Treatment of Neuropathic Pain with the Capsaicin 8% Patch: Is Pretreatment with Lidocaine Necessary?
|
|
|
- Sylvia Bates
- 5 years ago
- Views:
Transcription
1 ORIGINAL ARTICLE Treatment of Neuropathic Pain with the Capsaicin 8% Patch: Is Pretreatment with Lidocaine Necessary? Kai-Uwe Kern, MD*; Walburga Nowack, RN*; Chris Poole, PhD *Centre of Pain Medicine and Palliative Care, Wiesbaden, Germany; Pharmatelligence, Cardiff MediCentre, Cardiff, Wales, UK & Abstract: The capsaicin 8% patch can effectively treat neuropathic pain, but application can cause discomfort or a burning sensation. Until March 2013, it was recommended that patients be pretreated with a topical anesthetic, for example lidocaine, before capsaicin patch application. However, speculation existed over the need for pretreatment and its effectiveness in alleviating treatment-associated discomfort. This article compares tolerability to and efficacy of the capsaicin patch in pretreated and non-pretreated patients. All patients received a single capsaicin patch application. Pretreated patients received a lidocaine plaster before and intravenous lidocaine and metamizole infusions during capsaicin patch application. Pain levels, assessed using a Numeric Address correspondence and reprint requests to: Kai-Uwe Kern, MD, Institute of Pain Medicine, Pain Practice, Sonnenberger Str. 68, 65193, Wiesbaden, Germany. dr.kern@schmerzpraxis-wiesbaden.de Disclosures: Dr Kern is a consultant or presenter for Astellas, Berlin Chemie, Boehringer Ingelheim, Betapharm, Gr unenthal, Lilly, Medi Bayreuth, and Mundipharma and received LIIRA Grant support for clinical research from Pfizer. Pharmatelligence has received consultancy fees from Astellas, Grünenthal, and Pfizer. Dr Poole has also consulted for Astellas. Kai-Uwe Kern is no longer an employee at the Centre of Pain Medicine and Palliative Care and now works at the Institute of Pain Medicine, Pain Practice. Chris Poole is no longer an employee at Pharmatelligence and now works at Cardiff University. Submitted: April 16, 2013; Revision accepted: September 16, 2013 DOI /papr The Authors. Pain Practice published by Wiley Periodicals, Inc. on behalf of World Institute of Pain. This is an open access article under the terms of the Creative Commons Attribution-NonCommercial License, which permits use, distribution and reproduction in any medium, provided the original work is properly cited and is not used for commercial purposes /13/$15.00 Pain Practice, Volume 14, Issue 2, 2014 E42 E50 Rating Scale (NRS), were used to determine tolerability and efficacy. All patients (pretreated n = 32; non-pretreated n = 26) completed 100% of the intended capsaicin patch application duration. At the time of capsaicin patch removal, 69% of pretreated and 88% of non-pretreated patients reported an NRS score increase, which returned to baseline by 6 hours post-treatment. There was no significant difference in mean NRS score between patient groups at any time during or after capsaicin patch treatment. Response was similar between patient groups; capsaicin patch treatment provided rapid and significant pain reductions that were sustained over 12 weeks. The same proportion of pretreated and non-pretreated patients reported willingness to receive retreatment with the capsaicin patch. This analysis shows that the capsaicin 8% patch is generally tolerable, and the small discomfort associated with patch application is short-lived. Lidocaine pretreatment does not have a significant effect on tolerability, efficacy, or patient willingness to receive retreatment. & Key Words: capsaicin, nerve pain, neuralgia, nociceptors, peripheral nervous system, topical, tolerability, lidocaine pretreatment INTRODUCTION Neuropathic pain (NP), a chronic and disabling condition, affects up to 8% of the European population. 1 3 Treatments include tricyclic antidepressants, opioids, anticonvulsants, and topical capsaicin or lidocaine creams. 4,5 Many of these NP medications have limited efficacy, with typically fewer than 50% of patients
2 Capsaicin 8% Patch Application and Pretreatment E43 achieving satisfactory pain relief. 4 Additionally, cumbersome treatment regimens and unwanted systemic side effects are commonly associated with many medications, often resulting in patient intolerance to treatment and leading to poor compliance. 6 The capsaicin 8% patch (QUTENZA TM ) is indicated in Europe for the treatment of peripheral NP in nondiabetic adults either alone or in combination with other medicinal products for pain. Capsaicin causes excitation of the Transient Receptor Potential Vanilloid 1 (TRPV1) channels, expressed on nociceptors, leading to the generation of action potentials and a burning sensation in the skin. 7 However, administration of capsaicin at a high concentration leads to constant overstimulation of the nociceptors and results in a reversible reduction in epidermal nerve fiber density and defunctionalization of the nociceptors. The overall outcome of this is a reduction in NP Treatment with the capsaicin 8% patch is not associated with any systemic side effects, and a single application can provide significant relief from NP for up to 3 months Due to the mechanism of action of capsaicin, patients treated with the capsaicin 8% patch may experience a burning sensation during application. 7,11 Until recently, it was therefore recommended that, to alleviate any discomfort, patients receive pretreatment with a topical anesthetic, such as lidocaine or EMLA â, before patch application. However, there was speculation regarding the utility of topical anesthetics to block capsaicinrelated discomfort and, in March 2013, updates were made to the recommendations for pretreatment to state that the treatment area may be pretreated with a topical anesthetic or the patient might be administered an oral analgesic prior to patch application. 16 Amide-type anesthetics (such as lidocaine) prevent nerve impulse transmission through inhibition of sodium ion channels and so reduce the transmission of pain. 17 However, these sodium channels act independently of the TRPV1 receptors at peripheral nerve terminals. Consequently, TRPV1 can induce axonal depolarization and defunctionalization even in the presence of sodium channel blockade. 18,19 At the Centre of Pain Medicine and Palliative Care in Wiesbaden, Germany, the capsaicin 8% patch is routinely used to treat a variety of patients with NP. Initially, the capsaicin 8% patch was largely used to treat those patients who had been experiencing NP for a long time and had not responded well, or at all, to other medications. At this time, the original recommendations with regard to pretreatment were followed and all patients received pretreatment with lidocaine before application of the capsaicin 8% patch. As experience with the patch was gained by healthcare professionals at the clinic and positive effects of the treatment were observed, the capsaicin 8% patch was offered to new patients who were at an earlier point in their disease progression. The experience gained also affected how the healthcare professionals chose to manage treatmentassociated discomfort and raised questions around the necessity of pretreatment with lidocaine, ultimately resulting in the discontinuation of its routine use in this clinic. Here, the authors report an analysis of the tolerability and efficacy of treatment with the capsaicin 8% patch in patients at the clinic in Wiesbaden, comparing the experience of patients who did and did not receive lidocaine pretreatment prior to capsaicin 8% patch therapy. METHODS Patients All patients diagnosed with an NP condition and receiving treatment with the capsaicin 8% patch between May 2010 and May 2011 at the Centre of Pain Medicine and Palliative Care, Wiesbaden, Germany were included in the analysis. Ethical approval for this retrospective analysis was granted on December 13, 2011 from the Ethik-Kommission bei der Landes arztekammer Hessen. As this was a retrospective analysis, informed consent was not required and all treatments and assessments were consistent with the usual standard of care. Procedure The application procedure for the capsaicin patch was carried out as described previously. 20 Following pretreatment, as appropriate, the capsaicin 8% patch was applied for 30 minutes to the feet and for 60 minutes to all other areas of the body. Pretreatment. Patients receiving pretreatment were asked to apply a lidocaine patch (Versatis â ;Gr unenthal, Germany) 2 hours prior to capsaicin 8% patch application. Pretreated patients were provided with an intravenous infusion of lidocaine, 80 mg (Braun, Germany) for 1 hour during patch application, and
3 E44 KERN ET AL. the majority were also provided with an infusion of metamizole, 1 g (Lichtenstein, Germany) during capsaicin patch application. For those patients treated on the feet, the lidocaine infusion continued for 30 minutes after removal of the capsaicin patch. A few patients received lidocaine and metamizole infusions without lidocaine patch application. Patients not receiving pretreatment did not receive cleansing of the skin prior to capsaicin patch application as this was not necessary. Management of Treatment-related Discomfort Treatment-related discomfort was monitored during and after the application procedure. Patients who experienced discomfort during patch application could receive metamizole 1 g, and if further analgesia was required, piritramide 3.75 to 30 mg (Janssen-Cilag, Germany). After the capsaicin patch had been removed, patients received localized cooling to alleviate any continued discomfort. Pain Level Assessment Pain or discomfort pre- and postcapsaicin patch application was assessed using a Numeric Rating Scale (NRS), where patients were asked to score their pain on an integer scale of 0 to 10, with 0 meaning no pain and 10 representing worst imaginable pain. Using pain diaries, patients were asked to assess their pain for the week prior to treatment, immediately prior to the initiation of treatment, and at 0.5, 1, 6, 12, and 24 hours and 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 30, and 90 days after capsaicin 8% patch application. Tolerability Tolerability was assessed by the number of patients who completed the intended duration of capsaicin 8% patch application, by the change in pain score at the time of patch removal and up to 24 hours postpatch removal, and by the requirement for additional analgesics. The NRS score immediately prior to the initiation of treatment was used as the baseline in analysis of change in pain score. Efficacy Efficacy was assessed by the absolute change in NRS score from baseline, the percentage change in NRS score, and the proportion of responders, classified as patients who exhibit 30% reduction from baseline in pain score up to 90 days post-treatment. When assessing efficacy, the mean NRS score for the week prior to treatment was used as the baseline pain score. Statistical Analysis Differences in baseline characteristics were tested appropriately to their respective distributions. Given the ordinal properties of the NRS scale, between-group (pretreated and non-pretreated patients) differences in change in absolute pain score from baseline were first tested at each timepoint (24 hours and 30 and 90 days post-treatment) with the nonparametric Mann Whitney U-test, then collectively using repeated-measures ordinal logistic general estimating equation (GEE) modeling with multinomial distribution with a cumulative logit link function. As percentage change in pain exhibited an approximately normal distribution and pseudo-scale properties, initial between-group comparisons at the fixed timepoints were made using the independent samples Student s t-test, followed by repeated-measures linear GEE modeling, with normal distribution and an identity link function. Measurement time and pretreatment subgroup were modeled as fixed factors in all analyses. SPSS v20 (IBM Corp., Armonk, NY, USA) statistical software was used for all inferential testing. The threshold parameter significance was set at P 0.05 for all tests. Retreatment At 30 and 90 days post-treatment, patients were asked to record in their pain diary whether they would be happy to receive retreatment with the capsaicin 8% patch. From 10 weeks after initial treatment, patients could request retreatment with the capsaicin 8% patch if their pain had returned to around 80% of the baseline level. RESULTS Patients Between May 2010 and May 2011, 58 patients received a total of 82 capsaicin 8% patch treatments at the Centre of Pain Medicine and Palliative Care, Wiesbaden, Germany. Of these patients, 41 received 1 treatment, 12 patients were treated twice, 4 patients received three treatments, and 1 patient received five
4 Capsaicin 8% Patch Application and Pretreatment E45 treatments. Only data from the first capsaicin 8% patch treatment received by the 58 patients are included in the analyses reported here. Of these first treatments, 32 patients received pretreatment and 26 received no pretreatment. Among the pretreated patients, 21 received a lidocaine patch plus infusions of lidocaine and metamizole, 8 received only infusions of lidocaine and metamizole, while 3 patients received a lidocaine patch and a lidocaine infusion. Pretreatment was routinely carried out as part of the capsaicin 8% patch treatment procedure for the first 5 months of using the therapy at the center (between May and October 2010). In November 2010, the decision was made to utilize the capsaicin 8% patch without pretreatment where possible. This was a clinical decision based on the experience gained of the treatment procedure and after hearing the opinion of other experienced users of the capsaicin 8% patch who had routinely provided pretreatment. It was felt that it would be beneficial to remove the lengthy pretreatment step if possible and that this would not be detrimental to the patient or efficacy of the treatment. After the pretreatment step was removed, it soon became apparent that pretreatment is not necessary in most cases. A comparison at baseline showed that pretreated and non-pretreated patients were similar in most regards, Table 1. Baseline Characteristics of All Patients Non-pretreated (n = 26) Pretreated (n = 32) P value Male, n (%) 10 (38) 16 (50) Age, years (SD) 64.9 (15.3) 61.4 (14.8) Pain duration, years (SD) 2.2 (2.2) 5.5 (3.6) <0.001 Application time, n (%) 30 minutes 1 (4) 7 (22) 60 minutes 25 (96) 25 (78) NRS score prior to 4.8 (2.0) 5.3 (2.6) treatment (SD) NP diagnosis, n (%) Postherpetic neuralgia 15 (58) 10 (31) Mononeuropathy 4 (15) 6 (19) Polyneuropathy 1 (4) 3 (9) Postsurgical/post-trauma 4 (15) 5 (16) FBSS/postdicsectomy 0 4 (13) Other 2 (8) 4 (13) Concomitant medications for pain, n (%) Any 23 (88) 31 (97) Opioids 13 (50) 22 (69) Anticonvulsants 9 (35) 18 (56) Antidepressants 6 (23) 2 (6) Other 14 (54) 11 (34) Mean number of concomitant medications for pain taken by each patient (SD) 1.8 (1.4) 1.8 (0.9) FBSS, failed back surgery syndrome; SD, standard deviation. except for the mean duration of NP, which was significantly greater in the population of pretreated patients compared with non-pretreated patients (Table 1). Tolerability All patients completed 100% of the intended capsaicin 8% patch application time. At the time of capsaicin 8% removal, 22 (69%) pretreated patients and 23 (88%) non-pretreated patients reported an increase in NRS compared with NRS immediately prior to initiation of treatment, while 8 (25%) pretreated patients and 1 (4%) non-pretreated patient reported a decrease in NRS compared with NRS immediately prior to initiation of treatment (Figure 1). Of those patients reporting a decrease in NRS, 4 (44%) had received a 30-minute capsaicin patch application. A moderate increase in NRS (increase of > 5) was reported in 3 (9%) patients who received pretreatment and 4 (15%) patients who did not (Figure 1). During treatment, there was an increase in mean NRS score in both pretreated and non-pretreated patients, but this rapidly declined and by 6 hours after capsaicin 8% patch application, NRS scores had returned to baseline levels in both groups (Figure 2). There was no significant difference in mean NRS score between non-pretreated and pretreated patients at any time during or immediately after the treatment period, up to 24 hours postapplication. As analgesic responses are often not distributed equally around the mean, the data were analyzed using both parametric and nonparametric statistical methods. The same result was observed when using either test (Figure 2). This was corroborated by ordinal logistic GEE that revealed no significant parameter effect for pretreatment allocation (P = 0.508). In total, 34 patients requested additional analgesia during application of the capsaicin 8% patch due to treatment-related discomfort. Twenty-two patients (65%) received only nonopioid analgesia (either metamizole or ketoprofen), 3 (9%) received piritramide only, while 9 (26%) received both nonopioid and opioid analgesia. Despite the slightly higher increase in NRS among patients who did not receive pretreatment compared with those who did, there was no significant difference in nonopioid treatment. However, significantly, more pretreated patients than non-pretreated patients received piritramide during capsaicin 8% patch treatment (31% vs. 8%; P = 0.028; Table 2).
5 E46 KERN ET AL. Pretreated; 60-minute capsaicin patch application Pretreated; 30-minute capsaicin patch application 10 Non-pretreated; 60-minute capsaicin patch application Non-pretreated; 30-minute capsaicin patch application Change in Pain Score from Baseline at Patch Removal (NRS) Pretreated Non-pretreated Figure 1. Change in pain score from baseline at removal of capsaicin 8% patch. Data for individual patients receiving lidocaine pretreatment (n = 32) and receiving no pretreatment (n = 26) are shown, ordered by size of change in Numeric Rating Scale (NRS) score from baseline. Mean Pain Score (NRS) P = (0.078) P = (0.157) P = (0.342) P = P = (0.469) (0.510) Non-pretreated Pretreated P = (0.509) Time Since Patch Application (hours) Figure 2. Mean pain scores (SD) for pretreated and nonpretreated patients during the first 24 hours after capsaicin 8% patch treatment. P values for pretreated (n = 32) vs. nonpretreated (n = 26) patients were calculated using a t-test for equality of means, not assuming equal variance, and using the Mann Whitney U-test that compares ranks and is therefore not affected by the distribution of the dependent variable. P values from the Mann Whitney U-test are presented in brackets. Tolerability was also assessed in patients grouped according to their baseline NRS score, using the mean NRS for the week prior to treatment. The general trend appeared to be for patients with a higher baseline NRS score (7 to 10) to exhibit a decrease in NRS score from baseline at 0.5, 1, 6, and 12 hours after application of the capsaicin 8% patch (Figure S1). In patients with a lower baseline pain score (NRS 3 to 6), it was more common to see an increase in NRS from baseline at 30 and 60 minutes postpatch application and then a reduction at 6 and 12 hours postpatch application, returning to near or below baseline levels (Figure S1). There was no apparent difference between patients who received pretreatment and those who did not receive pretreatment. Adverse Events. In general, adverse events were limited to application site events, including erythema and treatment-related discomfort, which were mild and self-limited. One patient developed an allergic reaction distant from the application site, which required a physician consultation, and led to sleep disturbance the same night, 3 other patients required a physician consultation during treatment due to treatmentrelated discomfort, and 3 further patients reported sleep disturbance during the first night after treatment. Efficacy Following capsaicin 8% patch treatment, a reduction in NRS score of 30% was seen in 31% of pretreated Table 2. Requirement for Additional Pain Medication during Capsaicin 8% Patch Treatment Non-pretreated, n (%) Pretreated, n (%) P value* Odds ratio 95% CI Nonopioid pain medication 15 (58) 16 (50) to 2.08 Opioid pain medication (piritramide) 2 (8) 10 (31) to *Pretreated vs. non-pretreated; calculated using chi-squared test. CI, confidence interval.
6 Capsaicin 8% Patch Application and Pretreatment E47 A Change in Pain from Baseline (NRS) B Non-pretreated Pretreated Non-pretreated Pretreated (0.795) 10.0 (0.888) (0.440) (0.449) (0.633) (0.463) (0.718) (0.481) (0.037) (0.057) Time Since Treatment (days) Time Since Treatment (days) Change in Pain from Baseline (NRS), % Figure 3. Change in pain score from baseline in pretreated and non-pretreated patients. (A) Change in absolute Numeric Rating Scale (NRS) score and (B) percentage change in NRS score from baseline (mean pain score during the week before treatment) is shown. P values for pretreated (n = 32) vs. non-pretreated (n = 24) patients were calculated using a t-test for equality of means, not assuming equal variance, and using the Mann Whitney U-test that compares ranks and is therefore not affected by the distribution of the dependent variable. P values from the Mann Whitney U-test are presented in brackets. patients and 26% of non-pretreated patients after 30 days, and in 38% of pretreated patients and 30% of non-pretreated patients after 90 days. The difference between pretreated and non-pretreated patients was not significant at either timepoint (P = and P = 0.587, respectively). The pain reductions observed were rapid and sustained over 90 days (Figure 3). At initial timepoints, pretreated patients exhibited a greater reduction in pain score from baseline than non-pretreated patients. From Day 7 to Day 90, there was no difference between the Patients Willing to Receive Retreatment (%) Pretreated (n = 30) 57 Nonpretreated (n = 23) Responder (n = 19) Nonresponder (n = 34) Figure 4. Proportion of patients stating that they would be happy to receive capsaicin 8% patch retreatment when necessary. Patients were asked to record if they would be willing to receive capsaicin 8% patch retreatment 90 days after initial treatment. The proportion of pretreated and non-pretreated patients and the proportion of responders, classified as those patients with 30% decrease in NRS from baseline, and nonresponders willing to receive retreatment are shown two groups (Figure 3). Use of parametric tests demonstrated a significant difference between percentage reduction from baseline in NRS score at Day 3 posttreatment (Figure 3B). However, the nonparametric analysis found no significant difference at this timepoint. At all other timepoints, there was no significant difference regardless of the test used. These findings were corroborated by ordinal logistic GEE of absolute change in pain score and linear GEE of percentage change in pain score; in both instances, pretreatment allocation was not a significant predictor (P = and P = 0.244, respectively). Retreatment Thirty and 90 days after treatment, there was no difference between the number of pretreated and nonpretreated patients who would be happy to receive capsaicin 8% patch retreatment if and when necessary (Figure 4). A greater proportion of responders (patients with a 30% decrease in NRS from baseline) than nonresponders at 90 days stated that they would be willing to receive retreatment (84% vs. 41%; P = 0.002). DISCUSSION Observations in this real-life clinical setting suggest that the tolerability of capsaicin 8% patch application is similar regardless of whether patients have received
7 E48 KERN ET AL. lidocaine pretreatment or not. In particular, comparing patients receiving or not receiving pretreatment, a similar number requested pain relief with metamizole during capsaicin 8% patch application. Moreover, whether or not a patient had received pretreatment had no effect on the willingness of patients to receive retreatment with the capsaicin 8% patch, with the majority of patients stating they would be happy to receive retreatment when necessary. Interestingly, the data suggest that patients with more pain at baseline may tolerate application of the capsaicin 8% patch better than patients with less pain at baseline. It may be that patients who generally experience a higher level of NP do not find that the discomfort caused by the capsaicin 8% patch is strong enough to cause them more pain than they already experience, while patients with a lower baseline level of pain may be more sensitive to other stimuli that can cause discomfort. Whether or not patients received pretreatment also did not have any effect on the efficacy of capsaicin 8% patch treatment. Although pretreated patients displayed a greater reduction in pain than non-pretreated patients initially, there was no significant difference in pain reduction between the patient groups at timepoints from Day 7 through to Day 90. Thirty days after treatment, 31% of pretreated patients and 26% of non-pretreated patients had a reduction in NRS score of 30% compared with baseline. Due to the way in which we collected our patients data, it is not possible to carry out a direct comparison between efficacy of the capsaicin patch in our patients and data seen in the clinical trials, in which patients with postherpetic neuralgia received treatment with the capsaicin 8% patch. Nonetheless, our data may be considered to be comparable to those in the clinical trials, in which 42% to 46% of patients achieved a reduction of 30% in pain score during Weeks 2 to 8 compared with baseline. 12,14 While we chose to report response to capsaicin 8% patch treatment by analyzing the mean change from baseline in pain score to allow this informal comparison with clinical trial data, we note that analgesic response is not normally distributed around the mean, raising possible concerns regarding the use of parametric statistical methods. We therefore carried out additional analysis of the mean changes in pain score using nonparametric tests that compared differences in ranks of pain scores. This analysis demonstrated that the data are similar regardless of the technique used and supports the validity of the parametric analysis. This is in line with experience that nonparametric methods are normally consistent with parametric methods, unless the distribution of data is very substantially skewed. 21 It is interesting to note that of 8 pretreated patients showing a decrease from baseline in NRS at the time of patch removal, 4 had received a 30-minute treatment. These data are perhaps not that unexpected, as it is likely that the effects of the pretreatment medication on the patient s NP are still being felt after 30 minutes, but the effects will subside by 60 minutes. Moreover, we and others have noted that the onset of treatment-related discomfort usually occurs around 30 minutes after application of the capsaicin 8% patch. 22 The major disadvantage of this study is its retrospective nature and the potential for bias to have been introduced into the study. It could be argued that the analysis presented in this article was carried out to confirm an assumption already made when clinical practice was changed to stop pretreatment; that assumption being that pretreatment was not usually necessary. However, while we did feel at the time of changing clinical practice that pretreatment may be an unnecessary step, it was only after removal of the pretreatment step and observing patients responses that we decided that pretreatment was not necessary. Throughout the entire time period covered in the current analysis, the assessment measures remained the same for all patients, who were asked to score their pain on a rating of 0 to 10 at the same predefined timepoints post-treatment. All patients used the same 11-point pain rating scale and recorded their score in pain diaries. The retrospective analysis we present here is based on pain scores retrieved using these assessments, and therefore, the outcomes are largely independent of any predrawn conclusion. The results of the analysis presented here are likely to have been affected in part by the timeframe in which the data were collected. Our analysis covers the first year after which we started treating patients with the capsaicin 8% patch and discontinuation of the routine use of pretreatment occurred as we became more experienced with use of the patch. Thus, in general, patients receiving pretreatment were treated by staff less experienced in capsaicin patch use than those not receiving pretreatment. This factor is likely to account for the significantly greater number of pretreated than non-pretreated patients requesting treatment with opioid pain medication during capsaicin 8% patch application. Other limitations of this study include the small number of patients included within the analysis and the fact that the results are from a single center.
8 Capsaicin 8% Patch Application and Pretreatment E49 Nonetheless, these limitations do not affect the major conclusion of our analysis, namely that treatment of patients with the capsaicin 8% patch without analgesic pretreatment is well tolerated by the majority of patients. Indeed, we believe that improvements in the experience of patient management and treatment procedures may actually have more effect on the tolerability of capsaicin 8% patch treatment than lidocaine pretreatment. The importance of patient management to ensure good tolerability of capsaicin 8% patch treatment has been described in detail previously by a group of experienced users. 20 Our positive experience of treating patients with the capsaicin 8% patch without using lidocaine pretreatment means that we continue to do this routinely, with over 150 treatments carried out to date. The manner in which the data were collected is also likely to account for the significant difference in mean duration of NP between pretreated and non-pretreated patients. In general, when the capsaicin patch became available for the treatment of peripheral NP those patients with long-standing NP that was not effectively controlled by other medications were the first to receive this new treatment. There are practical benefits to not pretreating patients before application of the capsaicin 8% patch. These include a reduction in total treatment time, which would be advantageous to both patients and healthcare professionals, and a potential cost benefit, resulting from removal of the cost of the pretreatment medication(s). Further studies are warranted to investigate these benefits further. An additional point to note is that in March 2013, updates were made to the recommendations for pretreatment before capsaicin 8% patch application. The label now states that the treatment area may be pretreated with a topical anesthetic or the patient might be administered an oral analgesic prior to patch application. The findings from this analysis align with these updates in suggesting that pretreatment with a topical anesthetic may not be necessary when administering the capsaicin 8% patch. In conclusion, the data presented here from the Centre of Pain Medicine and Palliative Care in Wiesbaden indicate that the small increase in discomfort that is seen in most patients during capsaicin 8% patch application is short-lived and tolerable, regardless of whether the patient has received pretreatment with lidocaine or not, and it neither deters patients from accepting retreatment nor does it affect efficacy. ACKNOWLEDGMENTS Editorial assistance was provided by Adelphi Communications Ltd, supported by Astellas Pharma Europe Ltd. REFERENCES 1. Bouhassira D, Lanteri-Minet M, Attal N, Laurent B, Touboul C. Prevalence of chronic pain with neuropathic characteristics in the general population. Pain. 2008;136: Mailis Gagnon A, Furlan A, Lakha SF, Yegneswaran B. Systematic review of the prevalence of neuropathic pain. Eur J Pain. 2007;11(Suppl. 1):S202 S203 (Abstract 457). 3. Torrance N, Smith BH, Bennett MI, Lee AJ. The epidemiology of chronic pain of predominantly neuropathic origin. Results from a general population survey. J Pain. 2006;7: O Connor AB, Dworkin RH. Treatment of neuropathic pain: an overview of recent guidelines. Am J Med. 2009;122 (10 Suppl.):S22 S Stillman M. Clinical approach to patients with neuropathic pain. Cleve Clin J Med. 2006;73: Dworkin RH, O Connor AB, Audette J, et al. Recommendations for the pharmacological management of neuropathic pain: an overview and literature update. Mayo Clin Proc. 2010;85(3 Suppl.):S3 S Wallace M, Pappagallo M. Qutenza â : a capsaicin 8% patch for the management of postherpetic neuralgia. Expert Rev Neurother. 2011;11: Anand P, Bley K. Topical capsaicin for pain management: therapeutic potential and mechanisms of action of the new high-concentration capsaicin 8% patch. Br J Anaesth. 2011;107: Kennedy WR, Vanhove GF, Lu SP, et al. A randomized, controlled, open-label study of the long-term effects of NGX-4010, a high-concentration capsaicin patch, on epidermal nerve fiber density and sensory function in healthy volunteers. J Pain. 2010;11: Malmberg AB, Mizisin AP, Calcutt NA, et al. Reduced heat sensitivity and epidermal nerve fiber immunostaining following single applications of a high-concentration capsaicin patch. Pain. 2004;111: McCormack PL. Capsaicin dermal patch: in non-diabetic peripheral neuropathic pain. Drugs. 2010;70: Backonja M, Wallace MS, Blonsky ER, et al. NGX- 4010, a high-concentration capsaicin patch, for the treatment of postherpetic neuralgia: a randomised, double-blind study. Lancet Neurol. 2008;7: Backonja MM, Malan TP, Vanhove GF, Tobias JK. NGX-4010, a high-concentration capsaicin patch, for the treatment of postherpetic neuralgia: a randomized, doubleblind, controlled study with an open-label extension. Pain Med. 2010;11: Irving GA, Backonja MM, Dunteman E, et al. A multicenter, randomized, double-blind, controlled study of
9 E50 KERN ET AL. NGX-4010, a high-concentration capsaicin patch, for the treatment of postherpetic neuralgia. Pain Med. 2011;12: Simpson DM, Brown S, Tobias J. Controlled trial of high-concentration capsaicin patch for treatment of painful HIV neuropathy. Neurology. 2008;70: Astellas Pharma Europe Ltd. Summary of Product Characteristics: Qutenza 179 mg Cutaneous Patch. London, UK: European Medicines Agency; 2013 (cited 31 July 2013); Available from: WC pdf. 17. Medicines and Healthcare product Regulatory Agency (MHRA). LMX 4 lidocaine 4% w/w cream: PL 20685/0034. London, UK: MHRA; 2007 [cited 31 July 2013]; Available from: siteresources/con pdf. 18. Craft RM, Porreca F. Tetracaine attenuates irritancy without attenuating desensitization produced by intravesical resiniferatoxin in the rat. Pain. 1994;57: Lam VY, Wallace M, Schulteis G. Effects of lidocaine patch on intradermal capsaicin-induced pain: a double-blind, controlled trial. J Pain. 2011;12: England J, Wagner T, Kern KU, Roth-Daniek A, Sell A. The capsaicin 8% patch for peripheral neuropathic pain. Br J Nurs. 2011;20: Tanizaki H. Power comparison of non-parametric tests: small-sample properties from Monte Carlo experiments. J Appl Stat. 1997;24: Wagner T, Roth-Daniek A, Sell A, England J, Kern K-U. Capsaicin 8% patch for peripheral neuropathic pain: review of treatment best practice from real-world clinical experience. Pain Manag. 2012;2: SUPPORTING INFORMATION Additional Supporting Information may be found in the online version of this article: Figure S1. Mean change in pain score from baseline to 24 hours in pretreated and non-pretreated patients. Changes in mean absolute Numeric Rating Scale (NRS) score from baseline (NRS score immediately prior to capsaicin 8% patch treatment) to times during the first 24 hours following patch application were determined for patients grouped according to mean baseline NRS in the week before treatment with the capsaicin 8% patch.
Original Research Article The Capsaicin 8% Patch for Neuropathic Pain in Clinical Practice: A Retrospective Analysis
 bs_bs_banner Pain Medicine 2013; 14: 1202 1211 Wiley Periodicals, Inc. NEUROPATHIC PAIN SECTION Original Research Article The Capsaicin 8% Patch for Neuropathic Pain in Clinical Practice: A Retrospective
bs_bs_banner Pain Medicine 2013; 14: 1202 1211 Wiley Periodicals, Inc. NEUROPATHIC PAIN SECTION Original Research Article The Capsaicin 8% Patch for Neuropathic Pain in Clinical Practice: A Retrospective
Capsaicin cutaneous patch
 New Medicines Profile August 2010 Issue. 10/03 cutaneous patch Concise evaluated information to support the managed entry of new medicines in the NHS Summary cutaneous patch (Qutenza ) is licensed for
New Medicines Profile August 2010 Issue. 10/03 cutaneous patch Concise evaluated information to support the managed entry of new medicines in the NHS Summary cutaneous patch (Qutenza ) is licensed for
Patient s Guide. A single, 1-hour, localized treatment may provide 3 months of relief from post-shingles nerve pain.
 Patient s Guide A single, 1-hour, localized treatment may provide 3 months of relief from post-shingles nerve pain www..com is indicated for the management of neuropathic pain associated with postherpetic
Patient s Guide A single, 1-hour, localized treatment may provide 3 months of relief from post-shingles nerve pain www..com is indicated for the management of neuropathic pain associated with postherpetic
Brown et al. AIDS Research and Therapy 2013, 10:5
 Brown et al. AIDS Research and Therapy 213, 1:5 RESEARCH Open Access NGX-41, a capsaicin 8% patch, for the treatment of painful HIV-associated distal sensory polyneuropathy: integrated analysis of two
Brown et al. AIDS Research and Therapy 213, 1:5 RESEARCH Open Access NGX-41, a capsaicin 8% patch, for the treatment of painful HIV-associated distal sensory polyneuropathy: integrated analysis of two
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE SCOPE
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Neuropathic pain pharmacological management: the pharmacological management of neuropathic pain in adults in non-specialist
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Neuropathic pain pharmacological management: the pharmacological management of neuropathic pain in adults in non-specialist
Intravenous Lidocaine for Neuropathic Pain: A Retrospective Analysis of Tolerability and Efficacy
 Pain Medicine 2014; 2015; : 16: 531 536 Wiley Periodicals, Inc. NEUROPATHIC Intravenous Lidocaine PAIN SECTION for Neuropathic Pain: A Retrospective Analysis of Tolerability Brief and Efficacy Research
Pain Medicine 2014; 2015; : 16: 531 536 Wiley Periodicals, Inc. NEUROPATHIC Intravenous Lidocaine PAIN SECTION for Neuropathic Pain: A Retrospective Analysis of Tolerability Brief and Efficacy Research
Neuropathic Pain Treatment Guidelines
 Neuropathic Pain Treatment Guidelines Background Pain is an unpleasant sensory and emotional experience that can have a significant impact on a person s quality of life, general health, psychological health,
Neuropathic Pain Treatment Guidelines Background Pain is an unpleasant sensory and emotional experience that can have a significant impact on a person s quality of life, general health, psychological health,
Capsaicin 8% patch for treprostinil subcutaneous infusion site pain in pulmonary hypertension patients
 British Journal of Anaesthesia 112 (2): 337 47 (214) Advance Access publication 22 September 213. doi:1.193/bja/aet38 Capsaicin 8% patch for treprostinil subcutaneous infusion site pain in pulmonary hypertension
British Journal of Anaesthesia 112 (2): 337 47 (214) Advance Access publication 22 September 213. doi:1.193/bja/aet38 Capsaicin 8% patch for treprostinil subcutaneous infusion site pain in pulmonary hypertension
7 th November % of patients had lidocaine plasters prescribed for the licensed indication of post herpatic neuralgia
 Directorate of Integrated Care Health and Social Care Board 12-22 Linenhall Street Belfast BT2 8BS Tel : 028 90553782 Fax : 028 90553622 Web Site: www.hscboard.hscni.net 7 th November 2013 Dear colleague
Directorate of Integrated Care Health and Social Care Board 12-22 Linenhall Street Belfast BT2 8BS Tel : 028 90553782 Fax : 028 90553622 Web Site: www.hscboard.hscni.net 7 th November 2013 Dear colleague
Capsaicin 8% patch versus oral pregabalin in patients with peripheral neuropathic pain
 ORIGINAL ARTICLE Capsaicin 8% patch versus oral pregabalin in patients with peripheral neuropathic pain M. Haanp a a 1, G. Cruccu 2, T.J. Nurmikko 3, W.T. McBride 4, A. Docu Axelarad 5, A. Bosilkov 6,
ORIGINAL ARTICLE Capsaicin 8% patch versus oral pregabalin in patients with peripheral neuropathic pain M. Haanp a a 1, G. Cruccu 2, T.J. Nurmikko 3, W.T. McBride 4, A. Docu Axelarad 5, A. Bosilkov 6,
Scottish Medicines Consortium
 Scottish Medicines Consortium duloxetine 30mg and 60mg capsules (Cymbalta ) No. (285/06) Eli Lilly and Company Limited/Boehringer Ingelheim 4 August 2006 The Scottish Medicines Consortium has completed
Scottish Medicines Consortium duloxetine 30mg and 60mg capsules (Cymbalta ) No. (285/06) Eli Lilly and Company Limited/Boehringer Ingelheim 4 August 2006 The Scottish Medicines Consortium has completed
Assay Sensitivity.
 Assay Sensitivity Michael C. Rowbotham, MD Professor of Neurology UCSF-Mount Zion Pain Management Center Senior Scientist and IRB Chair, CPMC Research Institute Michael.Rowbotham@ucsf.edu Outline What
Assay Sensitivity Michael C. Rowbotham, MD Professor of Neurology UCSF-Mount Zion Pain Management Center Senior Scientist and IRB Chair, CPMC Research Institute Michael.Rowbotham@ucsf.edu Outline What
Capsaicin 8% patch repeat treatment in non-diabetic peripheral neuropathic pain: a 52- week, open-label, single-arm, safety study
 The Clinical Journal of Pain Publish Ahead of Print DOI:10.1097/AJP.0000000000000473 Capsaicin 8% patch repeat treatment in non-diabetic peripheral neuropathic pain: a 52- week, open-label, single-arm,
The Clinical Journal of Pain Publish Ahead of Print DOI:10.1097/AJP.0000000000000473 Capsaicin 8% patch repeat treatment in non-diabetic peripheral neuropathic pain: a 52- week, open-label, single-arm,
Neuropathic Pain in Palliative Care
 Neuropathic Pain in Palliative Care Neuropathic Pain in Advanced Cancer Affects 40% of patients Multiple concurrent pains are common Often complex pathophysiology with mixed components Nocioceptive Neuropathic
Neuropathic Pain in Palliative Care Neuropathic Pain in Advanced Cancer Affects 40% of patients Multiple concurrent pains are common Often complex pathophysiology with mixed components Nocioceptive Neuropathic
Scottish Medicines Consortium
 Scottish Medicines Consortium pregabalin, 25mg, 50mg, 75mg, 100mg, 150mg, 200mg, 225mg, 300mg capsules (Lyrica ) No. (389/07) Pfizer Limited 6 July 2007 The Scottish Medicines Consortium has completed
Scottish Medicines Consortium pregabalin, 25mg, 50mg, 75mg, 100mg, 150mg, 200mg, 225mg, 300mg capsules (Lyrica ) No. (389/07) Pfizer Limited 6 July 2007 The Scottish Medicines Consortium has completed
Medications for the Treatment of Neuropathic Pain
 Medications for the Treatment of Neuropathic Pain February 23, 2011 Jinny Tavee, MD Associate Professor Neurological Institute Cleveland Clinic Foundation Neuropathic Pain Pain, paresthesias, and sensory
Medications for the Treatment of Neuropathic Pain February 23, 2011 Jinny Tavee, MD Associate Professor Neurological Institute Cleveland Clinic Foundation Neuropathic Pain Pain, paresthesias, and sensory
Key words: Capsaicin, patch, CP, topical capsaicin, neuropathic pain, peripheral neuropathic pain, PNP, high concentration CP
 Pain Physician 2017; 20:27-35 ISSN 1533-3159 Randomized Trial Efficacy and Safety of 0.625% and 1.25% Capsaicin Patch in Peripheral Neuropathic Pain: Multi-Center, Randomized, and Semi-Double Blind Controlled
Pain Physician 2017; 20:27-35 ISSN 1533-3159 Randomized Trial Efficacy and Safety of 0.625% and 1.25% Capsaicin Patch in Peripheral Neuropathic Pain: Multi-Center, Randomized, and Semi-Double Blind Controlled
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Lyrica) Reference Number: HIM.PA.64 Effective Date: 12/14 Last Review Date: 08/17 Line of Business: Health Insurance Marketplace Revision Log See Important Reminder at the end of this
Clinical Policy: (Lyrica) Reference Number: HIM.PA.64 Effective Date: 12/14 Last Review Date: 08/17 Line of Business: Health Insurance Marketplace Revision Log See Important Reminder at the end of this
An Open-Label Study of the Lidocaine Patch 5% in Painful Idiopathic Sensory Polyneuropathy
 Blackwell Science, LtdOxford, UKPMEPain Medicine1526-2375American Academy of Pain MedicineSeptember/October 20056 5379384Original ArticleLidocaine Patch 5%Herrmann et al. PAIN MEDICINE Volume 6 Number
Blackwell Science, LtdOxford, UKPMEPain Medicine1526-2375American Academy of Pain MedicineSeptember/October 20056 5379384Original ArticleLidocaine Patch 5%Herrmann et al. PAIN MEDICINE Volume 6 Number
IF I M NOT TREATING WITH OPIOIDS, THEN WHAT AM I SUPPOSED TO USE?
 NON-OPIOID TREATMENT OPTIONS FOR CHRONIC PAIN Alison Knutson, PharmD, BCACP Medication Management Pharmacist Park Nicollet Creekside Clinic Dr. Knutson indicated no potential conflict of interest to this
NON-OPIOID TREATMENT OPTIONS FOR CHRONIC PAIN Alison Knutson, PharmD, BCACP Medication Management Pharmacist Park Nicollet Creekside Clinic Dr. Knutson indicated no potential conflict of interest to this
Emla (lidocaine 2.5% and prilocaine 2.5%), Lidocaine Topical 5%, Pliaglis Cream (lidocaine 7% and tetracaine 7%), Tetravex Gel (tetracaine 2%)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Lidocaine Topicals Page: 1 of 5 Last Review Date: September 20, 2018 Lidocaine Topicals Description
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Lidocaine Topicals Page: 1 of 5 Last Review Date: September 20, 2018 Lidocaine Topicals Description
Diagnosis and Treatment of Postherpetic Neuralgia
 J KMA Special Issue Diagnosis and Treatment of Postherpetic Neuralgia Myung Ha Yoon, MD Department of Anesthesiology and Pain Medicine, Chonnam National University Medical School E mail : mhyoon@jnu.ac.kr
J KMA Special Issue Diagnosis and Treatment of Postherpetic Neuralgia Myung Ha Yoon, MD Department of Anesthesiology and Pain Medicine, Chonnam National University Medical School E mail : mhyoon@jnu.ac.kr
Neuropathic pain (pain due to nerve damage)
 Neuropathic pain (pain due to nerve damage) Clinical Guideline Pain can be nociceptive, neuropathic or mixed. The neuropathic component of pain generally responds poorly to conventional analgesics. Consider
Neuropathic pain (pain due to nerve damage) Clinical Guideline Pain can be nociceptive, neuropathic or mixed. The neuropathic component of pain generally responds poorly to conventional analgesics. Consider
A review of Neuropathic Pain: From Guidelines to Clinical Practice
 Pain Ther (2017) 6 (Suppl 1):S35 S42 DOI 10.1007/s40122-017-0087-0 REVIEW A review of Neuropathic Pain: From Guidelines to Clinical Practice Giorgio Cruccu. Andrea Truini Received: October 12, 2017 Ó The
Pain Ther (2017) 6 (Suppl 1):S35 S42 DOI 10.1007/s40122-017-0087-0 REVIEW A review of Neuropathic Pain: From Guidelines to Clinical Practice Giorgio Cruccu. Andrea Truini Received: October 12, 2017 Ó The
Sponsor. Generic drug name. Trial indication(s) Protocol number. Protocol title. Phase of Drug Development. Study Start/End Dates
 Sponsor Novartis Generic drug name SAB378 [Naphthalen-1-yl-(4-pentyloxynaphthalen-1-yl)methanone] Trial indication(s) Postherpetic neuralgia (PHN) Protocol number CSAB378A2201 Protocol title A multicenter,
Sponsor Novartis Generic drug name SAB378 [Naphthalen-1-yl-(4-pentyloxynaphthalen-1-yl)methanone] Trial indication(s) Postherpetic neuralgia (PHN) Protocol number CSAB378A2201 Protocol title A multicenter,
A Review of Neuropathic Pain: From Diagnostic Tests to Mechanisms
 DOI 10.1007/s40122-017-0085-2 REVIEW A Review of Neuropathic Pain: From Diagnostic Tests to Mechanisms Andrea Truini Received: September 19, 2017 Ó The Author(s) 2017. This article is an open access publication
DOI 10.1007/s40122-017-0085-2 REVIEW A Review of Neuropathic Pain: From Diagnostic Tests to Mechanisms Andrea Truini Received: September 19, 2017 Ó The Author(s) 2017. This article is an open access publication
Effect of Intravenous Lidocaine on the Neuropathic Pain of Failed Back Surgery Syndrome
 Original Article Korean J Pain 2012 April; Vol. 25, No. 2: 94-98 pissn 2005-9159 eissn 2093-0569 http://dx.doi.org/10.3344/kjp.2012.25.2.94 Effect of Intravenous Lidocaine on the Neuropathic Pain of Failed
Original Article Korean J Pain 2012 April; Vol. 25, No. 2: 94-98 pissn 2005-9159 eissn 2093-0569 http://dx.doi.org/10.3344/kjp.2012.25.2.94 Effect of Intravenous Lidocaine on the Neuropathic Pain of Failed
This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data.
 abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data. The synopsis
abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data. The synopsis
Managing Diabetic Peripheral Neuropathic Pain
 Managing Diabetic Peripheral Neuropathic Pain Juzar Hooker Consulting Neurologist, Aga Khan University Hospital, Nairobi juzar.hooker@aku.edu Disclosure Eli lilly (have not driven, reviewed or controlled
Managing Diabetic Peripheral Neuropathic Pain Juzar Hooker Consulting Neurologist, Aga Khan University Hospital, Nairobi juzar.hooker@aku.edu Disclosure Eli lilly (have not driven, reviewed or controlled
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Lidoderm, ZTlido) Reference Number: CP.PMN.08 Effective Date: 09.01.06 Last Review Date: 08.18 Line of Business: Commercial, HIM, Medicaid Revision Log See Important Reminder at the end
Clinical Policy: (Lidoderm, ZTlido) Reference Number: CP.PMN.08 Effective Date: 09.01.06 Last Review Date: 08.18 Line of Business: Commercial, HIM, Medicaid Revision Log See Important Reminder at the end
New Medicines Profile
 New Medicines Profile August 2008 Issue. 08/07 Lidocaine 70mg/tetracaine 70mg medicated plaster (Rapydan ) Concise evaluated information to support the managed entry of new medicines in the NHS Summary
New Medicines Profile August 2008 Issue. 08/07 Lidocaine 70mg/tetracaine 70mg medicated plaster (Rapydan ) Concise evaluated information to support the managed entry of new medicines in the NHS Summary
Predictive Value of Lidocaine for Treatment Success of Oxcarbazepine in Patients with Neuropathic Pain Syndrome
 Pain Ther (2013) 2:49 56 DOI 10.1007/s40122-013-0007-x ORIGINAL RESEARCH Predictive Value of Lidocaine for Treatment Success of Oxcarbazepine in Patients with Neuropathic Pain Syndrome Sivan Schipper Andreas
Pain Ther (2013) 2:49 56 DOI 10.1007/s40122-013-0007-x ORIGINAL RESEARCH Predictive Value of Lidocaine for Treatment Success of Oxcarbazepine in Patients with Neuropathic Pain Syndrome Sivan Schipper Andreas
A Randomized, Double-Blind, Controlled Study of NGX-4010, a. Capsaicin 8% Dermal Patch, for the Treatment of Painful HIVassociated
 JAIDS Journal of Acquired Immune Deficiency Syndromes Publish Ahead of Print DOI: 10.1097/QAI.0b013e31823e31f7 A Randomized, Double-Blind, Controlled Study of NGX-4010, a Capsaicin 8% Dermal Patch, for
JAIDS Journal of Acquired Immune Deficiency Syndromes Publish Ahead of Print DOI: 10.1097/QAI.0b013e31823e31f7 A Randomized, Double-Blind, Controlled Study of NGX-4010, a Capsaicin 8% Dermal Patch, for
Non-Opioid Drugs to Treat Neuropathic Pain. March 2018
 Non-Opioid Drugs to Treat Neuropathic Pain Final Report March 2018 This report is intended only for state employees in states participating in the Drug Effectiveness Review Project (DERP). Do not distribute
Non-Opioid Drugs to Treat Neuropathic Pain Final Report March 2018 This report is intended only for state employees in states participating in the Drug Effectiveness Review Project (DERP). Do not distribute
Ingredient Efficacy Evaluation
 Ingredient Efficacy Evaluation Thousands of physicians nationwide trust LidoPro ointment for safe, effective, and convenient pain management. LidoPro contains ingredients that deliver anti-inflammatory
Ingredient Efficacy Evaluation Thousands of physicians nationwide trust LidoPro ointment for safe, effective, and convenient pain management. LidoPro contains ingredients that deliver anti-inflammatory
Summary of Results for Laypersons
 What was the Study Called? Summary of Results for Laypersons A Phase 2a Enriched Enrollment Randomized Withdrawal Study to Assess Analgesic Efficacy and Safety of ASP8477 in Subjects with Peripheral Neuropathic
What was the Study Called? Summary of Results for Laypersons A Phase 2a Enriched Enrollment Randomized Withdrawal Study to Assess Analgesic Efficacy and Safety of ASP8477 in Subjects with Peripheral Neuropathic
Advice following an Independent Review Panel (IRP)
 Scottish Medicines Consortium Advice following an Independent Review Panel (IRP) Pregabalin 25, 50, 75, 100, 150, 200 and 300mg capsules (Lyrica ) Pfizer No. 157/05 7 July 2006 The Scottish Medicines Consortium
Scottish Medicines Consortium Advice following an Independent Review Panel (IRP) Pregabalin 25, 50, 75, 100, 150, 200 and 300mg capsules (Lyrica ) Pfizer No. 157/05 7 July 2006 The Scottish Medicines Consortium
Accelerating the Development of Enhanced Pain Treatments March 25, Bermuda
 Accelerating the Development of Enhanced Pain Treatments March 25, 2011 - Bermuda Accelerating the Development of Enhanced Pain Treatments March 25, 2011 - Bermuda Proof-of-concept trials Ian Gilron, MD,
Accelerating the Development of Enhanced Pain Treatments March 25, 2011 - Bermuda Accelerating the Development of Enhanced Pain Treatments March 25, 2011 - Bermuda Proof-of-concept trials Ian Gilron, MD,
Statistics as a Tool. A set of tools for collecting, organizing, presenting and analyzing numerical facts or observations.
 Statistics as a Tool A set of tools for collecting, organizing, presenting and analyzing numerical facts or observations. Descriptive Statistics Numerical facts or observations that are organized describe
Statistics as a Tool A set of tools for collecting, organizing, presenting and analyzing numerical facts or observations. Descriptive Statistics Numerical facts or observations that are organized describe
Pathophysiology of Pain
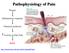 Pathophysiology of Pain Wound Inflammatory response Chemical mediators Activity in Pain Path PAIN http://neuroscience.uth.tmc.edu/s2/chapter08.html Chris Cohan, Ph.D. Dept. of Pathology/Anat Sci University
Pathophysiology of Pain Wound Inflammatory response Chemical mediators Activity in Pain Path PAIN http://neuroscience.uth.tmc.edu/s2/chapter08.html Chris Cohan, Ph.D. Dept. of Pathology/Anat Sci University
Choosing the Correct Statistical Test
 Choosing the Correct Statistical Test T racie O. Afifi, PhD Departments of Community Health Sciences & Psychiatry University of Manitoba Department of Community Health Sciences COLLEGE OF MEDICINE, FACULTY
Choosing the Correct Statistical Test T racie O. Afifi, PhD Departments of Community Health Sciences & Psychiatry University of Manitoba Department of Community Health Sciences COLLEGE OF MEDICINE, FACULTY
High-dose 8% capsaicin patch in treatment of chemotherapyinduced peripheral neuropathy: single-center experience
 Med Oncol (2017) 34:162 DOI 10.1007/s12032-017-1015-1 ORIGINAL PAPER High-dose 8% capsaicin patch in treatment of chemotherapyinduced peripheral neuropathy: single-center experience Iwona Filipczak-Bryniarska
Med Oncol (2017) 34:162 DOI 10.1007/s12032-017-1015-1 ORIGINAL PAPER High-dose 8% capsaicin patch in treatment of chemotherapyinduced peripheral neuropathy: single-center experience Iwona Filipczak-Bryniarska
Individual Study Table Referring to Part of Dossier: Volume: Page:
 2.0 Synopsis Abbott Laboratories Name of Study Drug: ABT-639 Name of Active Ingredient: ABT-639 Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority Use Only) Title
2.0 Synopsis Abbott Laboratories Name of Study Drug: ABT-639 Name of Active Ingredient: ABT-639 Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority Use Only) Title
COMPOUNDING PHARMACY SOLUTIONS PRESCRIPTION COMPOUNDING FOR PAIN MANAGEMENT
 JUNE 2012 COMPOUNDING PHARMACY SOLUTIONS PRESCRIPTION COMPOUNDING WWW.CPSRXS. COM We customize individual prescriptions for the specific needs of our patients. INSIDE THIS ISSUE: Acute Pain 2 Neuropathic
JUNE 2012 COMPOUNDING PHARMACY SOLUTIONS PRESCRIPTION COMPOUNDING WWW.CPSRXS. COM We customize individual prescriptions for the specific needs of our patients. INSIDE THIS ISSUE: Acute Pain 2 Neuropathic
NORLAND AVENUE PHARMACY PRESCRIPTION COMPOUNDING FOR PAIN MANAGEMENT
 NOVEMBER 2011 NORLAND AVENUE PHARMACY PRESCRIPTION COMPOUNDING N ORLANDA VENUEP HARMACY. COM We customize individual prescriptions for the specific needs of our patients. INSIDE THIS ISSUE: Sciatic Pain
NOVEMBER 2011 NORLAND AVENUE PHARMACY PRESCRIPTION COMPOUNDING N ORLANDA VENUEP HARMACY. COM We customize individual prescriptions for the specific needs of our patients. INSIDE THIS ISSUE: Sciatic Pain
Clinical Policy Title: Intravenous lidocaine infusion for neuropathic pain
 Clinical Policy Title: Intravenous lidocaine infusion for neuropathic pain Clinical Policy Number: 03.03.08 Effective Date: June 1, 2014 Initial Review Date: January 19, 2014 Most Recent Review Date: January
Clinical Policy Title: Intravenous lidocaine infusion for neuropathic pain Clinical Policy Number: 03.03.08 Effective Date: June 1, 2014 Initial Review Date: January 19, 2014 Most Recent Review Date: January
Peripheral Subcutaneous Field Stimulation
 Peripheral Subcutaneous Field Stimulation Policy Number: 7.01.139 Last Review: 9/2014 Origination: 7/2013 Next Review: 1/2015 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide
Peripheral Subcutaneous Field Stimulation Policy Number: 7.01.139 Last Review: 9/2014 Origination: 7/2013 Next Review: 1/2015 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide
GENERAL PAIN DEFINITIONS
 I. OVERVIEW GENERAL PAIN DEFINITIONS Charles E. Argoff, MD CHAPTER 1 1. What is pain? Some dictionaries define pain as An unpleasant sensation, occurring in varying degrees of severity as a consequence
I. OVERVIEW GENERAL PAIN DEFINITIONS Charles E. Argoff, MD CHAPTER 1 1. What is pain? Some dictionaries define pain as An unpleasant sensation, occurring in varying degrees of severity as a consequence
New Developments in the Treatment Algorithm for Peripheral Neuropathic Painpme_
 Pain Medicine 2011; 12: S100 S108 Wiley Periodicals, Inc. New Developments in the Treatment Algorithm for Peripheral Neuropathic Painpme_1160 100..108 Oscar de Leon-Casasola, MD School of Medicine and
Pain Medicine 2011; 12: S100 S108 Wiley Periodicals, Inc. New Developments in the Treatment Algorithm for Peripheral Neuropathic Painpme_1160 100..108 Oscar de Leon-Casasola, MD School of Medicine and
EFFECTS OF CAPSAICIN ON RAT SCIATIC NERVE IN VINCRISTINE-INDUCED NEUROPATHIC PAIN MODEL
 IJPSR (2013), Vol. 4, Issue 2 (Research Article) Received on 17 October, 2012; received in revised form, 29 November, 2012; accepted, 30 January, 2013 EFFECTS OF CAPSAICIN ON RAT SCIATIC NERVE IN VINCRISTINE-INDUCED
IJPSR (2013), Vol. 4, Issue 2 (Research Article) Received on 17 October, 2012; received in revised form, 29 November, 2012; accepted, 30 January, 2013 EFFECTS OF CAPSAICIN ON RAT SCIATIC NERVE IN VINCRISTINE-INDUCED
REFERENCE CODE GDHC406DFR PUBLICAT ION DATE APRIL 2014 LIDODERM/VERSATIS (NEUROPATHIC PAIN) - FORECAST AND MARKET ANALYSIS TO 2022
 REFERENCE CODE GDHC406DFR PUBLICAT ION DATE APRIL 2014 LIDODERM/VERSATIS (NEUROPATHIC PAIN) - Executive Summary Table below provides a summary of the key metrics for Lidoderm/Versatis in the 7MM neuropathic
REFERENCE CODE GDHC406DFR PUBLICAT ION DATE APRIL 2014 LIDODERM/VERSATIS (NEUROPATHIC PAIN) - Executive Summary Table below provides a summary of the key metrics for Lidoderm/Versatis in the 7MM neuropathic
Literature Scan: Topical Analgesics
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Gabapentin vs. Amitriptyline in Painful Diabetic Neuropathy: An Open-Label Pilot Study
 280 Journal of Pain and Symptom Management Vol. 20 No. 4 October 2000 Original Article Gabapentin vs. Amitriptyline in Painful Diabetic Neuropathy: An Open-Label Pilot Study Carlo Dallocchio, MD, Carlo
280 Journal of Pain and Symptom Management Vol. 20 No. 4 October 2000 Original Article Gabapentin vs. Amitriptyline in Painful Diabetic Neuropathy: An Open-Label Pilot Study Carlo Dallocchio, MD, Carlo
Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD
 Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD 1630 Main Street Suite 215 Chester, MD 410-571-9000 www.4-no-pain.com
Brian Kahan, D.O. FAAPMR, DABPM, DAOCRM, FIPP Center for Pain Medicine and Physiatric Rehabilitation 2002 Medical Parkway Suite 150 Annapolis, MD 1630 Main Street Suite 215 Chester, MD 410-571-9000 www.4-no-pain.com
Treatment of Neuropathic Pain: What Does the Evidence Say? or Just the Facts Ma am
 Treatment of Neuropathic Pain: What Does the Evidence Say? or Just the Facts Ma am Tim R Brown, PharmD, BCACP, FASHP Director of Clinical Pharmacotherapy Cleveland Clinic Akron General Center for Family
Treatment of Neuropathic Pain: What Does the Evidence Say? or Just the Facts Ma am Tim R Brown, PharmD, BCACP, FASHP Director of Clinical Pharmacotherapy Cleveland Clinic Akron General Center for Family
PAIN MEDICINE FOR THE NON-PAIN SPECIALIST 2017
 PAIN MEDICINE FOR THE NON-PAIN SPECIALIST 2017 FEBRUARY 16-18, 2017 JW MARRIOTT DESERT SPRINGS RESORT & SPA PALM DESERT, CALIFORNIA Learn the latest treatment strategies and multidisciplinary management
PAIN MEDICINE FOR THE NON-PAIN SPECIALIST 2017 FEBRUARY 16-18, 2017 JW MARRIOTT DESERT SPRINGS RESORT & SPA PALM DESERT, CALIFORNIA Learn the latest treatment strategies and multidisciplinary management
The New Types of Pharmacological and Other Treatments of Pain
 The New Types of Pharmacological and Other Treatments of Pain Prof. Richard Rokyta MD, PhD, DSc, FCMA Charles University in Prague, Third Faculty of Medicine, Department of Normal, Pathological and Clinical
The New Types of Pharmacological and Other Treatments of Pain Prof. Richard Rokyta MD, PhD, DSc, FCMA Charles University in Prague, Third Faculty of Medicine, Department of Normal, Pathological and Clinical
Pain Management in the
 Pain Management in the Elderly Meri Hix, PharmD, CGP, BCPS Associate Professor of Pharmacy Practice Midwestern University Chicago College of Pharmacy No conflicts of interest to declare Objectives Discuss
Pain Management in the Elderly Meri Hix, PharmD, CGP, BCPS Associate Professor of Pharmacy Practice Midwestern University Chicago College of Pharmacy No conflicts of interest to declare Objectives Discuss
Capsaicinoids in the treatment of neuropathic pain: a review
 501576TAN7110.1177/1756285613501576Therapeutic Advances in Neurological DisordersJF Peppin and M Pappagallo 2013501576 Therapeutic Advances in Neurological Disorders Review Capsaicinoids in the treatment
501576TAN7110.1177/1756285613501576Therapeutic Advances in Neurological DisordersJF Peppin and M Pappagallo 2013501576 Therapeutic Advances in Neurological Disorders Review Capsaicinoids in the treatment
Pain. Types of Pain. Types of Pain 8/21/2013
 Pain 1 Types of Pain Acute Pain Complex combination of sensory, perceptual, & emotional experiences as a result of a noxious stimulus Mediated by rapidly conducting nerve pathways & associated with increased
Pain 1 Types of Pain Acute Pain Complex combination of sensory, perceptual, & emotional experiences as a result of a noxious stimulus Mediated by rapidly conducting nerve pathways & associated with increased
CHAPTER 4 PAIN AND ITS MANAGEMENT
 CHAPTER 4 PAIN AND ITS MANAGEMENT Pain Definition: An unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms of such damage. Types of Pain
CHAPTER 4 PAIN AND ITS MANAGEMENT Pain Definition: An unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms of such damage. Types of Pain
16 year old with Disabling Chest Wall Pain after Thoracoscopic Talc Pleurodesis for Treatment of Recurrent Spontaneous Pneumothoraces
 16 year old with Disabling Chest Wall Pain after Thoracoscopic Talc Pleurodesis for Treatment of Recurrent Spontaneous Pneumothoraces Moderators: Kendra Grim, MD, Robert T. Wilder, MD, PhD Institution:
16 year old with Disabling Chest Wall Pain after Thoracoscopic Talc Pleurodesis for Treatment of Recurrent Spontaneous Pneumothoraces Moderators: Kendra Grim, MD, Robert T. Wilder, MD, PhD Institution:
REFERENCE CODE GDHC407DFR PUBLICAT ION DATE APRIL 2014 QUTENZA (NEUROPATHIC PAIN) - FORECAST AND MARKET ANALYSIS TO 2022
 REFERENCE CODE GDHC407DFR PUBLICAT ION DATE APRIL 2014 QUTENZA (NEUROPATHIC PAIN) - Executive Summary Table below provides a summary of the key metrics for Qutenza in the 7MM neuropathic pain markets during
REFERENCE CODE GDHC407DFR PUBLICAT ION DATE APRIL 2014 QUTENZA (NEUROPATHIC PAIN) - Executive Summary Table below provides a summary of the key metrics for Qutenza in the 7MM neuropathic pain markets during
Superiority of capsaicin 8% patch versus oral pregabalin on dynamic mechanical allodynia in patients with peripheral neuropathic pain
 SHORT COMMUNICATION Superiority of capsaicin 8% patch versus oral pregabalin on dynamic mechanical allodynia in patients with peripheral neuropathic pain G. Cruccu 1, T.J. Nurmikko 2, E. Ernault 3, F.K.
SHORT COMMUNICATION Superiority of capsaicin 8% patch versus oral pregabalin on dynamic mechanical allodynia in patients with peripheral neuropathic pain G. Cruccu 1, T.J. Nurmikko 2, E. Ernault 3, F.K.
NEUROPATHIC CANCER PAIN STANDARDS AND GUIDELINES
 NEUROPATHIC CANCER PAIN STANDARDS AND GUIDELINES GENERAL PRINCIPLES Neuropathic pain may be relieved in the majority of patients by multimodal management A careful history and examination are essential.
NEUROPATHIC CANCER PAIN STANDARDS AND GUIDELINES GENERAL PRINCIPLES Neuropathic pain may be relieved in the majority of patients by multimodal management A careful history and examination are essential.
Choose a category. You will be given the answer. You must give the correct question. Click to begin.
 Instructions for using this template. Remember this is Jeopardy, so where I have written Answer this is the prompt the students will see, and where I have Question should be the student s response. To
Instructions for using this template. Remember this is Jeopardy, so where I have written Answer this is the prompt the students will see, and where I have Question should be the student s response. To
Research Article The Need for Improved Management of Painful Diabetic Neuropathy in Primary Care
 Pain Research and Management Volume 2016, Article ID 1974863, 4 pages http://dx.doi.org/10.1155/2016/1974863 Research Article The Need for Improved Management of Painful Diabetic Neuropathy in Primary
Pain Research and Management Volume 2016, Article ID 1974863, 4 pages http://dx.doi.org/10.1155/2016/1974863 Research Article The Need for Improved Management of Painful Diabetic Neuropathy in Primary
Qutenza. Pain Management Team Patient Information Leaflet
 Qutenza Pain Management Team Patient Information Leaflet Introduction This leaflet is for people who are considering using Qutenza. It gives information on what Qutenza is, what the treatment involves
Qutenza Pain Management Team Patient Information Leaflet Introduction This leaflet is for people who are considering using Qutenza. It gives information on what Qutenza is, what the treatment involves
IL DOLORE NEOPLASTICO NEI SECONDARISMI OSSEI E SCRAMBLER THERAPY
 IL DOLORE NEOPLASTICO NEI SECONDARISMI OSSEI E SCRAMBLER THERAPY Ka#a Bencardino Niguarda Cancer Center, Grande Ospedale Metropolitano Niguarda, Milano, Italia Scrambler therapy Scrambler Therapy is a
IL DOLORE NEOPLASTICO NEI SECONDARISMI OSSEI E SCRAMBLER THERAPY Ka#a Bencardino Niguarda Cancer Center, Grande Ospedale Metropolitano Niguarda, Milano, Italia Scrambler therapy Scrambler Therapy is a
Peripheral Subcutaneous Field Stimulation
 Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
Applies to all products administered or underwritten by Blue Cross and Blue Shield of Louisiana and its subsidiary, HMO Louisiana, Inc.(collectively referred to as the Company ), unless otherwise provided
K30 case study: innovative treatment approach to myofascial neck pain. Marc Brodsky, MD UCLA Center for East-West Medicine May 27, 2008
 K30 case study: innovative treatment approach to myofascial neck pain Marc Brodsky, MD UCLA Center for East-West Medicine May 27, 2008 Patient presentation 24 yo female medical student with a oneyear h/o
K30 case study: innovative treatment approach to myofascial neck pain Marc Brodsky, MD UCLA Center for East-West Medicine May 27, 2008 Patient presentation 24 yo female medical student with a oneyear h/o
National Horizon Scanning Centre. Pregabalin (Lyrica) for fibromyalgia. September 2007
 Pregabalin (Lyrica) for fibromyalgia September 2007 This technology summary is based on information available at the time of research and a limited literature search. It is not intended to be a definitive
Pregabalin (Lyrica) for fibromyalgia September 2007 This technology summary is based on information available at the time of research and a limited literature search. It is not intended to be a definitive
Overview. Normally, the process is completely reversible.
 Overview Local anesthetics produce a transient and reversible loss of sensation (analgesia) in a circumscribed region of the body without loss of consciousness. Normally, the process is completely reversible.
Overview Local anesthetics produce a transient and reversible loss of sensation (analgesia) in a circumscribed region of the body without loss of consciousness. Normally, the process is completely reversible.
ABSTRACT. Original Articles /
 Original Articles / 550; 7(): 9-95 J Thai Rehabil Med 007; 7(): 9-95..,..,..,..,.. ABSTRACT Efficacy of topical capsaicin and methyl salicylate as an adjuvant therapy in chronic non-specific low back pain
Original Articles / 550; 7(): 9-95 J Thai Rehabil Med 007; 7(): 9-95..,..,..,..,.. ABSTRACT Efficacy of topical capsaicin and methyl salicylate as an adjuvant therapy in chronic non-specific low back pain
Use of Pregabalin for Postoperative Pain: Outcomes in 2 Trials
 Use of Pregabalin for Postoperative Pain: Outcomes in 2 Trials Jacques E. Chelly, 1 Neil Singla, 2 David R. Lionberger, 3 Henrik Kehlet, 4 Luis Sanin, 5 Jonathan Sporn, 5 Ruoyong Yang, 5 Raymond C. Cheung,
Use of Pregabalin for Postoperative Pain: Outcomes in 2 Trials Jacques E. Chelly, 1 Neil Singla, 2 David R. Lionberger, 3 Henrik Kehlet, 4 Luis Sanin, 5 Jonathan Sporn, 5 Ruoyong Yang, 5 Raymond C. Cheung,
Is Topical Clonazepam More Effective Than Oral Clonazepam in Treatment of Burning Mouth Syndrome (BMS)?
 Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2014 Is Topical Clonazepam More Effective
Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2014 Is Topical Clonazepam More Effective
Pharmacologic Aspect of Neuropathic Pain
 September 911, 2015, Banda Aceh, Indonesia Pharmacologic Aspect of Neuropathic Pain 1 Endang Mutiawati, 2 Imai Indra, 3 Syahrul, 4 *Mulyadi 1 Department of Neurology, Faculty of Medicine, Syiah Kuala University,
September 911, 2015, Banda Aceh, Indonesia Pharmacologic Aspect of Neuropathic Pain 1 Endang Mutiawati, 2 Imai Indra, 3 Syahrul, 4 *Mulyadi 1 Department of Neurology, Faculty of Medicine, Syiah Kuala University,
754 Journal of Pain and Symptom Management Vol. 42 No. 5 November 2011
 754 Journal of Pain and Symptom Management Vol. 42 No. 5 November 2011 Brief Report Characterization of Abdominal Pain During Methylnaltrexone Treatment of Opioid-Induced Constipation in Advanced Illness:
754 Journal of Pain and Symptom Management Vol. 42 No. 5 November 2011 Brief Report Characterization of Abdominal Pain During Methylnaltrexone Treatment of Opioid-Induced Constipation in Advanced Illness:
Setting The setting was primary and secondary care. The economic study was carried out in Canada.
 Cost-effectiveness of pregabalin for the management of neuropathic pain associated with diabetic peripheral neuropathy and postherpetic neuralgia: a Canadian perspective Tarride J E, Gordon A, Vera-Llonch
Cost-effectiveness of pregabalin for the management of neuropathic pain associated with diabetic peripheral neuropathy and postherpetic neuralgia: a Canadian perspective Tarride J E, Gordon A, Vera-Llonch
*Please see amendment for Pennsylvania Medicaid at the
 Number: 0725 Policy *Please see amendment for Pennsylvania Medicaid at the end of this CPB. I. Aetna considers the use of antivirals (oral), gabapentin, intrathecal corticosteroids, lidocaine patch, opioids
Number: 0725 Policy *Please see amendment for Pennsylvania Medicaid at the end of this CPB. I. Aetna considers the use of antivirals (oral), gabapentin, intrathecal corticosteroids, lidocaine patch, opioids
GABAPENTIN BNF Gabapentin is a chemical analogue of γ-aminobutyric acid (GABA) but does not act
 GABAPENTIN BNF 4.8.1 Class: Anti-epileptic. Indications: Adjunctive treatment for partial seizures with or without secondary generalisation; 1,2 neuropathic pain of any cause. 3 12 Pharmacology Gabapentin
GABAPENTIN BNF 4.8.1 Class: Anti-epileptic. Indications: Adjunctive treatment for partial seizures with or without secondary generalisation; 1,2 neuropathic pain of any cause. 3 12 Pharmacology Gabapentin
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Current Pharmacotherapy of Chronic Pain
 S16 Journal of Pain and Symptom Management Vol. 19 No. 1(Suppl.) January 2000 Proceedings Supplement NMDA-Receptor Antagonists: Evolving Role in Analgesia Current Pharmacotherapy of Chronic Pain Russell
S16 Journal of Pain and Symptom Management Vol. 19 No. 1(Suppl.) January 2000 Proceedings Supplement NMDA-Receptor Antagonists: Evolving Role in Analgesia Current Pharmacotherapy of Chronic Pain Russell
Efficacy of Levetiracetam: A Review of Three Pivotal Clinical Trials
 Epilepsia, 42(Suppl. 4):31 35, 2001 Blackwell Science, Inc. International League Against Epilepsy Efficacy of : A Review of Three Pivotal Clinical Trials Michael Privitera University of Cincinnati Medical
Epilepsia, 42(Suppl. 4):31 35, 2001 Blackwell Science, Inc. International League Against Epilepsy Efficacy of : A Review of Three Pivotal Clinical Trials Michael Privitera University of Cincinnati Medical
NIH Public Access Author Manuscript Ann Neurol. Author manuscript; available in PMC 2009 October 19.
 NIH Public Access Author Manuscript Published in final edited form as: Ann Neurol. 2009 March ; 65(3): 348 351. doi:10.1002/ana.21601. Sympathetic Block with Botulinum Toxin to Treat Complex Regional Pain
NIH Public Access Author Manuscript Published in final edited form as: Ann Neurol. 2009 March ; 65(3): 348 351. doi:10.1002/ana.21601. Sympathetic Block with Botulinum Toxin to Treat Complex Regional Pain
Approaches to Managing Neuropathic Pain. Nov 7, 2017
 1 Approaches to Managing Neuropathic Pain Nov 7, 2017 3 Learning objectives Review the current Canadian Guidelines on Neuropathic Pain Review the diagnosis and options available for the management of Neuropathic
1 Approaches to Managing Neuropathic Pain Nov 7, 2017 3 Learning objectives Review the current Canadian Guidelines on Neuropathic Pain Review the diagnosis and options available for the management of Neuropathic
Pain: Current Understanding of Assessment, Management, and Treatments. Sponsored by the American Pain Sciety
 Pain: Current Understanding of Assessment, Management, and Treatments Sponsored by the American Pain Sciety This program is supported by an unrestricted educational grant from NPC. For permission to reprint
Pain: Current Understanding of Assessment, Management, and Treatments Sponsored by the American Pain Sciety This program is supported by an unrestricted educational grant from NPC. For permission to reprint
Peripheral Subcutaneous Field Stimulation
 Peripheral Subcutaneous Field Stimulation Policy Number: 7.01.139 Last Review: 3/2018 Origination: 7/2013 Next Review: 9/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide
Peripheral Subcutaneous Field Stimulation Policy Number: 7.01.139 Last Review: 3/2018 Origination: 7/2013 Next Review: 9/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide
Acute Pain NETP: SEPTEMBER 2013 COHORT
 Acute Pain NETP: SEPTEMBER 2013 COHORT Pain & Suffering an unpleasant sensory & emotional experience associated with actual or potential tissue damage, or described in terms of such damage International
Acute Pain NETP: SEPTEMBER 2013 COHORT Pain & Suffering an unpleasant sensory & emotional experience associated with actual or potential tissue damage, or described in terms of such damage International
TITLE: Gabapentin for HIV-associated Neuropathic Pain: A Review of the Clinical Effectiveness
 TITLE: Gabapentin for HIV-associated Neuropathic Pain: A Review of the Clinical Effectiveness DATE: 22 January 2016 CONTEXT AND POLICY ISSUES Neuropathic pain can be severe and debilitating and results
TITLE: Gabapentin for HIV-associated Neuropathic Pain: A Review of the Clinical Effectiveness DATE: 22 January 2016 CONTEXT AND POLICY ISSUES Neuropathic pain can be severe and debilitating and results
KANSAS Kansas State Board of Healing Arts. Source: Kansas State Board of Healing Arts. Approved: October 17, 1998
 KANSAS Kansas State Board of Healing Arts Source: Kansas State Board of Healing Arts Approved: October 17, 1998 GUIDELINES FOR THE USE OF CONTROLLED SUBSTANCES FOR THE TREATMENT OF PAIN Section 1: Preamble
KANSAS Kansas State Board of Healing Arts Source: Kansas State Board of Healing Arts Approved: October 17, 1998 GUIDELINES FOR THE USE OF CONTROLLED SUBSTANCES FOR THE TREATMENT OF PAIN Section 1: Preamble
Neuropathic Pain. Scott Magnuson, MD Pain Management of North Idaho, PLLC
 Neuropathic Pain Scott Magnuson, MD Pain Management of North Idaho, PLLC Pain is our friend "An unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described
Neuropathic Pain Scott Magnuson, MD Pain Management of North Idaho, PLLC Pain is our friend "An unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described
A journey towards estimand specification in pain: motivation and challenges
 Biostatistics Neuroscience Franchise A journey towards estimand specification in pain: motivation and challenges Francesca Callegari, PhD PSI Meeting Reading, UK Sep 27 th, 2017 pdated Slide Quote We shall
Biostatistics Neuroscience Franchise A journey towards estimand specification in pain: motivation and challenges Francesca Callegari, PhD PSI Meeting Reading, UK Sep 27 th, 2017 pdated Slide Quote We shall
Topical capsaicin for pain management: therapeutic potential and mechanisms of action of the new high-concentration capsaicin 8% patch
 British Journal of Anaesthesia 107 (4): 490 502 (2011) Advance Access publication 17 August 2011. doi:10.1093/bja/aer260 REVIEW ARTICLE Topical capsaicin for pain management: therapeutic potential and
British Journal of Anaesthesia 107 (4): 490 502 (2011) Advance Access publication 17 August 2011. doi:10.1093/bja/aer260 REVIEW ARTICLE Topical capsaicin for pain management: therapeutic potential and
DOSAGE FORMS AND STRENGTHS Qutenza patch contains 8% capsaicin (640 mcg/cm2). Each patch contains a total of 179 mg of capsaicin.
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use QUTENZA safely and effectively. See full prescribing information for QUTENZA. QUTENZA (capsaicin)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use QUTENZA safely and effectively. See full prescribing information for QUTENZA. QUTENZA (capsaicin)
Clinical Trial Results with OROS Ò Hydromorphone
 Vol. 33 No. 2S February 2007 Journal of Pain and Symptom Management S25 Advances in the Long-Term Management of Chronic Pain: Recent Evidence with OROS Ò Hydromorphone, a Novel, Once-Daily, Long-Acting
Vol. 33 No. 2S February 2007 Journal of Pain and Symptom Management S25 Advances in the Long-Term Management of Chronic Pain: Recent Evidence with OROS Ò Hydromorphone, a Novel, Once-Daily, Long-Acting
MEDICAL POLICY SUBJECT: KETAMINE INFUSION THERAPY FOR THE TREATMENT OF CHRONIC PAIN SYNDROMES POLICY NUMBER: CATEGORY: Technology Assessment
 Clinical criteria used to make utilization review decisions are based on credible scientific evidence published in peer reviewed medical literature generally recognized by the medical community. Guidelines
Clinical criteria used to make utilization review decisions are based on credible scientific evidence published in peer reviewed medical literature generally recognized by the medical community. Guidelines
Sharon A Stephen, PhD, ARNP, ACHPN. September 23, 2014
 Sharon A Stephen, PhD, ARNP, ACHPN September 23, 2014 Case-based presentation selected to discuss: Pain assessment Barriers to adequate pain relief Pharmacologic interventions Non-Pharmacologic interventions
Sharon A Stephen, PhD, ARNP, ACHPN September 23, 2014 Case-based presentation selected to discuss: Pain assessment Barriers to adequate pain relief Pharmacologic interventions Non-Pharmacologic interventions
The Epidemiology of Chronic Pain of Predominantly Neuropathic Origin. Results From a General Population Survey
 The Journal of Pain, Vol 7, No 4 (April), 2006: pp 281-289 Available online at www.sciencedirect.com The Epidemiology of Chronic Pain of Predominantly Neuropathic Origin. Results From a General Population
The Journal of Pain, Vol 7, No 4 (April), 2006: pp 281-289 Available online at www.sciencedirect.com The Epidemiology of Chronic Pain of Predominantly Neuropathic Origin. Results From a General Population
