Macular edema (ME) results from a complex
|
|
|
- Sophie Lawrence
- 5 years ago
- Views:
Transcription
1 DIFFERENTIATING STEROID DELIVERY SYSTEMS FOR MACULAR EDEMA * Baruch D. Kuppermann, MD, PhD ABSTRACT Steroids can intervene with many of the processes that lead to macular edema (ME). Steroids are potent anti-inflammatory agents, but they also have anti-vascular endothelial growth factor activity. Triamcinolone acetonide is probably the most commonly used steroid for ME, but it may not be the ideal formulation for ME. This article discusses several new steroid delivery systems: the fluocinolone acetonide implants and dexamethasone intravitreal implant. Drug-delivery systems under investigation have 2 fundamental approaches and philosophies: longer-acting reservoir implants with good long-term control of disease but with potential for drug or suppressive side effects, and shorter-acting, biodegradable inserts that potentially expose the eye to less drug or suppressive side effects but may not control disease as well. Sustained-release ocular implants lead to drug concentration gradients in eye tissues in which drug concentration is highest near the implant. Models of drug gradients based on implant location suggest that the implant can be shifted further into the eye, perhaps partitioning the drug better in the posterior segment and minimizing the anterior segment exposure. (Adv Stud Ophthalmol. 2010;7(2):35-41) *Based on a presentation by Dr Kuppermann at a roundtable symposium held in Fort Lauderdale, Florida, on April 30, Professor of Ophthalmology and Biomedical Engineering, Gavin Herbert Eye Institute, University of California, Irvine, Irvine, California. Address correspondence to: Baruch D. Kuppermann, MD, PhD, Professor of Ophthalmology and Biomedical Engineering, Gavin Herbert Eye Institute, University of California, Irvine, MS 1 125, Irvine, CA bdkupper@uci.edu. Macular edema (ME) results from a complex pathway of events, originating from vascular disease (diabetes or central/branch retinal vein occlusion) or as primary inflammatory disease (uveitis). ME due to vascular disease is strongly associated with vascular endothelial growth factor (VEGF) levels, whereas ME due to uveitis is due primarily to inflammatory mediators, such as interleukin-1 and tumor necrosis factor-α. Steroids can intervene with many of the processes in both pathways that lead to ME. Steroids are potent anti-inflammatory agents, but they also have anti-vegf activity. DIFFERENTIATING STEROIDS Triamcinolone acetonide (TA) is probably the most commonly used steroid for ME, but is it the ideal formulation for ME? Corticosteroids are based on the cortisol structure, but they have several properties beyond chemical structure by which they can be distinguished, including anti-inflammatory potency, ability to translocate the glucocorticoid receptor complex to the nucleus, ability to transactivate or transrepress ligand-dependent gene sets and biologic responses, neuroprotection of the photoreceptors/retinal pigment epithelium, and direct cytotoxic effects. 1-6 The Table shows the relative potencies of corticosteroids, and clearly TA is not the most potent. 7 Although TA is used most consistently because of its formulation, drug-delivery systems are being developed that offer better choices for treating ME. Another important way to distinguish among steroids is by their side effects. For example, in the Diabetes Clinical Research Network (DRCR) Protocol B study, 1- and 4-mg TA (Trivaris; Allergan, Inc, Irvine, CA; a micronized and lyophylized injectable suspension formulation) was compared to focal/grid Johns Hopkins Advanced Studies in Ophthalmology 35
2 Table. Relative Potencies of Corticosteroids Corticosteroid Cortisone 0.8 Cortisol 1 Prednisone 4 Methylprednisolone 5 Triamcinolone 5 Betamethasone 25 Dexamethasone 25 Fluocinolone acetonide 25 Relative Potencies Adapted with permission from Hardman et al, eds. Goodman and Gilman's The Pharmacological Basis of Therapeutics. 10th ed. New York, NY: McGraw-Hill; photocoagulation in patients with diabetic ME (DME). The results showed important side effects with TA: glaucoma and cataracts. Intraocular pressure (IOP) increased from baseline by 10 mm Hg or more at any visit in 4%, 16%, and 33% of eyes in the 3 treatment groups, respectively, and cataract surgery was performed in 13%, 23%, and 51% of eyes in the 3 treatment groups, respectively. 8 Other steroids and steroid drug-delivery systems may show different incidences of cataract and glaucoma, which are the key ocular side effects associated with intraocular steroid use. Patient with Poor Visual Acuity and Diffuse DME in Both Eyes Harry W. Flynn, Jr, MD JO is a 43-year-old man with a 19-year history of non insulin-dependent diabetes. He is pseudophakic in both eyes and had both focal/grid laser and pan-retinal photocoagulation in both eyes. Visual acuity is 20/200 OD and 20/400 OS. Fundus photograph of the right eye reveals hard exudates in the center of the macula and a large circinate ring. Optical coherence tomography (OCT) imaging (right eye) reveals diffuse retinal thickening (>500 µm), with slight vitreoretinal interface abnormality or cystic changes. How would you treat his left eye? How would you treat his right eye? How would you manage this patient if your first-line therapy was not effective? For further discussion of the challenges in managing this real-life situation, please go to NEW STEROID DELIVERY SYSTEMS Triamcinolone acetonide is discussed in more detail elsewhere in this monograph. This article discusses new drug-delivery systems under development, that are either US Food and Drug Administration (FDA) approved or pending approval. FLUOCINOLONE The fluocinolone acetonide-containing sustainedrelease drug-delivery device (SR-DDD; Retisert; Bausch & Lomb, Rochester, NY) is surgically implanted through a 3.5-mm pars plana incision and delivers 0.59 mg of steroid total for up to 36 months. Fluocinolone is 25 times more potent than cortisol (Table), and its use in a chronic drug-delivery implant allows the usage of very low doses, which have been shown in clinical trials to be therapeutic. Currently, it is only FDA approved for uveitis. A phase III clinical trial in patients with DME is fully enrolled but approval has not yet been sought due to concerns about steroid-induced ocular side effects, particularly the high incidence of glaucoma, despite positive changes in visual acuity in some patients. After 3 years, 27.6% of patients who received the implant achieved at least 3 lines of improvement in visual acuity compared with 14.5% who received physician-determined standard of care (typically laser treatment). However, 18.9% and 15.9%, respectively, also lost at least 3 lines after 3 years of treatment. Safety data from the prescribing information show that eyes that receive the implant require significant monitoring. After 3 years, approximately 77% of those who received the implant required topical IOPlowering drops, and 37% required filtering operations. The formation of cataracts after implant is also nearly ubiquitous in patients with DME (over 90% after 3 years, compared with 15% in the fellow eye). Cataract formation rises dramatically after approximately 18 months with the implant. 9 Another fluocinolone acetonide-containing implant, which is an injectable SR-DDD (Iluvien; Alimera Sciences, Inc, Alpharetta, Georgia), uses a non-bioerodible polyimide tube (3.5 mm long, 0.37 mm diameter) to deliver the corticosteroid (0.2 µg or 0.5 µg daily over 36 or 18 months, respectively). Both injectable fluocinolone SR-DDD implants studied in the clinical trial contain approximately 50% the amount of fluocinolone as the surgically implanted 36 Vol. 7, No. 2 December 2010
3 SR-DDD. A 25-gauge inserter allows for a self-sealing wound, so the procedure can be done in multiple settings (ie, office, ambulatory surgery center, or hospital). The implant floats freely in the vitreous cavity, but tends to settle in the inferior vitreous base. The rationale for this product was to hopefully minimize ocular side effects such as cataract and glaucoma by further lowering the dose and to move the implant away from the eye wall and more into the inferior vitreous base. The bioerodible fluocinolone implant has been studied in 956 patients with DME in 2 multicenter, international, randomized, double-masked, dosefinding, controlled trials (same protocol) comparing the 2 implant doses with standard of care (laser therapy). Although the data are not yet published, initial analyses of the pooled data show that 28.7% and 28.6% (low- and high-dose implant) gained at least 15 Early Treatment Diabetic Retinopathy Study letters at month 24, compared with 16.2% of those receiving standard of care (P =.002 for both treatment groups vs control). The differences with implant compared to standard of care were observed as early as month 1. Similarly, reduction in excess foveal thickness (measured by OCT) was seen by month 1 and remained dramatically different between low-dose implant and control throughout the study. The rates of IOP increase were much lower than with the surgically implanted fluocinolone SR- DDD, with 16.3% and 21.6% of the low- and highdose groups having IOP greater than 30 mm Hg, compared with 2.7% in the control group. Additionally, rates of glaucoma surgery were much lower than with the surgically implanted fluocinolone SR-DDD, with 3.5% and 7.4% of the lowand high-dose groups, respectively, requiring 1 or more IOP-lowering surgeries, compared to 0.5% of the control group. However, cataract rates were very high similar (though slightly lower) than what was found with the surgically implanted fluocinolone SR-DDD. Cataract was reported in month 24 in eyes that were phakic at baseline in 80% and 87% of the low- and high-dose groups, respectively, compared with 43.6% of the control group. Cataract surgery by month 24 in eyes that were phakic at baseline was required in 74.9%, 84%, and 23.1% of patients, respectively. Thus, cataract again will be nearly ubiquitous in patients who receive this implant, a risk that needs to be discussed with the patient. Glaucoma appears to be more manageable. DEXAMETHASONE A dexamethasone intravitreal implant (Ozurdex; Allergan, Inc, Irvine, CA) has been approved by the FDA for the treatment of ME caused by retinal vein occlusion. In this implant, the drug is embedded into a biodegradable polymer (polylactic glycolic acid). As the polymer degrades, the drug elutes over time and the remaining polymer subsequently breaks down to carbon dioxide and water. (By contrast, with the bioerodible injectable fluocinolone SR-DDD, there is a residual husk of polymer permanently left behind in the eye.) This implant was studied in 2 doses (350 and 700 µg) and was implanted using a 20-gauge incision in the phase II trial, though in phase III and in the approved product, it is injected into the eye through a 22-gauge injector. A phase II study in patients with ME caused by a variety of diseases (ie, diabetes, retinal vein occlusion, uveitis, and Irvine-Gass syndrome post cataract surgery) showed significant vision improvement through 180 days with the high dose (Figure 1). 10 Patients with ME caused by uveitis or Irvine-Gass syndrome experienced particular benefit, but significant improvement in visual acuity was observed across all patient subtypes who received the 700-µg dexamethasone implant (Figure 2). 10 Increased IOP was observed in some patients (17% and 12.9% in the 350- and 700-µg groups, respectively, compared with 0% in the observation group), as was vitreous hemorrhage (21%, 21.8%, and 0%, respectively), but cataract was not (15%, 17.8%, and 12.4%, respectively). 10 (Please note: The phase III data leading to its approval are discussed in Dr Kuppermann s second article.) EMERGING CONCEPTS IN VITREAL DRUG DELIVERY It is important to realize that sustained-release ocular implants may lead to drug concentration gradients in eye tissues, in which drug concentration is highest near the implant, for at least moderate periods of time. Models of drug gradients based on implant location show that, for example, a fluocinolone implant sutured to the eye wall has low but measurable aqueous levels ( µg/ml) and higher macular levels ( µg/g). By simply moving the implant by 4 mm, perhaps similar to the placement with the dexamethasone intravitreal implant or injectable fluocinolone SR-DDD, the aqueous levels of drug are no longer measurable and the macular levels of drug are Johns Hopkins Advanced Studies in Ophthalmology 37
4 Figure 1. Improvements in Visual Acuity: Results from the PME Dexamethasone Intravitreal Implant Trial Patients, % Figure 2. Visual Acuity Improvements Across Subgroups (Day 90): Results from the Dexamethasone Intravitreal Implant Trial Patients with at least 10-letter improvement, % P < P values are 700 µg vs observation. Patients in the 700-µg treatment group received no rescue therapy between day 90 and 180. DDS = drug-delivery system; ETDRS = Early Treatment Diabetic Retinopathy Study; PME = persistent macular edema. Reprinted with permission from Kuppermann et al. Arch Ophthalmol. 2007;125: P = All Patie nts DM E RVO Uv e itis/irv ine - Gass Posurdex (Ozurdex [dexamethasone intravitreal implant]; Allergan, Inc, Irvine, CA) Phase II Surgical Implantation Study. DME = diabetic macular edema; RVO = retinal vein occlusion. Data from Kuppermann et al. Arch Ophthalmol. 2007;125: P = P = Day 90 Day 180 Day 90 Day 180 Gained >10 ETDRS letters Observation (n = 105) Dexamethasone -DDS 350 µg (n = 103) Dexamethasone -DDS 700 µg (n = 105) Gained >15 ETDRS letters fold greater ( µg/g). If there are drug gradients in vivo, we can take advantage of that and shift the implant further into the eye, and perhaps partition the drug better in the posterior segment and minimize the anterior segment exposure. Also, the pharmacokinetics of freely injected intravitreal drugs are altered dramatically based on whether the eye is vitrectomized. However, pharmacokinetic studies of these emerging drug-delivery systems show no difference between vitrectomized and nonvitrectomized eyes in terms of retinal drug concentrations. 11 Therefore, no longer does the vitreous act as a reservoir; rather, the implant is the reservoir. CONCLUSIONS Steroids are uniquely positioned to address the cascade of events leading to ME. Different steroids have varying potencies and toxicities. There are several ways to distinguish among the steroids used in ophthalmology, including chemical structure, anti-inflammatory potency, ability to translocate the glucocorticoid receptor complex to the nucleus, ability to transactivate or transrepress ligand-dependent gene sets and biologic responses, neuroprotection of the photoreceptors/retinal pigment epithelium, and direct cytotoxic effects. These differences may help to explain the differences among steroids in their safety and efficacy for the treatment of retinal disease. Applying our current understanding of glucocorticoid pharmacology to drug discovery may lead to the development of ocular hypertension- and cataract-free steroids. Drug-delivery systems under investigation have 2 fundamental approaches and philosophies: longer-acting reservoir implants with good long-term control of disease but with potential for drug or suppressive side effects, and shorter-acting, biodegradable inserts that potentially expose the eye to less drug or suppressive side effects but may not control disease as well. DISCUSSION STEROIDS IN VITRECTOMIZED EYES Dr Flynn: I often use steroids in vitrectomized eyes because steroid crystals have a longer effect in eyes with no formed vitreous compared to the rapid clearing with anti-vegf agents. These crystals may adhere to the surface of the retina but there appears to be no toxic effect. 38 Vol. 7, No. 2 December 2010
5 Dr Kuppermann: I also like to use TA post-vitrectomy, but because I am concerned about direct toxic effects of TA laying on the surface of the retina in vitrecomized eyes, I prefer using a TA sub-tenon s injection at the end of the case for the same purpose and I found reasonable effects from that. Thus, there are still ways to use steroids and try to mitigate some of the potential for steroid toxicity. TA FORMULATIONS Dr Flynn: The standard injectable solution TA (Kenalog; Bristol-Myers Squibb, Princeton, NJ), which includes preservatives, was used mainly in 2004 and We became concerned about toxicity of preservatives, and shifted toward using preservativefree compounded pharmacy-formulated preparation, which became very popular. Another brand of TA (Triescence; Alcon, Inc., Fort Worth, TX), which is an injectable suspension formulation, was more of a dispersive agent and was used often in surgery to help identify the vitreous. This product is popular among many surgeons. There is also the hydrogel-based injectable suspension formulation (Trivaris; Allergan, Inc, Irvine, CA), which was used in clinical trials such as DRCR and SCORE (Standard Care vs Corticosteroid for Retinal Vein Occlusion). The advantage of this formulation is that it forms a cohesive bolus of drug in the inferior formed vitreous and it is visible for months after the injection. Therefore we can choose from 3 different formulations of TA injectable. I think the retinal specialists generally have drifted away from the use of the standard TA injectable solution. I think it is relatively rarely used nowadays even though it is relatively inexpensive. Dr Nguyen: Do you choose different steroids based on different types of patients? Dr Campochiaro: Until we get genetic data, there are some clinical features that we can use to tailor treatment. One is, of course, we tend to exclude any patient who has underlying glaucoma from the use of any steroids including steroid inserts. In addition, patients who have a family history of glaucoma may be more likely to have a steroid response. We do not know whether it is possible to identify patients by a stress test. We know that if a patient responds with increased IOP to topical steroids, that he or she is more likely after an implant to have increased IOP. So, certainly, if a patient has any history of a steroid response after topical steroids or any type of a steroid administration, that would also make the patient less suitable as a candidate for steroid implant. Dr Davis: The stress test idea is useful when it is positive, but changing any of 3 factors dose, route of administration, and duration of exposure might change a patient s response. In the trials with the implantable device, patients were enrolled who had been exposed previously to regional corticosteroids and were not considered steroid responders, but some of them did have elevation of IOP from the device presumably related to the long duration of exposure with the device. Without genetic data, it is going to be difficult to know if someone is a responder, and even if they are genetically predisposed, dose, route, and exposure might still make a difference. In a situation where you have perhaps no other equally effective therapy, is the increase in IOP necessarily a contraindication for use of the agent? We can assess on an individual basis the risks and benefits of remaining untreated with a potentially extremely useful agent versus potentially requiring glaucoma surgery. Central acuity can be lost forever from cystoid ME, whereas glaucoma surgery is generally considered a manageable risk and a reasonably predictable surgery not a perfect surgery but one that can be offered as part of the management of a patient. Dr Campochiaro: It is unlikely that genetic testing or stress testing will identify 100% of patients who are steroid responders, but it would still be useful to help rule out a certain percentage of patients understanding that such things are only tools and do not guarantee that a patient will not experience increased IOP from injection of a steroid insert. Certainly the duration of steroid exposure is very important in terms of developing glaucoma. Dr Kuppermann: Most of us have migrated away from using the standard TA injectable solution because of the black box warning on the label and the fact that there is another formulation (TA injectable suspension [Alcon, Inc.]) that has an ocular indication. Dr Campochiaro: I still use TA injectable solution. I have not had many problems with it. It is certainly much less expensive, and it is more easily available than TA injectable suspension (Alcon, Inc). Has anybody seen any problems with this injectable suspension formulation when used for the treatment of ME? Several cases have been referred to me of patients who have had extreme dispersion of this formulation. It may not be as suitable as TA injectable solution, which tends to form a clump and get out of visual axes. Johns Hopkins Advanced Studies in Ophthalmology 39
6 Dr Kuppermann: Most of us shifted away from TA injectable solution approximately 3 or 4 years ago because there was a cluster of cases of sterile endophthalmitis. Thus, I started using a compounding pharmacy preservative-free formulation of TA. With the TA injectable solution, it was always visible after 1 month, frequently visible after 2 months, and sometimes visible after 3 months. Once I switched over to the compounding pharmacy formulation, most of the time you do not see it at all after 1 month. TA injectable suspension (Alcon, Inc.) is somewhere in between, meaning it is usually visible at 1 month but it does not seem to have the visual visible durability of TA injectable solution. Candidly, I am thinking of shifting back to TA injectable solution as well, because I believe it has the longest duration in the eye of all the TA formulations. Dr Flynn: TA injectable suspension (Alcon, Inc.) is always dispersive, especially in vitrectomized eyes. In anticipation of that event, I warn patients that for approximately 1 week, they will have a snowstorm with the drug dispersed in the vitreous cavity which definitely reduces their vision for a few days. This can even be significant when it disperses into a liquefied vitreous. TA injectable solution also can be dispersive but more often in large clumps. Dr Davis: I no longer use TA injectable solution for intravitreal injection. With 2 FDA-approved products for ocular inflammatory disease, I see no reason to use it. The TA injectable suspension formulated by Allergan, Inc is not commercially available, but it has an FDA label for ocular use. The TA injectable suspension formulated by Alcon, Inc. is available. One situation in which I have used TA injectable solution is for vitreous visualization, because the crystal size is different from the injectable suspension formulation (Alcon, Inc). In some cases you get better visualization of the vitreous. Because it is rinsed from the eye at the end of the vitrectomy, I am less concerned about the toxic effects that might occur from the vehicle and preservatives. Dr Kuppermann: Which is ironic because the primary label indication of that injectable suspension formulation is for visualization of the vitreous. You are saying TA injectable solution is better for that. Dr Do: Right now, I still use TA for some cases of ME. However, like most ophthalmologists, I am concerned about the risk of elevated IOP associated with TA. I will be curious to see if the decreased risk of elevated IOP with dexamethasone intravitreal implant holds up in the long term because the short-term studies have suggested there is less IOP elevation in dexamethasone-based SR-DDDs. Interestingly, in the injectable fluocinolone SR-DDD studies, there was not a bigger benefit with the higher dose than with the low dose, which was surprising because usually you see a greater reduction in retinal thickness or a greater improvement in visual acuity along with the higher dose of steroids. But the FAME (Fluocinolone Acetonide in DME) studies show that the dose and clinical response do not always correlate. In some steroid formulations, the lower dose may have better efficacy and decreased adverse events than the higher dose. Dr Kuppermann: That is a very good point, particularly with the drug-delivery systems, because with chronic drug delivery you are saturating the receptors even with low doses. The ranibizumab intravitreal injection (Lucentis; Genentech, Inc, South San Francisco, CA) dosing is partially defined so that there will be some residual drug at 28 days at therapeutic levels. We are massively overtreating all our patients with intravitreal injections, in the sense that immediately after an intravitreal injection the dose is much higher than needed, but we are more concerned with extending the therapeutic window at the tail end of the treatment window. Once you saturate the glucocorticoid or VEGF receptors, you are already getting all the benefit you need out of that and you can minimize some of the collateral effects with the chronic low dosing of drug-delivery systems. Dr Schmidt-Erfurth: We talk so much about different drug formulations that I think that we have forgotten that the real indication for steroid treatment is for inflammation. We have since learned that steroids may have some effect in other diseases such as vascular macular disease. But now that we have a whole spectrum of different types of drugs available, should we not use steroids for diseases that are inflammation-driven and try to find the best formulation with the longest durability and the lowest rate of side effects? And then for other diseases which are not primarily inflammatory, but for which we see that, for instance, anti-vegf compounds have a good effect, try to find the lowest dose with the best efficacy and lowest side effects for those? Dr Campochiaro: TA injectable suspension (Alcon, Inc.) is approved for use during surgery to 40 Vol. 7, No. 2 December 2010
7 visualize the vitreous. That should not make us feel any better about injecting it for ME; it has not been studied for that. It is not proven to be any safer than TA injectable solution. This whole issue about preservatives being the cause of sterile endophthalmitis seems to be incorrect. It appears to be more a matter of the crystals because preservative-free TA does it, the TA injectable suspension formulation does it, anything that has particulate matter does it. The difference is that some patients just respond to particulate matter in their vitreous differently such that they get a bigger inflammatory response. Thus I do not think we should leave the impression that there is any particular steroid preparation that has been well studied for use in ME in terms of the free steroids and that there is no reason why TA injectable suspension is any better than TA injectable solution. Dr Kuppermann: TA injectable suspension (Allergan, Inc) was studied in clinical trials. It is not commercially available, and because the hydrogel sequesters the crystals, it has not been associated with sterile endophthalmitis. But, because it is micronized, lyophilized, and has a small particle size, it may not have the durability and effect that we see with the TA injectable solution, which has the largest particle size of all the TA formulations. Dr Flynn: I think there was an overreaction to blaming the preservatives in TA injectable solution. We now have large trials (DRCR Network Protocols B and I) showing similar low rates of endophthalmitis with intravitreal steroids, particularly in patients with diabetes, when you would expect higher rates. We know now that inflammation is part of the mechanism for diabetic retinopathy, which is not just a retinal vascular disease. Inflammation is important. Dr Kuppermann: I am not sure that the separation of inflammatory diseases from VEGF-mediated diseases is quite true because we have seen that there is evidence that inflammation is part of all of these diseases, therefore that there may be a partial blurring of the differences between the pathophysiology of the various retinal diseases of interest. REFERENCES 1. Schaaf MJ, Lewis-Tuffin LJ, Cidlowski JA. Ligand-selective targeting of the glucocorticoid receptor to nuclear subdomains is associated with decreased receptor mobility. Mol Endocrinol. 2005;19: Parver LM, Auker CR, Fine BS, Doyle T. Dexamethasone protection against photochemical retina injury. Arch Ophthalmol. 1984;102: Reme CE, Grimm C, Hafezi F, et al. Why study rod cell death in retinal degenerations and how? Doc Ophthalmol. 2003;106: Wenzel A, Grimm C, Samardzija M, Reme CE. The genetic modifier Rpe65Leu(450): effect on light damage susceptibility in c-fos-deficient mice. Invest Ophthalmol Vis Sci. 2003;44: Wenzel A, Grimm C, Seeliger MW, et al. Prevention of photoreceptor apoptosis by activation of the glucocorticoid receptor. Invest Ophthalmol Vis Sci. 2001;42: Smit-McBride Z, Morse LS, Cessna C, et al. Global gene profiling of early and late induced changes in gene expression upon intravitreal injection of triamcinolone and dexamethasone in the mouse retina. Poster presented at: Annual Meeting of the Association for Research in Vision and Ophthalmology; May 3-7, 2009; Ft. Lauderdale, FL. Abstract/poster# 494/D Hardman JG, Limbird LE, Gilman AG, eds. Goodman and Gilman's The Pharmacological Basis of Therapeutics. 10th ed. New York, NY: McGraw-Hill; Diabetic Retinopathy Clinical Research Network. A randomized trial comparing intravitreal triamcinolone acetonide and focal/grid photocoagulation for diabetic macular edema. Ophthalmology. 2008;115: Fluocinolone acetonide intravitreal implant [prescribing information]. Rochester, NY: Bausch & Lomb; Kuppermann BD, Blumenkranz MS, Haller JS, et al. Randomized, controlled study of a novel intravitreous dexamethasone drug delivery system in patients with persistent macular edema. Arch Ophthalmol. 2007;125: Kuppermann BD, Chang-LIN JE, Burke JA, et al. Pharmacokinetics of dexamethasone after intravitreal implantation of 700 µg dexamethasone posterior segment drug delivery system in vitrectomized versus nonvitrectomized eyes. Presented at: 7th EURETINA Congress; May 17-20, 2007; Monte Carlo, Monaco. Johns Hopkins Advanced Studies in Ophthalmology 41
Macular edema (ME) is the most common
 MANAGEMENT OF RETINAL VEIN OCCLUSIONS * Peter A. Campochiaro, MD ABSTRACT Macular edema (ME) is the most common cause of reduced vision in patients with retinal vein occlusions (RVOs). The primary cause
MANAGEMENT OF RETINAL VEIN OCCLUSIONS * Peter A. Campochiaro, MD ABSTRACT Macular edema (ME) is the most common cause of reduced vision in patients with retinal vein occlusions (RVOs). The primary cause
ILUVIEN IN DIABETIC MACULAR ODEMA
 1 ILUVIEN IN DIABETIC MACULAR ODEMA Marie Tsaloumas Consultant Ophthalmic Surgeon Queen Elizabeth Hospital, Birmingham bars conference 2104 1 2 Declaration of interest I have sat on Advisory boards for
1 ILUVIEN IN DIABETIC MACULAR ODEMA Marie Tsaloumas Consultant Ophthalmic Surgeon Queen Elizabeth Hospital, Birmingham bars conference 2104 1 2 Declaration of interest I have sat on Advisory boards for
Abbreviated Drug Evaluation: Fluocinolone acetonide intravitreal implant (Retisert )
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35, Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
FEP Medical Policy Manual
 FEP Medical Policy Manual Last Review: September 20 Next Review: September 2017 Related Policies 9.03.21 Aqueous Shunts for Glaucoma Intravitreal Corticosteroid Implants Summary An intravitreal implant
FEP Medical Policy Manual Last Review: September 20 Next Review: September 2017 Related Policies 9.03.21 Aqueous Shunts for Glaucoma Intravitreal Corticosteroid Implants Summary An intravitreal implant
PRECISION PROGRAM. Injection Technique Quick-Reference Guide. Companion booklet for the Video Guide to Injection Technique
 Injection Technique Quick-Reference Guide PRECISION PROGRAM Companion booklet for the Video Guide to Injection Technique Available at www.ozurdexprecisionprogram.com Provides step-by-step directions with
Injection Technique Quick-Reference Guide PRECISION PROGRAM Companion booklet for the Video Guide to Injection Technique Available at www.ozurdexprecisionprogram.com Provides step-by-step directions with
Long-Term Follow-Up of Patient with Diabetic Macular Edema Receiving Fluocinolone Acetonide Intravitreal Implant
 Ophthalmol Ther (2015) 4:51 58 DOI 10.1007/s40123-015-0028-0 CASE REPORT Long-Term Follow-Up of Patient with Diabetic Macular Edema Receiving Fluocinolone Acetonide Intravitreal Implant Thomas Bertelmann
Ophthalmol Ther (2015) 4:51 58 DOI 10.1007/s40123-015-0028-0 CASE REPORT Long-Term Follow-Up of Patient with Diabetic Macular Edema Receiving Fluocinolone Acetonide Intravitreal Implant Thomas Bertelmann
Intravitreal Corticosteroid Implants. Description
 Subject: Intravitreal Corticosteroid Implants Page: 1 of 20 Last Review Status/Date: June 2015 Intravitreal Corticosteroid Implants Description An intravitreal implant is a drug delivery system, injected
Subject: Intravitreal Corticosteroid Implants Page: 1 of 20 Last Review Status/Date: June 2015 Intravitreal Corticosteroid Implants Description An intravitreal implant is a drug delivery system, injected
Intravitreal Corticosteroid Implants
 Intravitreal Corticosteroid Implants Policy Number: 9.03.23 Last Review: 4/2018 Origination: 07/2015 Next Review: 4/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage
Intravitreal Corticosteroid Implants Policy Number: 9.03.23 Last Review: 4/2018 Origination: 07/2015 Next Review: 4/2019 Policy Blue Cross and Blue Shield of Kansas City (Blue KC) will provide coverage
Diabetic maculopathy 11/ An update on. Miss Vasuki Sivagnanavel
 Miss Vasuki Sivagnanavel Consultant Ophthalmologist An update on Diabetic maculopathy Despite advances in the management of diabetes, diabetic retinopathy is already the commonest cause of blindness among
Miss Vasuki Sivagnanavel Consultant Ophthalmologist An update on Diabetic maculopathy Despite advances in the management of diabetes, diabetic retinopathy is already the commonest cause of blindness among
Intravitreal Corticosteroids in the Management of Diabetic Macular Edema
 Curr Ophthalmol Rep (2013) 1:144 149 DOI 10.1007/s40135-013-0015-3 DIABETIC RETINOPATHY: MEDICAL AND SURGICAL THERAPIES (PK KAISER, SECTION EDITOR) Intravitreal Corticosteroids in the Management of Diabetic
Curr Ophthalmol Rep (2013) 1:144 149 DOI 10.1007/s40135-013-0015-3 DIABETIC RETINOPATHY: MEDICAL AND SURGICAL THERAPIES (PK KAISER, SECTION EDITOR) Intravitreal Corticosteroids in the Management of Diabetic
FLUOCINOLONE ACETONIDE: STEROID LONG ACTING
 FLUOCINOLONE ACETONIDE: STEROID LONG ACTING Giuseppe Querques, MD PhD Department of Ophthalmology, IRCCS Ospedale San Raffaele, University Vita Salute San Raffaele, Milan, Italy Financial Disclosure ADVISORY
FLUOCINOLONE ACETONIDE: STEROID LONG ACTING Giuseppe Querques, MD PhD Department of Ophthalmology, IRCCS Ospedale San Raffaele, University Vita Salute San Raffaele, Milan, Italy Financial Disclosure ADVISORY
Clinical Trials in Diabetic Retinopathy. Harry W. Flynn Jr., M.D. Nidhi Relhan Batra, M.D.
 1 Clinical Trials in Diabetic Retinopathy 2018 Harry W. Flynn Jr., M.D. Nidhi Relhan Batra, M.D. Bascom Palmer Eye Institute 900 N.W. 17th Street Miami, FL 33136 Phone: (305) 326-6118 Fax: (305) 326-6417
1 Clinical Trials in Diabetic Retinopathy 2018 Harry W. Flynn Jr., M.D. Nidhi Relhan Batra, M.D. Bascom Palmer Eye Institute 900 N.W. 17th Street Miami, FL 33136 Phone: (305) 326-6118 Fax: (305) 326-6417
Diabetic Retinopathy: Managing the Extremes. J. Michael Jumper, MD West Coast Retina
 Diabetic Retinopathy: Managing the Extremes J. Michael Jumper, MD West Coast Retina Case 1: EC 65 y.o. HM No vision complaints Meds: Glyburide Metformin Pioglitazone Va: 20/20 OU 20/20 Case 2: HS 68 y.o.
Diabetic Retinopathy: Managing the Extremes J. Michael Jumper, MD West Coast Retina Case 1: EC 65 y.o. HM No vision complaints Meds: Glyburide Metformin Pioglitazone Va: 20/20 OU 20/20 Case 2: HS 68 y.o.
Sustained-Release Corticosteroid Options
 Sustained-Release Corticosteroid Options Mariana Cabrera, University of Miami Miller School of Medicine Steven Yeh, Emory University Thomas A Albini, University of Miami Miller School of Medicine Journal
Sustained-Release Corticosteroid Options Mariana Cabrera, University of Miami Miller School of Medicine Steven Yeh, Emory University Thomas A Albini, University of Miami Miller School of Medicine Journal
Medical Coverage Policy Intravitreal Corticosteroid Implants)
 Medical Coverage Policy Intravitreal Corticosteroid Implants) EFFECTIVE DATE:10 01 2015 POLICY LAST UPDATED: 02 07 2017 OVERVIEW An intravitreal implant is a drug delivery system, injected or surgically
Medical Coverage Policy Intravitreal Corticosteroid Implants) EFFECTIVE DATE:10 01 2015 POLICY LAST UPDATED: 02 07 2017 OVERVIEW An intravitreal implant is a drug delivery system, injected or surgically
EFFICACY OF INTRAVITREAL TRIAMCINOLONE ACETONIDE FOR THE TREATMENT OF DIABETIC MACULAR EDEMA
 Basrah Journal Of Surgery EFFICACY OF INTRAVITREAL TRIAMCINOLONE ACETONIDE FOR THE TREATMENT OF DIABETIC MACULAR EDEMA Salah Zuhair Abed Al-Asadi MB,ChB, FICMS, Lecturer, Department of Surgery, College
Basrah Journal Of Surgery EFFICACY OF INTRAVITREAL TRIAMCINOLONE ACETONIDE FOR THE TREATMENT OF DIABETIC MACULAR EDEMA Salah Zuhair Abed Al-Asadi MB,ChB, FICMS, Lecturer, Department of Surgery, College
ABSTRACT INTRODUCTION
 DOI 10.1007/s40123-017-0114-6 ORIGINAL RESEARCH Real-World Cost Savings Demonstrated by Switching Patients with Refractory Diabetic Macular Edema to Intravitreal Fluocinolone Acetonide (Iluvien): A Retrospective
DOI 10.1007/s40123-017-0114-6 ORIGINAL RESEARCH Real-World Cost Savings Demonstrated by Switching Patients with Refractory Diabetic Macular Edema to Intravitreal Fluocinolone Acetonide (Iluvien): A Retrospective
Jay M. Haynie, O.D.; F.A.A.O. Olympia Tacoma Renton Kennewick Washington
 Jay M. Haynie, O.D.; F.A.A.O. Olympia Tacoma Renton Kennewick Washington I Jay M. Haynie, OD, FAAO have received honoraria from the following companies: Reichert Technologies Notal Vision Carl Zeiss Meditec
Jay M. Haynie, O.D.; F.A.A.O. Olympia Tacoma Renton Kennewick Washington I Jay M. Haynie, OD, FAAO have received honoraria from the following companies: Reichert Technologies Notal Vision Carl Zeiss Meditec
Clinically Significant Macular Edema (CSME)
 Clinically Significant Macular Edema (CSME) 1 Clinically Significant Macular Edema (CSME) Sadrina T. Shaw OMT I Student July 26, 2014 Advisor: Dr. Uwaydat Clinically Significant Macular Edema (CSME) 2
Clinically Significant Macular Edema (CSME) 1 Clinically Significant Macular Edema (CSME) Sadrina T. Shaw OMT I Student July 26, 2014 Advisor: Dr. Uwaydat Clinically Significant Macular Edema (CSME) 2
Facts About Diabetic Eye Disease
 Facts About Diabetic Eye Disease Points to Remember 1. Diabetic eye disease comprises a group of eye conditions that affect people with diabetes. These conditions include diabetic retinopathy, diabetic
Facts About Diabetic Eye Disease Points to Remember 1. Diabetic eye disease comprises a group of eye conditions that affect people with diabetes. These conditions include diabetic retinopathy, diabetic
Diabetic Retinopatathy
 Diabetic Retinopatathy Jay M. Haynie, OD, FAAO Financial Disclosure I have received honoraria or am on the advisory board for the following companies: Carl Zeiss Meditec Arctic DX Macula Risk Advanced
Diabetic Retinopatathy Jay M. Haynie, OD, FAAO Financial Disclosure I have received honoraria or am on the advisory board for the following companies: Carl Zeiss Meditec Arctic DX Macula Risk Advanced
Retinal Vein Occlusion (RVO) Treatment pathway- Northeast England. Retinal Vein Occlusion (RVO) with Macular oedema (MO)
 Retinal Vein Occlusion (RVO) Treatment pathway- Northeast England (Royal Victoria Infirmary, Sunderland Eye Infirmary, James Cook University Hospital, Darlington Memorial Hospital, University Hospital
Retinal Vein Occlusion (RVO) Treatment pathway- Northeast England (Royal Victoria Infirmary, Sunderland Eye Infirmary, James Cook University Hospital, Darlington Memorial Hospital, University Hospital
Recalcitrant Diabetic Macular Oedema: Therapeutic Options
 December 2007 A. Giridhar et al. - Recalcitrant DME 451 CONSULTATION S E C T I O N Recalcitrant Diabetic Macular Oedema: Therapeutic Options Dr. Cyrus M Shroff 1, Dr. N S Muralidhar 2, Dr. R Narayanan
December 2007 A. Giridhar et al. - Recalcitrant DME 451 CONSULTATION S E C T I O N Recalcitrant Diabetic Macular Oedema: Therapeutic Options Dr. Cyrus M Shroff 1, Dr. N S Muralidhar 2, Dr. R Narayanan
INTRAVITREAL CORTICOSTEROIDS IN DIABETIC MACULAR EDEMA Pharmacokinetic Considerations
 Review INTRAVITREAL CORTICOSTEROIDS IN DIABETIC MACULAR EDEMA Pharmacokinetic Considerations YIT YANG, MBCHB, FRCOPHTH,* CLARE BAILEY, MD, FRCP, FRCOPHTH, ANAT LOEWENSTEIN, MD, MHA, PASCALE MASSIN, MD,
Review INTRAVITREAL CORTICOSTEROIDS IN DIABETIC MACULAR EDEMA Pharmacokinetic Considerations YIT YANG, MBCHB, FRCOPHTH,* CLARE BAILEY, MD, FRCP, FRCOPHTH, ANAT LOEWENSTEIN, MD, MHA, PASCALE MASSIN, MD,
All Steroids Are Equal But Some Are More Equal Than Others
 All Steroids Are Equal But Some Are More Equal Than Others Baruch D Kuppermann, MD, PhD Professor of Ophthalmology and Biomedical Engineering Director, Gavin Herbert Eye Institute University of California,
All Steroids Are Equal But Some Are More Equal Than Others Baruch D Kuppermann, MD, PhD Professor of Ophthalmology and Biomedical Engineering Director, Gavin Herbert Eye Institute University of California,
ORIGINAL RESEARCH ARTICLE
 EJO ISSN 1120-6721 Eur J Ophthalmol 2017; 27 (3): 357-362 DOI: 10.5301/ejo.5000929 ORIGINAL RESEARCH ARTICLE Diabetic macular edema outcomes in eyes treated with fluocinolone acetonide 0.2 µg/d intravitreal
EJO ISSN 1120-6721 Eur J Ophthalmol 2017; 27 (3): 357-362 DOI: 10.5301/ejo.5000929 ORIGINAL RESEARCH ARTICLE Diabetic macular edema outcomes in eyes treated with fluocinolone acetonide 0.2 µg/d intravitreal
Review Article Intravitreal Steroids for the Treatment of Retinal Diseases
 e Scientific World Journal, Article ID 989501, 14 pages http://dx.doi.org/10.1155/2014/989501 Review Article Intravitreal Steroids for the Treatment of Retinal Diseases Valentina Sarao, 1 Daniele Veritti,
e Scientific World Journal, Article ID 989501, 14 pages http://dx.doi.org/10.1155/2014/989501 Review Article Intravitreal Steroids for the Treatment of Retinal Diseases Valentina Sarao, 1 Daniele Veritti,
Corporate Medical Policy
 Corporate Medical Policy Intravitreal Implant File Name: Origination: Last CAP Review: Next CAP Review: Last Review: intravitreal_implant 11/2010 6/2017 6/2018 6/2017 Description of Procedure or Service
Corporate Medical Policy Intravitreal Implant File Name: Origination: Last CAP Review: Next CAP Review: Last Review: intravitreal_implant 11/2010 6/2017 6/2018 6/2017 Description of Procedure or Service
EU Regulatory workshop Ophthalmology clinical development and scientific advice. Industry view on DME and macular edema secondary to RVO
 EU Regulatory workshop Ophthalmology clinical development and scientific advice. Industry view on DME and macular edema secondary to RVO Yehia Hashad, M.D. Vice President and Global Therapeutic Area Head
EU Regulatory workshop Ophthalmology clinical development and scientific advice. Industry view on DME and macular edema secondary to RVO Yehia Hashad, M.D. Vice President and Global Therapeutic Area Head
Development of a dexamethasone intravitreal implant for the treatment of noninfectious posterior segment uveitis
 Ann. N.Y. Acad. Sci. ISSN 0077-8923 ANNALS OF THE NEW YORK ACADEMY OF SCIENCES Issue: Pharmaceutical Science to Improve the Human Condition: Prix Galien 2014 Development of a dexamethasone intravitreal
Ann. N.Y. Acad. Sci. ISSN 0077-8923 ANNALS OF THE NEW YORK ACADEMY OF SCIENCES Issue: Pharmaceutical Science to Improve the Human Condition: Prix Galien 2014 Development of a dexamethasone intravitreal
ROLE OF LASER PHOTOCOAGULATION VERSUS INTRAVITREAL TRIAMCINOLONE ACETONIDE IN ANGIOGRAPHIC MACULAR EDEMA IN DIABETES MELLITUS
 ORIGINAL ARTICLE ROLE OF LASER PHOTOCOAGULATION VERSUS INTRAVITREAL TRIAMCINOLONE ACETONIDE IN ANGIOGRAPHIC MACULAR EDEMA IN DIABETES MELLITUS Aggarwal Somesh VP 1, Shah Sonali N 2, Bharwada Rekha M 3,
ORIGINAL ARTICLE ROLE OF LASER PHOTOCOAGULATION VERSUS INTRAVITREAL TRIAMCINOLONE ACETONIDE IN ANGIOGRAPHIC MACULAR EDEMA IN DIABETES MELLITUS Aggarwal Somesh VP 1, Shah Sonali N 2, Bharwada Rekha M 3,
Case Report Inherent Challenges in Managing Long Standing Refractory Diabetic Macular Edema
 Cronicon OPEN ACCESS EC OPHTHALMOLOGY Case Report Inherent Challenges in Managing Long Standing Refractory Diabetic Macular Edema V Swetha E Jeganathan 1,2 * and Karen Madill 3 1 Department of Ophthalmology,
Cronicon OPEN ACCESS EC OPHTHALMOLOGY Case Report Inherent Challenges in Managing Long Standing Refractory Diabetic Macular Edema V Swetha E Jeganathan 1,2 * and Karen Madill 3 1 Department of Ophthalmology,
Paradigm Shift in the treatment of Diabetic Retinopathy. Haytham I. S. Salti, MD Associate Professor
 Paradigm Shift in the treatment of Diabetic Retinopathy Haytham I. S. Salti, MD Associate Professor Disclosure No financial interests related to the subject matter of this talk This presentation includes
Paradigm Shift in the treatment of Diabetic Retinopathy Haytham I. S. Salti, MD Associate Professor Disclosure No financial interests related to the subject matter of this talk This presentation includes
Diagnosis and treatment of diabetic retinopathy. Blake Cooper MD Ophthalmologist Vitreoretinal Surgeon Retina Associates Kansas City
 Diagnosis and treatment of diabetic retinopathy Blake Cooper MD Ophthalmologist Vitreoretinal Surgeon Retina Associates Kansas City Disclosures Consulted for Novo Nordisk 2017,2018. Will be discussing
Diagnosis and treatment of diabetic retinopathy Blake Cooper MD Ophthalmologist Vitreoretinal Surgeon Retina Associates Kansas City Disclosures Consulted for Novo Nordisk 2017,2018. Will be discussing
Information for Patients undergoing Intravitreal Triamcinolone Acetonide (Kenalog) Injection
 Information for Patients undergoing Intravitreal Triamcinolone Acetonide (Kenalog) Injection Kenalog/SS/ST/04.2012/v1.1 review 05.2013 Page 1 Introduction Your doctor has found that you have leakage of
Information for Patients undergoing Intravitreal Triamcinolone Acetonide (Kenalog) Injection Kenalog/SS/ST/04.2012/v1.1 review 05.2013 Page 1 Introduction Your doctor has found that you have leakage of
The Diabetic Retinopathy Clinical Research Network. Management of DME in Eyes with PDR
 The Diabetic Retinopathy Clinical Research Network Management of DME in Eyes with PDR 1 What Has Been Learned? Diabetic Retinopathy Treatment Protocol F: Results suggest that clinically meaningful differences
The Diabetic Retinopathy Clinical Research Network Management of DME in Eyes with PDR 1 What Has Been Learned? Diabetic Retinopathy Treatment Protocol F: Results suggest that clinically meaningful differences
Study of clinical significance of optical coherence tomography in diagnosis & management of diabetic macular edema
 Original Research Article Study of clinical significance of optical coherence tomography in diagnosis & management of diabetic macular edema Neha Kantilal Desai 1,*, Somesh Vedprakash Aggarwal 2, Sonali
Original Research Article Study of clinical significance of optical coherence tomography in diagnosis & management of diabetic macular edema Neha Kantilal Desai 1,*, Somesh Vedprakash Aggarwal 2, Sonali
Charles C. Wykoff MD PhD Rahul N. Khurana MD
 HDWallpapers Suprachoroidal Triamcinolone Acetonide with & without Intravitreal Aflibercept for DME: Results of the 6 Month Prospective Phase 1/2 Hulk trial Blanton Eye Institute Charles C. Wykoff MD PhD
HDWallpapers Suprachoroidal Triamcinolone Acetonide with & without Intravitreal Aflibercept for DME: Results of the 6 Month Prospective Phase 1/2 Hulk trial Blanton Eye Institute Charles C. Wykoff MD PhD
 Research Article http://www.alliedacademies.org/clinical-ophthalmology-and-vision-science/ A 2-year retrospective study of the treatment of retinal vein occlusion with dexamethasone 0.7 mg intravitreal
Research Article http://www.alliedacademies.org/clinical-ophthalmology-and-vision-science/ A 2-year retrospective study of the treatment of retinal vein occlusion with dexamethasone 0.7 mg intravitreal
Retina Diabetic Macular Edema
 Retina Diabetic Macular Edema ILUVIEN A New Approach to the Treatment of Diabetic Macula Edema Pravin U Dugel, MD 1 and Richard Parrish, MD 2 1. Retinal Consultants of Arizona, Phoenix, Arizona, US; Clinical
Retina Diabetic Macular Edema ILUVIEN A New Approach to the Treatment of Diabetic Macula Edema Pravin U Dugel, MD 1 and Richard Parrish, MD 2 1. Retinal Consultants of Arizona, Phoenix, Arizona, US; Clinical
84 Year Old with Rosacea
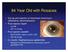 84 Year Old with Rosacea S/p tap and injection of intravitreal vancomycin, ceftazidime, dexamethasone Post-injection day#1 Va HM IOP 14 mmhg Post-injection week#3 BCVA 20/20-3 (plano +0.50 x 180) IOP 23
84 Year Old with Rosacea S/p tap and injection of intravitreal vancomycin, ceftazidime, dexamethasone Post-injection day#1 Va HM IOP 14 mmhg Post-injection week#3 BCVA 20/20-3 (plano +0.50 x 180) IOP 23
Intravitreal Injection
 for patients Eye Clinic Ipswich Hospital Tel: 01473 703230 Intravitreal Injection What is an intravitreal injection? An intravitreal injection is the injection of a drug into the vitreous body (the jelly
for patients Eye Clinic Ipswich Hospital Tel: 01473 703230 Intravitreal Injection What is an intravitreal injection? An intravitreal injection is the injection of a drug into the vitreous body (the jelly
Combination Treatment of Diabetic Macular Edema with Anti-Vascular Endothelial Growth Factor and Steroids: Analysis of DRCR.
 REVIEW ARTICLE Combination Treatment of Diabetic Macular Edema with Anti-Vascular Endothelial Growth Factor and Steroids: Analysis of DRCR.net Protocol U Cindy Ung 1, Kareem Moussa 1, Yoshihiro Yonekawa
REVIEW ARTICLE Combination Treatment of Diabetic Macular Edema with Anti-Vascular Endothelial Growth Factor and Steroids: Analysis of DRCR.net Protocol U Cindy Ung 1, Kareem Moussa 1, Yoshihiro Yonekawa
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE. Health Technology Appraisal. Aflibercept for treating diabetic macular oedema.
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Health Technology Appraisal Aflibercept for treating diabetic macular oedema Final scope Final remit/appraisal objective To appraise the clinical and cost
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Health Technology Appraisal Aflibercept for treating diabetic macular oedema Final scope Final remit/appraisal objective To appraise the clinical and cost
Diabetic retinopathy (DR) progressively
 FOCUS ON DIABETIC MACULAR EDEMA * Frederick L. Ferris III, MD ABSTRACT The current state of treatment for diabetic macular edema (DME) is focused on slowing the rate of vision loss through assessment of
FOCUS ON DIABETIC MACULAR EDEMA * Frederick L. Ferris III, MD ABSTRACT The current state of treatment for diabetic macular edema (DME) is focused on slowing the rate of vision loss through assessment of
Diabetic Retinopathy Clinical Research Network
 Diabetic Retinopathy Clinical Research Network A Randomized Trial Comparing Intravitreal Triamcinolone Acetonide and Laser Photocoagulation for Diabetic Macular Edema Version 6.0 July 20, 2007 Triamcinolone
Diabetic Retinopathy Clinical Research Network A Randomized Trial Comparing Intravitreal Triamcinolone Acetonide and Laser Photocoagulation for Diabetic Macular Edema Version 6.0 July 20, 2007 Triamcinolone
Original Effective Date: 10/24/2016. Subject: Intravitreal corticosteroid implants: Ozurdex (dexamethasone intravitreal implant)
 Subject: Intravitreal corticosteroid implants: Ozurdex (dexamethasone intravitreal implant) Original Effective Date: 10/24/2016 Policy Number: MCP-282 Revision Date(s): 12/13/2017 DISCLAIMER This Molina
Subject: Intravitreal corticosteroid implants: Ozurdex (dexamethasone intravitreal implant) Original Effective Date: 10/24/2016 Policy Number: MCP-282 Revision Date(s): 12/13/2017 DISCLAIMER This Molina
Scott M. Pfahler D.O. Dayton Vitreo-Retinal Associates AOCOO-HNS Palm Springs, CA 2012
 Scott M. Pfahler D.O. Dayton Vitreo-Retinal Associates AOCOO-HNS Palm Springs, CA 2012 Proliferative Diabetic Retinopathy Laser Treatments Medical Treatment Surgical Treatment Diabetic Macular Edema Laser
Scott M. Pfahler D.O. Dayton Vitreo-Retinal Associates AOCOO-HNS Palm Springs, CA 2012 Proliferative Diabetic Retinopathy Laser Treatments Medical Treatment Surgical Treatment Diabetic Macular Edema Laser
CENTENE PHARMACY AND THERAPEUTICS NEW DRUG REVIEW 2Q17 April May
 BRAND NAME Lucentis GENERIC NAME ranibizumab MANUFACTURER Genentech, Inc. DATE OF APPROVAL June 30, 2006 PRODUCT LAUNCH DATE July 13, 2006 REVIEW TYPE Review type 1 (RT1): New Drug Review Full review of
BRAND NAME Lucentis GENERIC NAME ranibizumab MANUFACTURER Genentech, Inc. DATE OF APPROVAL June 30, 2006 PRODUCT LAUNCH DATE July 13, 2006 REVIEW TYPE Review type 1 (RT1): New Drug Review Full review of
Targeting Inflammation in Diabetic Macular Edema: From Basic Science to Clinical Trials to Clinical Practice
 Targeting Inflammation in Diabetic Macular Edema: From Basic Science to Clinical Trials to Clinical Practice Baruch D Kuppermann, MD, PhD Professor of Ophthalmology and Biomedical Engineering Director,
Targeting Inflammation in Diabetic Macular Edema: From Basic Science to Clinical Trials to Clinical Practice Baruch D Kuppermann, MD, PhD Professor of Ophthalmology and Biomedical Engineering Director,
Intravitreal versus Posterior Subtenon Injection of Triamcinolone Acetonide for Diabetic Macular Edema
 Intravitreal versus Posterior Subtenon Injection of Triamcinolone Acetonide for Diabetic Macular Edema Young Jae Choi, MD, In Kyung Oh, MD, Jae Ryung Oh, MD, PhD, Kuhl Huh, MD, PhD Department of Ophthalmology,
Intravitreal versus Posterior Subtenon Injection of Triamcinolone Acetonide for Diabetic Macular Edema Young Jae Choi, MD, In Kyung Oh, MD, Jae Ryung Oh, MD, PhD, Kuhl Huh, MD, PhD Department of Ophthalmology,
psivida Transforms into Commercial Stage Specialty BioPharmaceutical Company AAO October 25, 2018 NASDAQ: EYPT
 psivida Transforms into Commercial Stage Specialty BioPharmaceutical Company OIS @ AAO October 25, 2018 NASDAQ: EYPT Forward Looking SAFE HARBOR STATEMENTS UNDER THE PRIVATE SECURITIES LITIGATION REFORM
psivida Transforms into Commercial Stage Specialty BioPharmaceutical Company OIS @ AAO October 25, 2018 NASDAQ: EYPT Forward Looking SAFE HARBOR STATEMENTS UNDER THE PRIVATE SECURITIES LITIGATION REFORM
An Update on Branch Retinal Vein Occlusion Treatment Studies. Amiee Ho, O.D. Pacific University College of Optometry
 An Update on Branch Retinal Vein Occlusion Treatment Studies Amiee Ho, O.D. Pacific University College of Optometry Course Description This course focuses on current treatment options available for macular
An Update on Branch Retinal Vein Occlusion Treatment Studies Amiee Ho, O.D. Pacific University College of Optometry Course Description This course focuses on current treatment options available for macular
COMPARISON OF INTRAVITREAL TRIAMCINOLONE INJECTION VS LASER PHOTOCOAGULATION IN ANGIOGRAPHIC MACULAR EDEMA IN DIABETIC RETINOPATHY
 Original Article COMPARISON OF INTRAVITREAL TRIAMCINOLONE INJECTION VS LASER PHOTOCOAGULATION IN ANGIOGRAPHIC MACULAR EDEMA IN DIABETIC RETINOPATHY Aggarwal Somesh V 1, Shah Sonali N 2, Bharwada Rekha
Original Article COMPARISON OF INTRAVITREAL TRIAMCINOLONE INJECTION VS LASER PHOTOCOAGULATION IN ANGIOGRAPHIC MACULAR EDEMA IN DIABETIC RETINOPATHY Aggarwal Somesh V 1, Shah Sonali N 2, Bharwada Rekha
A ROUNDTABLE DISCUSSION FEATURING:
 Jointly sponsored by the Dulaney Foundation and Retina Today. Supplement to October 2009 New and Emerging Treatment Paradigms for Macular Edema in Retinal Vein Occlusion and Diabetic Retinopathy A ROUNDTABLE
Jointly sponsored by the Dulaney Foundation and Retina Today. Supplement to October 2009 New and Emerging Treatment Paradigms for Macular Edema in Retinal Vein Occlusion and Diabetic Retinopathy A ROUNDTABLE
Ophthalmic VEGF Inhibitors. Eylea (aflibercept), Macugen (pegaptanib) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Ophthalmic VEGF Inhibitors Page: 1 of 5 Last Review Date: September 20, 2018 Ophthalmic VEGF Inhibitors
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Ophthalmic VEGF Inhibitors Page: 1 of 5 Last Review Date: September 20, 2018 Ophthalmic VEGF Inhibitors
SUMMARY. Heather Casparis, MD,* and Neil M. Bressler, MD MARINA AND ANCHOR
 The following are summaries of selected presentations and posters from the American Society of Retina Specialists and European VitreoRetinal Society Annual Meeting held September 9 13, 2006, in Cannes,
The following are summaries of selected presentations and posters from the American Society of Retina Specialists and European VitreoRetinal Society Annual Meeting held September 9 13, 2006, in Cannes,
Vascular Disease Ocular Manifestations of Systemic Hypertension
 Vascular Disease Ocular Manifestations of Systemic Hypertension Maynard L. Pohl, OD, FAAO Pacific Cataract & Laser Institute 10500 NE 8 th Street, Suite 1650 Bellevue, WA 98004 USA 425-462-7664 Cerebrovascular
Vascular Disease Ocular Manifestations of Systemic Hypertension Maynard L. Pohl, OD, FAAO Pacific Cataract & Laser Institute 10500 NE 8 th Street, Suite 1650 Bellevue, WA 98004 USA 425-462-7664 Cerebrovascular
Diabetic Macular Edema Treatment in the 21st Century
 Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Transcript Details This is a transcript of a continuing medical education (CME) activity accessible on the ReachMD network. Additional media formats for the activity and full activity details (including
Taiwan Journal of Ophthalmology
 Taiwan Journal of Ophthalmology 3 (2013) 163e167 Contents lists available at SciVerse ScienceDirect Taiwan Journal of Ophthalmology journal homepage: www.e-tjo.com Case report Intravitreal dexamethasone
Taiwan Journal of Ophthalmology 3 (2013) 163e167 Contents lists available at SciVerse ScienceDirect Taiwan Journal of Ophthalmology journal homepage: www.e-tjo.com Case report Intravitreal dexamethasone
1/25/2018. Case Management Strategies in Diabetic Retinopathy. Case Study #1: Severe DME. DDOS: 3/31/2016 Va 20/400. Disclosures
 Case Management Strategies in Diabetic Retinopathy Disclosures No financial conflict of interest Will discuss off label use of intraocular Bevacizumab (Avastin) for Diabetic Retinopathy Sundeep Dev, MD
Case Management Strategies in Diabetic Retinopathy Disclosures No financial conflict of interest Will discuss off label use of intraocular Bevacizumab (Avastin) for Diabetic Retinopathy Sundeep Dev, MD
Treatment of Retinal Vein Occlusion (RVO)
 Manchester Royal Eye Hospital Medical Retina Services Information for Patients Treatment of Retinal Vein Occlusion (RVO) What is a Retinal Vein Occlusion (RVO)? The retina is the light sensitive layer
Manchester Royal Eye Hospital Medical Retina Services Information for Patients Treatment of Retinal Vein Occlusion (RVO) What is a Retinal Vein Occlusion (RVO)? The retina is the light sensitive layer
Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME
 Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME Ophthalmology Innovation Summit @ AAO, October 25 2018 Megan Baldwin PhD, CEO & Managing Director Disclaimer Investment
Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME Ophthalmology Innovation Summit @ AAO, October 25 2018 Megan Baldwin PhD, CEO & Managing Director Disclaimer Investment
An updated review of long-term outcomes from randomized controlled trials in approved pharmaceuticals for diabetic macular edema
 Review Article An updated review of long-term outcomes from randomized controlled trials in approved pharmaceuticals for diabetic macular edema Jia-Kang Wang 1,2,3,4, Tzu-Lun Huang 1,5, Pei-Yuan Su 1,4,6,
Review Article An updated review of long-term outcomes from randomized controlled trials in approved pharmaceuticals for diabetic macular edema Jia-Kang Wang 1,2,3,4, Tzu-Lun Huang 1,5, Pei-Yuan Su 1,4,6,
A retrospective nonrandomized study was conducted at 3
 Department of Ophthalmology, Kangbuk Samsung Hospital, Sungkyunkwan University College of Medicine 1, Seoul, Korea Hangil Eye Hospital 2, Incheon, Korea Seoul National University Bundang Hospital 3, Seongnam,
Department of Ophthalmology, Kangbuk Samsung Hospital, Sungkyunkwan University College of Medicine 1, Seoul, Korea Hangil Eye Hospital 2, Incheon, Korea Seoul National University Bundang Hospital 3, Seongnam,
Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME
 Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME Ophthalmology Innovation Summit @ AAO, October 25 2018 Megan Baldwin PhD, CEO & Managing Director Disclaimer Investment
Clinical results of OPT-302 (VEGF-C/D Trap ) Combination Treatment in namd and DME Ophthalmology Innovation Summit @ AAO, October 25 2018 Megan Baldwin PhD, CEO & Managing Director Disclaimer Investment
Dexamethasone posterior segment drug delivery system (Ozurdex) for diabetic macular oedema
 Dexamethasone posterior segment drug delivery system (Ozurdex) for diabetic macular oedema This technology summary is based on information available at the time of research and a limited literature search.
Dexamethasone posterior segment drug delivery system (Ozurdex) for diabetic macular oedema This technology summary is based on information available at the time of research and a limited literature search.
 measure of your overall performance. An isolated glucose test is helpful to let you know what your sugar level is at one moment, but it doesn t tell you whether or not your diabetes is under adequate control
measure of your overall performance. An isolated glucose test is helpful to let you know what your sugar level is at one moment, but it doesn t tell you whether or not your diabetes is under adequate control
Research Article Differentiation between Good and Low-Responders to Intravitreal Ranibizumab for Macular Edema Secondary to Retinal Vein Occlusion
 Hindawi Publishing Corporation Journal of Ophthalmology Volume 2016, Article ID 9875741, 6 pages http://dx.doi.org/10.1155/2016/9875741 Research Article Differentiation between Good and Low-Responders
Hindawi Publishing Corporation Journal of Ophthalmology Volume 2016, Article ID 9875741, 6 pages http://dx.doi.org/10.1155/2016/9875741 Research Article Differentiation between Good and Low-Responders
Diabetic Retinopathy Clinical Research Network
 Diabetic Retinopathy Clinical Research Network Short-term Evaluation of Combination Corticosteroid+Anti- VEGF Treatment for Persistent Central-Involved Diabetic Macular Edema Following Anti-VEGF Therapy
Diabetic Retinopathy Clinical Research Network Short-term Evaluation of Combination Corticosteroid+Anti- VEGF Treatment for Persistent Central-Involved Diabetic Macular Edema Following Anti-VEGF Therapy
Diabetic Macular Oedema To treat or not to treat?
 Diabetic Macular Oedema To treat or not to treat? Ms Ranjit Sandhu MBBS MRCOphth MD FRCOphth Consultant Ophthalmic Surgeon Cataract Surgery, Medical Retina & Uveitis The Luton and Dunstable University
Diabetic Macular Oedema To treat or not to treat? Ms Ranjit Sandhu MBBS MRCOphth MD FRCOphth Consultant Ophthalmic Surgeon Cataract Surgery, Medical Retina & Uveitis The Luton and Dunstable University
Although photocoagulation and photodynamic PROCEEDINGS PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION *
 PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION Evangelos S. Gragoudas, MD ABSTRACT In December 24, the US Food and Drug Administration (FDA) approved pegaptanib sodium. Pegaptanib
PEGAPTANIB SODIUM FOR THE TREATMENT OF AGE-RELATED MACULAR DEGENERATION Evangelos S. Gragoudas, MD ABSTRACT In December 24, the US Food and Drug Administration (FDA) approved pegaptanib sodium. Pegaptanib
Despite our growing knowledge of the
 OVERVIEW OF AVAILABLE TREATMENTS FOR NEOVASCULAR AGE-RELATED MACULAR DEGENERATION * Carl D. Regillo, MD ABSTRACT Although potential treatments for neovascular age-related macular degeneration represent
OVERVIEW OF AVAILABLE TREATMENTS FOR NEOVASCULAR AGE-RELATED MACULAR DEGENERATION * Carl D. Regillo, MD ABSTRACT Although potential treatments for neovascular age-related macular degeneration represent
New developments in the treatment of diabetic macular edema: latest clinical evidence
 Review: Clinical Trial Outcomes New developments in the treatment of diabetic macular edema: latest clinical evidence Clin. Invest. (2012) 2(1), 89 105 Diabetic retinopathy is a major cause of blindness
Review: Clinical Trial Outcomes New developments in the treatment of diabetic macular edema: latest clinical evidence Clin. Invest. (2012) 2(1), 89 105 Diabetic retinopathy is a major cause of blindness
INTRAVITREAL IMPLANTS
 INTRAVITREAL IMPLANTS Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
INTRAVITREAL IMPLANTS Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and drugs
Low Illumination 3-D Heads-Up Vitrectomy for Diabetic Macular Edema
 Low Illumination 3-D Heads-Up Vitrectomy for Diabetic Macular Edema Yoshihiro Yonekawa, M.D., Bozho Todorich, M.D., Ph.D., Jeremy D. Wolfe, M.D. Yoshihiro Yonekawa, M.D., is a vitreoretinal surgeon at
Low Illumination 3-D Heads-Up Vitrectomy for Diabetic Macular Edema Yoshihiro Yonekawa, M.D., Bozho Todorich, M.D., Ph.D., Jeremy D. Wolfe, M.D. Yoshihiro Yonekawa, M.D., is a vitreoretinal surgeon at
Darren J. Bell, M.D. Texas A&M University College of Medicine College Station, Texas Doctor of Medicine
 Darren J. Bell, M.D. Education Texas A&M University College of Medicine College Station, Texas Doctor of Medicine 1997-2001 Texas A&M University College Station, Texas Bachelor of Science, Biomedical Engineering
Darren J. Bell, M.D. Education Texas A&M University College of Medicine College Station, Texas Doctor of Medicine 1997-2001 Texas A&M University College Station, Texas Bachelor of Science, Biomedical Engineering
Visual Acuity Outcomes in Diabetic Macular Edema With Fluocinolone Acetonide 0.2 µg/day Versus Ranibizumab Plus Deferred Laser (DRCR Protocol I)
 EXPERIMENTAL SCIENCE Visual Acuity Outcomes in Diabetic Macular Edema With Fluocinolone Acetonide 0.2 µg/day Versus Ranibizumab Plus Deferred Laser (DRCR Protocol I) Michael A. Singer, MD; Dan M. Miller,
EXPERIMENTAL SCIENCE Visual Acuity Outcomes in Diabetic Macular Edema With Fluocinolone Acetonide 0.2 µg/day Versus Ranibizumab Plus Deferred Laser (DRCR Protocol I) Michael A. Singer, MD; Dan M. Miller,
Suprachoroidal Triamcinolone Acetonide for Retinal Vein Occlusion: Results of the Tanzanite Study
 Suprachoroidal Triamcinolone Acetonide for Retinal Vein Occlusion: Results of the Tanzanite Study Peter A. Campochiaro, MD, 1 Charles C. Wykoff, MD, 2 David M. Brown, MD, 2 David S. Boyer, MD, 3 Mark Barakat,
Suprachoroidal Triamcinolone Acetonide for Retinal Vein Occlusion: Results of the Tanzanite Study Peter A. Campochiaro, MD, 1 Charles C. Wykoff, MD, 2 David M. Brown, MD, 2 David S. Boyer, MD, 3 Mark Barakat,
CURRENT TRENDS IN DIABETIC MACULAR EDEMA TREATMENT. Muge R. Kesen, MD
 CURRENT TRENDS IN DIABETIC MACULAR EDEMA TREATMENT Muge R. Kesen, MD No relevant financial interest or relationships DISCLOSURE OBJECTIVES Current trends (evidence based) Review of clinical trials Diabetic
CURRENT TRENDS IN DIABETIC MACULAR EDEMA TREATMENT Muge R. Kesen, MD No relevant financial interest or relationships DISCLOSURE OBJECTIVES Current trends (evidence based) Review of clinical trials Diabetic
Subclinical Diabetic Macular Edema Study
 Diabetic Retinopathy Clinical Research Network Subclinical Diabetic Macular Edema Study Version 2.0 March 21, 2006 Subclinical DME Protocol v2 0 3-21-06.doc Table of Contents Chapter 1. Background Information
Diabetic Retinopathy Clinical Research Network Subclinical Diabetic Macular Edema Study Version 2.0 March 21, 2006 Subclinical DME Protocol v2 0 3-21-06.doc Table of Contents Chapter 1. Background Information
Dexamethasone Intravitreal Implant Rescue Treatment for Bevacizumab Refractory Macular Edema Secondary to Branch Retinal Vein Occlusion
 pissn: 1011-8942 eissn: 2092-9382 Korean J Ophthalmol 2017;31(2):108-114 https://doi.org/10.3341/kjo.2017.31.2.108 Original Article Dexamethasone Intravitreal Implant Rescue Treatment for Bevacizumab Refractory
pissn: 1011-8942 eissn: 2092-9382 Korean J Ophthalmol 2017;31(2):108-114 https://doi.org/10.3341/kjo.2017.31.2.108 Original Article Dexamethasone Intravitreal Implant Rescue Treatment for Bevacizumab Refractory
Applications of Sustained Release Delivery Systems in Ocular Disease
 Applications of Sustained Release Delivery Systems in Ocular Disease Formulation and Delivery Systems For Peptide and Protein 2012 December 5, 2012 Christopher A. Rhodes, Ph.D. Principal, Christopher A
Applications of Sustained Release Delivery Systems in Ocular Disease Formulation and Delivery Systems For Peptide and Protein 2012 December 5, 2012 Christopher A. Rhodes, Ph.D. Principal, Christopher A
Learn Connect Succeed. JCAHPO Regional Meetings 2017
 Learn Connect Succeed JCAHPO Regional Meetings 2017 Intravitreal Injection Technique Updated Recommendations from an Expert Panel Sophie J. Bakri, M.D. Professor of Ophthalmology Vitreoretinal Diseases
Learn Connect Succeed JCAHPO Regional Meetings 2017 Intravitreal Injection Technique Updated Recommendations from an Expert Panel Sophie J. Bakri, M.D. Professor of Ophthalmology Vitreoretinal Diseases
RETINA TODAY. for Therapeutic Use in the Posterior Segment CME ACTIVITY. Jointly sponsored by the Dulaney Foundation and Retina Today.
 Supplement to CME ACTIVITY RETINA TODAY November/December 2010 Jointly sponsored by the Dulaney Foundation and Retina Today. for Therapeutic Use in the Posterior Segment Jointly sponsored by the Dulaney
Supplement to CME ACTIVITY RETINA TODAY November/December 2010 Jointly sponsored by the Dulaney Foundation and Retina Today. for Therapeutic Use in the Posterior Segment Jointly sponsored by the Dulaney
Diabetic eye disease. Diabetic retinopathy. Sam S. Dahr, M.D. Retina Center of Oklahoma.
 Diabetic eye disease Sam S. Dahr, M.D. Retina Center of Oklahoma www.rcoklahoma.com Downloaded from: The Retina (on 28 May 2007 12:48 AM) 2007 Elsevier Diabetic retinopathy Downloaded from: The Retina
Diabetic eye disease Sam S. Dahr, M.D. Retina Center of Oklahoma www.rcoklahoma.com Downloaded from: The Retina (on 28 May 2007 12:48 AM) 2007 Elsevier Diabetic retinopathy Downloaded from: The Retina
Dan Roberts. International Low Vision Support Group NEWSLETTER. This Month. In This Issue. 1: This Month 2-5: News
 In This Issue 1: This Month 2-5: News Eye & Body Complete from Biosyntrx Extra Strength Protection Call for information 800-688-6815 The ILVSG is sponsored by MD Foundation www.eyesight.org 888-633-3937
In This Issue 1: This Month 2-5: News Eye & Body Complete from Biosyntrx Extra Strength Protection Call for information 800-688-6815 The ILVSG is sponsored by MD Foundation www.eyesight.org 888-633-3937
Original Paper. Ophthalmic Res DOI: /
 Original Paper Received: December 19, 216 Accepted: December 19, 216 Published online: February 9, 217 A Nonrandomized, Open-Label, Multicenter, Phase 4 Pilot Study on the Effect and Safety of ILUVIEN
Original Paper Received: December 19, 216 Accepted: December 19, 216 Published online: February 9, 217 A Nonrandomized, Open-Label, Multicenter, Phase 4 Pilot Study on the Effect and Safety of ILUVIEN
Applying New Data to Improve the Standard of Care in Retinal Disease. With articles by Gaurav K. Shah, MD Carl D. Regillo, MD.
 Supplement to July/August 2012 CME Activity Applying New Data to Improve the Standard of Care in Retinal Disease With articles by Gaurav K. Shah, MD Carl D. Regillo, MD Release date: August 2012. Expiration
Supplement to July/August 2012 CME Activity Applying New Data to Improve the Standard of Care in Retinal Disease With articles by Gaurav K. Shah, MD Carl D. Regillo, MD Release date: August 2012. Expiration
Ophthalmology Macular Pathways
 Ophthalmology Macular Pathways Age related Macular Degeneration Diabetic Macular Oedema Macular Oedema secondary to Central Retinal Macular Oedema secondary to Branch Retinal CNV associated with pathological
Ophthalmology Macular Pathways Age related Macular Degeneration Diabetic Macular Oedema Macular Oedema secondary to Central Retinal Macular Oedema secondary to Branch Retinal CNV associated with pathological
Vascular Endothelial Growth Factor (VEGF) Inhibitors Ocular Use Drug Class Monograph (Medical Benefit)
 Vascular Endothelial Growth Factor (VEGF) Inhibitors Ocular Use Drug Class Monograph (Medical Benefit) Line of Business: Medi-Cal Effective Date: May 17, 2017 Revision Date: May 17, 2017 This policy has
Vascular Endothelial Growth Factor (VEGF) Inhibitors Ocular Use Drug Class Monograph (Medical Benefit) Line of Business: Medi-Cal Effective Date: May 17, 2017 Revision Date: May 17, 2017 This policy has
PART 1: GENERAL RETINAL ANATOMY
 PART 1: GENERAL RETINAL ANATOMY General Anatomy At Ora Serrata At Optic Nerve Head Fundoscopic View Of Normal Retina What Is So Special About Diabetic Retinopathy? The WHO definition of blindness is
PART 1: GENERAL RETINAL ANATOMY General Anatomy At Ora Serrata At Optic Nerve Head Fundoscopic View Of Normal Retina What Is So Special About Diabetic Retinopathy? The WHO definition of blindness is
A Patient s Guide to Diabetic Retinopathy
 Diabetic Retinopathy A Patient s Guide to Diabetic Retinopathy 840 Walnut Street, Philadelphia PA 19107 www.willseye.org Diabetic Retinopathy 1. Definition Diabetic retinopathy is a complication of diabetes
Diabetic Retinopathy A Patient s Guide to Diabetic Retinopathy 840 Walnut Street, Philadelphia PA 19107 www.willseye.org Diabetic Retinopathy 1. Definition Diabetic retinopathy is a complication of diabetes
Corporate Presentation August 2018
 Corporate Presentation August 218 Forward-Looking Statements This presentation contains forward-looking statements as defined in the Private Securities Litigation Reform Act of 1995, as amended. The words
Corporate Presentation August 218 Forward-Looking Statements This presentation contains forward-looking statements as defined in the Private Securities Litigation Reform Act of 1995, as amended. The words
Clinical Outcomes After Intravitreal Bevacizumab Injection for Diabetic Macular Edema
 Original Article Clinical Outcomes After Intravitreal Bevacizumab Injection for Diabetic Macular Edema Karen Joyce G. Castro, MD, Marie Joan V. Loy, MD International Eye Institute St. Luke s Medical Center
Original Article Clinical Outcomes After Intravitreal Bevacizumab Injection for Diabetic Macular Edema Karen Joyce G. Castro, MD, Marie Joan V. Loy, MD International Eye Institute St. Luke s Medical Center
Intravitreal Ranibizumab or Triamcinolone Acetonide in Combination with Laser Photocoagulation for Diabetic Macular Edema
 Diabetic Retinopathy Clinical Research Network Intravitreal Ranibizumab or Triamcinolone Acetonide in Combination with Laser Photocoagulation for Diabetic Macular Edema Version 6.0 May 21, 2010 Note: See
Diabetic Retinopathy Clinical Research Network Intravitreal Ranibizumab or Triamcinolone Acetonide in Combination with Laser Photocoagulation for Diabetic Macular Edema Version 6.0 May 21, 2010 Note: See
WGA. The Global Glaucoma Network
 The Global Glaucoma Network Fort Lauderdale April 30, 2005 Indications for Surgery 1. The decision for surgery should consider the risk/benefit ratio. Note: Although a lower IOP is generally considered
The Global Glaucoma Network Fort Lauderdale April 30, 2005 Indications for Surgery 1. The decision for surgery should consider the risk/benefit ratio. Note: Although a lower IOP is generally considered
Current Insight Into Retinal Disease Management: Focus on DME and Intravitreal Corticosteroids
 VIEW CASE VIDEOS ONLINE Supplement to March 2015 CME ACTIVITY Current Insight Into Retinal Disease Management: Focus on DME and Intravitreal Corticosteroids Allen C. Ho, MD, Moderator Michael D. Ober,
VIEW CASE VIDEOS ONLINE Supplement to March 2015 CME ACTIVITY Current Insight Into Retinal Disease Management: Focus on DME and Intravitreal Corticosteroids Allen C. Ho, MD, Moderator Michael D. Ober,
