BIO132 Chapter 27 Fluid, Electrolyte and Acid Base Balance Lecture Outline
|
|
|
- Willa Owen
- 6 years ago
- Views:
Transcription
1 BIO132 Chapter 27 Fluid, Electrolyte and Acid Base Balance Lecture Outline Fluid divisions 1. Extracellular fluid (ECF) 2. Intracellular fluid (ICF) Stabilization 1. Fluid balance 2. Electrolyte balance 3. Acid-Base balance Fluid & Electrolyte Balance Male ~60% H 2 O Female ~50% H 2 O 2/3 H 2 O ICF Electrolytes ECF: Na +, Cl -, HCO 3 - ICF: K +, Mg 2+, HPO 4 2-, proteins Rules: 1. Homeostasis: ECF 2. Plasma volume & osmotic concentration 3. Water follows salt 4. Content = gain + loss Hormones 1. Antidiuretic Hormone (ADH) Hypothalamus Release: osmotic concentration Effects: A. H 2 O conservation B. thirst 2. Aldosterone Adrenal cortex Release: A. K + Na + (ECF) B. renin ( BP volume) C. osmotic conc. filtrate Effect: Na + absorption & K + & collecting ducts Addison s Disease 3. Natriuretic Peptides ANP & BNP Release: heart stretch Effects: A. thirst B. block ADH C. block aldosterone Fluid Balance 1. Movement ECF: 20%plasma, 80% interstitial fluid A. hydrostatic pressure plasma IF B. colloid osmotic pressure IF plasma Edema 2. Exchange A. Water loss Obligatory: 2500ml/day urine, feces, insensible perspiration Sensible perspiration: 4L/hr Fever: 200ml/day/ C B. Water gain Drink: 1000ml/day Food: 1200ml/day Metabolism: 300ml/day Hyponatremia (hypotonic hydration) Cause: 1. ingestion/injection 2. blocked elimination 3. endocrine disorder Effect: ECF ICF Water intoxication Hypernatremia (dehydration) Hypovolemic shock 3. Shifts Equilibrium: osmosis ICF reserve Electrolyte Balance Importance: 1. water balance 2. cell functions 1. Sodium ECF: NaCl & NaHCO 3 GI intake vs. urine & perspiration loss Osmosis Too low: Renin-angiotensin Too high: Natriuretic peptides 2. Potassium ICF GI intake vs. urine loss Tubular secretion: A. K + in ECF = K + secretion B. ph in ECF = K + secretion C. aldosterone = Na + reabsorption and K + secretion Hypokalemia Cause: 1. inadequate intake 2. diuretics 3. excessive aldosterone 4. increased ph Effect: weakness, confusion Hyperkalemia Cause: 1. renal failure 2. diuretics 3. decreased ph Effect: arrhythmia, flaccid paralysis 3. Calcium Function: Skeleton Muscular & neural activity Amy Warenda Czura, Ph.D. 1 SCCC BIO132 Chapter 27 Handout
2 Blood clotting Cofactor Second messenger GI intake & skeleton vs. urine loss Too low: Parathyroid hormone & calcitriol Too high: Calcitonin Hypercalcemia Cause: hyper-parathyroidism cancer Effect: fatigue confusion arrhythmia calcification Hypocalcemia Cause: hypo-parathyroidism vitamin D deficiency renal failure Effect: spasms convulsions weak beats clotting osteoporosis 4. Magnesium Function: skeleton & ICF (cofactor) Excess: lethargy & coma Deficiency: convulsions 5. Phosphate Functions: Skeleton ATP Cofactor Nucleic acids 6. Chloride ECF Excess: metabolic acidosis Deficiency: metabolic alkalosis Acid Base Balance Acid: H + ph 0-7 Base OH - ph 7-14 Neutral ph 7, H + = OH - Strong acid/base Weak acid/base Acidosis ph < 7.35 Alkalosis ph > Types A. Volatile B. Fixed C. Organic 2. Control Buffers A. Protein Buffering System Amino acids Hemoglobin Buffering System Carbonic anhydrase Carbonic acid Bicarbonate B. Carbonic Acid-Bicarbonate Buffering ECF CO 2 + H 2 O H 2 CO 3 H + + HCO 3 - Requires: 1. CO 2 levels 2. respiration function 3. kidney function C. Phosphate Buffering System ICF H 2 PO 4 & NaPO 4 HPO Maintenance A. Respiratory Compensation CO 2 = ph CO 2 = ph B. Renal Compensation H + = ph HCO 3 - = ph 4. Disturbances A. buffers, lung, kidney B. cardiovascular C. CNS Respiratory acidosis CO 2 = ph Cardiac arrest, congestive heart failure Emphysema, pneumonia, pneumothorax Respiratory alkalosis CO 2 = ph Hyperventilation Metabolic acidosis H + = ph 1. Acid production Lactic acidosis hypoxia Ketoacidosis starvation, diabetes mellitus 2. Impaired excretion glomerulonephritis diuretics 3. Bicarbonate loss diarrhea Metabolic alkalosis HCO 3 - = ph Vomiting Antacids Age related changes water content renal compensation mineral reserves respiratory compensation Amy Warenda Czura, Ph.D. 2 SCCC BIO132 Chapter 27 Handout
3 FLUID, ELECTROLYTE AND ACID BASE BALANCE Extracellular Fluid (ECF) = interstitial fluid, plasma, lymph, CSF, synovial fluid, serous fluid, etc. Intracellular Fluid (ICF) = cytosol Homeostasis involves the regulation of composition and volume of both fluid divisions Stabilizing ECF and ICF involves: 1) Fluid Balance Must have equal gain (food & metabolism) and loss (urine & perspiration) of water 2) Electrolyte Balance Electrolytes = ions from dissociated compounds that will conduct an electrical charge in solution Must have equal gain (absorption in GI) and loss (urine in kidney and perspiration in skin) 3) Acid-Base Balance The production of hydrogen ions by metabolism must be matched by loss of these H + ions at the kidney (protons: H + ) and lungs (carbonic acid) Fluid and Electrolyte Balance (water and ions move together) - Average male ~ 60% H 2 O (more muscle which can be ~ 75% H 2 O) - Average female ~ 50% H 2 O (more adipose which is only ~ 10% H 2 O) - Most of the water in the body is found in the ICF (~ 2/3) - The electrolytes vary depending on the fluid division: ECF Principal cation = Na + Principal anions = Cl -, HCO 3 - ICF Principal cation = K +, Mg 2+ Principal anions = HPO 4 2- and negatively charged proteins -Although different ions dominate, both fluid divisions have the same osmotic concentrations. The ions cannot pass freely through cell membranes, but the water can by osmosis, and will move to equilibrium. Thus, solute/electrolyte concentrations of the fluid divisions will directly impact water distribution. The Four Rules of Regulation of Fluids and Electrolytes: 1) All homeostatic mechanisms for fluid composition respond to changes in the ECF -Receptors monitor the composition of plasma and CSF and trigger neural and endocrine mechanisms in response to change -Individual cells cannot be monitored and thus ICF has no direct impact 2) No receptors directly monitor fluid or electrolyte balance -Only plasma volume and osmotic concentration are monitored which give an indirect measure of fluid or electrolyte levels 3) Water follows salt -Cells cannot move water by active transport -Water will always move by osmosis and this movement cannot be stopped Amy Warenda Czura, Ph.D. 3 SCCC BIO132 Chapter 27 Handout
4 4) The body s content of water or electrolytes rises and falls with gain and loss to and from the environment Too much intake = high content in the body Too much loss = low content in the body Primary Regulatory Hormones: 1) Antidiuretic Hormone (ADH) - Osmoreceptors in the hypothalamus monitor the ECF and release ADH in response to high osmotic concentration (low water, high solute) - Osmotic concentration = ADH levels - Primary effects of ADH: A) stimulate water conservation at kidneys B) stimulate thirst center 2) Aldosterone - Released by the adrenal cortex to regulate Na + absorption and K + loss in the DCT and collecting system in the kidney - Retention of Na + will result in H 2 O conservation - Aldosterone is released in response to: A) high K + or low Na + in ECF (e.g. renal circulation) B) activation of the renin-angiotensin system due to a drop in BP or blood volume C) decline in kidney filtrate osmotic concentration at the DCT (more water less solutes) Addison s Disease = hypoaldosteronism: results in massive loss of NaCl and H 2 O in the urine; must adjust diet to compensate. 3) Natriuretic Peptides - ANP (atrial) and BNP (brain) are released in response to stretching of the heart wall - They function to reduce thirst and block release of ADH and aldosterone resulting in diuresis (fluid loss in the kidney) Fluid Balance: 1) Fluid movement within the ECF -Two important divisions of the ECF: 1) plasma ( ~ 20%) 2) interstitial fluid ( ~ 80%) -There is continuous flow between both: A) Hydrostatic pressure pushes water from the plasma into the interstitial fluid B) Colloid osmotic pressure draws water from the interstitial fluid to the plasma Edema = abnormal amount of water leaves the plasma and accumulates in the interstitial fluid 2) Fluid exchange with the environment A) Water losses ~ 2500 ml/day in urine, feces and insensible perspiration = obligatory water loss - Sensible perspiration: can reach up to 4 L/hr under extreme conditions - Fever: for each degree rise, insensible perspiration will increase by 200 ml/day Amy Warenda Czura, Ph.D. 4 SCCC BIO132 Chapter 27 Handout
5 B) Water Gains - Must match water losses or dehydration will result - Typical gain: ~1000 ml from drink ~1200 ml from food ~300 ml metabolic waste (Metabolic generation of water occurs from dehydration synthesis reactions and aerobic respiration in the mitochondria: water is the waste product of these types of reactions) - Water content is not easily measured, so ion content, particularly Na +, is measured and regulated instead Hyponatremia = hypotonic hydration: condition of low Na + concentration (i.e. excess water). Can be caused by: 1) Ingestion of a large volume of fresh water or injection of a hypotonic solution. 2) Inability to eliminate excess water at the kidney 3) Endocrine disorder (e.g. too much ADH) -This results in water moving from the ECF to the ICF causing cellular damage: water intoxication = cerebral edema and CNS dysfunction Hypernatremia = dehydration: condition of high Na + concentration (i.e. water depletion). -This results in decreased plasma volume and blood pressure that can lead to hypovolemic shock (inadequate circulation) 3) Fluid Shifts -Movement of water will occur between the ECF and ICF due to changes in osmotic concentrations -Water will always come to equilibrium: -If osmotic concentration of the ECF increases (becomes hypertonic) due to a loss of water but not electrolytes, water will leave the ICF -If osmotic concentration of ECF decreases (becomes hypotonic) due to a gain of water but not electrolytes, water will enter the ICF -The total amount of water is greater in the ICF than ECF: this allows the ICF to act as a reserve to accommodate changes in the ECF until hormones can restore homeostasis Electrolyte Balance: -Electrolyte balance is important because: 1) total electrolyte concentrations directly affect water balance 2) concentrations of individual electrolytes can affect cell functions -The two most important electrolytes are sodium and potassium: 1) Sodium Balance (normal blood values: meq/l *) -Na + is the dominant cation in the ECF -90% of the ECF osmotic concentration is due to sodium salts: NaCl and NaHCO 3 -The total amount of Na + in the ECF is due to a balance between Na + uptake in the digestive system and Na + excretion in urine and perspiration. Amy Warenda Czura, Ph.D. 5 SCCC BIO132 Chapter 27 Handout
6 -The overall sodium concentration in body fluids rarely changes because water always moves to compensate: e.g. high sodium levels in the blood will cause retention of water to maintain the same Na + concentration, but this results in a high blood volume (this is why salt is bad for hypertensive patients) -Minor gains and losses of Na + in the ECF are compensated by water in the ICF and later adjusted by hormonal activities: -ECF volume too low renin-angiotensin system is activated to conserve water and Na + -ECF volume too high natriuretic peptides released: block ADH and aldosterone resulting in water and Na + loss 2) Potassium Balance (normal blood values: meq/l) -K + is the dominant cation in the ICF (98% of the total body K + is inside cells) -The concentration of K + in the ECF depends on absorption in the GI vs. excretion in urine -The exchange pump at the kidney tubules secrete K + (or H + ) in order to reabsorb Na + -The rate of tubular secretion of K + in the kidney is controlled by three factors: 1. Changes in the K + concentration of the ECF K + in ECF = K + secretion 2. Changes in blood ph at low ph, H + is used for Na + reabsorption instead of K + at the exchange pump ph in ECF = K + secretion 3. Aldosterone levels aldosterone = Na + reabsorption and K + secretion Hypokalemia = low K + concentration in the ECF: will cause muscular weakness and mental confusion It can be caused by: 1. inadequate dietary K + intake 2. some diuretic drugs 3. excessive aldosterone 4. increased ph of ECF Hyperkalemia = high K + concentration in the ECF: will cause cardiac arrhythmia and flaccid paralysis It can be caused by: 1. renal failure 2. diuretics that block Na + reabsorption 3. a decline in ph 3) Other Electrolytes A) Calcium (normal blood values: meq/l or mg/dl) -Ca 2+ is the most abundant mineral in the body -99% is located in the skeleton for structure -Ca 2+ is important for: -muscular and neural cell activities -blood clotting -as a cofactor for enzymes -as a second messenger (intercellular signaling) Amy Warenda Czura, Ph.D. 6 SCCC BIO132 Chapter 27 Handout
7 -Ca 2+ homeostasis involves an interplay between skeletal reserves, uptake at the GI, and loss at the kidney -Parathyroid hormone and calcitriol function to raise blood Ca 2+ levels -Calcitonin functions to lower blood Ca 2+ levels Hypercalcemia = high Ca 2+ concentration in the ECF: can be due to hyper-parathyroidism or cancers. Can cause fatigue, confusion, cardiac arrhythmia, and calcification of organs Hypocalcemia = low Ca 2+ concentration in the ECF: can be due to hypo-parathyroidism, vitamin D deficiency, or renal failure. Can cause muscle spasms, convulsions, weak heartbeats, reduced clotting, and osteoporosis B) Magnesium (normal blood values: meq/l) -Most Mg 2+ is located in the skeleton -The remainder is located in the ICF -Mg 2+ is important as a cofactor for enzymes and as a structural component of the skeleton -Excess magnesium can cause lethargy and coma -Insufficient magnesium can cause convulsions C) Phosphate (normal blood values: 1-6 meq/l) -Free phosphate (HPO 4 2- ) is found in the ICF -It is used for: -mineralization of bone -formation of high energy compounds (ATP) -cofactors for enzymes -synthesis of nucleic acids -Phosphate tends to accompany calcium so physiological effects of excess or deficiency are related to calcium levels D) Chloride (normal blood values: meq/l) -Cl - is the most abundant anion in the ECF -The body has no use for it other than the fact that it travels with Na + -Excess can cause metabolic acidosis -Deficiencies can cause metabolic alkalosis Acid Base Balance Acid = a substance that dissociates to release H + ions Base = a substance that dissociates to release OH - ions or absorbs H + ions. The ph scale is used to measure the concentration of H + ions in a solution (ph = potential of Hydrogen ) Water is neutral: H + = OH -, ph 7 An acid solution (ph O - 7) has more H + than OH - A basic or alkaline solution (ph 7-14) has more OH - than H + Strong acids or bases dissociate completely in solution (e.g. HCl H + + Cl - ) Weak acids or bases do not completely dissociate: many molecules remain intact (e.g. H 2 CO 3 ) Amy Warenda Czura, Ph.D. 7 SCCC BIO132 Chapter 27 Handout
8 Normal ph of the ECF = Above or below this range will disrupt cell membranes and denature proteins Acidosis = ECF ph below 7.35 Alkalosis = ECF ph above 7.45 (Acidosis is the more common problem since metabolism generates acid waste products) 1) Types of Acids A) Volatile Acids can leave solution and enter the atmosphere e.g. CO 2 + H 2 O H 2 CO 3 H HCO 3 lungs blood B) Fixed Acids remain in solution until they are excreted e.g. sulfuric acid, phosphoric acid C) Organic Acids the products of metabolism These are usually metabolized into other wastes, but they can build up under anaerobic conditions or starvation e.g. Lactic acid, Ketone bodies 2) Mechanisms of ph Control Buffers = dissolved compounds that can remove H + ions to stabilize ph Usually buffers are a weak acid and its corresponding salt Three major buffering systems: A) The Protein Buffer System -Proteins are used to regulate ph in the ECF and ICF (most effective in the ICF) -Amino acids can be used to accept or release H + ions: -At typical body ph, most carboxyl groups exist as COO - and can accept H + ions if the ph begins to drop -Histidine and Cysteine remain as COOH at normal ph and can donate H + if the ph rises. -Most proteins can provide some degree of buffering with their carboxyl terminal. -Hemoglobin can have a great effect on blood ph The Hemoglobin (Hb) Buffering System (most effective in the ECF: blood) -In RBCs the enzyme carbonic anhydrase converts CO 2 into H 2 CO 3 which then dissociates -The H + remains inside RBCs, but the HCO - 3 enters the plasma where it can absorb excess H + B) The Carbonic Acid-Bicarbonate Buffer System -The most important buffer for the ECF (free in plasma or aided by Hb) -Carbon dioxide and water form carbonic acid which dissociates into hydrogen ions and bicarbonate ions -The bicarbonate ions can be used to absorb excess H + ions in the ECF and then can be released as CO 2 and H 2 O at the lungs -This buffering only works if: 1. CO 2 levels are normal 2. Respiration is functioning normally Amy Warenda Czura, Ph.D. 8 SCCC BIO132 Chapter 27 Handout
9 3. Free bicarbonate ions are available Bicarbonate ions can be generated from CO 2 + H 2 O or NaHCO 3 -, but to have free HCO 3 -, H + ions must be excreted at the kidney C) The Phosphate Buffer System -Phosphate is used to buffer ICF and urine -H 2 PO 4 or NaPO 4 can dissociate to generate HPO 4-2, which can absorb H + (as above, only as long as H + is excreted at the kidney) 3) Maintenance of Acid Base Balance -Buffering will only temporarily solve the H + problem: permanent removal as H 2 O at the lungs or through secretion at the kidney is necessary to maintain ph near neutral -ph homeostasis: A) Respiratory Compensation -Respiration rate is altered to control ph CO 2 = ph CO 2 = ph B) Renal Compensation -The rate of H + and HCO 3- secretion or reabsorption can be altered as necessary H + = ph HCO 3 - = ph 4) Disturbances of Acid Base Balance Many factors contribute: A) Disorders of buffers, respiratory performance or renal function B) Cardiovascular conditions that alter blood flow to the lungs and kidneys C) CNS disorders that effect cardiovascular or respiratory reflexes Respiratory acidosis = respiratory system fails to eliminate all CO 2 generated by the peripheral tissues causing a decline in ph. Can be caused by cardiac arrest, emphysema, congestive heart failure, pneumonia, pneumothorax, etc. Respiratory alkalosis = lungs remove too much CO 2 causing an increase in ph. Common result of hyperventilation due to anxiety or pain: this usually corrects itself Metabolic Acidosis 3 causes: 1. Production of fixed or organic acids A. Lactic acidosis results from hypoxia and is usually linked to respiratory acidosis B. Ketoacidosis results from starvation or diabetus mellitus 2. Impaired ability to excrete H + at kidneys, e.g. glomerulonephritis, diuretics 3. Severe bicarbonate loss, e.g. diarrhea: buffering agents from intestinal secretions are lost before they can be reabsorbed Metabolic Alkalosis = rare condition, caused by an increase in HCO 3 -. Secretion of HCl in the stomach releases HCO 3 - in the ECF. Severe vomiting will cause continuous acid production and loss. The corresponding HCO 3 - then accumulates in the ECF. Alkalosis can also result from chronic and excessive use of antacids. Amy Warenda Czura, Ph.D. 9 SCCC BIO132 Chapter 27 Handout
10 Age Related Changes: 1) reduced water content affects solute concentrations (elderly ~ 40% H 2 O) 2) reduced kidney function (loss of renal compensation) 3) loss of mineral reserves (Ca 2+, Mg 2+, HPO 4 2- ) 4) reduced lung function (loss of respiratory compensation) In case you ever need to know: *milliequivalents per liter (meq/l) - meq/l is a method of expressing concentration when the analytes are dissolved and disassociated in solution. meq/l is also equal to millimoles of charge per liter (mm+/l or mm-/l depending on valence). To express this as mg/l, divide the molecular weight of the ion (g/mole) by the valence (charge number) and multiply by the meq/l. e.g. Ca 2+ Calcium has a molecular weight of grams/mole Calcium has a valence of +2 The equivalent weight = (40.08grams/mole) / (2 equivalents/mole) = grams/eq (To convert to mg/meq you simply multiply g/eq by 1000 mg/g and divide by 1000 meq/eq, thus g/eq = mg/meq) If the blood contains 4.5 meq/l this is equal to 90 mg/l or 9 mg/100ml (100mL = 1dL) (40.08 / 2) x 4.5 = 90 Optional Computer Activity: Understanding Physiology to enhance comprehension of renal and respiratory compensation of acidosis and alkalosis: PhysioEx Exercise 47 (On the PhysioEx CD-ROM packaged with the Marieb lab book) pages P-116 to P-125 and P-169 (back of the book) in 8 th edition pages PEx-153 to PEx-164 (back of the book) in 9 th edition pages PEx-157 to PEx-168 (back of the book) in 10 th edition PhysioEx Exercise 10, pages PEx-149 to PEx-159 in 11 th & 12th editions Amy Warenda Czura, Ph.D. 10 SCCC BIO132 Chapter 27 Handout
Water, Electrolytes, and Acid-Base Balance
 Chapter 27 Water, Electrolytes, and Acid-Base Balance 1 Body Fluids Intracellular fluid compartment All fluids inside cells of body About 40% of total body weight Extracellular fluid compartment All fluids
Chapter 27 Water, Electrolytes, and Acid-Base Balance 1 Body Fluids Intracellular fluid compartment All fluids inside cells of body About 40% of total body weight Extracellular fluid compartment All fluids
Principles of Anatomy and Physiology
 Principles of Anatomy and Physiology 14 th Edition CHAPTER 27 Fluid, Electrolyte, and Acid Base Fluid Compartments and Fluid In adults, body fluids make up between 55% and 65% of total body mass. Body
Principles of Anatomy and Physiology 14 th Edition CHAPTER 27 Fluid, Electrolyte, and Acid Base Fluid Compartments and Fluid In adults, body fluids make up between 55% and 65% of total body mass. Body
CHAPTER 27 LECTURE OUTLINE
 CHAPTER 27 LECTURE OUTLINE I. INTRODUCTION A. Body fluid refers to body water and its dissolved substances. B. Regulatory mechanisms insure homeostasis of body fluids since their malfunction may seriously
CHAPTER 27 LECTURE OUTLINE I. INTRODUCTION A. Body fluid refers to body water and its dissolved substances. B. Regulatory mechanisms insure homeostasis of body fluids since their malfunction may seriously
Chapter 26 Fluid, Electrolyte, and Acid- Base Balance
 Chapter 26 Fluid, Electrolyte, and Acid- Base Balance 1 Body Water Content Infants: 73% or more water (low body fat, low bone mass) Adult males: ~60% water Adult females: ~50% water (higher fat content,
Chapter 26 Fluid, Electrolyte, and Acid- Base Balance 1 Body Water Content Infants: 73% or more water (low body fat, low bone mass) Adult males: ~60% water Adult females: ~50% water (higher fat content,
Fluid, Electrolyte, and Acid Base Balance
 25 Fluid, Electrolyte, and Acid Base Balance Lecture Presentation by Lori Garrett Note to the Instructor: For the third edition of Visual Anatomy & Physiology, we have updated our PowerPoints to fully
25 Fluid, Electrolyte, and Acid Base Balance Lecture Presentation by Lori Garrett Note to the Instructor: For the third edition of Visual Anatomy & Physiology, we have updated our PowerPoints to fully
Chapter 27: WATER, ELECTROLYTES, AND ACID-BASE BALANCE
 Chapter 27: WATER, ELECTROLYTES, AND ACID-BASE BALANCE I. RELATED TOPICS Integumentary system Cerebrospinal fluid Aqueous humor Digestive juices Feces Capillary dynamics Lymph circulation Edema Osmosis
Chapter 27: WATER, ELECTROLYTES, AND ACID-BASE BALANCE I. RELATED TOPICS Integumentary system Cerebrospinal fluid Aqueous humor Digestive juices Feces Capillary dynamics Lymph circulation Edema Osmosis
Acid-Base Balance 11/18/2011. Regulation of Potassium Balance. Regulation of Potassium Balance. Regulatory Site: Cortical Collecting Ducts.
 Influence of Other Hormones on Sodium Balance Acid-Base Balance Estrogens: Enhance NaCl reabsorption by renal tubules May cause water retention during menstrual cycles Are responsible for edema during
Influence of Other Hormones on Sodium Balance Acid-Base Balance Estrogens: Enhance NaCl reabsorption by renal tubules May cause water retention during menstrual cycles Are responsible for edema during
1. 09/07/16 Ch 1: Intro to Human A & P 1
 Table of Contents # Date Title Page # 1. 09/07/16 Ch 1: Intro to Human A & P 1 2. 09/19/16 Ch 18: Water, Electrolyte, and Acid-Base Balance 5 i 1 09/19/16 Chapter 18: Water, Electrolyte, and Acid-Base
Table of Contents # Date Title Page # 1. 09/07/16 Ch 1: Intro to Human A & P 1 2. 09/19/16 Ch 18: Water, Electrolyte, and Acid-Base Balance 5 i 1 09/19/16 Chapter 18: Water, Electrolyte, and Acid-Base
Chapter 24 Water, Electrolyte and Acid-Base Balance
 Chapter 24 Water, Electrolyte and Acid-Base Balance Total body water for 150 lb. male = 40L 65% ICF 35% ECF 25% tissue fluid 8% blood plasma, lymph 2% transcellular fluid (CSF, synovial fluid) Water Movement
Chapter 24 Water, Electrolyte and Acid-Base Balance Total body water for 150 lb. male = 40L 65% ICF 35% ECF 25% tissue fluid 8% blood plasma, lymph 2% transcellular fluid (CSF, synovial fluid) Water Movement
Acid Base Balance. Chapter 26 Balance. ph Imbalances. Acid Base Balance. CO 2 and ph. Carbonic Acid. Part 2. Acid/Base Balance
 Acid Base Balance Chapter 26 Balance Part 2. Acid/Base Balance Precisely balances production and loss of hydrogen ions (ph) The body generates acids during normal metabolism, tends to reduce ph Kidneys:
Acid Base Balance Chapter 26 Balance Part 2. Acid/Base Balance Precisely balances production and loss of hydrogen ions (ph) The body generates acids during normal metabolism, tends to reduce ph Kidneys:
Body water content. Fluid compartments. Regulation of water output. Water balance and ECF osmolallty. Regulation of water intake
 Body water content Infants have low body fat, low bone mass, and are 73% or more water Total water content declines throughout life Healthy males are about 60% water; females 50% This difference reflects
Body water content Infants have low body fat, low bone mass, and are 73% or more water Total water content declines throughout life Healthy males are about 60% water; females 50% This difference reflects
BIOL 2402 Fluid/Electrolyte Regulation
 Dr. Chris Doumen Collin County Community College BIOL 2402 Fluid/Electrolyte Regulation 1 Body Water Content On average, we are 50-60 % water For a 70 kg male = 40 liters water This water is divided into
Dr. Chris Doumen Collin County Community College BIOL 2402 Fluid/Electrolyte Regulation 1 Body Water Content On average, we are 50-60 % water For a 70 kg male = 40 liters water This water is divided into
Fluids and electrolytes
 Body Water Content Fluids and electrolytes Infants have low body fat, low bone mass, and are 73% or more water Total water content declines throughout life Healthy males are about 60% water; healthy females
Body Water Content Fluids and electrolytes Infants have low body fat, low bone mass, and are 73% or more water Total water content declines throughout life Healthy males are about 60% water; healthy females
Chapter 27: Fluid, Electrolyte, and Acid Base Balance
 Chapter 27: Fluid, Electrolyte, and Acid Base Balance Fluid, Electrolyte, and Acid Base Balance: An Overview, p. 995 Most of your body weight is water. Water accounts for up to 99 percent of the volume
Chapter 27: Fluid, Electrolyte, and Acid Base Balance Fluid, Electrolyte, and Acid Base Balance: An Overview, p. 995 Most of your body weight is water. Water accounts for up to 99 percent of the volume
ACID BASE BALANCE & BODY FLUID. Ani Retno Prijanti Renal and Body Fluids Module Juni 2008
 ACID BASE BALANCE & BODY FLUID Ani Retno Prijanti Renal and Body Fluids Module Juni 2008 1 Continuous Mixing of Body Fluids 2 Water Balance and ECF Osmolality To remain properly hydrated, water intake
ACID BASE BALANCE & BODY FLUID Ani Retno Prijanti Renal and Body Fluids Module Juni 2008 1 Continuous Mixing of Body Fluids 2 Water Balance and ECF Osmolality To remain properly hydrated, water intake
WATER, SODIUM AND POTASSIUM
 WATER, SODIUM AND POTASSIUM Attila Miseta Tamás Kőszegi Department of Laboratory Medicine, 2016 1 Average daily water intake and output of a normal adult 2 Approximate contributions to plasma osmolality
WATER, SODIUM AND POTASSIUM Attila Miseta Tamás Kőszegi Department of Laboratory Medicine, 2016 1 Average daily water intake and output of a normal adult 2 Approximate contributions to plasma osmolality
H 2 O, Electrolytes and Acid-Base Balance
 H 2 O, Electrolytes and Acid-Base Balance Body Fluids Intracellular Fluid Compartment All fluid inside the cells 40% of body weight Extracellular Fluid Compartment All fluid outside of cells 20% of body
H 2 O, Electrolytes and Acid-Base Balance Body Fluids Intracellular Fluid Compartment All fluid inside the cells 40% of body weight Extracellular Fluid Compartment All fluid outside of cells 20% of body
Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + )
 Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration in body fluids Precise regulation of ph at
Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration in body fluids Precise regulation of ph at
Chapter 19 The Urinary System Fluid and Electrolyte Balance
 Chapter 19 The Urinary System Fluid and Electrolyte Balance Chapter Outline The Concept of Balance Water Balance Sodium Balance Potassium Balance Calcium Balance Interactions between Fluid and Electrolyte
Chapter 19 The Urinary System Fluid and Electrolyte Balance Chapter Outline The Concept of Balance Water Balance Sodium Balance Potassium Balance Calcium Balance Interactions between Fluid and Electrolyte
Acid and Base Balance
 Acid and Base Balance 1 2 The Body and ph Homeostasis of ph is tightly controlled Extracellular fluid = 7.4 Blood = 7.35 7.45 < 7.35: Acidosis (acidemia) > 7.45: Alkalosis (alkalemia) < 6.8 or > 8.0: death
Acid and Base Balance 1 2 The Body and ph Homeostasis of ph is tightly controlled Extracellular fluid = 7.4 Blood = 7.35 7.45 < 7.35: Acidosis (acidemia) > 7.45: Alkalosis (alkalemia) < 6.8 or > 8.0: death
Major intra and extracellular ions Lec: 1
 Major intra and extracellular ions Lec: 1 The body fluids are solutions of inorganic and organic solutes. The concentration balance of the various components is maintained in order for the cell and tissue
Major intra and extracellular ions Lec: 1 The body fluids are solutions of inorganic and organic solutes. The concentration balance of the various components is maintained in order for the cell and tissue
BIOL 221 Chapter 26 Fluids & Electrolytes. 35 slides
 BIOL 221 Chapter 26 Fluids & Electrolytes 35 slides 1 Body Water Content Total Body Water is the percentage of a person s weight that is water. TBW can easily vary due to: gender males have higher TBW
BIOL 221 Chapter 26 Fluids & Electrolytes 35 slides 1 Body Water Content Total Body Water is the percentage of a person s weight that is water. TBW can easily vary due to: gender males have higher TBW
Body Water Content Total Body Water is the percentage of a person s weight that is water. TBW can easily vary due to: gender
 BIOL 221 Chapter 26 Fluids & Electrolytes 35 slides 1 Body Water Content Total Body Water is the percentage of a person s weight that is water. TBW can easily vary due to: gender males have higher TBW
BIOL 221 Chapter 26 Fluids & Electrolytes 35 slides 1 Body Water Content Total Body Water is the percentage of a person s weight that is water. TBW can easily vary due to: gender males have higher TBW
Acid-Base Balance Dr. Gary Mumaugh
 Acid-Base Balance Dr. Gary Mumaugh Introduction Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration
Acid-Base Balance Dr. Gary Mumaugh Introduction Acid-base balance is one of the most important of the body s homeostatic mechanisms Acid-base balance refers to regulation of hydrogen ion (H + ) concentration
Fluid and Electrolytes P A R T 4
 Fluid and Electrolytes P A R T 4 Mechanisms that control acid-base homeostasis Acids and bases continually enter and leave body Hydrogen ions also result from metabolic activity Acids Hydrogen ion donors
Fluid and Electrolytes P A R T 4 Mechanisms that control acid-base homeostasis Acids and bases continually enter and leave body Hydrogen ions also result from metabolic activity Acids Hydrogen ion donors
The Urinary System 15PART B. PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College
 PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College The Urinary System 15PART B Ureters Slender tubes attaching the kidney to the bladder Continuous with
PowerPoint Lecture Slide Presentation by Patty Bostwick-Taylor, Florence-Darlington Technical College The Urinary System 15PART B Ureters Slender tubes attaching the kidney to the bladder Continuous with
FLUID, ELECTROLYTES, AND ACID-BASE HOMEOSTASIS
 Chapter 27 1 BIOLOGY 2402 Anatomy and Physiology Lecture Chapter 27 FLUID, ELECTROLYTES, AND ACID-BASE HOMEOSTASIS Chapter 27 2 FLUID, ELECTROLYTES, AND ACID-BASE HOMEOSTAIS Body fluid refers to the body
Chapter 27 1 BIOLOGY 2402 Anatomy and Physiology Lecture Chapter 27 FLUID, ELECTROLYTES, AND ACID-BASE HOMEOSTASIS Chapter 27 2 FLUID, ELECTROLYTES, AND ACID-BASE HOMEOSTAIS Body fluid refers to the body
Body Water Content Total Body Water is the percentage of a person s weight that is water. TBW can easily vary due to: gender
 BIOL 221 Chapter 26 Fluids & Electrolytes 35 slides!1 Body Water Content Total Body Water is the percentage of a person s weight that is water. TBW can easily vary due to: gender males have higher TBW
BIOL 221 Chapter 26 Fluids & Electrolytes 35 slides!1 Body Water Content Total Body Water is the percentage of a person s weight that is water. TBW can easily vary due to: gender males have higher TBW
Body Water Content Infants have low body fat, low bone mass, and are 73% or more water Total water content declines throughout life Healthy males are
 Fluid, Electrolyte, and Acid-Base Balance Body Water Content Infants have low body fat, low bone mass, and are 73% or more water Total water content declines throughout life Healthy males are about 60%
Fluid, Electrolyte, and Acid-Base Balance Body Water Content Infants have low body fat, low bone mass, and are 73% or more water Total water content declines throughout life Healthy males are about 60%
UNIT VI: ACID BASE IMBALANCE
 UNIT VI: ACID BASE IMBALANCE 1 Objectives: Review the physiological mechanism responsible to regulate acid base balance in the body i.e.: Buffers (phosphate, hemoglobin, carbonate) Renal mechanism Respiratory
UNIT VI: ACID BASE IMBALANCE 1 Objectives: Review the physiological mechanism responsible to regulate acid base balance in the body i.e.: Buffers (phosphate, hemoglobin, carbonate) Renal mechanism Respiratory
5/18/2017. Specific Electrolytes. Sodium. Sodium. Sodium. Sodium. Sodium
 Specific Electrolytes Hyponatremia Hypervolemic Replacing water (not electrolytes) after perspiration Freshwater near-drowning Syndrome of Inappropriate ADH Secretion (SIADH) Hypovolemic GI disease (decreased
Specific Electrolytes Hyponatremia Hypervolemic Replacing water (not electrolytes) after perspiration Freshwater near-drowning Syndrome of Inappropriate ADH Secretion (SIADH) Hypovolemic GI disease (decreased
Acids, Bases, and Salts
 Acid / Base Balance Objectives Define an acid, a base, and the measure of ph. Discuss acid/base balance, the effects of acidosis or alkalosis on the body, and the mechanisms in place to maintain balance
Acid / Base Balance Objectives Define an acid, a base, and the measure of ph. Discuss acid/base balance, the effects of acidosis or alkalosis on the body, and the mechanisms in place to maintain balance
Chapter 26 Electrolyte & Acid-Base Balance
 Chapter 26 Electrolyte & Acid-Base Balance Slides by Barbara Heard and W. Rose. figures from Marieb & Hoehn 9 th ed. Portions copyright Pearson Education Major fluid compartments of the body Total body
Chapter 26 Electrolyte & Acid-Base Balance Slides by Barbara Heard and W. Rose. figures from Marieb & Hoehn 9 th ed. Portions copyright Pearson Education Major fluid compartments of the body Total body
1/3/2008. Karen Burke Priscilla LeMone Elaine Mohn-Brown. Medical-Surgical Nursing Care, 2e Karen Burke, Priscilla LeMone, and Elaine Mohn-Brown
 Medical-Surgical Nursing Care Second Edition Karen Burke Priscilla LeMone Elaine Mohn-Brown Chapter 7 Caring for Clients with Altered Fluid, Electrolyte, or Acid-Base Balance Water Primary component of
Medical-Surgical Nursing Care Second Edition Karen Burke Priscilla LeMone Elaine Mohn-Brown Chapter 7 Caring for Clients with Altered Fluid, Electrolyte, or Acid-Base Balance Water Primary component of
Renal Physiology. April, J. Mohan, PhD. Lecturer, Physiology Unit, Faculty of Medical Sciences, U.W.I., St Augustine.
 Renal Physiology April, 2011 J. Mohan, PhD. Lecturer, Physiology Unit, Faculty of Medical Sciences, U.W.I., St Augustine. Office : Room 105, Physiology Unit. References: Koeppen B.E. & Stanton B.A. (2010).
Renal Physiology April, 2011 J. Mohan, PhD. Lecturer, Physiology Unit, Faculty of Medical Sciences, U.W.I., St Augustine. Office : Room 105, Physiology Unit. References: Koeppen B.E. & Stanton B.A. (2010).
Amjad Bani Hani Ass.Prof. of Cardiac Surgery & Intensive Care FLUIDS AND ELECTROLYTES
 Amjad Bani Hani Ass.Prof. of Cardiac Surgery & Intensive Care FLUIDS AND ELECTROLYTES Body Water Content Water Balance: Normal 2500 2000 1500 1000 500 Metab Food Fluids Stool Breath Sweat Urine
Amjad Bani Hani Ass.Prof. of Cardiac Surgery & Intensive Care FLUIDS AND ELECTROLYTES Body Water Content Water Balance: Normal 2500 2000 1500 1000 500 Metab Food Fluids Stool Breath Sweat Urine
Part 1 The Cell and the Cellular Environment
 1 Chapter 3 Anatomy and Physiology Part 1 The Cell and the Cellular Environment 2 The Human Cell The is the fundamental unit of the human body. Cells contain all the necessary for life functions. 3 Cell
1 Chapter 3 Anatomy and Physiology Part 1 The Cell and the Cellular Environment 2 The Human Cell The is the fundamental unit of the human body. Cells contain all the necessary for life functions. 3 Cell
Chapter 2. Fluid, Electrolyte, and Acid-Base Imbalances
 Chapter 2 Fluid, Electrolyte, and Acid-Base Imbalances Review of Concepts and Processes The major component of the body is water. Water is located in these compartments: Intracellular fluid (ICF) compartment
Chapter 2 Fluid, Electrolyte, and Acid-Base Imbalances Review of Concepts and Processes The major component of the body is water. Water is located in these compartments: Intracellular fluid (ICF) compartment
Principles of Fluid Balance
 Principles of Fluid Balance I. The Cellular Environment: Fluids and Electrolytes A. Water 1. Total body water (TBW) = 60% of total body weight 2. Fluid Compartments in the Body a. Intracellular Compartment
Principles of Fluid Balance I. The Cellular Environment: Fluids and Electrolytes A. Water 1. Total body water (TBW) = 60% of total body weight 2. Fluid Compartments in the Body a. Intracellular Compartment
Renal Physiology Part II. Bio 219 Napa Valley College Dr. Adam Ross
 Renal Physiology Part II Bio 219 Napa Valley College Dr. Adam Ross Fluid and Electrolyte balance As we know from our previous studies: Water and ions need to be balanced in order to maintain proper homeostatic
Renal Physiology Part II Bio 219 Napa Valley College Dr. Adam Ross Fluid and Electrolyte balance As we know from our previous studies: Water and ions need to be balanced in order to maintain proper homeostatic
Chapter 20 8/23/2016. Fluids and Electrolytes. Fluid (Water) Fluid (Water) (Cont.) Functions
 Chapter 20 Fluids and Electrolytes All items and derived items 2015, 2011, 2006 by Mosby, Inc., an imprint of Elsevier Inc. All rights reserved. Fluid (Water) Functions Provides an extracellular transportation
Chapter 20 Fluids and Electrolytes All items and derived items 2015, 2011, 2006 by Mosby, Inc., an imprint of Elsevier Inc. All rights reserved. Fluid (Water) Functions Provides an extracellular transportation
Chapter 24 Lecture Outline
 Chapter 24 Lecture Outline See separate PowerPoint slides for all figures and tables preinserted into PowerPoint without notes. Copyright McGraw-Hill Education. Permission required for reproduction or
Chapter 24 Lecture Outline See separate PowerPoint slides for all figures and tables preinserted into PowerPoint without notes. Copyright McGraw-Hill Education. Permission required for reproduction or
Chapter 15 Fluid and Acid-Base Balance
 Chapter 15 Fluid and Acid-Base Balance by Dr. Jay M. Templin Brooks/Cole - Thomson Learning Fluid Balance Water constitutes ~60% of body weight. All cells and tissues are surrounded by an aqueous environment.
Chapter 15 Fluid and Acid-Base Balance by Dr. Jay M. Templin Brooks/Cole - Thomson Learning Fluid Balance Water constitutes ~60% of body weight. All cells and tissues are surrounded by an aqueous environment.
Acid-Base Balance * OpenStax
 OpenStax-CNX module: m46409 1 Acid-Base Balance * OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section, you will be
OpenStax-CNX module: m46409 1 Acid-Base Balance * OpenStax This work is produced by OpenStax-CNX and licensed under the Creative Commons Attribution License 3.0 By the end of this section, you will be
Fluid and electrolyte balance, imbalance
 Fluid and electrolyte balance, imbalance Body fluid The fluids are distributed throughout the body in various compartments. Body fluid is composed primarily of water Water is the solvent in which all solutes
Fluid and electrolyte balance, imbalance Body fluid The fluids are distributed throughout the body in various compartments. Body fluid is composed primarily of water Water is the solvent in which all solutes
Instrumental determination of electrolytes in urine. Amal Alamri
 Instrumental determination of electrolytes in urine Amal Alamri What is the Electrolytes? Electrolytes are positively and negatively chargedions, Found in Within body's cells extracellular fluids, including
Instrumental determination of electrolytes in urine Amal Alamri What is the Electrolytes? Electrolytes are positively and negatively chargedions, Found in Within body's cells extracellular fluids, including
SOCM Fluids Electrolytes and Replacement Products PFN: SOMRXL09. Terminal Learning Objective. References. Hours: 2.0 Last updated: November 2015
 SOCM Fluids Electrolytes and Replacement Products PFN: SOMRXL09 Hours: 2.0 Last updated: November 2015 Slide 1 Terminal Learning Objective Action: Communicate knowledge of Fluid, Electrolyte, and Acid
SOCM Fluids Electrolytes and Replacement Products PFN: SOMRXL09 Hours: 2.0 Last updated: November 2015 Slide 1 Terminal Learning Objective Action: Communicate knowledge of Fluid, Electrolyte, and Acid
Acid/Base Balance. the concentrations of these two ions affect the acidity or alkalinity of body fluids
 Acid/Base Balance some of most critical ions in body fluids are H + (hydrogen) and OH - (hydroxyl) ions the concentrations of these two ions affect the acidity or alkalinity of body fluids acidity/alkalinity
Acid/Base Balance some of most critical ions in body fluids are H + (hydrogen) and OH - (hydroxyl) ions the concentrations of these two ions affect the acidity or alkalinity of body fluids acidity/alkalinity
Body fluids. Lecture 13:
 Lecture 13: Body fluids Body fluids are distributed in compartments: A. Intracellular compartment: inside the cells of the body (two thirds) B. Extracellular compartment: (one third) it is divided into
Lecture 13: Body fluids Body fluids are distributed in compartments: A. Intracellular compartment: inside the cells of the body (two thirds) B. Extracellular compartment: (one third) it is divided into
Physio 12 -Summer 02 - Renal Physiology - Page 1
 Physiology 12 Kidney and Fluid regulation Guyton Ch 20, 21,22,23 Roles of the Kidney Regulation of body fluid osmolarity and electrolytes Regulation of acid-base balance (ph) Excretion of natural wastes
Physiology 12 Kidney and Fluid regulation Guyton Ch 20, 21,22,23 Roles of the Kidney Regulation of body fluid osmolarity and electrolytes Regulation of acid-base balance (ph) Excretion of natural wastes
Potassium regulation. -Kidney is a major regulator for potassium Homeostasis.
 Potassium regulation. -Kidney is a major regulator for potassium Homeostasis. Normal potassium intake, distribution, and output from the body. Effects of severe hyperkalemia Partial depolarization of cell
Potassium regulation. -Kidney is a major regulator for potassium Homeostasis. Normal potassium intake, distribution, and output from the body. Effects of severe hyperkalemia Partial depolarization of cell
Regulation of fluid and electrolytes balance
 Regulation of fluid and electrolytes balance Three Compartment Fluid Compartments Intracellular = Cytoplasmic (inside cells) Extracellular compartment is subdivided into Interstitial = Intercellular +
Regulation of fluid and electrolytes balance Three Compartment Fluid Compartments Intracellular = Cytoplasmic (inside cells) Extracellular compartment is subdivided into Interstitial = Intercellular +
RENAL FUNCTION An Overview
 RENAL FUNCTION An Overview UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY PBL MBBS II SEMINAR VJ. Temple 1 Kidneys
RENAL FUNCTION An Overview UNIVERSITY OF PNG SCHOOL OF MEDICINE AND HEALTH SCIENCES DIVISION OF BASIC MEDICAL SCIENCES DISCIPLINE OF BIOCHEMISTRY & MOLECULAR BIOLOGY PBL MBBS II SEMINAR VJ. Temple 1 Kidneys
Fluid, Electrolyte, and Acid Base Balance
 Chapter 26 Fluid, Electrolyte, and Acid Base Balance Bi 233 Body Water Content Largest component of the body Infants have low body fat, low bone mass, and are 73% or more water Healthy males are about
Chapter 26 Fluid, Electrolyte, and Acid Base Balance Bi 233 Body Water Content Largest component of the body Infants have low body fat, low bone mass, and are 73% or more water Healthy males are about
Acid Base Balance. Professor Dr. Raid M. H. Al-Salih. Clinical Chemistry Professor Dr. Raid M. H. Al-Salih
 Acid Base Balance 1 HYDROGEN ION CONCENTRATION and CONCEPT OF ph Blood hydrogen ion concentration (abbreviated [H + ]) is maintained within tight limits in health, with the normal concentration being between
Acid Base Balance 1 HYDROGEN ION CONCENTRATION and CONCEPT OF ph Blood hydrogen ion concentration (abbreviated [H + ]) is maintained within tight limits in health, with the normal concentration being between
Carbon Dioxide Transport. Carbon Dioxide. Carbon Dioxide Transport. Carbon Dioxide Transport - Plasma. Hydrolysis of Water
 Module H: Carbon Dioxide Transport Beachey Ch 9 & 10 Egan pp. 244-246, 281-284 Carbon Dioxide Transport At the end of today s session you will be able to : Describe the relationship free hydrogen ions
Module H: Carbon Dioxide Transport Beachey Ch 9 & 10 Egan pp. 244-246, 281-284 Carbon Dioxide Transport At the end of today s session you will be able to : Describe the relationship free hydrogen ions
Disclaimer. Chapter 3 Disorder of Water, Electrolyte and Acid-base Professor A. S. Alhomida. Disorder of Water and Electrolyte
 Disclaimer King Saud University College of Science Department of Biochemistry The texts, tables, figures and images contained in this course presentation (BCH 376) are not my own, they can be found on:
Disclaimer King Saud University College of Science Department of Biochemistry The texts, tables, figures and images contained in this course presentation (BCH 376) are not my own, they can be found on:
Acid-Base Imbalance-2 Lecture 9 (12/4/2015) Yanal A. Shafagoj MD. PhD
 AcidBase Imbalance2 Lecture 9 (12/4/2015) Yanal A. Shafagoj MD. PhD Introduction Disturbance in acidbase balance are common clinical problem that range in severity from mild to life threatening, the acute
AcidBase Imbalance2 Lecture 9 (12/4/2015) Yanal A. Shafagoj MD. PhD Introduction Disturbance in acidbase balance are common clinical problem that range in severity from mild to life threatening, the acute
The Urinary System PART B
 15 The Urinary System PART B PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB Urinary Bladder Smooth,
15 The Urinary System PART B PowerPoint Lecture Slide Presentation by Jerry L. Cook, Sam Houston University ESSENTIALS OF HUMAN ANATOMY & PHYSIOLOGY EIGHTH EDITION ELAINE N. MARIEB Urinary Bladder Smooth,
26 FLUID, ELECTROLYTE, AND ACID-BASE BALANCE
 CHAPTER 26 FLUID, ELECTROLYTE, AND ACID-BASE BALANCE 1173 26 FLUID, ELECTROLYTE, AND ACID-BASE BALANCE Figure 26.1 Venus Williams Perspiring on the Tennis Court The body has critically important mechanisms
CHAPTER 26 FLUID, ELECTROLYTE, AND ACID-BASE BALANCE 1173 26 FLUID, ELECTROLYTE, AND ACID-BASE BALANCE Figure 26.1 Venus Williams Perspiring on the Tennis Court The body has critically important mechanisms
Acid - base equilibrium
 Acid base equilibrium ph concept ph = log [H + ] ph [H+] 1 100 mmol/l D = 90 mmol/l 2 10 mmol/l D = 9 mmol/l 3 1 mmol/l 2 ph = log [H + ] 3 ph ph = log [H + ] ph of capillary blood norm: 7,35 7,45 Sorensen
Acid base equilibrium ph concept ph = log [H + ] ph [H+] 1 100 mmol/l D = 90 mmol/l 2 10 mmol/l D = 9 mmol/l 3 1 mmol/l 2 ph = log [H + ] 3 ph ph = log [H + ] ph of capillary blood norm: 7,35 7,45 Sorensen
BODY FLUID. Outline. Functions of body fluid Water distribution in the body Maintenance of body fluid. Regulation of fluid homeostasis
 BODY FLUID Nutritional Biochemistry Yue-Hwa Chen Dec 13, 2007 Chen 1 Outline Functions of body fluid Water distribution in the body Maintenance of body fluid Intake vs output Regulation of body fluid Fluid
BODY FLUID Nutritional Biochemistry Yue-Hwa Chen Dec 13, 2007 Chen 1 Outline Functions of body fluid Water distribution in the body Maintenance of body fluid Intake vs output Regulation of body fluid Fluid
12/7/10. Excretory System. The basic function of the excretory system is to regulate the volume and composition of body fluids by:
 Excretory System The basic function of the excretory system is to regulate the volume and composition of body fluids by: o o removing wastes returning needed substances to the body for reuse Body systems
Excretory System The basic function of the excretory system is to regulate the volume and composition of body fluids by: o o removing wastes returning needed substances to the body for reuse Body systems
Slide 1. Slide 2. Slide 3. Learning Outcomes. Acid base terminology ARTERIAL BLOOD GAS INTERPRETATION
 Slide 1 ARTERIAL BLOOD GAS INTERPRETATION David O Neill MSc BSc RN NMP FHEA Associate Lecturer (Non Medical Prescribing) Cardiff University Advanced Nurse Practitioner Respiratory Medicine Slide 2 Learning
Slide 1 ARTERIAL BLOOD GAS INTERPRETATION David O Neill MSc BSc RN NMP FHEA Associate Lecturer (Non Medical Prescribing) Cardiff University Advanced Nurse Practitioner Respiratory Medicine Slide 2 Learning
Other Factors Affecting GFR. Chapter 25. After Filtration. Reabsorption and Secretion. 5 Functions of the PCT
 Other Factors Affecting GFR Chapter 25 Part 2. Renal Physiology Nitric oxide vasodilator produced by the vascular endothelium Adenosine vasoconstrictor of renal vasculature Endothelin a powerful vasoconstrictor
Other Factors Affecting GFR Chapter 25 Part 2. Renal Physiology Nitric oxide vasodilator produced by the vascular endothelium Adenosine vasoconstrictor of renal vasculature Endothelin a powerful vasoconstrictor
Water compartments inside and outside cells maintain a balanced distribution of total body water.
 Chapter 9 Water Balance Chapter 9 Lesson 9.1 Key Concepts Water compartments inside and outside cells maintain a balanced distribution of total body water. The concentration of various solute particles
Chapter 9 Water Balance Chapter 9 Lesson 9.1 Key Concepts Water compartments inside and outside cells maintain a balanced distribution of total body water. The concentration of various solute particles
RENAL PHYSIOLOGY. Physiology Unit 4
 RENAL PHYSIOLOGY Physiology Unit 4 Renal Functions Primary Function is to regulate the chemistry of plasma through urine formation Additional Functions Regulate concentration of waste products Regulate
RENAL PHYSIOLOGY Physiology Unit 4 Renal Functions Primary Function is to regulate the chemistry of plasma through urine formation Additional Functions Regulate concentration of waste products Regulate
Acids and Bases their definitions and meanings
 Acids and Bases their definitions and meanings Molecules containing hydrogen atoms that can release hydrogen ions in solutions are referred to as acids. (HCl H + Cl ) (H 2 CO 3 H + HCO 3 ) A base is an
Acids and Bases their definitions and meanings Molecules containing hydrogen atoms that can release hydrogen ions in solutions are referred to as acids. (HCl H + Cl ) (H 2 CO 3 H + HCO 3 ) A base is an
Kidneys in regulation of homeostasis
 Kidneys in regulation of homeostasis Assoc. Prof. MUDr. Markéta Bébarová, Ph.D. Department of Physiology Faculty of Medicine, Masaryk University This presentation includes only the most important terms
Kidneys in regulation of homeostasis Assoc. Prof. MUDr. Markéta Bébarová, Ph.D. Department of Physiology Faculty of Medicine, Masaryk University This presentation includes only the most important terms
PARAMEDIC RESOURCE MANUAL
 ONTARIO BASE HOSPITAL GROUP PARAMEDIC RESOURCE MANUAL ACID-BASE BALANCE SECTION SIX Version 1.1 2010 Update PARAMEDIC RESOURCE MANUAL OBJECTIVES: ACID-BASE BALANCE The objectives indicate what you should
ONTARIO BASE HOSPITAL GROUP PARAMEDIC RESOURCE MANUAL ACID-BASE BALANCE SECTION SIX Version 1.1 2010 Update PARAMEDIC RESOURCE MANUAL OBJECTIVES: ACID-BASE BALANCE The objectives indicate what you should
Emergency Medical Training Services Emergency Medical Technician Paramedic Program Outlines Outline Topic: Patho Instructor Notes Revised: 11/2013
 Emergency Medical Training Services Emergency Medical Technician Paramedic Program Outlines Outline Topic: Patho Instructor Notes Revised: 11/2013 Cells form 4 basic tissue groups: 1. Epithelial 2. Connective
Emergency Medical Training Services Emergency Medical Technician Paramedic Program Outlines Outline Topic: Patho Instructor Notes Revised: 11/2013 Cells form 4 basic tissue groups: 1. Epithelial 2. Connective
BIPN100 F15 Human Physiology (Kristan) Lecture 18: Endocrine control of renal function. p. 1
 BIPN100 F15 Human Physiology (Kristan) Lecture 18: Endocrine control of renal function. p. 1 Terms you should understand by the end of this section: diuresis, antidiuresis, osmoreceptors, atrial stretch
BIPN100 F15 Human Physiology (Kristan) Lecture 18: Endocrine control of renal function. p. 1 Terms you should understand by the end of this section: diuresis, antidiuresis, osmoreceptors, atrial stretch
EXCRETION QUESTIONS. Use the following information to answer the next two questions.
 EXCRETION QUESTIONS Use the following information to answer the next two questions. 1. Filtration occurs at the area labeled A. V B. X C. Y D. Z 2. The antidiuretic hormone (vasopressin) acts on the area
EXCRETION QUESTIONS Use the following information to answer the next two questions. 1. Filtration occurs at the area labeled A. V B. X C. Y D. Z 2. The antidiuretic hormone (vasopressin) acts on the area
Questions? Homework due in lab 6. PreLab #6 HW 15 & 16 (follow directions, 6 points!)
 Questions? Homework due in lab 6 PreLab #6 HW 15 & 16 (follow directions, 6 points!) Part 3 Variations in Urine Formation Composition varies Fluid volume Solute concentration Variations in Urine Formation
Questions? Homework due in lab 6 PreLab #6 HW 15 & 16 (follow directions, 6 points!) Part 3 Variations in Urine Formation Composition varies Fluid volume Solute concentration Variations in Urine Formation
Disorders of Acid-Base
 Disorders of Acid-Base Balance Bởi: OpenStaxCollege Normal arterial blood ph is restricted to a very narrow range of 7.35 to 7.45. A person who has a blood ph below 7.35 is considered to be in acidosis
Disorders of Acid-Base Balance Bởi: OpenStaxCollege Normal arterial blood ph is restricted to a very narrow range of 7.35 to 7.45. A person who has a blood ph below 7.35 is considered to be in acidosis
Kidneys and Homeostasis
 16 The Urinary System The Urinary System OUTLINE: Eliminating Waste Components of the Urinary System Kidneys and Homeostasis Urination Urinary Tract Infections Eliminating Waste Excretion Elimination of
16 The Urinary System The Urinary System OUTLINE: Eliminating Waste Components of the Urinary System Kidneys and Homeostasis Urination Urinary Tract Infections Eliminating Waste Excretion Elimination of
Renal physiology D.HAMMOUDI.MD
 Renal physiology D.HAMMOUDI.MD Functions Regulating blood ionic composition Regulating blood ph Regulating blood volume Regulating blood pressure Produce calcitrol and erythropoietin Regulating blood glucose
Renal physiology D.HAMMOUDI.MD Functions Regulating blood ionic composition Regulating blood ph Regulating blood volume Regulating blood pressure Produce calcitrol and erythropoietin Regulating blood glucose
Chapter 10: Urinary System & Excretion
 Chapter 10: Urinary System & Excretion Organs of Urinary System Kidneys (2) form urine Ureters (2) Carry urine from kidneys to bladder Bladder Stores urine Urethra Carries urine from bladder to outside
Chapter 10: Urinary System & Excretion Organs of Urinary System Kidneys (2) form urine Ureters (2) Carry urine from kidneys to bladder Bladder Stores urine Urethra Carries urine from bladder to outside
NORMAL POTASSIUM DISTRIBUTION AND BALANCE
 NORMAL POTASSIUM DISTRIBUTION AND BALANCE 98% of body potassium is contained within cells, principally muscle cells, and is readily exchangeable. Only 2% is in ECF. Daily intake exceeds the amount in ECF.
NORMAL POTASSIUM DISTRIBUTION AND BALANCE 98% of body potassium is contained within cells, principally muscle cells, and is readily exchangeable. Only 2% is in ECF. Daily intake exceeds the amount in ECF.
The principal functions of the kidneys
 Renal physiology The principal functions of the kidneys Formation and excretion of urine Excretion of waste products, drugs, and toxins Regulation of body water and mineral content of the body Maintenance
Renal physiology The principal functions of the kidneys Formation and excretion of urine Excretion of waste products, drugs, and toxins Regulation of body water and mineral content of the body Maintenance
Osmotic Regulation and the Urinary System. Chapter 50
 Osmotic Regulation and the Urinary System Chapter 50 Challenge Questions Indicate the areas of the nephron that the following hormones target, and describe when and how the hormones elicit their actions.
Osmotic Regulation and the Urinary System Chapter 50 Challenge Questions Indicate the areas of the nephron that the following hormones target, and describe when and how the hormones elicit their actions.
Renal physiology V. Regulation of acid-base balance. Dr Alida Koorts BMS
 Renal physiology V Regulation of acidbase balance Dr Alida Koorts BMS 712 012 319 2921 akoorts@medic.up.ac.za Hydrogen ions (H + ): Concentration and origin Concentration in arterial blood, resting: [H
Renal physiology V Regulation of acidbase balance Dr Alida Koorts BMS 712 012 319 2921 akoorts@medic.up.ac.za Hydrogen ions (H + ): Concentration and origin Concentration in arterial blood, resting: [H
D fini n tion: p = = -log [H+] ph=7 me m an s 10-7 Mol M H+ + (100 nmol m /l); ) p ; H=8 me m an s 10-8 Mol M H+ + (10 (10 n nmol m /l) Nor
![D fini n tion: p = = -log [H+] ph=7 me m an s 10-7 Mol M H+ + (100 nmol m /l); ) p ; H=8 me m an s 10-8 Mol M H+ + (10 (10 n nmol m /l) Nor D fini n tion: p = = -log [H+] ph=7 me m an s 10-7 Mol M H+ + (100 nmol m /l); ) p ; H=8 me m an s 10-8 Mol M H+ + (10 (10 n nmol m /l) Nor](/thumbs/75/72756397.jpg) Definition: ph regulation ph = -log [H + ] ph=7 means 10-7 Mol H + (100 nmol/l); ph=8 means 10 Normal plasma value: 7.35-7.45; 7.45; (H Acidosis: ph7.45 Intracellular ph = 7.1-7.3
Definition: ph regulation ph = -log [H + ] ph=7 means 10-7 Mol H + (100 nmol/l); ph=8 means 10 Normal plasma value: 7.35-7.45; 7.45; (H Acidosis: ph7.45 Intracellular ph = 7.1-7.3
1. a)label the parts indicated above and give one function for structures Y and Z
 Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- renal cortex - X- renal medulla Y- renal pelvis collecting center of urine and then
Excretory System 1 1. Excretory System a)label the parts indicated above and give one function for structures Y and Z W- renal cortex - X- renal medulla Y- renal pelvis collecting center of urine and then
Fluid, Electrolyte, and Acid-Base Balance. Maintaining Water and Electrolyte Balance of Blood
 514 Essentials of Human Anatomy and Physiology Intracellular fluid volume = 25 L, 40% body weight Total body water volume = 40 L, 60% body weight Extracellular fluid (ECF) volume =15 L, 20% body weight
514 Essentials of Human Anatomy and Physiology Intracellular fluid volume = 25 L, 40% body weight Total body water volume = 40 L, 60% body weight Extracellular fluid (ECF) volume =15 L, 20% body weight
Fluid, electrolyte, and acid-base balance
 Fluid, electrolyte, and acid-base balance Chapter 50 Ra'eda Almashaqba 1 Fluid, electrolyte, and acid-base balance About 46% to 60%of the average adult's weight is water, which is vital to health and normal
Fluid, electrolyte, and acid-base balance Chapter 50 Ra'eda Almashaqba 1 Fluid, electrolyte, and acid-base balance About 46% to 60%of the average adult's weight is water, which is vital to health and normal
Calcium (Ca 2+ ) mg/dl
 Quick Guide to Laboratory Values Use this handy cheat-sheet to help you monitor laboratory values related to fluid and electrolyte status. Remember, normal values may vary according to techniques used
Quick Guide to Laboratory Values Use this handy cheat-sheet to help you monitor laboratory values related to fluid and electrolyte status. Remember, normal values may vary according to techniques used
Chapter 12. Excretion and the Interaction of Systems
 Chapter 12 Excretion and the Interaction of Systems 1 2 Goals for This Chapter 1. Identify the main structures and functions of the human excretory system 2. Explain the function of the nephron 3. Describe
Chapter 12 Excretion and the Interaction of Systems 1 2 Goals for This Chapter 1. Identify the main structures and functions of the human excretory system 2. Explain the function of the nephron 3. Describe
BCH 450 Biochemistry of Specialized Tissues
 BCH 450 Biochemistry of Specialized Tissues VII. Renal Structure, Function & Regulation Kidney Function 1. Regulate Extracellular fluid (ECF) (plasma and interstitial fluid) through formation of urine.
BCH 450 Biochemistry of Specialized Tissues VII. Renal Structure, Function & Regulation Kidney Function 1. Regulate Extracellular fluid (ECF) (plasma and interstitial fluid) through formation of urine.
1. Urinary System, General
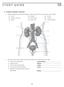 S T U D Y G U I D E 16 1. Urinary System, General a. Label the figure by placing the numbers of the structures in the spaces by the correct labels. 7 Aorta 6 Kidney 8 Ureter 2 Inferior vena cava 4 Renal
S T U D Y G U I D E 16 1. Urinary System, General a. Label the figure by placing the numbers of the structures in the spaces by the correct labels. 7 Aorta 6 Kidney 8 Ureter 2 Inferior vena cava 4 Renal
Renal Reabsorption & Secretion
 Renal Reabsorption & Secretion Topics for today: Nephron processing of filtrate Control of glomerular filtration Reabsorption and secretion Examples of solute clearance rates Hormones affecting kidney
Renal Reabsorption & Secretion Topics for today: Nephron processing of filtrate Control of glomerular filtration Reabsorption and secretion Examples of solute clearance rates Hormones affecting kidney
Introduction. Acids, Bases and ph; a review
 0 P a g e Introduction In this sheet, we discuss acidbase balance in our body and the role of kidneys in its establishment. Arrangement of topics is different from that of the lecture, to assure consistency
0 P a g e Introduction In this sheet, we discuss acidbase balance in our body and the role of kidneys in its establishment. Arrangement of topics is different from that of the lecture, to assure consistency
Metabolic Alkalosis: Vomiting
 RENAL ANL) ACID-BASE PHYSIOLOGY 213 Case 37 Metabolic Alkalosis: Vomiting Maria Cuervo is a 20-year-old philosophy major at a state university. When the "24-hour" stomach flu went around campus during
RENAL ANL) ACID-BASE PHYSIOLOGY 213 Case 37 Metabolic Alkalosis: Vomiting Maria Cuervo is a 20-year-old philosophy major at a state university. When the "24-hour" stomach flu went around campus during
The Urinary S. (Chp. 10) & Excretion. What are the functions of the urinary system? Maintenance of water-salt and acidbase
 10.1 Urinary system The Urinary S. (Chp. 10) & Excretion 10.1 Urinary system What are the functions of the urinary system? 1. Excretion of metabolic wastes (urea, uric acid & creatinine) 1. Maintenance
10.1 Urinary system The Urinary S. (Chp. 10) & Excretion 10.1 Urinary system What are the functions of the urinary system? 1. Excretion of metabolic wastes (urea, uric acid & creatinine) 1. Maintenance
ACID-BASE BALANCE URINE BLOOD AIR
 ACIDBASE BALANCE URINE BLOOD AIR H 2 PO 4 NH 4 HCO 3 KIDNEY H H HCO 3 CELLS Hb H LUNG H 2 CO 3 HHb CO 2 H 2 O ph = 7.4 [HCO 3 ] = 24 meq/l PCO 2 = 40 mm Hg CO 2 PRIMARY RENAL MECHANISMS INVOLVED IN ACIDBASE
ACIDBASE BALANCE URINE BLOOD AIR H 2 PO 4 NH 4 HCO 3 KIDNEY H H HCO 3 CELLS Hb H LUNG H 2 CO 3 HHb CO 2 H 2 O ph = 7.4 [HCO 3 ] = 24 meq/l PCO 2 = 40 mm Hg CO 2 PRIMARY RENAL MECHANISMS INVOLVED IN ACIDBASE
Physiology. Proper-es of The Body Fluid. Dr. Deniz Balcı.
 Physiology Proper-es of The Body Fluid Dr. Deniz Balcı deniz.balci@neu.edu.tr Outline 1 Volumes & Compartments of Body Fluids 2 Composi-on of Body Fluids 3 Fluid Movement between Compartments Reading Assignment
Physiology Proper-es of The Body Fluid Dr. Deniz Balcı deniz.balci@neu.edu.tr Outline 1 Volumes & Compartments of Body Fluids 2 Composi-on of Body Fluids 3 Fluid Movement between Compartments Reading Assignment
Fal Fal P h y s i o l o g y 6 1 1, S a n F r a n c i s c o S t a t e U n i v e r s i t y
 Fall 12 OSMOTIC REGULATION OF THE RENAL SYSTEM: Effects of fasting and ingestion of water, coke, or Gatorade on urine flow rate and specific gravity Dorette Franks The purpose of the physiology experiment
Fall 12 OSMOTIC REGULATION OF THE RENAL SYSTEM: Effects of fasting and ingestion of water, coke, or Gatorade on urine flow rate and specific gravity Dorette Franks The purpose of the physiology experiment
Technical University of Mombasa Faculty of Applied and Health Sciences
 Technical University of Mombasa Faculty of Applied and Health Sciences DEPARTMENT OF MEDICAL SCIENCES UNIVERSITY EXAMINATION FOR THE DEGREE OF BACHELOR OF MEDICAL LABORATORY SCIENCES BMLS 12S -Regular
Technical University of Mombasa Faculty of Applied and Health Sciences DEPARTMENT OF MEDICAL SCIENCES UNIVERSITY EXAMINATION FOR THE DEGREE OF BACHELOR OF MEDICAL LABORATORY SCIENCES BMLS 12S -Regular
Urinary System Organization. Urinary System Organization. The Kidneys. The Components of the Urinary System
 Urinary System Organization The Golden Rule: The Job of The Urinary System is to Maintain the Composition and Volume of ECF remember this & all else will fall in place! Functions of the Urinary System
Urinary System Organization The Golden Rule: The Job of The Urinary System is to Maintain the Composition and Volume of ECF remember this & all else will fall in place! Functions of the Urinary System
Ch 17 Physiology of the Kidneys
 Ch 17 Physiology of the Kidneys Review Anatomy on your own SLOs List and describe the 4 major functions of the kidneys. List and explain the 4 processes of the urinary system. Diagram the filtration barriers
Ch 17 Physiology of the Kidneys Review Anatomy on your own SLOs List and describe the 4 major functions of the kidneys. List and explain the 4 processes of the urinary system. Diagram the filtration barriers
