GUIDELINES FOR THE MANAGEMENT OF BLADDER CANCER
|
|
|
- Preston Barnett
- 6 years ago
- Views:
Transcription
1 GUIDELINES FOR THE MANAGEMENT OF BLADDER CANCER For approvals and version control see Document Management Record on page 9 Ref: AngCN-SSG-U4 Page 1 of 11 Approved and Published: Aug 2012
2 TABLE OF CONTENTS 1 Introduction Referrals Investigations, staging and treatment guidelines Initial tumour resection New non-muscle invasive Tumours Recurrent non-muscle invasive Tumours Muscle-invasive Tumours Cystectomy High risk disease post-cystectomy (vascular/lymphovascular invasion, pn+, M+, positive margins) Radiotherapy (RT) Radical RT Palliative RT Chemotherapy Follow up after radical treatment Cystectomy Radiotherapy Metastatic disease Upper Tract Transitional Cell Carcinoma Investigation Treatment Follow-up Clinical Trials Portfolio Evidence of Agreement... 9 Page 2 of 11 Approved and Published: Aug 2012
3 1 Introduction The purpose of this manual is to collate the numerous guidelines that exist, into a working manual for the management of bladder cancer within the Anglia Cancer Network. It should act as a summary guide for the management of patients with bladder cancer based on the available published evidence. Its scope is to aid all health practitioners involved in the patient from primary care and referral through treatment to follow up. However, as constant modifications are being made, these guidelines should only be used to give an indication of current management. They should not be used to treat patients without checking that changes have not been made. These guidelines have been developed by discussion between clinicians within the Anglia Cancer Network Urology Site Specific Group but without the establishment of a formal guideline development group. These guidelines have been endorsed by the Anglia Cancer Network Urology Site Specific Group. They will be reviewed and updated on an annual basis or more frequently as required. The guidelines are intended as a working document for daily practice. For more detailed sources, and for educational material, please refer to guidelines produced by e.g. NICE, BAUS, EAU, AUA. 2 Referrals For referrals from Primary and Secondary Care, see AngCN bladder cancer pathway Appendix 1 Bladder Pathway and DH suspected cancer guidance. 2.1 Investigations, staging and treatment guidelines Outpatient assessment of haematuria clinical history and examination FBC, C & E, PSA in men urine culture and consider cytology upper tract imaging (at local discretion) Classification and treatment for patients with bladder cancer. 2.2 Initial tumour resection TURBT under GA or regional anaesthesia Single dose of intravesical chemotherapy At the discretion of the treating consultant, those with very small papillary tumours, those with small indeterminate lesions, or those with obviously solid invasive tumours, may not receive intravesical chemotherapy All tumours classified by Stage (TNM) and Grade LMDT review 2.3 New non-muscle invasive Tumours ptag1 or ptag2 Flexible cystoscopy at three months At three month flexible cystoscopy, assign the patient to a recurrence risk group, as follows. Page 3 of 11 Approved and Published: Aug 2012
4 Initial resection Three month cystoscopy Recurrence risk group Solitary tumour and negative Low risk of recurrence Solitary tumour and positive Medium risk of recurrence or Multifocal tumour and negative Medium risk of recurrence Multifocal tumour and positive High risk of recurrence Subsequent management is recommended as follows: Recurrence risk group Plan Low flexible cystoscopy at one year from diagnosis then annually for seven years from diagnosis then if no recurrence, annual GP symptom review and urinalysis for life Medium intravesical chemotherapy after three month flexible cystoscopy if recurrence then three monthly flexible cystoscopy until one year from diagnosis then six monthly flexible cystoscopy until three years from diagnosis then annual flexible cystoscopy until seven years from diagnosis then if no recurrence, annual GP symptom review and urinalysis for life High six doses of intravesical chemo then 3 monthly flexible cystoscopy until one year from diagnosis then six monthly flexible cystoscopy until three years from diagnosis then annual flexible cystoscopy until seven years from diagnosis then if no recurrence, annual GP symptom review and urinalysis for life pt1 (all grades), ptag3 and ptis (Carcinoma in situ) Patients with high risk superficial bladder cancer (as designated in the 2008 Urology Measures, ptag3, pt1g3, T1G2, carcinoma in situ, and extensive, multifocal or recurrent G2) should be referred for discussion at the SMDT. To simplify the referral process, it has been agreed that the SMDT only requires a copy of the LMDT s outcome form and just the histology slide that demonstrates the relevant pathology. Relevant imaging should be sent where appropriate. The SMDT will confirm an Action Plan for each of these referred patients, For those patients who need to be counselled about the options of BCG and cystectomy, the SMDT will arrange an outpatient appointment at the Cancer centre for the patient to see one of the following Urologists who perform Radical Cystectomy: CUH NNUH Alexandra Colquhoun David Neal William Turner Edwin Ho Vivek Kumar Robert Mills Mark Rochester Krishna Sethia Page 4 of 11 Approved and Published: Aug 2012
5 Intravesical BCG for those patients recommended by the SMDT to have BCG An induction course of six weekly doses of BCG, with GA cystoscopy and biopsy six weeks after 1 st course, then Lamm's maintenance schedule (three instillations at three months, six months, then six monthly for a further three years), with three monthly flexible cystoscopy and cytology for two years, then six monthly for two years, then annually for life. It is recognised that not all patients will complete a full maintenance course due to side effects. In this case alterations to the doses and/or frequency maybe required. 2.4 Recurrent non-muscle invasive Tumours Initial ptag1 or ptag2 disease For single recurrence, resect/biopsy and diathermy, single post operative dose of intravesical chemotherapy if larger than 3 mm max diameter for multiple recurrences, resect/biopsy and diathermy, single post operative dose of intravesical chemotherapy LMDT review Consider full (6 week) course of intravesical chemotherapy / BCG Flexible cystoscopy at three months For large recurrences (>1cm), multiple (>5) or if worsening recurrence history and if patient is fit for radical surgery refer to SMDT to discuss cystectomy. For other patients treated with BCG (ptag3, pt1g2/3 or Cis) If recurrence, resect/biopsy & diathermy Discuss at LMDT If candidate for Radical Surgery to refer to SMDT for discussion of option of cystectomy 2.5 Muscle-invasive Tumours If a new tumour looks solid at initial flexible cystoscopy, arrange MRI before TURBT At TURBT, resect/debulk tumour and consider sending separate bladder tumour base for analysis TUR/cup biopsy of prostatic urethra to assess indication for urethrectomy Document bimanual examination of clinical stage LMDT discussion of histology Refer for discussion at SMDT if muscle-invasive or high risk superficial (as above) MRI pelvis and abdomen, if not done already Chest X-ray, alkaline phosphatase (bone scan, if elevated alkaline phosphatase or otherwise indicated) Discussion of cases at SMDT Assess suitability for neo-adjuvant chemotherapy. To consider options of Radical Cystectomy and Radical radiotherapy If not a suitable candidate for neo-adjuvant chemotherapy, arrange to see in CUH or NNUH Uro- Oncology Clinic if a surgical candidate, and with an Oncologist either locally, or at CUH or NNUH as appropriate. 2.6 Cystectomy Discuss the following: Neo-adjuvant chemotherapy Alternatives to cystectomy are radiotherapy or continued endoscopic management Page 5 of 11 Approved and Published: Aug 2012
6 Diversion/reconstruction options (all those possible for particular patient): Conduit Bladder substitute Catheterisable reservoir only if urethrectomy necessary Check GFR and estimate of differential renal function if any upper tract dilatation, consider if normal upper tracts Consider nephrostomy drainage if upper tract obstruction 2.7 High risk disease post-cystectomy (vascular/lymphovascular invasion, pn+, M+, positive margins) Review histology in SMDT Consider adjuvant chemotherapy 3 Radiotherapy (RT) 3.1 Radical RT Fit patients with muscle invasive disease: ideally those with no ureteric obstruction with small tumours away from trigone Younger fitter patients with limited lymphadenopathy (but no metastases) can be offered radical radiotherapy if good response to neo-adjuvant chemotherapy Patient keen on bladder preservation Preference over radical surgery: Concomitant disease increasing the risks of surgery e.g. cardiovascular disease Inability to manage a stoma 3.2 Palliative RT Palliative radiotherapy is to control symptoms, particularly pelvic pain (not cystitis) and haematuria in the following circumstances: frail patients metastatic disease 4 Chemotherapy Gem/CIS (Gemcitabine and cisplatin) is the current choice for network. See Network Approved Chemotherapy Regimen list and protocol. Restage with CT/MR and EUA towards the end of, or after chemotherapy Page 6 of 11 Approved and Published: Aug 2012
7 5 Follow up after radical treatment 5.1 Cystectomy Clinic six weeks post surgery to: exclude complications of surgery physical examination, serum haemoglobin, and creatinine Further follow-up depends on classification of recurrence risk: Low recurrence risk (worst pathology, T1 or better) Clinic three monthly for two years, then six monthly for two years, then annually for life FBC, C&E and CXR at each visit, PSA if prostate cancer at cystectomy Upper tract FU with US and Nuclear Medicine High recurrence risk (worst pathology, T2 or worse) As for low risk, with CT in first 6-12 weeks, and consideration of adjuvant chemotherapy 5.2 Radiotherapy GA cystoscopy at three months, then three monthly flexible cystoscopy for two years, then six monthly for two years, then annually for life 5.3 Metastatic disease discussion in SMDT staging/restaging consider chemotherapy and Palliative Care referral 6 Upper Tract Transitional Cell Carcinoma 6.1 Investigation as for Haematuria Clinic, and retrograde ureterogram/ureteroscopy and biopsy bone scintigram if elevated alkaline phosphatase or bone pain consider Nuclear medicine assessment of differential renal function 6.2 Treatment Open or laparoscopic nephroureterectomy Local excision or laser ablation are options for low volume pta G1 or G2 disease, after SMDT discussion Consider adjuvant chemotherapy if high risk disease, after SMDT discussion 6.3 Follow-up Check cystoscopy at three, six and 12 months, then annually if clear, until five years flexible ureterorenoscopy for life following local excision or laser ablation Imaging of remaining (contralateral) upper tract if symptomatic Page 7 of 11 Approved and Published: Aug 2012
8 7 Clinical Trials Portfolio The Manual for Cancer Services 2004 states that one of the responsibilities of the MDT Lead Clinician is To ensure mechanisms are in place to support entry of eligible patients into clinical trials. The infrastructure to support clinical trials activity within the Anglia Cancer Network is the responsibility of the two Cancer s (West Anglia Cancer (WACRN) and Anglia East Cancer (AECRN) which provides facilities enabling patient entry into clinical trials in all of its units. The Anglia Cancer Network Urology SSG committed to participation in high quality research studies and clinical trials. Whenever possible, patients should be considered for inclusion in local and national research studies and clinical trials. There is an NSSG agreed single list of clinical trials and research studies supported by the individual MDTs. This is updated at least annually. Further details and protocols are available as follow: Trust Contact Telephone Bedford West Anglia Cancer CUH West Anglia Cancer Hinchingbrooke West Anglia Cancer Ipswich Anglia East Cancer natalie.barber@nnuh.nhs.uk James Paget Anglia East Cancer natalie.barber@nnuh.nhs.uk King s Lynn West Anglia Cancer Norfolk & Anglia East Cancer natalie.barber@nnuh.nhs.uk Norwich Papworth West Anglia Cancer Peterborough West Anglia Cancer elisa.barter@pbh-tr.nhs.uk West Suffolk West Anglia Cancer Page 8 of 11 Approved and Published: Aug 2012
9 8 Evidence of Agreement These Clinical Guidelines have been agreed by: For comments / amendments to these guidelines, please contact: Name Hospital Tel. No Robert Brierly IHT Robert.Brierly@ipswichhospital.nhs.uk For copies of guidelines, please refer to the Anglia Cancer Network website: The Chair of the Network Board Name: Paul Watson Organisation: NHS Suffolk Date agreed: 28 July 2012 The Chair/Joint Chairs of the Urology NSSG Name: Robert Brierly Organisation: IHT Date agreed: 19 July 2012 Name: Kumar Vivekanandan Organisation: NNUHFT Date agreed: 19 July 2012 The NSSG Members This document was discussed at the Urology NSSG Meeting on 19 July 2012 and was agreed to by all members. Document management Document history Review 2 years period: Date placed on electronic library: Authors: Robert Brierly Document Owner: Version number as approved and published: 2 Unique identifier no.: Aug 2012 Anglia Cancer Network Tel: AngCN-SSG-U4 Monitoring the effectiveness of the Process a) Process for Monitoring compliance and Effectiveness - Review of compliance as determined by audit. Any non compliance to be presented by PQ Manager to the AngCN Business Meeting on an annual basis the minutes of this meeting are retained for a minimum of five years. b) Standards/Key Performance Indicators This process forms part of a quality system working to, but not accredited to, International Standard BS EN ISO 9001:2008. The effectiveness of the process will be monitored in accordance with the methods given in the quality manual, AngCN-QM. Equality and Diversity Statement Page 9 of 11 Approved and Published: Aug 2012
10 This document complies with the Suffolk PCT Equality and Diversity statement an EIA assessment is available on request to Anglia Cancer Network PQ Manager, Gibson Centre, Exning Road, Newmarket, CB8 7JG. Disclaimer It is your responsibility to check against the electronic library that this printed out copy is the most recent issue of this document. Please notify any changes required to the Anglia Cancer Network PQ Manager Page 10 of 11 Approved and Published: Aug 2012
11 Appendix 1 Bladder Pathway BLADDER CANCER PATHWAY (E&W) Final Flexi cysto FU Other Referrals i.e. non urgent 18ww referral (via cystoscopy) Result ptag1/2 OPA and OP instillations MMC Result ptag3/cis OPA BCG BCG Urgent GP suspected cancer referral (2WW) One-stop Haematuria Clinic TURBT LOCAL MULTIDISCIPLINARY TEAM (LMDT) MEETING Histology presented Decision to refer to SMDT Result pt1g2/3 OPA then 2nd look TURBT Result <pt1 Result >pt2 OPA Decision to treat (DTT) Options for Cystectomy Cystectomy +/- EUA beforehand Cystectomy +/- EUA beforehand Direct referrals for staging where appropriate Staging +/- CT/MRI/ GFR Result >pt2 OPA Decision to treat (DTT) SPECIALIST MULTIDISCIPLINARY (SMDT) MEETING OPA Decision to treat (DTT) Radiotherapy Neoadjuvant Chemotherapy SMDT / OPA to assess fitness for subsequent treatment Earliest clinically appropriate date, decision to treat (DTT) Radiotherapy +/- EUA beforehand Chemotherapy Palliative Care Palliative Care Consultant upgrade points e.g. referral meets NICE criteria; at first seen, during or after diagnostic tests; on or before MDT date & decision to treat date +62 days from these upgrade point dates Tertiary Referral (to SMDT) Consider Clinical Trial and Follow Up Referral to extended MDT services at any point in pathway e.g. Palliative care specialists, Specialist Nurses and AHPs, education about adverse effects of treatment; access to help and advice from all specialists; staff alert to psychosocial needs; therapists/local healthcare teams, Community Matron, Pharmacists (medicines usage review), Social Services, Mental Health Services, Housing benefits (Social Care and other agencies), GPs, Expert Patient Programme, Smoking Cessation, Alcohol Service (NORCAS), Voluntary organisations, information on prescription, Homeshield (over 60s) Day 0 (Referral) By Day 14 (1st seen) By Day 28 (LMDT meeting) By Day 42 (DTT) By Day 62 (treatment) Day 0 (DTT) By Day 31 (2nd treatment) Key: Unit / Centre Centre Access to specialist services GFOCW Elapsed time for follow up or presentation of recurrence, mets or predetermined gap between treatments Page 11 of 11 Approved and Published: Aug 2012
BLADDER CANCER PATHWAY (E&W) Version 1
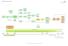 BLADDER CANCER PATHWAY (E&W) Version 1 Flexi cysto FU (via cystoscopy) ptag1/2 and OP instillations MMC ptag3/cis BCG BCG cancer One-stop Haematuria Clinic TURBT Histology presented Decision to refer to
BLADDER CANCER PATHWAY (E&W) Version 1 Flexi cysto FU (via cystoscopy) ptag1/2 and OP instillations MMC ptag3/cis BCG BCG cancer One-stop Haematuria Clinic TURBT Histology presented Decision to refer to
GUIDELINES FOR THE MANAGEMENT OF
 GUIDELINES FOR THE MANAGEMENT OF RENAL CANCER Date of endorsement: July 2011 Authors: Mr. RD Mills & Mr. WH Turner Ref: AngCN-SSG-U3 Page 1 of 14 Approved and Published: Aug 2011 Title: Guidelines for
GUIDELINES FOR THE MANAGEMENT OF RENAL CANCER Date of endorsement: July 2011 Authors: Mr. RD Mills & Mr. WH Turner Ref: AngCN-SSG-U3 Page 1 of 14 Approved and Published: Aug 2011 Title: Guidelines for
3.1 Investigations for Patients Presenting with Haematuria Table 1
 3.1 Investigations for Patients Presenting with Haematuria Table 1 Patients at risk of bacterial endocarditis should be given antibiotic prophylaxis as per local guidelines. Patients with heart valve replacements
3.1 Investigations for Patients Presenting with Haematuria Table 1 Patients at risk of bacterial endocarditis should be given antibiotic prophylaxis as per local guidelines. Patients with heart valve replacements
Brain and CNS tumours Presentation pathway
 Brain and CNS tumours Presentation pathway Ref: AngCN-SSG-BC16 Page 1 of 8 1 Background and Scope This presentation pathway deals with the pathway of referral from all aspects of primary care to hospitals
Brain and CNS tumours Presentation pathway Ref: AngCN-SSG-BC16 Page 1 of 8 1 Background and Scope This presentation pathway deals with the pathway of referral from all aspects of primary care to hospitals
Bladder Cancer Guidelines
 Bladder Cancer Guidelines Agreed by Urology CSG: October 2011 Review Date: September 2013 Bladder Cancer 1. Referral Guidelines The following patients should be considered as potentially having bladder
Bladder Cancer Guidelines Agreed by Urology CSG: October 2011 Review Date: September 2013 Bladder Cancer 1. Referral Guidelines The following patients should be considered as potentially having bladder
Guidelines for the Management of Bladder Cancer West Midlands Expert Advisory Group for Urological Cancer
 Guidelines for the Management of Bladder Cancer West Midlands Expert Advisory Group for Urological Cancer West Midlands Clinical Networks and Clinical Senate Coversheet for Network Expert Advisory Group
Guidelines for the Management of Bladder Cancer West Midlands Expert Advisory Group for Urological Cancer West Midlands Clinical Networks and Clinical Senate Coversheet for Network Expert Advisory Group
Appendix 4 Urology Care Pathways
 Appendix 4 Urology Care Pathways Cancer Care Pathways outline the steps and stages in the patient journey from referral through to diagnostics, staging, treatment, follow up, rehabilitation and if applicable
Appendix 4 Urology Care Pathways Cancer Care Pathways outline the steps and stages in the patient journey from referral through to diagnostics, staging, treatment, follow up, rehabilitation and if applicable
Guidelines for the Management of Bladder Cancer
 Guidelines for the Management of Bladder Cancer Date Approved by Network Governance July 2012 Date for Review July 2015 Changes Between Version 3 and 4 Sections 5.2 and 8 updated Page 1 of 9 1. Scope of
Guidelines for the Management of Bladder Cancer Date Approved by Network Governance July 2012 Date for Review July 2015 Changes Between Version 3 and 4 Sections 5.2 and 8 updated Page 1 of 9 1. Scope of
Chemotherapy Training and Assessment Policy. For Medical Prescribers and Pharmacy Verifiers
 Chemotherapy Training and Assessment Policy For Medical Prescribers and Pharmacy Verifiers For approvals and version control see Document Management Record on page 6 Doc Ref: AngCN-CCG-C36 Approved and
Chemotherapy Training and Assessment Policy For Medical Prescribers and Pharmacy Verifiers For approvals and version control see Document Management Record on page 6 Doc Ref: AngCN-CCG-C36 Approved and
Guidance for the Network Review of Chemotherapy Errors
 Guidance for the Network Review of Chemotherapy Errors For approvals and version control see Document Management Record on page 8 Doc Ref: AngCN-CCG-C31 Approved and published: March 2013 Page 1 of 8 Table
Guidance for the Network Review of Chemotherapy Errors For approvals and version control see Document Management Record on page 8 Doc Ref: AngCN-CCG-C31 Approved and published: March 2013 Page 1 of 8 Table
Guidelines for the Management of Renal Cancer West Midlands Expert Advisory Group for Urological Cancer
 Guidelines for the Management of Renal Cancer West Midlands Expert Advisory Group for Urological Cancer West Midlands Clinical Networks and Clinical Senate Coversheet for Network Expert Advisory Group
Guidelines for the Management of Renal Cancer West Midlands Expert Advisory Group for Urological Cancer West Midlands Clinical Networks and Clinical Senate Coversheet for Network Expert Advisory Group
MUSCLE - INVASIVE AND METASTATIC BLADDER CANCER
 10 MUSCLE - INVASIVE AND METASTATIC BLADDER CANCER Recommendations from the EAU Working Party on Muscle Invasive and Metastatic Bladder Cancer G. Jakse (chairman), F. Algaba, S. Fossa, A. Stenzl, C. Sternberg
10 MUSCLE - INVASIVE AND METASTATIC BLADDER CANCER Recommendations from the EAU Working Party on Muscle Invasive and Metastatic Bladder Cancer G. Jakse (chairman), F. Algaba, S. Fossa, A. Stenzl, C. Sternberg
Ref No: AngCN-SSG-Sa9. H:\Cancer Network\Tumour Site\Sarcoma\Peer Review\Active\AngCN-SSG- Sa9_v2_Anglia_Configuration_Sarcoma_Services.
 Anglia Cancer Network Configuration of Sarcoma Services Please note this document has only been partially approved. For further details, approvals and version control please see Document Management Record
Anglia Cancer Network Configuration of Sarcoma Services Please note this document has only been partially approved. For further details, approvals and version control please see Document Management Record
UROLOGY CANCER 2009 COMPARATIVE AUDIT REPORT
 Urological Cancer Audit 2009 SOUTH EAST SCOTLAND CANCER NETWORK PROSPECTIVE CANCER AUDIT UROLOGY CANCER 2009 COMPARATIVE AUDIT REPORT Dr Prasad Bollina, NHS Lothian SCAN Lead Urology Cancer Clinician Dr
Urological Cancer Audit 2009 SOUTH EAST SCOTLAND CANCER NETWORK PROSPECTIVE CANCER AUDIT UROLOGY CANCER 2009 COMPARATIVE AUDIT REPORT Dr Prasad Bollina, NHS Lothian SCAN Lead Urology Cancer Clinician Dr
Guideline for the Management of Vulval Cancer
 Version History Guideline for the Management of Vulval Cancer Version Date Brief Summary of Change Issued 2.0 20.02.08 Endorsed by the Governance Committee 2.1 19.11.10 Circulated at NSSG meeting 2.2 13.04.11
Version History Guideline for the Management of Vulval Cancer Version Date Brief Summary of Change Issued 2.0 20.02.08 Endorsed by the Governance Committee 2.1 19.11.10 Circulated at NSSG meeting 2.2 13.04.11
UROLOGICAL CANCER 2010 COMPARATIVE AUDIT REPORT
 SOUTH EAST SCOTLAND CANCER NETWORK PROSPECTIVE CANCER AUDIT UROLOGICAL CANCER 2010 COMPARATIVE AUDIT REPORT Dr Prasad Bollina, NHS Lothian SCAN Lead Urology Cancer Clinician Dr Prasad Bollina, NHS Lothian
SOUTH EAST SCOTLAND CANCER NETWORK PROSPECTIVE CANCER AUDIT UROLOGICAL CANCER 2010 COMPARATIVE AUDIT REPORT Dr Prasad Bollina, NHS Lothian SCAN Lead Urology Cancer Clinician Dr Prasad Bollina, NHS Lothian
Care of bladder cancer patients diagnosed in Northern Ireland 2010 & 2011 (Summary)
 Care of bladder cancer patients diagnosed in 2010 & 2011 (Summary) Bannon, F., Ranaghan, L., & Gavin, A. (2014). Care of bladder cancer patients diagnosed in 2010 & 2011 (Summary). N. Cancer Registry,
Care of bladder cancer patients diagnosed in 2010 & 2011 (Summary) Bannon, F., Ranaghan, L., & Gavin, A. (2014). Care of bladder cancer patients diagnosed in 2010 & 2011 (Summary). N. Cancer Registry,
Bladder Cancer in Primary Care. Dr Penny Kehagioglou Consultant Clinical Oncologist
 Bladder Cancer in Primary Care Dr Penny Kehagioglou Consultant Clinical Oncologist Objectives Patient presentation in primary care Investigating bladder cancer Management of bladder cancer Differential
Bladder Cancer in Primary Care Dr Penny Kehagioglou Consultant Clinical Oncologist Objectives Patient presentation in primary care Investigating bladder cancer Management of bladder cancer Differential
The innovative aspect is that it detects bladder cancer based on the novel biomarker, minichromosome maintenance complex component 5 (MCM5).
 pat hways ADXBLADDER for detecting bladder cancer Medtech innovation briefing Published: 12 April 2019 nice.org.uk/guidance/mib180 Summary The technology described in this briefing is ADXBLADDER. It is
pat hways ADXBLADDER for detecting bladder cancer Medtech innovation briefing Published: 12 April 2019 nice.org.uk/guidance/mib180 Summary The technology described in this briefing is ADXBLADDER. It is
Bladder tumour resection (TURBT): procedure-specific information
 PATIENT INFORMATION Bladder tumour resection (TURBT): procedure-specific information What is the evidence base for this information? This leaflet includes advice from consensus panels, the British Association
PATIENT INFORMATION Bladder tumour resection (TURBT): procedure-specific information What is the evidence base for this information? This leaflet includes advice from consensus panels, the British Association
Guidelines for Management of Penile Cancer
 Guidelines for Management of Penile Cancer Date Approved by Network Governance July 2012 Date for Review July 2015 Changes Between Versions 2 and 3 Sections 3, 5, 6 and 16 updated. Page 1 of 10 1. Scope
Guidelines for Management of Penile Cancer Date Approved by Network Governance July 2012 Date for Review July 2015 Changes Between Versions 2 and 3 Sections 3, 5, 6 and 16 updated. Page 1 of 10 1. Scope
INTERNAL VALIDATION REPORT (MULTI-DISCIPLINARY TEAM)
 INTERNAL VALIDATION REPORT (MULTI-DISCIPLINARY TEAM) Network Trust MDT NECN N Tees & Hartlepool North Tees And Hartlepool Date Self Assessment Completed 23rd July 2009 Date of IV Review 19th June 2009
INTERNAL VALIDATION REPORT (MULTI-DISCIPLINARY TEAM) Network Trust MDT NECN N Tees & Hartlepool North Tees And Hartlepool Date Self Assessment Completed 23rd July 2009 Date of IV Review 19th June 2009
West Midlands Sarcoma Advisory Group
 West Midlands Sarcoma Advisory Group Guideline for the Initial Investigation and Referral to Specialist Sarcoma Multi Disciplinary Team for Suspected Bone Sarcoma Version History Version Date Brief Summary
West Midlands Sarcoma Advisory Group Guideline for the Initial Investigation and Referral to Specialist Sarcoma Multi Disciplinary Team for Suspected Bone Sarcoma Version History Version Date Brief Summary
Skin SSG (Anglia East & Anglia West)
 Guidelines for Referrals between Skin LMDT and SMDT Skin SSG (Anglia East & Anglia West) Author: Dr Jennifer Garioch, Consultant Dermatologist Dr Pamela Todd, Consultant Dermatologist Approved by: Anglia
Guidelines for Referrals between Skin LMDT and SMDT Skin SSG (Anglia East & Anglia West) Author: Dr Jennifer Garioch, Consultant Dermatologist Dr Pamela Todd, Consultant Dermatologist Approved by: Anglia
Guidelines for the Management of Prostate Cancer West Midlands Expert Advisory Group for Urological Cancer
 Guidelines for the Management of Prostate Cancer West Midlands Expert Advisory Group for Urological Cancer West Midlands Clinical Networks and Clinical Senate Coversheet for Network Expert Advisory Group
Guidelines for the Management of Prostate Cancer West Midlands Expert Advisory Group for Urological Cancer West Midlands Clinical Networks and Clinical Senate Coversheet for Network Expert Advisory Group
Guidelines for the Management of Suspected Sarcoma in Primary Care
 Guidelines for the Management of Suspected Sarcoma in Primary Care Author: Anglia Cancer Network Sarcoma SSG Document Approved Date: 16-Dec-10 Review Date: December 2012 Ref Code: AngCN-SSG-Sa1 Status:
Guidelines for the Management of Suspected Sarcoma in Primary Care Author: Anglia Cancer Network Sarcoma SSG Document Approved Date: 16-Dec-10 Review Date: December 2012 Ref Code: AngCN-SSG-Sa1 Status:
LCA Lung Clinical Forum. 21 st October 2014
 LCA Lung Clinical Forum 21 st October 2014 Welcome Dr Liz Sawicka Chair - LCA Lung Pathway Group Succession planning Dr Kate Haire Consultant in Public Health Medicine, LCA Commissioning Intentions for
LCA Lung Clinical Forum 21 st October 2014 Welcome Dr Liz Sawicka Chair - LCA Lung Pathway Group Succession planning Dr Kate Haire Consultant in Public Health Medicine, LCA Commissioning Intentions for
Attachment #2 Overview of Follow-up
 Attachment #2 Overview of Follow-up Provided below is a general overview of follow-up and this may vary based on specific patient or cancer characteristics. Of note, Labs and imaging can be performed closer
Attachment #2 Overview of Follow-up Provided below is a general overview of follow-up and this may vary based on specific patient or cancer characteristics. Of note, Labs and imaging can be performed closer
Guideline for the Management of Patients Suitable for Immediate Breast Reconstruction
 Version History Guideline for the Management of Patients Suitable for Immediate Breast Reconstruction Version Summary of change Date Issued 2.0 Endorsed by the Governance Committee 20.02.08 2.1 Circulated
Version History Guideline for the Management of Patients Suitable for Immediate Breast Reconstruction Version Summary of change Date Issued 2.0 Endorsed by the Governance Committee 20.02.08 2.1 Circulated
Bladder cancer - suspected
 Background information Information resources for patients and carers Updates to this care map Bladder cancer - clinical presentation History Examination Consider differential diagnoses Clinical indications
Background information Information resources for patients and carers Updates to this care map Bladder cancer - clinical presentation History Examination Consider differential diagnoses Clinical indications
West Midlands Sarcoma Advisory Group
 West Midlands Sarcoma Advisory Group Guideline for the Initial Investigation and Referral to Sarcoma Specialist Multi Disciplinary Team for Suspected Sarcoma of Soft Tissue Extremities (limbs and trunk
West Midlands Sarcoma Advisory Group Guideline for the Initial Investigation and Referral to Sarcoma Specialist Multi Disciplinary Team for Suspected Sarcoma of Soft Tissue Extremities (limbs and trunk
SELF ASSESSMENT REPORT (MULTI-DISCIPLINARY TEAM)
 SELF ASSESSMENT REPORT (MULTI-DISCIPLINARY TEAM) Network Trust MDT MDT Lead Clinician ASWCN TAUNTON AND SOMERSET Taunton Lung MDT (11-2C-1) - 2011/12 Dr Sarah Foster Compliance Self Assessment LUNG MDT
SELF ASSESSMENT REPORT (MULTI-DISCIPLINARY TEAM) Network Trust MDT MDT Lead Clinician ASWCN TAUNTON AND SOMERSET Taunton Lung MDT (11-2C-1) - 2011/12 Dr Sarah Foster Compliance Self Assessment LUNG MDT
Costing report: Bladder cancer
 Putting NICE guidance into practice Costing report: Bladder cancer Implementing the NICE guideline on bladder cancer (NG2) Published: February 2015 Updated September 2015 to update the unit cost of transurethral
Putting NICE guidance into practice Costing report: Bladder cancer Implementing the NICE guideline on bladder cancer (NG2) Published: February 2015 Updated September 2015 to update the unit cost of transurethral
Shared Care Pathway for Soft Tissue Sarcomas Presenting to Site Specialised MDTs Lung /chest wall sarcomas incl. pulmonary metastatectomy Version 2
 Shared Care Pathway for Soft Tissue Sarcomas Presenting to Site Specialised MDTs Lung /chest wall sarcomas incl. pulmonary metastatectomy Version 2 Background Sarcomas that arise in the lung de novo are
Shared Care Pathway for Soft Tissue Sarcomas Presenting to Site Specialised MDTs Lung /chest wall sarcomas incl. pulmonary metastatectomy Version 2 Background Sarcomas that arise in the lung de novo are
Attachment #2 Overview of Follow-up
 Attachment #2 Overview of Follow-up Provided below is a general overview of follow-up and this may vary based on specific patient or cancer characteristics. Of note, Labs and imaging can be performed closer
Attachment #2 Overview of Follow-up Provided below is a general overview of follow-up and this may vary based on specific patient or cancer characteristics. Of note, Labs and imaging can be performed closer
Guideline for the Follow-up of Patients with Gynaecological Malignancies
 Guideline for the Follow-up of Patients with Gynaecological Malignancies Version History Version Date Summary of Change/Process 2.0 20.02.08 Endorsed by the Governance Committee 2.1 18.11.10 Circulated
Guideline for the Follow-up of Patients with Gynaecological Malignancies Version History Version Date Summary of Change/Process 2.0 20.02.08 Endorsed by the Governance Committee 2.1 18.11.10 Circulated
Inspection/examination of the ureter & biopsy : procedure-specific information
 PATIENT INFORMATION Inspection/examination of the ureter & biopsy : procedure-specific information What is the evidence base for this information? This leaflet includes advice from consensus panels, the
PATIENT INFORMATION Inspection/examination of the ureter & biopsy : procedure-specific information What is the evidence base for this information? This leaflet includes advice from consensus panels, the
MUSCLE-INVASIVE AND METASTATIC BLADDER CANCER
 MUSCLE-INVASIVE AND METASTATIC BLADDER CANCER (Text update March 2008) A. Stenzl (chairman), N.C. Cowan, M. De Santis, G. Jakse, M. Kuczyk, A.S. Merseburger, M.J. Ribal, A. Sherif, J.A. Witjes Introduction
MUSCLE-INVASIVE AND METASTATIC BLADDER CANCER (Text update March 2008) A. Stenzl (chairman), N.C. Cowan, M. De Santis, G. Jakse, M. Kuczyk, A.S. Merseburger, M.J. Ribal, A. Sherif, J.A. Witjes Introduction
North of Scotland Cancer Network Clinical Management Guideline for Metastatic Malignancy of Undefined Primary Origin (MUO)
 North of Scotland Cancer Network Clinical Management Guideline for Metastatic Malignancy of Undefined Primary Origin (MUO) UNCONTROLLED WHEN PRINTED DOCUMENT CONTROL Original Prepared by NMcL April 2016
North of Scotland Cancer Network Clinical Management Guideline for Metastatic Malignancy of Undefined Primary Origin (MUO) UNCONTROLLED WHEN PRINTED DOCUMENT CONTROL Original Prepared by NMcL April 2016
NICE Quality Standards and COF
 NICE Quality Standards and COF David Baldwin Consultant Respiratory Physician NUH Hon Senior Lecturer Nottingham University Clinical lead NICE lung cancer GL Chair NICE QS Topic Expert Group Quality Standards
NICE Quality Standards and COF David Baldwin Consultant Respiratory Physician NUH Hon Senior Lecturer Nottingham University Clinical lead NICE lung cancer GL Chair NICE QS Topic Expert Group Quality Standards
Work Programme/Service Delivery Plan 2010/2013
 Essex and East Suffolk Gynaecological Cancer Network Site Specific Group Work Programme/Service Delivery Plan 2010/2013 Version Number 1.2 Author Members of the NSSG Date Written June 2010 Reviewed May
Essex and East Suffolk Gynaecological Cancer Network Site Specific Group Work Programme/Service Delivery Plan 2010/2013 Version Number 1.2 Author Members of the NSSG Date Written June 2010 Reviewed May
Citation International journal of urology (2. Right which has been published in final f
 Title Novel constant-pressure irrigation of renal pelvic tumors after ipsila Nakamura, Kenji; Terada, Naoki; Sug Author(s) Toshinori; Matsui, Yoshiyuki; Imamu Kazutoshi; Kamba, Tomomi; Yoshimura Citation
Title Novel constant-pressure irrigation of renal pelvic tumors after ipsila Nakamura, Kenji; Terada, Naoki; Sug Author(s) Toshinori; Matsui, Yoshiyuki; Imamu Kazutoshi; Kamba, Tomomi; Yoshimura Citation
Cancer of Unknown Primary (CUP) Protocol
 1 Department of Oncology. Cancer of Unknown Primary (CUP) Protocol Version: Document type: Document sponsor Designation Document author [ s] Designation[s] Approving committee / Group Ratified by: Date
1 Department of Oncology. Cancer of Unknown Primary (CUP) Protocol Version: Document type: Document sponsor Designation Document author [ s] Designation[s] Approving committee / Group Ratified by: Date
Manchester Cancer Colorectal Pathway Board: Guidelines for management of colorectal hepatic metastases
 Manchester Cancer Colorectal Pathway Board: Guidelines for management of colorectal hepatic metastases Date: April 2015 Date for review: April 2018 1. Principles The recognised specialist HPB MDT for Greater
Manchester Cancer Colorectal Pathway Board: Guidelines for management of colorectal hepatic metastases Date: April 2015 Date for review: April 2018 1. Principles The recognised specialist HPB MDT for Greater
North of Scotland Cancer Network Clinical Management Guideline for Carcinoma of the Uterine Cervix
 THIS DOCUMENT North of Scotland Cancer Network Carcinoma of the Uterine Cervix UNCONTROLLED WHEN PRINTED DOCUMENT CONTROL Prepared by A Kennedy/AG Macdonald/Others Approved by NOT APPROVED Issue date April
THIS DOCUMENT North of Scotland Cancer Network Carcinoma of the Uterine Cervix UNCONTROLLED WHEN PRINTED DOCUMENT CONTROL Prepared by A Kennedy/AG Macdonald/Others Approved by NOT APPROVED Issue date April
Improving services for upper GI (OG) cancer Application template (Version 2)
 Trust Clinical lead Improving services for upper GI (OG) cancer Application template (Version 2) Managerial lead Date completed 14 June 2013 Barnet & Chase Farm Hospitals NHS Trust Dr Marta Carpani Upper
Trust Clinical lead Improving services for upper GI (OG) cancer Application template (Version 2) Managerial lead Date completed 14 June 2013 Barnet & Chase Farm Hospitals NHS Trust Dr Marta Carpani Upper
Intravesical immunotherapy (known as BCG therapy): procedure-specific information
 PATIENT INFORMATION Intravesical immunotherapy (known as BCG therapy): procedure-specific information What is the evidence base for this information? This leaflet includes advice from consensus panels,
PATIENT INFORMATION Intravesical immunotherapy (known as BCG therapy): procedure-specific information What is the evidence base for this information? This leaflet includes advice from consensus panels,
Update on Haematuria and Bladder Cancer
 Update on Haematuria and Bladder Cancer Hugh Mostafid FRCS(urol) FEBU Consultant Urologist, Royal Surrey County Hospital and Honorary Senior Lecturer, University of Surrey Guildford None Declarations Recent
Update on Haematuria and Bladder Cancer Hugh Mostafid FRCS(urol) FEBU Consultant Urologist, Royal Surrey County Hospital and Honorary Senior Lecturer, University of Surrey Guildford None Declarations Recent
NICE guideline Published: 25 February 2015 nice.org.uk/guidance/ng2
 Bladder cancer: diagnosis and management NICE guideline Published: 25 February 2015 nice.org.uk/guidance/ng2 NICE 2017. All rights reserved. Subject to Notice of rights (https://www.nice.org.uk/terms-and-conditions#notice-ofrights).
Bladder cancer: diagnosis and management NICE guideline Published: 25 February 2015 nice.org.uk/guidance/ng2 NICE 2017. All rights reserved. Subject to Notice of rights (https://www.nice.org.uk/terms-and-conditions#notice-ofrights).
Urological Tumours 1 Kidney tumours 2 Bladder tumours
 Urological Tumours 1 Kidney tumours 2 Bladder tumours Tim Bracey SpR Histopathology Derriford Hospital Kidney tumours What are we going to talk about?! Anatomy of urinary tract! Types of kidney tumours!
Urological Tumours 1 Kidney tumours 2 Bladder tumours Tim Bracey SpR Histopathology Derriford Hospital Kidney tumours What are we going to talk about?! Anatomy of urinary tract! Types of kidney tumours!
National Optimal Lung Cancer Pathway
 National Optimal Lung Cancer Pathway This document was produced by the Lung Clinical Expert Group 2017 Document Title: National Optimal Lung Cancer Pathway and Implementation Guide Date of issue: August
National Optimal Lung Cancer Pathway This document was produced by the Lung Clinical Expert Group 2017 Document Title: National Optimal Lung Cancer Pathway and Implementation Guide Date of issue: August
Shared Care Pathway for Soft Tissue Sarcomas Presenting to Site Specialised MDTs. Gynaecological sarcomas Version 1
 Shared Care Pathway for Soft Tissue Sarcomas Presenting to Site Specialised MDTs Gynaecological sarcomas Version 1 Background This guidance is to provide direction for the management of patients with sarcomas
Shared Care Pathway for Soft Tissue Sarcomas Presenting to Site Specialised MDTs Gynaecological sarcomas Version 1 Background This guidance is to provide direction for the management of patients with sarcomas
RECTAL CANCER APPARENT COMPLETE RESPONSE (acr) AFTER LONG COURSE CHEMORADIOTHERAPY
 COLORECTAL CLINICAL SUBGROUP RECTAL CANCER APPARENT COMPLETE RESPONSE (acr) AFTER LONG COURSE CHEMORADIOTHERAPY Finalised by: Dr Simon Gollins Mr Andrew Renehan Dr Mark Saunders Mr Nigel Scott Dr Shabbir
COLORECTAL CLINICAL SUBGROUP RECTAL CANCER APPARENT COMPLETE RESPONSE (acr) AFTER LONG COURSE CHEMORADIOTHERAPY Finalised by: Dr Simon Gollins Mr Andrew Renehan Dr Mark Saunders Mr Nigel Scott Dr Shabbir
Cancer of Unknown Primary Service
 Cancer of Unknown Primary Service Dr Maurice Fernando Consultant In Specialist Palliative Care and CUP lead Doncaster and Bassetlaw Hospitals NHS FT Wakefield meeting -14-07-2016 CUP service CUP MDT
Cancer of Unknown Primary Service Dr Maurice Fernando Consultant In Specialist Palliative Care and CUP lead Doncaster and Bassetlaw Hospitals NHS FT Wakefield meeting -14-07-2016 CUP service CUP MDT
Non Muscle Invasive Bladder Cancer. Primary and Recurrent TCC 4/10/2010. Two major consequences: Strategies: High-Risk NMI TCC
 Intravesical Therapy 2010-When, with What, When to Stop Friday, April 9, 2010 Ralph de VereWhite, MD Director, UC Davis Cancer Center Associate Dean for Cancer Programs Professor, Department of Urolgoy
Intravesical Therapy 2010-When, with What, When to Stop Friday, April 9, 2010 Ralph de VereWhite, MD Director, UC Davis Cancer Center Associate Dean for Cancer Programs Professor, Department of Urolgoy
SELF ASSESSMENT REPORT (MULTI-DISCIPLINARY TEAM)
 SELF ASSESSMENT REPORT (MULTI-DISCIPLINARY TEAM) Network Trust MDT MDT Lead Clinician GMCN ROYAL WOLVERHAMPTON HOSPITALS The Royal Wolverhampton Hospitals Trust Lung MDT (11-2C-1) - 2011/12 Dr Angela Morgan
SELF ASSESSMENT REPORT (MULTI-DISCIPLINARY TEAM) Network Trust MDT MDT Lead Clinician GMCN ROYAL WOLVERHAMPTON HOSPITALS The Royal Wolverhampton Hospitals Trust Lung MDT (11-2C-1) - 2011/12 Dr Angela Morgan
The group seemed largely happy with the proposed GDG membership. Additional comments and clarifications as below:
 Bladder Cancer Guideline Draft Scope Stakeholder Workshop: Group Notes Group 1 John Graham Andrew Champion Julia Verne Elise Hasler Nathan Bromham Suzanne Langley Sue Norman Lyndon Gommersall Dr Kate Williamson
Bladder Cancer Guideline Draft Scope Stakeholder Workshop: Group Notes Group 1 John Graham Andrew Champion Julia Verne Elise Hasler Nathan Bromham Suzanne Langley Sue Norman Lyndon Gommersall Dr Kate Williamson
Staging and Grading Last Updated Friday, 14 November 2008
 Staging and Grading Last Updated Friday, 14 November 2008 There is a staging graph below Blood in the urine is the most common indication that something is wrong. Often one will experience pain or difficulty
Staging and Grading Last Updated Friday, 14 November 2008 There is a staging graph below Blood in the urine is the most common indication that something is wrong. Often one will experience pain or difficulty
Glossary of Terms Primary Urethral Cancer
 Patient Information English Glossary of Terms Primary Urethral Cancer Advanced cancer A tumour that grows into deeper layers of tissue, adjacent organs, or surrounding muscles. Anaesthesia (general, spinal,
Patient Information English Glossary of Terms Primary Urethral Cancer Advanced cancer A tumour that grows into deeper layers of tissue, adjacent organs, or surrounding muscles. Anaesthesia (general, spinal,
Guidelines for the Management of Adult Patients with Urological Cancers 2013
 Guidelines for the Management of Adult Patients with Urological Cancers 2013 Version Control This is a controlled document please destroy all previous versions on receipt of a new version. Date Approved:
Guidelines for the Management of Adult Patients with Urological Cancers 2013 Version Control This is a controlled document please destroy all previous versions on receipt of a new version. Date Approved:
Percutaneous removal of kidney stone(s): procedurespecific information
 PATIENT INFORMATION Percutaneous removal of kidney stone(s): procedurespecific information What is the evidence base for this information? This leaflet includes advice from consensus panels, the British
PATIENT INFORMATION Percutaneous removal of kidney stone(s): procedurespecific information What is the evidence base for this information? This leaflet includes advice from consensus panels, the British
A patient with recurrent bladder cancer presents with the following history:
 MP/H Quiz A patient with recurrent bladder cancer presents with the following history: 9/23/06 TURB 1/12/07 TURB 4/1/07 TURB 7/12/07 TURB 11/14/07 Non-invasive papillary transitional cell carcinoma from
MP/H Quiz A patient with recurrent bladder cancer presents with the following history: 9/23/06 TURB 1/12/07 TURB 4/1/07 TURB 7/12/07 TURB 11/14/07 Non-invasive papillary transitional cell carcinoma from
Guideline for the Diagnosis of Breast Cancer
 Guideline for the Diagnosis of Breast Cancer Version History Version Date Brief Summary of Change Issued 2.0 May 2007 Approved by the Governance Committee 2.0 25.11.08 Discussed at the NSSG 2.1 5.12.08
Guideline for the Diagnosis of Breast Cancer Version History Version Date Brief Summary of Change Issued 2.0 May 2007 Approved by the Governance Committee 2.0 25.11.08 Discussed at the NSSG 2.1 5.12.08
When to Integrate Surgery for Metatstatic Urothelial Cancers
 When to Integrate Surgery for Metatstatic Urothelial Cancers Wade J. Sexton, M.D. Senior Member and Professor Department of Genitourinary Oncology Moffitt Cancer Center Case Presentation #1 67 yo male
When to Integrate Surgery for Metatstatic Urothelial Cancers Wade J. Sexton, M.D. Senior Member and Professor Department of Genitourinary Oncology Moffitt Cancer Center Case Presentation #1 67 yo male
PATHWAY MANAGEMENT OF METASTATIC SPINAL CORD COMPRESSION (MSCC) THE CHRISTIE, GREATER MANCHESTER & CHESHIRE
 PATHWAY MANAGEMENT OF METASTATIC SPINAL CORD COMPRESSION (MSCC) THE CHRISTIE, GREATER MANCHESTER & CHESHIRE Procedure Reference: Document Owner: Dr V. Misra Version: Accountable Committee: V3 MSCC Network
PATHWAY MANAGEMENT OF METASTATIC SPINAL CORD COMPRESSION (MSCC) THE CHRISTIE, GREATER MANCHESTER & CHESHIRE Procedure Reference: Document Owner: Dr V. Misra Version: Accountable Committee: V3 MSCC Network
CHEMO-RADIOTHERAPY FOR BLADDER CANCER. Dr Darren Mitchell Consultant Clinical Oncologist Northern Ireland Cancer Centre
 CHEMO-RADIOTHERAPY FOR BLADDER CANCER Dr Darren Mitchell Consultant Clinical Oncologist Northern Ireland Cancer Centre AIMS Muscle invasive disease Current Gold-Standard Rationale behind Chemo-Radiotherapy
CHEMO-RADIOTHERAPY FOR BLADDER CANCER Dr Darren Mitchell Consultant Clinical Oncologist Northern Ireland Cancer Centre AIMS Muscle invasive disease Current Gold-Standard Rationale behind Chemo-Radiotherapy
BLADDER CANCER: PATIENT INFORMATION
 BLADDER CANCER: PATIENT INFORMATION The bladder is the balloon like organ located in the pelvis that stores and empties urine. Urine is produced by the kidneys, is conducted to the bladder by the ureters,
BLADDER CANCER: PATIENT INFORMATION The bladder is the balloon like organ located in the pelvis that stores and empties urine. Urine is produced by the kidneys, is conducted to the bladder by the ureters,
Skin SSG (Anglia East & Anglia West)
 Skin SSG (Anglia East & Anglia West) Author: Dr Jennifer Garioch, Consultant Dermatologist Dr Pamela Todd, Consultant Dermatologist Approved by: Anglia Cancer Network Skin NSSG Approved on: Reviewed and
Skin SSG (Anglia East & Anglia West) Author: Dr Jennifer Garioch, Consultant Dermatologist Dr Pamela Todd, Consultant Dermatologist Approved by: Anglia Cancer Network Skin NSSG Approved on: Reviewed and
Non-Muscle Invasive Bladder Cancer BCG Failures: University of Iowa Hospitals and Clinics Experience. Paul Gellhaus Assistant Clinical Professor
 Non-Muscle Invasive Bladder Cancer BCG Failures: University of Iowa Hospitals and Clinics Experience Paul Gellhaus Assistant Clinical Professor Iowa??? none Disclosures Caveats Dr. Michael O Donnell
Non-Muscle Invasive Bladder Cancer BCG Failures: University of Iowa Hospitals and Clinics Experience Paul Gellhaus Assistant Clinical Professor Iowa??? none Disclosures Caveats Dr. Michael O Donnell
National Breast Cancer Audit next steps. Martin Lee
 National Breast Cancer Audit next steps Martin Lee National Cancer Audits Current Bowel Cancer Head & Neck Cancer Lung cancer Oesophagogastric cancer New Prostate Cancer - undergoing procurement Breast
National Breast Cancer Audit next steps Martin Lee National Cancer Audits Current Bowel Cancer Head & Neck Cancer Lung cancer Oesophagogastric cancer New Prostate Cancer - undergoing procurement Breast
North of Scotland Cancer Network Clinical Management Guideline for Oropharyngeal Cancer
 Nth of Scotland Cancer Netwk Clinical Management Guideline f Oropharyngeal Cancer UNCONTROLLED WHEN PRINTED Based on NHST CMG with further extensive consultation within NOSCAN DOCUMENT CONTROL Original
Nth of Scotland Cancer Netwk Clinical Management Guideline f Oropharyngeal Cancer UNCONTROLLED WHEN PRINTED Based on NHST CMG with further extensive consultation within NOSCAN DOCUMENT CONTROL Original
Case 1. Receives induction BCG weekly x 6 without significant toxicity Next step should be:
 Case 1 89 year old male with initial occurrence of gross hematuria Office flexible cystoscopy shows two papillary tumors with some surface necrosis Complete TURBT into muscle Florescence cysto shows two
Case 1 89 year old male with initial occurrence of gross hematuria Office flexible cystoscopy shows two papillary tumors with some surface necrosis Complete TURBT into muscle Florescence cysto shows two
Sarcoma. National Follow-up Guidelines
 North, South East and West of Scotland Cancer Networks Sarcoma National Managed Clinical Network Sarcoma National Follow-up Guidelines Prepared by Dr Ioanna Nixon, Dr Lucy Wall and Lindsay Campbell Approved
North, South East and West of Scotland Cancer Networks Sarcoma National Managed Clinical Network Sarcoma National Follow-up Guidelines Prepared by Dr Ioanna Nixon, Dr Lucy Wall and Lindsay Campbell Approved
Panel: A Case-based Approach to the Management of Bladder Cancer
 Panel: A Case-based Approach to the Management of Bladder Cancer ~ Moderator: Robert Donohue, MD Panel: David C. Beyer, MD E. David Crawford, MD Donald L. Lamm, MD Paul D. Maroni, MD TCC Cases Robert E.
Panel: A Case-based Approach to the Management of Bladder Cancer ~ Moderator: Robert Donohue, MD Panel: David C. Beyer, MD E. David Crawford, MD Donald L. Lamm, MD Paul D. Maroni, MD TCC Cases Robert E.
Information for Patients. Bladder Cancer. English
 Information for Patients Bladder Cancer English Table of contents What is the function of the bladder?... 3 What is bladder cancer?... 3 What causes bladder cancer?... 3 Stages of the disease... 3 Risk
Information for Patients Bladder Cancer English Table of contents What is the function of the bladder?... 3 What is bladder cancer?... 3 What causes bladder cancer?... 3 Stages of the disease... 3 Risk
Chemotherapy Treatment Algorithms for Urology Cancer
 Chemotherapy Treatment Algorithms for Urology Cancer Chemoradiation for bladder cancer; Chemotherapy algorithm for non TCC bladder cancer Squamous cell carcinoma; Chemotherapy Algorithm for Non Transitional
Chemotherapy Treatment Algorithms for Urology Cancer Chemoradiation for bladder cancer; Chemotherapy algorithm for non TCC bladder cancer Squamous cell carcinoma; Chemotherapy Algorithm for Non Transitional
One Stop Prostate Biopsy Protocol Author Consultation Date Approved
 One Stop Prostate Biopsy Protocol Author Consultation Date Approved Urology Nurse Practioner PROTOCOL FOR MEN ATTENDING A ONE STOP PROSTATE BIOPSY CLINIC RATIONALE Prostate cancer is the most common cancer
One Stop Prostate Biopsy Protocol Author Consultation Date Approved Urology Nurse Practioner PROTOCOL FOR MEN ATTENDING A ONE STOP PROSTATE BIOPSY CLINIC RATIONALE Prostate cancer is the most common cancer
Health Board/Region: All-Wales
 Peer Review: Cancer Sub-site: Gynaecology Health Board/Region: All-Wales Cycle: Second Date of review: February 2018 This report describes the findings and themes observed by clinical review panels during
Peer Review: Cancer Sub-site: Gynaecology Health Board/Region: All-Wales Cycle: Second Date of review: February 2018 This report describes the findings and themes observed by clinical review panels during
Trust Guideline for the inclusion of women at High Risk of Breast Cancer in the NHS Breast Screening Programme
 Trust Guideline for the inclusion of women at High Risk of Breast Cancer in the NHS Breast Screening Programme For Use in: By: For: Division responsible for document: Key words: Name and job title of document
Trust Guideline for the inclusion of women at High Risk of Breast Cancer in the NHS Breast Screening Programme For Use in: By: For: Division responsible for document: Key words: Name and job title of document
There are a number of national guidelines and performance standards which support the implementation of a straight to CT pathway.
 December 2015 CONTENTS Contents... 2 1 Introduction... 3 2 Case for Change... 3 3 Evidence... 3 3.1 National and regional policy... 3 3.2 Local audit... 4 4 Supporting Work Initiatives... 5 4.1 Identification
December 2015 CONTENTS Contents... 2 1 Introduction... 3 2 Case for Change... 3 3 Evidence... 3 3.1 National and regional policy... 3 3.2 Local audit... 4 4 Supporting Work Initiatives... 5 4.1 Identification
Somerset, Wiltshire, Avon and Gloucestershire (SWAG) Cancer Services. Cancer of Unknown Primary Network Site Specific Group. Clinical Guidelines
 Somerset, Wiltshire, Avon and Gloucestershire (SWAG) Cancer Services Cancer of Unknown Primary Network Site Specific Group Revision due: April 2019 Page 1 of 11 VERSION CONTROL THIS IS A CONTROLLED DOCUMENT.
Somerset, Wiltshire, Avon and Gloucestershire (SWAG) Cancer Services Cancer of Unknown Primary Network Site Specific Group Revision due: April 2019 Page 1 of 11 VERSION CONTROL THIS IS A CONTROLLED DOCUMENT.
London Cancer Urology Pathway Board
 Urology Pathway Board Date: 13/10/2016 Venue: Boardroom, UCLH @Westmoreland street, London, W1G 8PH Chair: John Hines, Pathway Director 1. Welcome, Apologies and minutes of last meeting Last meetings minutes
Urology Pathway Board Date: 13/10/2016 Venue: Boardroom, UCLH @Westmoreland street, London, W1G 8PH Chair: John Hines, Pathway Director 1. Welcome, Apologies and minutes of last meeting Last meetings minutes
Faster Cancer Treatment Indicators: Use cases
 Faster Cancer Treatment Indicators: Use cases 2014 Date: October 2014 Version: Owner: Status: v01 Ministry of Health Cancer Services Final Citation: Ministry of Health. 2014. Faster Cancer Treatment Indicators:
Faster Cancer Treatment Indicators: Use cases 2014 Date: October 2014 Version: Owner: Status: v01 Ministry of Health Cancer Services Final Citation: Ministry of Health. 2014. Faster Cancer Treatment Indicators:
Point-Counterpoint: Radiation & Bladder Cancer
 Radiation Plays a Major Role in Certain Stages of Bladder Cancer ~ David C. Beyer, MD Radiation Therapy; no role in management of bladder cancer Robert E. Donohue M.D. Denver VAMC University of Colorado
Radiation Plays a Major Role in Certain Stages of Bladder Cancer ~ David C. Beyer, MD Radiation Therapy; no role in management of bladder cancer Robert E. Donohue M.D. Denver VAMC University of Colorado
South East London Cancer Network GUIDELINES FOR THE MANAGEMENT OF UROLOGICAL CANCERS. June 2012
 South East London Cancer Network GUIDELINES FOR THE MANAGEMENT OF UROLOGICAL CANCERS June 2012 Review date: August 2013 1 SOUTH EAST LONDON CANCER NETWORK UROLOGY DIAGNOSTIC AND TREATMENT GUIDELINES Index
South East London Cancer Network GUIDELINES FOR THE MANAGEMENT OF UROLOGICAL CANCERS June 2012 Review date: August 2013 1 SOUTH EAST LONDON CANCER NETWORK UROLOGY DIAGNOSTIC AND TREATMENT GUIDELINES Index
Radical removal of the kidney (radical nephrectomy): procedure-specific information
 PATIENT INFORMATION Radical removal of the kidney (radical nephrectomy): procedure-specific information What is the evidence base for this information? This leaflet includes advice from consensus panels,
PATIENT INFORMATION Radical removal of the kidney (radical nephrectomy): procedure-specific information What is the evidence base for this information? This leaflet includes advice from consensus panels,
NMIBC. Piotr Jarzemski. Department of Urology Jan Biziel University Hospital Bydgoszcz, Poland
 NMIBC Piotr Jarzemski Department of Urology Jan Biziel University Hospital Bydgoszcz, Poland 71 year old male patient was admitted to the Department of Urology First TURBT - 2 months prior to the hospitalisation.
NMIBC Piotr Jarzemski Department of Urology Jan Biziel University Hospital Bydgoszcz, Poland 71 year old male patient was admitted to the Department of Urology First TURBT - 2 months prior to the hospitalisation.
Guideline for the referral to the Late Effects Multi Disciplinary Team
 Guideline for the referral to the Late Effects Multi Disciplinary Team Version History Version Date Summary of Change\Process 0.1 July 2010 Reviewed and approved by West Midlands Children s Cancer Network
Guideline for the referral to the Late Effects Multi Disciplinary Team Version History Version Date Summary of Change\Process 0.1 July 2010 Reviewed and approved by West Midlands Children s Cancer Network
North of Scotland Cancer Network Clinical Management Guideline for Endometrial Cancer
 THIS DOCUMENT North of Scotland Cancer Network Clinical Management Guideline for Endometrial Cancer Based on WOSCAN CMG with further extensive consultation within NOSCAN UNCONTROLLED WHEN PRINTED DOCUMENT
THIS DOCUMENT North of Scotland Cancer Network Clinical Management Guideline for Endometrial Cancer Based on WOSCAN CMG with further extensive consultation within NOSCAN UNCONTROLLED WHEN PRINTED DOCUMENT
Richard Watson, Chief Transformation Officer. Dr P Holloway, GP Clinical Lead for Cancer Lisa Parrish, Senior Transformation Lead
 GOVERNING BODY Agenda Item No. 08 Reference No. IESCCG 18-02 Date. 23 January 2018 Title Lead Chief Officer Author(s) Purpose Cancer Services Update Richard Watson, Chief Transformation Officer Dr P Holloway,
GOVERNING BODY Agenda Item No. 08 Reference No. IESCCG 18-02 Date. 23 January 2018 Title Lead Chief Officer Author(s) Purpose Cancer Services Update Richard Watson, Chief Transformation Officer Dr P Holloway,
National Optimal Lung Cancer Pathways. Dr Sadia Anwar Nottingham University Hospitals NHS Trust Clinical Lead for Lung Cancer
 National Optimal Lung Cancer Pathways Dr Sadia Anwar ttingham University Hospitals NHS Trust Clinical Lead for Lung Cancer Overview How NOLCP evolved How it relates to national guidance Pathways Implementation
National Optimal Lung Cancer Pathways Dr Sadia Anwar ttingham University Hospitals NHS Trust Clinical Lead for Lung Cancer Overview How NOLCP evolved How it relates to national guidance Pathways Implementation
The Urology One-Stop Clinic
 The Urology One-Stop Clinic Exceptional healthcare, personally delivered The aim of this leaflet is to answer any questions you may have about the Urology One-Stop Clinic. What is the Urology One-Stop
The Urology One-Stop Clinic Exceptional healthcare, personally delivered The aim of this leaflet is to answer any questions you may have about the Urology One-Stop Clinic. What is the Urology One-Stop
Bladder Cancer Canada November 21st, Bladder Cancer 2018: A brighter light at the end of the cystoscope
 Bladder Cancer Canada November 21st, 2018 Bladder Cancer 2018: A brighter light at the end of the cystoscope Chris Morash MD FRCSC Associate Professor, University of Ottawa Head, Urological Oncology Bladder
Bladder Cancer Canada November 21st, 2018 Bladder Cancer 2018: A brighter light at the end of the cystoscope Chris Morash MD FRCSC Associate Professor, University of Ottawa Head, Urological Oncology Bladder
A06/S(HSS)b Ex-vivo partial nephrectomy service (Adult)
 A06/S(HSS)b 2013/14 NHS STANDARD CONTRACT FOR EX-VIVO PARTIAL NEPHRECTOMY SERVICE (ADULT) PARTICULARS, SCHEDULE 2 THE SERVICES, A - SERVICE SPECIFICATION Service Specification No. Service Commissioner
A06/S(HSS)b 2013/14 NHS STANDARD CONTRACT FOR EX-VIVO PARTIAL NEPHRECTOMY SERVICE (ADULT) PARTICULARS, SCHEDULE 2 THE SERVICES, A - SERVICE SPECIFICATION Service Specification No. Service Commissioner
Upper GI Malignancies Imaging Guidelines for the Management of Gastric, Oesophageal & Pancreatic Cancers 2012
 Upper GI Malignancies Imaging Guidelines for the Management of Gastric, Oesophageal & Pancreatic Cancers 2012 Version Control This is a controlled document please destroy all previous versions on receipt
Upper GI Malignancies Imaging Guidelines for the Management of Gastric, Oesophageal & Pancreatic Cancers 2012 Version Control This is a controlled document please destroy all previous versions on receipt
Supra Network Sarcoma Advisory Group (SAG) Annual Report
 London and South East Sarcoma Network Supra Network Sarcoma Advisory Group (SAG) Annual Report 2011-2012 Hosted by Date: December 2012 Version: 2 Review Date: September 2013 London Cancer Integrated Cancer
London and South East Sarcoma Network Supra Network Sarcoma Advisory Group (SAG) Annual Report 2011-2012 Hosted by Date: December 2012 Version: 2 Review Date: September 2013 London Cancer Integrated Cancer
Pathway Gynaecology Cancer & Diagnostic Protocol for Inter Trust transfer
 NICaN Pathway Gynaecology Cancer & Diagnostic Protocol for Inter Trust transfer Timed schedules to enable the proactive management of the patient from point of receipt of referral to first definitive treatment
NICaN Pathway Gynaecology Cancer & Diagnostic Protocol for Inter Trust transfer Timed schedules to enable the proactive management of the patient from point of receipt of referral to first definitive treatment
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of electrically-stimulated intravesical chemotherapy for superficial bladder
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of electrically-stimulated intravesical chemotherapy for superficial bladder
Joseph H. Williams, MD Idaho Urologic Institute St. Alphonsus Regional Medical Center September 22, 2016
 BLADDER CANCER Joseph H. Williams, MD Idaho Urologic Institute St. Alphonsus Regional Medical Center September 22, 2016 BLADDER CANCER = UROTHELIAL CANCER Antiquated term is Transitional Cell Carcinoma
BLADDER CANCER Joseph H. Williams, MD Idaho Urologic Institute St. Alphonsus Regional Medical Center September 22, 2016 BLADDER CANCER = UROTHELIAL CANCER Antiquated term is Transitional Cell Carcinoma
