Literature Scan: Drugs for BPH
|
|
|
- Jack Francis
- 6 years ago
- Views:
Transcription
1 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon Phone Fax Literature Scan: Drugs for BPH Date of Review: July 2016 Date of Last Review: May 2014 Literature Search: May 2016 Current Status of PDL Class: See Appendix 1. Conclusions: One new guideline, 3 systematic reviews and one Food and Drug Administration (FDA) safety alert have been published for benign prostatic hypertrophy (BPH) therapies since the last review. 1 5 There is evidence that therapies for BPH all significantly improve the International Prostate Symptom Score (IPSS) compared to placebo by 3.69 to 7.06 points. Doxazocin and terazosin were associated with the most improved IPSS scores of 7.06 and 6.76, respectively. 1 The IPSS is a validated tool comprised of up to 35 points based on 8 questions. A minimally important difference is a decrease of 3 or more points; a moderate improvement is a decrease of 5 or more points; and marked improvement in symptoms is a decrease of 8 or more points. 2 Evidence from the Agency for Healthcare Research and Quality (AHRQ) found alpha 1 blockers to have similar or superior outcomes to newer therapies, or combination of therapies, for BPH. Alpha 1 blockers silodosin and tamsulosin changed IPSS scores by 7.8 and 7.2, respectively (weighted mean difference [WMD] 0.63; 95% CI, 1.62 to 0.36). 3 Other evidence showed tadalafil and tamsulosin resulted in 5.6 and 5.9 IPSS point reduction, respectively (WMD 0.07; 95% CI, 2.12 to 2.23). The addition of an alpha 1 blocker (tamsulosin or alfuzosin) to tadalafil provided an additional 1.6 point IPSS decrease over alpha 1 blocker monotherapy ( 10.4 vs. 8.6; WMD 2.01; 95% CI, 4.03 to 0.00). Similar differences were seen with the combination of tadalafil and finasteride compared to finasteride alone ( 5.5 vs. 4.5 points (CI not provided). Adverse events were more common with silodosin compared to placebo and tamsulosin. Tadalafil was associated with higher risk of adverse events compared to tamsulosin. 3 Guideline recommendations from the National Institute for Health and Care Excellence (NICE) support current preferred drug list (PDL) placement for drugs used in the treatment of BPH. 4 Recommendations: No changes are recommended to the PDL or prior authorization criteria for BPH treatments. Consider costs in executive session. Previous Conclusions/Conclusions: There is no new evidence for comparative effectiveness or harms outcomes between drugs used for BPH. No further review or research needed at this time. Author: Kathy Sentena, PharmD Date: July 2016
2 Methods: A Medline literature search for new systematic reviews and randomized controlled trials (RCTs) assessing clinically relevant outcomes to active controls, or placebo if needed, was conducted. A summary of the clinical trials is available in Appendix 2 with abstracts presented in Appendix 3. The Medline search strategy used for this literature scan is available in Appendix 4, which includes dates, search terms and limits used. The OHSU Drug Effectiveness Review Project, Agency for Healthcare Research and Quality (AHRQ), Cochrane Collection, National Institute for Health and Clinical Excellence (NICE), Department of Veterans Affairs, BMJ Clinical Evidence, and the Canadian Agency for Drugs and Technologies in Health (CADTH) resources were manually searched for high quality and relevant systematic reviews. When necessary, systematic reviews are critically appraised for quality using the AMSTAR tool and clinical practice guidelines using the AGREE tool. The FDA website was searched for new drug approvals, indications, and pertinent safety alerts. Finally, the AHRQ National Guideline Clearinghouse (NGC) was searched for updated and recent evidence based guidelines. The primary focus of the evidence is on high quality systematic reviews and evidence based guidelines. Randomized controlled trials will be emphasized if evidence is lacking or insufficient from those preferred sources. New Systematic Reviews: A systematic review evaluated the comparative effectiveness of monotherapies for the treatment of BPH. Drugs included in the review were the following: terazosin, doxazosin, tamsulosin, alfuzosin, silodosin, naftopidil, finasteride, dustasteride, tolterodine, fesoterodine, and solifenacin. 1 The primary outcome was change in IPSS. One hundred twenty four trials were included with two thirds having a treatment duration longer than one year. The median age of patients was 65 years with a median baseline IPSS score of with moderate to severe symptoms. 1 The evidence was rated as moderate in quality. In placebo comparisons, a network meta analysis found BPH therapies to decrease IPSS scores by 3.69 to 7.06 points. The largest decreases were found with doxazosin (absolute effect [AE] 7.06; 95% CI, to 3.71) and terazosin (AE 6.76; 95% CI, to 3.35). Significant IPSS changes were seen for all treatments except tolterodine and solifenacin. Most changes in IPSS were comparable to each other except for doxazocin and terazosin which were found to be significantly more effective than tamsulosin, alfuzosin, tadalafil, naftopidil, dutasteride, finasteride, tolterodine and solifenacin. 1 A significantly higher incidence of adverse effects (RR 1.33 to 2.10) was found with doxazosin, terazosin, silodosin, fesoterodine and tadalafil. Therapies that were associated with the highest incidence of withdrawals due to adverse events were: alfuzosin, terazosin, dutasteride, tolterodine, tadalafil, sildenafil and vardenafil. 1 A systematic review by AHRQ evaluated medications for symptom management associated with BPH. 3 Evidence was analyzed from 57 RCTs and 5 observational studies which included the following medications: silodosin, tolterodine, solifenacin, festoterodine, mirabegron, tadalafil and sildenafil. 3 In a review of alpha 1 blockers, 4 12 week trials found silodosin to be more effective than placebo in improving lower urinary tract symptoms (LUTS) caused by BPH with a weighted mean difference (WMD) in IPSS score of Silodosin was compared to tamsulosin in trials lasting 4 to 12 weeks and was found to reduce IPSS scores similar to tamsulosin ( 7.8 vs. 7.2) but with a degree of heterogeneity (I 2 = 76%). More adverse events were associated with silodosin compared to tamsulosin, however, withdrawal rates were similar. 3 For anticholinergics, combination therapy of tolterodine, solifenacin and fesoterodine combined with alpha 1 blockers were similar in efficacy to alpha 1 blocker monotherapy. There was insufficient evidence to compare the adverse events of combination versus monotherapy. 3 For phosphodiesterase 5 (PDE 5) inhibitors, tadalafil improved IPSS scores more than placebo in a pooled analysis of week trials (n=3516) of mostly white participants ( 5.5 vs. 3.4, respectively). Four small trials found tadalafil combined with an alpha 1 blocker to be more effective in symptom improvement than alpha 1 blocker monotherapy ( 10.4 and 8.6) respectively. 3 A one point improvement in IPSS scores was found with tadalafil/finasteride combination over finasteride monotherapy. Tadalafil was similar to tamsulosin in improving IPSS scores, 5.6 vs. 5.9, in men studied for 3 months. Evidence for sildenafil comparisons were deemed insufficient. Adverse events with tadalafil are similar to alpha 1 blockers but withdrawals related to adverse events are higher. 3 For
3 beta 3 adrenergic agonist, there was insufficient evidence on mirabegron to draw efficacy conclusions. 3 Limitations to the evidence were risk of performance and detection bias, limited applicability to patients over 70 years of age and high percentage of patients in PDE 5 inhibitor trials having erectile dysfunction. Long term effects on blood pressure, drug interactions and maintenance of symptom improvement has not been studied. A systematic review and meta analysis analyzed the efficacy and safety of dutasteride compared to placebo in men with symptomatic BPH. 5 Seven RCTs, 2 openlabel extension trials and 2 finasteride comparison trials were included (n=12,129). The primary outcome was the change in urinary symptoms assessed by changes in IPSS. Dutasteride was more effective in decreasing IPSS compared to placebo (weighted mean difference [WMD] 1.78, 95% CI, 3.01 to 0.55) but with significant heterogeneity (I 2 = 69%). 3 Symptom improvement was similar in dutasteride and finasteride comparisons. Dustasteride was associated with adverse events more commonly than placebo (RR 1.04; 95% CI, 1.00 to 1.07). Withdrawal rates were similar between groups. 5 New Guidelines: National Institute for Health and Care Excellence (NICE) In 2015 a guideline on the assessment and management of the lower urinary tract in men was released. 4 Recommendations as they pertain to drug treatment are: men with symptomatic lower urinary tract symptoms (LUTS) unresponsive to conservative management should be offered drug therapy with an alpha 1 blocker; men with enlarged prostates (estimated at 30 gms or PSA >1.4 ng/ml) and at risk of progression should be offered an 5 alpha reductase inhibitor; and combination therapy with an 5 alpha reductase inhibitor and an alpha 1 blocker should be offered to men with moderate to severe LUTS and enlarged prostate. Men with LUTS should not be offered a PDE 5 inhibitor for the sole treatment of LUTS. 4 New FDA Drug Approvals: None identified. New Formulations/Indications: None identified. New FDA Safety Alerts: In July of 2014 the FDA warned of the possibility of intraoperative floppy iris syndrome (IFIS) during cataract and glaucoma surgery in some patients on or previously treated with alpha 1 blockers, including tamsulosin. 6
4 References: 1. Yuan JQ, Mao C, Wong S, et al. Comparative effectiveness and safety of monodrug therapies for lower urinary tract symptoms associated with benign prostatic hyperplasia. Medicine; 94:e974. Doi: /MD Barry MJ, Williford WO, Chang Y et al. Benign prostatic hyperplasia specific health 30 status measures in clinical research: how much change in the American Urological 31 Association symptom index and the benign prostatic hyperplasia impact index is 32 perceptible to patients? J Urol 1995; 154: Brasure M, MacDonald R, Dahm P, et al. Newer medications for lower urinary tract symptoms attributed to benign prostatic hyperplasia: a review. Comparative Effectiveness Review No AHRQ Publication No. 16 EHC024 EF. Rockville, MD: Agency for Healthcare Research and Quality; May Accessed June 2, National Clinical Guideline Centre for Acute and Chronic Conditions. Lower urinary tract symptoms in men: assessment and management. London (UK): National Institute for Health and Care Excellence (NICE), June: no: Park T, Choi JY. Efficacy and safety of dutasteride for the treatment of symptomatic benign prostatic hyperplasia (BPH): a systematic review and metaanalysis. World J Urol 2014: 32: DOI: /s Food and Drug Administration. Flomax (tamsulosin) hydrochloride capsules safety warning. July 2014 Drug Safety Labeling Changes. Available at: Accessed April 30, Roehrborn C, Perez I, Roos E, et al. Efficacy and safety of a fixed dose combination of dutasteride and tamsulosin treatment (Duodart ) compared with watchful waiting with initiation of tamsulosin therapy if symptoms do not improve, both provided with lifestyle advice, in the management of treatmentnaïve men with moderately symptomatic benign prostatic hyperplasia: 2 year CONDUCT study results. BJU Int 2015;116: Appendix 1: Current Status on Preferred Drug List ROUTE FORMULATION BRAND GENERIC PDL ORAL CAP ER 24H FLOMAX TAMSULOSIN HCL Y ORAL CAP ER 24H TAMSULOSIN HCL TAMSULOSIN HCL Y ORAL CAPSULE TERAZOSIN HCL TERAZOSIN HCL Y ORAL TABLET CARDURA DOXAZOSIN MESYLATE Y ORAL TABLET DOXAZOSIN MESYLATE DOXAZOSIN MESYLATE Y ORAL TABLET FINASTERIDE FINASTERIDE Y ORAL TABLET PROSCAR FINASTERIDE Y ORAL CAPSULE AVODART DUTASTERIDE N ORAL CAPSULE DUTASTERIDE DUTASTERIDE N ORAL CAPSULE RAPAFLO SILODOSIN N ORAL TAB ER 24 CARDURA XL DOXAZOSIN MESYLATE N ORAL TAB ER 24H ALFUZOSIN HCL ER ALFUZOSIN HCL N ORAL TAB ER 24H UROXATRAL ALFUZOSIN HCL N ORAL CAPSULE JALYN DUTASTERIDE/TAMSULOSIN N
5 Appendix 2: New Clinical Trials A total of 347 citations were manually reviewed from the literature search. After further review, 346 trials were excluded because of wrong study design (observational), comparator (placebo), or outcome studied (non clinical). The remaining trial is briefly described in the table below. The full abstract is included in Appendix 3. Table 1: Description of Clinical Trial Study Comparison Population Primary Outcome Results Roehrborn, et al 7 OL, PG, RCT Dutasteride 0.5 mg and tamsulosin 0.4 mg vs. (D/T) Watchful waiting tamsulosin if no symptom improvement (WW) Treatment naïve men with moderately symptomatic BPH (IPSS score of 8 19) at risk of progression n = 742 Symptomatic improvement from baseline to 24 months, measured by IPSS Abbreviations: IPSS = International Prostate Symptom Score (IPSS); OL = open label; PG = parallel group; RCT = randomized controlled trial D/T: 5.4 points WW: 3.6 points (ETD 1.8; 95 % CI 2.5 to 1.2; P < 0.001) Appendix 3: Abstracts of Clinical Trials Roehrborn C, Perez I, Roos E, et al. Efficacy and safety of a fixed dose combination of dutasteride and tamsulosin treatment (Duodart ) compared with watchful waiting with initiation of tamsulosin therapy if symptoms do not improve, both provided with lifestyle advice, in the management of treatment naïve men with moderately symptomatic benign prostatic hyperplasia: 2 year CONDUCT study results. BJU Int 2015;116: Objective: To investigate whether a fixed dose combination (FDC) of 0.5 mg dutasteride and 0.4 mg tamsulosin is more effective than watchful waiting with protocol defined initiation of tamsulosin therapy if symptoms did not improve (WW All) in treatment naïve men with moderately symptomatic benign prostatic hyperplasia (BPH) at risk of progression. Methods: This was a multicentre, randomised, open label, parallel group study (NCT ) in 742 men with an International Prostate Symptom Score (IPSS) of 8 19, prostate volume 30 ml and total serum PSA level of 1.5 ng/ml. Patients were randomised to FDC (369 patients) or WW All (373) and followed for 24 months. All patients were given lifestyle advice. The primary endpoint was symptomatic improvement from baseline to 24 months, measured by the IPSS. Secondary outcomes included BPH clinical progression, impact on quality of life (QoL), and safety. Results: The change in IPSS at 24 months was significantly greater for FDC than WW All ( 5.4 vs 3.6 points, P < 0.001). With FDC, the risk of BPH progression was reduced by 43.1% (P < 0.001); 29% and 18% of men in the WW All and FDC groups had clinical progression, respectively, comprising symptomatic progression in most patients. Improvements in QoL (BPH Impact Index and question 8 of the IPSS) were seen in both groups but were significantly greater with FDC (P < 0.001). The safety profile of FDC was consistent with established profiles of dutasteride and tamsulosin. Conclusion: FDC therapy with dutasteride and tamsulosin, plus lifestyle advice, resulted in rapid and sustained improvements in men with moderate BPH symptoms at risk of progression with significantly greater symptom and QoL improvements and a significantly reduced risk of BPH progression compared with WW plus initiation of tamsulosin as per protocol.
6 Appendix 4: Medline Search Strategy Database(s): Ovid MEDLINE(R) without Revisions 1996 to April Week Search Strategy: # Searches Results Annotations 1 Doxazosin/ Finasteride/ Dutasteride/ silodosin.mp alfuzosin.mp tamsulosin.mp or 2 or 3 or 4 or 5 or limit 7 to (english language and humans and yr="2014 -Current") limit 8 to (clinical study or clinical trial, all or clinical trial, phase iii or clinical trial or comparative study or controlled clinical trial or meta-analysis or practice guideline or randomized controlled trial or systematic reviews) 143 Database(s): Ovid MEDLINE(R) without Revisions 1996 to April Week Search Strategy: # Searches Results Annotations 1 benign prostatic hypertrophy.mp. or Prostatic Hyperplasia/ limit 1 to (english language and humans) limit 2 to yr="2014 -Current" limit 3 to (clinical study or clinical trial, all or clinical trial, phase iii or clinical trial or controlled clinical trial or meta-analysis or practice guideline or randomized controlled trial or systematic reviews) 204
7 Appendix 5: Prior Authorization Criteria Benign Prostatic Hypertrophy (BPH) Medications Goal(s): BPH with urinary obstruction is an OHP-funded treatment only when post-void residuals are 150 ml or more. Restrict use for male pattern baldness and erectile dysfunction, which are not OHP-funded conditions. Length of Authorization: Up to 12 months Requires PA: Non-preferred drugs Covered Alternatives: Current PMPDP preferred drug list per OAR at Searchable site for Oregon FFS Drug Class listed at Approval Criteria 1. What diagnosis is being treated? Record ICD10 code 2. Will the prescriber consider switching to a preferred product? Message: Preferred products do not require a PA. Preferred products are evidence-based reviewed for comparative effectiveness and safety by the Oregon Pharmacy & Therapeutics Committee. Yes: Inform prescriber of covered alternatives in class. No: Go to #3 3. Is the request for continuation of therapy previously approved by the FFS program? Yes: Go to Renewal Criteria No: Go to #4
8 Approval Criteria 4. Is the request for an alpha-1 blocker, and does the patient have a diagnosis related to functional and mechanical disorders of the genitourinary system including bladder outlet obstruction? Yes: Go to #5 No: Go to #6 5. Has the patient tried and failed a 2-month trial of a preferred alpha-1 blocker? 6. Does the patient have a diagnosis of benign prostatic hypertrophy (BPH) or enlarged prostate with obstruction? 7. Does the patient have a diagnosis of unspecified urinary obstruction or BPH without obstruction? Yes: Approve an alpha-1 blocker for up to 12 months Yes: Approve for up to 12 months Yes: Pass to RPh. Deny; not funded by the OHP No: Pass to RPh. Deny until patient has tried and failed a covered alternative No: Go to #7 No: Pass to RPh. Go to #8 8. RPh Only: All other conditions need to be evaluated to see if diagnosis is funded: Funded: covered diagnoses related to prostate may be approved for 1 year. Not Funded: unfunded diagnoses (e.g., hair growth, erectile dysfunction) should be denied (not funded by the OHP). Alpha-1 blockers and 5-alpha reductase inhibitors may be used concurrently for BPH up to 1 year. Alpha-1 blockers may be discontinued once prostate is reduced to normal size. If urine retention (obstructive), ask for more specific diagnosis. Renewal Criteria 1. Is the request for an alpha-1 blocker and does the patient have a diagnosis related to functional and mechanical disorders of the genitourinary system including bladder outlet obstruction? 2. Has the patient also been taking a 5-alpha reductase inhibitor for the last year? Yes: Go to #2 No: Go to #3 Yes: Recommend against combination therapy exceeding 1 year. No: Approve for the shorter of 12 months or length of the prescription
9 3. Does the patient have a diagnosis of BPH or enlarged prostate with obstruction? 4. Does the patient have a diagnosis of unspecified urinary obstruction or benign prostatic hyperplasia without obstruction? Yes: Approve for up to 12 months No: Go to #4 Yes: Pass to RPh. Deny; not funded by the OHP No: Pass to RPh. Go to #5 5. RPh only: All other indications need to be evaluated as to whether they are a funded condition: Alpha Blockers and 5-alpha reductase inhibitors may be used concurrently for BPH up to 1 year. Alphablockers may be discontinued once prostate is reduced to normal size. If urine retention, obstructive, ask for more specific diagnosis. P&T Review: 7/16 (KS); 11/12; 9/10; 3/10; 5/08; 2/06 Implementation: 2/21/13; 1/1/11; 4/20/10; 5/22/08; 7/1/06; 9/30/05 If funded and clinic provides supporting literature, approve for up to 12 months. If non-funded, deny (not funded by the OHP).
Month/Year of Review: May 2014 Date of Last Review: November 2012 Source Document: OSU College of Pharmacy
 Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Copyright 2012 Oregon State University. All Rights
Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Copyright 2012 Oregon State University. All Rights
Literature Scan: Phosphate Binders
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Literature Scan: Erythropoiesis Stimulating Agents
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Literature Scan: Alzheimer s Drugs
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Literature Scan: Parenteral Antipsychotics
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Literature Scan: Antivirals for Herpes Simplex Virus
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
OHSU Drug Effectiveness Review Project Summary Report Long-Acting Insulins
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-2596
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-2596
Drug Class Literature Scan: Pancreatic Enzymes
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Literature Scan: Analgesics for Gout. Month/Year of Review: April 2015 Date of Last Review: January 2014
 Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301 1079 Phone 503 947 5220 Fax 503 947 1119 Copyright 2012 Oregon State University. All Rights
Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301 1079 Phone 503 947 5220 Fax 503 947 1119 Copyright 2012 Oregon State University. All Rights
National Institute for Health and Care Excellence. Lower Urinary Tract Symptoms Update Addendum Consultation Table 3 rd February 5 pm 3 rd March 2015
 British Association of Urological Surgeons British Association of Urological Surgeons National Institute for Health and Care Excellence Lower Urinary Tract Symptoms Update Addendum Consultation Table 3
British Association of Urological Surgeons British Association of Urological Surgeons National Institute for Health and Care Excellence Lower Urinary Tract Symptoms Update Addendum Consultation Table 3
Drug Class Literature Scan: Topical Antipsoriatics
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Centre for Clinical Practice Surveillance Programme
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Centre for Clinical Practice Surveillance Programme Clinical guideline CG97: The management of lower urinary tract symptoms in men Publication date May
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Centre for Clinical Practice Surveillance Programme Clinical guideline CG97: The management of lower urinary tract symptoms in men Publication date May
Literature Scan: Topical Analgesics
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Anticholinergics. COMT* Inhibitors. Dopaminergic Agents. Dopamine Agonists. Combination Product
 Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-945-5220 Fax 503-947-1119 Class Update: Parkinson s Drugs Month/Year of Review:
Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-945-5220 Fax 503-947-1119 Class Update: Parkinson s Drugs Month/Year of Review:
Class Update: Benign Prostatic Hyperplasia (BPH)
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Month/Year of Review: November 2014 Date of Last Review: June 2012 PDL Classes: Anti-anginals, Cardiovascular
 Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Copyright 2012 Oregon State University. All Rights
Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Copyright 2012 Oregon State University. All Rights
Benign Prostatic Hyperplasia (BPH) Important Papers / Landmark. Vijayan Manogran
 Benign Prostatic Hyperplasia (BPH) Important Papers / Landmark Studies Vijayan Manogran MTOPS & CombAT PLESS SMART ALTESS ALF-ONE VA PREDICT EPICS Landmark Studies MTOPS Medical Therapy of Prostatic Symptoms
Benign Prostatic Hyperplasia (BPH) Important Papers / Landmark Studies Vijayan Manogran MTOPS & CombAT PLESS SMART ALTESS ALF-ONE VA PREDICT EPICS Landmark Studies MTOPS Medical Therapy of Prostatic Symptoms
The Journal of International Medical Research 2012; 40:
 The Journal of International Medical Research 2012; 40: 899 908 Comparison of α-blocker Monotherapy and α-blocker Plus 5α-Reductase Inhibitor Combination Therapy Based on Prostate Volume for Treatment
The Journal of International Medical Research 2012; 40: 899 908 Comparison of α-blocker Monotherapy and α-blocker Plus 5α-Reductase Inhibitor Combination Therapy Based on Prostate Volume for Treatment
Victoria Sharp, MD, MBA, FAAFP. Clinical Professor of Urology and Family Medicine
 Victoria Sharp, MD, MBA, FAAFP Clinical Professor of Urology and Family Medicine Victoria Sharp, MD, MBA, FAAFP Market Chief Medial Officer AmeriHealth Caritas Family of Companies Office phone: (515) 330-3740
Victoria Sharp, MD, MBA, FAAFP Clinical Professor of Urology and Family Medicine Victoria Sharp, MD, MBA, FAAFP Market Chief Medial Officer AmeriHealth Caritas Family of Companies Office phone: (515) 330-3740
Index. urologic.theclinics.com. Note: Page numbers of article titles are in boldface type.
 Index Note: Page numbers of article titles are in boldface type. A Ablative therapies, transurethral needle ablation, Adverse events, sexual side effects of BPH Aging, and incidence of BPH associated with
Index Note: Page numbers of article titles are in boldface type. A Ablative therapies, transurethral needle ablation, Adverse events, sexual side effects of BPH Aging, and incidence of BPH associated with
The Evolution of Combination Therapy. US men eligible for BPH treatment * with projected population changes
 The Management of BPH & The Impact of Combination Therapy Results Combination of Avodart and Tamsulosin (CombAT) Medical Therapy of Prostate Symptoms (MTOPS) Dr. Jack Barkin, md, fics, facs, dabu, Mcert
The Management of BPH & The Impact of Combination Therapy Results Combination of Avodart and Tamsulosin (CombAT) Medical Therapy of Prostate Symptoms (MTOPS) Dr. Jack Barkin, md, fics, facs, dabu, Mcert
Management of male LUTS in general practice
 17 Management of male LUTS in general practice MARK J. SPEAKMAN AND FAITH MCMEEKIN The initial management of lower urinary tract symptoms in men is usually carried out in primary care. The authors explain
17 Management of male LUTS in general practice MARK J. SPEAKMAN AND FAITH MCMEEKIN The initial management of lower urinary tract symptoms in men is usually carried out in primary care. The authors explain
α-blocker Monotherapy and α-blocker Plus 5-Alpha-Reductase Inhibitor Combination Treatment in Benign Prostatic Hyperplasia; 10 Years Long-Term Results
 www.kjurology.org http://dx.doi.org/10.4111/kju.2012.53.4.248 Voiding Dysfunction α-blocker Monotherapy and α-blocker Plus 5-Alpha-Reductase Inhibitor Combination Treatment in Benign Prostatic Hyperplasia;
www.kjurology.org http://dx.doi.org/10.4111/kju.2012.53.4.248 Voiding Dysfunction α-blocker Monotherapy and α-blocker Plus 5-Alpha-Reductase Inhibitor Combination Treatment in Benign Prostatic Hyperplasia;
Prostate Disease. Chad Baxter, MD
 Prostate Disease Chad Baxter, MD Managing BPH and LUTS Chad Baxter, MD Department of Urology cbaxter@mednet.ucla.edu 33 nd Annual UCLA Intensive Course in Geriatric Medicine & Board Review Prevalence of
Prostate Disease Chad Baxter, MD Managing BPH and LUTS Chad Baxter, MD Department of Urology cbaxter@mednet.ucla.edu 33 nd Annual UCLA Intensive Course in Geriatric Medicine & Board Review Prevalence of
10/9/2015. Dana A. Brown, Pharm.D., BCPS Assistant Dean for Academics, Associate Professor of Pharmacy Practice Palm Beach Atlantic University
 Dana A. Brown, Pharm.D., BCPS Assistant Dean for Academics, Associate Professor of Pharmacy Practice Palm Beach Atlantic University 1. Explain the pathophysiology of benign prostatic hyperplasia (BPH),
Dana A. Brown, Pharm.D., BCPS Assistant Dean for Academics, Associate Professor of Pharmacy Practice Palm Beach Atlantic University 1. Explain the pathophysiology of benign prostatic hyperplasia (BPH),
Benign Prostatic Hyperplasia. Management of Benign Prostatic Hyperplasia. Goals of Therapy
 Benign Prostatic Hyperplasia Management of Benign Prostatic Hyperplasia Goals of Therapy Improve or abolish lower urinary tract symptoms (LUTS) Prevent or delay clinical progression of benign prostatic
Benign Prostatic Hyperplasia Management of Benign Prostatic Hyperplasia Goals of Therapy Improve or abolish lower urinary tract symptoms (LUTS) Prevent or delay clinical progression of benign prostatic
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: Reference Number: CP.HNCA.01 Effective Date: 11.16.16 Last Review Date: 04.18 Line of Business: Commercial - HNCA Revision Log See Important Reminder at the end of this policy for important
Clinical Policy: Reference Number: CP.HNCA.01 Effective Date: 11.16.16 Last Review Date: 04.18 Line of Business: Commercial - HNCA Revision Log See Important Reminder at the end of this policy for important
EVALUATION OF THE EFFICACY OF TADALAFIL IN IMPROVING LOWER URINARY TRACT SYMPTOMS IN PATIENTS WITH SYMPTOMATIC BENIGN PROSTATIC ENLARGEMENT
 Basrah Journal Of Surgery EVALUATION OF THE EFFICACY OF TADALAFIL IN IMPROVING LOWER URINARY TRACT SYMPTOMS IN PATIENTS WITH SYMPTOMATIC BENIGN PROSTATIC ENLARGEMENT MB, ChB, FIBMS, Assistant Professor
Basrah Journal Of Surgery EVALUATION OF THE EFFICACY OF TADALAFIL IN IMPROVING LOWER URINARY TRACT SYMPTOMS IN PATIENTS WITH SYMPTOMATIC BENIGN PROSTATIC ENLARGEMENT MB, ChB, FIBMS, Assistant Professor
Literature Scan: Antibiotics for Clostridium difficile Infection. Month/Year of Review: May 2015 Date of Last Review: April 2012
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Management of LUTS. Simon Woodhams February 2012
 Management of LUTS Simon Woodhams February 2012 The management of lower urinary tract symptoms (LUTS) in men Implementing NICE guidance May 2010 NICE clinical guideline 97 Background Lower urinary tract
Management of LUTS Simon Woodhams February 2012 The management of lower urinary tract symptoms (LUTS) in men Implementing NICE guidance May 2010 NICE clinical guideline 97 Background Lower urinary tract
Drug Effectiveness Review Project Literature Scan Summary. Month/Year of Review: January 2015 Date of Last Review: January 2013
 Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301 1079 Phone 503 947 5220 Fax 503 947 1119 Copyright 2012 Oregon State University. All Rights
Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301 1079 Phone 503 947 5220 Fax 503 947 1119 Copyright 2012 Oregon State University. All Rights
New Dosage Formulation. Month/Year of Review: June 2012 End date of literature search: May Dossier Received: No (Requested)
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Prior Authorization Criteria Update: Androgens, Topical and Parenteral
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Benign Prostatic Hyperplasia. Jay Lee, MD, FRCSC Clinical Associate Professor University of Calgary
 Benign Prostatic Hyperplasia Jay Lee, MD, FRCSC Clinical Associate Professor University of Calgary Copyright 2017 by Sea Courses Inc. All rights reserved. No part of this document may be reproduced, copied,
Benign Prostatic Hyperplasia Jay Lee, MD, FRCSC Clinical Associate Professor University of Calgary Copyright 2017 by Sea Courses Inc. All rights reserved. No part of this document may be reproduced, copied,
Evidence-based Practice Center Systematic Review Protocol
 Evidence-based Practice Center Systematic Review Protocol Project Title: Newer Medications for Lower Urinary Tract Symptoms associated with Benign Prostatic Hyperplasia I. Background and Objectives for
Evidence-based Practice Center Systematic Review Protocol Project Title: Newer Medications for Lower Urinary Tract Symptoms associated with Benign Prostatic Hyperplasia I. Background and Objectives for
Benign Prostatic Hyperplasia (BPH):
 Benign Prostatic Hyperplasia (BPH): Evidence Based Guidelines for Primary Care Providers Jeanne Martin, DNP, ANP-BC Objectives 1. Understand the pathophysiology and prevalence of BPH 2. Select the appropriate
Benign Prostatic Hyperplasia (BPH): Evidence Based Guidelines for Primary Care Providers Jeanne Martin, DNP, ANP-BC Objectives 1. Understand the pathophysiology and prevalence of BPH 2. Select the appropriate
OHSU Drug Effectiveness Review Project Summary Report Benzodiazepines
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Abbreviated Class Review: Long-Acting Injectable Antipsychotics
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Increasing Awareness, Diagnosis, and Treatment of BPH, LUTS, and EP
 Introduction to Enlarged Prostate E. David Crawford, MD Professor of Surgery (Urology) and Radiation Oncology Head, Urologic Oncology E. David Crawford Endowed Chair in Urologic Oncology University of
Introduction to Enlarged Prostate E. David Crawford, MD Professor of Surgery (Urology) and Radiation Oncology Head, Urologic Oncology E. David Crawford Endowed Chair in Urologic Oncology University of
New Treatment Modalities for Benign Prostatic Hyperplasia. Seung-June Oh, MD Department of Urology, Seoul National University Hospital
 New Treatment Modalities for Benign Prostatic Hyperplasia Seung-June Oh, MD Department of Urology, Seoul National University Hospital Options for Treating BPH Pharmacotherapy blocker Agents for BPH + OAB
New Treatment Modalities for Benign Prostatic Hyperplasia Seung-June Oh, MD Department of Urology, Seoul National University Hospital Options for Treating BPH Pharmacotherapy blocker Agents for BPH + OAB
Prior Authorization Update: Botulinum Toxins
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
H6D-MC-LVHR Clinical Study Report Synopsis Page LVHR Synopsis (LY450190)
 H6D-MC-LVHR Clinical Study Report Synopsis Page 1 2. LVHR Synopsis H6D-MC-LVHR Clinical Study Report Synopsis Page 2 Clinical Study Report Synopsis: Study H6D-MC-LVHR Title of Study: A Randomized, Double-Blind,
H6D-MC-LVHR Clinical Study Report Synopsis Page 1 2. LVHR Synopsis H6D-MC-LVHR Clinical Study Report Synopsis Page 2 Clinical Study Report Synopsis: Study H6D-MC-LVHR Title of Study: A Randomized, Double-Blind,
Systematic Review of Combination Drug Therapy for Non-neurogenic Male Lower Urinary Tract Symptoms
 EUROPEAN UROLOGY 64 (2013) 228 243 available at www.sciencedirect.com journal homepage: www.europeanurology.com Platinum Priority Collaborative Review Benign Prostatic Hyperplasia Editorial by Herbert
EUROPEAN UROLOGY 64 (2013) 228 243 available at www.sciencedirect.com journal homepage: www.europeanurology.com Platinum Priority Collaborative Review Benign Prostatic Hyperplasia Editorial by Herbert
Last Review Status/Date: December Summary
 Section: Surgery Effective Date: January 15, 2016 Subject: Prostatic Urethral Lift Page: 1 of 9 Last Review Status/Date: December 2015 Summary Benign prostatic hyperplasia (BPH) is a common condition in
Section: Surgery Effective Date: January 15, 2016 Subject: Prostatic Urethral Lift Page: 1 of 9 Last Review Status/Date: December 2015 Summary Benign prostatic hyperplasia (BPH) is a common condition in
Key words: Lower Urinary Tract Symptoms (LUTS), Prostatic Hyperplasia, Alpha-1 Adrenoceptor Antagonists, Tamsulosin, Terazosin.
 The Professional Medical Journal DOI: 10.17957/TPMJ/17.4102 ORIGINAL PROF-4102 PROSTATIC HYPERPLASIA; COMPARISON BETWEEN TAMSULOSIN AND TERAZOSIN FOR EFFICACY IN MEDICAL MANAGEMENT OF LOWER URINARY TRACT
The Professional Medical Journal DOI: 10.17957/TPMJ/17.4102 ORIGINAL PROF-4102 PROSTATIC HYPERPLASIA; COMPARISON BETWEEN TAMSULOSIN AND TERAZOSIN FOR EFFICACY IN MEDICAL MANAGEMENT OF LOWER URINARY TRACT
Chapter 2: Methodology
 Chapter 2: Methodology TABLE OF CONTENTS Introduction... 2 Study Selection and Data Abstraction... 2 Data Synthesis... 8 Guideline Development and Approvals... 9 Conflict of Interest... 10 Copyright 2010
Chapter 2: Methodology TABLE OF CONTENTS Introduction... 2 Study Selection and Data Abstraction... 2 Data Synthesis... 8 Guideline Development and Approvals... 9 Conflict of Interest... 10 Copyright 2010
Month/Year of Review: March 2014 Date of Last Review: April 2012
 Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Copyright 2012 Oregon State University. All Rights
Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Copyright 2012 Oregon State University. All Rights
Drug Class Literature Scan: Attention Deficit Hyperactivity Disorder
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-2596
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-2596
Abbreviated Class Review: Long-Acting Injectable Antipsychotics
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Single Technology Appraisal (STA) Tadalafil for the treatment of symptoms associated with benign prostatic hyperplasia
 Comment 1: the draft remit Appendix D - NICE s response to consultee and commentator comments on the draft scope and provisional matrix National Institute for Health and Clinical Excellence Single Technology
Comment 1: the draft remit Appendix D - NICE s response to consultee and commentator comments on the draft scope and provisional matrix National Institute for Health and Clinical Excellence Single Technology
Cialis. Cialis (tadalafil) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.06.02 Subject: Cialis Page: 1 of 5 Last Review Date: September 18, 2015 Cialis Description Cialis (tadalafil)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.06.02 Subject: Cialis Page: 1 of 5 Last Review Date: September 18, 2015 Cialis Description Cialis (tadalafil)
Prior Authorization Update: Nusinersen
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Combination Drug Therapy for Benign Prostatic Hyperplasia (BPH)
 The Annals of African Surgery www.sskenya.org Combination Drug Therapy for Benign Prostatic Hyperplasia (BPH) Author: Oliech J.S. FRCS, Affiliation: Department of Surgery, University of Nairobi. P.O. Box
The Annals of African Surgery www.sskenya.org Combination Drug Therapy for Benign Prostatic Hyperplasia (BPH) Author: Oliech J.S. FRCS, Affiliation: Department of Surgery, University of Nairobi. P.O. Box
Mini-Invasive Treatment in Urological Diseases Dott. Alberto Saita Responsabile Endourologia Istituto Clinico Humanitas - Rozzano
 Dipartimento di Urologia Direttore Prof. Giorgio Guazzoni Mini-Invasive Treatment in Urological Diseases Dott. Alberto Saita Responsabile Endourologia Istituto Clinico Humanitas - Rozzano alberto.saita@humanitas.it
Dipartimento di Urologia Direttore Prof. Giorgio Guazzoni Mini-Invasive Treatment in Urological Diseases Dott. Alberto Saita Responsabile Endourologia Istituto Clinico Humanitas - Rozzano alberto.saita@humanitas.it
Should flomax be taken at bedtime
 Should flomax be taken at bedtime Adults with radiopaque lower ureteral stones of 10 mm or smaller have received tamsulosin 0.4 mg PO at bedtime for 28 days or until definite stone passage (i.e. Prostate
Should flomax be taken at bedtime Adults with radiopaque lower ureteral stones of 10 mm or smaller have received tamsulosin 0.4 mg PO at bedtime for 28 days or until definite stone passage (i.e. Prostate
Drug Class Literature Scan: Antianginals
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Drug Effectiveness Review Project Summary Report Long acting Insulins
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Literature Scan: Anti-Parkinson s Agents
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
CONDUCT study A step further in changing treatment paradigm in BPH
 CONDUCT study A step further in changing treatment paradigm in BPH CLUJ,25.09.2015, Adriana Stoica, Medical Advisor GSK Toate informa,ile con,nute in aceasta prezentare sunt conforme cu Rezumatul caracteris,cilor
CONDUCT study A step further in changing treatment paradigm in BPH CLUJ,25.09.2015, Adriana Stoica, Medical Advisor GSK Toate informa,ile con,nute in aceasta prezentare sunt conforme cu Rezumatul caracteris,cilor
Cialis. Cialis (tadalafil) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.40.22 Subject: Cialis Page: 1 of 5 Last Review Date: September 15, 2017 Cialis Description Cialis (tadalafil)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.40.22 Subject: Cialis Page: 1 of 5 Last Review Date: September 15, 2017 Cialis Description Cialis (tadalafil)
Male Lower Urinary Tract Symptoms: Management in primary care and beyond. Daniel Cohen PhD FRCS(Urol) Consultant Urological Surgeon
 Male Lower Urinary Tract Symptoms: Management in primary care and beyond Daniel Cohen PhD FRCS(Urol) Consultant Urological Surgeon 1 LUTS Very common: 1/3 men over age of 50 have moderate to severe LUTS
Male Lower Urinary Tract Symptoms: Management in primary care and beyond Daniel Cohen PhD FRCS(Urol) Consultant Urological Surgeon 1 LUTS Very common: 1/3 men over age of 50 have moderate to severe LUTS
Class Update: Oral Antipsychotics
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
L ower urinary tract symptoms (LUTS) are a major health problem and are prevalent in men aged.45 years1 3.
 OPEN SUBJECT AREAS: UROGENITAL DISEASES DRUG DEVELOPMENT Received 23 October 2013 Accepted 15 January 2014 Published 4 February 2014 Correspondence and requests for materials should be addressed to P.H.
OPEN SUBJECT AREAS: UROGENITAL DISEASES DRUG DEVELOPMENT Received 23 October 2013 Accepted 15 January 2014 Published 4 February 2014 Correspondence and requests for materials should be addressed to P.H.
Medical Therapy for Benign Prostatic Hyperplasia Present and Future Impact
 REPORTS Medical Therapy for Benign Prostatic Hyperplasia Present and Future Impact Muta M. Issa, MD, MBA; Timothy S. Regan, BPharm, RPh, CPh Abstract The purpose of this manuscript is to provide clinicians,
REPORTS Medical Therapy for Benign Prostatic Hyperplasia Present and Future Impact Muta M. Issa, MD, MBA; Timothy S. Regan, BPharm, RPh, CPh Abstract The purpose of this manuscript is to provide clinicians,
Class Update: Tetracyclines
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Month/Year of Review: January 2012 Date of Last Review: September 2010
 Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-945-5220 Fax 503-947-1119 Month/Year of Review: January 2012 Date of Last Review:
Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-945-5220 Fax 503-947-1119 Month/Year of Review: January 2012 Date of Last Review:
Alpha-BlockerTherapy Can Be Withdrawn in the Majority of Men Following Initial CombinationTherapy with the Dual 5a-Reductase Inhibitor Dutasteride
 European Urology European Urology 44 (2003) 461 466 Alpha-BlockerTherapy Can Be Withdrawn in the Majority of Men Following Initial CombinationTherapy with the Dual 5a-Reductase Inhibitor Dutasteride J.
European Urology European Urology 44 (2003) 461 466 Alpha-BlockerTherapy Can Be Withdrawn in the Majority of Men Following Initial CombinationTherapy with the Dual 5a-Reductase Inhibitor Dutasteride J.
Benign Prostatic Hyperplasia (BPH) IPT VI Srikanth Kolluru, Ph.D
 Benign Prostatic yperplasia (BP) IPT VI Srikanth Kolluru, Ph.D. kolluru@tamhsc.edu, 361 221 0741 verview 1. Introduction and background of BP 2. Causes and common symptoms 3. Treatment options Learning
Benign Prostatic yperplasia (BP) IPT VI Srikanth Kolluru, Ph.D. kolluru@tamhsc.edu, 361 221 0741 verview 1. Introduction and background of BP 2. Causes and common symptoms 3. Treatment options Learning
ClinicalTrials.gov Identifier: sanofi-aventis. Sponsor/company:
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinicalTrials.gov
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinicalTrials.gov
Month/Year of Review: September 2013 Date of Last Review: February 2012
 Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Copyright 2012 Oregon State University. All Rights
Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119 Copyright 2012 Oregon State University. All Rights
Introduction. Original Article: Clinical Investigation
 International Journal of Urology (2015) 22, 582--587 doi: 10.1111/iju.12741 Original Article: Clinical Investigation Treatment satisfaction and clinically meaningful symptom improvement in men with lower
International Journal of Urology (2015) 22, 582--587 doi: 10.1111/iju.12741 Original Article: Clinical Investigation Treatment satisfaction and clinically meaningful symptom improvement in men with lower
Current drug management of BPH in primary care Claire Taylor MRCS, Charlotte Foley MRCS and Roger Kirby MA, MD, FRCS Urol
 Drug review BPH Current drug management of BPH in primary care Claire Taylor MRCS, Charlotte Foley MRCS and Roger Kirby MA, MD, FRCS Urol Skyline Imaging Ltd The range of drug treatments for BPH, alone
Drug review BPH Current drug management of BPH in primary care Claire Taylor MRCS, Charlotte Foley MRCS and Roger Kirby MA, MD, FRCS Urol Skyline Imaging Ltd The range of drug treatments for BPH, alone
Month/Year of Review: September 2012 Date of Last Review: September 2010
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
The Journal of International Medical Research 2005; 33:
 The Journal of International Medical Research 2005; 33: 562 573 A Comparative Study on the Safety and Efficacy of Tamsulosin and Alfuzosin in the Management of Symptomatic Benign Prostatic Hyperplasia:
The Journal of International Medical Research 2005; 33: 562 573 A Comparative Study on the Safety and Efficacy of Tamsulosin and Alfuzosin in the Management of Symptomatic Benign Prostatic Hyperplasia:
Benign Prostatic Hyperplasia (BPH)
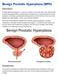 Benign Prostatic Hyperplasia (BPH) Definition Prostate gland enlargement is a common condition as men get older. Also called benign prostatic hyperplasia (BPH), prostate gland enlargement can cause bothersome
Benign Prostatic Hyperplasia (BPH) Definition Prostate gland enlargement is a common condition as men get older. Also called benign prostatic hyperplasia (BPH), prostate gland enlargement can cause bothersome
RESEARCH. Katrina Wilcox Hagberg, 1 Hozefa A Divan, 2 Rebecca Persson, 1 J Curtis Nickel, 3 Susan S Jick 1. open access
 open access Risk of erectile dysfunction associated with use of 5-α reductase inhibitors for benign prostatic hyperplasia or alopecia: population based studies using the Clinical Practice Research Datalink
open access Risk of erectile dysfunction associated with use of 5-α reductase inhibitors for benign prostatic hyperplasia or alopecia: population based studies using the Clinical Practice Research Datalink
REVIEW The role of combination medical therapy in benign prostatic hyperplasia
 (2008) 20, S33 S43 & 2008 Nature Publishing Group All rights reserved 0955-9930/08 $30.00 www.nature.com/ijir REVIEW The role of combination medical therapy in benign prostatic hyperplasia Department of
(2008) 20, S33 S43 & 2008 Nature Publishing Group All rights reserved 0955-9930/08 $30.00 www.nature.com/ijir REVIEW The role of combination medical therapy in benign prostatic hyperplasia Department of
Month/Year of Review: September 2012 Date of Last Review: September 2010
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Drug Class Review on Overactive Bladder Drugs
 Drug Class Review on Overactive Bladder Drugs Preliminary Scan Report #1 February 2014 Last Summary Review (June 2013) The Agency for Healthcare Research and Quality has not yet seen or approved this report
Drug Class Review on Overactive Bladder Drugs Preliminary Scan Report #1 February 2014 Last Summary Review (June 2013) The Agency for Healthcare Research and Quality has not yet seen or approved this report
Role of silodosin in patients with LUTS/BPE non responding to medical treatment with tamsulosin: a prospective, open-label, pilot study
 European Review for Medical and Pharmacological Sciences 2017; 21: 4941-4945 Role of silodosin in patients with LUTS/BPE non responding to medical treatment with tamsulosin: a prospective, open-label,
European Review for Medical and Pharmacological Sciences 2017; 21: 4941-4945 Role of silodosin in patients with LUTS/BPE non responding to medical treatment with tamsulosin: a prospective, open-label,
Therapeutic Class Overview Benign Prostatic Hyperplasia (BPH) Treatments
 Therapeutic Class Overview Benign Prostatic Hyperplasia (BPH) Treatments Therapeutic Class Overview/Summary: The agents approved for the treatment of signs and symptoms of benign prostatic hyperplasia
Therapeutic Class Overview Benign Prostatic Hyperplasia (BPH) Treatments Therapeutic Class Overview/Summary: The agents approved for the treatment of signs and symptoms of benign prostatic hyperplasia
Chapter 4: Research and Future Directions
 Chapter 4: Research and Future Directions Introduction Many of the future research needs listed in the 1994 Agency for Health Care Policy and Research (AHCPR) clinical practice guideline Benign Prostatic
Chapter 4: Research and Future Directions Introduction Many of the future research needs listed in the 1994 Agency for Health Care Policy and Research (AHCPR) clinical practice guideline Benign Prostatic
Recommended drug treatment of BPH in primary care Claire Taylor MRCS, Charlotte Foley MRCS and Roger Kirby MA, MD, FRCS Urol
 Drug review BPH Recommended drug treatment of BPH in primary care Claire Taylor MRCS, Charlotte Foley MRCS and Roger Kirby MA, MD, FRCS Urol Skyline Imaging Ltd The development of safe and effective drugs
Drug review BPH Recommended drug treatment of BPH in primary care Claire Taylor MRCS, Charlotte Foley MRCS and Roger Kirby MA, MD, FRCS Urol Skyline Imaging Ltd The development of safe and effective drugs
Date of Review: September 2016 Date of Last Review: September 2015
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Literature Scan: Triptans
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Benign Prostatic Hyperplasia. Shahideh Amini Pharm.D clinical pharmacy resident Tehran university of medical science Department of pharmacotherapy
 Benign Prostatic Hyperplasia Shahideh Amini Pharm.D clinical pharmacy resident Tehran university of medical science Department of pharmacotherapy Definition BPH is a common disorder that increases in frequency
Benign Prostatic Hyperplasia Shahideh Amini Pharm.D clinical pharmacy resident Tehran university of medical science Department of pharmacotherapy Definition BPH is a common disorder that increases in frequency
Index. urologic.theclinics.com. Note: Page numbers of article titles are in boldface type.
 Index Note: Page numbers of article titles are in boldface type. A Ablation, of prostate, holmium laser, 485 495 African prune tree (Pygeum africanum), 454 455 Alfuzosin, 445 446 Alpha-adrenergic agonists,
Index Note: Page numbers of article titles are in boldface type. A Ablation, of prostate, holmium laser, 485 495 African prune tree (Pygeum africanum), 454 455 Alfuzosin, 445 446 Alpha-adrenergic agonists,
REPORTS. Clinical and Economic Outcomes in Patients Treated for Enlarged Prostate
 Clinical and Economic Outcomes in Patients Treated for Enlarged Prostate Michael James Naslund, MD, MBA; Muta M. Issa, MD, MBA; Amy L. Grogg, PharmD; Michael T. Eaddy, PharmD, PhD; and Libby Black, PharmD
Clinical and Economic Outcomes in Patients Treated for Enlarged Prostate Michael James Naslund, MD, MBA; Muta M. Issa, MD, MBA; Amy L. Grogg, PharmD; Michael T. Eaddy, PharmD, PhD; and Libby Black, PharmD
IJBCP International Journal of Basic & Clinical Pharmacology
 Print ISSN: 2319-2003 Online ISSN: 2279-0780 IJBCP International Journal of Basic & Clinical Pharmacology DOI: http://dx.doi.org/10.18203/2319-2003.ijbcp20160772 Research Article Efficacy and safety of
Print ISSN: 2319-2003 Online ISSN: 2279-0780 IJBCP International Journal of Basic & Clinical Pharmacology DOI: http://dx.doi.org/10.18203/2319-2003.ijbcp20160772 Research Article Efficacy and safety of
Month/Year of Review: May 2014 Date of Last Review: September New Drug Evaluation: Sucrofferic Oxyhydroxide (Velphoro )
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Will Medical Management of Benign Prostatic Hyperplasia Result in Better or Worse Sexual Function in Men?
 Urol Sci 2011;22(1):14 18 MINI REVIEW Will Medical Management of Benign Prostatic Hyperplasia Result in Better or Worse Sexual Function in Men? Thomas I.S. Hwang 1,2,3 * 1 Shin Kong WHS Hospital, Taipei,
Urol Sci 2011;22(1):14 18 MINI REVIEW Will Medical Management of Benign Prostatic Hyperplasia Result in Better or Worse Sexual Function in Men? Thomas I.S. Hwang 1,2,3 * 1 Shin Kong WHS Hospital, Taipei,
Treating BPH: Comparing Rezum UroLift and HoLEP
 Treating BPH: Comparing Rezum UroLift and HoLEP Scott M. Cheney MD Mayo Clinic Arizona 2018 MFMER slide-1 Welcome to AZ 2018 MFMER slide-2 Outline Background on BPH, Rezum, Urolift, HoLEP AUA Guideline
Treating BPH: Comparing Rezum UroLift and HoLEP Scott M. Cheney MD Mayo Clinic Arizona 2018 MFMER slide-1 Welcome to AZ 2018 MFMER slide-2 Outline Background on BPH, Rezum, Urolift, HoLEP AUA Guideline
The Risk of Fracture with Taking Alpha Blockers for Treating Benign Prostatic Hyperplasia
 J Prev Med Public Health 2009;42(3):165-170 DOI: 103961/jpmph2009423165 The Risk of Fracture with Taking Alpha Blockers for Treating Benign Prostatic Hyperplasia Joongyub Lee 1) Nam-Kyoung Choi 13) Sun-Young
J Prev Med Public Health 2009;42(3):165-170 DOI: 103961/jpmph2009423165 The Risk of Fracture with Taking Alpha Blockers for Treating Benign Prostatic Hyperplasia Joongyub Lee 1) Nam-Kyoung Choi 13) Sun-Young
Prostatic Urethral Lift
 Protocol Prostatic Urethral Lift (701151) Medical Benefit Effective Date: 04/01/18 Next Review Date: 01/19 Preauthorization No Review Dates: 05/17, 01/18 Preauthorization is not required. The following
Protocol Prostatic Urethral Lift (701151) Medical Benefit Effective Date: 04/01/18 Next Review Date: 01/19 Preauthorization No Review Dates: 05/17, 01/18 Preauthorization is not required. The following
Therapeutic Strategies for Managing BPH Progression
 european urology supplements 5 (2006) 997 1003 available at www.sciencedirect.com journal homepage: www.europeanurology.com Therapeutic Strategies for Managing BPH Progression John M. Fitzpatrick a, *,
european urology supplements 5 (2006) 997 1003 available at www.sciencedirect.com journal homepage: www.europeanurology.com Therapeutic Strategies for Managing BPH Progression John M. Fitzpatrick a, *,
MMM. Topic The use of Tadalafil 5mg daily for the treatment of BPH-LUTS
 Dr Tan & Partners MMM Vol. 1 No. 1 Morbidity & Mortality Meeting 14 th November 2014 Introduction Topic The use of Tadalafil 5mg daily for the treatment of BPH-LUTS Tadalafil 5mg daily is a well established
Dr Tan & Partners MMM Vol. 1 No. 1 Morbidity & Mortality Meeting 14 th November 2014 Introduction Topic The use of Tadalafil 5mg daily for the treatment of BPH-LUTS Tadalafil 5mg daily is a well established
Literature Scan: NSAIDs
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
OHSU Drug Effectiveness Review Project Summary Report Benzodiazepines
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
