Approval / Acknowledgement Letter Ref No. / Diary no F.No.12-48/GSK/PAC- Synflorix/15-BD 25-Mar-15 2 GlaxoSmithKline Pharmaceuticals Limited.
|
|
|
- Beverly Long
- 6 years ago
- Views:
Transcription
1 1 GlaxoSmithKline Deletion of a n-significant in-process test limit during the purification process of the tetanus / diphtheria toxoids used in the manufacture of GSK's PCV vaccine [brand name: Synflorix] F /GSK/PAC- Synflorix/15-BD 25-Mar-15 2 GlaxoSmithKline Application for a Post Approval Change in Biological Product - To reclassify fill volume controls from in-process control to process parameter/measurements with alert and action levels performed during the filling process of GSK's MMRV vaccine - Measles, mumps, rubella and varicella vaccine (live) Ph. Eur. F /GSK/PAC- MMRV/15-BD 25-Mar-15 3 GlaxoSmithKline 4 GlaxoSmithKline 5 GlaxoSmithKline Asia Private Application for a Post Approval Change in Biological Product - Introduction of a mir to the Quality Control (QC) procedures for the final container volume determination and update to the final target fill volume controls for the diluent Water for Injection used for GSK's MMRV vaccine - Measles, mumps, rubella and varicella vaccine (live) Ph. Eur. I Addition of Tuas-Singapore site for manufacturing and QC testing of adsorbed movalent conjugate bulk for the 10th serotype (PS-5) used in GSK s Pneumococcal Polysaccharide Conjugate Vaccine (adsorbed) Ph. Eur.] [Brand Name: Synflorix] tification of a Post Approval Change in Biological Product - Deletion of a n-significant in-process test limit during the purification process of the tetanus toxoids used in the manufacture of GSK's Haemophilus Type b conjugated vaccine - Hib Vaccine (Brand Name: HIBERIX) F. NO /GSK/PAC- MMRV/15-BD 26-Mar-15 F /GSK/PAC- Synflorix/15-BD 28-Apr-15 F /GSK/PAC- Hibrix /15-BD 26-Jun-15
2 6 GlaxoSmithKline tification of Level Post Approval Changes for GSK's HBV vaccine - Hepatitis B Virus (rdna) Vaccine IP [Engerix B Paediatric/ Adult] - Correction of the compendial references to the analytical methods mentioned in the registration dossier 1. The purity by Sodium Dodecyl Sulphate Drug Substance: HBsAg PolyAcrylamide Gel Electrophoresis (SDS-PAGE) 2. The aluminium identity testing method for Aluminium Hydroxide gel or Al(OH)3 F /GSK/PAC- Rotarix/15-BD 01-Oct-15 7 GlaxoSmithKline Asia Private 8 GlaxoSmithKline Asia Private 9 GlaxoSmithKline tification of Post Approval Change (Level ) in Biological Product Reduction of PRN load on Q-Sepharose column Vaccines Impacted: - Diphtheria Tetanus and Pertussis (Acellular Component) Vaccine (Adsorbed) Ph. Eur. [Infanrix ] - Diphtheria, Tetanus and Pertussis (Acellular Component) Vaccine (Adsorbed, reduced antigens content) Ph. Eur. [Boostrix ] tification of a Post Approval Change for GSK's Human Rotavirus Vaccine (HRV) - Rotavirus Vaccine (Live Attenuated, Oral) IP [Brand Name: Rotarix] Change in the name of the diluent manufacturing site (intermediate site) from Glaxo Wellcome NDB to Aspen NDB tification of a Post Approval Change for GSK's MMRV Vaccine - Measles, Mumps, Rubella and Varicella vaccine (live) Ph. Eur. [Brand Name: Priorix Tetra] Change in the name of the diluent manufacturing site (intermediate site) from Glaxo Wellcome NDB to Aspen NDB DCGI Diary v-15 F /GSK/PAC- Rotarix/16-BD 18-Dec-15 DCGI Diary Dec-15
3 10 GlaxoSmithKline Asia Private 11 GlaxoSmithKline Asia Private 12 GlaxoSmithKline 13 GlaxoSmithKline & Correction of the compendial references to the analytical methods mentioned in the registration dossier -The assay and loss on drying tests of dipotassiumphoshate trihydrate -The sterility test by direct iculation -The formaldehyde content by spectrophotometry -The aluminium identity testing method for Aluminium Hydroxide gel or Al(OH)3 Vaccine impacted: Diphtheria, Tetanus and Pertussis (Acellular Component) Vaccine (Adsorbed, reduced antigens content) Ph. Eur. [BOOSTRIX] Application for Post Approval Change in Biological Product - Addition of an additional freeze-dryer at the registered Fidia manufacturing site (FIDIA Farmaceutici S.p.a., Padova, Italy) for the production of GSK's Measles, Mumps and Rubella Vaccine (live) IP [MMR] tification of a Post Approval Change in Biological Product - Addition of building SG05 for n-sterile further purification steps of the tetanus & diphtheria toxoids used as carrier protein in manufacture of GSK's pneumococcal conjugated vaccine [brand name: Synflorix] Application for the below post approval s 1. Use of building Wavre rd 30 (WN30) at the GSK biologicals, Wavre, Belgium site as an additional facility for formulation 2. Addition of alternative container for DP intermediate storage 1. Hepatitis A (Inactivated) and Hepatitis B (rdna) Vaccine (Adsorbed) IP [HAV-HAB] 2. Inactivated Hepatitis A Vaccine (Adsorbed) IP [HAV] F /GSK/PAC- Boostrix/15-BD 10-Jul-15 F /GSK/PAC- MMRV/15-BD 3-Aug-15 F /GSK/PAC- Synflorix/15-BD 17-Aug-15 F /GSK/PAC- Hep A&B/15-BD 1-Oct-15
4 14 GlaxoSmithKline 15 GlaxoSmithKline Asia Private Application for Post Approval Change in Biological Product - New reference standard for Hepatitis A antigen content by Enzyme Linked Immusorbent Assays (ELISA) and Potency hepatitis A antigen by ELISA assays performed as part of in process and QC release testing of the drug substance and during QC release testing Impacted vaccines: 1. Hepatitis A (Inactivated) and Hepatitis B (rdna) Vaccine (Adsorbed) IP [HAV-HAB] 2. Inactivated Hepatitis A Vaccine (Adsorbed) IP [HAV] Application for Post Approval Change in Biological Product Use of building Wavre rd 30 (WN30) at the GSK Biologicals, Wavre, Belgium site as an additional facility for formulation of Human Papillomavirus Vaccine rdna Ph. Eur. [CERVARIX] F /GSK/PAC- Hep.A/15-BD 26-Oct-15 F /GSK/PAC- Cervarix/ 15-BD dated 01-Dec Dec-15
5 16 GlaxoSmithKline 17 GlaxoSmithKline Asia Private Application of Post Approval Change in Biological Product Revision of the vial rubber stoppers release specifications for various lyophilized and liquid vaccines Vaccines Impacted: Pneumococcal Polysaccharide Conjugate Vaccine (adsorbed) Ph. Eur. [Synflorix ] Hepatitis A (Inactivated) and Hepatitis B (rdna) Vaccine (Adsorbed) IP [Twinrix Adult] Varicella Vaccine, Live IP [Varilrix ] Inactivated Hepatitis A Vaccine (Adsorbed) IP [Havrix 1440 (Adult)/720 (Junior)] Poliomyelitis Vaccine (Inactivated) IP [Poliorix] Hepatitis B Vaccine (rdna) IP (Genetically Engineered) [Engerix B 20 mcg/1 ml (Adult) / Engerix B 10 mcg/0.5 ml (Paediatric)] Measles, Mumps, Rubella and Varicella vaccine (Live) Ph. Eur. [Priorix Tetra] Application of Post Approval Change in Biological Product Revision of the vial rubber stoppers release specifications for various lyophilized and liquid vaccines Vaccines Impacted: - Human Papillomavirus Vaccine rdna Ph Eur. [Cervarix ] - Rotavirus Vaccine (Live Attenuated, Oral) IP [Rotarix ] - Diphtheria Tetanus and Pertussis (Acellular Component) Vaccine (Adsorbed) Ph. Eur. [Infanrix ] - Haemophilus type b conjugated vaccine IP [Hiberix ] - Meningococcal Polysaccharide Vaccine IP [Mencevax ACWY] - Measles, Mumps and Rubella Vaccine (live) IP [Priorix ] - Diphtheria, Tetanus and Pertussis (Acellular Component) Vaccine (Adsorbed, reduced antigens content) Ph. Eur. F /GSK/PAC- Many Vaccines/15-BD 09-Sep-15 F /GSK/PAC- Many Vaccines/15-BD 09-Sep-15
6 [Boostrix ] 18 GlaxoSmithKline Asia Private 19 GlaxoSmithKline Asia Private 20 GlaxoSmithKline Asia Private & Application for approval of Post Approval Changes for GSK's HRV vaccine - Rotavirus Vaccine (Live Attenuated, Oral) IP [ROTARIX] - Correction of the compendial references to the analytical methods mentioned in the registration dossier 1. The sterility test Mycoplasma by culture 2. The assay and loss on drying tests of dipotassium phosphate trihydrate Application for Level Post Approval Change - Change in the retest period (or shelf-life) for the drug substance, involving: Extension of Shelf life of Diphtheria Tetanus (DT) concentrate from 24 months to 36 months Impacted GSK vaccines: Diphtheria, Tetanus and Pertussis (Acellular Component) Vaccine (Adsorbed, reduced antigens content) (dtpa) [Brand Name: BoostrixJ Diphtheria, Tetanus and Pertussis (Acellular Component) Vaccine (Adsorbed) (DTPa) [Brand Name: lnfanrix] Correction of the compendial references to the analytical methods mentioned in the registration dossier -The assay and loss on drying tests of dipotassiumphoshate trihydrate -The sterility test by direct iculation -The formaldehyde content by spectrophotometry -The aluminium identity testing method for Aluminium Hydroxide gel or Al(OH)3 Vaccine impacted: Diphtheria, Tetanus and Pertussis (Acellular Component) Vaccine (Adsorbed) Ph. Eur. [INFANRIX] F /GSK/PAC- Rotarix/15-BD. 12-Feb-16 F /GSK/PAC- DT/15-BD 12-Feb-16 F /GSK/PAC- Infanrix/16-BD 16-Feb-16
7 21 GlaxoSmithKline Asia Private 22 GlaxoSmithKline 23 GlaxoSmithKline Asia Private Application for Post Approval Change in Biological Products - Replacement of dtpa reference vaccine used in mouse immugenicity assay for Acellular pertussis (Pa) antigens for GSK's dtpa vaccine - Diphtheria, Tetanus and Pertussis (Acellular Component) Vaccine (Adsorbed) Ph. Eur. [Brand Name: BOOSTRIXJ tification of Level Post-approval : Decommission of packaging and labeling activities in Pre-filled Syringes in Building DR A/B located in Dresden, Germany 1. Hepatitis A Inactivated) and Hepatitis B (rdna) Vaccine (Adsorbed) IP [Twinrix Adult] 2. Inactivated Hepatitis A Vaccine (Adsorbed) IP [Havrix Adult/Pediatric] Application for Post-Approval Change (Level ) in Biological Product - Change in the specification of raw material addition of a test of the foam breaking function of the antifoam emulsion used as a raw material 1. Diphtheria, Tetanus and Pertussis (Acellular Component) Vaccine (Adsorbed) Ph. Eur. [lnfanrix] 2. Diphtheria, Tetanus and Pertussis (Acellular Component) Vaccine (Adsorbed, Reduced Antigens content) Ph. Eur. [Boostrix] F /GSK/PACdTpa/16-BD 19-Feb-16 F /GSK/PAC- MV /16-BD 8-Mar-16 F /GSK/PAC- Boostrix & Infanrix/ 16- BD 8-Mar-16
8 24 GlaxoSmithKline Asia Private 25 GlaxoSmithKline 26 GlaxoSmithKline Asia Private & Application for Post Approval Change in Biological Product Correction of the compendial references to the analytical methods mentioned in the registration dossier for GSK s HPV vaccine [brand name: Cervarix] 1. Sterility test Mycoplasma by culture 2. Purity by SDS-PAGE 3. Aluminium identity test for Al(OH)3 gel 4. Protein content by total nitrogen assay tification of Level Ill Post Approval Changes for GSK's HAB vaccine- Hepatitis A (Inactivated) and Hepatitis B (rdna) Vaccine (Adsorbed) IP [Twinrix Adult] Introduction of the sterility tests by membrane filtration as Quality Control (QC) release test on adsorbed Hepatitis A antigen (ADFIHA) intermediate final bulk Introduction of the sterility tests by membrane filtration as Quality Control (QC) release test on adsorbed Hepatitis B antigen (HBsAg) concentrate (AHEF) intermediate final bulk Application for Post-Approval Change in Biological Product - Increase in the lifetime of the TFF membrane filter used in the manufacture of MPL powder, an excipient used in GSK s HPV Vaccine [brand name: Cervarix] F /GSK/PAC- HPV/16-BD dated 08- Mar Mar-16 F /GSK/PAC- HAB vaccine/16-bd 8-Mar-16 F /PAC- Cervarix/16-BD dated 08-Mar Mar-16
9 27 GlaxoSmithKline Asia Private 28 GlaxoSmithKline 29 GlaxoSmithKline I & tification of Level Post-approval : Decommission of packaging and labeling activities in Pre-filled Syringes in Building DR A/B located in Dresden, Germany 1. Diphtheria, Tetanus and Pertussis (Acellular Component) Vaccine (Adsorbed) Ph. Eur. [lnfanrix] 2. Diphtheria, Tetanus and Pertussis (Acellular Component) Vaccine (Adsorbed, Reduced Antigens content) Ph. Eur. [Boostrix] Application for Post-Approval Changes (Level I & Level ) in Biological Product - Scale up of microfluidised PS5 and PS19F bulks and shelf-life extension of microfluidised PS5 used in the manufacture of GSK s pneumococcal vaccine (Synflorix) & Application for Post Approval Changes in Biological Product Correction of the compendial references to the analytical methods used in the testing of raw material for DS and DS intermediate for GSK s pneumococcal polysaccharide conjugate vaccine [brand name: Synflorix] -Dipotassium phosphate trihydrate (K2PO4.3H2O) -Purified Tetanus Toxoid (TT) bulk DCGI Diary Mar-16 F /GSK/PAC- Synflorix/ 16-BD 8-Apr-16 F /GSK/PAC- Synflorix/ 16-BD 8-Apr GlaxoSmithKline & Application for approval of Post Approval Changes for GSK's HAV vaccine - Inactivated Hepatitis A Vaccine (Adsorbed) IP (HAVRIX) - Correction of the compendial references to the analytical methods mentioned in the registration dossier 1. The sterility test Mycoplasma by culture 2. The aluminum identity testing method for Aluminum Hydroxide gel or Al(OH)3 3. The determination of formaldehyde content by spectrophotometry F /GSK/PAC- HAV/16-BD 11-Apr-16
10 31 GlaxoSmithKline Asia Private 32 GlaxoSmithKline 33 GlaxoSmithKline tification of a Level Ill Post Approval Change for Suppression of formulation, filling and lyophilization activities in RX.16.1 site for Vials in Rixensart, Belgium 1. Rotavirus Vaccine (Live Attenuated, Oral) IP [Rotarix] tification of a Level Ill Post Approval Change for Suppression of formulation, filling and lyophilization activities in RX.16.1 site for Vials in Rixensart, Belgium 1. Inactivated Hepatitis A Vaccine (Adsorbed) IP [Havrix Adult/Pediatric] 2. Poliomyelitis Vaccine (Inactivated) IP [Poliorix] & Application for Post Approval Changes in Biological Product - Change in description test and introduction of nephelometry on Hepatitis B surface antigen (HBsAg) bulk of GSK's Hepatitis B containing vaccines. Impacted vaccines: 1. Hepatitis A (Inactivated) and Hepatitis B (rdna) Vaccine (Adsorbed) IP [HAV-HAB] 2. Hepatitis B Virus (rdna) Vaccine IP [HBV] F /GSK/PAC- Rotavirus/16-BD 18-Apr-16 F /GSK/PAC- Hep-A, Polio/16-BD 18-Apr-16 F /GSK/PAC- Hepatitis/16-BD 25-Apr GlaxoSmithKline Asia Private (requiring CDL review) Application for Level Post-Approval Change Change in the re test period (or shelf Life) for the drug substance, involving: Extension of Shelf Life of PRN bulk from 24 months to 36 months. Impacted Vaccines: Diphtheria, Tetanus and Pertussis (Acellular Component) Vaccine (Adsorbed, Reduced Antigen Content) Ph. Eur. [Brand Name: BOOSTRIX] Diphtheria, Tetanus and Pertussis (Acellular Component) F /GSK/PAC - MV/16-BD 4-May-16
11 Vaccine (Adsorbed) Ph. Eur. [INFANRIX] 35 GlaxoSmithKline Asia Private Replacement of the current in-house working reference serum (WRS) used in the in vitro part of the potency assay (one-dose test) for the acellular Pertussis (Pa) components of Pa-containing vaccines Impacted Vaccines: Diphtheria, Tetanus and Pertussis (Acellular Component) Vaccine (Adsorbed, Reduced Antigen Content) Ph. Eur. [Brand Name: BOOSTRIX] Diphtheria, Tetanus and Pertussis (Acellular Component) Vaccine (Adsorbed) Ph. Eur. [INFANRIX] F /GSK/PAC- Boostrix & Infanrix / 16- BD 4-May GlaxoSmithKline 37 GlaxoSmithKline Asia Private (requiring CDL review) Application for Post-Approval Change (Level ) in Biological Product Extension of shelf-life for GSK s pneumococcal polysaccharide conjugate vaccine (Synflorix) in Pre-Filled Syringe from 36 to 48months. tification of Post Approval Change (Level ) in Biological Product Introduction of the sterility test by membrane filtration as QC release test on ATTOAA intermediate used in the formulation of GSK s dtpa vaccine [brand name: Boostrix] F /GSK/PAC- Synflorix/ 16-BD 4-May-16 F /GSK/PAC- Boostrix/16-BD 13-May-16
12 38 GlaxoSmithKline & Application for Post-Approval Changes (Level and Level ) in Biological Product Removal of compendial references for analytical procedures o Sterility test Mycoplasma detection by culture method o Neomycin sulphate content by radial diffusion o Particle count test for diluent (WFI) Impacted vaccines: Measles, Mumps, Rubella and Varicella vaccine (Live) Ph. Eur. [Priorix Tetra] Varicella Vaccine, Live IP [Varilrix ] F /GSK/PAC- MMRV vaccine/ 16-BD 24-May GlaxoSmithKline Asia Private 40 GlaxoSmithKline 41 GlaxoSmithKline Asia Private & Application for Post-Approval Changes (Level and Level ) in Biological Product Removal of compendial references for analytical procedures used in GSK s MMR vaccine [brand name: Priorix] o Sterility test Mycoplasma detection by culture method o Neomycin sulphate content by radial diffusion o Particle count test for diluent (WFI) : tification of Post-Approval Change (Level ) in Biological Product - modification in the ph target and range used at the final ph adjustment during preparation of purified TT bulk, a carrier protein used in GSK s Pneumococcal Polysaccharide Conjugate Vaccine (adsorbed) Ph. Eur. [brand name: SYNFLORIX] tification of Post-Approval Change (Level ) in Biological Product Introduction of BioReliance, Rockville, MD, USA as additional QC site responsible to conduct the Rabbit Pyrogen assay test for MPL powder, an excipient used in GSK s HPV vaccine [brand name: Cervarix] F /GSK/PAC- MMR vaccine/16-bd 24-May-16 F /GSK/PAC- Synflorix/16-BD 3-Jun-16 F /GSK/PAC- HPV /136-BD 15-Jun-16
13 42 GlaxoSmithKline Asia Private 43 GlaxoSmithKline 44 GlaxoSmithKline Application for Post-Approval Change (Level ) in Biological Product Change of source of raw material cystine for Tetanus - Marburg 1. Diphtheria, Tetanus and Pertussis (Acellular Component) Vaccine (Adsorbed) Ph. Eur. [lnfanrix) 2. Diphtheria, Tetanus and Pertussis (Acellular Component) Vaccine (Adsorbed, Reduced Antigens content) Ph. Eur. [Boostrix) Application for Approval of a Level Post-Approval Change - Introduction of Maximum Storage Period for Hepatitis A (HAV) Iculum 1. Hepatitis A (Inactivated) and Hepatitis B (rdna) Vaccine (Adsorbed) IP [Twinrix Adult] 2. Inactivated Hepatitis A Vaccine (Adsorbed) IP [Havrix Adult/Pediatric] & Application for approval of Post Approval Changes for GSK's HAB vaccine - Hepatitis A (Inactivated) and Hepatitis B (rdna) Vaccine (Adsorbed) IP [Twinrix Adult] - Correction of the compendia} references to the analytical methods mentioned in the registration dossier 1. The sterility test Mycoplasma by culture 2. The purity by Sodium Dodecyl Sulphate PolyAcrylamide Gel Electrophoresis (SDS-PAGE) 3. The aluminum identity testing method for Aluminum Hydroxide gel or Al(OH)3 4. The determination of formaldehyde content by Spectrophotometry F /GSK/PAC- Boostrix & Infanrix/ 16- BD 27-Jun-16 F /GSK/PAC- Twinrix & Havrix/16-BD 8-Aug-16 F /GSK/PAC- HAV & HBV/ 16-BD 8-Aug-16
14 45 GlaxoSmithKline 46 GlaxoSmithKline 47 GlaxoSmithKline 48 GlaxoSmithKline 49 GlaxoSmithKline (requiring CDL review), but seeking approval and Application for Approval of a Level Post-Approval Change for correction of Compendial References to the Analytical methods mentioned in the Registration Dossier for GSK's HBV Containing Vaccines. Vaccine impacted: Hepatitis A Inactivated) and Hepatitis B (rdna) Vaccine (Adsorbed) IP [Twinrix Adult] Application for approval of Post-Approval Changes in Biological Product for GSK's Varicella vaccine [brand name: Varilrix] 1) Alignment of Varilrix table of composition in line with source country (Belgian) approval 2) Inclusion of the reference to Ph. Eur. mograph 0806 for Arginine Application for Post-Approval Change (Level ) in Biological Product Introduction of a shelf-life of 96 months at -196 C for the Varicella iculum used in the manufacturing process of GSK s MMRV and Varicella vaccines based on available stability data Vaccines Impacted: - MMRV vaccine [brand name: PRIORIX-TETRA] - Varicella vaccine [brand name: VARILRIX] Application for Post-Approval Changes (Level and Level ) in Biological Product - Change of specification for the antigenic activity performed at release and during stability evaluation of HBV bulk, a drug substance of GSK s HAB vaccine [brand name: TWINRIX ADULT] Application for Post-Approval Changes (Level ) Update to the description of QC release tests (Identity and Potency) performed during the manufacture of the rubella virus live attenuated bulk (DS) and on the final container (DP) F /GSK/PAC- Twinrix/16-BD 30-Aug-16 F /GSK/PAC- Varilrix/16-BD 30-Aug-16 F /GSK/PAC- MMRV & Varicella/ 16- BD 30-Aug-16 F /GSK/PAC- HAB/ 16-BD 22-Sep-16 F /GSK/PAC- MMRV I 16-BD 26-Sep-16
15 Vaccine Impacted: MMRV vaccine [brand name: PRIORIX-TETRA] 50 GlaxoSmithKline Asia Private 51 GlaxoSmithKline 52 GlaxoSmithKline Asia Private (requiring CDL review), category according to the guidance, category according to the guidance Application for Post-Approval Changes (Level ) Update to the description of QC release tests (Identity and Potency) performed during the manufacture of the rubella virus live attenuated bulk (DS) and on the final container (DP) Vaccine Impacted: MMR vaccine [brand name: PRIORIX] Application for Post Approval Changes (Level ) in Biological Product Update to the description within the registration files for internal controls, solutions and media Impacted vaccines: - Measles, Mumps, Rubella and Varicella Vaccine (Live) Ph. Eur. [Priorix-Tetra] - Varicella Vaccine, Live IP [Varilrix] Application for Post Approval Changes (Level ) in Biological Product Update to the description within the registration files for internal controls, solutions and media Impacted vaccine: - Measles, Mumps & Rubella Vaccine (Live) IP [Priorix] F /GSK/PAC- MMR/16-BD 26-Sep-16 F /GSK/PAC- MMRV & Varicella /16- BD 26-Sep-16 F /GSK/PAC- MMR/16-BD 26-Sep-16
16 53 GlaxoSmithKline Asia Private 54 GlaxoSmithKline Asia Private Application for Post-Approval Change (Level ) in Biological Product - Replacement of the current Pertactin (PRN) Reference Standard by a new proposed batch 1. Diphtheria, Tetanus and Pertussis (Acellular Component) Vaccine (Adsorbed, reduced antigens content) Ph. Eur. [brand name: BOOSTRIX] 2. Diphtheria, Tetanus and Pertussis (Acellular Component) Vaccine (Adsorbed) Ph. Eur. [brand name: INFANRIX] tification of a Level Post Approval Change in Biological Product- Tightening of Temperature Criterion of Diluent Used in GSK's Rotavirus Vaccine (Live Attenuated, Oral) IP [Rotatix]. F. / 12-64/GSK/PAC- Boostrix&Infanrix/16- BD 9-Dec-16 DCGI Diary Dec GlaxoSmithKline 56 GlaxoSmithKline Application for Approval of a Level Post Approval Change in Biological Products - Revision of Glass Vial Container Release Specifications for all Vaccine Formulations Filled in Glass Vial Containers. 1. Inactivated Hepatitis A Vaccine (Adsorbed) IP [Havrix 1440 (Adult)/720 (Junior)] 2. Poliomyelitis Vaccine (Inactivated) IP [Poliorix] 3. Measles, Mumps, Rubella and Varicella Vaccine (Live) Ph. Eur. [Priorix-Tetra] 4. Pneumococcal Polysaccharide Conjugate Vaccine (Adosrbed) Ph. Eur. [Synflorix] 5. Varicella Vaccine, Live IP [Varilrix] tification of Post Approval Change (Level ) in Biological Product Use of a new working seed of Streptococcus pneumoniae serotype 18C used to produce PS18C bulks, an intermediate in the production of GSK s pneumococcal vaccine (SYNFLORIX) F /GSK/PAC- Many vaccines/16-bd 21-Dec-16 DCGI Diary Dec-16
17 57 GlaxoSmithKline 58 GlaxoSmithKline Asia Private Application for Post-Approval Change (Level ) in Biological Product Extension of shelf-life for GSK s pneumococcal polysaccharide conjugate vaccine [SYNFLORIX] in 1-dose and 2- dose Vial from 36 to 48months Application for Approval of a Level Post Approval Change in Biological Products - Revision of Glass Vial Container Release Specifications for all Vaccine Formulations Filled in Glass Vial Containers. 1. Human Papillomavirus Vaccine rdna Ph. Eur. [Cervarix] 2. Measles, Mumps and Rubella Vaccine (Live) [Priorix] 3. Rotavirus Vaccine (Live Attenuated,-Oral) LP. [Rotarix] F / GSK/PAC- Synflorix/ 16-BD 28-Dec-16 F /GSK/PAC- Many vac/16-bd 9-Jan-17
Advisory Committee on Immunization Practices VACCINE ACRONYMS
 Vaccine Acronyms Page 1 of 5 Advisy Committee on Immunization Practices VACCINE ACRONYMS Vaccines Included in the Immunization Schedules f Children, Adolescents, and Adults Following is a table of standardized
Vaccine Acronyms Page 1 of 5 Advisy Committee on Immunization Practices VACCINE ACRONYMS Vaccines Included in the Immunization Schedules f Children, Adolescents, and Adults Following is a table of standardized
New Jersey Department of Health Vaccine Preventable Disease Program Childhood and Adolescent Recommended Vaccines
 New Jersey Department of Health Vaccine Preventable Disease Program Childhood and Adolescent Recommended Vaccines Antigens Vaccine Approved Age Daptacel Diphtheria, Tetanus, and acellular Pertussis (DTaP)
New Jersey Department of Health Vaccine Preventable Disease Program Childhood and Adolescent Recommended Vaccines Antigens Vaccine Approved Age Daptacel Diphtheria, Tetanus, and acellular Pertussis (DTaP)
TRICARE Retail Vaccination Program Vaccine List - September 2018*
 Diphtheria, tetanus and pertussis Diphtheria and tetanus toxoids adsorbed acellular pertussis adsorbed DT DTaP Diphtheria and tetanus toxoids adsorbed Daptacel, Infanrix Tetanus and diphtheria toxoids
Diphtheria, tetanus and pertussis Diphtheria and tetanus toxoids adsorbed acellular pertussis adsorbed DT DTaP Diphtheria and tetanus toxoids adsorbed Daptacel, Infanrix Tetanus and diphtheria toxoids
Changes to the National Immunisation Schedule
 17 December 2013 Changes to the National Immunisation Schedule PHARMAC is pleased to announce decisions related to the National Immunisation Schedule (NIS) that will take effect from 1 July 2014. This
17 December 2013 Changes to the National Immunisation Schedule PHARMAC is pleased to announce decisions related to the National Immunisation Schedule (NIS) that will take effect from 1 July 2014. This
Bharat Biotech. Hyderabad. Type of PAC (Supplement and Notifiable / Permission Granted by CDSCO) S. No Product
 Bharat Biotech Hyderabad S. No Product Type of PAC (Supplement and / Permission Granted by CDSCO) PAC Details Date of NOC in manufacturing process of Oral Polio F.No. 12-63/BHARAT/(PACOPV)/13-BD 30.08.2013
Bharat Biotech Hyderabad S. No Product Type of PAC (Supplement and / Permission Granted by CDSCO) PAC Details Date of NOC in manufacturing process of Oral Polio F.No. 12-63/BHARAT/(PACOPV)/13-BD 30.08.2013
Advisory Committee on Immunization Practices VACCINE ACRONYMS
 May 1, 2015 Vaccine Acronyms Page 1 of 5 Advisy Committee on Immunization Practices VACCINE ACRONYMS Vaccines Included in the Immunization Schedules f Children, Adolescents, and Adults Following is a table
May 1, 2015 Vaccine Acronyms Page 1 of 5 Advisy Committee on Immunization Practices VACCINE ACRONYMS Vaccines Included in the Immunization Schedules f Children, Adolescents, and Adults Following is a table
PREVENTIVE IMMUNIZATIONS. PREVENTIVE IMMUNIZATIONS These codes do not have a diagnosis code requirement for preventive benefits to apply.
 An immunization that does not fall under one of the exclusions in the Certificate of Coverage is considered covered after the following conditions are satisfied: (1) FDA approval; (2) explicit ACIP recommendation
An immunization that does not fall under one of the exclusions in the Certificate of Coverage is considered covered after the following conditions are satisfied: (1) FDA approval; (2) explicit ACIP recommendation
Healthy People 2020 objectives were released in 2010, with a 10-year horizon to achieve the goals by 2020.
 Appendix 1: Healthy People 2020 Immunization-related Objectives Healthy People provides science-based, 10-year national objectives for improving the health of all Americans. For three decades, Healthy
Appendix 1: Healthy People 2020 Immunization-related Objectives Healthy People provides science-based, 10-year national objectives for improving the health of all Americans. For three decades, Healthy
PREVENTIVE IMMUNIZATIONS. PREVENTIVE IMMUNIZATIONS These codes do not have a diagnosis code requirement for preventive benefits to apply.
 An immunization that does not fall under one of the exclusions in the Certificate of Coverage is considered covered after the following conditions are satisfied: (1) FDA approval; (2) explicit ACIP recommendation
An immunization that does not fall under one of the exclusions in the Certificate of Coverage is considered covered after the following conditions are satisfied: (1) FDA approval; (2) explicit ACIP recommendation
GENERAL IMMUNIZATION GUIDE FOR CHILDCARE PROVIDERS August 2018 **CHILD VACCINES** DIPHTHERIA, TETANUS, PERTUSSIS VACCINES
 GENERAL IMMUNIZATION GUIDE FOR CHILDCARE PROVIDERS August 2018 **CHILD VACCINES** DIPHTHERIA, TETANUS, PERTUSSIS VACCINES DTaP: Diphtheria, Tetanus, acellular Pertussis Vaccine Infanrix Licensed in 1997
GENERAL IMMUNIZATION GUIDE FOR CHILDCARE PROVIDERS August 2018 **CHILD VACCINES** DIPHTHERIA, TETANUS, PERTUSSIS VACCINES DTaP: Diphtheria, Tetanus, acellular Pertussis Vaccine Infanrix Licensed in 1997
STANWOOD-CAMANO SCHOOL DISTRICT REQUEST FOR PART-TIME ATTENDANCE OR ANCILLARY SERVICES FROM PRIVATE SCHOOL PUPIL
 STANWOOD-CAMANO SCHOOL DISTRICT REQUEST FOR PART-TIME ATTENDANCE OR ANCILLARY SERVICES FROM PRIVATE SCHOOL PUPIL 114 F2 Students Please complete ALL information: Name of Pupil Grade Address of Pupil City
STANWOOD-CAMANO SCHOOL DISTRICT REQUEST FOR PART-TIME ATTENDANCE OR ANCILLARY SERVICES FROM PRIVATE SCHOOL PUPIL 114 F2 Students Please complete ALL information: Name of Pupil Grade Address of Pupil City
Vaccine Label Examples
 Vaccine Label Examples With the large amount of vaccine carried in most clinics, staff can easily become confused about vaccines within the storage unit. Labeling the area where vaccines are stored can
Vaccine Label Examples With the large amount of vaccine carried in most clinics, staff can easily become confused about vaccines within the storage unit. Labeling the area where vaccines are stored can
VACCINE MANAGEMENT. Recommendations for Handling and Storage of Selected Biologicals. January 2001 DEPARTMENT OF HEALTH AND HUMAN SERVICES
 VACCINE MANAGEMENT Recommendations for Handling and Storage of Selected Biologicals January 2001 DEPARTMENT OF HEALTH AND HUMAN SERVICES DTaP: Diphtheria Toxoid, Tetanus Toxoid, Acellular Pertussis Vaccine
VACCINE MANAGEMENT Recommendations for Handling and Storage of Selected Biologicals January 2001 DEPARTMENT OF HEALTH AND HUMAN SERVICES DTaP: Diphtheria Toxoid, Tetanus Toxoid, Acellular Pertussis Vaccine
Vaccine Label Examples
 Vaccine Label Examples With the large amount of vaccine carried in most clinics, staff can easily become confused about vaccines within the storage unit. Labeling the area where vaccines are stored can
Vaccine Label Examples With the large amount of vaccine carried in most clinics, staff can easily become confused about vaccines within the storage unit. Labeling the area where vaccines are stored can
Vaccination Decision Making: What Providers Need to Know
 Objectives Vaccination Decision Making: What Providers Need to Know Kelli Smith, RN, BSN Iowa Department of Public Health Immunization Program Catch-up Schedule: how to most efficiently bring persons up-to-date
Objectives Vaccination Decision Making: What Providers Need to Know Kelli Smith, RN, BSN Iowa Department of Public Health Immunization Program Catch-up Schedule: how to most efficiently bring persons up-to-date
Hexavalent Vaccines: Hepatitis B antibody response and co-administration with other vaccines
 Viral Hepatitis Prevention Board Hanoi, 25 26 July 2018 Hexavalent Vaccines: Hepatitis B antibody response and co-administration with other vaccines Prof. Timo Vesikari Vaccine Research Center University
Viral Hepatitis Prevention Board Hanoi, 25 26 July 2018 Hexavalent Vaccines: Hepatitis B antibody response and co-administration with other vaccines Prof. Timo Vesikari Vaccine Research Center University
Detailed cost of vaccination/ Immunization in India (Birth to 10 years of age)
 Detailed cost of vaccination/ Immunization in India (Birth to 10 years of age) Glossary: IPV= Injectable Polio vaccine OPV= Oral polio vaccine (these are oral drops, which is also given in "Pulse Polio
Detailed cost of vaccination/ Immunization in India (Birth to 10 years of age) Glossary: IPV= Injectable Polio vaccine OPV= Oral polio vaccine (these are oral drops, which is also given in "Pulse Polio
I certify that the information provided on this form is correct and verifiable. Month Day Year
 Certificate of Immunization Status (CIS) DOH 48-0 January 05 Please print. See back for instructions on how to fill out this form or get it printed from the Immunization Information System. Child s Last
Certificate of Immunization Status (CIS) DOH 48-0 January 05 Please print. See back for instructions on how to fill out this form or get it printed from the Immunization Information System. Child s Last
Vaccinology 101 for Fellows
 Vaccinology 101 for Fellows Meg Fisher, MD Medical Director, The Children s Hospital Monmouth Medical Center An affiliate of the Saint Barnabas Health Care System Long Branch, NJ Disclosures I have no
Vaccinology 101 for Fellows Meg Fisher, MD Medical Director, The Children s Hospital Monmouth Medical Center An affiliate of the Saint Barnabas Health Care System Long Branch, NJ Disclosures I have no
Victorian immunisation catch-up tool for 10 to 19 year olds
 Victorian immunisation catch-up tool for 10 to 19 year olds For healthy children with no documented history of vaccine administration The objective of catch-up vaccination is to complete a course of age
Victorian immunisation catch-up tool for 10 to 19 year olds For healthy children with no documented history of vaccine administration The objective of catch-up vaccination is to complete a course of age
Coverage of Vaccines Medicaid and Child Health Plus Members
 Coverage of Vaccines Medicaid and Child Health Plus Members For children between the ages 0-18, routine recommended vaccinations are covered through Vaccines for Children program [VFC]. Fidelis Care will
Coverage of Vaccines Medicaid and Child Health Plus Members For children between the ages 0-18, routine recommended vaccinations are covered through Vaccines for Children program [VFC]. Fidelis Care will
Temperature Intelligence Solutions. Thermostability of Vaccines
 Thermostability of Vaccines Why are all vaccines sensitive to heat and some to freezing? Why is thermostability of vaccine important? What can be done to improve the thermostability of vaccines? Goals
Thermostability of Vaccines Why are all vaccines sensitive to heat and some to freezing? Why is thermostability of vaccine important? What can be done to improve the thermostability of vaccines? Goals
Modern Drug House.
 +91-8048941768 Modern Drug House https://www.moderndrughouse.in/ We are one of the leading suppliers and traders of a wide range of Vaccine and Pharmaceutical Products. Owing to its effectiveness, purity
+91-8048941768 Modern Drug House https://www.moderndrughouse.in/ We are one of the leading suppliers and traders of a wide range of Vaccine and Pharmaceutical Products. Owing to its effectiveness, purity
EARLY CHILDHOOD DEVELOPMENT & EDUCATION
 A division of Pawsitively Bassett, Inc. EARLY CHILDHOOD DEVELOPMENT & EDUCATION 08-09 67 N. Monroe St., Spokane WA 9908 www.nlcpreschool.com (509) 8-00 Dear Parents, Thank you, for your interest in the
A division of Pawsitively Bassett, Inc. EARLY CHILDHOOD DEVELOPMENT & EDUCATION 08-09 67 N. Monroe St., Spokane WA 9908 www.nlcpreschool.com (509) 8-00 Dear Parents, Thank you, for your interest in the
Syrian Programme Refugees Advice on assessment of immunisation status and recommendations for additional immunisation
 Syrian Programme Refugees Advice on assessment of immunisation status and recommendations for additional immunisation Background Information Immunisation services in Syria Syria had good immunisation services,
Syrian Programme Refugees Advice on assessment of immunisation status and recommendations for additional immunisation Background Information Immunisation services in Syria Syria had good immunisation services,
immunisation in New Zealand
 This appendix details the history of. Section A1.1 is a brief summary of when each vaccine was introduced to the National Immunisation Schedule (the Schedule). This summary includes vaccines which were
This appendix details the history of. Section A1.1 is a brief summary of when each vaccine was introduced to the National Immunisation Schedule (the Schedule). This summary includes vaccines which were
BabyJabs Vaccines. All vaccines are mercury- free We use aluminium- free vaccines wherever possible
 BabyJabs Vaccines All vaccines are mercury- free We use aluminium- free vaccines wherever possible BabyJabs is a dedicated children s immunisation service, offering a choice of single and small combination
BabyJabs Vaccines All vaccines are mercury- free We use aluminium- free vaccines wherever possible BabyJabs is a dedicated children s immunisation service, offering a choice of single and small combination
To demonstrate that DTPa-HBV-IPV/Hib-MenC-TT co-administered with 10Pn, is non-inferior to DTPa-HBV-IPV/Hib coadministered
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
All Kindergarteners and 4-6 year old transfer students. 4 doses DTP or DTaP 1 dose must be at or after 4 years of age. None
 Parents Vaccines Required for School Entry in Michigan Whenever children are brought into group settings, there is a chance for diseases to spread. Children must follow state vaccine laws in order to attend
Parents Vaccines Required for School Entry in Michigan Whenever children are brought into group settings, there is a chance for diseases to spread. Children must follow state vaccine laws in order to attend
Immunization Guidelines for the Use of State Supplied Vaccine April 18, 2013
 DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
BabyJabs Vaccines. All vaccines are mercury-free We use aluminium-free vaccines wherever possible
 BabyJabs Vaccines All vaccines are mercury-free We use aluminium-free vaccines wherever possible BabyJabs is a dedicated children s immunisation service, offering a choice of single and small combination
BabyJabs Vaccines All vaccines are mercury-free We use aluminium-free vaccines wherever possible BabyJabs is a dedicated children s immunisation service, offering a choice of single and small combination
For the use only of Registered Medical Practitioners or a Hospital or a Laboratory
 For the use only of Registered Medical Practitioners or a Hospital or a Laboratory INFANRIX HEXA Diphtheria, tetanus, pertussis (acellular component), hepatitis B (rdna), poliomyelitis (inactivated) and
For the use only of Registered Medical Practitioners or a Hospital or a Laboratory INFANRIX HEXA Diphtheria, tetanus, pertussis (acellular component), hepatitis B (rdna), poliomyelitis (inactivated) and
BabyJabs Vaccines. All vaccines are mercury-free We use aluminium-free vaccines wherever possible
 BabyJabs Vaccines All vaccines are mercury-free We use aluminium-free vaccines wherever possible BabyJabs is a dedicated children s immunisation service, offering a choice of single and small combination
BabyJabs Vaccines All vaccines are mercury-free We use aluminium-free vaccines wherever possible BabyJabs is a dedicated children s immunisation service, offering a choice of single and small combination
What DO the childhood immunization footnotes reveal? Questions and answers
 What DO the childhood immunization footnotes reveal? Questions and answers Stanley E. Grogg, DO, FACOP, FAAP he Advisory Committee on Immunization Practices (ACIP) recommends the childhood vaccination
What DO the childhood immunization footnotes reveal? Questions and answers Stanley E. Grogg, DO, FACOP, FAAP he Advisory Committee on Immunization Practices (ACIP) recommends the childhood vaccination
Objectives. Immunity. Childhood Immunization Risk of Non-Vaccinated Children 12/22/2015
 Childhood Immunization Risk of Non-Vaccinated Children Bertha P. Rojas, Pharm.D. PGY-1 Pharmacy Resident South Miami Hospital Objectives Understand the definition of herd immunity Identify vaccine-preventable
Childhood Immunization Risk of Non-Vaccinated Children Bertha P. Rojas, Pharm.D. PGY-1 Pharmacy Resident South Miami Hospital Objectives Understand the definition of herd immunity Identify vaccine-preventable
National Immunisation News
 July 2015 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Changes to the Primary Childhood Immunisation Programme BCG Shortage Pertussis Vaccine Mumps Order Vaccines
July 2015 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Changes to the Primary Childhood Immunisation Programme BCG Shortage Pertussis Vaccine Mumps Order Vaccines
Immunization Guidelines For the Use of State Supplied Vaccine July 1, 2011
 DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
Victorian immunisation catch-up tool for 10 to 19 year olds
 Victorian immunisation catch-up tool for 10 to 19 year olds January 2018 Guidelines for 10 to 19 year olds with no documented history of vaccine administration The objective of catch-up vaccination is
Victorian immunisation catch-up tool for 10 to 19 year olds January 2018 Guidelines for 10 to 19 year olds with no documented history of vaccine administration The objective of catch-up vaccination is
Vaccine ingredients. Fact sheet August 2017 page 1. Continued...
 Vaccine manufacture and composition is complex and tightly regulated to maximise safety. The safety of the individual components, and of the vaccine itself, must be demonstrated before a vaccine can be
Vaccine manufacture and composition is complex and tightly regulated to maximise safety. The safety of the individual components, and of the vaccine itself, must be demonstrated before a vaccine can be
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Infanrix hexa, Powder and suspension for suspension for injection. Diphtheria (D), tetanus (T), pertussis (acellular, component)
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Infanrix hexa, Powder and suspension for suspension for injection. Diphtheria (D), tetanus (T), pertussis (acellular, component)
Appendix An Assessment Tool to Determine the Validity of Vaccine Doses
 Appendix 4.4 - An Assessment Tool to Determine the Validity of Vaccine Doses Note: Refer to the Canadian Immunization Guide and New Brunswick (NB) immunization program directives for recommendations for
Appendix 4.4 - An Assessment Tool to Determine the Validity of Vaccine Doses Note: Refer to the Canadian Immunization Guide and New Brunswick (NB) immunization program directives for recommendations for
Immunise your child on time. It s their best protection
 Immunise your child on time It s their best protection If you are a parent or caregiver, this pamphlet is for you. It tells you about the recommended ages for your baby to receive their immunisations.
Immunise your child on time It s their best protection If you are a parent or caregiver, this pamphlet is for you. It tells you about the recommended ages for your baby to receive their immunisations.
A Review of Pediatric Vaccines Gary Overturf, M.D. Professor of Pediatrics, Inf. Dis. University of New Mexico
 Review of Pediatric and Adolescent Vaccines 2012 Idaho Society of Health- System Pharmacists Boise, ID April 28 th 2012 A Review of Pediatric Vaccines - 2012 Gary Overturf, M.D. Professor of Pediatrics,
Review of Pediatric and Adolescent Vaccines 2012 Idaho Society of Health- System Pharmacists Boise, ID April 28 th 2012 A Review of Pediatric Vaccines - 2012 Gary Overturf, M.D. Professor of Pediatrics,
NEW ZEALAND DATA SHEET
 1. PRODUCT NAME NEW ZEALAND DATA SHEET HIBERIX Haemophilus influenzae type b (Hib) powder and diluent for solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION After reconstitution, 1 dose
1. PRODUCT NAME NEW ZEALAND DATA SHEET HIBERIX Haemophilus influenzae type b (Hib) powder and diluent for solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION After reconstitution, 1 dose
National Immunisation News The newsletter of the HSE National Immunisation Office
 February 2015 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Changes to the PCI programme Uptake statistics Pneumovax name change Flu season Common queries School
February 2015 National Immunisation News The newsletter of the HSE National Immunisation Office CONTENTS Changes to the PCI programme Uptake statistics Pneumovax name change Flu season Common queries School
Vaccines and other immunological antimicrobial therapy 1
 Vaccines and other immunological antimicrobial therapy 1 Vaccines Vaccine: a biological preparation that provides active acquired immunity to a particular disease. Vaccine typically contains an agent that
Vaccines and other immunological antimicrobial therapy 1 Vaccines Vaccine: a biological preparation that provides active acquired immunity to a particular disease. Vaccine typically contains an agent that
CHILDHOOD VACCINATION
 EPI (3) Age of Child How and Where is it given? CHILDHOOD VACCINATION Nicolette du Plessis Block 10 28/02/2012 10 weeks DTaP-IPV/Hib (2) Diphtheria, Tetanus, Acellular pertussis, Inactivated polio vaccine,
EPI (3) Age of Child How and Where is it given? CHILDHOOD VACCINATION Nicolette du Plessis Block 10 28/02/2012 10 weeks DTaP-IPV/Hib (2) Diphtheria, Tetanus, Acellular pertussis, Inactivated polio vaccine,
Study No.: Title: Rationale: Phase: Study Period: Study Design Centers: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Childhood Immunisations Template Guide 2017
 Childhood Immunisations Template Guide 2017 1 Template Control Page Childhood Immunisations Template Guide Title Childhood Immunisations Author CEG Description Supports the schedule of Childhood Immunisations
Childhood Immunisations Template Guide 2017 1 Template Control Page Childhood Immunisations Template Guide Title Childhood Immunisations Author CEG Description Supports the schedule of Childhood Immunisations
Practical Applications of Immunology. Chapter 18
 Practical Applications of Immunology Chapter 18 I. Vaccines A. Definition A suspension of organisms or fractions of organisms that is used to induce immunity (immunologic memory). The mechanism of memory
Practical Applications of Immunology Chapter 18 I. Vaccines A. Definition A suspension of organisms or fractions of organisms that is used to induce immunity (immunologic memory). The mechanism of memory
National Immunisation News
 National Immunisation News The newsletter of the HSE National Immunisation Office July 2016 Changes to the Primary Childhood Immunisation Programme The National Immunisation Advisory Committee (NIAC) has
National Immunisation News The newsletter of the HSE National Immunisation Office July 2016 Changes to the Primary Childhood Immunisation Programme The National Immunisation Advisory Committee (NIAC) has
Manitoba Annual Immunization Surveillance Report
 Annual Immunization Surveillance Report January 1 to December 31, 2014 Epidemiology & Surveillance Public Branch Public and Primary Care Division, y Living and Seniors Released: January 2016 TABLE OF CONTENTS
Annual Immunization Surveillance Report January 1 to December 31, 2014 Epidemiology & Surveillance Public Branch Public and Primary Care Division, y Living and Seniors Released: January 2016 TABLE OF CONTENTS
Immunisation Policy. Country Children s Early Learning Ph: M:
 Immunisation Policy Published June 2018 Review Date December 2019 Sources Education and Care Services National Regulations, October 2017 Guide to the Education and Care Services National Law and the Education
Immunisation Policy Published June 2018 Review Date December 2019 Sources Education and Care Services National Regulations, October 2017 Guide to the Education and Care Services National Law and the Education
Childhood Immunisations Template Guide 2016
 Childhood Immunisations Template Guide 2016 1 Template Control Page Childhood Immunisations Template Guide Title Childhood Immunisations Author CEG Version 9 Description Supports the schedule of Childhood
Childhood Immunisations Template Guide 2016 1 Template Control Page Childhood Immunisations Template Guide Title Childhood Immunisations Author CEG Version 9 Description Supports the schedule of Childhood
3 rd dose. 3 rd or 4 th dose, see footnote 5. see footnote 13. for certain high-risk groups
 Figure 1. Recommended immunization schedule for persons aged 0 through 18 years 2013. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations must be read
Figure 1. Recommended immunization schedule for persons aged 0 through 18 years 2013. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations must be read
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Infanrix hexa, Powder and suspension for suspension for injection. Diphtheria (D), tetanus (T), pertussis (acellular, component)
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Infanrix hexa, Powder and suspension for suspension for injection. Diphtheria (D), tetanus (T), pertussis (acellular, component)
Early Learning Centre Immunisation Policy Legislation ACT Public Health Regulations (2000)
 Early Learning Centre Immunisation Policy Legislation ACT Public Health Regulations (2000) National Quality Standard / Education and Care Services National Regulations Standard 2.1 Each child s health
Early Learning Centre Immunisation Policy Legislation ACT Public Health Regulations (2000) National Quality Standard / Education and Care Services National Regulations Standard 2.1 Each child s health
Centers for Disease Control and Prevention. Final Revised Vaccine Information Materials for MMR
 This document is scheduled to be published in the Federal Register on 03/15/2018 and available online at https://federalregister.gov/d/2018-05299, and on FDsys.gov BILLING CODE: 4163-18-P DEPARTMENT OF
This document is scheduled to be published in the Federal Register on 03/15/2018 and available online at https://federalregister.gov/d/2018-05299, and on FDsys.gov BILLING CODE: 4163-18-P DEPARTMENT OF
Synflorix. Pneumococcal polysaccharide and Non-Typeable Haemophilus influenzae (NTHi) protein D conjugate vaccine, adsorbed
 Synflorix Pneumococcal polysaccharide and Non-Typeable Haemophilus influenzae (NTHi) protein D conjugate vaccine, adsorbed QUALITATIVE AND QUANTITATIVE COMPOSITION One dose (0.5 ml) contains 1 microgram
Synflorix Pneumococcal polysaccharide and Non-Typeable Haemophilus influenzae (NTHi) protein D conjugate vaccine, adsorbed QUALITATIVE AND QUANTITATIVE COMPOSITION One dose (0.5 ml) contains 1 microgram
Armed Services Blood Program Immunization List
 Immunization List NOTICE: The Department of Defense (DoD) assumes no risk for the use of this information by non-dod personnel, blood programs, or individual medical institutions. The use of this information
Immunization List NOTICE: The Department of Defense (DoD) assumes no risk for the use of this information by non-dod personnel, blood programs, or individual medical institutions. The use of this information
14: Immunological products and vaccines
 File name: Immunological Products and s Formulary BNF Original Date of issue: 30/10/2005 Last Reviewed: 23/11/2012 Version:4 Page 1 of 6 14: Immunological products and vaccines 14.1 Active immunity Live
File name: Immunological Products and s Formulary BNF Original Date of issue: 30/10/2005 Last Reviewed: 23/11/2012 Version:4 Page 1 of 6 14: Immunological products and vaccines 14.1 Active immunity Live
Immunization Guidelines for the Use of State Supplied Vaccine May 17, 2015
 DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
DTaP / DT DTaP/IPV/Hep B Combination (Pediarix ) Children from 6 weeks of age up to the 7 th birthday Children from 2 months of age up to the 7th birthday: Indicated for the primary doses of DTaP, IPV,
7.0 Nunavut Childhood and Adult Immunization Schedules and Catch-up Aids
 7.0 Nunavut Childhood and Adult Immunization Schedules and Catch-up Aids Contents Introduction Nunavut Recommended Childhood Immunization Schedule Nunavut Routine Adult Immunization Schedule Nunavut Immunization
7.0 Nunavut Childhood and Adult Immunization Schedules and Catch-up Aids Contents Introduction Nunavut Recommended Childhood Immunization Schedule Nunavut Routine Adult Immunization Schedule Nunavut Immunization
IMPORTANT: PLEASE READ
 PART III: CONSUMER INFORMATION combined hepatitis A (inactivated) and hepatitis B (recombinant) vaccine This leaflet is part III of a three-part "Product Monograph" published when was approved for sale
PART III: CONSUMER INFORMATION combined hepatitis A (inactivated) and hepatitis B (recombinant) vaccine This leaflet is part III of a three-part "Product Monograph" published when was approved for sale
For the use only of Registered Medical Practitioners or a Hospital or a Laboratory HIBERIX
 For the use only of Registered Medical Practitioners or a Hospital or a Laboratory HIBERIX Haemophilus type b conjugated vaccine IP 1. NAME OF THE MEDICINAL PRODUCT Haemophilus type b conjugated vaccine
For the use only of Registered Medical Practitioners or a Hospital or a Laboratory HIBERIX Haemophilus type b conjugated vaccine IP 1. NAME OF THE MEDICINAL PRODUCT Haemophilus type b conjugated vaccine
Vaccines, Not Just for Babies
 Vaccines, Not Just for Babies Meg Fisher, MD Medical Director Disclosures I have no relevant financial relationships with the manufacturers of any commercial products or commercial services discussed in
Vaccines, Not Just for Babies Meg Fisher, MD Medical Director Disclosures I have no relevant financial relationships with the manufacturers of any commercial products or commercial services discussed in
Diphtheria-Tetanus-Acellular Pertussis-Polio- Haemophilus influenzae type b Conjugate Combined Vaccine Biological Page (DTaP-IPV-Hib)
 Diphtheria-Tetanus-Acellular Pertussis-Polio- Haemophilus influenzae type b Conjugate Combined Vaccine Biological Page (DTaP-IPV-Hib) Section 7: Biological Product Information Standard #: 07.211 Created
Diphtheria-Tetanus-Acellular Pertussis-Polio- Haemophilus influenzae type b Conjugate Combined Vaccine Biological Page (DTaP-IPV-Hib) Section 7: Biological Product Information Standard #: 07.211 Created
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Infanrix hexa, Powder and suspension for suspension for injection. Diphtheria (D), tetanus (T), pertussis (acellular, component)
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Infanrix hexa, Powder and suspension for suspension for injection. Diphtheria (D), tetanus (T), pertussis (acellular, component)
Enhanced immunisation schedule Victoria
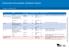 Children from March 2016 Age Disease Vaccine brand Birth Hepatitis B H-B-Vax-II Paediatric 2 months - from 6 weeks Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Reconstitute Site given Route
Children from March 2016 Age Disease Vaccine brand Birth Hepatitis B H-B-Vax-II Paediatric 2 months - from 6 weeks Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Reconstitute Site given Route
Annex 1. WHO Recommendations, Guidelines and other documents related to the manufacture and quality control of biological substances used in medicine
 WHO related to the manufacture and quality control of biological substances used in medicine WHO are intended to provide guidance to those responsible for the production of biological substances as well
WHO related to the manufacture and quality control of biological substances used in medicine WHO are intended to provide guidance to those responsible for the production of biological substances as well
SKAGIT VALLEY FAMILY YMCA
 Central Preschool Registration 2019-2020 Child s First : MI: Last : Address: City: State: Zip Home Phone: ( ) Gender: Birth : Age: Child lives w/ Mother/Guardian: Cell Phone: ( ) Employer: Work Phone:
Central Preschool Registration 2019-2020 Child s First : MI: Last : Address: City: State: Zip Home Phone: ( ) Gender: Birth : Age: Child lives w/ Mother/Guardian: Cell Phone: ( ) Employer: Work Phone:
CPT 2016 Code Changes
 CPT 2016 Code Changes Code Changes - Medicine New CPT 2016 New Codes Code Description 69209 Removal impacted cerumen using irrigation/lavage, unilateral 90620 Meningococcal recombinant protein and outer
CPT 2016 Code Changes Code Changes - Medicine New CPT 2016 New Codes Code Description 69209 Removal impacted cerumen using irrigation/lavage, unilateral 90620 Meningococcal recombinant protein and outer
Immunisation Subcommittee of PTAC Meeting held 28 October 2015
 Immunisation Subcommittee of PTAC Meeting held 28 October 2015 (minutes for web publishing) Immunisation Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology
Immunisation Subcommittee of PTAC Meeting held 28 October 2015 (minutes for web publishing) Immunisation Subcommittee minutes are published in accordance with the Terms of Reference for the Pharmacology
Child Care Registration Form
 Child Care Registration Form child entered care Child s name Last First Middle Name (Nickname) used Birthdate Street address City Zip code Child s parent/guardian name home phone # cell phone# child left
Child Care Registration Form child entered care Child s name Last First Middle Name (Nickname) used Birthdate Street address City Zip code Child s parent/guardian name home phone # cell phone# child left
Yukon Immunization Program Manual ADMENDMENTS & ADDITIONS
 2012 August Page 1 Manual ADMENDMENTS & ADDITIONS Number & 11-01 2011 June 09 11-02 2011 October 11 12-01 2012 Feb 9 Topic Additions to Yukon Immunization Program Manual Twinrix, Twinrix Junior, Menamune,
2012 August Page 1 Manual ADMENDMENTS & ADDITIONS Number & 11-01 2011 June 09 11-02 2011 October 11 12-01 2012 Feb 9 Topic Additions to Yukon Immunization Program Manual Twinrix, Twinrix Junior, Menamune,
Centers for Disease Control and Prevention. Proposed Revised Vaccine Information Materials for MMR
 This document is scheduled to be published in the Federal Register on 10/18/2016 and available online at https://federalregister.gov/d/2016-25144, and on FDsys.gov BILLING CODE: 4163-18-P DEPARTMENT OF
This document is scheduled to be published in the Federal Register on 10/18/2016 and available online at https://federalregister.gov/d/2016-25144, and on FDsys.gov BILLING CODE: 4163-18-P DEPARTMENT OF
Immunisation schedule Victoria
 Infants and children July 2018 Birth H-B-Vax-II Paediatric or Paediatric 2 months (from 6 weeks) and 4 months Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Haemophilus influenzae type b Anterolateral
Infants and children July 2018 Birth H-B-Vax-II Paediatric or Paediatric 2 months (from 6 weeks) and 4 months Diphtheria-tetanus-pertussis, poliomyelitis-hepatitis B- Haemophilus influenzae type b Anterolateral
NOTE: The above recommendations must be read along with the footnotes of this schedule.
 Figure 1. Recommended immunization schedule for persons aged 0 through 18 years 2013. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations must be read
Figure 1. Recommended immunization schedule for persons aged 0 through 18 years 2013. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations must be read
State of Alaska Child Care & School Immunization Requirements Packet Revised 08/2010
 State of Alaska Child Care & School Immunization Requirements Packet Revised 08/2010 ALASKA IMMUNIZATION PROGRAM (907) 269-8000 OR 1-888-430-4321 http://www.epi.hss.state.ak.us/id/immune.stm (This page
State of Alaska Child Care & School Immunization Requirements Packet Revised 08/2010 ALASKA IMMUNIZATION PROGRAM (907) 269-8000 OR 1-888-430-4321 http://www.epi.hss.state.ak.us/id/immune.stm (This page
Child Health and Disability Prevention (CHDP) Program Code Conversion
 Child Health and Disability Prevention (CHDP) Program Code Conversion Health s All s are effective for dates of service on or after July 1, 2017. (Select s for laboratory-only providers with effective
Child Health and Disability Prevention (CHDP) Program Code Conversion Health s All s are effective for dates of service on or after July 1, 2017. (Select s for laboratory-only providers with effective
- Infanrix hexa (DTPa-HBV-IPV/Hib): GSK Biologicals combined diphtheria, tetanus, acellular pertussis, hepatitis
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
PRODUCT MONOGRAPH NIMENRIX. Meningococcal polysaccharide groups A, C, W-135 and Y conjugate vaccine. Powder and diluent for solution for injection
 PRODUCT MONOGRAPH Meningococcal polysaccharide groups A, C, W-135 and Y conjugate vaccine Powder and diluent for solution for injection Active Immunizing Agent Pfizer Canada Inc. 17,300 Trans-Canada Highway
PRODUCT MONOGRAPH Meningococcal polysaccharide groups A, C, W-135 and Y conjugate vaccine Powder and diluent for solution for injection Active Immunizing Agent Pfizer Canada Inc. 17,300 Trans-Canada Highway
For the use only of a Registered Medical Practitioner or a Hospital or a Laboratory SYNFLORIX
 For the use only of a Registered Medical Practitioner or a Hospital or a Laboratory SYNFLORIX Pneumococcal Polysaccharide and Non-Typeable Haemophilus influenzae (NTHi) Protein D Conjugate Vaccine, adsorbed
For the use only of a Registered Medical Practitioner or a Hospital or a Laboratory SYNFLORIX Pneumococcal Polysaccharide and Non-Typeable Haemophilus influenzae (NTHi) Protein D Conjugate Vaccine, adsorbed
The Scientific Relevance of the ATT Today and from a historical Perspective
 www.pei.de The Scientific Relevance of the ATT Today and from a historical Perspective Dr. Klaus Cussler EPAA International Workshop Towards global harmonization of 3Rs in biologicals 15-16 September 2015
www.pei.de The Scientific Relevance of the ATT Today and from a historical Perspective Dr. Klaus Cussler EPAA International Workshop Towards global harmonization of 3Rs in biologicals 15-16 September 2015
PATH Quick Reference Guide: Coding for Pediatric Health HEDIS Measures
 PATH Quick Reference Guide: Coding for Pediatric Health HEDIS Measures This guide is designed to be a quick reference tool to help with medical coding of select Healthcare Effectiveness Data and Information
PATH Quick Reference Guide: Coding for Pediatric Health HEDIS Measures This guide is designed to be a quick reference tool to help with medical coding of select Healthcare Effectiveness Data and Information
Table of Contents. 1.0 INFANRIX hexa VACCINE...3
 Table of Contents 1.0 INFANRIX hexa VACCINE...3 1.1 What is INFANRIX hexa vaccine?...3 1.2 When will INFANRIX hexa be introduced?...3 1.3 What is the difference between INFANRIX hexa and Pediacel?...3
Table of Contents 1.0 INFANRIX hexa VACCINE...3 1.1 What is INFANRIX hexa vaccine?...3 1.2 When will INFANRIX hexa be introduced?...3 1.3 What is the difference between INFANRIX hexa and Pediacel?...3
Pentabio Vaccine (DTP-HB-Hib)
 SUMMARY OF PRODUCT CHARACTERISTICS Product Name Pharmaceutical Form Strength Presentation : Pentabio : Suspension for injection : 1, 5 and 10 doses : Box of 10 vials @ 0.5 ml Box of 10 vials @ 2.5 ml Box
SUMMARY OF PRODUCT CHARACTERISTICS Product Name Pharmaceutical Form Strength Presentation : Pentabio : Suspension for injection : 1, 5 and 10 doses : Box of 10 vials @ 0.5 ml Box of 10 vials @ 2.5 ml Box
1 Principles of Vaccination Immunology and Vaccine-Preventable Diseases... 1 Classification of Vaccines... 4 Selected References...
 1 Principles of Vaccination Immunology and Vaccine-Preventable Diseases... 1 Classification of Vaccines... 4 Selected References... 7 2 General Recommendations on Immunization Timing and Spacing of Vaccines...
1 Principles of Vaccination Immunology and Vaccine-Preventable Diseases... 1 Classification of Vaccines... 4 Selected References... 7 2 General Recommendations on Immunization Timing and Spacing of Vaccines...
Immunizations are among the most cost effective and widely used public health interventions.
 Focused Issue of This Month Recommended by the Korean Pediatric Society, 2008 Hoan Jong Lee, MD Department of Pediatrics, Seoul National University College of Medicine E mail : hoanlee@snu.ac.kr J Korean
Focused Issue of This Month Recommended by the Korean Pediatric Society, 2008 Hoan Jong Lee, MD Department of Pediatrics, Seoul National University College of Medicine E mail : hoanlee@snu.ac.kr J Korean
NOTE: The above recommendations must be read along with the footnotes of this schedule.
 Figure 1. Recommended immunization schedule for persons aged 0 through 18 years United States, 2014. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations
Figure 1. Recommended immunization schedule for persons aged 0 through 18 years United States, 2014. (FOR THOSE WHO FALL BEHIND OR START LATE, SEE THE CATCH-UP SCHEDULE [FIGURE 2]). These recommendations
Prequalification Team WHO PUBLIC INSPECTION REPORT Vaccine Manufacturer
 20, avenue Appia CH-1211 Geneva 27 Switzerland Tel central +41 22 791 2111 Fax central +41 22 791 3111 www.who.int Part 1: General information Name of Manufacturer Prequalification Team WHO PUBLIC INSPECTION
20, avenue Appia CH-1211 Geneva 27 Switzerland Tel central +41 22 791 2111 Fax central +41 22 791 3111 www.who.int Part 1: General information Name of Manufacturer Prequalification Team WHO PUBLIC INSPECTION
NEW ZEALAND DATA SHEET
 NEW ZEALAND DATA SHEET Name of the Medicinal Product MENITORIX Haemophilus type b and Neisseria meningitidis group C conjugate vaccine Presentation MENITORIX is presented as a powder and diluent for reconstitution
NEW ZEALAND DATA SHEET Name of the Medicinal Product MENITORIX Haemophilus type b and Neisseria meningitidis group C conjugate vaccine Presentation MENITORIX is presented as a powder and diluent for reconstitution
DATA SHEET PRIORIX-TETRA. Name of Medicinal Product
 DATA SHEET Name of Medicinal Product PRIORIX-TETRA Live attenuated measles, mumps, rubella and varicella vaccine Presentation PRIORIX-TETRA is a sterile lyophilised mixed preparation of the attenuated
DATA SHEET Name of Medicinal Product PRIORIX-TETRA Live attenuated measles, mumps, rubella and varicella vaccine Presentation PRIORIX-TETRA is a sterile lyophilised mixed preparation of the attenuated
K 6 th Express Billing Contract
 2017-2018 K 6 th Express Billing Contract Child Information: First Middle Initial Last Parent/Guardian Information: First Middle Initial Last Address Phone Requested Start Will you be receiving state/other
2017-2018 K 6 th Express Billing Contract Child Information: First Middle Initial Last Parent/Guardian Information: First Middle Initial Last Address Phone Requested Start Will you be receiving state/other
Benefit Interpretation
 Benefit Interpretation Subject: Part B vs. Part D Vaccines Issue Number: BI-039 Applies to: Medicare Advantage Effective Date: May 1, 2017 Attachments: Part B Vaccines Diagnosis Code Limits Table of Contents
Benefit Interpretation Subject: Part B vs. Part D Vaccines Issue Number: BI-039 Applies to: Medicare Advantage Effective Date: May 1, 2017 Attachments: Part B Vaccines Diagnosis Code Limits Table of Contents
K 6 th Express Billing Contract
 05-06 K 6 th Express Billing Contract Child Information: First Middle Initial Last Parent/Guardian Information: First Middle Initial Last Address Phone Requested Start Will you be receiving state/other
05-06 K 6 th Express Billing Contract Child Information: First Middle Initial Last Parent/Guardian Information: First Middle Initial Last Address Phone Requested Start Will you be receiving state/other
NEW ZEALAND DATA SHEET
 1. PRODUCT NAME NEW ZEALAND DATA SHEET INFANRIX HEXA Combined diphtheria-tetanus-acellular pertussis, hepatitis B, enhanced inactivated polio vaccine and Haemophilus influenzae type b powder and suspension
1. PRODUCT NAME NEW ZEALAND DATA SHEET INFANRIX HEXA Combined diphtheria-tetanus-acellular pertussis, hepatitis B, enhanced inactivated polio vaccine and Haemophilus influenzae type b powder and suspension
Use of Infanrix -IPV+Hib in the infant primary immunisation schedule
 Use of Infanrix -IPV+Hib in the infant primary immunisation schedule An update for registered healthcare practitioners July 2014 Quality Education for a Healthier Scotland 1 Acknowledgments Many thanks
Use of Infanrix -IPV+Hib in the infant primary immunisation schedule An update for registered healthcare practitioners July 2014 Quality Education for a Healthier Scotland 1 Acknowledgments Many thanks
PHARMAC invites proposals for the supply of various vaccines in New Zealand.
 15 February 2016 Dear Supplier REQUEST FOR PROPOSALS SUPPLY OF VARIOUS VACCINES PHARMAC invites proposals for the supply of various vaccines in New Zealand. This request for proposals (RFP) letter incorporates
15 February 2016 Dear Supplier REQUEST FOR PROPOSALS SUPPLY OF VARIOUS VACCINES PHARMAC invites proposals for the supply of various vaccines in New Zealand. This request for proposals (RFP) letter incorporates
