Alberta Health Services Infection Prevention and Control - Initiatives and Services. Surveillance Protocol January 12, 2010 Rev.
|
|
|
- Winifred Joseph
- 5 years ago
- Views:
Transcription
1 Alberta Health Services Infection Prevention and Control - Initiatives and Services Hospital Acquired Bloodstream Infections (HABSI) Hospital Wide- in Acute Care and Acute Rehabilitation Facilities Surveillance Protocol January 12, 2010 Rev. April 21, 2011
2 Table of Contents INTRODUCTION... 3 OBJECTIVES... 3 PATIENT POPULATION... 3 DENOMINATOR DATA... 3 CASE DEFINITION... 4 OUTCOME... 5 RISK FACTORS... 5 METHODOLOGY... 6 RATE CALCULATIONS... 7 BENCHMARKING... 7 REPORTING... 7 SURVEILLANCE PERIOD... 7 REFERENCES... 8 APPENDIX A: EXAMPLE OF LINE DAY COLLECTION FORM FOR ADULT/PEDIATRIC ICU... 9 APPENDIX B: EXAMPLE OF LINE DAY COLLECTION FORM FOR NICU APPENDIX C: CASE INCLUSION ALGORITHM APPENDIX D: EXAMPLE OF COLLECTION FORM AHS-HABSI Surveillance Protocol -revapril 2011.doc Page 2 of 16
3 INTRODUCTION Hospital acquired bloodstream infections (BSIs) are an important cause of morbidity and mortality in severely ill patients. Although these infections account for less than 10% of the total of all hospital acquired infections, they contribute to increased length of stay and a higher cost of care. Surveillance is an essential component of infection prevention and control. If carried out in a uniform manner, surveillance provides a measure of the burden of illness, establishes benchmark rates for internal and external comparison, identifies potential risk factors, and allows assessment of specific interventions. Surveillance of hospital acquired BSI (HABSI) is considered a measure of quality of care 1,2. OBJECTIVES 1. To establish site-specific rates for HABSI in the acute care/acute rehabilitation patient population in Alberta Health Services (AHS) facilities. 2. To compare pooled AHS HABSI rates to appropriate established benchmarks. 3. To detect clusters of HABSI. 4. To identify areas for improvement and/or requirements for change of practice. 5. To report HABSI rates quarterly to appropriate stakeholders and provincial government. PATIENT POPULATION Any acute care/acute rehabilitation inpatient who has an organism identified in a blood culture after admission, in any acute or acute rehab site/facility in AHS. DENOMINATOR DATA Numbers of inpatient admissions and inpatient days will be obtained from Costing and Benchmarking on a quarterly basis. Central Venous Catheter (CVC) line days (as denominator in ICUs only) Count and record the number of patients with one or more CVC* at approximately the same time once each day. A patient with more than one CVC counts as only one CVC day. (see Appendix A). Denominator data is collected by ICU personnel. * A CVC is a venous access device that terminates at or close to the heart or one of the great vessels e.g. aorta, pulmonary artery, superior vena cava, inferior vena cava, brachiocephalic, internal jugular, subclavian, external iliac and common femoral veins, and umbilical artery and vein. CVC include non-tunneled (standard) CVC, whether coated or not, peripherally inserted CVC (PIC), tunneled devices (e.g. Broviac, Hickman, tunneled hemodialysis line, etc.) and umbilical artery and vein catheters. Totally implanted devices such as Ports are NOT included. Pulmonary artery catheters are included as these are inserted via a central vein. Other arterial catheters are NOT included. Pacemaker leads and other non-infusion devices inserted into central blood vessels or the heart are NOT included.1 AHS-HABSI Surveillance Protocol -revapril 2011.doc Page 3 of 16
4 Neonate Specific data stratification This population is stratified into five distinct categories base on birth weight: 1. < 750g g g g 5. >2500g Further, this group is risk stratified by the type of catheter used to provide patient care: 1. CVC 2. Umbilical 3. Umbilical + CVC Count and record the number of patients of each birth weight group with one or more CVC and the number with one or more umbilical catheters at approximately the same time each day. If a patient has both an umbilical catheter and CVC(s), count as an umbilical catheter day. If line type is not able to be differentiated, but line days are being collected use Umbilical + CVC (see Appendix B). CASE DEFINITION 3 Note: All infections included in surveillance must be hospital acquired (HA), therefore the BSI must not present or incubating on the time of admission and if patient has been in hospital for less than 48 hours prior to the onset of the BSI, there must be compelling evidence that the infection is attributable to the hospital. Primary BSI: 1. Recognized pathogen cultured from one or more blood cultures, unrelated to infection at another site. OR 2. At least one of: fever greater than 38 C, chills, hypotension or signs of infection of catheter insertion site, tunnel or pocket. AND Common skin contaminant (e.g. diphtheroids, Bacillus spp, Propionibacterium spp, coagulase negative staphylococci or micrococci) cultured from two or more blood cultures drawn from separate sites or on separate occasions (i.e. from different venipunctures, no minimal time interval). Please refer to the CDC/NHSN surveillance definition of health care associated infection and criteria for specific types of infections in the acute care setting to as guidance in identifying infections at another site. Symptoms of systemic infection for patients 1 year: one of the following with no other recognized cause: fever (>30 C core), hypothermia (<36 C core), apnea, or bradycardia AHS-HABSI Surveillance Protocol -revapril 2011.doc Page 4 of 16
5 Secondary BSI: Bloodstream infection which is bacteriologically, temporally and clinically related to an infection with the same pathogen at another body site of the patient. CVC-associated BSI: If CVC in use at onset of BSI or within the 48 hours before the onset of BSI If CVC has been in situ for less than 48 hours prior to the onset of BSI, as there is no minimum time that the CVC must be in place in order for the BSI to be considered CVC related, there must be compelling evidence that the infection is CVC-associated If CVC removed greater than 48 hours before onset there must be compelling evidence that the infection was associated with the CVC ICU-associated BSI: BSI not present or incubating on admission to ICU If patient has been in ICU for less than 48 hours prior to the onset of BSI, there must be compelling evidence that the infection is attributable to the ICU BSI onset during ICU stay or within 48 hours of leaving ICU Relapse vs. New Infection Same microorganism (as best as can be determined by the data available - e.g. species, antibiotic sensitivity, etc.) isolated from a subsequent blood culture: Relapse - if less than 10 days from a negative culture OR less than 10 days from completion of appropriate antibiotic therapy, consider as a relapse and DO NOT REPORT. New Infection - if more than 10 days from a negative culture (if culture was done) and more than 10 days from completion of appropriate antibiotic therapy REPORT AS A NEW INFECTION. OUTCOME Thirty days following positive blood culture determine if patient is: discharged deceased still in health care facility RISK FACTORS Certain factors may be associated with blood stream infections in specific patient populations. Information about these factors may be documented as additional data for these patients. These data points are included on the surveillance form for healthcare facilities that choose to document these details for the relevant patient groups. These factors include: 1. Premature rupture of membranes (maternal) for neonates (PROM>24 hrs) 2. Total parenteral nutrition (TPN) 3. Intra-aortic balloon pump (IABP) 4. Extracorporeal membrane oxygenation (ECMO) AHS-HABSI Surveillance Protocol -revapril 2011.doc Page 5 of 16
6 METHODOLOGY Identification of patients with BSI: Daily (or, as a minimum, two to three times weekly) review of microbiology laboratory results by Infection Control Practitioner. An algorithm for case inclusion is available in Appendix C For each positive blood culture: determine if patient is an inpatient or was an inpatient within the 48 hours prior to the time the specimen was obtained. Special: if a patient, upon admission, is blood culture positive and has been in hospital within the last 48hrs include in surveillance as hospital acquired if other case criteria is met. If the organism cultured in blood sample is a common skin contaminant ensure clinical criteria (symptoms of infection) are met. If criteria for infection are not present, this sample should be considered a contaminated blood culture and the case is not included in surveillance data. Information may be obtained from a variety of sources including: patient or family members, patient hospital charts, nurses logs, laboratory reports, nursing and medical staff. Determine if case definition for HABSI is met. Determine if HABSI meets definition for primary or secondary bacteremia If patient meets definition for primary BSI, that is, no evidence of infection at another body site with the same pathogen at the same time. If primary BSI, determine is it attributable to the CVC or not. Review patient s chart to determine if a CVC was in place or removed within the preceding 48 hours. If CVC associated primary BSI determine if attributable to the ICU or not. Determine if they were an admitted patient in an ICU or discharged from the ICU within the preceding 48 hours. For secondary BSI, identify site of infection causing the BSI. Note: Please refer to the CDC/NHSN surveillance definition of health care associated infection and criteria for specific types of infections in the acute care setting as guidance in identifying infections at another site. Fill in the data collection form (Appendix D) Follow-up at 30 days to determine outcome and finalize data collection form. AHS-HABSI Surveillance Protocol -revapril 2011.doc Page 6 of 16
7 RATE CALCULATIONS HABSI rates will be calculated quarterly for each acute care/acute rehabilitation facility participating in surveillance. Technical Definitions HABSI rate Number of HABSIs Number patient admissions Number of HABSIs Number patient days X 1,000 X 10,000 Note using the above definition rates may be further analyzed to derive rates for primary, primary CVC, and secondary HABSI. CVC-associated BSI rate (ICU Only) Number of CVC associated HABSIs Number of CVC line days X 1,000 Note using the above definition rates may be further analyzed to derive rates for neonates by birth weight and line type. BENCHMARKING Pooled Alberta Health Services HABSI rates will be compared with rates from appropriate Canadian Nosocomial Surveillance Program (CNISP) comparators. REPORTING HABSI rates will be reported quarterly to appropriate stakeholders within 60 days following the end of each quarter. SURVEILLANCE PERIOD AHS-HABSI surveillance commenced with a protocol trial September, 2009 with full implementation October, AHS-HABSI Surveillance Protocol -revapril 2011.doc Page 7 of 16
8 REFERENCES 1. Canadian Nosocomial Infection Surveillance Program (CNISP) Central Venous Catheter-Associated Blood Stream Infections in Intensive Care Units and in Recipients of Haemapoeietic Steam Cell Transplants. Protocol. December 22, Centers for Disease Control and Prevention. Guidelines for the prevention of intravascular catheter-related infections. MMWR 2002; 51 (No. RR-10): Canadian Nosocomial Infection Surveillance Program (CNISP), Surveillance for Central Venous Catheter Associated Blood Stream Infections (CVC-BSI) in Intensive Care Units, Revised April 15 th, CDC/NHSN surveillance definition of health care associated infection and criteria for specific types of infections in the acute care setting. AJIC June, 2002; Vol. 36. No.5: AHS-HABSI Surveillance Protocol -revapril 2011.doc Page 8 of 16
9 APPENDIX A: Example of Line day Collection Form for Adult/Pediatric ICU Intensive Care Unit Central Line Bloodstream Infection Surveillance Number of Line Days Please indicate the number of patients with central lines in place by bed number (i.e. count only one central line, even if the patient has more than one central line). A central line is one that enters a central vein either directly e.g. subclavian, jugular or femoral vein; or is inserted via a peripheral vein and is threaded into a central vein e.g. umbilical artery/venous catheters, PICC lines, Broviac catheters. Arterial lines are excluded. Lines in place should be documented at the same time each day. If a patient is discharged from a bed and a new patient admitted, do not revise count. Month of Unit Hospital Bed# Total
10 This page is intentionally blank for two sided printing.
11 APPENDIX B: Example of Line Day Collection Form for NICU SURVEILLANCE FOR CVC-ASSOCIATED BSI IN NEONATAL INTENSIVE CARE UNITS Daily denominator collection form for Neonatal Intensive Care Unit (NICU) Month Year Date Birth weight 750 g # # Pts UC #CVC Birth weight g # # Pts UC #CVC Birth weight g # # Pts UC #CVC Birth weight g Birth weight >2500 g # # # # Pts UC #CVC Pts UC #CVC Total # Pts=number of infants # UC=number of infants with umbilical catheter # CVC=number of infants with 1 or more central venous lines If infant has both a UC & CVC, count as UC day AHS-HABSI Surveillance Protocol -revapril 2011.doc Page 11 of 16
12 THIS PAGE IS INTENTIONALLY BLANK FOR TWO SIDED PRINTING. AHS-HABSI Surveillance Protocol -revapril 2011.doc Page 12 of 16
13 APPENDIX C: Case Inclusion Algorithm AHS-HABSI Surveillance Protocol -revapril 2011.doc Page 13 of 16
14 This page is intentionally blank for two sided printing. AHS-HABSI Surveillance Protocol -revapril 2011.doc Page 14 of 16
15 NAME PHN# MED REC# APPENDIX D: Example of Collection Form 1. PATIENT DEMOGRAPHICS / Admission Details PHN: NAME: Last name: First Name Gender: M F Admit Date: MM/DD/YY DOB: MM/DD/YYYY Med.Rec./Hosp # Site/Facility D/C Date: MM/DD/YYYY Birth weight (BWT) <750g g g g > 2500g Gestational Age: (weeks) 2. BSI PROM>24 hrs TPN IABP ECMO Date of Infection: (MM-DD-YYYY) (Date 1 st +) Hemodialysis Dialysis Line CVC Associated BSI ICU Associated BSI Central Perm Graft Central Temp Fistula FACILITY: UNIT/Bed #: SERVICE: PROGRAM: 3. Infection Date of Infection: (MM-DD-YYYY) (Date 1 st +) Primary Bacteremia CVC Related Non CVC Related Maternal IV Device Arterial Line Peripheral Line No line CVC PICC Line Type CVC Umbilical Umbilical + CVC Secondary Bacteremia (see criteria for infection on reverse side of form) Burn Skin GI Not endoscopy related Superficial Incision GI endoscopy related Deep Incision Respiratory Tract Organ Space Pneumonia Other Urinary Tract ICU Admission Date MM/DD/YYYY ICU Discharge Date: MM/DD/YYYY Outcome: (Patient disposition 30 days following BSI) Discharged Deceased Alive, in hospital (inpatient) 4. Pathogen (Organism Isolated) Additional Comments: Total Time Expended on Data Collection: (in minutes) Entered in Data Base AHS-HABSI Data Collection Form January 2010
16 Guideline for Completing Blood Stream Infection Surveillance Form BSI must not present or incubating on the time of admission and if patient has been in hospital for less than 48 hours prior to the onset of the BSI, there must be compelling evidence that the infection is attributable to the hospital. Primary BSI 1. Recognized pathogen cultured from one or more blood cultures, unrelated to infection at another site. OR 2. At least one of: fever greater than 38 C, chills, hypotension (if aged less than 1 year: one of fever greater than 38 C, hypothermia, apnea or bradycardia), or signs of infection of catheter insertion site, tunnel or pocket. AND Common skin contaminant (e.g. diphtheroids, Bacillus spp, Propionibacterium spp, coagulase negative staphylococci or micrococci) cultured from two or more blood cultures drawn from separate sites or on separate occasions (i.e. from different venipunctures, no minimal time interval). OR 3. At least one of: fever greater than 38 C, chills, hypotension (if aged less than 1 year: one of fever >38 C, hypothermia < 36 C, apnea or bradycardia), or signs of infection of catheter insertion site, tunnel or pocket. AND Common skin contaminant (as above) cultured from one blood culture from a patient with an intravenous line and the physician institutes appropriate antimicrobial therapy. Secondary BSI: A bloodstream infection which is bacteriologically, temporally and clinically related to an infection (CDC definition for guidance) with the same pathogen at another body site of the patient. Relapse vs New Infection Same microorganism (as best as can be determined by the data available- e.g. species, antibiotic sensitivity, etc) isolated from a subsequent blood culture: Relapse - if less than 10 days from a negative culture OR less than 10 days from completion of appropriate antibiotic therapy, consider as a relapse and DO NOT REPORT. New Infection - if more than 10 days from a negative culture (if culture was done) and more than 10 days from completion of appropriate antibiotic therapy REPORT AS A NEW INFECTION. ICU-associated BSI BSI was not present on admission to ICU. BSI onset during ICU stay or within 48 hours of leaving ICU CVC-associated BSI CVC-associated if catheter in use at onset of BSI or within the 48 hours before the onset of BSI (if CVC removed 48 hours before onset there must be compelling evidence that the infection was associated with the CVC) Signs/Symptoms Infection General: Chills Fever Pus Respiratory: Dr. s Dx Inflammation WBC >10 x 10/L Cough Chest X-Ray Resp Assist Signs Sputum Trach Urinary: Dysuria/Frequency/Urgency Hematuria Pyuria Flank Pain CVA/SP Tenderness Straight Cath Foley Instrumentation Stents Neonates Alter Resp Poor Feed Diarrhea WCB < 4x 10L GI Diarrhea AHS- HABSI Data Collection Form January 2010
Dialysis Event Protocol
 Dialysis Event Protocol Introduction In 2009, more than 370,000 patients were treated with maintenance hemodialysis in the United States. 1 Hemodialysis patients require a vascular access, which can be
Dialysis Event Protocol Introduction In 2009, more than 370,000 patients were treated with maintenance hemodialysis in the United States. 1 Hemodialysis patients require a vascular access, which can be
2018 CNISP HAI Surveillance Case definitions
 2018 CNISP HAI Surveillance Case definitions The following case definitions for the surveillance of healthcare-associated infections (HAIs) are used by all acute-care hospitals that participate in the
2018 CNISP HAI Surveillance Case definitions The following case definitions for the surveillance of healthcare-associated infections (HAIs) are used by all acute-care hospitals that participate in the
Surveillance for Central Line Associated Blood Stream Infections (CLABSI) in Intensive Care Units
 Canadian Nosocomial Infection Surveillance Program Surveillance for Central Line Associated Blood Stream Infections (CLABSI) in Intensive Care Units 2017 CLABSI Surveillance Protocol Revised November 7
Canadian Nosocomial Infection Surveillance Program Surveillance for Central Line Associated Blood Stream Infections (CLABSI) in Intensive Care Units 2017 CLABSI Surveillance Protocol Revised November 7
Central Line Care and Management
 Central Line Care and Management What is a Central Line/ CVAD? (central venous access device) A vascular infusion device that terminates at or close to the heart or in one of the great vessels (aorta,
Central Line Care and Management What is a Central Line/ CVAD? (central venous access device) A vascular infusion device that terminates at or close to the heart or in one of the great vessels (aorta,
Q 1 (May-July 2016) Q 2 (August-October 2016) Q 3 (November 2016-January 2017)
 Adult ICU: May 2016-July 2017 Table 1a. Counts and rates of positive blood cultures and blood stream infections which meet the case definition in your critical care unit and for all adult critical care
Adult ICU: May 2016-July 2017 Table 1a. Counts and rates of positive blood cultures and blood stream infections which meet the case definition in your critical care unit and for all adult critical care
Understanding the Dialysis Event Protocol and Avoiding Common Reporting Mistakes Alicia Shugart, MA
 Understanding the Dialysis Event Protocol and Avoiding Common Reporting Mistakes Alicia Shugart, MA Public Health Analyst Topics Dialysis Event Protocol Collecting data Reporting a numerator and denominator
Understanding the Dialysis Event Protocol and Avoiding Common Reporting Mistakes Alicia Shugart, MA Public Health Analyst Topics Dialysis Event Protocol Collecting data Reporting a numerator and denominator
LABORATORY CONFIRMED BLOODSTREAM INFECTIONS (LCBI) Surveillance Definitions Case Studies
 LABORATORY CONFIRMED BLOODSTREAM INFECTIONS (LCBI) Surveillance Definitions Case Studies PRIMARY BSI Primary bloodstream infections (BSI) Organism cultured from the blood that is not related to an infection
LABORATORY CONFIRMED BLOODSTREAM INFECTIONS (LCBI) Surveillance Definitions Case Studies PRIMARY BSI Primary bloodstream infections (BSI) Organism cultured from the blood that is not related to an infection
INFECTION PREVENTION AND CONTROL
 INFECTION PREVENTION AND CONTROL Health Care-Associated Infection (HAI) Definitions May 28, 2012 The Capital Health Infection Prevention Control (IPAC) department conducts ongoing surveillance reports
INFECTION PREVENTION AND CONTROL Health Care-Associated Infection (HAI) Definitions May 28, 2012 The Capital Health Infection Prevention Control (IPAC) department conducts ongoing surveillance reports
Catheter-Associated Urinary Tract Infection (CAUTI) Event
 Catheter-Associated Urinary Tract Infection () Event Introduction: The urinary tract is the most common site of healthcare-associated infection, accounting for more than 30% of infections reported by acute
Catheter-Associated Urinary Tract Infection () Event Introduction: The urinary tract is the most common site of healthcare-associated infection, accounting for more than 30% of infections reported by acute
LABORATORY CONFIRMED BLOODSTREAM INFECTIONS (LCBI) Surveillance Definitions Case Studies
 LABORATORY CONFIRMED BLOODSTREAM INFECTIONS (LCBI) Surveillance Definitions Case Studies PRIMARY BSI Primary bloodstream infections (BSI) Organism cultured from the blood that is not related to an infection
LABORATORY CONFIRMED BLOODSTREAM INFECTIONS (LCBI) Surveillance Definitions Case Studies PRIMARY BSI Primary bloodstream infections (BSI) Organism cultured from the blood that is not related to an infection
Recommendations for Hospital Quality Measures in 2011:
 Pediatric Measures: Recommendations for Hospital Quality Measures in 2011: Based on the input of a group of healthcare stakeholders, the following new hospital measures are recommended: 1) Home Management
Pediatric Measures: Recommendations for Hospital Quality Measures in 2011: Based on the input of a group of healthcare stakeholders, the following new hospital measures are recommended: 1) Home Management
Catheter-Associated Urinary Tract Infection (CAUTI) Event
 Device-associated Events Catheter-Associated Urinary Tract Infection () Event Introduction: The urinary tract is the most common site of healthcare-associated infection, accounting for more than 30% of
Device-associated Events Catheter-Associated Urinary Tract Infection () Event Introduction: The urinary tract is the most common site of healthcare-associated infection, accounting for more than 30% of
Bloodstream Infection Event (Central Line-associated Bloodstream Infection and Non-central line-associated Bloodstream Infection)
 Bloodstream Infection Event (Central Line-associated Bloodstream Infection and Non-central line-associated Bloodstream Infection) Settings: Surveillance will occur in all inpatient locations in Kuwait
Bloodstream Infection Event (Central Line-associated Bloodstream Infection and Non-central line-associated Bloodstream Infection) Settings: Surveillance will occur in all inpatient locations in Kuwait
Management of Catheter Related Bloodstream Infection (CRBSI), including Antibiotic Lock Therapy.
 Management of Catheter Related Bloodstream Infection (CRBSI), including Antibiotic Lock Therapy. Written by: Dr K Gajee, Consultant Microbiologist Date: June 2017 Approved by: Drugs & Therapeutics Committee
Management of Catheter Related Bloodstream Infection (CRBSI), including Antibiotic Lock Therapy. Written by: Dr K Gajee, Consultant Microbiologist Date: June 2017 Approved by: Drugs & Therapeutics Committee
Healthcare-associated infections acquired in intensive care units
 SURVEILLANCE REPORT Annual Epidemiological Report for 2015 Healthcare-associated infections acquired in intensive care units Key facts In 2015, 11 788 (8.3%) of patients staying in an intensive care unit
SURVEILLANCE REPORT Annual Epidemiological Report for 2015 Healthcare-associated infections acquired in intensive care units Key facts In 2015, 11 788 (8.3%) of patients staying in an intensive care unit
IV therapy. By: Susan Mberenga, RN, MSN. Copyright 2016, 2013, 2010, 2006, 2002 by Saunders, an imprint of Elsevier Inc.
 IV therapy By: Susan Mberenga, RN, MSN 1 IV Therapy Types of solutions Isotonic Hypotonic Hypertonic Caution: Too rapid or excessive infusion of any IV fluid has the potential to cause serious problems
IV therapy By: Susan Mberenga, RN, MSN 1 IV Therapy Types of solutions Isotonic Hypotonic Hypertonic Caution: Too rapid or excessive infusion of any IV fluid has the potential to cause serious problems
American College of Surgeons Critical Care Review Course 2012: Infection Control
 American College of Surgeons Critical Care Review Course 2012: Infection Control Overview: I. Central line associated blood stream infection (CLABSI) II. Ventilator associated pneumonia (VAP) I. Central
American College of Surgeons Critical Care Review Course 2012: Infection Control Overview: I. Central line associated blood stream infection (CLABSI) II. Ventilator associated pneumonia (VAP) I. Central
Catheter-Associated Urinary Tract Infection (CAUTI) Event
 Catheter-Associated Urinary Tract Infection () Event Introduction: Urinary tract infections (UTIs) are tied with pneumonia as the second most common type of healthcare-associated infection, second only
Catheter-Associated Urinary Tract Infection () Event Introduction: Urinary tract infections (UTIs) are tied with pneumonia as the second most common type of healthcare-associated infection, second only
Enhanced EARS-Net Surveillance 2017 First Half
 1 Enhanced EARS-Net Surveillance 2017 First Half In this report Main results for 2017, first half Breakdown of factors by organism and resistance subtype Device-association Data quality assessment Key
1 Enhanced EARS-Net Surveillance 2017 First Half In this report Main results for 2017, first half Breakdown of factors by organism and resistance subtype Device-association Data quality assessment Key
Consideration of some other specific indications: Bacteremia
 European Medicines Agency Workshop on Antibacterials, London 7-8 February 2011 Consideration of some other specific indications: Bacteremia Harald Seifert Institut für Medizinische Mikrobiologie, Immunologie
European Medicines Agency Workshop on Antibacterials, London 7-8 February 2011 Consideration of some other specific indications: Bacteremia Harald Seifert Institut für Medizinische Mikrobiologie, Immunologie
The Impact of Healthcare Associated Infection (HCAI)
 Instructions for use Save this presentation Feel free to add or delete slides as necessary, change information to suit local needs and as new guidance or evidence is published Disclaimer: Whilst the working
Instructions for use Save this presentation Feel free to add or delete slides as necessary, change information to suit local needs and as new guidance or evidence is published Disclaimer: Whilst the working
National Healthcare Safety Network: Central Line-associated Bloodstream Case Studies Teresa C. Horan, MPH
 National Healthcare Safety Network: Central Line-associated Bloodstream Case Studies Teresa C. Horan, MPH National Center for Emerging and Zoonotic Infectious Diseases Division of Healthcare Quality Promotion
National Healthcare Safety Network: Central Line-associated Bloodstream Case Studies Teresa C. Horan, MPH National Center for Emerging and Zoonotic Infectious Diseases Division of Healthcare Quality Promotion
Unit 11. Objectives. Indications for IV Therapy. Intravenous Access Devices & Common IV Fluids. 3 categories. Maintenance Replacement Restoration
 Unit 11 Fluids, Electrolytes and Acid Base Imbalances Intravenous Access Devices & Common IV Fluids Objectives Review the purpose and types of intravenous (IV) therapy. Recall the nursing care related
Unit 11 Fluids, Electrolytes and Acid Base Imbalances Intravenous Access Devices & Common IV Fluids Objectives Review the purpose and types of intravenous (IV) therapy. Recall the nursing care related
IV Fluids. Nursing B23. Objectives. Serum Osmolality
 IV Fluids Nursing B23 Objectives Discuss the purpose of IV Discuss nursing interventions in IV therapy Identify complications of IV therapy Differentiate between peripheral line, central line, and PICC
IV Fluids Nursing B23 Objectives Discuss the purpose of IV Discuss nursing interventions in IV therapy Identify complications of IV therapy Differentiate between peripheral line, central line, and PICC
Prevent Bloodstream Infections by Using Appropriate Devices
 37 Prevent Bloodstream Infections by Using Appropriate Devices Situation Catheter-associated infections include exit, tunnel, pocket and bloodstream infections. In the United States, when these types of
37 Prevent Bloodstream Infections by Using Appropriate Devices Situation Catheter-associated infections include exit, tunnel, pocket and bloodstream infections. In the United States, when these types of
Mary Lou Garey MSN EMT-P MedFlight of Ohio
 Mary Lou Garey MSN EMT-P MedFlight of Ohio Function Prolonged and frequent access to venous circulation Allows for patient to carry on normal life; decrease number of needle sticks Medications, parenteral
Mary Lou Garey MSN EMT-P MedFlight of Ohio Function Prolonged and frequent access to venous circulation Allows for patient to carry on normal life; decrease number of needle sticks Medications, parenteral
The University of Toledo Medical Center and its Medical Staff
 Name of Policy: Policy Number: Department: 3364-109-GEN-705 Infection Control Medical Staff Hospital Administration Approving Officer: Responsible Agent: Scope: Chair, Infection Control Committee Chief
Name of Policy: Policy Number: Department: 3364-109-GEN-705 Infection Control Medical Staff Hospital Administration Approving Officer: Responsible Agent: Scope: Chair, Infection Control Committee Chief
IV Fluids Nursing B23 Objectives Serum Osmolality 275 to 295 Isotonic
 1 IV Fluids Nursing B23 2 Objectives 3 Serum Osmolality Serum osmolality solute concentration of a solution Higher osmolality means greater pulling power for water Normal serum osmolality is 275 to 295
1 IV Fluids Nursing B23 2 Objectives 3 Serum Osmolality Serum osmolality solute concentration of a solution Higher osmolality means greater pulling power for water Normal serum osmolality is 275 to 295
The Clinical Significance of Blood Cultures. Presented BY; Cindy Winfrey, MSN, RN, CIC, DON- LTC TM, VA- BC TM
 The Clinical Significance of Blood Cultures Presented BY; Cindy Winfrey, MSN, RN, CIC, DON- LTC TM, VA- BC TM OVERVIEW Blood cultures are considered an important laboratory tool used to diagnose serious
The Clinical Significance of Blood Cultures Presented BY; Cindy Winfrey, MSN, RN, CIC, DON- LTC TM, VA- BC TM OVERVIEW Blood cultures are considered an important laboratory tool used to diagnose serious
Overview of CVADs. Type of device commonly used. Dwell time Flushing requirement Associated complications. lumens
 Source: Clinical Skills Management of Vascular Access Devices Pre-course handbook. Adapted with permission from NHS Lothian Employee and Education Development Team. Overview of CVADs Type of device Veins
Source: Clinical Skills Management of Vascular Access Devices Pre-course handbook. Adapted with permission from NHS Lothian Employee and Education Development Team. Overview of CVADs Type of device Veins
CASE SCENARIO EXERCISE
 påçííáëü=pìêîéáää~ååé=çñ=eé~äíüå~êé ^ëëçåá~íéç=fåñéåíáçå=mêçöê~ããé CASE SCENARIO EXERCISE CATHETER-ASSOCIATED URINARY TRACT INFECTION SURVEILLANCE SCOTTISH SURVEILLANCE OF HEALTHCARE ASSOCIATED INFECTION
påçííáëü=pìêîéáää~ååé=çñ=eé~äíüå~êé ^ëëçåá~íéç=fåñéåíáçå=mêçöê~ããé CASE SCENARIO EXERCISE CATHETER-ASSOCIATED URINARY TRACT INFECTION SURVEILLANCE SCOTTISH SURVEILLANCE OF HEALTHCARE ASSOCIATED INFECTION
Blood Culture Collection and Interpretation
 Blood Culture Collection and Interpretation Catherine Ernst, RN,PBT(ASCP) Blood Cultures Indications for blood culture collection Proper method for blood culture collection Interpreting a blood culture
Blood Culture Collection and Interpretation Catherine Ernst, RN,PBT(ASCP) Blood Cultures Indications for blood culture collection Proper method for blood culture collection Interpreting a blood culture
Dialysis Event Reporting ESRD Network 8 LAN June 18, 2014
 Dialysis Event Reporting ESRD Network 8 LAN June 18, 2014 Meredith Kanago, MSPH Epidemiologist, Healthcare Associated Infections and Antimicrobial Resistance Program, Tennessee Department of Health Acknowledgements
Dialysis Event Reporting ESRD Network 8 LAN June 18, 2014 Meredith Kanago, MSPH Epidemiologist, Healthcare Associated Infections and Antimicrobial Resistance Program, Tennessee Department of Health Acknowledgements
PATIENT DEMOGRAPHICS. Surname. Given name. Pacific Islander (non-maori) ADMISSION DETAILS
 Reviewer / hospital Date review started PATIENT DEMOGRAPHICS MRN DOB Sex Patient sticky label if available, else enter details here Surname Post-code Given name Australian Aborigine / TSI Middle Eastern
Reviewer / hospital Date review started PATIENT DEMOGRAPHICS MRN DOB Sex Patient sticky label if available, else enter details here Surname Post-code Given name Australian Aborigine / TSI Middle Eastern
Proprietary Acute Care Indicators
 Proprietary Acute Care Indicators Indicator 1a: Device-Associated Infections in the Intensive Care Unit Central Line-Associated Bloodstream Infections in the APICU, CCU, MICU, M/S ICU, & SICU Ventilator-Associated
Proprietary Acute Care Indicators Indicator 1a: Device-Associated Infections in the Intensive Care Unit Central Line-Associated Bloodstream Infections in the APICU, CCU, MICU, M/S ICU, & SICU Ventilator-Associated
MANAGEMENT OF INTRAVASCULAR (IV) LINES AND THERAPY. All GCC Countries
 TITLE/DESCRIPTION: MANAGEMENT OF INTRAVASCULAR (IV) LINES AND THERAPY INDEX NUMBER: EFFECTIVE DATE: APPLIES TO: ISSUING AUTHORITY: 01/01/2009 01/01/2013 All GCC Countries GULF COOPERATION COUNCIL CENTRE
TITLE/DESCRIPTION: MANAGEMENT OF INTRAVASCULAR (IV) LINES AND THERAPY INDEX NUMBER: EFFECTIVE DATE: APPLIES TO: ISSUING AUTHORITY: 01/01/2009 01/01/2013 All GCC Countries GULF COOPERATION COUNCIL CENTRE
BLOODSTREAM INFECTIONS (BSI)
 BLOODSTREAM INFECTIONS (BSI) EPIDEMIOLOGY, PATHOPHYSIOLOGY, AND PREVENTION REBEKAH MOEHRING, MD, MPH dicon.medicine.duke.edu dason.medicine.duke.edu Disclosures Grants to Institution: CDC, AHRQ, CDC Foundation
BLOODSTREAM INFECTIONS (BSI) EPIDEMIOLOGY, PATHOPHYSIOLOGY, AND PREVENTION REBEKAH MOEHRING, MD, MPH dicon.medicine.duke.edu dason.medicine.duke.edu Disclosures Grants to Institution: CDC, AHRQ, CDC Foundation
Prevention of intravascular catheter-related infection in Ireland
 Prevention of intravascular catheter-related infection in Ireland Item type Authors Publisher Report SARI Prevention of Intravascular Catheter-related Infection Sub-Committee HSE Health Protection Surveillance
Prevention of intravascular catheter-related infection in Ireland Item type Authors Publisher Report SARI Prevention of Intravascular Catheter-related Infection Sub-Committee HSE Health Protection Surveillance
2018 HEMODIALYSIS CATHETERS CODING AND REIMBURSEMENT GUIDE
 2018 HEMODIALYSIS CATHETERS CODING AND REIMBURSEMENT GUIDE Contents Overview of Central Venous Access s for Hemodialysis 2 Procedures Using Hemodialysis s 2 Physician Reimbursement for Hemodialysis s 3
2018 HEMODIALYSIS CATHETERS CODING AND REIMBURSEMENT GUIDE Contents Overview of Central Venous Access s for Hemodialysis 2 Procedures Using Hemodialysis s 2 Physician Reimbursement for Hemodialysis s 3
Indicator Definition
 Hospital Acquired Clostridium difficile Infection (CDI) Rate Full data definition sign-off completed. Name of Measure Name of Measure (short) Domain Type of Measure Business Context Rationale Hospital
Hospital Acquired Clostridium difficile Infection (CDI) Rate Full data definition sign-off completed. Name of Measure Name of Measure (short) Domain Type of Measure Business Context Rationale Hospital
Prevention of Central Line-associated Bloodstream Infections (CLA-BSIs) associated with Arterial Catheters
 Prevention of Central Line-associated Bloodstream Infections (CLA-BSIs) associated with Arterial Catheters William R. Jarvis, M.D. President, Jason and Jarvis Associates, LLC Saxe Communications 2012 Sponsored
Prevention of Central Line-associated Bloodstream Infections (CLA-BSIs) associated with Arterial Catheters William R. Jarvis, M.D. President, Jason and Jarvis Associates, LLC Saxe Communications 2012 Sponsored
Key Definitions. Downloaded from
 Key Definitions In order to make sure that every member of the team is clear about the definitions that will be used, we have provided a list of key terms in the next few pages along with commonly used
Key Definitions In order to make sure that every member of the team is clear about the definitions that will be used, we have provided a list of key terms in the next few pages along with commonly used
Surgical Privileges Form: Vascular Surgery
 Surgical Form: Vascular Surgery Clinical Request Applicant s Name:. License No. (If Any):... Date:... Scope of Practice:. Facility:.. Place of Work:. CATEGORY I: GENERAL PRIVILEGES 1. Admitting privileges
Surgical Form: Vascular Surgery Clinical Request Applicant s Name:. License No. (If Any):... Date:... Scope of Practice:. Facility:.. Place of Work:. CATEGORY I: GENERAL PRIVILEGES 1. Admitting privileges
Appendix E: Overview of Vascular
 Appendix E: Overview of Vascular 56 Peripheral Short Catheter, less than 3 inches (7.5 cm) in length; over-the-needle catheter is most common. Inserted by percutaneous venipuncture, generally into a hand
Appendix E: Overview of Vascular 56 Peripheral Short Catheter, less than 3 inches (7.5 cm) in length; over-the-needle catheter is most common. Inserted by percutaneous venipuncture, generally into a hand
Prevention of CAUTI is discussed in the CDC/HICPAC document, Guideline for Prevention of Catheter-associated Urinary Tract Infection 4.
 Urinary Tract Infection (Catheter-Associated Urinary Tract Infection [CA] and Non-Catheter-Associated Urinary Tract Infection []) and Other Urinary System Infection [USI]) Events Introduction: Urinary
Urinary Tract Infection (Catheter-Associated Urinary Tract Infection [CA] and Non-Catheter-Associated Urinary Tract Infection []) and Other Urinary System Infection [USI]) Events Introduction: Urinary
HOSPITAL INFECTION CONTROL
 HOSPITAL INFECTION CONTROL Objectives To be able to define hospital acquired infections discuss the sources and routes of transmission of infections in a hospital describe methods of prevention and control
HOSPITAL INFECTION CONTROL Objectives To be able to define hospital acquired infections discuss the sources and routes of transmission of infections in a hospital describe methods of prevention and control
Haemodialysis central venous catheter-related sepsis management guideline Version 3. NAME M. Letheren Chair Clinical Effectiveness Advisory Group
 Lancashire Teaching Hospitals NHS Foundation Trust Haemodialysis central venous catheter-related sepsis management guideline Version 3 AUTHOR APPROVED BY DATE AUTH REF. NO NAME REBG/00018/July12 Michael
Lancashire Teaching Hospitals NHS Foundation Trust Haemodialysis central venous catheter-related sepsis management guideline Version 3 AUTHOR APPROVED BY DATE AUTH REF. NO NAME REBG/00018/July12 Michael
Healthcare Associated Infection Report February 2016 data
 Healthcare Associated Infection Report February 2016 data Section 1 Board Wide Issues Section 1 of the HAIRT covers Board wide infection prevention and control activity and actions. For reports on individual
Healthcare Associated Infection Report February 2016 data Section 1 Board Wide Issues Section 1 of the HAIRT covers Board wide infection prevention and control activity and actions. For reports on individual
Nursing Care of the Dialysis Patient. Adrian Hordon, MSN, RN
 Nursing Care of the Dialysis Patient Adrian Hordon, MSN, RN Understand principles of hemodialysis Recognize different access ports Identify side effects and complications Discuss nursing care for pre and
Nursing Care of the Dialysis Patient Adrian Hordon, MSN, RN Understand principles of hemodialysis Recognize different access ports Identify side effects and complications Discuss nursing care for pre and
APIC NHSN Webinar 9/8/2015. Topic Overview. Overall Learning Objectives
 APIC NHSN Webinar Janet Brooks, Cindy Gross, Denise Leaptrot, & Eileen Scalise Subject Matter Experts September 9, 2015 National Center for Emerging and Zoonotic Infectious Diseases Place Descriptor Here
APIC NHSN Webinar Janet Brooks, Cindy Gross, Denise Leaptrot, & Eileen Scalise Subject Matter Experts September 9, 2015 National Center for Emerging and Zoonotic Infectious Diseases Place Descriptor Here
2016 National Healthcare Safety Network (NHSN) Definition Update. Peg Gilbert, RN, MS, CIC. Quality Improvement Advisor, Regional Lead.
 2016 National Healthcare Safety Network (NHSN) Definition Update Peg Gilbert, RN, MS, CIC Quality Improvement Advisor, Regional Lead Objectives Relate the changes in the NHSN surveillance definitions for
2016 National Healthcare Safety Network (NHSN) Definition Update Peg Gilbert, RN, MS, CIC Quality Improvement Advisor, Regional Lead Objectives Relate the changes in the NHSN surveillance definitions for
Port Design. Page 1. Port Placement, Removal, and Management. Selecting a Vascular Access Device. Thomas M. Vesely, MD
 Non-Dialysis Procedures Port Placement, Removal, and Management Thomas M. Vesely, MD Saint Louis, Missouri Selecting a Vascular Access Device Duration of use Number of lumens Frequency used Blood flow
Non-Dialysis Procedures Port Placement, Removal, and Management Thomas M. Vesely, MD Saint Louis, Missouri Selecting a Vascular Access Device Duration of use Number of lumens Frequency used Blood flow
Infection Prevention and Control in Long Term Care Part 2
 Infection Prevention and Control in Long Term Care Part 2 Course ID: 1029 - Credit Hours: 2 Author(s) Bonnie Chustz,RN, BSN WCC Disclosures None Accreditation KLA Education Services LLC is accredited by
Infection Prevention and Control in Long Term Care Part 2 Course ID: 1029 - Credit Hours: 2 Author(s) Bonnie Chustz,RN, BSN WCC Disclosures None Accreditation KLA Education Services LLC is accredited by
Central Venous Access Devices. Stephanie Cunningham Amy Waters
 Central Venous Access Devices Stephanie Cunningham Amy Waters 5 Must Know Facts About CVAD s 1) What are CVAD s? 2) What are CVAD s used for? 3) How are these devices put in? 4) What are the complications
Central Venous Access Devices Stephanie Cunningham Amy Waters 5 Must Know Facts About CVAD s 1) What are CVAD s? 2) What are CVAD s used for? 3) How are these devices put in? 4) What are the complications
SURGICAL SITE INFECTIONS: SURVEILLANCE & PREVENTION
 SURGICAL SITE INFECTIONS: SURVEILLANCE & PREVENTION Facts There were an estimated 157,500 surgical site infections associated with inpatient surgeries in 2011. SSIs were the most common healthcare-associated
SURGICAL SITE INFECTIONS: SURVEILLANCE & PREVENTION Facts There were an estimated 157,500 surgical site infections associated with inpatient surgeries in 2011. SSIs were the most common healthcare-associated
5. Use of antibiotics, which disturbs balance of normal flora. 6. Poor nutritional status.
 Microbiology Chapter 5 Introduction to Pathogens 5:1 Changing Patterns of Disease In the past 100 years, since the discovery of the Germ Theory of Disease was accepted: We have learned that microbes cause
Microbiology Chapter 5 Introduction to Pathogens 5:1 Changing Patterns of Disease In the past 100 years, since the discovery of the Germ Theory of Disease was accepted: We have learned that microbes cause
Annex 4. Case definitions of infections
 Protocol for validation of PPS of HAIs and antimicrobial use in European LTCF TECHNICAL DOCUMENT Annex 4. Case definitions of infections Healthcare-associated infections and antimicrobial use in European
Protocol for validation of PPS of HAIs and antimicrobial use in European LTCF TECHNICAL DOCUMENT Annex 4. Case definitions of infections Healthcare-associated infections and antimicrobial use in European
(Facility Name and Address) (1D) Surveillance of Urinary Tract Infections in the Long-Term Care Setting
 Policy Number: 1D Date: 4/16/14 Version: 1 (1D) Surveillance of Urinary Tract Infections in the Long-Term Care Setting Introduction: One-quarter of the older adult population in the United States will
Policy Number: 1D Date: 4/16/14 Version: 1 (1D) Surveillance of Urinary Tract Infections in the Long-Term Care Setting Introduction: One-quarter of the older adult population in the United States will
Appendix B: Provincial Case Definitions for Reportable Diseases
 Ministry of Health and Long-Term Care Infectious Diseases Protocol Appendix B: Provincial Case Definitions for Reportable Diseases Disease: Carbapenemase-producing Enterobacteriaceae (CPE) infection or
Ministry of Health and Long-Term Care Infectious Diseases Protocol Appendix B: Provincial Case Definitions for Reportable Diseases Disease: Carbapenemase-producing Enterobacteriaceae (CPE) infection or
CRITICAL CARE SURVEILLANCE: CENTRAL VENOUS CATHETER RELATED INFECTIONS ALL WALES
 Welsh Healthcare Associated Infections Programme (WHAIP) Rhaglen Heintiau sy n Gysylltiedig a Gofal Iechyd Cymru (RHGGIC) CRITICAL CARE SURVEILLANCE: CENTRAL VENOUS CATHETER RELATED INFECTIONS ALL WALES
Welsh Healthcare Associated Infections Programme (WHAIP) Rhaglen Heintiau sy n Gysylltiedig a Gofal Iechyd Cymru (RHGGIC) CRITICAL CARE SURVEILLANCE: CENTRAL VENOUS CATHETER RELATED INFECTIONS ALL WALES
NHSN Catheter-Associated Urinary Tract Infection Surveillance in 2016
 NHSN Catheter-Associated Urinary Tract Infection Surveillance in 2016 Presented by: Susan Steeves MSN, RN, CNL, CIC June 28,2016 Taken from: NHSN Training 2016 March 3, 2016 Eileen Scalise, MSN, RN Nurse
NHSN Catheter-Associated Urinary Tract Infection Surveillance in 2016 Presented by: Susan Steeves MSN, RN, CNL, CIC June 28,2016 Taken from: NHSN Training 2016 March 3, 2016 Eileen Scalise, MSN, RN Nurse
Central Line-Associated Infections (CLABSI) Settings Toolkit
 Central Line-Associated Infections (CLABSI) in Non-Intensive Care Unit (non-icu) Settings Toolkit Activity C: ELC Prevention Collaboratives Alex Kallen, MD, MPH and Priti Patel, MD, MPH Division of Healthcare
Central Line-Associated Infections (CLABSI) in Non-Intensive Care Unit (non-icu) Settings Toolkit Activity C: ELC Prevention Collaboratives Alex Kallen, MD, MPH and Priti Patel, MD, MPH Division of Healthcare
Sustained CAUTI and CLABSI Improvements Using a Bundled Approach
 Sustained CAUTI and CLABSI Improvements Using a Bundled Approach Experiences of a Level 1 Trauma Center Angela Booth-Jones, PHD, MS, CIC Director, Infection Prevention 1 2 3 Miami Valley Hospital MVH has
Sustained CAUTI and CLABSI Improvements Using a Bundled Approach Experiences of a Level 1 Trauma Center Angela Booth-Jones, PHD, MS, CIC Director, Infection Prevention 1 2 3 Miami Valley Hospital MVH has
IR Central Venous Access [ ] Pre Procedure
![IR Central Venous Access [ ] Pre Procedure IR Central Venous Access [ ] Pre Procedure](/thumbs/90/104119758.jpg) IR Central Venous Access [1050200001] Pre Procedure Case Request/Scheduling Procedure Enter IR Case Request if not already completed (All hospitals except Grant Medical Center) [ ] Case Request IR Lab
IR Central Venous Access [1050200001] Pre Procedure Case Request/Scheduling Procedure Enter IR Case Request if not already completed (All hospitals except Grant Medical Center) [ ] Case Request IR Lab
Advanced Training Program Infection Prevention and Control By Dr. Ahmad Farouk EBFM, MRCGP, CIC
 Advanced Training Program Infection Prevention and Control By Dr. Ahmad Farouk EBFM, MRCGP, CIC Tel: +973 172 80 8 50 Mobile: +973 343 58 323 Fax: +973 a 11446 Address: BMMI Tower, Office 1423, 14 th Floor,
Advanced Training Program Infection Prevention and Control By Dr. Ahmad Farouk EBFM, MRCGP, CIC Tel: +973 172 80 8 50 Mobile: +973 343 58 323 Fax: +973 a 11446 Address: BMMI Tower, Office 1423, 14 th Floor,
Central Line Associated Blood Stream Infection (CLABSI) William Parker, PharmD, BCPS, CGP, CPh
 Central Line Associated Blood Stream Infection (CLABSI) William Parker, PharmD, BCPS, CGP, CPh Learning Objectives for Pharmacists Define Central Line Associated Blood Stream Infections (CLABSIs) and differentiate
Central Line Associated Blood Stream Infection (CLABSI) William Parker, PharmD, BCPS, CGP, CPh Learning Objectives for Pharmacists Define Central Line Associated Blood Stream Infections (CLABSIs) and differentiate
Catheter Associated Urinary Tract Infection
 Catheter Associated Urinary Tract Infection Facts about CAUTI 12% to 16% of adult patients will receive a urinary catheter during hospitalization. 4 th most common HAI Accounts for more than 12% of acute
Catheter Associated Urinary Tract Infection Facts about CAUTI 12% to 16% of adult patients will receive a urinary catheter during hospitalization. 4 th most common HAI Accounts for more than 12% of acute
PARENTERAL NUTRITION: VASCUAR ACCESS DEVICE SELECTION
 PARENTERAL NUTRITION: VASCUAR ACCESS DEVICE SELECTION Winifred Magambo-Gasana Vascular Access Nurse Practitioner Oxford University Hospitals NHS Foundation Trust Aim An overview of the range of Vascular
PARENTERAL NUTRITION: VASCUAR ACCESS DEVICE SELECTION Winifred Magambo-Gasana Vascular Access Nurse Practitioner Oxford University Hospitals NHS Foundation Trust Aim An overview of the range of Vascular
NEONATAL SEPSIS. Dalima Ari Wahono Astrawinata Departemen Patologi Klinik, FKUI-RSCM
 NEONATAL SEPSIS Dalima Ari Wahono Astrawinata Departemen Patologi Klinik, FKUI- Background Neonatal sepsis : Early-onset Late-onset Early-onset : mostly premature neonates Within 24 hours 85% 24-48 hours
NEONATAL SEPSIS Dalima Ari Wahono Astrawinata Departemen Patologi Klinik, FKUI- Background Neonatal sepsis : Early-onset Late-onset Early-onset : mostly premature neonates Within 24 hours 85% 24-48 hours
You have a what, inside you?
 Costal Emergency Medicine Conference You have a what, inside you? Less than mainstream medical devices encountered in the ED. Eric Ossmann, MD, FACEP Associate Professor Duke University Medical Center
Costal Emergency Medicine Conference You have a what, inside you? Less than mainstream medical devices encountered in the ED. Eric Ossmann, MD, FACEP Associate Professor Duke University Medical Center
You have a what, inside you?
 Costal Emergency Medicine Conference You have a what, inside you? Less than mainstream medical devices encountered in the ED. Eric Ossmann, MD, FACEP Associate Professor Duke University Medical Center
Costal Emergency Medicine Conference You have a what, inside you? Less than mainstream medical devices encountered in the ED. Eric Ossmann, MD, FACEP Associate Professor Duke University Medical Center
Catheter Associated Urinary Tract Infection
 Catheter Associated Urinary Tract Infection Facts about CAUTI 12% to 16% of adult patients will receive a urinary catheter during hospitalization. 4 th most common HAI Accounts for more than 12% of acute
Catheter Associated Urinary Tract Infection Facts about CAUTI 12% to 16% of adult patients will receive a urinary catheter during hospitalization. 4 th most common HAI Accounts for more than 12% of acute
Enhanced EARS-Net Surveillance REPORT FOR 2012 DATA
 Enhanced EARS-Net Surveillance REPORT FOR DATA 1 In this report Main results for Proposed changes to the enhanced programme Abbreviations Used Here BSI Bloodstream Infections CVC Central Venous Catheter
Enhanced EARS-Net Surveillance REPORT FOR DATA 1 In this report Main results for Proposed changes to the enhanced programme Abbreviations Used Here BSI Bloodstream Infections CVC Central Venous Catheter
MANAGEMENT OF HAEMODIALYSIS CATHETER RELATED BLOOD STREAM INFECTION
 MANAGEMENT OF HAEMODIALYSIS CATHETER RELATED BLOOD STREAM INFECTION RRCV CMG Renal and Transplant Service 1. Introduction Catheter related blood stream infection (CR-BSI) is a common complication in patients
MANAGEMENT OF HAEMODIALYSIS CATHETER RELATED BLOOD STREAM INFECTION RRCV CMG Renal and Transplant Service 1. Introduction Catheter related blood stream infection (CR-BSI) is a common complication in patients
Procedures/Risks:central venous catheter
 Procedures/Risks:central venous catheter Central Venous Catheter Placement Procedure: Placement of the central venous catheter will take place in the Interventional Radiology Department (IRD) at The Ohio
Procedures/Risks:central venous catheter Central Venous Catheter Placement Procedure: Placement of the central venous catheter will take place in the Interventional Radiology Department (IRD) at The Ohio
Prevention of CAUTI is discussed in the CDC/HICPAC document, Guideline for Prevention of Catheter-associated Urinary Tract Infection.
 Urinary Tract Infection (Catheter-Associated Urinary Tract Infection [CA] and Non-Catheter-Associated Urinary Tract Infection []) and Other Urinary System Infection [USI]) Events Introduction: Urinary
Urinary Tract Infection (Catheter-Associated Urinary Tract Infection [CA] and Non-Catheter-Associated Urinary Tract Infection []) and Other Urinary System Infection [USI]) Events Introduction: Urinary
Microbiology Laboratory Directors, Infection Preventionists, Primary Care Providers, Emergency Department Directors, Infectious Disease Physicians
 MEMORANDUM DATE: October 1, 2009 TO: FROM: SUBJECT: Microbiology Laboratory Directors, Infection Preventionists, Primary Care Providers, Emergency Department Directors, Infectious Disease Physicians Michael
MEMORANDUM DATE: October 1, 2009 TO: FROM: SUBJECT: Microbiology Laboratory Directors, Infection Preventionists, Primary Care Providers, Emergency Department Directors, Infectious Disease Physicians Michael
HAIs in the USA. March 23, 2017 Department of Public Health Sciences. Infectious Disease Epidemiology BMTRY 713 (Healthcare Associated Infection )
 Infectious Disease Epidemiology BMTRY 713 (A. Selassie, DrPH) Lecture 17 Healthcare-Associated Infections (HAIs) Learning Objectives 1. Describe magnitude and implication of HAIs 2. Identify the main risk
Infectious Disease Epidemiology BMTRY 713 (A. Selassie, DrPH) Lecture 17 Healthcare-Associated Infections (HAIs) Learning Objectives 1. Describe magnitude and implication of HAIs 2. Identify the main risk
Microbiological diagnosis of infective endocarditis; what is new?
 Microbiological diagnosis of infective endocarditis; what is new? Dr Amani El Kholy, MD Professor of Clinical Pathology (Microbiology), Faculty of Medicine, Cairo University ESC 2017 1 Objectives Lab Diagnostic
Microbiological diagnosis of infective endocarditis; what is new? Dr Amani El Kholy, MD Professor of Clinical Pathology (Microbiology), Faculty of Medicine, Cairo University ESC 2017 1 Objectives Lab Diagnostic
Bloodstream Infections
 GUIDE TO INFECTION CONTROL IN THE HOSPITAL CHAPTER NUMBER 30: Bloodstream Infections Authors Larry Lutwick MD Gonzalo Bearman MD, MPH Chapter Editor Ziad A. Memish, MD, FRCPC, FACP Topic Outline Definition
GUIDE TO INFECTION CONTROL IN THE HOSPITAL CHAPTER NUMBER 30: Bloodstream Infections Authors Larry Lutwick MD Gonzalo Bearman MD, MPH Chapter Editor Ziad A. Memish, MD, FRCPC, FACP Topic Outline Definition
Patient guide: pfm Nit-Occlud PDA coil occlusion system. Catheter occlusion of. Patent Ductus Arteriosus. with the
 Patient guide: Catheter occlusion of Patent Ductus Arteriosus with the pfm Nit-Occlud PDA coil occlusion system pfm Produkte für die Medizin - AG Wankelstr. 60 D - 50996 Cologne Phone: +49 (0) 2236 96
Patient guide: Catheter occlusion of Patent Ductus Arteriosus with the pfm Nit-Occlud PDA coil occlusion system pfm Produkte für die Medizin - AG Wankelstr. 60 D - 50996 Cologne Phone: +49 (0) 2236 96
Vascular Surgery Rotation Objectives for Junior Residents (PGY-1 and 2)
 Vascular Surgery Rotation Objectives for Junior Residents (PGY-1 and 2) Definition Vascular surgery is the specialty concerned with the diagnosis and management of congenital and acquired diseases of the
Vascular Surgery Rotation Objectives for Junior Residents (PGY-1 and 2) Definition Vascular surgery is the specialty concerned with the diagnosis and management of congenital and acquired diseases of the
Central venous catheters
 Follow the link from the online version of this article to obtain certified continuing medical education credits Central venous catheters Reston N Smith, 1 Jerry P Nolan 2 1 North Bristol NHS Trust, Bristol,
Follow the link from the online version of this article to obtain certified continuing medical education credits Central venous catheters Reston N Smith, 1 Jerry P Nolan 2 1 North Bristol NHS Trust, Bristol,
Reviews in Infection
 Reviews in Infection RIF 1(1):1-6 (2010) RIF ISSN:1837-6746 Original Research Infection related processes during haemodialysis: A study in General Hospital Haemodialysis unit Naser Hussain 1, *Mona F.
Reviews in Infection RIF 1(1):1-6 (2010) RIF ISSN:1837-6746 Original Research Infection related processes during haemodialysis: A study in General Hospital Haemodialysis unit Naser Hussain 1, *Mona F.
Infected cardiac-implantable electronic devices: diagnosis, and treatment
 Infected cardiac-implantable electronic devices: diagnosis, and treatment The incidence of infection following implantation of cardiac implantable electronic devices (CIEDs) is increasing at a faster rate
Infected cardiac-implantable electronic devices: diagnosis, and treatment The incidence of infection following implantation of cardiac implantable electronic devices (CIEDs) is increasing at a faster rate
Venous Access and Ports. Helen Starosta
 Venous Access and Ports Helen Starosta Venous access and ports Peripheral IV access Arterio-Venous Fistula Central venous access Peripherally Inserted Central Catheter (PICC) Non Tunnelled Central Venous
Venous Access and Ports Helen Starosta Venous access and ports Peripheral IV access Arterio-Venous Fistula Central venous access Peripherally Inserted Central Catheter (PICC) Non Tunnelled Central Venous
Central Venous Access Devices and Infection
 Central Venous Access Devices and Infection Dr Andrew Daley Microbiology & Infectious Diseases Women s & Children s Health Melbourne Background Types of infection! Local site infection! Blood stream infection!
Central Venous Access Devices and Infection Dr Andrew Daley Microbiology & Infectious Diseases Women s & Children s Health Melbourne Background Types of infection! Local site infection! Blood stream infection!
Experience. Initials Medical Staffing Network Rev. 06/15 F01201 NNICU 2 of 5
 Neonatal Intensive Care Unit Self Assessment Directions Please circle a value for each question to provide us and the interested facilities with an assessment of your clinical experience. These values
Neonatal Intensive Care Unit Self Assessment Directions Please circle a value for each question to provide us and the interested facilities with an assessment of your clinical experience. These values
Infectious Diseases in Clinical Practice February 2011 Lisa G. Winston, MD
 Infectious Diseases in Clinical Practice February 2011 Lisa G. Winston, MD A 60 year old man was hospitalized 2 months ago for CABG. His course was uncomplicated, and he was discharged after 5 days. He
Infectious Diseases in Clinical Practice February 2011 Lisa G. Winston, MD A 60 year old man was hospitalized 2 months ago for CABG. His course was uncomplicated, and he was discharged after 5 days. He
Canadian Nosocomial Infection Surveillance Program (CNISP)
 Canadian Nosocomial Infection Surveillance Program (CNISP) 2017 Surveillance Protocol for Carbapenem-Resistant Gram-Negatives (CRGN) in CNISP Healthcare Facilities December 29, 2017 FINAL Working Group:
Canadian Nosocomial Infection Surveillance Program (CNISP) 2017 Surveillance Protocol for Carbapenem-Resistant Gram-Negatives (CRGN) in CNISP Healthcare Facilities December 29, 2017 FINAL Working Group:
Blood cultures in ED. Dr Sebastian Chang MBBS FACEM
 Blood cultures in ED Dr Sebastian Chang MBBS FACEM Why do we care about blood cultures? blood cultures are the most direct method for detecting bacteraemia in patients a positive blood culture: 1. can
Blood cultures in ED Dr Sebastian Chang MBBS FACEM Why do we care about blood cultures? blood cultures are the most direct method for detecting bacteraemia in patients a positive blood culture: 1. can
A A U
 PVD Venous AUC Rating Sheet 2nd Round 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 Median I NI MADM Rating Agree Disagree Upper Extremity Venous Evaluation Table 1. Venous Duplex of the Upper Extremities for Patency
PVD Venous AUC Rating Sheet 2nd Round 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 Median I NI MADM Rating Agree Disagree Upper Extremity Venous Evaluation Table 1. Venous Duplex of the Upper Extremities for Patency
INFECTION SURVEILLANCE
 INFECTION SURVEILLANCE IN LONG-TERM CARE 5 TH ANNUAL WYOMING INFECTION PREVENTION CONFERENCE, 2015 Russ Forney, PhD, MT(ASCP), Mountain-Pacific Quality Health THE PROBLEM Long-Term Care Acute Care Time
INFECTION SURVEILLANCE IN LONG-TERM CARE 5 TH ANNUAL WYOMING INFECTION PREVENTION CONFERENCE, 2015 Russ Forney, PhD, MT(ASCP), Mountain-Pacific Quality Health THE PROBLEM Long-Term Care Acute Care Time
Quality & Hospital Acquired Conditions
 Quality & Hospital Acquired Conditions Rebecca Armbruster, DO, MS, FACOI Medical Director Resource Management Patricia Heys, BS Director of Infection Prevention & Control Sally Hinkle, DNP, MPA, RN Director
Quality & Hospital Acquired Conditions Rebecca Armbruster, DO, MS, FACOI Medical Director Resource Management Patricia Heys, BS Director of Infection Prevention & Control Sally Hinkle, DNP, MPA, RN Director
3M Tegaderm CHG Chlorhexidine Gluconate I.V. Securement Dressing Description 3M Tegaderm CHG Chlorhexidine Gluconate I.V. Securement Dressing is used
 3M Tegaderm CHG Chlorhexidine Gluconate I.V. Securement Dressing Description 3M Tegaderm CHG Chlorhexidine Gluconate I.V. Securement Dressing is used to cover and protect catheter sites and to secure devices
3M Tegaderm CHG Chlorhexidine Gluconate I.V. Securement Dressing Description 3M Tegaderm CHG Chlorhexidine Gluconate I.V. Securement Dressing is used to cover and protect catheter sites and to secure devices
Jun 15, CPT codes and are assigned for bilateral upper or lower extremity arterial assessments to check blood flow in relation to a
 Jun 15, 2009. CPT codes 93922 and 93923 are assigned for bilateral upper or lower extremity arterial assessments to check blood flow in relation to a blockage.. 93922 Noninvasive physiologic studies of
Jun 15, 2009. CPT codes 93922 and 93923 are assigned for bilateral upper or lower extremity arterial assessments to check blood flow in relation to a blockage.. 93922 Noninvasive physiologic studies of
Arterial Map of the Thorax, Abdomen and Pelvis 2017 Edition
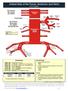 Arterial Map of the Thorax, Abdomen and Pelvis Angiography 75605 (-26) Aortography, thoracic 75625 (-26) Aortography, abdominal by serialography 75630 (-26) Aortography, abdominal + bilat iliofemoral 75705
Arterial Map of the Thorax, Abdomen and Pelvis Angiography 75605 (-26) Aortography, thoracic 75625 (-26) Aortography, abdominal by serialography 75630 (-26) Aortography, abdominal + bilat iliofemoral 75705
Vaxcel Implantable Ports Valved and Non-Valved. A Patient s Guide
 Vaxcel Implantable Ports Valved and Non-Valved A Patient s Guide Vaxcel Implantable Port This pamphlet provides some answers to questions you may have about your implantable port and how to care for it
Vaxcel Implantable Ports Valved and Non-Valved A Patient s Guide Vaxcel Implantable Port This pamphlet provides some answers to questions you may have about your implantable port and how to care for it
convey the clinical quality measure's title, number, owner/developer and contact
 CMS-0033-P 153 convey the clinical quality measure's title, number, owner/developer and contact information, and a link to existing electronic specifications where applicable. TABLE 20: Proposed Clinical
CMS-0033-P 153 convey the clinical quality measure's title, number, owner/developer and contact information, and a link to existing electronic specifications where applicable. TABLE 20: Proposed Clinical
Guidelines for the Prevention of Intravascular Catheter-Related Infections
 Guidelines for the Prevention of Intravascular Catheter-Related Infections Naomi P. O Grady, MD*; Mary Alexander, BS ; E. Patchen Dellinger, MD ; Julie L. Gerberding, MD, MPH ; Stephen O. Heard, MD ; Dennis
Guidelines for the Prevention of Intravascular Catheter-Related Infections Naomi P. O Grady, MD*; Mary Alexander, BS ; E. Patchen Dellinger, MD ; Julie L. Gerberding, MD, MPH ; Stephen O. Heard, MD ; Dennis
