ClinicalTrials.gov Identifier: NCT Sponsor/company: Sanofi-Aventis. Date: 08/02/ 2008
|
|
|
- Harvey Pope
- 5 years ago
- Views:
Transcription
1 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: Sanofi-Aventis ClinicalTrials.gov Identifier: NCT Generic drug name: Ramipril Study Code: HOE498_3501 Date: 08/02/ 2008 Title of the study: Investigator(s): Study center(s): Publications (reference): Study period: Date first patient enrolled: Date last patient completed: Objectives: A placebo-controlled, double-blind, randomized, multicenter study of ramipril 5 and 10 mg capsules and insulin infusion in subjects with unstable coronary syndromes. Richard Gallo, MD and Pierre Theroux, MD Montreal Heart Institute 5000 Belanger East Montreal, Qc Canada H1T 1C8 10 Sites No Publication 24-Jun Aug-2005 The co-primary objectives of the study are: Phase of development: IIIb No Objection Letter received 18 Sept 2003 To demonstrate that the acute administration of ramipril will control the inflammation process in patients with high-risk acute coronary syndrome (ACS) as assessed by the highly specific C-reactive protein (hscrp) blood levels To demonstrate that the normalization of blood glucose levels with intravenous insulin will improve the inflammation process during the acute phase of an ACS as assessed by tumor necrosing factor alpha (TNFa) blood levels Methodology: This was two by two design in which ACS patients were randomized to double-blind ramipril 10 mg or placebo (6 weeks)/ramipril 5 mg (2 weeks) for the first 8 weeks and randomized to open-label intravenous (IV) insulin therapy or conventional therapy for the first 72 hours or until a percutaneous coronary interventions (PCI) or a coronary artery bypass graft surgery (CABG). From weeks 8 to 12 all patients were to receive ramipril 10 mg. Blood was collected at randomization, 72 hours and 6 weeks and frozen to be assayed for hscrp and TNFa at the conclusion of the study. At 24 weeks a telephone call was made to determine if the subject was alive and had not experienced a myocardial infarction, stroke or urgent intervention for recurrent symptoms. 1
2 Number of Patients: Planned: 412 patie nts Randomized: 78 patients Treated: 78 patients Evaluated: Diagnosis and criteria for inclusion: Changes from baseline in hscrp and TNFa Safety: Treatment emergent adverse events (TEAEs) Male and female patien ts = 18 years presenting within 12 hours after the last episode of chest pain with either accelerating pattern of anginal pain, prolonged or recurrent anginal pain at rest or minimal effort or anginal pain at rest or minimal effort >48 hours after an acute myocardial infarction and electrocardiographic evidence of myocardial ischemia or abnormal cardiac markers while excluding having heart failure, diabetes mellitus requiring insulin therapy, or an active chronic inflammatory disease, auto-immune disease or cancer. This study was stopped when the last patient randomized completed 12 weeks of the study and before all patients reached 24 weeks of follow-up. Investigational product: ramipril ramipril insulin Dose: 5 mg 10 mg hospital pharmacy Administration: qd qd as required Duration of treatment: 12 weeks Duration of observation: 24 weeks Reference therapy: ramipril placebo conventional therapy Dose: Administration: Criteria for evaluation: Statistical methods: N/A Qd The current report is an abbreviated report, and as such, only the safety results are being presented in full. TEAEs were evaluate d, and analyzed using descriptive statistics. A TEAE is defined as any new adverse event (AE) or any existing AE which worsened following the first dose of study drug until 7 days following the last dose of study medication Descrip tive statistics using means, medians, standard deviations, and ranges for continuous data and numbers of patients and percentages for categorical data. 2
3 Summary: Since only 78 of the planned 412 subjects were randomized and treated, this abbreviated format was used to summarize the results. In addition, since the insulin was only used during the first 72 hours, and it was obtained from commercially available supplies in the hospital pharmacy this summary will only report the results with respect to ramipril. The study completion and demographic results with respect to insulin can be found in the attached tables. Study Completion Characteristic Statistic Ramipril Placebo Number of patients Number Withdrawn 19 (50.0) 16 (40.0) Duration of study drug (days)* Duration of follow-up (days) Reason for withdrawal Adverse event Treatment not required Death Lost to follow-up Other Mean (SD) Median Range Mean (SD) Median Range 71.4 (28.37) (55.38) (10.5) 0 ( 0.0) 2 (10.5) 2 (10.5) 13 (68.4) 67.5 (28.15) (59.67) (25.0) 1 ( 6.3) 0 ( 0.0) 2 (12.5) 9 (56.3) * - For the first 56 days in the placebo, subjects were receiving placebo, after which they received ramipril Demography Characteristic Statistic Ramipril Placebo Number of patients Number Age (years) Mean (SD) 61.5 (11.47) 62.8 (11.94) Median Range Males 32 (84.2) 30 (75.0) Race White 38 (100.0) 39 (97.5) Asian 0 ( 0.0) 1( 2.5) TEAEs were reported by 34 (89.5%) ramipril treated patients and 37 (92.5%) patients who received placebo. Possibly related TEAEs were reported by 5 (13.2%) ramipril treated patients and 8 (20.0%) patients who received placebo. The most frequently (>3 patients in any treatment group) reported TEAEs are found in the table below. All Treatment Emergent Events Reported by More than 3 Patients System Organ Class/Preferred Term Ramipril Placebo Number of patients
4 Any Event 34 (89.5%) 37 (92.5%) General disorders Chest pain Fatigue Edema peripheral Nervous system disorders Headache Dizziness Vascular disorders Hypotension Hematoma Hypertension Gastrointestinal disorders Nausea Constipation Cardiac disorders Angina pectoris Skin and subcutaneous tissue disorders Ecchymosis Psychiatric disorders Anxiety Injury and procedural complications Post procedural pain Metabolism and nutrition disorders 17 (44.7%) 15 (39.5%) 4 (10.5%) 17 (44.7%) 9 (23.7%) 8 (21.1%) 12 (31.6%) 8 (21.1%) 4 (10.5%) 11 (28.9%) 6 (15.8%) 9 (23.7%) 7 (18.4%) 5 (13.2%) 6 (15.8%) 4 (10.5%) 5 (13.2%) 3 ( 7.9%) 19 (47.5%) 14 (35.0%) 3 ( 7.5%) 4 (10.0%) 14 (35.0%) 7 (17.5%) 8 (20.0%) 19 (47.5%) 8 (20.0%) 7 (17.5%) 13 (32.5%) 4 (10.0%) 14 (35.0%) 8 (20.0%) 9 (22.5%) 4 (10.0%) 9 (22.5%) Hypoglycemia 2 ( 5.3%) 6 (15.0%) Serious TEAEs were reported by 7 (18.4%) treated patients and 7 (17.5%) patients who received placebo. No serious TEAEs was possibly related to study drug. A 78 white male ramipril treated patient went into cardiogenic shock day 4 of treatment and died. This event was not possibly related to study drug. Two (5.3%) ramipril 4
5 Date of report: 30 November 2007 treated patients and 2 (5.0%) patients who received placebo had hypoglycemic events that met the definitions for expedited reporting. That is to say that the symptomatic hypoglycemia was secondary to insulin infusion. Two (5.3%) ramipril treated patients and patients who received placebo experienced TEAEs that lead to the discontinuation of study drug. 5
6 LIST OF ABBREVIATIONS AND DEFINITION OF TERMS Abbreviation Definition ACS AE(s) CABG CRF(s) hscrp PCI PCSA SAE(s) TEAE(s) TNFa WHO Acute coronary syndrome Adverse Event(s) Coronary Artery Bypass Graft Surgery Case Report Form(s) Highly specific C-reactive protein Percutaneous Coronary Interventions Potentially Clinically Significant Abnormality Serious Adverse Event(s) Treatment Emergent Adverse Event Tumor necrosing factor alpha World Health Organization 6
Clopidogrel Date: 15 July 2008
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinicalTrials.gov
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinicalTrials.gov
Full Novartis CTRD Results Template
 Full Novartis CTRD Results Template Sponsor Novartis Generic Drug Name vildagliptin Therapeutic Area of Trial Type 2 diabetes Approved Indication Type 2 diabetes Protocol Number CLAF237A23138E1 Title A
Full Novartis CTRD Results Template Sponsor Novartis Generic Drug Name vildagliptin Therapeutic Area of Trial Type 2 diabetes Approved Indication Type 2 diabetes Protocol Number CLAF237A23138E1 Title A
SYNOPSIS 2/198 CSR_BDY-EFC5825-EN-E02. Name of company: TABULAR FORMAT (For National Authority Use only)
 SYNOPSIS Title of the study: A randomized, double-blind, placebo-controlled, parallel-group, fixed-dose (rimonabant 20 mg) multicenter study of long-term glycemic control with rimonabant in treatment-naïve
SYNOPSIS Title of the study: A randomized, double-blind, placebo-controlled, parallel-group, fixed-dose (rimonabant 20 mg) multicenter study of long-term glycemic control with rimonabant in treatment-naïve
Efficacy/pharmacodynamics: 85 Safety: 89
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor/Company: Sanofi Drug substance:
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor/Company: Sanofi Drug substance:
Clinical Trial Synopsis TL-OPI-518, NCT#
 Clinical Trial Synopsis, NCT# 00225264 Title of Study: A Double-Blind, Randomized, Comparator-Controlled Study in Subjects With Type 2 Diabetes Mellitus Comparing the Effects of Pioglitazone HCl vs Glimepiride
Clinical Trial Synopsis, NCT# 00225264 Title of Study: A Double-Blind, Randomized, Comparator-Controlled Study in Subjects With Type 2 Diabetes Mellitus Comparing the Effects of Pioglitazone HCl vs Glimepiride
Clinical Trial Synopsis TL-OPI-516, NCT#
 Clinical Trial Synopsis, NCT#00225277 Title of Study: A Double-Blind, Randomized, Comparator-Controlled Study in Subjects With Type 2 Diabetes Mellitus Comparing the Effects of Pioglitazone HCl Versus
Clinical Trial Synopsis, NCT#00225277 Title of Study: A Double-Blind, Randomized, Comparator-Controlled Study in Subjects With Type 2 Diabetes Mellitus Comparing the Effects of Pioglitazone HCl Versus
Sponsor / Company: Sanofi Drug substance(s): Insulin Glargine. According to template: QSD VERSION N 4.0 (07-JUN-2012) Page 1
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
Protocol GTC : A Randomized, Open Label, Parallel Design Study of Sevelamer Hydrochloride (Renagel ) in Chronic Kidney Disease Patients.
 Protocol GTC-68-208: A Randomized, Open Label, Parallel Design Study of Sevelamer Hydrochloride (Renagel ) in Chronic Kidney Disease Patients. These results are supplied for informational purposes only.
Protocol GTC-68-208: A Randomized, Open Label, Parallel Design Study of Sevelamer Hydrochloride (Renagel ) in Chronic Kidney Disease Patients. These results are supplied for informational purposes only.
Clinical Trial Results Summary Study EN3409-BUP-305
 Title of Study: A 52-Week, Open-Label, Long-Term Treatment Evaluation of the Safety and Efficacy of BEMA Buprenorphine in Subjects with Moderate to Severe Chronic Pain Coordinating Investigator: Martin
Title of Study: A 52-Week, Open-Label, Long-Term Treatment Evaluation of the Safety and Efficacy of BEMA Buprenorphine in Subjects with Moderate to Severe Chronic Pain Coordinating Investigator: Martin
SYNOPSIS. Administration: subcutaneous injection Batch number(s):
 SYNOPSIS Title of the study: A randomized, double-blind, placebo-controlled, 2-arm parallel-group, multicenter 24-week study followed by an extension assessing the efficacy and safety of AVE0010 on top
SYNOPSIS Title of the study: A randomized, double-blind, placebo-controlled, 2-arm parallel-group, multicenter 24-week study followed by an extension assessing the efficacy and safety of AVE0010 on top
Sponsor / Company: Sanofi Drug substance(s): Insulin Glargine (HOE901) Insulin Glulisine (HMR1964)
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
GSK Medicine: Study Number: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Study Code: Date: 27 July 2007
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: Generic drug name:
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: Generic drug name:
Clinical Trial Synopsis TL-OPI-525, NCT#
 Clinical Trial Synopsis, NCT#00762736 Title of Study: A Phase II, Double-Blind, Randomized, Placebo-Controlled, Proof-of-Concept Study of the Efficacy, Safety, and Tolerability of Pioglitazone HCl (ACTOS
Clinical Trial Synopsis, NCT#00762736 Title of Study: A Phase II, Double-Blind, Randomized, Placebo-Controlled, Proof-of-Concept Study of the Efficacy, Safety, and Tolerability of Pioglitazone HCl (ACTOS
Synopsis Style Clinical Study Report SR EFC10139 Version number: 1 (electronic 2.0)
 SYNOPSIS Title of the study: A randomized, double-blind, parallel-group, multicenter, multinational study to assess the long-term effect, over 1 year, of rimonabant 10 mg in comparison with rimonabant
SYNOPSIS Title of the study: A randomized, double-blind, parallel-group, multicenter, multinational study to assess the long-term effect, over 1 year, of rimonabant 10 mg in comparison with rimonabant
PFIZER INC. THERAPEUTIC AREA AND FDA APPROVED INDICATIONS: See USPI.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
These results are supplied for informational purposes only.
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinialTrials.gov
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinialTrials.gov
Number of patients: Planned: 403 Randomized: 391 Treated: 390 Efficacy: 363 (Full analysis set) 356 (Per-protocol set)
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company : sanofi-aventis
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company : sanofi-aventis
Study No.: LOV Title: Rationale: Phase: IIB Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary-
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Sponsor / Company: Sanofi Drug substance(s): AMARYL M (1/250 mg) / HOE490
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
ClinialTrials.gov Identifier: HOE901_4020 Insulin Glargine Date: Study Code: This was a multicenter study that was conducted at 59 US sites
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: Generic drug name:
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: Generic drug name:
PFIZER INC. THERAPEUTIC AREA AND FDA APPROVED INDICATIONS: See United States Package Insert (USPI)
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. Study Initiation Date and Completion Dates: 09 March 2000 to 09 August 2001.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centers: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
PFIZER INC. THERAPEUTIC AREA AND FDA APPROVED INDICATIONS: See United States Package Insert (USPI)
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Sponsor Novartis. Generic Drug Name Fluvastatin. Therapeutic Area of Trial Dyslipidemia
 Page 1 Sponsor Novartis Generic Drug Name Fluvastatin Therapeutic Area of Trial Dyslipidemia Approved Indication Therapeutic area and approved indications in Germany: Hypercholesterolemia (HC), combined
Page 1 Sponsor Novartis Generic Drug Name Fluvastatin Therapeutic Area of Trial Dyslipidemia Approved Indication Therapeutic area and approved indications in Germany: Hypercholesterolemia (HC), combined
Sponsor / Company: Sanofi Drug substance(s): Insulin Glargine. Study Identifiers: NCT
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
Sponsor: Sanofi Drug substance(s): SAR342434
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor: Sanofi Drug substance(s):
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor: Sanofi Drug substance(s):
Full Novartis CTRD Results Template
 Full Novartis CTRD Results Template Sponsor Novartis Generic Drug Name vildagliptin Therapeutic Area of Trial Type 2 diabetes Approved Indication Type 2 diabetes Protocol Number CLAF237A23137E1 Title A
Full Novartis CTRD Results Template Sponsor Novartis Generic Drug Name vildagliptin Therapeutic Area of Trial Type 2 diabetes Approved Indication Type 2 diabetes Protocol Number CLAF237A23137E1 Title A
Sponsor. Novartis Pharmaceuticals Corporation Generic Drug Name. Agomelatine Therapeutic Area of Trial. Major depressive disorder Approved Indication
 Clinical Trial Results Database Page 1 Sponsor Novartis Pharmaceuticals Corporation Generic Drug Name Therapeutic Area of Trial Major depressive disorder Approved Indication Investigational drug Study
Clinical Trial Results Database Page 1 Sponsor Novartis Pharmaceuticals Corporation Generic Drug Name Therapeutic Area of Trial Major depressive disorder Approved Indication Investigational drug Study
IRBES_R_04320 Generic drug name: Irbesartan + amlodipine Date: Study Code: 31 active centers: 20 in Korea, 7 in India and 4 in Philippines.
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinicalTrials.gov
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinicalTrials.gov
Sponsor / Company: Sanofi Drug substance(s): HOE901-U300 (insulin glargine) According to template: QSD VERSION N 4.0 (07-JUN-2012) Page 1
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
Supplementary material 1. Definitions of study endpoints (extracted from the Endpoint Validation Committee Charter) 1.
 Rationale, design, and baseline characteristics of the SIGNIFY trial: a randomized, double-blind, placebo-controlled trial of ivabradine in patients with stable coronary artery disease without clinical
Rationale, design, and baseline characteristics of the SIGNIFY trial: a randomized, double-blind, placebo-controlled trial of ivabradine in patients with stable coronary artery disease without clinical
ClinicalTrials.gov Identifier: sanofi-aventis. Sponsor/company:
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinicalTrials.gov
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinicalTrials.gov
23-Aug-2011 Lixisenatide (AVE0010) - EFC6014 Version number: 1 (electronic 1.0)
 SYNOPSIS Title of the study: A randomized, double-blind, placebo-controlled, parallel-group, multicenter 24-week study followed by an extension assessing the efficacy and safety of AVE0010 on top of metformin
SYNOPSIS Title of the study: A randomized, double-blind, placebo-controlled, parallel-group, multicenter 24-week study followed by an extension assessing the efficacy and safety of AVE0010 on top of metformin
Investigators, study sites Multicenter, 35 US sites. Coordinating Investigator: Richard Bergenstal, MD
 STUDY SYNOPSIS Study number Title HMR1964A/3502 Apidra (insulin glulisine) administered in a fixed-bolus regimen vs. variable-bolus regimen based on carbohydrate counting in adult subjects with type 2
STUDY SYNOPSIS Study number Title HMR1964A/3502 Apidra (insulin glulisine) administered in a fixed-bolus regimen vs. variable-bolus regimen based on carbohydrate counting in adult subjects with type 2
Clinical Trial Results Database Page 1
 Clinical Trial Results Database Page 1 Sponsor Novartis Pharmaceuticals Corporation Generic Drug Name Therapeutic Area of Trial Major Depressive Disorder (MDD) Approved Indication Treatment of major depressive
Clinical Trial Results Database Page 1 Sponsor Novartis Pharmaceuticals Corporation Generic Drug Name Therapeutic Area of Trial Major Depressive Disorder (MDD) Approved Indication Treatment of major depressive
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Clinical Trial Results Posting
 RD..3.2 V1. Page/Seite 1 of/von 5 CT Registry ID#: NCT2428 (ClinicalTrials.gov Identifier number) These results are supplied for informational purposes only. Prescribing decisions should be made based
RD..3.2 V1. Page/Seite 1 of/von 5 CT Registry ID#: NCT2428 (ClinicalTrials.gov Identifier number) These results are supplied for informational purposes only. Prescribing decisions should be made based
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Sponsor / Company: Sanofi Drug substance(s): Insulin Glargine (HOE901) Insulin Glulisine (HMR1964)
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
Sponsor: Sanofi Drug substance(s): Lantus /insulin glargine. Study Identifiers: U , NCT Study code: LANTUL07225
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor: Sanofi Drug substance(s):
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor: Sanofi Drug substance(s):
2.0 Synopsis. Adalimumab M Clinical Study Report R&D/04/900. (For National Authority Use Only) Referring to Part of Dossier: Volume:
 2. Synopsis Abbott Laboratories Name of Study Drug: Name of Active Ingredient: Title of Study: Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority Use Only) Phase
2. Synopsis Abbott Laboratories Name of Study Drug: Name of Active Ingredient: Title of Study: Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority Use Only) Phase
These results are supplied for informational purposes only.
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinialTrials.gov
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinialTrials.gov
Randomized comparison of single versus double mammary coronary artery bypass grafting: 5 year outcomes of the Arterial Revascularization Trial
 Randomized comparison of single versus double mammary coronary artery bypass grafting: 5 year outcomes of the Arterial Revascularization Trial Embargoed until 10:45 a.m. CT, Monday, Nov. 14, 2016 David
Randomized comparison of single versus double mammary coronary artery bypass grafting: 5 year outcomes of the Arterial Revascularization Trial Embargoed until 10:45 a.m. CT, Monday, Nov. 14, 2016 David
2.0 Synopsis. Adalimumab R&D/04/118. (For National Authority Use Only) Referring to Part of Dossier: Volume:
 2.0 Synopsis Abbott Laboratories Name of Study Drug: Adalimumab Name of Active Ingredient: Adalimumab Title of Study: Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority
2.0 Synopsis Abbott Laboratories Name of Study Drug: Adalimumab Name of Active Ingredient: Adalimumab Title of Study: Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority
SYNOPSIS. Clinical Study Report CN138002: Addendum 1. Individual Study Table Referring to the Dossier
 Name of Sponsor/Company: Bristol-Myers Squibb Name of Finished Product: Abilify Name of Active Ingredient: aripiprazole Individual Study Table Referring to the Dossier (For National Authority Use Only)
Name of Sponsor/Company: Bristol-Myers Squibb Name of Finished Product: Abilify Name of Active Ingredient: aripiprazole Individual Study Table Referring to the Dossier (For National Authority Use Only)
Sponsor / Company: Sanofi Drug substance(s): insulin glargine (HOE901) According to template: QSD VERSION N 4.0 (07-JUN-2012) Page 1
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
Study Number CAIN457C2302 (core study) and CAIN457C2302E1 (extension study)
 Clinical Trial Results Database Page 1 Sponsor Novartis Generic Drug Name Secukinumab Therapeutic Area of Trial Uveitis Approved Indication Investigational Study Number CC2302 (core study) and CC2302E1
Clinical Trial Results Database Page 1 Sponsor Novartis Generic Drug Name Secukinumab Therapeutic Area of Trial Uveitis Approved Indication Investigational Study Number CC2302 (core study) and CC2302E1
ClinialTrials.gov Identifier: sanofi-aventis. Sponsor/company: 07/November/2008
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinialTrials.gov
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinialTrials.gov
Sponsor Novartis. Generic Drug Name Vildagliptin/Metformin. Therapeutic Area of Trial Type 2 diabetes. Approved Indication Type 2 diabetes
 Clinical Trial Results Database Page 1 Sponsor Novartis Generic Drug Name Vildagliptin/Metformin Therapeutic Area of Trial Type 2 diabetes Approved Indication Type 2 diabetes Study Number CLMF237A2309
Clinical Trial Results Database Page 1 Sponsor Novartis Generic Drug Name Vildagliptin/Metformin Therapeutic Area of Trial Type 2 diabetes Approved Indication Type 2 diabetes Study Number CLMF237A2309
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Allergan Not Applicable AGN A Multi-Center, Double-Blind, Randomized, Placebo-Controlled, Multiple Dose, Parallel
 Peripheral Neuropathy Design, Dose Ranging Study of the Safety and Efficacy of AGN 203818 in Patients with Painful Diabetic 203818-004. A Multi-Center, Double-Blind, Randomized, Placebo-Controlled, Multiple
Peripheral Neuropathy Design, Dose Ranging Study of the Safety and Efficacy of AGN 203818 in Patients with Painful Diabetic 203818-004. A Multi-Center, Double-Blind, Randomized, Placebo-Controlled, Multiple
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
ClinicalTrials.gov Identifier: sanofi-aventis. Sponsor/company:
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinicalTrials.gov
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinicalTrials.gov
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
CARDIOVASCULAR SAFETY OF FEBUXOSTAT OR ALLOPURINOL IN PATIENTS WITH GOUT AND CARDIOVASCULAR DISEASE (The CARES Trial)
 CARDIOVASCULAR SAFETY OF FEBUXOSTAT OR ALLOPURINOL IN PATIENTS WITH GOUT AND CARDIOVASCULAR DISEASE (The CARES Trial) William B. White, MD, for the CARES Investigators Calhoun Cardiology Center University
CARDIOVASCULAR SAFETY OF FEBUXOSTAT OR ALLOPURINOL IN PATIENTS WITH GOUT AND CARDIOVASCULAR DISEASE (The CARES Trial) William B. White, MD, for the CARES Investigators Calhoun Cardiology Center University
PFIZER INC. Study Initiation Date and Completion Dates: Information not available (Date of Statistical Report: 16 May 2004)
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
Core Safety Profile. Pharmaceutical form(s)/strength: Film-coated tablets 1.25 mg, 2.5 mg, 3.75 mg, 5 mg, 7.5 mg and 10 mg. Date of FAR:
 Core Safety Profile Active substance: Bisoprolol Pharmaceutical form(s)/strength: Film-coated tablets 1.25 mg, 2.5 mg, 3.75 mg, 5 mg, 7.5 mg and 10 mg P - RMS: FI/H/PSUR/0002/002 Date of FAR: 13.12.2011
Core Safety Profile Active substance: Bisoprolol Pharmaceutical form(s)/strength: Film-coated tablets 1.25 mg, 2.5 mg, 3.75 mg, 5 mg, 7.5 mg and 10 mg P - RMS: FI/H/PSUR/0002/002 Date of FAR: 13.12.2011
Supplement materials:
 Supplement materials: Table S1: ICD-9 codes used to define prevalent comorbid conditions and incident conditions Comorbid condition ICD-9 code Hypertension 401-405 Diabetes mellitus 250.x Myocardial infarction
Supplement materials: Table S1: ICD-9 codes used to define prevalent comorbid conditions and incident conditions Comorbid condition ICD-9 code Hypertension 401-405 Diabetes mellitus 250.x Myocardial infarction
Study No. 178-CL-008 Report Final Version, 14 Dec 2006 Reissued Version, 18 Jul 2011 Astellas Pharma Europe B.V. Page 13 of 122
 Page 13 of 122 3 SYNOPSIS Title of study: (International) Study No: A randomized, double-blind, parallel group, proof-of-concept study of in comparison with placebo and tolterodine in patients with symptomatic
Page 13 of 122 3 SYNOPSIS Title of study: (International) Study No: A randomized, double-blind, parallel group, proof-of-concept study of in comparison with placebo and tolterodine in patients with symptomatic
Sponsor / Company: Sanofi Drug substance(s): SAR (iniparib)
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
Medicine Dr. Omed Lecture 2 Stable and Unstable Angina
 Medicine Dr. Omed Lecture 2 Stable and Unstable Angina Risk stratification in stable angina. High Risk; *post infarct angina, *poor effort tolerance, *ischemia at low workload, *left main or three vessel
Medicine Dr. Omed Lecture 2 Stable and Unstable Angina Risk stratification in stable angina. High Risk; *post infarct angina, *poor effort tolerance, *ischemia at low workload, *left main or three vessel
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
This was a randomized, double-blind, placebo-controlled, fixed-dose, parallel-group study.
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
PFIZER INC. THERAPEUTIC AREA AND FDA APPROVED INDICATIONS: See USPI.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
Supplementary Online Content
 Supplementary Online Content Valle JA, Tamez H, Abbott JD, et al. Contemporary use and trends in unprotected left main coronary artery percutaneous coronary intervention in the United States: an analysis
Supplementary Online Content Valle JA, Tamez H, Abbott JD, et al. Contemporary use and trends in unprotected left main coronary artery percutaneous coronary intervention in the United States: an analysis
DUKECATHR Dataset Dictionary
 DUKECATHR Dataset Dictionary Version of DUKECATH dataset for educational use that has been modified to be unsuitable for clinical research or publication (Created Date and Time: 28OCT16 14:35) Table of
DUKECATHR Dataset Dictionary Version of DUKECATH dataset for educational use that has been modified to be unsuitable for clinical research or publication (Created Date and Time: 28OCT16 14:35) Table of
PFIZER INC. PROTOCOL TITLE: Efficacy and Safety of the Authentic Recombinant Human Somatropin Genotropin in Children with Familial Short Stature
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
Bristol-Myers Squibb
 A Study of the Safety and Efficacy of plus Tenofovir in Adults with Chronic Hepatitis B Virus Infection with Previous Nucleoside/Nucleotide Treatment Failure () FINAL CLINICAL STUDY REPORT EUDRACT Number:
A Study of the Safety and Efficacy of plus Tenofovir in Adults with Chronic Hepatitis B Virus Infection with Previous Nucleoside/Nucleotide Treatment Failure () FINAL CLINICAL STUDY REPORT EUDRACT Number:
Study No.: Title: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
Cardiovascular Disorders Lecture 3 Coronar Artery Diseases
 Cardiovascular Disorders Lecture 3 Coronar Artery Diseases By Prof. El Sayed Abdel Fattah Eid Lecturer of Internal Medicine Delta University Coronary Heart Diseases It is the leading cause of death in
Cardiovascular Disorders Lecture 3 Coronar Artery Diseases By Prof. El Sayed Abdel Fattah Eid Lecturer of Internal Medicine Delta University Coronary Heart Diseases It is the leading cause of death in
Nilotinib AEs (adverse events) in CML population:
 Nilotinib AEs (adverse events) in CML population: The percentages below were taken from a randomized trial of nilotinib 300mg BID in newly diagnosed Ph+ CML patients (N=279) taken from the Tasigna 2017
Nilotinib AEs (adverse events) in CML population: The percentages below were taken from a randomized trial of nilotinib 300mg BID in newly diagnosed Ph+ CML patients (N=279) taken from the Tasigna 2017
Summary ID#7029. Clinical Study Summary: Study F1D-MC-HGKQ
 CT Registry ID# 7029 Page 1 Summary ID#7029 Clinical Study Summary: Study F1D-MC-HGKQ Clinical Study Report: Versus Divalproex and Placebo in the Treatment of Mild to Moderate Mania Associated with Bipolar
CT Registry ID# 7029 Page 1 Summary ID#7029 Clinical Study Summary: Study F1D-MC-HGKQ Clinical Study Report: Versus Divalproex and Placebo in the Treatment of Mild to Moderate Mania Associated with Bipolar
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
2.0 Synopsis. Adalimumab DE019 OLE (5-year) Clinical Study Report Amendment 1 R&D/06/095. (For National Authority Use Only)
 2.0 Synopsis Abbott Laboratories Name of Study Drug: Humira Name of Active Ingredient: Adalimumab Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority Use Only) Title
2.0 Synopsis Abbott Laboratories Name of Study Drug: Humira Name of Active Ingredient: Adalimumab Individual Study Table Referring to Part of Dossier: Volume: Page: (For National Authority Use Only) Title
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. PROPRIETARY DRUG NAME / GENERIC DRUG NAME: Advil / Ibuprofen
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. PROPRIETARY DRUG NAME / GENERIC DRUG NAME: Advil / Ibuprofen
Supplementary Table 1. Details of the components of the primary composite endpoint
 Supplementary Table 1. Details of the components of the primary composite endpoint 1. Death The cause of death will be defined by the underlying cause, not the immediate mode of death. Death will be classified
Supplementary Table 1. Details of the components of the primary composite endpoint 1. Death The cause of death will be defined by the underlying cause, not the immediate mode of death. Death will be classified
Adults With Diagnosed Diabetes
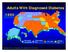 Adults With Diagnosed Diabetes 1990 No data available Less than 4% 4%-6% Above 6% Mokdad AH, et al. Diabetes Care. 2000;23(9):1278-1283. Adults With Diagnosed Diabetes 2000 4%-6% Above 6% Mokdad AH, et
Adults With Diagnosed Diabetes 1990 No data available Less than 4% 4%-6% Above 6% Mokdad AH, et al. Diabetes Care. 2000;23(9):1278-1283. Adults With Diagnosed Diabetes 2000 4%-6% Above 6% Mokdad AH, et
This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data.
 abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the
abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the
Blood Eosinophils and Response to Maintenance COPD Treatment: Data from the FLAME Trial. Online Data Supplement
 Blood Eosinophils and Response to Maintenance COPD Treatment: Data from the FLAME Trial Nicolas Roche, Kenneth R. Chapman, Claus F. Vogelmeier, Felix JF Herth, Chau Thach, Robert Fogel, Petter Olsson,
Blood Eosinophils and Response to Maintenance COPD Treatment: Data from the FLAME Trial Nicolas Roche, Kenneth R. Chapman, Claus F. Vogelmeier, Felix JF Herth, Chau Thach, Robert Fogel, Petter Olsson,
SYNOPSIS. Number of subjects: Planned: 22 Randomized: 23 Treated: 23. Evaluated: Pharmacodynamic: 22 Safety: 23 Pharmacokinetics: 22
 SYNOPSIS Title of the study: A randomized, cross-over, open, euglycemic clamp study on the relative bioavailability and activity of 0.6 U/kg insulin glargine and 20 µg lixisenatide, given as on-site mix
SYNOPSIS Title of the study: A randomized, cross-over, open, euglycemic clamp study on the relative bioavailability and activity of 0.6 U/kg insulin glargine and 20 µg lixisenatide, given as on-site mix
Study No.: Title: Rationale: Phase: Study Period: Study Design: Centres: Indication: Treatment: Objectives: Primary Outcome/Efficacy Variable:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
BRL /RSD-101RLL/1/CPMS-716. Report Synopsis
 Report Synopsis Study Title: A Multicenter, Open-label, Six-Month Extension Study to Assess the Long-term Safety of Paroxetine in Children and Adolescents with Major Depressive Disorder (MDD) or Obsessive-Compulsive
Report Synopsis Study Title: A Multicenter, Open-label, Six-Month Extension Study to Assess the Long-term Safety of Paroxetine in Children and Adolescents with Major Depressive Disorder (MDD) or Obsessive-Compulsive
Study No.:MPX Title: Rationale: Phase: IIB Study Period: Study Design: Centres: Indication: Treatment: Objectives:
 The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
The study listed may include approved and non-approved uses, formulations or treatment regimens. The results reported in any single study may not reflect the overall results obtained on studies of a product.
PFIZER INC. PROTOCOL TITLE: Growth hormone therapy in short children after renal transplantation for chronic renal failure in Germany.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
Previous Study Return to List Next Study
 A service of the U.S. National Institutes of Health Trial record 1 of 1 for: 42603ATT3013 Previous Study Return to List Next Study A Study to Evaluate Effectiveness and Safety of Prolonged Release OROS
A service of the U.S. National Institutes of Health Trial record 1 of 1 for: 42603ATT3013 Previous Study Return to List Next Study A Study to Evaluate Effectiveness and Safety of Prolonged Release OROS
Clinical Study Synopsis
 Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Clinical Study Synopsis This Clinical Study Synopsis is provided for patients and healthcare professionals to increase the transparency of Bayer's clinical research. This document is not intended to replace
Sponsor Novartis. Generic Drug Name. NA (not existing yet) Therapeutic Area of Trial Parkinson s Disease L-dopa induced dyskinesia
 Page 1 Sponsor Novartis Generic Drug Name NA (not existing yet) Therapeutic Area of Trial Parkinson s Disease L-dopa induced dyskinesia Approved Indication Investigational. Study Number CA2206 Title A
Page 1 Sponsor Novartis Generic Drug Name NA (not existing yet) Therapeutic Area of Trial Parkinson s Disease L-dopa induced dyskinesia Approved Indication Investigational. Study Number CA2206 Title A
PFIZER INC. PROPRIETARY DRUG NAME /GENERIC DRUG NAME: Cerebyx / Fosphenytoin Sodium
 PFIZER INC These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
PFIZER INC These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. For publications based on this study, see associated bibliography.
2.0 Synopsis. Choline fenofibrate capsules (ABT-335) M Clinical Study Report R&D/06/772. (For National Authority Use Only) Name of Study Drug:
 2.0 Synopsis Abbott Laboratories Individual Study Table Referring to Part of Dossier: (For National Authority Use Only) Name of Study Drug: Volume: Choline Fenofibrate (335) Name of Active Ingredient:
2.0 Synopsis Abbott Laboratories Individual Study Table Referring to Part of Dossier: (For National Authority Use Only) Name of Study Drug: Volume: Choline Fenofibrate (335) Name of Active Ingredient:
Sponsor / Company: Sanofi Drug substance(s): Docetaxel (Taxotere )
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription. Sponsor / Company: Sanofi Drug substance(s):
Off-white, round, with faceted edges, scored on one side and bearing the inscription IK10 (10mg) or IK20 (20mg).
 1 TRADE NAME OF THE MEDICINAL PRODUCT Ikorel 10mg and 20mg Tablet 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Nicorandil 10mg or 20mg For a full list of excipients, see section 6.1 3 PHARMACEUTICAL FORM
1 TRADE NAME OF THE MEDICINAL PRODUCT Ikorel 10mg and 20mg Tablet 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Nicorandil 10mg or 20mg For a full list of excipients, see section 6.1 3 PHARMACEUTICAL FORM
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert.
 PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. PROPRIETARY DRUG NAME / GENERIC DRUG NAME: Lyrica / Pregabalin
PFIZER INC. These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert. PROPRIETARY DRUG NAME / GENERIC DRUG NAME: Lyrica / Pregabalin
PHENTOLAMINE MESYLATE INJECTION SANDOZ STANDARD 5 mg/ ml THERAPEUTIC CLASSIFICATION Alpha-adrenoreceptor Blocker
 PACKAGE INSERT Pr PHENTOLAMINE MESYLATE INJECTION SANDOZ STANDARD 5 mg/ ml THERAPEUTIC CLASSIFICATION Alpha-adrenoreceptor Blocker ACTIONS AND CLINICAL PHARMACOLOGY Phentolamine produces an alpha-adrenergic
PACKAGE INSERT Pr PHENTOLAMINE MESYLATE INJECTION SANDOZ STANDARD 5 mg/ ml THERAPEUTIC CLASSIFICATION Alpha-adrenoreceptor Blocker ACTIONS AND CLINICAL PHARMACOLOGY Phentolamine produces an alpha-adrenergic
Supplementary Appendix
 Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Lembo AJ, Lacy BE, Zuckerman MJ, et al. Eluxadoline for irritable
Supplementary Appendix This appendix has been provided by the authors to give readers additional information about their work. Supplement to: Lembo AJ, Lacy BE, Zuckerman MJ, et al. Eluxadoline for irritable
Efficacy Study of Zoledronic Acid and Teriparatide Combination Therapy in Women With Osteoporosis
 A service of the U.S. National Institutes of Health Trial record 1 of 1 for: CZOL446H2409 Previous Study Return to List Next Study Efficacy Study of Zoledronic Acid and Combination Therapy in Women With
A service of the U.S. National Institutes of Health Trial record 1 of 1 for: CZOL446H2409 Previous Study Return to List Next Study Efficacy Study of Zoledronic Acid and Combination Therapy in Women With
ClinialTrials.gov Identifier: Sponsor/company: sanofi-aventis
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinialTrials.gov
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinialTrials.gov
