VACCINE ACTIVE SURVEILLANCE I
|
|
|
- Bernadette Stokes
- 5 years ago
- Views:
Transcription
1 VACCINE ACTIVE SURVEILLANCE I Caitlin N Dodd, MS Biostatistician, Global Child Health Cincinnati Children s Hospital Medical Center
2 CONTENTS M1 Compare epidemiological study methods Discuss the benefits of self controlled case series (SCCS) study methodology Discuss the application of SCCS
3 Slide 2 M1 These are really placeholders. Please change the Contents to suit your purpose. We will recommend that you use max of 30slides for this 60mins talk. MSH, 03/23/2012
4 SECTION ONE: COMPARISON OF STUDY METHODS Outline: Case Control Studies Cohort Studies
5 CASE CONTROL STUDIES Subjects who have experienced an event of interest or have been diagnosed with the condition of interest are matched to non diagnosed controls Popular because they are relatively inexpensive and can be completed quickly Prohibitive in that controls should ideally be matched across multiple characteristics
6 CASE CONTROL STUDIES Latent confounders cannot be controlled for by matching Case control studies can demonstrate association but do not demonstrate causation Vaccinated Unvaccinated Adverse a b event No AE c d Total a+c b+d
7 CASE CONTROL STUDIES Advantages Permit the study of rare diseases. Permit the study of diseases with long latency between exposure and manifestation. Can be launched and conducted over relatively short time periods. Relatively inexpensive as compared to cohort studies. Can study multiple potential causes of disease.
8 CASE CONTROL STUDIES Disadvantages Information on exposure and past history is primarily based on interview and may be subject to recall bias. Validation of information on exposure is difficult, or incomplete, or even impossible. By definition, concerned with one disease only. Cannot usually provide information on incidence rates of disease. Generally incomplete control of extraneous variables. Choice of appropriate control group may be difficult. Methodology may be hard to comprehend for non epidemiologists and correct interpretation of results may be difficult.
9 CASE CONTROL STUDIES VS. SCCS Self controlled case series can be thought of as a case control study in which subjects act as their own controls. Because of the underlying time scale (whether age or calendar time based), the self controlled case series is a more flexible methodology. Relative incidence can be estimated by season, exposure, or subject age
10 CASE CONTROL STUDIES VS. SCCS Exposed populations may be self selecting which may lead to inherent bias in a case control study The self controlled case series method is more computationally intensive and less intensive in terms of data collection.
11 CASE CONTROL STUDIES VS. SCCS Case control study provides relative risk = (a/a+c) / (b/b+d) SCCS provides an estimate of relative incidence associated with exposure using only a. Relative incidence associated with time varying covariates (season, age, etc.) are estimated using a and b.
12 COHORT STUDIES A cohort study follows a group of healthy individuals over time and then compares those subjects who experience the event of interest with those who do not experience the event of interest Analyzed by calculating incidence rate ratios for exposed and non exposed subjects, then using these to determine relative risk.
13 COHORT STUDIES Advantages: Allow complete information on the subject s exposure, including quality control of data, and experience thereafter. Provide a clear temporal sequence of exposure and disease. Give an opportunity to study multiple outcomes related to a specific exposure. Permit calculation of incidence rates (absolute risk) as well as relative risk. Methodology and results are easily understood by nonepidemiologists. Enable the study of relatively rare exposures.
14 COHORT STUDIES Disadvantages: Not suited for the study of extremely rare diseases because a large number of subjects is required. Not suitable when the time between exposure and disease manifestation is very long, although this can be overcome in historical cohort studies. Exposure patterns, for example the composition of oral contraceptives, may change during the course of the study and make the results irrelevant. Maintaining high rates of follow up can be difficult. Expensive to carry out because a large number of subjects is usually required. Baseline data may be sparse because the large number of subjects does not allow for long interviews.
15 COHORT STUDIES VS. SCCS In a SCCS study, exposure times are considered to be fixed and event times are random. The opposite is the case in a cohort study. Cohort studies estimate relative risk while SCCS studies estimate relative incidence.
16 COHORT STUDIES VS. SCCS Both allow for age and time effects Efficiency is lower in the SCCS because, by conditioning on the number of events, marginal information is lost for each individual Self controlled case series studies require far less time in patient follow up than cohort studies.
17 ADVANTAGES OF THE SELF-CONTROLLED CASE SERIES METHOD Based on cases only Provides consistent estimates of relative incidence Controls for fixed confounders Age and temporal variation in baseline can be allowed for Can have high efficiency relative to the retrospective cohort method.
18 LIMITATIONS OF THE SELF-CONTROLLED CASE SERIES METHOD It requires that the probability of exposure is not affected by the occurrence of an outcome event. Risk must be small for non recurrent events Only produces estimates of relative (not absolute) incidence Requires variability in the time or age of the event
19 APPLICATIONS Has typically been applied to vaccine safety questions Also: SSRIs and falls Long haul flights and Thromboembolism Beta blockers and heart failure
20 SECTION TWO: EXAMPLE OF STUDY USING SCCS Does oral polio vaccine cause intussusception in infants? Evidence from a sequence of three self controlled cases series studies in The United Kingdom Nick Andrews, et al.
21 NICK ANDREWS, ORAL POLIO VACCINE AND INTUSSUSCEPTION, 2001 Three analyses on 3 sets of linked hospital admissions data Analysis 1: exploratory, risk periods defined as days 0 13 after each of three doses Analysis 2: exploratory, risk periods days after each dose Analysis 3: formal hypothesis testing of increased risk in days following each dose
22 NICK ANDREWS, ORAL POLIO VACCINE AND INTUSSUSCEPTION, 2001 Methods Collected database data on all children age 28 to 365 days with an ICD code of intussusception For each child, date of birth, sex, postal code, hospital, hospital number, immunizations, dates of immunizations, and age in days at time of admission were collected. Validation of diagnosis was conducted on a subset of subjects
23 NICK ANDREWS, ORAL POLIO VACCINE AND INTUSSUSCEPTION, 2001 Statistical Analysis Variables used in the analysis: DOB, dates of vaccinations, dates of intussusceptions, region, and sex. Subjects with less than 3 recorded doses were excluded as were subjects with less than 21 days between doses Analyzed using the self controlled case series method, stratified by age for three data sets (HE1: January 1991 March 1997, HE2: April 1997 June1999, and GRPD: from the General Practice Research Database).
24 NICK ANDREWS, ORAL POLIO VACCINE AND INTUSSUSCEPTION, 2001 Descriptive statistics on 3 data sets
25 NICK ANDREWS, ORAL POLIO VACCINE AND INTUSSUSCEPTION, 2001 Results No evidence of increased relative incidence in the 0 13 days following vaccination Increased risk of intussusception in the days following the third dose of OPV (RI = 2.15, p = ) No increased risk when 14 days pre vaccination and periods after dose 1 and 2 included in the background risk in HE2 and GRPD data (RI = 1.03, 95% CI = )
26 NICK ANDREWS, ORAL POLIO VACCINE AND INTUSSUSCEPTION, 2001 Results Total cases in the risk periods with relative incidence (95% CI) by dose for the three data sets
27 NICK ANDREWS, ORAL POLIO VACCINE AND INTUSSUSCEPTION, 2001 Results Total cases in the risk periods with relative incidence (95% CI) for dose 3 with a pre vaccination low period:
28 NICK ANDREWS, ORAL POLIO VACCINE AND INTUSSUSCEPTION, 2001 Interpretation and Conclusions
29 SECTION THREE: APPLYING THE SCCS Outline: Events and exposures appropriate to the selfcontrolled case series Determining potential exposure, seasonal, and ageassociated risk windows Power and sample size
30 EVENTS AND EXPOSURES PAST STUDIES MMR vaccine and autism MMR vaccine and meningitis Influenza vaccine and asthma MMR vaccine and Idiopathic thrombocytopenic purpura OPV and intussusception Rotavirus vaccine and intussusception Infant vaccines and lower respiratory disease MMR vaccine and bacterial infections SRI antidepressants and hip fracture Air travel and venous thromboembolism Influenza vaccine and Bell s Palsy MMR vaccine and gait disturbance SRI antidepressants and myocardial infarction Nicotine replacement therapy and myocardial infarction, stroke, or death Bupropian and sudden death Acute infection and pulmonary embolism Dialysis for diabetes treatment and foot ulceration Influenza vaccine and Guillain Barre Syndrome MMR vaccination and convulsions Hepatitis B vaccine and demyelinating events Oral Bisphosphonates and Atrial fibrillation Thiazolidinediones and Fractures Prescription Medications and Motor vehicle crashes
31 EVENTS AND EXPOSURES Questions to consider What is a recurrence and what is a new event? How many doses are given? Is this a transient or chronic exposure? What determines the date of exposure? Of an event? Can cases be reliably verified? Is exposure history available? In what form? What is the risk period following exposure?
32 EVENTS AND EXPOSURES Risk periods: Chosen based on prior hypotheses, previous studies, and presumed biological mechanisms. If it is too long, too short or is placed so that it does not cover the true risk period then the relative incidence estimate may be biased toward the null
33 EXPOSURE, SEASON, AND AGE ASSOCIATED RISK Questions to consider Are exposure, event, or both seasonal? Does data exist to answer these questions? Do subjects represent an age group or specific range of ages? Is age data available? How many exposures? Will all subjects have the same number of exposures? Is risk after each exposure expected to be the same?
34 POWER AND SAMPLE SIZE An Example:
35 MODELING WITH THE CASE SERIES METHOD Example using the global GBS/H1N1 study Assessing risk of Guillain Barre Syndrome following vaccination with pandemic influenza vaccine Includes data from 15 countries Based upon increased risk seen in the swine flu pandemic
36 MODELING WITH THE CASE SERIES METHOD Multiple risk periods Multiple risk periods can be defined and the relative incidence in these periods can be estimated using the case series method For the Global GBS/H1N1 project, these periods are days 1 6, 7 21, and following vaccination Each of these periods is compared to the control period, meaning days during observation before exposure and days during observation following day 42 post vaccine.
37 MODELING WITH THE CASE SERIES METHOD Multiple events Requires a within individual independence assumption Total number of events within each time period are analyzed For the GBS study, multiple events are not analyzed. Additional GBS diagnoses within 6 months of the initial diagnosis are considered a relapse and only the first diagnosis is analyzed.
38 MODELING WITH THE CASE SERIES METHOD Unique and non independent events Poisson assumption fails when events are unique (as in the case of GBS). When the baseline rate for the event approaches 0, the case series method is valid Background rate of GBS is 10 to 20 cases per million population Poisson assumption fails when occurrence of one event increases the probability of subsequent events (recurrent GBS events) If the initiating event is rare, only the first event should be used Did not see recurrent GBS in the global data set
39 MODELING WITH THE CASE SERIES METHOD Event dependent exposures Most restrictive assumption of the case series method is that the occurrence of the event does not alter subsequent exposures Violated in the case of GBS and influenza vaccine Three approaches: Redefine the observation period to begin at the time of exposure and only include exposed individuals. Removing a pre exposure time period from the baseline The pseudo likelihood method
40 MODELING WITH THE CASE SERIES METHOD Covariates and interactions Although fixed covariates are controlled, covariates may be effect modifiers. GBS is more common in males and in older subjects Interactions with sex and age group will be included in the analysis of global data A likelihood ratio test can be conducted to determine if the interaction should be included in the model.
41 MODELING WITH THE CASE SERIES METHOD Multiple exposures (of the same vaccine) Multiple exposures (such as doses of childhood vaccines) can be modeled, each with a risk period following exposure. The estimates are relatively unchanged if the time between exposures is longer than the risk period for each exposure. This is allowed for in the analysis of GBS and H1N1 vaccine, although few subjects received 2 doses of pandemic vaccine. Risk windows do not overlap
42 MODELING WITH THE CASE SERIES METHOD Multiple exposures (of different vaccines) Multiple exposures (such as a seasonal and pandemic vaccine) can be modeled, each with a risk period following exposure. Risk windows in this situation may overlap In the analysis of GBS and H1N1, we plan to assess relative incidence associated with seasonal and pandemic vaccines Challenge in that these were not always received in the same order Necessary to write new code to accommodate this situation
43 MODELING WITH THE CASE SERIES METHOD Confounding between exposure and age Unexposed cases help to disentangle the age effect. Not applicable to the GBS study because GBS is not hypothesized to be age dependent on the weekly or monthly scale. Long and indefinite risk periods Confounding between age and exposure effects is more likely with long risk periods. Not applicable to the GBS study risk period is hypothesized to be 6 weeks at the most.
44 MODELING WITH THE CASE SERIES METHOD Temporal effects Using calendar time as the underlying time line Appropriate for influenza vaccination studies due to the seasonality of influenza and influenza vaccination Determining global seasonality cut points has been one of the main challenges of the global GBS/H1N1 study
45 CONCLUSION The self controlled case series method is a powerful tool for vaccine safety studies Vaccine safety researchers continue to improve and modify the method The global study has been completed. Next phase to include developing countries
Methodologies for vaccine safety surveillance. Nick Andrews, Statistics Unit Public Health England October 2015
 Methodologies for vaccine safety surveillance Nick Andrews, Statistics Unit Public Health England October 2015 Some dramas of the past 20 years which do you think turned out to be real adverse reactions?
Methodologies for vaccine safety surveillance Nick Andrews, Statistics Unit Public Health England October 2015 Some dramas of the past 20 years which do you think turned out to be real adverse reactions?
Population-based post-licensure safety surveillance
 Population-based post-licensure safety surveillance Nick Andrews, Public Health England With thanks to Paddy Farrington, Open University for contributing some SCCS slides ADVAC May 27 th 2016 Aims of the
Population-based post-licensure safety surveillance Nick Andrews, Public Health England With thanks to Paddy Farrington, Open University for contributing some SCCS slides ADVAC May 27 th 2016 Aims of the
INTRODUCTION TO EPIDEMIOLOGICAL STUDY DESIGNS PHUNLERD PIYARAJ, MD., MHS., PHD.
 INTRODUCTION TO EPIDEMIOLOGICAL STUDY DESIGNS PHUNLERD PIYARAJ, MD., MHS., PHD. 1 OBJECTIVES By the end of this section, you will be able to: Provide a definition of epidemiology Describe the major types
INTRODUCTION TO EPIDEMIOLOGICAL STUDY DESIGNS PHUNLERD PIYARAJ, MD., MHS., PHD. 1 OBJECTIVES By the end of this section, you will be able to: Provide a definition of epidemiology Describe the major types
Self-controlled case-series method
 Self-controlled case-series method Presented by StellaMay Gwini Biostatistical Consultancy Platform, DEPM Objectives To describe the self-controlled case-series method Highlight possible uses of SCCS within
Self-controlled case-series method Presented by StellaMay Gwini Biostatistical Consultancy Platform, DEPM Objectives To describe the self-controlled case-series method Highlight possible uses of SCCS within
Confounding in influenza VE studies in seniors, and possible solutions
 Confounding in influenza VE studies in seniors, and possible solutions Michael L. Jackson Group Health Research Institute 4 th December, 2012 1 Outline Focus is on non-specific outcomes (e.g. community-acquired
Confounding in influenza VE studies in seniors, and possible solutions Michael L. Jackson Group Health Research Institute 4 th December, 2012 1 Outline Focus is on non-specific outcomes (e.g. community-acquired
Assessing Vaccine Safety Post Licensure. Neal A. Halsey Johns Hopkins University
 Assessing Vaccine Safety Post Licensure Neal A. Halsey Johns Hopkins University Inactivated Respiratory Syncytial Virus Vaccine: 1960 s Formalin inactivated Administered to infants Minimal reactions Induced
Assessing Vaccine Safety Post Licensure Neal A. Halsey Johns Hopkins University Inactivated Respiratory Syncytial Virus Vaccine: 1960 s Formalin inactivated Administered to infants Minimal reactions Induced
Assessing the Safety of Vaccines at the FDA: Pre- and Post-Licensure Evaluation
 Assessing the Safety of Vaccines at the FDA: Pre- and Post-Licensure Evaluation Karen Farizo, M.D. Office of Vaccines Research and Review Center for Biologics Evaluation and Research US Food and Drug Administration
Assessing the Safety of Vaccines at the FDA: Pre- and Post-Licensure Evaluation Karen Farizo, M.D. Office of Vaccines Research and Review Center for Biologics Evaluation and Research US Food and Drug Administration
Answer keys for Assignment 4: Measures of disease frequency
 Answer keys for Assignment 4: Measures of disease frequency (The correct answer is underlined in bold text) 1. This statistic is used to estimate the risk of acquiring a disease. It may be measured as
Answer keys for Assignment 4: Measures of disease frequency (The correct answer is underlined in bold text) 1. This statistic is used to estimate the risk of acquiring a disease. It may be measured as
Self controlled case series methods: an alternative to standard epidemiological study designs
 open access Self controlled case series methods: an alternative to standard epidemiological study designs Irene Petersen,, Ian Douglas, Heather Whitaker Department of Primary Care and Population Health,
open access Self controlled case series methods: an alternative to standard epidemiological study designs Irene Petersen,, Ian Douglas, Heather Whitaker Department of Primary Care and Population Health,
Epidemiology: Overview of Key Concepts and Study Design. Polly Marchbanks
 Epidemiology: Overview of Key Concepts and Study Design Polly Marchbanks Lecture Outline (1) Key epidemiologic concepts - Definition - What epi is not - What epi is - Process of epi research Lecture Outline
Epidemiology: Overview of Key Concepts and Study Design Polly Marchbanks Lecture Outline (1) Key epidemiologic concepts - Definition - What epi is not - What epi is - Process of epi research Lecture Outline
Studies of Guillain-Barré syndrome (GBS) after influenza vaccination
 Studies of Guillain-Barré syndrome (GBS) after influenza vaccination Institute of Medicine Immunization Safety Review Committee March 13, 2003 Robert T. Chen MD MA Immunization Safety Branch National Immunization
Studies of Guillain-Barré syndrome (GBS) after influenza vaccination Institute of Medicine Immunization Safety Review Committee March 13, 2003 Robert T. Chen MD MA Immunization Safety Branch National Immunization
Incidence of GBS and CIDP following influenza vaccination
 Incidence of GBS and CIDP following influenza vaccination 1 National Influenza Immunization Program (NIIP) Analysis of the national surveillance data on GBS cases between October 1, 1976 and January 31,
Incidence of GBS and CIDP following influenza vaccination 1 National Influenza Immunization Program (NIIP) Analysis of the national surveillance data on GBS cases between October 1, 1976 and January 31,
Bias and confounding special issues. Outline for evaluation of bias
 EPIDEMIOLOGI BIAS special issues and discussion of paper April 2009 Søren Friis Institut for Epidemiologisk Kræftforskning Kræftens Bekæmpelse AGENDA Bias and confounding special issues Confounding by
EPIDEMIOLOGI BIAS special issues and discussion of paper April 2009 Søren Friis Institut for Epidemiologisk Kræftforskning Kræftens Bekæmpelse AGENDA Bias and confounding special issues Confounding by
Overview of Anticipated Procedures for Active Surveillance of New Medical Products
 info@mini-sentinel.org 1 Overview of Anticipated Procedures for Active Surveillance of New Medical Products Elizabeth Chrischilles, PhD Mini-Sentinel Protocol Core January 31, 2013 info@mini-sentinel.org
info@mini-sentinel.org 1 Overview of Anticipated Procedures for Active Surveillance of New Medical Products Elizabeth Chrischilles, PhD Mini-Sentinel Protocol Core January 31, 2013 info@mini-sentinel.org
Tuning Epidemiological Study Design Methods for Exploratory Data Analysis in Real World Data
 Tuning Epidemiological Study Design Methods for Exploratory Data Analysis in Real World Data Andrew Bate Senior Director, Epidemiology Group Lead, Analytics 15th Annual Meeting of the International Society
Tuning Epidemiological Study Design Methods for Exploratory Data Analysis in Real World Data Andrew Bate Senior Director, Epidemiology Group Lead, Analytics 15th Annual Meeting of the International Society
3. Factors such as race, age, sex, and a person s physiological state are all considered determinants of disease. a. True
 / False 1. Epidemiology is the basic science of public health. LEARNING OBJECTIVES: CNIA.BOYL.17.2.1 - Define epidemiology. 2. Within the field of epidemiology, the term distribution refers to the relationship
/ False 1. Epidemiology is the basic science of public health. LEARNING OBJECTIVES: CNIA.BOYL.17.2.1 - Define epidemiology. 2. Within the field of epidemiology, the term distribution refers to the relationship
Using the Self-Controlled Risk Interval (SCRI) Method to Study Vaccine Safety
 info@sentinelsystem.org 1 Using the Self-Controlled Risk Interval (SCRI) Method to Study Vaccine Safety W. Katherine Yih, PhD, MPH Sentinel/PRISM Epidemiology of Vaccine Safety ICPE pre-conference course,
info@sentinelsystem.org 1 Using the Self-Controlled Risk Interval (SCRI) Method to Study Vaccine Safety W. Katherine Yih, PhD, MPH Sentinel/PRISM Epidemiology of Vaccine Safety ICPE pre-conference course,
Confounding Bias: Stratification
 OUTLINE: Confounding- cont. Generalizability Reproducibility Effect modification Confounding Bias: Stratification Example 1: Association between place of residence & Chronic bronchitis Residence Chronic
OUTLINE: Confounding- cont. Generalizability Reproducibility Effect modification Confounding Bias: Stratification Example 1: Association between place of residence & Chronic bronchitis Residence Chronic
1 Case Control Studies
 Statistics April 11, 2013 Debdeep Pati 1 Case Control Studies 1. Case-control (retrospective) study: Select cases, Select controls, Compare cases and controls 2. Cutaneous melanoma (Autier, 1996). 420
Statistics April 11, 2013 Debdeep Pati 1 Case Control Studies 1. Case-control (retrospective) study: Select cases, Select controls, Compare cases and controls 2. Cutaneous melanoma (Autier, 1996). 420
OBSERVATIONAL MEDICAL OUTCOMES PARTNERSHIP
 OBSERVATIONAL Patient-centered observational analytics: New directions toward studying the effects of medical products Patrick Ryan on behalf of OMOP Research Team May 22, 2012 Observational Medical Outcomes
OBSERVATIONAL Patient-centered observational analytics: New directions toward studying the effects of medical products Patrick Ryan on behalf of OMOP Research Team May 22, 2012 Observational Medical Outcomes
Influenza vaccine effectiveness assessment in the UK. Nick Andrews, Statistics Unit, Health Protection Agency
 Influenza vaccine effectiveness assessment in the UK Nick Andrews, Statistics Unit, Health Protection Agency 1 Outline Introduction The UK swabbing schemes Assessment by the test negative case control
Influenza vaccine effectiveness assessment in the UK Nick Andrews, Statistics Unit, Health Protection Agency 1 Outline Introduction The UK swabbing schemes Assessment by the test negative case control
Incorporating Clinical Information into the Label
 ECC Population Health Group LLC expertise-driven consulting in global maternal-child health & pharmacoepidemiology Incorporating Clinical Information into the Label Labels without Categories: A Workshop
ECC Population Health Group LLC expertise-driven consulting in global maternal-child health & pharmacoepidemiology Incorporating Clinical Information into the Label Labels without Categories: A Workshop
Case-Control Studies
 Case-Control Studies Marc Schenker M.D., M.P.H Dept. of Public Health Sciences UC Davis Marc Schenker M.D., M.P.H, UC Davis 1 Case-Control Studies OBJECTIVES After this session, you will be familiar with:
Case-Control Studies Marc Schenker M.D., M.P.H Dept. of Public Health Sciences UC Davis Marc Schenker M.D., M.P.H, UC Davis 1 Case-Control Studies OBJECTIVES After this session, you will be familiar with:
Epidemiologic Methods and Counting Infections: The Basics of Surveillance
 Epidemiologic Methods and Counting Infections: The Basics of Surveillance Ebbing Lautenbach, MD, MPH, MSCE University of Pennsylvania School of Medicine Nothing to disclose PENN Outline Definitions / Historical
Epidemiologic Methods and Counting Infections: The Basics of Surveillance Ebbing Lautenbach, MD, MPH, MSCE University of Pennsylvania School of Medicine Nothing to disclose PENN Outline Definitions / Historical
Influenza vaccine and GBS
 Influenza vaccine and GBS Tamar Lasky, PhD DHHS/NIH/NICHD Presented to the Institute of Medicine Immunization Safety Review Committee March 13, 2003 Guillain-Barré Syndrome and the 1992-93 and 1993-94
Influenza vaccine and GBS Tamar Lasky, PhD DHHS/NIH/NICHD Presented to the Institute of Medicine Immunization Safety Review Committee March 13, 2003 Guillain-Barré Syndrome and the 1992-93 and 1993-94
Using negative control outcomes to identify biased study design: A self-controlled case series example. James Weaver 1,2.
 Using negative control outcomes to identify biased study design: A self-controlled case series example James Weaver 1,2 1Janssen Research & Development, LLC, Raritan, NJ, USA 2 Observational Health Data
Using negative control outcomes to identify biased study design: A self-controlled case series example James Weaver 1,2 1Janssen Research & Development, LLC, Raritan, NJ, USA 2 Observational Health Data
Trial Designs. Professor Peter Cameron
 Trial Designs Professor Peter Cameron OVERVIEW Review of Observational methods Principles of experimental design applied to observational studies Population Selection Looking for bias Inference Analysis
Trial Designs Professor Peter Cameron OVERVIEW Review of Observational methods Principles of experimental design applied to observational studies Population Selection Looking for bias Inference Analysis
Interim WHO guidance for the surveillance of human infection with swine influenza A(H1N1) virus
 Interim WHO guidance for the surveillance of human infection with swine influenza A(H1N1) virus 29 April 2009 Introduction The audiences for this guidance document are the National Focal Points for the
Interim WHO guidance for the surveillance of human infection with swine influenza A(H1N1) virus 29 April 2009 Introduction The audiences for this guidance document are the National Focal Points for the
Role of vaccination and the evaluation of LSD control programmes
 Role of vaccination and the evaluation of LSD control programmes Dr Nick Lyons FAO Workshop: LSD prevention and control Tbilisi, Georgia, 11th November 2015 Vaccines and protection Susceptible Infected
Role of vaccination and the evaluation of LSD control programmes Dr Nick Lyons FAO Workshop: LSD prevention and control Tbilisi, Georgia, 11th November 2015 Vaccines and protection Susceptible Infected
Following the health of half a million participants
 Following the health of half a million participants Cathie Sudlow UK Biobank Scientific Conference London, June 2018 Follow-up of participants in very large prospective cohorts Aim: identify a wide range
Following the health of half a million participants Cathie Sudlow UK Biobank Scientific Conference London, June 2018 Follow-up of participants in very large prospective cohorts Aim: identify a wide range
H1N1 Vaccine Based on CDCs ACIP Meeting, July 29, 2009
 August 6, 2009 H1N1 Vaccine Based on CDCs ACIP Meeting, July 29, 2009 CDC s Advisory Committee on Immunization Practices (ACIP), a panel made up of medical and public health experts, met July 29, 2009,
August 6, 2009 H1N1 Vaccine Based on CDCs ACIP Meeting, July 29, 2009 CDC s Advisory Committee on Immunization Practices (ACIP), a panel made up of medical and public health experts, met July 29, 2009,
Influenza Vaccine Safety Monitoring Update
 Influenza Vaccine Safety Monitoring Update Advisory Committee on Immunization Practices October 28, 2010 Tom Shimabukuro, MD, MPH, MBA Immunization Safety Office Division of Healthcare Quality Promotion,
Influenza Vaccine Safety Monitoring Update Advisory Committee on Immunization Practices October 28, 2010 Tom Shimabukuro, MD, MPH, MBA Immunization Safety Office Division of Healthcare Quality Promotion,
California Department of Public Health
 State of California Health and Human Services Agency California Department of Public Health MARK B HORTON, MD, MSPH Director ARNOLD SCHWARZENEGGER Governor 2009 H1N1 and Seasonal Influenza Guidance for
State of California Health and Human Services Agency California Department of Public Health MARK B HORTON, MD, MSPH Director ARNOLD SCHWARZENEGGER Governor 2009 H1N1 and Seasonal Influenza Guidance for
DECLARATION OF CONFLICT OF INTEREST
 DECLARATION OF CONFLICT OF INTEREST Warfarin and the risk of major bleeding events in patients with atrial fibrillation: a population-based study Laurent Azoulay PhD 1,2, Sophie Dell Aniello MSc 1, Teresa
DECLARATION OF CONFLICT OF INTEREST Warfarin and the risk of major bleeding events in patients with atrial fibrillation: a population-based study Laurent Azoulay PhD 1,2, Sophie Dell Aniello MSc 1, Teresa
Lessons Learned from Adverse Events and Assessment of Causal Relationships. Neal A. Halsey Johns Hopkins University
 Lessons Learned from Adverse Events and Assessment of Causal Relationships Neal A. Halsey Johns Hopkins University Lessons Learned 1000s of lessons learned Many vaccines not successful Some caused harm
Lessons Learned from Adverse Events and Assessment of Causal Relationships Neal A. Halsey Johns Hopkins University Lessons Learned 1000s of lessons learned Many vaccines not successful Some caused harm
Vaccine reaction history
 Questionnaire to consider partial or complete medical exemption from vaccination Vaccine reaction history This form should be copied and completed for each vaccine reaction, whether it is for the patient
Questionnaire to consider partial or complete medical exemption from vaccination Vaccine reaction history This form should be copied and completed for each vaccine reaction, whether it is for the patient
Swine Flu. Media Briefing. 13 January 2011
 Swine Flu Media Briefing 13 January 2011 Overview Systems for monitoring swine flu Pattern of flu in Northern Ireland Updated swine flu deaths Vaccination programme Systems for Monitoring Flu Special arrangements
Swine Flu Media Briefing 13 January 2011 Overview Systems for monitoring swine flu Pattern of flu in Northern Ireland Updated swine flu deaths Vaccination programme Systems for Monitoring Flu Special arrangements
Basic Epidemiology: Study Types. Learning Objectives. The Five Ws of Epidemiology. At the end of this presentation, participants will be able to:
 Basic Epidemiology: Study Types John Kobayashi, MD, MPH August 12, 2009 Learning Objectives At the end of this presentation, participants will be able to: Define some key concepts in epidemiology Describe
Basic Epidemiology: Study Types John Kobayashi, MD, MPH August 12, 2009 Learning Objectives At the end of this presentation, participants will be able to: Define some key concepts in epidemiology Describe
Study Designs in Epidemiology
 Study Designs in Epidemiology Dr. Sireen Alkhaldi, BDS, MPH, DrPH First semester 2017/ 2018 Department of Family and Community Medicine School of Medicine/ The University of Jordan Epidemiologic Study
Study Designs in Epidemiology Dr. Sireen Alkhaldi, BDS, MPH, DrPH First semester 2017/ 2018 Department of Family and Community Medicine School of Medicine/ The University of Jordan Epidemiologic Study
Study design. Chapter 64. Research Methodology S.B. MARTINS, A. ZIN, W. ZIN
 Chapter 64 Study design S.B. MARTINS, A. ZIN, W. ZIN Research Methodology Scientific methodology comprises a set of rules and procedures to investigate the phenomena of interest. These rules and procedures
Chapter 64 Study design S.B. MARTINS, A. ZIN, W. ZIN Research Methodology Scientific methodology comprises a set of rules and procedures to investigate the phenomena of interest. These rules and procedures
Should i get the H1N1 Vaccine?
 Should i get the H1N1 Vaccine? 2 comments and 10 questions.. 12 slides Dr. Mike Evans Associate Professor, Family & Community Medicine, University of Toronto, Staff Physician, St. Michael s Hospital Director,
Should i get the H1N1 Vaccine? 2 comments and 10 questions.. 12 slides Dr. Mike Evans Associate Professor, Family & Community Medicine, University of Toronto, Staff Physician, St. Michael s Hospital Director,
RESEARCH. Unintended effects of statins in men and women in England and Wales: population based cohort study using the QResearch database
 Unintended effects of statins in men and women in England and Wales: population based cohort study using the QResearch database Julia Hippisley-Cox, professor of clinical epidemiology and general practice,
Unintended effects of statins in men and women in England and Wales: population based cohort study using the QResearch database Julia Hippisley-Cox, professor of clinical epidemiology and general practice,
Measure of Association Examples of measure of association
 Measure of Association Examples of measure of association Epidemiologists usually use relative differences to assess causal association (Table 3-1). Table 3 1 Types of Measures of Association Used in Analytic
Measure of Association Examples of measure of association Epidemiologists usually use relative differences to assess causal association (Table 3-1). Table 3 1 Types of Measures of Association Used in Analytic
US Efforts in Vaccine Safety. Roger Baxter, MD California Immunization Coalition Summit 2011
 US Efforts in Vaccine Safety Roger Baxter, MD California Immunization Coalition Summit 2011 Conflicts of interest I receive research grants from Sanofi Pasteur, GSK, Merck, MedImmune, Novartis, Pfizer,
US Efforts in Vaccine Safety Roger Baxter, MD California Immunization Coalition Summit 2011 Conflicts of interest I receive research grants from Sanofi Pasteur, GSK, Merck, MedImmune, Novartis, Pfizer,
HARM. Definition modified from the IHI definition of Harm by the QUEST Harm Workgroup
 Indonesia Clinical epidemiology and Evidence Based Medicine (ICE(ICE-EBM) HARM Jarir At Thobari (FK UGM) Efficacious Medical intervention Harm HARM Unintended physical injury resulting from or contributed
Indonesia Clinical epidemiology and Evidence Based Medicine (ICE(ICE-EBM) HARM Jarir At Thobari (FK UGM) Efficacious Medical intervention Harm HARM Unintended physical injury resulting from or contributed
observational studies Descriptive studies
 form one stage within this broader sequence, which begins with laboratory studies using animal models, thence to human testing: Phase I: The new drug or treatment is tested in a small group of people for
form one stage within this broader sequence, which begins with laboratory studies using animal models, thence to human testing: Phase I: The new drug or treatment is tested in a small group of people for
Vaccinology 2017 Hanoi, Vietnam October 2017
 Vaccinology 2017 Hanoi, Vietnam October 2017 Active surveillance to assess vaccine benefits and risks Associate Professor Kristine Macartney National Centre for Immunisation Research and Surveillance University
Vaccinology 2017 Hanoi, Vietnam October 2017 Active surveillance to assess vaccine benefits and risks Associate Professor Kristine Macartney National Centre for Immunisation Research and Surveillance University
Epidemiologic Study Designs. (RCTs)
 Epidemiologic Study Designs Epidemiologic Study Designs Experimental (RCTs) Observational Analytical Descriptive Case-Control Cohort + cross-sectional & ecologic Epidemiologic Study Designs Descriptive
Epidemiologic Study Designs Epidemiologic Study Designs Experimental (RCTs) Observational Analytical Descriptive Case-Control Cohort + cross-sectional & ecologic Epidemiologic Study Designs Descriptive
Nationwide Oral Poliovirus Vaccination Campaign and the Incidence of Guillain-Barre Syndrome
 American Journal of Epidemiology Copyright O 1998 by The John3 Hopkins University School of Hygiene and Public Health AD rights reserved Vol. 147, No. 1 Printed In USA. Nationwide Oral Poliovirus Vaccination
American Journal of Epidemiology Copyright O 1998 by The John3 Hopkins University School of Hygiene and Public Health AD rights reserved Vol. 147, No. 1 Printed In USA. Nationwide Oral Poliovirus Vaccination
Combined oral contraceptives and risk of venous thromboembolism: nested case control studies using the QResearch and the CPRD databases
 Combined oral contraceptives and risk of venous thromboembolism: nested case control studies using the QResearch and the CPRD databases Yana Vinogradova, Carol Coupland, Julia Hippisley-Cox Web appendix:
Combined oral contraceptives and risk of venous thromboembolism: nested case control studies using the QResearch and the CPRD databases Yana Vinogradova, Carol Coupland, Julia Hippisley-Cox Web appendix:
What is Statistics? Ronghui (Lily) Xu (UCSD) An Introduction to Statistics 1 / 23
 What is Statistics? Some definitions of Statistics: The science of Statistics is essentially a branch of Applied Mathematics, and maybe regarded as mathematics applied to observational data. Fisher The
What is Statistics? Some definitions of Statistics: The science of Statistics is essentially a branch of Applied Mathematics, and maybe regarded as mathematics applied to observational data. Fisher The
Clinical Research Design and Conduction
 Clinical Research Design and Conduction Study Design M Mohsen Ibrahim, MD Cardiac Department Cairo University Algorithm for classification of types of clinical research Grimes DA and Schulz KF. Lancet
Clinical Research Design and Conduction Study Design M Mohsen Ibrahim, MD Cardiac Department Cairo University Algorithm for classification of types of clinical research Grimes DA and Schulz KF. Lancet
Observational Study Designs. Review. Today. Measures of disease occurrence. Cohort Studies
 Observational Study Designs Denise Boudreau, PhD Center for Health Studies Group Health Cooperative Today Review cohort studies Case-control studies Design Identifying cases and controls Measuring exposure
Observational Study Designs Denise Boudreau, PhD Center for Health Studies Group Health Cooperative Today Review cohort studies Case-control studies Design Identifying cases and controls Measuring exposure
Supplementary Online Content
 1 Supplementary Online Content Friedman DJ, Piccini JP, Wang T, et al. Association between left atrial appendage occlusion and readmission for thromboembolism among patients with atrial fibrillation undergoing
1 Supplementary Online Content Friedman DJ, Piccini JP, Wang T, et al. Association between left atrial appendage occlusion and readmission for thromboembolism among patients with atrial fibrillation undergoing
Purpose. Study Designs. Objectives. Observational Studies. Analytic Studies
 Purpose Study Designs H.S. Teitelbaum, DO, PhD, MPH, FAOCOPM AOCOPM Annual Meeting Introduce notions of study design Clarify common terminology used with description and interpretation of information collected
Purpose Study Designs H.S. Teitelbaum, DO, PhD, MPH, FAOCOPM AOCOPM Annual Meeting Introduce notions of study design Clarify common terminology used with description and interpretation of information collected
Pros and Cons of Clinical Trials vs. Observational Studies. Beth Devine PCORP Summer Institute July 14, 2015
 Pros and Cons of Clinical Trials vs. Observational Studies Beth Devine PCORP Summer Institute July 14, 2015 Outline Randomized Trials Definitions: Pragmatic-explanatory continuum indicator summary (PRECIS)
Pros and Cons of Clinical Trials vs. Observational Studies Beth Devine PCORP Summer Institute July 14, 2015 Outline Randomized Trials Definitions: Pragmatic-explanatory continuum indicator summary (PRECIS)
Background Describing epidemics Modelling epidemics Predicting epidemics Manipulating epidemics
 Mathematical Epidemiology of Infectious Diseases David Earn Background Describing epidemics Modelling epidemics Predicting epidemics Manipulating epidemics McMaster University Background Describing epidemics
Mathematical Epidemiology of Infectious Diseases David Earn Background Describing epidemics Modelling epidemics Predicting epidemics Manipulating epidemics McMaster University Background Describing epidemics
Health Studies 315. Clinical Epidemiology: Evidence of Risk and Harm
 Health Studies 315 Clinical Epidemiology: Evidence of Risk and Harm 1 Patients encounter (possibly) risky exposures Alcohol during pregnancy (fetal risk) Electromagnetic fields (cancer risk) Vasectomy
Health Studies 315 Clinical Epidemiology: Evidence of Risk and Harm 1 Patients encounter (possibly) risky exposures Alcohol during pregnancy (fetal risk) Electromagnetic fields (cancer risk) Vasectomy
Determinants of quality: Factors that lower or increase the quality of evidence
 Determinants of quality: Factors that lower or increase the quality of evidence GRADE Workshop CBO, NHG and Dutch Cochrane Centre CBO, April 17th, 2013 Outline The GRADE approach: step by step Factors
Determinants of quality: Factors that lower or increase the quality of evidence GRADE Workshop CBO, NHG and Dutch Cochrane Centre CBO, April 17th, 2013 Outline The GRADE approach: step by step Factors
Influenza & Other Modelling Activities at the Public Health Agency of Canada (PHAC)
 Influenza & Other Modelling Activities at the Public Health Agency of Canada (PHAC) Teresa Mersereau and Dena Schanzer, Public Health Agency of Canada Pan-InfORM and NCCID Co-hosted Workshop: Use of Mathematical
Influenza & Other Modelling Activities at the Public Health Agency of Canada (PHAC) Teresa Mersereau and Dena Schanzer, Public Health Agency of Canada Pan-InfORM and NCCID Co-hosted Workshop: Use of Mathematical
Case Studies in Ecology and Evolution. 10 The population biology of infectious disease
 10 The population biology of infectious disease In 1918 and 1919 a pandemic strain of influenza swept around the globe. It is estimated that 500 million people became infected with this strain of the flu
10 The population biology of infectious disease In 1918 and 1919 a pandemic strain of influenza swept around the globe. It is estimated that 500 million people became infected with this strain of the flu
Vaccine Safety Datalink (VSD)
 Vaccine Safety Datalink (VSD) Overview Immunization Safety Branch National Immunization Program Institute of Medicine (IOM) Reports on Vaccine Safety "Many gaps and limitations" in current knowledge +
Vaccine Safety Datalink (VSD) Overview Immunization Safety Branch National Immunization Program Institute of Medicine (IOM) Reports on Vaccine Safety "Many gaps and limitations" in current knowledge +
Design of Observational and Experimental Clinical Studies Schulz 9/17/2008. The worst-taught taught
 Faculty disclosure Design of Observational and Experimental Clinical Studies Kenneth F., PhD Dr. has no financial affiliations to disclose Note: Additional disclosure information is located within the
Faculty disclosure Design of Observational and Experimental Clinical Studies Kenneth F., PhD Dr. has no financial affiliations to disclose Note: Additional disclosure information is located within the
Vaccine Safety: Its everyone s business! PHO Rounds: Nov 19, 2013
 Vaccine Safety: Its everyone s business! PHO Rounds: Nov 19, 2013 Tara Harris, Nurse Consultant Shelley Deeks, Medical Director Immunization and Vaccine Preventable Diseases 1 Learning objectives 1. Describe
Vaccine Safety: Its everyone s business! PHO Rounds: Nov 19, 2013 Tara Harris, Nurse Consultant Shelley Deeks, Medical Director Immunization and Vaccine Preventable Diseases 1 Learning objectives 1. Describe
Epidemiologic study designs
 Epidemiologic study designs and critical appraisal of scientific papers Rolf H.H. Groenwold, MD, PhD Bio sketch MD, PhD in epidemiology Associate professor of Epidemiology at UMC Utrecht Research focus
Epidemiologic study designs and critical appraisal of scientific papers Rolf H.H. Groenwold, MD, PhD Bio sketch MD, PhD in epidemiology Associate professor of Epidemiology at UMC Utrecht Research focus
Asian Journal of Research in Biological and Pharmaceutical Sciences Journal home page:
 Review Article ISSN: 2349 4492 Asian Journal of Research in Biological and Pharmaceutical Sciences Journal home page: www.ajrbps.com REVIEW ON EPIDEMIOLOGICAL STUDY DESIGNS V.J. Divya *1, A. Vikneswari
Review Article ISSN: 2349 4492 Asian Journal of Research in Biological and Pharmaceutical Sciences Journal home page: www.ajrbps.com REVIEW ON EPIDEMIOLOGICAL STUDY DESIGNS V.J. Divya *1, A. Vikneswari
Vaccines for Children
 Vaccines for Children 0-9 old Our goal is to offer your family the best care possible, which includes making sure your child is up to date on all vaccines. DTaP (Diptheria, Tetanus, Pertussis) Vaccine
Vaccines for Children 0-9 old Our goal is to offer your family the best care possible, which includes making sure your child is up to date on all vaccines. DTaP (Diptheria, Tetanus, Pertussis) Vaccine
Trends in: Flu Immunization
 Trends in: Flu Immunization October 2015 Influenza is transmitted from person to person through droplets released from an infected person when they sneeze, cough or talk.[1] Although most people recover
Trends in: Flu Immunization October 2015 Influenza is transmitted from person to person through droplets released from an infected person when they sneeze, cough or talk.[1] Although most people recover
Risk and Uncertainty. The Precautionary Principle: Immunisation safety: risks, benefits and precautions.
 Risk and Uncertainty The Precautionary Principle: Immunisation safety: risks, benefits and precautions. Prof. David Salisbury CB Director of Immunisation, Department of Health. The precautionary principle
Risk and Uncertainty The Precautionary Principle: Immunisation safety: risks, benefits and precautions. Prof. David Salisbury CB Director of Immunisation, Department of Health. The precautionary principle
Approaches to Assessing Intussusception Risk in Developing Countries
 Approaches to Assessing Intussusception Risk in Developing Countries Jacqueline Tate Centers for Disease Control and Prevention Atlanta, GA, USA National Center for Immunization & Respiratory Diseases
Approaches to Assessing Intussusception Risk in Developing Countries Jacqueline Tate Centers for Disease Control and Prevention Atlanta, GA, USA National Center for Immunization & Respiratory Diseases
Drug prescriptions (Pharm) Exposure (36/48 months)
 ANNEX SECTION PART A - Study design: Figure 1 overview of the study design Drug prescriptions (Pharm) 2006-2010 Exposure (36/48 months) flexible time windows (e.g. 90 days) time index date (hospital discharge)
ANNEX SECTION PART A - Study design: Figure 1 overview of the study design Drug prescriptions (Pharm) 2006-2010 Exposure (36/48 months) flexible time windows (e.g. 90 days) time index date (hospital discharge)
Perspectives on Improving International Vaccine Safety
 Perspectives on Improving International Vaccine Safety Robert Ball, MD, MPH, ScM Director, Office of Biostatistics and Epidemiology Center for Biologics Evaluation and Research U.S. Food and Drug Administration
Perspectives on Improving International Vaccine Safety Robert Ball, MD, MPH, ScM Director, Office of Biostatistics and Epidemiology Center for Biologics Evaluation and Research U.S. Food and Drug Administration
Safety of Vaccines Used for Routine Immunization in the United States Executive Summary
 Evidence Report/Technology Assessment Number 215 Safety of s Used for Routine Immunization in the United States Executive Summary Background s are considered one of the greatest public health achievements
Evidence Report/Technology Assessment Number 215 Safety of s Used for Routine Immunization in the United States Executive Summary Background s are considered one of the greatest public health achievements
Trends in Pneumonia and Influenza Morbidity and Mortality
 Trends in Pneumonia and Influenza Morbidity and Mortality American Lung Association Epidemiology and Statistics Unit Research and Health Education Division November 2015 Page intentionally left blank Introduction
Trends in Pneumonia and Influenza Morbidity and Mortality American Lung Association Epidemiology and Statistics Unit Research and Health Education Division November 2015 Page intentionally left blank Introduction
Evidence-Based Medicine Journal Club. A Primer in Statistics, Study Design, and Epidemiology. August, 2013
 Evidence-Based Medicine Journal Club A Primer in Statistics, Study Design, and Epidemiology August, 2013 Rationale for EBM Conscientious, explicit, and judicious use Beyond clinical experience and physiologic
Evidence-Based Medicine Journal Club A Primer in Statistics, Study Design, and Epidemiology August, 2013 Rationale for EBM Conscientious, explicit, and judicious use Beyond clinical experience and physiologic
Vaccine-Preventable Diseases in Colorado s Children 2009 Sean O Leary MD, Carl Armon PhD, Joni Reynolds, RNC, MSN, James Todd MD
 State of the Health of Colorado s Children Vaccine-Preventable Diseases in Colorado s Children 29 Sean O Leary MD, Carl Armon PhD, Joni Reynolds, RNC, MSN, James Todd MD Vaccines have been highly effective
State of the Health of Colorado s Children Vaccine-Preventable Diseases in Colorado s Children 29 Sean O Leary MD, Carl Armon PhD, Joni Reynolds, RNC, MSN, James Todd MD Vaccines have been highly effective
Breast Cancer After Treatment of Hodgkin's Disease.
 Breast Cancer After Treatment of Hodgkin's Disease. Hancock SL, Tucker MA, Hoppe R Journal of the National Cancer Institute 85(1):25-31, 1993 Introduction The risks of second malignancy are increased in
Breast Cancer After Treatment of Hodgkin's Disease. Hancock SL, Tucker MA, Hoppe R Journal of the National Cancer Institute 85(1):25-31, 1993 Introduction The risks of second malignancy are increased in
2017 HEDIS Measures. PREVENTIVE SCREENING 2017 Measure Quality Indicator
 PREVENTIVE SCREENING Childhood Immunization Children who turn 2 during the Adolescent Immunization Adolescents who turn 13 during the Lead Screening Children who turn 2 during the Breast Cancer Screening
PREVENTIVE SCREENING Childhood Immunization Children who turn 2 during the Adolescent Immunization Adolescents who turn 13 during the Lead Screening Children who turn 2 during the Breast Cancer Screening
TECHNICAL DOCUMENT. Protocol for cluster investigations to measure influenza vaccine effectiveness in the EU/EEA.
 TECHNICAL DOCUMENT Protocol for cluster investigations to measure influenza vaccine effectiveness in the EU/EEA www.ecdc.europa.eu ECDC TECHNICAL DOCUMENT Protocol for cluster investigations to measure
TECHNICAL DOCUMENT Protocol for cluster investigations to measure influenza vaccine effectiveness in the EU/EEA www.ecdc.europa.eu ECDC TECHNICAL DOCUMENT Protocol for cluster investigations to measure
Clinical Quality Measures
 Core Measures Preventive Care and Screening Measure Pair: a. Tobacco Use Assessment, b. Tobacco Cessation Intervention. Percentage of patients aged 18 years and older who have been seen for at least 2
Core Measures Preventive Care and Screening Measure Pair: a. Tobacco Use Assessment, b. Tobacco Cessation Intervention. Percentage of patients aged 18 years and older who have been seen for at least 2
Sentinel Initiative Public Workshop. The Brookings Institution Marriott at Metro Center Washington, DC Tuesday, January 14, 2014
 Sentinel Initiative Public Workshop The Brookings Institution Marriott at Metro Center Washington, DC Tuesday, January 14, 2014 State of CBER s Mini-Sentinel Activities Michael Nguyen, MD Division of Epidemiology
Sentinel Initiative Public Workshop The Brookings Institution Marriott at Metro Center Washington, DC Tuesday, January 14, 2014 State of CBER s Mini-Sentinel Activities Michael Nguyen, MD Division of Epidemiology
LA SIERRA UNIVERSITY Department of Health and Exercise Science Fall 2011
 LA SIERRA UNIVERSITY Department of Health and Exercise Science Fall 2011 Course: HLED 467 Principles of Epidemiology (3 units) Instructor: Khaled Bahjri, MD, MPH (Assistant Professor) Office: 24871 Taylor
LA SIERRA UNIVERSITY Department of Health and Exercise Science Fall 2011 Course: HLED 467 Principles of Epidemiology (3 units) Instructor: Khaled Bahjri, MD, MPH (Assistant Professor) Office: 24871 Taylor
Vaccine Safety: the risk of undermining our successes at the global level
 Vaccine Safety: the risk of undermining our successes at the global level Pan-American Vaccine Safety Summit May 10, 2012 1 nsafe vaccine can have serious consequences Safety crises derail immunization
Vaccine Safety: the risk of undermining our successes at the global level Pan-American Vaccine Safety Summit May 10, 2012 1 nsafe vaccine can have serious consequences Safety crises derail immunization
Use of combined oral contraceptives and risk of venous thromboembolism: nested case-control studies using the QResearch and CPRD databases
 open access Use of combined oral contraceptives and risk of venous thromboembolism: nested case-control studies using the and databases Yana Vinogradova, Carol Coupland, Julia Hippisley-Cox Division of
open access Use of combined oral contraceptives and risk of venous thromboembolism: nested case-control studies using the and databases Yana Vinogradova, Carol Coupland, Julia Hippisley-Cox Division of
Vaccines: The Real Story
 Vaccines: The Real Story by, Ph.D. Qualifications Ph.D. in Nuclear Chemistry from the University of Rochester University Professor From 199-1995 NSF-Sponsored Scientist with More Than $2, In Research Grants
Vaccines: The Real Story by, Ph.D. Qualifications Ph.D. in Nuclear Chemistry from the University of Rochester University Professor From 199-1995 NSF-Sponsored Scientist with More Than $2, In Research Grants
Module 2: Fundamentals of Epidemiology Issues of Interpretation. Slide 1: Introduction. Slide 2: Acknowledgements. Slide 3: Presentation Objectives
 Slide 1: Introduction Issues of of Interpretation in in Epidemiologic Studies Developed through APTR Initiative to Enhance Prevention and Population Health Developed Education through in collaboration
Slide 1: Introduction Issues of of Interpretation in in Epidemiologic Studies Developed through APTR Initiative to Enhance Prevention and Population Health Developed Education through in collaboration
Deaths/yr Efficacy Use Prev Deaths/yr. Influenza 36,000 70% 60% 18,000. Pneumonia 40,000 60% 40% 20,000 HBV 6,000 90% 30% 4,000
 Tetanus, Diptheria, Pertussis,! Measles, Mumps, Rubella, Varicella, HPV, Polio Meningococcus, Pneumococcus,! Influenza, Hepatitis B, Hepatitis A,! H influenza, Rabies, Typhoid,! Yellow Fever, Japanese
Tetanus, Diptheria, Pertussis,! Measles, Mumps, Rubella, Varicella, HPV, Polio Meningococcus, Pneumococcus,! Influenza, Hepatitis B, Hepatitis A,! H influenza, Rabies, Typhoid,! Yellow Fever, Japanese
Observational study II
 Observational study II (QUANTITATIVE METHOD IV) หล กส ตรเวชศาสตร ช มชน RACM 302 1 AIMS Observational study and analytic study designs : Case-control study Cross-sectional study Ecological study Calculate
Observational study II (QUANTITATIVE METHOD IV) หล กส ตรเวชศาสตร ช มชน RACM 302 1 AIMS Observational study and analytic study designs : Case-control study Cross-sectional study Ecological study Calculate
Study Designs in Epidemiology
 Study Designs in Epidemiology Headlines Epidemiological research Classification of designs Qualitative methods Quantitative methods Choice of design Epidemiological Research Lab research: applies knowledge
Study Designs in Epidemiology Headlines Epidemiological research Classification of designs Qualitative methods Quantitative methods Choice of design Epidemiological Research Lab research: applies knowledge
UNDERSTANDING EPIDEMIOLOGICAL STUDIES. Csaba P Kovesdy, MD FASN Salem VA Medical Center, Salem VA University of Virginia, Charlottesville VA
 UNDERSTANDING EPIDEMIOLOGICAL STUDIES Csaba P Kovesdy, MD FASN Salem VA Medical Center, Salem VA University of Virginia, Charlottesville VA Study design in epidemiological research: Summary Observational
UNDERSTANDING EPIDEMIOLOGICAL STUDIES Csaba P Kovesdy, MD FASN Salem VA Medical Center, Salem VA University of Virginia, Charlottesville VA Study design in epidemiological research: Summary Observational
Table 1. Proposed Measures for Use in Establishing Quality Performance Standards that ACOs Must Meet for Shared Savings
 CMS-1345-P 174 Table 1. Proposed Measures for Use in Establishing Quality Performance Standards that ACOs Must Meet for Shared Savings AIM: Better Care for Individuals 1. Patient/Care Giver Experience
CMS-1345-P 174 Table 1. Proposed Measures for Use in Establishing Quality Performance Standards that ACOs Must Meet for Shared Savings AIM: Better Care for Individuals 1. Patient/Care Giver Experience
What is the risk of venous thromboembolism (VTE) in patients treated by an Outpatient Parenteral Antimicrobial Therapy (OPAT) Service?
 What is the risk of venous thromboembolism (VTE) in patients treated by an Outpatient Parenteral Antimicrobial Therapy (OPAT) Service? David A. Barr; Sharon Irvine; Neil D. Ritchie; Jay McCutcheon; R.
What is the risk of venous thromboembolism (VTE) in patients treated by an Outpatient Parenteral Antimicrobial Therapy (OPAT) Service? David A. Barr; Sharon Irvine; Neil D. Ritchie; Jay McCutcheon; R.
What is Influenza? Patricia Daly MD, FRCPC Medical Health Officer and Medical Director of Communicable Disease Control
 Vancouver Coastal Health & The Vancouver Coastal Health Research Institute presents: On Call with VGH Experts Lecture Series The Flu and You What is Influenza? Patricia Daly MD, FRCPC Medical Health Officer
Vancouver Coastal Health & The Vancouver Coastal Health Research Institute presents: On Call with VGH Experts Lecture Series The Flu and You What is Influenza? Patricia Daly MD, FRCPC Medical Health Officer
proposed set to a required subset of 3 to 5 measures based on the availability of electronic
 CMS-0033-P 143 proposed set to a required subset of 3 to 5 measures based on the availability of electronic measure specifications and comments received. We propose to require for 2011 and 2012 that EP's
CMS-0033-P 143 proposed set to a required subset of 3 to 5 measures based on the availability of electronic measure specifications and comments received. We propose to require for 2011 and 2012 that EP's
MMR vaccine and idiopathic thrombocytopaenic purpura
 Blackwell Science, LtdOxford, UKBCPBritish Journal of Clinical Pharmacology0306-5251Blackwell Publishing 200355Short ReportMMR vaccine and idiopathic thrombocytopaenic purpurac. Black et al. MMR vaccine
Blackwell Science, LtdOxford, UKBCPBritish Journal of Clinical Pharmacology0306-5251Blackwell Publishing 200355Short ReportMMR vaccine and idiopathic thrombocytopaenic purpurac. Black et al. MMR vaccine
Is There An Association?
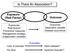 Is There An Association? Exposure (Risk Factor) Outcome Exposures Risk factors Preventive measures Management strategy Independent variables Outcomes Dependent variable Disease occurrence Lack of exercise
Is There An Association? Exposure (Risk Factor) Outcome Exposures Risk factors Preventive measures Management strategy Independent variables Outcomes Dependent variable Disease occurrence Lack of exercise
Intervention vs. Observational Trials:
 Intervention vs. Observational Trials: A Brief Introduction Introduction Just like an architect translates a vision for a building into blueprints, so an investigator translates their research idea into
Intervention vs. Observational Trials: A Brief Introduction Introduction Just like an architect translates a vision for a building into blueprints, so an investigator translates their research idea into
Handout 1: Introduction to the Research Process and Study Design STAT 335 Fall 2016
 DESIGNING OBSERVATIONAL STUDIES As we have discussed, for the purpose of establishing cause-and-effect relationships, observational studies have a distinct disadvantage in comparison to randomized comparative
DESIGNING OBSERVATIONAL STUDIES As we have discussed, for the purpose of establishing cause-and-effect relationships, observational studies have a distinct disadvantage in comparison to randomized comparative
115 remained abstinent. 140 remained abstinent. Relapsed Remained abstinent Total
 Chapter 10 Exercises 1. Intent-to-treat analysis: Example 1 In a randomized controlled trial to determine whether the nicotine patch reduces the risk of relapse among smokers who have committed to quit,
Chapter 10 Exercises 1. Intent-to-treat analysis: Example 1 In a randomized controlled trial to determine whether the nicotine patch reduces the risk of relapse among smokers who have committed to quit,
Data and Approaches in National and International Immunization Studies. Emory University, Schools of Public Health & Medicine & CHRSE
 Data and Approaches in National and International Immunization Studies Saad B. Omer, MBBS MPH PhD Emory University, Schools of Public Health & Medicine & CHRSE Outline Characteristics of vaccine refusers
Data and Approaches in National and International Immunization Studies Saad B. Omer, MBBS MPH PhD Emory University, Schools of Public Health & Medicine & CHRSE Outline Characteristics of vaccine refusers
