Rapid Visible Improvements in Onychomycosis: An Open-Label Clinical Investigation of Kerasal Nail TM
|
|
|
- Gwendolyn Cameron Taylor
- 5 years ago
- Views:
Transcription
1 Journal of Cosmetics, Dermatological Sciences and Applications, 2017, 7, ISSN Online: ISSN Print: Rapid Visible Improvements in Onychomycosis: An Open-Label Clinical Investigation of Kerasal Nail TM Jiun Yit Pan 1, Chris Lixian Tan 2, Anders Toft 3* 1 National Skin Centre, Singapore City, Singapore 2 Division of Dermatology, National University Hospital, Singapore City, Singapore 3 Moberg Pharma AB, Bromma, Sweden How to cite this paper: Pan, J.Y., Tan, C.L. and Toft, A. (2017) Rapid Visible Improvements in Onychomycosis: An Open-Label Clinical Investigation of Kerasal Nail TM. Journal of Cosmetics, Dermatological Sciences and Applications, 7, Received: February 4, 2017 Accepted: March 10, 2017 Published: March 13, 2017 Copyright 2017 by authors and Scientific Research Publishing Inc. This work is licensed under the Creative Commons Attribution International License (CC BY 4.0). Open Access Abstract Traditional treatments for onychomycosis, a fungal disease primarily affecting the toenails, are associated with poor tolerability or inadequate efficacy. Moreover, because many patients find onychomycosis embarrassing, achieving rapid cosmetic improvements is desirable. Kerasal Nail TM is a marketed topical treatment containing propylene glycol, urea and lactic acid. The ability of Kerasal Nail to improve the appearance of an affected nail was investigated in a 4-week open-label study. Patients aged 21 years and with 25% to 75% of a big toenail or thumbnail (the target nail) affected by clinically suspected onychomycosis (n = 65) were instructed to apply Kerasal Nail to all their affected nails once daily for 4 weeks. After 1, 2 and 4 weeks of treatment, they assessed the appearance of the target nail on a 4-point scale, with a score of 2 indicating a minimum of some improvement. They also assessed the nail for improvements in thickening, discoloration, brittleness and softness. Adverse events (AEs) occurring between administration of the first dose of Kerasal Nail and the end of the 4-week treatment period were recorded. 85% of patients (95% confidence interval [CI] 74 to 92) achieved some improvement in the target nail at 4 weeks. Improvement of the target nail was reported by 65% of patients (95% CI 52 to 76) after 1 week of treatment and by 82% of patients (95% CI 70 to 90) after 2 weeks. 55% of patients achieved some improvement in discoloration in the first week of treatment. After 4 weeks of treatment, discoloration, thickening, brittleness and softness were each improved in more than half of patients. Treatment-related AEs were limited to two cases of administration site pain. Overall treatment compliance was 99%. In summary, Kerasal Nail produced very rapid improvements in nail appearance in patients with onychomycosis and was well tolerated. DOI: /jcdsa March 13, 2017
2 Keywords Onychomycosis, Kerasal Nail, Asian, Topical, 4 Weeks 1. Introduction Onychomycosis, a fungal infection of the toenails and (less frequently) the fingernails, is characterized by discoloration of the nail plate, subungual hyperkeratosis/debris and onycholysis (detachment of all or part of the nail plate from the nail bed). It has been estimated to affect as many as a fifth of people in Europe and East Asia [1]. Its prevalence is higher in females than in males [2] and increases with age [1] [3]. Onychomycosis does not resolve spontaneously and if left untreated it can be transmitted to family members [4]. While not dangerous per se, it often causes pain and discomfort and can worsen health-related quality of life, particularly in patients with persistent symptoms [5]. It can be difficult to cure, with successful treatment taking up to 1 year [1]. Moreover, the relapse rate may be as high as 25% [6]. Treatment options for onychomycosis include laser therapy, removal of the affected nail by surgery or with chemicals, and various antifungal agents, applied topically or consumed systemically. Oral antifungals such as terbinafine and itraconazole are effective but cause unpleasant side effects such as headache, nausea, diarrhea and skin reactions [7]. They also have the potential for adverse liver effects and drug interactions, which may limit their use in the elderly the population that is most likely to suffer from onychomycosis [8]. Conversely, topical agents such as amorolfine and ciclopirox, normally applied to the nail plate as lacquers, do not generally cause systemic toxicity but have inadequate efficacy because of their limited ability to penetrate the nail plate [9] [10]. Kerasal Nail TM is a marketed topical treatment containing propylene glycol, urea and lactic acid. In one early study, a different test solution containing these same three ingredients cured or yielded improvements in 21 of 23 patients with culture-positive onychomycosis when used for 2 to 6 months [11]. A subsequent 24-week randomized double-blind placebo-controlled study in 493 patients with distal subungual onychomycosis investigated K101, which is similar to Kerasal Nail but has a higher urea content (20% vs 9.5%) [12]. The mycological cure rate was higher for K101 versus placebo and a greater proportion of patients treated with K101 reported improvement in their disease. Even after successful therapy with systemic or topical antifungals, affected nails may not have a normal appearance because clearance of signs of infection typically requires turnover of the nail plate [13]. Patients with onychomycosis often suffer from embarrassment and other psychosocial problems [14] [15] [16] and would therefore benefit from alternative treatments that rapidly enhance the cosmetic appearance of affected nails. Because approximately three-quarters of patients treated with K101 in the 24-week study reported at least some im- 58
3 provement by week 8 [12], a further study specifically investigated improvement in appearance after 8 weeks of K101 treatment in 75 patients with onychomycosis [17]. The proportion of patients who reported at least some improvement in an affected nail was 77% at 2 weeks, 88% at 4 weeks and 92% at 8 weeks. No adverse events (AEs) judged to be related to K101 were reported. The aim of the present study was to investigate the effects of Kerasal Nail in terms of overall appearance and specific physical attributes during and after 4 weeks of treatment. 2. Methods An open-label, single-arm baseline-controlled 4-week post-marketing clinical investigation to evaluate the ability of Kerasal Nail to provide rapid improvements in nail appearance in patients with onychomycosis was conducted at two sites in Singapore (a university hospital and a dermatology clinic) in Kerasal Nail is a clear colorless solution whose ingredients are propylene glycol, urea, lactic acid, disodium EDTA, glycerin, sodium hydroxide and water. It is supplied in a plastic tube with a silicone tip to facilitate application. The final study protocol (dated 20 Oct 2015) and the patient information and consent form (version 1.1, dated 23 Dec 2015) were approved, in writing, by the National Healthcare Group (NHG) Domain Specific Review Board (DSRB) on 08 Jan 2016, before the first patient was enrolled in the study. The study was conducted in accordance with the Declaration of Helsinki and ICH E6 Good Clinical Practice. Male and female subjects who fulfilled the following inclusion criteria were invited to participate: i) age 21 years, ii) 25% to 75% of a big toenail or thumbnail (the target nail) affected by clinically suspected onychomycosis and (iii) willingness and ability to provide signed written informed consent. The following exclusion criteria were applied: i) proximal subungual onychomycosis, ii) other conditions known to cause an abnormal nail appearance, iii) topical treatment of the nail with an antifungal medication or systemic use of an antifungal treatment in the previous month, iv) participation in another clinical study of an investigational drug or device during the previous 4 weeks and v) known allergy to any of the ingredients in the study treatment. The study comprised a screening visit and follow-up visits at 1, 2 and 4 weeks. At the screening visit, patients were assessed for demographics, medical history and prior and concomitant medications and underwent a physical examination of the hands and feet. They received instructions on how to use Kerasal Nail and applied it to all their affected nails under supervision. They were instructed to thereafter apply Kerasal Nail to all affected nails once daily at bedtime for 4 weeks. Compliance was assessed at each follow-up visit by asking patients how many days per week on average they had applied the study treatment since the previous study visit. At screening, the target nail was selected for follow-up assessments. Images of the target nail were captured by standardized photography at the screening visit 59
4 and the 1-, 2- and 4-week follow-up visits. After 1, 2 and 4 weeks of treatment, patients assessed the appearance of the target nail compared to baseline using a 4-point global assessment scale: 1 = No improvement, 2 = Some improvement, 3 = Clear improvement and 4 = Very good improvement. They were also asked whether, compared to baseline, the target nail had become i) less thickened, ii) less discolored, iii) less brittle and iv) softer (Yes/No/Cannot determine). The primary endpoint was the proportion of patients who experienced at least some improvement in the target nail after 4 weeks of treatment, based on a score of 2 on the global assessment scale. The secondary endpoints were the proportions of patients who experienced i) at least some improvement in the target nail at 1 and 2 weeks and ii) improvements in thickening, discoloration, brittleness and softness at 1, 2 and 4 weeks. AEs that occurred between administration of the first dose of Kerasal Nail and the end of the 4-week treatment period, whether judged to be related to the study treatment or not, were recorded at each study visit. AEs were classified based on severity, seriousness and relationship to study treatment. All patients who used the study product at least once were included in the safety set; those who additionally underwent at least one post-baseline assessment were included in the full analysis set (FAS). The per-protocol set (PPS) comprised patients who had i) at least 80% treatment compliance during the 4-week study period, ii) no major protocol deviations and iii) a score for the global assessment scale at 4 weeks. The primary efficacy analysis was based on the FAS; missing values were imputed by the last observation carried forward method. If a value for the 1-week follow-up visit was missing, the baseline value was imputed. Based on previous studies of similar products, it was determined that a success rate of 50% with a 95% confidence interval of ±0.123 would be sufficient to confirm early visible improvements in target nail appearance (success being defined as a score of 2 on the 4-point global assessment scale after 4 weeks of treatment). It was calculated that 65 patients would need to be enrolled to achieve this. Statistical analyses were performed using STATA 14.0 (StataCorp LP, Texas, USA). Continuous data are presented as descriptive statistics: number of observations (n), mean and standard deviation (SD). Categorical data are presented as frequency (n) and percentage (%). Two-sided 95% confidence intervals (CIs) were computed using the Clopper-Pearson (exact) method. No adjustments were made for possible differences according to study site. 3. Results 68 subjects were screened and 65 of them were enrolled in the study. The first patient was enrolled on 19 Jan 2016 and the last patient completed the study on 15 Apr All 65 participants completed the study and were included in the FAS and safety set. Eight patients were excluded from the PPS because of inadequate treatment compliance (n = 1) or major protocol deviations (n = 7). Mean 60
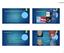
5 treatment compliance was 99% overall (median 100%, range 76 to 100) and did not deteriorate over the course of the study (Table 1). The mean age of participants was 53 years (median 53 years, range 26 to 86) and 29 (45%) were female (Table 2). As shown in Table 3, 85% of patients (55/65) achieved at least some visible improvement in the target nail at 4 weeks (95% CI 74 to 92). 65% of patients (42/65) experienced some improvement of the target nail after 1 week of treatment (95% CI 52 to 76) and 82% of them (53/65) experienced some improvement after 2 weeks (95% CI 70 to 90). Figure 1 shows examples of improvements in a big toenail and a thumbnail during the 4-week treatment period. More than half of patients achieved some improvement in discoloration in the first week of treatment (Table 4). Approximately half of patients showed reduced nail thickening by the end of the second week of treatment. After 4 weeks of treatment, discoloration, thickening, brittleness and softness were improved in more than half of patients (range 51% for brittleness to 72% for discoloration) (Table 4). There were 3 AEs in 3 patients (5%). All AEs were mild and none of them were serious. Two of the 3 AEs were pain at the administration site assessed as probably related to the study product; the other AE was a case of folliculitis judged not to be related to the study product. No patients discontinued treatment or were withdrawn from the study because of AEs. Table 1. Treatment compliance according to information provided at the 1-, 2- and 4-week follow-up visits (safety set). Treatment period Compliance Week 1 a Mean (SD) 99.6 (2.5) Median 100 Range 86 to 100 Week 2 Mean (SD) 98.7 (9.0) Median 100 Range 29 to 100 Weeks 3 to 4 Mean (SD) 99.8 (1.8) Median 100 Range 86 to 100 Weeks 1 to 4 Mean (SD) 99.3 (3.1) Median 100 Range 76 to 100 a Data missing for one patient. 61
6 Table 2. Demographics and baseline characteristics (full analysis set/safety set). N = 65 Age (years) Mean (SD) 52.7 (13.0) Median 53 Range 26 to 86 Missing (n) 0 Sex, n (%) Female 29 (45) Male 36 (55) Race, n (%) Asian or Pacific Islander 64 (98) Caucasian 1 (2) Fingernails/toenails, n (%) Normal 0 (0) Abnormal 65 (100) Skin on hands/feet, n (%) Normal 57 (88) Abnormal 8 (12) Table 3. Changes in overall appearance of the target nail based on global assessment scale score (full analysis set, last observation carried forward). Appearance of target nail compared to baseline 1 week 2 weeks 4 weeks n % n % n % 1. No improvement Some improvement Clear improvement Very good improvement At least some improvement Percentage values are correct to the nearest whole number. The values for 1 week and 2 weeks add up to 99 (rather than 100) because of rounding. (a) (b) Figure 1. Visible improvements in target nails from baseline to 4 weeks. (a) A left big toenail. (b) A left thumbnail. 62
7 Table 4. Proportions of patients experiencing improvements in individual target nail attributes after 1, 2 and 4 weeks of treatment (full analysis set, last observation carried forward). Target nail attribute 1 week 2 weeks 4 weeks n % 95% CI a n % 95% CI a n % 95% CI a Thickening to to to 80 Discoloration to to to 83 Brittleness to to to 63 Softness to to to 75 a For the % values. 4. Discussion In this 4-week open-label study, 85% of patients with onychomycosis reported at least some improvement in the appearance of an affected nail after treatment with Kerasal Nail for 4 weeks (the primary endpoint). 65% of patients experienced visible improvement after just 1 week of treatment and 82% after 2 weeks. These values are comparable to those from the 8-week study of K101, in which 77% of patients reported at least some visible improvement at 2 weeks and 88% at 4 weeks [17]. As with K101 [17], improvements in thickening, discoloration, brittleness and softness were observed at 4 weeks, with discoloration the first physical attribute to show improvement in more than half of participants. Previous studies have highlighted the embarrassment and stigmatization many onychomycosis patients suffer. In a US telephone survey of 258 patients with confirmed onychomycosis, 74% of participants reported a feeling of embarrassment [14]. Other studies describe low self-esteem and social isolation in patients [15]. In a recent online survey, 1017 Hong Kong residents were shown photographs of people with and without onychomycosis and asked questions about them. Respondents were less likely to engage in sporting or social activities with onychomycosis patients and more likely to feel uncomfortable in their presence [16]. Rapid cosmetic improvements such as those seen here with Kerasal Nail are important for onychomycosis patients to feel better about themselves and to avoid being or feeling stigmatized. Because sustained treatment is typically needed to cure onychomycosis, adherence to therapy is very important. While the factors determining treatment adherence in onychomycosis are poorly understood, one study found that frequency of therapy was a key consideration for patients [18]. Another study reported that the main reasons for non-adherence include AEs and patients misinterpreting visible improvements as evidence that they were already cured [17]. In the present study, patient-rated improvements in affected nails were achieved with few treatment-related AEs. As with K101 [17], mean overall adherence was extremely high (99%). This may both explain the rapid results and reflect the convenience and ease of use of Kerasal Nail. The only treatment-related AEs in the present study were transient pain at the administration site in 2 patients. There were no treatment-related AEs in the 63
8 8-week study of K101 [17], in which, as with Kerasal Nail in the present study, the treatment was applied using a silicone tip. The silicone tip may facilitate application to the nail, thereby limiting accidental contact with the adjacent skin. This in turn may prevent local irritation and pain such as that seen in 6% of participants in the 24-week study of K101 [12], in which K101 was applied dropwise. Limitations of the present study include the lack of an active or placebo control group and the associated lack of blinding. Participants were aware that they were receiving an active treatment and this may have affected their scoring of the target nail. This is important given that all the study s efficacy variables were subjective. Also, the participants were recruited in Singapore at a single university hospital and a single dermatology clinic and may therefore not be representative of the spectrum of onychomycosis patients in South-East Asia or even Singapore. Furthermore, 64 of the 65 participants had Asian or Pacific Islander as their race, so the results of the present study may not necessarily be generalizable to patients of other races. However, the results were similar to those obtained in the 8-week study of K101 [17], which was conducted in Sweden. This indicates that Kerasal Nail is equally effective in populations of different ethnicities. Moreover, the patient sample in the present study covered both sexes and a wide age range (26 to 86 years). 5. Conclusion In conclusion, Kerasal Nail elicited rapid effects in patients with onychomycosis, with two-thirds of patients reporting improvements in affected nails after just 1 week of treatment. The excellent overall treatment compliance suggests that the study participants felt motivated to adhere to treatment, possibly because of the effectiveness, tolerability and convenience of using Kerasal Nail. This is important because achieving optimal therapeutic results will typically take much longer compared to the early visible effects seen in this study. Users of Kerasal Nail should therefore be cautioned against discontinuing treatment prematurely. Acknowledgements P. J. Y. thanks Dr. Benson Yeo, Dr. Joel Lim and Dr. Teoh Yee Leng for helping to recruit patients, and Yan Qin, clinical trial coordinator at the National Skin Centre, Singapore. C. L. T. thanks Dr. Derrick Aw and Dr. Aditi Sobti for helping to recruit patients, and Mr. Nick Choi, clinical trial coordinator at the Division of Dermatology, National University Hospital, Singapore. A. T, on behalf of Moberg Pharma AB, thanks the patients and staff who participated in the study. The authors also acknowledge Stephen Gilliver of TFS AB for medical writing services, paid for by Moberg Pharma AB. Moberg Pharma AB funded the study and supplied the study product. Execution of the study, data analysis and reporting were outsourced to a contract resource organization. References [1] Ghannoum, M. and Isham, N. (2014) Fungal Nail Infections (Onychomycosis): A 64
9 Never-Ending Story? PLoS Pathogens, 10, e [2] Gupta, A.K., Jain, H.C., Lynde, C.W., Macdonald, P., Cooper, E.A. and Summerbell, R.C. (2000) Prevalence and Epidemiology of Onychomycosis in Patients Visiting Physicians Offices: A Multicenter Canadian Survey of 15,000 Patients. Journal of the American Academy of Dermatology, 43, [3] Elewski, B.E. and Charif, M.A. (1997) Prevalence of Onychomycosis in Patients Attending a Dermatology Clinic in Northeastern Ohio for Other Conditions. Archives of Dermatology, 133, [4] Tosti, A. and Elewski, B.E. (2016) Onychomycosis: Practical Approaches to Minimize Relapse and Recurrence. Skin Appendage Disorders, 2, [5] Milobratović, D., Janković, S., Vukičević, J., Marinković, J., Janković, J. and Railić, Z. (2013) Quality of Life in Patients with Toenail Onychomycosis. Mycoses, 56, [6] Scher, R.K. and Baran, R. (2003) Onychomycosis in Clinical Practice: Factors Contributing to Recurrence. British Journal of Dermatology, 149, [7] Gupta, A.K. and Shear, N.H. (2000) A Risk-Benefit Assessment of the Newer Oral Antifungal Agents Used to Treat Onychomycosis. Drug Safety, 22, [8] Elewski, B. and Tavakkol, A. (2005) Safety and Tolerability of Oral Antifungal Agents in the Treatment of Fungal Nail Disease: A Proven Reality. Therapeutics and Clinical Risk Management, 1, [9] Lauharanta, J. (1992) Comparative Efficacy and Safety of Amorolfine Nail Lacquer 2% versus 5% Once Weekly. Clinical and Experimental Dermatology, 17, [10] Alley, M.R.K., Baker, S.J., Beutner, K.R. and Plattner, J. (2007) Recent Progress on the Topical Therapy of Onychomycosis. Expert Opinion on Investigational Drugs, 16, [11] Faergemann, J. and Swanbeck, G. (1989) Treatment of Onychomycosis with a Propylene Glycol-Urea-Lactic Acid Solution. Mycoses, 32, [12] Emtestam, L., Kaaman, T. and Rensfeldt, K. (2012) Treatment of Distal Subungual Onychomycosis with a Topical Preparation of Urea, Propylene Glycol and Lactic Acid: Results of a 24-Week, Double-Blind, Placebo-Controlled Study. Mycoses, 55, [13] Scher, R.K., Tavakkol, A., Sigurgeirsson, B., Hay, R.J., Joseph, W.S., Tosti, A., Fleckman, P., Ghannoum, M., Armstrong, D.G., Markinson, B.C. and Elewski, B.E. (2007) Onychomycosis: Diagnosis and Definition of Cure. Journal of the American Academy of Dermatology, 56, [14] Drake, L.A., Scher, R.K., Smith, E.B., Faich, G.A., Smith, S.L., Hong, J.J. and Stiller, M.J. (1998) Effect of Onychomycosis on Quality of Life. Journal of the American Academy of Dermatology, 38, [15] Chacon, A., Franca, K., Fernandez, A. and Nouri, K. (2013) Psychosocial Impact of Onychomycosis: A Review. International Journal of Dermatology, 52,
10 [16] Chan, H.H., Wong, E.T. and Yeung, C.K. (2014) Psychosocial Perception of Adults with Onychomycosis: A Blinded, Controlled Comparison of 1017 Adult Hong Kong Residents with or without Onychomycosis. BioPsychoSocial Medicine, 8, [17] Faergemann, J., Gullstrand, S. and Rensfeldt, K. (2011) Early and Visible Improvements after Application of K101 in the Appearance of Nails Discoloured and Deformed by Onychomycosis. Journal of Cosmetics, Dermatological Sciences and Applications, 1, [18] Nolting, S.K., Sanchez Carazo, S., De Boulle, K. and Lambert, J.R. (1998) Oral Treatment Schedules for Onychomycosis: A Study of Patient Preference. International Journal of Dermatology, 37, Submit or recommend next manuscript to SCIRP and we will provide best service for you: Accepting pre-submission inquiries through , Facebook, LinkedIn, Twitter, etc. A wide selection of journals (inclusive of 9 subjects, more than 200 journals) Providing 24-hour high-quality service User-friendly online submission system Fair and swift peer-review system Efficient typesetting and proofreading procedure Display of the result of downloads and visits, as well as the number of cited articles Maximum dissemination of your research work Submit your manuscript at: Or contact jcdsa@scirp.org 66
Diagnosis,Therapy and Prophylaxis of Fungal Diseases
 mycoses Diagnosis,Therapy and Prophylaxis of Fungal Diseases Original article Treatment of distal subungual onychomycosis with a topical preparation of urea, propylene glycol and lactic acid: results of
mycoses Diagnosis,Therapy and Prophylaxis of Fungal Diseases Original article Treatment of distal subungual onychomycosis with a topical preparation of urea, propylene glycol and lactic acid: results of
Fungal Resistance, Biofilm, and Its Impact In the Management of Nail Infection
 Fungal Resistance, Biofilm, and Its Impact In the Management of Nail Infection Faculty Raza Aly, PhD, MPH Professor Emeritus University of California Medical Center (MSSF) Professor, Dermatology Faculty
Fungal Resistance, Biofilm, and Its Impact In the Management of Nail Infection Faculty Raza Aly, PhD, MPH Professor Emeritus University of California Medical Center (MSSF) Professor, Dermatology Faculty
Classification. Distal & Lateral Subungual OM. White Superficial OM. Proximal Subungual OM. Candidal OM. Total dystrophic OM
 Onychomycosis Commonest dermatological condition Definition: Infection of the nail caused by fungi that include dermatophytes, non-dermatophyte moulds and yeasts (mainly Candida). 80% of all OM affects
Onychomycosis Commonest dermatological condition Definition: Infection of the nail caused by fungi that include dermatophytes, non-dermatophyte moulds and yeasts (mainly Candida). 80% of all OM affects
Clinical Trial Designs for Topical Antifungal Treatments of Onychomycosis and Implications on Clinical Practice
 Clinical Trial Designs for Topical Antifungal Treatments of Onychomycosis and Implications on Clinical Practice Phoebe Rich, MD; Tracey C. Vlahovic, DPM; Warren S. Joseph, DPM; Lee T. Zane, MD; Steve B.
Clinical Trial Designs for Topical Antifungal Treatments of Onychomycosis and Implications on Clinical Practice Phoebe Rich, MD; Tracey C. Vlahovic, DPM; Warren S. Joseph, DPM; Lee T. Zane, MD; Steve B.
Laser Treatment of Onychomycosis. Description
 Subject: Laser Treatment of Onychomycosis Page: 1 of 8 Last Review Status/Date: September 2014 Laser Treatment of Onychomycosis Description Onychomycosis is a common fungal infection of the nail. Currently
Subject: Laser Treatment of Onychomycosis Page: 1 of 8 Last Review Status/Date: September 2014 Laser Treatment of Onychomycosis Description Onychomycosis is a common fungal infection of the nail. Currently
Clinical Evaluation of Safety and Efficacy of a New Topical Treatment for Onychomycosis
 October 2011 1186 Volume 10 Issue 10 Copyright 2011 ORIGINAL ARTICLES Clinical Evaluation of Safety and Efficacy of a New Topical Treatment for Onychomycosis Adnan Nasir MD,a Beth Goldstein MD,b Martin
October 2011 1186 Volume 10 Issue 10 Copyright 2011 ORIGINAL ARTICLES Clinical Evaluation of Safety and Efficacy of a New Topical Treatment for Onychomycosis Adnan Nasir MD,a Beth Goldstein MD,b Martin
UPDATE ON ONYCHOMYCOSIS TREATMENTS
 UPDATE ON ONYCHOMYCOSIS TREATMENTS Tracey C. Vlahovic, DPM FFPM RCPS (Glasg) Clinical Professor, Dept. of Podiatric Medicine Temple University School of Podiatric Medicine, Philadelphia, PA CONFLICTS OF
UPDATE ON ONYCHOMYCOSIS TREATMENTS Tracey C. Vlahovic, DPM FFPM RCPS (Glasg) Clinical Professor, Dept. of Podiatric Medicine Temple University School of Podiatric Medicine, Philadelphia, PA CONFLICTS OF
The US Onychomycosis Market (2017 Edition) July 2017
 The US Onychomycosis Market (2017 Edition) July 2017 The US Onychomycosis Market Report Scope of the Report The report entitled The US Onychomycosis Market (2017 Edition), provides analysis of the US onychomycosis
The US Onychomycosis Market (2017 Edition) July 2017 The US Onychomycosis Market Report Scope of the Report The report entitled The US Onychomycosis Market (2017 Edition), provides analysis of the US onychomycosis
 Page 1 of 5 Management strategies for onychomycosis in special patient populations Phoebe Rich, Nathaniel J. Jellinek and David M. Pariser Dermatology News. 46.8 (Aug. 2015): ps13. Copyright: COPYRIGHT
Page 1 of 5 Management strategies for onychomycosis in special patient populations Phoebe Rich, Nathaniel J. Jellinek and David M. Pariser Dermatology News. 46.8 (Aug. 2015): ps13. Copyright: COPYRIGHT
A class IIa medical device intended for mild-to-moderate fungal nail infection PRODUCT MONOGRAPH
 A class IIa medical device intended for mild-to-moderate fungal nail infection PRODUCT MONOGRAPH AWB-2052628721 Date of Preparation March 2017 Introduction to Bayer Bayer is a Life Science company with
A class IIa medical device intended for mild-to-moderate fungal nail infection PRODUCT MONOGRAPH AWB-2052628721 Date of Preparation March 2017 Introduction to Bayer Bayer is a Life Science company with
2.0 Synopsis. Adalimumab R&D/04/118. (For National Authority Use Only) Referring to Part of Dossier: Volume:
 2.0 Synopsis Abbott Laboratories Name of Study Drug: Adalimumab Name of Active Ingredient: Adalimumab Title of Study: Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority
2.0 Synopsis Abbott Laboratories Name of Study Drug: Adalimumab Name of Active Ingredient: Adalimumab Title of Study: Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority
DISTAL LATERAL SUBUNGUAL ONYCHOMYCOSIS
 Boni E. Elewski, MD James Elder Professor of Dermatology University of Alabama RESEARCH GRANTS -TO UNIVERSITY Dusa, Meiji, Valeant, Viamet Amgen, Abbvie, Boehringer Ingelheim, Celgene, Lilly, Merck, Novartis,
Boni E. Elewski, MD James Elder Professor of Dermatology University of Alabama RESEARCH GRANTS -TO UNIVERSITY Dusa, Meiji, Valeant, Viamet Amgen, Abbvie, Boehringer Ingelheim, Celgene, Lilly, Merck, Novartis,
Therapeutics for the Clinician. Efinaconazole Solution 10%: Topical Antifungal Therapy for Toenail Onychomycosis CUTIS
 Solution 10%: Topical Antifungal Therapy for Toenail Onychomycosis Antonella Tosti, MD Practice Points Toenail onychomycosis is a common disease with limited treatment options, as treatment failures and
Solution 10%: Topical Antifungal Therapy for Toenail Onychomycosis Antonella Tosti, MD Practice Points Toenail onychomycosis is a common disease with limited treatment options, as treatment failures and
Laser Treatment of Onychomycosis
 Laser Treatment of Onychomycosis Policy Number: 2.01.89 Last Review: 5/2018 Origination: 11/2015 Next Review: 11/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide coverage
Laser Treatment of Onychomycosis Policy Number: 2.01.89 Last Review: 5/2018 Origination: 11/2015 Next Review: 11/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will not provide coverage
TRANSPARENCY COMMITTEE OPINION. 27 January 2010
 The legally binding text is the original French version TRAPARENCY COMMITTEE OPINION 27 January 2010 80 mg/g, medicated nail lacquer B/1 glass vial of 3.3 ml (CIP: 395 010-4) B/1 glass vial of 6.6 ml (CIP:
The legally binding text is the original French version TRAPARENCY COMMITTEE OPINION 27 January 2010 80 mg/g, medicated nail lacquer B/1 glass vial of 3.3 ml (CIP: 395 010-4) B/1 glass vial of 6.6 ml (CIP:
The Safety of Walking Space for the Elderly People Living in Communities in Beijing, China
 Open Journal of Social Sciences, 2017, 5, 153-158 http://www.scirp.org/journal/jss ISSN Online: 2327-5960 ISSN Print: 2327-5952 The Safety of Walking Space for the Elderly People Living in Communities
Open Journal of Social Sciences, 2017, 5, 153-158 http://www.scirp.org/journal/jss ISSN Online: 2327-5960 ISSN Print: 2327-5952 The Safety of Walking Space for the Elderly People Living in Communities
Medical Policy. MP Laser Treatment of Onychomycosis
 Medical Policy MP 2.01.89 BCBSA Ref. Policy: 2.01.89 Last Review: 12/27/2017 Effective Date: 12/27/2017 Section: Medicine Related Policies 2.01.71 Nonpharmacologic Treatment of Rosacea 9.01.502 Experimental
Medical Policy MP 2.01.89 BCBSA Ref. Policy: 2.01.89 Last Review: 12/27/2017 Effective Date: 12/27/2017 Section: Medicine Related Policies 2.01.71 Nonpharmacologic Treatment of Rosacea 9.01.502 Experimental
RESULTS. TABLE 1 FOOT, TOE, AND TOENAIL EXAMINATION CONDITION (Number of patients with)
 Over the several decades that I have been in practice I have been struggling to provide my patients with onychomycosis an effective treatment for this pervasive and embarrassing disease. I have had several
Over the several decades that I have been in practice I have been struggling to provide my patients with onychomycosis an effective treatment for this pervasive and embarrassing disease. I have had several
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
INTRODUCTION. Rapid clearance of sub-ungual onychomycosis by controlled micro penetration and topical terbinafine solution using the Clearanail device
 Rapid clearance of sub-ungual onychomycosis by controlled micro penetration and topical terbinafine solution using the Clearanail device Dr Ivan Bristow, Faculty of Health Sciences, University of Southampton
Rapid clearance of sub-ungual onychomycosis by controlled micro penetration and topical terbinafine solution using the Clearanail device Dr Ivan Bristow, Faculty of Health Sciences, University of Southampton
Clinical clearing of moderate and severe onychomycosis with the Nd:YAG 1064nm laser and post treatment prevention with tolnaftate
 Clinical clearing of moderate and severe onychomycosis with the Nd:YAG 1064nm laser and post treatment prevention with tolnaftate by Myron A. Bodman DPM 1*, Marie Mantini Blazer DPM 1, Bryan D. Caldwell
Clinical clearing of moderate and severe onychomycosis with the Nd:YAG 1064nm laser and post treatment prevention with tolnaftate by Myron A. Bodman DPM 1*, Marie Mantini Blazer DPM 1, Bryan D. Caldwell
2.0 Synopsis. Adalimumab M Clinical Study Report R&D/04/900. (For National Authority Use Only) Referring to Part of Dossier: Volume:
 2. Synopsis Abbott Laboratories Name of Study Drug: Name of Active Ingredient: Title of Study: Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority Use Only) Phase
2. Synopsis Abbott Laboratories Name of Study Drug: Name of Active Ingredient: Title of Study: Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority Use Only) Phase
overview of current strategies for monotherapy and combination therapy
 ORIGINAL ARTICLE JEADV (2005) 19, 21 29 DOI: 10.1111/j.1468-3083.2004.00988.x Topical antifungal drugs for the treatment of onychomycosis: an Blackwell Publishing, Ltd. overview of current strategies for
ORIGINAL ARTICLE JEADV (2005) 19, 21 29 DOI: 10.1111/j.1468-3083.2004.00988.x Topical antifungal drugs for the treatment of onychomycosis: an Blackwell Publishing, Ltd. overview of current strategies for
Clinical Study Synopsis
 Clinical Study Synopsis This document is not intended to replace the advice of a healthcare professional and should not be considered as a recommendation. Patients should always seek medical advice before
Clinical Study Synopsis This document is not intended to replace the advice of a healthcare professional and should not be considered as a recommendation. Patients should always seek medical advice before
TIME TO CLEAN IT UP. TOENAIL FUNGUS Still your dirty secret? WITH JUBLIA FIGHTING TOENAIL FUNGUS CAN BE FUN
 TOENAIL FUNGUS Still your dirty secret? TIME TO CLEAN IT UP WITH JUBLIA JubliApp FIGHTING TOENAIL FUNGUS CAN BE FUN Download JubliApp for tools, tips, and support Learn more at JubliApp.com Visit JubliaRx.com
TOENAIL FUNGUS Still your dirty secret? TIME TO CLEAN IT UP WITH JUBLIA JubliApp FIGHTING TOENAIL FUNGUS CAN BE FUN Download JubliApp for tools, tips, and support Learn more at JubliApp.com Visit JubliaRx.com
Corporate Medical Policy
 Corporate Medical Policy Laser Treatment of Onychomycosis File Name: Origination: Last CAP Review: Next CAP Review: Last Review: laser_treatment_of_onychomycosis 5/2013 11/2017 11/2018 11/2017 Description
Corporate Medical Policy Laser Treatment of Onychomycosis File Name: Origination: Last CAP Review: Next CAP Review: Last Review: laser_treatment_of_onychomycosis 5/2013 11/2017 11/2018 11/2017 Description
Source of effectiveness data The effectiveness evidence was derived from a systematic review of published studies.
 Pharmacoeconomic analysis of ciclopirox nail lacquer solution 8% and the new oral antifungal agents used to treat dermatophyte toe onychomycosis in the United States Gupta A K Record Status This is a critical
Pharmacoeconomic analysis of ciclopirox nail lacquer solution 8% and the new oral antifungal agents used to treat dermatophyte toe onychomycosis in the United States Gupta A K Record Status This is a critical
Background 1/29/2018. Lasers and Lights for Onychomycosis KEYVAN NOURI, MD. Keyvan Nouri, M.D. White superficial. onychomycosis
 Lasers and Lights for Onychomycosis KEYVAN NOURI, MD Keyvan Nouri, M.D. Professor of Dermatology, Ophthalmology, Otolaryngology & Surgery Louis C. Skinner, Jr., M.D. Endowed Chair in Dermatology Richard
Lasers and Lights for Onychomycosis KEYVAN NOURI, MD Keyvan Nouri, M.D. Professor of Dermatology, Ophthalmology, Otolaryngology & Surgery Louis C. Skinner, Jr., M.D. Endowed Chair in Dermatology Richard
Modelling A decision tree was used to estimate benefits and costs of alternative agents.
 Economic evaluation of antifungal agents in the treatment of toenail onychomycosis in Germany Van Doorslaer E K, Tormans G, Gupta A K, Van Rossem K, Eggleston A, Dubois D J, De Doncker P, Haneke E Record
Economic evaluation of antifungal agents in the treatment of toenail onychomycosis in Germany Van Doorslaer E K, Tormans G, Gupta A K, Van Rossem K, Eggleston A, Dubois D J, De Doncker P, Haneke E Record
ORIGINAL INVESTIGATION. Boni E. Elewski, MD; James Leyden, MD; Michael G. Rinaldi, PhD; Ercem Atillasoy, MD
 Office Practice Based Confirmation of Onychomycosis A US Nationwide Prospective Survey ORIGINAL INVESTIGATION Boni E. Elewski, MD; James Leyden, MD; Michael G. Rinaldi, PhD; Ercem Atillasoy, MD Background:
Office Practice Based Confirmation of Onychomycosis A US Nationwide Prospective Survey ORIGINAL INVESTIGATION Boni E. Elewski, MD; James Leyden, MD; Michael G. Rinaldi, PhD; Ercem Atillasoy, MD Background:
STUDY. A New Classification System for Grading the Severity of Onychomycosis
 STUDY A New Classification System for Grading the Severity of Onychomycosis Onychomycosis Severity Index Caitlin Carney, MD; Antonella Tosti, MD; Ralph Daniel, MD; Richard Scher, MD; Phoebe Rich, MD; Jamie
STUDY A New Classification System for Grading the Severity of Onychomycosis Onychomycosis Severity Index Caitlin Carney, MD; Antonella Tosti, MD; Ralph Daniel, MD; Richard Scher, MD; Phoebe Rich, MD; Jamie
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
BJD British Journal of Dermatology. Summary. What s already known about this topic? CLINICAL TRIALS
 CLINICAL TRIALS BJD British Journal of Dermatology Efficacy of topical resin lacquer, amorolfine and oral terbinafine for treating toenail onychomycosis: a prospective, randomized, controlled, investigator-blinded,
CLINICAL TRIALS BJD British Journal of Dermatology Efficacy of topical resin lacquer, amorolfine and oral terbinafine for treating toenail onychomycosis: a prospective, randomized, controlled, investigator-blinded,
Kent Academic Repository
 Kent Academic Repository Full text document (pdf) Citation for published version Stoeber, Joachim and Luszczynska, Aleksandra (2010) Impact factor stories: Anxiety, Stress, & Coping. Editors' Bulletin,
Kent Academic Repository Full text document (pdf) Citation for published version Stoeber, Joachim and Luszczynska, Aleksandra (2010) Impact factor stories: Anxiety, Stress, & Coping. Editors' Bulletin,
PFIZER INC. GENERIC DRUG NAME and/or COMPOUND NUMBER: 5% Spironolactone Cream/ PF
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
Moberg Pharma AB Providing Unique Products in Underserved Niches through Commercial and Innovation Excellence
 Moberg Pharma AB Providing Unique Products in Underserved Niches through Commercial and Innovation Excellence Presentation at Remium Capital Market Day, December 2, 2014 Peter Wolpert, CEO & Founder 0
Moberg Pharma AB Providing Unique Products in Underserved Niches through Commercial and Innovation Excellence Presentation at Remium Capital Market Day, December 2, 2014 Peter Wolpert, CEO & Founder 0
Effects of Tube Depth and Infusion Rate of Continuous Humidification by Endotracheal Intubation on Humidification Effect
 Open Journal of Nursing, 2017, 7, 123-127 http://www.scirp.org/journal/ojn ISSN Online: 2162-5344 ISSN Print: 2162-5336 Effects of Tube Depth and Infusion Rate of Continuous Humidification by Endotracheal
Open Journal of Nursing, 2017, 7, 123-127 http://www.scirp.org/journal/ojn ISSN Online: 2162-5344 ISSN Print: 2162-5336 Effects of Tube Depth and Infusion Rate of Continuous Humidification by Endotracheal
Investigation and Analysis on Current Situation of Coal-Burning Fluorosis Prevalence
 Open Journal of Epidemiology, 2017, 7, 115-123 http://www.scirp.org/journal/ojepi ISSN Online: 2165-7467 ISSN Print: 2165-7459 Investigation and Analysis on Current Situation of Coal-Burning Fluorosis
Open Journal of Epidemiology, 2017, 7, 115-123 http://www.scirp.org/journal/ojepi ISSN Online: 2165-7467 ISSN Print: 2165-7459 Investigation and Analysis on Current Situation of Coal-Burning Fluorosis
J. Hibler, D.O. OhioHealth - O Bleness Memorial Hospital, Athens, Ohio. AOCD Annual Conference Orlando, Florida
 J. Hibler, D.O. OhioHealth - O Bleness Memorial Hospital, Athens, Ohio AOCD Annual Conference Orlando, Florida 10.18.15 A) Onychodystrophy B) Onychogryphosis Question dogma Michael Conroy, MD C) Onychomycosis
J. Hibler, D.O. OhioHealth - O Bleness Memorial Hospital, Athens, Ohio AOCD Annual Conference Orlando, Florida 10.18.15 A) Onychodystrophy B) Onychogryphosis Question dogma Michael Conroy, MD C) Onychomycosis
Investor Presentation December 2012
 Investor Presentation December 2012 1 Safe Harbor Statement Certain items in this presentation and other matters discussed today or answers that may be given to questions asked could constitute forwardlooking
Investor Presentation December 2012 1 Safe Harbor Statement Certain items in this presentation and other matters discussed today or answers that may be given to questions asked could constitute forwardlooking
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
Activity of TDT 067 (Terbinafine in Transfersome) against Agents of Onychomycosis, as Determined by Minimum Inhibitory and Fungicidal Concentrations
 JOURNAL OF CLINICAL MICROBIOLOGY, May 2011, p. 1716 1720 Vol. 49, No. 5 0095-1137/11/$12.00 doi:10.1128/jcm.00083-11 Copyright 2011, American Society for Microbiology. All Rights Reserved. Activity of
JOURNAL OF CLINICAL MICROBIOLOGY, May 2011, p. 1716 1720 Vol. 49, No. 5 0095-1137/11/$12.00 doi:10.1128/jcm.00083-11 Copyright 2011, American Society for Microbiology. All Rights Reserved. Activity of
Study Number CAIN457C2302 (core study) and CAIN457C2302E1 (extension study)
 Clinical Trial Results Database Page 1 Sponsor Novartis Generic Drug Name Secukinumab Therapeutic Area of Trial Uveitis Approved Indication Investigational Study Number CC2302 (core study) and CC2302E1
Clinical Trial Results Database Page 1 Sponsor Novartis Generic Drug Name Secukinumab Therapeutic Area of Trial Uveitis Approved Indication Investigational Study Number CC2302 (core study) and CC2302E1
TIME TO CLEAN IT UP. TOENAIL FUNGUS Still your dirty secret? WITH JUBLIA
 TOENAIL FUNGUS Still your dirty secret? TIME TO CLEAN IT UP WITH JUBLIA PRESCRIBED BRAND FOR TOENAIL FUNGUS Visit JubliaRx.com for a $25 Instant Savings Offer* at Walgreens and other participating independent
TOENAIL FUNGUS Still your dirty secret? TIME TO CLEAN IT UP WITH JUBLIA PRESCRIBED BRAND FOR TOENAIL FUNGUS Visit JubliaRx.com for a $25 Instant Savings Offer* at Walgreens and other participating independent
Scottish Medicines Consortium
 Scottish Medicines Consortium imiquimod 5% cream (Aldara) No. (385/07) Meda Pharmaceuticals Ltd 04 April 2008 The Scottish Medicines Consortium has completed its assessment of the above product and advises
Scottish Medicines Consortium imiquimod 5% cream (Aldara) No. (385/07) Meda Pharmaceuticals Ltd 04 April 2008 The Scottish Medicines Consortium has completed its assessment of the above product and advises
Pharmacokinetics of three doses of once-weekly fluconazole (150, 300, and 450 mg) in distal subungual onychomycosis of the toenail
 SUPPORTED BY AN EDUCATIONAL GRANT FROM U.S. PHARMACEUTICALS, PFIZER INC. Pharmacokinetics of three doses of once-weekly fluconazole (150, 300, and 450 mg) in distal subungual onychomycosis of the toenail
SUPPORTED BY AN EDUCATIONAL GRANT FROM U.S. PHARMACEUTICALS, PFIZER INC. Pharmacokinetics of three doses of once-weekly fluconazole (150, 300, and 450 mg) in distal subungual onychomycosis of the toenail
Acrylate Allergy to Fake Nails: Unique Presentations of Onchodystrophy and Subungual Hyperkeratosis
 University of Vermont ScholarWorks @ UVM Family Medicine Block Clerkship, Student Projects College of Medicine 2017 Acrylate Allergy to Fake Nails: Unique Presentations of Onchodystrophy and Subungual
University of Vermont ScholarWorks @ UVM Family Medicine Block Clerkship, Student Projects College of Medicine 2017 Acrylate Allergy to Fake Nails: Unique Presentations of Onchodystrophy and Subungual
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
mg 25 mg mg 25 mg mg 100 mg 1
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Market Access CTR Summary
 Market Access CTR Summary Study No.: BEL114246 Title: Efficacy of Belimumab Treatment in a Subpopulation of Systemic Lupus Erythematosus (SLE) Patients: A Pooled Analysis of the HGS1006-C1056 (BLISS-52)
Market Access CTR Summary Study No.: BEL114246 Title: Efficacy of Belimumab Treatment in a Subpopulation of Systemic Lupus Erythematosus (SLE) Patients: A Pooled Analysis of the HGS1006-C1056 (BLISS-52)
Sponsor / Company: Sanofi Drug substance(s): HOE901-U300 (insulin glargine) According to template: QSD VERSION N 4.0 (07-JUN-2012) Page 1
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
A proven treatment for fungal nail infection
 T: 020 8472 9496 E: info@easthampodiatry.co.uk www.easthampodiatry.co.uk A proven treatment for fungal nail infection N A I L a s e r Trust NOVEON Nailaser to give you confidence in your toes Fungal nail
T: 020 8472 9496 E: info@easthampodiatry.co.uk www.easthampodiatry.co.uk A proven treatment for fungal nail infection N A I L a s e r Trust NOVEON Nailaser to give you confidence in your toes Fungal nail
How to cure toenail fungus
 P ford residence southampton, ny How to cure toenail fungus What Makes Zeta Clear The Best Fungal Nail Treatment. Zeta Clear is more effective than any other toenail fungus cream or home treatment out
P ford residence southampton, ny How to cure toenail fungus What Makes Zeta Clear The Best Fungal Nail Treatment. Zeta Clear is more effective than any other toenail fungus cream or home treatment out
Ann Dermatol Vol. 29, No. 6,
 pissn 1013-9087ㆍeISSN 2005-3894 Ann Dermatol Vol. 29, No. 6, 2017 https://doi.org/10.5021/ad.2017.29.6.699 ORIGINAL ARTICLE Randomized Clinical Trial to Evaluate the Efficacy and Safety of Combination
pissn 1013-9087ㆍeISSN 2005-3894 Ann Dermatol Vol. 29, No. 6, 2017 https://doi.org/10.5021/ad.2017.29.6.699 ORIGINAL ARTICLE Randomized Clinical Trial to Evaluate the Efficacy and Safety of Combination
International Journal of Scientific & Engineering Research Volume 9, Issue 4, April ISSN Laser Treatment of Onychomycosis
 International Journal of Scientific & Engineering Research Volume 9, Issue 4, April-2018 712 Laser Treatment of Onychomycosis Olga Olisova, MD, PhD 1, Igor Pinson, MD, PhD 2, Ekaterina Anpilogova, MD 1
International Journal of Scientific & Engineering Research Volume 9, Issue 4, April-2018 712 Laser Treatment of Onychomycosis Olga Olisova, MD, PhD 1, Igor Pinson, MD, PhD 2, Ekaterina Anpilogova, MD 1
To order reprints or e-prints of JDD articles please contact JDD
 June 2017 534 VOLUME 16 ISSUE 6 Copyright 2017 ORIGINAL ARTICLE Journal of Drugs in Dermatology The Efficacy and Safety of Azelaic Acid 15% Foam in the Treatment of Truncal Acne Vulgaris Lauren K. Hoffman
June 2017 534 VOLUME 16 ISSUE 6 Copyright 2017 ORIGINAL ARTICLE Journal of Drugs in Dermatology The Efficacy and Safety of Azelaic Acid 15% Foam in the Treatment of Truncal Acne Vulgaris Lauren K. Hoffman
Kenneth W. Mahaffey, MD and Keith AA Fox, MB ChB
 Once-daily oral direct factor Xa inhibition Compared with vitamin K antagonism for prevention of stroke and Embolism Trial in Atrial Fibrillation Kenneth W. Mahaffey, MD and Keith AA Fox, MB ChB on behalf
Once-daily oral direct factor Xa inhibition Compared with vitamin K antagonism for prevention of stroke and Embolism Trial in Atrial Fibrillation Kenneth W. Mahaffey, MD and Keith AA Fox, MB ChB on behalf
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
In 2005, the estimated number of
 Treatment of Onychomycosis in Diabetic Patients Jason A. Winston and Jami L. Miller, MD In 2005, the estimated number of Americans with diabetes was 20.8 million people, with an additional 1.5 million
Treatment of Onychomycosis in Diabetic Patients Jason A. Winston and Jami L. Miller, MD In 2005, the estimated number of Americans with diabetes was 20.8 million people, with an additional 1.5 million
Estimates of Influenza Vaccination Coverage among Adults United States, Flu Season
 Estimates of Influenza Vaccination Coverage among Adults United States, 2017 18 Flu Season On This Page Summary Methods Results Discussion Figure 1 Figure 2 Figure 3 Figure 4 Table 1 Additional Estimates
Estimates of Influenza Vaccination Coverage among Adults United States, 2017 18 Flu Season On This Page Summary Methods Results Discussion Figure 1 Figure 2 Figure 3 Figure 4 Table 1 Additional Estimates
Epidemiology of Venous Thromboembolism in the East. Heng Joo NG Department of Haematology Singapore General Hospital Singapore
 Epidemiology of Venous Thromboembolism in the East Heng Joo NG Department of Haematology Singapore General Hospital Singapore Early Literature Tinckler LF Br Med J 1964;1:502 Propagating a notion.. Hwang
Epidemiology of Venous Thromboembolism in the East Heng Joo NG Department of Haematology Singapore General Hospital Singapore Early Literature Tinckler LF Br Med J 1964;1:502 Propagating a notion.. Hwang
AR101 peanut allergy immunotherapy for adult and paediatric patients
 AR101 peanut allergy immunotherapy for adult and paediatric patients NIHRIO (HSRIC) ID: 11815 NIHR Innovation Observatory Evidence Briefing: May 2017 NICE ID: 8773 LAY SUMMARY Food allergy occurs when
AR101 peanut allergy immunotherapy for adult and paediatric patients NIHRIO (HSRIC) ID: 11815 NIHR Innovation Observatory Evidence Briefing: May 2017 NICE ID: 8773 LAY SUMMARY Food allergy occurs when
PFIZER INC. Study Initiation Date and Completion Dates: Information not available (Date of Statistical Report: 16 May 2004)
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
THE MARKET-LEADING RESOURCE FOR THE GLOBAL BIOANALYTICAL COMMUNITY BIOANALYSIS ZONE 2018 MEDIA PACK
 THE MARKET-LEADING RESOURCE FOR THE GLOBAL BIOANALYTICAL COMMUNITY BIOANALYSIS ZONE 2018 MEDIA PACK Sarah Mayes: s.mayes@future-science.com Dionne Murray: d.murray@future-science.com +44 (0)20 8371 6090
THE MARKET-LEADING RESOURCE FOR THE GLOBAL BIOANALYTICAL COMMUNITY BIOANALYSIS ZONE 2018 MEDIA PACK Sarah Mayes: s.mayes@future-science.com Dionne Murray: d.murray@future-science.com +44 (0)20 8371 6090
Product: Denosumab (AMG 162) Abbreviated Clinical Study Report: (Extension Phase Results) Date: 24 August 2010 Page Page 2 of 2 of
 Product: Denosumab (MG 162) bbreviated Clinical Study Report: 20040114 (Extension Phase Results) Date: 24 ugust 2010 Page Page 2 of 2 of 1314 55 2. SYNOPSIS Name of Sponsor: mgen Inc. Name of Finished
Product: Denosumab (MG 162) bbreviated Clinical Study Report: 20040114 (Extension Phase Results) Date: 24 ugust 2010 Page Page 2 of 2 of 1314 55 2. SYNOPSIS Name of Sponsor: mgen Inc. Name of Finished
Efficacy of Levetiracetam: A Review of Three Pivotal Clinical Trials
 Epilepsia, 42(Suppl. 4):31 35, 2001 Blackwell Science, Inc. International League Against Epilepsy Efficacy of : A Review of Three Pivotal Clinical Trials Michael Privitera University of Cincinnati Medical
Epilepsia, 42(Suppl. 4):31 35, 2001 Blackwell Science, Inc. International League Against Epilepsy Efficacy of : A Review of Three Pivotal Clinical Trials Michael Privitera University of Cincinnati Medical
GENERAL INFORMATION. Adverse Event (AE) Definition (ICH GUIDELINES E6 FOR GCP 1.2):
 Make copies of the blank SAE report form as needed. Retain originals with confirmation of all information faxed to DMID Pharmacovigilance Group Clinical Research Operations and Management Support (CROMS
Make copies of the blank SAE report form as needed. Retain originals with confirmation of all information faxed to DMID Pharmacovigilance Group Clinical Research Operations and Management Support (CROMS
CASE STUDY ADULT CASES OF ONYCHOMYCOSIS. Warren S. Joseph, DPM, FIDSA
 ADULT CASES OF ONYCHOMYCOSIS Warren S. Joseph, DPM, FIDSA BACKGROUND (CASE 1) A 41-year-old woman presented with no complaints of pain, but she was concerned about discoloration of her toenails. She has
ADULT CASES OF ONYCHOMYCOSIS Warren S. Joseph, DPM, FIDSA BACKGROUND (CASE 1) A 41-year-old woman presented with no complaints of pain, but she was concerned about discoloration of her toenails. She has
The Post Stroke Checklist
 Singapore National Stroke Association, 2014 Stroke Club 1 The Post Stroke Checklist Christopher Chen, FRCP, FAMS Department of Pharmacology, National University of Singapore Singapore Singapore National
Singapore National Stroke Association, 2014 Stroke Club 1 The Post Stroke Checklist Christopher Chen, FRCP, FAMS Department of Pharmacology, National University of Singapore Singapore Singapore National
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Trial Synopsis TL , NCT#
 Clinical Trial Synopsis, NCT#00492011 Title of Study: A Randomized, Double-Blind, Placebo-Controlled, Parallel-Group Study to Evaluate the Ability of Ramelteon 1 mg, 4 mg, and 8 mg to Alleviate the Insomnia
Clinical Trial Synopsis, NCT#00492011 Title of Study: A Randomized, Double-Blind, Placebo-Controlled, Parallel-Group Study to Evaluate the Ability of Ramelteon 1 mg, 4 mg, and 8 mg to Alleviate the Insomnia
Clinical Trial Results Database Page 1
 Clinical Trial Results Database Page 1 Sponsor Novartis Pharmaceuticals Corporation Generic Drug Name Therapeutic Area of Trial Major Depressive Disorder (MDD) Approved Indication Treatment of major depressive
Clinical Trial Results Database Page 1 Sponsor Novartis Pharmaceuticals Corporation Generic Drug Name Therapeutic Area of Trial Major Depressive Disorder (MDD) Approved Indication Treatment of major depressive
Clinical Trial Report Synopsis
 Clinical Trial Report Synopsis Efficacy and Safety of Ingenol Mebutate Gel in Field Treatment of Actinic Keratosis on Full Face, Balding Scalp or Approximately 250 cm 2 on the Chest Design of trial: Part
Clinical Trial Report Synopsis Efficacy and Safety of Ingenol Mebutate Gel in Field Treatment of Actinic Keratosis on Full Face, Balding Scalp or Approximately 250 cm 2 on the Chest Design of trial: Part
BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE (JPC)
 BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE (JPC) April 2014 Review April 2017 Bulletin 197: Dapoxetine for Premature Ejaculation JPC Recommendations: To support the East of England Priorities Advisory
BEDFORDSHIRE AND LUTON JOINT PRESCRIBING COMMITTEE (JPC) April 2014 Review April 2017 Bulletin 197: Dapoxetine for Premature Ejaculation JPC Recommendations: To support the East of England Priorities Advisory
Clinical Study Synopsis for Public Disclosure
 Clinical Study Synopsis for Public Disclosure These results are supplied for informational purposes only in the interest of scientific disclosure. The synopsis may include approved and non-approved uses,
Clinical Study Synopsis for Public Disclosure These results are supplied for informational purposes only in the interest of scientific disclosure. The synopsis may include approved and non-approved uses,
Amruta Mohite 1, Anil Gugle 2*, Rahul Kote 3 and Vikrant Jadhav 4
 MVP Journal of Medical Sciences, Vol 2(2), 76-80, July-December 2015 A Comparative Study of Efficacy, Safety and Relapse Rate of Three Drugs; Systemic Ketoconazole, Systemic Itraconazole and Topical Oxiconazole
MVP Journal of Medical Sciences, Vol 2(2), 76-80, July-December 2015 A Comparative Study of Efficacy, Safety and Relapse Rate of Three Drugs; Systemic Ketoconazole, Systemic Itraconazole and Topical Oxiconazole
Establishment of Dose Reference Levels for Nuclear Medicine in Sudan
 Open Journal of Radiology, 2016, 6, 258-263 http://www.scirp.org/journal/ojrad ISSN Online: 2164-3032 ISSN Print: 2164-3024 Establishment of Dose Reference Levels for Nuclear Medicine in Sudan Wadah M.
Open Journal of Radiology, 2016, 6, 258-263 http://www.scirp.org/journal/ojrad ISSN Online: 2164-3032 ISSN Print: 2164-3024 Establishment of Dose Reference Levels for Nuclear Medicine in Sudan Wadah M.
XP23829 PHASE 2 PSORIASIS TRIAL PRELIMINARY TOPLINE DATA PRESENTATION SEPTEMBER 15, 2015 COPYRIGHT 2015 XENOPORT, INC. ALL RIGHTS RESERVED.
 XP23829 PHASE 2 PSORIASIS TRIAL PRELIMINARY TOPLINE DATA PRESENTATION SEPTEMBER 15, 2015 COPYRIGHT 2015 XENOPORT, INC. ALL RIGHTS RESERVED. SAFE HARBOR DISCLAIMER These slides and the accompanying oral
XP23829 PHASE 2 PSORIASIS TRIAL PRELIMINARY TOPLINE DATA PRESENTATION SEPTEMBER 15, 2015 COPYRIGHT 2015 XENOPORT, INC. ALL RIGHTS RESERVED. SAFE HARBOR DISCLAIMER These slides and the accompanying oral
Title. Establishment of a novel model of onychomycosis in the rabbit. for evaluation of antifungal agents. Running title
 AAC Accepts, published online ahead of print on May 0 Antimicrob. Agents Chemother. doi:./aac.00- Copyright 0, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights Reserved.
AAC Accepts, published online ahead of print on May 0 Antimicrob. Agents Chemother. doi:./aac.00- Copyright 0, American Society for Microbiology and/or the Listed Authors/Institutions. All Rights Reserved.
The effectiveness of telephone reminders and SMS messages on compliance with colorectal cancer screening: an open-label, randomized controlled trial
 Page1 of 5 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 The effectiveness of telephone reminders and SMS messages on compliance with colorectal cancer screening: an
Page1 of 5 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 30 The effectiveness of telephone reminders and SMS messages on compliance with colorectal cancer screening: an
OTEZLA (Apremilast) Showed Meaningful Improvements in Clinical and Quality of Life Measures of Psoriasis Beyond Those Captured by Assessing Skin Alone
 OTEZLA (Apremilast) Showed Meaningful Improvements in Clinical and Quality of Life Measures of Psoriasis Beyond Those Captured by Assessing Skin Alone Patients with moderate to severe plaque psoriasis
OTEZLA (Apremilast) Showed Meaningful Improvements in Clinical and Quality of Life Measures of Psoriasis Beyond Those Captured by Assessing Skin Alone Patients with moderate to severe plaque psoriasis
Use and Perception of Scientific Medical Reprints
 TITLE OF PRESENTATION Use and Perception of Scientific Medical Reprints Elsevier Health Panel Research Report Date August 2017 I 2 Report Structure Research Objectives, Methodology & Sample Executive Summary
TITLE OF PRESENTATION Use and Perception of Scientific Medical Reprints Elsevier Health Panel Research Report Date August 2017 I 2 Report Structure Research Objectives, Methodology & Sample Executive Summary
Moberg Pharma AB Providing Unique Products in Underserved Niches through Commercial and Innovation Excellence
 Moberg Pharma AB Providing Unique Products in Underserved Niches through Commercial and Innovation Excellence Presentation at Jefferies Global Healthcare Conference, November 20, 2014 Peter Wolpert, CEO
Moberg Pharma AB Providing Unique Products in Underserved Niches through Commercial and Innovation Excellence Presentation at Jefferies Global Healthcare Conference, November 20, 2014 Peter Wolpert, CEO
 PATCH Analysis Plan v1.2.doc Prophylactic Antibiotics for the Treatment of Cellulitis at Home: PATCH Analysis Plan for PATCH I and PATCH II Authors: Angela Crook, Andrew Nunn, James Mason and Kim Thomas,
PATCH Analysis Plan v1.2.doc Prophylactic Antibiotics for the Treatment of Cellulitis at Home: PATCH Analysis Plan for PATCH I and PATCH II Authors: Angela Crook, Andrew Nunn, James Mason and Kim Thomas,
Migraine: Developing Drugs for Acute Treatment Guidance for Industry
 Migraine: Developing Drugs for Acute Treatment Guidance for Industry U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) February 2018
Migraine: Developing Drugs for Acute Treatment Guidance for Industry U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) February 2018
Scottish Medicines Consortium
 Scottish Medicines Consortium ustekinumab, 45mg solution for injection (Stelara ) No. (572/09) Janssen-Cilag Ltd 15 January 2010 The Scottish Medicines Consortium (SMC) has completed its assessment of
Scottish Medicines Consortium ustekinumab, 45mg solution for injection (Stelara ) No. (572/09) Janssen-Cilag Ltd 15 January 2010 The Scottish Medicines Consortium (SMC) has completed its assessment of
Clinical and Mycologic Characteristics of Onychomycosis in Diabetic Patients
 Acta Dermatovenerologica Croatica 2010;18(2):84-91 CLINICAL ARTICLE Clinical and Mycologic Characteristics of Onychomycosis in Diabetic Patients Nawaf Al-Mutairi, Bayoumy Ibrahim Eassa, Dhuha Abdullah
Acta Dermatovenerologica Croatica 2010;18(2):84-91 CLINICAL ARTICLE Clinical and Mycologic Characteristics of Onychomycosis in Diabetic Patients Nawaf Al-Mutairi, Bayoumy Ibrahim Eassa, Dhuha Abdullah
Supplementary Online Content
 Supplementary Online Content Foley P, Gordon K, Griffiths, CEM, et al. Efficacy of guselkumab compared with adalimumab and placebo for psoriasis in specific body regions: a secondary analysis of 2 randomized
Supplementary Online Content Foley P, Gordon K, Griffiths, CEM, et al. Efficacy of guselkumab compared with adalimumab and placebo for psoriasis in specific body regions: a secondary analysis of 2 randomized
Study No. 178-CL-008 Report Final Version, 14 Dec 2006 Reissued Version, 18 Jul 2011 Astellas Pharma Europe B.V. Page 13 of 122
 Page 13 of 122 3 SYNOPSIS Title of study: (International) Study No: A randomized, double-blind, parallel group, proof-of-concept study of in comparison with placebo and tolterodine in patients with symptomatic
Page 13 of 122 3 SYNOPSIS Title of study: (International) Study No: A randomized, double-blind, parallel group, proof-of-concept study of in comparison with placebo and tolterodine in patients with symptomatic
Scottish Medicines Consortium
 Scottish Medicines Consortium levetiracetam, 250, 500, 750 and 1000mg tablets and levetiracetam oral solution 100mg/ml (Keppra ) No. (394/07) UCB Pharma Limited 10 August 2007 The Scottish Medicines Consortium
Scottish Medicines Consortium levetiracetam, 250, 500, 750 and 1000mg tablets and levetiracetam oral solution 100mg/ml (Keppra ) No. (394/07) UCB Pharma Limited 10 August 2007 The Scottish Medicines Consortium
Clinical Trial Report Synopsis
 This document has been do\vnloaded from \v ww.leo-pharma.com subject to the terms of use state on the website. It contains data and results regarding approved and non-approved uses, formulations or treatment
This document has been do\vnloaded from \v ww.leo-pharma.com subject to the terms of use state on the website. It contains data and results regarding approved and non-approved uses, formulations or treatment
16.9 hours. Not reported 19.9 hours. Metabolism: Not known Elimination: Not known. Efficacy:
 Brand Name: Luzu Generic Name: luliconazole Manufacturer 1 : Medicis Pharmaceutical Corporation Drug Class 1,2,3,4 : Imidazole antifungal, topical Uses Labeled Uses 1,2,3,4 : Topical treatment of interdigital
Brand Name: Luzu Generic Name: luliconazole Manufacturer 1 : Medicis Pharmaceutical Corporation Drug Class 1,2,3,4 : Imidazole antifungal, topical Uses Labeled Uses 1,2,3,4 : Topical treatment of interdigital
Public Assessment Report Scientific discussion. Ovixan (mometasone furoate) SE/H/1088/01/DC
 Public Assessment Report Scientific discussion Ovixan (mometasone furoate) SE/H/1088/01/DC This module reflects the scientific discussion for the approval of Ovixan. The procedure was finalised at 2012-12-14.
Public Assessment Report Scientific discussion Ovixan (mometasone furoate) SE/H/1088/01/DC This module reflects the scientific discussion for the approval of Ovixan. The procedure was finalised at 2012-12-14.
Wellness Coaching for People with Prediabetes
 Wellness Coaching for People with Prediabetes PUBLIC HEALTH RESEARCH, PRACTICE, AND POLICY Volume 12, E207 NOVEMBER 2015 ORIGINAL RESEARCH Wellness Coaching for People With Prediabetes: A Randomized Encouragement
Wellness Coaching for People with Prediabetes PUBLIC HEALTH RESEARCH, PRACTICE, AND POLICY Volume 12, E207 NOVEMBER 2015 ORIGINAL RESEARCH Wellness Coaching for People With Prediabetes: A Randomized Encouragement
PFIZER INC. Study Initiation Date and Completion Dates: 09 March 2000 to 09 August 2001.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
The Potential Psychological Impact of Skin Conditions
 DOI 10.1007/s13555-016-0169-7 REVIEW The Potential Psychological Impact of Skin Conditions Ari Tuckman Received: August 11, 2016 The Author(s) 2017. This article is published with open access at Springerlink.com
DOI 10.1007/s13555-016-0169-7 REVIEW The Potential Psychological Impact of Skin Conditions Ari Tuckman Received: August 11, 2016 The Author(s) 2017. This article is published with open access at Springerlink.com
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Screening for ovarian cancer Kehoe, Sean
 Screening for ovarian cancer Kehoe, Sean DOI: 10.1016/j.maturitas.2015.05.009 License: Creative Commons: Attribution-NonCommercial-NoDerivs (CC BY-NC-ND) Document Version Peer reviewed version Citation
Screening for ovarian cancer Kehoe, Sean DOI: 10.1016/j.maturitas.2015.05.009 License: Creative Commons: Attribution-NonCommercial-NoDerivs (CC BY-NC-ND) Document Version Peer reviewed version Citation
