(51) Int Cl.: A61K 9/00 ( ) A61K 31/573 ( ) A61P 27/02 ( )
|
|
|
- Marvin York
- 6 years ago
- Views:
Transcription
1 (19) TEPZZ 4ZZ_4B_T (11) EP B1 (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent: Bulletin 13/17 (21) Application number: (22) Date of filing: (1) Int Cl.: A61K 9/00 (06.01) A61K 31/73 (06.01) A61P 27/02 (06.01) (86) International application number: PCT/JP/ (87) International publication number: WO 11/ ( Gazette 11/03) (4) EYE DROP WITH DIFLUPREDNATE FOR MACULAR EDEMA TREATMENT AUGENTROPFEN MIT DIFLUPREDNAT ZUR BEHANDLUNG VON MAKULAÖDEM COLLYRE À BASE DE DIFLUPREDNATE POUR LE TRAITEMENT DE LOEDÈME MACULAIRE (84) Designated Contracting States: AL AT BE BG CH CY CZ DE DK EE ES FI FR GB GR HR HU IE IS IT LI LT LU LV MC MK MT NL NO PL PT RO SE SI SK SM TR (74) Representative: von Kreisler Selting Werner Deichmannhaus am Dom Bahnhofsvorplatz Köln (DE) EP B1 () Priority: JP (43) Date of publication of application: Bulletin 11/27 (73) Proprietor: Yamagata University Yamagata-shi, Yamagata (JP) (72) Inventors: YAMASHITA, Hidetoshi Yamagata-shi Yamagata (JP) YAMAMOTO, Teiko Toshima-ku, Tokyo (JP) GOTO, Sakiko Yamagata-shi Yamagata (JP) ABE, Sachi Yamagata-shi Yamagata (JP) KIRII, Eriko Yamagata-shi Yamagata (JP) OKUMURA, Atsushi Suita-shi Osaka, 602 (JP) (6) References cited: EP-A WO-A1-0/ DIFLUPREDNATE OPHTHALMIC EMULSION 0 0% (DUREZOL) STUDY GROUP ET AL: "Difluprednate ophthalmic emulsion 0.0% for postoperative inflammation and pain" JOURNAL CATARACT AND REFRACTIVE SURGERY, SURGERY, FAIRFAX, VA LNKD- DOI:.16/J.JCRS , vol. 3, no. 1, 1 January 09 ( ), pages 26-34, XP ISSN: [retrieved on ] MARTIDIS ADAM ET AL: "Intravitreal triamcinolone for refractory diabetic macular edema." May 02 (02-0), OPHTHALMOLOGY MAY 02 LNKD- PUBMED: , VOL. 9, NR., PAGE(S) 9-927, XP ISSN: page 926, right-hand column, last paragraph SAKIKO NAKANO ET AL: "Steroid eye drop treatment (difluprednate ophthalmic emulsion) is effective in reducing refractory diabetic macular edema" GRAEFE S ARCHIVE FOR CLINICAL AND EXPERIMENTAL OPHTHALMOLOGY ; INCORPORATING GERMAN JOURNAL OF OPHTHALMOLOGY, SPRINGER, BERLIN, DE, vol. 248, no. 6, 24 February (-02-24), pages 80-8, XP ISSN: X Note: Within nine months of the publication of the mention of the grant of the European patent in the European Patent Bulletin, any person may give notice to the European Patent Office of opposition to that patent, in accordance with the Implementing Regulations. Notice of opposition shall not be deemed to have been filed until the opposition fee has been paid. (Art. 99(1) European Patent Convention). Printed by Jouve, 7001 PARIS (FR)
2 Description TECHNICAL FIELD OF THE INVENTION [0001] The present invention relates to an eye drop containing difluprednate as an active ingredient for use in the treatment of macular edema. BACKGROUND OF THE INVENTION [0002] Macular edema is swelling of retinal macula, and the edema occurs due to a liquid diapedesis from the retinal blood vessels. The blood leaks from weak blood vessel walls and enters into extremely small regions of retinal macula rich in retina cone which is a nerve terminal that detects color and the vision during the day relies on. Next, images become blurred in the center of the central field or right beside the center. The visual acuity decreases progressively for months. Diabetic retinopathy, retinal blood vessel obstruction, ocular inflammation and age-related macular degeneration are all associated with the macular edema. Retinal macula is sometimes damaged by maculatumentia after removal of crystalline lens for the treatment of cataract. As the conventional therapy of macular edema, photocoagulation by laser irradiation, vitreous surgery and systemic administration, intravitreal administration and sub-tenon administration of steroid and the like have been performed. The photocoagulation by laser irradiation closes the blood vessel permitting liquid diapedesis, and decreases swelling of macula. However, attention should be paid in laser irradiation to avoid extremely vulnerable fovea. If the fovea should be injured by this surgery, the central visual field may be damaged. Moreover, plural laser surgeries are often required to eliminate swelling. While vitreous surgery is applied to a case for which a laser surgery is ineffective, it is associated with high tissue-invasive potential, sometimes causing problems of post-surgery complications. In addition, the administration of steroid is reported to be useful. While systemic administration of steroid is possible for the treatment of ocular diseases, in general, it often causes side effects which are too severe for ophthalmologic uses. Therefore, intravitreal administration and sub-tenon administration, which are topical administrations, have also been studied. Although intravitreal administration can solve some problems associated with systemic administration, intravitreal administration of existing ophthalmic compositions can cause ocular hypertension, steroid glaucoma and posterior subcapsular cataract when steroid is administered. Also, intravitreal administration of steroid sometimes causes post-surgery complications. sub-tenon administration is often used in clinical practice to decrease the tissue-invasive potential of intravitreal administration and burden on patients. While administration of steroid decreases the tissue-invasive potential as compared to vitreous surgery, it is still associated with the problems of post-surgery complications. Administration by instillation is an administration method with high merit since it has low tissue-invasive potential. Examples of the treatment of ophthalmic diseases by instillation of steroid include use of a 0.1% betamethasone ophthalmic solution for anti-inflammatory diseases (blepharitis, conjunctivitis, keratitis, scleritis, episcleritis, anterior ocular segmentuveitis, postoperative inflammation) in the external eye and the anterior ocular segment. Moreover, WO 07/0227 describes the possibility of application of instillation and the like of various steroids and corticosteroid antagonists to the treatment of various ophthalmic diseases such as macular degeneration, glaucoma, macular edema, age-related macular degeneration, retina angiogenesis, diabetic retinopathy, iritis, posterior eye segmentuveitis and the like, while decreasing the side effects of steroid. However, only few cases of effectiveness of instillation for macular edema of the retina in clinical practice have been reported, and there is only one report of volume reduction of retinal macular edema by administration of 0.1% betamethasone ophthalmic solution for 2 weeks to one month, 6 times per day (The 43rd Annual Congress of Japanese Retina and Vitreous Society 016-, 04). Thus, the treatment of macular edema by eye drop has not been performed. SUMMARY OF THE INVENTION Problems to be Solved by the Invention 0 [0003] The present invention aims to provide an eye drop for the treatment of macular edema. Means of Solving the Problems [0004] The present inventors have conducted intensive studies in an attempt to solve the aforementioned problems and found that administration of an eye drop containing difluprednate as an active ingredient improves the symptoms of decreased visual acuity, increased foveal retinal thickness and the like due to macular edema, which resulted in the completion of the present invention. [000] Accordingly, the present invention provides the following: 2
3 1 (1) An eye drop containing difluprednate for use in the treatment of macular edema. (2) The eye drop for use in the treatment according to (1), wherein the macular edema is refractory macular edema. (3) The eye drop for use in the treatment according to (1) or (2), which is an emulsion eye drop. (4) The eye drop for use in the treatment according to any one of (1) to (3), wherein the concentration of difluprednate is % (w/v). () The eye drop for use in the treatment according to any one of (1) to (4), comprising % (w/v) of difluprednate, and castor oil, polysorbate 80, concentrated glycerin, sodium acetate, boric acid, sodium edetate and sorbic acid. (6) The eye drop for use in the treatment according to any one of (1) to (), wherein the concentration of difluprednate is % (w/v), and the eye drop is administered for at least 1 week, 4 times per day, in a dose of - 0 ml per administration. (6) The eye drop for use in the treatment according to any one of (1) to(), wherein the concentration of difluprednate is % (w/v), and the eye drop is administered for 1-4 weeks, 4 times per day, in a dose of - 0 ml per administration. (8) The eye drop for use in the treatment according to any one of (1) to (), wherein the concentration of difluprednate is % (w/v), and the eye drop is administered 4 times per day for 4 weeks from the start of the administration and twice per day after 4 weeks, in a dose of - 0 ml per administration. (9) The eye drop for use in the treatment according to any one of (1) to (), wherein the concentration of difluprednate is % (w/v), and the eye drop is administered for not longer than 12 weeks, 4 times per day for 4 weeks from the start of the administration and twice per day after 4 weeks, in a dose of - 0 ml per administration. () The eye drop for use in the treatment according to any one of (1) to (), wherein the concentration of difluprednate is % (w/v), and the eye drop is administered for at least 12 weeks, 4 times per day for 4 weeks from the start of the administration and twice per day after 4 weeks, in a dose of - 0 ml per administration. 2 Effect of the Invention [0006] According to the present invention, macular edema can be treated with an eye drop. Particularly, the eye drop of the present invention is useful for treating refractory macular edema. EMBODIMENT OF THE INVENTION [0007] The present invention relates to an eye drop for use in the treatment macular edema, which eye drop contains difluprednate as an active ingredient, [0008] Difluprednate (6α,9α-difluoroprednisolone 17-butyrate 21-acetate) is a steroidal anti-inflammatory drug like betamethasone phosphate and triamcinolone, and is known to show a superior anti-inflammatory action by transdermal administration. In addition, an eye drop thereof is used for the prophylaxis or treatment of inflammation after surgery and for the prophylaxis or treatment of ocular pain after surgery. [0009] Macular edema is swelling of retinal macula, and the edema occurs due to a liquid leakage from the retinal blood vessels. Macular edema is observed as increased foveal retinal thickness of macula. While the foveal retinal thickness of healthy human varies depending on the individual differences and age, the average value thereof is reported to be.178 mm (Br J Ophthalmol Sep; 82(9): 03-6). For the treatment of macular edema, photocoagulation by laser irradiation, vitreous surgery, systemic administration of steroid, intravitreal administration and sub-tenon administration have conventionally been employed as mentioned above, and a certain level of therapeutic effect is found by these treatment methods in some macular edema. On the other hand, it is known that these conventional treatment methods cannot provide a sufficient effect for some macular edema. In the present invention, refractory macular edema refers to macular edema for which a sufficient effect cannot be afforded by the conventionally-employed treatments (e.g., any one or more of photocoagulation by irradiation, vitreous surgery, systemic administration of steroid, intravitreal administration and sub-tenon administration). [00] In the present invention, a sufficient treatment effect means that the foveal retinal thickness decreases by not less than %. In addition, improvement of logmar value by not less than 0.2 unit is also considered a sufficient effect. This is a general diagnostic criterium in ophthalmic treatments (FDA/NEI protocol (NEI/FDA Ophthalmic Clinical Trial Design and Endpoints Meeting, 06)). [0011] The logmar (the log of the minimum angle of resolution) value shows the ability of the eye (visual acuity) to distinguish the minimum separable acuity, which can be determined by logarithmically converting the values measured using a decimal visual acuity chart. [0012] The foveal retinal thickness is a value from the internal limiting membrane to the visual cell inner segment-outer segment junction in the fovea, and can be measured by Cirrus OCT (registered trade mark, Carl Zeiss), 3D OCT-00 (registered trade mark, TOPCON CORPORATION). 3
4 [0013] The eye drop containing difluprednate as an active ingredient relating to the present invention may take any form of emulsion or suspension. In view of the tissue transitivity of difluprednate, particularly preferred is an emulsion eye drop in the form of an emulsion. Examples of such emulsion eye drop include Durezol (registered trade mark, Sirion Therapeutics, USA). Durezol is a preparation obtained by emulsifying a mixture of difluprednate and predetermined subcomponents for the purpose of enabling difluprednate, the active ingredient, to appropriately penetrate into the eyeball and act on the affected part. When the eye drop of the present invention is an emulsion, it can be prepared according to, for example, US Patent No. 6,114,319 (JP-B-34364). When the eye drop of the present invention is a suspension, it can be prepared according to, for example, US Patent No.,6,848 (JP-B ). [0014] The concentration of difluprednate contained in the eye drop of the present invention is preferably % (w/v), more preferably % (w/v), particularly preferably 0.0% (w/v). [001] The eye drop of the present invention can be used after mixing with various known pharmaceutically acceptable substances appropriately selected with the aim of adjusting the tissue transitivity upon instillation. Examples of the pharmaceutically acceptable components include oils (e.g., castor oil, peanuts oil, cottonseed oil, soybean oil, olive oil, medium-chain triglyceride), solvents (e.g., saline, sterilization purified water), stabilizers (e.g., sodium edetate, citric acid), emulsifiers (e.g., polyvinylpyrrolidone), suspending agents (e.g., hydroxypropylmethylcellulose, methylcellulose, hydroxymethylcellulose), surfactants (e.g., polysorbate 80, polyoxyethylene hydrogenated castor oil), preservatives (e.g., benzalkonium chloride, parabens, chlorobutanol), buffers (e.g., boric acid, borax (sodium borate), sodium acetate, citrate buffer, phosphate buffer ), isotonicity agents (e.g., sodium chloride, glycerol, mannitol), and ph adjusters (e.g., hydrochloric acid, sodium hydroxide). These various known substances can be appropriately selected and used according to the object. Particularly, when the eye drop of the present invention is used as an emulsion, it desirably contains a surfactant as an emulsifier. As the surfactant, a nonionic surfactant and the like can be added. Examples of the nonionic surfactant include polyoxyethylene hydrogenated castor oils or polyoxyethylene sorbitan fatty acid ester, preferably sorbitan polyoxyethylene monooleates, polyoxyethylene sorbitan monolaurates, sorbitan polyoxyethylene monopalmitates, and sorbitan polyoxyethylene monostearates. Among these, castor oil and polysorbate 80 are preferably contained. Examples of other components that can be contained include concentrated glycerin, sodium acetate, boric acid, sodium edetate and sorbic acid. Particularly preferred are concentrated glycerin, sodium acetate, boric acid, sodium edetate and sorbic acid, which are preferably contained as necessary. [0016] The eye drop of the present invention can be safely administered to mammals (human, dog, rabbit, bovine, horse, monkey, cat, sheep etc.). [0017] While the dose of the eye drop of the present invention varies depending on the level of condition, age and body weight of patient, for example, an eye drop containing % (w/v) of difluprednate is preferably administered to an adult 2-4 times per day by instillation of 1-2 drops (preferably 1 drop: - 0 ml) per administration. In addition, while the dosing period of the eye drop of the present invention varies depending on the level of condition, for example, it is at least 1 week, preferably 1-4 weeks, more preferably not less than 4 weeks, more preferably not longer than 12 weeks. Depending on the level of condition, however, administration exceeding 12 weeks may be performed. [0018] In a preferable administration mode, for example, the dosing period of the eye drop of the present invention with a difluprednate concentration of % (w/v) is at least 1 week, the administration frequency is 4 times per day, and the dose is - 0 ml per administration. In another preferable administration mode, for example, the dosing period of the eye drop of the present invention with a difluprednate concentration of % (w/v) is 1-4 weeks, the administration frequency is 4 times per day, and the dose is - 0 ml per administration. In a still another preferable administration mode, the administration frequency of the eye drop of the present invention with a difluprednate concentration of % (w/v) is 4 times per day for 4 weeks from the start of the administration, twice per day after 4 weeks, and the dose is - 0 ml per administration. In a yet another preferable administration mode, the dosing period of the eye drop of the present invention with a difluprednate concentration of % (w/v) is not longer than 12 weeks, the administration frequency is 4 times per day for 4 weeks from the start of the administration, twice per day after 4 weeks, and the dose is - 0 ml per administration. In other preferable administration mode, the dosing period of the eye drop of the present invention with a difluprednate concentration of % (w/v) is at least 12 weeks, the administration frequency is 4 times per day for 4 weeks from the start of the administration, twice per day after 4 weeks, and the dose is - 0 ml per administration. 4
5 EXAMPLES (Example 1) 1 [0019] The following results were obtained by the clinical tests (UMIN ) approved by the Ethics Committee of the Yamagata University Faculty of Medicine. For confirmation of the efficacy of the eye drop of the present invention, the eyes of 11 patients with macular edema (16 eyes) were treated by instillation of 0.0% (w/v) difluprednate emulsion eye drop (Durezol). The patient group instilled with Durezol included patients who received treatment by sub-tenon administration of triamcinolone (n=8), intravitreal administration of triamcinolone (n=4), vitreous surgery (n=) or intravitreal administration of bevacizumab (n=2) (with therapeutic duplication). In addition, photocoagulation was applied to 12 eyes, and an intraocular lens was implanted in 11 eyes. Thus, the patients subjected to the treatment were those who had underwent conventionally-known treatments of macular edema, and were in various conditions particularly due to vitreous surgery and intraocular lens implant. They were patients diagnosed with refractory macular edema who resist improvement of retinal edema near fovea even by these treatments and still have a greater foveal retinal thickness than healthy human. The clinical test was performed by instillation of Durezol to all patients after lapse of not less than 3 months from the previous treatment. The instillation of Durezol was performed 4 times per day for the first 1 month, twice per day for 2 months thereafter. In addition, the dose per administration was 1 drop (about - 0 ml). The formulation of Durezol is as follows. 2 difluprednate castor oil polysorbate 80 concentrated glycerin sodium acetate boric acid sodium edetate sorbic acid sterile purified water 0.0 g.0 g 4.0 g 2.2 g 0.0 g 0.1 g 0.02 g 0.1 g total amount 0 ml [00] As a control group, 17 eyes of 9 patients from among patients with the onset of diabetic macular edema were adopted, who were comparable to the Durezol administration group in the age, sex, duration of diabetes and the level of retinopathy. The control group consists of patients before undergoing treatments of macular edema, such as sub-tenon administration of steroid, intravitreal administration of steroid, vitreous surgery and the like. This control group was administered with a 0.1% (w/v) ophthalmic solution of betamethasone sodium phosphate (Rinderon A, registered trade mark) 6 times per day for 1 month. The betamethasone phosphate eye drop is a steroid preparation generally used as an anti-inflammatory agent for anterior ocular segment in the same manner as an eye drop containing difluprednate. The above-mentioned administration conditions of Rinderon A are those for the administration of Rinderon A in a test (steroid responder test) previously confirming the level of intraocular pressure increase caused by sub-tenon administration or intravitreal administration of steroid such as triamcinolone and the like for the treatment of macular edema. The steroid responder test aims to test the sensitivity of individual patient to steroid by measuring the level of intraocular pressure increase resulting from the action, inside the eyeball, of betamethasone phosphate administered by instillation, and it clarifies whether or not macular edema can be treated by intravitreal administration and the like of steroid. [0021] The patients in the Examples of the present invention and the patients in the control group were subjected to the visual acuity test and measurement of decrease rate of the foveal retina thickness using Cirrus OCT (Carl Zeiss), according to the FDA/NEI protocol (NEI/FDA Ophthalmic Clinical Trial Design and Endpoints Meeting, 06). For the judgment of effectiveness of the instillation treatment, improvement of not less than 0.2 unit in the logmar value by instillation was judged to be effective for improvement of visual acuity, or not less than % of decrease rate of the foveal retinal thickness was judged to be effective. In addition, the effectiveness rate was calculated as a proportion (%) of the number of cases effective for the improvement of visual acuity and foveal retinal thickness decrease rate to the total number of cases. [0022] Table 1 shows the effects of the Durezol instillation and Rinderon A instillation on the foveal retinal thickness decrease rate in macular edema. The effectiveness rate of the foveal retinal thickness decrease is 37.% in the Durezol administration group at the time point of 1 month from the start of the administration, which clearly shows that the symptom of macular edema is improved. In contrast, in the Rinderon A administration group (control group) (Rinderon A is used as an anti-inflammatory agent for the anterior ocular segment like Durezol), a decrease in the foveal retinal thickness was found in a small portion of patients; however, the effectiveness rate to the total number of patients was
6 1 2 extremely low as compared to the Durezol administration group. Considering that the patients of the Durezol administration group are difficult to treat for edema as compared to the patients of the Rinderon A administration group, it is clear that the eye drop of difluprednate of the present invention shows efficacy not obtainable by a betamethasone phosphate eye drop, which is a similar steroid preparation. In addition, it is clear that the difluprednate eye drop is also effective for refractory macular edema, which is difficult to treat by conventional treatment methods. Table 2 shows time-course changes in the effect of Durezol instillation on the foveal retinal thickness decrease rate. Continuous instillation of Durezol to macular edema patients provides continuous increase in the effectiveness rate, continuous decrease in the average retina thickness of the patients, and administration for 3 months can afford an extremely high effectiveness rate of 61.%. While the patients of the Durezol administration group had experience of various conventionally-known treatments before the Durezol instillation treatment, similar effectiveness was achieved by the Durezol administration regardless of the treatment history such as vitreous surgery and the like. This indicates that the instilled difluprednate preparation directly acts on the edema site of the macula part. The effect of the eye drop of the present invention is not limited for refractory macular edema, and a similar edema-improving effect is also expected in general patients with macular edema, whose conditions can be improved more easily. Table 3 shows the effect of Durezol for improvement of visual acuity in macular edema. Table 4 shows time course changes of the effectiveness rate by the Durezol administration in the improvement of visual acuity. Improvement of visual acuity by the treatment of macular edema requires a comparatively long time after improvement of edema in the macula part, and therefore, it is generally difficult to confirm improvement of visual acuity during a short-term treatment. However, apparent improvement of visual acuity was found in a short time in a part of the Durezol administration group, thus showing the effectiveness of the difluprednate eye drop as a method for treating macular edema. On the other hand, an effectiveness rate relative to the improvement of visual acuity was not confirmed in the control Rinderon A administration group. Rinderon A is a steroid used by instillation as an anti-inflammatory agent for the anterior ocular segment, as well as for a steroid responder test in patients with macular edema, as mentioned above, and the results are consistent with the absence of improvement of visual acuity in the steroid responder test. [0023] [Table 1] effectiveness rate after 1 month from start of administration Rinderon A.9% Durezol 37.% 3 [0024] [Table 2] after administration effectiveness rate average value (mm) of foveal retinal thickness 1 week 37.% month 37.% months 3.3% months 61.% [002] 0 [Table 3] effectiveness rate after 1 month from start of administration Rinderon A 0.0% Durezol 31% [0026] 6
7 [Table 4] after administration effectiveness rate 1 week 13% 1 month 31% 2 months 13% 3 months 31% INDUSTRIAL APPLICABILITY 1 [0027] An eye drop containing difluprednate of the present invention as an active ingredient has enabled improvement of edema in macular edema patients by instillation, which is an administration method with low tissue invasiveness as compared to conventional methods employed in clinical practice. The effects on foveal retinal thickness and visual acuity are superior to those of a betamethasone phosphate eye drop, for which experimental example of instillation administration has been reported. Moreover, effective results can also be obtained in patients with recurrent refractory macular edema, who received sub-tenon administration or intravitreal administration of triamcinolone, vitreous surgery or intravitreal administration of bevacizumab in the past. Claims 1. An eye drop containing difluprednate for use in the treatment of macular edema The eye drop for use in the treatment according to claim 1, wherein the macular edema is refractory macular edema. 3. The eye drop for use in the treatment according to claim 1 or 2, which is an emulsion eye drop. 4. The eye drop for use in the treatment according to any one of claims 1 to 3, wherein the concentration of difluprednate is % (w/v). 3. The eye drop for use in the treatment according to any one of claims 1 to 4, comprising % (w/v) of difluprednate, and castor oil, polysorbate 80, concentrated glycerin, sodium acetate, boric acid, sodium edetate and sorbic acid. 6. The eye drop for use in the treatment according to any one of claims 1 to, wherein the concentration of difluprednate is % (w/v), and the eye drop is administered for at least 1 week, 4 times per day, in a dose of - 0 ml per administration The eye drop for use in the treatment according to any one of claims 1 to, wherein the concentration of difluprednate is % (w/v), and the eye drop is administered for 1-4 weeks, 4 times per day, in a dose of - 0 ml per administration. 8. The eye drop for use in the treatment according to any one of claims 1 to, wherein the concentration of difluprednate is % (w/v), and the eye drop is administered 4 times per day for 4 weeks from the start of the administration and twice per day after 4 weeks, in a dose of - 0 ml per administration The eye drop for use in the treatment according to any one of claims 1 to, wherein the concentration of difluprednate is % (w/v), and the eye drop is administered for not longer than 12 weeks, 4 times per day for 4 weeks from the start of the administration and twice per day after 4 weeks, in a dose of - 0 ml per administration.. The eye drop for use in the treatment according to any one of claims 1 to, wherein the concentration of difluprednate is % (w/v), and the eye drop is administered for at least 12 weeks, 4 times per day for 4 weeks from the start of the administration and twice per day after 4 weeks, in a dose of - 0 ml per administration. 7
8 Patentansprüche 1. Augentropfen, die Difluprednat enthalten, zur Verwendung bei der Behandlung von Makulaödem. 2. Augentropfen zur Verwendung bei der Behandlung gemäß Anspruch 1, wobei das Makulaödem ein therapierefraktäres Makulaödem ist. 3. Augentropfen zur Verwendung bei der Behandlung gemäß Anspruch 1 oder 2, die als Emulsion vorliegen. 4. Augentropfen zur Verwendung bei der Behandlung gemäß einem der Ansprüche 1 bis 3, wobei die Konzentration von Difluprednat 0,00 bis 0,1% (w/v) beträgt. 1. Augentropfen zur Verwendung bei der Behandlung gemäß einem der Ansprüche 1 bis 4, umfassend 0,00 bis 0,1% (w/v) Difluprednat sowie Ricinusöl, Polysorbat 80, konzentriertes Glycerin, Natriumacetat, Borsäure, Natriumedetat und Sorbinsäure Augentropfen zur Verwendung bei der Behandlung gemäß einem der Ansprüche 1 bis, wobei die Konzentration von Difluprednat 0,00 bis 0,1% (w/v) beträgt und die Augentropfen wenigstens 1 Woche lang viermal am Tag in einer Dosis von bis 0 ml pro Verabreichung verabreicht werden. 7. Augentropfen zur Verwendung bei der Behandlung gemäß einem der Ansprüche 1 bis, wobei die Konzentration von Difluprednat 0,00 bis 0,1% (w/v) beträgt und die Augentropfen 1 bis 4 Wochen lang viermal am Tag in einer Dosis von bis 0 ml pro Verabreichung verabreicht werden. 8. Augentropfen zur Verwendung bei der Behandlung gemäß einem der Ansprüche 1 bis, wobei die Konzentration von Difluprednat 0,00 bis 0,1% (w/v) beträgt und die Augentropfen ab Beginn der Verabreichung 4 Wochen lang viermal am Tag und nach 4 Wochen zweimal am Tag in einer Dosis von bis 0 ml pro Verabreichung verabreicht werden. 9. Augentropfen zur Verwendung bei der Behandlung gemäß einem der Ansprüche 1 bis, wobei die Konzentration von Difluprednat 0,00 bis 0,1% (w/v) beträgt und die Augentropfen nicht länger als 12 Wochen verabreicht werden, und zwar ab Beginn der Verabreichung 4 Wochen lang viermal am Tag und nach 4 Wochen zweimal am Tag in einer Dosis von bis 0 ml pro Verabreichung.. Augentropfen zur Verwendung bei der Behandlung gemäß einem der Ansprüche 1 bis, wobei die Konzentration von Difluprednat 0,00 bis 0,1% (w/v) beträgt und die Augentropfen wenigstens 12 Wochen lang verabreicht werden, und zwar ab Beginn der Verabreichung 4 Wochen lang viermal am Tag und nach 4 Wochen zweimal am Tag in einer Dosis von bis 0 ml pro Verabreichung. Revendications 1. Goutte oculaire contenant du difluprednate pour son utilisation dans le traitement de l oedème maculaire Goutte oculaire pour son utilisation dans le traitement selon la revendication 1, dans laquelle l oedème maculaire est un oedème maculaire réfractaire Goutte oculaire pour son utilisation dans le traitement selon la revendication 1 ou 2, qui est une goutte oculaire par émulsion. 4. Goutte oculaire pour son utilisation dans le traitement selon l une quelconque des revendications 1 à 3, dans laquelle la concentration de difluprednate est de 0,00 à 0,1 % (p/v).. Goutte oculaire pour son utilisation dans le traitement selon l une quelconque des revendications 1 à 4, comprenant 0,00 à 0,1 % (p/v) de difluprednate, et de l huile de ricin, du polysorbate 80, de la glycérine concentrée, de l acétate de sodium, de l acide borique, de l édétate de sodium et de l acide sorbique. 6. Goutte oculaire pour son utilisation dans le traitement selon l une quelconque des revendications 1 à, dans laquelle 8
9 la concentration de difluprednate est de 0,00 à 0,1 % (p/v) et la goutte oculaire est administrée pendant au moins 1 semaine, 4 fois par jour, dans une dose de à 0 ml par administration. 7. Goutte oculaire pour son utilisation dans le traitement selon l une quelconque des revendications 1 à, dans laquelle la concentration de difluprednate est de 0,00 à 0,1 % (p/v) et la goutte oculaire est administrée pendant 1 à 4 semaines, 4 fois par jour, dans une dose de à 0 ml par administration. 8. Goutte oculaire pour son utilisation dans le traitement selon l une quelconque des revendications 1 à, dans laquelle la concentration de difluprednate est de 0,00 à 0,1 % (p/v) et la goutte oculaire est administrée 4 fois par jour pendant 4 semaines à compter du début de l administration et deux fois par jour après 4 semaines, dans une dose de à 0 ml par administration Goutte oculaire pour son utilisation dans le traitement selon l une quelconque des revendications 1 à, dans laquelle la concentration de difluprednate est de 0,00 à 0,1 % (p/v) et la goutte oculaire est administrée pendant au plus 12 semaines, 4 fois par jour pendant 4 semaines à compter du début de l administration et deux fois par jour après 4 semaines, dans une dose de à 0 ml par administration.. Goutte oculaire pour son utilisation dans le traitement selon l une quelconque des revendications 1 à, dans laquelle la concentration de difluprednate est de 0,00 à 0,1 % (p/v) et la goutte oculaire est administrée pendant au moins 12 semaines, 4 fois par jour pendant 4 semaines à compter du début de l administration et deux fois par jour après 4 semaines, dans une dose de à 0 ml par administration
10 REFERENCES CITED IN THE DESCRIPTION This list of references cited by the applicant is for the reader s convenience only. It does not form part of the European patent document. Even though great care has been taken in compiling the references, errors or omissions cannot be excluded and the EPO disclaims all liability in this regard. Patent documents cited in the description WO A [0002] US A [0013] JP B [0013] US 6848 A [0013] JP B [0013] Non-patent literature cited in the description The 43rd Annual Congress of Japanese Retina and Vitreous Society 016-, 04 [0002] Br J Ophthalmol., September 1998, vol. 82 (9), 03-6 [0009] NEI/FDA Ophthalmic Clinical Trial Design and Endpoints Meeting, 06 [00]
TEPZZ _7584 A_T EP A1 (19) (11) EP A1 (12) EUROPEAN PATENT APPLICATION. (51) Int Cl.: A61K 9/00 ( ) A61K 31/439 (2006.
 (19) TEPZZ _784 A_T (11) EP 3 17 842 A1 (12) EUROPEAN PATENT APPLICATION (43) Date of publication: 07.06.17 Bulletin 17/23 (1) Int Cl.: A61K 9/00 (06.01) A61K 31/439 (06.01) (21) Application number: 1197874.9
(19) TEPZZ _784 A_T (11) EP 3 17 842 A1 (12) EUROPEAN PATENT APPLICATION (43) Date of publication: 07.06.17 Bulletin 17/23 (1) Int Cl.: A61K 9/00 (06.01) A61K 31/439 (06.01) (21) Application number: 1197874.9
(51) Int Cl.: A61K 9/00 ( ) A61K 31/198 ( ) A61K 47/26 ( )
 (19) TEPZZ 8Z6B_T (11) EP 2 8 06 B1 (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent: 08.06.16 Bulletin 16/23 (21) Application number: 1171684.2 (22) Date
(19) TEPZZ 8Z6B_T (11) EP 2 8 06 B1 (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent: 08.06.16 Bulletin 16/23 (21) Application number: 1171684.2 (22) Date
EP A2 (19) (11) EP A2 (12) EUROPEAN PATENT APPLICATION. (43) Date of publication: Bulletin 2011/42
 (19) (12) EUROPEAN PATENT APPLICATION (11) EP 2 377 14 A2 (43) Date of publication: 19..11 Bulletin 11/42 (21) Application number: 10329.0 (1) Int Cl.: A61K 9/00 (06.01) A61K 31/13 (06.01) A61K 31/167
(19) (12) EUROPEAN PATENT APPLICATION (11) EP 2 377 14 A2 (43) Date of publication: 19..11 Bulletin 11/42 (21) Application number: 10329.0 (1) Int Cl.: A61K 9/00 (06.01) A61K 31/13 (06.01) A61K 31/167
TEPZZ 9Z4Z79B_T EP B1 (19) (11) EP B1 (12) EUROPEAN PATENT SPECIFICATION
 (19) TEPZZ 9Z4Z79B_T (11) EP 2 904 079 B1 (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent: 29.03.17 Bulletin 17/13 (21) Application number: 1377374.0 (22)
(19) TEPZZ 9Z4Z79B_T (11) EP 2 904 079 B1 (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent: 29.03.17 Bulletin 17/13 (21) Application number: 1377374.0 (22)
TEPZZ 85_Z 4A_T EP A1 (19) (11) EP A1 (12) EUROPEAN PATENT APPLICATION
 (19) TEPZZ 8_Z 4A_T (11) EP 2 81 034 A1 (12) EUROPEAN PATENT APPLICATION (43) Date of publication: 2.03.1 Bulletin 1/13 (21) Application number: 1022.2 (1) Int Cl.: A61C /02 (06.01) A61C 19/04 (06.01)
(19) TEPZZ 8_Z 4A_T (11) EP 2 81 034 A1 (12) EUROPEAN PATENT APPLICATION (43) Date of publication: 2.03.1 Bulletin 1/13 (21) Application number: 1022.2 (1) Int Cl.: A61C /02 (06.01) A61C 19/04 (06.01)
(51) Int Cl.: A61K 31/77 ( ) A61P 1/06 ( ) A61P 1/10 ( )
 (19) (11) EP 1 326 92 B1 (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent: 23.07.08 Bulletin 08/ (21) Application number: 0197466.2 (22) Date of filing:
(19) (11) EP 1 326 92 B1 (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent: 23.07.08 Bulletin 08/ (21) Application number: 0197466.2 (22) Date of filing:
*EP A1* EP A1 (19) (11) EP A1. (12) EUROPEAN PATENT APPLICATION published in accordance with Art.
 (19) Europäisches Patentamt European Patent Office Office européen des brevets *EP00147089A1* (11) EP 1 47 089 A1 (12) EUROPEAN PATENT APPLICATION published in accordance with Art. 18(3) EPC (43) Date
(19) Europäisches Patentamt European Patent Office Office européen des brevets *EP00147089A1* (11) EP 1 47 089 A1 (12) EUROPEAN PATENT APPLICATION published in accordance with Art. 18(3) EPC (43) Date
(51) Int Cl.: A61K 9/20 ( ) A61K 31/41 ( )
 (19) TEPZZ 94677_A_T (11) (12) EUROPEAN PATENT APPLICATION (43) Date of publication: 2.11.1 Bulletin 1/48 (1) Int Cl.: A61K 9/ (06.01) A61K 31/41 (06.01) (21) Application number: 116790.3 (22) Date of
(19) TEPZZ 94677_A_T (11) (12) EUROPEAN PATENT APPLICATION (43) Date of publication: 2.11.1 Bulletin 1/48 (1) Int Cl.: A61K 9/ (06.01) A61K 31/41 (06.01) (21) Application number: 116790.3 (22) Date of
TEPZZ _ 849 A_T EP A1 (19) (11) EP A1. (12) EUROPEAN PATENT APPLICATION published in accordance with Art.
 (19) TEPZZ _ 849 A_T (11) EP 3 138 493 A1 (12) EUROPEAN PATENT APPLICATION published in accordance with Art. 3(4) EPC (43) Date of publication: 08.03.17 Bulletin 17/ (21) Application number: 786271. (22)
(19) TEPZZ _ 849 A_T (11) EP 3 138 493 A1 (12) EUROPEAN PATENT APPLICATION published in accordance with Art. 3(4) EPC (43) Date of publication: 08.03.17 Bulletin 17/ (21) Application number: 786271. (22)
(51) Int Cl.: A61F 9/007 ( ) A61B 17/32 ( )
 (19) (11) EP 1 733 707 B1 (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent: 03.09.2008 Bulletin 2008/36 (1) Int Cl.: A61F 9/007 (2006.01) A61B 17/32 (2006.01)
(19) (11) EP 1 733 707 B1 (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent: 03.09.2008 Bulletin 2008/36 (1) Int Cl.: A61F 9/007 (2006.01) A61B 17/32 (2006.01)
EP A2 (19) (11) EP A2 (12) EUROPEAN PATENT APPLICATION. (43) Date of publication: Bulletin 2009/16
 (19) (12) EUROPEAN PATENT APPLICATION (11) EP 2 047 749 A2 (43) Date of publication: 1.04.09 Bulletin 09/16 (21) Application number: 0838028.0 (22) Date of filing: 07..08 (84) Designated Contracting States:
(19) (12) EUROPEAN PATENT APPLICATION (11) EP 2 047 749 A2 (43) Date of publication: 1.04.09 Bulletin 09/16 (21) Application number: 0838028.0 (22) Date of filing: 07..08 (84) Designated Contracting States:
EFFICACY OF INTRAVITREAL TRIAMCINOLONE ACETONIDE FOR THE TREATMENT OF DIABETIC MACULAR EDEMA
 Basrah Journal Of Surgery EFFICACY OF INTRAVITREAL TRIAMCINOLONE ACETONIDE FOR THE TREATMENT OF DIABETIC MACULAR EDEMA Salah Zuhair Abed Al-Asadi MB,ChB, FICMS, Lecturer, Department of Surgery, College
Basrah Journal Of Surgery EFFICACY OF INTRAVITREAL TRIAMCINOLONE ACETONIDE FOR THE TREATMENT OF DIABETIC MACULAR EDEMA Salah Zuhair Abed Al-Asadi MB,ChB, FICMS, Lecturer, Department of Surgery, College
nepafenac 1mg/mL eye drops, suspension (Nevanac ) SMC No. (813/12) Alcon Laboratories (UK) Ltd
 nepafenac 1mg/mL eye drops, suspension (Nevanac ) SMC No. (813/12) Alcon Laboratories (UK) Ltd 05 October 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
nepafenac 1mg/mL eye drops, suspension (Nevanac ) SMC No. (813/12) Alcon Laboratories (UK) Ltd 05 October 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product
NEPTUNE RED BANK BRICK
 NEPTUNE RED BANK BRICK Diabetes & The Eye Diabetics are more likely to develop Cataracts at a younger age. Diabetics are twice as likely to develop Glaucoma when compared to non-diabetics. The primary
NEPTUNE RED BANK BRICK Diabetes & The Eye Diabetics are more likely to develop Cataracts at a younger age. Diabetics are twice as likely to develop Glaucoma when compared to non-diabetics. The primary
EP A1 (19) (11) EP A1 (12) EUROPEAN PATENT APPLICATION. (43) Date of publication: Bulletin 2012/28
 (19) (12) EUROPEAN PATENT APPLICATION (11) EP 2 474 288 A1 (43) Date of publication: 11.07.2012 Bulletin 2012/28 (51) Int Cl.: A61F 2/40 (2006.01) A61F 2/30 (2006.01) (21) Application number: 11150615.0
(19) (12) EUROPEAN PATENT APPLICATION (11) EP 2 474 288 A1 (43) Date of publication: 11.07.2012 Bulletin 2012/28 (51) Int Cl.: A61F 2/40 (2006.01) A61F 2/30 (2006.01) (21) Application number: 11150615.0
TEPZZ Z568 B_T EP B1 (19) (11) EP B1 (12) EUROPEAN PATENT SPECIFICATION
 (19) TEPZZ Z68 B_T (11) (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent: 22.03.17 Bulletin 17/12 (21) Application number: 07794121.9 (22) Date of filing:.08.07
(19) TEPZZ Z68 B_T (11) (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent: 22.03.17 Bulletin 17/12 (21) Application number: 07794121.9 (22) Date of filing:.08.07
(51) Int Cl.: A23L 33/19 ( ) A61K 35/20 ( ) A23L 33/10 ( ) A61P 3/10 ( )
 (19) TEPZZ 768 B_T (11) EP 2 768 323 B1 (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent: 14.06.2017 Bulletin 2017/24 (21) Application number: 12778093.0
(19) TEPZZ 768 B_T (11) EP 2 768 323 B1 (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent: 14.06.2017 Bulletin 2017/24 (21) Application number: 12778093.0
(51) Int Cl.: C12N 1/20 ( ) A61K 35/74 ( ) A61K 39/04 ( ) A61P 35/00 ( ) A61K 39/00 ( )
 (19) TEPZZ 66 _8B_T (11) EP 2 663 318 B1 (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent: 18.03.1 Bulletin 1/12 (21) Application number: 12702318.2 (22)
(19) TEPZZ 66 _8B_T (11) EP 2 663 318 B1 (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent: 18.03.1 Bulletin 1/12 (21) Application number: 12702318.2 (22)
Clinically Significant Macular Edema (CSME)
 Clinically Significant Macular Edema (CSME) 1 Clinically Significant Macular Edema (CSME) Sadrina T. Shaw OMT I Student July 26, 2014 Advisor: Dr. Uwaydat Clinically Significant Macular Edema (CSME) 2
Clinically Significant Macular Edema (CSME) 1 Clinically Significant Macular Edema (CSME) Sadrina T. Shaw OMT I Student July 26, 2014 Advisor: Dr. Uwaydat Clinically Significant Macular Edema (CSME) 2
EP A1 (19) (11) EP A1. (12) EUROPEAN PATENT APPLICATION published in accordance with Art. 153(4) EPC
 (19) (12) EUROPEAN PATENT APPLICATION published in accordance with Art. 13(4) EPC (11) EP 2 07 001 A1 (43) Date of publication: 01.07.2009 Bulletin 2009/27 (21) Application number: 07834499.1 (22) Date
(19) (12) EUROPEAN PATENT APPLICATION published in accordance with Art. 13(4) EPC (11) EP 2 07 001 A1 (43) Date of publication: 01.07.2009 Bulletin 2009/27 (21) Application number: 07834499.1 (22) Date
EP B2 (19) (11) EP B2. (12) NEW EUROPEAN PATENT SPECIFICATION After opposition procedure
 (19) (12) NEW EUROPEAN PATENT SPECIFICATION After opposition procedure (11) EP 0 92 789 B2 (4) Date of publication and mention of the opposition decision: 17.06.09 Bulletin 09/2 (1) Int Cl.: A61K 31/702
(19) (12) NEW EUROPEAN PATENT SPECIFICATION After opposition procedure (11) EP 0 92 789 B2 (4) Date of publication and mention of the opposition decision: 17.06.09 Bulletin 09/2 (1) Int Cl.: A61K 31/702
(51) Int Cl.: A23L 2/66 ( )
 (19) (12) NEW EUROPEAN PATENT SPECIFICATION (11) EP 0 967 892 B2 (4) Date of publication and mention of the opposition decision:..07 Bulletin 07/41 (4) Mention of the grant of the patent: 29.12.04 Bulletin
(19) (12) NEW EUROPEAN PATENT SPECIFICATION (11) EP 0 967 892 B2 (4) Date of publication and mention of the opposition decision:..07 Bulletin 07/41 (4) Mention of the grant of the patent: 29.12.04 Bulletin
ROLE OF LASER PHOTOCOAGULATION VERSUS INTRAVITREAL TRIAMCINOLONE ACETONIDE IN ANGIOGRAPHIC MACULAR EDEMA IN DIABETES MELLITUS
 ORIGINAL ARTICLE ROLE OF LASER PHOTOCOAGULATION VERSUS INTRAVITREAL TRIAMCINOLONE ACETONIDE IN ANGIOGRAPHIC MACULAR EDEMA IN DIABETES MELLITUS Aggarwal Somesh VP 1, Shah Sonali N 2, Bharwada Rekha M 3,
ORIGINAL ARTICLE ROLE OF LASER PHOTOCOAGULATION VERSUS INTRAVITREAL TRIAMCINOLONE ACETONIDE IN ANGIOGRAPHIC MACULAR EDEMA IN DIABETES MELLITUS Aggarwal Somesh VP 1, Shah Sonali N 2, Bharwada Rekha M 3,
Vitrectomy for Diabetic Cystoid Macular Edema
 Vitrectomy for Diabetic Cystoid Macular Edema Yukihiro Sato, Zeon Lee and Hiroyuki Shimada Department of Ophthalmology, Nihon University School of Medicine, Tokyo, Japan Purpose: We evaluated visual outcomes
Vitrectomy for Diabetic Cystoid Macular Edema Yukihiro Sato, Zeon Lee and Hiroyuki Shimada Department of Ophthalmology, Nihon University School of Medicine, Tokyo, Japan Purpose: We evaluated visual outcomes
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE. Health Technology Appraisal. Aflibercept for treating diabetic macular oedema.
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Health Technology Appraisal Aflibercept for treating diabetic macular oedema Final scope Final remit/appraisal objective To appraise the clinical and cost
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Health Technology Appraisal Aflibercept for treating diabetic macular oedema Final scope Final remit/appraisal objective To appraise the clinical and cost
(51) Int Cl.: A61K 33/24 ( ) A61P 9/00 ( ) A61P 9/10 ( ) A61P 41/00 ( ) A61P 43/00 ( )
 (19) TEPZZ 68 4B_T (11) EP 2 6 834 B1 (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent: 26.11.2014 Bulletin 2014/48 (21) Application number: 12189630.2
(19) TEPZZ 68 4B_T (11) EP 2 6 834 B1 (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent: 26.11.2014 Bulletin 2014/48 (21) Application number: 12189630.2
Efficacy of intravitreal bevacizumab (Avastin TM ) for shortterm treatment of diabetic macular edema
 111 ORIGINAL Efficacy of intravitreal bevacizumab (Avastin TM ) for shortterm treatment of diabetic macular edema Toshihiko Nagasawa, Takeshi Naito, Shingo Matsushita, Hiroyuki Sato, Takashi Katome, and
111 ORIGINAL Efficacy of intravitreal bevacizumab (Avastin TM ) for shortterm treatment of diabetic macular edema Toshihiko Nagasawa, Takeshi Naito, Shingo Matsushita, Hiroyuki Sato, Takashi Katome, and
(43) Publication date: 04 September 2014 ( ) (22) Filing Date: 27 February 2014 ( )
 (54) Title (EN): INCOHERENT TYPE-III MATERIALS FOR CHARGE CARRIERS CONTROL DEVICES (54) Title (FR): MATÉRIAUX DE TYPE III INCOHÉRENTS POUR DISPOSITIFS DE RÉGULATION DE PORTEURS DE CHARGES (72) Inventor(s):
(54) Title (EN): INCOHERENT TYPE-III MATERIALS FOR CHARGE CARRIERS CONTROL DEVICES (54) Title (FR): MATÉRIAUX DE TYPE III INCOHÉRENTS POUR DISPOSITIFS DE RÉGULATION DE PORTEURS DE CHARGES (72) Inventor(s):
Facts About Diabetic Eye Disease
 Facts About Diabetic Eye Disease Points to Remember 1. Diabetic eye disease comprises a group of eye conditions that affect people with diabetes. These conditions include diabetic retinopathy, diabetic
Facts About Diabetic Eye Disease Points to Remember 1. Diabetic eye disease comprises a group of eye conditions that affect people with diabetes. These conditions include diabetic retinopathy, diabetic
Information for Patients undergoing Intravitreal Triamcinolone Acetonide (Kenalog) Injection
 Information for Patients undergoing Intravitreal Triamcinolone Acetonide (Kenalog) Injection Kenalog/SS/ST/04.2012/v1.1 review 05.2013 Page 1 Introduction Your doctor has found that you have leakage of
Information for Patients undergoing Intravitreal Triamcinolone Acetonide (Kenalog) Injection Kenalog/SS/ST/04.2012/v1.1 review 05.2013 Page 1 Introduction Your doctor has found that you have leakage of
TEPZZ Z697_5A_T EP A1 (19) (11) EP A1 (12) EUROPEAN PATENT APPLICATION. (51) Int Cl.: A61K 9/20 ( ) A61K 31/135 (2006.
 (19) TEPZZ Z697_A_T (11) EP 3 069 7 A1 (12) EUROPEAN PATENT APPLICATION (43) Date of publication: 21.09.16 Bulletin 16/38 (1) Int Cl.: A61K 9/ (06.01) A61K 31/13 (06.01) (21) Application number: 160172.1
(19) TEPZZ Z697_A_T (11) EP 3 069 7 A1 (12) EUROPEAN PATENT APPLICATION (43) Date of publication: 21.09.16 Bulletin 16/38 (1) Int Cl.: A61K 9/ (06.01) A61K 31/13 (06.01) (21) Application number: 160172.1
A Patient s Guide to Diabetic Retinopathy
 Diabetic Retinopathy A Patient s Guide to Diabetic Retinopathy 840 Walnut Street, Philadelphia PA 19107 www.willseye.org Diabetic Retinopathy 1. Definition Diabetic retinopathy is a complication of diabetes
Diabetic Retinopathy A Patient s Guide to Diabetic Retinopathy 840 Walnut Street, Philadelphia PA 19107 www.willseye.org Diabetic Retinopathy 1. Definition Diabetic retinopathy is a complication of diabetes
Assisting in Ophthalmology. Copyright 2011, 2007, 2003, 1999 by Saunders, an imprint of Elsevier Inc. All rights reserved.
 Assisting in Ophthalmology Learning Objectives Define, spell, and pronounce the terms listed in the vocabulary. Apply critical thinking skills in performing patient assessment and care. Explain the differences
Assisting in Ophthalmology Learning Objectives Define, spell, and pronounce the terms listed in the vocabulary. Apply critical thinking skills in performing patient assessment and care. Explain the differences
Ophthalmology. Ophthalmology Services
 Ophthalmology Ophthalmology Services The Ophthalmology service offers the latest and most comprehensive eye care for patients. With a dedicated team of eye surgeons and consultants, we treat vision problems
Ophthalmology Ophthalmology Services The Ophthalmology service offers the latest and most comprehensive eye care for patients. With a dedicated team of eye surgeons and consultants, we treat vision problems
COMPARISON OF INTRAVITREAL TRIAMCINOLONE INJECTION VS LASER PHOTOCOAGULATION IN ANGIOGRAPHIC MACULAR EDEMA IN DIABETIC RETINOPATHY
 Original Article COMPARISON OF INTRAVITREAL TRIAMCINOLONE INJECTION VS LASER PHOTOCOAGULATION IN ANGIOGRAPHIC MACULAR EDEMA IN DIABETIC RETINOPATHY Aggarwal Somesh V 1, Shah Sonali N 2, Bharwada Rekha
Original Article COMPARISON OF INTRAVITREAL TRIAMCINOLONE INJECTION VS LASER PHOTOCOAGULATION IN ANGIOGRAPHIC MACULAR EDEMA IN DIABETIC RETINOPATHY Aggarwal Somesh V 1, Shah Sonali N 2, Bharwada Rekha
TEPZZ 857Z_4B_T EP B1 (19) (11) EP B1 (12) EUROPEAN PATENT SPECIFICATION
 (19) TEPZZ 87Z_4B_T (11) EP 2 87 014 B1 (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent: 22.06.16 Bulletin 16/2 (21) Application number: 14199791.6 (1)
(19) TEPZZ 87Z_4B_T (11) EP 2 87 014 B1 (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent: 22.06.16 Bulletin 16/2 (21) Application number: 14199791.6 (1)
Evaluation of Changes of Macular Thickness in Diabetic Retinopathy after Cataract Surgery
 pissn: 1011-8942 eissn: 2092-9382 Korean J Ophthalmol 2011;25(4):238-242 DOI: 10.3341/kjo.2011.25.4.238 Evaluation of Changes of Macular Thickness in Diabetic Retinopathy after Cataract Surgery Original
pissn: 1011-8942 eissn: 2092-9382 Korean J Ophthalmol 2011;25(4):238-242 DOI: 10.3341/kjo.2011.25.4.238 Evaluation of Changes of Macular Thickness in Diabetic Retinopathy after Cataract Surgery Original
A retrospective nonrandomized study was conducted at 3
 Department of Ophthalmology, Kangbuk Samsung Hospital, Sungkyunkwan University College of Medicine 1, Seoul, Korea Hangil Eye Hospital 2, Incheon, Korea Seoul National University Bundang Hospital 3, Seongnam,
Department of Ophthalmology, Kangbuk Samsung Hospital, Sungkyunkwan University College of Medicine 1, Seoul, Korea Hangil Eye Hospital 2, Incheon, Korea Seoul National University Bundang Hospital 3, Seongnam,
Intravitreal versus Posterior Subtenon Injection of Triamcinolone Acetonide for Diabetic Macular Edema
 Intravitreal versus Posterior Subtenon Injection of Triamcinolone Acetonide for Diabetic Macular Edema Young Jae Choi, MD, In Kyung Oh, MD, Jae Ryung Oh, MD, PhD, Kuhl Huh, MD, PhD Department of Ophthalmology,
Intravitreal versus Posterior Subtenon Injection of Triamcinolone Acetonide for Diabetic Macular Edema Young Jae Choi, MD, In Kyung Oh, MD, Jae Ryung Oh, MD, PhD, Kuhl Huh, MD, PhD Department of Ophthalmology,
Case report 12/10/2014. Delphine Lam ; Dr Mayer Srour Service d ophtalmologie Professeur E.Souied Université Paris Est
 Case report 12/10/2014 Delphine Lam ; Dr Mayer Srour Service d ophtalmologie Professeur E.Souied Medical history Man, 75 years old Complaint: Vision loss in left eye in June 2014 Past ophthalmologic history:
Case report 12/10/2014 Delphine Lam ; Dr Mayer Srour Service d ophtalmologie Professeur E.Souied Medical history Man, 75 years old Complaint: Vision loss in left eye in June 2014 Past ophthalmologic history:
Diabetic retinopathy damage to the blood vessels in the retina. Cataract clouding of the eye s lens. Cataracts develop at an earlier age in people
 Diabetic Retinopathy What is diabetic eye disease? Diabetic eye disease refers to a group of eye problems that people with diabetes may face as a complication of diabetes. All can cause severe vision loss
Diabetic Retinopathy What is diabetic eye disease? Diabetic eye disease refers to a group of eye problems that people with diabetes may face as a complication of diabetes. All can cause severe vision loss
(12) United States Patent (10) Patent No.: US 6,440,428 B1
 USOO64.40428B1 (12) United States Patent (10) Patent No.: US 6,440,428 B1 Leander et al. (45) Date of Patent: *Aug. 27, 2002 (54) PODOPHYLLOTOXIN PREPARATION 4,235,889 A 11/1980 Evers... 514/863 CONTAINING
USOO64.40428B1 (12) United States Patent (10) Patent No.: US 6,440,428 B1 Leander et al. (45) Date of Patent: *Aug. 27, 2002 (54) PODOPHYLLOTOXIN PREPARATION 4,235,889 A 11/1980 Evers... 514/863 CONTAINING
EP B1 (19) (11) EP B1 (12) EUROPEAN PATENT SPECIFICATION
 (19) (11) EP 1 747 781 B1 (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent: 17.02. Bulletin /07 (21) Application number: 074849.2 (22) Date of filing: 13.0.0
(19) (11) EP 1 747 781 B1 (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent: 17.02. Bulletin /07 (21) Application number: 074849.2 (22) Date of filing: 13.0.0
PRODUCT MONOGRAPH. (Fluorometholone 0.1% Ophthalmic Suspension), USP. Corticosteroid
 PRODUCT MONOGRAPH Pr Sandoz Fluorometholone (Fluorometholone 0.1% Ophthalmic Suspension), USP Corticosteroid Sandoz Canada Inc., Date of Revision: June 21, 2012 145 Jules-Léger Boucherville, QC, Canada
PRODUCT MONOGRAPH Pr Sandoz Fluorometholone (Fluorometholone 0.1% Ophthalmic Suspension), USP Corticosteroid Sandoz Canada Inc., Date of Revision: June 21, 2012 145 Jules-Léger Boucherville, QC, Canada
What you can expect with OZURDEX
 Important Information About Macular Edema Following Branch or Central Retinal Vein Occlusion (RVO) and Treatment For patients with RVO What you can expect with OZURDEX Approved Use OZURDEX (dexamethasone
Important Information About Macular Edema Following Branch or Central Retinal Vein Occlusion (RVO) and Treatment For patients with RVO What you can expect with OZURDEX Approved Use OZURDEX (dexamethasone
Work Sheet And Course Hand Out
 Work Sheet And Course Hand Out This course provides the primary care health professional with a basic understanding of the eye, its function and the assessment of common sight- and non-sight threatening
Work Sheet And Course Hand Out This course provides the primary care health professional with a basic understanding of the eye, its function and the assessment of common sight- and non-sight threatening
Optometric Postoperative Cataract Surgery Management
 Financial Disclosures Optometric Postoperative Cataract Surgery Management David Dinh, OD Oak Cliff Eye Clinic Dallas Eye Consultants March 10, 2015 Comanagement Joint cooperation between two or more specialists
Financial Disclosures Optometric Postoperative Cataract Surgery Management David Dinh, OD Oak Cliff Eye Clinic Dallas Eye Consultants March 10, 2015 Comanagement Joint cooperation between two or more specialists
 measure of your overall performance. An isolated glucose test is helpful to let you know what your sugar level is at one moment, but it doesn t tell you whether or not your diabetes is under adequate control
measure of your overall performance. An isolated glucose test is helpful to let you know what your sugar level is at one moment, but it doesn t tell you whether or not your diabetes is under adequate control
(51) Int Cl.: A23D 7/00 ( ) A23D 7/015 ( ) C11C 3/04 ( ) (56) References cited:
 (19) (12) EUROPEAN PATENT SPECIFICATION (11) EP 1 931 211 B1 (4) Date of publication and mention of the grant of the patent:.12.08 Bulletin 08/0 (21) Application number: 0677699.0 (22) Date of filing:
(19) (12) EUROPEAN PATENT SPECIFICATION (11) EP 1 931 211 B1 (4) Date of publication and mention of the grant of the patent:.12.08 Bulletin 08/0 (21) Application number: 0677699.0 (22) Date of filing:
PRECISION PROGRAM. Injection Technique Quick-Reference Guide. Companion booklet for the Video Guide to Injection Technique
 Injection Technique Quick-Reference Guide PRECISION PROGRAM Companion booklet for the Video Guide to Injection Technique Available at www.ozurdexprecisionprogram.com Provides step-by-step directions with
Injection Technique Quick-Reference Guide PRECISION PROGRAM Companion booklet for the Video Guide to Injection Technique Available at www.ozurdexprecisionprogram.com Provides step-by-step directions with
Diabetic Retinopathy A Presentation for the Public
 Diabetic Retinopathy A Presentation for the Public Ray M. Balyeat, MD The Eye Institute Tulsa, Oklahoma The Healthy Eye Light rays enter the eye through the cornea, pupil and lens. These light rays are
Diabetic Retinopathy A Presentation for the Public Ray M. Balyeat, MD The Eye Institute Tulsa, Oklahoma The Healthy Eye Light rays enter the eye through the cornea, pupil and lens. These light rays are
Yoshiro Minami 1*, Taiji Nagaoka 2, Akihiro Ishibazawa 1,2 and Akitoshi Yoshida 2
 Minami et al. BMC Ophthalmology (2017) 17:90 DOI 10.1186/s12886-017-0485-4 RESEARCH ARTICLE Open Access Correlation between short- and long-term effects of intravitreal ranibizumab therapy on macular edema
Minami et al. BMC Ophthalmology (2017) 17:90 DOI 10.1186/s12886-017-0485-4 RESEARCH ARTICLE Open Access Correlation between short- and long-term effects of intravitreal ranibizumab therapy on macular edema
An A to Z guide on Epiretinal Membranes (ERMs) Paris Tranos PhD,ICO,FRCS OPHTHALMICA Vitreoretinal & Uveitis Department
 An A to Z guide on Epiretinal Membranes (ERMs) Paris Tranos PhD,ICO,FRCS OPHTHALMICA Vitreoretinal & Uveitis Department Types of ERM Natural history OCT prognostic factors ERM with co-existing pathology
An A to Z guide on Epiretinal Membranes (ERMs) Paris Tranos PhD,ICO,FRCS OPHTHALMICA Vitreoretinal & Uveitis Department Types of ERM Natural history OCT prognostic factors ERM with co-existing pathology
*EP A1* EP A1 (19) (11) EP A1 (12) EUROPEAN PATENT APPLICATION. (43) Date of publication: Bulletin 2004/12
 (19) Europäisches Patentamt European Patent Office Office européen des brevets *EP001398311A1* (11) EP 1 398 311 A1 (12) EUROPEAN PATENT APPLICATION (43) Date of publication: 17.03.04 Bulletin 04/12 (1)
(19) Europäisches Patentamt European Patent Office Office européen des brevets *EP001398311A1* (11) EP 1 398 311 A1 (12) EUROPEAN PATENT APPLICATION (43) Date of publication: 17.03.04 Bulletin 04/12 (1)
SUPPLEMENTARY INFORMATION
 SUPPLEMENTARY INFORMATION Contents METHODS... 2 Inclusion and exclusion criteria... 2 Supplementary table S1... 2 Assessment of abnormal ocular signs and symptoms... 3 Supplementary table S2... 3 Ocular
SUPPLEMENTARY INFORMATION Contents METHODS... 2 Inclusion and exclusion criteria... 2 Supplementary table S1... 2 Assessment of abnormal ocular signs and symptoms... 3 Supplementary table S2... 3 Ocular
Prednisolone Sodium Phosphate Ophthalmic Solution USP, 1% (Sterile) Rx only
 Prednisolone Sodium Phosphate Ophthalmic Solution USP, 1% (Sterile) Rx only DESCRIPTION Prednisolone Sodium Phosphate Ophthalmic Solution, 1%, is a sterile solution for ophthalmic administration having
Prednisolone Sodium Phosphate Ophthalmic Solution USP, 1% (Sterile) Rx only DESCRIPTION Prednisolone Sodium Phosphate Ophthalmic Solution, 1%, is a sterile solution for ophthalmic administration having
AUSTRALIAN PRODUCT INFORMATION FLAREX (FLUOROMETHOLONE ACETATE) EYE DROPS SUSPENSION
 AUSTRALIAN PRODUCT INFORMATION FLAREX (FLUOROMETHOLONE ACETATE) EYE DROPS SUSPENSION 1 NAME OF THE MEDICINE Fluorometholone acetate. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION The active ingredient in
AUSTRALIAN PRODUCT INFORMATION FLAREX (FLUOROMETHOLONE ACETATE) EYE DROPS SUSPENSION 1 NAME OF THE MEDICINE Fluorometholone acetate. 2 QUALITATIVE AND QUANTITATIVE COMPOSITION The active ingredient in
EP B1 (19) (11) EP B1 (12) EUROPEAN PATENT SPECIFICATION
 (19) (11) EP 2 124 94 B1 (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent: 20.04.2011 Bulletin 2011/16 (21) Application number: 0786831.7 (22) Date of filing:
(19) (11) EP 2 124 94 B1 (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent: 20.04.2011 Bulletin 2011/16 (21) Application number: 0786831.7 (22) Date of filing:
Misdiagnosed Vogt-Koyanagi-Harada (VKH) disease and atypical central serous chorioretinopathy (CSC)
 HPTER 12 Misdiagnosed Vogt-Koyanagi-Harada (VKH) disease and atypical central serous chorioretinopathy (S) linical Features VKH disease is a bilateral granulomatous panuveitis often associated with exudative
HPTER 12 Misdiagnosed Vogt-Koyanagi-Harada (VKH) disease and atypical central serous chorioretinopathy (S) linical Features VKH disease is a bilateral granulomatous panuveitis often associated with exudative
(12) United States Patent (10) Patent No.: US 6,365,596 B1
 USOO63696B1 (12) United States Patent (10) Patent No.: US 6,365,596 B1 Valenti (45) Date of Patent: Apr. 2, 2002 (54) ORAL PHARMACEUTICAL COMPOSITIONS (51) Int. Cl."... A61K 31/44 CONTAINING BUPRENORPHIN
USOO63696B1 (12) United States Patent (10) Patent No.: US 6,365,596 B1 Valenti (45) Date of Patent: Apr. 2, 2002 (54) ORAL PHARMACEUTICAL COMPOSITIONS (51) Int. Cl."... A61K 31/44 CONTAINING BUPRENORPHIN
(43) International Publication Date. 15 July 2010 ( ) WO 2010/ A3. (19) World Intellectual Property Organization International Bureau
 (12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (43) International Publication Date (10) International
(12) INTERNATIONAL APPLICATION PUBLISHED UNDER THE PATENT COOPERATION TREATY (PCT) (19) World Intellectual Property Organization International Bureau (43) International Publication Date (10) International
Diabetic Retinopathy
 Diabetic Retinopathy Diabetes mellitus is one of the leading causes of irreversible blindness worldwide. In the United States, it is the most common cause of blindness in people younger than 65 years.
Diabetic Retinopathy Diabetes mellitus is one of the leading causes of irreversible blindness worldwide. In the United States, it is the most common cause of blindness in people younger than 65 years.
TEPZZ Z6_56_B_T EP B1 (19) Zusammensetzungen zur Behandlung von Krebs Compositions servant à traiter un cancer (11) EP B1
 (19) TEPZZ Z6_6_B_T (11) EP 2 061 61 B1 (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent:.07.13 Bulletin 13/28 (21) Application number: 07837326.3 (22)
(19) TEPZZ Z6_6_B_T (11) EP 2 061 61 B1 (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent:.07.13 Bulletin 13/28 (21) Application number: 07837326.3 (22)
RVO RETINAL VEIN OCCLUSION
 RVO RETINAL VEIN OCCLUSION A guide to understanding RVO Take some time to learn about RVO - it may help you hold on to your vision Retinal vein occlusion is a common disorder of the retina and a leading
RVO RETINAL VEIN OCCLUSION A guide to understanding RVO Take some time to learn about RVO - it may help you hold on to your vision Retinal vein occlusion is a common disorder of the retina and a leading
Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial
 ASX and Media Release 3 January 2018 Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial Melbourne, Australia; January 3 2018 Opthea Limited (ASX:OPT), a late stage biopharmaceutical company
ASX and Media Release 3 January 2018 Opthea Initiates OPT-302 Diabetic Macular Edema Clinical Trial Melbourne, Australia; January 3 2018 Opthea Limited (ASX:OPT), a late stage biopharmaceutical company
Recalcitrant Diabetic Macular Oedema: Therapeutic Options
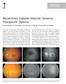 December 2007 A. Giridhar et al. - Recalcitrant DME 451 CONSULTATION S E C T I O N Recalcitrant Diabetic Macular Oedema: Therapeutic Options Dr. Cyrus M Shroff 1, Dr. N S Muralidhar 2, Dr. R Narayanan
December 2007 A. Giridhar et al. - Recalcitrant DME 451 CONSULTATION S E C T I O N Recalcitrant Diabetic Macular Oedema: Therapeutic Options Dr. Cyrus M Shroff 1, Dr. N S Muralidhar 2, Dr. R Narayanan
INVELTYS (loteprednol etabonate ophthalmic suspension) 1%, for topical ophthalmic use Initial U.S. Approval: 1998
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use INVELTYS safely and effectively. See full prescribing information for INVELTYS. INVELTYS (loteprednol
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use INVELTYS safely and effectively. See full prescribing information for INVELTYS. INVELTYS (loteprednol
(51) Int Cl.: A61N 1/20 ( )
 (19) TEPZZ _ B_T (11) EP 2 1 323 B1 (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent: 08.0.13 Bulletin 13/19 (21) Application number: 0877312.2 (22) Date
(19) TEPZZ _ B_T (11) EP 2 1 323 B1 (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent: 08.0.13 Bulletin 13/19 (21) Application number: 0877312.2 (22) Date
Date approved: 04/18/18. Approved by: Pharmacy and Therapeutics Quality Management Subcommittee Effective Date: Department of Origin: Pharmacy
 Integrated Healthcare Services and Criteria Document: Reference #: PC/V001 Page: 1 of 9 PRODUCT APPLICATION: PreferredOne Administrative Services, Inc. (PAS) ERISA PreferredOne Administrative Services,
Integrated Healthcare Services and Criteria Document: Reference #: PC/V001 Page: 1 of 9 PRODUCT APPLICATION: PreferredOne Administrative Services, Inc. (PAS) ERISA PreferredOne Administrative Services,
Update on management of Anterior Uveitis
 Update on management of Anterior Uveitis Parthopratim Dutta Majumder Senior Consultant, Department of Uvea & Intraocular Inflammation Medical Research Foundation, Sankara Nethralaya ABCD of Treating a
Update on management of Anterior Uveitis Parthopratim Dutta Majumder Senior Consultant, Department of Uvea & Intraocular Inflammation Medical Research Foundation, Sankara Nethralaya ABCD of Treating a
Retinal Vein Occlusion (RVO) Treatment pathway- Northeast England. Retinal Vein Occlusion (RVO) with Macular oedema (MO)
 Retinal Vein Occlusion (RVO) Treatment pathway- Northeast England (Royal Victoria Infirmary, Sunderland Eye Infirmary, James Cook University Hospital, Darlington Memorial Hospital, University Hospital
Retinal Vein Occlusion (RVO) Treatment pathway- Northeast England (Royal Victoria Infirmary, Sunderland Eye Infirmary, James Cook University Hospital, Darlington Memorial Hospital, University Hospital
Retina Conference. Janelle Fassbender, MD, PhD University of Louisville Department of Ophthalmology and Visual Sciences 09/04/2014
 Retina Conference Janelle Fassbender, MD, PhD University of Louisville Department of Ophthalmology and Visual Sciences 09/04/2014 Subjective CC/HPI: 64 year old Caucasian female referred by outside ophthalmologist
Retina Conference Janelle Fassbender, MD, PhD University of Louisville Department of Ophthalmology and Visual Sciences 09/04/2014 Subjective CC/HPI: 64 year old Caucasian female referred by outside ophthalmologist
NEW ZEALAND DATA SHEET 1. PRODUCT NAME
 NEW ZEALAND DATA SHEET 1. PRODUCT NAME Flucon fluorometholone 0.1% Eye Drops Suspension 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of Flucon contains 1.0 mg of fluorometholone (0.1% w/v). Excipient
NEW ZEALAND DATA SHEET 1. PRODUCT NAME Flucon fluorometholone 0.1% Eye Drops Suspension 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of Flucon contains 1.0 mg of fluorometholone (0.1% w/v). Excipient
ILUVIEN IN DIABETIC MACULAR ODEMA
 1 ILUVIEN IN DIABETIC MACULAR ODEMA Marie Tsaloumas Consultant Ophthalmic Surgeon Queen Elizabeth Hospital, Birmingham bars conference 2104 1 2 Declaration of interest I have sat on Advisory boards for
1 ILUVIEN IN DIABETIC MACULAR ODEMA Marie Tsaloumas Consultant Ophthalmic Surgeon Queen Elizabeth Hospital, Birmingham bars conference 2104 1 2 Declaration of interest I have sat on Advisory boards for
The effect of a single intravitreal implantation of dexamethasone on the fellow eye in bilateral non-infectious uveitis case report
 European Review for Medical and Pharmacological Sciences The effect of a single intravitreal implantation of dexamethasone on the fellow eye in bilateral non-infectious uveitis case report J. CISZEWSKA,
European Review for Medical and Pharmacological Sciences The effect of a single intravitreal implantation of dexamethasone on the fellow eye in bilateral non-infectious uveitis case report J. CISZEWSKA,
Test Bank for Medical Surgical Nursing An Integrated Approach 3rd Edition by White
 Test Bank for Medical Surgical Nursing An Integrated Approach 3rd Edition by White Link full download : http://testbankair.com/download/test-bank-for-medical-surgical-nursing-anintegrated-approach-3rd-edition-by-white/
Test Bank for Medical Surgical Nursing An Integrated Approach 3rd Edition by White Link full download : http://testbankair.com/download/test-bank-for-medical-surgical-nursing-anintegrated-approach-3rd-edition-by-white/
Moncef Khairallah, MD
 Moncef Khairallah, MD Department of Ophthalmology, Fattouma Bourguiba University Hospital Faculty of Medicine, University of Monastir Monastir, Tunisia INTRODUCTION IU: anatomic form of uveitis involving
Moncef Khairallah, MD Department of Ophthalmology, Fattouma Bourguiba University Hospital Faculty of Medicine, University of Monastir Monastir, Tunisia INTRODUCTION IU: anatomic form of uveitis involving
Optical Coherence Tomograpic Features in Idiopathic Retinitis, Vasculitis, Aneurysms and Neuroretinitis (IRVAN)
 Columbia International Publishing Journal of Ophthalmic Research (2014) Research Article Optical Coherence Tomograpic Features in Idiopathic Retinitis, Vasculitis, Aneurysms and Neuroretinitis (IRVAN)
Columbia International Publishing Journal of Ophthalmic Research (2014) Research Article Optical Coherence Tomograpic Features in Idiopathic Retinitis, Vasculitis, Aneurysms and Neuroretinitis (IRVAN)
Choroidal Neovascularization in Sympathetic Ophthalmia
 Choroidal Neovascularization in Sympathetic Ophthalmia Lucia Sobrin, Miguel Cordero Coma, C. Stephen Foster Case Report A 49-year-old man presented after a ruptured globe repair of his left eye status
Choroidal Neovascularization in Sympathetic Ophthalmia Lucia Sobrin, Miguel Cordero Coma, C. Stephen Foster Case Report A 49-year-old man presented after a ruptured globe repair of his left eye status
Age-related macular degeneration (AMD) and RANIBIZUMAB (Lucentis)
 Aggiornamento biotecnologico: MoAb anti VEGF Age-related macular degeneration (AMD) and RANIBIZUMAB (Lucentis) Giulia Germena AMD DRY (non-neovascular) Atrophic cell death in the macula No leakage WET
Aggiornamento biotecnologico: MoAb anti VEGF Age-related macular degeneration (AMD) and RANIBIZUMAB (Lucentis) Giulia Germena AMD DRY (non-neovascular) Atrophic cell death in the macula No leakage WET
Diabetes & Your Eyes
 Diabetes & Your Eyes Diabetes is a disease that occurs when the pancreas does not secrete enough insulin or the body is unable to process it properly. Insulin is the hormone that regulates the level of
Diabetes & Your Eyes Diabetes is a disease that occurs when the pancreas does not secrete enough insulin or the body is unable to process it properly. Insulin is the hormone that regulates the level of
Scrub In. What is the function of vitreous humor? What does the pupil do when exposed to bright light? a. Maintain eye shape and provide color vision
 Scrub In What is the function of vitreous humor? a. Maintain eye shape and provide color vision b. Maintain eye shape and refract light rays c. Provide night vision and color vision d. Provide night vision
Scrub In What is the function of vitreous humor? a. Maintain eye shape and provide color vision b. Maintain eye shape and refract light rays c. Provide night vision and color vision d. Provide night vision
Eyes on Diabetics: How to Avoid Blindness in Diabetic Patient
 Eyes on Diabetics: How to Avoid Blindness in Diabetic Patient Rova Virgana FK Unpad Pusat Mata Nasional RS Mata Cicendo Bandung Eye Center (Hospital and Clinic) PIT IDI Jabar 2018 Keys Facts from WHO
Eyes on Diabetics: How to Avoid Blindness in Diabetic Patient Rova Virgana FK Unpad Pusat Mata Nasional RS Mata Cicendo Bandung Eye Center (Hospital and Clinic) PIT IDI Jabar 2018 Keys Facts from WHO
Yasser R. Serag, MD Tamer Wasfi, MD El- Saied El-Dessoukey, MD Magdi S. Moussa, MD Anselm Kampik, MD
 Microperimetric Evaluation of Brilliant Blue G- assisted Internal Limiting Membrane Peeling By Yasser R. Serag, MD Tamer Wasfi, MD El- Saied El-Dessoukey, MD Magdi S. Moussa, MD Anselm Kampik, MD The internal
Microperimetric Evaluation of Brilliant Blue G- assisted Internal Limiting Membrane Peeling By Yasser R. Serag, MD Tamer Wasfi, MD El- Saied El-Dessoukey, MD Magdi S. Moussa, MD Anselm Kampik, MD The internal
Diagnosis and treatment of diabetic retinopathy. Blake Cooper MD Ophthalmologist Vitreoretinal Surgeon Retina Associates Kansas City
 Diagnosis and treatment of diabetic retinopathy Blake Cooper MD Ophthalmologist Vitreoretinal Surgeon Retina Associates Kansas City Disclosures Consulted for Novo Nordisk 2017,2018. Will be discussing
Diagnosis and treatment of diabetic retinopathy Blake Cooper MD Ophthalmologist Vitreoretinal Surgeon Retina Associates Kansas City Disclosures Consulted for Novo Nordisk 2017,2018. Will be discussing
What's hot and current in ophthalmology. ... and what is missing?
 What's hot and current in ophthalmology... and what is missing? 30 April 2014 Cross-sectional review of ophthalmology clinical trials 2 Sources Online searches, BioPharmClinica, clinicaltrials.gov, EU
What's hot and current in ophthalmology... and what is missing? 30 April 2014 Cross-sectional review of ophthalmology clinical trials 2 Sources Online searches, BioPharmClinica, clinicaltrials.gov, EU
New Developments in the treatment of Diabetic Retinopathy
 New Developments in the treatment of Diabetic Retinopathy B. Jeroen Klevering University Medical Centre Nijmegen - The Netherlands Topics Management of diabetic retinopathy Interventions a. primary (prevention)
New Developments in the treatment of Diabetic Retinopathy B. Jeroen Klevering University Medical Centre Nijmegen - The Netherlands Topics Management of diabetic retinopathy Interventions a. primary (prevention)
Advances in OCT Murray Fingeret, OD
 Disclosures Advances in OCT Murray Fingeret, OD Consultant Alcon, Allergan, Bausch & Lomb, Carl Zeiss Meditec, Diopsys, Heidelberg Engineering, Reichert, Topcon Currently Approved OCT Devices OCT Devices
Disclosures Advances in OCT Murray Fingeret, OD Consultant Alcon, Allergan, Bausch & Lomb, Carl Zeiss Meditec, Diopsys, Heidelberg Engineering, Reichert, Topcon Currently Approved OCT Devices OCT Devices
Optical Coherence Tomography: Pearls for the Anterior Segment Surgeon Basic Science Michael Stewart, M.D.
 Optical Coherence Tomography: Pearls for the Anterior Segment Surgeon Basic Science Michael Stewart, M.D. Disclosure OCT Optical Coherence Tomography No relevant financial relationships I will refer to
Optical Coherence Tomography: Pearls for the Anterior Segment Surgeon Basic Science Michael Stewart, M.D. Disclosure OCT Optical Coherence Tomography No relevant financial relationships I will refer to
(51) Int Cl.: A61K 47/18 ( ) A61K 47/20 ( ) A61K 47/40 ( ) A61P 29/00 ( ) (72) Inventor: Penkler, Lawrence John
 (19) (11) EP 1 767 219 B1 (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent: 13.01. Bulletin /02 (1) Int Cl.: A61K 47/18 (06.01) A61K 47/ (06.01) A61K 47/
(19) (11) EP 1 767 219 B1 (12) EUROPEAN PATENT SPECIFICATION (4) Date of publication and mention of the grant of the patent: 13.01. Bulletin /02 (1) Int Cl.: A61K 47/18 (06.01) A61K 47/ (06.01) A61K 47/
Calypso Application. License for card and portable objects.
 Calypso Application License for card and portable objects. I N N O V A T R O N CalypsoLicense page 1 / 6 Innovatron, 27 rue de Bassano, 75008 Paris, France 1. License Policy General Presentation Innovatron
Calypso Application License for card and portable objects. I N N O V A T R O N CalypsoLicense page 1 / 6 Innovatron, 27 rue de Bassano, 75008 Paris, France 1. License Policy General Presentation Innovatron
Rising to the Challenge of Satisfying Unmet Medical Needs. Back-of-the-eye. Main Back-of-the-Eye Diseases. Uveitis. Behcet s disease.
 Feature Rising to the Challenge of Satisfying Unmet Medical Needs Eyeing Further Specialization Back-of-the-eye Diseases Main Back-of-the-Eye Diseases Age-related macular degeneration Uveitis Behcet s
Feature Rising to the Challenge of Satisfying Unmet Medical Needs Eyeing Further Specialization Back-of-the-eye Diseases Main Back-of-the-Eye Diseases Age-related macular degeneration Uveitis Behcet s
FEP Medical Policy Manual
 FEP Medical Policy Manual Last Review: September 20 Next Review: September 2017 Related Policies 9.03.21 Aqueous Shunts for Glaucoma Intravitreal Corticosteroid Implants Summary An intravitreal implant
FEP Medical Policy Manual Last Review: September 20 Next Review: September 2017 Related Policies 9.03.21 Aqueous Shunts for Glaucoma Intravitreal Corticosteroid Implants Summary An intravitreal implant
The Human Eye. Cornea Iris. Pupil. Lens. Retina
 The Retina Thin layer of light-sensitive tissue at the back of the eye (the film of the camera). Light rays are focused on the retina then transmitted to the brain. The macula is the very small area in
The Retina Thin layer of light-sensitive tissue at the back of the eye (the film of the camera). Light rays are focused on the retina then transmitted to the brain. The macula is the very small area in
GENERAL INFORMATION CORNEAL TRANSPLANTATION
 GENERAL INFORMATION CORNEAL TRANSPLANTATION WHAT IS CORNEAL TRANSPLANTATION? A corneal transplant is an operation where a damaged or diseased cornea is replaced with donated, healthy tissue. Also called
GENERAL INFORMATION CORNEAL TRANSPLANTATION WHAT IS CORNEAL TRANSPLANTATION? A corneal transplant is an operation where a damaged or diseased cornea is replaced with donated, healthy tissue. Also called
Pharmaceutical Pricing & Reimbursement Information. PPRI Project Co-ordination Sabine Vogler, Gesundheit Österreich GmbH
 PPRI - Comparative Analysis PPRI Project Co-ordination Sabine Vogler, Gesundheit Österreich GmbH PPRI Conference Vienna, 29 June 2007 PPRI Conference, Vienna, 29 June 2007 1 Comparative Analysis - Outline
PPRI - Comparative Analysis PPRI Project Co-ordination Sabine Vogler, Gesundheit Österreich GmbH PPRI Conference Vienna, 29 June 2007 PPRI Conference, Vienna, 29 June 2007 1 Comparative Analysis - Outline
RETINAL CONDITIONS RETINAL CONDITIONS
 GENERAL INFORMATION RETINAL CONDITIONS RETINAL CONDITIONS WHAT ARE RETINAL CONDITIONS? Retinal conditions affect the light-sensitive tissue at the back of eye known as the retina. They include diseases
GENERAL INFORMATION RETINAL CONDITIONS RETINAL CONDITIONS WHAT ARE RETINAL CONDITIONS? Retinal conditions affect the light-sensitive tissue at the back of eye known as the retina. They include diseases
Diabetic Macular Edema Treatment in the 21st Century
 Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
VMA at the macula resulting in VMT
 Ocriplasmina for pharmacologic treatment in VMT Teresio Avitabile 1 Introduction PVD is a normal, physiologic process that occurs with aging; however, in some cases, PVD is incomplete Incomplete PVD localized
Ocriplasmina for pharmacologic treatment in VMT Teresio Avitabile 1 Introduction PVD is a normal, physiologic process that occurs with aging; however, in some cases, PVD is incomplete Incomplete PVD localized
