79956F Kitano et al_bi754091bi TiP_JSMO 2018 (NB).pdf 1 13/07/ :55:06 C M Y CM MY CY CMY K
|
|
|
- William Harrington
- 5 years ago
- Views:
Transcription
1 79956F Kitano et al_bi754091bi TiP_JSMO 2018 (NB).pdf 1 13/07/ :55:06 C M Y CM MY CY CMY K
2 Phase 1 trial of an anti-pd-1 mab BI ± an anti-lag-3 mab BI in Asian patients with advanced solid tumors Shigehisa Kitano, 1 Noriko Kobayashi, 1 Mabrouk Elgadi, 2 Ralph Graeser, 3 Miaomiao Ge, 4 Kosuke Ishida, 5 Akiko Harada, 5 Yoshifumi Tachibana, 5 Noboru Yamamoto 1 1 Department of Experimental Therapeutics, National Cancer Center Hospital, Tokyo, Japan; 2 Boehringer Ingelheim Canada Ltd., Burlington, ON, Canada; 3 Boehringer Ingelheim Pharma GmbH & Co. KG, Biberach, Germany; 4 Boehringer Ingelheim International Pharmaceuticals Inc., Ridgefield, CT, USA; 5 Nippon Boehringer Ingelheim Co. Ltd., Tokyo, Japan Presented at 2018 Japanese Society of Medical Oncology Annual Meeting (JSMO), July 19 21, 2018, Kobe, Japan
3 Introduction Background Signaling via the immune checkpoint molecules PD-1 and LAG-3 can lead to T-cell dysfunction and prevent antitumor responses 1 Dual inhibition of PD-1 and LAG-3 may synergistically restore T-cell activation and enhance antitumor immune responses 1,2 Combined treatment with an anti-pd-1 monoclonal antibody (mab) and an anti- LAG-3 mab may achieve greater rates and durations of response compared with anti-pd-1 mab monotherapy Anti-PD-1/anti-LAG-3 mab combinations have demonstrated encouraging preclinical 3 and clinical 4 antitumor activity Preliminary study results in patients with advanced solid tumors demonstrate that BI is safe and well-tolerated up to 400 mg, with evidence of antitumor activity 5 In this Phase I trial (NCT ), the anti-pd-1 mab BI will be evaluated in Asian patients with advanced, unresectable, and/or metastatic solid tumors (as monotherapy and together with the anti-lag-3 mab, BI ) mab, monoclonal antibody
4 Introduction (cont d) Objectives Assess the safety, tolerability and PK of BI with and without BI Determine the MTD and/or RP2D of BI monotherapy, and BI plus BI Assess the efficacy of the RP2D of BI plus BI in patients with G/EJC, EC or HCC EC, esophageal cancer; G/EJC, gastric/esophagogastric junction cancer; HCC, hepatocellular cancer; MTD, maximum tolerated dose; PK, pharmacokinetics; RP2D, recommended Phase II dose
5 Eligibility criteria Key inclusion criteria Adult patients Measurable lesions, according to Response Evaluation Criteria in Solid Tumors (RECIST) v1.1 Parts 1 and 2: confirmed diagnosis of advanced, unresectable, and/or metastatic solid tumors (any type) Part 3 (Cohorts 1 3): 1 line of systemic therapy (excluding adjuvant), no prior anti-pd-1/pd-l1 mab therapy Part 3 (Cohort 4): disease progression on or after prior anti-pd-1/pd-l1 mab therapy mab, monoclonal antibody; RECIST, Response Evaluation Criteria in Solid Tumors
6 Eligibility criteria (cont d) Key exclusion criteria Antitumor treatment within 4 weeks or 5 half-life periods (whichever is shorter) Part 2 and 3 only: prior anti-lag-3 therapy Inadequate organ function or bone marrow reserve (based on laboratory values) Immunosuppressive corticosteroids (>10 mg/day prednisone, or equivalent) <4 weeks prior to first dose of treatment History of pneumonitis (in last 5 years), HIV or interstitial lung disease Active autoimmune disease Active infection requiring systemic treatment
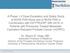
7 Study design Open-label, single-arm, Phase I trial in patients with advanced solid tumors Part 1 Part 2 BI mg via iv infusion; Q3W RP2D BI BI Starting doses: 240 mg from Part 1 and 80 mg via iv infusion; Q3W Japanese patients with advanced solid tumors of any type RP2D Q3W, once every three weeks
8 Study design (cont d) Part 3 BI BI (at RP2D) ~140 Cohort 1 Cohort 2 Cohort 3 Gastric/ esophagogastric junction cancer Esophageal cancer Hepatocellular cancer Asian patients (including Japan, Taiwan and Korea) No previous anti-pd-1 / PD-L1 mab therapy 1 line of systemic therapy Cohort 4 Gastric/esophagogastric junction, esophageal, or hepatocellular cancer Asian patients (including Japan, Taiwan and Korea) Failure of previous anti-pd-1 / PD-L1 mab therapy mab, monoclonal antibody; RP2D, recommended Phase II dose
9 Endpoints Primary endpoints Parts 1 and 2 MTD of BI and BI BI , based on dose-limiting toxicities in Cycle 1 Part 3 Investigator assessed objective response according to RECIST v1.1 MTD, maximum tolerated dose; RECIST, Response Evaluation Criteria in Solid Tumors
10 Endpoints (cont d) Secondary endpoints Parts 1 and 2 BI ; BI BI plasma C max and AUC Parts 1 and 2 Investigator assessed objective response according to RECIST v1.1 AUC, area under curve; C max, maximum drug concentration; RECIST, Response Evaluation Criteria in Solid Tumors
11 Endpoints (cont d) Secondary endpoints Part 3 Duration of response From Date of first documented investigator-assessed PR or CR according to RECIST v1.1 To Date of progressive disease or death Part 3 Disease control (investigator-assessed PR, CR or stable disease according to RECIST v1.1) CR, complete response; PR, partial response; RECIST, Response Evaluation Criteria in Solid Tumors
12 Key points Objectives: Parts 1 and 2 Determine safety, tolerability, PK, and MTD/RP2D of BI monotherapy and BI BI combination therapy Part 3 Explore the efficacy, and further investigate safety, tolerability and PK of BI BI in patients with G/EJC, EC or HCC Study design: Open-label, single-arm, Phase I trial in Asian patients with advanced, unresectable, and/or metastatic solid tumors EC, esophageal cancer; G/EJC, gastric/esophagogastric junction cancer; HCC, hepatocellular cancer; MTD, maximum tolerated dose; PK, pharmacokinetics; RP2D, recommended Phase II dose
13 Current status Enrollment opened in February, 2018 Target enrollment is 164 patients from Asian countries including Japan Part 1 is complete; no dose limiting toxicities were reported, RP2D of BI confirmed as 240 mg Part 2 opens for recruitment in July 2018 RP2D, recommended Phase II dose
14 References 1. Ascierto PA, et al. J Clin Oncol 2017;35: Blackburn SD, et al. Nat Immunol 2009;10: Woo S-R, et al. 2012;72: Ascierto PA, et al. Ann Oncol 2017;28(Suppl. 5):mdx Johnson M, et al. J Clin Oncol 2018;36(Suppl. 5):212 Acknowledgments This study is funded by Boehringer Ingelheim. The authors were fully responsible for all content and editorial decisions, were involved at all stages of poster development and have approved the final version. Medical writing assistance, supported financially by Boehringer Ingelheim, was provided by Mike Simpson of GeoMed, an Ashfield company, part of UDG Healthcare plc, during the development of this poster. *Corresponding author address: skitano@ncc.go.jp These materials are for personal use only and may not be reproduced without written permission of the authors and the appropriate copyright permissions.
Cohort D Xentuzumab + abemaciclib + fulvestrant (500 mg/month) Key exclusion criteria RP2D-4 NSCLC. Cohort F Xentuzumab + abemaciclib + fulvestrant
 A phase Ib trial of xentuzumab and abemaciclib in patients with locally advanced or metastatic solid tumors, including hormone receptor-positive, HER2-negative breast cancer (plus endocrine therapy) Douglas
A phase Ib trial of xentuzumab and abemaciclib in patients with locally advanced or metastatic solid tumors, including hormone receptor-positive, HER2-negative breast cancer (plus endocrine therapy) Douglas
Screening, Baseline Characteristics. BI and BI IV q3w. DLTs monitored throughout the study
 Phase I study of BI 754111 (anti-lag-3) plus BI 754091(anti-PD-1) in patients (pts) with advanced solid cancers, followed by expansion in pts with microsatellite stable metastatic colorectal cancer (mcrc),
Phase I study of BI 754111 (anti-lag-3) plus BI 754091(anti-PD-1) in patients (pts) with advanced solid cancers, followed by expansion in pts with microsatellite stable metastatic colorectal cancer (mcrc),
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
National Taiwan University Hospital and National Taiwan University Cancer Center, Taipei, Taiwan;
 Competing central nervous system or systemic progression analysis for patients with EGFR mutation-positive NSCLC receiving afatinib in LUX-Lung 3, 6, and 7 James C.-H. Yang, 1 Yi-Long Wu, 2 Vera Hirsh,
Competing central nervous system or systemic progression analysis for patients with EGFR mutation-positive NSCLC receiving afatinib in LUX-Lung 3, 6, and 7 James C.-H. Yang, 1 Yi-Long Wu, 2 Vera Hirsh,
Competing CNS or systemic progression analysis for EGFR mutation-positive NSCLC patients on afatinib in LUX-Lung 3, 6, and 7
 Competing CNS or systemic progression analysis for EGFR mutation-positive NSCLC patients on afatinib in LUX-Lung 3, 6, and 7 Diego Kaen, 1 James C.-H. Yang, 2 Yi-Long Wu, 3 Vera Hirsh, 4 Kenneth O Byrne,
Competing CNS or systemic progression analysis for EGFR mutation-positive NSCLC patients on afatinib in LUX-Lung 3, 6, and 7 Diego Kaen, 1 James C.-H. Yang, 2 Yi-Long Wu, 3 Vera Hirsh, 4 Kenneth O Byrne,
Post-marketing observational study of Japanese patients with EGFR mutation-positive (EGFRm+) NSCLC treated with daily afatinib (final report)
 Post-marketing observational study of Japanese patients with EGFR mutation-positive (EGFRm+) NSCLC treated with daily afatinib (final report) Nobuyuki Yamamoto, 1 Toshihiro Nukiwa, 2 Yoichi Nakanishi,
Post-marketing observational study of Japanese patients with EGFR mutation-positive (EGFRm+) NSCLC treated with daily afatinib (final report) Nobuyuki Yamamoto, 1 Toshihiro Nukiwa, 2 Yoichi Nakanishi,
Afatinib in patients with EGFR mutation-positive NSCLC harboring uncommon mutations: overview of clinical data
 Afatinib in patients with EGFR mutation-positive NSCLC harboring uncommon mutations: overview of clinical data Oscar Arrieta, 1 Pedro De Marchi, 2 Nobuyuki Yamamoto, 3 Chong-Jen Yu, 4 Sai-Hong I Ou, 5
Afatinib in patients with EGFR mutation-positive NSCLC harboring uncommon mutations: overview of clinical data Oscar Arrieta, 1 Pedro De Marchi, 2 Nobuyuki Yamamoto, 3 Chong-Jen Yu, 4 Sai-Hong I Ou, 5
Sequential treatment with afatinib and osimertinib in real-world patients with EGFR mutation-positive advanced NSCLC: the GioTag study
 Scan the QR code for an electronic copy of the poster and supplementary material Sequential treatment with afatinib and osimertinib in real-world patients with EGFR mutation-positive advanced NSCLC: the
Scan the QR code for an electronic copy of the poster and supplementary material Sequential treatment with afatinib and osimertinib in real-world patients with EGFR mutation-positive advanced NSCLC: the
Presented at the European Society of Medical Oncology (ESMO), Munich, Germany, October 2018
 Effectiveness of afatinib in clinical practice first results of the GIDEON trial: a prospective non-interventional study in EGFR-mutated NSCLC in Germany Wolfgang M. Brueckl 1 *, Eckart Laack 2, Martin
Effectiveness of afatinib in clinical practice first results of the GIDEON trial: a prospective non-interventional study in EGFR-mutated NSCLC in Germany Wolfgang M. Brueckl 1 *, Eckart Laack 2, Martin
Nivolumab in combination with ipilimumab in metastatic renal cell carcinoma (mrcc): Results of a phase I trial
 Nivolumab in combination with ipilimumab in metastatic renal cell carcinoma (mrcc): Results of a phase I trial H. Hammers, E.R. Plimack, J.R. Infante, M.S. Ernstoff, B. Rini, D.F. McDermott, A. Razak,
Nivolumab in combination with ipilimumab in metastatic renal cell carcinoma (mrcc): Results of a phase I trial H. Hammers, E.R. Plimack, J.R. Infante, M.S. Ernstoff, B. Rini, D.F. McDermott, A. Razak,
Impact of ERBB mutations on clinical outcomes in afatinib- or erlotinib-treated patients with SqCC of the lung
 Impact of ERBB mutations on clinical outcomes in afatinib- or erlotinib-treated patients with SqCC of the lung Glenwood D. Goss, Enriqueta Felip, 2 Manuel Cobo, 3 Shun Lu, 4 Konstantinos Syrigos, 5 Ki
Impact of ERBB mutations on clinical outcomes in afatinib- or erlotinib-treated patients with SqCC of the lung Glenwood D. Goss, Enriqueta Felip, 2 Manuel Cobo, 3 Shun Lu, 4 Konstantinos Syrigos, 5 Ki
More cancer patients are being treated with immunotherapy, but
 Bristol-Myers Squibb and Five Prime Present Phase 1a/1b Data Evaluating Cabiralizumab (anti-csf-1 receptor antibody) with Opdivo (nivolumab) in Patients with Advanced Solid Tumors PRINCETON, N.J. & SOUTH
Bristol-Myers Squibb and Five Prime Present Phase 1a/1b Data Evaluating Cabiralizumab (anti-csf-1 receptor antibody) with Opdivo (nivolumab) in Patients with Advanced Solid Tumors PRINCETON, N.J. & SOUTH
Bristol-Myers Squibb, Braine-l Alleud, Belgium; 12 MD Anderson Cancer Center, Houston, TX, USA
 3531 Combination of nivolumab (NIVO) + ipilimumab (IPI) in the treatment of patients (pts) with deficient DNA mismatch repair (dmmr)/high microsatellite instability (MSI-H) metastatic colorectal cancer
3531 Combination of nivolumab (NIVO) + ipilimumab (IPI) in the treatment of patients (pts) with deficient DNA mismatch repair (dmmr)/high microsatellite instability (MSI-H) metastatic colorectal cancer
TGFβR1 Kinase Inhibitor
 TGFβR1 Kinase Inhibitor Galunisertib, LY2157299 H 2 0 Derived from Prud homme GJ 1 ; Flavell RA, et al. 2 Drug Discovery Platform: Cancer Angiogenesis and Tumor Microenvironment/Immuno-Oncology A Phase
TGFβR1 Kinase Inhibitor Galunisertib, LY2157299 H 2 0 Derived from Prud homme GJ 1 ; Flavell RA, et al. 2 Drug Discovery Platform: Cancer Angiogenesis and Tumor Microenvironment/Immuno-Oncology A Phase
MELANOMA METASTASICO: NUEVAS COMBINACIONES. Dr Ana Arance MD PhD Oncología Médica Hospital Clínic Barcelona
 MELANOMA METASTASICO: NUEVAS COMBINACIONES Dr Ana Arance MD PhD Oncología Médica Hospital Clínic Barcelona Summary of OS accross clinical trials in patients with metastatic melanoma Ugurel et al. Eur J
MELANOMA METASTASICO: NUEVAS COMBINACIONES Dr Ana Arance MD PhD Oncología Médica Hospital Clínic Barcelona Summary of OS accross clinical trials in patients with metastatic melanoma Ugurel et al. Eur J
(generic name: ipilimumab) Injection 50 mg ( Yervoy ), a human anti-human CTLA-4 monoclonal. August 21, 2018
 August 21, 2018 Opdivo Approved for Supplemental Applications for Expanded Indications of Malignant Pleural Mesothelioma and Adjuvant Treatment of Melanoma, Change in Dosage and Administration (D&A) of
August 21, 2018 Opdivo Approved for Supplemental Applications for Expanded Indications of Malignant Pleural Mesothelioma and Adjuvant Treatment of Melanoma, Change in Dosage and Administration (D&A) of
Pembrolizumab for Patients With PD-L1 Positive Advanced Carcinoid or Pancreatic Neuroendocrine Tumors: Results From the KEYNOTE-028 Study
 Pembrolizumab for Patients With PD-L1 Positive Advanced Carcinoid or Pancreatic Neuroendocrine Tumors: Results From the KEYNOTE-28 Study Abstract 427O Mehnert JM, Bergsland E, O Neil BH, Santoro A, Schellens
Pembrolizumab for Patients With PD-L1 Positive Advanced Carcinoid or Pancreatic Neuroendocrine Tumors: Results From the KEYNOTE-28 Study Abstract 427O Mehnert JM, Bergsland E, O Neil BH, Santoro A, Schellens
Afatinib dose adjustment: Effect on safety, efficacy and patient-reported outcomes in the LUX-Lung 3/6 trials in EGFRm+ NSCLC
 Afatinib dose adjustment: Effect on safety, efficacy and patient-reported outcomes in the LUX-Lung 3/6 trials in EGFRm+ NSCLC Vera Hirsh, 1 Eng-Huat Tan, 2 Yi-Long Wu, 3 Lecia V. Sequist, 4 Caicun Zhou,
Afatinib dose adjustment: Effect on safety, efficacy and patient-reported outcomes in the LUX-Lung 3/6 trials in EGFRm+ NSCLC Vera Hirsh, 1 Eng-Huat Tan, 2 Yi-Long Wu, 3 Lecia V. Sequist, 4 Caicun Zhou,
TGFβR1 Kinase Inhibitor
 TGFβR1 Kinase Inhibitor Galunisertib, LY2157299 H 2 0 Prud homme GJ 1 ; Flavell RA, et al 2 Drug Discovery Platform: Cancer Angiogenesis and Tumor Microenvironment/Immuno-Oncology A Phase 1b/2 Dose-Escalation
TGFβR1 Kinase Inhibitor Galunisertib, LY2157299 H 2 0 Prud homme GJ 1 ; Flavell RA, et al 2 Drug Discovery Platform: Cancer Angiogenesis and Tumor Microenvironment/Immuno-Oncology A Phase 1b/2 Dose-Escalation
CONSIDERATIONS IN DEVELOPMENT OF PEMBROLIZUMAB IN MSI-H CANCERS
 CONSIDERATIONS IN DEVELOPMENT OF PEMBROLIZUMAB IN MSI-H CANCERS December 2017 Christine K. Gause, Ph.D Executive Director, Biostatistics. 2 Microsatellite Instability-High Cancer - USPI KEYTRUDA is indicated
CONSIDERATIONS IN DEVELOPMENT OF PEMBROLIZUMAB IN MSI-H CANCERS December 2017 Christine K. Gause, Ph.D Executive Director, Biostatistics. 2 Microsatellite Instability-High Cancer - USPI KEYTRUDA is indicated
Division of Hematology-Oncology, Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, South Korea
 Antitumor Activity and Safety of FPA144, an ADCCenhanced, FGFR2b Isoform-Selective Monoclonal Antibody, in Patients with FGFR2b + Gastric Cancer and Advanced Solid Tumors Jeeyun Lee 1, Johanna Bendell
Antitumor Activity and Safety of FPA144, an ADCCenhanced, FGFR2b Isoform-Selective Monoclonal Antibody, in Patients with FGFR2b + Gastric Cancer and Advanced Solid Tumors Jeeyun Lee 1, Johanna Bendell
ESMO 2017, Madrid, Spain Dr. Loredana Vecchione Charite Comprehensive Cancer Center, Berlin HIGHLIGHTS ON CANCERS OF THE UPPER GI TRACT
 ESMO 2017, Madrid, Spain Dr. Loredana Vecchione Charite Comprehensive Cancer Center, Berlin HIGHLIGHTS ON CANCERS OF THE UPPER GI TRACT DOCETAXEL, OXALIPLATIN AND FLUOROURACIL/LEUCOVORIN (FLOT) FOR RESECTABLE
ESMO 2017, Madrid, Spain Dr. Loredana Vecchione Charite Comprehensive Cancer Center, Berlin HIGHLIGHTS ON CANCERS OF THE UPPER GI TRACT DOCETAXEL, OXALIPLATIN AND FLUOROURACIL/LEUCOVORIN (FLOT) FOR RESECTABLE
Study No Title : Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Presentation by Dr. Thomas Yau on behalf of his co-authors
 4078 First presented at the American Society of Clinical Oncology (ASCO) 2016 Annual Meeting, Chicago, Illinois, USA, June 3-7, 2016. Reused with permission from the American Society of Clinical Oncology
4078 First presented at the American Society of Clinical Oncology (ASCO) 2016 Annual Meeting, Chicago, Illinois, USA, June 3-7, 2016. Reused with permission from the American Society of Clinical Oncology
Overall survival with afatinib versus chemotherapy in patients with NSCLC harboring common EGFR
 Overall survival with afatinib versus chemotherapy in patients with NSCLC harboring common EGFR mutations: subgroup analyses by race/ethnicity in LUX-Lung 3 and LUX-Lung 6 Yi-Long Wu, 1 Lecia V Sequist,
Overall survival with afatinib versus chemotherapy in patients with NSCLC harboring common EGFR mutations: subgroup analyses by race/ethnicity in LUX-Lung 3 and LUX-Lung 6 Yi-Long Wu, 1 Lecia V Sequist,
Clinical Study Synopsis for Public Disclosure
 abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis - which is part of
abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis - which is part of
A randomized phase 2 trial of CRLX101 in combination with bevacizumab in patients with metastatic renal cell carcinoma (mrcc) vs standard of care
 A randomized phase 2 trial of CRLX101 in combination with bevacizumab in patients with metastatic renal cell carcinoma (mrcc) vs standard of care Martin H. Voss 1, Thomas Hutson 2, Arif Hussain 3, Ulka
A randomized phase 2 trial of CRLX101 in combination with bevacizumab in patients with metastatic renal cell carcinoma (mrcc) vs standard of care Martin H. Voss 1, Thomas Hutson 2, Arif Hussain 3, Ulka
CheckMate 012: Safety and Efficacy of First Line Nivolumab and Ipilimumab in Advanced Non-Small Cell Lung Cancer
 CheckMate 12: Safety and Efficacy of First Line Nivolumab and Ipilimumab in Advanced Non-Small Cell Lung Cancer Abstract 31 Hellmann MD, Gettinger SN, Goldman J, Brahmer J, Borghaei H, Chow LQ, Ready NE,
CheckMate 12: Safety and Efficacy of First Line Nivolumab and Ipilimumab in Advanced Non-Small Cell Lung Cancer Abstract 31 Hellmann MD, Gettinger SN, Goldman J, Brahmer J, Borghaei H, Chow LQ, Ready NE,
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
Now Available: Final Rule for FDAAA 801 and NIH Policy on Clinical Trial Reporting
 A service of the U.S. National Institutes of Health Now Available: Final Rule for FDAAA 801 and NIH Policy on Clinical Trial Reporting Trial record 1 of 1 for: Keynote 355 Previous Study Return to List
A service of the U.S. National Institutes of Health Now Available: Final Rule for FDAAA 801 and NIH Policy on Clinical Trial Reporting Trial record 1 of 1 for: Keynote 355 Previous Study Return to List
PHASE 1 CLINICAL TRIALS: DESIGNS AND CONSIDERATIONS. Sarit Assouline, MD, MSc, FRCPC Associate professor, McGill University Jewish General Hospital
 PHASE 1 CLINICAL TRIALS: DESIGNS AND CONSIDERATIONS Sarit Assouline, MD, MSc, FRCPC Associate professor, McGill University Jewish General Hospital Outline Introduction Goal of Phase I studies in oncology
PHASE 1 CLINICAL TRIALS: DESIGNS AND CONSIDERATIONS Sarit Assouline, MD, MSc, FRCPC Associate professor, McGill University Jewish General Hospital Outline Introduction Goal of Phase I studies in oncology
AFATINIB FOLLOWED BY OSIMERTINIB IN REAL-WORLD PATIENTS WITH EGFR MUTATION-POSITIVE NSCLC: AN OBSERVATIONAL STUDY
 AFATINIB FOLLOWED BY OSIMERTINIB IN REAL-WORLD PATIENTS WITH EGFR MUTATION-POSITIVE NSCLC: AN OBSERVATIONAL STUDY Maximilian J. Hochmair, 1 Alessandro Morabito, 2 Desiree Hao, 3 Cheng-Ta Yang, 4 Ross A.
AFATINIB FOLLOWED BY OSIMERTINIB IN REAL-WORLD PATIENTS WITH EGFR MUTATION-POSITIVE NSCLC: AN OBSERVATIONAL STUDY Maximilian J. Hochmair, 1 Alessandro Morabito, 2 Desiree Hao, 3 Cheng-Ta Yang, 4 Ross A.
Challenges in Distinguishing Clinical Signals to Support Development Decisions: Case Studies
 Challenges in Distinguishing Clinical Signals to Support Development Decisions: Case Studies David Feltquate MD, PhD Head of Early Clinical Development, Oncology Bristol-Myers Squibb, Princeton, NJ Challenges
Challenges in Distinguishing Clinical Signals to Support Development Decisions: Case Studies David Feltquate MD, PhD Head of Early Clinical Development, Oncology Bristol-Myers Squibb, Princeton, NJ Challenges
A phase II umbrella trial in patients with relapsed ovarian cancer
 A phase II umbrella trial in patients with relapsed ovarian cancer NSGO-OV-UMB1 ENGOT-OV30 Sponsor: NSGO Relapsed ovarian cancer Treatment until disease progression A phase II umbrella trial in patients
A phase II umbrella trial in patients with relapsed ovarian cancer NSGO-OV-UMB1 ENGOT-OV30 Sponsor: NSGO Relapsed ovarian cancer Treatment until disease progression A phase II umbrella trial in patients
PI3K/mTOR Dual Inhibitor
 PI3K/mTOR Dual Inhibitor LY3023414 Courtney KD, et al 1 Drug Discovery Platform: Cancer Cell Signaling A Double-Blinded, Placebo-Controlled, Randomized Phase II Study of Enzalutamide With or Without the
PI3K/mTOR Dual Inhibitor LY3023414 Courtney KD, et al 1 Drug Discovery Platform: Cancer Cell Signaling A Double-Blinded, Placebo-Controlled, Randomized Phase II Study of Enzalutamide With or Without the
Merck Pfizer Alliance Strategy in gynecologic oncology
 Merck Pfizer Alliance Strategy in gynecologic oncology Lenka Kostková, MD., PhD. GCIG CCRN Educational symposium and Clinical Trials Workshop Bucharest, February 3 rd, 2018 RO/AVEOV/1217/0001 Avelumab
Merck Pfizer Alliance Strategy in gynecologic oncology Lenka Kostková, MD., PhD. GCIG CCRN Educational symposium and Clinical Trials Workshop Bucharest, February 3 rd, 2018 RO/AVEOV/1217/0001 Avelumab
Nuevas vías de administración de Trastuzumab MIQUEL ÀNGEL SEGUÍ PALMER
 Nuevas vías de administración de Trastuzumab MIQUEL ÀNGEL SEGUÍ PALMER Trastuzumab, a humanized monoclonal antibody against the extracellular domain of the HER2 receptor, is the standard-of-care treatment
Nuevas vías de administración de Trastuzumab MIQUEL ÀNGEL SEGUÍ PALMER Trastuzumab, a humanized monoclonal antibody against the extracellular domain of the HER2 receptor, is the standard-of-care treatment
Supplementary Online Content
 Supplementary Online Content Powles T, O Donnell PH, Massard C, et al. Efficacy and safety of durvalumab in locally advanced or metastatic urothelial carcinoma: updated results from a phase 1/2 openlabel
Supplementary Online Content Powles T, O Donnell PH, Massard C, et al. Efficacy and safety of durvalumab in locally advanced or metastatic urothelial carcinoma: updated results from a phase 1/2 openlabel
Presenter Disclosure Information
 Presenter Disclosure Information Tara C. Gangadhar, M.D. The following relationships exist related to this presentation: Research funding (Institution): Incyte Corporation and Merck & Co., Inc Preliminary
Presenter Disclosure Information Tara C. Gangadhar, M.D. The following relationships exist related to this presentation: Research funding (Institution): Incyte Corporation and Merck & Co., Inc Preliminary
Phase I and Pharmacodynamic Study of High-Dose NGR-hTNF in Patients
 Phase I and Pharmacodynamic Study of High-Dose NGR-hTNF in Patients with Refractory Solid Tumors P. A. Zucali 1, A. Santoro 1, M. Simonelli 1, F. De Vincenzo 1, E. Lorenzi 1, L. Rimassa 1, L. Balzarini
Phase I and Pharmacodynamic Study of High-Dose NGR-hTNF in Patients with Refractory Solid Tumors P. A. Zucali 1, A. Santoro 1, M. Simonelli 1, F. De Vincenzo 1, E. Lorenzi 1, L. Rimassa 1, L. Balzarini
First Phase 3 Results Presented for a PD-1 Immune Checkpoint Inhibitor
 September 30, 2014 Positive Phase 3 Data for Opdivo (nivolumab) in Advanced Melanoma Patients Previously Treated with Yervoy @ (ipilimumab) Presented at the ESMO 2014 Congress First Phase 3 Results Presented
September 30, 2014 Positive Phase 3 Data for Opdivo (nivolumab) in Advanced Melanoma Patients Previously Treated with Yervoy @ (ipilimumab) Presented at the ESMO 2014 Congress First Phase 3 Results Presented
STUDY DESIGN. VMP 6-week cycles, total of 9 cycles. Figure 2. Alcyone study design. Countries housing study sites are shaded in gold.
 TPS8608 A Randomized Open-label Study of Bortezomib, Melphalan, And Prednisone (VMP) Versus Daratumumab () Plus VMP in Patients With Previously Untreated Multiple Myeloma (MM) Who Are Ineligible for High-dose
TPS8608 A Randomized Open-label Study of Bortezomib, Melphalan, And Prednisone (VMP) Versus Daratumumab () Plus VMP in Patients With Previously Untreated Multiple Myeloma (MM) Who Are Ineligible for High-dose
Attached from the following page is the press release made by BMS for your information.
 June 2, 2015 Phase I/II Opdivo (nivolumab) Trial Shows Bristol-Myers Squibb s PD-1 Immune Checkpoint Inhibitor is First to Demonstrate Anti-Tumor Activity In Patients With Hepatocellular Carcinoma (PRINCETON,
June 2, 2015 Phase I/II Opdivo (nivolumab) Trial Shows Bristol-Myers Squibb s PD-1 Immune Checkpoint Inhibitor is First to Demonstrate Anti-Tumor Activity In Patients With Hepatocellular Carcinoma (PRINCETON,
ULTIMATE GBG 95 UC-0140/1606 BIG UnLock The IMmune cells ATtraction in ER+ breast cancer
 ULTIMATE GBG 95 UC-0140/1606 BIG 16-01 UnLock The IMmune cells ATtraction in ER+ breast cancer A PHASE II TRIAL TESTING DURVALUMAB COMBINED WITH ENDOCRINE THERAPY IN PATIENTS WITH ER+/HER2- BREAST CANCER
ULTIMATE GBG 95 UC-0140/1606 BIG 16-01 UnLock The IMmune cells ATtraction in ER+ breast cancer A PHASE II TRIAL TESTING DURVALUMAB COMBINED WITH ENDOCRINE THERAPY IN PATIENTS WITH ER+/HER2- BREAST CANCER
Primary Endpoint The primary endpoint is overall survival, measured as the time in weeks from randomization to date of death due to any cause.
 CASE STUDY Randomized, Double-Blind, Phase III Trial of NES-822 plus AMO-1002 vs. AMO-1002 alone as first-line therapy in patients with advanced pancreatic cancer This is a multicenter, randomized Phase
CASE STUDY Randomized, Double-Blind, Phase III Trial of NES-822 plus AMO-1002 vs. AMO-1002 alone as first-line therapy in patients with advanced pancreatic cancer This is a multicenter, randomized Phase
ASCO 2014 Highlights*
 ASCO 214 Highlights* Investor Meeting June 2, 214 *American Society of Clinical Oncology, May 3 June 3, 214 Forward-Looking Information During this meeting, we will make statements about the Company s
ASCO 214 Highlights* Investor Meeting June 2, 214 *American Society of Clinical Oncology, May 3 June 3, 214 Forward-Looking Information During this meeting, we will make statements about the Company s
Transforming science into medicine
 Transforming science into medicine 2 Forward-looking statements This presentation contains forward-looking statements. These statements include words like may, expects, believes, plans, scheduled, and
Transforming science into medicine 2 Forward-looking statements This presentation contains forward-looking statements. These statements include words like may, expects, believes, plans, scheduled, and
Atezolizumab Is a Humanized Anti-PDL1 Antibody That Inhibits the Binding of PD-L1 to PD-1 and B7.1
 Phase II, Single-Arm Trial (BIRCH) of Atezolizumab as First-Line or Subsequent Therapy for Locally Advanced or Metastatic PD-L1-Selected Non-Small Cell Lung Cancer (NSCLC) Abstract 16LBA Besse B, Johnson
Phase II, Single-Arm Trial (BIRCH) of Atezolizumab as First-Line or Subsequent Therapy for Locally Advanced or Metastatic PD-L1-Selected Non-Small Cell Lung Cancer (NSCLC) Abstract 16LBA Besse B, Johnson
R&D Conference Call. CHUGAI PHARMACEUTICAL CO., LTD. Department Manager of Oncology Lifecycle Management Dept. Megumi Uzu.
 R&D Conference Call CHUGAI PHARMACEUTICAL CO., LTD. Department Manager of Oncology Lifecycle Management Dept. Megumi Uzu July 4, 2016 Forward-Looking Statements This presentation may include forward-looking
R&D Conference Call CHUGAI PHARMACEUTICAL CO., LTD. Department Manager of Oncology Lifecycle Management Dept. Megumi Uzu July 4, 2016 Forward-Looking Statements This presentation may include forward-looking
GASTRIC & PANCREATIC CANCER
 GASTRIC & PANCREATIC CANCER ASCO HIGHLIGHTS 2005 Fadi Sami Farhat, MD Head of Hematology Oncology Division Hammoud Hospital University Medical Center Saida Lebanon Tel: +961 3 753 155 E-Mail: drfadi@drfadi.org
GASTRIC & PANCREATIC CANCER ASCO HIGHLIGHTS 2005 Fadi Sami Farhat, MD Head of Hematology Oncology Division Hammoud Hospital University Medical Center Saida Lebanon Tel: +961 3 753 155 E-Mail: drfadi@drfadi.org
Nivolumab: esperienze italiane nel carcinoma polmonare avanzato
 NSCLC avanzato: quali novità nel 2018? Negrar, 30 Ottobre 2018 Nivolumab: esperienze italiane nel carcinoma polmonare avanzato Francesco Grossi UOC Oncologia Medica Fondazione IRCCS Ca Granda Ospedale
NSCLC avanzato: quali novità nel 2018? Negrar, 30 Ottobre 2018 Nivolumab: esperienze italiane nel carcinoma polmonare avanzato Francesco Grossi UOC Oncologia Medica Fondazione IRCCS Ca Granda Ospedale
Clinical Pipeline Highlights
 Clinical Pipeline Highlights Geoffrey M. Nichol, M.D., M.B.A. Senior Vice President, Product Development Medarex, Inc. R&D Day December 9, 25 MDX-7: Anti-PSMA HuMAb for Prostate Cancer Biology PSMA expressed
Clinical Pipeline Highlights Geoffrey M. Nichol, M.D., M.B.A. Senior Vice President, Product Development Medarex, Inc. R&D Day December 9, 25 MDX-7: Anti-PSMA HuMAb for Prostate Cancer Biology PSMA expressed
The Current Prostate Cancer Landscape
 A Phase 1-2 Dose-Escalation and Safety Study of ADXS-PSA Alone and of ADXS-PSA in Combination with KEYTRUDA (MK-3475) in Patients with Previously Treated Metastatic Castration-Resistant Prostate Cancer
A Phase 1-2 Dose-Escalation and Safety Study of ADXS-PSA Alone and of ADXS-PSA in Combination with KEYTRUDA (MK-3475) in Patients with Previously Treated Metastatic Castration-Resistant Prostate Cancer
2019 ASCO-SITC. Nektar Therapeutics Investor & Analyst Call. March 1, 2019
 Nektar Therapeutics Investor & Analyst Call March 1, 2019 This presentation includes forward-looking statements regarding Nektar s proprietary drug candidates, the timing of the start and conclusion of
Nektar Therapeutics Investor & Analyst Call March 1, 2019 This presentation includes forward-looking statements regarding Nektar s proprietary drug candidates, the timing of the start and conclusion of
This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data.
 abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the
abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the
Clinical and statistical considerations of bridging approaches to alternative dosing regimen or formulations
 Clinical and statistical considerations of bridging approaches to alternative dosing regimen or formulations Dominik Heinzmann, PhD Global Development Team Leader Biostatistics Manager F. Hoffmann-La Roche,
Clinical and statistical considerations of bridging approaches to alternative dosing regimen or formulations Dominik Heinzmann, PhD Global Development Team Leader Biostatistics Manager F. Hoffmann-La Roche,
METRIC Study Key Eligibility Criteria
 The METRIC Study METRIC Study Key Eligibility Criteria The pivotal METRIC Study is evaluating glembatumumab vedotin in patients with gpnmb overexpressing metastatic triple-negative breast cancer (TNBC).
The METRIC Study METRIC Study Key Eligibility Criteria The pivotal METRIC Study is evaluating glembatumumab vedotin in patients with gpnmb overexpressing metastatic triple-negative breast cancer (TNBC).
My name is Dr. David Ilson, Professor of Medicine at Memorial Sloan Kettering Cancer Center and Weill Cornell Medical Center in New York, New York.
 Welcome to this CME/CE-certified activity entitled, Integrating the Latest Advances Into Clinical Experience: Data and Expert Insights From the 2016 Meeting on Gastrointestinal Cancers in San Francisco.
Welcome to this CME/CE-certified activity entitled, Integrating the Latest Advances Into Clinical Experience: Data and Expert Insights From the 2016 Meeting on Gastrointestinal Cancers in San Francisco.
Cisplatin plus Gemcitabine versus Gemcitabine for Biliary Tract Cancer. Valle J et al. N Engl J Med 2010;362(14):
 Cisplatin plus Gemcitabine versus Gemcitabine for Biliary Tract Cancer Valle J et al. N Engl J Med 2010;362(14):1273-81. Introduction > Biliary tract cancers (BTC: cholangiocarcinoma, gall bladder cancer,
Cisplatin plus Gemcitabine versus Gemcitabine for Biliary Tract Cancer Valle J et al. N Engl J Med 2010;362(14):1273-81. Introduction > Biliary tract cancers (BTC: cholangiocarcinoma, gall bladder cancer,
The 2010 Gastrointestinal Cancers Symposium Oral Abstract Session: Cancers of the Pancreas, Small Bowel and Hepatobilliary Tract
 The 2010 Gastrointestinal Cancers Symposium : Cancers of the Pancreas, Small Bowel and Hepatobilliary Tract Abstract #131: Phase I study of MK 0646 (dalotuzumab), a humanized monoclonal antibody against
The 2010 Gastrointestinal Cancers Symposium : Cancers of the Pancreas, Small Bowel and Hepatobilliary Tract Abstract #131: Phase I study of MK 0646 (dalotuzumab), a humanized monoclonal antibody against
PTAC meeting held on 5 & 6 May (minutes for web publishing)
 PTAC meeting held on 5 & 6 May 2016 (minutes for web publishing) PTAC minutes are published in accordance with the Terms of Reference for the Pharmacology and Therapeutics Advisory Committee (PTAC) and
PTAC meeting held on 5 & 6 May 2016 (minutes for web publishing) PTAC minutes are published in accordance with the Terms of Reference for the Pharmacology and Therapeutics Advisory Committee (PTAC) and
Investor Update. Basel, 21 October 2018
 Investor Update Basel, 21 October 2018 Updated data showed that Roche's Tecentriq in combination with Avastin shrank tumours in people with unresectable or advanced hepatocellular carcinoma (HCC) Data
Investor Update Basel, 21 October 2018 Updated data showed that Roche's Tecentriq in combination with Avastin shrank tumours in people with unresectable or advanced hepatocellular carcinoma (HCC) Data
At Fox Chase Cancer Centre during study participation
 Long-Term Outcome of a Phase 1 Study of the Investigational Oral Proteasome Inhibitor Ixazomib at the Recommended Phase 3 Dose in Patients with Relapsed or Refractory Systemic Light-Chain (AL) Amyloidosis
Long-Term Outcome of a Phase 1 Study of the Investigational Oral Proteasome Inhibitor Ixazomib at the Recommended Phase 3 Dose in Patients with Relapsed or Refractory Systemic Light-Chain (AL) Amyloidosis
Evan J. Lipson, M.D.
 Update on treatment for Merkel cell, cutaneous squamous cell and basal cell cancers Evan J. Lipson, M.D. The Johns Hopkins University School of Medicine Bloomberg~Kimmel Institute for Cancer Immunotherapy
Update on treatment for Merkel cell, cutaneous squamous cell and basal cell cancers Evan J. Lipson, M.D. The Johns Hopkins University School of Medicine Bloomberg~Kimmel Institute for Cancer Immunotherapy
A Phase I Study of Oral ABL001 in Patients With CML or Ph+ ALL
 A service of the U.S. National Institutes of Health Now Available: Final Rule for FDAAA 801 and NIH Policy on Clinical Trial Reporting Trial record 1 of 1 for: CABL001X2101 Previous Study Return to List
A service of the U.S. National Institutes of Health Now Available: Final Rule for FDAAA 801 and NIH Policy on Clinical Trial Reporting Trial record 1 of 1 for: CABL001X2101 Previous Study Return to List
Avelumab in M etastatic or Locally Advanced Solid Tumors (JAVELIN Solid Tumor)
 A service of the U.S. National Institutes of Health Trial record 1 of 1 for: NCT01772004 Previous Study Return to List Next Study Avelumab in M etastatic or Locally Advanced Solid Tumors (JAVELIN Solid
A service of the U.S. National Institutes of Health Trial record 1 of 1 for: NCT01772004 Previous Study Return to List Next Study Avelumab in M etastatic or Locally Advanced Solid Tumors (JAVELIN Solid
Metastatic NSCLC: Expanding Role of Immunotherapy. Evan W. Alley, MD, PhD Abramson Cancer Center at Penn Presbyterian
 Metastatic NSCLC: Expanding Role of Immunotherapy Evan W. Alley, MD, PhD Abramson Cancer Center at Penn Presbyterian Disclosures: No relevant disclosures Please note that some of the studies reported in
Metastatic NSCLC: Expanding Role of Immunotherapy Evan W. Alley, MD, PhD Abramson Cancer Center at Penn Presbyterian Disclosures: No relevant disclosures Please note that some of the studies reported in
Keytruda. Keytruda (pembrolizumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.21.50 Subject: Keytruda Page: 1 of 9 Last Review Date: November 30, 2018 Keytruda Description Keytruda
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.21.50 Subject: Keytruda Page: 1 of 9 Last Review Date: November 30, 2018 Keytruda Description Keytruda
Immunotherapy for Breast Cancer. Aurelio B. Castrellon Medical Oncology Memorial Healthcare System
 Immunotherapy for Breast Cancer Aurelio B. Castrellon Medical Oncology Memorial Healthcare System Conflicts Research support : Cascadian therapeutics, Puma biotechnology, Odonate therapeutics, Pfizer,
Immunotherapy for Breast Cancer Aurelio B. Castrellon Medical Oncology Memorial Healthcare System Conflicts Research support : Cascadian therapeutics, Puma biotechnology, Odonate therapeutics, Pfizer,
Chemotherapy and Immunotherapy in Combination Non-Small Cell Lung Cancer (NSCLC)
 Chemotherapy and Immunotherapy in Combination Non-Small Cell Lung Cancer (NSCLC) Jeffrey Crawford, MD George Barth Geller Professor for Research in Cancer Co-Program Leader, Solid Tumor Therapeutics Program
Chemotherapy and Immunotherapy in Combination Non-Small Cell Lung Cancer (NSCLC) Jeffrey Crawford, MD George Barth Geller Professor for Research in Cancer Co-Program Leader, Solid Tumor Therapeutics Program
This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data.
 abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the
abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the
1.0 Abstract. Title. Keywords. Adalimumab, Rheumatoid Arthritis, Effectiveness, Safety. Rationale and Background
 1.0 Abstract Title Assessment of the safety of adalimumab in rheumatoid arthritis (RA) patients showing rapid progression of structural damage of the joints, who have no prior history of treatment with
1.0 Abstract Title Assessment of the safety of adalimumab in rheumatoid arthritis (RA) patients showing rapid progression of structural damage of the joints, who have no prior history of treatment with
ICONIC: Biologic and clinical activity of first in class ICOS agonist antibody JTX /- nivolumab (nivo) in patients with advanced cancers
 ICONIC: Biologic and clinical activity of first in class ICOS agonist antibody JTX-211 +/- nivolumab (nivo) in patients with advanced cancers Timothy A. Yap 1, Howard A. Burris 2, Shivaani Kummar 3, Gerald
ICONIC: Biologic and clinical activity of first in class ICOS agonist antibody JTX-211 +/- nivolumab (nivo) in patients with advanced cancers Timothy A. Yap 1, Howard A. Burris 2, Shivaani Kummar 3, Gerald
Attached from the following page is the press release made by BMS for your information.
 June 2, 2015 Opdivo (nivolumab) Demonstrates Superior Survival Compared to Standard of Care (docetaxel) for Previously-Treated Squamous Non-Small Cell Lung Cancer in Phase III Trial (PRINCETON, NJ, May
June 2, 2015 Opdivo (nivolumab) Demonstrates Superior Survival Compared to Standard of Care (docetaxel) for Previously-Treated Squamous Non-Small Cell Lung Cancer in Phase III Trial (PRINCETON, NJ, May
Citi Global Healthcare Conference
 Citi Global Healthcare Conference Francis Cuss, MB BChir, FRCP Chief Scientific Officer February 24, 2014 1 Forward-Looking Information During this meeting, we will make statements about the Company s
Citi Global Healthcare Conference Francis Cuss, MB BChir, FRCP Chief Scientific Officer February 24, 2014 1 Forward-Looking Information During this meeting, we will make statements about the Company s
2016 Year-End Results and Conference Call. March 14, 2017
 2016 Year-End Results and Conference Call March 14, 2017 Forward Looking Statement This communication contains "forward-looking" statements within the meaning of the Private Securities Litigation Reform
2016 Year-End Results and Conference Call March 14, 2017 Forward Looking Statement This communication contains "forward-looking" statements within the meaning of the Private Securities Litigation Reform
VEGF Receptor-2 Antagonist
 VEGF Receptor-2 Antagonist Ramucirumab, LY3009806, IMC-1121B Derived from Adams RH and Alitalo K 1 ; Hicklin DJ and Ellis LM. 2 Drug Discovery Platform: Cancer Angiogenesis and Tumor Microenvironment RAINFALL:
VEGF Receptor-2 Antagonist Ramucirumab, LY3009806, IMC-1121B Derived from Adams RH and Alitalo K 1 ; Hicklin DJ and Ellis LM. 2 Drug Discovery Platform: Cancer Angiogenesis and Tumor Microenvironment RAINFALL:
Phase I Trial of Siplizumab in CD-2 Positive Lymphoproliferative Disease
 Phase I Trial of Siplizumab in CD-2 Positive Lymphoproliferative Disease J E Janik 1, D O Mahony 1, JC Morris 1, L Moses 1, D O Hagan 1, W Gao 1, M A Stetler-Stevenson 1, M Taylor 2, L Hammershaimb 2 T
Phase I Trial of Siplizumab in CD-2 Positive Lymphoproliferative Disease J E Janik 1, D O Mahony 1, JC Morris 1, L Moses 1, D O Hagan 1, W Gao 1, M A Stetler-Stevenson 1, M Taylor 2, L Hammershaimb 2 T
Phase 1 Study Combining Anti-PD-L1 (MEDI4736) With BRAF (Dabrafenib) and/or MEK (Trametinib) Inhibitors in Advanced Melanoma
 Phase 1 Study Combining Anti-PD-L1 (MEDI4736) With BRAF (Dabrafenib) and/or MEK (Trametinib) Inhibitors in Advanced Melanoma Abstract #3003 Ribas A, Butler M, Lutzky J, Lawrence D, Robert C, Miller W,
Phase 1 Study Combining Anti-PD-L1 (MEDI4736) With BRAF (Dabrafenib) and/or MEK (Trametinib) Inhibitors in Advanced Melanoma Abstract #3003 Ribas A, Butler M, Lutzky J, Lawrence D, Robert C, Miller W,
Attached from the following page is the press release made by BMS for your information.
 June 22, 2015 European Commission Approves Bristol-Myers Squibb s Opdivo (nivolumab), the First and Only PD-1 Checkpoint Inhibitor Approved in Europe, for Both First-Line and Previously-Treated Advanced
June 22, 2015 European Commission Approves Bristol-Myers Squibb s Opdivo (nivolumab), the First and Only PD-1 Checkpoint Inhibitor Approved in Europe, for Both First-Line and Previously-Treated Advanced
BAVARIAN NORDIC BIO DEUTSCHLAND PRESENTATION OCTOBER 2014 CSE/OMX:BAVA, OTC:BVNRY
 BAVARIAN NORDIC BIO DEUTSCHLAND PRESENTATION OCTOBER 2014 CSE/OMX:BAVA, OTC:BVNRY This presentation includes "forward-looking statements" that involve risks, uncertainties and other factors, many of which
BAVARIAN NORDIC BIO DEUTSCHLAND PRESENTATION OCTOBER 2014 CSE/OMX:BAVA, OTC:BVNRY This presentation includes "forward-looking statements" that involve risks, uncertainties and other factors, many of which
Idera Pharmaceuticals
 Idera Pharmaceuticals ILLUMINATE-204 Clinical Data Update December 2018 Forward Looking Statements and Other Important Cautions This presentation contains forward-looking statements within the meaning
Idera Pharmaceuticals ILLUMINATE-204 Clinical Data Update December 2018 Forward Looking Statements and Other Important Cautions This presentation contains forward-looking statements within the meaning
Osimertinib Activity in Patients With Leptomeningeal Disease From Non-Small Cell Lung Cancer: Updated Results From the BLOOM Study
 Osimertinib Activity in Patients With Leptomeningeal Disease From Non-Small Cell Lung Cancer: Updated Results From the BLOOM Study Abstract 9002 Yang JC, Kim DW, Kim SW, Cho BC, Lee JS, Ye X, Yin X, Yang
Osimertinib Activity in Patients With Leptomeningeal Disease From Non-Small Cell Lung Cancer: Updated Results From the BLOOM Study Abstract 9002 Yang JC, Kim DW, Kim SW, Cho BC, Lee JS, Ye X, Yin X, Yang
GBR 1302: EFFECT OF CD3-HER2, A BISPECIFIC T CELL ENGAGER ANTIBODY, IN TRASTUZUMAB-RESISTANT CANCERS
 GBR 1302: EFFECT OF CD3-HER2, A BISPECIFIC T CELL ENGAGER ANTIBODY, IN TRASTUZUMAB-RESISTANT CANCERS JONATHAN BACK 1 ; MARTIN WERMKE 2 ; JULIE MACOIN 1 ; AMELIE CROSET 1 ; JOHN KAUH 3 ; VENKATESHWAR REDDY
GBR 1302: EFFECT OF CD3-HER2, A BISPECIFIC T CELL ENGAGER ANTIBODY, IN TRASTUZUMAB-RESISTANT CANCERS JONATHAN BACK 1 ; MARTIN WERMKE 2 ; JULIE MACOIN 1 ; AMELIE CROSET 1 ; JOHN KAUH 3 ; VENKATESHWAR REDDY
Weitere Kombinationspartner der Immunotherapie
 1 Weitere Kombinationspartner der Immunotherapie Rolf Stahel University Hospital of Zürich Zürich, 9.12.216 2 Immunotherapy in a multimodality approach NSCLC Advanced disease Checkpoint inhibitors for
1 Weitere Kombinationspartner der Immunotherapie Rolf Stahel University Hospital of Zürich Zürich, 9.12.216 2 Immunotherapy in a multimodality approach NSCLC Advanced disease Checkpoint inhibitors for
New targets in endometrial and ovarian cancer
 New targets in endometrial and ovarian cancer SAMO Interdisciplinary Workshop on Gynecologic Tumors Luzern, January 16-17, 2016 C. Sessa IOSI Bellinzona Outline New targets in ovarian cancer - Cell cycle
New targets in endometrial and ovarian cancer SAMO Interdisciplinary Workshop on Gynecologic Tumors Luzern, January 16-17, 2016 C. Sessa IOSI Bellinzona Outline New targets in ovarian cancer - Cell cycle
SYNOPSIS. Issue Date: 31 July 2013
 SYNOPSIS Issue Date: 31 July 2013 Name of Sponsor/Company Name of Finished Product Name of Active Ingredient(s) Janssen Research & Development, LLC YONDELIS Trabectedin (R279741) Protocol No.: ET743-OVC-1003
SYNOPSIS Issue Date: 31 July 2013 Name of Sponsor/Company Name of Finished Product Name of Active Ingredient(s) Janssen Research & Development, LLC YONDELIS Trabectedin (R279741) Protocol No.: ET743-OVC-1003
Novel EGFR TKI Theliatinib: An Open Label, Dose Escalation Phase I Clinical Trial
 Novel EGFR TKI Theliatinib: An Open Label, Dose Escalation Phase I Clinical Trial 2014-309-00CH1 Presenter: Jifang Gong, Beijing Cancer Hospital Lin Shen 1, Li Zhang 2, Hongyun Zhao 2, Wenfeng Fang 2,
Novel EGFR TKI Theliatinib: An Open Label, Dose Escalation Phase I Clinical Trial 2014-309-00CH1 Presenter: Jifang Gong, Beijing Cancer Hospital Lin Shen 1, Li Zhang 2, Hongyun Zhao 2, Wenfeng Fang 2,
Open Clinical Trials: What s Out There Now Paula D. Ryan, MD, PhD
 Open Clinical Trials: What s Out There Now Paula D. Ryan, MD, PhD Hanahan and Weinberg, 2000 Acquired Capabilities of Cancer Clinical Trials When should I consider a clinical trial? How do I find the right
Open Clinical Trials: What s Out There Now Paula D. Ryan, MD, PhD Hanahan and Weinberg, 2000 Acquired Capabilities of Cancer Clinical Trials When should I consider a clinical trial? How do I find the right
Evolution of Early Phase Trials: Clinical Trial Design in the Modern Era
 Evolution of Early Phase Trials: Clinical Trial Design in the Modern Era Shivaani Kummar, MD, FACP Professor of Medicine (Oncology) Director, Phase I Clinical Research Program Co-Director, Translational
Evolution of Early Phase Trials: Clinical Trial Design in the Modern Era Shivaani Kummar, MD, FACP Professor of Medicine (Oncology) Director, Phase I Clinical Research Program Co-Director, Translational
mg 25 mg mg 25 mg mg 100 mg 1
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Liver and Biliary Tract Cancers Critical Review
 Liver and Biliary Tract Cancers Critical Review Lorenza Rimassa Oncologia Medica e Ematologia Humanitas Cancer Center Humanitas Research Hospital Rozzano (Milano) Critical review Oral presentations Melero
Liver and Biliary Tract Cancers Critical Review Lorenza Rimassa Oncologia Medica e Ematologia Humanitas Cancer Center Humanitas Research Hospital Rozzano (Milano) Critical review Oral presentations Melero
Summary of late stage clinical trials. As of Jul. 22, 2016
 Summary of late stage clinical trials As of Jul. 22, 2016 List of abbreviations AE BID DLT GFR iv MTD ORR OS PD PFS PK po PPK Q2W Q3W Q4W QD QW sc TID Adverse Events Twice daily Dose Limiting Toxicity
Summary of late stage clinical trials As of Jul. 22, 2016 List of abbreviations AE BID DLT GFR iv MTD ORR OS PD PFS PK po PPK Q2W Q3W Q4W QD QW sc TID Adverse Events Twice daily Dose Limiting Toxicity
2018 F. Hoffmann-La Roche Ltd. All rights reserved.
 PRELIMINARY EVIDENCE FOR PHARMACODYNAMIC EFFECTS OF RG7916 IN JEWELFISH A STUDY IN PATIENTS WITH SPINAL MUSCULAR ATROPHY WHO PREVIOUSLY PARTICIPATED IN A STUDY WITH ANOTHER SMN2-SPLICING TARGETING THERAPY
PRELIMINARY EVIDENCE FOR PHARMACODYNAMIC EFFECTS OF RG7916 IN JEWELFISH A STUDY IN PATIENTS WITH SPINAL MUSCULAR ATROPHY WHO PREVIOUSLY PARTICIPATED IN A STUDY WITH ANOTHER SMN2-SPLICING TARGETING THERAPY
Pembrolizumab in Relapsed/Refractory Classical Hodgkin Lymphoma: Phase 2 KEYNOTE-087 Study
 Pembrolizumab in Relapsed/Refractory Classical Hodgkin Lymphoma: Phase 2 KEYNOTE-087 Study Craig H. Moskowitz, 1 Pier Luigi Zinzani, 2 Michelle A. Fanale, 3 Philippe Armand, 4 Nathalie Johnson, 5 John
Pembrolizumab in Relapsed/Refractory Classical Hodgkin Lymphoma: Phase 2 KEYNOTE-087 Study Craig H. Moskowitz, 1 Pier Luigi Zinzani, 2 Michelle A. Fanale, 3 Philippe Armand, 4 Nathalie Johnson, 5 John
ASCO Annual Meeting 2013, May 31 June , Chicago, IL
 Phase I Study of Safety and Pharmacokinetics of GDC-0917, an Antagonist of Inhibitor of Apoptosis Proteins in Patients with Refractory Solid Tumors or Lymphoma 1 A Tolcher, 1 K Papadopoulos, 1 A Patnaik,
Phase I Study of Safety and Pharmacokinetics of GDC-0917, an Antagonist of Inhibitor of Apoptosis Proteins in Patients with Refractory Solid Tumors or Lymphoma 1 A Tolcher, 1 K Papadopoulos, 1 A Patnaik,
U.S. Food and Drug Administration Accepts Supplemental Biologics License Application. for Opdivo (nivolumab)
 September 3, 2015 U.S. Food and Drug Administration Accepts Supplemental Biologics License Application for Opdivo (nivolumab) in Previously Treated Non-Squamous Non-Small Cell Lung Cancer Patients (PRINCETON,
September 3, 2015 U.S. Food and Drug Administration Accepts Supplemental Biologics License Application for Opdivo (nivolumab) in Previously Treated Non-Squamous Non-Small Cell Lung Cancer Patients (PRINCETON,
La revolución de la inmunoterapia: dónde la posicionamos? Javier Puente, MD, PhD
 La revolución de la inmunoterapia: dónde la posicionamos? Javier Puente, MD, PhD Hospital Universitario Clinico San Carlos Medical Oncology Department Thoracic & Urological Cancer Unit Complutense University
La revolución de la inmunoterapia: dónde la posicionamos? Javier Puente, MD, PhD Hospital Universitario Clinico San Carlos Medical Oncology Department Thoracic & Urological Cancer Unit Complutense University
B I ABOUT BI DISEASE AREA & MECHANISM OF ACTION. For journalists outside UK/US/Canada only B A C K G R O U N D E R
 For journalists outside UK/US/Canada only B I 1 4 8 2 6 9 4 1. About BI 1482694 2. Disease area & mechanism of action 3. Development status 4. Data overview 1. ABOUT BI 1482694 BI 1482694* (HM61713**)
For journalists outside UK/US/Canada only B I 1 4 8 2 6 9 4 1. About BI 1482694 2. Disease area & mechanism of action 3. Development status 4. Data overview 1. ABOUT BI 1482694 BI 1482694* (HM61713**)
Clinical activity of BLU-285, a highly potent and selective KIT/PDGFRα inhibitor designed to treat gastrointestinal stromal tumor (GIST)
 Clinical activity of BLU-285, a highly potent and selective KIT/PDGFRα inhibitor designed to treat gastrointestinal stromal tumor (GIST) Michael Heinrich 1, Robin Jones 2, Margaret von Mehren 3, Patrick
Clinical activity of BLU-285, a highly potent and selective KIT/PDGFRα inhibitor designed to treat gastrointestinal stromal tumor (GIST) Michael Heinrich 1, Robin Jones 2, Margaret von Mehren 3, Patrick
