Name: Mode of Study: Student Number: Qualification: Instruction: Answer all the questions on the questions paper.
|
|
|
- Wilfrid Hicks
- 5 years ago
- Views:
Transcription
1 Name: Mode of Study: Student Number: Qualification: Instruction: Answer all the questions on the questions paper. SECTION A: BIOLOGY QUESTION 1 [20 MARKS] Question Type: Multiple Choices. Each answer equals 2 marks. 1.1 A substance needed by the body for growth, energy, repair and maintenance is called a. [2] A. nutrient B. carbohydrate C. calorie D. lipid 1.2 All of the following are nutrients found in food except. [2] A. proteins B. carbohydrates C. plasma D. vitamins 1.3 A diet high in saturated fats can be linked to which of the following? [2] A. kidney failure B. rickets C. anorexia D. cardiovascular disease 1.4 A mineral that the body needs to work properly especially to absorb Vitamin D is. [2] A. calcium B. iron C. magnesium D. lead 1.5 Fibre cannot actually be digested; however, it is an important part of our diet for various reasons. One of the following reasons is wrong, identify which. [2] A. As it remains undigested it passes through the entire gut from mouth to anus and thus keeps food moving smoothly through our system. B. It causes constipation. C. The fibre absorbs poisonous waste from the digested food. D. High fibre diets are believed to reduce the risk of heart disease, bowel cancer and cholesterol in the body. Page 1 of 6
2 1.6 One function of a carbohydrate is. [2] A. keep the heart functioning smoothly B. store and transport genetic material C. to provide the body with immediate energy D. control the rate of reactions 1.7 The three types of carbohydrates are. [2] A. glycerol, polysaccharide, monosaccharide B. disaccharide, monosaccharide, glycerol C. glycerol, monosaccharide, polysaccharide D. monosaccharide, polysaccharide, disaccharide 1.8 What elements make up a carbohydrate? [2] A. hydrogen, calcium, oxygen B. carbon, potassium, oxygen C. hydrogen, oxygen, carbon D. calcium, potassium, oxygen 1.9 Glycogen, a polysaccharide, in your liver may be broken down to glucose by the process of. [2] A. hydrolysis B. dehydration synthesis C. condensation D. isomerization 1.10 Water makes up roughly 65% of our body weight. We take in water when we eat and drink. All the following statements are correct except [2] A. Water is important because chemical reactions in our cells take place in water. B. Waste products are passed out of our bodies in blood. C. Our blood transports substances that are dissolved in water D. Water is in sweat that cools us down SECTION B: CHEMISTRY QUESTION 1 [20 MARKS] Question Type: Multiple Choices. Each answer equals 2 marks. 1.1 In the metric system, the basic unit for mass is the. [2] A. Gram B. Pound C. Ton D. Kilogram Page 2 of 6
3 1.2 Which is NOT a unit of measurement in the SI system? [2] A. inch B. meter C. g/ml D. g/cm How many millimeters are in 24 cm? [2] A. 240 B C. 24,000 D. 240, The amount of space an object occupies is known as its. [2] A. density B. volume C. mass D. time 1.5 The ability of a measurement to be consistently reproduced is referred to as. A. accuracy B. qualitative C. quantitative D. precision 1.6 The amount of gravitational force on an object is known as its. [2] A. mass B. mole C. weight D. area 1.7 What is the density of a 61.8 g cube that is 2.3 cm wide, 4.0 cm tall, and 3.2 cm long? [2] A. 6.5 g/cm 3 B. 2.1 g/cm 3 C g/cm 3 D. None of the above 1.8 Which of the following measurements has the greatest number of significant figures? [2] A ml B ml C ml D x 10 9 ml [2] Page 3 of 6
4 1.9 If the temperature of a freezer is equal to o F, what would the temperature of a freezer be in o C? [2] A o C B o C C o C D. None of the above 1.10 a cyclist travels a distance of 0.5 Km in a time of 0.5 hours. The speed of the cyclist in SI units is equal to. [2] A m/s B. 0.3 m/s 2 C. 1 Km/h D. None of the above SECTION C: PHYSICS QUESTION 1 [20 MARKS] Question Type: Multiple Choices. Each answer equals 2 marks. 1. A disturbance that transfers energy through a medium from one location to the other is known as [2] (a) Gas (b) Sound (c) Potential energy (d) Wave 2. Waves that are produced by means of charged particle accelerations [2] (a) Mechanical waves (b) Ocean waves (c) Electromagnetic waves (d) S-Type earthquake waves 3. Which type of wave vibrates forming 90 o to the direction of motion of the wave? [2] (a) Transverse wave (b) Longitudinal wave (c) Both transverse and longitudinal wave (d) None of the above Page 4 of 6
5 4. A distance between any two repeating points on a wave [2] (a) Amplitude (b) Wavelength (c) Velocity (d) Frequency 5. Mechanical waves travel slower through [2] (a) Gas (b) Solid (c) Liquid (d) Denser mediums 6. What is the wavelength of a sound wave with a frequency of 0.05 KHz? (Speed of sound is 900 Km/h). [2] (a) 230 m (b) 50 Hz (c) 250 m/s (d) 5 m 7. A sound wave in a steel rail has a frequency of 620 Hz and a wavelength of 10.5 x 10 6 μm. What is the speed of sound in steel? [2] (a) 6.51 x 10 9 m/s (b) 6.51 x 10 3 m/s (c) 1.69 x 10 4 m/s (d) 5.90 x 10-5 m/s 8. Energy transferred from the sun to the surface of the earth is known as [2] (a) Sound energy (b) Solar energy (c) Earthquake (d) Potential energy 9. Earthquake waves travel in the form of [2] (a) Microwaves (b) Longitudinal waves (c) Light waves (d) Transverse waves Page 5 of 6
6 10. When molecules are given extra space and allowed to expand. [2] (a) Rarefaction (b) Compression (c) Crest (d) Trough TOTAL MARKS: 60 THE END Page 6 of 6
Digestive and Excretory Systems
 Digestive and Excretory Systems Homeostasis Q: How are the materials that enter and leave your body related to the processes that maintain homeostasis? 30.1 How is the human body organized and regulated?
Digestive and Excretory Systems Homeostasis Q: How are the materials that enter and leave your body related to the processes that maintain homeostasis? 30.1 How is the human body organized and regulated?
namibia UniVERSITY OF SCIEnCE AnD TECHnOLOGY
 ' I namibia UniVERSITY OF SCIEnCE AnD TECHnOLOGY FACULTY OF HEALTH AND APPLIED SCIENCES DEPARTMENT OF NATURAL AND APPLIED SCIENCES QUALIFICATION: QUALIFICATION CODE: LEVEL: 4 COURSE CODE: BSC410S COURSE
' I namibia UniVERSITY OF SCIEnCE AnD TECHnOLOGY FACULTY OF HEALTH AND APPLIED SCIENCES DEPARTMENT OF NATURAL AND APPLIED SCIENCES QUALIFICATION: QUALIFICATION CODE: LEVEL: 4 COURSE CODE: BSC410S COURSE
Choosing What You Eat and Why. Chapter 1 BIOL1400 Dr. Mohamad H. Termos
 Choosing What You Eat and Why Chapter 1 BIOL1400 Dr. Mohamad H. Termos Objectives Following this lecture, you should be able to describe: - Nutrition definition - Sources of nutrients - Energy sources
Choosing What You Eat and Why Chapter 1 BIOL1400 Dr. Mohamad H. Termos Objectives Following this lecture, you should be able to describe: - Nutrition definition - Sources of nutrients - Energy sources
30.1 Organization of the Human Body
 30.1 Organization of the Human Body Organization of the Body The levels of organization in the body include cells, tissues, organs, and organ systems. At each level of organization, these parts of the
30.1 Organization of the Human Body Organization of the Body The levels of organization in the body include cells, tissues, organs, and organ systems. At each level of organization, these parts of the
Nutrition, Nutrition, Nutrition! Because food is life! Oh, I m hungry!
 Nutrition, Nutrition, Nutrition! Because food is life! Oh, I m hungry! Topics of Study 1. What is metabolism? 2. Energy and chemical changes 3. Nutrients needed for a healthy lifestyle 4. Calories and
Nutrition, Nutrition, Nutrition! Because food is life! Oh, I m hungry! Topics of Study 1. What is metabolism? 2. Energy and chemical changes 3. Nutrients needed for a healthy lifestyle 4. Calories and
UNIT 2 DIABETES REVIEW
 UNIT 2 DIABETES REVIEW Pancreas is unable to make insulin. Therefore, glucose cannot get into the cells for energy. Insulin is made, but cell receptors do not work at getting recognizing that insulin.
UNIT 2 DIABETES REVIEW Pancreas is unable to make insulin. Therefore, glucose cannot get into the cells for energy. Insulin is made, but cell receptors do not work at getting recognizing that insulin.
Ch 2 Molecules of life
 Ch 2 Molecules of life Think about (Ch 2, p.2) 1. Water is essential to life. If there is water on a planet, it is possible that life may exist on the planet. 2. Water makes up the largest percentage by
Ch 2 Molecules of life Think about (Ch 2, p.2) 1. Water is essential to life. If there is water on a planet, it is possible that life may exist on the planet. 2. Water makes up the largest percentage by
Note-Taking Strategy. You will receive another guided note sheet to record all notes. Anything that is green should be recorded.
 Note-Taking Strategy You will receive another guided note sheet to record all notes. Anything that is green should be recorded. Giving Your Body What It Needs Each of the six nutrients has a specific job
Note-Taking Strategy You will receive another guided note sheet to record all notes. Anything that is green should be recorded. Giving Your Body What It Needs Each of the six nutrients has a specific job
Lesson 3 Understanding Nutrients and Their Importance
 Unit B Understanding Animal Body Systems Lesson 3 Understanding Nutrients and Their Importance 1 Terms Balanced ration Carbohydrates Complex carbohydrates Disaccharides Essential nutrients Ether Fat Fat-soluble
Unit B Understanding Animal Body Systems Lesson 3 Understanding Nutrients and Their Importance 1 Terms Balanced ration Carbohydrates Complex carbohydrates Disaccharides Essential nutrients Ether Fat Fat-soluble
Proteins. Biomolecules. Nucleic Acids. The Building Blocks of Life
 Proteins Biomolecules Nucleic Acids The Building Blocks of Life Carbohydrates Lipids Biomolecules are Organic Molecules 1. Organic molecules that are Carbon based (at least 1 Carbon molecule and often
Proteins Biomolecules Nucleic Acids The Building Blocks of Life Carbohydrates Lipids Biomolecules are Organic Molecules 1. Organic molecules that are Carbon based (at least 1 Carbon molecule and often
UGRC 145: FOOD AND NUTRITION IN EVERYDAY LIFE
 UGRC 145: FOOD AND NUTRITION IN EVERYDAY LIFE Session 2 MACRONUTRIENTS Lecturer: PROF. MATILDA STEINER-ASIEDU, SBS, CBAS; University of Ghana, Email: tillysteiner@gmail.com College of Education School
UGRC 145: FOOD AND NUTRITION IN EVERYDAY LIFE Session 2 MACRONUTRIENTS Lecturer: PROF. MATILDA STEINER-ASIEDU, SBS, CBAS; University of Ghana, Email: tillysteiner@gmail.com College of Education School
Importance of Nutrition
 The EAT WELL Plate Canada s food guide Food pyramid Importance of Nutrition Energy for body metabolism (nerve impulses, contraction of muscles, repair and replacement of cells Raw materials for building
The EAT WELL Plate Canada s food guide Food pyramid Importance of Nutrition Energy for body metabolism (nerve impulses, contraction of muscles, repair and replacement of cells Raw materials for building
Nutrients The substances in food that promote normal growth, maintenance, and repair in your body are called nutrients.
 Lesson 1 Nutrition and Your Health What Is Nutrition? Nutrients The substances in food that promote normal growth, maintenance, and repair in your body are called nutrients. Nutrition Nutrition is the
Lesson 1 Nutrition and Your Health What Is Nutrition? Nutrients The substances in food that promote normal growth, maintenance, and repair in your body are called nutrients. Nutrition Nutrition is the
Topic 3.1 Nutrients. - Lipids are an essential part of the and are a part of cell in the body.
 Name: Topic 3.1 Nutrients Date: IB SEHS 3.1.1. List the macronutrients and micronutrients Macronutrients: - lipid (fat) - carbohydrate - protein - water (says the book) Micronutrients: - vitamins - minerals
Name: Topic 3.1 Nutrients Date: IB SEHS 3.1.1. List the macronutrients and micronutrients Macronutrients: - lipid (fat) - carbohydrate - protein - water (says the book) Micronutrients: - vitamins - minerals
Name Date Class. 2. Is the following sentence true or false? Food is required for the body to. maintain homeostasis, keeping a steady internal state.
 CHAPTER 11 FOOD AND DIGESTION SECTION 11 1 Food and Energy (pages 370-380) This section tells about the six nutrients needed by the body. It also describes the Food Guide Pyramid and how to read labels
CHAPTER 11 FOOD AND DIGESTION SECTION 11 1 Food and Energy (pages 370-380) This section tells about the six nutrients needed by the body. It also describes the Food Guide Pyramid and how to read labels
Chapter 2 Part 3: Organic and Inorganic Compounds
 Chapter 2 Part 3: Organic and Inorganic Compounds Objectives: 1) List the major groups of inorganic chemicals common in cells. 2) Describe the functions of various types of inorganic chemicals in cells.
Chapter 2 Part 3: Organic and Inorganic Compounds Objectives: 1) List the major groups of inorganic chemicals common in cells. 2) Describe the functions of various types of inorganic chemicals in cells.
Nutrition is the study of the nutrients in food and how they nourish the body.
 Chapter 2 Nutrients Copyright 2011 by the National Restaurant Association Educational Foundation (NRAEF) and published by Pearson Education, Inc. All rights reserved. People need certain nutrients on a
Chapter 2 Nutrients Copyright 2011 by the National Restaurant Association Educational Foundation (NRAEF) and published by Pearson Education, Inc. All rights reserved. People need certain nutrients on a
The six elements that make up 99.9% of all living things include
 The six elements that make up 99.9% of all living things include 1. C, K, O, N, Ca and S 2. C, P, S, H, O and N 3. C, P, K, I, O and N 4. N, O, P, H, S and T 75% 15% 1 C, K, O, N, Ca and S C, P, S, H,
The six elements that make up 99.9% of all living things include 1. C, K, O, N, Ca and S 2. C, P, S, H, O and N 3. C, P, K, I, O and N 4. N, O, P, H, S and T 75% 15% 1 C, K, O, N, Ca and S C, P, S, H,
Chapter 6, Part Read Activity 6A - Choosing a Meal and orally attempt the procedure and discussion on page 99.
 Science 9 Unit 1 Worksheet Chapter 6, Part 1. 1. Read Activity 6A - Choosing a Meal and orally attempt the procedure and discussion on page 99. 2. Your body is made up of,,,, and many other materials.
Science 9 Unit 1 Worksheet Chapter 6, Part 1. 1. Read Activity 6A - Choosing a Meal and orally attempt the procedure and discussion on page 99. 2. Your body is made up of,,,, and many other materials.
NUTRITION. Professor Andrea Garrison Biology 11 Illustrations 2010 Pearson Education, Inc.
 NUTRITION Professor Andrea Garrison Biology 11 Illustrations 2010 Pearson Education, Inc. Proper Diet Carbohydrates Proteins Lipids (fats) Water Vitamins Fiber Inorganic salts Nutrition 2 Carbohydrates
NUTRITION Professor Andrea Garrison Biology 11 Illustrations 2010 Pearson Education, Inc. Proper Diet Carbohydrates Proteins Lipids (fats) Water Vitamins Fiber Inorganic salts Nutrition 2 Carbohydrates
What is food made of?
 What is food made of? Food: Nutrients and Food Any substance that is ingested (eaten) and sustains life Meat, fish, nuts, fruits, vegetables, grain products, etc. Nutrients: Food is broken down into substances
What is food made of? Food: Nutrients and Food Any substance that is ingested (eaten) and sustains life Meat, fish, nuts, fruits, vegetables, grain products, etc. Nutrients: Food is broken down into substances
CHEMISTRY OF LIFE 30 JANUARY 2013
 CHEMISTRY OF LIFE 30 JANUARY 2013 Lesson Description In this lesson, we will: Investigate the structure and function of molecules that are essential for life. Key Concepts Terminology A molecule is any
CHEMISTRY OF LIFE 30 JANUARY 2013 Lesson Description In this lesson, we will: Investigate the structure and function of molecules that are essential for life. Key Concepts Terminology A molecule is any
Digestion and Human Health
 Digestion and Human Health The Molecules of Living Systems There are three main fluid components in your body Cytoplasm in your cells Fluid between your cells Fluid in your blood The also contain many
Digestion and Human Health The Molecules of Living Systems There are three main fluid components in your body Cytoplasm in your cells Fluid between your cells Fluid in your blood The also contain many
Function, maintenance and protection
 2. Food and nutrients 2.3 The role of nutrients VITAMINS 2.3.3 Function, maintenance and protection Vitamins and minerals do not provide energy but they play a basic role in how the body functions. We
2. Food and nutrients 2.3 The role of nutrients VITAMINS 2.3.3 Function, maintenance and protection Vitamins and minerals do not provide energy but they play a basic role in how the body functions. We
I. ROLE OF CARBON IN ORGANISMS: Organic compounds = compounds that contain carbon Ex: Carbohydrates, lipids, proteins
 I. ROLE OF CARBON IN ORGANISMS: Organic compounds = compounds that contain carbon Ex: Carbohydrates, lipids, proteins Inorganic compounds = compounds that DO NOT contain carbon Ex: Vitamins, minerals,
I. ROLE OF CARBON IN ORGANISMS: Organic compounds = compounds that contain carbon Ex: Carbohydrates, lipids, proteins Inorganic compounds = compounds that DO NOT contain carbon Ex: Vitamins, minerals,
Biomolecules. Unit 3
 Biomolecules Unit 3 Atoms Elements Compounds Periodic Table What are biomolecules? Monomers vs Polymers Carbohydrates Lipids Proteins Nucleic Acids Minerals Vitamins Enzymes Triglycerides Chemical Reactions
Biomolecules Unit 3 Atoms Elements Compounds Periodic Table What are biomolecules? Monomers vs Polymers Carbohydrates Lipids Proteins Nucleic Acids Minerals Vitamins Enzymes Triglycerides Chemical Reactions
CHEMISTRY OF LIFE 05 FEBRUARY 2014
 CHEMISTRY OF LIFE 05 FEBRUARY 2014 In this lesson we will: Lesson Description Discuss inorganic compounds and their importance Discuss organic compounds and their biological importance. Summary Inorganic
CHEMISTRY OF LIFE 05 FEBRUARY 2014 In this lesson we will: Lesson Description Discuss inorganic compounds and their importance Discuss organic compounds and their biological importance. Summary Inorganic
Nutrients and Digestion
 Nutrients and Digestion Nutrition what is needed to be taken in to keep the body healthy Essential Nutrients Carbohydrates Fats Proteins Minerals Vitamins Water Carbohydrates Types of sugars combined in
Nutrients and Digestion Nutrition what is needed to be taken in to keep the body healthy Essential Nutrients Carbohydrates Fats Proteins Minerals Vitamins Water Carbohydrates Types of sugars combined in
Assignment #1: Biological Molecules & the Chemistry of Life
 Assignment #1: Biological Molecules & the Chemistry of Life A. Important Inorganic Molecules Water 1. Explain why water is considered a polar molecule. The partial negative charge of the oxygen and the
Assignment #1: Biological Molecules & the Chemistry of Life A. Important Inorganic Molecules Water 1. Explain why water is considered a polar molecule. The partial negative charge of the oxygen and the
Six Nutrients. Nutrients: substances in food that your body needs to stay healthy. Carbohydrates Protein Fat Minerals Vitamins Water
 Nutrients Six Nutrients Nutrients: substances in food that your body needs to stay healthy Carbohydrates Protein Fat Minerals Vitamins Water Water Function: most essential nutrient Helps digest and absorb
Nutrients Six Nutrients Nutrients: substances in food that your body needs to stay healthy Carbohydrates Protein Fat Minerals Vitamins Water Water Function: most essential nutrient Helps digest and absorb
Carbohydrates, Lipids, Proteins, and Nucleic Acids
 Carbohydrates, Lipids, Proteins, and Nucleic Acids Is it made of carbohydrates? Organic compounds composed of carbon, hydrogen, and oxygen in a 1:2:1 ratio. A carbohydrate with 6 carbon atoms would have
Carbohydrates, Lipids, Proteins, and Nucleic Acids Is it made of carbohydrates? Organic compounds composed of carbon, hydrogen, and oxygen in a 1:2:1 ratio. A carbohydrate with 6 carbon atoms would have
½ cup of CHEX MIX contains 13 g of carbs = 4% daily value. How much more can you have the rest of the day??? _4_ = X X= 325 g
 BIOCHEMISTRY ½ cup of CHEX MIX contains 13 g of carbs = 4% daily value. How much more can you have the rest of the day??? _4_ = 13 100 X X= 325 g These spinach imposters contain less than 2 percent of
BIOCHEMISTRY ½ cup of CHEX MIX contains 13 g of carbs = 4% daily value. How much more can you have the rest of the day??? _4_ = 13 100 X X= 325 g These spinach imposters contain less than 2 percent of
INTERMEDIATE 1 1 Food and Diet. These elements are present in compounds - not as free elements.
 INTERMEDIATE 1 1 Food and Diet FOOD AND DIET The main elements present in the human body are: Hydrogen Oxygen Nitrogen Carbon These elements are present in compounds - not as free elements. Unlike plants,
INTERMEDIATE 1 1 Food and Diet FOOD AND DIET The main elements present in the human body are: Hydrogen Oxygen Nitrogen Carbon These elements are present in compounds - not as free elements. Unlike plants,
Name Date Class. This section tells about the six nutrients needed by the body and the usefulness of the Fook Guide Pyramid and food labels.
 Food and Digestion Name Date Class Food and Energy This section tells about the six nutrients needed by the body and the usefulness of the Fook Guide Pyramid and food labels. Use Target Reading Skills
Food and Digestion Name Date Class Food and Energy This section tells about the six nutrients needed by the body and the usefulness of the Fook Guide Pyramid and food labels. Use Target Reading Skills
Environmental Literacy Project Michigan State University. Lesson 2.3: Materials Plants Are Made Of
 Environmental Literacy Project Michigan State University Lesson 2.3: Materials Plants Are Made Of Benchmark Scale Power of Ten Large Scale: Farm field Decimal Style Large scale Larger 10 5 10 4 10 3 Larger
Environmental Literacy Project Michigan State University Lesson 2.3: Materials Plants Are Made Of Benchmark Scale Power of Ten Large Scale: Farm field Decimal Style Large scale Larger 10 5 10 4 10 3 Larger
Cell Organization, Nutrition, Digestion, and Excretion
 Cell Organization, Nutrition, Digestion, and Excretion Cell Organization: 1. Put these terms in order from least complex to most complex: Tissue, cell, organ, organelle, organism, organ system 1. Organelle
Cell Organization, Nutrition, Digestion, and Excretion Cell Organization: 1. Put these terms in order from least complex to most complex: Tissue, cell, organ, organelle, organism, organ system 1. Organelle
1.3.1 Function of Food. Why do we need food?
 1.3.1 Function of Food Why do we need food? Need to know The Function of Food Three reasons for requiring food 2 Food is needed for: 1.Energy 2.Growth of new cells and Repair of existing cells, tissues,
1.3.1 Function of Food Why do we need food? Need to know The Function of Food Three reasons for requiring food 2 Food is needed for: 1.Energy 2.Growth of new cells and Repair of existing cells, tissues,
Properties of Water. 1. The graph shows the relationship between the rate of enzyme action and ph for three enzymes: pepsin, urease, and trypsin.
 Name: ate: 1. The graph shows the relationship between the rate of enzyme action and ph for three enzymes: pepsin, urease, and trypsin. 1. Which of these enzymes function in the most similar ph range?.
Name: ate: 1. The graph shows the relationship between the rate of enzyme action and ph for three enzymes: pepsin, urease, and trypsin. 1. Which of these enzymes function in the most similar ph range?.
Classes of Nutrients A Diet
 Ch. 7 Notes Section 1: What is Nutrition? is the science or study of food and the ways the body uses food. are substances in food that provide energy or help form body tissues and are necessary for life
Ch. 7 Notes Section 1: What is Nutrition? is the science or study of food and the ways the body uses food. are substances in food that provide energy or help form body tissues and are necessary for life
Proteins. Biomolecules. Nucleic Acids. The Building Blocks of Life
 Proteins Biomolecules Nucleic Acids The Building Blocks of Life Carbohydrates Lipids Biomolecules are 1. Organic molecules that are (at least 1 Carbon molecule and often chains of Carbon) They all contain.
Proteins Biomolecules Nucleic Acids The Building Blocks of Life Carbohydrates Lipids Biomolecules are 1. Organic molecules that are (at least 1 Carbon molecule and often chains of Carbon) They all contain.
30.1 Organization of the Human Body
 30.1 Organization of the Human Body Lesson Objectives Describe how the human body is organized. Explain homeostasis. Lesson Summary Organization of the Body The levels of organization in a multicellular
30.1 Organization of the Human Body Lesson Objectives Describe how the human body is organized. Explain homeostasis. Lesson Summary Organization of the Body The levels of organization in a multicellular
BIOLOGICAL MOLECULES REVIEW-UNIT 1 1. The factor being tested in an experiment is the A. data. B. variable. C. conclusion. D. observation. 2.
 BIOLOGICAL MOLECULES REVIEW-UNIT 1 1. The factor being tested in an experiment is the A. data. B. variable. C. conclusion. D. observation. 2. A possible explanation for an event that occurs in nature is
BIOLOGICAL MOLECULES REVIEW-UNIT 1 1. The factor being tested in an experiment is the A. data. B. variable. C. conclusion. D. observation. 2. A possible explanation for an event that occurs in nature is
What are the most common elements in living organisms? What is the difference between monomers, dimers and polymers?
 What do each of these terms mean? Atom Molecule Element Compound Organic Inorganic What are the most common elements in living organisms? What are the roles of magnesium, iron, phosphate and calcium in
What do each of these terms mean? Atom Molecule Element Compound Organic Inorganic What are the most common elements in living organisms? What are the roles of magnesium, iron, phosphate and calcium in
Essential Components of Food
 Essential Components of Food The elements of life living things are mostly (98%) made of 6 elements: C carbon H hydrogen O oxygen P phosphorus N nitrogen S sulphur -each element makes a specific number
Essential Components of Food The elements of life living things are mostly (98%) made of 6 elements: C carbon H hydrogen O oxygen P phosphorus N nitrogen S sulphur -each element makes a specific number
Biomolecules. The chemistry of life
 Biomolecules The chemistry of life Knowing Word Parts can help you remember big words in Biochem Mono one, single Di two, double Poly many, much Hydro water (think: hydrate) Bio related to life (think:
Biomolecules The chemistry of life Knowing Word Parts can help you remember big words in Biochem Mono one, single Di two, double Poly many, much Hydro water (think: hydrate) Bio related to life (think:
Chapter 15 Food and Digestion
 Chapter 15 Food and Digestion 15.1A Food and Energy Functions of Nutrients 1. 2. 3. 4. Calories = amt. of energy in food RDA depends on age, gender, size and activity level Types of Nutrients (includes
Chapter 15 Food and Digestion 15.1A Food and Energy Functions of Nutrients 1. 2. 3. 4. Calories = amt. of energy in food RDA depends on age, gender, size and activity level Types of Nutrients (includes
Human Digestion. Junior Science
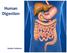 Human Digestion Junior Science True or False The word stomach comes from the Greek meaning Throat. Butterflies in the stomach have nothing to do with the stomach. It is possible to live without your stomach.
Human Digestion Junior Science True or False The word stomach comes from the Greek meaning Throat. Butterflies in the stomach have nothing to do with the stomach. It is possible to live without your stomach.
Thursday, September 5. Why are carbohydrates important for us to consume?
 Bell Work: Thursday, September 5 Carbon Video Why are carbohydrates important for us to consume? 1 O 2 CARBON What is it...and WY is it important? Carbon the building block of life, basis for most molecules
Bell Work: Thursday, September 5 Carbon Video Why are carbohydrates important for us to consume? 1 O 2 CARBON What is it...and WY is it important? Carbon the building block of life, basis for most molecules
Chapter 3: Biochemistry Adapted from PPT by S. Edwards. By PresenterMedia.com
 Chapter 3: Biochemistry Adapted from PPT by S. Edwards By PresenterMedia.com CARBON COMPOUNDS CHAPTER 3 SECTION 1 By PresenterMedia.com Compounds LOOK NO Carbon!!! ORGANIC COMPOUNDS Compounds that contain
Chapter 3: Biochemistry Adapted from PPT by S. Edwards By PresenterMedia.com CARBON COMPOUNDS CHAPTER 3 SECTION 1 By PresenterMedia.com Compounds LOOK NO Carbon!!! ORGANIC COMPOUNDS Compounds that contain
Biochemistry. Definition-
 Biochemistry Notes Biochemistry Definition- the scientific study of the chemical composition of living matter AND of the chemical processes that go on in living organisms. Biochemistry Facts 1. The human
Biochemistry Notes Biochemistry Definition- the scientific study of the chemical composition of living matter AND of the chemical processes that go on in living organisms. Biochemistry Facts 1. The human
Biochemistry: Macromolecules
 1 Biology: Macromolecules 2 Carbohydrates Carbohydrate organic compound containing carbon, hydrogen, & oxygen in a 1:2:1 ratio Meaning: hydrated carbon ratio of h:0 is 2:1 (same as in water) Source: plants
1 Biology: Macromolecules 2 Carbohydrates Carbohydrate organic compound containing carbon, hydrogen, & oxygen in a 1:2:1 ratio Meaning: hydrated carbon ratio of h:0 is 2:1 (same as in water) Source: plants
Digestive System. Science 7 HMS 2016
 Digestive System Science 7 HMS 2016 ??? What is the role of the digestive system? 1. Breaks down food into molecules the body can use 2. Molecules are absorbed into the blood stream to be carried to the
Digestive System Science 7 HMS 2016 ??? What is the role of the digestive system? 1. Breaks down food into molecules the body can use 2. Molecules are absorbed into the blood stream to be carried to the
NUTRITION. Understanding Nutrients and Their Importance
 NUTRITION Understanding Nutrients and Their Importance What are the essential nutrients for animal production? Good nutrition can increase feed efficiency and the rate of gain for animals. Animals must
NUTRITION Understanding Nutrients and Their Importance What are the essential nutrients for animal production? Good nutrition can increase feed efficiency and the rate of gain for animals. Animals must
FOOD. Why do we need food? What's in our food? There are 3 trace elements, Iron (Fe), Copper (Cu) and Zinc (Zn).
 Why do we need food? FOOD 1. As a source of energy keeps our cells and us alive. 2. To make chemicals for our metabolic reactions. 3. As raw materials for growth and repair of our cells and body. What's
Why do we need food? FOOD 1. As a source of energy keeps our cells and us alive. 2. To make chemicals for our metabolic reactions. 3. As raw materials for growth and repair of our cells and body. What's
Section 4: Exercise Physiology. Diet and nutrition and their effect on physical activity and performance
 Section 4: Exercise Physiology Diet and nutrition and their effect on physical activity and performance Learning Objectives 1. Identify the seven classes of food as: carbohydrates, fats, proteins, vitamins,
Section 4: Exercise Physiology Diet and nutrition and their effect on physical activity and performance Learning Objectives 1. Identify the seven classes of food as: carbohydrates, fats, proteins, vitamins,
Copy into Note Packet and Return to Teacher Section 3 Chemistry of Cells
 Copy into Note Packet and Return to Teacher Section 3 Chemistry of Cells Objectives Summarize the characteristics of organic compounds. Compare the structures and function of different types of biomolecules.
Copy into Note Packet and Return to Teacher Section 3 Chemistry of Cells Objectives Summarize the characteristics of organic compounds. Compare the structures and function of different types of biomolecules.
BIOLOGY 111. CHAPTER 2: The Chemistry of Life Biological Molecules
 BIOLOGY 111 CHAPTER 2: The Chemistry of Life Biological Molecules The Chemistry of Life : Learning Outcomes 2.4) Describe the significance of carbon in forming the basis of the four classes of biological
BIOLOGY 111 CHAPTER 2: The Chemistry of Life Biological Molecules The Chemistry of Life : Learning Outcomes 2.4) Describe the significance of carbon in forming the basis of the four classes of biological
Biochemistry Name: Practice Questions
 Name: Practice Questions 1. Carbohydrate molecules A and B come in contact with the cell membrane of the same cell. Molecule A passes through the membrane readily, but molecule B does not. It is most likely
Name: Practice Questions 1. Carbohydrate molecules A and B come in contact with the cell membrane of the same cell. Molecule A passes through the membrane readily, but molecule B does not. It is most likely
1.1.1 Protein. 1 Quiz: Protein. 1. The main reason why the body needs protein is for growth, repair and maintenance.
 1 Quiz: Protein 1.1.1 Protein 1. The main reason why the body needs protein is for growth, repair and maintenance. 2. An example of a protein alternative is mycoprotein, e.g. Quorn. 3. An example of a
1 Quiz: Protein 1.1.1 Protein 1. The main reason why the body needs protein is for growth, repair and maintenance. 2. An example of a protein alternative is mycoprotein, e.g. Quorn. 3. An example of a
6 Nutrients Essential for Life
 6 Nutrients Essential for Life Mind Moo-Ver SWBAT identify the 6 essential nutrients for life QOD: What does ph measure Give an example of an acidic substance, a basic substance and a neutral substance
6 Nutrients Essential for Life Mind Moo-Ver SWBAT identify the 6 essential nutrients for life QOD: What does ph measure Give an example of an acidic substance, a basic substance and a neutral substance
The chemistry of life
 The chemistry of life All living organisms are comprised of organic molecules. Organic molecules contain CARBON and HYDROGEN which is not true of inorganic molecules. Carbon is central to life on Earth
The chemistry of life All living organisms are comprised of organic molecules. Organic molecules contain CARBON and HYDROGEN which is not true of inorganic molecules. Carbon is central to life on Earth
AGENDA for 12/03/13 AGENDA: HOMEWORK: Due Thurs, OBJECTIVES:
 AGENDA for 12/03/13 AGENDA: 1. Go over 2.1 Quiz 2. 2.2.2: Food Labels Analyzing Food Labels Assessing Nutritional Daily Requirements OBJECTIVES: 1. Analyze food labels to determine the nutritional content
AGENDA for 12/03/13 AGENDA: 1. Go over 2.1 Quiz 2. 2.2.2: Food Labels Analyzing Food Labels Assessing Nutritional Daily Requirements OBJECTIVES: 1. Analyze food labels to determine the nutritional content
Organic Compounds. B-3.5 Students will be able to summarize the functions of proteins, carbohydrates, and fats in the human body.
 Organic Compounds B-3.4 tudents will be able to summarize how the structures of organic molecules (including proteins, carbohydrates, and fats) are related to their relative caloric values. B-3.5 tudents
Organic Compounds B-3.4 tudents will be able to summarize how the structures of organic molecules (including proteins, carbohydrates, and fats) are related to their relative caloric values. B-3.5 tudents
NUTRITION: THE STUDY OF HOW THE BODY UTILIZES THE FOOD WE EAT
 NUTRITION: THE STUDY OF HOW THE BODY UTILIZES THE FOOD WE EAT NUTRIENT: a chemical found in food that is needed for life and growth, & the building blocks to make your body function correctly! WHAT DO
NUTRITION: THE STUDY OF HOW THE BODY UTILIZES THE FOOD WE EAT NUTRIENT: a chemical found in food that is needed for life and growth, & the building blocks to make your body function correctly! WHAT DO
Chapter 15 Food and Digestion
 Chapter 15 Food and Digestion Activity: Use Qualitative Observations (5 senses) to describe: What happens when you see candy? How does it smell? How do you chomp it into smaller pieces or swallow candy
Chapter 15 Food and Digestion Activity: Use Qualitative Observations (5 senses) to describe: What happens when you see candy? How does it smell? How do you chomp it into smaller pieces or swallow candy
BCH 445 Biochemistry of nutrition Dr. Mohamed Saad Daoud
 BCH 445 Biochemistry of nutrition Dr. Mohamed Saad Daoud 1 Carbohydrates Carbohydrates: Compounds composed of carbon, oxygen, and hydrogen arranged as monosaccharides or multiples of monosaccharides. Most,
BCH 445 Biochemistry of nutrition Dr. Mohamed Saad Daoud 1 Carbohydrates Carbohydrates: Compounds composed of carbon, oxygen, and hydrogen arranged as monosaccharides or multiples of monosaccharides. Most,
Objective 4- Digestion
 Objective 4- Digestion 1. Describe why cells require nutrients Supply energy for metabolism (cell activities), matter for synthesis of new materials, cell reproduction, secretion and to regulate cell processes
Objective 4- Digestion 1. Describe why cells require nutrients Supply energy for metabolism (cell activities), matter for synthesis of new materials, cell reproduction, secretion and to regulate cell processes
Human Nutrition. How our diet determines Health & Wellness
 Human Nutrition How our diet determines Health & Wellness Nutrition The study of food How it nourishes our body How it influences health A mix of Biological and Social Science Relatively new Pragmatic
Human Nutrition How our diet determines Health & Wellness Nutrition The study of food How it nourishes our body How it influences health A mix of Biological and Social Science Relatively new Pragmatic
Learning Target: Describe characteristics and functions of carbohydrates, lipids, and proteins. Compare and contrast the classes of organic
 Learning Target: Describe characteristics and functions of carbohydrates, lipids, and proteins. Compare and contrast the classes of organic compounds. What are inorganic molecules? Molecules that CANNOT
Learning Target: Describe characteristics and functions of carbohydrates, lipids, and proteins. Compare and contrast the classes of organic compounds. What are inorganic molecules? Molecules that CANNOT
Carbon. Has four valence electrons Can bond with many elements. Can bond to other carbon atoms. Hydrogen, Oxygen, Phosphorus, Sulfur, and Nitrogen
 Organic Compounds Carbon Has four valence electrons Can bond with many elements Hydrogen, Oxygen, Phosphorus, Sulfur, and Nitrogen Can bond to other carbon atoms Gives carbon the ability to form chains
Organic Compounds Carbon Has four valence electrons Can bond with many elements Hydrogen, Oxygen, Phosphorus, Sulfur, and Nitrogen Can bond to other carbon atoms Gives carbon the ability to form chains
Chemical Formulas. Chemical Formula CH 3 COCHCHOCHClCHNH Lewis Dot Structure
 Biochemistry . Chemical Formulas A chemical formula represents the chemical makeup of a compound. It shows the numbers and kinds of atoms present in a compound. It is a kind of shorthand that scientists
Biochemistry . Chemical Formulas A chemical formula represents the chemical makeup of a compound. It shows the numbers and kinds of atoms present in a compound. It is a kind of shorthand that scientists
Do Now: Sort the following into the order of life from smallest to largest:
 Do Now: Sort the following into the order of life from smallest to largest: organ, molecule, atom, organelle, cell, organ system, tissue, organism Correct Order: atom, molecule, organelle, cell, tissue,
Do Now: Sort the following into the order of life from smallest to largest: organ, molecule, atom, organelle, cell, organ system, tissue, organism Correct Order: atom, molecule, organelle, cell, tissue,
Ms. Golub & Ms. Sahar Date: Unit 2- Test #1
 Name Ms. Golub & Ms. Sahar Date: Unit 2- Test #1 1. The interaction between guard cells and a leaf opening would not be involved in A) diffusion of carbon dioxide B) maintaining homeostasis C) heterotrophic
Name Ms. Golub & Ms. Sahar Date: Unit 2- Test #1 1. The interaction between guard cells and a leaf opening would not be involved in A) diffusion of carbon dioxide B) maintaining homeostasis C) heterotrophic
Biology Chapter 2 Review
 Biology Chapter 2 Review Vocabulary: Define the following words on a separate piece of paper. Element Compound Ion Ionic Bond Covalent Bond Molecule Hydrogen Bon Cohesion Adhesion Solution Solute Solvent
Biology Chapter 2 Review Vocabulary: Define the following words on a separate piece of paper. Element Compound Ion Ionic Bond Covalent Bond Molecule Hydrogen Bon Cohesion Adhesion Solution Solute Solvent
The Digestive and Excretory Systems
 BC Science 8 CH02 11/5/06 11:39 AM Page 26 The Digestive and Excretory Systems Textbook pages 64 81 Before You Read Section 2.2 Summary Many structures and organs work together to digest food. What parts
BC Science 8 CH02 11/5/06 11:39 AM Page 26 The Digestive and Excretory Systems Textbook pages 64 81 Before You Read Section 2.2 Summary Many structures and organs work together to digest food. What parts
Chapter 2 pt 2. Atoms, Molecules, and Life. Gregory Ahearn. John Crocker. Including the lecture Materials of
 Chapter 2 pt 2 Atoms, Molecules, and Life Including the lecture Materials of Gregory Ahearn University of North Florida with amendments and additions by John Crocker Copyright 2009 Pearson Education, Inc..
Chapter 2 pt 2 Atoms, Molecules, and Life Including the lecture Materials of Gregory Ahearn University of North Florida with amendments and additions by John Crocker Copyright 2009 Pearson Education, Inc..
BIOMOLECULES. Ms. Bosse Fall 2015
 BIOMOLECULES Ms. Bosse Fall 2015 Biology Biology is the study of the living world. Bio = life Major Molecules of Life Macromolecules giant molecules found in living cells; made from thousands of smaller
BIOMOLECULES Ms. Bosse Fall 2015 Biology Biology is the study of the living world. Bio = life Major Molecules of Life Macromolecules giant molecules found in living cells; made from thousands of smaller
I. ROLE OF CARBON IN ORGANISMS:
 Name: Period: Date: I. ROLE OF CARBON IN ORGANISMS: = compounds that contain carbon Ex: Carbohydrates, lipids, proteins = compounds that DO NOT contain carbon Ex: Vitamins, minerals, water Carbon forms
Name: Period: Date: I. ROLE OF CARBON IN ORGANISMS: = compounds that contain carbon Ex: Carbohydrates, lipids, proteins = compounds that DO NOT contain carbon Ex: Vitamins, minerals, water Carbon forms
Biology 12. Biochemistry. Water - a polar molecule Water (H 2 O) is held together by covalent bonds.
 Biology 12 Biochemistry Water - a polar molecule Water (H 2 O) is held together by covalent bonds. Electrons in these bonds spend more time circulating around the larger Oxygen atom than the smaller Hydrogen
Biology 12 Biochemistry Water - a polar molecule Water (H 2 O) is held together by covalent bonds. Electrons in these bonds spend more time circulating around the larger Oxygen atom than the smaller Hydrogen
Biomolecules. 1) Carbohydrate Facts. Types of Biomolecules. Carbohydrate Facts. What are Biomolecules? 12/25/13. Two Types of Carbohydrates
 Biomolecules What are Biomolecules? Organic compounds made by living things Also called biochemicals Some are very large There are thousands of different biomolecules, but are separated into 4 categories
Biomolecules What are Biomolecules? Organic compounds made by living things Also called biochemicals Some are very large There are thousands of different biomolecules, but are separated into 4 categories
Nutrition. Chapter 45. Reada Almashagba
 Nutrition Chapter 45 1 Nutrition: - Nutrient are organic substances found in food and are required for body function - No one food provide all essential nutrient Major function of nutrition: providing
Nutrition Chapter 45 1 Nutrition: - Nutrient are organic substances found in food and are required for body function - No one food provide all essential nutrient Major function of nutrition: providing
Dehydration Synthesis and Hydrolysis Reactions. ne_content/animations/reaction_types.ht ml
 Glucose Molecule Macromolecules Carbohydrates, proteins, and nucleic acids are polymers Polymers long molecules made from building blocks linked by covalent bonds Monomers the building blocks to polymers
Glucose Molecule Macromolecules Carbohydrates, proteins, and nucleic acids are polymers Polymers long molecules made from building blocks linked by covalent bonds Monomers the building blocks to polymers
Railways (Group-D & ALP) Science Questions with solution
 Railways (Group-D & ALP) Science Questions with solution RRB Science MCQs Quiz 2 Directions: Study the following questions carefully and choose the right answer: 1. Which of the following non-alcoholic
Railways (Group-D & ALP) Science Questions with solution RRB Science MCQs Quiz 2 Directions: Study the following questions carefully and choose the right answer: 1. Which of the following non-alcoholic
When people don t eat enough complex carbohydrates they don t have enough energy and feel tired and less alert. They also may not get enough fiber.
 Carbohydrates Carbohydrates are compounds that come from plants and contain carbon, hydrogen, and oxygen. These nutrients supply energy, which all living things need. Carbohydrates are the body s most
Carbohydrates Carbohydrates are compounds that come from plants and contain carbon, hydrogen, and oxygen. These nutrients supply energy, which all living things need. Carbohydrates are the body s most
LIP I I P D I S & PROTEINS
 LIPIDS & PROTEINS I. LIPIDS: Foods: butter, oil, Crisco, lard Commonly called fats & oils Contain more C-H bonds and less O atoms than carbohydrates. Ex: C 57 H 110 O 6 Nonpolar; therefore repel water
LIPIDS & PROTEINS I. LIPIDS: Foods: butter, oil, Crisco, lard Commonly called fats & oils Contain more C-H bonds and less O atoms than carbohydrates. Ex: C 57 H 110 O 6 Nonpolar; therefore repel water
UNIT 3 CHEMISTRY OF LIFE NOTES Chapter 6 pg
 UNIT 3 CHEMISTRY OF LIFE NOTES Chapter 6 pg. 146-173 Name Date Class Warm-up: List the percentages of each: Total Fats Saturated Fats Carbohydrates Protein What biomolecule would cholesterol be classified
UNIT 3 CHEMISTRY OF LIFE NOTES Chapter 6 pg. 146-173 Name Date Class Warm-up: List the percentages of each: Total Fats Saturated Fats Carbohydrates Protein What biomolecule would cholesterol be classified
The building blocks for this molecule are A) amino acids B) simple sugars C) fats D) molecular bases
 1. Base your answer to the following question on the diagram below and on your knowledge of biology. The diagram represents a portion of a starch molecule. The building blocks for this molecule are A)
1. Base your answer to the following question on the diagram below and on your knowledge of biology. The diagram represents a portion of a starch molecule. The building blocks for this molecule are A)
Chapter Three (Biochemistry)
 Chapter Three (Biochemistry) 1 SECTION ONE: CARBON COMPOUNDS CARBON BONDING All compounds can be classified in two broad categories: organic compounds and inorganic compounds. Organic compounds are made
Chapter Three (Biochemistry) 1 SECTION ONE: CARBON COMPOUNDS CARBON BONDING All compounds can be classified in two broad categories: organic compounds and inorganic compounds. Organic compounds are made
Nutrition, part 2. Because 1 part isn t enough!
 Nutrition, part 2 Because 1 part isn t enough! 4. Calories and Caloric Intake Calories per gram of our Macro and Micro nutrients Macro Carbohydrates: 4 cal/g Fats: 9 cal/g Proteins: 4 cal/g Micro Vitamins:
Nutrition, part 2 Because 1 part isn t enough! 4. Calories and Caloric Intake Calories per gram of our Macro and Micro nutrients Macro Carbohydrates: 4 cal/g Fats: 9 cal/g Proteins: 4 cal/g Micro Vitamins:
Mid Term Review. 1. step 1, only 3. both step 1 and step 2 2. step 2, only 4. neither step 1 nor step 2
 Name Mid Term Review 1. Diagrams, tables, and graphs are used by scientists mainly to 1. design a research plan for an experiment 3. organize data 2. test a hypothesis 4. predict the independent variable
Name Mid Term Review 1. Diagrams, tables, and graphs are used by scientists mainly to 1. design a research plan for an experiment 3. organize data 2. test a hypothesis 4. predict the independent variable
General, Organic & Biological Chemistry, 5e (Timberlake) Chapter 2 Chemistry and Measurements. 2.1 Multiple-Choice Questions
 General Organic and Biological Chemistry Structures of Life 5th Edition Timberlake TEST BANK Full download at: https://testbankreal.com/wp-admin/post.php?post=1729&action=edit General, Organic & Biological
General Organic and Biological Chemistry Structures of Life 5th Edition Timberlake TEST BANK Full download at: https://testbankreal.com/wp-admin/post.php?post=1729&action=edit General, Organic & Biological
5. Groups A and B in the table below contain molecular formulas of compounds.
 1. Which group consists entirely of organic molecules? A) protein, oxygen, fat B) protein, starch, fat C) water, carbon dioxide, oxygen D) water, starch, protein 2. Which statement describes starches,
1. Which group consists entirely of organic molecules? A) protein, oxygen, fat B) protein, starch, fat C) water, carbon dioxide, oxygen D) water, starch, protein 2. Which statement describes starches,
CHEMISTRY OF LIFE 13 MARCH 2013
 CHEMISTRY OF LIFE 13 MARCH 2013 Lesson Description In this lesson, we revise: How molecules are classified The importance of water How to test samples for glucose and starch Key Concepts Terminology A
CHEMISTRY OF LIFE 13 MARCH 2013 Lesson Description In this lesson, we revise: How molecules are classified The importance of water How to test samples for glucose and starch Key Concepts Terminology A
(*) (*) Ingestion, digestion, absorption, and elimination. Uptake of nutrients by body cells (intestine)
 Human Digestive System Food is pushed along the digestive tract by peristalsis the rhythmic waves of contraction of smooth muscles in the wall of the canal Accessory glands. Main stages of food processing
Human Digestive System Food is pushed along the digestive tract by peristalsis the rhythmic waves of contraction of smooth muscles in the wall of the canal Accessory glands. Main stages of food processing
Organic Molecules. 1. The structural formulas shown represent certain organic compounds found in living cells.
 Name: ate: 1. The structural formulas shown represent certain organic compounds found in living cells. 1. (1) () (3) Which formula represents a monosaccharide? (4) (5). 1.. 3. 5. Which formula represents
Name: ate: 1. The structural formulas shown represent certain organic compounds found in living cells. 1. (1) () (3) Which formula represents a monosaccharide? (4) (5). 1.. 3. 5. Which formula represents
Neatness 0 1 Accuracy Completeness Lab Class Procedure Total Lab Score
 New Paltz High School Science Department Name:.... Due Date:... Lab Title:.Big Ol Biomolecules..Lab #... Lab Partners: Your Lab Score will be based on the following: Neatness: All labs must be well-written
New Paltz High School Science Department Name:.... Due Date:... Lab Title:.Big Ol Biomolecules..Lab #... Lab Partners: Your Lab Score will be based on the following: Neatness: All labs must be well-written
2-3 Carbon Compounds 10/22/2013. The Chemistry of Carbon. More Carbon. Chemistry (cont) More Macromolecules. Macromolecules
 The Chemistry of Carbon 2-3 Carbon Compounds Because of carbons 4 valence electrons it can form covalent bonds with many other elements (octet rule) 2 Chemistry (cont) Plus, it can bond with itself More
The Chemistry of Carbon 2-3 Carbon Compounds Because of carbons 4 valence electrons it can form covalent bonds with many other elements (octet rule) 2 Chemistry (cont) Plus, it can bond with itself More
Organic Compounds. Biology-CP Mrs. Bradbury
 Organic Compounds Biology-CP Mrs. Bradbury Carbon Chemistry The compounds that form the cells and tissues of the body are produced from similar compounds in the foods you eat. Common to most foods and
Organic Compounds Biology-CP Mrs. Bradbury Carbon Chemistry The compounds that form the cells and tissues of the body are produced from similar compounds in the foods you eat. Common to most foods and
Carbon Compounds (2.3) (Part 1 - Carbohydrates)
 Carbon Compounds (2.3) (Part 1 - Carbohydrates) The Chemistry of Carbon (Organic Chemistry) Organic Chemistry: The study of compounds that contain bonds between carbon atoms. Carbon can bond with many
Carbon Compounds (2.3) (Part 1 - Carbohydrates) The Chemistry of Carbon (Organic Chemistry) Organic Chemistry: The study of compounds that contain bonds between carbon atoms. Carbon can bond with many
What is a Biomolecule?
 Biology Unit 03 What is a Biomolecule? Organic molecule made by living organisms Consists mostly of carbon (C), hydrogen (H), and oxygen (O) But wait What is an Organic Molecule? Organic Molecules: Contain
Biology Unit 03 What is a Biomolecule? Organic molecule made by living organisms Consists mostly of carbon (C), hydrogen (H), and oxygen (O) But wait What is an Organic Molecule? Organic Molecules: Contain
