EEA JCDs - overview. Period starts at: Period ends at: 08/04/ /04/ April Subcommittee 1 EEA JCD. Adoption Date. Confirmed EIF Date
|
|
|
- Lambert Richards
- 5 years ago
- Views:
Transcription
1 s - overview Overview of adopted EEA Joint Committee decisions. This report provides detailed information on; number, adoption date, confirmed entry into force date, placement in the EEA Agreement, publication in the Official Journal and the celex number of the incorporated acts. Period starts at: Period ends at: 08/04/ /04/ April 2014 Subcommittee 1 EIF 1 033/ /04/ /04/2014 Annex I Veterinary and Phytosanitary Matters Publication Annex / Protocol Celex Title Compliance EU 2 034/ /04/ /04/2014 Annex I Veterinary and Phytosanitary Matters 32013D0076 Commission Implementing Decision 2013/76/EU of 4 February 2013 amending Decision 2009/719/EC authorising certain Member States to revise their annual BSE monitoring programmes 26/02/ R R R R1078 Commission Implementing Regulation (EU) 1040/2013 of 24 October 2013 concerning the authorisation of a preparation of endo-1,4-beta-xylanase produced by Trichoderma reesei (MUCL 49755) and endo-1,3(4)-beta-glucanase produced by Trichoderma reesei (MUCL 49754) as a feed additive for pigs for fattening and minor porcine species for fattening other than Sus scrofa domesticus and turkeys for fattening (holder of authorisation Aveve NV) Commission Implementing Regulation (EU) 1055/2013 of 25 October 2013 concerning the authorisation of a preparation of orthophosphoric acid as a feed additive for all animal species Commission Implementing Regulation (EU) 1060/2013 of 29 October 2013 concerning the authorisation of bentonite as a feed additive for all animal species Commission Implementing Regulation (EU) 1078/2013 of 31 October 2013 concerning the authorisation of fumaric acid as a feed additive for all animal species 32013R1113 Commission Implementing Regulation (EU) 1113/2013 of 7 vember 2013 concerning the authorisation of preparations of Lactobacillus plantarum NCIMB 40027, Lactobacillus buchneri DSM 22501, Lactobacillus buchneri NCIMB 40788/CNCM I-4323, Lactobacillus buchneri LN 40177/ATCC PTA-6138, and Lactobacillus buchneri LN 4637/ATCC PTA as feed additives for all animal species 19/11/ /11/ /11/ /11/2013 1
2 EIF Publication Annex / Protocol Celex Title Compliance EU 32013R1222 Commission Implementing Regulation (EU) 1222/2013 of 29 vember 2013 concerning the authorisation of propionic acid, sodium propionate and ammonium propionate as feed additives for ruminants, pigs and poultry 20/12/ / /04/ /04/2014 Annex I Veterinary and Phytosanitary Matters 4 036/ /04/ /04/2014 Annex II Technical Regulations, 32012L0001 Commission Implementing Directive 2012/1/EU of 6 January 2012 amending Annex I to Council Directive 66/402/EEC as regards the conditions to be satisfied by the crop Oryza sativa 32013L0057 Commission Implementing Directive 2013/57/EU of 20 vember 2013 amending Directives 2003/90/EC and 2003/91/EC setting out implementing measures for the purposes of Article 7 of Council Directive 2002/53/EC and Article 7 of Council Directive 2002/55/EC respectively, as regards the characteristics to be covered as a minimum by the examination and the minimum conditions for examining certain varieties of agricultural plant species and vegetable species 13/01/ /07/ R0480 Commission Implementing Regulation (EU) 480/2013 of 24 May 2013 amending Implementing Regulation (EU) 788/2012 as regards the period of analysis of certain pesticides performed on a voluntary basis 32013R0772 Commission Regulation (EU) 772/2013 of 8 August 2013 amending Annexes II, III and V to Regulation (EC) 396/2005 of the European Parliament and of the Council as regards maximum residue levels for diphenylamine in or on certain products 32013R0777 Commission Regulation (EU) 777/2013 of 12 August 2013 amending Annexes II, III and V to Regulation (EC) 396/2005 of the European Parliament and of the Council as regards maximum residue levels for clodinafop, clomazone, diuron, ethalfluralin, ioxynil, iprovalicarb, maleic hydrazide, mepanipyrim, metconazole, prosulfocarb and tepraloxydim in or on certain products 32013R0834 Commission Regulation (EU) 834/2013 of 30 August 2013 amending Annexes II and III to Regulation (EC) 396/2005 of the European Parliament and of the Council as regards maximum residue levels for acequinocyl, bixafen, diazinon, difenoconazole, etoxazole, fenhexamid, fludioxonil, isopyrazam, lambda-cyhalothrin, profenofos and prothioconazole in or on certain products 16/06/ /03/ /03/ /09/2013 2
3 EIF 5 037/ /04/ /04/2014 Annex II Technical Regulations, Publication Annex / Protocol Celex Title Compliance EU 32013R0985 Commission Regulation (EU) 985/2013 of 14 October 2013 amending and correcting Annex I to Regulation (EC) 1334/2008 of the European Parliament and of the Council as regards certain flavouring substances 04/11/ / /04/ /04/2014 Annex II Technical Regulations, 32013R0195 Commission Regulation (EU) 195/2013 of 7 March 2013 amending Directive 2007/46/EC of the European Parliament and of the Council and Commission Regulation (EC) 692/2008 as concerns innovative technologies for reducing CO2 emissions from light passenger and commercial vehicles 29/03/ / /04/ /04/2014 Annex II Technical Regulations, 32013R0817 Commission Regulation (EU) 817/2013 of 28 August 2013 amending Annexes II and III to Regulation (EC) 1333/2008 of the European Parliament and of the Council and the Annex to Commission Regulation (EU) 231/2012 as regards Octenyl succinic acid modified gum arabic 32013R0818 Commission Regulation (EU) 818/2013 of 28 August 2013 amending Annex III to Regulation (EC) 1333/2008 of the European Parliament and of the Council as regards the use of Sucrose esters of fatty acids (E 473) in flavourings for water based clear flavoured drinks 32013R0851 Commission Regulation (EU) 851/2013 of 3 September 2013 authorising certain health claims made on foods, other than those referring to the reduction of disease risk and to children s development and health and amending Regulation (EU) 432/ /09/ /09/ /09/ R0489 Commission Implementing Regulation (EU) 489/2013 of 27 May 2013 amending the Annex to Regulation (EU) 37/2010 on pharmacologically active substances and their classification regarding maximum residue limits in foodstuffs of animal origin, as regards the substance double stranded ribonucleic acid homologous to viral ribonucleic acid coding for part of the coat protein and part of the intergenic region of the Israel Acute Paralysis Virus 18/06/2013 3
4 EIF 8 040/ /04/ /04/2014 Annex II Technical Regulations, Publication Annex / Protocol Celex Title Compliance EU 9 041/ /04/ /04/2014 Annex II Technical Regulations, 32013D0301 Commission Implementing Decision 2013/301/EU of 11 June 2013 amending Implementing Decision 2012/715/EU establishing a list of third countries with a regulatory framework applicable to active substances for medicinal products for human use and the respective control and enforcement activities ensuring a level of protection of public health equivalent to that in the Union 16/06/ / /04/ /04/2014 Annex II Technical Regulations, 32013H0172 Commission Recommendation of 5 April 2013 on a common framework for a unique device identification system of medical devices in the Union 32013R0198 Commission Implementing Regulation (EU) 198/2013 of 7 March 2013 on the selection of a symbol for the purpose of identifying medicinal products for human use that are subject to additional monitoring 32013R0220 Commission Regulation (EU) 220/2013 of 13 March 2013 amending Council Regulation (EC) 297/95 as regards the adjustment of the fees of the European Medicines Agency to the inflation rate 29/03/ /04/ / /04/2014 Annex II Technical Regulations, 32013R0463 Commission Regulation (EU) 463/2013 of 17 May 2013 amending Regulation (EC) 2003/2003 of the European Parliament and of the Council relating to fertilisers for the purposes of adapting Annexes I, II and IV thereto to technical progress 16/06/ R0837 Commission Delegated Regulation (EU) 837/2013 of 25 June 2013 amending Annex III to Regulation (EU) 528/2012 of the European Parliament and of the Council as regards the information requirements for authorisation of biocidal products 32013R0945 Commission Implementing Regulation (EU) 945/2013 of 2 October 2013 to approve cypermethrin as an existing active substance for use in biocidal products for product-type R0955 Commission Implementing Regulation (EU) 955/2013 of 4 October 2013 to approve propiconazole as an existing active substance for use in biocidal products for product-type 9 23/09/ /10/ /10/2013 4
5 EIF / /04/2014 Annex II Technical Regulations, Publication Annex / Protocol Celex Title Compliance EU 32013R R R R R R R R1039 Commission Implementing Regulation (EU) 1032/2013 of 24 October 2013 approving bromoacetic acid as an existing active substance for use in biocidal products for product-type 4 Commission Implementing Regulation (EU) 1033/2013 of 24 October 2013 approving copper sulphate pentahydrate as an existing active substance for use in biocidal products for product-type 2 Commission Implementing Regulation (EU) 1034/2013 of 24 October 2013 approving aluminium phosphide releasing phosphine as an active substance for use in biocidal products for product type 20 Commission Implementing Regulation (EU) 1035/2013 of 24 October 2013 approving benzoic acid as an existing active substance for use in biocidal products for product-types 3 and 4 Commission Implementing Regulation (EU) 1036/2013 of 24 October 2013 approving etofenprox as an existing active substance for use in biocidal products for product-type 18 Commission Implementing Regulation (EU) 1037/2013 of 24 October 2013 approving IPBC as an existing active substance for use in biocidal products for product-type 6 Commission Implementing Regulation (EU) 1038/2013 of 24 October 2013 approving tebuconazole as an existing active substance for use in biocidal products for product-types 7 and 10 Commission Implementing Regulation (EU) 1039/2013 of 24 October 2013 modifying the approval of nonanoic acid as an existing active substance for use in biocidal products for product-type R0354 Commission Implementing Regulation (EU) 354/2013 of 18 April 2013 on changes of biocidal products authorised in accordance with Regulation (EU) 528/2012 of the European Parliament and of the Council 32013R0564 Commission Implementing Regulation (EU) 564/2013 of 18 June 2013 on the fees and charges payable to the European Chemicals Agency pursuant to Regulation (EU) 528/2012 of the European Parliament and of the Council concerning the making available on the market and use of biocidal products 08/05/ /07/2013 5
6 EIF / /04/ /04/2014 Annex II Technical Regulations, Publication Annex / Protocol Celex Title Compliance EU / /04/ /04/2014 Annex II Technical Regulations, 32013R0758 Commission Regulation (EU) 758/2013 of 7 August 2013 correcting Annex VI to Regulation (EC) 1272/2008 of the European Parliament and of the Council on classification, labelling and packaging of substances and mixtures 32013R0944 Commission Regulation (EU) 944/2013 of 2 October 2013 amending, for the purposes of its adaptation to technical and scientific progress, Regulation (EC) 1272/2008 of the European Parliament and of the Council on classification, labelling and packaging of substances and mixtures 13/08/ /10/ / /04/ /04/2014 Annex II Technical Regulations, 32013D R1197 Commission Implementing Decision 674/2013 of 25 vember 2013 on Guidelines on Annex I to Regulation (EC) 1223/2009 of the European Parliament and of the Council on cosmetic products Commission Regulation (EU) 1197/2013 of 25 vember 2013 amending Annex III to Regulation (EC) 1223/2009 of the European Parliament and of the Council on cosmetic products 16/12/ /12/2013 Subcommittee 1: 15 JCDs, 42 Acts. Subcommittee / /04/ /04/2014 Annex XIII Transport 32013L0052 Commission Directive 2013/52/EU of 30 October 2013 amending Council Directive 96/98/EC on marine equipment 04/12/ R R0886 COMMISSION DELEGATED REGULATION (EU) 885/2013 of 15 May 2013 supplementing ITS Directive 2010/40/EU of the European Parliament and of the Council with regard to the provision of information services for safe and secure parking places for trucks and commercial vehicles COMMISSION DELEGATED REGULATION (EU) 886/2013 of 15 May 2013 supplementing Directive 2010/40/EU of the European Parliament and of the Council with regard to data and procedures for the provision, where possible, of road safety-related minimum universal traffic information free of charge to users 01/10/ /10/2013 6
7 EIF / /04/ /04/2014 Annex XIII Transport Publication Annex / Protocol Celex Title Compliance EU / /04/ /04/2014 Annex XIII Transport 32013L0049 Commission Directive 2013/49/EU of 11 October 2013 amending Annex II to Directive 2006/87/EC of the European Parliament and of the Council laying down technical requirements for inland waterway vessels 32013R0909 Commission Implementing Regulation (EU) 909/2013 of 10 September 2013 on the technical specifications for the electronic chart display and information system for inland navigation (Inland ECDIS) referred to in Directive 2005/44/EC of the European Parliament and of the Council 03/11/ /03/2016 Subcommittee 2: 3 JCDs, 5 Acts. Subcommittee / /04/ /04/2014 Annex VI Social Security 32013R1264 Commission Implementing Regulation (EU) 1264/2013 of 3 December 2013 amending Regulation (EC) 474/2006 establishing the Community list of air carriers which are subject to an operating ban within the Community 07/12/2013 Subcommittee 3: 1 JCDs, 1 Acts. Subcommittee / /04/2014 Annex XX Environment 52013XX0927 (01) Administrative Commission for the Coordination of Social Security Systems Decision S9 of 20 June 2013 concerning refund procedures for the implementation of Articles 35 and 41 of Regulation (EC) 883/ /09/ D0641 Commission Decision 2013/641/EU of 7 vember 2013 establishing the ecological criteria for the award of the EU Ecolabel for flushing toilets and urinals 32013R0782 Commission Regulation (EU) 782/2013 of 14 August 2013 amending Annex III to Regulation (EU) 66/2010 of the European Parliament and of the Council on the EU Ecolabel 09/11/ /09/2013 7
8 EIF / /04/ /04/2014 Annex XX Environment Protocol 02 on products excluded from the scope of the Agreement in accordance with Article 8(3)(a) Publication Annex / Protocol Celex Title Compliance EU / /04/ /04/2014 Annex XXI Statistics 32013R0815 Commission Regulation (EU) 815/2013 of 27 August 2013 amending Regulation (EC) 748/2009 on the list of aircraft operators that performed an aviation activity listed in Annex I to Directive 2003/87/EC of the European Parliament and of the Council on or after 1 January 2006 specifying the administering Member State for each aircraft operator to take into consideration the accession of Croatia to the European Union 32014R0100 Commission Regulation (EU) 100/2014 of 5 February 2014 amending Regulation (EC) 748/2009 on the list of aircraft operators that performed an aviation activity listed in Annex I to Directive 2003/87/EC of the European Parliament and of the Council on or after 1 January 2006 specifying the administering Member State for each aircraft operator 07/09/ /02/2014 Subcommittee 4: 3 JCDs, 5 Acts R0549 Regulation (EU) 549/2013 of the European Parliament and of the Council of 21 May 2013 on the European system of national and regional accounts in the European Union 16/07/2013 Total number of JCDs : 22 Total number of Acts : 53 8
EEA JCDs - overview 08/11/ /11/2013. Period starts at: Period ends at: 08 November Subcommittee 1. Confirmed EIF Date
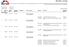 s - overview Overview of adopted EEA Joint Committee decisions. This report provides detailed information on; number, adoption date, confirmed entry into force date, placement in the EEA Agreement, publication
s - overview Overview of adopted EEA Joint Committee decisions. This report provides detailed information on; number, adoption date, confirmed entry into force date, placement in the EEA Agreement, publication
5.1 EEA JCDs - Overview of adopted JCDs
 5.1 s - Overview of adopted JCDs Overview of adopted EEA Joint Committee Decisions and EU acts contained therein. The report by default shows EEA Joint Committee Decisions adopted during the period chosen,
5.1 s - Overview of adopted JCDs Overview of adopted EEA Joint Committee Decisions and EU acts contained therein. The report by default shows EEA Joint Committee Decisions adopted during the period chosen,
Section Animal Nutrition 24 SEPTEMBER SEPTEMBER 2012 AGENDA
 EUROPEAN COMMISSION HEALTH & CONSUMERS DIRECTORATE-GENERAL SANCO G Ares (2012) 1108941 STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH Section Animal Nutrition 24 SEPTEMBER 2012-25 SEPTEMBER 2012
EUROPEAN COMMISSION HEALTH & CONSUMERS DIRECTORATE-GENERAL SANCO G Ares (2012) 1108941 STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH Section Animal Nutrition 24 SEPTEMBER 2012-25 SEPTEMBER 2012
EUROPEAN COMMISSION. STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH Section Animal Nutrition 16 DECEMBER DECEMBER 2013 AGENDA
 EUROPEAN COMMISSION HEALTH AND CONSUMERS DIRECTORATE-GENERAL SANCO G (2013) 3790404 STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH Section Animal Nutrition 16 DECEMBER 2013-17 DECEMBER 2013 AGENDA
EUROPEAN COMMISSION HEALTH AND CONSUMERS DIRECTORATE-GENERAL SANCO G (2013) 3790404 STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH Section Animal Nutrition 16 DECEMBER 2013-17 DECEMBER 2013 AGENDA
Delegations will find attached document D042445/02.
 Council of the European Union Brussels, 11 December 2015 (OR. en) 15273/15 DLEG 164 AGRI 670 SAN 437 COVER NOTE From: European Commission date of receipt: 11 December 2015 To: No. Cion doc.: D042445/02
Council of the European Union Brussels, 11 December 2015 (OR. en) 15273/15 DLEG 164 AGRI 670 SAN 437 COVER NOTE From: European Commission date of receipt: 11 December 2015 To: No. Cion doc.: D042445/02
COMMISSION DELEGATED REGULATION (EU).../... of XXX
 EUROPEAN COMMISSION Brussels, XXX SANTE/10355/2015 (POOL/E4/2015/10355/10355-EN. doc) [...1(2015) XXX draft COMMISSION DELEGATED REGULATION (EU).../... of XXX supplementmg Regulation (EU) No 609/2013 of
EUROPEAN COMMISSION Brussels, XXX SANTE/10355/2015 (POOL/E4/2015/10355/10355-EN. doc) [...1(2015) XXX draft COMMISSION DELEGATED REGULATION (EU).../... of XXX supplementmg Regulation (EU) No 609/2013 of
REPORT FROM THE COMMISSION TO THE EUROPEAN PARLIAMENT AND THE COUNCIL
 EUROPEAN COMMISSION Brussels, 22.8.2017 COM(2017) 438 final REPORT FROM THE COMMISSION TO THE EUROPEAN PARLIAMENT AND THE COUNCIL on the exercise of the delegation conferred on the Commission pursuant
EUROPEAN COMMISSION Brussels, 22.8.2017 COM(2017) 438 final REPORT FROM THE COMMISSION TO THE EUROPEAN PARLIAMENT AND THE COUNCIL on the exercise of the delegation conferred on the Commission pursuant
GLP in the European Union Ecolabel detergents, GLP and accreditation
 GLP in the European Union Ecolabel detergents, GLP and accreditation Maik Schmahl Brussels, 25/03/2010 Chemicals Unit Outline What is GLP? How has it developed? The role of the Member States, the European
GLP in the European Union Ecolabel detergents, GLP and accreditation Maik Schmahl Brussels, 25/03/2010 Chemicals Unit Outline What is GLP? How has it developed? The role of the Member States, the European
EUROPEAN COMMISSION. Standing Committee on Plants, Animals, Food and Feed Section Animal Nutrition 27 JUNE JUNE 2018 AGENDA
 EUROPEAN COMMISSION Health and Food Safety Directorate General sante.ddg2.g.5(2018)2959871 Standing Committee on Plants, Animals, Food and Feed Section Animal Nutrition 27 JUNE 2018-29 JUNE 2018 CIRCABC
EUROPEAN COMMISSION Health and Food Safety Directorate General sante.ddg2.g.5(2018)2959871 Standing Committee on Plants, Animals, Food and Feed Section Animal Nutrition 27 JUNE 2018-29 JUNE 2018 CIRCABC
FoodDrinkEurope Position on GLP studies
 FoodDrinkEurope Position on GLP studies Content of the presentation Introduction to GLP Importance of GLP for the food industry Use of GLP based on the example of novel foods Conclusions GLP what is it?
FoodDrinkEurope Position on GLP studies Content of the presentation Introduction to GLP Importance of GLP for the food industry Use of GLP based on the example of novel foods Conclusions GLP what is it?
EUROPEAN COMMISSION. Standing Committee on Plants, Animals, Food and Feed Section Animal Nutrition 14 NOVEMBER NOVEMBER 2016 AGENDA
 EUROPEAN COMMISSION HEALTH AND FOOD SAFETY DIRECTORATE-GENERAL Ares (2016) 6549653 Standing Committee on Plants, Animals, Food and Feed Section Animal Nutrition 14 NOVEMBER 2016-15 NOVEMBER 2016 CIRCABC
EUROPEAN COMMISSION HEALTH AND FOOD SAFETY DIRECTORATE-GENERAL Ares (2016) 6549653 Standing Committee on Plants, Animals, Food and Feed Section Animal Nutrition 14 NOVEMBER 2016-15 NOVEMBER 2016 CIRCABC
HEAVY METALS RESTRICTIONS OF EU ELV DIRECTIVE. STATUS OF ACTUAL ANNEX II REVISIONS.
 Department, date HEAVY METALS RESTRICTIONS OF EU ELV DIRECTIVE. STATUS OF ACTUAL ANNEX II REVISIONS. CLEPA Material Regulation Conference 2017, 17. May 2017, Reutlingen R. Hoock, BMW Group on behalf of
Department, date HEAVY METALS RESTRICTIONS OF EU ELV DIRECTIVE. STATUS OF ACTUAL ANNEX II REVISIONS. CLEPA Material Regulation Conference 2017, 17. May 2017, Reutlingen R. Hoock, BMW Group on behalf of
COMMISSION IMPLEMENTING DECISION. of
 EUROPEAN COMMISSION Brussels, 4.7.2017 C(2017) 4462 final COMMISSION IMPLEMENTING DECISION of 4.7.2017 on the identification of bis(2-ethylhexyl) phthalate (DEHP), dibutyl phthalate (DBP), benzyl butyl
EUROPEAN COMMISSION Brussels, 4.7.2017 C(2017) 4462 final COMMISSION IMPLEMENTING DECISION of 4.7.2017 on the identification of bis(2-ethylhexyl) phthalate (DEHP), dibutyl phthalate (DBP), benzyl butyl
COMMISSION REGULATION (EU) / of XXX
 Ref. Ares(2016)5616438-28/09/2016 EUROPEAN COMMISSION Brussels, XXX [ ](2016) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annex III to Directive 2008/98/EC of the European Parliament and of
Ref. Ares(2016)5616438-28/09/2016 EUROPEAN COMMISSION Brussels, XXX [ ](2016) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annex III to Directive 2008/98/EC of the European Parliament and of
Official Journal of the European Union REGULATIONS
 L 259/2 REGULATIONS COMMISSION DELEGATED REGULATION (EU) 2017/1798 of 2 June 2017 supplementing Regulation (EU) No 609/2013 of the European Parliament and of the Council as regards the specific compositional
L 259/2 REGULATIONS COMMISSION DELEGATED REGULATION (EU) 2017/1798 of 2 June 2017 supplementing Regulation (EU) No 609/2013 of the European Parliament and of the Council as regards the specific compositional
EUROPEAN COMMISSION SUMMARY REPORT OF THE STANDING COMMITTEE ON PLANTS, ANIMALS, FOOD AND FEED HELD IN BRUSSELS ON 10 FEBRUARY 2015
 EUROPEAN COMMISSION HEALTH AND FOOD SAFETY DIRECTORATE-GENERAL Brussels sante.ddg2.g.dir(2015)934853 SUMMARY REPORT OF THE STANDING COMMITTEE ON PLANTS, ANIMALS, FOOD AND FEED HELD IN BRUSSELS ON 10 FEBRUARY
EUROPEAN COMMISSION HEALTH AND FOOD SAFETY DIRECTORATE-GENERAL Brussels sante.ddg2.g.dir(2015)934853 SUMMARY REPORT OF THE STANDING COMMITTEE ON PLANTS, ANIMALS, FOOD AND FEED HELD IN BRUSSELS ON 10 FEBRUARY
COMMISSION REGULATION (EU) No /.. of XXX
 EUROPEAN COMMISSION Brussels, XXX SANCO/11214/2014 (POOL/E3/2014/11214/11214-EN.doc) D036822/04 [ ](2014) XXX draft COMMISSION REGULATION (EU) No /.. of XXX amending and correcting Annexes II and III to
EUROPEAN COMMISSION Brussels, XXX SANCO/11214/2014 (POOL/E3/2014/11214/11214-EN.doc) D036822/04 [ ](2014) XXX draft COMMISSION REGULATION (EU) No /.. of XXX amending and correcting Annexes II and III to
HEALTH & CONSUMERS DIRECTORATE-GENERAL
 EUROPEAN COMMISSION HEALTH & CONSUMERS DIRECTORATE-GENERAL Ares (2011) 1223178 SUMMARY RECORD OF THE STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH HELD ON 21 OCTOBER 2011 IN BRUSSELS (Animal Nutrition
EUROPEAN COMMISSION HEALTH & CONSUMERS DIRECTORATE-GENERAL Ares (2011) 1223178 SUMMARY RECORD OF THE STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH HELD ON 21 OCTOBER 2011 IN BRUSSELS (Animal Nutrition
SUMMARY RECORD OF THE STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH HELD ON FEBRUARY 2012 IN BRUSSELS (Animal Nutrition Section)
 EUROPEAN COMMISSION HEALTH & CONSUMERS DIRECTORATE-GENERAL Ares (2012) 195747 SUMMARY RECORD OF THE STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH HELD ON 16-17 FEBRUARY 2012 IN BRUSSELS (Animal
EUROPEAN COMMISSION HEALTH & CONSUMERS DIRECTORATE-GENERAL Ares (2012) 195747 SUMMARY RECORD OF THE STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH HELD ON 16-17 FEBRUARY 2012 IN BRUSSELS (Animal
COMMISSION REGULATION (EU) / of XXX
 EUROPEAN COMMISSION Brussels, XXX SANTE/10893/2018 Rev. 1 [ ](2018) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annexes II, III, IV and V to Regulation (EC) No 396/2005 of the European Parliament
EUROPEAN COMMISSION Brussels, XXX SANTE/10893/2018 Rev. 1 [ ](2018) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annexes II, III, IV and V to Regulation (EC) No 396/2005 of the European Parliament
Official Journal of the European Union
 23.2.2018 EN L 53/69 COMMISSION IMPLEMENTING REGULATION (EU) 2018/243 of 15 February 2018 concerning the authorisation of 3-hydroxybutan-2-one, pentan-2,3-dione, 3,5-dimethyl cyclopentan-1,2-dione, hexan-3,4-dione,
23.2.2018 EN L 53/69 COMMISSION IMPLEMENTING REGULATION (EU) 2018/243 of 15 February 2018 concerning the authorisation of 3-hydroxybutan-2-one, pentan-2,3-dione, 3,5-dimethyl cyclopentan-1,2-dione, hexan-3,4-dione,
L 108/2 Official Journal of the European Union (8) Potassium chromate meets the criteria for classification as carcinogenic and mutagenic Re
 18.4.2013 Official Journal of the European Union L 108/1 II (Non-legislative acts) REGULATIONS COMMISSION REGULATION (EU) No 348/2013 of 17 April 2013 amending Annex XIV to Regulation (EC) No 1907/2006
18.4.2013 Official Journal of the European Union L 108/1 II (Non-legislative acts) REGULATIONS COMMISSION REGULATION (EU) No 348/2013 of 17 April 2013 amending Annex XIV to Regulation (EC) No 1907/2006
COMMISSION IMPLEMENTING REGULATION (EU)
 EN 31.10.2013 Official Journal of the European Union L 289/33 COMMISSION IMPLEMENTING REGULATION (EU) No 1060/2013 of 29 October 2013 concerning the of bentonite as a feed additive for all animal (Text
EN 31.10.2013 Official Journal of the European Union L 289/33 COMMISSION IMPLEMENTING REGULATION (EU) No 1060/2013 of 29 October 2013 concerning the of bentonite as a feed additive for all animal (Text
EUROPEAN COMMISSION. Standing Committee on Plants, Animals, Food and Feed Section Animal Nutrition April 2019 AGENDA
 EUROPEAN COMMISSION Health and Food Safety Directorate General sante.ddg2.g.5(2019)1889069 Standing Committee on Plants, Animals, Food and Feed Section Animal Nutrition 09-11 April 2019 CIRCABC Link: https://circabc.europa.eu/w/browse/b032e44c-8425-427d-a36e-6cbedc052ae1
EUROPEAN COMMISSION Health and Food Safety Directorate General sante.ddg2.g.5(2019)1889069 Standing Committee on Plants, Animals, Food and Feed Section Animal Nutrition 09-11 April 2019 CIRCABC Link: https://circabc.europa.eu/w/browse/b032e44c-8425-427d-a36e-6cbedc052ae1
DRAFT COMMISSION DELEGATED REGULATION (EU) /... of XXX
 EUROPEAN COMMISSION Brussels, XXX C(2016) 3752 projet DRAFT COMMISSION DELEGATED REGULATION (EU) /... of XXX setting out scientific criteria for the determination of endocrine-disrupting properties pursuant
EUROPEAN COMMISSION Brussels, XXX C(2016) 3752 projet DRAFT COMMISSION DELEGATED REGULATION (EU) /... of XXX setting out scientific criteria for the determination of endocrine-disrupting properties pursuant
Official Journal of the European Union
 L 230/8 EN 25.8.2016 COMMISSION REGULATION (EU) 2016/1413 of 24 August 2016 amending Regulation (EU) No 432/2012 establishing a list of permitted health claims made on foods other than those referring
L 230/8 EN 25.8.2016 COMMISSION REGULATION (EU) 2016/1413 of 24 August 2016 amending Regulation (EU) No 432/2012 establishing a list of permitted health claims made on foods other than those referring
EUROPEAN COMMISSION HEALTH & CONSUMERS DIRECTORATE-GENERAL
 EUROPEAN COMMISSION HEALTH & CONSUMERS DIRECTORATE-GENERAL Brussels, SANCO G (2013) 3607634 SUMMARY REPORT OF THE STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH HELD IN BRUSSELS ON 17 OCTOBER 2013-18
EUROPEAN COMMISSION HEALTH & CONSUMERS DIRECTORATE-GENERAL Brussels, SANCO G (2013) 3607634 SUMMARY REPORT OF THE STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH HELD IN BRUSSELS ON 17 OCTOBER 2013-18
Food Additives in the European Union
 Food Additives in Europe http://www.reading.ac.uk/foodlaw/additive.htm Σελίδα 1 από 8 Department of Food and Nutritional Sciences, University of Reading, UK (Note: This page is copyright - D.J.Jukes 2013
Food Additives in Europe http://www.reading.ac.uk/foodlaw/additive.htm Σελίδα 1 από 8 Department of Food and Nutritional Sciences, University of Reading, UK (Note: This page is copyright - D.J.Jukes 2013
EUROPEAN COMMISSION. Standing Committee on Plants, Animals, Food and Feed Section Animal Nutrition 24 APRIL APRIL 2018 AGENDA
 EUROPEAN COMMISSION Health and Food Safety Directorate General sante.ddg2.g.5(2018)1746188 Standing Committee on Plants, Animals, Food and Feed Section Animal Nutrition 24 APRIL 2018-25 APRIL 2018 CIRCABC
EUROPEAN COMMISSION Health and Food Safety Directorate General sante.ddg2.g.5(2018)1746188 Standing Committee on Plants, Animals, Food and Feed Section Animal Nutrition 24 APRIL 2018-25 APRIL 2018 CIRCABC
Council of the European Union Brussels, 6 February 2017 (OR. en)
 Council of the European Union Brussels, 6 February 2017 (OR. en) 5966/17 AGRILEG 28 VETER 11 COVER NOTE From: European Commission date of receipt: 3 February 2017 To: General Secretariat of the Council
Council of the European Union Brussels, 6 February 2017 (OR. en) 5966/17 AGRILEG 28 VETER 11 COVER NOTE From: European Commission date of receipt: 3 February 2017 To: General Secretariat of the Council
Council of the European Union Brussels, 12 November 2015 (OR. en)
 Council of the European Union Brussels, 12 November 2015 (OR. en) 14050/15 AGRILEG 222 DENLEG 147 VETER 102 COVER NOTE From: European Commission date of receipt: 12 November 2015 To: No. Cion doc.: D039258/04
Council of the European Union Brussels, 12 November 2015 (OR. en) 14050/15 AGRILEG 222 DENLEG 147 VETER 102 COVER NOTE From: European Commission date of receipt: 12 November 2015 To: No. Cion doc.: D039258/04
L 243/10 Official Journal of the European Union
 L 243/10 Official Journal of the European Union 15.7.2004 COMMISSION REGULATION (EC) No 1288/2004 of 14 July 2004 concerning the permanent authorisation of certain additives and the provisional authorisation
L 243/10 Official Journal of the European Union 15.7.2004 COMMISSION REGULATION (EC) No 1288/2004 of 14 July 2004 concerning the permanent authorisation of certain additives and the provisional authorisation
DIRECTIVES. (Text with EEA relevance)
 L 238/44 DIRECTIVES COMMISSION DIRECTIVE (EU) 2017/1572 of 15 September 2017 supplementing Directive 2001/83/EC of the European Parliament and of the Council as regards the principles and guidelines of
L 238/44 DIRECTIVES COMMISSION DIRECTIVE (EU) 2017/1572 of 15 September 2017 supplementing Directive 2001/83/EC of the European Parliament and of the Council as regards the principles and guidelines of
Official Journal of the European Union L 318/19
 6.12.2005 Official Journal of the European Union L 318/19 COMMISSION DIRECTIVE 2005/87/EC of 5 December 2005 amending Annex I to Directive 2002/32/EC of the European Parliament and of the Council on undesirable
6.12.2005 Official Journal of the European Union L 318/19 COMMISSION DIRECTIVE 2005/87/EC of 5 December 2005 amending Annex I to Directive 2002/32/EC of the European Parliament and of the Council on undesirable
European Union legislation on Food additives, Food enzymes, Extractions solvents and Food flavourings
 European Union legislation on Food additives, Food enzymes, Extractions solvents and Food flavourings European Commission, DG, Unit E3 Chemicals, contaminants and pesticides Serbia-Screening meeting on
European Union legislation on Food additives, Food enzymes, Extractions solvents and Food flavourings European Commission, DG, Unit E3 Chemicals, contaminants and pesticides Serbia-Screening meeting on
SUMMARY MINUTES OF THE MEETING OF THE STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH. Brussels, February 2007
 SUMMARY MINUTES OF THE MEETING OF THE STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH ANIMAL NUTRITION SECTION Brussels, 22-23 February 2007 1. Feed Additives 1.1. Application under Regulation (EC)
SUMMARY MINUTES OF THE MEETING OF THE STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH ANIMAL NUTRITION SECTION Brussels, 22-23 February 2007 1. Feed Additives 1.1. Application under Regulation (EC)
EUROPEAN COMMISSION SUMMARY REPORT OF THE STANDING COMMITTEE ON PLANTS, ANIMALS, FOOD AND FEED HELD IN BRUSSELS ON 08 MARCH MARCH 2016
 EUROPEAN COMMISSION HEALTH AND FOOD SAFETY DIRECTORATE-GENERAL Ares (2016) 1328021 SUMMARY REPORT OF THE STANDING COMMITTEE ON PLANTS, ANIMALS, FOOD AND FEED HELD IN BRUSSELS ON 08 MARCH 2016-09 MARCH
EUROPEAN COMMISSION HEALTH AND FOOD SAFETY DIRECTORATE-GENERAL Ares (2016) 1328021 SUMMARY REPORT OF THE STANDING COMMITTEE ON PLANTS, ANIMALS, FOOD AND FEED HELD IN BRUSSELS ON 08 MARCH 2016-09 MARCH
Delegations will find attached document D056135/03.
 Council of the European Union Brussels, 1 June 2018 (OR. en) 9586/18 DLEG 46 SAN 175 AGRI 260 COVER NOTE From: European Commission date of receipt: 30 May 2018 To: No. Cion doc.: D056135/03 Subject: General
Council of the European Union Brussels, 1 June 2018 (OR. en) 9586/18 DLEG 46 SAN 175 AGRI 260 COVER NOTE From: European Commission date of receipt: 30 May 2018 To: No. Cion doc.: D056135/03 Subject: General
(Text with EEA relevance)
 L 253/36 EN COMMISSION REGULATION (EU) 2018/1497 of 8 October 2018 amending Annex II to Regulation (EC) No 1333/2008 of the European Parliament and of the Council as regards food category 17 and the use
L 253/36 EN COMMISSION REGULATION (EU) 2018/1497 of 8 October 2018 amending Annex II to Regulation (EC) No 1333/2008 of the European Parliament and of the Council as regards food category 17 and the use
Delegations will find attached document D051559/03.
 Council of the European Union Brussels, 9 November 2017 (OR. en) 14153/17 DLEG 98 AGRI 614 SAN 406 COVER NOTE From: European Commission date of receipt: 8 November 2017 To: No. Cion doc.: D051559/03 Subject:
Council of the European Union Brussels, 9 November 2017 (OR. en) 14153/17 DLEG 98 AGRI 614 SAN 406 COVER NOTE From: European Commission date of receipt: 8 November 2017 To: No. Cion doc.: D051559/03 Subject:
COMMISSION REGULATION (EU) / of XXX
 Ref. Ares(2019)141469-10/01/2019 EUROPEAN COMMISSION Brussels, XXX [ ](2018) XXX draft COMMISSION REGULATION (EU) / of XXX amending, for the purposes of its adaptation to technical and scientific progress,
Ref. Ares(2019)141469-10/01/2019 EUROPEAN COMMISSION Brussels, XXX [ ](2018) XXX draft COMMISSION REGULATION (EU) / of XXX amending, for the purposes of its adaptation to technical and scientific progress,
EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL. (adopted on 19 June 2002)
 EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL Directorate C - Scientific Opinions C2 - Management of scientific committees; scientific co-operation and networks REPORT OF THE SCIENTIFIC
EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL Directorate C - Scientific Opinions C2 - Management of scientific committees; scientific co-operation and networks REPORT OF THE SCIENTIFIC
COUNCIL OF THE EUROPEAN UNION. Brussels, 7 September 2009 (OR. en) 11261/09 Interinstitutional File: 2008/0002 (COD) DENLEG 51 CODEC 893
 COUNCIL OF THE EUROPEAN UNION Brussels, 7 September 2009 (OR. en) 11261/09 Interinstitutional File: 2008/0002 (COD) DLEG 51 CODEC 893 LEGISLATIVE ACTS AND OTHER INSTRUMTS Subject: Common Position with
COUNCIL OF THE EUROPEAN UNION Brussels, 7 September 2009 (OR. en) 11261/09 Interinstitutional File: 2008/0002 (COD) DLEG 51 CODEC 893 LEGISLATIVE ACTS AND OTHER INSTRUMTS Subject: Common Position with
Official Journal of the European Union
 24.7.2018 EN L 186/3 COMMISSION IMPLEMENTING REGULATION (EU) 2018/1039 of 23 July 2018 concerning the of Copper(II) diacetate monohydrate, Copper(II) carbonate dihydroxy monohydrate, Copper(II) chloride
24.7.2018 EN L 186/3 COMMISSION IMPLEMENTING REGULATION (EU) 2018/1039 of 23 July 2018 concerning the of Copper(II) diacetate monohydrate, Copper(II) carbonate dihydroxy monohydrate, Copper(II) chloride
13267/11 PM/tl 1 DG I
 COUNCIL OF THE EUROPEAN UNION Brussels, 27 July 2011 13267/11 DLEG 109 COVER NOTE from: European Commission date of receipt: 22 July 2011 to: General Secretariat of the Council No Cion doc.: D015369/01
COUNCIL OF THE EUROPEAN UNION Brussels, 27 July 2011 13267/11 DLEG 109 COVER NOTE from: European Commission date of receipt: 22 July 2011 to: General Secretariat of the Council No Cion doc.: D015369/01
Official Journal of the European Union L 40/19
 11.2.2009 Official Journal of the European Union L 40/19 COMMISSION DIRECTIVE 2009/8/EC of 10 February 2009 amending Annex I to Directive 2002/32/EC of the European Parliament and of the Council as regards
11.2.2009 Official Journal of the European Union L 40/19 COMMISSION DIRECTIVE 2009/8/EC of 10 February 2009 amending Annex I to Directive 2002/32/EC of the European Parliament and of the Council as regards
COMMISSION REGULATION (EU) / of XXX
 EUROPEAN COMMISSION Brussels, XXX SANTE/11992/2017 Rev. 0 [ ](2017) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annex II to Regulation (EC) No 1107/2009 by setting out scientific criteria for
EUROPEAN COMMISSION Brussels, XXX SANTE/11992/2017 Rev. 0 [ ](2017) XXX draft COMMISSION REGULATION (EU) / of XXX amending Annex II to Regulation (EC) No 1107/2009 by setting out scientific criteria for
SUMMARY RECORD OF THE STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH HELD ON DECEMBER 2009 IN BRUSSELS (Section Animal Nutrition)
 EUROPEAN COMMISSION HEALTH & CONSUMERS DIRECTORATE-GENERAL D(2009)411967 SUMMARY RECORD OF THE STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH HELD ON 17-18 DECEMBER 2009 IN BRUSSELS (Section Animal
EUROPEAN COMMISSION HEALTH & CONSUMERS DIRECTORATE-GENERAL D(2009)411967 SUMMARY RECORD OF THE STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH HELD ON 17-18 DECEMBER 2009 IN BRUSSELS (Section Animal
EUROPEAN COMMISSION SUMMARY REPORT OF THE STANDING COMMITTEE ON PLANTS, ANIMALS, FOOD AND FEED HELD IN BRUSSELS ON 23 APRIL APRIL 2015
 EUROPEAN COMMISSION HEALTH AND FOOD SAFETY DIRECTORATE-GENERAL Ares (2015) 4630466 SUMMARY REPORT OF THE STANDING COMMITTEE ON PLANTS, ANIMALS, FOOD AND FEED HELD IN BRUSSELS ON 23 APRIL 2015-24 APRIL
EUROPEAN COMMISSION HEALTH AND FOOD SAFETY DIRECTORATE-GENERAL Ares (2015) 4630466 SUMMARY REPORT OF THE STANDING COMMITTEE ON PLANTS, ANIMALS, FOOD AND FEED HELD IN BRUSSELS ON 23 APRIL 2015-24 APRIL
Council of the European Union Brussels, 24 March 2015 (OR. en)
 Council of the European Union Brussels, 24 March 2015 (OR. en) 7498/15 DLEG 43 AGRI 154 SAN 84 COVER NOTE From: European Commission date of receipt: 24 March 2015 To: No. Cion doc.: D038008/02 Subject:
Council of the European Union Brussels, 24 March 2015 (OR. en) 7498/15 DLEG 43 AGRI 154 SAN 84 COVER NOTE From: European Commission date of receipt: 24 March 2015 To: No. Cion doc.: D038008/02 Subject:
(Non-legislative acts) REGULATIONS
 EN 23.5.2013 Official Journal of the European Union L 136/1 II (Non-legislative acts) REGULATIONS COMMISSION IMPLEMENTING REGULATION (EU) No 469/2013 of 22 May 2013 concerning the of DL-methionine, DL-methionine
EN 23.5.2013 Official Journal of the European Union L 136/1 II (Non-legislative acts) REGULATIONS COMMISSION IMPLEMENTING REGULATION (EU) No 469/2013 of 22 May 2013 concerning the of DL-methionine, DL-methionine
COMMISSION DELEGATED REGULATION (EU) /... of XXX
 EUROPEAN COMMISSION Brussels, XXX SANTE/11174/2016 (POOL/E1/2016/E1/11174/11174- EN.doc) [ ](2017) XXX draft COMMISSION DELEGATED REGULATION (EU) /... of XXX amending the Annex to Regulation (EU) No 609/2013
EUROPEAN COMMISSION Brussels, XXX SANTE/11174/2016 (POOL/E1/2016/E1/11174/11174- EN.doc) [ ](2017) XXX draft COMMISSION DELEGATED REGULATION (EU) /... of XXX amending the Annex to Regulation (EU) No 609/2013
HEALTH & CONSUMERS DIRECTORATE-GENERAL
 EUROPEAN COMMISSION HEALTH & CONSUMERS DIRECTORATE-GENERAL Brussels, SANCO G Ares (2012) 1293315 SUMMARY REPORT OF THE STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH HELD IN BRUSSELS ON 24 SEPTEMBER
EUROPEAN COMMISSION HEALTH & CONSUMERS DIRECTORATE-GENERAL Brussels, SANCO G Ares (2012) 1293315 SUMMARY REPORT OF THE STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH HELD IN BRUSSELS ON 24 SEPTEMBER
COMMISSION REGULATION (EU)
 29.8.2013 Official Journal of the European Union L 230/7 COMMISSION REGULATION (EU) No 817/2013 of 28 August 2013 amending Annexes II and III to Regulation (EC) No 1333/2008 of the European Parliament
29.8.2013 Official Journal of the European Union L 230/7 COMMISSION REGULATION (EU) No 817/2013 of 28 August 2013 amending Annexes II and III to Regulation (EC) No 1333/2008 of the European Parliament
HEALTH & CONSUMERS DIRECTORATE-GENERAL
 EUROPEAN COMMISSION HEALTH & CONSUMERS DIRECTORATE-GENERAL Brussels, SANCO E 2707357 SUMMARY REPORT OF THE STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH HELD IN BRUSSELS ON 22 APRIL 2013-23 APRIL
EUROPEAN COMMISSION HEALTH & CONSUMERS DIRECTORATE-GENERAL Brussels, SANCO E 2707357 SUMMARY REPORT OF THE STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH HELD IN BRUSSELS ON 22 APRIL 2013-23 APRIL
COMMISSION REGULATION (EU) / of XXX
 Ref. Ares(2017)4140854-23/08/2017 EUROPEAN COMMISSION Brussels, XXX SANTE/10578/2017 Rev. 2 CIS (POOL/E2/2017/10578/10578R2-EN CIS.doc) [ ](2017) XXX draft COMMISSION REGULATION (EU) / of XXX on the use
Ref. Ares(2017)4140854-23/08/2017 EUROPEAN COMMISSION Brussels, XXX SANTE/10578/2017 Rev. 2 CIS (POOL/E2/2017/10578/10578R2-EN CIS.doc) [ ](2017) XXX draft COMMISSION REGULATION (EU) / of XXX on the use
EUROPEAN COMMISSION. Standing Committee on Plants, Animals, Food and Feed Section Animal Nutrition 23 JUNE JUNE 2016 AGENDA
 EUROPEAN COMMISSION HEALTH AND FOOD SAFETY DIRECTORATE-GENERAL Ares (2016) 3148606 Standing Committee on Plants, Animals, Food and Feed Section Animal Nutrition 23 JUNE 2016-24 JUNE 2016 CIRCABC Link:
EUROPEAN COMMISSION HEALTH AND FOOD SAFETY DIRECTORATE-GENERAL Ares (2016) 3148606 Standing Committee on Plants, Animals, Food and Feed Section Animal Nutrition 23 JUNE 2016-24 JUNE 2016 CIRCABC Link:
COMMISSION IMPLEMENTING DECISION. of
 EUROPEAN COMMISSION Brussels, 14-04-2011 C(2011)2799 COMMISSION IMPLEMENTING DECISION of 14-04-2011 granting marketing authorisation under Regulation (EC) No 726/2004 of the European Parliament and of
EUROPEAN COMMISSION Brussels, 14-04-2011 C(2011)2799 COMMISSION IMPLEMENTING DECISION of 14-04-2011 granting marketing authorisation under Regulation (EC) No 726/2004 of the European Parliament and of
COMMISSION DELEGATED REGULATION (EU) /... of XXX
 EUROPEAN COMMISSION Brussels, XXX SANTE/11481/2018 CIS (POOL/E1/2018/11481/11481- CIS.doc) [ ](2018) XXX draft COMMISSION DELEGATED REGULATION (EU) /... of XXX amending Commission Delegated Regulation
EUROPEAN COMMISSION Brussels, XXX SANTE/11481/2018 CIS (POOL/E1/2018/11481/11481- CIS.doc) [ ](2018) XXX draft COMMISSION DELEGATED REGULATION (EU) /... of XXX amending Commission Delegated Regulation
2003 No. 301 FOOD. Made th June 2003 Coming into operation 21st July 2003
 STATUTORY RULES OF NORTHERN IRELAND 2003 No. 301 FOOD Specified Sugar Products Regulations (Northern Ireland) 2003 Made - - - - 10th June 2003 Coming into operation 21st July 2003 The Department of Health,
STATUTORY RULES OF NORTHERN IRELAND 2003 No. 301 FOOD Specified Sugar Products Regulations (Northern Ireland) 2003 Made - - - - 10th June 2003 Coming into operation 21st July 2003 The Department of Health,
COMMISSION REGULATION (EU)
 L 89/36 Official Journal of the European Union 25.3.2014 COMMISSION REGULATION (EU) No 298/2014 of 21 March 2014 amending Annex II to Regulation (EC) No 1333/2008 of the European Parliament and of the
L 89/36 Official Journal of the European Union 25.3.2014 COMMISSION REGULATION (EU) No 298/2014 of 21 March 2014 amending Annex II to Regulation (EC) No 1333/2008 of the European Parliament and of the
COMMISSION REGULATION (EU) / of XXX
 Ref. Ares(2017)4140854-23/08/2017 EUROPEAN COMMISSION Brussels, XXX SANTE/10578/2017 Rev. 2 CIS (POOL/E2/2017/10578/10578R2-EN CIS.doc) [ ](2017) XXX draft COMMISSION REGULATION (EU) / of XXX on the use
Ref. Ares(2017)4140854-23/08/2017 EUROPEAN COMMISSION Brussels, XXX SANTE/10578/2017 Rev. 2 CIS (POOL/E2/2017/10578/10578R2-EN CIS.doc) [ ](2017) XXX draft COMMISSION REGULATION (EU) / of XXX on the use
Act on Narcotic Drugs and Psychotropic Substances and Precursors thereof
 Issuer: Riigikogu Type: act In force from: 13.05.2016 In force until: 22.11.2018 Translation published: 06.05.2016 Act on Narcotic Drugs and Psychotropic Substances and Precursors thereof Amended by the
Issuer: Riigikogu Type: act In force from: 13.05.2016 In force until: 22.11.2018 Translation published: 06.05.2016 Act on Narcotic Drugs and Psychotropic Substances and Precursors thereof Amended by the
Please find below the Commission Communication and the Commission Recommendation for the substance zinc sulphate
 Please find below the Commission Communication and the Commission Recommendation for the substance zinc sulphate CAS No: 7733-02-0 EINECS No: 231-793-3 20.6.2008 EN C 155/1 II (Information) INFORMATION
Please find below the Commission Communication and the Commission Recommendation for the substance zinc sulphate CAS No: 7733-02-0 EINECS No: 231-793-3 20.6.2008 EN C 155/1 II (Information) INFORMATION
REGULATIONS. REGULATION (EC) No 1272/2008 OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL. of 16 December 2008
 31.12.2008 EN Official Journal of the European Union L 353/1 I (Acts adopted under the EC Treaty/Euratom Treaty whose publication is obligatory) REGULATIONS REGULATION (EC) No 1272/2008 OF THE EUROPEAN
31.12.2008 EN Official Journal of the European Union L 353/1 I (Acts adopted under the EC Treaty/Euratom Treaty whose publication is obligatory) REGULATIONS REGULATION (EC) No 1272/2008 OF THE EUROPEAN
COMMISSION REGULATION (EC)
 20.5.2009 Official Journal of the European Union L 124/3 COMMISSION REGULATION (EC) No 411/2009 of 18 May 2009 amending Regulation (EC) No 798/2008 laying down a list of third countries, territories, zones
20.5.2009 Official Journal of the European Union L 124/3 COMMISSION REGULATION (EC) No 411/2009 of 18 May 2009 amending Regulation (EC) No 798/2008 laying down a list of third countries, territories, zones
Health & Consumer Protection. EC legislation on food. Olga Solomon Unit E3
 Health & Consumer Protection Directorate-General EC legislation on food additives Olga Solomon Unit E3 1 What is a food additive? Food additives are substances: not normally consumed as food or a characteristic
Health & Consumer Protection Directorate-General EC legislation on food additives Olga Solomon Unit E3 1 What is a food additive? Food additives are substances: not normally consumed as food or a characteristic
EUROPEAN COMMISSION. Standing Committee on Plants, Animals, Food and Feed Section Animal Nutrition 23 MAY MAY 2016 AGENDA
 EUROPEAN COMMISSION HEALTH AND FOOD SAFETY DIRECTORATE-GENERAL Ares (2016) 2537654 Standing Committee on Plants, Animals, Food and Feed Section Animal Nutrition 23 MAY 2016-24 MAY 2016 CIRCABC Link: https://circabc.europa.eu/w/browse/e360a1b3-c86e-4696-8122-f8c95d0412c5
EUROPEAN COMMISSION HEALTH AND FOOD SAFETY DIRECTORATE-GENERAL Ares (2016) 2537654 Standing Committee on Plants, Animals, Food and Feed Section Animal Nutrition 23 MAY 2016-24 MAY 2016 CIRCABC Link: https://circabc.europa.eu/w/browse/e360a1b3-c86e-4696-8122-f8c95d0412c5
COMMISSION DELEGATED REGULATION (EU) /... of XXX
 EUROPEAN COMMISSION Brussels, XXX SANTE/11481/2018 CIS (POOL/E1/2018/11481/11481-EN CIS.doc) [ ](2018) XXX draft COMMISSION DELEGATED REGULATION (EU) /... of XXX amending Commission Delegated Regulation
EUROPEAN COMMISSION Brussels, XXX SANTE/11481/2018 CIS (POOL/E1/2018/11481/11481-EN CIS.doc) [ ](2018) XXX draft COMMISSION DELEGATED REGULATION (EU) /... of XXX amending Commission Delegated Regulation
COMMISSION OF THE EUROPEAN COMMUNITIES COMMISSION DECISION. of 18-XII-2007
 COMMISSION OF THE EUROPEAN COMMUNITIES COMMISSION DECISION of 18-XII-2007 Brussels, 18-XII-2007 C(2007)6737 NOT FOR PUBLICATION granting marketing authorisation under Regulation (EC) n 726/2004 of the
COMMISSION OF THE EUROPEAN COMMUNITIES COMMISSION DECISION of 18-XII-2007 Brussels, 18-XII-2007 C(2007)6737 NOT FOR PUBLICATION granting marketing authorisation under Regulation (EC) n 726/2004 of the
(Text with EEA relevance)
 L 274/51 COMMISSION IMPLEMENTING REGULATION (EU) 2018/1647 of 31 October 2018 authorising the placing on the market of egg membrane hydrolysate as a novel food under Regulation (EU) 2015/2283 of the European
L 274/51 COMMISSION IMPLEMENTING REGULATION (EU) 2018/1647 of 31 October 2018 authorising the placing on the market of egg membrane hydrolysate as a novel food under Regulation (EU) 2015/2283 of the European
REGULATION concerning Marketing Authorisations for Natural Medicinal Products and Registration of Traditional Herbal Medicinal Products, No. 142/2011.
 REGULATION concerning Marketing Authorisations for Natural Medicinal Products and Registration of Traditional Herbal Medicinal Products, No. 142/2011. CHAPTER I Scope and definitions. Article 1 Scope.
REGULATION concerning Marketing Authorisations for Natural Medicinal Products and Registration of Traditional Herbal Medicinal Products, No. 142/2011. CHAPTER I Scope and definitions. Article 1 Scope.
COMMISSION REGULATION (EU) / of XXX
 ² EUROPEAN COMMISSION Brussels, XXX SANTE/00131/2015 REV1 [ ](2015) XXX draft COMMISSION REGULATION (EU) / of XXX amending Section E of Chapter V of Annex IV to Regulation (EC) No 999/2001 of the European
² EUROPEAN COMMISSION Brussels, XXX SANTE/00131/2015 REV1 [ ](2015) XXX draft COMMISSION REGULATION (EU) / of XXX amending Section E of Chapter V of Annex IV to Regulation (EC) No 999/2001 of the European
STATUTORY INSTRUMENTS. S.I. No. 724 of 2011 FOOD SAFETY AUTHORITY OF IRELAND ACT 1998 (AMENDMENT OF FIRST AND SECOND SCHEDULES) ORDER 2011
 STATUTORY INSTRUMENTS. S.I. No. 724 of 2011 FOOD SAFETY AUTHORITY OF IRELAND ACT 1998 (AMENDMENT OF FIRST AND SECOND SCHEDULES) ORDER 2011 (Prn. A11/2427) 2 [724] S.I. No. 724 of 2011 FOOD SAFETY AUTHORITY
STATUTORY INSTRUMENTS. S.I. No. 724 of 2011 FOOD SAFETY AUTHORITY OF IRELAND ACT 1998 (AMENDMENT OF FIRST AND SECOND SCHEDULES) ORDER 2011 (Prn. A11/2427) 2 [724] S.I. No. 724 of 2011 FOOD SAFETY AUTHORITY
(Text with EEA relevance) (2014/798/EU)
 L 330/50 COMMISSION IMPLEMTING DECISION of 13 November 2014 amending Annex F to Council Directive 64/432/EEC as regards the format of the model health certificates for intra-union trade in bovine animals
L 330/50 COMMISSION IMPLEMTING DECISION of 13 November 2014 amending Annex F to Council Directive 64/432/EEC as regards the format of the model health certificates for intra-union trade in bovine animals
Official Journal of the European Union. (Non-legislative acts) REGULATIONS
 22.8.2017 EN L 216/1 II (Non-legislative acts) REGULATIONS COMMISSION IMPLEMENTING REGULATION (EU) 2017/1490 of 21 August 2017 concerning the of manganous chloride tetrahydrate, manganese (II) oxide, manganous
22.8.2017 EN L 216/1 II (Non-legislative acts) REGULATIONS COMMISSION IMPLEMENTING REGULATION (EU) 2017/1490 of 21 August 2017 concerning the of manganous chloride tetrahydrate, manganese (II) oxide, manganous
COMMISSION OF THE EUROPEAN COMMUNITIES COMMISSION DECISION. of 29-VIII-2008
 COMMISSION OF THE EUROPEAN COMMUNITIES COMMISSION DECISION of 29-VIII-2008 Brussels, 29-VIII-2008 C(2008)4783 NOT FOR PUBLICATION granting marketing authorisation under Regulation (EC) No 726/2004 of the
COMMISSION OF THE EUROPEAN COMMUNITIES COMMISSION DECISION of 29-VIII-2008 Brussels, 29-VIII-2008 C(2008)4783 NOT FOR PUBLICATION granting marketing authorisation under Regulation (EC) No 726/2004 of the
(Text with EEA relevance)
 23.10.2014 EN L 304/81 COMMISSION REGULATION (EU) No 1123/2014 of 22 October 2014 amending Directive 2008/38/EC establishing a list of intended uses of feedingstuffs for particular s (Text with EEA relevance)
23.10.2014 EN L 304/81 COMMISSION REGULATION (EU) No 1123/2014 of 22 October 2014 amending Directive 2008/38/EC establishing a list of intended uses of feedingstuffs for particular s (Text with EEA relevance)
COMMISSION OF THE EUROPEAN COMMUNITIES COMMISSION DECISION. of
 COMMISSION OF THE EUROPEAN COMMUNITIES Brussels, 5.10.2009 C(2009)7726 COMMISSION DECISION of 5.10.2009 granting marketing authorisation under Regulation (EC) No 726/2004 of the European Parliament and
COMMISSION OF THE EUROPEAN COMMUNITIES Brussels, 5.10.2009 C(2009)7726 COMMISSION DECISION of 5.10.2009 granting marketing authorisation under Regulation (EC) No 726/2004 of the European Parliament and
CED GUIDELINES TO INTERPRET AND IMPLEMENT COUNCIL DIRECTIVE 2011/84/EU ON TOOTH WHITENING PRODUCTS
 CED GUIDELINES TO INTERPRET AND IMPLEMENT COUNCIL DIRECTIVE 2011/84/EU ON TOOTH WHITENING PRODUCTS I INTRODUCTION This document provides guidance for interpreting and implementing the Council Directive
CED GUIDELINES TO INTERPRET AND IMPLEMENT COUNCIL DIRECTIVE 2011/84/EU ON TOOTH WHITENING PRODUCTS I INTRODUCTION This document provides guidance for interpreting and implementing the Council Directive
13480/12 PM/ns 1 DG B 4B
 COUNCIL OF THE EUROPEAN UNION Brussels, 10 September 2012 13480/12 DENLEG 80 AGRI 553 COVER NOTE from: European Commission date of receipt: 22 August 2012 to: General Secretariat of the Council No Cion
COUNCIL OF THE EUROPEAN UNION Brussels, 10 September 2012 13480/12 DENLEG 80 AGRI 553 COVER NOTE from: European Commission date of receipt: 22 August 2012 to: General Secretariat of the Council No Cion
WORKING DOCUMENT DOES NOT NECESSARILY REPRESENT THE VIEWS OF THE EUROPEAN COMMISSION SERVICES
 EUROPEAN COMMISSION Brussels, XXX SANTE/11728/2016 [ ](2016) XXX draft of XXX in certain raw apricot kernels and derived products (Text with EEA relevance) WORKING DOCUMT DOES NOT NECESSARILY REPREST THE
EUROPEAN COMMISSION Brussels, XXX SANTE/11728/2016 [ ](2016) XXX draft of XXX in certain raw apricot kernels and derived products (Text with EEA relevance) WORKING DOCUMT DOES NOT NECESSARILY REPREST THE
This document is meant purely as a documentation tool and the institutions do not assume any liability for its contents
 2007R1394 EN 02.07.2012 001.001 1 This document is meant purely as a documentation tool and the institutions do not assume any liability for its contents B REGULATION (EC) No 1394/2007 OF THE EUROPEAN
2007R1394 EN 02.07.2012 001.001 1 This document is meant purely as a documentation tool and the institutions do not assume any liability for its contents B REGULATION (EC) No 1394/2007 OF THE EUROPEAN
COMMISSION IMPLEMENTING REGULATION (EU) /... of XXX
 EUROPEAN COMMISSION Brussels, XXX [ ](2018) XXX draft COMMISSION IMPLEMENTING REGULATION (EU) /... of XXX amending Regulation (EC) No 889/2008 laying down detailed rules for the implementation of Council
EUROPEAN COMMISSION Brussels, XXX [ ](2018) XXX draft COMMISSION IMPLEMENTING REGULATION (EU) /... of XXX amending Regulation (EC) No 889/2008 laying down detailed rules for the implementation of Council
COMMISSION REGULATION (EU)
 11.3.2011 Official Journal of the European Union L 64/15 COMMISSION REGULATION (EU) No 234/2011 of 10 March 2011 implementing Regulation (EC) No 1331/2008 of the European Parliament and of the Council
11.3.2011 Official Journal of the European Union L 64/15 COMMISSION REGULATION (EU) No 234/2011 of 10 March 2011 implementing Regulation (EC) No 1331/2008 of the European Parliament and of the Council
EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL. (Adopted on 22 January 2003)
 EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL Directorate C - Scientific Opinions C2 - Management of scientific committees; scientific co-operation and networks REPORT OF THE SCIENTIFIC
EUROPEAN COMMISSION HEALTH & CONSUMER PROTECTION DIRECTORATE-GENERAL Directorate C - Scientific Opinions C2 - Management of scientific committees; scientific co-operation and networks REPORT OF THE SCIENTIFIC
(Non-legislative acts) REGULATIONS
 17.1.2013 Official Journal of the European Union L 13/1 II (Non-legislative acts) REGULATIONS COMMISSION REGULATION (EU) No 25/2013 of 16 January 2013 amending Annexes II and III to Regulation (EC) No
17.1.2013 Official Journal of the European Union L 13/1 II (Non-legislative acts) REGULATIONS COMMISSION REGULATION (EU) No 25/2013 of 16 January 2013 amending Annexes II and III to Regulation (EC) No
Council of the European Union Brussels, 14 February 2017 (OR. en)
 Council of the European Union Brussels, 14 February 2017 (OR. en) 6300/17 AGRILEG 43 VETER 17 COVER NOTE From: European Commission date of receipt: 13 February 2017 To: General Secretariat of the Council
Council of the European Union Brussels, 14 February 2017 (OR. en) 6300/17 AGRILEG 43 VETER 17 COVER NOTE From: European Commission date of receipt: 13 February 2017 To: General Secretariat of the Council
EUROPEAN COMMISSION SUMMARY REPORT OF THE STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH HELD IN BRUSSELS ON 17 FEBRUARY FEBRUARY 2014
 EUROPEAN COMMISSION HEALTH AND CONSUMERS DIRECTORATE-GENERAL Brussels, SANCO G (2014) 394592 SUMMARY REPORT OF THE STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH HELD IN BRUSSELS ON 17 FEBRUARY
EUROPEAN COMMISSION HEALTH AND CONSUMERS DIRECTORATE-GENERAL Brussels, SANCO G (2014) 394592 SUMMARY REPORT OF THE STANDING COMMITTEE ON THE FOOD CHAIN AND ANIMAL HEALTH HELD IN BRUSSELS ON 17 FEBRUARY
EUROPEAN COMMISSION DIRECTORATE-GENERAL FOR HEALTH AND FOOD SAFETY
 EUROPEAN COMMISSION DIRECTORATE-GENERAL FOR HEALTH AND FOOD SAFETY Food and feed safety, innovation Pesticides and biocides COMMISSION STAFF WORKING DOCUMENT 1 Basic Substance diammonium phosphate SANTE/12351/2015
EUROPEAN COMMISSION DIRECTORATE-GENERAL FOR HEALTH AND FOOD SAFETY Food and feed safety, innovation Pesticides and biocides COMMISSION STAFF WORKING DOCUMENT 1 Basic Substance diammonium phosphate SANTE/12351/2015
DIRECTIVE 2004/24/EC OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL. of 31 March 2004
 30.4.2004 Official Journal of the European Union L 136/85 DIRECTIVE 2004/24/EC OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL of 31 March 2004 amending, as regards traditional herbal medicinal products,
30.4.2004 Official Journal of the European Union L 136/85 DIRECTIVE 2004/24/EC OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL of 31 March 2004 amending, as regards traditional herbal medicinal products,
National Food Administration s Regulations on the Use of a Particular Symbol 1 ;
 National Food Administration s Regulations on the Use of a Particular Symbol 1 ; Translation by the Swedish National Food Administration March 2006 This translation is provided for information and guidance
National Food Administration s Regulations on the Use of a Particular Symbol 1 ; Translation by the Swedish National Food Administration March 2006 This translation is provided for information and guidance
Official Journal of the European Union. (Non-legislative acts) REGULATIONS
 21.2.2015 L 50/1 II (Non-legislative acts) REGULATIONS COMMISSION REGULATION (EU) 2015/282 of 20 February 2015 amending Annexes VIII, IX and X to Regulation (EC) No 1907/2006 of the European Parliament
21.2.2015 L 50/1 II (Non-legislative acts) REGULATIONS COMMISSION REGULATION (EU) 2015/282 of 20 February 2015 amending Annexes VIII, IX and X to Regulation (EC) No 1907/2006 of the European Parliament
(Text with EEA relevance)
 5.10.2018 L 251/13 COMMISSION REGULATION (EU) 2018/1481 of 4 October 2018 amending Annexes II and III to Regulation (EC) No 1333/2008 of the European Parliament and of the Council and the Annex to Commission
5.10.2018 L 251/13 COMMISSION REGULATION (EU) 2018/1481 of 4 October 2018 amending Annexes II and III to Regulation (EC) No 1333/2008 of the European Parliament and of the Council and the Annex to Commission
(Text with EEA relevance)
 L 204/36 EN COMMISSION IMPLEMENTING REGULATION (EU) 2018/1122 of 10 August 2018 authorising the placing on the market of pyrroloquinoline quinone disodium salt as a novel food under Regulation (EU) 2015/2283
L 204/36 EN COMMISSION IMPLEMENTING REGULATION (EU) 2018/1122 of 10 August 2018 authorising the placing on the market of pyrroloquinoline quinone disodium salt as a novel food under Regulation (EU) 2015/2283
THE FOOD AND FEED IMPORTS (EU EXIT) REGULATIONS 2018 THE MATERIALS AND ARTICLES IN CONTACT WITH FOOD (EU EXIT) REGULATIONS 2018
 Minister for Public Health, Sport and Wellbeing Joe FitzPatrick MSP T: 0300 244 4000 E: scottish.ministers@gov.scot Lewis Macdonald MSP Convenor Health and Sport Committee Scottish Parliament Edinburgh
Minister for Public Health, Sport and Wellbeing Joe FitzPatrick MSP T: 0300 244 4000 E: scottish.ministers@gov.scot Lewis Macdonald MSP Convenor Health and Sport Committee Scottish Parliament Edinburgh
COMMISSION OF THE EUROPEAN COMMUNITIES COMMISSION STAFF WORKING DOCUMENT
 EN EN EN COMMISSION OF THE EUROPEAN COMMUNITIES Brussels, 17.10.2007 SEC(2007) 1411 PART I COMMISSION STAFF WORKING DOCUMENT Monitoring of Pesticide Residues in Products of Plant Origin in the European
EN EN EN COMMISSION OF THE EUROPEAN COMMUNITIES Brussels, 17.10.2007 SEC(2007) 1411 PART I COMMISSION STAFF WORKING DOCUMENT Monitoring of Pesticide Residues in Products of Plant Origin in the European
REASONED OPINION. European Food Safety Authority 2, 3. European Food Safety Authority (EFSA), Parma, Italy
 EFSA Journal 2012;10(7):2841 REASONED OPINION Reasoned opinion on the review of the existing maximum residue levels (MRLs) for paraffin oil (CAS 64742-54-7) according to Article 12 of Regulation (EC) No
EFSA Journal 2012;10(7):2841 REASONED OPINION Reasoned opinion on the review of the existing maximum residue levels (MRLs) for paraffin oil (CAS 64742-54-7) according to Article 12 of Regulation (EC) No
6961/13 PM/ns 1 DG B 4B
 COUNCIL OF THE EUROPEAN UNION Brussels, 4 March 2013 6961/13 DENLEG 19 AGRI 132 COVER NOTE from: European Commission date of receipt: 27 February 2013 to: General Secretariat of the Council No Cion doc.:
COUNCIL OF THE EUROPEAN UNION Brussels, 4 March 2013 6961/13 DENLEG 19 AGRI 132 COVER NOTE from: European Commission date of receipt: 27 February 2013 to: General Secretariat of the Council No Cion doc.:
EUROPEAN UNION. Brussels, 15 October 2007 (OR. en) 2005/0227 (COD) PE-CONS 3627/07 MI 149 ECO 83 SAN 123 CODEC 621
 EUROPEAN UNION THE EUROPEAN PARLIAMT Brussels, 15 October 2007 (OR. en) THE COUNCIL 2005/0227 (COD) PE-CONS 3627/07 MI 149 ECO 83 SAN 123 CODEC 621 LEGISLATIVE ACTS AND OTHER INSTRUMTS Subject: Regulation
EUROPEAN UNION THE EUROPEAN PARLIAMT Brussels, 15 October 2007 (OR. en) THE COUNCIL 2005/0227 (COD) PE-CONS 3627/07 MI 149 ECO 83 SAN 123 CODEC 621 LEGISLATIVE ACTS AND OTHER INSTRUMTS Subject: Regulation
