CLINICAL GUIDELINES ID TAG
|
|
|
- Claude Armstrong
- 5 years ago
- Views:
Transcription
1 Title: Author: Speciality/ Division: Directorate: Date Uploaded: Review Date: September 2019 CG ID TAG CG0423 CLINICAL GUIDELINES ID TAG Clinical Guideline for Alteplase in intra-arterial thrombolysis for treating acute peripheral arterial occlusion Alaistair Lewis, Richard McConville, Joan Morton, Jayne Agnew Radiology/ Surgery Acute Version 1.0 September 2016 Page 1
2 Clinical Guideline for Alteplase in intra- arterial thrombolysis for treating acute peripheral arterial occlusion Thrombolytic therapy is used in the management of acute arterial ischaemia. The principle objective of thrombolytic therapy is to remove pathological thromboemboli and to facilitate restoration of vascular patency. Thrombolytic therapy is contraindicated in any patient with a high risk of haemorrhage. The Working Party on Thrombolysis in the Management of Limb Ischemia divided contraindications into absolute, major and minor (see table 1) Table 1 Contraindications to intra-arterial thrombolysis Absolute 1. Established cerebrovascular event (including TIAs within last 2 months) 2. Active bleeding diathesis 3. Recent gastrointestinal bleeding (<10 days) 4. Neurosurgery (intracranial, spinal) within last 3 months 5. Intracranial trauma within last 3 months Relative major 1. Cardiopulmonary resuscitation within last 10 days 2. Major nonvascular surgery or trauma within last 10 days 3. Uncontrolled hypertension >180 mm Hg systolic or >110 mm Hg DIASTOLIC 4. Puncture of noncompressible vessel 5. Intracranial tumour 6. Recent eye surgery Minor 1. Hepatic failure particularly those with coagulopathy 2. Bacterial endocarditis 3. Pregnancy 4. Diabetic haemorrhagic retinopathy The following conditions are also listed as contraindications to administration of alteplase by the manufacturer and should be evaluated on a case-by-case basis significant bleeding disorder at present or within the past 6 months known haemorrhagic diathesis patients receiving effective oral anticoagulant treatment, e.g. warfarin, apixaban manifest or recent severe or dangerous bleeding known history of or suspected intracranial haemorrhage suspected subarachnoid haemorrhage or condition after subarachnoid haemorrhage from aneurysm any history of central nervous system damage (i.e. neoplasm, aneurysm, intracranial or spinal surgery) recent (less than 10 days) traumatic external heart massage, obstetrical delivery, recent puncture of a noncompressible blood-vessel (e.g. subclavian or jugular vein puncture) severe uncontrolled arterial hypertension Version 1.0 September 2016 Page 2
3 bacterial endocarditis, pericarditis acute pancreatitis documented ulcerative gastrointestinal disease during the last 3 months, oesophageal varices, arterial-aneurysm, arterial/venous malformations neoplasm with increased bleeding risk severe liver disease, including hepatic failure, cirrhosis, portal hypertension (oesophageal varices) and active hepatitis major surgery or significant trauma in past 3 months any known history of haemorrhagic stroke or stroke of unknown origin known history of ischaemic stroke or transient ischaemic attack (TIA) in the preceding 6 months platelet count of below 100,000/mm 3 This is an unlicensed use of alteplase. The patient s coagulation screen must be checked prior to the procedure. The PT should be within 9-13 seconds. If the result is outside this range the procedure is contraindicated. Following a diagnostic angiogram a sheath is inserted into the affected artery and a catheter inserted through the sheath with its tip positioned in close proximity to the thrombus, ideally within the thrombus if possible. This allows direct delivery of alteplase to the thrombus. A local hypercoagulable state may be present in the vessel undergoing thrombolysis. It is standard practice to concomitantly administer a low dose of unfractionated heparin to prevent immediate rethrombosis of the treated vessel and to prevent pericatheter thrombosis. The unfractionated heparin is infused via the sheath side arm with the aim of producing only mild prolongation of the APTT (NOT full anticoagulation). The radiologist/ surgeon must prescribe the intra-arterial alteplase and intra-arterial unfractionated heparin on the Trust Intra-arterial thrombolysis prescription chart. Radiology staff will initiate the alteplase and unfractionated heparin infusions and commence the intra-arterial thrombolysis monitoring sheet. Alteplase is administered via the intra-arterial catheter and unfractionated heparin via the sheath sidearm. Intra-arterial thrombolysis regime 1. Reconstitute the contents of a 20mg vial of alteplase with 20ml of water for injection to give a solution with a final concentration of 1mg/ml. This should be gently mixed and vigorous agitation or shaking should be avoided to prevent foam formation. 2. A loading dose of 2ml (2mg) of the reconstituted solution is administered by slow bolus injection into the arterial catheter by the interventional radiologist when the catheter is in position ml (10mg) of the reconstituted solution should be further diluted. Withdraw 10ml from a 50ml infusion bag of sodium chloride 0.9% and then add 10ml of the reconstituted alteplase. This gives a final concentration of 10mg in 50ml (1mg in 5ml). The infusion bag must be labelled and signed by two qualified practitioners. This solution has an expiry date of 8 hours at room temperature. (The remaining reconstituted alteplase solution should be discarded) Version 1.0 September 2016 Page 3
4 4. This solution is infused initially at 10ml per hour (2mg /hr) for two hours. Then it is infused as a maintenance infusion via the intra-arterial catheter at a rate of 5ml per hour (1mg/hr) thereafter. 5. For subsequent alteplase infusions reconstitute the contents of a 20mg vial of alteplase with 20ml of water for injection. The reconstituted solution is stable for 24 hours at 2-8 o C. Further dilute 10ml of the reconstituted solution. Remove 10ml from a 50ml infusion bag of sodium chloride 0.9% and then add 10ml of the reconstituted alteplase solution. This gives a final concentration of 10mg in 50ml (1mg in 5ml). This solution has an expiry date of 8 hours at room temperature. This solution should be infused at 5ml per hour (1mg/hr) until further radiology review (up to 24 hours). 6. Label the intra-arterial line 7. An intra-arterial thrombolysis monitoring sheet will be initiated in radiology and transferred back to the ward with the patient where ward nursing staff will continue to record the infusion rate and volume. Radiology staff will mark the position of the catheter in relation to the sheath to detect if line displacement occurs. If the position of the catheter moves more than 1cm, ward staff must contact the patient s consultant or radiologist immediately Unfractionated heparin 1. Unfractionated heparin is administered with alteplase to prevent re-occlusion after thrombolysis and should be given via the sheath side arm to reduce the risk of pericatheter thrombosis. 2. Draw up 25mls of 1,000 units per ml unfractionated heparin solution (25,000units). The syringe must be labelled and signed by two qualified practitioners. 3. Infuse the solution via the sheath side arm at a rate of 1mls per hour (1000 units/ hr). This infusion rate is NOT to be adjusted. There is no requirement for APTT monitoring with this regime. 4. An intra-arterial thrombolysis monitoring sheet will be initiated in radiology and transferred back to the ward with the patient where nursing staff must continue to record the infusion rate and volume. Duration of therapy The patient will return to radiology and undergo subsequent angiography within the next 24 hours in order to determine the effectiveness of thrombolysis. The decision to discontinue thrombolysis will be made by the radiologist based on angiography results. Nursing Observations and Patient Care Due to the increased risk of haemorrhage and shock during intra-arterial thrombolysis the patient should be strictly confined to bed rest and kept lying as flat as possible. Version 1.0 September 2016 Page 4
5 Intra-muscular injections should be avoided. Patient observations (blood pressure, heart rate, temperature, respiratory rate) should be recorded every 30 minutes for the first 2 hours and then every hour until thrombolysis is discontinued. A coagulation screen should be checked at 4 hours after commencing therapy, the purpose of which is to ensure hypersensitivity to heparin is not present. If the APPT is less than 45 seconds, the heparin infusion may continue without change. If the APPT is greater than 45 seconds, contact the named consultant directly for advice. The patient should be closely monitored for any signs of bleeding particularly at the arterial catheter site. Persistently low blood pressure and/or tachycardia may indicate haemorrhage and necessitates pausing the alteplase and unfractionated heparin infusions as soon as possible until this has been resolved. The catheter site should be monitored on an hourly basis and observations recorded on the intra-arterial thrombolysis monitoring sheet. Special consideration should be given to any bruising or hard swelling around the site which should be treated as haematoma by applying firm pressure and seeking medical advice. Once intra-arterial thrombolysis has been stopped the arterial catheter should be removed by radiology. The sheath should be removed by radiology following the angiogram. A sterile transparent dressing should then be placed over the site to allow observation and monitoring for haemorrhage and haematoma. These observations should continue every half hour to hourly for at least 4 hours and then 2 hourly after that and recorded on the thrombolysis monitoring sheet. References 1. Actilyse. Summary of Product Characteristics. Boehringer Ingelheim. Date of text revision 23 rd September Available at Accessed 26 th October Actilyse monograph Medusa Injectable Medicines Guide. Available at Accessed 26th October Working Party on Thrombolysis in the Management of Limb Ischemia. Thrombolysis in the Management of Lower Limb Peripheral Arterial Occlusion A Consensus Document. Journal of Vascular and Interventional Radiology 2003;7:S337-S349. Version 1.0 September 2016 Page 5
6 Intra-arterial thrombolysis prescription sheet Allergies/ Medicine sensitivities This section must be completed before prescribing and administration except in exceptional circumstances Medicine/allergen Date of Type of e.g. rash Signature/ designation/ date Write in CAPITAL LETTERS or use addressograph Patient s name: Health and Care no. Date of birth: or No known allergies (please tick) Signature/ Designation: Date: Hospital: Ward: Consultant: Initial prescription (radiology department use) Date Drug Route Rate Radiologist/surgeon Alteplase 2mg loading Via arterial Bolus dose Alteplase 10mg made up to 50ml with sodium chloride 0.9% (10mg in 50ml) Unfractionated heparin 25ml 1000units/ml catheter Via arterial catheter Via sheath side arm 10ml per hour for 2 hours 1ml per hour (1000 units/hr) Given by Witness Ongoing prescription (until further radiology review) Drug Alteplase 10mg Start date Rate Route 5ml per hour Arterial catheter Dilution Make up to 50ml with sodium chloride 0.9% Radiologist/ surgeon Date, time,, witness Date, time,, witness Date, time,, witness Pharmacy Drug Unfractionated heparin 25ml 1000units/ml Start date Pharmacy Additional comments Solution expires 8hrs after reconstitution Rate Route 1ml per hour Sheath side arm Dilution Radiologist/ surgeon Additional comments No requirement to monitor APTT Version 1.0 September 2016 Page 6
7 Allergies/ Medicine sensitivities This section must be completed before prescribing and administration except in exceptional circumstances Medicine/allergen Date of Intra-arterial thrombolysis monitoring sheet Type of e.g. rash Signature/ designation/ date Write in CAPITAL LETTERS or use addressograph Patient s name: Health and Care no. Date of birth: or No known allergies (please tick) Signature/ Designation: Date: Hospital: Ward: Consultant: Date of thrombolysis: Line labelled: Yes / No Date/ Time Alteplase Infusion rate Alteplase Infusion volume Heparin infusion rate Heparin infusion volume Line insertion site OK Position of catheter to sheath on insertion: Yes/No Line secure Limb check Pulse present Signature Print name Post removal check Date/ Time Insertion site checked Bleeding Signature Print name Version 1.0 September 2016 Page 7
BEAUMONT HOSPITAL. Beaumont Hospital Department of Nephrology and Renal Nursing ADMINISTRATION OF ACTILYSE. Guideline Number: 13. Guideline Version: B
 BEAUMONT HOSPITAL Beaumont Hospital Department of Nephrology and Renal Nursing Guideline Name: GUIDELINES FOR THE ADMINISTRATION OF ACTILYSE Guideline Number: 13 Guideline Version: B Developed By: Betty
BEAUMONT HOSPITAL Beaumont Hospital Department of Nephrology and Renal Nursing Guideline Name: GUIDELINES FOR THE ADMINISTRATION OF ACTILYSE Guideline Number: 13 Guideline Version: B Developed By: Betty
NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOLS. ACTIVASE (t-pa) INFUSION PROTOCOL FOR ACUTE MYOCARDIAL INFARCTION
 NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOLS ACTIVASE (t-pa) FOR ACUTE MYOCARDIAL INFARCTION I. PURPOSE: A. To reduce the extent of myocardial infarction by lysing the clot in
NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOLS ACTIVASE (t-pa) FOR ACUTE MYOCARDIAL INFARCTION I. PURPOSE: A. To reduce the extent of myocardial infarction by lysing the clot in
Thrombolysis administration
 Thrombolysis administration Liz Mackey Stroke Nurse Practitioner Western Health Sunshine & Footscray Hospital, Melbourne Thanks ASNEN committee members Skye Coote, Acute Stroke Nurse, Eastern Health (slide
Thrombolysis administration Liz Mackey Stroke Nurse Practitioner Western Health Sunshine & Footscray Hospital, Melbourne Thanks ASNEN committee members Skye Coote, Acute Stroke Nurse, Eastern Health (slide
Thrombolysis for acute ischaemic stroke Rapid Assessment Protocol NORTHERN IRELAND Regional Protocol (Version 002 July 08)
 Thrombolysis for acute ischaemic stroke Rapid Assessment Protocol NORTHERN IRELAND Regional Protocol (Version 002 July 08) Patient Details Time of onset? Capillary Blood glucose 2.8-22.2 mmol/l? Blood
Thrombolysis for acute ischaemic stroke Rapid Assessment Protocol NORTHERN IRELAND Regional Protocol (Version 002 July 08) Patient Details Time of onset? Capillary Blood glucose 2.8-22.2 mmol/l? Blood
NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOLS. ACUTE CEREBROVASCULAR ACCIDENT TPA (ACTIVASE /alteplase) FOR THROMBOLYSIS
 NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOLS ACUTE CEREBROVASCULAR ACCIDENT TPA (ACTIVASE /alteplase) FOR THROMBOLYSIS I. Purpose : A. To reduce morbidity and mortality associated
NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOLS ACUTE CEREBROVASCULAR ACCIDENT TPA (ACTIVASE /alteplase) FOR THROMBOLYSIS I. Purpose : A. To reduce morbidity and mortality associated
Thrombolysis Assessment
 Thrombolysis Assessment Brief Clinical Summary of symptom onset of arrival of patient of assessment BP GCS BM If BM
Thrombolysis Assessment Brief Clinical Summary of symptom onset of arrival of patient of assessment BP GCS BM If BM
GUIDELINES FOR THE EARLY MANAGEMENT OF PATIENTS WITH ACUTE ISCHEMIC STROKE
 2018 UPDATE QUICK SHEET 2018 American Heart Association GUIDELINES FOR THE EARLY MANAGEMENT OF PATIENTS WITH ACUTE ISCHEMIC STROKE A Summary for Healthcare Professionals from the American Heart Association/American
2018 UPDATE QUICK SHEET 2018 American Heart Association GUIDELINES FOR THE EARLY MANAGEMENT OF PATIENTS WITH ACUTE ISCHEMIC STROKE A Summary for Healthcare Professionals from the American Heart Association/American
How to give thrombolysis in acute myocardial infarction
 Page 1 of 6 How to give thrombolysis in acute myocardial infarction Original article: Michael Tam In the major urban hospitals, there will be little place for thrombolysis in acute STEMI (STelevation myocardial
Page 1 of 6 How to give thrombolysis in acute myocardial infarction Original article: Michael Tam In the major urban hospitals, there will be little place for thrombolysis in acute STEMI (STelevation myocardial
METALYSE Tenecteplase
 METALYSE Tenecteplase Consumer Medicine Information What is in this leaflet This leaflet answers some common questions about Metalyse. It does not contain all the available information. It does not take
METALYSE Tenecteplase Consumer Medicine Information What is in this leaflet This leaflet answers some common questions about Metalyse. It does not contain all the available information. It does not take
PACKAGE LEAFLET: INFORMATION FOR THE USER. Actilyse Cathflo 2 mg, poeder voor oplossing voor injectie en infusie. Alteplase
 PACKAGE LEAFLET 1 PACKAGE LEAFLET: INFORMATION FOR THE USER Actilyse Cathflo 2 mg, poeder voor oplossing voor injectie en infusie Alteplase Read all of this leaflet carefully before you start using this
PACKAGE LEAFLET 1 PACKAGE LEAFLET: INFORMATION FOR THE USER Actilyse Cathflo 2 mg, poeder voor oplossing voor injectie en infusie Alteplase Read all of this leaflet carefully before you start using this
Acute Stroke Protocols Modified- What s New in 2013
 Acute Stroke Protocols Modified- What s New in 2013 KUMAR RAJAMANI, MD, DM. Vascular Neurologist-MSN Associate Professor of Neurology WSU School of Medicine. Saturday, September 21, 2013 Crystal Mountain
Acute Stroke Protocols Modified- What s New in 2013 KUMAR RAJAMANI, MD, DM. Vascular Neurologist-MSN Associate Professor of Neurology WSU School of Medicine. Saturday, September 21, 2013 Crystal Mountain
9/24/2013. Thrombolytics in 2013: Never Say Never. September 19 th, 2013 Scott M Lilly, MD PhD. Clinical Case
 September 19 th, 2013 Scott M Lilly, MD PhD Thrombolytics in 2013: Never Say Never Clinical Case 2 1 Evolution of STEMI Therapy The importance of absolute rest in bed for several days is clear James B
September 19 th, 2013 Scott M Lilly, MD PhD Thrombolytics in 2013: Never Say Never Clinical Case 2 1 Evolution of STEMI Therapy The importance of absolute rest in bed for several days is clear James B
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Tenecteplase Boehringer Ingelheim Pharma KG 6,000 units Powder and solvent for solution for injection 2. QUALITATIVE AND QUANTITATIVE
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Tenecteplase Boehringer Ingelheim Pharma KG 6,000 units Powder and solvent for solution for injection 2. QUALITATIVE AND QUANTITATIVE
PE Pathway. The charts are listed as follows:
 PE Pathway This document comprises 6 simple flow charts to assist clinicians in the investigation and treatment of suspected or confirmed Acute Pulmonary Emboli. The pathway has been put together using
PE Pathway This document comprises 6 simple flow charts to assist clinicians in the investigation and treatment of suspected or confirmed Acute Pulmonary Emboli. The pathway has been put together using
ACCESS CENTER:
 ACCESS CENTER: 1-877-367-8855 Emergency Specialty Services: BRAIN ATTACK Criteria: Stroke symptom onset time less than 6 hours Referring Emergency Department Patient Information Data: Time last known normal:
ACCESS CENTER: 1-877-367-8855 Emergency Specialty Services: BRAIN ATTACK Criteria: Stroke symptom onset time less than 6 hours Referring Emergency Department Patient Information Data: Time last known normal:
ST Elevation Myocardial Infarction (STEMI) Reperfusion Order Set
 Form Title Form Number CH-0454 2018, Alberta Health Services, CKCM This work is licensed under the Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License. The license does not
Form Title Form Number CH-0454 2018, Alberta Health Services, CKCM This work is licensed under the Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License. The license does not
Primary Stroke Center Quality & Performance Measures
 Primary Stroke Center Quality & Performance Measures This section of the manual contains information related to the quality performance of Primary Stroke Centers. Brain Attack Coalition Definitions Recognition
Primary Stroke Center Quality & Performance Measures This section of the manual contains information related to the quality performance of Primary Stroke Centers. Brain Attack Coalition Definitions Recognition
Shared Care Protocol for the Prescription and Supply of Low Molecular Weight Heparins
 Tameside Hospital NHS Foundation Trust and NHS Tameside and Glossop Shared Care Protocol for the Prescription and Supply of Low Molecular Weight Heparins Version 5.2 Version: 5.2 Authorised by: Joint Medicines
Tameside Hospital NHS Foundation Trust and NHS Tameside and Glossop Shared Care Protocol for the Prescription and Supply of Low Molecular Weight Heparins Version 5.2 Version: 5.2 Authorised by: Joint Medicines
AGWS Stroke Thrombolysis Clinical Profoma
 AGWS Stroke Thrombolysis Clinical Profoma Incorporating Salisbury NHS Foundation Trust guidance Date: On Arrival: Affix patient label here) GCS NIHSS Score: Pulse SaO on Air Give O only if < 95 % on Air
AGWS Stroke Thrombolysis Clinical Profoma Incorporating Salisbury NHS Foundation Trust guidance Date: On Arrival: Affix patient label here) GCS NIHSS Score: Pulse SaO on Air Give O only if < 95 % on Air
P-RMS: LT/H/PSUR/0004/001
 Core Safety Profile Active substance: Dalteparine Pharmaceutical form(s)/strength: Solution for injection, 2500 I.U./0.2ml, 2500 I.U./ml, 5000 I.U./0.2ml, 7500 I.U./0.3ml, 7500 I.U./0.75ml, 10000 I.U./0.4ml,
Core Safety Profile Active substance: Dalteparine Pharmaceutical form(s)/strength: Solution for injection, 2500 I.U./0.2ml, 2500 I.U./ml, 5000 I.U./0.2ml, 7500 I.U./0.3ml, 7500 I.U./0.75ml, 10000 I.U./0.4ml,
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Actilyse Powder and solvent for solution for injection and infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 vial with powder
SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Actilyse Powder and solvent for solution for injection and infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION 1 vial with powder
Cardiac Catheter Labs Intravenous Drug Therapy Guide
 Title of Guideline (must include the word Guideline (not protocol, policy, procedure etc) Contact Name and Job Title (author) Cardiac Catheter Lab IV Medicines Guideline Helen Buxton ( Senior Cath Lab
Title of Guideline (must include the word Guideline (not protocol, policy, procedure etc) Contact Name and Job Title (author) Cardiac Catheter Lab IV Medicines Guideline Helen Buxton ( Senior Cath Lab
Southern Trust Home IV Service. Guidelines for the administration of IV antibiotics
 Southern Trust Home IV Service Guidelines for the administration of IV antibiotics Title: Author: CLINICAL GUIDELINES ID TAG Antibiotic guidelines - Southern Trust Home IV service guidelines for the administration
Southern Trust Home IV Service Guidelines for the administration of IV antibiotics Title: Author: CLINICAL GUIDELINES ID TAG Antibiotic guidelines - Southern Trust Home IV service guidelines for the administration
NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOLS EPTIFIBATIDE (INTEGRILIN) PROTOCOL
 NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE S EPTIFIBATIDE (INTEGRILIN) I. PURPOSE: A. Integrilin (Eptifibatide) is a specific and potent inhibitor of the platelet receptor glycoprotein
NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE S EPTIFIBATIDE (INTEGRILIN) I. PURPOSE: A. Integrilin (Eptifibatide) is a specific and potent inhibitor of the platelet receptor glycoprotein
MANAGEMENT OF BLEEDING AND EXCESSIVE ANTICOAGULATION IN ADULTS RECEIVING ANTI-COAGULANTS
 Hairmyres Hospital MANAGEMENT OF BLEEDING AND EXCESSIVE ANTICOAGULATION IN ADULTS RECEIVING ANTI-COAGULANTS Bleeding in patients on anticoagulants, even in the absence of over-anticoagulation, can be life-threatening
Hairmyres Hospital MANAGEMENT OF BLEEDING AND EXCESSIVE ANTICOAGULATION IN ADULTS RECEIVING ANTI-COAGULANTS Bleeding in patients on anticoagulants, even in the absence of over-anticoagulation, can be life-threatening
ANNEX III LABELLING AND PACKAGE LEAFLET
 ANNEX III LABELLING AND PACKAGE LEAFLET 1 B. PACKAGE LEAFLET 2 PACKAGE LEAFLET Read all of this leaflet carefully before you start taking this medicine. - Keep this leaflet. You may need to read it again.
ANNEX III LABELLING AND PACKAGE LEAFLET 1 B. PACKAGE LEAFLET 2 PACKAGE LEAFLET Read all of this leaflet carefully before you start taking this medicine. - Keep this leaflet. You may need to read it again.
Ultrasound-enhanced, catheter-directed thrombolysis for pulmonary embolism
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Ultrasound-enhanced, catheter-directed thrombolysis for pulmonary embolism A pulmonary embolism (PE) is
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Interventional procedure consultation document Ultrasound-enhanced, catheter-directed thrombolysis for pulmonary embolism A pulmonary embolism (PE) is
Neonatal Parenteral Nutrition Guideline Dr M Hogan, Maire Cullen ANNP, Una Toland Ward Manager, Sandra Kilpatrick Neonatal Pharmacist
 CLINICAL GUIDELINES ID TAG Title: Author: Designation: Speciality / Division: Directorate: Neonatal Parenteral Nutrition Guideline Dr M Hogan, Maire Cullen ANNP, Una Toland Ward Manager, Sandra Kilpatrick
CLINICAL GUIDELINES ID TAG Title: Author: Designation: Speciality / Division: Directorate: Neonatal Parenteral Nutrition Guideline Dr M Hogan, Maire Cullen ANNP, Una Toland Ward Manager, Sandra Kilpatrick
DRUG GUIDELINE. HYDRALAZINE (Intravenous severe hypertension in pregnancy)
 DRUG GUIDELINE HYDRALAZINE (Intravenous severe hypertension SCOPE (Area): FOR USE IN: Labour Ward, HDU, Theatre and ED EXCLUSIONS: Paediatrics (seek Paediatrician advice) and other general wards. SCOPE
DRUG GUIDELINE HYDRALAZINE (Intravenous severe hypertension SCOPE (Area): FOR USE IN: Labour Ward, HDU, Theatre and ED EXCLUSIONS: Paediatrics (seek Paediatrician advice) and other general wards. SCOPE
Thrombolysis Delivery, Care, and Monitoring. 5 Acute Trusts - 6 Primary Care Trusts Ambulance Trust 4 Local Authorities
 Thrombolysis Delivery, Care, and Monitoring Documentation & Pathways Need to follow locally agreed policies and procedures Follow thrombolysis pathway? Need to complete Sits database Weight Dose matters!
Thrombolysis Delivery, Care, and Monitoring Documentation & Pathways Need to follow locally agreed policies and procedures Follow thrombolysis pathway? Need to complete Sits database Weight Dose matters!
Appendix IV - Prescribing Guidance for Apixaban
 Appendix IV - Prescribing Guidance for Apixaban Patient Factors Dose of Apixaban If your patient has any of the following MAJOR risk factors: Hypersensitivity to the active substance or to any of the excipients
Appendix IV - Prescribing Guidance for Apixaban Patient Factors Dose of Apixaban If your patient has any of the following MAJOR risk factors: Hypersensitivity to the active substance or to any of the excipients
RETAVASE (reteplase) for injection, for intravenous use Initial U.S. Approval: 1996
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use RETAVASE safely and effectively. See full prescribing information for RETAVASE. RETAVASE (reteplase)
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use RETAVASE safely and effectively. See full prescribing information for RETAVASE. RETAVASE (reteplase)
Suspected Deep Vein Thrombosis (DVT) Assessment
 CHI no... First name... DOB... /... /... Last name... Sex: c M c F Address...... Telephone... or attach addressograph label here Hospital/Location: c Hairmyres c Monklands c Wishaw Other (specify)... Ward/Base...
CHI no... First name... DOB... /... /... Last name... Sex: c M c F Address...... Telephone... or attach addressograph label here Hospital/Location: c Hairmyres c Monklands c Wishaw Other (specify)... Ward/Base...
NUH Emergency Department. Guideline for Diagnosis & Treatment of PE (Massive and Non-Massive) in Adults only
 NUH Emergency Department Guideline for Diagnosis & Treatment of PE (Massive and n-massive) in Adults only Contents Suspected Pulmonary Embolus in the ED Flow Chart Page 1 Administration of Thrombolysis
NUH Emergency Department Guideline for Diagnosis & Treatment of PE (Massive and n-massive) in Adults only Contents Suspected Pulmonary Embolus in the ED Flow Chart Page 1 Administration of Thrombolysis
Nursing Management Pre /Post Thrombolysis in Stroke
 Craigavon Area Hospital Guidelines for Nursing Management Pre /Post Thrombolysis in Stroke 1. A senior nurse in the stroke unit will be required to monitor the patients condition post Thrombolysis on a
Craigavon Area Hospital Guidelines for Nursing Management Pre /Post Thrombolysis in Stroke 1. A senior nurse in the stroke unit will be required to monitor the patients condition post Thrombolysis on a
Primary Stroke Center Acute Stroke Transfer Guidelines When to Consider a Transfer:
 When to Consider a Transfer: Hemorrhagic Stroke Large volume intracerebral hematoma greater than 5cm on CT Concern for expanding hematoma Rapidly declining mental status, especially requiring intubation
When to Consider a Transfer: Hemorrhagic Stroke Large volume intracerebral hematoma greater than 5cm on CT Concern for expanding hematoma Rapidly declining mental status, especially requiring intubation
Edoxaban For preventing stroke and systemic embolism in people with non-valvular atrial fibrillation (NICE TA 355)
 Rationale for Initiation, Continuation and Discontinuation (RICaD) Edoxaban For preventing stroke and systemic embolism in people with non-valvular atrial fibrillation (NICE TA 355) This document supports
Rationale for Initiation, Continuation and Discontinuation (RICaD) Edoxaban For preventing stroke and systemic embolism in people with non-valvular atrial fibrillation (NICE TA 355) This document supports
Active date July Ratification date: Review date January 2014 Applies to: Staff managing patients on warfarin. Exclusions:
 Guideline Title: Guidelines for the management of warfarin reversal [key words : Beriplex, Octaplex, PCC, vitamin K, anticoagulant, anticoagulation] Authors: Dr Sarah Allford, Consultant Haematologist
Guideline Title: Guidelines for the management of warfarin reversal [key words : Beriplex, Octaplex, PCC, vitamin K, anticoagulant, anticoagulation] Authors: Dr Sarah Allford, Consultant Haematologist
Online Supplementary Data. Country Number of centers Number of patients randomized
 A Randomized, Double-Blind, -Controlled, Phase-2B Study to Evaluate the Safety and Efficacy of Recombinant Human Soluble Thrombomodulin, ART-123, in Patients with Sepsis and Suspected Disseminated Intravascular
A Randomized, Double-Blind, -Controlled, Phase-2B Study to Evaluate the Safety and Efficacy of Recombinant Human Soluble Thrombomodulin, ART-123, in Patients with Sepsis and Suspected Disseminated Intravascular
THE CARE AND MAINTENANCE OF TOTALLY IMPLANTED VENOUS ACCESS DEVICES (PORTS)
 Children s Hospital for Wales Department of Paediatric Respiratory Medicine and Cystic Fibrosis THE CARE AND MAINTENANCE OF TOTALLY IMPLANTED VENOUS ACCESS DEVICES (PORTS) This protocol applies to the
Children s Hospital for Wales Department of Paediatric Respiratory Medicine and Cystic Fibrosis THE CARE AND MAINTENANCE OF TOTALLY IMPLANTED VENOUS ACCESS DEVICES (PORTS) This protocol applies to the
Package leaflet: Information for the user. ReoPro 2 mg/ml solution for injection or infusion. abciximab
 Package leaflet: Information for the user ReoPro 2 mg/ml solution for injection or infusion abciximab Read all of this leaflet carefully before you start using this medicine because it contains important
Package leaflet: Information for the user ReoPro 2 mg/ml solution for injection or infusion abciximab Read all of this leaflet carefully before you start using this medicine because it contains important
Competency Title: Caring for a patient with an arterial line
 Appendix 2 Competency Title: Caring for a patient with an arterial line Aims and objectives Following completion of this competency document the practitioner will be able to: Discuss the indications for
Appendix 2 Competency Title: Caring for a patient with an arterial line Aims and objectives Following completion of this competency document the practitioner will be able to: Discuss the indications for
Management of Acute Myocardial Infarction
 Management of Acute Myocardial Infarction Prof. Hossam Kandil Professor of Cardiology Cairo University ST Elevation Acute Myocardial Infarction Aims Of Management Emergency care (Pre-hospital) Early care
Management of Acute Myocardial Infarction Prof. Hossam Kandil Professor of Cardiology Cairo University ST Elevation Acute Myocardial Infarction Aims Of Management Emergency care (Pre-hospital) Early care
Inhixa (Enoxaparin Sodium)
 Inhixa (Enoxaparin Sodium) P R E V ENTIS SAFETY D E V I C E P R E V E N T I S I S A N AU TO M AT I C N E E D L E S H I E L D I N G S Y S T E M, W H I C H H A S A C O V E R T H AT E X T E N D S O V E R
Inhixa (Enoxaparin Sodium) P R E V ENTIS SAFETY D E V I C E P R E V E N T I S I S A N AU TO M AT I C N E E D L E S H I E L D I N G S Y S T E M, W H I C H H A S A C O V E R T H AT E X T E N D S O V E R
IR Central Venous Access [ ] Pre Procedure
![IR Central Venous Access [ ] Pre Procedure IR Central Venous Access [ ] Pre Procedure](/thumbs/90/104119758.jpg) IR Central Venous Access [1050200001] Pre Procedure Case Request/Scheduling Procedure Enter IR Case Request if not already completed (All hospitals except Grant Medical Center) [ ] Case Request IR Lab
IR Central Venous Access [1050200001] Pre Procedure Case Request/Scheduling Procedure Enter IR Case Request if not already completed (All hospitals except Grant Medical Center) [ ] Case Request IR Lab
INTRAVENOUS HYDRALAZINE POLICY
 PURPOSE INTRAVENOUS HYDRALAZINE POLICY The purpose of this policy is to: provide safe and effective care for women establish a local approach to care that is evidence based and consistent inform good decision
PURPOSE INTRAVENOUS HYDRALAZINE POLICY The purpose of this policy is to: provide safe and effective care for women establish a local approach to care that is evidence based and consistent inform good decision
DVT - initial management NSCCG
 Background information Information resources for patients and carers Updates to this care map Synonyms Below knee DVT and bleeding risks Patient with confirmed DVT Scan confirms superficial thrombophlebitis
Background information Information resources for patients and carers Updates to this care map Synonyms Below knee DVT and bleeding risks Patient with confirmed DVT Scan confirms superficial thrombophlebitis
Emergency Department Management of Acute Ischemic Stroke
 Emergency Department Management of Acute Ischemic Stroke R. Jason Thurman, MD Associate Professor of Emergency Medicine and Neurosurgery Associate Director, Vanderbilt Stroke Center Vanderbilt University,
Emergency Department Management of Acute Ischemic Stroke R. Jason Thurman, MD Associate Professor of Emergency Medicine and Neurosurgery Associate Director, Vanderbilt Stroke Center Vanderbilt University,
Is Thrombolysis Only for a Crisis?
 Is Thrombolysis Only for a Crisis? December 19, 2017 Is Thrombolysis Only for a Crisis? Indications for Thrombolytic Therapy in Patients with Acute Pulmonary Embolism Case Scenario A 28 year old woman
Is Thrombolysis Only for a Crisis? December 19, 2017 Is Thrombolysis Only for a Crisis? Indications for Thrombolytic Therapy in Patients with Acute Pulmonary Embolism Case Scenario A 28 year old woman
Diagnosis and Management of Acute Myocardial Infarction
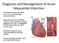 Diagnosis and Management of Acute Myocardial Infarction Acute Myocardial Infarction (AMI) occurs as a result of prolonged myocardial ischemia Atherosclerosis leads to endothelial rupture or erosion that
Diagnosis and Management of Acute Myocardial Infarction Acute Myocardial Infarction (AMI) occurs as a result of prolonged myocardial ischemia Atherosclerosis leads to endothelial rupture or erosion that
Edoxaban Treatment and secondary prevention of deep vein thrombosis and/or pulmonary embolism (NICE TA354)
 Rationale for Initiation, Continuation and Discontinuation (RICaD) Edoxaban Treatment and secondary prevention of deep vein thrombosis and/or pulmonary embolism (NICE TA354) This document supports the
Rationale for Initiation, Continuation and Discontinuation (RICaD) Edoxaban Treatment and secondary prevention of deep vein thrombosis and/or pulmonary embolism (NICE TA354) This document supports the
If viewing a printed copy of this policy, please note it could be expired. Got to to view current policies.
 If viewing a printed copy of this policy, please note it could be expired. Got to www.fairview.org/fhipolicies to view current policies. Department Policy Code: D: PC-5555 Entity: Fairview Pharmacy Services
If viewing a printed copy of this policy, please note it could be expired. Got to www.fairview.org/fhipolicies to view current policies. Department Policy Code: D: PC-5555 Entity: Fairview Pharmacy Services
Home Care Services HomeMed MedEQUIP Michigan Visiting Care Michigan Visiting Nurses Wheelchair Seating Service PROCEDURE
 UNIVERSITY OF MICHIGAN HOSPITALS AND HEALTH CENTERS UMHHC-HCS: 253.054 First Approved Date: 3/2010 Home Care Services HomeMed MedEQUIP Michigan Visiting Care Michigan Visiting Nurses Wheelchair Seating
UNIVERSITY OF MICHIGAN HOSPITALS AND HEALTH CENTERS UMHHC-HCS: 253.054 First Approved Date: 3/2010 Home Care Services HomeMed MedEQUIP Michigan Visiting Care Michigan Visiting Nurses Wheelchair Seating
Dr Ben Turner. Consultant Neurologist and Honorary Senior Lecturer Barts and The London NHS Trust London Bridge Hospital
 Stroke Management Dr Ben Turner Consultant Neurologist and Honorary Senior Lecturer Barts and The London NHS Trust London Bridge Hospital Introduction Stroke is the major cause of disability in the developed
Stroke Management Dr Ben Turner Consultant Neurologist and Honorary Senior Lecturer Barts and The London NHS Trust London Bridge Hospital Introduction Stroke is the major cause of disability in the developed
Alliance A Symptomatic brain radionecrosis after receiving radiosurgery for
 RANDOMIZED PHASE II STUDY: CORTICOSTEROIDS + BEVACIZUMAB VS. CORTICOSTEROIDS + PLACEBO (BEST) FOR RADIONECROSIS AFTER RADIOSURGERY FOR BRAIN METASTASES Pre-registration Eligibility Criteria Required Initial
RANDOMIZED PHASE II STUDY: CORTICOSTEROIDS + BEVACIZUMAB VS. CORTICOSTEROIDS + PLACEBO (BEST) FOR RADIONECROSIS AFTER RADIOSURGERY FOR BRAIN METASTASES Pre-registration Eligibility Criteria Required Initial
CAUTION: You must refer to the intranet for the most recent version of this procedural document.
 Procedure for the use of Intravenous Iron Dextran (CosmoFer ) Sharepoint Location Sharepoint Index Directory Clinical Policies and Guidelines General Policies and Guidelines/ Haematology And blood transfusion
Procedure for the use of Intravenous Iron Dextran (CosmoFer ) Sharepoint Location Sharepoint Index Directory Clinical Policies and Guidelines General Policies and Guidelines/ Haematology And blood transfusion
Stroke in the ED. Dr. William Whiteley. Scottish Senior Clinical Fellow University of Edinburgh Consultant Neurologist NHS Lothian
 Stroke in the ED Dr. William Whiteley Scottish Senior Clinical Fellow University of Edinburgh Consultant Neurologist NHS Lothian 2016 RCP Guideline for Stroke RCP guidelines for acute ischaemic stroke
Stroke in the ED Dr. William Whiteley Scottish Senior Clinical Fellow University of Edinburgh Consultant Neurologist NHS Lothian 2016 RCP Guideline for Stroke RCP guidelines for acute ischaemic stroke
PREOPERATIVE ANAEMIA PATHWAY
 PREOPERATIVE ANAEMIA PATHWAY Surname: Patient ID No. Forename: DOB: / / Age: NHS Number: Likes to be called: Address: Tel. No. Religion/Spirituality: Next of Kin: Name GP Name: GP Practice: Planned Operation:
PREOPERATIVE ANAEMIA PATHWAY Surname: Patient ID No. Forename: DOB: / / Age: NHS Number: Likes to be called: Address: Tel. No. Religion/Spirituality: Next of Kin: Name GP Name: GP Practice: Planned Operation:
Medicines Management Group
 Title SHARED CARE PROTOCOL for Extended Treatment of symptomatic VTE and prevention of its recurrence in Cancer Patients with Solid and Haematological Tumours Scope: Dalteparin (Fragmin ) may be considered
Title SHARED CARE PROTOCOL for Extended Treatment of symptomatic VTE and prevention of its recurrence in Cancer Patients with Solid and Haematological Tumours Scope: Dalteparin (Fragmin ) may be considered
Paravertebral policy. The Acute pain Management Dept, UCLH
 UCLH PARAVERTEBRAL BLOCK (ADULTS) POLICY Paravertebral policy. The Acute pain Management Dept, UCLH DEFINITION A Paravertebral block is a method of providing effective analgesia using a local anaesthetic.
UCLH PARAVERTEBRAL BLOCK (ADULTS) POLICY Paravertebral policy. The Acute pain Management Dept, UCLH DEFINITION A Paravertebral block is a method of providing effective analgesia using a local anaesthetic.
Step-by-step instructions for intravenous (iv) infusions for patients with:
 Step-by-step instructions for intravenous (iv) infusions for patients with: Rheumatoid Arthritis (RA) Systemic Juvenile Idiopathic Arthritis (sjia) Polyarticular Juvenile Idiopathic Arthritis (pjia) Please
Step-by-step instructions for intravenous (iv) infusions for patients with: Rheumatoid Arthritis (RA) Systemic Juvenile Idiopathic Arthritis (sjia) Polyarticular Juvenile Idiopathic Arthritis (pjia) Please
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service
 M AY. 6. 2011 10:37 A M F D A - C D R H - O D E - P M O N O. 4147 P. 1 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control
M AY. 6. 2011 10:37 A M F D A - C D R H - O D E - P M O N O. 4147 P. 1 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control
ENDOVASCULAR THERAPIES FOR ACUTE STROKE
 ENDOVASCULAR THERAPIES FOR ACUTE STROKE Cerebral Arteriogram Cerebral Anatomy Cerebral Anatomy Brain Imaging Acute Ischemic Stroke (AIS) Therapy Main goal is to restore blood flow and improve perfusion
ENDOVASCULAR THERAPIES FOR ACUTE STROKE Cerebral Arteriogram Cerebral Anatomy Cerebral Anatomy Brain Imaging Acute Ischemic Stroke (AIS) Therapy Main goal is to restore blood flow and improve perfusion
Pathology Service User Guide Haematology
 Pathology Service User Guide Haematology St Richard s This section of the Pathology Service User Guide includes: Anticoagulant Therapy Information about the Anticoagulant Clinic Low Molecular Weight Heparin
Pathology Service User Guide Haematology St Richard s This section of the Pathology Service User Guide includes: Anticoagulant Therapy Information about the Anticoagulant Clinic Low Molecular Weight Heparin
St George Hospital Renal Department Guideline: INTERNAL ONLY ANTICOAGULATION - COMMENCEMENT OF HAEMODIALYSIS
 ANTICOAGULATION - COMMENCEMENT OF HAEMODIALYSIS Summary Aim: To prevent clotting of the extracorporeal circuit during haemodialysis If there are no contraindications, heparin can be used. In the first
ANTICOAGULATION - COMMENCEMENT OF HAEMODIALYSIS Summary Aim: To prevent clotting of the extracorporeal circuit during haemodialysis If there are no contraindications, heparin can be used. In the first
Venous Thromboembolism Prophylaxis
 Approved by: Venous Thromboembolism Prophylaxis Vice President and Chief Medical Officer; and Vice President and Chief Operating Officer Corporate Policy & Procedures Manual Number: Date Approved January
Approved by: Venous Thromboembolism Prophylaxis Vice President and Chief Medical Officer; and Vice President and Chief Operating Officer Corporate Policy & Procedures Manual Number: Date Approved January
Policy for the use of intravenous Iron Dextran (CosmoFer )
 Policy for the use of intravenous Iron Dextran (CosmoFer ) Sharepoint Location Clinical Policies and Guidelines Sharepoint Index Directory General Policies and Guidelines Sub Area Haematology and Blood
Policy for the use of intravenous Iron Dextran (CosmoFer ) Sharepoint Location Clinical Policies and Guidelines Sharepoint Index Directory General Policies and Guidelines Sub Area Haematology and Blood
Protocol for IV rtpa Treatment of Acute Ischemic Stroke
 Protocol for IV rtpa Treatment of Acute Ischemic Stroke Acute stroke management is progressing very rapidly. Our team offers several options for acute stroke therapy, including endovascular therapy and
Protocol for IV rtpa Treatment of Acute Ischemic Stroke Acute stroke management is progressing very rapidly. Our team offers several options for acute stroke therapy, including endovascular therapy and
NICE Guidance: Venous thromboembolism (deep vein thrombosis and pulmonary embolism) in patients admitted to hospital 1
 The College of Emergency Medicine Patron: HRH The Princess Royal Churchill House Tel +44 (0)207 404 1999 35 Red Lion Square Fax +44 (0)207 067 1267 London WC1R 4SG www.collemergencymed.ac.uk CLINICAL EFFECTIVENESS
The College of Emergency Medicine Patron: HRH The Princess Royal Churchill House Tel +44 (0)207 404 1999 35 Red Lion Square Fax +44 (0)207 067 1267 London WC1R 4SG www.collemergencymed.ac.uk CLINICAL EFFECTIVENESS
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Metalyse 8,000 units powder and solvent for solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each vial contains
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT Metalyse 8,000 units powder and solvent for solution for injection 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each vial contains
DOCUMENT CONTROL PAGE
 DOCUMENT CONTROL PAGE Title Title: UNDERGOING SPINAL DEFORMITY SURGERY Version: 2 Reference Number: Supersedes Supersedes: all other versions Description of Amendment(s): Revision of analgesia requirements
DOCUMENT CONTROL PAGE Title Title: UNDERGOING SPINAL DEFORMITY SURGERY Version: 2 Reference Number: Supersedes Supersedes: all other versions Description of Amendment(s): Revision of analgesia requirements
o Unenhanced Head CT
 Drip & Ship Protocol Acute Stroke Ready Hospital (ASRH) Duluth Area Primary Stroke Center (St. Luke s & St. Mary s Essentia) PATIENT LABEL Patient displays strokelike symptoms EMS/ED CSS > 0 Glucose >
Drip & Ship Protocol Acute Stroke Ready Hospital (ASRH) Duluth Area Primary Stroke Center (St. Luke s & St. Mary s Essentia) PATIENT LABEL Patient displays strokelike symptoms EMS/ED CSS > 0 Glucose >
MEDICAL RECORD. Last and given name(s)... Personal Identification Number PESEL.. Residence address... Phone number..
 MEDICAL RECORD Last and given name(s)... Personal Identification Number PESEL.. Residence address... Phone number The person authorised by the patient to be contacted and receive information concerning
MEDICAL RECORD Last and given name(s)... Personal Identification Number PESEL.. Residence address... Phone number The person authorised by the patient to be contacted and receive information concerning
The Multi arm Optimization of Stroke Thrombolysis (MOST) Trial
 The Multi arm Optimization of Stroke Thrombolysis (MOST) Trial Study Team Principal Investigators: Opeolu Adeoye, University of Cincinnati (Lead) Andrew Barreto, University of Texas Houston Jim Grotta,
The Multi arm Optimization of Stroke Thrombolysis (MOST) Trial Study Team Principal Investigators: Opeolu Adeoye, University of Cincinnati (Lead) Andrew Barreto, University of Texas Houston Jim Grotta,
Standard operating procedures for preparation and administration of intramuscular injections. No Action Rationale
 Standard operating procedures for preparation and administration of intramuscular injections Preparation Overview No Action Rationale 1 Collect and check all equipment 2 Check that the packaging of all
Standard operating procedures for preparation and administration of intramuscular injections Preparation Overview No Action Rationale 1 Collect and check all equipment 2 Check that the packaging of all
Nursing Process Focus: Patients Receiving Heparin
 Obtain complete heath history including allergies, drug history and possible drug Assess baseline coagulation studies and CBC. Assess for history of bleeding disorders, GI bleeding, cerebral bleed, recent
Obtain complete heath history including allergies, drug history and possible drug Assess baseline coagulation studies and CBC. Assess for history of bleeding disorders, GI bleeding, cerebral bleed, recent
Angiogram, angioplasty and stenting
 Angiogram, angioplasty and stenting Who is this leaflet for? This leaflet is for people who are having: an angiogram angioplasty (with or without a stent) An angiogram (also called an arteriogram or angiography)
Angiogram, angioplasty and stenting Who is this leaflet for? This leaflet is for people who are having: an angiogram angioplasty (with or without a stent) An angiogram (also called an arteriogram or angiography)
Catheter-Directed Thrombolysis for Acute Limb Ischemia. Hwan Jun Jae MD Seoul National University Hospital Seoul, Korea
 Catheter-Directed Thrombolysis for Acute Limb Ischemia Hwan Jun Jae MD Seoul National University Hospital Seoul, Korea Disclosure Speaker name: Hwan Jun Jae... I have the following potential conflicts
Catheter-Directed Thrombolysis for Acute Limb Ischemia Hwan Jun Jae MD Seoul National University Hospital Seoul, Korea Disclosure Speaker name: Hwan Jun Jae... I have the following potential conflicts
Intravenous Iloprost Guidelines. November 2020
 Index No: MMG31 (Formerly MM31) Intravenous Iloprost Guidelines Version: 3.0 Date ratified: Ratified by: (Name of Committee) Name of originator/author, job title and department: November 2017 Medicines
Index No: MMG31 (Formerly MM31) Intravenous Iloprost Guidelines Version: 3.0 Date ratified: Ratified by: (Name of Committee) Name of originator/author, job title and department: November 2017 Medicines
Unrestricted. Dr ppooransari fellowship of perenatalogy
 Unrestricted Dr ppooransari fellowship of perenatalogy Assessment of severity of hemorrhage Significant drops in blood pressure are generally not manifested until substantial bleeding has occurred, and
Unrestricted Dr ppooransari fellowship of perenatalogy Assessment of severity of hemorrhage Significant drops in blood pressure are generally not manifested until substantial bleeding has occurred, and
Children & Young People s Directorate Paediatric-Neonatal Guidelines Checklist & Version Control Sheet
 1 Children & Young People s Directorate Paediatric-Neonatal Guidelines Checklist & Version Control Sheet 1 Name of Guideline / Policy/ Procedure MANAGEMENT OF ACUTE PAEDIATRIC ASTHMA Purpose of Procedure/
1 Children & Young People s Directorate Paediatric-Neonatal Guidelines Checklist & Version Control Sheet 1 Name of Guideline / Policy/ Procedure MANAGEMENT OF ACUTE PAEDIATRIC ASTHMA Purpose of Procedure/
Guideline scope Stroke and transient ischaemic attack in over 16s: diagnosis and initial management (update)
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Guideline scope Stroke and transient ischaemic attack in over s: diagnosis and initial management (update) 0 0 This will update the NICE on stroke and
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Guideline scope Stroke and transient ischaemic attack in over s: diagnosis and initial management (update) 0 0 This will update the NICE on stroke and
Venous interventions in DVT
 Venous interventions in DVT Sriram Narayanan Chief of Vascular and Endovascular Surgery, Tan Tock Seng Hospital A/Prof of Surgery, National University of Singapore ANTI-COAGULATION LMWH Warfarin x 6m Acute
Venous interventions in DVT Sriram Narayanan Chief of Vascular and Endovascular Surgery, Tan Tock Seng Hospital A/Prof of Surgery, National University of Singapore ANTI-COAGULATION LMWH Warfarin x 6m Acute
Package leaflet: Information for the patient. Trasylol 10,000 KIU/ml solution for injection or infusion. Aprotinin
 Package leaflet: Information for the patient Trasylol 10,000 KIU/ml solution for injection or infusion Aprotinin Read all of this leaflet carefully before you are given this medicine because it contains
Package leaflet: Information for the patient Trasylol 10,000 KIU/ml solution for injection or infusion Aprotinin Read all of this leaflet carefully before you are given this medicine because it contains
Standard Operational Procedure. Drainage of Malignant Ascites (Abdominal Paracentesis)
 Standard Operational Procedure Drainage of Malignant Ascites (Abdominal Paracentesis) Background Cancers that involve the peritoneum can cause fluid to build up within the abdominal cavity. This is most
Standard Operational Procedure Drainage of Malignant Ascites (Abdominal Paracentesis) Background Cancers that involve the peritoneum can cause fluid to build up within the abdominal cavity. This is most
Thrombolysis, adjunctive pharmacology and interventions
 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation ESC Annual Congress Munich, 2012 Thrombolysis, adjunctive pharmacology and interventions
ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation ESC Annual Congress Munich, 2012 Thrombolysis, adjunctive pharmacology and interventions
Clinical Competency Name: Post-operative use of Metaraminol in Recovery
 Clinical Competency Name: Post-operative use of Metaraminol in Recovery Trainee name: Title: Ward or department: Method of assessment: Clinical assessor: Name: Title: Professional assessor: Name: Title:
Clinical Competency Name: Post-operative use of Metaraminol in Recovery Trainee name: Title: Ward or department: Method of assessment: Clinical assessor: Name: Title: Professional assessor: Name: Title:
Xarelto (rivaroxaban) Prescriber Guide
 Xarelto (rivaroxaban) Prescriber Guide October 2018 PP-XAR-IE-0031 Contents Patient Alert Card 4 Dosing Recommendations 4 Stroke prevention in adult patients with non-valvular atrial fibrillation 4 Patients
Xarelto (rivaroxaban) Prescriber Guide October 2018 PP-XAR-IE-0031 Contents Patient Alert Card 4 Dosing Recommendations 4 Stroke prevention in adult patients with non-valvular atrial fibrillation 4 Patients
METALYSE. (Tenecteplase)
 METALYSE (Tenecteplase) NAME OF THE MEDICINE METALYSE (tenecteplase) CAS Registry Number 191588-94-0 DESCRIPTION Tenecteplase is a tissue plasminogen activator (t-pa) produced by recombinant DNA technology,
METALYSE (Tenecteplase) NAME OF THE MEDICINE METALYSE (tenecteplase) CAS Registry Number 191588-94-0 DESCRIPTION Tenecteplase is a tissue plasminogen activator (t-pa) produced by recombinant DNA technology,
Section 6 Intra Aortic Balloon Pump
 Section 6 Intra Aortic Balloon Pump The Intra Aortic Balloon Pump (IABP) The balloon is synthetic and is made for single use only. It is threaded into the aorta, usually via a femoral approach. The balloon
Section 6 Intra Aortic Balloon Pump The Intra Aortic Balloon Pump (IABP) The balloon is synthetic and is made for single use only. It is threaded into the aorta, usually via a femoral approach. The balloon
Routine, Every 2 hours, Starting today, If temperature greater than 38.5 C initiate Evaluation for Possible Sepsis Physician Order #829
 Height Weight Allergies General Vital Signs [X] Frequent vital signs Indication: Q15 minutes x (# of occurrences): 4 Q30 minutes x (# of occurrences): 4 Q1 hour x (# of occurrences): Q2 hours x (# of occurrences):
Height Weight Allergies General Vital Signs [X] Frequent vital signs Indication: Q15 minutes x (# of occurrences): 4 Q30 minutes x (# of occurrences): 4 Q1 hour x (# of occurrences): Q2 hours x (# of occurrences):
A step-by-step preparation guide
 A step-by-step preparation guide For needle and needle-free systems This guide provides detailed instructions on the reconstitution, dilution, and storage of VELETRI. It is intended to be used after your
A step-by-step preparation guide For needle and needle-free systems This guide provides detailed instructions on the reconstitution, dilution, and storage of VELETRI. It is intended to be used after your
October 2017 Pulmonary Embolism
 October 2017 Pulmonary Embolism Prof. Ahmed BaHammam, FRCP, FCCP Professor of Medicine College of Medicine King Saud University 1 Objectives Epidemiology Pathophysiology Diagnosis Massive PE Treatment
October 2017 Pulmonary Embolism Prof. Ahmed BaHammam, FRCP, FCCP Professor of Medicine College of Medicine King Saud University 1 Objectives Epidemiology Pathophysiology Diagnosis Massive PE Treatment
Transfusion Requirements and Management in Trauma RACHEL JACK
 Transfusion Requirements and Management in Trauma RACHEL JACK Overview Haemostatic resuscitation Massive Transfusion Protocol Overview of NBA research guidelines Haemostatic resuscitation Permissive hypotension
Transfusion Requirements and Management in Trauma RACHEL JACK Overview Haemostatic resuscitation Massive Transfusion Protocol Overview of NBA research guidelines Haemostatic resuscitation Permissive hypotension
North East Essex Medicines Management Committee
 Colchester Hospital University NHS Foundation Trust North East Essex Clinical Commissioning Group North East Essex Medicines Management Committee ORAL ANTICOAGULANT (Vit K antagonist only) MANAGEMENT GUIDELINES
Colchester Hospital University NHS Foundation Trust North East Essex Clinical Commissioning Group North East Essex Medicines Management Committee ORAL ANTICOAGULANT (Vit K antagonist only) MANAGEMENT GUIDELINES
Elements for a public summary. VI.2.1 Overview of disease epidemiology
 VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology Coronary artery disease and as anticoagulant (inhibiting the clotting of the blood) in patients undergoing surgery to treat blockages
VI.2 Elements for a public summary VI.2.1 Overview of disease epidemiology Coronary artery disease and as anticoagulant (inhibiting the clotting of the blood) in patients undergoing surgery to treat blockages
BLEEDING EMERGENCY. Always apply pressure directly over the bleeding site with your thumb or use an inverted bottle top to localise the pressure
 Bleeding between dialysis sessions is very rare and there are almost always warning signs before this happens. Warning signs include:- Infection of the fistula Bulging area, usually with shiny skin and
Bleeding between dialysis sessions is very rare and there are almost always warning signs before this happens. Warning signs include:- Infection of the fistula Bulging area, usually with shiny skin and
RadRx Your Prescription for Accurate Coding & Reimbursement Copyright All Rights Reserved.
 Interventional Radiology Coding Case Studies Prepared by Stacie L. Buck, RHIA, CCS-P, RCC, CIRCC, AAPC Fellow President & Senior Consultant Week of October 29, 2018 Mesenteric Arteriogram & Thrombectomy/Thrombolysis
Interventional Radiology Coding Case Studies Prepared by Stacie L. Buck, RHIA, CCS-P, RCC, CIRCC, AAPC Fellow President & Senior Consultant Week of October 29, 2018 Mesenteric Arteriogram & Thrombectomy/Thrombolysis
Consultation Group: Cardiology Consultants Clinical lead for cardiology: Dr Andrew Hannah. Review Date: Uncontrolled when printed.
 NHS Grampian Staff Guidelines For The In-Hospital Management Of Unstable Angina And Non-ST-Segment- Elevation Myocardial Infarction Patients (17 Years And Older) Co-ordinators: Cardiology Specialist Clinical
NHS Grampian Staff Guidelines For The In-Hospital Management Of Unstable Angina And Non-ST-Segment- Elevation Myocardial Infarction Patients (17 Years And Older) Co-ordinators: Cardiology Specialist Clinical
