Oncofocus. Patient Test Report
|
|
|
- Miles Parks
- 5 years ago
- Views:
Transcription
1 Oncofocus Patient Test Report
2 Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 24 May of 14 ONC17 Surname Requesting Clinician Forename DOB Date requested Gender Histology # Female 02689/A1 Lung Tumour % Primary site Adenocarcinoma Tumour % Tumour subtype Soft Tissue (macrodissected) Tissue Type 20-25% - Comment: 237 genes were targeted using 2530 unique amplicons covering oncogenes, fusion genes, genes susceptible to copy number variation and tumour suppressors. Actionable genetic variants detected by Oncofocus are linked to 582 anti-cancer targeted therapies. The RNA extracted from this sample was of optimal quality. The DNA extracted form the sample was of borderline quality. The Oncofocus assay on which the sample was run met all assay specific quality metrics for detecting oncogenes, fusion genes and tumour suppressors, however, copy number variation analysis was not possible. The following actionable variants were detected: Variant Summary Sample Cancer Type: Non-Small Cell Lung Cancer In this cancer type In other cancer type In this cancer type and other cancer types Contraindicated Both for use and contraindicated No evidence Gene Variant EMA US-FDA ESMO US-NCCN Global Clinical Trials ERBB2 p.(ala775_gly776insyvma) c.2313_2324dup (also known as p.(e770_a771insayvm) c.2324_2325insatacgtgatggc) (2) (15) EMA: European Medicine Agency, US-FDA: United States-Food and Drug Administration, ESMO: European Society for Medical Oncology, US-NCCN: United States-National Comprehensive Cancer Network. Numbers in parentheses indicate the number of relevant therapies with evidence.hotspot variants with >10% alternate allele reads, and in >10 unique reads are classified as detected with an assay sensitivity and positive predictive value(ppv) of 92%. Copy number variants; amplifications of CN> 6 with the 5% confidence value of 4 after normalization and deletions with 95% CI 1 are classified as present when the tumour% >50% with a sensitivity of 80% and PPV 100%. Gene Fusions are reported when occurring in >20 counts and meeting the thresholds of assay specific internal RNA quality control with a sensitivity of 92% and PPV of 99%. Supplementary technical information is available upon request. DISCLAIMER: The data presented here is a result of the curation of published data sources, but may not be exhaustive. The data version is (003).
3 Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 24 May of 14 Relevant Therapy Summary In this cancer type In other cancer type In this cancer type and other cancer types Contraindicated Both for use and contraindicated No evidence ERBB2 p.(e770_a771insayvm) c.2324_2325insatacgtgatggc Relevant Therapy EMA US-FDA ESMO US-NCCN Global Clinical Trials* afatinib (II) trastuzumab ado-trastuzumab emtansine (II) afatinib + chemotherapy (II) AZD-8931, erlotinib + chemotherapy (II) erlotinib + chemotherapy (II) lapatinib (II) luminespib (II) neratinib (II) pertuzumab + trastuzumab (II) pyrotinib (II) AP32788 (I/II) pirotinib (I/II) selumetinib + vistusertib (I/II) everolimus + trastuzumab + letrozole (I) varlitinib + chemotherapy (I) * Most advanced phase (IV, III, II/III, II, I/II, I) is shown and multiple clinical trials may be available. See global clinical trials section in the pages to follow.
4 Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 24 May of 14 Current US-NCCN Information In this cancer type In other cancer type In this cancer type and other cancer types Contraindicated US-NCCN information is current as of For the most up-to-date information, search For NCCN International Adaptations & Translations, search ERBB2 p.(e770_a771insayvm) c.2324_2325insatacgtgatggc afatinib US-NCCN Recommendation category: 2B Population segment (Line of therapy): NSCLC (Not specified) Reference: NCCN Guidelines - NCCN-Non-Small Cell Lung Cancer [Version ] trastuzumab US-NCCN Recommendation category: 2B Population segment (Line of therapy): NSCLC (Not specified) Reference: NCCN Guidelines - NCCN-Non-Small Cell Lung Cancer [Version ]
5 Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 24 May of 14 Current Global Clinical Trials Information Global Clinical Trials information is current as of For the most up-to-date information regarding a particular trial, search by NCT ID or search local clinical trials authority website by local identifier listed in 'Other identifiers'. ERBB2 p.(e770_a771insayvm) c.2324_2325insatacgtgatggc No NCT ID - see other identifier(s) A Study of Trastuzumab Emtansine in Patients With HER2-Positive, Recurrent Metastatic Non-Small Cell Lung Cancer Variant class: ERBB2 exon 20 insertion Other identifiers: HER2-CS-2, TrialTroveID , UMIN Population segments: ALK, EGFR, Second line or greater/refractory/relapsed, Stage III, Stage IV Therapy: ado-trastuzumab emtansine Location: Japan NCT Afatinib in Pretreated Patients With Advanced NSCLC Harbouring HER2 Exon 20 Mutations Variant class: ERBB2 exon 20 mutation Other identifiers: , ETOP 7-14, ETOP NICHE, EudraCT Number: , NICHE, REec , TrialTroveID Population segments: Second line or greater/refractory/relapsed, Squamous Cell, Stage III, Stage IV Therapy: afatinib Locations: Germany, Netherlands, Spain, Switzerland NCT A Multi-center Phase II Study of AUY922 in Patients with Stage IV Non-small Cell Lung Cancer (NSCLC) With Driver Molecular Alterations Other Than Sensitive EGFR Mutation, Who have Progressed after one Line of Systemic Therapy Variant class: ERBB2 exon 20 mutation Other identifiers: , CAUY922ATW02T( MIPD), TrialTroveID Population segments: ALK, EGFR, KRAS, Second line or greater/refractory/relapsed, Stage IV Therapy: luminespib Location: Taiwan
6 Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 24 May of 14 ERBB2 p.(e770_a771insayvm) c.2324_2325insatacgtgatggc (continued) NCT An Open-label, Multicenter, Multinational, Phase II Study Exploring the Efficacy and Safety of Neratinib Therapy in Patients With Solid Tumors With Activating HER2, HER3 or EGFR Mutations or With EGFR Gene Amplification. Variant class: ERBB2 activating mutation Other identifiers: , , CTA733, EudraCT Number: , IRAS ID: , NCI , PUMA-NER-5201, REec , SUMMIT, SUMMIT basket, TrialTroveID Population segments: EGFR, Estrogen receptor positive, HER2 positive, Progesterone receptor positive, Second line or greater/refractory/relapsed, Stage IV Therapy: neratinib Locations: Australia, Finland, Israel, Italy, Republic of Korea, Spain, United Kingdom, United States US States: CA, FL, MA, MO, MS, NJ, NY, TN, TX US Contact: Puma Biotechnology Clinical Operations [ ; ClinicalTrials@pumabiotechnology.com] NCT A Phase II Study of Afatinib in Patients With Advanced NSCLC Harboring HER2 Mutations, Previously Treated With Chemotherapy Other identifiers: CTR , , TrialTroveID Population segments: Second line or greater/refractory/relapsed, Stage III, Stage IV Therapy: afatinib + chemotherapy Location: Malaysia NCT Single Arm Phase II Clinical Trial to Investigate the Efficacy and Safety of Pyrotinib as a Single Agent in HER2 Mutation Advanced Non-small Cell Lung Cancer Patients Who Failed to Previous at Least 2nd Line Treatments Other identifiers: FK1406, TrialTroveID Population segments: Second line or greater/refractory/relapsed, Stage III, Stage IV Therapy: pyrotinib Location: China
7 Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 24 May of 14 ERBB2 p.(e770_a771insayvm) c.2324_2325insatacgtgatggc (continued) NCT Intergroup Trial UNICANCER UC / IFCT 1301: SAFIR02_Lung - Evaluation of the Efficacy of High Throughput Genome Analysis as a Therapeutic Decision Tool for Patients With Metastatic Non-small Cell Lung Cancer Variant class: ERBB2 aberration Other identifiers: EudraCT Number: , IFCT-1301 SAFIR02 Lung, SAFIR02 Lung ITTC-1301, SAFIR02_Lung, TrialTroveID , UC / IFCT 1301 Population segments: Adenocarcinoma, First line, Large Cell, Maintenance/ Consolidation, Second line or greater/refractory/relapsed, Squamous Cell, Stage III, Stage IV Therapies: AZD-8931, erlotinib + chemotherapy Location: France NCT A Phase I/II Study Of The Safety, Pharmacokinetics, And Anti-Tumor Activity Of The Oral EGFR/HER2 Inhibitor AP32788 In Non-Small Cell Lung Cancer Variant class: ERBB2 exon 20 insertion Other identifiers: , AP , NCI , TrialTroveID Population segments: CNS mets, EGFR, Second line or greater/refractory/relapsed, Stage III, Stage IV Phase: I/II Therapy: AP32788 US States: CA, CO, MA, NY, TN, VA US Contact: Dr. Shuanglian Li [ ; Shuanglian.Li@ariad.com] NCT A Phase Ib/IIa Study of AZD2014 in Combination With Selumetinib in Patients With Advanced Cancers. Variant class: ERBB2 aberration Other identifiers: QM, EudraCT Number: , IRAS ID , Torcmek, TrialTroveID , UKCRN ID:18725 Population segments: EGFR, FGFR, HER2 negative, HER2 positive, KRAS, Second line or greater/refractory/relapsed, Squamous Cell, Stage III, Stage IV, Triple receptor negative Phase: I/II Therapy: selumetinib + vistusertib Location: United Kingdom
8 Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 24 May of 14 ERBB2 p.(e770_a771insayvm) c.2324_2325insatacgtgatggc (continued) NCT A Phase II Trial of Ado-Trastuzumab Emtansine for Patients With HER2 Amplified or Mutant Cancers Cancer type: Lung Cancer Variant class: ERBB2 activating mutation Other identifiers: , TrialTroveID Population segments: First line, Stage III, Stage IV Therapy: ado-trastuzumab emtansine US State: NY US Contact: Multiple contacts: See for complete list of contacts. NCT Molecular Analysis for Therapy Choice (MATCH) Variant class: ERBB2 activating mutation Other identifiers: , CTSU/EAY131, EAY131, EAY131-A, EAY131-B, EAY131-E, EAY131-F, EAY131-G, EAY131-H, EAY131-I, EAY131-MATCH, EAY131-N, EAY131-P, EAY131-Q, EAY131-R, EAY131-S1, EAY131-S2, EAY131-T, EAY131-U, EAY131-V, EAY131- X, ECOGEAY131-M, MATCH, NCI , NCI-MATCH, TrialTroveID Population segments: (N/A), Aggressive, ALK, Classical, EGFR, HER2 positive, Indolent, Nodular lymphocyte-predominant, Second line or greater/refractory/relapsed, Stage III, Stage IV Therapy: afatinib US States: AK, AL, AR, AZ, CA, CO, CT, DC, DE, FL, GA, HI, IA, ID, IL, IN, KS, KY, LA, MA, MD, ME, MI, MN, MO, MS, MT, NC, ND, NE, NH, NJ, NM, NV, NY, OH, OK, OR, PA, RI, SC, SD, TN, TX, UT, VA, WA, WI, WV, WY US Contact: Multiple contacts: See for complete list of contacts.
9 Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 24 May of 14 ERBB2 p.(e770_a771insayvm) c.2324_2325insatacgtgatggc (continued) NCT My Pathway: An Open Label Phase IIa Study Evaluating Trastuzumab/ Pertuzumab, Erlotinib, Vemurafenib/ Cobimetinib, and Vismodegib in Patients Who Have Advanced Solid Tumors With Mutations or Gene Expression Abnormalities Predictive of Response to One of These Agents Variant class: ERBB2 activating mutation Other identifiers: , , AAAN9701, J1480, ML28897, ML28897/PRO 02, ML28897PRO/02, My Pathway, NCI , TrialTroveID Population segments: Second line or greater/refractory/relapsed, Stage III, Stage IV Therapy: pertuzumab + trastuzumab US States: AR, AZ, CA, CO, FL, GA, IL, MD, MN, NC, ND, NY, OH, OK, OR, PA, SD, TN, TX, VA, WA US Contact: Hoffmann-La Roche, Study Director [ ; global.rochegenentechtrials@roche.com] NCT Phase II Clinical Trial Treating Relapsed/ Recurrent/Refractory Pediatric Solid Tumors With the Genomically-Targeted Agent Erlotinib in Combination With Temozolomide Other identifiers: , NCI , TrialTroveID Population segments: (N/A), Pediatric or Adolescent, Second line or greater/refractory/ Relapsed Therapy: erlotinib + chemotherapy US State: MO US Contact: Dr. Robert Hayashi [ ; Hayashi_R@kids.wustl.edu] NCT A Two-period, Multicenter, Randomized, Open-label, Phase II Study Evaluating the Clinical Benefit of a Maintenance Treatment Targeting Tumor Molecular Alterations in Patients With Progressive Locally-advanced or Metastatic Solid Tumors MOST: My own specific treatment Other identifiers: ET12-081, EudraCT number: , MOST, ProfiLER, TrialTroveID Population segments: Maintenance/Consolidation, Second line or greater/refractory/ Relapsed, Stage III, Stage IV Therapy: lapatinib Location: France
10 Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 24 May of 14 ERBB2 p.(e770_a771insayvm) c.2324_2325insatacgtgatggc (continued) NCT A Phase 1 Study of KBP-5209 in Patients With Advanced Solid Tumors Variant class: ERBB aberration Other identifiers: , 5209-CPK-1001, NCI , TrialTroveID Population segments: EGFR, Second line or greater/refractory/relapsed, Stage III, Stage IV Phase: I/II Therapy: pirotinib US States: IN, TX, UT US Contact: Matthew S. Hunt [ ; Matthew.hunt2@covance.com] NCT Combination Treatment With Everolimus, Letrozole and Trastuzumab in Hormone Receptor and HER2/Neu-positive Patients With Advanced Metastatic Breast Cancer and Other Solid Tumors: Evaluating Synergy and Overcoming Resistance Other identifiers: , NCI , TrialTroveID Population segments: First line, HER2 positive, Maintenance/Consolidation, Second line or greater/refractory/relapsed, Stage III, Stage IV Other inclusion criteria: ER positive and/or PR positive Phase: I Therapy: everolimus + trastuzumab + letrozole US State: TX US Contact: Dr. Filip Janku [ ] No NCT ID - see other identifier(s) Phase I Clinical Study With Advanced Solid Tumors KBP-5209 Treatment Other identifiers: 5209-CPK-1002, CTR , TrialTroveID Population segments: EGFR, Second line or greater/refractory/relapsed, Stage III, Stage IV Phase: I Therapy: pirotinib Location: China
11 Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 24 May of 14 ERBB2 p.(e770_a771insayvm) c.2324_2325insatacgtgatggc (continued) NCT A Two-part Phase I, Open Label, Dose Escalation Study to Evaluate the Safety, Tolerability and Pharmacokinetics of Pyrotinib in Patients Whose Disease Progressed on Prior HER2 Targeted Therapy Other identifiers: SHRUS 1001, TrialTroveID Population segments: HER2 positive, Second line or greater/refractory/relapsed, Stage III, Stage IV Phase: I Therapy: pyrotinib US State: TX US Contact: Multiple contacts: See for complete list of contacts. NCT Phase I Study to Evaluate the Safety and Tolerability of ASLAN001 in Combination with Oxaliplatin and Capecitabine or Oxaliplatin and 5-FU with Leucovorin Variant class: ERBB2 aberration Other identifiers: ASLAN SG, TrialTroveID Population segments: Second line or greater/refractory/relapsed, Stage IV Exclusion criteria variant class: EGFR T790M mutation Phase: I Therapy: varlitinib + chemotherapy Location: Singapore
12 Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 24 May of 14 Appendix: Evidence Summary by Variant Class A variant class hierarchy was created to summarize gene variants with associated clinical evidence. Evidence items refers to citations across the different global data sources. ERBB2 p.(e770_a771insayvm) c.2324_2325insatacgtgatggc Variant Class Evidence Items ERBB aberration 1 ERBB2 aberration 3 ERBB2 mutation 9 ERBB2 activating mutation 4 ERBB2 exon 20 insertion 2 ERBB2 exon 20 mutation 2 ERBB2 exon 20 insertion 2 Appendix: Variant Details DNA Sequence Variants Gene Amino Acid Change Coding Variant ID ERBB2 p.(e770_a771insayv M) c.2324_2325insatac GTGATGGC Allele Frequency Transcript Variant Effect Gene Class Variant Class COSM % NM_ nonframeshift Insertion Gain of Function Hotspot
13
Oncofocus. Patient Test Report
 Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 24 May 2017 1 of 14 ONC17 Surname Requesting Clinician Forename DOB Date requested Gender Histology
Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 24 May 2017 1 of 14 ONC17 Surname Requesting Clinician Forename DOB Date requested Gender Histology
Oncofocus. Patient Test Report
 Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson BMS: Tiffany Haddow Date: 14 Dec 2016 1 of 18 Comment: The DNA and RNA extracted from this sample were of optimal quality. The Oncofocus
Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson BMS: Tiffany Haddow Date: 14 Dec 2016 1 of 18 Comment: The DNA and RNA extracted from this sample were of optimal quality. The Oncofocus
Oncofocus. Patient Test Report
 Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 06 Jun 2017 1 of 13 ONC17 Surname Requesting Clinician Forename DOB Date requested Gender Female Histology
Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 06 Jun 2017 1 of 13 ONC17 Surname Requesting Clinician Forename DOB Date requested Gender Female Histology
Oncofocus. Patient Test Report
 Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson BMS: Tiffany Haddow Date: 14 Dec 2016 1 of 18 Comment: The DNA and RNA extracted from this sample were of optimal quality. The Oncofocus
Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson BMS: Tiffany Haddow Date: 14 Dec 2016 1 of 18 Comment: The DNA and RNA extracted from this sample were of optimal quality. The Oncofocus
Oncofocus. Patient Test Report
 Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson BMS: Tiffany Haddow Date: 22 Dec 2016 1 of 4 Comment: The DNA and RNA extracted from this sample were of optimal quality. The Oncofocus
Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson BMS: Tiffany Haddow Date: 22 Dec 2016 1 of 4 Comment: The DNA and RNA extracted from this sample were of optimal quality. The Oncofocus
Oncofocus. Patient Test Report
 Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 29 Mar 2017 1 of 8 Surname Forename DOB Gender Histology # Primary site Tumour subtype Tissue Type
Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 29 Mar 2017 1 of 8 Surname Forename DOB Gender Histology # Primary site Tumour subtype Tissue Type
Oncofocus. Patient Test Report
 Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 08 Feb 2017 1 of 6 Comment: The DNA and RNA extracted from this sample were of optimal quantity. The
Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 08 Feb 2017 1 of 6 Comment: The DNA and RNA extracted from this sample were of optimal quantity. The
Oncofocus. Patient Test Report
 Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 04 Jan 2018 1 of 25 ONC17 Surname Requester Forename Contact details DOB Date requested Gender Histology
Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 04 Jan 2018 1 of 25 ONC17 Surname Requester Forename Contact details DOB Date requested Gender Histology
Oncofocus. Patient Test Report
 Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 04 May 2017 1 of 13 Surname Forename DOB Gender Histology # Primary site Tumour subtype Tissue Type
Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 04 May 2017 1 of 13 Surname Forename DOB Gender Histology # Primary site Tumour subtype Tissue Type
Oncofocus. Patient Test Report
 Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 06 Apr 2017 1 of 16 Surname Forename DOB Gender Histology # Primary site Tumour subtype Tissue Type
Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 06 Apr 2017 1 of 16 Surname Forename DOB Gender Histology # Primary site Tumour subtype Tissue Type
Oncofocus. Patient Test Report
 Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Katherine Marquis Date: 05 Oct 2016 1 of 8 The DNA and RNA extracted from this sample were of optimal quality. The Oncofocus
Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Katherine Marquis Date: 05 Oct 2016 1 of 8 The DNA and RNA extracted from this sample were of optimal quality. The Oncofocus
Oncofocus. Patient Test Report
 Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson BMS: Tiffany Haddow Date: 30 Nov 2016 1 of 11 Comment: The DNA and RNA extracted from this sample were of optimal quality. The Oncofocus
Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson BMS: Tiffany Haddow Date: 30 Nov 2016 1 of 11 Comment: The DNA and RNA extracted from this sample were of optimal quality. The Oncofocus
Oncofocus. Patient Test Report
 Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 04 May 2017 1 of 28 Surname Forename DOB Gender Histology # Primary site Tumour subtype Tissue Type
Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 04 May 2017 1 of 28 Surname Forename DOB Gender Histology # Primary site Tumour subtype Tissue Type
Oncofocus. Patient Test Report
 Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 04 May 2017 1 of 28 Surname Forename DOB Gender Histology # Primary site Tumour subtype Tissue Type
Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 04 May 2017 1 of 28 Surname Forename DOB Gender Histology # Primary site Tumour subtype Tissue Type
Oncofocus. Patient Test Report
 Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 04 May 2017 1 of 13 Surname Forename DOB Gender Histology # Primary site Tumour subtype Tissue Type
Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 04 May 2017 1 of 13 Surname Forename DOB Gender Histology # Primary site Tumour subtype Tissue Type
Oncofocus. Patient Test Report
 Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 18 Jan 2017 1 of 25 Comment: The DNA and RNA extracted from this sample were of optimal quality. The
Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 18 Jan 2017 1 of 25 Comment: The DNA and RNA extracted from this sample were of optimal quality. The
Oncofocus. Patient Test Report
 Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 21 Apr 2017 1 of 25 ONC17 Surname Requesting Clinician Forename DOB Date requested Gender Histology
Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Tiffany Haddow Date: 21 Apr 2017 1 of 25 ONC17 Surname Requesting Clinician Forename DOB Date requested Gender Histology
Oncofocus. Patient Test Report
 Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Keeley Monsen Date: 26 May 2017 1 of 26 ONC17 Surname Requesting Clinician Forename DOB Date requested Gender Male Histology
Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Keeley Monsen Date: 26 May 2017 1 of 26 ONC17 Surname Requesting Clinician Forename DOB Date requested Gender Male Histology
Oncofocus. Patient Test Report
 Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Katherine Marquis Oncologica UK Ltd Date: 27 Jun 2016 1 of 36 Comment: The DNA and RNA extracted from this sample were of
Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Katherine Marquis Oncologica UK Ltd Date: 27 Jun 2016 1 of 36 Comment: The DNA and RNA extracted from this sample were of
Oncofocus. Patient Test Report
 Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Katherine Marquis Oncologica UK Ltd Date: 05 Aug 2016 1 of 18 Comment: The DNA and RNA extracted from this sample were of
Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson Senior BMS: Katherine Marquis Oncologica UK Ltd Date: 05 Aug 2016 1 of 18 Comment: The DNA and RNA extracted from this sample were of
Federation of State Boards of Physical Therapy Jurisdiction Licensure Reference Guide Topic: Retaking NPTE
 The table below lists the requirements for retaking the National Physical Therapy Exam (NPTE) for each jurisdiction. Summary Number of attempts on NPTE limited? 16 27 Number of attempts allowed before
The table below lists the requirements for retaking the National Physical Therapy Exam (NPTE) for each jurisdiction. Summary Number of attempts on NPTE limited? 16 27 Number of attempts allowed before
Workforce Data The American Board of Pediatrics
 Workforce Data 2009-2010 The American Board of Pediatrics Caution. Before using this report as a resource, please read the information below! Please use caution when comparing data in this version of the
Workforce Data 2009-2010 The American Board of Pediatrics Caution. Before using this report as a resource, please read the information below! Please use caution when comparing data in this version of the
Medical Laboratory Accredited to ISO15189:2012. Oncofocus. Precision Oncology ONCOFOCUS TEST REPORT
 Medical Laboratory Accredited to ISO15189:2012 Oncofocus Precision Oncology ONCOFOCUS TEST REPORT Lead Clinical Scientist: - Pre-Reg Clinical Scientist: - Date: 1 of 15 Surname Forename DOB Gender Histology
Medical Laboratory Accredited to ISO15189:2012 Oncofocus Precision Oncology ONCOFOCUS TEST REPORT Lead Clinical Scientist: - Pre-Reg Clinical Scientist: - Date: 1 of 15 Surname Forename DOB Gender Histology
Oncofocus. Patient Test Report
 Oncofocus Patient Test Report Oncofocus Patient Test Report Oncologica UK Ltd, Suite 15-16, The Science Village, Chesterford Research Park, Cambridge, CB10 1XL www.oncologica.com - Tel: +44 (0)1223 785327
Oncofocus Patient Test Report Oncofocus Patient Test Report Oncologica UK Ltd, Suite 15-16, The Science Village, Chesterford Research Park, Cambridge, CB10 1XL www.oncologica.com - Tel: +44 (0)1223 785327
Oncofocus. Patient Test Report
 Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson BMS: Tiffany Haddow Date: 18 Jan 2017 1 of 30 Comment: The DNA and RNA extracted from this sample were of optimal quality. The Oncofocus
Oncofocus Patient Test Report Lead Clinical Scientist: Keeda Snelson BMS: Tiffany Haddow Date: 18 Jan 2017 1 of 30 Comment: The DNA and RNA extracted from this sample were of optimal quality. The Oncofocus
Federation of State Boards of Physical Therapy Jurisdiction Licensure Reference Guide Topic: Direct Access
 Each licensing authority indicates the level of direct access allowed in the jurisdiction and the type of limitations that apply to this access. There are two tables: Types and Limits Referrals TYPES AND
Each licensing authority indicates the level of direct access allowed in the jurisdiction and the type of limitations that apply to this access. There are two tables: Types and Limits Referrals TYPES AND
Financial Impact of Lung Cancer in West Virginia
 Financial Impact of Lung Cancer in West Virginia John Deskins, Ph.D. Christiadi, Ph.D. Sara Harper November 2018 Bureau of Business & Economic Research College of Business & Economics West Virginia University
Financial Impact of Lung Cancer in West Virginia John Deskins, Ph.D. Christiadi, Ph.D. Sara Harper November 2018 Bureau of Business & Economic Research College of Business & Economics West Virginia University
Federation of State Boards of Physical Therapy Jurisdiction Licensure Reference Guide Topic: Direct Access
 Each licensing authority indicates the level of direct access allowed in the jurisdiction and the type of limitations that apply to this access. There are two tables: Types and Limits Referrals TYPES AND
Each licensing authority indicates the level of direct access allowed in the jurisdiction and the type of limitations that apply to this access. There are two tables: Types and Limits Referrals TYPES AND
RECOVERY SUPPORT SERVICES IN STATES
 RECOVERY SUPPORT SERVICES IN STATES An analysis of State recovery support services using the 16 17 Substance Abuse Block Grant (SABG) Behavioral Health Assessment and Plan THIS PROJECT IS BEING SUPPORTED
RECOVERY SUPPORT SERVICES IN STATES An analysis of State recovery support services using the 16 17 Substance Abuse Block Grant (SABG) Behavioral Health Assessment and Plan THIS PROJECT IS BEING SUPPORTED
Personalized Gene Profile
 Sequence Variants: 6 4 ALTERATION MUTANT FRACTION FDA GUIDANCE (for indication) FDA GUIDANCE (for other indications) TRIALS (details below) c.38g>a; p.g13d 21.0% Cetuximab Contraindicated 22 Panitumumab
Sequence Variants: 6 4 ALTERATION MUTANT FRACTION FDA GUIDANCE (for indication) FDA GUIDANCE (for other indications) TRIALS (details below) c.38g>a; p.g13d 21.0% Cetuximab Contraindicated 22 Panitumumab
Oncofocus. Patient Test Report
 Oncofocus Patient Test Report Senior BMS: Katherine Marquis Date: 04 Apr 2016 17:01:50 PM 1 of 18 Comment: The DNA and RNA extracted from this sample were of optimal quality. The Oncofocus assay on which
Oncofocus Patient Test Report Senior BMS: Katherine Marquis Date: 04 Apr 2016 17:01:50 PM 1 of 18 Comment: The DNA and RNA extracted from this sample were of optimal quality. The Oncofocus assay on which
2016 COMMUNITY SURVEY
 1 Epilepsy Innovation Institute (Ei ) 016 COMMUNITY SURVEY INTRODUCTION From September 8th to November 9th, 016, epilepsy.com hosted a survey that asked the community the following: What are the aspects
1 Epilepsy Innovation Institute (Ei ) 016 COMMUNITY SURVEY INTRODUCTION From September 8th to November 9th, 016, epilepsy.com hosted a survey that asked the community the following: What are the aspects
Federation of State Boards of Physical Therapy Jurisdiction Licensure Reference Guide Topic: Foreign Educated Physical Therapists
 Requirements for Licensure Summary: Number of Jurisdictions that Require: Educational Credentials Review 50 Graduation from a program equivalent to CAPTE 37 Eligibility to practice in the country in which
Requirements for Licensure Summary: Number of Jurisdictions that Require: Educational Credentials Review 50 Graduation from a program equivalent to CAPTE 37 Eligibility to practice in the country in which
Evidence-Based Policymaking: Investing in Programs that Work
 Evidence-Based Policymaking: Investing in Programs that Work August 4, 2015 The Policy Challenge Though policymakers want to make the best choices, the process often relies on inertia and anecdote Very
Evidence-Based Policymaking: Investing in Programs that Work August 4, 2015 The Policy Challenge Though policymakers want to make the best choices, the process often relies on inertia and anecdote Very
How to Get Paid for Doing EBD
 How to Get Paid for Doing EBD Robert D. Compton, DDS President Robert Compton, DDS Executive Director DentaQuest Institute Disclosure DentaQuest Institute President DentaQuest Benefits Senior VP & CDO
How to Get Paid for Doing EBD Robert D. Compton, DDS President Robert Compton, DDS Executive Director DentaQuest Institute Disclosure DentaQuest Institute President DentaQuest Benefits Senior VP & CDO
State of California Department of Justice. Bureau of Narcotic Enforcement
 State of California Department of Justice Bureau of Narcotic Enforcement Prescription Drugs in the U.S. At least half of all Americans take one prescription drug regularly, with one in six taking three
State of California Department of Justice Bureau of Narcotic Enforcement Prescription Drugs in the U.S. At least half of all Americans take one prescription drug regularly, with one in six taking three
Different Types of Cancer
 Different Types of Cancer Cancer can originate almost anywhere in the body. Sarcomas (connective tissue) Ø arise from cells found in the supporting tissues of the body such as bone, cartilage, fat, connective
Different Types of Cancer Cancer can originate almost anywhere in the body. Sarcomas (connective tissue) Ø arise from cells found in the supporting tissues of the body such as bone, cartilage, fat, connective
Federation of State Boards of Physical Therapy Jurisdiction Licensure Reference Guide Topic: Direct Access
 Each licensing authority indicates the level of direct access allowed in the jurisdiction and the type of limitations that apply to this access. There are three tables: Types and Limits Specific Limits
Each licensing authority indicates the level of direct access allowed in the jurisdiction and the type of limitations that apply to this access. There are three tables: Types and Limits Specific Limits
Federation of State Boards of Physical Therapy Jurisdiction Licensure Reference Guide Topic: Foreign Educated PTs and PTAs
 PT Requirements for Licensure Summary: Number of Jurisdictions that Require: Educational Credentials Review 50 from a program equivalent to CAPTE 37 Eligibility to practice in the country in which education
PT Requirements for Licensure Summary: Number of Jurisdictions that Require: Educational Credentials Review 50 from a program equivalent to CAPTE 37 Eligibility to practice in the country in which education
Tecentriq (atezolizumab) (Intravenous)
 Tecentriq (atezolizumab) (Intravenous) Last Review Date: 06/01/2018 Date of Origin: 06/28/2016 Document Number: IC-0278 Dates Reviewed: 06/2016, 08/2016, 10/2016, 02/2017, 04/2017, 08/2017, 11/2017, 02/2018,
Tecentriq (atezolizumab) (Intravenous) Last Review Date: 06/01/2018 Date of Origin: 06/28/2016 Document Number: IC-0278 Dates Reviewed: 06/2016, 08/2016, 10/2016, 02/2017, 04/2017, 08/2017, 11/2017, 02/2018,
BY-STATE MENTAL HEALTH SERVICES AND EXPENDITURES IN MEDICAID, 1999
 STATE-BY BY-STATE MENTAL HEALTH SERVICES AND EXPENDITURES IN MEDICAID, 1999 James Verdier,, Ann Cherlow,, and Allison Barrett Mathematica Policy Research, Inc. Jeffrey Buck and Judith Teich Substance Abuse
STATE-BY BY-STATE MENTAL HEALTH SERVICES AND EXPENDITURES IN MEDICAID, 1999 James Verdier,, Ann Cherlow,, and Allison Barrett Mathematica Policy Research, Inc. Jeffrey Buck and Judith Teich Substance Abuse
Dates Reviewed: 12/2012, 3/2013, 6/2013, 9/2013, 11/2013, 12/2013, 3/2014, 6/2014, 9/2014, 12/2014,
 Perjeta (pertuzumab) Last Review Date: 5/30/2017 Date of Origin: 11/01/2012 Document Number: IC-0096 Dates Reviewed: 12/2012, 3/2013, 6/2013, 9/2013, 11/2013, 12/2013, 3/2014, 6/2014, 9/2014, 12/2014,
Perjeta (pertuzumab) Last Review Date: 5/30/2017 Date of Origin: 11/01/2012 Document Number: IC-0096 Dates Reviewed: 12/2012, 3/2013, 6/2013, 9/2013, 11/2013, 12/2013, 3/2014, 6/2014, 9/2014, 12/2014,
USA National Mental Healthcare Nonprofit Exempt Organization Financial Analysis as of December 14, 2015 January 24, 2016 ANSA-H2
 USA National Mental Healthcare Nonprofit Exempt Organization Financial Analysis as of December 14, 2015 January 24, 2016 ANSA-H2 Prepared by David Yoo, HanaSoul Consulting, Omaha, Nebraska dcyoo@cox.net
USA National Mental Healthcare Nonprofit Exempt Organization Financial Analysis as of December 14, 2015 January 24, 2016 ANSA-H2 Prepared by David Yoo, HanaSoul Consulting, Omaha, Nebraska dcyoo@cox.net
Overview of the HHS National Network of Quitlines Initiative
 Overview of the HHS National Network of Quitlines Initiative Prepared for the 2005 National Oral Health Conference 6th Joint Meeting of ASTDD and AAPHD Barbara Z. Park, RDH, MPH May 2, 2005 Background
Overview of the HHS National Network of Quitlines Initiative Prepared for the 2005 National Oral Health Conference 6th Joint Meeting of ASTDD and AAPHD Barbara Z. Park, RDH, MPH May 2, 2005 Background
Using Cancer Registry Data to Estimate the Percentage of Melanomas Attributable to UV Exposure
 Using Cancer Registry Data to Estimate the Percentage of Melanomas Attributable to UV Exposure Meg Watson, MPH Epidemiologist NAACCR Annual Conference June 16, 2016 National Center for Chronic Disease
Using Cancer Registry Data to Estimate the Percentage of Melanomas Attributable to UV Exposure Meg Watson, MPH Epidemiologist NAACCR Annual Conference June 16, 2016 National Center for Chronic Disease
Overview and Findings from ASTHO s IIS Interstate Data Sharing Meeting
 Overview and Findings from ASTHO s IIS Interstate Data Sharing Meeting Kim Martin Association of State and Territorial Health Officials (ASTHO) May 12, 2015 The Need for IIS Interstate Data Exchange States,
Overview and Findings from ASTHO s IIS Interstate Data Sharing Meeting Kim Martin Association of State and Territorial Health Officials (ASTHO) May 12, 2015 The Need for IIS Interstate Data Exchange States,
THREE BIG IMPACT ISSUES
 THREE BIG IMPACT ISSUES Tim McAfee, MD, MPH Director CDC Office on Smoking and Health Presented at the National Cancer Policy Forum Workshop on Reducing Tobacco-Related Cancer Incidence and Mortality June
THREE BIG IMPACT ISSUES Tim McAfee, MD, MPH Director CDC Office on Smoking and Health Presented at the National Cancer Policy Forum Workshop on Reducing Tobacco-Related Cancer Incidence and Mortality June
Lung Cancer Genetics: Common Mutations and How to Treat Them David J. Kwiatkowski, MD, PhD. Mount Carrigain 2/4/17
 Lung Cancer Genetics: Common Mutations and How to Treat Them David J. Kwiatkowski, MD, PhD Mount Carrigain 2/4/17 Histology Adenocarcinoma: Mixed subtype, acinar, papillary, solid, micropapillary, lepidic
Lung Cancer Genetics: Common Mutations and How to Treat Them David J. Kwiatkowski, MD, PhD Mount Carrigain 2/4/17 Histology Adenocarcinoma: Mixed subtype, acinar, papillary, solid, micropapillary, lepidic
LaTanya Runnells, Ph.D Program Manager December 6, 2016
 LaTanya Runnells, Ph.D Program Manager December 6, 2016 Mental Health First Aid USA is coordinated by the National Council for Behavioral Health, the Maryland Department of Health and Mental Hygiene, and
LaTanya Runnells, Ph.D Program Manager December 6, 2016 Mental Health First Aid USA is coordinated by the National Council for Behavioral Health, the Maryland Department of Health and Mental Hygiene, and
Federation of State Boards of Physical Therapy Jurisdiction Licensure Reference Guide Topic: License Renewal. License Renewal on Birthdays
 The table below lists information on the term and renewal date for each jurisdiction. Summary License Term 1 26 2 Renewal Date One date 31 Birthdays 6 Half in even years 4 4 License Term AL AK AZ AR CA
The table below lists information on the term and renewal date for each jurisdiction. Summary License Term 1 26 2 Renewal Date One date 31 Birthdays 6 Half in even years 4 4 License Term AL AK AZ AR CA
ANNUAL REPORT EXECUTIVE SUMMARY. The full report is available at DECEMBER 2017
 ANNUAL REPORT EXECUTIVE SUMMARY DECEMBER 2017 The full report is available at www.americashealthrankings.org EXECUTIVE SUMMARY OVERVIEW America s Health Rankings presents its 28th Annual Report, providing
ANNUAL REPORT EXECUTIVE SUMMARY DECEMBER 2017 The full report is available at www.americashealthrankings.org EXECUTIVE SUMMARY OVERVIEW America s Health Rankings presents its 28th Annual Report, providing
What is the Objective of the DQA in Developing Performance Measures. Robert Compton, DDS Executive Director
 What is the Objective of the DQA in Developing Performance Measures Robert Compton, DDS Executive Director EBD Champions Conference May 9-10, 2014 DISCLOSURE Disclosure on DentaQuest Benefits ~ 20 million
What is the Objective of the DQA in Developing Performance Measures Robert Compton, DDS Executive Director EBD Champions Conference May 9-10, 2014 DISCLOSURE Disclosure on DentaQuest Benefits ~ 20 million
Dental ER Visits: Evidence of a Failed System. Shelly Gehshan AACDP Conference April 29, 2012
 Dental ER Visits: Evidence of a Failed System Shelly Gehshan AACDP Conference April 29, 2012 Overview of Pew s s findings Preventable dental conditions were the primary diagnosis in 830,590 visits to hospital
Dental ER Visits: Evidence of a Failed System Shelly Gehshan AACDP Conference April 29, 2012 Overview of Pew s s findings Preventable dental conditions were the primary diagnosis in 830,590 visits to hospital
Improving Cancer Surveillance and Mortality Data for AI/AN Populations
 Improving Cancer Surveillance and Mortality Data for AI/AN Populations Melissa A. Jim, MPH (Diné) Epidemiologist, Cancer Surveillance Branch Assigned to IHS Division of Epidemiology and Disease Prevention
Improving Cancer Surveillance and Mortality Data for AI/AN Populations Melissa A. Jim, MPH (Diné) Epidemiologist, Cancer Surveillance Branch Assigned to IHS Division of Epidemiology and Disease Prevention
State Tobacco Control Programs
 State Tobacco Control Programs National Cancer Policy Forum Workshop Reducing Tobacco-Related Cancer Incidence and Mortality Karla S. Sneegas, MPH Chief Program Services Branch CDC Office on Smoking and
State Tobacco Control Programs National Cancer Policy Forum Workshop Reducing Tobacco-Related Cancer Incidence and Mortality Karla S. Sneegas, MPH Chief Program Services Branch CDC Office on Smoking and
Arkansas Prescription Monitoring Program
 Arkansas Prescription Monitoring Program FY 2016 Third Quarter Report January-March 2016 Arkansas Prescription Monitoring Program Quarterly Report January March, Fiscal year 2016 Act 304 of 2011 authorized
Arkansas Prescription Monitoring Program FY 2016 Third Quarter Report January-March 2016 Arkansas Prescription Monitoring Program Quarterly Report January March, Fiscal year 2016 Act 304 of 2011 authorized
Cyramza (ramucirumab) (Intravenous)
 Cyramza (ramucirumab) (Intravenous) Document Number: IC 0199 Last Review Date: 5/1/2018 Date of Origin: 06/24/2014 Dates Reviewed: 09/2014, 01/2015, 05/2015, 11/2015, 04/2016, 08/2016, 11/2016, 05/2017,
Cyramza (ramucirumab) (Intravenous) Document Number: IC 0199 Last Review Date: 5/1/2018 Date of Origin: 06/24/2014 Dates Reviewed: 09/2014, 01/2015, 05/2015, 11/2015, 04/2016, 08/2016, 11/2016, 05/2017,
Improving Oral Health:
 Improving Oral Health: New Tools for State Policy Makers Cassie Yarbrough, MPP Lead Public Policy Analyst Health Policy Institute Lansing, MI June 1, 2017 The ADA Health Policy Institute 2017 American
Improving Oral Health: New Tools for State Policy Makers Cassie Yarbrough, MPP Lead Public Policy Analyst Health Policy Institute Lansing, MI June 1, 2017 The ADA Health Policy Institute 2017 American
Christie Eheman, PhD NAACCR 6/2012
 Christie Eheman, PhD NAACCR 6/2012 Comparative Effectiveness Research CDC National Program of Cancer Registries Christie Eheman, PhD NAACCR 6/2012 CDC - Fran Michaud, Dave Butterworth, Linda Mulvihill,
Christie Eheman, PhD NAACCR 6/2012 Comparative Effectiveness Research CDC National Program of Cancer Registries Christie Eheman, PhD NAACCR 6/2012 CDC - Fran Michaud, Dave Butterworth, Linda Mulvihill,
Use of molecular surveillance data to identify clusters of recent and rapid HIV transmission
 National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention Use of molecular surveillance data to identify clusters of recent and rapid HIV transmission Alexa Oster, MD Acting Lead, Incidence
National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention Use of molecular surveillance data to identify clusters of recent and rapid HIV transmission Alexa Oster, MD Acting Lead, Incidence
Arkansas Prescription Monitoring Program
 Arkansas Prescription Monitoring Program FY 2017 Second Quarter Report October December 2016 Arkansas Prescription Monitoring Program Quarterly Report October December, Fiscal year 2017 Act 304 of 2011
Arkansas Prescription Monitoring Program FY 2017 Second Quarter Report October December 2016 Arkansas Prescription Monitoring Program Quarterly Report October December, Fiscal year 2017 Act 304 of 2011
Imfinzi (durvalumab) (Intravenous)
 Imfinzi (durvalumab) (Intravenous) Last Review Date: 09/05/2018 Date of Origin: 05/30/2017 Dates Reviewed: 05/2017, 08/2017, 11/2017, 02/2018, 05/2018, 09/2018 Document Number: IC-0301 I. Length of Authorization
Imfinzi (durvalumab) (Intravenous) Last Review Date: 09/05/2018 Date of Origin: 05/30/2017 Dates Reviewed: 05/2017, 08/2017, 11/2017, 02/2018, 05/2018, 09/2018 Document Number: IC-0301 I. Length of Authorization
Consensus and Collaboration
 Consensus and Collaboration John Morton, MD, MPH, FACS, FASMBS Chief, Bariatric & Minimally Invasive Surgery Stanford School of Medicine Past-President, American Society of Metabolic and Bariatric Surgery,
Consensus and Collaboration John Morton, MD, MPH, FACS, FASMBS Chief, Bariatric & Minimally Invasive Surgery Stanford School of Medicine Past-President, American Society of Metabolic and Bariatric Surgery,
A National and Statewide Perspective on the Opioid Crisis
 Insert CIO Name Here A National and Statewide Perspective on the Opioid Crisis Amy Peeples, MPA Deputy Director, National Center for Injury Prevention and Control Centers for Disease Control and Prevention
Insert CIO Name Here A National and Statewide Perspective on the Opioid Crisis Amy Peeples, MPA Deputy Director, National Center for Injury Prevention and Control Centers for Disease Control and Prevention
The Affordable Care Act and HIV: What are the Implications?
 The Affordable Care Act and HIV: What are the Implications? 2013 National Black AIDS Institute Webinar Series September 18, 2013 Jen Kates, Kaiser Family Foundation The Challenge Figure 1 30 years into
The Affordable Care Act and HIV: What are the Implications? 2013 National Black AIDS Institute Webinar Series September 18, 2013 Jen Kates, Kaiser Family Foundation The Challenge Figure 1 30 years into
Strategies to Increase Hepatitis C Treatment Within ADAPs
 Strategies to Increase Hepatitis C Treatment Within ADAPs Amanda Bowes National Alliance of State & Territorial AIDS Directors (NASTAD) Disclosures Presenter(s) has no financial interest to disclose. Learning
Strategies to Increase Hepatitis C Treatment Within ADAPs Amanda Bowes National Alliance of State & Territorial AIDS Directors (NASTAD) Disclosures Presenter(s) has no financial interest to disclose. Learning
Expedited Partner Therapy (EPT)/Patient Deliver Partner Therapy (PDPT)
 Expedited Partner Therapy (EPT)/Patient Deliver Partner Therapy (PDPT) David Johnson STD Disparities Coordinator, Office of Health Equity Division of STD Prevention National Center for HIV, Viral Hepatitis,
Expedited Partner Therapy (EPT)/Patient Deliver Partner Therapy (PDPT) David Johnson STD Disparities Coordinator, Office of Health Equity Division of STD Prevention National Center for HIV, Viral Hepatitis,
Considerations for State Obesity Policy
 Considerations for State Obesity Policy Scott Kahan, MD, MPH Faculty, Johns Hopkins Bloomberg School of Public Health Director, National Center for Weight & Wellness Clinical Director, STOP Obesity Alliance,
Considerations for State Obesity Policy Scott Kahan, MD, MPH Faculty, Johns Hopkins Bloomberg School of Public Health Director, National Center for Weight & Wellness Clinical Director, STOP Obesity Alliance,
Marijuana and driving in the United States: prevalence, risks, and laws
 Marijuana and driving in the United States: prevalence, risks, and laws Lifesavers National Conference 2016 Long Beach, California April 4, 2016 Anne T. McCartt iihs.org IIHS is an independent, nonprofit
Marijuana and driving in the United States: prevalence, risks, and laws Lifesavers National Conference 2016 Long Beach, California April 4, 2016 Anne T. McCartt iihs.org IIHS is an independent, nonprofit
Report to Congressional Defense Committees
 Report to Congressional Defense Committees The Department of Defense Comprehensive Autism Care Demonstration Quarterly Report to Congress Second Quarter, Fiscal Year 2017 In Response to: Senate Report
Report to Congressional Defense Committees The Department of Defense Comprehensive Autism Care Demonstration Quarterly Report to Congress Second Quarter, Fiscal Year 2017 In Response to: Senate Report
7/6/2015. Cancer Related Deaths: United States. Management of NSCLC TODAY. Emerging mutations as predictive biomarkers in lung cancer: Overview
 Emerging mutations as predictive biomarkers in lung cancer: Overview Kirtee Raparia, MD Assistant Professor of Pathology Cancer Related Deaths: United States Men Lung and bronchus 28% Prostate 10% Colon
Emerging mutations as predictive biomarkers in lung cancer: Overview Kirtee Raparia, MD Assistant Professor of Pathology Cancer Related Deaths: United States Men Lung and bronchus 28% Prostate 10% Colon
Approach to Cancer Prevention through Policy, Systems, and Environmental Change in the U.S.
 Approach to Cancer Prevention through Policy, Systems, and Environmental Change in the U.S. Marcus Plescia, MD, MPH Director, Division of Cancer Prevention and Control Centers for Disease Control and Prevention
Approach to Cancer Prevention through Policy, Systems, and Environmental Change in the U.S. Marcus Plescia, MD, MPH Director, Division of Cancer Prevention and Control Centers for Disease Control and Prevention
RAISE Network Webinar Series. Asian Smokers Quitline (ASQ): Promoting Cessation in Our Communities. March 17, :00 pm 2:00 pm PT
 RAISE Network Webinar Series Asian Smokers Quitline (ASQ): Promoting Cessation in Our Communities March 17, 2015 1:00 pm 2:00 pm PT National Council of Asian Pacific Americans Website: www.ncapaonline.org
RAISE Network Webinar Series Asian Smokers Quitline (ASQ): Promoting Cessation in Our Communities March 17, 2015 1:00 pm 2:00 pm PT National Council of Asian Pacific Americans Website: www.ncapaonline.org
Black Women s Access to Health Insurance
 FACT SHEET Black Women s Access to Health Insurance APRIL 2018 Data released by the U.S. Census Bureau show that, despite significant health insurance gains since the Affordable Care Act (ACA) was implemented,
FACT SHEET Black Women s Access to Health Insurance APRIL 2018 Data released by the U.S. Census Bureau show that, despite significant health insurance gains since the Affordable Care Act (ACA) was implemented,
Voluntary Mental Health Treatment Laws for Minors & Length of Inpatient Stay. Tori Lallemont MPH Thesis: Maternal & Child Health June 6, 2007
 Voluntary Mental Health Treatment Laws for Minors & Length of Inpatient Stay Tori Lallemont MPH Thesis: Maternal & Child Health June 6, 2007 Introduction 1997: Nearly 300,000 children were admitted to
Voluntary Mental Health Treatment Laws for Minors & Length of Inpatient Stay Tori Lallemont MPH Thesis: Maternal & Child Health June 6, 2007 Introduction 1997: Nearly 300,000 children were admitted to
AMERICAN IMMUNIZATION REGISTRY ASSOCIATION A L I S O N C H I, P R O G R A M D I R E C T O R
 AMERICAN IMMUNIZATION REGISTRY ASSOCIATION A L I S O N C H I, P R O G R A M D I R E C T O R WHAT IS AIRA A way to work together to develop standards, best practices, develop and share strategies and provide
AMERICAN IMMUNIZATION REGISTRY ASSOCIATION A L I S O N C H I, P R O G R A M D I R E C T O R WHAT IS AIRA A way to work together to develop standards, best practices, develop and share strategies and provide
Q1 What is your age?
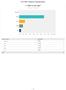 Q What is your age? Answ ered: 9 Skipped: Under 3 3-33 34-37 38-4 4+ Under 3 3-33 34-37 38-4 4+ 68.42% 3 5.79% 3.53% 2 5.26% Total 9 / 35 Q2 What is your gender? Answ ered: 9 Skipped: Male Female Male
Q What is your age? Answ ered: 9 Skipped: Under 3 3-33 34-37 38-4 4+ Under 3 3-33 34-37 38-4 4+ 68.42% 3 5.79% 3.53% 2 5.26% Total 9 / 35 Q2 What is your gender? Answ ered: 9 Skipped: Male Female Male
ADEA Survey of Dental School Seniors, 2015 Graduating Class Tables Report
 ADEA Survey of Dental School Seniors, 2015 Graduating Class Tables Report Published March 2016 Suggested Citation American Dental Education Association. (March 2016). ADEA Survey of Dental School Seniors,
ADEA Survey of Dental School Seniors, 2015 Graduating Class Tables Report Published March 2016 Suggested Citation American Dental Education Association. (March 2016). ADEA Survey of Dental School Seniors,
Eylea (aflibercept) Document Number: IC-0026
 Eylea (aflibercept) Document Number: IC-0026 Last Review Date: 3/1/2018 Date of Origin: 02/07/2013 Dates Reviewed: 03/07/2013, 06/2013, 09/2013, 12/2013, 03/2014, 06/2014, 09/2014, 12/2014, 03/2015, 04/2015,
Eylea (aflibercept) Document Number: IC-0026 Last Review Date: 3/1/2018 Date of Origin: 02/07/2013 Dates Reviewed: 03/07/2013, 06/2013, 09/2013, 12/2013, 03/2014, 06/2014, 09/2014, 12/2014, 03/2015, 04/2015,
Rituxan Hycela (rituximab and hyaluronidase human) (Subcutaneous)
 Rituxan Hycela (rituximab and hyaluronidase human) (Subcutaneous) Document Number: IC-0322 Last Review Date: 02/06/2018 Date of Origin: 7/20/2010 Dates Reviewed: 09/2010, 12/2010, 02/2011, 03/2011, 05/2011,
Rituxan Hycela (rituximab and hyaluronidase human) (Subcutaneous) Document Number: IC-0322 Last Review Date: 02/06/2018 Date of Origin: 7/20/2010 Dates Reviewed: 09/2010, 12/2010, 02/2011, 03/2011, 05/2011,
How Often Do Americans Eat Vegetarian Meals? And How Many Adults in the U.S. Are Vegetarian? Posted on May 29, 2015 by The VRG Blog Editor
 How Often Do Americans Eat Vegetarian Meals? And How Many Adults in the U.S. Are Vegetarian? Posted on May 29, 2015 by The VRG Blog Editor The Vegetarian Resource Group asks in a 2015 National Survey Conducted
How Often Do Americans Eat Vegetarian Meals? And How Many Adults in the U.S. Are Vegetarian? Posted on May 29, 2015 by The VRG Blog Editor The Vegetarian Resource Group asks in a 2015 National Survey Conducted
Shu-Hong Zhu, PhD University of California, San Diego INTRODUCING THE ASIAN SMOKERS QUITLINE (ASQ)
 Shu-Hong Zhu, PhD University of California, San Diego INTRODUCING THE ASIAN SMOKERS QUITLINE (ASQ) THE REACH AND EFFICACY OF A MULTILINGUAL TELEPHONE QUITLINE FOR ASIAN SMOKERS English Quitlines in 1992
Shu-Hong Zhu, PhD University of California, San Diego INTRODUCING THE ASIAN SMOKERS QUITLINE (ASQ) THE REACH AND EFFICACY OF A MULTILINGUAL TELEPHONE QUITLINE FOR ASIAN SMOKERS English Quitlines in 1992
Comprehensive Genomic Profiling, in record time. Accurate. Clinically Proven. Fast.
 Comprehensive Genomic Profiling, in record time Accurate. ly Proven. Fast. PCDx advantages Comprehensive genomic profiling, in record time PCDx Comprehensive Genomic Profiling (CGP) provides precise information
Comprehensive Genomic Profiling, in record time Accurate. ly Proven. Fast. PCDx advantages Comprehensive genomic profiling, in record time PCDx Comprehensive Genomic Profiling (CGP) provides precise information
Yervoy (ipilmumab) Last Review Date: 03/25/2014 Date of Origin: 11/28/2011. Prior Auth Available: Post-Service Edit:
 Yervoy (ipilmumab) Date of Origin: 11/28/2011 Prior Auth Available: Post-Service Edit: Dates Reviewed: 12/13/2011, 03/2012, 06/19/2012, 09/06/2012, 12/06/2012, 05/16/2013, 06/06/2013, 09/05/2013, 12/05/2013,
Yervoy (ipilmumab) Date of Origin: 11/28/2011 Prior Auth Available: Post-Service Edit: Dates Reviewed: 12/13/2011, 03/2012, 06/19/2012, 09/06/2012, 12/06/2012, 05/16/2013, 06/06/2013, 09/05/2013, 12/05/2013,
Michael A. Preston, Ph.D., M.P.H. University of Arkansas for Medical
 Michael A. Preston, Ph.D., M.P.H. University of Arkansas for Medical Sciences mapreston@uams.edu @MDonP Community Health Centers of Arkansas Annual Conference Little Rock, AR 27 September 2018 No Financial
Michael A. Preston, Ph.D., M.P.H. University of Arkansas for Medical Sciences mapreston@uams.edu @MDonP Community Health Centers of Arkansas Annual Conference Little Rock, AR 27 September 2018 No Financial
Reducing Barriers to Risk Appropriate Cancer Genetics Services: Current Strategies
 Reducing Barriers to Risk Appropriate Cancer Genetics Services: Current Strategies Kara J. Milliron, MS, CGC Mark D. Pearlman, MD Disclosure I am a contract genetic counselor with Informed DNA, Inc. The
Reducing Barriers to Risk Appropriate Cancer Genetics Services: Current Strategies Kara J. Milliron, MS, CGC Mark D. Pearlman, MD Disclosure I am a contract genetic counselor with Informed DNA, Inc. The
2018 Perinatal Hepatitis B Summit. Eva Hansson, RN, MSN Perinatal Hepatitis B Prevention Coordinator
 2018 Perinatal Hepatitis B Summit Eva Hansson, RN, MSN Perinatal Hepatitis B Prevention Coordinator Overview of Summit Summary of perinatal Hepatitis B in the US & Texas ACIP Hepatitis B Prevention recommendations
2018 Perinatal Hepatitis B Summit Eva Hansson, RN, MSN Perinatal Hepatitis B Prevention Coordinator Overview of Summit Summary of perinatal Hepatitis B in the US & Texas ACIP Hepatitis B Prevention recommendations
Program Overview. Karen L. Swartz, M.D. Johns Hopkins University School of Medicine
 Program Overview Karen L. Swartz, M.D. Johns Hopkins University School of Medicine 0 has generously funded the expansion of ADAP in Washington DC and Texas Objectives Why was ADAP created? What ADAP is
Program Overview Karen L. Swartz, M.D. Johns Hopkins University School of Medicine 0 has generously funded the expansion of ADAP in Washington DC and Texas Objectives Why was ADAP created? What ADAP is
StrandAdvantage Tissue-Specific Cancer Genomic Tests. Empowering Crucial First-Line Therapy Decisions for Your Patient
 StrandAdvantage Tissue-Specific Cancer Genomic Tests Empowering Crucial First-Line Therapy Decisions for Your Patient Harness the power of precision medicine with StrandAdvantage Precision medicine in
StrandAdvantage Tissue-Specific Cancer Genomic Tests Empowering Crucial First-Line Therapy Decisions for Your Patient Harness the power of precision medicine with StrandAdvantage Precision medicine in
Alzheimer s Association Clinical Studies Initiative
 Alzheimer s Association Clinical Studies Initiative Presented at the October 4, 2007 meeting on Recruitment and Retention Challenges and Opportunities For the Alzheimer Disease Centers By Paula Moore Director,
Alzheimer s Association Clinical Studies Initiative Presented at the October 4, 2007 meeting on Recruitment and Retention Challenges and Opportunities For the Alzheimer Disease Centers By Paula Moore Director,
Director s Update Brief novel 2009-H1N1. Tuesday 21 JUL EDT Day 95. Week of: Explaining the burden of disease and aligning resources
 Director s Update Brief novel 9-H1N1 Tuesday 1 JUL 9 815 EDT Day 95 Week of: Explaining the burden of disease and aligning resources Key Events novel 9-H1N1 Declarations WHO: Pandemic Phase (11 JUN 9 1
Director s Update Brief novel 9-H1N1 Tuesday 1 JUL 9 815 EDT Day 95 Week of: Explaining the burden of disease and aligning resources Key Events novel 9-H1N1 Declarations WHO: Pandemic Phase (11 JUN 9 1
Mexico. April August 2009: first wave
 Partners in Prepare edness Conference H1N1 Planning, Re esponse, Recovery April 6, 2010 Overview H1N1 Event April 24: first cases appeared in US and Mexico April 26: public health emergency declared April
Partners in Prepare edness Conference H1N1 Planning, Re esponse, Recovery April 6, 2010 Overview H1N1 Event April 24: first cases appeared in US and Mexico April 26: public health emergency declared April
CDC s National Comprehensive Cancer Control Program (NCCCP): 2010 Priorities and New Program Opportunities
 CDC s National Comprehensive Cancer Control Program (NCCCP): 2010 Priorities and New Program Opportunities Laura Seeff MD Chief, Comprehensive Cancer Control Branch Division of Cancer Prevention and Control
CDC s National Comprehensive Cancer Control Program (NCCCP): 2010 Priorities and New Program Opportunities Laura Seeff MD Chief, Comprehensive Cancer Control Branch Division of Cancer Prevention and Control
Number of fatal work injuries,
 Number of fatal work injuries, 1992 2010 Number of fatal work injuries 7,000 6,217 6,331 6,632 6,275 6,202 6,238 6,055 6,054 5,920 5,915 6,000 5,534 5,575 5,764 5,734 5,840 5,657 5,214 5,000 4,551 4,690
Number of fatal work injuries, 1992 2010 Number of fatal work injuries 7,000 6,217 6,331 6,632 6,275 6,202 6,238 6,055 6,054 5,920 5,915 6,000 5,534 5,575 5,764 5,734 5,840 5,657 5,214 5,000 4,551 4,690
The Right Hit. DEVELOPING EFFECTIVE MEDIA STRATEGY AT SYRINGE SERVICES PROGRAMS
 The Right Hit. DEVELOPING EFFECTIVE MEDIA STRATEGY AT SYRINGE SERVICES PROGRAMS AIDS United Presenters Jess Tilley Executive Director, HRH413 Kyle Barbour, M.D. Resident Physician, University of Rochester,
The Right Hit. DEVELOPING EFFECTIVE MEDIA STRATEGY AT SYRINGE SERVICES PROGRAMS AIDS United Presenters Jess Tilley Executive Director, HRH413 Kyle Barbour, M.D. Resident Physician, University of Rochester,
Awareness and Education Survey, 2012 Education & Best Practices Committee
 Awareness and Education Survey, 2012 Education & Best Practices Committee October 7, 2012 Wolfram Blattner President, North Texas Prostate Cancer Coalition (NTxPCC) Note: All of the Comments are my own
Awareness and Education Survey, 2012 Education & Best Practices Committee October 7, 2012 Wolfram Blattner President, North Texas Prostate Cancer Coalition (NTxPCC) Note: All of the Comments are my own
The Growing Health and Economic Burden of Older Adult Falls- Recent CDC Research
 The Growing Health and Economic Burden of Older Adult Falls- Recent CDC Research Gwen Bergen, PhD gjb8@cdc.gov Division of Unintentional Injury Prevention National Center for Injury Prevention and Control
The Growing Health and Economic Burden of Older Adult Falls- Recent CDC Research Gwen Bergen, PhD gjb8@cdc.gov Division of Unintentional Injury Prevention National Center for Injury Prevention and Control
MEDICAID FINANCING OF HPV VACCINE: Access for Low-Income Women
 MEDICAID FINANCING OF HPV VACCINE: Access for Low-Income Women For the American Public Health Association s 135 th Annual Meeting Alexandra Stewart, JD Department of Health Policy November 7, 2007 1 The
MEDICAID FINANCING OF HPV VACCINE: Access for Low-Income Women For the American Public Health Association s 135 th Annual Meeting Alexandra Stewart, JD Department of Health Policy November 7, 2007 1 The
SNAP Outreach within Food Banks: A View From The Ground
 SNAP Outreach within Food Banks: A View From The Ground Shana Alford, Feeding America Colleen Heflin, University of Missouri Elaine Waxman, Feeding America FEEDING AMERICA + PARTNER NAME PARTNERSHIP DISCUSSION
SNAP Outreach within Food Banks: A View From The Ground Shana Alford, Feeding America Colleen Heflin, University of Missouri Elaine Waxman, Feeding America FEEDING AMERICA + PARTNER NAME PARTNERSHIP DISCUSSION
Women s Health Coverage: Stalled Progress
 FACT SHEET Women s Health Coverage: Stalled Progress SEPTEMBER 2018 New data from the U.S. Census Bureau show that 1 in 10 women lack access to health insurance. This year, progress in reducing the number
FACT SHEET Women s Health Coverage: Stalled Progress SEPTEMBER 2018 New data from the U.S. Census Bureau show that 1 in 10 women lack access to health insurance. This year, progress in reducing the number
