Supplementary Appendix
|
|
|
- Robert Barton
- 6 years ago
- Views:
Transcription
1 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Wanner C, Inzucchi SE, Lachin JM, et al. Empagliflozin and progression of kidney disease in type 2 diabetes. N Engl J Med 2016;375: DOI: /NEJMoa
2 Supplement to: Wanner C, Inzucchi SE, Lachin J, et al. Empagliflozin and Progression of Kidney Disease in Type 2 Diabetes. N Engl J Med. Figure S1. Pre-specified subgroup analyses for incident or worsening nephropathy Figure S2. Incident or worsening nephropathy with empagliflozin 10 mg, empagliflozin 25 mg and placebo... 3 Figure S3. Incident or worsening nephropathy or cardiovascular death with empagliflozin and placebo... 4 Figure S4. Incident or worsening nephropathy with empagliflozin and placebo in patients with prevalent kidney disease defined as egfr (MDRD) <60 ml/min/1.73m 2 and/or macroalbuminuria (urine albumin-to-creatinine ratio >300 mg/g) at baseline... 6 Figure S5. Composite of doubling of serum creatinine (accompanied by egfr [MDRD] 45 ml/min/1.73m 2 ), initiation of renal replacement therapy, or death due to renal disease in patients with macroalbuminuria (urine albumin-to-creatinine ratio >300 mg/g) at baseline... 7 Figure S6. Post-hoc subgroup analyses of doubling of serum creatinine, initiation of renal replacement therapy, or death due to renal disease Figure S7. egfr according to CKD-EPI formula over 192 weeks in patients with egfr <60 ml/min/1.73m 2 and 60 ml/min/1.73m 2 at baseline Figure S8. Adverse events with empagliflozin compared with placebo Figure S9. Acute renal failure and acute kidney injury with empagliflozin and placebo Table S1. Baseline characteristics of patients with egfr (MDRD) <60 and 60 ml/min/1.73m 2 at baseline Table S2. Cardiovascular medications introduced post-baseline in patients with egfr (MDRD) <60 and 60 ml/min/1.73m 2 at baseline Table S3. Composite microvascular outcome with empagliflozin compared with placebo Table S4. Incident or worsening nephropathy and incident or worsening nephropathy or cardiovascular death: pre-specified analysis and post-hoc sensitivity analysis Table S5. Incident or worsening nephropathy and incident or worsening nephropathy or cardiovascular death with empagliflozin compared with placebo in patients with prevalent kidney disease defined as egfr (MDRD) <60 ml/min/1.73m 2 and/or macroalbuminuria (urine albuminto-creatinine ratio >300 mg/g) at baseline Table S6. Adverse events in patients with egfr (MDRD) <60 and 60 ml/min/1.73m 2 at baseline Table S7. Complicated urinary tract infections by MedDRA preferred term in patients with egfr (MDRD) <60 and 60 ml/min/1.73m 2 at baseline Table S8. Changes in clinical laboratory parameters in patients with egfr (MDRD) <60 and 60 ml/min/1.73m 2 at baseline
3 Figure S1. Pre-specified subgroup analyses for incident or worsening nephropathy. Cox regression analyses in patients treated with 1 dose of study drug. p-value is for test of homogeneity of treatment group difference among subgroups (test for treatment group by covariate interaction) with no adjustment for multiple testing. ACE, angiotensin-converting enzyme. ARB, angiotensin-receptor blocker. DBP, diastolic blood pressure. SBP, systolic blood pressure. 2
4 Figure S2. Incident or worsening nephropathy with empagliflozin 10 mg, empagliflozin 25 mg and placebo Kaplan-Meier estimates of time to first occurrence of incident or worsening nephropathy in the empagliflozin 10 mg, empagliflozin 25 mg and placebo groups in patients treated with 1 dose of study drug. Hazard ratio is based on Cox regression analysis. Pre-specified analyses. 3
5 Figure S3. Incident or worsening nephropathy or cardiovascular death with empagliflozin and placebo Kaplan-Meier estimates of time to first occurrence of incident or worsening nephropathy or cardiovascular death in the empagliflozin pooled and placebo groups (Panel A) and in the empagliflozin 10 mg, empagliflozin 25 mg and placebo groups (Panel B) in patients treated with 1 dose of study drug. Hazard ratios are based on Cox regression analyses. Pre-specified analyses. A Empagliflozin pooled and placebo groups 4
6 B Empagliflozin 10 mg, empagliflozin 25 mg and placebo groups 5
7 Figure S4. Incident or worsening nephropathy with empagliflozin and placebo in patients with prevalent kidney disease defined as egfr (MDRD) <60 ml/min/1.73m 2 and/or macroalbuminuria (urine albumin-to-creatinine ratio >300 mg/g) at baseline Kaplan-Meier estimates of time to first occurrence of incident or worsening nephropathy in the empagliflozin and placebo groups in patients with prevalent kidney disease treated with 1 dose of study drug. Hazard ratio is based on Cox regression analysis. Post-hoc analyses. egfr, estimated glomerular filtration rate. MDRD, Modification of Diet in Renal Disease. 6
8 Figure S5. Composite of doubling of serum creatinine (accompanied by egfr [MDRD] 45 ml/min/1.73m 2 ), initiation of renal replacement therapy, or death due to renal disease in patients with macroalbuminuria (urine albumin-to-creatinine ratio >300 mg/g) at baseline Kaplan-Meier estimates in empagliflozin and placebo groups in patients with macroalbuminuria (urine albumin-to-creatinine ratio >300 mg/g) at baseline treated with 1 dose of study drug. Hazard ratio is based on Cox regression analysis. Post-hoc analyses. egfr, estimated glomerular filtration rate. MDRD, Modification of Diet in Renal Disease. 7
9 Figure S6. Post-hoc subgroup analyses of doubling of serum creatinine, initiation of renal replacement therapy, or death due to renal disease. Cox regression analyses in patients treated with 1 dose of study drug. p-value is for test of homogeneity of treatment group difference among subgroups (test for treatment group by covariate interaction) with no adjustment for multiple testing. 8
10 Figure S7. egfr according to CKD-EPI formula over 192 weeks in patients with egfr <60 ml/min/1.73m 2 and 60 ml/min/1.73m 2 at baseline. egfr according to CKD-EPI formula over 192 weeks in patients with egfr <60 ml/min/1.73m2 at baseline (Panel A) and 60 ml/min/1.73m2 at baseline (Panel B). egfr category at baseline is according to the MDRD formula. Post-hoc analysis. Mixed model repeated measures analysis, including baseline glycated hemoglobin as a linear covariate and region, baseline egfr (MDRD), baseline body mass index, the last week a patient could have had egfr measurements, treatment, visit, visit by treatment interaction, baseline glycated hemoglobin by visit interaction, baseline egfr by visit interaction, baseline egfr by treatment interaction and baseline egfr by visit by treatment interaction as fixed effects, in patients treated with 1 dose of study drug. egfr, estimated glomerular filtration rate; CKD-EPI, Chronic Kidney Disease Epidemiology Collaboration; MDRD, Modification of Diet in Renal Disease. A Patients with egfr <60 ml/min/1.73m 2 at baseline 9
11 B Patients with egfr 60 ml/min/1.73m 2 at baseline 10
12 Figure S8. Adverse events with empagliflozin compared with placebo. Incidence rate ratios (based on rate per 100 patient-years) for empagliflozin compared with placebo based on post-hoc assessments. Data are from patients treated with 1 dose of study drug based on events that occurred during treatment or 7 days after the last intake of study drug. 11
13 * Plasma glucose 70 mg/dl and/or requiring assistance. Based on 79 MedDRA preferred terms. Pyelonephritis, urosepsis or serious adverse event consistent with urinary tract infection. Based on 88 MedDRA preferred terms. Based on 8 MedDRA preferred terms. ǁ Based on the narrow standardized MedDRA query acute renal failure. ** Based on 1 standardized MedDRA query. Based on 62 MedDRA preferred terms. Based on 2 MedDRA preferred terms. egfr, estimated glomerular filtration rate. MDRD, Modification of Diet in Renal Disease. MedDRA, Medical Dictionary for Regulatory Activities. 12
14 Figure S9. Acute renal failure and acute kidney injury with empagliflozin and placebo. Kaplan-Meier estimates of time to first occurrence of acute renal failure based on the narrow standardized MedDRA query acute renal failure and time to first occurrence of acute kidney injury (MedDRA preferred term) in the empagliflozin and placebo groups in patients treated with 1 dose of study drug based on events that occurred during treatment or 7 days after the last intake of study drug. Post-hoc analyses. MedDRA, Medical Dictionary for Regulatory Activities. 13
15 Table S1. Baseline characteristics of patients with egfr (MDRD) <60 and 60 ml/min/1.73m 2 at baseline Patients with egfr (MDRD) <60 ml/min/1.73m 2 at baseline Patients with egfr (MDRD) 60 ml/min/1.73m 2 at baseline Characteristic* Placebo (N=607) Empagliflozin (N=1212) Placebo (N=1726) Empagliflozin (N=3473) Age years 67.1± ± ± ±8.5 Male no. (%) 418 (68.9) 816 (67.3) 1262 (73.1) 2518 (72.5) Race no. (%) White 444 (73.1) 878 (72.4) 1234 (71.5) 2524 (72.7) Asian 132 (21.7) 265 (21.9) 379 (22.0) 741 (21.3) Black/African-American 27 (4.4) 56 (4.6) 93 (5.4) 181 (5.2) Other/Missing 4 (0.7) 13 (1.1) 20 (1.2) 27 (0.8) Ethnicity no. (%) Not Hispanic or Latino 507 (83.5) 1000 (82.5) 1405 (81.4) 2835 (81.6) Hispanic or Latino 99 (16.3) 211 (17.4) 319 (18.5) 634 (18.3) Missing 1 (0.2) 1 (0.1) 2 (0.1) 4 (0.1) Region no. (%) Europe 210 (34.6) 418 (34.5) 749 (43.4) 1508 (43.4) North America (plus Australia and 175 (28.8) 329 (27.1) 287 (16.6) 603 (17.4) New Zealand) Asia 113 (18.6) 232 (19.1) 337 (19.5) 665 (19.1) Latin America 79 (13.0) 174 (14.4) 281 (16.3) 545 (15.7) Africa 30 (4.9) 59 (4.9) 72 (4.2) 152 (4.4) Weight kg 87.1± ± ± ±18.7 Body mass index kg/m ± ± ± ±5.2 CV risk factor no. (%) 601 (99.0) 1203 (99.3) 1706 (98.8) 3452 (99.4) Coronary artery disease 482 (79.4) 938 (77.4) 1281 (74.2) 2606 (75.0) Multi-vessel coronary artery 331 (54.5) 610 (50.3) 769 (44.6) 1569 (45.2) disease History of myocardial infarction 264 (43.5) 559 (46.1) 819 (47.5) 1630 (46.9) Coronary artery bypass graft 196 (32.3) 380 (31.4) 367 (21.3) 795 (22.9) History of stroke 156 (25.7) 293 (24.2) 397 (23.0) 791 (22.8) Peripheral artery disease 130 (21.4) 314 (25.9) 349 (20.2) 667 (19.2) Single vessel coronary artery 54 (8.9) 108 (8.9) 184 (10.7) 390 (11.2) 14
16 disease Cardiac failure 89 (14.7) 174 (14.4) 155 (9.0) 288 (8.3) Glycated hemoglobin ǁ % 8.03± ± ± ±0.84 Time since diagnosis of type 2 diabetes no. (%) 1 years 9 (1.5) 23 (1.9) 43 (2.5) 105 (3.0) >1 to 5 years 55 (9.1) 121 (10.0) 316 (18.3) 591 (17.0) >5 to 10 years 121 (19.9) 274 (22.6) 450 (26.1) 901 (25.9) >10 years 422 (69.5) 794 (65.5) 917 (53.1) 1876 (54.0) Glucose-lowering therapy no. (%) Medication taken alone or in combination Metformin 369 (60.8) 711 (58.7) 1365 (79.1) 2746 (79.1) Sulfonylurea 234 (38.6) 480 (39.6) 758 (43.9) 1534 (44.2) Thiazolidinedione 27 (4.4) 62 (5.1) 74 (4.3) 136 (3.9) Insulin 357 (58.8) 699 (57.7) 778 (45.1) 1551 (44.7) Monotherapy 217 (35.7) 430 (35.5) 474 (27.5) 950 (27.4) Dual therapy 279 (46.0) 530 (43.7) 869 (50.3) 1727 (49.7) Anti-hypertensive therapy no. (%) 590 (97.2) 1186 (97.9) 1631 (94.5) 3259 (93.8) Angiotensin-converting enzyme 502 (82.7) 1031 (85.1) 1366 (79.1) 2766 (79.6) inhibitors/angiotensin receptor blockers Beta-blockers 415 (68.4) 829 (68.4) 1083 (62.7) 2226 (64.1) Diuretics 355 (58.5) 710 (58.6) 633 (36.7) 1336 (38.5) Calcium channel blockers 227 (37.4) 446 (36.8) 561 (32.5) 1082 (31.2) Mineralocorticoid receptor 48 (7.9) 119 (9.8) 88 (5.1) 186 (5.4) antagonists Renin inhibitors 5 (0.8) 12 (1.0) 14 (0.8) 15 (0.4) Other 70 (11.5) 144 (11.9) 121 (7.0) 239 (6.9) Lipid-lowering therapy no. (%) 496 (81.7) 1024 (84.5) 1368 (79.3) 2795 (80.5) Statins 461 (75.9) 966 (79.7) 1312 (76.0) 2663 (76.7) Fibrates 80 (13.2) 173 (14.3) 119 (6.9) 258 (7.4) Ezetimibe 31 (5.1) 43 (3.5) 50 (2.9) 146 (4.2) Niacin 15 (2.5) 22 (1.8) 20 (1.2) 69 (2.0) Other 72 (11.9) 127 (10.5) 103 (6.0) 238 (6.9) 15
17 Anti-coagulants no. (%) 552 (90.9) 1087 (89.7) 1538 (89.1) 3074 (88.5) Acetylsalicylic acid 495 (81.5) 981 (80.9) 1432 (83.0) 2894 (83.3) Clopidogrel 56 (9.2) 133 (11.0) 193 (11.2) 361 (10.4) Vitamin K antagonists 63 (10.4) 100 (8.3) 93 (5.4) 166 (4.8) Systolic blood pressure mmhg 136.4± ± ± ±16.6 Diastolic blood pressure mmhg 74.6± ± ± ±9.5 Total cholesterol mg/dl** 163.0± ± ± ±44.4 Low density lipoprotein cholesterol 85.0± ± ± ±36.0 mg/dl High density lipoprotein cholesterol 42.9± ± ± ±11.7 mg/dl** Triglycerides mg/dl** 180.4± ± ± ±136.4 Estimated glomerular filtration rate 48.6± ± ± ±17.1 (MDRD) ml/min/1.73m 2 Urine albumin-to-creatinine ratio no. (%) <30 mg/g 283 (46.6) 566 (46.7) 1099 (63.7) 2223 (64.0) 30 to 300 mg/g 205 (33.8) 411 (33.9) 470 (27.2) 926 (26.7) >300 mg/g 115 (18.9) 223 (18.4) 145 (8.4) 286 (8.2) Missing 4 (0.7) 12 (1.0) 12 (0.7) 38 (1.1) * Plus minus values are means ± SD. Body mass index is the weight in kilograms divided by the square of the height in meters. Information was not available for one patient in the placebo group with 60 ml/min/1.73m 2 at baseline. Information was not available for one patient in the placebo group and one patient in the empagliflozin 25 mg group with 60 ml/min/1.73m 2 at baseline. Based on the narrow standardized MedDRA query cardiac failure. ǁ Information was not available for one patient in the empagliflozin 10 mg group with 60 ml/min/1.73m 2 at baseline. ** Placebo n=598 and empagliflozin n=1202 for patients with egfr <60 ml/min/1.73m 2 at baseline; placebo n=1711 and empagliflozin n=3423 for patients with egfr 60 ml/min/1.73m 2 at baseline. Placebo n=598 and empagliflozin n=1201 for patients with egfr <60 ml/min/1.73m 2 at baseline; placebo n=1711 and empagliflozin n=3421 for patients with egfr 60 ml/min/1.73m 2 at baseline. Post-hoc assessment: p<0.05 for difference between empagliflozin and placebo based on Chi-square test for binary/categorical variables or t-test for continuous variables. MDRD, Modification of Diet in Renal Disease. MedDRA, Medical Dictionary for Regulatory Activities. 16
18 Table S2. Cardiovascular medications introduced post-baseline in patients with egfr (MDRD) <60 and 60 ml/min/1.73m 2 at baseline Patients with egfr <60 ml/min/1.73m 2 at baseline Placebo Empagliflozin Patients with egfr 60 ml/min/1.73m 2 at baseline Placebo Empagliflozin (N = 607) (N = 1212) (N = 1726) (N = 3473) Anti-hypertensive therapies no. (%) 359 (59.1) 624 (51.5) 831 (48.1) 1463 (42.1) Angiotensin-converting enzyme 202 (33.3) 359 (29.6) 500 (29.0) 864 (24.9) inhibitors/angiotensin receptor blockers Beta-blockers 136 (22.4) 256 (21.1) 345 (20.0) 602 (17.3) Diuretics 227 (37.4) 327 (27.0) 381 (22.1) 572 (16.5) Calcium channel blockers 137 (22.6) 231 (19.1) 344 (19.9) 441 (12.7) Mineralocorticoid receptor 54 (8.9) 64 (5.3) 82 (4.8) 113 (3.3) antagonists Renin inhibitors 2 (0.3) 4 (0.3) 4 (0.2) 5 (0.1) Other 62 (10.2) 106 (8.7) 103 (6.0) 168 (4.8) Lipid-lowering therapies no. (%) 207 (34.1) 354 (29.2) 512 (29.7) 1012 (29.1) Statins 169 (27.8) 289 (23.8) 432 (25.0) 856 (24.6) Fibrates 41 (6.8) 53 (4.4) 87 (5.0) 158 (4.5) Ezetimibe 17 (2.8) 25 (2.1) 32 (1.9) 69 (2.0) Niacin 5 (0.8) 7 (0.6) 10 (0.6) 16 (0.5) Other 24 (4.0) 35 (2.9) 47 (2.7) 74 (2.1) Anti-coagulants no. (%) 213 (35.1) 391 (32.3) 495 (28.7) 949 (27.3) Acetylsalicylic acid 127 (20.9) 238 (19.6) 327 (18.9) 602 (17.3) Clopidogrel 36 (5.9) 82 (6.8) 98 (5.7) 179 (5.2) Vitamin K antagonists 43 (7.1) 60 (5.0) 59 (3.4) 99 (2.9) Patients treated with 1 dose of study drug. Cardiovascular medications introduced post-baseline irrespective of study medication intake. MDRD, Modification of Diet in Renal Disease. 17
19 Table S3. Composite microvascular outcome with empagliflozin compared with placebo Placebo No. with event/ analyzed (%) Rate/1000 patient-yr No. with event/ analyzed (%) Empagliflozin Rate/1000 patient-yr Hazard ratio (95% CI) p-value Composite microvascular 424/2068 (20.5) /4132 (14.0) (0.54, 0.70) <0.001 outcome Initiation of laser 29/2333 (1.2) /4687 (0.9) (0.43, 1.12) therapy for retinopathy Vitreous hemorrhage 16/2333 (0.7) /4687 (0.6) (0.51, 1.71) Diabetes-related 2/2333 (0.1) 0.3 4/4687 (0.1) 0.3 blindness* Incident or worsening nephropathy 388/2061 (18.8) /4124 (12.7) (0.53, 0.70) <0.001 Prespecified Cox regression analyses in patients treated with 1 dose of study drug. *Hazard ratio and 95% CI were not analyzed as the total number of patients with events was <14. 18
20 Table S4. Incident or worsening nephropathy and incident or worsening nephropathy or cardiovascular death: pre-specified analysis and post-hoc sensitivity analysis Incident or worsening nephropathy Placebo Empagliflozin Hazard ratio No. with event/ Rate/1000 No. with event/ Rate/1000 (95% CI) analyzed (%) patient-yr analyzed (%) patient-yr p-value Pre-specified analysis* 388/2061 (18.8) /4124 (12.7) (0.53, 0.70) <0.001 Sensitivity analysis 388/2333 (16.6) /4687 (11.2) (0.53, 0.69) <0.001 Incident or worsening nephropathy or cardiovascular death Pre-specified analysis* 497/2102 (23.6) /4170 (16.2) (0.55, 0.69) <0.001 Sensitivity analysis 497/2333 (21.3) /4687 (14.4) (0.54, 0.69) <0.001 * Patients treated with 1 dose of study drug not considering patients with macroalbuminuria at baseline and/or patients without baseline and/or post-baseline serum creatinine measurements and/or patients without post-baseline urine albumin-to-creatinine ratio measurements (unless they experienced at least one of the other components of the composite renal outcome). All patients treated with 1 dose of study drug. 19
21 Table S5. Incident or worsening nephropathy and incident or worsening nephropathy or cardiovascular death with empagliflozin compared with placebo in patients with prevalent kidney disease defined as egfr (MDRD) <60 ml/min/1.73m 2 and/or macroalbuminuria (urine albumin-to-creatinine ratio >300 mg/g) at baseline Placebo No. with event/ analyzed (%) Rate/1000 patient-yr Empagliflozin No. with event/ Rate/1000 analyzed (%) patient-yr Hazard ratio (95% CI) p-value Incident or worsening 161/507 (31.8) /998 (20.7) (0.47, 0.71) <0.001 nephropathy Incident or worsening nephropathy or cardiovascular death 213/539 (39.5) /1039 (27.9) (0.53, 0.76) <0.001 Post-hoc Cox regression analyses in patients with prevalent kidney disease treated with 1 dose of study drug. egfr, estimated glomerular filtration rate. MDRD, Modification of Diet in Renal Disease. 20
22 Table S6. Adverse events in patients with egfr (MDRD) <60 and 60 ml/min/1.73m 2 at baseline. Event Patients with egfr (MDRD) <60 ml/min/1.73m 2 at baseline Placebo Pooled empagliflozin (N = 607) (N = 1212) No. (%) Rate per No. (%) Rate per with one or 100-patient with one or 100-patient more event years more event years Patients with egfr (MDRD) 60 ml/min/1.73m 2 at baseline Placebo Pooled empagliflozin (N = 1726) (N = 3473) No. (%) Rate per No. (%) Rate per with one or 100-patient with one or 100-patient more event years more event years Any adverse event 577 (95.1) (91.3) (90.5) (89.9) Rate ratio (95% CI) 0.70 ( ) 0.88 ( ) Severe adverse event 208 (34.3) (29.7) (22.2) (21.3) 9.1 Rate ratio (95% CI) 0.81 ( ) 0.92 ( ) Serious adverse event 321 (52.9) (45.5) (38.6) (35.6) 17.0 Rate ratio (95% CI) 0.78 ( ) 0.87 ( ) Death 40 (6.6) (5.6) (4.6) (3.1) 1.2 Rate ratio (95% CI) 0.82 ( ) 0.66 ( ) Adverse event leading to 167 (27.5) (22.9) (16.6) (15.4) 6.2 discontinuation Rate ratio (95% CI) 0.79 ( ) 0.90 ( ) Confirmed hypoglycemic 233 (38.4) (32.3) (24.2) (26.3) 12.8 adverse event* Rate ratio (95% CI) 0.78 ( ) 1.10 ( ) Requiring assistance 18 (3.0) (1.9) (1.0) (1.2) 0.4 Rate ratio (95% CI) 0.62 ( ) 1.06 ( ) Event consistent with urinary tract infection 132 (21.7) (22.9) (16.9) (16.2) 6.9 Rate ratio (95% CI) 1.06 ( ) 0.92 ( ) 21
23 Complicated urinary tract 17 (2.8) (3.1) (1.4) (1.3) 0.5 infection Rate ratio (95% CI) ( ) ( ) Event consistent with genital 10 (1.6) (5.3) (1.9) (6.8) 2.7 infection Rate ratio (95% CI) ( ) ( ) Event consistent with volume 49 (8.1) (6.7) (3.8) (4.5) 1.8 depletion Rate ratio (95% CI) ( ) ( ) Acute renal failure ǁ 87 (14.3) (11.2) (3.9) (3.2) 1.2 Rate ratio (95% CI) 0.75 ( ) Acute kidney injury 22 (3.6) (2.1) (0.9) (0.5) 0.2 Diabetic ketoacidosis** 1 (0.2) (0.2) (0.1) <0.1 Thromboembolic event 7 (1.2) (1.1) (0.8) (0.5) ( ) Rate ratio (95% CI) 0.89 ( ) 0.63 ( ) Bone fracture 32 (5.3) (4.7) (3.4) (3.5) 1.4 Rate ratio (95% CI) 0.86 ( ) 0.99 ( ) Hyperkalemia 42 (6.9) (3.9) (2.1) (1.3) 0.5 Rate ratio (95% CI) 0.53 ( ) 0.61 ( ) Data are from patients treated with 1 dose of study drug based on events that occurred during treatment or 7 days after the last intake of study drug. Post-hoc assessments. * Plasma glucose 70 mg/dl and/or requiring assistance. Based on 79 MedDRA preferred terms. Pyelonephritis, urosepsis or serious adverse event consistent with urinary tract infection. Based on 88 MedDRA preferred terms. Based on 8 MedDRA preferred terms. ǁ Based on the narrow standardized MedDRA query acute renal failure. ** Based on 4 MedDRA preferred terms. Incidence rate ratio and 95% confidence interval not calculated due to low number of events. Based on 1 standardized MedDRA query Based on 62 MedDRA preferred terms. Based on 2 MedDRA preferred terms. egfr, estimated glomerular filtration rate. MDRD, Modification of Diet in Renal Disease. 22
24 Table S7. Complicated urinary tract infections by MedDRA preferred term in patients with egfr (MDRD) <60 and 60 ml/min/1.73m 2 at baseline Patients with egfr (MDRD) <60 ml/min/1.73m 2 at baseline Patients with egfr (MDRD) 60 ml/min/1.73m 2 at baseline Placebo (N = 607) Empagliflozin (N = 1212) Placebo (N = 1726) Empagliflozin (N = 3473) no. (%) with one or more event Complicated urinary tract infection 17 (2.8) 37 (3.1) 24 (1.4) 45 (1.3) Urinary tract infection 7 (1.2) 15 (1.2) 9 (0.5) 14 (0.4) Urosepsis 2 (0.3) 9 (0.7) 1 (0.1) 8 (0.2) Pyelonephritis 2 (0.3) 4 (0.3) 2 (0.1) 9 (0.3) Pyelonephritis chronic 4 (0.7) 5 (0.4) 6 (0.3) 5 (0.1) Pyelonephritis acute 1 (0.2) 4 (0.3) 5 (0.3) 4 (0.1) Cystitis (0.1) 0 Kidney infection 2 (0.3) 1 (0.1) 0 3 (0.1) Urinary tract infection fungal 0 2 (0.2) 0 1 (<0.1) Cystitis bacterial (0.1) 0 Escherichia urinary tract infection 1 (0.2) Urinary tract infection (<0.1) pseudomonal Cystitis glandularis (<0.1) Cystitis hemorrhagic (0.1) 0 Nephritis (<0.1) Data are from patients treated with 1 dose of study drug based on events that occurred during treatment or 7 days after the last intake of study medication. Complicated urinary tract infection defined as pyelonephritis, urosepsis or serious adverse event consistent with urinary tract infection. MedDRA, Medical Dictionary for Regulatory Activities. egfr, estimated glomerular filtration rate. MDRD, Modification of Diet in Renal Disease. 23
25 Table S8. Changes in clinical laboratory parameters in patients with egfr (MDRD) <60 and 60 ml/min/1.73m 2 at baseline. Patients with egfr (MDRD) <60 ml/min/1.73m 2 at baseline Patients with egfr (MDRD) 60 ml/min/1.73m 2 at baseline Placebo Empagliflozin Placebo Empagliflozin Baseline Change from baseline Baseline Change from baseline Baseline Change from baseline Baseline Change from baseline Hematocrit, % 40.3 ± ± ± ± ± ± ± ± 5.2 Hemoglobin, g/dl 13.1 ± ± ± ± ± ± ± ± 1.3 Aspartate 15 ± 12 0 ± ± 10-1 ± ± 12 1 ± ± 10 0 ± 23 aminotransferase, U/L Alanine aminotransferase, 17 ± 13-1 ± ± 10-1 ± ± 14 0 ± ± 12-2 ± 19 U/L Alkaline phosphatase, U/L 66 ± 35 8 ± ± 36 6 ± ± 31 4 ± ± 31 2 ± 26 Electrolytes Sodium, meq/l 141 ± 2 0 ± ± 2 0 ± ± 2 0 ± ± 2 0 ± 2 Potassium, meq/l 4.4 ± ± ± ± ± ± ± ± 0.4 Calcium, mg/dl 9.8 ± ± ± ± ± ± ± ± 0.5 Magnesium, meq/l 1.7 ± ± ± ± ± ± ± ± 0.2 Chloride, meq/l 102 ± 2-1 ± ± 2-1 ± ± 2-1 ± ± 2-1 ± 2 Phosphate, mg/dl 3.7 ± ± ± ± ± ± ± ± 0.3 Plus-minus values are means ± SD and data are normalized to a standard reference range. Changes from baseline are the last measurement 3 days after the last intake of study medication. Data are from patients treated with 1 dose of study drug with a baseline and on-treatment measurement. Conversion factors: sodium, potassium, chloride and phosphate: 1 meq/l = 1 mmol/l; calcium: 1 mg/dl = 0.25 mmol/l; magnesium: 1 meq/l = 0.5 mmol/l. egfr, estimated glomerular filtration rate. MDRD, Modification of Diet in Renal Disease. Hematocrit: Placebo n=587 and empagliflozin n=1141 for patients with egfr <60 ml/min/1.73m 2 at baseline; placebo n=1671 and empagliflozin n=3355 for patients with egfr 60 ml/min/1.73m 2 at baseline. Hemoglobin: Placebo n=589 and empagliflozin n=1145 for patients with egfr <60 ml/min/1.73m 2 at baseline; placebo n=1674 and empagliflozin n=3366 for patients with egfr 60 ml/min/1.73m 2 at baseline. 24
26 Aspartate aminotransferase, alanine aminotransferase and alkaline phosphatase: Placebo n=603 and empagliflozin n=1182 for patients with egfr <60 ml/min/1.73m 2 at baseline; placebo n=1710 (or n=1709 for aspartate aminotransferase) and empagliflozin n=3430 for patients with egfr 60 ml/min/1.73m 2 at baseline. Electrolytes: Placebo n=588 and empagliflozin n=1150 (or n=1151 for potassium) for patients with egfr <60 ml/min/1.73m 2 at baseline; placebo n=1673 (or n=1672 for potassium) and empagliflozin n=3370 for patients with egfr 60 ml/min/1.73m 2 at baseline. 25
egfr > 50 (n = 13,916)
 Saxagliptin and Cardiovascular Risk in Patients with Type 2 Diabetes Mellitus and Moderate or Severe Renal Impairment: Observations from the SAVOR-TIMI 53 Trial Supplementary Table 1. Characteristics according
Saxagliptin and Cardiovascular Risk in Patients with Type 2 Diabetes Mellitus and Moderate or Severe Renal Impairment: Observations from the SAVOR-TIMI 53 Trial Supplementary Table 1. Characteristics according
This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data.
 abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the
abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the
Supplementary Table 1. Patient demographics and baseline characteristics (treated patients).
 Supplementary Table 1. Patient demographics and baseline characteristics (treated patients). Placebo (n=188) 10 mg (n=186) 25 mg (n=189) Total (n=563) Gender, n (%) Male 75 (40) 97 (52) 84 (44) 256 (45)
Supplementary Table 1. Patient demographics and baseline characteristics (treated patients). Placebo (n=188) 10 mg (n=186) 25 mg (n=189) Total (n=563) Gender, n (%) Male 75 (40) 97 (52) 84 (44) 256 (45)
PROTEZIONE DAL DANNO RENALE NEL DIABETE TIPO 2: RUOLO DEI NUOVI FARMACI. Massimo Boemi UOC Malattie Metaboliche e Diabetologia IRCCS INRCA Ancona
 PROTEZIONE DAL DANNO RENALE NEL DIABETE TIPO 2: RUOLO DEI NUOVI FARMACI Massimo Boemi UOC Malattie Metaboliche e Diabetologia IRCCS INRCA Ancona Disclosure Dr Massimo Boemi has been granted as speaker
PROTEZIONE DAL DANNO RENALE NEL DIABETE TIPO 2: RUOLO DEI NUOVI FARMACI Massimo Boemi UOC Malattie Metaboliche e Diabetologia IRCCS INRCA Ancona Disclosure Dr Massimo Boemi has been granted as speaker
Supplementary Online Content
 Supplementary Online Content Afkarian M, Zelnick L, Hall YN, et al. Clinical manifestations of kidney disease among US adults with diabetes, 1988-2014. JAMA. doi:10.1001/jama.2016.10924 emethods efigure
Supplementary Online Content Afkarian M, Zelnick L, Hall YN, et al. Clinical manifestations of kidney disease among US adults with diabetes, 1988-2014. JAMA. doi:10.1001/jama.2016.10924 emethods efigure
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Solomon SD, Uno H, Lewis EF, et al. Erythropoietic response
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Solomon SD, Uno H, Lewis EF, et al. Erythropoietic response
The EMPA-REG OUTCOME trial: Design and results. David Fitchett, MD University of Toronto, Canada
 The EMPA-REG OUTCOME trial: Design and results David Fitchett, MD University of Toronto, Canada Asian Cardio Diabetes Forum April 23 24, 2016 Kuala Lumpur, Malaysia Life Expectancy Is Reduced by ~12 Years
The EMPA-REG OUTCOME trial: Design and results David Fitchett, MD University of Toronto, Canada Asian Cardio Diabetes Forum April 23 24, 2016 Kuala Lumpur, Malaysia Life Expectancy Is Reduced by ~12 Years
Data Analysis Plan for assessing clinical efficacy and safety of ER niacin/laropiprant in the HPS2-THRIVE trial
 Data Analysis Plan for assessing clinical efficacy and safety of ER niacin/laropiprant in the HPS2-THRIVE trial 1 Background This Data Analysis Plan describes the strategy, rationale and statistical methods
Data Analysis Plan for assessing clinical efficacy and safety of ER niacin/laropiprant in the HPS2-THRIVE trial 1 Background This Data Analysis Plan describes the strategy, rationale and statistical methods
Supplementary Table 1. Criteria for selection of normal control individuals among healthy volunteers
 Supplementary Table 1. Criteria for selection of normal control individuals among healthy volunteers Medical parameters Cut-off values BMI (kg/m 2 ) 25.0 Waist (cm) (Men and Women) (Men) 85, (Women) 90
Supplementary Table 1. Criteria for selection of normal control individuals among healthy volunteers Medical parameters Cut-off values BMI (kg/m 2 ) 25.0 Waist (cm) (Men and Women) (Men) 85, (Women) 90
GALECTIN-3 PREDICTS LONG TERM CARDIOVASCULAR DEATH IN HIGH-RISK CORONARY ARTERY DISEASE PATIENTS
 GALECTIN-3 PREDICTS LONG TERM CARDIOVASCULAR DEATH IN HIGH-RISK CORONARY ARTERY DISEASE PATIENTS Table of Contents List of authors pag 2 Supplemental figure I pag 3 Supplemental figure II pag 4 Supplemental
GALECTIN-3 PREDICTS LONG TERM CARDIOVASCULAR DEATH IN HIGH-RISK CORONARY ARTERY DISEASE PATIENTS Table of Contents List of authors pag 2 Supplemental figure I pag 3 Supplemental figure II pag 4 Supplemental
A factorial randomized trial of blood pressure lowering and intensive glucose control in 11,140 patients with type 2 diabetes
 A factorial randomized trial of blood pressure lowering and intensive glucose control in 11,140 patients with type 2 diabetes Hypotheses: Among individuals with type 2 diabetes, the risks of major microvascular
A factorial randomized trial of blood pressure lowering and intensive glucose control in 11,140 patients with type 2 diabetes Hypotheses: Among individuals with type 2 diabetes, the risks of major microvascular
Dapagliflozin and cardiovascular outcomes in type 2
 EARN 3 FREE CPD POINTS diabetes Leader in digital CPD for Southern African healthcare professionals Dapagliflozin and cardiovascular outcomes in type 2 diabetes Introduction People with type 2 diabetes
EARN 3 FREE CPD POINTS diabetes Leader in digital CPD for Southern African healthcare professionals Dapagliflozin and cardiovascular outcomes in type 2 diabetes Introduction People with type 2 diabetes
Effects of Lowering LDL Cholesterol on Progression of Kidney Disease
 Effects of Lowering LDL Cholesterol on Progression of Kidney Disease Richard Haynes, David Lewis, Jonathan Emberson, Christina Reith, Lawrence Agodoa, Alan Cass, Jonathan C. Craig, Dick de Zeeuw, Bo Feldt-Rasmussen,
Effects of Lowering LDL Cholesterol on Progression of Kidney Disease Richard Haynes, David Lewis, Jonathan Emberson, Christina Reith, Lawrence Agodoa, Alan Cass, Jonathan C. Craig, Dick de Zeeuw, Bo Feldt-Rasmussen,
Supplementary Online Content
 Supplementary Online Content Xu X, Qin X, Li Y, et al. Efficacy of folic acid therapy on the progression of chronic kidney disease: the Renal Substudy of the China Stroke Primary Prevention Trial. JAMA
Supplementary Online Content Xu X, Qin X, Li Y, et al. Efficacy of folic acid therapy on the progression of chronic kidney disease: the Renal Substudy of the China Stroke Primary Prevention Trial. JAMA
Metformin should be considered in all patients with type 2 diabetes unless contra-indicated
 November 2001 N P S National Prescribing Service Limited PPR fifteen Prescribing Practice Review PPR Managing type 2 diabetes For General Practice Key messages Metformin should be considered in all patients
November 2001 N P S National Prescribing Service Limited PPR fifteen Prescribing Practice Review PPR Managing type 2 diabetes For General Practice Key messages Metformin should be considered in all patients
Empagliflozin: Role in Treatment Options for Patients with Type 2 Diabetes Mellitus
 Diabetes Ther (2017) 8:33 53 DOI 10.1007/s13300-016-0211-x REVIEW Empagliflozin: Role in Treatment Options for Patients with Type 2 Diabetes Mellitus John E. Anderson. Eugene E. Wright Jr.. Charles F.
Diabetes Ther (2017) 8:33 53 DOI 10.1007/s13300-016-0211-x REVIEW Empagliflozin: Role in Treatment Options for Patients with Type 2 Diabetes Mellitus John E. Anderson. Eugene E. Wright Jr.. Charles F.
Supplementary Online Content
 Supplementary Online Content Tangri N, Stevens LA, Griffith J, et al. A predictive model for progression of chronic kidney disease to kidney failure. JAMA. 2011;305(15):1553-1559. eequation. Applying the
Supplementary Online Content Tangri N, Stevens LA, Griffith J, et al. A predictive model for progression of chronic kidney disease to kidney failure. JAMA. 2011;305(15):1553-1559. eequation. Applying the
Disclosures. Diabetes and Cardiovascular Risk Management. Learning Objectives. Atherosclerotic Cardiovascular Disease
 Disclosures Diabetes and Cardiovascular Risk Management Tony Hampton, MD, MBA Medical Director Advocate Aurora Operating System Advocate Aurora Healthcare Downers Grove, IL No conflicts or disclosures
Disclosures Diabetes and Cardiovascular Risk Management Tony Hampton, MD, MBA Medical Director Advocate Aurora Operating System Advocate Aurora Healthcare Downers Grove, IL No conflicts or disclosures
Supplementary Table 1. Baseline Characteristics by Quintiles of Systolic and Diastolic Blood Pressures
 Supplementary Data Supplementary Table 1. Baseline Characteristics by Quintiles of Systolic and Diastolic Blood Pressures Quintiles of Systolic Blood Pressure Quintiles of Diastolic Blood Pressure Q1 Q2
Supplementary Data Supplementary Table 1. Baseline Characteristics by Quintiles of Systolic and Diastolic Blood Pressures Quintiles of Systolic Blood Pressure Quintiles of Diastolic Blood Pressure Q1 Q2
Antihypertensive Trial Design ALLHAT
 1 U.S. Department of Health and Human Services Major Outcomes in High Risk Hypertensive Patients Randomized to Angiotensin-Converting Enzyme Inhibitor or Calcium Channel Blocker vs Diuretic National Institutes
1 U.S. Department of Health and Human Services Major Outcomes in High Risk Hypertensive Patients Randomized to Angiotensin-Converting Enzyme Inhibitor or Calcium Channel Blocker vs Diuretic National Institutes
Condition/Procedure Measure Compliance Criteria Reference Attribution Method
 Premium Specialty: Cardiology Credentialed Specialties include: Cardiac Diagnostic, Cardiology, Cardiovascular Disease, Clinical Cardiac Electrophysiology, and Interventional Cardiology This document is
Premium Specialty: Cardiology Credentialed Specialties include: Cardiac Diagnostic, Cardiology, Cardiovascular Disease, Clinical Cardiac Electrophysiology, and Interventional Cardiology This document is
Diabetes and Hypertension
 Diabetes and Hypertension M.Nakhjvani,M.D Tehran University of Medical Sciences 20-8-96 Hypertension Common DM comorbidity Prevalence depends on diabetes type, age, BMI, ethnicity Major risk factor for
Diabetes and Hypertension M.Nakhjvani,M.D Tehran University of Medical Sciences 20-8-96 Hypertension Common DM comorbidity Prevalence depends on diabetes type, age, BMI, ethnicity Major risk factor for
Metabolic Syndrome and Chronic Kidney Disease
 Metabolic Syndrome and Chronic Kidney Disease Definition of Metabolic Syndrome National Cholesterol Education Program (NCEP) Adult Treatment Panel (ATP) III Abdominal obesity, defined as a waist circumference
Metabolic Syndrome and Chronic Kidney Disease Definition of Metabolic Syndrome National Cholesterol Education Program (NCEP) Adult Treatment Panel (ATP) III Abdominal obesity, defined as a waist circumference
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Serra AL, Poster D, Kistler AD, et al. Sirolimus and kidney
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Serra AL, Poster D, Kistler AD, et al. Sirolimus and kidney
Long-Term Care Updates
 Long-Term Care Updates January 2016 By Yunuo (Enora) Wu, PharmD Chronic kidney disease (CKD) is defined as kidney damage (including structural or functional abnormalities) or glomerular filtration rate
Long-Term Care Updates January 2016 By Yunuo (Enora) Wu, PharmD Chronic kidney disease (CKD) is defined as kidney damage (including structural or functional abnormalities) or glomerular filtration rate
Prevention And Treatment of Diabetic Nephropathy. MOH Clinical Practice Guidelines 3/2006 Dr Stephen Chew Tec Huan
 Prevention And Treatment of Diabetic Nephropathy MOH Clinical Practice Guidelines 3/2006 Dr Stephen Chew Tec Huan Prevention Tight glucose control reduces the development of diabetic nephropathy Progression
Prevention And Treatment of Diabetic Nephropathy MOH Clinical Practice Guidelines 3/2006 Dr Stephen Chew Tec Huan Prevention Tight glucose control reduces the development of diabetic nephropathy Progression
Individual Study Table Referring to Part of Dossier: Volume: Page:
 Synopsis Abbott Laboratories Name of Study Drug: Paricalcitol Capsules (ABT-358) (Zemplar ) Name of Active Ingredient: Paricalcitol Individual Study Table Referring to Part of Dossier: Volume: Page: (For
Synopsis Abbott Laboratories Name of Study Drug: Paricalcitol Capsules (ABT-358) (Zemplar ) Name of Active Ingredient: Paricalcitol Individual Study Table Referring to Part of Dossier: Volume: Page: (For
It is well understood that patients with type 2 diabetes (T2D) are
 INFORMATION for the PHARMACIST This article was sponsored by Boehringer Ingelheim and Lilly. Addressing the Risk of Cardiovascular Mortality in Adult Patients with Type 2 Diabetes and Established Cardiovascular
INFORMATION for the PHARMACIST This article was sponsored by Boehringer Ingelheim and Lilly. Addressing the Risk of Cardiovascular Mortality in Adult Patients with Type 2 Diabetes and Established Cardiovascular
Empagliflozin and Progression of Kidney Disease in Type 2 Diabetes
 The new england journal of medicine Original Article and Progression of Kidney Disease in Type 2 Diabetes Christoph Wanner, M.D., Silvio E. Inzucchi, M.D., John M. Lachin, Sc.D., David Fitchett, M.D.,
The new england journal of medicine Original Article and Progression of Kidney Disease in Type 2 Diabetes Christoph Wanner, M.D., Silvio E. Inzucchi, M.D., John M. Lachin, Sc.D., David Fitchett, M.D.,
1. Albuminuria an early sign of glomerular damage and renal disease. albuminuria
 1. Albuminuria an early sign of glomerular damage and renal disease albuminuria Cardio-renal continuum REGRESS Target organ damage Asymptomatic CKD New risk factors Atherosclerosis Target organ damage
1. Albuminuria an early sign of glomerular damage and renal disease albuminuria Cardio-renal continuum REGRESS Target organ damage Asymptomatic CKD New risk factors Atherosclerosis Target organ damage
SUPPLEMENTARY DATA. Supplementary Figure S1. Cohort definition flow chart.
 Supplementary Figure S1. Cohort definition flow chart. Supplementary Table S1. Baseline characteristics of study population grouped according to having developed incident CKD during the follow-up or not
Supplementary Figure S1. Cohort definition flow chart. Supplementary Table S1. Baseline characteristics of study population grouped according to having developed incident CKD during the follow-up or not
Hypertension Update Clinical Controversies Regarding Age and Race
 Hypertension Update Clinical Controversies Regarding Age and Race Allison Helmer, PharmD, BCACP Assistant Clinical Professor Auburn University Harrison School of Pharmacy July 22, 2017 DISCLOSURE/CONFLICT
Hypertension Update Clinical Controversies Regarding Age and Race Allison Helmer, PharmD, BCACP Assistant Clinical Professor Auburn University Harrison School of Pharmacy July 22, 2017 DISCLOSURE/CONFLICT
Renal Protection Staying on Target
 Update Staying on Target James Barton, MD, FRCPC As presented at the University of Saskatchewan's Management of Diabetes & Its Complications (May 2004) Gwen s case Gwen, 49, asks you to take on her primary
Update Staying on Target James Barton, MD, FRCPC As presented at the University of Saskatchewan's Management of Diabetes & Its Complications (May 2004) Gwen s case Gwen, 49, asks you to take on her primary
Trial to Reduce. Aranesp* Therapy. Cardiovascular Events with
 Trial to Reduce Cardiovascular Events with Aranesp* Therapy John J.V. McMurray, Hajime Uno, Petr Jarolim, Akshay S. Desai, Dick de Zeeuw, Kai-Uwe Eckardt, Peter Ivanovich, Andrew S. Levey, Eldrin F. Lewis,
Trial to Reduce Cardiovascular Events with Aranesp* Therapy John J.V. McMurray, Hajime Uno, Petr Jarolim, Akshay S. Desai, Dick de Zeeuw, Kai-Uwe Eckardt, Peter Ivanovich, Andrew S. Levey, Eldrin F. Lewis,
Stages of Chronic Kidney Disease (CKD)
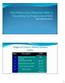 Early Treatment is the Key Stages of Chronic Kidney Disease (CKD) Stage Description GFR (ml/min/1.73 m 2 ) >90 1 Kidney damage with normal or GFR 2 Mild decrease in GFR 60-89 3 Moderate decrease in GFR
Early Treatment is the Key Stages of Chronic Kidney Disease (CKD) Stage Description GFR (ml/min/1.73 m 2 ) >90 1 Kidney damage with normal or GFR 2 Mild decrease in GFR 60-89 3 Moderate decrease in GFR
JNC Evidence-Based Guidelines for the Management of High Blood Pressure in Adults
 JNC 8 2014 Evidence-Based Guidelines for the Management of High Blood Pressure in Adults Table of Contents Why Do We Treat Hypertension? Blood Pressure Treatment Goals Initial Therapy Strength of Recommendation
JNC 8 2014 Evidence-Based Guidelines for the Management of High Blood Pressure in Adults Table of Contents Why Do We Treat Hypertension? Blood Pressure Treatment Goals Initial Therapy Strength of Recommendation
Effect of Saxagliptin on Renal Outcomes in the SAVOR TIMI- 53 study- Appendixes:
 Effect of Saxagliptin on Renal Outcomes in the SAVOR TIMI- 53 study- Appendixes: Appendix Table 1: Number and % of patients at the saxagliptin and placebo arms, according to egfr and on treatment ACR groups
Effect of Saxagliptin on Renal Outcomes in the SAVOR TIMI- 53 study- Appendixes: Appendix Table 1: Number and % of patients at the saxagliptin and placebo arms, according to egfr and on treatment ACR groups
BASELINE CHARACTERISTICS OF THE STUDY POPULATION
 Study Summary DAILY ORAL SODIUM BICARBONATE PRESERVES GLOMERULAR FILTRATION RATE BY SLOWING ITS DECLINE IN EARLY HYPERTENSIVE NEPHROPATHY This was a 5-year, single-center, prospective, randomized, placebo-controlled,
Study Summary DAILY ORAL SODIUM BICARBONATE PRESERVES GLOMERULAR FILTRATION RATE BY SLOWING ITS DECLINE IN EARLY HYPERTENSIVE NEPHROPATHY This was a 5-year, single-center, prospective, randomized, placebo-controlled,
Online Appendix (JACC )
 Beta blockers in Heart Failure Collaborative Group Online Appendix (JACC013117-0413) Heart rate, heart rhythm and prognostic effect of beta-blockers in heart failure: individual-patient data meta-analysis
Beta blockers in Heart Failure Collaborative Group Online Appendix (JACC013117-0413) Heart rate, heart rhythm and prognostic effect of beta-blockers in heart failure: individual-patient data meta-analysis
Adult Diabetes Clinician Guide NOVEMBER 2017
 Kaiser Permanente National CLINICAL PRACTICE GUIDELINES Adult Diabetes Clinician Guide Introduction NOVEMBER 2017 This evidence-based guideline summary is based on the 2017 KP National Diabetes Guideline.
Kaiser Permanente National CLINICAL PRACTICE GUIDELINES Adult Diabetes Clinician Guide Introduction NOVEMBER 2017 This evidence-based guideline summary is based on the 2017 KP National Diabetes Guideline.
QUICK REFERENCE FOR HEALTHCARE PROVIDERS
 KEY MESSAGES 1 SCREENING CRITERIA Screen: Patients with DM and/or hypertension at least yearly. Consider screening patients with: Age >65 years old Family history of stage 5 CKD or hereditary kidney disease
KEY MESSAGES 1 SCREENING CRITERIA Screen: Patients with DM and/or hypertension at least yearly. Consider screening patients with: Age >65 years old Family history of stage 5 CKD or hereditary kidney disease
CKD FOR INTERNISTS. Dr Ahmed Hossain Associate professor Medicine Sir Salimullah Medical College
 CKD FOR INTERNISTS Dr Ahmed Hossain Associate professor Medicine Sir Salimullah Medical College INTRODUCTION In 2002, the National Kidney Foundation s Kidney Disease Outcomes Quality Initiative(KDOQI)
CKD FOR INTERNISTS Dr Ahmed Hossain Associate professor Medicine Sir Salimullah Medical College INTRODUCTION In 2002, the National Kidney Foundation s Kidney Disease Outcomes Quality Initiative(KDOQI)
LEADER Liraglutide and cardiovascular outcomes in type 2 diabetes
 LEADER Liraglutide and cardiovascular outcomes in type 2 diabetes Presented at DSBS seminar on mediation analysis August 18 th Søren Rasmussen, Novo Nordisk. LEADER CV outcome study To determine the effect
LEADER Liraglutide and cardiovascular outcomes in type 2 diabetes Presented at DSBS seminar on mediation analysis August 18 th Søren Rasmussen, Novo Nordisk. LEADER CV outcome study To determine the effect
STABILITY Stabilization of Atherosclerotic plaque By Initiation of darapladib TherapY. Harvey D White on behalf of The STABILITY Investigators
 STABILITY Stabilization of Atherosclerotic plaque By Initiation of darapladib TherapY Harvey D White on behalf of The STABILITY Investigators Lipoprotein- associated Phospholipase A 2 (Lp-PLA 2 ) activity:
STABILITY Stabilization of Atherosclerotic plaque By Initiation of darapladib TherapY Harvey D White on behalf of The STABILITY Investigators Lipoprotein- associated Phospholipase A 2 (Lp-PLA 2 ) activity:
ADVANCE post trial ObservatioNal Study
 Hot Topics in Diabetes 50 th EASD, Vienna 2014 ADVANCE post trial ObservatioNal Study Sophia Zoungas The George Institute The University of Sydney Rationale and Study Design Sophia Zoungas The George Institute
Hot Topics in Diabetes 50 th EASD, Vienna 2014 ADVANCE post trial ObservatioNal Study Sophia Zoungas The George Institute The University of Sydney Rationale and Study Design Sophia Zoungas The George Institute
Clinical Trial Synopsis TL-OPI-525, NCT#
 Clinical Trial Synopsis, NCT#00762736 Title of Study: A Phase II, Double-Blind, Randomized, Placebo-Controlled, Proof-of-Concept Study of the Efficacy, Safety, and Tolerability of Pioglitazone HCl (ACTOS
Clinical Trial Synopsis, NCT#00762736 Title of Study: A Phase II, Double-Blind, Randomized, Placebo-Controlled, Proof-of-Concept Study of the Efficacy, Safety, and Tolerability of Pioglitazone HCl (ACTOS
DISCLOSURES OUTLINE OUTLINE 9/29/2014 ANTI-HYPERTENSIVE MANAGEMENT OF CHRONIC KIDNEY DISEASE
 ANTI-HYPERTENSIVE MANAGEMENT OF CHRONIC KIDNEY DISEASE DISCLOSURES Editor-in-Chief- Nephrology- UpToDate- (Wolters Klewer) Richard J. Glassock, MD, MACP Geffen School of Medicine at UCLA 1 st Annual Internal
ANTI-HYPERTENSIVE MANAGEMENT OF CHRONIC KIDNEY DISEASE DISCLOSURES Editor-in-Chief- Nephrology- UpToDate- (Wolters Klewer) Richard J. Glassock, MD, MACP Geffen School of Medicine at UCLA 1 st Annual Internal
VA/DoD Clinical Practice Guideline for the Diagnosis and Management of Hypertension - Pocket Guide Update 2004 Revision July 2005
 VA/DoD Clinical Practice Guideline for the Diagnosis and Management of Hypertension - Pocket Guide Update 2004 Revision July 2005 1 Any adult in the health care system 2 Obtain blood pressure (BP) (Reliable,
VA/DoD Clinical Practice Guideline for the Diagnosis and Management of Hypertension - Pocket Guide Update 2004 Revision July 2005 1 Any adult in the health care system 2 Obtain blood pressure (BP) (Reliable,
Supplementary Online Content
 Supplementary Online Content Larsen JR, Vedtofte L, Jakobsen MSL, et al. Effect of liraglutide treatment on prediabetes and overweight or obesity in clozapine- or olanzapine-treated patients with schizophrenia
Supplementary Online Content Larsen JR, Vedtofte L, Jakobsen MSL, et al. Effect of liraglutide treatment on prediabetes and overweight or obesity in clozapine- or olanzapine-treated patients with schizophrenia
New Hypertension Guideline Recommendations for Adults July 7, :45-9:30am
 Advances in Cardiovascular Disease 30 th Annual Convention and Reunion UERM-CMAA, Inc. Annual Convention and Scientific Meeting July 5-8, 2018 New Hypertension Guideline Recommendations for Adults July
Advances in Cardiovascular Disease 30 th Annual Convention and Reunion UERM-CMAA, Inc. Annual Convention and Scientific Meeting July 5-8, 2018 New Hypertension Guideline Recommendations for Adults July
Cedars Sinai Diabetes. Michael A. Weber
 Cedars Sinai Diabetes Michael A. Weber Speaker Disclosures I disclose that I am a Consultant for: Ablative Solutions, Boston Scientific, Boehringer Ingelheim, Eli Lilly, Forest, Medtronics, Novartis, ReCor
Cedars Sinai Diabetes Michael A. Weber Speaker Disclosures I disclose that I am a Consultant for: Ablative Solutions, Boston Scientific, Boehringer Ingelheim, Eli Lilly, Forest, Medtronics, Novartis, ReCor
CANVAS Program Independent commentary
 CANVAS Program Independent commentary Cliff Bailey Aston University, Birmingham, UK 2017 Disclosures and disclaimers Clifford J Bailey CJB has attended advisory boards, undertaken ad hoc consultancy, received
CANVAS Program Independent commentary Cliff Bailey Aston University, Birmingham, UK 2017 Disclosures and disclaimers Clifford J Bailey CJB has attended advisory boards, undertaken ad hoc consultancy, received
A n aly tical m e t h o d s
 a A n aly tical m e t h o d s If I didn t go to the screening at Farmers Market I would not have known about my kidney problems. I am grateful to the whole staff. They were very professional. Thank you.
a A n aly tical m e t h o d s If I didn t go to the screening at Farmers Market I would not have known about my kidney problems. I am grateful to the whole staff. They were very professional. Thank you.
Long-Term Complications of Diabetes Mellitus Macrovascular Complication
 Long-Term Complications of Diabetes Mellitus Macrovascular Complication Sung Hee Choi MD, PhD Professor, Seoul National University College of Medicine, SNUBH, Bundang Hospital Diabetes = CVD equivalent
Long-Term Complications of Diabetes Mellitus Macrovascular Complication Sung Hee Choi MD, PhD Professor, Seoul National University College of Medicine, SNUBH, Bundang Hospital Diabetes = CVD equivalent
Managing Chronic Kidney Disease: Reducing Risk for CKD Progression
 Managing Chronic Kidney Disease: Reducing Risk for CKD Progression Arasu Gopinath, MD Clinical Nephrologist, Medical Director, Jordan Landing Dialysis Center Objectives: Identify the most important risks
Managing Chronic Kidney Disease: Reducing Risk for CKD Progression Arasu Gopinath, MD Clinical Nephrologist, Medical Director, Jordan Landing Dialysis Center Objectives: Identify the most important risks
Drug Class Monograph
 Drug Class Monograph Class: Sodium-Glucose Co-Transporter 2 (SGLT-2) Inhibitors Drugs: Farxiga (dapagliflozin), Invokamet (canagliflozin/metformin), Invokana (canagliflozin), Jardiance (empagliflozin),
Drug Class Monograph Class: Sodium-Glucose Co-Transporter 2 (SGLT-2) Inhibitors Drugs: Farxiga (dapagliflozin), Invokamet (canagliflozin/metformin), Invokana (canagliflozin), Jardiance (empagliflozin),
American Academy of Insurance Medicine
 American Academy of Insurance Medicine October 2012 Dr. Alison Moy Liberty Mutual Dr. John Kirkpatrick Thrivent Financial for Lutherans 1 59 year old male, diagnosed with T2DM six months ago Nonsmoker
American Academy of Insurance Medicine October 2012 Dr. Alison Moy Liberty Mutual Dr. John Kirkpatrick Thrivent Financial for Lutherans 1 59 year old male, diagnosed with T2DM six months ago Nonsmoker
SUPPLEMENTARY DATA. Supplementary Table 1. Baseline Patient Characteristics
 Supplementary Table 1. Baseline Patient Characteristics Normally distributed data are presented as mean (±SD), data that were not of a normal distribution are presented as median (ICR). The baseline characteristics
Supplementary Table 1. Baseline Patient Characteristics Normally distributed data are presented as mean (±SD), data that were not of a normal distribution are presented as median (ICR). The baseline characteristics
Cardiovascular Diabetology. Open Access ORIGINAL INVESTIGATION. C. R. L. Cardoso 1, N. C. Leite 1, C. B. M. Moram 2 and G. F.
 https://doi.org/10.1186/s12933-018-0677-0 Cardiovascular Diabetology ORIGINAL INVESTIGATION Open Access Long term visit to visit glycemic variability as predictor of micro and macrovascular complications
https://doi.org/10.1186/s12933-018-0677-0 Cardiovascular Diabetology ORIGINAL INVESTIGATION Open Access Long term visit to visit glycemic variability as predictor of micro and macrovascular complications
Diabetes in Renal Patients. Contents. Understanding Diabetic Nephropathy
 Diabetes in Renal Patients Contents Understanding Diabetic Nephropathy What effect does CKD have on a patient s diabetic control? Diabetic Drugs in CKD and Dialysis Patients Hyper and Hypoglycaemia in
Diabetes in Renal Patients Contents Understanding Diabetic Nephropathy What effect does CKD have on a patient s diabetic control? Diabetic Drugs in CKD and Dialysis Patients Hyper and Hypoglycaemia in
CHRONIC RENAL FAILURE: WHAT THE PRIMARY CARE CAN OFFER. The annual conference of the Lebanese Society of Family Medicine October 2017 Dr Hiba AZAR
 CHRONIC RENAL FAILURE: WHAT THE PRIMARY CARE CAN OFFER The annual conference of the Lebanese Society of Family Medicine October 2017 Dr Hiba AZAR OUTLINE: A journey through CKD Screening for CKD: The why,
CHRONIC RENAL FAILURE: WHAT THE PRIMARY CARE CAN OFFER The annual conference of the Lebanese Society of Family Medicine October 2017 Dr Hiba AZAR OUTLINE: A journey through CKD Screening for CKD: The why,
BASELINE CHARACTERISTICS OF THE STUDY POPULATION
 COMPARISON OF TREATING METABOLIC ACIDOSIS IN CKD STAGE 4 HYPERTENSIVE KIDNEY DISEASE WITH FRUITS & VEGETABLES OR SODIUM BICARBONATE This was a 1-year, single-center, prospective, randomized, interventional
COMPARISON OF TREATING METABOLIC ACIDOSIS IN CKD STAGE 4 HYPERTENSIVE KIDNEY DISEASE WITH FRUITS & VEGETABLES OR SODIUM BICARBONATE This was a 1-year, single-center, prospective, randomized, interventional
Index. Note: Page numbers of article titles are in boldface type.
 Heart Failure Clin 2 (2006) 101 105 Index Note: Page numbers of article titles are in boldface type. A ACE inhibitors, in diabetic hypertension, 30 31 Adipokines, cardiovascular events related to, 6 Advanced
Heart Failure Clin 2 (2006) 101 105 Index Note: Page numbers of article titles are in boldface type. A ACE inhibitors, in diabetic hypertension, 30 31 Adipokines, cardiovascular events related to, 6 Advanced
The Diabetes Kidney Disease Connection Missouri Foundation for Health February 26, 2009
 The Diabetes Kidney Disease Connection Missouri Foundation for Health February 26, 2009 Teresa Northcutt, RN BSN Primaris Program Manager, Prevention - CKD MO-09-01-CKD This material was prepared by Primaris,
The Diabetes Kidney Disease Connection Missouri Foundation for Health February 26, 2009 Teresa Northcutt, RN BSN Primaris Program Manager, Prevention - CKD MO-09-01-CKD This material was prepared by Primaris,
Diabetes and Kidney Disease. Kris Bentley Renal Nurse practitioner 2018
 Diabetes and Kidney Disease Kris Bentley Renal Nurse practitioner 2018 Aims Develop an understanding of Chronic Kidney Disease Understand how diabetes impacts on your kidneys Be able to recognise the risk
Diabetes and Kidney Disease Kris Bentley Renal Nurse practitioner 2018 Aims Develop an understanding of Chronic Kidney Disease Understand how diabetes impacts on your kidneys Be able to recognise the risk
The CARI Guidelines Caring for Australasians with Renal Impairment. ACE Inhibitor and Angiotensin II Antagonist Combination Treatment GUIDELINES
 ACE Inhibitor and Angiotensin II Antagonist Combination Treatment Date written: September 2004 Final submission: September 2005 Author: Kathy Nicholls GUIDELINES No recommendations possible based on Level
ACE Inhibitor and Angiotensin II Antagonist Combination Treatment Date written: September 2004 Final submission: September 2005 Author: Kathy Nicholls GUIDELINES No recommendations possible based on Level
Supplementary Online Content
 Supplementary Online Content Clark WF, Sontrop JM, Huang S-H et al. Effect of Coaching to Increase Water Intake on Kidney Function Decline in Adults With Chronic Kidney Disease: the CKD WIT Randomized
Supplementary Online Content Clark WF, Sontrop JM, Huang S-H et al. Effect of Coaching to Increase Water Intake on Kidney Function Decline in Adults With Chronic Kidney Disease: the CKD WIT Randomized
Safety profile of Liraglutide: Recent Updates. Mohammadreza Rostamzadeh,M.D.
 Safety profile of Liraglutide: Recent Updates Mohammadreza Rostamzadeh,M.D. Pancreatitis: Victoza post-marketing experience: spontaneous reports of pancreatitis For the majority of the cases, there is
Safety profile of Liraglutide: Recent Updates Mohammadreza Rostamzadeh,M.D. Pancreatitis: Victoza post-marketing experience: spontaneous reports of pancreatitis For the majority of the cases, there is
Supplementary Online Content
 Supplementary Online Content Pedersen SB, Langsted A, Nordestgaard BG. Nonfasting mild-to-moderate hypertriglyceridemia and risk of acute pancreatitis. JAMA Intern Med. Published online November 7, 2016.
Supplementary Online Content Pedersen SB, Langsted A, Nordestgaard BG. Nonfasting mild-to-moderate hypertriglyceridemia and risk of acute pancreatitis. JAMA Intern Med. Published online November 7, 2016.
Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes
 Original Article, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes Bernard Zinman, M.D., Christoph Wanner, M.D., John M. Lachin, Sc.D., David Fitchett, M.D., Erich Bluhmki, Ph.D., Stefan Hantel,
Original Article, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes Bernard Zinman, M.D., Christoph Wanner, M.D., John M. Lachin, Sc.D., David Fitchett, M.D., Erich Bluhmki, Ph.D., Stefan Hantel,
EDMS #4298 Version 1.0
 Randomized EValuation of the Effects of Anacetrapib through Lipid-modification (HPS3/TIMI55 REVEAL) Data Analysis Plan Version 1.0 1 Table of Contents 1 VERSION HISTORY... 2 2 INTRODUCTION... 3 2.1 BACKGROUND...
Randomized EValuation of the Effects of Anacetrapib through Lipid-modification (HPS3/TIMI55 REVEAL) Data Analysis Plan Version 1.0 1 Table of Contents 1 VERSION HISTORY... 2 2 INTRODUCTION... 3 2.1 BACKGROUND...
ABCD and Renal Association Clinical Guidelines for Diabetic Nephropathy-CKD. Management of Dyslipidaemia and Hypertension in Adults Dr Peter Winocour
 ABCD and Renal Association Clinical Guidelines for Diabetic Nephropathy-CKD. Management of Dyslipidaemia and Hypertension in Adults Dr Peter Winocour Dr Indranil Dasgupta Rationale No national practical
ABCD and Renal Association Clinical Guidelines for Diabetic Nephropathy-CKD. Management of Dyslipidaemia and Hypertension in Adults Dr Peter Winocour Dr Indranil Dasgupta Rationale No national practical
Kidney and heart: dangerous liaisons. Luis M. RUILOPE (Madrid, Spain)
 Kidney and heart: dangerous liaisons Luis M. RUILOPE (Madrid, Spain) Type 2 diabetes and renal disease: impact on cardiovascular outcomes The "heavyweights" of modifiable CVD risk factors Hypertension
Kidney and heart: dangerous liaisons Luis M. RUILOPE (Madrid, Spain) Type 2 diabetes and renal disease: impact on cardiovascular outcomes The "heavyweights" of modifiable CVD risk factors Hypertension
Chapter Two Renal function measures in the adolescent NHANES population
 0 Chapter Two Renal function measures in the adolescent NHANES population In youth acquire that which may restore the damage of old age; and if you are mindful that old age has wisdom for its food, you
0 Chapter Two Renal function measures in the adolescent NHANES population In youth acquire that which may restore the damage of old age; and if you are mindful that old age has wisdom for its food, you
ALLHAT RENAL DISEASE OUTCOMES IN HYPERTENSIVE PATIENTS STRATIFIED INTO 4 GROUPS BY BASELINE GLOMERULAR FILTRATION RATE (GFR)
 1 RENAL DISEASE OUTCOMES IN HYPERTENSIVE PATIENTS STRATIFIED INTO 4 GROUPS BY BASELINE GLOMERULAR FILTRATION RATE (GFR) 6 / 5 / 1006-1 2 Introduction Hypertension is the second most common cause of end-stage
1 RENAL DISEASE OUTCOMES IN HYPERTENSIVE PATIENTS STRATIFIED INTO 4 GROUPS BY BASELINE GLOMERULAR FILTRATION RATE (GFR) 6 / 5 / 1006-1 2 Introduction Hypertension is the second most common cause of end-stage
Multiphasic Blood Analysis
 Understanding Your Multiphasic Blood Analysis Test Results Mon General thanks you for participating in the multiphasic blood analysis. This test can be an early warning of health problems, including coronary
Understanding Your Multiphasic Blood Analysis Test Results Mon General thanks you for participating in the multiphasic blood analysis. This test can be an early warning of health problems, including coronary
Beta-blockers in Patients with Mid-range Left Ventricular Ejection Fraction after AMI Improved Clinical Outcomes
 Beta-blockers in Patients with Mid-range Left Ventricular Ejection Fraction after AMI Improved Clinical Outcomes Seung-Jae Joo and other KAMIR-NIH investigators Department of Cardiology, Jeju National
Beta-blockers in Patients with Mid-range Left Ventricular Ejection Fraction after AMI Improved Clinical Outcomes Seung-Jae Joo and other KAMIR-NIH investigators Department of Cardiology, Jeju National
S150 KEEP Analytical Methods. American Journal of Kidney Diseases, Vol 55, No 3, Suppl 2, 2010:pp S150-S153
 S150 KEEP 2009 Analytical Methods American Journal of Kidney Diseases, Vol 55, No 3, Suppl 2, 2010:pp S150-S153 S151 The Kidney Early Evaluation program (KEEP) is a free, communitybased health screening
S150 KEEP 2009 Analytical Methods American Journal of Kidney Diseases, Vol 55, No 3, Suppl 2, 2010:pp S150-S153 S151 The Kidney Early Evaluation program (KEEP) is a free, communitybased health screening
HYPERTENSION GUIDELINES WHERE ARE WE IN 2014
 HYPERTENSION GUIDELINES WHERE ARE WE IN 2014 Donald J. DiPette MD FACP Special Assistant to the Provost for Health Affairs Distinguished Health Sciences Professor University of South Carolina University
HYPERTENSION GUIDELINES WHERE ARE WE IN 2014 Donald J. DiPette MD FACP Special Assistant to the Provost for Health Affairs Distinguished Health Sciences Professor University of South Carolina University
Have you seen a patient like Elaine *?
 (linagliptin) 5mg tablets Have you seen a patient like Elaine *? *Hypothetical patient profile Elaine * : 60 years old Housewife *Hypothetical patient profile ELAINE*: T2D Patient with early signs of kidney
(linagliptin) 5mg tablets Have you seen a patient like Elaine *? *Hypothetical patient profile Elaine * : 60 years old Housewife *Hypothetical patient profile ELAINE*: T2D Patient with early signs of kidney
Lowering blood pressure (BP) in patients with type 2
 Average Clinician-Measured Blood Pressures and Cardiovascular Outcomes in Patients With Type 2 Diabetes Mellitus and Ischemic Heart Disease in the EXAMINE Trial William B. White, MD; Fatima Jalil, MD;
Average Clinician-Measured Blood Pressures and Cardiovascular Outcomes in Patients With Type 2 Diabetes Mellitus and Ischemic Heart Disease in the EXAMINE Trial William B. White, MD; Fatima Jalil, MD;
Supplement materials:
 Supplement materials: Table S1: ICD-9 codes used to define prevalent comorbid conditions and incident conditions Comorbid condition ICD-9 code Hypertension 401-405 Diabetes mellitus 250.x Myocardial infarction
Supplement materials: Table S1: ICD-9 codes used to define prevalent comorbid conditions and incident conditions Comorbid condition ICD-9 code Hypertension 401-405 Diabetes mellitus 250.x Myocardial infarction
The ABCs (A1C, BP and Cholesterol) of Diabetes
 The ABCs (A1C, BP and Cholesterol) of Diabetes Gregg Simonson, PhD Director, Professional Training and Consulting International Diabetes Center; Adjunct Assistant Professor, University of Minnesota Department
The ABCs (A1C, BP and Cholesterol) of Diabetes Gregg Simonson, PhD Director, Professional Training and Consulting International Diabetes Center; Adjunct Assistant Professor, University of Minnesota Department
Update on Current Trends in Hypertension Management
 Friday General Session Update on Current Trends in Hypertension Management Shawna Nesbitt, MD Associate Dean, Minority Student Affairs Associate Professor, Department of Internal Medicine Office of Student
Friday General Session Update on Current Trends in Hypertension Management Shawna Nesbitt, MD Associate Dean, Minority Student Affairs Associate Professor, Department of Internal Medicine Office of Student
Table 1 Baseline characteristics of 60 hemodialysis patients with atrial fibrillation and warfarin use
 Table 1 Baseline characteristics of 60 hemodialysis patients with atrial fibrillation and warfarin use Baseline characteristics Users (n = 28) Non-users (n = 32) P value Age (years) 67.8 (9.4) 68.4 (8.5)
Table 1 Baseline characteristics of 60 hemodialysis patients with atrial fibrillation and warfarin use Baseline characteristics Users (n = 28) Non-users (n = 32) P value Age (years) 67.8 (9.4) 68.4 (8.5)
SCIENTIFIC STUDY REPORT
 PAGE 1 18-NOV-2016 SCIENTIFIC STUDY REPORT Study Title: Real-Life Effectiveness and Care Patterns of Diabetes Management The RECAP-DM Study 1 EXECUTIVE SUMMARY Introduction: Despite the well-established
PAGE 1 18-NOV-2016 SCIENTIFIC STUDY REPORT Study Title: Real-Life Effectiveness and Care Patterns of Diabetes Management The RECAP-DM Study 1 EXECUTIVE SUMMARY Introduction: Despite the well-established
DISCLOSURE PHARMACIST OBJECTIVES 9/30/2014 JNC 8: A REVIEW OF THE LONG-AWAITED/MUCH-ANTICIPATED HYPERTENSION GUIDELINES. I have nothing to disclose.
 JNC 8: A REVIEW OF THE LONG-AWAITED/MUCH-ANTICIPATED HYPERTENSION GUIDELINES Tiffany Dickey, PharmD Assistant Professor, UAMS COP Clinical Pharmacy Specialist, Mercy Hospital Northwest AR DISCLOSURE I
JNC 8: A REVIEW OF THE LONG-AWAITED/MUCH-ANTICIPATED HYPERTENSION GUIDELINES Tiffany Dickey, PharmD Assistant Professor, UAMS COP Clinical Pharmacy Specialist, Mercy Hospital Northwest AR DISCLOSURE I
Cardiovascular Benefits of Two Classes of Antihyperglycemic Medications
 Cardiovascular Benefits of Two Classes of Antihyperglycemic Medications Nathan Woolever, Pharm.D., Resident Pharmacist Pharmacy Grand Rounds November 6 th, 2018 Franciscan Healthcare La Crosse, WI 2017
Cardiovascular Benefits of Two Classes of Antihyperglycemic Medications Nathan Woolever, Pharm.D., Resident Pharmacist Pharmacy Grand Rounds November 6 th, 2018 Franciscan Healthcare La Crosse, WI 2017
Treatment Of Preserved Cardiac Function Heart Failure with an Aldosterone antagonist (TOPCAT) AHA Nov 18, 2014 Update on Randomized Trials
 Treatment Of Preserved Cardiac Function Heart Failure with an Aldosterone antagonist (TOPCAT) AHA Nov 18, 2014 Update on Randomized Trials Marc A. Pfeffer, MD, PhD; Brian Claggett, PhD; Susan F. Assmann,
Treatment Of Preserved Cardiac Function Heart Failure with an Aldosterone antagonist (TOPCAT) AHA Nov 18, 2014 Update on Randomized Trials Marc A. Pfeffer, MD, PhD; Brian Claggett, PhD; Susan F. Assmann,
What s In the New Hypertension Guidelines?
 American College of Physicians Ohio/Air Force Chapters 2018 Scientific Meeting Columbus, OH October 5, 2018 What s In the New Hypertension Guidelines? Max C. Reif, MD, FACP Objectives: At the end of the
American College of Physicians Ohio/Air Force Chapters 2018 Scientific Meeting Columbus, OH October 5, 2018 What s In the New Hypertension Guidelines? Max C. Reif, MD, FACP Objectives: At the end of the
Sodium-Glucose Co-Transporter 2 (SGLT-2) Inhibitors Drug Class Prior Authorization Protocol
 Sodium-Glucose Co-Transporter 2 (SGLT-2) Inhibitors Drug Class Prior Authorization Protocol Line of Business: Medicaid P&T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has
Sodium-Glucose Co-Transporter 2 (SGLT-2) Inhibitors Drug Class Prior Authorization Protocol Line of Business: Medicaid P&T Approval Date: February 21, 2018 Effective Date: April 1, 2018 This policy has
Resistant hypertension (RH), that is, blood pressure (BP)
 Resistant Hypertension, Time-Updated Blood Pressure Values and Renal Outcome in Type 2 Diabetes Mellitus Francesca Viazzi, MD; Pamela Piscitelli, MD; Antonio Ceriello, MD; Paola Fioretto, MD; Carlo Giorda,
Resistant Hypertension, Time-Updated Blood Pressure Values and Renal Outcome in Type 2 Diabetes Mellitus Francesca Viazzi, MD; Pamela Piscitelli, MD; Antonio Ceriello, MD; Paola Fioretto, MD; Carlo Giorda,
Chemistry Reference Ranges and Critical Values
 Alanine Aminotransferase (ALT, SGPT) 3-9 years 9-18 years 1-9 years 9-18 years 10-25 U/L 10-35 U/L 10-30 U/L 10-25 U/L 10-30 U/L 10-35 U/L 10-25 U/L 10-35 U/L 10-25 U/L 10-20 U/L 10-35 U/L Albumin 0-6
Alanine Aminotransferase (ALT, SGPT) 3-9 years 9-18 years 1-9 years 9-18 years 10-25 U/L 10-35 U/L 10-30 U/L 10-25 U/L 10-30 U/L 10-35 U/L 10-25 U/L 10-35 U/L 10-25 U/L 10-20 U/L 10-35 U/L Albumin 0-6
Abbreviations DPP-IV dipeptidyl peptidase IV DREAM Diabetes REduction Assessment with ramipril and rosiglitazone
 Index Abbreviations DPP-IV dipeptidyl peptidase IV DREAM Diabetes REduction Assessment with ramipril and rosiglitazone Medication GAD glutamic acid decarboxylase GLP-1 glucagon-like peptide 1 NPH neutral
Index Abbreviations DPP-IV dipeptidyl peptidase IV DREAM Diabetes REduction Assessment with ramipril and rosiglitazone Medication GAD glutamic acid decarboxylase GLP-1 glucagon-like peptide 1 NPH neutral
hypertension Head of prevention and control of CVD disease office Ministry of heath
 hypertension t. Samavat MD,Cadiologist,MPH Head of prevention and control of CVD disease office Ministry of heath RECOMMENDATIONS FOR HYPERTENSION DIAGNOSIS, ASSESSMENT, AND TREATMENT Definition of hypertension
hypertension t. Samavat MD,Cadiologist,MPH Head of prevention and control of CVD disease office Ministry of heath RECOMMENDATIONS FOR HYPERTENSION DIAGNOSIS, ASSESSMENT, AND TREATMENT Definition of hypertension
Chemistry Reference Ranges and Critical Values
 Alanine Aminotransferase (ALT, SGPT) 3-9 years 9-18 years 1-9 years 9-18 years 10-30 U/L 10-30 U/L 10-20 U/L Albumin 0-6 days 6 days - 37 months 37 months - 7 years 7-20 years 2.6-3.6 g/dl 3.4-4.2 g/dl
Alanine Aminotransferase (ALT, SGPT) 3-9 years 9-18 years 1-9 years 9-18 years 10-30 U/L 10-30 U/L 10-20 U/L Albumin 0-6 days 6 days - 37 months 37 months - 7 years 7-20 years 2.6-3.6 g/dl 3.4-4.2 g/dl
Variability in drug response: towards more personalized diabetes care Petrykiv, Sergei
 University of Groningen Variability in drug response: towards more personalized diabetes care Petrykiv, Sergei IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you
University of Groningen Variability in drug response: towards more personalized diabetes care Petrykiv, Sergei IMPORTANT NOTE: You are advised to consult the publisher's version (publisher's PDF) if you

 www.usrds.org www.usrds.org 1 1,749 + (2,032) 1,563 to
www.usrds.org www.usrds.org 1 1,749 + (2,032) 1,563 to