Feidhmeannacht na Seirbhíse Sláinte
|
|
|
- Anabel Lindsey
- 5 years ago
- Views:
Transcription
1 Feidhmeannacht na Seirbhíse Sláinte Health Service Executive Feidhmeannacht na Seirbhíse Sláinte Seirbhís Aisíocaíochta Cúraim Phríomhúil Bealach amach 5 an M50 An Bóthar Thuaidh Fionnghlas Baile Átha Cliath 11 D11 XKF3 Guthán: (01) Facs: (01) Health Service Executive Primary Care Reimbursement Service Exit 5, M50 North Road Finglas Dublin 11 D11 XKF3 Tel: (01) Fax: (01) Circular 046/16 16 th September 2016 Dear Doctor, The Medicines Management Programme (MMP) will be announcing two further preferred drugs this week in relation to beta blockers and calcium channel blockers. This follows a comprehensive review of the literature and a consultation period which provided valuable submissions from a variety of stakeholders. Beta Blocker preferred drug is Bisoprolol Calcium Channel Blocker preferred drug is Amlodipine A detailed document outlining the reasons for the selection of these preferred drugs along with Prescribing Tips will be published on the MMP website Further information on the Preferred Drugs Imitative and the MMP can also be found here. Please find enclosed a copy of communication from Professor Michael Barry, National Clinical Lead, Medicines Management Programme. I am also using this opportunity to enclose another document which may be of interest: A copy of a Clinical Advisory Note from the Early Warning Emerging Trends Subcommittee of the National Advisory Committee on Drugs and Alcohol. Thank you for your cooperation on this initiative. Yours faithfully, Anne Marie Hoey Primary Care Reimbursement & Eligibility
2 16/09/2016 Re: Preferred Drugs Initiative Dear Colleagues, The HSE Medicines Management Programmes (MMP) preferred drug initiative aims to promote safe, effective and cost-effective prescribing. We will be announcing two further preferred drugs later this week in relation to beta blockers and calcium channel blockers. This follows a comprehensive review of the literature and a consultation period during which we received valuable submissions from a variety of stakeholders. Factors which impacted on our choice of agent included the licensed therapeutic indication(s), clinical outcome data, international clinical guidelines, cost, patient-related factors and prescribing trends in Ireland. Beta Blockers: when initiating therapy with a beta blocker or if a change in therapy is being considered for patients with heart failure/angina or as add on therapy for hypertension: Preferred Beta Blocker is BISOPROLOL Calcium Channel Blockers (CCB): when initiating therapy with a calcium channel blocker or if a change in therapy is being considered for the treatment of hypertension/stable angina: Preferred CCB is AMLODIPINE A detailed document outlining the reasons for the selection of these preferred drugs along with Prescribing Tips will be published on the MMP website shortly. We hope that this document will be a useful reference source for prescribers. Choosing to prescribe the preferred drug will enhance quality prescribing while also delivering real savings for the healthcare system and for patients who pay for their medicines. We welcome your on-going feedback and support which will help to shape the development of the MMP with the aim of creating a prescribing resource for all prescribers. We have now named Preferred Drugs in 10 therapeutic areas. Statins SIMVASTATIN Proton Pump Inhibitor LANSOPRAZOLE ACE inhibitor RAMIPRIL ARB CANDESARTAN SSRI CITALOPRAM SNRI VENLAFAXINE Oral anticoagulant WARFARIN (NOAC: APIXABAN) Antimuscarinics TOLTERODINE SR Beta Blocker BISOPROLOL Calcium Channel Blocker AMLODIPINE I wish to thank you for your continued support of the Preferred Drug Initiative and for more information on the Medicines Management Programme please visit: With best wishes, Prof Michael Barry, National Clinical Lead, Medicines Management Programme
3 Clinical Advisory To: From: Prescribing Doctors, Pharmacists, Emergency Department Doctors and Emergency Department Nurses The Early Warning Emerging Trends Subcommittee of the National Advisory Committee on Drugs and Alcohol Date: June 2016 Subject: Medicines containing PREGABALIN and GABAPENTIN: being used abused and misused recreationally Dear Healthcare Professional, Call for vigilance and reporting The Early Warning and Emerging Trends (EWET) subcommittee 1, in agreement with the Department of Health wishes to highlight the need for vigilance when prescribing and dispensing pregabalin and gabapentin as both of these drugs present a risk of addiction and a potential for illegal diversion and medicinal misuse. Prescribers should always undertake a risk benefit assessment prior to prescribing either of these medicines for patients under their care. Summary The EWET subcommittee wishes to inform you of the serious concern regarding the misuse of prescription-only medicines containing PREGABALIN (Pregabalin, Lyrica, Brieka) and to a lesser extent, GABAPENTIN (Gabapentin, Gabin, Neurontin, Neurostil) and the exponential rise in prescribing rates of these medicines. Over the past 2 years the EWET has received a number of anecdotal reports on the misuse of pregabalin and to a lesser extent the misuse of gabapentin. Recently this information has been bolstered with evidenced based reports about the misuse of these medicines for recreational use and through their appearance in post mortem toxicology screening tests. Furthermore, dispensing data from the PCRS over the past 5 years shows a dramatic and continued increase in the volume of dispensed pregabalin, suggesting that those who are misusing these drugs may be sourcing them through legal as well as illegal channels. While this dispensing increase may be partly attributable to the greater number of authorised indications for pregabalin, it appears from the emerging evidence that the misuse of this drug may also be a significant factor in the escalating numbers of pregabalin items being dispensed in Ireland.
4 Background Pregabalin and gabapentin were originally developed and authorised as treatments for epilepsy. However, further research has shown that it can also be beneficial in treating other conditions, e.g. generalised anxiety disorder and neuropathic pain. Initially it was considered that pregabalin and gabapentin had a low potential for misuse, however the EWET Subcommittee is aware of concerns and reports that suggest that abuse of these medicines is on the increase. In 2013 there were 33 drug-related deaths in England and Wales where pregabalin was mentioned on the death certificate. Of 10 patients attending a Belfast hospital following recreational pregabalin abuse, six presented with seizures. Information as to the numbers of deaths related to pregabalin in Ireland is not available at this time. Evidence on the misuse of Pregabalin and harm potential A study into the potential for misuse of pregabalin was conducted by researchers at the HSE National Drug Treatment Centre Laboratory in Dublin between June and August The study found that of 440 people tested, 39 tested positive for pregabalin, representing 9.2% of the total sample, indicating that the misuse of pregabalin is a serious emerging issue. Only 10 patients from this group had been prescribed the drug. These findings have been published in the Irish Medical Journal Pregabalin Abuse amongst Opioid Substitution Treatment Patients 2. A report published in the UK Emergency Medical Journal in 2013 entitled Lyrica Nights- Recreational Pregabalin Abuse in an Urban Emergency Department J Millar, S Sadasivan, N Weatherup, S Lutton, which was based on the outcomes from a one year review of all patients presenting to the Emergency Department of the Royal Victoria Hospital, Belfast after recreational drug abuse. Those who admitted to pregabalin usage were identified and case notes were reviewed. The study found that in Belfast, emergency departments (ED) have witnessed a recent increase in the number of patients presenting after recreational abuse of pregabalin. Patients, state that the medication induces a state similar to drunkenness, hence the street name Budweiser's Patients are either taking tablets whole or cutting [, crushing] and snorting them. 60% of patients in this case series presented to the ED with seizures and 20% required ICU admission. Harm potential related to the recreational use of pregabalin and gabapentin include: physical dependencies; central nervous system depression, resulting in drowsiness, sedation, and respiratory depression; pregabalin and gabapentin related mortalities. The Therapeutics Today newsletter dated May 2013, No. 5, produced by the National Medicines Information Centre at St. James s Hospital (SJH) Dublin 8 and Dept of Therapeutics Trinity College,included a Focus on Pregabalin to highlight that patients may experience withdrawal symptoms after both short and long-term pregabalin therapy 3.
5 Known issues in other jurisdictions United Kingdom: In 2013, there were 19 deaths implicated with pregabalin and 17 deaths implicated with gabapentin in the UK. In September 2015, the UK Office for National Statistics (ONS) released registrations information on deaths related to drug related poisoning, which highlighted a significant increase in deaths from 2012 onwards. In 2014, there were 38 deaths where pregabalin was mentioned on the deceaseds death certificate; and, 26 deaths where gabapentin was mentioned on the deceaseds death certificate 4. Germany: Since 2008 pregabalin abuse and dependence has been reported with increasing frequency to a German medical regulatory body (BfArM) 5. Finland: The University of Helsinki has undertaken an assessment of pregabalin and gabapentin in opioid overdose deaths, noting that pregabalin abuse with high doses is increasingly common and can be fatal when combined with opioids. USA: In 2005 the Drug Enforcement Administration placed pregabalin under Schedule V of the Controlled Substances Act; citing that the abuse of pregabalin may lead to limited physical dependence or psychological dependence relative to the drugs or other substances in Schedule IV 6. Please remember that any suspected adverse events should be reported to the Health Products Regulatory Authority (formerly the Irish Medicines Board), via HPRA Pharmacovigilance, Earlsfort Terrace, IRL - Dublin 2; Tel: ; Fax: Website: medsafety@hpra.ie. References 1. Early Warning Emerging Trends Subcommittee (EWET) are a subcommittee of the National Advisory Committee on Drugs and Alcohol and comprises representation from the following organisations: o An Garda Síochána, o HSE Addiction Services, o Health Research Board, o Forensic Science Ireland, o Department of Justice and Equality, o Department of Health, o Medical Bureau of Road Safety, o State Laboratory, o HSE, o National Poisons Information Centre, o Revenue Customs Service, o Health Products Regulatory Authority, o Frontline Services Provider, o Academic Expert.
6 2. Pregabalin Abuse amongst Opioid Substitution Treatment Patients S McNamara, S Stokes, R Kilduff, A Shine, HSE National Drug Treatment Centre, McCarthy Centre, Pearse Street, Dublin 2; The Irish Medical Journal, November December 2015 Vol. 108 No Today2013/Therapeutics%20Today%20May%20% pdf 4. Advice_-Pregabalin_and_gabapentin.pdf
Reimbursement of Lidocaine 5% Plasters (Versatis ) effective 1 st September 2017
 Feidhmeannacht na Seirbhíse Sláinte, Seirbhís Aisíocaíochta Cúraim Phríomhúil Bealach amach 5 an M50, An Bóthar Thuaidh, Fionnghlas Baile Átha Cliath 11, D11 XKF3 Guthán: (01) 864 7100 Facs: (01) 834 3589
Feidhmeannacht na Seirbhíse Sláinte, Seirbhís Aisíocaíochta Cúraim Phríomhúil Bealach amach 5 an M50, An Bóthar Thuaidh, Fionnghlas Baile Átha Cliath 11, D11 XKF3 Guthán: (01) 864 7100 Facs: (01) 834 3589
Feidhmeannacht na Seirbhíse Sláinte
 Feidhmeannacht na Seirbhíse Sláinte Health Service Executive Feidhmeannacht na Seirbhíse Sláinte Seirbhís Aisíocaíochta Cúraim Phríomhúil Bealach amach 5 an M50 An Bóthar Thuaidh Fionnghlas Baile Átha
Feidhmeannacht na Seirbhíse Sláinte Health Service Executive Feidhmeannacht na Seirbhíse Sláinte Seirbhís Aisíocaíochta Cúraim Phríomhúil Bealach amach 5 an M50 An Bóthar Thuaidh Fionnghlas Baile Átha
Oral Nutritional Supplements (ONS)
 Feidhmeannacht na Seirbhíse Sláinte, Seirbhís Aisíocaíochta Cúraim Phríomhúil Bealach amach 5 an M50, An Bóthar Thuaidh, Fionnghlas Baile Átha Cliath 11, D11 XKF3 Guthán: (01) 864 7100 Facs: (01) 834 3589
Feidhmeannacht na Seirbhíse Sláinte, Seirbhís Aisíocaíochta Cúraim Phríomhúil Bealach amach 5 an M50, An Bóthar Thuaidh, Fionnghlas Baile Átha Cliath 11, D11 XKF3 Guthán: (01) 864 7100 Facs: (01) 834 3589
Circular May High Tech Hub Ordering and Management System
 Feidhmeannacht na Seirbhíse Sláinte, Seirbhís Aisíocaíochta Cúraim Phríomhúil Bealach amach 5 an M50, An Bóthar Thuaidh, Fionnghlas Baile Átha Cliath 11, D11 XKF3 Guthán: (01) 864 7100 Facs: (01) 834 3589
Feidhmeannacht na Seirbhíse Sláinte, Seirbhís Aisíocaíochta Cúraim Phríomhúil Bealach amach 5 an M50, An Bóthar Thuaidh, Fionnghlas Baile Átha Cliath 11, D11 XKF3 Guthán: (01) 864 7100 Facs: (01) 834 3589
Feidhmeannacht na Seirbhíse Sláinte
 Feidhmeannacht na Seirbhíse Sláinte Health Service Executive Feidhmeannacht na Seirbhíse Sláinte Seirbhís Aisíocaíochta Cúraim Phríomhúil Plás J5 Lárionad Gnó na Páirce Thuaidh Bealach Amach 5, M50 An
Feidhmeannacht na Seirbhíse Sláinte Health Service Executive Feidhmeannacht na Seirbhíse Sláinte Seirbhís Aisíocaíochta Cúraim Phríomhúil Plás J5 Lárionad Gnó na Páirce Thuaidh Bealach Amach 5, M50 An
Feidhmeannacht na Seirbhíse Sláinte
 Feidhmeannacht na Seirbhíse Sláinte Health Service Executive Feidhmeannacht na Seirbhíse Sláinte Seirbhís Aisíoca Príomhchúraim Bealach amach 5 an M50 An Bóthar Thuaidh Fionnghlas Baile Átha Cliath 11
Feidhmeannacht na Seirbhíse Sláinte Health Service Executive Feidhmeannacht na Seirbhíse Sláinte Seirbhís Aisíoca Príomhchúraim Bealach amach 5 an M50 An Bóthar Thuaidh Fionnghlas Baile Átha Cliath 11
Feidhmeannacht na Seirbhíse Sláinte
 Feidhmeannacht na Seirbhíse Sláinte Health Service Executive TITLE FORENAME SURNAME ADDRESS 1 ADDRESS 2 ADDRESS 3 COUNTY Feidhmeannacht na Seirbhíse Sláinte Seirbhís Aisíoca Príomhchúraim Bealach amach
Feidhmeannacht na Seirbhíse Sláinte Health Service Executive TITLE FORENAME SURNAME ADDRESS 1 ADDRESS 2 ADDRESS 3 COUNTY Feidhmeannacht na Seirbhíse Sláinte Seirbhís Aisíoca Príomhchúraim Bealach amach
Circular No. 028/ st December Dear Dentist,
 Feidhmeannacht na Seirbhíse Sláinte Health Service Executive Seirbhís Aisíoca Príomhchúraim Primary Care Reimbursement Service Aonaid 1-5 Urlár na Talún Units 1-5 Ground Floor Oifigí na Páirce Thuaidh
Feidhmeannacht na Seirbhíse Sláinte Health Service Executive Seirbhís Aisíoca Príomhchúraim Primary Care Reimbursement Service Aonaid 1-5 Urlár na Talún Units 1-5 Ground Floor Oifigí na Páirce Thuaidh
Medicines Management Programme Update
 Medicines Management Programme Update Sarah Clarke Chief II Pharmacist and Programme Manager Medicines Management Programme (MMP) www.hse.ie/yourmedicines Millions ( ) The Medicines Management Programme
Medicines Management Programme Update Sarah Clarke Chief II Pharmacist and Programme Manager Medicines Management Programme (MMP) www.hse.ie/yourmedicines Millions ( ) The Medicines Management Programme
What is pregabalin? Pregabalin tablets. Pregabalin misuse. National Drug Treatment Centre Research. Administration
 What is pregabalin? Pregabalin is a prescription drug used to manage a number of long-term conditions, including epilepsy, neuropathic pain and generalised anxiety disorder. Similar to benzodiazepines,
What is pregabalin? Pregabalin is a prescription drug used to manage a number of long-term conditions, including epilepsy, neuropathic pain and generalised anxiety disorder. Similar to benzodiazepines,
National Medicines Management Programme
 National Medicines Management Programme Michael Barry 28 th November 2013 The Medicines Management Programme Multi-disciplinary Medicines Management Programme (MMP) headed by the National Medicines Information
National Medicines Management Programme Michael Barry 28 th November 2013 The Medicines Management Programme Multi-disciplinary Medicines Management Programme (MMP) headed by the National Medicines Information
Feidhmeannacht na Seirbhíse Sláinte
 Feidhmeannacht na Seirbhíse Sláinte Health Service Executive Feidhmeannacht na Seirbhíse Sláinte Seirbhís Aisíocaíochta Cúraim Phríomhúil Plás J5 Lárionad Gnó na Páirce Thuaidh Bealach Amach 5, M50 An
Feidhmeannacht na Seirbhíse Sláinte Health Service Executive Feidhmeannacht na Seirbhíse Sláinte Seirbhís Aisíocaíochta Cúraim Phríomhúil Plás J5 Lárionad Gnó na Páirce Thuaidh Bealach Amach 5, M50 An
Primary Care Reimbursement Service. Statistical Analysis of Claims and Payments
 Primary Care Reimbursement Service Statistical Analysis of Claims and Payments Feidhmeannacht na Seirbhíse Sláinte Seirbhís Aisíoca Príomhchúraim Bealach amach 5 an M50 An Bóthar Thuaidh Fionnghlas Baile
Primary Care Reimbursement Service Statistical Analysis of Claims and Payments Feidhmeannacht na Seirbhíse Sláinte Seirbhís Aisíoca Príomhchúraim Bealach amach 5 an M50 An Bóthar Thuaidh Fionnghlas Baile
Medicines Management Programme Update
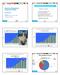 The Medicines Management Programme (MMP) Medicines Management Programme Update Michael Barry Clinical Lead HSE-Medicines Management Programme (MMP) www.hse.ie/yourmedicines What are our values? Ensuring
The Medicines Management Programme (MMP) Medicines Management Programme Update Michael Barry Clinical Lead HSE-Medicines Management Programme (MMP) www.hse.ie/yourmedicines What are our values? Ensuring
Re: Handling of gabapentin and pregabalin as Schedule 3 Controlled Drugs in health and justice commissioned services
 To: Health and justice commissioners and providers Public Health England health and justice leads Her Majesty s Prison and Probation Service Home Office immigration removal centre leads Health and justice
To: Health and justice commissioners and providers Public Health England health and justice leads Her Majesty s Prison and Probation Service Home Office immigration removal centre leads Health and justice
DRUG RELATED DEATHS AND PREGABALIN. Dr Abhishek Goli INSPIRE CGL
 DRUG RELATED DEATHS AND PREGABALIN Dr Abhishek Goli INSPIRE CGL INTRODUCTION 1. Overview on drug related deaths 2. Pregabalin and Gabapentin abuse potential 3. Benzos POTENTIAL FACTORS Increase in availability
DRUG RELATED DEATHS AND PREGABALIN Dr Abhishek Goli INSPIRE CGL INTRODUCTION 1. Overview on drug related deaths 2. Pregabalin and Gabapentin abuse potential 3. Benzos POTENTIAL FACTORS Increase in availability
Advice for prescribers on the risk of the misuse of pregabalin and gabapentin
 Advice for prescribers on the risk of the misuse of pregabalin and gabapentin Purpose of this advice This document has been produced by healthcare professionals with support from policy observers to provide:
Advice for prescribers on the risk of the misuse of pregabalin and gabapentin Purpose of this advice This document has been produced by healthcare professionals with support from policy observers to provide:
PRESCRIBING BY RADIOGRAPHERS: A VISION PAPER
 PRESCRIBING BY RADIOGRAPHERS: A VISION PAPER 1 INTRODUCTION 1.1 The Review of Prescribing, Supply & Administration of Medicines (Crown II) Final Report was submitted to The Secretary of State for Health
PRESCRIBING BY RADIOGRAPHERS: A VISION PAPER 1 INTRODUCTION 1.1 The Review of Prescribing, Supply & Administration of Medicines (Crown II) Final Report was submitted to The Secretary of State for Health
Non-Prescription Medicinal Products Containing Codeine: Guidance for Pharmacists on Safe Supply to Patients
 Non-Prescription Medicinal Products Containing Codeine: Guidance for Pharmacists on Safe Supply to Patients Pharmaceutical Society of Ireland Version 3 October 2017 Updates made following the enactment
Non-Prescription Medicinal Products Containing Codeine: Guidance for Pharmacists on Safe Supply to Patients Pharmaceutical Society of Ireland Version 3 October 2017 Updates made following the enactment
Safe Prescribing and Dispensing of Controlled Drugs. Joint Guidance Medical Council and Pharmaceutical Society of Ireland
 Safe Prescribing and Dispensing of Controlled Drugs Joint Guidance Medical Council and Pharmaceutical Society of Ireland Purpose of this Guidance Purpose of this Guidance This resource aims to facilitate
Safe Prescribing and Dispensing of Controlled Drugs Joint Guidance Medical Council and Pharmaceutical Society of Ireland Purpose of this Guidance Purpose of this Guidance This resource aims to facilitate
Substance use and misuse
 An open learning programme for pharmacists and pharmacy technicians Substance use and misuse Educational solutions for the NHS pharmacy workforce DLP 160 Contents iii About CPPE open learning programmes
An open learning programme for pharmacists and pharmacy technicians Substance use and misuse Educational solutions for the NHS pharmacy workforce DLP 160 Contents iii About CPPE open learning programmes
The Sustainability of Irish Pharmaceutical Expenditure. Valerie Walshe Ph.D. Economist
 The Sustainability of Irish Pharmaceutical Expenditure Valerie Walshe Ph.D. Economist valerie.walshe@hse.ie February 2015 Content Overview of Irish Health Services Community Drug Prescribing Trends Driving
The Sustainability of Irish Pharmaceutical Expenditure Valerie Walshe Ph.D. Economist valerie.walshe@hse.ie February 2015 Content Overview of Irish Health Services Community Drug Prescribing Trends Driving
Zofran tablets contain a medicine called ondansetron. This belongs to a group of medicines called anti-emetics.
 Package Leaflet: Information for the User Zofran 4 mg or 8 mg Film coated Tablets Ondansetron Read all of this leaflet carefully before you start taking this medicine because it contains important information
Package Leaflet: Information for the User Zofran 4 mg or 8 mg Film coated Tablets Ondansetron Read all of this leaflet carefully before you start taking this medicine because it contains important information
Cynthia B. Jones, Director Department of Medical Assistance Services (DMAS)
 Department of Medical Assistance Services 600 East Broad Street, Suite 1300 Richmond, Virginia 23219 MEDICAID MEMO http://www.dmas.state.va.us TO: All Prescribing Providers, Pharmacists, and Managed Care
Department of Medical Assistance Services 600 East Broad Street, Suite 1300 Richmond, Virginia 23219 MEDICAID MEMO http://www.dmas.state.va.us TO: All Prescribing Providers, Pharmacists, and Managed Care
medicines and side effects
 human medicines our advice on medicines and side effects medicines title and side effects Medicines can help people live longer and healthier lives. They can help cure or treat an illness or disease and
human medicines our advice on medicines and side effects medicines title and side effects Medicines can help people live longer and healthier lives. They can help cure or treat an illness or disease and
Service Level Agreement for the Provision of Level 1 Substance Misuse Services from a Community Pharmacy under contract to NHS Grampian
 Service Level Agreement for the Provision of Level 1 Substance Misuse Services from a Community Pharmacy under contract to NHS Grampian 1. Introduction The provision of Substance Misuse (SM) services through
Service Level Agreement for the Provision of Level 1 Substance Misuse Services from a Community Pharmacy under contract to NHS Grampian 1. Introduction The provision of Substance Misuse (SM) services through
PLEASE READ Important Patient Safety Information Approved by HPRA
 PLEASE READ Important Patient Safety Information Approved by HPRA Valproate (Epilim ): NEW restrictions on use PREGNANCY PREVENTION PROGRAMME to be put in place. 16 April 2018 Dear Healthcare professional,
PLEASE READ Important Patient Safety Information Approved by HPRA Valproate (Epilim ): NEW restrictions on use PREGNANCY PREVENTION PROGRAMME to be put in place. 16 April 2018 Dear Healthcare professional,
Ref: E 007. PGEU Response. Consultation on measures for improving the recognition of medical prescriptions issued in another Member State
 Ref:11.11.24E 007 PGEU Response Consultation on measures for improving the recognition of medical prescriptions issued in another Member State PGEU The Pharmaceutical Group of the European Union (PGEU)
Ref:11.11.24E 007 PGEU Response Consultation on measures for improving the recognition of medical prescriptions issued in another Member State PGEU The Pharmaceutical Group of the European Union (PGEU)
Guide to Interchangeable Medicines
 Guide to Interchangeable Medicines AUT-G0115-6 01 JUNE 2018 This guide does not purport to be an interpretation of law and/or regulations and is for guidance purposes only. CONTENTS 1 BACKGROUND 3 1.1
Guide to Interchangeable Medicines AUT-G0115-6 01 JUNE 2018 This guide does not purport to be an interpretation of law and/or regulations and is for guidance purposes only. CONTENTS 1 BACKGROUND 3 1.1
2004-L SEPTEMBER
 BULLETIN INTELLIGENCE Buprenorphine: Potential for Abuse Product No. 2004-L0424-013 SEPTEMBER 2004 U. S. D E P A R T M E N T O F J U S T I C E NDIC Within the past 2 years buprenorphine a Schedule III
BULLETIN INTELLIGENCE Buprenorphine: Potential for Abuse Product No. 2004-L0424-013 SEPTEMBER 2004 U. S. D E P A R T M E N T O F J U S T I C E NDIC Within the past 2 years buprenorphine a Schedule III
Appearance before House of Commons Standing Committee on Health as part of its Study on the Government s Role in Addressing Prescription Drug Abuse
 Opening Statement Appearance before House of Commons Standing Committee on Health as part of its Study on the Government s Role in Addressing Prescription Drug Abuse Dr. Chris Simpson, President-elect
Opening Statement Appearance before House of Commons Standing Committee on Health as part of its Study on the Government s Role in Addressing Prescription Drug Abuse Dr. Chris Simpson, President-elect
2 What you need to know before you use Zofran Suppositories
 Package Leaflet: Information for the User Zofran 16 mg Suppositories ondansetron Read all of this leaflet carefully before you start using this medicine because it contains important information for you.
Package Leaflet: Information for the User Zofran 16 mg Suppositories ondansetron Read all of this leaflet carefully before you start using this medicine because it contains important information for you.
PLEASE READ Important Patient Safety Information Approved by HPRA
 PLEASE READ Important Patient Safety Information Approved by HPRA Valproate (Epilim ): NEW restrictions on use PREGNANCY PREVENTION PROGRAMME to be put in place. 16 April 2018 Dear Pharmacist, This letter
PLEASE READ Important Patient Safety Information Approved by HPRA Valproate (Epilim ): NEW restrictions on use PREGNANCY PREVENTION PROGRAMME to be put in place. 16 April 2018 Dear Pharmacist, This letter
Package leaflet: Information for the user. Carmellose sodium
 Package leaflet: Information for the user CELLUVISC 0.5% w/v, eye drops, solution, unit dose Carmellose sodium Read all of this leaflet carefully before you start using this medicine because it contains
Package leaflet: Information for the user CELLUVISC 0.5% w/v, eye drops, solution, unit dose Carmellose sodium Read all of this leaflet carefully before you start using this medicine because it contains
PATIENT INFORMATION BOOKLET
 PATIENT INFORMATION BOOKLET GETTING STARTED WITH YOUR MEDICINES TIVICAY (dolutegravir) + lamivudine Tivicay is subject to additional monitoring. This will allow quick identification of new safety information.
PATIENT INFORMATION BOOKLET GETTING STARTED WITH YOUR MEDICINES TIVICAY (dolutegravir) + lamivudine Tivicay is subject to additional monitoring. This will allow quick identification of new safety information.
Zofran syrup contains a medicine called ondansetron. This belongs to a group of medicines called anti-emetics.
 Package Leaflet: Information for the User Zofran 4 mg/5 ml Syrup Ondansetron Read all of this leaflet carefully before you start taking this medicine because it contains important information for you.
Package Leaflet: Information for the User Zofran 4 mg/5 ml Syrup Ondansetron Read all of this leaflet carefully before you start taking this medicine because it contains important information for you.
REPORT OF THE MEDICAL BUREAU OF ROAD SAFETY FOR THE YEAR ENDED 31 ST DECEMBER 1999
 REPORT OF THE MEDICAL BUREAU OF ROAD SAFETY FOR THE YEAR ENDED 31 ST DECEMBER 1999 1 STAFF DETAILS The Department of Forensic Medicine/Medical Bureau of Road Safety currently has a total of sixteen members
REPORT OF THE MEDICAL BUREAU OF ROAD SAFETY FOR THE YEAR ENDED 31 ST DECEMBER 1999 1 STAFF DETAILS The Department of Forensic Medicine/Medical Bureau of Road Safety currently has a total of sixteen members
1. What Betahistine dihydrochloride tablets is and what it is used for
 Package leaflet: Information for the user Betahistine dihydrochloride 8 mg tablets Betahistine dihydrochloride 16 mg tablets Betahistine dihydrochloride Read all of this leaflet carefully before you start
Package leaflet: Information for the user Betahistine dihydrochloride 8 mg tablets Betahistine dihydrochloride 16 mg tablets Betahistine dihydrochloride Read all of this leaflet carefully before you start
solutions MEDICATION MIS MANAGEMENT and Chronic Pain 10/4/2016 Opioid Abuse: Current Data Opioid Abuse: Current Data
 MEDICATION MIS MANAGEMENT and Chronic Pain solutions Opioid Abuse: Current Data Americans consume 80% of the global supply of opioids This includes 99% of the world s hydrocodone and 2/3s of the world
MEDICATION MIS MANAGEMENT and Chronic Pain solutions Opioid Abuse: Current Data Americans consume 80% of the global supply of opioids This includes 99% of the world s hydrocodone and 2/3s of the world
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE SCOPE
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Drug Misuse: opiate detoxification of drug misusers in the community, hospital and prison. 1.1 Short title Drug misuse detoxification
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Drug Misuse: opiate detoxification of drug misusers in the community, hospital and prison. 1.1 Short title Drug misuse detoxification
Claire Gilbert, Tony Margetts, Gilda Nunez, Bryony Sedgwick, Tim Allison. Thanks to Hull Public Health, Prof. Marks and Paul Smith
 There is something in the heroin: responding to a rise in drug related deaths and the emergence of fentanyl and carfentanil in a local illicit drug market Claire Gilbert, Tony Margetts, Gilda Nunez, Bryony
There is something in the heroin: responding to a rise in drug related deaths and the emergence of fentanyl and carfentanil in a local illicit drug market Claire Gilbert, Tony Margetts, Gilda Nunez, Bryony
CONFIRMED INFLUENZA ISOLATES IN NORTHERN IRELAND - NICE GUIDANCE ON USE OF ANTIVIRALS NOW APPLIES
 From the Chief Medical Officer Dr Michael McBride HSS(MD) 34/2018 FOR ACTION Chief Executives, Public Health Agency/Health & Social Care Board/HSC Trusts/ NIAS GP Medical Advisers, Health & Social Care
From the Chief Medical Officer Dr Michael McBride HSS(MD) 34/2018 FOR ACTION Chief Executives, Public Health Agency/Health & Social Care Board/HSC Trusts/ NIAS GP Medical Advisers, Health & Social Care
Consultation on renaming the Local anaesthetics and Prescription only medicines annotations for chiropodists / podiatrists
 Consultation on renaming the Local anaesthetics and Prescription only medicines annotations for chiropodists / podiatrists 1. Introduction 1.1 We are the Health and Care Professions Council (HCPC). This
Consultation on renaming the Local anaesthetics and Prescription only medicines annotations for chiropodists / podiatrists 1. Introduction 1.1 We are the Health and Care Professions Council (HCPC). This
Guidance for Pharmacists on the Safe Supply of Non-Prescription Levonorgestrel 1500mcg for Emergency Hormonal Contraception
 Guidance for Pharmacists on the Safe Supply of Non-Prescription Levonorgestrel 1500mcg for Emergency Hormonal Contraception Pharmaceutical Society of Ireland Version 4 December 2016 Updates made following
Guidance for Pharmacists on the Safe Supply of Non-Prescription Levonorgestrel 1500mcg for Emergency Hormonal Contraception Pharmaceutical Society of Ireland Version 4 December 2016 Updates made following
Package leaflet: Information for the User Clarityn 10 mg tablets Loratadine
 Package leaflet: Information for the User Clarityn 10 mg tablets Loratadine Read all of this leaflet carefully before you start taking this medicine because it contains important information for you. Always
Package leaflet: Information for the User Clarityn 10 mg tablets Loratadine Read all of this leaflet carefully before you start taking this medicine because it contains important information for you. Always
Package leaflet: Information for the user. Lidocaine Hydrochloride 1% w/v Solution for Injection Lidocaine Hydrochloride
 Package leaflet: Information for the user Lidocaine Hydrochloride 1% w/v Solution for Injection Lidocaine Hydrochloride Read all of this leaflet carefully before you start using this medicine because it
Package leaflet: Information for the user Lidocaine Hydrochloride 1% w/v Solution for Injection Lidocaine Hydrochloride Read all of this leaflet carefully before you start using this medicine because it
Primary Health Networks
 Primary Health Networks Drug and Alcohol Treatment Activity Work Plan 2016-17 to 2018-19 Hunter New England & Central Coast Please note: This Activity Work Plan was developed in response to the HNECC PHN
Primary Health Networks Drug and Alcohol Treatment Activity Work Plan 2016-17 to 2018-19 Hunter New England & Central Coast Please note: This Activity Work Plan was developed in response to the HNECC PHN
Table of Contents Interim Report of the OxyContin Task Force, Newfoundland & Labrador, January 30, 2004
 OXYCONTIN TASK FORCE INTERIM REPORT January 30, 2004 Submitted to Hon. Elizabeth Marshall, Minister of Health & Community Services, Government of Newfoundland and Labrador Table of Contents INTRODUCTION
OXYCONTIN TASK FORCE INTERIM REPORT January 30, 2004 Submitted to Hon. Elizabeth Marshall, Minister of Health & Community Services, Government of Newfoundland and Labrador Table of Contents INTRODUCTION
NHS WALES PRIMARY CARE PRESCRIBING ANALYSIS FOR TRAMADOL DATA TO SEPTEMBER 2014
 NHS WALES PRIMARY CARE PRESCRIBING ANALYSIS FOR TRAMADOL DATA TO SEPTEMBER 2014 March 2015 This report has been prepared by the Welsh Analytical Prescribing Support Unit (WAPSU), part of the All Wales
NHS WALES PRIMARY CARE PRESCRIBING ANALYSIS FOR TRAMADOL DATA TO SEPTEMBER 2014 March 2015 This report has been prepared by the Welsh Analytical Prescribing Support Unit (WAPSU), part of the All Wales
MARYLAND BOARD OF PHARMACY
 MARYLAND BOARD OF PHARMACY CONTRACEPTION REGULATIONSAnd Other Legislative Initiatives THE ROAD FROM CONCEPTUALIZATION TO IMPLEMENTATION Deena Speights-Napata Executive Director Maryland Board of Pharmacy
MARYLAND BOARD OF PHARMACY CONTRACEPTION REGULATIONSAnd Other Legislative Initiatives THE ROAD FROM CONCEPTUALIZATION TO IMPLEMENTATION Deena Speights-Napata Executive Director Maryland Board of Pharmacy
Primary Care Reimbursement Service STATISTICAL ANALYSIS OF CLAIMS AND PAYMENTS 2010
 Primary Care Reimbursement Service STATISTICAL ANALYSIS OF CLAIMS AND PAYMENTS 2010 Feidhmeannacht na Seirbhíse Sláinte Seirbhís Aisíoca Príomhchúraim Bealach amach 5 an M50 An Bóthar Thuaidh Fionnghlas
Primary Care Reimbursement Service STATISTICAL ANALYSIS OF CLAIMS AND PAYMENTS 2010 Feidhmeannacht na Seirbhíse Sláinte Seirbhís Aisíoca Príomhchúraim Bealach amach 5 an M50 An Bóthar Thuaidh Fionnghlas
What is drug diversion?
 Learning Objectives Identify the impact of drug diversion on patients and healthcare organizations Explain the common points of risks and methods of drug diversion in healthcare s controlled substance
Learning Objectives Identify the impact of drug diversion on patients and healthcare organizations Explain the common points of risks and methods of drug diversion in healthcare s controlled substance
PYRIDOSTIGMINE LIME TM 60MG TABLETS Pyridostigmine bromide
 PACKAGE LEAFLET PYRIDOSTIGMINE LIME TM 60MG TABLETS Pyridostigmine bromide Patient Information Leaflet READ ALL OF THIS LEAFLET CAREFULLY BEFORE YOU START USING THIS MEDICINE. Keep this leaflet. You may
PACKAGE LEAFLET PYRIDOSTIGMINE LIME TM 60MG TABLETS Pyridostigmine bromide Patient Information Leaflet READ ALL OF THIS LEAFLET CAREFULLY BEFORE YOU START USING THIS MEDICINE. Keep this leaflet. You may
Modernized Reference Drug Program
 Modernized Reference Drug Program Monitoring Report For the period ending May 31, 2017 Issued: November 2017 Published by: Pharmaceutical Services Division and Health Sector Information, Analysis and Reporting
Modernized Reference Drug Program Monitoring Report For the period ending May 31, 2017 Issued: November 2017 Published by: Pharmaceutical Services Division and Health Sector Information, Analysis and Reporting
Dear DEA. Howard A. Heit, MD, FACP, FASAM,* Edward Covington, MD, and Patricia M. Good
 PAIN MEDICINE Volume 5 Number 3 2004,* Edward Covington, MD, and Patricia M. Good *Georgetown University, Washington, District of Columbia; Cleveland Clinic Foundation, Cleveland, Ohio; Office of Diversion
PAIN MEDICINE Volume 5 Number 3 2004,* Edward Covington, MD, and Patricia M. Good *Georgetown University, Washington, District of Columbia; Cleveland Clinic Foundation, Cleveland, Ohio; Office of Diversion
Chief Dental Officer England: Advice on commercial practice of online registered dental practitioners selling patient group directions for NHS
 Chief Dental Officer England: Advice on commercial practice of online registered dental practitioners selling patient group directions for NHS England commissioned services. Chief Dental Officer England:
Chief Dental Officer England: Advice on commercial practice of online registered dental practitioners selling patient group directions for NHS England commissioned services. Chief Dental Officer England:
TEEN EDUCATION. Medication Safety for Teens.
 Slide 1 Welcome to today s program, My Generation Rx: Medication Safety for Teens. You may be asking yourself, What is My Generation Rx? My Generation Rx encourages teens to incorporate the Generation
Slide 1 Welcome to today s program, My Generation Rx: Medication Safety for Teens. You may be asking yourself, What is My Generation Rx? My Generation Rx encourages teens to incorporate the Generation
Template Standard Operating Procedure For: Handling of Midazolam and other controlled drugs in Dental Practices
 Name of Dental Practice : Objectives To ensure implementation of the regulations and guidance on safe and secure handling of midazolam and other controlled drugs (CDs) Scope To cover all aspects of obtaining
Name of Dental Practice : Objectives To ensure implementation of the regulations and guidance on safe and secure handling of midazolam and other controlled drugs (CDs) Scope To cover all aspects of obtaining
Safe Medication Practices
 Safe medication practices for life. Slide 1 Safe Medication Practices Facilitator Talking Points Welcome to today s program, Generation Rx: Safe Medication Practices for Life. You may be asking yourself,
Safe medication practices for life. Slide 1 Safe Medication Practices Facilitator Talking Points Welcome to today s program, Generation Rx: Safe Medication Practices for Life. You may be asking yourself,
Package leaflet: Information for the patient. Heparin Sodium BP 1000 IU/L in 0.9% w/v Sodium Chloride IV Infusion. Active substance: Heparin Sodium
 Package leaflet: Information for the patient Heparin Sodium BP 1000 IU/L in 0.9% w/v Sodium Chloride IV Infusion Active substance: Heparin Sodium Read all of this leaflet carefully before you are given
Package leaflet: Information for the patient Heparin Sodium BP 1000 IU/L in 0.9% w/v Sodium Chloride IV Infusion Active substance: Heparin Sodium Read all of this leaflet carefully before you are given
Ontario s Narcotics Strategy
 Ontario s Narcotics Strategy Ontario Public Drug Programs Ministry of Health and Long-Term Care January 31, 2012 Ontario Harm Reduction Distribution Program Conference 2012 1 Background The Need for Action
Ontario s Narcotics Strategy Ontario Public Drug Programs Ministry of Health and Long-Term Care January 31, 2012 Ontario Harm Reduction Distribution Program Conference 2012 1 Background The Need for Action
THE EXPERT ADVISORY COMMITTEE ON DRUGS (EACD) ADVICE TO THE MINISTER ON:
 THE EXPERT ADVISORY COMMITTEE ON DRUGS (EACD) ADVICE TO THE MINISTER ON: BENZYLPIPERAZINE (BZP) April 2004 1 CONTENTS Executive Summary 3 Recommendations 4 Substances identification Similarity to known
THE EXPERT ADVISORY COMMITTEE ON DRUGS (EACD) ADVICE TO THE MINISTER ON: BENZYLPIPERAZINE (BZP) April 2004 1 CONTENTS Executive Summary 3 Recommendations 4 Substances identification Similarity to known
SUBJECT: Opioid Overdose and Intranasal Naloxone Training for Law Enforcement: Train the Trainer Session: Queens County October 17, 2014
 ANDREW M. CUOMO GOVERNOR STATE OF NEW YORK DIVISION OF CRIMINAL JUSTICE SERVICES Alfred E. Smith Office Building 80 South Swan Street Albany, New York 12210 http://criminaljustice.ny.gov MICHAEL C. GREEN
ANDREW M. CUOMO GOVERNOR STATE OF NEW YORK DIVISION OF CRIMINAL JUSTICE SERVICES Alfred E. Smith Office Building 80 South Swan Street Albany, New York 12210 http://criminaljustice.ny.gov MICHAEL C. GREEN
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE SCOPE
 NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Drug Misuse: opiate detoxification of drug misusers in the community, hospital and prison. 1.1 Short title Drug misuse detoxification
NATIONAL INSTITUTE FOR HEALTH AND CLINICAL EXCELLENCE 1 Guideline title SCOPE Drug Misuse: opiate detoxification of drug misusers in the community, hospital and prison. 1.1 Short title Drug misuse detoxification
Missouri Guidelines for the Use of Controlled Substances for the Treatment of Pain
 Substances for the Treatment of Pain Effective January 2007, the Board of Healing Arts appointed a Task Force to review the current statutes, rules and guidelines regarding the treatment of pain. This
Substances for the Treatment of Pain Effective January 2007, the Board of Healing Arts appointed a Task Force to review the current statutes, rules and guidelines regarding the treatment of pain. This
ACCG Mental Health Summit
 ACCG Mental Health Summit Sheila Pierce, Opioid Program Coordinator Director, Prescription Drug Management Program Discussion 1. Overview Opioid Problem in GA - Video 2. DPH Opioid Program 3. Priorities
ACCG Mental Health Summit Sheila Pierce, Opioid Program Coordinator Director, Prescription Drug Management Program Discussion 1. Overview Opioid Problem in GA - Video 2. DPH Opioid Program 3. Priorities
Minister s Opioid Emergency Response Commission Recommendations to the Minister Updated July 5, 2018
 The Minister s Opioid Emergency Response Commission was established May 31, 2017 to support the Government of Alberta s urgent response to the opioid crisis. As part of its mandate, the Commission is responsible
The Minister s Opioid Emergency Response Commission was established May 31, 2017 to support the Government of Alberta s urgent response to the opioid crisis. As part of its mandate, the Commission is responsible
VALPROATE. (Epilim, Depakote ) Patient Information Booklet
 VALPROATE (Epilim, Depakote ) Patient Information Booklet This booklet contains key information about the risks of valproate in pregnancy. This booklet is for you, if you are a girl or a woman taking any
VALPROATE (Epilim, Depakote ) Patient Information Booklet This booklet contains key information about the risks of valproate in pregnancy. This booklet is for you, if you are a girl or a woman taking any
Questionnaire 1 of 5: Cannabis Plant and Cannabis Resin. Introduction
 Introduction This is one of five questionnaires that Member States are invited to complete ahead of the 40th WHO Expert Committee on Drug Dependence. This questionnaire will ask you about cannabis plant
Introduction This is one of five questionnaires that Member States are invited to complete ahead of the 40th WHO Expert Committee on Drug Dependence. This questionnaire will ask you about cannabis plant
REAL TIME MONITORING OF PRESCRIPTION MEDICINES BEING A HEALTHY STATE
 BEING A HEALTHY STATE 2036 WILL MARK OUR STATE S BICENTENARY By the time our State turns 200 years old, I want South Australia to be a place of prosperity. Planning and delivering on my vision for a better
BEING A HEALTHY STATE 2036 WILL MARK OUR STATE S BICENTENARY By the time our State turns 200 years old, I want South Australia to be a place of prosperity. Planning and delivering on my vision for a better
A guide to essential gluten-free foods available from the pharmacy
 A guide to essential gluten-free foods available from the pharmacy This information guide has been produced by For further information: This resource has been reviewed by the British Dietetic Association.
A guide to essential gluten-free foods available from the pharmacy This information guide has been produced by For further information: This resource has been reviewed by the British Dietetic Association.
Volume 9; Number 3 March 2015 PRESCRIBING AND DISPENSING PREGABALIN FOR NEUROPATHIC PAIN
 Arden and Greater East Midlands Commissioning Support Unit in association with Lincolnshire Clinical Commissioning Groups, Lincolnshire Community Health Services, United Lincolnshire Hospitals Trust and
Arden and Greater East Midlands Commissioning Support Unit in association with Lincolnshire Clinical Commissioning Groups, Lincolnshire Community Health Services, United Lincolnshire Hospitals Trust and
ALCOHOL RELATED DEATHS REGISTERED IN NORTHERN IRELAND ( )
 ALCOHOL RELATED DEATHS REGISTERED IN NORTHERN IRELAND (1999-2009) 9.30am Thursday, 16 December 2010 Introduction 1. This report looks at the most recent official death registration data available on alcohol
ALCOHOL RELATED DEATHS REGISTERED IN NORTHERN IRELAND (1999-2009) 9.30am Thursday, 16 December 2010 Introduction 1. This report looks at the most recent official death registration data available on alcohol
SHARED CARE AGREEMENT: MELATONIN (CHILDREN)
 NB: This document should be read in conjunction with the current Summary of Product Characteristics (SPC) where appropriate. DRUG AND INDICATION: Generic drug name: Formulations: MELATONIN 3mg immediate
NB: This document should be read in conjunction with the current Summary of Product Characteristics (SPC) where appropriate. DRUG AND INDICATION: Generic drug name: Formulations: MELATONIN 3mg immediate
Prescribing of anti-osteoporotic therapies following the use of Proton Pump Inhibitors in general practice
 Prescribing of anti-osteoporotic therapies following the use of Proton Pump Inhibitors in general practice B. McGowan, K. Bennett, J. Feely Department of Pharmacology & Therapeutics, Trinity Centre for
Prescribing of anti-osteoporotic therapies following the use of Proton Pump Inhibitors in general practice B. McGowan, K. Bennett, J. Feely Department of Pharmacology & Therapeutics, Trinity Centre for
Course Outline. Code: PAR302 Title: Substance Misuse and Toxicology
 Faculty: School: Teaching Session: Semester 2 Year: 2018 Course Outline Code: PAR302 Title: Substance Misuse and Toxicology Science, Health, Education and Engineering Nursing, Midwifery & Paramedicine
Faculty: School: Teaching Session: Semester 2 Year: 2018 Course Outline Code: PAR302 Title: Substance Misuse and Toxicology Science, Health, Education and Engineering Nursing, Midwifery & Paramedicine
Substance-Exposed Newborn
 Substance-Exposed Newborn Definition: Substance-exposed newborn as a maltreatment occurs when a child is exposed to a controlled substance or alcohol prenatally. Exposure to a controlled substance or alcohol
Substance-Exposed Newborn Definition: Substance-exposed newborn as a maltreatment occurs when a child is exposed to a controlled substance or alcohol prenatally. Exposure to a controlled substance or alcohol
FATAL POISONINGS BY MEDICINAL OPIOIDS IN FINLAND
 FATAL POISONINGS BY MEDICINAL OPIOIDS IN FINLAND ERKKI VUORI 12.-13.11.2012 HJELT INSTITUUTTI OIKEUSLÄÄKETIETEEN OSASTO FINLAND IS AN IDEAL COUNTRY FOR POST- MORTEM TOXICOLOGICAL INVESTIGATIONS High autopsy
FATAL POISONINGS BY MEDICINAL OPIOIDS IN FINLAND ERKKI VUORI 12.-13.11.2012 HJELT INSTITUUTTI OIKEUSLÄÄKETIETEEN OSASTO FINLAND IS AN IDEAL COUNTRY FOR POST- MORTEM TOXICOLOGICAL INVESTIGATIONS High autopsy
Package leaflet: Information for the patient. Selincro 18 mg film-coated tablets nalmefene
 Package leaflet: Information for the patient Selincro 18 mg film-coated tablets nalmefene Read all of this leaflet carefully before you start taking this medicine because it contains important information
Package leaflet: Information for the patient Selincro 18 mg film-coated tablets nalmefene Read all of this leaflet carefully before you start taking this medicine because it contains important information
Kansas Department of Health and Environment (KDHE) Kansas Data-Driven Prevention Initiative Request for Proposal (RFP) Fiscal Year 2019
 Kansas Department of Health and Environment (KDHE) Kansas Data-Driven Prevention Initiative Request for Proposal (RFP) Fiscal Year 2019 Kansas Department of Health and Environment Bureau of Health Promotion
Kansas Department of Health and Environment (KDHE) Kansas Data-Driven Prevention Initiative Request for Proposal (RFP) Fiscal Year 2019 Kansas Department of Health and Environment Bureau of Health Promotion
Summary of Product Characteristics
 Summary of Product Characteristics 1 NAME OF THE MEDICINAL PRODUCT Atrosan Devil's Claw film-coated tablets 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each film-coated tablet contains 480 mg of extract
Summary of Product Characteristics 1 NAME OF THE MEDICINAL PRODUCT Atrosan Devil's Claw film-coated tablets 2 QUALITATIVE AND QUANTITATIVE COMPOSITION Each film-coated tablet contains 480 mg of extract
Take Home Naloxone Report on supply and use to reverse an overdose
 Take Home Naloxone Report on supply and use to reverse an overdose 2012-2016 1 Contents 1. Introduction 2. How the Take Home Naloxone Programme works 3. How information is collected 4. Patients supplied
Take Home Naloxone Report on supply and use to reverse an overdose 2012-2016 1 Contents 1. Introduction 2. How the Take Home Naloxone Programme works 3. How information is collected 4. Patients supplied
Prescribing Dilemmas. Sue Mulvenna Head of Pharmacy and CD Accountable Officer South Region SW 19 th May
 Prescribing Dilemmas Sue Mulvenna Head of Pharmacy and CD Accountable Officer South Region SW 19 th May 2016 The four principles of medicines optimisation Aim to understand the patient s experience Evidence
Prescribing Dilemmas Sue Mulvenna Head of Pharmacy and CD Accountable Officer South Region SW 19 th May 2016 The four principles of medicines optimisation Aim to understand the patient s experience Evidence
Medicinal cannabis. What is medicinal cannabis? What are cannabinoids? The endocannabinoid system
 Medicinal cannabis What is medicinal cannabis? Broadly speaking, medicinal cannabis is cannabis prescribed to relieve the symptoms of a medical condition, such as epilepsy. It is important to make the
Medicinal cannabis What is medicinal cannabis? Broadly speaking, medicinal cannabis is cannabis prescribed to relieve the symptoms of a medical condition, such as epilepsy. It is important to make the
Sodium Chloride 0.9% w/v Intravenous Infusion BP Solution for Infusion Sodium chloride
 Package leaflet: Information for the user Sodium Chloride 0.9% w/v Intravenous Infusion BP Solution for Infusion Sodium chloride Read all of this leaflet carefully before using this medicine because it
Package leaflet: Information for the user Sodium Chloride 0.9% w/v Intravenous Infusion BP Solution for Infusion Sodium chloride Read all of this leaflet carefully before using this medicine because it
Alcohol and Drug Commissioning Framework for Northern Ireland Consultation Questionnaire.
 Alcohol and Drug Commissioning Framework for Northern Ireland 2013-16 Consultation Questionnaire. This questionnaire has been designed to help stakeholders respond to the above framework. Written responses
Alcohol and Drug Commissioning Framework for Northern Ireland 2013-16 Consultation Questionnaire. This questionnaire has been designed to help stakeholders respond to the above framework. Written responses
Drug Misuse and Dependence Guidelines on Clinical Management
 Department of Health Scottish Office Department of Health Welsh Office Department of Health and Social Services, Northern Ireland Drug Misuse and Dependence Guidelines on Clinical Management An Executive
Department of Health Scottish Office Department of Health Welsh Office Department of Health and Social Services, Northern Ireland Drug Misuse and Dependence Guidelines on Clinical Management An Executive
Package leaflet: Information for the patient. Konakion MM Ampoules 10mg/ml solution for injection and oral solution Phytomenadione (vitamin K 1 )
 Package leaflet: Information for the patient Konakion MM Ampoules 10mg/ml solution for injection and oral solution Phytomenadione (vitamin K 1 ) Roche Read all of this leaflet carefully before you start
Package leaflet: Information for the patient Konakion MM Ampoules 10mg/ml solution for injection and oral solution Phytomenadione (vitamin K 1 ) Roche Read all of this leaflet carefully before you start
Information & statistics related to alcohol & drug misuse and community pharmacybased brief advice & intervention
 Information & statistics related to alcohol & drug misuse and community pharmacybased brief advice & intervention 1 INTRODUCTION Alcohol and Drug Misuse and their related harms cost our society hundreds
Information & statistics related to alcohol & drug misuse and community pharmacybased brief advice & intervention 1 INTRODUCTION Alcohol and Drug Misuse and their related harms cost our society hundreds
Package leaflet: Information for the user. Colecalciferol 3000 IU/ml Oral Solution (colecalciferol)
 Package leaflet: Information for the user Colecalciferol 3000 IU/ml Oral Solution (colecalciferol) Read all of this leaflet carefully before you start taking this medicine because it contains important
Package leaflet: Information for the user Colecalciferol 3000 IU/ml Oral Solution (colecalciferol) Read all of this leaflet carefully before you start taking this medicine because it contains important
Package leaflet: Information for the user. Ondansetron 4mg/5ml Syrup (ondansetron as ondansetron hydrochloride dihydrate)
 Package leaflet: Information for the user Ondansetron 4mg/5ml Syrup (ondansetron as ondansetron hydrochloride dihydrate) Read all of this leaflet carefully before you start taking this medicine because
Package leaflet: Information for the user Ondansetron 4mg/5ml Syrup (ondansetron as ondansetron hydrochloride dihydrate) Read all of this leaflet carefully before you start taking this medicine because
Patient and Family Agreement on Opioids
 Patient and Family Agreement on Opioids We care about our patients and are committed to their recovery and wellness. We offer our patients medications and options for various services to keep them from
Patient and Family Agreement on Opioids We care about our patients and are committed to their recovery and wellness. We offer our patients medications and options for various services to keep them from
PACKAGE LEAFLET: INFORMATION FOR THE USER Paracodin 0.20% w/w Syrup (dihydrocodeine hydrogen tartrate)
 PACKAGE LEAFLET: INFORMATION FOR THE USER Paracodin 0.20% w/w Syrup (dihydrocodeine hydrogen tartrate) Read all of this leaflet carefully before you start taking this medicine because it contains important
PACKAGE LEAFLET: INFORMATION FOR THE USER Paracodin 0.20% w/w Syrup (dihydrocodeine hydrogen tartrate) Read all of this leaflet carefully before you start taking this medicine because it contains important
Provider Education & Utilization Initiatives Alliance of States with PMPs East Regional Meeting
 Provider Education & Utilization Initiatives Alliance of States with PMPs East Regional Meeting Baltimore, MD April 5, 2011 Massachusetts Department of Public Health MA Prescription Monitoring Program
Provider Education & Utilization Initiatives Alliance of States with PMPs East Regional Meeting Baltimore, MD April 5, 2011 Massachusetts Department of Public Health MA Prescription Monitoring Program
Less pain in my life helps me get back to living.
 Eligible patients pay as little as $0 for BELBUCA Less pain in my life helps me get back to living. BELBUCA is: RETHINK RELIEF A strong prescription pain medicine that contains an opioid (narcotic) that
Eligible patients pay as little as $0 for BELBUCA Less pain in my life helps me get back to living. BELBUCA is: RETHINK RELIEF A strong prescription pain medicine that contains an opioid (narcotic) that
PACKAGE LEAFLET: INFORMATION FOR THE USER. Glucose Intravenous Infusion BP 10% w/v solution for infusion Glucose (as glucose monohydrate)
 PACKAGE LEAFLET: INFORMATION FOR THE USER Glucose Intravenous Infusion BP 10% w/v solution for infusion Glucose (as glucose monohydrate) Read all of this leaflet carefully before you start using this medicine
PACKAGE LEAFLET: INFORMATION FOR THE USER Glucose Intravenous Infusion BP 10% w/v solution for infusion Glucose (as glucose monohydrate) Read all of this leaflet carefully before you start using this medicine
Consent to research. A draft for consultation
 Consent to research A draft for consultation 1 Consent to research About the guidance Our guidance Consent: patients and doctors making decisions together (2008) 1 sets out the principles of good practice
Consent to research A draft for consultation 1 Consent to research About the guidance Our guidance Consent: patients and doctors making decisions together (2008) 1 sets out the principles of good practice
Learn how MORPHABOND ER can help manage your pain
 Your healthcare provider has prescribed Learn how can help manage your pain What is? MORPHABOND ER (morphine sulfate) extended-release tablets, CII is a strong prescription pain medicine that contains
Your healthcare provider has prescribed Learn how can help manage your pain What is? MORPHABOND ER (morphine sulfate) extended-release tablets, CII is a strong prescription pain medicine that contains
H NDS-ONHealth. Prescription Drug Abuse. Drug overdose death rates in the United States have more than tripled since 1990 and have never been higher.
 H NDS-ONHealth Health Wave Newsletter, October 2013 Visit us on our website at www.healthwaveinc.com Drug overdose death rates in the United States have more than tripled since 1990 and have never been
H NDS-ONHealth Health Wave Newsletter, October 2013 Visit us on our website at www.healthwaveinc.com Drug overdose death rates in the United States have more than tripled since 1990 and have never been
