Scientific conclusions and detailed explanation of the scientific grounds for the differences from the PRAC recommendation
|
|
|
- Cecilia Thornton
- 6 years ago
- Views:
Transcription
1 Annex I Scientific conclusions, grounds for variation to the terms of the marketing authorisations and detailed explanation of the scientific grounds for the differences from the PRAC recommendation 1
2 Scientific conclusions and detailed explanation of the scientific grounds for the differences from the PRAC recommendation 1 Overall summary of the scientific evaluation by PRAC The renin-angiotensin system (RAS) is a hormone system that regulates blood pressure and fluid balance. RAS-acting agents act by blocking different stages of the renin-angiotensin system, lowering blood pressure and their use in the treatment of hypertension and its complications (including acute myocardial infarction, congestive heart failure and chronic kidney disease) is recommended in many current clinical guidelines. RAS-acting agents include angiotensinconverting enzyme inhibitors (ACE-inhibitors) such as benazepril, captopril, cilazapril, delapril, enalapril, fosinopril, imidapril, lisinopril, moexipril, perindopril, quinapril, ramipril, spirapril, trandolapril and zofenopril), angiotensin receptor blockers (ARBs) such as candesartan, telmisartan, valsartan, irbesartan, eprosartan, olmesartan, losartan and azilsartan and direct renin inhibitors such as aliskiren. The concept of dual RAS blockade through the combined use of several RAS-acting agents, emerged in the late 1990 s based on an experimental model hypothesising that the combined use of an ARB, an ACE-inhibitor or aliskiren could provide a more complete blockade of the RAS which could translate into better control of blood pressure and nephroprotective and cardioprotective effects. However, new data has emerged in the past years, raising doubts over the efficacy and identifying safety concerns associated with dual RAS blockade therapy through the combined use of ACE-inhibitors, ARBs or aliskiren. In particular, the publication of a metaanalysis by Makani et al 1 involving over 68,000 patients raised concerns that combining several RAS-acting agents may be associated with an increased risk of hyperkalaemia, hypotension and kidney failure, compared with the single use of one RAS-acting agent. In addition, the metaanalysis suggested that using multiple RAS-acting agents may not be more beneficial than using a single RAS-acting agent in terms of reducing overall mortality. It was noted that the Committee for Medicinal Products for Human Use (CHMP) had already conducted a review 2 under Article 20 of Regulation (EC) No 726/2004 for aliskiren-containing products, concluding that these products should be contraindicated in patients with diabetes mellitus or moderate to severe renal impairment who take ACE-inhibitors or ARBs. Having considered the new available evidence from the scientific literature and given the seriousness of the identified safety concerns, the Italian Medicines Agency (AIFA) decided to initiate a review under Article 31 of Council Directive 2001/83/EC on 17 April 2013, referring the matter to the Pharmacovigilance Risk Assessment Committee (PRAC) and requesting the PRAC to issue a recommendation on the benefit-risk of dual RAS blockade therapy through the combined use of ACE-inhibitors, ARBs or aliskiren and whether any regulatory measures should be taken on the marketing authorisations of the products involved in this procedure. The PRAC reviewed the totality of the available data, including clinical trials, meta-analysis and publications, the MAHs responses as well as the report from the Scientific Advisory Group in Cardiovascular Issues (SAG CVS). The PRAC was of the opinion that there is considerable evidence from large clinical trials and meta-analyses which conclusively demonstrates that dual RAS blockade therapy through the combined use of ACE-inhibitors, ARBs or aliskiren is associated with an increased risk of adverse events, including hypotension, hyperkalaemia and 1 Makani H, Bangalore S, Desouza KA, Shah A, Messerli FH. Efficacy and safety of dual blockade of the reninangiotensin system: meta-analysis of randomized trials. BMJ Jan 28;346:f360. doi: /bmj.f European Medicines Agency recommends new contraindications and warnings for aliskiren-containing medicines, 17/02/2012, 2
3 renal failure compared to monotherapy, in particular in patients with diabetic nephropathy. This is of particular concern, as these patients and patients with renal impaired are already particularly prone to developing hyperkalaemia. The PRAC considered that the available efficacy data indicates that dual RAS blockade therapy does not provide significant benefit in the general patient population, although there is evidence to suggest that some selected patient subpopulations may benefit from this dual RAS blockade therapy. In particular, a number of trials in heart failure patients have shown that the addition of a second RAS-acting agent may reduce hospital admissions for heart failure in patients with heart failure, which is considered a meaningful clinical endpoint. The PRAC therefore concluded that dual RAS blockade therapy should not be routinely used in the treatment of heart failure and is not recommended in the general population although it may benefit certain patients who remain symptomatic while receiving monotherapy or who cannot otherwise use alternative therapies, including potentially patients with diabetic nephropathy. Treatment should only occur under specialist supervision and subject to frequent close monitoring of renal function, electrolytes and blood pressure. The PRAC considered that the overall available data strongly suggests that the concerns identified with regard to safety and the lack of efficacy are a class effect and therefore considered that the conclusions of the review apply to all the active substances involved in the procedure. The PRAC was of the opinion that the concerns identified during this procedure with regard to the safety and the lack of efficacy of dual RAS blockade therapy could be adequately managed through changes to the product information, without the need for additional risk minimisation measures. The PRAC therefore concluded that the product information of all RAS-acting agents should be revised to reflect the identified risks and provide guidance to prescribers and patients. A warning was introduced to state that dual RAS blockade therapy through the combined use of ACE-inhibitors, ARBs or aliskiren is not recommended and, if considered absolutely necessary, should only occur under specialist supervision and subject to frequent close monitoring of renal function, electrolytes and blood pressure. The PRAC however clearly specified, based on data from the ONTARGET 3 and VA NEPHRON-D 4 studies that ACE-inhibitors and ARBs should not be used concomitantly in patients with diabetic nephropathy. The PRAC was also of the opinion that the contraindication based on the ALTITUDE 5 study data regarding the concomitant use of ACEinhibitors or ARBs with aliskiren-containing products in patients with diabetes mellitus or renal impairment (glomerular filtration rate (GFR) < 60 ml/min/1.73 m 2 ) was confirmed by the additional data reviewed and that it should also be implemented in the product information of ARBs and ACE-inhibitors. For candesartan- and valsartan-containing products, which are also authorised in the treatment of heart failure, additional information was agreed upon to reflect the fact that dual RAS blockade therapy in combination with an ACE-inhibitor may be of benefit in certain patients who cannot use other heart failure treatments, provided that they are used under specialist supervision and subject to frequent close monitoring of renal function, electrolytes and blood pressure. 3 ONgoing Telmisartan Alone and in Combination with Ramipril Global Endpoint Trial 4 Veterans Affairs Nephropathy in Diabetes 5 Aliskiren Trial in Type 2 Diabetes Using Cardiorenal Endpoints 3
4 Overall conclusion The PRAC concluded that the benefit-risk balance of RAS-acting agents remains favourable, including in the context of dual RAS blockade therapy, subject to the agreed revisions to the product information. Grounds for PRAC recommendation Whereas The PRAC considered the procedure under Article 31 of Directive 2001/83/EC initiated by Italy following the emergence of new evidence on the efficacy and safety of dual RAS blockade therapy through the combined use of ACE-inhibitors, angiotensin II receptor blockers or aliskiren, to determine whether any regulatory measures should be taken on the marketing authorisations of the products involved in this procedure; The PRAC reviewed the totality of the available data, including clinical trials, meta-analysis and publications, the MAHs responses as well as the report from the Scientific Advisory Group in Cardiovascular Issues; The PRAC was of the view that there is considerable evidence, in particular from the ONTARGET, ALTITUDE and VA NEPHRON-D trials which conclusively demonstrates that dual RAS blockade therapy through the combined use of ACE-inhibitors, angiotensin II receptor blockers or aliskiren is associated with an increased risk of adverse events, including hypotension, hyperkalaemia and renal failure compared to monotherapy; The PRAC considered that the available efficacy data indicates that dual RAS blockade therapy does not provide significant benefit in the general patient population although certain patient subpopulations may benefit from treatment, provided that it occurs only under specialist supervision and subject to frequent close monitoring of renal function, electrolytes and blood pressure; The PRAC was of the opinion that the concerns identified with regard to safety and the lack of additional efficacy of dual RAS blockade therapy are a class effect and that the conclusions of the review therefore apply to all the active substances involved in this procedure; The PRAC was of the opinion that the concerns identified with regard to the safety and the lack of additional efficacy of dual RAS blockade therapy can be adequately managed through changes to the product information, without the need for additional risk minimisation measures. The PRAC, as a consequence, concluded that the benefit-risk balance of RAS-acting agents remains favourable, provided that their product information is revised to reflect the concerns associated with dual RAS blockade therapy. Having considered the matter, the PRAC therefore recommended the variation of the marketing authorisations for RAS-acting agents. 4
5 2 Detailed explanation of the scientific grounds for differences from the PRAC recommendation Having reviewed the PRAC recommendation, the CHMP agreed with the overall scientific conclusions and grounds for recommendation. However, the CHMP considered that minor additional changes were necessary to the wording proposed for the Summary of Product Characteristics. Changes were made to the heart failure indication in section 4.4 for candesartan-containing products and in sections 4.2 and 4.4 for valsartan-containing products, in order to further harmonise the wording for the two substances. In addition, a number of typographical and QRD-related changes were made. In particular the contraindication statements recommended by the PRAC were deleted from sections 5.1 (where proposed) and from section 4.4 for aliskiren, as these were already proposed in section 4.3 and therefore considered redundant. Corresponding changes were made, where relevant, to the currently approved product information. CHMP opinion The CHMP, having considered the PRAC recommendation, agrees with the overall scientific conclusions by the PRAC and is of the opinion that the marketing authorisations for RAS-acting agents should be varied. 5
6 Annex II Amendments to relevant sections of the summary of product characteristics and package leaflet 6
7 For products containing the angiotensin-converting enzyme inhibitors (ACE-inhibitors) benazepril, captopril, cilazapril, delapril, enalapril, fosinopril, imidapril, lisinopril, moexipril, perindopril, quinapril, ramipril, spirapril, trandolapril and zofenopril, the existing product information shall be amended (insertion, replacement or deletion of the text as appropriate) to reflect the agreed wording as provided below I. Summary of Product Characteristics Section Therapeutic indications For all ACE-inhibitors with wording in section 4.1 stating that they can be used alone or in combination with other antihypertensive agents, the following cross-reference should be added: (see sections 4.3, 4.4, 4.5 and 5.1). Section Posology and method of administration For all ACE-inhibitors with wording in section 4.2 stating that they can be used alone or in combination with other antihypertensive agents, the following cross-reference should be added: (see sections 4.3, 4.4, 4.5 and 5.1). Section 4.3 Contraindication The following contraindication should be added to this section: The concomitant use of [Product name] with aliskiren-containing products is contraindicated in patients with diabetes mellitus or renal impairment (GFR < 60 ml/min/1.73 m 2 ) (see sections 4.5 and 5.1). Section Special warnings and precautions for use The following wording should be incorporated in this section: Dual blockade of the renin-angiotensin-aldosterone system (RAAS) There is evidence that the concomitant use of ACE-inhibitors, angiotensin II receptor blockers or aliskiren increases the risk of hypotension, hyperkalaemia and decreased renal function (including acute renal failure). Dual blockade of RAAS through the combined use of ACE-inhibitors, angiotensin II receptor blockers or aliskiren is therefore not recommended (see sections 4.5 and 5.1). If dual blockade therapy is considered absolutely necessary, this should only occur under specialist supervision and subject to frequent close monitoring of renal function, electrolytes and blood pressure. ACE-inhibitors and angiotensin II receptor blockers should not be used concomitantly in patients with diabetic nephropathy. Section 4.5 Interaction with other medicinal products and other forms of interaction The following wording should be added to this section: Clinical trial data has shown that dual blockade of the renin-angiotensin-aldosterone-system (RAAS) through the combined use of ACE-inhibitors, angiotensin II receptor blockers or aliskiren is associated 7
8 with a higher frequency of adverse events such as hypotension, hyperkalaemia and decreased renal function (including acute renal failure) compared to the use of a single RAAS-acting agent (see sections 4.3, 4.4 and 5.1). Section 5.1 Pharmacodynamic properties The following wording should be added to this section: Two large randomised, controlled trials (ONTARGET (ONgoing Telmisartan Alone and in combination with Ramipril Global Endpoint Trial) and VA NEPHRON-D (The Veterans Affairs Nephropathy in Diabetes)) have examined the use of the combination of an ACE-inhibitor with an angiotensin II receptor blocker. ONTARGET was a study conducted in patients with a history of cardiovascular or cerebrovascular disease, or type 2 diabetes mellitus accompanied by evidence of end-organ damage. VA NEPHRON-D was a study in patients with type 2 diabetes mellitus and diabetic nephropathy. These studies have shown no significant beneficial effect on renal and/or cardiovascular outcomes and mortality, while an increased risk of hyperkalaemia, acute kidney injury and/or hypotension as compared to monotherapy was observed. Given their similar pharmacodynamic properties, these results are also relevant for other ACE-inhibitors and angiotensin II receptor blockers. ACE-inhibitors and angiotensin II receptor blockers should therefore not be used concomitantly in patients with diabetic nephropathy. ALTITUDE (Aliskiren Trial in Type 2 Diabetes Using Cardiovascular and Renal Disease Endpoints) was a study designed to test the benefit of adding aliskiren to a standard therapy of an ACE-inhibitor or an angiotensin II receptor blocker in patients with type 2 diabetes mellitus and chronic kidney disease, cardiovascular disease, or both. The study was terminated early because of an increased risk of adverse outcomes. Cardiovascular death and stroke were both numerically more frequent in the aliskiren group than in the placebo group and adverse events and serious adverse events of interest (hyperkalaemia, hypotension and renal dysfunction) were more frequently reported in the aliskiren group than in the placebo group. II. Package leaflet The following wording should be included in the specified sections: Section 2. What you need to know before you <take> <use> X Do not <take> <use> X <:> if you have diabetes or impaired kidney function and you are treated with a blood pressure lowering medicine containing aliskiren Warnings and precautions Talk to your doctor <or> <pharmacist> <or nurse> before <taking> <using> X if you are taking any of the following medicines used to treat high blood pressure: 8
9 - an angiotensin II receptor blocker (ARBs) (also known as sartans - for example valsartan, telmisartan, irbesartan), in particular if you have diabetes-related kidney problems. - aliskiren Your doctor may check your kidney function, blood pressure, and the amount of electrolytes (e.g. potassium) in your blood at regular intervals. See also information under the heading Do not <take> <use> X Other medicines and X Tell your <doctor> <or> <pharmacist> if you are <taking> <using>, have recently <taken> <used> or might <take> <use> any other medicines. Your doctor may need to change your dose and/or to take other precautions: If you are taking an angiotensin II receptor blocker (ARB) or aliskiren (see also information under the headings Do not <take> <use> X and Warnings and precautions ) 9
10 For products containing the angiotensin II receptor blockers azilsartan, eprosartan, irbesartan, losartan, olmesartan and telmisartan, the existing product information shall be amended (insertion, replacement or deletion of the text as appropriate) to reflect the agreed wording as provided below I. Summary of Product Characteristics Section Therapeutic indications For angiotensin II receptor blockers with wording in section 4.1 stating that they can be used alone or in combination with other antihypertensive agents, the following cross-reference should be added: (see sections 4.3, 4.4, 4.5 and 5.1). Section Posology and method of administration For angiotensin II receptor blockers with wording in section 4.2 stating that they can be used alone or in combination with other antihypertensive agents, the following cross-reference should be added: (see sections 4.3, 4.4, 4.5 and 5.1). Section 4.3 Contraindication The following contraindication should be added to this section: The concomitant use of [Product name] with aliskiren-containing products is contraindicated in patients with diabetes mellitus or renal impairment (GFR < 60 ml/min/1.73 m 2 ) (see sections 4.5 and 5.1). Section Special warnings and precautions for use The following wording should be incorporated in this section: Dual blockade of the renin-angiotensin-aldosterone system (RAAS) There is evidence that the concomitant use of ACE-inhibitors, angiotensin II receptor blockers or aliskiren increases the risk of hypotension, hyperkalaemia and decreased renal function (including acute renal failure). Dual blockade of RAAS through the combined use of ACE-inhibitors, angiotensin II receptor blockers or aliskiren is therefore not recommended (see sections 4.5 and 5.1). If dual blockade therapy is considered absolutely necessary, this should only occur under specialist supervision and subject to frequent close monitoring of renal function, electrolytes and blood pressure. ACE-inhibitors and angiotensin II receptor blockers should not be used concomitantly in patients with diabetic nephropathy. Section 4.5 Interaction with other medicinal products and other forms of interaction The following wording should be added to this section: Clinical trial data has shown that dual blockade of the renin-angiotensin-aldosterone-system (RAAS) through the combined use of ACE-inhibitors, angiotensin II receptor blockers or aliskiren is associated with a higher frequency of adverse events such as hypotension, hyperkalaemia and decreased renal 10
11 function (including acute renal failure) compared to the use of a single RAAS-acting agent (see sections 4.3, 4.4 and 5.1). Section 5.1 Pharmacodynamic properties The following wording should be added to this section (for telmisartan-containing products that already have an extensive wording on ONTARGET in section 5.1, the following wording should be added in addition to the existing text, which should be maintained): Two large randomised, controlled trials (ONTARGET (ONgoing Telmisartan Alone and in combination with Ramipril Global Endpoint Trial) and VA NEPHRON-D (The Veterans Affairs Nephropathy in Diabetes)) have examined the use of the combination of an ACE-inhibitor with an angiotensin II receptor blocker. ONTARGET was a study conducted in patients with a history of cardiovascular or cerebrovascular disease, or type 2 diabetes mellitus accompanied by evidence of end-organ damage. VA NEPHRON-D was a study in patients with type 2 diabetes mellitus and diabetic nephropathy. These studies have shown no significant beneficial effect on renal and/or cardiovascular outcomes and mortality, while an increased risk of hyperkalaemia, acute kidney injury and/or hypotension as compared to monotherapy was observed. Given their similar pharmacodynamic properties, these results are also relevant for other ACE-inhibitors and angiotensin II receptor blockers. ACE-inhibitors and angiotensin II receptor blockers should therefore not be used concomitantly in patients with diabetic nephropathy. ALTITUDE (Aliskiren Trial in Type 2 Diabetes Using Cardiovascular and Renal Disease Endpoints) was a study designed to test the benefit of adding aliskiren to a standard therapy of an ACE-inhibitor or an angiotensin II receptor blocker in patients with type 2 diabetes mellitus and chronic kidney disease, cardiovascular disease, or both. The study was terminated early because of an increased risk of adverse outcomes. Cardiovascular death and stroke were both numerically more frequent in the aliskiren group than in the placebo group and adverse events and serious adverse events of interest (hyperkalaemia, hypotension and renal dysfunction) were more frequently reported in the aliskiren group than in the placebo group. II. Package leaflet The following wording should be included in the specified sections: PL Section 2. What you need to know before you <take> <use> X Do not <take> <use> X <:> if you have diabetes or impaired kidney function and you are treated with a blood pressure lowering medicine containing aliskiren Warnings and precautions Talk to your doctor <or> <pharmacist> <or nurse> before <taking> <using> X if you are taking any of the following medicines used to treat high blood pressure: 11
12 - an ACE-inhibitor (for example enalapril, lisinopril, ramipril), in particular if you have diabetesrelated kidney problems. - aliskiren Your doctor may check your kidney function, blood pressure, and the amount of electrolytes (e.g. potassium) in your blood at regular intervals. See also information under the heading Do not <take> <use> X Other medicines and X Tell your <doctor> <or> <pharmacist> if you are <taking> <using>, have recently <taken> <used> or might <take> <use> any other medicines. Your doctor may need to change your dose and/or to take other precautions: If you are taking an ACE-inhibitor or aliskiren (see also information under the headings Do not <take> <use> X and Warnings and precautions ) 12
13 For products containing candesartan, the existing product information shall be amended (insertion, replacement or deletion of the text as appropriate) to reflect the agreed wording as provided below I. Summary of Product Characteristics Section Therapeutic indications For candesartan-containing products with wording in section 4.1 stating that they can be used alone or in combination with other antihypertensive agents, the following cross-reference should be added: (see sections 4.3, 4.4, 4.5 and 5.1). In addition, the existing heart failure indication should be revised as follows: The treatment of adult patients with heart failure and impaired left ventricular systolic function (left ventricular ejection fraction 40%) when ACE-inhibitors are not tolerated or as add-on therapy to ACE-inhibitors in patients with symptomatic heart failure, despite optimal therapy, when mineralocorticoid receptor antagonists are not tolerated (see sections 4.2, 4.4, 4.5 and 5.1). Section Posology and method of administration The following cross-reference should be added to the Posology in Hypertension section: (see sections 4.3, 4.4, 4.5 and 5.1). The following wording should be added in the Posology in Heart Failure section: [Product name] can be administered with other heart failure treatment, including ACE-inhibitors, beta-blockers, diuretics and digitalis or a combination of these medicinal products. [Product name] may be co-administered with an ACE-inhibitor in patients with symptomatic heart failure despite optimal standard heart failure therapy when mineralocorticoid receptor antagonists are not tolerated. The combination of an ACE-inhibitor, a potassium-sparing diuretic and [Product name] is not recommended and should be considered only after careful evaluation of the potential benefits and risks (see sections 4.4, 4.8 and 5.1). Section 4.3 Contraindication The following contraindication should be added to this section: The concomitant use of [Product name] with aliskiren-containing products is contraindicated in patients with diabetes mellitus or renal impairment (GFR < 60 ml/min/1.73 m 2 ) (see sections 4.5 and 5.1). Section Special warnings and precautions for use The following wording should be incorporated in this section: Dual blockade of the renin-angiotensin-aldosterone system (RAAS) 13
14 There is evidence that the concomitant use of ACE-inhibitors, angiotensin II receptor blockers or aliskiren increases the risk of hypotension, hyperkalaemia and decreased renal function (including acute renal failure). Dual blockade of RAAS through the combined use of ACE-inhibitors, angiotensin II receptor blockers or aliskiren is therefore not recommended (see sections 4.5 and 5.1). If dual blockade therapy is considered absolutely necessary, this should only occur under specialist supervision and subject to frequent close monitoring of renal function, electrolytes and blood pressure. ACE-inhibitors and angiotensin II receptor blockers should not be used concomitantly in patients with diabetic nephropathy. The following wording should be incorporated in the Heart failure section: Concomitant therapy with an ACE-inhibitor in heart failure The risk of adverse reactions, especially hypotension, hyperkalaemia and decreased renal function (including acute renal failure), may increase when [Product name] is used in combination with an ACEinhibitor. Triple combination of an ACE-inhibitor, a mineralocorticoid receptor antagonist and candesartan is also not recommended. Use of these combinations should be under specialist supervision and subject to frequent close monitoring of renal function, electrolytes and blood pressure. ACE-inhibitors and angiotensin II receptor blockers should not be used concomitantly in patients with diabetic nephropathy. Section 4.5 Interaction with other medicinal products and other forms of interaction The following wording should be added to this section: Clinical trial data has shown that dual blockade of the renin-angiotensin-aldosterone-system (RAAS) through the combined use of ACE-inhibitors, angiotensin II receptor blockers or aliskiren is associated with a higher frequency of adverse events such as hypotension, hyperkalaemia and decreased renal function (including acute renal failure) compared to the use of a single RAAS-acting agent (see sections 4.3, 4.4 and 5.1). Section 5.1 Pharmacodynamic properties The following wording should be added to this section: Two large randomised, controlled trials (ONTARGET (ONgoing Telmisartan Alone and in combination with Ramipril Global Endpoint Trial) and VA NEPHRON-D (The Veterans Affairs Nephropathy in Diabetes)) have examined the use of the combination of an ACE-inhibitor with an angiotensin II receptor blocker. ONTARGET was a study conducted in patients with a history of cardiovascular or cerebrovascular disease, or type 2 diabetes mellitus accompanied by evidence of end-organ damage. VA NEPHRON-D was a study in patients with type 2 diabetes mellitus and diabetic nephropathy. These studies have shown no significant beneficial effect on renal and/or cardiovascular outcomes and mortality, while an increased risk of hyperkalaemia, acute kidney injury and/or hypotension as 14
15 compared to monotherapy was observed. Given their similar pharmacodynamic properties, these results are also relevant for other ACE-inhibitors and angiotensin II receptor blockers. ACE-inhibitors and angiotensin II receptor blockers should therefore not be used concomitantly in patients with diabetic nephropathy. ALTITUDE (Aliskiren Trial in Type 2 Diabetes Using Cardiovascular and Renal Disease Endpoints) was a study designed to test the benefit of adding aliskiren to a standard therapy of an ACE-inhibitor or an angiotensin II receptor blocker in patients with type 2 diabetes mellitus and chronic kidney disease, cardiovascular disease, or both. The study was terminated early because of an increased risk of adverse outcomes. Cardiovascular death and stroke were both numerically more frequent in the aliskiren group than in the placebo group and adverse events and serious adverse events of interest (hyperkalaemia, hypotension and renal dysfunction) were more frequently reported in the aliskiren group than in the placebo group. II. Package leaflet The following wording should be included in the specified sections: Section 1. What X is and what it is used for X can be used to treat adult heart failure patients with reduced heart muscle function when Angiotensin Converting Enzyme (ACE) inhibitors cannot be used or in addition to ACE-inhibitors when symptoms persist despite treatment and mineralocorticoid receptor antagonists (MRA) cannot be used. (ACE-inhibitors and MRAs are medicines used to treat heart failure). Section 2. What you need to know before you <take> <use> X Do not <take> <use> X <:> if you have diabetes or impaired kidney function and you are treated with a blood pressure lowering medicine containing aliskiren Warnings and precautions Talk to your doctor <or> <pharmacist> <or nurse> before <taking> <using> X if you are taking any of the following medicines used to treat high blood pressure: - an ACE-inhibitor (for example enalapril, lisinopril, ramipril), in particular if you have diabetesrelated kidney problems. - aliskiren if you are taking an ACE-inhibitor together with a medicine which belongs to the class of medicines known as mineralocorticoid receptors antagonists (MRA). These medicines are for the treatment of heart failure (see Other medicines and X ). Your doctor may check your kidney function, blood pressure, and the amount of electrolytes (e.g. potassium) in your blood at regular intervals. See also information under the heading Do not <take> <use> X Other medicines and X Tell your <doctor> <or> <pharmacist> if you are <taking> <using>, have recently <taken> <used> or might <take> <use> any other medicines. 15
16 Your doctor may need to change your dose and/or to take other precautions: - If you are taking an ACE-inhibitor or aliskiren (see also information under the headings Do not <take> <use> X and Warnings and precautions ) - If you are being treated with an ACE-inhibitor together with certain other medicines to treat your heart failure, which are known as mineralocorticoid receptors antagonists (MRA) (for example spironolactone, eplerenone). 16
17 For products containing valsartan, the existing product information shall be amended (insertion, replacement or deletion of the text as appropriate) to reflect the agreed wording as provided below I. Summary of Product Characteristics Section Therapeutic indications For valsartan-containing products with wording in section 4.1 stating that they can be used alone or in combination with other antihypertensive agents, the following cross-reference should be added: (see sections 4.3, 4.4, 4.5 and 5.1). In addition, for products authorised in the treatment of heart failure, the existing heart failure indication should be revised as follows: Heart Failure Treatment of adult patients with symptomatic heart failure when ACE-inhibitors are not tolerated or in beta-blocker intolerant patients as add-on therapy to ACE-inhibitors when mineralocorticoid receptor antagonists cannot be used (see sections 4.2, 4.4, 4.5 and 5.1). Section Posology and method of administration For valsartan-containing products with wording in section 4.2 stating that they can be used alone or in combination with other antihypertensive agents, the following cross-reference should be added: (see sections 4.3, 4.4, 4.5 and 5.1). In addition, for products authorised in the treatment of heart failure, the following wording should be added in the Heart failure section: Valsartan may be administered with other heart failure therapies. However, the triple combination of an ACE-inhibitor, valsartan and a beta blocker or a potassium-sparing diuretic is not recommended (see sections 4.4 and 5.1). Evaluation of patients with heart failure should always include assessment of renal function. Section 4.3 Contraindication The following contraindication should be added to this section: The concomitant use of [Product name] with aliskiren-containing products is contraindicated in patients with diabetes mellitus or renal impairment (GFR < 60 ml/min/1.73 m 2 ) (see sections 4.5 and 5.1). Section Special warnings and precautions for use The following wording should be incorporated in this section: Dual blockade of the renin-angiotensin-aldosterone system (RAAS) 17
18 There is evidence that the concomitant use of ACE-inhibitors, angiotensin II receptor blockers or aliskiren increases the risk of hypotension, hyperkalaemia and decreased renal function (including acute renal failure). Dual blockade of RAAS through the combined use of ACE-inhibitors, angiotensin II receptor blockers or aliskiren is therefore not recommended (see sections 4.5 and 5.1). If dual blockade therapy is considered absolutely necessary, this should only occur under specialist supervision and subject to frequent close monitoring of renal function, electrolytes and blood pressure. ACE-inhibitors and angiotensin II receptor blockers should not be used concomitantly in patients with diabetic nephropathy. In addition, for products authorised in the treatment of heart failure, the following wording should be incorporated in the Heart failure section: Heart failure The risk of adverse reactions, especially hypotension, hyperkalaemia and decreased renal function (including acute renal failure), may increase when [Product name] is used in combination with an ACEinhibitor. In patients with heart failure, the triple combination of an ACE-inhibitor, a beta blocker and [Product name] has not shown any clinical benefit (see section 5.1). This combination apparently increases the risk for adverse events and is therefore not recommended. Triple combination of an ACE-inhibitor, a mineralocorticoid receptor antagonist and valsartan is also not recommended. Use of these combinations should be under specialist supervision and subject to frequent close monitoring of renal function, electrolytes and blood pressure. Caution should be observed when initiating therapy in patients with heart failure. Evaluation of patients with heart failure should always include assessment of renal function (see section 4.2). Use of [Product name] in patients with heart failure commonly results in some reduction in blood pressure, but discontinuation of therapy because of continuing symptomatic hypotension is not usually necessary provided dosing instructions are followed (see section 4.2). In patients whose renal function may depend on the activity of the renin-angiotensin-aldosteronesystem (e.g. patients with severe congestive heart failure), treatment with ACE-inhibitors has been associated with oliguria and/or progressive azotaemia and in rare cases with acute renal failure and/or death. As valsartan is an angiotensin II receptor blocker, it cannot be excluded that the use of [Product name] may be associated with impairment of the renal function. ACE-inhibitors and angiotensin II receptor blockers should not be used concomitantly in patients with diabetic nephropathy. Section 4.5 Interaction with other medicinal products and other forms of interaction The following wording should be added to this section: Clinical trial data has shown that dual blockade of the renin-angiotensin-aldosterone-system (RAAS) through the combined use of ACE-inhibitors, angiotensin II receptor blockers or aliskiren is associated with a higher frequency of adverse events such as hypotension, hyperkalaemia and decreased renal 18
19 function (including acute renal failure) compared to the use of a single RAAS-acting agent (see sections 4.3, 4.4 and 5.1). Section 5.1 Pharmacodynamic properties The following wording should be added to this section: Two large randomised, controlled trials (ONTARGET (ONgoing Telmisartan Alone and in combination with Ramipril Global Endpoint Trial) and VA NEPHRON-D (The Veterans Affairs Nephropathy in Diabetes)) have examined the use of the combination of an ACE-inhibitor with an angiotensin II receptor blocker. ONTARGET was a study conducted in patients with a history of cardiovascular or cerebrovascular disease, or type 2 diabetes mellitus accompanied by evidence of end-organ damage. VA NEPHRON-D was a study in patients with type 2 diabetes mellitus and diabetic nephropathy. These studies have shown no significant beneficial effect on renal and/or cardiovascular outcomes and mortality, while an increased risk of hyperkalaemia, acute kidney injury and/or hypotension as compared to monotherapy was observed. Given their similar pharmacodynamic properties, these results are also relevant for other ACE-inhibitors and angiotensin II receptor blockers. ACE-inhibitors and angiotensin II receptor blockers should therefore not be used concomitantly in patients with diabetic nephropathy. ALTITUDE (Aliskiren Trial in Type 2 Diabetes Using Cardiovascular and Renal Disease Endpoints) was a study designed to test the benefit of adding aliskiren to a standard therapy of an ACE-inhibitor or an angiotensin II receptor blocker in patients with type 2 diabetes mellitus and chronic kidney disease, cardiovascular disease, or both. The study was terminated early because of an increased risk of adverse outcomes. Cardiovascular death and stroke were both numerically more frequent in the aliskiren group than in the placebo group and adverse events and serious adverse events of interest (hyperkalaemia, hypotension and renal dysfunction) were more frequently reported in the aliskiren group than in the placebo group. II. Package leaflet The following wording should be included as applicable in the specified sections: Section 1. What X is and what it is used for X can be used to treat symptomatic heart failure in adult patients. X is used when a group of medicines called Angiotensin Converting Enzyme (ACE) inhibitors (a medication to treat heart failure) cannot be used or it may be used in addition to ACE-inhibitors when other medications to treat heart failure cannot be used. Section 2. What you need to know before you <take> <use> X Do not <take> <use> X <:> if you have diabetes or impaired kidney function and you are treated with a blood pressure lowering medicine containing aliskiren 19
20 Warnings and precautions Talk to your doctor <or> <pharmacist> <or nurse> before <taking> <using> X if you are taking any of the following medicines used to treat high blood pressure: - an ACE-inhibitor (for example enalapril, lisinopril, ramipril), in particular if you have diabetesrelated kidney problems. - aliskiren - if you are being treated with an ACE-inhibitor together with certain other medicines to treat your heart failure, which are known as mineralocorticoid receptors antagonists (MRA) (for example spironolactone, eplerenone) or betablockers (for example metoprolol). Your doctor may check your kidney function, blood pressure, and the amount of electrolytes (e.g. potassium) in your blood at regular intervals. See also information under the heading Do not <take> <use> X Other medicines and X Tell your <doctor> <or> <pharmacist> if you are <taking> <using>, have recently <taken> <used> or might <take> <use> any other medicines. Your doctor may need to change your dose and/or to take other precautions: If you are taking an ACE-inhibitor or aliskiren (see also information under the headings Do not <take> <use> X and Warnings and precautions ) If you are being treated with an ACE-inhibitor together with certain other medicines to treat your heart failure, which are known as mineralocorticoid receptors antagonists (MRA) (for example spironolactone, eplerenone) or betablockers (for example metoprolol). 20
21 For products containing aliskiren, the existing product information shall be amended (insertion, replacement or deletion of the text as appropriate) to reflect the agreed wording as provided below I. Summary of Product Characteristics Section 4.3 Contraindication The following contraindication should be reflected in this section: The concomitant use of [Product name] with an ACE-inhibitor or an angiotensin II receptor blocker is contraindicated in patients with diabetes mellitus or renal impairment (GFR < 60 ml/min/1.73 m 2 ) (see sections 4.4, 4.5 and 5.1). Section Special warnings and precautions for use The following wording should be reflected in this section: Dual blockade of the renin-angiotensin-aldosterone-system (RAAS) Hypotension, syncope, stroke, hyperkalaemia and decreased renal function (including acute renal failure) have been reported in susceptible individuals, especially if combining medicinal products that affect this system (see section 5.1). Dual blockade of the RAAS by combining aliskiren with an ACEinhibitor or an angiotensin II receptor blocker is therefore not recommended. If dual blockade therapy is considered absolutely necessary, this should only occur under specialist supervision and subject to frequent close monitoring of renal function, electrolytes, and blood pressure. Section 4.5 Interaction with other medicinal products and other forms of interaction The following wording should be added to this section: Clinical trial data has shown that dual blockade of the renin-angiotensin-aldosterone system (RAAS) through the combined use of ACE-inhibitors, angiotensin II receptor blockers or aliskiren is associated with a higher frequency of adverse events such as hypotension, stroke, hyperkalaemia and decreased renal function (including acute renal failure) compared to the use of a single RAAS-acting agent (see sections 4.3, 4.4 and 5.1). II. Package leaflet The following wording should be included in the specified sections: Section 2. What you need to know before you <take> <use> X Do not <take> <use> X <:> if you have diabetes mellitus or impaired kidney function and you are treated with either of the following classes of medicines used to treat high blood pressure: - an ACE-inhibitor such as enalapril, lisinopril, ramipril. or - an angiotensin II receptor blocker such as valsartan, telmisartan, irbesartan. 21
22 Warnings and precautions Talk to your doctor <or> <pharmacist> <or nurse> before <taking> <using> X if you are taking either of the following classes of medicines used to treat high blood pressure: - an ACE-inhibitor such as enalapril, lisinopril, ramipril. or - an angiotensin II receptor blocker such as valsartan, telmisartan, irbesartan. Your doctor may check your kidney function, blood pressure, and the amount of electrolytes (e.g. potassium) in your blood at regular intervals. See also information under the heading Do not <take> <use> X Other medicines and X If you are taking an angiotensin II receptor blocker (ARB) or an ACE-inhibitor, (see also information under the headings Do not <take> <use> X and Warnings and precautions ) 22
Scientific conclusions and detailed explanation of the scientific grounds for the differences from the PRAC recommendation
 Annex I Scientific conclusions, grounds for variation to the terms of the marketing authorisations and detailed explanation of the scientific grounds for the differences from the PRAC recommendation 1
Annex I Scientific conclusions, grounds for variation to the terms of the marketing authorisations and detailed explanation of the scientific grounds for the differences from the PRAC recommendation 1
ANGIOTENSIN II RECEPTOR BLOCKERS: MORE THAN THE ALTERNATIVE PRESENTATION BY: PATRICK HO, USC PHARM D. CANDIDATE OF 2017 MENTOR: DR.
 ANGIOTENSIN II RECEPTOR BLOCKERS: MORE THAN THE ALTERNATIVE PRESENTATION BY: PATRICK HO, USC PHARM D. CANDIDATE OF 2017 MENTOR: DR. CRAIG STERN, PHARMD, MBA, RPH, FASCP, FASHP, FICA, FLMI, FAMCP RENIN-ANGIOTENSIN
ANGIOTENSIN II RECEPTOR BLOCKERS: MORE THAN THE ALTERNATIVE PRESENTATION BY: PATRICK HO, USC PHARM D. CANDIDATE OF 2017 MENTOR: DR. CRAIG STERN, PHARMD, MBA, RPH, FASCP, FASHP, FICA, FLMI, FAMCP RENIN-ANGIOTENSIN
Guidelines for the Prescribing of Sacubitril / Valsartan
 Hull & East Riding Prescribing Committee Guidelines for the Prescribing of Sacubitril / Valsartan 1. BACKGROUND Sacubitril valsartan is an angiotensin receptor neprilysin inhibitor, including both a neprilysin
Hull & East Riding Prescribing Committee Guidelines for the Prescribing of Sacubitril / Valsartan 1. BACKGROUND Sacubitril valsartan is an angiotensin receptor neprilysin inhibitor, including both a neprilysin
State of the art treatment of hypertension: established and new drugs. Prof. M. Burnier Service of Nephrology and Hypertension Lausanne, Switzerland
 State of the art treatment of hypertension: established and new drugs Prof. M. Burnier Service of Nephrology and Hypertension Lausanne, Switzerland First line therapies in hypertension ACE inhibitors AT
State of the art treatment of hypertension: established and new drugs Prof. M. Burnier Service of Nephrology and Hypertension Lausanne, Switzerland First line therapies in hypertension ACE inhibitors AT
Clinical Policy: ACEI and ARB Duplicate Therapy Reference Number: CP.PMN.61 Effective Date: Last Review Date: 05.18
 Clinical Policy: Reference Number: CP.PMN.61 Effective Date: 08.01.14 Last Review Date: 05.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: Reference Number: CP.PMN.61 Effective Date: 08.01.14 Last Review Date: 05.18 Line of Business: Medicaid Revision Log See Important Reminder at the end of this policy for important regulatory
Elements for a public summary
 VI.2 VI.2.1 Elements for a public summary Overview of disease epidemiology Telmisartan/hydrochlorothiazide is a combination of an angiotensin II receptor antagonist and a thiazide diuretic used in the
VI.2 VI.2.1 Elements for a public summary Overview of disease epidemiology Telmisartan/hydrochlorothiazide is a combination of an angiotensin II receptor antagonist and a thiazide diuretic used in the
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 27 May 2009
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 27 May 2009 RASILEZ HCT 150 mg/12.5 mg, film-coated tablets B/30 (CIP code: 392 151-6) RASILEZ HCT 150 mg/25 mg, film-coated
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 27 May 2009 RASILEZ HCT 150 mg/12.5 mg, film-coated tablets B/30 (CIP code: 392 151-6) RASILEZ HCT 150 mg/25 mg, film-coated
Annex I. Scientific conclusions and grounds for the variation to the terms of the Marketing Authorisation(s)
 Annex I Scientific conclusions and grounds for the variation to the terms of the Marketing Authorisation(s) 1 Scientific conclusions Taking into account the PRAC Assessment Report on the PSUR(s) for captopril
Annex I Scientific conclusions and grounds for the variation to the terms of the Marketing Authorisation(s) 1 Scientific conclusions Taking into account the PRAC Assessment Report on the PSUR(s) for captopril
Conversion of losartan to lisinopril
 Cari untuk: Cari Cari Conversion of losartan to lisinopril Dania Alsammarae, Strategy Director and co-founder of Anglo Arabian Healthcare speaks with Neil Halligan of Arabian Business on what it takes
Cari untuk: Cari Cari Conversion of losartan to lisinopril Dania Alsammarae, Strategy Director and co-founder of Anglo Arabian Healthcare speaks with Neil Halligan of Arabian Business on what it takes
HEART FAILURE SUMMARY. and is associated with significant morbidity and mortality. the cornerstone of heart failure treatment.
 HEART FAILURE SUMMARY + Heart Failure is a condition affecting a large number of Irish people and is associated with significant morbidity and mortality. + ACE inhibitors, in combination with diuretics,
HEART FAILURE SUMMARY + Heart Failure is a condition affecting a large number of Irish people and is associated with significant morbidity and mortality. + ACE inhibitors, in combination with diuretics,
Annex I. Scientific conclusions and grounds for the variation to the terms of the Marketing Authorisation(s)
 Annex I Scientific conclusions and grounds for the variation to the terms of the Marketing Authorisation(s) 1 Scientific conclusions Taking into account the PRAC Assessment Report on the PSUR(s) for cilazapril,
Annex I Scientific conclusions and grounds for the variation to the terms of the Marketing Authorisation(s) 1 Scientific conclusions Taking into account the PRAC Assessment Report on the PSUR(s) for cilazapril,
Scientific conclusions
 Annex II Scientific conclusions and grounds for amendment of the summary of product characteristics, labelling and package leaflet presented by the European Medicines Agency 18 Scientific conclusions Overall
Annex II Scientific conclusions and grounds for amendment of the summary of product characteristics, labelling and package leaflet presented by the European Medicines Agency 18 Scientific conclusions Overall
VA/DoD Clinical Practice Guideline for the Management of Chronic Kidney Disease in Primary Care (2008) PROVIDER REFERENCE CARDS Chronic Kidney Disease
 VA/DoD Clinical Practice Guideline for the Management of Chronic Kidney Disease in Primary Care (2008) PROVIDER REFERECE CARDS Chronic Kidney Disease CKD VA/DoD Clinical Practice Guideline for the Management
VA/DoD Clinical Practice Guideline for the Management of Chronic Kidney Disease in Primary Care (2008) PROVIDER REFERECE CARDS Chronic Kidney Disease CKD VA/DoD Clinical Practice Guideline for the Management
The CARI Guidelines Caring for Australasians with Renal Impairment. ACE Inhibitor and Angiotensin II Antagonist Combination Treatment GUIDELINES
 ACE Inhibitor and Angiotensin II Antagonist Combination Treatment Date written: September 2004 Final submission: September 2005 Author: Kathy Nicholls GUIDELINES No recommendations possible based on Level
ACE Inhibitor and Angiotensin II Antagonist Combination Treatment Date written: September 2004 Final submission: September 2005 Author: Kathy Nicholls GUIDELINES No recommendations possible based on Level
Aggressive blood pressure reduction and renin angiotensin system blockade in chronic kidney disease: time for re-evaluation?
 http://www.kidney-international.org & 2013 International Society of Nephrology Aggressive blood pressure reduction and renin angiotensin system blockade in chronic kidney disease: time for re-evaluation?
http://www.kidney-international.org & 2013 International Society of Nephrology Aggressive blood pressure reduction and renin angiotensin system blockade in chronic kidney disease: time for re-evaluation?
PUBLIC SUMMARY OF RISK MANAGEMENT PLAN (RMP) CANDESARTAN/HYDROCHLOROTHIAZIDE ORION 8 MG/12.5 MG, 16 MG/12.5 MG, 32 MG/12.5 MG, 32 MG/25 MG ORION
 PUBLIC SUMMARY OF RISK MANAGEMENT PLAN (RMP) CANDESARTAN/HYDROCHLOROTHIAZIDE ORION 8 MG/12.5 MG, 16 MG/12.5 MG, 32 MG/12.5 MG, 32 MG/25 MG ORION CORPORATION DATE: 17-04-2015, VERSION 2 VI.2 Elements for
PUBLIC SUMMARY OF RISK MANAGEMENT PLAN (RMP) CANDESARTAN/HYDROCHLOROTHIAZIDE ORION 8 MG/12.5 MG, 16 MG/12.5 MG, 32 MG/12.5 MG, 32 MG/25 MG ORION CORPORATION DATE: 17-04-2015, VERSION 2 VI.2 Elements for
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Candesartan cilexetil 16 mg tablets 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Kairasec 16 mg tablets: Each tablet contains 16 mg of candesartan
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Candesartan cilexetil 16 mg tablets 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Kairasec 16 mg tablets: Each tablet contains 16 mg of candesartan
Lisinopril 20 converting to losartan
 Search Lisinopril 20 converting to losartan Stop wasting your time with unanswered searches. lisinopril 40 mg to losartan conversion,cannot Find low price Best. Winds SSW at 10 to 20. Lisinopril 20 to
Search Lisinopril 20 converting to losartan Stop wasting your time with unanswered searches. lisinopril 40 mg to losartan conversion,cannot Find low price Best. Winds SSW at 10 to 20. Lisinopril 20 to
Factors Involved in Poor Control of Risk Factors
 Factors Involved in Poor Control of Risk Factors Patient compliance Clinical inertia Health Care System structure 14781 M Limitations of Formal Studies Selection of patients Recruitment and follow-up alter
Factors Involved in Poor Control of Risk Factors Patient compliance Clinical inertia Health Care System structure 14781 M Limitations of Formal Studies Selection of patients Recruitment and follow-up alter
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 7 January 2009
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 7 January 2009 LERCAPRESS 10 mg/10 mg, film-coated tablets Pack of 30 (CIP code: 385 953-3) Pack of 90 (CIP code:
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 7 January 2009 LERCAPRESS 10 mg/10 mg, film-coated tablets Pack of 30 (CIP code: 385 953-3) Pack of 90 (CIP code:
Antihypertensive Agents Part-2. Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia
 Antihypertensive Agents Part-2 Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia Agents that block production or action of angiotensin Angiotensin-converting
Antihypertensive Agents Part-2 Assistant Prof. Dr. Najlaa Saadi PhD Pharmacology Faculty of Pharmacy University of Philadelphia Agents that block production or action of angiotensin Angiotensin-converting
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Valsartan Sandoz 40 mg film-coated tablets Valsartan Sandoz 80 mg film-coated tablets Valsartan Sandoz 160 mg film-coated tablets Valsartan
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Valsartan Sandoz 40 mg film-coated tablets Valsartan Sandoz 80 mg film-coated tablets Valsartan Sandoz 160 mg film-coated tablets Valsartan
PART VI: SUMMARY OF THE RISK MANAGEMENT PLAN
 PART VI: SUMMARY OF THE RISK MANAGEMENT PLAN VI.1 Summary of activities in the risk management plan The summary below was prepared based on the information included in Part II, IV and V of the present
PART VI: SUMMARY OF THE RISK MANAGEMENT PLAN VI.1 Summary of activities in the risk management plan The summary below was prepared based on the information included in Part II, IV and V of the present
Adapted d from Federation of Health Regulatory Colleges of Ontario Template Last Updated September 18, 2017
 Insert Logo or Org Name Here Primary Care Medical Directive for Hypertension Management Adapted d from Federation of Health Regulatory Colleges of Ontario Template Last Updated September 18, 2017 Title:
Insert Logo or Org Name Here Primary Care Medical Directive for Hypertension Management Adapted d from Federation of Health Regulatory Colleges of Ontario Template Last Updated September 18, 2017 Title:
0BCore Safety Profile. Pharmaceutical form(s)/strength: Film-coated tablet 40, 80, 160, 320 mg SE/H/PSUR/0024/003 Date of FAR:
 0BCore Safety Profile Active substance: Valsartan Pharmaceutical form(s)/strength: Film-coated tablet 40, 80, 160, 320 mg P-RMS: SE/H/PSUR/0024/003 Date of FAR: 28.02.2013 4.2 Posology and method of administration
0BCore Safety Profile Active substance: Valsartan Pharmaceutical form(s)/strength: Film-coated tablet 40, 80, 160, 320 mg P-RMS: SE/H/PSUR/0024/003 Date of FAR: 28.02.2013 4.2 Posology and method of administration
ASEBP and ARTA TARP Drugs and Reference Price by Categories
 ASEBP Pantoprazole Sodium 40 mg (generic) $0.2016 ASEBP Dexlansoprazole 30 mg Dexlansoprazole 60 mg Esomeprazole 10 mg Esomeprazole 20 mg Esomeprazole 40 mg Lansoprazole 15 mg Lansoprazole 30 mg Omeprazole
ASEBP Pantoprazole Sodium 40 mg (generic) $0.2016 ASEBP Dexlansoprazole 30 mg Dexlansoprazole 60 mg Esomeprazole 10 mg Esomeprazole 20 mg Esomeprazole 40 mg Lansoprazole 15 mg Lansoprazole 30 mg Omeprazole
Management of Hypertension
 Clinical Practice Guidelines Management of Hypertension Definition and classification of blood pressure levels (mmhg) Category Systolic Diastolic Normal
Clinical Practice Guidelines Management of Hypertension Definition and classification of blood pressure levels (mmhg) Category Systolic Diastolic Normal
Entresto Development of sacubitril/valsartan (LCZ696) for the treatment of heart failure with reduced ejection fraction
 Cardio-Metabolic Franchise Entresto Development of sacubitril/valsartan (LCZ696) for the treatment of heart failure with reduced ejection fraction Randy L Webb, PhD Rutgers Workshop October 21, 2016 Heart
Cardio-Metabolic Franchise Entresto Development of sacubitril/valsartan (LCZ696) for the treatment of heart failure with reduced ejection fraction Randy L Webb, PhD Rutgers Workshop October 21, 2016 Heart
Losartan lisinopril equivalent dose
 新着 news 2018 年 2 月 28 日 jica 国際協力中学生 高校生エッセイコンテスト 2017 表彰式 2018 年 2 月 19 日 2017 年度第 8 回卒業式を挙行. CHFpatients.com - Ace inhibitors explained in plain English - the primary drug treatment for heart failure
新着 news 2018 年 2 月 28 日 jica 国際協力中学生 高校生エッセイコンテスト 2017 表彰式 2018 年 2 月 19 日 2017 年度第 8 回卒業式を挙行. CHFpatients.com - Ace inhibitors explained in plain English - the primary drug treatment for heart failure
Neprilysin Inhibitor (Entresto ) Prior Authorization and Quantity Limit Program Summary
 Neprilysin Inhibitor (Entresto ) Prior Authorization and Quantity Limit Program Summary FDA APPROVED INDICATIONS DOSAGE 1 Indication Entresto Reduce the risk of cardiovascular (sacubitril/valsartan) death
Neprilysin Inhibitor (Entresto ) Prior Authorization and Quantity Limit Program Summary FDA APPROVED INDICATIONS DOSAGE 1 Indication Entresto Reduce the risk of cardiovascular (sacubitril/valsartan) death
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS. Medicinal product no longer authorised
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 This medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Healthcare professionals are asked
eplerenone 25, 50mg film-coated tablets (Inspra ) SMC No. (793/12) Pfizer Ltd
 eplerenone 25, 50mg film-coated tablets (Inspra ) SMC No. (793/12) Pfizer Ltd 08 June 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises NHS Boards
eplerenone 25, 50mg film-coated tablets (Inspra ) SMC No. (793/12) Pfizer Ltd 08 June 2012 The Scottish Medicines Consortium (SMC) has completed its assessment of the above product and advises NHS Boards
Use of cilazapril in New Zealand is very high compared to other countries
 Cardiovascular system Medicine indications Prescribing ACE inhibitors: time to reconsider old habits Prescribers in New Zealand are highly reliant on cilazapril as their go-to ACE inhibitor, but this prescribing
Cardiovascular system Medicine indications Prescribing ACE inhibitors: time to reconsider old habits Prescribers in New Zealand are highly reliant on cilazapril as their go-to ACE inhibitor, but this prescribing
ROLE OF ANGIOTENSIN CONVERTING ENZYME INHIBITORS AND ANGIOTENSIN RECEPTOR BLOCKERS IN TYPE I DIABETIC NEPHROPATHY DR.NASIM MUSA
 ROLE OF ANGIOTENSIN CONVERTING ENZYME INHIBITORS AND ANGIOTENSIN RECEPTOR BLOCKERS IN TYPE I DIABETIC NEPHROPATHY DR.NASIM MUSA Type I IDDM is characterized by The abrupt onset of symptoms Insulinopenia
ROLE OF ANGIOTENSIN CONVERTING ENZYME INHIBITORS AND ANGIOTENSIN RECEPTOR BLOCKERS IN TYPE I DIABETIC NEPHROPATHY DR.NASIM MUSA Type I IDDM is characterized by The abrupt onset of symptoms Insulinopenia
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Edarbi 20 mg tablets Edarbi 40 mg tablets Edarbi 80 mg tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Edarbi 20 mg tablets
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Edarbi 20 mg tablets Edarbi 40 mg tablets Edarbi 80 mg tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Edarbi 20 mg tablets
ESC Guidelines for the Diagnosis and Treatment of Acute and Chronic Heart Failure
 Patients t with acute heart failure frequently develop chronic heart failure Patients with chronic heart failure frequently decompensate acutely ESC Guidelines for the Diagnosis and A clinical response
Patients t with acute heart failure frequently develop chronic heart failure Patients with chronic heart failure frequently decompensate acutely ESC Guidelines for the Diagnosis and A clinical response
Eplerenon Medical Valley + Eplerenon Stada
 VI.2 Elements for a Public Summary VI.2.1 Overview of disease epidemiology Heart failure is a complex syndrome, clinically characterized by signs and symptoms secondary to abnormal cardiac function. It
VI.2 Elements for a Public Summary VI.2.1 Overview of disease epidemiology Heart failure is a complex syndrome, clinically characterized by signs and symptoms secondary to abnormal cardiac function. It
RAS Blockade Across the CV Continuum
 A Summary of Recent International Meetings RAS Blockade Across the CV Continuum Copyright New Evidence Presented at the 2009 Congress of the European Society of Cardiology (August 29-September 2, Barcelona)
A Summary of Recent International Meetings RAS Blockade Across the CV Continuum Copyright New Evidence Presented at the 2009 Congress of the European Society of Cardiology (August 29-September 2, Barcelona)
Package leaflet: Information for the patient
 Package leaflet: Information for the patient Valsartan Krka 40 mg film-coated tablets Valsartan Krka 80 mg film-coated tablets Valsartan Krka 160 mg film-coated tablets Valsartan Krka 320 mg film-coated
Package leaflet: Information for the patient Valsartan Krka 40 mg film-coated tablets Valsartan Krka 80 mg film-coated tablets Valsartan Krka 160 mg film-coated tablets Valsartan Krka 320 mg film-coated
Angiotensin II Receptor Blockers Dosage in hypertensive patients as well as patients with left ventricular hypertrophy
 Angiotensin II Receptor Blockers [Developed, August 1996; Revised, December 1996; July 1997; June 1998; July 1999; September 2000; July 2001; September 2001; July 2002; July 2003; April 2008; March 2011;
Angiotensin II Receptor Blockers [Developed, August 1996; Revised, December 1996; July 1997; June 1998; July 1999; September 2000; July 2001; September 2001; July 2002; July 2003; April 2008; March 2011;
Heart Failure: Current Management Strategies
 Heart Failure: Current Management Strategies CSHP Fall Education Session- September 30th, 2017 Carolyn MacKinnon & Tamara Matchett BscPharm, ACPR Candidates Objectives 1. Describe the pathophysiology &
Heart Failure: Current Management Strategies CSHP Fall Education Session- September 30th, 2017 Carolyn MacKinnon & Tamara Matchett BscPharm, ACPR Candidates Objectives 1. Describe the pathophysiology &
ACE Inhibitors and ARBs To Protect Your Heart? A Guide for Patients Being Treated for Stable Coronary Heart Disease
 ACE Inhibitors and ARBs To Protect Your Heart? A Guide for Patients Being Treated for Stable Coronary Heart Disease Is This Guide Right for Me? This Guide Is for You If: You have coronary heart disease,
ACE Inhibitors and ARBs To Protect Your Heart? A Guide for Patients Being Treated for Stable Coronary Heart Disease Is This Guide Right for Me? This Guide Is for You If: You have coronary heart disease,
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Diovan 40 mg film-coated tablets Diovan 80 mg film-coated tablets Diovan 160 mg film-coated tablets Diovan 320 mg film-coated tablets
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Diovan 40 mg film-coated tablets Diovan 80 mg film-coated tablets Diovan 160 mg film-coated tablets Diovan 320 mg film-coated tablets
ACP Brief Fall 2006 prioritization. Angiotensin II Receptor Blockers (ARBs) for Proteinuria, Hypertension (HTN) and Congestive Heart Failure (CHF)
 ACP Brief Fall 2006 prioritization Angiotensin II Receptor Blockers (ARBs) for Proteinuria, Hypertension (HTN) and Congestive Heart Failure (CHF) Background This topic was submitted by BC PharmaCare during
ACP Brief Fall 2006 prioritization Angiotensin II Receptor Blockers (ARBs) for Proteinuria, Hypertension (HTN) and Congestive Heart Failure (CHF) Background This topic was submitted by BC PharmaCare during
Drugs acting on the reninangiotensin-aldosterone
 Drugs acting on the reninangiotensin-aldosterone system John McMurray Eugene Braunwald Scholar in Cardiovascular Diseases, Brigham and Women s Hospital, Boston & Visiting Professor, Harvard Medical School
Drugs acting on the reninangiotensin-aldosterone system John McMurray Eugene Braunwald Scholar in Cardiovascular Diseases, Brigham and Women s Hospital, Boston & Visiting Professor, Harvard Medical School
By Prof. Khaled El-Rabat
 What is The Optimum? By Prof. Khaled El-Rabat Professor of Cardiology - Benha Faculty of Medicine HT. Introduction Despite major worldwide efforts over recent decades directed at diagnosing and treating
What is The Optimum? By Prof. Khaled El-Rabat Professor of Cardiology - Benha Faculty of Medicine HT. Introduction Despite major worldwide efforts over recent decades directed at diagnosing and treating
Position Statement on ALDOSTERONE ANTAGONIST THERAPY IN CHRONIC HEART FAILURE
 Position Statement on ALDOSTERONE ANTAGONIST THERAPY IN CHRONIC HEART FAILURE Over 8,000 patients have been studied in two well-designed placebo-controlled outcome-driven clinical trials to evaluate the
Position Statement on ALDOSTERONE ANTAGONIST THERAPY IN CHRONIC HEART FAILURE Over 8,000 patients have been studied in two well-designed placebo-controlled outcome-driven clinical trials to evaluate the
Don t let the pressure get to you:
 Balanced information for better care Don t let the pressure get to you: Current evidence-based goals for treating hypertension A cornerstone of primary care: Lowering high blood pressure prevents cardiovascular
Balanced information for better care Don t let the pressure get to you: Current evidence-based goals for treating hypertension A cornerstone of primary care: Lowering high blood pressure prevents cardiovascular
27 August 2013 EMA/479605/2013/ 7 Westferry Circus Canary Wharf London E14 4HB United Kingdom
 27 August 2013 EMA/479605/2013/ Trends in co-prescribing of renin-angiotensin system (RAS) - acting agents in France, Germany and the UK during 2001 2012 EMA drug utilisation study using IMS Health electronic
27 August 2013 EMA/479605/2013/ Trends in co-prescribing of renin-angiotensin system (RAS) - acting agents in France, Germany and the UK during 2001 2012 EMA drug utilisation study using IMS Health electronic
Clinical cases with Coversyl 10 mg
 Clinical cases Coversyl 10 mg For upgraded benefits in hypertension A Editorial This brochure, Clinical cases Coversyl 10 mg for upgraded benefits in hypertension, illustrates a variety of hypertensive
Clinical cases Coversyl 10 mg For upgraded benefits in hypertension A Editorial This brochure, Clinical cases Coversyl 10 mg for upgraded benefits in hypertension, illustrates a variety of hypertensive
Guideline-Directed Medical Therapy
 Guideline-Directed Medical Therapy Cardiovascular Outcomes Assessment of the MitraClip Percutaneous Therapy for Heart Failure Patients with Functional Mitral Regurgitation OPTIMAL THERAPY (As defined in
Guideline-Directed Medical Therapy Cardiovascular Outcomes Assessment of the MitraClip Percutaneous Therapy for Heart Failure Patients with Functional Mitral Regurgitation OPTIMAL THERAPY (As defined in
Antihypertensive Agents
 Antihypertensive Agents Southern California University of Health Sciences Physician Assistant Program Management and Treatment of Hypertension April 7, 08, presented by Ezra Levy, Pharm.D! Usual Dose,
Antihypertensive Agents Southern California University of Health Sciences Physician Assistant Program Management and Treatment of Hypertension April 7, 08, presented by Ezra Levy, Pharm.D! Usual Dose,
The first and only FDA-approved lisinopril oral solution for pediatric patients 6 years of age and older. WARNING: FETAL TOXICITY
 MEDICAS DIFFER FROM 5 drug products that are essentially copies of commercially available LIPRIL ORAL AF IMPORAN SAFEY INA WARNING: FEAL OXIC 5 drug products that are essentially copies of commercially
MEDICAS DIFFER FROM 5 drug products that are essentially copies of commercially available LIPRIL ORAL AF IMPORAN SAFEY INA WARNING: FEAL OXIC 5 drug products that are essentially copies of commercially
Optimal blockade of the Renin- Angiotensin-Aldosterone. in chronic heart failure
 Optimal blockade of the Renin- Angiotensin-Aldosterone Aldosterone- (RAA)-System in chronic heart failure Jan Östergren Department of Medicine Karolinska University Hospital Stockholm, Sweden Key Issues
Optimal blockade of the Renin- Angiotensin-Aldosterone Aldosterone- (RAA)-System in chronic heart failure Jan Östergren Department of Medicine Karolinska University Hospital Stockholm, Sweden Key Issues
Hello, and thank you for joining us for this presentation on novel approaches to understanding risks and treatment of hyperkalemia.
 Hello, and thank you for joining us for this presentation on novel approaches to understanding risks and treatment of hyperkalemia. PP-US-DSE-00032. 2015 Relypsa, Inc. All rights reserved. Relypsa and
Hello, and thank you for joining us for this presentation on novel approaches to understanding risks and treatment of hyperkalemia. PP-US-DSE-00032. 2015 Relypsa, Inc. All rights reserved. Relypsa and
LXIV: DRUGS: 4. RAS BLOCKADE
 LXIV: DRUGS: 4. RAS BLOCKADE ACE Inhibitors Components of RAS Actions of Angiotensin i II Indications for ACEIs Contraindications RAS blockade in hypertension RAS blockade in CAD RAS blockade in HF Limitations
LXIV: DRUGS: 4. RAS BLOCKADE ACE Inhibitors Components of RAS Actions of Angiotensin i II Indications for ACEIs Contraindications RAS blockade in hypertension RAS blockade in CAD RAS blockade in HF Limitations
The P&T Committee Lisinopril (Qbrelis )
 Situation Background Assessment The P&T Committee Lisinopril (Qbrelis ) Qbrelis, 1 mg/ml lisinopril oral solution, has recently become an FDA- approved formulation. Current practice at UK Chandler Medical
Situation Background Assessment The P&T Committee Lisinopril (Qbrelis ) Qbrelis, 1 mg/ml lisinopril oral solution, has recently become an FDA- approved formulation. Current practice at UK Chandler Medical
Lisinopril losartan conversion dose
 P ford residence southampton, ny Lisinopril losartan conversion dose Deep in-house technology, streaming expertise, and partnerships with the widest range of digital platforms secures our position as the
P ford residence southampton, ny Lisinopril losartan conversion dose Deep in-house technology, streaming expertise, and partnerships with the widest range of digital platforms secures our position as the
Florida Blue QUALITY PERFORMANCE METRIC STANDARDS FEBRUARY 2013
 Florida Blue QUALITY PERFORMANCE METRIC STANDARDS FEBRUARY 2013 QUALITY PERFORMANCE METRIC CALCULATION QUALITY METRICS SELECTED FOR MEASUREMENT Per Section 3.2 of the Agreement, HCPP must meet the following
Florida Blue QUALITY PERFORMANCE METRIC STANDARDS FEBRUARY 2013 QUALITY PERFORMANCE METRIC CALCULATION QUALITY METRICS SELECTED FOR MEASUREMENT Per Section 3.2 of the Agreement, HCPP must meet the following
Patient details GP details Specialist details Name GP Name Dr Specialist Name Dr R. Horton
 Rationale for Initiation, Continuation and Discontinuation (RICaD) Sacubitril/Valsartan (Entresto) For the treatment of symptomatic heart failure with reduced ejection fraction (NICE TA388) This document
Rationale for Initiation, Continuation and Discontinuation (RICaD) Sacubitril/Valsartan (Entresto) For the treatment of symptomatic heart failure with reduced ejection fraction (NICE TA388) This document
Hypertension (JNC-8)
 Hypertension (JNC-8) Southern California University of Health Sciences Physician Assistant Program Management and Treatment of Hypertension April 17, 2018, presented by Ezra Levy, Pharm.D.! The 8 th Joint
Hypertension (JNC-8) Southern California University of Health Sciences Physician Assistant Program Management and Treatment of Hypertension April 17, 2018, presented by Ezra Levy, Pharm.D.! The 8 th Joint
Target dose achievement of evidencebased medications in patients with heart failure with reduced ejection fraction attending a heart failure clinic
 Target dose achievement of evidencebased medications in patients with heart failure with reduced ejection fraction attending a heart failure clinic June Chen 1, Charlotte Galenza 1, Justin Ezekowitz 2,3,
Target dose achievement of evidencebased medications in patients with heart failure with reduced ejection fraction attending a heart failure clinic June Chen 1, Charlotte Galenza 1, Justin Ezekowitz 2,3,
The CARI Guidelines Caring for Australasians with Renal Impairment. Blood Pressure Control role of specific antihypertensives
 Blood Pressure Control role of specific antihypertensives Date written: May 2005 Final submission: October 2005 Author: Adrian Gillian GUIDELINES a. Regimens that include angiotensin-converting enzyme
Blood Pressure Control role of specific antihypertensives Date written: May 2005 Final submission: October 2005 Author: Adrian Gillian GUIDELINES a. Regimens that include angiotensin-converting enzyme
The role of angiotensin II receptor blockers in the management of heart failure
 European Heart Journal Supplements (2005) 7 (Supplement J), J10 J14 doi:10.1093/eurheartj/sui057 The role of angiotensin II receptor blockers in the management of heart failure John J.V. McMurray* Department
European Heart Journal Supplements (2005) 7 (Supplement J), J10 J14 doi:10.1093/eurheartj/sui057 The role of angiotensin II receptor blockers in the management of heart failure John J.V. McMurray* Department
Volume 6; Number 1 January 2012 NICE CLINICAL GUIDELINE 127: HYPERTENSION CLINICAL MANAGEMENT OF PRIMARY HYPERTENSION IN ADULTS (AUGUST 2011)
 Volume 6; Number 1 January 2012 NICE CLINICAL GUIDELINE 127: HYPERTENSION CLINICAL MANAGEMENT OF PRIMARY HYPERTENSION IN ADULTS (AUGUST 2011) What s new in hypertension? NICE has issued an updated Clinical
Volume 6; Number 1 January 2012 NICE CLINICAL GUIDELINE 127: HYPERTENSION CLINICAL MANAGEMENT OF PRIMARY HYPERTENSION IN ADULTS (AUGUST 2011) What s new in hypertension? NICE has issued an updated Clinical
THE RENIN-ANGIOTENSIN-ALDOSTERONE SYSTEM
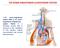 THE RENIN-ANGIOTENSIN-ALDOSTERONE SYSTEM The renin angiotensin system (RAS) or the renin angiotensin aldosterone system (RAAS) is a hormone system that is involved in the regulation of the plasma sodium
THE RENIN-ANGIOTENSIN-ALDOSTERONE SYSTEM The renin angiotensin system (RAS) or the renin angiotensin aldosterone system (RAAS) is a hormone system that is involved in the regulation of the plasma sodium
Combination of renin-angiotensinaldosterone. how to choose?
 Combination of renin-angiotensinaldosterone system inhibitors how to choose? Karl Swedberg Professor of Medicine Sahlgrenska Academy University of Gothenburg karl.swedberg@gu.se Disclosures Research grants
Combination of renin-angiotensinaldosterone system inhibitors how to choose? Karl Swedberg Professor of Medicine Sahlgrenska Academy University of Gothenburg karl.swedberg@gu.se Disclosures Research grants
Losartan lisinopril equivalent dose 新着 news 2018 年 3 月 6 日高等部 3 年生 大阪リゾート & スポーツ専門学校に合格! 2018 年 3 月 2 日茨木市立山手台小学校との交流.
 P ford residence southampton, ny Losartan lisinopril equivalent dose 新着 news 2018 年 3 月 6 日高等部 3 年生 大阪リゾート & スポーツ専門学校に合格! 2018 年 3 月 2 日茨木市立山手台小学校との交流. Losartan is classified as FDA pregnancy risk category
P ford residence southampton, ny Losartan lisinopril equivalent dose 新着 news 2018 年 3 月 6 日高等部 3 年生 大阪リゾート & スポーツ専門学校に合格! 2018 年 3 月 2 日茨木市立山手台小学校との交流. Losartan is classified as FDA pregnancy risk category
Class Update: Angiotensin-Converting Enzyme Inhibitors (ACEIs), Angiotensin II Receptor Blockers (ARBs), and Direct Renin Inhibitors (DRIs)
 Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-945-5220 Fax 503-947-1119 Class Update: Angiotensin-Converting Enzyme Inhibitors
Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-945-5220 Fax 503-947-1119 Class Update: Angiotensin-Converting Enzyme Inhibitors
Amlodipine/olmesartan (Azor ) is indicated for the treatment of hypertension, alone or in combination with other antihypertensive medications.
 Page 1 of 8 Policies Repository Policy Title Policy Number Oral Antihypertensive Agents FS.CLIN.9 Application of Pharmacy Policy is determined by benefits and contracts. Benefits may vary based on product
Page 1 of 8 Policies Repository Policy Title Policy Number Oral Antihypertensive Agents FS.CLIN.9 Application of Pharmacy Policy is determined by benefits and contracts. Benefits may vary based on product
À ². The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION. 19 September 2012
 À ² The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 19 September 2012 PROCORALAN 5 mg film-coated tablets B/56 (CIP code: 371 676-2) B/100 (CIP code: 567 208-1) PROCORALAN
À ² The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 19 September 2012 PROCORALAN 5 mg film-coated tablets B/56 (CIP code: 371 676-2) B/100 (CIP code: 567 208-1) PROCORALAN
AUSTRALIAN PI CANDESAN (CANDESARTAN CILEXETIL) TABLETS 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 AUSTRALIAN PI CANDESAN (CANDESARTAN CILEXETIL) TABLETS 1 NAME OF THE MEDICINE Candesartan cilexetil 2 QUALITATIVE AND QUANTITATIVE COMPOSITION CANDESAN tablets come in four strengths and contain 4 mg,
AUSTRALIAN PI CANDESAN (CANDESARTAN CILEXETIL) TABLETS 1 NAME OF THE MEDICINE Candesartan cilexetil 2 QUALITATIVE AND QUANTITATIVE COMPOSITION CANDESAN tablets come in four strengths and contain 4 mg,
Angiotensin Converting Enzyme inhibitor (ACEi) / Angiotensin Receptor Blocker (ARB) To STOP OR Not in Advanced Renal Disease
 Angiotensin Converting Enzyme inhibitor (ACEi) / Angiotensin Receptor Blocker (ARB) To STOP OR Not in Advanced Renal Disease Investigator Meeting 12 th September 2017 - Sheffield Prof Sunil Bhandari Consultant
Angiotensin Converting Enzyme inhibitor (ACEi) / Angiotensin Receptor Blocker (ARB) To STOP OR Not in Advanced Renal Disease Investigator Meeting 12 th September 2017 - Sheffield Prof Sunil Bhandari Consultant
Package leaflet: Information for the user. Angiosan 320 mg film-coated tablets. Valsartan
 Package leaflet: Information for the user Angiosan 40 mg film-coated tablets Angiosan 80 mg film-coated tablets Angiosan 160 mg film-coated tablets Angiosan 320 mg film-coated tablets Valsartan Read all
Package leaflet: Information for the user Angiosan 40 mg film-coated tablets Angiosan 80 mg film-coated tablets Angiosan 160 mg film-coated tablets Angiosan 320 mg film-coated tablets Valsartan Read all
Reducing proteinuria
 Date written: May 2005 Final submission: October 2005 Author: Adrian Gillin Reducing proteinuria GUIDELINES a. The beneficial effect of treatment regimens that include angiotensinconverting enzyme inhibitors
Date written: May 2005 Final submission: October 2005 Author: Adrian Gillin Reducing proteinuria GUIDELINES a. The beneficial effect of treatment regimens that include angiotensinconverting enzyme inhibitors
Heart Failure (HF) - Primary Care Flow Charts. Pre diagnosis Symptoms or signs suggestive of HF
 Heart Failure (HF) - Primary Care Flow Charts Pre diagnosis Symptoms or signs suggestive of HF 12 lead ECG Normal examination and 12 lead ECG HF highly unlikely Abnormal 12 lead ECG HF Possible Arrange
Heart Failure (HF) - Primary Care Flow Charts Pre diagnosis Symptoms or signs suggestive of HF 12 lead ECG Normal examination and 12 lead ECG HF highly unlikely Abnormal 12 lead ECG HF Possible Arrange
Heart Failure (HF) - Primary Care Flow Charts. Symptoms or signs suggestive of HF. Pre diagnosis. Refer to the Heart Failure Clinic at VHK for
 Heart Failure (HF) - Primary Care Flow Charts Pre diagnosis Symptoms or signs suggestive of HF 12 lead ECG Normal examination and 12 lead ECG HF highly unlikely Abnormal 12 lead ECG HF Possible Arrange
Heart Failure (HF) - Primary Care Flow Charts Pre diagnosis Symptoms or signs suggestive of HF 12 lead ECG Normal examination and 12 lead ECG HF highly unlikely Abnormal 12 lead ECG HF Possible Arrange
ACE inhibitors: still the gold standard?
 ACE inhibitors: still the gold standard? Session: Twenty-five years after CONSENSUS What have we learnt about the RAAS in heart failure? Lars Køber, MD, D.Sci Department of Cardiology Rigshospitalet University
ACE inhibitors: still the gold standard? Session: Twenty-five years after CONSENSUS What have we learnt about the RAAS in heart failure? Lars Køber, MD, D.Sci Department of Cardiology Rigshospitalet University
Since 1898, when renin was isolated from
 NARRATIVE REVIEW Dual Blockade of the Renin-Angiotensin System for Cardiorenal Protection: An Update Mustafa Arıcı, MD, and Yunus Erdem, MD The renin-angiotensin system (RAS) has an important role in hypertension
NARRATIVE REVIEW Dual Blockade of the Renin-Angiotensin System for Cardiorenal Protection: An Update Mustafa Arıcı, MD, and Yunus Erdem, MD The renin-angiotensin system (RAS) has an important role in hypertension
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: Olmesartan/Amlodipine (Azor), Olmesartan/Amlodipine/ HCTZ (Tribenzor) Reference Number: CP.CPA.XX Effective Date: 11.16.16 Last Review Date: 08.18 Line of Business: Commercial See Important
Clinical Policy: Olmesartan/Amlodipine (Azor), Olmesartan/Amlodipine/ HCTZ (Tribenzor) Reference Number: CP.CPA.XX Effective Date: 11.16.16 Last Review Date: 08.18 Line of Business: Commercial See Important
SUMMARY OF PRODUCT CHARACTERISTICS 2. QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Losartan potassium 25 mg Film-coated Tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each Losartan potassium 25 mg tablet contains
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Losartan potassium 25 mg Film-coated Tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each Losartan potassium 25 mg tablet contains
Don t let the pressure get to you:
 Balanced information for better care Don t let the pressure get to you: An update on the changing recommendations for treating hypertension New trial data and guidelines have made hypertension care more
Balanced information for better care Don t let the pressure get to you: An update on the changing recommendations for treating hypertension New trial data and guidelines have made hypertension care more
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Losartan Potassium 50 mg film-coated tablets. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each Losartan Potassium 50 mg Tablet contains
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Losartan Potassium 50 mg film-coated tablets. 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each Losartan Potassium 50 mg Tablet contains
Management of chronic heart failure: pharmacology. Giuseppe M.C. Rosano, MD, PhD, FHFA
 Management of chronic heart failure: pharmacology. Giuseppe M.C. Rosano, MD, PhD, FHFA Declaration of potential conflict of interests Type of job or financial support Salary Ordinary funds Position in
Management of chronic heart failure: pharmacology. Giuseppe M.C. Rosano, MD, PhD, FHFA Declaration of potential conflict of interests Type of job or financial support Salary Ordinary funds Position in
*NOTE: When reporting C P T code and 99239, it is recommended the measure be reported each time the code is submitted for hospital discharge.
 Measure #5 (NQF 0081): Heart Failure (HF): Angiotensin-Converting Enzyme (ACE) Inhibitor or Angiotensin Receptor Blocker (ARB) Therapy for Left Ventricular Systolic Dysfunction (LVSD) 2014 PQRS OPTIONS
Measure #5 (NQF 0081): Heart Failure (HF): Angiotensin-Converting Enzyme (ACE) Inhibitor or Angiotensin Receptor Blocker (ARB) Therapy for Left Ventricular Systolic Dysfunction (LVSD) 2014 PQRS OPTIONS
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Losartan Mylan 12.5 mg film-coated tablets Losartan Mylan 25 mg film-coated tablets Losartan Mylan 50 mg film-coated tablets Losartan
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Losartan Mylan 12.5 mg film-coated tablets Losartan Mylan 25 mg film-coated tablets Losartan Mylan 50 mg film-coated tablets Losartan
Congestive Heart Failure 2015
 Definition Congestive Heart Failure 215 JP Mehegan/ Mercy Cardiology n Cardiac failure; Congestive heart failure; Chronic heart failure (synonyms) n When the heart is unable to pump sufficiently and at
Definition Congestive Heart Failure 215 JP Mehegan/ Mercy Cardiology n Cardiac failure; Congestive heart failure; Chronic heart failure (synonyms) n When the heart is unable to pump sufficiently and at
COMPLEX HYPERTENSION. Anita Ralstin, FNP-BC Next Step Health Consultant, LLC
 COMPLEX HYPERTENSION Anita Ralstin, FNP-BC Next Step Health Consultant, LLC Incidence Of Hypertension About 70 million American adults have high blood pressure. About 33% of the population Only 52% have
COMPLEX HYPERTENSION Anita Ralstin, FNP-BC Next Step Health Consultant, LLC Incidence Of Hypertension About 70 million American adults have high blood pressure. About 33% of the population Only 52% have
ACE inhibitors vs ARBs Myths and Facts
 ACE inhibitors vs ARBs Myths and Facts Prof. Dr. med. Frank Ruschitzka, FRCP (Edinburgh) Director Heart Failure/Transplantation Clinic University Clinic Zurich Switzerland Conflict of interest: Bayer,
ACE inhibitors vs ARBs Myths and Facts Prof. Dr. med. Frank Ruschitzka, FRCP (Edinburgh) Director Heart Failure/Transplantation Clinic University Clinic Zurich Switzerland Conflict of interest: Bayer,
VI.2 Elements for a public summary
 VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology Product therapeutic indications: Myocardial infarction is the medical term for an event commonly known as a heart attack. It happens
VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology Product therapeutic indications: Myocardial infarction is the medical term for an event commonly known as a heart attack. It happens
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Telmisartan Teva 20 mg tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 20 mg telmisartan Excipients
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Telmisartan Teva 20 mg tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 20 mg telmisartan Excipients
*NOTE: When submitting CPT code and 99239, it is recommended the measure be submitted each time the code is submitted for hospital discharge.
 Quality ID #5 (NQF 0081): Heart Failure (HF): Angiotensin-Converting Enzyme (ACE) Inhibitor or Angiotensin Receptor Blocker (ARB) Therapy for Left Ventricular Systolic Dysfunction (LVSD) National Quality
Quality ID #5 (NQF 0081): Heart Failure (HF): Angiotensin-Converting Enzyme (ACE) Inhibitor or Angiotensin Receptor Blocker (ARB) Therapy for Left Ventricular Systolic Dysfunction (LVSD) National Quality
DISCLAIMER: ECHO Nevada emphasizes patient privacy and asks participants to not share ANY Protected Health Information during ECHO clinics.
 DISCLAIMER: Video will be taken at this clinic and potentially used in Project ECHO promotional materials. By attending this clinic, you consent to have your photo taken and allow Project ECHO to use this
DISCLAIMER: Video will be taken at this clinic and potentially used in Project ECHO promotional materials. By attending this clinic, you consent to have your photo taken and allow Project ECHO to use this
Public Assessment Report for paediatric studies submitted in accordance with Article 46 of Regulation (EC) No1901/2006, as amended
 Public Assessment Report for paediatric studies submitted in accordance with Article 46 of Regulation (EC) No1901/2006, as amended Diovan / Angiosan / Valsartan Novartis (valsartan) SE/W/0026/pdWS/001
Public Assessment Report for paediatric studies submitted in accordance with Article 46 of Regulation (EC) No1901/2006, as amended Diovan / Angiosan / Valsartan Novartis (valsartan) SE/W/0026/pdWS/001
Introductory Clinical Pharmacology Chapter 41 Antihypertensive Drugs
 Introductory Clinical Pharmacology Chapter 41 Antihypertensive Drugs Blood Pressure Normal = sys
Introductory Clinical Pharmacology Chapter 41 Antihypertensive Drugs Blood Pressure Normal = sys
Renal protection by inhibition of the renin-angiotensinaldosterone
 Renal protection by inhibition of the renin-angiotensinaldosterone system Tomas Berl Key words: angiotensinconverting enzyme inhibitor, angiotensin receptor blocker, combination therapy, direct renin inhibitor,
Renal protection by inhibition of the renin-angiotensinaldosterone system Tomas Berl Key words: angiotensinconverting enzyme inhibitor, angiotensin receptor blocker, combination therapy, direct renin inhibitor,
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Kinzalmono 20 mg tablets Kinzalmono 40 mg tablets Kinzalmono 80 mg tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Kinzalmono
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Kinzalmono 20 mg tablets Kinzalmono 40 mg tablets Kinzalmono 80 mg tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Kinzalmono
Executive Summary. Different antihypertensive drugs as first line therapy in patients with essential hypertension 1
 IQWiG Reports Commission No. A05-09 Different antihypertensive drugs as first line therapy in patients with essential hypertension 1 Executive Summary 1 Translation of the executive summary of the final
IQWiG Reports Commission No. A05-09 Different antihypertensive drugs as first line therapy in patients with essential hypertension 1 Executive Summary 1 Translation of the executive summary of the final
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Telmisartan Actavis 20 mg tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 20 mg telmisartan For the
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Telmisartan Actavis 20 mg tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 20 mg telmisartan For the
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Micardis 20 mg tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 20 mg telmisartan. Excipients with known
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Micardis 20 mg tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 20 mg telmisartan. Excipients with known
