PRODUCT INFORMATION Panadeine EXTRA
|
|
|
- Gillian Sharp
- 6 years ago
- Views:
Transcription
1 PRODUCT INFORMATION Panadeine EXTRA COMPOSITION Each caplet brand of capsule-shaped tablet contains: Paracetamol 500 mg Codeine phosphate 15 mg and Maize Starch Purified Talc Pregelatinised Maize Starch Povidone Stearic Acid Potassium Sorbate Magnesium Stearate MATERIAL OF ANIMAL ORIGIN None ACTIONS Analgesic and antipyretic. Paracetamol's analgesic mechanism of action has not been fully elucidated but may involve blocking impulse generation at the bradykinin-sensitive chemoreceptors that evoke pain. The antipyretic effect of paracetamol rises from its ability to block the action of prostaglandin synthetase and so prevent the synthesis of prostaglandins in response to the pyrogen stimulus in the region of the anterior hypothalamus. Codeine acts centrally. It produces analgesia by dulling the response to painful stimuli at several loci in the CNS. This causes an alteration in the sensation and affective response of pain. There is evidence to suggest that a combination of paracetamol with codeine is superior in analgesic action to either drug administered alone. PANADEINE EXTRA Product Information Page 1
2 PHARMACOKINETICS Paracetamol After oral administration, paracetamol is absorbed rapidly and completely from the small intestine; peak plasma levels occur 30 to 120 minutes after administration. Paracetamol is uniformly distributed throughout most body fluids; the apparent volume of distribution is 1 to 1.2 L/kg. Paracetamol can cross the placenta and is excreted in milk. Plasma protein binding is negligible at usual therapeutic concentrations but increases with increasing concentrations. Paracetamol is metabolised by the hepatic microsomal enzyme system. In adults at therapeutic doses, paracetamol is mainly conjugated with glucuronide (45-55%) or sulfate (20-30%). A minor proportion (less than 20%) is metabolised to catechol derivatives and mercapturic acid compounds via oxidation. Paracetamol is metabolised differently by infants and children compared to adults, the sulfate conjugate being predominant. Paracetamol is excreted in the urine mainly as the glucuronide and sulphate conjugates. Less than 5% is excreted as unchanged paracetamol with 85-90% of the administered dose eliminated in the urine within 24 hours of ingestion. The elimination half-life varies from 1 to 4 hours. Food intake delays paracetamol absorption. Codeine Codeine has about one-sixth of morphine's analgesic activity. It is well absorbed from the gastrointestinal tract and does not interfere with paracetamol absorption. It is metabolised in the liver to morphine and norcodeine, which with codeine, are excreted in the urine, partly as conjugates with glucuronic acid. Excretion is almost complete within 24 hours. Patients who metabolise drugs poorly via CYP2D6 are likely to obtain reduced benefit from codeine due to reduced formation of the active metabolite. This may be the case in about 8% of patients. INDICATIONS Temporary relief of moderate to severe acute pain associated with strong headaches, migraine headaches, dental surgery or toothache, menstrual pain and sports injuries (eg. backaches and muscular pain). PANADEINE EXTRA Product Information Page 2
3 CONTRAINDICATIONS Hypersensitivity to paracetamol or codeine or other ingredients (see composition). PRECAUTIONS Panadeine EXTRA should be administered with caution to patients with hepatic or renal dysfunction. Codeine should be used with caution in patients with CNS depression or decreased respiratory reserve. Prolonged use of high doses of codeine may produce dependence. Due to the preparation's sedative action, impairment of the mental and/or physical abilities required for the performance of potentially hazardous activities may occur. Hence children engaging in bike riding and other hazardous activities should be supervised to avoid potential harm. Adults should not drive, operate machinery or drink alcohol whilst taking this medication. Use in Pregnancy Category A Use in Lactation When Panadeine EXTRA is administered to a nursing mother, alternative arrangements should be made for feeding the infant. Drug Interactions Anticoagulant dosage may require reduction if Panadeine EXTRA medication is prolonged. Paracetamol absorption is increased by drugs which increase gastric emptying, eg. metoclopramide, and decreased by drugs which decrease gastric emptying, eg. propantheline, antidepressants with anticholinergic properties and narcotic analgesics. Paracetamol may increase chloramphenicol concentrations. The likelihood of paracetamol toxicity may be increased by the concomitant use of enzyme-inducing agents such as alcohol or anti-epileptic drugs. It is possible that interactions could occur between drugs that can inhibit CYP2D6 (such as quinidine, phenothiazines and antipsychotic agents) and codeine. Concurrent administration of sedatives or tranquillisers may enhance the potential respiratory depressant effects of codeine. PANADEINE EXTRA Product Information Page 3
4 ADVERSE REACTIONS Paracetamol Reports of adverse reactions are rare. Although the following reactions have been reported, a causal relationship to the administration of paracetamol has been neither confirmed nor refuted: dyspepsia, nausea, allergic and haematological reactions. Codeine Nausea and vomiting, constipation, dizziness and drowsiness have been reported at therapeutic doses. Very rarely, skin rashes may occur in patients hypersensitive to codeine. Prolonged use of large doses of codeine may result in physiological dependence. DOSAGE AND ADMINISTRATION Adults and Children over 12 years 2 caplets four times a day if required (maximum 8 caplets in 24 hours). Not recommended for children under 12 years of age. OVERDOSAGE Symptoms Toxic symptoms include vomiting, abdominal pain, hypotension, sweating, central stimulation with exhilaration and convulsions in children, drowsiness, respiratory depression, cyanosis and coma. The most serious adverse effect of acute overdosage of paracetamol is a dose-dependent, potentially fatal hepatic necrosis. In adults, hepatotoxicity may occur after ingestion of a single dose of 10 to 15 g (30 tablets) of paracetamol; a dose of 25 g (50 tablets) or more is potentially fatal. Symptoms during the first two days of acute poisoning by paracetamol do not reflect the potential seriousness of the intoxication. Major manifestations of liver failure such as jaundice, hypoglycaemia and metabolic acidosis may take at least three days to develop. In case of overdosage, contact the Poisons Information Centre ( ). PRESENTATION Panadeine EXTRA caplets Capsule-shaped tablets (white, marked PAN 15): 12s and 24s (retail pack) and 6s (health professional sample pack). PANADEINE EXTRA Product Information Page 4
5 POISON SCHEDULE S3: Pharmacist Only Medicine SPONSOR GlaxoSmithKline Australia Pty Ltd (t/a GlaxoSmithKline Consumer Healthcare) 82 Hughes Avenue Ermington NSW 2115 TGA approved: November 2009 Panadeine and Caplets are registered trade marks of the GlaxoSmithKline group of companies. PANADEINE EXTRA Product Information Page 5
PRODUCT INFORMATION CODAPANE XTRA Paracetamol 500 mg and Codeine Phosphate 15 mg Tablets
 PRODUCT INFORMATION CODAPANE XTRA Paracetamol 500 mg and Codeine Phosphate 15 mg Tablets NAME OF THE MEDICINE Active Ingredients: Paracetamol and Codeine Phosphate Paracetamol: Molecular Formula: C 8 H
PRODUCT INFORMATION CODAPANE XTRA Paracetamol 500 mg and Codeine Phosphate 15 mg Tablets NAME OF THE MEDICINE Active Ingredients: Paracetamol and Codeine Phosphate Paracetamol: Molecular Formula: C 8 H
Panadeine Tablet, Caplets and Rapid Soluble tablets PRODUCT INFORMATION
 Panadeine Tablet, Caplets and Rapid Soluble tablets PRODUCT INFORMATION DESCRIPTION Active Ingredients Paracetamol 500 mg Codeine Phosphate 8 mg Excipients Tablets - Starch - maize, talc - purified, stearic
Panadeine Tablet, Caplets and Rapid Soluble tablets PRODUCT INFORMATION DESCRIPTION Active Ingredients Paracetamol 500 mg Codeine Phosphate 8 mg Excipients Tablets - Starch - maize, talc - purified, stearic
PANADOL EXTRA PRODUCT INFORMATION
 PANADOL EXTRA PRODUCT INFORMATION DESCRIPTION Active Ingredients: Paracetamol 500 mg and Caffeine 65mg per tablet. Excipients: Maize starch Starch - Pregelatinised maize Povidone Potassium sorbate Purified
PANADOL EXTRA PRODUCT INFORMATION DESCRIPTION Active Ingredients: Paracetamol 500 mg and Caffeine 65mg per tablet. Excipients: Maize starch Starch - Pregelatinised maize Povidone Potassium sorbate Purified
Molecular formula: Molecular weight: C 8 H 9 NO 2 CAS Registry no.:
 Parapane Paracetamol PRODUCT INFORMATION NAME OF THE MEDICINE Active ingredient: Chemical name: Paracetamol N-(4-hydroxyphenyl) Structural formula: Molecular formula: 151.20 Molecular weight: C 8 H 9 NO
Parapane Paracetamol PRODUCT INFORMATION NAME OF THE MEDICINE Active ingredient: Chemical name: Paracetamol N-(4-hydroxyphenyl) Structural formula: Molecular formula: 151.20 Molecular weight: C 8 H 9 NO
PRODUCT INFORMATION. (RS)-N,N-Dimethyl-2-[(2-methylphenyl)phenylmethoxy]ethanamine dihydrogen 2-hydroxypropane-1,2,3-tricarboxylate
![PRODUCT INFORMATION. (RS)-N,N-Dimethyl-2-[(2-methylphenyl)phenylmethoxy]ethanamine dihydrogen 2-hydroxypropane-1,2,3-tricarboxylate PRODUCT INFORMATION. (RS)-N,N-Dimethyl-2-[(2-methylphenyl)phenylmethoxy]ethanamine dihydrogen 2-hydroxypropane-1,2,3-tricarboxylate](/thumbs/81/82665370.jpg) NORGESIC Orphenadrine citrate and paracetamol PRODUCT INFORMATION NAME OF THE MEDICINE Active ingredient: Chemical name: CAS number: 4682-36-4 Chemical structure: Orphenadrine citrate (RS)-N,N-Dimethyl-2-[(2-methylphenyl)phenylmethoxy]ethanamine
NORGESIC Orphenadrine citrate and paracetamol PRODUCT INFORMATION NAME OF THE MEDICINE Active ingredient: Chemical name: CAS number: 4682-36-4 Chemical structure: Orphenadrine citrate (RS)-N,N-Dimethyl-2-[(2-methylphenyl)phenylmethoxy]ethanamine
PANADOL COLD & FLU RELIEF ORIGINAL FORMULA is a white, capsule-shaped tablet with flat edges, one face marked with C&F, PANADOL on the other.
 Product description PANADOL COLD & FLU RELIEF ORIGINAL FORMULA is a white, capsule-shaped tablet with flat edges, one face marked with C&F, PANADOL on the other. Ingredients Active Ingredients: Paracetamol
Product description PANADOL COLD & FLU RELIEF ORIGINAL FORMULA is a white, capsule-shaped tablet with flat edges, one face marked with C&F, PANADOL on the other. Ingredients Active Ingredients: Paracetamol
PRODUCT INFORMATION PANADEINE FORTE
 PRODUCT INFORMATION PANADEINE FORTE NAME OF THE MEDICINE Non-proprietary Name Paracetamol and codeine phosphate DESCRIPTION Each capsule-shaped tablet contains Paracetamol 500 mg, Codeine phosphate 30
PRODUCT INFORMATION PANADEINE FORTE NAME OF THE MEDICINE Non-proprietary Name Paracetamol and codeine phosphate DESCRIPTION Each capsule-shaped tablet contains Paracetamol 500 mg, Codeine phosphate 30
PRODUCT INFORMATION PRODEINE 15 H 3 CO. Paracetamol MW Codeine phosphate MW
 PRODUCT INFORMATION PRODEINE 15 NAME OF THE MEDICINE Non-proprietary Name Paracetamol and codeine phosphate. Chemical Structure HO O H N CH 3 NH CH 3 H H 3 CO Paracetamol MW 151.17 Codeine phosphate MW
PRODUCT INFORMATION PRODEINE 15 NAME OF THE MEDICINE Non-proprietary Name Paracetamol and codeine phosphate. Chemical Structure HO O H N CH 3 NH CH 3 H H 3 CO Paracetamol MW 151.17 Codeine phosphate MW
DATA SHEET. PANADOL Mini Caps Capsule shaped tablet with a gelatin coating which is one half green and the other half white.
 PANADOL TABLETS PANADOL MINI CAPS DATA SHEET Proprietary (Trade) Name: PANADOL Active ingredient: Paracetamol (BP) 500 mg/tablet PRESENTATIONS White, film-coated tablet with bevelled edge, shallow convex,
PANADOL TABLETS PANADOL MINI CAPS DATA SHEET Proprietary (Trade) Name: PANADOL Active ingredient: Paracetamol (BP) 500 mg/tablet PRESENTATIONS White, film-coated tablet with bevelled edge, shallow convex,
PRODUCT INFORMATION PANADOL TABLETS PANADOL MINI CAPS PANADOL SUPPOSITORIES
 PRODUCT INFORMATION PANADOL TABLETS PANADOL MINI CAPS PANADOL SUPPOSITORIES NAME OF THE MEDICINE Active ingredients Chemical structure CAS Registry Number Paracetamol 103-90-2 DESCRIPTION Paracetamol is
PRODUCT INFORMATION PANADOL TABLETS PANADOL MINI CAPS PANADOL SUPPOSITORIES NAME OF THE MEDICINE Active ingredients Chemical structure CAS Registry Number Paracetamol 103-90-2 DESCRIPTION Paracetamol is
Active ingredients: Paracetamol Codeine phosphate. Chemical names: N-(4-Hydroxyphenyl)acetamide 4,5a-epoxy-3-methoxy-17-methyl-7,8-
 Codapane Paracetamol / Codeine phosphate PRODUCT INFORMATION NAME OF THE MEDICINE Active ingredients: Paracetamol Codeine phosphate Chemical names: N-(4-Hydroxyphenyl)acetamide 4,5a-epoxy-3-methoxy-17-methyl-7,8-
Codapane Paracetamol / Codeine phosphate PRODUCT INFORMATION NAME OF THE MEDICINE Active ingredients: Paracetamol Codeine phosphate Chemical names: N-(4-Hydroxyphenyl)acetamide 4,5a-epoxy-3-methoxy-17-methyl-7,8-
PANADOL OSTEO PRODUCT INFORMATION
 PANADOL OSTEO PRODUCT INFORMATION NAME OF THE MEDICINE Active ingredients Chemical structure CAS Registry Number Paracetamol 103-90-2 DESCRIPTION White to off-white film coated capsule shaped tablets with
PANADOL OSTEO PRODUCT INFORMATION NAME OF THE MEDICINE Active ingredients Chemical structure CAS Registry Number Paracetamol 103-90-2 DESCRIPTION White to off-white film coated capsule shaped tablets with
NEW ZEALAND DATA SHEET
 1. PRODUCT NAME PANADOL TABLETS, Paracetamol 500 mg, film coated tablet PANADOL MINI CAPS, Paracetamol 500 mg, coated tablet 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Active ingredient: Paracetamol (BP)
1. PRODUCT NAME PANADOL TABLETS, Paracetamol 500 mg, film coated tablet PANADOL MINI CAPS, Paracetamol 500 mg, coated tablet 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Active ingredient: Paracetamol (BP)
PRODUCT INFORMATION ANAGRAINE. Each ANAGRAINE tablet contains 5 mg metoclopramide and 500 mg paracetamol.
 PRODUCT INFORMATION ANAGRAINE COMPOSITION Each ANAGRAINE tablet contains 5 mg metoclopramide and 500 mg paracetamol. ACTIONS Metoclopramide stimulates gastrointestinal tract motility and accelerates gastric
PRODUCT INFORMATION ANAGRAINE COMPOSITION Each ANAGRAINE tablet contains 5 mg metoclopramide and 500 mg paracetamol. ACTIONS Metoclopramide stimulates gastrointestinal tract motility and accelerates gastric
AUSTRALIAN PRODUCT INFORMATION PANADOL OPTIZORB TABLETS (PARACETAMOL) TABLETS PANADOL OPTIZORB CAPLETS (PARACETAMOL) TABLETS
 AUSTRALIAN PRODUCT INFORMATION PANADOL OPTIZORB TABLETS (PARACETAMOL) TABLETS PANADOL OPTIZORB CAPLETS (PARACETAMOL) TABLETS 1 NAME OF THE MEDICINE Paracetamol 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
AUSTRALIAN PRODUCT INFORMATION PANADOL OPTIZORB TABLETS (PARACETAMOL) TABLETS PANADOL OPTIZORB CAPLETS (PARACETAMOL) TABLETS 1 NAME OF THE MEDICINE Paracetamol 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
Your Pharmacy Paracetamol Plus, capsule shaped tablets (caplets)
 New Zealand Data Sheet Product Description Your Pharmacy Plus, capsule shaped tablets (caplets) 500 mg per tablet & Codeine Phosphate 10 mg per tablet Blister packs of white, capsule-shaped tablets Composition
New Zealand Data Sheet Product Description Your Pharmacy Plus, capsule shaped tablets (caplets) 500 mg per tablet & Codeine Phosphate 10 mg per tablet Blister packs of white, capsule-shaped tablets Composition
AUSTRALIAN PRODUCT INFORMATION, PAINSTOP DAY-TIME PAIN RELIEVER (PARACETAMOL 120MG IN 5ML AND CODEINE PHOSPHATE HEMIHYDRATE 5MG IN 5ML)
 AUSTRALIAN PRODUCT INFORMATION, PAINSTOP DAY-TIME PAIN RELIEVER (PARACETAMOL 120MG IN 5ML AND CODEINE PHOSPHATE HEMIHYDRATE 5MG IN 5ML) 1. NAME OF THE MEDICINE Paracetamol and codeine phosphate hemihydrate.
AUSTRALIAN PRODUCT INFORMATION, PAINSTOP DAY-TIME PAIN RELIEVER (PARACETAMOL 120MG IN 5ML AND CODEINE PHOSPHATE HEMIHYDRATE 5MG IN 5ML) 1. NAME OF THE MEDICINE Paracetamol and codeine phosphate hemihydrate.
2. QUALITATIVE AND QUANTITATIVE COMPOSITION. Each capsule contains PARACETAMOL 500mg For a full list of excipients, see section 6.1.
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT PARACETAMOL 500mg CAPSULES Boots Paracetamol 500mg Capsules 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each capsule contains PARACETAMOL
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT PARACETAMOL 500mg CAPSULES Boots Paracetamol 500mg Capsules 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each capsule contains PARACETAMOL
PRODUCT INFORMATION. Codral* Original Cold & Flu Tablets
 PRODUCT INFORMATION Codral* Original Cold & Flu Tablets Product description Codral* Original Cold & Flu tablets contain paracetamol 500 mg, pseudoephedrine hydrochloride 30 mg and codeine phosphate 6 mg.
PRODUCT INFORMATION Codral* Original Cold & Flu Tablets Product description Codral* Original Cold & Flu tablets contain paracetamol 500 mg, pseudoephedrine hydrochloride 30 mg and codeine phosphate 6 mg.
PANADOL SINUS DAY & NIGHT TABLETS PRODUCT INFORMATION
 Product description PANADOL SINUS DAY tablet is a white, film-coated capsule-shaped tablet with flat edges, front and back faces marked with DAY. PANADOL SINUS NIGHT tablet is a white, film-coated round
Product description PANADOL SINUS DAY tablet is a white, film-coated capsule-shaped tablet with flat edges, front and back faces marked with DAY. PANADOL SINUS NIGHT tablet is a white, film-coated round
PARACOD Tablets (Paracetamol + Codeine phosphate)
 Published on: 22 Sep 2014 PARACOD Tablets (Paracetamol + Codeine phosphate) Composition PARACOD Tablets Each effervescent tablet contains: Paracetamol IP...650 mg Codeine Phosphate IP... 30 mg Dosage Form/s
Published on: 22 Sep 2014 PARACOD Tablets (Paracetamol + Codeine phosphate) Composition PARACOD Tablets Each effervescent tablet contains: Paracetamol IP...650 mg Codeine Phosphate IP... 30 mg Dosage Form/s
PACKAGE INSERT TEMPLATE FOR PARACETAMOL SUPPOSITORIES
 PACKAGE INSERT TEMPLATE FOR PARACETAMOL SUPPOSITORIES Brand or Product Name [Product name] Suppositories mg Name and Strength of Active Substance(s) Eg Paracetamol 500mg Paracetamol 250mg Paracetamol 125mg
PACKAGE INSERT TEMPLATE FOR PARACETAMOL SUPPOSITORIES Brand or Product Name [Product name] Suppositories mg Name and Strength of Active Substance(s) Eg Paracetamol 500mg Paracetamol 250mg Paracetamol 125mg
PRODUCT INFORMATION PANADOL COLD & FLU MAX HOT LEMON POWDER NAME OF THE MEDICINE. Chemical structure: CAS 2 Registry Number: DESCRIPTION
 PRODUCT INFORMATION PANADOL COLD & FLU MAX HOT LEMON POWDER NAME OF THE MEDICINE Active ingredient: Paracetamol Chemical structure: CAS 2 Registry Number: 103-90-2 DESCRIPTION Paracetamol is a white, crystalline
PRODUCT INFORMATION PANADOL COLD & FLU MAX HOT LEMON POWDER NAME OF THE MEDICINE Active ingredient: Paracetamol Chemical structure: CAS 2 Registry Number: 103-90-2 DESCRIPTION Paracetamol is a white, crystalline
NEW ZEALAND DATASHEET
 NEW ZEALAND DATASHEET COLDREX HOT REMEDY COLD & FLU HOT LEMON Powder for Oral Solution Paracetamol (BP) 1000mg/sachet Presentation Pale yellow, free flowing heterogeneous powder with and odour of lemon
NEW ZEALAND DATASHEET COLDREX HOT REMEDY COLD & FLU HOT LEMON Powder for Oral Solution Paracetamol (BP) 1000mg/sachet Presentation Pale yellow, free flowing heterogeneous powder with and odour of lemon
NEW ZEALAND DATA SHEET. Each Paracetamol + Codeine contains 500 mg of paracetamol and 8 mg of codeine phosphate.
 NEW ZEALAND DATA SHEET PARACETAMOL + CODEINE 1. Product Name Paracetamol + Codeine, 500 mg + 8 mg, tablets. 2. Qualitative and Quantitative Composition Each Paracetamol + Codeine contains 500 mg of paracetamol
NEW ZEALAND DATA SHEET PARACETAMOL + CODEINE 1. Product Name Paracetamol + Codeine, 500 mg + 8 mg, tablets. 2. Qualitative and Quantitative Composition Each Paracetamol + Codeine contains 500 mg of paracetamol
PRODUCT INFORMATION. Sudafed* Sinus Day + Night Relief Tablets
 PRODUCT INFORMATION Sudafed* Sinus Day + Night Relief Tablets Product description Sudafed* Sinus Day + Night Relief tablets contain two separate formulations: day tablets and night tablets. Each Sudafed*
PRODUCT INFORMATION Sudafed* Sinus Day + Night Relief Tablets Product description Sudafed* Sinus Day + Night Relief tablets contain two separate formulations: day tablets and night tablets. Each Sudafed*
PRODUCT INFORMATION APOHEALTH PARACETAMOL PLUS CODEINE & CALMATIVE paracetamol 500mg codeine phosphate 10mg and doxylamine succinate 5.
 Product Information Page 1 PRODUCT INFORMATION APOHEALTH PARACETAMOL PLUS CODEINE & CALMATIVE paracetamol 500mg codeine phosphate 10mg and doxylamine succinate 5.1mg tablets NAME OF THE MEDICINE Paracetamol
Product Information Page 1 PRODUCT INFORMATION APOHEALTH PARACETAMOL PLUS CODEINE & CALMATIVE paracetamol 500mg codeine phosphate 10mg and doxylamine succinate 5.1mg tablets NAME OF THE MEDICINE Paracetamol
Metabolism Paracetamol is metabolised in the liver and excreted in the urine mainly as glucuronide and sulphate conjugates.
 FEBRAMOL Composition Febramol 150 Suppositories Each suppository contains Paracetamol 150 mg. Suppositories, Tablets & Syrup Febramol 300 Suppositories Each suppository contains Paracetamol 300 mg. Each
FEBRAMOL Composition Febramol 150 Suppositories Each suppository contains Paracetamol 150 mg. Suppositories, Tablets & Syrup Febramol 300 Suppositories Each suppository contains Paracetamol 300 mg. Each
PANADOL COLD & FLU MAX HOT LEMON Powder for Oral Solution DATA SHEET
 PANADOL COLD & FLU MAX HOT LEMON Powder for Oral Solution Paracetamol (BP) 1000mg/sachet DATA SHEET Presentation Pale yellow, free flowing heterogeneous powder with and odour of lemon Indications Fast,
PANADOL COLD & FLU MAX HOT LEMON Powder for Oral Solution Paracetamol (BP) 1000mg/sachet DATA SHEET Presentation Pale yellow, free flowing heterogeneous powder with and odour of lemon Indications Fast,
PRODUCT INFORMATION. Active ingredients Chemical structure CAS Registry Number. Paracetamol
 PRODUCT INFORMATION PANADOL SINUS PAIN & CONGESTION RELIEF CAPLETS PANADOL COLD & FLU PLUS DECONGESTANT CAPLETS NAME OF THE MEDICINE Active ingredients Chemical structure CAS Registry Number Paracetamol
PRODUCT INFORMATION PANADOL SINUS PAIN & CONGESTION RELIEF CAPLETS PANADOL COLD & FLU PLUS DECONGESTANT CAPLETS NAME OF THE MEDICINE Active ingredients Chemical structure CAS Registry Number Paracetamol
NEW ZEALAND DATA SHEET
 NEW ZEALAND DATA SHEET 1. PRODUCT NAME PANADOL Extra (reformulation), Paracetamol 500 mg and Caffeine 65 mg, film coated tablet 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Active Ingredients: Paracetamol
NEW ZEALAND DATA SHEET 1. PRODUCT NAME PANADOL Extra (reformulation), Paracetamol 500 mg and Caffeine 65 mg, film coated tablet 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Active Ingredients: Paracetamol
PRODUCT INFORMATION. Sudafed* Sinus Day + Night Relief Tablets
 PRODUCT INFORMATION Sudafed* Sinus Day + Night Relief Tablets Name of the Medicine Paracetamol CAS 2 Registry Number: 103-90-2 Pseudoephedrine Hydrochloride CAS 2 Registry Number: 345-78-8 Triprolidine
PRODUCT INFORMATION Sudafed* Sinus Day + Night Relief Tablets Name of the Medicine Paracetamol CAS 2 Registry Number: 103-90-2 Pseudoephedrine Hydrochloride CAS 2 Registry Number: 345-78-8 Triprolidine
DATA SHEET. Paracetamol (BP) 500 mg and Phenylephrine Hydrochloride (BP) 5 mg
 COLDREX PE PHENYLEPHRINE SINUS COLDREX PE PHENYLEPHRINE CONGESTION CLEAR PANADOL SINUS PAIN & CONGESTION RELIEF PANADOL COLD & FLU MAX + DECONGESTANT DATA SHEET Paracetamol (BP) 500 mg and Phenylephrine
COLDREX PE PHENYLEPHRINE SINUS COLDREX PE PHENYLEPHRINE CONGESTION CLEAR PANADOL SINUS PAIN & CONGESTION RELIEF PANADOL COLD & FLU MAX + DECONGESTANT DATA SHEET Paracetamol (BP) 500 mg and Phenylephrine
PRODUCT INFORMATION APO-Paracetamol/Codeine 500/30
 PRODUCT INFORMATION APO-Paracetamol/Codeine 500/30 NAME OF THE MEDICINE Non-proprietary Name Paracetamol and codeine phosphate Chemical Structure Paracetamol MW 151.17 Codeine phosphate MW 406.37 CAS Numbers
PRODUCT INFORMATION APO-Paracetamol/Codeine 500/30 NAME OF THE MEDICINE Non-proprietary Name Paracetamol and codeine phosphate Chemical Structure Paracetamol MW 151.17 Codeine phosphate MW 406.37 CAS Numbers
Codeine Phosphate Hemihydrate. Tablet: Solpadol Caplets are white capsule shaped tablets, marked SOLPADOL on one side.
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Solpadol Caplets Solpadol Capsules Solpadol 30mg/500mg Effervescent Tablets 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Active Constituents
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Solpadol Caplets Solpadol Capsules Solpadol 30mg/500mg Effervescent Tablets 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Active Constituents
Chemists Own Strong Pain Extra Paracetamol and Codeine Phosphate
 Chemists Own Strong Pain Extra Paracetamol and Codeine Phosphate Consumer Medicine Information(CMI) Please read this information before you start taking this medicine. What is in this CMI? This CMI answers
Chemists Own Strong Pain Extra Paracetamol and Codeine Phosphate Consumer Medicine Information(CMI) Please read this information before you start taking this medicine. What is in this CMI? This CMI answers
Paracetamol, codeine phosphate hemihydrate and promethazine hydrochloride.
 AUSTRALIAN PRODUCT INFORMATION, PAINSTOP NIGHT-TIME PAIN RELIEVER (PARACETAMOL 120MG IN 5ML,CODEINE PHOSPHATE HEMIHYDRATE 5MG IN 5ML AND PROMETHAZINE HYDROCHLORIDE 6.5MG IN 5ML) 1. NAME OF THE MEDICINE
AUSTRALIAN PRODUCT INFORMATION, PAINSTOP NIGHT-TIME PAIN RELIEVER (PARACETAMOL 120MG IN 5ML,CODEINE PHOSPHATE HEMIHYDRATE 5MG IN 5ML AND PROMETHAZINE HYDROCHLORIDE 6.5MG IN 5ML) 1. NAME OF THE MEDICINE
RAFEN PLUS Ibuprofen 200mg/Codeine phosphate 12.8mg tablets PRODUCT INFORMATION
 RAFEN PLUS Ibuprofen 200mg/Codeine phosphate 12.8mg tablets PRODUCT INFORMATION Composition RAFEN PLUS tablet is a white to off-white capsule-shaped, biconvex tablet, containing ibuprofen 200mg and codeine
RAFEN PLUS Ibuprofen 200mg/Codeine phosphate 12.8mg tablets PRODUCT INFORMATION Composition RAFEN PLUS tablet is a white to off-white capsule-shaped, biconvex tablet, containing ibuprofen 200mg and codeine
APO-PARACETAMOL/CODEINE 500/30
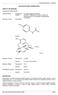 APO-PARACETAMOL/CODEINE 500/30 NAME OF THE MEDICINE Paracetamol/Codeine 500/30. Chemical Name: Paracetamol: N-(4-Hydroxyphenyl) acetamide Codeine: (5R, 6S)-7, 8-didehydro-4,5-epoxy-3-methoxy-Nmethylmorphinan-6-ol
APO-PARACETAMOL/CODEINE 500/30 NAME OF THE MEDICINE Paracetamol/Codeine 500/30. Chemical Name: Paracetamol: N-(4-Hydroxyphenyl) acetamide Codeine: (5R, 6S)-7, 8-didehydro-4,5-epoxy-3-methoxy-Nmethylmorphinan-6-ol
NEW ZEALAND DATA SHEET ACUPAN TM. 3. PHARMACEUTICAL FORM White, round, biconvex, film-coated tablets (7 mm diameter) engraved APN on one face.
 1. PRODUCT NAME ACUPAN 30 mg tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains nefopam hydrochloride 30 mg. For a full list of excipients, see section 6.1. 3. PHARMACEUTICAL FORM
1. PRODUCT NAME ACUPAN 30 mg tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains nefopam hydrochloride 30 mg. For a full list of excipients, see section 6.1. 3. PHARMACEUTICAL FORM
PRODUCT INFORMATION. Sudafed* Sinus + Allergy & Pain Relief Tablets
 PRODUCT INFORMATION Sudafed* Sinus + Allergy & Pain Relief Tablets Name of the Medicine Paracetamol CAS 2 Registry Number: 103-90-2 Pseudoephedrine Hydrochloride CAS 2 Registry Number: 345-78-8 Triprolidine
PRODUCT INFORMATION Sudafed* Sinus + Allergy & Pain Relief Tablets Name of the Medicine Paracetamol CAS 2 Registry Number: 103-90-2 Pseudoephedrine Hydrochloride CAS 2 Registry Number: 345-78-8 Triprolidine
SANDOMIGRAN (pizotifen malate)
 SANDOMIGRAN (pizotifen malate) S N CH 3 Pizotifen. COOH CH OH CH 2 COOH MALATE DESCRIPTION Pizotifen is a cycloheptathiophene derivative structurally related to cyproheptadine and the tricyclic antidepressants.
SANDOMIGRAN (pizotifen malate) S N CH 3 Pizotifen. COOH CH OH CH 2 COOH MALATE DESCRIPTION Pizotifen is a cycloheptathiophene derivative structurally related to cyproheptadine and the tricyclic antidepressants.
NOTOPAIN CAPLETS. Diclofenac Sodium + Paracetamol. Composition. Each tablet contains: Diclofenac Sodium BP 50mg Paracetamol BP 500mg.
 NOTOPAIN CAPLETS Diclofenac Sodium + Paracetamol Composition Each tablet contains: Diclofenac Sodium BP 50mg Paracetamol BP 500mg Pharmacology Phamacodynamics Diclofenac relieves pain and inflammation
NOTOPAIN CAPLETS Diclofenac Sodium + Paracetamol Composition Each tablet contains: Diclofenac Sodium BP 50mg Paracetamol BP 500mg Pharmacology Phamacodynamics Diclofenac relieves pain and inflammation
PRODUCT INFORMATION LEAFLET
 PRODUCT INFORMATION LEAFLET 1. Product Name Brand Name: Crocin Cold & Flu Max Generic Name: Paracetamol, Caffeine and Phenylephrine Hydrochloride Tablets 2. Qualitative & Quantitative Composition Each
PRODUCT INFORMATION LEAFLET 1. Product Name Brand Name: Crocin Cold & Flu Max Generic Name: Paracetamol, Caffeine and Phenylephrine Hydrochloride Tablets 2. Qualitative & Quantitative Composition Each
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT 2. QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT PARACETAMOL 500mg CAPSULES 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Paracetamol 500mg For the full list of excipients see section6.1.
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT PARACETAMOL 500mg CAPSULES 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Paracetamol 500mg For the full list of excipients see section6.1.
Summary of Product Characteristics
 1 NAME OF THE MEDICINAL PRODUCT Tipol 75mg Suppositories Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each suppository contains 75mg of paracetamol For a full list of excipients,
1 NAME OF THE MEDICINAL PRODUCT Tipol 75mg Suppositories Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each suppository contains 75mg of paracetamol For a full list of excipients,
CHILDREN S PANADOL COLOURFREE SUSPENSION PANADOL SUPPOSITORIES 125 MG PANADOL SUPPOSITORIES 250 MG DATA SHEET. Proprietary (Trade) Name: PANADOL
 CHILDREN S PANADOL COLOURFREE SUSPENSION PANADOL SUPPOSITORIES 125 MG PANADOL SUPPOSITORIES 250 MG DATA SHEET Proprietary (Trade) Name: PANADOL Active ingredient: Paracetamol (BP) 120 mg/5ml (Children
CHILDREN S PANADOL COLOURFREE SUSPENSION PANADOL SUPPOSITORIES 125 MG PANADOL SUPPOSITORIES 250 MG DATA SHEET Proprietary (Trade) Name: PANADOL Active ingredient: Paracetamol (BP) 120 mg/5ml (Children
PRODUCT INFORMATION. SUDAFED Sinus 12 Hour Relief Tablets
 PRODUCT INFORMATION SUDAFED Sinus 12 Hour Relief Tablets NAME OF THE MEDICINE Pseudoephedrine Hydrochloride CAS 2 Registry Number: 345-78-8 DESCRIPTION SUDAFED Sinus 12 Hour Relief prolonged-release tablets
PRODUCT INFORMATION SUDAFED Sinus 12 Hour Relief Tablets NAME OF THE MEDICINE Pseudoephedrine Hydrochloride CAS 2 Registry Number: 345-78-8 DESCRIPTION SUDAFED Sinus 12 Hour Relief prolonged-release tablets
PRODUCT INFORMATION PRODEINE FORTE H 3 CO. Paracetamol MW Codeine phosphate hemihydrate MW
 PRODUCT INFORMATION PRODEINE FORTE NAME OF THE MEDICINE Australian Approved Name Paracetamol and codeine phosphate hemihydrate Chemical Structure H N CH 3 HO O H.H 3 PO 4. 1/2 H 2 0 NH CH 3 H 3 CO Paracetamol
PRODUCT INFORMATION PRODEINE FORTE NAME OF THE MEDICINE Australian Approved Name Paracetamol and codeine phosphate hemihydrate Chemical Structure H N CH 3 HO O H.H 3 PO 4. 1/2 H 2 0 NH CH 3 H 3 CO Paracetamol
SCHEDULING STATUS: S0 For pack sizes of 24 tablets or less. For pack sizes of more than 24 tablets
 SCHEDULING STATUS: S0 For pack sizes of 24 tablets or less S1 For pack sizes of more than 24 tablets PROPRIETARY NAME: AND DOSAGE FORM PANADO MELTABS (Tablets) COMPOSITION: Each tablet contains 500 mg
SCHEDULING STATUS: S0 For pack sizes of 24 tablets or less S1 For pack sizes of more than 24 tablets PROPRIETARY NAME: AND DOSAGE FORM PANADO MELTABS (Tablets) COMPOSITION: Each tablet contains 500 mg
PRODUCT INFORMATION. Sudafed* Sinus + Anti-inflammatory Pain Relief Caplets
 PRODUCT INFORMATION Sudafed* Sinus + Anti-inflammatory Pain Relief Caplets Product description Sudafed* Sinus + Anti-inflammatory Pain Relief caplets contain pseudoephedrine hydrochloride 30 mg and ibuprofen
PRODUCT INFORMATION Sudafed* Sinus + Anti-inflammatory Pain Relief Caplets Product description Sudafed* Sinus + Anti-inflammatory Pain Relief caplets contain pseudoephedrine hydrochloride 30 mg and ibuprofen
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 1. Name of Medicinal Product Co-Codamol 8/500 mg Capsules 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Paracetamol 500.0 mg Codeine Phosphate 8.0 mg For excipients, see section 6.1 3 PHARMACEUTICAL FORM
1. Name of Medicinal Product Co-Codamol 8/500 mg Capsules 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Paracetamol 500.0 mg Codeine Phosphate 8.0 mg For excipients, see section 6.1 3 PHARMACEUTICAL FORM
PRODUCT INFORMATION. Active ingredients Chemical structure CAS Registry Number. Paracetamol
 PRODUCT INFORMATION PANADOL COLD & FLU MAX + DECONGESTANT HOT LEMON POWDER NAME OF THE MEDICINE Active ingredients Chemical structure CAS Registry Number Paracetamol 103-90-2 Phenylephrine Hydrochloride
PRODUCT INFORMATION PANADOL COLD & FLU MAX + DECONGESTANT HOT LEMON POWDER NAME OF THE MEDICINE Active ingredients Chemical structure CAS Registry Number Paracetamol 103-90-2 Phenylephrine Hydrochloride
PRODUCT INFORMATION MERSYNDOL FORTE TABLETS
 PRODUCT INFORMATION MERSYNDOL FORTE TABLETS NAME OF THE MEDICINE Non-proprietary Name Paracetamol, codeine phosphate and doxylamine succinate DESCRIPTION Each tablet contains paracetamol 450 mg, codeine
PRODUCT INFORMATION MERSYNDOL FORTE TABLETS NAME OF THE MEDICINE Non-proprietary Name Paracetamol, codeine phosphate and doxylamine succinate DESCRIPTION Each tablet contains paracetamol 450 mg, codeine
Metomax Paracetamol and Metoclopramide (as hydrochloride)
 Metomax Paracetamol and Metoclopramide (as hydrochloride) PRODUCT INFORMATION Name of the Medicine Active ingredients: Paracetamol Metoclopramide hydrochloride Structural formulas: Chemical names: N-(4-hydroxyphenyl)acetamide
Metomax Paracetamol and Metoclopramide (as hydrochloride) PRODUCT INFORMATION Name of the Medicine Active ingredients: Paracetamol Metoclopramide hydrochloride Structural formulas: Chemical names: N-(4-hydroxyphenyl)acetamide
Migraleve, Migraleve Pink and Migraleve Yellow Product Information
 Migraleve, Migraleve Pink and Migraleve Yellow Product Information Adverse events should be reported. Reporting forms and information can be found at www.mhra.gov.uk/yellowcard Adverse events should also
Migraleve, Migraleve Pink and Migraleve Yellow Product Information Adverse events should be reported. Reporting forms and information can be found at www.mhra.gov.uk/yellowcard Adverse events should also
PRODUCT INFORMATION ACTACODE
 PRODUCT INFORMATION ACTACODE NAME OF THE MEDICINE Actacode codeine linctus contains codeine phosphate 5 mg/ml. Codeine phosphate is (5R,6S)-7,8-didehydro-4,5-epoxy-3-methoxy-N-methylmorphinan-6-ol dihydrogen
PRODUCT INFORMATION ACTACODE NAME OF THE MEDICINE Actacode codeine linctus contains codeine phosphate 5 mg/ml. Codeine phosphate is (5R,6S)-7,8-didehydro-4,5-epoxy-3-methoxy-N-methylmorphinan-6-ol dihydrogen
2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Paracetamol 80mg Suppositories 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each suppository contains 80mg Paracetamol For a full list of
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Paracetamol 80mg Suppositories 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each suppository contains 80mg Paracetamol For a full list of
White capsule-shaped tablets (caplets) with flat edges, one face is embossed with sun graphic within an oval.
 PANADOL SINUS PAIN & CONGESTION RELIEF DAY & NIGHT CAPLETS DATA SHEET Paracetamol (BP) 500 mg, Phenylephrine Hydrochloride (BP) 5 mg, Chlorpheniramine Maleate (BP) 2 mg Presentation DAY Caplets: NIGHT
PANADOL SINUS PAIN & CONGESTION RELIEF DAY & NIGHT CAPLETS DATA SHEET Paracetamol (BP) 500 mg, Phenylephrine Hydrochloride (BP) 5 mg, Chlorpheniramine Maleate (BP) 2 mg Presentation DAY Caplets: NIGHT
Farmadol. Paracetamol 10 mg/ml INFUSION SOLUTION
 Farmadol Paracetamol 10 mg/ml INFUSION SOLUTION Composition Each ml contains: Paracetamol 10 mg Pharmacology Pharmacodynamic properties The precise mechanism of the analgesic and antipyretic properties
Farmadol Paracetamol 10 mg/ml INFUSION SOLUTION Composition Each ml contains: Paracetamol 10 mg Pharmacology Pharmacodynamic properties The precise mechanism of the analgesic and antipyretic properties
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT ALVERINE Mayoly Spindler, 60 mg hard capsules 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each capsule contains 60 mg of alverine citrate.
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT ALVERINE Mayoly Spindler, 60 mg hard capsules 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each capsule contains 60 mg of alverine citrate.
NEW ZEALAND DATA SHEET
 1. PRODUCT NAME Sudomyl, Tablet, 60 mg 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Name and strength of the active substance Pseudoephedrine Hydrochloride 60mg Excipient(s) with known effect For the full
1. PRODUCT NAME Sudomyl, Tablet, 60 mg 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Name and strength of the active substance Pseudoephedrine Hydrochloride 60mg Excipient(s) with known effect For the full
Each 5ml of Sinarest LP New Syrup contains: Phenylephrine
 Composition: Each 5ml of Sinarest LP New Syrup contains: Paracetamol Phenylephrine Levocetrizine 250mg 5mg 1.25mg Pharmacokinetic properties: Paracetamol is readily absorbed from the gastrointestinal tract
Composition: Each 5ml of Sinarest LP New Syrup contains: Paracetamol Phenylephrine Levocetrizine 250mg 5mg 1.25mg Pharmacokinetic properties: Paracetamol is readily absorbed from the gastrointestinal tract
DOMCET Tablet / Suspension (Domperidone maleate + Paracetamol)
 Published on: 23 Sep 2014 DOMCET Tablet / Suspension ( maleate + ) Composition Tablets Each film- coated tablet contains: Maleate equivalent to.. 10 mg.. 325 mg Suspension Each 5 ml of suspension contains:
Published on: 23 Sep 2014 DOMCET Tablet / Suspension ( maleate + ) Composition Tablets Each film- coated tablet contains: Maleate equivalent to.. 10 mg.. 325 mg Suspension Each 5 ml of suspension contains:
Summary of Product Characteristics
 1 NAME OF THE MEDICINAL PRODUCT Maxilief Effervescent Tablets Paracetamol 500mg Codeine Phosphate Hemihydrate 8mg Caffeine 30mg Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
1 NAME OF THE MEDICINAL PRODUCT Maxilief Effervescent Tablets Paracetamol 500mg Codeine Phosphate Hemihydrate 8mg Caffeine 30mg Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
PRODUCT INFORMATION. Active ingredients Chemical structure CAS Registry Number. Paracetamol
 PRODUCT INFORMATION PANADOL SINUS PAIN & CONGESTION RELIEF DAY & NIGHT CAPLETS NAME OF THE MEDICINE Active ingredients Chemical structure CAS Registry Number Paracetamol 103-90-2 Phenylephrine Hydrochloride
PRODUCT INFORMATION PANADOL SINUS PAIN & CONGESTION RELIEF DAY & NIGHT CAPLETS NAME OF THE MEDICINE Active ingredients Chemical structure CAS Registry Number Paracetamol 103-90-2 Phenylephrine Hydrochloride
PRODUCT INFORMATION. Ammonium chloride is an expectorant that has an irritant effect on mucous membranes.
 PRODUCT INFORMATION BENADRYL Original Oral Liquid (New Formula) Name of the Medicine Diphenhydramine hydrochloride Ammonium chloride The chemical name for diphenhydramine hydrochloride is 2-(diphenylmethoxy)-N,Ndimethylethanamine
PRODUCT INFORMATION BENADRYL Original Oral Liquid (New Formula) Name of the Medicine Diphenhydramine hydrochloride Ammonium chloride The chemical name for diphenhydramine hydrochloride is 2-(diphenylmethoxy)-N,Ndimethylethanamine
APO-Paracetamol/Codeine 500/30 Contains the active ingredients, paracetamol and codeine phosphate
 APO-Paracetamol/Codeine 500/30 Contains the active ingredients, paracetamol and codeine phosphate Consumer Medicine Information For a copy of a large print leaflet, Ph: 1800 195 055 What is in this leaflet
APO-Paracetamol/Codeine 500/30 Contains the active ingredients, paracetamol and codeine phosphate Consumer Medicine Information For a copy of a large print leaflet, Ph: 1800 195 055 What is in this leaflet
PROFESSIONAL INFORMATION
 SCHEDULING STATUS: S1 PROPRIETARY NAME AND DOSAGE FORM: Aleve Tablets COMPOSITION: Each tablet contains naproxen sodium 220 mg (equivalent to 200 mg naproxen) CATEGORY AND CLASS: A / 2.7 Antipyretic or
SCHEDULING STATUS: S1 PROPRIETARY NAME AND DOSAGE FORM: Aleve Tablets COMPOSITION: Each tablet contains naproxen sodium 220 mg (equivalent to 200 mg naproxen) CATEGORY AND CLASS: A / 2.7 Antipyretic or
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Comfora 595 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One film-coated tablet contains: glucosamine sulphate
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Comfora 595 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION One film-coated tablet contains: glucosamine sulphate
Evolution continues. Year: 1940 Fuel: Electricity Speed: 32 km/h. Year: 1998 Fuel: Electricity Speed: 118 km/h
 Year: 1940 Fuel: Electricity Speed: 32 km/h Year: 1998 Fuel: Electricity Speed: 118 km/h Year: 2008 Fuel: Electricity Speed: 210 km/h Year: 2016 Fuel: Electricity Speed: 250 km/h Evolution continues The
Year: 1940 Fuel: Electricity Speed: 32 km/h Year: 1998 Fuel: Electricity Speed: 118 km/h Year: 2008 Fuel: Electricity Speed: 210 km/h Year: 2016 Fuel: Electricity Speed: 250 km/h Evolution continues The
PRODUCT INFORMATION H 3 CO. paracetamol MW codeine phosphate hemihydrate MW
 RODUCT INFORMATION RODEINE NAME OF THE MEDICINE Non-proprietary Name aracetamol, codeine phosphate hemihydrate Chemical Structure H N CH 3 HO O H.H 3 O 4. 1/2 H 2 0 NH CH 3 H 3 CO paracetamol MW 151.17
RODUCT INFORMATION RODEINE NAME OF THE MEDICINE Non-proprietary Name aracetamol, codeine phosphate hemihydrate Chemical Structure H N CH 3 HO O H.H 3 O 4. 1/2 H 2 0 NH CH 3 H 3 CO paracetamol MW 151.17
SUMMARY OF PRODUCT CHARACTERISTICS 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Co-Codamol 8mg/500mg Tablets Boots Paracetamol and Codeine Tablets 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Paracetamol 500mg Codeine
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Co-Codamol 8mg/500mg Tablets Boots Paracetamol and Codeine Tablets 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Paracetamol 500mg Codeine
Summary of Product Characteristics
 1 NAME OF THE MEDICINAL PRODUCT Panadol Baby 120 mg/5 ml, Oral Suspension. Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each 5 ml spoonful of suspension contains paracetamol
1 NAME OF THE MEDICINAL PRODUCT Panadol Baby 120 mg/5 ml, Oral Suspension. Summary of Product Characteristics 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each 5 ml spoonful of suspension contains paracetamol
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Cyklonova 500 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Tranexamic acid 500 mg. For the full list of excipients,
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT Cyklonova 500 mg film-coated tablets 2. QUALITATIVE AND QUANTITATIVE COMPOSITION Tranexamic acid 500 mg. For the full list of excipients,
LEAFLET: INFORMATION FOR THE USER. Freshalgin 500th mg tablets Metamizole sodium (M etamizol sodium)
 LEAFLET: INFORMATION FOR THE USER Freshalgin 500th mg tablets Metamizole sodium (M etamizol sodium) Read this leaflet carefully before you start taking this medicine. This medicine is available without
LEAFLET: INFORMATION FOR THE USER Freshalgin 500th mg tablets Metamizole sodium (M etamizol sodium) Read this leaflet carefully before you start taking this medicine. This medicine is available without
1. What Solpadol is and what it is used for
 PATIENT INFORMATION LEAFLET SOLPADOL 30mg/500mg CAPLETS Codeine Phosphate and Paracetamol Read all of this leaflet carefully before you start taking this medicine. Keep this leaflet. You may need to read
PATIENT INFORMATION LEAFLET SOLPADOL 30mg/500mg CAPLETS Codeine Phosphate and Paracetamol Read all of this leaflet carefully before you start taking this medicine. Keep this leaflet. You may need to read
Immodium / loprarmide
 Immodium / loprarmide IMODIUM (loperamide hydrochloride) is indicated for the control and symptomatic relief of acute nonspecific diarrhea and of chronic diarrhea associated with inflammatory bowel disease.
Immodium / loprarmide IMODIUM (loperamide hydrochloride) is indicated for the control and symptomatic relief of acute nonspecific diarrhea and of chronic diarrhea associated with inflammatory bowel disease.
PRODUCT INFORMATION. AKINETON (biperiden hydrochloride 2 mg tablets)
 PRODUCT INFORMATION AKINETON (biperiden hydrochloride 2 mg tablets) NAME OF THE MEDICINE Non-proprietary name: biperiden hydrochloride Structural formula (biperiden): Chemical name (biperiden): α-5-norbornen-2-yl-α-phenyl-1-piperidine
PRODUCT INFORMATION AKINETON (biperiden hydrochloride 2 mg tablets) NAME OF THE MEDICINE Non-proprietary name: biperiden hydrochloride Structural formula (biperiden): Chemical name (biperiden): α-5-norbornen-2-yl-α-phenyl-1-piperidine
PRODUCT INFORMATION. Parke Davis* Day & Night Original Cough Cold & Flu Capsules
 PRODUCT INFORMATION Parke Davis* Day & Night Original Cough Cold & Flu Capsules Product description Parke Davis* Day & Night Original Cough Cold & Flu capsules contain two separate formulations: day capsules
PRODUCT INFORMATION Parke Davis* Day & Night Original Cough Cold & Flu Capsules Product description Parke Davis* Day & Night Original Cough Cold & Flu capsules contain two separate formulations: day capsules
New Zealand Data Sheet. Arrow-Bendrofluazide Bendroflumethiazide (also known as Bendrofluazide) Tablets 2.5mg and 5mg
 New Zealand Data Sheet Arrow-Bendrofluazide Bendroflumethiazide (also known as Bendrofluazide) Tablets 2.5mg and 5mg Description Arrow-Bendrofluazide Tablets contain 2.5 and 5 mg of the active ingredient
New Zealand Data Sheet Arrow-Bendrofluazide Bendroflumethiazide (also known as Bendrofluazide) Tablets 2.5mg and 5mg Description Arrow-Bendrofluazide Tablets contain 2.5 and 5 mg of the active ingredient
BRICANYL INJECTION. terbutaline sulfate PRODUCT INFORMATION
 BRICANYL INJECTION terbutaline sulfate PRODUCT INFORMATION NAME OF THE MEDICINE Terbutaline sulfate, 2-(tert-butylamino)-1-(3,5-dihydroxyphenyl) ethanol sulfate, a sympathomimetic bronchodilator with a
BRICANYL INJECTION terbutaline sulfate PRODUCT INFORMATION NAME OF THE MEDICINE Terbutaline sulfate, 2-(tert-butylamino)-1-(3,5-dihydroxyphenyl) ethanol sulfate, a sympathomimetic bronchodilator with a
SUMMARY OF PRODUCT CHARACTERISTICS
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT PARACETAMOL 500mg CAPSULES 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 500mg of paracetamol. Excipients with known
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT PARACETAMOL 500mg CAPSULES 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each tablet contains 500mg of paracetamol. Excipients with known
CONSUMER MEDICINE INFORMATION Please read this information before you start using this medicine.
 Panadol Sinus Pain & Congestion Relief Day & Night Caplets Day caplet: 500 mg Paracetamol / 5 mg Phenylephrine hydrochloride; Night caplet: 500 mg Paracetamol / 5 mg Phenylephrine hydrochloride / 2 mg
Panadol Sinus Pain & Congestion Relief Day & Night Caplets Day caplet: 500 mg Paracetamol / 5 mg Phenylephrine hydrochloride; Night caplet: 500 mg Paracetamol / 5 mg Phenylephrine hydrochloride / 2 mg
PANADOL Night Caplets 500mg Paracetamol & 25 mg Diphenhydramine Hydrochloride
 PANADOL Night Caplets 500mg Paracetamol & 25 mg Diphenhydramine Hydrochloride Consumer Medicine Information What is in this leaflet? This leaflet answers some common questions about PANADOL Night. It does
PANADOL Night Caplets 500mg Paracetamol & 25 mg Diphenhydramine Hydrochloride Consumer Medicine Information What is in this leaflet? This leaflet answers some common questions about PANADOL Night. It does
PRODUCT INFORMATION. Codral* Original Cold & Flu + Cough Day & Night Capsules
 PRODUCT INFORMATION Codral* Original Cold & Flu + Cough Day & Night Capsules Description Codral* Original Cold & Flu + Cough Day & Night capsules contain two separate formulations: day capsules and night
PRODUCT INFORMATION Codral* Original Cold & Flu + Cough Day & Night Capsules Description Codral* Original Cold & Flu + Cough Day & Night capsules contain two separate formulations: day capsules and night
PATIENT INFORMATION LEAFLET: Lancap 15 mg and 30 mg. Please read this leaflet carefully before taking LANCAP capsules.
 PATIENT INFORMATION LEAFLET: Lancap 15 mg and 30 mg Please read this leaflet carefully before taking LANCAP capsules. Keep this leaflet. You may need to read it again. If you have any further questions,
PATIENT INFORMATION LEAFLET: Lancap 15 mg and 30 mg Please read this leaflet carefully before taking LANCAP capsules. Keep this leaflet. You may need to read it again. If you have any further questions,
SUMMARY OF PRODUCT CHARACTERISTICS 2 QUALITATIVE AND QUANTITATIVE COMPOSITION
 SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Paracetamol 500mg Capsules Boots Paracetamol 500mg Capsules 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each capsule contains paracetamol
SUMMARY OF PRODUCT CHARACTERISTICS 1 NAME OF THE MEDICINAL PRODUCT Paracetamol 500mg Capsules Boots Paracetamol 500mg Capsules 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each capsule contains paracetamol
AUSTRALIAN PRODUCT INFORMATION APO-BETAHISTINE (BETAHISTINE DIHYDROCHLORIDE) 2 AND 3 QUALITATIVE AND QUANTITATIVE COMPOSITION AND PHARMACEUTICAL FORM
 AUSTRALIAN PRODUCT INFORMATION APO-BETAHISTINE (BETAHISTINE DIHYDROCHLORIDE) 1 NAME OF THE MEDICINE Betahistine dihydrochloride. 2 AND 3 QUALITATIVE AND QUANTITATIVE COMPOSITION AND PHARMACEUTICAL FORM
AUSTRALIAN PRODUCT INFORMATION APO-BETAHISTINE (BETAHISTINE DIHYDROCHLORIDE) 1 NAME OF THE MEDICINE Betahistine dihydrochloride. 2 AND 3 QUALITATIVE AND QUANTITATIVE COMPOSITION AND PHARMACEUTICAL FORM
MOVICOL Liquid Orange Flavour Concentrate for Oral Solution (macrogol 3350)
 MOVICOL Liquid Orange Flavour Concentrate for Oral Solution (macrogol 3350) NAME OF THE MEDICINE: MOVICOL Liquid Orange Flavour, Concentrate for Oral Solution. DESCRIPTION: A clear colourless solution.
MOVICOL Liquid Orange Flavour Concentrate for Oral Solution (macrogol 3350) NAME OF THE MEDICINE: MOVICOL Liquid Orange Flavour, Concentrate for Oral Solution. DESCRIPTION: A clear colourless solution.
CLINICAL PHARMACOLOGY
 robaxin / robaxin -7 50 (methocarbamol tablets, USP) Rx Only DESCRIPT ION robaxin /robaxin -750 (methocarbamol tablets, USP), a carbamate derivative of guaifenesin, is a central nervous system (CNS) depressant
robaxin / robaxin -7 50 (methocarbamol tablets, USP) Rx Only DESCRIPT ION robaxin /robaxin -750 (methocarbamol tablets, USP), a carbamate derivative of guaifenesin, is a central nervous system (CNS) depressant
TRAPADOL INJECTION FOR I.V./I.M. USE ONLY
 TRAPADOL INJECTION FOR I.V./I.M. USE ONLY Composition : Each 2ml. contains : Tramadol Hydrochloride I.P. Water for injection I.P. 100mg. q.s. CLINICAL PHARMACOLOGY : Pharmacodynamics Tramadol is a centrally
TRAPADOL INJECTION FOR I.V./I.M. USE ONLY Composition : Each 2ml. contains : Tramadol Hydrochloride I.P. Water for injection I.P. 100mg. q.s. CLINICAL PHARMACOLOGY : Pharmacodynamics Tramadol is a centrally
PRESCRIBING INFORMATION mg primaquine phosphate equivalent to 15 mg primaquine base. Antimalarial
 PRESCRIBING INFORMATION Pr PRIMAQUINE primaquine phosphate tablets USP 26.3 mg primaquine phosphate equivalent to 15 mg primaquine base Antimalarial sanofi-aventis Canada Inc. 2150 St. Elzear Blvd. West
PRESCRIBING INFORMATION Pr PRIMAQUINE primaquine phosphate tablets USP 26.3 mg primaquine phosphate equivalent to 15 mg primaquine base Antimalarial sanofi-aventis Canada Inc. 2150 St. Elzear Blvd. West
SUMMARY OF PRODUCT CHARACTERISTICS FOR BENZODIAZEPINES AS ANXIOLYTICS OR HYPNOTICS
 SUMMARY OF PRODUCT CHARACTERISTICS FOR BENZODIAZEPINES AS ANXIOLYTICS OR HYPNOTICS Guideline Title Summary of Product Characteristics for Benzodiazepines as Anxiolytics or Hypnotics Legislative basis Directive
SUMMARY OF PRODUCT CHARACTERISTICS FOR BENZODIAZEPINES AS ANXIOLYTICS OR HYPNOTICS Guideline Title Summary of Product Characteristics for Benzodiazepines as Anxiolytics or Hypnotics Legislative basis Directive
Nicotinic Acid Nicotinic Acid
 Nicotinic Acid Nicotinic Acid PRODUCT INFORMATION NAME OF THE MEDICINE Active ingredient: Nicotinic acid BP 250 mg. Structural formula: Molecular weight: 123.1 CAS Registry no.: 59-67-6 DESCRIPTION Inactive
Nicotinic Acid Nicotinic Acid PRODUCT INFORMATION NAME OF THE MEDICINE Active ingredient: Nicotinic acid BP 250 mg. Structural formula: Molecular weight: 123.1 CAS Registry no.: 59-67-6 DESCRIPTION Inactive
MESULID 100 REVISED PRODUCT INFORMATION
 MESULID 100 REVISED PRODUCT INFORMATION COMPOSITION Each caplet of MESULID 100 contains: Nimesulide 100 mg. Inactive Ingredients Lactose, microcrystalline cellulose, sodium starch glycolate, hydrogenated
MESULID 100 REVISED PRODUCT INFORMATION COMPOSITION Each caplet of MESULID 100 contains: Nimesulide 100 mg. Inactive Ingredients Lactose, microcrystalline cellulose, sodium starch glycolate, hydrogenated
PRODUCT INFORMATION. Benadryl* for the Family Nightime Oral Liquid
 Product description PRODUCT INFORMATION Benadryl* for the Family Nightime Oral Liquid Each 5 ml of Benadryl* Nightime oral liquid contains dextromethorphan hydrobromide 10 mg and diphenhydramine hydrochloride
Product description PRODUCT INFORMATION Benadryl* for the Family Nightime Oral Liquid Each 5 ml of Benadryl* Nightime oral liquid contains dextromethorphan hydrobromide 10 mg and diphenhydramine hydrochloride
Panadol Cold & Flu Relief + Cough Caplets Paracetamol 500mg, Dextromethorphan Hydrobromide 15mg, Phenylephrine Hydrochloride 5mg;
 Panadol Cold & Flu Relief + Cough Caplets Paracetamol 500mg, Dextromethorphan Hydrobromide 15mg, Phenylephrine Hydrochloride 5mg; Consumer Medicine Information What is in this leaflet This leaflet answers
Panadol Cold & Flu Relief + Cough Caplets Paracetamol 500mg, Dextromethorphan Hydrobromide 15mg, Phenylephrine Hydrochloride 5mg; Consumer Medicine Information What is in this leaflet This leaflet answers
