Single Use Curlew TM Multiple Biopsy Forceps
|
|
|
- Arabella Wilkins
- 5 years ago
- Views:
Transcription
1 Single Use Curlew TM Multiple Biopsy Forceps 13 SPECIMEN WITH METAL STORAGE CYLINDER With In Situ Fixation, Batch or Single Specimen Collection US Patents 5,782,747; 5,980,468; 6,071,248; foreign patents and pending INSTRUCTIONS FOR USE Read this document in its entirety prior to use Rx Only Caution: Federal (USA) Law restricts this device to sale by or on the order of a physician Warning: This Device is Sharp! Use same care as for any other surgical device to avoid punctures or cuts to hands, gloves, etc. Caution: A thorough understanding of the technical principles, clinical application and risks associated with multiple biopsy is necessary before using this instrument Indications: Biopsy forceps are intended for use in collecting tissue samples for histological examination. Page 1 of 8
2 Contraindications: None known within the scope of the indicated use. Warning: For single use only. Do not reuse, reprocess or sterilize. Reuse, reprocessing or re-sterilization may compromise the structural integrity of the device and/ or lead to device failure which, in turn, may result in patient injury, illness or death. Inspection: Warning: Inspect the package containing the forceps. If any damage is noticed, do not use the instrument as sterility may be compromised. Peel open the pouch and remove the forceps. Visually inspect the forceps for kinks, loose or broken parts. With forceps coiled in 8 inch loops: (2) for 230cm; (1) for 160cm; (1) for 110cm, verify forceps operate freely with no hesitation, and smoothly with no grinding, friction or clicking. Warning: Do not use if any abnormalities are found or if the instrument is not operating properly. Device Overview The Curlew Multiple Biopsy forceps collects and stores up to 13 biopsies 2.0mm in diameter in a separable perforated metal storage cylinder for in situ fixation and transport to pathology with minimum handling and staff exposure to human material, sharps and solvents. In situ fixation is fixation of the entire 13 specimen biopsy series within the metal storage cylinder. This minimizes the work, exposure to sharps and human material previously required to separate the biopsies from the biopsy device in endoscopy. The metal storage cylinder maintains, collects and orients stored specimens in acquisition order as documented in the Curlew Multiple Biopsy Endoscopy Log (Attached to last page of the Pathology Laboratory Instructions) through histopathological processing with minimum handling in pathology. Multiple sections are mounted on slides in acquisition order for reading by the pathologist. Curlew Multiple Biopsy forceps requires a minimum 3.2mm endoscope channel. For biopsy the jaws are pushed out of the storage cylinder and inserted into the tissue to surround the specimen. During jaw closure and retraction, biopsies are cut, captured, oriented and collected in the storage cylinder in acquisition order. This action: (1) retains specimens in the storage cylinder Page 2 of 8
3 during and after endoscope withdrawal, during in situ fixation and transport to pathology; (2) protects staff during multiple biopsy use; (3) prevents endoscope instrument channel damage during passage and withdrawal. The Curlew label on the pouch and fixative vial indicate the maximum number of specimens collected in a single endoscopic pass and held in the storage cylinder for in situ fixation. The Curlew Multiple Biopsy Endoscopy Log (Attached to last page of the Pathology Laboratory Instructions) completed by the endoscopist identifies the number of specimens, biopsy sites, acquisition order and indications to the pathologist. When in situ fixation is not used, numerous multiple biopsy passes are supported. Operation: For hot multiple biopsy forceps; connect an Olympus compatible active cord to the instrument and ensure the connection is tight. Inspect the active cord for kinks, bends, breaks and exposed wires. If any abnormality is found, do not use the cord. The cord should snap onto the handle. If the cord is loose, call your sales representative who can provide you with the proper cord. Connect the other end of the cord to the electrosurgical generator and follow the manufacturer s instructions regarding operation and proper grounding. NOTE: IT IS IMPORTANT TO USE AN OLYMPUS COMPATIBLE PLUG. OTHER PLUG CONFIGURATIONS CAN LEAD TO FAULTY OPERATION. Warning: Connection to equipment not complying with EN could compromise patient safety. Prepare the Curlew Multiple Biopsy Endoscopy Log (Attached to last page of the Pathology Laboratory Instructions) to record the site, sequence and indication of each biopsy. Extend the biopsy jaws by gently pulling back on the thumb loop and retract by advancing the thumb loop to the stop to confirm easy biopsy action. Page 3 of 8
4 Do not open and close the jaws without straightening the shaft as that may alter the precise biopsy action. Observe that the jaws retract fully into the biopsy storage cylinder when the thumb loop is advanced to the handle stop. Full jaw retraction ensures that the endoscope instrument channel is not damaged during passage or withdrawal and that the biopsy specimens are retained in the storage cylinder during withdrawal from the endoscope. Close the forceps jaws before inserting the instrument into a 3.2mm endoscope channel. Use short motions when inserting the forceps to avoid kinking. If the scope is equipped with an elevator, lower the elevator fully before loading. If resistance is met when the bending tip of the forceps is angulated, release the bending angle slightly until the instrument passes smoothly. Do not force the forceps into an angulated scope as this could damage the endoscope. To take a biopsy, place the instrument tip close to the desired biopsy site and gently retract the thumb loop to extend the jaws while visualizing the procedure through the scope. Advance the open jaws into the tissue to surround the biopsy site. Close the jaws by gently advancing the thumb loop to the handle stop. The closing jaws cut, capture, orient and store the specimen in the storage cylinder, in the order of acquisition. Inspect the biopsy site for the bleeding mucosal defect indicating biospy capture. Capture failure is caused by not inserting the open jaws into the tissue as with a biopsy pin. Note the biopsy site, sequence and indication on Page 4 of 8
5 the Curlew Multiple Biopsy Endoscopy Log (Attached to last page of the Pathology Laboratory Instructions). Repeat until up to 13 biopsies, or the desired number of biopsies, are obtained. Caution: Do not use excessive force to close the jaws as this could damage the instrument. When the biopsy sequence is complete, fully retract the jaws into the biopsy storage cylinder and slowly withdraw the instrument from the scope while keeping the jaws closed. If the scope has a forceps elevator, it must be lowered before withdrawing the forceps. If the jaws will not close completely, do not force the forceps out of the endoscope. Remove the endoscope with the forceps still in place and then manually close the jaws before removing the instrument from the scope. Retrieval and Fixation of Batched or Individual Multiple Biopsy Specimens: After removing the biopsy instrument from the endoscope, keep the jaws closed inside the metal storage cylinder by pressing the thumb loop against the handle stop. This prevents loss of the specimens and protects staff from contact with sharps, jaws and potentially infectious tissue or fluid. With storage cylinder over the fixative vial, retract the thumb loop to open the storage cylinder. Cut the black cable ½ inch from the metal storage cylinder with a heavy wire cutter. (Do not cut or damage the metal cylinder as this may prevent specimen removal in pathology.) Place the metal storage cylinder in labeled fixative vial for transport to pathology. Affix a matching label to the Curlew Multiple Biopsy Endoscopy Log (Attached to last page of the Pathology Laboratory Instructions) for transport to pathology. Page 5 of 8
6 Batch Collection of Specimens without in situ fixation: (Irrigation of specimen batches from the storage cylinder into fixative) Prepare a blunt 20G needle on a 3ml syringe filled with fixative. Remove Multiple Biopsy forceps device from the endoscope and hold the storage tip over the fixative vial. Retract the thumb loop to open the storage cylinder. Identify an open proximal storage cylinder hole (not blocked by forceps mechanism). Insert irrigation needle in an open proximal storage cylinder hole. Irrigate specimens into fixative. Examine the storage cylinder to ensure all specimens have been removed. Repeat if specimens remain. Single Specimen Collection without in situ fixation: Each biopsy specimen is held within the biopsy jaws until pushed into the storage cylinder by the subsequent biopsy. Individual specimens may be collected from the sharp biopsy jaws by removing the Multiple Biopsy forceps from the endoscope, opening the jaws and placing the specimen in fixative. Disposal: Page 6 of 8
7 Warning: After use, this product may be a potential biohazard. Handle or dispose of it in accordance with accepted medical practice and applicable local, state and federal laws and regulations. Storage: Store sterile instruments at room temperature. Do not expose to solvents, radiation, UV light or any other substance or environment that could compromise the sterility of the unit. Warranty: Endo-Therapeutics Inc. warrants that the instrument will be free from defects in materials and workmanship for use in a single procedure. This warranty does not apply to use beyond the expiration date. This device is not intended to be reprocessed. The user assumes responsibility for any consequences related to the use of reprocessed instruments. Endo- Therapeutics obligations under this warranty are limited to the replacement of the instrument. Page 7 of 8
8 Symbols used on labeling Consult Instruction for Use Warning - Indicates a hazardous situation which, if not avoided, could result in death or serious injury. Caution - Indicates a hazardous situation which, if not avoided, could result in minor or moderate injury. Single use Do not reuse Ethylene Oxide Sterilization Catalog Number / Product Code Lot Number Use by Date Rx Only Federal (USA) Law restricts this device to sale by or on the order of a physician Authorized Representative Manufacturer Page 8 of 8
Initial placement 24FR Pull PEG kit REORDER NO:
 Initial placement 24FR Pull PEG kit REORDER NO: 00710805 INSTRUCTIONS FOR USE 1 of 5 These products have been manufactured not to include latex. Intended Use: The Initial placement 24FR Pull PEG kit is
Initial placement 24FR Pull PEG kit REORDER NO: 00710805 INSTRUCTIONS FOR USE 1 of 5 These products have been manufactured not to include latex. Intended Use: The Initial placement 24FR Pull PEG kit is
Initial placement 20FR Guidewire PEG kit REORDER NO:
 Initial placement 20FR Guidewire PEG kit REORDER NO: 00710802 INSTRUCTIONS FOR USE 1 of 5 These products have been manufactured not to include latex. Intended Use: The Initial placement 20FR Guidewire
Initial placement 20FR Guidewire PEG kit REORDER NO: 00710802 INSTRUCTIONS FOR USE 1 of 5 These products have been manufactured not to include latex. Intended Use: The Initial placement 20FR Guidewire
St. Jude Medical 8-Channel Adapter. Clinician's Manual
 St. Jude Medical 8-Channel Adapter Clinician's Manual CAUTION: Federal (USA) law restricts this device to sale by or on the order of a physician. For U.S. California Only: Proposition 65, a State of California
St. Jude Medical 8-Channel Adapter Clinician's Manual CAUTION: Federal (USA) law restricts this device to sale by or on the order of a physician. For U.S. California Only: Proposition 65, a State of California
CLARIVEIN INFUSION CATHETER
 CLARIVEIN INFUSION CATHETER General Product Description Overview The ClariVein Infusion Catheter (ClariVein -IC) is an infusion catheter system designed to introduce physician-specified medicaments into
CLARIVEIN INFUSION CATHETER General Product Description Overview The ClariVein Infusion Catheter (ClariVein -IC) is an infusion catheter system designed to introduce physician-specified medicaments into
Stratis INSTRUCTIONS FOR USE. Needle-free Injection System. 0.5mL volume (+/-5%)
 Stratis Needle-free Injection System INSTRUCTIONS FOR USE 0.5mL volume (+/-5%) Stratis English Symbols Glossary (Note: All symbols are derived from ISO 15223-1, Medical Devices - Symbols to be used with
Stratis Needle-free Injection System INSTRUCTIONS FOR USE 0.5mL volume (+/-5%) Stratis English Symbols Glossary (Note: All symbols are derived from ISO 15223-1, Medical Devices - Symbols to be used with
Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter
 Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter Caution: Federal (USA) law restricts this device to sale by or on the order of a physician. DEVICE DESCRIPTION
Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter Caution: Federal (USA) law restricts this device to sale by or on the order of a physician. DEVICE DESCRIPTION
AXS Catalyst Distal Access Catheter
 AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
CAREFULLY READ ALL INSTRUCTIONS PRIOR TO USE
 CAREFULLY READ ALL INSTRUCTIONS PRIOR TO USE INDICATIONS FOR USE The LATERA Absorbable Nasal Implant is indicated for supporting upper and lower lateral nasal cartilage. CAUTION: Federal law restricts
CAREFULLY READ ALL INSTRUCTIONS PRIOR TO USE INDICATIONS FOR USE The LATERA Absorbable Nasal Implant is indicated for supporting upper and lower lateral nasal cartilage. CAUTION: Federal law restricts
Advisor FL Circular Mapping Catheter, Sensor Enabled D-AVSE-D10-F20, D-AVSE-DF10-F20, D-AVSE-D10-F15, D-AVSE-DF10-F15. Instructions for Use
 Advisor FL Circular Mapping Catheter, Sensor Enabled D-AVSE-D10-F20, D-AVSE-DF10-F20, D-AVSE-D10-F15, D-AVSE-DF10-F15 Instructions for Use For U.S. California Only: Proposition 65, a State of California
Advisor FL Circular Mapping Catheter, Sensor Enabled D-AVSE-D10-F20, D-AVSE-DF10-F20, D-AVSE-D10-F15, D-AVSE-DF10-F15 Instructions for Use For U.S. California Only: Proposition 65, a State of California
Device Preparation (all steps to be performed per standard interventional technique)
 sertera Biopsy Device Instructions for Use Please read all information carefully. Failure to properly follow the instructions may lead to unintended consequences. Important: This package insert is designed
sertera Biopsy Device Instructions for Use Please read all information carefully. Failure to properly follow the instructions may lead to unintended consequences. Important: This package insert is designed
Endorectal Balloon (ERB) Endorectal Balloon (ERB) Instructions for Use
 Endorectal Balloon (ERB) Instructions for Use GRF-0004-00 Rev. 08 Page 1 of 9 Contents Instructions for Use... 3 Indications for Use... 3 Contraindications... 3 Storage... 4 Warnings... 4 Accessories Provided...
Endorectal Balloon (ERB) Instructions for Use GRF-0004-00 Rev. 08 Page 1 of 9 Contents Instructions for Use... 3 Indications for Use... 3 Contraindications... 3 Storage... 4 Warnings... 4 Accessories Provided...
Radux StandTall Instructions for Use Sheath Extender and Securement Clasp
 Radux StandTall Instructions for Use Sheath Extender and Securement Clasp USA Caution Federal (USA) law restricts this device to sale by or on the order of a health care professional. Caution The Radux
Radux StandTall Instructions for Use Sheath Extender and Securement Clasp USA Caution Federal (USA) law restricts this device to sale by or on the order of a health care professional. Caution The Radux
RD180 SP TECHNOLOGY GUIDE READ THIS PRODUCT INSERT THOROUGHLY BEFORE USE
 RD180 SP TECHNOLOGY GUIDE READ THIS PRODUCT INSERT THOROUGHLY BEFORE USE FIG. 1 RD180 SP - THE RUNNING DEVICE 1 2 6 3 7 4 5 1 NEEDLE 2 FERRULE 3 SUTURE 4 DEVICE TIP 5 ANGLED SHAFT 6 HANDLE 7 PINK LEVER
RD180 SP TECHNOLOGY GUIDE READ THIS PRODUCT INSERT THOROUGHLY BEFORE USE FIG. 1 RD180 SP - THE RUNNING DEVICE 1 2 6 3 7 4 5 1 NEEDLE 2 FERRULE 3 SUTURE 4 DEVICE TIP 5 ANGLED SHAFT 6 HANDLE 7 PINK LEVER
Instructions for Use Reprocessed Arthroscopic Shavers
 Reprocessed by Instructions for Use Reprocessed Arthroscopic Shavers Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a physician. STERILE
Reprocessed by Instructions for Use Reprocessed Arthroscopic Shavers Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a physician. STERILE
Directions For Use. All directions should be read before use
 Directions For Use All directions should be read before use DEVICE DESCRIPTION: The CLEANER.XT Rotational Thrombectomy System is a percutaneous, 6Fr catheter based system (single piece construction) that
Directions For Use All directions should be read before use DEVICE DESCRIPTION: The CLEANER.XT Rotational Thrombectomy System is a percutaneous, 6Fr catheter based system (single piece construction) that
Instructions for Use Reprocessed ENDOPATH XCEL Bladeless Endoscopic Trocars and Cannulas. Reprocessed Device for Single Use.
 Reprocessed by Instructions for Use Reprocessed ENDOPATH XCEL Bladeless Endoscopic Trocars and Cannulas Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by
Reprocessed by Instructions for Use Reprocessed ENDOPATH XCEL Bladeless Endoscopic Trocars and Cannulas Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by
ER REBOA Catheter. Instructions for Use
 ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
1 Description. 2 Indications. 3 Warnings ASPIRATION CATHETER
 Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
The Ceterix NovoStitch Disposable Suture Passer. Do Not Reuse
 The Ceterix NovoStitch Disposable Suture Passer Ceterix Orthopaedics 959 Hamilton Avenue Menlo Park, CA 94025 Customer Service: (650) 241-1748 IK Sterile D Do Not Reuse i See Instructions for Use THE CETERIX
The Ceterix NovoStitch Disposable Suture Passer Ceterix Orthopaedics 959 Hamilton Avenue Menlo Park, CA 94025 Customer Service: (650) 241-1748 IK Sterile D Do Not Reuse i See Instructions for Use THE CETERIX
Technique Guide Small Bone Fusion System
 Technique Guide Small Bone Fusion System The Pinit Plate Small Bone Fusion System is a super low profile, modular bone plate and screw system designed to stabilize a bunionectomy with a medial to lateral
Technique Guide Small Bone Fusion System The Pinit Plate Small Bone Fusion System is a super low profile, modular bone plate and screw system designed to stabilize a bunionectomy with a medial to lateral
Cardiva Catalyst III INSTRUCTIONS FOR USE
 Cardiva Catalyst III INSTRUCTIONS FOR USE IFU 2469 Rev. G CAUTION Federal (USA) law restricts this device to sale by or on the order of a physician. For single use only GRAPHICAL SYMBOLS ON THE CARDIVA
Cardiva Catalyst III INSTRUCTIONS FOR USE IFU 2469 Rev. G CAUTION Federal (USA) law restricts this device to sale by or on the order of a physician. For single use only GRAPHICAL SYMBOLS ON THE CARDIVA
Instructions for Use Reprocessed Ethicon ENDOPATH XCEL TM Bladeless Trocar with OPTIVIEW Technology
 Reprocessed by Instructions for Use Reprocessed Ethicon ENDOPATH XCEL TM Bladeless Trocar with OPTIVIEW Technology Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device
Reprocessed by Instructions for Use Reprocessed Ethicon ENDOPATH XCEL TM Bladeless Trocar with OPTIVIEW Technology Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device
Ponsky * PEG Safety System - "Pull" Bard * PEG Safety System - "Guidewire" Information for Use
 Ponsky * PEG Safety System - "Pull" Bard * PEG Safety System - "Guidewire" Information for Use Rx only Single patient use DEHP-Free This product and package do not contain natural rubber latex STERILE
Ponsky * PEG Safety System - "Pull" Bard * PEG Safety System - "Guidewire" Information for Use Rx only Single patient use DEHP-Free This product and package do not contain natural rubber latex STERILE
Instructions for Use Enbrel (en-brel) (etanercept) for injection, for subcutaneous use Multiple-dose Vial
 Instructions for Use Enbrel (en-brel) (etanercept) for injection, for subcutaneous use Multiple-dose Vial How do I prepare and give an injection with Enbrel multiple-dose vial? A multiple-dose vial contains
Instructions for Use Enbrel (en-brel) (etanercept) for injection, for subcutaneous use Multiple-dose Vial How do I prepare and give an injection with Enbrel multiple-dose vial? A multiple-dose vial contains
The following languages are included in this packet:
 OSTEOSET XR BONE VOID FILLER 150841-0 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) 中文 - Chinese
OSTEOSET XR BONE VOID FILLER 150841-0 The following languages are included in this packet: English (en) Deutsch (de) Nederlands (nl) Français (fr) Español (es) Italiano (it) Português (pt) 中文 - Chinese
Step by Step Description Use of Bonopty Coaxial Bone Biopsy System
 Step by Step Description Use of Bonopty Coaxial Bone Biopsy System Before use, please read the Instructions For Use which accompany the product for indications, contraindications, warnings and precautions.
Step by Step Description Use of Bonopty Coaxial Bone Biopsy System Before use, please read the Instructions For Use which accompany the product for indications, contraindications, warnings and precautions.
Step by step description - Use of Bonopty Bone Biopsy System, 12 gauge
 Step by step description - Use of Bonopty Bone Biopsy System, 12 gauge Before use, please read the Instructions For Use which accompany the product for indications, contraindications, warnings and precautions.
Step by step description - Use of Bonopty Bone Biopsy System, 12 gauge Before use, please read the Instructions For Use which accompany the product for indications, contraindications, warnings and precautions.
Pinit Plate Small Bone Fusion System Bone Plate & Screw System
 Pinit Plate Small Bone Fusion System Bone Plate & Screw System Description The Pinit Plate Small Bone Fusion System consists of 2-hole bone plates made available in three length options and two thickness
Pinit Plate Small Bone Fusion System Bone Plate & Screw System Description The Pinit Plate Small Bone Fusion System consists of 2-hole bone plates made available in three length options and two thickness
TJF-160F/VF Cleaning and Disinfection Checklist
 TJF-160F/VF Cleaning and Disinfection Checklist TJF-160F/VF Cleaning and Disinfection Checklist This checklist is used to evaluate and confirm if cleaning and disinfection of the TJF-160F/VF has been performed
TJF-160F/VF Cleaning and Disinfection Checklist TJF-160F/VF Cleaning and Disinfection Checklist This checklist is used to evaluate and confirm if cleaning and disinfection of the TJF-160F/VF has been performed
Step by step description - Use of Bonopty Bone Biopsy System, 14 gauge
 Step by step description - Use of Bonopty Bone Biopsy System, 14 gauge Before use, please read the Instructions For Use which accompany the product for indications, contraindications, warnings and precautions.
Step by step description - Use of Bonopty Bone Biopsy System, 14 gauge Before use, please read the Instructions For Use which accompany the product for indications, contraindications, warnings and precautions.
TruGuard Custom Tongue and Jaw Positioner Instructions for Use
 TruGuard Custom Tongue and Jaw Positioner Instructions for Use Caution: Federal Law restricts this device to sale by or on the order of a licensed Physician or Radiation Therapist. DEVICE DESCRIPTION Bionix
TruGuard Custom Tongue and Jaw Positioner Instructions for Use Caution: Federal Law restricts this device to sale by or on the order of a licensed Physician or Radiation Therapist. DEVICE DESCRIPTION Bionix
with Blom-Singer Gel Cap Insertion System
 with Blom-Singer Gel Cap Insertion System MEDICAL PROFESSIONAL Instructions For Use R2 37269-02G 37269-02G Effective January 2016 Blom-Singer and InHealth Technologies are registered trademarks of Freudenberg
with Blom-Singer Gel Cap Insertion System MEDICAL PROFESSIONAL Instructions For Use R2 37269-02G 37269-02G Effective January 2016 Blom-Singer and InHealth Technologies are registered trademarks of Freudenberg
Passer. Sterile. D i. Do Not PASSER DESCRIPTION. The Ceterix. Page 1
 The Ceterix NovoStitch Disposable Suture Passer Ceterix Orthopaedic cs 959 Hamilton Avenue Menlo Park, CA 94025 Customer Service: (650) 241 1748 IK D i Sterile Do Not Reusee See Instructions for Use THE
The Ceterix NovoStitch Disposable Suture Passer Ceterix Orthopaedic cs 959 Hamilton Avenue Menlo Park, CA 94025 Customer Service: (650) 241 1748 IK D i Sterile Do Not Reusee See Instructions for Use THE
Peel-Apart Percutaneous Introducer Kits for
 Bard Access Systems Peel-Apart Percutaneous Introducer Kits for Table of Contents Contents Page Bard Implanted Ports Hickman*, Leonard*, Broviac*, Tenckhoff*, and Groshong* Catheters Introduction....................................
Bard Access Systems Peel-Apart Percutaneous Introducer Kits for Table of Contents Contents Page Bard Implanted Ports Hickman*, Leonard*, Broviac*, Tenckhoff*, and Groshong* Catheters Introduction....................................
INSTRUCTIONS FOR USE AND CARE
 DIAMONDS INSTRUCTIONS FOR USE AND CARE DIAMONDS, MULTI-USE, FRICTION GRIP Quala Diamonds are a rotary cutting device made of stainless steel and coated with diamond particles on the working end. It is
DIAMONDS INSTRUCTIONS FOR USE AND CARE DIAMONDS, MULTI-USE, FRICTION GRIP Quala Diamonds are a rotary cutting device made of stainless steel and coated with diamond particles on the working end. It is
PRO-DENSE Bone Graft Substitute The following languages are included in this packet:
 EN PRO-DENSE Bone Graft Substitute 133486-11 The following languages are included in this packet: English (en) For additional languages, visit our website www.wmt.com Then click on the Prescribing Information
EN PRO-DENSE Bone Graft Substitute 133486-11 The following languages are included in this packet: English (en) For additional languages, visit our website www.wmt.com Then click on the Prescribing Information
AXIOS Stent and Electrocautery Enhanced Delivery System. Quick Reference Guide
 AXIOS Stent and Electrocautery Enhanced Delivery System Quick Reference Guide AXIOS Stent (front and side views) UPN Codes Flange Diameter (mm) Lumen Diamter (mm) Saddle Length (mm) Catheter OD (Fr) Catheter
AXIOS Stent and Electrocautery Enhanced Delivery System Quick Reference Guide AXIOS Stent (front and side views) UPN Codes Flange Diameter (mm) Lumen Diamter (mm) Saddle Length (mm) Catheter OD (Fr) Catheter
Tissue Acquisition. Introducing our large range of single use accessories for the collection of histology and cytology in the GI tract.
 Tissue Acquisition Introducing our large range of single use accessories for the collection of histology and cytology in the GI tract. MAKING A DIFFERENCE TO HEALTH Infinity ERCP Sampling Device STIFFER
Tissue Acquisition Introducing our large range of single use accessories for the collection of histology and cytology in the GI tract. MAKING A DIFFERENCE TO HEALTH Infinity ERCP Sampling Device STIFFER
Technique Guide KISSloc Suture System
 Technique Guide KISSloc Suture System The KISSloc Suture System consists of a strong self-cinching suture assembly, a unique load-dispersing Arrow Plate and procedure specific instrumentation. Simple,
Technique Guide KISSloc Suture System The KISSloc Suture System consists of a strong self-cinching suture assembly, a unique load-dispersing Arrow Plate and procedure specific instrumentation. Simple,
Lotus transducer Instructions for use
 Lotus transducer Instructions for use Instructions for use transducer Before using the product, please read the following information thoroughly Important This document provides instructions for using
Lotus transducer Instructions for use Instructions for use transducer Before using the product, please read the following information thoroughly Important This document provides instructions for using
PRO-DENSE BONE GRAFT SUBSTITUTE
 PRO-DENSE BONE GRAFT SUBSTITUTE 152917-0 The following languages are included in this packet: For additional languages, visit our website www.wright.com. Then click on the option. For additional information
PRO-DENSE BONE GRAFT SUBSTITUTE 152917-0 The following languages are included in this packet: For additional languages, visit our website www.wright.com. Then click on the option. For additional information
for the ideal insertion of peritoneal dialysis catheter (patent pending)
 for the ideal insertion of peritoneal dialysis catheter (patent pending) CAUTION: U.S. federal law restricts this device to sale by or on the order of a properly licensed practitioner. DEVICE DESCRIPTION
for the ideal insertion of peritoneal dialysis catheter (patent pending) CAUTION: U.S. federal law restricts this device to sale by or on the order of a properly licensed practitioner. DEVICE DESCRIPTION
Directions For Use. All directions should be read before use. Page 1 of 8
 Directions For Use All directions should be read before use Page 1 of 8 WARNING: For single use only. Do not reuse, reprocess or re-sterilize. Reuse, reprocessing or re-sterilization may compromise the
Directions For Use All directions should be read before use Page 1 of 8 WARNING: For single use only. Do not reuse, reprocess or re-sterilize. Reuse, reprocessing or re-sterilization may compromise the
Figure 1: Revolution TM Peripheral Atherectomy System Diagram. Table 1: Revolution TM Peripheral Atherectomy System Specifications Minimum Burr
 Instructions For Use All instructions should be read before use CAUTION Investigational Device. Limited by Federal (or United States) law to investigational use. DEVICE DESCRIPTION: The Revolution TM Peripheral
Instructions For Use All instructions should be read before use CAUTION Investigational Device. Limited by Federal (or United States) law to investigational use. DEVICE DESCRIPTION: The Revolution TM Peripheral
WallFlex Biliary Metal Stents
 WallFlex Biliary Metal Stents Please Note: These steps are intended to be for general guidance only. For complete instructions for use, indications, contraindications and warnings, please refer to the
WallFlex Biliary Metal Stents Please Note: These steps are intended to be for general guidance only. For complete instructions for use, indications, contraindications and warnings, please refer to the
low ProfIle neuro PlaTIng system
 low ProfIle neuro PlaTIng system surgical TeChnIque Table of Contents Introduction Low Profile Neuro Cranial Plating System 2 Surgical Technique Technique 5 Product Information Low Profile Neuro Plates
low ProfIle neuro PlaTIng system surgical TeChnIque Table of Contents Introduction Low Profile Neuro Cranial Plating System 2 Surgical Technique Technique 5 Product Information Low Profile Neuro Plates
Lag Screw Device Intended for symphyseal fracture fixation of the mandible
 Lag Screw Device Intended for symphyseal fracture fixation of the mandible SUrgicaL TecHNiqUe Lag Screw Device Intended for symphyseal fracture fixation of the mandible Simplifies the lag screw fixation
Lag Screw Device Intended for symphyseal fracture fixation of the mandible SUrgicaL TecHNiqUe Lag Screw Device Intended for symphyseal fracture fixation of the mandible Simplifies the lag screw fixation
Instructions for Use HEMLIBRA (hem-lee-bruh) (emicizumab-kxwh) injection, for subcutaneous use
 Instructions for Use HEMLIBRA (hem-lee-bruh) (emicizumab-kxwh) injection, for subcutaneous use Be sure that you read, understand, and follow the Instructions for Use before injecting HEMLIBRA. Your healthcare
Instructions for Use HEMLIBRA (hem-lee-bruh) (emicizumab-kxwh) injection, for subcutaneous use Be sure that you read, understand, and follow the Instructions for Use before injecting HEMLIBRA. Your healthcare
Low Profile Neuro Plating System. Surgical Technique
 Low Profile Neuro Plating System Surgical Technique TABLE OF CONTENTS INTRODUCTION Low Profile Neuro Plating System 2 SURGICAL TECHNIQUE Technique 5 PRODUCT INFORMATION Low Profile Neuro Plates 10 Low
Low Profile Neuro Plating System Surgical Technique TABLE OF CONTENTS INTRODUCTION Low Profile Neuro Plating System 2 SURGICAL TECHNIQUE Technique 5 PRODUCT INFORMATION Low Profile Neuro Plates 10 Low
Absorbable Woven Polyglycolic Acid Mesh Tube (Absorbable Nerve Conduit Tube) INSTRUCTIONS FOR USE 2 6
 Absorbable Woven Polyglycolic Acid Mesh Tube (Absorbable Nerve Conduit Tube) INSTRUCTIONS FOR USE 2 6 1 0086 SYMBOL DEFINITIONS ENGLISH Do not Reuse Consult Instructions For Use Ethylene Oxide Sterilized
Absorbable Woven Polyglycolic Acid Mesh Tube (Absorbable Nerve Conduit Tube) INSTRUCTIONS FOR USE 2 6 1 0086 SYMBOL DEFINITIONS ENGLISH Do not Reuse Consult Instructions For Use Ethylene Oxide Sterilized
Inspection Window. Syringe Body
 Guide to Parts Before use Instructions for Use UDENYCA (yoo-den-i-kah) (pegfilgrastim-cbqv) Injection Single-Dose Prefilled Syringe Inspection Window Needle Cap Needle Label Plunger Plunger Head Needle
Guide to Parts Before use Instructions for Use UDENYCA (yoo-den-i-kah) (pegfilgrastim-cbqv) Injection Single-Dose Prefilled Syringe Inspection Window Needle Cap Needle Label Plunger Plunger Head Needle
3.5 mm Locking Attachment Plate
 For Treatment of Periprosthetic Fractures 3.5 mm Locking Attachment Plate Surgical Technique Table of Contents Introduction 3.5 mm Locking Attachment Plate 2 Indications 4 Surgical Technique Preparation
For Treatment of Periprosthetic Fractures 3.5 mm Locking Attachment Plate Surgical Technique Table of Contents Introduction 3.5 mm Locking Attachment Plate 2 Indications 4 Surgical Technique Preparation
Peritoneal Drainage System
 ASEPT Drainage Parts and Accessories (Provided separately, see package label for contents.) ASEPT Pleural Drainage System 622289 (1 each) (includes ASEPT drainage catheter and insertion kit) ASEPT Peritoneal
ASEPT Drainage Parts and Accessories (Provided separately, see package label for contents.) ASEPT Pleural Drainage System 622289 (1 each) (includes ASEPT drainage catheter and insertion kit) ASEPT Peritoneal
Visit Linvatec.com today to learn more. Surgical Technique: Sequential Meniscal Running Stitch
 ORDERING INFORMATION Sequent Meniscal Repair System MR004S MR007S MR004C MR007C SC047D Straight Needle, 4 Implants Straight Needle, 7 Implants Curved Needle, 4 Implants Curved Needle, 7 implants Sequent
ORDERING INFORMATION Sequent Meniscal Repair System MR004S MR007S MR004C MR007C SC047D Straight Needle, 4 Implants Straight Needle, 7 Implants Curved Needle, 4 Implants Curved Needle, 7 implants Sequent
Sharps Container (not included) 1 Gather and check supplies. Gather supplies
 Instructions for Use BENLYSTA (ben-list-ah) (belimumab) injection, for subcutaneous use Prefilled Syringe Read this Instructions for Use before you start to use BENLYSTA and each time you refill your prescription.
Instructions for Use BENLYSTA (ben-list-ah) (belimumab) injection, for subcutaneous use Prefilled Syringe Read this Instructions for Use before you start to use BENLYSTA and each time you refill your prescription.
CATHETER ACCESS KIT. For use with Prometra Programmable Infusion Systems
 CATHETER ACCESS KIT Caution: Federal (USA) Law restricts this device to sale by or on the order of a physician. Table of Contents Contents... 3 Description... 3 Indications... 3 Contraindications... 3
CATHETER ACCESS KIT Caution: Federal (USA) Law restricts this device to sale by or on the order of a physician. Table of Contents Contents... 3 Description... 3 Indications... 3 Contraindications... 3
Correction System. Surgical Technique
 Nextra Hammertoe Correction System Surgical Technique Maximized Bone Purchase* Stable and Secure Phalanx Optimized Screw Design Adjustable Bone-to-Bone Apposition Progressive Ratchet Tightening Mechanism
Nextra Hammertoe Correction System Surgical Technique Maximized Bone Purchase* Stable and Secure Phalanx Optimized Screw Design Adjustable Bone-to-Bone Apposition Progressive Ratchet Tightening Mechanism
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device. Instructions for Use
 The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
Altus Product description / Instructions for Use IFU 6 Rev. 1
 Content: 1 Introduction... 2 1.1 Legend... 2 1.2 Indication... 2 1.3 Contraindication... 2 1.4 Field of Application... 2 1.5 Sterilization... 2 1.6 Storage... 2 1.7 Allergies... 2 2 SYSTEM DESCRIPTION...
Content: 1 Introduction... 2 1.1 Legend... 2 1.2 Indication... 2 1.3 Contraindication... 2 1.4 Field of Application... 2 1.5 Sterilization... 2 1.6 Storage... 2 1.7 Allergies... 2 2 SYSTEM DESCRIPTION...
Doc no: IFU/CPL Issue date: Rev no: 00 Rev date: 1
 FUTURA SURGICARE PVT LTD INSTRUCTION FOR USE Brand name: PROGUT Material: Catgut Plain. Absorbable surgical suture U.S.P Description: Progut surgical catgut suture is a sterile absorbable suture composed
FUTURA SURGICARE PVT LTD INSTRUCTION FOR USE Brand name: PROGUT Material: Catgut Plain. Absorbable surgical suture U.S.P Description: Progut surgical catgut suture is a sterile absorbable suture composed
Oral Specimen Collection Device
 Oral Specimen Collection Device For In Vitro Diagnostic Use NAME AND INTENDED USE The Intercept Oral Specimen Collection Device is intended for use in the collection, preservation and transport of oral
Oral Specimen Collection Device For In Vitro Diagnostic Use NAME AND INTENDED USE The Intercept Oral Specimen Collection Device is intended for use in the collection, preservation and transport of oral
INSTRUCTIONS FOR PREPARING AND GIVING AN INJECTION OF ENBREL POWDER
 INSTRUCTIONS FOR PREPARING AND GIVING AN INJECTION OF ENBREL POWDER Introduction The following instructions explain how to prepare and inject Enbrel powder for injection. Please read the instructions carefully
INSTRUCTIONS FOR PREPARING AND GIVING AN INJECTION OF ENBREL POWDER Introduction The following instructions explain how to prepare and inject Enbrel powder for injection. Please read the instructions carefully
Marrow Cellution Bone Marrow Aspirate (BMA) and Stem Cell Harvesting System
 SURGICAL TECHNIQUE Marrow Cellution Bone Marrow Aspirate (BMA) and Stem Cell Harvesting System Medtronic is the most empowering source of biologics solutions offering an unmatched combination of therapies,
SURGICAL TECHNIQUE Marrow Cellution Bone Marrow Aspirate (BMA) and Stem Cell Harvesting System Medtronic is the most empowering source of biologics solutions offering an unmatched combination of therapies,
INSTRUCTIONS FOR USE
 Comfort Injects 50 Comfort-in INSTRUCTIONS FOR USE English user manual 1 Introduction Thank you for choosing the Comfort in Needle free Injection system, developed with your comfort and convenience in
Comfort Injects 50 Comfort-in INSTRUCTIONS FOR USE English user manual 1 Introduction Thank you for choosing the Comfort in Needle free Injection system, developed with your comfort and convenience in
SYMBOLOGY. = Manufacturer. = Manufacturing Serial Number. = Manufacturing Lot Number = Catalog Number LOT REF. = Do not resterilize Single Use Only
 damage or expenses directly or indirectly arising from use of this product. This warranty is in lieu of and excludes all other warranties not expressly set forth herein, whether express or implied by operation
damage or expenses directly or indirectly arising from use of this product. This warranty is in lieu of and excludes all other warranties not expressly set forth herein, whether express or implied by operation
BACTISEAL Endoscopic Ventricular Catheter (REF & )
 BACTISEAL Endoscopic Ventricular Catheter (REF 82-3087 & 82-3088) LCN 206966-001/E 2011 2017 DePuy Synthes. All rights reserved. Revised 07/17 206966-001-E.indd 1 ENGLISH IMPORTANT INFORMATION Please Read
BACTISEAL Endoscopic Ventricular Catheter (REF 82-3087 & 82-3088) LCN 206966-001/E 2011 2017 DePuy Synthes. All rights reserved. Revised 07/17 206966-001-E.indd 1 ENGLISH IMPORTANT INFORMATION Please Read
RECOMMENDED INSTRUCTIONS FOR USE
 Rapid Exchange PTCA Dilatation Catheter RECOMMENDED INSTRUCTIONS FOR USE Available in diameters 1.25mm to 4.5mm and in lengths 09mm to 40mm Caution: This device should be used only by physicians trained
Rapid Exchange PTCA Dilatation Catheter RECOMMENDED INSTRUCTIONS FOR USE Available in diameters 1.25mm to 4.5mm and in lengths 09mm to 40mm Caution: This device should be used only by physicians trained
SURGICAL TECHNIQUE GUIDE
 DANGER indicates an imminently hazardous situation which, if not avoided, will result in death or serious injury. WARNING indicates a potentially hazardous situation which, if not avoided, could result
DANGER indicates an imminently hazardous situation which, if not avoided, will result in death or serious injury. WARNING indicates a potentially hazardous situation which, if not avoided, could result
How to Use ENBREL : Vial Adapter Method
 How to Use ENBREL : Vial Adapter Method SETTING UP FOR AN INJECTION Select a clean, well-lit, flat working surface, such as a table. Take the ENBREL dose tray out of the refrigerator and place it on your
How to Use ENBREL : Vial Adapter Method SETTING UP FOR AN INJECTION Select a clean, well-lit, flat working surface, such as a table. Take the ENBREL dose tray out of the refrigerator and place it on your
Integra SURGICAL TECHNIQUE. MemoFix Super Elastic Nitinol Staple System. Super Elastic Nitinol Staple System
 Integra MemoFix Super Elastic Nitinol Staple System SURGICAL TECHNIQUE Super Elastic Nitinol Staple System Table of Contents Design Rationale...2 System Features...2 Indications...2 Contraindications...2
Integra MemoFix Super Elastic Nitinol Staple System SURGICAL TECHNIQUE Super Elastic Nitinol Staple System Table of Contents Design Rationale...2 System Features...2 Indications...2 Contraindications...2
Guidance for. implant removal. Straumann Dental Implant System
 Guidance for implant removal Straumann Dental Implant System The ITI (International Team for Implantology) is academic partner of Institut Straumann AG in the areas of research and education. CONTENTS
Guidance for implant removal Straumann Dental Implant System The ITI (International Team for Implantology) is academic partner of Institut Straumann AG in the areas of research and education. CONTENTS
Instructions for Use. For use with. 10 mg vial
 Instructions for Use For use with 10 mg vial Table of Contents Parts of the ZOMA-Jet 10... 1 Supplies you will need to mix a ZOMACTON 10 mg Vial... 2 Mix a ZOMACTON 10 mg vial... 3 Reset the ZOMA-Jet 10...
Instructions for Use For use with 10 mg vial Table of Contents Parts of the ZOMA-Jet 10... 1 Supplies you will need to mix a ZOMACTON 10 mg Vial... 2 Mix a ZOMACTON 10 mg vial... 3 Reset the ZOMA-Jet 10...
PORTO 2 VENT CPAP OS. Operator s Manual. PORTO 2VENT CPAP OS System Operator s Manual Part Number Rev I
 PORTO 2 VENT CPAP OS Operator s Manual 1 2 TABLE of CONTENTS 1. Introduction 3. Operating Instructions 1a. Definitions 3a. Setting the CPAP Level 1b. General Description 3b. Applying the Breathing Circuit
PORTO 2 VENT CPAP OS Operator s Manual 1 2 TABLE of CONTENTS 1. Introduction 3. Operating Instructions 1a. Definitions 3a. Setting the CPAP Level 1b. General Description 3b. Applying the Breathing Circuit
Instructions for Use Neulasta (nu-las-tah) (pegfilgrastim) Injection, for subcutaneous use Single-Dose Prefilled Syringe. Plunger rod Used plunger rod
 Instructions for Use Neulasta (nu-las-tah) (pegfilgrastim) Injection, for subcutaneous use Single-Dose Prefilled Syringe Guide to parts Before use After use Plunger rod Used plunger rod Finger grip Label
Instructions for Use Neulasta (nu-las-tah) (pegfilgrastim) Injection, for subcutaneous use Single-Dose Prefilled Syringe Guide to parts Before use After use Plunger rod Used plunger rod Finger grip Label
Instructions. Fibrlok Optical Fiber Splicing System. Issue 6, August 1991
 Instructions TM Fibrlok Optical Fiber Splicing System Issue 6, August 1991 1.0 General The 3M brand Fibrlok TM Optical Fiber Splicing System provides permanent mechanical splices for single or multi mode
Instructions TM Fibrlok Optical Fiber Splicing System Issue 6, August 1991 1.0 General The 3M brand Fibrlok TM Optical Fiber Splicing System provides permanent mechanical splices for single or multi mode
8 Performing Medical Procedures
 8 The Terason usmart3200t Ultrasound System can aid in performing medical procedures such as biopsies. Depending on whether you purchased the additional equipment required for these procedures, you may
8 The Terason usmart3200t Ultrasound System can aid in performing medical procedures such as biopsies. Depending on whether you purchased the additional equipment required for these procedures, you may
Transjugular liver access and biopsy
 Transjugular liver access and biopsy An illustrated guide Illustrations by Lisa Clark LABS LIVER ACCESS AND BIOPSY SET MEDICAL Support your procedures by using products that are specifically designed for
Transjugular liver access and biopsy An illustrated guide Illustrations by Lisa Clark LABS LIVER ACCESS AND BIOPSY SET MEDICAL Support your procedures by using products that are specifically designed for
Instructions for Use io-flex Probe
 DEVICE DESCRIPTION: The is an accessory of the Amendia io-flex System, which includes the MicroBlade Shaver device. The io-flex Probe is designed to access the neural foramen. It is comprised of an outer
DEVICE DESCRIPTION: The is an accessory of the Amendia io-flex System, which includes the MicroBlade Shaver device. The io-flex Probe is designed to access the neural foramen. It is comprised of an outer
CrossFix II. All-Suture, All-Inside Meniscal Repair System. Surgical Technique
 CrossFix II All-Suture, All-Inside Meniscal Repair System Surgical Technique 3 CrossFix II All- Suture Repair System Surgical Technique All-Inside Repair with Inside-Out Results The CrossFix II Meniscal
CrossFix II All-Suture, All-Inside Meniscal Repair System Surgical Technique 3 CrossFix II All- Suture Repair System Surgical Technique All-Inside Repair with Inside-Out Results The CrossFix II Meniscal
COR. Precision Targeting Cartilage Repair System. Arthroscopic Technique for Repair of Osteochondral Defects
 COR Precision Targeting Cartilage Repair System Arthroscopic Technique for Repair of Osteochondral Defects Planning the Procedure An 18-gauge spinal needle is initially used to plan a perpendicular approach
COR Precision Targeting Cartilage Repair System Arthroscopic Technique for Repair of Osteochondral Defects Planning the Procedure An 18-gauge spinal needle is initially used to plan a perpendicular approach
The Enbrel SureClick autoinjector is a single-dose prefilled autoinjector. It contains one 50 mg dose of Enbrel.
 Instructions for Use Welcome! The Enbrel SureClick autoinjector is a single-dose prefilled autoinjector. It contains one 50 mg dose of Enbrel. Your healthcare provider has prescribed Enbrel SureClick autoinjector
Instructions for Use Welcome! The Enbrel SureClick autoinjector is a single-dose prefilled autoinjector. It contains one 50 mg dose of Enbrel. Your healthcare provider has prescribed Enbrel SureClick autoinjector
Instructions for Use 1
 Instructions for Use 1 Instructions for Use IMPORTANT! Please read before use. Description The Sniper is a dual lumen catheter comprised of an inner guidewire/infusion lumen and an outer, annular lumen
Instructions for Use 1 Instructions for Use IMPORTANT! Please read before use. Description The Sniper is a dual lumen catheter comprised of an inner guidewire/infusion lumen and an outer, annular lumen
Instructions for Use. Welcome!
 Instructions for Use Welcome! The AMJEVITA SureClick autoinjector is a single-use prefilled autoinjector. Consult your doctor if you have any questions about your dose. Your doctor has prescribed AMJEVITA
Instructions for Use Welcome! The AMJEVITA SureClick autoinjector is a single-use prefilled autoinjector. Consult your doctor if you have any questions about your dose. Your doctor has prescribed AMJEVITA
Instructions for Use Enbrel (en-brel) (etanercept) injection, for subcutaneous use Single-dose Prefilled Syringe
 Instructions for Use Enbrel (en-brel) (etanercept) injection, for subcutaneous use Single-dose Prefilled Syringe How do I prepare and give an injection with Enbrel Single-dose Prefilled Syringe? There
Instructions for Use Enbrel (en-brel) (etanercept) injection, for subcutaneous use Single-dose Prefilled Syringe How do I prepare and give an injection with Enbrel Single-dose Prefilled Syringe? There
MEDICAL CORPORATION Asbury Rd., P.O. Box 758, Farmingdale, NJ USA Fax
 MEDICAL CORPORATION 5206 Asbury Rd., P.O. Box 758, Farmingdale, NJ 07727 USA 732-938-2266 800-323-4035 Fax 732-938-2399 MYO/WIRE Temporary Cardiac Pacing Wire System MYO/WIRE temporary cardiac pacing wires.
MEDICAL CORPORATION 5206 Asbury Rd., P.O. Box 758, Farmingdale, NJ 07727 USA 732-938-2266 800-323-4035 Fax 732-938-2399 MYO/WIRE Temporary Cardiac Pacing Wire System MYO/WIRE temporary cardiac pacing wires.
Arthroscopic Shoulder Repair Using the Smith & Nephew KINSA Suture Anchor
 Shoulder Series Technique Guide *smith&nephew KINSA Suture Anchor Arthroscopic Shoulder Repair Using the Smith & Nephew KINSA Suture Anchor Reviewed by: Larry D. Field, MD Mississippi Sports Medicine and
Shoulder Series Technique Guide *smith&nephew KINSA Suture Anchor Arthroscopic Shoulder Repair Using the Smith & Nephew KINSA Suture Anchor Reviewed by: Larry D. Field, MD Mississippi Sports Medicine and
Instructions for Use Reprocessed 3D Diagnostic Ultrasound Catheters
 Reprocessed by Instructions for Use Reprocessed 3D Diagnostic Ultrasound Catheters Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a
Reprocessed by Instructions for Use Reprocessed 3D Diagnostic Ultrasound Catheters Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a
Quick Reference Guide
 Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER
 Page 1 of 5 NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER STERILE. SINGLE USE ONLY. Sterilized with ethylene oxide gas. Non pyrogenic. Do not resterilize. Do not use opened or damaged packages.
Page 1 of 5 NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER STERILE. SINGLE USE ONLY. Sterilized with ethylene oxide gas. Non pyrogenic. Do not resterilize. Do not use opened or damaged packages.
For the Attention of the Operating Surgeon: IMPORTANT INFORMATION ON THE MATRIXRIB FIXATION SYSTEM
 For the Attention of the Operating Surgeon: IMPORTANT INFORMATION ON THE MATRIXRIB FIXATION SYSTEM 10/16 GP2685-E-CAN DESCRIPTION The MatrixRIB Fixation System consists of locking plates, locking screws,
For the Attention of the Operating Surgeon: IMPORTANT INFORMATION ON THE MATRIXRIB FIXATION SYSTEM 10/16 GP2685-E-CAN DESCRIPTION The MatrixRIB Fixation System consists of locking plates, locking screws,
ASEPT. Pleural Drainage System INSTRUCTIONS FOR USE REF LOT STERILE EO. Manufactured for: 824 Twelfth Avenue Bethlehem, PA
 ASEPT Pleural Drainage System LS-00116-01-AB 017-11 INSTRUCTIONS FOR USE Do not reuse STERILIZE Do not resterilize 30 C Rx only 15 C REF Store at controlled room temperature 15-30 C (59-86 F) Keep away
ASEPT Pleural Drainage System LS-00116-01-AB 017-11 INSTRUCTIONS FOR USE Do not reuse STERILIZE Do not resterilize 30 C Rx only 15 C REF Store at controlled room temperature 15-30 C (59-86 F) Keep away
ASEPT. Pleural Drainage System INSTRUCTIONS FOR USE. Rx only REF LOT. STERILE EO Sterilized using ethylene oxide
 ASEPT Pleural Drainage System INSTRUCTIONS FOR USE L1171/D REV 2014-02 2 Do not reuse 2 STERILIZE Do not resterilize Store at room temperature STERILE EO Sterilized using ethylene oxide Rx only Do not
ASEPT Pleural Drainage System INSTRUCTIONS FOR USE L1171/D REV 2014-02 2 Do not reuse 2 STERILIZE Do not resterilize Store at room temperature STERILE EO Sterilized using ethylene oxide Rx only Do not
FS MTS. Micro Periodontal Tissue Scissors
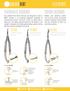 FEATHERLITE SCISSORS Our patented Flex Series Scissors are designed to have a 300% increase in its shearing angulation compared to conventional scissors. The result is that, no mater what is being cut,
FEATHERLITE SCISSORS Our patented Flex Series Scissors are designed to have a 300% increase in its shearing angulation compared to conventional scissors. The result is that, no mater what is being cut,
Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a physician.
 Page 1 of 5 Reprocessed by Instructions for Use Reprocessed Ultrasound Catheters Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a physician.
Page 1 of 5 Reprocessed by Instructions for Use Reprocessed Ultrasound Catheters Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a physician.
NONGYNECOLOGICAL CYTOLOGY FINE NEEDLE ASPIRATION SPECIMENS
 NONGYNECOLOGICAL CYTOLOGY FINE NEEDLE ASPIRATION SPECIMENS I. Purpose Fine needle aspiration of mass lesions is commonly utilized in the detection and characterization of a variety of malignant diseases.
NONGYNECOLOGICAL CYTOLOGY FINE NEEDLE ASPIRATION SPECIMENS I. Purpose Fine needle aspiration of mass lesions is commonly utilized in the detection and characterization of a variety of malignant diseases.
Doc no: IFU/CC Issue date: Rev no: 04 Rev date:
 FUTURA SURGICARE PVT LTD INSTRUCTION FOR USE Brand name: PROGUT Material: Catgut Chromic Absorbable surgical suture U.S.P Description: Progut surgical catgut suture is a sterile absorbable suture composed
FUTURA SURGICARE PVT LTD INSTRUCTION FOR USE Brand name: PROGUT Material: Catgut Chromic Absorbable surgical suture U.S.P Description: Progut surgical catgut suture is a sterile absorbable suture composed
eptfe Vascular Graft Instructions for Use
 TM eptfe Vascular Graft Instructions for Use R Instructions For Use Caution: Federal (U.S.A) law restricts this device to sale by or on the order of a physician. Device Description, Indications, Contraindications,
TM eptfe Vascular Graft Instructions for Use R Instructions For Use Caution: Federal (U.S.A) law restricts this device to sale by or on the order of a physician. Device Description, Indications, Contraindications,
Aspira* Peritoneal Drainage Catheter
 Aspira* Peritoneal Drainage Catheter Instructions For Use Access Systems Product Description: The Aspira* Peritoneal Drainage Catheter is a tunneled, long-term catheter used to drain accumulated fluid
Aspira* Peritoneal Drainage Catheter Instructions For Use Access Systems Product Description: The Aspira* Peritoneal Drainage Catheter is a tunneled, long-term catheter used to drain accumulated fluid
