Pipe Cleaners in the Pipeline: Mechanisms of action for novel cardiovascular drugs on the clinical horizon
|
|
|
- Myrtle Todd
- 5 years ago
- Views:
Transcription
1 Cardiovascular Pipeline in Decline Pipe Cleaners in the Pipeline: Mechanisms of action for novel cardiovascular drugs on the clinical horizon Matthew J. Campen, Ph.D., M.S.P.H. UNM Regents Professor of Pharmaceutical Sciences Reasons for Candidate Failure JACC Basic Transl Sci. 1, ; 2016 Heart Failure: Early Stages Novel Drugs by Disease Class Hypertension and Heart Failure Entresto Ularitide Hyperlipidemia Mipomersen MTTP Inhibitors PCSK9 Inhibitors Heart Failure: Early Stages Compensatory Mechanisms are progressively activated: Increased Sympathetic Activity Improves cardiac performance Fluid Retention (RAAS activation) Increases volume return to the heart Myocardial Hypertrophy Stimulated by Angiotensin II, epinephrine Entresto (Valsartan/Sacubitril) Dual-acting Angiotensin Receptor Blocker (valsartan) plus Neprilysin inhibitor (sacubitril) Sacubitril is a prodrug that is hydrolyzed to active inhibitor Entresto is approved (2015) for NYHA II-IV heart failure patients with reduced ejection fraction (HFrEF) 1
2 Percent Baseline AT-II Pressure Response 6/19/2017 Angiotensin II AT 1 Receptor Blockers (ARB) 1. Angiotensin II can also be formed directly from angiotensin I and angiotensinogen without going through ACE pathway. Neprilysin (Neutral Endopeptidase) 2. AT 1 receptor vasoconstriction, sympathetic activation, cell growth, sodium and fluid retention. 3. AT 2 receptor vasodilation & inhibition of cell growth 4. Blockage of AT1 Inactive Metabolites a. feedback inhibition on renin b. levels of plasma renin angiotensin II c. stimulation of AT2 & vasodilation. Campbell. Nature Reviews Cardiology 14, , 2017 Preclinical Data Supporting the Value of a Neprilysin Inhibitor and ARB in Reducing Blood Pressure PARADIGM-HF Trial Reported Clear Benefits of Entresto Compared to ACEi, Alone Vehicle Telmisartan (0.1 mg/kg) Candoxatril (10 mg/kg) Telmi + Candox ( mg/kg) Time (Hour) McMurray et al., NEJM, 371: , 2014 Decompensation in Heart Failure Entresto (Valsartan/Sacubitril) Side effects are consistent with an ARB Angioedema Hypotension Hyperkalemia Renal failure In the PARADIGM-HF clinical trial, discontinuation due to side effects of Entresto was 10%, compared to 12% with enalapril 2
3 Management of Acute Decompensation Evidence for Congestion Orthopnea High venous pressure Distended jugular vein Edema (WET) Ascites (fluid in abdomen) Valsalva Square Wave Abdominal-Jugular reflex Evidence for Low Perfusion Narrow pulse pressure Cool extremities Sleepiness ACEi hypotension Declining serum Na + Renal dysfunction Urodilatin Ularitide (Cardiorentis AG) Atrial Natriuretic Peptide analogue Indicated for acute cardiac failure (decompensation) Selective venous vasodilator Reduces cardiac wall stress Reduces pulmonary congestion and associated sympotoms Ularitide Benefits Short-Term Symptomology in Acute Cardiac Decompensation; No Long-Term Benefits Urodilatin Ularitide (Cardiorentis AG) Acts on a specific receptor (NPR-A) Activates particulate guanylyl cyclase to produce cyclic GMP cgmp stimulates vasodilation (venous) and diuresis (kidney) Anker et al., Eur Heart J. 36: , Packer et al, NEJM. 376: , 2017 Components of an Advanced, Vulnerable Arterial Plaque Uptake, Processing and Fate of Cholesterol and Lipids Glossary: LPL = lipoprotein lipase HL = hepatic lipase FFA = free fatty acids 3
4 Cholesterol Feedback Signaling Mipomersen (Kynamro) 1 st in class injectable 2 nd gen antisense oligo linked by phosphorothioate instead of phosphodiester Sugar molecules are also modified Inhibits ApoB mrna from transcribing FDA approved for homozygous familial hypercholesterolemia (Genzyme via ISIS) Other antisense oligos and microrna are in development for various targets Action of Antisense Oligos Bays. Clinical Lipidology, 44: , 2009 Thomas et al., JACC. 62: , 2013 Mipomersen Reversibly Reduces LDL-C, Lp(a) and ApoB Mipomersen Reversibly Elevates Serum ALT, Interacts with Statins May interact with statins Risk of liver fat accumulation because VLDL production/secreyion is blocked Thomas et al., JACC. 62: , 2013 Thomas et al., JACC. 62: ,
5 Novel Pathways: MTTP Inhibition Novel Pathways: MTTP Inhibition Microsomal triglyceride transfer protein (MTTP) is essential for transferring triglycerides to: ApoB48 in enterocytes ApoB100 in Hepatocytes Inhibition of MTTP therefore blocks production of Chylomicrons and VLDL Lomitapide (Juxtapid) is an MTTP inhibitor CYP3A4 metabolized; GI effects are most commonly reported side effect Multicenter, double-blind, 12-week trial, included 84 patients with hypercholesterolemia. Randomly assigned ezetimibe 10 mg daily (n = 29) Lopitamide (AEGR) 5 mg daily for the first 4 weeks, 7.5 mg daily for the second 4 weeks and 10 mg daily for the last 4 weeks (n = 28) Ezetimibe 10 mg daily and AEGR-733 administered with the dose titration described above (n = 28) Lopitamide efficacy comparable to ezetimibe, slightly more adverse events (but mild and considered safe) Approved in 2012 Samaha et al., Nature Clinical Practice Cardiovascular Medicine 2008 Novel Pathways: PCSK9 inhibition Proprotein convertase subtilisin/kexin type 9 Binds to LDLR and induces endosomal uptake and degradation Reduced LDLR=increased circulating LDL PCSK9 antibodies are being tested as therapeutics: alirocumab, Praluent (Aventis/Regeneron) evolocumab, Repatha TM (Amgen) bococizumab (Phase 3) Approved for heterozygous familial hypercholesterolemia clinical atherosclerotic cardiovascular disease who require additional lowering of LDL-C PCSK9 Diagram Betteridge. Nature Reviews Endocrinology 9:76-78, Calculated LDL Cholesterol Levels over Time (Intention-to-Treat Analysis). Adverse Events of Interest and Laboratory Values: Safety Analysis. Robinson JG et al. N Engl J Med 2015;372: Robinson JG et al. N Engl J Med 2015;372:
6 Cumulative Incidence of Cardiovascular Events. HDL Good Cholesterol HDL are protective lipoproteins that decrease the risk of CHD HDL participates in reverse cholesterol transport, the process by which excess cholesterol is acquired from cells and transferred to the liver for excretion HDL also has putative anti-inflammatory, antioxidative, platelet antiaggregatory, anticoagulant, and profibrinolytic activities HDL contains the antioxidant enzymes Paraoxonase PAF acetylhydrolase Glutathione peroxidase Sabatine MS et al. N Engl J Med 2015;372: HDL Physiological Role HDL -Three proposed beneficial effects: 1. Increases reverse cholesterol transport. 2. Enhances NO and PG effects on smooth muscle and platelets 3. Inhibits platelet and monocyte binding to damaged endothelium cells. CETP: cholesteryl ester transfer protein Besler et al., EMBO Molecular Medicine, 4: , 2012 ILLUMINATE Study Enrolled >15k patients with coronary artery disease Compared Torcetrapib + Atorvastatin vs atorvastatin alone Mean increase of 72.1% in high-density lipoprotein cholesterol (HDL-C) Mean decrease of 24.9% in LDL-C Small mean decrease of 9% in triglycerides compared with baseline (all P <.001 vs atorvastatin only). Significant in Blood Pressure (mean = 5.4 mmhg) Significant in mortality Dalcetrapib Hoffman LaRoche Dal-OUTCOMES Trial HDL increased, but no clinical benefit NEJM 367: ,
7 DEFINE Trial (Determining the Efficacy and Tolerability of CETP Inhibition with Anacetrapib) Anacetrapib Merck CETP inhibitor Increase HDL 138% Decreased LDL 36% No increase in BP or adverse events Will HDL Targeting Lead to Clinical Benefits? HDL is not a simple molecule, but rather a complex subcellular particle with lipids and proteins with diverse functions Oxidative environments can impair those functions and actually make HDL a pro-inflammatory lipoparticle Proteomic, lipidomic studies are ongoing to better understand the link to CVD Currently overall cholesterol efflux, as a functional marker, is the best link between CVD and HDL Besler et al., EMBO Molecular Medicine, 4: , 2012 Cholesterol Efflux Capacity Summary While the cardiovascular drug pipeline is declining, novel drugs continue to achieve FDA approval New technologies (antisense oligos, antibodies) will play a larger role due to specificity of targets, but will also face challenges related to route of administration and bioavailability. Identification of novel pathways, such as MTTP and PCSK9, will be crucial for maintaining a healthy drug discovery pipeline Traditional targets and biomarkers will change as we get new information from big data GWAS and omic approaches The future ain t what it used to be. Yogi Berra Khera and Rader. ATVB, 33: ,
2017 Summer MAOFP Update
 2017 Summer MAOFP Update. Cardiology Update 2017 Landmark Trials Change Practice Guidelines David J. Strobl, DO, FNLA Heart Failure: Epidemiology More than 4 million patients affected 400,000 new cases
2017 Summer MAOFP Update. Cardiology Update 2017 Landmark Trials Change Practice Guidelines David J. Strobl, DO, FNLA Heart Failure: Epidemiology More than 4 million patients affected 400,000 new cases
Entresto Development of sacubitril/valsartan (LCZ696) for the treatment of heart failure with reduced ejection fraction
 Cardio-Metabolic Franchise Entresto Development of sacubitril/valsartan (LCZ696) for the treatment of heart failure with reduced ejection fraction Randy L Webb, PhD Rutgers Workshop October 21, 2016 Heart
Cardio-Metabolic Franchise Entresto Development of sacubitril/valsartan (LCZ696) for the treatment of heart failure with reduced ejection fraction Randy L Webb, PhD Rutgers Workshop October 21, 2016 Heart
Lipid Therapy: Statins and Beyond. Ivan Anderson, MD RIHVH Cardiology
 Lipid Therapy: Statins and Beyond Ivan Anderson, MD RIHVH Cardiology Outline The cholesterol hypothesis and lipid metabolism The Guidelines 4 Groups that Benefit from Lipid therapy Initiation and monitoring
Lipid Therapy: Statins and Beyond Ivan Anderson, MD RIHVH Cardiology Outline The cholesterol hypothesis and lipid metabolism The Guidelines 4 Groups that Benefit from Lipid therapy Initiation and monitoring
Juxtapid. Juxtapid (lomitapide) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Juxtapid Page: 1 of 6 Last Review Date: September 20, 2018 Juxtapid Description Juxtapid (lomitapide)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Juxtapid Page: 1 of 6 Last Review Date: September 20, 2018 Juxtapid Description Juxtapid (lomitapide)
Drug Class Monograph
 Drug Class Monograph Class: Proprotein Convertase Subtilisin/Kexin Type 9 (PCSK9) Inhibitor Drugs: Praluent (alirocumab), Repatha (evolocumab) Line of Business: Medi-Cal Effective Date: February 17, 2016
Drug Class Monograph Class: Proprotein Convertase Subtilisin/Kexin Type 9 (PCSK9) Inhibitor Drugs: Praluent (alirocumab), Repatha (evolocumab) Line of Business: Medi-Cal Effective Date: February 17, 2016
Update in Cardiology What s Hot in 2017?
 Update in Cardiology What s Hot in 2017? Mark R. Milunski, MD, FACC, FACP Chief, Cardiology Section Orlando Veterans Affairs Medical Center Associate Professor of Medicine University of Central Florida
Update in Cardiology What s Hot in 2017? Mark R. Milunski, MD, FACC, FACP Chief, Cardiology Section Orlando Veterans Affairs Medical Center Associate Professor of Medicine University of Central Florida
Cardiovascular Controversies: Emerging Therapies for Lowering Cardiovascular Risk
 Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Disclosure of Relationships
 Disclosure of Relationships Over the past 12 months Dr Ruilope has served as Consultant and Speakers Bureau member of Astra-Zeneca, Bayer, Daiichi-Sankyo, Menarini, Novartis, Otsuka, Pfizer, Relypsa, Servier
Disclosure of Relationships Over the past 12 months Dr Ruilope has served as Consultant and Speakers Bureau member of Astra-Zeneca, Bayer, Daiichi-Sankyo, Menarini, Novartis, Otsuka, Pfizer, Relypsa, Servier
Marshall Tulloch-Reid, MD, MPhil, DSc, FACE Epidemiology Research Unit Tropical Medicine Research Institute The University of the West Indies, Mona,
 Marshall Tulloch-Reid, MD, MPhil, DSc, FACE Epidemiology Research Unit Tropical Medicine Research Institute The University of the West Indies, Mona, Jamaica At the end of this presentation the participant
Marshall Tulloch-Reid, MD, MPhil, DSc, FACE Epidemiology Research Unit Tropical Medicine Research Institute The University of the West Indies, Mona, Jamaica At the end of this presentation the participant
Lipid Lowering in Patients at High Risk for Cardiovascular Disease
 Lipid Lowering in Patients at High Risk for Cardiovascular Disease Prof. John J.P. Kastelein, MD PhD FESC Dept. of Vascular Medicine Academic Medical Center / University of Amsterdam The Netherlands Novel
Lipid Lowering in Patients at High Risk for Cardiovascular Disease Prof. John J.P. Kastelein, MD PhD FESC Dept. of Vascular Medicine Academic Medical Center / University of Amsterdam The Netherlands Novel
PCSK9 Inhibitors Current Status
 PCSK9 Inhibitors Current Status Ryan T. Whitney, MD FACC Bryan Heart Spring Conference 2016 Disclosures, Conflicts, and Nefarious Connections I own no stock in the companies mentioned in this talk. I am
PCSK9 Inhibitors Current Status Ryan T. Whitney, MD FACC Bryan Heart Spring Conference 2016 Disclosures, Conflicts, and Nefarious Connections I own no stock in the companies mentioned in this talk. I am
PCSK9 Agents Drug Class Prior Authorization Protocol
 PCSK9 Agents Drug Class Prior Authorization Protocol Line of Business: Medicaid P & T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has been developed through review of medical
PCSK9 Agents Drug Class Prior Authorization Protocol Line of Business: Medicaid P & T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has been developed through review of medical
PCSK9 Inhibitors Current Status
 PCSK9 Inhibitors Current Status Ryan T. Whitney, MD FACC Bryan Heart Fall Conference 2015 Disclosures, Conflicts, and Nefarious Connections I own no stock in the companies mentioned in this talk. I am
PCSK9 Inhibitors Current Status Ryan T. Whitney, MD FACC Bryan Heart Fall Conference 2015 Disclosures, Conflicts, and Nefarious Connections I own no stock in the companies mentioned in this talk. I am
Kynamro. Kynamro (mipomersen) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.40.02 Subject: Kynamro Page: 1 of 5 Last Review Date: September 15, 2017 Kynamro Description Kynamro
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.40.02 Subject: Kynamro Page: 1 of 5 Last Review Date: September 15, 2017 Kynamro Description Kynamro
PCSK9 Inhibitors and Modulators
 PCSK9 Inhibitors and Modulators Pam R. Taub MD, FACC Director of Step Family Cardiac Rehabilitation and Wellness Center Associate Professor of Medicine UC San Diego Health System Disclosures Speaker s
PCSK9 Inhibitors and Modulators Pam R. Taub MD, FACC Director of Step Family Cardiac Rehabilitation and Wellness Center Associate Professor of Medicine UC San Diego Health System Disclosures Speaker s
Treating HF Patients with ARNI s Why, When and How?
 Treating HF Patients with ARNI s Why, When and How? 19 th Annual San Diego Heart Failure Symposium for Primary Care Physicians January 11-12, 2019 La Jolla, CA Barry Greenberg M.D. Distinguished Professor
Treating HF Patients with ARNI s Why, When and How? 19 th Annual San Diego Heart Failure Symposium for Primary Care Physicians January 11-12, 2019 La Jolla, CA Barry Greenberg M.D. Distinguished Professor
Modern Lipid Management:
 Modern Lipid Management: New Drugs, New Targets, New Hope Kirk U. Knowlton, M.D Director of Cardiovascular Research Co Chief of Cardiology Why lower LDL C in those without evidence of CAD (primary prevention)
Modern Lipid Management: New Drugs, New Targets, New Hope Kirk U. Knowlton, M.D Director of Cardiovascular Research Co Chief of Cardiology Why lower LDL C in those without evidence of CAD (primary prevention)
Overview & Update on the Utilization of the Natriuretic Peptides in Heart Failure
 June 28, 2016 Overview & Update on the Utilization of the Natriuretic Peptides in Heart Failure Linda C. Rogers, PhD, DABCC, FACB. Agenda Overview of the Natriuretic Peptides and Efficacy studies Similarities
June 28, 2016 Overview & Update on the Utilization of the Natriuretic Peptides in Heart Failure Linda C. Rogers, PhD, DABCC, FACB. Agenda Overview of the Natriuretic Peptides and Efficacy studies Similarities
ADMINISTRATIVE POLICY AND PROCEDURE
 ADMINISTRATIVE POLICY PROCEDURE Policy #: Subject: PCSK9 INHIBITS (ex: Repatha) Section: Care Management Effective Date: January 1, 2015 Revision Date(s): NA Review Date(s): NA Responsible Parties: Patryce
ADMINISTRATIVE POLICY PROCEDURE Policy #: Subject: PCSK9 INHIBITS (ex: Repatha) Section: Care Management Effective Date: January 1, 2015 Revision Date(s): NA Review Date(s): NA Responsible Parties: Patryce
Joslin Diabetes Center Advances in Diabetes and Thyroid Disease 2013 Consensus and Controversy in Diabetic Dyslipidemia
 Consensus and Controversy in Diabetes and Dyslipidemia Om P. Ganda MD Director, Lipid Clinic Joslin diabetes Center Boston, MA, USA CVD Outcomes in DM vs non- DM 102 Prospective studies; 698, 782 people,
Consensus and Controversy in Diabetes and Dyslipidemia Om P. Ganda MD Director, Lipid Clinic Joslin diabetes Center Boston, MA, USA CVD Outcomes in DM vs non- DM 102 Prospective studies; 698, 782 people,
PCSK9 INHIBITORS, ARNIS, FUNNY CHANNEL INHIBITORS: ARE YOU SURE THESE ARE REALLY CARDIAC DRUGS?
 PCSK9 INHIBITORS, ARNIS, FUNNY CHANNEL INHIBITORS: ARE YOU SURE THESE ARE REALLY CARDIAC DRUGS? ALLORIE SMITH, PHARM.D. CARDIOVASCULAR UPDATE CONFERENCE FEBRUARY 9, 2012 OBJECTIVES Explain the pharmacology
PCSK9 INHIBITORS, ARNIS, FUNNY CHANNEL INHIBITORS: ARE YOU SURE THESE ARE REALLY CARDIAC DRUGS? ALLORIE SMITH, PHARM.D. CARDIOVASCULAR UPDATE CONFERENCE FEBRUARY 9, 2012 OBJECTIVES Explain the pharmacology
ARxCH. Annual Review of Changes in Healthcare. Entresto: An Overview for Pharmacists
 Entresto: An Overview for Pharmacists David Comshaw, PharmD Candidate 2019 1 Gyen Musgrave, PharmD Candidate 2019 1 Suzanne Surowiec, PharmD, BCACP 1 Jason Guy, PharmD 1 1 University of Findlay College
Entresto: An Overview for Pharmacists David Comshaw, PharmD Candidate 2019 1 Gyen Musgrave, PharmD Candidate 2019 1 Suzanne Surowiec, PharmD, BCACP 1 Jason Guy, PharmD 1 1 University of Findlay College
REPATHA (PCSK9 INHIBITORS)
 REPATHA (PCSK9 INHIBITS) Indications: PCSK9 Inhibitors are indicated for treatment of adults with heterozygous familial hypercholesterolemia (HeFH) or clinical atherosclerotic cardiovascular disease as
REPATHA (PCSK9 INHIBITS) Indications: PCSK9 Inhibitors are indicated for treatment of adults with heterozygous familial hypercholesterolemia (HeFH) or clinical atherosclerotic cardiovascular disease as
Request for Prior Authorization for PCSK9 inhibitor therapy Website Form Submit request via: Fax
 Request for Prior Authorization for PCSK9 inhibitor therapy Website Form www.highmarkhealthoptions.com Submit request via: Fax - 1-855-476-4158 PCSK9 is a protein that reduces the hepatic removal of low-density
Request for Prior Authorization for PCSK9 inhibitor therapy Website Form www.highmarkhealthoptions.com Submit request via: Fax - 1-855-476-4158 PCSK9 is a protein that reduces the hepatic removal of low-density
Copyright 2011, 2007 by Mosby, Inc., an affiliate of Elsevier Inc. Normal Cardiac Anatomy
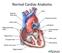 Mosby,, an affiliate of Elsevier Normal Cardiac Anatomy Impaired cardiac pumping Results in vasoconstriction & fluid retention Characterized by ventricular dysfunction, reduced exercise tolerance, diminished
Mosby,, an affiliate of Elsevier Normal Cardiac Anatomy Impaired cardiac pumping Results in vasoconstriction & fluid retention Characterized by ventricular dysfunction, reduced exercise tolerance, diminished
Accumulating Clinical data on PCSK9 Inhibition: Key Lessons and Challenges
 ESC 2015 London Accumulating Clinical data on PCSK9 Inhibition: Key Lessons and Challenges Paul M Ridker, MD, MPH Eugene Braunwald Professor of Medicine Harvard Medical School Director, Center for Cardiovascular
ESC 2015 London Accumulating Clinical data on PCSK9 Inhibition: Key Lessons and Challenges Paul M Ridker, MD, MPH Eugene Braunwald Professor of Medicine Harvard Medical School Director, Center for Cardiovascular
EVOLOCUMAB Generic Brand HICL GCN Exception/Other EVOLOCUMAB REPATHA 42378
 Generic Brand HICL GCN Exception/Other EVOLOCUMAB REPATHA 42378 This drug requires a written request for prior authorization. All requests for Repatha (evolocumab) require review by a pharmacist prior
Generic Brand HICL GCN Exception/Other EVOLOCUMAB REPATHA 42378 This drug requires a written request for prior authorization. All requests for Repatha (evolocumab) require review by a pharmacist prior
Heart Failure. Subjective SOB (shortness of breath) Peripheral edema. Orthopnea (2-3 pillows) PND (paroxysmal nocturnal dyspnea)
 Pharmacology I. Definitions A. Heart Failure (HF) Heart Failure Ezra Levy, Pharm.D. HF Results when one or both ventricles are unable to pump sufficient blood to meet the body s needs There are 2 types
Pharmacology I. Definitions A. Heart Failure (HF) Heart Failure Ezra Levy, Pharm.D. HF Results when one or both ventricles are unable to pump sufficient blood to meet the body s needs There are 2 types
The inhibition of CETP: From simply raising HDL-c to promoting cholesterol efflux and lowering of atherogenic lipoproteins Prof Dr J Wouter Jukema
 The inhibition of CETP: From simply raising HDL-c to promoting cholesterol efflux and lowering of atherogenic lipoproteins Prof Dr J Wouter Jukema Dept Cardiology, Leiden University Medical Center, Leiden,
The inhibition of CETP: From simply raising HDL-c to promoting cholesterol efflux and lowering of atherogenic lipoproteins Prof Dr J Wouter Jukema Dept Cardiology, Leiden University Medical Center, Leiden,
Clinical Policy: Mipomersen (Kynamro) Reference Number: ERX.SPMN.186 Effective Date: 01/2017
 Clinical Policy: (Kynamro) Reference Number: ERX.SPMN.186 Effective Date: 01/2017 Last Review Date: Revision Log See Important Reminder at the end of this policy for important regulatory and legal information.
Clinical Policy: (Kynamro) Reference Number: ERX.SPMN.186 Effective Date: 01/2017 Last Review Date: Revision Log See Important Reminder at the end of this policy for important regulatory and legal information.
Clinical Policy: Lomitapide (Juxtapid) Reference Number: ERX.SPA.170 Effective Date:
 Clinical Policy: (Juxtapid) Reference Number: ERX.SPA.170 Effective Date: 01.11.17 Last Review Date: 11.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Juxtapid) Reference Number: ERX.SPA.170 Effective Date: 01.11.17 Last Review Date: 11.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
ACCP Cardiology PRN Journal Club
 ACCP Cardiology PRN Journal Club Announcements Next journal club Thursday, Dec. 14 th at 3:00 PM EST PACIFY Trial Effects of IV Fentanyl on Ticagrelor Absorption and Platelet Inhibition Among Patients
ACCP Cardiology PRN Journal Club Announcements Next journal club Thursday, Dec. 14 th at 3:00 PM EST PACIFY Trial Effects of IV Fentanyl on Ticagrelor Absorption and Platelet Inhibition Among Patients
Disclosures for Presenter
 A Comparison of Angiotensin Receptor- Neprilysin Inhibition (ARNI) With ACE Inhibition in the Long-Term Treatment of Chronic Heart Failure With a Reduced Ejection Fraction Milton Packer, John J.V. McMurray,
A Comparison of Angiotensin Receptor- Neprilysin Inhibition (ARNI) With ACE Inhibition in the Long-Term Treatment of Chronic Heart Failure With a Reduced Ejection Fraction Milton Packer, John J.V. McMurray,
CETP inhibition: pros and cons. Philip Barter The Heart Research Institute Sydney, Australia
 CETP inhibition: pros and cons Philip Barter The Heart Research Institute Sydney, Australia Philip Barter Disclosures Received honorariums for lectures, consultancies or membership of advisory boards from:
CETP inhibition: pros and cons Philip Barter The Heart Research Institute Sydney, Australia Philip Barter Disclosures Received honorariums for lectures, consultancies or membership of advisory boards from:
Repatha. Repatha (evolocumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.16.08 Subject: Repatha Page: 1 of 8 Last Review Date: September 18, 2015 Repatha Description Repatha
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.16.08 Subject: Repatha Page: 1 of 8 Last Review Date: September 18, 2015 Repatha Description Repatha
LCZ696: LA NUOVA RIVOLUZIONE NELLA TERAPIA DELLO SCOMPENSO CARDIACO. Dario Leosco Università di Napoli Federico II
 LCZ696: LA NUOVA RIVOLUZIONE NELLA TERAPIA DELLO SCOMPENSO CARDIACO Dario Leosco Università di Napoli Federico II Projected changes in cardiovascular diseases CVD Deaths Increase 33% CVD DALYS 22% CAD
LCZ696: LA NUOVA RIVOLUZIONE NELLA TERAPIA DELLO SCOMPENSO CARDIACO Dario Leosco Università di Napoli Federico II Projected changes in cardiovascular diseases CVD Deaths Increase 33% CVD DALYS 22% CAD
ANGIOTENSIN RECEPTOR-NEPRILYSIN INHIBITORS IN HEART FAILURE FROM CHD
 ANGIOTENSIN RECEPTOR-NEPRILYSIN INHIBITORS IN HEART FAILURE FROM CHD Karen Stout, MD FACC Professor, Medicine/Pediatrics University of Washington Seattle, WA USA No disclosures Case 35 year old man with
ANGIOTENSIN RECEPTOR-NEPRILYSIN INHIBITORS IN HEART FAILURE FROM CHD Karen Stout, MD FACC Professor, Medicine/Pediatrics University of Washington Seattle, WA USA No disclosures Case 35 year old man with
PCSK9 INHIBITORS NEW DRUGS IN CARDIOLOGY 7/6/ ANNUAL MEETING OBJECTIVES PCSK9 AND HYPERCHOLESTEROLEMIA PRESENTATION OUTLINE
 NEW DRUGS IN CARDIOLOGY LORI FIALLO, PHARMD.,BCPS AQ-CARDIOLOGY PCSK9 INHIBITORS OBJECTIVES Identify recent FDA approvals in cardiology Describe the impact of recent FDA approvals on current practice Design
NEW DRUGS IN CARDIOLOGY LORI FIALLO, PHARMD.,BCPS AQ-CARDIOLOGY PCSK9 INHIBITORS OBJECTIVES Identify recent FDA approvals in cardiology Describe the impact of recent FDA approvals on current practice Design
Pharmacy Management Drug Policy
 SUBJECT: ; Praluent (alirocumab), Repatha (evolocumab) POLICY NUMBER: Pharmacy-61 EFFECTIVE DATE: 8/15 LAST REVIEW DATE: 9/22/2017 If the member s subscriber contract excludes coverage for a specific service
SUBJECT: ; Praluent (alirocumab), Repatha (evolocumab) POLICY NUMBER: Pharmacy-61 EFFECTIVE DATE: 8/15 LAST REVIEW DATE: 9/22/2017 If the member s subscriber contract excludes coverage for a specific service
What Role do the New PCSK9 Inhibitors Have in Lipid Lowering Treatment?
 What Role do the New PCSK9 Inhibitors Have in Lipid Lowering Treatment? Jennifer G. Robinson, MD, MPH Professor, Departments of Epidemiology & Medicine Director, Prevention Intervention Center University
What Role do the New PCSK9 Inhibitors Have in Lipid Lowering Treatment? Jennifer G. Robinson, MD, MPH Professor, Departments of Epidemiology & Medicine Director, Prevention Intervention Center University
Juxtapid (lomitapide)
 Juxtapid (lomitapide) Policy Number: 5.01.599 Last Review: 06/2018 Origination: 07/2015 Next Review: 06/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage for Juxtapid
Juxtapid (lomitapide) Policy Number: 5.01.599 Last Review: 06/2018 Origination: 07/2015 Next Review: 06/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage for Juxtapid
Familial Hypercholesterolemia
 Familial Hypercholesterolemia Dr.Ramzi Al-Mohammadi Assistant Professor of Medicine Interventional Cardiologist, Advanced HF and Transplant Consultant Classification of Hyperlipedemia Primary hyperlipedemia:
Familial Hypercholesterolemia Dr.Ramzi Al-Mohammadi Assistant Professor of Medicine Interventional Cardiologist, Advanced HF and Transplant Consultant Classification of Hyperlipedemia Primary hyperlipedemia:
REVIEW ARTICLE. Sacubitril/valsartan Use for the Hospitalist Mitchell Padkins 1, James Hart 1, Rachel Littrell 2
 Sacubitril/valsartan Use for the Hospitalist Mitchell Padkins 1, James Hart 1, Rachel Littrell 2 1 University of Missouri School of Medicine, Columbia, MO 2 Division of Cardiovascular Medicine, Department
Sacubitril/valsartan Use for the Hospitalist Mitchell Padkins 1, James Hart 1, Rachel Littrell 2 1 University of Missouri School of Medicine, Columbia, MO 2 Division of Cardiovascular Medicine, Department
LXIV: DRUGS: 4. RAS BLOCKADE
 LXIV: DRUGS: 4. RAS BLOCKADE ACE Inhibitors Components of RAS Actions of Angiotensin i II Indications for ACEIs Contraindications RAS blockade in hypertension RAS blockade in CAD RAS blockade in HF Limitations
LXIV: DRUGS: 4. RAS BLOCKADE ACE Inhibitors Components of RAS Actions of Angiotensin i II Indications for ACEIs Contraindications RAS blockade in hypertension RAS blockade in CAD RAS blockade in HF Limitations
Disclosures. This speaker has indicated there are no relevant financial relationships to be disclosed.
 Disclosures This speaker has indicated there are no relevant financial relationships to be disclosed. And the Beat Goes On: New Medications for Heart Failure Alison M. Walton, PharmD, BCPS The Case of
Disclosures This speaker has indicated there are no relevant financial relationships to be disclosed. And the Beat Goes On: New Medications for Heart Failure Alison M. Walton, PharmD, BCPS The Case of
Clinical Policy: Evolocumab (Repatha) Reference Number: ERX.SPMN.184 Effective Date: 01/2017
 Clinical Policy: (Repatha) Reference Number: ERX.SPMN.184 Effective Date: 01/2017 Last Review Date: Revision Log See Important Reminder at the end of this policy for important regulatory and legal information.
Clinical Policy: (Repatha) Reference Number: ERX.SPMN.184 Effective Date: 01/2017 Last Review Date: Revision Log See Important Reminder at the end of this policy for important regulatory and legal information.
From PARADIGM-HF to Clinical Practice. Waleed AlHabeeb, MD, MHA Associate Professor of Medicine President of the Saudi Heart Failure Group
 From PARADIGM-HF to Clinical Practice Waleed AlHabeeb, MD, MHA Associate Professor of Medicine President of the Saudi Heart Failure Group PARADIGM-HF: Inclusion Criteria Chronic HF NYHA FC II IV with LVEF
From PARADIGM-HF to Clinical Practice Waleed AlHabeeb, MD, MHA Associate Professor of Medicine President of the Saudi Heart Failure Group PARADIGM-HF: Inclusion Criteria Chronic HF NYHA FC II IV with LVEF
UnitedHealthcare Pharmacy Clinical Pharmacy Programs
 UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2017 P 2063-8 Program Prior Authorization/Medical Necessity Medication Repatha (evolocumab) P&T Approval Date 5/2015, 9/2015, 11/2015,
UnitedHealthcare Pharmacy Clinical Pharmacy Programs Program Number 2017 P 2063-8 Program Prior Authorization/Medical Necessity Medication Repatha (evolocumab) P&T Approval Date 5/2015, 9/2015, 11/2015,
New Approaches to Lower LDL-C
 New Approaches to Lower LDL-C CSIM 27 October 2016 Jacques Genest MD Cardiovascular Health Across the Lifespan Program McGill University Health Center Disclosure J. Genest MD 2016 Advisory Board, Speaker
New Approaches to Lower LDL-C CSIM 27 October 2016 Jacques Genest MD Cardiovascular Health Across the Lifespan Program McGill University Health Center Disclosure J. Genest MD 2016 Advisory Board, Speaker
Repatha. Repatha (evolocumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.40.08 Subject: Repatha Page: 1 of 8 Last Review Date: December 2, 2016 Repatha Description Repatha (evolocumab)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.40.08 Subject: Repatha Page: 1 of 8 Last Review Date: December 2, 2016 Repatha Description Repatha (evolocumab)
ENTRESTO (sacubitril and valsartan) oral tablet
 ENTRESTO (sacubitril and valsartan) oral tablet Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This
ENTRESTO (sacubitril and valsartan) oral tablet Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This
Alirocumab Treatment Effect Did Not Differ Between Patients With and Without Low HDL-C or High Triglyceride Levels in Phase 3 trials
 Alirocumab Treatment Effect Did Not Differ Between Patients With and Without Low HDL-C or High Triglyceride Levels in Phase 3 trials G. Kees Hovingh, 1 Richard Ceska, 2 Michael Louie, 3 Pascal Minini,
Alirocumab Treatment Effect Did Not Differ Between Patients With and Without Low HDL-C or High Triglyceride Levels in Phase 3 trials G. Kees Hovingh, 1 Richard Ceska, 2 Michael Louie, 3 Pascal Minini,
Cholesteryl ester transfer protein inhibitors - what have we learnt? Philip Barter The Heart Research Institute Sydney, Australia
 Cholesteryl ester transfer protein inhibitors - what have we learnt? Philip Barter The Heart Research Institute Sydney, Australia Philip Barter Disclosures Received honorariums for lectures, consultancies
Cholesteryl ester transfer protein inhibitors - what have we learnt? Philip Barter The Heart Research Institute Sydney, Australia Philip Barter Disclosures Received honorariums for lectures, consultancies
Satish K Surabhi, MD.FACC,FSCAI,RPVI Medical Director, Cardiac Cath Labs AnMed Health Heart & Vascular Care
 Satish K Surabhi, MD.FACC,FSCAI,RPVI Medical Director, Cardiac Cath Labs AnMed Health Heart & Vascular Care None Fig. 1. Progression of Heart Failure.With each hospitalization for acute heart failure,
Satish K Surabhi, MD.FACC,FSCAI,RPVI Medical Director, Cardiac Cath Labs AnMed Health Heart & Vascular Care None Fig. 1. Progression of Heart Failure.With each hospitalization for acute heart failure,
B. Patient has not reached the percentage reduction goal with statin therapy
 Managing Cardiovascular Risk: The Importance of Lowering LDL Cholesterol and Reaching Treatment Goals for LDL Cholesterol The Role of the Pharmacist Learning Objectives 1. Review the role of lipid levels
Managing Cardiovascular Risk: The Importance of Lowering LDL Cholesterol and Reaching Treatment Goals for LDL Cholesterol The Role of the Pharmacist Learning Objectives 1. Review the role of lipid levels
Best Lipid Treatments
 Best Lipid Treatments Pam R. Taub MD, FACC Director of Step Family Cardiac Rehabilitation and Wellness Center Associate Professor of Medicine UC San Diego Health System Overview of Talk Review of pathogenesis
Best Lipid Treatments Pam R. Taub MD, FACC Director of Step Family Cardiac Rehabilitation and Wellness Center Associate Professor of Medicine UC San Diego Health System Overview of Talk Review of pathogenesis
DYSLIPIDEMIA. Michael Brändle, Stefan Bilz
 DYSLIPIDEMIA Michael Brändle, Stefan Bilz Cardiovascular risk in patients with DM Current guidelines with emphasis on patients with DM Familial Hypercholesterolemia PCSK9-inhibitors Primary Prevention
DYSLIPIDEMIA Michael Brändle, Stefan Bilz Cardiovascular risk in patients with DM Current guidelines with emphasis on patients with DM Familial Hypercholesterolemia PCSK9-inhibitors Primary Prevention
Combination of renin-angiotensinaldosterone. how to choose?
 Combination of renin-angiotensinaldosterone system inhibitors how to choose? Karl Swedberg Professor of Medicine Sahlgrenska Academy University of Gothenburg karl.swedberg@gu.se Disclosures Research grants
Combination of renin-angiotensinaldosterone system inhibitors how to choose? Karl Swedberg Professor of Medicine Sahlgrenska Academy University of Gothenburg karl.swedberg@gu.se Disclosures Research grants
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Repatha) Reference Number: HIM.PA.SP46 Effective Date: 01.01.18 Last Review Date: Line of Business: Health Insurance Marketplace Revision Log See Important Reminder at the end of this
Clinical Policy: (Repatha) Reference Number: HIM.PA.SP46 Effective Date: 01.01.18 Last Review Date: Line of Business: Health Insurance Marketplace Revision Log See Important Reminder at the end of this
What s New in Heart Failure? Marie-France Gauthier, BSc, PharmD, ACPR Clinical Pharmacist at Montfort Hospital
 What s New in Heart Failure? Marie-France Gauthier, BSc, PharmD, ACPR Clinical Pharmacist at Montfort Hospital Disclosures I have no current or past relationships with commercial entities Learning objectives
What s New in Heart Failure? Marie-France Gauthier, BSc, PharmD, ACPR Clinical Pharmacist at Montfort Hospital Disclosures I have no current or past relationships with commercial entities Learning objectives
Repatha. Repatha (evolocumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.40.08 Subject: Repatha Page: 1 of 9 Last Review Date: September 15, 2017 Repatha Description Repatha
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.40.08 Subject: Repatha Page: 1 of 9 Last Review Date: September 15, 2017 Repatha Description Repatha
New Drugs for Lipid Management
 New Drugs for Lipid Management John Larry, MD Assistant Professor-Clinical Section Chief, OSU East Cardiovascular Medicine Department of Internal Medicine Division of Cardiovascular Medicine The Ohio State
New Drugs for Lipid Management John Larry, MD Assistant Professor-Clinical Section Chief, OSU East Cardiovascular Medicine Department of Internal Medicine Division of Cardiovascular Medicine The Ohio State
New Drugs for Lipid Management
 New Drugs for Lipid Management John Larry, MD Assistant Professor-Clinical Section Chief, OSU East Cardiovascular Medicine Department of Internal Medicine Division of Cardiovascular Medicine The Ohio State
New Drugs for Lipid Management John Larry, MD Assistant Professor-Clinical Section Chief, OSU East Cardiovascular Medicine Department of Internal Medicine Division of Cardiovascular Medicine The Ohio State
Beyond ACE-inhibitors for Heart Failure. Jacob Townsend, MD NCVH Birmingham 2015
 Beyond ACE-inhibitors for Heart Failure Jacob Townsend, MD NCVH Birmingham 2015 % Decrease in Mortality Current Therapy HFrEF 0% Angiotensin receptor blocker ACE inhibitor Beta blocker Mineralocorticoid
Beyond ACE-inhibitors for Heart Failure Jacob Townsend, MD NCVH Birmingham 2015 % Decrease in Mortality Current Therapy HFrEF 0% Angiotensin receptor blocker ACE inhibitor Beta blocker Mineralocorticoid
Repatha. Repatha (evolocumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.40.08 Subject: Repatha Page: 1 of 9 Last Review Date: November 30, 2018 Repatha Description Repatha (evolocumab)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.40.08 Subject: Repatha Page: 1 of 9 Last Review Date: November 30, 2018 Repatha Description Repatha (evolocumab)
Drug Class Prior Authorization Criteria PCSK9 Inhibitors
 Drug Class Prior Authorization Criteria PCSK9 Inhibitors Line of Business: Medicaid P & T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has been developed through review of
Drug Class Prior Authorization Criteria PCSK9 Inhibitors Line of Business: Medicaid P & T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has been developed through review of
Rationale and Practical Aspects of Sacubitril- Valsartan and Ivabradine Use in Heart Failure Patients
 Rationale and Practical Aspects of Sacubitril- Valsartan and Ivabradine Use in Heart Failure Patients Javed Butler, MD, MPH, MBA Patrick H. Lehan Professor of Medicine Professor of Physiology Chairman,
Rationale and Practical Aspects of Sacubitril- Valsartan and Ivabradine Use in Heart Failure Patients Javed Butler, MD, MPH, MBA Patrick H. Lehan Professor of Medicine Professor of Physiology Chairman,
LCZ696 A First-in-Class Angiotensin Receptor Neprilysin Inhibitor
 The Angiotensin Receptor Neprilysin Inhibitor LCZ696 in Heart Failure with Preserved Ejection Fraction The Prospective comparison of ARNI with ARB on Management Of heart failure with preserved ejection
The Angiotensin Receptor Neprilysin Inhibitor LCZ696 in Heart Failure with Preserved Ejection Fraction The Prospective comparison of ARNI with ARB on Management Of heart failure with preserved ejection
Disclosures. Overview. Goal statement. Advances in Chronic Heart Failure Management 5/22/17
 Disclosures Advances in Chronic Heart Failure Management I have nothing to disclose Van N Selby, MD UCSF Advanced Heart Failure Program May 22, 2017 Goal statement To review recently-approved therapies
Disclosures Advances in Chronic Heart Failure Management I have nothing to disclose Van N Selby, MD UCSF Advanced Heart Failure Program May 22, 2017 Goal statement To review recently-approved therapies
Disclosures. Advances in Chronic Heart Failure Management 6/12/2017. Van N Selby, MD UCSF Advanced Heart Failure Program June 19, 2017
 Advances in Chronic Heart Failure Management Van N Selby, MD UCSF Advanced Heart Failure Program June 19, 2017 I have nothing to disclose Disclosures 1 Goal statement To review recently-approved therapies
Advances in Chronic Heart Failure Management Van N Selby, MD UCSF Advanced Heart Failure Program June 19, 2017 I have nothing to disclose Disclosures 1 Goal statement To review recently-approved therapies
Heart Failure: Current Management Strategies
 Heart Failure: Current Management Strategies CSHP Fall Education Session- September 30th, 2017 Carolyn MacKinnon & Tamara Matchett BscPharm, ACPR Candidates Objectives 1. Describe the pathophysiology &
Heart Failure: Current Management Strategies CSHP Fall Education Session- September 30th, 2017 Carolyn MacKinnon & Tamara Matchett BscPharm, ACPR Candidates Objectives 1. Describe the pathophysiology &
Proprotein Convertase Subtilisin/Kexin type 9(PCSK9) Inhibitors Prior Authorization with Quantity Limit Program Summary
 Proprotein Convertase Subtilisin/Kexin type 9(PCSK9) Inhibitors Prior Authorization with Quantity Limit Program Summary Proprotein Convertase Subtilisin/Kexin type 9(PCSK9) Inhibitors Prior Authorization
Proprotein Convertase Subtilisin/Kexin type 9(PCSK9) Inhibitors Prior Authorization with Quantity Limit Program Summary Proprotein Convertase Subtilisin/Kexin type 9(PCSK9) Inhibitors Prior Authorization
Neprilysin Inhibitor (Entresto ) Prior Authorization and Quantity Limit Program Summary
 Neprilysin Inhibitor (Entresto ) Prior Authorization and Quantity Limit Program Summary FDA APPROVED INDICATIONS DOSAGE 1 Indication Entresto Reduce the risk of cardiovascular (sacubitril/valsartan) death
Neprilysin Inhibitor (Entresto ) Prior Authorization and Quantity Limit Program Summary FDA APPROVED INDICATIONS DOSAGE 1 Indication Entresto Reduce the risk of cardiovascular (sacubitril/valsartan) death
Current Cholesterol Guidelines and Treatment of Residual Risk COPYRIGHT. J. Peter Oettgen, MD
 Current Cholesterol Guidelines and Treatment of Residual Risk J. Peter Oettgen, MD Associate Professor of Medicine Harvard Medical School Director, Preventive Cardiology Beth Israel Deaconess Medical Center
Current Cholesterol Guidelines and Treatment of Residual Risk J. Peter Oettgen, MD Associate Professor of Medicine Harvard Medical School Director, Preventive Cardiology Beth Israel Deaconess Medical Center
THE LATEST IN CARDIOVASCULAR RISK LIPID MANAGEMENT
 THE LATEST IN CARDIOVASCULAR RISK LIPID MANAGEMENT Thomas F. Whayne, Jr, MD, PhD, FACC Professor of Medicine (Cardiology) Gill Heart Institute University of Kentucky April 2016 E-mail: twhayn0@uky.edu
THE LATEST IN CARDIOVASCULAR RISK LIPID MANAGEMENT Thomas F. Whayne, Jr, MD, PhD, FACC Professor of Medicine (Cardiology) Gill Heart Institute University of Kentucky April 2016 E-mail: twhayn0@uky.edu
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Proprotein Convertase Subtilisin/kexin type 9 Page 1 of 24 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Proprotein Convertase Subtilisin/kexin type 9 (PCSK9)
Proprotein Convertase Subtilisin/kexin type 9 Page 1 of 24 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Proprotein Convertase Subtilisin/kexin type 9 (PCSK9)
New Cardiovascular Drugs 2015 CAM F. CAMPBELL MD FACC MERCY CARDIOLOGY 2/6/2015
 New Cardiovascular Drugs 2015 CAM F. CAMPBELL MD FACC MERCY CARDIOLOGY 2/6/2015 Disclosures Financial: None Personal: Married, 4 children, 2 grandchildren, 1 grandchild on the way Trivia: I have sailed
New Cardiovascular Drugs 2015 CAM F. CAMPBELL MD FACC MERCY CARDIOLOGY 2/6/2015 Disclosures Financial: None Personal: Married, 4 children, 2 grandchildren, 1 grandchild on the way Trivia: I have sailed
Estimated 5.7 million Americans with HF. 915, 000 new HF cases annually, HF incidence approaches
 Heart Failure: Management of a Chronic Disease Jenny Bauerly RN, CHFN, APRN-BC Heart Failure (HF) Definition A complex clinical syndrome that can result from any structural or functional cardiac disorder
Heart Failure: Management of a Chronic Disease Jenny Bauerly RN, CHFN, APRN-BC Heart Failure (HF) Definition A complex clinical syndrome that can result from any structural or functional cardiac disorder
Long-Term Care Updates
 Long-Term Care Updates July 2015 By Amy Friedman Wilson, PharmD Heart failure (HF) is a clinical condition in which ventricular filling or ejection of blood is structurally or functionally impaired. 1
Long-Term Care Updates July 2015 By Amy Friedman Wilson, PharmD Heart failure (HF) is a clinical condition in which ventricular filling or ejection of blood is structurally or functionally impaired. 1
Prof. John Chapman, MD, PhD, DSc
 Prof. John Chapman, MD, PhD, DSc Director of the Dyslipidemia and Atherosclerosis Research Unit of the National Institute for Health and Medical Research (INSERM) at the Pitié-Salpétrière Hospital in Paris
Prof. John Chapman, MD, PhD, DSc Director of the Dyslipidemia and Atherosclerosis Research Unit of the National Institute for Health and Medical Research (INSERM) at the Pitié-Salpétrière Hospital in Paris
Cardiovascular Protection and the RAS
 Cardiovascular Protection and the RAS Katalin Kauser, MD, PhD, DSc Senior Associate Director, Boehringer Ingelheim Pharmaceutical Inc. Micardis Product Pipeline Scientific Support Ridgefield, CT, USA Cardiovascular
Cardiovascular Protection and the RAS Katalin Kauser, MD, PhD, DSc Senior Associate Director, Boehringer Ingelheim Pharmaceutical Inc. Micardis Product Pipeline Scientific Support Ridgefield, CT, USA Cardiovascular
1/4/18. Heart Failure Guideline Review and Update. Disclosure. Pharmacist Objectives. Pharmacy Technician Objectives. What is Heart Failure?
 Disclosure Heart Failure Guideline Review and Update I have had no financial relationship over the past 12 months with any commercial sponsor with a vested interest in this presentation. Natalie Beiter,
Disclosure Heart Failure Guideline Review and Update I have had no financial relationship over the past 12 months with any commercial sponsor with a vested interest in this presentation. Natalie Beiter,
Metabolism and Atherogenic Properties of LDL
 Metabolism and Atherogenic Properties of LDL Manfredi Rizzo, MD, PhD Associate Professor of Internal Medicine Faculty of Medicine, University of Palermo, Italy & Affiliate Associate Professor of Internal
Metabolism and Atherogenic Properties of LDL Manfredi Rizzo, MD, PhD Associate Professor of Internal Medicine Faculty of Medicine, University of Palermo, Italy & Affiliate Associate Professor of Internal
Clinical Policy: Evolocumab (Repatha) Reference Number: ERX.SPA.169 Effective Date:
 Clinical Policy: (Repatha) Reference Number: ERX.SPA.169 Effective Date: 01.11.17 Last Review Date: 11.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Clinical Policy: (Repatha) Reference Number: ERX.SPA.169 Effective Date: 01.11.17 Last Review Date: 11.17 Revision Log See Important Reminder at the end of this policy for important regulatory and legal
Evolving Concepts on Lipid Management from Ezetimibe (IMPROVE IT) to PCSK9 Inhibitors
 Evolving Concepts on Lipid Management from Ezetimibe (IMPROVE IT) to PCSK9 Inhibitors Sidney C. Smith, Jr. MD, FACC, FAHA, FESC Professor of Medicine/Cardiology University of North Carolina at Chapel Hill
Evolving Concepts on Lipid Management from Ezetimibe (IMPROVE IT) to PCSK9 Inhibitors Sidney C. Smith, Jr. MD, FACC, FAHA, FESC Professor of Medicine/Cardiology University of North Carolina at Chapel Hill
SGK 2016 Session: Postgraduate Course in Heart Failure Lausanne, 15. June 2016 Heart Failure Guidelines 2016
 SGK 2016 Session: Postgraduate Course in Heart Failure Lausanne, 15. June 2016 Heart Failure Guidelines 2016 Matthias Nägele, MD University Hospital Zurich Disclosures I have nothing to disclose. The new
SGK 2016 Session: Postgraduate Course in Heart Failure Lausanne, 15. June 2016 Heart Failure Guidelines 2016 Matthias Nägele, MD University Hospital Zurich Disclosures I have nothing to disclose. The new
Part 1 Risk Factors and Atherosclerosis. LO1. Define the Different Forms of CVD
 Week 3: Cardiovascular Disease Learning Outcomes: 1. Define the difference forms of CVD 2. Describe the various risk factors of CVD 3. Describe atherosclerosis and its stages 4. Describe the role of oxidation,
Week 3: Cardiovascular Disease Learning Outcomes: 1. Define the difference forms of CVD 2. Describe the various risk factors of CVD 3. Describe atherosclerosis and its stages 4. Describe the role of oxidation,
Drug Prior Authorization Guideline PCSK9 Inhibitors -
 Drug Prior Authorization Guideline PCSK9 Inhibitors - REPATHA (evolocumab) PRALUENT (alirocumab) PA9911 Covered Service: Yes when meets criteria below Prior Authorization Required: Yes-as shown below Additional
Drug Prior Authorization Guideline PCSK9 Inhibitors - REPATHA (evolocumab) PRALUENT (alirocumab) PA9911 Covered Service: Yes when meets criteria below Prior Authorization Required: Yes-as shown below Additional
New Winners in the World of Heart Failure. Laura Steffens PharmD Candidate 2016 CICU Presentation August 12, 2015
 New Winners in the World of Heart Failure Laura Steffens PharmD Candidate 2016 CICU Presentation August 12, 2015 Jessup 2014 Shaking Things Up 2003: FDA approved eplerenone for the treatment of heart failure
New Winners in the World of Heart Failure Laura Steffens PharmD Candidate 2016 CICU Presentation August 12, 2015 Jessup 2014 Shaking Things Up 2003: FDA approved eplerenone for the treatment of heart failure
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Proprotein Convertase Subtilisin/kexin type 9 Page 1 of 22 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Proprotein Convertase Subtilisin/kexin type 9 (PCSK9)
Proprotein Convertase Subtilisin/kexin type 9 Page 1 of 22 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: Proprotein Convertase Subtilisin/kexin type 9 (PCSK9)
 http://noodlemaz.wordpress.com/category/science/cancer/ Outline Introduction Serious nature of Cardiovascular Disease (CVD) How to prevent CVD? The disease process Damage and plaque development Current
http://noodlemaz.wordpress.com/category/science/cancer/ Outline Introduction Serious nature of Cardiovascular Disease (CVD) How to prevent CVD? The disease process Damage and plaque development Current
Making War on Cholesterol with New Weapons: How Low Can We/Should We Go? Shaun Goodman
 Making War on Cholesterol with New Weapons: How Low Can We/Should We Go? Shaun Goodman Disclosures Research grant support, speaker/consulting honoraria: Sanofi and Regeneron Including ODYSSEY Outcomes
Making War on Cholesterol with New Weapons: How Low Can We/Should We Go? Shaun Goodman Disclosures Research grant support, speaker/consulting honoraria: Sanofi and Regeneron Including ODYSSEY Outcomes
PCSK9 antibodies: A new therapeutic option for the treatment of hypercholesterolemia
 : 262-267, 2017 Περίληψη Διάλεξης PCSK9 antibodies: A new therapeutic option for the treatment of hypercholesterolemia I. Gouni-Bethold Polyclinic for Endocrinology, Diabetes, and Preventive Medicine University
: 262-267, 2017 Περίληψη Διάλεξης PCSK9 antibodies: A new therapeutic option for the treatment of hypercholesterolemia I. Gouni-Bethold Polyclinic for Endocrinology, Diabetes, and Preventive Medicine University
Praluent. Praluent (alirocumab) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.40.06 Subject: Praluent Page: 1 of 10 Last Review Date: September 20, 2018 Praluent Description Praluent
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.40.06 Subject: Praluent Page: 1 of 10 Last Review Date: September 20, 2018 Praluent Description Praluent
Review of guidelines for management of dyslipidemia in diabetic patients
 2012 international Conference on Diabetes and metabolism (ICDM) Review of guidelines for management of dyslipidemia in diabetic patients Nan Hee Kim, MD, PhD Department of Internal Medicine, Korea University
2012 international Conference on Diabetes and metabolism (ICDM) Review of guidelines for management of dyslipidemia in diabetic patients Nan Hee Kim, MD, PhD Department of Internal Medicine, Korea University
Cardiac Drugs - The Missing Links (Drugs we should have in New Zealand)
 Pharmac: Devices and Drugs Heart drugs the missing links John Elliott Chairs: Rob Doughty & Rajesh Nair Cardiac Drugs - The Missing Links (Drugs we should have in New Zealand) John Elliott University of
Pharmac: Devices and Drugs Heart drugs the missing links John Elliott Chairs: Rob Doughty & Rajesh Nair Cardiac Drugs - The Missing Links (Drugs we should have in New Zealand) John Elliott University of
Heart Failure Clinician Guide JANUARY 2016
 Kaiser Permanente National CLINICAL PRACTICE GUIDELINES Heart Failure Clinician Guide JANUARY 2016 Introduction This evidence-based guideline summary is based on the 2016 National Heart Failure Guideline.
Kaiser Permanente National CLINICAL PRACTICE GUIDELINES Heart Failure Clinician Guide JANUARY 2016 Introduction This evidence-based guideline summary is based on the 2016 National Heart Failure Guideline.
New Trials. Iain Squire. Professor of Cardiovascular Medicine University of Leicester. Chair, BSH
 New Trials Iain Squire Professor of Cardiovascular Medicine University of Leicester Chair, BSH BSH Heart Failure Day for Revalidation and Training 2017 Presentation title: New Trials Speaker: Iain Squire
New Trials Iain Squire Professor of Cardiovascular Medicine University of Leicester Chair, BSH BSH Heart Failure Day for Revalidation and Training 2017 Presentation title: New Trials Speaker: Iain Squire
Lipid/Lipoprotein Structure and Metabolism (Overview)
 Lipid/Lipoprotein Structure and Metabolism (Overview) Philip Barter President, International Atherosclerosis Society Centre for Vascular Research University of New South Wales Sydney, Australia Disclosures
Lipid/Lipoprotein Structure and Metabolism (Overview) Philip Barter President, International Atherosclerosis Society Centre for Vascular Research University of New South Wales Sydney, Australia Disclosures
