NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOLS EPTIFIBATIDE (INTEGRILIN) PROTOCOL
|
|
|
- Lenard Hodge
- 6 years ago
- Views:
Transcription
1 NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE S EPTIFIBATIDE (INTEGRILIN) I. PURPOSE: A. Integrilin (Eptifibatide) is a specific and potent inhibitor of the platelet receptor glycoprotein IIb-IIIa, preventing binding of fibrinogen to GP IIb-IIIa, thus helping to prevent platelet aggregation. B. Integrilin (Eptifibatide) is indicated for the treatment of patients with acute coronary syndrome (unstable angina, non Q MI), including patients who are to be managed medically and those undergoing percutaneous coronary intervention (PCI). II. PATIENT SELECTION: The patient must meet some or all of the following criteria: A. Chest pain at rest (> 10 minutes duration) within the previous 24 hours. B. ECG abnormalities within 12 hours, defined as: ST segment depression > 0.5 mm, or T wave inversion > 1 mm, or ST segment elevation between 0.6 mm and 1 mm, or Positive CK-MB assay, or Positive Troponin T or I C. Percutaneous coronary intervention is planned within hours (angioplasty, atherectomy, stent placement). Date of Original: 4/99 Reviewed Revised 12/01 3/08 3/08 3/09 3/09 9/11 9/11 9/ /14 10/15 10/16 6/17 7
2 PAGE 2 III. CONTRAINDICATIONS: Because Integrilin (Eptifibatide) increases the risk of bleeding, it is contraindicated in the following situations: A. A history of bleeding diathesis, or evidence of active abnormal bleeding within the previous 30 days. B. Severe hypertension (systolic blood pressure > 200mmHg or diastolic blood pressure > 110mmHg) not adequately controlled on antihypertensive therapy. C. Major surgery within preceding 6 weeks. D. History of stroke within 30 days, or any history of hemorrhagic stroke. E. Current or planned administration of another parenteral GP IIb-IIIa inhibitor. F. Platelet count < 100,000/mm3. G. Serum creatinine > 2 mg/dl (for the 180 mcg/kg bolus and the 2 mcg/kg/min infusion) or > 4mg/dL (for the 135 mcg/kg bolus and the 0.5 mcg/kg/min infusion). H. Dependency on renal dialysis. I. Known hypersensitivity to any component of the product. 7a EPTIFIBATICLE (INTEGRILLIN)
3 PAGE 3 IV. SPECIAL CONSIDERATIONS: A. At least two primary, preferably three, I.V. infusion lines, or per M.D. order, will be established prior to dosing with Integrilin (Eptifibatide) minimize venipunctures. B. Bleeding is the most common complication encountered during Integrilin (Eptifibatide) therapy. If bleeding cannot be controlled with pressure, infusion of etifibatide and concomitant heparin should be discontinued.. C. Arterial and venous punctures, IM injections, and the use of urinary catheters, nasotracheal intubation, and nasogastric tubes should be minimized. When obtaining I.V. access, noncompressible sites (e.g., subclavian or jugular veins) should be avoided. D. Before infusion of Integrilin eptifibatide, the following lab tests should be performed to identify pre-existing hemostatic abnormalities: hemoglobin and hematocrit, platelet count, serum creatinine, and PT/PTT. Lab testing to be order by physician. E. The PTT should be maintained between 50 and 70 seconds unless PCI is to be performed. During PCI, the ACT should be maintained between seconds. F. The PTT should be checked prior to arterial sheath removal. The sheath should not be removed unless the PTT is < 45 seconds. 7b
4 PAGE 4 G. In patients treated with heparin, bleeding can be minimized by close monitoring of the PTT. H. If the patient experiences a confirmed platelet decrease to < 100,000mm3, eptifibatide and heparin should be discontinued and the condition appropriately monitored and treated. I. In clinical studies, patients were enrolled up to 94 years of age, with no apparent difference in efficacy between the older and younger patients treated with eptifibatide. The incidence of bleeding complications was higher in the elderly patients, with the incremental risk of eptifibatideassociated bleeding greater in the older patients. J. Use toothettes for oral care. V. PROCEDURE: A. The patient and/or patient s family should be well informed by the physician of the risk of potential bleeding complications with the administration of Integrilin (Eptifibatide). B. After orders have been received from the cardiologist for the administration of Integrilin (Eptifibatide), all labs, per physician orders, should be drawn and lines placed prior to dosing the patient. C. Continue therapy with Heparin and aspirin as ordered by the physician during administration of Integrilin (Eptifibatide). Follow the IV Heparin protocol for PTT parameters/times to maintain target PTT between 50 and 70 seconds. 7c
5 PAGE 5 VI. DOSAGE AND ADMINISTRATION: A. The recommended adult dosage of eptifibatide in patients with acute coronary syndrome is an IV bolus of 180 mcg/kg as soon as possible following diagnosis, followed by a continuous infusion of 2 mcg/kg/min until hospital discharge or initiation of CABG surgery, up to 72 hours. B. If a patient is to undergo PCI while receiving eptifibatide, consideration can be given to decreasing the infusion rate to 0.5 mcg/kg/min at the time of the procedure. C. In patients not presenting with acute coronary syndrome, and undergoing PCI, the recommended adult dosage of eptifibatide is an IV bolus of 135 mcg/kg, administered immediately before the initiation of PCI, followed by a continuous infusion of 0.5 mcg/kg/min for hours. D. Instructions for administration: 1. Integrilin (eptifibatide) solution should be inspected visually for particulate matter and discoloration prior to administration. 2. Integrilin may be administered in the same IV line as alteplase, atropine, dobutamine, heparin, lidocaine, meperidine, metoprolol, midazolam, morphine, nitroglycerin, or verapamil. It should not be administered through the same IV line as furosemide. 3. Integrilin (eptifibatide) may be administered in the same IV line with 0.9% NaCl or 0.9% NaCl 5% dextrose. The infusion may also contain up to 40mEq/L of potassium chloride. 4. The bolus dose of Integrilin (eptifibatide) should be withdrawn from the 10-ml vial into a syringe, and administered IV push over 1-2 minutes. Each 10-ml vial contains 20mg eptifibatide in sterile solution. 7d
6 PAGE 6 5. Immediately following the bolus dose administration, a continuous infusion of Integrilin (eptifibatide) should be initiated. When using an IV infusion pump, Integrilin (eptifibatide) should be administered undiluted directly from the 100-ml vial. This vial should be spiked with a vented infusion set, with care taken to center the spike within the circle on the stopper top. Each 100-ml vial contains 75 mg eptifibatide in sterile solution. 6. Integrilin (eptifibatide) is to be administered by volume according to patient weight. 7. Integrilin (eptifibatide) vials should be refrigerated at degrees F. 8. The physician will determine and order the bolus and infusion rate of his/her choice. 7e
7 INTEGRILIN (EPTIFIBATIDE) DOSING CHART FOR PATIENTS WITH ACUTE CORONARY SYNDROME (180 mcg/kg Bolus and 2 mcg/kg/min Infusion) Patient Bolus Infusion Weight Volume Rate (kg) (2 mg/ml) (0.75 mg/ml) ml 6ml/hr > INTEGRILIN (EPTIFIBATIDE) DOSING CHART BY WEIGHT FOR PATIENTS WITHOUT ACUTE CORONARY SYNDROMES UNDERGOING PCI (135 mcg/kg Bolus and 0.5 mcg/kg/min Infusion) Patient Bolus Infusion Weight Volume Rate (kg) (2 mg/ml) (0.75 mg/ml) ml 2 ml/hr g
PRODUCT MONOGRAPH. Pr INTEGRILIN. Eptifibatide Injection Intravenous Solution. 2 mg/ml bolus Injection 0.75 mg/ml Injection (as infused)
 PRODUCT MONOGRAPH Pr INTEGRILIN Injection Intravenous Solution 2 mg/ml bolus Injection 0.75 mg/ml Injection (as infused) Platelet Aggregation Inhibitor Merck Canada Inc. Date of Preparation: 16750 route
PRODUCT MONOGRAPH Pr INTEGRILIN Injection Intravenous Solution 2 mg/ml bolus Injection 0.75 mg/ml Injection (as infused) Platelet Aggregation Inhibitor Merck Canada Inc. Date of Preparation: 16750 route
BOLUS VIAL + INFUSION
 INDICATIONS AND USAGE AGGRASTAT is indicated to reduce the rate of thrombotic cardiovascular events (combined endpoint of death, myocardial infarction, or refractory ischemia/ repeat cardiac procedure)
INDICATIONS AND USAGE AGGRASTAT is indicated to reduce the rate of thrombotic cardiovascular events (combined endpoint of death, myocardial infarction, or refractory ischemia/ repeat cardiac procedure)
DOSING AND ADMINISTRATION
 DOSING AND ADMINISTRATION USING 100 ML VIAL OR 250 ML BAG (50 MCG/ML UNITS) 1 2 BOLUS 25 mcg/kg within 5 minutes INFUSION CrCl >60 ml/min: 0.15 mcg/kg/min CrCl
DOSING AND ADMINISTRATION USING 100 ML VIAL OR 250 ML BAG (50 MCG/ML UNITS) 1 2 BOLUS 25 mcg/kg within 5 minutes INFUSION CrCl >60 ml/min: 0.15 mcg/kg/min CrCl
NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOLS. ACTIVASE (t-pa) INFUSION PROTOCOL FOR ACUTE MYOCARDIAL INFARCTION
 NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOLS ACTIVASE (t-pa) FOR ACUTE MYOCARDIAL INFARCTION I. PURPOSE: A. To reduce the extent of myocardial infarction by lysing the clot in
NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOLS ACTIVASE (t-pa) FOR ACUTE MYOCARDIAL INFARCTION I. PURPOSE: A. To reduce the extent of myocardial infarction by lysing the clot in
PRE-MIXED BAGS DOSING AND ADMINISTRATION. Rate (ml/hr)
 Weight (lb) Weight (kg) PRE-MIXED BAGS DOSING AND ADMINISTRATION 1 2 BOLUS Using Pre-Mixed Bags 25 mcg/kg within 5 minutes Volume (ml) Rate (ml/hr) INFUSION Using Pre-Mixed Bags CrCl >60 ml/min: 0.15 mcg/kg/min
Weight (lb) Weight (kg) PRE-MIXED BAGS DOSING AND ADMINISTRATION 1 2 BOLUS Using Pre-Mixed Bags 25 mcg/kg within 5 minutes Volume (ml) Rate (ml/hr) INFUSION Using Pre-Mixed Bags CrCl >60 ml/min: 0.15 mcg/kg/min
AGGRASTAT (tirofiban hydrochloride) injection, for intravenous use Initial U.S. Approval: 1998
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use AGGRASTAT (tirofiban hydrochloride) safely and effectively. See full prescribing information for
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use AGGRASTAT (tirofiban hydrochloride) safely and effectively. See full prescribing information for
CONTRAINDICATIONS INDICATIONS AND USAGE WARNINGS AND PRECAUTIONS ADVERSE REACTIONS DOSAGE AND ADMINISTRATION DRUG INTERACTIONS
 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use AGGRASTAT (tirofiban hydrochloride) safely and effectively. See full prescribing information for
HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use AGGRASTAT (tirofiban hydrochloride) safely and effectively. See full prescribing information for
NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOLS. ACUTE CEREBROVASCULAR ACCIDENT TPA (ACTIVASE /alteplase) FOR THROMBOLYSIS
 NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOLS ACUTE CEREBROVASCULAR ACCIDENT TPA (ACTIVASE /alteplase) FOR THROMBOLYSIS I. Purpose : A. To reduce morbidity and mortality associated
NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOLS ACUTE CEREBROVASCULAR ACCIDENT TPA (ACTIVASE /alteplase) FOR THROMBOLYSIS I. Purpose : A. To reduce morbidity and mortality associated
Management of Acute Myocardial Infarction
 Management of Acute Myocardial Infarction Prof. Hossam Kandil Professor of Cardiology Cairo University ST Elevation Acute Myocardial Infarction Aims Of Management Emergency care (Pre-hospital) Early care
Management of Acute Myocardial Infarction Prof. Hossam Kandil Professor of Cardiology Cairo University ST Elevation Acute Myocardial Infarction Aims Of Management Emergency care (Pre-hospital) Early care
NEW ZEALAND DATA SHEET INTEGRILIN SOLUTION FOR INJECTION. INTEGRILIN Eptifibatide (20mg in 10mL and 75mg in 100mL) solution for injection.
 NEW ZEALAND DATA SHEET INTEGRILIN SOLUTION FOR INJECTION NAME OF MEDICINE INTEGRILIN Eptifibatide (20mg in 10mL and 75mg in 100mL) solution for injection. PRESENTATION INTEGRILIN is a clear, colourless
NEW ZEALAND DATA SHEET INTEGRILIN SOLUTION FOR INJECTION NAME OF MEDICINE INTEGRILIN Eptifibatide (20mg in 10mL and 75mg in 100mL) solution for injection. PRESENTATION INTEGRILIN is a clear, colourless
Learning Objectives. Epidemiology of Acute Coronary Syndrome
 Cardiovascular Update: Antiplatelet therapy in acute coronary syndromes PHILLIP WEEKS, PHARM.D., BCPS-AQ CARDIOLOGY Learning Objectives Interpret guidelines as they relate to constructing an antiplatelet
Cardiovascular Update: Antiplatelet therapy in acute coronary syndromes PHILLIP WEEKS, PHARM.D., BCPS-AQ CARDIOLOGY Learning Objectives Interpret guidelines as they relate to constructing an antiplatelet
6/1/18 LEARNING OBJECTIVES PATIENT POPULATION PRESENTATIONS
 PREVENTING HOSPITAL READMISSIONS IN CARDIOVASCULAR PATIENTS Christina Cortez Perry, MSN, FNP-C, CCCC Cardiology Coordinator- Corpus Christi Medical Center 1 2 LEARNING OBJECTIVES Identify the target patient
PREVENTING HOSPITAL READMISSIONS IN CARDIOVASCULAR PATIENTS Christina Cortez Perry, MSN, FNP-C, CCCC Cardiology Coordinator- Corpus Christi Medical Center 1 2 LEARNING OBJECTIVES Identify the target patient
Inhixa (Enoxaparin Sodium)
 Inhixa (Enoxaparin Sodium) P R E V ENTIS SAFETY D E V I C E P R E V E N T I S I S A N AU TO M AT I C N E E D L E S H I E L D I N G S Y S T E M, W H I C H H A S A C O V E R T H AT E X T E N D S O V E R
Inhixa (Enoxaparin Sodium) P R E V ENTIS SAFETY D E V I C E P R E V E N T I S I S A N AU TO M AT I C N E E D L E S H I E L D I N G S Y S T E M, W H I C H H A S A C O V E R T H AT E X T E N D S O V E R
BIVASTAT Injection (Bivalirudin)
 Published on: 1 Aug 2014 BIVASTAT Injection (Bivalirudin) Compostion BIVASTAT Each vial contains: Bivalirudin.. 250 mg Excipients.q.s. Supplied with 5 ml Sterile Water for Injection, IP Dosage Form Sterile
Published on: 1 Aug 2014 BIVASTAT Injection (Bivalirudin) Compostion BIVASTAT Each vial contains: Bivalirudin.. 250 mg Excipients.q.s. Supplied with 5 ml Sterile Water for Injection, IP Dosage Form Sterile
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS
 ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT INTEGRILIN 0.75 mg/ml solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of solution for infusion contains
ANNEX I SUMMARY OF PRODUCT CHARACTERISTICS 1 1. NAME OF THE MEDICINAL PRODUCT INTEGRILIN 0.75 mg/ml solution for infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each ml of solution for infusion contains
Cangrelor: Is it the new CHAMPION for PCI? Robert Barcelona, PharmD, BCPS Clinical Pharmacy Specialist, Cardiac Intensive Care Unit November 13, 2015
 Cangrelor: Is it the new CHAMPION for PCI? Robert Barcelona, PharmD, BCPS Clinical Pharmacy Specialist, Cardiac Intensive Care Unit November 13, 2015 Objectives Review the pharmacology and pharmacokinetic
Cangrelor: Is it the new CHAMPION for PCI? Robert Barcelona, PharmD, BCPS Clinical Pharmacy Specialist, Cardiac Intensive Care Unit November 13, 2015 Objectives Review the pharmacology and pharmacokinetic
GUIDELINES FOR THE EARLY MANAGEMENT OF PATIENTS WITH ACUTE ISCHEMIC STROKE
 2018 UPDATE QUICK SHEET 2018 American Heart Association GUIDELINES FOR THE EARLY MANAGEMENT OF PATIENTS WITH ACUTE ISCHEMIC STROKE A Summary for Healthcare Professionals from the American Heart Association/American
2018 UPDATE QUICK SHEET 2018 American Heart Association GUIDELINES FOR THE EARLY MANAGEMENT OF PATIENTS WITH ACUTE ISCHEMIC STROKE A Summary for Healthcare Professionals from the American Heart Association/American
Objectives. Treatment of ACS. Early Invasive Strategy. UA/NSTEMI General Concepts. UA/NSTEMI Initial Therapy/Antithrombotic
 Objectives Treatment of ACS Michael P. Gulseth, Pharm. D., BCPS Pharmacotherapy II Spring 2006 Define early invasive strategy and what patients typically receive this approach Compare/contrast the medications
Objectives Treatment of ACS Michael P. Gulseth, Pharm. D., BCPS Pharmacotherapy II Spring 2006 Define early invasive strategy and what patients typically receive this approach Compare/contrast the medications
Continuing Medical Education Post-Test
 Continuing Medical Education Post-Test Based on the information presented in this monograph, please choose one correct response for each of the following questions or statements. Record your answers on
Continuing Medical Education Post-Test Based on the information presented in this monograph, please choose one correct response for each of the following questions or statements. Record your answers on
Acute coronary syndromes
 Acute coronary syndromes 1 Acute coronary syndromes Acute coronary syndromes results primarily from diminished myocardial blood flow secondary to an occlusive or partially occlusive coronary artery thrombus.
Acute coronary syndromes 1 Acute coronary syndromes Acute coronary syndromes results primarily from diminished myocardial blood flow secondary to an occlusive or partially occlusive coronary artery thrombus.
ST. DOMINIC-JACKSON MEMORIAL HOSPITAL JACKSON, MISSISSIPPI. CONTINUOUS RENAL REPLACEMENT THERAPY (CRRT) HEPARIN ANTICOAGULATION Page 1 of 5
 HEPARIN ANTICOAGULATION Page 1 of 5 Pharmacy Mnemonic: CRRTHEP1 PATIENT DATA: DIAGNOSIS: AKI ESRD Other: WEIGHT: Today: kg Admission Weight:: kg Dry Weight: kg Access TYPE: Temporary Dialysis Catheter
HEPARIN ANTICOAGULATION Page 1 of 5 Pharmacy Mnemonic: CRRTHEP1 PATIENT DATA: DIAGNOSIS: AKI ESRD Other: WEIGHT: Today: kg Admission Weight:: kg Dry Weight: kg Access TYPE: Temporary Dialysis Catheter
When the learner has completed this module, she/he will be able to:
 Thrombolytics and Myocardial Infarction WWW.RN.ORG Reviewed September 2017, Expires September 2019 Provider Information and Specifics available on our Website Unauthorized Distribution Prohibited 2017
Thrombolytics and Myocardial Infarction WWW.RN.ORG Reviewed September 2017, Expires September 2019 Provider Information and Specifics available on our Website Unauthorized Distribution Prohibited 2017
6 th Floor and 7 East Nurses Guide Intravenous Drip List Approved for RN Administration University of Kentucky Chandler Medical Center
 RATE Abciximab (Reopro) Alteplase (tpa, Activase) All units 6S and 6W ONLY Platelet aggregation inhibitor Thrombolytic agent Bolus: 0.25 mg/kg IV over 5 min Infusion: 0.125 0.9 mg/kg (max 90 mg); 10% of
RATE Abciximab (Reopro) Alteplase (tpa, Activase) All units 6S and 6W ONLY Platelet aggregation inhibitor Thrombolytic agent Bolus: 0.25 mg/kg IV over 5 min Infusion: 0.125 0.9 mg/kg (max 90 mg); 10% of
Nanik Hatsakorzian Pharm.D/MPH
 Pharm.D/MPH 2014 1 Therapeutics FDA indication & Dosing Clinical Pearls Anticoagulants Heparin Antiphospholipid antibody syndrome Cerebral thromboembolism Prosthetic heart valve Acute coronary syndrome
Pharm.D/MPH 2014 1 Therapeutics FDA indication & Dosing Clinical Pearls Anticoagulants Heparin Antiphospholipid antibody syndrome Cerebral thromboembolism Prosthetic heart valve Acute coronary syndrome
CLINICAL GUIDELINES ID TAG
 Title: Author: Speciality/ Division: Directorate: Date Uploaded: Review Date: September 2019 CG ID TAG CG0423 CLINICAL GUIDELINES ID TAG Clinical Guideline for Alteplase in intra-arterial thrombolysis
Title: Author: Speciality/ Division: Directorate: Date Uploaded: Review Date: September 2019 CG ID TAG CG0423 CLINICAL GUIDELINES ID TAG Clinical Guideline for Alteplase in intra-arterial thrombolysis
ADULT POST NEUROLOGIC INTERVENTION ORDERS 2 of 4
 9 Actual 9 Estimated DOWNTIME INTERVENTION 1 of 4 Weight kg 9 Actual 9 Estimated Height cm ALLERGIES: REFER TO ALLERGY PROFILE/ POWERCHART Admit to Dr.: Bed Type: Dx: ( ) Check, circle and/or fill in all
9 Actual 9 Estimated DOWNTIME INTERVENTION 1 of 4 Weight kg 9 Actual 9 Estimated Height cm ALLERGIES: REFER TO ALLERGY PROFILE/ POWERCHART Admit to Dr.: Bed Type: Dx: ( ) Check, circle and/or fill in all
Guideline for STEMI. Reperfusion at a PCI-Capable Hospital
 MANSOURA. 2015 Guideline for STEMI Reperfusion at a PCI-Capable Hospital Mahmoud Yossof MANSOURA 2015 Reperfusion Therapy for Patients with STEMI *Patients with cardiogenic shock or severe heart failure
MANSOURA. 2015 Guideline for STEMI Reperfusion at a PCI-Capable Hospital Mahmoud Yossof MANSOURA 2015 Reperfusion Therapy for Patients with STEMI *Patients with cardiogenic shock or severe heart failure
Unstable angina and NSTEMI
 Issue date: March 2010 Unstable angina and NSTEMI The early management of unstable angina and non-st-segment-elevation myocardial infarction This guideline updates and replaces recommendations for the
Issue date: March 2010 Unstable angina and NSTEMI The early management of unstable angina and non-st-segment-elevation myocardial infarction This guideline updates and replaces recommendations for the
Diagnosis and Management of Acute Myocardial Infarction
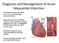 Diagnosis and Management of Acute Myocardial Infarction Acute Myocardial Infarction (AMI) occurs as a result of prolonged myocardial ischemia Atherosclerosis leads to endothelial rupture or erosion that
Diagnosis and Management of Acute Myocardial Infarction Acute Myocardial Infarction (AMI) occurs as a result of prolonged myocardial ischemia Atherosclerosis leads to endothelial rupture or erosion that
Timing of Surgery After Percutaneous Coronary Intervention
 Timing of Surgery After Percutaneous Coronary Intervention Deepak Talreja, MD, FACC Bayview/EVMS/Sentara Outline/Highlights Timing of elective surgery What to do with medications Stopping anti-platelet
Timing of Surgery After Percutaneous Coronary Intervention Deepak Talreja, MD, FACC Bayview/EVMS/Sentara Outline/Highlights Timing of elective surgery What to do with medications Stopping anti-platelet
Cardiovascular Disorders Lecture 3 Coronar Artery Diseases
 Cardiovascular Disorders Lecture 3 Coronar Artery Diseases By Prof. El Sayed Abdel Fattah Eid Lecturer of Internal Medicine Delta University Coronary Heart Diseases It is the leading cause of death in
Cardiovascular Disorders Lecture 3 Coronar Artery Diseases By Prof. El Sayed Abdel Fattah Eid Lecturer of Internal Medicine Delta University Coronary Heart Diseases It is the leading cause of death in
Drug Max dose approved for IVP Dilution Rate Monitoring Parameters. Dilution not necessary (Available in prefilled syringe)
 Drug Max dose approved for IVP Dilution Rate Monitoring Parameters Acetazolamide 500 mg Reconstitute with at least 5ml sterile water (max concentration should not exceed 100mg/ml) 100-500 mg/min Hypotension
Drug Max dose approved for IVP Dilution Rate Monitoring Parameters Acetazolamide 500 mg Reconstitute with at least 5ml sterile water (max concentration should not exceed 100mg/ml) 100-500 mg/min Hypotension
Physician Orders ADULT
 Physician Orders ADULT attach patient label here Order Set: ED Non-STEMI Unstable Angina Orders Height: cm Weight: kg Allergies: No known allergies Medication allergy(s): Latex allergy Other: Admission/Transfer/Discharge
Physician Orders ADULT attach patient label here Order Set: ED Non-STEMI Unstable Angina Orders Height: cm Weight: kg Allergies: No known allergies Medication allergy(s): Latex allergy Other: Admission/Transfer/Discharge
DATA SHEET. REOPRO vial 5 ml contains 10 mg abciximab rmc. REOPRO is a clear, colourless, sterile solution for intravenous use.
 DATA SHEET REOPRO Abciximab rmc, solution for injection, vials 2 mg/ml. Presentation REOPRO vial 5 ml contains 10 mg abciximab rmc. REOPRO is a clear, colourless, sterile solution for intravenous use.
DATA SHEET REOPRO Abciximab rmc, solution for injection, vials 2 mg/ml. Presentation REOPRO vial 5 ml contains 10 mg abciximab rmc. REOPRO is a clear, colourless, sterile solution for intravenous use.
SUPPLEMENTAL MATERIAL
 SUPPLEMENTAL MATERIAL Table S1: Number and percentage of patients by age category Distribution of age Age
SUPPLEMENTAL MATERIAL Table S1: Number and percentage of patients by age category Distribution of age Age
PRODUCT INFORMATION INTEGRILIN INJECTION
 PRODUCT INFORMATION INTEGRILIN INJECTION NAME OF THE MEDICINE Eptifibatide Chemical structure: Chemical Name: CAS Registry No.: 157630-07-4 cyclo(s-s)-mercaptopropionyl-l-homoarginylglycyl-l-aspartyl-
PRODUCT INFORMATION INTEGRILIN INJECTION NAME OF THE MEDICINE Eptifibatide Chemical structure: Chemical Name: CAS Registry No.: 157630-07-4 cyclo(s-s)-mercaptopropionyl-l-homoarginylglycyl-l-aspartyl-
Cardiac Catheter Labs Intravenous Drug Therapy Guide
 Title of Guideline (must include the word Guideline (not protocol, policy, procedure etc) Contact Name and Job Title (author) Cardiac Catheter Lab IV Medicines Guideline Helen Buxton ( Senior Cath Lab
Title of Guideline (must include the word Guideline (not protocol, policy, procedure etc) Contact Name and Job Title (author) Cardiac Catheter Lab IV Medicines Guideline Helen Buxton ( Senior Cath Lab
Platelet-fibrin clot. 50Kd STEMI. Abciximab. Video of a IIb/IIIa inhibitor in action. Unstable Angina and non-stsegment
 Objectives IIb/IIIa, Vitamin K, and Direct Thrombin Inhibition in Cardiology Michael Gulseth, Pharm. D., BCPS Assistant Professor, Duluth Pharmacy 6122 February 14, 2005 Describe the pharmacology, kinetics,
Objectives IIb/IIIa, Vitamin K, and Direct Thrombin Inhibition in Cardiology Michael Gulseth, Pharm. D., BCPS Assistant Professor, Duluth Pharmacy 6122 February 14, 2005 Describe the pharmacology, kinetics,
APPENDIX F: CASE REPORT FORM
 APPENDIX F: CASE REPORT FORM Instruction: Complete this form to notify all ACS admissions at your centre to National Cardiovascular Disease Registry. Where check boxes are provided, check ( ) one or more
APPENDIX F: CASE REPORT FORM Instruction: Complete this form to notify all ACS admissions at your centre to National Cardiovascular Disease Registry. Where check boxes are provided, check ( ) one or more
Package leaflet: Information for the user. ReoPro 2 mg/ml solution for injection or infusion. abciximab
 Package leaflet: Information for the user ReoPro 2 mg/ml solution for injection or infusion abciximab Read all of this leaflet carefully before you start using this medicine because it contains important
Package leaflet: Information for the user ReoPro 2 mg/ml solution for injection or infusion abciximab Read all of this leaflet carefully before you start using this medicine because it contains important
Clopidogrel Date: 15 July 2008
 These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinicalTrials.gov
These results are supplied for informational purposes only. Prescribing decisions should be made based on the approved package insert in the country of prescription Sponsor/company: sanofi-aventis ClinicalTrials.gov
How to give thrombolysis in acute myocardial infarction
 Page 1 of 6 How to give thrombolysis in acute myocardial infarction Original article: Michael Tam In the major urban hospitals, there will be little place for thrombolysis in acute STEMI (STelevation myocardial
Page 1 of 6 How to give thrombolysis in acute myocardial infarction Original article: Michael Tam In the major urban hospitals, there will be little place for thrombolysis in acute STEMI (STelevation myocardial
FastTest. You ve read the book now test yourself
 FastTest You ve read the book...... now test yourself To ensure you have learned the key points that will improve your patient care, read the authors questions below. The answers will refer you back to
FastTest You ve read the book...... now test yourself To ensure you have learned the key points that will improve your patient care, read the authors questions below. The answers will refer you back to
Nitroglycerin and Heparin Drip Interfacility Protocols
 Nitroglycerin and Heparin Drip Interfacility Protocols EMS Protocol This protocol applies to nitroglycerin and Heparin drips that are initiated at the transferring facility prior to transport and are not
Nitroglycerin and Heparin Drip Interfacility Protocols EMS Protocol This protocol applies to nitroglycerin and Heparin drips that are initiated at the transferring facility prior to transport and are not
PACKAGE LEAFLET: INFORMATION FOR THE USER. Aggrastat (250 micrograms/ml) concentrate for solution for infusion tirofiban
 PACKAGE LEAFLET: INFORMATION FOR THE USER Aggrastat (250 micrograms/ml) concentrate for solution for infusion tirofiban Read all of this leaflet carefully before you start using this medicine because it
PACKAGE LEAFLET: INFORMATION FOR THE USER Aggrastat (250 micrograms/ml) concentrate for solution for infusion tirofiban Read all of this leaflet carefully before you start using this medicine because it
S PECIAL A RTICLE. Combination Antiplatelet Therapy: Implications for Pharmacists
 S PECIAL A RTICLE Combination Antiplatelet Therapy: Implications for Pharmacists Robert L. Talbert, Pharm.D., FCCP, Sarah A. Spinler, Pharm.D., FCCP, Jean M. Nappi, Pharm.D., FCCP, and Michael B. Bottorff,
S PECIAL A RTICLE Combination Antiplatelet Therapy: Implications for Pharmacists Robert L. Talbert, Pharm.D., FCCP, Sarah A. Spinler, Pharm.D., FCCP, Jean M. Nappi, Pharm.D., FCCP, and Michael B. Bottorff,
Evidence-Based Management of CAD: Last Decade Trials and Updated Guidelines
 Evidence-Based Management of CAD: Last Decade Trials and Updated Guidelines Enrico Ferrari, MD Cardiac Surgery Unit Cardiocentro Ticino Foundation Lugano, Switzerland Conflict of Interests No conflict
Evidence-Based Management of CAD: Last Decade Trials and Updated Guidelines Enrico Ferrari, MD Cardiac Surgery Unit Cardiocentro Ticino Foundation Lugano, Switzerland Conflict of Interests No conflict
ADULT DRUG REFERENCE Drug Indication Adult Dosage Precautions / Comments
 ADENOSINE Paroxysmal SVT 1 st Dose 6 mg rapid IV 2 nd & 3 rd Doses 12 mg rapid IV push Follow each dose with rapid bolus of 20 ml NS May cause transient heart block or asystole. Side effects include chest
ADENOSINE Paroxysmal SVT 1 st Dose 6 mg rapid IV 2 nd & 3 rd Doses 12 mg rapid IV push Follow each dose with rapid bolus of 20 ml NS May cause transient heart block or asystole. Side effects include chest
Medicine Dr. Omed Lecture 2 Stable and Unstable Angina
 Medicine Dr. Omed Lecture 2 Stable and Unstable Angina Risk stratification in stable angina. High Risk; *post infarct angina, *poor effort tolerance, *ischemia at low workload, *left main or three vessel
Medicine Dr. Omed Lecture 2 Stable and Unstable Angina Risk stratification in stable angina. High Risk; *post infarct angina, *poor effort tolerance, *ischemia at low workload, *left main or three vessel
Diagnosis: Allergies with reaction type:
 Patient Name: Diagnosis: Allergies with reaction type: ICU Stroke-Ischemic S/P tpa Version 2 5/29/14 This order set is designed to be used with an admission set or for a patient already admitted Nursing
Patient Name: Diagnosis: Allergies with reaction type: ICU Stroke-Ischemic S/P tpa Version 2 5/29/14 This order set is designed to be used with an admission set or for a patient already admitted Nursing
Acute Coronary Syndrome (ACS) Initial Evaluation and Management
 Acute Coronary Syndrome (ACS) Initial Evaluation and Management Symptoms of Possible ACS Chest discomfort with or without radiation to the arm(s), jaw, or epigastrium Short of breath Weakness Diaphoresis
Acute Coronary Syndrome (ACS) Initial Evaluation and Management Symptoms of Possible ACS Chest discomfort with or without radiation to the arm(s), jaw, or epigastrium Short of breath Weakness Diaphoresis
NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOL USE OF PROPOFOL (DIPRIVAN) FOR VENTILATOR MANAGEMENT
 NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOL I. PURPOSE: To provide guidelines for the administration of Propofol, which is an anesthetic agent, indicated for the continuous intravenous
NURSING DEPARTMENT CRITICAL CARE POLICY MANUAL CRITICAL CARE PROTOCOL I. PURPOSE: To provide guidelines for the administration of Propofol, which is an anesthetic agent, indicated for the continuous intravenous
UTMB DEPARTMENT OF PHARMACY POLICY AND PROCEDURES Section Medication Use ADMINISTRATION OF MEDICATION BY IV PUSH
 Page 1 of 8 ADMINISTRATION OF MEDICATION BY IV PUSH AUDIENCE POLICY This document is directed to Registered Nurses (RNs), Licensed Vocational Nurses (LVN s), and physicians. The following delineates the
Page 1 of 8 ADMINISTRATION OF MEDICATION BY IV PUSH AUDIENCE POLICY This document is directed to Registered Nurses (RNs), Licensed Vocational Nurses (LVN s), and physicians. The following delineates the
2016 Physician Quality Reporting System Data Collection Form: Coronary Artery Bypass Graft (CABG) (for patients aged 18 years and older)
 2016 Physician Quality Reporting System Data Collection Form: Coronary Artery Bypass Graft (CABG) (for patients aged 18 years and older) IMPORTANT: Any measure with a 0% performance rate (100% for inverse
2016 Physician Quality Reporting System Data Collection Form: Coronary Artery Bypass Graft (CABG) (for patients aged 18 years and older) IMPORTANT: Any measure with a 0% performance rate (100% for inverse
NOTICES DEPARTMENT OF HEALTH
 NOTICES DEPARTMENT OF HEALTH Approved Drugs for ALS Ambulance Services [42 Pa.B. 4229] [Saturday, July 7, 2012] Under 28 Pa. Code 1005.11(b) (relating to drug use, control and security), the following
NOTICES DEPARTMENT OF HEALTH Approved Drugs for ALS Ambulance Services [42 Pa.B. 4229] [Saturday, July 7, 2012] Under 28 Pa. Code 1005.11(b) (relating to drug use, control and security), the following
Chest Pain Acute Coronary Syndrome Version 4 4/10/17 This order set is designed to be used with an admission set or for a patient already admitted
 Patient Name: Diagnosis: Allergies with reaction type: Chest Pain Acute Coronary Syndrome Version 4 4/10/17 This order set is designed to be used with an admission set or for a patient already admitted
Patient Name: Diagnosis: Allergies with reaction type: Chest Pain Acute Coronary Syndrome Version 4 4/10/17 This order set is designed to be used with an admission set or for a patient already admitted
Atrial Fibrillation Version 2 11/4/15 This order set must be used with an admission order set if patient not already admitted.
 Patient Name: Diagnosis: Allergies with reaction type: Atrial Fibrillation Version 2 11/4/15 This order set must be used with an admission order set if patient not already admitted. Telemetry Medical Telemetry:
Patient Name: Diagnosis: Allergies with reaction type: Atrial Fibrillation Version 2 11/4/15 This order set must be used with an admission order set if patient not already admitted. Telemetry Medical Telemetry:
Chapter 10 Worksheet Blood Pressure and Antithrombotic Agents
 Complete the following. 1. A layer of cells lines each vessel in the vascular system. This layer is a passive barrier that keeps cells and proteins from going into tissues; it also contains substances
Complete the following. 1. A layer of cells lines each vessel in the vascular system. This layer is a passive barrier that keeps cells and proteins from going into tissues; it also contains substances
Supplementary Table 1. Details of the components of the primary composite endpoint
 Supplementary Table 1. Details of the components of the primary composite endpoint 1. Death The cause of death will be defined by the underlying cause, not the immediate mode of death. Death will be classified
Supplementary Table 1. Details of the components of the primary composite endpoint 1. Death The cause of death will be defined by the underlying cause, not the immediate mode of death. Death will be classified
Belinda Green, Cardiologist, SDHB, 2016
 Acute Coronary syndromes All STEMI ALL Non STEMI Unstable angina Belinda Green, Cardiologist, SDHB, 2016 Thrombus in proximal LAD Underlying pathophysiology Be very afraid for your patient Wellens
Acute Coronary syndromes All STEMI ALL Non STEMI Unstable angina Belinda Green, Cardiologist, SDHB, 2016 Thrombus in proximal LAD Underlying pathophysiology Be very afraid for your patient Wellens
PRODUCT INFORMATION. Concentrate for IV infusion. AGGRASTAT (tirofiban hydrochloride) DESCRIPTION
 ARS-012001s 1 PRODUCT INFORMATION Concentrate for IV infusion AGGRASTAT (tirofiban hydrochloride) DESCRIPTION AGGRASTAT (tirofiban hydrochloride, MSD), a non-peptide antagonist of the platelet glycoprotein
ARS-012001s 1 PRODUCT INFORMATION Concentrate for IV infusion AGGRASTAT (tirofiban hydrochloride) DESCRIPTION AGGRASTAT (tirofiban hydrochloride, MSD), a non-peptide antagonist of the platelet glycoprotein
Treatment Strategies for the Prevention of Ischemic Complications in Patients Undergoing Percutaneous Coronary Intervention with Stent Placement
 Treatment Strategies for the Prevention of Ischemic Complications in Patients Undergoing Percutaneous Coronary Intervention with Stent Placement Pharmaceutical Care Project Outcomes Literature Evaluation
Treatment Strategies for the Prevention of Ischemic Complications in Patients Undergoing Percutaneous Coronary Intervention with Stent Placement Pharmaceutical Care Project Outcomes Literature Evaluation
PHYSICIAN SIGNATURE DATE TIME DRUG ALLERGIES WT: KG
 Available at ALL facilities Non Categorized SUB ED Chest Pain: STEMI Protocol(SUB)* SUB ED Chest Pain: STEMI Protocol Lab Orders(SUB)* ED Rainbow Tubes(SUB)* ***Reminder: Order ED Rainbow Tubes (SUB) as
Available at ALL facilities Non Categorized SUB ED Chest Pain: STEMI Protocol(SUB)* SUB ED Chest Pain: STEMI Protocol Lab Orders(SUB)* ED Rainbow Tubes(SUB)* ***Reminder: Order ED Rainbow Tubes (SUB) as
PACKAGE LEAFLET: INFORMATION FOR THE USER. Aggrastat (50 micrograms/ml) solution for infusion tirofiban
 PACKAGE LEAFLET: INFORMATION FOR THE USER Aggrastat (50 micrograms/ml) solution for infusion tirofiban Read all of this leaflet carefully before you start using this medicine because it contains important
PACKAGE LEAFLET: INFORMATION FOR THE USER Aggrastat (50 micrograms/ml) solution for infusion tirofiban Read all of this leaflet carefully before you start using this medicine because it contains important
Medical Management of Acute Coronary Syndrome: The roles of a noncardiologist. Norbert Lingling D. Uy, MD Professor of Medicine UERMMMCI
 Medical Management of Acute Coronary Syndrome: The roles of a noncardiologist physician Norbert Lingling D. Uy, MD Professor of Medicine UERMMMCI Outcome objectives of the discussion: At the end of the
Medical Management of Acute Coronary Syndrome: The roles of a noncardiologist physician Norbert Lingling D. Uy, MD Professor of Medicine UERMMMCI Outcome objectives of the discussion: At the end of the
Access Site Complications. Nick Cheshire MD FRCS, Professor of Vascular Surgery Imperial College Healthcare St Mary s Hospital Campus
 Access Site Complications Nick Cheshire MD FRCS, Professor of Vascular Surgery Imperial College Healthcare St Mary s Hospital Campus Surgeons & Cardiologists Interventional Cardiology 2008 Eye of a Hawk,
Access Site Complications Nick Cheshire MD FRCS, Professor of Vascular Surgery Imperial College Healthcare St Mary s Hospital Campus Surgeons & Cardiologists Interventional Cardiology 2008 Eye of a Hawk,
FULL PRESCRIBING INFORMATION: CONTENTS* 1 INDICATIONS AND USAGE. 1.1 Percutaneous Transluminal Coronary Angioplasty (PTCA)
 451129B/Revised: June 2016 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use BIVALIRUDIN FOR INJECTION safely and effectively. See full prescribing
451129B/Revised: June 2016 HIGHLIGHTS OF PRESCRIBING INFORMATION These highlights do not include all the information needed to use BIVALIRUDIN FOR INJECTION safely and effectively. See full prescribing
PEDIATRIC LIFEGIFT BRAIN DEATH PLAN
 PHYSICIAN S Diagnosis Weight Allergies Admit/Discharge/Transfer THIS PLAN IS TO BE ED ONLY ON THE LIFEGIFT ENCOUNTER, WITH DR LIFEGIFT AS THE ATTENDING. Patient Status Pt Status: Inpatient (Inpatient only
PHYSICIAN S Diagnosis Weight Allergies Admit/Discharge/Transfer THIS PLAN IS TO BE ED ONLY ON THE LIFEGIFT ENCOUNTER, WITH DR LIFEGIFT AS THE ATTENDING. Patient Status Pt Status: Inpatient (Inpatient only
Continuing Medical Education Post-Test
 Continuing Medical Education Post-Test Based on the information presented in this monograph, please choose one correct response for each of the following questions or statements. Record your answers on
Continuing Medical Education Post-Test Based on the information presented in this monograph, please choose one correct response for each of the following questions or statements. Record your answers on
STANDARDIZED PROCEDURE CARDIAC STRESS TESTING-DOBUTAMINE INFUSION (Adult)
 I. Definition: This test is performed to evaluate for cardiac ischemia, arrhythmias, and/or response to exercise. II. Background Information A. Setting: The setting (inpatient vs outpatient) and population
I. Definition: This test is performed to evaluate for cardiac ischemia, arrhythmias, and/or response to exercise. II. Background Information A. Setting: The setting (inpatient vs outpatient) and population
IR Central Venous Access [ ] Pre Procedure
![IR Central Venous Access [ ] Pre Procedure IR Central Venous Access [ ] Pre Procedure](/thumbs/90/104119758.jpg) IR Central Venous Access [1050200001] Pre Procedure Case Request/Scheduling Procedure Enter IR Case Request if not already completed (All hospitals except Grant Medical Center) [ ] Case Request IR Lab
IR Central Venous Access [1050200001] Pre Procedure Case Request/Scheduling Procedure Enter IR Case Request if not already completed (All hospitals except Grant Medical Center) [ ] Case Request IR Lab
Thrombolysis administration
 Thrombolysis administration Liz Mackey Stroke Nurse Practitioner Western Health Sunshine & Footscray Hospital, Melbourne Thanks ASNEN committee members Skye Coote, Acute Stroke Nurse, Eastern Health (slide
Thrombolysis administration Liz Mackey Stroke Nurse Practitioner Western Health Sunshine & Footscray Hospital, Melbourne Thanks ASNEN committee members Skye Coote, Acute Stroke Nurse, Eastern Health (slide
Therapeutic Hypothermia for Post Cardiac Arrest Plan Initial Orders
 Arrest Plan Initial Orders Weight Allergies Therapeutic Hypothermia Guidelines ***Required to continue with ordering Plan.*** Strict Intake and Output q1h, throughout cooling and re warming. Set Up for
Arrest Plan Initial Orders Weight Allergies Therapeutic Hypothermia Guidelines ***Required to continue with ordering Plan.*** Strict Intake and Output q1h, throughout cooling and re warming. Set Up for
Patient characteristics Intervention Comparison Length of followup
 ANTICOAGULANT CHAPTER Low Molecular Weight Heparins compared with Unfractionated Heparin Ref ID: 1903 Reference Antman EM, McCabe CH, Gurfinkel EP et al. Enoxaparin prevents death and cardiac ischemic
ANTICOAGULANT CHAPTER Low Molecular Weight Heparins compared with Unfractionated Heparin Ref ID: 1903 Reference Antman EM, McCabe CH, Gurfinkel EP et al. Enoxaparin prevents death and cardiac ischemic
Case report. Open Access. Abstract
 Open Access Case report A retrospective analysis and case series of glycoprotein IIb/IIIa inhibitor associated diffuse alveolar hemorrhage: two case reports Hima Mikkilineni, Steven R Bruhl, William R
Open Access Case report A retrospective analysis and case series of glycoprotein IIb/IIIa inhibitor associated diffuse alveolar hemorrhage: two case reports Hima Mikkilineni, Steven R Bruhl, William R
Medical Management of Acute Heart Failure
 Critical Care Medicine and Trauma Medical Management of Acute Heart Failure Mary O. Gray, MD, FAHA Associate Professor of Medicine University of California, San Francisco Staff Cardiologist and Training
Critical Care Medicine and Trauma Medical Management of Acute Heart Failure Mary O. Gray, MD, FAHA Associate Professor of Medicine University of California, San Francisco Staff Cardiologist and Training
DAY1_CARDIOVASCULAR PRACTICE QUESTIONS
 DAY1_CARDIOVASCULAR PRACTICE QUESTIONS 1 P age 1. A 59-year-old male is admitted complaining of chest pain and dyspnea. ST elevation and T-wave inversion were seen on the ECG in V2, V3, and V4. IV thrombolytic
DAY1_CARDIOVASCULAR PRACTICE QUESTIONS 1 P age 1. A 59-year-old male is admitted complaining of chest pain and dyspnea. ST elevation and T-wave inversion were seen on the ECG in V2, V3, and V4. IV thrombolytic
2010, Metzler Helfried
 Perioperative Strategies in Patients on Dual Antiplatelet Drug Therapy: Noncardiac Surgery H. Metzler Department of Anaesthesiology and Intensive Care Medicine Medical University of Graz, Austria What
Perioperative Strategies in Patients on Dual Antiplatelet Drug Therapy: Noncardiac Surgery H. Metzler Department of Anaesthesiology and Intensive Care Medicine Medical University of Graz, Austria What
ST Elevation Myocardial Infarction (STEMI) Reperfusion Order Set
 Form Title Form Number CH-0454 2018, Alberta Health Services, CKCM This work is licensed under the Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License. The license does not
Form Title Form Number CH-0454 2018, Alberta Health Services, CKCM This work is licensed under the Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License. The license does not
New Antithrombotic and Antiplatelet Drugs in CAD : (Factor Xa inhibitors, Direct Thrombin inhibitors and Prasugrel)
 New Antithrombotic and Antiplatelet Drugs in CAD : (Factor Xa inhibitors, Direct Thrombin inhibitors and Prasugrel) Limitations and Advantages of UFH and LMWH Biological limitations of UFH : 1. immune-mediated
New Antithrombotic and Antiplatelet Drugs in CAD : (Factor Xa inhibitors, Direct Thrombin inhibitors and Prasugrel) Limitations and Advantages of UFH and LMWH Biological limitations of UFH : 1. immune-mediated
Scottish Medicines Consortium
 Scottish Medicines Consortium bivalirudin 250mg for injection or infusion (Angiox ) (156/05) Nycomed UK Ltd No. 4 February, 2005 The Scottish Medicines Consortium has completed its assessment of the above
Scottish Medicines Consortium bivalirudin 250mg for injection or infusion (Angiox ) (156/05) Nycomed UK Ltd No. 4 February, 2005 The Scottish Medicines Consortium has completed its assessment of the above
** Medication exercises ** NICU Phase II
 ** Medication exercises ** NICU Phase II A) Baby A has had a bowel resection six hours ago. She weighs 3 kg. Postop, she is quite agitated and she is on a fentanyl drip at 2 micrograms/kg/hr (#1). She
** Medication exercises ** NICU Phase II A) Baby A has had a bowel resection six hours ago. She weighs 3 kg. Postop, she is quite agitated and she is on a fentanyl drip at 2 micrograms/kg/hr (#1). She
Cardiac Catheterization, Coronary Angiogram, and Percutaneous Coronary Intervention (PCI)
 Cardiac Catheterization, Coronary Angiogram, and Percutaneous Coronary Intervention (PCI) Cardiac Catheterization, Coronary Angiogram, and Percutaneous Coronary Intervention (PCI) Your doctor has recommended
Cardiac Catheterization, Coronary Angiogram, and Percutaneous Coronary Intervention (PCI) Cardiac Catheterization, Coronary Angiogram, and Percutaneous Coronary Intervention (PCI) Your doctor has recommended
DOBUTamine INJECTION, USP R x only
 DOBUTamine INJECTION, USP R x only DESCRIPTION Dobutamine Injection, USP is 1,2-benzenediol, 4-[2-[[3-(4-hydro-xyphenyl)-1- methylpropyl]amino]ethyl]-hydrochloride, (±). It is a synthetic catecholamine.
DOBUTamine INJECTION, USP R x only DESCRIPTION Dobutamine Injection, USP is 1,2-benzenediol, 4-[2-[[3-(4-hydro-xyphenyl)-1- methylpropyl]amino]ethyl]-hydrochloride, (±). It is a synthetic catecholamine.
Cardiovascular Concerns in Intermediate Care
 Cardiovascular Concerns in Intermediate Care GINA ST. JEAN RN, MSN, CCRN-CSC CLINICAL NURSE EDUCATOR HEART AND & CRITICAL AND INTERMEDIATE CARE Objectives: Identify how to do a thorough assessment of the
Cardiovascular Concerns in Intermediate Care GINA ST. JEAN RN, MSN, CCRN-CSC CLINICAL NURSE EDUCATOR HEART AND & CRITICAL AND INTERMEDIATE CARE Objectives: Identify how to do a thorough assessment of the
PHYSICIAN SIGNATURE DATE TIME DRUG ALLERGIES WT: KG
 Available at: BMC-B BMC-D BMC-N BMC-S Activity Activity Bedrest with BRP, with assistance at nurse's discretion (DEF)* Ambulate with Assistance Diet Communication Order Patient to remain NPO while in PACU
Available at: BMC-B BMC-D BMC-N BMC-S Activity Activity Bedrest with BRP, with assistance at nurse's discretion (DEF)* Ambulate with Assistance Diet Communication Order Patient to remain NPO while in PACU
ARGATROBAN Injection PRESCRIBING INFORMATION
 ARGATROBAN Injection PRESCRIBING INFORMATION DESCRIPTION Argatroban is a synthetic direct thrombin inhibitor derived from L-arginine. The chemical name for Argatroban is 1-[5-[(aminoiminomethyl)amino]-1-oxo-2-[[(1,2,3,4-tetrahydro-3-
ARGATROBAN Injection PRESCRIBING INFORMATION DESCRIPTION Argatroban is a synthetic direct thrombin inhibitor derived from L-arginine. The chemical name for Argatroban is 1-[5-[(aminoiminomethyl)amino]-1-oxo-2-[[(1,2,3,4-tetrahydro-3-
bivalirudin 250mg powder for concentrate for solution for injection or infusion (Angiox) SMC No. (638/10) The Medicines Company
 bivalirudin 250mg powder for concentrate for solution for injection or infusion (Angiox) SMC No. (638/10) The Medicines Company 06 August 2010 The Scottish Medicines Consortium (SMC) has completed its
bivalirudin 250mg powder for concentrate for solution for injection or infusion (Angiox) SMC No. (638/10) The Medicines Company 06 August 2010 The Scottish Medicines Consortium (SMC) has completed its
Preoperative Cardiac Risk Assessment: Approach & Guidelines
 Preoperative Cardiac Risk Assessment: Approach & Guidelines By, Liam Morris, MD., FACC (02/03/18) CPG : Clinical Practice Guidelines GDMT : Guidelines Directed Medical Therapy GWC : Guideline Writing Committee
Preoperative Cardiac Risk Assessment: Approach & Guidelines By, Liam Morris, MD., FACC (02/03/18) CPG : Clinical Practice Guidelines GDMT : Guidelines Directed Medical Therapy GWC : Guideline Writing Committee
Clopidogrel and ASA after CABG for NSTEMI
 Clopidogrel and ASA after CABG for NSTEMI May 17, 2007 Justin Lee Pharmacy Resident University Health Network Objectives At the end of this session, you should be able to: Explain the rationale for antiplatelet
Clopidogrel and ASA after CABG for NSTEMI May 17, 2007 Justin Lee Pharmacy Resident University Health Network Objectives At the end of this session, you should be able to: Explain the rationale for antiplatelet
TRANSPARENCY COMMITTEE OPINION. 29 April 2009
 The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 29 April 2009 ANGIOX 250 mg, powder for concentrate for solution for injection or infusion by the intravenous route
The legally binding text is the original French version TRANSPARENCY COMMITTEE OPINION 29 April 2009 ANGIOX 250 mg, powder for concentrate for solution for injection or infusion by the intravenous route
Clinical Lessons from BMC2-PCI
 Clinical Lessons from BMC2-PCI The Blue Cross Blue Shield of Michigan Cardiovascular Consortium Hitinder Gurm, M.D. University of Michigan Overview 32 papers since inception 10 papers published this year
Clinical Lessons from BMC2-PCI The Blue Cross Blue Shield of Michigan Cardiovascular Consortium Hitinder Gurm, M.D. University of Michigan Overview 32 papers since inception 10 papers published this year
Clinical Therapeutics/Volume 31, Number 8, 2009
 Clinical Therapeutics/Volume 31, Number 8, 2009 Assessment of the Bioequivalence of Brand and Biogeneric Formulations of Abciximab for the Treatment of Acute Coronary Syndrome: A Prospective, Open-Label,
Clinical Therapeutics/Volume 31, Number 8, 2009 Assessment of the Bioequivalence of Brand and Biogeneric Formulations of Abciximab for the Treatment of Acute Coronary Syndrome: A Prospective, Open-Label,
Blanchard Valley Hospital Pharmacy Code Blue Overview
 Blanchard Valley Hospital Pharmacy Code Blue Overview Adapted from Gary Spanik, RPh Edited by Kate Reeves, Pharm D, RPh Jon Manocchio, Pharm D, RPh Pharmacist Responsibilities Be aware of basic ACLS tenets
Blanchard Valley Hospital Pharmacy Code Blue Overview Adapted from Gary Spanik, RPh Edited by Kate Reeves, Pharm D, RPh Jon Manocchio, Pharm D, RPh Pharmacist Responsibilities Be aware of basic ACLS tenets
* * FORM REV. 02/2019 Page 1 of 4. TNKASE (tenecteplase) / ACUTE STEMI ORDERS SCHEDULED MEDICATIONS:
 1. Is this a CMS inpatient only procedure? Yes, admit as inpatient, proceed to # 3 No, proceed to # 2 2. Do you expect that the patient s condition will require a hospital stay that will cross two midnights
1. Is this a CMS inpatient only procedure? Yes, admit as inpatient, proceed to # 3 No, proceed to # 2 2. Do you expect that the patient s condition will require a hospital stay that will cross two midnights
Update on Antithrombotic Therapy in Acute Coronary Syndrome
 Update on Antithrombotic Therapy in Acute Coronary Syndrome Laura Tsang November 13, 2006 Objectives: By the end of this session, you should understand: The role of antithrombotics in ACS Their mechanisms
Update on Antithrombotic Therapy in Acute Coronary Syndrome Laura Tsang November 13, 2006 Objectives: By the end of this session, you should understand: The role of antithrombotics in ACS Their mechanisms
** Medication exercises ** NICU Phase II
 ** Medication exercises ** NICU Phase II A) Baby A has had a bowel resection six hours ago. She weighs 3 kg. Post-op, she is quite agitated and she is on a fentanyl drip at 2 micrograms/kg/hr (#1). She
** Medication exercises ** NICU Phase II A) Baby A has had a bowel resection six hours ago. She weighs 3 kg. Post-op, she is quite agitated and she is on a fentanyl drip at 2 micrograms/kg/hr (#1). She
POST-OP CARDIAC SURGERY PHYSICIAN S ORDER SHEET USE BALLPOINT PEN ONLY. CARDIAC INTENSIVE CARE UNIT
 PHYSICIAN S SHEET Automatically Activate, if not in agreement, cross out and initial Activated by Checking Box ALLERGIES: None known YES Patient s Height: Patient s Weight: ALL MEDICATION and INTRAVENOUS
PHYSICIAN S SHEET Automatically Activate, if not in agreement, cross out and initial Activated by Checking Box ALLERGIES: None known YES Patient s Height: Patient s Weight: ALL MEDICATION and INTRAVENOUS
Cardiology. Self Learning Package. Module 5: Pharmacology: Treatment of Acute Coronary. Prevention
 Cardiology Self Learning Package Module 5: Pharmacology: Treatment of Acute Coronary Syndromes, Module 5: Pharmacology: Hyperlipidaemia, Treatment of Acute Coronary Hypertension, Symdrome, Hyperlipidaemia,
Cardiology Self Learning Package Module 5: Pharmacology: Treatment of Acute Coronary Syndromes, Module 5: Pharmacology: Hyperlipidaemia, Treatment of Acute Coronary Hypertension, Symdrome, Hyperlipidaemia,
What oral antiplatelet therapy would you choose? a) ASA alone b) ASA + Clopidogrel c) ASA + Prasugrel d) ASA + Ticagrelor
 76 year old female Prior Hypertension, Hyperlipidemia, Smoking On Hydrochlorothiazide, Atorvastatin New onset chest discomfort; 2 episodes in past 24 hours Heart rate 122/min; BP 170/92 mm Hg, Killip Class
76 year old female Prior Hypertension, Hyperlipidemia, Smoking On Hydrochlorothiazide, Atorvastatin New onset chest discomfort; 2 episodes in past 24 hours Heart rate 122/min; BP 170/92 mm Hg, Killip Class
