MEDICAL POLICY EFFECTIVE DATE: 05/19/11 REVISED DATE: 05/24/12, 05/23/13 ARCHIVED DATE: 05/22/14 EDITED DATE: 05/28/15, 05/25/16, 05/18/17, 05/17/18
|
|
|
- Felicity Fields
- 6 years ago
- Views:
Transcription
1 MEDICAL POLICY SUBJECT: NON-INVASIVE HELICOBACTER PAGE: 1 OF: 6 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including an Essential Plan product, covers a specific service, medical policy criteria apply to the benefit. If a Medicare product covers a specific service, and there is no national or local Medicare coverage decision for the service, medical policy criteria apply to the benefit. POLICY STATEMENT: Based upon our criteria and assessment of the peer-reviewed literature: I. Testing for H pylori infection using either an urea breath test (UBT 13 C or 14 C) or a stool antigen test (HpSA ) has been medically proven to be effective and is medically appropriate for the following: A. Patients, aged 55 years or younger, with uninvestigated dyspeptic symptoms who have no alarm features suggestive of cancer or ulcer complications (e.g., bleeding, anemia, unexplained weight loss, vomiting, dysphagia); B. Determining eradication after antibiotic therapy in any of the following circumstances: 1. Patients with active peptic ulcer disease (PUD) or who have received treatment for H. pylori PUD; 2. Patients with persistent dyspeptic symptoms after an appropriate course of treatment; 3. Patients with associated mucosa-associated lymphoid tissue (MALT) lymphoma; or 4. Patients who have undergone resection for early gastric cancer. C. As part of the preoperative work-up for patients undergoing a bariatric procedure. II. Screening for H. pylori infection in the absence of upper gastrointestinal symptoms is considered not medically necessary (except as stated above). III. Simultaneous or concurrent testing using UBT and HpSA is considered not medically necessary. Refer to Administrative policy #AP-11, Helicobacter Pylori (H. pylori) Serology Testing. POLICY GUIDELINES: I. The American College of Gastroenterology guidelines recommend that diagnostic testing for H. pylori infection should only be performed if treatment is intended for positive results. II. Dyspepsia associated with alarm features (e.g., bleeding, anemia, unexplained weight loss, vomiting, dysphagia, odynophagia, early satiety, family history of gastrointestinal cancer, previous esophagogastric malignancy) or new onset dyspepsia symptoms in persons older than age 55 years usually requires an upper endoscopy. III. When confirmation of eradication is necessary, testing should be performed no sooner than 4 weeks after completion of treatment. DESCRIPTION: Helicobacter pylori (H. pylori) is a spiral shaped bacterium that is found in the gastric mucus layer or adherent to the epithelial lining of the stomach. Helicobacter pylori (H. pylori) remains one of the most common worldwide human infections and is associated with a number of important upper gastrointestinal (GI) conditions including chronic gastritis, peptic ulcer disease, and gastric malignancy. The pathogenic role of H. pylori in peptic ulcer disease, both duodenal and gastric, is well-recognized. Nearly 95% of patients with duodenal ulcers and 80 % of patients with gastric ulcers are found to be infected with H. pylori. Dyspepsia is clinically defined as nausea, epigastric pain or discomfort experienced on more than seven days of a fourweek period. Factors that affect the management of dyspepsia include the patient s age, routine use of NSAIDs, and presence of any alarm symptoms. Alarm symptoms are identified as melena, hematemesis, persistent vomiting, A nonprofit independent licensee of the BlueCross BlueShield Association
2 PAGE: 2 OF: 6 anemia, acute onset of total dysphagia or involuntary weight loss greater than 5%. The test and-treat strategy for H. pylori has been endorsed for the management of uninvestigated dyspepsia by a number of organizations, including the American Gastroenterological Association and the American College of Gastroenterology. The methods of diagnostic testing for H. pylori can be divided into those that do and those that do not require endoscopy. Endoscopic methods for testing include histology, rapid urease testing, culture and polymerase chain reaction (not widely available for clinical use in the United States). Nonendoscopic diagnostic tests include: antibody tests, urea breath tests, and stool/fecal antigen tests. Antibody testing relies upon the detection of IgG antibodies specific to H. pylori in serum, whole blood, or urine. IgG antibodies to H. pylori typically become present approximately 21 days after infection and can remain present long after eradication. The urea breath test identifies active H. pylori infection by way of the organism s urease activity. In a UBT, the patient is given an oral preparation of either nonradioisotope carbon-13- (13C-) labeled urea, or radioactive isotope carbon-14- (14C-) labeled urea. In the presence of H. pylori infection, bacterial urease metabolizes the urea to produce labeled carbon dioxide (CO 2 ) and ammonia. The labeled carbon diffuses into the bloodstream and is excreted by the lungs. Patients are required to be off anti-microbials and bismuth for 2 weeks prior to UBT testing. Fasting for one hour prior to testing is also required. The stool /fecal antigen test is based on the passage of H. pylori bacteria and antigens in the gastrointestinal tract, identifies H. pylori antigen in the stool by enzyme immunoassay with the use of polyclonal anti-h. pylori antibody. If stool antigen testing is used, no special requirements are needed by the patient such as fasting or stopping medications. The American College of Gastroenterology no longer recommends serology for the detection of H pylori infection. Several factors limit the usefulness of antibody testing in clinical practice. A meta-analysis evaluated the performance characteristics of several commercially available quantitative serological assays and found their overall sensitivity and specificity to be 85% and 79%, respectively, with no differences between the different assays. It is very important to understand that the positive predictive value (PPV) of antibody testing is greatly influenced by the prevalence of H. pylori infection. In regions where the prevalence of H. pylori is high, such as urban areas or communities with large immigrant populations, the PPV is reasonably good. However, in a community setting with a prevalence of approximately 20% as is the case in much of the United States, though a negative antibody test suggests the absence of infection, a positive test has no value in predicting the presence of an active infection. Therefore in low prevalence populations, antibody tests should be avoided. Further, antibody tests developed using antigens from one region of the world may not perform well when applied to patients in another part of the world. Finally, antibody tests are of little benefit in documenting eradication as results can remain positive for years following successful cure of the infection. RATIONALE: UBT The UBT Breath Collection Kit has been cleared for marketing by the FDA. Exalenz Bioscience Ltd has also obtained FDA approval for marketing its BreathID system for the detection of H pylori bacteria. UBT systems are intended for use in the qualitative detection of H. pylori and as an aid in the initial diagnosis and post-treatment monitoring of H. pylori infection in pediatric patients and adult patients (e.g., age 3 and older). The test may be used to monitor treatment if used at least four weeks following completion of therapy. Esophagogastroduodenal (EGD) endoscopy with biopsy is considered the reference method for the diagnosis of Helicobacter pylori (H. pylori). The overall body of literature suggests that noninvasive testing with the urea breath test (UBT) is as effective as endoscopy in managing select patients with uncomplicated upper gastrointestinal symptoms. Overall, the sensitivity and specificity found in studies investigating the diagnostic performance of UBTs have been found to be exceeding 95% in most studies. Test reproducibility has been found to be excellent. The UBT also provides an accurate means of post-treatment testing. HpSA HpSA has been cleared by the FDA for use in both pediatric patients and adult patients. H. pylori stool antigen (HpSA ) testing provides an acceptable alternative to UBT and is FDA cleared for use in the initial diagnosis,
3 PAGE: 3 OF: 6 therapeutic monitoring, eradication confirmation both adults and children. Reported sensitivity and specificity found in studies are 96.1% and 95.7%, respectively. When testing for H. pylori in populations with a low pretest probability of infection, the HpSA provides greater accuracy than serologic testing with only a modest increase in incremental costs. CODES: Number Description Eligibility for reimbursement is based upon the benefits set forth in the member s subscriber contract. CODES MAY NOT BE COVERED UNDER ALL CIRCUMSTANCES. PLEASE READ THE POLICY AND GUIDELINES STATEMENTS CAREFULLY. Codes may not be all inclusive as the AMA and CMS code updates may occur more frequently than policy updates. Code Key: Experimental/Investigational = (E/I), Not medically necessary/ appropriate = (NMN). CPT: Urea breath test, C-14 (isotopic); acquisition for analysis HCPCS: analysis Helicobacter pylori; breath test analysis for urease activity, non-radioactive (e.g., C-13) drug administration and sample collection Infectious agent antigen detection by enzyme immunoassay technique qualitative or semiquantitative, multiple step method; Helicobacter pylori, stool No specific code(s) Copyright 2018 American Medical Association, Chicago, IL ICD10: B96.81 Helicobacter pylori (H. pylori) as the cause of diseases classified elsewhere C16.0-C16.9 Malignant neoplasm stomach (code range) C82.50 Diffuse follicle center lymphoma, unspecified site C82.59 Diffuse follicle center lymphoma, extranodal and solid organ sites C84.90 Mature T/NK-cell lymphomas, unspecified, unspecified site C84.99 Mature T/NK-cell lymphomas, unspecified, extranodal and solid organ sites C84.A0 C84.Z0 C84.Z9 Cutaneous T-cell lymphoma, unspecified, unspecified site Other mature T/NK-cell lymphomas, unspecified site Other mature T/NK-cell lymphomas, extranodal and solid organ sites C85.10 Unspecified B-cell lymphoma, unspecified site C85.19 Unspecified B-cell lymphoma, extranodal and solid organ sites C85.20 Mediastinal (thymic) large B-cell lymphoma, unspecified site C85.29 Mediastinal (thymic) large B-cell lymphoma, extranodal and solid organ sites C85.80 Other specified types of non-hodgkin lymphoma, unspecified site C85.89 Other specified types of non-hodgkin lymphoma, extranodal and solid organ sites C85.90 Non-Hodgkin lymphoma, unspecified, unspecified site C85.99 Non-Hodgkin lymphoma, unspecified, extranodal and solid organ sites C86.4 Blastic NK-cell lymphoma
4 PAGE: 4 OF: 6 K25.0-K25.9 K26.0-K26.9 K27.0-K27.9 K28.0-K28.9 K K29.91 K30 REFERENCES: Gastric Ulcer (code range) Duodenal Ulcer (code range) Peptic Ulcer (code range) Gastrojejunal Ulcer (code range) Gastritis (code range) Functional dyspepsia Z87.11 Personal history of peptic ulcer disease This medical policy had previously been deleted in 2005 based upon an administrative decision. This medical policy was reactivated and underwent an extensive update in 2011 based on a new Utilization Management initiative. Ables AZ, et al. Update on helicobacter pylori treatment. Amer Fam Physician 2007 [ accessed 4/5/18. *Aksoy DY, et al. Evaluation of helicobacter pylori stool antigen test (HpSA) for the detection of helicobacter pylori infection and comparison with other methods. Hepatogastroenterol 2003 Jul-Aug;50(52): *Arents NL, et al. The accuracy of the Helicobacter Pylori stool antigen test in diagnosing H. Pylori in treated and untreated patients. Eur J Gastroenterol Hepatol 2001 Apr;13(4): *BlueCross BlueShield Association. Urea breath test for diagnosis of Helicobacter pylori. Medical Policy Reference Manual Policy # Jul 17; archived 2010 Nov 10. *Chey WD, et al. American College of Gastroenterology guideline on the management of Helicobacter pylori infection. Am J Gastroenterol 2007 Aug;102(8): *Chey WD, et al. A comparison of three fingerstick, whole blood antibody tests for helicobacter pylori infection: a United States, multicenter trial. Am J Gastroenterol 1999 Jun;94(6): Crowley E, et al. How to use Helicobacter pylori testing in paediatric practice. Arch Dis Child Educ Pract Edu 2013 Feb;98(1): Delaney BC, et al. Helicobacter pylori test and treat versus proton pump inhibitor in initial treatment of dyspepsia in primary care: multicentre randomized controlled trial(mrc-cube trial). BMJ 2008 Mar 22;336(7645): *Delaney B, et al. Initial management strategies for dyspepsia. Cochrane Database Syst Rev 2005(4):CD *Delaney BC, et al. Randomised controlled trial of Helicobacter pylori testing and endoscopy for dyspepsia in primary care. BMJ 2001 Apr 14;322(7291): *Elitsur Y, et al. Stool antigen test for diagnosis of helicobacter pylori infection in children with symptomatic disease: a prospective study. J Pediatr Gastroenterol Nutr 2004 Jul;39(1):64-7. Elitsur Y, et al. Urea breath test in children: the United States prospective, multicenter study. Helicobacter 2009 Apr;14(2): *Garza-Gonzalez E, et al. Comparison of endoscopy-based and serum-based methods for the diagnosis of helicobacter pylori. Can J Gastroenterol 2003 Feb;17(2): Graham DY. Efficient identification and evaluation of effective helicobacter pylori therapies. Clin Gastroenterol Hepatol 2009 Feb;7(2): Graham DY, et al. Clinical practice: diagnosis and evaluation of dyspepsia. J Clin Gastroenterol 2010 Mar;44(3):
5 PAGE: 5 OF: 6 Guarner J, et al. Helicobacter pylori diagnostic tests in children: review of the literature form Eur J Pediatr 2010 Jan;169(1): *Institute of Clinical Systems Improvement (ICSI). Health care guideline: Initial management of dyspepsia and GERD [ accessed 4/15/14. *Jarbol DE, et al. proton pump inhibitor or testing for Helicobacter pylori as the first step for patients presenting with dyspepsia? A cluster-randomized trial. Am J Gastroenterol 2006 Jun;101(6): Journal of Pediatric Gastroenterology and Nutrition. Evidenced-based guidelines from ESGPHAN and NASPGHAN for Helicobacter pylori infection in children. [ accessed 4/5/18. Leal YA, et al. 13C-urea breath test for the diagnosis of Helicobacter pylori infection in children: a systematic review and meta-analysis. Helicobacter 2011 Aug;16(4): Leal YA, et al. Utility of stool sample-based tests for the diagnosis of helicobacter pylori infection in children. J Pediatr Gastroenterol Nutr 2011 Jun;52(6): *Li Y, et al. Clinical value of helicobacter pylori stool antigen test, ImmunoCard STAT HpSA, for detecting H pylori infection. World J Gastroenterol 2004 Mar;10(6): National Institute for Clinical Excellence (NICE). Dyspepsia; managing dyspepsia in adults in primary care.cg [ accessed 4/15/14. Pathak CM, et al. 14C-urea breath test is safe for pediatric patients. Nucl Med Commun 2010 Sep;31(9): *Perri F, et al. Helicobacter pylori antigen stool test and 13C-urea breath test in patients after eradication treatments. Am J Gastroenterol 2002 Nov;97(11): Queiroz DM, et al. Helicobacter pylori infection in infants and toddlers on South America: concordance between [13C] urea breath test and monoclonal H. pylori stool antigen test. J Clin Microbiol 2013 Nov;51(11): *Schilling D, et al. Diagnostic accuracy of 13 C-urea breath test in the diagnosis of Helicobacter pylori infection in patients with partial gastric resection due to peptic ulcer disease: a prospective multicenter study. Digestion 2001;63(1):8-13. Sudraba A, et al. performance of routine helicobacter pylori tests in patients with atrophic gastritis. J Gastrointest Liver Dis 2011 Dec;20(4): Tain XY, et al. Diagnostic performance of urea breath test, rapid urea test, and histology for helicobacter pylori infection in patients with partial gastrectomy: a meta-analysis. J Clin Gastroenterol 2012 Apr;46(4): *Talley NJ, et al. Guidelines for the management of dyspepsia. Am J Gastroenterol 2005 Oct;100(10): *Talley NJ, et al. American Gastroenterological Association. Technical review on the evaluation of dyspepsia. Gastroenterology 2005 Nov;129(5): U.S. Food and Drug Administration. FDA news release. FDA approves first helicobacter pylori test for children Feb 24 [ accessed 4/5/18. *Vakil N, et al. How to test for Helicobacter pylori in Cleve Clin J Med 2005 May;72 Suppl 2:S8-13. Valle PC, et al. Managing dyspepsia in the young adult patient: effects of different tests for helicobacter pylori in a testan-scope approach. Scan J Gastroenterol 2013 Aug;48(8): Velayos B, et al. Accuracy of urea breath test performed immediately after emergency endoscopy in peptic ulcer bleeding. Dig Dis Sci 2012 Jul;57(7):
6 PAGE: 6 OF: 6 Wardi J, et al. A rapid continuous-real-time 13C-urea test for the detection of helicobacter pylori in patients after partial gastrectomy. J Clin Gastroenterol 2012 Apr;46(4): *Weingart V, et al. Sensitivity of a novel stool antigen test for the detection of helicobacter pylori in adult out-patients before and after eradication therapy. J Clin Microbiol 2004 Mar;42(3): * key article KEY WORDS: Helicobacter pylori, HpSA, H pylori, Urea breath test CMS COVERAGE FOR MEDICARE PRODUCT MEMBERS Based upon our review, Helicobacter pylori testing is not addressed in National or regional CMS coverage determinations or policies.
Helicobacter Pylori Testing HELICOBACTER PYLORI TESTING HS-131. Policy Number: HS-131. Original Effective Date: 9/17/2009
 Easy Choice Health Plan, Inc. Harmony Health Plan of Illinois, Inc. Missouri Care, Inc. Ohana Health Plan, a plan offered by WellCare Health Insurance of Arizona, Inc. WellCare Health Insurance of Illinois,
Easy Choice Health Plan, Inc. Harmony Health Plan of Illinois, Inc. Missouri Care, Inc. Ohana Health Plan, a plan offered by WellCare Health Insurance of Arizona, Inc. WellCare Health Insurance of Illinois,
Clinical Policy: Helicobacter Pylori Serology Testing Reference Number: CP.MP.153
 Clinical Policy: Reference Number: CP.MP.153 Effective Date: 12/17 Last Review Date: 12/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
Clinical Policy: Reference Number: CP.MP.153 Effective Date: 12/17 Last Review Date: 12/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory and
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: helicobacter_pylori_testing 01/01/2019 N/A 01/01/2020 01/01/2019 Policy Effective April 1, 2019 Description
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: helicobacter_pylori_testing 01/01/2019 N/A 01/01/2020 01/01/2019 Policy Effective April 1, 2019 Description
Clinical Policy Title: Noninvasive testing for H. pylori
 Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.05 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: August 17, 2017 Next
Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.05 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: August 17, 2017 Next
Clinical Policy Title: Noninvasive testing for H. pylori
 Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.04 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: August 17, 2016 Next
Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.04 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: August 17, 2016 Next
Clinical Policy Title: Noninvasive testing for H. pylori
 Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.04 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: August 17, 2016 Next
Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.04 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: August 17, 2016 Next
Clinical Policy Title: Noninvasive testing for H. pylori
 Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.05 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: July 3, 2018 Next
Clinical Policy Title: Noninvasive testing for H. pylori Clinical Policy Number: 08.01.05 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: July 3, 2018 Next
Clinical Policy Title: Breath Testing for H. Pylori
 Clinical Policy Title: Breath Testing for H. Pylori Clinical Policy Number: 08.01.04 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: August 19, 2015 Next Review
Clinical Policy Title: Breath Testing for H. Pylori Clinical Policy Number: 08.01.04 Effective Date: January 1, 2016 Initial Review Date: August 19, 2015 Most Recent Review Date: August 19, 2015 Next Review
Urea Breath Test for Diagnosis of Helicobactor pylori. Original Policy Date 12:2013
 MP 2.04.04 Urea Breath Test for Diagnosis of Helicobactor pylori Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date 12:2013 Return to Medical Policy Index
MP 2.04.04 Urea Breath Test for Diagnosis of Helicobactor pylori Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date 12:2013 Return to Medical Policy Index
MEDICAL POLICY SUBJECT: MAGNETIC ESOPHAGEAL RING/ MAGNETIC SPHINCTER AUGMENTATION FOR THE TREATMENT OF GASTROESOPHAGEAL REFLUX DISEASE (GERD)
 MEDICAL POLICY SUBJECT: MAGNETIC ESOPHAGEAL RING/ MAGNETIC SPHINCTER PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial
MEDICAL POLICY SUBJECT: MAGNETIC ESOPHAGEAL RING/ MAGNETIC SPHINCTER PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial
Peptic Ulcer Disease Update
 Peptic Ulcer Disease Update Col Pat Storms RAM 2005 Disclosure Information 84th Annual AsMA Scientific Meeting Col Patrick Storms I have no financial relationships to disclose. I will discuss the following
Peptic Ulcer Disease Update Col Pat Storms RAM 2005 Disclosure Information 84th Annual AsMA Scientific Meeting Col Patrick Storms I have no financial relationships to disclose. I will discuss the following
Disclosures. Co-founder and Chief Science Officer, TechLab
 H. pylori testing Disclosures Co-founder and Chief Science Officer, TechLab Learning Objectives Evaluate the appropriate testing methodology by balancing performance, economics, and workflow. Discuss the
H. pylori testing Disclosures Co-founder and Chief Science Officer, TechLab Learning Objectives Evaluate the appropriate testing methodology by balancing performance, economics, and workflow. Discuss the
American College of Gastroenterology Guideline on the Management of Helicobacter pylori Infection
 American Journal of Gastroenterology ISSN 0002-9270 C 2007 by Am. Coll. of Gastroenterology doi: 10.1111/j.1572-0241.2007.01393.x Published by Blackwell Publishing American College of Gastroenterology
American Journal of Gastroenterology ISSN 0002-9270 C 2007 by Am. Coll. of Gastroenterology doi: 10.1111/j.1572-0241.2007.01393.x Published by Blackwell Publishing American College of Gastroenterology
MEDICAL POLICY SUBJECT: TRANSRECTAL ULTRASOUND (TRUS)
 MEDICAL POLICY SUBJECT: TRANSRECTAL ULTRASOUND 06/16/05, 05/18/06, 03/15/07, 02/21/08 PAGE: 1 OF: 5 If the member's subscriber contract excludes coverage for a specific service it is not covered under
MEDICAL POLICY SUBJECT: TRANSRECTAL ULTRASOUND 06/16/05, 05/18/06, 03/15/07, 02/21/08 PAGE: 1 OF: 5 If the member's subscriber contract excludes coverage for a specific service it is not covered under
Treatment of Helicobacter pylori Infection
 Treatment of Helicobacter pylori Infection Epidemiology of H. pylori infection (North America) Which are the high risk groups? Epidemiology of H. pylori infection (North America) Which are the high risk
Treatment of Helicobacter pylori Infection Epidemiology of H. pylori infection (North America) Which are the high risk groups? Epidemiology of H. pylori infection (North America) Which are the high risk
Helicobacter 2008;13:1-6. Am J Gastroent 2007;102: Am J of Med 2004;117:31-35.
 An Update on Helicobacter pylori and Its Treatment Trenika Mitchell, PharmD, BCPS Clinical Assistant Professor University of Kentucky College of Pharmacy October 18, 2008 Objectives Review the epidemiology
An Update on Helicobacter pylori and Its Treatment Trenika Mitchell, PharmD, BCPS Clinical Assistant Professor University of Kentucky College of Pharmacy October 18, 2008 Objectives Review the epidemiology
MEDICAL POLICY SUBJECT: URINARY TUMOR MARKERS FOR BLADDER CANCER. POLICY NUMBER: CATEGORY: Technology Assessment
 MEDICAL POLICY SUBJECT: URINARY TUMOR MARKERS FOR PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
MEDICAL POLICY SUBJECT: URINARY TUMOR MARKERS FOR PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
ACG Clinical Guideline: Treatment of Helicobacter pylori Infection
 ACG Clinical Guideline: Treatment of Helicobacter pylori Infection William D. Chey, MD, FACG 1, Grigorios I. Leontiadis, MD, PhD 2, Colin W. Howden, MD, FACG 3 and Steven F. Moss, MD, FACG 4 1 Division
ACG Clinical Guideline: Treatment of Helicobacter pylori Infection William D. Chey, MD, FACG 1, Grigorios I. Leontiadis, MD, PhD 2, Colin W. Howden, MD, FACG 3 and Steven F. Moss, MD, FACG 4 1 Division
Helicobacter and gastritis
 1 Helicobacter and gastritis Dr. Hala Al Daghistani Helicobacter pylori is a spiral-shaped gram-negative rod. H. pylori is associated with antral gastritis, duodenal (peptic) ulcer disease, gastric ulcers,
1 Helicobacter and gastritis Dr. Hala Al Daghistani Helicobacter pylori is a spiral-shaped gram-negative rod. H. pylori is associated with antral gastritis, duodenal (peptic) ulcer disease, gastric ulcers,
MEDICAL POLICY SUBJECT: MAMMOGRAPHY: COMPUTER- AIDED DETECTION (CAD) POLICY NUMBER: CATEGORY: Technology Assessment
 MEDICAL POLICY SUBJECT: MAMMOGRAPHY: COMPUTER- PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
MEDICAL POLICY SUBJECT: MAMMOGRAPHY: COMPUTER- PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
What is the status of Sequential Therapy Versus Standard Triple- Drug Therapy in peptic ulcer disease in eradicating H pylori?
 What is the status of Sequential Therapy Versus Standard Triple- Drug Therapy in peptic ulcer disease in eradicating H pylori? Sequential Therapy Versus Standard Triple- Drug Therapy for Helicobacter pylori
What is the status of Sequential Therapy Versus Standard Triple- Drug Therapy in peptic ulcer disease in eradicating H pylori? Sequential Therapy Versus Standard Triple- Drug Therapy for Helicobacter pylori
PEPTIC ULCER DISEASE JOHN R SALTZMAN, MD. Director of Endoscopy Brigham and Women s Hospital Professor of Medicine Harvard Medical School
 PEPTIC ULCER DISEASE JOHN R SALTZMAN, MD Director of Endoscopy Brigham and Women s Hospital Professor of Medicine Harvard Medical School No disclosures Disclosures Overview Causes of peptic ulcer disease
PEPTIC ULCER DISEASE JOHN R SALTZMAN, MD Director of Endoscopy Brigham and Women s Hospital Professor of Medicine Harvard Medical School No disclosures Disclosures Overview Causes of peptic ulcer disease
Evaluation of a new rapid immunoassay for the detection of Helicobacter pylori in faeces: a prospective pilot study
 Aliment Pharmacol Ther 2005; 21: 485 489. doi: 10.1111/j.1365-2036.2005.02355.x Evaluation of a new rapid immunoassay for the detection of Helicobacter pylori in faeces: a prospective pilot study L. TREVISANI*,
Aliment Pharmacol Ther 2005; 21: 485 489. doi: 10.1111/j.1365-2036.2005.02355.x Evaluation of a new rapid immunoassay for the detection of Helicobacter pylori in faeces: a prospective pilot study L. TREVISANI*,
Acid-Peptic Diseases of the Stomach and Duodenum Including Helicobacter pylori and NSAIDs Prof. Sheila Crowe
 Acid-Peptic Diseases of the Stomach and Duodenum Including Helicobacter pylori and NSAIDs 1 Division of Gastroenterology UC San Diego School of Medicine Clinical presentations of Helicobacter pylori infection
Acid-Peptic Diseases of the Stomach and Duodenum Including Helicobacter pylori and NSAIDs 1 Division of Gastroenterology UC San Diego School of Medicine Clinical presentations of Helicobacter pylori infection
Management of dyspepsia and of Helicobacter pylori infection
 Management of dyspepsia and of Helicobacter pylori infection The University of Nottingham John Atherton Wolfson Digestive Diseases Centre University of Nottingham, UK Community management of dyspepsia
Management of dyspepsia and of Helicobacter pylori infection The University of Nottingham John Atherton Wolfson Digestive Diseases Centre University of Nottingham, UK Community management of dyspepsia
The significance of Helicobacter pylori in the approach of dyspepsia in primary care Arents, Nicolaas Lodevikus Augustinus
 University of Groningen The significance of Helicobacter pylori in the approach of dyspepsia in primary care Arents, Nicolaas Lodevikus Augustinus IMPORTANT NOTE: You are advised to consult the publisher's
University of Groningen The significance of Helicobacter pylori in the approach of dyspepsia in primary care Arents, Nicolaas Lodevikus Augustinus IMPORTANT NOTE: You are advised to consult the publisher's
DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) MANAGEMENT OF DYSPEPSIA
 DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) MANAGEMENT OF DYSPEPSIA o Patients of any age with ALARM signs should be referred through the 2-week referral system o Routine endoscopic investigation
DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) MANAGEMENT OF DYSPEPSIA o Patients of any age with ALARM signs should be referred through the 2-week referral system o Routine endoscopic investigation
instrument. When 13C-UBT positive value is greater than or equal to / - 0.4, the the subject can be 1. Data and methods details are as follows:
 Application of 13 C-urea breath test in screening helicobacter pylori infection during health examination in Chengdu, Sichuan YANG Yan-hua. LIU Yu-ping. CHENG You-fu, SHUAI Ping. LU Qiao. ZHENG Xiao-xia,
Application of 13 C-urea breath test in screening helicobacter pylori infection during health examination in Chengdu, Sichuan YANG Yan-hua. LIU Yu-ping. CHENG You-fu, SHUAI Ping. LU Qiao. ZHENG Xiao-xia,
EDUCATION PRACTICE. Persistent Helicobacter pylori Infection After a Course of Antimicrobial Therapy What s Next? Clinical Scenario.
 CLINICAL GASTROENTEROLOGY AND HEPATOLOGY 2008;6:1086 1090 EDUCATION PRACTICE Persistent Helicobacter pylori Infection After a Course of Antimicrobial Therapy What s Next? RICHARD J. SAAD* and WILLIAM D.
CLINICAL GASTROENTEROLOGY AND HEPATOLOGY 2008;6:1086 1090 EDUCATION PRACTICE Persistent Helicobacter pylori Infection After a Course of Antimicrobial Therapy What s Next? RICHARD J. SAAD* and WILLIAM D.
Helicobacter pylori: Diagnosis, treatment and risks of untreated infection
 Helicobacter pylori: Diagnosis, treatment and risks of untreated infection Klaus Mönkemüller Department of Gastroenterology, Hepatology und Infectius Diseases Otto-von-Guericke University, Magdeburg bb
Helicobacter pylori: Diagnosis, treatment and risks of untreated infection Klaus Mönkemüller Department of Gastroenterology, Hepatology und Infectius Diseases Otto-von-Guericke University, Magdeburg bb
National Digestive Diseases Information Clearinghouse
 Gastritis National Digestive Diseases Information Clearinghouse U.S. Department of Health and Human Services NATIONAL INSTITUTES OF HEALTH What is gastritis? Gastritis is a condition in which the stomach
Gastritis National Digestive Diseases Information Clearinghouse U.S. Department of Health and Human Services NATIONAL INSTITUTES OF HEALTH What is gastritis? Gastritis is a condition in which the stomach
Type of intervention Diagnosis. Economic study type Cost-effectiveness analysis.
 Noninvasive tests as a substitute for histology in the diagnosis of Helicobacter pylori infection Hahn M, Fennerty M B, Corless C L, Magaret N, Lieberman D A, Faigel D O Record Status This is a critical
Noninvasive tests as a substitute for histology in the diagnosis of Helicobacter pylori infection Hahn M, Fennerty M B, Corless C L, Magaret N, Lieberman D A, Faigel D O Record Status This is a critical
Fecoprevalence and determinants of Helicobacter pylor infection among asymptomatic children in Myanmar
 International Journal of Gastroenterology, Hepatology, Transplant & Nutrition Original Article Fecoprevalence and determinants of Helicobacter pylor infection among asymptomatic children in Myanmar Hnin
International Journal of Gastroenterology, Hepatology, Transplant & Nutrition Original Article Fecoprevalence and determinants of Helicobacter pylor infection among asymptomatic children in Myanmar Hnin
Helicobacter pylori 幽門螺旋桿菌 馬偕紀念醫院新竹分院一般內科, 肝膽腸胃科陳重助醫師
 Helicobacter pylori 幽門螺旋桿菌 馬偕紀念醫院新竹分院一般內科, 肝膽腸胃科陳重助醫師 Hp : Helicobacter pylori Part 1. Pathophysiology and immune response Pathogenesis of Hp infection Part 2. Clinical manifestation Part 3. Dx tests for
Helicobacter pylori 幽門螺旋桿菌 馬偕紀念醫院新竹分院一般內科, 肝膽腸胃科陳重助醫師 Hp : Helicobacter pylori Part 1. Pathophysiology and immune response Pathogenesis of Hp infection Part 2. Clinical manifestation Part 3. Dx tests for
June By: Reza Gholami
 ACG/CAG guideline on Management of Dyspepsia June 2017 By: Reza Gholami DEFINITION OF DYSPEPSIA AND SCOPE OF THE GUIDELINE Dyspepsia was originally defined as any symptoms referable to the upper gastrointestinal
ACG/CAG guideline on Management of Dyspepsia June 2017 By: Reza Gholami DEFINITION OF DYSPEPSIA AND SCOPE OF THE GUIDELINE Dyspepsia was originally defined as any symptoms referable to the upper gastrointestinal
CHAPTER 18. PEPTIC ULCER DISEASE, SELF-ASSESSMENT QUESTIONS. 1. Which of the following is not a common cause of peptic ulcer disease (PUD)?
 CHAPTER 18. PEPTIC ULCER DISEASE, SELF-ASSESSMENT QUESTIONS 1. Which of the following is not a common cause of peptic ulcer disease (PUD)? A. Chronic alcohol ingestion B. Nonsteroidal antiinflammatory
CHAPTER 18. PEPTIC ULCER DISEASE, SELF-ASSESSMENT QUESTIONS 1. Which of the following is not a common cause of peptic ulcer disease (PUD)? A. Chronic alcohol ingestion B. Nonsteroidal antiinflammatory
Management of Dyspepsia
 MPharm Programme Management of Dyspepsia Slide 1 of 28 Learning Objectives Understand the principles and wider implications underpinning evidence based therapeutics in the key clinical specialities Objectively
MPharm Programme Management of Dyspepsia Slide 1 of 28 Learning Objectives Understand the principles and wider implications underpinning evidence based therapeutics in the key clinical specialities Objectively
The Nobel Prize in Physiology or Medicine for 2005
 The Nobel Prize in Physiology or Medicine for 2005 jointly to Barry J. Marshall and J. Robin Warren for their discovery of "the bacterium Helicobacter pylori and its role in gastritis and peptic ulcer
The Nobel Prize in Physiology or Medicine for 2005 jointly to Barry J. Marshall and J. Robin Warren for their discovery of "the bacterium Helicobacter pylori and its role in gastritis and peptic ulcer
Helicobacter pylori:an Emerging Pathogen
 Bacteriology at UW-Madison Bacteriology 330 Home Page Helicobacter pylori:an Emerging Pathogen by Karrie Holston, Department of Bacteriology University of Wisconsin-Madison Description of Helicobacter
Bacteriology at UW-Madison Bacteriology 330 Home Page Helicobacter pylori:an Emerging Pathogen by Karrie Holston, Department of Bacteriology University of Wisconsin-Madison Description of Helicobacter
Non-Ulcer Dyspepsia: what is it? What can we do with these patients? Overview. Dyspepsia Definition. Functional Dyspepsia. Dyspepsia the Basics
 Non-Ulcer : what is it? What can we do with these patients? Temporal Changes and Geographic Variations in Developing Peptic Ulcer Disease Gastric Cancer 1900 Eamonn M M Quigley MD FACG Alimentary Pharmabiotic
Non-Ulcer : what is it? What can we do with these patients? Temporal Changes and Geographic Variations in Developing Peptic Ulcer Disease Gastric Cancer 1900 Eamonn M M Quigley MD FACG Alimentary Pharmabiotic
Intragastric Acidification Reduces the Occurrence of False-Negative Urea Breath Test Results in Patients Taking a Proton Pump Inhibitor
 THE AMERICAN JOURNAL OF GASTROENTEROLOGY Vol. 96, No. 4, 2001 2001 by Am. Coll. of Gastroenterology ISSN 0002-9270/01/$20.00 Published by Elsevier Science Inc. PII S0002-9270(01)02250-X Intragastric Acidification
THE AMERICAN JOURNAL OF GASTROENTEROLOGY Vol. 96, No. 4, 2001 2001 by Am. Coll. of Gastroenterology ISSN 0002-9270/01/$20.00 Published by Elsevier Science Inc. PII S0002-9270(01)02250-X Intragastric Acidification
MEDICAL POLICY SUBJECT: GASTRIC ELECTRICAL STIMULATION
 MEDICAL POLICY SUBJECT: GASTRIC ELECTRICAL PAGE: 1 OF: 6 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including an
MEDICAL POLICY SUBJECT: GASTRIC ELECTRICAL PAGE: 1 OF: 6 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including an
Proton Pump Inhibitors Drug Class Prior Authorization Protocol
 Proton Pump Inhibitors Drug Class Prior Authorization Protocol Line of Business: Medi-Cal P&T Approval Date: November 15, 2017 Effective Date: January 1, 2018 This policy has been developed through review
Proton Pump Inhibitors Drug Class Prior Authorization Protocol Line of Business: Medi-Cal P&T Approval Date: November 15, 2017 Effective Date: January 1, 2018 This policy has been developed through review
MEDICAL POLICY SUBJECT: COGNITIVE REHABILITATION. POLICY NUMBER: CATEGORY: Therapy/Rehabilitation
 MEDICAL POLICY SUBJECT: COGNITIVE REHABILITATION PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product (including
MEDICAL POLICY SUBJECT: COGNITIVE REHABILITATION PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product (including
H. pylori Antigen ELISA Kit
 H. pylori Antigen ELISA Kit Catalog Number KA3142 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
H. pylori Antigen ELISA Kit Catalog Number KA3142 96 assays Version: 04 Intended for research use only www.abnova.com Table of Contents Introduction... 3 Intended Use... 3 Background... 3 Principle of
TRANSPARENCY COMMITTEE OPINION. 13 December 2006
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 December 2006 HELIKIT 75 mg, powder for oral solution CIP : 343 132-1 Applicant : MAYOLY SPINDLER 13 Curea anhydrous
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 13 December 2006 HELIKIT 75 mg, powder for oral solution CIP : 343 132-1 Applicant : MAYOLY SPINDLER 13 Curea anhydrous
Treatment and Screening of H. pylori Infection in Alaskan Populations
 Treatment and Screening of H. pylori Infection in Alaskan Populations Matthew F. Deraedt, Pharm.D. Lieutenant United States Public Health Service Alaska Native Medical Center Pharmacy Practice PGY-1 Resident
Treatment and Screening of H. pylori Infection in Alaskan Populations Matthew F. Deraedt, Pharm.D. Lieutenant United States Public Health Service Alaska Native Medical Center Pharmacy Practice PGY-1 Resident
MANAGEMENT OF DYSPEPSIA AND GASTRO-OESOPHAGEAL REFLUX DISEASE (GORD)
 DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) MANAGEMENT OF DYSPEPSIA AND GASTRO-OESOPHAGEAL REFLUX DISEASE (GORD) Routine endoscopic investigation of patients of any age, presenting with dyspepsia
DERBYSHIRE JOINT AREA PRESCRIBING COMMITTEE (JAPC) MANAGEMENT OF DYSPEPSIA AND GASTRO-OESOPHAGEAL REFLUX DISEASE (GORD) Routine endoscopic investigation of patients of any age, presenting with dyspepsia
Comparison of the Accuracy of Two Commercial Rapid Urase Tests, CLOtest and Pronto Dry, in Detecting Helicobacter pylori Infection ABSTRACT
 Original Article Rojborwonwitaya J, Vijitjunyakul N THAI J GASTROENTEROL 2005 Vol. 6 No. 2 May - Aug. 2005 55 Comparison of the Accuracy of Two Commercial Rapid Urase Tests, CLOtest and Pronto Dry, in
Original Article Rojborwonwitaya J, Vijitjunyakul N THAI J GASTROENTEROL 2005 Vol. 6 No. 2 May - Aug. 2005 55 Comparison of the Accuracy of Two Commercial Rapid Urase Tests, CLOtest and Pronto Dry, in
University Medical Center at Brackenridge. Gastroenterology Clinic Worksheet
 Gastroenterology Clinic Worksheet 1. GI Bleeding (occult or symptomatic) a. CBC b. Iron, Ferritin b. Medication history 2. Iron Deficiency Anemia and no evident source (if no iron deficiency consider hematological
Gastroenterology Clinic Worksheet 1. GI Bleeding (occult or symptomatic) a. CBC b. Iron, Ferritin b. Medication history 2. Iron Deficiency Anemia and no evident source (if no iron deficiency consider hematological
6/25/ % 20% 50% 19% Functional Dyspepsia Peptic Ulcer GERD Cancer Other
 Peptic Ulcer Disease and Dyspepsia John M. Inadomi, MD Professor of Medicine UCSF Chief, Clinical Gastroenterology San Francisco General Hospital Case History 49 y/o woman complains of several months of
Peptic Ulcer Disease and Dyspepsia John M. Inadomi, MD Professor of Medicine UCSF Chief, Clinical Gastroenterology San Francisco General Hospital Case History 49 y/o woman complains of several months of
One-third of adults experience pain or discomfort in
 GASTROENTEROLOGY 2002;122:1270 1285 Dyspepsia Management in Primary Care: A Decision Analysis of Competing Strategies BRENNAN M. R. SPIEGEL,* NIMISH B. VAKIL, and JOSHUA J. OFMAN*, *Department of Medicine
GASTROENTEROLOGY 2002;122:1270 1285 Dyspepsia Management in Primary Care: A Decision Analysis of Competing Strategies BRENNAN M. R. SPIEGEL,* NIMISH B. VAKIL, and JOSHUA J. OFMAN*, *Department of Medicine
Gilles Jequier. Commercial Director Organobalance, a Novozymes Company
 "Latest clinical Evidences showing that a proprietary Lactobacillus reuteri Strain can reduce the Symptoms associated with a Helicobacter pylori Infection" Gilles Jequier Commercial Director Organobalance,
"Latest clinical Evidences showing that a proprietary Lactobacillus reuteri Strain can reduce the Symptoms associated with a Helicobacter pylori Infection" Gilles Jequier Commercial Director Organobalance,
H.pylori IgA Cat #
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
GI update. Common conditions and concerns my patients frequently asked about
 GI update Common conditions and concerns my patients frequently asked about Specific conditions I ll try to cover today 1. Colon polyps, colorectal cancer and colonoscopy 2. Crohn s disease 3. Peptic ulcer
GI update Common conditions and concerns my patients frequently asked about Specific conditions I ll try to cover today 1. Colon polyps, colorectal cancer and colonoscopy 2. Crohn s disease 3. Peptic ulcer
Helicobacter Pylori Test-and-Treat Strategy for Management of Dyspepsia: A Comprehensive Review
 Citation: (2013) 4, e32; doi:10.1038/ctg.2013.3 & 2013 the American College of Gastroenterology All rights reserved 2155-384X/13 www.nature.com/ctg CLINICAL/NARRATIVE REVIEW Helicobacter Pylori Test-and-Treat
Citation: (2013) 4, e32; doi:10.1038/ctg.2013.3 & 2013 the American College of Gastroenterology All rights reserved 2155-384X/13 www.nature.com/ctg CLINICAL/NARRATIVE REVIEW Helicobacter Pylori Test-and-Treat
Investigating dyspepsia Rocco Maurizio Zagari, Lorenzo Fuccio, Franco Bazzoli
 DATE: 11/5/2008-10:27:27 ID:(BMJ)zagr584193 /(Jouve)bmj-001985 DOI: 10.1136/bmj.a1400 Topic(s): Type: InSection:review-article For the full versions of these articles see bmj.com CLINICAL REVIEW Investigating
DATE: 11/5/2008-10:27:27 ID:(BMJ)zagr584193 /(Jouve)bmj-001985 DOI: 10.1136/bmj.a1400 Topic(s): Type: InSection:review-article For the full versions of these articles see bmj.com CLINICAL REVIEW Investigating
H. pylori IgM CLIA kit
 H. pylori IgM CLIA kit Cat. No.:DEEL0251 Pkg.Size:96 tests Intended use Helicobacter pylori IgM Chemiluminescence ELISA is intended for use in evaluating the serologic status to H. pylori infection in
H. pylori IgM CLIA kit Cat. No.:DEEL0251 Pkg.Size:96 tests Intended use Helicobacter pylori IgM Chemiluminescence ELISA is intended for use in evaluating the serologic status to H. pylori infection in
H.Pylori IgG
 DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
DIAGNOSTIC AUTOMATION, INC. 21250 Califa Street, Suite 102 and116, Woodland Hills, CA 91367 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com
H.Pylori IgG Cat # 1503Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
COMPARISON OF ONCE-A-DAY VERSUS TWICE-A-DAY CLARITHROMYCIN IN TRIPLE THERAPY FOR HELICOBACTER PYLORI ERADICATION
 Phil J Gastroenterol 2006; 2: 25-29 COMPARISON OF ONCE-A-DAY VERSUS TWICE-A-DAY CLARITHROMYCIN IN TRIPLE THERAPY FOR HELICOBACTER PYLORI ERADICATION Marianne P Collado, Ma Fatima P Calida, Peter P Sy,
Phil J Gastroenterol 2006; 2: 25-29 COMPARISON OF ONCE-A-DAY VERSUS TWICE-A-DAY CLARITHROMYCIN IN TRIPLE THERAPY FOR HELICOBACTER PYLORI ERADICATION Marianne P Collado, Ma Fatima P Calida, Peter P Sy,
Summary HTA. Efficacy and cost-effectiveness of the
 Summary HTA HTA-Report Summary Efficacy and cost-effectiveness of the 13 C-urea breath test as the primary diagnostic investigation for the detection of Helicobacter pylori infection compared to invasive
Summary HTA HTA-Report Summary Efficacy and cost-effectiveness of the 13 C-urea breath test as the primary diagnostic investigation for the detection of Helicobacter pylori infection compared to invasive
Medical Policy. MP Ingestible ph and Pressure Capsule
 Medical Policy BCBSA Ref. Policy: 2.01.81 Last Review: 11/15/2018 Effective Date: 11/15/2018 Section: Medicine Related Policies 2.01.20 Esophageal ph Monitoring 6.01.33 Wireless Capsule Endoscopy as a
Medical Policy BCBSA Ref. Policy: 2.01.81 Last Review: 11/15/2018 Effective Date: 11/15/2018 Section: Medicine Related Policies 2.01.20 Esophageal ph Monitoring 6.01.33 Wireless Capsule Endoscopy as a
See external label 2 C-8 C Σ=96 tests Cat # 1505Z. MICROWELL ELISA H.Pylori IgA Cat # 1505Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Epidemiology of Peptic Ulcer Disease
 Epidemiology of Peptic Ulcer Disease Introduction Peptic Ulcer Disease (PUD) is disruption of the mucosal integrity of the stomach and/or duodenum leading to a local defect or excavation due to active
Epidemiology of Peptic Ulcer Disease Introduction Peptic Ulcer Disease (PUD) is disruption of the mucosal integrity of the stomach and/or duodenum leading to a local defect or excavation due to active
Efficacy of standard triple therapy versus bismuth-based quadruple therapy for eradication of Helicobacter Pylori infection
 Efficacy of standard triple therapy versus bismuth-based quadruple therapy for eradication of Helicobacter Pylori infection Ramin Talaie Modarress Hospital, Shahid Beheshti University of Medical Sciences,
Efficacy of standard triple therapy versus bismuth-based quadruple therapy for eradication of Helicobacter Pylori infection Ramin Talaie Modarress Hospital, Shahid Beheshti University of Medical Sciences,
Outline. Definition (s) Epidemiology Pathophysiology Management With an emphasis on recent developments
 Chronic Dyspepsia Eamonn M M Quigley MD FRCP FACP MACG FRCPI Lynda K and David M Underwood Center for Digestive Disorders Houston Methodist Hospital Houston, Texas Outline Definition (s) Epidemiology Pathophysiology
Chronic Dyspepsia Eamonn M M Quigley MD FRCP FACP MACG FRCPI Lynda K and David M Underwood Center for Digestive Disorders Houston Methodist Hospital Houston, Texas Outline Definition (s) Epidemiology Pathophysiology
SUBJECT: LIMB PNEUMATIC COMPRESSION EFFECTIVE DATE: 06/27/13 DEVICES FOR VENOUS REVISED DATE: 06/26/14 THROMBOEMBOLISM PROPHYLAXIS
 MEDICAL POLICY SUBJECT: LIMB PNEUMATIC COMPRESSION PAGE: 1 OF: 5 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases, medical
MEDICAL POLICY SUBJECT: LIMB PNEUMATIC COMPRESSION PAGE: 1 OF: 5 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases, medical
Magaji et al., Afr. J. Infect. Dis. 2(2): THE ROLE OF AN AGGRESSIVE FACTOR IN PEPTIC ULCER DISEASE (PUD)
 Magaji et al., Afr. J. Infect. Dis. 2(2): 80-84 80 Review Paper ISSN: 2006-0165 2008 THE ROLE OF AN AGGRESSIVE FACTOR IN PEPTIC ULCER DISEASE (PUD) Magaji, R.A. 1,b *, Tanko, Y. 2 and Magaji, G.M. 3 Afr.
Magaji et al., Afr. J. Infect. Dis. 2(2): 80-84 80 Review Paper ISSN: 2006-0165 2008 THE ROLE OF AN AGGRESSIVE FACTOR IN PEPTIC ULCER DISEASE (PUD) Magaji, R.A. 1,b *, Tanko, Y. 2 and Magaji, G.M. 3 Afr.
Prevpac Pylera Omeclamox-Pak
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.21 Subject: -Pak Page: 1 of 5 Last Review Date: September 20, 2018 -Pak Description (lansoprazole,
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.50.21 Subject: -Pak Page: 1 of 5 Last Review Date: September 20, 2018 -Pak Description (lansoprazole,
MEDICAL POLICY SUBJECT: TRANSMYOCARDIAL REVASCULARIZATION
 MEDICAL POLICY SUBJECT: TRANSMYOCARDIAL 7/21/05, 05/18/06, 03/15/07, 02/21/08,, PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply.
MEDICAL POLICY SUBJECT: TRANSMYOCARDIAL 7/21/05, 05/18/06, 03/15/07, 02/21/08,, PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply.
594 Lewin, Weinstein, and Riddell s Gastrointestinal Pathology and Its Clinical Implications
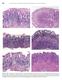 594 Lewin, Weinstein, and Riddell s Gastrointestinal Pathology and Its Clinical Implications Figure 13-20. Stages in the natural history of H. pylori. Biopsies from the antrum are on the left and the oxyntic
594 Lewin, Weinstein, and Riddell s Gastrointestinal Pathology and Its Clinical Implications Figure 13-20. Stages in the natural history of H. pylori. Biopsies from the antrum are on the left and the oxyntic
H. pylori IgM. Cat # H. pylori IgM ELISA. ELISA: Enzyme Linked Immunosorbent Assay. ELISA - Indirect; Antigen Coated Plate
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com H. pylori
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com H. pylori
THE PREVALENCE OF HELICBACTER PYLORI AMONG PATIENTS COMPLAINING FROM ABDOMINAL PAIN
 THE PREVALENCE OF HELICBACTER PYLORI AMONG PATIENTS COMPLAINING FROM ABDOMINAL PAIN Ahed J. Al-Khatib Jordan University of Science and Technology, Jordan Ahmed Saber Abu-zaiton Al-albayt University Abstract
THE PREVALENCE OF HELICBACTER PYLORI AMONG PATIENTS COMPLAINING FROM ABDOMINAL PAIN Ahed J. Al-Khatib Jordan University of Science and Technology, Jordan Ahmed Saber Abu-zaiton Al-albayt University Abstract
Association of Helicobacter pylori infection with Atrophic gastritis in patients with Dyspepsia
 ADVANCES IN BIORESEARCH Adv. Biores., Vol 8 [3] May 2017: 137-141 2017 Society of Education, India Print ISSN 0976-4585; Online ISSN 2277-1573 Journal s URL:http://www.soeagra.com/abr.html CODEN: ABRDC3
ADVANCES IN BIORESEARCH Adv. Biores., Vol 8 [3] May 2017: 137-141 2017 Society of Education, India Print ISSN 0976-4585; Online ISSN 2277-1573 Journal s URL:http://www.soeagra.com/abr.html CODEN: ABRDC3
Setting The setting was primary and secondary care. The economic study was carried out in the UK.
 Helicobacter pylori "test and treat" or endoscopy for managing dyspepsia: an individual patient data meta-analysis Ford A C, Qume M, Moayyedi P, Arents N L, Lassen A T, Logan R F, McColl K E, Myres P,
Helicobacter pylori "test and treat" or endoscopy for managing dyspepsia: an individual patient data meta-analysis Ford A C, Qume M, Moayyedi P, Arents N L, Lassen A T, Logan R F, McColl K E, Myres P,
Updates in Evaluation and Management of Dyspepsia and H. Pylori Infection
 Updates in Evaluation and Management of Dyspepsia and H. Pylori Infection Isabel Lee, MD Associate Professor of Health Sciences UCSF Department of Family and Community Medicine Disclosures None 2 Session
Updates in Evaluation and Management of Dyspepsia and H. Pylori Infection Isabel Lee, MD Associate Professor of Health Sciences UCSF Department of Family and Community Medicine Disclosures None 2 Session
Comparative study of invasive methods for diagnosis of Helicobacter pylori in humans
 ISSN: 2319-7706 Volume 2 Number 7 (2013) pp. 63-68 http://www.ijcmas.com Original Research Article Comparative study of invasive methods for diagnosis of Helicobacter pylori in humans V.Subbukesavaraja
ISSN: 2319-7706 Volume 2 Number 7 (2013) pp. 63-68 http://www.ijcmas.com Original Research Article Comparative study of invasive methods for diagnosis of Helicobacter pylori in humans V.Subbukesavaraja
MEDICAL POLICY SUBJECT: NUTRITIONAL THERAPY
 MEDICAL POLICY PAGE: 1 OF: 6 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases, medical policy criteria are not applied.
MEDICAL POLICY PAGE: 1 OF: 6 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases, medical policy criteria are not applied.
MEDICAL POLICY SUBJECT: GENOTYPING OR PHENOTYPING FOR THIOPURINE METHYLTRANSFERASE (TPMT) FOR PATIENTS TREATED WITH AZATHIOPRINE (6-MP)
 MEDICAL POLICY SUBJECT: GENOTYPING OR PHENOTYPING PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product (including
MEDICAL POLICY SUBJECT: GENOTYPING OR PHENOTYPING PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product (including
KEYWORDS Dyspepsia, Acid Peptic Disease, Helicobacter Pylori, Urease, Giemsa, Peptic Ulcer, Non-Ulcer Dyspepsia.
 INCIDENCE OF HELICOBACTER PYLORI WITH ACID PEPTIC DISEASE AND MALIGNANT CONDITIONS OF UPPER GASTROINTESTINAL TRACT IN A TERTIARY CENTRE - A PROSPECTIVE STUDY Karunamoorthy Rajachidambaram 1, Dinkaran Kaarthesan
INCIDENCE OF HELICOBACTER PYLORI WITH ACID PEPTIC DISEASE AND MALIGNANT CONDITIONS OF UPPER GASTROINTESTINAL TRACT IN A TERTIARY CENTRE - A PROSPECTIVE STUDY Karunamoorthy Rajachidambaram 1, Dinkaran Kaarthesan
SCIENTIFIC DISCUSSION
 SCIENTIFIC DISCUSSION This module reflects the initial scientific discussion and scientific discussion on procedures, which have been finalised before 1 October 2001. For scientific information on procedures
SCIENTIFIC DISCUSSION This module reflects the initial scientific discussion and scientific discussion on procedures, which have been finalised before 1 October 2001. For scientific information on procedures
Correlation Between Endoscopic and Histological Findings in Different Gastroduodenal Lesion and its Association with Helicobacter Pylori
 ORIGINAL ARTICLE Correlation Between Endoscopic and Histological Findings in Different Gastroduodenal Lesion and its Association with Helicobacter Pylori *A. Sultana 1, SM Badruddoza 2, F Rahman 3 1 Dr.
ORIGINAL ARTICLE Correlation Between Endoscopic and Histological Findings in Different Gastroduodenal Lesion and its Association with Helicobacter Pylori *A. Sultana 1, SM Badruddoza 2, F Rahman 3 1 Dr.
H.Pylori IgM Cat # 1504Z
 DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
DIAGNOSTIC AUTOMATION, INC. 23961 Craftsman Road, Suite D/E/F, Calabasas, CA 91302 Tel: (818) 591-3030 Fax: (818) 591-8383 onestep@rapidtest.com technicalsupport@rapidtest.com www.rapidtest.com See external
Peptic ulcer disease. Nomin-Erdene. D SOM-531
 Peptic ulcer disease Nomin-Erdene. D SOM-531 Learning objectives Stomach gross anatomy PUD Epidemiology Pathogenesis Clinical manifestation Diagnosing Treatment Complicated ulcer disease Surgical procedures
Peptic ulcer disease Nomin-Erdene. D SOM-531 Learning objectives Stomach gross anatomy PUD Epidemiology Pathogenesis Clinical manifestation Diagnosing Treatment Complicated ulcer disease Surgical procedures
MEDICAL POLICY SUBJECT: COCHLEAR IMPLANTS AND AUDITORY BRAINSTEM IMPLANTS. POLICY NUMBER: CATEGORY: Technology Assessment
 MEDICAL POLICY PAGE: 1 OF: 5 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases, medical policy criteria are not applied.
MEDICAL POLICY PAGE: 1 OF: 5 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases, medical policy criteria are not applied.
Diagnostic value of rapid Helicobacter pylori stool antigen test.
 Diagnostic value of rapid Helicobacter pylori stool antigen test Mohamed Abdel-moghny Moustafa 1, Sameh Ahmed Abdel-bary 1, Eslam Safwat 1, Mohamed Elnemr 2 1 internal Medicine Department, Faculty of Medicine,
Diagnostic value of rapid Helicobacter pylori stool antigen test Mohamed Abdel-moghny Moustafa 1, Sameh Ahmed Abdel-bary 1, Eslam Safwat 1, Mohamed Elnemr 2 1 internal Medicine Department, Faculty of Medicine,
MEDICAL POLICY SUBJECT: ENHANCED EXTERNAL COUNTERPULSATION
 MEDICAL POLICY PAGE: 1 OF: 4 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases, medical policy criteria are not applied.
MEDICAL POLICY PAGE: 1 OF: 4 If the member's subscriber contract excludes coverage for a specific service it is not covered under that contract. In such cases, medical policy criteria are not applied.
HCPCS Codes (Alphanumeric, CPT AMA) ICD-9-CM Codes Covered by Medicare Program
 HCPCS s (Alphanumeric, CPT AMA) 82272 Blood, occult, by peroxidase activity (e.g., guaiac), qualitative, feces, 1-3 simultaneous determinations, performed for other than colorectal neoplasm screening ICD-9-CM
HCPCS s (Alphanumeric, CPT AMA) 82272 Blood, occult, by peroxidase activity (e.g., guaiac), qualitative, feces, 1-3 simultaneous determinations, performed for other than colorectal neoplasm screening ICD-9-CM
MEDICAL POLICY. SUBJECT: ISOLATED LIMB PERFUSION and INFUSION
 MEDICAL POLICY SUBJECT: ISOLATED LIMB PERFUSION and PAGE: 1 OF: 6 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product (including
MEDICAL POLICY SUBJECT: ISOLATED LIMB PERFUSION and PAGE: 1 OF: 6 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product (including
Research Article Performance of Routine Helicobacter pylori Invasive Tests in Patients with Dyspepsia
 Gastroenterology Research and Practice Volume 2013, Article ID 184806, 5 pages http://dx.doi.org/10.1155/2013/184806 Research Article Performance of Routine Helicobacter pylori Invasive Tests in Patients
Gastroenterology Research and Practice Volume 2013, Article ID 184806, 5 pages http://dx.doi.org/10.1155/2013/184806 Research Article Performance of Routine Helicobacter pylori Invasive Tests in Patients
Low Dose Furazolidone for Eradication of H- pylori Instead of Clarithromycin: A Clinical Trial
 Global Journal of Health Science; Vol. 7, No. 1; 2015 ISSN 1916-9736 E-ISSN 1916-9744 Published by Canadian Center of Science and Education Low Dose Furazolidone for Eradication of H- pylori Instead of
Global Journal of Health Science; Vol. 7, No. 1; 2015 ISSN 1916-9736 E-ISSN 1916-9744 Published by Canadian Center of Science and Education Low Dose Furazolidone for Eradication of H- pylori Instead of
1. Appropriateness of Gastroscopy: Dyspepsia 1
 Special Topic 579 1. Appropriateness of Gastroscopy: Dyspepsia 1 F. Froehlich *, M. Bochud **, J.-J. Gonvers*, R.W. Dubois***, J.-P. Vader **, V. Wietlisbach ***, B. Burnand ** * Policlinique Médicale
Special Topic 579 1. Appropriateness of Gastroscopy: Dyspepsia 1 F. Froehlich *, M. Bochud **, J.-J. Gonvers*, R.W. Dubois***, J.-P. Vader **, V. Wietlisbach ***, B. Burnand ** * Policlinique Médicale
Index. Note: Page numbers of article titles are in boldface type.
 Note: Page numbers of article titles are in boldface type. A Adherence, to bismuth quadruple therapy, 543 546 Adjuvant therapy, probiotics as, 567 569 Age factors, in gastric cancer, 611 612, 616 AID protein,
Note: Page numbers of article titles are in boldface type. A Adherence, to bismuth quadruple therapy, 543 546 Adjuvant therapy, probiotics as, 567 569 Age factors, in gastric cancer, 611 612, 616 AID protein,
Peptic ulcer disease Disorders of the esophagus
 Peptic ulcer disease Disorders of the esophagus Peptic ulcer disease Burning epigastric pain Exacerbated by fasting Improved with meals Ulcer: disruption of mucosal integrity >5 mm in size, with depth
Peptic ulcer disease Disorders of the esophagus Peptic ulcer disease Burning epigastric pain Exacerbated by fasting Improved with meals Ulcer: disruption of mucosal integrity >5 mm in size, with depth
MEDICAL POLICY SUBJECT: PSYCHOLOGICAL TESTING. POLICY NUMBER: CATEGORY: Behavioral Health
 MEDICAL POLICY SUBJECT: PSYCHOLOGICAL TESTING PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
MEDICAL POLICY SUBJECT: PSYCHOLOGICAL TESTING PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
Management of Helicobacter pylori positive dyspepsia in GPs practice in Slovakia.
 Management of Helicobacter pylori positive dyspepsia in GPs practice in Slovakia. www.vpl.sk P. Lipták, L. Kostková,.I.Vaverková,J.Bendová, M.Fedorová, S.ostrovska, D.Tarinova, I.Machacova, E.Sedlakova,
Management of Helicobacter pylori positive dyspepsia in GPs practice in Slovakia. www.vpl.sk P. Lipták, L. Kostková,.I.Vaverková,J.Bendová, M.Fedorová, S.ostrovska, D.Tarinova, I.Machacova, E.Sedlakova,
Treating H. pylori in 2016
 Treating H. pylori in 2016 William D. Chey, MD, FACG Professor of Medicine University of Michigan The Case: A 38 yo Russian man presents with recurrent epigastric pain which occurs after meals and sometimes
Treating H. pylori in 2016 William D. Chey, MD, FACG Professor of Medicine University of Michigan The Case: A 38 yo Russian man presents with recurrent epigastric pain which occurs after meals and sometimes
MEDICAL POLICY SUBJECT: TUMOR CHEMORESISTANCE AND CHEMOSENSITIVITY ASSAYS. POLICY NUMBER: CATEGORY: Laboratory Test
 MEDICAL POLICY SUBJECT: TUMOR CHEMORESISTANCE AND PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
MEDICAL POLICY SUBJECT: TUMOR CHEMORESISTANCE AND PAGE: 1 OF: 5 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
