Transcranial Magnetic Stimulation
|
|
|
- Regina Todd
- 5 years ago
- Views:
Transcription
1 Transcranial Magnetic Stimulation Date of Origin: 7/24/2018 Last Review Date: 7/24/2018 Effective Date: 08/01/18 Dates Reviewed: 7/24/2018 Developed By: Medical Necessity Criteria Committee I. Description Transcranial magnetic stimulation (TMS), including repetitive transcranial magnetic stimulation (rtms) and deep transcranial magnetic stimulation (dtms), is a noninvasive method of delivering electrical stimulation to the brain. It uses a specifically designed magnetic coil that is placed in contact with the scalp to generate rapidly alternating magnetic fields and produces electrical stimulation of cortical neurons. The procedure takes approximately minutes and is generally administered daily over a four to seven week period. TMS can be performed in an office setting as it does not require anesthesia and does not induce a convulsion. Imaging studies have shown a decrease in activity of the left dorsolateral prefrontal cortex (DLPFC) in depressed patients, and early studies suggested that high frequency (e.g., 5 10 Hz) TMS of the left DLPFC had antidepressant effects. The FDA approved TMS in October 2008 for use in the treatment of treatment-refractory major depressive disorder (TRD) based on the results of a multisite randomized controlled clinical trial. Scientific literature supports the safety and effectiveness of TMS in treating TRD. It is believed to be generally less effective than Electroconvulsive Therapy (ECT) but with a much more benign side-effect profile. Data do not show an advantage for either rtms or dtms compared with the other. A typical course of TMS is 5 days a week for 6 weeks (total of 30 sessions), followed by a 3 week taper of 3 TMS treatments in week 1; 2 TMS treatments the next week, and; 1 TMS treatment in the last week. The role of follow-up or maintenance treatment has not been established in the literature and there is not yet a generally accepted protocol for maintenance treatment. As a result, maintenance TMS is considered by some to be experimental/investigational. Given that TRD is a chronic condition, some patients with a positive response to TMS may benefit from reintroduction or some form of booster sessions. Clinical judgment with consideration of patient history and resources, response to treatment, and appropriateness of other treatments is necessary in order to weigh the appropriateness of additional treatment beyond the acute course. The Food and Drug Administration (FDA) has approved several transcranial magnetic stimulation (TMS) systems and devices (e.g., NeuroStar TMS Therapy System (Neuronetics, Inc.), Brainsway Deep TMS System (Brainsway Ltd.), Magstim Rapid2 Therapy System (Magstim Company Limited), MagVita TMS Therapy System (Tonica Elektronik A/S)). Moda Health Medical Necessity Criteria Page 1/5
2 II. Criteria: CWQI BHC-0014 A. Initiation Criteria: Authorization for Initiation of Transcranial magnetic stimulation (TMS) is indicated by ALL of the following: 1. Confirmed diagnosis of severe major depressive disorder (single episode or recurrent) documented by a standardized evidence based depression rating scale that reliably measures depressive symptoms. 2. The patient is age of 18 or older. 3. The patient has had adequate trials of other treatments with inadequate response documented on a standardized measurement tool, including ALL of the following: a) Appropriate pharmacological treatment consisting of at least four different psychopharmacologic agents with adequate dose and duration (or discontinued due to intolerable side-effects) including (1) At least one augmentation trial; and (2) At least two different agent classes. b) An adequate trial of an evidenced based psychotherapy within the current depressive episode; AND 4. Treating provider has ruled out the presence of contraindications such as active substance abuse, seizure disorder, or any medications, implants or devices that may compromise the safety or efficacy of the procedure: B. Continued Care Criteria: Authorization extending a standard course of TMS beyond 36 sessions is indicated by ALL of the following: 1. The patient has shown a positive response to treatment as evidenced by a reduction in depressive symptoms on a standardized measurement tool. 2. Additional visits are needed due to a need for re-mapping or other confounding factors. 3. The treatment plan includes a plan for completing treatment with the lowest appropriate number of additional sessions. C. Reintroduction Criteria: Authorization for reintroducing TMS after completion of an acute episode of treatment is indicated by ALL of the following: 1. Clinical need for additional treatment is demonstrated by 1 or more of the following: a) Recurrence of symptoms meeting criteria for initial authorization of TMS; or b) Patient history demonstrating a substantial risk of deterioration that can t be prevented by other means such as pharmacological and/or psychotherapeutic intervention. 2. A reasonable expectation that additional treatment will produce clinical benefit 3. Other treatment approaches (e.g., psychotherapy, pharmacotherapy) are employed concurrently as appropriate; AND 4. Treatment is provided at the lowest number of sessions required to maintain/continue improvement. Moda Health Medical Necessity Criteria Page 2/5
3 III. Information Submitted with the Prior Authorization Request: Psychiatric evaluation including diagnosis, symptomatology, results of standardized evidence based depression rating scale, treatment history (psychotherapeutic and pharmacologic), screening for contraindications and requested services. IV. CPT or HCPC codes covered: Codes Description Therapeutic repetitive transcranial magnetic stimulation (TMS) treatment; initial, including cortical mapping, motor threshold determination, delivery and management Therapeutic repetitive transcranial magnetic stimulation (TMS) treatment; subsequent delivery and management, per session Therapeutic repetitive transcranial magnetic stimulation (TMS) treatment; subsequent motor threshold redetermination with delivery and management. V. Annual Review History Review Date Revisions Effective Date 8/1/2018 New criteria 8/1/2018 VI. References Avery DH et al. (2008). Transcranial Magnetic Stimulation in the Acute Treatment of Major Depressive Disorder: Clinical Response in an Open-Label Extension Trial. J Clin Psychiatry 69(3): Connolly KR, Helmer A, Cristancho MA, Cristancho P, O'Reardon JP (2012). Effectiveness of transcranial magnetic stimulation in clinical practice post-fda approval in the United States: results observed with the first 100 consecutive cases of depression at an academic medical center. J Clin Psychiatry. 73(4). Dunner, DL, et al. (2014). A Multisite, Naturalistic, Observational Study of Transcranial Magnetic Stimulation for Patients With Pharmacoresistant Major Depressive Disorder: Durability of Benefit Over a 1-Year Follow-Up Period. J Clin Psychiatry 75(12): Fitzgerald PB, Grace N, Hoy KE, Bailey M, Daskalakis ZJ, (2013). An open label trial of clustered maintenance rtms for patients with refractory depression. Brain Stimul. 6(3): Moda Health Medical Necessity Criteria Page 3/5
4 George, MS et al. (2010). Daily Left Prefrontal Transcranial Magnetic Stimulation Therapy for Major Depressive Disorder: A Sham-Controlled Randomized Trial. Arch Gen Psychiatry. 67(5): Kedzior KK, Reitz SK, Azorina V, Loo C. (2015). Durability of the antidepressant effect of the highfrequency repetitive transcranial magnetic stimulation (rtms) In the absence of maintenance treatment in major depression: a systematic review and meta-analysis of 16 double-blind, randomized, shamcontrolled trials. Depress Anxiety. 32(3): Kelly, MS et al. (2017). Initial Response to Transcranial Magnetic Stimulation Treatment for Depression Predicts Subsequent Response. J Neuropsychiatry Clin Neurosci. 29(2): Levkovitz, Y et al. (2015). Efficacy and safety of deep transcranial magnetic stimulation for major depression: a prospective multicenter randomized controlled trial. World Psychiatry. 14: O'Reardon JP, Blumner KH, Peshek AD, Pradilla RR, Pimiento PC (2005). Long-term maintenance therapy for major depressive disorder with rtms. J Clin Psychiatry. 66(12): O Reardon JP et al. (2007). Efficacy and Safety of Transcranial Magnetic Stimulation in the Acute Treatment of Major Depression: A Multisite Randomized Controlled Trial. Biol Psychiatry. 62: Richieri R et al. (2013). Maintenance transcranial magnetic stimulation reduces depression relapse: a propensity-adjusted analysis. J Affect Disord. 151(1): VII. Appendix 1 Applicable ICD-10 diagnosis codes: Codes Description F33.2 Major depressive disorder, Recurrent, Severe F32.2 Major depressive disorder, Single episode, Severe VIII. Appendix 1 Centers for Medicare and Medicaid Services (CMS) Medicare coverage for outpatient (Part B) drugs is outlined in the Medicare Benefit Policy Manual (Pub ), Chapter 15, 50 Drugs and Biologicals. In addition, National Coverage Determination (NCD) and Local Coverage Determinations (LCDs) may exist and compliance with these policies is required where applicable. They can be found at: Additional indications may be covered at the discretion of the health plan. Medicare Part B Covered Diagnosis Codes (applicable to existing NCD/LCD): Moda Health Medical Necessity Criteria Page 4/5
5 Jurisdiction(s): 5, 8 Wisconsin Physicians Service Insurance Corporation: L34641 NCD/LCD Document (s): NCD/LCD Document (s): Transcranial Magnetic Stimulation (TMS) L34641 Medicare Part B Administrative Contractor (MAC) Jurisdictions Jurisdiction Applicable State/US Territory Contractor F (2 & 3) AK, WA, OR, ID, ND, SD, MT, WY, UT, AZ Noridian Healthcare Solutions, LLC Moda Health Medical Necessity Criteria Page 5/5
Todd Hutton, M.D. Karl Lanocha, M.D Richard Bermudes, M.D. Kimberly Cress, M.D.
 Transcranial Magnetic Stimulation: What You Need To Know Todd Hutton, M.D. Karl Lanocha, M.D Richard Bermudes, M.D. Kimberly Cress, M.D. Disclosures Todd Hutton, M.D. Speaker for Neuronetics Board of Directors,
Transcranial Magnetic Stimulation: What You Need To Know Todd Hutton, M.D. Karl Lanocha, M.D Richard Bermudes, M.D. Kimberly Cress, M.D. Disclosures Todd Hutton, M.D. Speaker for Neuronetics Board of Directors,
Intrathecal Opioid Therapy for Management of Chronic Pain
 Intrathecal Opioid Therapy for Management of Chronic Pain Date of Origin: 01/2000 Last Review Date: 09/27/2017 Effective Date: 09/27/2017 Dates Reviewed: 11/2002, 12/2003, 12/2004, 12/2005, 12/2006, 12/2007,
Intrathecal Opioid Therapy for Management of Chronic Pain Date of Origin: 01/2000 Last Review Date: 09/27/2017 Effective Date: 09/27/2017 Dates Reviewed: 11/2002, 12/2003, 12/2004, 12/2005, 12/2006, 12/2007,
Repetitive Transcranial Magnetic Stimulation as a for Treatment of Refractory Depression and other Psychiatric/Neurologic Disorders
 Repetitive Transcranial Magnetic Stimulation as a for Treatment of Refractory Depression and other Psychiatric/Neurologic Disorders Policy Number: Original Effective Date: MM.12.015 08/01/2014 Line(s)
Repetitive Transcranial Magnetic Stimulation as a for Treatment of Refractory Depression and other Psychiatric/Neurologic Disorders Policy Number: Original Effective Date: MM.12.015 08/01/2014 Line(s)
Continuous Glucose Monitoring (CGM)
 Continuous Glucose Monitoring (CGM) Date of Origin: 02/2001 Last Review Date: 07/26/2017 Effective Date: 07/26/2017 Dates Reviewed: 04/2004, 04/2005, 03/2006, 11/2006, 12/2007, 03/2008, 09/2008, 04/2009,
Continuous Glucose Monitoring (CGM) Date of Origin: 02/2001 Last Review Date: 07/26/2017 Effective Date: 07/26/2017 Dates Reviewed: 04/2004, 04/2005, 03/2006, 11/2006, 12/2007, 03/2008, 09/2008, 04/2009,
Anesthesia for Routine Gastrointestinal Endoscopic Procedures (Additional description)
 Anesthesia for Routine Gastrointestinal Endoscopic Procedures (Additional description) Date of Origin: 05/2012 Last Review Date: 12/06/2017 Effective Date: 01/01/2018 Dates Reviewed: 12/2013, 11/2014,
Anesthesia for Routine Gastrointestinal Endoscopic Procedures (Additional description) Date of Origin: 05/2012 Last Review Date: 12/06/2017 Effective Date: 01/01/2018 Dates Reviewed: 12/2013, 11/2014,
Krystexxa (pegloticase) Document Number: IC-0158
 Krystexxa (pegloticase) Document Number: IC-0158 Last Review Date: 06/27/2017 Date of Origin: 02/07/20103 Dates Reviewed: 11/2013, 08/2014, 07/2015, 07/2016, 09/2016, 12/2016, 03/2017, 06/2017 I. Length
Krystexxa (pegloticase) Document Number: IC-0158 Last Review Date: 06/27/2017 Date of Origin: 02/07/20103 Dates Reviewed: 11/2013, 08/2014, 07/2015, 07/2016, 09/2016, 12/2016, 03/2017, 06/2017 I. Length
Eylea (aflibercept) Document Number: IC-0026
 Eylea (aflibercept) Document Number: IC-0026 Last Review Date: 3/1/2018 Date of Origin: 02/07/2013 Dates Reviewed: 03/07/2013, 06/2013, 09/2013, 12/2013, 03/2014, 06/2014, 09/2014, 12/2014, 03/2015, 04/2015,
Eylea (aflibercept) Document Number: IC-0026 Last Review Date: 3/1/2018 Date of Origin: 02/07/2013 Dates Reviewed: 03/07/2013, 06/2013, 09/2013, 12/2013, 03/2014, 06/2014, 09/2014, 12/2014, 03/2015, 04/2015,
Intravitreal Avastin (Bevacizumab)
 Intravitreal Avastin (Bevacizumab) Date of Origin: 10/18/2018 Last Review Date: 10/18/2018 Effective Date: 10/18/2018 Dates Reviewed: 10/2018 Developed By: Medical Criteria Committee I. Length of Authorization
Intravitreal Avastin (Bevacizumab) Date of Origin: 10/18/2018 Last Review Date: 10/18/2018 Effective Date: 10/18/2018 Dates Reviewed: 10/2018 Developed By: Medical Criteria Committee I. Length of Authorization
Original Effective Date: 8/28/2013. Subject: Transcranial Magnetic Stimulation for the Treatment of Major Depression
 Subject: Transcranial Magnetic Stimulation for the Treatment of Major Depression Guidance Number: MCG-104 Revision Date(s): Original Effective Date: 8/28/2013 Medical Coverage Guidance Approval Date: 8/28/2013
Subject: Transcranial Magnetic Stimulation for the Treatment of Major Depression Guidance Number: MCG-104 Revision Date(s): Original Effective Date: 8/28/2013 Medical Coverage Guidance Approval Date: 8/28/2013
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Transcranial Magnetic Stimulation (TMS) Page 1 of 21 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: See also: Transcranial Magnetic Stimulation (TMS) Vagus Nerve
Transcranial Magnetic Stimulation (TMS) Page 1 of 21 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: See also: Transcranial Magnetic Stimulation (TMS) Vagus Nerve
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Transcranial Magnetic Stimulation (TMS) Page 1 of 24 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: See also: Transcranial Magnetic Stimulation (TMS) Vagus Nerve
Transcranial Magnetic Stimulation (TMS) Page 1 of 24 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: See also: Transcranial Magnetic Stimulation (TMS) Vagus Nerve
Velcade (bortezomib) Document Number: IC-0137
 Velcade (bortezomib) Document Number: IC-0137 Last Review Date: 11/21/2017 Date of Origin: 11/28/2011 Dates Reviewed: 12/2011, 03/2012, 06/2012, 09/2012, 12/2012, 03/2013, 06/2013, 09/2013, 12/2013, 03/2014,
Velcade (bortezomib) Document Number: IC-0137 Last Review Date: 11/21/2017 Date of Origin: 11/28/2011 Dates Reviewed: 12/2011, 03/2012, 06/2012, 09/2012, 12/2012, 03/2013, 06/2013, 09/2013, 12/2013, 03/2014,
Transcranial Magnetic Stimulation for the Treatment of Depression
 Transcranial Magnetic Stimulation for the Treatment of Depression Paul E. Holtzheimer, MD Associate Professor Departments of Psychiatry and Surgery Geisel School of Medicine at Dartmouth Dartmouth-Hitchcock
Transcranial Magnetic Stimulation for the Treatment of Depression Paul E. Holtzheimer, MD Associate Professor Departments of Psychiatry and Surgery Geisel School of Medicine at Dartmouth Dartmouth-Hitchcock
Treatment of Depression: A Brief History
 2500 82nd Place Urbandale, Ia Treatment of Depression: A Brief History 1960 s to Present Community Based Services SSRI Antidepressants SNRI Antidepressants 1970 s to present 20% of patients do not respond
2500 82nd Place Urbandale, Ia Treatment of Depression: A Brief History 1960 s to Present Community Based Services SSRI Antidepressants SNRI Antidepressants 1970 s to present 20% of patients do not respond
Dates Reviewed: 12/2012, 3/2013, 6/2013, 9/2013, 11/2013, 12/2013, 3/2014, 6/2014, 9/2014, 12/2014,
 Perjeta (pertuzumab) Last Review Date: 5/30/2017 Date of Origin: 11/01/2012 Document Number: IC-0096 Dates Reviewed: 12/2012, 3/2013, 6/2013, 9/2013, 11/2013, 12/2013, 3/2014, 6/2014, 9/2014, 12/2014,
Perjeta (pertuzumab) Last Review Date: 5/30/2017 Date of Origin: 11/01/2012 Document Number: IC-0096 Dates Reviewed: 12/2012, 3/2013, 6/2013, 9/2013, 11/2013, 12/2013, 3/2014, 6/2014, 9/2014, 12/2014,
Transcranial Magnetic Stimulation and Cranial Electrical Stimulation (CES) as a Treatment of Depression and Other Psychiatric/Neurologic Disorders
 Transcranial Magnetic Stimulation and Cranial Electrical Stimulation (CES) as a Treatment of Depression and Other Psychiatric/Neurologic Disorders Policy Number: 2.01.50 Last Review: 10/2017 Origination:
Transcranial Magnetic Stimulation and Cranial Electrical Stimulation (CES) as a Treatment of Depression and Other Psychiatric/Neurologic Disorders Policy Number: 2.01.50 Last Review: 10/2017 Origination:
Herniated Disc Treatment Non-covered Procedures
 Herniated Disc Treatment Non-covered Procedures Date of Origin: 03/2004 Last Review Date: 03/22/2017 Effective Date: 03/22/2017 Dates Reviewed: 02/2005, 02/2006, 02/2007, 03/2008, 01/2009, 04/2009, 02/2011,
Herniated Disc Treatment Non-covered Procedures Date of Origin: 03/2004 Last Review Date: 03/22/2017 Effective Date: 03/22/2017 Dates Reviewed: 02/2005, 02/2006, 02/2007, 03/2008, 01/2009, 04/2009, 02/2011,
Imfinzi (durvalumab) (Intravenous)
 Imfinzi (durvalumab) (Intravenous) Last Review Date: 09/05/2018 Date of Origin: 05/30/2017 Dates Reviewed: 05/2017, 08/2017, 11/2017, 02/2018, 05/2018, 09/2018 Document Number: IC-0301 I. Length of Authorization
Imfinzi (durvalumab) (Intravenous) Last Review Date: 09/05/2018 Date of Origin: 05/30/2017 Dates Reviewed: 05/2017, 08/2017, 11/2017, 02/2018, 05/2018, 09/2018 Document Number: IC-0301 I. Length of Authorization
HCT Medical Policy. Transcranial Magnetic Stimulation (TMS) for Major Depression Policy # 121 Current Effective Date: 05/24/2016.
 HCT Medical Policy Transcranial Magnetic Stimulation (TMS) for Major Depression Policy # 121 Current Effective Date: 05/24/2016 Medical Policies are developed by HealthyCT to assist in administering plan
HCT Medical Policy Transcranial Magnetic Stimulation (TMS) for Major Depression Policy # 121 Current Effective Date: 05/24/2016 Medical Policies are developed by HealthyCT to assist in administering plan
Pulmonary Rehabilitation
 Pulmonary Rehabilitation Date of Origin: 06/2005 Last Review Date: 10/25/2017 Effective Date: 10/25/2017 Dates Reviewed: 05/2006, 05/2007, 05/2008, 11/2009, 02/2011, 01/2012, 08/2013, 07/2014, 09/2015,
Pulmonary Rehabilitation Date of Origin: 06/2005 Last Review Date: 10/25/2017 Effective Date: 10/25/2017 Dates Reviewed: 05/2006, 05/2007, 05/2008, 11/2009, 02/2011, 01/2012, 08/2013, 07/2014, 09/2015,
Tecentriq (atezolizumab) (Intravenous)
 Tecentriq (atezolizumab) (Intravenous) Last Review Date: 06/01/2018 Date of Origin: 06/28/2016 Document Number: IC-0278 Dates Reviewed: 06/2016, 08/2016, 10/2016, 02/2017, 04/2017, 08/2017, 11/2017, 02/2018,
Tecentriq (atezolizumab) (Intravenous) Last Review Date: 06/01/2018 Date of Origin: 06/28/2016 Document Number: IC-0278 Dates Reviewed: 06/2016, 08/2016, 10/2016, 02/2017, 04/2017, 08/2017, 11/2017, 02/2018,
Yervoy (ipilmumab) Last Review Date: 03/25/2014 Date of Origin: 11/28/2011. Prior Auth Available: Post-Service Edit:
 Yervoy (ipilmumab) Date of Origin: 11/28/2011 Prior Auth Available: Post-Service Edit: Dates Reviewed: 12/13/2011, 03/2012, 06/19/2012, 09/06/2012, 12/06/2012, 05/16/2013, 06/06/2013, 09/05/2013, 12/05/2013,
Yervoy (ipilmumab) Date of Origin: 11/28/2011 Prior Auth Available: Post-Service Edit: Dates Reviewed: 12/13/2011, 03/2012, 06/19/2012, 09/06/2012, 12/06/2012, 05/16/2013, 06/06/2013, 09/05/2013, 12/05/2013,
Statement on Repetitive Transcranial Magnetic Stimulation for Depression. Position statement CERT03/17
 Statement on Repetitive Transcranial Magnetic Stimulation for Depression Position statement CERT03/17 Approved by the Royal College of Psychiatrists, Committee on ECT and Related Treatments: February 2017
Statement on Repetitive Transcranial Magnetic Stimulation for Depression Position statement CERT03/17 Approved by the Royal College of Psychiatrists, Committee on ECT and Related Treatments: February 2017
Neuromodulation Approaches to Treatment Resistant Depression
 1 Alternative Treatments: Neuromodulation Approaches to Treatment Resistant Depression Audrey R. Tyrka, MD, PhD Assistant Professor Brown University Department of Psychiatry Associate Chief, Mood Disorders
1 Alternative Treatments: Neuromodulation Approaches to Treatment Resistant Depression Audrey R. Tyrka, MD, PhD Assistant Professor Brown University Department of Psychiatry Associate Chief, Mood Disorders
TRANSCRANIAL MAGNETIC STIMULATION
 TRANSCRANIAL MAGNETIC STIMULATION Optum Behavioral Clinical Policy Policy Number: BH727BCPTMS_112017 Effective Date: November, 2017 Table of Contents Page BENEFIT CONSIDERATIONS... 1 COVERAGE RATIONALE...
TRANSCRANIAL MAGNETIC STIMULATION Optum Behavioral Clinical Policy Policy Number: BH727BCPTMS_112017 Effective Date: November, 2017 Table of Contents Page BENEFIT CONSIDERATIONS... 1 COVERAGE RATIONALE...
Rituxan Hycela (rituximab and hyaluronidase human) (Subcutaneous)
 Rituxan Hycela (rituximab and hyaluronidase human) (Subcutaneous) Document Number: IC-0322 Last Review Date: 02/06/2018 Date of Origin: 7/20/2010 Dates Reviewed: 09/2010, 12/2010, 02/2011, 03/2011, 05/2011,
Rituxan Hycela (rituximab and hyaluronidase human) (Subcutaneous) Document Number: IC-0322 Last Review Date: 02/06/2018 Date of Origin: 7/20/2010 Dates Reviewed: 09/2010, 12/2010, 02/2011, 03/2011, 05/2011,
Repetitive Transcranial Magnetic Stimulation (rtms)
 Page 1 of 33 Medical Policies - Mental Health Repetitive Transcranial Magnetic Stimulation (rtms) Print Number: PSY301.015 Effective Date: 07-01-2014 Coverage: Initial rtms Treatment Repetitive transcranial
Page 1 of 33 Medical Policies - Mental Health Repetitive Transcranial Magnetic Stimulation (rtms) Print Number: PSY301.015 Effective Date: 07-01-2014 Coverage: Initial rtms Treatment Repetitive transcranial
Frequently Asked Questions FAQS. NeuroStar TMS Therapies
 Frequently Asked Questions FAQS NeuroStar TMS Therapies Provided by Dr Terrence A. Boyadjis MD 790 E Market Street Suite 245 West Chester, PA 19382 610.738.9576 FAQS About TMS Therapies Page 1 NeuroStar
Frequently Asked Questions FAQS NeuroStar TMS Therapies Provided by Dr Terrence A. Boyadjis MD 790 E Market Street Suite 245 West Chester, PA 19382 610.738.9576 FAQS About TMS Therapies Page 1 NeuroStar
Clinical Policy: Transcranial Magnetic Stimulation
 Clinical Policy: Reference Number: CP.MP.172 Last Review Date: 12/18 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and legal information. Description
Clinical Policy: Reference Number: CP.MP.172 Last Review Date: 12/18 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and legal information. Description
Subject: Transcranial Magnetic Stimulation
 01-93875-18 Original Effective Date: 06/15/02 Reviewed: 12/06/18 Revised: 12/15/18 Subject: Transcranial Magnetic Stimulation THIS MEDICAL COVERAGE GUIDELINE IS NOT AN AUTHORIZATION, CERTIFICATION, EXPLANATION
01-93875-18 Original Effective Date: 06/15/02 Reviewed: 12/06/18 Revised: 12/15/18 Subject: Transcranial Magnetic Stimulation THIS MEDICAL COVERAGE GUIDELINE IS NOT AN AUTHORIZATION, CERTIFICATION, EXPLANATION
High Frequency Chest Wall Oscillating Devices (HFCWO) (Airway Clearance Systems)
 High Frequency Chest Wall Oscillating Devices (HFCWO) (Airway Clearance Systems) Date of Origin: 05/2015 Last Review Date: 07/26/2017 Effective Date: 07/26/2017 Dates Reviewed: 07/2016 Developed By: Medical
High Frequency Chest Wall Oscillating Devices (HFCWO) (Airway Clearance Systems) Date of Origin: 05/2015 Last Review Date: 07/26/2017 Effective Date: 07/26/2017 Dates Reviewed: 07/2016 Developed By: Medical
Document Number: IC I. Length of Authorization. Dosing Limits
 Hyaluronic Acid Derivatives: Durolane, Euflexxa, Gel-One, GelSyn-3, GenVisc 850, Hyalgan, Hymovis, Monovisc, Orthovisc, Supartz/Supartz FX, Synojoynt, Synvisc, & Synvisc-One, TriVisc, Visco-3 (Intra-articular)
Hyaluronic Acid Derivatives: Durolane, Euflexxa, Gel-One, GelSyn-3, GenVisc 850, Hyalgan, Hymovis, Monovisc, Orthovisc, Supartz/Supartz FX, Synojoynt, Synvisc, & Synvisc-One, TriVisc, Visco-3 (Intra-articular)
Continuous Glucose Monitoring (CGM)
 Continuous Glucose Monitoring (CGM) Date of Origin: 02/2001 Last Review Date: 08/22/2018 Effective Date: 08/22/2018 Dates Reviewed: 04/2004, 04/2005, 03/2006, 11/2006, 12/2007, 03/2008, 09/2008, 04/2009,
Continuous Glucose Monitoring (CGM) Date of Origin: 02/2001 Last Review Date: 08/22/2018 Effective Date: 08/22/2018 Dates Reviewed: 04/2004, 04/2005, 03/2006, 11/2006, 12/2007, 03/2008, 09/2008, 04/2009,
Transcranial Magnetic Stimulation (TMS)
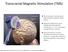 Transcranial Magnetic Stimulation (TMS) The treatment coil produces MRI-strength magnetic field pulses. Magnetic field pulses pass unimpeded through the cranium for 2-3 cm. and induce a small electric
Transcranial Magnetic Stimulation (TMS) The treatment coil produces MRI-strength magnetic field pulses. Magnetic field pulses pass unimpeded through the cranium for 2-3 cm. and induce a small electric
Cyramza (ramucirumab) (Intravenous)
 Cyramza (ramucirumab) (Intravenous) Document Number: IC 0199 Last Review Date: 5/1/2018 Date of Origin: 06/24/2014 Dates Reviewed: 09/2014, 01/2015, 05/2015, 11/2015, 04/2016, 08/2016, 11/2016, 05/2017,
Cyramza (ramucirumab) (Intravenous) Document Number: IC 0199 Last Review Date: 5/1/2018 Date of Origin: 06/24/2014 Dates Reviewed: 09/2014, 01/2015, 05/2015, 11/2015, 04/2016, 08/2016, 11/2016, 05/2017,
Case 2:15-cv MWF-JPR Document Filed 03/20/17 Page 1 of 36 Page ID #:388 EXHIBIT 1
 Case 2:15-cv-07074-MWF-JPR Document 65-10 Filed 03/20/17 Page 1 of 36 Page ID #:388 EXHIBIT 1 Case 2:15-cv-07074-MWF-JPR Document 65-10 Filed 03/20/17 Page 2 of 36 Page ID #:389 Cigna Medical Coverage
Case 2:15-cv-07074-MWF-JPR Document 65-10 Filed 03/20/17 Page 1 of 36 Page ID #:388 EXHIBIT 1 Case 2:15-cv-07074-MWF-JPR Document 65-10 Filed 03/20/17 Page 2 of 36 Page ID #:389 Cigna Medical Coverage
MEDICAL POLICY SUBJECT: TRANSCRANIAL MAGNETIC STIMULATION
 MEDICAL POLICY SUBJECT: TRANSCRANIAL MAGNETIC CATEGORY: Technology Assessment 12/18/14, 10/15/15, 12/15/16, 12/21/17 PAGE: 1 OF: 14 If a product excludes coverage for a service, it is not covered, and
MEDICAL POLICY SUBJECT: TRANSCRANIAL MAGNETIC CATEGORY: Technology Assessment 12/18/14, 10/15/15, 12/15/16, 12/21/17 PAGE: 1 OF: 14 If a product excludes coverage for a service, it is not covered, and
Vagus Nerve Stimulation (VNS)
 Vagus Nerve Stimulation (VNS) Date of Origin: 12/2008 Last Review Date: 06/27/2018 Effective Date: 07/01/2018 Dates Reviewed: 07/2010, 07/2011, 07/2012, 05/2013, 07/2014, 01/2016, 01/2017, 06/2018 Developed
Vagus Nerve Stimulation (VNS) Date of Origin: 12/2008 Last Review Date: 06/27/2018 Effective Date: 07/01/2018 Dates Reviewed: 07/2010, 07/2011, 07/2012, 05/2013, 07/2014, 01/2016, 01/2017, 06/2018 Developed
Medical Policy An Independent Licensee of the Blue Cross and Blue Shield Association
 of Depression and Other Psychiatric Disorders Page 1 of 26 Medical Policy An Independent Licensee of the Blue Cross and Blue Shield Association Title: See also: Transcranial Magnetic Stimulation (TMS)
of Depression and Other Psychiatric Disorders Page 1 of 26 Medical Policy An Independent Licensee of the Blue Cross and Blue Shield Association Title: See also: Transcranial Magnetic Stimulation (TMS)
NOW I M A NEUROSTAR. Let Your Best Self Shine
 NOW I M A NEUROSTAR. Let Your Best Self Shine It s Time for a New You NeuroStar Advanced Therapy is an in-office treatment that requires no anesthesia or sedation. You are awake and alert during treatment
NOW I M A NEUROSTAR. Let Your Best Self Shine It s Time for a New You NeuroStar Advanced Therapy is an in-office treatment that requires no anesthesia or sedation. You are awake and alert during treatment
Nonpharmacologic Interventions for Treatment-Resistant Depression. Public Meeting December 9, 2011
 Nonpharmacologic Interventions for Treatment-Resistant Depression Public Meeting December 9, 2011 New England CEPAC Goal: To improve the application of evidence to guide practice and policy in New England
Nonpharmacologic Interventions for Treatment-Resistant Depression Public Meeting December 9, 2011 New England CEPAC Goal: To improve the application of evidence to guide practice and policy in New England
Colony Stimulating Factors: Zarxio (filgrastim sndz) (Subcutaneous/Intravenous)
 Colony Stimulating Factors: Zarxio (filgrastim sndz) (Subcutaneous/Intravenous) Document Number: IC 0245 Last Review Date: 5/1/2018 Date of Origin: 03/31/2015 Dates Reviewed: 03/2015, 05/2015, 08/2015,
Colony Stimulating Factors: Zarxio (filgrastim sndz) (Subcutaneous/Intravenous) Document Number: IC 0245 Last Review Date: 5/1/2018 Date of Origin: 03/31/2015 Dates Reviewed: 03/2015, 05/2015, 08/2015,
Oscar G. Morales. MD Founding Director McLean Hospital TMS
 Institute of Medicine of the National Academies Non-Invasive Neuromodulation of the Central Nervous System: A Workshop Washington, DC. March 2 and 3, 2015 Session IV: Reimbursement Oscar G. Morales. MD
Institute of Medicine of the National Academies Non-Invasive Neuromodulation of the Central Nervous System: A Workshop Washington, DC. March 2 and 3, 2015 Session IV: Reimbursement Oscar G. Morales. MD
Treatment or Removal of Benign Skin Lesions
 Treatment or Removal of Benign Skin Lesions Date of Origin: 10/26/2016 Last Review Date: 12/15/2017 Effective Date: 10/25/2017 Dates Reviewed: 10/2016, 10/2017, 12/15/2017 Developed By: Medical Necessity
Treatment or Removal of Benign Skin Lesions Date of Origin: 10/26/2016 Last Review Date: 12/15/2017 Effective Date: 10/25/2017 Dates Reviewed: 10/2016, 10/2017, 12/15/2017 Developed By: Medical Necessity
Transcranial Magnetic Stimulation: Scientific Underpinnings and Practical Applications
 Transcranial Magnetic Stimulation: Scientific Underpinnings and Practical Applications Jon Draud, MS, MD Clinical Professor of Psychiatry University of Tennessee College of Medicine Memphis, Tennessee
Transcranial Magnetic Stimulation: Scientific Underpinnings and Practical Applications Jon Draud, MS, MD Clinical Professor of Psychiatry University of Tennessee College of Medicine Memphis, Tennessee
Trelstar (triptorelin) (Intramuscular)
 Trelstar (triptorelin) (Intramuscular) Last Review Date: 02/06/2018 Date of Origin: 11/28/2011 Document Number: IC-0131 Dates Reviewed: 12/2011, 03/2012, 06/19/2012, 09/2012, 12/2012, 03/2013, 06/2013,
Trelstar (triptorelin) (Intramuscular) Last Review Date: 02/06/2018 Date of Origin: 11/28/2011 Document Number: IC-0131 Dates Reviewed: 12/2011, 03/2012, 06/19/2012, 09/2012, 12/2012, 03/2013, 06/2013,
Cardiac Disease Screening Lipid Profile
 Cardiac Disease Screening Lipid Profile Date of Origin: 04/2003 Last Review Date: 02/27/2019 Effective Date: 03/01/2019 Dates Reviewed: 01/2004, 04/2005, 12/2005, 06/2006, 06/2007, 07/2008, 6/2011, 05/2012,
Cardiac Disease Screening Lipid Profile Date of Origin: 04/2003 Last Review Date: 02/27/2019 Effective Date: 03/01/2019 Dates Reviewed: 01/2004, 04/2005, 12/2005, 06/2006, 06/2007, 07/2008, 6/2011, 05/2012,
Colony Stimulating Factors: Nivestym (filgrastim-aafi) (Subcutaneous/Intravenous)
 Colony Stimulating Factors: Nivestym (filgrastim-aafi) (Subcutaneous/Intravenous) Document Number: MODA-0375 Last Review Date: 08/06/2018 Date of Origin: 08/06/2018 Dates Reviewed: 08/2018 I. Length of
Colony Stimulating Factors: Nivestym (filgrastim-aafi) (Subcutaneous/Intravenous) Document Number: MODA-0375 Last Review Date: 08/06/2018 Date of Origin: 08/06/2018 Dates Reviewed: 08/2018 I. Length of
FDA CLEARS NEUROSTAR TMS THERAPY FOR THE TREATMENT OF DEPRESSION
 FOR IMMEDIATE RELEASE FDA CLEARS NEUROSTAR TMS THERAPY FOR THE TREATMENT OF DEPRESSION First and Only Non-systemic and Non-invasive Treatment Cleared for Patients Who Have Not Benefited From Prior Antidepressant
FOR IMMEDIATE RELEASE FDA CLEARS NEUROSTAR TMS THERAPY FOR THE TREATMENT OF DEPRESSION First and Only Non-systemic and Non-invasive Treatment Cleared for Patients Who Have Not Benefited From Prior Antidepressant
LEASE DO NOT COPY. Setting up atms Clinic
 Setting up atms Clinic Daniel Press, M.D. Assistant Professor in Neurology, Harvard Medical School and Beth Israel Deaconess Medical Center Contents Safety and training of personnel Equipment Certification
Setting up atms Clinic Daniel Press, M.D. Assistant Professor in Neurology, Harvard Medical School and Beth Israel Deaconess Medical Center Contents Safety and training of personnel Equipment Certification
Transcranial Magnetic Stimulation and Cranial Electrical Stimulation (CES) as a Treatment of Depression and Other Psychiatric/Neurologic Disorders
 Transcranial Magnetic Stimulation and Cranial Electrical Stimulation (CES) as a Treatment of Depression and Other Psychiatric/Neurologic Disorders Policy Number: 2.01.50 Last Review: 10/2014 Origination:
Transcranial Magnetic Stimulation and Cranial Electrical Stimulation (CES) as a Treatment of Depression and Other Psychiatric/Neurologic Disorders Policy Number: 2.01.50 Last Review: 10/2014 Origination:
Patient Manual Brainsway Deep Transcranial Magnetic Stimulation (Deep TMS) System for Treatment of Major Depressive Disorder
 Dr. Zahida Tayyib www.mvptms.com Patient Manual Brainsway Deep Transcranial Magnetic Stimulation (Deep TMS) System for Treatment of Major Depressive Disorder If you are considering Brainsway Deep treatment
Dr. Zahida Tayyib www.mvptms.com Patient Manual Brainsway Deep Transcranial Magnetic Stimulation (Deep TMS) System for Treatment of Major Depressive Disorder If you are considering Brainsway Deep treatment
Transcranial Magnetic Stimulation as a Treatment of Depression and Other Psychiatric/Neurologic Disorders Corporate Medical Policy
 Transcranial Magnetic Stimulation as a Treatment of Depression and Other Psychiatric/Neurologic Disorders Corporate Medical Policy File name: Transcranial Magnetic Stimulation as a Treatment of Depression
Transcranial Magnetic Stimulation as a Treatment of Depression and Other Psychiatric/Neurologic Disorders Corporate Medical Policy File name: Transcranial Magnetic Stimulation as a Treatment of Depression
Reduction Mammoplasty
 Reduction Mammoplasty Date of Origin: 02/1999 Last Review Date: 06/28/2017 Effective Date: 06/28/2017 Dates Reviewed: 05/1999, 10/2000, 09/2001, 03/2002, 05/2002, 08/2002, 10/2003, 10/2004, 09/2005, 11/2005,
Reduction Mammoplasty Date of Origin: 02/1999 Last Review Date: 06/28/2017 Effective Date: 06/28/2017 Dates Reviewed: 05/1999, 10/2000, 09/2001, 03/2002, 05/2002, 08/2002, 10/2003, 10/2004, 09/2005, 11/2005,
Cardiac Disease Screening Lipid Profile
 Cardiac Disease Screening Lipid Profile Date of Origin: 04/2003 Last Review Date: 08/23/2017 Effective Date: 08/23/2017 Dates Reviewed: 01/2004, 04/2005, 12/2005, 06/2006, 06/2007, 07/2008, 6/2011, 05/2012,
Cardiac Disease Screening Lipid Profile Date of Origin: 04/2003 Last Review Date: 08/23/2017 Effective Date: 08/23/2017 Dates Reviewed: 01/2004, 04/2005, 12/2005, 06/2006, 06/2007, 07/2008, 6/2011, 05/2012,
DEEP TMS TREATMENT CONSENT FORM
 DEEP TMS TREATMENT CONSENT FORM My doctor has recommended that I receive treatment with Brainsway Deep TMS. The nature of this treatment, including the benefits and risks that I may experience, has been
DEEP TMS TREATMENT CONSENT FORM My doctor has recommended that I receive treatment with Brainsway Deep TMS. The nature of this treatment, including the benefits and risks that I may experience, has been
Ilaris (canakinumab) (Subcutaneous)
 Ilaris (canakinumab) (Subcutaneous) Last Review Date: 08/02/2018 Date of Origin: 11/07/2013 Dates Reviewed: 08/2014, 07/2015, 07/2016, 10/2016, 10/2017, 08/2018 Document Number: IC-0177 I. Length of Authorization
Ilaris (canakinumab) (Subcutaneous) Last Review Date: 08/02/2018 Date of Origin: 11/07/2013 Dates Reviewed: 08/2014, 07/2015, 07/2016, 10/2016, 10/2017, 08/2018 Document Number: IC-0177 I. Length of Authorization
Setting up atms Clinic. Daniel Press, M.D. Assistant Professor in Neurology, Harvard Medical School and Beth Israel Deaconess Medical Center
 Setting up atms Clinic Daniel Press, M.D. Assistant Professor in Neurology, Harvard Medical School and Beth Israel Deaconess Medical Center Contents Safety and training of personnel Equipment Certification
Setting up atms Clinic Daniel Press, M.D. Assistant Professor in Neurology, Harvard Medical School and Beth Israel Deaconess Medical Center Contents Safety and training of personnel Equipment Certification
Trelstar Depot (triptorelin)
 Dates Reviewed: 12/13/2011, 03/2012, 06/19/2012, 09/06/2012, 12/06/2012, 03/07/2013, 06/06/2013, Date of Origin: 11/28/2011 09/05/2013, 12/05/2013, 03/25/2014 Prior Auth Available: Post-service edit: The
Dates Reviewed: 12/13/2011, 03/2012, 06/19/2012, 09/06/2012, 12/06/2012, 03/07/2013, 06/06/2013, Date of Origin: 11/28/2011 09/05/2013, 12/05/2013, 03/25/2014 Prior Auth Available: Post-service edit: The
ADMINISTRATIVE POLICY AND PROCEDURE
 Page 1 of 6 SECTION: Medical SUBJECT: Electroconvulsive Therapy (ECT) DATE OF ORIGIN: 10/1/96 REVIEW DATES: 7/1/98, 10/1/99, 7/1/02, 7/1/04, 10/1/05, 5/1/09, 1/3/13, 7/1/15, 8/1/16 EFFECTIVE DATE: 3/24/17
Page 1 of 6 SECTION: Medical SUBJECT: Electroconvulsive Therapy (ECT) DATE OF ORIGIN: 10/1/96 REVIEW DATES: 7/1/98, 10/1/99, 7/1/02, 7/1/04, 10/1/05, 5/1/09, 1/3/13, 7/1/15, 8/1/16 EFFECTIVE DATE: 3/24/17
Patient Education Brief. NeuroStar TMS Therapies
 Patient Education Brief NeuroStar TMS Therapies Provided by Dr Terrence A. Boyadjis MD 790 E Market Street Suite 245 West Chester, PA 19382 610.738.9576 About Depression Depression is a serious medical
Patient Education Brief NeuroStar TMS Therapies Provided by Dr Terrence A. Boyadjis MD 790 E Market Street Suite 245 West Chester, PA 19382 610.738.9576 About Depression Depression is a serious medical
Erythropoiesis Stimulating Agents (ESAs): Aranesp (darbepoetin alfa) (Subcutaneous/Intravenous) *NON DIALYSIS* Document Number: IC 0242
 Erythropoiesis Stimulating Agents (ESAs): Aranesp (darbepoetin alfa) (Subcutaneous/Intravenous) *NON DIALYSIS* Document Number: IC 0242 Last Review Date: 05/01/2018 Date of Origin: 10/17/2008 Dates Reviewed:
Erythropoiesis Stimulating Agents (ESAs): Aranesp (darbepoetin alfa) (Subcutaneous/Intravenous) *NON DIALYSIS* Document Number: IC 0242 Last Review Date: 05/01/2018 Date of Origin: 10/17/2008 Dates Reviewed:
Alimta (pemetrexed) Document Number: IC 0007
 Alimta (pemetrexed) Document Number: IC 0007 Last Review Date: 05/01/2018 Date of Origin: 07/20/2010 Dates Reviewed: 09/2010, 12/2010, 03/2011, 06/2011,0 9/2011, 12/2011, 03/2012, 06/2012, 09/2012, 12/2012,
Alimta (pemetrexed) Document Number: IC 0007 Last Review Date: 05/01/2018 Date of Origin: 07/20/2010 Dates Reviewed: 09/2010, 12/2010, 03/2011, 06/2011,0 9/2011, 12/2011, 03/2012, 06/2012, 09/2012, 12/2012,
What is Repetitive Transcranial Magnetic Stimulation?
 rtms for Refractory Depression: Findings and Future Jonathan Downar, MD PhD Asst Professor, Dept of Psychiatry University of Toronto, Canada Co-Director, rtms Clinic Toronto Western Hospital University
rtms for Refractory Depression: Findings and Future Jonathan Downar, MD PhD Asst Professor, Dept of Psychiatry University of Toronto, Canada Co-Director, rtms Clinic Toronto Western Hospital University
Administration of repetitive transcranial magnetic stimulation (rtms)
 Professional Practice Guideline 16 Administration of repetitive transcranial magnetic stimulation (rtms) November 2018 Authorising Body: Responsible Committee/Department: Responsible Department: Document
Professional Practice Guideline 16 Administration of repetitive transcranial magnetic stimulation (rtms) November 2018 Authorising Body: Responsible Committee/Department: Responsible Department: Document
Brain Stimulation. Berry S. Anderson, PhD, RN Mary Rosedale, PhD, PMHNP-BC, NEA-BC Theresa Kormos, PMHCNS-BC Cindy Brown, BSN, RN
 Brain Stimulation American Psychiatric Nurses Association 24th Annual Conference October 16, 2010 Kentucky International Convention Center Louisville, Kentucky 1 Berry S. Anderson, PhD, RN Mary Rosedale,
Brain Stimulation American Psychiatric Nurses Association 24th Annual Conference October 16, 2010 Kentucky International Convention Center Louisville, Kentucky 1 Berry S. Anderson, PhD, RN Mary Rosedale,
Surgical Treatment for Achalasia
 Surgical Treatment for Achalasia Date of Origin: 08/2015 Last Review Date: 08/23/2017 Effective Date: 08/23/2017 Dates Reviewed: 08/2016, 08/2017 Developed By: Medical Necessity Criteria Committee I. For
Surgical Treatment for Achalasia Date of Origin: 08/2015 Last Review Date: 08/23/2017 Effective Date: 08/23/2017 Dates Reviewed: 08/2016, 08/2017 Developed By: Medical Necessity Criteria Committee I. For
Transcranial magnetic stimulation in clinical practice
 BJPsych Advances (2016), vol. 22, 373 379 doi: 10.1192/apt.bp.115.015206 Transcranial magnetic stimulation in clinical practice Sheila Hardy, Lorraine Bastick, Alex O Neill-Kerr, Priyadharshini Sabesan,
BJPsych Advances (2016), vol. 22, 373 379 doi: 10.1192/apt.bp.115.015206 Transcranial magnetic stimulation in clinical practice Sheila Hardy, Lorraine Bastick, Alex O Neill-Kerr, Priyadharshini Sabesan,
Effective Date: July 27, 2016
 Medical Review Criteria Transcranial Magnetic Stimulation Effective Date: July 27, 2016 Subject: Transcranial Magnetic Stimulation Policy: An initial course of left prefrontal TMS 1 is covered for: Patients
Medical Review Criteria Transcranial Magnetic Stimulation Effective Date: July 27, 2016 Subject: Transcranial Magnetic Stimulation Policy: An initial course of left prefrontal TMS 1 is covered for: Patients
IEHP UM Subcommittee Approved Authorization Guidelines Electroconvulsive Therapy- ECT
 Electroconvulsive Therapy- ECT Policy: IEHP considers ECT medically necessary for members with the following disorders: 1. Unipolar and bipolar depression. 2. Bipolar mania. 3. Psychotic disorders including
Electroconvulsive Therapy- ECT Policy: IEHP considers ECT medically necessary for members with the following disorders: 1. Unipolar and bipolar depression. 2. Bipolar mania. 3. Psychotic disorders including
Setting up a TMS Treatment Program
 Setting up a TMS Treatment Program Alvaro Pascual-Leone, M.D., Ph.D. Professor in Neurology Harvard Medical School Beth Israel Deaconess Medical Center Daniel Cohen, M.D., M.M.Sc. Instructor in Neurology
Setting up a TMS Treatment Program Alvaro Pascual-Leone, M.D., Ph.D. Professor in Neurology Harvard Medical School Beth Israel Deaconess Medical Center Daniel Cohen, M.D., M.M.Sc. Instructor in Neurology
HHS Public Access Author manuscript J Neuropsychiatry Clin Neurosci. Author manuscript; available in PMC 2017 September 11.
 HHS Public Access Author manuscript Published in final edited form as: J Neuropsychiatry Clin Neurosci. 2017 ; 29(2): 179 182. doi:10.1176/appi.neuropsych.16100181. Initial Response to Transcranial Magnetic
HHS Public Access Author manuscript Published in final edited form as: J Neuropsychiatry Clin Neurosci. 2017 ; 29(2): 179 182. doi:10.1176/appi.neuropsych.16100181. Initial Response to Transcranial Magnetic
The Clinical Application of Transcranial Magnetic Stimulation
 The Clinical Application of Transcranial Magnetic Stimulation Todd M. Hutton, MD, FAPA ABSTRACT Transcranial magnetic stimulation (TMS) provides psychiatrists with a new treatment offering in clinical
The Clinical Application of Transcranial Magnetic Stimulation Todd M. Hutton, MD, FAPA ABSTRACT Transcranial magnetic stimulation (TMS) provides psychiatrists with a new treatment offering in clinical
Repetitive transcranial magnetic stimulation for depression
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Repetitive transcranial magnetic stimulation for depression Depression causes low mood or sadness that can
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Repetitive transcranial magnetic stimulation for depression Depression causes low mood or sadness that can
Efficacy and Safety of Transcranial Magnetic Stimulation in Patients with Depression
 Showa Univ J Med Sci 30 1, 97 106, March 2018 Original Efficacy and Safety of Transcranial Magnetic Stimulation in Patients with Depression Yu KAWAGUCHI 1 and Akira IWANAMI 2 Abstract : Transcranial magnetic
Showa Univ J Med Sci 30 1, 97 106, March 2018 Original Efficacy and Safety of Transcranial Magnetic Stimulation in Patients with Depression Yu KAWAGUCHI 1 and Akira IWANAMI 2 Abstract : Transcranial magnetic
Bringing. to patients with depression
 Bringing to patients with depression Hope is knowing remission from depression is possible Depression is a serious illness that affects approximately 350 million people worldwide. 1 While medications may
Bringing to patients with depression Hope is knowing remission from depression is possible Depression is a serious illness that affects approximately 350 million people worldwide. 1 While medications may
2018 Otsuka Pharmaceutical Development & Commercialization, Inc., Rockville, MD Lundbeck, LLC.
 Neuromodulation Techniques: State of the Science Philip G. Janicak, MD Adjunct Professor Department of Psychiatry and Behavioral Sciences, Northwestern University Feinberg School of Medicine Consultant
Neuromodulation Techniques: State of the Science Philip G. Janicak, MD Adjunct Professor Department of Psychiatry and Behavioral Sciences, Northwestern University Feinberg School of Medicine Consultant
Transcranial magnetic stimulation has become an increasingly
 Within-Session Mood Changes From TMS in Depressed Patients Tobias Dang, M.D. David H. Avery, M.D. Joan Russo, Ph.D. Transcranial magnetic stimulation has become an increasingly well studied tool in neuropsychiatry.
Within-Session Mood Changes From TMS in Depressed Patients Tobias Dang, M.D. David H. Avery, M.D. Joan Russo, Ph.D. Transcranial magnetic stimulation has become an increasingly well studied tool in neuropsychiatry.
Therapeutic Neuromodulation: Overview of a Novel Treatment Platform
 Therapeutic Neuromodulation: Overview of a Novel Treatment Platform David G. Brock, MD; and Mark A. Demitrack, MD Shutterstock David G. Brock, MD, is Medical Director, Neuronetics, Inc. Mark A. Demitrack,
Therapeutic Neuromodulation: Overview of a Novel Treatment Platform David G. Brock, MD; and Mark A. Demitrack, MD Shutterstock David G. Brock, MD, is Medical Director, Neuronetics, Inc. Mark A. Demitrack,
TRANSCRANIAL MAGNETIC STIMULATION: CLINICAL UPDATE FOR PSYCHIATRIC APPLICATIONS ANNA MAZUR, PH.D. OSU DEPT. OF PSYCHIATRY AND BEHAVIORAL SCIENCES
 TRANSCRANIAL MAGNETIC STIMULATION: CLINICAL UPDATE FOR PSYCHIATRIC APPLICATIONS ANNA MAZUR, PH.D. OSU DEPT. OF PSYCHIATRY AND BEHAVIORAL SCIENCES FINANCIAL DISCLOSURE I have financial relationships to
TRANSCRANIAL MAGNETIC STIMULATION: CLINICAL UPDATE FOR PSYCHIATRIC APPLICATIONS ANNA MAZUR, PH.D. OSU DEPT. OF PSYCHIATRY AND BEHAVIORAL SCIENCES FINANCIAL DISCLOSURE I have financial relationships to
Review Paper. Introduction
 Review Paper Unilateral and bilateral repetitive transcranial magnetic stimulation for treatment-resistant depression: a meta-analysis of randomized controlled trials over decades Shayan Sehatzadeh, MD,
Review Paper Unilateral and bilateral repetitive transcranial magnetic stimulation for treatment-resistant depression: a meta-analysis of randomized controlled trials over decades Shayan Sehatzadeh, MD,
Review. M. Gross 1, L. Nakamura 1, A. Pascual-Leone 2 F. Fregni 2 1 Department of Psychiatry, University of S¼o Paulo, S¼o
 Acta Psychiatr Scand 2007: 116: 165 173 All rights reserved DOI: 10.1111/j.1600-0447.2007.01049.x Copyright Ó 2007 The Authors Journal Compilation Ó 2007 Blackwell Munksgaard ACTA PSYCHIATRICA SCANDINAVICA
Acta Psychiatr Scand 2007: 116: 165 173 All rights reserved DOI: 10.1111/j.1600-0447.2007.01049.x Copyright Ó 2007 The Authors Journal Compilation Ó 2007 Blackwell Munksgaard ACTA PSYCHIATRICA SCANDINAVICA
POLICY TITLE: Transcranial Magnetic Stimulation (TMS)
 Departmental Policy POLICY NO.: 200.02.101P POLICY TITLE: Transcranial Magnetic Stimulation (TMS) Submitted by: Daniel Castellanos, MD Title: Founding Chair, Department of Psychiatry & Behavioral Health
Departmental Policy POLICY NO.: 200.02.101P POLICY TITLE: Transcranial Magnetic Stimulation (TMS) Submitted by: Daniel Castellanos, MD Title: Founding Chair, Department of Psychiatry & Behavioral Health
A Patient s Guide: Pursuing Health Care. Reimbursement. for NeuroStar TMS Therapy
 A Patient s Guide: Pursuing Health Care Reimbursement for NeuroStar TMS Therapy Partnering With Your Healthcare Provider Your healthcare provider is a medical professional who is trained to assess your
A Patient s Guide: Pursuing Health Care Reimbursement for NeuroStar TMS Therapy Partnering With Your Healthcare Provider Your healthcare provider is a medical professional who is trained to assess your
Remission From Depression Is Possible
 Remission From Depression Is Possible About Depression Over 350 million people worldwide suffer from depression 1 While medications may help manage symptoms of depression, many patients are not satisfied
Remission From Depression Is Possible About Depression Over 350 million people worldwide suffer from depression 1 While medications may help manage symptoms of depression, many patients are not satisfied
The Evidence Base for rtms Reimbursement
 The Evidence Base for rtms Reimbursement Bradley N. Gaynes, MD, MPH Professor of Psychiatry Associate Chair of Research Training and Education Institute of Medicine Workshop March 3, 2015 What do we know,
The Evidence Base for rtms Reimbursement Bradley N. Gaynes, MD, MPH Professor of Psychiatry Associate Chair of Research Training and Education Institute of Medicine Workshop March 3, 2015 What do we know,
Knee Cartilage Transplants
 Knee Cartilage Transplants Date of Origin: 3/2005 Last Review Date: 8/23/2017 Effective Date: 8/23/2017 Dates Reviewed: Developed By: Medical Necessity Criteria Committee I. Description Allograft transplants
Knee Cartilage Transplants Date of Origin: 3/2005 Last Review Date: 8/23/2017 Effective Date: 8/23/2017 Dates Reviewed: Developed By: Medical Necessity Criteria Committee I. Description Allograft transplants
Coverage Summary. Wound Treatments
 Coverage Summary Wound Treatments Policy Number: W-001 Products: UnitedHealthcare Medicare Advantage Plans Original Approval Date: 02/18/2009 Approved by: UnitedHealthcare Medicare Benefit Interpretation
Coverage Summary Wound Treatments Policy Number: W-001 Products: UnitedHealthcare Medicare Advantage Plans Original Approval Date: 02/18/2009 Approved by: UnitedHealthcare Medicare Benefit Interpretation
BRAIN STIMULATION AN ALTERNATIVE TO DRUG THERAPY IN MATERNAL DEPRESSION?
 BRAIN STIMULATION AN ALTERNATIVE TO DRUG THERAPY IN MATERNAL DEPRESSION? Kira Stein, MD Medical Director West Coast Life Center Sherman Oaks, California CA Maternal Mental Health Initiative - 2013 2013
BRAIN STIMULATION AN ALTERNATIVE TO DRUG THERAPY IN MATERNAL DEPRESSION? Kira Stein, MD Medical Director West Coast Life Center Sherman Oaks, California CA Maternal Mental Health Initiative - 2013 2013
BRAINSWAY DEEP TMS SYSTEM
 510(K) SUMMARY JAN 0 72013 BRAINSWAY DEEP TMS SYSTEM Applicant Name: Company Name: Address: 510(k) Number K122288 Brainsway Ltd. 19 Hartumn St. (Bynet Bldg) Har Hotzvim, Jerusalem, ISRAEL 91451 Tel: +972-2-5813
510(K) SUMMARY JAN 0 72013 BRAINSWAY DEEP TMS SYSTEM Applicant Name: Company Name: Address: 510(k) Number K122288 Brainsway Ltd. 19 Hartumn St. (Bynet Bldg) Har Hotzvim, Jerusalem, ISRAEL 91451 Tel: +972-2-5813
rtms Versus ECT The Future of Neuromodulation & Brain Stimulation Therapies
 rtms Versus ECT The Future of Neuromodulation & Brain Stimulation Therapies rtms Treatment Equipment rtms Equipment Positioning of coil ECT & rtms Innovative Treatment Options for Patients Only a fraction
rtms Versus ECT The Future of Neuromodulation & Brain Stimulation Therapies rtms Treatment Equipment rtms Equipment Positioning of coil ECT & rtms Innovative Treatment Options for Patients Only a fraction
MEDICAL POLICY EFFECTIVE DATE: 04/28/11 REVISED DATE: 04/26/12, 04/25/13, 04/24/14, 06/25/15, 06/22/16, 06/22/17
 MEDICAL POLICY SUBJECT: STANDARD DIALECTICAL BEHAVIOR A nonprofit independent licensee of the BlueCross BlueShield Association PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered,
MEDICAL POLICY SUBJECT: STANDARD DIALECTICAL BEHAVIOR A nonprofit independent licensee of the BlueCross BlueShield Association PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered,
Interventions for Relapsing Depression: TMS, ECT, and Ketamine
 Interventions for Relapsing Depression: TMS, ECT, and Ketamine MDD top cause of disability; suicide only top-10 cause of death currently increasing. 10-15% lifetime prevalence A third of patients do not
Interventions for Relapsing Depression: TMS, ECT, and Ketamine MDD top cause of disability; suicide only top-10 cause of death currently increasing. 10-15% lifetime prevalence A third of patients do not
Interventions for Relapsing Depression: TMS, ECT, and Ketamine
 Interventions for Relapsing Depression: TMS, ECT, and Ketamine MDD top cause of disability; suicide only top-10 cause of death currently increasing. 10-15% lifetime prevalence A third of patients do not
Interventions for Relapsing Depression: TMS, ECT, and Ketamine MDD top cause of disability; suicide only top-10 cause of death currently increasing. 10-15% lifetime prevalence A third of patients do not
The Next Chapter in Brain Stimulation Therapy
 The Next Chapter in Brain Stimulation Therapy Outline I. Depression and Treatment Resistant Depression Barriers to Treatment Treatment Options Cost and Impact of Depression II. Electroconvulsive Therapy
The Next Chapter in Brain Stimulation Therapy Outline I. Depression and Treatment Resistant Depression Barriers to Treatment Treatment Options Cost and Impact of Depression II. Electroconvulsive Therapy
TRANSCRANIAL MAGNETIC STIMULATION
 MEDICAL POLICY TRANSCRANIAL MAGNETIC STIMULATION Policy Number: 2013T0536F Effective Date: December 1, 2013 Table of Contents COVERAGE RATIONALE... BACKGROUND... CLINICAL EVIDENCE... U.S. FOOD AND DRUG
MEDICAL POLICY TRANSCRANIAL MAGNETIC STIMULATION Policy Number: 2013T0536F Effective Date: December 1, 2013 Table of Contents COVERAGE RATIONALE... BACKGROUND... CLINICAL EVIDENCE... U.S. FOOD AND DRUG
obsessive-compulsive disorder; OCD
 9(1), 50 56, 2017 OCD OCD DBSECT tdcsrtms DBS ECT 6 tdcs rtms obsessive-compulsive disorder; OCD 2013 OCD 2012 deep brain stimulation; DBS electroconvulsive therapy; ECT transcranial direct current stimulation;
9(1), 50 56, 2017 OCD OCD DBSECT tdcsrtms DBS ECT 6 tdcs rtms obsessive-compulsive disorder; OCD 2013 OCD 2012 deep brain stimulation; DBS electroconvulsive therapy; ECT transcranial direct current stimulation;
Prolia /Xgeva (denosumab) Document Number: IC-0098
 / (denosumab) Document Number: IC-0098 Last Review Date: 5/30/2017 Date of Origin: 11/28/2011 Dates Reviewed: 12/2011, 03/2012, 06/2012, 09/2012, 12/2012, 03/2013, 06/2013, 03/2014, 06/2014, 09/2014, 12/2014,
/ (denosumab) Document Number: IC-0098 Last Review Date: 5/30/2017 Date of Origin: 11/28/2011 Dates Reviewed: 12/2011, 03/2012, 06/2012, 09/2012, 12/2012, 03/2013, 06/2013, 03/2014, 06/2014, 09/2014, 12/2014,
Objectives. Objectives. A practice review. 02-Nov-16 MAJOR DEPRESSIVE DISORDER: NEW DEVELOPMENTS AND PRACTICAL IMPLICATIONS
 MAJOR DEPRESSIVE DISORDER: NEW DEVELOPMENTS AND PRACTICAL IMPLICATIONS Jon-Paul Khoo What is treatment resistance really? Database review 328 consecutive non-remitted MDD patients referred for private
MAJOR DEPRESSIVE DISORDER: NEW DEVELOPMENTS AND PRACTICAL IMPLICATIONS Jon-Paul Khoo What is treatment resistance really? Database review 328 consecutive non-remitted MDD patients referred for private
