John Wiley and Sons A/S Published by Blackwell Publishing Ltd. BIPOLAR DISORDERS Differences in resting corticolimbic functional connectivity in bipolar I
Bipolar Disorders 2013 Original Article Ó 2013 John Wiley and Sons A/S Published by Blackwell Publishing Ltd. BIPOLAR DISORDERS Differences in resting corticolimbic functional connectivity in bipolar I
The Critical Relationship between the Timing of Stimulus Presentation and Data Acquisition in Blocked Designs with fmri
 NeuroImage 10, 36 44 (1999) Article ID nimg.1999.0447, available online at http://www.idealibrary.com on The Critical Relationship between the Timing of Stimulus Presentation and Data Acquisition in Blocked
NeuroImage 10, 36 44 (1999) Article ID nimg.1999.0447, available online at http://www.idealibrary.com on The Critical Relationship between the Timing of Stimulus Presentation and Data Acquisition in Blocked
Differential contributions of subregions of medial temporal lobe to memory system in. amnestic mild cognitive impairment: insights from fmri study
 Differential contributions of subregions of medial temporal lobe to memory system in amnestic mild cognitive impairment: insights from fmri study Jiu Chen 1, *, Xujun Duan 2, *, Hao Shu 1, Zan Wang 1,
Differential contributions of subregions of medial temporal lobe to memory system in amnestic mild cognitive impairment: insights from fmri study Jiu Chen 1, *, Xujun Duan 2, *, Hao Shu 1, Zan Wang 1,
Neural correlates of recognition memory of social information in people with schizophrenia
 Research Paper Neural correlates of recognition memory of social information in people with schizophrenia Philippe-Olivier Harvey, PhD; Martin Lepage, PhD Department of Psychiatry, McGill University, Montréal,
Research Paper Neural correlates of recognition memory of social information in people with schizophrenia Philippe-Olivier Harvey, PhD; Martin Lepage, PhD Department of Psychiatry, McGill University, Montréal,
Maintaining Force Control Despite Changes in Emotional Context Engages Dorsomedial Prefrontal and Premotor Cortex
 Cerebral Cortex Advance Access published June 15, 2011 Cerebral Cortex doi:10.1093/cercor/bhr141 Maintaining Force Control Despite Changes in Emotional Context Engages Dorsomedial Prefrontal and Premotor
Cerebral Cortex Advance Access published June 15, 2011 Cerebral Cortex doi:10.1093/cercor/bhr141 Maintaining Force Control Despite Changes in Emotional Context Engages Dorsomedial Prefrontal and Premotor
The neural correlates of belief-bias inhibition: The impact of logic training
 The neural correlates of belief-bias inhibition: The impact of logic training Item type Authors Citation DOI Publisher Journal Rights Article Luo, Junlong; Tang, Xiaochen; Zhang, Entao; Stupple, Edward
The neural correlates of belief-bias inhibition: The impact of logic training Item type Authors Citation DOI Publisher Journal Rights Article Luo, Junlong; Tang, Xiaochen; Zhang, Entao; Stupple, Edward
Title: Pinpointing resilience in Bipolar Disorder
 Title: Pinpointing resilience in Bipolar Disorder 1. AIM OF THE RESEARCH AND BRIEF BACKGROUND Bipolar disorder (BD) is a mood disorder characterised by episodes of depression and mania. It ranks as one
Title: Pinpointing resilience in Bipolar Disorder 1. AIM OF THE RESEARCH AND BRIEF BACKGROUND Bipolar disorder (BD) is a mood disorder characterised by episodes of depression and mania. It ranks as one
Common Neural Substrates for Ordinal Representation in Short-Term Memory, Numerical and Alphabetical Cognition
 Common Neural Substrates for Ordinal Representation in Short-Term Memory, Numerical and Alphabetical Cognition Lucie Attout 1 *, Wim Fias 2, Eric Salmon 3, Steve Majerus 1 1 Department of Psychology -
Common Neural Substrates for Ordinal Representation in Short-Term Memory, Numerical and Alphabetical Cognition Lucie Attout 1 *, Wim Fias 2, Eric Salmon 3, Steve Majerus 1 1 Department of Psychology -
Title:Atypical language organization in temporal lobe epilepsy revealed by a passive semantic paradigm
 Author's response to reviews Title:Atypical language organization in temporal lobe epilepsy revealed by a passive semantic paradigm Authors: Julia Miro (juliamirollado@gmail.com) Pablo Ripollès (pablo.ripolles.vidal@gmail.com)
Author's response to reviews Title:Atypical language organization in temporal lobe epilepsy revealed by a passive semantic paradigm Authors: Julia Miro (juliamirollado@gmail.com) Pablo Ripollès (pablo.ripolles.vidal@gmail.com)
The Hippocampus Remains Activated over the Long Term for the Retrieval of Truly Episodic Memories
 The Hippocampus Remains Activated over the Long Term for the Retrieval of Truly Episodic Memories Caroline Harand 1,2,3,4, Françoise Bertran 1,2,3,5, Renaud La Joie 1,2,3,4, Brigitte Landeau 1,2,3,4, Florence
The Hippocampus Remains Activated over the Long Term for the Retrieval of Truly Episodic Memories Caroline Harand 1,2,3,4, Françoise Bertran 1,2,3,5, Renaud La Joie 1,2,3,4, Brigitte Landeau 1,2,3,4, Florence
