Different regional patterns of cortical thinning in. Alzheimer s disease and frontotemporal dementia (FTD) are
|
|
|
- Chad Greer
- 5 years ago
- Views:
Transcription
1 doi: /brain/awm016 Brain (2007), 130, 1159^1166 Different regional patterns of cortical thinning in Alzheimer s disease and frontotemporal dementia An-Tao Du, 1, * Norbert Schuff, 1,2 Joel H. Kramer, 3 Howard J. Rosen, 4 Maria Luisa Gorno-Tempini, 4 Katherine Rankin, 4 Bruce L. Miller 4 and Michael W. Weiner 1^5 1 Center for Imaging of Neurodegenerative Diseases, Department of Veterans Affairs Medical Center, Departments of 2 Radiology, 3 Psychiatry, 4 Neurology and 5 Medicine, University of California, San Francisco, CA, USA Correspondence to: An-Tao Du, MD, Center for Imaging of Neurodegenerative Diseases, VA Medical Center, 4150, Clement street, San Francisco, CA 94121, USA antao.du@ucsf.edu *Present address: Department of Neurology, Wayne State University, Detroit, MI, USA antao@wayne.edu Alzheimer s disease and frontotemporal dementia (FTD) can be difficult to differentiate clinically because of overlapping symptoms. Distinguishing the two dementias based on volumetric measurements of brain atrophy with MRI has been only partially successful. Whether MRI measurements of cortical thinning improve the differentiation between Alzheimer s disease and FTD is unclear. In this study, we measured cortical thickness using a set of automated tools (Freesurfer) to reconstruct the brain s cortical surface from T 1 -weighted structural MRI data in 22 patients with Alzheimer s disease, 19 patients with FTD and 23 cognitively normal subjects.the goals were to detect the characteristic patterns of cortical thinning in these two types of dementia, to test the relationship between cortical thickness and cognitive impairment, to determine if measurement of cortical thickness is better than that of cortical volume for differentiating between these dementias and normal ageing and improving the classification of Alzheimer s disease and FTD based on neuropsychological scores alone. Compared to cognitively normal subjects, Alzheimer s disease patients had a thinner cortex primarily in bilateral, frontal, parietal, temporal and occipital lobes (P50.001), while FTD patients had a thinner cortex in bilateral, frontal and temporal regions and some thinning in inferior parietal regions and the posterior cingulate (P50.001). Compared to FTD patients, Alzheimer s disease patients had a thinner cortex (P50.001) in parts of bilateral parietal and precuneus regions. Cognitive impairment was negatively correlated with cortical thickness of frontal, parietal and temporal lobes in Alzheimer s disease, while similar correlations were not significant in FTD. Measurement of cortical thickness was similar to that of cortical volume in differentiating between normal ageing, Alzheimer s disease and FTD. Furthermore, cortical thickness measurements significantly improved the classification between Alzheimer s disease and FTD based on neuropsychological scores alone, including the Mini-Mental State Examination and a modified version of the Trail-Making Test. In conclusion, the characteristic patterns of cortical thinning in Alzheimer s disease and FTD suggest that cortical thickness may be a useful surrogate marker for these types of dementia. Keywords: Alzheimer s disease; frontotemporal dementia; cortical thickness; cortical volume Abbreviations: FTD, frontotemporal dementia; CN, cognitively normal; MMSE, Mini-Mental State Examination; CDR, Clinical Dementia Rating scale; TMT, Trail-Making Test Received September 16, Revised December 5, Accepted January 17, Advance Access publication March 12, 2007 Introduction Alzheimer s disease and frontotemporal dementia (FTD) are sometimes difficult to differentiate clinically because of overlapping symptoms (McKhann et al., 1984; Neary et al., 1998; Siri et al., 2001). Definite diagnosis requires histopathological examination of brain tissue. Although structural MRI data depict characteristic patterns of brain atrophy in Alzheimer s disease and FTD, aiding a differential diagnosis between the dementias (Kitagaki et al., 1998; Frisoni et al., 1999; Laakso et al., 2000; Rosen et al., 2002; Gee et al., 2003; Grossman et al., 2004; ß The Author (2007). Published by Oxford University Press on behalf ofthe Guarantors of Brain. Allrights reserved. For Permissions, please journals.permissions@oxfordjournals.org
2 1160 Brain (2007), 130, 1159^1166 A.-T. Du et al. Lipton et al., 2004; Whitwell et al., 2005), a complete division based on MRI has not been accomplished. Histopathological studies reported that Alzheimer s disease and FTD pathologies are associated with damage to specific cortical layers, e.g. layer II of the entorhinal cortex and layer III of the neocortex in Alzheimer s disease and layer III and V of frontal and temporal lobes in FTD (Pearson et al., 1985; Lewis et al., 1987; Gomez-Isla et al., 1996; Kersaitis et al., 2004). Although current MRI methods lack the power to resolve individual cortical layers, these histological observations raise the possibility that MRI-based examination of cortical thickness may be more specific than volumetric measurements for a differential diagnosis between Alzheimer s disease and FTD. However, since the cortex is a highly folded structure and its surface is rarely positioned perpendicular to any of the cardinal axes, measurements of cortical thickness are difficult, especially in presence of pathological alterations. Techniques have been recently developed for measuring cortical thickness in MRI using automated surface reconstruction, transformation and high-resolution intersubject alignment procedures (Fischl et al., 1999; Dale et al., 1999; Fischl and Dale, 2000). In addition, a recent MRI study of cortical thickness showed thinning of the cortex in broad brain regions such as medial temporal lobe, frontal and parietal lobes in patients with Alzheimer s disease when compared to cognitively normal (CN) subjects, consistent with the expected pathological pattern of Alzheimer s disease (Lerch et al., 2005). However, to what extent these patterns in Alzheimer s disease are dissociable from other dementias has not been established. Furthermore, MRI reports of cortical thinning in FTD are sparse. Therefore the main goal of this study was to determine the characteristic pattern of cortical thinning in FTD compared to CN and differences in cortical thickness between FTD and Alzheimer s disease. The second goal was to explore the relationship between cortical thickness and severity of cognitive impairment in Alzheimer s disease and FTD. Lastly, we compared the diagnostic value of assessing cortical thinning versus cortical volume loss for differentiating between normal ageing, Alzheimer s disease and FTD and tested if measurement of cortical thickness improves the classification between Alzheimer s disease and FTD based on neuropsychological scores alone. Material and methods Subjects Twenty-three CN subjects, 22 patients diagnosed with Alzheimer s disease and 19 patients diagnosed with FTD were included in the study (Table 1). The majority of subjects in this study are identical to those reported in our previous perfusion study (Du et al., 2006) except for two patients with Alzheimer s disease, two patients with FTD and two CN subjects, who had MRI of inferior quality, not suitable for reliable tissue segmentation and spatial normalization processing with Freesurfer ( The patients with FTD and Alzheimer s disease were recruited Table 1 Demographics Controls AD FTD Number (F/M) 23 (14/9) 22(8/14) 19 (3/16) Age (years) MMSE y CDR box score Modified trail y P50.01 between AD vs CN or FTD vs CN; y P50.01 between Alzheimer s disease and FTD. In units of number of corrected lines per minute. from the Memory and Aging Center of the University of California, San Francisco as described in detail in our previous paper (Du et al., 2006). All patients were diagnosed based upon information obtained from an extensive clinical history and physical examination. FTD was diagnosed according to the consensus criteria established by Neary et al. (1998). Patients with FTD who had motor neuron disease-related symptoms were excluded. Patients with Alzheimer s disease were diagnosed according to the criteria of the National Institute of Neurological and Communicative Disorders and Stroke- Alzheimer s Disease and Related Disorders Association (NINCDS/ADRDA) (McKhann et al., 1984). All subjects received a standard battery of neuropsychological tests, including assessment of global cognitive impairment using the Mini-Mental State Examination (MMSE) (Folstein et al., 1975) scores and global functional impairment using the Clinical Dementia Rating (CDR) scale (Morris, 1993). A modified version of the Trail-Making Test (TMT) was used to assess executive functions (Rosen et al., 2004). MRI data were visually inspected by a radiologist to rule out major neuropathologies other than neurodegeneration, such as tumour, stroke and severe white matter disease. All subjects or their guardians gave written informed consent before participating in the study, which was approved by the Committees of Human Research at the University of California and the VA Medical Center at San Francisco. Data acquisition and processing MRI data were obtained on a 1.5 T Siemens Vision TM System (Siemens Inc., Iselin NJ), using a standard quadrature head coil. Structural MRI data were acquired using a double spin echo (DSE) sequence and a volumetric magnetization-prepared rapid gradient echo (MPRAGE) T 1 -weighted sequence. The parameters of DSE images were: TR/TE1/TE2 ¼ 2500/20/80 ms timing, mm 2 in-plane resolution, and about 50 contiguous 3.00 mm thick axial slices oriented along the optic nerve as seen from a sagittal scout MR image. The parameters of MPRAGE T 1 -weighted images were: TR/TE/TI ¼ 10/7/300 ms timing, 15 flip angle, mm 2 in-plane resolution, and 1.40 mm thick coronal partitions and oriented orthogonal to the image planes of DSE. The construction cortical surface was based on 3D MPRAGE images using Freesurfer ( software. The detailed procedure for the surface construction with Freesurfer has been described and validated in previous papers (Fischl et al., 1999; Dale et al., 1999; Fischl and Dale, 2000). In brief, the procedure involves segmentation of white matter, tessellation of the grey/white matter junction, inflation of the folded surface
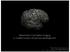
3 Cortical thinning in AD and FTD Brain (2007), 130, 1159^ tessellation patterns and automatic correction of topological defects in the resulting manifold. This surface is then used as the starting point for a deformable surface algorithm designed to find the grey/ white and pial surfaces with submillimetre precision. This method uses both intensity and continuity information from the surfaces in the deformation procedure in order to interpolate surface locations for regions in which the MRI image is ambiguous. For each subject, thickness of the cortical ribbon was computed on a uniform grid with 1 mm spacing across both cortical hemispheres, with the thickness being defined by the shortest distance between the grey/white and pial surface models (Fischl and Dale, 2000), providing in essence estimates of submillimetre differences. Thickness measures were mapped to the inflated surface of each subject s brain reconstruction, allowing visualization of data across the entire cortical surface. All images were aligned to a common surface template using a highresolution surface-based averaging technique that aligned cortical folding patterns. Regions of interest (ROI) on a standard brain (Desikan et al., 2006) were mapped back to each participant s native image space using a high-dimensional spherical morphing procedure to find the homologous regions across subjects. Then volume and mean thickness of cortical grey matter in each ROI were determined. Finally, cortical thickness was smoothed with a 10-mm full width at half height Gaussian kernel to reduce local variations in the measurements for further analysis. Statistics The comparison of regional cortical thickness variations between groups was tested regionally unbiased vertex-by-vertex using analysis of covariance (ANCOVA) with adjustments for age and sex effects. Significance was set to a P-value of without correction for family-wise errors. In addition to the regionally unbiased approach, ROIs measured cortical thickness in frontal, parietal and temporal lobes were also compared between the groups using ANCOVA with age and sex as covariates. Furthermore, the ROI measurements were used to test the relationship between cortical thickness in frontal, parietal and temporal lobe regions and cognitive function. Relationships between neuropsychological scores and MRI measures were tested using Pearson correlation statistics. The discriminatory powers of cortical thickness, cortical volume and neuropsychological tests were tested by leave-one-out cross-validations of the logistic regressions. In addition, the comparison of cortical thickness and volume for differentiating between CN, Alzheimer s disease and FTD was tested by comparing the area under receiver operator characteristic analysis with Mann Whitney tests (DeLong et al., 1988). The significance level of the tests was a Results The comparison of cortical thickness in Alzheimer s disease and FTD versus CN Figure 1 depicts the regional pattern of cortical thinning in patients with Alzheimer s disease compared to CN subjects, showing for the Alzheimer s disease significant cortical thinning in broad regions of bilateral frontal, parietal, temporal and occipital lobes (P50.001), while the sensorimotor cortical regions are spared. The most significant thinning in Alzheimer s disease involved bilateral medial temporal, temporoparietal regions, the posterior cingulate and the precuneus. There was no brain region in patients with Alzheimer s disease where the cortex was significantly thicker than in CN subjects. Figure 2 depicts the regional pattern of cortical thinning in patients with FTD compared to CN subjects, showing for the FTD significant cortical thinning in bilateral frontal and temporal regions, and in some inferior parietal regions and the posterior cingulate Fig. 1 Regional variation of cortical thickness in Alzheimer s disease compared to controls. The colour-code for P-values is on a logarithmic scale of 1^7. Warmer colours (positive values) represent cortical thinning; cooler colours (negative values) represent cortical thickening.
4 1162 Brain (2007), 130, 1159^1166 A.-T. Du et al. (P50.001). The most significant thinning in FTD involved bilateral prefrontal regions, the anterior and posterior cingulate and right anterior temporal region. Similar to Alzheimer s disease, there was no brain region in FTD patients where the cortex was significantly thicker than in CN subjects. Compared to patients with FTD, patients with Alzheimer s disease had a thinner cortex (P50.001) in parts of bilateral parietal and precuneus regions and in left temporal and occipital regions. In contrast, patients with FTD exhibited no significant regions of cortical thinning when compared to patients with Alzheimer s disease. Comparison of cortical thickness between Alzheimer s disease and FTD Figure 3 depicts regional differences of cortical thickness between patients with Alzheimer s disease and FTD. ROI analysis of cortical thickness in frontal, parietal and temporal lobes Average cortical thickness in the frontal, temporal and parietal lobes is listed by group in Table 2. Consistent with Fig. 2 Regional variation of cortical thickness in FTD compared to controls. The colour-coding for P-values is on a logarithmic scale of 1^5. Warmer colours (positive values) represent cortical thinning; cooler colours (negative values) represent cortical thickening. Fig. 3 Regional variations of cortical thickness between Alzheimer s disease and FTD. The colour-coding is identical to that shown in Fig. 2.
5 Cortical thinning in AD and FTD Brain (2007), 130, 1159^ the vertex-by-vertex analysis, patients with Alzheimer s disease and FTD had thinner cortices in the frontal, temporal and parietal lobes than CN subjects (all P50.001), independent of age and gender. Furthermore, patients with Alzheimer s disease had a thinner parietal cortex than patients with FTD (P50.001), while differences between Alzheimer s disease and FTD in the frontal and temporal cortices were not significant. Similar to the vertexby-vertex analysis, FTD patients exhibited no significant regions of cortical thinning when compared to patients with Alzheimer s disease. Since in the clinical FTD group, there is a greater risk that older patients could be false positive where the true pathological diagnosis is actually Alzheimer s disease and parietal cortical thinning is considered a feature of Alzheimer s disease, we tested in FTD separately the extent to which age explains parietal cortical thinning, but found no significant age effect (P ¼ 0.17). Furthermore, we compared the neuropsychological profile of FTD patients having a cortical thickness thicker than the median parietal cortical thickness (1.83 mm) with those having a cortical thickness thinner than the median parietal cortical thickness, but found no significant difference in MMSE, CDR or TMT scores (all P40.8) between the two groups. Relationship between cortical thickness and cognitive function The relationship between cortical thickness and cognitive functions were tested including only Alzheimer s disease and FTD patients. MMSE scores were positively correlated with cortical thickness of the frontal (r ¼ 0.50, P50.05), temporal (r ¼ 0.48, P50.05) and parietal lobes (r ¼ 0.46, P50.05) in Alzheimer s disease, but not in FTD (all P40.5). In Alzheimer s disease, CDR box scores were negatively correlated with cortical thickness of parietal lobe (r ¼ 0.45, P50.05), but not that of frontal and temporal lobes, in contrast to MMSE scores. In FTD, no significant correlations were found between CDR box scores and cortical thickness, similar to the results for MMSE. Since the distributions of both MMSE and CDR box scores were skewed towards higher dementia severity, we repeated the analyses of correlations for log-transformed MMSE and CDR data but obtained similar results. Furthermore, executive function, as measured with TMT, was not Table 2 Average cortical thickness (in mm) and standard deviation of frontal, parietal and temporal lobes in CN, Alzheimer s disease and FTD Frontal Temporal Parietal CN Alzheimer s disease FTD y P between Alzheimer s disease vs CN or between FTD vs CN; y P between Alzheimer s disease and FTD. significantly correlated with cortical thickness of frontal, parietal or temporal lobe in patients with FTD (all regions P40.16). Classification between groups Results of group classifications using either volume or thickness of frontal, temporal or parietal cortex are summarized in Table 3. This shows that Alzheimer s disease patients could be distinguished from controls with 87 to 99% accuracy, based on either volume or thickness of frontal, parietal and temporal cortex. In comparison, FTD patients could be distinguished from controls with 74 to 94% accuracy based on either volume or thickness of frontal, parietal and temporal cortex. Moreover, Alzheimer s disease and FTD patients could be distinguished from each other with 76 to 83% accuracy, based on either volume or thickness of parietal cortex. However, whether volume or thickness was used for classification made no significant difference (all P40.3). Note, volume and thickness of frontal and temporal cortex were not used for classification, because differences in these regions between Alzheimer s disease and FTD were not significant. Using MMSE scores alone correctly (P50.01) separated FTD and Alzheimer s disease patients with an overall classification of 72 2%. Using TMT scores alone correctly (P50.01) separated FTD and Alzheimer s disease patients with an overall classification of 76 2%. Adding parietal cortical thickness to the MMSE scores significantly improved (P50.01) overall classification between Alzheimer s disease and FTD to 78 2%. Similarly, adding parietal cortical thickness to TMT scores significantly improved (P ¼ 0.02) overall classification between Alzheimer s disease and FTD to 84 2%. Discussion The major findings of this study are: (i) Alzheimer s disease is associated with cortical thinning primarily in the frontal, parietal, temporal and occipital lobes, while a different regional pattern of cortical thinning is found in FTD, Table 3 Overall classification between CN, FTD and AD based on logistic regressions and leave-one-out cross validations using either cortical thickness or volumes of frontal, parietal and temporal lobes Cortical measure Alzheimer s disease vs CN (%) FTD vs CN (%) Alzheimer s disease vs FTD (%) Volume Frontal ^a Parietal Temporal ^a Frontal ^a Thickness Parietal Temporal ^a a No difference between AD vs FTD.
6 1164 Brain (2007), 130, 1159^1166 A.-T. Du et al. involving primarily the frontal and temporal lobes. The pattern of cortical thinning in each disease is consistent with previous MRI studies using volumetric measurements of brain atrophy and also with histopathological findings of the brain s selective vulnerability to Alzheimer s disease and FTD; (ii) dementia severity is negatively correlated with cortical thickness in Alzheimer s disease, while comparable correlations in FTD were not significant; (iii) measurement of cortical thickness provided similar accuracy as that of cortical volume for differentiating between CN, Alzheimer s disease and FTD and significantly improved the classification between Alzheimer s disease and FTD based on neuropsychological scores alone. The finding of characteristic patterns of cortical thinning in Alzheimer s disease replicates the previous study (Lerch et al., 2005) and is consistent with the pattern of tissue loss reported by histopathological and volumetric MRI studies (Braak and Braak, 1995, 1998; Baron et al., 2001). Furthermore, we found that FTD is associated with a characteristic regional pattern of cortical thinning in frontal, temporal regions and parietal lobe, and also with greatest cortical atrophy in prefrontal regions, and less cortical atrophy in parietal regions. The findings in FTD are consistent with previous pathological reports and volumetric MRI studies (Rosen et al., 2002; Broe et al., 2003; Grossman et al., 2004; Whitwell et al., 2005). In particular, prominent thinning of frontal and temporal cortex in FTD is in agreement with autopsy findings from macroscopic examinations of FTD brains showing consistently frontal and temporal and, less commonly, parietal atrophy (Dickson, 2001; Kersaitis et al., 2004). Furthermore, this study showed that FTD was associated with the cortical thinning in both orbital and medial frontal cortices with a similar severity, which is also consistent with the previous pathological study that FTD is associated with the orbital and medial frontal cortices in the early stage (Broe et al., 2003). However, the previous VBM studies (Rosen et al., 2005; Williams et al., 2005) have shown that in FTD, the behaviour change is related to grey matter loss in the medial frontal cortex other than the orbital frontal cortex. More studies may be needed to elucidate the relationship between the behaviour change and cortical atrophy with cortical thickness measurement in FTD. While patients with Alzheimer s disease had thinner cortices on MRI than FTD patients, specifically in the precuneus and the parietal lobe regions, no cortical region in FTD was significantly thinner than in Alzheimer s disease. The finding that Alzheimer s disease and FTD patients showed similar levels of cortical thinning in the frontal lobe is surprising, given that greater brain atrophy in frontal regions in FTD than Alzheimer s disease has previously been reported (Grossman et al., 2004). Different demographics of patients including stage and severity of cognitive impairment may explain the different findings. For example, in the previous MRI study (Grossman et al., 2004), patients with Alzheimer s disease or FTD were matched for dementia severity based solely on MMSE scores, which assesses predominantly memory impairments but less executive functions. In fact, a previous neuropsychological study demonstrated that FTD patients could be more impaired in judgement and problem solving than Alzheimer s disease patients when MMSE scores of the patients are matched (Rosen et al., 2004). It is therefore conceivable that previous findings of more frontal lobe atrophy in FTD than in Alzheimer s disease can be explained at least in part by greater executive dysfunction in some of FTD patients than in Alzheimer s disease patients. It is also possible that Alzheimer s disease heterogeneity may lead to different results, depending whether the frontal cortex is involved in the disease process or not. Although there was no difference in cortical thinning in the frontal regions between Alzheimer s disease and FTD in this study, we found that Alzheimer s disease was associated with a thinner cortex in the dorsolateral frontal cortex than the orbital and medial frontal cortices (Fig. 1), while FTD was associated with a similar cortical thinning in the dorsolateral, orbital and medial frontal cortices (Fig. 2). Taken together, these results suggest that while there are areas of overlap between the atrophic patterns of the two dementias, their patterns are dissociable and measurements of cortical thickness may be a useful surrogate marker for Alzheimer s disease and FTD. In this study, we found that FTD was associated with cortical thinning in inferior parietal lobes and the posterior cingulate cortex, which are prominent regions affected by Alzheimer s disease pathology. Other MRI studies using voxel-based morphometry found no significant structural abnormalities in these regions in FTD (Rosen et al., 2002; Gee et al., 2003; Whitwell et al., 2005). There are several possible explanations for the discrepancy. First, cortical thickness measurements may be more sensitive than voxelbased morphometry in detecting cortical alterations. Second, older patients with FTD may have concomitant Alzheimer s disease pathology or could be false negatively classified as Alzheimer s disease patients. However, we did not find systematic differences in brain atrophy between younger and older FTD patients; nor did we discover differences in the neurocognitive characteristics between FTD patients with more parietal atrophy and less parietal atrophy. Nonetheless, only autopsy can provide conclusive results for comorbidity of Alzheimer s disease pathology in some patients with FTD symptoms. Another finding is that regional cortical thinning correlated with dementia severity in Alzheimer s disease but no significant correlations were seen in FTD. It is unclear why correlations show a disease selective effect. One reason could be that severity of FTD, which presents with predominantly behavioural problems, is not accurately reflected in MMSE and CDR tests, which assess primarily cognitive functions, though other studies have used MMSE and CDR together to compare dementia severity between Alzheimer s disease and FTD (Likeman et al., 2005). Previous pathological studies showed that FTD impacts
7 Cortical thinning in AD and FTD Brain (2007), 130, 1159^ also white matter and several subcortical nuclei such as the thalamus in addition to cortical involvement (Mann and South, 1993; Broe et al., 2003; Schofield et al., 2003). Thus, damage of white matter and subcortical nuclei may play a role in cognitive impairment in FTD. However, both dementias are neuropathologically defined by cortical neuronal changes, thus involvement of white matter is not surprising, because neuronal damage implies damage to the white matter as the white matter reflects the neuronal axons and dendrites that are affected in Alzheimer s disease as well as in FTD. More studies involving novel MRI techniques, such as diffusion imaging that is more sensitive to white matter changes than volumetric MRI are needed to evaluate the difference of white matter and subcortical nuclei between Alzheimer s disease and FTD. In addition, the negative finding between cortical thickness and cognitive function may be due to the small range of MMSE and CDR scores in FTD patients, which makes detecting a relationship between cortical thickness and cognitive functions unreliable. More studies of FTD patients with a broader spread of dementia severity are needed to evaluate a potential relationship between cortical thickness and cognitive functions. The best discriminator between Alzheimer s disease and FTD was parietal lobe atrophy in Alzheimer s disease. This is in agreement with a recent diagnostic MRI study, which reported for a range of pathology confirmed dementia cases that only posterior greater than anterior gradient of atrophy was highly specific for Alzheimer s disease when compared to other dementias, including FTD (Likeman et al., 2005). Overall, however, we found no significant improvement in correctly classifying CN, Alzheimer s disease and FTD from cortical thickness measurement as compared to volumetric measurements of the cortex. We expected that measurements of cortical thinning would be less confounded by underlying white matter atrophy than measurements of cortical volume. Our argument was based on the assumption that the inner surface area of grey matter, which is interfaced with the surface area of white matter, might shrink as a consequence of white matter atrophy or white matter lesions, hence impacting computations of volume measurements more than computations of cortical thickness. The negative outcome could be due to the fact that thickness measurements are intrinsically limited by the finite resolution of MRI, diminishing the advantage of assessing cortical thickness versus cortical volume. Studies at higher magnetic fields that can afford higher image resolution may overcome this limitation. Although cortical thickness and volume measurements provided similar divisions between Alzheimer s disease and FTD, cortical thickness measurement significantly improved the classification between the two diseases based on neuropsychological scores alone. In addition, measurement of cortical thickness provided a similar distinction between Alzheimer s disease and FTD patients than biomarkers such as CSF based tau and isoprostane and better classifications than CSF amyloid beta (1 42), as reported in the previous study (Grossman et al., 2005). Taken together, this suggests that cortical thickness is a useful marker for differentiating the illnesses. A major limitation of this study is that the diagnosis of dementia and its type was made clinically without autopsy confirmation. Therefore, it is possible that some of the patients with FTD had also Alzheimer s disease, vice versa or had other causes of dementia. The inclusion of patients at a more advanced stage of disease may reduce the relevance of our findings to the earliest detection of dementia and differentiation between Alzheimer s disease and FTD. Studies including early stage patients may be more useful in evaluating the clinical diagnostic value of cortical thickness. The relatively young age of patients with Alzheimer s disease, used to match the average age of the patients with FTD, may limit the projection of the findings to an older Alzheimer s disease population. Acknowledgements This work was supported in part by a research applications grant of the Department of Veterans Affairs REAP, NIH grants AG10897, a Program Project Grant from the National Institute of Aging (PO1AG19724), ADRC (P50-AG023501) and a Network grant from the Hillblom Foundation. We also thank Dr Fischl et al. in Massachusetts General Hospital for assistance with use of Freesurfer. References Baron JC, Chetelat G, Desgranges B, et al. In vivo mapping of gray matter loss with voxel-based morphometry in mild Alzheimer s disease. Neuroimage 2001; 14: Braak H, Braak E. Staging of Alzheimer s disease-related neurofibrillary changes. Neurobiol Aging 1995; 16: Braak H, Braak E. Evolution of neuronal changes in the course of Alzheimer s disease. J Neural Transm Suppl 1998; 53: Broe M, Hodges JR, Schofield E, Shepherd CE, Kril JJ, Halliday GM. Staging disease severity in pathologically confirmed cases of frontotemporal dementia. Neurology 2003; 60: Dale AM, Fischl B, Sereno MI. Cortical surface-based analysis. I. Segmentation and surface reconstruction. Neuroimage 1999; 9: DeLong ER, DeLong DM, Clarke-Pearson DL. Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics 1988; 44: Desikan RS, Segonne F, Fischl B, et al. An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. Neuroimage 2006; 31: Dickson DW. Neuropathology of Pick s disease. Neurology 2001; 56: S16 S20. Du AT, Jahng GH, Hayasaka S, et al. Hypoperfusion in frontotemporal dementia and Alzheimer s disease by arterial spin labeling MRI. Neurology 2006; 67: Fischl B, Dale AM. Measuring the thickness of the human cerebral cortex from magnetic resonance images. Proc Natl Acad Sci U S A 2000; 97: Fischl B, Sereno MI, Dale AM. Cortical surface-based analysis. II: inflation, flattening, and a surface-based coordinate system. Neuroimage 1999; 9: Folstein MF, Folstein SE, McHugh PR. Mini-mental state. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 1975; 12:
8 1166 Brain (2007), 130, 1159^1166 A.-T. Du et al. Frisoni GB, Laakso MP, Beltramello A, et al. Hippocampal and entorhinal cortex atrophy in frontotemporal dementia and Alzheimer s disease. Neurology 1999; 52: Gee J, Ding L, Xie Z, Lin M, DeVita C, Grossman M. Alzheimer s disease and frontotemporal dementia exhibit distinct atrophy-behavior correlates: a computer-assisted imaging study. Acad Radiol 2003; 10: Gomez-Isla T, Price JL, McKeel DW Jr, Morris JC, Growdon JH, Hyman BT. Profound loss of layer II entorhinal cortex neurons occurs in very mild Alzheimer s disease. J Neurosci 1996; 16: Grossman M, McMillan C, Moore P, et al. What s in a name: voxel-based morphometric analyses of MRI and naming difficulty in Alzheimer s disease, frontotemporal dementia and corticobasal degeneration. Brain 2004; 127: Grossman M, Farmer J, Leight S, et al. Cerebrospinal fluid profile in frontotemporal dementia and Alzheimer s disease. Ann Neurol 2005; 57: Kersaitis C, Halliday GM, Kril JJ. Regional and cellular pathology in frontotemporal dementia: relationship to stage of disease in cases with and without Pick bodies. Acta Neuropathol (Berl) 2004; 108: Kitagaki H, Mori E, Yamaji S, et al. Frontotemporal dementia and Alzheimer disease: evaluation of cortical atrophy with automated hemispheric surface display generated with MR images. Radiology 1998; 208: Laakso MP, Frisoni GB, Kononen M, et al. Hippocampus and entorhinal cortex in frontotemporal dementia and Alzheimer s disease: a morphometric MRI study. Biol Psychiatr 2000; 47: Lerch JP, Pruessner JC, Zijdenbos A, Hampel H, Teipel SJ, Evans AC. Focal decline of cortical thickness in Alzheimer s disease identified by computational neuroanatomy. Cereb Cortex 2005; 15: Lewis DA, Campbell MJ, Terry RD, Morrison JH. Laminar and regional distributions of neurofibrillary tangles and neuritic plaques in Alzheimer s disease: a quantitative study of visual and auditory cortices. J Neurosci 1987; 7: Likeman M, Anderson VM, Stevens JM, et al. Visual assessment of atrophy on magnetic resonance imaging in the diagnosis of pathologically confirmed young-onset dementias. Arch Neurol 2005; 62: Lipton AM, Benavides R, Hynan LS, et al. Lateralization on neuroimaging does not differentiate frontotemporal lobar degeneration from Alzheimer s disease. Dement Geriatr Cogn Disord 2004; 17: Mann DM, South PW. The topographic distribution of brain atrophy in frontal lobe dementia. Acta Neuropathol (Berl) 1993; 85: McKhann G, Drachman D, Folstein M, Katzman R, Price D, Stadlan EM. Clinical diagnosis of Alzheimer s disease: report of the NINCDS- ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer s Disease. Neurology 1984; 34: Morris JC. The clinical dementia rating (CDR): current version and scoring rules. Neurology 1993; 43: Neary D, Snowden JS, Gustafson L, et al. Frontotemporal lobar degeneration: a consensus on clinical diagnostic criteria. Neurology 1998; 51: Pearson RC, Esiri MM, Hiorns RW, Wilcock GK, Powell TP. Anatomical correlates of the distribution of the pathological changes in the neocortex in Alzheimer disease. Proc Natl Acad Sci USA 1985; 82: Rosen HJ, Gorno-Tempini ML, Goldman WP, et al. Patterns of brain atrophy in frontotemporal dementia and semantic dementia. Neurology 2002; 58: Rosen HJ, Narvaez JM, Hallam B, et al. Neuropsychological and functional measures of severity in Alzheimer disease, frontotemporal dementia, and semantic dementia. Alzheimer Dis Assoc Disord 2004; 18: Rosen HJ, Allison SC, Schauer GF, Gorno-Tempini ML, Weiner MW, Miller BL. Neuroanatomical correlates of behavioural disorders in dementia. Brain 2005; 128: Schofield E, Kersaitis C, Shepherd CE, Kril JJ, Halliday GM. Severity of gliosis in Pick s disease and frontotemporal lobar degeneration: tau-positive glia differentiate these disorders. Brain 2003; 126: Siri S, Benaglio I, Frigerio A, Binetti G, Cappa SF. A brief neuropsychological assessment for the differential diagnosis between frontotemporal dementia and Alzheimer s disease. Eur J Neurol 2001; 8: Whitwell JL, Josephs KA, Rossor MN, et al. Magnetic resonance imaging signatures of tissue pathology in frontotemporal dementia. Arch Neurol 2005; 62: Williams GB, Nestor PJ, Hodges JR. Neural correlates of semantic and behavioural deficits in frontotemporal dementia. Neuroimage 2005; 24:
Neuropathology of Neurodegenerative Disorders Prof. Jillian Kril
 Neurodegenerative disorders to be discussed Alzheimer s disease Lewy body diseases Frontotemporal dementia and other tauopathies Huntington s disease Motor Neuron Disease 2 Neuropathology of neurodegeneration
Neurodegenerative disorders to be discussed Alzheimer s disease Lewy body diseases Frontotemporal dementia and other tauopathies Huntington s disease Motor Neuron Disease 2 Neuropathology of neurodegeneration
Automated detection of abnormal changes in cortical thickness: A tool to help diagnosis in neocortical focal epilepsy
 Automated detection of abnormal changes in cortical thickness: A tool to help diagnosis in neocortical focal epilepsy 1. Introduction Epilepsy is a common neurological disorder, which affects about 1 %
Automated detection of abnormal changes in cortical thickness: A tool to help diagnosis in neocortical focal epilepsy 1. Introduction Epilepsy is a common neurological disorder, which affects about 1 %
ORIGINAL CONTRIBUTION. Deformation-Based Morphometry Reveals Brain Atrophy in Frontotemporal Dementia
 ORIGINAL CONTRIBUTION Deformation-Based Morphometry Reveals Brain Atrophy in Frontotemporal Dementia Valerie A. Cardenas, PhD; Adam L. Boxer, MD, PhD; Linda L. Chao, PhD; Maria L. Gorno-Tempini, MD, PhD;
ORIGINAL CONTRIBUTION Deformation-Based Morphometry Reveals Brain Atrophy in Frontotemporal Dementia Valerie A. Cardenas, PhD; Adam L. Boxer, MD, PhD; Linda L. Chao, PhD; Maria L. Gorno-Tempini, MD, PhD;
Role of TDP-43 in Non-Alzheimer s and Alzheimer s Neurodegenerative Diseases
 Role of TDP-43 in Non-Alzheimer s and Alzheimer s Neurodegenerative Diseases Keith A. Josephs, MD, MST, MSc Professor of Neurology 13th Annual Mild Cognitive Impairment (MCI) Symposium: Alzheimer and Non-Alzheimer
Role of TDP-43 in Non-Alzheimer s and Alzheimer s Neurodegenerative Diseases Keith A. Josephs, MD, MST, MSc Professor of Neurology 13th Annual Mild Cognitive Impairment (MCI) Symposium: Alzheimer and Non-Alzheimer
Published February 2, 2012 as /ajnr.A2935
 Published February 2, 2012 as 10.3174/ajnr.A2935 ORIGINAL RESEARCH H. Matsuda S. Mizumura K. Nemoto F. Yamashita E. Imabayashi N. Sato T. Asada Automatic Voxel-Based Morphometry of Structural MRI by SPM8
Published February 2, 2012 as 10.3174/ajnr.A2935 ORIGINAL RESEARCH H. Matsuda S. Mizumura K. Nemoto F. Yamashita E. Imabayashi N. Sato T. Asada Automatic Voxel-Based Morphometry of Structural MRI by SPM8
Alzheimer s Disease And Frontotemporal Dementia Exhibit Distinct Atrophy Behavior Correlates: A Computer-Assisted Imaging Study 1
 Alzheimer s Disease And Frontotemporal Dementia Exhibit Distinct Atrophy Behavior Correlates: A Computer-Assisted Imaging Study 1 James Gee, PhD, Lijun Ding, PhD, Zhiyong Xie, PhD, Michael Lin, MS, Christian
Alzheimer s Disease And Frontotemporal Dementia Exhibit Distinct Atrophy Behavior Correlates: A Computer-Assisted Imaging Study 1 James Gee, PhD, Lijun Ding, PhD, Zhiyong Xie, PhD, Michael Lin, MS, Christian
Amyotrophic lateral sclerosis (ALS) is a progressive neurodegenerative
 ORIGINAL RESEARCH E. Matsusue S. Sugihara S. Fujii T. Kinoshita T. Nakano E. Ohama T. Ogawa Cerebral Cortical and White Matter Lesions in Amyotrophic Lateral Sclerosis with Dementia: Correlation with MR
ORIGINAL RESEARCH E. Matsusue S. Sugihara S. Fujii T. Kinoshita T. Nakano E. Ohama T. Ogawa Cerebral Cortical and White Matter Lesions in Amyotrophic Lateral Sclerosis with Dementia: Correlation with MR
O Connor 1. Appendix e-1
 O Connor 1 Appendix e-1 Neuropsychiatric assessment The Cambridge Behavioural Inventory Revised (CBI-R) 1, 2 is a proxy behavioural questionnaire that has been extensively used in studies involving FTD
O Connor 1 Appendix e-1 Neuropsychiatric assessment The Cambridge Behavioural Inventory Revised (CBI-R) 1, 2 is a proxy behavioural questionnaire that has been extensively used in studies involving FTD
Fully-automated volumetric MRI with normative ranges: Translation to clinical practice
 Behavioural Neurology 21 (2009) 21 28 21 DOI 10.3233/BEN-2009-0226 IOS Press Fully-automated volumetric MRI with normative ranges: Translation to clinical practice J.B. Brewer Department of Radiology and
Behavioural Neurology 21 (2009) 21 28 21 DOI 10.3233/BEN-2009-0226 IOS Press Fully-automated volumetric MRI with normative ranges: Translation to clinical practice J.B. Brewer Department of Radiology and
Diffusion Tensor Imaging in Dementia. Howard Rosen UCSF Department of Neurology Memory and Aging Center
 Diffusion Tensor Imaging in Dementia Howard Rosen UCSF Department of Neurology Memory and Aging Center www.memory.ucsf.edu Overview Examples of DTI findings in Alzheimer s disease And other dementias Explore
Diffusion Tensor Imaging in Dementia Howard Rosen UCSF Department of Neurology Memory and Aging Center www.memory.ucsf.edu Overview Examples of DTI findings in Alzheimer s disease And other dementias Explore
ORIGINAL CONTRIBUTION
 ORIGINAL CONTRIBUTION Frontotemporal Lobar Degeneration Without Lobar Atrophy Keith A. Josephs, MST, MD; Jennifer L. Whitwell, PhD; Clifford R. Jack, MD; Joseph E. Parisi, MD; Dennis W. Dickson, MD Background:
ORIGINAL CONTRIBUTION Frontotemporal Lobar Degeneration Without Lobar Atrophy Keith A. Josephs, MST, MD; Jennifer L. Whitwell, PhD; Clifford R. Jack, MD; Joseph E. Parisi, MD; Dennis W. Dickson, MD Background:
DISCLOSURES. Objectives. THE EPIDEMIC of 21 st Century. Clinical Assessment of Cognition: New & Emerging Tools for Diagnosing Dementia NONE TO REPORT
 Clinical Assessment of Cognition: New & Emerging Tools for Diagnosing Dementia DISCLOSURES NONE TO REPORT Freddi Segal Gidan, PA, PhD USC Keck School of Medicine Rancho/USC California Alzheimers Disease
Clinical Assessment of Cognition: New & Emerging Tools for Diagnosing Dementia DISCLOSURES NONE TO REPORT Freddi Segal Gidan, PA, PhD USC Keck School of Medicine Rancho/USC California Alzheimers Disease
Supplementary Information Methods Subjects The study was comprised of 84 chronic pain patients with either chronic back pain (CBP) or osteoarthritis
 Supplementary Information Methods Subjects The study was comprised of 84 chronic pain patients with either chronic back pain (CBP) or osteoarthritis (OA). All subjects provided informed consent to procedures
Supplementary Information Methods Subjects The study was comprised of 84 chronic pain patients with either chronic back pain (CBP) or osteoarthritis (OA). All subjects provided informed consent to procedures
Discriminative Analysis for Image-Based Population Comparisons
 Discriminative Analysis for Image-Based Population Comparisons Polina Golland 1,BruceFischl 2, Mona Spiridon 3, Nancy Kanwisher 3, Randy L. Buckner 4, Martha E. Shenton 5, Ron Kikinis 6, and W. Eric L.
Discriminative Analysis for Image-Based Population Comparisons Polina Golland 1,BruceFischl 2, Mona Spiridon 3, Nancy Kanwisher 3, Randy L. Buckner 4, Martha E. Shenton 5, Ron Kikinis 6, and W. Eric L.
Yin-Hui Siow MD, FRCPC Director of Nuclear Medicine Southlake Regional Health Centre
 Yin-Hui Siow MD, FRCPC Director of Nuclear Medicine Southlake Regional Health Centre Today Introduction to CT Introduction to MRI Introduction to nuclear medicine Imaging the dementias The Brain ~ 1.5
Yin-Hui Siow MD, FRCPC Director of Nuclear Medicine Southlake Regional Health Centre Today Introduction to CT Introduction to MRI Introduction to nuclear medicine Imaging the dementias The Brain ~ 1.5
Discriminative Analysis for Image-Based Studies
 Discriminative Analysis for Image-Based Studies Polina Golland 1, Bruce Fischl 2, Mona Spiridon 3, Nancy Kanwisher 3, Randy L. Buckner 4, Martha E. Shenton 5, Ron Kikinis 6, Anders Dale 2, and W. Eric
Discriminative Analysis for Image-Based Studies Polina Golland 1, Bruce Fischl 2, Mona Spiridon 3, Nancy Kanwisher 3, Randy L. Buckner 4, Martha E. Shenton 5, Ron Kikinis 6, Anders Dale 2, and W. Eric
Piano playing skills in a patient with frontotemporal dementia: A longitudinal case study
 International Symposium on Performance Science ISBN 978-94-90306-01-4 The Author 2009, Published by the AEC All rights reserved Piano playing skills in a patient with frontotemporal dementia: A longitudinal
International Symposium on Performance Science ISBN 978-94-90306-01-4 The Author 2009, Published by the AEC All rights reserved Piano playing skills in a patient with frontotemporal dementia: A longitudinal
SUPPLEMENTARY INFORMATION In format provided by Frank et al. (JULY 2010)
 Table 1 Imaging bios for Alzheimer s Visual rating High correlation with Multicenter studies have Accuracy for longitudinal hippocampus volume (R 2 been performed, but changes only at chance about 0.9,
Table 1 Imaging bios for Alzheimer s Visual rating High correlation with Multicenter studies have Accuracy for longitudinal hippocampus volume (R 2 been performed, but changes only at chance about 0.9,
fmri and Voxel-based Morphometry in Detection of Early Stages of Alzheimer's Disease
 fmri and Voxel-based Morphometry in Detection of Early Stages of Alzheimer's Disease Andrey V. Sokolov 1,3, Sergey V. Vorobyev 2, Aleksandr Yu. Efimtcev 1,3, Viacheslav S. Dekan 1,3, Gennadiy E. Trufanov
fmri and Voxel-based Morphometry in Detection of Early Stages of Alzheimer's Disease Andrey V. Sokolov 1,3, Sergey V. Vorobyev 2, Aleksandr Yu. Efimtcev 1,3, Viacheslav S. Dekan 1,3, Gennadiy E. Trufanov
Visual Rating Scale Reference Material. Lorna Harper Dementia Research Centre University College London
 Visual Rating Scale Reference Material Lorna Harper Dementia Research Centre University College London Background The reference materials included in this document were compiled and used in relation to
Visual Rating Scale Reference Material Lorna Harper Dementia Research Centre University College London Background The reference materials included in this document were compiled and used in relation to
Structural And Functional Integration: Why all imaging requires you to be a structural imager. David H. Salat
 Structural And Functional Integration: Why all imaging requires you to be a structural imager David H. Salat salat@nmr.mgh.harvard.edu Salat:StructFunct:HST.583:2015 Structural Information is Critical
Structural And Functional Integration: Why all imaging requires you to be a structural imager David H. Salat salat@nmr.mgh.harvard.edu Salat:StructFunct:HST.583:2015 Structural Information is Critical
Quantitative Neuroimaging- Gray and white matter Alteration in Multiple Sclerosis. Lior Or-Bach Instructors: Prof. Anat Achiron Dr.
 Quantitative Neuroimaging- Gray and white matter Alteration in Multiple Sclerosis Lior Or-Bach Instructors: Prof. Anat Achiron Dr. Shmulik Miron INTRODUCTION Multiple Sclerosis general background Gray
Quantitative Neuroimaging- Gray and white matter Alteration in Multiple Sclerosis Lior Or-Bach Instructors: Prof. Anat Achiron Dr. Shmulik Miron INTRODUCTION Multiple Sclerosis general background Gray
Round table: Moderator; Fereshteh Sedaghat, MD, PhD Brain Mapping in Dementias and Non-invasive Neurostimulation
 Round table: Moderator; Fereshteh Sedaghat, MD, PhD Brain Mapping in Dementias and Non-invasive Neurostimulation 1. Reflection of Mild Cognitive Impairment (MCI) and Dementias by Molecular Imaging, PET
Round table: Moderator; Fereshteh Sedaghat, MD, PhD Brain Mapping in Dementias and Non-invasive Neurostimulation 1. Reflection of Mild Cognitive Impairment (MCI) and Dementias by Molecular Imaging, PET
MRI of Pathological Aging Brain
 MRI of Pathological Aging Brain Yukio Miki Department of Radiology, Osaka City University A variety of pathological changes occur in the brain with aging, and many of these changes can be identified by
MRI of Pathological Aging Brain Yukio Miki Department of Radiology, Osaka City University A variety of pathological changes occur in the brain with aging, and many of these changes can be identified by
review of existing studies on ASL in dementia Marion Smits, MD PhD
 review of existing studies on ASL in dementia Marion Smits, MD PhD Associate Professor of Neuroradiology Department of Radiology, Erasmus MC, Rotterdam (NL) Alzheimer Centre South-West Netherlands, Rotterdam
review of existing studies on ASL in dementia Marion Smits, MD PhD Associate Professor of Neuroradiology Department of Radiology, Erasmus MC, Rotterdam (NL) Alzheimer Centre South-West Netherlands, Rotterdam
Imaging of Alzheimer s Disease: State of the Art
 July 2015 Imaging of Alzheimer s Disease: State of the Art Neir Eshel, Harvard Medical School Year IV Outline Our patient Definition of dementia Alzheimer s disease Epidemiology Diagnosis Stages of progression
July 2015 Imaging of Alzheimer s Disease: State of the Art Neir Eshel, Harvard Medical School Year IV Outline Our patient Definition of dementia Alzheimer s disease Epidemiology Diagnosis Stages of progression
Dementia. Stephen S. Flitman, MD Medical Director 21st Century Neurology
 Dementia Stephen S. Flitman, MD Medical Director 21st Century Neurology www.neurozone.org Dementia is a syndrome Progressive memory loss, plus Progressive loss of one or more cognitive functions: Language
Dementia Stephen S. Flitman, MD Medical Director 21st Century Neurology www.neurozone.org Dementia is a syndrome Progressive memory loss, plus Progressive loss of one or more cognitive functions: Language
Neuro degenerative PET image from FDG, amyloid to Tau
 Neuro degenerative PET image from FDG, amyloid to Tau Kun Ju Lin ( ) MD, Ph.D Department of Nuclear Medicine and Molecular Imaging Center, Chang Gung Memorial Hospital ( ) Department of Medical Imaging
Neuro degenerative PET image from FDG, amyloid to Tau Kun Ju Lin ( ) MD, Ph.D Department of Nuclear Medicine and Molecular Imaging Center, Chang Gung Memorial Hospital ( ) Department of Medical Imaging
FRONTOTEMPORAL DEGENERATION: OVERVIEW, TRENDS AND DEVELOPMENTS
 FRONTOTEMPORAL DEGENERATION: OVERVIEW, TRENDS AND DEVELOPMENTS Norman L. Foster, M.D. Director, Center for Alzheimer s Care, Imaging and Research Chief, Division of Cognitive Neurology, Department of Neurology
FRONTOTEMPORAL DEGENERATION: OVERVIEW, TRENDS AND DEVELOPMENTS Norman L. Foster, M.D. Director, Center for Alzheimer s Care, Imaging and Research Chief, Division of Cognitive Neurology, Department of Neurology
Differential diagnosis of Frontotemporal Dementia FTLD using visual rating scales
 Differential diagnosis of Frontotemporal Dementia FTLD using visual rating scales Poster No.: C-0491 Congress: ECR 2016 Type: Scientific Exhibit Authors: S. Manouvelou 1, G. ANYFANTAKIS 2, V. Koutoulidis
Differential diagnosis of Frontotemporal Dementia FTLD using visual rating scales Poster No.: C-0491 Congress: ECR 2016 Type: Scientific Exhibit Authors: S. Manouvelou 1, G. ANYFANTAKIS 2, V. Koutoulidis
Automated Whole Brain Segmentation Using FreeSurfer
 Automated Whole Brain Segmentation Using FreeSurfer https://surfer.nmr.mgh.harvard.edu/ FreeSurfer (FS) is a free software package developed at the Martinos Center for Biomedical Imaging used for three
Automated Whole Brain Segmentation Using FreeSurfer https://surfer.nmr.mgh.harvard.edu/ FreeSurfer (FS) is a free software package developed at the Martinos Center for Biomedical Imaging used for three
Cortical hypoperfusion in Parkinson's disease assessed with arterial spin labeling MRI
 Cortical hypoperfusion in Parkinson's disease assessed with arterial spin labeling MRI Poster No.: C-0609 Congress: ECR 2013 Type: Scientific Exhibit Authors: S. Aoki, K. Kamagata, Y. Motoi, K. Kamiya,
Cortical hypoperfusion in Parkinson's disease assessed with arterial spin labeling MRI Poster No.: C-0609 Congress: ECR 2013 Type: Scientific Exhibit Authors: S. Aoki, K. Kamagata, Y. Motoi, K. Kamiya,
ORIGINAL CONTRIBUTION. Regional Distribution of Neuritic Plaques in the Nondemented Elderly and Subjects With Very Mild Alzheimer Disease
 ORIGINAL CONTRIBUTION Regional Distribution of Neuritic Plaques in the Nondemented Elderly and Subjects With Very Mild Alzheimer Disease Vahram Haroutunian, PhD; Daniel P. Perl, MD; Dushyant P. Purohit,
ORIGINAL CONTRIBUTION Regional Distribution of Neuritic Plaques in the Nondemented Elderly and Subjects With Very Mild Alzheimer Disease Vahram Haroutunian, PhD; Daniel P. Perl, MD; Dushyant P. Purohit,
Mild Cognitive Impairment (MCI)
 October 19, 2018 Mild Cognitive Impairment (MCI) Yonas E. Geda, MD, MSc Professor of Neurology and Psychiatry Consultant, Departments of Psychiatry & Psychology, and Neurology Mayo Clinic College of Medicine
October 19, 2018 Mild Cognitive Impairment (MCI) Yonas E. Geda, MD, MSc Professor of Neurology and Psychiatry Consultant, Departments of Psychiatry & Psychology, and Neurology Mayo Clinic College of Medicine
Supplementary Online Content
 Supplementary Online Content Gregg NM, Kim AE, Gurol ME, et al. Incidental cerebral microbleeds and cerebral blood flow in elderly individuals. JAMA Neurol. Published online July 13, 2015. doi:10.1001/jamaneurol.2015.1359.
Supplementary Online Content Gregg NM, Kim AE, Gurol ME, et al. Incidental cerebral microbleeds and cerebral blood flow in elderly individuals. JAMA Neurol. Published online July 13, 2015. doi:10.1001/jamaneurol.2015.1359.
SWI including phase and magnitude images
 On-line Table: MRI imaging recommendation and summary of key features Sequence Pathologies Visible Key Features T1 volumetric high-resolution whole-brain reformatted in axial, coronal, and sagittal planes
On-line Table: MRI imaging recommendation and summary of key features Sequence Pathologies Visible Key Features T1 volumetric high-resolution whole-brain reformatted in axial, coronal, and sagittal planes
Diagnosis before NIA AA The impact of FDG PET in. Diagnosis after NIA AA Neuropathology and PET image 2015/10/16
 The impact of FDG PET in degenerative dementia diagnosis Jung Lung, Hsu MD, Ph.D (Utrecht) Section of dementia and cognitive impairment Department of Neurology Chang Gung Memorial Hospital, Linkou, Taipei
The impact of FDG PET in degenerative dementia diagnosis Jung Lung, Hsu MD, Ph.D (Utrecht) Section of dementia and cognitive impairment Department of Neurology Chang Gung Memorial Hospital, Linkou, Taipei
FDG-PET e parkinsonismi
 Parkinsonismi FDG-PET e parkinsonismi Valentina Berti Dipartimento di Scienze Biomediche, Sperimentali e Cliniche Sez. Medicina Nucleare Università degli Studi di Firenze History 140 PubMed: FDG AND parkinsonism
Parkinsonismi FDG-PET e parkinsonismi Valentina Berti Dipartimento di Scienze Biomediche, Sperimentali e Cliniche Sez. Medicina Nucleare Università degli Studi di Firenze History 140 PubMed: FDG AND parkinsonism
Assessing Brain Volumes Using MorphoBox Prototype
 MAGNETOM Flash (68) 2/207 33 Assessing Brain Volumes Using MorphoBox Prototype Alexis Roche,2,3 ; Bénédicte Maréchal,2,3 ; Tobias Kober,2,3 ; Gunnar Krueger 4 ; Patric Hagmann ; Philippe Maeder ; Reto
MAGNETOM Flash (68) 2/207 33 Assessing Brain Volumes Using MorphoBox Prototype Alexis Roche,2,3 ; Bénédicte Maréchal,2,3 ; Tobias Kober,2,3 ; Gunnar Krueger 4 ; Patric Hagmann ; Philippe Maeder ; Reto
Frontotemporal dementia and dementia with Lewy bodies in a case-control study of Alzheimer s disease
 International Psychogeriatrics: page 1 of 8 C 2009 International Psychogeriatric Association doi:10.1017/s1041610209009454 Frontotemporal dementia and dementia with Lewy bodies in a case-control study
International Psychogeriatrics: page 1 of 8 C 2009 International Psychogeriatric Association doi:10.1017/s1041610209009454 Frontotemporal dementia and dementia with Lewy bodies in a case-control study
Characterizing Anatomical Variability And Alzheimer s Disease Related Cortical Thinning in the Medial Temporal Lobe
 Characterizing Anatomical Variability And Alzheimer s Disease Related Cortical Thinning in the Medial Temporal Lobe Long Xie, Laura Wisse, Sandhitsu Das, Ranjit Ittyerah, Jiancong Wang, David Wolk, Paul
Characterizing Anatomical Variability And Alzheimer s Disease Related Cortical Thinning in the Medial Temporal Lobe Long Xie, Laura Wisse, Sandhitsu Das, Ranjit Ittyerah, Jiancong Wang, David Wolk, Paul
Neuroanatomical correlates of depressive symptoms in newly diagnosed Parkinson s disease patients
 Neurology Asia 2018; 23(3) : 239 244 Neuroanatomical correlates of depressive symptoms in newly diagnosed Parkinson s disease patients * 1,2 Seong-il Oh MD, * 1,2 Eun Joo Chung MD, 3 Song-I Chun PhD, 3
Neurology Asia 2018; 23(3) : 239 244 Neuroanatomical correlates of depressive symptoms in newly diagnosed Parkinson s disease patients * 1,2 Seong-il Oh MD, * 1,2 Eun Joo Chung MD, 3 Song-I Chun PhD, 3
Baseline Characteristics of Patients Attending the Memory Clinic Serving the South Shore of Boston
 Article ID: ISSN 2046-1690 Baseline Characteristics of Patients Attending the www.thealzcenter.org Memory Clinic Serving the South Shore of Boston Corresponding Author: Dr. Anil K Nair, Chief of Neurology,
Article ID: ISSN 2046-1690 Baseline Characteristics of Patients Attending the www.thealzcenter.org Memory Clinic Serving the South Shore of Boston Corresponding Author: Dr. Anil K Nair, Chief of Neurology,
Advanced multimodal imaging in malformations of cortical development
 Advanced multimodal imaging in malformations of cortical development Seok Jun Hong (sjhong@bic.mni.mcgill.ca) NOEL Neuroimaging of Epilepsy Lab MICA Multimodal Imaging and Connectome Analysis Lab w4 w5
Advanced multimodal imaging in malformations of cortical development Seok Jun Hong (sjhong@bic.mni.mcgill.ca) NOEL Neuroimaging of Epilepsy Lab MICA Multimodal Imaging and Connectome Analysis Lab w4 w5
Brain imaging for the diagnosis of people with suspected dementia
 Why do we undertake brain imaging in dementia? Brain imaging for the diagnosis of people with suspected dementia Not just because guidelines tell us to! Exclude other causes for dementia Help confirm diagnosis
Why do we undertake brain imaging in dementia? Brain imaging for the diagnosis of people with suspected dementia Not just because guidelines tell us to! Exclude other causes for dementia Help confirm diagnosis
Estimating the Validity of the Korean Version of Expanded Clinical Dementia Rating (CDR) Scale
 Estimating the Validity of the Korean Version of Expanded Clinical Dementia Rating (CDR) Scale Seong Hye Choi, M.D.*, Duk L. Na, M.D., Byung Hwa Lee, M.A., Dong-Seog Hahm, M.D., Jee Hyang Jeong, M.D.,
Estimating the Validity of the Korean Version of Expanded Clinical Dementia Rating (CDR) Scale Seong Hye Choi, M.D.*, Duk L. Na, M.D., Byung Hwa Lee, M.A., Dong-Seog Hahm, M.D., Jee Hyang Jeong, M.D.,
Clinicopathologic and genetic aspects of hippocampal sclerosis. Dennis W. Dickson, MD Mayo Clinic, Jacksonville, Florida USA
 Clinicopathologic and genetic aspects of hippocampal sclerosis Dennis W. Dickson, MD Mayo Clinic, Jacksonville, Florida USA The hippocampus in health & disease A major structure of the medial temporal
Clinicopathologic and genetic aspects of hippocampal sclerosis Dennis W. Dickson, MD Mayo Clinic, Jacksonville, Florida USA The hippocampus in health & disease A major structure of the medial temporal
ORIGINAL CONTRIBUTION. Application of Automated Medial Temporal Lobe Atrophy Scale to Alzheimer Disease
 ORIGINAL CONTRIBUTION Application of Automated Medial Temporal Lobe Atrophy Scale to Alzheimer Disease Basil H. Ridha, MRCP; Josephine Barnes, MA, PhD; Laura A. van de Pol, MD; Jonathan M. Schott, MD,
ORIGINAL CONTRIBUTION Application of Automated Medial Temporal Lobe Atrophy Scale to Alzheimer Disease Basil H. Ridha, MRCP; Josephine Barnes, MA, PhD; Laura A. van de Pol, MD; Jonathan M. Schott, MD,
Alzheimer disease is the most common cause of dementia in
 ORIGINAL RESEARCH BRAIN Automated Segmentation of Hippocampal Subfields in Drug-Naïve Patients with Alzheimer Disease H.K. Lim, S.C. Hong, W.S. Jung, K.J. Ahn, W.Y. Won, C. Hahn, I.S. Kim, and C.U. Lee
ORIGINAL RESEARCH BRAIN Automated Segmentation of Hippocampal Subfields in Drug-Naïve Patients with Alzheimer Disease H.K. Lim, S.C. Hong, W.S. Jung, K.J. Ahn, W.Y. Won, C. Hahn, I.S. Kim, and C.U. Lee
ORIGINAL CONTRIBUTION. Neurofibrillary Tangles in Nondemented Elderly Subjects and Mild Alzheimer Disease
 ORIGINAL CONTRIBUTION Neurofibrillary Tangles in Nondemented Elderly Subjects and Mild Alzheimer Disease Vahram Haroutunian, PhD; Dushyant P. Purohit, MD; Daniel P. Perl, MD; Deborah Marin, MD; Khalid
ORIGINAL CONTRIBUTION Neurofibrillary Tangles in Nondemented Elderly Subjects and Mild Alzheimer Disease Vahram Haroutunian, PhD; Dushyant P. Purohit, MD; Daniel P. Perl, MD; Deborah Marin, MD; Khalid
Neuro-Imaging in dementia: using MRI in routine work-up Prof. Philip Scheltens
 Neuro-Imaging in dementia: Philip Scheltens Alzheimer Center VU University Medical Center Amsterdam The Netherlands 1 Outline of talk Current guidelines Imaging used to exclude disease Specific patterns
Neuro-Imaging in dementia: Philip Scheltens Alzheimer Center VU University Medical Center Amsterdam The Netherlands 1 Outline of talk Current guidelines Imaging used to exclude disease Specific patterns
Cover Page. The handle holds various files of this Leiden University dissertation
 Cover Page The handle http://hdl.handle.net/1887/39720 holds various files of this Leiden University dissertation Author: Hafkemeijer, Anne Title: Brain networks in aging and dementia Issue Date: 2016-05-26
Cover Page The handle http://hdl.handle.net/1887/39720 holds various files of this Leiden University dissertation Author: Hafkemeijer, Anne Title: Brain networks in aging and dementia Issue Date: 2016-05-26
Structural MRI in Frontotemporal Dementia: Comparisons between Hippocampal Volumetry, Tensor- Based Morphometry and Voxel-Based Morphometry
 : Comparisons between Hippocampal Volumetry, Tensor- Based Morphometry and Voxel-Based Morphometry Miguel Ángel Muñoz-Ruiz 1,Päivi Hartikainen 1,2, Juha Koikkalainen 3, Robin Wolz 4, Valtteri Julkunen
: Comparisons between Hippocampal Volumetry, Tensor- Based Morphometry and Voxel-Based Morphometry Miguel Ángel Muñoz-Ruiz 1,Päivi Hartikainen 1,2, Juha Koikkalainen 3, Robin Wolz 4, Valtteri Julkunen
White matter hyperintensities correlate with neuropsychiatric manifestations of Alzheimer s disease and frontotemporal lobar degeneration
 White matter hyperintensities correlate with neuropsychiatric manifestations of Alzheimer s disease and frontotemporal lobar degeneration Annual Scientific Meeting Canadian Geriatric Society Philippe Desmarais,
White matter hyperintensities correlate with neuropsychiatric manifestations of Alzheimer s disease and frontotemporal lobar degeneration Annual Scientific Meeting Canadian Geriatric Society Philippe Desmarais,
DEMENTIA 101: WHAT IS HAPPENING IN THE BRAIN? Philip L. Rambo, PhD
 DEMENTIA 101: WHAT IS HAPPENING IN THE BRAIN? Philip L. Rambo, PhD OBJECTIVES Terminology/Dementia Basics Most Common Types Defining features Neuro-anatomical/pathological underpinnings Neuro-cognitive
DEMENTIA 101: WHAT IS HAPPENING IN THE BRAIN? Philip L. Rambo, PhD OBJECTIVES Terminology/Dementia Basics Most Common Types Defining features Neuro-anatomical/pathological underpinnings Neuro-cognitive
Is DTI Increasing the Connectivity Between the Magnet Suite and the Clinic?
 Current Literature In Clinical Science Is DTI Increasing the Connectivity Between the Magnet Suite and the Clinic? Spatial Patterns of Water Diffusion Along White Matter Tracts in Temporal Lobe Epilepsy.
Current Literature In Clinical Science Is DTI Increasing the Connectivity Between the Magnet Suite and the Clinic? Spatial Patterns of Water Diffusion Along White Matter Tracts in Temporal Lobe Epilepsy.
Funding: NIDCF UL1 DE019583, NIA RL1 AG032119, NINDS RL1 NS062412, NIDA TL1 DA
 The Effect of Cognitive Functioning, Age, and Molecular Variables on Brain Structure Among Carriers of the Fragile X Premutation: Deformation Based Morphometry Study Naomi J. Goodrich-Hunsaker*, Ling M.
The Effect of Cognitive Functioning, Age, and Molecular Variables on Brain Structure Among Carriers of the Fragile X Premutation: Deformation Based Morphometry Study Naomi J. Goodrich-Hunsaker*, Ling M.
Anosognosia, or loss of insight into one s cognitive
 REGULAR ARTICLES Anosognosia Is a Significant Predictor of Apathy in Alzheimer s Disease Sergio E. Starkstein, M.D., Ph.D. Simone Brockman, M.A. David Bruce, M.D. Gustavo Petracca, M.D. Anosognosia and
REGULAR ARTICLES Anosognosia Is a Significant Predictor of Apathy in Alzheimer s Disease Sergio E. Starkstein, M.D., Ph.D. Simone Brockman, M.A. David Bruce, M.D. Gustavo Petracca, M.D. Anosognosia and
DOI: /brain/awh075 Brain (2004), 127, 628±649
 DOI: 10.1093/brain/awh075 Brain (2004), 127, 628±649 What's in a name: voxel-based morphometric analyses of MRI and naming dif culty in Alzheimer's disease, frontotemporal dementia and corticobasal degeneration
DOI: 10.1093/brain/awh075 Brain (2004), 127, 628±649 What's in a name: voxel-based morphometric analyses of MRI and naming dif culty in Alzheimer's disease, frontotemporal dementia and corticobasal degeneration
Differentiation of semantic dementia and Alzheimer s disease using the Addenbrooke s Cognitive Examination (ACE)
 INTERNATIONAL JOURNAL OF GERIATRIC PSYCHIATRY Int J Geriatr Psychiatry 2008; 23: 370 375. Published online 4 September 2007 in Wiley InterScience (www.interscience.wiley.com).1887 Differentiation of semantic
INTERNATIONAL JOURNAL OF GERIATRIC PSYCHIATRY Int J Geriatr Psychiatry 2008; 23: 370 375. Published online 4 September 2007 in Wiley InterScience (www.interscience.wiley.com).1887 Differentiation of semantic
Neuroimaging for Diagnosis of Psychiatric Disorders
 Psychiatric Disorder Neuroimaging for Diagnosis of Psychiatric Disorders JMAJ 45(12): 538 544, 2002 Yoshio HIRAYASU Associate Professor, Department of Neuropsychiatry Kyorin University School of Medicine
Psychiatric Disorder Neuroimaging for Diagnosis of Psychiatric Disorders JMAJ 45(12): 538 544, 2002 Yoshio HIRAYASU Associate Professor, Department of Neuropsychiatry Kyorin University School of Medicine
Washington University: Setting the Stage for Secondary Prevention Trials in Alzheimer Disease
 Washington University: Setting the Stage for Secondary Prevention Trials in Alzheimer Disease John C. Morris, MD Harvey A. and Dorismae Hacker Friedman Distinguished Professor of Neurology Disclosure Statement
Washington University: Setting the Stage for Secondary Prevention Trials in Alzheimer Disease John C. Morris, MD Harvey A. and Dorismae Hacker Friedman Distinguished Professor of Neurology Disclosure Statement
Diffusion Tensor Imaging in Psychiatry
 2003 KHBM DTI in Psychiatry Diffusion Tensor Imaging in Psychiatry KHBM 2003. 11. 21. 서울대학교 의과대학 정신과학교실 권준수 Neuropsychiatric conditions DTI has been studied in Alzheimer s disease Schizophrenia Alcoholism
2003 KHBM DTI in Psychiatry Diffusion Tensor Imaging in Psychiatry KHBM 2003. 11. 21. 서울대학교 의과대학 정신과학교실 권준수 Neuropsychiatric conditions DTI has been studied in Alzheimer s disease Schizophrenia Alcoholism
Neural Correlates of Episodic Memory in Behavioral Variant Frontotemporal Dementia
 Journal of Alzheimer s Disease 24 (2011) 261 268 DOI 10.3233/JAD-2010-101668 IOS Press 261 Neural Correlates of Episodic Memory in Behavioral Variant Frontotemporal Dementia Catherine Pennington a, John
Journal of Alzheimer s Disease 24 (2011) 261 268 DOI 10.3233/JAD-2010-101668 IOS Press 261 Neural Correlates of Episodic Memory in Behavioral Variant Frontotemporal Dementia Catherine Pennington a, John
CISC 3250 Systems Neuroscience
 CISC 3250 Systems Neuroscience Levels of organization Central Nervous System 1m 10 11 neurons Neural systems and neuroanatomy Systems 10cm Networks 1mm Neurons 100μm 10 8 neurons Professor Daniel Leeds
CISC 3250 Systems Neuroscience Levels of organization Central Nervous System 1m 10 11 neurons Neural systems and neuroanatomy Systems 10cm Networks 1mm Neurons 100μm 10 8 neurons Professor Daniel Leeds
How preserved is episodic memory in behavioral variant frontotemporal dementia?
 How preserved is episodic memory in behavioral variant frontotemporal dementia? M. Hornberger, PhD O. Piguet, PhD A.J. Graham, PhD, MRCP P.J. Nestor, MD, FRACP J.R. Hodges, MD, FRCP Address correspondence
How preserved is episodic memory in behavioral variant frontotemporal dementia? M. Hornberger, PhD O. Piguet, PhD A.J. Graham, PhD, MRCP P.J. Nestor, MD, FRACP J.R. Hodges, MD, FRCP Address correspondence
Neuroimaging for dementia diagnosis. Guidance from the London Dementia Clinical Network
 Neuroimaging for dementia diagnosis Guidance from the London Dementia Clinical Network Authors Dr Stephen Orleans-Foli Consultant Psychiatrist, West London Mental Health NHS Trust Dr Jeremy Isaacs Consultant
Neuroimaging for dementia diagnosis Guidance from the London Dementia Clinical Network Authors Dr Stephen Orleans-Foli Consultant Psychiatrist, West London Mental Health NHS Trust Dr Jeremy Isaacs Consultant
Brain gray matter volume changes associated with motor symptoms in patients with Parkinson s disease
 Kang et al. Chinese Neurosurgical Journal (2015) 1:9 DOI 10.1186/s41016-015-0003-6 RESEARCH Open Access Brain gray matter volume changes associated with motor symptoms in patients with Parkinson s disease
Kang et al. Chinese Neurosurgical Journal (2015) 1:9 DOI 10.1186/s41016-015-0003-6 RESEARCH Open Access Brain gray matter volume changes associated with motor symptoms in patients with Parkinson s disease
Cover Page. The handle holds various files of this Leiden University dissertation
 Cover Page The handle http://hdl.handle.net/1887/26921 holds various files of this Leiden University dissertation Author: Doan, Nhat Trung Title: Quantitative analysis of human brain MR images at ultrahigh
Cover Page The handle http://hdl.handle.net/1887/26921 holds various files of this Leiden University dissertation Author: Doan, Nhat Trung Title: Quantitative analysis of human brain MR images at ultrahigh
Procedia - Social and Behavioral Sciences 159 ( 2014 ) WCPCG 2014
 Available online at www.sciencedirect.com ScienceDirect Procedia - Social and Behavioral Sciences 159 ( 2014 ) 743 748 WCPCG 2014 Differences in Visuospatial Cognition Performance and Regional Brain Activation
Available online at www.sciencedirect.com ScienceDirect Procedia - Social and Behavioral Sciences 159 ( 2014 ) 743 748 WCPCG 2014 Differences in Visuospatial Cognition Performance and Regional Brain Activation
Supplementary Online Content
 Supplementary Online Content Hooshmand B, Magialasche F, Kalpouzos G, et al. Association of vitamin B, folate, and sulfur amino acids with brain magnetic resonance imaging measures in older adults: a longitudinal
Supplementary Online Content Hooshmand B, Magialasche F, Kalpouzos G, et al. Association of vitamin B, folate, and sulfur amino acids with brain magnetic resonance imaging measures in older adults: a longitudinal
Investigating the impact of midlife obesity on Alzheimer s disease (AD) pathology in a mouse model of AD
 Brain@McGill Prize for Neuroscience Undergraduate Research Colleen Rollins Supervisor: Dr. Mallar Chakravarty Revised: August 8, 2017 Investigating the impact of midlife obesity on Alzheimer s disease
Brain@McGill Prize for Neuroscience Undergraduate Research Colleen Rollins Supervisor: Dr. Mallar Chakravarty Revised: August 8, 2017 Investigating the impact of midlife obesity on Alzheimer s disease
Regulatory Challenges across Dementia Subtypes European View
 Regulatory Challenges across Dementia Subtypes European View Population definition including Early disease at risk Endpoints in POC studies Endpoints in pivotal trials 1 Disclaimer No CoI The opinions
Regulatory Challenges across Dementia Subtypes European View Population definition including Early disease at risk Endpoints in POC studies Endpoints in pivotal trials 1 Disclaimer No CoI The opinions
Dementia mimicking Alzheimer s disease Owing to a tau mutation: CSF and PET findings
 Dementia mimicking Alzheimer s disease Owing to a tau mutation: CSF and PET findings Chapter 4.2 N. Tolboom E.L.G.E. Koedam J.M. Schott M. Yaqub M.A. Blankenstein F. Barkhof Y.A.L. Pijnenburg A.A. Lammertsma
Dementia mimicking Alzheimer s disease Owing to a tau mutation: CSF and PET findings Chapter 4.2 N. Tolboom E.L.G.E. Koedam J.M. Schott M. Yaqub M.A. Blankenstein F. Barkhof Y.A.L. Pijnenburg A.A. Lammertsma
Hallucinations and conscious access to visual inputs in Parkinson s disease
 Supplemental informations Hallucinations and conscious access to visual inputs in Parkinson s disease Stéphanie Lefebvre, PhD^1,2, Guillaume Baille, MD^4, Renaud Jardri MD, PhD 1,2 Lucie Plomhause, PhD
Supplemental informations Hallucinations and conscious access to visual inputs in Parkinson s disease Stéphanie Lefebvre, PhD^1,2, Guillaume Baille, MD^4, Renaud Jardri MD, PhD 1,2 Lucie Plomhause, PhD
Classification and Statistical Analysis of Auditory FMRI Data Using Linear Discriminative Analysis and Quadratic Discriminative Analysis
 International Journal of Innovative Research in Computer Science & Technology (IJIRCST) ISSN: 2347-5552, Volume-2, Issue-6, November-2014 Classification and Statistical Analysis of Auditory FMRI Data Using
International Journal of Innovative Research in Computer Science & Technology (IJIRCST) ISSN: 2347-5552, Volume-2, Issue-6, November-2014 Classification and Statistical Analysis of Auditory FMRI Data Using
Right and Left Medial Orbitofrontal Volumes Show an Opposite Relationship to Agreeableness in FTD
 Dement Geriatr Cogn Disord 2004;17:328 332 DOI: 10.1159/000077165 Right and Left Medial Orbitofrontal Volumes Show an Opposite Relationship to Agreeableness in FTD Katherine P. Rankin a Howard J. Rosen
Dement Geriatr Cogn Disord 2004;17:328 332 DOI: 10.1159/000077165 Right and Left Medial Orbitofrontal Volumes Show an Opposite Relationship to Agreeableness in FTD Katherine P. Rankin a Howard J. Rosen
Assessing Cognition. Andreana Haley, Ph.D. Department of Psychology University of Texas at Austin
 Assessing Cognition Andreana Haley, Ph.D. Department of Psychology University of Texas at Austin Agenda Topic: Cognitive Aging Assessing Cognition Diagnosing Cognitive Impairment Dementia Diagnoses Symptoms
Assessing Cognition Andreana Haley, Ph.D. Department of Psychology University of Texas at Austin Agenda Topic: Cognitive Aging Assessing Cognition Diagnosing Cognitive Impairment Dementia Diagnoses Symptoms
Voxel-based morphometry in clinical neurosciences
 Voxel-based morphometry in clinical neurosciences Ph.D. Thesis Ádám Feldmann Department of Behavioural Sciences Leader of Doctoral School: Prof. Dr.Sámuel Komoly, D.Sc. Program leader: Prof. Dr.Sámuel
Voxel-based morphometry in clinical neurosciences Ph.D. Thesis Ádám Feldmann Department of Behavioural Sciences Leader of Doctoral School: Prof. Dr.Sámuel Komoly, D.Sc. Program leader: Prof. Dr.Sámuel
Detection of Mild Cognitive Impairment using Image Differences and Clinical Features
 Detection of Mild Cognitive Impairment using Image Differences and Clinical Features L I N L I S C H O O L O F C O M P U T I N G C L E M S O N U N I V E R S I T Y Copyright notice Many of the images in
Detection of Mild Cognitive Impairment using Image Differences and Clinical Features L I N L I S C H O O L O F C O M P U T I N G C L E M S O N U N I V E R S I T Y Copyright notice Many of the images in
A new rapid landmark-based regional MRI segmentation method of the brain
 Journal of the Neurological Sciences 194 (2002) 35 40 www.elsevier.com/locate/jns A new rapid landmark-based regional MRI segmentation method of the brain A.L.W. Bokde a, S.J. Teipel a, *, Y. Zebuhr a,
Journal of the Neurological Sciences 194 (2002) 35 40 www.elsevier.com/locate/jns A new rapid landmark-based regional MRI segmentation method of the brain A.L.W. Bokde a, S.J. Teipel a, *, Y. Zebuhr a,
Caring Sheet #11: Alzheimer s Disease:
 CARING SHEETS: Caring Sheet #11: Alzheimer s Disease: A Summary of Information and Intervention Suggestions with an Emphasis on Cognition By Shelly E. Weaverdyck, PhD Introduction This caring sheet focuses
CARING SHEETS: Caring Sheet #11: Alzheimer s Disease: A Summary of Information and Intervention Suggestions with an Emphasis on Cognition By Shelly E. Weaverdyck, PhD Introduction This caring sheet focuses
Four Tissue Segmentation in ADNI II
 Four Tissue Segmentation in ADNI II Charles DeCarli, MD, Pauline Maillard, PhD, Evan Fletcher, PhD Department of Neurology and Center for Neuroscience, University of California at Davis Summary Table of
Four Tissue Segmentation in ADNI II Charles DeCarli, MD, Pauline Maillard, PhD, Evan Fletcher, PhD Department of Neurology and Center for Neuroscience, University of California at Davis Summary Table of
The current state of healthcare for Normal Aging, Mild Cognitive Impairment, & Alzheimer s Disease
 The current state of healthcare for Normal Aging, g, Mild Cognitive Impairment, & Alzheimer s Disease William Rodman Shankle, MS MD FACP Director, Alzheimer s Program, Hoag Neurosciences Institute Neurologist,
The current state of healthcare for Normal Aging, g, Mild Cognitive Impairment, & Alzheimer s Disease William Rodman Shankle, MS MD FACP Director, Alzheimer s Program, Hoag Neurosciences Institute Neurologist,
FDG-PET improves accuracy in distinguishing frontotemporal dementia and Alzheimer s disease
 doi:10.1093/brain/awm177 Brain (2007), 130,2616^2635 FDG-PET improves accuracy in distinguishing frontotemporal dementia and Alzheimer s disease Norman L. Foster, 1 Judith L. Heidebrink, 2,4 Christopher
doi:10.1093/brain/awm177 Brain (2007), 130,2616^2635 FDG-PET improves accuracy in distinguishing frontotemporal dementia and Alzheimer s disease Norman L. Foster, 1 Judith L. Heidebrink, 2,4 Christopher
Department of Radiology, Weill Cornell Medical College, 525 East 68th Street, New York, NY 10065, USA 3
 Neurology Research International Volume 2012, Article ID 718150, 6 pages doi:10.1155/2012/718150 Research Article A Pilot Study of Quantitative MRI Measurements of Ventricular Volume and Cortical Atrophy
Neurology Research International Volume 2012, Article ID 718150, 6 pages doi:10.1155/2012/718150 Research Article A Pilot Study of Quantitative MRI Measurements of Ventricular Volume and Cortical Atrophy
CHAPTER 5 NEUROPSYCHOLOGICAL PROFILE OF ALZHEIMER S DISEASE
 CHAPTER 5 NEUROPSYCHOLOGICAL PROFILE OF ALZHEIMER S DISEASE 5.1 GENERAL BACKGROUND Neuropsychological assessment plays a crucial role in the assessment of cognitive decline in older age. In India, there
CHAPTER 5 NEUROPSYCHOLOGICAL PROFILE OF ALZHEIMER S DISEASE 5.1 GENERAL BACKGROUND Neuropsychological assessment plays a crucial role in the assessment of cognitive decline in older age. In India, there
Human Brain Myelination from Birth to 4.5 Years
 Human Brain Myelination from Birth to 4.5 Years B. Aubert-Broche, V. Fonov, I. Leppert, G.B. Pike, and D.L. Collins Montreal Neurological Institute, McGill University, Montreal, Canada Abstract. The myelination
Human Brain Myelination from Birth to 4.5 Years B. Aubert-Broche, V. Fonov, I. Leppert, G.B. Pike, and D.L. Collins Montreal Neurological Institute, McGill University, Montreal, Canada Abstract. The myelination
Form D1: Clinician Diagnosis
 Initial Visit Packet Form D: Clinician Diagnosis NACC Uniform Data Set (UDS) ADC name: Subject ID: Form date: / / Visit #: Examiner s initials: INSTRUCTIONS: This form is to be completed by the clinician.
Initial Visit Packet Form D: Clinician Diagnosis NACC Uniform Data Set (UDS) ADC name: Subject ID: Form date: / / Visit #: Examiner s initials: INSTRUCTIONS: This form is to be completed by the clinician.
Update on functional brain imaging in Movement Disorders
 Update on functional brain imaging in Movement Disorders Mario Masellis, MSc, MD, FRCPC, PhD Assistant Professor & Clinician-Scientist Sunnybrook Health Sciences Centre University of Toronto 53 rd CNSF
Update on functional brain imaging in Movement Disorders Mario Masellis, MSc, MD, FRCPC, PhD Assistant Professor & Clinician-Scientist Sunnybrook Health Sciences Centre University of Toronto 53 rd CNSF
P. Hitchcock, Ph.D. Department of Cell and Developmental Biology Kellogg Eye Center. Wednesday, 16 March 2009, 1:00p.m. 2:00p.m.
 Normal CNS, Special Senses, Head and Neck TOPIC: CEREBRAL HEMISPHERES FACULTY: LECTURE: READING: P. Hitchcock, Ph.D. Department of Cell and Developmental Biology Kellogg Eye Center Wednesday, 16 March
Normal CNS, Special Senses, Head and Neck TOPIC: CEREBRAL HEMISPHERES FACULTY: LECTURE: READING: P. Hitchcock, Ph.D. Department of Cell and Developmental Biology Kellogg Eye Center Wednesday, 16 March
Supplementary Online Content
 Supplementary Online Content Redlich R, Opel N, Grotegerd D, et al. Prediction of individual response to electroconvulsive therapy via machine learning on structural magnetic resonance imaging data. JAMA
Supplementary Online Content Redlich R, Opel N, Grotegerd D, et al. Prediction of individual response to electroconvulsive therapy via machine learning on structural magnetic resonance imaging data. JAMA
Brain tissue and white matter lesion volume analysis in diabetes mellitus type 2
 Brain tissue and white matter lesion volume analysis in diabetes mellitus type 2 C. Jongen J. van der Grond L.J. Kappelle G.J. Biessels M.A. Viergever J.P.W. Pluim On behalf of the Utrecht Diabetic Encephalopathy
Brain tissue and white matter lesion volume analysis in diabetes mellitus type 2 C. Jongen J. van der Grond L.J. Kappelle G.J. Biessels M.A. Viergever J.P.W. Pluim On behalf of the Utrecht Diabetic Encephalopathy
Introduction, use of imaging and current guidelines. John O Brien Professor of Old Age Psychiatry University of Cambridge
 Introduction, use of imaging and current guidelines John O Brien Professor of Old Age Psychiatry University of Cambridge Why do we undertake brain imaging in AD and other dementias? Exclude other causes
Introduction, use of imaging and current guidelines John O Brien Professor of Old Age Psychiatry University of Cambridge Why do we undertake brain imaging in AD and other dementias? Exclude other causes
CASE 49. What type of memory is available for conscious retrieval? Which part of the brain stores semantic (factual) memories?
 CASE 49 A 43-year-old woman is brought to her primary care physician by her family because of concerns about her forgetfulness. The patient has a history of Down syndrome but no other medical problems.
CASE 49 A 43-year-old woman is brought to her primary care physician by her family because of concerns about her forgetfulness. The patient has a history of Down syndrome but no other medical problems.
Online appendices are unedited and posted as supplied by the authors. SUPPLEMENTARY MATERIAL
 Appendix 1 to Sehmbi M, Rowley CD, Minuzzi L, et al. Age-related deficits in intracortical myelination in young adults with bipolar SUPPLEMENTARY MATERIAL Supplementary Methods Intracortical Myelin (ICM)
Appendix 1 to Sehmbi M, Rowley CD, Minuzzi L, et al. Age-related deficits in intracortical myelination in young adults with bipolar SUPPLEMENTARY MATERIAL Supplementary Methods Intracortical Myelin (ICM)
Differential contributions of subregions of medial temporal lobe to memory system in. amnestic mild cognitive impairment: insights from fmri study
 Differential contributions of subregions of medial temporal lobe to memory system in amnestic mild cognitive impairment: insights from fmri study Jiu Chen 1, *, Xujun Duan 2, *, Hao Shu 1, Zan Wang 1,
Differential contributions of subregions of medial temporal lobe to memory system in amnestic mild cognitive impairment: insights from fmri study Jiu Chen 1, *, Xujun Duan 2, *, Hao Shu 1, Zan Wang 1,
ORIGINAL CONTRIBUTION. Comparison of the Short Test of Mental Status and the Mini-Mental State Examination in Mild Cognitive Impairment
 ORIGINAL CONTRIBUTION Comparison of the Short Test of Mental Status and the Mini-Mental State Examination in Mild Cognitive Impairment David F. Tang-Wai, MDCM; David S. Knopman, MD; Yonas E. Geda, MD;
ORIGINAL CONTRIBUTION Comparison of the Short Test of Mental Status and the Mini-Mental State Examination in Mild Cognitive Impairment David F. Tang-Wai, MDCM; David S. Knopman, MD; Yonas E. Geda, MD;
Visualization strategies for major white matter tracts identified by diffusion tensor imaging for intraoperative use
 International Congress Series 1281 (2005) 793 797 www.ics-elsevier.com Visualization strategies for major white matter tracts identified by diffusion tensor imaging for intraoperative use Ch. Nimsky a,b,
International Congress Series 1281 (2005) 793 797 www.ics-elsevier.com Visualization strategies for major white matter tracts identified by diffusion tensor imaging for intraoperative use Ch. Nimsky a,b,
Twelve right-handed subjects between the ages of 22 and 30 were recruited from the
 Supplementary Methods Materials & Methods Subjects Twelve right-handed subjects between the ages of 22 and 30 were recruited from the Dartmouth community. All subjects were native speakers of English,
Supplementary Methods Materials & Methods Subjects Twelve right-handed subjects between the ages of 22 and 30 were recruited from the Dartmouth community. All subjects were native speakers of English,
