Virtual Lectures Planning Committee Disclosure Summary
|
|
|
- Pierce Simon
- 5 years ago
- Views:
Transcription
1 Mayo Medical Laboratories Virtual Lectures 2014 MFMER MFMER Virtual Lectures Planning Committee Disclosure Summary As a provider accredited by ACCME, College of Medicine, Mayo Clinic (Mayo School of CPD) must ensure balance, independence, objectivity and scientific rigor in its educational activities. Course Director(s), Planning Committee Members, Faculty, and all others who are in a position to control the content of this educational activity are required to disclose all relevant financial relationships with any commercial interest related to the subject matter of the educational activity. Safeguards against commercial bias have been put in place. Faculty also will disclose any off label and/or investigational use of pharmaceuticals or instruments discussed in their presentation. Disclosure of these relevant financial relationships will be published in activity materials so those participants in the activity may formulate their own judgments regarding the presentation. Listed below are individuals with control of the content of this program who have disclosed Relevant financial relationship(s) with industry: None No relevant financial relationship(s) with industry: Joaquin Garcia, MD program presenter Curtis Hanson, MD program planning committee Sharon Preuss program planning committee Bobbi Pritt, MD, MSc, DTMH program presenter, program planning committee Cara Schmidt program planning committee References to off-label and/or investigational usage(s) of pharmaceuticals or instruments in their presentation: None MFMER 1
2 Test Utilization in the Clinical Microbiology Lab What Works and What Doesn t Bobbi Pritt, M.D. Medical Director, Clinical Parasitology Laboratory Mayo Clinic, Rochester, Minnesota, U.S.A MFMER DISCLOSURES Relevant Financial Relationship(s) None Off-Label Usage None 2016 MFMER
3 Optimizing Test Utilization in Clinical Microbiology Similar challenges as other areas of the clinical laboratory Some unique considerations: Testing is dependent on numerous clinical factors Exposure history: Geographic location/travel Contact with infected individuals or animals Immune status (HIV/AIDS, HTLV-1, chemotherapy) Current outbreaks Presence of instrumentation (e.g. catheters, prosthetic joints) 2016 MFMER Fecal Parasite testing as an example Complex testing options Traditional method for identification of parasites in feces is the Ova and Parasite examination (O&P) Appropriate for identifying many of the exotic parasites Less sensitive method for detecting the more common intestinal parasites in the United States (Giardia and Cryptosporidium) Requires up to 7 stool specimens for optimal detection of Giardia 2016 MFMER
4 Additional Detection Methods Antigen detection (Immunoassays) for Giardia and Cryptosporidium Multiplex PCR for bacterial, viral and parasitic pathogens Special staining methods: Modified acid fast stain for the coccidia Modified trichrome stain for microsporidia Individual PCR, culture and serologic tests 2016 MFMER Too many options! Common approaches: Order all of the tests! Order the routine O&P that s what I learned in medical school I don t know what my patient has so I want the most inclusive test Preferred approach: Order of selective tests based on the clinical presentation and risk factors. Solution: An algorithm was developed based on published guidelines and clinician input 2016 MFMER
5 MFMER A real-life experience The situation: the lab supervisor felt that the O&P was over-ordered and that physicians did not use our algorithm My approach: Step 1 examine the data 2016 MFMER
6 Stool Parasite Exam Test volumes Only <5% of our specimens were positive O&P Giardia EIA Cryptosporidium EIA Jan-05 Mar-05 May-05 Jul-05 Sep-05 Nov-05 Jan-06 Mar-06 May-06 Jul-06 Sep-06 Nov-06 Jan-07 Mar-07 May-07 Jul-07 Sep-07 Nov-07 Jan-08 Mar-08 May-08 Jul-08 Sept-08 Nov-08 Jan-09 Mar-09 May MFMER A real-life experience continued Step 2 perform chart review Most O&P orders were incorrectly placed Patients were actually more likely to have Giardia or Cryptosporidium for which EIAs were recommended. Step 3 Talk to providers Most did not know that an algorithm was available for guiding orders Assessment The algorithm was not being used to guide O&P testing Plan Provide education 2016 MFMER
7 30 min Educational Video 60 min Educational Program Ongoing education To in-house physicians And residents O&P Educational Bulletin Giardia EIA Cryptosporidium EIA Mar 07 May-07 Jul-07 Sep-07 Nov-07 Jan-08 Mar-08 May-08 Jul-08 Sept-08 Nov-08 Jan-09 Mar-09 May-09 Jul-09 Sep-09 Nov-09 Jan-10 Mar-10 May-10 Jul-10 Sept-10 Nov-10 Jan-11 Mar-11 May-11 Jul-11 Sept-11 Nov-11 Jan-12 Mar MFMER Conclusion Educational efforts are not enough to change ordering practices Why? Messages don t reach the right people Habit/patterns are hard to break Regular influx of new trainees and physicians 2016 MFMER
8 Can we turn this algorithm into a cascade? Cascade definition: a sequence of tests that are performed by the lab based on results of preceding tests. The cascade should be based on accepted guidelines and the components need to be transparent to providers MFMER MFMER
9 Use of the Cascade Allows for efficient test ordering and significant cost savings Provider places an order for the algorithm Serum is drawn and sent to the laboratory ( send and hold ) The laboratory performs a cascade of tests based on the algorithm A la carte ordering is still available, but most clinicians choose to use the algorithm 2016 MFMER Cost Comparison Comparison of Testing Options Celiac Disease Serology Cascade: Weighted Average Price $ Total package if ordered a la carte $ Mayo Foundation for Medical Education and Research. All Rights Reserved MFMER
10 Laboratory-Driven Algorithms Why this works: Only serum is required Initial limited laboratory testing drives additional testing Hence, the Laboratory has control over the algorithm rather than the Clinician 2016 MFMER Can we turn this algorithm into a cascade? The most important question: can we get the information we need from our clinicians to guide testing? 2016 MFMER
11 Necessary information to determine if testing for intestinal parasites is even warranted 2016 MFMER From a doctor s office: Stool specimen for Giardia antigen testing 2016 MFMER
12 Form for patients to complete: Have been in car for 1 hr 40 min 2016 MFMER Challenges in implementing laboratorydriven cascades for intestinal parasites Initial AND sequential testing depends on clinical factors The laboratory has a difficult time getting this information Therefore, laboratory professionals have little control over testing Need to rely on the ordering clinician to use the algorithm 2016 MFMER
13 How do we get the ordering clinician to use the algorithm? 2016 MFMER FINAL SOLUTION: Use the Ordering System 2016 MFMER
14 Use of the Test Ordering System Provides an opportunity to influence the provider at the point of order, usually before a specimen is obtained from the patient This is the best place to intervene! You can use the ordering system to: Provide education Collect information Make it easy to do the right thing and hard to do the wrong thing 2016 MFMER Experience from our practice: We have noticed that tests at the top of the ordering screen get ordered more often than those at the bottom. Therefore, listing tests alphabetically may guide users to the first test they see (which might not be the correct one). A recommended approach: Put Common tests at the top of your order requisition or ordering screen. This is what we now do for all microbiology tests MFMER
15 Stool (Micro) Next Cancel Guide Comments Common Procedures C. difficile Toxin PCR Select Either Enteric Pathogens Culture OR PCR (not both) Bacterial Enteric Pathogens Culture, Stool 8098 ***Includes Shiga Toxin PCR Bacterial Enteric Pathogens PCR ***Rapid test for Salmonella, Shigella, Yersinia, Campylobacter, Shiga Toxin ***Reflexive culture performed if positive Fecal Leukocytes 8046 Giardia Ag Cryptosporidium Ag Rotavirus Ag 8886 Additional procedures Helicobacter pylori Ag Nocardia Stain Vibrio Culture VRE PCR ***For infection control purposes Acid Fast Smear for Mycobacterium 8213 M. tuberculosis Complex PCR Mycobacterial Culture 8205 Fungal Smear Fungal Culture, Routine Adenovirus PCR Viral Culture, Non Respiratory Cyclospora Stain Microsporidia Stain ***Microsporidia found primarily in immunocompromised patients Parasitic Examination 9216 ***Includes Fecal Leukocytes ***Recommend three collections, place new order for each 1 day 2 day 3 day Previous Order Screen Nothing to call these Out; no reference to algorithm Box around parasitic exam highlights this test Stool (Micro) Next Cancel Guide Comments Common Procedures C. difficile Toxin PCR Select Either Enteric Pathogens Culture OR PCR (not both) Bacterial Enteric Pathogens Culture, Stool 8098 ***Includes Shiga Toxin PCR Bacterial Enteric Pathogens PCR ***Rapid test for Salmonella, Shigella, Yersinia, Campylobacter, Shiga Toxin ***Reflexive culture performed if positive Fecal Leukocytes 8046 Fecal Parasite Testing for diarrhea (See Guide for Algorithm) Giardia Ag Cryptosporidium Ag Rotavirus Ag 8886 Additional procedures Helicobacter pylori Ag Nocardia Stain Vibrio Culture VRE PCR ***For infection control purposes Acid Fast Smear for Mycobacterium 8213 M. tuberculosis Complex PCR Mycobacterial Culture 8205 Fungal Smear Fungal Culture, Routine Adenovirus PCR Viral Culture, Non Respiratory Cyclospora Stain Microsporidia Stain ***Microsporidia found primarily in immunocompromised patients Parasitic Examination 9216 (order only if appropriate exposure history) ***See Guide for Algorithm ***Recommend three collections, place new order for each 1 day 2 day 3 day New Order Screen We ve seen a sustained 20% decrease in orders compared to previous years since implementing these changes 15
16 Conclusions Changing the order that tests appear is relatively simple method to achieve a modest change in ordering patterns. Consider: Have you seen your ordering screens? Do you know how your tests appear to your providers? How are your tests currently listed? Are the tests simply listed alphabetically or in a way designed to guide use? Can anyone order any test? 2016 MFMER Options for Ordering System Interventions Low Complexity Removing tests Changing test names Moderate Complexity Changing the lay-out of the order screens/forms Adding helper text High Complexity Set up IT rules (Dr. Donato s inpatient test rules) Require information to be entered at the time of ordering 2016 MFMER
17 EXAMPLE: Ordering urine bacterial culture on patients with indwelling Foley catheter A reason must be checked for this order to be placed or the physician can t place the order MFMER A twist to the Ova and Parasite story Implementation of multiplex PCR tests for bacteria, viruses and parasites 2016 MFMER
18 GI Pathogens Panel Simultaneously tests for 22 pathogens Multiplex PCR Testing is performed in 1 hour Includes testing for: Giardia duodenalis Cryptosporidium spp. Cyclospora cayetanensis Entamoeba histolytica Expensive! 2016 MFMER Special considerations Not all patients with diarrhea need testing How to guide provider ordering? Need to avoid duplicate testing for the same pathogens E.g. culture, EIA, individual PCR 2016 MFMER
19 How we accomplished this Created an algorithm with our ID partners Embedded the algorithm into the ordering system, along with helper text Embedded the algorithm into AskMayoExpert, a widely-used clinician support tool Created IT rules to prevent duplicate testing Also use manual cancellation in the lab for duplicates Education there s still a role! 2016 MFMER Laboratory Testing for Infectious Causes of Diarrhea Community-acquired diarrhea, <7 days duration WITHOUT warning signs or risk factors for severe disease 1,2 Community-acquired diarrhea 7 days duration OR Travel-related diarrhea OR Diarrhea with warning signs/risk factors for severe disease 1 Health care-associated diarrhea (onset after the 3 rd inpatient day) or patients with recent antibiotic use Testing not generally indicated If diarrhea persists: GIP / Gastrointestinal Pathogen Panel, PCR, Feces 3 Consider OAP / Parasitic Examination if traveler with >2 weeks of symptoms 4 NEGATIVE POSITIVE CDFRP / Clostridium difficile Toxin Molecular Detection, PCR, Feces NEGATIVE If diarrhea persists: No additional testing required unless clinical picture indicates If diarrhea persists: Consider: STL / Enteric Pathogens Culture, Stool GIAR / Giardia Antigen, Feces LCMSP / Microsporidia species, Molecular Detection, PCR (immunocompromised patients) OAP / Parasitic Examination 4 Use clinical judgment to guide the need for additional testing. 1 Warning signs and risk factors for severe disease include fever, bloody diarrhea, dysentery, severe abdominal pain, dehydration, hospitalization, and immunocompromised state. 2 During the summer, consider ordering STFRP / Shiga toxin, Molecular detection, PCR, Feces on children with diarrhea even if they don t have frankly bloody diarrhea, are not toxic-appearing, and diarrhea has been present <7 days. 3 GI Pathogen Panel tests for common bacterial, viral and parasitic cases of diarrhea 4 Submit 3 stool collected on separate days for maximum sensitivity Note: in outbreak scenarios with a known organism, consider ordering a specific test for that organism (CYCL / Cyclospora Stain, CRYPS / Cryptosporidium Antigen, Feces, GIAR / Giardia Antigen, Feces, bacterial stool culture) Mayo Foundation for Medical Education and Research (MFMER), All rights reserved. MAYO, Mayo Medical Laboratories and the triple-shield Mayo logo are trademarks and/or service marks of MFMER MFMER /
20 Laboratory Testing for Infectious Causes of Diarrhea Community-acquired diarrhea, <7 days duration WITHOUT warning signs or risk factors for severe diseases 1,2 Community-acquired diarrhea, <7 days duration WITHOUT warning signs or risk factors for severe disease 1,2 Testing not generally indicated 1 Warning signs and risk factors for severe disease include fever, bloody diarrhea, dysentery, severe abdominal pain, dehydration, hospitalization, and immunocompromised state. 2 During the summer, consider ordering STFRP / Shiga toxin, Molecular detection, PCR, Feces on children with diarrhea even if they don t have frankly bloody diarrhea, are not toxic-appearing, and diarrhea has been present <7 days MFMER Laboratory Testing for Infectious Causes of Diarrhea Community-acquired diarrhea, <7 days duration WITHOUT warning signs or risk factors for severe diseases 1,2 Community-acquired GIP diarrhea, / Gastrointestinal <7 days duration WITHOUT Pathogen warning Panel, signs or PCR, Feces 3 risk factors for severe Consider diseases OAP 1,2 / Parasitic If diarrhea persists: Examination if traveler with >2 weeks of symptoms 4 Testing not generally indicated 3 1 Warning GI Pathogen signs Panel and risk tests factors for common for severe bacterial, disease viral include and parasitic fever, bloody cases diarrhea, of dysentery, severe abdominal pain, 4 dehydration, Submit 3 stool hospitalization, collected on separate and immunocompromised days for maximum state. sensitivity 2 During the summer, consider ordering STFRP / Shiga toxin, Molecular detection, PCR, Feces on children with diarrhea even if they don t have frankly bloody diarrhea, are not toxic-appearing, and diarrhea has been present <7 days MFMER
21 Laboratory Testing for Infectious Causes of Diarrhea Community-acquired diarrhea, <7 days duration WITHOUT warning signs or risk factors for severe diseases 1,2 Community-acquired diarrhea 7 days duration OR Travel-related diarrhea OR Diarrhea with warning signs/risk factors for severe disease 1 Health care-associated diarrhea (onset after the 3 rd inpatient day) or patients with recent antibiotic use Testing not generally indicated If diarrhea persists: GIP / Gastrointestinal Pathogen Panel, PCR, Feces 3 Consider OAP / Parasitic Examination if traveler with >2 weeks of symptoms 4 NEGATIVE POSITIVE CDFRP / Clostridium difficile Toxin Molecular Detection, PCR, Feces NEGATIVE If diarrhea persists: No additional testing required unless clinical picture indicates If diarrhea persists: Consider: STL / Enteric Pathogens Culture, Stool GIAR / Giardia Antigen, Feces LCMSP / Microsporidia species, Molecular Detection, PCR (immunocompromised patients) OAP / Parasitic Examination 4 Use clinical judgment to guide the need for additional testing. 1 Warning signs and risk factors for severe disease include fever, bloody diarrhea, dysentery, severe abdominal pain, dehydration, hospitalization, and immunocompromised state. 2 During the summer, consider ordering STFRP / Shiga toxin, Molecular detection, PCR, Feces on children with diarrhea even if they don t have frankly bloody diarrhea, are not toxic-appearing, and diarrhea has been present <7 days. 3 GI Pathogen Panel tests for common bacterial, viral and parasitic cases of diarrhea 4 Submit 3 stool collected on separate days for maximum sensitivity Note: in outbreak scenarios with a known organism, consider ordering a specific test for that organism (CYCL / Cyclospora Stain, CRYPS / Cryptosporidium Antigen, Feces, GIAR / Giardia Antigen, Feces, bacterial stool culture) Mayo Foundation for Medical Education and Research (MFMER), All rights reserved. MAYO, Mayo Medical Laboratories and the triple-shield Mayo logo are trademarks and/or service marks of MFMER MFMER /2015 Laboratory Testing for Infectious Causes of Diarrhea Community-acquired diarrhea 7 days duration OR Travel-related diarrhea OR Diarrhea with warning signs/risk factors for severe disease 1 Community-acquired diarrhea 7 days duration OR Travel-related diarrhea OR Diarrhea with warning signs/risk factors for severe disease 1 GIP / Gastrointestinal Pathogen Panel, PCR, Feces 3 Consider OAP / Parasitic Examination if traveler with >2 weeks of symptoms 4 1 Warning signs and risk factors for severe disease include fever, bloody diarrhea, dysentery, severe abdominal pain, dehydration, hospitalization, and immunocompromised state. 3 GI Pathogen Panel tests for common bacterial, viral and parasitic cases of diarrhea 4 Submit 3 stool collected on separate days for maximum sensitivity 2016 MFMER
22 Laboratory Testing for Infectious Causes of Diarrhea Community-acquired diarrhea 7 days duration OR Travel-related diarrhea OR Diarrhea with warning signs/risk factors for severe disease 1 Community-acquired diarrhea 7 days duration OR NEGATIVE POSITIVE Travel-related diarrhea OR If diarrhea persists: Diarrhea with warning signs/risk factors for severe disease 1 No additional testing required unless clinical picture indicates Consider: STL / Enteric Pathogens Culture, Stool GIAR / Giardia Antigen, Feces LCMSP >2 / weeks Microsporidia of symptoms species, Molecular 4 Detection, PCR (immunocompromised patients) OAP / Parasitic Examination 4 GIP / Gastrointestinal Pathogen Panel, PCR, Feces 3 Consider OAP / Parasitic Examination if traveler with 3 GI Pathogen Panel tests for common bacterial, viral and parasitic cases of diarrhea 4 Submit 3 stool collected on separate days for maximum sensitivity 2016 MFMER Laboratory Testing for Infectious Causes of Diarrhea Community-acquired diarrhea, <7 days duration WITHOUT warning signs or risk factors for severe diseases 1,2 Community-acquired diarrhea 7 days duration OR Travel-related diarrhea OR Diarrhea with warning signs/risk factors for severe disease 1 Health care-associated diarrhea (onset after the 3 rd inpatient day) or patients with recent antibiotic use Testing not generally indicated If diarrhea persists: GIP / Gastrointestinal Pathogen Panel, PCR, Feces 3 Consider OAP / Parasitic Examination if traveler with >2 weeks of symptoms 4 NEGATIVE POSITIVE CDFRP / Clostridium difficile Toxin Molecular Detection, PCR, Feces NEGATIVE If diarrhea persists: No additional testing required unless clinical picture indicates If diarrhea persists: Consider: STL / Enteric Pathogens Culture, Stool GIAR / Giardia Antigen, Feces LCMSP / Microsporidia species, Molecular Detection, PCR (immunocompromised patients) OAP / Parasitic Examination 4 Use clinical judgment to guide the need for additional testing. 1 Warning signs and risk factors for severe disease include fever, bloody diarrhea, dysentery, severe abdominal pain, dehydration, hospitalization, and immunocompromised state. 2 During the summer, consider ordering STFRP / Shiga toxin, Molecular detection, PCR, Feces on children with diarrhea even if they don t have frankly bloody diarrhea, are not toxic-appearing, and diarrhea has been present <7 days. 3 GI Pathogen Panel tests for common bacterial, viral and parasitic cases of diarrhea 4 Submit 3 stool collected on separate days for maximum sensitivity Note: in outbreak scenarios with a known organism, consider ordering a specific test for that organism (CYCL / Cyclospora Stain, CRYPS / Cryptosporidium Antigen, Feces, GIAR / Giardia Antigen, Feces, bacterial stool culture) Mayo Foundation for Medical Education and Research (MFMER), All rights reserved. MAYO, Mayo Medical Laboratories and the triple-shield Mayo logo are trademarks and/or service marks of MFMER MFMER /
23 Laboratory Testing for Infectious Causes of Diarrhea Health care-associated diarrhea (onset after the 3 rd inpatient day) or patients with recent antibiotic use Health care-associated diarrhea (onset after the 3 rd inpatient day) or patients with recent antibiotic use CDFRP / Clostridium difficile Toxin Molecular Detection, PCR, Feces 2016 MFMER Laboratory Testing for Infectious Causes of Diarrhea Health care-associated diarrhea (onset after the 3 rd inpatient day) or patients with recent antibiotic use Health care-associated diarrhea (onset POSITIVE after the 3 rd NEGATIVE inpatient day) or patients with recent antibiotic use No additional testing required unless clinical picture indicates If diarrhea persists: CDFRP / Clostridium difficile Toxin Molecular Detection, PCR, Feces Use clinical judgment to guide the need for additional testing MFMER
24 Laboratory Testing for Infectious Causes of Diarrhea Community-acquired diarrhea, <7 days duration WITHOUT warning signs or risk factors for severe diseases 1,2 Community-acquired diarrhea 7 days duration OR Travel-related diarrhea OR Diarrhea with warning signs/risk factors for severe disease 1 Health care-associated diarrhea (onset after the 3 rd inpatient day) or patients with recent antibiotic use Note: in outbreak scenarios with a known organism, Testing not generally indicated GIP / Gastrointestinal Pathogen Panel, PCR, Feces consider ordering a specific test for that organism 3 CDFRP / Clostridium difficile Toxin Consider OAP / Parasitic Examination if traveler with Molecular Detection, PCR, Feces >2 weeks of symptoms If diarrhea persists: (CYCL / Cyclospora Stain, 4 CRYPS / Cryptosporidium NEGATIVE POSITIVE NEGATIVE Antigen, Feces, GIAR / Giardia Antigen, Feces, If diarrhea persists: No additional testing required If diarrhea persists: unless clinical picture indicates bacterial stool culture) Consider: STL / Enteric Pathogens Culture, Stool GIAR / Giardia Antigen, Feces LCMSP / Microsporidia species, Molecular Detection, PCR (immunocompromised patients) OAP / Parasitic Examination 4 Use clinical judgment to guide the need for additional testing. 1 Warning signs and risk factors for severe disease include fever, bloody diarrhea, dysentery, severe abdominal pain, dehydration, hospitalization, and immunocompromised state. 2 During the summer, consider ordering STFRP / Shiga toxin, Molecular detection, PCR, Feces on children with diarrhea even if they don t have frankly bloody diarrhea, are not toxic-appearing, and diarrhea has been present <7 days. 3 GI Pathogen Panel tests for common bacterial, viral and parasitic cases of diarrhea 4 Submit 3 stool collected on separate days for maximum sensitivity Note: in outbreak scenarios with a known organism, consider ordering a specific test for that organism (CYCL / Cyclospora Stain, CRYPS / Cryptosporidium Antigen, Feces, GIAR / Giardia Antigen, Feces, bacterial stool culture) Mayo Foundation for Medical Education and Research (MFMER), All rights reserved. MAYO, Mayo Medical Laboratories and the triple-shield Mayo logo are trademarks and/or service marks of MFMER MFMER /2015 Test volumes since implementation GI Panel implemented on October 12, 2015 Stopped flipping testing for the following EIAs at the end of March Tests performed Antigen Volumes Use your DATA! CRYPS GIAR ROTA 2016 MFMER
25 Additional Approaches (NOT STOOL!) 2016 MFMER Use Residents and Fellows to control use of expensive molecular multiplex platforms Multiplex Respiratory PCR panel: Lab staff first confirm the medical service that ordered the test is one of the approved services Providers who are NOT on the approved list will receive a call from the Clinical Microbiology fellow Then a discussion about testing ensues, based on a script that we created 2016 MFMER
26 Script for ordering intervention The Clinical Microbiology lab received a nasopharyngeal swab collected from your patient, <patient name>, that included an order for the Multiplex Respiratory Pathogen PCR Panel. I just wanted to provide some information before we proceed with testing. First, I wanted to make sure you were aware that this test is very expensive and also tests for a number of viral pathogens for which there is no treatment available. Can you tell me a little bit about the patient s clinical presentation so that we can determine whether this is the right test? Based on the patient s clinical presentation, there may be some molecular alternative tests that may be more appropriate and cost effective (e.g. Influenza A/B PCR, RSV PCR) 2016 MFMER Whatever approaches you use, be sure to capture your data Share data with lab and institutional leadership and attach dollar amounts (money saved, potential for savings) This is an excellent approach for getting the support you need to start and/or continue utilization efforts Gather pre- and post-intervention data You may find out that your intervention isn t working (time to try something new) Share ordering data with your clinicians This can be very powerful for identifying outliers clinicians who order far more tests than the others and getting them to change their practices 2016 MFMER
27 Summary There are many different approaches for tackling test utilization issues some with more impact than others. It is important to identify and partner with clinicianchampions Create and implement algorithms based on data and best practice The test ordering system can be a powerful tool to guide test usage. Don t aim for perfection any progress is good! Monitor your results and share successes with leadership 2016 MFMER Thank you! 2016 MFMER
28 Test Utilization Strategies in Anatomic Pathology Joaquin J. Garcia, MD Vice Chair of Laboratories, Anatomic Pathology Mayo Clinic Rochester 2016 MFMER Disclosures Relevant financial relationship(s) with industry: None MFMER
29 Laboratory Medicine & Pathology Cytogenetics Microbiology Biochemistry Anatomic Pathology Transfusion Hematopathology Molecular Genetics 2016 MFMER Pathology Anatomic Pathology 2016 MFMER
30 Laboratory Medicine: Chain of Events Laboratory Requisition Interpretation 2016 MFMER Laboratory Medicine: Chain of Command Clinician Pathologist 2016 MFMER
31 Anatomic Pathology: Chain of Events Laboratory Laboratory Requisition Laboratory Interpretation 2016 MFMER Anatomic Pathology: Chain of Command Clinician Trainee Technician Pathologist 2016 MFMER
32 Anatomic Pathology: Chain of Command 2016 MFMER Anatomic Pathology: Behavior Modifiers Experience Training Preference Confusion Fear 2016 MFMER
33 Tissue Screening Tissue Submission Immunohistochemistry Education 2016 MFMER Education 5% 2016 MFMER
34 Education 16,000 Slides 2016 MFMER Education 4,000 Slides 2016 MFMER
35 Immunohistochemistry 2016 MFMER Immunohistochemistry 300,000 Slides 2016 MFMER
36 Immunohistochemistry 100,000 Slides 2016 MFMER Tissue Submission 2016 MFMER
37 Surgical Margin Assessment Tissue Submission 2016 MFMER MFMER
38 2016 MFMER MFMER
39 2016 MFMER MFMER
40 2016 MFMER Tangential Method Perpendicular Method 2016 MFMER
41 2016 MFMER Tangential Method Perpendicular Method 2016 MFMER
42 2016 MFMER BCS 18 mos 414 Margin Assess 259 Positive Margin 2016 MFMER
43 Perpendicular Tangential 2016 MFMER Surgical Margin Assessment Tissue Submission 3-fold Slide Volume Decrease 2016 MFMER
44 Tissue Screening 2016 MFMER Genetic Testing Approval Process Tissue Screening 2016 MFMER
45 Cytogenetics Microbiology Biochemistry Anatomic Pathology Transfusion Hematopathology Molecular Genetics 2016 MFMER Cytogenetics Microbiology Anatomic Pathology Hematopathology Molecular Genetics 2016 MFMER
46 H&E Mucicarmine p MFMER H&E Mucicarmine p MFMER
47 H&E p63 FISH 2016 MFMER Cytogenetics Anatomic Pathology Molecular Genetics 2016 MFMER
48 100 Cases/Day 5 Cancelled 5 Modified Tissue Screening 2016 MFMER Tissue Screening Tissue Submission Immunohistochemistry Education 2016 MFMER
49 Question Answer Discuss 2016 MFMER
The Cost-effectiveness of a GI PCR panel in Detecting Necessary to Treat Infections
 The Cost-effectiveness of a GI PCR panel in Detecting Necessary to Treat Infections Annie L. Andrews MD, MSCR Annie N. Simpson PhD Kit N. Simpson DrPH Daniel C. Williams MD, MSCR The authors have nothing
The Cost-effectiveness of a GI PCR panel in Detecting Necessary to Treat Infections Annie L. Andrews MD, MSCR Annie N. Simpson PhD Kit N. Simpson DrPH Daniel C. Williams MD, MSCR The authors have nothing
Dear Healthcare Provider, The information contained here may be very important to your practice. Please take a moment to review this document.
 February 2018 Dear Healthcare Provider, The information contained here may be very important to your practice. Please take a moment to review this document. TEST BULLETIN CHLAMYDIA/GONORRHEA SPECIMEN COLLECTION
February 2018 Dear Healthcare Provider, The information contained here may be very important to your practice. Please take a moment to review this document. TEST BULLETIN CHLAMYDIA/GONORRHEA SPECIMEN COLLECTION
Advances in Gastrointestinal Pathogen Detection
 Advances in Gastrointestinal Pathogen Detection Erin McElvania TeKippe, Ph.D., D(ABMM) Director of Clinical Microbiology Children s Health System, Assistant Professor of Pathology and Pediatrics UT Southwestern
Advances in Gastrointestinal Pathogen Detection Erin McElvania TeKippe, Ph.D., D(ABMM) Director of Clinical Microbiology Children s Health System, Assistant Professor of Pathology and Pediatrics UT Southwestern
MICROBIOLOGY SPECIMEN COLLECTION MANUAL
 Lee Memorial Health System Lee County, FL CLINICAL LABORATORY MICROBIOLOGY SPECIMEN COLLECTION MANUAL ACID FAST CULTURE Specimen Type see Specimen Chart ACID FAST STAIN see Specimen Chart Acid Fast stain
Lee Memorial Health System Lee County, FL CLINICAL LABORATORY MICROBIOLOGY SPECIMEN COLLECTION MANUAL ACID FAST CULTURE Specimen Type see Specimen Chart ACID FAST STAIN see Specimen Chart Acid Fast stain
Collection (Specimen Source Required on all tests) Sputum: >5 ml required. First morning specimen preferred.
 Type Acid Fast (Mycobacteria) Sputum: >5 ml required. First morning specimen preferred. For blood, sodium heparin tube preferred. Lithium heparin acceptable. Do not centrifuge.. delay. Swabs are not appropriate
Type Acid Fast (Mycobacteria) Sputum: >5 ml required. First morning specimen preferred. For blood, sodium heparin tube preferred. Lithium heparin acceptable. Do not centrifuge.. delay. Swabs are not appropriate
DISCLOSURE Relevant relationships with commercial entities Wyeth (received advisory board & speaker honoraria) Potential for conflicts of interest wit
 GASTROENTERITIS DISCLOSURE Relevant relationships with commercial entities Wyeth (received advisory board & speaker honoraria) Potential for conflicts of interest within this presentation fidaxomicin (which
GASTROENTERITIS DISCLOSURE Relevant relationships with commercial entities Wyeth (received advisory board & speaker honoraria) Potential for conflicts of interest within this presentation fidaxomicin (which
33. I will recommend this primer to my colleagues. A. Strongly Agree D. Disagree B. Agree E. Strongly Disagree C. Neither agree nor disagree
 27. The primer increased my ability to recognize foodborne illnesses and increased the likelihood that I will consider such illnesses in my patients. 28. The primer increased my knowledge and skills in
27. The primer increased my ability to recognize foodborne illnesses and increased the likelihood that I will consider such illnesses in my patients. 28. The primer increased my knowledge and skills in
Virtual Lectures Planning Committee Disclosure Summary
 Mayo Medical Laboratories Virtual Lectures 2014 MFMER MFMER Virtual Lectures Planning Committee Disclosure Summary As a provider accredited by ACCME, College of Medicine, Mayo Clinic (Mayo School of CPD)
Mayo Medical Laboratories Virtual Lectures 2014 MFMER MFMER Virtual Lectures Planning Committee Disclosure Summary As a provider accredited by ACCME, College of Medicine, Mayo Clinic (Mayo School of CPD)
Infectious Disease Diagnostics in the Geisinger Health System
 10X Essentials Infectious Disease Diagnostics in the Geisinger Health System SUMMARY Choose the more sensitive Giardia and Cryptosporidium EIA (GEIA and CRYIA) on most Epic order sets. OAP (Ova and Parasite)
10X Essentials Infectious Disease Diagnostics in the Geisinger Health System SUMMARY Choose the more sensitive Giardia and Cryptosporidium EIA (GEIA and CRYIA) on most Epic order sets. OAP (Ova and Parasite)
Foodborne Disease in the Region of Peel
 Foodborne Disease in the Region of Peel HIGHLIGHTS The incidence of selected foodborne diseases was generally higher in Peel than in Ontario between 1993 and 22. A higher incidence was observed in Peel
Foodborne Disease in the Region of Peel HIGHLIGHTS The incidence of selected foodborne diseases was generally higher in Peel than in Ontario between 1993 and 22. A higher incidence was observed in Peel
PAMET Continuing Education 2016
 PAMET Continuing Education 2016 Agent of gastroenteritis Medium/method] used for routine screening/detection in stool samples Salmonella, Shigella, MacConkey, Hektoen, Bismuth sulfite,etc. Plesiomonas
PAMET Continuing Education 2016 Agent of gastroenteritis Medium/method] used for routine screening/detection in stool samples Salmonella, Shigella, MacConkey, Hektoen, Bismuth sulfite,etc. Plesiomonas
Clinical Microbiology CLS 2019 Clinical Microbiology Laboratory Scheme Application Form
 Laboratory Scheme Application Form complete all sections below and return to LGC Standards Proficiency Testing by email, fax or post. Returning customer Lab ID: Purchase order no.: (compulsory) Round Despatch
Laboratory Scheme Application Form complete all sections below and return to LGC Standards Proficiency Testing by email, fax or post. Returning customer Lab ID: Purchase order no.: (compulsory) Round Despatch
IDSA Diarrhea Guidelines. Larry Pickering, MD, FAAP, FIDSA, FPIDS
 IDSA Diarrhea Guidelines Larry Pickering, MD, FAAP, FIDSA, FPIDS Title Sub-caption Infectious Diseases Society of America Strategic Priorities Establishing state of the art clinical guidelines Advocating
IDSA Diarrhea Guidelines Larry Pickering, MD, FAAP, FIDSA, FPIDS Title Sub-caption Infectious Diseases Society of America Strategic Priorities Establishing state of the art clinical guidelines Advocating
Attendees will understand the early experience and clinical impact of GI multiplex PCR diagnostics in children
 Participants will understand the role of a comprehensive business case in considering the introduction of novel technology affecting multiple areas of the laboratory Attendees will learn about the impact
Participants will understand the role of a comprehensive business case in considering the introduction of novel technology affecting multiple areas of the laboratory Attendees will learn about the impact
Bacterial Enteropathogens Faecal PCR and Culture Results
 Bacterial Enteropathogens - 2019 Faecal PCR and Culture Results Because most diarrhoeal illness is self-limiting, microbiological investigation is usually not indicated. It should however be considered
Bacterial Enteropathogens - 2019 Faecal PCR and Culture Results Because most diarrhoeal illness is self-limiting, microbiological investigation is usually not indicated. It should however be considered
Stool Collection Guidelines
 Stool Collection Guidelines Your child s stool (bowel movement) must be tested so we can plan the treatment for your child. You will need to collect the stool specimen at home. Bring it to a Laboratory
Stool Collection Guidelines Your child s stool (bowel movement) must be tested so we can plan the treatment for your child. You will need to collect the stool specimen at home. Bring it to a Laboratory
PATHOGEN DETECTION WITH THE FILMARRAY
 PATHOGEN DETECTION WITH THE FILMARRAY The System Sample-to-Answer in an Hour Single sample Multiple samples The FilmArray integrates sample preparation, amplification, detection, and analysis all into
PATHOGEN DETECTION WITH THE FILMARRAY The System Sample-to-Answer in an Hour Single sample Multiple samples The FilmArray integrates sample preparation, amplification, detection, and analysis all into
3base The Science Behind EasyScreen Molecular Diagnostic Assays. Nucleic Acid Conversion Technology
 3base The Science Behind EasyScreen Molecular Diagnostic Assays Nucleic Acid Conversion Technology 1 Topics 3base Technology Overview Technical Advantages of 3base Validation of 3base Specificity Validation
3base The Science Behind EasyScreen Molecular Diagnostic Assays Nucleic Acid Conversion Technology 1 Topics 3base Technology Overview Technical Advantages of 3base Validation of 3base Specificity Validation
IP Lab Webinar 8/23/2012
 2 What Infection Preventionists need to know about the Laboratory Anne Maher, MS, M(ASCP), CIC Richard VanEnk PhD, CIC 1 Objectives Describe what the laboratory can do for you; common laboratory tests
2 What Infection Preventionists need to know about the Laboratory Anne Maher, MS, M(ASCP), CIC Richard VanEnk PhD, CIC 1 Objectives Describe what the laboratory can do for you; common laboratory tests
The Changing Paradigm of the Laboratory Diagnosis of Gastroenteritis
 Disclosures The Changing Paradigm of the Laboratory Diagnosis of Gastroenteritis Melissa B. Miller, PhD, D(ABMM) Professor, Pathology and Laboratory Medicine UNC School of Medicine Director, Clinical Molecular
Disclosures The Changing Paradigm of the Laboratory Diagnosis of Gastroenteritis Melissa B. Miller, PhD, D(ABMM) Professor, Pathology and Laboratory Medicine UNC School of Medicine Director, Clinical Molecular
CLIENT NOTICE UPDATE - August 24, *** NEW COLLECTION CONTAINTER for PARASITE BY PCR ***
 CLIENT NOTICE UPDATE - August 24, 2017 LifeLabs Introduces PCR Testing For Detection Of Parasites In Stool Samples On August 8 th, 2017, LifeLabs Medical Laboratories has started detection of parasites
CLIENT NOTICE UPDATE - August 24, 2017 LifeLabs Introduces PCR Testing For Detection Of Parasites In Stool Samples On August 8 th, 2017, LifeLabs Medical Laboratories has started detection of parasites
Laboratory Bulletin...
 Laboratory Bulletin... Updates and Information from Rex Healthcare and Rex Outreach August/September 1998 Issue Number 34 Change in Glycohemoglobin Reporting Glycohemoglobin determination is valuable in
Laboratory Bulletin... Updates and Information from Rex Healthcare and Rex Outreach August/September 1998 Issue Number 34 Change in Glycohemoglobin Reporting Glycohemoglobin determination is valuable in
New Molecular Diagnostic Assays for Solana Performance Assessment, Workflow Analysis, and Clinical Utility
 New Molecular Diagnostic Assays for Solana Performance Assessment, Workflow Analysis, and Clinical Utility Gerald A. Capraro, Ph.D., D(ABMM) Director, Clinical Microbiology Carolinas Pathology Group Atrium
New Molecular Diagnostic Assays for Solana Performance Assessment, Workflow Analysis, and Clinical Utility Gerald A. Capraro, Ph.D., D(ABMM) Director, Clinical Microbiology Carolinas Pathology Group Atrium
Immunohistochemical Confirmation of Infections
 Immunohistochemical Confirmation of Infections Danny A. Milner, Jr, MD, MSc, FCAP The Brigham and Women s Hospital Harvard Medical School Boston, Masschusetts USA Judicious Use of Immunohistochemistry
Immunohistochemical Confirmation of Infections Danny A. Milner, Jr, MD, MSc, FCAP The Brigham and Women s Hospital Harvard Medical School Boston, Masschusetts USA Judicious Use of Immunohistochemistry
Fecal Me This, Fecal Me That Where Are We Now With Giardia and Cryptosporidium Diagnostics?
 Fecal Me This, Fecal Me That Where Are We Now With Giardia and Cryptosporidium Diagnostics? Shelley Miller, PhD, D(ABMM) Clinical Instructor Dept of Pathology & Lab Medicine UCLA Objectives Discuss giardiasis
Fecal Me This, Fecal Me That Where Are We Now With Giardia and Cryptosporidium Diagnostics? Shelley Miller, PhD, D(ABMM) Clinical Instructor Dept of Pathology & Lab Medicine UCLA Objectives Discuss giardiasis
Gastrointestinal Pathogen Panel Guidance Authors: Trevor Van Schooneveld, MD, Kiri Rolek, PharmD, BCPS, Paul Fey PhD, Mark Rupp, MD
 Gastrointestinal Pathogen Panel Guidance Authors: Trevor Van Schooneveld, MD, Kiri Rolek, PharmD, BCPS, Paul Fey PhD, Mark Rupp, MD Background: Many pathogens, including bacteria, parasites, and viruses
Gastrointestinal Pathogen Panel Guidance Authors: Trevor Van Schooneveld, MD, Kiri Rolek, PharmD, BCPS, Paul Fey PhD, Mark Rupp, MD Background: Many pathogens, including bacteria, parasites, and viruses
Hompes Method. Practitioner Training Level II. Lesson Seven Part A DRG Pathogen Plus Interpretation
 Hompes Method Practitioner Training Level II Lesson Seven Part A DRG Pathogen Plus Interpretation Health for the People Ltd not for reuse without expressed permission Hompes Method is a trading name of
Hompes Method Practitioner Training Level II Lesson Seven Part A DRG Pathogen Plus Interpretation Health for the People Ltd not for reuse without expressed permission Hompes Method is a trading name of
Microbiology EQA Product Portfolio
 Labquality EQAS Microbiology EQA Product Portfolio Clinically relevant external quality assessment program for microbiology Bacterial serology Bacteriology Mycology Parasitology Preanalytics Virology Labquality
Labquality EQAS Microbiology EQA Product Portfolio Clinically relevant external quality assessment program for microbiology Bacterial serology Bacteriology Mycology Parasitology Preanalytics Virology Labquality
MICROBIOLOGICAL TESTING IN PICU
 MICROBIOLOGICAL TESTING IN PICU This is a guideline for the taking of microbiological samples in PICU to diagnose or exclude infection. The diagnosis of infection requires: Ruling out non-infectious causes
MICROBIOLOGICAL TESTING IN PICU This is a guideline for the taking of microbiological samples in PICU to diagnose or exclude infection. The diagnosis of infection requires: Ruling out non-infectious causes
Ever wonder what s really happening on the inside?
 For Practitioners Ever wonder what s really happening on the inside? Are your patients suffering from diarrhea, constipation, bloating, gas or indigestion? Rocky Mountain Analytical is now offering Gut-Well
For Practitioners Ever wonder what s really happening on the inside? Are your patients suffering from diarrhea, constipation, bloating, gas or indigestion? Rocky Mountain Analytical is now offering Gut-Well
The Netherlands Donor Feces Bank a. it takes stool to get better
 The Netherlands Donor Feces Bank a it takes stool to get better H.W. Verspaget Dept. of Biobanking and Dept. of Gastroenterology Leiden University Medical Center The Netherlands Fecal MicrobiotaTransplantation
The Netherlands Donor Feces Bank a it takes stool to get better H.W. Verspaget Dept. of Biobanking and Dept. of Gastroenterology Leiden University Medical Center The Netherlands Fecal MicrobiotaTransplantation
FOOD BORNE INFECTIONS
 Food Poisoning Food poisoning Advisory commitee on Microbiological safety of food (ACMSF, UK) has defined food poisoning as : An acute illness with gastrointestinal or neurological manifestation affecting
Food Poisoning Food poisoning Advisory commitee on Microbiological safety of food (ACMSF, UK) has defined food poisoning as : An acute illness with gastrointestinal or neurological manifestation affecting
Test Requested Specimen Ordering Recommendations
 Microbiology Essentials Culture and Sensitivity (C&S) Urine C&S Catheter Surgical (excluding kidney aspirates) Voided Requisition requirements o Specific method of collection MUST be indicated o Indicate
Microbiology Essentials Culture and Sensitivity (C&S) Urine C&S Catheter Surgical (excluding kidney aspirates) Voided Requisition requirements o Specific method of collection MUST be indicated o Indicate
Acute Gastroenteritis
 Acute Gastroenteritis Definitions Epidemiology Clinical presentation Diagnostics Therapy Erdélyi, Dániel 2018.04.10. Definitions Acute gastroenteritis (AGE): Loose or liquid stools 3 times daily (better:
Acute Gastroenteritis Definitions Epidemiology Clinical presentation Diagnostics Therapy Erdélyi, Dániel 2018.04.10. Definitions Acute gastroenteritis (AGE): Loose or liquid stools 3 times daily (better:
DEPARTMENT OF MICROBIOLOGY IMPORTANT NOTICE TO USERS Turnaround Times (TATs) for Microbiology Investigations
 Dear User, ISSUE: M008 DEPARTMENT OF MICROBIOLOGY IMPORTANT NOTICE TO USERS Turnaround Times (TATs) for Microbiology Investigations In order to comply with national quality guidance and as part of our
Dear User, ISSUE: M008 DEPARTMENT OF MICROBIOLOGY IMPORTANT NOTICE TO USERS Turnaround Times (TATs) for Microbiology Investigations In order to comply with national quality guidance and as part of our
Gastrointestinal Disease from 2007 to 2014
 Data Requested by Amber Erickson, Epidemiologist, North Central Health District Gastrointestinal Disease from 2007 to 2014 North Central Health District Aemon Weaver, Epidemiology Intern, NCHD September
Data Requested by Amber Erickson, Epidemiologist, North Central Health District Gastrointestinal Disease from 2007 to 2014 North Central Health District Aemon Weaver, Epidemiology Intern, NCHD September
OpenBiome Quality Metrics
 OpenBiome Quality Metrics Donor Assessment Stool Collection & Production Controls Quality Assurance Purpose This section summarizes the assays and process controls that OpenBiome has developed to ensure
OpenBiome Quality Metrics Donor Assessment Stool Collection & Production Controls Quality Assurance Purpose This section summarizes the assays and process controls that OpenBiome has developed to ensure
Potential Reimbursement CPT Codes
 BioFire FilmArray Blood Culture Identification (BCID) Panel Medicare All targets (n) 87150 n x * *BioFire BCID Panel is comprised of 27 total targets. The number of targets allowed for reimbursement may
BioFire FilmArray Blood Culture Identification (BCID) Panel Medicare All targets (n) 87150 n x * *BioFire BCID Panel is comprised of 27 total targets. The number of targets allowed for reimbursement may
Laboratory Investigation of. Infectious Diarrhoea. Quiz Feedback. bpac nz better medicine
 Laboratory Investigation of Infectious Diarrhoea Quiz Feedback bpac nz better medicine GP Review Panel: Dr Janine Bailey, Motueka Dr Stephen Kuzmich, Wellington Dr Randall Sturm, Auckland Dr Neil Whittaker,
Laboratory Investigation of Infectious Diarrhoea Quiz Feedback bpac nz better medicine GP Review Panel: Dr Janine Bailey, Motueka Dr Stephen Kuzmich, Wellington Dr Randall Sturm, Auckland Dr Neil Whittaker,
SHIGA-TOXIN PRODUCING ESCHERICHIA COLI STEC Update. Roshan Reporter, MD, MPH Rita Bagby, PS-PHN Leticia Martinez, PS-PHN
 SHIGA-TOXIN PRODUCING ESCHERICHIA COLI STEC Update Roshan Reporter, MD, MPH Rita Bagby, PS-PHN Leticia Martinez, PS-PHN Objectives At the conclusion of this presentation the participant should be able
SHIGA-TOXIN PRODUCING ESCHERICHIA COLI STEC Update Roshan Reporter, MD, MPH Rita Bagby, PS-PHN Leticia Martinez, PS-PHN Objectives At the conclusion of this presentation the participant should be able
BILLING RATES - FY 17/18
 Meat-Poultry Inspection Department Cross Utilization "Regular Time" per hour $22.93 USDA Meat Inspection Cross Utilization "Over Time Rate" per hour $25.40 USDA Meat Inspection Overtime Rates per hour
Meat-Poultry Inspection Department Cross Utilization "Regular Time" per hour $22.93 USDA Meat Inspection Cross Utilization "Over Time Rate" per hour $25.40 USDA Meat Inspection Overtime Rates per hour
REVISED ESTIMATE OF FOOD-BORNE ILLNESS IN CANADA
 REVISED ESTIMATE OF FOOD-BORNE ILLNESS IN CANADA M. Kate Thomas, Regan Murray, Logan Flockhart, Frank Pollari, Aamir Fazil, Katarina Pintar, Andrea Nesbitt, Barbara Marshall BioM&S May 2, 2014 Outline
REVISED ESTIMATE OF FOOD-BORNE ILLNESS IN CANADA M. Kate Thomas, Regan Murray, Logan Flockhart, Frank Pollari, Aamir Fazil, Katarina Pintar, Andrea Nesbitt, Barbara Marshall BioM&S May 2, 2014 Outline
PARASITOLOGY CASE HISTORY 15 (HISTOLOGY) (Lynne S. Garcia)
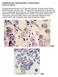 PARASITOLOGY CASE HISTORY 15 (HISTOLOGY) (Lynne S. Garcia) A biopsy was performed on a 27-year-old man with no known travel history, presenting with a perianal ulcer. The specimen was preserved in formalin
PARASITOLOGY CASE HISTORY 15 (HISTOLOGY) (Lynne S. Garcia) A biopsy was performed on a 27-year-old man with no known travel history, presenting with a perianal ulcer. The specimen was preserved in formalin
The Value and Complexity of Infection Studies or Why we Bug the Data Managers.
 The Value and Complexity of Infection Studies or Why we Bug the Data Managers. Marcie Tomblyn, MD, MS, Scientific Director Manisha Kukreja, MBBS, MPH, Biostatistician Infection and Immune Reconstitution
The Value and Complexity of Infection Studies or Why we Bug the Data Managers. Marcie Tomblyn, MD, MS, Scientific Director Manisha Kukreja, MBBS, MPH, Biostatistician Infection and Immune Reconstitution
Rapid-VIDITEST Enterovirus
 Rapid-VIDITEST Enterovirus A rapid one step Enterovirus Card test for the qualitative detection of Enterovirus antigens in human feces. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365,
Rapid-VIDITEST Enterovirus A rapid one step Enterovirus Card test for the qualitative detection of Enterovirus antigens in human feces. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365,
INFECTIOUS DISEASE. Page 2
 Infectious disease Advantages OF TESTING INFECTIOUS DISEASE We are in the middle of a paradigm shift in infectious disease diagnostic testing. As we move from targeted infectious disease testing to a syndromic
Infectious disease Advantages OF TESTING INFECTIOUS DISEASE We are in the middle of a paradigm shift in infectious disease diagnostic testing. As we move from targeted infectious disease testing to a syndromic
35.0. ug/ml Trough. 8.0 Carbamazepine 15.0 ug/ml Digoxin 2.5 ng/ml Gentamicin Peak Random Trough
 The following are a list by department of Critical (Panic) Values: CHEMISTRY TEST LESS THAN GREATER THAN UNITS Glucose 0-7days 40 200 mg/dl Glucose > 7 days 50 400 mg/dl Glucose, CSF 25 mg/dl Sodium 121
The following are a list by department of Critical (Panic) Values: CHEMISTRY TEST LESS THAN GREATER THAN UNITS Glucose 0-7days 40 200 mg/dl Glucose > 7 days 50 400 mg/dl Glucose, CSF 25 mg/dl Sodium 121
American Association of Bioanalysts 5615 Kirby Drive, Suite 870 Houston, TX
 Q3 2018 Parasitology American Association of Bioanalysts 5615 Kirby Drive, Suite 870 Houston, TX 77005 800-234-5315 281-436-5357 Specimen 1 Referees Extent 1 Extent 2 Total Few to 534 Giardia lamblia Many
Q3 2018 Parasitology American Association of Bioanalysts 5615 Kirby Drive, Suite 870 Houston, TX 77005 800-234-5315 281-436-5357 Specimen 1 Referees Extent 1 Extent 2 Total Few to 534 Giardia lamblia Many
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Luton and Dunstable University Hospital NHS Foundation Trust Lewsey Road Luton Bedfordshire LU4 0DZ Contact: Pauline Philip Tel: +44 (0)1582
2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Luton and Dunstable University Hospital NHS Foundation Trust Lewsey Road Luton Bedfordshire LU4 0DZ Contact: Pauline Philip Tel: +44 (0)1582
Saliva & Feces analysis
 Saliva & Feces analysis Saliva analysis What is Saliva? Watery substance present in humans and animals and secreted by salivary glands and aids in digestion It is a filtrate of plasma Contains electrolytes,
Saliva & Feces analysis Saliva analysis What is Saliva? Watery substance present in humans and animals and secreted by salivary glands and aids in digestion It is a filtrate of plasma Contains electrolytes,
CONTROL OF VIRAL GASTROENTERITIS OUTBREAKS IN CALIFORNIA LONG-TERM CARE FACILITIES
 CONTROL OF VIRAL GASTROENTERITIS OUTBREAKS IN CALIFORNIA LONG-TERM CARE FACILITIES California Department of Health Services Division of Communicable Disease Control In Conjunction with Licensing and Certification
CONTROL OF VIRAL GASTROENTERITIS OUTBREAKS IN CALIFORNIA LONG-TERM CARE FACILITIES California Department of Health Services Division of Communicable Disease Control In Conjunction with Licensing and Certification
Pathogen specific exclusion criteria for people at increased risk of transmitting an infection to others
 Appendix 2: disease March 2018 Exclusion/Restriction Cases of most enteric disease should be considered infectious and should remain off work/school until 48 hours after symptoms have ceased. Certain individuals
Appendix 2: disease March 2018 Exclusion/Restriction Cases of most enteric disease should be considered infectious and should remain off work/school until 48 hours after symptoms have ceased. Certain individuals
Abstract. Introduction
 DECEMBER FEBRUARY 2017 2018 What How can causes we keep lizards kids brains from dying to from change diarrhea? size? Authors: Authors: Susan Crow, Meghan Pawlowski, Manyowa Meki, Patricia Lara LaDage,
DECEMBER FEBRUARY 2017 2018 What How can causes we keep lizards kids brains from dying to from change diarrhea? size? Authors: Authors: Susan Crow, Meghan Pawlowski, Manyowa Meki, Patricia Lara LaDage,
Patient: Ima Sample. Accession: Shiloh Rd, Ste 101. Collected: 2/10/2018. Received: 2/12/2018 Alpharetta GA
 GI-MAP TM DNA Stool Analysis Patient: Ima Sample Accession: 20180212-0001 5895 Shiloh Rd, Ste 101 Collected: 2/10/2018 Received: 2/12/2018 Alpharetta GA 30005 877-485-5336 DOB: 7/11/1981 Completed: Ordered
GI-MAP TM DNA Stool Analysis Patient: Ima Sample Accession: 20180212-0001 5895 Shiloh Rd, Ste 101 Collected: 2/10/2018 Received: 2/12/2018 Alpharetta GA 30005 877-485-5336 DOB: 7/11/1981 Completed: Ordered
Lesson 1.5. The Usual Suspects. Estimated time: Two 50 min periods. Instructional overview. Instructional objectives. Assessment
 Lesson 1.5 The Usual Suspects Estimated time: Two 50 min periods Instructional overview Using information from peer presentations, students will identify the ten most common foodborne pathogens and recognize
Lesson 1.5 The Usual Suspects Estimated time: Two 50 min periods Instructional overview Using information from peer presentations, students will identify the ten most common foodborne pathogens and recognize
BILLING RATES - FY 18/19
 Meat-Poultry Inspection Department Cross Utilization "Regular Time" per hour $22.93 USDA Meat Inspection Cross Utilization "Over Time Rate" per hour $25.40 USDA Meat Inspection Overtime Rates per hour
Meat-Poultry Inspection Department Cross Utilization "Regular Time" per hour $22.93 USDA Meat Inspection Cross Utilization "Over Time Rate" per hour $25.40 USDA Meat Inspection Overtime Rates per hour
COMPREHENSIVE STOOL ANALYSIS
 COMPREHENSIVE STOOL ANALYSIS Intestinal parasites: Normal value = 0 (not marked) Reference range: 0 (negative) - 4 (heavy presence) Specimens fixed and transported in SAF and concentrated using CONSED
COMPREHENSIVE STOOL ANALYSIS Intestinal parasites: Normal value = 0 (not marked) Reference range: 0 (negative) - 4 (heavy presence) Specimens fixed and transported in SAF and concentrated using CONSED
Specimen Type. Usual Tests Ordered
 Specimen Type ABSCESS Open Closed BITE WOUND BLOOD CULTURE BLOOD CULTURE, FUNGUS BLOOD CULTURE, AFB Usual Tests Ordered CXBLD Blood Culture CXFNB Culture, Fungal, Blood CAFBB Mycobacterial Culture, Blood
Specimen Type ABSCESS Open Closed BITE WOUND BLOOD CULTURE BLOOD CULTURE, FUNGUS BLOOD CULTURE, AFB Usual Tests Ordered CXBLD Blood Culture CXFNB Culture, Fungal, Blood CAFBB Mycobacterial Culture, Blood
Rapid-VIDITEST. Rota-Adeno. One Step Rotavirus and Adenovirus Antigen Blister test. Instruction manual
 Rapid-VIDITEST Rota-Adeno One Step Rotavirus and Adenovirus Antigen Blister test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec, 252 42 Jesenice, Czech Republic, Tel.: +420
Rapid-VIDITEST Rota-Adeno One Step Rotavirus and Adenovirus Antigen Blister test. Instruction manual Producer: VIDIA spol. s r.o., Nad Safinou II 365, Vestec, 252 42 Jesenice, Czech Republic, Tel.: +420
FOODBORNE INFECTIONS. Caroline Charlier-Woerther March 2017
 FOODBORNE INFECTIONS Caroline Charlier-Woerther March 2017 LEARNING OBJECTIVES Know the pathogens involved in diarrheas Know the basics of management of diarrhea Know the main patterns of listeriosis and
FOODBORNE INFECTIONS Caroline Charlier-Woerther March 2017 LEARNING OBJECTIVES Know the pathogens involved in diarrheas Know the basics of management of diarrhea Know the main patterns of listeriosis and
Specimen Type. Usual Tests Ordered
 Specimen Type ABSCESS Open Closed BITE WOUND BLOOD CULTURE BLOOD CULTURE, FUNGUS BLOOD CULTURE, AFB Usual Tests Ordered CXBLD Blood Culture CXFNB Culture, Fungal, Blood CAFBB Mycobacterial Culture, Blood
Specimen Type ABSCESS Open Closed BITE WOUND BLOOD CULTURE BLOOD CULTURE, FUNGUS BLOOD CULTURE, AFB Usual Tests Ordered CXBLD Blood Culture CXFNB Culture, Fungal, Blood CAFBB Mycobacterial Culture, Blood
Bacterial Enteric Infections Detected by Culture-Independent Diagnostic Tests FoodNet, United States,
 Bacterial Enteric Infections Detected by Culture-Independent Diagnostic Tests FoodNet, United States, 2012 2014 Martha Iwamoto, MD 1, Jennifer Y. Huang, MPH 1, Alicia B. Cronquist, MPH 2, Carlota Medus,
Bacterial Enteric Infections Detected by Culture-Independent Diagnostic Tests FoodNet, United States, 2012 2014 Martha Iwamoto, MD 1, Jennifer Y. Huang, MPH 1, Alicia B. Cronquist, MPH 2, Carlota Medus,
Specimen Type. Usual Tests Ordered
 Specimen Type ABSCESS Open Closed BITE WOUND BLOOD CULTURE BLOOD CULTURE, FUNGUS BLOOD CULTURE, AFB Usual Tests Ordered CXBLD Blood Culture CXFNB Culture, Fungal, Blood CAFBB Mycobacterial Culture, Blood
Specimen Type ABSCESS Open Closed BITE WOUND BLOOD CULTURE BLOOD CULTURE, FUNGUS BLOOD CULTURE, AFB Usual Tests Ordered CXBLD Blood Culture CXFNB Culture, Fungal, Blood CAFBB Mycobacterial Culture, Blood
Reflex Test Protocols
 UCH Clinical Laboratory Reflex Test Protocols Blood Bank Prepare RBCs for Transfusion (aka Crossmatch) N/A No Antibody Screen ordered Antibody Screen Direct Antiglobulin Test (DAT) Polyspecific DAT Positive
UCH Clinical Laboratory Reflex Test Protocols Blood Bank Prepare RBCs for Transfusion (aka Crossmatch) N/A No Antibody Screen ordered Antibody Screen Direct Antiglobulin Test (DAT) Polyspecific DAT Positive
Schedule of Accreditation issued by United Kingdom Accreditation Service 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK
 2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Issue date: 17 August 2016 Worcestershire Royal Hospital Contact: Peter Jackson Microbiology Laboratory Tel: +44 (0) 1905 760192 Ext 30659
2 Pine Trees, Chertsey Lane, Staines-upon-Thames, TW18 3HR, UK Issue date: 17 August 2016 Worcestershire Royal Hospital Contact: Peter Jackson Microbiology Laboratory Tel: +44 (0) 1905 760192 Ext 30659
Rev. 6/18/2018. Specimen Type. Usual Tests Ordered
 Specimen Type ABSCESS Open Closed BITE WOUND BLOOD CULTURE BLOOD CULTURE, FUNGUS BLOOD CULTURE, AFB Usual Tests Ordered CXBLD Blood Culture CXFNB Culture, Fungal, Blood CAFBB Mycobacterial Culture, Blood
Specimen Type ABSCESS Open Closed BITE WOUND BLOOD CULTURE BLOOD CULTURE, FUNGUS BLOOD CULTURE, AFB Usual Tests Ordered CXBLD Blood Culture CXFNB Culture, Fungal, Blood CAFBB Mycobacterial Culture, Blood
Bacterial Enteric Pathogens: Clostridium difficile, Salmonella, Shigella, Escherichia coli, and others
 GUIDE TO INFECTION CONTROL IN THE HOSPITAL CHAPTER 48 Bacterial Enteric Pathogens: Clostridium difficile, Salmonella, Shigella, Escherichia coli, and others Authors Olivier Vandenberg, MD, PhD Michèle
GUIDE TO INFECTION CONTROL IN THE HOSPITAL CHAPTER 48 Bacterial Enteric Pathogens: Clostridium difficile, Salmonella, Shigella, Escherichia coli, and others Authors Olivier Vandenberg, MD, PhD Michèle
Reading material: MEDICAL MICROBIOLOGY
 1 MEDICAL MICROBIOLOGY Pathology Residents rotate for at least three months in Medical Microbiology. These rotations are designed to introduce the resident to Medical Microbiology and allow them to become
1 MEDICAL MICROBIOLOGY Pathology Residents rotate for at least three months in Medical Microbiology. These rotations are designed to introduce the resident to Medical Microbiology and allow them to become
Isolation Precautions in Clinics
 Purpose Audience General principles Possible Exposures To define isolation precautions in a clinic setting. Clinics Isolation status should be determined primarily by the suspected disease and/or pathogen.
Purpose Audience General principles Possible Exposures To define isolation precautions in a clinic setting. Clinics Isolation status should be determined primarily by the suspected disease and/or pathogen.
LABORATORY TRENDS. Recent Cluster of Locally-Acquired Cyclospora BC PUBLIC HEALTH MICROBIOLOGY & REFERENCE LABORATORY. Vancouver, BC.
 BC PUBLIC HEALTH In this Issue: Recent Cluster of Locally-Acquired Cyclospora The single-cell protozoan parasite Cyclospora cayetanensis is a rare but well known cause of diarrheal disease. Infections
BC PUBLIC HEALTH In this Issue: Recent Cluster of Locally-Acquired Cyclospora The single-cell protozoan parasite Cyclospora cayetanensis is a rare but well known cause of diarrheal disease. Infections
Anton van Leeuwenhoek. Protozoa: This is what he saw in his own stool sample. Morphology 10/14/2009. Protozoans that cause diarrheal disease
 Access to safe drinking water is everyone s right Anton van Leeuwenhoek Protozoa: Protozoans that cause diarrheal disease This is what he saw in his own stool sample 1. Giardia lamblia 2. Entameba histolytica
Access to safe drinking water is everyone s right Anton van Leeuwenhoek Protozoa: Protozoans that cause diarrheal disease This is what he saw in his own stool sample 1. Giardia lamblia 2. Entameba histolytica
Outline. Cryptococcosis Pneumocystosis Diarrhea. Case Histories: HIV Related- Opportunistic Infections in 2015
 AU Edited: 05/06/15 Case Histories: HIV Related- Opportunistic Infections in 2015 Henry Masur, MD Clinical Professor of Medicine George Washington University School of Medicine Bethesda, Maryland Washington,
AU Edited: 05/06/15 Case Histories: HIV Related- Opportunistic Infections in 2015 Henry Masur, MD Clinical Professor of Medicine George Washington University School of Medicine Bethesda, Maryland Washington,
Access to safe drinking water is everyone s right. Protozoans that cause diarrheal disease
 Access to safe drinking water is everyone s right Protozoa: Protozoans that cause diarrheal disease 1. Giardia lamblia 2. Entameba histolytica 3. Cryptosporidium parvum 4. Cyclospora cayetanensis 1 Giardia
Access to safe drinking water is everyone s right Protozoa: Protozoans that cause diarrheal disease 1. Giardia lamblia 2. Entameba histolytica 3. Cryptosporidium parvum 4. Cyclospora cayetanensis 1 Giardia
Reflex Test Protocols
 Blood Bank Prepare RBCs for Transfusion (aka Crossmatch) N/A No Antibody Screen ordered Antibody Screen Direct Antiglobulin Test (DAT) Polyspecific DAT Positive polyspecific DAT IgG DAT, C3 DAT Fetal Cell
Blood Bank Prepare RBCs for Transfusion (aka Crossmatch) N/A No Antibody Screen ordered Antibody Screen Direct Antiglobulin Test (DAT) Polyspecific DAT Positive polyspecific DAT IgG DAT, C3 DAT Fetal Cell
AHS Laboratory Services Central Zone Guide to Lab Services Manual Cultures/Procedures
 AEROBIC CULTURE Routine culture and routine aerobic culture are synonymous terms. Aerobic culture attempts to identify bacteria which grow in the presence of oxygen and are associated with potential infection
AEROBIC CULTURE Routine culture and routine aerobic culture are synonymous terms. Aerobic culture attempts to identify bacteria which grow in the presence of oxygen and are associated with potential infection
2015 Update in Infectious Diseases: New Tools in Diagnostic Microbiology
 2015 Update in Infectious Diseases: New Tools in Diagnostic Microbiology Bert K. Lopansri, MD Chief, Intermountain Division of Infectious Diseases Medical Director, Central Microbiology Lab Conflicts of
2015 Update in Infectious Diseases: New Tools in Diagnostic Microbiology Bert K. Lopansri, MD Chief, Intermountain Division of Infectious Diseases Medical Director, Central Microbiology Lab Conflicts of
Collection Container Category Source Tests Order Codes
 Nasal Nares culture for MRSA screen ONLY (Outpatient) NSL Room BD BBL Dual Culture Swab (Red Cap) Ear Ear culture EARC Room Eye Eye Culture EYEC Room Cultures Vaginal Genital Culture GEN or GC Room Superficial
Nasal Nares culture for MRSA screen ONLY (Outpatient) NSL Room BD BBL Dual Culture Swab (Red Cap) Ear Ear culture EARC Room Eye Eye Culture EYEC Room Cultures Vaginal Genital Culture GEN or GC Room Superficial
Meeting the Challenge of Changing Diagnostic Testing Practices and the Impact on Public Health Surveillance
 National Center for Emerging and Zoonotic Infectious Diseases Meeting the Challenge of Changing Diagnostic Testing Practices and the Impact on Public Health Surveillance Aimee Geissler, PhD, MPH FoodNet
National Center for Emerging and Zoonotic Infectious Diseases Meeting the Challenge of Changing Diagnostic Testing Practices and the Impact on Public Health Surveillance Aimee Geissler, PhD, MPH FoodNet
Proficiency Testing Materials for use with FilmArray Panels
 Proficiency Testing Materials for use with FilmArray Panels Introduction This document provides a listing of proficiency testing materials that have been tested at BioFire and are compatible with the FilmArray
Proficiency Testing Materials for use with FilmArray Panels Introduction This document provides a listing of proficiency testing materials that have been tested at BioFire and are compatible with the FilmArray
Clostridium difficile infection surveillance: Applying the case definition
 Clostridium difficile infection surveillance: Applying the case definition PICNet Conference March 3 rd 2016 Presented by: Tara Leigh Donovan, MSc Managing Consultant (Former Epidemiologist) 1 Disclaimer
Clostridium difficile infection surveillance: Applying the case definition PICNet Conference March 3 rd 2016 Presented by: Tara Leigh Donovan, MSc Managing Consultant (Former Epidemiologist) 1 Disclaimer
Viral gastroenteritis Gastrointestinal infections caused by viruses are the most common and the most contagious.3
 CMA Today Quick Clinic May/Jun 2017 (Issue 3/Volume 50) GI woes Gastroenteritis affects patients at home and abroad By Nancy Solomon We expect patients to come into the office a few times a year with that
CMA Today Quick Clinic May/Jun 2017 (Issue 3/Volume 50) GI woes Gastroenteritis affects patients at home and abroad By Nancy Solomon We expect patients to come into the office a few times a year with that
Update on infections with and clinical lab guidelines for Shiga toxin-producing E. coli (STEC) in the United States
 Update on infections with and clinical lab guidelines for Shiga toxin-producing E. coli (STEC) in the United States Patricia M. Griffin, MD Enteric Diseases Epidemiology Branch Centers for Disease Control
Update on infections with and clinical lab guidelines for Shiga toxin-producing E. coli (STEC) in the United States Patricia M. Griffin, MD Enteric Diseases Epidemiology Branch Centers for Disease Control
Infection Control Manual Residential Care Part 3 Infection Control Standards IC6: Additional Precautions
 IC6: 0110 Appendix I Selection Table Infection Control Manual esidential Care IC6: Additional Legend: outine Practice * reportable to Public Health C - Contact ** reportable by Lab D - Droplet A - Airborne
IC6: 0110 Appendix I Selection Table Infection Control Manual esidential Care IC6: Additional Legend: outine Practice * reportable to Public Health C - Contact ** reportable by Lab D - Droplet A - Airborne
Communicable diseases. Gastrointestinal track infection. Sarkhell Araz MSc. Public health/epidemiology
 Communicable diseases Gastrointestinal track infection Sarkhell Araz MSc. Public health/epidemiology Communicable diseases : Refer to diseases that can be transmitted and make people ill. They are caused
Communicable diseases Gastrointestinal track infection Sarkhell Araz MSc. Public health/epidemiology Communicable diseases : Refer to diseases that can be transmitted and make people ill. They are caused
Proficiency Testing Materials for use with BioFire FilmArray Panels
 Proficiency Materials for use with BioFire FilmArray Panels Introduction This document provides a listing of proficiency testing materials that have been tested at BioFire and are compatible with the BioFire
Proficiency Materials for use with BioFire FilmArray Panels Introduction This document provides a listing of proficiency testing materials that have been tested at BioFire and are compatible with the BioFire
Hot Topics in Infectious Diseases: Enteric Infections in the Arctic
 Hot Topics in Infectious Diseases: Enteric Infections in the Arctic Tobey Audcent MD, FRCPC Department of Pediatrics Children s Hospital of Eastern Ontario 6 th International Meeting on Indigenous Child
Hot Topics in Infectious Diseases: Enteric Infections in the Arctic Tobey Audcent MD, FRCPC Department of Pediatrics Children s Hospital of Eastern Ontario 6 th International Meeting on Indigenous Child
EDUCATIONAL COMMENTARY CLOSTRIDIUM DIFFICILE UPDATE
 EDUCATIONAL COMMENTARY CLOSTRIDIUM DIFFICILE UPDATE Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE CME/CMLE credits click
EDUCATIONAL COMMENTARY CLOSTRIDIUM DIFFICILE UPDATE Educational commentary is provided through our affiliation with the American Society for Clinical Pathology (ASCP). To obtain FREE CME/CMLE credits click
Microbiology- One Lab s Transformation from Ordinary to Extraordinary
 Microbiology- One Lab s Transformation from Ordinary to Extraordinary Christy Saum, MT (ASCP) Laboratory Specialist-Microbiology Bryan Medical Center Bryan Medical Center-East In the Beginning In the Beginning
Microbiology- One Lab s Transformation from Ordinary to Extraordinary Christy Saum, MT (ASCP) Laboratory Specialist-Microbiology Bryan Medical Center Bryan Medical Center-East In the Beginning In the Beginning
Epidemiology of Diarrheal Diseases. Robert Black, MD, MPH Johns Hopkins University
 This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike License. Your use of this material constitutes acceptance of that license and the conditions of use of materials on this
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike License. Your use of this material constitutes acceptance of that license and the conditions of use of materials on this
PROTOZOAN ENTERIC INFECTION IN AIDS RELATED DIARRHEA IN THAILAND
 PROTOZOAN ENTERIC INFECTION IN AIDS RELATED DIARRHEA IN THAILAND Duangdao Waywa 1, Siriporn Kongkriengdaj 1, Suparp Chaidatch 1, Surapee Tiengrim 1 Boonchai Kowadisaiburana 2, Suchada Chaikachonpat 1,
PROTOZOAN ENTERIC INFECTION IN AIDS RELATED DIARRHEA IN THAILAND Duangdao Waywa 1, Siriporn Kongkriengdaj 1, Suparp Chaidatch 1, Surapee Tiengrim 1 Boonchai Kowadisaiburana 2, Suchada Chaikachonpat 1,
Management of Needlestick and Mucous Membrane Exposures To Blood/Body Fluids
 Management of Needlestick and Mucous Membrane Exposures To Blood/Body Fluids August 2004 Table Of Contents Protocol - Management of Needlestick and Mucous Membrane Exposures to Blood/Body Fluids Assessment
Management of Needlestick and Mucous Membrane Exposures To Blood/Body Fluids August 2004 Table Of Contents Protocol - Management of Needlestick and Mucous Membrane Exposures to Blood/Body Fluids Assessment
Influenza-Associated Pediatric Mortality rev Jan 2018
 rev Jan 2018 Infectious Agent Influenza A, B or C virus BASIC EPIDEMIOLOGY Transmission Transmission occurs via droplet spread. After a person infected with influenza coughs, sneezes, or talks, influenza
rev Jan 2018 Infectious Agent Influenza A, B or C virus BASIC EPIDEMIOLOGY Transmission Transmission occurs via droplet spread. After a person infected with influenza coughs, sneezes, or talks, influenza
Guidance for obtaining faecal specimens from patients with diarrhoea (Background information)
 Guidance for obtaining faecal specimens from patients with diarrhoea (Background information) Version 1.0 Date of Issue: January 2009 Review Date: January 2010 Page 1 of 11 Contents 1. Introduction...
Guidance for obtaining faecal specimens from patients with diarrhoea (Background information) Version 1.0 Date of Issue: January 2009 Review Date: January 2010 Page 1 of 11 Contents 1. Introduction...
Perform Gram stain only. Select Survey D5, Gram Stain
 www.cap.org Bacteriology This flowchart is provided as a guide for ordering the appropriate Bacteriology Surveys for your laboratory s testing menu. In order to meet CLIA requirements for the subspecialty
www.cap.org Bacteriology This flowchart is provided as a guide for ordering the appropriate Bacteriology Surveys for your laboratory s testing menu. In order to meet CLIA requirements for the subspecialty
Interpretation At-a-Glance Bacteriology. Parasitology
 Patient: EMILY TET DOB: December 31, 1980 ex: F MN: 0001558266 Order Number: M9301014 Completed: January 31, 2019 eceived: January 30, 2019 Collected: January 30, 2019 Test Office Test POD Test MD, DO,
Patient: EMILY TET DOB: December 31, 1980 ex: F MN: 0001558266 Order Number: M9301014 Completed: January 31, 2019 eceived: January 30, 2019 Collected: January 30, 2019 Test Office Test POD Test MD, DO,
Gastroenteritis Outbreaks rev Apr 2017
 rev Apr 2017 Gastroenteritis is common, and so are gastroenteritis outbreaks. Rapid investigation of outbreaks of unknown etiology is critical for the identification of contaminated food vehicles or other
rev Apr 2017 Gastroenteritis is common, and so are gastroenteritis outbreaks. Rapid investigation of outbreaks of unknown etiology is critical for the identification of contaminated food vehicles or other
H E A L T H & R E S E A R C H. Innovating the future
 H E A L T H & R E S E A R C H 1 Index Company profile Export Countries Product Range: clinical diagnosis Technology New Launching: Shigella Shigella dysenteriae Calprotectin HAV Products pipeline Norovirus
H E A L T H & R E S E A R C H 1 Index Company profile Export Countries Product Range: clinical diagnosis Technology New Launching: Shigella Shigella dysenteriae Calprotectin HAV Products pipeline Norovirus
a) decide whether an investigation can be carried out (sample(s) or other evidence is available for analysis)
 ENT-1 ENTERIC DISEASE Background At the Surveillance Advisory Group workshop held in March 1999, it was agreed that all reported cases of enteric disease, whether via doctor notification, self-report or
ENT-1 ENTERIC DISEASE Background At the Surveillance Advisory Group workshop held in March 1999, it was agreed that all reported cases of enteric disease, whether via doctor notification, self-report or
A New Multiplex Real-time PCR Assay For Detection Of Intestinal Parasites
 A New Multiplex Real-time PCR Assay For Detection Of Intestinal Parasites Dr. Andreas Simons Worldwide provider of diagnostic assay solutions Offers a variety of test kit methodologies R-Biopharm Headquarters
A New Multiplex Real-time PCR Assay For Detection Of Intestinal Parasites Dr. Andreas Simons Worldwide provider of diagnostic assay solutions Offers a variety of test kit methodologies R-Biopharm Headquarters
