Instructions for Use ANGIOGUARD XP Emboli Capture Guidewire System
|
|
|
- Philomena Morrison
- 6 years ago
- Views:
Transcription
1 Instructions for Use ANGIOGUARD XP Emboli Capture Guidewire System 1
2 2 This page has been
3 This page has been 3
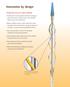
4 CAUTION: Federal (USA) law restricts this device to sale by or on the order of a physician. Read all instructions carefully. Failure to properly follow the instructions, warnings and precautions may lead to serious consequences or injury to the patient. STERILE. The ANGIOGUARD XP Emboli Capture Guidewire System is sterilized with ethylene oxide gas. Nonpyrogenic. Radiopaque. FOR ONE USE ONLY. DO NOT RESTERILIZE. Store in a cool, dark, dry place. 1.0 Device Name The device brand name is the ANGIOGUARD XP Emboli Capture Guidewire. 2.0 Description The ANGIOGUARD XP Emboli Capture Guidewire system consists of the following components: A 300 cm long,.014" disposable guidewire with an integrated deployable filter basket near the distal end to trap and capture emboli and a torque/locking device on its proximal end; A filter introducer; A deployment sheath; A capture sheath; and A guidewire introducer. The filter basket consists of a thin, porous membrane supported by a fine metal skeleton. The membrane contains numerous 100- micron pores to ensure adequate perfusion. Radiopaque markers are located proximal and distal to the filter basket to aid in placement of the device using fluoroscopy. Radiopaque markers are also located on every other strut of the wire filter basket just proximal to the filter material. The filter introducer component is used to flush the deployment sheath and to load the filter basket into the deployment sheath prior to use. The deployment sheath maintains the filter basket in the closed position. The torque component locks the sheath in place on the wire. The system is then inserted into the body. Upon reaching the anatomical site, the deployment sheath is removed and the filter deploys inside the arterial lumen. With the device in place, the carotid stenting procedure may then be performed. The separate capture sheath, which has a radiopaque marker at its distal end, is used to capture and retrieve the proximal (uncovered) portion of the filter basket. The capture sheath is advanced over the guidewire inside the patient. When the capture sheath s distal radiopaque marker band and the basket s proximal marker band touch, the basket is captured. The filter membrane is not captured by the sheath. The torque device (same one used for delivery) is connected to the capture sheath and tightened on the guidewire to ensure the filter basket with the trapped emboli remain captured when removing the guidewire/ capture sheath assembly from the patient. The following device schematic correlates to the preceding device description. The ANGIOGUARD XP Emboli Capture Guidewire is provided as noted in Table 1 below. Table 1 - ANGIOGUARD XP Emboli Capture Guidewire System CATALOG CATALOG GUIDEWIRE SYSTEM FILTER BASKET CODES CODES DIAMETER LENGTH DIAMETER Medium Extra (in) (cm) (mm) Support Support MC EC MC EC MC EC MC EC MC EC
5 The ANGIOGUARD TM XP Emboli Capture Guidewire is packaged as noted in Figures 1 and Indications for Use The ANGIOGUARD XP Emboli Capture Guidewire is indicated for use as a guidewire and embolic protection system to contain and remove embolic material (thrombus/debris) while performing carotid artery angioplasty and stenting procedures in carotid arteries. The diameter of the artery at the site of filter basket placement should be from 3 mm to 7.5 mm (see Section 9.3 of these instructions for basket/vessel sizing). 4.0 Contraindications Use of the ANGIOGUARD XP Emboli Capture Guidewire is contraindicated in the following patients: 1. Patients in whom antiplatelet and or anticoagulation therapy is contraindicated. 2. Patients in whom the guide catheter is unable to be placed. 3. Patients with uncorrected bleeding disorders. 4. Patients with known allergies to Nitinol. 5. Lesions in the ostium of the common carotid artery. 5.0 Warnings 5.1 General Warnings 1. Only physicians who have received appropriate training for carotid stenting and who are familiar with the principles, clinical applications, complications, side effects and hazards commonly associated with carotid interventional procedures should use this device. 2. The safety and efficacy of ANGIOGUARD have not been demonstrated with carotid stent systems other than the Cordis PRECISE stent. 3. Overstretching of the artery may result in rupture and life-threatening bleeding. 4. Patient ACT of >300 seconds needs to be maintained during ANGIOGUARD XP Emboli Capture Guidewire deployment. (See Section 9.1 of these instructions.) 5. The safety and effectiveness of this device as an emboli protection system has not been established in the coronary, cerebral, or peripheral vasculature, other than carotid arteries. 5.2 Patient Selection Warnings 6. Safety and effectiveness of the angioplasty and carotid stenting procedure has NOT yet been established in patients with the characteristics noted below. Lesion Characteristics: Patients with evidence of intraluminal thrombus thought to increase the risk of plaque fragmentation and distal embolization. Patients whose lesion(s) may require more than two stents. Patients with total occlusion of the target vessel. Patients with lesions of the ostium of the common carotid. Patients with highly calcified lesions resistant to PTA. Concurrent treatment of bilateral lesions. Patient Characteristics: Patients at low-to-moderate risk for adverse events from carotid endarterectomy. Patients experiencing acute ischemic neurologic stroke or who experienced a stroke within 48 hours. Patients with an intracranial mass lesion (i.e., abscess, tumor, or infection) or aneurysm (> 9 mm). Patients with arterio-venous malformations in the territory of the target carotid artery. Patients with coagulopathies. Patients with poor renal function, who, in the physician s opinion, may be at high risk for a reaction to contrast medium. Patients with perforated vessels evidenced by extravasation of contrast media. Patients with aneurysmal dilation immediately proximal or distal to the lesion. Pregnant patients or patients under the age of 18. Access Characteristics: Patients with known peripheral vascular, supra-aortic or internal carotid artery tortuosity that would preclude the use of catheter-based techniques. Patients in whom femoral or brachial access is not possible. 7. Risk of distal embolization may be higher if the ANGIOGUARD XP Emboli Capture Guidewire is not used during carotid stenting procedures. 5.3 Device Use Warnings 8. Do not use the device if there are abnormalities in the sterile barrier (e.g. broken seal, torn or breached barrier) or product. 9. This device is intended for one-time use only. Do not resterilize and/or reuse. Structural integrity and/or function may be impaired through reuse or cleaning. 10. Use the ANGIOGUARD XP Emboli Capture Guidewire prior to the "Use By" date specified on the package. 11. Observe all guidewire movement in the vessels using fluoroscopic guidance. 12. DO NOT TORQUE THE GUIDEWIRE. Do not torque a guidewire without observing corresponding movement of the tip; otherwise, vessel trauma could occur. Torquing a guidewire against resistance may cause guidewire damage and/or guidewire tip separation. Always advance or withdraw the guidewire slowly. Never push, auger, withdraw or torque a guidewire that meets resistance. Resistance may be felt and/or observed using fluoroscopy by noting any buckling of the guidewire tip. Determine the cause of resistance under fluoroscopy and take the necessary remedial action. 13.Before the guidewire is moved, tip movement should be examined using fluoroscopy. 14. Perform all exchanges slowly to prevent air from entering the catheter system. 15.When introducing the guidewire, confirm that the guiding catheter or interventional sheath introducer tip is free within the vessel lumen and not against the vessel wall. 5
6 Failure to do so may result in vessel trauma upon guidewire exit from the tip. Use the radiopaque marker of the interventional device to confirm position. 6.0 Precautions 6.1 Pre-Procedure Precautions 1. The ANGIOGUARD TM XP Emboli Capture Guidewire is supplied STERILE and is intended for single use only. Do not re-sterilize and/or reuse the device. 2. Use the ANGIOGUARD XP Emboli Capture Guidewire System prior to the "Use By" date printed on the package. 3. Guidewires are delicate instruments and should be handled carefully. Prior to use, and when possible during the procedure, inspect the guidewire carefully for coil separation, bends, kinks, or damage of the filter basket assembly. 4. Do not use if opened or damaged. 5. Confirm the compatibility of the ANGIOGUARD XP Emboli Capture Guidewire with the interventional device before actual use. 6.2 Device Use Precautions 6. If distal perfusion of dye is significantly reduced or no dye is perfusing past the distal marker band of the filter basket, the ANGIOGUARD XP Emboli Capture Guidewire may have reached its maximum capacity to contain emboli. Remove and replace with a new ANGIOGUARD XP system per Section 11.0 of these instructions. 7. Do not attempt to close the filter basket with the Deployment Sheath. The ANGIOGUARD XP Emboli Capture Guidewire should only be removed using the Capture Sheath. 8. Care during diagnostic or interventional device exchanges must be practiced to minimize movement of the guidewire/filter basket. 9. Use caution when withdrawing ANGIOGUARD XP Emboli Capture Guidewire through the deployed stent. 6
7 7.0 Adverse Events 7.1 Observed Adverse Events Carotid stenting with distal protection was conducted on a total of 573 patients with carotid artery disease who were at high risk for adverse events from carotid endarterectomy (CEA) in the SAPPHIRE clinical study (Stenting and Angioplasty with Protection in Patients at High Risk for Endarterectomy). The study was conducted to evaluate the safety and effectiveness of the Cordis PRECISE Nitinol Stent System and the ANGIOGUARD TM XP Emboli Capture Guidewire. The study included a randomized arm that compared stent patients to CEA patients (334 patients). The study also included a non-randomized stent arm for patients who met the same entry criteria as the randomized patients, but who were determined by the surgeon at the study site to be at too high a risk for adverse events from carotid endarterectomy (406 patients). Patients meeting the same inclusion criteria as the randomized patients, but determined by the interventionalist to be inappropriate for stent treatment, were entered into a non-randomized surgical arm (7 patients). The major adverse event (MAE) rate in all study arms was defined as death, stroke, or MI (Q-wave or non-q-wave) to 30 days and death or ipsilateral stroke from 31 days to 360 days. Only 7 patients were enrolled in the non-randomized surgical arm of the SAPPHIRE study. The 360-day MAE rate for these patients was 14.3%. Thirty-day complications in the SAPPHIRE Randomized Study arm (167 stent patients vs. 167 CEA patients) and the non-randomized stent arm (406 patients) are shown in Table 2. Differences in 30-day event rates in-hospital vs. out-of-hospital for major adverse events (MAE) and Transient Ischemic Attack (TIA) are also provided in Table 2, which follows. All acute bradycardia and hypotensive events occurred in-hospital. Differences in 30- day event rates observed in-hospital vs. out-of-hospital for MAE and TIA are also provided in Table 2, which follows. Table 2 - Randomized & Non-Randomized Patient Events to 30 Days 30-Day Complications Randomized Stent Randomized CEA Non-Randomized (In-Hospital vs. Out-of-Hospital) (N=167) (N=167) Stent (N=406) In-Hosp Out-of-Hosp In-Hosp Out-of-Hosp In-Hosp Out-of-Hosp MAE 4.2% (7) 1.2% (2) 7.2% (12) 3.0% (5) 3.2% (13) 3.7% (15) Death (All Cause) 0.0% (0) 1.2% (2) 1.2% (2) 1.2% (2) 1.2% (5) 1.0% (4) Myocardial Infarction (Q or Non-Q) 2.4% (4) 0.0% (0) 4.8% (8) 1.2% (2) 1.0% (4) 0.7% (3) Stroke 3.6% (6) 0.0% (0) 2.4% (4) 0.6% (1) 2.5% (10) 2.5% (10) Transient Ischemic Attack (TIA) 3.6% (6) 0.0% (0) 2.4% (4) 0.0% (0) 3.2% (13) 2.2% (9) Randomized Stent Randomized CEA Non-Randomized 30-Day Complications (N=167) (N=167) P-value* Stent (N=406) Death (All Cause), Any Stroke or MI 4.8% (8) 9.6% (16) % (28) Death (All Cause) 1.2% (2) 2.4% (4) % (9) Myocardial Infarction (Q or Non-Q) 2.4% (4) 6.0% (10) % (7) Q Wave MI 0.0% (0) 1.2% (2) % (1) Non-Q Wave MI 2.4% (4) 4.8% (8) % (6) Stroke 3.6% (6) 3.0% (5) > % (20) Major Ipsilateral Stroke 0.6% (1) 1.2% (2) > % (10) Major Non-Ipsilateral Stroke 0.6% (1) 0.6% (1) > % (2) Minor Ipsilateral Stroke 2.4% (4) 0.6% (1) % (7) Minor Non-Ipsilateral Stroke 0.6% (1) 0.6% (1) > % (2) Transient Ischemic Attack (TIA) 3.6% (6) 2.4% (4) % (22) Target Lesion Revascularization 0.0% (0) 0.0% (0) - 0.5% (2) Surgery 0.0% (0) 0.0% (0) - 0.0% (0) PTA 0.0% (0) 0.0% (0) - 0.5% (2) Target Vessel Revascularization (not involving the Target Lesion) 0.0% (0) 0.0% (0) - 0.0% (0) Surgery 0.0% (0) 0.0% (0) - 0.0% (0) PTA 0.0% (0) 0.0% (0) - 0.0% (0) Major Bleeding 1 9.0% (15) 10.2% (17) % (52) Cranial Nerve Injury 0.0% (0) 4.2% (7) % (0) Severe Hypotension 17.4% (29) 3.0% (5) < % (61) Bradycardia 8.4% (14) 3.0% (5) % (13) Vascular Complications 2 5.4% (9) N/A - 2.5% (10) Device/Procedure Related Adverse Events 3 0.0% (0) % (0) * P-value displayed refers to comparison of randomized arms. (1) Major Bleeding: Any non-access site-related bleeding resulting in a 25% or more decline in HCT or requiring transfusion. (2) Vascular Complications: Events related to bleeding or vascular injury at the percutaneous access site. (3) There were no device or procedure related events. In 17 of 19 initial stent delivery failures, a subsequent attempt was successful. In one case, the patient was treated with CEA. In the other case, the patient was treated with balloon angioplasty alone. One stent fracture was noted from one-year ultrasound films, with no adverse effect to the patient. 7
8 8 7.2 Potential Adverse Events Adverse events that may be associated with the use of the ANGIOGUARD TM XP Emboli Capture Guidewire, in conjunction with the Cordis PRECISE Nitinol Stent System include, but may not be limited to: air embolism allergic/anaphylactoid reaction aneurysm angina/coronary ischemia arrhythmia (including bradycardia, possibly requiring need for a temporary or permanent pacemaker) arterial occlusion/restenosis of the treated vessel arterial occlusion/thrombus, at puncture site arterial occlusion/thrombus, remote from puncture site arteriovenous fistula bacteremia or septicemia cerebral edema damage to implanted stent(s) death embolization, arterial emergent repeat hospital intervention fever GI bleeding from anticoagulation/antiplatelet medication hematoma bleed, puncture site hematoma bleed, remote site hemorrhage hyperperfusion syndrome hypotension/hypertension infection intimal injury/dissection ischemia/infarction of tissue/organ local infection and pain at insertion site malposition (failure to deploy filter basket at intended site) myocardial infarction pain pseudoaneurysm renal failure seizure severe unilateral headache stroke transient ischemic attack vasospasm venous occlusion/thrombosis, at puncture site venous occlusion/thrombosis, remote from puncture site vessel rupture, dissection, perforation 7.3 Device Related Adverse Event Reporting Any adverse event (clinical incident) involving the ANGIOGUARD XP Emboli Capture Guidewire should be reported to Cordis Corporation immediately. To report an incident, call the Product Quality Services Department at , option Clinical Study Information 8.1 Objectives The primary objective of the pivotal clinical study (SAPPHIRE) was to compare the safety and effectiveness of the Cordis PRECISE Nitinol Stent Systems, used in conjunction with the ANGIOGUARD XP Emboli Capture Guidewire, to carotid endarterectomy (CEA) in the treatment of carotid artery disease in patients at increased risk for adverse events from CEA. Study hypotheses examined whether the major adverse events (MAE) rate of randomized stent patients was not inferior to randomized CEA patients. Safety evaluations included assessments of major clinical events occurring during the procedure, prior to discharge, within 30 days, six months, one year and every 12 months thereafter for a total of three years; access site vascular complications; independent neurological assessments at 24 hours, 30 days, six months and one year post procedure. Effectiveness evaluations included assessments of successful stent deployment at the target lesion; less than 30% residual diameter stenosis at the completion of the procedure as measured by carotid angiography; and restenosis (> 50%) as determined by carotid ultrasound at 30 days, six months and one year post procedure and every 12 months thereafter for a total of three years. 8.2 Study Design The pivotal SAPPHIRE study was a multi-center, prospective, randomized, triangular sequential trial comparing patients at increased risk for adverse events from CEA who received a stent to a surgical (CEA) control. The safety and effectiveness of the Cordis PRECISE Nitinol Stent System, used in conjunction with the ANGIOGUARD XP Emboli Capture Guidewire in the treatment of de novo or restenotic obstructive carotid artery disease in these patients, was evaluated. The study also included a non-randomized stent arm, which included those patients who met entry criteria but who were determined by the surgeon at the study site to be at too high a risk for adverse outcomes from surgery and therefore inappropriate for randomization. Likewise, patients meeting the entry criteria, but determined by the interventionalist to be unacceptable candidates for stenting and therefore not randomizable, had the option of entering a non-randomized surgical arm. SAPPHIRE entry criteria were identical for all patients. All patients were evaluated to determine whether they met the entry criteria by a multi-disciplinary team consisting of a neurologist, interventionalist, and vascular surgeon. Patients meeting the criteria were either randomized to treatment by stent or CEA, or placed into the non-randomized stent or CEA arms, based on the medical judgment of the interventionalist and surgeon as noted above. Patients who were entered into this study were either asymptomatic with a > 80% diameter stenosis or symptomatic with a > 50% diameter stenosis. Symptomatic patients were defined as those patients who have one or more TIAs, characterized by distinct focal neurological dysfunction or monocular blindness with clearing of signs and symptoms within 24 hours or one or more completed strokes with persistence of symptoms or signs for more than 24 hours. In addition, ALL patients must also have had at least one anatomic or co-morbid risk factor placing them at high-risk for adverse events from CEA. These risk factors are as follows: Congestive Heart Failure (Class III/IV), and/or known severe left ventricular dysfunction <30% Open-heart surgery within 6 weeks Recent myocardial infarction (>24 hours and <4 weeks) Unstable angina (CCS class III/IV) Synchronous severe cardiac and carotid disease requiring open heart surgery and carotid revascularization Severe pulmonary disease to include any of the following: - Chronic oxygen therapy - Resting P02 of < 60 mmhg - Baseline hematocrit > 50% - FEV1 or DLCO < 50% of normal Contralateral carotid occlusion Contralateral laryngeal palsy Post-radiation treatment Previous CEA recurrent stenosis High cervical ICA lesions CCA lesions below the clavicle Severe tandem lesions Abnormal stress test The primary endpoint was a composite of MAE including death, any stroke, or myocardial infarction (MI), in the first 30 days following treatment and death or ipsilateral stroke between 31 days and 12 months. An independent Clinical Events Committee adjudicated all MAE s and other events. Endpoints were analyzed on an intent-to-treat basis. A total of 747 patients were enrolled in the SAPPHIRE study at 29 centers in the United States. The randomized population included 334 patients (167 stent/167 CEA), 310 of who were treated per protocol. The primary reasons why the remaining 24 patients were not treated were: 1) Eleven patients withdrew consent; 2) Six patients were found not to meet inclusion criteria subsequent to randomization; 3) Five patients conditions deteriorated and they became too high a risk for any treatment; and 4) Two patients were randomized to surgery that was never performed. The non-randomized stent arm included 406 patients and the non-randomized CEA arm included seven patients.
9 Follow-up evaluations were scheduled at 30 days, six months and one year post-procedure, and annually thereafter for three years. Imaging data provided in this summary are based on findings from two independent centralized Core Laboratories, which reviewed ultrasound and angiographic films. A third independent laboratory analyzed trapped material contained in a percentage of all ANGIOGUARD TM XP filter baskets. A Clinical Events Committee (CEC) adjudicated all clinical events and an independent Data Safety Monitoring Board (DSMB) monitored safety. 8.3 Patient Demographics Table 3 provides the subject characteristics of randomized patients and non-randomized stent patients enrolled in the SAPPHIRE trial. Table 3 - SAPPHIRE Patient Demographics* Non-Randomized Patient Characteristics Randomized Stent Randomized CEA P-value** Stent Age (Years) % Male 66.9% (111/166) 67.1% (108/161) % (261/406) Diabetes 25.3% (42/166) 27.5% (44/160) % (125/406) Coronary Artery Disease 85.8% (133/155) 75.5% (111/147) % (259/376) Previous PTCA (Coronary) 34.8% (56/161) 23.4% (37/158) % (83/392) Previous CABG 43.4% (72/166) 30.8% (49/159) % (128/406) Previous Q-Wave or Non-Q-Wave MI 29.7% (46/155) 35.3% (54/153) % (122/365) Angina at a Low Workload or Unstable Angina 24.1% (20/83) 14.7% (11/75) % (41/130) Congestive Heart Failure 17.5% (29/166) 17.4% (28/161) % (74/406) Coexistent Severe Coronary Artery Disease 15.9% (26/164) 16.5% (26/158) % (51/400) Requiring Carotid and Coronary Revascularization Systolic Blood Pressure History of Dyslipidemia 78.5% (128/163) 76.9% (123/160) % (289/391) Previous CEA/Recurrent Stenosis 22.6% (37/164) 22.2% (35/158) % (151/401) Post-Radiation Treatment 4.3% (7/164) 5.7% (9/158) % (64/401) Prior CEA 28.3% (47/166) 26.7% (43/161) % (183/405) Contralateral Carotid Occlusion 23.6% (39/165) 25.3% (40/158) % (65/400) History of Stroke 27.1% (45/166) 23.8% (38/160) % (129/399) History of TIA 31.1% (50/161) 34.0% (53/156) % (138/400) High Cervical ICA Lesions 4.3% (7/164) 4.4% (7/158) % (51/401) CCA Lesions Below the Clavicle 0.0% (0/164) 0.0% (0/158) - 3.0% (12/401) Other Co-morbid Risk Factors Precluding CEA 0.0% (0/164) 0.0% (0/160) - 7.9% (32/404) Renal Insufficiency 6.0% (10/166) 7.5% (12/160) % (30/405) Current Cigarette Use 16.9% (27/160) 16.4% (26/159) % (54/399) Patients >80 years 19.3% (32/166) 20.5% (33/161) % (78/406) * The denominator represents the total number of responses to a question in the case report form. **P-value displayed refers to comparison of randomized arms. 8.4 Study Results The 360-day major adverse events (MAE) rate, defined as death, stroke, or MI (Q-wave or non-q-wave) to 30 days and death or ipsilateral stroke from 31 days to 360 days was 12.0% for the randomized stent patients compared with 19.2% for the control group. These results demonstrate noninferiority (p=0.004) of carotid stenting to carotid endarterectomy (CEA) with the pre-specified non-inferiority delta of 3%. The MAE rate at 360 days for the non-randomized stent patients was 15.8%. In a test of the primary endpoint against the Objective Performance Criteria (OPC), despite the fact that the rate was numerically less than the OPC plus the delta, the p- value was found to be In a test of the MAE rate when post 30-day non-neurological deaths are not included, the p-value was found to be < The causes of these non-neurological deaths are well documented, and consist of cardiac deaths, cancer deaths, renal failure, and respiratory failure. A comparison of the non-randomized stent arm and the randomized CEA arm was conducted utilizing a propensity score analysis that accounted for baseline imbalances due to the non-randomized (i.e., more observational) nature of group membership. The analysis found the treatment difference (non-randomized stent minus CEA) in 360-day MAE was 5.3%, with an adjusted 95% confidence interval of 13.4% to 3.0%. Thus, after adjusting for the higher risk of patients in the non-randomized stent arm, 360-day MAE outcomes were non-inferior to the CEA arm of the randomized study within a 3% delta. Principal safety and effectiveness results to 360 days are presented in Table 4, which follows. The cumulative percentage of MAE through 360-days for the randomized and non-randomized stent patients is presented in Figure 3, which follows. Non-randomized CEA patient event rates are not provided in Table 4 since only seven patients were enrolled in that study arm and the data are insufficient for statistical analysis. For informational purposes, the MAE rate for non-randomized CEA patients to 360 days was 14.3% (1/7). Figures 4 and 5 present the cumulative percentage of MAE through 360 days for randomized asymptomatic and symptomatic patients. 9
10 Table 4 - Principal Safety & Effectiveness Results To 360 Days (Intent to Treat) Safety Measures & Other Clinical Events Randomized Stent Randomized CEA P-value* Non-Randomized to 360 Days (N=167) (N=167) Stent (N=406) MAE % (20/167) 19.2%(32/167) % (64/406) Death (All Cause) 7.2% (12/167) 12.6% (21/167) % (41/406) Stroke 6.0% (10/167) 7.2% (12/167) % (37/406) Major Ipsilateral Stroke 0.6% (1/167) 3.0%(5/167) % (13/406) Minor Ipsilateral Stroke 3.6% (6/167) 1.8% (3/167) % (16/406) Myocardial Infarction (Q or Non-Q) 3.0% (5/167) 7.2% (12/167) % (11/406) TIA 6.6% (11/167) 3.0% (5/167) % (28/406) Major Bleeding 2 9.0% (15/167) 10.2% (17/167) % (54/406) Cranial Nerve Injury 0.0% (0/167) 4.8% (8/167) % (0/406) Severe Hypotension 17.4% (29/167) 3.0% (5/167) < % (63/406) Bradycardia 8.4% (14/167) 3.0% (5/167) % (14/406) Vascular Complications 3 5.4% (9/167) N/A - 2.5% (10/406) Device/Procedure Related Adverse Events 4 0.0% (0) 0.0% (0) - 0.0% (0) Efficacy Measures Randomized Randomized P-Value* Non-Randomized Stent (N=167) CEA (N=167) Stent (N=406) Lesion Success % (145/158) N/A N/A 90.4% (368/407) Procedure Success % (140/159) N/A N/A 87.9% (355/404) Device Success % (145/159) N/A N/A 89.6% (363/405) ANGIOGUARD TM Success % (152/159) N/A N/A 91.6% (372/406) Post-Procedure In-Lesion Minimal Lumen Diameter (MLD in mm) Mean+SD (N) (147) (385) Range (min, max) (2.1,7.3) N/A N/A (2.0, 8.1) Post-Procedure In-Lesion Percent Diameter Stenosis (%DS) 9 Mean+SD (N) (147) (385) Range (min, max) (1.5, 49.3) N/A N/A (-12.1, 64.7) Post-Procedure In-Stent Minimal Lumen Diameter (MLD in mm) Mean+SD (N) (147) (381) Range (min, max) (2.1, 7.9) N/A N/A (2.2, 8.1) Post-Procedure In-Stent Percent Diameter Stenosis (%DS) 10 Mean+SD (N) (147) (381) Range (min, max) (-42.0, 46.6) N/A N/A (-34.9, 43.8) Binary Ultrasound In-Vessel Restenosis at 360 days % (24/122) 31.3% (30/96) % (78/282) Binary Ultrasound In-Stent Restenosis at 360 days % (19/122) 13.5% (13/96) % (52/282) Cumulative % of TLR at 360 days** % 4.3% % Cumulative % of MAE 1 at 360 days** 12.2% 20.1% % Numbers are % (counts/sample size). *P-value displayed refers to comparison of randomized arms. **Cumulative percentage estimates are by Kaplan-Meier methods with standard error estimates by Peto formula. (1) Major Adverse Events (MAE) = Death, MI or stroke to 30 days and death or ipsilateral stroke from days. (2) Major Bleeding = Any non-access site related bleeding resulting in a 25% or more decline in HCT or requiring transfusion. (3) Vascular Complications = Events related to bleeding or vascular injury at the percutaneous access site. (4) There were no device or procedure related events. In 17 of 19 initial stent delivery failures, a subsequent attempt was successful. In one case, the patient was treated with CEA. In the other case, the patient was treated with balloon angioplasty alone. One stent fracture was noted from one-year ultrasound films, with no adverse effect to the patient. (5) Lesion Success = The attainment of a final residual stenosis of <30% using any percutaneous method. If no in-stent measurements were available, in-lesion measurements were used, and if no QCA was available, visual estimates were used. (6) Procedure Success = The attainment of a final residual stenosis of <30% and no in-hospital MAE. If no in-stent measurements were available, in-lesion measurements were used, and if no QCA was available, visual estimates were used. (7) Device Success = The attainment of a final residual stenosis of <30% using only the assigned device. If no in-stent measurements were available, in-lesion measurements were used, and if no QCA was available, visual estimates were used. (8) ANGIOGUARD TM Success = Successful deployment and retrieval of the ANGIOGUARD TM device. (9) In-lesion % DS Measurement = Defined as the % diameter stenosis either within the stented segment or within 5 mm proximal or distal to the stent edges. (10) In-stent % DS Measurement = Defined as the % diameter stenosis within the stented segment. (11) Binary Restenosis is defined by Ultrasound as % diameter stenosis >50%. (12) TLR = Target Lesion Revascularization 10
11 Figure 3 Cumulative Percentage of MAE* at 360 days Stent Time After Procedure (Days) Randomized Patients N at risk % with events CEA N at risk % with events Test Between Groups Log-Rank P-Values Time After Procedure (Days) Non-Randomized Patients Stent N at risk % with events * Major Adverse Events (MAE) = Death, MI or stroke to 30 days and death or ipsilateral stroke from days. Figure 4 Cumulative Percentage of MAE* at 360 days Asymptomatic Randomized Stent and CEA Patients Time After Procedure (Days) Stent N at risk % with events CEA N at risk % with events Test Between Groups Log-Rank P-Value * Major Adverse Events (MAE) = Death, MI or stroke to 30 days and death or ipsilateral stroke from days. 11
12 Figure 5 Cumulative Percentage of MAE* at 360 Days Symptomatic Randomized Stent & CEA Patients Time After Procedure (Days) Stent N at risk % with events CEA N at risk % with events Test Between Groups Log-Rank P-Values * Major Adverse Events (MAE) = Death, MI or stroke to 30 days and death or ipsilateral stroke from days. Basket Content Analysis - A pathology core lab analyzed the contents of 294 ANGIOGUARD TM XP filter baskets from the non-randomized stent arm of the SAPPHIRE trial and determined that 59.5% (175/294) of the baskets contained material that had been captured during the carotid stenting procedure. Physicians reported that there was visible material present in 56% of the 393 baskets inspected in the non-randomized stent arm and in 72.2% of the 158 baskets inspected in the randomized stent arm. 30-Day Stroke Rate of Carotid Artery Stenting with and without ANGIOGUARD XP Emboli Capture Guidewire - Two non-randomized studies utilizing the PRECISE stent (and its predecessor, the S.M.A.R.T. stent) were conducted in Europe (CASCADE) and in the United States (U.S. Feasibility Study). Thirty-one (31) of 131 patients in CASCADE and 85 of 261 patients in the U.S. Feasibility study were treated with stenting in conjunction with the ANGIOGUARD embolic protection device while the remaining patients were treated with stenting alone. Because the number of patients in each trial is small, an exploratory analysis was performed in which the data for 30- day stroke from these two trials were combined. 30-day stroke rates for patients treated by stenting alone and for patients treated by stenting with distal embolic protection were analyzed in a post-hoc analysis; combined rates of 30- day stroke were 8.6% for the 266 patients treated with stenting alone and 2.6% for the 116 patients treated by stenting with distal embolic protection. In this analysis, the difference between these two rates (an absolute reduction of 6% and a relative reduction of 70%) is significant with a p- value of Day Stroke Rate of Carotid Artery Stenting with and without the ANGIOGUARD XP Emboli Capture Guidewire TM TM %
13 9.0 Directions for Use Only physicians who have received appropriate training for carotid stenting and who are familiar with the principles, clinical applications, complications, side effects and hazards commonly associated with carotid interventional procedures should use this device. 9.1 Peri-Procedural Care It is recommended that heparin (intravenous) be given during the procedure immediately after guiding catheter cannulation. The initial bolus dose of heparin should be approximately 3,000 to 5,000 units (with necessary weight adjustments). Additional bolus doses of heparin should be given to maintain an ACT near 300 seconds during the entire procedure. No heparin should be given after the procedure until hemostasis at the puncture site is achieved. 9.2 Pre-Procedure The percutaneous placement of the ANGIOGUARD TM XP Emboli Capture Guidewire in a stenotic or obstructed carotid artery should be done in an angiography procedure room. Angiography should be performed to map out the extent of the lesion(s) and the collateral flow. If thrombus is present, do not proceed. Access vessels must be sufficiently patent or sufficiently recanalized, to proceed with further intervention. Patient preparation and sterile precautions should be the same as for any angioplasty procedure. a) Inject contrast media Perform a percutaneous angiogram using standard technique. b) Identify and mark the lesion Fluoroscopically identify and mark the lesion, observing the most distal level of the stenosis. 9.3 Device Selection and Preparation CAUTION: The ANGIOGUARD XP Emboli Capture Guidewire is supplied STERILE and is intended for single use only. DO NOT resterilize and/or reuse the device. Do not use if opened or damaged. CAUTION: Use the ANGIOGUARD XP Emboli Capture Guidewire system prior to the "Use By" date printed on the package. CAUTION: Guidewires are delicate instruments and should be handled carefully. Prior to use, and when possible during the procedure, inspect the guidewire carefully for coil separation, bends, kinks, or damage of the filter basket assembly. Do not use defective equipment. CAUTION: Confirm the compatibility of the ANGIOGUARD XP Emboli Capture Guidewire with the interventional device before actual use. Select the appropriate diameter filter basket in accordance with Table 5, which follows. Table 5 Filter Basket Sizing Recommendation Nominal Filter Basket Size Recommended Vessel Size 4.0 mm 3.0 mm < Vessel < 3.5 mm 5.0 mm 3.5 mm < Vessel < 4.5 mm 6.0 mm 4.5 mm < Vessel < 5.5 mm 7.0 mm 5.5 mm < Vessel < 6.5 mm 8.0 mm 6.5 mm < Vessel < 7.5 mm 2. During shipping, the deployment sheath tip may become disengaged from the filter basket introducer. Verify that the deployment sheath tip is engaged. If not, engage manually by inserting the deployment sheath tip into the filter basket introducer. 3. Fill a 5 ml Luer lock syringe with sterile saline and purge all air. 4. Attach syringe to Luer lock hub on the end of the filter introducer. 5. Inject 5 ml of saline to purge all air from the deployment sheath and filter basket (ensure distal tip of the deployment sheath is inside the filter basket introducer tip prior to purging the system). You should see saline dripping from the proximal end of the deployment sheath (inside the coil dispenser). 6. Disconnect syringe. 7. Remove the two proximal (closest to the torque device) anti-migration clips holding the guidewire in the dispenser coil. 8. Ensure the torque device is locked onto the guidewire. 9. Gripping the torque device in one hand and the coil dispenser in the other, pull on the wire until the basket is completely docked into the tip of the deployment sheath. When completely docked, approximately half the filter basket will still be visible out of the end of the deployment sheath. 10.After the deployment sheath is completely docked, remove the remaining distal anti-migration clip (closest to the filter introducer) and continue to pull back on the torque device to disengage the docked deployment sheath/guidewire assembly from the filter introducer. 11.Loosen the torque device. 12.While holding the torque device in one hand, gradually pull back on the guidewire with the other hand. Continue to pull the guidewire through the torque device until the proximal end of the deployment sheath is visible at the proximal end of the torque device. 13.Tighten the torque device onto the deployment sheath to secure the deployment sheath to the guidewire. NOTE: If desired, the torque device can be carefully moved to another position on the sheath and retightened. 14.The filter basket, now loaded inside the deployment sheath tip and secured to the guidewire with the torque device, can be completely removed from the coil dispenser. 15.The device is now prepped and ready for use. 16.Capture Sheath After the interventional or diagnostic procedure is complete, attach a 5 ml Luer lock syringe, prepared as described in step 3 of this section, to the capture sheath hub and flush with 5 ml of sterile saline. You should see saline dripping out of the other end of the capture sheath (inside the coil dispenser). Remove the syringe and then remove the capture sheath from the coil dispenser. The capture sheath is now ready for use. 9.4 Filter Deployment and Capture Procedure WARNING: Patient ACT of >300 seconds needs to be maintained during ANGIOGUARD XP Emboli Capture Guidewire deployment. (See Section 9.1 of these instructions.) WARNING: Observe all guidewire movement in the vessels using fluoroscopic guidance. WARNING: Before the guidewire is moved, the tip movement should be examined using fluoroscopy. 1. The packaging contains two plastic coil-tubing dispensers (see Figures 1 and 2). One contains the Guidewire with Filter Basket, Filter Basket Introducer, Peel-away Guidewire Introducer, Torque Device, and yellow Deployment Sheath. The second contains the blue Capture Sheath, which will not be needed until the filter basket is ready to be removed from the vessel. Place the Capture Sheath aside until needed. 13
14 14 WARNING: DO NOT TORQUE THE GUIDEWIRE. Do not torque a guidewire without observing corresponding movement of the tip; otherwise, vessel trauma could occur. Torquing a guidewire against resistance may cause guidewire damage and/or guidewire tip separation. Always advance or withdraw the guidewire slowly. Never push, auger, withdraw or torque a guidewire that meets resistance. Resistance may be felt and/or observed using fluoroscopy by noting any buckling of the guidewire tip. Determine the cause of resistance under fluoroscopy and take the necessary remedial action. 1. Insert the peel-away guidewire introducer into the interventional sheath introducer or the hemostatic valve of the Y connector attached to the guiding catheter. Ensure the hemostatic valve is fully opened before inserting the guidewire introducer. 2. Carefully insert the deployment sheath/guidewire assembly through the peel-away guidewire introducer and into the guiding catheter or the interventional sheath introducer. An 8F (2.7 mm) with a.088" minimum ID guiding catheter or a 6F (2.0 mm) interventional sheath introducer is recommended for ease of removal. WARNING: When introducing the guidewire, confirm that the guiding catheter or interventional sheath introducer tip is free within the vessel lumen and not against the vessel wall. Failure to do so may result in vessel trauma upon guidewire exit from the tip. Use the radiopaque marker of the interventional device to confirm position. 3. To remove the peel-away guidewire introducer, retract the peel-away guidewire introducer from the interventional sheath introducer or hemostatic valve of the Y connector attached to the guiding catheter. Snap open the peel-away guidewire introducer hub and peel apart the entire introducer shaft. 4. Advance the guidewire through the guide catheter or interventional sheath introducer until it is proximal to the tip of the guiding catheter or interventional sheath. 5. Tighten the hemostatic valve slightly to reduce the flow of blood from around the deployment sheath/guidewire assembly. Ensure that movement of the guidewire is still possible. 6. Using fluoroscopy, advance the deployment sheath/ guidewire assembly out further from the guide catheter or interventional sheath introducer. Use the torque device to steer the guidewire across the lesion. NOTE: With reference to step 7 of this section, allow sufficient space between the lesion and filter basket to prevent interference with the distal tips of all other diagnostic and interventional devices to be used throughout the procedure. 7. Position the filter basket of the ANGIOGUARD TM XP Emboli Capture Guidewire so both distal and proximal radiopaque markers are distal to the lesion to be treated. 8. After achieving the appropriate filter basket position in the vessel, remove the torque device from the deployment sheath/guidewire assembly. NOTE: With reference to step 9 of this section, always maintain filter basket position during deployment sheath removal. NOTE: With reference to step 9 of this section, never attempt to recapture the filter basket using the deployment sheath. If repositioning of the device is necessary, use the capture sheath and reposition as described in step 14 of this section. NOTE: With reference to step 9 of this section, the distal tip of the deployment sheath is larger in diameter than the sheath body. The hemovalve may need to be adjusted to allow clearance for the larger distal tip. 9. While maintaining the deployment sheath/guidewire assembly position, deploy the filter basket by sliding the deployment sheath proximally. Making sure to maintain the guidewire position, continue to remove the deployment sheath by sliding the sheath proximally until it is totally removed from the guidewire. 10.Close the hemovalve. 11.The filter basket of the ANGIOGUARD XP Emboli Capture Guidewire is now fully deployed within the vessel. Confirm full deployment by fluoroscopy. The marker bands on the filter basket struts should be fully apposed to the vessel wall. 12.Using fluoroscopy, inject dye to determine that there is adequate flow distal to the filter basket (or distal band of the guidewire) and that the guidewire remains in the proper position. 13.Once filter basket position is confirmed,.014 in. (0.36 mm) compatible interventional devices can be loaded through the open hemovalve on to the guidewire for treatment of the lesion. Use the standard technique of over the guidewire or single operator exchange while treating the lesion. Be careful not to move the filter basket during exchanges. CAUTION: With reference to step 14 of this section, never attempt to close the filter basket with the deployment sheath. The ANGIOGUARD XP Emboli Capture Guidewire should only be removed using the Capture Sheath. 14.Once the lesion is treated and all of the interventional or diagnostic devices have been removed, thread the prepped (per step 16 of section 9.3) capture sheath over the proximal end of the guidewire. Open the hemovalve and advance the capture sheath until the distal marker on the sheath lines up with the proximal marker on the filter basket. This will close the filter basket. CAUTION: Use caution when withdrawing the ANGIOGUARD XP Emboli Capture Guidewire through the deployed stent. NOTE: With reference to step 15 of this section, do not try removing the captured system by only pulling on the capture sheath. NOTE: With reference to step 15 of this section, never pull the captured system into the guiding catheter or interventional sheath introducer if there is resistance. If resistance is encountered, reposition the capture sheath to ensure that the filter basket is properly seated into the capture sheath. Using fluoroscopy, verify capture sheath position by checking marker band alignment between the capture sheath and guidewire and confirm filter basket closure by ensuring reduction of the filter basket diameter shown by the radiopaque strut markers. 15.Place the torque device over the guidewire and attach the Luer lock to the capture sheath hub. With the torque device locked to the capture sheath, tighten the torque device to the guidewire. Then remove the system by pulling the guidewire proximal to the Capture Sheath hub through the guide catheter or interventional sheath introducer and out of the hemostasis valve as a single unit. Care should be taken when pulling the captured basket through the open hemovalve to avoid potential release of captured emboli within the guiding catheter How to Detect if the ANGIOGUARD XP Emboli Capture Guidewire is Full 1. Once the ANGIOGUARD XP Emboli Capture Guidewire is deployed in the vessel, it may capture emboli during the entire time of the interventional procedure. Therefore, it is recommended to check the status of the ANGIOGUARD XP Emboli Capture Guidewire during regular intervals during the intervention. 2. Using fluoroscopy, perform a distal dye injection through the guide catheter or interventional sheath introducer and observe the flow of dye distal to the filter basket or distal marker on the guidewire.
15 CAUTION: If the distal perfusion of dye is significantly reduced or no dye is perfusing past the filter basket or distal marker band, the ANGIOGUARD TM XP Emboli Capture Guidewire may have reached its capacity to contain emboli. If there is a severe reduction in distal dye perfusion, it is recommended to exchange the ANGIOGUARD XP Emboli Capture Guidewire for a new one How to Exchange a Full ANGIOGUARD XP Emboli Capture Guidewire WARNING: Perform all exchanges slowly to prevent air from entering the catheter system. CAUTION: The ANGIOGUARD XP Emboli Capture Guidewire should only be removed using the Capture Sheath and should be removed through the guide catheter or interventional sheath introducer only. See Section 9.4, steps 14 and 15 of these instructions for proper filter basket capture and system removal. To exchange an ANGIOGUARD XP Emboli Capture Guidewire that has captured its capacity of emboli: 1. Remove all interventional devices from the ANGIOGUARD XP Emboli Capture Guidewire. 2. Prep the Capture Sheath as outlined in Section 9.3, step 16 of these instructions. 3. Back-load the Capture Sheath over the proximal end of the ANGIOGUARD XP Emboli Capture Guidewire. Advance the Capture Sheath until the distal marker on the sheath lines up with the proximal marker on the guidewire as described in Section 9.4, step 14 of these instructions. 4. After the Capture Sheath is in position, using fluoroscopy, confirm filter basket closure by ensuring reduction of the filter basket diameter shown by the radiopaque strut markers, keeping in mind that the emboli that have been captured may not allow the ANGIOGUARD XP Emboli Capture Guidewire to reach its initial low profile. 5. Open the hemovalve to allow the ANGIOGUARD XP Emboli Capture Guidewire free movement and to assure that the ANGIOGUARD XP Emboli Capture Guidewire is not damaged as it is removed since it may contain emboli. 6. Remove the ANGIOGUARD XP Emboli Capture Guidewire and Capture Sheath as described in Section 9.4, steps 14 and Deploy a new ANGIOGUARD XP Emboli Capture Guidewire as described in Section 9.3, steps 1 through 16 and Section 9.4, steps 1 through 15. DISCLAIMER OF WARRANTY AND LIMITATION OF REMEDY THERE IS NO EXPRESS OR IMPLIED WARRANTY, INCLUDING WITHOUT LIMITATION ANY IMPLIED WARRANTY OF MERCHANTABILITY OR FITNESS FOR A PARTICULAR PURPOSE, ON THE CORDIS PRODUCT(S) DESCRIBED IN THIS PUBLICATION. UNDER NO CIRCUMSTANCES SHALL CORDIS BE LIABLE FOR ANY DIRECT, INCIDENTAL, OR CONSEQUENTIAL DAMAGES OTHER THAN AS EXPRESSLY PROVIDED BY SPECIFIC LAW. NO PERSON HAS THE AUTHORITY TO BIND CORDIS TO ANY REPRESENTATION OR WARRANTY EXCEPT AS SPECIFICALLY SET FORTH HEREIN. Descriptions or specifications in Cordis printed matter, including this publication, are meant solely to generally describe the product at the time of manufacture and do not constitute any express warranties. Cordis Corporation will not be responsible for any direct, incidental, or consequential damages resulting from reuse of the product. Protected under one or more of the following U.S. Patents: 6,391,044; 6,428,559; 6,485,501; 6,540,768; and other U.S. and foreign patents pending Patient Information A Patient Brochure, which includes information on carotid artery disease, the carotid stent implant procedure, and the ANGIOGUARD XP Emboli Capture Guidewire is available from Cordis and can be obtained by accessing or by contacting Cordis at Label Designation/How Supplied Interventional.014" (0.36 mm) compatible devices mean: Cordis PRECISE Nitinol Stent System Angioplasty balloons The ANGIOGUARD XP Emboli Capture Guidewire is supplied sterile (by ethylene oxide gas) and is intended for ONE USE ONLY. 15
DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service
 M AY. 6. 2011 10:37 A M F D A - C D R H - O D E - P M O N O. 4147 P. 1 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control
M AY. 6. 2011 10:37 A M F D A - C D R H - O D E - P M O N O. 4147 P. 1 DEPARTMENT OF HEALTH & HUMAN SERVICES Public Health Service Food and Drug Administration 10903 New Hampshire Avenue Document Control
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device. Instructions for Use
 The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
The SplitWire Percutaneous Transluminal Angioplasty Scoring Device Instructions for Use Contents Contains one (1) SplitWire device. Sterile. Sterilized with ethylene oxide gas. Radiopaque. For single use
1 Description. 2 Indications. 3 Warnings ASPIRATION CATHETER
 Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
Page 1 of 5 ASPIRATION CATHETER Carefully read all instructions prior to use, observe all warnings and precautions noted throughout these instructions. Failure to do so may result in complications. STERILE.
ER REBOA Catheter. Instructions for Use
 ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
ER REBOA Catheter Instructions for Use Prytime Medical Devices, Inc. 229 N. Main Street Boerne, TX 78006, USA feedback@prytimemedical.com www.prytimemedical.com US 1 210 340 0116 U.S. and Foreign Patents
Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System
 100000017144.3 100000017144.3 Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System Figure 1.065" (1.65 mm) OR.078" (1.98 mm) 16 4 13 10 11 15 8 3 14.062" (1.57 mm) OR.065" (1.65 mm).038" (0.97
100000017144.3 100000017144.3 Instructions for Use Cordis PRECISE PRO Rx Nitinol Stent System Figure 1.065" (1.65 mm) OR.078" (1.98 mm) 16 4 13 10 11 15 8 3 14.062" (1.57 mm) OR.065" (1.65 mm).038" (0.97
RECOMMENDED INSTRUCTIONS FOR USE
 Rapid Exchange PTCA Dilatation Catheter RECOMMENDED INSTRUCTIONS FOR USE Available in diameters 1.25mm to 4.5mm and in lengths 09mm to 40mm Caution: This device should be used only by physicians trained
Rapid Exchange PTCA Dilatation Catheter RECOMMENDED INSTRUCTIONS FOR USE Available in diameters 1.25mm to 4.5mm and in lengths 09mm to 40mm Caution: This device should be used only by physicians trained
AXS Catalyst Distal Access Catheter
 AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
AXS Catalyst Distal Access Catheter Directions for Use 2 AXS Catalyst Distal Access Catheter ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning Contents
NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER
 Page 1 of 5 NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER STERILE. SINGLE USE ONLY. Sterilized with ethylene oxide gas. Non pyrogenic. Do not resterilize. Do not use opened or damaged packages.
Page 1 of 5 NON-COMPLIANT PTCA RAPID EXCHANGE DILATATION CATHETER STERILE. SINGLE USE ONLY. Sterilized with ethylene oxide gas. Non pyrogenic. Do not resterilize. Do not use opened or damaged packages.
Single Pass MCA Revascularization with Trevo
 Single Pass MCA Revascularization with Trevo By Concentric Medical ProVueTM Retriever Case Reported By Sascha Prothmann, MD Klinikum rechts der Isar, Munich, Germany Images contained herein courtesy of
Single Pass MCA Revascularization with Trevo By Concentric Medical ProVueTM Retriever Case Reported By Sascha Prothmann, MD Klinikum rechts der Isar, Munich, Germany Images contained herein courtesy of
Directions For Use. All directions should be read before use
 Directions For Use All directions should be read before use DEVICE DESCRIPTION: The CLEANER.XT Rotational Thrombectomy System is a percutaneous, 6Fr catheter based system (single piece construction) that
Directions For Use All directions should be read before use DEVICE DESCRIPTION: The CLEANER.XT Rotational Thrombectomy System is a percutaneous, 6Fr catheter based system (single piece construction) that
CAROTID ARTERY ANGIOPLASTY
 CAROTID ARTERY ANGIOPLASTY Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical Coverage Guideline
CAROTID ARTERY ANGIOPLASTY Coverage for services, procedures, medical devices and drugs are dependent upon benefit eligibility as outlined in the member's specific benefit plan. This Medical Coverage Guideline
CLARIVEIN INFUSION CATHETER
 CLARIVEIN INFUSION CATHETER General Product Description Overview The ClariVein Infusion Catheter (ClariVein -IC) is an infusion catheter system designed to introduce physician-specified medicaments into
CLARIVEIN INFUSION CATHETER General Product Description Overview The ClariVein Infusion Catheter (ClariVein -IC) is an infusion catheter system designed to introduce physician-specified medicaments into
Figure 1: Revolution TM Peripheral Atherectomy System Diagram. Table 1: Revolution TM Peripheral Atherectomy System Specifications Minimum Burr
 Instructions For Use All instructions should be read before use CAUTION Investigational Device. Limited by Federal (or United States) law to investigational use. DEVICE DESCRIPTION: The Revolution TM Peripheral
Instructions For Use All instructions should be read before use CAUTION Investigational Device. Limited by Federal (or United States) law to investigational use. DEVICE DESCRIPTION: The Revolution TM Peripheral
1 Comparison of Warfarin and Aspirin for Symptomatic Intracranial Arterial Stenosis. N Engl J Med 352;13, March 31, 2005
 The risk of ischemic stroke in patients with Intracranial Atherosclerotic Disease (ICAD) ranges from 7 to 24%. 1,2 Developed specifically for the treatment of ICAD, the Wingspan Stent System and Gateway
The risk of ischemic stroke in patients with Intracranial Atherosclerotic Disease (ICAD) ranges from 7 to 24%. 1,2 Developed specifically for the treatment of ICAD, the Wingspan Stent System and Gateway
Quick Reference Guide
 Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
Quick Reference Guide Indications for Use The AFX Endovascular AAA System is indicated for endovascular treatment in patients with AAA. The devices are indicated for patients with suitable aneurysm morphology
Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter
 Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter Caution: Federal (USA) law restricts this device to sale by or on the order of a physician. DEVICE DESCRIPTION
Instructions for Use Reprocessed LASSO Circular Mapping Diagnostic Electrophysiology (EP) Catheter Caution: Federal (USA) law restricts this device to sale by or on the order of a physician. DEVICE DESCRIPTION
PTA Balloon Dilatation Catheter
 PTA Balloon Dilatation Catheter March 2016 1/6(3214-0) Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions for use Keep
PTA Balloon Dilatation Catheter March 2016 1/6(3214-0) Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions for use Keep
Cardiva Catalyst III INSTRUCTIONS FOR USE
 Cardiva Catalyst III INSTRUCTIONS FOR USE IFU 2469 Rev. G CAUTION Federal (USA) law restricts this device to sale by or on the order of a physician. For single use only GRAPHICAL SYMBOLS ON THE CARDIVA
Cardiva Catalyst III INSTRUCTIONS FOR USE IFU 2469 Rev. G CAUTION Federal (USA) law restricts this device to sale by or on the order of a physician. For single use only GRAPHICAL SYMBOLS ON THE CARDIVA
Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU)
 Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU) For use with the Shockwave Medical, Inc. IVL Generator and Connector Cable Indication for Use The Shockwave Medical IVL System
Peripheral Intravascular Lithotripsy (IVL) Catheter Instructions for Use (IFU) For use with the Shockwave Medical, Inc. IVL Generator and Connector Cable Indication for Use The Shockwave Medical IVL System
Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM
 Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM Innovating for life. CONFORMABILIT Y 6 F S H E AT H C O M PAT I B I L I T Y THE ASSURANT COBALT ILIAC STENT, WITH ITS UNIQUE COBALT CHROMIUM MODULAR
Assurant Cobalt Iliac BALLOON EXPANDABLE STENT SYSTEM Innovating for life. CONFORMABILIT Y 6 F S H E AT H C O M PAT I B I L I T Y THE ASSURANT COBALT ILIAC STENT, WITH ITS UNIQUE COBALT CHROMIUM MODULAR
PTCA Dilatation Catheter. Instructions for Use
 PTCA Dilatation Catheter Instructions for Use Rx ONLY Caution: Federal law restricts this device to sale by or on the order of a physician STERILE. Sterilized with ethylene oxide gas Distributed by Medinol
PTCA Dilatation Catheter Instructions for Use Rx ONLY Caution: Federal law restricts this device to sale by or on the order of a physician STERILE. Sterilized with ethylene oxide gas Distributed by Medinol
System.
 System www.penumbrainc.com POD System Case Examples 45 cm of POD Packing Coil in 1.5 to 3 mm diameter dilating vessel Bronchial Artery Embolization Dr. Amit Kakkar and Dr. Aksim Rivera, Bronx, NY Inflow:
System www.penumbrainc.com POD System Case Examples 45 cm of POD Packing Coil in 1.5 to 3 mm diameter dilating vessel Bronchial Artery Embolization Dr. Amit Kakkar and Dr. Aksim Rivera, Bronx, NY Inflow:
Endorectal Balloon (ERB) Endorectal Balloon (ERB) Instructions for Use
 Endorectal Balloon (ERB) Instructions for Use GRF-0004-00 Rev. 08 Page 1 of 9 Contents Instructions for Use... 3 Indications for Use... 3 Contraindications... 3 Storage... 4 Warnings... 4 Accessories Provided...
Endorectal Balloon (ERB) Instructions for Use GRF-0004-00 Rev. 08 Page 1 of 9 Contents Instructions for Use... 3 Indications for Use... 3 Contraindications... 3 Storage... 4 Warnings... 4 Accessories Provided...
PTA Balloon Dilatation Catheter
 PTA Balloon Dilatation Catheter 1/6(3211-0) February 2016 TIS-488-06272017 Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions
PTA Balloon Dilatation Catheter 1/6(3211-0) February 2016 TIS-488-06272017 Catalogue number Serial number Lot number Use by Do not reuse Do not resterilize Do not use if package is damaged Consult instructions
New Trials in Progress: ACT 1. Jon Matsumura, MD Cannes, France June 28, 2008
 New Trials in Progress: ACT 1 Jon Matsumura, MD Cannes, France June 28, 2008 Faculty Disclosure I disclose the following financial relationships: Consultant, CAS training director, and/or research grants
New Trials in Progress: ACT 1 Jon Matsumura, MD Cannes, France June 28, 2008 Faculty Disclosure I disclose the following financial relationships: Consultant, CAS training director, and/or research grants
Balloon in Balloon (BIB )
 Balloon in Balloon (BIB ) Stent Placement Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed
Balloon in Balloon (BIB ) Stent Placement Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed
PTCA Dilatation Catheter. Instructions for Use
 PTCA Dilatation Catheter Instructions for Use Rx ONLY Caution: Federal law restricts this device to sale by or on the order of a physician STERILE. Sterilized with ethylene oxide gas Distributed by Medinol
PTCA Dilatation Catheter Instructions for Use Rx ONLY Caution: Federal law restricts this device to sale by or on the order of a physician STERILE. Sterilized with ethylene oxide gas Distributed by Medinol
Directions For Use. All directions should be read before use. Page 1 of 8
 Directions For Use All directions should be read before use Page 1 of 8 WARNING: For single use only. Do not reuse, reprocess or re-sterilize. Reuse, reprocessing or re-sterilization may compromise the
Directions For Use All directions should be read before use Page 1 of 8 WARNING: For single use only. Do not reuse, reprocess or re-sterilize. Reuse, reprocessing or re-sterilization may compromise the
INSTRUCTIONS FOR USE
 PERICARDIUM COVERED STENT (PCS) INSTRUCTIONS FOR USE STERILE - Ethylene Oxide Sterilized. For single use only. Do not re-sterilize. THE CLINICAL TECHNIQUES AND PROCEDURES DESCRIBED HERE DO NOT REPRESENT
PERICARDIUM COVERED STENT (PCS) INSTRUCTIONS FOR USE STERILE - Ethylene Oxide Sterilized. For single use only. Do not re-sterilize. THE CLINICAL TECHNIQUES AND PROCEDURES DESCRIBED HERE DO NOT REPRESENT
DURABLE. CONSISTENT. SAFE. IN.PACT Admiral Drug-Coated Balloon
 DURABLE. CONSISTENT. SAFE. Drug-Coated Balloon THE SCIENCE BEHIND THE OUTCOMES DCB has proven, long-term durable outcomes across multiple clinical trials, as well as across complex patient and lesion types.
DURABLE. CONSISTENT. SAFE. Drug-Coated Balloon THE SCIENCE BEHIND THE OUTCOMES DCB has proven, long-term durable outcomes across multiple clinical trials, as well as across complex patient and lesion types.
Ruby Coil. Large Volume Detachable Coils
 Ruby Coil Ruby Case Examples 38 mm Hepatic Artery Aneurysm Pulmonary AVM 2 Coils Dr. James Benenati Miami Cardiac and Vascular Institute, FL Y90 Embolization Type 2 Endoleak Dr. J Moskovitz Florida Hospital,
Ruby Coil Ruby Case Examples 38 mm Hepatic Artery Aneurysm Pulmonary AVM 2 Coils Dr. James Benenati Miami Cardiac and Vascular Institute, FL Y90 Embolization Type 2 Endoleak Dr. J Moskovitz Florida Hospital,
Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a physician.
 Page 1 of 5 Reprocessed by Instructions for Use Reprocessed Ultrasound Catheters Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a physician.
Page 1 of 5 Reprocessed by Instructions for Use Reprocessed Ultrasound Catheters Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a physician.
Radux StandTall Instructions for Use Sheath Extender and Securement Clasp
 Radux StandTall Instructions for Use Sheath Extender and Securement Clasp USA Caution Federal (USA) law restricts this device to sale by or on the order of a health care professional. Caution The Radux
Radux StandTall Instructions for Use Sheath Extender and Securement Clasp USA Caution Federal (USA) law restricts this device to sale by or on the order of a health care professional. Caution The Radux
Express LD Iliac. Premounted Stent System ONLY. over-the-wire
 50455844-01 STERILE - DO NOT RESTERILIZE - SINGLE USE ONLY ONLY 2017-03 < EN > Express LD Iliac over-the-wire Premounted Stent System Caution: Federal Law (USA) restricts this device to sale by or on the
50455844-01 STERILE - DO NOT RESTERILIZE - SINGLE USE ONLY ONLY 2017-03 < EN > Express LD Iliac over-the-wire Premounted Stent System Caution: Federal Law (USA) restricts this device to sale by or on the
Current Non-Surgical Cardiac Interventions. By Pam Bayles, RN, BSN
 Current Non-Surgical Cardiac Interventions By Pam Bayles, RN, BSN Balloon Angioplasty & DES A balloon-tipped catheter was first used in 1964 to treat a cause of atherosclerotic disease in a patient s leg
Current Non-Surgical Cardiac Interventions By Pam Bayles, RN, BSN Balloon Angioplasty & DES A balloon-tipped catheter was first used in 1964 to treat a cause of atherosclerotic disease in a patient s leg
Azur CX 35 Peripheral Coil System (Detachable) Instructions for Use
 Azur CX 35 Peripheral Coil System (Detachable) Instructions for Use DEVICE DESCRIPTION The Detachable Azur CX 35 Peripheral Coil System (Azur system) consists of a coil implant attached to a delivery system.
Azur CX 35 Peripheral Coil System (Detachable) Instructions for Use DEVICE DESCRIPTION The Detachable Azur CX 35 Peripheral Coil System (Azur system) consists of a coil implant attached to a delivery system.
Instructions for Use Reprocessed 3D Diagnostic Ultrasound Catheters
 Reprocessed by Instructions for Use Reprocessed 3D Diagnostic Ultrasound Catheters Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a
Reprocessed by Instructions for Use Reprocessed 3D Diagnostic Ultrasound Catheters Reprocessed Device for Single Use Caution: Federal (U.S.A.) law restricts this device to sale by or on the order of a
Balloon in Balloon (BIB )
 in (BIB ) Dilatation Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed by: B. Braun
in (BIB ) Dilatation Catheter INSTRUCTIONS FOR USE CAUTION: FEDERAL (USA) LAW RESTRICTS THIS DEVICE TO SALE BY OR ON THE ORDER OF A PHYSICIAN. Read all instructions prior to use. Distributed by: B. Braun
Understanding aneurysms and flow diversion treatment
 Surpass Streamline Flow Diverter See package insert for complete indications, contraindications, warnings and instructions for use. INTENDED USE / INDICATIONS FOR USE The Surpass Streamline Flow Diverter
Surpass Streamline Flow Diverter See package insert for complete indications, contraindications, warnings and instructions for use. INTENDED USE / INDICATIONS FOR USE The Surpass Streamline Flow Diverter
Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully
 Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully supported by self-expanding z-stents H&L-B
Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full thickness woven polyester graft material Fully supported by self-expanding z-stents H&L-B
Emerge. PTCA Dilatation Catheter ONLY. monorail. over-the-wire
 90990229-01 Emerge monorail over-the-wire PTCA Dilatation Catheter Table of Contents 2014-09 < EN > warning...1 device description...1 Table 1. Emerge Balloon Coatings...1 Contents...1 Intended use/indications
90990229-01 Emerge monorail over-the-wire PTCA Dilatation Catheter Table of Contents 2014-09 < EN > warning...1 device description...1 Table 1. Emerge Balloon Coatings...1 Contents...1 Intended use/indications
INSTRUCTIONS FOR USE FOR: English
 INSTRUCTIONS FOR USE FOR: English INSTRUCTIONS FOR USE FOR: Carefully read all instructions prior to use. Observe all warnings and precautions noted throughout these instructions. Failure to do so may
INSTRUCTIONS FOR USE FOR: English INSTRUCTIONS FOR USE FOR: Carefully read all instructions prior to use. Observe all warnings and precautions noted throughout these instructions. Failure to do so may
Single Use Curlew TM Multiple Biopsy Forceps
 Single Use Curlew TM Multiple Biopsy Forceps 13 SPECIMEN WITH METAL STORAGE CYLINDER With In Situ Fixation, Batch or Single Specimen Collection US Patents 5,782,747; 5,980,468; 6,071,248; foreign patents
Single Use Curlew TM Multiple Biopsy Forceps 13 SPECIMEN WITH METAL STORAGE CYLINDER With In Situ Fixation, Batch or Single Specimen Collection US Patents 5,782,747; 5,980,468; 6,071,248; foreign patents
Instructions for Use 1
 Instructions for Use 1 Instructions for Use IMPORTANT! Please read before use. Description The Sniper is a dual lumen catheter comprised of an inner guidewire/infusion lumen and an outer, annular lumen
Instructions for Use 1 Instructions for Use IMPORTANT! Please read before use. Description The Sniper is a dual lumen catheter comprised of an inner guidewire/infusion lumen and an outer, annular lumen
COIL SYSTEM ORDERING INFORMATION
 EMBOLIZATION SYSTEM RUBY COIL SYSTEM FRAME with Ruby Standard FILL with Ruby Soft Now with 3 & 40 mm sizes! SYSTEM ANCHOR with Now designed to embolize 3 4 mm vessels! PACK with Coil Now with WAVE Shape
EMBOLIZATION SYSTEM RUBY COIL SYSTEM FRAME with Ruby Standard FILL with Ruby Soft Now with 3 & 40 mm sizes! SYSTEM ANCHOR with Now designed to embolize 3 4 mm vessels! PACK with Coil Now with WAVE Shape
Advanced Innovation for Exceptional Performance
 Advanced Innovation for Exceptional Performance longest BALLOON LENGTHS ON THE MARKET Bard Peripheral Vascular Ultraverse 014 300 mm Boston Scientific Coyote 220 mm Cordis SLEEK RX 220 mm Aviator Plus
Advanced Innovation for Exceptional Performance longest BALLOON LENGTHS ON THE MARKET Bard Peripheral Vascular Ultraverse 014 300 mm Boston Scientific Coyote 220 mm Cordis SLEEK RX 220 mm Aviator Plus
Neuroform EZ Stent System
 Neuroform EZ Stent System Directions for Use 2 Neuroform EZ Stent System 1 cm 1.9 cm 135 cm 185 cm ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning
Neuroform EZ Stent System Directions for Use 2 Neuroform EZ Stent System 1 cm 1.9 cm 135 cm 185 cm ONLY Caution: Federal Law (USA) restricts this device to sale by or on the order of a physician. warning
B05680 Vers. 6/ Figure 2. LifeStent Vascular Stent
 B05680 Vers. 6/12-10 BARD LIFESTENT Stent and Delivery System Vascular Application Recommended Guidewire Length Table Catheter Working Length Recommended Guidewire Length 130 cm 300 cm 80 cm 260 cm 160
B05680 Vers. 6/12-10 BARD LIFESTENT Stent and Delivery System Vascular Application Recommended Guidewire Length Table Catheter Working Length Recommended Guidewire Length 130 cm 300 cm 80 cm 260 cm 160
Cook Medical. Zenith Flex AAA Endovascular Graft with Z-Trak Introduction System Physician Training
 Cook Medical Zenith Flex AAA Endovascular Graft with Z-Trak Introduction System Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full-thickness,
Cook Medical Zenith Flex AAA Endovascular Graft with Z-Trak Introduction System Physician Training Bifurcated system Proximal suprarenal stent Modular (aortic main body and two iliac legs) Full-thickness,
Ancillary Components with Z-Trak Introduction System
 Ancillary Components with Z-Trak Introduction System Zenith Flex AAA Endovascular Graft Ancillary Components Converter Converters can be used to convert a bifurcated graft into an aortouniiliac graft if
Ancillary Components with Z-Trak Introduction System Zenith Flex AAA Endovascular Graft Ancillary Components Converter Converters can be used to convert a bifurcated graft into an aortouniiliac graft if
Phoenix Atherectomy System Product Overview /LB
 Phoenix Atherectomy System Product Overview Phoenix Atherectomy System- The next generation of atherectomy The first hybrid atherectomy system available Combines the benefits of existing atherectomy systems
Phoenix Atherectomy System Product Overview Phoenix Atherectomy System- The next generation of atherectomy The first hybrid atherectomy system available Combines the benefits of existing atherectomy systems
MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES
 MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES PUSHING BOUNDARIES Terumo Interventional Systems is committed to your success with innovative procedural solutions and ongoing support for your most challenging
MAXIMIZE RADIAL SOLUTIONS TO PERIPHERAL CHALLENGES PUSHING BOUNDARIES Terumo Interventional Systems is committed to your success with innovative procedural solutions and ongoing support for your most challenging
Pocket Reference Guide For (CTO) Technologies
 to receive our latest news and key activities. Pocket Reference Guide For (CTO) Technologies LinkedIn page Follow us on CORDIS EMEA Table of Contents Essential prescribing information for both FRONTRUNNER
to receive our latest news and key activities. Pocket Reference Guide For (CTO) Technologies LinkedIn page Follow us on CORDIS EMEA Table of Contents Essential prescribing information for both FRONTRUNNER
ASAHI Corsair INSTRUCTIONS FOR USE
 ASAHI Corsair INSTRUCTIONS FOR USE SYMBOLS Legal manufacturer Do not use if package is damaged GC Minimum GC I.D. REF Catalogue number Caution, consult accompanying documents Maximum injection pressure
ASAHI Corsair INSTRUCTIONS FOR USE SYMBOLS Legal manufacturer Do not use if package is damaged GC Minimum GC I.D. REF Catalogue number Caution, consult accompanying documents Maximum injection pressure
Nit-Occlud. Coil System for PDA Closure IMPLANTATION POCKET GUIDE. Rx only CV / B. Braun Interventional Systems Inc.
 Refer to the Nit-Occlud PDA Instructions for Use for relevant warnings, precautions, complications and contraindications. This device has been designed for single use only. Nit-Occlud Coil System for PDA
Refer to the Nit-Occlud PDA Instructions for Use for relevant warnings, precautions, complications and contraindications. This device has been designed for single use only. Nit-Occlud Coil System for PDA
Z-MED II BALLOON AORTIC AND PULMONIC VALVULOPLASTY CATHETER. Instructions for Use
 Z-MED II BALLOON AORTIC AND PULMONIC VALVULOPLASTY CATHETER Instructions for Use CAUTION: Federal (USA) Law restricts this device to sale by or on the order of a physician. Manufactured for: B. Braun Interventional
Z-MED II BALLOON AORTIC AND PULMONIC VALVULOPLASTY CATHETER Instructions for Use CAUTION: Federal (USA) Law restricts this device to sale by or on the order of a physician. Manufactured for: B. Braun Interventional
Innovation by design. Technology that sets a new standard
 Innovation by design Technology that sets a new standard Flexible nitinol scoring element with three rectangular spiral struts works in tandem with a semi-compliant balloon to score the target lesion Balloon
Innovation by design Technology that sets a new standard Flexible nitinol scoring element with three rectangular spiral struts works in tandem with a semi-compliant balloon to score the target lesion Balloon
TOBA II 12-Month Results Tack Optimized Balloon Angioplasty
 TOBA II 12-Month Results Tack Optimized Balloon Angioplasty William Gray, MD System Chief, Cardiovascular Division Main Line Health, Philadelphia, PA Dissection: The Primary Mechanism of Angioplasty Lesions
TOBA II 12-Month Results Tack Optimized Balloon Angioplasty William Gray, MD System Chief, Cardiovascular Division Main Line Health, Philadelphia, PA Dissection: The Primary Mechanism of Angioplasty Lesions
Sustained Release. Superior Results.
 ELUVIA Drug-Eluting Vascular Stent System Sustained Release. Superior Results. Superiority determined in Post Hoc Superiority Analysis. 12-Month Primary Patency rate of 86.8% in the Eluvia arm (n=309)
ELUVIA Drug-Eluting Vascular Stent System Sustained Release. Superior Results. Superiority determined in Post Hoc Superiority Analysis. 12-Month Primary Patency rate of 86.8% in the Eluvia arm (n=309)
Carotid Artery Stenting
 Carotid Artery Stenting JESSICA MITCHELL, ACNP CENTRAL ILLINOIS RADIOLOGICAL ASSOCIATES External Carotid Artery (ECA) can easily be identified from Internal Carotid Artery (ICA) by noticing the branches.
Carotid Artery Stenting JESSICA MITCHELL, ACNP CENTRAL ILLINOIS RADIOLOGICAL ASSOCIATES External Carotid Artery (ECA) can easily be identified from Internal Carotid Artery (ICA) by noticing the branches.
CAROTID ARTERY ANGIOPLASTY
 CAROTID ARTERY ANGIOPLASTY Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and
CAROTID ARTERY ANGIOPLASTY Non-Discrimination Statement and Multi-Language Interpreter Services information are located at the end of this document. Coverage for services, procedures, medical devices and
Vascu-PICC WITH CUFF PERIPHERALLY INSERTED CENTRAL VEIN ACCESS CATHETER INSTRUCTIONS FOR USE
 Vascu-PICC WITH CUFF PERIPHERALLY INSERTED CENTRAL VEIN ACCESS CATHETER INSTRUCTIONS FOR USE INDICATIONS FOR USE: The Vascu-PICC with cuff Peripherally Inserted Central Vein Catheters are designed for
Vascu-PICC WITH CUFF PERIPHERALLY INSERTED CENTRAL VEIN ACCESS CATHETER INSTRUCTIONS FOR USE INDICATIONS FOR USE: The Vascu-PICC with cuff Peripherally Inserted Central Vein Catheters are designed for
Talent Abdominal Stent Graft
 Talent Abdominal with THE Xcelerant Hydro Delivery System Expanding the Indications for EVAR Treat More Patients Short Necks The Talent Abdominal is the only FDA-approved device for proximal aortic neck
Talent Abdominal with THE Xcelerant Hydro Delivery System Expanding the Indications for EVAR Treat More Patients Short Necks The Talent Abdominal is the only FDA-approved device for proximal aortic neck
MULTILAYER FLOW MODULATOR (MFM ) FOR PERIPHERAL ANEURYSMS TREATMENT (LOWER LIMBS, VISCERALS AND SUBCLAVIAN)
 0459 MULTILAYER FLOW MODULATOR (MFM ) FOR PERIPHERAL ANEURYSMS TREATMENT (LOWER LIMBS, VISCERALS AND SUBCLAVIAN) WITH ITS ANGIOGRAPHIC CONTROL DELIVERY SYSTEM INSTRUCTIONS FOR USE (I.F.U.) English Français
0459 MULTILAYER FLOW MODULATOR (MFM ) FOR PERIPHERAL ANEURYSMS TREATMENT (LOWER LIMBS, VISCERALS AND SUBCLAVIAN) WITH ITS ANGIOGRAPHIC CONTROL DELIVERY SYSTEM INSTRUCTIONS FOR USE (I.F.U.) English Français
Wolverine Coronary Cutting Balloon
 50482216-01 2017-02 < en > Wolverine Coronary MONORAIL OVER-THE-WIRE Microsurgical Dilatation Device Table of Contents Warning...1 device description...1 Contents...1 Quantity Material...1 Intended use/indications
50482216-01 2017-02 < en > Wolverine Coronary MONORAIL OVER-THE-WIRE Microsurgical Dilatation Device Table of Contents Warning...1 device description...1 Contents...1 Quantity Material...1 Intended use/indications
Peel-Apart Percutaneous Introducer Kits for
 Bard Access Systems Peel-Apart Percutaneous Introducer Kits for Table of Contents Contents Page Bard Implanted Ports Hickman*, Leonard*, Broviac*, Tenckhoff*, and Groshong* Catheters Introduction....................................
Bard Access Systems Peel-Apart Percutaneous Introducer Kits for Table of Contents Contents Page Bard Implanted Ports Hickman*, Leonard*, Broviac*, Tenckhoff*, and Groshong* Catheters Introduction....................................
VERNACULAR Trial & Clinical Experience with the VENOVO Venous Stent
 Stephen Black, MD VERNACULAR Trial & Clinical Experience with the VENOVO Venous Stent 1 Speaker Disclaimers The speakers presentation today is on behalf of Bard Peripheral Vascular, Inc. Any discussion
Stephen Black, MD VERNACULAR Trial & Clinical Experience with the VENOVO Venous Stent 1 Speaker Disclaimers The speakers presentation today is on behalf of Bard Peripheral Vascular, Inc. Any discussion
REBEL. Platinum Chromium Coronary Stent System. Patient Information Guide
 REBEL Patient Information Guide REBEL PATIENT INFORMATION GUIDE You have recently had a REBEL bare metal stent implanted in the coronary arteries of your heart. The following information is important for
REBEL Patient Information Guide REBEL PATIENT INFORMATION GUIDE You have recently had a REBEL bare metal stent implanted in the coronary arteries of your heart. The following information is important for
Subclavian artery Stenting
 Subclavian artery Stenting Etiology Atherosclerosis Takayasu s arteritis Fibromuscular dysplasia Giant Cell Arteritis Radiation-induced Vascular Injury Thoracic Outlet Syndrome Neurofibromatosis Incidence
Subclavian artery Stenting Etiology Atherosclerosis Takayasu s arteritis Fibromuscular dysplasia Giant Cell Arteritis Radiation-induced Vascular Injury Thoracic Outlet Syndrome Neurofibromatosis Incidence
Atherectomy: Jetstream and Directional. George S. Chrysant, M.D.
 Atherectomy: Jetstream and Directional George S. Chrysant, M.D. Disclosures Abbott Vascular: MAB, consultant, proctor Abiomed: consultant Boston Scientific: MAB, consultant, proctor Medicines Company:
Atherectomy: Jetstream and Directional George S. Chrysant, M.D. Disclosures Abbott Vascular: MAB, consultant, proctor Abiomed: consultant Boston Scientific: MAB, consultant, proctor Medicines Company:
Neuroform Microdelivery Stent System
 Neuroform Microdelivery Stent System Patient Information Booklet TABLE OF CONTENTS What is the Purpose of This Booklet?...2 What is a Wide Neck, Intracranial Aneurysm?...2 What is the Treatment for Wide
Neuroform Microdelivery Stent System Patient Information Booklet TABLE OF CONTENTS What is the Purpose of This Booklet?...2 What is a Wide Neck, Intracranial Aneurysm?...2 What is the Treatment for Wide
Peter A. Soukas, M.D., FACC, FSVM, FSCAI, RPVI
 Peter A. Soukas, M.D., FACC, FSVM, FSCAI, RPVI Director, Peripheral Vascular Interventional Laboratory Director, Vascular & Endovascular Medicine Fellowship Program Assistant Professor of Medicine The
Peter A. Soukas, M.D., FACC, FSVM, FSCAI, RPVI Director, Peripheral Vascular Interventional Laboratory Director, Vascular & Endovascular Medicine Fellowship Program Assistant Professor of Medicine The
Per-Q-Cath* PICC Catheters with Excalibur Introducer* System
 Bard Access Systems Per-Q-Cath* PICC and Catheters with Excalibur Introducer* System Instructions For Use Table of Contents Table of Contents Page Contents 1 Product Description, Indications & Contraindications
Bard Access Systems Per-Q-Cath* PICC and Catheters with Excalibur Introducer* System Instructions For Use Table of Contents Table of Contents Page Contents 1 Product Description, Indications & Contraindications
Carotid Artery Stenting
 Carotid Artery Stenting Natural history of the carotid stenosis Asymptomatic 80% carotid stenosis - 6% risk of stroke / year Symptomatic carotid stenosis have 10% risk of CVA at one year and 40% at 5 years
Carotid Artery Stenting Natural history of the carotid stenosis Asymptomatic 80% carotid stenosis - 6% risk of stroke / year Symptomatic carotid stenosis have 10% risk of CVA at one year and 40% at 5 years
SYMBOLOGY. = Manufacturer. = Manufacturing Serial Number. = Manufacturing Lot Number = Catalog Number LOT REF. = Do not resterilize Single Use Only
 damage or expenses directly or indirectly arising from use of this product. This warranty is in lieu of and excludes all other warranties not expressly set forth herein, whether express or implied by operation
damage or expenses directly or indirectly arising from use of this product. This warranty is in lieu of and excludes all other warranties not expressly set forth herein, whether express or implied by operation
Carotid Artery Stenting (CAS) Pathophysiology. Technical Considerations. Plaque characteristics: relevant concepts. CAS and CEA
 Carotid Artery Stenting (CAS) Carotid Artery Stenting for Stroke Risk Reduction Matthew A. Corriere MD, MS, RPVI Assistant Professor of Surgery Department of Vascular and Endovascular Surgery Rationale:
Carotid Artery Stenting (CAS) Carotid Artery Stenting for Stroke Risk Reduction Matthew A. Corriere MD, MS, RPVI Assistant Professor of Surgery Department of Vascular and Endovascular Surgery Rationale:
Instructions for Use
 Instructions for Use B05692 Vers.4/09-11 LifeStent Solo Vascular Stent System Figure 1 CAUTION: U.S. federal law restricts this device to sale by or on the order of a physician This device is supplied
Instructions for Use B05692 Vers.4/09-11 LifeStent Solo Vascular Stent System Figure 1 CAUTION: U.S. federal law restricts this device to sale by or on the order of a physician This device is supplied
INSTRUCTIONS FOR USE FOR:
 INSTRUCTIONS FOR USE FOR: en English cs Čeština da Dansk nl Nederlands fi Suomi fr Français de Deutsch el Ελληνικά hu Magyar it Italiano lt Lietuvių no Norsk pl Polski pt Português es Español sv Svenska
INSTRUCTIONS FOR USE FOR: en English cs Čeština da Dansk nl Nederlands fi Suomi fr Français de Deutsch el Ελληνικά hu Magyar it Italiano lt Lietuvių no Norsk pl Polski pt Português es Español sv Svenska
Carotid Revascularization
 Options for Carotid Disease Carotid Revascularization Wayne Causey, MD 2 nd Year Vascular Surgery Fellow Best medical therapy, Carotid Endarterectomy, and Carotid Stenting Who benefits from best medical
Options for Carotid Disease Carotid Revascularization Wayne Causey, MD 2 nd Year Vascular Surgery Fellow Best medical therapy, Carotid Endarterectomy, and Carotid Stenting Who benefits from best medical
DISRUPT PAD. (( Data Summary )) DISRUPT PAD Data Summary SPL Rev. B 2016 Shockwave Medical Inc. All rights reserved.
 DISRUPT PAD (( Data Summary )) DISRUPT PAD Data Summary SPL 60971 Rev. B 1 Summary of the key findings from the DISRUPT PAD Study 99% of femoropopliteal lesions treated were moderately or severely calcified.
DISRUPT PAD (( Data Summary )) DISRUPT PAD Data Summary SPL 60971 Rev. B 1 Summary of the key findings from the DISRUPT PAD Study 99% of femoropopliteal lesions treated were moderately or severely calcified.
ROTABLATORTM. Peripheral. Rotational Atherectomy System. Quick Reference Cards
 Peripheral ROTABLATORTM Rotational Atherectomy System Quick Reference Cards Technical Assistance: 1.800.949.6708 Customer Service: 1.888.272.1001 Complaints: 1.800.811.3211 Setup Video: www.bostonscientific.com/rotablatorsetup
Peripheral ROTABLATORTM Rotational Atherectomy System Quick Reference Cards Technical Assistance: 1.800.949.6708 Customer Service: 1.888.272.1001 Complaints: 1.800.811.3211 Setup Video: www.bostonscientific.com/rotablatorsetup
Treatment Considerations for Carotid Artery Stenosis. Danielle Zielinski, RN, MSN, ACNP Rush University Neurosurgery
 Treatment Considerations for Carotid Artery Stenosis Danielle Zielinski, RN, MSN, ACNP Rush University Neurosurgery 4.29.2016 There is no actual or potential conflict of interest in regards to this presentation
Treatment Considerations for Carotid Artery Stenosis Danielle Zielinski, RN, MSN, ACNP Rush University Neurosurgery 4.29.2016 There is no actual or potential conflict of interest in regards to this presentation
Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING
 Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING 1 2013 Spectranetics. All Rights Reserved. Approved for External
Excimer Laser for Coronary Intervention: Case Study RADIAL APPROACH: CORONARY LASER ATHERECTOMY FOR CTO OF THE LAD FOLLOWED BY PTCA NO STENTING 1 2013 Spectranetics. All Rights Reserved. Approved for External
INSTRUCTIONS FOR USE (IFU)
 INSTRUCTIONS FOR USE (IFU) B05696 Vers.3/11-09 BARD E LUMINEXX Vascular Stent Delivery System Diagrams BARD S.A.F.E. Delivery System with the PERFORMAXX Grip BARD S.A.F.E. Delivery System after removal
INSTRUCTIONS FOR USE (IFU) B05696 Vers.3/11-09 BARD E LUMINEXX Vascular Stent Delivery System Diagrams BARD S.A.F.E. Delivery System with the PERFORMAXX Grip BARD S.A.F.E. Delivery System after removal
Polyurethane Dual Lumen Occlusion Catheter English Instructions for Use. Polyurethane Dual Lumen Occlusion Catheter
 Polyurethane Dual Lumen Occlusion Catheter English Instructions for Use Polyurethane Dual Lumen Occlusion Catheter Polyurethane Dual Lumen Occlusion Catheter (Model Numbers 4F-2L-40, 4F-2L-80, 5F-2L-40,
Polyurethane Dual Lumen Occlusion Catheter English Instructions for Use Polyurethane Dual Lumen Occlusion Catheter Polyurethane Dual Lumen Occlusion Catheter (Model Numbers 4F-2L-40, 4F-2L-80, 5F-2L-40,
MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES. Medtronic Further. Together
 DRUG-COATED BALL0ON TREATMENT FOR PATIENTS WITH INTERMITTENT CLAUDICATION: INSIGHTS FROM THE IN.PACT GLOBAL FULL CLINICAL COHORT MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES Medtronic Further. Together
DRUG-COATED BALL0ON TREATMENT FOR PATIENTS WITH INTERMITTENT CLAUDICATION: INSIGHTS FROM THE IN.PACT GLOBAL FULL CLINICAL COHORT MICHAEL R. JAFF, DO MASSACHUSETTS, UNITED STATES Medtronic Further. Together
Complex ilio-caval revascularization in chronic venous obstruction with the Venovo Stent. Michael K. W. Lichtenberg MD, FESC
 Complex ilio-caval revascularization in chronic venous obstruction with the Venovo Stent Michael K. W. Lichtenberg MD, FESC German Venous Center Arnsberg, Germany Not available for sale or distribution
Complex ilio-caval revascularization in chronic venous obstruction with the Venovo Stent Michael K. W. Lichtenberg MD, FESC German Venous Center Arnsberg, Germany Not available for sale or distribution
The Challenge of Accurately Stenting Aorto-Ostial Lesions
 The Challenge of Accurately Stenting Aorto-Ostial Lesions In a retrospective study of 100 patients in whom stents were placed using angiographic landmarks, correct stent positioning was achieved in only
The Challenge of Accurately Stenting Aorto-Ostial Lesions In a retrospective study of 100 patients in whom stents were placed using angiographic landmarks, correct stent positioning was achieved in only
a physician-initiated study investigating the RoadSaver stent in carotid lesions Dr. Michel Bosiers
 The study a physician-initiated study investigating the RoadSaver stent in carotid lesions Dr. Michel Bosiers Conflict of interest have the following potential conflicts of interest to report: Consulting
The study a physician-initiated study investigating the RoadSaver stent in carotid lesions Dr. Michel Bosiers Conflict of interest have the following potential conflicts of interest to report: Consulting
CATHETER ACCESS KIT. For use with Prometra Programmable Infusion Systems
 CATHETER ACCESS KIT Caution: Federal (USA) Law restricts this device to sale by or on the order of a physician. Table of Contents Contents... 3 Description... 3 Indications... 3 Contraindications... 3
CATHETER ACCESS KIT Caution: Federal (USA) Law restricts this device to sale by or on the order of a physician. Table of Contents Contents... 3 Description... 3 Indications... 3 Contraindications... 3
Vivek R. Deshmukh, MD Director, Cerebrovascular and Endovascular Neurosurgery Chairman, Department of Neurosurgery Providence Brain and Spine
 Vivek R. Deshmukh, MD Director, Cerebrovascular and Endovascular Neurosurgery Chairman, Department of Neurosurgery Providence Brain and Spine Institute The Oregon Clinic Disclosure I declare that neither
Vivek R. Deshmukh, MD Director, Cerebrovascular and Endovascular Neurosurgery Chairman, Department of Neurosurgery Providence Brain and Spine Institute The Oregon Clinic Disclosure I declare that neither
Bard LifeStar Vascular Stent System
 Bard LifeStar Vascular Stent System Instructions For Use (IFU) Bard LifeStar Vascular Stent System Delivery System Diagram 8x60 B05685 Vers.2/12-11 1 Instructions for use Read the Bard LifeStar Vascular
Bard LifeStar Vascular Stent System Instructions For Use (IFU) Bard LifeStar Vascular Stent System Delivery System Diagram 8x60 B05685 Vers.2/12-11 1 Instructions for use Read the Bard LifeStar Vascular
CAREFULLY READ ALL INSTRUCTIONS PRIOR TO USE
 CAREFULLY READ ALL INSTRUCTIONS PRIOR TO USE INDICATIONS FOR USE The LATERA Absorbable Nasal Implant is indicated for supporting upper and lower lateral nasal cartilage. CAUTION: Federal law restricts
CAREFULLY READ ALL INSTRUCTIONS PRIOR TO USE INDICATIONS FOR USE The LATERA Absorbable Nasal Implant is indicated for supporting upper and lower lateral nasal cartilage. CAUTION: Federal law restricts
Chronic Total Occlusion (CTO) Technologies
 to receive our latest news and key activities. Chronic Total Occlusion (CTO) Technologies Re-open vital channels LinkedIn page Follow us on CORDIS EMEA OUTBACK LTD Re-Entry Catheter True Lumen Re-Entry
to receive our latest news and key activities. Chronic Total Occlusion (CTO) Technologies Re-open vital channels LinkedIn page Follow us on CORDIS EMEA OUTBACK LTD Re-Entry Catheter True Lumen Re-Entry
PERFORMANCE YOU CAN TRUST. EverFlex Self-expanding Peripheral Stent with Entrust Delivery System
 PERFORMANCE YOU CAN TRUST EverFlex Self-expanding Peripheral Stent with Entrust Delivery System The Entrust Delivery System is designed to provide improved patient outcomes and procedural efficiency. The
PERFORMANCE YOU CAN TRUST EverFlex Self-expanding Peripheral Stent with Entrust Delivery System The Entrust Delivery System is designed to provide improved patient outcomes and procedural efficiency. The
Extracranial Carotid Artery Stenting With or Without Distal Protection Device
 Extracranial Carotid Artery Stenting With or Without Distal Protection Device Eak-Kyun Shin MD. Professor of Medicine Division of Cardiology, Heart Center, Gil Medical Center Gacheon Medical School Incheon,
Extracranial Carotid Artery Stenting With or Without Distal Protection Device Eak-Kyun Shin MD. Professor of Medicine Division of Cardiology, Heart Center, Gil Medical Center Gacheon Medical School Incheon,
Maximise Clinical Safety*. Cordis ExoSeal. The novel approach to secure and easy closure. Vascular Closure Device CORDIS EXOSEAL VCD
 Cordis ExoSeal Vascular Closure Device Cardiology and Endovascular CORDIS EXOSEAL VCD Maximise Clinical Safety*. The novel approach to secure and easy closure. * Clinical data from the ECLIPSE Trial indicates
Cordis ExoSeal Vascular Closure Device Cardiology and Endovascular CORDIS EXOSEAL VCD Maximise Clinical Safety*. The novel approach to secure and easy closure. * Clinical data from the ECLIPSE Trial indicates
Prevention and Management of Cardiac Adverse Event
 Prevention and Management of Cardiac Adverse Event Carlo Cernetti Department of Interventional Cardiology Mirano (Italy) Cannes MEEC 14 June 2007 Are these risks factors of Haemodynamic Instability
Prevention and Management of Cardiac Adverse Event Carlo Cernetti Department of Interventional Cardiology Mirano (Italy) Cannes MEEC 14 June 2007 Are these risks factors of Haemodynamic Instability
Patient guide: pfm Nit-Occlud PDA coil occlusion system. Catheter occlusion of. Patent Ductus Arteriosus. with the
 Patient guide: Catheter occlusion of Patent Ductus Arteriosus with the pfm Nit-Occlud PDA coil occlusion system pfm Produkte für die Medizin - AG Wankelstr. 60 D - 50996 Cologne Phone: +49 (0) 2236 96
Patient guide: Catheter occlusion of Patent Ductus Arteriosus with the pfm Nit-Occlud PDA coil occlusion system pfm Produkte für die Medizin - AG Wankelstr. 60 D - 50996 Cologne Phone: +49 (0) 2236 96
