Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider
|
|
|
- Lee Terry
- 5 years ago
- Views:
Transcription
1 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISEASE/CONDITION POPULATION TRIAD Submitting laboratory: Cambridge RGC Approved: September Disease/condition approved name and symbol as published on the OMIM database (alternative names will be listed on the UKGTN website) 2. OMIM number for disease/condition Disease/condition please provide a brief description of the characteristics of the disease/condition and prognosis for affected individuals. Please provide this information in laymen s terms. 46, XY SEX REVERSAL 3; SRXY3 46,XY disorder of sex development (46,XY DSD) is characterized by a 46, XY karyotype and ambiguous genitalia with mild to severe penoscrotal hypospadias with or without chordee, dysgenetic testes, reduced to no sperm production, and müllerian structures that range from absent to presence of a fully developed uterus and fallopian tubes. 46, XY complete gonadal dysgenesis (46, XY CGD) is characterized by a 46, XY karyotype, normal female external genitalia, completely undeveloped ( streak ) gonads, no sperm production, and presence of normal müllerian structures. 4. Disease/condition mode of inheritance Autosomal recessive/dominant 5. Gene approved name(s) and symbol as Nuclear receptor subfamily 5, group A, Member 1 published on HUGO database (alternative NR5A1 names will be listed on the UKGTN website) 6. OMIM number for gene(s) Gene description(s) NR5A1 is an orphan nuclear receptor which regulates multiple genes involved in adrenal and gonadal development. It has 6 functioning exons (2,3,4,5,6,7) split into 10 PCR reactions. 7b. Number of amplicons to provide this 10 amplicons test 7c. MolU/Cyto band that this test is assigned to These would all be in band D 8. Mutational spectrum for which you test We will screen for nonsense and missense mutations, including details of known common splice site mutations, small insertions and deletions in mutations these genes. We will also offer testing for whole exon deletions and duplications. 9. Technical method(s) Fluorescent sequence analysis using mutation 10. Validation process Please explain how this test has been validated for use in your laboratory 11a. Are you providing this test already? Yes surveyor. MLPA analysis using MRC Holland kit. Sequence and dosage analysis is used for mutation testing in a wide range of disorders in our laboratory therefore these methods are already validated within the lab.
2 11b. If yes, how many reports have you produced? 11c. Number of reports mutation positive 6 11d. Number of reports mutation negative For how long have you been providing this service? 13a. Is there specialised local clinical/research expertise for this disease? 25 on a research basis 2 years Yes 13b. If yes, please provide details Professor Hughes in University of Cambridge Dept Paediatrics in conjunction with John Achermann, Institute of Child Health, London. 14. Are you testing for other genes/diseases/conditions closely allied to this one? Please give details Your current activity If applicable - How many tests do you currently provide annually in your laboratory? Yes. Androgen Receptor, SRD5A2, HSD17B3 and SRY in addition of NR5A1 will complete a package of candidate gene analysis relevant to a molecular diagnosis of XY DSD. 15a. Index cases 25 15b. Family members where mutation is 10 known Your capacity if Gene Dossier approved We will be able to meet the capacity for the UK How many tests will you be able to provide annually in your laboratory if this gene dossier is approved and recommended for NHS funding? 16a. Index cases 16b. Family members where mutation is known Based on experience how many tests will be required nationally (UK wide) per annum? Please identify the information on which this is based 17a. Index cases 50 17b. Family members where mutation is 30 known 18. National activity (England, Scotland, Wales & Northern Ireland) If your laboratory is unable to provide the full national need please could you provide information on how the national requirement may be met. For example, are you aware of any other labs (UKGTN members or otherwise) offering this test to NHS patients on a local area basis only? This question has been included in order to gauge if there could be any issues in equity of access for NHS patients. It is appreciated that some laboratories may not be able to answer this question. If this is the case please write unknown Based on Cambridge DSD database gonadal dysgenesis cases, this number is constantly changing as the parameters for the phenotype are wide.
3 EPIDEMIOLOGY 19. Estimated prevalence of condition in the general UK population Please identify the information on which this is based 20. Estimated gene frequency (Carrier frequency or allele frequency) Please identify the information on which this is based 21. Estimated penetrance Please identify the information on which this is based 22. Estimated prevalence of condition in the target population. The target population is the group of people that meet the minimum criteria as listed in the Testing Criteria. Unknown Not known N/A Estimated prevalence is difficult to state based on the definition of the disease condition quoted in Item 3. Overall, the prevalence of NR5A1 mutations in patients with a clinical diagnosis of gonadal dysgenesis is 10-15% from published studies. The Cambridge series of mutations found in 6 patients with partial gonadal dysgenesis (based on a robust clinical diagnosis) reports a higher prevalence of 24%. INTENDED USE 23. Please tick the relevant clinical purpose of testing Diagnosis Yes No Treatment x Yes No Prognosis & management Yes No Presymptomatic testing Yes x No Carrier testing for family members Yes No Prenatal testing Yes No
4 TEST CHARACTERISTICS 24. Analytical sensitivity and specificity This should be based on your own laboratory data for the specific test being applied for or the analytical sensitivity and specificity of the method/technique to be used in the case of a test yet to be set up. >99% for uni-directional sequencing analysed with Mutation Surveyor 25. Clinical sensitivity and specificity of test in target population The clinical sensitivity of a test is the probability of a positive test result when condition is known to be present; the clinical specificity is the probability of a negative test result when disease is known to be absent. The denominator in this case is the number with the disease (for sensitivity) or the number without condition (for specificity). Not possible to provide figures because of difficulties currently in defining the denominator in view of the widely defined clinical phenotype (see 22). 26. Clinical validity (positive and negative predictive value in the target population) The clinical validity of a genetic test is a measure of how well the test predicts the presence or absence of the phenotype, clinical condition or predisposition. It is measured by its positive predictive value (the probability of getting the condition given a positive test) and negative predictive value (the probability of not getting the condition given a negative test). As in Testing pathway for tests where more than one gene is to be tested Please include your testing strategy if more than one gene will be tested and data on the expected proportions of positive results for each part of the process. Please illustrate this with a flow diagram. The clinical entry point would be abnormal genitalia at birth with biochemical/imaging tests suggestive of gonadal dysgenesis. If there is evidence of adrenal insufficiency, this would raise the stakes for a mutation. At an older age, problems occurring at puberty with virilisation of an XY child raised female would be the clinical entry point. How analysis of NR5A1 may inter-relate with testing other genes is illustrated in the flow diagram. Ambiguous genitalia (XY) Virilisation at puberty (XY female) Normal androgens Abnormal androgens USP 5 -reductase Normal USP AR, SRD5A2 No uterus Uterus SRD5A2 HSD17B3, AR, NR5A1 Normal HSD17133 SRY, NR5A1 NR5A1 NB: Increasingly, it is becoming evident that NR5A1 should be tested early in the investigation pathway.
5 CLINICAL UTILITY 28. How will the test add to the management of the patient or alter clinical outcome? Establishing a specific diagnosis is important in XY DSD. This provides data for genetic counselling and decisions on sex assignment (this has life-long implications). Gonadal tumour risk is genemutation dependent (high in SRY and probably NR5A1 mutations, low in HSD17B3, SRD5A2). This in turn informs decisions about gonadectomy and its timing. There is a risk of life-threatening adrenal insufficiency associated with an NR5A1 mutation. The management and clinical outcome implications are self-evidence in this context. 29. How will the availability of this test impact on patient and family life? Families want to have a diagnosis. Especially important in XY DSD where the cause is still unknown in at least 40% of cases. NR5A1 analysis will improve that yield. 30. Benefits of the test Please provide a summary of the overall benefits of this test. Diagnosis, accurate genetic counselling, appropriate sex assignment, tumour risk. 31. Is there an alternative means of diagnosis or prediction that does not involve molecular diagnosis? If so (and in particular if there is a biochemical test), please state the added advantage of the molecular test. The indirect tests are too non-specific; currently will result in defining a general category of gonadal dysgenesis. 32. Please describe any specific ethical, legal or social issues with this particular test. None that is different to testing for other candidate genes in DSD. 33. The Testing Criteria must be completed where Testing Criteria are not already available. If Testing Criteria are available, do you agree with them Yes/No If No: Please propose alternative Testing Criteria AND please explain here the reasons for the changes. 34. Savings or investment per annum in the diagnostic pathway based on national expected activity, cost of diagnostics avoided and cost of genetic test. Please show calculations. Avoid a step-wise analysis of other candidate genes. For example, analysing AR, HSD17B3 and SRD5A2 would be an unnecessary cost of 1300 per patient investigated. A conservative annual saving would be 26,000 (based on 20 patient samples). Since the phenotype associated with NR5A1 mutations can be so variable, the flow diagram in box 27 is a suggested outline that will at least target those 46, XY DSD genes where mutations are associated with a more specific phenotype. Consequently, analysing AR, HSD17B3 and SRD5A2 when not supported by adequate phenotyping (clinical and biochemical) would generate an unnecessary cost of 1300 per patient investigated. An estimated annual saving would be 65,000.
6 35. List the diagnostic tests/procedures that would no longer be required with costs Costs and type of imaging procedures: Pelvic US and MRI 1200 Costs and types of laboratory pathology tests: Hormone analyses 500 (other than molecular/cyto genetic proposed in this gene dossier) Costs and types of physiological tests (e.g. ECG) N/A Cost and types of other investigations/procedures (e.g. biopsy): Occasional 400 biopsy Total cost tests/procedures no longer required REAL LIFE CASE STUDY In collaboration with the clinical lead, describe a real case example to illustrate how the test would improve patient experience. A 14 year old girl started to virilise at puberty. Investigations showed a 46, XY karyotype and testosterone was elevated, increasing further with HCG stimulation. The differential included partial androgen insensitivity syndrome, 5 -reductase deficiency and 17β-hydroxysteroid dehydrogenase deficiency. A likely genetic cause was strengthened by her 12 year old sister also have a 46, XY karyotype. Several urinary steroid analyses were performed, numerous imaging studies to identify the site of gonads and the internal genital anatomy. Sequencing of the AR, SRD5A2 and HSD17B3 genes was normal. During the investigative period extending over 18 months, clinical management was a challenge to control the effects of virilisation which can be irreversible (eg, voice deepening, jaw enlargement). Analysis of the NR5A1 gene revealed a heterozygous point mutation (Gln329X) in exon 5 resulting in a premature stop codon. There was now a specific diagnosis and explanation for virilisation in 2 affected sisters. Both are at risk of adrenal insufficiency; assessment of adrenal reserve by ACTH stimulation is currently normal but will need to be monitored regularly. 37. For the case example, if there are cost savings, please provide these below: Figures are based on cost savings for both affected sisters. PRE GENETIC TEST Costs and type of imaging procedures US, MRI 2000 Costs and type of laboratory pathology tests USP, plasma steroids 3000 (other than molecular/cyto genetic proposed in this gene dossier) Costs and type of physiological tests (e.g. ECG) Cost and type of other investigations/procedures (e.g. biopsy) - gonads 1200 Cost outpatient consultations (genetics and non genetics) 2600 Total cost pre genetic test 8800 POST GENETIC TEST Costs and type of imaging procedures Costs and types laboratory pathology tests monitoring steroid levels 600 (other than molecular/cyto genetic proposed in this gene dossier) Cost of genetic test proposing in this gene dossier Carrier detection 300 Costs and type of physiological tests (e.g. ECG) Cost and type of other investigations/procedures (e.g. biopsy) Cost outpatient consultations (genetics and non genetics) 1200 Total cost post genetic test Estimated savings for case example described 7000 approximately
7 UKGTN Testing Criteria Approved name and symbol of disease/condition(s): 46,XY Sex Reversal 3; SRXY3 Approved name and symbol of gene(s): nuclear receptor subfamily 5, group A, member 1 NR5A1 OMIM number(s): OMIM number(s): Patient name: Patient postcode: Date of birth: NHS number: Name of referrer: Title/Position: Lab ID: Referrals will only be accepted from one of the following: Referrer Clinical geneticist Endocrinologist (paediatric/adult) Tick if this refers to you. Minimum criteria required for testing to be appropriate as stated in the Gene Dossier: Criteria 46,XY karyotype AND evidence of gonadal dysgenesis (uterus, FSH, low testosterone, low AMH, gonadal histology if available). At risk family members where familial mutation is known Tick if this patient meets criteria If the sample does not fulfil the clinical criteria or you are not one of the specified types of referrer and you still feel that testing should be performed please contact the laboratory to discuss testing of the sample
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISEASE/CONDITION POPULATION TRIAD Submitting laboratory: London North East RGC GOSH Approved: September
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISEASE/CONDITION POPULATION TRIAD Submitting laboratory: London North East RGC GOSH Approved: September
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISORDER/CONDITION POPULATION TRIAD Submitting laboratory: Exeter RGC Approved: Sept 2013 1. Disorder/condition
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISORDER/CONDITION POPULATION TRIAD Submitting laboratory: Exeter RGC Approved: Sept 2013 1. Disorder/condition
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Epileptic encephalopathy, early infantile 4. OMIM number for disease 612164 Disease
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Epileptic encephalopathy, early infantile 4. OMIM number for disease 612164 Disease
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISEASE/CONDITION POPULATION TRIAD Submitting laboratory: Birmingham RGC Approved: September 2012
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISEASE/CONDITION POPULATION TRIAD Submitting laboratory: Birmingham RGC Approved: September 2012
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name HEMOCHROMATOSIS, TYPE 4; HFE4 OMIM number for disease #606069 Disease alternative
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name HEMOCHROMATOSIS, TYPE 4; HFE4 OMIM number for disease #606069 Disease alternative
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Leber congenital amaurosis OMIM number for disease 204000 Disease alternative
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Leber congenital amaurosis OMIM number for disease 204000 Disease alternative
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISEASE/CONDITION POPULATION TRIAD Submitting laboratory: London North East RGC GOSH Approved: September
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISEASE/CONDITION POPULATION TRIAD Submitting laboratory: London North East RGC GOSH Approved: September
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Choroideremia OMIM number for disease 303100 Disease alternative names please
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Choroideremia OMIM number for disease 303100 Disease alternative names please
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Genetic Causes of Hypothyroidism 1. Loss of function mutations in TSHR cause thyroid
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Genetic Causes of Hypothyroidism 1. Loss of function mutations in TSHR cause thyroid
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Amyotrophic Lateral Sclerosis 10 (ALS10) and Amyotrophic Lateral Sclerosis 6 (ALS6)
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Amyotrophic Lateral Sclerosis 10 (ALS10) and Amyotrophic Lateral Sclerosis 6 (ALS6)
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISEASE/CONDITION POPULATION TRIAD Submitting laboratory: Manchester RGC Approved: September 2013
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISEASE/CONDITION POPULATION TRIAD Submitting laboratory: Manchester RGC Approved: September 2013
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Parkinson disease 8, automsomal dominant OMIM number for disease 607060 Disease
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Parkinson disease 8, automsomal dominant OMIM number for disease 607060 Disease
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names you wish listed) (A)-Testing
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names you wish listed) (A)-Testing
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name OMIM number for disease 231300 Disease alternative names Please provide any alternative
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name OMIM number for disease 231300 Disease alternative names Please provide any alternative
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name OMIM number for disease Disease alternative names please provide any alternative
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name OMIM number for disease Disease alternative names please provide any alternative
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names Osteogenesis Imperfecta
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names Osteogenesis Imperfecta
Submitting Laboratory: London NE RGC GOSH
 Submitting laboratory: London NE RGC GOSH 1. Disorder/condition approved name (please provide UK spelling if different from US) and symbol as published on the OMIM database (alternative names will be listed
Submitting laboratory: London NE RGC GOSH 1. Disorder/condition approved name (please provide UK spelling if different from US) and symbol as published on the OMIM database (alternative names will be listed
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names you wish listed) Glucocorticoid-remediable
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names you wish listed) Glucocorticoid-remediable
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name OMIM number for disease 147920 Disease alternative names Please provide any alternative
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name OMIM number for disease 147920 Disease alternative names Please provide any alternative
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISORDER/CONDITION POPULATION TRIAD Submitting laboratory: Cardiff SAS Porphyria Approved: Sept
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISORDER/CONDITION POPULATION TRIAD Submitting laboratory: Cardiff SAS Porphyria Approved: Sept
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Loeys-Dietz Syndrome OMIM number for disease 609192; 608967; 610380; 610168 Disease
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Loeys-Dietz Syndrome OMIM number for disease 609192; 608967; 610380; 610168 Disease
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names you wish listed) (A)-Testing
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names you wish listed) (A)-Testing
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names you wish listed) (A)-Testing
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names you wish listed) (A)-Testing
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names you wish listed) (A)-Testing
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names you wish listed) (A)-Testing
Application to be an additional provider for existing test on the NHS Directory of Molecular Genetic Testing Additional Provider form
 Application to be an additional provider for existing test on the NHS Directory of Molecular Genetic Testing Additional Provider form Disease: Gene: Cystic Fibrosis (CF) (carrier testing in reproductive
Application to be an additional provider for existing test on the NHS Directory of Molecular Genetic Testing Additional Provider form Disease: Gene: Cystic Fibrosis (CF) (carrier testing in reproductive
When testes make no testosterone: Identifying a rare cause of 46, XY female phenotype in adulthood
 When testes make no testosterone: Identifying a rare cause of 46, XY female phenotype in adulthood Gardner DG, Shoback D. Greenspan's Basic & Clinical Endocrinology, 10e; 2017 Sira Korpaisarn, MD Endocrinology
When testes make no testosterone: Identifying a rare cause of 46, XY female phenotype in adulthood Gardner DG, Shoback D. Greenspan's Basic & Clinical Endocrinology, 10e; 2017 Sira Korpaisarn, MD Endocrinology
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Polycystic Kidney Disease, Autosomal Dominant OMIM number for disease 173900 Disease
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name Polycystic Kidney Disease, Autosomal Dominant OMIM number for disease 173900 Disease
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names you wish listed) (A)-Testing
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name and description (please provide any alternative names you wish listed) (A)-Testing
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name OMIM number for disease 312060 Disease alternative names please provide any alternative
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Test Disease Population Triad Disease name OMIM number for disease 312060 Disease alternative names please provide any alternative
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Submitting laboratory: Bristol RGC 1. Disorder/condition approved name and symbol as published on the OMIM database (alternative
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Submitting laboratory: Bristol RGC 1. Disorder/condition approved name and symbol as published on the OMIM database (alternative
Disordered Sex Differentiation Mixed gonadal dysgenesis Congenital adrenal hyperplasia Mixed gonadal dysgenesis
 Disordered Sex Differentiation DSD has superceded intersex in describing genital anomalies in childhood DSD results from hormonal imbalances due to (i) abnormal genetic status, (ii) enzyme defects, or
Disordered Sex Differentiation DSD has superceded intersex in describing genital anomalies in childhood DSD results from hormonal imbalances due to (i) abnormal genetic status, (ii) enzyme defects, or
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISORDER/CONDITION POPULATION TRIAD Submitting laboratory: Bristol RGC Approved: Sept 2013 1. Disorder/condition
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider TEST DISORDER/CONDITION POPULATION TRIAD Submitting laboratory: Bristol RGC Approved: Sept 2013 1. Disorder/condition
46,XY Female: SRY and AR Basis: Genotype & Phenotype Correlation
 Original Article Indian Journal of Genetics and Molecular Research 53 Volume 1 Number 2, July - December 2012 46,XY Female: SRY and AR Basis: Genotype & Phenotype Correlation Amudha S.*, MSc; Sayee Rajangam**,
Original Article Indian Journal of Genetics and Molecular Research 53 Volume 1 Number 2, July - December 2012 46,XY Female: SRY and AR Basis: Genotype & Phenotype Correlation Amudha S.*, MSc; Sayee Rajangam**,
Let s Talk About Hormones!
 Let s Talk About Hormones! This lesson was created by Serena Reves and Nichelle Penney, with materials from the BCTF and The Pride Education Network. Hormones are responsible for the regulation of many
Let s Talk About Hormones! This lesson was created by Serena Reves and Nichelle Penney, with materials from the BCTF and The Pride Education Network. Hormones are responsible for the regulation of many
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider Submitting laboratory: Sheffield RGC 1. Disorder/condition approved name (please provide UK spelling
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider Submitting laboratory: Sheffield RGC 1. Disorder/condition approved name (please provide UK spelling
DEVELOPMENT (DSD) 1 4 DISORDERS OF SEX
 Wit JM, Ranke MB, Kelnar CJH (eds): ESPE classification of paediatric endocrine diagnosis. 4. Disorders of sex development (DSD). Horm Res 2007;68(suppl 2):21 24 ESPE Code Diagnosis OMIM ICD 10 4 DISORDERS
Wit JM, Ranke MB, Kelnar CJH (eds): ESPE classification of paediatric endocrine diagnosis. 4. Disorders of sex development (DSD). Horm Res 2007;68(suppl 2):21 24 ESPE Code Diagnosis OMIM ICD 10 4 DISORDERS
IN SUMMARY HST 071 NORMAL & ABNORMAL SEXUAL DIFFERENTIATION Fetal Sex Differentiation Postnatal Diagnosis and Management of Intersex Abnormalities
 Harvard-MIT Division of Health Sciences and Technology HST.071: Human Reproductive Biology Course Director: Professor Henry Klapholz IN SUMMARY HST 071 Title: Fetal Sex Differentiation Postnatal Diagnosis
Harvard-MIT Division of Health Sciences and Technology HST.071: Human Reproductive Biology Course Director: Professor Henry Klapholz IN SUMMARY HST 071 Title: Fetal Sex Differentiation Postnatal Diagnosis
A National Model of Care for Paediatric Healthcare Services in Ireland Chapter 28: Paediatric Gynaecology
 A National Model of Care for Paediatric Healthcare Services in Ireland Chapter 28: Paediatric Gynaecology Clinical Strategy and Programmes Division Table of Contents 28.0 Introduction 2 28.1 Current Service
A National Model of Care for Paediatric Healthcare Services in Ireland Chapter 28: Paediatric Gynaecology Clinical Strategy and Programmes Division Table of Contents 28.0 Introduction 2 28.1 Current Service
Management of gonads in DSD
 Management of gonads in DSD Martine Cools, paediatric endocrinologist, Katja Wolffenbuttel and Piet Hoebeke, paediatric urologists, all at University Hospital Ghent, Belgium Sten Drop, paediatric endocrinologist
Management of gonads in DSD Martine Cools, paediatric endocrinologist, Katja Wolffenbuttel and Piet Hoebeke, paediatric urologists, all at University Hospital Ghent, Belgium Sten Drop, paediatric endocrinologist
Approach to Disorders of Sex Development (DSD)
 Approach to Disorders of Sex Development (DSD) Old name: The Approach to Intersex Disorders Dr. Abdulmoein Al-Agha, FRCP Ass. Professor & Consultant Pediatric Endocrinologist, KAUH, Erfan Hospital & Ibn
Approach to Disorders of Sex Development (DSD) Old name: The Approach to Intersex Disorders Dr. Abdulmoein Al-Agha, FRCP Ass. Professor & Consultant Pediatric Endocrinologist, KAUH, Erfan Hospital & Ibn
PHYSIOLOGY AND PATHOLOGY OF SEXUAL DIFFERENTIATION
 PHYSIOLOGY AND PATHOLOGY OF SEXUAL DIFFERENTIATION Prof. Dr med. Jolanta Słowikowska-Hilczer Department of Andrology and Reproductive Endocrinology Medical University of Łódź, Poland Sexual determination
PHYSIOLOGY AND PATHOLOGY OF SEXUAL DIFFERENTIATION Prof. Dr med. Jolanta Słowikowska-Hilczer Department of Andrology and Reproductive Endocrinology Medical University of Łódź, Poland Sexual determination
Clinical Standards for Service Planning in PAG
 The British Society for Paediatric & Adolescent Gynaecology. Clinical Standards for Service Planning in PAG Introduction The management of young children and adolescents with gynaecological problems (aged
The British Society for Paediatric & Adolescent Gynaecology. Clinical Standards for Service Planning in PAG Introduction The management of young children and adolescents with gynaecological problems (aged
Chapter 18 Development. Sexual Differentiation
 Chapter 18 Development Sexual Differentiation There Are Many Levels of Sex Determination Chromosomal Sex Gonadal Sex Internal Sex Organs External Sex Organs Brain Sex Gender Identity Gender Preference
Chapter 18 Development Sexual Differentiation There Are Many Levels of Sex Determination Chromosomal Sex Gonadal Sex Internal Sex Organs External Sex Organs Brain Sex Gender Identity Gender Preference
Bios 90/95. Jennifer Swann, PhD
 Sexual Differentiation Fall 2007 Bios 90/95 Jennifer Swann, PhD Dept Biol Sci, Lehigh University Why have sexes? What determines sex? Environment Genetics Hormones What causes these differences? The true
Sexual Differentiation Fall 2007 Bios 90/95 Jennifer Swann, PhD Dept Biol Sci, Lehigh University Why have sexes? What determines sex? Environment Genetics Hormones What causes these differences? The true
DEFINITION: Masculinization of external genitalia in patients with normal 46XX karyotype.
 INTERSEX DISORDERS DEFINITION: Masculinization of external genitalia in patients with normal 46XX karyotype. - Degree of masculinization variable: - mild clitoromegaly - complete fusion of labia folds
INTERSEX DISORDERS DEFINITION: Masculinization of external genitalia in patients with normal 46XX karyotype. - Degree of masculinization variable: - mild clitoromegaly - complete fusion of labia folds
FLASH CARDS. Kalat s Book Chapter 11 Alphabetical
 FLASH CARDS www.biologicalpsych.com Kalat s Book Chapter 11 Alphabetical alpha-fetoprotein alpha-fetoprotein Alpha-Fetal Protein (AFP) or alpha-1- fetoprotein. During a prenatal sensitive period, estradiol
FLASH CARDS www.biologicalpsych.com Kalat s Book Chapter 11 Alphabetical alpha-fetoprotein alpha-fetoprotein Alpha-Fetal Protein (AFP) or alpha-1- fetoprotein. During a prenatal sensitive period, estradiol
AMBIGUOUS GENITALIA. Dr. HAKIMI, SpAK. Dr. MELDA DELIANA, SpAK
 AMBIGUOUS GENITALIA (DISORDERS OF SEXUAL DEVELOPMENT) Dr. HAKIMI, SpAK Dr. MELDA DELIANA, SpAK Dr. SISKA MAYASARI LUBIS, SpA Pediatric Endocrinology division USU/H. ADAM MALIK HOSPITAL 1 INTRODUCTION Normal
AMBIGUOUS GENITALIA (DISORDERS OF SEXUAL DEVELOPMENT) Dr. HAKIMI, SpAK Dr. MELDA DELIANA, SpAK Dr. SISKA MAYASARI LUBIS, SpA Pediatric Endocrinology division USU/H. ADAM MALIK HOSPITAL 1 INTRODUCTION Normal
1) Intersexuality - Dr. Huda
 1) Intersexuality - Dr. Huda DSD (Disorders of sex development) occur when there is disruption of either: Gonadal differentiation Fetal sex steroid production or action Mullerian abnormalities and Wolffian
1) Intersexuality - Dr. Huda DSD (Disorders of sex development) occur when there is disruption of either: Gonadal differentiation Fetal sex steroid production or action Mullerian abnormalities and Wolffian
MRC-Holland MLPA. Description version 08; 07 May 2015
 mix P185-C1 Intersex Lot C1-0611: As compared to the previous version B2 (lot B2-0311), s for CYP21A2 have been removed and s for the CXorf21 gene as well as additional s for NR0B1, NR5A1 and the Y chromosome
mix P185-C1 Intersex Lot C1-0611: As compared to the previous version B2 (lot B2-0311), s for CYP21A2 have been removed and s for the CXorf21 gene as well as additional s for NR0B1, NR5A1 and the Y chromosome
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider Submitting laboratory: Leeds RGC 1. Disorder/condition approved name (please provide UK spelling if different
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider Submitting laboratory: Leeds RGC 1. Disorder/condition approved name (please provide UK spelling if different
Intersex Genital Mutilations in ICD-10 Zwischengeschlecht.org / StopIGM.org 2014 (v2.1)
 Intersex Genital Mutilations in ICD-10 Zwischengeschlecht.org / StopIGM.org 2014 (v2.1) ICD-10 Codes and Descriptions: http://apps.who.int/classifications/icd10/browse/2010/en 1. Reference: 17 Most Common
Intersex Genital Mutilations in ICD-10 Zwischengeschlecht.org / StopIGM.org 2014 (v2.1) ICD-10 Codes and Descriptions: http://apps.who.int/classifications/icd10/browse/2010/en 1. Reference: 17 Most Common
SALSA MLPA probemix P185-C2 Intersex Lot C2-1015: As compared to the previous version C1 (lot C1-0611), the lengths of four probes have been adjusted.
 mix P185-C2 Intersex Lot C2-1015: As compared to the previous version C1 (lot C1-0611), the lengths of four s have been adjusted. The sex-determining region on chromosome Y (SRY) is the most important
mix P185-C2 Intersex Lot C2-1015: As compared to the previous version C1 (lot C1-0611), the lengths of four s have been adjusted. The sex-determining region on chromosome Y (SRY) is the most important
Sex chromosomes and sex determination
 Sex chromosomes and sex determination History (1) 1940-ties Alfred Jost embryo-surgical experiments on gonads gonadal sex; male gonadal sex presence of testes; female gonadal sex lack of testes. History
Sex chromosomes and sex determination History (1) 1940-ties Alfred Jost embryo-surgical experiments on gonads gonadal sex; male gonadal sex presence of testes; female gonadal sex lack of testes. History
GENDER James Bier
 GENDER 2005-2008 James Bier Objectives 1. State the method of determining gender in several genetic systems. 2. List the three regions of the Y chromosome. 3. Describe the events that promote sexual development
GENDER 2005-2008 James Bier Objectives 1. State the method of determining gender in several genetic systems. 2. List the three regions of the Y chromosome. 3. Describe the events that promote sexual development
UCL. Gerard Conway. Treatment of AIS patients in multidisciplinary teams in UK. Tratamiento del SIA en equipos multidisciplinares en el Reino Unido
 UCL Treatment of AIS patients in multidisciplinary teams in UK Tratamiento del SIA en equipos multidisciplinares en el Reino Unido Gerard Conway London, UK University College London Hospitals University
UCL Treatment of AIS patients in multidisciplinary teams in UK Tratamiento del SIA en equipos multidisciplinares en el Reino Unido Gerard Conway London, UK University College London Hospitals University
Disclosure. Session Objectives. I have no actual or potential conflict of interest in relation to this program/presentation.
 46, XY Female: A Case of Complete Androgen Insensitivity Syndrome (CAIS) MICHELLE MCLOUGHLINMSN, CRNP, CPNP-AC THE CHILDREN S HOSPITAL OF PHILADELPHIA DIVISION OF ENDOCRINOLOGY AND DIABETES Disclosure
46, XY Female: A Case of Complete Androgen Insensitivity Syndrome (CAIS) MICHELLE MCLOUGHLINMSN, CRNP, CPNP-AC THE CHILDREN S HOSPITAL OF PHILADELPHIA DIVISION OF ENDOCRINOLOGY AND DIABETES Disclosure
Best Practice & Research Clinical Endocrinology & Metabolism
 Best Practice & Research Clinical Endocrinology & Metabolism 24 (2010) 335 354 Contents lists available at ScienceDirect Best Practice & Research Clinical Endocrinology & Metabolism journal homepage: www.elsevier.com/locate/beem
Best Practice & Research Clinical Endocrinology & Metabolism 24 (2010) 335 354 Contents lists available at ScienceDirect Best Practice & Research Clinical Endocrinology & Metabolism journal homepage: www.elsevier.com/locate/beem
46,XY disorders of sex development (DSD)
 Clinical Endocrinology (2009) 70, 173 187 doi: 10.1111/j.1365-2265.2008.03392.x CLINICAL PRACTICE UPDATE Blackwell Publishing Ltd 46,XY disorders of sex development (DSD) Berenice Bilharinho Mendonca,
Clinical Endocrinology (2009) 70, 173 187 doi: 10.1111/j.1365-2265.2008.03392.x CLINICAL PRACTICE UPDATE Blackwell Publishing Ltd 46,XY disorders of sex development (DSD) Berenice Bilharinho Mendonca,
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Submitting laboratory: Bristol RGC 1. Disorder/condition approved name and symbol as published on the OMIM database (alternative
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier Submitting laboratory: Bristol RGC 1. Disorder/condition approved name and symbol as published on the OMIM database (alternative
2. Which male target tissues respond to testosterone, and which require dihydrotestosterone?
 308 PHYSIOLOGY CASES AND PROBLEMS Case 56 Male Pseudohermaphroditism: Sa-Reductase Deficiency Fourteen years ago, Wally and Wanda Garvey, who live in rural North Carolina, had their first child. The baby
308 PHYSIOLOGY CASES AND PROBLEMS Case 56 Male Pseudohermaphroditism: Sa-Reductase Deficiency Fourteen years ago, Wally and Wanda Garvey, who live in rural North Carolina, had their first child. The baby
Intersex is a group of conditions where there is a discrepancy between the external genitals and the internal genitals (the testes and ovaries).
 Intersex to use the sharing features on this page, please enable JavaScript. Share on facebookshare on IwitterBookmark & SharePrintcr-lnendly version Intersex is a group of conditions where
Intersex to use the sharing features on this page, please enable JavaScript. Share on facebookshare on IwitterBookmark & SharePrintcr-lnendly version Intersex is a group of conditions where
Sexual Differentiation Fall 2008 Bios 90
 Sexual Differentiation Fall 2008 Bios 90 Jennifer Swann, Professor Dept Biol Sci, Lehigh University Asexual reproduction Budding : offspring develop as a growth on the body of the parent jellyfishes, echinoderms,
Sexual Differentiation Fall 2008 Bios 90 Jennifer Swann, Professor Dept Biol Sci, Lehigh University Asexual reproduction Budding : offspring develop as a growth on the body of the parent jellyfishes, echinoderms,
Goals. Disorders of Sex Development (Intersex): An Overview. Joshua May, MD Pediatric Endocrinology
 Disorders of Sex Development (Intersex): An Overview Joshua May, MD Pediatric Endocrinology Murphy, et al., J Ped Adol Gynecol, 2011 Goals Objectives: Participants will be able to: 1. Apply the medical
Disorders of Sex Development (Intersex): An Overview Joshua May, MD Pediatric Endocrinology Murphy, et al., J Ped Adol Gynecol, 2011 Goals Objectives: Participants will be able to: 1. Apply the medical
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider Submitting laboratory: Birmingham RGC 1. Disorder/condition approved name (please provide UK spelling
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider Submitting laboratory: Birmingham RGC 1. Disorder/condition approved name (please provide UK spelling
State of the art review in gonadal dysgenesis: challenges in diagnosis and management
 McCann-Crosby et al. International Journal of Pediatric Endocrinology 2014, 2014:4 PES REVIEW State of the art review in gonadal dysgenesis: challenges in diagnosis and management Open Access Bonnie McCann-Crosby
McCann-Crosby et al. International Journal of Pediatric Endocrinology 2014, 2014:4 PES REVIEW State of the art review in gonadal dysgenesis: challenges in diagnosis and management Open Access Bonnie McCann-Crosby
Ch 20: Reproduction. Keypoints: Human Chromosomes Gametogenesis Fertilization Early development Parturition
 Ch 20: Reproduction Keypoints: Human Chromosomes Gametogenesis Fertilization Early development Parturition SLOs Contrast mitosis/meiosis, haploid/diploid, autosomes/sex chromosomes. Outline the hormonal
Ch 20: Reproduction Keypoints: Human Chromosomes Gametogenesis Fertilization Early development Parturition SLOs Contrast mitosis/meiosis, haploid/diploid, autosomes/sex chromosomes. Outline the hormonal
Information leaflet for patients and families. Congenital Adrenal Hyperplasia (CAH)
 Information leaflet for patients and families Congenital Adrenal Hyperplasia (CAH) What is CAH? CAH is a disorder causing impaired hormone production from the adrenal glands. Hormones are chemical messengers
Information leaflet for patients and families Congenital Adrenal Hyperplasia (CAH) What is CAH? CAH is a disorder causing impaired hormone production from the adrenal glands. Hormones are chemical messengers
Androgen insensitivity syndrome: a survey of diagnostic procedures and management in the UK
 Archives of Disease in Childhood 1997;77:305 309 305 Department of Paediatrics, University of Cambridge, Addenbrookes Hospital Y Teoh D M Williams M N Patterson I A Hughes University College London Hospitals,
Archives of Disease in Childhood 1997;77:305 309 305 Department of Paediatrics, University of Cambridge, Addenbrookes Hospital Y Teoh D M Williams M N Patterson I A Hughes University College London Hospitals,
172 Subject Index. Aromatase deficiency (cont.) paracrine and intracrine actions, 19 severe estrogen deficiency, 21 variations, 22
 Subject Index A ACTH-induced hypertension, 111 112 Androgen insensitivity syndrome (AIS), 63 androgen receptor (AR), 59 bone mineral apparent density (BMAD), 60 61 CAIS subjects, 59 61 estradiol levels,
Subject Index A ACTH-induced hypertension, 111 112 Androgen insensitivity syndrome (AIS), 63 androgen receptor (AR), 59 bone mineral apparent density (BMAD), 60 61 CAIS subjects, 59 61 estradiol levels,
Supplemental Data: Detailed Characteristics of Patients with MKRN3. Patient 1 was born after an uneventful pregnancy. She presented in our
 1 2 Supplemental Data: Detailed Characteristics of Patients with MKRN3 Mutations 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 Patient 1 was born after an uneventful pregnancy. She presented
1 2 Supplemental Data: Detailed Characteristics of Patients with MKRN3 Mutations 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 Patient 1 was born after an uneventful pregnancy. She presented
Mutations. A2 Biology For WJEC
 12. Mutation is a change in the amount, arrangement or structure in the DNA of an organism. 13. There are two types of mutations, chromosome mutations and gene mutations. Mutations A2 Biology For WJEC
12. Mutation is a change in the amount, arrangement or structure in the DNA of an organism. 13. There are two types of mutations, chromosome mutations and gene mutations. Mutations A2 Biology For WJEC
The Biology of Sex: How We Become Male or Female.
 The Biology of Sex: How We Become Male or Female. Dr. Tamatha Barbeau, Dept. of Biology Guest Lecture for Gender 200 March 2017 Objectives: 1. Sex vs. Gender defined. 2. Biological sex based on inheritance
The Biology of Sex: How We Become Male or Female. Dr. Tamatha Barbeau, Dept. of Biology Guest Lecture for Gender 200 March 2017 Objectives: 1. Sex vs. Gender defined. 2. Biological sex based on inheritance
Case Based Urology Learning Program
 Case Based Urology Learning Program Resident s Corner: UROLOGY Case Number 3 CBULP 2011 019 Case Based Urology Learning Program Editor: Associate Editors: Manager: Case Contributors: Steven C. Campbell,
Case Based Urology Learning Program Resident s Corner: UROLOGY Case Number 3 CBULP 2011 019 Case Based Urology Learning Program Editor: Associate Editors: Manager: Case Contributors: Steven C. Campbell,
Corporate Medical Policy
 Corporate Medical Policy Invasive Prenatal (Fetal) Diagnostic Testing File Name: Origination: Last CAP Review: Next CAP Review: Last Review: invasive_prenatal_(fetal)_diagnostic_testing 12/2014 3/2018
Corporate Medical Policy Invasive Prenatal (Fetal) Diagnostic Testing File Name: Origination: Last CAP Review: Next CAP Review: Last Review: invasive_prenatal_(fetal)_diagnostic_testing 12/2014 3/2018
Stump the Professors. Professor Panelists 4/13/2015. Challenger. Kylie Fowler, MD
 4/13/2015 Stump the Professors Professor Panelists Cynthia Holland-Hall MD, MPH Mary Anne Jamieson, MD Julie Strickland, MD, MPH Challenger Kylie Fowler, MD Medical degree: University of Washington School
4/13/2015 Stump the Professors Professor Panelists Cynthia Holland-Hall MD, MPH Mary Anne Jamieson, MD Julie Strickland, MD, MPH Challenger Kylie Fowler, MD Medical degree: University of Washington School
COMPLETE GONADAL DYSGENESIS WITH XY CHROMOSOMAL CONSTITUTION
 Tipar Cap coada final.qxd 1/22/2007 11:57 PM Page 465 Case report COMPLETE GONADAL DYSGENESIS WITH XY CHROMOSOMAL CONSTITUTION Dorina Stoicanescu *,1, Valerica Belengeanu 1, Dana Amzar 2, Cristina Popa
Tipar Cap coada final.qxd 1/22/2007 11:57 PM Page 465 Case report COMPLETE GONADAL DYSGENESIS WITH XY CHROMOSOMAL CONSTITUTION Dorina Stoicanescu *,1, Valerica Belengeanu 1, Dana Amzar 2, Cristina Popa
Disorder name: Congenital Adrenal Hyperplasia Acronym: CAH
 Genetic Fact Sheets for Parents Draft Disorders Screening, Technology, and Research in Genetics is a multi-state project to improve information about the financial, ethical, legal, and social issues surrounding
Genetic Fact Sheets for Parents Draft Disorders Screening, Technology, and Research in Genetics is a multi-state project to improve information about the financial, ethical, legal, and social issues surrounding
Disorders of Sex Development: not Always Endocrine Disorders
 ARC Journal of Diabetes and Endocrinology Volume 2, Issue 1, 2016, PP 14-18 ISSN 2455-5983 (Online) http://dx.doi.org/10.20431/2455-5983.0201002 www.arcjournals.org Disorders of Sex Development: not Always
ARC Journal of Diabetes and Endocrinology Volume 2, Issue 1, 2016, PP 14-18 ISSN 2455-5983 (Online) http://dx.doi.org/10.20431/2455-5983.0201002 www.arcjournals.org Disorders of Sex Development: not Always
Chromosomes and Human Inheritance. Chapter 11
 Chromosomes and Human Inheritance Chapter 11 11.1 Human Chromosomes Human body cells have 23 pairs of homologous chromosomes 22 pairs of autosomes 1 pair of sex chromosomes Autosomes and Sex Chromosomes
Chromosomes and Human Inheritance Chapter 11 11.1 Human Chromosomes Human body cells have 23 pairs of homologous chromosomes 22 pairs of autosomes 1 pair of sex chromosomes Autosomes and Sex Chromosomes
When menarche does not occur a pediatric view
 Peter Bang, MD, PhD, MSci Div of Pediatrics Dept of Clinical and Experimental Medicine Faculty of Health Sciences Linköping University peter.bang@liu.se Mobile +46708226491 Svensk Förening för Obstetrik
Peter Bang, MD, PhD, MSci Div of Pediatrics Dept of Clinical and Experimental Medicine Faculty of Health Sciences Linköping University peter.bang@liu.se Mobile +46708226491 Svensk Förening för Obstetrik
UKGTN Testing Criteria
 Test name: Neonatal Diabetes 22 Gene Panel UKGTN Testing Criteria Approved name and symbol of disorder/condition(s): See Appendix 1 Approved name and symbol of gene(s): See Appendix 1 number(s): number(s):
Test name: Neonatal Diabetes 22 Gene Panel UKGTN Testing Criteria Approved name and symbol of disorder/condition(s): See Appendix 1 Approved name and symbol of gene(s): See Appendix 1 number(s): number(s):
Martin Ritzén. bioscience explained Vol 7 No 2. Girl or boy: What guides gender development and how can this be a problem within
 Martin Ritzén Professor emeritus, Karolinska Institutet, Stockholm, Sweden Girl or boy: What guides gender development and how can this be a problem within sport? Introduction During the 2009 athletics
Martin Ritzén Professor emeritus, Karolinska Institutet, Stockholm, Sweden Girl or boy: What guides gender development and how can this be a problem within sport? Introduction During the 2009 athletics
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE. Proposed Highly Specialised Technology Evaluation
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Proposed Highly Specialised Technology Evaluation Drisapersen for treating Duchenne muscular Draft scope (pre-referral) Draft remit/evaluation objective
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Proposed Highly Specialised Technology Evaluation Drisapersen for treating Duchenne muscular Draft scope (pre-referral) Draft remit/evaluation objective
CLINICAL GENETICS 1. GENERAL ASPECTS OF THE SPECIALISATION. 1.1 Purpose of the specialisation
 CLINICAL GENETICS 1. GENERAL ASPECTS OF THE SPECIALISATION 1.1 Purpose of the specialisation The specialisation is an education and learning process in which trainee specialists (residents) acquire theoretical
CLINICAL GENETICS 1. GENERAL ASPECTS OF THE SPECIALISATION 1.1 Purpose of the specialisation The specialisation is an education and learning process in which trainee specialists (residents) acquire theoretical
SHARED CARE PRESCRIBING GUIDELINE Triptorelin (Gonapeptyl Depot 3.75 mg TM, Decapeptyl SR mg TM ) for precocious puberty
 WORKING IN PARTNERSHIP WITH SHARED CARE PRESCRIBING GUIDELINE Triptorelin (Gonapeptyl Depot 3.75 mg TM, Decapeptyl SR 11.25 mg TM ) for precocious puberty NHS Surrey s Medicines Management Committee classification:
WORKING IN PARTNERSHIP WITH SHARED CARE PRESCRIBING GUIDELINE Triptorelin (Gonapeptyl Depot 3.75 mg TM, Decapeptyl SR 11.25 mg TM ) for precocious puberty NHS Surrey s Medicines Management Committee classification:
C. Patrick Shahan, MD University of Tennessee Health Science Center Department of Surgery
 C. Patrick Shahan, MD University of Tennessee Health Science Center Department of Surgery Drop use of hermaphrodite and derivatives 1 in 15,000 live births Congenital Adrenal Hyperplasia Mixed Gonadal
C. Patrick Shahan, MD University of Tennessee Health Science Center Department of Surgery Drop use of hermaphrodite and derivatives 1 in 15,000 live births Congenital Adrenal Hyperplasia Mixed Gonadal
Sexual Development. 6 Stages of Development
 6 Sexual Development 6 Stages of Development Development passes through distinct stages, the first of which is fertilization, when one sperm enters one ovum. To enter an ovum, a sperm must undergo the
6 Sexual Development 6 Stages of Development Development passes through distinct stages, the first of which is fertilization, when one sperm enters one ovum. To enter an ovum, a sperm must undergo the
Analysis of the Sex-determining Region of the Y Chromosome (SRY) in a Case of 46, XX True Hermaphrodite
 Clin Pediatr Endocrinol 1994; 3(2): 91-95 Copyright (C) 1994 by The Japanese Society for Pediatric Endocrinology Analysis of the Sex-determining Region of the Y Chromosome (SRY) in a Case of 46, XX True
Clin Pediatr Endocrinol 1994; 3(2): 91-95 Copyright (C) 1994 by The Japanese Society for Pediatric Endocrinology Analysis of the Sex-determining Region of the Y Chromosome (SRY) in a Case of 46, XX True
MEDICAL GENOMICS LABORATORY. Next-Gen Sequencing and Deletion/Duplication Analysis of NF1 Only (NF1-NG)
 Next-Gen Sequencing and Deletion/Duplication Analysis of NF1 Only (NF1-NG) Ordering Information Acceptable specimen types: Fresh blood sample (3-6 ml EDTA; no time limitations associated with receipt)
Next-Gen Sequencing and Deletion/Duplication Analysis of NF1 Only (NF1-NG) Ordering Information Acceptable specimen types: Fresh blood sample (3-6 ml EDTA; no time limitations associated with receipt)
Long Term Follow-Up Clinical Guidelines for X-linked Adrenoleukodystrophy
 Long Term Follow-Up Clinical Guidelines for X-linked Adrenoleukodystrophy Gerald Raymond, M.D. Department of Pediatrics and Neurology Penn State Medical Center Hershey, PA June 20, 2018 Disclosure Information
Long Term Follow-Up Clinical Guidelines for X-linked Adrenoleukodystrophy Gerald Raymond, M.D. Department of Pediatrics and Neurology Penn State Medical Center Hershey, PA June 20, 2018 Disclosure Information
REPRODUCCIÓN. La idea fija. Copyright 2004 Pearson Education, Inc., publishing as Benjamin Cummings
 REPRODUCCIÓN La idea fija How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development, birth
REPRODUCCIÓN La idea fija How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development, birth
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider
 Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider Submitting laboratory: London NE RGC GOSH 1. Disorder/condition approved name (please provide UK spelling
Proposal form for the evaluation of a genetic test for NHS Service Gene Dossier/Additional Provider Submitting laboratory: London NE RGC GOSH 1. Disorder/condition approved name (please provide UK spelling
An easily missed diagnosis: 17-alpha-hydroxylase/17,20-lyase deficiency
 The Turkish Journal of Pediatrics 2015; 57: 277-281 Case Report An easily missed diagnosis: 17-alpha-hydroxylase/17,20-lyase deficiency Banu Küçükemre-Aydın 1, Özlem Öğrendil-Yanar 1, Ilmay Bilge 2, Firdevs
The Turkish Journal of Pediatrics 2015; 57: 277-281 Case Report An easily missed diagnosis: 17-alpha-hydroxylase/17,20-lyase deficiency Banu Küçükemre-Aydın 1, Özlem Öğrendil-Yanar 1, Ilmay Bilge 2, Firdevs
SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY
 1 SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY PBL SEMINAR: SEX HORMONES PART 1 An Overview What are steroid hormones? Steroid
1 SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY PBL SEMINAR: SEX HORMONES PART 1 An Overview What are steroid hormones? Steroid
Ambiguous genitalia (Disorders of sex development); is any case in which the external genitalia do not appear completely male or completely female.
 Ambiguous genitalia (Disorders of sex development); is any case in which the external genitalia do not appear completely male or completely female. Disorders of sex development (DSD); True hermaphrodite;
Ambiguous genitalia (Disorders of sex development); is any case in which the external genitalia do not appear completely male or completely female. Disorders of sex development (DSD); True hermaphrodite;
6.6 Hormones, homeostasis and reproduction
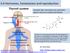 6.6 Hormones, homeostasis and reproduction Essential idea: Hormones are used when signals need to be widely distributed. Thyroxin is a hormone produced by the thyroid gland. It's key role is in controlling
6.6 Hormones, homeostasis and reproduction Essential idea: Hormones are used when signals need to be widely distributed. Thyroxin is a hormone produced by the thyroid gland. It's key role is in controlling
Clinical Guideline ADRENARCHE MANAGEMENT OF CHILDREN PRESENTING WITH SIGNS OF EARLY ONSET PUBIC HAIR/BODY ODOUR/ACNE
 Clinical Guideline ADRENARCHE MANAGEMENT OF CHILDREN PRESENTING WITH SIGNS OF EARLY ONSET PUBIC HAIR/BODY ODOUR/ACNE Includes guidance for the distinction between adrenarche, precocious puberty and other
Clinical Guideline ADRENARCHE MANAGEMENT OF CHILDREN PRESENTING WITH SIGNS OF EARLY ONSET PUBIC HAIR/BODY ODOUR/ACNE Includes guidance for the distinction between adrenarche, precocious puberty and other
Men s consent to the use and storage of sperm or embryos for surrogacy
 HFEA MSG form Men s consent to the use and storage of sperm or embryos for surrogacy About this form This form is produced by the Human Fertilisation and Embryology Authority (HFEA), the UK s independent
HFEA MSG form Men s consent to the use and storage of sperm or embryos for surrogacy About this form This form is produced by the Human Fertilisation and Embryology Authority (HFEA), the UK s independent
SISTEMA REPRODUCTOR (LA IDEA FIJA) Copyright 2004 Pearson Education, Inc., publishing as Benjamin Cummings
 SISTEMA REPRODUCTOR (LA IDEA FIJA) How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development,
SISTEMA REPRODUCTOR (LA IDEA FIJA) How male and female reproductive systems differentiate The reproductive organs and how they work How gametes are produced and fertilized Pregnancy, stages of development,
Analysis of exon 1 mutation in NR5A1 gene of Ambiguous Genitalia patients using SSCP analysis
 International Journal of ChemTech Research CODEN (USA): IJCRGG ISSN : 0974-4290 Vol.6, No.8, pp 4015-4021, September 2014 ICSET-2014 [6th 7th May 2014] International Conference on Science, Engineering
International Journal of ChemTech Research CODEN (USA): IJCRGG ISSN : 0974-4290 Vol.6, No.8, pp 4015-4021, September 2014 ICSET-2014 [6th 7th May 2014] International Conference on Science, Engineering
