ROME Update from DDW and ACG 2014
|
|
|
- Betty Gaines
- 5 years ago
- Views:
Transcription
1 ROME Update from DDW and ACG
2 Rome Faculty Douglas Drossman, MD University of North Carolina Chapel Hill, NC Lin Chang, MD David Geffen School of Medicine at UCLA Los Angeles, CA 2
3 This Program is Supported by Educational Grants from: 3
4 Attention The abstracts discussed in this presentation were presented at Digestive Disease Week As such, they have not been subjected to a full peer-review process. These abstracts discuss uses of particular agents that are outside of their indications and/or agents that have not yet received approval for the uses discussed herein. 4
5 Nortriptyline for Idiopathic Gastroparesis: A Multicenter, Randomized, Double-Masked, Placebo-Controlled Trial (NORIG) DD- #1 Parkman HP, Van Natta M, Abell TL et al. Abstract 5 5
6 Tricyclic Antidepressant Actions in FGIDs Antidepressant action Visceral analgesia Changes in motility Smooth muscle relaxation FGID=functional gastrointestinal disorder Adapted from Rome Foundation Functional GI Disorders Specialty Modules. 6 6
7 Background and Methods Background Tricyclic antidepressants (TCAs) are frequently used as neuromodulators for treatment of nausea, vomiting, and abdominal pain in patients with gastroparesis This practice is not yet based on evidence from well-designed clinical trials Study design 15-week, multicenter, randomized, placebo-controlled, double-blind, parallel group, dose escalation trial Patients 130 patients with confirmed gastroparesis with delayed gastric emptying by scintigraphy and symptom scores >21 on Gastroparesis Cardinal Symptom Index (GCSI) Treatments Nortriptyline (escalated at 3-week intervals: 10, 25, 60, 75 mg/day) Placebo Primary end point Decrease from the baseline GCSI score of 50% on 2 consecutive visits over 15 weeks of follow-up 7
8 Efficacy Results Overall symptomatic improvement did not differ between nortriptyline and control patients 21% 23% Patients (%) 8
9 Results: Additional End Points and Safety Nortriptyline was associated with: Greater decrease in nausea (P=.04) and abdominal pain (P=.03) at 3 weeks but not after Greater improvement in appetite (P=.03) and ability to finish a meal (P=.08) at week 15 Greater increase in body mass index (0.5 vs -0.2 kg/m 2 ; P=.03) Improved symptoms in subgroup with impaired satiety testing (<250 ml Ensure; P=.06)) Safety Nortriptyline was associated with a high rate of discontinuation (19 vs 6 placebo patients) Adverse effects were the primary reason for discontinuation 9
10 Conclusions In this first adequately powered randomized clinical trial of a neuromodulator in idiopathic gastroparesis, nortriptyline did not improve overall symptom endpoint in idiopathic gastroparesis over a 15 week period Improvements in appetite, satiety and BMI were noted An early improvement in nausea and abdominal pain was seen early in the trial, but this was not sustained Nortriptyline was associated with early treatment discontinuation in 29% (19 vs 6 placebo) patients 10
11 The Functional Dyspepsia Treatment Trial: Key Results DD- #2 Locke GR et el. Abstract
12 Methods Design: Prospective, randomized, double-blind parallel group trial: 3 arms Placebo Amitriptyline 50 mg Escitalopram 10 mg Patients completed questionnaires, nutrient drink test, gastroesophageal study, SPECT, and blood testing before and after treatment Screening Treatment Follow-up Week 0 wks 12 wks 36 wks Q Q Randomization GI testing GI testing SPECT=single photon emission computed tomography. 12
13 Results: Primary Endpoint P= % 40% 38% Patients with ulcer-like but not dysmotility-like disease improved with amitriptyline 13
14 Conclusions Amitriptyline, but not escitalopram, was efficacious for the global symptoms of functional dyspepsia Treatment effect of amitriptyline was greatest in the ulcer-like functional dyspepsia subgroup and in males Response was not predicted by patient characteristics (eg, anxiety, BMI) or baseline GI physiology test results 14
15 Do Co-Morbid Mental Disorders Impact Differently on Perceived Symptom Severity in Patients With Gastrointestinal Diseases? A Comparison of Functional Dyspepsia and Intestinal Graft-Versus-Host Disease LC- #3 Adam B, Klisanin V, Junne FP et al. Abstract Sa
16 Background and Methods Background Anxiety and depression are associated with functional dyspepsia (FD) but their role in symptom severity in organic gastrointestinal diseases is not well known. Study design This study compared the role of anxiety and depression in a functional GI disorder, functional dyspepsia, and an organic GI disease, graft-vs.-host disease which is a major cause of non-relapse mortality and morbidity. GVHD can occur after allogenic stem cell transplantation. 16
17 Background and Methods Study design (cont.) 67 patients with GVHD and 45 patients with Rome III + FD were studied. GI symptoms were assessed using a standardized Gastrointestinal Symptom Score for epigastric pain, cramps, fullness, early satiety, nausea and vomiting Anxiety and depression were measured using the Hospital Anxiety and Depression (HAD) scale HAD scores of 0-7 were considered to be indicative of no psychological co-morbidity HAD scores between 8-10 were categorized as probably anxiety and depression Scores >= 11 were indicative of relevant anxiety and depression 17
18 Results Probable Anxiety Relevant Anxiety Probable Depression Relevant Depression agvhd 12/67 (17.9%) 6/67 (9.0%) 4/67 (6.0%) 8/67 (11.9%) FD 12/45 (26.7%) 8/45 (17.7%) 11/45 (24.4%) 6/45 (13.3%) Overall anxiety and depression scores did not differ between FD and GVHD 18
19 Results Anxiety in GVHD correlated with: Severity of pain (r=.25, P=.04) Fullness (r=.43, P<.001) Satiety (r=.40, P=.001) Nausea (r=.36, P=.003) Vomiting (r=.40, P=.001) Anxiety in functional dyspepsia correlated with: Pain (r=.44, P<.028) Fullness (r=.38, P<.001) Satiety (r=.23, P=.03) Vomiting (r=.32, P=.002) 19
20 Results Functional dyspepsia patients without anxiety showed significantly more severe cramps and nausea but less vomiting compared to GVHD without anxiety In FD patients with anxiety, pain was more prominent while vomiting was less evident compared to GVHD with anxiety 20
21 Results Depression in GVHD was associated with: Pain (r=.58, P.001) Cramps (r=.53, P<.001) Fullness (r=.64, P<.001) Satiety (r=.50, P<.001) Nausea (r=.43, P<.001) Vomiting (r=.48, P<.001) Depression was associated with: Pain (r=.23, P<.001) 21
22 Conclusions While patients with functional dyspepsia experience more pain, vomiting is more severe in GVHD Psychological co-morbidity is closely linked to symptom severity in both functional dyspepsia and organic GI disease Anxiety and depression appeared to be associated with pain in FD and a number of GI symptoms in GVHD These differences may indicate different underlying pathophysiological mechanisms rather than psychologically driven factors 22
23 Mirtazapine Improves Early Satiation, Nutrient Intake, Weight Recovery, and Quality of Life in Functional Dyspepsia With Weight Loss: A Double-Blind, Randomized, Placebo-Controlled Pilot Study DD- #4 Ly HG, Carbone F, Holvoet L et al. Abstract
24 Background and Methods Background In up to 40% of cases, weight loss accompanies functional dyspepsia symptoms (eg, early satiation, postprandial fullness, epigastric pain/burning) Study design 8-week, randomized, placebo-controlled study Patients 34 functional dyspepsia patients who lost 10% of original body weight without major depression or anxiety disorder Treatments Mirtazapine 15 mg orally in the evening Placebo End points Functional dyspepsia symptom severity (8 symptoms score 0-3) Weight Functional dyspepsia-specific quality of life Results of nutrient challenge test 24
25 Results Outcome Week 0 vs Week 8 Mirtazapine Week 0 vs Week 8 Placebo Mirtazapine vs Placebo Total Symptoms 10.9 ± 0.9 vs 7.5 ± 1.1 P= ± 0.9 vs 9.9 ± 1.2 NS -3.3 ± 0.9 vs 2.1 ± 1.0 NS Early Satiety 1.9 ± 0.2 vs 1.1 ± 0.26 P= ± 0.2 vs 1.7 ± 0.2 NS -0.8 ± 0.2 vs 0.27 ± 0.2 P=.005 Quality of Life 31.7 ± 1.8 vs 24.5 ± 2.6 P= ± 0.38 vs 30.5 ± 2.9 NS -7.3 ± 1.8 vs 0.1 ± 1.9 P=.030 Weight (kg) 67.0 ± 3.5 vs 68.8 ± 3.8 P= ± 2.7 vs 58.4 ± 2.8 NS 4.2 ± 0.8 vs -0.1 ± 0.4 P=.0002 Nutrient Intake (cal) ± 42.3 vs ± 72.3 P= ± 75.0 vs ± 85.7 NS ± 55.0 vs ± 38.1 P=
26 Results Dyspeptic symptoms Placebo Mirtazapine 15.0 Symptom score Week
27 Conclusions Mirtazapine significantly improves early satiation and quality of life in FD with weight loss The favorable effect of mirtazapine is associated with improved nutrient intake and superior recovery of weight loss 27
28 Effect of the H1-Receptor Antagonist Ebastin on Visceral Perception and Clinical Symptoms in IBS LC- #5 Van Wanrooji S, Wouters MM, Van Oudenhove L et al. Abstract
29 Background and Methods Background Previous studies suggest that ketotifen (mast cell stabilizer/h1 antagonist) improves visceral hypersensitivity and IBS symptoms but doesn t appear to have this effect by decreasing mast cell mediators and therefore may have a beneficial effect via H1 blockade Study design 12-week, randomized, placebo-controlled study evaluating the effects of the H1-antagonist ebastin in IBS 55 Rome III+ IBS patients stratified by normal or increased visceral sensitivity determined by barostat balloon distension Measured visceral sensitivity before and after treatment 29
30 Background and Methods Primary end point Reduction in visual analog score during +21 mm Hg distensions of 15 points Symptom measures served as secondary endpoints IBS symptoms by subjective global assessment, Gastrointestinal Symptom Rating Scale (GSRS), and health-related quality of life (IBS-QOL) scored before, during, and after treatment 30
31 Results 55 IBS patients (62% women, mean age = 35 yrs) Visceral hypersensitivity was seen in 45% Symptom scores evoked by rectal distension were not significantly influenced by ebastin 31
32 Results SGA Abdominal pain (100mm VAS) Figure 1A. SGA abdominal pain (mass x SGA) run-in N=26 Placebo N=21 Ebastin Treatment Period by Treatment Interaction Effect (P=.03) s1 s2 w1 w2 w3 w4 w5 w6 w7 w8 w9 w10 w1-w12: weeks of treatment w13 & w14: run-out post treatment SGA = Subjects Global Assessment (100mm visual analog scale [VAS]). Linear mixed models: treatment and time as categorical and continuous independent variables, respectively. A time-by-treatment effect was added to the test difference in evolution over time (linear slopes) between both groups. Ebastin (linear slope effect = -1.35, p<0.0001), placebo (slope -0.29, p=0.4) Ebastin resulted in a significant reduction in abdominal pain (P=.03) run-out w11 w12 w13 w14 32
33 Results SGA of relief - % with at least considerable relief Figure 1B. N=26 Placebo N=24 Ebastin 50 * follow up % of patients with at least considerable relief w1 w2 w3 w4 w5 w6 w7 w8 w9 w10 w11 w12 w13 w14 Weeks of treatment w1-w12 Post treatment w13 & w14 Figure 1B. Percentage of patients with at least considerable relief of global symptoms SGA = Subjects Global Assessment; *p<0.05x2-test. Significantly more patients treated with ebastin vs. placebo had at least considerable relief of symptoms (46% vs 12%; P=0.001) No difference in other GI symptoms by the GSRS IBS-QOL scores showed improvement with ebastin vs. placebo 33
34 Conclusions Trial failed to show a significant effect on visceral perception assessed by rectal distension Ebastin resulted in a significant improvement in global symptom relief, abdominal pain, and quality of life compared to placebo These data suggest a poor relationship between barostat findings and clinical response Study suggests that H1-receptor blockade may represent an effective treatment for IBS Ebastin is not available in the US 34
35 Randomized, Placebo-Controlled Trial of Glutamine for the Treatment of Diarrhea-Predominant IBS DD- #6 Basra S, Verne GN, Zhou Q. Abstract
36 Background and Methods Background IBS symptoms (abdominal pain, diarrhea, urgency) may be related to intestinal hyperpermeability IBS-D patients have decreased intestinal glutamine synthase levels Study design 8-week, randomized, double-blind, placebo-controlled study Patients 61 patients with IBS-D Patients completed a IBS Symptom Severity Scale (IBSS), lactulose/mannitol intestinal permeability test and intestinal biopsies for Claudin-1 expression before and after study Treatments Glutamine 10 g orally three times daily Placebo 36
37 Results Significant improvements (P<.01) were observed in: Abdominal pain Bloating Diarrhea Intestinal permeability was restored in patients who received glutamine (P<.05) A positive correlation was observed between improvement in IBS symptoms and colonic Caludin-1 expression following glutamine therapy (r=0.7) 37
38 Conclusions Oral glutamine supplementation improves gastrointestinal symptoms and restores normal intestinal permeability in D-IBS patients Improvements in symptoms and intestinal permeability correlated with Claudin-1 expression Glutamine may be a useful therapeutic agent to treat D-IBS patients who have intestinal hyperpermeability 38
39 Efficacy of Ibodutant, a Selective Antagonist of Neurokinin 2 Receptors, in IBS-D: Results of a Double- Blind, Placebo-Controlled, Parallel-Group Phase II Study (IRIS-2) LC- #7 Tack JF, Dochev YS, Bochenek A et al. Abstract
40 Background and Methods Background Ibodutant is a selective antagonist of neurokinin 2 (NK2) receptors in clinical development In animal models, ibodutant did not alter sensorimotor function, but reverted intestinal hypermotility and hyperalgesia 40
41 The NK2 Receptor: Role in Gastrointestinal Function G-protein-linked receptors Three distinct subtypes (neurokinin 1, 2, 3) Physiologic ligand: Tachykinins (excitatory neuropeptides) Substance P Neurokinin A Neurokinin B Sources of tachykinins Intrinsic neurons located in myenteric plexus, projecting to the muscle layers Intrinsic neurons located in the submucous plexus, projecting to the mucosa and submucosal blood vessels Vannuchi MG, Evangelista S. Biomolecular Concepts. 2013;4:
42 Methods Study design Multinational, double-blind, dose-finding, placebo controlled study with 2-week treatment-free run-in and 8 week treatment period Patients 559 patients with IBS-D (Rome III criteria)- 60% women 25% loose/watery stool, 3 stools/day, at least moderate abdominal pain/discomfort on 3 days during run-in period Treatments Placebo or ibodutant 1 mg, 3 mg, 10 mg once daily (1:1:1:1 ratio) Primary end point: Combined response of satisfactory relief of overall IBS symptoms and abdominal pain/discomfort (yes/no) in 6 of 8 weeks 42
43 Baseline Characteristics Parameter Mean age Mean abdominal pain intensity Mean number of bowel movements 46 years Moderate to severe 4.5/day Stool consistency* 5.7 *According to Bristol Stool Scale (Type 1 = separate hard lumps; Type 7 = entirely liquid) 43
44 Efficacy Results Trend toward increased response with escalating ibodutant dose Best efficacy with highest (10 mg) dose Patients (%) Responders by 75% Rule (Overall Population) * Placebo Ibodutant 1 mg Ibodutant 3 mg Ibodutant 10 mg *P=0.032 vs placebo (not significant after Hochberg correction for multiple testing). 44
45 Efficacy Results Responders by 75% Rule (Males vs Females) 50 Males Females * Patients (%) Placebo Ibodutant 1 mg Ibodutant 3 mg Ibodutant 10 mg *P=.003 vs placebo. 45
46 Safety 94% of patients completed the study Incidence of adverse events <10% 5 serious adverse events occurred 46
47 Conclusions The NK2 receptor plays a demonstrated role in modulating symptoms of IBS In this well-designed study, ibodutant demonstrated a dose-dependent trend toward efficacy in the overall population 75% rule end point difficult to achieve This effect was driven by positive outcomes in women, with little if any effect in men 47
48 Plecanatide, a Novel Guanylate Cyclase C (GC-C) Receptor Antagonist, is Efficacious and Safe in Patients With Chronic Idiopathic Constipation: Results from a 951-Patient, 12-Week, Multicenter Trial LC- #8 Miner PB, Surowitz R, Fogel R et al. Abstract 925G 48
49 Background and Methods Background Plecanatide is a GC-C receptor agonist; mimics effect of the natriuretic peptide uroguanylin Study design 12-week, randomized, placebo controlled study Patients 951 CIC patients (Rome III criteria) <3 complete spontaneous bowel movements (CSBMs)/week at baseline Treatments Plecanatide (0.3, 1, 3 mg) orally once daily Placebo once daily Primary end point Number of CSBMs/week Weekly responder: >3CSBMs/wk + increase of 1 CSBM from baseline 49
50 Efficacy Results PBO n=234 PLE 0.3 mg n=237 PLE 1 mg n=238 PLE 3 mg n=237 Responders for 9 weeks (%) * * Responders including last 3 of 4 weeks * ** Median time to first SBM (hours) * 21.3* 12.5* Change in CSBM frequency from baseline Change in SBM frequency from baseline * 1.76* 2.13* * 2.23* 2.88* Change in stool consistency (BSFQ) * 2.01* Change in straining score * -2.07* *Significant difference vs placebo. BSFQ=Bristol Stool Form Questionnaire 50
51 Safety Results PBO n=234 PLE 0.3 mg n=237 PLE 1 mg n=238 PLE 3 mg n=237 Serious adverse events 5 (2.1%) 1 (0.4%) 1 (0.4%) 2 (0.8%) Patients with TEAE(s) 96 (40.7%) 99 (41.8%) 103 (43.3%) 106 (44.7%) Diarrhea with TEAE(s) 3 (1.3%) 13 (5.5%) 20 (8.4%) 23 (9.7%) Patient withdrew due to TEAE Patient withdrew due to diarrhea 8 (3.4%) 9 (3.8%) 16 (6.7%) 13 (5.5%) 1 (0.4%) 2 (0.8%) 8 (3.4%) 7 (3.0%) TEAE=Treatment-emergent adverse event 51
52 Conclusions The 3-mg dose of plecanatide appears effective, safe, and well-tolerated Dose-related efficacy was shown in this trial 52
53 Qualitative Assessment of Symptom Experience in Patients With IBS for the Development of Patient-Reported Outcome Instruments LC- #9 Baird MJ, Carson R, Ervin C et al. Abstract Su
54 Background and Methods Background Aim There are currently no patient-reported outcome (PRO) measures that are accepted as well-defined and reliable by the FDA Conduct qualitative research with IBS patients to elicit their symptom experience in accordance with the FDA-mandated guidance for development of a PRO instrument Study design Face-to-face interviews conducted in a representative sample of patients with physician-confirmed IBS Patients Irritable bowel syndrome with diarrhea (IBS-D) (n=17) Irritable bowel syndrome with constipation (IBS-C) (n=14) Irritable bowel syndrome with mixed symptoms (IBS-M) (n=18) 54
55 Results: Spontaneously Reported IBS Concepts by Subtype with 50% Reporting Frequency Concepts Spontaneously Reported: n(%) IBS-D (n=17) IBS-C (n=14) IBS-M (n=18) Diarrhea: 17 (100%) Constipation: 14 (100%) Constipation: 18 (100%) Loose or Watery Stools: 16 (94%) Infrequent BMs: 14 (100%) Abdominal Pain: 17 (94%) Urgency: 15 (88%) Bloating: 13 (93%) Infrequent BMs: 17 (94%) Abdominal Pain: 15 (88%) Can t go: 12 (86%) Diarrhea: 16 (89%) Cramping: 12 (71%) Abdominal Pain: 11 (79%) Recurrent BMs: 16 (89%) Recurrent BMs: 12 (71%) Small Stools: 11 (79%) Loose or Watery Stools: 15 (83%) Too Frequent BMs: 12 (71%) Straining: 10 (71%) Too Frequent BMs: 14 (78%) Gas: 10 (59%) Gas: 9 (64%) Cramping: 13 (72%) Abdominal Discomfort: 9 (53%) Abdominal Discomfort: 9 (64%) Urgency: 13 (72%) BM = Bowel Movement Feeling of Fullness: 8 (57%) Can t go: 12 (67%) Hard Stools: 8 (57%) Long Time in Bathroom: 12 (67%) Unsuccessful Attempts: 12 (67%) Bloating: 11 (61%) Hard Stools: 11 (61%) Straining: 10 (56%) 55
56 Results: Summary of Single Most Bothersome IBS Symptom Symptom Number of Patients Reporting Symptom as the Most Bothersome IBS-D (n=17) IBS-C (n=14) a IBS-M (n=18) b Total (N=49) Abdominal Pain Urgency Cramping Bloating Infrequent BMs Abdominal discomfort Gas Too frequent BMs Accidents Loose or watery stools Hard Stools Recurrent BMs Vomitting Contractions BM = Bowel Movement a One IBS-C participant did not report a most bothersome symptom. b Two IBS-M participants reported two most bothersome symptoms. 56
57 Conclusions IBS patients report a range of gastrointestinal symptoms that includes but is not limited to abdominal pain and abnormal stool consistency and frequency This is not a test of a proposed PRO instrument Preliminary step in identifying symptom domains that will determine which items are developed for the PRO None of the findings are surprising Pain, urgency, cramping, and bloating are likely to be heavily weighted in the eventual PRO instrument Sample size is relatively small given the implications for clinical trials 57
58 Efficacy and Safety of Naloxegol in Patients With Opioid- Induced Constipation: Results from 2 Prospective, Randomized, Controlled Trials DD- #10 Chey WD, Webster L, Sostek M et al. Abstract
59 Study Design (KODIAC-04 and KODIAC-05) Screening Period (5 14 days) OIC Confirmation Period* (14 days) Randomized Treatment Period (12 weeks) Posttreatment (2 weeks) Placebo Naloxegol 12.5 mg/day Naloxegol 25 mg/day (Visit 1) (Visit 2) (Visits 3 8) Randomization (1:1:1) (Visit 3) EOT (Visit 8) Follow-up (Visit 9) Outpatients with OIC aged 18 to <85 years receiving oral morphine equivalent: mg/day for 4 weeks before screening for noncancer pain *Diagnosis of OIC and opioid dose stability confirmed by ediary (provided at screening). Bisacodyl (10 15 mg; maximum of 3 doses/episode) and one-time use enema permitted as rescue. Patients could either complete the follow-up visit or enroll in a safety extension study. EOT=end of treatment; OIC=opioid-induced constipation. 59
60 Patients (%) Primary Endpoint: Responder Rates (Weeks 1 12) ITT Population Placebo Naloxegol 12.5 mg/day Naloxegol 25 mg/day 40.8 * n=214kodiac-04 n=213 n=214 n=232 KODIAC-05 n=232 n=232 P vs placebo No. needed to treat NS 9.7 Primary efficacy endpoint: 3 SBM/week with 1 SBM/week increase over baseline for 9 of the 12 weeks and 3 of the last 4 weeks of treatment *Significant vs placebo under the multiple testing procedure. Calculated as (% responders naloxegol % responders placebo). 60
61 Safety KODIAC-04 KODIAC-05 Any adverse event (AE)* Discontinuation due to an AE Placebo Naloxegol 12.5 mg/day Naloxegol 25 mg/day Placebo Naloxegol 12.5 mg/day Naloxegol 25 mg/day 46.9% 49.3% 61.2% 58.9% 59.6% 69.0% 5.6% 4.3% 10.3% 5.2% 5.2% 10.3% Deaths 0% 0.9% 0% 0% 0% 0% Serious AE 5.2% 5.2% 3.3% 5.2% 6.1% 3.4% *Most frequent adverse events were gastrointestinal-related 61
62 Conclusions In patients with OIC, naloxegol treatment increased the frequency of SBMs and resulted in significantly more OIC responders vs placebo This effect was maintained over the 12-week treatment period There was no reversal or reduction of opioid-mediated analgesia as reflected by daily pain scores and opioid dose Naloxegol was generally well tolerated and safe with the most commonly reported AEs were GI-related and were found more in the higher dose of naloxegol Naloxegol is in late-phase clinical development 62
63 Dyssynergic Defecation Can Be Diagnosed by Questionnaire and Physical Examination LC- #11 Chiarioni G, Kim SM, Whitehead WE et al. Abstract Sa
64 Background and Methods Background Dyssynergic defecation (DD) has a distinct pathologic mechanism vs other subtypes of constipation Diagnosis currently depends on costly, invasive tests, not widely available Aim Develop a questionnaire-based methodology for diagnosing dyssynergic defecation Patients 238 patients with refractory constipation Assessments Questionnaire and physical exam (PE) Anorectal manometry (ARM) Balloon evacuation test (BET) Defecography 64
65 Methods Mechanical obstruction (MO) if rectal prolapse on physical exam or abnormal defecography + BET >2 min DD if paradoxical contraction or failure to relax pelvic floor muscles on ARM + BET >2 min Slow transit constipation (STC) if >5/24 sitzmarkers remained 5 days after ingestion + BET >2 min Normal transit constipation is absence of MO, DD and STC Predictive variables were symptoms and physical exam findings 65
66 Results 238 patients divided into 2 groups of 119- test sample and validation sample Avg age= 45 yrs, 92% women DD=102, STC= 31, MP=37, NTC=63, 5 not classifiable Best predictor of DD was question on which muscles used to defecate- response of anus identified 91% of DD PE finding of anal sphincter relaxing on straining correctly identified 97.8% of patients without DD Learning Sample Validation Sample Sensitivity Specificity Sensitivity Specificity Questionnaire: Incomplete Evacuation Anal Blockage Digital Maneuvers Squeeze anus to defecate Physical Examination: Abd wall contracts to defecate Anal sphincter relaxes to defecate
67 Conclusions Asking patients which muscles they use to defecate has an overall accuracy of 87% for distinguishing patients with dyssynergic defecation from those with other types of constipation This question could be used in epidemiological studies to estimate the prevalence of dyssynergic defecation, in primary care to guide treatment, and in pharmaceutical trials to select patients for inclusion 67
68 Epidemiology of Fecal Incontinence in US Adults From 2005 to 2010: Prevalence, Trends, and Risk Factors LC- #12 Ditah IC, Devaki P, Jaiyeoba CO et al. Abstract Sa
69 Background and Methods Background Fecal incontinence (FI) is a common condition that can lead to poor self-image, social isolation and poor QOL The prevalence of fecal incontinence may be increasing especially with our aging population Aim To estimate the prevalence, analyze trends and identify risk factors for FI among non-industrialized US adults from Study design Data from NHANES (National Health and Nutrition Examination Survey) used to estimate prevalence FI Participants 14,759 participants in NHANES 69
70 Overall Prevalence of Fecal Incontinence / / /
71 Additional Results Fecal incontinence occurred weekly in 1.13% of study participants Fecal incontinence was statistically significantly more common in women (9.45%) than men (7.27%) Fecal incontinence increased statistically significantly with age 2.91% in subjects aged 20 to 29 years 16.16% among those aged 70 years Independent risk factors for FI: >55 years, diabetes, female gender, poor health status, urge incontinence, non-hispanic whites, >21 stools/week and loose/watery stools 71
72 Risk Factors for Fecal Incontinence Risk Factor Univariate analysis P value Multivariate analysis P value Age Groups (yrs) Baseline (1.21, 2.40) (0.50, 1.75) > (2.24, 4.34) < (0.84, 2.69) > (3.49, 5.95) < (1.12, 3.54) 0.02 > (4.73, 8.76) < (1.10, 4.36) Body Mass Index <25 Baseline (0.79, 1.14) > (0.65, 1.25) >0.05 > (1.08, 1.56) (0.76, 1.41) >0.05 Self rated health Good Baseline Poor 2.14 (1.79, 2.55) < (1.14, 1.81) Gender Male Baseline Female 1.28 (1.11, 1.47) (0.51, 0.89) Ethnicity Hispanic Baseline Non Hispanic White 1.15 (0.98, 1.35) > (1.16, 2.26) Non Hispanic Black 1.12 (0.89, 1.41) > (0.73, 1.74) >0.05 Urinary Incontinence No Baseline Yes 1.65 (1.27, 2.14) < (1.23, 2.22) Diabetes No Baseline Yes 1.20 (1.06, 1.35) (1.17, 2.17) Frequency of Bowel Movement/week < 3 Baseline (0.50, 0.90) (0.59, 1.51) >0.05 > (1.32, 2.59) (1.42, 4.28) Stool consistency Usual Baseline Hard/lumpy 1.11 (0.82, 1.50) > (0.51, 1.44) >0.05 Loose/watery 3.52 (2.84, 4.36) < (1.76, 3.53) <
73 Conclusions Fecal incontinence is a prevalent problem among noninstitutionalized US adults, and rose steadily from 2005 to 2010 Age is a strong risk factor Diabetes and chronic diarrhea are potential modifiable risk factors This is not the largest population-based study of fecal incontinence, but it is the most representative of the overall population Limitation: Study pooled males and females in multivariate analyses despite substantial differences in risk factors between genders 73
Emerging Treatments for IBS-C and Clinical Trial Endpoints
 Emerging Treatments for IBS-C and Clinical Trial Endpoints Lin Chang, M.D. Oppenheimer Family Center for Neurobiology of Stress David Geffen School of Medicine at UCLA Learning Objectives Describe current
Emerging Treatments for IBS-C and Clinical Trial Endpoints Lin Chang, M.D. Oppenheimer Family Center for Neurobiology of Stress David Geffen School of Medicine at UCLA Learning Objectives Describe current
William Chey, MD University of Michigan Ann Arbor, MI
 Lin Chang, MD David Geffen School of Medicine at UCLA Los Angeles, CA William Chey, MD University of Michigan Ann Arbor, MI Mark Pimentel, MD Cedars-Sinai Medical Center Los Angeles, CA Accredited by Jointly
Lin Chang, MD David Geffen School of Medicine at UCLA Los Angeles, CA William Chey, MD University of Michigan Ann Arbor, MI Mark Pimentel, MD Cedars-Sinai Medical Center Los Angeles, CA Accredited by Jointly
SUPPLEMENTARY INFORMATION Associated with
 Table1: Rome III and Rome IV diagnostic criteria for IBS, functional constipation and functional dyspepsia. Rome III diagnostic criteria 1,2 Rome IV diagnostic criteria 3,4 Diagnostic criteria for IBS
Table1: Rome III and Rome IV diagnostic criteria for IBS, functional constipation and functional dyspepsia. Rome III diagnostic criteria 1,2 Rome IV diagnostic criteria 3,4 Diagnostic criteria for IBS
IBS: overview and assessment of pain outcomes and implications for inclusion criteria
 IBS: overview and assessment of pain outcomes and implications for inclusion criteria William D. Chey, MD Professor of Medicine University of Michigan What is the Irritable Bowel Syndrome Symptom based
IBS: overview and assessment of pain outcomes and implications for inclusion criteria William D. Chey, MD Professor of Medicine University of Michigan What is the Irritable Bowel Syndrome Symptom based
Nicholas J. Talley, MD University of Newcastle Callaghan, NSW Australia. Mark Pimentel, MD Cedars-Sinai Medical Center Los Angeles, CA
 Lin Chang, MD David Geffen School of Medicine at UCLA Los Angeles, CA Mark Pimentel, MD Cedars-Sinai Medical Center Los Angeles, CA Nicholas J. Talley, MD University of Newcastle Callaghan, NSW Australia
Lin Chang, MD David Geffen School of Medicine at UCLA Los Angeles, CA Mark Pimentel, MD Cedars-Sinai Medical Center Los Angeles, CA Nicholas J. Talley, MD University of Newcastle Callaghan, NSW Australia
CHAPTER 11 Functional Gastrointestinal Disorders (FGID) Mr. Ashok Kumar Dept of Pharmacy Practice SRM College of Pharmacy SRM University
 CHAPTER 11 Functional Gastrointestinal Disorders (FGID) Mr. Ashok Kumar Dept of Pharmacy Practice SRM College of Pharmacy SRM University 1 Definition of FGID Chronic and recurrent symptoms of the gastrointestinal
CHAPTER 11 Functional Gastrointestinal Disorders (FGID) Mr. Ashok Kumar Dept of Pharmacy Practice SRM College of Pharmacy SRM University 1 Definition of FGID Chronic and recurrent symptoms of the gastrointestinal
Oral Methylnaltrexone for the. Constipation in Patients with Chronic Non-cancer Pain
 Oral Methylnaltrexone for the Treatment of Opioid-induced Constipation in Patients with Chronic Non-cancer Pain Richard L. Rauck, 1 John F. Peppin, 2 Robert J.Israel, 3 Jennifer Carpenito, 3 Jeffrey Cohn,
Oral Methylnaltrexone for the Treatment of Opioid-induced Constipation in Patients with Chronic Non-cancer Pain Richard L. Rauck, 1 John F. Peppin, 2 Robert J.Israel, 3 Jennifer Carpenito, 3 Jeffrey Cohn,
Outline. Definition (s) Epidemiology Pathophysiology Management With an emphasis on recent developments
 Chronic Dyspepsia Eamonn M M Quigley MD FRCP FACP MACG FRCPI Lynda K and David M Underwood Center for Digestive Disorders Houston Methodist Hospital Houston, Texas Outline Definition (s) Epidemiology Pathophysiology
Chronic Dyspepsia Eamonn M M Quigley MD FRCP FACP MACG FRCPI Lynda K and David M Underwood Center for Digestive Disorders Houston Methodist Hospital Houston, Texas Outline Definition (s) Epidemiology Pathophysiology
Biofeedback for Pelvic Floor Disorders and Incontinence
 The UNC Center for Functional GI & Motility Disorders www.med.unc.edu/ibs Biofeedback for Pelvic Floor Disorders and Incontinence Olafur S. Palsson, Psy.D. Associate Professor of Medicine UNC Center for
The UNC Center for Functional GI & Motility Disorders www.med.unc.edu/ibs Biofeedback for Pelvic Floor Disorders and Incontinence Olafur S. Palsson, Psy.D. Associate Professor of Medicine UNC Center for
Irritable Bowel Syndrome. Mustafa Giaffer March 2017
 Irritable Bowel Syndrome Mustafa Giaffer March 2017 Introduction First described in 1771. 50% of patients present
Irritable Bowel Syndrome Mustafa Giaffer March 2017 Introduction First described in 1771. 50% of patients present
Functional Dyspepsia
 Functional Dyspepsia American College of Gastroenterology Boston Massachusetts, June 2015 Brian E. Lacy, PhD, MD, FACG Professor of Medicine Geisel School of Medicine at Dartmouth Chief, Section of Gastroenterology
Functional Dyspepsia American College of Gastroenterology Boston Massachusetts, June 2015 Brian E. Lacy, PhD, MD, FACG Professor of Medicine Geisel School of Medicine at Dartmouth Chief, Section of Gastroenterology
UBS Global Healthcare Conference May 19, 2014
 UBS Global Healthcare Conference May 19, 2014 Safe Harbor Statement This presentation may contain forward-looking statements within the meaning of Section 27A of the Securities Act of 1933 and Section
UBS Global Healthcare Conference May 19, 2014 Safe Harbor Statement This presentation may contain forward-looking statements within the meaning of Section 27A of the Securities Act of 1933 and Section
WARNING: RISK OF SERIOUS DEHYDRATION IN PEDIATRIC PATIENTS
 June 5, 2018 Synergy Pharmaceuticals Highlights New Data at Digestive Disease Week (DDW) 2018 Linking Uroguanylin Deficiency to Chronic Idiopathic Constipation (CIC) and Irritable Bowel Syndrome with Constipation
June 5, 2018 Synergy Pharmaceuticals Highlights New Data at Digestive Disease Week (DDW) 2018 Linking Uroguanylin Deficiency to Chronic Idiopathic Constipation (CIC) and Irritable Bowel Syndrome with Constipation
Effects of baseline abdominal pain and bloating on response to lubiprostone in patients with irritable bowel syndrome with constipation
 Alimentary Pharmacology and Therapeutics Effects of baseline abdominal pain and bloating on response to lubiprostone in patients with irritable bowel syndrome with constipation L. Chang*, W. D. Chey, D.
Alimentary Pharmacology and Therapeutics Effects of baseline abdominal pain and bloating on response to lubiprostone in patients with irritable bowel syndrome with constipation L. Chang*, W. D. Chey, D.
Functional Heartburn and Dyspepsia
 Functional Heartburn and Dyspepsia Nicholas Shaheen, MD, MPH Center for Esophageal Diseases and Swallowing University of North Carolina Objectives Understand the means of diagnosing functional heartburn
Functional Heartburn and Dyspepsia Nicholas Shaheen, MD, MPH Center for Esophageal Diseases and Swallowing University of North Carolina Objectives Understand the means of diagnosing functional heartburn
Opioid-Induced Constipation
 Objectives Opioid-Induced Constipation Brianna Jansma, PharmD Alex Smith, PharmD Megan Robinson, PharmD Summarize epidemiology of opioid-induced constipation (OIC) Understand opiates effects on the gastrointestinal
Objectives Opioid-Induced Constipation Brianna Jansma, PharmD Alex Smith, PharmD Megan Robinson, PharmD Summarize epidemiology of opioid-induced constipation (OIC) Understand opiates effects on the gastrointestinal
June By: Reza Gholami
 ACG/CAG guideline on Management of Dyspepsia June 2017 By: Reza Gholami DEFINITION OF DYSPEPSIA AND SCOPE OF THE GUIDELINE Dyspepsia was originally defined as any symptoms referable to the upper gastrointestinal
ACG/CAG guideline on Management of Dyspepsia June 2017 By: Reza Gholami DEFINITION OF DYSPEPSIA AND SCOPE OF THE GUIDELINE Dyspepsia was originally defined as any symptoms referable to the upper gastrointestinal
Number of studies. Endoscopic finding. Number of subjects. Pooled prevalence 95% CI
 Clinical Approach to the Patient t with Dyspepsia William D. Chey, MD, FACG Professor of Medicine University of Michigan Prevalence of Endoscopic Findings in Individuals with Dyspepsia Systematic Review
Clinical Approach to the Patient t with Dyspepsia William D. Chey, MD, FACG Professor of Medicine University of Michigan Prevalence of Endoscopic Findings in Individuals with Dyspepsia Systematic Review
OPIOID-INDUCED CONSTIPATION DR ANDREW DAVIES
 OPIOID-INDUCED CONSTIPATION DR ANDREW DAVIES Introduction Introduction Mean faecal weight 128 g / cap / day Mean range 51-796 g Absolute range 15-1505 g Main factors affecting mass are caloric intake,
OPIOID-INDUCED CONSTIPATION DR ANDREW DAVIES Introduction Introduction Mean faecal weight 128 g / cap / day Mean range 51-796 g Absolute range 15-1505 g Main factors affecting mass are caloric intake,
Daniel Canafax, PharmD VP, Clinical Research Theravance, Inc.
 Demonstrates Improvement in Bowel Movement Frequency and Bristol Stool Scores in a Phase 2b Study of Patients with Opioid-Induced Constipation (OIC) Ross Vickery, PhD, 1 Yu-Ping Li, PhD, 1 Ullrich Schwertschlag,
Demonstrates Improvement in Bowel Movement Frequency and Bristol Stool Scores in a Phase 2b Study of Patients with Opioid-Induced Constipation (OIC) Ross Vickery, PhD, 1 Yu-Ping Li, PhD, 1 Ullrich Schwertschlag,
OCTOBER 7-10 PHILADELPHIA, PENNSYLVANIA
 OMED 17 OCTOBER 7-10 PHILADELPHIA, PENNSYLVANIA 29.5 Category 1-A CME credits anticipated ACOFP / AOA s 122 nd Annual Osteopathic Medical Conference & Exposition Joint Session with ACOFP and Cleveland
OMED 17 OCTOBER 7-10 PHILADELPHIA, PENNSYLVANIA 29.5 Category 1-A CME credits anticipated ACOFP / AOA s 122 nd Annual Osteopathic Medical Conference & Exposition Joint Session with ACOFP and Cleveland
Elderly Man With Chronic Constipation
 Elderly Man With Chronic Constipation Linda Nguyen, MD Director, Neurogastroenterology and Motility Clinical Assistant Professor Stanford University Overview Normal bowel function Defining Constipation:
Elderly Man With Chronic Constipation Linda Nguyen, MD Director, Neurogastroenterology and Motility Clinical Assistant Professor Stanford University Overview Normal bowel function Defining Constipation:
Xifaxan, Lotronex and Viberzi Prior Authorization and Quantity Limit Program Summary
 Xifaxan, Lotronex and Viberzi Prior Authorization and Quantity Limit Program Summary FDA APPROVED INDICATIONS DOSAGE 1,2 Lotronex (alosetron) a Indication For women with severe diarrheapredominant irritable
Xifaxan, Lotronex and Viberzi Prior Authorization and Quantity Limit Program Summary FDA APPROVED INDICATIONS DOSAGE 1,2 Lotronex (alosetron) a Indication For women with severe diarrheapredominant irritable
Synergy Pharmaceuticals TRULANCE (Plecanatide) Receives U.S. FDA Approval for the Treatment of Adults with Chronic Idiopathic Constipation
 January 19, 2017 Synergy Pharmaceuticals TRULANCE (Plecanatide) Receives U.S. FDA Approval for the Treatment of Adults with Chronic Idiopathic Constipation NEW YORK--(BUSINESS WIRE)-- Synergy Pharmaceuticals
January 19, 2017 Synergy Pharmaceuticals TRULANCE (Plecanatide) Receives U.S. FDA Approval for the Treatment of Adults with Chronic Idiopathic Constipation NEW YORK--(BUSINESS WIRE)-- Synergy Pharmaceuticals
Constipation An Overview. Definition Physiology of GI tract Etiology Assessment Treatment
 CONSTIPATION Constipation An Overview Definition Physiology of GI tract Etiology Assessment Treatment Definition Constipation = the infrequent passage of hard feces Definition of Infrequent The meaning
CONSTIPATION Constipation An Overview Definition Physiology of GI tract Etiology Assessment Treatment Definition Constipation = the infrequent passage of hard feces Definition of Infrequent The meaning
William D Chey, 1 Anthony J Lembo, 2 James A Phillips, 3 David P Rosenbaum 4
 Efficacy and safety of tenapanor in patients with constipationpredominant irritable bowel syndrome: a 12-week, double-blind, placebocontrolled, randomized phase 2b trial William D Chey, 1 Anthony J Lembo,
Efficacy and safety of tenapanor in patients with constipationpredominant irritable bowel syndrome: a 12-week, double-blind, placebocontrolled, randomized phase 2b trial William D Chey, 1 Anthony J Lembo,
Our evidence. Your expertise. SmartPill : The data you need to evaluate motility disorders.
 Our evidence. Your expertise. SmartPill : The data you need to evaluate motility disorders. SmartPill benefits your practice: Convenient performed right in your office Test standardization Provides direct
Our evidence. Your expertise. SmartPill : The data you need to evaluate motility disorders. SmartPill benefits your practice: Convenient performed right in your office Test standardization Provides direct
Bloating, Flatulence, and
 A 45-Year-Old Man With Recurrent Abdominal Pain, Bloating, Flatulence, and Intermittent Loose Stools Anthony J. Lembo, MD Associate Professor of Medicine Harvard Medical School Director, GI Motility Laboratory
A 45-Year-Old Man With Recurrent Abdominal Pain, Bloating, Flatulence, and Intermittent Loose Stools Anthony J. Lembo, MD Associate Professor of Medicine Harvard Medical School Director, GI Motility Laboratory
Efficacy and Safety of Lubiprostone. Laura Wozniak February 23, 2010 K30 Monthly Journal Club
 Efficacy and Safety of Lubiprostone Laura Wozniak February 23, 2010 K30 Monthly Journal Club Objectives Brief overview of constipation Review of article Discussion Constipation in Children 3-5% of all
Efficacy and Safety of Lubiprostone Laura Wozniak February 23, 2010 K30 Monthly Journal Club Objectives Brief overview of constipation Review of article Discussion Constipation in Children 3-5% of all
PELVIC PAIN : Gastroenterological Conditions
 PELVIC PAIN : Gastroenterological Conditions Departman Tarih Prof. A. Melih OZEL, MD Department of Gastroenterology Anadolu Medical Center Hospital Gebze Kocaeli - TURKEY Presentation plan 15 min. Introduction
PELVIC PAIN : Gastroenterological Conditions Departman Tarih Prof. A. Melih OZEL, MD Department of Gastroenterology Anadolu Medical Center Hospital Gebze Kocaeli - TURKEY Presentation plan 15 min. Introduction
Evidence-based Treatment Strategies for
 Evidence-based Treatment Strategies for Chronic Constipation William D. Chey, MD Professor of Medicine University of Michigan Rome III criteria*: Chronic constipation Must include 2 of the following (>25%
Evidence-based Treatment Strategies for Chronic Constipation William D. Chey, MD Professor of Medicine University of Michigan Rome III criteria*: Chronic constipation Must include 2 of the following (>25%
... SELECTED ABSTRACTS...
 ... SELECTED ABSTRACTS... The following abstracts, from medical journals containing literature on irritable bowel syndrome, were selected for their relevance to this supplement. A Technical Review for
... SELECTED ABSTRACTS... The following abstracts, from medical journals containing literature on irritable bowel syndrome, were selected for their relevance to this supplement. A Technical Review for
SYMPROIC (naldemedine tosylate) oral capsule
 SYMPROIC (naldemedine tosylate) oral capsule Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This
SYMPROIC (naldemedine tosylate) oral capsule Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This
Slide #43. Functional Disorders - An Update 11/8/ MA ACP Annual Scientific Meeting. Functional Disorders: An Update
 Functional Disorders: An Update Anthony Lembo, M.D. Associate Professor of Medicine Beth Israel Deaconess Medical Center Harvard Medical School Boston, MA Disclosure of Financial Relationships Anthony
Functional Disorders: An Update Anthony Lembo, M.D. Associate Professor of Medicine Beth Israel Deaconess Medical Center Harvard Medical School Boston, MA Disclosure of Financial Relationships Anthony
What Is Constipation?
 CONSTIPATION What Is Constipation? Constipation is when you have infrequent or hard-to-pass bowel movements (meaning they are painful or you have to strain), have hard stools or feel like your bowel movements
CONSTIPATION What Is Constipation? Constipation is when you have infrequent or hard-to-pass bowel movements (meaning they are painful or you have to strain), have hard stools or feel like your bowel movements
FUNCTIONAL DISORDERS ROME IV CRITERIA AND DIAGNOSTIC QUESTIONNAIRE DDW 2016,REVIEW. Presented by; Marjan Mokhtare Gastroenterologist, IUMS
 FUNCTIONAL DISORDERS ROME IV CRITERIA AND DIAGNOSTIC QUESTIONNAIRE DDW 2016,REVIEW Presented by; Marjan Mokhtare Gastroenterologist, IUMS Functional gastrointestinal disorders (FGIDs), the most common
FUNCTIONAL DISORDERS ROME IV CRITERIA AND DIAGNOSTIC QUESTIONNAIRE DDW 2016,REVIEW Presented by; Marjan Mokhtare Gastroenterologist, IUMS Functional gastrointestinal disorders (FGIDs), the most common
IBS - Definition. Chronic functional disorder of GI generally characterized by:
 IBS - Definition Chronic functional disorder of GI generally characterized by: 3500 3000 No. of Publications 2500 2000 1500 1000 Irritable Bowel syndrome Irritable Bowel Syndrome 500 0 1968-1977 1978-1987
IBS - Definition Chronic functional disorder of GI generally characterized by: 3500 3000 No. of Publications 2500 2000 1500 1000 Irritable Bowel syndrome Irritable Bowel Syndrome 500 0 1968-1977 1978-1987
ACG Clinical Guideline: Management of Benign Anorectal Disorders
 ACG Clinical Guideline: Management of Benign Anorectal Disorders Arnold Wald, MD, MACG 1, Adil E. Bharucha, MBBS, MD 2, Bard C. Cosman, MD, MPH, FASCRS 3 and William E. Whitehead, PhD, MACG 4 1 Division
ACG Clinical Guideline: Management of Benign Anorectal Disorders Arnold Wald, MD, MACG 1, Adil E. Bharucha, MBBS, MD 2, Bard C. Cosman, MD, MPH, FASCRS 3 and William E. Whitehead, PhD, MACG 4 1 Division
Drossman Gastroenterology 55 Vilcom Center Drive Boyd Hall, Suite 110 Chapel Hill, NC 27514
 Drossman Gastroenterology 55 Vilcom Center Drive Boyd Hall, Suite 110 Chapel Hill, NC 27514 Normal Gastrointestinal Motility and Function "Motility" is an unfamiliar word to many people; it is used primarily
Drossman Gastroenterology 55 Vilcom Center Drive Boyd Hall, Suite 110 Chapel Hill, NC 27514 Normal Gastrointestinal Motility and Function "Motility" is an unfamiliar word to many people; it is used primarily
UNDERSTANDING IBS AND CC Implications for diagnosis and management
 UNDERSTANDING IBS AND CC Implications for diagnosis and management J. TACK, M.D., Ph.D. Department of Gastroenterology University Hospitals, K.U. Leuven Leuven, Belgium TYPES OF GASTROINTESTINAL DISORDERS
UNDERSTANDING IBS AND CC Implications for diagnosis and management J. TACK, M.D., Ph.D. Department of Gastroenterology University Hospitals, K.U. Leuven Leuven, Belgium TYPES OF GASTROINTESTINAL DISORDERS
Common Gastrointestinal Problems in the Elderly
 Common Gastrointestinal Problems in the Elderly Brian Viviano, D.O. Objectives Understand the pathophysiology, clinical manifestations, diagnosis and management of GI diseases of the elderly. Differentiate
Common Gastrointestinal Problems in the Elderly Brian Viviano, D.O. Objectives Understand the pathophysiology, clinical manifestations, diagnosis and management of GI diseases of the elderly. Differentiate
Irritable Bowel Syndrome Now. George M. Logan, MD Friday, May 5, :35 4:05 PM
 Irritable Bowel Syndrome Now George M. Logan, MD Friday, May 5, 2017 3:35 4:05 PM Dr. Logan indicated no potential conflict of interest to this presentation. He does not intend to discuss any unapproved/investigative
Irritable Bowel Syndrome Now George M. Logan, MD Friday, May 5, 2017 3:35 4:05 PM Dr. Logan indicated no potential conflict of interest to this presentation. He does not intend to discuss any unapproved/investigative
A Guide to Gastrointestinal Motility Disorders
 A Guide to Gastrointestinal Motility Disorders Albert J. Bredenoord André Smout Jan Tack A Guide to Gastrointestinal Motility Disorders Albert J. Bredenoord Gastroenterology and Hepatology Academic Medical
A Guide to Gastrointestinal Motility Disorders Albert J. Bredenoord André Smout Jan Tack A Guide to Gastrointestinal Motility Disorders Albert J. Bredenoord Gastroenterology and Hepatology Academic Medical
Constipation Information Leaflet THE DIGESTIVE SYSTEM. gutscharity.org.uk
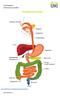 THE DIGESTIVE SYSTEM http://healthfavo.com/digestive-system-for-kids.html This factsheet is about Constipation Constipation is a symptom that can mean different things to different people but the usual
THE DIGESTIVE SYSTEM http://healthfavo.com/digestive-system-for-kids.html This factsheet is about Constipation Constipation is a symptom that can mean different things to different people but the usual
Lessons learned along the path to qualification of an IBS outcome measure*
 Lessons learned along the path to qualification of an IBS outcome measure* Stephen Joel Coons, PhD Patient-Reported Outcome (PRO) Consortium Critical Path Institute IMMPACT-XX WASHINGTON, DC July 14, 2017
Lessons learned along the path to qualification of an IBS outcome measure* Stephen Joel Coons, PhD Patient-Reported Outcome (PRO) Consortium Critical Path Institute IMMPACT-XX WASHINGTON, DC July 14, 2017
PELVIC PAIN : Gastroenterological Conditions
 PELVIC PAIN : Gastroenterological Conditions Departman Tarih Prof. A. Melih OZEL, MD Department of Gastroenterology Anadolu Medical Center Hospital Gebze Kocaeli - TURKEY Presentation plan 15 min. Introduction
PELVIC PAIN : Gastroenterological Conditions Departman Tarih Prof. A. Melih OZEL, MD Department of Gastroenterology Anadolu Medical Center Hospital Gebze Kocaeli - TURKEY Presentation plan 15 min. Introduction
PRODUCT MONOGRAPH. Linaclotide capsules. 72 mcg, 145 mcg and 290 mcg linaclotide. Guanylate Cyclase-C Agonist
 PRODUCT MONOGRAPH Pr CONSTELLA Linaclotide capsules 72 mcg, 145 mcg and 290 mcg linaclotide Guanylate Cyclase-C Agonist Allergan Inc. 85 Enterprise Blvd., Suite 500 Markham, Ontario L6G 0B5 Date of Preparation:
PRODUCT MONOGRAPH Pr CONSTELLA Linaclotide capsules 72 mcg, 145 mcg and 290 mcg linaclotide Guanylate Cyclase-C Agonist Allergan Inc. 85 Enterprise Blvd., Suite 500 Markham, Ontario L6G 0B5 Date of Preparation:
David Leff, DO. April 13, Disclosure. I have the following financial relationships to disclosure:
 David Leff, DO AOMA 94 th Annual Convention April 13, 2016 Disclosure I have the following financial relationships to disclosure: Speaker s Bureau: Allergan Labs, Takeda Pharmaceutical, Valeant Pharmaceutical
David Leff, DO AOMA 94 th Annual Convention April 13, 2016 Disclosure I have the following financial relationships to disclosure: Speaker s Bureau: Allergan Labs, Takeda Pharmaceutical, Valeant Pharmaceutical
Objectives. Identify age-related changes in the gastrointestinal tract
 The Primary Care Physician s Approach to Gastrointestinal Complaints in the Elderly Brian Viviano, D.O. Medical Associates of Erie Objectives Identify age-related changes in the gastrointestinal tract
The Primary Care Physician s Approach to Gastrointestinal Complaints in the Elderly Brian Viviano, D.O. Medical Associates of Erie Objectives Identify age-related changes in the gastrointestinal tract
Irritable Bowel Syndrome and Chronic Constipation
 Title slide - part 1 Irritable Bowel Syndrome and Chronic Constipation IBS - Physiologic Research Stress affects GI function Meals Pain / motility Time Line of Physiologic Research in IBS Pain sensitivit
Title slide - part 1 Irritable Bowel Syndrome and Chronic Constipation IBS - Physiologic Research Stress affects GI function Meals Pain / motility Time Line of Physiologic Research in IBS Pain sensitivit
Intestinal, non-intestinal, and extra-digestive response to linaclotide in patients with IBS-C: results at Week 4 predict sustained response
 Intestinal, non-intestinal, and extra-digestive response to linaclotide in patients with IBS-C: results at Week 4 predict sustained response Blanca Serrano, 1 Silvia Delgado-Aros, 2 Fermín Mearin, 3 Constanza
Intestinal, non-intestinal, and extra-digestive response to linaclotide in patients with IBS-C: results at Week 4 predict sustained response Blanca Serrano, 1 Silvia Delgado-Aros, 2 Fermín Mearin, 3 Constanza
Definition, Pathogenesis, and Management of That Cursed Dyspepsia
 Clinical Gastroenterology and Hepatology 2018;16:467 479 Definition, Pathogenesis, and Management of That Cursed Dyspepsia Pramoda Koduru, Malcolm Irani, and Eamonn M. M. Quigley IM F1 김영기 Dyspepsia Umbrella
Clinical Gastroenterology and Hepatology 2018;16:467 479 Definition, Pathogenesis, and Management of That Cursed Dyspepsia Pramoda Koduru, Malcolm Irani, and Eamonn M. M. Quigley IM F1 김영기 Dyspepsia Umbrella
Constipation: pathophysiology and management
 REVIEW C URRENT OPINION Constipation: pathophysiology and management Arnold Wald Purpose of review Continuing advances in pharmaceutical development are providing an expanding array of treatment approaches
REVIEW C URRENT OPINION Constipation: pathophysiology and management Arnold Wald Purpose of review Continuing advances in pharmaceutical development are providing an expanding array of treatment approaches
Chronic constipation in the elderly
 Chronic constipation in the elderly 1 Dec,2011 R 2 Natta Asanaleykha Epidemiology Definition Scope The impact of chronic constipation in the elderly Pathophysiology Evaluation the elderly patient with
Chronic constipation in the elderly 1 Dec,2011 R 2 Natta Asanaleykha Epidemiology Definition Scope The impact of chronic constipation in the elderly Pathophysiology Evaluation the elderly patient with
Evaluation of Efficacy Variables in Clinical Study of Irritable Bowel Syndrome with Diarrhea
 Evaluation of Efficacy Variables in Clinical Study of Irritable Bowel Syndrome with Diarrhea January 2018 Motoko IDA Evaluation of Efficacy Variables in Clinical Study of Irritable Bowel Syndrome with
Evaluation of Efficacy Variables in Clinical Study of Irritable Bowel Syndrome with Diarrhea January 2018 Motoko IDA Evaluation of Efficacy Variables in Clinical Study of Irritable Bowel Syndrome with
Does the Injection of Botulinum Toxin Improve Symptoms in Patients With Gastroparesis?
 Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2014 Does the Injection of Botulinum Toxin
Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2014 Does the Injection of Botulinum Toxin
Constipation. What is constipation? What is the criteria for having constipation? What are the different types of constipation?
 What is constipation? is defined as having a bowel movement less than 3 times per week. It is usually associated with hard stools or difficulty passing stools. You may have pain while passing stools or
What is constipation? is defined as having a bowel movement less than 3 times per week. It is usually associated with hard stools or difficulty passing stools. You may have pain while passing stools or
Jointly sponsored by Purdue University College of Pharmacy and the Gi Health Foundation. This activity is supported by Salix Pharmaceuticals.
 Accredited by Jointly sponsored by Purdue University College of Pharmacy and the Gi Health Foundation. This activity is supported by Salix Pharmaceuticals. Mark Pimentel, MD Cedars-Sinai Medical Center
Accredited by Jointly sponsored by Purdue University College of Pharmacy and the Gi Health Foundation. This activity is supported by Salix Pharmaceuticals. Mark Pimentel, MD Cedars-Sinai Medical Center
IRONWOOD AND FOREST ANNOUNCE POSITIVE LINACLOTIDE RESULTS FROM PHASE 3 TRIAL IN PATIENTS WITH IRRITABLE BOWEL SYNDROME WITH CONSTIPATION
 FOR IMMEDIATE RELEASE Ironwood Contact: Forest Contact: Susan Brady Frank J. Murdolo Corporate Communications Vice President, Investor Relations 617.621.8304 212.224.6714 sbrady@ironwoodpharma.com frank.murdolo@frx.com
FOR IMMEDIATE RELEASE Ironwood Contact: Forest Contact: Susan Brady Frank J. Murdolo Corporate Communications Vice President, Investor Relations 617.621.8304 212.224.6714 sbrady@ironwoodpharma.com frank.murdolo@frx.com
ACCIDENTAL BOWEL LEAKAGE: A PRACTICAL APPROACH TO EVALUATION. Tristi W. Muir, MD Chair, Department of OB/GYN Houston Methodist Hospital
 ACCIDENTAL BOWEL LEAKAGE: A PRACTICAL APPROACH TO EVALUATION Tristi W. Muir, MD Chair, Department of OB/GYN Houston Methodist Hospital Accidental Bowel Leakage What Gets the Woman into Your Office 67%
ACCIDENTAL BOWEL LEAKAGE: A PRACTICAL APPROACH TO EVALUATION Tristi W. Muir, MD Chair, Department of OB/GYN Houston Methodist Hospital Accidental Bowel Leakage What Gets the Woman into Your Office 67%
Mark Pimentel, MD Cedars-Sinai Medical Center Los Angeles, CA
 Anthony J. Lembo, M.D. Harvard Medical School Boston, MA Mark Pimentel, MD Cedars-Sinai Medical Center Los Angeles, CA Nicholas J. Talley, MD University of Newcastle Callaghan, NSW Australia Accredited
Anthony J. Lembo, M.D. Harvard Medical School Boston, MA Mark Pimentel, MD Cedars-Sinai Medical Center Los Angeles, CA Nicholas J. Talley, MD University of Newcastle Callaghan, NSW Australia Accredited
The Opportunity: c-ibs and pain relief with confidence YKP10811
 The Opportunity: c-ibs and pain relief with confidence YKP10811 1 TABLE OF CONTENTS Profile Summary Clinical Data Mode of Action Pharmacologic Profile Safety and Toxicity Profile ADME Overview vs. Competitors
The Opportunity: c-ibs and pain relief with confidence YKP10811 1 TABLE OF CONTENTS Profile Summary Clinical Data Mode of Action Pharmacologic Profile Safety and Toxicity Profile ADME Overview vs. Competitors
CENTENE PHARMACY AND THERAPEUTICS DRUG REVIEW 2Q17 April May
 BRAND NAME Trulance GENERIC NAME Plecanatide MANUFACTURER Synergy Pharmaceuticals, Inc. DATE OF APPROVAL January 19, 2017 PRODUCT LAUNCH DATE Anticipated in 1Q2017 REVIEW TYPE Review type 1 (RT1): New
BRAND NAME Trulance GENERIC NAME Plecanatide MANUFACTURER Synergy Pharmaceuticals, Inc. DATE OF APPROVAL January 19, 2017 PRODUCT LAUNCH DATE Anticipated in 1Q2017 REVIEW TYPE Review type 1 (RT1): New
Do Probiotics Provide Adequate Relief From Overall Symptoms, Including Abdominal Pain and Bloating, in Adults With Irritable Bowel Syndrome?
 Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2015 Do Probiotics Provide Adequate Relief
Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2015 Do Probiotics Provide Adequate Relief
Primary Management of Irritable Bowel Syndrome
 Primary Management of Irritable Bowel Syndrome Jasmine Zia, MD Acting Instructor, Division of Gastroenterology Current Concepts in Drug Therapy CME Course April 23, 2015 Irritable Bowel Syndrome (IBS)
Primary Management of Irritable Bowel Syndrome Jasmine Zia, MD Acting Instructor, Division of Gastroenterology Current Concepts in Drug Therapy CME Course April 23, 2015 Irritable Bowel Syndrome (IBS)
CENTENE PHARMACY AND THERAPEUTICS DRUG REVIEW 3Q17 July August
 BRAND NAME Symproic GENERIC NAME Naldemedine MANUFACTURER Shionogi Inc. DATE OF APPROVAL March 23, 2017 PRODUCT LAUNCH DATE Anticipated to launch mid-summer 2017 REVIEW TYPE Review type 1 (RT1): New Drug
BRAND NAME Symproic GENERIC NAME Naldemedine MANUFACTURER Shionogi Inc. DATE OF APPROVAL March 23, 2017 PRODUCT LAUNCH DATE Anticipated to launch mid-summer 2017 REVIEW TYPE Review type 1 (RT1): New Drug
Understanding & Alleviating Constipation. Living (Well!) with Gastroparesis Program Warm-Up Class
 Understanding & Alleviating Constipation Living (Well!) with Gastroparesis Program Warm-Up Class Please Remember The information presented is for educational purposes only and is in no way intended as
Understanding & Alleviating Constipation Living (Well!) with Gastroparesis Program Warm-Up Class Please Remember The information presented is for educational purposes only and is in no way intended as
Dr. Patsy Smyth, FNP-BC
 Dr. Patsy Smyth, FNP-BC Gastroparesis literally translated means stomach paralysis. Gastroparesis is a syndrome characterized by delayed gastric emptying in absence of mechanical obstruction of the stomach.
Dr. Patsy Smyth, FNP-BC Gastroparesis literally translated means stomach paralysis. Gastroparesis is a syndrome characterized by delayed gastric emptying in absence of mechanical obstruction of the stomach.
Constipation. (Medical Aspects)
 Constipation (Medical Aspects) By Dr. Ehab Abdel Khalik MD. Anatomy of the anorectum The rectum is 12-15 15 cm. long. It connects with the sigmoid colon by the rectosigmoid junction which is believed to
Constipation (Medical Aspects) By Dr. Ehab Abdel Khalik MD. Anatomy of the anorectum The rectum is 12-15 15 cm. long. It connects with the sigmoid colon by the rectosigmoid junction which is believed to
Relief, true to form
 For adults with Chronic Idiopathic Constipation (CIC) or Irritable Bowel Syndrome with Constipation (IBS-C) Relief, true to form When It Comes to Constipation Relief, Form Matters Trulance can help you
For adults with Chronic Idiopathic Constipation (CIC) or Irritable Bowel Syndrome with Constipation (IBS-C) Relief, true to form When It Comes to Constipation Relief, Form Matters Trulance can help you
pissn: eissn: Journal of Neurogastroenterology and Motility
 JNM J Neurogastroenterol Motil, Vol. 20 No. 3 July, 2014 pissn: 2093-0879 eissn: 2093-0887 http://dx.doi.org/10.5056/jnm14022 Original Article Effects of Chili Treatment on Gastrointestinal and Rectal
JNM J Neurogastroenterol Motil, Vol. 20 No. 3 July, 2014 pissn: 2093-0879 eissn: 2093-0887 http://dx.doi.org/10.5056/jnm14022 Original Article Effects of Chili Treatment on Gastrointestinal and Rectal
Irritable Bowel Syndrome
 Irritable Bowel Syndrome Functional gastrointestinal disorders Definition A variable combination of chronic or recurrent gastrointestinal symptoms (attributed to the pharynx, esophagus, stomach, biliary
Irritable Bowel Syndrome Functional gastrointestinal disorders Definition A variable combination of chronic or recurrent gastrointestinal symptoms (attributed to the pharynx, esophagus, stomach, biliary
Linaclotide Improves Abdominal Pain and Bowel Habits in a Phase IIb Study of Patients With Irritable Bowel Syndrome With Constipation
 GASTROENTEROLOGY 2010;139:1877 1886 CLINICAL Linaclotide Improves Abdominal Pain and Bowel Habits in a Phase IIb Study of Patients With Irritable Bowel Syndrome With Constipation JEFFREY M. JOHNSTON,*
GASTROENTEROLOGY 2010;139:1877 1886 CLINICAL Linaclotide Improves Abdominal Pain and Bowel Habits in a Phase IIb Study of Patients With Irritable Bowel Syndrome With Constipation JEFFREY M. JOHNSTON,*
MANAGEMENT OF CHRONIC CONSTIPATION BEYOND LAXATIVES
 Enrique Rey Professor of Medicine Head. Department of Digestive Diseases Hospital Clínico San Carlos Complutense University Madrid, Spain MANAGEMENT OF CHRONIC CONSTIPATION BEYOND LAXATIVES CONSTIPATION:
Enrique Rey Professor of Medicine Head. Department of Digestive Diseases Hospital Clínico San Carlos Complutense University Madrid, Spain MANAGEMENT OF CHRONIC CONSTIPATION BEYOND LAXATIVES CONSTIPATION:
GI Pharmacology -4 Irritable Bowel Syndrome and Antiemetics. Dr. Alia Shatanawi
 GI Pharmacology -4 Irritable Bowel Syndrome and Antiemetics Dr. Alia Shatanawi 11-04-2018 Drugs used in Irritable Bowel Syndrome Idiopathic, chronic, relapsing disorder characterized by abdominal discomfort
GI Pharmacology -4 Irritable Bowel Syndrome and Antiemetics Dr. Alia Shatanawi 11-04-2018 Drugs used in Irritable Bowel Syndrome Idiopathic, chronic, relapsing disorder characterized by abdominal discomfort
FOOT OFF THE BRAKES. Kerri Novak MD MSc FRCPC. Chronic Constipation: Taking the Foot off the Brakes Dr. Kerri Novak
 CHRONIC CONSTIPATION: TAKING THE FOOT OFF THE BRAKES Kerri Novak MD MSc FRCPC www.seacourses.com 1 OUTLINE Epidemiology i Quality of life Approach Therapies www.seacourses.com 2 DEFINING CHRONIC CONSTIPATION
CHRONIC CONSTIPATION: TAKING THE FOOT OFF THE BRAKES Kerri Novak MD MSc FRCPC www.seacourses.com 1 OUTLINE Epidemiology i Quality of life Approach Therapies www.seacourses.com 2 DEFINING CHRONIC CONSTIPATION
A Case of Fecal Incontinence: Medical and Interventional Treatment Options
 A Case of Fecal Incontinence: Medical and Interventional Treatment Options HPI JP is a 69 year-old F with a 12-month history of FI. Her symptoms began after a colonoscopy She has been experiencing passive
A Case of Fecal Incontinence: Medical and Interventional Treatment Options HPI JP is a 69 year-old F with a 12-month history of FI. Her symptoms began after a colonoscopy She has been experiencing passive
Fecal Incontinence. What is fecal incontinence?
 Scan for mobile link. Fecal Incontinence Fecal incontinence is the inability to control the passage of waste material from the body. It may be associated with constipation or diarrhea and typically occurs
Scan for mobile link. Fecal Incontinence Fecal incontinence is the inability to control the passage of waste material from the body. It may be associated with constipation or diarrhea and typically occurs
Current Pharmacological Treatment Options in Chronic Constipation and IBS with Constipation
 Current Pharmacological Treatment Options in Chronic Constipation and IBS with Constipation Anthony Lembo, M.D. Associate Professor of Medicine Harvard Medical School Director, GI Motility Center Beth
Current Pharmacological Treatment Options in Chronic Constipation and IBS with Constipation Anthony Lembo, M.D. Associate Professor of Medicine Harvard Medical School Director, GI Motility Center Beth
Constipation. H. David Vargas, MD. Overview
 Constipation H. David Vargas, MD Overview Constipation is a very common complaint affecting upwards of 15% of all Americans. Fortunately, constipation usually is simple to avoid and easy to treat when
Constipation H. David Vargas, MD Overview Constipation is a very common complaint affecting upwards of 15% of all Americans. Fortunately, constipation usually is simple to avoid and easy to treat when
10/10/16. Disclosures. Educational Objectives
 Nimish Vakil, MD, FACP, FACG, AGAF, FASGE Clinical Adjunct Professor University of Wisconsin Madison, Wisconsin Disclosures All faculty, course directors, planning committee, content reviewers and others
Nimish Vakil, MD, FACP, FACG, AGAF, FASGE Clinical Adjunct Professor University of Wisconsin Madison, Wisconsin Disclosures All faculty, course directors, planning committee, content reviewers and others
TREATMENT SOCIETY GUIDELINES FOR CONSTIPATION: WHAT IS NEW? FUNCTIONAL CONSTIPATION
 SOCIETY GUIDELINES FOR CONSTIPATION: WHAT IS NEW? Samuel Nurko MD MPH Center for Motility and Functional Gastrointestinal Disorders FUNCTIONAL CONSTIPATION One of the most common functional GI disorders
SOCIETY GUIDELINES FOR CONSTIPATION: WHAT IS NEW? Samuel Nurko MD MPH Center for Motility and Functional Gastrointestinal Disorders FUNCTIONAL CONSTIPATION One of the most common functional GI disorders
Linaclotide: A new drug for the treatment of chronic constipation and irritable bowel syndrome with constipation
 Review Article Linaclotide: A new drug for the treatment of chronic constipation and irritable bowel syndrome with constipation United European Gastroenterology Journal 1(1) 7 20! Author(s) 2013 Reprints
Review Article Linaclotide: A new drug for the treatment of chronic constipation and irritable bowel syndrome with constipation United European Gastroenterology Journal 1(1) 7 20! Author(s) 2013 Reprints
Paediatric constipation and functional non-retentive faecal soiling Voskuijl, W.P.
 UvA-DARE (Digital Academic Repository) Paediatric constipation and functional non-retentive faecal soiling Voskuijl, W.P. Link to publication Citation for published version (APA): Voskuijl, W. P. (2005).
UvA-DARE (Digital Academic Repository) Paediatric constipation and functional non-retentive faecal soiling Voskuijl, W.P. Link to publication Citation for published version (APA): Voskuijl, W. P. (2005).
Pharmacotherapy for IBS
 Pharmacotherapy for IBS Brooks D. Cash, M.D., FACG Chief, Gastroenterology Professor of Medicine University of South Alabama Director, GI Physiology, USA Medical Center Mobile, AL Disclosures I have served
Pharmacotherapy for IBS Brooks D. Cash, M.D., FACG Chief, Gastroenterology Professor of Medicine University of South Alabama Director, GI Physiology, USA Medical Center Mobile, AL Disclosures I have served
IBS Irritable Bowel syndrome Therapeutics II PHCL 430
 Salman Bin AbdulAziz University College Of Pharmacy IBS Irritable Bowel syndrome Therapeutics II PHCL 430 Email:- ahmedadel.pharmd@gmail.com Ahmed A AlAmer PharmD R.S is 32-year-old woman experiences intermittent
Salman Bin AbdulAziz University College Of Pharmacy IBS Irritable Bowel syndrome Therapeutics II PHCL 430 Email:- ahmedadel.pharmd@gmail.com Ahmed A AlAmer PharmD R.S is 32-year-old woman experiences intermittent
ARDELYX REPORTS POSITIVE T3MPO-2 PHASE 3 TRIAL RESULTS IN IBS-C
 ARDELYX REPORTS POSITIVE T3MPO-2 PHASE 3 TRIAL RESULTS IN IBS-C OCTOBER 11, 2017 NASDAQ: ARDX FORWARD-LOOKING STATEMENTS To the extent that statements contained in this presentation are not descriptions
ARDELYX REPORTS POSITIVE T3MPO-2 PHASE 3 TRIAL RESULTS IN IBS-C OCTOBER 11, 2017 NASDAQ: ARDX FORWARD-LOOKING STATEMENTS To the extent that statements contained in this presentation are not descriptions
6/25/ % 20% 50% 19% Functional Dyspepsia Peptic Ulcer GERD Cancer Other
 Peptic Ulcer Disease and Dyspepsia John M. Inadomi, MD Professor of Medicine UCSF Chief, Clinical Gastroenterology San Francisco General Hospital Case History 49 y/o woman complains of several months of
Peptic Ulcer Disease and Dyspepsia John M. Inadomi, MD Professor of Medicine UCSF Chief, Clinical Gastroenterology San Francisco General Hospital Case History 49 y/o woman complains of several months of
Irritable Bowel Syndrome and Chronic Constipation. Treatment of IBS. Susan Lucak, M.D. Columbia University Medical Center
 Ti tl e s l i d e - p a rt 1 Irritable Bowel Syndrome and Chronic Constipation Susan Lucak, M.D. Columbia University Medical Center Treatment of IBS Abdominal pain / discomfort Antispasmodics Antidepressants
Ti tl e s l i d e - p a rt 1 Irritable Bowel Syndrome and Chronic Constipation Susan Lucak, M.D. Columbia University Medical Center Treatment of IBS Abdominal pain / discomfort Antispasmodics Antidepressants
Medical Policy. MP Ingestible ph and Pressure Capsule
 Medical Policy BCBSA Ref. Policy: 2.01.81 Last Review: 11/15/2018 Effective Date: 11/15/2018 Section: Medicine Related Policies 2.01.20 Esophageal ph Monitoring 6.01.33 Wireless Capsule Endoscopy as a
Medical Policy BCBSA Ref. Policy: 2.01.81 Last Review: 11/15/2018 Effective Date: 11/15/2018 Section: Medicine Related Policies 2.01.20 Esophageal ph Monitoring 6.01.33 Wireless Capsule Endoscopy as a
Tenapanor for irritable bowel syndrome with constipation
 NIHR Innovation Observatory Evidence Briefing: February 2018 Tenapanor for irritable bowel syndrome with constipation NIHRIO (HSRIC) ID: 6704 NICE ID: 9736 LAY SUMMARY Irritable bowel syndrome with constipation
NIHR Innovation Observatory Evidence Briefing: February 2018 Tenapanor for irritable bowel syndrome with constipation NIHRIO (HSRIC) ID: 6704 NICE ID: 9736 LAY SUMMARY Irritable bowel syndrome with constipation
GASTROPARESIS. C. Prakash Gyawali, MD Professor of Medicine Washington University in St. Louis
 GASTROPARESIS C. Prakash Gyawali, MD Professor of Medicine Washington University in St. Louis Symptom Definitions Nausea: a subjective feeling of wanting to vomit Vomiting: forceful expulsion of gastroduodenal
GASTROPARESIS C. Prakash Gyawali, MD Professor of Medicine Washington University in St. Louis Symptom Definitions Nausea: a subjective feeling of wanting to vomit Vomiting: forceful expulsion of gastroduodenal
Lead team presentation Eluxadoline for treating irritable bowel syndrome with diarrhoea (STA)
 Public slides Lead team presentation Eluxadoline for treating irritable bowel syndrome with diarrhoea (STA) 1 st Appraisal Committee meeting Committee B, 25th January 2017 Lead team: Nigel Westwood, Anne
Public slides Lead team presentation Eluxadoline for treating irritable bowel syndrome with diarrhoea (STA) 1 st Appraisal Committee meeting Committee B, 25th January 2017 Lead team: Nigel Westwood, Anne
daily; available as 10- mg g PO
 Overview of the PRN: The Pain and Palliative Care PRN of ACCP is an organization of pharmacy practitioners, clinical scientists, pharmacy educators, and others. Its mission is to advance pain and palliative
Overview of the PRN: The Pain and Palliative Care PRN of ACCP is an organization of pharmacy practitioners, clinical scientists, pharmacy educators, and others. Its mission is to advance pain and palliative
A Nursing Assessment Tool for Adults With Fecal Incontinence
 Journal of Wound, Ostomy and Continence Nursing 2000, 279- A Nursing Assessment Tool for Adults With Fecal Incontinence Christine Norton, MA, RN, and Sonya Chelvanayagam, MSc, RN Abstract Fecal incontinence
Journal of Wound, Ostomy and Continence Nursing 2000, 279- A Nursing Assessment Tool for Adults With Fecal Incontinence Christine Norton, MA, RN, and Sonya Chelvanayagam, MSc, RN Abstract Fecal incontinence
APDW 2016 Poster No. a90312
 APDW 2016 Poster No. a90312 SYN-010, a Proprietary Modified-Release Formulation of Lovastatin Lactone, Lowered Breath Methane and Improved Stool Frequency in Patients with IBS-C Results of a multi-center,
APDW 2016 Poster No. a90312 SYN-010, a Proprietary Modified-Release Formulation of Lovastatin Lactone, Lowered Breath Methane and Improved Stool Frequency in Patients with IBS-C Results of a multi-center,
3D Dynamic Ultrasound In Obstructed Defecation
 3D Dynamic Ultrasound In Obstructed Defecation By Ramy Salahudin Abdelkader Assist. Lecturer of General Surgery Cairo University Introduction Pelvic floor is complex system, with passive and active components
3D Dynamic Ultrasound In Obstructed Defecation By Ramy Salahudin Abdelkader Assist. Lecturer of General Surgery Cairo University Introduction Pelvic floor is complex system, with passive and active components
At the outset, we want to clear up some terminology issues. IBS is COPYRIGHTED MATERIAL. What Is IBS?
 1 What Is IBS? At the outset, we want to clear up some terminology issues. IBS is the abbreviation that doctors use for irritable bowel syndrome, often when they are talking about people with IBS. We will
1 What Is IBS? At the outset, we want to clear up some terminology issues. IBS is the abbreviation that doctors use for irritable bowel syndrome, often when they are talking about people with IBS. We will
