Formulary Chemotherapy Agents: (Current as of 6/2018) Therapeutic Class
|
|
|
- Gordon Cole
- 5 years ago
- Views:
Transcription
1 MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY Cancer LAST REVIEW 9/11/18 THERAPEUTIC CLASS Oncology REVIEW HISTORY 5/17, 5/16 LOB AFFECTED Medi-Cal (MONTH/YEAR) This policy has been developed through review of medical literature, consideration of medical necessity, generally accepted medical practice standards, and approved by the HPSJ Pharmacy and Therapeutic Advisory Committee. OVERVIEW Oncology medications account for one of the largest drug category spend in the United States. In more recent years, drug innovative companies have had tremendous success releasing numerous new drugs on the market each year with the latest goal to develop targeted cancer tumor cells as opposed to the traditional, cytotoxic chemotherapy agents. The below criteria, limits, and requirements for HPSJ s formulary oncology agents. **Note: This coverage policy strictly reviews the agents used specifically for the treatment or management of cancer. For Antiemetic Agents, please refer to Coverage Policy Gastrointestinal Disorders Nausea. For ESAs (i.e. Epogen), please refer to Coverage Policy Renal Anemia. Formulary Chemotherapy Agents: (Current as of 6/2018) Therapeutic Class Alkylating Agents Anti-androgens Antineoplastic Antibiotic Generic Name (Brand Name) Altretamine (Hexalen) Busulfan (Myleran, Busulfex), Carmustine (BiCNU) Chlorambucil (Leukeran) Cyclophosphamide Cyclophosphamide Vial Estramustine (Emcyt) Hydroxyurea (Hydrea) Lomustine (Ceenu) Melphalan (Alkeran) Oxaliplatin (Eloxatin) Procarbazine (Matulane) Available Strengths Formulary Limits Average Cost per 30 days 50mg PA, SP $3,604.52* PO: 2mg : 60mg/10ml PA, SP $ * 100mg Vial PA, SP $3084.3* 2mg PA, SP $368.89* 25mg, 50mg -- $ mg -- $ mg PA, SP $3,372.60* 500mg -- $ mg, 40mg, 100mg PA, SP $1, mg PA, SP $2,335.5* 50mg/10ml, 100mg/20ml PA, SP $352.92* 50mg PA $2, Streptozocin (Zanosar) 1gram PA, SP $ Temozolomide (Temodar), Bicalutamide (Casodex) Nilutamide (Nilandron) Flutamide (Euflex) Bleomycin Sulfate (Blenoxane) Dactinomycin (Cosmegen) Mitomycin (Mutamycin) PO: 5mg, 20mg, 100mg, 140mg, 280mg 250mg : 100mg PA, SP $1, mg -- $ mg PA, SP $8,696.34* 125mg PA, SP $ unit, 30 unit PA, SP $440.00* 0.5 mg PA, SP $3,040.50* 5mg, 20mg, 40mg PA, SP $1,415.64* Coverage Policy Oncology - Cancer Page 1
2 Monoclonal Antibody Anti-metabolites Anti-CD20 Anti- HER2+ Anti-PD- L1 EGFR Inhibitor VEGF Inhibitor Aromatase Inhibitors MEK inhibitor mtor Kinase Inhibitor Topoisomerase I Inhibitor Topoisomerase II Inhibitor Angiogenesis Inhibitor Rituximab (Rituxan) Pertuzumab (Perjeta) Trastuzumab (Herceptin) Atezolizumab (Tecentriq) Avelumab (Bavencio) 10mg/ml PA, SP $12,624.69* 420mg/14ml PA, SP $18,045.00* 150mg, 440mg PA, SP $9,644.09* 1200 mg/20 ml PA, SP $10,656.65* 200 mg/10 ml PA, SP $1,859.52* Durvalumab (Imfinzi) 120mg/2.4ml, 500mg/10ml PA,SP $11,774.00* Cetuximab (Erbitux) Necitumumab (Portrazza) Panitumumab (Vectibix) Bevacizumab (Avastin) Capecitabine (Xeloda) Flurouracil (Carac, Efudex) Cream, Solution Mercaptopurine Methotrexate, SQ Pemetrexed (Alimta) Thioguanine (Tabloid) Anastrozole Exemestane (Aromasin) Letrozole Cobimetinib Fumarate (Cotellic) Trametinib Dimethyl Sulfoxide (Mekinist) Everolimus (Afinitor) Irinotecan HCl (Camptosar) Irinotecan Liposomal (Onivyde) Topotecan (Hycamtin), Etoposide (Etopophos), Vial Mitoxantrone Vial Daunorubicin (Cerubidine) Doxorubicin (Adriamycin) Epirubicin (Ellence) Idarubicin (Idamycin) Valrubicin (Valstar) Lenalidomide (Revlimid) 200mg/100ml PA, SP $17,583.58* 800mg/50ml PA, SP $20,372.00* 100mg/5ml PA, SP $11,969.40* 100 mg/4 ml, 400 mg/16 ml PA, SP $3,346.00* 150mg, 500mg PA, SP $1,552.78* Cream: 0.05%, 0.1%, 0.5% Solution: 0.2%, 0.5% -- $ mg -- $95.35 PO: 2.5mg SQ: 25mg/ml -- $ mg, 500mg PA, SP $7,022.47* 40mg PA, SP $902.39* 1mg -- $ mg PA, SP $ mg -- $ mg PA, SP $6,677.33* 0.5mg, 2mg PA, SP, 2mg $10,832.36* 2.5mg, 5mg 7.5mg, 10mg PA, SP 14, mg/2ml, 300mg/15ml PA, SP $ mg/10ml PA, SP $13,234.26* PO: 0.25mg 1mg : 4mg/4mL PO: 50mg : 100mg PA, SP $9,589.39* PA, SP $154.77* 2mg/ml PA, SP $121.75* 5 mg/ml PA, SP -- 10mg, 50mg, 2mg/ml PA, SP $ mg, 200mg, 50mg/25ml PA, SP $13,909.20* 1mg/ml PA, SP $ mg/ml PA, SP $6, mg, 5mg, 10mg 15mg, 20mg, 25mg PA, SP $16,060.65* Coverage Policy Oncology - Cancer Page 2
3 GnRH Agonist Antineoplastic-Enzyme Inhibitors Antineoplastic-Histone Deacetylase Inhibitor Antineoplastic-Estrogen Receptor Antagonist Retinoic Acid Derivative Taxane Derivative Vinca Alkaloids Antineoplastic- Miscellaneous Goserelin Acetate (Zoladex) Implant Histrelin Acetate (Vantas) Kit Leuprolide Acetate (Eligard) Syringe Leuprolide Acetate (Lupron Depot) Syringe Kit Leuprolide Acetate Kit Triptorelin Pamoate (Trelstar) Syringe Bortezomib (Velcade) Vial Dasatinib (Sprycel) Erlotinib (Tarceva) Imatinib (Gleevec) Lapatinib (Tykerb) Nilotinib (Tasigna) Sorafenib (Nexavar) Sunitinib (Sutent) Vorinostat (Zolinza) Fulvestrant (Faslodex) Intramuscular injection Tamoxifen Oral Solution, Tormifene (Fareston) 3.6mg, 10.8mg PA, SP $ mg PA, SP $4,615.43* 7.5mg, 22.5mg, 30mg, 45mg PA, SP $ mg, 22.5mg, 30mg PA, SP $4, mg/0.2ml PA, SP $ mg/2ml, 11.25mg/2ml PA, SP $975.89* 3.5mg PA, SP $17,299.30* 20mg, 50mg, 70mg 80mg 100mg, 140mg PA, SP $13,046.35* 25mg, 100mg, 150mg PA, SP $8,166.55* 100mg, 400mg PA, SP $4,847.05* 250mg PA, SP $4,852.90* 250mg PA, SP $12,053.15* 200mg PA, SP $22,404.44* 12.5mg, 25mg, 50mg PA, SP $20, mg PA, SP $ 18,011.52* 250mg/5ml NF $1,375.5* IR: 10mg, 20mg Soln: 10mg/5ml -- Soln is NF $ mg NF $1, Bexarotene (Targretin) 75 mg PA, SP $35,800 Tretinoin (Vesanoid) Docetaxel (Docefrez) Paclitaxel Protein-Bound (Abraxane) Vinblastine Sulfate Vincristine Sulfate (Vincasar PFS) Vincristine Sulfate Liposomal (Marqibo) Vinorelbine Tartrate (Navelbine) Asparaginase (Erwinaze) Estramustine (Emcyt) Ixabepilone (Ixempra) Megestrol Acetate Mitotane (Lysodren) 10mg PA, SP $2, mg, 80mg PA, SP $139.59* 100mg PA, SP $3,385.20* 1mg/ml PA, SP $ mg/ml PA, SP $ mg/31ml PA, SP -- 10mg/ml PA, SP $2,593.00* 10,000 Unit PA, SP $18,237.40* 140mg PA, SP $3,372.60* 15mg, 45mg PA, SP $ 18,011.84* 20mg, 40mg -- $ mg PA, SP $2,294.78* Coverage Policy Oncology - Cancer Page 3
4 Chemotherapy Rescue Agents Filgrastim (Neupogen, Granix, Zarxio) Syringe, Peg-Filgrastim (Neulasta) Syringe Leucovorin Calcium 300mcg/ml, 300mcg/0.5ml, 480mcg/0.8ml, 480mcg/1.6ml PA, SP $2,290.31* 6mg/0.6ml PA, SP $5,489.94* 5mg, 10mg, 15mg, 25mg -- $49.62 PA = Prior Authorization Required SP = Restricted to Specialty Pharmacy * Based on standard pricing (AWP or MAC); for weight dosing, used 70kg or 1.73m 2 to estimate standard dose EGFR=Epidermal Growth Factor Receptor VEGF=Vascular Endothelial Growth Factor MEK mitogen-activated extracellular kinase EVALUATION CRITERIA FOR APPROVAL/EXCEPTION CONSIDERATION For agents that do not have established prior authorization criteria or agents that are Non- Formulary, HPSJ will make the determination based on the National Comprehensive Cancer Network (NCCN) Guidelines and Medical Necessity criteria as described in HPSJ Medical Review Guidelines (UM06) see below for details. The following general Medical Necessity criteria are used when there are no diagnosis-or procedurespecific criteria applicable to the situation. All criteria below must be met for the service to be considered medically necessary. 1. The services are prescribed by a licensed health care practitioner practicing within the scope of his/her license in the context of his/her treatment of the individual. 2. The services are safe, effective, and consistent with nationally accepted standards of medical practice. 3. The services are not experimental or investigational. 4. The services are individualized, specific, and consistent with the individual s signs, symptoms, history, and diagnosis. 5. The services follow peer reviewed evidence based literature that support medical necessity. These services are reasonably expected, in a clinically meaningful way, to: i. Help restore or maintain the individual s health, or ii. iii. Improve or prevent deterioration of the individual s disorder or condition, or Delay progression of a disorder or condition characterized by a progressively deteriorating course when that disorder or condition is the focus of treatment for this episode of care. 6. The individual complies with the essential elements of treatment. 7. The services are not primarily for the convenience of the individual, practitioner, caregiver, family, or another party. 8. Services are not being sought as a way to potentially avoid legal proceedings, incarceration, or other legal consequences. 9. The services are not predominantly domiciliary or custodial. 10. No exclusionary criteria are met. Medications Submitting UM (Medical) Authorization vs. Pharmacy Authorization: Most medications can be covered under both medical and pharmacy benefits depending the setting of administration. For medications that is to be dispensed through a LTC pharmacy or outpatient pharmacy, please submit a pharmacy authorization. For all other administration settings (including buy-and-bill), please submit a UM authorization. How to submit a PHARMACY (RX) prior authorization form for review: 1. Submit request through HPSJ s Pharmacy Medication Prior Authorization Request form which can be obtained from 2. Include clinic notes documenting diagnosis, past treatment history, and any pertinent laboratory tests 3. Fax both the completed prior authorization form and the clinic documents to HPSJ Pharmacy Department: Coverage Policy Oncology - Cancer Page 4
5 How to submit a MEDICAL (UM) prior authorization form for review: 1. Submit request through HPSJ s Medical Authorization Request form which can be obtained from 2. Include clinic notes documenting diagnosis, past treatment history, and any pertinent laboratory tests 3. Fax both the completed prior authorization form and the clinic documents to HPSJ Medical Department: Colony Stimulating Factors 1 st line filgrastim-sndz (Zarxio), peg-filgrastim (Neulasta); 2 nd line filgrastim (Neupogen), tbo-filgrastim (Granix) 1 st line filgrastim-sndz (Zarxio), peg-filgrastim (Neulasta) Coverage Criteria: Medical necessity Required Information for Approval: filgrastim-sndz (Zarxio), peg-filgrastim (Neulasta) will be approved based upon medical necessity. Other Notes: None 2 nd line filgrastim (Neupogen), tbo-filgrastim (Granix) Coverage Criteria: filgrastim (Neupogen), tbo-filgrastim (Granix) are reserved for documentation of treatment failure of a 1 st line agent (filgrastim-sndz (Zarxio), peg-filgrastim (Neulasta). Required Information for Approval: Drug refill history showing fill(s) of a 1 st line agent Other Notes: None Alkylating Agents Cylophosphamide, Hydroxyurea Coverage Criteria: None Required Information for Approval: N/A Altretamine, Busulfan, Carmustine, Chlorambucil, Estramustine, Lomustine, Mephalan, Oxaliplatin, Procarbazine, Temozolomide, Streptozocin Coverage Criteria: PA required, approval is determined by medical necessity criteria Anti-androgens Bicalumide Coverage Criteria: None Required Information for Approval: N/A Nilutamide, Flutamide Coverage Policy Oncology - Cancer Page 5
6 Antibiotics Bleomycin Sulfate, Dactinomycin and Mitomycin Coverage Criteria: PA required, approval is determined by medical necessity criteria Anti-Monoclonal Antibodies Rituximab, Pertuzumab, Trastuzumab, Atezolizumab, Avelumab, Durvalumab, Cetuximab, Necitumumab, Panitumumab and Bevacizumab Coverage Criteria: PA required, approval is determined by medical necessity criteria Anti-metabolites Flurouracil, Mercaptopurine and Methotrexate Coverage Criteria: None Required Information for Approval: N/A Capecitabine, Premetrexed, Thioguanine Aromatase Inhibitors Anastrazole, Letrozole Coverage Criteria: None Required Information for Approval: N/A Exemestane MEK Inhibitors Cobimetinib, Trametinib Coverage Policy Oncology - Cancer Page 6
7 mtor Inhibitors Everolimus Topoisomerase I Inhibitor Irinotecan, Topotecan Topoisomerase II Inhibitor Etoposide, Mitoxantrone, Daunorubicin, Doxorubicin, Epirubicin, Idarubicin, Valrubicin Angiogenesis Inhibitor Lenalidomide GnRH Agonist Goserelin, Histrelin, Leuprolide, Triptorelin Enzyme Inhibitors Bortezomib, Dastinib, Erlotinib, Imatinib, Lapatinib, Nilotinib, Sorafenib, Sunitinib Coverage Policy Oncology - Cancer Page 7
8 Histone Deacetylase Inhibitor Vorinostat Estrogen Receptor Antagonist Fulvestrant, Tamoxifen, Tormifene Retinoic Acid Derivative Bexarotene, Tretinoin Vinca Alkaloids Vinblastine Sulfate, Vincristine Sulfate, Vinorelbine Tartrate Antineoplastic - Miscellaneous Asparaginase, Estramustine, Ixabepilone, Megestrol Acetate, Mitotane REFERENCES 1. Blackwell K, Semiglazov V, Krasnozhon D, et al. Comparison of EP2006, a filgrastim biosimilar, to the reference: a phase III, randomized, double-blind clinical study in the prevention of severe neutropenia in patients with breast cancer receiving myelosuppressive chemotherapy. Ann Oncol. 2015;26(9): REVIEW & EDIT HISTORY Document Changes Reference Date P&T Chairman Creation of Policy HPSJ Coverage Policy Oncology Cancer /2016 Johnathan Yeh, PharmD 05.docx Update of Policy HPSJ Coverage Policy Oncology Cancer /2017 Johnathan Yeh, PharmD 05.docx Update of Policy HPSJ Coverage Policy Oncology Cancer docx 9/2018 Johnathan Yeh, PharmD Note: All changes are approved by the HPSJ P&T Committee before incorporation into the utilization policy Coverage Policy Oncology - Cancer Page 8
Acute Lymphocytic Leukemia
 Acute Lymphocytic Leukemia Splenectomy (Removal of Spleen) 6-Mercaptopurine (Purinethol, 6-MP) Alemtuzumab (Campath ) Arsenic Trioxide (Trisenox ) Bendamustine (Treanda ) Bexarotene (Targretin ) Bleomycin
Acute Lymphocytic Leukemia Splenectomy (Removal of Spleen) 6-Mercaptopurine (Purinethol, 6-MP) Alemtuzumab (Campath ) Arsenic Trioxide (Trisenox ) Bendamustine (Treanda ) Bexarotene (Targretin ) Bleomycin
ICON Formulary - October 2018 Legend - ICON Protocols Essential (previously Standard), Core, Enhanced Core, Enhanced Enhanced
 ICON Formulary - October 2018 Legend - ICON Protocols Essential (previously Standard), Core, Enhanced Core, Enhanced Enhanced Class Medicine Name Nappi Strength Form Size Route Abiraterone Acetate ZYTIGA
ICON Formulary - October 2018 Legend - ICON Protocols Essential (previously Standard), Core, Enhanced Core, Enhanced Enhanced Class Medicine Name Nappi Strength Form Size Route Abiraterone Acetate ZYTIGA
Chemotherapy 101 for Radiation Oncology Workers
 Chemotherapy 101 for Radiation Oncology Workers James Sinclair, M.D. CCARE/Medical Director Scripps Cancer Center March 9, 2013 Category 1) Alkylating agents Alkylating agents directly damage DNA to prevent
Chemotherapy 101 for Radiation Oncology Workers James Sinclair, M.D. CCARE/Medical Director Scripps Cancer Center March 9, 2013 Category 1) Alkylating agents Alkylating agents directly damage DNA to prevent
Medication Review. Cancer Chemotherapy Drugs. Pharmacy Technician Training Systems Passassured, LLC
 Medication Review Cancer Chemotherapy Drugs Pharmacy Technician Training Systems Passassured, LLC Medication Review, Cancer Chemotherapy Drugs PassAssured's Pharmacy Technician Training Program Medication
Medication Review Cancer Chemotherapy Drugs Pharmacy Technician Training Systems Passassured, LLC Medication Review, Cancer Chemotherapy Drugs PassAssured's Pharmacy Technician Training Program Medication
HCPCS Code/ generic (Brand) Name J7506. J8520 capecitabine (Xeloda) 1. J8521 capecitabine. J8530 cyclophosphamide. (Cytoxan) 1
 drug and administration compendia Current Price () J7506 Prednisone, oral, per 5 mg 12/1/07 $0.07 $0.19 N/A prednisone 2 J8520 capecitabine (Xeloda) 1 Capecitabine, oral, 150 mg 8/1/07 $5.79 $4.59 N/A
drug and administration compendia Current Price () J7506 Prednisone, oral, per 5 mg 12/1/07 $0.07 $0.19 N/A prednisone 2 J8520 capecitabine (Xeloda) 1 Capecitabine, oral, 150 mg 8/1/07 $5.79 $4.59 N/A
MEDICAL NECESSITY GUIDELINE
 PAGE: 1 of 10 IMPORTANT REMINDER This Clinical Policy has been developed by appropriately experienced and licensed health care professionals based on a thorough review and consideration of generally accepted
PAGE: 1 of 10 IMPORTANT REMINDER This Clinical Policy has been developed by appropriately experienced and licensed health care professionals based on a thorough review and consideration of generally accepted
Overview of Cancer. Laura Bingell RN Transition Center Nurse for MFP (607)
 Overview of Cancer Laura Bingell RN Transition Center Nurse for MFP (607)962-8225 lbingell@ilny.org What is Cancer? A collection of related diseases in which some of the body s cells begin to divide abnormally
Overview of Cancer Laura Bingell RN Transition Center Nurse for MFP (607)962-8225 lbingell@ilny.org What is Cancer? A collection of related diseases in which some of the body s cells begin to divide abnormally
West of Scotland Cancer Network Guideline for Managing Chemotherapy Induced Nausea and Vomiting
 West of Scotland Cancer Network Guideline for Managing Chemotherapy Induced Nausea and Vomiting Definitions Acute nausea and vomiting Delayed nausea and vomiting Anticipatory nausea and vomiting Initial
West of Scotland Cancer Network Guideline for Managing Chemotherapy Induced Nausea and Vomiting Definitions Acute nausea and vomiting Delayed nausea and vomiting Anticipatory nausea and vomiting Initial
VI.2 Elements for a Public Summary VI.2.1 Overview of Disease Epidemiology Acute Nausea and Vomiting (N&V) Etiologies:
 VI.2 Elements for a Public Summary VI.2.1 Overview of Disease Epidemiology Acute Nausea and Vomiting (N&V) Incidence: The incidence of acute and delayed N&V was investigated in highly and moderately emetogenic
VI.2 Elements for a Public Summary VI.2.1 Overview of Disease Epidemiology Acute Nausea and Vomiting (N&V) Incidence: The incidence of acute and delayed N&V was investigated in highly and moderately emetogenic
To help doctors give their patients the best possible care, the American
 Patient Information Resources from ASCO What to Know ASCO s Guideline on Preventing Vomiting Caused by Cancer Treatment SEPTEMBER 2011 KEY MESSAGES The risk of nausea and vomiting depends on the specific
Patient Information Resources from ASCO What to Know ASCO s Guideline on Preventing Vomiting Caused by Cancer Treatment SEPTEMBER 2011 KEY MESSAGES The risk of nausea and vomiting depends on the specific
Guidelines on Chemotherapy-induced Nausea and Vomiting in Pediatric Cancer Patients
 Guidelines on Chemotherapy-induced Nausea Vomiting in Pediatric Cancer Patients COG Supportive Care Endorsed Guidelines Click here to see all the COG Supportive Care Endorsed Guidelines. DISCLAIMER For
Guidelines on Chemotherapy-induced Nausea Vomiting in Pediatric Cancer Patients COG Supportive Care Endorsed Guidelines Click here to see all the COG Supportive Care Endorsed Guidelines. DISCLAIMER For
Objective: To provide a standard procedure for the recycling of unused medication and the disposal of medicines across all BCPFT Hospital sites.
 WARDS/DEPARTMENTS By: 0 01/09/019 1 of 3 Objective: To provide a standard procedure for the recycling of unused medication and the disposal of medicines across all BCPFT Hospital sites. Scope: All Black
WARDS/DEPARTMENTS By: 0 01/09/019 1 of 3 Objective: To provide a standard procedure for the recycling of unused medication and the disposal of medicines across all BCPFT Hospital sites. Scope: All Black
MABCAMPATH mg 3 INJ GENZYME BIOPHARMACEUTICALS. MABCAMPBATH mg 3 INJ GENZYME BIOPHARMACEUTICALS
 ICON Formulary - November 2016 Class Medicine Name Nappi Strength Form Size Route Manufacturer Abiraterone Acetate ZYTIGA 250 720690001 250 mg 120 ORAL JANSSEN PHARMACEUTICALS Aldesleukin CHIRON IL-2 18
ICON Formulary - November 2016 Class Medicine Name Nappi Strength Form Size Route Manufacturer Abiraterone Acetate ZYTIGA 250 720690001 250 mg 120 ORAL JANSSEN PHARMACEUTICALS Aldesleukin CHIRON IL-2 18
Introduction to Antineoplastic Prescribing
 Introduction to Antineoplastic Prescribing Robert Bradbury, R.Ph., BCPS Clinical Coordinator H. Lee Moffitt Cancer Center Objectives Meet the following goals concerning antineoplastic prescribing: Understand
Introduction to Antineoplastic Prescribing Robert Bradbury, R.Ph., BCPS Clinical Coordinator H. Lee Moffitt Cancer Center Objectives Meet the following goals concerning antineoplastic prescribing: Understand
Bladder Cancer Pathways (Urothelial)
 Bladder Cancer Pathways (Urothelial) Patient Name: Date of Birth: Member Number: Treatment Start Date: ICD-10 Code: Pathology: Stage: 0a 0is I II III IV Recurrent Line of Treatment: Neoadjuvant/Pre-Op
Bladder Cancer Pathways (Urothelial) Patient Name: Date of Birth: Member Number: Treatment Start Date: ICD-10 Code: Pathology: Stage: 0a 0is I II III IV Recurrent Line of Treatment: Neoadjuvant/Pre-Op
Description The following are synthetic cannabinoids requiring prior authorization: dronabinol (Marinol, Syndros ), nabilone (Cesamet )
 Clinical Policy: Nabilone (Cesamet), Dronabinol (Marinol, Syndros) Reference Number: CP.CPA.242 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important
Clinical Policy: Nabilone (Cesamet), Dronabinol (Marinol, Syndros) Reference Number: CP.CPA.242 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important
Who is Bearing the Cost?
 THE COS ST T OF CANCE R DRUGS IN CANADA PA R T 2 Who is Bearing the Cost? KONG KHOO, ROSEMARY COLUCCI, WILLIAM HRYNIUK, ROBERT KAMINO, TANIA REDINA AND COLLEEN SAVAGE Introduction Last year we reported
THE COS ST T OF CANCE R DRUGS IN CANADA PA R T 2 Who is Bearing the Cost? KONG KHOO, ROSEMARY COLUCCI, WILLIAM HRYNIUK, ROBERT KAMINO, TANIA REDINA AND COLLEEN SAVAGE Introduction Last year we reported
Bladder Cancer (Urothelial) Pathways
 Bladder Cancer (Urothelial) Pathways Patient Name: Date of Birth: Member Number: Treatment Start Date: ICD-10 Code: Pathology: Stage: 0a 0is I II III IV Recurrent Line of Treatment: Neoadjuvant/Pre-Op
Bladder Cancer (Urothelial) Pathways Patient Name: Date of Birth: Member Number: Treatment Start Date: ICD-10 Code: Pathology: Stage: 0a 0is I II III IV Recurrent Line of Treatment: Neoadjuvant/Pre-Op
SCI. SickKids-Caribbean Initiative Enhancing Capacity for Care in Paediatric Cancer and Blood Disorders
 1.0 Introduction The (SCI) is a not-for-profit collaboration between the Hospital for Sick Children (SickKids), Toronto, Canada, and seven Caribbean health care institutions across six countries that strive
1.0 Introduction The (SCI) is a not-for-profit collaboration between the Hospital for Sick Children (SickKids), Toronto, Canada, and seven Caribbean health care institutions across six countries that strive
COST CONSIDERATIONS Union for International Cancer Control 2014 Review of Cancer Medicines on the WHO List of Essential Medicines!!!!!!!!!
 UICCEMLCostingScenarios BackoftheEnvelope Calculations PreparedforWorkingGroupSession:19621November2014,Geneva MethodsSummary We have chosen a conservative approach, calculating cost per vial. We have
UICCEMLCostingScenarios BackoftheEnvelope Calculations PreparedforWorkingGroupSession:19621November2014,Geneva MethodsSummary We have chosen a conservative approach, calculating cost per vial. We have
Azacitidine Vidaza Non-transplant myelodysplastic syndrome Funded Funded Funded Funded Funded Funded Not Funded
 Provincial Fundin Summary The interim Joint Oncoloy Dru Review (ijodr) was the precursor oncoloy dru review process prior to pcodr, which provided evidence-based recommendation for cancer treatments from
Provincial Fundin Summary The interim Joint Oncoloy Dru Review (ijodr) was the precursor oncoloy dru review process prior to pcodr, which provided evidence-based recommendation for cancer treatments from
Ovarian Cancer. compendia TREATMENT OF
 drug & compendia TREATMENT OF Ovarian Cancer With each publication, ManagedCare Oncology s drug & Compendia highlights a single medication or a group of medications that could be utilized in the management
drug & compendia TREATMENT OF Ovarian Cancer With each publication, ManagedCare Oncology s drug & Compendia highlights a single medication or a group of medications that could be utilized in the management
 Exhibit B United States Patent Application 20020012663 Kind Code A1 Waksal, Harlan W. January 31, 2002 Treatment of refractory human tumors with epidermal growth factor receptor antagonists Abstract A
Exhibit B United States Patent Application 20020012663 Kind Code A1 Waksal, Harlan W. January 31, 2002 Treatment of refractory human tumors with epidermal growth factor receptor antagonists Abstract A
DRUG PROPERTIES YOU NEED TO KNOW
 Dr. Janet Fitzakerley Summer 2013 Med 6541 Hematopoiesis and Host Defences jfitzake@d.umn.edu www.d.umn.edu/~jfitzake Page 1 of 11 DRUG PROPERTIES YOU NEED TO KNOW 1. Mechanism of action a. chemical class
Dr. Janet Fitzakerley Summer 2013 Med 6541 Hematopoiesis and Host Defences jfitzake@d.umn.edu www.d.umn.edu/~jfitzake Page 1 of 11 DRUG PROPERTIES YOU NEED TO KNOW 1. Mechanism of action a. chemical class
Form 2023 R2.0: Ovarian Cancer Pre-HSCT Data
 Key Fields Sequence Number Date Received: - - CIBMTR Center Number: CIBMTR Recipient ID: Today's Date: - - Date of HSCT for which this form is being completed: - - HSCT type: (check all that apply) Autologous
Key Fields Sequence Number Date Received: - - CIBMTR Center Number: CIBMTR Recipient ID: Today's Date: - - Date of HSCT for which this form is being completed: - - HSCT type: (check all that apply) Autologous
Protocol Number Tumour Group Protocol Name on NCCP website 22/02/ Lung Afatinib Monotherapy 244 Gastrointestinal Regorafenib Monotherapy
 Last Updated 22-Feb-18 Date of last update Protocol Number Tumour Group Protocol Name on NCCP website 22/02/2018 221 Afatinib Monotherapy 244 Gastrointestinal Regorafenib Monotherapy 249 Gynaecology Intrathecal
Last Updated 22-Feb-18 Date of last update Protocol Number Tumour Group Protocol Name on NCCP website 22/02/2018 221 Afatinib Monotherapy 244 Gastrointestinal Regorafenib Monotherapy 249 Gynaecology Intrathecal
REGULATORY INFORMATION
 REGULATORY*INFORMATION/*CANCER*MEDICINES* Summary'of'regulatory'status'of'the'medicines' As#recommended#in#the#EML#Secretariat s#instructions#regarding#applications#for#the#addition#of#new# medicines#
REGULATORY*INFORMATION/*CANCER*MEDICINES* Summary'of'regulatory'status'of'the'medicines' As#recommended#in#the#EML#Secretariat s#instructions#regarding#applications#for#the#addition#of#new# medicines#
Protocol Number Intrathecal Methotrexate for CNS 01/02/2018 Prophylaxis in GTN Gynaecology 249
 Last updated Feb 9, 2018 Revision due Protocol Name on NCCP website Tumour Group Protocol Number Intrathecal Methotrexate for CNS 01/02/2018 Prophylaxis in GTN Gynaecology 249 Two Day Etoposide CISplatin
Last updated Feb 9, 2018 Revision due Protocol Name on NCCP website Tumour Group Protocol Number Intrathecal Methotrexate for CNS 01/02/2018 Prophylaxis in GTN Gynaecology 249 Two Day Etoposide CISplatin
APPHON/ROPPHA Guideline for the Prevention and Management of Chemotherapy Induced Nausea and Vomiting in Children with Cancer
 APPHON/ROPPHA Guideline for the Prevention and Management of Chemotherapy Induced Nausea and Vomiting in Children with Cancer 5850/5980 University Avenue, PO Box 9700, Halifax, N.S. B3K 6R8 PEDIATRIC HEMATOLOGY/ONCOLOGY
APPHON/ROPPHA Guideline for the Prevention and Management of Chemotherapy Induced Nausea and Vomiting in Children with Cancer 5850/5980 University Avenue, PO Box 9700, Halifax, N.S. B3K 6R8 PEDIATRIC HEMATOLOGY/ONCOLOGY
Drug Name. J0129 Injection, abatacept (Orencia ), 10 mg Effective 01/01/2014. J0178 Injection, aflibercept (Eylea ), 1 mg Effective 04/01/2015
 J0129 Injection, abatacept (Orencia ), 10 J0178 Injection, aflibercept (Eylea ), 1 J0256 J0257 J0585 J0586 J0587 J0588 J0597 J0641 J0717 J0800 Injection, alpha 1-proteinase inhibitor, human (Aralast NP,
J0129 Injection, abatacept (Orencia ), 10 J0178 Injection, aflibercept (Eylea ), 1 J0256 J0257 J0585 J0586 J0587 J0588 J0597 J0641 J0717 J0800 Injection, alpha 1-proteinase inhibitor, human (Aralast NP,
UNDERWRITING GUIDE. Secure Advantage HOSPITAL COVERAGE. The Insurance Policy That Pays You Cash In The Event Of A Covered Hospitalization
 UNDERWRITING GUIDE Secure Advantage HOSPITAL COVERAGE The Insurance Policy That Pays You Cash In The Event Of A Covered Hospitalization TABLE OF CONTENTS Contents Page Secure Advantage Underwriting Guide...2
UNDERWRITING GUIDE Secure Advantage HOSPITAL COVERAGE The Insurance Policy That Pays You Cash In The Event Of A Covered Hospitalization TABLE OF CONTENTS Contents Page Secure Advantage Underwriting Guide...2
Adverse effects of anticancer drugs (Antimetabolites agents, Alkylating agents, Antimicrotubule agents, Miscellaneous agents, Immune therapies and
 35 Adverse effects of anticancer drugs (Antimetabolites agents, Alkylating agents, Antimicrotubule agents, Miscellaneous agents, Immune therapies and Biologically directed therapies ) 1 1- Nausea and vomiting
35 Adverse effects of anticancer drugs (Antimetabolites agents, Alkylating agents, Antimicrotubule agents, Miscellaneous agents, Immune therapies and Biologically directed therapies ) 1 1- Nausea and vomiting
GUIDELINES FOR ANTIEMETIC USE IN ONCOLOGY SUMMARY CLASSIFICATION
 GUIDELINES FOR ANTIEMETIC USE IN ONCOLOGY SUMMARY More than half of all cancer patients experience nausea or vomiting during the course of their treatment. If nausea or vomiting becomes severe enough,
GUIDELINES FOR ANTIEMETIC USE IN ONCOLOGY SUMMARY More than half of all cancer patients experience nausea or vomiting during the course of their treatment. If nausea or vomiting becomes severe enough,
MASCC Guidelines for Antiemetic control: An update
 MASCC / ISOO 17 th International Symposium Supportive Care in Cancer June 30 July 2, 2005 / Geneva, Switzerland MASCC Guidelines for Antiemetic control: An update Sussanne Börjeson, RN, PhD Linköping University,
MASCC / ISOO 17 th International Symposium Supportive Care in Cancer June 30 July 2, 2005 / Geneva, Switzerland MASCC Guidelines for Antiemetic control: An update Sussanne Börjeson, RN, PhD Linköping University,
Leukemia. Treatment of. compendia. Associated ICD-9-CM Codes: Drug & Administration. managedcareoncology.com
 Drug & compendia Treatment of Leukemia With each publication, Managed Care Oncology s Drug & Compendia highlights a single medication or a group of medications that could be utilized in the management
Drug & compendia Treatment of Leukemia With each publication, Managed Care Oncology s Drug & Compendia highlights a single medication or a group of medications that could be utilized in the management
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: Reference Number: CP.CPA.223 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: Reference Number: CP.CPA.223 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy for important regulatory
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Verzenio) Reference Number: CP.PHAR.355 Effective Date: 10.24.17 Last Review Date: 02.19 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this
Clinical Policy: (Verzenio) Reference Number: CP.PHAR.355 Effective Date: 10.24.17 Last Review Date: 02.19 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this
DRUG PROPERTIES YOU NEED TO KNOW
 jfitzake@d.umn.edu www.d.umn.edu/~jfitzake Page 1 of 8 DRUG PROPERTIES YOU NEED TO KNOW 1. Mechanism of action a. chemical class b. resistance 2. Pharmacokinetics 3. Therapeutic uses 4. Major side effects/toxicities
jfitzake@d.umn.edu www.d.umn.edu/~jfitzake Page 1 of 8 DRUG PROPERTIES YOU NEED TO KNOW 1. Mechanism of action a. chemical class b. resistance 2. Pharmacokinetics 3. Therapeutic uses 4. Major side effects/toxicities
1 The Cancer Programs Regulation (AR 242/98) is amended by this Regulation.
 Alberta Regulation 18/2005 Cancer Programs Act AMENDMENT REGULATION Filed: February 22, 2005 For information only: Made by the Minister of Health and Wellness (M.O. 9/2005) on February 17, 2005 pursuant
Alberta Regulation 18/2005 Cancer Programs Act AMENDMENT REGULATION Filed: February 22, 2005 For information only: Made by the Minister of Health and Wellness (M.O. 9/2005) on February 17, 2005 pursuant
Hazard Meds: Navigating the Warning Beacons
 Hazard Meds: Navigating the Warning Beacons Disclosures I have nothing to disclose at this time. Mark Twohey, Pharm.D., BCOP Palmetto Health Richland March 24, 2012 Objectives Understand the reasons for
Hazard Meds: Navigating the Warning Beacons Disclosures I have nothing to disclose at this time. Mark Twohey, Pharm.D., BCOP Palmetto Health Richland March 24, 2012 Objectives Understand the reasons for
Guidelines for the Use of Anti-Emetics with Chemotherapy
 Guidelines for the Use of Anti-Emetics with The purpose of this document is to provide guidance on the rational use of anti-emetics for prevention and treatment of chemotherapy-induced nausea and vomiting
Guidelines for the Use of Anti-Emetics with The purpose of this document is to provide guidance on the rational use of anti-emetics for prevention and treatment of chemotherapy-induced nausea and vomiting
EquiCash UNDERWRITING GUIDE. Equitable &You. The Policy That Pays You Cash In The Event You Have To Be Hospitalized HOSPITAL PLAN
 UNDERWRITING GUIDE EquiCash HOSPITAL PLAN The Policy That Pays You Cash In The Event You Have To Be Hospitalized Rev. 10/13 Equitable &You... Committed To Caring TABLE OF CONTENTS Contents Page EquiCash
UNDERWRITING GUIDE EquiCash HOSPITAL PLAN The Policy That Pays You Cash In The Event You Have To Be Hospitalized Rev. 10/13 Equitable &You... Committed To Caring TABLE OF CONTENTS Contents Page EquiCash
Guideline for Classification of the Acute Emetogenic Potential of Antineoplastic Medication in Pediatric Cancer Patients
 Guideline for Classification of the Acute Emetogenic Potential of Antineoplastic Medication in Pediatric Cancer Patients POGO Antineoplastic Induced Nausea and Vomiting Guideline Development Panel: L.
Guideline for Classification of the Acute Emetogenic Potential of Antineoplastic Medication in Pediatric Cancer Patients POGO Antineoplastic Induced Nausea and Vomiting Guideline Development Panel: L.
Clinical Policy: Lapatinib (Tykerb) Reference Number: CP.PHAR.79 Effective Date: Last Review Date: 11.17
 Clinical Policy: (Tykerb) Reference Number: CP.PHAR.79 Effective Date: 10.01.11 Last Review Date: 11.17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: (Tykerb) Reference Number: CP.PHAR.79 Effective Date: 10.01.11 Last Review Date: 11.17 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important
Chemotherapy Agents 101
 Chemotherapy Agents 101 Jumana Ashy, Pharm D, BCOP Judith Misas, PharmD Candidate 2018 Jeremy Hutchinson, PharmD Sady Castance, PharmD September 12, 2017 1 Learning Objectives At the end of the presentation
Chemotherapy Agents 101 Jumana Ashy, Pharm D, BCOP Judith Misas, PharmD Candidate 2018 Jeremy Hutchinson, PharmD Sady Castance, PharmD September 12, 2017 1 Learning Objectives At the end of the presentation
DOXOrubicin, Cyclophosphamide (AC 60/600) 21 day followed by weekly PACLitaxel (80) Therapy (AC-T) 261 CARBOplatin (AUC4-6) Monotherapy-21 days
 Last updated Oct 17, 2018 Tumour Group Protocol Number Protocol Name on NCCP website Breast 200 Trastuzumab (IV) Monotherapy 21 days 201 Trastuzumab (IV) Monotherapy 7 days 202 DOCEtaxel Monotherapy 100mg/m2
Last updated Oct 17, 2018 Tumour Group Protocol Number Protocol Name on NCCP website Breast 200 Trastuzumab (IV) Monotherapy 21 days 201 Trastuzumab (IV) Monotherapy 7 days 202 DOCEtaxel Monotherapy 100mg/m2
Genetics in Cancer Therapy. Raju Kucherlapati, Ph.D. Harvard Medical School
 Genetics in Cancer Therapy Raju Kucherlapati, Ph.D. Harvard Medical School RK Financial interests I do not intend to discuss an off-label use of a product during this activity AVEO Pharmaceuticals KEW
Genetics in Cancer Therapy Raju Kucherlapati, Ph.D. Harvard Medical School RK Financial interests I do not intend to discuss an off-label use of a product during this activity AVEO Pharmaceuticals KEW
Prevention and Management of chemo-and radiotherapy-induced nausea and vomiting
 Prevention and Management of chemo-and radiotherapy-induced nausea and vomiting Focusing on the updated MASCC/ESMO guidelines Karin Jordan Department of Hematology and Oncology, University of Heidelberg
Prevention and Management of chemo-and radiotherapy-induced nausea and vomiting Focusing on the updated MASCC/ESMO guidelines Karin Jordan Department of Hematology and Oncology, University of Heidelberg
Primary malignant neoplasms, not lymphatic or hematopoietic. Secondary malignant neoplasms (i.e.metastatic) Malignant neoplasm, unknown site
 Supplementary Table 1. ICD-9-CM codes used to define cancer ICD-9 Diagnosis code 140.xx-172.xx 174.xx-195.xx 196.xx 198.xx 199.xx 200.xx-208.xx Description Primary malignant neoplasms, not lymphatic or
Supplementary Table 1. ICD-9-CM codes used to define cancer ICD-9 Diagnosis code 140.xx-172.xx 174.xx-195.xx 196.xx 198.xx 199.xx 200.xx-208.xx Description Primary malignant neoplasms, not lymphatic or
Objectives. Principles of Cancer Treatment. Cancer Treatment Modalities. Cancer Treatment Modalities
 Objectives Principles of Cancer Treatment Juanita Madison, RN, MN, AOCN Oncology Clinical Nurse Specialist Seattle Cancer Care Alliance Describe the principles of cancer treatment Surgery Chemotherapy
Objectives Principles of Cancer Treatment Juanita Madison, RN, MN, AOCN Oncology Clinical Nurse Specialist Seattle Cancer Care Alliance Describe the principles of cancer treatment Surgery Chemotherapy
FREEDOM OF INFORMATION SOUTH EAST SCOTLAND CANCER NETWORK
 Dear Date 06/02/09 Your Ref Our Ref RM/1220 Enquiries to Richard Mutch Extension 89441 Direct Line 0131-536-9441 Direct Fax 0131-536-9009 Email richard.mutch@lhnhslothian.scot.nhs.uk FREEDOM OF INFORMATION
Dear Date 06/02/09 Your Ref Our Ref RM/1220 Enquiries to Richard Mutch Extension 89441 Direct Line 0131-536-9441 Direct Fax 0131-536-9009 Email richard.mutch@lhnhslothian.scot.nhs.uk FREEDOM OF INFORMATION
Clinical Policy: Pemetrexed (Alimta) Reference Number: CP.PHAR.368 Effective Date: Last Review Date: Line of Business: Medicaid
 Clinical Policy: (Alimta) Reference Number: CP.PHAR.368 Effective Date: 10.31.17 Last Review Date: 02.18 Line of Business: Medicaid Coding Implications Revision Log See Important Reminder at the end of
Clinical Policy: (Alimta) Reference Number: CP.PHAR.368 Effective Date: 10.31.17 Last Review Date: 02.18 Line of Business: Medicaid Coding Implications Revision Log See Important Reminder at the end of
UP Health System Marquette Medication Guideline High Alert Drugs
 1 ALTEPLASE SOLR 100 MG ACTIVASE 305010 50242008527 TRUE ALTEPLASE SOLR 50 MG ACTIVASE 305008 50242004413 TRUE AMIODARONE INJ 50 MG/ML CORDARONE IV 305035 63323061609 TRUE AMIODARONE INJ 50 MG/ML CORDARONE
1 ALTEPLASE SOLR 100 MG ACTIVASE 305010 50242008527 TRUE ALTEPLASE SOLR 50 MG ACTIVASE 305008 50242004413 TRUE AMIODARONE INJ 50 MG/ML CORDARONE IV 305035 63323061609 TRUE AMIODARONE INJ 50 MG/ML CORDARONE
Access To Cancer Drugs In Medicare Part D: Formulary Placement And Beneficiary Cost Sharing In 2006
 Access To Cancer Drugs In Medicare Part D: Formulary Placement And eneficiary Cost Sharing In 2006 Medicare Part D beneficiaries have access to many more cancer drugs than ever before, with low cost sharing.
Access To Cancer Drugs In Medicare Part D: Formulary Placement And eneficiary Cost Sharing In 2006 Medicare Part D beneficiaries have access to many more cancer drugs than ever before, with low cost sharing.
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Verzenio) Reference Number: CP.PHAR.355 Effective Date: 10.24.17 Last Review Date: 05.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this
Clinical Policy: (Verzenio) Reference Number: CP.PHAR.355 Effective Date: 10.24.17 Last Review Date: 05.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this
Provider Administered Drug Program (PADP) and Physician Administered Drug VPSS List
 Provider Administered Drug Program (PADP) and Physician Administered Drug VPSS List Code Drug Name Effective and/or Term Date J0129 Injection, abatacept (Orencia ), 10 mg J0178 Injection, aflibercept (Eylea
Provider Administered Drug Program (PADP) and Physician Administered Drug VPSS List Code Drug Name Effective and/or Term Date J0129 Injection, abatacept (Orencia ), 10 mg J0178 Injection, aflibercept (Eylea
Anastrozole. Active Ingredient. Brand Name: Arimidex Strength: 1 mg Dosage Form: Tablets Pack Size: 28 Tabs Company: AstraZeneca
 Super Specialty Drugs by Anastrozole Arimidex 1 mg 28 Tabs AstraZeneca Armotraz 1 mg 10 Tabs Cipla Anastrozole Anabrez 1 mg 100 Tabs Sun Decitabine Dacogen 50 mg 1 Vial Eisai Inc. Erlotinib Erlocip-150
Super Specialty Drugs by Anastrozole Arimidex 1 mg 28 Tabs AstraZeneca Armotraz 1 mg 10 Tabs Cipla Anastrozole Anabrez 1 mg 100 Tabs Sun Decitabine Dacogen 50 mg 1 Vial Eisai Inc. Erlotinib Erlocip-150
Vivida Health Specialty Pharmacy Drugs (Injectable) Prior-Authorization Requirements Effective 1/1/19
 Vivida Health Specialty Pharmacy Drugs (Injectable) Prior-Authorization Requirements Effective 1/1/19 All Non-Par Provider Requests Requires Authorization Regardless of Service J0178 J0180 J0202 J0205
Vivida Health Specialty Pharmacy Drugs (Injectable) Prior-Authorization Requirements Effective 1/1/19 All Non-Par Provider Requests Requires Authorization Regardless of Service J0178 J0180 J0202 J0205
Guideline Update on Antiemetics
 Guideline Update on Antiemetics Clinical Practice Guideline Special Announcements Please check www.asco.org/guidelines/antiemetics for current FDA alert(s) and safety announcement(s) on antiemetics 2 Introduction
Guideline Update on Antiemetics Clinical Practice Guideline Special Announcements Please check www.asco.org/guidelines/antiemetics for current FDA alert(s) and safety announcement(s) on antiemetics 2 Introduction
Committee Approval Date: December 12, 2014 Next Review Date: July 2015
 Medication Policy Manual Policy No: dru378 Topic: Akynzeo, netupitant/palonosetron Date of Origin: December 12, 2014 Committee Approval Date: December 12, 2014 Next Review Date: July 2015 Effective Date:
Medication Policy Manual Policy No: dru378 Topic: Akynzeo, netupitant/palonosetron Date of Origin: December 12, 2014 Committee Approval Date: December 12, 2014 Next Review Date: July 2015 Effective Date:
ALKYLATING AGENTS (22)
 Serving the United States, Canada and North Central America MEMBER OF TEXAS HEALTH CARE ENTITIES A PRIVATE MEMBERSHIP MEDICAL ASSOCIATION 3105 MAIN STREET, ROWLETT, TX 75088 TEL: 214-299-9449 office@rgccusa.com
Serving the United States, Canada and North Central America MEMBER OF TEXAS HEALTH CARE ENTITIES A PRIVATE MEMBERSHIP MEDICAL ASSOCIATION 3105 MAIN STREET, ROWLETT, TX 75088 TEL: 214-299-9449 office@rgccusa.com
Current and Emerging Therapeutic Options in the Management of Chemotherapy-Induced Nausea and Vomiting (CINV) Objectives
 Current and Emerging Therapeutic Options in the Management of Chemotherapy-Induced Nausea and Vomiting (CINV) Susan Urba, M.D. University of Michigan Comprehensive Cancer Center Objectives Mechanisms of
Current and Emerging Therapeutic Options in the Management of Chemotherapy-Induced Nausea and Vomiting (CINV) Susan Urba, M.D. University of Michigan Comprehensive Cancer Center Objectives Mechanisms of
Adjuvant Systemic Therapy in Early Stage Breast Cancer
 Adjuvant Systemic Therapy in Early Stage Breast Cancer Julie R. Gralow, M.D. Director, Breast Medical Oncology Jill Bennett Endowed Professor of Breast Cancer Professor, Global Health University of Washington
Adjuvant Systemic Therapy in Early Stage Breast Cancer Julie R. Gralow, M.D. Director, Breast Medical Oncology Jill Bennett Endowed Professor of Breast Cancer Professor, Global Health University of Washington
Part B payment for drugs in Medicare: Phase 1 of CMS s proposed pilot and its impact on oncology care
 Part B payment for drugs in Medicare: Phase 1 of CMS s proposed pilot and its impact on oncology care Raina H. Jain, Stephen M. Schleicher, Coral L. Atoria, Peter B. Bach Executive Summary The recent pilot
Part B payment for drugs in Medicare: Phase 1 of CMS s proposed pilot and its impact on oncology care Raina H. Jain, Stephen M. Schleicher, Coral L. Atoria, Peter B. Bach Executive Summary The recent pilot
Fundamentals of Pharmacology for Veterinary Technicians Chapter 20
 Figure 20-1 Figure 20-2 Figure 20-3A Figure 20-3B Figure 20-4 Table 20-1 Antineoplastic Agents Used for Chemotherapy in Animals Drug Type Drug Category Examples Cell-Cycle Effect CCNS Alkylating agents:
Figure 20-1 Figure 20-2 Figure 20-3A Figure 20-3B Figure 20-4 Table 20-1 Antineoplastic Agents Used for Chemotherapy in Animals Drug Type Drug Category Examples Cell-Cycle Effect CCNS Alkylating agents:
CPT Service Description Effective Date
 Medical Oncology Program Review Code List 2 nd Quarter 2018 This list is subject to change once per quarter. Changes will be posted to the BCBSNC website at www.bcbsnc.com by the 10th day of January, April,
Medical Oncology Program Review Code List 2 nd Quarter 2018 This list is subject to change once per quarter. Changes will be posted to the BCBSNC website at www.bcbsnc.com by the 10th day of January, April,
Part B payment for drugs in Medicare 0
 Part B payment for drugs in Medicare 0 Introduction: The recent pilot proposal 1 on Part B drug payment from the Center for Medicare and Medicaid Innovation of CMS has met strong resistance 2 from the
Part B payment for drugs in Medicare 0 Introduction: The recent pilot proposal 1 on Part B drug payment from the Center for Medicare and Medicaid Innovation of CMS has met strong resistance 2 from the
Treatment Options for Breast Cancer in Low- and Middle-Income Countries: Adjuvant and Metastatic Systemic Therapy
 Women s Empowerment Cancer Advocacy Network (WE CAN) Conference Bucharest, Romania October 2015 Treatment Options for Breast Cancer in Low- and Middle-Income Countries: Adjuvant and Metastatic Systemic
Women s Empowerment Cancer Advocacy Network (WE CAN) Conference Bucharest, Romania October 2015 Treatment Options for Breast Cancer in Low- and Middle-Income Countries: Adjuvant and Metastatic Systemic
Subject: Palonosetron Hydrochloride (Aloxi )
 09-J0000-87 Original Effective Date: 02/15/09 Reviewed: 07/09/14 Revised: 03/15/18 Subject: Palonosetron Hydrochloride (Aloxi ) THIS MEDICAL COVERAGE GUIDELINE IS NOT AN AUTHORIZATION, CERTIFICATION, EXPLANATION
09-J0000-87 Original Effective Date: 02/15/09 Reviewed: 07/09/14 Revised: 03/15/18 Subject: Palonosetron Hydrochloride (Aloxi ) THIS MEDICAL COVERAGE GUIDELINE IS NOT AN AUTHORIZATION, CERTIFICATION, EXPLANATION
Prior Authorization Proposal: Oncology Agents
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
ASSESSMENT OF THE PAEDIATRIC NEEDS CHEMOTHERAPY PRODUCTS (PART I) DISCLAIMER
 European Medicines Agency Evaluation of Medicines for Human Use London, September 2006 Doc. Ref.: EMEA/384641/2006 ASSESSMENT OF THE PAEDIATRIC NEEDS CHEMOTHERAPY PRODUCTS (PART I) DISCLAIMER The Paediatric
European Medicines Agency Evaluation of Medicines for Human Use London, September 2006 Doc. Ref.: EMEA/384641/2006 ASSESSMENT OF THE PAEDIATRIC NEEDS CHEMOTHERAPY PRODUCTS (PART I) DISCLAIMER The Paediatric
Medications currently available to treat Multiple Myeloma include: Current Code Price (AWP) Effective Date. Code Price (AWP)
 drug and administration compendia Compendia information developed and maintained by With each publication Managed Care Oncology s Drug & Compendia will highlight a single medication or a group of medications
drug and administration compendia Compendia information developed and maintained by With each publication Managed Care Oncology s Drug & Compendia will highlight a single medication or a group of medications
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Zofran, Zuplenz) Reference Number: CP.CPA.173 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Medicaid Medi-Cal Revision Log See Important Reminder at the end of this
Clinical Policy: (Zofran, Zuplenz) Reference Number: CP.CPA.173 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Medicaid Medi-Cal Revision Log See Important Reminder at the end of this
Safe Practices in Oral Anticancer Medications: Implications for Community Pharmacy
 Safe Practices in Oral Anticancer Medications: Implications for Community Pharmacy Faculty Disclosures Dr. Holle has provided content expertise on patient education materials for Innocrin Pharmaceuticals
Safe Practices in Oral Anticancer Medications: Implications for Community Pharmacy Faculty Disclosures Dr. Holle has provided content expertise on patient education materials for Innocrin Pharmaceuticals
INJECTION, INOTUZUMAB OZOGAMICIN, 0.1 MG [BESPONSA ] [C CODES FOR FACILITY USE ONLY]
![INJECTION, INOTUZUMAB OZOGAMICIN, 0.1 MG [BESPONSA ] [C CODES FOR FACILITY USE ONLY] INJECTION, INOTUZUMAB OZOGAMICIN, 0.1 MG [BESPONSA ] [C CODES FOR FACILITY USE ONLY]](/thumbs/85/92112268.jpg) Commercial Medical Oncology Program Review Code List 3rd Quarter 2018 This list is subject to change once per quarter. Changes will be posted to the BCBSNC website at www.bcbsnc.com by the 10th day of
Commercial Medical Oncology Program Review Code List 3rd Quarter 2018 This list is subject to change once per quarter. Changes will be posted to the BCBSNC website at www.bcbsnc.com by the 10th day of
AD regimen, 576 adrenal steroid inhibitor. aminoglutethimide, 23 25
 A 640 Index Index abiraterone acetate, 5 7 Abraxane. See albuminbound paclitaxel ABVD regimen, 57, 540 ABV regimen, 519 520 AC docetaxel regimen, 476, 478 acetaminophen busulfan and, 71 AC regimen, 475,
A 640 Index Index abiraterone acetate, 5 7 Abraxane. See albuminbound paclitaxel ABVD regimen, 57, 540 ABV regimen, 519 520 AC docetaxel regimen, 476, 478 acetaminophen busulfan and, 71 AC regimen, 475,
Adult Intravenous Systemic Anticancer Therapy (SACT) Section A. SUMMARY of SCHEME QIPP Reference
 CA2 Nationally standardised Dose banding for Adult Intravenous Anticancer Therapy (SACT) Scheme Name CA2: Nationally Standardised Dose Banding for Adult Intravenous Systemic Anticancer Therapy (SACT) Section
CA2 Nationally standardised Dose banding for Adult Intravenous Anticancer Therapy (SACT) Scheme Name CA2: Nationally Standardised Dose Banding for Adult Intravenous Systemic Anticancer Therapy (SACT) Section
CANCER CHEMOTHERAPY Michael Lea
 CANCER CHEMOTHERAPY 2010 Michael Lea Cancer Chemotherapy - Lecture Outline 1. Targets for cancer chemotherapy 2. Classification of anticancer drugs 3. Cell cycle specificity 4. Drug resistance 5. New approaches
CANCER CHEMOTHERAPY 2010 Michael Lea Cancer Chemotherapy - Lecture Outline 1. Targets for cancer chemotherapy 2. Classification of anticancer drugs 3. Cell cycle specificity 4. Drug resistance 5. New approaches
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Stivarga) Reference Number: CP.CPA.157 Effective Date: 11.16.17 Last Review Date: 11.17 Line of Business: Medicaid Medi-Cal Revision Log See Important Reminder at the end of this policy
Clinical Policy: (Stivarga) Reference Number: CP.CPA.157 Effective Date: 11.16.17 Last Review Date: 11.17 Line of Business: Medicaid Medi-Cal Revision Log See Important Reminder at the end of this policy
DRUGS YOU NEED TO KNOW
 jfitzake@d.umn.edu Page 1 of 7 DRUGS YOU NEED TO KNOW ALKYLATING AGENTS (blue cards) BUSULFAN CARMUSTINE (BCNU) CYCLOPHOSPHAMIDE DACARBAZINE LOMUSTINE (CCNU) MECHLORETHAMINE MELPHALAN THIOTEPA NATURAL
jfitzake@d.umn.edu Page 1 of 7 DRUGS YOU NEED TO KNOW ALKYLATING AGENTS (blue cards) BUSULFAN CARMUSTINE (BCNU) CYCLOPHOSPHAMIDE DACARBAZINE LOMUSTINE (CCNU) MECHLORETHAMINE MELPHALAN THIOTEPA NATURAL
Part B payment for drugs in Medicare 0
 Part B payment for drugs in Medicare 0 Introduction: The recent pilot proposal 1 on Part B drug payment from the Center for Medicare and Medicaid Innovation of CMS has met strong resistance 2 from the
Part B payment for drugs in Medicare 0 Introduction: The recent pilot proposal 1 on Part B drug payment from the Center for Medicare and Medicaid Innovation of CMS has met strong resistance 2 from the
through the cell cycle. However, how we administer drugs also depends on the combinations that we give and the doses that we give.
 Hello and welcome to this lecture. My name is Hillary Prescott. I am a Clinical Pharmacy Specialist at The University of Texas MD Anderson Cancer Center. My colleague, Jeff Bryan and I have prepared this
Hello and welcome to this lecture. My name is Hillary Prescott. I am a Clinical Pharmacy Specialist at The University of Texas MD Anderson Cancer Center. My colleague, Jeff Bryan and I have prepared this
ERLEADA (apalutamide) oral tablet
 ERLEADA (apalutamide) oral tablet Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy Coverage
ERLEADA (apalutamide) oral tablet Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy Coverage
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Lynparza) Reference Number: CP.PHAR.360 Effective Date: 10.03.17 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this
Clinical Policy: (Lynparza) Reference Number: CP.PHAR.360 Effective Date: 10.03.17 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Revision Log See Important Reminder at the end of this
Supplementary Figures
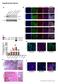 Supplementary Figures Supplementary Figure 1. Characterization of the generated hipsc lines a) Western blot analysis indicating the efficient downregulation of p53 upon knockdown in three hipsc lines (
Supplementary Figures Supplementary Figure 1. Characterization of the generated hipsc lines a) Western blot analysis indicating the efficient downregulation of p53 upon knockdown in three hipsc lines (
Emetogenicity level 1. Emetogenicity level 2
 Emetogenicity level 1 15 mins Pre-Chemo Maxalon 10mg po During chemo and Post Chemo 3 days Maxalon10mg po 8 hourly Increase Maxalon 20mg po 8 hourly Change to Cyclizine 50mg po 8 hourly 3 days If nausea
Emetogenicity level 1 15 mins Pre-Chemo Maxalon 10mg po During chemo and Post Chemo 3 days Maxalon10mg po 8 hourly Increase Maxalon 20mg po 8 hourly Change to Cyclizine 50mg po 8 hourly 3 days If nausea
Additional information to support. The National Patient Safety Agency s Rapid Response Report Risks of incorrect dosing of oral anti-cancer medicines
 Additional information to support The National Patient Safety Agency s Rapid Response Report Risks of incorrect dosing of oral anti-cancer medicines Reference: NPSA/2008/RRR001 - issued on 22 January 2008
Additional information to support The National Patient Safety Agency s Rapid Response Report Risks of incorrect dosing of oral anti-cancer medicines Reference: NPSA/2008/RRR001 - issued on 22 January 2008
Job title: Consultant Pharmacist/Advanced Practice Pharmacist
 Title : Guidelines for the Use of Antiemetics Purpose: To provide trust-wide guidance on the safe and effective use of antiemetics for the prevention and treatment of chemotherapy and radiotherapy induced
Title : Guidelines for the Use of Antiemetics Purpose: To provide trust-wide guidance on the safe and effective use of antiemetics for the prevention and treatment of chemotherapy and radiotherapy induced
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Stivarga) Reference Number: CP.PHAR.107 Effective Date: 12.01.12 Last Review Date: 05.18 Line of Business: Commercial, HIM, Medicaid Revision Log See Important Reminder at the end of
Clinical Policy: (Stivarga) Reference Number: CP.PHAR.107 Effective Date: 12.01.12 Last Review Date: 05.18 Line of Business: Commercial, HIM, Medicaid Revision Log See Important Reminder at the end of
PHARMACY PEARLS CHANIN WRIGHT, PHARMD
 PHARMACY PEARLS CHANIN WRIGHT, PHARMD OBJECTIVES From the pearls heard, discuss with someone near you What is something you can apply to your setting Traumatic brain injury in children Goals Correct and
PHARMACY PEARLS CHANIN WRIGHT, PHARMD OBJECTIVES From the pearls heard, discuss with someone near you What is something you can apply to your setting Traumatic brain injury in children Goals Correct and
ORAL ONCOLOGY CRITERIA
 ORAL ONCOLOGY CRITERIA LENGTH OF AUTHORIZATION: Varies; Maximum of one year REVIEW CRITERIA: Drug Name Indication & Dosage Age Limit Quantity per day AFINITOR (everolimus) AFINITOR DISPERZ (everolimus)
ORAL ONCOLOGY CRITERIA LENGTH OF AUTHORIZATION: Varies; Maximum of one year REVIEW CRITERIA: Drug Name Indication & Dosage Age Limit Quantity per day AFINITOR (everolimus) AFINITOR DISPERZ (everolimus)
Haematology, Oncology and Palliative Care Directorate.
 Anticancer Treatment for Administration on the Somerset Mobile Chemotherapy Unit The table below details the suitability of different types of anticancer treatment for administration on the Somerset Mobile
Anticancer Treatment for Administration on the Somerset Mobile Chemotherapy Unit The table below details the suitability of different types of anticancer treatment for administration on the Somerset Mobile
4. Shown below is the structure of doxorubicin (Adriamycin). What is true about this agent?
 Midterm 2: 3 points each (except final question worth 1 point 1. A useful regimen for treating colorectal cancer is FOLFIRI. What is true about this regimen? A. The regimen includes folinic acid, 5-fluorouracil
Midterm 2: 3 points each (except final question worth 1 point 1. A useful regimen for treating colorectal cancer is FOLFIRI. What is true about this regimen? A. The regimen includes folinic acid, 5-fluorouracil
Hazardous Medication List
 Hazardous Medication List REDUCING OCCUPATIONAL EXPOSURE TO HAZARDOUS MEDICATION FOR ALL STAFF Table of Contents Hazardous Medication List Key Points... 1 Hazardous Medication List... 2 Special Handling
Hazardous Medication List REDUCING OCCUPATIONAL EXPOSURE TO HAZARDOUS MEDICATION FOR ALL STAFF Table of Contents Hazardous Medication List Key Points... 1 Hazardous Medication List... 2 Special Handling
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Self Administered Oncology Agents Page 1 of 13 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Self Administered Oncology Agents Prime Therapeutics will review Prior
Self Administered Oncology Agents Page 1 of 13 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Self Administered Oncology Agents Prime Therapeutics will review Prior
CancerPACT Cancer Patients Alliance for Clinical Trials
 TM CancerPACT Cancer Patients Alliance for Clinical Trials Listing of Ongoing Cancer Clinical Trials in the Sacramento Area Winter 2008 I. Solid Tumors 1. Breast p.1 2. Central Nervous System p.2 3. Gastrointestinal
TM CancerPACT Cancer Patients Alliance for Clinical Trials Listing of Ongoing Cancer Clinical Trials in the Sacramento Area Winter 2008 I. Solid Tumors 1. Breast p.1 2. Central Nervous System p.2 3. Gastrointestinal
Chapter 2 Clinical Anticancer Drugs for Cancer Treatment
 Chapter 2 Clinical Anticancer Drugs for Cancer Treatment Besides cytoreductive surgery and radiotherapy, chemotherapy is the most widely used therapeutic strategy in combating cancer. The high incidence
Chapter 2 Clinical Anticancer Drugs for Cancer Treatment Besides cytoreductive surgery and radiotherapy, chemotherapy is the most widely used therapeutic strategy in combating cancer. The high incidence
Generic (Brand) Strength & Dosage form Fml Limit Cost per Rx Notes 5-HT3 Antagonists
 MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY Nausea LAST REVIEW 9/11/2018 THERAPEUTIC CLASS Gastrointestinal Disorders REVIEW HISTORY 12/16, 11/15, 11/07 LOB AFFECTED
MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY Nausea LAST REVIEW 9/11/2018 THERAPEUTIC CLASS Gastrointestinal Disorders REVIEW HISTORY 12/16, 11/15, 11/07 LOB AFFECTED
