PHARMACY PEARLS CHANIN WRIGHT, PHARMD
|
|
|
- Corey Holmes
- 6 years ago
- Views:
Transcription
1 PHARMACY PEARLS CHANIN WRIGHT, PHARMD
2 OBJECTIVES From the pearls heard, discuss with someone near you What is something you can apply to your setting
3
4 Traumatic brain injury in children Goals Correct and prevent early hypotension and hypoxia Establish an airway What drug should you use Ketamine = decrease ICP and increase CPP CPP = MAP - ICP Document GCS prior to medications J Neurosurg Pediatr 2009;4:40-6
5 TBI Mannitol Although mannitol is commonly used in the management of raised ICP in pediatric TBI, no studies meeting inclusion criteria were identified for use as evidence for this topic
6 TBI Hypertonic saline Should be considered for the treatment of severe TBI associated with intracranial hypertension There is insufficient evidence to support or refute the use of hypertonic saline >3% Dose: ml/kg or CI: ml/kg Minimum dose needed to maintain ICP <20 should be used Serum osmolarity should be maintained <360
7 TBI Pentobarbital Should be considered for refractory ICP Barbiturate coma LD: -2 mg/kg CI: 1- mg/kg/hr Serum levels : mg/l (send out) Myocardial suppression Dopamine, epinephrine Long term effects on neurological outcomes are unknown
8 TBI 2012 guidelines The use of corticosteroids are not recommended to improve the outcome or reduce ICP for children with TBI No studies showing efficacy GI bleeding Glucose intolerance Electrolyte abnormalities infection
9 Post Traumatic Seizures Occurs early within days of injury Pediatrics at greater risk than adults <2 years old 3 fold increased risk than children between 2 and 12 Greatest risk < days post injury GCS <10 Cortical contusion Depressed skull fracture Subdural, epidural, intracerebral hematoma Penetrating head wound Seizures in the 1 st 24 hours post injury
10 Post Traumatic Seizures Phenytoin vs. placebo 3.6% vs. 14.2% Phenytoin for the 1 st days post injury in patients who had no seizure on presentation Subsequent trials have shown similar results Levetiracetam 2 studies concluded equal efficacy compared to phenytoin At this time can t be recommended over phenytoin
11 TBI DVT prophylaxis Consider if 30 kg > or any sign of puberty SQ heparin or SQ enoxaparin May be started within 48 hours of TBI
12 Emergency Medicine PEARLS Intranasal midazolam 0.2 mg/kg Imaging, positioning 0. mg/kg Laceration repair Ortho procedures IV line placement Maximum of 10 mg /dose or repeat administration
13 IN Midazolam May burn with administration Few patients complain or have memory of burning Shows no significant respiratory depression Give a max of 1 ml per nare Rec mg/ml conc Allows for a rapid, non-invasive method for mild to moderate sedation
14 Adenosine IO Dosing : 0.1 mg/kg 0.3 mg/kg Max of 6 mg initially, and 12mg on repeat Extremely short half-life (<10 sec) Reduce initial dose if post-cardiac transplant patients On dipyridamole or carbamazepine Given through a central line Increase dose if on theophylline or caffeine
15 Adenosine IO Case report of administration through IO 11 day old with SVT Converted with IV adenosine 0.1 mg/kg IO adenosine converted to NSR Converted to SVT Additional 0.1 mg/kg IO needed
16 Adenosine IO Case report 2 month old with SVT Converted with IV adenosine 0.0 mg/kg, 0.1 mg/kg, 0.2 mg/kg IO no conversion 0.1 mg/kg given IV peripherally converted to NSR
17 Adenosine IO Case report 4 month old Converted with IV adenosine 0.1 mg/kg, 0.2 mg/kg no conversion 0.1 mg/kg, 0.2 mg/kg femorally caused transient asystole, conversion to NSR, then return to SVT
18 Methylene Blue for Joint Injuries Suspected open joint injuries extravasation would be highly indicative of open joint injury Use 1% lidocaine first (0.6ml/kg max) 1 ml methylene blue in 00 ml NS instilled into the joint space Adverse effects Injection of methylene blue may cause effusion Contraindications Known hypersensitivity to the drug Severe renal impairment G6PD deficiency Methemoglobinemia Topical skin discoloration can be treated with a weak bleach solution
19 SCIWORA Spinal cord injury w/o radiographic abnormality Over 6% cases occur in children <8 years old High dose methylprednisolone NOT routinely recommended Fluid resuscitation and pressor therapy alpha agonists i.e.. Norepinephrine or phenylephrine
20 Tissue entrapment by zipper
21 Tissue entrapment by zipper Before reaching for the wire cutters which freak out a grown man let alone a year old Try a liberal amount of mineral oil to the area and waiting -10 minutes. Often the tissue will slide out easily! If not, a dose of IN midazolam may be in order before the wire cutters are brought out!
22 SVC syndrome Superior vena cava syndrome Encircled by lymph nodes responsible for draining thoracic cavity Most often seen in kids with Hodgkin's or non- Hodgkin's Lymphoma Massive adenopathy obstructs SVC, resulting in dilated chest and neck veins, facial edema, drowsiness, stupor All intravenous therapy must be via the lower extremities SVC obstruction results in poor circulation in the upper extremities and torso Drug distribution is restricted and there is an increased risk of thrombus formation
23 Topical Analgesia Many available products Aerosols Creams Gels Ointments Injections Compounds
24 Topicals Product selection depends on procedure Is the skin broken or intact What is the surface area involved What is your timeframe Three common products Lidocaine 2.% with prilocaine 2.% oil in water eutectic mixture Lidocaine 4% liposomal cream LET gel
25 Topicals Intact skin Open skin Occlusive dressing OOA 20 OOA 30 OOA 60 DOA 60 EMLA X X X X 4% X +/- X X LET X X DOA 120
26 TOPICALS Road rash Can make lido-lube Lidocaine 3-4 mg/kg (as1 or 2% solution for injection) Large tube of sterile surgical lubricant Use small emesis basin Mix in lubricant until the consistency is a thin gel Slather over area, wait 20 min, then scrub
27
28 Hazardous Drugs National Institute for Occupational Safety and Health (NIOSH) Goal: to prevent the occupational exposure to antineoplastic/hazardous drug in health care setting Recommend standard or universal precautions Chemotherapy, antivirals, hormones, bioengineered, miscellaneous
29 Hazardous Drugs Drug Source AHFS Acitretin Aldesleukin Ambrisentan Alefacept 6 Pharmacologic-therapeutic classification 88:04 Vitamin A 4, 24:12.92 Vasodilating agents, miscellaneous 84:92 Skin and mucous membrane agents, miscellaneous Alitretinoin 3,4, 84:92 Skin and mucous membrane agents, miscellaneous Altretamine Amsacrine 3, Not in AHFS (antineoplastic agent) Anastrozole 1, Arsenic trioxide Asparaginase Azacitidine 3, Azathioprine 2,3, 92:44 Immunosuppressant agents Bacillus Calmette-Guerin (BCG) 1,2,4 80:12 Vaccines Bendamustine HCl Bexarotene 2,3,4,
30 Bicalutamide Bleomycin Bortezomib Bosentan Busulfan Cabergoline agonists Capecitabine Carbamazepine Carboplatin Carmustine Cetrorelix acetate 1, 6 6 Chlorambucil Chloramphenicol 1, Choriogonadotropin alfa Cidofovir 3, Cisplatin Cladribine Clofarabine 6 Clonazepam Colchicine Cyclophosphamide 24:12.92 Vasodilating agents, miscellaneous 10:00 Antineoplastic agents 28: Ergot-derivative dopamine receptor 28:12.92 Anticonvulsants, miscellaneous 92:40 Gonadotropin-releasing hormone antagonists 8:12.08 Chloramphenicols 68:18 Gonadotropins 8:18.32 Nucleosides and nucleotides 28:12.08 Benzodiazepines 92:16 Antigout agents
31 Cyclosporin 1 Cytarabine Dacarbazine Dactinomycin Dasatinib 6 Daunorubicin HCl Decitibine 6 Degarelix Denileukin 3,4, Diethylstilbestrol Dinoprostone Docetaxel Doxorubicin Dronedarone HCl Dutasteride Entecavir 6 Entecavir 6 Epirubicin Ergonovine/methylergonovine Estradiol 1, Estramustine phosphate Estrogen-progestin combination 92:44 Immunosuppressive agents Not in AHFS (nonsteroidal synthetic estrogen) 6:00 Oxytocics 24:04.04 Antiarrythmics 92:08 -alpha reductase inhibitors 8:18.32 Nucleosides and nucleotides 8:18.32 Nucleosides and nucleotides 6:00 Oxytocics 68:16.04 Estrogens 68:12 Contraceptives
32 Estrogens, conjugated Estrogens, esterified Estrone Estropipate Etoposide Everolimus Exemestane Finasteride Floxuridine Fludarabine Fluorouracil Fluoxymesterone Flutamide Fulvestrant Ganciclovir Ganirelix acetate antagonists Gemcitabine Gemtuzumab ozogamicin Gonadotropin, chorionic Goserelin Hydroxyurea Idarubicin Ifosfamide 1, 1,3, 1,2, 68:16.04 Estrogens 68:16.04 Estrogens 68:16.04 Estrogens 68:16.04 Estrogens 10:00 Antineoplastic agents 10:00 Antineoplastic agents 92:08 -alpha reductase inhibitors 68:08 Androgens 8:18.32 Nucleosides and nucleotides 92:40 Gonadotropin-releasing hormone 1,3,4, 1,2, 68:18 Gonadotropins
33 Imatinib mesylate 1,3,4, Irinotecan HCl Ixabepilone Leflunomide 3, Lenalidomide 6 Letrozole 1, Leuprolide acetate 1,2, Lomustine Mechlorethamine Medroxyprogesterone acetat 6 Megestrol 1, Melphalan Menotropins Mercaptopurine Methotrexate Methyltestosterone Mifepristone Mitomycin Mitotane 1,4, Mitoxantrone HCl Mycophenolate mofetil 1,3, Mycophenolic acid Nafarelin 92:36 Disease-modifying antirheumatic agents 92:20 Biologic response modifiers 68:32 Progestins 68:18 Gonadotropins 68:08 Androgens 6:00 Oxytocics 92:44 Immunosuppressive agents 92:44 Immunosuppressive agents 68:18 Gonadotropins
34 Nelarabine 6 Nilotinib Nilutamide 1, Oxaliplatin 1,3,4, Oxcarbazepine Oxytocin Paclitaxel Palifermin 6 Paroxetine** 6, Pazopanib HCl Pegaspargase Pemetrexed 6 Pentamidine isethionate 1,2,3, Pentetate calcium trisodium Pentostatin Phenoxybenzamine HCl blocking agents Pipobroman 3, Plerixafor Podofilox agents (mitotic inhibitor) Podophyllum resin miscellaneous 28:12.92 Anticonvulsants, miscellaneous 6:00 Oxytocics 84:16 Cell stimulants and proliferants 28: Selective serotonin uptake inhibitors 8:30.92 Antiprotozoals, miscellaneous 6 Not in AHFS 12: Non-selective alpha-adrenergic Not in AHFS (antineoplastic agent) 20:16 Hematopoietic agents 84:92 Miscellaneous skin and mucous membrane 84:92 Skin and mucous membrane agents,
35 Pralatrexate Procarbazine Progesterone Progestins Raloxifene Rasagiline mesylate Ribavirin Risperidone Romidepsin Sirolimus Sorafenib Streptozocin Sunitinib malate Tacrolimus Tamoxifen Televancin Temozolomide Temsirolimus Teniposide Testolactone Testosterone Tetracycline HCl 6 1,2, , 1,2, 3,4, 68:32 Progestins 68:12 Contraceptives 68:16.12 Estrogen agonists-antagonists 28:36 Antiparkinsonian agents 8:18.32 Nucleosides and nucleotides 28: Atypical antipsychotics 92:44 Immunosuppressive agents 92:44 Immunosuppressive agents 8: Glycopeptides 68:08 Androgens 8:12.24 Tetracyclines
36 Thalidomide Thioguanine Thiotepa Topotecan Toremifene citrate Tretinoin Trifluridine Triptorelin Uracil mustard Valganciclovir Valproic acid/ divalproex Na Valrubicin Vidarabine Vigabatrin Vinblastine sulfate Vincristine sulfate Vinorelbine tartrate Vorinostat Zidovudine Ziprasidone HCl Zoledronic acid Zonisamide 1,3, 1, 1,2,3, 1,2, 3, 1,3, 1,2,3, 1,2, 6 1,2, 6 92:20 Biologic response modifiers 84:16 Cell stimulants and proliferants 2:04.20 Antivirals Not in AHFS (antineoplastic agent) 8:18.32 Nucleosides and nucleotides 28:12.92 Anticonvulsants, miscellaneous Not in AHFS 28:12.92 Anticonvulsants, miscellaneous 8:18:08 Antiretroviral agents 28: Atypical antipsychotics 92:24 Bone resorption inhibitors 28:12.92 Anticonvulsants, miscellaneous
37 HD Residue 2010 study drugs studied 3 hospitals % of pharmacy wipes contaminated 43% of nursing wipes contaminated Connor, TH. JOEM 10/2010
38
39
40 HD PEARLS OSHA has no standards but has generated 3 guidelines in 1986, 199, 1999 Employers have a general duty to provide a workplace free from recognized, serious hazards Requires training programs and worker protection
41 HD PEARLS Handle materials safely Use spill kits Label appropriately Line surfaces Bag for transport
42 HD PEARLS Wipe, don t spray spraying the vial may move the HD to other items or areas Put gloves on and take off carefully! Sanitize with wipes Gowns get contaminated- don t save! Saving may result in drug transfer
43 So, what do you know? Who needs to know it? Share what you know!
Objective: To provide a standard procedure for the recycling of unused medication and the disposal of medicines across all BCPFT Hospital sites.
 WARDS/DEPARTMENTS By: 0 01/09/019 1 of 3 Objective: To provide a standard procedure for the recycling of unused medication and the disposal of medicines across all BCPFT Hospital sites. Scope: All Black
WARDS/DEPARTMENTS By: 0 01/09/019 1 of 3 Objective: To provide a standard procedure for the recycling of unused medication and the disposal of medicines across all BCPFT Hospital sites. Scope: All Black
Hazardous Medication List
 Hazardous Medication List REDUCING OCCUPATIONAL EXPOSURE TO HAZARDOUS MEDICATION FOR ALL STAFF Table of Contents Hazardous Medication List Key Points... 1 Hazardous Medication List... 2 Special Handling
Hazardous Medication List REDUCING OCCUPATIONAL EXPOSURE TO HAZARDOUS MEDICATION FOR ALL STAFF Table of Contents Hazardous Medication List Key Points... 1 Hazardous Medication List... 2 Special Handling
 Exhibit B United States Patent Application 20020012663 Kind Code A1 Waksal, Harlan W. January 31, 2002 Treatment of refractory human tumors with epidermal growth factor receptor antagonists Abstract A
Exhibit B United States Patent Application 20020012663 Kind Code A1 Waksal, Harlan W. January 31, 2002 Treatment of refractory human tumors with epidermal growth factor receptor antagonists Abstract A
Guidelines on Chemotherapy-induced Nausea and Vomiting in Pediatric Cancer Patients
 Guidelines on Chemotherapy-induced Nausea Vomiting in Pediatric Cancer Patients COG Supportive Care Endorsed Guidelines Click here to see all the COG Supportive Care Endorsed Guidelines. DISCLAIMER For
Guidelines on Chemotherapy-induced Nausea Vomiting in Pediatric Cancer Patients COG Supportive Care Endorsed Guidelines Click here to see all the COG Supportive Care Endorsed Guidelines. DISCLAIMER For
MEDICAL NECESSITY GUIDELINE
 PAGE: 1 of 10 IMPORTANT REMINDER This Clinical Policy has been developed by appropriately experienced and licensed health care professionals based on a thorough review and consideration of generally accepted
PAGE: 1 of 10 IMPORTANT REMINDER This Clinical Policy has been developed by appropriately experienced and licensed health care professionals based on a thorough review and consideration of generally accepted
West of Scotland Cancer Network Guideline for Managing Chemotherapy Induced Nausea and Vomiting
 West of Scotland Cancer Network Guideline for Managing Chemotherapy Induced Nausea and Vomiting Definitions Acute nausea and vomiting Delayed nausea and vomiting Anticipatory nausea and vomiting Initial
West of Scotland Cancer Network Guideline for Managing Chemotherapy Induced Nausea and Vomiting Definitions Acute nausea and vomiting Delayed nausea and vomiting Anticipatory nausea and vomiting Initial
Primary malignant neoplasms, not lymphatic or hematopoietic. Secondary malignant neoplasms (i.e.metastatic) Malignant neoplasm, unknown site
 Supplementary Table 1. ICD-9-CM codes used to define cancer ICD-9 Diagnosis code 140.xx-172.xx 174.xx-195.xx 196.xx 198.xx 199.xx 200.xx-208.xx Description Primary malignant neoplasms, not lymphatic or
Supplementary Table 1. ICD-9-CM codes used to define cancer ICD-9 Diagnosis code 140.xx-172.xx 174.xx-195.xx 196.xx 198.xx 199.xx 200.xx-208.xx Description Primary malignant neoplasms, not lymphatic or
SCI. SickKids-Caribbean Initiative Enhancing Capacity for Care in Paediatric Cancer and Blood Disorders
 1.0 Introduction The (SCI) is a not-for-profit collaboration between the Hospital for Sick Children (SickKids), Toronto, Canada, and seven Caribbean health care institutions across six countries that strive
1.0 Introduction The (SCI) is a not-for-profit collaboration between the Hospital for Sick Children (SickKids), Toronto, Canada, and seven Caribbean health care institutions across six countries that strive
Guideline for Classification of the Acute Emetogenic Potential of Antineoplastic Medication in Pediatric Cancer Patients
 Guideline for Classification of the Acute Emetogenic Potential of Antineoplastic Medication in Pediatric Cancer Patients POGO Antineoplastic Induced Nausea and Vomiting Guideline Development Panel: L.
Guideline for Classification of the Acute Emetogenic Potential of Antineoplastic Medication in Pediatric Cancer Patients POGO Antineoplastic Induced Nausea and Vomiting Guideline Development Panel: L.
Use of Prophylactic Growth Factors and Antimicrobials in Elderly Patients with Cancer: A
 Supportive Care in Cancer Use of Prophylactic Growth Factors and Antimicrobials in Elderly Patients with Cancer: A Systematic Review of the Medicare Database Romina Sosa, Shuling Li, Julia T. Molony, Jiannong
Supportive Care in Cancer Use of Prophylactic Growth Factors and Antimicrobials in Elderly Patients with Cancer: A Systematic Review of the Medicare Database Romina Sosa, Shuling Li, Julia T. Molony, Jiannong
Medication Review. Cancer Chemotherapy Drugs. Pharmacy Technician Training Systems Passassured, LLC
 Medication Review Cancer Chemotherapy Drugs Pharmacy Technician Training Systems Passassured, LLC Medication Review, Cancer Chemotherapy Drugs PassAssured's Pharmacy Technician Training Program Medication
Medication Review Cancer Chemotherapy Drugs Pharmacy Technician Training Systems Passassured, LLC Medication Review, Cancer Chemotherapy Drugs PassAssured's Pharmacy Technician Training Program Medication
APPHON/ROPPHA Guideline for the Prevention and Management of Chemotherapy Induced Nausea and Vomiting in Children with Cancer
 APPHON/ROPPHA Guideline for the Prevention and Management of Chemotherapy Induced Nausea and Vomiting in Children with Cancer 5850/5980 University Avenue, PO Box 9700, Halifax, N.S. B3K 6R8 PEDIATRIC HEMATOLOGY/ONCOLOGY
APPHON/ROPPHA Guideline for the Prevention and Management of Chemotherapy Induced Nausea and Vomiting in Children with Cancer 5850/5980 University Avenue, PO Box 9700, Halifax, N.S. B3K 6R8 PEDIATRIC HEMATOLOGY/ONCOLOGY
Subject: Palonosetron Hydrochloride (Aloxi )
 09-J0000-87 Original Effective Date: 02/15/09 Reviewed: 07/09/14 Revised: 03/15/18 Subject: Palonosetron Hydrochloride (Aloxi ) THIS MEDICAL COVERAGE GUIDELINE IS NOT AN AUTHORIZATION, CERTIFICATION, EXPLANATION
09-J0000-87 Original Effective Date: 02/15/09 Reviewed: 07/09/14 Revised: 03/15/18 Subject: Palonosetron Hydrochloride (Aloxi ) THIS MEDICAL COVERAGE GUIDELINE IS NOT AN AUTHORIZATION, CERTIFICATION, EXPLANATION
Supplementary Figures
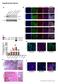 Supplementary Figures Supplementary Figure 1. Characterization of the generated hipsc lines a) Western blot analysis indicating the efficient downregulation of p53 upon knockdown in three hipsc lines (
Supplementary Figures Supplementary Figure 1. Characterization of the generated hipsc lines a) Western blot analysis indicating the efficient downregulation of p53 upon knockdown in three hipsc lines (
Description The following are synthetic cannabinoids requiring prior authorization: dronabinol (Marinol, Syndros ), nabilone (Cesamet )
 Clinical Policy: Nabilone (Cesamet), Dronabinol (Marinol, Syndros) Reference Number: CP.CPA.242 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important
Clinical Policy: Nabilone (Cesamet), Dronabinol (Marinol, Syndros) Reference Number: CP.CPA.242 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important
MASCC Guidelines for Antiemetic control: An update
 MASCC / ISOO 17 th International Symposium Supportive Care in Cancer June 30 July 2, 2005 / Geneva, Switzerland MASCC Guidelines for Antiemetic control: An update Sussanne Börjeson, RN, PhD Linköping University,
MASCC / ISOO 17 th International Symposium Supportive Care in Cancer June 30 July 2, 2005 / Geneva, Switzerland MASCC Guidelines for Antiemetic control: An update Sussanne Börjeson, RN, PhD Linköping University,
GUIDELINES FOR ANTIEMETIC USE IN ONCOLOGY SUMMARY CLASSIFICATION
 GUIDELINES FOR ANTIEMETIC USE IN ONCOLOGY SUMMARY More than half of all cancer patients experience nausea or vomiting during the course of their treatment. If nausea or vomiting becomes severe enough,
GUIDELINES FOR ANTIEMETIC USE IN ONCOLOGY SUMMARY More than half of all cancer patients experience nausea or vomiting during the course of their treatment. If nausea or vomiting becomes severe enough,
Objectives 5/19/2017. Hazardous Drugs Sunnybrook Health Sciences Centre s Experience. Hazardous Drug Committee at Sunnybrook
 s Sunnybrook Health Sciences Centre s Experience Romina Marchesano Drug Information Pharmacist Sunnybrook Health Sciences Centre Objectives Review: SHSC approach to NIOSHs list of s and USP / NAPRA
s Sunnybrook Health Sciences Centre s Experience Romina Marchesano Drug Information Pharmacist Sunnybrook Health Sciences Centre Objectives Review: SHSC approach to NIOSHs list of s and USP / NAPRA
1 The Cancer Programs Regulation (AR 242/98) is amended by this Regulation.
 Alberta Regulation 18/2005 Cancer Programs Act AMENDMENT REGULATION Filed: February 22, 2005 For information only: Made by the Minister of Health and Wellness (M.O. 9/2005) on February 17, 2005 pursuant
Alberta Regulation 18/2005 Cancer Programs Act AMENDMENT REGULATION Filed: February 22, 2005 For information only: Made by the Minister of Health and Wellness (M.O. 9/2005) on February 17, 2005 pursuant
DRUG PROPERTIES YOU NEED TO KNOW
 jfitzake@d.umn.edu www.d.umn.edu/~jfitzake Page 1 of 8 DRUG PROPERTIES YOU NEED TO KNOW 1. Mechanism of action a. chemical class b. resistance 2. Pharmacokinetics 3. Therapeutic uses 4. Major side effects/toxicities
jfitzake@d.umn.edu www.d.umn.edu/~jfitzake Page 1 of 8 DRUG PROPERTIES YOU NEED TO KNOW 1. Mechanism of action a. chemical class b. resistance 2. Pharmacokinetics 3. Therapeutic uses 4. Major side effects/toxicities
ICON Formulary - October 2018 Legend - ICON Protocols Essential (previously Standard), Core, Enhanced Core, Enhanced Enhanced
 ICON Formulary - October 2018 Legend - ICON Protocols Essential (previously Standard), Core, Enhanced Core, Enhanced Enhanced Class Medicine Name Nappi Strength Form Size Route Abiraterone Acetate ZYTIGA
ICON Formulary - October 2018 Legend - ICON Protocols Essential (previously Standard), Core, Enhanced Core, Enhanced Enhanced Class Medicine Name Nappi Strength Form Size Route Abiraterone Acetate ZYTIGA
Vivida Health Specialty Pharmacy Drugs (Injectable) Prior-Authorization Requirements Effective 1/1/19
 Vivida Health Specialty Pharmacy Drugs (Injectable) Prior-Authorization Requirements Effective 1/1/19 All Non-Par Provider Requests Requires Authorization Regardless of Service J0178 J0180 J0202 J0205
Vivida Health Specialty Pharmacy Drugs (Injectable) Prior-Authorization Requirements Effective 1/1/19 All Non-Par Provider Requests Requires Authorization Regardless of Service J0178 J0180 J0202 J0205
Adverse effects of anticancer drugs (Antimetabolites agents, Alkylating agents, Antimicrotubule agents, Miscellaneous agents, Immune therapies and
 35 Adverse effects of anticancer drugs (Antimetabolites agents, Alkylating agents, Antimicrotubule agents, Miscellaneous agents, Immune therapies and Biologically directed therapies ) 1 1- Nausea and vomiting
35 Adverse effects of anticancer drugs (Antimetabolites agents, Alkylating agents, Antimicrotubule agents, Miscellaneous agents, Immune therapies and Biologically directed therapies ) 1 1- Nausea and vomiting
Guidelines for the Use of Anti-Emetics with Chemotherapy
 Guidelines for the Use of Anti-Emetics with The purpose of this document is to provide guidance on the rational use of anti-emetics for prevention and treatment of chemotherapy-induced nausea and vomiting
Guidelines for the Use of Anti-Emetics with The purpose of this document is to provide guidance on the rational use of anti-emetics for prevention and treatment of chemotherapy-induced nausea and vomiting
Managements of Chemotherpay Induded Nausea and Vomiting
 REVIEW ARTICLE Managements of Chemotherpay Induded Nausea and Vomiting Department of Surgery, The Catholic University of Korea Sung Geun Kim 23 24 Sung Geun Kim Korean Journal of Clinical Oncology Summer
REVIEW ARTICLE Managements of Chemotherpay Induded Nausea and Vomiting Department of Surgery, The Catholic University of Korea Sung Geun Kim 23 24 Sung Geun Kim Korean Journal of Clinical Oncology Summer
Guideline Update on Antiemetics
 Guideline Update on Antiemetics Clinical Practice Guideline Special Announcements Please check www.asco.org/guidelines/antiemetics for current FDA alert(s) and safety announcement(s) on antiemetics 2 Introduction
Guideline Update on Antiemetics Clinical Practice Guideline Special Announcements Please check www.asco.org/guidelines/antiemetics for current FDA alert(s) and safety announcement(s) on antiemetics 2 Introduction
Emetogenicity level 1. Emetogenicity level 2
 Emetogenicity level 1 15 mins Pre-Chemo Maxalon 10mg po During chemo and Post Chemo 3 days Maxalon10mg po 8 hourly Increase Maxalon 20mg po 8 hourly Change to Cyclizine 50mg po 8 hourly 3 days If nausea
Emetogenicity level 1 15 mins Pre-Chemo Maxalon 10mg po During chemo and Post Chemo 3 days Maxalon10mg po 8 hourly Increase Maxalon 20mg po 8 hourly Change to Cyclizine 50mg po 8 hourly 3 days If nausea
Formulary Chemotherapy Agents: (Current as of 6/2018) Therapeutic Class
 MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY Cancer LAST REVIEW 9/11/18 THERAPEUTIC CLASS Oncology REVIEW HISTORY 5/17, 5/16 LOB AFFECTED Medi-Cal (MONTH/YEAR) This policy
MEDICATION COVERAGE POLICY PHARMACY AND THERAPEUTICS ADVISORY COMMITTEE POLICY Cancer LAST REVIEW 9/11/18 THERAPEUTIC CLASS Oncology REVIEW HISTORY 5/17, 5/16 LOB AFFECTED Medi-Cal (MONTH/YEAR) This policy
DRUG PROPERTIES YOU NEED TO KNOW
 Dr. Janet Fitzakerley Summer 2013 Med 6541 Hematopoiesis and Host Defences jfitzake@d.umn.edu www.d.umn.edu/~jfitzake Page 1 of 11 DRUG PROPERTIES YOU NEED TO KNOW 1. Mechanism of action a. chemical class
Dr. Janet Fitzakerley Summer 2013 Med 6541 Hematopoiesis and Host Defences jfitzake@d.umn.edu www.d.umn.edu/~jfitzake Page 1 of 11 DRUG PROPERTIES YOU NEED TO KNOW 1. Mechanism of action a. chemical class
Current and Emerging Therapeutic Options in the Management of Chemotherapy-Induced Nausea and Vomiting (CINV) Objectives
 Current and Emerging Therapeutic Options in the Management of Chemotherapy-Induced Nausea and Vomiting (CINV) Susan Urba, M.D. University of Michigan Comprehensive Cancer Center Objectives Mechanisms of
Current and Emerging Therapeutic Options in the Management of Chemotherapy-Induced Nausea and Vomiting (CINV) Susan Urba, M.D. University of Michigan Comprehensive Cancer Center Objectives Mechanisms of
Subject: Fosnetupitant-Palonosetron (Akynzeo) IV
 09-J3000-01 Original Effective Date: 06/15/18 Reviewed: 05/09/18 Revised: 01/01/19 Subject: Fosnetupitant-Palonosetron (Akynzeo) IV THIS MEDICAL COVERAGE GUIDELINE IS NOT AN AUTHORIZATION, CERTIFICATION,
09-J3000-01 Original Effective Date: 06/15/18 Reviewed: 05/09/18 Revised: 01/01/19 Subject: Fosnetupitant-Palonosetron (Akynzeo) IV THIS MEDICAL COVERAGE GUIDELINE IS NOT AN AUTHORIZATION, CERTIFICATION,
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: Reference Number: CP.CPA.223 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy for important regulatory
Clinical Policy: Reference Number: CP.CPA.223 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Commercial Revision Log See Important Reminder at the end of this policy for important regulatory
DOXOrubicin, Cyclophosphamide (AC 60/600) 21 day followed by weekly PACLitaxel (80) Therapy (AC-T) 261 CARBOplatin (AUC4-6) Monotherapy-21 days
 Last updated Oct 17, 2018 Tumour Group Protocol Number Protocol Name on NCCP website Breast 200 Trastuzumab (IV) Monotherapy 21 days 201 Trastuzumab (IV) Monotherapy 7 days 202 DOCEtaxel Monotherapy 100mg/m2
Last updated Oct 17, 2018 Tumour Group Protocol Number Protocol Name on NCCP website Breast 200 Trastuzumab (IV) Monotherapy 21 days 201 Trastuzumab (IV) Monotherapy 7 days 202 DOCEtaxel Monotherapy 100mg/m2
1 17 ACITRETIN 10MG CAP 20, ,000 14,000 4, ACITRETIN 25MG CAP 50, ,000 35,000 10,000
 ردیف کد ژنریک نام ژنریک مبلغ پوشش وزارت بهداشت مبلغ پوشش بیمه مبلغ پرداخت بیمار درصد پرداخت بیمار قیمت اعالمی وزارت بهداشت نام تجاری 1 17 ACITRETIN 10MG CAP 20,000 10.0 2,000 14,000 4,000 2 18 ACITRETIN
ردیف کد ژنریک نام ژنریک مبلغ پوشش وزارت بهداشت مبلغ پوشش بیمه مبلغ پرداخت بیمار درصد پرداخت بیمار قیمت اعالمی وزارت بهداشت نام تجاری 1 17 ACITRETIN 10MG CAP 20,000 10.0 2,000 14,000 4,000 2 18 ACITRETIN
Part B payment for drugs in Medicare 0
 Part B payment for drugs in Medicare 0 Introduction: The recent pilot proposal 1 on Part B drug payment from the Center for Medicare and Medicaid Innovation of CMS has met strong resistance 2 from the
Part B payment for drugs in Medicare 0 Introduction: The recent pilot proposal 1 on Part B drug payment from the Center for Medicare and Medicaid Innovation of CMS has met strong resistance 2 from the
Prevention and Management of chemo-and radiotherapy-induced nausea and vomiting
 Prevention and Management of chemo-and radiotherapy-induced nausea and vomiting Focusing on the updated MASCC/ESMO guidelines Karin Jordan Department of Hematology and Oncology, University of Heidelberg
Prevention and Management of chemo-and radiotherapy-induced nausea and vomiting Focusing on the updated MASCC/ESMO guidelines Karin Jordan Department of Hematology and Oncology, University of Heidelberg
DRUGS YOU NEED TO KNOW
 jfitzake@d.umn.edu Page 1 of 7 DRUGS YOU NEED TO KNOW ALKYLATING AGENTS (blue cards) BUSULFAN CARMUSTINE (BCNU) CYCLOPHOSPHAMIDE DACARBAZINE LOMUSTINE (CCNU) MECHLORETHAMINE MELPHALAN THIOTEPA NATURAL
jfitzake@d.umn.edu Page 1 of 7 DRUGS YOU NEED TO KNOW ALKYLATING AGENTS (blue cards) BUSULFAN CARMUSTINE (BCNU) CYCLOPHOSPHAMIDE DACARBAZINE LOMUSTINE (CCNU) MECHLORETHAMINE MELPHALAN THIOTEPA NATURAL
DRUG EXTRAVASATION. Vesicants. Irritants
 DRUG EXTRAVASATION Vesicants Irritants Vesicants Antineoplastic drugs Amsacrine Dactinomycin Daunorubicin Docetaxel (rare) Doxorubicin Epirubicin Idarubicin Mechlorethamine Mitomycin Oxaliplatin (rare)
DRUG EXTRAVASATION Vesicants Irritants Vesicants Antineoplastic drugs Amsacrine Dactinomycin Daunorubicin Docetaxel (rare) Doxorubicin Epirubicin Idarubicin Mechlorethamine Mitomycin Oxaliplatin (rare)
Part B payment for drugs in Medicare: Phase 1 of CMS s proposed pilot and its impact on oncology care
 Part B payment for drugs in Medicare: Phase 1 of CMS s proposed pilot and its impact on oncology care Raina H. Jain, Stephen M. Schleicher, Coral L. Atoria, Peter B. Bach Executive Summary The recent pilot
Part B payment for drugs in Medicare: Phase 1 of CMS s proposed pilot and its impact on oncology care Raina H. Jain, Stephen M. Schleicher, Coral L. Atoria, Peter B. Bach Executive Summary The recent pilot
Exposure Control Plan for Cytotoxic Substances
 Exposure Control Plan Department of www.rms.ubc.ca UBC-RMS-OHS-ECP-17-001 Effective date: December 8, 2017 Review date: NA Supersedes: NA 1. PURPOSE Exposure Control Plan for Cytotoxic Substances The University
Exposure Control Plan Department of www.rms.ubc.ca UBC-RMS-OHS-ECP-17-001 Effective date: December 8, 2017 Review date: NA Supersedes: NA 1. PURPOSE Exposure Control Plan for Cytotoxic Substances The University
AD regimen, 576 adrenal steroid inhibitor. aminoglutethimide, 23 25
 A 640 Index Index abiraterone acetate, 5 7 Abraxane. See albuminbound paclitaxel ABVD regimen, 57, 540 ABV regimen, 519 520 AC docetaxel regimen, 476, 478 acetaminophen busulfan and, 71 AC regimen, 475,
A 640 Index Index abiraterone acetate, 5 7 Abraxane. See albuminbound paclitaxel ABVD regimen, 57, 540 ABV regimen, 519 520 AC docetaxel regimen, 476, 478 acetaminophen busulfan and, 71 AC regimen, 475,
COST CONSIDERATIONS Union for International Cancer Control 2014 Review of Cancer Medicines on the WHO List of Essential Medicines!!!!!!!!!
 UICCEMLCostingScenarios BackoftheEnvelope Calculations PreparedforWorkingGroupSession:19621November2014,Geneva MethodsSummary We have chosen a conservative approach, calculating cost per vial. We have
UICCEMLCostingScenarios BackoftheEnvelope Calculations PreparedforWorkingGroupSession:19621November2014,Geneva MethodsSummary We have chosen a conservative approach, calculating cost per vial. We have
See Important Reminder at the end of this policy for important regulatory and legal information.
 Clinical Policy: (Zofran, Zuplenz) Reference Number: CP.CPA.173 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Medicaid Medi-Cal Revision Log See Important Reminder at the end of this
Clinical Policy: (Zofran, Zuplenz) Reference Number: CP.CPA.173 Effective Date: 11.16.16 Last Review Date: 11.17 Line of Business: Medicaid Medi-Cal Revision Log See Important Reminder at the end of this
Part B payment for drugs in Medicare 0
 Part B payment for drugs in Medicare 0 Introduction: The recent pilot proposal 1 on Part B drug payment from the Center for Medicare and Medicaid Innovation of CMS has met strong resistance 2 from the
Part B payment for drugs in Medicare 0 Introduction: The recent pilot proposal 1 on Part B drug payment from the Center for Medicare and Medicaid Innovation of CMS has met strong resistance 2 from the
Subject: NK-1 receptor antagonist injectable therapy (Emend, Cinvanti, Varubi )
 09-J2000-60 Original Effective Date: 06/15/16 Reviewed: 04/11/18 Revised: 01/01/19 Subject: NK-1 receptor antagonist injectable therapy (Emend, Cinvanti, Varubi ) THIS MEDICAL COVERAGE GUIDELINE IS NOT
09-J2000-60 Original Effective Date: 06/15/16 Reviewed: 04/11/18 Revised: 01/01/19 Subject: NK-1 receptor antagonist injectable therapy (Emend, Cinvanti, Varubi ) THIS MEDICAL COVERAGE GUIDELINE IS NOT
ANTIEMETICS UTILIZATION MANAGEMENT CRITERIA
 ANTIEMETICS [Akynzeo, Anzemet, Cesamet, Emend, Sancuso, Varubi, Zuplenz ] DRUG CLASS: UTILIZATION MANAGEMENT CRITERIA 5-HT 3 Receptor Antagonists 5-HT 3 Receptor Antagonist and Substance P/Neurokinin (NK
ANTIEMETICS [Akynzeo, Anzemet, Cesamet, Emend, Sancuso, Varubi, Zuplenz ] DRUG CLASS: UTILIZATION MANAGEMENT CRITERIA 5-HT 3 Receptor Antagonists 5-HT 3 Receptor Antagonist and Substance P/Neurokinin (NK
Committee Approval Date: December 12, 2014 Next Review Date: July 2015
 Medication Policy Manual Policy No: dru378 Topic: Akynzeo, netupitant/palonosetron Date of Origin: December 12, 2014 Committee Approval Date: December 12, 2014 Next Review Date: July 2015 Effective Date:
Medication Policy Manual Policy No: dru378 Topic: Akynzeo, netupitant/palonosetron Date of Origin: December 12, 2014 Committee Approval Date: December 12, 2014 Next Review Date: July 2015 Effective Date:
Acute Lymphocytic Leukemia
 Acute Lymphocytic Leukemia Splenectomy (Removal of Spleen) 6-Mercaptopurine (Purinethol, 6-MP) Alemtuzumab (Campath ) Arsenic Trioxide (Trisenox ) Bendamustine (Treanda ) Bexarotene (Targretin ) Bleomycin
Acute Lymphocytic Leukemia Splenectomy (Removal of Spleen) 6-Mercaptopurine (Purinethol, 6-MP) Alemtuzumab (Campath ) Arsenic Trioxide (Trisenox ) Bendamustine (Treanda ) Bexarotene (Targretin ) Bleomycin
1. Purpose Documentation Description.1 4. References 8 5. Cross-References.8 6. Development of the guideline.8
 CLINICAL GUIDELINE For use in (clinical areas): For use by (staff groups): For use for (patients): Document owner: Status: Oncology/Haematology Unit (excluding Paediatrics) Oncologists, Haematologists,
CLINICAL GUIDELINE For use in (clinical areas): For use by (staff groups): For use for (patients): Document owner: Status: Oncology/Haematology Unit (excluding Paediatrics) Oncologists, Haematologists,
ELECTRONIC HEALTH RECORD (EHR) ENHANCEMENTS FOR MARCH 15, 2016 SUMMARY
 ELECTRONIC HEALTH RECORD (EHR) ENHANCEMENTS FOR MARCH 15, 2016 SUMMARY Problem Opening PACS Images on ipads or ibooks has Been Fixed Changes have been made in PROD to enable user credentials to be passed
ELECTRONIC HEALTH RECORD (EHR) ENHANCEMENTS FOR MARCH 15, 2016 SUMMARY Problem Opening PACS Images on ipads or ibooks has Been Fixed Changes have been made in PROD to enable user credentials to be passed
Clinical Tools and Resources for Self-Study and Patient Education
 Clinical Tools and Resources for Self-Study and Patient Education CHEMOTHERAPY-INDUCED NAUSEA AND VOMITING CLINICIAN'S RESOURCE GUIDE The clinical tools and resources contained herein are provided as educational
Clinical Tools and Resources for Self-Study and Patient Education CHEMOTHERAPY-INDUCED NAUSEA AND VOMITING CLINICIAN'S RESOURCE GUIDE The clinical tools and resources contained herein are provided as educational
Protocol Number Tumour Group Protocol Name on NCCP website 22/02/ Lung Afatinib Monotherapy 244 Gastrointestinal Regorafenib Monotherapy
 Last Updated 22-Feb-18 Date of last update Protocol Number Tumour Group Protocol Name on NCCP website 22/02/2018 221 Afatinib Monotherapy 244 Gastrointestinal Regorafenib Monotherapy 249 Gynaecology Intrathecal
Last Updated 22-Feb-18 Date of last update Protocol Number Tumour Group Protocol Name on NCCP website 22/02/2018 221 Afatinib Monotherapy 244 Gastrointestinal Regorafenib Monotherapy 249 Gynaecology Intrathecal
Protocol Number Intrathecal Methotrexate for CNS 01/02/2018 Prophylaxis in GTN Gynaecology 249
 Last updated Feb 9, 2018 Revision due Protocol Name on NCCP website Tumour Group Protocol Number Intrathecal Methotrexate for CNS 01/02/2018 Prophylaxis in GTN Gynaecology 249 Two Day Etoposide CISplatin
Last updated Feb 9, 2018 Revision due Protocol Name on NCCP website Tumour Group Protocol Number Intrathecal Methotrexate for CNS 01/02/2018 Prophylaxis in GTN Gynaecology 249 Two Day Etoposide CISplatin
Chemotherapy 101 for Radiation Oncology Workers
 Chemotherapy 101 for Radiation Oncology Workers James Sinclair, M.D. CCARE/Medical Director Scripps Cancer Center March 9, 2013 Category 1) Alkylating agents Alkylating agents directly damage DNA to prevent
Chemotherapy 101 for Radiation Oncology Workers James Sinclair, M.D. CCARE/Medical Director Scripps Cancer Center March 9, 2013 Category 1) Alkylating agents Alkylating agents directly damage DNA to prevent
J1556 INJECTION, IMMUNE GLOBULIN (BIVIGAM) 500 MG $ J1559 INJECTION, IMMUNE GLOBULIN (HIZENTRA) 100 MG $14.364
 G0333 INITIAL 30-DAY SUPPLY AS A BENEFICIARY $57.000 J0133 INJECTION, ACYCLOVIR 5 MG $0.470 J0285 INJECTION, AMPHOTERICIN B 50 MG $10.280 J0287 INJECTION, AMPHOTERICIN B LIPID COMPLEX 10 MG $21.850 J0288
G0333 INITIAL 30-DAY SUPPLY AS A BENEFICIARY $57.000 J0133 INJECTION, ACYCLOVIR 5 MG $0.470 J0285 INJECTION, AMPHOTERICIN B 50 MG $10.280 J0287 INJECTION, AMPHOTERICIN B LIPID COMPLEX 10 MG $21.850 J0288
OVERVIEW OF COMMENTS RECEIVED ON LIST OF PAEDIATRIC NEEDS ONCOLOGY I (CYTOTOXIC THERAPY)
 European Medicines Agency Evaluation of Medicines for Human Use London, September 2006 Doc. Ref. OVERVIEW OF COMMENTS RECEIVED ON LIST OF PAEDIATRIC NEEDS ONCOLOGY I (CYTOTOXIC THERAPY) Table 1: Organisations
European Medicines Agency Evaluation of Medicines for Human Use London, September 2006 Doc. Ref. OVERVIEW OF COMMENTS RECEIVED ON LIST OF PAEDIATRIC NEEDS ONCOLOGY I (CYTOTOXIC THERAPY) Table 1: Organisations
CHEMOTHERAPY/ ANTICANCER DRUGS/ NEOPLASTIC AGENTS
 CHEMOTHERAPY/ 1 Oncology, Misc, Vaccines ANTICANCER DRUGS/ NEOPLASTIC AGENTS Important Terms: Cancer: A group of diseases involving abnormal cell growth with the potential to invade or spread to other
CHEMOTHERAPY/ 1 Oncology, Misc, Vaccines ANTICANCER DRUGS/ NEOPLASTIC AGENTS Important Terms: Cancer: A group of diseases involving abnormal cell growth with the potential to invade or spread to other
Antiemesis. NCCN Clinical Practice Guidelines in Oncology. Antiemesis. Version Continue
 Table of Contents Clinical in Oncology Version 2.2010 Continue www.nccn.org Version 2.2010, 04/07/2010 2010 National Comprehensive Cancer Network, Inc. All rights reserved. These guidelines and this illustration
Table of Contents Clinical in Oncology Version 2.2010 Continue www.nccn.org Version 2.2010, 04/07/2010 2010 National Comprehensive Cancer Network, Inc. All rights reserved. These guidelines and this illustration
The AngCN Antiemetic Guidelines
 The AngCN Antiemetic Guidelines Guidelines for the Management of Nausea and Vomiting in Adult Patients Receiving Chemotherapy and/or Radiotherapy AngCN Document Reference: AngCN-CCG-C31 CONTENTS 1.0 Introduction
The AngCN Antiemetic Guidelines Guidelines for the Management of Nausea and Vomiting in Adult Patients Receiving Chemotherapy and/or Radiotherapy AngCN Document Reference: AngCN-CCG-C31 CONTENTS 1.0 Introduction
Adult Intravenous Systemic Anticancer Therapy (SACT) Section A. SUMMARY of SCHEME QIPP Reference
 CA2 Nationally standardised Dose banding for Adult Intravenous Anticancer Therapy (SACT) Scheme Name CA2: Nationally Standardised Dose Banding for Adult Intravenous Systemic Anticancer Therapy (SACT) Section
CA2 Nationally standardised Dose banding for Adult Intravenous Anticancer Therapy (SACT) Scheme Name CA2: Nationally Standardised Dose Banding for Adult Intravenous Systemic Anticancer Therapy (SACT) Section
through the cell cycle. However, how we administer drugs also depends on the combinations that we give and the doses that we give.
 Hello and welcome to this lecture. My name is Hillary Prescott. I am a Clinical Pharmacy Specialist at The University of Texas MD Anderson Cancer Center. My colleague, Jeff Bryan and I have prepared this
Hello and welcome to this lecture. My name is Hillary Prescott. I am a Clinical Pharmacy Specialist at The University of Texas MD Anderson Cancer Center. My colleague, Jeff Bryan and I have prepared this
DME MAC Jurisdiction C Drug Fees, Pharmacy Dispensing Fees and Pharmacy Supply Fees Effective 01/01/2018 through 03/31/2018
 G0333 PHARMACY DISPENSING FEE FOR INHALATION DRUG(S); INITIAL 30-DAY SUPPLY AS A BENEFICIARY $57.000 J0133 INJECTION, ACYCLOVIR 5 MG $0.068 J0285 INJECTION, AMPHOTERICIN B 50 MG $32.987 J0287 INJECTION,
G0333 PHARMACY DISPENSING FEE FOR INHALATION DRUG(S); INITIAL 30-DAY SUPPLY AS A BENEFICIARY $57.000 J0133 INJECTION, ACYCLOVIR 5 MG $0.068 J0285 INJECTION, AMPHOTERICIN B 50 MG $32.987 J0287 INJECTION,
DME MAC Jurisdiction B Drug Fees, Pharmacy Dispensing Fees and Pharmacy Supply Fees Effective 01/01/2019 through 03/31/2019
 G0333 PHARMACY DISPENSING FEE FOR INHALATION DRUG(S); INITIAL 30-DAY SUPPLY AS A BENEFICIARY $57.000 J0133 INJECTION, ACYCLOVIR 5 MG $0.048 J0285 INJECTION, AMPHOTERICIN B 50 MG $31.668 J0287 INJECTION,
G0333 PHARMACY DISPENSING FEE FOR INHALATION DRUG(S); INITIAL 30-DAY SUPPLY AS A BENEFICIARY $57.000 J0133 INJECTION, ACYCLOVIR 5 MG $0.048 J0285 INJECTION, AMPHOTERICIN B 50 MG $31.668 J0287 INJECTION,
FOR A HIGHER DEGREE OF CONFIDENCE
 SELECTING THE RIGHT HAND PROTECTION WHEN WORKING WITH CHEMOTHERAPY DRUGS. > Introduction Manufacturing specialized products in a pharmaceutical manufacturing environment has a specific set of requirements
SELECTING THE RIGHT HAND PROTECTION WHEN WORKING WITH CHEMOTHERAPY DRUGS. > Introduction Manufacturing specialized products in a pharmaceutical manufacturing environment has a specific set of requirements
Job title: Consultant Pharmacist/Advanced Practice Pharmacist
 Title : Guidelines for the Use of Antiemetics Purpose: To provide trust-wide guidance on the safe and effective use of antiemetics for the prevention and treatment of chemotherapy and radiotherapy induced
Title : Guidelines for the Use of Antiemetics Purpose: To provide trust-wide guidance on the safe and effective use of antiemetics for the prevention and treatment of chemotherapy and radiotherapy induced
REVIEW Guideline for the Classification of the Acute Emetogenic Potential of Antineoplastic Medication in Pediatric Cancer Patients
 Pediatr Blood Cancer 2011;57:191 198 REVIEW Guideline for the Classification of the Acute Emetogenic Potential of Antineoplastic Medication in Pediatric Cancer Patients L. Lee Dupuis, MScPhm, ACPR, FCSHP,
Pediatr Blood Cancer 2011;57:191 198 REVIEW Guideline for the Classification of the Acute Emetogenic Potential of Antineoplastic Medication in Pediatric Cancer Patients L. Lee Dupuis, MScPhm, ACPR, FCSHP,
VI.2 Elements for a Public Summary VI.2.1 Overview of Disease Epidemiology Acute Nausea and Vomiting (N&V) Etiologies:
 VI.2 Elements for a Public Summary VI.2.1 Overview of Disease Epidemiology Acute Nausea and Vomiting (N&V) Incidence: The incidence of acute and delayed N&V was investigated in highly and moderately emetogenic
VI.2 Elements for a Public Summary VI.2.1 Overview of Disease Epidemiology Acute Nausea and Vomiting (N&V) Incidence: The incidence of acute and delayed N&V was investigated in highly and moderately emetogenic
Corporate Medical Policy
 Antiemetic Injection Therapy Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: antiemetic_injection_therapy 5/2015 3/2017 3/2018 3/2017 Description of Procedure
Antiemetic Injection Therapy Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: antiemetic_injection_therapy 5/2015 3/2017 3/2018 3/2017 Description of Procedure
Prevention and Management of cancer disease and of chemo-and radiotherapyinduced nausea and vomiting
 Prevention and Management of cancer disease and of chemo-and radiotherapyinduced nausea and vomiting Focusing on the updated MASCC/ESMO guidelines Karin Jordan Department of Hematology and Oncology, University
Prevention and Management of cancer disease and of chemo-and radiotherapyinduced nausea and vomiting Focusing on the updated MASCC/ESMO guidelines Karin Jordan Department of Hematology and Oncology, University
Trust Guideline for Prevention and Control of Chemotherapy and Radiotherapy Induced Nausea and Vomiting in Adults
 A Clinical Guideline For Use in: Organisation-wide By: For: Key words: Name of document author: Job title of document author: Name of document author s Line Manager: Job title of author s Line Manager:
A Clinical Guideline For Use in: Organisation-wide By: For: Key words: Name of document author: Job title of document author: Name of document author s Line Manager: Job title of author s Line Manager:
ASSESSMENT OF THE PAEDIATRIC NEEDS CHEMOTHERAPY PRODUCTS (PART I) DISCLAIMER
 European Medicines Agency Evaluation of Medicines for Human Use London, September 2006 Doc. Ref.: EMEA/384641/2006 ASSESSMENT OF THE PAEDIATRIC NEEDS CHEMOTHERAPY PRODUCTS (PART I) DISCLAIMER The Paediatric
European Medicines Agency Evaluation of Medicines for Human Use London, September 2006 Doc. Ref.: EMEA/384641/2006 ASSESSMENT OF THE PAEDIATRIC NEEDS CHEMOTHERAPY PRODUCTS (PART I) DISCLAIMER The Paediatric
Northern Cancer Alliance
 Northern Cancer Alliance Anti-emetic Guidelines for Chemotherapy Induced Nausea and Vomiting (CINV) Adult Oncology & Haematology Document Control Document Title: Antiemetic Guidelines for CINV NESCN v2.2
Northern Cancer Alliance Anti-emetic Guidelines for Chemotherapy Induced Nausea and Vomiting (CINV) Adult Oncology & Haematology Document Control Document Title: Antiemetic Guidelines for CINV NESCN v2.2
Patient 1: Patient 2:
 Appendix A Compiled by Dr. Raymond Ngeh and Dr. Robert Luk Clinical notes and PET/CT scan images of eleven patients: 1. Middle age woman has cancer of the pancreas in the body of the gland. After just
Appendix A Compiled by Dr. Raymond Ngeh and Dr. Robert Luk Clinical notes and PET/CT scan images of eleven patients: 1. Middle age woman has cancer of the pancreas in the body of the gland. After just
NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines ) Antiemesis. Version NCCN.org. Continue
 NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines ) Version 1.2011 NCCN.org Continue Note: All recommendations are category 2A unless otherwise indicated. Clinical Trials: NCCN believes that
NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines ) Version 1.2011 NCCN.org Continue Note: All recommendations are category 2A unless otherwise indicated. Clinical Trials: NCCN believes that
The following are J Code requirements
 The following are J Code requirements J Codes 20610 Arthrocentesis, aspiration and/or injection; major joint or bursa (eg, shoulder, hip, knee joint, subacromial bursa) A9579 Injection, gadolinium based
The following are J Code requirements J Codes 20610 Arthrocentesis, aspiration and/or injection; major joint or bursa (eg, shoulder, hip, knee joint, subacromial bursa) A9579 Injection, gadolinium based
Product Name Strength Dosage Form Therapeutic Area Dossier Status. Aceclofenac 100mg Film coated Tablets Analgesia-inflammation Approved
 Product Name Strength Dosage Form Therapeutic Area Dossier Status Aceclofenac 100mg Film coated Tablets Analgesia-inflammation Approved Alendronate Sodium 10/70mg Tablets Osteoporosis Approved Anastrozole
Product Name Strength Dosage Form Therapeutic Area Dossier Status Aceclofenac 100mg Film coated Tablets Analgesia-inflammation Approved Alendronate Sodium 10/70mg Tablets Osteoporosis Approved Anastrozole
Chemotherapy Induced Nausea and Vomiting (CINV) Anti-emetic Guidelines
 North of England Cancer Network Chemotherapy Induced Nausea and Vomiting (CINV) Anti-emetic Guidelines Adult Oncology & Haematology Quality and safety for every patient every time For more information
North of England Cancer Network Chemotherapy Induced Nausea and Vomiting (CINV) Anti-emetic Guidelines Adult Oncology & Haematology Quality and safety for every patient every time For more information
Hodgkin and Non-Hodgkin Lymphoma (LYM) Post-Infusion Data
 Hodgkin and Non-Hodgkin Lymphoma (LYM) Post-Infusion Data Registry Use Only Sequence Number: Date Received: CIBMTR Center Number: CIBMTR Research ID: Event date: / / Visit 100 day 6 months 1 year 2 years
Hodgkin and Non-Hodgkin Lymphoma (LYM) Post-Infusion Data Registry Use Only Sequence Number: Date Received: CIBMTR Center Number: CIBMTR Research ID: Event date: / / Visit 100 day 6 months 1 year 2 years
Antiemetic Guidelines. Guidelines for the Management of Nausea and Vomiting in Adult Patients Receiving Chemotherapy and/or Radiotherapy
 Antiemetic Guidelines Guidelines for the Management of Nausea and Vomiting in Adult Patients Receiving Chemotherapy and/or Radiotherapy For approvals and version control see Document Management Record
Antiemetic Guidelines Guidelines for the Management of Nausea and Vomiting in Adult Patients Receiving Chemotherapy and/or Radiotherapy For approvals and version control see Document Management Record
Haematology, Oncology and Palliative Care Directorate.
 Anticancer Treatment for Administration on the Somerset Mobile Chemotherapy Unit The table below details the suitability of different types of anticancer treatment for administration on the Somerset Mobile
Anticancer Treatment for Administration on the Somerset Mobile Chemotherapy Unit The table below details the suitability of different types of anticancer treatment for administration on the Somerset Mobile
Standard Regimens for Haematology
 Regimens for Haematology ChlVPP Chlorambucil 6mg/m 2 PO D1 to 14 Vinblastine 6mg/m 2 (max 10mg) IV on D1 & 8 Procarbazine 100mg/m 2 PO on D1 to 14 Prednisolone 40mg PO D1 to 14 ABVD Doxorubicin 25mg/m
Regimens for Haematology ChlVPP Chlorambucil 6mg/m 2 PO D1 to 14 Vinblastine 6mg/m 2 (max 10mg) IV on D1 & 8 Procarbazine 100mg/m 2 PO on D1 to 14 Prednisolone 40mg PO D1 to 14 ABVD Doxorubicin 25mg/m
It is a malignancy originating from breast tissue
 59 Breast cancer 1 It is a malignancy originating from breast tissue including both early stages which are potentially curable, and metastatic breast cancer (MBC) which is usually incurable. Most breast
59 Breast cancer 1 It is a malignancy originating from breast tissue including both early stages which are potentially curable, and metastatic breast cancer (MBC) which is usually incurable. Most breast
Disclosure. Objectives. Objectives. Oncologic Emergency. Classification of Emergencies 1 3/2/2015
 Disclosure Oncologic Emergencies and Acute Supportive Care of the Critically Ill Oncology Patient By: Kimberly Regis, Pharm.D. Broward Health Medical Center Pharmacy Resident 2014-2015 I do not have a
Disclosure Oncologic Emergencies and Acute Supportive Care of the Critically Ill Oncology Patient By: Kimberly Regis, Pharm.D. Broward Health Medical Center Pharmacy Resident 2014-2015 I do not have a
ANTIEMETIC GUIDELINES: MASCC/ESMO
 Open Issues for CINV Do we reliably measure that? Do we control nausea optimally? Are guidelines useful for oral therapies related nausea and vomiting? Breakthrough and refractory nausea and vomiting:
Open Issues for CINV Do we reliably measure that? Do we control nausea optimally? Are guidelines useful for oral therapies related nausea and vomiting? Breakthrough and refractory nausea and vomiting:
SELECTING THE RIGHT HAND PROTECTION WHEN WORKING WITH CHEMOTHERAPY DRUGS
 SELECTING THE RIGHT HAND PROTECTION WHEN WORKING WITH CHEMOTHERAPY DRUGS SELECTING THE RIGHT HAND PROTECTION WHEN WORKING WITH CHEMOTHERAPY DRUGS Manufacturing specialized products in a pharmaceutical
SELECTING THE RIGHT HAND PROTECTION WHEN WORKING WITH CHEMOTHERAPY DRUGS SELECTING THE RIGHT HAND PROTECTION WHEN WORKING WITH CHEMOTHERAPY DRUGS Manufacturing specialized products in a pharmaceutical
SARASOTA MEMORIAL HOSPITAL
 SARASOTA MEMORIAL HOSPITAL TITLE: NURSING PROCEDURE DATE: REVIEWED: PAGES: 03/86 7/18 1 of 13 PS1094 ISSUED FOR: Nursing RESPONSIBILITY: RN Chemo Qualified PURPOSE: To provide the RN with specific steps
SARASOTA MEMORIAL HOSPITAL TITLE: NURSING PROCEDURE DATE: REVIEWED: PAGES: 03/86 7/18 1 of 13 PS1094 ISSUED FOR: Nursing RESPONSIBILITY: RN Chemo Qualified PURPOSE: To provide the RN with specific steps
HCPCS Code/ generic (Brand) Name J7506. J8520 capecitabine (Xeloda) 1. J8521 capecitabine. J8530 cyclophosphamide. (Cytoxan) 1
 drug and administration compendia Current Price () J7506 Prednisone, oral, per 5 mg 12/1/07 $0.07 $0.19 N/A prednisone 2 J8520 capecitabine (Xeloda) 1 Capecitabine, oral, 150 mg 8/1/07 $5.79 $4.59 N/A
drug and administration compendia Current Price () J7506 Prednisone, oral, per 5 mg 12/1/07 $0.07 $0.19 N/A prednisone 2 J8520 capecitabine (Xeloda) 1 Capecitabine, oral, 150 mg 8/1/07 $5.79 $4.59 N/A
Title Developed By. Implementation Contact person(s) Review Date Group Responsible. NICaN Policy: Management of Chemotherapy Extravasation; Version 3
 Title Developed By Management of Chemotherapy Extravasation NICaN Regional Pharmacy Group Version History Version 3: 2009 (See appendix 6) Version 2: January 2006 Version 1: June 2005 Consultation Period
Title Developed By Management of Chemotherapy Extravasation NICaN Regional Pharmacy Group Version History Version 3: 2009 (See appendix 6) Version 2: January 2006 Version 1: June 2005 Consultation Period
Richard J. Gralla, MD Medical Director Quality of Life Research Associates New York, NY
 Oncology Consultations Improving the Management of Chemotherapy-Induced Nausea and Vomiting (CINV) A CE-Certified Activity Featuring Consultations With Supported by an educational grant from Eisai. Dannemiller
Oncology Consultations Improving the Management of Chemotherapy-Induced Nausea and Vomiting (CINV) A CE-Certified Activity Featuring Consultations With Supported by an educational grant from Eisai. Dannemiller
MABCAMPATH mg 3 INJ GENZYME BIOPHARMACEUTICALS. MABCAMPBATH mg 3 INJ GENZYME BIOPHARMACEUTICALS
 ICON Formulary - November 2016 Class Medicine Name Nappi Strength Form Size Route Manufacturer Abiraterone Acetate ZYTIGA 250 720690001 250 mg 120 ORAL JANSSEN PHARMACEUTICALS Aldesleukin CHIRON IL-2 18
ICON Formulary - November 2016 Class Medicine Name Nappi Strength Form Size Route Manufacturer Abiraterone Acetate ZYTIGA 250 720690001 250 mg 120 ORAL JANSSEN PHARMACEUTICALS Aldesleukin CHIRON IL-2 18
Recognising and reducing the risk of chemotherapy extravasation
 Recognising and reducing the risk of chemotherapy extravasation When chemotherapy drugs leak from the veins it can cause serious injury to the patient, greatly heighten their fears of undergoing future
Recognising and reducing the risk of chemotherapy extravasation When chemotherapy drugs leak from the veins it can cause serious injury to the patient, greatly heighten their fears of undergoing future
Cancer Chemotherapy. Munir Gharaibeh MD, PhD, MHPE School of Medicine, The University of Jordan December 2018
 Cancer Chemotherapy Munir Gharaibeh MD, PhD, MHPE School of Medicine, The University of Jordan December 2018 االدوية Polyfunctional Alkylating Agents االستخالبية متعددة الوظائف Not cell cycle-specific.
Cancer Chemotherapy Munir Gharaibeh MD, PhD, MHPE School of Medicine, The University of Jordan December 2018 االدوية Polyfunctional Alkylating Agents االستخالبية متعددة الوظائف Not cell cycle-specific.
Systemic Anticancer Therapy Drug Interactions Table
 Systemic Anticancer Therapy Drug Interactions Table 1 This guide is intended to cover the most common interactions involving Systemic Anticancer therapies, e.g. cytotoxic agents, monoclonal antibodies
Systemic Anticancer Therapy Drug Interactions Table 1 This guide is intended to cover the most common interactions involving Systemic Anticancer therapies, e.g. cytotoxic agents, monoclonal antibodies
Year III Pharm D. Dr. V. Chitra
 Year III Pharm D. Dr. V. Chitra 50.2 Rang Etiolopathology Cell Cycle = Growth, Division 18_01_cell_cycle.jpg Characteristics of Cancer Cells The problem: Cancer cells divide rapidly (cell cycle is accelerated)
Year III Pharm D. Dr. V. Chitra 50.2 Rang Etiolopathology Cell Cycle = Growth, Division 18_01_cell_cycle.jpg Characteristics of Cancer Cells The problem: Cancer cells divide rapidly (cell cycle is accelerated)
Prior Authorization Proposal: Oncology Agents
 Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Copyright 2012 Oregon State University. All Rights Reserved Drug Use Research & Management Program Oregon State University, 500 Summer Street NE, E35 Salem, Oregon 97301-1079 Phone 503-947-5220 Fax 503-947-1119
Fundamentals of Pharmacology for Veterinary Technicians Chapter 20
 Figure 20-1 Figure 20-2 Figure 20-3A Figure 20-3B Figure 20-4 Table 20-1 Antineoplastic Agents Used for Chemotherapy in Animals Drug Type Drug Category Examples Cell-Cycle Effect CCNS Alkylating agents:
Figure 20-1 Figure 20-2 Figure 20-3A Figure 20-3B Figure 20-4 Table 20-1 Antineoplastic Agents Used for Chemotherapy in Animals Drug Type Drug Category Examples Cell-Cycle Effect CCNS Alkylating agents:
PRACTICE GUIDELINE. DEFINITIONS: Mild head injury: Glasgow Coma Scale* (GCS) score Moderate head injury: GCS 9-12 Severe head injury: GCS 3-8
 PRACTICE GUIDELINE Effective Date: 9-1-2012 Manual Reference: Deaconess Trauma Services TITLE: TRAUMATIC BRAIN INJURY GUIDELINE OBJECTIVE: To provide practice management guidelines for traumatic brain
PRACTICE GUIDELINE Effective Date: 9-1-2012 Manual Reference: Deaconess Trauma Services TITLE: TRAUMATIC BRAIN INJURY GUIDELINE OBJECTIVE: To provide practice management guidelines for traumatic brain
Index. derm.theclinics.com. Note: Page numbers of article titles are in boldface type.
 Note: Page numbers of article titles are in boldface type. A Acitretin and cutaneous T-cell lymphomas, 716 719, 722, 725 ADCC. See Antibody-dependent cell-mediated cytotoxicity. Adjunctive therapies and
Note: Page numbers of article titles are in boldface type. A Acitretin and cutaneous T-cell lymphomas, 716 719, 722, 725 ADCC. See Antibody-dependent cell-mediated cytotoxicity. Adjunctive therapies and
