RNA preparation from extracted paraffin cores:
|
|
|
- Elmer Rodgers
- 5 years ago
- Views:
Transcription
1 Supplementary methods, Nielsen et al., A comparison of PAM50 intrinsic subtyping with immunohistochemistry and clinical prognostic factors in tamoxifen-treated estrogen receptor positive breast cancer. RNA preparation from extracted paraffin cores: RNA was recovered using the High Pure RNA Paraffin Kit including an on-column DNase I treatment to remove any residual DNA (Roche Applied Science, Indianapolis IN). RNA yields were assessed using an ND-1000 Spectrophotometer (NanoDrop Technologies, Rockland DE), and samples used for analysis where the concentration of total RNA exceeded 25 ng/ul and yield exceed 1.2 ug. qrt-pcr: cdna synthesis was completed using a mixture of random hexamers and gene-specific primers, and qpcr was performed with the Roche LightCycler 480 instrument as previously described (1, 2). Each 384-well plate contained samples and a calibrator in triplicate with 2.5 ng cdna and 10 ng cdna, respectively per reaction. A tumor sample was considered of insufficient quality if any of the reference controls (ACTB, PSMC4, RPLP0, MRPL19, or SF3A1) failed. Since the expression of one of the 50 discriminator genes may not be detected by PCR due to a technical failure or due to the biology of the sample (i.e. that gene is not expressed by the tumor at a measureable level) we developed validation rules to distinguish between these possibilities. A technical failure occurred if 1) the calibrator failed in all replicates, 2) the calibrator Cps were out of range for that gene, or 3) all calibrators had secondary melting peaks greater than three-quarter the height of the specific melting peak. If the negative and calibrators passed, then a gene could be assigned a low copy value. The low copy was a Cp=40 for that gene. This value was assigned if either 1) all 3 sample replicates had Cp=0, or 2) all sample replicates had a Cp>35 with non-specific melting peaks, or 3) there was PCR amplification with a Cp>38. If both low copy and a measurable copy number value were assigned for sample replicates, then the measurable copy value was used. Subtype assignment by the 50 gene classifier Gene expression microarrays have been extensively used to characterize gene expression variation in breast cancer. The predominant variation has been characterized as 5 intrinsic subtypes of breast cancer that have been repeatedly observed (3-5). A 50 gene qrt-pcr assay has been constructed to provide stable and highly repeatable subtype classification in relevant tissue specimens (FFPE and frozen). A complete description of the development and initial evaluation of this assay was described in Parker et al.(1). Development of the classifier began with a set of over 200 samples with 20k expression measurements by microarray and 160 genes measured by qrt-pcr. The microarray data allowed identification of gold
2 standard specimens representing each of the five primary breast cancer subtypes. The set of 160 qrt- PCR genes was then optimized to identify a reduced subset of genes from which accurate subtype classification could still be performed. A set of 50 genes was finalized using cross-validation. The 50 genes, corresponding centroids, and other parameters of the nearest centroid breast cancer intrinsic subtype classifier are available in Parker et al. (1). The qrt-pcr assay consists of these 50 genes and an additional 5 housekeeper genes used for sample normalization. Analysis by qrt-pcr is performed by first normalizing the raw Ct values to gene specific technical controls followed by normalization to the sample controls (housekeepers) (6). The distance to each centroid is calculated using Spearman's rank correlation. The centroid associated with the largest positive correlation value is assigned as the subtype of the sample. This protocol produces reproducible subtype correlations and subtype assignment independently for each sample. Risk models of the 50 gene based subtype Subtype classification of a breast tumor may provide insight into the prognosis and illuminate potential treatment strategies. However, variation exists within each subtype, and this variation carries useful prognostic information. Thus the prognostic value of the gene expression information is not limited to the final subtype assignment. Development of the ROR risk of relapse models consisted of assignment of the five subtype correlations to each sample as described above. This was performed using set of node negative patients with no adjuvant systemic therapy. The four tumor subtype (normal-like excluded) correlations were related to relapse free survival as additive terms in a Cox model. Ridge regression was performed in the context of cross-validation to optimize the weights for each term. This model of the four tumor subtype correlations (equation 1) provides the subtype based risk of relapse (ROR-S): (equation 1) ROR-S = 0.05*Basal *Her2-0.34*LumA *LumB (1) Tumor size was found to be an independent prognostic factor and was included in the model as an additive term. Tumor size was limited to binary information ( <=2cm vs >2cm) as this was the data available in the training set. The weights were re-learned using the same process to produce a combined model of tumor size and subtype. This model was previously termed ROR-C (1) and is given in equation 2. The current manuscript has renamed this metric from ROR-C to ROR-T to distinguish this combined model from other combined models that are evaluated. (equation 2) ROR-T = ROR-C = 0.05*Basal *Her2-0.23*LumA *LumB *T (1) Additional models were explored using the same process as was used to develop these models. The process of model development and the training data for these novel models is identical to that described above (and more completely in (1)) for ROR-S and ROR-T. The motivation behind these models
3 was to incorporate proliferation information as an independent factor with respect to subtype. The 50 gene set contains many genes that are known markers of proliferation. A set of 11 proliferation genes (table M1) was highly correlated (r > 0.8) in the qrt-pcr and microarray training sets described in Parker et al [1]. Table M1 Genes in proliferation index GeneName UniGene EntrezGene BIRC5 Hs CCNB1 Hs CDC20 Hs NUF2 Hs CEP55 Hs NDC80 Hs MKI67 Hs PTTG1 Hs RRM2 Hs TYMS Hs UBE2C Hs These 11 genes were summarized by averaging the normalized expression estimates in each sample. This proliferation signature index was modeled as an additional term to the four subtypes to produce ROR-P (equation 3), and in addition to the model of four subtypes and tumor size to produce ROR-PT (equation 4). (equation 3) ROR-P = *Basal + 0.7*Her2-0.95*LumA *LumB *Prolif (equation 4) ROR-PT = *Basal *Her2-0.9*LumA *LumB *T *Prolif Thresholds for all four models were identified in the training set using the same process. The distribution of risk scores from each model was stratified by the subtype assignment. The low risk threshold for each model was assigned as the minimum of all Luminal B scores, and the high risk threshold was the maximum of all Luminal A scores. Luminal gene signature Evaluation of the information content of ER protein and ESR1 gene expression are compared to a summarized luminal gene expression signature. This gene expression signature was constructed in a similar fashion to the proliferation signature. Specifically, a set of 8 genes (table M2) was highly correlated (r > 0.8) in qrt-pcr and microarray training sets from Parker et al [1] and clearly
4 distinguished luminal tumors. These 8 genes were summarized by averaging the normalized expression estimates in each sample.
5 Table M2 Genes in luminal signature GeneName UniGene EntrezGene BAG1 Hs ESR1 Hs FOXA1 Hs GPR160 Hs NAT1 Hs MAPT Hs MLPH Hs PGR Hs Risk models based on standard clinical data and immunohistochemically-determined intrinsic subtype The accuracy of risk classification is an important measure for comparing the various models. Other variables such as standard clinical risk factors and immunohistochemistry (IHC) based subtype may also be compared in this context if appropriate models are available. Pathological grade and IHC based subtype assignments are correlated with the 50 gene based subtype assignments, and independent of pathological stage with respect to prognostic information (1, 7). Formal models of these variables are needed to evaluate which of these three biologic measures best compliments pathologic stage to produce the most accurate classification. The online decision making tool Adjuvant! Online ( was used to generate breast cancer specific survival (BCSS) estimates for each patient in this cohort (8). The clinical and pathologic factors used to generate the risk estimates were age, tumor size, tumor grade, ER status, number of positive lymph nodes, and lymphovascular invasion. A model of immunohistochemical subtype and tumor size (IHC-T) was constructed following the same process outlined for development of the ROR scores. Specifically, the two categorical variables were treated as additive terms in the Cox models, fit with Ridge regression, and optimized with crossvalidation. Cross-validation was performed by randomly selecting 2/3 of the samples for training with the remaining 1/3 used for validation. The process was repeated 100 times to evaluate model performance. IHC subtype definitions for Luminal A and B follow the protocol described in Cheang et al. (7), and incorporate the information from ER, PR, HER2 and Ki67 immunohistochemistry: Luminal A = (ER or PR) positive and HER2 negative and Ki67 <= 13%; Luminal B = (ER or PR) positive and (HER2 positive or Ki67 > 13%); HER2 = ER negative and PR negative and HER2 positive; basal = ER negative and PR negative and HER2 negative and (ck5/6 or EGFR) positive. The data set used for training these models has been previously described (6) and was derived concurrently with the test set data on the same tissue microarray series (making the technical aspects of immunohistochemistry performance and scoring identical between training and test set). This training
6 cohort consists of 1545 node negative samples from patients who received no adjuvant systemic therapy. Tumor size, IHC subtype, pathological grade, and lymphovascular invasion were significant predictors (p < 0.05) of relapse free survival in standard Cox multivariable analysis. Grade-T and IHC-T models were evaluated during cross-validation. As with the ROR models, relapse free survival, where any relapse was considered an event, was used for training. Accuracy estimates from cross-validation are illustrated in Figure M1. The final risk scores assigned to each possible level of a sample are provided in table M3. These values were assigned to the estrogen receptor positive, tamoxifen-treated series samples (described in the main paper) as appropriate for the comparative evaluations presented. Figure M1. Accuracy estimates from cross-validation of pathological models in the training set. The C-index was calculated during each of 100 rounds of cross-validation from the "left-out" node negative samples. The bar height represents the mean C-index and corresponding standard error. Table M3 IHC-T IHC subtype Tumor size Risk Score Luminal A <= 2cm 0 Luminal A > 2cm 0.23 Luminal B <= 2cm 0.4 Luminal B > 2cm 0.64 Her2 <= 2cm 0.44 Her2 > 2cm 0.67 Basal <= 2cm 0.15 Basal > 2cm 0.38
7 Supplementary References 1. Parker JS, Mullins M, Cheang MC, et al. Supervised risk predictor of breast cancer based on intrinsic subtypes. J Clin Oncol 2009;27: Mullins M, Perreard L, Quackenbush JF, et al. Agreement in breast cancer classification between microarray and quantitative reverse transcription PCR from fresh-frozen and formalin-fixed, paraffinembedded tissues. Clin Chem 2007;53: Perou CM, Sorlie T, Eisen MB, et al. Molecular portraits of human breast tumours. Nature 2000;406: Sorlie T, Tibshirani R, Parker J, et al. Repeated observation of breast tumor subtypes in independent gene expression data sets. Proc Natl Acad Sci U S A 2003;100: Hu Z, Fan C, Oh DS, et al. The molecular portraits of breast tumors are conserved across microarray platforms. BMC Genomics 2006;7: Perreard L, Fan C, Quackenbush JF, et al. Classification and risk stratification of invasive breast carcinomas using a real-time quantitative RT-PCR assay. Breast Cancer Res 2006;8:R Cheang MC, Voduc D, Bajdik C, et al. Basal-like breast cancer defined by five biomarkers has superior prognostic value than triple-negative phenotype. Clin Cancer Res 2008;14: Ravdin PM, Siminoff LA, Davis GJ, et al. Computer program to assist in making decisions about adjuvant therapy for women with early breast cancer. J Clin Oncol 2001;19:
Breast cancer classification: beyond the intrinsic molecular subtypes
 Breast cancer classification: beyond the intrinsic molecular subtypes Britta Weigelt, PhD Signal Transduction Laboratory CRUK London Research Institute Summary Breast cancer heterogeneity Molecular classification
Breast cancer classification: beyond the intrinsic molecular subtypes Britta Weigelt, PhD Signal Transduction Laboratory CRUK London Research Institute Summary Breast cancer heterogeneity Molecular classification
Ki67 Index, HER2 Status, and Prognosis of Patients With Luminal B Breast Cancer
 ARTICLE Ki67 Index, HER2 Status, and Prognosis of Patients With Breast Cancer Maggie C. U. Cheang, Stephen K. Chia, David Voduc, Dongxia Gao, Samuel Leung, Jacqueline Snider, Mark Watson, Sherri Davies,
ARTICLE Ki67 Index, HER2 Status, and Prognosis of Patients With Breast Cancer Maggie C. U. Cheang, Stephen K. Chia, David Voduc, Dongxia Gao, Samuel Leung, Jacqueline Snider, Mark Watson, Sherri Davies,
A new way of looking at breast cancer tumour biology
 A new way of looking at breast cancer tumour biology Contents Intrinsic subtypes of breast cancer 3 Gene expression assays 3 Basis of the Prosigna test 4 Information provided by Prosigna 5 The accuracy
A new way of looking at breast cancer tumour biology Contents Intrinsic subtypes of breast cancer 3 Gene expression assays 3 Basis of the Prosigna test 4 Information provided by Prosigna 5 The accuracy
Comparison of Triple Negative Breast Cancer between Asian and Western Data Sets
 2010 IEEE International Conference on Bioinformatics and Biomedicine Workshops Comparison of Triple Negative Breast Cancer between Asian and Western Data Sets Lee H. Chen Bioinformatics and Biostatistics
2010 IEEE International Conference on Bioinformatics and Biomedicine Workshops Comparison of Triple Negative Breast Cancer between Asian and Western Data Sets Lee H. Chen Bioinformatics and Biostatistics
Alternate Gene Signatures, or Not
 Charles M. Perou, Ph.D. Departments of Genetics and Pathology Lineberger Comprehensive Cancer Center University of North Carolina Chapel Hill, North Carolina, USA Alternate Gene Signatures, or Not Charles
Charles M. Perou, Ph.D. Departments of Genetics and Pathology Lineberger Comprehensive Cancer Center University of North Carolina Chapel Hill, North Carolina, USA Alternate Gene Signatures, or Not Charles
Immunohistochemical classification of breast tumours
 Immunohistochemical classification of breast tumours Workshop in Diagnostic Immunohistochemistry September 19 th - 21 th 2018 Anne-Vibeke Lænkholm Department of Surgical Pathology, Zealand University Hospital,
Immunohistochemical classification of breast tumours Workshop in Diagnostic Immunohistochemistry September 19 th - 21 th 2018 Anne-Vibeke Lænkholm Department of Surgical Pathology, Zealand University Hospital,
CLINICAL IMPLEMENTATION OF BREAST CANCER GENOMICS. Joel Stanley Parker
 CLINICAL IMPLEMENTATION OF BREAST CANCER GENOMICS Joel Stanley Parker A dissertation submitted to the faculty of the University of North Carolina at Chapel Hill in partial fulfillment of the requirements
CLINICAL IMPLEMENTATION OF BREAST CANCER GENOMICS Joel Stanley Parker A dissertation submitted to the faculty of the University of North Carolina at Chapel Hill in partial fulfillment of the requirements
Breast cancer: Molecular STAGING classification and testing. Korourian A : AP,CP ; MD,PHD(Molecular medicine)
 Breast cancer: Molecular STAGING classification and testing Korourian A : AP,CP ; MD,PHD(Molecular medicine) Breast Cancer Theory: Halsted Operative breast cancer is a local-regional disease The positive
Breast cancer: Molecular STAGING classification and testing Korourian A : AP,CP ; MD,PHD(Molecular medicine) Breast Cancer Theory: Halsted Operative breast cancer is a local-regional disease The positive
Contemporary Classification of Breast Cancer
 Contemporary Classification of Breast Cancer Laura C. Collins, M.D. Vice Chair of Anatomic Pathology Professor of Pathology Beth Israel Deaconess Medical Center and Harvard Medical School Boston, MA Outline
Contemporary Classification of Breast Cancer Laura C. Collins, M.D. Vice Chair of Anatomic Pathology Professor of Pathology Beth Israel Deaconess Medical Center and Harvard Medical School Boston, MA Outline
Development and verification of the PAM50-based Prosigna breast cancer gene signature assay
 Washington University School of Medicine Digital Commons@Becker Open Access Publications 2015 Development and verification of the PAM50-based Prosigna breast cancer gene signature assay Jacqueline Snider
Washington University School of Medicine Digital Commons@Becker Open Access Publications 2015 Development and verification of the PAM50-based Prosigna breast cancer gene signature assay Jacqueline Snider
Comparison of prognostic signatures for ER positive breast cancer in TransATAC:
 Comparison of prognostic signatures for ER positive breast cancer in TransATAC: EndoPredict, a high performance test in node negative and node positive disease Ivana Sestak, PhD Centre for Cancer Prevention
Comparison of prognostic signatures for ER positive breast cancer in TransATAC: EndoPredict, a high performance test in node negative and node positive disease Ivana Sestak, PhD Centre for Cancer Prevention
Gene Signatures in Breast Cancer: Moving Beyond ER, PR, and HER2? Lisa A. Carey, M.D. University of North Carolina USA
 Gene Signatures in Breast Cancer: Moving Beyond ER, PR, and HER2? Lisa A. Carey, M.D. University of North Carolina USA When Are Biomarkers Ready To Use? Same Rules for Gene Expression Panels Key elements
Gene Signatures in Breast Cancer: Moving Beyond ER, PR, and HER2? Lisa A. Carey, M.D. University of North Carolina USA When Are Biomarkers Ready To Use? Same Rules for Gene Expression Panels Key elements
Molecular in vitro diagnostic test for the quantitative detection of the mrna expression of ERBB2, ESR1, PGR and MKI67 in breast cancer tissue.
 Innovation for your breast cancer diagnostics PGR G ATA G C G A C G AT C G A A A G A A G T TA G ATA G C G A C G AT C G A A A G A A G T TA G ATA G C G A C G AT C G A A A G A A G T TA G ATA G C G A C ERBB2
Innovation for your breast cancer diagnostics PGR G ATA G C G A C G AT C G A A A G A A G T TA G ATA G C G A C G AT C G A A A G A A G T TA G ATA G C G A C G AT C G A A A G A A G T TA G ATA G C G A C ERBB2
FISH mcgh Karyotyping ISH RT-PCR. Expression arrays RNA. Tissue microarrays Protein arrays MS. Protein IHC
 Classification of Breast Cancer in the Molecular Era Susan J. Done University Health Network, Toronto Why classify? Prognosis Prediction of response to therapy Pathogenesis Invasive breast cancer can have
Classification of Breast Cancer in the Molecular Era Susan J. Done University Health Network, Toronto Why classify? Prognosis Prediction of response to therapy Pathogenesis Invasive breast cancer can have
FAQs for UK Pathology Departments
 FAQs for UK Pathology Departments This is an educational piece written for Healthcare Professionals FAQs for UK Pathology Departments If you would like to discuss any of the listed FAQs further, or have
FAQs for UK Pathology Departments This is an educational piece written for Healthcare Professionals FAQs for UK Pathology Departments If you would like to discuss any of the listed FAQs further, or have
Profili Genici e Personalizzazione del trattamento adiuvante nel carcinoma mammario G. RICCIARDI
 Profili Genici e Personalizzazione del trattamento adiuvante nel carcinoma mammario G. RICCIARDI UOC Oncologia Medica, A.O. Papardo, Messina Dir. Prof. V. Adamo BREAST CANCER Brain Adjuvant Medical Therapies
Profili Genici e Personalizzazione del trattamento adiuvante nel carcinoma mammario G. RICCIARDI UOC Oncologia Medica, A.O. Papardo, Messina Dir. Prof. V. Adamo BREAST CANCER Brain Adjuvant Medical Therapies
Roadmap for Developing and Validating Therapeutically Relevant Genomic Classifiers. Richard Simon, J Clin Oncol 23:
 Roadmap for Developing and Validating Therapeutically Relevant Genomic Classifiers. Richard Simon, J Clin Oncol 23:7332-7341 Presented by Deming Mi 7/25/2006 Major reasons for few prognostic factors to
Roadmap for Developing and Validating Therapeutically Relevant Genomic Classifiers. Richard Simon, J Clin Oncol 23:7332-7341 Presented by Deming Mi 7/25/2006 Major reasons for few prognostic factors to
Assessment of Risk Recurrence: Adjuvant Online, OncotypeDx & Mammaprint
 Assessment of Risk Recurrence: Adjuvant Online, OncotypeDx & Mammaprint William J. Gradishar, MD Professor of Medicine Robert H. Lurie Comprehensive Cancer Center of Northwestern University Classical
Assessment of Risk Recurrence: Adjuvant Online, OncotypeDx & Mammaprint William J. Gradishar, MD Professor of Medicine Robert H. Lurie Comprehensive Cancer Center of Northwestern University Classical
Relevancia práctica de la clasificación de subtipos intrínsecos en cáncer de mama Miguel Martín Instituto de Investigación Sanitaria Gregorio Marañón
 Relevancia práctica de la clasificación de subtipos intrínsecos en cáncer de mama Miguel Martín Instituto de Investigación Sanitaria Gregorio Marañón Universidad Complutense Madrid The new technologies
Relevancia práctica de la clasificación de subtipos intrínsecos en cáncer de mama Miguel Martín Instituto de Investigación Sanitaria Gregorio Marañón Universidad Complutense Madrid The new technologies
30 years of progress in cancer research
 Breast Cancer Molecular Knowledge Integrated in Clinical Practice Personalized Medicine Laura J. Esserman UCSF Comprehensive Cancer Center Retreat Breast Cancer Management Advances 80-90s 1) Screening
Breast Cancer Molecular Knowledge Integrated in Clinical Practice Personalized Medicine Laura J. Esserman UCSF Comprehensive Cancer Center Retreat Breast Cancer Management Advances 80-90s 1) Screening
Genomic Analyses across Six Cancer Types Identify Basal-like Breast Cancer as a Unique Molecular Entity
 Genomic Analyses across Six Cancer Types Identify Basal-like Breast Cancer as a Unique Molecular Entity Aleix Prat, Barbara Adamo, Cheng Fan, Vicente Peg, Maria Vidal, Patricia Galván, Ana Vivancos, Paolo
Genomic Analyses across Six Cancer Types Identify Basal-like Breast Cancer as a Unique Molecular Entity Aleix Prat, Barbara Adamo, Cheng Fan, Vicente Peg, Maria Vidal, Patricia Galván, Ana Vivancos, Paolo
Prosigna BREAST CANCER PROGNOSTIC GENE SIGNATURE ASSAY
 Prosigna BREAST CANCER PROGNOSTIC GENE SIGNATURE ASSAY Methodology The test is based on the reported 50-gene classifier algorithm originally named PAM50 and is performed on the ncounter Dx Analysis System
Prosigna BREAST CANCER PROGNOSTIC GENE SIGNATURE ASSAY Methodology The test is based on the reported 50-gene classifier algorithm originally named PAM50 and is performed on the ncounter Dx Analysis System
Prosigna BREAST CANCER PROGNOSTIC GENE SIGNATURE ASSAY
 Prosigna BREAST CANCER PROGNOSTIC GENE SIGNATURE ASSAY GENE EXPRESSION PROFILING WITH PROSIGNA What is Prosigna? Prosigna Breast Cancer Prognostic Gene Signature Assay is an FDA-approved assay which provides
Prosigna BREAST CANCER PROGNOSTIC GENE SIGNATURE ASSAY GENE EXPRESSION PROFILING WITH PROSIGNA What is Prosigna? Prosigna Breast Cancer Prognostic Gene Signature Assay is an FDA-approved assay which provides
Triple Negative Breast Cancer
 Triple Negative Breast Cancer Prof. Dr. Pornchai O-charoenrat Division of Head-Neck & Breast Surgery Department of Surgery Faculty of Medicine Siriraj Hospital Breast Cancer Classification Traditional
Triple Negative Breast Cancer Prof. Dr. Pornchai O-charoenrat Division of Head-Neck & Breast Surgery Department of Surgery Faculty of Medicine Siriraj Hospital Breast Cancer Classification Traditional
Role of Genomic Profiling in (Minimally) Node Positive Breast Cancer
 Role of Genomic Profiling in (Minimally) Node Positive Breast Cancer Kathy S. Albain, MD, FACP Professor of Medicine Dean s Scholar Loyola University Chicago Stritch School of Medicine Cardinal Bernardin
Role of Genomic Profiling in (Minimally) Node Positive Breast Cancer Kathy S. Albain, MD, FACP Professor of Medicine Dean s Scholar Loyola University Chicago Stritch School of Medicine Cardinal Bernardin
Implications of Progesterone Receptor Status for the Biology and Prognosis of Breast Cancers
 日大医誌 75 (1): 10 15 (2016) 10 Original Article Implications of Progesterone Receptor Status for the Biology and Prognosis of Breast Cancers Naotaka Uchida 1), Yasuki Matsui 1), Takeshi Notsu 1) and Manabu
日大医誌 75 (1): 10 15 (2016) 10 Original Article Implications of Progesterone Receptor Status for the Biology and Prognosis of Breast Cancers Naotaka Uchida 1), Yasuki Matsui 1), Takeshi Notsu 1) and Manabu
Key points about expression profile intrinsic subtypes. Analytically robust expression profiles from FFPE sections using Nanostring technology
 Analytically robust expression profiles from FFPE sections using Nanostring technology Torsten O. Nielsen, MD/PhD, FRCPC Professor of Pathology and Laboratory Medicine University of British Columbia, Vancouver,
Analytically robust expression profiles from FFPE sections using Nanostring technology Torsten O. Nielsen, MD/PhD, FRCPC Professor of Pathology and Laboratory Medicine University of British Columbia, Vancouver,
CHARLES M. PEROU. The University of North Carolina at Chapel Hill, Chapel Hill, North Carolina, USA
 The Oncologist Molecular Stratification of Triple-Negative Breast Cancers CHARLES M. PEROU The University of North Carolina at Chapel Hill, Chapel Hill, North Carolina, USA Key Words. Triple-negative breast
The Oncologist Molecular Stratification of Triple-Negative Breast Cancers CHARLES M. PEROU The University of North Carolina at Chapel Hill, Chapel Hill, North Carolina, USA Key Words. Triple-negative breast
Reporting of Breast Cancer Do s and Don ts
 Reporting of Breast Cancer Do s and Don ts 7 th SGH Annual Breast Pathology Course Professor Michael Bilous Conjoint Professor Western Sydney University Consultant Pathologist, Australian Clinical Labs,
Reporting of Breast Cancer Do s and Don ts 7 th SGH Annual Breast Pathology Course Professor Michael Bilous Conjoint Professor Western Sydney University Consultant Pathologist, Australian Clinical Labs,
Molecular in vitro diagnostic test for the quantitative detection of the mrna expression of ERBB2, ESR1, PGR and MKI67 in breast cancer tissue.
 Innovation for your breast cancer diagnostics Now valid ated on: -Roche cob as z 480 A nalyzer -Roche L ightcycle r 480 II - ABI 750 0 Fast -Versant kpcr Cyc ler PGR ESR1 MKI67 Molecular in vitro diagnostic
Innovation for your breast cancer diagnostics Now valid ated on: -Roche cob as z 480 A nalyzer -Roche L ightcycle r 480 II - ABI 750 0 Fast -Versant kpcr Cyc ler PGR ESR1 MKI67 Molecular in vitro diagnostic
Adjuvant endocrine therapy (essentials in ER positive early breast cancer)
 Adjuvant endocrine therapy (essentials in ER positive early breast cancer) Giuseppe Curigliano MD, PhD Breast Cancer Program Division of Experimental Therapeutics Outline Picking optimal adjuvant endocrine
Adjuvant endocrine therapy (essentials in ER positive early breast cancer) Giuseppe Curigliano MD, PhD Breast Cancer Program Division of Experimental Therapeutics Outline Picking optimal adjuvant endocrine
CLINICOPATHOLOGIC FEATURES AND MOLECULAR SUBTYPES OF BREAST CANCER IN FEZ-MEKNES REGION (MOROCCO): A STUDY OF 390 PATIENTS
 CLINICOPATHOLOGIC FEATURES AND MOLECULAR SUBTYPES OF BREAST CANCER IN FEZ-MEKNES REGION (MOROCCO): A STUDY OF 390 PATIENTS S. Chahbouni¹, A. Amarti 2, N. Hammas 3, L. Chbani 3, H. El fatemi 3 1 Department
CLINICOPATHOLOGIC FEATURES AND MOLECULAR SUBTYPES OF BREAST CANCER IN FEZ-MEKNES REGION (MOROCCO): A STUDY OF 390 PATIENTS S. Chahbouni¹, A. Amarti 2, N. Hammas 3, L. Chbani 3, H. El fatemi 3 1 Department
OVERVIEW OF GENE EXPRESSION-BASED TESTS IN EARLY BREAST CANCER
 OVERVIEW OF GENE EXPRESSION-BASED TESTS IN EARLY BREAST CANCER Aleix Prat, MD PhD Medical Oncology Department Hospital Clínic of Barcelona University of Barcelona esmo.org Disclosures Advisory role for
OVERVIEW OF GENE EXPRESSION-BASED TESTS IN EARLY BREAST CANCER Aleix Prat, MD PhD Medical Oncology Department Hospital Clínic of Barcelona University of Barcelona esmo.org Disclosures Advisory role for
Concordance among Gene-Expression Based Predictors for Breast Cancer
 The new england journal of medicine original article Concordance among Gene-Expression Based Predictors for Breast Cancer Cheng Fan, M.S., Daniel S. Oh, Ph.D., Lodewyk Wessels, Ph.D., Britta Weigelt, Ph.D.,
The new england journal of medicine original article Concordance among Gene-Expression Based Predictors for Breast Cancer Cheng Fan, M.S., Daniel S. Oh, Ph.D., Lodewyk Wessels, Ph.D., Britta Weigelt, Ph.D.,
RECENT ADVANCES IN THE MOLECULAR DIAGNOSIS OF BREAST CANCER
 Technology Transfer in Diagnostic Pathology. 6th Central European Regional Meeting. Cytopathology. Balatonfüred, Hungary, April 7-9, 2011. RECENT ADVANCES IN THE MOLECULAR DIAGNOSIS OF BREAST CANCER Philippe
Technology Transfer in Diagnostic Pathology. 6th Central European Regional Meeting. Cytopathology. Balatonfüred, Hungary, April 7-9, 2011. RECENT ADVANCES IN THE MOLECULAR DIAGNOSIS OF BREAST CANCER Philippe
Multigene Testing in NCCN Breast Cancer Treatment Guidelines, v1.2011
 Multigene Testing in NCCN Breast Cancer Treatment Guidelines, v1.2011 Robert W. Carlson, M.D. Professor of Medicine Stanford University Chair, NCCN Breast Cancer Treatment Guidelines Panel Selection of
Multigene Testing in NCCN Breast Cancer Treatment Guidelines, v1.2011 Robert W. Carlson, M.D. Professor of Medicine Stanford University Chair, NCCN Breast Cancer Treatment Guidelines Panel Selection of
Table S2. Expression of PRMT7 in clinical breast carcinoma samples
 Table S2. Expression of PRMT7 in clinical breast carcinoma samples (All data were obtained from cancer microarray database Oncomine.) Analysis type* Analysis Class(number sampels) 1 2 3 4 Correlation (up/down)#
Table S2. Expression of PRMT7 in clinical breast carcinoma samples (All data were obtained from cancer microarray database Oncomine.) Analysis type* Analysis Class(number sampels) 1 2 3 4 Correlation (up/down)#
Only Estrogen receptor positive is not enough to predict the prognosis of breast cancer
 Young Investigator Award, Global Breast Cancer Conference 2018 Only Estrogen receptor positive is not enough to predict the prognosis of breast cancer ㅑ Running head: Revisiting estrogen positive tumors
Young Investigator Award, Global Breast Cancer Conference 2018 Only Estrogen receptor positive is not enough to predict the prognosis of breast cancer ㅑ Running head: Revisiting estrogen positive tumors
Package AIMS. June 29, 2018
 Type Package Package AIMS June 29, 2018 Title AIMS : Absolute Assignment of Breast Cancer Intrinsic Molecular Subtype Version 1.12.0 Date 2014-06-25 Description This package contains the AIMS implementation.
Type Package Package AIMS June 29, 2018 Title AIMS : Absolute Assignment of Breast Cancer Intrinsic Molecular Subtype Version 1.12.0 Date 2014-06-25 Description This package contains the AIMS implementation.
Molecular subclasses of breast cancer: how do we define them? The IMPAKT 2012 Working Group Statement
 Annals of Oncology 23: 2997 3006, 2012 doi:10.1093/annonc/mds586 Molecular subclasses of breast cancer: how do we define them? The IMPAKT 2012 Working Group Statement S. Guiu 1, S. Michiels 2, F. André
Annals of Oncology 23: 2997 3006, 2012 doi:10.1093/annonc/mds586 Molecular subclasses of breast cancer: how do we define them? The IMPAKT 2012 Working Group Statement S. Guiu 1, S. Michiels 2, F. André
UK Interdisciplinary Breast Cancer Symposium. Should lobular phenotype be considered when deciding treatment? Michael J Kerin
 UK Interdisciplinary Breast Cancer Symposium Should lobular phenotype be considered when deciding treatment? Michael J Kerin Professor of Surgery National University of Ireland, Galway and Galway University
UK Interdisciplinary Breast Cancer Symposium Should lobular phenotype be considered when deciding treatment? Michael J Kerin Professor of Surgery National University of Ireland, Galway and Galway University
Breast cancer in elderly patients (70 years and older): The University of Tennessee Medical Center at Knoxville 10 year experience
 Breast cancer in elderly patients (70 years and older): The University of Tennessee Medical Center at Knoxville 10 year experience Curzon M, Curzon C, Heidel RE, Desai P, McLoughlin J, Panella T, Bell
Breast cancer in elderly patients (70 years and older): The University of Tennessee Medical Center at Knoxville 10 year experience Curzon M, Curzon C, Heidel RE, Desai P, McLoughlin J, Panella T, Bell
Current Status of Biomarkers (including DNA Tumor Markers and Immunohistochemistry in the Laboratory Diagnosis of Tumors)
 Current Status of Biomarkers (including DNA Tumor Markers and Immunohistochemistry in the Laboratory Diagnosis of Tumors) Kael Mikesell, DO McKay-Dee Hospital May 14, 2015 Outline Update to DNA Testing
Current Status of Biomarkers (including DNA Tumor Markers and Immunohistochemistry in the Laboratory Diagnosis of Tumors) Kael Mikesell, DO McKay-Dee Hospital May 14, 2015 Outline Update to DNA Testing
Present Role of Immunohistochemistry in the. Subtypes. Beppe Viale European Institute of Oncology University of Milan Milan-Italy
 Present Role of Immunohistochemistry in the Classification of Molecular Subtypes Beppe Viale European Institute of Oncology University of Milan Milan-Italy We know it is many diseases Breast cancer is
Present Role of Immunohistochemistry in the Classification of Molecular Subtypes Beppe Viale European Institute of Oncology University of Milan Milan-Italy We know it is many diseases Breast cancer is
Morphological and Molecular Typing of breast Cancer
 Morphological and Molecular Typing of breast Cancer Ian Ellis Molecular Medical Sciences, University of Nottingham Department of Histopathology, Nottingham University Hospitals NHS Trust Histological
Morphological and Molecular Typing of breast Cancer Ian Ellis Molecular Medical Sciences, University of Nottingham Department of Histopathology, Nottingham University Hospitals NHS Trust Histological
Question 1 A. ER-, PR-, HER+ B. ER+, PR+, HER2- C. ER-, PR+, HER2- D. ER-, PR-, HER2- E. ER-, PR+, HER2+
 Triple Negative Breast Cancer Laura C. Collins, M.D. Department of Pathology Beth Israel Deaconess Medical Center and Harvard Medical School, Boston, MA Question 1 The tumor depicted on the next slide
Triple Negative Breast Cancer Laura C. Collins, M.D. Department of Pathology Beth Israel Deaconess Medical Center and Harvard Medical School, Boston, MA Question 1 The tumor depicted on the next slide
BreastPRS is a gene expression assay that stratifies intermediaterisk Oncotype DX patients into high- or low-risk for disease recurrence
 Breast Cancer Res Treat (2013) 139:705 715 DOI 10.1007/s10549-013-2604-0 PRECLINICAL STUDY BreastPRS is a gene expression assay that stratifies intermediaterisk Oncotype DX patients into high- or low-risk
Breast Cancer Res Treat (2013) 139:705 715 DOI 10.1007/s10549-013-2604-0 PRECLINICAL STUDY BreastPRS is a gene expression assay that stratifies intermediaterisk Oncotype DX patients into high- or low-risk
Adjuvan Chemotherapy in Breast Cancer
 Adjuvan Chemotherapy in Breast Cancer Prof Dr Adnan Aydıner Istanbul University, Oncology Institute aa1 Slide 1 aa1 adnan aydiner; 17.02.2008 15-Year Reductions in Recurrence and Disease-Specific Mortality
Adjuvan Chemotherapy in Breast Cancer Prof Dr Adnan Aydıner Istanbul University, Oncology Institute aa1 Slide 1 aa1 adnan aydiner; 17.02.2008 15-Year Reductions in Recurrence and Disease-Specific Mortality
Is Gene Expression Profiling the Best Method for Selecting Systemic Therapy in EBC? Norman Wolmark Miami March 8, 2013
 Is Gene Expression Profiling the Best Method for Selecting Systemic Therapy in EBC? Norman Wolmark Miami March 8, 2013 Changing Phases claudin low Lum A Lum B Basal Her2 NIH Consensus Development Panel,
Is Gene Expression Profiling the Best Method for Selecting Systemic Therapy in EBC? Norman Wolmark Miami March 8, 2013 Changing Phases claudin low Lum A Lum B Basal Her2 NIH Consensus Development Panel,
Supplementary webappendix
 Supplementary webappendix This webappendix formed part of the original submission and has been peer reviewed. We post it as supplied by the authors. Supplement to: Kratz JR, He J, Van Den Eeden SK, et
Supplementary webappendix This webappendix formed part of the original submission and has been peer reviewed. We post it as supplied by the authors. Supplement to: Kratz JR, He J, Van Den Eeden SK, et
Immunohistochemical Analysis of ER, PR, Her2 and CK5/6 in Infiltrative Breast Carcinomas in Indian Patients
 Immunohistochemical Analysis of Infiltrative Breast Carcinomas in Indian Patients RESEARCH COMMUNICATION Immunohistochemical Analysis of ER, PR, Her2 and CK5/6 in Infiltrative Breast Carcinomas in Indian
Immunohistochemical Analysis of Infiltrative Breast Carcinomas in Indian Patients RESEARCH COMMUNICATION Immunohistochemical Analysis of ER, PR, Her2 and CK5/6 in Infiltrative Breast Carcinomas in Indian
Emerging Data on Multianalyte Algorithm Assays in Breast Cancer: A Clinical Review
 June 2015 Volume 13, Issue 6, Supplement 6 Faculty Ralph V. Boccia, MD Medical Director Center for Cancer and Blood Disorders Bethesda, Maryland Clinical Associate Professor of Medicine Georgetown University
June 2015 Volume 13, Issue 6, Supplement 6 Faculty Ralph V. Boccia, MD Medical Director Center for Cancer and Blood Disorders Bethesda, Maryland Clinical Associate Professor of Medicine Georgetown University
Breast cancer pathology
 Breast cancer pathology Giancarlo Pruneri, M.D. National Cancer Institute (INT) Milan University of Milan, School of Medicine Giancarlo.Pruneri@unimi.it Currently accepted prognostic/predictive parameters
Breast cancer pathology Giancarlo Pruneri, M.D. National Cancer Institute (INT) Milan University of Milan, School of Medicine Giancarlo.Pruneri@unimi.it Currently accepted prognostic/predictive parameters
Predictive Assays in Radiation Therapy
 Outline Predictive Assays in Radiation Therapy Radiation Biology Introduction Early predictive assays Recent trends in predictive assays Examples for specific tumors Summary Lecture 4-23-2014 Introduction
Outline Predictive Assays in Radiation Therapy Radiation Biology Introduction Early predictive assays Recent trends in predictive assays Examples for specific tumors Summary Lecture 4-23-2014 Introduction
Layered-IHC (L-IHC): A novel and robust approach to multiplexed immunohistochemistry So many markers and so little tissue
 Page 1 The need for multiplex detection of tissue biomarkers. There is a constant and growing demand for increased biomarker analysis in human tissue specimens. Analysis of tissue biomarkers is key to
Page 1 The need for multiplex detection of tissue biomarkers. There is a constant and growing demand for increased biomarker analysis in human tissue specimens. Analysis of tissue biomarkers is key to
Post Neoadjuvant therapy: issues in interpretation
 Post Neoadjuvant therapy: issues in interpretation Disclosure: Overview D Prognostic features in assessment of post treatment specimens: Tumor size Cellularity Grade Receptors LN Neoadjuvant chemotherapy:
Post Neoadjuvant therapy: issues in interpretation Disclosure: Overview D Prognostic features in assessment of post treatment specimens: Tumor size Cellularity Grade Receptors LN Neoadjuvant chemotherapy:
Diagnosis and Treatment of Patients with Primary and Metastatic Breast Cancer. Pathology. AGO e. V. in der DGGG e.v. sowie in der DKG e.v.
 Diagnosis and Treatment of Patients with Primary and Metastatic Breast Cancer Pathology Pathology Versions 2004 2017: Blohmer / Costa / Fehm / Friedrichs / Huober / Kreipe / Lück / Schneeweis / Sinn /
Diagnosis and Treatment of Patients with Primary and Metastatic Breast Cancer Pathology Pathology Versions 2004 2017: Blohmer / Costa / Fehm / Friedrichs / Huober / Kreipe / Lück / Schneeweis / Sinn /
EGFR as paradoxical predictor of chemosensitivity and outcome among triple-negative breast cancer
 ONCOLOGY REPORTS 21: 413-417, 2009 413 EGFR as paradoxical predictor of chemosensitivity and outcome among triple-negative breast cancer HIROKO NOGI 1, TADASHI KOBAYASHI 2, MASAFUMI SUZUKI 3, ISAO TABEI
ONCOLOGY REPORTS 21: 413-417, 2009 413 EGFR as paradoxical predictor of chemosensitivity and outcome among triple-negative breast cancer HIROKO NOGI 1, TADASHI KOBAYASHI 2, MASAFUMI SUZUKI 3, ISAO TABEI
Molecular Characterization of Breast Cancer: The Clinical Significance
 Molecular Characterization of : The Clinical Significance Shahla Masood, M.D. Professor and Chair Department of Pathology and Laboratory Medicine University of Florida College of Medicine-Jacksonville
Molecular Characterization of : The Clinical Significance Shahla Masood, M.D. Professor and Chair Department of Pathology and Laboratory Medicine University of Florida College of Medicine-Jacksonville
AIMS: Absolute Assignment of Breast Cancer Intrinsic Molecular Subtype
 AIMS: Absolute Assignment of Breast Cancer Intrinsic Molecular Subtype Eric R. Paquet (eric.r.paquet@gmail.com), Michael T. Hallett (michael.t.hallett@mcgill.ca) 1 1 Department of Biochemistry, Breast
AIMS: Absolute Assignment of Breast Cancer Intrinsic Molecular Subtype Eric R. Paquet (eric.r.paquet@gmail.com), Michael T. Hallett (michael.t.hallett@mcgill.ca) 1 1 Department of Biochemistry, Breast
A Prospective Comparison of the 21-Gene Recurrence Score and the PAM50-Based Prosigna in Estrogen Receptor-Positive Early-Stage Breast Cancer
 Adv Ther (2015) 32:1237 1247 DOI 10.1007/s12325-015-0269-2 ORIGINAL RESEARCH A Prospective Comparison of the 21-Gene Recurrence Score and the PAM50-Based Prosigna in Estrogen Receptor-Positive Early-Stage
Adv Ther (2015) 32:1237 1247 DOI 10.1007/s12325-015-0269-2 ORIGINAL RESEARCH A Prospective Comparison of the 21-Gene Recurrence Score and the PAM50-Based Prosigna in Estrogen Receptor-Positive Early-Stage
Corporate Medical Policy
 Corporate Medical Policy Microarray-based Gene Expression Testing for Cancers of Unknown File Name: Origination: Last CAP Review: Next CAP Review: Last Review: microarray-based_gene_expression_testing_for_cancers_of_unknown_primary
Corporate Medical Policy Microarray-based Gene Expression Testing for Cancers of Unknown File Name: Origination: Last CAP Review: Next CAP Review: Last Review: microarray-based_gene_expression_testing_for_cancers_of_unknown_primary
Breast Cancer Subtypes and the Risk of Local and Regional Relapse
 VOLUME 28 NUMBER 10 APRIL 1 2010 JOURNAL OF CLINICAL ONCOLOGY O R I G I N A L R E P O R T Breast Cancer s and the Risk of Local and Regional Relapse K. David Voduc, Maggie C.U. Cheang, Scott Tyldesley,
VOLUME 28 NUMBER 10 APRIL 1 2010 JOURNAL OF CLINICAL ONCOLOGY O R I G I N A L R E P O R T Breast Cancer s and the Risk of Local and Regional Relapse K. David Voduc, Maggie C.U. Cheang, Scott Tyldesley,
Current Status and Future Development of Tools for Prognosis and Prediction - USA
 Current Status and Future Development of Tools for Prognosis and Prediction - USA John H. Ward, MD Professor of Medicine University of Utah School of Medicine Huntsman Cancer Institute October18, 2009
Current Status and Future Development of Tools for Prognosis and Prediction - USA John H. Ward, MD Professor of Medicine University of Utah School of Medicine Huntsman Cancer Institute October18, 2009
ncounter Assay Automated Process Immobilize and align reporter for image collecting and barcode counting ncounter Prep Station
 ncounter Assay ncounter Prep Station Automated Process Hybridize Reporter to RNA Remove excess reporters Bind reporter to surface Immobilize and align reporter Image surface Count codes Immobilize and
ncounter Assay ncounter Prep Station Automated Process Hybridize Reporter to RNA Remove excess reporters Bind reporter to surface Immobilize and align reporter Image surface Count codes Immobilize and
Genomic Profiling of Tumors and Loco-Regional Recurrence
 1 Genomic Profiling of Tumors and Loco-Regional Recurrence Terry Mamounas, M.D., M.P.H., F.A.C.S. Medical Director, Comprehensive Breast Program UF Health Cancer Center at Orlando Health Professor of Surgery,
1 Genomic Profiling of Tumors and Loco-Regional Recurrence Terry Mamounas, M.D., M.P.H., F.A.C.S. Medical Director, Comprehensive Breast Program UF Health Cancer Center at Orlando Health Professor of Surgery,
Heterogeneidad tumoral. Federico Rojo
 Heterogeneidad tumoral Federico Rojo Outline of the presentation Definition and evidences Intertumor heterogeneity Spatial and temporal intratumor heterogeneity Clinical implications of tumor heterogeneity.
Heterogeneidad tumoral Federico Rojo Outline of the presentation Definition and evidences Intertumor heterogeneity Spatial and temporal intratumor heterogeneity Clinical implications of tumor heterogeneity.
Cover Page. The handle holds various files of this Leiden University dissertation
 Cover Page The handle http://hdl.handle.net/1887/55957 holds various files of this Leiden University dissertation Author: Dekker T.J.A. Title: Optimizing breast cancer survival models based on conventional
Cover Page The handle http://hdl.handle.net/1887/55957 holds various files of this Leiden University dissertation Author: Dekker T.J.A. Title: Optimizing breast cancer survival models based on conventional
Luminal A and B Where are we? (or lost in translation?)
 Luminal A and B Where are we? (or lost in translation?) Emiel J. Rutgers The Netherlands Cancer Institute Antoni Van Leeuwenhoek Hospital Amsterdam How to determine adjuvant or neoadjuvant treatment for
Luminal A and B Where are we? (or lost in translation?) Emiel J. Rutgers The Netherlands Cancer Institute Antoni Van Leeuwenhoek Hospital Amsterdam How to determine adjuvant or neoadjuvant treatment for
Applications of IHC. Determination of the primary site in metastatic tumors of unknown origin
 Applications of IHC Determination of the primary site in metastatic tumors of unknown origin Classification of tumors that appear 'undifferentiated' by standard light microscopy Precise classification
Applications of IHC Determination of the primary site in metastatic tumors of unknown origin Classification of tumors that appear 'undifferentiated' by standard light microscopy Precise classification
TABLE OF CONTENTS. Executive Summary The EndoPredict Test Intended Use Population Breast Cancer Clinical Dilemma. Analytical Validity
 Clinical Dossier TABLE OF CONTENTS Executive Summary The EndoPredict Test Intended Use Population Breast Cancer Clinical Dilemma 1 2 3 Analytical Validity 4 Clinical Validity 4 Clinical Utility 7 Medicare
Clinical Dossier TABLE OF CONTENTS Executive Summary The EndoPredict Test Intended Use Population Breast Cancer Clinical Dilemma 1 2 3 Analytical Validity 4 Clinical Validity 4 Clinical Utility 7 Medicare
Controversies in Breast Pathology ELENA PROVENZANO ADDENBROOKES HOSPITAL, CAMBRIDGE
 Controversies in Breast Pathology ELENA PROVENZANO ADDENBROOKES HOSPITAL, CAMBRIDGE Neoadjuvant Chemotherapy Indications: Management of locally advanced invasive breast cancers including inflammatory breast
Controversies in Breast Pathology ELENA PROVENZANO ADDENBROOKES HOSPITAL, CAMBRIDGE Neoadjuvant Chemotherapy Indications: Management of locally advanced invasive breast cancers including inflammatory breast
10/15/2012. Biologic Subtypes of TNBC. Topics. Topics. Histopathology Molecular pathology Clinical relevance
 Biologic Subtypes of TNBC Andrea L. Richardson M.D. Ph.D. Brigham and Women s Hospital Dana-Farber Cancer Institute Harvard Medical School Boston, MA Topics Histopathology Molecular pathology Clinical
Biologic Subtypes of TNBC Andrea L. Richardson M.D. Ph.D. Brigham and Women s Hospital Dana-Farber Cancer Institute Harvard Medical School Boston, MA Topics Histopathology Molecular pathology Clinical
Refining Prognosis of Early Stage Lung Cancer by Molecular Features (Part 2): Early Steps in Molecularly Defined Prognosis
 5/17/13 Refining Prognosis of Early Stage Lung Cancer by Molecular Features (Part 2): Early Steps in Molecularly Defined Prognosis Johannes Kratz, MD Post-doctoral Fellow, Thoracic Oncology Laboratory
5/17/13 Refining Prognosis of Early Stage Lung Cancer by Molecular Features (Part 2): Early Steps in Molecularly Defined Prognosis Johannes Kratz, MD Post-doctoral Fellow, Thoracic Oncology Laboratory
Modern classification of breast cancer-should we stick with morphology or convert to molecular profiles?
 Modern classification of breast cancer-should we stick with morphology or convert to molecular profiles? Ian Ellis Professor of Cancer Pathology Molecular Medical Sciences University of Nottingham Dept
Modern classification of breast cancer-should we stick with morphology or convert to molecular profiles? Ian Ellis Professor of Cancer Pathology Molecular Medical Sciences University of Nottingham Dept
The intended place in therapy would be to replace IHC testing in people with early-stage breast cancer.
 pat hways MammaTyper in vitro diagnostic test for determining breast cancer subtypes Medtech innovation briefing Published: 18 January 2018 nice.org.uk/guidance/mib135 Summary The technology described
pat hways MammaTyper in vitro diagnostic test for determining breast cancer subtypes Medtech innovation briefing Published: 18 January 2018 nice.org.uk/guidance/mib135 Summary The technology described
Editorial Process: Submission:11/30/2017 Acceptance:01/04/2019
 RESEARCH ARTICLE Editorial Process: Submission:11/30/2017 Acceptance:01/04/2019 in Non-Metastatic Triple-Negative Breast Cancer O Al jarroudi*, A Zaimi, S A Brahmi, S Afqir Abstract Introduction: Triple-negative
RESEARCH ARTICLE Editorial Process: Submission:11/30/2017 Acceptance:01/04/2019 in Non-Metastatic Triple-Negative Breast Cancer O Al jarroudi*, A Zaimi, S A Brahmi, S Afqir Abstract Introduction: Triple-negative
Supplementary Online Content
 Supplementary Online Content Fumagalli D, Venet D, Ignatiadis M, et al. RNA Sequencing to predict response to neoadjuvant anti-her2 therapy: a secondary analysis of the NeoALTTO randomized clinical trial.
Supplementary Online Content Fumagalli D, Venet D, Ignatiadis M, et al. RNA Sequencing to predict response to neoadjuvant anti-her2 therapy: a secondary analysis of the NeoALTTO randomized clinical trial.
Molecular classification of breast cancer implications for pathologists. Sarah E Pinder
 Molecular classification of breast cancer implications for pathologists Sarah E Pinder Courtesy of CW Elston Histological types Breast Cancer Special Types 17 morphological special types 25-30% of all
Molecular classification of breast cancer implications for pathologists Sarah E Pinder Courtesy of CW Elston Histological types Breast Cancer Special Types 17 morphological special types 25-30% of all
ncounter Assay Automated Process Capture & Reporter Probes Bind reporter to surface Remove excess reporters Hybridize CodeSet to RNA
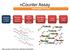 ncounter Assay Automated Process Hybridize CodeSet to RNA Remove excess reporters Bind reporter to surface Immobilize and align reporter Image surface Count codes mrna Capture & Reporter Probes slides
ncounter Assay Automated Process Hybridize CodeSet to RNA Remove excess reporters Bind reporter to surface Immobilize and align reporter Image surface Count codes mrna Capture & Reporter Probes slides
Proteomic Biomarker Discovery in Breast Cancer
 Proteomic Biomarker Discovery in Breast Cancer Rob Baxter Laboratory for Cellular and Diagnostic Proteomics Kolling Institute, University of Sydney, Royal North Shore Hospital robert.baxter@sydney.edu.au
Proteomic Biomarker Discovery in Breast Cancer Rob Baxter Laboratory for Cellular and Diagnostic Proteomics Kolling Institute, University of Sydney, Royal North Shore Hospital robert.baxter@sydney.edu.au
8/8/2011. PONDERing the Need to TAILOR Adjuvant Chemotherapy in ER+ Node Positive Breast Cancer. Overview
 Overview PONDERing the Need to TAILOR Adjuvant in ER+ Node Positive Breast Cancer Jennifer K. Litton, M.D. Assistant Professor The University of Texas M. D. Anderson Cancer Center Using multigene assay
Overview PONDERing the Need to TAILOR Adjuvant in ER+ Node Positive Breast Cancer Jennifer K. Litton, M.D. Assistant Professor The University of Texas M. D. Anderson Cancer Center Using multigene assay
Type: Evidence Based Evidence Quality: High Strength of Recommendation: Strong
 Clinical Question 1: For women with early-stage invasive breast cancer and with known estrogen and progesterone receptor (ER/PgR) and human epidermal growth factor receptor 2 (HER2 status), which other
Clinical Question 1: For women with early-stage invasive breast cancer and with known estrogen and progesterone receptor (ER/PgR) and human epidermal growth factor receptor 2 (HER2 status), which other
Oncotype DX testing in node-positive disease
 Should gene array assays be routinely used in node positive disease? Yes Christy A. Russell, MD University of Southern California Oncotype DX testing in node-positive disease 1 Validity of the Oncotype
Should gene array assays be routinely used in node positive disease? Yes Christy A. Russell, MD University of Southern California Oncotype DX testing in node-positive disease 1 Validity of the Oncotype
Molecular Classification of Iraqi Breast Cancer Patients and Its Correlation with Patients Profile
 Original Article Molecular Classification of Iraqi Breast Cancer Patients and Its Correlation * Manwar A Al-Naqqash* Mustafa K Jassim** MBChB, DMRT MBChB, DMRT, MSc Pathology MBChB Fac Med Baghdad 2016;
Original Article Molecular Classification of Iraqi Breast Cancer Patients and Its Correlation * Manwar A Al-Naqqash* Mustafa K Jassim** MBChB, DMRT MBChB, DMRT, MSc Pathology MBChB Fac Med Baghdad 2016;
Response to Paclitaxel in Node-positive Triple Negative Breast Cancer
 J Korean Surg Soc 2010;79:173-179 DOI: 10.4174/jkss.2010.79.3.173 원 저 Response to Paclitaxel in Node-positive Triple Negative Breast Cancer Department of Surgery, Kosin University College of Medicine,
J Korean Surg Soc 2010;79:173-179 DOI: 10.4174/jkss.2010.79.3.173 원 저 Response to Paclitaxel in Node-positive Triple Negative Breast Cancer Department of Surgery, Kosin University College of Medicine,
Study Finds Lung Cancer Subtype Test from UNC, Startup GeneCentric to be Accurate, Reproducible
 Study Finds Lung Cancer Subtype Test from UNC, Startup GeneCentric to be Accurate, Reproducible May 30, 2013 By Ben Butkus A team led by clinical researchers from the University of North Carolina, Chapel
Study Finds Lung Cancer Subtype Test from UNC, Startup GeneCentric to be Accurate, Reproducible May 30, 2013 By Ben Butkus A team led by clinical researchers from the University of North Carolina, Chapel
National Medical Policy
 National Medical Policy Subject: Policy Number: MammaPrint, Prosigna and Other Gene Expression Tests for Breast Cancer NMP520 Effective Date*: May 2013 Updated: June 2017 This National Medical Policy is
National Medical Policy Subject: Policy Number: MammaPrint, Prosigna and Other Gene Expression Tests for Breast Cancer NMP520 Effective Date*: May 2013 Updated: June 2017 This National Medical Policy is
Quality Assurance and Quality Control in the Pathology Dept.
 Quality Assurance and Quality Control in the Pathology Dept. Judith Sandbank M.D. Pathology Assaf-Harofeh Medical Center ISRAEL jsandbank@asaf.health.gov.il 2 nd IBDC, 9 th February, 2012 Pathology as
Quality Assurance and Quality Control in the Pathology Dept. Judith Sandbank M.D. Pathology Assaf-Harofeh Medical Center ISRAEL jsandbank@asaf.health.gov.il 2 nd IBDC, 9 th February, 2012 Pathology as
MammaPrint, the story of the 70-gene profile
 MammaPrint, the story of the 70-gene profile René Bernards Professor of Molecular Carcinogenesis The Netherlands Cancer Institute Amsterdam Chief Scientific Officer Agendia Amsterdam The breast cancer
MammaPrint, the story of the 70-gene profile René Bernards Professor of Molecular Carcinogenesis The Netherlands Cancer Institute Amsterdam Chief Scientific Officer Agendia Amsterdam The breast cancer
Molecular Classification of Breast Cancer Implications for Therapy Selection
 Molecular Classification of Breast Cancer Implications for Therapy Selection Karen Gelmon MD FRCPC Professor Medicine, UBC Medical Oncologist, BC Cancer Agency Summary Describe the current molecular classifications
Molecular Classification of Breast Cancer Implications for Therapy Selection Karen Gelmon MD FRCPC Professor Medicine, UBC Medical Oncologist, BC Cancer Agency Summary Describe the current molecular classifications
SUPPLEMENTARY INFORMATION
 doi:.38/nature8975 SUPPLEMENTAL TEXT Unique association of HOTAIR with patient outcome To determine whether the expression of other HOX lincrnas in addition to HOTAIR can predict patient outcome, we measured
doi:.38/nature8975 SUPPLEMENTAL TEXT Unique association of HOTAIR with patient outcome To determine whether the expression of other HOX lincrnas in addition to HOTAIR can predict patient outcome, we measured
Breast Cancer: ASCO Poster Review
 Breast Cancer: ASCO Poster Review Carmen Criscitiello, MD, PhD Istituto Europeo di Oncologia Milano HER2+ SUBTYPE Research questions in early HER2+ BC De-escalation of toxicity without compromising efficacy
Breast Cancer: ASCO Poster Review Carmen Criscitiello, MD, PhD Istituto Europeo di Oncologia Milano HER2+ SUBTYPE Research questions in early HER2+ BC De-escalation of toxicity without compromising efficacy
SALSA MLPA KIT P078-B1 Breast Tumour Lot 0210, 0109
 SALSA MLPA KIT P078-B1 Breast Tumour Lot 0210, 0109 This P078-B1 Breast Tumour probemix contains probes for several genes (including ERBB2, BIRC5, MYC, TOP2A, ESR1, MTDH, CCND1, CCNE1, EGFR and C11orf30)
SALSA MLPA KIT P078-B1 Breast Tumour Lot 0210, 0109 This P078-B1 Breast Tumour probemix contains probes for several genes (including ERBB2, BIRC5, MYC, TOP2A, ESR1, MTDH, CCND1, CCNE1, EGFR and C11orf30)
J Clin Oncol 24: by American Society of Clinical Oncology INTRODUCTION
 VOLUME 24 NUMBER 36 DECEMBER 20 2006 JOURNAL OF CLINICAL ONCOLOGY O R I G I N A L R E P O R T Locoregional Relapse and Distant Metastasis in Conservatively Managed Triple Negative Early-Stage Breast Cancer
VOLUME 24 NUMBER 36 DECEMBER 20 2006 JOURNAL OF CLINICAL ONCOLOGY O R I G I N A L R E P O R T Locoregional Relapse and Distant Metastasis in Conservatively Managed Triple Negative Early-Stage Breast Cancer
Clinical Policy Title: Breast cancer index genetic testing
 Clinical Policy Title: Breast cancer index genetic testing Clinical Policy Number: 02.01.22 Effective Date: January 1, 2017 Initial Review Date: October 19, 2016 Most Recent Review Date: October 19, 2016
Clinical Policy Title: Breast cancer index genetic testing Clinical Policy Number: 02.01.22 Effective Date: January 1, 2017 Initial Review Date: October 19, 2016 Most Recent Review Date: October 19, 2016
SYSTEMIC THERAPY OPTIONS FOR BREAST CANCER IN 2014
 SYSTEMIC THERAPY OPTIONS FOR BREAST Oncology Day 2014 CANCER IN 2014 Dr. Katherine Enright, M.D., M.P.H., F.R.C.P.(C) Katherine.enright@Trilliumhealthpartners.ca OBJECTIVES 1. Outline an approach to the
SYSTEMIC THERAPY OPTIONS FOR BREAST Oncology Day 2014 CANCER IN 2014 Dr. Katherine Enright, M.D., M.P.H., F.R.C.P.(C) Katherine.enright@Trilliumhealthpartners.ca OBJECTIVES 1. Outline an approach to the
> 6000 Mutations in Melanoma. Tests That Cay Be Employed. FISH for Additions/Deletions. Comparative Genomic Hybridization
 Winter Clinical 2017: The Assessment and Diagnosis of Melanoma Whitney A. High, MD, JD, MEng Associate Professor, Dermatology & Pathology Director of Dermatopathology (Dermatology) University of Colorado
Winter Clinical 2017: The Assessment and Diagnosis of Melanoma Whitney A. High, MD, JD, MEng Associate Professor, Dermatology & Pathology Director of Dermatopathology (Dermatology) University of Colorado
Genetic alterations of histone lysine methyltransferases and their significance in breast cancer
 Genetic alterations of histone lysine methyltransferases and their significance in breast cancer Supplementary Materials and Methods Phylogenetic tree of the HMT superfamily The phylogeny outlined in the
Genetic alterations of histone lysine methyltransferases and their significance in breast cancer Supplementary Materials and Methods Phylogenetic tree of the HMT superfamily The phylogeny outlined in the
Triple-Negative Breast Cancer Time to Slice and Dice? Carsten Denkert, MD Charité University Hospital Berlin, Germany
 Triple-Negative Breast Cancer Time to Slice and Dice? Carsten Denkert, MD Charité University Hospital Berlin, Germany Triple-Negative Breast Cancer (TNBC) 2018 Presentation Outline The molecular heterogeneity
Triple-Negative Breast Cancer Time to Slice and Dice? Carsten Denkert, MD Charité University Hospital Berlin, Germany Triple-Negative Breast Cancer (TNBC) 2018 Presentation Outline The molecular heterogeneity
