UDK : 61 ISSN
|
|
|
- Frank Barton
- 6 years ago
- Views:
Transcription
1 J Med Biochem 2016; 35 (1) DOI: /jomb UDK : 61 ISSN J Med Biochem 35: 26 33, 2016 Original paper Originalni nau~ni rad INFLUENCE OF CYP2C19*2 GENE VARIANT ON THERAPEUTIC RESPONSE DURING CLOPIDOGREL TREATMENT IN PATIENTS WITH CAROTID ARTERY STENOSIS UTICAJ CYP2C19*2 VARIJANTE GENA NA TERAPIJSKI ODGOVOR U TOKU PRIMENE KLOPIDOGRELA KOD BOLESNIKA SA STENOZOM KAROTIDNE ARTERIJE Dragana Ba~kovi} 1, Svetlana Ignjatovi} 1,2, Ljiljana Raki}evi} 3, Jelena Kusi}-Ti{ma 3, Dragica Radojkovi} 3, Branko ^alija 4, Evgenija Strugarevi} 4, \or e Radak 4,5, Mirjana Kova~ 5,6 1 University of Belgrade Faculty of Pharmacy, Belgrade, Serbia 2 Center for Medical Biochemistry, Clinical Center of Serbia 3 Institute of Molecular Genetics and Genetic Engineering, University of Belgrade, Serbia 4 Institute for Cardiovascular Diseases Dedinje, Belgrade, Serbia 5 Faculty of Medicine, University of Belgrade, Serbia 6 Blood Transfusion Institute of Serbia, Hemostasis Department, Belgrade, Serbia Summary Background: Despite the proven clinical effect of oral antiplatelet drugs, a considerable number of patients do not have an adequate response to clopidogrel. The aim of our study was to determine the influence of CYP2C19*2 loss-of-function variant allele on clopidogrel responsiveness in patients with carotid artery stenosis. Methods: One hundred and twelve patients with carotid artery stenosis undergoing endarterectomy were included in this one-year prospective study. All of them received clopidogrel (75 mg daily) for at least 30 days after the intervention. They were followed from the moment of hospital admission. CYP2C19*2 genotyping was performed by TaqMan Assay. The influence of CYP2C19*2 variant allele on clopidogrel platelet reactivity was determined using multiple-electrode aggregometry (MEA). Results: Genotyping results showed that 82 (73.2%) patients were homozygous for wild type, 29 (25.9%) were heterozygous for the CYP2C19*2 allele and 1 (0.9%) was CYP2C19*2 homozygous. After 24 hours, among those with the wild type 29.3% were clopidogrel responders, and in those with the CYP2C19*2 alleles 10%. In the wild type group, 74.4% were clopidogrel responders after 7 days of Kratak sadr`aj Uvod: I pored dokazanog klini~kog efekta oralne antiagregacijske terapije, zna~ajan broj pacijenata nema adekvatan odgovor na primenjeni klopidogrel. Cilj na{e studije je bio da se utvrdi uticaj prisutne CYP2C19*2 varijante gena na terapijski odgovor u toku primene klopidogrela kod pacijenata sa stenozom karotidne arterije. Metode: U jednogodi{nju prospektivnu studiju uklju~eno je 112 pacijenata sa stenozom karotidne arterije kod kojih je izvr{ena endarterektomija. Posle operativnog zahvata, pa - cijenti su primali 75 mg dnevno klopidogrela u trajanju od najmanje mesec dana. Svi ispitanici su pra}eni od momenta prijema. Za CYP2C19 genotipizaciju kori{}en je TaqMan test. Uticaj CYP2C19*2 alela na trombocitnu reaktivnost ispitivan je primenom multiple-electrode aggregometry (MEA). Rezultati: Rezultati genotipizacije su pokazali da su 82 (73,2%) ispitanika homozigoti za wild-type, 29 (25,9%) heterozigoti za CYP2C19*2 alel, dok je 1 (0,9%) bio ho mo - zigot za CYP2C19*2. Nakon 24 sata, u grupi sa wild-type genotipom 29,3% ispitanika dali su odgovor na klopidogrel, a u grupi sa CYP2C19*2 varijantom gena 10% ispitanika. U grupi sa wild-type genotipom, 74,4% ispita nika su imali terapijski odgovor nakon 7 dana, odnosno 82,9% Address for correspondence: Dragana Backovic Faculty of Pharmacy, University of Belgrade Vojvode Stepe 450, Belgrade, Serbia Tel: Fax: draganabackoviccªgmail.com Abbreviations: CYP (the hepatic cytochrome P450), PCI (per cutaneous coronary intervention), CYP2C19*2; CYP2C19*3 (genetic polymorphism), MEA (multiple electrode aggregometry), AUC (area under curve), PCR (polymerase chain reaction), BMI (body mass index), HDL (high density lipoprotein), CEA (carotid endarterectomy), CCBs (calcium channel blockers).
2 J Med Biochem 2016; 35 (1) 27 taking the drug; 82.9% after 30 days of clopidogrel introduction, respectively. In patients with the CYP2C19*2 alleles the number of responders increased up to 46.7% after 7 days; 53.3% after 30 days of taking the drug, respectively. The risk for being a low-responder is higher for the patients heterozygous for the CYP2C19*2 allele vs. wildtype (OR 4.250, 95% CI , P<0.01). Conclusions: The CYP2C19*2 loss-of-function variant allele has significant influence on clopidogrel response in patients with carotid artery stenosis undergoing endarterectomy. Keywords: clopidogrel, CYP2C19*2, platelet reactivity nakon 30 dana od primene klopidogrela. U grupi sa CYP2C19*2 alelom broj ispitanika sa terapijskim odgo - vorom raste do 46,7% nakon 7 dana, odnosno do 53,3% nakon 30 dana od primene klopidogrela. Rizik za slab od - govor je ve}i kod nosilaca CYP2C19*2 alela u odnosu na nenosioce (wild-type) (OR 4,250, 95% CI , P<0,01). Zaklju~ak: CYP2C19*2 varijanta gena zna~ajno uti~e na terapijski odgovor u toku primene klopidogrela kod boles - nika sa stenozom karotidne arterije kod kojih je izvr{ena endarterektomija. Klju~ne re~i: klopidogrel, CYP2C19*2 varijanta gena, reaktivnost trombocita Introduction Platelets play a key role in the development of thrombotic complications in patients with atherosclerotic vascular disease (1). Oral antiplatelet drugs, aspirin and clopidogrel are the cornerstone therapy in conditions characterized by risk for arterial thrombosis (1). They are widely used as treatment for patients with acute coronary syndrome and for stent thrombosis prevention in patients undergoing percutaneous coronary intervention (2). Regarding patients undergoing carotid endarterectomy, perioperative anti - thrombotic therapy should consist of aspirin alone, while the addition of clopidogrel should be considered for high risk patients (3). Despite their proven clinical effect, a considerable number of patients do not have an adequate response to aspirin, clopidogrel or both (4). Ten to fifteen percent of patients receiving these medications have recurrent atherothrombotic events while receiving treatment (2, 5). Clopidogrel is a second-generation oral thieno - pyridine. It is a prodrug which is converted into its active metabolite by the isoforms of hepatic cyto - hrome P450 (CYP) in a two-step process. The active metabolite irreversibly inhibits ADP-stimulated platelet aggregation covalently binding to the purinergic P2Y12 receptor on the platelet surface (2). Approximately 85% is hydrolized into an inactive carboxylic acid derivative compound by esterase and 15% of the administered clopidogrel form the active clopidogrel thiol metabolite. The CYP2C19 enzyme is involved in both oxidative steps of clopidogrel metabolism (2, 4). Several pharmacogenomic studies de - monstrated that CYP2C19 variant alleles affect the pharmacokinetics and pharmacodynamics of clopidogrel and influence the interindividual heterogenity of the clopidogrel response (2). Variant alleles for which reduced or loss of function have been shown comprise CYP2C19*2 to CYP2C19*8. The CYP2C19*2 gene variant is a G681A mutation in exon 5 which encodes for a cryptic splice variant, while a G636A mutation in exon 4 results in a premature stop codon in CYP2C19*3 (6). The CYP2C19*2 is the most frequent variant allele in the Caucasian and the African population while CYP2C19*3 to CYP2C19*8 alleles have minor clinical impact due to their low frequency (<1%) (6). Individuals who are carriers of CYP2C19*2 and CYP2C19*3 alleles have a reduced metabolism of clopidogrel into its active metabolite and reduced ex vivo platelet reactivity, which leads to an increased risk of adverse cardiovascular events (5). The CYP2C19*17 allelic variant, a C806T mutation in exon 5, is responsible for an increased catalytic activity. Carriage of CYP2C19*17 allele(s) results in an increased platelet inhibition by clopidogrel due to an enhanced formation of the active metabolite which could lead to an increased risk of bleeding (6). So far, studies have examined whether clopidogrel treatment is affected by CYP2C19 variant alleles in patients with acute coronary syndrome (7, 8), myo - cardial infarction (9), coronary artery disease (10, 11) and in patients undergoing percutaneous coronary intervention (PCI) (12 16). According to the published data, the presence of CYP2C19*2 variant allele(s) leads to a reduction in clopidogrel-induced platelet inhibition. The aim of our study was to determine the influence of CYP2C19*2 loss-of-function variant allele on clopidogrel responsiveness in patients with carotid artery stenosis undergoing endarterectomy. Materials and Methods Patients From June 2012 to October 2013, 112 consecutive patients with carotid artery stenosis undergoing elective endarterectomy (CEA) at Institute for Cardio - vascular Diseases»Dedinje«, Belgrade, were enrolled in the prospective study. The patients characteristics are show in Table I. Related to the study criteria, all participants who received clopidogrel (75 mg daily) for at least 30 days from intervention were eligible for the study. Exclusion criteria were renal and/or liver dysfunction, diabetes mellitus, preoperative platelet count below 150x10 9 /L, intake of antiplatelet drugs other than
3 28 Ba~kovi} et al.: CYP2C19*2 gene variant and therapeutic response to clopidogrel Table I Baseline characteristics of patients. Age, years (mean ± SD) 66.2 ± 8.13 Gender, n (%) Male 66 (58.9) Female 46 (41.1) BMI, n (%) < (81.3) > (18.8) Risk factors, n (%) Smoking status/yes 84 (75.0) Hypercholesterolemia/yes 40 (35.7) Hypertriglyceridemia/yes 26 (23.2) Positive family history of cardiovascular disease/yes, n (%) 41 (36.6) Presence of stenosis of both ACI, n (%) 71 (63.4) Previous stroke or TIA, n (%) 40 (35.7) Stroke/yes 33 (82.5) TIA/yes 7 (17.5) Laboratory data (mean ± SD) Fibrinogen, g/l 4.6±1.1 Leukocytes, 10-9 /L 7.0±1.9 Hemoglobin, g/l 136.5±12.3 Platelets, 10-9 /L 219±57 Co-medication, n (%) Aspirin 112 (100) Beta blocker 41 (36.6) ACE inhibitor 64 (57.1) Calcium channel blocker 35 (31.3) Angiotensin II receptor antagonist 4 (3.6) Statins 53 (47.3) SD (standard deviation), BMI (body mass index, kg/m 2 ), CVD (cardiovascular diseases), ACI (internal carotid artery), TIA (transitory ischemic attack), ASA (acetylic salicylic acid), ACE (angiotensin converting enzyme). aspirin and clopidogrel. On admission patients were initially treated with aspirin. During their hospital stay after elective CEA, in all of them a dual combination of aspirin and clopidogrel of 75 mg was continued. The patients were followed up for 12 months from the moment of hospital admission. Patient compliance was assessed by telephone interviews. The outcomes that were evaluated during follow up were TIA, stroke and death. The research protocol was approved by the Ethics Committee of the Institute for Cardiovascular Di seases»dedinje«. All patients gave written in for - med consent for genotype determination and platelet function testing prior to study inclusion. Blood sampling For whole blood impedance aggregometry, blood samples were collected in tubes containing heparin as anticoagulant (BD, Swingdon, UK). Blood samples were kept at room temperature for approximately 30 min before platelet function testing. For genetic testing, blood samples were collected in tubes containing sodium citrate as anticoagulant (BD, Swingdon, UK). Platelet function testing For platelet function testing multiple electrode aggregometry (MEA) is used, which has been performed using the impedance aggregometer (Multi- plate analyzer, Roche Diagnostics, Mannheim, Ger - many). ADP-induced platelet aggregation was used to analyze the effect of clopidogrel. The in strument continuously measures the impedance change, which is proportional to the amount of pla telet aggregates on the electrode wires. The area under the aggregation curve (AUC) was used to express the aggregation response over the measured time (AU*min). Platelet aggregation was measured three times after intervention: 24 h after the first dose (75 mg) of clopidogrel, after 7 days and 30 days of taking the drug. In relation to the published results (17, 18), we performed the first measurement after 24 hours of clopidogrel in order to determine whether a lower clopidogrel dose (75 mg) affected platelet aggregation enough to point the difference in respect to the CY2C19*2 carriers status. We prospectively defined low response to clopidogrel by setting a cutoff point at the upper quartile (25%) of measurements by MEA 30 days after clopidogrel introduction. The pointed cutoff value of 575 AU*min was used in the assessment of therapeutic response at all three time points of measurements. Genotyping Genomic DNA was isolated from whole blood using QIAamp DNA Blood Mini Kit (QIAGEN, Hilden, Germany) according to the supplier s instruction. Genotyping for CYP2C19*2 variant allele was performed by TaqMan Drug Metabolism Genotyping Assay for SNP rs (Thermo Fisher Scientific, Life Technologies, Carlsbad, CA, US), on a 7500 Real Time PCR System (Applied Biosystems, Foster City, CA, USA), according to the manufacturer s protocol. The results were analyzed by Applied Biosystems 7500 Real-Time PCR System Sequence Detection Software v1.4.1.
4 J Med Biochem 2016; 35 (1) 29 Statistical methodology The normality of continuous variables was evaluated using the Kolmogorov-Smirnoff test. According to the Kolmodorov-Smirnoff test, all data showed normal distribution and continuous variables are presented as mean and standard deviation (SD). Categorical variables are presented as numbers or percentages and they were compared using Pearson chi-square test. Testing of the difference in ADP values at three different moments and in two groups was performed using two way parametric ANOVA with the Bonferroni test for post hoc comparson. Finally, for estimating predictors of low responsiveness, the binary logistic regression model was used. A P value < 0.05 was considered to indicate a significant difference. Statistical analyses in all applied methods were performed using SPSS version 20.0 (SPSS Inc., Chicago, Illinois). Results Genotyping results showed that 82 (73.2%) patients were homozygous for wild type, 29 (25.9%) were heterozygous for the CYP2C19*2 variant allele and 1 (0.9%) was homozygous for CYP2C19*2. Thus, the wild type genotype frequency was 73.2% vs. 26.8% for CYP2C19*2 carriers (Table II). Mean and SD values of ADP-induced platelet aggregation across CYP2C19 genotypes measured 24 h after the first dose of clopidogrel, after 7 and 30 days of taking the drug are presented in Figure 1. The values of platelet aggregation were statistically significantly different between the two genotype groups, P<0.01. As showed in the Figure 1, platelet aggrega- Table II Prevalence of CYP2C19 genotypes in study population. Genotype n (%) Wild type (CYP2C19*1/*1) 82 (73.2) Heterozygous for CYP2C19*2 29 (25.9) Homozygous for CYP2C19*2 1 (0.9) CYP (the hepatic cytochrome P 450) ADP (AU*min) ADP 24h ADP 7 day ADP 30 day 0 Wild type Heterozygote (1/2*) Figure 1 ADP-induced platelet aggregation across CYP2C19 genotypes measured 24 h after the first clopidogrel dose, after 7 days and after 30 days of clopidogrel treatment. The bottom of the box plots for ADP-induced platelet inhibition mark the 25 th percentile, the median lines mark the 50 th percentile, the top of the boxes mark the 75 th percentile, the bottom and top of the vertical lines mark the 5 th and 95 th percentile.
5 30 Ba~kovi} et al.: CYP2C19*2 gene variant and therapeutic response to clopidogrel Table III ADP-induced platelet aggregation values over time across CYP2C19 genotypes. ADP 24h, mean (SD)# ADP 7day, mean (SD)# P value Wild type (189.71) (191.30) <0.001 Heterozygous for CYP2C19* (165.08) (232.18) ADP 7day, mean (SD)# ADP 30 day, mean (SD)# P value Wild type (191.30) (183.47) <0.001 Heterozygous for CYP2C19* (232.18) (257.06) ADP 24h, mean (SD)# ADP 30 day, mean (SD)# P value Wild type (189.71) (183.47) <0.001 Heterozygous for CYP2C19* (165.08) (257.06) <0.001 #Platelet aggregation was expressed as AU*min Table IV Correlation between genotype and phenotype. After 24 hours of taking the drug P value Wild type Heterozygous for CYP2C19* clopidogrel responder, n (%) 24 (29.3) 3 (10.0) clopidogrel low-responder, n (%) 58 (70.7) 27 (90.0) After 7 days of taking the drug Wild type Heterozygous for CYP2C19* clopidogrel responder, n (%) 61 (74.4) 14 (46.7) clopidogrel low-responder, n (%) 21 (25.6) 16 (53.3) After 30 days of taking the drug Wild type Heterozygous for CYP2C19* clopidogrel responder, n (%) 68 (82.9) 16 (53.3) clopidogrel low-responder, n (%) 14 (17.1) 14 (46.7) tion was significantly higher in patients carrying the CYP2C19*2 allele than in the group of non-carriers in all three measurements; P<0.01. Analysis of the numerical values of platelet aggregation also showed significant difference in relation to genotype. In the group with wild type, ADPinduced platelet aggregation showed a tendency of decline over time. In patients with the CYP2C19*2 allele, the decline of platelet aggregation was ob - served after 7 days of clopidogrel and remained largely unchanged at the last measurement performed after 30 days of clopidogrel intake (Table III). The correlation between CYP2C19 genotype and therapeutic response to clopidogrel was showed in Table IV. The cutoff value for post-treatment mea - surements defining the upper quartile (25%) of patients was 575 AU*min. According to this cutoff value, after 24 hours, in the group with the wild type 29.3% of patients were clopidogrel responders, compared to only 10% from the group with the CYP2C19*2 variant, P= In the wild type group, 74.4% were clopidogrel responders after 7 days of taking the drug; 82.9% after 30 days of clopidogrel introduction, respectively. In patients with the
6 J Med Biochem 2016; 35 (1) 31 CYP2C19*2 alleles the number of responders in - creased up to 46.7% after 7 days; 53.3% after 30 days of taking the drug, respectively. So, we notice a significant difference between the two genotype groups at the last two measurements of platelet reactivity, P=0.006, P=0.001 respectively. The patient who was homozygous for CYP2C19*2 had no therapeutic response from the beginning of the treatment. The risk for being a lowresponder is higher for subjects heterozygous for the CYP2C19*2 allele vs. wild type (OR 4.250, 95% CI ). In the logistic regression model, the CYP2C19*2 variant allele was found to be an independent predictor of low responsiveness to clopidogrel, P<0.01. In the follow up period, stroke occurred in 2 patients (1.8%). Both of them are carriers of the CYP2C19*2 allele. Due to the size of the group who developed stroke, we could not use any appropriate statistical method in order to test whether there is an influence of the CYP2C19*2 variant allele on the development of stroke. Discussion To the best of our knowledge, this is the first study in which the laboratory response to clopidogrel was assessed to evaluate the relationship between the CYP2C19*2 loss-of-function allele and platelet aggregation in patients with carotid artery stenosis undergoing CEA, who received antiplatelet treatment with clopidogrel. In our study group, 26.8% of patients are CYP2C19*2 carriers, which is in line with the previously published data where 27.1% of patients were carriers of at least one variant allele (6). We have to note that our data are the first results relating to the CYP2C19*2 variant allele frequency in a Serbian po p - ulation. The results from our study demonstrated that patients carrying the CYP2C19*2 allele had significantly higher ADP-induced platelet aggregation compared to the non-carriers. This is in accordance with the results from previously published studies, which gave evidence of a relationship between CYP2C19 variant alleles and reduced le vels of the active clopidogrel me tabolite in the plasma (7, 10) and reduced platelet inhibition (7, 9, 10, 12 14, 19 22) which cause poor or absence of responsiveness to clopidogrel and ina de quate protection from ischemic events (7, 9, 10, 13, 14). Analysis of the numerical values of platelet aggregation also showed significant difference in relation to genotype. Specifically, in the group with wild type ADP-induced platelet aggregation showed a tendency of decline over time, while in the group with the CYP2C19*2 variant allele the decline in value of platelet aggregation was observed 7 days after clopidogrel treatment and remained largely unchanged. In our study population, one patient was homo - zygous for CYP2C19*2 and he had no therapeutic response from the beginning of clopidogrel treatment. Our results agree with data reported by others, where platelet aggregation values in CYP2C19*2/*2 patients are significantly higher than in CYP2C19 *1/*2 patients and the wild type genotype (13, 19). In our study among patients carrying the CYP2C19*2 allele, 46.7% were clopidogrel lowresponders after 30 days of taking the drug. In contrast, only 27% of patients with the loss-of-function polymorphism were classified as clopidogrel lowresponders in the Pe gasus-pci study (16). Unlike the Pegasus-PCI study, our study included a smaller number of patients with different clinical presentation who were administered a lower dose of clopidogrel (75 mg). Also, a possible reason for the difference be - tween Pegasus-PCI and our study arises from the fact that the interval between clopidogrel intake and blood sampling was different. Additionally, PCI leads to the release of multiple coagulation mediators, which additionally activate platelets (16). Statistical analysis of our results showed that the risk for being a clopidogrel low-responder is 4.25-fold higher for patients heterozygous for the CYP2C19*2 allele compared to those homozygous for the wild type. Similar results were presented in previous published studies, too (19, 21, 23). We have to note that there are contrasting conclusions in the literature about the predictive value of CYP2C19*2 variant allele as well as linking pharmacodynamic response to the drug and clinical outcomes. These uncertainties are likely due to the inaccuracies and lack of standardization of platelet functional tests that have been used to detect lowresponders to clopidogrel, as well as the absence of a universally accepted and validated cutoff value for platelet function (24). Also, it has been well documented that platelet reactivity is influenced by clinical presentation and that choice of the best cutoff value is disease specific. This approach would have allowed a more accurate identification of low-responders to clopidogrel and prediction of cardiovascular outcomes during treatment (15, 24). Further, we should stress the possible effect of co-medications on clopidogrel response. The data concerning statin clopidogrel interactions are inconsistent. In vitro studies have showed that the conversion of clopidogrel into the active metabolite is inhibited by atorvastatin, but in vivo studies in healthy volunteers could not confirm an interaction between lipophilic statins and clopidogrel, especially when evaluations were performed with patients receiving maintenance therapy or after they had been given a 600 mg loading dose of clopidogrel (26 28). More - over, a recent study showed that the addition of highdose atorvastatin (80 mg) for 30 days significantly improved the pharmacodynamic effects of double-
7 32 Ba~kovi} et al.: CYP2C19*2 gene variant and therapeutic response to clopidogrel dose clopidogrel, reducing platelet reactivity and improving optimal clopidogrel response in statin naïve patients with high-on treatment platelet reactivity on standard-dose clopidogrel (28). Some calcium channel blockers (CCBs) have strong inhibitory effects on the drug transporter p-glycoprotein (P-gp), which may cause de creased intestinal efflux of clopidogrel (29). Harmsze et al. evaluated the effect of co-administation of Pgp-inhibiting CCBs (verapamil, nifedipin, diltiazem, barnidipine) and non-pgp-inhibiting CCBs (amlo di pine) on clopidogrel platelet reactivity in patients on dual antiplatelet therapy undergoing PCI. According to the results, only amplodipine was associated with a high risk of clopidogrel poor response (30). Among the limitations of our study that should be discussed is the fact that this is an observational single-center study that involved a relatively small number of patients and as such it should be considered as a pilot study. Also, in order to obtain clear results about the influence of CYP2C19*2 loss-offunction genotype on clopidogrel responsiveness, we did not include the patients with diabetes mellitus. In addition, we assessed only the impact of one allelic variant of the CYP2C19 isoenzyme on clopidogrel responsiveness. Conclusion The results from our study showed that the CYP2C19*2 loss-of-function variant allele has significant influence on clopidogrel response in patients with carotid artery stenosis undergoing endarterectomy. Acknowledgments: The Ministry of Education, Science and Technological Development of the Republic of Serbia supported this study on the basis of contracts No and No Conflict of interest statement The authors stated that they have no conflicts of interest regarding the publication of this article. References 1. Spiliopoulos S, Pastromas G, Katsanos K, Kitrou P, Kar - na batidis D, Siablis D. Platelet responsiveness to clopidogrel treatment after peripheral endovascular procedures; The PRECLOP study: Clinical impact and optimal cut off value of on-treatment high platelet reactivity. J Am Coll Cardiol 2013; 61: Yin T, Miyata T. Pharmacogenomics of clopidogrel: Evi - dence and perspective. Thromb Res 2011; 128: Paciaroni M, Bogousslavsky J. Antithrombotic therapy in carotid artery stenosis: an update. Eur Neurol 2015; 73: Würtz M, Lordkipanidzé M, Grove EL. Pharmaco ge no mics in cardiovascular disease: focus on aspirin and ADP re ce - ptor antagonist. J Thromb Haemost 2013; 11: Ahmad T, Voora D, Becker RC. The pharmacogenetics of antiplatelet agents: towards personalized therapy? Nat Rev Cardiol 2011; 8: Trenk D, Hochholzer W. Genetics of platelet inhibitor treatment. Br J Clin Pharmacol 2014; 77: Mega JL, Close SL, Wiviott SD, Shen L, Hockett RD, Brandt JT, et al. Cytochrome P-450 polymorphisms and response to clopidogrel. N Engl J Med 2009; 360: Stimpfle F, Karathanos A, Droppa M, Metzger J, Rath D, Müller K, et al. Impact of point-of-care testing for CYP2C19 on platelet inhibition in patients with acute co - r onary syndrome and early dual antiplatelet therapy in the emergency setting. Thromb Res 2014; 134: Simon T, Verstuyft C, Mary-Krause M, Quteineh L, Drouet E, Méneveau N, et al., for the French Registry of Acute ST-Elevation and Non-ST-Elevation Myocardial Infarction (FAST-MI) Investigators. Genetic Determinants of response to clopidogrel and cardiovascular events. N Engl J Med 2009; 360: Varenhorst C, James S, Erlinge D, Brandt JT, Braun OÖ, Man M, et al. Genetic variation of CYP2C19 affects both pharmacokinetic and pharmacodynamic responses to clopidogrel but not prasugrel in aspirin-treated patients with coronary artery disease. Eur Heart J 2009; 30: Erlinge D, James S, Duvvuru S, Jakubowski JA, Wagner H, Varenhorst C, et al. Clopidogrel metaboliser status based on point-of-care CYP2C19 genetic testing in patients with coronary artery disease. Thromb Haemost 2014; 111: Campo G, Parrinello G, Ferraresi P, Lunghi B, Tebaldi M, Miccoli M, et al. Prospective evaluation of on-clopidogrel platelet reactivity over time in patients treated with percutaneous coronary intervention. J Am Coll Cardiol 2011; 57: Shuldiner AR, O Connell JR, Bliden KP, Gandhi A, Ryan K, Horenstein RB, et al. Association of Cytochrome P450 2C19 genotype with the antiplatelet effect and clinical efficacy of clopidogrel therapy. JAMA 2009; 302: Trenk D, Hochholzer W, Fromm MF, Chialda LE, Pahl A, Valina CM, et al. Cytochrome P450 2C19 681G>A polymorphism and high on-clopidogrel platelet reactivity associated with 1-year clinical outcome of elective percutaneous coronary intervention with drug-eluting or baremetal stents. J Am Coll Cardiol 2008; 51:
8 J Med Biochem 2016; 35 (1) Campo G, Fileti L, De Cesare N, Meliga E, Furgieri A, Russo F, et al. on behalf of the 3T/2R Investigators. Long-term clinical outcome based on aspirin and clopidogrel responsiveness status after elective percutaneous coronary intervention: a 3T/2R (Tailoring Treatment With Tirofiban in Patients Showing Resistance to Aspirin and/or Resistance to Clopidogrel) trial substudy. J Am Coll Cardiol 2010; 56: Siller-Matula JM, Delle-Karth G, Lang IM, Neunteufl T, Kozinski M, Kubica J, et al. Phenotyping vs. genotyping for prediction of clopidogrel efficacy and safety: the PEGASUS-PCI study. J Thromb Haemost 2012; 10: Mrdovic I, Savic L, Krljanac G, Asanin M, Perunicic J, Antonijevic N, et al. Impact of high post-loading platelet aggregation on 30-day clinical outcomes after primary percutaneous coronary intervention. The antiplatelet regimen tailoring after primary PCI (ART-PCI) trial. Int J Cardiol 2013; 167: Chirumamilla AP, Maehara A, Mintz GS, Mehran R, Kanwal S, Weisz G, et al. High platelet reactivity on clopidogrel therapy correlates with increased coronary atherosclerosis and calcification: a volumetric intravascular ultrasound study. JACC Cardiovasc Imaging 2012; 5: Sibbing D, Gebhard D, Koch W, Braun S, Stegherr J, Morath T, et al. Isolated and interactive impact of common CYP2C19 genetic variants on the platelet effect of chronic clopidogrel therapy. J Thromb Haemost 2010; 8: Holmes MV, Perel P, Shah T, Hingorani AD, Casas JP. CYP2C19 genotype, clopidogrel metabolism, platelet function and cardiovascular events: a systematic review and meta-analysis. JAMA 2011; 306: Nakata T, Miyahara M, Nakatani K, Wada H, Tanigawa T, Komada F, et al., for the McLORDD group. Relationship between CYP2C19 loss-of-function polymorphism and platelet reactivities with clopidogrel treatment in Japanese patients undergoing coronary stent implantation. Circ J 2013; 77: Tatarunas V, Jankauskiene L, Kupstyte N, Skipskis V, Gustiene O, Grybauskas P, et al. The role of clinical parameters and CYP2C19 G681 and CYP4F2 G1347A polymorphisms on platelet reactivity during dual antiplatelet therapy. Blood Coagul Fibrinolysis 2014; 25: Zhang L, Chen Y, Jin Y, Qu F, Li J, Ma C, et al. Genetic determinants of high on-treatment platelet reactivity in clopidogrel treated Chinese patients. Thromb Res 2013; 132: Cattaneo M. Response variability to clopidogrel: is tailored treatment, based on laboratory testing, the right solution? J Thromb Haemost 2012; 10: Siller-Matula JM, Trenk D, Krahenbuhl S, Michelson AD, Delle-Karth G. Clinical implications of drug-drug interactions with P2Y12 receptor inhibitors. J Thromb Haemost 2014; 12: Pavlovi} S, Zuki} B, Stojiljkovi} Petrovi} M. Molecular genetic markers as a basis for personalized medicine. J Med Biochem 2014; 33: Stankovi} S, A{anin M, Majki}-Singh N. The pharmacogenetics of cardiovascular drugs. J Med Biochem 2014; 33: Leoncini M, Toso A, Maioli M, Bellandi F. Statin and clopidogrel pharmacological interaction. G Ital Cardiol (Rome) 2013; 14: , 29. Wang ZY, Chen M, Zhu LL, Yu LS, Zeng S, Xiang MX, et al. Pharmacokinetic drug interactions with clopidogrel: updated review and risk management in combination therapy. Ther Clin Risk Manag 2015; 11: Harmsze AM, Robijns K, van Werkum JW, Breet NJ, Hackeng CM, ten Berg JM, et al. The use of amlodipine, but not of P-glycoprotein inhibiting calcium channel blockers is associated with clopidogrel poor-response. Thromb Haemost 2010; 103: Received: April 16, 2015 Accepted: June 16, 2015
JOINT MEETING OF CORONARY REVASCULARIZATION 2014 TIONG LEE LEN SENIOR RESEARCH PHARMACIST CLINICAL RESEARCH CENTER, SARAWAK GENERAL HOSPITAL
 The Effect of Non Steady State and Steady State Clopidogrel Carboxylic Acid Plasma Concentration on Clopidogrel Responsiveness in Patients Planned For Percutaneous Coronary Intervention JOINT MEETING OF
The Effect of Non Steady State and Steady State Clopidogrel Carboxylic Acid Plasma Concentration on Clopidogrel Responsiveness in Patients Planned For Percutaneous Coronary Intervention JOINT MEETING OF
New insights in stent thrombosis: Platelet function monitoring. Franz-Josef Neumann Herz-Zentrum Bad Krozingen
 New insights in stent thrombosis: Platelet function monitoring Franz-Josef Neumann Herz-Zentrum Bad Krozingen New insights in stent thrombosis: Platelet function monitoring Variability of residual platelet
New insights in stent thrombosis: Platelet function monitoring Franz-Josef Neumann Herz-Zentrum Bad Krozingen New insights in stent thrombosis: Platelet function monitoring Variability of residual platelet
Thrombosis Research active studies
 Thrombosis Research active studies A Pharmacodynamic Study Comparing Prasugrel Versus Ticagrelor in Patients With Coronary Artery Disease Undergoing PCI With CYP2C19 Loss-of-function Genotypes: A Feasibility
Thrombosis Research active studies A Pharmacodynamic Study Comparing Prasugrel Versus Ticagrelor in Patients With Coronary Artery Disease Undergoing PCI With CYP2C19 Loss-of-function Genotypes: A Feasibility
What hematologists should know about VerifyNow
 What hematologists should know about VerifyNow Hematology fellows conference 12/13/2013 Presenter: Christina Fitzmaurice, MD, MPH Discussant: Daniel Sabath, MD, PhD HMC consult patient 54 yo woman admitted
What hematologists should know about VerifyNow Hematology fellows conference 12/13/2013 Presenter: Christina Fitzmaurice, MD, MPH Discussant: Daniel Sabath, MD, PhD HMC consult patient 54 yo woman admitted
Cost is not a barrier to implementing clopidogrel pharmacogenetics. Larisa H. Cavallari, Pharm.D., and Glen T. Schumock, Pharm.D., M.B.A.
 Cost is not a barrier to implementing clopidogrel pharmacogenetics Larisa H. Cavallari, Pharm.D., and Glen T. Schumock, Pharm.D., M.B.A. From the Department of Pharmacy Practice (both authors), and the
Cost is not a barrier to implementing clopidogrel pharmacogenetics Larisa H. Cavallari, Pharm.D., and Glen T. Schumock, Pharm.D., M.B.A. From the Department of Pharmacy Practice (both authors), and the
Role of Clopidogrel in Acute Coronary Syndromes. Hossam Kandil,, MD. Professor of Cardiology Cairo University
 Role of Clopidogrel in Acute Coronary Syndromes Hossam Kandil,, MD Professor of Cardiology Cairo University ACS Treatment Strategies Reperfusion/Revascularization Therapy Thrombolysis PCI (with/ without
Role of Clopidogrel in Acute Coronary Syndromes Hossam Kandil,, MD Professor of Cardiology Cairo University ACS Treatment Strategies Reperfusion/Revascularization Therapy Thrombolysis PCI (with/ without
Session Objectives. Clopidogrel Resistance. Clopidogrel (Plavix )
 Session Objectives New Antithrombotics and Real Time Genetic Testing: Their Role in the Vascular Patient Margaret C. Fang, MD, MPH Associate Professor of Medicine Division of Hospital Medicine Medical
Session Objectives New Antithrombotics and Real Time Genetic Testing: Their Role in the Vascular Patient Margaret C. Fang, MD, MPH Associate Professor of Medicine Division of Hospital Medicine Medical
ΠΑΝΕΠΙΣΤΗΜΙΟ ΙΩΑΝΝΙΝΩΝ. Εξατοµικευµένη αντιαιµοπεταλιακή αγωγή. Ποιο είναι το µέλλον?
 ΠΑΝΕΠΙΣΤΗΜΙΟ ΙΩΑΝΝΙΝΩΝ ΕΡΕΥΝΗΤΙΚΟ ΚΕΝΤΡΟ ΑΘΗΡΟΘΡΟΜΒΩΣΗΣ Εξατοµικευµένη αντιαιµοπεταλιακή αγωγή. Ποιο είναι το µέλλον? Αλέξανδρος Δ. Τσελέπης, MD, PhD Καθηγητής Βιοχηµείας - Κλινικής Χηµείας Disclosures
ΠΑΝΕΠΙΣΤΗΜΙΟ ΙΩΑΝΝΙΝΩΝ ΕΡΕΥΝΗΤΙΚΟ ΚΕΝΤΡΟ ΑΘΗΡΟΘΡΟΜΒΩΣΗΣ Εξατοµικευµένη αντιαιµοπεταλιακή αγωγή. Ποιο είναι το µέλλον? Αλέξανδρος Δ. Τσελέπης, MD, PhD Καθηγητής Βιοχηµείας - Κλινικής Χηµείας Disclosures
Pharmacogenomics with Clopidogrel: Does One Size Fit All?
 Pharmacogenomics with Clopidogrel: Does One Size Fit All? Leah A. Sabato, PharmD PGY-1 Pharmacy Practice Resident UC Health - University of Cincinnati Medical Center leah.sabato@uchealth.com April 2015
Pharmacogenomics with Clopidogrel: Does One Size Fit All? Leah A. Sabato, PharmD PGY-1 Pharmacy Practice Resident UC Health - University of Cincinnati Medical Center leah.sabato@uchealth.com April 2015
Cytochrome P450 interactions
 Cytochrome P450 interactions Learning objectives After completing this activity, pharmacists should be able to: Explain the mechanism of action of clopidogrel-ppi interaction Assess the risks and benefits
Cytochrome P450 interactions Learning objectives After completing this activity, pharmacists should be able to: Explain the mechanism of action of clopidogrel-ppi interaction Assess the risks and benefits
GENNARO SARDELLA MD, FACC,FESC
 PhaRmacodynamic Effects of Switching therapy in PCI patients with high on Treatment platelet reactivity and genotype variation: high Clopidogrel dose versus Prasugrel (RESET GENE Study). (ClinicalTrials.gov
PhaRmacodynamic Effects of Switching therapy in PCI patients with high on Treatment platelet reactivity and genotype variation: high Clopidogrel dose versus Prasugrel (RESET GENE Study). (ClinicalTrials.gov
INDIVIDUALIZED MEDICINE
 CENTER FOR INDIVIDUALIZED MEDICINE Clopidogrel Pharmacogenetics Can We Impact Clinical Practice? Michael E. Farkouh, MD, MSc Peter Munk Cardiac Centre University of Toronto Naveen Pereira MD Mayo Clinic
CENTER FOR INDIVIDUALIZED MEDICINE Clopidogrel Pharmacogenetics Can We Impact Clinical Practice? Michael E. Farkouh, MD, MSc Peter Munk Cardiac Centre University of Toronto Naveen Pereira MD Mayo Clinic
Stephan Windecker Department of Cardiology Swiss Cardiovascular Center and Clinical Trials Unit Bern Bern University Hospital, Switzerland
 Advances in Antiplatelet Therapy in PCI and ACS Stephan Windecker Department of Cardiology Swiss Cardiovascular Center and Clinical Trials Unit Bern Bern University Hospital, Switzerland Targets for Platelet
Advances in Antiplatelet Therapy in PCI and ACS Stephan Windecker Department of Cardiology Swiss Cardiovascular Center and Clinical Trials Unit Bern Bern University Hospital, Switzerland Targets for Platelet
USING OF DUAL ANTIPLATELET THERAPY IN POST PCI PATIENTS TO REDUCE CORONARY STENT THROMBOSIS: A REVIEW OF GENETIC TEST AND TEG
 USING OF DUAL ANTIPLATELET THERAPY IN POST PCI PATIENTS TO REDUCE CORONARY STENT THROMBOSIS: A REVIEW OF GENETIC TEST AND TEG Armel Kemwhoua Youssa 1, Mingming Yang 1,2 and Genshan Ma 1,2* 1 College of
USING OF DUAL ANTIPLATELET THERAPY IN POST PCI PATIENTS TO REDUCE CORONARY STENT THROMBOSIS: A REVIEW OF GENETIC TEST AND TEG Armel Kemwhoua Youssa 1, Mingming Yang 1,2 and Genshan Ma 1,2* 1 College of
Surveying the Landscape of Oral Antiplatelet Therapy in Acute Coronary Syndrome Management
 Surveying the Landscape of Oral Antiplatelet Therapy in Acute Coronary Syndrome Management Jeffrey S Berger, MD, MS Assistant Professor of Medicine and Surgery Director of Cardiovascular Thrombosis Disclosures
Surveying the Landscape of Oral Antiplatelet Therapy in Acute Coronary Syndrome Management Jeffrey S Berger, MD, MS Assistant Professor of Medicine and Surgery Director of Cardiovascular Thrombosis Disclosures
and Ticagrelor Professor of Medicine (Cardiology), Georgetown University Associate Director, Division of Cardiology, Washington Hospital Center
 Role of Genotyping and Point-of-Care of Testing in Clopidogrel, Prasugrel, and Ticagrelor Ron Waksman, MD Ron Waksman, MD Professor of Medicine (Cardiology), Georgetown University Associate Director, Division
Role of Genotyping and Point-of-Care of Testing in Clopidogrel, Prasugrel, and Ticagrelor Ron Waksman, MD Ron Waksman, MD Professor of Medicine (Cardiology), Georgetown University Associate Director, Division
ACCP Cardiology PRN Journal Club
 ACCP Cardiology PRN Journal Club 1 Optimising Crossover from Ticagrelor to Clopidogrel in Patients with Acute Coronary Syndrome [CAPITAL OPTI-CROSS] Monique Conway, PharmD, BCPS PGY-2 Cardiology Pharmacy
ACCP Cardiology PRN Journal Club 1 Optimising Crossover from Ticagrelor to Clopidogrel in Patients with Acute Coronary Syndrome [CAPITAL OPTI-CROSS] Monique Conway, PharmD, BCPS PGY-2 Cardiology Pharmacy
Multiplate analyzer Cut-off-values ADPtest and ASPItest
 Multiplate analyzer Cut-off-values ADPtest and ASPItest Cut-off values ADPtest The cut-off values were established in hirudin blood samples. Healthy individuals AUC (U) 1 Patient on ADP receptor antagonist
Multiplate analyzer Cut-off-values ADPtest and ASPItest Cut-off values ADPtest The cut-off values were established in hirudin blood samples. Healthy individuals AUC (U) 1 Patient on ADP receptor antagonist
Genetic Causes of Clopidogrel Nonresponsiveness: Which Ones Really Count?
 Genetic Causes of Clopidogrel Nonresponsiveness: Which Ones Really Count? Kathryn M. Momary, Pharm.D., Michael P. Dorsch, Pharm.D., M.S., and Eric R. Bates, M.D. Clopidogrel decreases the morbidity and
Genetic Causes of Clopidogrel Nonresponsiveness: Which Ones Really Count? Kathryn M. Momary, Pharm.D., Michael P. Dorsch, Pharm.D., M.S., and Eric R. Bates, M.D. Clopidogrel decreases the morbidity and
Impact of CYP2C19 and ABCB1 SNPs on outcomes with ticagrelor versus clopidogrel in acute coronary syndromes: a PLATO genetic substudy
 Impact of CYP2C19 and ABCB1 SNPs on outcomes with ticagrelor versus clopidogrel in acute coronary syndromes: a PLATO genetic substudy Lars Wallentin, Stefan James, Robert F Storey, Martin Armstrong, Bryan
Impact of CYP2C19 and ABCB1 SNPs on outcomes with ticagrelor versus clopidogrel in acute coronary syndromes: a PLATO genetic substudy Lars Wallentin, Stefan James, Robert F Storey, Martin Armstrong, Bryan
Platelet function testing to guide P2Y 12 -inhibitor treatment in ACS patients after PCI: insights from a national program in Hungary
 Platelet function testing to guide P2Y 12 -inhibitor treatment in ACS patients after PCI: insights from a national program in Hungary Dániel Aradi MD PhD Interventional Cardiologist Assistant professor
Platelet function testing to guide P2Y 12 -inhibitor treatment in ACS patients after PCI: insights from a national program in Hungary Dániel Aradi MD PhD Interventional Cardiologist Assistant professor
Abstract Background: Methods: Results: Conclusions:
 Two-Year Clinical and Angiographic Outcomes of Overlapping Sirolimusversus Paclitaxel- Eluting Stents in the Treatment of Diffuse Long Coronary Lesions Kang-Yin Chen 1,2, Seung-Woon Rha 1, Yong-Jian Li
Two-Year Clinical and Angiographic Outcomes of Overlapping Sirolimusversus Paclitaxel- Eluting Stents in the Treatment of Diffuse Long Coronary Lesions Kang-Yin Chen 1,2, Seung-Woon Rha 1, Yong-Jian Li
Clopidogrel Use in ACS and PCI: Clinical Trial Update
 Clopidogrel Use in ACS and PCI: Clinical Trial Update Matthew J. Price MD Director, Cardiac Catheterization Laboratory, Scripps Clinic, La Jolla, CA Assistant Professor, Scripps Translational Science Institute
Clopidogrel Use in ACS and PCI: Clinical Trial Update Matthew J. Price MD Director, Cardiac Catheterization Laboratory, Scripps Clinic, La Jolla, CA Assistant Professor, Scripps Translational Science Institute
APPLICATION OF GENETIC TESTING FOR CYP 450 POLYMORPHISM TO PREDICT RESPONSE TO CLOPIDOGREL INPATIENT UNDERGOING PCI
 INDIAN JOURNAL OF CARDIOVASCULAR DISEASES JOURNAL in women (IJCD) 2017 VOL 2 ISSUE 2 ORIGINAL ARTICLE 1 APPLICATION OF GENETIC TESTING FOR CYP 450 POLYMORPHISM TO PREDICT RESPONSE TO CLOPIDOGREL INPATIENT
INDIAN JOURNAL OF CARDIOVASCULAR DISEASES JOURNAL in women (IJCD) 2017 VOL 2 ISSUE 2 ORIGINAL ARTICLE 1 APPLICATION OF GENETIC TESTING FOR CYP 450 POLYMORPHISM TO PREDICT RESPONSE TO CLOPIDOGREL INPATIENT
Point-of-Care Genetic Testing for Tailored Anti-Platelet Therapy Ready for Prime Time?
 Point-of-Care Genetic Testing for Tailored Anti-Platelet Therapy Ready for Prime Time? Robert G. Wilkins, M.D., F.A.C.C., F.S.V.M New Cardiovascular Horizons May 29, 2015 No Disclosures Dual Anti-Platelet
Point-of-Care Genetic Testing for Tailored Anti-Platelet Therapy Ready for Prime Time? Robert G. Wilkins, M.D., F.A.C.C., F.S.V.M New Cardiovascular Horizons May 29, 2015 No Disclosures Dual Anti-Platelet
July ACCP Cardiology PRN Journal Club 7/23/2018
 July ACCP Cardiology PRN Journal Club 7/23/2018 Dr. Michael Plazak Dr. Michael Plazak is a PGY2 Cardiology Pharmacy Resident at the University of Maryland School of Pharmacy. He graduated from the University
July ACCP Cardiology PRN Journal Club 7/23/2018 Dr. Michael Plazak Dr. Michael Plazak is a PGY2 Cardiology Pharmacy Resident at the University of Maryland School of Pharmacy. He graduated from the University
OUTPATIENT ANTITHROMBOTIC MANAGEMENT POST NON-ST ELEVATION ACUTE CORONARY SYNDROME. TARGET AUDIENCE: All Canadian health care professionals.
 OUTPATIENT ANTITHROMBOTIC MANAGEMENT POST NON-ST ELEVATION ACUTE CORONARY SYNDROME TARGET AUDIENCE: All Canadian health care professionals. OBJECTIVE: To review the use of antiplatelet agents and oral
OUTPATIENT ANTITHROMBOTIC MANAGEMENT POST NON-ST ELEVATION ACUTE CORONARY SYNDROME TARGET AUDIENCE: All Canadian health care professionals. OBJECTIVE: To review the use of antiplatelet agents and oral
Utilization of a CYP2C19 genotype-guided antiplatelet treatment algorithm over time in patients undergoing percutaneous coronary intervention
 Utilization of a CYP2C19 genotype-guided antiplatelet treatment algorithm over time in patients undergoing percutaneous coronary intervention Alexandra Cervantes, PharmD Candidate Faculty Mentor: Craig
Utilization of a CYP2C19 genotype-guided antiplatelet treatment algorithm over time in patients undergoing percutaneous coronary intervention Alexandra Cervantes, PharmD Candidate Faculty Mentor: Craig
Prasugrel a step ahead in antiplatelet therapy
 Prasugrel a step ahead in antiplatelet therapy VS Srinath, MD (Med), DNB (Cardiology) The burden of atherosclerotic disease in the United States and across the world is vast. Although the symptoms of atherosclerosis
Prasugrel a step ahead in antiplatelet therapy VS Srinath, MD (Med), DNB (Cardiology) The burden of atherosclerotic disease in the United States and across the world is vast. Although the symptoms of atherosclerosis
Prasugrel: Son of Clopidogrel or Distant Cousin? Disclosures. Objectives
 Prasugrel: Son of Clopidogrel or Distant Cousin? By John J. Bon, Pharm.D., BCPS Lead Clinical Pharmacist, Critical Care Summa Health System Disclosures I have no actual or potential conflict of interest
Prasugrel: Son of Clopidogrel or Distant Cousin? By John J. Bon, Pharm.D., BCPS Lead Clinical Pharmacist, Critical Care Summa Health System Disclosures I have no actual or potential conflict of interest
Controversies in PCI A young cardiologist s perspective
 Controversies in PCI A young cardiologist s perspective Antiplatelet Tx, PLT function monitoring should be mandatory CONTRA M. Valgimigli, MD, PhD Ferrara, Italy Euro-PCR Session @ ESC August 30th 2010,
Controversies in PCI A young cardiologist s perspective Antiplatelet Tx, PLT function monitoring should be mandatory CONTRA M. Valgimigli, MD, PhD Ferrara, Italy Euro-PCR Session @ ESC August 30th 2010,
Do We Need Platelet Function Assays?
 Do We Need Platelet Function Assays? Matthew J. Price MD Director, Cardiac Catheterization Laboratory Scripps Clinic, La Jolla, CA The Antiplatelet Effect of Clopidogrel Varies Widely Among Individuals
Do We Need Platelet Function Assays? Matthew J. Price MD Director, Cardiac Catheterization Laboratory Scripps Clinic, La Jolla, CA The Antiplatelet Effect of Clopidogrel Varies Widely Among Individuals
Joint Meeting of Coronary Revascularization 8 th to 9 th December 2017
 B Joint Meeting of Coronary Revascularization 8 th to 9 th December 2017 A Novel Clinical Application Combining Genotyping and Platetlet Function Testing In Patients With Acs - A Case Series Shirley Tan
B Joint Meeting of Coronary Revascularization 8 th to 9 th December 2017 A Novel Clinical Application Combining Genotyping and Platetlet Function Testing In Patients With Acs - A Case Series Shirley Tan
Speaker s name: Thomas Cuisset, MD, PhD
 Speaker s name: Thomas Cuisset, MD, PhD X I have the following potential conflicts of interest to report: x Consulting: Daiichi Sankyo, Eli Lilly Employment in industry Stockholder of a healthcare company
Speaker s name: Thomas Cuisset, MD, PhD X I have the following potential conflicts of interest to report: x Consulting: Daiichi Sankyo, Eli Lilly Employment in industry Stockholder of a healthcare company
Antiplatelet agents treatment
 Session III Comprehensive management of diabetic patients Antiplatelet agents treatment Chonnam National University Hospital Department of Internal Medicine Dong-Hyeok Cho CONTENTS Introduction Prothrombotic
Session III Comprehensive management of diabetic patients Antiplatelet agents treatment Chonnam National University Hospital Department of Internal Medicine Dong-Hyeok Cho CONTENTS Introduction Prothrombotic
A PRAGMATIC RANDOMIZED TRIAL OF CYP2C19 GENOTYPING IMPLEMENTATION FOLLOWING PERCUTANEOUS CORONARY INTERVENTION (PCI)
 A PRAGMATIC RANDOMIZED TRIAL OF CYP2C19 GENOTYPING IMPLEMENTATION FOLLOWING PERCUTANEOUS CORONARY INTERVENTION (PCI) Sony Tuteja, PharmD, MS Twitter @sony_tuteja Perelman School of Medicine at the University
A PRAGMATIC RANDOMIZED TRIAL OF CYP2C19 GENOTYPING IMPLEMENTATION FOLLOWING PERCUTANEOUS CORONARY INTERVENTION (PCI) Sony Tuteja, PharmD, MS Twitter @sony_tuteja Perelman School of Medicine at the University
Personalized Medicine in Real Time
 LABORATORY OF PERSONALIZED HEALTH LPH Personalized Medicine in Real Time DNA-Guided Clopidogrel (Plavix ) Management Pharmacogenetic Foundations and Case Study 1 Clopidogrel (Plavix ) Leading AntiPlatelet
LABORATORY OF PERSONALIZED HEALTH LPH Personalized Medicine in Real Time DNA-Guided Clopidogrel (Plavix ) Management Pharmacogenetic Foundations and Case Study 1 Clopidogrel (Plavix ) Leading AntiPlatelet
Clopidogrel vs New Antiplatelet Therapy (Prasugrel) Adnan Kastrati, MD Deutsches Herzzentrum, Technische Universität München, Germany
 Clopidogrel vs New Antiplatelet Therapy () Adnan Kastrati, MD Deutsches Herzzentrum, Technische Universität München, Germany Seoul, April 3, 21 Dual Antiplatelet Therapy for Stenting MACE, % 12 1 8 6 In
Clopidogrel vs New Antiplatelet Therapy () Adnan Kastrati, MD Deutsches Herzzentrum, Technische Universität München, Germany Seoul, April 3, 21 Dual Antiplatelet Therapy for Stenting MACE, % 12 1 8 6 In
Proton Pump Inhibitors increase Cardiovascular risk in patient taking Clopidogrel
 Proton Pump Inhibitors increase Cardiovascular risk in patient taking Clopidogrel Dr.A.K.M. Aminul Hoque Assoc. Prof. of Medicine. Dhaka Medical College. Clopidogrel Metabolism Clopidogrel is an inactive
Proton Pump Inhibitors increase Cardiovascular risk in patient taking Clopidogrel Dr.A.K.M. Aminul Hoque Assoc. Prof. of Medicine. Dhaka Medical College. Clopidogrel Metabolism Clopidogrel is an inactive
Pharmacogenomics of antiplatelet drugs
 PLATELET DISORDERS:MANAGING TOO MANY,TOO FEW Pharmacogenomics of antiplatelet drugs Marc S. Sabatine 1 and Jessica L. Mega 1 1 TIMI Study Group, Division of Cardiovascular Medicine, Brigham and Women s
PLATELET DISORDERS:MANAGING TOO MANY,TOO FEW Pharmacogenomics of antiplatelet drugs Marc S. Sabatine 1 and Jessica L. Mega 1 1 TIMI Study Group, Division of Cardiovascular Medicine, Brigham and Women s
Ticagrelor compared with clopidogrel in patients with acute coronary syndromes the PLATO trial
 compared with clopidogrel in patients with acute coronary syndromes the PLATO trial August 30, 2009 at 08.00 CET PLATO background In NSTE-ACS and STEMI, current guidelines recommend 12 months aspirin and
compared with clopidogrel in patients with acute coronary syndromes the PLATO trial August 30, 2009 at 08.00 CET PLATO background In NSTE-ACS and STEMI, current guidelines recommend 12 months aspirin and
Utility of Pharmacogenomics to Identify and Limit CV Risk. Christopher B. Granger, MD
 Utility of Pharmacogenomics to Identify and Limit CV Risk Christopher B. Granger, MD Disclosure Research contracts: AstraZeneca, Novartis, GSK, Sanofi-Aventis, BMS, Pfizer, The Medicines Company, and Boehringer
Utility of Pharmacogenomics to Identify and Limit CV Risk Christopher B. Granger, MD Disclosure Research contracts: AstraZeneca, Novartis, GSK, Sanofi-Aventis, BMS, Pfizer, The Medicines Company, and Boehringer
Personalized Antiplatelet Therapy: State of the Art
 Personalized Antiplatelet Therapy: State of the Art Paul A. Gurbel, M.D. Sinai Center for Thrombosis Research Associate Professor of Medicine Johns Hopkins University School of Medicine Baltimore, Maryland,
Personalized Antiplatelet Therapy: State of the Art Paul A. Gurbel, M.D. Sinai Center for Thrombosis Research Associate Professor of Medicine Johns Hopkins University School of Medicine Baltimore, Maryland,
Optimal antiplatelet and anticoagulant therapy for patients treated in STEMI network
 Torino 6 Joint meeting with Mayo Clinic Great Innovation in Cardiology 14-15 Ottobre 2010 Optimal antiplatelet and anticoagulant therapy for patients treated in STEMI network Diego Ardissino Ischemic vs
Torino 6 Joint meeting with Mayo Clinic Great Innovation in Cardiology 14-15 Ottobre 2010 Optimal antiplatelet and anticoagulant therapy for patients treated in STEMI network Diego Ardissino Ischemic vs
Effect of genetic and coexisting polymorphisms on platelet response to clopidogrel in Chinese Han patients with acute coronary syndrome
 c Indian Academy of Sciences RESEARCH ARTICLE Effect of genetic and coexisting polymorphisms on platelet response to clopidogrel in Chinese Han patients with acute coronary syndrome XU LIU 1,2,YULUO 3,YANLAI
c Indian Academy of Sciences RESEARCH ARTICLE Effect of genetic and coexisting polymorphisms on platelet response to clopidogrel in Chinese Han patients with acute coronary syndrome XU LIU 1,2,YULUO 3,YANLAI
תרופות מעכבות טסיות חדשות ד"ר אלי לב מנהל שרות הצנתורים ח השרון מרכז רפואי רבין
 תרופות מעכבות טסיות חדשות ד"ר אלי לב מנהל שרות הצנתורים ח השרון בי""י מרכז רפואי רבין 1. Why should clopidogrel be replaced? 2. Prasugrel 3. Ticagrelor 4. Conclusions CURE TRIAL ACS pts 20 % reduction
תרופות מעכבות טסיות חדשות ד"ר אלי לב מנהל שרות הצנתורים ח השרון בי""י מרכז רפואי רבין 1. Why should clopidogrel be replaced? 2. Prasugrel 3. Ticagrelor 4. Conclusions CURE TRIAL ACS pts 20 % reduction
3/23/2017. Angelika Cyganska, PharmD Austin T. Wilson, MS, PharmD Candidate Europace Oct;14(10): Epub 2012 Aug 24.
 Angelika Cyganska, PharmD Austin T. Wilson, MS, PharmD Candidate 2017 Explain the efficacy and safety of triple therapy, in regards to thromboembolic and bleeding risk Summarize the guideline recommendations
Angelika Cyganska, PharmD Austin T. Wilson, MS, PharmD Candidate 2017 Explain the efficacy and safety of triple therapy, in regards to thromboembolic and bleeding risk Summarize the guideline recommendations
Drug Eluting Stents Sometimes Fail ESC Stockholm 29 Set 2010 Stent Thrombosis Alaide Chieffo
 Drug Eluting Stents Sometimes Fail ESC Stockholm 29 Set 2010 Stent Thrombosis 11.45-12.07 Alaide Chieffo San Raffaele Scientific Institute, Milan, Italy Historical Perspective 25 20 15 10 5 0 Serruys 1991
Drug Eluting Stents Sometimes Fail ESC Stockholm 29 Set 2010 Stent Thrombosis 11.45-12.07 Alaide Chieffo San Raffaele Scientific Institute, Milan, Italy Historical Perspective 25 20 15 10 5 0 Serruys 1991
Angelika Cyganska, PharmD Austin T. Wilson, MS, PharmD Candidate 2017
 Angelika Cyganska, PharmD Austin T. Wilson, MS, PharmD Candidate 2017 Explain the efficacy and safety of triple therapy, in regards to thromboembolic and bleeding risk Summarize the guideline recommendations
Angelika Cyganska, PharmD Austin T. Wilson, MS, PharmD Candidate 2017 Explain the efficacy and safety of triple therapy, in regards to thromboembolic and bleeding risk Summarize the guideline recommendations
FACTOR Xa AND PAR-1 BLOCKER : ATLAS-2, APPRAISE-2 & TRACER TRIALS
 New Horizons In Atherothrombosis Treatment 2012 순환기춘계학술대회 FACTOR Xa AND PAR-1 BLOCKER : ATLAS-2, APPRAISE-2 & TRACER TRIALS Division of Cardiology, Jeonbuk National University Medical School Jei Keon Chae,
New Horizons In Atherothrombosis Treatment 2012 순환기춘계학술대회 FACTOR Xa AND PAR-1 BLOCKER : ATLAS-2, APPRAISE-2 & TRACER TRIALS Division of Cardiology, Jeonbuk National University Medical School Jei Keon Chae,
IMMATURE PLATELETS CLINICAL USE
 HAEMATOLOGY FEBRUARY 217 WHITE PAPER IMMATURE PLATELETS CLINICAL USE Identifying poor antiplatelet drug response and its risks early on Platelets are important cells for repairing endothelial lesions,
HAEMATOLOGY FEBRUARY 217 WHITE PAPER IMMATURE PLATELETS CLINICAL USE Identifying poor antiplatelet drug response and its risks early on Platelets are important cells for repairing endothelial lesions,
CYP2C19 polymorphisms in acute coronary syndrome patients undergoing clopidogrel therapy in Zhengzhou population
 CYP2C19 polymorphisms in acute coronary syndrome patients undergoing clopidogrel therapy in Zhengzhou population Y.M. Guo 1, Z.C. Zhao 1, L. Zhang 1, H.Z. Li 1, Z. Li 2 and H.L. Sun 1 1 Department of Cardiovascular
CYP2C19 polymorphisms in acute coronary syndrome patients undergoing clopidogrel therapy in Zhengzhou population Y.M. Guo 1, Z.C. Zhao 1, L. Zhang 1, H.Z. Li 1, Z. Li 2 and H.L. Sun 1 1 Department of Cardiovascular
Journal of the American College of Cardiology Vol. 50, No. 19, by the American College of Cardiology Foundation ISSN /07/$32.
 Journal of the American College of Cardiology Vol. 50, No. 19, 2007 2007 by the American College of Cardiology Foundation ISSN 0735-1097/07/$32.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2007.07.058
Journal of the American College of Cardiology Vol. 50, No. 19, 2007 2007 by the American College of Cardiology Foundation ISSN 0735-1097/07/$32.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2007.07.058
Learning Objectives. Epidemiology of Acute Coronary Syndrome
 Cardiovascular Update: Antiplatelet therapy in acute coronary syndromes PHILLIP WEEKS, PHARM.D., BCPS-AQ CARDIOLOGY Learning Objectives Interpret guidelines as they relate to constructing an antiplatelet
Cardiovascular Update: Antiplatelet therapy in acute coronary syndromes PHILLIP WEEKS, PHARM.D., BCPS-AQ CARDIOLOGY Learning Objectives Interpret guidelines as they relate to constructing an antiplatelet
Belinda Green, Cardiologist, SDHB, 2016
 Acute Coronary syndromes All STEMI ALL Non STEMI Unstable angina Belinda Green, Cardiologist, SDHB, 2016 Thrombus in proximal LAD Underlying pathophysiology Be very afraid for your patient Wellens
Acute Coronary syndromes All STEMI ALL Non STEMI Unstable angina Belinda Green, Cardiologist, SDHB, 2016 Thrombus in proximal LAD Underlying pathophysiology Be very afraid for your patient Wellens
Cangrelor: Is it the new CHAMPION for PCI? Robert Barcelona, PharmD, BCPS Clinical Pharmacy Specialist, Cardiac Intensive Care Unit November 13, 2015
 Cangrelor: Is it the new CHAMPION for PCI? Robert Barcelona, PharmD, BCPS Clinical Pharmacy Specialist, Cardiac Intensive Care Unit November 13, 2015 Objectives Review the pharmacology and pharmacokinetic
Cangrelor: Is it the new CHAMPION for PCI? Robert Barcelona, PharmD, BCPS Clinical Pharmacy Specialist, Cardiac Intensive Care Unit November 13, 2015 Objectives Review the pharmacology and pharmacokinetic
Effectiveness of Clopidogrel Dose Escalation to Normalize Active Metabolite Exposure and Antiplatelet Effects in CYP2C19 Poor Metabolizers
 Special Section: Clopidogrel Editor's Choice Effectiveness of Clopidogrel Dose Escalation to Normalize Active Metabolite Exposure and Antiplatelet Effects in CYP2C19 Poor Metabolizers The Journal of Clinical
Special Section: Clopidogrel Editor's Choice Effectiveness of Clopidogrel Dose Escalation to Normalize Active Metabolite Exposure and Antiplatelet Effects in CYP2C19 Poor Metabolizers The Journal of Clinical
Case Report Simultaneous Two-Vessel Subacute Stent Thrombosis Caused by Clopidogrel Resistance from CYP2C19 Polymorphism
 Case Reports in Medicine Volume 2016, Article ID 2312078, 4 pages http://dx.doi.org/10.1155/2016/2312078 Case Report Simultaneous Two-Vessel Subacute Stent Thrombosis Caused by Clopidogrel Resistance from
Case Reports in Medicine Volume 2016, Article ID 2312078, 4 pages http://dx.doi.org/10.1155/2016/2312078 Case Report Simultaneous Two-Vessel Subacute Stent Thrombosis Caused by Clopidogrel Resistance from
Clopidogrel is the second-leading drug sold worldwide,
 CONTROVERSIES IN CARDIOVASCULAR MEDICINE Do Platelet Function Testing and Genotyping Improve Outcome in Patients Treated With Antithrombotic Agents? The Role of Platelet Reactivity and Genotype Testing
CONTROVERSIES IN CARDIOVASCULAR MEDICINE Do Platelet Function Testing and Genotyping Improve Outcome in Patients Treated With Antithrombotic Agents? The Role of Platelet Reactivity and Genotype Testing
Clinical Cardiology and Cardiovascular Medicine
 Volume 1 Issue 1 P: 104 Page 1 of 7 Clinical Cardiology and Cardiovascular Medicine Review Article The Impact of Cytochrome P450 2C19 Polymorphism on Cardiovascular Events in Indonesian Patients with Coronary
Volume 1 Issue 1 P: 104 Page 1 of 7 Clinical Cardiology and Cardiovascular Medicine Review Article The Impact of Cytochrome P450 2C19 Polymorphism on Cardiovascular Events in Indonesian Patients with Coronary
Cytochrome P-450 Polymorphisms and Response to Clopidogrel
 The new england journal of medicine original article Cytochrome P-45 Polymorphisms and Response to Clopidogrel Jessica L. Mega, M.D., M.P.H., Sandra L. Close, Ph.D., Stephen D. Wiviott, M.D., Lei Shen,
The new england journal of medicine original article Cytochrome P-45 Polymorphisms and Response to Clopidogrel Jessica L. Mega, M.D., M.P.H., Sandra L. Close, Ph.D., Stephen D. Wiviott, M.D., Lei Shen,
Balancing Efficacy and Safety of P2Y12 Inhibitors for ACS Patients
 SYP.CLO-A.16.07.01 Balancing Efficacy and Safety of P2Y12 Inhibitors for ACS Patients dr. Hariadi Hariawan, Sp.PD, Sp.JP (K) TOPICS Efficacy Safety Consideration from Currently Available Antiplatelet Agents
SYP.CLO-A.16.07.01 Balancing Efficacy and Safety of P2Y12 Inhibitors for ACS Patients dr. Hariadi Hariawan, Sp.PD, Sp.JP (K) TOPICS Efficacy Safety Consideration from Currently Available Antiplatelet Agents
Clopidogrel Response Variability and Platelet Function Testing: Should Routine Practice Be Changed in Interventional Cardiology?
 Clopidogrel Response Variability and Platelet Function Testing: Should Routine Practice Be Changed in Interventional Cardiology? Matthew J. Price MD, FACC Director, Cardiac Catheterization Laboratory Scripps
Clopidogrel Response Variability and Platelet Function Testing: Should Routine Practice Be Changed in Interventional Cardiology? Matthew J. Price MD, FACC Director, Cardiac Catheterization Laboratory Scripps
Evaluation of Clopidogrel Resistance. in ischemic stroke patients.
 ORIGINAL ARTICLE Evaluation of Clopidogrel Resistance in Ischemic Stroke Patients Takuya Fukuoka, Daisuke Furuya, Hidetaka Takeda, Tomohisa Dembo, Harumitu Nagoya, Yuji Kato, Ichiro Deguchi, Hajime Maruyama,
ORIGINAL ARTICLE Evaluation of Clopidogrel Resistance in Ischemic Stroke Patients Takuya Fukuoka, Daisuke Furuya, Hidetaka Takeda, Tomohisa Dembo, Harumitu Nagoya, Yuji Kato, Ichiro Deguchi, Hajime Maruyama,
Ticagrelor compared with clopidogrel in patients with acute coronary syndromes the PLATelet Inhibition and patient Outcomes trial
 compared with clopidogrel in patients with acute coronary syndromes the PLATelet Inhibition and patient Outcomes trial Outcomes in patients with and planned PCI Ph.Gabriel Steg*, Stefan James, Robert A
compared with clopidogrel in patients with acute coronary syndromes the PLATelet Inhibition and patient Outcomes trial Outcomes in patients with and planned PCI Ph.Gabriel Steg*, Stefan James, Robert A
The Changing Landscape of Managing Patients with PAD- Update on the Evidence and Practice of Care in Patients with Peripheral Artery Disease
 Interventional Cardiology and Cath Labs The Changing Landscape of Managing Patients with PAD- Update on the Evidence and Practice of Care in Patients with Peripheral Artery Disease Manesh R. Patel MD Chief,
Interventional Cardiology and Cath Labs The Changing Landscape of Managing Patients with PAD- Update on the Evidence and Practice of Care in Patients with Peripheral Artery Disease Manesh R. Patel MD Chief,
Upcoming Evidence and Practice of Optimal Antiplatelet Therapy in DES Era?
 Upcoming Evidence and Practice of ptimal Antiplatelet Therapy in DES Era? Polymorphism in Metabolism of Clopidogrel and Its Clinical Implications and Management Alexandra Lansky MD Columbia University
Upcoming Evidence and Practice of ptimal Antiplatelet Therapy in DES Era? Polymorphism in Metabolism of Clopidogrel and Its Clinical Implications and Management Alexandra Lansky MD Columbia University
QUT Digital Repository:
 QUT Digital Repository: http://eprints.qut.edu.au/ This is the author s version of this journal article. Published as: Doggrell, Sheila (2010) New drugs for the treatment of coronary artery syndromes.
QUT Digital Repository: http://eprints.qut.edu.au/ This is the author s version of this journal article. Published as: Doggrell, Sheila (2010) New drugs for the treatment of coronary artery syndromes.
Antithrombotic Therapy in Patients with Atrial Fibrillation
 Antithrombotic Therapy in Patients with Atrial Fibrillation June Soo Kim, M.D., Ph.D. Department of Medicine Cardiac & Vascular Center, Samsung Medical Center Sungkyunkwan University School of Medicine
Antithrombotic Therapy in Patients with Atrial Fibrillation June Soo Kim, M.D., Ph.D. Department of Medicine Cardiac & Vascular Center, Samsung Medical Center Sungkyunkwan University School of Medicine
Cardiovascular pharmacogenomics: ready for prime time?
 Cardiovascular pharmacogenomics: ready for prime time? Simon de Denus, pharmacist, MSc (Pharm), PhD Université de Montréal Beaulieu-Saucier Chair in Pharmacogenomics Assistant professor, Faculty of Pharmacy,
Cardiovascular pharmacogenomics: ready for prime time? Simon de Denus, pharmacist, MSc (Pharm), PhD Université de Montréal Beaulieu-Saucier Chair in Pharmacogenomics Assistant professor, Faculty of Pharmacy,
Oral anticoagulation/antiplatelet therapy in the secondary prevention of ACS patients the cost of reducing death!
 Oral anticoagulation/antiplatelet therapy in the secondary prevention of ACS patients the cost of reducing death! Robert C. Welsh, MD, FRCPC Associate Professor of Medicine Director, Adult Cardiac Catheterization
Oral anticoagulation/antiplatelet therapy in the secondary prevention of ACS patients the cost of reducing death! Robert C. Welsh, MD, FRCPC Associate Professor of Medicine Director, Adult Cardiac Catheterization
The Pharmacogenetics of Clopidogrel
 The Pharmacogenetics of Clopidogrel CANNeCTIN Cutting-Edge Pharmacogenetics Symposium May 22, 2009 Marc S. Sabatine, MD, MPH Investigator, TIMI Study Group Associate Physician, Cardiovascular Division,
The Pharmacogenetics of Clopidogrel CANNeCTIN Cutting-Edge Pharmacogenetics Symposium May 22, 2009 Marc S. Sabatine, MD, MPH Investigator, TIMI Study Group Associate Physician, Cardiovascular Division,
Pharmacy Drug Class Review
 Pharmacy Drug Class Review September 22, 2009 Disclaimer: Specific agents may have variations Oral antiplatelet agents Focus on dual therapy, genetic variants, and proton pump inhibitors 1. Antiplatelet
Pharmacy Drug Class Review September 22, 2009 Disclaimer: Specific agents may have variations Oral antiplatelet agents Focus on dual therapy, genetic variants, and proton pump inhibitors 1. Antiplatelet
ORIGINAL ARTICLE INTRODUCTION
 ORIGINAL ARTICLE Trends of Platelet Inhibition in Different Clopidogrel Pretreatment Patterns in Malaysian Patients Undergoing Elective Percutaneous Coronary Intervention Wen Ni Tiong, MSc, Melissa Mejin,
ORIGINAL ARTICLE Trends of Platelet Inhibition in Different Clopidogrel Pretreatment Patterns in Malaysian Patients Undergoing Elective Percutaneous Coronary Intervention Wen Ni Tiong, MSc, Melissa Mejin,
P2Y 12 blockade. To load or not to load before the cath lab?
 UPDATE ON ANTITHROMBOTICS IN ACUTE CORONARY SYNDROMES P2Y 12 blockade. To load or not to load before the cath lab? Franz-Josef Neumann Personal: None Institutional: Conflict of Interest Speaker honoraria,
UPDATE ON ANTITHROMBOTICS IN ACUTE CORONARY SYNDROMES P2Y 12 blockade. To load or not to load before the cath lab? Franz-Josef Neumann Personal: None Institutional: Conflict of Interest Speaker honoraria,
Clinical Pharmacogenetics Implementation Consortium Guidelines for Cytochrome P450-2C19 (CYP2C19) Genotype and Clopidogrel Therapy
 nature publishing group Clinical Pharmacogenetics Implementation Consortium Guidelines for Cytochrome P450-2C19 (CYP2C19) Genotype and Clopidogrel Therapy SA Scott 1, K Sangkuhl 2, EE Gardner 3, CM Stein
nature publishing group Clinical Pharmacogenetics Implementation Consortium Guidelines for Cytochrome P450-2C19 (CYP2C19) Genotype and Clopidogrel Therapy SA Scott 1, K Sangkuhl 2, EE Gardner 3, CM Stein
DECLARATION OF CONFLICT OF INTEREST. Lecture fees: AstraZeneca, Ely Lilly, Merck.
 DECLARATION OF CONFLICT OF INTEREST Lecture fees: AstraZeneca, Ely Lilly, Merck. Risk of stopping dual therapy. S D Kristensen, FESC Aarhus Denmark Acute coronary syndrome: coronary thrombus Platelets
DECLARATION OF CONFLICT OF INTEREST Lecture fees: AstraZeneca, Ely Lilly, Merck. Risk of stopping dual therapy. S D Kristensen, FESC Aarhus Denmark Acute coronary syndrome: coronary thrombus Platelets
Αντιαιμοπεταλιακη αγωγη (ποια, πο τε και για πο σο)
 Αντιαιμοπεταλιακη αγωγη (ποια, πο τε και για πο σο) Dimitrios Alexopoulos, MD, FESC, FACC Cardiology Department, Patras University Hospital, Patras, Rio, Greece. Patras University Hospital I, Dimitrios
Αντιαιμοπεταλιακη αγωγη (ποια, πο τε και για πο σο) Dimitrios Alexopoulos, MD, FESC, FACC Cardiology Department, Patras University Hospital, Patras, Rio, Greece. Patras University Hospital I, Dimitrios
Περιφερική Αρτηριακή Νόσος Νόσος Καρωτίδων
 Νεώτερα δεδομένα στην αντιαιμοπεταλιακή αγωγή που θα επηρεάσουν τις μελλοντικές κατευθυντήριες οδηγίες Περιφερική Αρτηριακή Νόσος Νόσος Καρωτίδων Μιλτιάδης Ματσάγκας, MD, PhD, FEBVS Αναπληρωτής Καθηγητής
Νεώτερα δεδομένα στην αντιαιμοπεταλιακή αγωγή που θα επηρεάσουν τις μελλοντικές κατευθυντήριες οδηγίες Περιφερική Αρτηριακή Νόσος Νόσος Καρωτίδων Μιλτιάδης Ματσάγκας, MD, PhD, FEBVS Αναπληρωτής Καθηγητής
Why and How Should We Switch Clopidogrel to Prasugrel?
 Case Presentation Why and How Should We Switch Clopidogrel to Prasugrel? Shaul Atar Western Galilee Medical Center Nahariya, ISRAEL Case Description A 67 Y. Old Pt. admitted to IM with anginal CP. DM,
Case Presentation Why and How Should We Switch Clopidogrel to Prasugrel? Shaul Atar Western Galilee Medical Center Nahariya, ISRAEL Case Description A 67 Y. Old Pt. admitted to IM with anginal CP. DM,
An Update on Oral Anti-platelet therapy in patients with non-st Myocardial Infarction. Disclosures
 An Update on Oral Anti-platelet therapy in patients with non-st Myocardial Infarction R. Scott Wright, MD, FACC, FESC, FAHA, Professor of Medicine Mayo Clinic Fall Managed Care Forum November 2013 3098590-1
An Update on Oral Anti-platelet therapy in patients with non-st Myocardial Infarction R. Scott Wright, MD, FACC, FESC, FAHA, Professor of Medicine Mayo Clinic Fall Managed Care Forum November 2013 3098590-1
Is There A LIfe for DES after discontinuation of Clopidogrel
 EMBARGOED UNTIL 3:45pm CT, Sunday, 11/16/14 - FOR MEDIA BACKGROUND ONLY - Do Not Publish Chicago 2014 Is There A LIfe for DES after discontinuation of Clopidogrel Six-month versus 24-month dual antiplatelet
EMBARGOED UNTIL 3:45pm CT, Sunday, 11/16/14 - FOR MEDIA BACKGROUND ONLY - Do Not Publish Chicago 2014 Is There A LIfe for DES after discontinuation of Clopidogrel Six-month versus 24-month dual antiplatelet
Preoperative Management of Patients Receiving Antithrombotics
 Preoperative Management of Patients Receiving Antithrombotics Bleeding complications remain an important concern for most surgical procedures. Attempts to minimize the risk of these complications by removing
Preoperative Management of Patients Receiving Antithrombotics Bleeding complications remain an important concern for most surgical procedures. Attempts to minimize the risk of these complications by removing
ISAR-CHOICE 2. Randomized Trial of 75 mg vs 150 mg of Daily Clopidogrel in Patients Undergoing PCI
 ISAR-CHOICE 2 Randomized Trial of 75 mg vs 150 mg of Daily Clopidogrel in Patients Undergoing PCI Adnan Kastrati Deutsches Herzzentrum, Munich, GERMANY Variability in Platelet Response to Clopidogrel Serebruany
ISAR-CHOICE 2 Randomized Trial of 75 mg vs 150 mg of Daily Clopidogrel in Patients Undergoing PCI Adnan Kastrati Deutsches Herzzentrum, Munich, GERMANY Variability in Platelet Response to Clopidogrel Serebruany
The Challenge. Warfarin or Novel Oral Anti-Coagulants in the PCI patient? Anticoagulation/Stroke
 Anticoagulation/Stroke Warfarin v new oral anticoagulants post PCI Warfarin or Novel Oral Anti-Coagulants in the PCI patient? Gerry Devlin Chairs: Phillip Matsis & Tony Scott Gerry Devlin Honorary Associate
Anticoagulation/Stroke Warfarin v new oral anticoagulants post PCI Warfarin or Novel Oral Anti-Coagulants in the PCI patient? Gerry Devlin Chairs: Phillip Matsis & Tony Scott Gerry Devlin Honorary Associate
PROTON PUMP INHIBITOR AND CLOPIDOGREL INTERACTION: Am J Gastroenterol Jan;105(1): Epub 2009 Nov 10.
 PROTON PUMP INHIBITOR AND CLOPIDOGREL INTERACTION: FACT OR FICTION? 本檔僅供內部教學使用檔案內所使用之照片之版權仍屬於原期刊公開使用時, 須獲得原期刊之同意授權 Am J Gastroenterol. 2010 Jan;105(1):34-41. Epub 2009 Nov 10. Introduction Current consensus
PROTON PUMP INHIBITOR AND CLOPIDOGREL INTERACTION: FACT OR FICTION? 本檔僅供內部教學使用檔案內所使用之照片之版權仍屬於原期刊公開使用時, 須獲得原期刊之同意授權 Am J Gastroenterol. 2010 Jan;105(1):34-41. Epub 2009 Nov 10. Introduction Current consensus
The Korean Society of Cardiology COI Disclosure
 The Korean Society of Cardiology COI Disclosure Name of First Author: Yongwhi Park The authors have no financial conflicts of interest to disclose concerning the presentation 2017 Annual Spring Scientific
The Korean Society of Cardiology COI Disclosure Name of First Author: Yongwhi Park The authors have no financial conflicts of interest to disclose concerning the presentation 2017 Annual Spring Scientific
Disclosures. Theodore A. Bass MD, FSCAI. The following relationships exist related to this presentation. None
 SCAI Fellows Course December 10, 2013 Disclosures Theodore A. Bass MD, FSCAI The following relationships exist related to this presentation None Current Controversies on DAPT in PCI Which drug? When to
SCAI Fellows Course December 10, 2013 Disclosures Theodore A. Bass MD, FSCAI The following relationships exist related to this presentation None Current Controversies on DAPT in PCI Which drug? When to
It is well recognized that there is substantial interindividual. Original Article
 Original Article CYP2C19 Genotype Has a Greater Effect on Adverse Cardiovascular Outcomes Following Percutaneous Coronary Intervention and in Asian Populations Treated With Clopidogrel A Meta-Analysis
Original Article CYP2C19 Genotype Has a Greater Effect on Adverse Cardiovascular Outcomes Following Percutaneous Coronary Intervention and in Asian Populations Treated With Clopidogrel A Meta-Analysis
Calcium-Channel Blockers Attenuate the Antiplatelet Effect of Clopidogrel
 ORIGINAL RESEARCH ARTICLE Calcium-Channel Blockers Attenuate the Antiplatelet Effect of Clopidogrel Thomas Gremmel, 1 Markus Durstberger, 1 Beate Eichelberger, 2 Renate Koppensteiner 1 & Simon Panzer 2
ORIGINAL RESEARCH ARTICLE Calcium-Channel Blockers Attenuate the Antiplatelet Effect of Clopidogrel Thomas Gremmel, 1 Markus Durstberger, 1 Beate Eichelberger, 2 Renate Koppensteiner 1 & Simon Panzer 2
Journal of the American College of Cardiology Vol. 45, No. 9, by the American College of Cardiology Foundation ISSN /05/$30.
 Journal of the American College of Cardiology Vol. 45, No. 9, 2005 2005 by the American College of Cardiology Foundation ISSN 0735-1097/05/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2005.01.030
Journal of the American College of Cardiology Vol. 45, No. 9, 2005 2005 by the American College of Cardiology Foundation ISSN 0735-1097/05/$30.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2005.01.030
Investigating Real-World Clopidogrel Pharmacogenetics in Stroke Using a Bioresource Linked to Electronic Medical Records
 Investigating Real-World Clopidogrel Pharmacogenetics in Stroke Using a Bioresource Linked to Electronic Medical Records Aleksi Tornio 1, Rob Flynn 2, Steve Morant 2, Elena Velten 2, Colin N. A. Palmer
Investigating Real-World Clopidogrel Pharmacogenetics in Stroke Using a Bioresource Linked to Electronic Medical Records Aleksi Tornio 1, Rob Flynn 2, Steve Morant 2, Elena Velten 2, Colin N. A. Palmer
CYP2C19 Phenotype, Stent Thrombosis, Myocardial Infarction, and Mortality in Patients with Coronary Stent Placement in a Chinese Population
 CYP2C19 Phenotype, Stent Thrombosis, Myocardial Infarction, and Mortality in Patients with Coronary Stent Placement in a Chinese Population Xiang Xie 1, Yi-Tong Ma 1 *, Yi-Ning Yang 1, Xiao-Mei Li 1, Xiang
CYP2C19 Phenotype, Stent Thrombosis, Myocardial Infarction, and Mortality in Patients with Coronary Stent Placement in a Chinese Population Xiang Xie 1, Yi-Tong Ma 1 *, Yi-Ning Yang 1, Xiao-Mei Li 1, Xiang
Adults With Diagnosed Diabetes
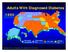 Adults With Diagnosed Diabetes 1990 No data available Less than 4% 4%-6% Above 6% Mokdad AH, et al. Diabetes Care. 2000;23(9):1278-1283. Adults With Diagnosed Diabetes 2000 4%-6% Above 6% Mokdad AH, et
Adults With Diagnosed Diabetes 1990 No data available Less than 4% 4%-6% Above 6% Mokdad AH, et al. Diabetes Care. 2000;23(9):1278-1283. Adults With Diagnosed Diabetes 2000 4%-6% Above 6% Mokdad AH, et
2010, Metzler Helfried
 Perioperative Strategies in Patients on Dual Antiplatelet Drug Therapy: Noncardiac Surgery H. Metzler Department of Anaesthesiology and Intensive Care Medicine Medical University of Graz, Austria What
Perioperative Strategies in Patients on Dual Antiplatelet Drug Therapy: Noncardiac Surgery H. Metzler Department of Anaesthesiology and Intensive Care Medicine Medical University of Graz, Austria What
Comparison of Omeprazole and Pantoprazole Influence on a High 150-mg Clopidogrel Maintenance Dose
 Journal of the American College of Cardiology Vol. 54, No. 13, 2009 2009 by the American College of Cardiology Foundation ISSN 0735-1097/09/$36.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2009.05.050
Journal of the American College of Cardiology Vol. 54, No. 13, 2009 2009 by the American College of Cardiology Foundation ISSN 0735-1097/09/$36.00 Published by Elsevier Inc. doi:10.1016/j.jacc.2009.05.050
Korea University Guro Hospital, Seoul, Korea * Chonnam National University Hospital, Gwangju, Korea
 Left Main Disease versus Non Left Main Disease in Acute Myocardial Infarction Patients in Real world Clinical Practice : Lessons from Korea Acute Myocardial Infarction Registry (KAMIR) Seung-Woon Rha*,
Left Main Disease versus Non Left Main Disease in Acute Myocardial Infarction Patients in Real world Clinical Practice : Lessons from Korea Acute Myocardial Infarction Registry (KAMIR) Seung-Woon Rha*,
Diversity of platelet function and genetic polymorphism in clopidogrel-treated Chinese patients
 Diversity of platelet function and genetic polymorphism in clopidogrel-treated Chinese patients B. Sun 1, J. Li 2, M. Dong 1, L. Yang 2, C. Wu 1, L. Zhu 1 and Y.L. Cong 2 1 Clinical Laboratory, The 309th
Diversity of platelet function and genetic polymorphism in clopidogrel-treated Chinese patients B. Sun 1, J. Li 2, M. Dong 1, L. Yang 2, C. Wu 1, L. Zhu 1 and Y.L. Cong 2 1 Clinical Laboratory, The 309th
Quale terapia antiaggregante nello STEMI? Prasugrel vs ticagrelor
 Quale terapia antiaggregante nello STEMI? Prasugrel vs ticagrelor Leonardo Bolognese Cardiovascular Department, Arezzo, Italy Platelet Reactivity in Patients with STEMI Undergoing Primary PCI Campo G et
Quale terapia antiaggregante nello STEMI? Prasugrel vs ticagrelor Leonardo Bolognese Cardiovascular Department, Arezzo, Italy Platelet Reactivity in Patients with STEMI Undergoing Primary PCI Campo G et
