Clinical Trials Related to Age Related Macular Degeneration
|
|
|
- Lynette Cobb
- 5 years ago
- Views:
Transcription
1 Clinical Trials Related to Age Related Macular Degeneration Kirti Singh MD, DNB, FRCS Kirti Singh MD, DNB, FRCS, Pooja Jain MBBS, Nitasha Ahir MBBS, Divya Jain MD, DNB Guru Nanak Eye Centre, Maulana Azad Medical College, New Delhi AREDS (Age Related Eye Disease Study) 1 To assess the clinical course, prognosis, and risk factors of age-related macular degeneration (AMD) and cataract. A prospective multicenter randomized clinical trial involving 3640 participants. People at high risk of developing advanced stages of AMD lowered their risk by about 25% when treated with a high-dose combination of vitamin C, vitamin E, beta-carotene, and zinc. People with intermediate or advanced AMD in one eye only, the micro nutrients reduced risk of vision loss caused by advanced AMD by 19%. However if only early AMD, the supplements did not provide any apparent benefit. AREDS 2 (Age Related eye disease study 2) 1. Whether oral supplementation with lutien (10mg/ kg), zeaxanthin (2 mg/d) and /or omega -3 long chain polyunsaturated fatty acids (DHA, EFA) (1gm/d) will decrease the risk of progression to advanced AMD, as compared to placebo. 2. Evaluate the effect of eliminating beta carotene and reduced zinc from the original AREDS formulation on the development and progression of AMD patients were enrolled for the study from September Conclusion: No over all additional benefit from adding omega 3 fatty acids or lutein and zeaxanthin to the formulation. However the study did find benefits in two sugroups of patients those not given beta carotene and those with very little lutein and zeaxanthin in their diets. Macular Photocoagulation Study (MPS) 2 To evaluate if argon and krypton laser is of benefit in preventing or delaying loss of central vision in patients with Choroidal Neovascular Membrane (CNVM) due to AMD, presumed ocular histoplasmosis (POH), and idiopathic neovascular membranes (INVM) Method: MPS consisted of 3 sets of Randomized Controlled Trial (RCTs). a). Argon Study: Used argon blue-green laser photocoagulation on leaking abnormal blood vessels in CNVM outside the fovea [200 to 2500µ from center of Foveal avascular zone (FAZ)] in the three conditions: AMD, POH, and INVM b). Krypton Study: To assess whether krypton red laser photocoagulation of CNVM lesions with posterior border 1 to 199µ from centre of FAZ is of benefit in preventing or delaying large losses of vision in patients with AMD, POH, and INVM. c). Foveal Study: To assess whether laser photocoagulation benefits in preventing or delaying further vision loss in patients with new or recurrent CNVM under the centre of FAZ. In all 3 arms, eyes with well-demarcated areas of classic CNV, defined by Fluorescein angiography, had a better visual prognosis when treated with laser photocoagulation versus observation. However subfoveal CNVM benefited more from laser treatment vs observation. Laser treatment for subfoveal CNV caused more visual loss in the immediate post laser period vs controls. www. dosonline.org l 47
2 However, the amount of visual loss in control group at 1yr was same in treatment group. After 1yr, the control group exhibited more visual loss than the treated group. TAP (Treatment of AMD with Photodynamic therapy) 3 Purpose To evaluate the efficacy of Visudyne therapy in patients with subfoveal CNVM secondary to AMD with predominantly classic CNV. 609 patients with AMD and subfoveal CNV with a classic component and visual acuity (VA) between 6/12 and 6/60 underwent a 2:1 randomisation between treatment and control (sham treatment).retreatment was applied to zones of persistent or new leakage at 3 monthly visits. Visudyne therapy sustained VA, stabilized contrast sensitivity and preserved the quality of vision in patients with predominantly classic CNV (irrespective of lesion size) Twice as many patients treated with Visudyne therapy retained vision (loss of less than 3 lines of VA) compared with placebo.visudyne therapy halted disease progression and impeded growth of CNVM. Treatment offers no advantage in minimally classic subgroup. VIP (Verteporfin in photodynamic therapy) 4 To evaluate the efficacy of Visudyne therapy in occult with no classic CNVM or presumed early onset CNVM and good visual acuity. Patients of AMD, with subfoveal CNV lesions measuring no greater than 5400 µ in greatest linear dimension with either a) Occult with no classic choroidal neovascularization, BCVA score of at least 50 (Snellen equivalent approximately 20/100), and evidence of haemorrhage or recent disease progression; or b) Evidence of classic CNVM with a BCVA score of at least 70 (better than a Snellen equivalent of approximately 20/40); were assigned randomly (2:1) to verteporfin therapy or placebo therapy. At follow-up examinations every 3 months, retreatment with the same regimen was applied if angiography showed fluorescein leakage. The main outcome measure was at least moderate vision loss, that is, a loss of at least 15 letters (approximately 3 lines), adhering to an intent-to-treat analysis with the last observation carried forward to impute for missing data. Visudyne therapy sustained VA, stabilized contrast sensitivity and preserved the quality of vision in patients with occult with no classic CNV. Benefit was most pronounced in smaller lesions (< 4 MPS DA: macular photocoagulation study disc area) or lower levels of visual acuity (< 20/50) Visudyne therapy halted the disease progression and impeded lesion growth. Fewer patients developed the more aggressive classic component of CNV. VIM trial (Verteporfin in Minimally classic CNVM) 5 To evaluate the potential benefits of reducing the light fluence in eyes with minimally classic CNVM. 117 patients were randomly assigned (1:1:1) to a) verteporfin infusion (6 mg/m²) and light application with an reduced fluence (RF) rate (300 mw/cm²) for 83 seconds (light dose of 25 J/cm²) or b) an standard fluence (SF) rate (600 mw/cm²) for 83 seconds (light dose of 50 J/cm²) or c) to placebo infusion with RF or SF. Patients with Visudyne therapy with either a reduced or standard light fluence had significantly better vision outcomes and lesser conversion to predominantly classic CNVM. VISION trial (VEGF Inhibition Study In Ocular Neovascularisation) 6 Vision-Year 1 To evaluate the usefulness of Pegaptanib in treating neovascular AMD. Entire spectrum of AMD, as encountered in clinical practice was included. Prospective, double-masked study, Phase 3, placebo-controlled trial patients randomized to sham (0 mg), 0.3 mg, 1.0 mg, or 3.0 mg of drug. Intravitreal injections were given every 6 weeks. The Primary endpoint was proportion of patients avoiding 3 lines / 15 letters (EDTRS) of vision loss at one year. Secondary endpoints were visual gain (>15 letters of vision) and/or mean change from baseline visual acuity. Primary endpoint: achieved by 70% of treated (0.3mg) vs. 55% of controls (p<0.001) representing a 27% increase in responders over controls. Secondary endpoints: Risk of severe loss of vision (>30 letters ETDRS) was seen in10% of treated cases vs. 22% in control eyes. Visual gain (>15 letters) was better in treated eyes at 6% (in 0.3mg), 7% (in 1.0 mg), 4% (in 3.0mg) vs. 2% of controls. 48 l DOS Times - Vol. 19, No. 3 September, 2013
3 Change in visual acuity defined as any gain in vision (>0 letters) was seen in 33% of treated vs 10% of control eyes. Combined analysis for the Primary endpoint showed that Pegaptanib appeared to be efficacious at all doses tested (0.3mg, 1.0mg and 3.0mg). The FDA reviewed this data and approved the lowest dose of Pegaptanib, 0.3mg, for a broad label (all subtypes). Vision - Year 2 To evaluate the efficacy of one year of pegaptanib therapy vs. two years of pegaptanib therapy patients (out of 1186) continued in the VISION study for a second year. Patients in treatment arms were re-randomized to observation (i.e. treated for year one, observed for year two) OR continue Pegaptanib injections in the same dose group. Patients in the sham arm were re-randomized to receive Pegaptanib or continue to receive sham. Primary endpoint was: Time until > 3 lines or 15 letters of vision was lost. Secondary end points were: Mean change in VA at week 102, proportion of eyes becoming legally blind (<6/60) and proportion of patients losing >15 letters at week 102. Intravitreal injections of 0.3mg Pegaptanib are effective in treating exudative AMD. The therapeutic benefit is maintained regardless of lesion composition or size. Treatment benefit is greater for two years of sustained treatment rather than one year. Pegaptanib did not show statistically significant benefit among patients with occult disease or in lesions greater than 4 disc areas regardless of composition. Continuing visual benefit was observed in patients who were randomized to receive therapy with Pegaptanib in year 2 of the VISION. trials when compared with 2 years usual care or cessation of therapy at year 1. Minimally Classic/Occult Trial of the Anti-VEGF Antibody Ranibizumab in the Treatment of Neovascular AMD (MARINA) 7 To evaluate the efficacy of Ranibizumab in CNVM Phase III, multi-centre, randomised, double masked, sham injection-controlled trial, involving 716 patients, who were randomised to intravitreal Ranibizumab 0.3 or 0.5mg or sham treatment once a month for 24 months Maintaining vision: Patients losing < 15 letters vision was seen in 95% (452/478) of treated vs. 62 % (148/238) of control group (p<.0001) Improving vision: Gain of 15 letters was seen in 25% (59/238) of 0.3 mg Ranibizumab vs. 34% (81/240) of 0.5 mg of Ranibizumab versus 5% (11/238) of controls. The difference between treated (both doses) and control in mean change in visual acuity from baseline was 17 letters. Visual acuity of > 6/12 at 12 months was seen in 40 % (188/478) of treated vs. 11% (26/238) of control patients. At 12 months on an average 7 letters gained in treated group vs letters lost by control group. Intravitreal Ranibizumab 0.3 or 0.5mg was beneficial in arresting visual loss due to minimally classic or occult CNVM. Anti-VEGF antibody for the treatment of predominantly classic choroidal neovascularisation in AMD (ANCHOR) 8 Comparing two different doses of Ranibizumab to verteporfin photodynamic therapy (PDT) in treating subfoveal neovascular macular degeneration. A Phase III, randomised, multi-centre double-masked active treatment-controlled trial. It enrolled 423 patients and randomised 1:1:1 to either PDT plus sham injection or to placebo PDT plus Ranibizumab (0.3 mg or 0.5 mg) monthly for 24 months. Fluorescein angiography was done every 3 months to determine the need for additional PDT or placebo PDT. Inclusion criteria were patients with predominantly classic CNV and VA of 6/12 6/96. Primary endpoint was proportion of subjects losing <15 ETDRS letters at 1 year and secondary endpoint was change in VA from baseline. Maintaining vision: Patients losing <15 letters at 1 year was 94% in 0.3 mg Ranibizumab group vs. 96% in 0.5mg Ranibizumab vs. 64% in PDT group (p < ). Improving vision: Patients gaining 15 letters was 36% in 0.3 mg Ranibizumab, 40% in 0.5 mg Ranibizumab and 6% in PDT (p < ). www. dosonline.org l 49
4 On average, 9 and 11 letters were gained with 0.3 mg and 0.5 mg Ranibizumab respectively vs. 10 letters lost in PDT group (p<0.0001). Severe visual loss was seen in 13% of PDT group versus none occurred in Ranibizumab groups. Leakage on FFA decreased by 2 disc areas (DA) in Ranibizumab group and increased by 1/3 DA in PDT group (p<0.0001). Ranibizumab provided greater clinical benefit than verteporfin PDT in patients with ARMD with new-onset, predominantly classic CNVM. Rates of serious adverse events were low. ProNTO (Prospective OCT Imaging of Patients with Neovascular AMD Treated with Intra-Ocular Ranibizumab) 9 Whether dosing on the basis of fluid in the macula results in fewer injections but has similar visual acuity outcomes to a monthly dosing regimen. 3 consecutive monthly injections of 0.5 mg Ranibizumab were given. Monitoring for anatomical and functional effects was done by OCT and FFA respectively. Injections were repeated only if required. After 1 year, the mean improvement in VA from baseline was 9.3 letters (p<0.001), and at 2 years it was 11.1 letters (p<0.001). Around35% eyes gained at least 15 letters of VA at 1 year and 43% gained at least 15 letters at 2 years. VA outcomes comparable with the outcomes from the phase III clinical studies, but fewer intravitreal injections were required. Ongoing studies with Ranibizumab: FOCUS, PIER, HORIZON, PROTECT etc. SANA trial- Systemic Avastin for neovascular AMD To evaluate the short-term safety of systemic bevacizumab and its effects on vision and subfoveal CNV in patients with neovascular AMD single-center, uncontrolled clinical study. AMD patients with subfoveal CNV (N = 9) and best-corrected VA letter scores of 70 to 20 (approximate Snellen equivalent, 20/40-20/400). Patients were treated at baseline with an infusion of bevacizumab (5 mg/kg), followed by 1 or 2 additional doses given at 2-week intervals. Safety assessments were performed at all visits. Conclusion: Significant improvement in VA and decreased retinal thickness (OCT) were seen. The systemic side effects led to the exploratory use of intravitreal injection of bevacizumab to treat CNV in AMD patients. Comparison of ARMD treatment trials (CATT) 10 Purpose To evaluate effects of Ranibizumab (Lucentis) and Bevacizumab (Avastin) when administered monthly or as needed for 2 years To describe the impact of switching to as needed treatment after 1 year of monthly treatment. CATT enrolled 1200 participants aged 50 and older with subfoveal CNV secondary to AMD in at least one eye and visual acuity between 20/40 and 20/320, inclusive. CATT participants completed a total of 24 monthly study visits for 2 years. All participants were assigned randomly to one of 4 treatment groups. 1) Lucentis on a fixed schedule of every 4 weeks for 1 year; at 1 year, re-randomization to Lucentis every 4 weeks or to variable dosing. 2) Avastin on a fixed schedule of every 4 weeks for 1 year; at 1 year, re-randomization to Avastin every 4 weeks or to variable dosing. 3) Lucentis on variable dosing for 2 years; i.e., after initial treatment, monthly evaluation for treatment based on signs of lesion activity. 4) Avastin on variable dosing for 2 years; i.e., after initial treatment, monthly evaluation for treatment based on signs of lesion activity. Those who were assigned to fixed monthly dosing groups would receive treatment at every visit. Those assigned to variable dosing groups were evaluated for treatment at every visit. If lesion activity was seen, the participant received a study treatment. In same regimen for 2 years, mean gain VA was similar for both groups (bevacizumab and Ranibizumab). The difference in gain of VA was 1.4 letters (p= 0.21, not significant.) Mean gain was greater for monthly than for as needed treatment (difference 2.4 letters, p=0.046) The proportion of patients with 1 or more systemic serious adverse events was higher with bevacizumab than Ranibizumab. Conclusion: Ranibizumab and bevacizumab had similar effects on VA over 2 year period. Treatment as needed resulted in less gain in VA, whether instituted at enrollment or after 1 year of monthly treatment. No differences between the 50 l DOS Times - Vol. 19, No. 3 September, 2013
5 two drugs in rates of death or arterio-thrombotic events was noted. VEGF Trap Eye in Phase 3 trial Investigate efficacy and safety of Aflibercept in wet AMD. Phase 1 and 2 trials showed that monthly or bimonthly Aflibercept was not inferior to monthly Ranibizumab in preventing vision loss (< 15 letter loss). The visual gain and safety was comparable with the two treatments. Year 2 treatment involved monthly pro re nata ( as needed) with required injections every 3 months and maintained vision gains from the first year, with an average of 4.2 injections of Aflibercept and 4.7 injections of Ranibizumab. References 1. AREDS Study Group. A randomized, placebo-controlled, clinical trial of high-dose supplementation with vitamins C and E, beta carotene, and zinc for age related macular degeneration and vision loss: AREDS report no. 8. Arch Ophthalmol. 2001; 119: Macular Photocoagulation Study Group. Subfoveal neovascular lesions in age-related macular degeneration. Guidelines for evaluation and treatment in the macular photocoagulation study. Arch Ophthalmol ; 109: Treatment of Age-related Macular Degeneration with Photodynamic Therapy (TAP) Study Group. Photodynamic therapy of subfoveal choroidal neovascularisation in age related macular degeneration with Verteprofin: one-year results of 2 randomized clinical trials: TAP report 1. Arch Ophthalmol. 1999;117: VIP Study Group. Verteprofin therapy of subfoveal choroidal neovascularisation in age-related macular degeneration: twoyear results of a randomized clinical trial including lesions with occult with no classic choroidal neovascularisation: Verteprofin in Photodynamic Therapy (VIP) report 2. Am J Ophthalmol. 2001; 131: Treatment of Age-Related Macular Degeneration with Photodynamic Therapy (TAP) Study Group. Photodynamic therapy of subfoveal choroidal neovascularisation in age-related macular degeneration with verteprofin: one-year results of 2 randomized clinical trials-tap report1. Arch Ophthalmol. 1999; 117: VEGF Inhibition Study in Ocular Neovascularization (V.I.S.I.O.N.) Clinical Trial Group. Year 2 efficacy results of 2 randomized controlled clinical trials of Pegaptanib for Neovascular Age-Related Macular Degeneration. Ophthalmology 2006 Sep; 113(9): Rosenfeld PJ, Brown DM, Heier JS, et al. MARINA Study Group. Ranibizumab for neovascular age-related macular degeneration. N Engl J Med Oct 5;355(14): Brown DM, Kaiser PK, Michels M, Soubrane G, et al. ANCHOR Study Group. Ranibizumab versus verteporfin for neovascular agerelated macular degeneration. N Engl J Med. 2006;355: Lalwani GA, Fung AE, Michels S, et al. An OCT-guided variabledosing regimen with Ranibizumab (Lucentis) in neovascular AMD: two year results of the PrONTO study. Poster presented at: Annual Meeting of the Association for Research in Vision and Ophthalmology; May 7, 2007; Fort Lauderdale, Fla. 10. The CATT Research Group. Ranibizumab and bevacizumab for treatment of neovascular age-related macular degeneration: 2-year results. Ophthalmology. Epub 2012 May 1. www. dosonline.org l 51
SUMMARY. Heather Casparis, MD,* and Neil M. Bressler, MD MARINA AND ANCHOR
 The following are summaries of selected presentations and posters from the American Society of Retina Specialists and European VitreoRetinal Society Annual Meeting held September 9 13, 2006, in Cannes,
The following are summaries of selected presentations and posters from the American Society of Retina Specialists and European VitreoRetinal Society Annual Meeting held September 9 13, 2006, in Cannes,
FEP Medical Policy Manual
 FEP Medical Policy Manual Last Review: September 2016 Next Review: September 2017 Related Policies 9.03.20 Intraocular Radiation Therapy for Age-Related Macular Degeneration Photodynamic Therapy for Choroidal
FEP Medical Policy Manual Last Review: September 2016 Next Review: September 2017 Related Policies 9.03.20 Intraocular Radiation Therapy for Age-Related Macular Degeneration Photodynamic Therapy for Choroidal
CENTENE PHARMACY AND THERAPEUTICS NEW DRUG REVIEW 2Q17 April May
 BRAND NAME Lucentis GENERIC NAME ranibizumab MANUFACTURER Genentech, Inc. DATE OF APPROVAL June 30, 2006 PRODUCT LAUNCH DATE July 13, 2006 REVIEW TYPE Review type 1 (RT1): New Drug Review Full review of
BRAND NAME Lucentis GENERIC NAME ranibizumab MANUFACTURER Genentech, Inc. DATE OF APPROVAL June 30, 2006 PRODUCT LAUNCH DATE July 13, 2006 REVIEW TYPE Review type 1 (RT1): New Drug Review Full review of
The limited number of currently approved
 CLINICAL TRIALS OF VERTEPORFIN AND PEGAPTANIB: WHAT ARE THE RESULTS? * William F. Mieler, MD ABSTRACT Currently available treatment options for the management of choroidal neovascularization (CNV) in age-related
CLINICAL TRIALS OF VERTEPORFIN AND PEGAPTANIB: WHAT ARE THE RESULTS? * William F. Mieler, MD ABSTRACT Currently available treatment options for the management of choroidal neovascularization (CNV) in age-related
Management of Neovascular AMD
 Kapusta AMD Part 1 Management of Neovascular AMD Dr. Michael A. Kapusta, MD, FRCSC Ophthalmologist in Chief Jewish General Hospital Vitreoretinal Surgeon 1 FINANCIAL DISCLOSURES Consulting honoraria Bayer,
Kapusta AMD Part 1 Management of Neovascular AMD Dr. Michael A. Kapusta, MD, FRCSC Ophthalmologist in Chief Jewish General Hospital Vitreoretinal Surgeon 1 FINANCIAL DISCLOSURES Consulting honoraria Bayer,
These issues are covered in more detail below.
 26.3.07 Comments from Novartis on the Assessment Report for the Health Technology Appraisal of Pegaptinib and Ranibizumab for the treatment of age-related macular degeneration In general we feel that the
26.3.07 Comments from Novartis on the Assessment Report for the Health Technology Appraisal of Pegaptinib and Ranibizumab for the treatment of age-related macular degeneration In general we feel that the
A Treat and Extend Regimen Using Ranibizumab for Neovascular Age-Related Macular Degeneration
 A Treat and Extend Regimen Using Ranibizumab for Neovascular Age-Related Macular Degeneration Clinical and Economic Impact Omesh P. Gupta, MD, MBA, Gary Shienbaum, MD, Avni H. Patel, MD, Christopher Fecarotta,
A Treat and Extend Regimen Using Ranibizumab for Neovascular Age-Related Macular Degeneration Clinical and Economic Impact Omesh P. Gupta, MD, MBA, Gary Shienbaum, MD, Avni H. Patel, MD, Christopher Fecarotta,
Despite our growing knowledge of the
 OVERVIEW OF AVAILABLE TREATMENTS FOR NEOVASCULAR AGE-RELATED MACULAR DEGENERATION * Carl D. Regillo, MD ABSTRACT Although potential treatments for neovascular age-related macular degeneration represent
OVERVIEW OF AVAILABLE TREATMENTS FOR NEOVASCULAR AGE-RELATED MACULAR DEGENERATION * Carl D. Regillo, MD ABSTRACT Although potential treatments for neovascular age-related macular degeneration represent
Ranibizumab and pegaptanib for the treatment of agerelated macular degeneration
 Ranibizumab and pegaptanib for the treatment of agerelated Issued: August 2008 last modified: May 2012 guidance.nice.org.uk/ta155 NHS Evidence has accredited the process used by the Centre for Health Technology
Ranibizumab and pegaptanib for the treatment of agerelated Issued: August 2008 last modified: May 2012 guidance.nice.org.uk/ta155 NHS Evidence has accredited the process used by the Centre for Health Technology
NEOVASCULAR AGE-RELATED MACULAR DEGENERation
 Randomized, Double-Masked, -Controlled Trial of Ranibizumab for Neovascular Age-Related Macular Degeneration: PIER Study Year 2 PREMA ABRAHAM, HUIBIN YUE, AND LAURA WILSON PURPOSE: To evaluate efficacy
Randomized, Double-Masked, -Controlled Trial of Ranibizumab for Neovascular Age-Related Macular Degeneration: PIER Study Year 2 PREMA ABRAHAM, HUIBIN YUE, AND LAURA WILSON PURPOSE: To evaluate efficacy
Clinical Study Fixed Monthly versus Less Frequent Ranibizumab Dosing and Predictors of Visual Response in Exudative Age-Related Macular Degeneration
 Ophthalmology Volume 12, Article ID 69641, 8 pages doi:1.1155/12/69641 Clinical Study Fixed Monthly versus Less Frequent Ranibizumab Dosing and Predictors of Visual Response in Exudative Age-Related Macular
Ophthalmology Volume 12, Article ID 69641, 8 pages doi:1.1155/12/69641 Clinical Study Fixed Monthly versus Less Frequent Ranibizumab Dosing and Predictors of Visual Response in Exudative Age-Related Macular
Description. Section: Other Effective Date: July 15, Subsection: Vision Original Policy Date: December 7, 2011 Subject: Page: 1 of 23
 Last Review Status/Date: June 2015 Page: 1 of 23 Description Photodynamic therapy (PDT) is a treatment modality designed to selectively occlude ocular choroidal neovascular tissue. The therapy is a 2-step
Last Review Status/Date: June 2015 Page: 1 of 23 Description Photodynamic therapy (PDT) is a treatment modality designed to selectively occlude ocular choroidal neovascular tissue. The therapy is a 2-step
VERTEPORFIN IN PHOTODYNAMIC THERAPY STUDY GROUP
 Verteporfin Therapy of Subfoveal Choroidal Neovascularization in Age-related Macular Degeneration: Two-year Results of a Randomized Clinical Trial Including Lesions With Occult With No Classic Choroidal
Verteporfin Therapy of Subfoveal Choroidal Neovascularization in Age-related Macular Degeneration: Two-year Results of a Randomized Clinical Trial Including Lesions With Occult With No Classic Choroidal
Photodynamic Therapy for Choroidal Neovascularization
 Photodynamic Therapy for Choroidal Neovascularization Policy Number: 9.03.08 Last Review: 10/2014 Origination: 10/2000 Next Review: 10/2015 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will
Photodynamic Therapy for Choroidal Neovascularization Policy Number: 9.03.08 Last Review: 10/2014 Origination: 10/2000 Next Review: 10/2015 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will
Instudies of vascular endothelial growth factor
 MONITORING THERAPEUTIC EFFICACY IN THE ANTI-VEGF ERA* SriniVas R. Sadda, MD ABSTRACT One of the most important questions with vascular endothelial growth factor (VEGF) inhibitors for age-related macular
MONITORING THERAPEUTIC EFFICACY IN THE ANTI-VEGF ERA* SriniVas R. Sadda, MD ABSTRACT One of the most important questions with vascular endothelial growth factor (VEGF) inhibitors for age-related macular
Technology appraisal guidance Published: 27 August 2008 nice.org.uk/guidance/ta155
 Ranibizumab and pegaptanib for the treatment of age-related macular degeneration Technology appraisal guidance Published: 27 August 2008 nice.org.uk/guidance/ta155 NICE 2018. All rights reserved. Subject
Ranibizumab and pegaptanib for the treatment of age-related macular degeneration Technology appraisal guidance Published: 27 August 2008 nice.org.uk/guidance/ta155 NICE 2018. All rights reserved. Subject
Anti-VEGF drugs & CATT Results. in AMD. Apollo Hospitals, Hyderabad. Mallika Goyal, MD
 Anti-VEGF drugs & CATT Results in AMD Mallika Goyal, MD Apollo Hospitals, Hyderabad Vascular Endothelial Growth Factor Napoleone Ferrara at Genentech 1989 identified & cloned the gene Protein that causes
Anti-VEGF drugs & CATT Results in AMD Mallika Goyal, MD Apollo Hospitals, Hyderabad Vascular Endothelial Growth Factor Napoleone Ferrara at Genentech 1989 identified & cloned the gene Protein that causes
ANTIVASCULAR ENDOTHELIAL GROWTH FACTOR
 THE ROLE OF RANIBIZUMAB * Michael L. Klein, MD ABSTRACT The positive 1-year results of ranibizumab trials reported at the 23rd Annual Meeting of the American Society of Retina Specialists have generated
THE ROLE OF RANIBIZUMAB * Michael L. Klein, MD ABSTRACT The positive 1-year results of ranibizumab trials reported at the 23rd Annual Meeting of the American Society of Retina Specialists have generated
Age-related macular degeneration (AMD) and RANIBIZUMAB (Lucentis)
 Aggiornamento biotecnologico: MoAb anti VEGF Age-related macular degeneration (AMD) and RANIBIZUMAB (Lucentis) Giulia Germena AMD DRY (non-neovascular) Atrophic cell death in the macula No leakage WET
Aggiornamento biotecnologico: MoAb anti VEGF Age-related macular degeneration (AMD) and RANIBIZUMAB (Lucentis) Giulia Germena AMD DRY (non-neovascular) Atrophic cell death in the macula No leakage WET
Anti VEGF Agents in Retinal Disorders Current Scenario
 Retina Anti VEGF Agents in Retinal Disorders Current Scenario Charu Gupta MS Charu Gupta MS, Cyrus M. Shroff MD Shroff Eye Centre, New Delhi T is a group of proteins involved in the regulation of angiogenesis,
Retina Anti VEGF Agents in Retinal Disorders Current Scenario Charu Gupta MS Charu Gupta MS, Cyrus M. Shroff MD Shroff Eye Centre, New Delhi T is a group of proteins involved in the regulation of angiogenesis,
Subgroup Analysis of the MARINA Study of Ranibizumab in Neovascular Age-Related Macular Degeneration
 Subgroup Analysis of the MARINA Study of in Neovascular Age-Related Macular Degeneration David S. Boyer, MD, 1 Andrew N. Antoszyk, MD, 2 Carl C. Awh, MD, 3 Robert B. Bhisitkul, MD, PhD, 4 Howard Shapiro,
Subgroup Analysis of the MARINA Study of in Neovascular Age-Related Macular Degeneration David S. Boyer, MD, 1 Andrew N. Antoszyk, MD, 2 Carl C. Awh, MD, 3 Robert B. Bhisitkul, MD, PhD, 4 Howard Shapiro,
CLINICAL SCIENCES. Verteporfin Therapy for Subfoveal Choroidal Neovascularization in Age-Related Macular Degeneration
 CLINICAL SCIENCES Verteporfin Therapy for Subfoveal Choroidal Neovascularization in Age-Related Macular Degeneration Three-Year Results of an Open-Label Extension of 2 Randomized Clinical Trials TAP Report
CLINICAL SCIENCES Verteporfin Therapy for Subfoveal Choroidal Neovascularization in Age-Related Macular Degeneration Three-Year Results of an Open-Label Extension of 2 Randomized Clinical Trials TAP Report
Risk factors of a reduced response to ranibizumab treatment for neovascular age-related macular degeneration evaluation in a clinical setting
 Korb et al. BMC Ophthalmology 2013, 13:84 RESEARCH ARTICLE Open Access Risk factors of a reduced response to ranibizumab treatment for neovascular age-related macular degeneration evaluation in a clinical
Korb et al. BMC Ophthalmology 2013, 13:84 RESEARCH ARTICLE Open Access Risk factors of a reduced response to ranibizumab treatment for neovascular age-related macular degeneration evaluation in a clinical
Coding Implications Revision Log. See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: Verteporfin (Visudyne) Reference Number: CP.PHAR.187 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important
Clinical Policy: Verteporfin (Visudyne) Reference Number: CP.PHAR.187 Effective Date: 03.16 Last Review Date: 02.18 Line of Business: Commercial, Medicaid Coding Implications Revision Log See Important
MEDICAL POLICY SUBJECT: PHOTODYNAMIC THERAPY FOR EFFECTIVE DATE: 07/20/00 SUBFOVEAL CHOROIDAL NEOVASCULARIZATION
 MEDICAL POLICY SUBJECT: PHOTODYNAMIC THERAPY FOR PAGE: 1 OF: 8 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
MEDICAL POLICY SUBJECT: PHOTODYNAMIC THERAPY FOR PAGE: 1 OF: 8 If a product excludes coverage for a service, it is not covered, and medical policy criteria do not apply. If a commercial product, including
Although photocoagulation and photodynamic PROCEEDINGS PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION *
 PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION Evangelos S. Gragoudas, MD ABSTRACT In December 24, the US Food and Drug Administration (FDA) approved pegaptanib sodium. Pegaptanib
PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION Evangelos S. Gragoudas, MD ABSTRACT In December 24, the US Food and Drug Administration (FDA) approved pegaptanib sodium. Pegaptanib
London Medicines Evaluation Network Review
 London Medicines Evaluation Network Review Evidence for initiating intravitreal bevacizumab for the management of wet age-related macular degeneration (wet-amd) in eyes with vision better than 6/12 November
London Medicines Evaluation Network Review Evidence for initiating intravitreal bevacizumab for the management of wet age-related macular degeneration (wet-amd) in eyes with vision better than 6/12 November
International Journal of Health Sciences and Research ISSN:
 International Journal of Health Sciences and Research www.ijhsr.org ISSN: 2249-9571 Original Research Article A Multivariate Analysis of Intravitreal Injection of Anti-VEGF Bevacizumab in the Treatment
International Journal of Health Sciences and Research www.ijhsr.org ISSN: 2249-9571 Original Research Article A Multivariate Analysis of Intravitreal Injection of Anti-VEGF Bevacizumab in the Treatment
Prevention and treatment of agerelated macular degeneration
 THERAPY REVIEW Prevention and treatment of agerelated macular degeneration SARAH HORTON AND CATHERINE GULY Age-related macular degeneration (AMD) is a common cause of visual loss in older people and GPs
THERAPY REVIEW Prevention and treatment of agerelated macular degeneration SARAH HORTON AND CATHERINE GULY Age-related macular degeneration (AMD) is a common cause of visual loss in older people and GPs
Retina Conference. Janelle Fassbender, MD, PhD University of Louisville Department of Ophthalmology and Visual Sciences 09/04/2014
 Retina Conference Janelle Fassbender, MD, PhD University of Louisville Department of Ophthalmology and Visual Sciences 09/04/2014 Subjective CC/HPI: 64 year old Caucasian female referred by outside ophthalmologist
Retina Conference Janelle Fassbender, MD, PhD University of Louisville Department of Ophthalmology and Visual Sciences 09/04/2014 Subjective CC/HPI: 64 year old Caucasian female referred by outside ophthalmologist
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists. International authors and editors
 We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 3,700 108,500 1.7 M Open access books available International authors and editors Downloads Our
We are IntechOpen, the world s leading publisher of Open Access books Built by scientists, for scientists 3,700 108,500 1.7 M Open access books available International authors and editors Downloads Our
Ophthalmology Macular Pathways
 Ophthalmology Macular Pathways Age related Macular Degeneration Diabetic Macular Oedema Macular Oedema secondary to Central Retinal Macular Oedema secondary to Branch Retinal CNV associated with pathological
Ophthalmology Macular Pathways Age related Macular Degeneration Diabetic Macular Oedema Macular Oedema secondary to Central Retinal Macular Oedema secondary to Branch Retinal CNV associated with pathological
Optimizing the Anti-VEGF Treatment Strategy for Neovascular Age-Related Macular Degeneration: From Clinical Trials to Real-Life Requirements
 Perspective DOI: 10.1167/tvst.4.3.6 Optimizing the Anti-VEGF Treatment Strategy for Neovascular Age-Related Macular Degeneration: From Clinical Trials to Real-Life Requirements Irmela Mantel 1,2 1 Department
Perspective DOI: 10.1167/tvst.4.3.6 Optimizing the Anti-VEGF Treatment Strategy for Neovascular Age-Related Macular Degeneration: From Clinical Trials to Real-Life Requirements Irmela Mantel 1,2 1 Department
FEP Medical Policy Manual
 FEP Medical Policy Manual Last Review: September 2016 Next Review: September 2017 Related Policies 6.01.10 Stereotactic Radiosurgery and Stereotactic Body Radiotherapy 8.01.10 Charged-Particle (Proton
FEP Medical Policy Manual Last Review: September 2016 Next Review: September 2017 Related Policies 6.01.10 Stereotactic Radiosurgery and Stereotactic Body Radiotherapy 8.01.10 Charged-Particle (Proton
Effectiveness of Ranibizumab for Neovascular Age-related Macular Degeneration using Clinician Determined Retreatment Strategy
 Effectiveness of Ranibizumab for Neovascular Age-related Macular Degeneration using Clinician Determined Retreatment Strategy Anupma Kumar, Jayashree N Sahni, Alexandros N Stangos, Claudio Campa, Simon
Effectiveness of Ranibizumab for Neovascular Age-related Macular Degeneration using Clinician Determined Retreatment Strategy Anupma Kumar, Jayashree N Sahni, Alexandros N Stangos, Claudio Campa, Simon
There are no published randomized, double-blind trials comparing aflibercept to other therapies in neovascular AMD.
 Subject: Eylea (aflibercept) Original Effective Date: 7/11/2014 Policy Number: MCP-191 Revision Date(s): 10/11/2016 Review Date(s): 12/16/2015; 10/11/2016, 6/22/2017, 7/10/2018 DISCLAIMER This Medical
Subject: Eylea (aflibercept) Original Effective Date: 7/11/2014 Policy Number: MCP-191 Revision Date(s): 10/11/2016 Review Date(s): 12/16/2015; 10/11/2016, 6/22/2017, 7/10/2018 DISCLAIMER This Medical
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE GUIDANCE EXECUTIVE (GE)
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE GUIDANCE EXECUTIVE (GE) Review of TA68; The clinical and cost effectiveness of photodynamic therapy for age-related macular degeneration TA68 guidance
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE GUIDANCE EXECUTIVE (GE) Review of TA68; The clinical and cost effectiveness of photodynamic therapy for age-related macular degeneration TA68 guidance
Computerized Model of Cost-Utility Analysis for Treatment of Age-Related Macular Degeneration
 Computerized Model of Cost-Utility Analysis for Treatment of Age-Related Macular Degeneration E. C. Fletcher, MRCOphth, 1 R. J. Lade, PhD, MBA, 2 T. Adewoyin, MRCOphth, 1 N. V. Chong, FRCOphth, MD 1 Purpose:
Computerized Model of Cost-Utility Analysis for Treatment of Age-Related Macular Degeneration E. C. Fletcher, MRCOphth, 1 R. J. Lade, PhD, MBA, 2 T. Adewoyin, MRCOphth, 1 N. V. Chong, FRCOphth, MD 1 Purpose:
CLINICAL STUDY. S Borooah 1, VS Jeganathan 1, A-M Ambrecht 1, D Oladiwura 2, M Gavin 2, B Dhillon 1 and P Cackett 1
 (2015) 29, 1156 1161 2015 Macmillan Publishers Limited All rights reserved 0950-222X/15 www.nature.com/eye CLINICAL STUDY Long-term visual outcomes of intravitreal ranibizumab treatment for wet age-related
(2015) 29, 1156 1161 2015 Macmillan Publishers Limited All rights reserved 0950-222X/15 www.nature.com/eye CLINICAL STUDY Long-term visual outcomes of intravitreal ranibizumab treatment for wet age-related
Long-term Management of AMD. Motasem Al-latayfeh, MD Assistant Prof. Ophthalmology Hashemite University Jordan
 Long-term Management of AMD Motasem Al-latayfeh, MD Assistant Prof. Ophthalmology Hashemite University Jordan DEFINITION 1 Age-related macular degeneration (AMD) is a disorder of the macula characterized
Long-term Management of AMD Motasem Al-latayfeh, MD Assistant Prof. Ophthalmology Hashemite University Jordan DEFINITION 1 Age-related macular degeneration (AMD) is a disorder of the macula characterized
Description. Section: Other Effective Date: July 15, Subsection: Vision Original Policy Date: September 13, 2011 Subject: Page: 1 of 18
 Last Review Status/Date: June 2014 Page: 1 of 18 Description Angiogenesis inhibitors (e.g., ranibizumab, bevacizumab, pegaptanib, aflibercept) are being evaluated for the treatment of disorders of choroidal
Last Review Status/Date: June 2014 Page: 1 of 18 Description Angiogenesis inhibitors (e.g., ranibizumab, bevacizumab, pegaptanib, aflibercept) are being evaluated for the treatment of disorders of choroidal
Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial
 ASX and Media Release 3 January 2018 Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial Melbourne, Australia; January 3 2018 Opthea Limited (ASX:OPT), a late stage biopharmaceutical company
ASX and Media Release 3 January 2018 Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial Melbourne, Australia; January 3 2018 Opthea Limited (ASX:OPT), a late stage biopharmaceutical company
Macular Degeneration. John Christoforidis, MD Susie Chang, MD. Assistant Professors Department of Ophthalmology Ohio State University
 Macular Degeneration John Christoforidis, MD Susie Chang, MD Assistant Professors Department of Ophthalmology Ohio State University 1. Retina 2. Optic Chiasm 3. LGN 4. Temporal lobe Courtesy of Ben Glasgow,
Macular Degeneration John Christoforidis, MD Susie Chang, MD Assistant Professors Department of Ophthalmology Ohio State University 1. Retina 2. Optic Chiasm 3. LGN 4. Temporal lobe Courtesy of Ben Glasgow,
The Foundation WHAT IS THE RETINA?
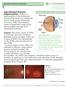 Age-Related Macular Degeneration (AMD) is a deterioration of the retina and choroid that leads to a substantial loss in visual acuity (sharpness of vision). AMD is the leading cause of significant visual
Age-Related Macular Degeneration (AMD) is a deterioration of the retina and choroid that leads to a substantial loss in visual acuity (sharpness of vision). AMD is the leading cause of significant visual
MONTHLY ADMINISTERED INTRAVITREOUS PHARmacotherapy
 Polypoidal Choroidal Vasculopathy Masquerading as Neovascular Age-Related Macular Degeneration Refractory to Ranibizumab ALEXANDROS N. STANGOS, JAGDEEP SINGH GANDHI, JAYASHREE NAIR-SAHNI, HEINRICH HEIMANN,
Polypoidal Choroidal Vasculopathy Masquerading as Neovascular Age-Related Macular Degeneration Refractory to Ranibizumab ALEXANDROS N. STANGOS, JAGDEEP SINGH GANDHI, JAYASHREE NAIR-SAHNI, HEINRICH HEIMANN,
Optical Coherence Tomography Monitoring Strategies for A-VEGF Treated Age-Related Macular Degeneration: An Evidence-Based Analysis
 Optical Coherence Tomography Monitoring Strategies for A-VEGF Treated Age-Related Macular Degeneration: An Evidence-Based Analysis G Pron August 2014 Ontario Health Technology Assessment Series; Vol. 14:
Optical Coherence Tomography Monitoring Strategies for A-VEGF Treated Age-Related Macular Degeneration: An Evidence-Based Analysis G Pron August 2014 Ontario Health Technology Assessment Series; Vol. 14:
Angiogenesis and Role of Anti-VEGF Therapy
 Review Article Angiogenesis and Role of Anti-VEGF Therapy P.S. Mahar, Azfar N. Hanfi, Aimal Khan Pak J Ophthalmol 2009, Vol. 25 No. 3.....................................................................................................
Review Article Angiogenesis and Role of Anti-VEGF Therapy P.S. Mahar, Azfar N. Hanfi, Aimal Khan Pak J Ophthalmol 2009, Vol. 25 No. 3.....................................................................................................
Aflibercept in wet age-related macular degeneration: a perspective review
 446007TAJ342040622312446007M Ohr and PK KaiserTherapeutic Advances in Chronic Disease 2012 Therapeutic Advances in Chronic Disease Review ibercept in wet age-related macular degeneration: a perspective
446007TAJ342040622312446007M Ohr and PK KaiserTherapeutic Advances in Chronic Disease 2012 Therapeutic Advances in Chronic Disease Review ibercept in wet age-related macular degeneration: a perspective
CASE STUDIES CURRENT AND FUTURE TREATMENT OPTIONS FOR NEOVASCULAR AGE-RELATED MACULAR DEGENERATION * Discussion led by Neil M.
 CURRENT AND FUTURE TREATMENT OPTIONS FOR NEOVASCULAR AGE-RELATED MACULAR DEGENERATION * Discussion led by Neil M. Bressler, MD Dr Bressler: The overview articles in this monograph summarize safety, efficacy,
CURRENT AND FUTURE TREATMENT OPTIONS FOR NEOVASCULAR AGE-RELATED MACULAR DEGENERATION * Discussion led by Neil M. Bressler, MD Dr Bressler: The overview articles in this monograph summarize safety, efficacy,
Digital Journal of Ophthalmology Digital Journal of Ophthalmology. Abstract
 Original Articles Combined intravitreal ranibizumab and verteporfin photodynamic therapy versus ranibizumab alone for the treatment of age-related macular degeneration Rosalia Giustolisi, MD, Nicoletta
Original Articles Combined intravitreal ranibizumab and verteporfin photodynamic therapy versus ranibizumab alone for the treatment of age-related macular degeneration Rosalia Giustolisi, MD, Nicoletta
The Natural History of Occult Choroidal Neovascularisation Associated With Age-related Macular Degeneration. A Systematic Review
 Review Article 145 The Natural History of Occult Choroidal Neovascularisation Associated With Age-related Macular Degeneration. A Systematic Review Antonio Polito, 1 MD, Miriam Isola, 2 MHS, Paolo Lanzetta,
Review Article 145 The Natural History of Occult Choroidal Neovascularisation Associated With Age-related Macular Degeneration. A Systematic Review Antonio Polito, 1 MD, Miriam Isola, 2 MHS, Paolo Lanzetta,
2/1/2017. Agenda. David F. Williams, MD, MBA. History of Current Treatment. Current Treatment/Wet AMD Anti-VEGF Drugs. History of Current Treatment
 UPDATE ON CLINICAL TRIALS FOR AMD David F. Williams, MD, MBA Agenda Brief History of Current Rx Recently Completed/New CTs: Wet AMD Recently Completed/New CTs: Dry AMD Current Treatment/Wet AMD Anti-VEGF
UPDATE ON CLINICAL TRIALS FOR AMD David F. Williams, MD, MBA Agenda Brief History of Current Rx Recently Completed/New CTs: Wet AMD Recently Completed/New CTs: Dry AMD Current Treatment/Wet AMD Anti-VEGF
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Efficacy of Anti-VEGF Agents in the Treatment of Age-Related Macular Degeneration
 Efficacy of Anti-VEGF Agents in the Treatment of Age-Related Macular Degeneration Marilita M. Moschos Abstract- Purpose: To evaluate by OCT and mf-erg the macular function in eyes with CNV due to ARMD
Efficacy of Anti-VEGF Agents in the Treatment of Age-Related Macular Degeneration Marilita M. Moschos Abstract- Purpose: To evaluate by OCT and mf-erg the macular function in eyes with CNV due to ARMD
Evolving European guidance on the medical management of neovascular age related macular degeneration
 Evolving European guidance on the medical management of neovascular age related macular degeneration U Chakravarthy, G Soubrane, F Bandello, V Chong, C Creuzot-Garcher, S A Dimitrakos, II, J-F Korobelnik,
Evolving European guidance on the medical management of neovascular age related macular degeneration U Chakravarthy, G Soubrane, F Bandello, V Chong, C Creuzot-Garcher, S A Dimitrakos, II, J-F Korobelnik,
Name of Policy: Bevacizumab, Avastin
 Name of Policy: Bevacizumab, Avastin Policy #: 377 Latest Review Date: September 2013 Category: Pharmacology Policy Grade: B Background/Definitions: As a general rule, benefits are payable under Blue Cross
Name of Policy: Bevacizumab, Avastin Policy #: 377 Latest Review Date: September 2013 Category: Pharmacology Policy Grade: B Background/Definitions: As a general rule, benefits are payable under Blue Cross
Treat and Extend Dosing Regimen with Anti-vascular Endothelial Growth Factor Agents for Neovascular Age-related Macular Degeneration
 REVIEW ARTICLE Treat and Extend Dosing Regimen with Anti-vascular Endothelial Growth Factor Agents for Neovascular Age-related Macular Degeneration Karen M. Wai, Rishi P. Singh Department of Ophthalmology,
REVIEW ARTICLE Treat and Extend Dosing Regimen with Anti-vascular Endothelial Growth Factor Agents for Neovascular Age-related Macular Degeneration Karen M. Wai, Rishi P. Singh Department of Ophthalmology,
Systemic bevacizumab (Avastin) therapy for exudative neovascular age-related macular degeneration. The BEAT-AMD-Study
 1 Department of Ophthalmology, The Ludwig Boltzmann Institute for Retinology and Biomicroscopic Lasersurgery, Rudolf Foundation Clinic, Vienna, Austria; 2 Department of Internal Medicine, Oncology, Rudolf
1 Department of Ophthalmology, The Ludwig Boltzmann Institute for Retinology and Biomicroscopic Lasersurgery, Rudolf Foundation Clinic, Vienna, Austria; 2 Department of Internal Medicine, Oncology, Rudolf
Age-related Macular Degeneration (AMD) Update. Michael W. Stewart, MD Professor and Chairman Department of Ophthalmology Mayo Clinic Florida
 Age-related Macular Degeneration (AMD) Update Michael W. Stewart, MD Professor and Chairman Department of Ophthalmology Mayo Clinic Florida Disclosure Alkahest Consultant Allergan Research Support Bayer
Age-related Macular Degeneration (AMD) Update Michael W. Stewart, MD Professor and Chairman Department of Ophthalmology Mayo Clinic Florida Disclosure Alkahest Consultant Allergan Research Support Bayer
Submission from the Royal College of Ophthalmologists. the National Institute for Health and Clinical Excellence. Ranibizumab and pegaptanib
 Submission from the Royal College of Ophthalmologists to the National Institute for Health and Clinical Excellence. Health Technology Appraisal Ranibizumab and pegaptanib for the treatment of Age related
Submission from the Royal College of Ophthalmologists to the National Institute for Health and Clinical Excellence. Health Technology Appraisal Ranibizumab and pegaptanib for the treatment of Age related
From Outdated to Updated: A Review of Important Clinical Trials in Ocular Disease from 2014
 From Outdated to Updated: A Review of Important Clinical Trials in Ocular Disease from 2014 1. This course is designed to review the important ophthalmic literature that was released between October 2013
From Outdated to Updated: A Review of Important Clinical Trials in Ocular Disease from 2014 1. This course is designed to review the important ophthalmic literature that was released between October 2013
Intraocular Radiation Therapy for Age-Related Macular Degeneration
 Medical Policy Manual Medicine, Policy No. 134 Intraocular Radiation Therapy for Age-Related Macular Degeneration Next Review: April 2019 Last Review: June 2018 Effective: August 1, 2018 IMPORTANT REMINDER
Medical Policy Manual Medicine, Policy No. 134 Intraocular Radiation Therapy for Age-Related Macular Degeneration Next Review: April 2019 Last Review: June 2018 Effective: August 1, 2018 IMPORTANT REMINDER
Retinal Pigment Epithelial Tears (Rips) in the ERA of Anti Vegf - When and Why?
 Original Article Retinal Pigment Epithelial Tears (Rips) in the ERA of Anti Vegf - When and Why? Dr Sreelekha S. MS, DNB, DO, FRCS, Dr Manoj Soman DNB, FICO, FRCS, Dr. Unni Nair MS, FRCS, Dr. Ruminder
Original Article Retinal Pigment Epithelial Tears (Rips) in the ERA of Anti Vegf - When and Why? Dr Sreelekha S. MS, DNB, DO, FRCS, Dr Manoj Soman DNB, FICO, FRCS, Dr. Unni Nair MS, FRCS, Dr. Ruminder
P. Stavrakas MD PhD. Consultant Ophthalmologist and vitreoretinal surgeon 2 nd Department of Ophthalmology University of Athens
 P. Stavrakas MD PhD Consultant Ophthalmologist and vitreoretinal surgeon 2 nd Department of Ophthalmology University of Athens Primary diagnose of exudative AMD Anti-VEGF monotherapy treatment Loss of
P. Stavrakas MD PhD Consultant Ophthalmologist and vitreoretinal surgeon 2 nd Department of Ophthalmology University of Athens Primary diagnose of exudative AMD Anti-VEGF monotherapy treatment Loss of
THE ROLE OF anti-vegf IN DIABETIC RETINOPATHY AND AGE RELATED MACULAR DEGENERATION
 THE ROLE OF anti-vegf IN DIABETIC RETINOPATHY AND AGE RELATED MACULAR DEGENERATION MOESTIDJAB DEPARTMENT OF OPHTHALMOLOGY SCHOOL OF MEDICINE AIRLANGGA UNIVERSITY DR SOETOMO HOSPITAL SURABAYA INTRODUCTION
THE ROLE OF anti-vegf IN DIABETIC RETINOPATHY AND AGE RELATED MACULAR DEGENERATION MOESTIDJAB DEPARTMENT OF OPHTHALMOLOGY SCHOOL OF MEDICINE AIRLANGGA UNIVERSITY DR SOETOMO HOSPITAL SURABAYA INTRODUCTION
Dr Dianne Sharp Ophthalmologist Retina Specialists, Parnell Greenlane Clinical Centre
 Dr Dianne Sharp Ophthalmologist Retina Specialists, Parnell Greenlane Clinical Centre 11:00-11:55 WS #115: The Revolution in Macular Degeneration Management 12:05-13:00 WS #127: The Revolution in Macular
Dr Dianne Sharp Ophthalmologist Retina Specialists, Parnell Greenlane Clinical Centre 11:00-11:55 WS #115: The Revolution in Macular Degeneration Management 12:05-13:00 WS #127: The Revolution in Macular
Retinal pigment epithelial tears after intravitreal bevacizumab injection for exudative age-related macular degeneration.
 Thomas Jefferson University Jefferson Digital Commons Wills Eye Institute Papers Wills Eye Institute 4-1-2008 Retinal pigment epithelial tears after intravitreal bevacizumab injection for exudative age-related
Thomas Jefferson University Jefferson Digital Commons Wills Eye Institute Papers Wills Eye Institute 4-1-2008 Retinal pigment epithelial tears after intravitreal bevacizumab injection for exudative age-related
Outcomes of Anti-VEGF Therapy for Neovascular Age-Related Macular Degeneration in Routine Clinical Practice
 Outcomes of Anti-VEGF Therapy for Neovascular Age-Related Macular Degeneration in Routine Clinical Practice Andrew A. Moshfeghi, MD, MBA 1 ; John D. Pitcher, MD 2,3 ; Genevieve Lucas, B. Comm. 4 ; Nick
Outcomes of Anti-VEGF Therapy for Neovascular Age-Related Macular Degeneration in Routine Clinical Practice Andrew A. Moshfeghi, MD, MBA 1 ; John D. Pitcher, MD 2,3 ; Genevieve Lucas, B. Comm. 4 ; Nick
Drug Class Update: Vascular Endothelial Growth Factors
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
CDR. Ranibizumab Lucentis Novartis Pharmaceuticals Canada Inc. Overview of CDR Clinical and Pharmacoeconomic Reports
 Overview of CDR Clinical and Pharmacoeconomic Reports CDR August 2008 Ranibizumab Lucentis Novartis Pharmaceuticals Canada Inc. Indication Age-related Macular Degeneration (AMD) Cite as: Common Drug Review.
Overview of CDR Clinical and Pharmacoeconomic Reports CDR August 2008 Ranibizumab Lucentis Novartis Pharmaceuticals Canada Inc. Indication Age-related Macular Degeneration (AMD) Cite as: Common Drug Review.
Good News for The Macular Generation. Dr Andrew Thompson FRANZCO
 Good News for The Macular Generation Dr Andrew Thompson FRANZCO Anti-VEGFs for wet MD are one of the greatest advances in medicine in past decade Prof. Gary Brown Director Wills Eye Institue Retina Service
Good News for The Macular Generation Dr Andrew Thompson FRANZCO Anti-VEGFs for wet MD are one of the greatest advances in medicine in past decade Prof. Gary Brown Director Wills Eye Institue Retina Service
Diabetic Retinopathy: Recent Advances in Treatment and Treatment Approaches
 Diabetic Retinopathy: Recent Advances in Treatment and Treatment Approaches Dr. David Wong Associate Professor Retina Specialist, Department of Ophthalmology & Vision Sciences, University of Toronto, Canada
Diabetic Retinopathy: Recent Advances in Treatment and Treatment Approaches Dr. David Wong Associate Professor Retina Specialist, Department of Ophthalmology & Vision Sciences, University of Toronto, Canada
Review Article Anti-VEGF Therapy and the Retina: An Update
 Journal of Ophthalmology Volume 2015, Article ID 627674, 13 pages http://dx.doi.org/10.1155/2015/627674 Review Article Anti-VEGF Therapy and the Retina: An Update Vikas Tah, 1 Harry O. Orlans, 2 Jonathan
Journal of Ophthalmology Volume 2015, Article ID 627674, 13 pages http://dx.doi.org/10.1155/2015/627674 Review Article Anti-VEGF Therapy and the Retina: An Update Vikas Tah, 1 Harry O. Orlans, 2 Jonathan
Section 21, Ophthalmological Preparations. Ranibizumab (Lucentis ) Addition
 Application for inclusion in the WHO Essential Medicines List Section 21, Ophthalmological Preparations Ranibizumab (Lucentis ) Addition Document type: Document status: Application for inclusion in WHO
Application for inclusion in the WHO Essential Medicines List Section 21, Ophthalmological Preparations Ranibizumab (Lucentis ) Addition Document type: Document status: Application for inclusion in WHO
Five-year Outcomes of Ranibizumab in Neovascular Age-related Macular Degeneration: Real Life Clinical Experience
 pissn: 1011-8942 eissn: 2092-9382 Korean J Ophthalmol 2017;31(5):424-430 https://doi.org/10.3341/kjo.2016.0125 Original Article Five-year Outcomes of Ranibizumab in Neovascular Age-related Macular Degeneration:
pissn: 1011-8942 eissn: 2092-9382 Korean J Ophthalmol 2017;31(5):424-430 https://doi.org/10.3341/kjo.2016.0125 Original Article Five-year Outcomes of Ranibizumab in Neovascular Age-related Macular Degeneration:
Intraocular Radiotherapy for Age- Related Macular Degeneration
 Intraocular Radiotherapy for Age- Related Macular Degeneration Policy Number: 9.03.20 Last Review: 9/2017 Origination: 9/2008 Next Review: 9/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue
Intraocular Radiotherapy for Age- Related Macular Degeneration Policy Number: 9.03.20 Last Review: 9/2017 Origination: 9/2008 Next Review: 9/2018 Policy Blue Cross and Blue Shield of Kansas City (Blue
Clinical Study Effects of Intravitreal Ranibizumab Injection on Chinese Patients with Wet Age-Related Macular Degeneration: 5-Year Follow-Up Results
 Ophthalmology Volume 2016, Article ID 6538192, 5 pages http://dx.doi.org/10.1155/2016/6538192 Clinical Study Effects of Intravitreal Ranibizumab Injection on Chinese Patients with Wet Age-Related Macular
Ophthalmology Volume 2016, Article ID 6538192, 5 pages http://dx.doi.org/10.1155/2016/6538192 Clinical Study Effects of Intravitreal Ranibizumab Injection on Chinese Patients with Wet Age-Related Macular
Persistent Macular Thickening After Ranibizumab Treatment for Diabetic Macular Edema With Vision Impairment
 9:30 AM Persistent Macular Thickening After Ranibizumab Treatment for Diabetic Macular Edema With Vision Impairment Lee Jampol, MD OBJECTIVE To assess subsequent visual and anatomic outcomes of eyes with
9:30 AM Persistent Macular Thickening After Ranibizumab Treatment for Diabetic Macular Edema With Vision Impairment Lee Jampol, MD OBJECTIVE To assess subsequent visual and anatomic outcomes of eyes with
Clinical Policy: Verteporfin (Visudyne) Reference Number: CP.PHAR.187
 Clinical Policy: (Visudyne) Reference Number: CP.PHAR.187 Effective Date: 03/16 Last Review Date: 03/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: (Visudyne) Reference Number: CP.PHAR.187 Effective Date: 03/16 Last Review Date: 03/17 Coding Implications Revision Log See Important Reminder at the end of this policy for important regulatory
Intravitreal ranibizumab and bevacizumab for the treatment of nonsubfoveal choroidal neovascularization in age-related macular degeneration
 Intravitreal ranibizumab and bevacizumab for the treatment of nonsubfoveal choroidal neovascularization in age-related macular degeneration Ranibizumab e bevacizumab intravítreo no tratamento da neovascularização
Intravitreal ranibizumab and bevacizumab for the treatment of nonsubfoveal choroidal neovascularization in age-related macular degeneration Ranibizumab e bevacizumab intravítreo no tratamento da neovascularização
Short-term outcomes of switching anti-vegf agents in eyes with treatment-resistant wet AMD
 Batioglu et al. BMC Ophthalmology (2015) 15:40 DOI 10.1186/s12886-015-0025-z RESEARCH ARTICLE Open Access Short-term outcomes of switching anti-vegf agents in eyes with treatment-resistant wet AMD Figen
Batioglu et al. BMC Ophthalmology (2015) 15:40 DOI 10.1186/s12886-015-0025-z RESEARCH ARTICLE Open Access Short-term outcomes of switching anti-vegf agents in eyes with treatment-resistant wet AMD Figen
1.1 Definition of intervention
 1.1 Definition of intervention In Australia vision loss is predominantly a problem of ageing, the presence of which can significantly impact on the quality of life of older Australians. The prevalence
1.1 Definition of intervention In Australia vision loss is predominantly a problem of ageing, the presence of which can significantly impact on the quality of life of older Australians. The prevalence
The Era of anti- - - VEGF Kirk L. Halvorson, OD
 The Era of anti- - - VEGF Kirk L. Halvorson, OD Introduction: Anti- - - Vascular Endothelial Growth Factor (Anti- - - VEGF) medication is a relatively a new line of medications used in treating a variety
The Era of anti- - - VEGF Kirk L. Halvorson, OD Introduction: Anti- - - Vascular Endothelial Growth Factor (Anti- - - VEGF) medication is a relatively a new line of medications used in treating a variety
A Little Physics. How Does It Work? Radiation Therapy for Choroidal Neovascularisation in AMD A Review. => cell death
 Radiation Therapy for Choroidal Neovascularisation in AMD A Review Jen Anikina ST4 Hillingdon Hospital A Little Physics One gray is the absorption of one joule of ionizing radiation energy per kilogram
Radiation Therapy for Choroidal Neovascularisation in AMD A Review Jen Anikina ST4 Hillingdon Hospital A Little Physics One gray is the absorption of one joule of ionizing radiation energy per kilogram
LUCENTIS ranibizumab (rbe)
 1 LUCENTIS ranibizumab (rbe) NAME OF THE MEDICINE Active ingredient: Ranibizumab Chemical name: Immunoglobulin G1, anti-(human vascular endothelial growth factor) Fab fragment (human-mouse monoclonal rhufab
1 LUCENTIS ranibizumab (rbe) NAME OF THE MEDICINE Active ingredient: Ranibizumab Chemical name: Immunoglobulin G1, anti-(human vascular endothelial growth factor) Fab fragment (human-mouse monoclonal rhufab
TREATMENT STRATEGIES FOR CHORIORETINAL VASCULAR DISEASES: ADVANTAGES AND DISADVANTAGES OF INDIVIDUALISED THERAPY
 TREATMENT STRATEGIES FOR CHORIORETINAL VASCULAR DISEASES: ADVANTAGES AND DISADVANTAGES OF INDIVIDUALISED THERAPY *Michael W. Stewart Professor and Chairman, Mayo School of Medicine, Department of Ophthalmology,
TREATMENT STRATEGIES FOR CHORIORETINAL VASCULAR DISEASES: ADVANTAGES AND DISADVANTAGES OF INDIVIDUALISED THERAPY *Michael W. Stewart Professor and Chairman, Mayo School of Medicine, Department of Ophthalmology,
Comparison of Age-related Macular Degeneration Treatments Trials. CATT: Lucentis - Avastin Trial. Manual of Procedures
 Comparison of Age-related Macular Degeneration Treatments Trials CATT: Lucentis - Avastin Trial Manual of Procedures January 2011 COMPARISON OF AGE-RELATED MACULAR DEGENERATION TREATMENTS TRIALS (CATT)
Comparison of Age-related Macular Degeneration Treatments Trials CATT: Lucentis - Avastin Trial Manual of Procedures January 2011 COMPARISON OF AGE-RELATED MACULAR DEGENERATION TREATMENTS TRIALS (CATT)
VEGF Trap-Eye for the treatment of neovascular age-related macular degeneration
 Drug Evaluation 1 4 5 6 7 8 9 10 11 1 1 14 15 16 17 18 19 0 1 4 5 6 7 8 9 0 1 4 5 6 7 8 9 40 41 4 4 44 45 46 47 48 49 50 51 5 5 54 1. Introduction. Background. Conclusion 4. Expert opinion VEGF Trap-Eye
Drug Evaluation 1 4 5 6 7 8 9 10 11 1 1 14 15 16 17 18 19 0 1 4 5 6 7 8 9 0 1 4 5 6 7 8 9 40 41 4 4 44 45 46 47 48 49 50 51 5 5 54 1. Introduction. Background. Conclusion 4. Expert opinion VEGF Trap-Eye
DATE: 11 March 2014 CONTEXT AND POLICY ISSUES
 TITLE: Long-Term Use of Ranibizumab for the Treatment of Age-Related Macular Degeneration: A Review of the Clinical and Cost-Effectiveness and Guidelines DATE: 11 March 2014 CONTEXT AND POLICY ISSUES Age-related
TITLE: Long-Term Use of Ranibizumab for the Treatment of Age-Related Macular Degeneration: A Review of the Clinical and Cost-Effectiveness and Guidelines DATE: 11 March 2014 CONTEXT AND POLICY ISSUES Age-related
Digital Journal of Ophthalmology, Vol. 21 Digital Journal of Ophthalmology, Vol. 21
 Original Article Feasibility and efficacy of a mass switch from ranibizumab (Lucentis) to bevacizumab (Avastin) for treatment of neovascular age-related macular degeneration Michael T. Andreoli, MD, a,b
Original Article Feasibility and efficacy of a mass switch from ranibizumab (Lucentis) to bevacizumab (Avastin) for treatment of neovascular age-related macular degeneration Michael T. Andreoli, MD, a,b
Stabilization of visual acuity with photodynamic therapy in eyes with chorioretinal anastomoses
 Graefe s Arch Clin Exp Ophthalmol (2004) 242:368 376 CLINICAL INVESTIGATION DOI 10.1007/s00417-003-0844-0 Rufino M. Silva José R. Faria de Abreu António Travassos José G. Cunha-Vaz Stabilization of visual
Graefe s Arch Clin Exp Ophthalmol (2004) 242:368 376 CLINICAL INVESTIGATION DOI 10.1007/s00417-003-0844-0 Rufino M. Silva José R. Faria de Abreu António Travassos José G. Cunha-Vaz Stabilization of visual
Opinion 4 December 2013
 The legally binding text is the original French version TRANSPARENCY COMMITTEE Opinion 4 December 2013 LUCENTIS 10 mg/ml, solution for injection Vial of 0.23 ml (CIP: 34009 378 101 5 9) Applicant: Novartis
The legally binding text is the original French version TRANSPARENCY COMMITTEE Opinion 4 December 2013 LUCENTIS 10 mg/ml, solution for injection Vial of 0.23 ml (CIP: 34009 378 101 5 9) Applicant: Novartis
Ranibizumab and Bevacizumab for Treatment of Neovascular Age-related Macular Degeneration
 and for Treatment of Neovascular Age-related Macular Degeneration Two-Year Results Comparison of Age-related Macular Degeneration Treatments Trials (CATT) Research Group* Writing Committee: Daniel F. Martin,
and for Treatment of Neovascular Age-related Macular Degeneration Two-Year Results Comparison of Age-related Macular Degeneration Treatments Trials (CATT) Research Group* Writing Committee: Daniel F. Martin,
Growth Conference. October 19, May 2013
 Dawson Corporate James Securities Presentation 3rd Annual Small Cap Growth Conference October 19, 2017 May 2013 Safe Harbor This document contains forward-looking statements within the meaning of the "safe
Dawson Corporate James Securities Presentation 3rd Annual Small Cap Growth Conference October 19, 2017 May 2013 Safe Harbor This document contains forward-looking statements within the meaning of the "safe
A prospective nonrandomized clinical study on exudative age related macular degeneration
 Original Article A prospective nonrandomized clinical study on exudative age related macular degeneration Ayakutty Muni Raja, Siddharam Janti, Charanya Chendilnathan, Adnan Matheen Department of Ophthalmology,
Original Article A prospective nonrandomized clinical study on exudative age related macular degeneration Ayakutty Muni Raja, Siddharam Janti, Charanya Chendilnathan, Adnan Matheen Department of Ophthalmology,
AGE RELATED MACULAR DEGENERATION (AMD) PREFERRED PRACTICE PATTERNS (PPP) Philippines: 2016
 AGE RELATED MACULAR DEGENERATION (AMD) PREFERRED PRACTICE PATTERNS (PPP) Philippines: 2016 The Age Related Macular Degeneration (AMD) Preferred Practice Patterns (PPP) Philippines: 2016 was prepared by
AGE RELATED MACULAR DEGENERATION (AMD) PREFERRED PRACTICE PATTERNS (PPP) Philippines: 2016 The Age Related Macular Degeneration (AMD) Preferred Practice Patterns (PPP) Philippines: 2016 was prepared by
Class Update: Vascular Endothelial Growth Factors
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME
 Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME Ophthalmology Innovation Summit @ AAO, October 25 2018 Megan Baldwin PhD, CEO & Managing Director Disclaimer Investment
Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME Ophthalmology Innovation Summit @ AAO, October 25 2018 Megan Baldwin PhD, CEO & Managing Director Disclaimer Investment
VASCULAR ENDOTHELIAL GROWTH FACTOR INHIBITORS
 VASCULAR ENDOTHELIAL GROWTH FACTOR INHIBITORS FOR OPHTHALMIC USE Policy Number: 2016D0001B Effective Date: January 1, 2016 Table of Contents: Page: Cross Reference Policy: POLICY DESCRIPTION 2 ZALTRAP
VASCULAR ENDOTHELIAL GROWTH FACTOR INHIBITORS FOR OPHTHALMIC USE Policy Number: 2016D0001B Effective Date: January 1, 2016 Table of Contents: Page: Cross Reference Policy: POLICY DESCRIPTION 2 ZALTRAP
Curriculum Vitae Ryan M. Rich, M.D.
 Retina Consultants of Southern Colorado, P.C. 3030 North Circle Drive, Suite 301 Colorado Springs, CO 80909 (719) 473-9595 DOB: March 20, 1972 Citizenship: USA CURRENT POSITION 2008-present Retina Consultants
Retina Consultants of Southern Colorado, P.C. 3030 North Circle Drive, Suite 301 Colorado Springs, CO 80909 (719) 473-9595 DOB: March 20, 1972 Citizenship: USA CURRENT POSITION 2008-present Retina Consultants
