ISSUES WITH THE PRESENT FDA ON THE MATTER OF 510(k) CLEARANCE
|
|
|
- Lynne Gertrude Harrison
- 6 years ago
- Views:
Transcription
1 PRESENTATION TO IOM COMMITTEE ON 510(k) ISSUES WITH THE PRESENT FDA ON THE MATTER OF 510(k) CLEARANCE Robert E. Fischell, Sc. D. Founder and Chief Technology Officer Neuralieve, Inc. Washington, DC July 28,
2 PRESENTATION TO IOM COMMITTEE ON 510(K) THERE IS A GREAT CONCERN IN THE MEDICAL DEVICE INDUSTRY THAT THE FDA IS SEEKING WAYS TO DELAY AND DISRUPT THE CLEARANCE OF NEW MEDICAL DEVICES UNDER THE 510(k) REGULATIONS 2
3 PRESENTATION TO IOM COMMITTEE ON 510(K) THIS PRESENTATION IS AN EXAMPLE OF HOW THE FDA IS SLOWING THE APPROVAL OF A NEW MEDICAL DEVICE THAT CAN BRING RELIEF, WITHOUT SIDE EFFECTS, TO MILLIONS OF AMERICANS WHO SUFFER FROM MIGRAINE HEADACHES 3
4 NEURALIEVE, INC. NEURALIEVE, INC. WAS FORMED IN THE YEAR 2000 TO DEVELOP AN EXTERNAL DEVICE TO RELIEVE MIGRAINE HEADACHES WITHOUT THE USE OF DRUGS THE EXTERNAL DEVICE PRODUCES AN INTENSE MAGNETIC PULSE ONTO THE BRAIN WHICH CAUSES AN ELECTRICAL PULSE THAT CAUSES DEPOLARIZATION OF NEURONS WHICH HAS BEEN SHOWN TO DISRUPT MIGRAINE SYMPTONS THIS TREATMENT IS CALLED TRANSCRANIAL MAGNETIC STIMULATION OR TMS
5 About Transcranial Magnetic Stimulation (TMS) TMS machines have been used to study brain function for more than 30 years in tens of thousands of patients Neuralieve uses a single pulse TMS that has been cited by the NIH to be Harmless In 2008, the prior FDA cleared a Neuronetics, Inc. de novo 510(k) application to treat depression using a 50% stronger magnetic pulse compared to Neuralieve, and the Neuronetics NeuroStar device provides 30,000 to 75,000 times as many pulses per treatment as compared to the Neuralieve device In 2008 Neuralieve completed a randomized clinical study of TMS in migraine patients with aura that showed that the Neuralieve device for the treatment of migraine headache is both safe and effective
6 Effective in Treating Any Pain Pain Free Rate at 2 hours for Any Baseline Pain P = P = P = P = Full Analysis Set = ITT Full Analysis N =
7 Sustained Pain Free Rates Full Analysis Set (ITT) 2-24h 2-48h Sustained Pain Free 7
8 Symptom-Free Rates at 2 hours Significant improvement for photophobia, phonophobia and nausea P = P = P =
9 TMS Comparable Efficacy to Triptans when used after pain begins When treated during moderate / severe pain TMS 2h Pain Free Triptans 2h Pain Free P=0.019 Percent of patients 35.0% 7.4% N = Adelman, JA., Meta-analysis of oral triptan therapy JMPC 1, Jan-Feb
10 TMS is a Safer Treatment TMS scores highest in patient satisfaction measure SNAE = Sustained pain free with No Adverse Events Efficacy: % patients sustained pain 24 hrs (SPF) Tolerability: (1-AE) % Adverse Events (AE) SNAE score: % SPF(1-AE) Triptan Average 20.0% 64.3% 12.8% TMS 29.3% b 95.1% bb 27.8% bbb Higher SNAE scores = better treatment outcome Source: Triptan SNAE Scores - Use of the Sustained Pain-Free Plus No Adverse Events Endpoint in Clinical Trials of Triptans in Acute Migraine; Dodick et al CNS Drugs 2007 TMS SNAE Scores- Neuralieve Migraine with Aura Study Data, 2008
11 Immediate Target Market Triptan Contra-Indicated Patients Estimated 5.5M patients cannot be treated SAFELY with triptans Pregnancy t 5M pregnancies per year in US ~ 1M lactation (assumes 75% women nursing for 3 months) t 25% migraine prevalence in child bearing age women 1.5M potential patients can not be served by triptans Patients with cardiovascular or pulmonary risk factors t 10 million angina patients (largest group) t Assume 12% migraine prevalence general population 2M potential patients can not be served by triptans Patients taking anti-depressants (SSRI, SNRI; e.g.: Prozac, Zoloft) t 17 million people in US suffer major depression t Migraine sufferers 5 times more likely to develop major depression t Fatal triptan interaction warning from FDA* t Assume 12% migraine prevalence general population (likely higher) 2M potential patients cannot be served by triptan t BECAUSE OF THE FDA, 5.5M AMERICAN PATIENTS CANNOT HAVE THEIR SEVERE HEADACHES RELIEVED WITH THE NEURALIEVE TMS DEVICE BUT MUST SUFFER THE PAIN OF REPEATED MIGRAINE HEADACHES
12 PROBLEMS WITH FDA CLEARANCE Trial that was designed with the help of the FDA is completed successfully June (k) submitted July 2008 with 2 predicates FDA denies predicates in October 2008 De novo 510(k) filed in November 2008 FDA missed reuired 60 day deadline to respond and there was no recourse to get them to respond Summer 2009 FDA branch chief states that because the TMS device would be very popular they must have a higher standard of efficacy for it to be approved. FDA states that we should have a panel meeting to review the data (like for a high risk PMA type device); This is unprecedented. November 2009 FDA delays panel until February 2010
13 PROBLEMS WITH FDA CLEARANCE January 2010 FDA denies our application, no panel, says the device failed to show that it was effective, cites concerns about nausea and safety and now wants a PMA with a huge additional study April 2010 The Lancet medical journal publishes the trial results, authored by the current American Headache Society (AHS) President, the past AHS President and the President of the International Headache Society, saying that TMS is a safe and effective cure for migraine headaches The thought leader in migraine for Germany publishes an editorial in the same Lancet issue stating that the trial was very well designed May 2010 Neuralieve receives a letter from the world leaders in migraine headache that strongly disagrees with the FDA conclusions that the device is not safe and effective June 2010 HEADACHE, the journal of the American Headache Society publishes a Review Article with the President of that Society as the lead author with the conclusion; The safety of stms in clinical practice, including as an acute migraine headache treatment, is supported by biological, empirical, and clinical trial evidence.
14 CONCLUSIONS REGARDING TMS FOR MIGRAINE There is no longer an appeals process that works in the FDA to overrule the lowest level of review, thus the company must commercialize overseas to raise money and cannot hire Americans into good paying jobs as a result More than 5 million American patients who cannot take migraine medications are denied this safe and effective therapy and therefore must suffer 10s of millions of severe migraine headaches without help from a device that has been proven in clinical studies to be safe and effective Some Americans will be able to go to Europe to get the device to treat their migraine headaches; but the vast majority of American migraine patients will not have ready access to the TMS device
15 TMS FOR TREATING MIGRAINE HEADACHE CONCLUSION TIME FOR QUESTIONS?
A Patient s Guide: Pursuing Health Care. Reimbursement. for NeuroStar TMS Therapy
 A Patient s Guide: Pursuing Health Care Reimbursement for NeuroStar TMS Therapy Partnering With Your Healthcare Provider Your healthcare provider is a medical professional who is trained to assess your
A Patient s Guide: Pursuing Health Care Reimbursement for NeuroStar TMS Therapy Partnering With Your Healthcare Provider Your healthcare provider is a medical professional who is trained to assess your
Bringing. to patients with depression
 Bringing to patients with depression Hope is knowing remission from depression is possible Depression is a serious illness that affects approximately 350 million people worldwide. 1 While medications may
Bringing to patients with depression Hope is knowing remission from depression is possible Depression is a serious illness that affects approximately 350 million people worldwide. 1 While medications may
FDA CLEARS NEUROSTAR TMS THERAPY FOR THE TREATMENT OF DEPRESSION
 FOR IMMEDIATE RELEASE FDA CLEARS NEUROSTAR TMS THERAPY FOR THE TREATMENT OF DEPRESSION First and Only Non-systemic and Non-invasive Treatment Cleared for Patients Who Have Not Benefited From Prior Antidepressant
FOR IMMEDIATE RELEASE FDA CLEARS NEUROSTAR TMS THERAPY FOR THE TREATMENT OF DEPRESSION First and Only Non-systemic and Non-invasive Treatment Cleared for Patients Who Have Not Benefited From Prior Antidepressant
Frequently Asked Questions FAQS. NeuroStar TMS Therapies
 Frequently Asked Questions FAQS NeuroStar TMS Therapies Provided by Dr Terrence A. Boyadjis MD 790 E Market Street Suite 245 West Chester, PA 19382 610.738.9576 FAQS About TMS Therapies Page 1 NeuroStar
Frequently Asked Questions FAQS NeuroStar TMS Therapies Provided by Dr Terrence A. Boyadjis MD 790 E Market Street Suite 245 West Chester, PA 19382 610.738.9576 FAQS About TMS Therapies Page 1 NeuroStar
What is new in the migraine world! Modar Khalil Consultant neurologist Hull Royal Infirmary
 What is new in the migraine world! Modar Khalil Consultant neurologist Hull Royal Infirmary Overview Understanding the burden Commonly used terms Acute therapy What we currently have What we are going
What is new in the migraine world! Modar Khalil Consultant neurologist Hull Royal Infirmary Overview Understanding the burden Commonly used terms Acute therapy What we currently have What we are going
a proven non-drug treatment for depression Get back to well without medication side effects right in your doctor s office.
 a proven non-drug treatment for depression Get back to well......without medication side effects......right in your doctor s office. Is NeuroStar TMS Therapy right for you? 1) Are you are taking medication
a proven non-drug treatment for depression Get back to well......without medication side effects......right in your doctor s office. Is NeuroStar TMS Therapy right for you? 1) Are you are taking medication
Transcranial Magnetic Stimulation and Cranial Electrical Stimulation (CES) as a Treatment of Depression and Other Psychiatric/Neurologic Disorders
 Transcranial Magnetic Stimulation and Cranial Electrical Stimulation (CES) as a Treatment of Depression and Other Psychiatric/Neurologic Disorders Policy Number: 2.01.50 Last Review: 10/2017 Origination:
Transcranial Magnetic Stimulation and Cranial Electrical Stimulation (CES) as a Treatment of Depression and Other Psychiatric/Neurologic Disorders Policy Number: 2.01.50 Last Review: 10/2017 Origination:
tens_(transcutaneous_electrical_nerve_stimulator) 7/ / / /2014 This policy is NOT effective until January 13, 2015
 Corporate Medical Policy TENS (Transcutaneous Electrical Nerve Stimulator) File Name: Origination: Last CAP Review: Next CAP Review: Last Review: tens_(transcutaneous_electrical_nerve_stimulator) 7/1982
Corporate Medical Policy TENS (Transcutaneous Electrical Nerve Stimulator) File Name: Origination: Last CAP Review: Next CAP Review: Last Review: tens_(transcutaneous_electrical_nerve_stimulator) 7/1982
Remission From Depression Is Possible
 Remission From Depression Is Possible About Depression Over 350 million people worldwide suffer from depression 1 While medications may help manage symptoms of depression, many patients are not satisfied
Remission From Depression Is Possible About Depression Over 350 million people worldwide suffer from depression 1 While medications may help manage symptoms of depression, many patients are not satisfied
34 th Annual J.P. Morgan Healthcare Conference
 JANUARY 2016 34 th Annual J.P. Morgan Healthcare Conference [ 1 ] Forward Looking Statements SPECIAL NOTE REGARDING FORWARD LOOKING STATEMENTS In addition to historical information, this presentation contains
JANUARY 2016 34 th Annual J.P. Morgan Healthcare Conference [ 1 ] Forward Looking Statements SPECIAL NOTE REGARDING FORWARD LOOKING STATEMENTS In addition to historical information, this presentation contains
Migraine: Developing Drugs for Acute Treatment Guidance for Industry
 Migraine: Developing Drugs for Acute Treatment Guidance for Industry U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) February 2018
Migraine: Developing Drugs for Acute Treatment Guidance for Industry U.S. Department of Health and Human Services Food and Drug Administration Center for Drug Evaluation and Research (CDER) February 2018
NOW I M A NEUROSTAR. Let Your Best Self Shine
 NOW I M A NEUROSTAR. Let Your Best Self Shine It s Time for a New You NeuroStar Advanced Therapy is an in-office treatment that requires no anesthesia or sedation. You are awake and alert during treatment
NOW I M A NEUROSTAR. Let Your Best Self Shine It s Time for a New You NeuroStar Advanced Therapy is an in-office treatment that requires no anesthesia or sedation. You are awake and alert during treatment
Transcranial Magnetic Stimulation (TMS)
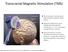 Transcranial Magnetic Stimulation (TMS) The treatment coil produces MRI-strength magnetic field pulses. Magnetic field pulses pass unimpeded through the cranium for 2-3 cm. and induce a small electric
Transcranial Magnetic Stimulation (TMS) The treatment coil produces MRI-strength magnetic field pulses. Magnetic field pulses pass unimpeded through the cranium for 2-3 cm. and induce a small electric
Repetitive Transcranial Magnetic Stimulation as a for Treatment of Refractory Depression and other Psychiatric/Neurologic Disorders
 Repetitive Transcranial Magnetic Stimulation as a for Treatment of Refractory Depression and other Psychiatric/Neurologic Disorders Policy Number: Original Effective Date: MM.12.015 08/01/2014 Line(s)
Repetitive Transcranial Magnetic Stimulation as a for Treatment of Refractory Depression and other Psychiatric/Neurologic Disorders Policy Number: Original Effective Date: MM.12.015 08/01/2014 Line(s)
Does maxalt cause high blood pressure
 Does maxalt cause high blood pressure The Borg System is 100 % Does maxalt cause high blood pressure You should not use this medicine if you have uncontrolled high blood pressure, heart problems, a history
Does maxalt cause high blood pressure The Borg System is 100 % Does maxalt cause high blood pressure You should not use this medicine if you have uncontrolled high blood pressure, heart problems, a history
Disclosures. Triptans for Kids 5/16/13
 5/16/13 Disclosures Triptans for Kids Amy A. Gelfand, MD GelfandA@neuropeds.ucsf.edu Departments of Neurology and Pediatrics UCSF Child Neurology and Headache Center I receive grant funding from: NIH/NINDS
5/16/13 Disclosures Triptans for Kids Amy A. Gelfand, MD GelfandA@neuropeds.ucsf.edu Departments of Neurology and Pediatrics UCSF Child Neurology and Headache Center I receive grant funding from: NIH/NINDS
Primer: Medical Device User Fee Amendments Han Zhong l September 2011
 Primer: Medical Device User Fee Amendments Han Zhong l September 2011 Introduction The Medical Device User Fee and Modernization Act (MDUFMA or MDUFA) is a set of agreements between the Food and Drug Administration
Primer: Medical Device User Fee Amendments Han Zhong l September 2011 Introduction The Medical Device User Fee and Modernization Act (MDUFMA or MDUFA) is a set of agreements between the Food and Drug Administration
Transcranial Magnetic Stimulation for the Treatment of Depression
 Transcranial Magnetic Stimulation for the Treatment of Depression Paul E. Holtzheimer, MD Associate Professor Departments of Psychiatry and Surgery Geisel School of Medicine at Dartmouth Dartmouth-Hitchcock
Transcranial Magnetic Stimulation for the Treatment of Depression Paul E. Holtzheimer, MD Associate Professor Departments of Psychiatry and Surgery Geisel School of Medicine at Dartmouth Dartmouth-Hitchcock
NEURONETICS. 510(k) SUMMARY NEUROSTAR TMS THERAPY SYSTEM'
 NEURONETICS 510(k) SUMMARY NEUROSTAR TMS THERAPY SYSTEM' 510(k) Owner: Neuronetics, Inc. DEC 1 6 Z008 1 Great Valley Parkway, Suite 2 Malvern, PA 19355 Phone: 610-640-4202 Fax: 610-640-4206 Company Contact:
NEURONETICS 510(k) SUMMARY NEUROSTAR TMS THERAPY SYSTEM' 510(k) Owner: Neuronetics, Inc. DEC 1 6 Z008 1 Great Valley Parkway, Suite 2 Malvern, PA 19355 Phone: 610-640-4202 Fax: 610-640-4206 Company Contact:
BMS for the Acute Treatment of Migraine: A Double-Blind, Randomized, Placebo Controlled, Dose-Ranging Trial
 BMS-927711 for the Acute Treatment of Migraine: A Double-Blind, Randomized, Placebo Controlled, Dose-Ranging Trial Ronald Marcus, MD Chief Medical Officer Spinifex Pharmaceuticals Migraine: Case Study
BMS-927711 for the Acute Treatment of Migraine: A Double-Blind, Randomized, Placebo Controlled, Dose-Ranging Trial Ronald Marcus, MD Chief Medical Officer Spinifex Pharmaceuticals Migraine: Case Study
Triptans: Nonresponse, Recurrence, and Serious AEs for Many Patients
 Efficacy, Safety, and Tolerability of Rimegepant 75 mg, an Oral CGRP Receptor Antagonist, for the Acute Treatment of Migraine: Results from a Phase 3, Double-Blind, Randomized, Placebo-Controlled Trial,
Efficacy, Safety, and Tolerability of Rimegepant 75 mg, an Oral CGRP Receptor Antagonist, for the Acute Treatment of Migraine: Results from a Phase 3, Double-Blind, Randomized, Placebo-Controlled Trial,
Ana Podgorac Belgrade, May 2012
 Headache and reproductive life Ana Podgorac Belgrade, May 2012 52 years old woman, English teacher in primary school, married, mother of two, with a history of migraine without aura. Over the last 6 months
Headache and reproductive life Ana Podgorac Belgrade, May 2012 52 years old woman, English teacher in primary school, married, mother of two, with a history of migraine without aura. Over the last 6 months
Supraorbital nerve stimulation Cefaly Device - FDA Approved for migraine prevention (also being investigated as acute therapy)
 NEUROSTIMULATION/NEUROMODULATION UPDATE Meyer and Renee Luskin Andrew Charles, M.D. Professor Luskin Chair in Migraine and Headache Studies Director, UCLA Goldberg Migraine Program David Geffen School
NEUROSTIMULATION/NEUROMODULATION UPDATE Meyer and Renee Luskin Andrew Charles, M.D. Professor Luskin Chair in Migraine and Headache Studies Director, UCLA Goldberg Migraine Program David Geffen School
Patient Education Brief. NeuroStar TMS Therapies
 Patient Education Brief NeuroStar TMS Therapies Provided by Dr Terrence A. Boyadjis MD 790 E Market Street Suite 245 West Chester, PA 19382 610.738.9576 About Depression Depression is a serious medical
Patient Education Brief NeuroStar TMS Therapies Provided by Dr Terrence A. Boyadjis MD 790 E Market Street Suite 245 West Chester, PA 19382 610.738.9576 About Depression Depression is a serious medical
Corporate Medical Policy
 Corporate Medical Policy TENS (Transcutaneous Electrical Nerve Stimulator) File Name: Origination: Last CAP Review: Next CAP Review: Last Review: tens_(transcutaneous_electrical_nerve_stimulator) 7/1982
Corporate Medical Policy TENS (Transcutaneous Electrical Nerve Stimulator) File Name: Origination: Last CAP Review: Next CAP Review: Last Review: tens_(transcutaneous_electrical_nerve_stimulator) 7/1982
Brain Stimulation. Berry S. Anderson, PhD, RN Mary Rosedale, PhD, PMHNP-BC, NEA-BC Theresa Kormos, PMHCNS-BC Cindy Brown, BSN, RN
 Brain Stimulation American Psychiatric Nurses Association 24th Annual Conference October 16, 2010 Kentucky International Convention Center Louisville, Kentucky 1 Berry S. Anderson, PhD, RN Mary Rosedale,
Brain Stimulation American Psychiatric Nurses Association 24th Annual Conference October 16, 2010 Kentucky International Convention Center Louisville, Kentucky 1 Berry S. Anderson, PhD, RN Mary Rosedale,
Dubai Standards of Care (Migraine)
 Dubai Standards of Care 2018 (Migraine) Preface Migraine is one of the most common problem dealt with in daily practice. In Dubai, the management of migraine is done through various different strategies.
Dubai Standards of Care 2018 (Migraine) Preface Migraine is one of the most common problem dealt with in daily practice. In Dubai, the management of migraine is done through various different strategies.
David L. Rosen, B.S. Pharm., J.D. Partner, Adv. and FDA Regulatory Practice Co-chair, Life Sciences Industry Team Washington, DC
 SUCCESSFULLY DEALING WITH BUSINESS, REGULATORY AND IP CHALLENGES IN ISRAELI BIOMED COMPANIES: Current FDA Issues Impacting the Regulation of BioMed Companies David L. Rosen, B.S. Pharm., J.D. Partner,
SUCCESSFULLY DEALING WITH BUSINESS, REGULATORY AND IP CHALLENGES IN ISRAELI BIOMED COMPANIES: Current FDA Issues Impacting the Regulation of BioMed Companies David L. Rosen, B.S. Pharm., J.D. Partner,
Commercial-Stage Bioelectronic Medicine Company 1 st FDA-cleared non-invasive vagus nerve stimulation platform therapy
 Commercial-Stage Bioelectronic Medicine Company 1 st FDA-cleared non-invasive vagus nerve stimulation platform therapy 2018 electrocore. All rights reserved. 1 Disclaimer This presentation may contain
Commercial-Stage Bioelectronic Medicine Company 1 st FDA-cleared non-invasive vagus nerve stimulation platform therapy 2018 electrocore. All rights reserved. 1 Disclaimer This presentation may contain
Forward Looking Statements and Further Information
 JANUARY 2015 [ 1 ] Forward Looking Statements and Further Information SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS This presentation contains forward-looking statements. All statements other than
JANUARY 2015 [ 1 ] Forward Looking Statements and Further Information SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS This presentation contains forward-looking statements. All statements other than
Adult & Pediatric Patients. Stanford Health Care, Division Pain Medicine
 Acute Treatment Strategies in Adult & Pediatric Patients Theresa Mallick Searle, MS, RN BC, ANP BC Disclosures Speakers Bureau: Allergan, Depomed Acute Treatment Strategies in Adult & Pediatric Patients
Acute Treatment Strategies in Adult & Pediatric Patients Theresa Mallick Searle, MS, RN BC, ANP BC Disclosures Speakers Bureau: Allergan, Depomed Acute Treatment Strategies in Adult & Pediatric Patients
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Transcranial Magnetic Stimulation (TMS) Page 1 of 21 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: See also: Transcranial Magnetic Stimulation (TMS) Vagus Nerve
Transcranial Magnetic Stimulation (TMS) Page 1 of 21 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: See also: Transcranial Magnetic Stimulation (TMS) Vagus Nerve
Study No.:MPX Title: Rationale: Phase: IIB Study Period: Study Design: Centres: Indication: Treatment: Objectives:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Headache Master School Japan-Osaka 2016 (HMSJ-Osaka2016) October 23, II. Management of Refractory Headaches
 Headache Master School Japan-Osaka 2016 (HMSJ-Osaka2016) October 23, 2016 II. Management of Refractory Headaches Case presentation 1: A case of intractable daily-persistent headache Keio University School
Headache Master School Japan-Osaka 2016 (HMSJ-Osaka2016) October 23, 2016 II. Management of Refractory Headaches Case presentation 1: A case of intractable daily-persistent headache Keio University School
10/17/2017 CHRONIC MIGRAINES BOTOX: TO INJECT OR NOT INJECT? IN CHRONIC MIGRAINE PROPHYLAXIS OBJECTIVES PATIENT CASE EPIDEMIOLOGY EPIDEMIOLOGY
 BOTOX: TO INJECT OR NOT INJECT? IN CHRONIC MIGRAINE PROPHYLAXIS OBJECTIVES JENNIFER SHIN, PHARMD PGY2 AMBULATORY CARE PHARMACY RESIDENT COMMUNITYCARE HEALTH CENTERS PHARMACOTHERAPY ROUNDS OCTOBER 20, 2017
BOTOX: TO INJECT OR NOT INJECT? IN CHRONIC MIGRAINE PROPHYLAXIS OBJECTIVES JENNIFER SHIN, PHARMD PGY2 AMBULATORY CARE PHARMACY RESIDENT COMMUNITYCARE HEALTH CENTERS PHARMACOTHERAPY ROUNDS OCTOBER 20, 2017
Lasmiditan (200 mg and 100 mg) Compared to Placebo for Acute Treatment of Migraine
 (200 mg and 100 mg) Compared to for Acute Treatment of Migraine Bernice Kuca, M.S. 1 ; Linda A. Wietecha, B.S.N., M.S. 2 ; Paul H. Berg, M.S. 2 ; Sheena K. Aurora, M.D. 2 1 CoLucid Pharmaceuticals, Inc.,
(200 mg and 100 mg) Compared to for Acute Treatment of Migraine Bernice Kuca, M.S. 1 ; Linda A. Wietecha, B.S.N., M.S. 2 ; Paul H. Berg, M.S. 2 ; Sheena K. Aurora, M.D. 2 1 CoLucid Pharmaceuticals, Inc.,
CMA Response: Health Canada s Medical Marihuana Regulatory Proposal. Submitted to the Office of Controlled Substances Health Canada.
 CMA Response: Health Canada s Medical Marihuana Regulatory Proposal Submitted to the Office of Controlled Substances Health Canada February 28, 2013 A healthy population and a vibrant medical profession
CMA Response: Health Canada s Medical Marihuana Regulatory Proposal Submitted to the Office of Controlled Substances Health Canada February 28, 2013 A healthy population and a vibrant medical profession
Becky Stuckey 4 th November 2017 Mercure Exeter
 Becky Stuckey 4 th November 2017 Mercure Exeter There are currently 30 headache nurses nationally Approximately 37 Neurologist led headache clinics in England. Headache should be ideally managed in primary
Becky Stuckey 4 th November 2017 Mercure Exeter There are currently 30 headache nurses nationally Approximately 37 Neurologist led headache clinics in England. Headache should be ideally managed in primary
OCTOBER 7-10 PHILADELPHIA, PENNSYLVANIA
 OMED 17 OCTOBER 7-10 PHILADELPHIA, PENNSYLVANIA 29.5 Category 1-A CME credits anticipated ACOFP / AOA s 122 nd Annual Osteopathic Medical Conference & Exposition Joint Session with ACOFP, ACONP and AOAAM:
OMED 17 OCTOBER 7-10 PHILADELPHIA, PENNSYLVANIA 29.5 Category 1-A CME credits anticipated ACOFP / AOA s 122 nd Annual Osteopathic Medical Conference & Exposition Joint Session with ACOFP, ACONP and AOAAM:
Use of Standards in Substantial Equivalence Determinations
 Guidance for Industry and for FDA Staff Use of Standards in Substantial Equivalence Determinations Document issued on: March 12, 2000 U.S. Department Of Health And Human Services Food and Drug Administration
Guidance for Industry and for FDA Staff Use of Standards in Substantial Equivalence Determinations Document issued on: March 12, 2000 U.S. Department Of Health And Human Services Food and Drug Administration
Transcranial Magnetic Stimulation and Cranial Electrical Stimulation (CES) as a Treatment of Depression and Other Psychiatric/Neurologic Disorders
 Transcranial Magnetic Stimulation and Cranial Electrical Stimulation (CES) as a Treatment of Depression and Other Psychiatric/Neurologic Disorders Policy Number: 2.01.50 Last Review: 10/2014 Origination:
Transcranial Magnetic Stimulation and Cranial Electrical Stimulation (CES) as a Treatment of Depression and Other Psychiatric/Neurologic Disorders Policy Number: 2.01.50 Last Review: 10/2014 Origination:
MIGRAINE A MYSTERY HEADACHE
 MIGRAINE A MYSTERY HEADACHE The migraine is a chronic neurological disease that is characterized by moderate to severe episodes of headache that is mostly associated with other central nervous system (CNS)
MIGRAINE A MYSTERY HEADACHE The migraine is a chronic neurological disease that is characterized by moderate to severe episodes of headache that is mostly associated with other central nervous system (CNS)
Medical Policy An independent licensee of the Blue Cross Blue Shield Association
 Transcranial Magnetic Stimulation (TMS) Page 1 of 24 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: See also: Transcranial Magnetic Stimulation (TMS) Vagus Nerve
Transcranial Magnetic Stimulation (TMS) Page 1 of 24 Medical Policy An independent licensee of the Blue Cross Blue Shield Association Title: See also: Transcranial Magnetic Stimulation (TMS) Vagus Nerve
FDA Regulation of Diagnostic Tests Jeffrey N. Gibbs Hyman, Phelps & McNamara, P.C. Washington, DC
 AIPLA Annual Meeting Joint Biotechnology Committee/ Special Committee on FDA Law Program October 21, 2010 Marriott Wardman Park Hotel Washington, DC FDA Regulation of Diagnostic Tests Jeffrey N. Gibbs
AIPLA Annual Meeting Joint Biotechnology Committee/ Special Committee on FDA Law Program October 21, 2010 Marriott Wardman Park Hotel Washington, DC FDA Regulation of Diagnostic Tests Jeffrey N. Gibbs
Transcranial Magnetic Stimulation as a Treatment of Depression and Other Psychiatric/Neurologic Disorders Corporate Medical Policy
 Transcranial Magnetic Stimulation as a Treatment of Depression and Other Psychiatric/Neurologic Disorders Corporate Medical Policy File name: Transcranial Magnetic Stimulation as a Treatment of Depression
Transcranial Magnetic Stimulation as a Treatment of Depression and Other Psychiatric/Neurologic Disorders Corporate Medical Policy File name: Transcranial Magnetic Stimulation as a Treatment of Depression
Migraine much more than just a headache
 Migraine much more than just a headache Session hosted by Teva UK Limited PUU4 11:15 12:15 UK/NHSS/18/0021b Date of Preparation: August 2018 The views expressed in this presentation are those of the speaker
Migraine much more than just a headache Session hosted by Teva UK Limited PUU4 11:15 12:15 UK/NHSS/18/0021b Date of Preparation: August 2018 The views expressed in this presentation are those of the speaker
What is the Effectiveness of OnabotulinumtoxinA (Botox ) in Reducing the Number of Chronic Migraines (CM) in Patients Years Old?
 Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2013 What is the Effectiveness of OnabotulinumtoxinA
Philadelphia College of Osteopathic Medicine DigitalCommons@PCOM PCOM Physician Assistant Studies Student Scholarship Student Dissertations, Theses and Papers 2013 What is the Effectiveness of OnabotulinumtoxinA
Guidance for Industry Migraine: Developing Drugs for Acute Treatment
 Guidance for Industry Migraine: Developing Drugs for Acute Treatment DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this draft
Guidance for Industry Migraine: Developing Drugs for Acute Treatment DRAFT GUIDANCE This guidance document is being distributed for comment purposes only. Comments and suggestions regarding this draft
AVEO and Astellas Report Final Overall Survival Results from TIVO-1
 AVEO and Astellas Report Final Overall Survival Results from TIVO-1 - Median Overall Survival of 28.8 Months Reported for Tivozanib in Patients with Advanced Kidney Cancer - CAMBRIDGE, Mass. and TOKYO,
AVEO and Astellas Report Final Overall Survival Results from TIVO-1 - Median Overall Survival of 28.8 Months Reported for Tivozanib in Patients with Advanced Kidney Cancer - CAMBRIDGE, Mass. and TOKYO,
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centers: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Key CDRH Regulatory Initiatives
 Key CDRH Regulatory Initiatives Revamping the Submission Process Creating New Strategies Ralph F. Hall University of Minnesota Law School Counsel Faegre Baker & Daniels May 8, 2012 Purpose of Today s Program
Key CDRH Regulatory Initiatives Revamping the Submission Process Creating New Strategies Ralph F. Hall University of Minnesota Law School Counsel Faegre Baker & Daniels May 8, 2012 Purpose of Today s Program
Rimegepant Pivotal Phase 3 Trial Results - Conference Call March 26, Biohaven
 Rimegepant Pivotal Phase 3 Trial Results - Conference Call March 26, 2018 1 Forward-Looking Statement This presentation contains forward-looking statements, including: statements about our plans to develop
Rimegepant Pivotal Phase 3 Trial Results - Conference Call March 26, 2018 1 Forward-Looking Statement This presentation contains forward-looking statements, including: statements about our plans to develop
Prevention and Treatment of Migraines CAITLIN BARNES, PHARM.D. CANDIDATE AMBULATORY CARE JOE CAMMILLERI, PHARM.D. NATOHYA MALLORY, PHARM.D.
 Prevention and Treatment of Migraines CAITLIN BARNES, PHARM.D. CANDIDATE AMBULATORY CARE JOE CAMMILLERI, PHARM.D. NATOHYA MALLORY, PHARM.D. Objectives Present patient case Review epidemiology/pathophysiology
Prevention and Treatment of Migraines CAITLIN BARNES, PHARM.D. CANDIDATE AMBULATORY CARE JOE CAMMILLERI, PHARM.D. NATOHYA MALLORY, PHARM.D. Objectives Present patient case Review epidemiology/pathophysiology
Bright Nights: Understanding Depression
 Bright Nights: Understanding Depression KEVIN SETHI, MD MARCH 18 TH, 2019 Objectives Features of Depression Facts and Myths Treatment Options Medications Neuromodulation Cognitive Behavioral Therapy Mindfulness
Bright Nights: Understanding Depression KEVIN SETHI, MD MARCH 18 TH, 2019 Objectives Features of Depression Facts and Myths Treatment Options Medications Neuromodulation Cognitive Behavioral Therapy Mindfulness
Neuro-MS/D DIAGNOSTICS REHABILITATION TREATMENT STIMULATION. Transcranial Magnetic Stimulator. of motor disorders after the stroke
 Neuro-MS/D Transcranial Magnetic Stimulator DIAGNOSTICS of corticospinal pathway pathology REHABILITATION of motor disorders after the stroke TREATMENT of depression and Parkinson s disease STIMULATION
Neuro-MS/D Transcranial Magnetic Stimulator DIAGNOSTICS of corticospinal pathway pathology REHABILITATION of motor disorders after the stroke TREATMENT of depression and Parkinson s disease STIMULATION
Chronic Migraine in Primary Care. December 11 th, 2017 Werner J. Becker University of Calgary
 Chronic Migraine in Primary Care December 11 th, 2017 Werner J. Becker University of Calgary Disclosures Faculty: Werner J. Becker Relationships with commercial interests: Grants/Research Support: Clinical
Chronic Migraine in Primary Care December 11 th, 2017 Werner J. Becker University of Calgary Disclosures Faculty: Werner J. Becker Relationships with commercial interests: Grants/Research Support: Clinical
Comments of the Patient, Consumer, and Public Health Coalition. Strengthening the Center for Devices and Radiological Health s 510(k) Review Process
 March 19, 2010 Division of Dockets Management (HFA-305) Food and Drug Administration 5630 Fishers Lane, Room 1061 Rockville, MD 20852 Comments of the Patient, Consumer, and Public Health Coalition on Strengthening
March 19, 2010 Division of Dockets Management (HFA-305) Food and Drug Administration 5630 Fishers Lane, Room 1061 Rockville, MD 20852 Comments of the Patient, Consumer, and Public Health Coalition on Strengthening
Controlling Migraine Pain
 Migraine Stats Controlling Migraine Pain Alan Zacharias, M.D. Associated Neurologists, Boulder Community Health 303-622-3365 Women 15% Men 5% Usually starts in 2 nd and 3 rd Decade Major Impact on days
Migraine Stats Controlling Migraine Pain Alan Zacharias, M.D. Associated Neurologists, Boulder Community Health 303-622-3365 Women 15% Men 5% Usually starts in 2 nd and 3 rd Decade Major Impact on days
Joint Meeting. and the Peripheral and Central Nervous System Drugs Advisory Committee of the Center for Drug Evaluation and Research
 Testimony to the Department of Health and Human Services Food and Drug Administration Joint Meeting of the Dental Products Panel of the Medical Devices Advisory Committee of the Center for Devices and
Testimony to the Department of Health and Human Services Food and Drug Administration Joint Meeting of the Dental Products Panel of the Medical Devices Advisory Committee of the Center for Devices and
Scientific Panel MindFreedom Support Coalition International 454 Willamette, Suite 216 PO Box Eugene, OR USA
 Scientific Panel MindFreedom Support Coalition International 454 Willamette, Suite 216 PO Box 11284 Eugene, OR 97440-3484 USA 20 September 2004 Cathryn M. Clary, MD, MBA, Vice President Psychiatry, Neurology,
Scientific Panel MindFreedom Support Coalition International 454 Willamette, Suite 216 PO Box 11284 Eugene, OR 97440-3484 USA 20 September 2004 Cathryn M. Clary, MD, MBA, Vice President Psychiatry, Neurology,
1/25/2018 ARE CGRP ANTAGONISTS ANY BETTER THAN CURRENT EVIDENCE BASED TREATMENTS? Disclosures: Objectives: Headache Division
 ARE CGRP ANTAGONISTS ANY BETTER THAN CURRENT EVIDENCE BASED TREATMENTS? Lawrence C Newman, MD, FAHS, FAAN Clinical Professor of Neurology Disclosures: Advisory Board: Alder, Allergan, Amgen, Lilly, Supernus,
ARE CGRP ANTAGONISTS ANY BETTER THAN CURRENT EVIDENCE BASED TREATMENTS? Lawrence C Newman, MD, FAHS, FAAN Clinical Professor of Neurology Disclosures: Advisory Board: Alder, Allergan, Amgen, Lilly, Supernus,
Evidence-based Practice And Standards For Medical Devices. Rita F. Redberg, MD, MSc, FACC, FAHA University of California, San Francisco
 Evidence-based Practice And Standards For Medical Devices Rita F. Redberg, MD, MSc, FACC, FAHA University of California, San Francisco 2 3 Regulatory Principles Data Transparency and Accessibility High
Evidence-based Practice And Standards For Medical Devices Rita F. Redberg, MD, MSc, FACC, FAHA University of California, San Francisco 2 3 Regulatory Principles Data Transparency and Accessibility High
What is in this leaflet
 Package leaflet: Information for the user 2.5 mg film-coated tablets Naratriptan Read all of this leaflet carefully before you start taking this medicine because it contains important information for you.
Package leaflet: Information for the user 2.5 mg film-coated tablets Naratriptan Read all of this leaflet carefully before you start taking this medicine because it contains important information for you.
HRSA Grant: D54HP Project Investigator: Carol Monson, DO, MS, FACOFP
 Michigan State University College of Osteopathic Medicine Family and Community Medicine and Internal Medicine Division of Geriatrics www.com.msu.edu/fcm HRSA Grant: D54HP23284-05-00 Project Investigator:
Michigan State University College of Osteopathic Medicine Family and Community Medicine and Internal Medicine Division of Geriatrics www.com.msu.edu/fcm HRSA Grant: D54HP23284-05-00 Project Investigator:
Position Specification
 Position Specification Empirical Spine, Inc. Title: Vice President, Clinical & Regulatory Affairs Opportunity: The Vice President, Clinical & Regulatory Affairs will play a key role in the success of this
Position Specification Empirical Spine, Inc. Title: Vice President, Clinical & Regulatory Affairs Opportunity: The Vice President, Clinical & Regulatory Affairs will play a key role in the success of this
IRB review of device studies
 Washington University School of Medicine Digital Commons@Becker 2011 Device Advice from the FDA 2011 Conferences 2011 IRB review of device studies Jonathan M. Green Washington University School of Medicine
Washington University School of Medicine Digital Commons@Becker 2011 Device Advice from the FDA 2011 Conferences 2011 IRB review of device studies Jonathan M. Green Washington University School of Medicine
Path to Value and Profitability
 Path to Value and Profitability June 4, 2015 Tim Clackson, Ph.D. President of R&D, Chief Scientific Officer ARIAD Pharmaceuticals, Inc. Elsa So Non-small cell lung cancer ARIAD clinical trial patient Some
Path to Value and Profitability June 4, 2015 Tim Clackson, Ph.D. President of R&D, Chief Scientific Officer ARIAD Pharmaceuticals, Inc. Elsa So Non-small cell lung cancer ARIAD clinical trial patient Some
Medical Devices and the Public s Health: The FDA 510(k) Clearance Process at 35 Years. Written Statement of
 Medical Devices and the Public s Health: The FDA 510(k) Clearance Process at 35 Years Written Statement of Dr. David R. Challoner Vice President for Health Affairs, Emeritus University of Florida and Chair,
Medical Devices and the Public s Health: The FDA 510(k) Clearance Process at 35 Years Written Statement of Dr. David R. Challoner Vice President for Health Affairs, Emeritus University of Florida and Chair,
Clinical Trials. Hans-Christoph Diener Senior Professor of Clinical Neuroscienes Medical Faculty University Duisburg-Essen Germany
 Clinical Trials Hans-Christoph Diener Senior Professor of Clinical Neuroscienes Medical Faculty University Duisburg-Essen Germany Conflict of Interest Statement German Research Council German Ministry
Clinical Trials Hans-Christoph Diener Senior Professor of Clinical Neuroscienes Medical Faculty University Duisburg-Essen Germany Conflict of Interest Statement German Research Council German Ministry
What? Who? When? Where? How?
 Who? What? When? Where? How? WHO As she stands at the window wiping tears from her eyes, a mother feels relief watching her child play with friends outside. Not long before, that same mother would never
Who? What? When? Where? How? WHO As she stands at the window wiping tears from her eyes, a mother feels relief watching her child play with friends outside. Not long before, that same mother would never
Current Migraine Treatment Therapy. Daniel Kassicieh, DO, FAAN
 Current Migraine Treatment Therapy Daniel Kassicieh, DO, FAAN Migraine a Disease Process Migraines are a chronic disease process similar to many other chronic medical conditions Migraine has a low mortality
Current Migraine Treatment Therapy Daniel Kassicieh, DO, FAAN Migraine a Disease Process Migraines are a chronic disease process similar to many other chronic medical conditions Migraine has a low mortality
Strategies in Migraine Care
 Strategies in Migraine Care Julie L. Roth, MD Rhode Island Hospital Assistant Professor, Neurology The Warren Alpert Medical School of Brown University March 28, 2015 Financial Disclosures None. Objectives
Strategies in Migraine Care Julie L. Roth, MD Rhode Island Hospital Assistant Professor, Neurology The Warren Alpert Medical School of Brown University March 28, 2015 Financial Disclosures None. Objectives
Summary A LOOK AT ELAGOLIX FOR ENDOMETRIOSIS JULY 2018
 JULY 2018 WHAT IS ENDOMETRIOSIS? Endometriosis is a chronic gynecological condition with symptoms that include painful menstrual periods, nonmenstrual pelvic pain, and pain during intercourse, as well
JULY 2018 WHAT IS ENDOMETRIOSIS? Endometriosis is a chronic gynecological condition with symptoms that include painful menstrual periods, nonmenstrual pelvic pain, and pain during intercourse, as well
The Challenge of Treating Pain
 FDA Charge to the Committee: FDA Opioid Action Plan and Incorporating the Broader Public Health Impact into the Formal Risk-Benefit Assessment for Opioids Robert M. Califf, MD Commissioner of Food and
FDA Charge to the Committee: FDA Opioid Action Plan and Incorporating the Broader Public Health Impact into the Formal Risk-Benefit Assessment for Opioids Robert M. Califf, MD Commissioner of Food and
Regulatory Status FDA approved indication: Migranal Nasal Spray is indicated for the acute treatment of migraine headaches with or without aura (1).
 Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.60 Subject: Migranal Nasal Spray Page: 1 of 5 Last Review Date: November 30, 2018 Migranal Nasal Spray
Federal Employee Program 1310 G Street, N.W. Washington, D.C. 20005 202.942.1000 Fax 202.942.1125 5.70.60 Subject: Migranal Nasal Spray Page: 1 of 5 Last Review Date: November 30, 2018 Migranal Nasal Spray
Phone: +44 (0) or BioPortfolio Limited
 Europe Neuromodulation Devices Market by Procedure [Internal Stimulation (DBS, GES, SNS, SCS, VNS), and External Stimulation (TENS, TMS)], and by Geography Analysis & Forecast to 2019 Europe Neuromodulation
Europe Neuromodulation Devices Market by Procedure [Internal Stimulation (DBS, GES, SNS, SCS, VNS), and External Stimulation (TENS, TMS)], and by Geography Analysis & Forecast to 2019 Europe Neuromodulation
Todd Hutton, M.D. Karl Lanocha, M.D Richard Bermudes, M.D. Kimberly Cress, M.D.
 Transcranial Magnetic Stimulation: What You Need To Know Todd Hutton, M.D. Karl Lanocha, M.D Richard Bermudes, M.D. Kimberly Cress, M.D. Disclosures Todd Hutton, M.D. Speaker for Neuronetics Board of Directors,
Transcranial Magnetic Stimulation: What You Need To Know Todd Hutton, M.D. Karl Lanocha, M.D Richard Bermudes, M.D. Kimberly Cress, M.D. Disclosures Todd Hutton, M.D. Speaker for Neuronetics Board of Directors,
WHY THE US GOVERNMENT REQUIRES WARNING LABELS ON TOOTHPASTE
 WHY THE US GOVERNMENT REQUIRES WARNING LABELS ON TOOTHPASTE WARNING: Keep out of the reach of children under 6 years of age. If you accidentally swallow more than used for bushing, seek professional assistance
WHY THE US GOVERNMENT REQUIRES WARNING LABELS ON TOOTHPASTE WARNING: Keep out of the reach of children under 6 years of age. If you accidentally swallow more than used for bushing, seek professional assistance
Medical Policy. Description/Scope. Position Statement. Rationale
 Subject: Document#: Current Effective Date: 06/28/2017 Status: Reviewed Last Review Date: 05/04/2017 Description/Scope This document addresses occipital nerve stimulation (ONS), which involves delivering
Subject: Document#: Current Effective Date: 06/28/2017 Status: Reviewed Last Review Date: 05/04/2017 Description/Scope This document addresses occipital nerve stimulation (ONS), which involves delivering
September 28, Division of Dockets Management (HFA-305) Food and Drug Administration 5630 Fishers Lane, Room 1061 Rockville, MD 20852
 September 28, 2011 Division of Dockets Management (HFA-305) Food and Drug Administration 5630 Fishers Lane, Room 1061 Rockville, MD 20852 Re: Docket No. : Center for Devices and Radiological Health 510(k)
September 28, 2011 Division of Dockets Management (HFA-305) Food and Drug Administration 5630 Fishers Lane, Room 1061 Rockville, MD 20852 Re: Docket No. : Center for Devices and Radiological Health 510(k)
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: vagus_nerve_stimulation 6/1998 5/2017 5/2018 5/2017 Description of Procedure or Service Stimulation of the
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: vagus_nerve_stimulation 6/1998 5/2017 5/2018 5/2017 Description of Procedure or Service Stimulation of the
March 14, The Food and Drug Administration (FDA) is facing a serious crisis that has significant implications for the nation s health.
 March 14, 2007 An Open Letter to: Chairman Edward Kennedy and Senator Mike Enzi, Chairman John Dingell and Representative Joe Barton Members of the Senate Health, Education, Labor and Pensions Committee
March 14, 2007 An Open Letter to: Chairman Edward Kennedy and Senator Mike Enzi, Chairman John Dingell and Representative Joe Barton Members of the Senate Health, Education, Labor and Pensions Committee
The Emperor s New Drugs: Medication and Placebo in the Treatment of Depression
 The Emperor s New Drugs: Medication and Placebo in the Treatment of Depression Irving Kirsch, PhD Associate Director, Program in Placebo Studies Harvard Medical School Professor Emeritus of Psychology
The Emperor s New Drugs: Medication and Placebo in the Treatment of Depression Irving Kirsch, PhD Associate Director, Program in Placebo Studies Harvard Medical School Professor Emeritus of Psychology
Conference Call for Investment Community. Nov 19, 2018
 Conference Call for Investment Community Nov 19, 2018 Agenda for Today s Call Topic Safe Harbor Statement Opening Remarks AR101 for Peanut Allergy Speaker Laura Hansen, PhD, VP, Investor Relations Jayson
Conference Call for Investment Community Nov 19, 2018 Agenda for Today s Call Topic Safe Harbor Statement Opening Remarks AR101 for Peanut Allergy Speaker Laura Hansen, PhD, VP, Investor Relations Jayson
QiG INS Conference May 22, 2011
 Strategy & Vision Growth Electrochem Medical Device Evolution QiG Cardio vascular QiG INS Conference May 22, 2011 Greatbatch Medical Neuro modulation 1 GREATBATCH TODAY 2010 Revenue by Product Line NYSE:
Strategy & Vision Growth Electrochem Medical Device Evolution QiG Cardio vascular QiG INS Conference May 22, 2011 Greatbatch Medical Neuro modulation 1 GREATBATCH TODAY 2010 Revenue by Product Line NYSE:
Paediatric headaches. Dr Jaycen Cruickshank Director of Clinical Training Ballarat Health Services. Brevity, levity, repetition
 Paediatric headaches Dr Jaycen Cruickshank Director of Clinical Training Ballarat Health Services Brevity, levity, repetition Paediatric)headache?)! Headache!in!children!is!not!that!common.!The!question!is!which!headaches!do!I!
Paediatric headaches Dr Jaycen Cruickshank Director of Clinical Training Ballarat Health Services Brevity, levity, repetition Paediatric)headache?)! Headache!in!children!is!not!that!common.!The!question!is!which!headaches!do!I!
BRAIN STIMULATION AN ALTERNATIVE TO DRUG THERAPY IN MATERNAL DEPRESSION?
 BRAIN STIMULATION AN ALTERNATIVE TO DRUG THERAPY IN MATERNAL DEPRESSION? Kira Stein, MD Medical Director West Coast Life Center Sherman Oaks, California CA Maternal Mental Health Initiative - 2013 2013
BRAIN STIMULATION AN ALTERNATIVE TO DRUG THERAPY IN MATERNAL DEPRESSION? Kira Stein, MD Medical Director West Coast Life Center Sherman Oaks, California CA Maternal Mental Health Initiative - 2013 2013
Medical Policy An Independent Licensee of the Blue Cross and Blue Shield Association
 of Depression and Other Psychiatric Disorders Page 1 of 26 Medical Policy An Independent Licensee of the Blue Cross and Blue Shield Association Title: See also: Transcranial Magnetic Stimulation (TMS)
of Depression and Other Psychiatric Disorders Page 1 of 26 Medical Policy An Independent Licensee of the Blue Cross and Blue Shield Association Title: See also: Transcranial Magnetic Stimulation (TMS)
Executive Summary. Overall conclusions of this report include:
 Executive Summary On November 23, 1998, 46 states settled their lawsuits against the nation s major tobacco companies to recover tobacco-related health care costs, joining four states Mississippi, Texas,
Executive Summary On November 23, 1998, 46 states settled their lawsuits against the nation s major tobacco companies to recover tobacco-related health care costs, joining four states Mississippi, Texas,
Toradol and vicodin. Toradol and vicodin
 Toradol and vicodin The Borg System is 100 % Toradol and vicodin Jan 11, 2017. An AFC lineman says he believes the use of painkillers such as Vicodin or OxyContin is now much more widespread throughout
Toradol and vicodin The Borg System is 100 % Toradol and vicodin Jan 11, 2017. An AFC lineman says he believes the use of painkillers such as Vicodin or OxyContin is now much more widespread throughout
The Coming Gamification of Fitness
 The Coming Gamification of Fitness Vikram Biyani (NetApp) Gregory Corrado (Google) Stacie Hibino (Samsung) Damian Tompkin (Yahoo) Chris Wayne (Yahoo) Fitness Crisis Americans are overweight and paying
The Coming Gamification of Fitness Vikram Biyani (NetApp) Gregory Corrado (Google) Stacie Hibino (Samsung) Damian Tompkin (Yahoo) Chris Wayne (Yahoo) Fitness Crisis Americans are overweight and paying
2/17/2018. Prof. Steven S. Saliterman Department of Biomedical Engineering, University of Minnesota
 Department of Biomedical Engineering, University of Minnesota http://saliterman.umn.edu/ FDA Authority Medical Device Act of 1976 FDA Modernization Act of 1997 Federal Food, Drug and Cosmetic Act (FFDCA)
Department of Biomedical Engineering, University of Minnesota http://saliterman.umn.edu/ FDA Authority Medical Device Act of 1976 FDA Modernization Act of 1997 Federal Food, Drug and Cosmetic Act (FFDCA)
Prof. Steven S. Saliterman. Department of Biomedical Engineering, University of Minnesota
 Department of Biomedical Engineering, University of Minnesota http://saliterman.umn.edu/ FDA Authority Medical Device Act of 1976 FDA Modernization Act of 1997 Federal Food, Drug and Cosmetic Act (FFDCA)
Department of Biomedical Engineering, University of Minnesota http://saliterman.umn.edu/ FDA Authority Medical Device Act of 1976 FDA Modernization Act of 1997 Federal Food, Drug and Cosmetic Act (FFDCA)
Mainstay Medical Announces Headline Results from ReActiv8-B Clinical Study
 Mainstay Medical Announces Headline Results from ReActiv8-B Clinical Study Responder rates at 120 days for treatment and active control groups were 56% vs. 47%; statistically significant difference on
Mainstay Medical Announces Headline Results from ReActiv8-B Clinical Study Responder rates at 120 days for treatment and active control groups were 56% vs. 47%; statistically significant difference on
Corporate Medical Policy
 Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: facet_joint_denervation 6/2009 4/2017 4/2018 4/2017 Description of Procedure or Service Facet joint denervation
Corporate Medical Policy File Name: Origination: Last CAP Review: Next CAP Review: Last Review: facet_joint_denervation 6/2009 4/2017 4/2018 4/2017 Description of Procedure or Service Facet joint denervation
Oscar G. Morales. MD Founding Director McLean Hospital TMS
 Institute of Medicine of the National Academies Non-Invasive Neuromodulation of the Central Nervous System: A Workshop Washington, DC. March 2 and 3, 2015 Session IV: Reimbursement Oscar G. Morales. MD
Institute of Medicine of the National Academies Non-Invasive Neuromodulation of the Central Nervous System: A Workshop Washington, DC. March 2 and 3, 2015 Session IV: Reimbursement Oscar G. Morales. MD
Benign Headache and Diagnosis Pathophysiology. Dr Andrew J Dowson Director of the King s Headache Service King s College Hospital London
 Benign Headache and Diagnosis Pathophysiology Dr Andrew J Dowson Director of the King s Headache Service King s College Hospital London Case histories Case 1 Male, 46 y - Truck driver - Headaches since
Benign Headache and Diagnosis Pathophysiology Dr Andrew J Dowson Director of the King s Headache Service King s College Hospital London Case histories Case 1 Male, 46 y - Truck driver - Headaches since
Another name for tylenol 300 with codeine 30
 Cari untuk: Cari Cari Another name for tylenol 300 with codeine 30 1-2-2018 Physician reviewed Tylenol with Codeine #3 patient information - includes Tylenol with Codeine #3 description, dosage and directions.
Cari untuk: Cari Cari Another name for tylenol 300 with codeine 30 1-2-2018 Physician reviewed Tylenol with Codeine #3 patient information - includes Tylenol with Codeine #3 description, dosage and directions.
BRAINSWAY DEEP TMS SYSTEM
 510(K) SUMMARY JAN 0 72013 BRAINSWAY DEEP TMS SYSTEM Applicant Name: Company Name: Address: 510(k) Number K122288 Brainsway Ltd. 19 Hartumn St. (Bynet Bldg) Har Hotzvim, Jerusalem, ISRAEL 91451 Tel: +972-2-5813
510(K) SUMMARY JAN 0 72013 BRAINSWAY DEEP TMS SYSTEM Applicant Name: Company Name: Address: 510(k) Number K122288 Brainsway Ltd. 19 Hartumn St. (Bynet Bldg) Har Hotzvim, Jerusalem, ISRAEL 91451 Tel: +972-2-5813
