4 PARENTERAL THERAPY
|
|
|
- Silvester York
- 6 years ago
- Views:
Transcription
1 WOMEN AND NEWBORN HEALTH SERVICE CLINICAL GUIDELINES SECTION A: GUIDELINES RELEVANT TO OBSTETRICS AND GYNAECOLOGY 4 PARENTERAL THERAPY 4.11 TOTAL PARENTERAL NUTRITION Authorised by: OGCCU Review Team: OGCCU /Pharmacy / Dietetics 4.11 TOTAL PARENTERAL NUTRITION AIMS To provide nutritional support via the intravenous route, when oral or enteral routes are inadequate, inaccessible or non functional 1. To minimise the potential catheter related, metabolic and infectious complications associated with total parenteral nutrition (TPN). BACKGROUND Total Parenteral Nutrition (TPN) is the formulation of nutritional components for intravenous delivery. Included are carbohydrates, amino acids, fats including essential fatty acids, electrolytes, vitamins, minerals, trace elements, water and other additives. TPN is indicated when there is an inability to provide adequate nutrition via the oral / enteral route and is administered continuously until enteral feeding can be re-established. TPN is continuously infused via a controlled infusion pump into a high flow vein, usually the superior vena cava adjacent to the right atrium. KEY POINTS 1. Observe: Hand hygiene before and after any manipulation of vascular access devices or catheter sites 1, 2 (Level I) See Infection Control Manual, Policy 2.4, Hand Hygiene. An aseptic technique. Adhere strictly to the principles of asepsis at all times to prevent contamination of the solution, line and central venous access device.tpn solutions are an ideal growth medium for bacteria and fungi. Standard precautions. See Infection Control Manual, Policy 2.1, Standard Precautions 2 The standard TPN base solution available is SmofKabiven. Alternatively a TPN solution can be made specifically for a patient to meet each unique clinical requirement. This is coordinated daily by the multi disciplinary team comprising dietetic, pharmacy, medical and nursing staff to improve overall quality of care 1, Electrolyte, phosphate and magnesium imbalances must be corrected prior to commencement of TPN to provide optimal care and to prevent refeeding syndrome. 4. Blood glucose levels must be assessed prior to therapy and monitored according to this guideline throughout therapy. DPMS Ref: 7491 All guidelines should be read in conjunction with the Disclaimer at the beginning of this manual Page 1 of 6
2 5. Administration of hypertonic TPN solutions (concentrations of Dextrose greater than 10% / Protein 5%) require central venous catheter access with confirmed catheter tip placement in the superior vena cava 2, 4, 5 This allows rapid dilution of the solution. Peripherally inserted central catheter, non tunnelled central venous catheter (CVC), tunnelled CVC s (e.g. Hickman/ Brovac/ Groshong catheters) or implanted ports are all suitable. 6. Administration of TPN should be via a dedicated CVC or, if a multiple lumen catheter is in situ, a designated lumen identified for this use 5. This catheter/lumen should not be used for blood sampling as per MR741. Although undesirable, where no other access is available, bolus medications may be given. Consult pharmacy for compatibility and administration advice. Use the proximal port and flush pre and post bolus 6. TPN solution and the line shall be discarded at or before the expiry date and time (< 24 hours). 7. TPN solution is incompatible with blood transfusions. Alternative intravenous access or a proximal port of a multi lumen catheter 7 should be used. 8. Monitor: Blood glucose levels (BGL) every 6 hours during rate changes. Once stable, monitor once daily. Base line weight 8 then twice weekly. Vital signs Fluid balance Catheter insertion site for sepsis, migration, and to ensure an intact dressing. For signs of infection, fluid and electrolyte imbalance and nutritional status while receiving TPN. Commencement and cessation of TPN infusions should be planned and at a controlled rate in liaison with the RMO. Administration of TPN shall be via a volumetric device. PARENTERAL NUTRITION FLUID Prescribed daily (Parenteral Nutrition Order MR 741) after review of biochemistry results. Pharmacy requires this prescription prior to 1100 hours to enable the solution to be manufactured under laminar flow conditions 5. Infusion rates are titrated on commencing and discontinuation of therapy to prevent fluctuation in blood glucose levels 8 - as per MR741. A volumetric pump is required 5, 9. TPN solution: o May include some drugs, for example insulin. o Protect by a light sensitive cover. o Non standard bags must be stored in the refrigerator and removed 60 minutes prior to infusing to bring to ambient temperature 5. 5, 10 o Should be completed within 24 hours of commencement DPMS Ref: 7491 All guidelines should be read in conjunction with the Disclaimer at the beginning of this manual Page 2 of 5
3 Ceasing TPN abruptly may result in rebound hypoglycaemia 7. Dextrose 10% is suggested for use if TPN is interrupted or unavailable 8. COMPLICATIONS Catheter related: Includes site infection and sepsis. Observe site and report pyrexia and altered BSL. Insertion related pneumothorax, air embolism, thrombosis Metabolic alterations: Biochemical monitoring is required to prevent or treat the following: o Hypo/hyperglycaemia o Hyponatraemia o Hypo/hypercalcaemia o Hypomagnesaemia o Hypophosphataemia PROCEDURE Ensure daily morning blood samples are collected for the monitoring of biochemistry while the patient is receiving TPN. Ensure the biochemistry forms are marked priority TPN. The RMO shall review the daily biochemistry results and adjust the TPN prescription accordingly in collaboration with the ward Pharmacist. Ensure the central venous access is present and patent for TPN administration. ADMINISTRATION The usual commencement rate is 20mL per hour, although this may vary according to the patient s clinical condition. The rate increases by 20mL per hour every 1-2 hours up to a maximum rate of 80mL per hour. The total volume of TPN mixture is infused at an hourly rate over a 24 hour period. Two midwives / nurses check that the prescription form is signed by the prescriber. TPN must be checked at the bedside by two nurses/ midwives and the patient must be identified by three indicators (e.g. name, DOB. Address or URMN) Check the patient s identification band against the patient label on the Parenteral Nutrition request form (MR 741) and TPN bag. Check the TPN solution including o Total volume o Administration rate. o Expiry date and time. Inspect the TPN bag, line and filter to ensure all is intact and free of particulate matter. If the TPN bag is not intact or particulate matter is present do not connect the TPN to the patient. DPMS Ref: 7491 All guidelines should be read in conjunction with the Disclaimer at the beginning of this manual Page 3 of 5
4 For lipid containing TPN ensure there are no visible fat globules Document the time the infusion was commenced and completed on the Parenteral Nutrition request form (MR 741). Both midwives / nurses must sign the MR 741. Retain the blue plastic bag to cover the fluid bag for the duration of the infusion. TPN CONNECTION PROCEDURE Equipment Non sterile gloves Sodium chloride 0.9% 10mL flush 10mL syringe 1% chlorhexidine and 70% isopropyl alcohol swabs x 3 Volumetric infusion pump TPN solution with attached, primed infusion line and 1.2 micron filter Blue plastic bag. PROCEDURE TPN should be removed from the refrigerator approximately 30 minutes prior to connection to allow the solution to come to room temperature. Perform hand hygiene. Visually inspect the infusion for signs of precipitation, discolouration, leakage, separation or contamination. ( Contact the pharmacist if there are any concerns regarding the above). Observe the CVC site, prior to the connection, for signs of infection and extravasation. Perform hand hygiene. Don non sterile gloves. Observe a strict aseptic technique. Prepare and cleanse the selected port with the 2% chlorhexidine and 70% isopropyl alcohol swab 11,12 and allow to air dry. Flush the selected vascular access port with sodium chloride 0.9% 10mL. Using a non touch technique connect the TPN line to the dedicated central venous port and secure the Luer-Lok connection. With the SmofKabiven bag this is the blue infusion port with the arrow flag. On the non standard bag it is the middle port. Remove gloves and discard appropriately. Perform hand hygiene. Check the prescribed rate on the Parenteral Nutrition request form (MR 741). Programme the volumetric infusion pump to the prescribed rate and commence the infusion. The initial TPN rate is usually 20mL per hour, increasing by approximately 20mL / hour every 1-2 hours up to a maximum rate of 80mL per hour. Monitor the blood glucose every 6 hours during rate changes. Once stable, monitor once daily. DPMS Ref: 7491 All guidelines should be read in conjunction with the Disclaimer at the beginning of this manual Page 4 of 5
5 Be aware of the daily target fluid input of each patient. Consider the patient s total daily fluid intake (including IV, enteral and oral) in the 24 hours prior to starting the TPN. Measure and monitor the total daily fluid output. Maintain an accurate fluid balance chart. Liaise with the RMO for the monitoring of electrolytes and management of results. Document the TPN volumes infused on the fluid balance chart or ASCU chart (MR731). Record the commencement of the TPN in the patient s Integrated Progress Notes. MONITORING THE PATIENT WHEN ESTABLISHING TPN. Monitor the patient s heart rate, blood pressure and temperature every 4 hours or more frequently as determined by the patient s clinical status for the duration of the infusion. Do not perform a BP every 4 hours on patients with low platelets or bleeding disorders unless clinically indicated. Weigh the patient prior to commencing the TPN, then weekly at a regular time. Regular weights assist in the assessment of adequate nutrition and daily fluid balance. Perform BGLs 6 hourly on initiation of TPN. Inform the RMO if the BGL is outside normal range. Monitor the biochemistry daily and PRN. CEASING TPN. Follow the rate reduction protocol as prescribed by the RMO. Each change must be checked, dated and the time recorded and signed by two midwives / nurses. Document the rate change on the fluid balance chart and integrated progress notes. Once the TPN is ceased and the line disconnected, flush the port with 10mL 0.9% sodium chloride. Ports may then be used for other infusions. When disconnecting the line, always use a posiflow or clamp the lumen to prevent the risk of air embolus. REFERENCES 1. National Institute for Health and Clinical Excellence. Nutrition support in adults. Clinical Guideline 32. London: National Health Service; 2006 February. 2. Intravenous Nurses Society Parenteral Nutrition. Journal of Infusion Nursing 2006; 29((1)): S Centre for Disease Control. Guidelines for the Prevention of Intravascular Catheter-Related Infections. Morbidity and Mortality Weekly Report 2002; 51(RR10). 4. Scottish Intercollegiate Guidelines Network. Postoperative management in adults. Postoperative nutrition, Edinburgh: National Health Service Scotland August. 5. Royal College of Nursing. Infusion therapies In: Standards for Infusion Therapy, 2005; 2nd edition: p47. DPMS Ref: 7491 All guidelines should be read in conjunction with the Disclaimer at the beginning of this manual Page 5 of 5
6 6. Louey Melissa. Clinical Information Service. Intervention: Total parenteral Nutrition, Joanna Briggs Institute; th September. 7. Morrisey Nancy A. Gastrointestinal Intubation and Special Nutritional Modalities, In: Smeltzer Suzanne C. BBG. Editor. Brunner & Suddarth's Textbook of Medical-Surgical Nursing, Philadelphia: Lippincott Williams & Wilkins; p. Chapter 36 p Hagle Mary GD, Kassulke., Laak K., Yale B,. Patient-Specific Therapies. Parenteral Nutrition, In: Weinstein Sharon M, editor. Plumer's Principles & Practice of Intravenous Therapy, Eighth ed. Philadelphia: Lippincott Williams & Wilkins; p. Ch 16. P Gianino Stephanie, Seltzer Rene, Eisenberg Patti. The ABCs of TPN. RN 1996; 59(2): The Joanna Briggs Institute, Intervention: Central Venous Implantable Ports - Needling, 10th August, 11. Ghosh D, Neild P. Parenteral nutrition. Clinical Medicine December 1, 2010;10(6): Pittiruti M, Hamilton H, Biffi R, et al. ESPEN Guidelines on Parenteral Nutrition: Central Venous Catheters (access, care,diagnosis and therapy of complications). Clinical nutrition (Edinburgh, Scotland). 2009;28(4): ACKNOWLEDGEMENT Sir Charles Gairdner Hospital. Clinical Practice Guideline No 7: Total Parenteral Nutrition.2013 DPMS Ref: 7491 All guidelines should be read in conjunction with the Disclaimer at the beginning of this manual Page 6 of 5
IV Fluids Nursing B23 Objectives Serum Osmolality 275 to 295 Isotonic
 1 IV Fluids Nursing B23 2 Objectives 3 Serum Osmolality Serum osmolality solute concentration of a solution Higher osmolality means greater pulling power for water Normal serum osmolality is 275 to 295
1 IV Fluids Nursing B23 2 Objectives 3 Serum Osmolality Serum osmolality solute concentration of a solution Higher osmolality means greater pulling power for water Normal serum osmolality is 275 to 295
IV Fluids. Nursing B23. Objectives. Serum Osmolality
 IV Fluids Nursing B23 Objectives Discuss the purpose of IV Discuss nursing interventions in IV therapy Identify complications of IV therapy Differentiate between peripheral line, central line, and PICC
IV Fluids Nursing B23 Objectives Discuss the purpose of IV Discuss nursing interventions in IV therapy Identify complications of IV therapy Differentiate between peripheral line, central line, and PICC
Central Line Care and Management
 Central Line Care and Management What is a Central Line/ CVAD? (central venous access device) A vascular infusion device that terminates at or close to the heart or in one of the great vessels (aorta,
Central Line Care and Management What is a Central Line/ CVAD? (central venous access device) A vascular infusion device that terminates at or close to the heart or in one of the great vessels (aorta,
CENTRAL VENOUS ACCESS DEVICES. BETHANY COLTON
 CENTRAL VENOUS ACCESS DEVICES. BETHANY COLTON Aims and Objectives To know what central venous access devices (CVAD) are. Types of CVADS used in haematology. To understand why we use them To know the complications
CENTRAL VENOUS ACCESS DEVICES. BETHANY COLTON Aims and Objectives To know what central venous access devices (CVAD) are. Types of CVADS used in haematology. To understand why we use them To know the complications
Neonatal Parenteral Nutrition Guideline Dr M Hogan, Maire Cullen ANNP, Una Toland Ward Manager, Sandra Kilpatrick Neonatal Pharmacist
 CLINICAL GUIDELINES ID TAG Title: Author: Designation: Speciality / Division: Directorate: Neonatal Parenteral Nutrition Guideline Dr M Hogan, Maire Cullen ANNP, Una Toland Ward Manager, Sandra Kilpatrick
CLINICAL GUIDELINES ID TAG Title: Author: Designation: Speciality / Division: Directorate: Neonatal Parenteral Nutrition Guideline Dr M Hogan, Maire Cullen ANNP, Una Toland Ward Manager, Sandra Kilpatrick
BPG 03: Continuous Renal Replacement Therapy (CRRT)
 BPG 03: Continuous Renal Replacement Therapy (CRRT) Statement of Best Practice Patient s requiring Continuous Renal Replacement Therapy (CRRT) will receive appropriate therapy to meet their individual
BPG 03: Continuous Renal Replacement Therapy (CRRT) Statement of Best Practice Patient s requiring Continuous Renal Replacement Therapy (CRRT) will receive appropriate therapy to meet their individual
SARASOTA MEMORIAL HOSPITAL
 SARASOTA MEMORIAL HOSPITAL NURSING PROCEDURE TITLE: ISSUED FOR: PARENTERAL NUTRITION AND INTRAVENOUS FAT EMULSION (ADULT AND PEDIATRICS) Nursing DATE: REVIEWED: PAGES: RESPONSIBILITY: RN, LPN II 12/80
SARASOTA MEMORIAL HOSPITAL NURSING PROCEDURE TITLE: ISSUED FOR: PARENTERAL NUTRITION AND INTRAVENOUS FAT EMULSION (ADULT AND PEDIATRICS) Nursing DATE: REVIEWED: PAGES: RESPONSIBILITY: RN, LPN II 12/80
Chapter 8 ADMINISTRATION OF BLOOD COMPONENTS
 Chapter 8 ADMINISTRATION OF BLOOD COMPONENTS PRACTICE POINTS Give the right blood product to the right patient at the right time. Failure to correctly check the patient or the pack can be fatal. At the
Chapter 8 ADMINISTRATION OF BLOOD COMPONENTS PRACTICE POINTS Give the right blood product to the right patient at the right time. Failure to correctly check the patient or the pack can be fatal. At the
If viewing a printed copy of this policy, please note it could be expired. Got to to view current policies.
 If viewing a printed copy of this policy, please note it could be expired. Got to www.fairview.org/fhipolicies to view current policies. Department Policy Code: D: PC-5575 Entity: Fairview Pharmacy Services
If viewing a printed copy of this policy, please note it could be expired. Got to www.fairview.org/fhipolicies to view current policies. Department Policy Code: D: PC-5575 Entity: Fairview Pharmacy Services
Jo Kuehn, RN, MSN, CPHQ Jenell Westhoven, RN, BSN
 Jo Kuehn, RN, MSN, CPHQ Jenell Westhoven, RN, BSN Disclosure Information Intravenous Therapy and Parenteral Nutrition Administration: Nursing In- Focus Jo Kuehn, RN, MSN, CPHQ and Jenell Westhoven, RN,
Jo Kuehn, RN, MSN, CPHQ Jenell Westhoven, RN, BSN Disclosure Information Intravenous Therapy and Parenteral Nutrition Administration: Nursing In- Focus Jo Kuehn, RN, MSN, CPHQ and Jenell Westhoven, RN,
PRESCRIBING INFORMATION. Dextrose Injection USP. (Concentrated Dextrose for Intravenous Administration) 50% (500 mg/ml) Fluid and Nutrient Replenisher
 PRESCRIBING INFORMATION Dextrose Injection USP (Concentrated Dextrose for Intravenous Administration) 50% (500 mg/ml) Fluid and Nutrient Replenisher Pfizer Canada Inc. 17300 Trans-Canada Highway Kirkland,
PRESCRIBING INFORMATION Dextrose Injection USP (Concentrated Dextrose for Intravenous Administration) 50% (500 mg/ml) Fluid and Nutrient Replenisher Pfizer Canada Inc. 17300 Trans-Canada Highway Kirkland,
ATI Skills Modules Checklist for Central Venous Access Devices
 For faculty use only Educator s name Score Date ATI Skills Modules Checklist for Central Venous Access Devices Student s name Date Verify order Patient record Assess for procedure need Identify, gather,
For faculty use only Educator s name Score Date ATI Skills Modules Checklist for Central Venous Access Devices Student s name Date Verify order Patient record Assess for procedure need Identify, gather,
Overview of CVADs. Type of device commonly used. Dwell time Flushing requirement Associated complications. lumens
 Source: Clinical Skills Management of Vascular Access Devices Pre-course handbook. Adapted with permission from NHS Lothian Employee and Education Development Team. Overview of CVADs Type of device Veins
Source: Clinical Skills Management of Vascular Access Devices Pre-course handbook. Adapted with permission from NHS Lothian Employee and Education Development Team. Overview of CVADs Type of device Veins
IV therapy. By: Susan Mberenga, RN, MSN. Copyright 2016, 2013, 2010, 2006, 2002 by Saunders, an imprint of Elsevier Inc.
 IV therapy By: Susan Mberenga, RN, MSN 1 IV Therapy Types of solutions Isotonic Hypotonic Hypertonic Caution: Too rapid or excessive infusion of any IV fluid has the potential to cause serious problems
IV therapy By: Susan Mberenga, RN, MSN 1 IV Therapy Types of solutions Isotonic Hypotonic Hypertonic Caution: Too rapid or excessive infusion of any IV fluid has the potential to cause serious problems
IR Central Venous Access [ ] Pre Procedure
![IR Central Venous Access [ ] Pre Procedure IR Central Venous Access [ ] Pre Procedure](/thumbs/90/104119758.jpg) IR Central Venous Access [1050200001] Pre Procedure Case Request/Scheduling Procedure Enter IR Case Request if not already completed (All hospitals except Grant Medical Center) [ ] Case Request IR Lab
IR Central Venous Access [1050200001] Pre Procedure Case Request/Scheduling Procedure Enter IR Case Request if not already completed (All hospitals except Grant Medical Center) [ ] Case Request IR Lab
If viewing a printed copy of this policy, please note it could be expired. Got to to view current policies.
 If viewing a printed copy of this policy, please note it could be expired. Got to www.fairview.org/fhipolicies to view current policies. Department Policy Code: D: PC-5555 Entity: Fairview Pharmacy Services
If viewing a printed copy of this policy, please note it could be expired. Got to www.fairview.org/fhipolicies to view current policies. Department Policy Code: D: PC-5555 Entity: Fairview Pharmacy Services
Emergency clamp should always be readily available in case of accidental catheter fracture
 Note: Please see individual policies for further information. Flushing best practice: Always use a 10 diameter syringe or larger when first accessing and when flushing vascular access device (VAD) Use
Note: Please see individual policies for further information. Flushing best practice: Always use a 10 diameter syringe or larger when first accessing and when flushing vascular access device (VAD) Use
NOTE: The first appearance of terms in bold in the body of this document (except titles) are defined terms please refer to the Definitions section.
 TITLE PARENTERAL NUTRITION ADMINISTRATION AND MONITORING SCOPE Provincial: Acute Care APPROVAL AUTHORITY Clinical Operations Executive Committee SPONSOR Pharmacy Services, Nutrition Services, and Health
TITLE PARENTERAL NUTRITION ADMINISTRATION AND MONITORING SCOPE Provincial: Acute Care APPROVAL AUTHORITY Clinical Operations Executive Committee SPONSOR Pharmacy Services, Nutrition Services, and Health
Curraheen, Co. Cork. Guidelines on the Management and Care of Central Venous Access Devices
 Curraheen, Co. Cork. Guidelines on the Management and Care of Central Venous Access Devices Date re-approved: 27 th Jan 2015. Version No: 2 Revision Due: 2018 Index code: CLIN028 Disclaimer: The information
Curraheen, Co. Cork. Guidelines on the Management and Care of Central Venous Access Devices Date re-approved: 27 th Jan 2015. Version No: 2 Revision Due: 2018 Index code: CLIN028 Disclaimer: The information
Unit 11. Objectives. Indications for IV Therapy. Intravenous Access Devices & Common IV Fluids. 3 categories. Maintenance Replacement Restoration
 Unit 11 Fluids, Electrolytes and Acid Base Imbalances Intravenous Access Devices & Common IV Fluids Objectives Review the purpose and types of intravenous (IV) therapy. Recall the nursing care related
Unit 11 Fluids, Electrolytes and Acid Base Imbalances Intravenous Access Devices & Common IV Fluids Objectives Review the purpose and types of intravenous (IV) therapy. Recall the nursing care related
PARENTERAL NUTRITION
 PARENTERAL NUTRITION DEFINITION Parenteral nutrition [(PN) or total parenteral nutrition (TPN)] is the intravenous infusion of some or all nutrients for tissue maintenance, metabolic requirements and growth
PARENTERAL NUTRITION DEFINITION Parenteral nutrition [(PN) or total parenteral nutrition (TPN)] is the intravenous infusion of some or all nutrients for tissue maintenance, metabolic requirements and growth
PATIENT CARE PLAN FOR CARE OF PERIPHERAL MIDLINE. Manufacturers specific recommendations should be noted and adhered to by individual practitioners.
 PATIENT CARE PLAN FOR CARE OF PERIPHERAL MIDLINE The care plan is designed to be used in conjunction with CINS Guidelines for vascular devices. Manufacturers specific recommendations should be noted and
PATIENT CARE PLAN FOR CARE OF PERIPHERAL MIDLINE The care plan is designed to be used in conjunction with CINS Guidelines for vascular devices. Manufacturers specific recommendations should be noted and
TRANSFUSION OF BLOOD COMPONENTS ADMINISTRATION. All blood components are administered according to BOP DHB Policy and NZBS Guidelines.
 STANDARDS All blood components are administered according to BOP DHB Policy and NZBS Guidelines. EQUIPMENT IV administration set with 260 micron filter either integrated blood filter; or add on blood filter
STANDARDS All blood components are administered according to BOP DHB Policy and NZBS Guidelines. EQUIPMENT IV administration set with 260 micron filter either integrated blood filter; or add on blood filter
Chapter 64 Administration of Injectable Medications
 Chapter 64 Administration of Injectable Medications Injectable Medications Injection or intravenous (IV) infusion More invasive than administration by mouth, rectum, or through the skin Common methods
Chapter 64 Administration of Injectable Medications Injectable Medications Injection or intravenous (IV) infusion More invasive than administration by mouth, rectum, or through the skin Common methods
Mary Lou Garey MSN EMT-P MedFlight of Ohio
 Mary Lou Garey MSN EMT-P MedFlight of Ohio Function Prolonged and frequent access to venous circulation Allows for patient to carry on normal life; decrease number of needle sticks Medications, parenteral
Mary Lou Garey MSN EMT-P MedFlight of Ohio Function Prolonged and frequent access to venous circulation Allows for patient to carry on normal life; decrease number of needle sticks Medications, parenteral
SARASOTA MEMORIAL HOSPITAL NURSING PROCEDURE
 SARASOTA MEMORIAL HOSPITAL NURSING PROCEDURE TITLE: ISSUED FOR: INTERMITTENT FLUID AND MEDICATION THERAPY Nursing DATE: REVIEWED: PAGES: RESPONSIBILITY: RN, LPN I, LPN II Per Job Description 03/81 8/09
SARASOTA MEMORIAL HOSPITAL NURSING PROCEDURE TITLE: ISSUED FOR: INTERMITTENT FLUID AND MEDICATION THERAPY Nursing DATE: REVIEWED: PAGES: RESPONSIBILITY: RN, LPN I, LPN II Per Job Description 03/81 8/09
10/28/2015. Disclaimer. Disclaimer
 Disclaimer Omnicare, Inc., as a provider of Infusion Pharmacy Services, is committed to the establishment and maintenance of the highest quality of care in infusion therapy services. This Infusion Therapy
Disclaimer Omnicare, Inc., as a provider of Infusion Pharmacy Services, is committed to the establishment and maintenance of the highest quality of care in infusion therapy services. This Infusion Therapy
MANITOBA RENAL PROGRAM
 MANITOBA RENAL PROGRAM SUBJECT Use of Closed Needleless Access Device with Hemodialysis Central Venous Catheters (CVC) SECTION CODE 30.20.04 30.20 Vascular Access AUTHORIZATION Professional Advisory Committee,
MANITOBA RENAL PROGRAM SUBJECT Use of Closed Needleless Access Device with Hemodialysis Central Venous Catheters (CVC) SECTION CODE 30.20.04 30.20 Vascular Access AUTHORIZATION Professional Advisory Committee,
X: Infusion Therapy. Saskatchewan Association of Licensed Practical Nurses, Competency Profile for LPNs, 3rd Ed. 201
 X: Infusion Therapy Saskatchewan Association of Licensed Practical Nurses, Competency Profile for LPNs, 3rd Ed. 201 Competency: X-1 Principles of X-1-1 X-1-2 X-1-3 X-1-4 X-1-5 Demonstrate knowledge and
X: Infusion Therapy Saskatchewan Association of Licensed Practical Nurses, Competency Profile for LPNs, 3rd Ed. 201 Competency: X-1 Principles of X-1-1 X-1-2 X-1-3 X-1-4 X-1-5 Demonstrate knowledge and
Department Policy. Code: D:PC Entity: Fairview Pharmacy Services. Department: Fairview Home Infusion. Manual: Policy and Procedure Manual
 Entity: Fairview Pharmacy Services Department: Fairview Home Infusion Manual: Policy and Procedure Manual Category: Home Infusion Subject: Midline (Extended Dwell Peripheral) Catheter Care and Management
Entity: Fairview Pharmacy Services Department: Fairview Home Infusion Manual: Policy and Procedure Manual Category: Home Infusion Subject: Midline (Extended Dwell Peripheral) Catheter Care and Management
Education for Self Administration of Intravenous Therapy HOME IV THERAPY PICC. Portacath
 HOME IV THERAPY PICC Portacath Who To contact Cardio-Respiratory Integrated Specialist Services (CRISS) Office hours 0800 1630 hours Ph: 364 0167 Weekends and after hours, phone Christchurch Hospital operator
HOME IV THERAPY PICC Portacath Who To contact Cardio-Respiratory Integrated Specialist Services (CRISS) Office hours 0800 1630 hours Ph: 364 0167 Weekends and after hours, phone Christchurch Hospital operator
Methods of Nutrition Support KNH 406
 Methods of Nutrition Support KNH 406 Malnutrition 30 50% of hospitalized patients 95% of nursing home patients Resulting in reduced quality of life & increased health care costs May be remedied by providing
Methods of Nutrition Support KNH 406 Malnutrition 30 50% of hospitalized patients 95% of nursing home patients Resulting in reduced quality of life & increased health care costs May be remedied by providing
Central Venous Access Devices. Stephanie Cunningham Amy Waters
 Central Venous Access Devices Stephanie Cunningham Amy Waters 5 Must Know Facts About CVAD s 1) What are CVAD s? 2) What are CVAD s used for? 3) How are these devices put in? 4) What are the complications
Central Venous Access Devices Stephanie Cunningham Amy Waters 5 Must Know Facts About CVAD s 1) What are CVAD s? 2) What are CVAD s used for? 3) How are these devices put in? 4) What are the complications
SUMMARY OF PRODUCT CHARACTERISTICS. Synthamin 14, 8.5% Amino Acid Intravenous Infusion
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Synthamin 14, 8.5% Amino Acid Intravenous Infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION L-Leucine Ph. Eur 0.620% w/v L-Isoleucine
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Synthamin 14, 8.5% Amino Acid Intravenous Infusion 2. QUALITATIVE AND QUANTITATIVE COMPOSITION L-Leucine Ph. Eur 0.620% w/v L-Isoleucine
IDPN. A Piece of the Puzzle for Dialysis Patients Nutrition. Toll Free: Toll Free:
 IDPN A Piece of the Puzzle for Dialysis Patients Nutrition Toll Free: 844-633-3448 Toll Free: 844-633-3448 Executive Infusion is pleased to announce our nutritional services which include Intradialytic
IDPN A Piece of the Puzzle for Dialysis Patients Nutrition Toll Free: 844-633-3448 Toll Free: 844-633-3448 Executive Infusion is pleased to announce our nutritional services which include Intradialytic
IV Drug Delivery Systems used in Cancer Care
 IV Drug Delivery Systems used in Cancer Care Cheri Constantino-Shor, RN, MSN, CRNI Seattle Cancer Care Alliance Nursing Staff Development Coordinator Presentation Objective Describe drug delivery devices
IV Drug Delivery Systems used in Cancer Care Cheri Constantino-Shor, RN, MSN, CRNI Seattle Cancer Care Alliance Nursing Staff Development Coordinator Presentation Objective Describe drug delivery devices
TOTAL PARENTERAL NUTRITION
 TOTAL PARENTERAL NUTRITION Indication See algorithm. Timing Start TPN as indicated on algorithm 1. There is no need to build up TPN volume. The volume of TPN (including lipids) should equate to the total
TOTAL PARENTERAL NUTRITION Indication See algorithm. Timing Start TPN as indicated on algorithm 1. There is no need to build up TPN volume. The volume of TPN (including lipids) should equate to the total
PACKAGE LEAFLET: INFORMATION FOR THE USER. Glucose Intravenous Infusion BP 10% w/v solution for infusion Glucose (as glucose monohydrate)
 PACKAGE LEAFLET: INFORMATION FOR THE USER Glucose Intravenous Infusion BP 10% w/v solution for infusion Glucose (as glucose monohydrate) Read all of this leaflet carefully before you start using this medicine
PACKAGE LEAFLET: INFORMATION FOR THE USER Glucose Intravenous Infusion BP 10% w/v solution for infusion Glucose (as glucose monohydrate) Read all of this leaflet carefully before you start using this medicine
Vascular access device selection & placement. Alisa Seangleulur, MD Anesthesiology Department, Faculty of Medicine, Thammasat University
 Vascular access device selection & placement Alisa Seangleulur, MD Anesthesiology Department, Faculty of Medicine, Thammasat University How to make the right choice of vascular access device.. Peripheral
Vascular access device selection & placement Alisa Seangleulur, MD Anesthesiology Department, Faculty of Medicine, Thammasat University How to make the right choice of vascular access device.. Peripheral
NOTTINGHAM CHILDREN S HOSPITAL Nursing Guideline. P07 Care and Management of Children with a Totally Implantable Venous Access Device (Portacath).
 NOTTINGHAM CHILDREN S HOSPITAL Nursing Guideline P07 Care and Management of Children with a Totally Implantable Venous Access Device (Portacath). Introduction A Portacath is a central venous access device
NOTTINGHAM CHILDREN S HOSPITAL Nursing Guideline P07 Care and Management of Children with a Totally Implantable Venous Access Device (Portacath). Introduction A Portacath is a central venous access device
Appendix E: Overview of Vascular
 Appendix E: Overview of Vascular 56 Peripheral Short Catheter, less than 3 inches (7.5 cm) in length; over-the-needle catheter is most common. Inserted by percutaneous venipuncture, generally into a hand
Appendix E: Overview of Vascular 56 Peripheral Short Catheter, less than 3 inches (7.5 cm) in length; over-the-needle catheter is most common. Inserted by percutaneous venipuncture, generally into a hand
PHARMACOLOGY AND PHARMACOKINETICS
 DRUG GUIDELINE Insulin, human neutral (Actrapid ) Intravenous Infusion for SCOPE (Area): FOR USE IN: Critical Care Unit, Emergency Department and Operating Suite EXCLUSIONS: Paediatrics (seek Paediatrician
DRUG GUIDELINE Insulin, human neutral (Actrapid ) Intravenous Infusion for SCOPE (Area): FOR USE IN: Critical Care Unit, Emergency Department and Operating Suite EXCLUSIONS: Paediatrics (seek Paediatrician
Guideline scope Neonatal parenteral nutrition
 NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Guideline scope Neonatal parenteral nutrition The Department of Health in England has asked NICE to develop a new guideline on parenteral nutrition in
NATIONAL INSTITUTE FOR HEALTH AND CARE EXCELLENCE Guideline scope Neonatal parenteral nutrition The Department of Health in England has asked NICE to develop a new guideline on parenteral nutrition in
Central Venous Catheter Insertion: Assisting
 Approved by: Central Venous Catheter Insertion: Assisting Gail Cameron Senior Director, Operations, Maternal, Neonatal & Child Health Programs Dr. Santiago Ensenat Medical Director, Neonatology Neonatal
Approved by: Central Venous Catheter Insertion: Assisting Gail Cameron Senior Director, Operations, Maternal, Neonatal & Child Health Programs Dr. Santiago Ensenat Medical Director, Neonatology Neonatal
Parenteral Nutrition in the Hospitalized Patient: which VAD, which policies? Rob Dawson DNP, MSA, APRN, ACNP-BC, VA-BC USA
 Parenteral Nutrition in the Hospitalized Patient: which VAD, which policies? Rob Dawson DNP, MSA, APRN, ACNP-BC, VA-BC USA Disclosure Consultant, Vascular Access Consultants LLC: Analogic, Inc / BK Ultrasound
Parenteral Nutrition in the Hospitalized Patient: which VAD, which policies? Rob Dawson DNP, MSA, APRN, ACNP-BC, VA-BC USA Disclosure Consultant, Vascular Access Consultants LLC: Analogic, Inc / BK Ultrasound
DRUG GUIDELINE. HYDRALAZINE (Intravenous severe hypertension in pregnancy)
 DRUG GUIDELINE HYDRALAZINE (Intravenous severe hypertension SCOPE (Area): FOR USE IN: Labour Ward, HDU, Theatre and ED EXCLUSIONS: Paediatrics (seek Paediatrician advice) and other general wards. SCOPE
DRUG GUIDELINE HYDRALAZINE (Intravenous severe hypertension SCOPE (Area): FOR USE IN: Labour Ward, HDU, Theatre and ED EXCLUSIONS: Paediatrics (seek Paediatrician advice) and other general wards. SCOPE
Peripheral Intravenous Devices
 SH CP 140 Peripheral Intravenous Devices Procedures for Peripheral intravenous cannula and Midline intravenous catheters Summary: Keywords (minimum of 5): (To assist policy search engine) Target Audience:
SH CP 140 Peripheral Intravenous Devices Procedures for Peripheral intravenous cannula and Midline intravenous catheters Summary: Keywords (minimum of 5): (To assist policy search engine) Target Audience:
Successful IV Starts Revised February 2014
 Successful IV Starts Revised February 2014 Why Intravenous Therapy? Used for access to the body s circulation Indications: Administer fluids, blood, medications, and nutrition Obtain laboratory specimens
Successful IV Starts Revised February 2014 Why Intravenous Therapy? Used for access to the body s circulation Indications: Administer fluids, blood, medications, and nutrition Obtain laboratory specimens
TYPE 1 DIABETES MELLITIS CARE OF WOMEN IN BIRTHING SUITE
 TYPE 1 DIABETES MELLITIS CARE OF WOMEN IN BIRTHING SUITE DEFINITION Type 1 Diabetes: described as a total lack of insulin produced by the pancreas for the requirements of the tissues. If left untreated,
TYPE 1 DIABETES MELLITIS CARE OF WOMEN IN BIRTHING SUITE DEFINITION Type 1 Diabetes: described as a total lack of insulin produced by the pancreas for the requirements of the tissues. If left untreated,
Home Total Parenteral Nutrition
 PATIENT & CAREGIVER EDUCATION Home Total Parenteral Nutrition This information will help teach you and your caregiver how to give total parenteral nutrition (TPN) at home. You will also receive a copy
PATIENT & CAREGIVER EDUCATION Home Total Parenteral Nutrition This information will help teach you and your caregiver how to give total parenteral nutrition (TPN) at home. You will also receive a copy
PROCEDURE FOR THE CARE AND MAINTENANCE OF CENTRAL INTRAVENOUS ACCESS DEVICES
 PROCEDURE FOR THE CARE AND MAINTENANCE OF CENTRAL INTRAVENOUS ACCESS DEVICES First Issued Issue Version Purpose of Issue/Description of Change Planned Review Date One To ensure a safe and effective procedure
PROCEDURE FOR THE CARE AND MAINTENANCE OF CENTRAL INTRAVENOUS ACCESS DEVICES First Issued Issue Version Purpose of Issue/Description of Change Planned Review Date One To ensure a safe and effective procedure
Guidelines for the Care and Maintenance of Intravenous Access Devices in Paediatric Patients
 Guidelines for the Care and Maintenance of Intravenous Access Devices in Paediatric Patients These guidelines have been adapted for local use from the Collaborative Intravenous Nursing Services regional
Guidelines for the Care and Maintenance of Intravenous Access Devices in Paediatric Patients These guidelines have been adapted for local use from the Collaborative Intravenous Nursing Services regional
Network Policy for the Care and Management of Central Venous Access Devices (CVAD) in Adults.
 Network Policy for the Care and Management of Central Venous Access Devices (CVAD) in Adults. Quality and safety for every patient every time Document Control Prepared By Sharon Harper Wendy Anderson Issue
Network Policy for the Care and Management of Central Venous Access Devices (CVAD) in Adults. Quality and safety for every patient every time Document Control Prepared By Sharon Harper Wendy Anderson Issue
Policy for the safe handling and administration of cytotoxic drugs for Children, Teenagers and Young Adults with Cancer
 Policy for the safe handling and administration of cytotoxic drugs for Children, Teenagers and Young Adults with Cancer Introduction Cytotoxic drugs are used to treat cancer and a number of other disorders.
Policy for the safe handling and administration of cytotoxic drugs for Children, Teenagers and Young Adults with Cancer Introduction Cytotoxic drugs are used to treat cancer and a number of other disorders.
Children s Acute Transport Service
 Children s Acute Transport Service Vascular Access Document Control Information Author Ramnarayan Author Position Consultant, CATS Document Owner Polke Document Owner Position CATS Co-ordinator Document
Children s Acute Transport Service Vascular Access Document Control Information Author Ramnarayan Author Position Consultant, CATS Document Owner Polke Document Owner Position CATS Co-ordinator Document
MANAGEMENT OF INTRAVASCULAR (IV) LINES AND THERAPY. All GCC Countries
 TITLE/DESCRIPTION: MANAGEMENT OF INTRAVASCULAR (IV) LINES AND THERAPY INDEX NUMBER: EFFECTIVE DATE: APPLIES TO: ISSUING AUTHORITY: 01/01/2009 01/01/2013 All GCC Countries GULF COOPERATION COUNCIL CENTRE
TITLE/DESCRIPTION: MANAGEMENT OF INTRAVASCULAR (IV) LINES AND THERAPY INDEX NUMBER: EFFECTIVE DATE: APPLIES TO: ISSUING AUTHORITY: 01/01/2009 01/01/2013 All GCC Countries GULF COOPERATION COUNCIL CENTRE
Pharmacy Instructions for Preparation
 MARQIBO (vincristine sulfate LIPOSOME injection) Pharmacy Instructions for Preparation Important Information for Preparation 1 The instructions for the constitution of MARQIBO are provided in each kit.
MARQIBO (vincristine sulfate LIPOSOME injection) Pharmacy Instructions for Preparation Important Information for Preparation 1 The instructions for the constitution of MARQIBO are provided in each kit.
SARASOTA MEMORIAL HOSPITAL NURSING PROCEDURE
 SARASOTA MEMORIAL HOSPITAL NURSING PROCEDURE TITLE: ISSUED FOR: Nursing DATE: REVIEWED: PAGES: 11/78 4/18 1 of 10 RESPONSIBILITY: RN (for chemo), LPN (nonchemo) PURPOSE: OBJECTIVE: To introduce medication
SARASOTA MEMORIAL HOSPITAL NURSING PROCEDURE TITLE: ISSUED FOR: Nursing DATE: REVIEWED: PAGES: 11/78 4/18 1 of 10 RESPONSIBILITY: RN (for chemo), LPN (nonchemo) PURPOSE: OBJECTIVE: To introduce medication
Step-by-step instructions for intravenous (iv) infusions for patients with:
 Step-by-step instructions for intravenous (iv) infusions for patients with: Rheumatoid Arthritis (RA) Systemic Juvenile Idiopathic Arthritis (sjia) Polyarticular Juvenile Idiopathic Arthritis (pjia) Please
Step-by-step instructions for intravenous (iv) infusions for patients with: Rheumatoid Arthritis (RA) Systemic Juvenile Idiopathic Arthritis (sjia) Polyarticular Juvenile Idiopathic Arthritis (pjia) Please
GESTATIONAL DIABETES (DIET/INSULIN/ METFORMIN) CARE OF WOMEN IN BIRTHING SUITE
 GESTATIONAL DIABETES (DIET/INSULIN/ METFORMIN) CARE OF WOMEN IN BIRTHING SUITE DEFINITION A disorder characterised by hyperglycaemia first recognised during pregnancy due to increased insulin resistance
GESTATIONAL DIABETES (DIET/INSULIN/ METFORMIN) CARE OF WOMEN IN BIRTHING SUITE DEFINITION A disorder characterised by hyperglycaemia first recognised during pregnancy due to increased insulin resistance
IV Therapy January, 08 Tip of the Month
 Every Hub Every Time IV Therapy January, 08 Tip of the Month Every Hub Every Time No matter what the occasion, SCRUB the catheter ports every single time before access. Evidence Supports SCRUBBING using
Every Hub Every Time IV Therapy January, 08 Tip of the Month Every Hub Every Time No matter what the occasion, SCRUB the catheter ports every single time before access. Evidence Supports SCRUBBING using
IV Link Staff. Infection Prevention & Control A Learning Package for IV Link Staff
 IV Link Staff Infection Prevention & Control A Learning Package for IV Link Staff Purpose This learning package provides key infection prevention messages and direction for knowledge that you require as
IV Link Staff Infection Prevention & Control A Learning Package for IV Link Staff Purpose This learning package provides key infection prevention messages and direction for knowledge that you require as
The University of Toledo Medical Center and its Medical Staff
 Name of Policy: Policy Number: Department: 3364-109-GEN-705 Infection Control Medical Staff Hospital Administration Approving Officer: Responsible Agent: Scope: Chair, Infection Control Committee Chief
Name of Policy: Policy Number: Department: 3364-109-GEN-705 Infection Control Medical Staff Hospital Administration Approving Officer: Responsible Agent: Scope: Chair, Infection Control Committee Chief
SARASOTA MEMORIAL HOSPITAL NURSING PROCEDURE
 SARASOTA MEMORIAL HOSPITAL NURSING PROCEDURE TITLE: ISSUED FOR: ADMINISTRATION OF A FEEDING (CONTINUOUS OR INTERMITTENT) OR MEDICATION VIA A GASTROSTOMY TUBE-ADULT Nursing DATE: REVIEWED: PAGES: 07/82
SARASOTA MEMORIAL HOSPITAL NURSING PROCEDURE TITLE: ISSUED FOR: ADMINISTRATION OF A FEEDING (CONTINUOUS OR INTERMITTENT) OR MEDICATION VIA A GASTROSTOMY TUBE-ADULT Nursing DATE: REVIEWED: PAGES: 07/82
Venepuncture and Cannulation. Louise Smith Clinical Nurse Specialist
 Venepuncture and Cannulation Louise Smith Clinical Nurse Specialist Outcomes By the end of this session you will be aware of: Basic anatomy Preparation procedures including patient identification Equipment
Venepuncture and Cannulation Louise Smith Clinical Nurse Specialist Outcomes By the end of this session you will be aware of: Basic anatomy Preparation procedures including patient identification Equipment
CATHETER ACCESS KIT. For use with Prometra Programmable Infusion Systems
 CATHETER ACCESS KIT Caution: Federal (USA) Law restricts this device to sale by or on the order of a physician. Table of Contents Contents... 3 Description... 3 Indications... 3 Contraindications... 3
CATHETER ACCESS KIT Caution: Federal (USA) Law restricts this device to sale by or on the order of a physician. Table of Contents Contents... 3 Description... 3 Indications... 3 Contraindications... 3
CONTROLLED DOCUMENT. Guidelines for the use of subcutaneous hydration in palliative care (hypodermoclysis) Controlled Document Number: CG259
 Guidelines for the use of subcutaneous hydration in palliative care (hypodermoclysis) CONTROLLED DOCUMENT CATEGORY: CLASSIFICATION: Controlled Document Number: Version Number: 1 Controlled Document Sponsor:
Guidelines for the use of subcutaneous hydration in palliative care (hypodermoclysis) CONTROLLED DOCUMENT CATEGORY: CLASSIFICATION: Controlled Document Number: Version Number: 1 Controlled Document Sponsor:
High Impact Intervention Peripheral intravenous cannula care bundle
 High Impact Intervention Peripheral intravenous cannula care bundle Aim To reduce the incidence of peripheral intravenous cannula infections. Introduction The aim of the care bundle, as set out in this
High Impact Intervention Peripheral intravenous cannula care bundle Aim To reduce the incidence of peripheral intravenous cannula infections. Introduction The aim of the care bundle, as set out in this
Infusion Skills Competency Checklist To be used at annual skills fair or at any other time for IV Competency
 Employee Profile Infusion Skills Checklist Last Name First Name Middle Initial Employee Number Employee Discipline Check one: RN LPN Per state specific LPN Practice Acts Direct Supervisor s Name: Date
Employee Profile Infusion Skills Checklist Last Name First Name Middle Initial Employee Number Employee Discipline Check one: RN LPN Per state specific LPN Practice Acts Direct Supervisor s Name: Date
You have a what, inside you?
 Costal Emergency Medicine Conference You have a what, inside you? Less than mainstream medical devices encountered in the ED. Eric Ossmann, MD, FACEP Associate Professor Duke University Medical Center
Costal Emergency Medicine Conference You have a what, inside you? Less than mainstream medical devices encountered in the ED. Eric Ossmann, MD, FACEP Associate Professor Duke University Medical Center
INTRAVENOUS HYDRALAZINE POLICY
 PURPOSE INTRAVENOUS HYDRALAZINE POLICY The purpose of this policy is to: provide safe and effective care for women establish a local approach to care that is evidence based and consistent inform good decision
PURPOSE INTRAVENOUS HYDRALAZINE POLICY The purpose of this policy is to: provide safe and effective care for women establish a local approach to care that is evidence based and consistent inform good decision
ASEPTIC DISPENSING TECHNIQUES/DRUG- NUTRIENT INTERACTIONS
 ASEPTIC DISPENSING TECHNIQUES/DRUG- NUTRIENT INTERACTIONS HARBANS KAUR DHILLON PRESIDENT OF PENSMA KUALA LUMPUR MALAYSIA PENSA 2007 University Malaya Medical Centre Kuala Lumpur ASEPTIC DISPENSING Ppn
ASEPTIC DISPENSING TECHNIQUES/DRUG- NUTRIENT INTERACTIONS HARBANS KAUR DHILLON PRESIDENT OF PENSMA KUALA LUMPUR MALAYSIA PENSA 2007 University Malaya Medical Centre Kuala Lumpur ASEPTIC DISPENSING Ppn
You have a what, inside you?
 Costal Emergency Medicine Conference You have a what, inside you? Less than mainstream medical devices encountered in the ED. Eric Ossmann, MD, FACEP Associate Professor Duke University Medical Center
Costal Emergency Medicine Conference You have a what, inside you? Less than mainstream medical devices encountered in the ED. Eric Ossmann, MD, FACEP Associate Professor Duke University Medical Center
Intravenous therapy and medication. ELCT Health Department Quality Assurance 2007
 Intravenous therapy and medication ELCT Health Department Quality Assurance 2007 Intravenous therapy and medication the procedure is absolutely sterile, everything should be handled remaining sterility
Intravenous therapy and medication ELCT Health Department Quality Assurance 2007 Intravenous therapy and medication the procedure is absolutely sterile, everything should be handled remaining sterility
Title The Prescription of Adult Inpatient Parenteral Nutrition (PN) Guideline. Author s job title Consultant Gastroenterologist Directorate Medicine
 Document Control Title The Prescription of Adult Inpatient Parenteral Nutrition (PN) Guideline Author Author s job title Consultant Gastroenterologist Directorate Medicine Department Medicine Version Date
Document Control Title The Prescription of Adult Inpatient Parenteral Nutrition (PN) Guideline Author Author s job title Consultant Gastroenterologist Directorate Medicine Department Medicine Version Date
The Impact of Catheter Occlusion in Central Line Associated Bloodstream Infections M A R C H 15, 2017
 The Impact of Catheter Occlusion in Central Line Associated Bloodstream Infections D A R C Y DOELLMAN M S N, RN, CRNI, VA - BC M A R C H 15, 2017 LOUISVILLE, KENTUCKY Cincinnati Children s Hospital 642
The Impact of Catheter Occlusion in Central Line Associated Bloodstream Infections D A R C Y DOELLMAN M S N, RN, CRNI, VA - BC M A R C H 15, 2017 LOUISVILLE, KENTUCKY Cincinnati Children s Hospital 642
BLOOD CULTURE POLICY FOR PAEDIATRICS
 BLOOD CULTURE POLICY FOR PAEDIATRICS 1. INTRODUCTION Blood culture to detect bacteraemia is an important investigation with major implications for the diagnosis of patients with infection and the selection
BLOOD CULTURE POLICY FOR PAEDIATRICS 1. INTRODUCTION Blood culture to detect bacteraemia is an important investigation with major implications for the diagnosis of patients with infection and the selection
CONTINUOUS RENAL REPLACEMENT THERAPY (CRRT)
 TABLE OF CONTENTS The purpose of this practice support document is to outline the procedures and guidelines related to care of the patient having continuous renal replacement therapy (CRRT) in the pediatric
TABLE OF CONTENTS The purpose of this practice support document is to outline the procedures and guidelines related to care of the patient having continuous renal replacement therapy (CRRT) in the pediatric
Value Life Lifecath Midli n e uide to Lifecath Midline rse s G u N A
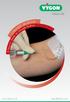 Value Life Lifecath Midl ine A Nurse s Guide to Lifecath Midline www.vygon.co.uk vygon@vygon.co.uk Useful Information Lifecath Midline Lifecath Midline Catheter Code: 1296 Peelable Cannula Introducer
Value Life Lifecath Midl ine A Nurse s Guide to Lifecath Midline www.vygon.co.uk vygon@vygon.co.uk Useful Information Lifecath Midline Lifecath Midline Catheter Code: 1296 Peelable Cannula Introducer
STANDARDIZED PROCEDURE REPROGRAMMING AND REFILLING INTRATHECAL BACLOFEN PUMPS and ACCESSING THE CATHETER ACCESS PORT (Adult,Peds)
 I. Definition The purpose of this procedure is to allow the Advanced Health Practitioner (AHP) to reprogram and refill intrathecal Baclofen pumps, as well as access the catheter access port for those AHPs
I. Definition The purpose of this procedure is to allow the Advanced Health Practitioner (AHP) to reprogram and refill intrathecal Baclofen pumps, as well as access the catheter access port for those AHPs
Policies and Procedures
 Policies and Procedures Title: CENTRAL VENOUS CATHETERS CARE OF PERIPHERALLY INSERTED CENTRAL CATHETERS (S) - accessing - dressing changes - tubing and adapter changes - flushing Authorization: [x] SHR
Policies and Procedures Title: CENTRAL VENOUS CATHETERS CARE OF PERIPHERALLY INSERTED CENTRAL CATHETERS (S) - accessing - dressing changes - tubing and adapter changes - flushing Authorization: [x] SHR
Common Infusions for Neonatal Use
 Common Infusions for Neonatal Use Document Title and Reference : Common Infusions for Neonatal Use Main Author (s) Dr N B Soni Ratified by: LSC CEG Date Ratified: May 2016 Review Date: May 2018 Version:
Common Infusions for Neonatal Use Document Title and Reference : Common Infusions for Neonatal Use Main Author (s) Dr N B Soni Ratified by: LSC CEG Date Ratified: May 2016 Review Date: May 2018 Version:
Home Care Services HomeMed MedEQUIP Michigan Visiting Care Michigan Visiting Nurses Wheelchair Seating Service PROCEDURE
 UNIVERSITY OF MICHIGAN HOSPITALS AND HEALTH CENTERS UMHHC-HCS: 253.054 First Approved Date: 3/2010 Home Care Services HomeMed MedEQUIP Michigan Visiting Care Michigan Visiting Nurses Wheelchair Seating
UNIVERSITY OF MICHIGAN HOSPITALS AND HEALTH CENTERS UMHHC-HCS: 253.054 First Approved Date: 3/2010 Home Care Services HomeMed MedEQUIP Michigan Visiting Care Michigan Visiting Nurses Wheelchair Seating
Policies and Procedures LPN Additional Competency: Care of Short Term, Tunneled, Implanted Central Venous Catheters
 Policies and Procedures LPN Additional Competency: Care of Short Term, Tunneled, Implanted Central Venous Catheters Title: CENTRAL VENOUS CATHETERS SHORT TERM, TUNNELED, IMPLANTED - CARE OF - accessing
Policies and Procedures LPN Additional Competency: Care of Short Term, Tunneled, Implanted Central Venous Catheters Title: CENTRAL VENOUS CATHETERS SHORT TERM, TUNNELED, IMPLANTED - CARE OF - accessing
CHEMOTHERAPY NETWORK GROUP POLICY FOR ADMINISTRATION OF CYTOTOXIC CHEMOTHERAPY
 CHEMOTHERAPY NETWORK GROUP POLICY FOR ADMINISTRATION OF CYTOTOXIC CHEMOTHERAPY Version 4.0 March 2016 Review date March 2018 Introduction It is the purpose of this policy to provide clear guidelines that
CHEMOTHERAPY NETWORK GROUP POLICY FOR ADMINISTRATION OF CYTOTOXIC CHEMOTHERAPY Version 4.0 March 2016 Review date March 2018 Introduction It is the purpose of this policy to provide clear guidelines that
Direct Intravenous (IV) Medication Administration Procedure
 Approved by: Chief Medical Officer; and Chief Operating Officer Direct Intravenous (IV) Medication Administration Procedure Corporate Policy & Procedures Manual Number: VII-B-310 Date Approved January
Approved by: Chief Medical Officer; and Chief Operating Officer Direct Intravenous (IV) Medication Administration Procedure Corporate Policy & Procedures Manual Number: VII-B-310 Date Approved January
Peripheral Cannulation
 Contents... 1 Purpose... 1 Scope... 1 Associated documents... 1 Certification Requirements for RN, EN, RM, IV and Anaesthetic Technicians... 1 Peripheral IV Cannulation Requirements... 2 Vein Assessment:...
Contents... 1 Purpose... 1 Scope... 1 Associated documents... 1 Certification Requirements for RN, EN, RM, IV and Anaesthetic Technicians... 1 Peripheral IV Cannulation Requirements... 2 Vein Assessment:...
PATIENT CARE MANUAL POLICY
 PATIENT CARE MANUAL POLICY NUMBER VII-A-15 PAGE 1 OF 3 APPROVED BY: CATEGORY: Vice President and Senior Operating Officer; Rural Health Services & Professional Practice Lead Body Systems; Central Nervous
PATIENT CARE MANUAL POLICY NUMBER VII-A-15 PAGE 1 OF 3 APPROVED BY: CATEGORY: Vice President and Senior Operating Officer; Rural Health Services & Professional Practice Lead Body Systems; Central Nervous
Standard Operating Procedures Clinical and Translational Research Center
 Standard Operating Procedures Clinical and Translational Research Center Title: Approved By: Number of Pages: Frequently Sampled Intravenous Glucose Tolerance Test (FSIVGTT) Effective March 19, 2012 Date:
Standard Operating Procedures Clinical and Translational Research Center Title: Approved By: Number of Pages: Frequently Sampled Intravenous Glucose Tolerance Test (FSIVGTT) Effective March 19, 2012 Date:
Total Parenteral Nutrition and Enteral Nutrition in the Home. Original Policy Date 12:2013
 MP 1.02.01 Total Parenteral Nutrition and Enteral Nutrition in the Home Medical Policy Section Durable Medical Equipment Issue Original Policy Date Last Review Status/Date Return to Medical Policy Index
MP 1.02.01 Total Parenteral Nutrition and Enteral Nutrition in the Home Medical Policy Section Durable Medical Equipment Issue Original Policy Date Last Review Status/Date Return to Medical Policy Index
Effect of changing lipid formulation in Parenteral Nutrition in the Newborn Experimental Pathology BSc
 Effect of changing lipid formulation in Parenteral Nutrition in the Newborn Experimental Pathology BSc Word count: 6939 0 CONTENTS Abstract...2 Acknowledgements...3 Introduction...4 Materials and Methods...11
Effect of changing lipid formulation in Parenteral Nutrition in the Newborn Experimental Pathology BSc Word count: 6939 0 CONTENTS Abstract...2 Acknowledgements...3 Introduction...4 Materials and Methods...11
Central Venous Catheter Care and Maintenance (includes catheter troubleshooting guide)
 Central Venous Catheter Care and Maintenance (includes catheter troubleshooting guide) A Guide for Patients in the Home Phone Number: Nurse/Contact: Central Venous Catheters This manual is a guide for
Central Venous Catheter Care and Maintenance (includes catheter troubleshooting guide) A Guide for Patients in the Home Phone Number: Nurse/Contact: Central Venous Catheters This manual is a guide for
Venous Access and Ports. Helen Starosta
 Venous Access and Ports Helen Starosta Venous access and ports Peripheral IV access Arterio-Venous Fistula Central venous access Peripherally Inserted Central Catheter (PICC) Non Tunnelled Central Venous
Venous Access and Ports Helen Starosta Venous access and ports Peripheral IV access Arterio-Venous Fistula Central venous access Peripherally Inserted Central Catheter (PICC) Non Tunnelled Central Venous
Parenteral Nutrition Policy
 Parenteral Nutrition Policy This is a new procedural document, please read in full Did you print this document yourself? The Trust discourages the retention of hard copies of policies and can only guarantee
Parenteral Nutrition Policy This is a new procedural document, please read in full Did you print this document yourself? The Trust discourages the retention of hard copies of policies and can only guarantee
Optimal nutrition in critically ill children
 Optimal nutrition in critically ill children MODULE 4 Good safety practices required for nutritional therapy Developed by The Asia Pacific Middle East Consensus Working Group on Nutrition Therapy in the
Optimal nutrition in critically ill children MODULE 4 Good safety practices required for nutritional therapy Developed by The Asia Pacific Middle East Consensus Working Group on Nutrition Therapy in the
4.0 Definitions. Subcutaneous: is defined as the loose connective tissue between the epidermis and the muscle.
 Status: Protocol: Specifies the procedures to be followed, only in exceptional circumstances should these not be followed. Title: Protocol on subcutaneous (SC) fluids (hypodermoclysis) administration Written
Status: Protocol: Specifies the procedures to be followed, only in exceptional circumstances should these not be followed. Title: Protocol on subcutaneous (SC) fluids (hypodermoclysis) administration Written
NHS GREATER GLASGOW & CLYDE CONTROL OF INFECTION COMMITTEE STANDARD OPERATING PROCEDURE (SOP) INSERTION & MAINTENANCE OF INDWELLING
 Page Page 1 of 6 AIM STATEMENT REQUIREMENTS LOCATION TIMING PROCEDURE To minimise the risk of secondary infection as a result of urinary catheterisation. A urinary catheter bypasses the body s normal defence
Page Page 1 of 6 AIM STATEMENT REQUIREMENTS LOCATION TIMING PROCEDURE To minimise the risk of secondary infection as a result of urinary catheterisation. A urinary catheter bypasses the body s normal defence
Education for self administration of intravenous therapy HOME IV THERAPY. 30 minute - Baxter Pump Tobramycin
 HOME IV THERAPY Tobramycin Tobramycin Check the order on the drug chart This can change when the results from your blood test come through. Your doctor will change the order, if required. A copy of the
HOME IV THERAPY Tobramycin Tobramycin Check the order on the drug chart This can change when the results from your blood test come through. Your doctor will change the order, if required. A copy of the
Intravenous Catheter Complications
 Vascular Access Device-Related Infection Inadequate skin antisepsis prior to VAD insertion Acute onset of fever, chills, and hypotension. No other apparent source of Notify Prescriber immediately Obtain
Vascular Access Device-Related Infection Inadequate skin antisepsis prior to VAD insertion Acute onset of fever, chills, and hypotension. No other apparent source of Notify Prescriber immediately Obtain
