Bone Morphogenetic Protein. Description
|
|
|
- Todd Wilson
- 6 years ago
- Views:
Transcription
1 Subject: Bone Morphogenetic Protein Page: 1 of 13 Last Review Status/Date: March 2015 Bone Morphogenetic Protein Description Two recombinant human bone morphogenetic proteins (rhbmps) are now commercially available, rhbmp-2, applied with an absorbable collagen sponge (InFUSE, Medtronic, Memphis, TN) and rhbmp-7, applied in putty (OP-1 ). These products have been investigated as an alternative to bone autografting in a variety of clinical situations, including spinal fusions, internal fixation of fractures, treatment of bone defects, and reconstruction of maxillofacial conditions. Background Bone morphogenetic proteins (BMPs) are members of the family of transforming growth factors. At present, some 20 different BMPs have been identified, all with varying degrees of tissue stimulating properties. RhBMPs are delivered to the bone grafting site as part of a surgical procedure; a variety of carrier and delivery systems has been investigated. Carrier systems, which are absorbed over time, function to maintain the concentration of the rhbmp at the treatment site; provide temporary scaffolding for osteogenesis; and prevent extraneous bone formation. Carrier systems have included inorganic material, synthetic polymer, natural polymers, and bone allograft. The rhbmp and carrier may be inserted via a delivery system, which may also function to provide mechanical support. The carrier and delivery system are important variables in the clinical use of rhbmps, and different clinical applications, such as long bone nonunion, or interbody or intertransverse fusion, have been evaluated with different carriers and delivery systems. For example, rhbmp putty with pedicle and screw devices are used for instrumented intertransverse fusion (posterolateral fusion; PLF), while rhbmp in a collagen sponge with bone dowels or interbody cages are used for interbody spinal fusion. In addition, interbody fusion of the lumbar spine can be approached from an anterior (anterior lumbar interbody fusion; ALIF), lateral (XLIF), or posterior direction (PLIF or TLIF); see appendix. Surgical procedures may include decompression of the spinal canal and insertion of pedicle screws and rods to increase stability of the spine. Posterior approaches (PLIF and TLIF) allow decompression (via laminotomies and facetectomies) for treatment of spinal canal pathology (e.g., spinal stenosis, lateral recess and foraminal stenosis, synovial cysts, hypertrophic ligamentum flavum) along with stabilization of the spine and are differentiated from instrumented or noninstrumented posterolateral intertransverse fusion (PLF), which involves the transverse processes. Due to the proximity of these procedures to the spinal canal, risks associated with ectopic bone formation are increased (e.g., radiculopathies). Increased risk of bone
2 Subject: Bone Morphogenetic Protein Page: 2 of 13 resorption around rhbmp grafts, heterotopic bone formation, epidural cyst formation, and seromas has also been postulated. Regulatory Status At the present time, two rhbmps and associated carrier/delivery systems have received approval from the U.S. Food and Drug Administration (FDA). The InFUSE system consists of rhbmp-2 on an absorbable collagen sponge carrier. The labeled indications for these devices are summarized here. OP-1 consists of rhbmp-7 and bovine collagen, which is reconstituted with saline to form a paste. The addition of carboxymethylcellulose forms putty. 1. InFUSE Bone Graft in conjunction with 1 of 2 interbody fusion devices, i.e., either the LT-Cage Lumbar Tapered Fusion Device or the Inter Fix RP Threaded Fusion device. This device received FDA approval through the premarket approval (PMA) process: The device is indicated for spinal fusion procedures in skeletally mature patients with degenerative disc disease (DDD) at 1 level from L2-S1. DDD is defined as discogenic back pain with degeneration of the disc confirmed by patient history, function deficit, and/or neurologic deficit and radiographic studies. These DDD patients may also have up to grade I spondylolisthesis at the involved level or retrolisthesis. The InFUSE Bone Graft/LT-CAGE devices are to be implanted via an anterior open or a laparoscopic approach. The InFUSE Bone Graft/INTER FIX Threaded Fusion Device; and InFUSE Bone Graft/INTER FIX RP Threaded Fusion Device are to be implanted via an anterior open approach only. Patients receiving the InFUSE Bone Graft/Interbody Fusion Device should have had at least 6 months of nonoperative treatment prior to treatment with the InFUSE Bone Graft/Interbody Fusion Device. (Note: A collagen sponge consists of the carrier, while the interbody fusion device is a delivery system. Use with posterior or transforaminal lumbar interbody fusion is considered offlabel.) For the treatment of acute, open fractures of the tibial shaft For sinus augmentations, and for localized alveolar ridge augmentations for defects associated with extraction sockets (P050053, March 2007) 2. OP-1 (Stryker Biotech, Hopkinton, MA) has received 2 FDA approvals through the Humanitarian Device Exemption (HDE) process. HDE is available to devices intended for fewer than 4,000 patients per year. As part of this process, the manufacturer is not required to demonstrate unequivocal benefit but only probable benefit. OP-1 received the following labeled indications: OP-1 Implant is indicated for use as an alternative to autograft in recalcitrant long bone nonunions where use of autograft is unfeasible and alternative treatments have failed. OP-1 Putty is indicated for use as an alternative to autograft in compromised patients requiring revision posterolateral (intertransverse) lumbar spinal fusion, for which autologous bone and bone marrow harvest are not feasible or are not expected to promote fusion. Examples of compromising factors include osteoporosis, smoking and diabetes. Stryker Biotech recently sought FDA permission to expand use of OP-1 Putty to include use in uninstrumented posterolateral lumbar spinal fusion for the treatment of lumbar spondylolithesis. In
3 Subject: Bone Morphogenetic Protein Page: 3 of 13 March 2009, an FDA advisory committee voted 6-1 against recommending the expanded approval. Olympus Biotech Corp., a subsidiary of Olympus Corp., acquired OP-1 assets in In 2014, Olympus Corp. closed Olympus Biotech operations in the United States and discontinued sales of the Olympus Biotech products in this country. Both OP-1 and InFUSE Bone Graft/LT-Cage Lumbar Tapered Fusion device are contraindicated in patients who are pregnant, may be allergic to any of the materials contained in the devices, have an infection near the area of the surgical incision, have had a tumor removed from the area of the implantation site or currently have a tumor in that area, or who are skeletally immature. In July 2008, the FDA issued a public health notification regarding life-threatening complications associated with recombinant human bone morphogenetic protein in cervical spine fusion. The FDA had received reports of complications with the use of rhbmp in cervical spine fusion. (1) These complications were associated with swelling of neck and throat tissue, which resulted in compression of the airway and/or neurologic structures in the neck. Some reports described difficulty swallowing, breathing, or speaking. Severe dysphagia following cervical spine fusion using rhbmp products has also been reported in the literature. As stated in the public health notification, the safety and effectiveness of rhbmp in the cervical spine have not been demonstrated, and these products are not approved by the FDA for this use. In 2011 Medtronic received a nonapprovable letter from the FDA for AMPLIFY. The AMPLIFY rhbmp-2 Matrix utilizes a higher dose of rhbmp (2.0 mg/ml) with a compression-resistant carrier and is being evaluated for posterolateral fusion of single level lumbar (L2-S1) degenerative disc disease. Related Policies Ultrasound Accelerated Fracture Healing Device Recombinant and Autologous Platelet-Derived Growth Factors as a Treatment of Wound Healing and Other Conditions Electrical Bone Growth Stimulation of the Appendicular Skeleton Electrical Stimulation of the Spine as an Adjunct to Spinal Fusion Procedures Policy *This policy statement applies to clinical review performed for pre-service (Prior Approval, Precertification, Advanced Benefit Determination, etc.) and/or post-service claims. Use of recombinant human bone morphogenetic protein-2 (rhbmp-2, InFUSE) may be considered medically necessary in skeletally mature patients: For anterior lumbar interbody fusion procedures when use of autograft is unfeasible. For instrumented posterolateral intertransverse spinal fusion procedures when use of autograft is unfeasible. For the treatment of acute, open fracture of the tibial shaft, when use of autograft is unfeasible.
4 Subject: Bone Morphogenetic Protein Page: 4 of 13 Use of recombinant human bone morphogenetic protein-7 (rhbmp-7, OP-1) may be considered medically necessary in skeletally mature patients: As an alternative to autograft in compromised patients (e.g., osteoporosis, tobacco use, or diabetes) requiring noninstrumented revision posterolateral intertransverse lumbar spinal fusion, for whom autologous bone and bone marrow harvest are not feasible or are not expected to promote fusion.* For recalcitrant long-bone nonunions where use of autograft is unfeasible and alternative conservative treatments have failed.* Bone morphogenetic protein (rhbmp-2 or rhbmp-7) is considered not medically necessary for all other indications, including but not limited to spinal fusion when use of autograft is feasible. *FDA approved under a Humanitarian Device Exemption (HDE). OP-1 is no longer sold in the United States. Policy Guidelines Use of iliac crest bone graft (ICBG) may be considered unfeasible due to situations that may include, but are not limited to, prior harvesting of ICBG or need for a greater quantity of ICBG than available (e.g., for multi-level fusion). There is not a consensus for the definition of nonunions. One proposed definition is failure of progression of fracture healing for at least 3 consecutive months (and at least 6 months following the fracture) accompanied by clinical symptoms of delayed/nonunion (pain, difficulty weight bearing).(2) The following patient selection criteria are described for policies No (ultrasound) and (electrical stimulation) in the treatment of nonunions: At least 3 months have passed since the date of the fracture, AND serial radiographs have confirmed that no progressive signs of healing have occurred, AND the fracture gap is 1 cm or less, AND the patient can be adequately immobilized and is of an age when he/she is likely to comply with non-weight bearing. A recalcitrant nonunion would thus be considered to be a nonunion with a larger fracture gap (e.g., greater than 1 cm) or a nonunion that has persisted for a longer duration of time with no response to conservative treatment (e.g., 3 months of ultrasound or electrical stimulation). Rationale At the time of the writing of this policy, randomized clinical trials supported the use of recombinant human bone morphogenetic protein-2 (rhbmp-2) in the treatment of interbody spinal fusion when used in conjunction with a tapered cage and also in the treatment of open tibial fractures. (3) In addition, a randomized study supported the use of rhbmp-7 in the treatment of recalcitrant nonunions of the long bones. (4) It should be noted that the majority of trials were designed to show that use of rhbmp is
5 Subject: Bone Morphogenetic Protein Page: 5 of 13 equivalent (not superior) to autologous bone grafting. Although the proposed advantage of rhbmp is the elimination of a separate incision site required for harvesting of autologous bone graft and the associated pain and morbidity secondary to this procedure, a 2011 study by Howard and colleagues raises questions about the magnitude of pain observed with iliac crest bone graft (ICBG) harvesting. (5) In this study, 112 patients who had an instrumented posterolateral lumbar fusion at 1 or 2 levels were seen at a tertiary spine center for a routine postoperative visit. Iliac crest bone graft was harvested in 53 patients (47.3%) through the midline incision used for lumbar fusion and recombinant human bone morphogenetic protein (rhbmp-2) was used in 59 patients (52.7%) with no graft harvest. An independent investigator who was not directly involved in the care of the patient and was unaware of the type of bone graft used in the fusion examined the patient for tenderness over the surgical site, as well as the left and right posterior iliac crest. At a mean follow-up of 41 months (range months), there was no significant difference between the groups in the proportion of patients complaining of tenderness over either iliac crest (3.8 vs. 3.6 on a 10-point scale). While 54% of patients complained of tenderness over one or both iliac crests, only 10 patients (9% of 112) had pain over the same crest from which the graft was harvested (mean pain score of 4.4). Spinal Fusion In 2013, 2 systematic reviews on the effectiveness and harms of recombinant human bone morphogenetic protein-2 (rhbmp-2) in spine fusion were published. (6, 7) These 2 systematic reviews of patient-level data followed a 2011 U.S. Senate investigation of industry influence on Infuse clinical studies and a systematic review by Carragee and colleagues of emerging safety concerns with rhbmp-2. (8, 9) The systematic review by Carragee et al. compared conclusions regarding safety and efficacy from the 13 published rhbmp-2 industry-sponsored trials with available U.S. Food and Drug Administration (FDA) data summaries, subsequent studies, and databases. Evaluation of the original trials suggested methodologic bias against the control group in the study design (discarding local bone graft and failure to prepare facets for arthrodesis) and potential bias (overestimation of harm) in the reporting of iliac crest donor site pain. Comparison between the published studies and FDA documents revealed internal inconsistencies and adverse events that were not reported in the published articles. Both of the 2013 studies conducted meta-analyses on individual patient data, both published and unpublished, that was provided by the manufacturer through the Yale University Open Data Access (YODA) Project. One meta-analysis was conducted by Simmonds and colleagues from the University of York in the United Kingdom; the other was by Fu and colleagues from the Oregon Health and Science University. The meta-analysis by Simmonds et al. included patient-level data from 12 randomized controlled trials (RCTs, n=1,408), regardless of spinal level or surgical approach, and adverse event data from an additional 35 observational studies. (6) rhbmp-2 increased the rate of radiographic fusion by 12% compared to ICBG, with substantial heterogeneity across trials. A small improvement in the Oswestry Disability Index (ODI, 3.5 percentage points) did not reach the previously defined threshold for a clinically significant effect. The review also found a small improvement in back pain (1 point on a 20- point scale) and SF-36 physical component score (PCS, 1.9 percentage points). There was no significant difference between the groups for leg pain. There was a potential for bias in the pain and
6 Subject: Bone Morphogenetic Protein Page: 6 of 13 functional outcomes since outcomes were patient-reported and patients were not blinded to the treatment received. Overall, the increase in successful fusion at up to 24 months did not appear to be associated with a clinically significant reduction in pain. The meta-analysis by Fu et al. included individual-patient data from 13 RCTs (n=1,981) and 31 cohort studies. (7) The review found moderate evidence of no consistent differences between rhbmp-2 and ICBG in overall success, fusion rates, or other effectiveness measures for anterior lumbar interbody fusion (ALIF) or posterolateral fusion (PLF). A small RCT and 3 cohort studies revealed no difference in effectiveness outcomes between rhbmp and ICBG for anterior cervical fusion. Reporting in the original published trials was found to be biased, with journal publications selecting analyses and results that favored rhbmp over ICBG. Both studies found that cancer risk may be increased with rhbmp-2, although the number of events was low and there was heterogeneity in the types of cancer. In the Simmonds analysis, combined analysis revealed a relative risk of 1.84 for cancer in the BMP group, but this increased rate did not reach statistical significance (95% CI: ). Fu et al. performed a combined analysis of cancer incidence at 24 months and 48 months post-treatment. At 24 months, there was a significant increase in cancer for the BMP group (risk ratio [RR]: 3.45, 95% CI: ), and at 48 months, there was a smaller increase that did not reach statistical significance (RR: 1.82, 95% CI: ). Other adverse events were also increased for the BMP group. Simmonds et al. found a higher incidence of early back and leg pain with rhbmp-2 in the analysis of patient-level data The studies consistently reported increased rates of heterotopic bone formation, leg pain/radiculitis, osteolysis and dysphagia, but combined analysis for these outcomes was not performed. The Fu study reported that BMP-2 was associated with a nonsignificantly increased risk for urogenital problems when used for anterior lumbar fusion and an increased risk for wound complications and dysphagia when used for anterior cervical spine fusion. Fu et al. documented that the information on adverse events in the published literature was incomplete in comparison to the total amount of information available. Off-label use of BMP can include multiple levels and dosages greater than the FDA-approved dose of rhbmp-2 for single-level fusion. In 2013, Carragee et al. assessed cancer risk after high-dose rhbmp- 2 (40 mg) using publicly available data from the pivotal, multicenter, randomized controlled trial of AMPLIFY (n=463).(10) The study found an increase in the incidence of cancer, a reduction in the time to first cancer, and a greater number of patients with multiple cancers. For example, at 2 years there were 15 new cancer events in 11 patients in the rhbmp-2 group compared with 2 new cancer events in 2 patients treated with autogenous bone graft, with an incidence rate ratio of When calculated in terms of the number of patients with one or more cancer events 2 years after surgery, the incidence rate per 100 person-years was 2.54 in the rhbmp-2 group compared with 0.50 in the control group, and the incidence rate ratio was The mean time to development of cancer was 17.5 months after use of rhbmp-2 compared with 31.8 months in the controls. Three patients in the rhbmp-2 group and none in the control group developed multiple new cancers.
7 Subject: Bone Morphogenetic Protein Page: 7 of 13 Long-Bone Fractures and Nonunions A 2010 Cochrane review evaluated the effectiveness and costs of rhbmp on fracture healing in acute fractures and nonunions compared with standards of care. (11) The literature was searched to October 2008, and 11 RCTs (976 participants) and 4 economic evaluations were included in the review. The times to fracture healing were comparable between the rhbmp and control groups. There was some evidence for increased healing rates, mainly for open tibial fractures without secondary procedures (risk ratio [RR]: 1.19). Three trials indicated that fewer secondary procedures were required for acute fractures treated with rhbmp (RR: 0.65). The authors concluded that limited evidence suggests that rhbmp may be more effective than standard of care for acute tibial fracture healing; however, the use of rhbmp for treating nonunion remains unclear (RR: 1.02). In 2014, Lyon et al reported a manufacturer-funded randomized double-blind trial of injectable rhbmp-2 in a calcium phosphate matrix for closed tibial diaphyseal fractures. (12) The study had a target enrollment of 600 patients but was stopped after interim analysis with 387 patients enrolled. Addition of the injectable rhbmp-2 paste to the standard of reamed intramedullary nail fixation did not shorten the time to fracture healing, resulting in the study termination due to futility. Oral and Maxillofacial Procedures The 2010 AHRQ technology assessment on the state of the evidence of on-label and off-label use of rhbmp (13) included the following conclusions: The strength of the body of evidence on clinical outcomes is moderate that rhbmp-2 does not provide an advantage in prosthesis implantation and functional loading compared to autograft plus allograft bone. There is also moderate evidence that oral sensory loss associated with autograft bone harvest can be avoided by use of rhbmp2. Through April 30, 2011, the FDA s Manufacturer and User Facility Device Experience (MAUDE) received 83 reports of adverse events involving rhbmp-2 in oral and maxillofacial operations. (14) rhbmp-2 was used off-label in 66.3% of these cases and included reconstruction of the mandible after fracture or cancer and alveolar cleft repair. The most frequently reported adverse events were local edema/pain, surgical site infections/wound complications, and graft failure. Overall, the evidence does not support a health benefit of rhbmp in oral and maxillofacial procedures. Additional Applications There has been research interest in the following applications: management of early stages of osteonecrosis of the vascular head, as an adjunct to hip arthroplasty to restore bone defects in the acetabulum or femoral shaft, and as an adjunct to distraction osteogenesis (i.e., Ilizarov procedure). (13, 14) The literature regarding these applications consists of small case series; no controlled trials have been identified.
8 Subject: Bone Morphogenetic Protein Page: 8 of 13 Ongoing Clinical Trials A search of online site clinicaltrials.gov in July 2013 identified several ongoing studies. Of particular interest is an industry-sponsored Phase II randomized controlled dose finding study of intra-articular BMP-7 for osteoarthritis of the knee (NCT ). The study lists an enrollment of 355 subjects and is described as completed as of January As of July 2013, no publications from this study have been identified. Practice Guidelines and Position Statements Guidelines on lumbar spinal fusion from the American Association of Neurological Surgeons (AANS) and the Congress of Neurological Surgeons (CNS) were updated in AANS/CNS gave a grade B recommendation (multiple level II studies) for the use of rhbmp-2 as a substitute for autologous iliac crest bone for anterior lumbar interbody fusion and single-level posterolateral instrumented fusion. Grade C recommendations were made for rhbmp-2 as an option for PLIF and TLIF, posterolateral fusion in patients older than 60 years, and as a graft extender for either instrumented or noninstrumented posterolateral fusions. AANS/CNS also gave a grade C recommendation (based on multiple level IV and V studies) that the use of rhbmp-2 as a graft option has been associated with a unique constellation of complications of which the surgeon should be aware when considering the use of this graft extender/substitute. U.S. Preventive Services Task Force Recommendations Not applicable. Summary In 2013, 2 systematic reviews on recombinant human bone morphogenetic protein-2 (rhbmp-2) that used manufacturer-provided individual patient data were published. Overall, these systematic reviews found little to no benefit of rhbmp-2 over iliac crest bone graft for spinal fusion, with an uncertain risk of harm. The small benefits reported do not support the widespread use of rhbmp-2, but do leave the possibility that rhbmp-2 may lead to clinically significant improvements in selected subgroups, such as patients in whom use of iliac crest bone graft (ICBG) is unfeasible and have a high risk of fusion failure. While there was a low adverse event rate overall, concerns remain about the possibility of increased adverse event rates with rhbmp-2, including cancer. Based on this new evidence, it is not possible to conclude that the small benefits of rhbmp-2 outweigh the risks. Therefore, rhbmp-2 is considered to be not medically necessary when use of ICBG is feasible. In cases where use of ICBG is not feasible, such as when previous bone harvest has been performed, the benefit of rhbmp in promoting fusion will likely outweigh the adverse effects, and therefore rhbmp-2 may be considered medically necessary. The U.S. Food and Drug Administration s humanitarian device exemptions (HDE) for rhbmp-7 state that use is restricted to patients in whom autologous bone and bone marrow harvest are not feasible or are not expected to promote to promote fusion. Therefore, the policy on rhbmp-7 remains unchanged. As of 2014, rhbmp-7 is no longer marketed in the United States.
9 Subject: Bone Morphogenetic Protein Page: 9 of 13 Use of rhbmp has not been shown to be as beneficial as the established alternative (iliac crest bone graft, ICBG) and evidence is insufficient to permit conclusions concerning the effect of rhbmp and is considered investigational for other indications, including (but not limited to): Cervical spinal fusions Posterior or transforaminal lumbar interbody spinal fusion (considered investigational because of safety concerns related to ectopic bone formation in the spinal canal); Treatment of noninstrumented posterolateral intertransverse spinal fusion when autograft is feasible and expected to promote fusion; An alternative or adjunct to bone grafting in other locations, (considered investigational due to the lack of controlled trials to support it efficacy). Medicare National Coverage The Centers for Medicare and Medicaid Services (CMS) has established an add-on to the diagnosisrelated group (DRG) payment to cover a portion of the cost of new technologies during the 2-year period before charge data for the technologies are incorporated into the DRG weights. To qualify, a technology must be new, must provide verifiable improvement in the treatment or diagnosis of beneficiaries, and the mean standardized charge for treatment using the new technology must be at least 1 standard deviation above the mean standardized charge for treating the same case without the new technology. In 2004, CMS concluded that the InFUSE Bone Graft/LT-CAGE met these criteria and will receive an add-on payment to DRGs 497 or 498. Medtronic, the manufacturer of the InFUSE device, has applied for a new technology add-on payment for the FDA-approved indication of treatment of open acute fractures of the tibial shaft. References 1. U.S. Food and Drug Administration FDA Public Health Notification: Life-threatening Complications Associated with Recombinant Human Bone Morphogenetic Protein in Cervical Spine Fusion. Available online at: htm Last accessed September Bhandari M, Fong K, Sprague S et al. Variability in the definition and perceived causes of delayed unions and nonunions: a cross-sectional, multinational survey of orthopaedic surgeons. J Bone Joint Surg Am 2012; 94(15):e U.S. Food and Drug Administration. Summary of Safety and Effectiveness. InFUSE Bone Graft/LT-Cage Lumbar Tapered Fusion Device. Available online at: Last accessed November Govender S, Csimma C, Genant HK et al. Recombinant human bone morphogenetic protein-2 for treatment of open tibial fractures: a prospective, controlled, randomized study of four hundred and fifty patients. J Bone Joint Surg Am 2002; 84-A (12): Howard JM, Glassman SD, Carreon LY. Posterior iliac crest pain after posterolateral fusion with or without iliac crest graft harvest. Spine J 2011; 11(6):534-7.
10 Subject: Bone Morphogenetic Protein Page: 10 of Simmonds MC, Brown JV, Heirs MK et al. Safety and Effectiveness of Recombinant Human Bone Morphogenetic Protein-2 for Spinal Fusion: A Meta-analysis of Individual-Participant Data. Ann Intern Med 2013; 158(12): Fu R, Selph S, McDonagh M et al. Effectiveness and Harms of Recombinant Human Bone Morphogenetic Protein-2 in Spine Fusion: A Systematic Review and Meta-analysis. Ann Intern Med 2013; 158(12): Carragee EJ, Hurwitz EL, Weiner BK. A critical review of recombinant human bone morphogenetic protein-2 trials in spinal surgery: emerging safety concerns and lessons learned. Spine J 2011; 11(6): United States Senate Finance Committee. Staff report on Medtronic's influence on INFUSE clinical studies. Int J Occup Environ Health 2013; 19(2): Carragee EJ, Chu G, Rohatgi R et al. Cancer risk after use of recombinant bone morphogenetic protein-2 for spinal arthrodesis. J Bone Joint Surg Am 2013; 95(17): Garrison KR, Shemilt I, Donell S et al. Bone morphogenetic protein (BMP) for fracture healing in adults. Cochrane Database Syst Rev 2010; (6):CD Lyon T, Scheele W, Bhandari M, et al. Efficacy and safety of recombinant human bone morphogenetic protein-2/calcium phosphate matrix for closed tibial diaphyseal fracture: a double-blind, randomized, controlled phase- II/III trial. J Bone Joint Surg Am. Dec ;95(23): PMID Ratko T BS, Samson D, et al Bone Morphogenetic Protein: The State of the Evidence of On- Label and Off-Label Use. AHRQ Technology Assessment Reports 2010; Accessed October, Woo EJ. Adverse events reported after the use of recombinant human bone morphogenetic protein 2. J Oral Maxillofac Surg 2012; 70(4): Valentin-Opran A, Wozney J, Csimma C et al. Clinical evaluation of recombinant human bone morphogenetic protein-2. Clin Orthop Relat Res 2002; 395: Ratko T BS, Samson D, et al Bone Morphogenetic Protein: The State of the Evidence of On- Label and Off-Label Use. AHRQ Technology Assessment Reports 2010; Accessed October, Kaiser MG, Groff MW, Watters WC, 3rd, et al. Guideline update for the performance of fusion procedures for degenerative disease of the lumbar spine. Part 16: bone graft extenders and substitutes as an adjunct for lumbar fusion. J Neurosurg Spine. 2014; 21(1): Accessed October 6, Policy History Date Action Reason June 2012 New Policy March 2013 Update Policy Policy updated with literature review; references added; policy statements unchanged. March 2014 Update Policy Policy updated with literature review, one FDA-approved indication that had been omitted was reinserted: Treatment of tibial shaft with BMP-2 (when autograft is unfeasible added); return to use of FDA language regarding treatment of noninstrumented revision
11 Subject: Bone Morphogenetic Protein Page: 11 of 13 posterolateral intertransverse lumbar spinal fusion with BMP-7 where use of autograft is unfeasible. March 2015 Update Policy Policy updated with literature review through October 6, 2014;references 12 and 17 added; policy statements unchanged. Appendix added. Keywords Bone Morphogenetic Protein Infuse, Bone Morphogenetic Protein OP-1, Bone Morphogenetic Protein BMP, BMP-2, BMP-7 InFuse Recombinant human bone morphogenetic protein This policy was approved by the FEP Pharmacy and Medical Policy Committee on March 20, 2015 and is effective April 15, Signature on file Deborah M. Smith, MD, MPH
12 Subject: Bone Morphogenetic Protein Page: 12 of 13 Appendix Procedures used for lumbar interbody fusion differ primarily in the direction of approach to the spine, ie, from the front (anterior), from the back (posterior or transforaminal) or from the side (lateral). An alternative approach to interbody fusion is arthrodesis of the transverse processes alone (posterolateral), which does not fuse the adjoining vertebral bodies. Circumferential fusion fuses both the adjacent vertebral bodies and the transverse processes, typically using both an anterior and posterior approach to the spine. Open and Minimally Invasive Approaches to Lumbar Interbody Fusion Procedure Access Approach Visualization Anterior (ALIF) Open, MI, or laparoscopic Transperitoneal or retroperitoneal Direct, endoscopic or laparoscopic with fluoroscopic guidance Posterior (PLIF) Open or MI Incision centered over spine with laminectomy/laminotomy and retraction of nerve Transforaminal (TLIF) Open or MI Offset from spine, through the intervertebral foramen via unilateral facetectomy Lateral Extreme lateral (XLIF) Direct lateral (DLIF) MI Retroperitoneal through transpsoas Direct, endoscopic or microscopic, with fluoroscopic guidance Direct, endoscopic or microscopic, with fluoroscopic guidance Direct, with neurologic monitoring and fluoroscopic guidance LIF: lumbar interbody fusion; MI: minimally invasive. Anterior Lumbar Interbody Fusion Anterior access provides direct visualization of the disc space, potentially allowing a more complete discectomy and better fusion than lateral or posterior approaches. An anterior approach avoids trauma to the paraspinal musculature, epidural scarring, traction on nerve roots, and dural tears. However, the retraction of the great vessels, peritoneal contents, and superior hypogastric sympathetic plexus with a peritoneal or retroperitoneal approach place these structures at risk of iatrogenic injury. Access to the posterior space for the treatment of nerve compression is also limited. Laparoscopic anterior lumbar interbody fusion has also been investigated.
13 Subject: Bone Morphogenetic Protein Page: 13 of 13 Posterior Lumbar Interbody Fusion Posterior lumbar interbody fusion (PLIF) can be performed through either a traditional open procedure with a midline incision or with a minimally invasive approach using bilateral paramedian incisions. In the open procedure, the midline muscle attachments are divided along the central incision to facilitate wide muscle retraction and laminectomy. In minimally invasive PLIF, tubular retractors may be used to open smaller central bilateral working channels to access the pedicles and foramen. Minimally invasive PLIF typically involves partial laminotomies and facetectomies. The decompression allows treatment of spinal canal pathology (e.g., spinal stenosis, lateral recess and foraminal stenosis, synovial cysts, hypertrophic ligamentum flavum), as well as stabilization of the spine through interbody fusion. Transforaminal Lumbar Interbody Fusion Transforaminal lumbar interbody fusion (TLIF) is differentiated from the more traditional bilateral PLIF by a unilateral approach to the disc space through the intervertebral foramen. In minimally invasive TLIF, a single incision about 2 to 3 cm in length is made approximately 3 cm lateral to the midline. A tubular retractor is docked on the facet joint complex and a facetectomy with partial laminectomy is performed. Less dural retraction is needed with access through the foramen via unilateral facetectomy, and contralateral scar formation is eliminated. TLIF provides access to the posterior elements along with the intervertebral disc space. Lateral Interbody Fusion Lateral interbody fusion (e.g., extreme lateral interbody fusion or direct lateral interbody fusion) uses specialized retractors in a minimally invasive, lateral approach to the anterior spine through the psoas. In comparison with anterior lumbar interbody fusion, the lateral approach does not risk injury to the peritoneum or great vessels. However, exposure to the spine may be more limited, and dissection of the psoas major places the nerves of the lumbar plexus at risk. Electromyographic monitoring and dissection predominantly within the anterior psoas major may be utilized to reduce the risk of nerve root injury. These various factors decrease the ability to perform a complete discectomy and address pathology of the posterior elements. Circumferential Fusion Circumferential fusion is 360 processes, typically through an anterior and posterior approach. bodies fusion and that transverse joins vertebrae by their entire Posterolateral Fusion Posterolateral fusion is a procedure where the transverse processes of the involved segments are decorticated and covered with a mixture of bone autograft or allograft.
Medical Policy Bone Morphogenetic Protein Section 7.0 Surgery. Description. Related Policies. Policy
 7.01.100 Bone Morphogenetic Protein Section 7.0 Surgery Effective Date January 30, 2015 Subsection 7.01 Surgery Original Policy Date March 1, 2005 Next Review Date January 2016 Description Two recombinant
7.01.100 Bone Morphogenetic Protein Section 7.0 Surgery Effective Date January 30, 2015 Subsection 7.01 Surgery Original Policy Date March 1, 2005 Next Review Date January 2016 Description Two recombinant
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: bone_morphogenetic_protein 10/2014 2/2018 2/2019 2/2018 Description of Procedure or Service Bone morphogenetic
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: bone_morphogenetic_protein 10/2014 2/2018 2/2019 2/2018 Description of Procedure or Service Bone morphogenetic
Bone Morphogenetic Protein
 Bone Morphogenetic Protein Policy Number: 7.01.100 Last Review: 12/2017 Origination: 5/2006 Next Review: 5/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage for bone
Bone Morphogenetic Protein Policy Number: 7.01.100 Last Review: 12/2017 Origination: 5/2006 Next Review: 5/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage for bone
Original Policy Date
 MP 7.01.80 Bone Morphogenetic Protein Medical Policy Section Surgery Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search/12/2013 Return to Medical Policy
MP 7.01.80 Bone Morphogenetic Protein Medical Policy Section Surgery Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search/12/2013 Return to Medical Policy
January 2015 Review: January 2017
 BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE (JPC) January 2015 Review: January 2017 Bulletin 209: Bone Morphogenetic Proteins (BMP) JPC Recommendations: To support the East of England Priorities
BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE (JPC) January 2015 Review: January 2017 Bulletin 209: Bone Morphogenetic Proteins (BMP) JPC Recommendations: To support the East of England Priorities
Facet Arthroplasty. Policy Number: Last Review: 9/2018 Origination: 9/2009 Next Review: 3/2019
 Facet Arthroplasty Policy Number: 7.01.120 Last Review: 9/2018 Origination: 9/2009 Next Review: 3/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide coverage for total facet
Facet Arthroplasty Policy Number: 7.01.120 Last Review: 9/2018 Origination: 9/2009 Next Review: 3/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide coverage for total facet
BONE OR SOFT TISSUE HEALING AND FUSION ENHANCEMENT PRODUCTS
 BONE OR SOFT TISSUE HEALING AND FUSION ENHANCEMENT PRODUCTS UnitedHealthcare Commercial Medical Policy Policy Number: 2017T0410S Effective Date: March 1, 2017 Table of Contents Page INSTRUCTIONS FOR USE...
BONE OR SOFT TISSUE HEALING AND FUSION ENHANCEMENT PRODUCTS UnitedHealthcare Commercial Medical Policy Policy Number: 2017T0410S Effective Date: March 1, 2017 Table of Contents Page INSTRUCTIONS FOR USE...
The most common surgery for chronic low back pain. Review
 Review Annals of Internal Medicine Effectiveness and Harms of Recombinant Human Bone Morphogenetic Protein-2 in Spine Fusion A Systematic Review and Meta-analysis Rongwei Fu, PhD; Shelley Selph, MD; Marian
Review Annals of Internal Medicine Effectiveness and Harms of Recombinant Human Bone Morphogenetic Protein-2 in Spine Fusion A Systematic Review and Meta-analysis Rongwei Fu, PhD; Shelley Selph, MD; Marian
Corporate Medical Policy
 Corporate Medical Policy Image-Guided Minimally Invasive Decompression (IG-MLD) for File Name: Origination: Last CAP Review: Next CAP Review: Last Review: image-guided_minimally_invasive_decompression_for_spinal_stenosis
Corporate Medical Policy Image-Guided Minimally Invasive Decompression (IG-MLD) for File Name: Origination: Last CAP Review: Next CAP Review: Last Review: image-guided_minimally_invasive_decompression_for_spinal_stenosis
Surgical Neurology International
 Surgical Neurology International OPEN ACCESS For entire Editorial Board visit : http://www.surgicalneurologyint.com Editor-in-Chief: James I. Ausman, MD, PhD University of California, Los Angeles, CA,
Surgical Neurology International OPEN ACCESS For entire Editorial Board visit : http://www.surgicalneurologyint.com Editor-in-Chief: James I. Ausman, MD, PhD University of California, Los Angeles, CA,
BMP s: The future of nonunion treatment or do we have a problem?
 BMP s: The future of nonunion treatment or do we have a problem? Michael D. McKee, MD, FRCS(C) Professor, Division of Orthopaedics, St. Michael s Hospital, University of Toronto, Toronto, Canada Disclosure
BMP s: The future of nonunion treatment or do we have a problem? Michael D. McKee, MD, FRCS(C) Professor, Division of Orthopaedics, St. Michael s Hospital, University of Toronto, Toronto, Canada Disclosure
CERVICAL PROCEDURES PHYSICIAN CODING
 CERVICAL PROCEDURES PHYSICIAN CODING Anterior Cervical Discectomy with Interbody Fusion (ACDF) Anterior interbody fusion, with discectomy and decompression; cervical below C2 22551 first interspace 22552
CERVICAL PROCEDURES PHYSICIAN CODING Anterior Cervical Discectomy with Interbody Fusion (ACDF) Anterior interbody fusion, with discectomy and decompression; cervical below C2 22551 first interspace 22552
Corporate Medical Policy
 Corporate Medical Policy Axial Lumbosacral Interbody Fusion File Name: Origination: Last CAP Review: Next CAP Review: Last Review: axial_lumbosacral_interbody_fusion 6/2009 10/2017 10/2018 10/2017 Description
Corporate Medical Policy Axial Lumbosacral Interbody Fusion File Name: Origination: Last CAP Review: Next CAP Review: Last Review: axial_lumbosacral_interbody_fusion 6/2009 10/2017 10/2018 10/2017 Description
ProDisc-L Total Disc Replacement. IDE Clinical Study.
 ProDisc-L Total Disc Replacement. IDE Clinical Study. A multi-center, prospective, randomized clinical trial. Instruments and implants approved by the AO Foundation Table of Contents Indications, Contraindications
ProDisc-L Total Disc Replacement. IDE Clinical Study. A multi-center, prospective, randomized clinical trial. Instruments and implants approved by the AO Foundation Table of Contents Indications, Contraindications
INFUSE Bone Graft/ LT-CAGE Lumbar Tapered Fusion Device. Patient Information
 INFUSE Bone Graft/ LT-CAGE Lumbar Tapered Fusion Device Patient Information Patient Information Brochure This Patient Guide is designed to help you make an informed decision about treatment for your back
INFUSE Bone Graft/ LT-CAGE Lumbar Tapered Fusion Device Patient Information Patient Information Brochure This Patient Guide is designed to help you make an informed decision about treatment for your back
U.S. MARKET FOR MINIMALLY INVASIVE SPINAL IMPLANTS
 U.S. MARKET FOR MINIMALLY INVASIVE SPINAL IMPLANTS idata_usmis15_rpt Published in December 2014 By idata Research Inc., 2014 idata Research Inc. Suite 308 4211 Kingsway Burnaby, British Columbia, Canada,
U.S. MARKET FOR MINIMALLY INVASIVE SPINAL IMPLANTS idata_usmis15_rpt Published in December 2014 By idata Research Inc., 2014 idata Research Inc. Suite 308 4211 Kingsway Burnaby, British Columbia, Canada,
5/19/2017. Interspinous Process Fixation with the Minuteman G3. What is the Minuteman G3. How Does it Work?
 Interspinous Process Fixation with the Minuteman G3 LLOYDINE J. JACOBS, MD CASTELLVI SPINE MEETING MAY 13, 2017 What is the Minuteman G3 The world s first spinous process plating system that is: Minimally
Interspinous Process Fixation with the Minuteman G3 LLOYDINE J. JACOBS, MD CASTELLVI SPINE MEETING MAY 13, 2017 What is the Minuteman G3 The world s first spinous process plating system that is: Minimally
CPT CODING EXAMPLES FUSION PROCEDURES. Anterior Lumbar Interbody Fusion (ALIF)
 CPT CODING EXAMPLES This list represents coding examples for common spine procedures. The information can also be used in conjunction with the Medicare Fee Calculator on http://www.cms.gov/apps/physician-fee-schedule/
CPT CODING EXAMPLES This list represents coding examples for common spine procedures. The information can also be used in conjunction with the Medicare Fee Calculator on http://www.cms.gov/apps/physician-fee-schedule/
Alamo T Transforaminal Lumbar Interbody System Surgical Technique
 Transforaminal Lumbar Interbody System Surgical Technique Table of Contents Indications and Device Description.............. 1 Alamo T Implant Features and Instruments...........2 Surgical Technique......................
Transforaminal Lumbar Interbody System Surgical Technique Table of Contents Indications and Device Description.............. 1 Alamo T Implant Features and Instruments...........2 Surgical Technique......................
FEP Medical Policy Manual
 FEP Medical Policy Manual Effective Date: July 15, 2017 Related Policies: 1.01.05 Ultrasound Accelerated Fracture Healing Device 7.01.07 Electrical Bone Growth Stimulation of the Appendicular Skeleton
FEP Medical Policy Manual Effective Date: July 15, 2017 Related Policies: 1.01.05 Ultrasound Accelerated Fracture Healing Device 7.01.07 Electrical Bone Growth Stimulation of the Appendicular Skeleton
Patient Information MIS LLIF. Lateral Lumbar Interbody Fusion Using Minimally Invasive Surgical Techniques
 Patient Information MIS LLIF Lateral Lumbar Interbody Fusion Using Minimally Invasive Surgical Techniques Table of Contents Anatomy of Spine....2 General Conditions of the Spine....4 What is Spondylolisthesis....5
Patient Information MIS LLIF Lateral Lumbar Interbody Fusion Using Minimally Invasive Surgical Techniques Table of Contents Anatomy of Spine....2 General Conditions of the Spine....4 What is Spondylolisthesis....5
Medical Policy Recombinant Human Bone Morphogenetic Protein (rhbmp) for Bone Fusion
 Medical Policy Recombinant Human Bone Morphogenetic Protein (rhbmp) for Bone Fusion Subject: Recombinant Human Bone Morphogenetic Protein (rhbmp) for Bone Fusion Background: Bone morphogenetic proteins
Medical Policy Recombinant Human Bone Morphogenetic Protein (rhbmp) for Bone Fusion Subject: Recombinant Human Bone Morphogenetic Protein (rhbmp) for Bone Fusion Background: Bone morphogenetic proteins
Populations Interventions Comparators Outcomes Individuals: With lumbar spinal stenosis
 Image-Guided Minimally Invasive Decompression for Spinal (701126) (Formerly Image-Guided Minimally Invasive Lumbar Decompression for Spinal ) Medical Benefit Effective Date: 10/01/17 Next Review Date:
Image-Guided Minimally Invasive Decompression for Spinal (701126) (Formerly Image-Guided Minimally Invasive Lumbar Decompression for Spinal ) Medical Benefit Effective Date: 10/01/17 Next Review Date:
PART III IN HOSPITAL ON CALL ANESTHESIA COVERAGE
 Anesthesia g) A consultation may not be claimed where the patient is referred to the anesthetist for the sole purpose of providing post-operative Patient Controlled Analgesia. h) Tariff 8406 may not be
Anesthesia g) A consultation may not be claimed where the patient is referred to the anesthetist for the sole purpose of providing post-operative Patient Controlled Analgesia. h) Tariff 8406 may not be
Disclosure. Endochondral Healing 4/28/2016. Fracture Healing Stimulation Where s the Evidence?
 Fracture Healing Stimulation Where s the Evidence? Thomas J Moore MD Atlanta Trauma Symposium April 23, 2016 Disclosure Member: AAOS Committee on Biologics Reviewer: Clinical Orthopaedics Related Research
Fracture Healing Stimulation Where s the Evidence? Thomas J Moore MD Atlanta Trauma Symposium April 23, 2016 Disclosure Member: AAOS Committee on Biologics Reviewer: Clinical Orthopaedics Related Research
Lumbar Spinal Fusion Corporate Medical Policy
 Lumbar Spinal Fusion Corporate Medical Policy File name: Lumbar Spinal Fusion File code: UM.SURG.15 Origination: 09/01/2016 Last Review: 09/29/2016 Next Review: 09/29/2017 Effective Date: 01/01/2017 Populations
Lumbar Spinal Fusion Corporate Medical Policy File name: Lumbar Spinal Fusion File code: UM.SURG.15 Origination: 09/01/2016 Last Review: 09/29/2016 Next Review: 09/29/2017 Effective Date: 01/01/2017 Populations
T.L.I.F. Surgical Technique. Featuring the T.L.I.F. SG Instruments, VG2 PLIF Allograft, and the MONARCH Spine System.
 Surgical Technique T.L.I.F. Transforaminal Lumbar Interbody Fusion Featuring the T.L.I.F. SG Instruments, VG2 PLIF Allograft, and the MONARCH Spine System. CONSULTING SURGEON Todd Albert, M.D. Rothman
Surgical Technique T.L.I.F. Transforaminal Lumbar Interbody Fusion Featuring the T.L.I.F. SG Instruments, VG2 PLIF Allograft, and the MONARCH Spine System. CONSULTING SURGEON Todd Albert, M.D. Rothman
Royal Oak IBFD System Surgical Technique Posterior Lumbar Interbody Fusion (PLIF)
 Royal Oak IBFD System Surgical Technique Posterior Lumbar Interbody Fusion (PLIF) Preoperative Planning Preoperative planning is necessary for the correct selection of lumbar interbody fusion devices.
Royal Oak IBFD System Surgical Technique Posterior Lumbar Interbody Fusion (PLIF) Preoperative Planning Preoperative planning is necessary for the correct selection of lumbar interbody fusion devices.
Topic: Percutaneous Axial Anterior Lumbar Fusion Date of Origin: June Section: Surgery Last Reviewed Date: June 2013
 Medical Policy Manual Topic: Percutaneous Axial Anterior Lumbar Fusion Date of Origin: June 2007 Section: Surgery Last Reviewed Date: June 2013 Policy No: 157 Effective Date: August 1, 2013 IMPORTANT REMINDER
Medical Policy Manual Topic: Percutaneous Axial Anterior Lumbar Fusion Date of Origin: June 2007 Section: Surgery Last Reviewed Date: June 2013 Policy No: 157 Effective Date: August 1, 2013 IMPORTANT REMINDER
Is unilateral pedicle screw fixation superior than bilateral pedicle screw fixation for lumbar degenerative diseases: a meta-analysis
 Lu et al. Journal of Orthopaedic Surgery and Research (2018) 13:296 https://doi.org/10.1186/s13018-018-1004-x SYSTEMATIC REVIEW Open Access Is unilateral pedicle screw fixation superior than bilateral
Lu et al. Journal of Orthopaedic Surgery and Research (2018) 13:296 https://doi.org/10.1186/s13018-018-1004-x SYSTEMATIC REVIEW Open Access Is unilateral pedicle screw fixation superior than bilateral
Degenerative L4-5 SPONDYLOLISTHESIS with Stenosis: Laminectomy and Postero-Lateral Fusion. Rick C. Sasso MD
 Degenerative L4-5 SPONDYLOLISTHESIS with Stenosis: Laminectomy and Postero-Lateral Fusion Rick C. Sasso MD Professor Chief of Surgery Clinical Orthopaedic Surgery University School of Medicine Disclosure:
Degenerative L4-5 SPONDYLOLISTHESIS with Stenosis: Laminectomy and Postero-Lateral Fusion Rick C. Sasso MD Professor Chief of Surgery Clinical Orthopaedic Surgery University School of Medicine Disclosure:
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Lumbar Spinal Fusion Page 1 of 28 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: See Also: Lumbar Spinal Fusion Interspinous Fixation (Fusion) Devices http://www.bcbsks.com/customerservice/providers/medicalpolicies/policies.shtml
Lumbar Spinal Fusion Page 1 of 28 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: See Also: Lumbar Spinal Fusion Interspinous Fixation (Fusion) Devices http://www.bcbsks.com/customerservice/providers/medicalpolicies/policies.shtml
Patient Information MIS LLIF. Lateral Lumbar Interbody Fusion Using Minimally Invasive Surgical Techniques
 Patient Information MIS LLIF Lateral Lumbar Interbody Fusion Using Minimally Invasive Surgical Techniques Table of Contents Anatomy of Spine...2 General Conditions of the Spine....4 What is Spondylolisthesis....5
Patient Information MIS LLIF Lateral Lumbar Interbody Fusion Using Minimally Invasive Surgical Techniques Table of Contents Anatomy of Spine...2 General Conditions of the Spine....4 What is Spondylolisthesis....5
Corporate Medical Policy
 Corporate Medical Policy Epidural Steroid Injections for Back Pain File Name: Origination: Last CAP Review: Next CAP Review: Last Review: epidural_steroid_injections_for_back_pain 2/2016 4/2017 4/2018
Corporate Medical Policy Epidural Steroid Injections for Back Pain File Name: Origination: Last CAP Review: Next CAP Review: Last Review: epidural_steroid_injections_for_back_pain 2/2016 4/2017 4/2018
The Fusion of. Strength, Design. Performance. and
 The Fusion of Strength, Design and Performance Interbody Fusion Platform Interbody Fusion Platform The DePuy Spine Interbody Fusion platform is designed to provide an open architecture implant in a load-sharing
The Fusion of Strength, Design and Performance Interbody Fusion Platform Interbody Fusion Platform The DePuy Spine Interbody Fusion platform is designed to provide an open architecture implant in a load-sharing
Microendoscope-assisted posterior lumbar interbody fusion: a technical note
 Original Study Microendoscope-assisted posterior lumbar interbody fusion: a technical note Hirohiko Inanami 1, Fumiko Saiki 1, Yasushi Oshima 2 1 Department of Orthopaedic Surgery, Inanami Spine and Joint
Original Study Microendoscope-assisted posterior lumbar interbody fusion: a technical note Hirohiko Inanami 1, Fumiko Saiki 1, Yasushi Oshima 2 1 Department of Orthopaedic Surgery, Inanami Spine and Joint
Original Date: October 2015 LUMBAR SPINAL FUSION FOR
 National Imaging Associates, Inc. Clinical guidelines Original Date: October 2015 LUMBAR SPINAL FUSION FOR Page 1 of 9 INSTABILITY AND DEGENERATIVE DISC CONDITIONS FOR CMS (MEDICARE) MEMBERS ONLY CPT4
National Imaging Associates, Inc. Clinical guidelines Original Date: October 2015 LUMBAR SPINAL FUSION FOR Page 1 of 9 INSTABILITY AND DEGENERATIVE DISC CONDITIONS FOR CMS (MEDICARE) MEMBERS ONLY CPT4
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Lumbar Spinal Fusion Page 1 of 29 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: See Also: Lumbar Spinal Fusion Interspinous Fixation (Fusion) Devices http://www.bcbsks.com/customerservice/providers/medicalpolicies/policies.shtml
Lumbar Spinal Fusion Page 1 of 29 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: See Also: Lumbar Spinal Fusion Interspinous Fixation (Fusion) Devices http://www.bcbsks.com/customerservice/providers/medicalpolicies/policies.shtml
Surgical Technique. Apache Posterior Lumbar Interbody Fusion Apache Transforaminal Lumbar Interbody Fusion
 Surgical Technique Apache Posterior Lumbar Interbody Fusion Apache Transforaminal Lumbar Interbody Fusion 2 Table of Contents Page Preoperative Planning 4 Patient Positioning 5 Disc Exposure 5 Disc and
Surgical Technique Apache Posterior Lumbar Interbody Fusion Apache Transforaminal Lumbar Interbody Fusion 2 Table of Contents Page Preoperative Planning 4 Patient Positioning 5 Disc Exposure 5 Disc and
Most cells in the human body have an assigned purpose. They are liver cells, fat cells, bone cells,
 What is a Stem Cell? Most cells in the human body have an assigned purpose. They are liver cells, fat cells, bone cells, and so on. These cells can replicate more of their own kind of cell, but they cannot
What is a Stem Cell? Most cells in the human body have an assigned purpose. They are liver cells, fat cells, bone cells, and so on. These cells can replicate more of their own kind of cell, but they cannot
5/27/2016. Stand-Alone Lumbar Lateral Interbody Fusion (LLIF) vs. Supplemental Fixation. Disclosures. LLIF Approach
 Stand-Alone Lumbar Lateral Interbody Fusion (LLIF) vs. Supplemental Fixation Joseph M. Zavatsky, M.D. Spine & Scoliosis Specialists Tampa, FL Disclosures Consultant - Zimmer / Biomet, DePuy Synthes Spine,
Stand-Alone Lumbar Lateral Interbody Fusion (LLIF) vs. Supplemental Fixation Joseph M. Zavatsky, M.D. Spine & Scoliosis Specialists Tampa, FL Disclosures Consultant - Zimmer / Biomet, DePuy Synthes Spine,
Populations Interventions Comparators Outcomes Individuals: With lumbar spinal stenosis
 Image-Guided Minimally Invasive Decompression for Spinal (701126) Medical Benefit Effective Date: 10/01/18 Next Review Date: 07/19 Preauthorization No Review Dates: 09/10, 07/11, 07/12, 07/13, 07/14, 07/15,
Image-Guided Minimally Invasive Decompression for Spinal (701126) Medical Benefit Effective Date: 10/01/18 Next Review Date: 07/19 Preauthorization No Review Dates: 09/10, 07/11, 07/12, 07/13, 07/14, 07/15,
Codes for Back and Spinal Procedures
 20930 Allograft for spine surgery only; morselized 20931 Allograft for spine surgery only; structural 20936 Autograft for spine surgery only (includes harvesting the graft); local (eg, ribs, spinous process,
20930 Allograft for spine surgery only; morselized 20931 Allograft for spine surgery only; structural 20936 Autograft for spine surgery only (includes harvesting the graft); local (eg, ribs, spinous process,
Interspinous Fusion Devices. Midterm results. ROME SPINE 2012, 7th International Meeting Rome, 6-7 December 2012
 Interspinous Fusion Devices. Midterm results. ROME SPINE 2012, 7th International Meeting Rome, 6-7 December 2012 Posterior distraction and decompression Secure Fixation and Stabilization Integrated Bone
Interspinous Fusion Devices. Midterm results. ROME SPINE 2012, 7th International Meeting Rome, 6-7 December 2012 Posterior distraction and decompression Secure Fixation and Stabilization Integrated Bone
T.L.I.F. Transforaminal Lumbar Interbody Fusion
 T.L.I.F. Transforaminal Lumbar Interbody Fusion Cover Surgical Header Technique Sub Guide header Introduction (T.L.I.F. ) technique has gained wide acceptance Additionally, the T.L.I.F. procedure avoids
T.L.I.F. Transforaminal Lumbar Interbody Fusion Cover Surgical Header Technique Sub Guide header Introduction (T.L.I.F. ) technique has gained wide acceptance Additionally, the T.L.I.F. procedure avoids
Replacement Code for Interbody Cage for Disc
 +22851 vs. +20931 October 22, 2015 We ve been told we cannot bill +22851 and +20931 with the ACDF code, 22551. Is this true? It is true if you are thinking about reporting +22851 (intervertebral device)
+22851 vs. +20931 October 22, 2015 We ve been told we cannot bill +22851 and +20931 with the ACDF code, 22551. Is this true? It is true if you are thinking about reporting +22851 (intervertebral device)
Anterior cervical diskectomy icd 10 procedure code
 Home Anterior cervical diskectomy icd 10 procedure code Access to discounts at hundreds of restaurants, travel destinations, retail stores, and service providers. AAPC members also have opportunities to
Home Anterior cervical diskectomy icd 10 procedure code Access to discounts at hundreds of restaurants, travel destinations, retail stores, and service providers. AAPC members also have opportunities to
Sinus Augmentation Studies Methods and Definition
 FDA approved indications for Infuse Bone Graft in the Maxillofacial Skeleton Alveolar Ridge Augmentation (Buccal Wall Defects) in the Maxilla Maxillary Sinus Floor augmentation Sinus Augmentation Studies
FDA approved indications for Infuse Bone Graft in the Maxillofacial Skeleton Alveolar Ridge Augmentation (Buccal Wall Defects) in the Maxilla Maxillary Sinus Floor augmentation Sinus Augmentation Studies
Lumbar Laminotomy DEFINING APPROPRIATE COVERAGE POSITIONS NASS COVERAGE POLICY RECOMMENDATIONS TASKFORCE
 NASS COVERAGE POLICY RECOMMENDATIONS Lumbar Laminotomy DEFINING APPROPRIATE COVERAGE POSITIONS North American Spine Society 7075 Veterans Blvd. Burr Ridge, IL 60527 TASKFORCE Introduction North American
NASS COVERAGE POLICY RECOMMENDATIONS Lumbar Laminotomy DEFINING APPROPRIATE COVERAGE POSITIONS North American Spine Society 7075 Veterans Blvd. Burr Ridge, IL 60527 TASKFORCE Introduction North American
Spinal Surgery coding Pain Coding
 Spinal Surgery coding Pain Coding CASE STUDIES, DISCUSSION ROBIN INGALLS-FITZGERALD, CCS, CPC, FCS, CEDC, CEMC CEO/PRESIDENT MEDICAL MANAGEMENT AND REIMBURSEMENT SPECIALISTS, LLC Agenda Discuss spinal
Spinal Surgery coding Pain Coding CASE STUDIES, DISCUSSION ROBIN INGALLS-FITZGERALD, CCS, CPC, FCS, CEDC, CEMC CEO/PRESIDENT MEDICAL MANAGEMENT AND REIMBURSEMENT SPECIALISTS, LLC Agenda Discuss spinal
Axial Lumbosacral Interbody Fusion. Description
 Section: Surgery Effective Date: April 15, 2014 Subject: Axial Lumbosacral Interbody Fusion Page: 1 of 6 Last Review Status/Date: March 2014 Axial Lumbosacral Interbody Fusion Description Axial lumbosacral
Section: Surgery Effective Date: April 15, 2014 Subject: Axial Lumbosacral Interbody Fusion Page: 1 of 6 Last Review Status/Date: March 2014 Axial Lumbosacral Interbody Fusion Description Axial lumbosacral
Electrical Bone Growth Stimulators (Osteogenic Stimulation)
 Medical Policy Manual Durable Medical Equipment, Policy No. 83.11 Electrical Bone Growth Stimulators (Osteogenic Stimulation) Next Review: July 2018 Last Review: September 2017 Effective: October 1, 2017
Medical Policy Manual Durable Medical Equipment, Policy No. 83.11 Electrical Bone Growth Stimulators (Osteogenic Stimulation) Next Review: July 2018 Last Review: September 2017 Effective: October 1, 2017
nva Anterior Lumbar Interbody Fusion System
 nva Anterior Lumbar Interbody Fusion System 1 IMPORTANT INFORMATION FOR PHYSICIANS, SURGEONS, AND/OR STAFF The nv a, nv p, and nv t are an intervertebral body fusion device used in the lumbar spine following
nva Anterior Lumbar Interbody Fusion System 1 IMPORTANT INFORMATION FOR PHYSICIANS, SURGEONS, AND/OR STAFF The nv a, nv p, and nv t are an intervertebral body fusion device used in the lumbar spine following
3D titanium interbody fusion cages sharx. White Paper
 3D titanium interbody fusion cages sharx (SLM selective laser melted) Goal of the study: Does the sharx intervertebral cage due to innovative material, new design, and lordotic shape solve some problems
3D titanium interbody fusion cages sharx (SLM selective laser melted) Goal of the study: Does the sharx intervertebral cage due to innovative material, new design, and lordotic shape solve some problems
Patient Information Brochure
 Patient Information Brochure Patient Information Brochure This brochure is designed to help you make an informed decision about your surgery. Please read this entire document carefully. Keep this document
Patient Information Brochure Patient Information Brochure This brochure is designed to help you make an informed decision about your surgery. Please read this entire document carefully. Keep this document
nvt Transforaminal Lumbar Interbody Fusion System
 nvt Transforaminal Lumbar Interbody Fusion System 1 IMPORTANT INFORMATION FOR PHYSICIANS, SURGEONS, AND/OR STAFF The nv a, nv p, and nv t are an intervertebral body fusion device used in the lumbar spine
nvt Transforaminal Lumbar Interbody Fusion System 1 IMPORTANT INFORMATION FOR PHYSICIANS, SURGEONS, AND/OR STAFF The nv a, nv p, and nv t are an intervertebral body fusion device used in the lumbar spine
nvp Posterior Lumbar Interbody Fusion System
 nvp Posterior Lumbar Interbody Fusion System 1 IMPORTANT INFORMATION FOR PHYSICIANS, SURGEONS, AND/OR STAFF The nv a, nv p, and nv t are an intervertebral body fusion device used in the lumbar spine following
nvp Posterior Lumbar Interbody Fusion System 1 IMPORTANT INFORMATION FOR PHYSICIANS, SURGEONS, AND/OR STAFF The nv a, nv p, and nv t are an intervertebral body fusion device used in the lumbar spine following
Procedure Coding Made Simple Five principles will help you capture appropriate charges for spine surgeries.
 Coding/Billing By Kim Pollock, MS, RN, MBA, CPC Procedure Coding Made Simple Five principles will help you capture appropriate charges for spine surgeries. It seems like coding spine cases is as complicated
Coding/Billing By Kim Pollock, MS, RN, MBA, CPC Procedure Coding Made Simple Five principles will help you capture appropriate charges for spine surgeries. It seems like coding spine cases is as complicated
Degenerative spondylolisthesis at the L4 L5 in a 32-year-old female with previous fusion for idiopathic scoliosis: A case report
 Journal of Orthopaedic Surgery 2003: 11(2): 202 206 Degenerative spondylolisthesis at the L4 L5 in a 32-year-old female with previous fusion for idiopathic scoliosis: A case report RB Winter Clinical Professor,
Journal of Orthopaedic Surgery 2003: 11(2): 202 206 Degenerative spondylolisthesis at the L4 L5 in a 32-year-old female with previous fusion for idiopathic scoliosis: A case report RB Winter Clinical Professor,
PATIENT: DOB: TODAY S DATE:
 1. I have been strongly advised to carefully read and consider this operative permit. I realize that it is important that I understand this material. I also understand that if certain sections are not
1. I have been strongly advised to carefully read and consider this operative permit. I realize that it is important that I understand this material. I also understand that if certain sections are not
Spine Tango annual report 2012
 DOI 10.1007/s00586-013-2943-x SPINE TANGO REPORT 2012 Spine Tango annual report 2012 M. Neukamp G. Perler T. Pigott E. Munting M. Aebi C. Röder Received: 31 July 2013 / Published online: 30 August 2013
DOI 10.1007/s00586-013-2943-x SPINE TANGO REPORT 2012 Spine Tango annual report 2012 M. Neukamp G. Perler T. Pigott E. Munting M. Aebi C. Röder Received: 31 July 2013 / Published online: 30 August 2013
PATIENT INFORMATION BROCHURE
 PATIENT INFORMATION BROCHURE PATIENT INFORMATION BROCHURE This brochure is designed to help you make an informed decision about your surgery. Please read this entire document carefully. Keep this document
PATIENT INFORMATION BROCHURE PATIENT INFORMATION BROCHURE This brochure is designed to help you make an informed decision about your surgery. Please read this entire document carefully. Keep this document
Anterior and Lateral Lumbar Minimally Invasive Approaches: How to Choose
 Anterior and Lateral Lumbar Minimally Invasive Approaches: How to Choose Lukas P. Zebala, MD Assistant Professor Washington University School of Medicine St. Louis, MO Disclosures Consultant: K2M, Inc.
Anterior and Lateral Lumbar Minimally Invasive Approaches: How to Choose Lukas P. Zebala, MD Assistant Professor Washington University School of Medicine St. Louis, MO Disclosures Consultant: K2M, Inc.
ILIF Interlaminar Lumbar Instrumented Fusion. Anton Thompkins, M.D.
 ILIF Interlaminar Lumbar Instrumented Fusion Anton Thompkins, M.D. Anton Thompkins, M.D. EDUCATION: BS, Biology, DePauw University, Greencastle, IN MD, University of Cincinnati College of Medicine RESIDENCY:
ILIF Interlaminar Lumbar Instrumented Fusion Anton Thompkins, M.D. Anton Thompkins, M.D. EDUCATION: BS, Biology, DePauw University, Greencastle, IN MD, University of Cincinnati College of Medicine RESIDENCY:
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of lateral interbody fusion in the lumbar spine for low back pain As a person gets
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE INTERVENTIONAL PROCEDURES PROGRAMME Interventional procedure overview of lateral interbody fusion in the lumbar spine for low back pain As a person gets
Bare Bones. Use of Systemic Osteoporosis Drugs in Select High Risk Patients Undergoing Spine Surgery
 Use of Systemic Osteoporosis Drugs in Select High Risk Patients Undergoing Spine Surgery 30th Annual Metabolic Bone Disease Society Meeting Denver, CO Oct 11, 2018 Bare Bones 2000 Francesca Pera Kelly
Use of Systemic Osteoporosis Drugs in Select High Risk Patients Undergoing Spine Surgery 30th Annual Metabolic Bone Disease Society Meeting Denver, CO Oct 11, 2018 Bare Bones 2000 Francesca Pera Kelly
22110 vertebral segment; cervical vertebral segment; thoracic vertebral segment; lumbar
 The following codes are authorized by Palladian Health for applicable product lines. Visit palladianhealth.com to request authorization and to access guidelines. Palladian Musculoskeletal Program Codes
The following codes are authorized by Palladian Health for applicable product lines. Visit palladianhealth.com to request authorization and to access guidelines. Palladian Musculoskeletal Program Codes
2012 CPT Coding Update AANS/CNS Joint Section on Disorders of the Spine and Peripheral Nerves
 2012 CPT Coding Update AANS/CNS Joint Section on Disorders of the Spine and Peripheral Nerves Joseph S. Cheng, M.D., M.S. Associate Professor of Neurological Surgery, Orthopedic Surgery, and Rehabilitation
2012 CPT Coding Update AANS/CNS Joint Section on Disorders of the Spine and Peripheral Nerves Joseph S. Cheng, M.D., M.S. Associate Professor of Neurological Surgery, Orthopedic Surgery, and Rehabilitation
ACDF. Anterior Cervical Discectomy and Fusion. An introduction to
 An introduction to ACDF Anterior Cervical Discectomy and Fusion This booklet provides general information on ACDF. It is not meant to replace any personal conversations that you might wish to have with
An introduction to ACDF Anterior Cervical Discectomy and Fusion This booklet provides general information on ACDF. It is not meant to replace any personal conversations that you might wish to have with
EFSPINE CERVICAL COMBINED SET DISC PROTHESIS ORGANIZER BOX
 EFSPINE CERVICAL COMBINED SET INSTRUMENTS CERVICAL CAGE & DISC PROTHESIS ORGANIZER BOX Cervical Thoracic Thoraco - Lumbar Sacral EFSPINE CERVICAL COMBINED SET CERVICAL IMPLANTS INTRODUCTION Cervical Disc
EFSPINE CERVICAL COMBINED SET INSTRUMENTS CERVICAL CAGE & DISC PROTHESIS ORGANIZER BOX Cervical Thoracic Thoraco - Lumbar Sacral EFSPINE CERVICAL COMBINED SET CERVICAL IMPLANTS INTRODUCTION Cervical Disc
Medical Policy An independent licensee of the
 Interspinous Fixation (Fusion) Devices Page 1 of 8 Medical Policy An independent licensee of the Title: See Also: Interspinous Fixation (Fusion) Devices Lumbar Spine Fusion http://www.bcbsks.com/customerservice/providers/medicalpolicies/policies.shtml
Interspinous Fixation (Fusion) Devices Page 1 of 8 Medical Policy An independent licensee of the Title: See Also: Interspinous Fixation (Fusion) Devices Lumbar Spine Fusion http://www.bcbsks.com/customerservice/providers/medicalpolicies/policies.shtml
POSTERIOR CERVICAL FUSION
 AN INTRODUCTION TO PCF POSTERIOR CERVICAL FUSION This booklet provides general information on the Posterior Cervical Fusion (PCF) surgical procedure for you to discuss with your physician. It is not meant
AN INTRODUCTION TO PCF POSTERIOR CERVICAL FUSION This booklet provides general information on the Posterior Cervical Fusion (PCF) surgical procedure for you to discuss with your physician. It is not meant
Anterior Cervical Discectomy and Fusion Surgery
 Disclaimer This movie is an educational resource only and should not be used to manage orthopaedic health. All decisions about the management of orthopaedic conditions must be made in conjunction with
Disclaimer This movie is an educational resource only and should not be used to manage orthopaedic health. All decisions about the management of orthopaedic conditions must be made in conjunction with
Spine A Preliminary Study of the Efficacy of Beta-Tricalcium Phosphate as a Bone Graft Expander for Instrumented Posterolateral Fusions
 Bibliography Spine Epstein, N.E., A Preliminary Study of the Efficacy of Beta-Tricalcium Phosphate as a Bone Graft Expander for Instrumented Posterolateral Fusions. Journal of Spinal Disorders and Techniques,
Bibliography Spine Epstein, N.E., A Preliminary Study of the Efficacy of Beta-Tricalcium Phosphate as a Bone Graft Expander for Instrumented Posterolateral Fusions. Journal of Spinal Disorders and Techniques,
1105 two (2) vertebrae... 1, add on per additional vertebra
 SPINE STAGE OPERATIONS Staged operations shall be paid at 100% for the first stage and 85% for the second stage. Where the second stage pays a higher fee 100% shall be paid and the first stage shall be
SPINE STAGE OPERATIONS Staged operations shall be paid at 100% for the first stage and 85% for the second stage. Where the second stage pays a higher fee 100% shall be paid and the first stage shall be
PARADIGM SPINE. Minimally Invasive Lumbar Fusion. Interlaminar Stabilization
 PARADIGM SPINE Minimally Invasive Lumbar Fusion Interlaminar Stabilization 2 A UNIQUE MIS ALTERNATIVE TO PEDICLE SCREW FIXATION The Gold Standard The combined use of surgical decompression and different
PARADIGM SPINE Minimally Invasive Lumbar Fusion Interlaminar Stabilization 2 A UNIQUE MIS ALTERNATIVE TO PEDICLE SCREW FIXATION The Gold Standard The combined use of surgical decompression and different
Medical Policy Original Effective Date: Revised Date: Page 1 of 11
 Page 1 of 11 Content Disclaimer Description Coverage Determination Clinical Indications Lumbar Spine Surgery Lumbar Spine Surgery Description Indication Coding Lumbar Spinal Fusion (single level)surgery
Page 1 of 11 Content Disclaimer Description Coverage Determination Clinical Indications Lumbar Spine Surgery Lumbar Spine Surgery Description Indication Coding Lumbar Spinal Fusion (single level)surgery
Prosthetic intervertebral disc replacement in the cervical spine: Cost-effectiveness compared with cervical discectomy with or without vertebral
 Prosthetic intervertebral disc replacement in the cervical spine: Cost-effectiveness compared with cervical discectomy with or without vertebral fusion Author: William Horsley NHS North East Treatment
Prosthetic intervertebral disc replacement in the cervical spine: Cost-effectiveness compared with cervical discectomy with or without vertebral fusion Author: William Horsley NHS North East Treatment
Original Policy Date
 MP 7.01.110 Axial Lumbosacral Interbody Fusion Medical Policy Section Surgery Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search12/2013 Return to Medical
MP 7.01.110 Axial Lumbosacral Interbody Fusion Medical Policy Section Surgery Issue 12/2013 Original Policy Date 12/2013 Last Review Status/Date Reviewed with literature search12/2013 Return to Medical
A PROSPECTIVE STUDY OF INCIDENTAL DURAL TEARS IN MICROENDOSCOPIC LUMBAR DECOMPRESSION SURGERY: INCIDENCE AND OUTCOMES
 A PROSPECTIVE STUDY OF INCIDENTAL DURAL TEARS IN MICROENDOSCOPIC LUMBAR DECOMPRESSION SURGERY: INCIDENCE AND OUTCOMES Takahiro Tsutsumimoto, Mutsuki Yui, Masashi Uehara, Hiroki Ohba, Hiroshi Ohta, Hidemi
A PROSPECTIVE STUDY OF INCIDENTAL DURAL TEARS IN MICROENDOSCOPIC LUMBAR DECOMPRESSION SURGERY: INCIDENCE AND OUTCOMES Takahiro Tsutsumimoto, Mutsuki Yui, Masashi Uehara, Hiroki Ohba, Hiroshi Ohta, Hidemi
Patient Information MIS TLIF. Transforaminal Lumbar Interbody Fusion Using Minimally Invasive Surgical Techniques
 Patient Information MIS TLIF Transforaminal Lumbar Interbody Fusion Using Minimally Invasive Surgical Techniques MIS TLIF Table of Contents Anatomy of Spine..............................................
Patient Information MIS TLIF Transforaminal Lumbar Interbody Fusion Using Minimally Invasive Surgical Techniques MIS TLIF Table of Contents Anatomy of Spine..............................................
Corporate Medical Policy
 Corporate Medical Policy Electrical Bone Growth Stimulation File Name: Origination: Last CAP Review: Next CAP Review: Last Review: electrical_bone_growth_stimulation 4/1981 6/2017 6/2018 6/2017 Description
Corporate Medical Policy Electrical Bone Growth Stimulation File Name: Origination: Last CAP Review: Next CAP Review: Last Review: electrical_bone_growth_stimulation 4/1981 6/2017 6/2018 6/2017 Description
Disclosures 5/27/2016. Narrowing of the spinal canal or neuroforamina causing a symptomatic compression of the neural element.
 Radiculopathy Saggital MRI view Contained disc extrusion uplifting PLL from bone Terrence Julien, MD, FACS, (blue FAANS arrows) Associate Professor of Neurosurgery System Director, Surgical Neuro-Oncology
Radiculopathy Saggital MRI view Contained disc extrusion uplifting PLL from bone Terrence Julien, MD, FACS, (blue FAANS arrows) Associate Professor of Neurosurgery System Director, Surgical Neuro-Oncology
TABLE OF CONTENTS. ShurFit Anterior Cervical Interbody Fusion (ACIF) System Overview 2. Implant Specifications 3. Instrument Features 4
 Surgical Technique TABLE OF CONTENTS ShurFit Anterior Cervical Interbody Fusion (ACIF) System Overview 2 Product Highlights 2 Indications 2 Implant Specifications 3 Instrument Features 4 Surgical Technique
Surgical Technique TABLE OF CONTENTS ShurFit Anterior Cervical Interbody Fusion (ACIF) System Overview 2 Product Highlights 2 Indications 2 Implant Specifications 3 Instrument Features 4 Surgical Technique
Abstract Study Design Systematic review. John C. France 1 James M. Schuster 2 Katherine Moran 3 Joseph R. Dettori 3
 THIEME GLOBAL SPINE JOURNAL Systematic Review 195 Iliac Crest Bone Graft in Lumbar Fusion: The Effectiveness and Safety Compared with Local Bone Graft, and Graft Site Morbidity Comparing a Single-Incision
THIEME GLOBAL SPINE JOURNAL Systematic Review 195 Iliac Crest Bone Graft in Lumbar Fusion: The Effectiveness and Safety Compared with Local Bone Graft, and Graft Site Morbidity Comparing a Single-Incision
Comparison of Clinical Outcomes Following Minimally Invasive Lateral Interbody Fusion Stratified by Preoperative Diagnosis
 Comparison of Clinical Outcomes Following Minimally Invasive Lateral Interbody Fusion Stratified by Preoperative Diagnosis Kaveh Khajavi, MD, FACS Alessandria Y. Shen, MSPH Anthony Hutchison, MSN Disclosures
Comparison of Clinical Outcomes Following Minimally Invasive Lateral Interbody Fusion Stratified by Preoperative Diagnosis Kaveh Khajavi, MD, FACS Alessandria Y. Shen, MSPH Anthony Hutchison, MSN Disclosures
Corporate Medical Policy
 Corporate Medical Policy Interspinous and Interlaminar Stabilization/Distraction Devices File Name: Origination: Last CAP Review: Next CAP Review: Last Review: interspinous_and_interlaminar_stabilization-distraction_devices
Corporate Medical Policy Interspinous and Interlaminar Stabilization/Distraction Devices File Name: Origination: Last CAP Review: Next CAP Review: Last Review: interspinous_and_interlaminar_stabilization-distraction_devices
Lumbar Disc Replacement FAQs
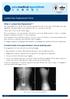 ISO 9001:2015 FS 550968 What is Lumbar Disc Replacement? The intervertebral discs are the shock absorbers between the bones of the spine. Unfortunately, they often degenerate tearing, bursting, or just
ISO 9001:2015 FS 550968 What is Lumbar Disc Replacement? The intervertebral discs are the shock absorbers between the bones of the spine. Unfortunately, they often degenerate tearing, bursting, or just
This procedure lacks scientific evidence of effectiveness, and is not covered.
 ARBenefits Approval: 09-21-2011 Effective Date: 01-01-2012 Revision Date: Code(s): 0275T Medical Policy Title: Minimally Invasive, Image-Guided Lumbar Decompression for Spinal Stenosis Document: ARB0186
ARBenefits Approval: 09-21-2011 Effective Date: 01-01-2012 Revision Date: Code(s): 0275T Medical Policy Title: Minimally Invasive, Image-Guided Lumbar Decompression for Spinal Stenosis Document: ARB0186
SURGICAL TECHNIQUE MANUAL. InterFuse T
 1 CONTENTS InterFuse T Product Description 3 Indications for Use 3 X-Ray Marker Locations 4 Product Specifications 4 Instrument Set 5 Step 1 Preoperative Planning 8 Patient Positioning 8 Step 2 Disc Removal
1 CONTENTS InterFuse T Product Description 3 Indications for Use 3 X-Ray Marker Locations 4 Product Specifications 4 Instrument Set 5 Step 1 Preoperative Planning 8 Patient Positioning 8 Step 2 Disc Removal
Subject: Non-Invasive Electrical Bone Growth Stimulators (EBGS)
 09-E0000-22 Original Effective Date: 06/15/00 Reviewed: 04/28/16 Revised: 05/15/16 Subject: Non-Invasive Electrical Bone Growth Stimulators (EBGS) THIS MEDICAL COVERAGE GUIDELINE IS NOT AN AUTHORIZATION,
09-E0000-22 Original Effective Date: 06/15/00 Reviewed: 04/28/16 Revised: 05/15/16 Subject: Non-Invasive Electrical Bone Growth Stimulators (EBGS) THIS MEDICAL COVERAGE GUIDELINE IS NOT AN AUTHORIZATION,
Surgical Technique. Apache Anterior Lumbar Interbody Fusion
 Surgical Technique Apache Anterior Lumbar Interbody Fusion 2 Table of Contents Page Preoperative Planning 4 Patient Positioning 4 Disc and Endplate Preparation 4 Distraction/Size Selection 5 Implantation
Surgical Technique Apache Anterior Lumbar Interbody Fusion 2 Table of Contents Page Preoperative Planning 4 Patient Positioning 4 Disc and Endplate Preparation 4 Distraction/Size Selection 5 Implantation
Patient Information. Spinal Fusion Using the ST360 or Silhouette Pedicle Screw System
 Patient Information Spinal Fusion Using the ST360 or Silhouette Pedicle Screw System Spinal Fusion Using the ST360 or Silhouette Pedicle Screw System Your doctor has recommended spinal fusion surgery using
Patient Information Spinal Fusion Using the ST360 or Silhouette Pedicle Screw System Spinal Fusion Using the ST360 or Silhouette Pedicle Screw System Your doctor has recommended spinal fusion surgery using
PROCEDURES WE PERFORM
 PROCEDURES WE PERFORM Decompression, Stabilization, and More North American Spine offers a family of advanced, minimally invasive procedures that are highly effective in treating most forms of chronic
PROCEDURES WE PERFORM Decompression, Stabilization, and More North American Spine offers a family of advanced, minimally invasive procedures that are highly effective in treating most forms of chronic
Regions of the Spine
 Anatomy The spine is a very complex mechanical structure that is highly flexible yet very strong and stable. In the normal spine, regardless of your position or activity, including sleeping, there is always
Anatomy The spine is a very complex mechanical structure that is highly flexible yet very strong and stable. In the normal spine, regardless of your position or activity, including sleeping, there is always
Fusion in MISS. Guillem Saló Bru, MD, Phd
 Fusion in MISS. Guillem Saló Bru, MD, Phd Orthopaedic Department. Spine Unit. Hospital del Mar. Barcelona. Associated Professor. Universitat Autónoma de Barcelona. Introduction. Spinal fusion: treatment
Fusion in MISS. Guillem Saló Bru, MD, Phd Orthopaedic Department. Spine Unit. Hospital del Mar. Barcelona. Associated Professor. Universitat Autónoma de Barcelona. Introduction. Spinal fusion: treatment
The Role of Surgery in the Treatment of Low Back Pain and Radiculopathy. Christian Etter, MD, Spine Surgeon Zürich, Switzerland
 The Role of Surgery in the Treatment of Low Back Pain and Radiculopathy Christian Etter, MD, Spine Surgeon Zürich, Switzerland WW Fusion Volume by Disorder 2004E % Tumor/Trauma 11% Deformity 15% Degeneration
The Role of Surgery in the Treatment of Low Back Pain and Radiculopathy Christian Etter, MD, Spine Surgeon Zürich, Switzerland WW Fusion Volume by Disorder 2004E % Tumor/Trauma 11% Deformity 15% Degeneration
Original Article Management of Single Level Lumbar Degenerative Spondylolisthesis: Decompression Alone or Decompression and Fusion
 Egyptian Journal of Neurosurgery Volume 9 / No. 4 / October - December 014 51-56 Original Article Management of Single Level Lumbar Degenerative Spondylolisthesis: Decompression Alone or Decompression
Egyptian Journal of Neurosurgery Volume 9 / No. 4 / October - December 014 51-56 Original Article Management of Single Level Lumbar Degenerative Spondylolisthesis: Decompression Alone or Decompression
