Rizatriptan in migraineurs with unilateral cranial autonomic symptoms: a double-blind trial
|
|
|
- Agnes Hicks
- 5 years ago
- Views:
Transcription
1 J Headache Pain (2012) 13: DOI /s y CLINICAL TRIALS Rizatriptan in migraineurs with unilateral cranial autonomic symptoms: a double-blind trial Piero Barbanti Luisa Fofi Valentina Dall Armi Cinzia Aurilia Gabriella Egeo Nicola Vanacore Stefano Bonassi Received: 3 February 2012 / Accepted: 12 March 2012 / Published online: 30 March 2012 The Author(s) This article is published with open access at Springerlink.com Abstract The objective and background is to confirm in a double-blind, placebo-controlled study the high triptan response rates we had previously reported in an open study in migraine patients with unilateral cranial autonomic symptoms. In this randomized, double-blind, placebocontrolled study 80 migraineurs with unilateral cranial autonomic symptoms were assigned to receive rizatriptan 10 mg wafer or placebo (ratio 1:1) and treated for a single moderate or severe migraine attack. The primary endpoints were pain freedom at 2 h and total migraine freedom at 2 h. Secondary endpoints included pain relief, no associated symptoms and sustained pain freedom or relief. Significantly more patients reported pain freedom at 2 h after taking rizatriptan (54 %) than after placebo (8 %) (therapeutic gain 46 % [28 %; 64 %]; P \ 0.001). Similarly, significantly more patients reported total migraine freedom at 2 h after rizatriptan (51 %) than after placebo (8 %) (therapeutic gain 43 % [26 %; 61 %]; P \ 0.001). Rizatriptan was also more effective than placebo on most secondary endpoints. We confirm in a placebo-controlled study our previous data suggesting that the presence of unilateral cranial autonomic symptoms in migraineurs predicts a P. Barbanti (&) L. Fofi C. Aurilia G. Egeo Headache and Pain Unit, Department of Neurological, Motor and Sensorial Sciences, IRCCS San Raffaele Pisana, Via della Pisana 235, Rome, Italy piero.barbanti@sanraffaele.it V. Dall Armi S. Bonassi Unit of Clinical and Molecular Epidemiology, IRCCS San Raffaele Pisana, Via della Pisana 235, Rome, Italy N. Vanacore National Center of Epidemiology, National Institute of Health, Rome, Italy positive response to triptans, probably owing to intense trigeminal peripheral afferent activation which strongly recruits peripheral neurovascular 5-HT1B/1D receptors. Acute and preventive pharmacological trials in migraine should focus also on this subset of migraine patients. Keywords Migraine Cranial autonomic symptoms Trigemino-autonomic reflex Treatment Rizatriptan Introduction Migraine pain depends on trigeminovascular system activation that induces vasoactive neuropeptide release from trigeminal perivascular axons leading to neurogenic inflammation that stimulates meningeal sensory fibers and transmits nociceptive information centrally, along the trigeminal axons, to the trigeminal nucleus caudalis, and from there rostrally to the thalamus and cortex [1]. In some migraineurs, activating the trigeminovascular system may trigger the efferent parasympathetic arm of the trigeminoautonomic reflex [2]. In these migraineurs, whose prevalence ranges from 26.4 % in the general migraine population to 45.8 % in patients attending a Headache Center, the clinical hallmarks are unilateral cranial autonomic symptoms (UAs) such as conjunctival injection, lacrimation, nasal congestion/ rhinorrhea, ptosis, eyelid swelling or forehead/facial sweating, singly or combined. Migraine headache is usually more strictly unilateral and more severe in patients with UAs than in the general migraine population [3, 4]. In an open study with sumatriptan 50 mg, we previously suggested that UAs in migraineurs may predict a positive response to triptans [5]. Their possible predictive value received further support from a study describing detectable serum vasoactive intestinal polypeptide (VIP), the
2 408 J Headache Pain (2012) 13: biochemical marker of parasympathetic activation, in the external jugular blood in one-half of migraine patients who responded to rizatriptan [6]. The complex yet clinically important issue of whether UAs predict triptan responses in migraine therefore awaits confirmatory data from a placebo-controlled study. Having this information would allow more tailored therapy for treating acute migraine. In this study, to find out more about triptan response rates in patients with UAs, in a randomized, double-blind, placebo-controlled, parallel-group study we used oral rizatriptan 10 mg, one of the most commonly used and effective triptans [7], for acute therapy in a consecutive series of patients with migraine and UAs. To do so we chose stringent primary outcome measures for treatment efficacy including pain and total migraine freedom at 2 h [8]. We also ensured that patients waited before taking oral rizatriptan until their headache became moderate to severe. Methods Study population and design For this randomized, double-blind, placebo-controlled, parallel-group, outpatient study to assess the efficacy of rizatriptan 10 mg wafer in treating a single acute migraine attack in migraineurs with UAs, patients were consecutively recruited from our Headache and Pain Unit. Patients were eligible for the study if they were C18 years of age, had a history of migraine with or without aura for at least 1 year, and in the 2 months before screening had experienced 1 8 moderate or severe migraine attacks per month [9]. Patients taking migraine prevention medication were allowed to enter the study if their prescribed daily dose had remained unchanged during the 3 months before screening. Patients taking propranolol, methysergide, serotonin norepinephrine reuptake inhibitors, selective serotonin reuptake inhibitors, or monoamine oxidase inhibitors within 14 months of the screening visit were not eligible. Patients with history or clinical evidence of ischemic heart disease or symptoms or findings consistent with ischemic heart disease, coronary artery vasospasm, or other significant underlying cardiovascular disease and those with clinical, laboratory, or electrocardiographic evidence of uncontrolled hypertension, uncontrolled diabetes, or significant pulmonary, renal, hepatic, endocrine, or other systemic disease were also excluded. Patients attended the hospital for a screening visit to assess eligibility and undertake physical examinations. The interview determined whether patients experienced UAs by asking the following question: During the migraine attack do you also have at least one of the following symptoms: unilateral conjunctival injection, lacrimation, nasal congestion/rhinorrhea, ptosis, eyelid swelling or forehead/facial sweating? [3]. Patients who met all the study entry criteria were enrolled and randomly allocated to receive either rizatriptan 10 mg wafer or placebo (ratio 1:1). Patients were encouraged to take migraine medication as soon as their migraine headache became moderate or severe. If the moderate or severe migraine headache persisted 2 h after dosing, or recurred within 24 h, patients had the option of taking their own rescue medication but triptans and ergot derivatives were prohibited for 24 h after study medication intake. This protocol was approved by the institutional review board at San Raffaele Pisana Scientific Institute and have therefore been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki. All patients gave their informed consent prior to their inclusion in the study. Data collection During the 24 h after taking the initial dose of study medication, patients recorded subjective assessments of pain severity, presence or absence of associated symptoms, use of rescue medication, and the onset of, if applicable, headache recurrence at specified time points in a paper migraine diary. Subjective adverse experiences were recorded in the diary and rated as mild, moderate or severe. Patients were asked to return to the study site as soon as possible and[7 days after treatment to allow physicians to review the diary, assess medication compliance and monitor tolerability. Headache severity was recorded using a four-grade scale (no pain, mild pain, moderate pain, severe pain) at six time points, baseline (time of taking study drug) and at 0.5, 1, 1.5, 2, and 24 h thereafter. The presence or absence of associated symptoms (nausea, vomiting, photophobia, or phonophobia) was recorded at the same time points as the headache severity ratings. For those patients who had pain relief (pain reduction to mild or none) or pain freedom (no pain) at 2 h, another variable recorded was the presence or absence of headache worsening (recurrence) within 2 24 h. In all patients we also recorded use of rescue medication within 24 h. Tolerability and safety were assessed by asking patients to report spontaneous adverse events (AEs). Outcome measures for efficacy The primary endpoints were pain freedom at 2 h and total migraine freedom (pain freedom and absence of associated symptoms) at 2 h. Secondary endpoints were pain freedom at 0.5, 1 and 1.5 h, pain relief at 0.5, 1, 1.5 and 2 h; absence of nausea, photophobia and phonophobia at 0.5, 1, 1.5 and 2 h; 2 24 h sustained pain relief (pain relief from 2 to 24 h
3 J Headache Pain (2012) 13: without rescue medication); and 2 24 h sustained pain freedom (pain freedom from 2 to 24 h without rescue medication). Randomization sequence generation The random allocation sequence, including details of any restrictions was produced by Computer Generated Masked Allocation Schedule, Blocking Factor: 4. The allocation sequence was generated by the Pharmaceutical Research and Development Labeling System, Merck & Co., USA. Numbered containers were used to implement the random allocation sequence. The sequence was concealed until unblinding was necessary. The principal investigator assigned participants to the groups, following the masked allocation schedule numbers. All the participants, those administering the treatments and those assessing the outcomes were blinded to group assignment. The success of blinding was guaranteed using similar shaped containers and tablets and by the sealed masked allocation schedule. Sample size calculation Sample size was calculated assuming that the therapeutic gain over placebo for migraineurs with UAs treated with rizatriptan 10 mg is similar to that reported in the general migraine population, i.e., 31 % pain freedom at 2 h and 33 % pain relief at 2 h [10]. Given the limited published evidence assessing the effect of rizatriptan 10 mg on the rate of total migraine freedom, the number of patients to be recruited was estimated according to the effect of rizatriptan 10 mg on pain freedom at 2 h. Assuming a type I error rate of 0.05 %, a statistical power of 90 %, and a pain freedom rate at 2 h of 41 % in the treated group versus 10 % in the placebo group [10], we estimated that the study would require a minimum number of 78 patients (39 to be treated with rizatriptan 10 mg and 39 with placebo). A total number of 100 patients was considered for inclusion in the study (Fig. 1). Statistical analysis The presence of heterogeneity between the two treatment groups according to demographic and baseline characteristics was assessed using the unpaired t test for continuous variables, and the Pearson s Chi-square test (or Fisher s exact test with expected cell frequencies \5 %) for categorical variables. A binomial regression analysis was run to test the effect of rizatriptan 10 mg versus placebo, and the therapeutic gain (defined as the change in outcome induced by rizatriptan 10 mg minus the placebo effect) with its relative 95 % confidence interval (CI), was estimated for all outcome measures. The absence of nausea, phonophobia and photophobia after treatment was analyzed in a subset of individuals who experienced these symptoms at baseline (when the attack began). To remove the effect of rescue medication, data were analyzed after excluding patients who took a rescue medication before the scheduled time point of outcome measurement. A P value\0.05 was the threshold for statistical significance. To take into account the type I error inflation generated by multiple comparisons, the Bonferroni correction was applied (new threshold for type I error \5 %: P \ ). STATA/SE V10 was used for all statistical analyses. Fig. 1 Flow-chart of patients 100 screened 10 did not meet entry criteria 1 declined to participate 89 randomized 45 assigned to rizatriptan 10 mg 44 assigned to placebo 4 not treated (no event) 5 not treated (no event) 41 assessed for efficacy 39 assessed for efficacy
4 410 J Headache Pain (2012) 13: Table 1 Socio-demographic and clinical characteristics of migraine patients with unilateral cranial autonomic symptoms Continuous variables are reported as mean ± SD and categorical variables as frequencies (%) Statistically significant P values (P \ 0.05) are in bold MwA migraine without aura, MA migraine with aura, UAs unilateral cranial autonomic symptoms Rizatriptan 10 mg (41 patients) Placebo (39 patients) P values Sex Female 33 (80 %) 34 (87 %) Male 8 (20 %) 5 (13 %) Age (years) ± ± Body mass index ± ± Illness duration (years) ± ± Family history of migraine 34 (82.9 %) 27 (69.2 %) MwA 39 (95.1 %) 36 (92.3 %) MwA? MA 2 (4.9 %) 3 (7.7 %) Attack/month 5.68 ± ± Attack duration (h) B24 20 (48.8 %) 21 (53.8 %) [24 21 (51.2 %) 18 (46.2 %) Pain location Unilateral, alternating side 12 (29.3 %) 7 (18 %) Unilateral, same side 25 (61 %) 28 (71.8 %) Bilateral 4 (9.8 %) 4 (10.3 %) Pain quality Pulsating 29 (70.7 %) 24 (61.5 %) Pressing 5 (12.2 %) 10 (25.7 %) Other 7 (17.1 %) 5 (12.8 %) Pain intensity Moderate 29 (70.7 %) 26 (66.7 %) Severe 12 (29.3 %) 13 (33.3 %) UAs (n) (62.5 %) 26 (70.3 %) [1 15 (37.5 %) 11 (29.7 %) Presence of allodynia 18 (43.9 %) 23 (59.0 %) Triptan naïve 18 (43.9 %) 24 (61.5 %) Current prophylaxis 6 (14.6 %) 15 (28.4 %) Menopause 7 (17.1 %) 6 (16.2 %) Oral contraceptives 4 (9.8 %) 3 (8.1 %) Comorbidities 18 (43.9 %) 18 (46.1 %) Results Efficacy Of the 100 outpatients screened, 10 failed to meet all the inclusion criteria, 1 declined to participate and 89 were randomly assigned to rizatriptan 10 mg wafer (45 patients) or placebo (44 patients). Four patients in the rizatriptan group and 5 patients in the placebo group were excluded from the efficacy assessment because they lacked a qualifying event (i.e., moderately or severely intense migraine attack) (Fig. 1). Demographic features and baseline characteristics were similar in the active drug and placebo groups (Table 1). Binomial regression analysis showed that a significantly larger percentage of patients assigned to rizatriptan than to placebo reported pain freedom at 2 h post dosing (54 % [95 % CI 38, 70 %] vs. 8 % [95 % CI -1, 17 %]) (P \ 0.001) (Fig. 2) and total migraine freedom at 2 h post dosing (51 % [95 % CI 36, 67 %] vs. 8 % [95 % CI -1, 17 %]) (P \ 0.001) (Fig. 3). Active treatment was also more effective than placebo on all the other outcome measures, pain free at 1.5 h, pain relief at, 1.5 and 2 h, no nausea at 2 h, no photophobia at 1.5 and 2 h, 2 24 h sustained pain relief and 2 24 h sustained pain
5 J Headache Pain (2012) 13: Fig. 2 Pain freedom at various time points after oral rizatriptan 10 mg (diamonds) intake during an acute migraine attack. Squares indicate the therapeutic gain (rizatriptan response placebo response). Asterisks indicate a statistically significant (P \ 0.05) therapeutic gain Pain freedom (%) p<0.001* p=0.026* p<0.001* p=0.087 p= minutes 1 hour 1.5 hours 2 hours hours Fig. 3 Total migraine freedom at various time points after oral rizatriptan 10 mg (diamonds) intake during an acute migraine attack. Squares indicate the therapeutic gain (rizatriptan response placebo response). Asterisks indicate a statistically significant (P \ 0.05) therapeutic gain Total migraine freedom (%) p=0.004* p<0.001* p=0.004* p=0.986 p= minutes 1 hour 1 hour 1.5 hours 1.5 hours 2 hours 2 hours 24 hours freedom (Table 2). Someother endpoints failed to reach statistical significance after Bonferroni correction, i.e., pain relief at 1 h, total migraine free at 1.5 h, no phonophobia at h. The recurrence rate was 17.4 % among rizatriptan responders and 25 % among placebo responders. Patients assigned to rizatriptan resorted to rescue medication less frequently than those assigned to placebo (15 vs. 41 %). Tolerability and reported adverse events Although the study primarily investigated efficacy, when we calculated the total number of AEs in each group before patients used rescue medication, the incidence of AEs was similar for rizatriptan and placebo (12 and 10 %) (Table 3). All the AEs were rated as mild. Discussion This randomized, double-blind, placebo-controlled parallel-group trial using rizatriptan confirms the results obtained in our previous open study using sumatriptan showing that the presence of UAs in migraineurs predicts highly positive response rates to triptans [5]. Designed as a conventional acute intervention during a moderate-tosevere migraine attack, the present study shows that rizatriptan is consistently more effective than placebo in achieving pain freedom at 2 h and total migraine freedom at 2 h in patients with UAs. Rizatriptan starts to relieve pain 1 h after dosing, achieves pain freedom, total migraine freedom, no photophobia, and no phonophobia at 1.5 h, and eliminates nausea at 2 h. Equally important clinically,
6 412 J Headache Pain (2012) 13: Table 2 Summary of efficacy data for rizatriptan 10 mg and placebo: primary and secondary endpoints Rizatriptan (n = 41 pts) Placebo (n = 39 pts) Therapeutic gain P value Pain freedom 0.5 h 1/40 (3 % [-2 %; 7 %]) 1/39 (3 % [-2 %;8%]) 0%[-7 %; 7 %] h 5/40 (13 % [2 %; 23 %]) 1/39 (3 % [-2 %; 8 %]) 10 % [-1 %; 21 %] h 16/40 (40 % [25 %; 55 %]) 3/39 (8 % [-1 %; 16 %]) 32 % [15 %; 50 %] <0.001* 2 h 21/39 (54 % [38 %; 70 %]) 3/38 (8 % [-1 %; 17 %]) 46 % [28 %; 64 %] <0.001* Pain relief 0.5 h 1/40 (3 % [-2 %; 7 %]) 1/39 (3 % [-2 %;8%]) 0%[-7 %; 7 %] h 8/40 (20 % [8 %; 32 %]) 1/39 (3 % [-2 %; 8 %]) 17 % [4 %; 31 %] h 20/40 (50 % [35 %; 66 %]) 3/39 (8 % [-1 %; 16 %]) 42 % [25 %; 60 %] <0.001* 2 h 23/39 (59 % [44 %; 74 %]) 4/38 (11 % [1 %; 20 %]) 49 % [30 %; 67 %] <0.001* Total migraine freedom 0.5 h 1/40 (3 % [-2 %; 7 %]) 1/39 (3 % [-2 %;8%]) 0%[-7 %; 7 %] h 3/40 (8 % [-1 %; 16 %]) 1/39 (3 % [-2 %;8%]) 5%[-5 %; 15 %] h 13/40 (33 % [18 %; 47 %]) 3/39 (8 % [-1 %; 16 %]) 25 % [8 %; 42 %] h 20/39 (51 % [36 %; 67 %]) 3/38 (8 % [-1 %; 17 %]) 43 % [26 %; 61 %] <0.001* No nausea 0.5 h 3/22 (14 % [-1 %; 28 %]) 2/23 (9 % [-3 %;20%]) 5%[-14 %; 23 %] h 8/22 (36 % [16 %; 57 %]) 13/23 (13 % [-1 %; 27 %]) 23 % [-1 %; 48 %] h 10/22 (46 % [25 %; 66 %]) 5/23 (22 % [5 %; 39 %]) 24 % [-3 %; 51 %] h 13/21 (62 % [41 %; 83 %]) 3/23 (13 % [-1 %; 27 %]) 49 % [24 %; 74 %] <0.001* No photophobia 0.5 h 3/31 (10 % [-1 %; 20 %]) 1/25 (4 % [-4 %;12%]) 6%[-7 %; 19 %] h 6/31 (19 % [6 %; 33 %]) 1/25 (4 % [-4 %; 12 %]) 15 % [-1 %; 31 %] h 14/22 (45 % [28 %; 63 %]) 1/25 (4 % [-4 %; 12 %]) 41 % [22 %; 60 %] <0.001* 2 h 16/30 (53 % [36 %; 71 %]) 2/24 (8 % [-3 %; 19 %]) 45 % [24 %; 66 %] <0.001* No phonophobia 0.5 h 1/29 (4 % [-3 %; 10 %]) 2/25 (8 % [-3 %; 19 %]) -5 %[-17 %; 8 %] h 6/29 (21 % [6 %; 35 %]) 2/25 (8 % [-3 %; 19 %]) 13 % [-6 %; 31 %] h 13/29 (45 % [27 %; 63 %]) 4/25 (16 % [2 %; 30 %]) 29 % [6 %; 52 %] h 14/28 (50 % [32 %; 69 %]) 4/24 (17 % [2 %; 32 %]) 33 % [10 %; 57 %] SPF 16/40 (40 % [25 %; 55 %]) 1/39 (3 % [-2 %; 8 %]) 37 % [22 %; 53 %] <0.001* 2 24 SPR 18/40 (45 % [30 %; 60 %]) 2/39 (5 % [-2 %; 12 %]) 40 % [23 %; 57 %] <0.001* Data are number of patients (% [95 % confidence interval]). Therapeutic gain = rizatriptan efficacy-placebo efficacy. Statistically significant P values (P \ 0.05) are in bold n, number of treated patients; 2 24 SPF, 2 24 h sustained pain freedom; 2 24 SPR, 2 24 h sustained pain relief * Type I error \5 % after Bonferroni correction for multiple comparison rizatriptan is better than placebo for inducing 2 24 h sustained pain relief and 2 24 h sustained pain freedom. The distinctive finding in our study is the high therapeutic gain for both primary and secondary endpoints. Although our study was not designed to investigate whether migraine patients with UAs respond better to triptans than those without, it offers meaningful evidence-based data on outcomes for comparison. When compared with the therapeutic gain reported in two meta-analyses [10, 11] investigating rizatriptan efficacy in a general migraine population, our study yielded a 15 % absolute increase for 2 h pain freedom (46 vs. 31 %), 15 % for 2 h total migraine freedom (43 vs. 28 %),16 % for 2 h pain relief (49 vs. 33 %), 19 % for 2 24 h sustained pain freedom (37 vs. 18 %), 21 % for 2 24 h sustained pain relief (40 vs. 19 %), 28 % for eliminating nausea (49 vs. 21 %), 17 % for eliminating photophobia (45 vs. 28 %), and 7 % for eliminating phonophobia (33 vs. 26 %). Another interesting finding for clinical and research purposes was that in our patients, all of whom treated their migraine headache only when it became moderate to severe, the therapeutic gain for 2 h pain freedom and 2 24 h sustained pain free (46 and 37 %) almost matched that reported for early treatment (48 and 43 %) [12].
7 J Headache Pain (2012) 13: Table 3 Adverse events in both treatment groups Rizatriptan 10 mg (n = 41 patients) Placebo (n = 39 patients) Any 5 (12 %) 4 (10 %) Nausea 1 (2 %) 2 (5 %) Somnolence 2 (5 %) 0 (0 %) Dizziness 2 (5 %) 0 (0 %) Fatigue 0 (0 %) 1 (2.5 %) Tachycardia 0 (0 %) 1 (2.5 %) The unexpectedly low placebo effect we found in this study is difficult to explain. Although age is a major variable predicting a placebo effect, patients over 50 years of age being less likely to respond to placebo and more likely to respond to rizatriptan [13], in our study age had no predictive effect because our patients mean age was about 40 years. Another possibility is that the low placebo effect and the high rizatriptan response at least partly depended on the fact that many of our patients (61.5 %) were prior triptan users and could therefore discriminate better between placebo and drug (had already experienced triptanrelated adverse events) or be triptan responders. Another unanswered question is why the incidence of rizatriptanrelated AEs was similar in the treated and placebo groups. When we screened for UAs in the migraineurs recruited for the study, some reported UAs spontaneously whereas others reported them only after specific questioning. UAs are more frequent in patients with migraine than might be believed. A population-based study using combined postal mail and telephone interviews showed that 26.9 % of migraine patients report at least one of the UAs during their migraine attack regularly, whereas a survey with face-toface interviews in a headache center discloses UAs in 45.8 % of migraineurs [3, 4]. The presence of UAs depends upon activating the trigeminal autonomic reflex, a physiological response intended to protect ocular and nasal tissue integrity from harmful stimuli. The trigeminal autonomic reflex consists of functional connections between trigeminal afferent fibers and parasympathetic efferents which arise from the superior salivatory nucleus, exit the brainstem via the seventh cranial nerve, traverse the geniculate ganglion and synapse in the sphenopalatine, otic and carotid miniganglia, thereby providing secretomotor innervation to structures such as the lacrimal glands and nasal mucosa [2]. In migraineurs without UAs only the trigeminal afferent arm is active, whereas in patients with UAs the efferent reflex parasympathetic arm is active as well. Migraineurs with UAs experience their headache predominantly on one side, report enhanced pain intensity and have a more frequent facial pain distribution than patients without UAs. Pain severity also correlates weakly with the number of UAs [4, 14]. It is noteworthy that clinical features allow to easily distinguish migraineurs with UAs from patients affected by migraine cluster headache (a debated syndrome characterized by a headache with predominant symptoms of migraine with at least one major timing factor plus three lesser features of cluster headache, or five lesser features of cluster headache ) and also by trigeminal autonomic cephalgias or sinus pathology [15]. A plausible explanation for our patients remarkable response to rizatriptan is that intense trigeminal peripheral afferent activation or sensitization in migraine patients with UAs strongly recruits peripheral neurovascular 5-HT1B/1D receptors, those targeted by triptans. The greater headache severity in these patients presumably reaches the pain threshold above which the autonomic reflex discharges and triggers UAs. This hypothesis receives support also from the clinical finding that patients usually report experiencing UAs when their migraine headache peaks [4]. The precise pathophysiological mechanism underlying trigemino-autonomic reflex activation in migraine is still unclear and could also involve other functional or anatomical peculiarities, or both, in the trigeminal and cranial parasympathetic systems [16]. Our study helps identify a more tailored strategy for treating acute migraine. The search for strategies to improve responsiveness to triptans, given that these antimigraine drugs fail to achieve good results in many individuals when studies consider strong endpoints (e.g., 2 h pain or migraine freedom), identifies as a crucial issue treatment timing. Treating an attack early widens responsiveness to triptans in allodynic patients by preventing central sensitization from developing [17]. Our findings also emphasize the importance of precisely characterizing the migraine phenotype to predict migraine responses. Here we indicate that simple and relatively common clinical features, namely UAs, may have a positive predictive value in rizatriptan therapy and, probably, in triptan therapy. Whether these observations apply also to other acute or preventive migraine medications merits further research. Despite its strong point as a randomized, double-blind, placebo-controlled pharmacological trial in migraine patients with UAs, our study has limitations, for example the lack of direct comparison between migraine patients with and without UAs and the fact that we did not enrol only triptan-naïve patients. In conclusion, migraine patients with UAs, a frequent yet often underdiagnosed category, are a clinically homogeneous migraine population who share a very good response to rizatriptan (and probably to other triptans) even when they use this drug to treat migraine headache that is already moderate or severe. We suggest that pharmacological trials for acute or preventive migraine medications should focus also on this subset of migraine patients.
8 414 J Headache Pain (2012) 13: Acknowledgments Thanks are due to Mrs. Alice Crossman for English revision. This work was supported by a research grant from the Investigator Initiated Studies Program of Merck Sharp & Dohme Corp. Clinical Registration Number: NCT ClinicalTrial.gov. Conflict of interest PB has received grants or research support and honoraria or both for lecturing from, has been a consultant and scientific adviser for Merck, Janssen Pharmaceutical Products, Lusofarmaco and Allergan. Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited. References 1. Goadsby PJ, Charbit AR, Andreou AP, Akerman S, Holland PR (2009) Neurobiology of migraine. Neuroscience 161(2): Goadsby PJ, Edvinsson L, Ekman R (1990) Vasoactive peptide release in the extracerebral circulation of humans during migraine headache. Ann Neurol 28(2): Barbanti P, Fabbrini G, Pesare M, Vanacore N, Cerbo R (2002) Unilateral cranial autonomic symptoms in migraine. Cephalalgia 22(4): Obermann M, Yoon MS, Dommes P, Kuznetsova J, Maschke M, Weimar C et al (2007) Prevalence of trigeminal autonomic symptoms in migraine: a population-based study. Cephalalgia 27(6): Barbanti P, Fabbrini G, Vanacore N, Pesare M, Buzzi MG (2003) Sumatriptan in migraine with unilateral cranial autonomic symptoms: an open study. Headache 43(4): Sarchielli P, Pini LA, Zanchin G, Alberti A, Maggioni F, Rossi C et al (2006) Clinical-biochemical correlates of migraine attacks in rizatriptan responders and non-responders. Cephalalgia 26(3): Ferrari MD, Roon KI, Lipton RB, Goadsby PJ (2001) Oral triptans (serotonin 5-HT(1B/1D) agonists) in acute migraine treatment: a meta-analysis of 53 trials. Lancet 358(9294): Edmeads J (2005) Defining response in migraine: which endpoints are important? Eur Neurol 53(1): Headache Classification Subcommittee of the International Headache Society (2004) The International Classification of Headache Disorders, 2nd edn. Cephalalgia 24(Suppl): Ferrari MD, Loder E, McCarroll KA, Lines CR (2001) Metaanalysis of rizatriptan in randomized controlled clinical trials. Cephalalgia 21(2): Rodgers AJ, Hustad CM, Cady RK, Martin VT, Winner P, Ramsey KE, Ho TW (2011) Total migraine freedom, a potential primary endpoint to assess acute treatment in migraine: comparison to the current FDA requirement using the complete rizatriptan study database. Headache 51(3): Mathew NT, Kailasam J, Meadors L (2004) Early treatment of migraine with rizatriptan: a placebo-controlled study. Headache 44(7): Ho TW, Fan X, Rodgers A, Lines C, Winner P, Shapiro RE (2009) Age effects on placebo response rates in clinical trials of acute agents for migraine: pooled analysis of rizatriptan trials in adults. Cephalalgia 29(7): Yoon M-S, Mueller D, Hansen N, Poitz F, Slomke M, Dommes P et al (2010) Prevalence of facial pain in migraine (8.9 %): a population-based study. Cephalalgia 30(1): Solomon S, Karfunkel P, Guglielmo KM (1985) Migraine cluster headache syndrome. Headache 25(5): Thomsen LL, Olesen J (1995) The autonomic nervous system and the regulation of arterial tone in migraine. Clin Auton Res 5(5): D Amico D, Moschiano F, Bussone G (2006) Early treatment of migraine attacks with triptans: a strategy to enhance outcomes and patient satisfaction? Expert Rev Neurother 6(7):
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Value of postmarketing surveillance studies in achieving a complete picture of antimigraine agents: using almotriptan as an example
 J Headache Pain (2006) 7:27 33 DOI 10.1007/s10194-006-0266-6 ORIGINAL Julio Pascual Hans-Christoph Diener Hélène Massiou Value of postmarketing surveillance studies in achieving a complete picture of antimigraine
J Headache Pain (2006) 7:27 33 DOI 10.1007/s10194-006-0266-6 ORIGINAL Julio Pascual Hans-Christoph Diener Hélène Massiou Value of postmarketing surveillance studies in achieving a complete picture of antimigraine
Despite the widespread use of triptans ... REPORTS... Almotriptan: A Review of Pharmacology, Clinical Efficacy, and Tolerability
 ... REPORTS... Almotriptan: A Review of Pharmacology, Clinical Efficacy, and Tolerability Randal L. Von Seggern, PharmD, BCPS Abstract Objective: This article summarizes preclinical and clinical data for
... REPORTS... Almotriptan: A Review of Pharmacology, Clinical Efficacy, and Tolerability Randal L. Von Seggern, PharmD, BCPS Abstract Objective: This article summarizes preclinical and clinical data for
Setting The setting of the study was primary care. The economic study was conducted in the USA.
 A comparison of the cost-effectiveness of almotriptan and sumatriptan in the treatment of acute migraine using a composite efficacy/tolerability end point Williams P, Reeder C E Record Status This is a
A comparison of the cost-effectiveness of almotriptan and sumatriptan in the treatment of acute migraine using a composite efficacy/tolerability end point Williams P, Reeder C E Record Status This is a
ACUTE TREATMENT FOR MIGRAINE. Cristina Tassorelli
 The European Headache School 2012 ACUTE TREATMENT FOR MIGRAINE Cristina Tassorelli Headache Science Centre, IRCCS Neurological Institute C. Mondino Foundation - Pavia University Centre for Adaptive Disorders
The European Headache School 2012 ACUTE TREATMENT FOR MIGRAINE Cristina Tassorelli Headache Science Centre, IRCCS Neurological Institute C. Mondino Foundation - Pavia University Centre for Adaptive Disorders
A New Era of Migraine Management: The Challenging Landscape in Prevention
 Provided by MediCom Worldwide, Inc. Supported by an educational grant from Teva Pharmaceuticals What is a Neuropeptide? Small chains of amino acids released by neural cells (neurons or glial cells) to
Provided by MediCom Worldwide, Inc. Supported by an educational grant from Teva Pharmaceuticals What is a Neuropeptide? Small chains of amino acids released by neural cells (neurons or glial cells) to
Lasmiditan (200 mg and 100 mg) Compared to Placebo for Acute Treatment of Migraine
 (200 mg and 100 mg) Compared to for Acute Treatment of Migraine Bernice Kuca, M.S. 1 ; Linda A. Wietecha, B.S.N., M.S. 2 ; Paul H. Berg, M.S. 2 ; Sheena K. Aurora, M.D. 2 1 CoLucid Pharmaceuticals, Inc.,
(200 mg and 100 mg) Compared to for Acute Treatment of Migraine Bernice Kuca, M.S. 1 ; Linda A. Wietecha, B.S.N., M.S. 2 ; Paul H. Berg, M.S. 2 ; Sheena K. Aurora, M.D. 2 1 CoLucid Pharmaceuticals, Inc.,
Migraine: Developing Drugs for Acute Treatment Guidance for Industry
 Migraine: Developing Drugs for Acute Treatment Guidance for Industry U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) February 2018
Migraine: Developing Drugs for Acute Treatment Guidance for Industry U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) February 2018
ONZETRA XSAIL (sumatriptan) nasal powder
 ONZETRA XSAIL (sumatriptan) nasal powder Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy
ONZETRA XSAIL (sumatriptan) nasal powder Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Pharmacy
Migraine Migraine Age Specific Prevalence in the United States. Headache International Headache Society Classification
 28 Primary Care Medicine Principles and Practice 29 October 28 Professor Peter J. Goadsby Peter.Goadsby@headache.ucsf.edu Department of Neurology Headache International Headache Society Classification
28 Primary Care Medicine Principles and Practice 29 October 28 Professor Peter J. Goadsby Peter.Goadsby@headache.ucsf.edu Department of Neurology Headache International Headache Society Classification
Disclosures. Triptans for Kids 5/16/13
 5/16/13 Disclosures Triptans for Kids Amy A. Gelfand, MD GelfandA@neuropeds.ucsf.edu Departments of Neurology and Pediatrics UCSF Child Neurology and Headache Center I receive grant funding from: NIH/NINDS
5/16/13 Disclosures Triptans for Kids Amy A. Gelfand, MD GelfandA@neuropeds.ucsf.edu Departments of Neurology and Pediatrics UCSF Child Neurology and Headache Center I receive grant funding from: NIH/NINDS
Clinical case. Clinical case 3/15/2018 OVERVIEW. Refractory headaches and update on novel treatment. Refractory headache.
 OVERVIEW Refractory headaches and update on novel treatment Definition of refractory headache Treatment approach Medications Neuromodulation In the pipeline Juliette Preston, MD OHSU Headache Center Refractory
OVERVIEW Refractory headaches and update on novel treatment Definition of refractory headache Treatment approach Medications Neuromodulation In the pipeline Juliette Preston, MD OHSU Headache Center Refractory
SUMAVEL DOSEPRO (sumatriptan succinate) solution for injection
 SUMAVEL DOSEPRO (sumatriptan succinate) solution for injection Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit
SUMAVEL DOSEPRO (sumatriptan succinate) solution for injection Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit
A new questionnaire for assessment of adverse events associated with triptans: methods of assessment influence the results. Preliminary results
 J Headache Pain (2004) 5:S112 S116 DOI 10.1007/s10194-004-0123-4 Michele Feleppa Fred D. Sheftell Luciana Ciannella Amedeo D Alessio Giancarlo Apice Nino N. Capobianco Donato M.T. Saracino Walter Di Iorio
J Headache Pain (2004) 5:S112 S116 DOI 10.1007/s10194-004-0123-4 Michele Feleppa Fred D. Sheftell Luciana Ciannella Amedeo D Alessio Giancarlo Apice Nino N. Capobianco Donato M.T. Saracino Walter Di Iorio
The use of combination therapies in the acute management of migraine
 REVIEW The use of combination therapies in the acute management of migraine Abouch Valenty Krymchantowski Headache Center of Rio, Rio de Janeiro, Brazil; Outpatient Headache Unit of the Instituto de Neurologia
REVIEW The use of combination therapies in the acute management of migraine Abouch Valenty Krymchantowski Headache Center of Rio, Rio de Janeiro, Brazil; Outpatient Headache Unit of the Instituto de Neurologia
Mark W. Green, MD, FAAN
 Mark W. Green, MD, FAAN Professor of Neurology, Anesthesiology, and Rehabilitation Medicine Director of Headache and Pain Medicine Icahn School of Medicine at Mt Sinai New York Pain-sensitive structures
Mark W. Green, MD, FAAN Professor of Neurology, Anesthesiology, and Rehabilitation Medicine Director of Headache and Pain Medicine Icahn School of Medicine at Mt Sinai New York Pain-sensitive structures
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centers: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Cluster headache (CH): epidemiology, classification and clinical picture
 Cluster headache (CH): epidemiology, classification and clinical picture Toomas Toomsoo, M.D. Head of the Center of Neurology East Tallinn Central Hospital 1 INTRODUCTION Cluster headache - known as trigeminal
Cluster headache (CH): epidemiology, classification and clinical picture Toomas Toomsoo, M.D. Head of the Center of Neurology East Tallinn Central Hospital 1 INTRODUCTION Cluster headache - known as trigeminal
...SELECTED ABSTRACTS...
 The following abstracts, from medical journals containing literature on migraine management, were selected for their relevance to this Special Report supplement. Two Sumatriptan Studies Two double-blind
The following abstracts, from medical journals containing literature on migraine management, were selected for their relevance to this Special Report supplement. Two Sumatriptan Studies Two double-blind
Eletriptan vs sumatriptan: A double-blind, placebo-controlled, multiple migraine attack study
 Eletriptan vs sumatriptan: A double-blind, placebo-controlled, multiple migraine attack study G. Sandrini, M. Färkkilä, G. Burgess, et al. Neurology 2002;59;1210-1217 DOI 10.1212/WNL.59.8.1210 This information
Eletriptan vs sumatriptan: A double-blind, placebo-controlled, multiple migraine attack study G. Sandrini, M. Färkkilä, G. Burgess, et al. Neurology 2002;59;1210-1217 DOI 10.1212/WNL.59.8.1210 This information
Disclosures. Objectives 6/2/2017
 Classification: Migraine and Trigeminal Autonomic Cephalalgias Lauren Doyle Strauss, DO, FAHS Assistant Professor, Child Neurology Assistant Director, Child Neurology Residency @StraussHeadache No disclosures
Classification: Migraine and Trigeminal Autonomic Cephalalgias Lauren Doyle Strauss, DO, FAHS Assistant Professor, Child Neurology Assistant Director, Child Neurology Residency @StraussHeadache No disclosures
Rizatriptan vs. ibuprofen in migraine: a randomised placebo-controlled trial
 J Headache Pain (2007) 8:175 179 DOI 10.1007/s10194-007-0386-7 ORIGINAL Usha Kant Misra Jayantee Kalita Rama Kant Yadav Rizatriptan vs. ibuprofen in migraine: a randomised placebo-controlled trial Received:
J Headache Pain (2007) 8:175 179 DOI 10.1007/s10194-007-0386-7 ORIGINAL Usha Kant Misra Jayantee Kalita Rama Kant Yadav Rizatriptan vs. ibuprofen in migraine: a randomised placebo-controlled trial Received:
Zolmitriptan is effective and well tolerated in Japanese patients with migraine: a dose response study
 is effective and well tolerated in Japanese patients with migraine: a dose response study F Sakai 1, M Iwata 2, K Tashiro 3, Y Itoyama 4, S Tsuji 5, Y Fukuuchi 6, G Sobue 7, K Nakashima 8 & M Morimatsu
is effective and well tolerated in Japanese patients with migraine: a dose response study F Sakai 1, M Iwata 2, K Tashiro 3, Y Itoyama 4, S Tsuji 5, Y Fukuuchi 6, G Sobue 7, K Nakashima 8 & M Morimatsu
ADVANCES IN MIGRAINE MANAGEMENT
 ADVANCES IN MIGRAINE MANAGEMENT Joanna Girard Katzman, M.D.MSPH Assistant Professor, Dept. of Neurology Project ECHO, Chronic Pain Program University of New Mexico Outline Migraine throughout the decades
ADVANCES IN MIGRAINE MANAGEMENT Joanna Girard Katzman, M.D.MSPH Assistant Professor, Dept. of Neurology Project ECHO, Chronic Pain Program University of New Mexico Outline Migraine throughout the decades
Migraine Controversies in Women s Health. Professor Peter J. Goadsby 5 December Department of Neurology
 Migraine 2008 Controversies in Women s Health 5 December 2008 Professor Peter J. Goadsby Peter.Goadsby@headache.ucsf.edu Department of Neurology Headache International Headache Society Classification Primary
Migraine 2008 Controversies in Women s Health 5 December 2008 Professor Peter J. Goadsby Peter.Goadsby@headache.ucsf.edu Department of Neurology Headache International Headache Society Classification Primary
Tears of Pain SUNCT and SUNA A/PROFESSOR ARUN AGGARWAL RPAH PAIN MANAGEMENT CENTRE
 Tears of Pain SUNCT and SUNA A/PROFESSOR ARUN AGGARWAL RPAH PAIN MANAGEMENT CENTRE IHS Classification 1989 (updated 2004) Primary Headaches 4 categories Migraine Tension-type Cluster and other trigeminal
Tears of Pain SUNCT and SUNA A/PROFESSOR ARUN AGGARWAL RPAH PAIN MANAGEMENT CENTRE IHS Classification 1989 (updated 2004) Primary Headaches 4 categories Migraine Tension-type Cluster and other trigeminal
Guidance for Industry Migraine: Developing Drugs for Acute Treatment
 Guidance for Industry Migraine: Developing Drugs for Acute Treatment DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this draft
Guidance for Industry Migraine: Developing Drugs for Acute Treatment DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this draft
MEASURE #1: MEDICATION PRESCRIBED FOR ACUTE MIGRAINE ATTACK Headache
 MEASURE #1: MEDICATION PRESCRIBED FOR ACUTE MIGRAINE ATTACK Headache Measure Description Percentage of patients age 12 years and older with a diagnosis of migraine who were prescribed a guideline recommended
MEASURE #1: MEDICATION PRESCRIBED FOR ACUTE MIGRAINE ATTACK Headache Measure Description Percentage of patients age 12 years and older with a diagnosis of migraine who were prescribed a guideline recommended
The main features of the TACs:
 Pathophysiology of trigeminal autonomic cephalalgias Jasna Zidverc Trajković Headache center The main features of the TACs: 1. Trigeminal distribution of the pain 2. Ipsilateral cranial autonomic features
Pathophysiology of trigeminal autonomic cephalalgias Jasna Zidverc Trajković Headache center The main features of the TACs: 1. Trigeminal distribution of the pain 2. Ipsilateral cranial autonomic features
The Clinical Profile of Sumatriptan: Cluster Headache Key Words
 Paper Eur Neurol 1994;34(suppl 2):35-39 P.J. Peter J. Goadsby Department of Neurology, The Prince Henry Hospital, Little Bay, Sydney, NSW, Australia The Clinical Profile of Sumatriptan: Cluster Headache
Paper Eur Neurol 1994;34(suppl 2):35-39 P.J. Peter J. Goadsby Department of Neurology, The Prince Henry Hospital, Little Bay, Sydney, NSW, Australia The Clinical Profile of Sumatriptan: Cluster Headache
Zolmitriptan nasal spray provides fast relief of migraine symptoms and is preferred by patients: a Swedish study of preference in clinical practice
 J Headache Pain (2004) 5:237 242 DOI 10.1007/s10194-004-0132-3 ORIGINAL Carl G.H. Dahlöf Mattias Linde Erika Kerekes Zolmitriptan nasal spray provides fast relief of migraine symptoms and is preferred
J Headache Pain (2004) 5:237 242 DOI 10.1007/s10194-004-0132-3 ORIGINAL Carl G.H. Dahlöf Mattias Linde Erika Kerekes Zolmitriptan nasal spray provides fast relief of migraine symptoms and is preferred
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
HEADACHE PATHOPHYSIOLOGY
 HEADACHE PATHOPHYSIOLOGY Andrew Charles, M.D. Professor Director, UCLA Goldberg Migraine Program Meyer and Renee Luskin Chair in Migraine and Headache Studies Director, Headache Research and Treatment
HEADACHE PATHOPHYSIOLOGY Andrew Charles, M.D. Professor Director, UCLA Goldberg Migraine Program Meyer and Renee Luskin Chair in Migraine and Headache Studies Director, Headache Research and Treatment
Cluster headache associated with acute maxillary sinusitis.
 Cluster headache associated with acute maxillary sinusitis. Edvardsson, Bengt Published in: SpringerPlus DOI: 10.1186/2193-1801-2-509 2013 Link to publication Citation for published version (APA): Edvardsson,
Cluster headache associated with acute maxillary sinusitis. Edvardsson, Bengt Published in: SpringerPlus DOI: 10.1186/2193-1801-2-509 2013 Link to publication Citation for published version (APA): Edvardsson,
Chronic Migraine in Primary Care. December 11 th, 2017 Werner J. Becker University of Calgary
 Chronic Migraine in Primary Care December 11 th, 2017 Werner J. Becker University of Calgary Disclosures Faculty: Werner J. Becker Relationships with commercial interests: Grants/Research Support: Clinical
Chronic Migraine in Primary Care December 11 th, 2017 Werner J. Becker University of Calgary Disclosures Faculty: Werner J. Becker Relationships with commercial interests: Grants/Research Support: Clinical
FREQUENCY OF CRANIAL AUTONOMIC SYMPTOMS (CAS) IN MIGRAINE PATIENTS
 O R I G I N A L A R T I C L E FREQUENCY OF CRANIAL AUTONOMIC SYMPTOMS (CAS) IN MIGRAINE PATIENTS Sadaf Nasir 1, Naureen Abrar 2, Khalid Sher 3, Shahnaz 4 1.Dr. Sadaf Nasir. Senior Registrar, Department
O R I G I N A L A R T I C L E FREQUENCY OF CRANIAL AUTONOMIC SYMPTOMS (CAS) IN MIGRAINE PATIENTS Sadaf Nasir 1, Naureen Abrar 2, Khalid Sher 3, Shahnaz 4 1.Dr. Sadaf Nasir. Senior Registrar, Department
Migraine and hormonal contraceptives
 Migraine and hormonal contraceptives Department of Community Medicine, Systems Epidemiology University of Tromsø, November 2017 Nora Stensland Bugge Medical research student Presentation outline What is
Migraine and hormonal contraceptives Department of Community Medicine, Systems Epidemiology University of Tromsø, November 2017 Nora Stensland Bugge Medical research student Presentation outline What is
Triptans: Nonresponse, Recurrence, and Serious AEs for Many Patients
 Efficacy, Safety, and Tolerability of Rimegepant 75 mg, an Oral CGRP Receptor Antagonist, for the Acute Treatment of Migraine: Results from a Phase 3, Double-Blind, Randomized, Placebo-Controlled Trial,
Efficacy, Safety, and Tolerability of Rimegepant 75 mg, an Oral CGRP Receptor Antagonist, for the Acute Treatment of Migraine: Results from a Phase 3, Double-Blind, Randomized, Placebo-Controlled Trial,
MIGRAINE A MYSTERY HEADACHE
 MIGRAINE A MYSTERY HEADACHE The migraine is a chronic neurological disease that is characterized by moderate to severe episodes of headache that is mostly associated with other central nervous system (CNS)
MIGRAINE A MYSTERY HEADACHE The migraine is a chronic neurological disease that is characterized by moderate to severe episodes of headache that is mostly associated with other central nervous system (CNS)
Sumatriptan Tablets, Nasal Spray (Imitrex), Nasal Powder (Onzetra Xsail), sumatriptan and naproxen sodium (Treximet tablets)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 0 Subject: Sumatriptan Page: 1 of 6 Last Review Date: November 30, 2018 Sumatriptan Description Sumatriptan
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 0 Subject: Sumatriptan Page: 1 of 6 Last Review Date: November 30, 2018 Sumatriptan Description Sumatriptan
Migraine strikes study: factors in patients decision to treat early
 J Headache Pain (2009) 10:93 99 DOI 10.1007/s10194-008-0091-1 ORIGINAL Migraine strikes study: factors in patients decision to treat early Wendy Golden Æ Judith K. Evans Æ Henry Hu Received: 17 October
J Headache Pain (2009) 10:93 99 DOI 10.1007/s10194-008-0091-1 ORIGINAL Migraine strikes study: factors in patients decision to treat early Wendy Golden Æ Judith K. Evans Æ Henry Hu Received: 17 October
Early treatment of a migraine attack while pain is still mild increases the efficacy of sumatriptan
 Blackwell Science, LtdOxford, UKCHACephalalgia1468-2982Blackwell Science, 20042411925933Original ArticleEarly treatment of migraine with sumatriptanj Scholpp et al. Early treatment of a migraine attack
Blackwell Science, LtdOxford, UKCHACephalalgia1468-2982Blackwell Science, 20042411925933Original ArticleEarly treatment of migraine with sumatriptanj Scholpp et al. Early treatment of a migraine attack
Embriologically, The head is formed from the first two cervical segments (except the mandible, which is formed by the third). The first and second
 Embriologically, The head is formed from the first two cervical segments (except the mandible, which is formed by the third). The first and second cervical vertebra are also derived from these two segments.
Embriologically, The head is formed from the first two cervical segments (except the mandible, which is formed by the third). The first and second cervical vertebra are also derived from these two segments.
Sumatriptan Tablets, Nasal Spray (Imitrex), Nasal Powder (Onzetra Xsail), sumatriptan and naproxen sodium (Treximet tablets)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 05.70.10 Subject: Sumatriptan Page: 1 of 6 Last Review Date: March 16, 2018 Sumatriptan Description Sumatriptan
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 05.70.10 Subject: Sumatriptan Page: 1 of 6 Last Review Date: March 16, 2018 Sumatriptan Description Sumatriptan
MIGRAINE A CAUSE OF INTENSE THROBBING; A MINI REVIEW
 IJBPAS, January, 2016, 5(1): 87-92 ISSN: 2277 4998 MIGRAINE A CAUSE OF INTENSE THROBBING; A MINI REVIEW MUHAMMAD ZAMAN 1*, RABIA HASSAN 1, MUHAMMAD SHAFEEQ UR RAHMAN 2, MUHAMMAD HAFIZ ARSHAD 3, SYED ATIF
IJBPAS, January, 2016, 5(1): 87-92 ISSN: 2277 4998 MIGRAINE A CAUSE OF INTENSE THROBBING; A MINI REVIEW MUHAMMAD ZAMAN 1*, RABIA HASSAN 1, MUHAMMAD SHAFEEQ UR RAHMAN 2, MUHAMMAD HAFIZ ARSHAD 3, SYED ATIF
Update on Diagnosis and Management of Migraines
 Update on Diagnosis and Management of Migraines Joel J. Heidelbaugh, MD, FAAFP, FACG Clinical Professor Departments of Family Medicine and Urology University of Michigan Learning Objectives To distinguish
Update on Diagnosis and Management of Migraines Joel J. Heidelbaugh, MD, FAAFP, FACG Clinical Professor Departments of Family Medicine and Urology University of Michigan Learning Objectives To distinguish
Drug Class Review on Triptans
 Drug Class Review on UPDATED FINAL REPORT #1 December 2003 Mark Helfand, MD, MPH Kim Peterson, MS Oregon Evidence-based Practice Center Oregon Health & Science University Table of Contents Introduction
Drug Class Review on UPDATED FINAL REPORT #1 December 2003 Mark Helfand, MD, MPH Kim Peterson, MS Oregon Evidence-based Practice Center Oregon Health & Science University Table of Contents Introduction
Headache Assessment In Primary Eye Care
 Headache Assessment In Primary Eye Care Spencer Johnson, O.D., F.A.A.O. Northeastern State University Oklahoma College of Optometry johns137@nsuok.edu Course Objectives Review headache classification Understand
Headache Assessment In Primary Eye Care Spencer Johnson, O.D., F.A.A.O. Northeastern State University Oklahoma College of Optometry johns137@nsuok.edu Course Objectives Review headache classification Understand
CGRP, MONOCLONAL ANTIBODIES AND SMALL MOLECULES (-GEPANTS)
 CGRP, MONOCLONAL ANTIBODIES AND SMALL MOLECULES (-GEPANTS) Hans-Christoph Diener Senior Professor of Clinical Neurosciences University Duisburg-Essen Germany CGRP, Monoclonal Antibodies and Small Molecules
CGRP, MONOCLONAL ANTIBODIES AND SMALL MOLECULES (-GEPANTS) Hans-Christoph Diener Senior Professor of Clinical Neurosciences University Duisburg-Essen Germany CGRP, Monoclonal Antibodies and Small Molecules
The Role of Allergies and Sinus Disorders in Headache & Facial Pain
 The Role of Allergies and Sinus Disorders in Headache & Facial Pain Vincent T. Martin, MD Professor of Medicine Co-director of the Headache & Facial Pain Center University of Cincinnati College of Medicine
The Role of Allergies and Sinus Disorders in Headache & Facial Pain Vincent T. Martin, MD Professor of Medicine Co-director of the Headache & Facial Pain Center University of Cincinnati College of Medicine
Study No.:MPX Title: Rationale: Phase: IIB Study Period: Study Design: Centres: Indication: Treatment: Objectives:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Zomig. Zomig / Zomig-ZMT (zolmitriptan) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.22 Subject: Zomig Page: 1 of 5 Last Review Date: November 30, 2018 Zomig Description Zomig / Zomig-ZMT
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.22 Subject: Zomig Page: 1 of 5 Last Review Date: November 30, 2018 Zomig Description Zomig / Zomig-ZMT
Zomig. Zomig / Zomig-ZMT (zolmitriptan) Description
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.22 Subject: Zomig Page: 1 of 5 Last Review Date: March 16, 2018 Zomig Description Zomig / Zomig-ZMT
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.22 Subject: Zomig Page: 1 of 5 Last Review Date: March 16, 2018 Zomig Description Zomig / Zomig-ZMT
Drug Class Review on the Triptans
 Drug Class Review on the Final Report March 7, 2003 Expires October 31, 2003 Mark Helfand, MD, MPH Kim Peterson, MS Produced by Oregon Health & Science University 3181 SW Sam Jackson Park Road Mailcode:
Drug Class Review on the Final Report March 7, 2003 Expires October 31, 2003 Mark Helfand, MD, MPH Kim Peterson, MS Produced by Oregon Health & Science University 3181 SW Sam Jackson Park Road Mailcode:
Cluster headache in women: clinical characteristics and comparison with cluster headache in men
 J Neurol Neurosurg Psychiatry 2001;70:613 617 613 Department of Neurology, JeVerson Headache Center, Thomas University Hospital, 111 South 11th Street, Gibbon Building, Suite 8130, Philadelphia, Pennsylvania
J Neurol Neurosurg Psychiatry 2001;70:613 617 613 Department of Neurology, JeVerson Headache Center, Thomas University Hospital, 111 South 11th Street, Gibbon Building, Suite 8130, Philadelphia, Pennsylvania
Supraorbital nerve stimulation Cefaly Device - FDA Approved for migraine prevention (also being investigated as acute therapy)
 NEUROSTIMULATION/NEUROMODULATION UPDATE Meyer and Renee Luskin Andrew Charles, M.D. Professor Luskin Chair in Migraine and Headache Studies Director, UCLA Goldberg Migraine Program David Geffen School
NEUROSTIMULATION/NEUROMODULATION UPDATE Meyer and Renee Luskin Andrew Charles, M.D. Professor Luskin Chair in Migraine and Headache Studies Director, UCLA Goldberg Migraine Program David Geffen School
Headache. Section 1. Migraine headache. Clinical presentation
 Section 1 Headache Migraine headache 1 Clinical presentation It is important to recognize just how significant a problem migraine headache is. It has been estimated that migraine affects 11% of the United
Section 1 Headache Migraine headache 1 Clinical presentation It is important to recognize just how significant a problem migraine headache is. It has been estimated that migraine affects 11% of the United
Is the Migraid device an asset in the non-pharmacologic treatment of migraine?
 Acta neurol. belg., 2007, 107, 40-46 Is the Migraid device an asset in the non-pharmacologic treatment of migraine? Herman PIETERSE 1,3, Joop A. M. KUSTER 2 and Luc M. VAN BORTEL 1 1 Heymans Institute
Acta neurol. belg., 2007, 107, 40-46 Is the Migraid device an asset in the non-pharmacologic treatment of migraine? Herman PIETERSE 1,3, Joop A. M. KUSTER 2 and Luc M. VAN BORTEL 1 1 Heymans Institute
Drug Class Review on Triptans
 Drug Class Review on Triptans Final Report Update 3 November 2005 Original Report Date: March 2003 Update 1 Report Date: December 2003 Update 2 Report Date: September 2004 A literature scan of this topic
Drug Class Review on Triptans Final Report Update 3 November 2005 Original Report Date: March 2003 Update 1 Report Date: December 2003 Update 2 Report Date: September 2004 A literature scan of this topic
medications. This was an openlabel study consisting of patients with migraines who historically failed to respond to oral triptan
 J Headache Pain (2007) 8:13 18 DOI 10.1007/s10194-007-0354-7 ORIGINAL Seymour Diamond Fred G. Freitag Alexander Feoktistov George Nissan Sumatriptan 6 mg subcutaneous as an effective migraine treatment
J Headache Pain (2007) 8:13 18 DOI 10.1007/s10194-007-0354-7 ORIGINAL Seymour Diamond Fred G. Freitag Alexander Feoktistov George Nissan Sumatriptan 6 mg subcutaneous as an effective migraine treatment
Adult & Pediatric Patients. Stanford Health Care, Division Pain Medicine
 Acute Treatment Strategies in Adult & Pediatric Patients Theresa Mallick Searle, MS, RN BC, ANP BC Disclosures Speakers Bureau: Allergan, Depomed Acute Treatment Strategies in Adult & Pediatric Patients
Acute Treatment Strategies in Adult & Pediatric Patients Theresa Mallick Searle, MS, RN BC, ANP BC Disclosures Speakers Bureau: Allergan, Depomed Acute Treatment Strategies in Adult & Pediatric Patients
Treatment satisfaction with zolmitriptan nasal spray for migraine in a real life setting: results from phase two of the REALIZE study
 J Headache Pain (2005) 6:405 411 DOI 10.1007/s10194-005-0237-3 ORIGINAL Marek Gawel Jürgen Aschoff Arne May Bruce R. Charlesworth on behalf of the REALIZE Study Group Treatment satisfaction with zolmitriptan
J Headache Pain (2005) 6:405 411 DOI 10.1007/s10194-005-0237-3 ORIGINAL Marek Gawel Jürgen Aschoff Arne May Bruce R. Charlesworth on behalf of the REALIZE Study Group Treatment satisfaction with zolmitriptan
Atenolol in the prophylaxis of chronic migraine: a 3-month open-label study
 Edvardsson SpringerPlus 2013, 2:479 a SpringerOpen Journal RESEARCH Open Access Atenolol in the prophylaxis of chronic migraine: a 3-month open-label study Bengt Edvardsson Abstract Background: Chronic
Edvardsson SpringerPlus 2013, 2:479 a SpringerOpen Journal RESEARCH Open Access Atenolol in the prophylaxis of chronic migraine: a 3-month open-label study Bengt Edvardsson Abstract Background: Chronic
Greg Book. September 25, 2007 Olin Neuropsychiatry Research Center
 Migraine Pathogenesis Greg Book September 25, 2007 Olin Neuropsychiatry Research Center Migraine Migraine i affects approximately 19.2% of females and 6.6% of males in the United States. (Patel, Bigal
Migraine Pathogenesis Greg Book September 25, 2007 Olin Neuropsychiatry Research Center Migraine Migraine i affects approximately 19.2% of females and 6.6% of males in the United States. (Patel, Bigal
What is the Effectiveness of OnabotulinumtoxinA (Botox ) in Reducing the Number of Chronic Migraines (CM) in Patients Years Old?
 Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2013 What is the Effectiveness of OnabotulinumtoxinA
Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2013 What is the Effectiveness of OnabotulinumtoxinA
Specific Objectives A. Topics to be lectured and discussed at the plenary sessions
 Specific Objectives A. Topics to be lectured and discussed at the plenary sessions 0. Introduction: Good morning ICHD-III! Let s start at the very beginning. When you read you begin with A-B-C, so when
Specific Objectives A. Topics to be lectured and discussed at the plenary sessions 0. Introduction: Good morning ICHD-III! Let s start at the very beginning. When you read you begin with A-B-C, so when
Headache Master School Japan-Osaka 2016 (HMSJ-Osaka2016) October 23, II. Management of Refractory Headaches
 Headache Master School Japan-Osaka 2016 (HMSJ-Osaka2016) October 23, 2016 II. Management of Refractory Headaches Case presentation 1: A case of intractable daily-persistent headache Keio University School
Headache Master School Japan-Osaka 2016 (HMSJ-Osaka2016) October 23, 2016 II. Management of Refractory Headaches Case presentation 1: A case of intractable daily-persistent headache Keio University School
Migraine Pathophysiology. Robert E. Shapiro, MD, PhD
 Migraine Pathophysiology Robert E. Shapiro, MD, PhD Disclosures Eli Lilly Member, Clinical Trials Data Monitoring Committee Learning Objectives By the end of this course participants will be able to describe:
Migraine Pathophysiology Robert E. Shapiro, MD, PhD Disclosures Eli Lilly Member, Clinical Trials Data Monitoring Committee Learning Objectives By the end of this course participants will be able to describe:
Trigeminal Autonomic Cephalalgias. Disclosures. Objectives 6/20/2018. Rashmi Halker Singh, MD FAHS UCNS Review Course June 2018
 Trigeminal Autonomic Cephalalgias Rashmi Halker Singh, MD FAHS UCNS Review Course June 2018 Disclosures Honoraria from Allergan and Amgen for advisory board, Current Neurology and Neuroscience Reports
Trigeminal Autonomic Cephalalgias Rashmi Halker Singh, MD FAHS UCNS Review Course June 2018 Disclosures Honoraria from Allergan and Amgen for advisory board, Current Neurology and Neuroscience Reports
Prevention and Treatment of Migraines CAITLIN BARNES, PHARM.D. CANDIDATE AMBULATORY CARE JOE CAMMILLERI, PHARM.D. NATOHYA MALLORY, PHARM.D.
 Prevention and Treatment of Migraines CAITLIN BARNES, PHARM.D. CANDIDATE AMBULATORY CARE JOE CAMMILLERI, PHARM.D. NATOHYA MALLORY, PHARM.D. Objectives Present patient case Review epidemiology/pathophysiology
Prevention and Treatment of Migraines CAITLIN BARNES, PHARM.D. CANDIDATE AMBULATORY CARE JOE CAMMILLERI, PHARM.D. NATOHYA MALLORY, PHARM.D. Objectives Present patient case Review epidemiology/pathophysiology
New Zealand Data Sheet
 Arrow - Sumatriptan Sumatriptan Injection 6mg/0.5mL Presentation New Zealand Data Sheet ARROW SUMATRIPTAN Injection 6mg/0.5mL is a clear, colourless to pale yellow solution in a prefilled syringe. Uses
Arrow - Sumatriptan Sumatriptan Injection 6mg/0.5mL Presentation New Zealand Data Sheet ARROW SUMATRIPTAN Injection 6mg/0.5mL is a clear, colourless to pale yellow solution in a prefilled syringe. Uses
An economic evaluation of rizatriptan in the treatment of migraine Thompson M, Gawel M, Desjardins B, Ferko N, Grima D
 An economic evaluation of rizatriptan in the treatment of migraine Thompson M, Gawel M, Desjardins B, Ferko N, Grima D Record Status This is a critical abstract of an economic evaluation that meets the
An economic evaluation of rizatriptan in the treatment of migraine Thompson M, Gawel M, Desjardins B, Ferko N, Grima D Record Status This is a critical abstract of an economic evaluation that meets the
Cost-effectiveness of almotriptan and rizatriptan in the treatment of acute migraine Williams P, Reeder C E
 Cost-effectiveness of almotriptan and rizatriptan in the treatment of acute migraine Williams P, Reeder C E Record Status This is a critical abstract of an economic evaluation that meets the criteria for
Cost-effectiveness of almotriptan and rizatriptan in the treatment of acute migraine Williams P, Reeder C E Record Status This is a critical abstract of an economic evaluation that meets the criteria for
Application of ICHD 2nd edition criteria for primary headaches with the aid of a computerised, structured medical record for the specialist
 J Headache Pain (2005) 6:205 210 DOI 10.1007/s10194-005-0186-x ICHD-II: EVALUATION AND PROPOSALS Paola Sarchielli Mauro Pedini Andrea Alberti Cristiana Rossi Antonio Baldi Ilenia Corbelli Paolo Calabresi
J Headache Pain (2005) 6:205 210 DOI 10.1007/s10194-005-0186-x ICHD-II: EVALUATION AND PROPOSALS Paola Sarchielli Mauro Pedini Andrea Alberti Cristiana Rossi Antonio Baldi Ilenia Corbelli Paolo Calabresi
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 CGRP Page 1 of 8 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: CGRP (calcitonin gene-related peptide) Prime Therapeutics will review Prior Authorization requests
CGRP Page 1 of 8 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: CGRP (calcitonin gene-related peptide) Prime Therapeutics will review Prior Authorization requests
By Nathan Hall Associate Editor
 By Nathan Hall Associate Editor 34 Practical Neurology March 2005 These new rules may change the definition of head pain, but some practitioners may find the new guidelines themselves to be a source of
By Nathan Hall Associate Editor 34 Practical Neurology March 2005 These new rules may change the definition of head pain, but some practitioners may find the new guidelines themselves to be a source of
Long-Term Care Updates
 Long-Term Care Updates September 2017 By Lindsay Slowiczek, PharmD Migraines are often considered to be a condition affecting younger or middle-aged patients, during which patients experience episodic,
Long-Term Care Updates September 2017 By Lindsay Slowiczek, PharmD Migraines are often considered to be a condition affecting younger or middle-aged patients, during which patients experience episodic,
Migraine is a primary headache disorder ... REPORTS... Migraine: Diagnosis, Management, and New Treatment Options
 ... REPORTS... Migraine: Diagnosis, Management, and New Treatment Options R. Michael Gallagher, DO; and F. Michael Cutrer, MD Abstract Objective: The safety and tolerability of medications used to treat
... REPORTS... Migraine: Diagnosis, Management, and New Treatment Options R. Michael Gallagher, DO; and F. Michael Cutrer, MD Abstract Objective: The safety and tolerability of medications used to treat
MEASURE #4: Overuse of Barbiturate Containing Medications for Primary Headache Disorders Headache
 MEASURE #4: Overuse of Barbiturate Containing Medications for Primary Headache Disorders Headache Measure Description Percentage of patients age 18 years old and older with a diagnosis of primary headache
MEASURE #4: Overuse of Barbiturate Containing Medications for Primary Headache Disorders Headache Measure Description Percentage of patients age 18 years old and older with a diagnosis of primary headache
They are updated regularly as new NICE guidance is published. To view the latest version of this NICE Pathway see:
 bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
bring together everything NICE says on a topic in an interactive flowchart. are interactive and designed to be used online. They are updated regularly as new NICE guidance is published. To view the latest
Sumatriptan (50 and 100 mg) in repeated migraine attacks: a patient preference study
 J Headache Pain () 1:33 38 Springer-Verlag ORIGINAL ARTICLE Giorgio Zanchin Maurizio Roncolato Laura Fabbri Sumatriptan (5 and mg) in repeated migraine attacks: a patient preference study Received: 29
J Headache Pain () 1:33 38 Springer-Verlag ORIGINAL ARTICLE Giorgio Zanchin Maurizio Roncolato Laura Fabbri Sumatriptan (5 and mg) in repeated migraine attacks: a patient preference study Received: 29
MEDICAL POLICY I. POLICY II. PRODUCT VARIATIONS TOP III. DESCRIPTION/BACKGROUND POLICY TITLE SPHENOPALATINE GANGLION BLOCK POLICY NUMBER MP-4.
 Original Issue Date (Created): 2/1/2018 Most Recent Review Date (Revised): 7/5/2018 Effective Date: 9/1/2018 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT VARIATIONS DISCLAIMER
Original Issue Date (Created): 2/1/2018 Most Recent Review Date (Revised): 7/5/2018 Effective Date: 9/1/2018 POLICY PRODUCT VARIATIONS DESCRIPTION/BACKGROUND RATIONALE DEFINITIONS BENEFIT VARIATIONS DISCLAIMER
Faculty Disclosures. Learning Objectives. Acute Treatment Strategies
 WWW.AMERICANHEADACHESOCIETY.ORG Acute Treatment Strategies Content developed by: Lawrence C. Newman, MD, FAHS Donna Gutterman, PharmD Faculty Disclosures LAWRENCE C. NEWMAN, MD, FAHS Dr. Newman has received
WWW.AMERICANHEADACHESOCIETY.ORG Acute Treatment Strategies Content developed by: Lawrence C. Newman, MD, FAHS Donna Gutterman, PharmD Faculty Disclosures LAWRENCE C. NEWMAN, MD, FAHS Dr. Newman has received
2.0 Synopsis. ABT-711 M Clinical Study Report R&D/06/573. (For National Authority Use Only) to Part of Dossier: Volume:
 2.0 Synopsis Abbott Laboratories Name of Study Drug: Depakote ER Name of Active Ingredient: Divalproex sodium (ABT-711) Individual Study Table Referring to Part of Dossier: Volume: Page: (For National
2.0 Synopsis Abbott Laboratories Name of Study Drug: Depakote ER Name of Active Ingredient: Divalproex sodium (ABT-711) Individual Study Table Referring to Part of Dossier: Volume: Page: (For National
Sumatriptan Injection (Imitrex / Alsuma / Sumavel / Zembrace)
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.11 Subject: Sumatriptan Injection Page: 1 of 6 Last Review Date: March 16, 2018 Sumatriptan Injection
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.11 Subject: Sumatriptan Injection Page: 1 of 6 Last Review Date: March 16, 2018 Sumatriptan Injection
Anti-Migraine Agents
 DRUG POLICY BENEFIT APPLICATION Anti-Migraine Agents Benefit determinations are based on the applicable contract language in effect at the time the services were rendered. Exclusions, limitations or exceptions
DRUG POLICY BENEFIT APPLICATION Anti-Migraine Agents Benefit determinations are based on the applicable contract language in effect at the time the services were rendered. Exclusions, limitations or exceptions
Calcitonin Gene Related Peptide Receptor Antagonist BIBN 4096 BS for the Acute Treatment of Migraine
 The new england journal of medicine original article Calcitonin Gene Related Peptide Receptor Antagonist BIBN 4096 BS for the Acute Treatment of Migraine Jes Olesen, M.D., Hans-Christoph Diener, M.D.,
The new england journal of medicine original article Calcitonin Gene Related Peptide Receptor Antagonist BIBN 4096 BS for the Acute Treatment of Migraine Jes Olesen, M.D., Hans-Christoph Diener, M.D.,
Preventive Effect of Greater Occipital Nerve Block on Severity and Frequency of Migraine Headache
 Global Journal of Health Science; Vol. 6, No. 6; 2014 ISSN 1916-9736 E-ISSN 1916-9744 Published by Canadian Center of Science and Education Preventive Effect of Greater Occipital Nerve Block on Severity
Global Journal of Health Science; Vol. 6, No. 6; 2014 ISSN 1916-9736 E-ISSN 1916-9744 Published by Canadian Center of Science and Education Preventive Effect of Greater Occipital Nerve Block on Severity
Sphenopalatine Ganglion Block or Other Intranasal Blockage for Headache
 Sphenopalatine Ganglion Block or Other Intranasal Blockage for Headache Policy Number: 7.01.159 Last Review: 7/1/18 Origination: 7/2017 Next Review: 1/1/19 Policy Blue Cross and Blue Shield of Kansas City
Sphenopalatine Ganglion Block or Other Intranasal Blockage for Headache Policy Number: 7.01.159 Last Review: 7/1/18 Origination: 7/2017 Next Review: 1/1/19 Policy Blue Cross and Blue Shield of Kansas City
Maxalt. Maxalt / Maxalt-MLT (rizatriptan) Description. Section: Prescription Drugs Effective Date: April 1, 2016
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Maxalt Page: 1 of 5 Last Review Date: March 18, 2016 Maxalt Description Maxalt / Maxalt-MLT (rizatriptan)
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 Subject: Maxalt Page: 1 of 5 Last Review Date: March 18, 2016 Maxalt Description Maxalt / Maxalt-MLT (rizatriptan)
Specific features of migraine syndrome in children
 J Headache Pain (2006) 7:206 210 DOI 10.1007/s10194-006-0312-4 RAPID COMMUNICATION Marija Knezevic-Pogancev Specific features of migraine syndrome in children Received: 16 April 2006 Accepted in revised
J Headache Pain (2006) 7:206 210 DOI 10.1007/s10194-006-0312-4 RAPID COMMUNICATION Marija Knezevic-Pogancev Specific features of migraine syndrome in children Received: 16 April 2006 Accepted in revised
Clinical Commissioning Policy Statement: Sphenopalatine Ganglion Stimulation for Refractory Chronic Cluster Headache (Adults)
 Clinical Commissioning Policy Statement: Sphenopalatine Ganglion Stimulation for Refractory Chronic Cluster Headache (Adults) NHS England Reference: 170083P 1 Contents 1. Plain Language Summary... 3 2.
Clinical Commissioning Policy Statement: Sphenopalatine Ganglion Stimulation for Refractory Chronic Cluster Headache (Adults) NHS England Reference: 170083P 1 Contents 1. Plain Language Summary... 3 2.
Outpatient Headache Care Guideline
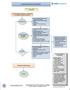 1 Outpatient Care Guideline Inclusion criteria: children > 3 yrs with headaches Is urgent emergency department, neuroimaging, or Neurology consultation indicated? Referral to ED if: New severe headache
1 Outpatient Care Guideline Inclusion criteria: children > 3 yrs with headaches Is urgent emergency department, neuroimaging, or Neurology consultation indicated? Referral to ED if: New severe headache
Advances in the Treatment of Migraine
 Advances in the Treatment of Migraine C. Philip O Carroll, M.D. Director Neurobehavioral Medicine Hoag Neurosciences Institute Guyuron B Headache, 2015;55:1464-1473 I m sorry your head hurts, sweetie.is
Advances in the Treatment of Migraine C. Philip O Carroll, M.D. Director Neurobehavioral Medicine Hoag Neurosciences Institute Guyuron B Headache, 2015;55:1464-1473 I m sorry your head hurts, sweetie.is
Cluster headache and paroxysmal hemicrania: differential diagnosis
 Blackwell Science, LtdOxford, UKCHACephalalgia0333-1024Blackwell Science, 2004254244248Original ArticleCH and PH: differential diagnosisj Zidverc-Trajkovic et al. Cluster headache and paroxysmal hemicrania:
Blackwell Science, LtdOxford, UKCHACephalalgia0333-1024Blackwell Science, 2004254244248Original ArticleCH and PH: differential diagnosisj Zidverc-Trajkovic et al. Cluster headache and paroxysmal hemicrania:
Disease Description and Statistics Current State: Medicare Denial of Coverage Expert Opinion Clinical Studies Gap in Medical Care Cost to US
 Oxygen Therapy Disease Description and Statistics Current State: Medicare Denial of Coverage Expert Opinion Clinical Studies Gap in Medical Care Cost to US Taxpayers Recommendations Cluster headaches are
Oxygen Therapy Disease Description and Statistics Current State: Medicare Denial of Coverage Expert Opinion Clinical Studies Gap in Medical Care Cost to US Taxpayers Recommendations Cluster headaches are
Core Safety Profile. Pharmaceutical form(s)/strength: Tablets and Oral lyophilisates (wafers) 5 and 10 mg NL/H/PSUR/0002/001 Date of FAR:
 Core Safety Profile Active substance: Rizatriptan Pharmaceutical form(s)/strength: Tablets and Oral lyophilisates (wafers) 5 and 10 mg P RMS: NL/H/PSUR/0002/001 Date of FAR: 23.05.2011 4.3 Contraindications
Core Safety Profile Active substance: Rizatriptan Pharmaceutical form(s)/strength: Tablets and Oral lyophilisates (wafers) 5 and 10 mg P RMS: NL/H/PSUR/0002/001 Date of FAR: 23.05.2011 4.3 Contraindications
Preventive Effect of Greater Occipital Nerve Block on Severity and Frequency of Migraine Headache
 Proceeding S.Z.P.G.M.I. Vol: 31(2): pp. 75-79, 2017. Preventive effect of Greater Occipital Nerve Block on Severity and Frequency of Migraine Headache Dr. Syed Mehmood Ali, Dr. Mudassar Aslam, Dr. Dawood
Proceeding S.Z.P.G.M.I. Vol: 31(2): pp. 75-79, 2017. Preventive effect of Greater Occipital Nerve Block on Severity and Frequency of Migraine Headache Dr. Syed Mehmood Ali, Dr. Mudassar Aslam, Dr. Dawood
Headache Mary D. Hughes, MD Neuroscience Associates
 Headache Mary D. Hughes, MD Neuroscience Associates Case 1 22 year old female presents with recurrent headaches. She has had headaches for the past 3 years. They start on the right side of her head and
Headache Mary D. Hughes, MD Neuroscience Associates Case 1 22 year old female presents with recurrent headaches. She has had headaches for the past 3 years. They start on the right side of her head and
Post-Operative Cluster Headache Following Carotid Endarterectomy
 Review Received: October 31, 2016 Accepted: January 11, 2017 Published online: February 3, 2017 Post-Operative Cluster Headache Following Carotid Endarterectomy Thijs H.T. Dirkx Peter J. Koehler Department
Review Received: October 31, 2016 Accepted: January 11, 2017 Published online: February 3, 2017 Post-Operative Cluster Headache Following Carotid Endarterectomy Thijs H.T. Dirkx Peter J. Koehler Department
