SAFETY GOGGLES MUST BE WORN AT ALL TIMES!
|
|
|
- Bertha Hall
- 6 years ago
- Views:
Transcription
1 SUGAR ME UP, DUDE! PURPOSE: To determine the density of five sugar reference solutions To determine the density of your two beverages, using the references as a guide To graph density versus percent sucrose for your sugar solutions, and understand various aspects of graphing To calculate percent error in order to determine your uncertainty in measurement BACKGROUND INFORMATION: Have you ever been to the ocean? Does it seem that you can float or swim much easier in the ocean than in a swimming pool? Seawater is denser than freshwater due to the presence of dissolved salt in the ocean. As a result, our buoyancy ability to float is greater in salt water than in plain water. Have you ever tried to take a regular soda and float it in water? How about a diet soda? What happens? What factors determine the density of a solution? Can the density of a solution be used to determine how much of a particular substance is dissolved in it? MATERIALS USED: Distilled Water Two Beverages of Choice Digital balance 140 ml Beaker 10 ml Pipette Pipetter Your Brain Sugar reference solutions 25 ml of each (0%, 5%, 10%, 15%, 20%) SAFETY GOGGLES MUST BE WORN AT ALL TIMES! PROCEDURE - PART A: 1. Make sure you have 6 sigma bottles at your station marked 0%, 5%, 10%, 15%, 20%, and distilled H 2 O rinse, that they are filled with sugar solutions and distilled H 2 O. 2. Place an empty 140 ml beaker in the balance and hit the tare button. The balance should read 0.00g, as if there is nothing on the balance at all. 3. Draw up ml of the 0% sugar solution (distilled water duh!) into a pipette and transfer the liquid to the empty beaker. 4. Record the mass of the solution in Data Table A. Remember what the balance is giving you is now just the mass of the solution, with out the beaker! 5. Dispose of the solution into the sink. Rinse the beaker and pipette with distilled water, and repeat steps 1-4 for each of your four sugar reference solutions. Make sure you rezero the balance before each new mass measurement! 6. Calculate the density of each solution and record in Data Table A. Include these calculations on a separate sheet of paper! Hint: Since the volume is always ml, the calculation should be easy you don t need a calculator!
2 PROCEDURE - PART B: 1. Use the procedure from Part A to determine the density of two beverages of your choice. Pour a small sample of your two beverages into two new sigma bottles. Rinse your beaker with distilled water, and record all mass and volume data in Data Table B. Make sure that you rinse your pipette and beaker each time, especially between the two beverages. DATA: DATA TABLE A: DENSITY OF REFERENCE SOLUTIONS Solution Mass (g) Volume (ml) Density (g/ml) 0% sugar 5% sugar 10% sugar 15% sugar 20% sugar DATA TABLE B: BEVERAGE DENSITIES Beverage Mass (g) Volume (ml) Density (g/ml) RESULTS DATA TABLE: BEVERAGE DENSITIES AND CALCULATED % SUGAR CONCENTRATION Beverage Density (g/ml) % Sugar Concentration GRAPHS/DATA TABLES: 1. Plot density versus concentration for the five reference solutions on a graph. The concentration is the independent variable, and the density is the dependent variable. Do you know where those go on a graph? Ask yourself what independent and dependent mean. After the points are graphed, you want to connect them with a line of best fit. This is a line that connects as many points as possible together because they share a relationship. You may graph with Logger Pro it has this feature, and will do it automatically. It is referred to as a linear fit. It is under the analyze option on the menu, and is also a separate button on the button bar.
3 2. Using the information provided to you by your graph, write an equation for the line on your graph. You will use this equation to solve for your unknown soda concentration. Use this equation to do the calculation in #3 below. ATTACH THE GRAPH TO YOUR LAB REPORT! CALCULATIONS: (use full units, and include all calculations on A SEPARATE SHEET OF PAPER!) 1. Show the calculations that you did above for the density of each sugar sample, and for your two soda samples. All calculations need to be included on a separate calculations page, with each section numbered and labeled. 2. Calculate the actual accepted value of the sugar concentration for both of your beverages, using the nutritional information and your measured density value. Record both the nutritional information and the actual percent sugar concentration on your calculations page and label them. 3. Calculate the unknown concentration of both of your beverages using the equation for your line, y = mx +b. Use this concentration to figure out the percent error below. 4. Use the following equation to calculate the percent error for sugar content for both of your beverages. Enter the percent errors for both beverages in your Results Data Table. % ERROR = MEASURED VALUE ACCEPTED VALUE X 100% ACCEPTED VALUE CONCLUSION QUESTIONS: 1. What was your measured density for pure water (0% sugar solution)? The density of water is quoted as 1.00 g/ml, but this precise value is for 4 0 C. Comment on why your measured density might be higher or lower than this. 2. This lab looks at the relationship between the density of a beverage and its sugar content. What assumption is made concerning the other ingredients in the beverage and their effect on the density of the solution? Do you think this a valid assumption? Explain.
4 3. a. When plotting data such as that obtained in this experiment, why is it not appropriate to connect the dots? b. What type of connection did we draw between the points plotted on the graph? c. Why do all of the points not fall on a straight line together? d. Comment on the sources of error in this experiment and their likely effect on the results. e. What is the benefit in being able to interpolate other unknown values from a line of best fit? 4. The main ingredient in soda, besides water, is obviously sugar. Look at your densities for each sugar solution, and each beverage. a. Are your beverages and solutions more or less dense than water? b. What would happen if you were to take a full can of regular soda and place it into a bucket of water? c. Hypothesize as to why a can of diet soda will float! 5. Let s say that an unknown beverage has a density of 1.03 g/ml. Calculate its % concentration using your equation from your graph below.
5 PRE-LAB QUESTIONS 1. If the following mass and volume data are used to calculate the density of a solution, how many significant figures are allowed in the calculated density? Mass of solution grams, volume of solution 8.27 ml. 2. Calculate the density of the solution described in Question #1. 3. According to its nutritional label, orange soda contains 49 grams of sugar per 355 ml serving. If the density of the beverage is /ml, what is the percent sugar concentration in orange soda? Hint this is a two-step problem! First, use the density to convert the 355 ml of soda to grams. Then, calculate the percent sugar in the soda.
Beverage Density Lab Sugar Content Analysis (Due Nov. 11)
 Beverage Density Lab Sugar Content Analysis (Due Nov. 11) Introduction: Nutritionists have recently raised concerns about the increasing popularity of sodas, fruit drinks and other beverages dur to their
Beverage Density Lab Sugar Content Analysis (Due Nov. 11) Introduction: Nutritionists have recently raised concerns about the increasing popularity of sodas, fruit drinks and other beverages dur to their
Beverage Density Lab
 Name: date: Beverage Density Lab Introduction: It is known that popular beverages often contain large amounts of. Nutritionists have raised concern in recent years as to the high levels of in these drinks
Name: date: Beverage Density Lab Introduction: It is known that popular beverages often contain large amounts of. Nutritionists have raised concern in recent years as to the high levels of in these drinks
Experiment 1: Scientific Measurements and Introduction to Excel
 Experiment 1: Scientific Measurements and Introduction to Excel Reading: Chapter 1 of your textbook and this lab handout. Learning Goals for Experiment 1: To use a scientific notebook as a primary record
Experiment 1: Scientific Measurements and Introduction to Excel Reading: Chapter 1 of your textbook and this lab handout. Learning Goals for Experiment 1: To use a scientific notebook as a primary record
Experimental Procedure
 1 of 7 9/12/2018, 2:19 PM https://www.sciencebuddies.org/science-fair-projects/project-ideas/humbio_p043/human-biology-health/iron-rich-foods (http://www.sciencebuddies.org/science-fair-projects /project-ideas/humbio_p043/human-biology-health/iron-rich-foods)
1 of 7 9/12/2018, 2:19 PM https://www.sciencebuddies.org/science-fair-projects/project-ideas/humbio_p043/human-biology-health/iron-rich-foods (http://www.sciencebuddies.org/science-fair-projects /project-ideas/humbio_p043/human-biology-health/iron-rich-foods)
MiSP Solubility Lab L3
 MiSP Solubility Lab L3 Name Date In today s lab you will be working in groups to determine whether sugar or salt dissolves more quickly in water. The rate at which different substances dissolve depends
MiSP Solubility Lab L3 Name Date In today s lab you will be working in groups to determine whether sugar or salt dissolves more quickly in water. The rate at which different substances dissolve depends
Lactose, Sucrose, and Glucose: How Many Sugars are in Your Smoothie?
 Lactose, Sucrose, and Glucose: How Many Sugars are in Your Smoothie? https://www.sciencebuddies.org/science-fair-projects/project-ideas/foodsci_p073/cooking-food-science/lactose-sucrose-and-glucose-how-many-sugars-arein-your-smoothie
Lactose, Sucrose, and Glucose: How Many Sugars are in Your Smoothie? https://www.sciencebuddies.org/science-fair-projects/project-ideas/foodsci_p073/cooking-food-science/lactose-sucrose-and-glucose-how-many-sugars-arein-your-smoothie
Titration of Synthesized Aspirin A continuation of the aspirin synthesis lab
 Titration of Synthesized Aspirin A continuation of the aspirin synthesis lab In this lab, you will determine the percent purity of your product from the aspirin synthesis using an acid-base titration.
Titration of Synthesized Aspirin A continuation of the aspirin synthesis lab In this lab, you will determine the percent purity of your product from the aspirin synthesis using an acid-base titration.
Coke Floats (Or Does It?)
 Date Lab Time Name Objective Coke Floats (Or Does It?) Scientific measurements will be introduced using common soft drinks as examples. Studentcollected data will be pooled leading to a discussion of accuracy
Date Lab Time Name Objective Coke Floats (Or Does It?) Scientific measurements will be introduced using common soft drinks as examples. Studentcollected data will be pooled leading to a discussion of accuracy
Name: Date: Solubility Lab - Worksheet #3 Level 1
 Name: Date: Solubility Lab - Worksheet #3 Level 1 In today s lab you will be working in groups to determine whether sugar or salt dissolves more quickly in water. The rate at which different substances
Name: Date: Solubility Lab - Worksheet #3 Level 1 In today s lab you will be working in groups to determine whether sugar or salt dissolves more quickly in water. The rate at which different substances
Osmosis. Evaluation copy
 Osmosis Computer 1B In order to survive, all organisms need to move molecules in and out of their cells. Molecules such as gases (e.g., O 2, CO 2 ), water, food, and wastes pass across the cell membrane.
Osmosis Computer 1B In order to survive, all organisms need to move molecules in and out of their cells. Molecules such as gases (e.g., O 2, CO 2 ), water, food, and wastes pass across the cell membrane.
Chemistry 151 Last Updated: Dec Lab 2: Measurements & Chemical Changes
 Chemistry 151 Last Updated: Dec. 2013 Lab 2: Measurements & Chemical Changes Introduction This lab will serve as an introduction to some of the basic techniques and skills you ll be using in this course
Chemistry 151 Last Updated: Dec. 2013 Lab 2: Measurements & Chemical Changes Introduction This lab will serve as an introduction to some of the basic techniques and skills you ll be using in this course
Diffusion and Osmosis Lab AP LAB 4
 Diffusion and Osmosis Lab AP LAB 4 Part 1: Surface Area and Cell Size Which do you think has a greater influence on the rate of diffusion in a cell surface area or volume? You will calculate surface are-to-volume
Diffusion and Osmosis Lab AP LAB 4 Part 1: Surface Area and Cell Size Which do you think has a greater influence on the rate of diffusion in a cell surface area or volume? You will calculate surface are-to-volume
A Penny for Your Thoughts: Scientific Measurements and Introduction to Excel
 A Penny for Your Thoughts: Scientific Measurements and Introduction to Excel Pre-lab Assignment: Introduction Reading: 1. Chapter sections 1.4 through 1.6 in your course text. 2. This lab handout. Questions:
A Penny for Your Thoughts: Scientific Measurements and Introduction to Excel Pre-lab Assignment: Introduction Reading: 1. Chapter sections 1.4 through 1.6 in your course text. 2. This lab handout. Questions:
Cell Membranes: Diffusion and Osmosis
 STO-112 Cell Membranes: Diffusion and Osmosis Part 1: Diffusion Diffusion is a process by which molecules move into or out of cells. To diffuse into or out of a cell, molecules must pass through the cell
STO-112 Cell Membranes: Diffusion and Osmosis Part 1: Diffusion Diffusion is a process by which molecules move into or out of cells. To diffuse into or out of a cell, molecules must pass through the cell
Experiment 1: Scientific Measurements and Introduction to Excel
 Experiment 1: Scientific Measurements and Introduction to Excel Reading: Chapter 1 of your textbook and this lab handout. Learning Goals for Experiment 1: To use a scientific notebook as a primary record
Experiment 1: Scientific Measurements and Introduction to Excel Reading: Chapter 1 of your textbook and this lab handout. Learning Goals for Experiment 1: To use a scientific notebook as a primary record
Egg-speriment With a Cell
 Name Date Class Cell Structure and Function Standards Investigation Overview S 7.1.a Standards Investigation Egg-speriment With a Cell One of the cell structures you will be learning about is the cell
Name Date Class Cell Structure and Function Standards Investigation Overview S 7.1.a Standards Investigation Egg-speriment With a Cell One of the cell structures you will be learning about is the cell
Lab: Acids and Bases
 Lab: Acids and Bases Many common household solutions contain acids and bases. Acid-base indicators, such as litmus and red cabbage juice, turn different colors in acidic and basic solutions. They can,
Lab: Acids and Bases Many common household solutions contain acids and bases. Acid-base indicators, such as litmus and red cabbage juice, turn different colors in acidic and basic solutions. They can,
LAB 04 Diffusion and Osmosis
 LAB 04 Diffusion and Osmosis Objectives: Describe the physical mechanisms of diffusion and osmosis. Understand the relationship between surface area and rate of diffusion. Describe how molar concentration
LAB 04 Diffusion and Osmosis Objectives: Describe the physical mechanisms of diffusion and osmosis. Understand the relationship between surface area and rate of diffusion. Describe how molar concentration
PROTEIN LAB BASED ON THE RESEARCH OF DR. RICHARD LONDRAVILLE
 PROTEIN LAB BASED ON THE RESEARCH OF DR. RICHARD LONDRAVILLE LAB OUTLINE- WEEK ONE PROCCESS WORMS FOR ANALYSIS SET UP A STANDARD CURVE WITH KNOWN CONCENTRATIONS PERFORM PROTEIN ASSAYS ON WORM SECTION INTERPRET
PROTEIN LAB BASED ON THE RESEARCH OF DR. RICHARD LONDRAVILLE LAB OUTLINE- WEEK ONE PROCCESS WORMS FOR ANALYSIS SET UP A STANDARD CURVE WITH KNOWN CONCENTRATIONS PERFORM PROTEIN ASSAYS ON WORM SECTION INTERPRET
Activity # 4. Measurement of Enzyme Activity
 Activity # 4. Measurement of Enzyme Activity Adapted from Experimental Molecular Biology Lab Manual, Stephen H. Munroe, Marquette University Week 1 Protein Extraction, Concentration Measurement & Instrument
Activity # 4. Measurement of Enzyme Activity Adapted from Experimental Molecular Biology Lab Manual, Stephen H. Munroe, Marquette University Week 1 Protein Extraction, Concentration Measurement & Instrument
Concentration of Sugar in Commercial Beverages
 Concentration of Sugar in Commercial Beverages Chemistry Period 3 Crater High School Rylee Jakabosky April 6th, 2016 Introduction According to an article by Everyday Health, 28.8% of young boys and 29.7%
Concentration of Sugar in Commercial Beverages Chemistry Period 3 Crater High School Rylee Jakabosky April 6th, 2016 Introduction According to an article by Everyday Health, 28.8% of young boys and 29.7%
SINKING SODA SURPRISE
 SINKING SODA SURPRISE Next: Materials and Explanations www.stevespanglerscience.com Then: Step-by-Step Photo Sequence SINKING SODA SURPRISE www.stevespanglerscience.com Which of your favorite sodas will
SINKING SODA SURPRISE Next: Materials and Explanations www.stevespanglerscience.com Then: Step-by-Step Photo Sequence SINKING SODA SURPRISE www.stevespanglerscience.com Which of your favorite sodas will
could be dissolved in 100 g of water at the given unsaturated, saturated, or supersaturated?
 Worksheet: Solubility Graphs Use the provided solubility graph to answer the following questions: For questions 1 4 an amount of solute is given, and a temperature is stated. If all of the solute could
Worksheet: Solubility Graphs Use the provided solubility graph to answer the following questions: For questions 1 4 an amount of solute is given, and a temperature is stated. If all of the solute could
Evaluation copy. Enzyme Action: Testing Catalase Activity. Computer
 Enzyme Action: Testing Catalase Activity Computer 6A Many organisms can decompose hydrogen peroxide (H 2 O 2 ) enzymatically. Enzymes are globular proteins, responsible for most of the chemical activities
Enzyme Action: Testing Catalase Activity Computer 6A Many organisms can decompose hydrogen peroxide (H 2 O 2 ) enzymatically. Enzymes are globular proteins, responsible for most of the chemical activities
Osmosis. Computer OBJECTIVES
 Osmosis Computer 22 In order to survive, all organisms need to move molecules in and out of their cells. Molecules such as gases (e.g., O 2, CO 2 ), water, food, and wastes pass across the cell membrane.
Osmosis Computer 22 In order to survive, all organisms need to move molecules in and out of their cells. Molecules such as gases (e.g., O 2, CO 2 ), water, food, and wastes pass across the cell membrane.
Enzyme Action: Testing Catalase Activity
 Enzyme Action: Testing Catalase Activity LabQuest 6A Many organisms can decompose hydrogen peroxide (H 2 O 2 ) enzymatically. Enzymes are globular proteins, responsible for most of the chemical activities
Enzyme Action: Testing Catalase Activity LabQuest 6A Many organisms can decompose hydrogen peroxide (H 2 O 2 ) enzymatically. Enzymes are globular proteins, responsible for most of the chemical activities
To understand osmosis, we must focus on the behavior of the solvent, not the solute.
 GCC CHM 130LL Osmosis and Dialysis Purpose: The purpose of this experiment is to observe the closely related phenomena of osmosis and diffusion as it relates to dialysis. It is hoped that you will be able
GCC CHM 130LL Osmosis and Dialysis Purpose: The purpose of this experiment is to observe the closely related phenomena of osmosis and diffusion as it relates to dialysis. It is hoped that you will be able
BIOLOGY 1101 LAB 1: OSMOSIS & DIFFUSION. READING: Please read pages & in your text prior to lab.
 BIOLOGY 1101 LAB 1: OSMOSIS & DIFFUSION READING: Please read pages 27-31 & 83-86 in your text prior to lab. INTRODUCTION: All living things depend on water. A water molecule is made up of an oxygen atom
BIOLOGY 1101 LAB 1: OSMOSIS & DIFFUSION READING: Please read pages 27-31 & 83-86 in your text prior to lab. INTRODUCTION: All living things depend on water. A water molecule is made up of an oxygen atom
Natural Selection Simulation: Predation and Coloration
 Name Period Date Natural Selection Simulation: Predation and Coloration This simulation was invented by G. Ledyard Stebbins, a pioneer in the evolution of plants. The purpose of the game is to illustrate
Name Period Date Natural Selection Simulation: Predation and Coloration This simulation was invented by G. Ledyard Stebbins, a pioneer in the evolution of plants. The purpose of the game is to illustrate
Diffusion and Osmosis
 Diffusion and Osmosis OBJECTIVES: 1. To explore how different molecules move by diffusion and osmosis through semi-permeable membranes. 2. To understand how concentration affects the movement of substances
Diffusion and Osmosis OBJECTIVES: 1. To explore how different molecules move by diffusion and osmosis through semi-permeable membranes. 2. To understand how concentration affects the movement of substances
Pre-Lab Exercises Lab 11: Drug Chemistry
 Pre-Lab Exercises Lab 11: Drug Chemistry Name Date Section 1. Describe how a non-steroidal anti-inflammatory drug (NSAID) reduces pain and inflammation. 2. The active ingredient in an aspirin tablet is
Pre-Lab Exercises Lab 11: Drug Chemistry Name Date Section 1. Describe how a non-steroidal anti-inflammatory drug (NSAID) reduces pain and inflammation. 2. The active ingredient in an aspirin tablet is
Diffusion and Osmosis
 Diffusion and Osmosis Introduction: In this exercise you will measure diffusion of small molecules through dialysis tubing, an example of a semi permeable membrane. The movement of a solute through a semi
Diffusion and Osmosis Introduction: In this exercise you will measure diffusion of small molecules through dialysis tubing, an example of a semi permeable membrane. The movement of a solute through a semi
Determining the Concentration of Iron in Vitamin Supplements
 Teacher Guide Determining the Concentration of Iron in Vitamin Supplements Background Information Colorimetry is the science of measuring color. Colorimetry is a useful technique for determining the concentration
Teacher Guide Determining the Concentration of Iron in Vitamin Supplements Background Information Colorimetry is the science of measuring color. Colorimetry is a useful technique for determining the concentration
Using Freezing-Point Depression to Find Molecular Weight. Evaluation copy
 Using Freezing-Point Depression to Find Molecular Weight Computer 4 When a solute is dissolved in a solvent, the freezing temperature is lowered in proportion to the number of moles of solute added. This
Using Freezing-Point Depression to Find Molecular Weight Computer 4 When a solute is dissolved in a solvent, the freezing temperature is lowered in proportion to the number of moles of solute added. This
David Huang! AP Biology! Oct. 4,2013! AP Biology Osmosis Laboratory Analysis! Introduction:!! There are several different methods for the
 David Huang AP Biology Oct. 4,2013 AP Biology Osmosis Laboratory Analysis Introduction: There are several different methods for the transportation of molecules across the phospholipid bilayer. These transportation
David Huang AP Biology Oct. 4,2013 AP Biology Osmosis Laboratory Analysis Introduction: There are several different methods for the transportation of molecules across the phospholipid bilayer. These transportation
Activity Sheet 1 Testing for Vitamin C- Part One
 Activity Sheet 1 Testing for - Part One Student Name: Purpose: To test the level of vitamin C in a variety of fruit juices. Procedure: Part 1 Testing Solution 1. Use the vitamin C solution for this experiment.
Activity Sheet 1 Testing for - Part One Student Name: Purpose: To test the level of vitamin C in a variety of fruit juices. Procedure: Part 1 Testing Solution 1. Use the vitamin C solution for this experiment.
Enzyme Action: Testing Catalase Activity
 Enzyme Action: Testing Catalase Activity Many organisms can decompose hydrogen peroxide (H 2 O 2 ) enzymatically. Enzymes are globular proteins, responsible for most of the chemical activities of living
Enzyme Action: Testing Catalase Activity Many organisms can decompose hydrogen peroxide (H 2 O 2 ) enzymatically. Enzymes are globular proteins, responsible for most of the chemical activities of living
AP Lab Four: Water Potential and Osmosis
 AP Biology AP Lab Four: Water Potential and Osmosis Name Atoms and molecules are constantly in motion, bumping off of membranes, barriers, each other, without end. The results of this among other phenomena
AP Biology AP Lab Four: Water Potential and Osmosis Name Atoms and molecules are constantly in motion, bumping off of membranes, barriers, each other, without end. The results of this among other phenomena
Sodium chloride Graduated cylinder Digital scale Flask/beaker Water Weighing boat Paper towels Directions. Task
 Saline solution is a mixture of sodium chloride, or salt, in water. Saline solution is normally 0.9% sodium chloride since 0.9% is also the normal concentration of saline within body fluids. An increase
Saline solution is a mixture of sodium chloride, or salt, in water. Saline solution is normally 0.9% sodium chloride since 0.9% is also the normal concentration of saline within body fluids. An increase
LAB Potato Cores Honors Biology, Newton North High
 Name Date Block LAB Potato Cores Honors Biology, Newton North High BACKGROUND: Osmosis is a type of passive transport. No input of energy is needed in order for water to pass through a selectively permeable
Name Date Block LAB Potato Cores Honors Biology, Newton North High BACKGROUND: Osmosis is a type of passive transport. No input of energy is needed in order for water to pass through a selectively permeable
Sugar Concentration in Sodas. Chemistry Period 3 Crater School of Business Innovation and Science Mallory Heard April 15th, 2016
 Sugar Concentration in Sodas Chemistry Period 3 Crater School of Business Innovation and Science Mallory Heard April 15th, 2016 Introduction Obesity is becoming a very big problem in America; 30% of the
Sugar Concentration in Sodas Chemistry Period 3 Crater School of Business Innovation and Science Mallory Heard April 15th, 2016 Introduction Obesity is becoming a very big problem in America; 30% of the
Biology Cell Unit Homework Packet #3
 Biology Cell Unit Homework Packet #3 Name DUE: Hour HW #5 Egg Demo Drawings Analysis HW #6 Elodea Drawings lab Analysis HW #7 Cell Questions Membrane and Transport HW #8 Questions / 5 possible points Homework
Biology Cell Unit Homework Packet #3 Name DUE: Hour HW #5 Egg Demo Drawings Analysis HW #6 Elodea Drawings lab Analysis HW #7 Cell Questions Membrane and Transport HW #8 Questions / 5 possible points Homework
Biology Cell Unit Homework Packet #3
 Biology Cell Unit Homework Packet #3 Name DUE: Hour HW #5 Egg Demo Drawings Analysis HW #6 Elodea Drawings lab Analysis HW #7 Cell Questions Membrane and Transport HW #8 Questions / 5 possible points Homework
Biology Cell Unit Homework Packet #3 Name DUE: Hour HW #5 Egg Demo Drawings Analysis HW #6 Elodea Drawings lab Analysis HW #7 Cell Questions Membrane and Transport HW #8 Questions / 5 possible points Homework
Experimental Procedure
 1 of 6 10/3/2018, 2:39 PM https://www.sciencebuddies.org/science-fair-projects/project-ideas/biomed_p012/medical-biotechnology/stopping-fungus-with-antifungal-medicines (http://www.sciencebuddies.org/science-fair-projects/project-ideas/biomed_p012/medical-biotechnology/stopping-fungus-with-antifungal-medicines)
1 of 6 10/3/2018, 2:39 PM https://www.sciencebuddies.org/science-fair-projects/project-ideas/biomed_p012/medical-biotechnology/stopping-fungus-with-antifungal-medicines (http://www.sciencebuddies.org/science-fair-projects/project-ideas/biomed_p012/medical-biotechnology/stopping-fungus-with-antifungal-medicines)
Organic Compounds in the Foods
 Organic Compounds in the Foods Purpose: This lab activity will help you understand the chemical composition (i.e., carbohydrates, proteins, and fats) of the foods that you eat. Materials we will be using:
Organic Compounds in the Foods Purpose: This lab activity will help you understand the chemical composition (i.e., carbohydrates, proteins, and fats) of the foods that you eat. Materials we will be using:
Lab Ch 6 Mole Buffet Lab Activity
 Lab Partners: Prelab must be complete before entering lab!!! (no prelab = you receive zero points lab) Show the calculations for molar mass for the following chemicals: (Show all work, units, and correct
Lab Partners: Prelab must be complete before entering lab!!! (no prelab = you receive zero points lab) Show the calculations for molar mass for the following chemicals: (Show all work, units, and correct
Catalytic Activity of Enzymes
 Catalytic Activity of Enzymes Introduction Enzymes are biological molecules that catalyze (speed up) chemical reactions. You could call enzymes the Builders and Do-ers in the cell; without them, life could
Catalytic Activity of Enzymes Introduction Enzymes are biological molecules that catalyze (speed up) chemical reactions. You could call enzymes the Builders and Do-ers in the cell; without them, life could
STATION 4: TONICITY due to OSMOSIS / Turgor Pressure in Plants
 STATION 4: TONICITY due to OSMOSIS / Turgor Pressure in Plants Tonicity is the concentration of solutions that determines the direction water will move across a semi-permeable membrane. A solution is a
STATION 4: TONICITY due to OSMOSIS / Turgor Pressure in Plants Tonicity is the concentration of solutions that determines the direction water will move across a semi-permeable membrane. A solution is a
EXPERIMENT 4 TITRATION OF AN UNKNOWN ACID
 EXPERIMENT 4 TITRATION OF AN UNKNOWN ACID The reaction of an acid and a base to form a salt and water is known as neutralization. In this experiment you will titrate an known amount of KHP with an unknown
EXPERIMENT 4 TITRATION OF AN UNKNOWN ACID The reaction of an acid and a base to form a salt and water is known as neutralization. In this experiment you will titrate an known amount of KHP with an unknown
Cell Processes: Osmosis
 www.mathbench.umd.edu./modules-au Osmosis May 2015 page 1 Cell Processes: Osmosis URL: http://mathbench.org.au/cellular-processes/osmosis/ Learning Outcomes After completing this module you should be able
www.mathbench.umd.edu./modules-au Osmosis May 2015 page 1 Cell Processes: Osmosis URL: http://mathbench.org.au/cellular-processes/osmosis/ Learning Outcomes After completing this module you should be able
How would you prepare 455 grams of an aqueous solution that is 6.50% sodium sulfate by mass?
 62 How would you prepare 455 grams of an aqueous solution that is 6.50% sodium sulfate by mass? Start a concentration calculation by writing the definition of the unit(s) you're using! We know everything
62 How would you prepare 455 grams of an aqueous solution that is 6.50% sodium sulfate by mass? Start a concentration calculation by writing the definition of the unit(s) you're using! We know everything
How much salt? Materials: Duckweed- you will need 5 plants for each container. Daphnia enough so that you can put 5-10 tweezers
 Name Date How much salt? Background: You are a resident of a town where the local stream has a concentration of sodium chloride of 50. You want to find out whether this amount of pollution affects the
Name Date How much salt? Background: You are a resident of a town where the local stream has a concentration of sodium chloride of 50. You want to find out whether this amount of pollution affects the
Determining the Molecular Mass of an Unknown Acid by Titration
 Determining the Molecular Mass of an Unknown Acid by Titration Objectives: To perform an analytical titration. To standardize a basic solution. To determine the equivalent mass of an unknown acid. Background:
Determining the Molecular Mass of an Unknown Acid by Titration Objectives: To perform an analytical titration. To standardize a basic solution. To determine the equivalent mass of an unknown acid. Background:
Distilled Water Balance Ruler Plastic wrap
 The following lab taken from: http://www.utsouthwestern.edu/edumedia/edufiles/education_training/programs/stars/osmosis-demo-lab.pdf Background Osmosis is the process whereby water moves across a cell
The following lab taken from: http://www.utsouthwestern.edu/edumedia/edufiles/education_training/programs/stars/osmosis-demo-lab.pdf Background Osmosis is the process whereby water moves across a cell
ENZYME ACTION: TESTING CATALASE ACTIVITY
 Name Date Period ENZYME ACTION: TESTING CATALASE ACTIVITY Many organisms can decompose hydrogen peroxide (H 2 O 2 ) enzymatically. Enzymes are globular proteins, responsible for most of the chemical activities
Name Date Period ENZYME ACTION: TESTING CATALASE ACTIVITY Many organisms can decompose hydrogen peroxide (H 2 O 2 ) enzymatically. Enzymes are globular proteins, responsible for most of the chemical activities
Organic Molecule Composition of Milk: Lab Investigation
 Name: Organic Molecule Composition of Milk: Lab Investigation Introduction & Background Milk & milk products have been a major food source from earliest recorded history. Milk is a natural, nutritionally
Name: Organic Molecule Composition of Milk: Lab Investigation Introduction & Background Milk & milk products have been a major food source from earliest recorded history. Milk is a natural, nutritionally
Lab #3 Potentiometric Titration of Soda Ash (after Christian, p , p ) (phenolphthalein)
 Lab #3 Potentiometric Titration of Soda Ash (after Christian, p.692-694, p.718-720) I: INTRODUCTION In this lab, an unknown sample of soda ash (a crude mixture of sodium carbonate) will be titrated with
Lab #3 Potentiometric Titration of Soda Ash (after Christian, p.692-694, p.718-720) I: INTRODUCTION In this lab, an unknown sample of soda ash (a crude mixture of sodium carbonate) will be titrated with
Name: Bio A.P. Lab Diffusion & Osmosis
 Name: Bio A.P. Lab Diffusion & Osmosis BACKGROUND: Many aspects of the life of a cell depend on the fact that atoms and molecules are constantly in motion (kinetic energy). This kinetic energy results
Name: Bio A.P. Lab Diffusion & Osmosis BACKGROUND: Many aspects of the life of a cell depend on the fact that atoms and molecules are constantly in motion (kinetic energy). This kinetic energy results
TRACKS Lesson Plan. Choosing healthy beverages Rethink Your Drink Grade 5 8 Boys Club
 TRACKS Lesson Plan Choosing healthy beverages Rethink Your Drink Grade 5 8 Boys Club I. Nutrition Education Goal & Objective: Goal 1: Students will comprehend concepts consistent with USDA guidance related
TRACKS Lesson Plan Choosing healthy beverages Rethink Your Drink Grade 5 8 Boys Club I. Nutrition Education Goal & Objective: Goal 1: Students will comprehend concepts consistent with USDA guidance related
Activity 5.2 Using chemical change to identify an unknown
 Activity 5.2 Using chemical change to identify an unknown How can you identify an unknown powder? In this activity, students will develop a method to test five similar-looking powders with four test liquids.
Activity 5.2 Using chemical change to identify an unknown How can you identify an unknown powder? In this activity, students will develop a method to test five similar-looking powders with four test liquids.
TOXICOLOGY PROTOCOLS PROTOCOL 1. SERIAL DILUTIONS. Objective To make a serial dilution for use in dose/response bioassays.
 TOXICOLOGY PROTOCOLS PROTOCOL 1. SERIAL DILUTIONS Objective To make a serial dilution for use in dose/response bioassays. Background The idea behind a bioassay is that the test organism will respond in
TOXICOLOGY PROTOCOLS PROTOCOL 1. SERIAL DILUTIONS Objective To make a serial dilution for use in dose/response bioassays. Background The idea behind a bioassay is that the test organism will respond in
EXPERIMENT. Titration of the Weak Acid Potassium Hydrogen Phthalate (KHP)
 INTRODUCTION EXPERIMENT Titration of the Weak Acid Potassium Hydrogen Phthalate (KHP) Materials generally considered to possess acidic and/or basic properties are widely distributed in nature and range
INTRODUCTION EXPERIMENT Titration of the Weak Acid Potassium Hydrogen Phthalate (KHP) Materials generally considered to possess acidic and/or basic properties are widely distributed in nature and range
BIO 322/122L Laboratory Plant Water Relations
 BIO 322/122L Laboratory Plant Water Relations I. Water Potential. The cytoplasm of the plant cell, with its enclosed vacuole, is contained within a membrane that is more permeable to water than to most
BIO 322/122L Laboratory Plant Water Relations I. Water Potential. The cytoplasm of the plant cell, with its enclosed vacuole, is contained within a membrane that is more permeable to water than to most
High School Lesson Plan
 High School Lesson Plan Choosing Healthy Beverages Rethink Your Drink Grades 9-12 I. Lesson Objectives: A. Students will explain the importance of water and hydration. B. Students will identify healthy
High School Lesson Plan Choosing Healthy Beverages Rethink Your Drink Grades 9-12 I. Lesson Objectives: A. Students will explain the importance of water and hydration. B. Students will identify healthy
BIO 12 UNIT 04: The Cell Membrane BCLN Rev. July, 2015
 Project 1: Osmosis Lab Name: Potential Credits: /50 Project Goals: to gain and demonstrate a better understanding of osmosis Instructions Please read through the Unit 4 Lessons, paying particular attention
Project 1: Osmosis Lab Name: Potential Credits: /50 Project Goals: to gain and demonstrate a better understanding of osmosis Instructions Please read through the Unit 4 Lessons, paying particular attention
Cell Diffusion & Permeability: See-Through Eggs Student Advanced Version
 Cell Diffusion & Permeability: See-Through Eggs Student Advanced Version In this lab, students will learn about the permeability of the cell membrane. By studying the ability of a shell-less egg to absorb
Cell Diffusion & Permeability: See-Through Eggs Student Advanced Version In this lab, students will learn about the permeability of the cell membrane. By studying the ability of a shell-less egg to absorb
Name: There are two things that will determine which particles will pass through and which will not:
 18 Diffusion and Osmosis in Living Systems Name: Problem: How do substances move into and out of cells? Introduction: In order for cells to carry on their life processes, they must take in materials and
18 Diffusion and Osmosis in Living Systems Name: Problem: How do substances move into and out of cells? Introduction: In order for cells to carry on their life processes, they must take in materials and
3 To gain experience monitoring a titration with a ph electrode and determining the equivalence point.
 Titrations PURPOSE To determine the concentration of acetic acid in vinegar. GOALS 1 To perform an acid-base titration. 2 To gain experience titrating carefully to a visible endpoint. 3 To gain experience
Titrations PURPOSE To determine the concentration of acetic acid in vinegar. GOALS 1 To perform an acid-base titration. 2 To gain experience titrating carefully to a visible endpoint. 3 To gain experience
DIFFUSON AND OSMOSIS INTRODUCTION diffusion concentration gradient. net osmosis water potential active transport
 DIFFUSON AND OSMOSIS NAME DATE INTRODUCTION The life of a cell is dependent on efficiently moving material into and out of the cell across the cell membrane. Raw materials such as oxygen and sugars needed
DIFFUSON AND OSMOSIS NAME DATE INTRODUCTION The life of a cell is dependent on efficiently moving material into and out of the cell across the cell membrane. Raw materials such as oxygen and sugars needed
Enzyme Analysis using Tyrosinase. Evaluation copy
 Enzyme Analysis using Tyrosinase Computer 15 Enzymes are molecules that regulate the chemical reactions that occur in all living organisms. Almost all enzymes are globular proteins that act as catalysts,
Enzyme Analysis using Tyrosinase Computer 15 Enzymes are molecules that regulate the chemical reactions that occur in all living organisms. Almost all enzymes are globular proteins that act as catalysts,
USING STATCRUNCH TO CONSTRUCT CONFIDENCE INTERVALS and CALCULATE SAMPLE SIZE
 USING STATCRUNCH TO CONSTRUCT CONFIDENCE INTERVALS and CALCULATE SAMPLE SIZE Using StatCrunch for confidence intervals (CI s) is super easy. As you can see in the assignments, I cover 9.2 before 9.1 because
USING STATCRUNCH TO CONSTRUCT CONFIDENCE INTERVALS and CALCULATE SAMPLE SIZE Using StatCrunch for confidence intervals (CI s) is super easy. As you can see in the assignments, I cover 9.2 before 9.1 because
Activity 4.2 Dissolving a substance in different liquids
 Activity 4.2 Does colored sugar dissolve equally well in water, vegetable oil, and alcohol? In the introductory story on Activity sheet 4.1, the student added drink mix to different liquids. Many drink
Activity 4.2 Does colored sugar dissolve equally well in water, vegetable oil, and alcohol? In the introductory story on Activity sheet 4.1, the student added drink mix to different liquids. Many drink
2. is a set of principles intended to help sustain a habitable planet.
 Chapter 2 Tools of the Trade 1 Multiple Choice 2-1 Safe, Ethical Handling of Chemicals and Waste 1. Which of the following statements are TRUE? I Organic solvents, concentrated acids, and concentrated
Chapter 2 Tools of the Trade 1 Multiple Choice 2-1 Safe, Ethical Handling of Chemicals and Waste 1. Which of the following statements are TRUE? I Organic solvents, concentrated acids, and concentrated
Enzyme Action: Testing Catalase Activity
 Enzyme Action: Testing Catalase Activity Pennsylvania Science Standards: S11.A.1.1.4 S11.A.1.3.1 S11.A.2.2.2.1 S11.A.2.2.2.2 Keystone Eligible Content Bio.B.4.1.1, Bio.B.4.1.2, and Bio.B.4.2.5 Introduction
Enzyme Action: Testing Catalase Activity Pennsylvania Science Standards: S11.A.1.1.4 S11.A.1.3.1 S11.A.2.2.2.1 S11.A.2.2.2.2 Keystone Eligible Content Bio.B.4.1.1, Bio.B.4.1.2, and Bio.B.4.2.5 Introduction
Evaluation copy 17B. Enzyme Action: Testing Catalase Activity. Computer
 Enzyme Action: Testing Catalase Activity Computer 17B Many organisms can decompose hydrogen peroxide (H 2 O 2 ) enzymatically. Enzymes are globular proteins, responsible for most of the chemical activities
Enzyme Action: Testing Catalase Activity Computer 17B Many organisms can decompose hydrogen peroxide (H 2 O 2 ) enzymatically. Enzymes are globular proteins, responsible for most of the chemical activities
Station 1: Med School Acceptance
 Station 1: Med School Acceptance The competition for students applying to medical school is tough, but how tough is it? Most medical school and allied health programs have a set number of positions available
Station 1: Med School Acceptance The competition for students applying to medical school is tough, but how tough is it? Most medical school and allied health programs have a set number of positions available
Lab 7: Heat and Calorimetry
 Concepts to explore: Gain applicable knowledge about calories Compare the calorie content of food samples Introduction Have you ever roasted marshmallows over a campfire? You have probably heard that foods
Concepts to explore: Gain applicable knowledge about calories Compare the calorie content of food samples Introduction Have you ever roasted marshmallows over a campfire? You have probably heard that foods
MOVEMENT PREPARATION LAB. Name: Score: Activity I: Predictability of the correct response choice & Influence of Pre cueing
 MOVEMENT PREPARATION LAB Name: Score: Activity I: Predictability of the correct response choice & Influence of Pre cueing Introduction: Research evidence has consistently shown that as the predictability
MOVEMENT PREPARATION LAB Name: Score: Activity I: Predictability of the correct response choice & Influence of Pre cueing Introduction: Research evidence has consistently shown that as the predictability
Inspirational chemistry 97. Index sheets. Rhubarb contains oxalic acid, which has the formula C H 3. O + 2 Mn CO 2
 Inspirational chemistry 97 Rates and rhubarb Index 4.1.3 3 sheets Rhubarb contains oxalic acid, which has the formula C 2 H 2 O 4 : O OH C C HO Figure 1 Structural formula of oxalic acid Oxalic acid reacts
Inspirational chemistry 97 Rates and rhubarb Index 4.1.3 3 sheets Rhubarb contains oxalic acid, which has the formula C 2 H 2 O 4 : O OH C C HO Figure 1 Structural formula of oxalic acid Oxalic acid reacts
AP Biology Lab 1c Water Potential
 Page 1 of 9 AP Biology Lab 1c Water Potential In this part of the exercise you will use potato cores placed in different molar concentrations of sucrose in order to determine the water potential of potato
Page 1 of 9 AP Biology Lab 1c Water Potential In this part of the exercise you will use potato cores placed in different molar concentrations of sucrose in order to determine the water potential of potato
Mass Measurements. Did you know that we use weights and measures every day? Chapter 1: Weights & Measures. Weights & Measures
 Chapter 1: Mass Measurements Did you know that we use weights and measures every day? People use weights and measures all the time. Whether you are stepping onto the scale at the doctor s office, measuring
Chapter 1: Mass Measurements Did you know that we use weights and measures every day? People use weights and measures all the time. Whether you are stepping onto the scale at the doctor s office, measuring
Scientific Inquiry Review
 Scientific Inquiry Review Adapted from Regentsprep.org Be able to USE AND APPLY the steps of the scientific method to solve a problem and design an experiment: Scientific Method: 1. Make observations about
Scientific Inquiry Review Adapted from Regentsprep.org Be able to USE AND APPLY the steps of the scientific method to solve a problem and design an experiment: Scientific Method: 1. Make observations about
Introductory Laboratory 0: Buffon's Needle
 Introductory Laboratory 0: Buffon's Needle Introduction: In geometry, the circumference of a circle divided by its diameter is a fundamental constant, denoted by the symbol ππ. Curiously, this number has
Introductory Laboratory 0: Buffon's Needle Introduction: In geometry, the circumference of a circle divided by its diameter is a fundamental constant, denoted by the symbol ππ. Curiously, this number has
Rate of Decomposition of Hydrogen Peroxide as a Function of Catalase. Concentration
 Rate of Decomposition of Hydrogen Peroxide as a Function of Catalase Concentration Performed: Herrin High School September 11, 2017 Mr. Johns INTRODUCTION Purpose: To determine how the concentration of
Rate of Decomposition of Hydrogen Peroxide as a Function of Catalase Concentration Performed: Herrin High School September 11, 2017 Mr. Johns INTRODUCTION Purpose: To determine how the concentration of
1SCIENTIFIC METHOD PART A. THE SCIENTIFIC METHOD
 1SCIENTIFIC METHOD LEARNING OUTCOMES Upon successful completion of this lab, you will be able to: Describe the steps of the scientific method Formulate research questions, hypotheses, and predictions Design
1SCIENTIFIC METHOD LEARNING OUTCOMES Upon successful completion of this lab, you will be able to: Describe the steps of the scientific method Formulate research questions, hypotheses, and predictions Design
Observing Osmosis Lab
 Observing Osmosis Lab Background Information: Molecules are in constant motion, and tend to move from areas of higher concentrations to areas of lower concentrations. Diffusion is defined as the movement
Observing Osmosis Lab Background Information: Molecules are in constant motion, and tend to move from areas of higher concentrations to areas of lower concentrations. Diffusion is defined as the movement
Name Date. In this lab investigation you will investigate the movement of water through a selectively permeable membrane.
 This lab will be hand-written in your data book AP Osmosis Labs Part A (was done in previous a previous class: Dialysis tube + Starch + Glucose) Part B: Osmosis Unknowns In this lab investigation you will
This lab will be hand-written in your data book AP Osmosis Labs Part A (was done in previous a previous class: Dialysis tube + Starch + Glucose) Part B: Osmosis Unknowns In this lab investigation you will
Am I Getting Anywhere? Physics of Blood Flow-Velocity. Student Data Page, Activity 3C
 Am I Getting Anywhere? Physics of Blood Flow-Velocity Student Data Page, Activity 3C Getting Started: 1. Go to the section of the model assigned to you to watch for leaks. 2. Turn on the pump. 3. Allow
Am I Getting Anywhere? Physics of Blood Flow-Velocity Student Data Page, Activity 3C Getting Started: 1. Go to the section of the model assigned to you to watch for leaks. 2. Turn on the pump. 3. Allow
Name: Date Block Selective Permeability
 LAB Name: Date Block Selective Permeability OBJECTIVES: Observe the selective permeability of an artificial membrane. Observe diffusion of substances across an artificial membrane. Devise a model for the
LAB Name: Date Block Selective Permeability OBJECTIVES: Observe the selective permeability of an artificial membrane. Observe diffusion of substances across an artificial membrane. Devise a model for the
Pre-lab Homework Lab 6: Photosynthesis & Cellular Respiration
 Lab Section: Name: Pre-lab Homework After reading over the lab and the topics of photosynthesis and cellular respiration from your textbook, answer these questions to be turned in at the beginning of the
Lab Section: Name: Pre-lab Homework After reading over the lab and the topics of photosynthesis and cellular respiration from your textbook, answer these questions to be turned in at the beginning of the
Carbohydrates. Objectives. Background. Experiment 6
 1 of 6 3/15/2011 7:27 PM Experiment 6 Carbohydrates Objectives During this experiment you will look at some of the physical and chemical properties of carbohydrates. Many of the carbohydrates, especially
1 of 6 3/15/2011 7:27 PM Experiment 6 Carbohydrates Objectives During this experiment you will look at some of the physical and chemical properties of carbohydrates. Many of the carbohydrates, especially
1. All cells have a that acts as a between the outside and inside of the cell.
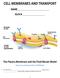 https://www.youtube.com/watch?v=cnbzdcibegy 1. All cells have a that acts as a between the outside and inside of the cell. 1 2. Cell membranes are primarily made of which are large molecules. 3. It is
https://www.youtube.com/watch?v=cnbzdcibegy 1. All cells have a that acts as a between the outside and inside of the cell. 1 2. Cell membranes are primarily made of which are large molecules. 3. It is
Mass Measurements. Did you know that we use weights and measures every day? Chapter 1: Weights & Measures. Weights & Measures
 Chapter 1: Mass Measurements Did you know that we use weights and measures every day? People use weights and measures all the time. Whether you are stepping onto the scale at the doctor s office, measuring
Chapter 1: Mass Measurements Did you know that we use weights and measures every day? People use weights and measures all the time. Whether you are stepping onto the scale at the doctor s office, measuring
Egg-speriment (Osmosis Lab) 2009
 Purpose/Objectives: Osmosis can have important consequences for the cell. The purpose of this lab is to study the effects of osmosis on a cell that is submerged in different aqueous environments: vinegar,
Purpose/Objectives: Osmosis can have important consequences for the cell. The purpose of this lab is to study the effects of osmosis on a cell that is submerged in different aqueous environments: vinegar,
Testing the capacity of biological buffer systems to resist changes in ph By P. H. Meter
 Testing the capacity of biological buffer systems to resist changes in ph By P. H. Meter Personal Engagement and Exploration Research Question: How do plant and animal tissues respond to changes in ph?
Testing the capacity of biological buffer systems to resist changes in ph By P. H. Meter Personal Engagement and Exploration Research Question: How do plant and animal tissues respond to changes in ph?
Toxicity testing. Introduction
 Toxicity testing Lab. Objective: To familiarize the student with the concepts and techniques of bioassay utilized in toxicity testing. To introduce students to the calculation of LC 5 values. Toxicity
Toxicity testing Lab. Objective: To familiarize the student with the concepts and techniques of bioassay utilized in toxicity testing. To introduce students to the calculation of LC 5 values. Toxicity
Bouncing Ball Lab. Name
 Bouncing Ball Lab Name Scientists use an organized set of steps when they solve problems or perform investigations. This organized set of steps is called the Scientific Method. There are many versions
Bouncing Ball Lab Name Scientists use an organized set of steps when they solve problems or perform investigations. This organized set of steps is called the Scientific Method. There are many versions
GN , CCNE: How Sweet is Your Drink?
 GN-000-28, CCNE: How Sweet is Your Drink? Client-centered nutrition education uses methods like group discussions and hands-on activities to engage participants in learning. This outline starts with a
GN-000-28, CCNE: How Sweet is Your Drink? Client-centered nutrition education uses methods like group discussions and hands-on activities to engage participants in learning. This outline starts with a
