Supplementary Figure 1. SybII and Ceb are sorted to distinct vesicle populations in astrocytes. Nature Neuroscience: doi: /nn.
|
|
|
- Lesley Daniel
- 6 years ago
- Views:
Transcription
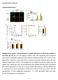
1 Supplementary Figure 1 SybII and Ceb are sorted to distinct vesicle populations in astrocytes.
2 (a) Exemplary images for cultured astrocytes co-immunolabeled with SybII and Ceb antibodies. SybII accumulates at the cell s periphery, whereas Ceb is scattered throughout the cytoplasm. Note the lack of colocalization between the two v-snare proteins as depicted in the higher magnification image. (b) Ceb colocalizes with the large dense core (LDCV) marker protein NPY. (c) Coimmunolabeling of the vesicular glutamate transporter (vglut) and SybII shows a high degree of colocalization between the two proteins. (d) Cytofluorograms (pixel plotted in grey values) for the indicated groups underline the lack of colocalization between SybII and Ceb (r 2 =0,34) and a good colocalization between Ceb and NPY (r 2 =0,84) and SybII and vglut (r 2 =0,94). (e) Line scan analyses for the indicated antigens (dashed lines in a-c). (f) Syb and Ceb colocalize with their respective cargo significantly better than with the other v-snare variant (Data were collected from 3 preparations; SybII vs Ceb, n=12; Manders coeff.: Ceb vs NPY, n=11, p<0.0001; sybii vs vglut, n=12, p<0.0001, one-way ANOVA versus SybII vs Ceb). All data are represented as mean ± s.e.m.
3 Supplementary Figure 2 Antibodies are specific for their respective antigens. (a) Confocal images of sybiiko astrocytes co-immunolabelled with affinity purified antibodies for sybii and ceb. Note that sybii staining is abolished in ko astrocytes. (b) Exemplary images of co-staining with ceb and sybii in cebko astrocytes. Ceb staining is abrogated in cebko cells. (c) Coimmunolabeling of NPY and ceb in NPYko astrocytes. NPY immunoreactivity is abolished in NPYko astrocytes. (d) Line scan analyses for the indicated antigens (dashed lines in a-c).
4 Supplementary Figure 3 Ca 2+ signaling is unaffected in v-snare-deficient astrocytes. (a,b) Confocal high magnification images of a dko astrocyte transfected with either cebmrfp (a) or sybiimrfp (b) and subsequently immunolabeled with NPY. Lentiviral expressed ceb-mrfp and endogenous NPY display a high degree of colocalization (Pearson s coefficient: 0.98±0.02; n=12), whereas sybii-mrfp and NPY show little colocalization (Pearson s coefficient: 0.54±0.001; n=11). (c) Exemplary confocal images of astrocytes loaded with Fluo4AM (10 µm) before (upper image) and
5 after stimulation (lower image) with 1 mm glutamate. (d) Time-course of stimulus dependent increase in Fluo4AM fluorescence in WT, sybiiko, cebko, dko, dko+ceb and dko+sybii. No increase could be observed in WT cells treated with mglur5 antagonist MPEP (20 µm) and unstimulated cells. (e) Quantification of Fluo4AM fluorescence at the signal maximum for all groups in (d). No difference in the maximum fluorescence could be observed for any of the conditions. Note that in the absence of a stimulus or pre-incubation of cells with MPEP a metabotropic glutamate receptor antagonist abolished increase in Fluo4AM (Data were collected from 3 preparations; WT, n=17; sybiiko, n=18, p=0.48; cebko, n=21, p=0.38; dko, n=22, p=0.34; dko+sybii, n=15 p=0.41; dko+ceb, n=19, p=0.42; +MPEP, n=21, p<0.0001; not stim., n=22, p<0.0001, one-way ANOVA vs WT). ***p< All data are represented as mean ± s.e.m.
6 Supplementary Figure 4 iglusnfr monitors glutamate release in astrocytes and neurons. (a) Exemplary image of an astrocyte transfected with iglusnfr during stimulation with 20 µm DHPG. Arrows point to quantal-like glutamate release. Dashed lines indicate the cellular borders of the
7 astrocyte, where they could be visualized. (b) Exemplary traces of single regions of interest in WT, sybiiko and cebko astrocytes. WT and cebko cells respond to DHPG with discrete, quantal-like fluorescent signals monitoring glutamate release. The slowly developing fluorescence offset is likely due to increase in ambient glutamate. No fluorescence increase could be detected in sybiiko astrocytes. (c) Glutamate secretion is abolished in sybiiko, dko astrocytes and in the presence of 50 µm MPEP, but remained unchanged in cebko cells (vs WT, cebko, p=0.996; sybiiko, p<0.001; dko, p<0.001; WT+MPEP, p<0.001; one-way ANOVA). (d) iglusnfr expression is unchanged between the groups (versus WT: cebko, p=0.133; sybiiko, p=0.130; dko, p=0.141; WT+MPEP, p=0.198; one-way ANOVA). (e) The amplitude of the transient glutamate flashes is unaltered between WT and cebko cells (events: WT, 648; cebko, 668; threshold: 5xSD of noise, WT, ±1.3x10-3 ; cebko, ±3.2x10-3 ). Inset displays the average of 10 exemplary events. Bar, 0.1 F/F 0, 320 ms. (f) WT and cebko cells continuously secrete glutamate during DHPG application. Data was collected from 3 preparations; WT, n=13; cebko, n=12; sybiiko, n=13, dko, n=11, WT+MPEP, n=9. (g) Exemplary images of autaptic hippocampal neurons transfected with iglusnfr before (upper panel: F 0 ) and during electrical stimulation (lower panel: F). (h) Representative traces of iglusnfr fluorescence in WT and sybiiko neurons. Top trace indicates the stimulation frequency. Note the lack of fluorescence increase in the sybiiko neurons where glutamatergic signaling is abolished. (i) Mean fluorescence increase upon single action potentials (p= ). (j) Expression level is unchanged in sybiiko neurons. Data was collected from WT, n=9; sybiiko, n=7, p=48 from 2 preparations, student s t-test. ***p< All data are represented as mean ± s.e.m.
8 Supplementary Figure 5 Loss of gliotransmitter secretion differentially affects the pool of readily releasable vesicles and paired pulse ratio. (a) Representative traces of the average postsynaptic response to 5s application of hypertonic solution (500 mm sucrose) from neurons cultured on WT, sybiiko and cebko astrocytes. (b,c) v- SNARE dependent secretion affects the readily releasable pool charge and release probability (RRP, sucrose: sybiiko, p<0.001; cebko, p<0.001; P R : sybiiko, p<0.001; cebko, p=0.77, one-way ANOVA vs WT.). Data was collected from recordings with WT (n=20), sybiiko (n=19) and cebko (n=19) 3 preparations. (d) Normalized EPSCs measured with an inter-stimulation-interval of 50ms. (e) Paired pulse ratio is only increased for neurons grown on sybiiko astrocytes. Data was extracted from the first two EPSCs of the 20 Hz train (amplitude ratio: EPSC2/EPSC1) and collected from 3
9 preparations, WT, n=62; sybiiko, n=28; cebko, n=35; dko, n=29 (ANOVA vs wt: sybiiko, p=0.009; cebko,p=0.43; dko, p=0.5). All data are represented as mean ± s.e.m.
10 Supplementary Figure 6 Spontaneous release is unaffected by extracellular application of NPY and ATP.
11 (a) Representative recordings of quantal signaling from neurons grown on WT or cebko astrocytes treated daily with either the vehicle or 10 nm NPY. (b) mepsc frequency, amplitude and charge are unchanged for the indicated groups (Data were collected from 3 preparations; WT+vehicle, n=24; cebko+vehicle, n=25; cebko+npy, n=26; for mepsc frequency, amplitude and charge all p-values ranged between and 0.935, one-way ANOVA vs WT+vehicle). (c) Exemplary traces of spontaneous mepscs recorded in neurons plated on WT and cebko astrocytes treated daily either with 10 nm NPY/ATP in the medium or a vehicle. (d) mepsc frequency, amplitude and charge are unaffected by NPY/ATP co-application (Data were collected from 3 preparations; WT+vehicle, n=19; cebko+vehicle, n=20; cebko+npy/atp, n=21; for mepsc frequency, amplitude and charge all p- Values ranged between and 0.969, one-way ANOVA vs WT+vehicle). (e) Representative recordings of mepscs from WT neurons grown on WT and cebko astrocytes, treated with 150 nm DPCPX/BIIE0246 (Data were collected from 3 preparations; adenosine A1 and NPY Y2 receptor antagonists; WT+vehicle, n=24; cebko+vehicle, n=19; WT+150 nm, DPCPX/BIIE0246, n=18; cebko+150 nm DPCPX/BIIE0246, n=20). (f) mepsc frequency, amplitude and charge for the conditions described in (e) were unaffected by the treatment (for mepsc frequency, amplitude and charge all p-values ranged between 0.21 and 0.982, one-way ANOVA vs WT+vehicle). All data are represented as mean ± s.e.m.
12 Supplementary Figure 7 Tonic ATP/NPY and glutamate release from astrocytes regulates synaptic transmission.
13 (a) Consecutive trains of APs (20Hz; interval time 1 min) recorded either in the presence of Ringer s solution or ATP/NPY (10 µm). (b,c) Amplitude of the first evoked response and RRP size were significantly reduced in the presence of ATP/NPY in neurons grown on WT and cebko astrocytes. Data was collected from 2 preparations. (Ringer: WT, n=9; amp: p=0,641, RRP, p=1,0; cebko, n=9 amp: p=0,706; RRP, p=0,691; ATP/NPY: WT, n=9; amp: p<0.001; RRP, p<0.001; cebko, n=9; amp: p<0.001, RRP: p<0.001, t-test) (d) Acute application of the A1 antagonist DPCPX and NPY-receptor antagonist BIIE0246 (indicated by the bar) strongly and reversibly augmented the evoked EPSC amplitude (recorded at 0.2 Hz) of neurons grown on WT astrocytes. No effect was seen for neurons plated on cebko astrocytes. Scale bar, 2nA, 20ms (e,f) The EPSC amplitude is specifically increased for WT, but not for cebko astrocytes (WT,DPCPX/BIIW0246: p<0.001; cebko, DPCPX/BIIE0246 p=0,37; t-test). Data were collected from 3 preparations, WT, n=16; cebko, n=14 cells; ***p< (g) Acute application of the kainate receptor antagonist NS-102 (indicated by the bar) strongly suppressed the EPSC amplitude (recorded at 0.2 Hz) in neurons grown on WT astrocytes to levels of cells plated with sybiiko cells. No changes were observed in sybiiko cells; bar, 2nA; 15ms. (h,i) EPSC amplitude is only decreased during NS-102 application in neurons grown on WT astrocytes. NS-102 had no effect on neurons grown on sybiiko cells (WT, NS-102: p<0.001, recovery, p=0,272; sybiiko, NS-102 p=0.419; recovery, p=0.482, t-test). Data was collected from 3 preparations from WT, n=17 and sybiiko, n=19 cells. ***p<0.001, t-test. All data are represented as mean ± s.e.m.
14 Supplementary Figure 8 Loss of distinct secretion pathways in astrocytes modulates the number of readily releasable vesicles. (a) Schematic drawing of the experimental protocol. Neurons were electrically stimulated with the
15 patch pipette (20Hz/2s) while fluorescence changes were monitored at 10Hz simultaneously. At the end of the recording cells were perfused with NH 4 Cl to unquench syn-ph fluorescence. (b) Exemplary recordings of quantal signaling from neurons transfected with Syn-pH grown without glia, on sybiiko or cebko astrocytes. (c) mepsc frequency is strongly reduced in cells grown without or on sybiiko astrocytes (mepsc freq: no glia, p<0.001; sybiiko, p<0.001; cebko, p=0.53; mepsc ampl.: no glia, p=0.79; sybiiko, p=0.82; cebko, p=0.83; mepsc charge: no glia, p=0.73; sybiiko, p=0.76; cebko, p=0.68, one-way ANOVA vs WT) (d) Time course for the decline of the evoked amplitudes. (e,f) v- SNARE mediated astrocytic secretion similarly affects the evoked amplitude (1 st EPSC) and RRP (first EPSC ampl.: no glia, p<0.001; sybiiko, p=0.0031; cebko, p<0.001; RRP charge: no glia, p<0.001; sybiiko, p<0.001; cebko, p<0.001, one-way ANOVA vs WT). Data was collected from 4 preparations; WT, n=20; sybiiko, n=17; cebko, n=16; non-glia, n=9; not stimulated, n=5; **p<0.01, ***p< All data are represented as mean ± s.e.m.
16 Supplementary Figure 9 v-snare-dependent gliotransmitters release does not influence the number of synapses. (a) Exemplary confocal images of autaptic neuronal cultures grown without glia cells or on WT, sybiiko, cebko or dko astrocytes immunolabelled with the presynaptic marker protein anti-bassoon.
17 (b) The number of synapses per microisland is reduced in the absence of astrocytes but not affected by their v-snare deficiency (no glia, p<0.001; sybiiko, p=0.994; cebko, p=0.994; dko, p=0.985; oneway ANOVA vs WT). Data was collected from 3 preparations, WT, n=17; no-glia, n=16; sybiiko, n=19; cebko, n=19; dko, n=19; ***p<0.001 All data are represented as mean ± s.e.m.
18 Supplementary Figure 10 Proposed model for v-snare-dependent gliotransmitter secretion pathways modulating synaptic efficacy. (a) Astrocytes release NPY and ATP through a ceb-dependent secretion pathway. NPY acts on presynaptic NPY Y2 receptors and ATP (rapidly converted into adenosine (ado)) may act on presynaptic adenosine A1 receptors to decrease the RRP. (b) SybII mediated glutamate release from astrocytic SLMVs exerts its potentiating effect of synaptic signaling (increasing the RRP size and P r )
19 through activation of presynaptic kainate receptors (KAR) or NMDA(NR2B) 2 and mglur receptors 6. (a)+(b) In the full protein context of WT cells the antagonistic effects of the v-snare dependent secretion pathways compensate each other (as evidenced by the dko phenotype) to increase the dynamic range of synaptic signaling.
Supplementary Materials for VAMP4 directs synaptic vesicles to a pool that selectively maintains asynchronous neurotransmission
 Supplementary Materials for VAMP4 directs synaptic vesicles to a pool that selectively maintains asynchronous neurotransmission Jesica Raingo, Mikhail Khvotchev, Pei Liu, Frederic Darios, Ying C. Li, Denise
Supplementary Materials for VAMP4 directs synaptic vesicles to a pool that selectively maintains asynchronous neurotransmission Jesica Raingo, Mikhail Khvotchev, Pei Liu, Frederic Darios, Ying C. Li, Denise
Astrocyte signaling controls spike timing-dependent depression at neocortical synapses
 Supplementary Information Astrocyte signaling controls spike timing-dependent depression at neocortical synapses Rogier Min and Thomas Nevian Department of Physiology, University of Berne, Bern, Switzerland
Supplementary Information Astrocyte signaling controls spike timing-dependent depression at neocortical synapses Rogier Min and Thomas Nevian Department of Physiology, University of Berne, Bern, Switzerland
Supporting Information
 ATP from synaptic terminals and astrocytes regulates NMDA receptors and synaptic plasticity through PSD- 95 multi- protein complex U.Lalo, O.Palygin, A.Verkhratsky, S.G.N. Grant and Y. Pankratov Supporting
ATP from synaptic terminals and astrocytes regulates NMDA receptors and synaptic plasticity through PSD- 95 multi- protein complex U.Lalo, O.Palygin, A.Verkhratsky, S.G.N. Grant and Y. Pankratov Supporting
SUPPLEMENTARY INFORMATION. Supplementary Figure 1
 SUPPLEMENTARY INFORMATION Supplementary Figure 1 The supralinear events evoked in CA3 pyramidal cells fulfill the criteria for NMDA spikes, exhibiting a threshold, sensitivity to NMDAR blockade, and all-or-none
SUPPLEMENTARY INFORMATION Supplementary Figure 1 The supralinear events evoked in CA3 pyramidal cells fulfill the criteria for NMDA spikes, exhibiting a threshold, sensitivity to NMDAR blockade, and all-or-none
Supplementary Figure 1. Basic properties of compound EPSPs at
 Supplementary Figure 1. Basic properties of compound EPSPs at hippocampal CA3 CA3 cell synapses. (a) EPSPs were evoked by extracellular stimulation of the recurrent collaterals and pharmacologically isolated
Supplementary Figure 1. Basic properties of compound EPSPs at hippocampal CA3 CA3 cell synapses. (a) EPSPs were evoked by extracellular stimulation of the recurrent collaterals and pharmacologically isolated
SUPPLEMENTARY INFORMATION
 Supplementary Figure 1. Normal AMPAR-mediated fepsp input-output curve in CA3-Psen cdko mice. Input-output curves, which are plotted initial slopes of the evoked fepsp as function of the amplitude of the
Supplementary Figure 1. Normal AMPAR-mediated fepsp input-output curve in CA3-Psen cdko mice. Input-output curves, which are plotted initial slopes of the evoked fepsp as function of the amplitude of the
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/317/5841/183/dc1 Supporting Online Material for Astrocytes Potentiate Transmitter Release at Single Hippocampal Synapses Gertrudis Perea and Alfonso Araque* *To whom
www.sciencemag.org/cgi/content/full/317/5841/183/dc1 Supporting Online Material for Astrocytes Potentiate Transmitter Release at Single Hippocampal Synapses Gertrudis Perea and Alfonso Araque* *To whom
Supplementary Information
 Supplementary Information D-Serine regulates cerebellar LTD and motor coordination through the 2 glutamate receptor Wataru Kakegawa, Yurika Miyoshi, Kenji Hamase, Shinji Matsuda, Keiko Matsuda, Kazuhisa
Supplementary Information D-Serine regulates cerebellar LTD and motor coordination through the 2 glutamate receptor Wataru Kakegawa, Yurika Miyoshi, Kenji Hamase, Shinji Matsuda, Keiko Matsuda, Kazuhisa
File name: Supplementary Information Description: Supplementary Figures, Supplementary Table and Supplementary References
 File name: Supplementary Information Description: Supplementary Figures, Supplementary Table and Supplementary References File name: Supplementary Data 1 Description: Summary datasheets showing the spatial
File name: Supplementary Information Description: Supplementary Figures, Supplementary Table and Supplementary References File name: Supplementary Data 1 Description: Summary datasheets showing the spatial
Neuroscience 201A (2016) - Problems in Synaptic Physiology
 Question 1: The record below in A shows an EPSC recorded from a cerebellar granule cell following stimulation (at the gap in the record) of a mossy fiber input. These responses are, then, evoked by stimulation.
Question 1: The record below in A shows an EPSC recorded from a cerebellar granule cell following stimulation (at the gap in the record) of a mossy fiber input. These responses are, then, evoked by stimulation.
Dep. Control Time (min)
 aa Control Dep. RP 1s 1 mv 2s 1 mv b % potentiation of IPSP 2 15 1 5 Dep. * 1 2 3 4 Time (min) Supplementary Figure 1. Rebound potentiation of IPSPs in PCs. a, IPSPs recorded with a K + gluconate pipette
aa Control Dep. RP 1s 1 mv 2s 1 mv b % potentiation of IPSP 2 15 1 5 Dep. * 1 2 3 4 Time (min) Supplementary Figure 1. Rebound potentiation of IPSPs in PCs. a, IPSPs recorded with a K + gluconate pipette
Supplementary Figure 1
 Supplementary Figure 1 Supplementary Figure 1 SNARE Probes for FRET/2pFLIM Analysis Used in the Present Study. mturquoise (mtq) and Venus (Ven) are in blue and yellow, respectively. The soluble N-ethylmaleimide-sensitive
Supplementary Figure 1 Supplementary Figure 1 SNARE Probes for FRET/2pFLIM Analysis Used in the Present Study. mturquoise (mtq) and Venus (Ven) are in blue and yellow, respectively. The soluble N-ethylmaleimide-sensitive
Supplementary Figure 1. Microglia do not show signs of classical immune activation following MD a-b. Images showing immunoreactivity for MHCII (a)
 1 Supplementary Figure 1. Microglia do not show signs of classical immune activation following MD a-b. Images showing immunoreactivity for MHCII (a) and CD45 (b) in fixed sections of binocular visual cortex
1 Supplementary Figure 1. Microglia do not show signs of classical immune activation following MD a-b. Images showing immunoreactivity for MHCII (a) and CD45 (b) in fixed sections of binocular visual cortex
Nature Neuroscience: doi: /nn Supplementary Figure 1
 Supplementary Figure 1 Drd1a-Cre driven ChR2 expression in the SCN. (a) Low-magnification image of a representative Drd1a-ChR2 coronal brain section (n = 2) showing endogenous tdtomato fluorescence (magenta).
Supplementary Figure 1 Drd1a-Cre driven ChR2 expression in the SCN. (a) Low-magnification image of a representative Drd1a-ChR2 coronal brain section (n = 2) showing endogenous tdtomato fluorescence (magenta).
How Nicotinic Signaling Shapes Neural Networks
 How Nicotinic Signaling Shapes Neural Networks Darwin K. Berg Division of Biological Sciences University of California, San Diego Nicotinic Cholinergic Signaling Uses the transmitter ACh to activate cation-selective
How Nicotinic Signaling Shapes Neural Networks Darwin K. Berg Division of Biological Sciences University of California, San Diego Nicotinic Cholinergic Signaling Uses the transmitter ACh to activate cation-selective
A genetically targeted optical sensor to monitor calcium signals in astrocyte processes
 A genetically targeted optical sensor to monitor calcium signals in astrocyte processes 1 Eiji Shigetomi, 1 Sebastian Kracun, 2 Michael V. Sofroniew & 1,2 *Baljit S. Khakh Ψ 1 Departments of Physiology
A genetically targeted optical sensor to monitor calcium signals in astrocyte processes 1 Eiji Shigetomi, 1 Sebastian Kracun, 2 Michael V. Sofroniew & 1,2 *Baljit S. Khakh Ψ 1 Departments of Physiology
Supporting Online Material for
 www.sciencemag.org/cgi/content/full/312/5779/1533/dc1 Supporting Online Material for Long-Term Potentiation of Neuron-Glia Synapses Mediated by Ca 2+ - Permeable AMPA Receptors Woo-Ping Ge, Xiu-Juan Yang,
www.sciencemag.org/cgi/content/full/312/5779/1533/dc1 Supporting Online Material for Long-Term Potentiation of Neuron-Glia Synapses Mediated by Ca 2+ - Permeable AMPA Receptors Woo-Ping Ge, Xiu-Juan Yang,
Tyrodes solution in a custom-built imaging chamber as described previously. Images were acquired
 Supplemental Material Supplemental Methods Electrical stimulation of CX-G3-labeled hippocampal neurons Following 5 min incubation in 0.5 µm CX-G3 and washes, 18-20 DIV neurons were imaged in normal Tyrodes
Supplemental Material Supplemental Methods Electrical stimulation of CX-G3-labeled hippocampal neurons Following 5 min incubation in 0.5 µm CX-G3 and washes, 18-20 DIV neurons were imaged in normal Tyrodes
Supplementary Figure 1) GABAergic enhancement by leptin hyperpolarizes POMC neurons A) Representative recording samples showing the membrane
 Supplementary Figure 1) GABAergic enhancement by leptin hyperpolarizes POMC neurons A) Representative recording samples showing the membrane potential recorded from POMC neurons following treatment with
Supplementary Figure 1) GABAergic enhancement by leptin hyperpolarizes POMC neurons A) Representative recording samples showing the membrane potential recorded from POMC neurons following treatment with
Supplemental Information. Differential Regulation. of Evoked and Spontaneous Release. by Presynaptic NMDA Receptors
 Neuron, Volume 96 Supplemental Information Differential Regulation of Evoked and Spontaneous Release by Presynaptic NMDA Receptors Therése Abrahamsson, hristina You hien hou, Si Ying Li, Adamo Mancino,
Neuron, Volume 96 Supplemental Information Differential Regulation of Evoked and Spontaneous Release by Presynaptic NMDA Receptors Therése Abrahamsson, hristina You hien hou, Si Ying Li, Adamo Mancino,
Supplemental Information. Ca V 2.2 Gates Calcium-Independent. but Voltage-Dependent Secretion. in Mammalian Sensory Neurons
 Neuron, Volume 96 Supplemental Information Ca V 2.2 Gates Calcium-Independent but Voltage-Dependent Secretion in Mammalian Sensory Neurons Zuying Chai, Changhe Wang, Rong Huang, Yuan Wang, Xiaoyu Zhang,
Neuron, Volume 96 Supplemental Information Ca V 2.2 Gates Calcium-Independent but Voltage-Dependent Secretion in Mammalian Sensory Neurons Zuying Chai, Changhe Wang, Rong Huang, Yuan Wang, Xiaoyu Zhang,
Cd k5 Ph o s p h o r y l at i o n o f Mu n c18-1 En h a n c e s Sy n a p t i c
 Cd k5 Ph o s p h o r y l at i o n o f Mu n c18-1 En h a n c e s Sy n a p t i c Tr a n s m i s s i o n Sabine K Schmitz 1 Matthijs Verhage 1 Ruud F Toonen 1 1 Functional Genomics, Center for Neurogenomics
Cd k5 Ph o s p h o r y l at i o n o f Mu n c18-1 En h a n c e s Sy n a p t i c Tr a n s m i s s i o n Sabine K Schmitz 1 Matthijs Verhage 1 Ruud F Toonen 1 1 Functional Genomics, Center for Neurogenomics
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION doi:10.1038/nature11306 Supplementary Figures Supplementary Figure 1. Basic characterization of GFP+ RGLs in the dentate gyrus of adult nestin-gfp mice. a, Sample confocal images
SUPPLEMENTARY INFORMATION doi:10.1038/nature11306 Supplementary Figures Supplementary Figure 1. Basic characterization of GFP+ RGLs in the dentate gyrus of adult nestin-gfp mice. a, Sample confocal images
Fig. S4. Current-voltage relations of iglurs. A-C: time courses of currents evoked by 100 ms pulses
 Fig. S1. Immunohistochemical detection of iglur2 protein in single islet cells. A: α cells identified using glucagon-specific antibody express the iglur2 subtype of AMPA receptor. 24 out of 26 identified
Fig. S1. Immunohistochemical detection of iglur2 protein in single islet cells. A: α cells identified using glucagon-specific antibody express the iglur2 subtype of AMPA receptor. 24 out of 26 identified
Supplemental information Acid-sensing ion channel 1a contributes to hippocampal LTP inducibility through multiple mechanisms
 Supplemental information Acid-sensing ion channel 1a contributes to hippocampal LTP inducibility through multiple mechanisms Ming-Gang Liu, Hu-Song Li, Wei-Guang Li, Yan-Jiao Wu, Shi-Ning Deng, Chen Huang,
Supplemental information Acid-sensing ion channel 1a contributes to hippocampal LTP inducibility through multiple mechanisms Ming-Gang Liu, Hu-Song Li, Wei-Guang Li, Yan-Jiao Wu, Shi-Ning Deng, Chen Huang,
Nature Neuroscience: doi: /nn Supplementary Figure 1
 Supplementary Figure 1 Subcellular segregation of VGluT2-IR and TH-IR within the same VGluT2-TH axon (wild type rats). (a-e) Serial sections of a dual VGluT2-TH labeled axon. This axon (blue outline) has
Supplementary Figure 1 Subcellular segregation of VGluT2-IR and TH-IR within the same VGluT2-TH axon (wild type rats). (a-e) Serial sections of a dual VGluT2-TH labeled axon. This axon (blue outline) has
Adenosine A 2A Receptors Are Essential for Long-Term Potentiation of NMDA-EPSCs at Hippocampal Mossy Fiber Synapses
 Article Adenosine A 2A Receptors Are Essential for Long-Term Potentiation of NMDA-EPSCs at Hippocampal Mossy Fiber Synapses Nelson Rebola, 1,3 Rafael Lujan, 2 Rodrigo A. Cunha, 1 and Christophe Mulle 3,
Article Adenosine A 2A Receptors Are Essential for Long-Term Potentiation of NMDA-EPSCs at Hippocampal Mossy Fiber Synapses Nelson Rebola, 1,3 Rafael Lujan, 2 Rodrigo A. Cunha, 1 and Christophe Mulle 3,
Light-evoked hyperpolarization and silencing of neurons by conjugated polymers
 Light-evoked hyperpolarization and silencing of neurons by conjugated polymers Paul Feyen 1,, Elisabetta Colombo 1,2,, Duco Endeman 1, Mattia Nova 1, Lucia Laudato 2, Nicola Martino 2,3, Maria Rosa Antognazza
Light-evoked hyperpolarization and silencing of neurons by conjugated polymers Paul Feyen 1,, Elisabetta Colombo 1,2,, Duco Endeman 1, Mattia Nova 1, Lucia Laudato 2, Nicola Martino 2,3, Maria Rosa Antognazza
Supplementary figure 1: LII/III GIN-cells show morphological characteristics of MC
 1 2 1 3 Supplementary figure 1: LII/III GIN-cells show morphological characteristics of MC 4 5 6 7 (a) Reconstructions of LII/III GIN-cells with somato-dendritic compartments in orange and axonal arborizations
1 2 1 3 Supplementary figure 1: LII/III GIN-cells show morphological characteristics of MC 4 5 6 7 (a) Reconstructions of LII/III GIN-cells with somato-dendritic compartments in orange and axonal arborizations
The mammalian cochlea possesses two classes of afferent neurons and two classes of efferent neurons.
 1 2 The mammalian cochlea possesses two classes of afferent neurons and two classes of efferent neurons. Type I afferents contact single inner hair cells to provide acoustic analysis as we know it. Type
1 2 The mammalian cochlea possesses two classes of afferent neurons and two classes of efferent neurons. Type I afferents contact single inner hair cells to provide acoustic analysis as we know it. Type
SUPPLEMENTARY INFORMATION
 doi: 10.1038/nature06994 A phosphatase cascade by which rewarding stimuli control nucleosomal response A. Stipanovich*, E. Valjent*, M. Matamales*, A. Nishi, J.H. Ahn, M. Maroteaux, J. Bertran-Gonzalez,
doi: 10.1038/nature06994 A phosphatase cascade by which rewarding stimuli control nucleosomal response A. Stipanovich*, E. Valjent*, M. Matamales*, A. Nishi, J.H. Ahn, M. Maroteaux, J. Bertran-Gonzalez,
Ube3a is required for experience-dependent maturation of the neocortex
 Ube3a is required for experience-dependent maturation of the neocortex Koji Yashiro, Thorfinn T. Riday, Kathryn H. Condon, Adam C. Roberts, Danilo R. Bernardo, Rohit Prakash, Richard J. Weinberg, Michael
Ube3a is required for experience-dependent maturation of the neocortex Koji Yashiro, Thorfinn T. Riday, Kathryn H. Condon, Adam C. Roberts, Danilo R. Bernardo, Rohit Prakash, Richard J. Weinberg, Michael
SUPPLEMENTARY INFORMATION
 doi: 1.138/nature6416 Supplementary Notes Spine Ca 2+ signals produced by glutamate uncaging We imaged uncaging-evoked [Ca 2+ ] transients in neurons loaded with a green Ca 2+ - sensitive indicator (G;
doi: 1.138/nature6416 Supplementary Notes Spine Ca 2+ signals produced by glutamate uncaging We imaged uncaging-evoked [Ca 2+ ] transients in neurons loaded with a green Ca 2+ - sensitive indicator (G;
Enhancement of synaptic transmission by cyclic AMP modulation of presynaptic I h channels. Vahri Beaumont and Robert S. Zucker
 Enhancement of synaptic transmission by cyclic AMP modulation of presynaptic I h channels Vahri Beaumont and Robert S. Zucker Background I h channels discovered in 1976 (Noma A. and Irisawa H.) Voltage-gated
Enhancement of synaptic transmission by cyclic AMP modulation of presynaptic I h channels Vahri Beaumont and Robert S. Zucker Background I h channels discovered in 1976 (Noma A. and Irisawa H.) Voltage-gated
Supplementary Figure 1 hlrrk2 promotes CAP dependent protein translation.
 ` Supplementary Figure 1 hlrrk2 promotes CAP dependent protein translation. (a) Overexpression of hlrrk2 in HeLa cells enhances total protein synthesis in [35S] methionine/cysteine incorporation assays.
` Supplementary Figure 1 hlrrk2 promotes CAP dependent protein translation. (a) Overexpression of hlrrk2 in HeLa cells enhances total protein synthesis in [35S] methionine/cysteine incorporation assays.
Supplementary Figure 1
 8w Pia II/III IV V VI PV EYFP EYFP PV EYFP PV d PV EYFP Supplementary Figure a Spike probability x - PV-Cre d Spike probability x - RS RS b e Spike probability Spike probability.6......8..... FS FS c f
8w Pia II/III IV V VI PV EYFP EYFP PV EYFP PV d PV EYFP Supplementary Figure a Spike probability x - PV-Cre d Spike probability x - RS RS b e Spike probability Spike probability.6......8..... FS FS c f
The effects of temperature on vesicular supply and release in autaptic cultures of rat and mouse hippocampal neurons
 Journal of Physiology (2002), 539.2, pp. 523 535 DOI: 10.1013/jphysiol.2001.013277 The Physiological Society 2002 www.jphysiol.org The effects of temperature on vesicular supply and release in autaptic
Journal of Physiology (2002), 539.2, pp. 523 535 DOI: 10.1013/jphysiol.2001.013277 The Physiological Society 2002 www.jphysiol.org The effects of temperature on vesicular supply and release in autaptic
Supplementary Figure 1. GABA depolarizes the majority of immature neurons in the
 Supplementary Figure 1. GABA depolarizes the majority of immature neurons in the upper cortical layers at P3 4 in vivo. (a b) Cell-attached current-clamp recordings illustrate responses to puff-applied
Supplementary Figure 1. GABA depolarizes the majority of immature neurons in the upper cortical layers at P3 4 in vivo. (a b) Cell-attached current-clamp recordings illustrate responses to puff-applied
Wenqin Hu, Cuiping Tian, Tun Li, Mingpo Yang, Han Hou & Yousheng Shu
 Distinct contributions of Na v 1.6 and Na v 1.2 in action potential initiation and backpropagation Wenqin Hu, Cuiping Tian, Tun Li, Mingpo Yang, Han Hou & Yousheng Shu Supplementary figure and legend Supplementary
Distinct contributions of Na v 1.6 and Na v 1.2 in action potential initiation and backpropagation Wenqin Hu, Cuiping Tian, Tun Li, Mingpo Yang, Han Hou & Yousheng Shu Supplementary figure and legend Supplementary
Supplemental Information. Memory-Relevant Mushroom Body. Output Synapses Are Cholinergic
 Neuron, Volume 89 Supplemental Information Memory-Relevant Mushroom Body Output Synapses Are Cholinergic Oliver Barnstedt, David Owald, Johannes Felsenberg, Ruth Brain, John-Paul Moszynski, Clifford B.
Neuron, Volume 89 Supplemental Information Memory-Relevant Mushroom Body Output Synapses Are Cholinergic Oliver Barnstedt, David Owald, Johannes Felsenberg, Ruth Brain, John-Paul Moszynski, Clifford B.
Supplementary Figure 1. SDS-FRL localization of CB 1 in the distal CA3 area of the rat hippocampus. (a-d) Axon terminals (t) in stratum pyramidale
 Supplementary Figure 1. SDS-FRL localization of CB 1 in the distal CA3 area of the rat hippocampus. (a-d) Axon terminals (t) in stratum pyramidale (b) show stronger immunolabeling for CB 1 than those in
Supplementary Figure 1. SDS-FRL localization of CB 1 in the distal CA3 area of the rat hippocampus. (a-d) Axon terminals (t) in stratum pyramidale (b) show stronger immunolabeling for CB 1 than those in
Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS)
 Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS) and their exosomes (EXO) in resting (REST) and activated
Supplementary Figure S1. Venn diagram analysis of mrna microarray data and mirna target analysis. (a) Western blot analysis of T lymphoblasts (CLS) and their exosomes (EXO) in resting (REST) and activated
1. Microfluidic device characteristics and cell compartmentalization
 Supplementary figures: 1. Microfluidic device characteristics and cell compartmentalization Microfluidic channels were molded in PDMS (Fig. S1 A and B) over silicon masters, which were structured with
Supplementary figures: 1. Microfluidic device characteristics and cell compartmentalization Microfluidic channels were molded in PDMS (Fig. S1 A and B) over silicon masters, which were structured with
Supplementary Figure 1
 Supplementary Figure 1 Arcuate ChIEF-tdTomato neurons expressed TH These micrographs show that TH-Cre-ChIEF-tdTomato (magenta), expressed by AAV in a TH-Cre mouse, were immunostained with TH (green) in
Supplementary Figure 1 Arcuate ChIEF-tdTomato neurons expressed TH These micrographs show that TH-Cre-ChIEF-tdTomato (magenta), expressed by AAV in a TH-Cre mouse, were immunostained with TH (green) in
Supplemental Information. Dorsal Raphe Dual Serotonin-Glutamate Neurons. Drive Reward by Establishing Excitatory Synapses
 Cell Reports, Volume 26 Supplemental Information Dorsal Raphe Dual Serotonin-Glutamate Neurons Drive Reward by Establishing Excitatory Synapses on VTA Mesoaccumbens Dopamine Neurons Hui-Ling Wang, Shiliang
Cell Reports, Volume 26 Supplemental Information Dorsal Raphe Dual Serotonin-Glutamate Neurons Drive Reward by Establishing Excitatory Synapses on VTA Mesoaccumbens Dopamine Neurons Hui-Ling Wang, Shiliang
-51mV 30s 3mV. n=14 n=4 p=0.4. Depolarization (mv) 3
 Supplementary Figure 1 a optoβ 2 -AR b ChR2-51mV 30s 3mV -50mV 30s 3mV c 4 n=14 n=4 p=0.4 Depolarization (mv) 3 2 1 0 Both optogenetic actuators, optoβ 2 AR and ChR2, were effective in stimulating astrocytes
Supplementary Figure 1 a optoβ 2 -AR b ChR2-51mV 30s 3mV -50mV 30s 3mV c 4 n=14 n=4 p=0.4 Depolarization (mv) 3 2 1 0 Both optogenetic actuators, optoβ 2 AR and ChR2, were effective in stimulating astrocytes
Title: Plasticity of intrinsic excitability in mature granule cells of the dentate gyrus
 Title: Plasticity of intrinsic excitability in mature granule cells of the dentate gyrus Authors: Jeffrey Lopez-Rojas a1, Martin Heine b1 and Michael R. Kreutz ac1 a Research Group Neuroplasticity, b Research
Title: Plasticity of intrinsic excitability in mature granule cells of the dentate gyrus Authors: Jeffrey Lopez-Rojas a1, Martin Heine b1 and Michael R. Kreutz ac1 a Research Group Neuroplasticity, b Research
Nature Neuroscience: doi: /nn Supplementary Figure 1. Large-scale calcium imaging in vivo.
 Supplementary Figure 1 Large-scale calcium imaging in vivo. (a) Schematic illustration of the in vivo camera imaging set-up for large-scale calcium imaging. (b) High-magnification two-photon image from
Supplementary Figure 1 Large-scale calcium imaging in vivo. (a) Schematic illustration of the in vivo camera imaging set-up for large-scale calcium imaging. (b) High-magnification two-photon image from
1.0. FSL NMDAR-fEPSP 0.8. amplitude (mv) Intensity (µa) 2.0 SD FSL Time (ms)
 a 2.5 1. AMPAR-fEPSP slope (mv/ms) 2. 1. NMDAR-fEPSP amplitude (mv).8.6.4.5.2. 2 4 6 8. 1 2 3 4 5 Intensity (µa) Intensity (µa) b 2. PPF Ratio (fepsp2/fepsp1) 1..5. 5 1 2 5 Time (ms) Supplementary Figure
a 2.5 1. AMPAR-fEPSP slope (mv/ms) 2. 1. NMDAR-fEPSP amplitude (mv).8.6.4.5.2. 2 4 6 8. 1 2 3 4 5 Intensity (µa) Intensity (µa) b 2. PPF Ratio (fepsp2/fepsp1) 1..5. 5 1 2 5 Time (ms) Supplementary Figure
Hormonal gain control of a medial preoptic area social reward circuit
 CORRECTION NOTICE Nat. Neurosci. 20, 449 458 (2017) Hormonal gain control of a medial preoptic area social reward circuit Jenna A McHenry, James M Otis, Mark A Rossi, J Elliott Robinson, Oksana Kosyk,
CORRECTION NOTICE Nat. Neurosci. 20, 449 458 (2017) Hormonal gain control of a medial preoptic area social reward circuit Jenna A McHenry, James M Otis, Mark A Rossi, J Elliott Robinson, Oksana Kosyk,
Bidirectional NMDA receptor plasticity controls CA3 output and heterosynaptic metaplasticity
 Bidirectional NMDA receptor plasticity controls CA output and heterosynaptic metaplasticity David L. Hunt, Nagore Puente, Pedro Grandes, Pablo E. Castillo a NMDAR EPSC (pa) - - -8-6 -4 - st 5 nd 5 b NMDAR
Bidirectional NMDA receptor plasticity controls CA output and heterosynaptic metaplasticity David L. Hunt, Nagore Puente, Pedro Grandes, Pablo E. Castillo a NMDAR EPSC (pa) - - -8-6 -4 - st 5 nd 5 b NMDAR
genome edited transient transfection, CMV promoter
 Supplementary Figure 1. In the absence of new protein translation, overexpressed caveolin-1-gfp is degraded faster than caveolin-1-gfp expressed from the endogenous caveolin 1 locus % loss of total caveolin-1-gfp
Supplementary Figure 1. In the absence of new protein translation, overexpressed caveolin-1-gfp is degraded faster than caveolin-1-gfp expressed from the endogenous caveolin 1 locus % loss of total caveolin-1-gfp
Nature Neuroscience: doi: /nn Supplementary Figure 1
 Supplementary Figure 1 Bidirectional optogenetic modulation of the tonic activity of CEA PKCδ + neurons in vitro. a, Top, Cell-attached voltage recording illustrating the blue light-induced increase in
Supplementary Figure 1 Bidirectional optogenetic modulation of the tonic activity of CEA PKCδ + neurons in vitro. a, Top, Cell-attached voltage recording illustrating the blue light-induced increase in
Synaptic Transmission: Ionic and Metabotropic
 Synaptic Transmission: Ionic and Metabotropic D. Purves et al. Neuroscience (Sinauer Assoc.) Chapters 5, 6, 7. C. Koch. Biophysics of Computation (Oxford) Chapter 4. J.G. Nicholls et al. From Neuron to
Synaptic Transmission: Ionic and Metabotropic D. Purves et al. Neuroscience (Sinauer Assoc.) Chapters 5, 6, 7. C. Koch. Biophysics of Computation (Oxford) Chapter 4. J.G. Nicholls et al. From Neuron to
Supplementary Figure 1
 Supplementary Figure 1 Miniature microdrive, spike sorting and sleep stage detection. a, A movable recording probe with 8-tetrodes (32-channels). It weighs ~1g. b, A mouse implanted with 8 tetrodes in
Supplementary Figure 1 Miniature microdrive, spike sorting and sleep stage detection. a, A movable recording probe with 8-tetrodes (32-channels). It weighs ~1g. b, A mouse implanted with 8 tetrodes in
SUPPLEMENTARY INFORMATION
 Supplementary Figure 1. Behavioural effects of ketamine in non-stressed and stressed mice. Naive C57BL/6 adult male mice (n=10/group) were given a single dose of saline vehicle or ketamine (3.0 mg/kg,
Supplementary Figure 1. Behavioural effects of ketamine in non-stressed and stressed mice. Naive C57BL/6 adult male mice (n=10/group) were given a single dose of saline vehicle or ketamine (3.0 mg/kg,
TGF-β Signaling Regulates Neuronal C1q Expression and Developmental Synaptic Refinement
 Supplementary Information Title: TGF-β Signaling Regulates Neuronal C1q Expression and Developmental Synaptic Refinement Authors: Allison R. Bialas and Beth Stevens Supplemental Figure 1. In vitro characterization
Supplementary Information Title: TGF-β Signaling Regulates Neuronal C1q Expression and Developmental Synaptic Refinement Authors: Allison R. Bialas and Beth Stevens Supplemental Figure 1. In vitro characterization
Nature Neuroscience: doi: /nn Supplementary Figure 1. Diverse anorexigenic signals induce c-fos expression in CEl PKC-δ + neurons
 Supplementary Figure 1 Diverse anorexigenic signals induce c-fos expression in CEl PKC-δ + neurons a-c. Quantification of CEl c-fos expression in mice intraperitoneal injected with anorexigenic drugs (a),
Supplementary Figure 1 Diverse anorexigenic signals induce c-fos expression in CEl PKC-δ + neurons a-c. Quantification of CEl c-fos expression in mice intraperitoneal injected with anorexigenic drugs (a),
BIPN 140 Problem Set 6
 BIPN 140 Problem Set 6 1) The hippocampus is a cortical structure in the medial portion of the temporal lobe (medial temporal lobe in primates. a) What is the main function of the hippocampus? The hippocampus
BIPN 140 Problem Set 6 1) The hippocampus is a cortical structure in the medial portion of the temporal lobe (medial temporal lobe in primates. a) What is the main function of the hippocampus? The hippocampus
Authors: K. L. Arendt, M. Royo, M. Fernández-Monreal, S. Knafo, C. N. Petrok, J.
 SUPPLEMENTARY INFORMATION Title: PIP 3 controls synaptic function by maintaining AMPA receptor clustering at the postsynaptic membrane Authors: K. L. Arendt, M. Royo, M. Fernández-Monreal, S. Knafo, C.
SUPPLEMENTARY INFORMATION Title: PIP 3 controls synaptic function by maintaining AMPA receptor clustering at the postsynaptic membrane Authors: K. L. Arendt, M. Royo, M. Fernández-Monreal, S. Knafo, C.
BIPN 140 Problem Set 6
 BIPN 140 Problem Set 6 1) Hippocampus is a cortical structure in the medial portion of the temporal lobe (medial temporal lobe in primates. a) What is the main function of the hippocampus? The hippocampus
BIPN 140 Problem Set 6 1) Hippocampus is a cortical structure in the medial portion of the temporal lobe (medial temporal lobe in primates. a) What is the main function of the hippocampus? The hippocampus
SUPPLEMENTARY INFORMATION
 DOI: 10.1038/ncb2566 Figure S1 CDKL5 protein expression pattern and localization in mouse brain. (a) Multiple-tissue western blot from a postnatal day (P) 21 mouse probed with an antibody against CDKL5.
DOI: 10.1038/ncb2566 Figure S1 CDKL5 protein expression pattern and localization in mouse brain. (a) Multiple-tissue western blot from a postnatal day (P) 21 mouse probed with an antibody against CDKL5.
SUPPLEMENTARY INFORMATION
 doi:1.138/nature1139 a d Whisker angle (deg) Whisking repeatability Control Muscimol.4.3.2.1 -.1 8 4-4 1 2 3 4 Performance (d') Pole 8 4-4 1 2 3 4 5 Time (s) b Mean protraction angle (deg) e Hit rate (p
doi:1.138/nature1139 a d Whisker angle (deg) Whisking repeatability Control Muscimol.4.3.2.1 -.1 8 4-4 1 2 3 4 Performance (d') Pole 8 4-4 1 2 3 4 5 Time (s) b Mean protraction angle (deg) e Hit rate (p
Synaptotagmin-7-Mediated Asynchronous Release Boosts High-Fidelity Synchronous Transmission at a Central Synapse
 Article Synaptotagmin-7-Mediated Asynchronous Release Boosts High-Fidelity Synchronous Transmission at a Central Synapse Highlights d Syt7 KO does not alter fast release or short-term plasticity of calyx
Article Synaptotagmin-7-Mediated Asynchronous Release Boosts High-Fidelity Synchronous Transmission at a Central Synapse Highlights d Syt7 KO does not alter fast release or short-term plasticity of calyx
Nature Neuroscience: doi: /nn Supplementary Figure 1
 Supplementary Figure 1 Relative expression of K IR2.1 transcript to enos was reduced 29-fold in capillaries from knockout animals. Relative expression of K IR2.1 transcript to enos was reduced 29-fold
Supplementary Figure 1 Relative expression of K IR2.1 transcript to enos was reduced 29-fold in capillaries from knockout animals. Relative expression of K IR2.1 transcript to enos was reduced 29-fold
Supplementary Figures
 Supplementary Figures Supplementary Figure 1 Characterization of stable expression of GlucB and sshbira in the CT26 cell line (a) Live cell imaging of stable CT26 cells expressing green fluorescent protein
Supplementary Figures Supplementary Figure 1 Characterization of stable expression of GlucB and sshbira in the CT26 cell line (a) Live cell imaging of stable CT26 cells expressing green fluorescent protein
Dopamine in Ube3a m-/p+ mice. Online Supplemental Material
 Online Supplemental Material S1 Supplemental Figure 1. Schematic of rate-dependent intracranial self-stimulation (ICSS) (A) Mice implanted with monopolar stimulating electrodes to the medial forebrain
Online Supplemental Material S1 Supplemental Figure 1. Schematic of rate-dependent intracranial self-stimulation (ICSS) (A) Mice implanted with monopolar stimulating electrodes to the medial forebrain
Alterations in Synaptic Strength Preceding Axon Withdrawal
 Alterations in Synaptic Strength Preceding Axon Withdrawal H. Colman, J. Nabekura, J.W. Lichtman presented by Ana Fiallos Synaptic Transmission at the Neuromuscular Junction Motor neurons with cell bodies
Alterations in Synaptic Strength Preceding Axon Withdrawal H. Colman, J. Nabekura, J.W. Lichtman presented by Ana Fiallos Synaptic Transmission at the Neuromuscular Junction Motor neurons with cell bodies
Lack of GPR88 enhances medium spiny neuron activity and alters. motor- and cue- dependent behaviors
 Lack of GPR88 enhances medium spiny neuron activity and alters motor- and cue- dependent behaviors Albert Quintana, Elisenda Sanz, Wengang Wang, Granville P. Storey, Ali D. Güler Matthew J. Wanat, Bryan
Lack of GPR88 enhances medium spiny neuron activity and alters motor- and cue- dependent behaviors Albert Quintana, Elisenda Sanz, Wengang Wang, Granville P. Storey, Ali D. Güler Matthew J. Wanat, Bryan
CELLULAR NEUROPHYSIOLOGY
 CELLULAR NEUROPHYSIOLOGY CONSTANCE HAMMOND 4. SYNAPTIC TRANSMISSION II: GLUTAMATERGIC TRANSMISSION Video 4-1: Observations and glutamate receptor channels Synaptic transmission II 1 Constance Hammond Observation
CELLULAR NEUROPHYSIOLOGY CONSTANCE HAMMOND 4. SYNAPTIC TRANSMISSION II: GLUTAMATERGIC TRANSMISSION Video 4-1: Observations and glutamate receptor channels Synaptic transmission II 1 Constance Hammond Observation
Activity-Induced Rapid Synaptic Maturation Mediated by Presynaptic Cdc42 Signaling
 Neuron 50, 401 414, May 4, 2006 ª2006 Elsevier Inc. DOI 10.1016/j.neuron.2006.03.017 Activity-Induced Rapid Synaptic Maturation Mediated by Presynaptic Cdc42 Signaling Wanhua Shen, 1,2,5 Bei Wu, 1,2,5
Neuron 50, 401 414, May 4, 2006 ª2006 Elsevier Inc. DOI 10.1016/j.neuron.2006.03.017 Activity-Induced Rapid Synaptic Maturation Mediated by Presynaptic Cdc42 Signaling Wanhua Shen, 1,2,5 Bei Wu, 1,2,5
Nature Methods: doi: /nmeth Supplementary Figure 1. Activity in turtle dorsal cortex is sparse.
 Supplementary Figure 1 Activity in turtle dorsal cortex is sparse. a. Probability distribution of firing rates across the population (notice log scale) in our data. The range of firing rates is wide but
Supplementary Figure 1 Activity in turtle dorsal cortex is sparse. a. Probability distribution of firing rates across the population (notice log scale) in our data. The range of firing rates is wide but
Rescue of mutant rhodopsin traffic by metformin-induced AMPK activation accelerates photoreceptor degeneration Athanasiou et al
 Supplementary Material Rescue of mutant rhodopsin traffic by metformin-induced AMPK activation accelerates photoreceptor degeneration Athanasiou et al Supplementary Figure 1. AICAR improves P23H rod opsin
Supplementary Material Rescue of mutant rhodopsin traffic by metformin-induced AMPK activation accelerates photoreceptor degeneration Athanasiou et al Supplementary Figure 1. AICAR improves P23H rod opsin
Nature Neuroscience: doi: /nn.4335
 Supplementary Figure 1 Cholinergic neurons projecting to the VTA are concentrated in the caudal mesopontine region. (a) Schematic showing the sites of retrograde tracer injections in the VTA: cholera toxin
Supplementary Figure 1 Cholinergic neurons projecting to the VTA are concentrated in the caudal mesopontine region. (a) Schematic showing the sites of retrograde tracer injections in the VTA: cholera toxin
Social transmission and buffering of synaptic changes after stress
 SUPPLEMENTARY INFORMATION Articles https://doi.org/10.1038/s41593-017-0044-6 In the format provided by the authors and unedited. Social transmission and buffering of synaptic changes after stress Toni-Lee
SUPPLEMENTARY INFORMATION Articles https://doi.org/10.1038/s41593-017-0044-6 In the format provided by the authors and unedited. Social transmission and buffering of synaptic changes after stress Toni-Lee
Nature Neuroscience: doi: /nn Supplementary Figure 1. Trial structure for go/no-go behavior
 Supplementary Figure 1 Trial structure for go/no-go behavior a, Overall timeline of experiments. Day 1: A1 mapping, injection of AAV1-SYN-GCAMP6s, cranial window and headpost implantation. Water restriction
Supplementary Figure 1 Trial structure for go/no-go behavior a, Overall timeline of experiments. Day 1: A1 mapping, injection of AAV1-SYN-GCAMP6s, cranial window and headpost implantation. Water restriction
Supplementary Figure 1
 Supplementary Figure 1 Supplementary Figure 1. Short latency of the fepsp evoked in CA3 by electrical stimulation of perforant path inputs (a) Single and superimposed representative perforant pathway-ca3
Supplementary Figure 1 Supplementary Figure 1. Short latency of the fepsp evoked in CA3 by electrical stimulation of perforant path inputs (a) Single and superimposed representative perforant pathway-ca3
Supplementary Table I Blood pressure and heart rate measurements pre- and post-stroke
 SUPPLEMENTARY INFORMATION doi:10.1038/nature09511 Supplementary Table I Blood pressure and heart rate measurements pre- and post-stroke Pre Post 7-days Systolic Diastolic BPM Systolic Diastolic BPM Systolic
SUPPLEMENTARY INFORMATION doi:10.1038/nature09511 Supplementary Table I Blood pressure and heart rate measurements pre- and post-stroke Pre Post 7-days Systolic Diastolic BPM Systolic Diastolic BPM Systolic
Supplementary Figure 1. Identification of the type II spiral ganglion neurons (SGN) via immunofluorescence of peripherin protein (PRPH).
 Supplementary Figure 1. Identification of the type II spiral ganglion neurons (SGN) via immunofluorescence of peripherin protein (PRPH). (a), (b), PRPH immunolabelling of cryosections from post-natal day
Supplementary Figure 1. Identification of the type II spiral ganglion neurons (SGN) via immunofluorescence of peripherin protein (PRPH). (a), (b), PRPH immunolabelling of cryosections from post-natal day
Ivy/Neurogliaform Interneurons Coordinate Activity in the Neurogenic Niche
 Ivy/Neurogliaform Interneurons Coordinate Activity in the Neurogenic Niche Sean J. Markwardt, Cristina V. Dieni, Jacques I. Wadiche & Linda Overstreet-Wadiche Supplementary Methods. Animals We used hemizygous
Ivy/Neurogliaform Interneurons Coordinate Activity in the Neurogenic Niche Sean J. Markwardt, Cristina V. Dieni, Jacques I. Wadiche & Linda Overstreet-Wadiche Supplementary Methods. Animals We used hemizygous
1) Drop off in the Bi 150 box outside Baxter 331 or to the head TA (jcolas).
 Bi/CNS/NB 150 Problem Set 3 Due: Tuesday, Oct. 27, at 4:30 pm Instructions: 1) Drop off in the Bi 150 box outside Baxter 331 or e-mail to the head TA (jcolas). 2) Submit with this cover page. 3) Use a
Bi/CNS/NB 150 Problem Set 3 Due: Tuesday, Oct. 27, at 4:30 pm Instructions: 1) Drop off in the Bi 150 box outside Baxter 331 or e-mail to the head TA (jcolas). 2) Submit with this cover page. 3) Use a
Chapter 5 subtitles GABAergic synaptic transmission
 CELLULAR NEUROPHYSIOLOGY CONSTANCE HAMMOND Chapter 5 subtitles GABAergic synaptic transmission INTRODUCTION (2:57) In this fifth chapter, you will learn how the binding of the GABA neurotransmitter to
CELLULAR NEUROPHYSIOLOGY CONSTANCE HAMMOND Chapter 5 subtitles GABAergic synaptic transmission INTRODUCTION (2:57) In this fifth chapter, you will learn how the binding of the GABA neurotransmitter to
Competition between Phasic and Asynchronous Release for Recovered Synaptic Vesicles at Developing Hippocampal Autaptic Synapses
 420 The Journal of Neuroscience, January 14, 2004 24(2):420 433 Cellular/Molecular Competition between Phasic and Asynchronous Release for Recovered Synaptic Vesicles at Developing Hippocampal Autaptic
420 The Journal of Neuroscience, January 14, 2004 24(2):420 433 Cellular/Molecular Competition between Phasic and Asynchronous Release for Recovered Synaptic Vesicles at Developing Hippocampal Autaptic
Supplementary Figure 1
 Supplementary Figure 1 Localization of virus injections. (a) Schematic showing the approximate center of AAV-DIO-ChR2-YFP injection sites in the NAc of Dyn-cre mice (n=8 mice, 16 injections; caudate/putamen,
Supplementary Figure 1 Localization of virus injections. (a) Schematic showing the approximate center of AAV-DIO-ChR2-YFP injection sites in the NAc of Dyn-cre mice (n=8 mice, 16 injections; caudate/putamen,
Supplementary Figure 1. mir124 does not change neuron morphology and synaptic
 Supplementary Figure 1. mir124 does not change neuron morphology and synaptic density. Hippocampal neurons were transfected with mir124 (containing DsRed) or DsRed as a control. 2 d after transfection,
Supplementary Figure 1. mir124 does not change neuron morphology and synaptic density. Hippocampal neurons were transfected with mir124 (containing DsRed) or DsRed as a control. 2 d after transfection,
1) Drop off in the Bi 150 box outside Baxter 331 or to the head TA (jcolas).
 Bi/CNS/NB 150 Problem Set 3 Due: Tuesday, Oct. 27, at 4:30 pm Instructions: 1) Drop off in the Bi 150 box outside Baxter 331 or e-mail to the head TA (jcolas). 2) Submit with this cover page. 3) Use a
Bi/CNS/NB 150 Problem Set 3 Due: Tuesday, Oct. 27, at 4:30 pm Instructions: 1) Drop off in the Bi 150 box outside Baxter 331 or e-mail to the head TA (jcolas). 2) Submit with this cover page. 3) Use a
Supplementary Figure 1. Using DNA barcode-labeled MHC multimers to generate TCR fingerprints
 Supplementary Figure 1 Using DNA barcode-labeled MHC multimers to generate TCR fingerprints (a) Schematic overview of the workflow behind a TCR fingerprint. Each peptide position of the original peptide
Supplementary Figure 1 Using DNA barcode-labeled MHC multimers to generate TCR fingerprints (a) Schematic overview of the workflow behind a TCR fingerprint. Each peptide position of the original peptide
NS200: In vitro electrophysiology section September 11th, 2013
 NS200: In vitro electrophysiology section September 11th, 2013 Quynh Anh Nguyen, 4 th Year Nicoll Lab quynhanh.nguyen@ucsf.edu N276 Genentech Hall, Mission Bay Outline Part I: Theory Review of circuit
NS200: In vitro electrophysiology section September 11th, 2013 Quynh Anh Nguyen, 4 th Year Nicoll Lab quynhanh.nguyen@ucsf.edu N276 Genentech Hall, Mission Bay Outline Part I: Theory Review of circuit
Bioscience in the 21st century
 Bioscience in the 21st century Neurons, Synapses, and Signaling Dr. Michael Burger Outline: 1. Why neuroscience? 2. The neuron 3. Action potentials 4. Synapses 5. Organization of the nervous system 6.
Bioscience in the 21st century Neurons, Synapses, and Signaling Dr. Michael Burger Outline: 1. Why neuroscience? 2. The neuron 3. Action potentials 4. Synapses 5. Organization of the nervous system 6.
Chapter 3 subtitles Action potentials
 CELLULAR NEUROPHYSIOLOGY CONSTANCE HAMMOND Chapter 3 subtitles Action potentials Introduction (3:15) This third chapter explains the calcium current triggered by the arrival of the action potential in
CELLULAR NEUROPHYSIOLOGY CONSTANCE HAMMOND Chapter 3 subtitles Action potentials Introduction (3:15) This third chapter explains the calcium current triggered by the arrival of the action potential in
Autonomous Function of Synaptotagmin 1 in Triggering Synchronous Release Independent of Asynchronous Release
 Neuron, Vol. 48, 547 554, November 23, 2005, Copyright ª2005 by Elsevier Inc. DOI 10.1016/j.neuron.2005.09.006 Autonomous Function of Synaptotagmin 1 in Triggering Synchronous Release Independent of Asynchronous
Neuron, Vol. 48, 547 554, November 23, 2005, Copyright ª2005 by Elsevier Inc. DOI 10.1016/j.neuron.2005.09.006 Autonomous Function of Synaptotagmin 1 in Triggering Synchronous Release Independent of Asynchronous
T H E J O U R N A L O F C E L L B I O L O G Y
 Supplemental material Chen et al., http://www.jcb.org/cgi/content/full/jcb.201210119/dc1 T H E J O U R N A L O F C E L L B I O L O G Y Figure S1. Lack of fast reversibility of UVR8 dissociation. (A) HEK293T
Supplemental material Chen et al., http://www.jcb.org/cgi/content/full/jcb.201210119/dc1 T H E J O U R N A L O F C E L L B I O L O G Y Figure S1. Lack of fast reversibility of UVR8 dissociation. (A) HEK293T
Activity Dependent Changes At the Developing Neuromuscular Junction
 Activity Dependent Changes At the Developing Neuromuscular Junction (slides 16, 17 and 18 have been slightly modified for clarity) MCP Lecture 2-3 9.013/7.68 04 Neuromuscular Junction Development 1. Muscle
Activity Dependent Changes At the Developing Neuromuscular Junction (slides 16, 17 and 18 have been slightly modified for clarity) MCP Lecture 2-3 9.013/7.68 04 Neuromuscular Junction Development 1. Muscle
Cellular Neurobiology / BIPN 140
 SECOND MIDTERM EXAMINATION Fall, 2015 GENERAL INSTRUCTIONS 1. Please write your name on ALL 6 pages. 2. Please answer each question IN THE SPACE ALLOTTED. 1) /10 pts 2) /10 pts 3) /15 pts 4) /15 pts 5)
SECOND MIDTERM EXAMINATION Fall, 2015 GENERAL INSTRUCTIONS 1. Please write your name on ALL 6 pages. 2. Please answer each question IN THE SPACE ALLOTTED. 1) /10 pts 2) /10 pts 3) /15 pts 4) /15 pts 5)
BIPN140 Lecture 8: Synaptic Transmission II
 BIPN140 Lecture 8: Synaptic Transmission II 1. Postsynaptic Receptors: Metabotropic & Ionotropic 2. Postsynaptic Responses (Postsynaptic Potentials, PSPs) 3. Neurotransmitters Su (FA16) Chemical Synapse:
BIPN140 Lecture 8: Synaptic Transmission II 1. Postsynaptic Receptors: Metabotropic & Ionotropic 2. Postsynaptic Responses (Postsynaptic Potentials, PSPs) 3. Neurotransmitters Su (FA16) Chemical Synapse:
Two distinct mechanisms for experiencedependent
 SUPPLEMENTARY INFORMATION Articles https://doi.org/10.1038/s41593-018-0150-0 In the format provided by the authors and unedited. Two distinct mechanisms for experiencedependent homeostasis Michelle C.
SUPPLEMENTARY INFORMATION Articles https://doi.org/10.1038/s41593-018-0150-0 In the format provided by the authors and unedited. Two distinct mechanisms for experiencedependent homeostasis Michelle C.
Presynaptic NMDA receptor control of spontaneous and evoked activity By: Sally Si Ying Li Supervisor: Jesper Sjöström
 Presynaptic NMDA receptor control of spontaneous and evoked activity By: Sally Si Ying Li Supervisor: Jesper Sjöström NMDA receptors are traditionally known to function as post-synaptic coincidence detectors.
Presynaptic NMDA receptor control of spontaneous and evoked activity By: Sally Si Ying Li Supervisor: Jesper Sjöström NMDA receptors are traditionally known to function as post-synaptic coincidence detectors.
LETTERS. Allosteric modulation of the presynaptic Ca 21 sensor for vesicle fusion. Xuelin Lou 1, Volker Scheuss 1 & Ralf Schneggenburger 1
 Vol 435 26 May 2005 doi:10.1038/nature03568 Allosteric modulation of the presynaptic Ca 21 sensor for vesicle fusion Xuelin Lou 1, Volker Scheuss 1 & Ralf Schneggenburger 1 Neurotransmitter release is
Vol 435 26 May 2005 doi:10.1038/nature03568 Allosteric modulation of the presynaptic Ca 21 sensor for vesicle fusion Xuelin Lou 1, Volker Scheuss 1 & Ralf Schneggenburger 1 Neurotransmitter release is
Nature Biotechnology: doi: /nbt.3828
 Supplementary Figure 1 Development of a FRET-based MCS. (a) Linker and MA2 modification are indicated by single letter amino acid code. indicates deletion of amino acids and N or C indicate the terminus
Supplementary Figure 1 Development of a FRET-based MCS. (a) Linker and MA2 modification are indicated by single letter amino acid code. indicates deletion of amino acids and N or C indicate the terminus
Nature Neuroscience: doi: /nn Supplementary Figure 1
 Supplementary Figure 1 Atlas representations of the midcingulate (MCC) region targeted in this study compared against the anterior cingulate (ACC) region commonly reported. Coronal sections are shown on
Supplementary Figure 1 Atlas representations of the midcingulate (MCC) region targeted in this study compared against the anterior cingulate (ACC) region commonly reported. Coronal sections are shown on
