CYP2D6: mirtazapine 2001/2002/2003
|
|
|
- Steven Pitts
- 5 years ago
- Views:
Transcription
1 CYP2D6: mirtazapine 2001/2002/200 Cl or = oral clearance,=c ss = steady state concentration, EM = extensive metaboliser, IM = intermediate metaboliser, MR = metabolic ratio, NS = non-significant, PM = poor metaboliser, S = significant, UM = ultra-rapid metaboliser The table below follows the KNMP definitions for EM, PM, IM and UM. Therefore, the definitions of EM, PM, IM and UM used in the table below may differ from the definition used by the authors in the article. Source Code Effect Comments ref. 1 Ramaekers JG et al. Residual effects of esmirtazapine on actual driving performance: overall findings and an exploratory analysis into the role of CYP2D6 phenotype. Psychopharmacology 2011;215: PM: B A total of 2 healthy volunteers in a cross-over study received S-mirtazapine 1.5 or.5 mg or placebo in the evening for 7 days. The deviation in distance from the side of the lane in a driving ability test was measured eleven hours after the first and last dose. Relevant co-medication was excluded. Phenotyping: 7x PM, 25x EM+IM+UM. PM versus EM+IM+UM: - increase in the percentage of volunteers who stopped prematurely due to mirtazapine-related adverse events (from 0% to 1%) (NS) - for both phenotypes, an effect on the ability to drive comparable to an alcohol concentration of 0.5 mg/ml cannot be ruled out following a single dose of.5 mg - for PM, an effect on the ability to drive comparable to an alcohol concentration of 0.5 mg/ml cannot be ruled out following a single dose of 1.5 mg or a repeated dose of.5 mg; this can be ruled out for EM+IM+UM - increase in the plasma concentration on day 8 by 80% (from 0.5 to 0.97 ng/ml) at a dose of 1.5 mg/day (NS) - increase in the plasma concentration on day 8 by 9% (from 1.52 to 2.12 ng/ml) at a dose of.5 mg/day (NS) NOTE: Genotype unknown. Exploratory analysis in a small group of poor CYP 2D6 metabolizers suggested that these subjects are more sensitive to the impairing effects of esmirtazapine on car driving. mirtazapine versus EM+IM+UM: PM: % ref. 2 Borobia AM et al. Influence of sex and CYP2D6 genotype on mirtazapine disposition, evaluated in Spanish healthy volunteers. Pharmacol Res 2009;59: NOTE: The doses used (intended for insomnia) are much lower than the doses used for depression. A total of 68 healthy volunteers received a single dose of mirtazapine 0 mg in both parts of a crossover study (various, bio-equivalent formulations). Co-medication was excluded. Genotyping: - x EM (2x *1/*1, 1x (*1/*)x2, 1x *1/*9) - 26x IM (2x gene dose 1 (18x *1/*, 2x *1/*5, x *1/*6) and 2x gene dose 0.5 (1x */*9, 1x */*9)) - 7x PM (6x */*, 1x */*6) - 1x UM ((*1/*1)x2) Gene dose 1 versus EM+UM: - AUC decreased by 16% (from to ng.hour/ml) (NS) - increase in the dose-corrected and weightcorrected AUC, predicted using a Both CYP2D6 genotype group and sex influence the disposition of mirtazapine in healthy volunteers. 1
2 ref. 2, continuation IM+PM: A pharmacokinetic model, by 6.% (from to ng.hour/ml) (S for the trend) (PM + gene dose 0.5) versus EM+UM: - AUC decreased by 25% (from to ng.hour/ml) (NS) - increase in the dose-corrected and weightcorrected AUC, predicted using a pharmacokinetic model, by 5% (from to ng.hour/ml) (S for the trend) Gene dose 1: 106% ref. Lind AB et al. Steady-state concentrations of mirtazapine, N- desmethylmirtazapine, 8-hydroxymirtazapine and their enantiomers in relation to cytochrome P50 2D6 genotype, age and smoking behaviour. Clin Pharmacokinet 2009;8: PM: A UM: AA NOTE: Genotyping was performed for * through *7, *9 and gene duplication. A total of 95 patients were treated with mirtazapine 0 mg/day for weeks, substrates and strong inhibitors of CYP2D6 as co-medication were excluded. Correction was performed for gender, age and smoking. Genotyping: - 56x EM (*1/*1) - 0x IM (x *1/*, 19x *1/*, 5x *1/*5, 2x *1/*6) - 6x PM (x */*, x */*5) - x UM (*1/*1xN with N 2) IM versus - increase in the median C ss of mirtazapine by.% (from 122 to 126 nmol/l) (NS) - increase in the median C ss of S(+)-mirtazapine by 9% (from 28 to 9 nmol/l) (S) - decrease in the median C ss of R(-)-mirtazapine by 1.1% (from 91 to 90 nmol/l) (NS) PM versus - increase in the median C ss of mirtazapine by 57% (from 122 to 192 nmol/l) (NS) - increase in the median C ss of S(+)-mirtazapine by 111% (from 28 to 59 nmol/l) (S) - increase in the median C ss of R(-)-mirtazapine by 9.9% (from 91 to 100 nmol/l) (NS) UM versus - increase in the median C ss of mirtazapine by.1% (from 122 to 127 nmol/l) (NS) - decrease in the median C ss of S(+)-mirtazapine by 25% (from 28 to 21 nmol/l) (NS) - increase in the median C ss of R(-)-mirtazapine by 16% (from 91 to 106 nmol/l) (NS) This study is the first to show the impact of the CYP2D6 genotype on steady-state serum concentrations of the enantiomers of mirtazapine and its metabolites. mirtazapine versus PM: 157% IM: 211% UM: 10% ref. Brockmöller J et al. Pharmacokinetics of mirtazapine: enantioselective effects of the CYP2D6 ultra rapid metabolizer genotype and correlation with adverse effects. Clin Pharmacol Ther 2007;81: PM: A UM: A NOTE: Genotyping was performed for * through *6 and gene duplication. The samples obtained in Kirchheiner, 200 were analysed for enantioselective effects. S(+)-mirtazapine: - decrease in AUC from PM to EM to UM ( 17.7, 9.9 and 6.7 mg.min/l respectively) (S) - increase in Cl or from PM to EM to UM ( 1., 2. and. L/min respectively) (S) - the clearance is higher than can be explained by the hepatic blood flow (1-1.6 L/min): therefore, a first pass effect occurs - plasma concentration correlates with sedation (S) R(-)-mirtazapine: - no decrease in AUC from PM to EM to UM (.1, 29. and 0. mg.min/l respectively) (S) - no increase in Cl or from PM to EM to UM ( 0.66, 0.77 and 0.7 L/min respectively) (S) In ultrarapid metabolizers, one might consider administering higher doses to achieve equivalent sedative and antidepressive effects, but one encounters the dilemma that the cardiovascularly active R(-) enantiomer will not be equally ultrarapidly eliminated. PM: 12% 2
3 ref., continuation ref. 5 Johnson M et al. A poor metabolizer for cytochromes P50 2D6 and 2C19: a case report on antidepressant treatment. CNS Spectr 2006;11: ref. 6 Stephan PL et al. Adverse drug reactions following nonresponse in a depressed patient with CYP2D6 deficiency and low CYP A/5 activity. Pharmacopsychiatry 2006;9: ref. 7 Kirchheiner J et al. Impact of the CYP2D6 ultrarapid metabolizer genotype on mirtazapine pharmacokinetics and adverse events in healthy volunteers. J Clin Psychopharmacol 200;2: UM: A PM: B - plasma concentration correlates with sedation and to a reduction in heart rate and blood pressure hours after administration (S) Both enantiomers: - no difference in plasma concentrations of the desmethyl metabolite between PM, EM and UM - plasma concentrations of the desmethyl metabolites were lower than those of the mother substance A female patient stopped taking venlafaxine, amitriptyline and escitalopram due to side effects. The patient was found to be CYP2D6 */* and CYP2C19 *2/*2. The patient s condition improved in the hospital, after the dose was set to mirtazapine 5 mg/day and hydroxyzine 50 mg/day. There was no followup after discharge and plasma concentrations were not determined. A 7-year-old male exhibited multiple side effects during treatment with clomipramine and quetiapine. The patient was found to be a PM for CYP2D6 (*/*6), EM for CYP2C19 and had a low CYPA/5 activity. The patient was previously treated with mirtazapine (for weeks, maximum dose 60 mg/day). The patient exhibited no clinical response to mirtazapine, side effects that formed a reason to perform genotyping were only reported for clomipramine and quetiapine. A total of 25 healthy, male volunteers (12x EM (gene dose 2), 10x UM (gene dose ), x PM (gene dose 0)) received a single dose of mirtazapine 5 mg. UM versus - AUC mirtazapine decreased from 1.1 to 0.9 mg.hour/l (S by 20%). - Cl or mirtazapine increased from 9.7 to 9.8 L/hour (S by 25%) PM versus - AUC mirtazapine increased from 1.1 to 2.2 mg.hour/l (S by 98%) - Cl or mirtazapine decreased from 9.7 to 20.1 L/hour (S by 9%) - 10 hours after administration, the PMs were still experiencing dry mouth, i.e. a longer period of side effects There was a significant correlation of the mirtazapine plasma concentration with reduced blood pressure, but not with heart rate or QT interval. All volunteers experienced significant sedation. The AUC of desmethylmirtazapine did not differ significantly between PM, EM and UM. Population pharmacokinetics analysis predicts that in carriers of 0, 1, 2 and active CYP2D6 alleles, 0%, 25%, 9% and 55% of Cl or mirtazapine are caused by CYP2D6. UM: 9% PM: 198% UM: 80% ref. 8 Grasmäder K et al. NOTE: Genotyping was performed for the alleles *, *, *5 and *6 and for gene duplication. Plasma concentrations were determined on a weekly basis in 9 patients (29x EM, 1x PM, 18x
4 Population pharmacokinetic analysis of mirtazapine. Eur J Clin Pharmacol 200;60:7-80. ref. 8, continuation IM and 1x UM) on mirtazapine. A total of 22 patients received CYP2D6 substrates as comedication, 10 patients received CYP2D6 inhibitors. A population pharmacokinetics model was constructed with the data. The PM and UM had no abnormal plasma concentrations ( and 6 ng/ml respectively) and were added to the IMs and EMs respectively for the model construction. The resulting model predicts a 26.% lower clearance for IMs than for EMs. The article did not contain any raw data. The variability of mirtazapine plasma concentrations in clinical routine is caused to a relevant degree by CYP2D6. This should be taken into account when a special clinical situation, such as co-morbidity and add-on medication, demands careful dosing of this drug. ref. 9 Grasmäder K et al. Impact of polymorphisms of cytochrome-p50 isoenzymes 2C9, 2C19 and 2D6 on plasma concentrations and clinical effects of antidepressants in a naturalistic clinical setting. Eur J Clin Pharmacol 200;60:29-6. ref. 10 Murphy GM Jr et al. Pharmacogenetics of antidepressant medication intolerance. Am J Psychiatry 200;160: UM: AA (PM+IM): AA NOTE: Genotyping was performed for the alleles *2,*, *, *5, *6, *7, *8 and *9 and for gene duplication. In this study, the *9 allele was categorised with the null alleles. A total of 16 Caucasian patients on antidepressants, including on mirtazapine (dose unknown) were genotyped. Of the patients on mirtazapine, one was a PM and one was a UM, the other 1 were either IM or EM. Relevant comedication was not excluded. The mean dose-corrected C ss of mirtazapine was 0.82 ng/ml per mg of dosed doxepin. For the PM, the dose-corrected plasma concentration was 28% higher than the mean. The PM had no relevant side effects. For the UM, the dose-corrected plasma concentration was % higher than the mean. The UM did have relevant side effects. Out of a total of 26 elderly patients, 121 patients (9x EM # +UM, 27x PM+IM) in a mirtazapine versus paroxetine trial received mirtazapine (15 mg/day for 2 weeks, followed by 0 mg/day for 2 weeks, followed by 0 or 5 mg/day for weeks.) PM+IM compared to EM # +UM: - no significant difference in final dose (1.5 versus 1.22 mg/day) - no significant difference in plasma concentration on day 28 (9.8 versus 0.55 ng/ml) - slightly lower percentage of women (. versus 52.1%) - no difference in three depression scores - lower score for the severity of side effects (NS; 8.6 versus 9.7) - lower percentage of patients who stopped treatment due to side effects (NS; 7. versus 18.1%) Analysis of variance demonstrated no significant interaction between co-medication and effects of the CYP2D6 genotype on the severity of side effects. EM # : In this study, gene dose 1 0 was considered as an EM instead of as an IM. The same results were obtained if gene dose 1 0 was considered as an IM instead of an EM. mirtazapine versus EM (+ IM): PM: 128% UM: 10% CYP2D6 genotype did not predict treatment outcome. Risk group -
5 Comments date of literature search: 11 May 2012 Dutch Pharmacogenetic Working Group decision Phenotype Code Gene-drug interaction Action Date PM B Yes No 27 November 2012 IM A Yes No UM A Yes No Justification of choices: Due to the relatively wide therapeutic range of mirtazapine, the kinetic consequences do not appear to result in clinical effects. One study with a single dose administration found a longer duration of the adverse event dry mouth for PMs. Another study involving a very low dose of S-mirtazapine found an effect on the ability to drive for PMs, but not for IM+EM+UM. However, in therapeutic doses, mirtazapine does have a severe negative effect on the ability to drive, also for non-pms. No significant clinical effects were observed in the other studies. Mechanism: Mirtazapine is converted by CYPA to the metabolite N-desmethylmirtazapine, which has a pharmacological activity that is -6% that of mirtazapine. Mirtazapine is mainly converted by CYP2D6 and CYP1A2 to inactive hydroxy metabolites. This conversion primarily involves the S(+)-enantiomer. Both enantiomers play a role in the side effect sedation, only the R(-)-enantiomer plays a role in the effects on blood pressure and heart rate. The S(+)-enantiomer is probably more therapeutically effective than the R(-)- enantiomer. 5
Pharmacogenetics of antidepressants and antipsychotics: the contribution of allelic variations to the phenotype of drug response
 (2004) 9, 442 473 & 2004 Nature Publishing Group All rights reserved 1359-4184/04 $25.00 www.nature.com/mp FEATURE REVIEW Pharmacogenetics of antidepressants and antipsychotics: the contribution of allelic
(2004) 9, 442 473 & 2004 Nature Publishing Group All rights reserved 1359-4184/04 $25.00 www.nature.com/mp FEATURE REVIEW Pharmacogenetics of antidepressants and antipsychotics: the contribution of allelic
Guidelines/Supporting Studies* FDA Label Information Additional Information/Commentsxc` Gene(s)/Level of evidence
 Drug Gene(s)/Level of evidence Guidelines/Supporting Studies* FDA Label Information Additional Information/Commentsxc` Haloperidol CYP2D6 ( SLC6A5 ( 2D6: DPWG guidelines Reduce dose by 50% in PMs Aripiprazole
Drug Gene(s)/Level of evidence Guidelines/Supporting Studies* FDA Label Information Additional Information/Commentsxc` Haloperidol CYP2D6 ( SLC6A5 ( 2D6: DPWG guidelines Reduce dose by 50% in PMs Aripiprazole
Pharmacogenomics of Antidepressant Medications
 Research Reviews: Pharmacy and Pharmaceutical Sciences e-issn: 2320-1215 www.rroij.com Pharmacogenomics of Antidepressant Medications Amanpreet Kooner and Inder Sehgal* California Health Sciences, University
Research Reviews: Pharmacy and Pharmaceutical Sciences e-issn: 2320-1215 www.rroij.com Pharmacogenomics of Antidepressant Medications Amanpreet Kooner and Inder Sehgal* California Health Sciences, University
Risperidone Case 1: Drug-Drug Interactions
 Risperidone Case 1: Drug-Drug Interactions 1-14-16 de Leon & Bork (a resident) J Clin Psychiatry 1997;58:450-1 http://www.ncbi.nlm.nih.gov/pubmed/9375597 Jose de Leon, MD Educational Objectives At the
Risperidone Case 1: Drug-Drug Interactions 1-14-16 de Leon & Bork (a resident) J Clin Psychiatry 1997;58:450-1 http://www.ncbi.nlm.nih.gov/pubmed/9375597 Jose de Leon, MD Educational Objectives At the
Mental Health DNA Insight WHITE PAPER
 Mental Health DNA Insight WHITE PAPER JULY 2016 Mental Health DNA Insight / White Paper Mental Health DNA Insight Pathway Genomics Mental Health DNA Insight test is aimed to help psychiatrists, neurologists,
Mental Health DNA Insight WHITE PAPER JULY 2016 Mental Health DNA Insight / White Paper Mental Health DNA Insight Pathway Genomics Mental Health DNA Insight test is aimed to help psychiatrists, neurologists,
Pharmacogenetics of Codeine. Lily Mulugeta, Pharm.D Office of Clinical Pharmacology Pediatric Group FDA
 Pharmacogenetics of Codeine Lily Mulugeta, Pharm.D Office of Clinical Pharmacology Pediatric Group FDA 1 Codeine Overview Naturally occurring opium alkaloid Demethylated to morphine for analgesic effect
Pharmacogenetics of Codeine Lily Mulugeta, Pharm.D Office of Clinical Pharmacology Pediatric Group FDA 1 Codeine Overview Naturally occurring opium alkaloid Demethylated to morphine for analgesic effect
Two decades of clinical pharmacogenetic testing - Where do we stand?
 Two decades of clinical pharmacogenetic testing - Where do we stand? Marja-Liisa Dahl, MD PhD, Professor Dept of Clinical Pharmacology Karolinska University Hospital/Karolinska Institutet Stockholm, Sweden
Two decades of clinical pharmacogenetic testing - Where do we stand? Marja-Liisa Dahl, MD PhD, Professor Dept of Clinical Pharmacology Karolinska University Hospital/Karolinska Institutet Stockholm, Sweden
General Discussion 4
 General Discussion 4 General Discussion 115 Introduction Psychiatry is considered to be one of the first medical disciplines that will implement pharmacogenetic testing in daily clinical practice. The
General Discussion 4 General Discussion 115 Introduction Psychiatry is considered to be one of the first medical disciplines that will implement pharmacogenetic testing in daily clinical practice. The
Cytochrome P450 Drug Interaction Table Flockhart Table
 Cytochrome P450 Drug Interaction Table Flockhart Table The table contains lists of drugs in columns under the designation of specific cytochrome P450 isoforms. CYTOCHROME P450 DRUG INTERACTION TABLE A
Cytochrome P450 Drug Interaction Table Flockhart Table The table contains lists of drugs in columns under the designation of specific cytochrome P450 isoforms. CYTOCHROME P450 DRUG INTERACTION TABLE A
A unified in vivomodelingapproach for quantitative prediction of the impact of gene polymorphism and drug interactions on drug exposure
 A unified in vivomodelingapproach for quantitative prediction of the impact of gene polymorphism and drug interactions on drug exposure Sylvain Goutelle, Michel Tod, Laurent Bourguignon, Nathalie Bleyzac,
A unified in vivomodelingapproach for quantitative prediction of the impact of gene polymorphism and drug interactions on drug exposure Sylvain Goutelle, Michel Tod, Laurent Bourguignon, Nathalie Bleyzac,
Implementing Pharmacogenetic Testing Into Clinical Laboratories. Elaine Lyon, PhD, & Gwen McMillin, PhD University of Utah ARUP Laboratories
 Implementing Pharmacogenetic Testing Into Clinical Laboratories Elaine Lyon, PhD, & Gwen McMillin, PhD University of Utah ARUP Laboratories Key learning objectives Recognize decisions in implementing pharmacogenetic
Implementing Pharmacogenetic Testing Into Clinical Laboratories Elaine Lyon, PhD, & Gwen McMillin, PhD University of Utah ARUP Laboratories Key learning objectives Recognize decisions in implementing pharmacogenetic
Pharmacogenetic Testing in Psychiatry Jose de Leon, MD ( )
 Pharmacogenetic Testing in Psychiatry Jose de Leon, MD (12-01-15) Conflicts of Interest (See more details on conflict of interest in the first presentation Training Psychiatrists to Think like Pharmacologists
Pharmacogenetic Testing in Psychiatry Jose de Leon, MD (12-01-15) Conflicts of Interest (See more details on conflict of interest in the first presentation Training Psychiatrists to Think like Pharmacologists
Therapeutic drug monitoring in neuropsychopharmacology: does it hold its promises?
 Therapeutic drug monitoring in neuropsychopharmacology: does it hold its promises? Prof. Dr. Christoph Hiemke Psychiatrische Klinik und Poliklinik Universität Mainz Psychopharmacotherapy Psychiatric Patient
Therapeutic drug monitoring in neuropsychopharmacology: does it hold its promises? Prof. Dr. Christoph Hiemke Psychiatrische Klinik und Poliklinik Universität Mainz Psychopharmacotherapy Psychiatric Patient
Pharmacogenetics: DNA analysis. to explain / predict. response to drug therapy. Maurizio Ferrari & Ron van Schaik
 Maurizio Ferrari & Ron van Schaik Workshop IFCC Kuala Lumpur November 19, 2012 Predictive, Preventive and Personalized Medicine Part II: Pharmacogenetics l r.vanschaik@erasmusmc.nl Pharmacogenetics: DNA
Maurizio Ferrari & Ron van Schaik Workshop IFCC Kuala Lumpur November 19, 2012 Predictive, Preventive and Personalized Medicine Part II: Pharmacogenetics l r.vanschaik@erasmusmc.nl Pharmacogenetics: DNA
Pharmacokinetics, pharmacodynamics and pharmacogenetics of aripiprazole and olanzapine in healthy subjects
 Pharmacokinetics, pharmacodynamics and pharmacogenetics of aripiprazole and olanzapine in healthy subjects Dr. Francisco Abad Santos 16 June 2017 Atypical antipsychotics or second generation antipsychotics
Pharmacokinetics, pharmacodynamics and pharmacogenetics of aripiprazole and olanzapine in healthy subjects Dr. Francisco Abad Santos 16 June 2017 Atypical antipsychotics or second generation antipsychotics
Impact of CYP2C19 genetics on pharmacokinetic variability of escitalopram and sertraline - a study based on therapeutic drug monitoring data
 Impact of CYP2C19 genetics on pharmacokinetic variability of escitalopram and sertraline - a study based on therapeutic drug monitoring data Ida Rudberg 2009 Department of Psychopharmacology Diakonhjemmet
Impact of CYP2C19 genetics on pharmacokinetic variability of escitalopram and sertraline - a study based on therapeutic drug monitoring data Ida Rudberg 2009 Department of Psychopharmacology Diakonhjemmet
Pharmacogenetics in: Primary Care. Bradley T. Wajda D.O.
 Pharmacogenetics in: Primary Care Bradley T. Wajda D.O. Pharmacogenomics Defined Pharmacogenomics uses information about a person s genetic makeup, or genome, to choose the drugs and drug doses that are
Pharmacogenetics in: Primary Care Bradley T. Wajda D.O. Pharmacogenomics Defined Pharmacogenomics uses information about a person s genetic makeup, or genome, to choose the drugs and drug doses that are
6. DOSE ADJUSTMENTS BASED ON PHARMACOGENETICS OF CYP450 ENZYMES
 6. DOSE ADJUSTMENTS BASED ON PHARMACOGENETICS OF CYP450 ENZYMES Ron H.N. van Schaik Dept. Clinical Chemistry, Erasmus MC, Rotterdam, The Netherlands 6.1 Introduction In today s medicine, drug therapy represents
6. DOSE ADJUSTMENTS BASED ON PHARMACOGENETICS OF CYP450 ENZYMES Ron H.N. van Schaik Dept. Clinical Chemistry, Erasmus MC, Rotterdam, The Netherlands 6.1 Introduction In today s medicine, drug therapy represents
WHAT S NEW. Vilazodone (Viibryd ) Vilazodone - Dosing ANTIDEPRESSANT UPDATE: What s New? The Cardiac Debate The Efficacy Debate?Pharmacogenomics?
 ANTIDEPRESSANT UPDATE: What s New? The Cardiac Debate The Efficacy Debate?Pharmacogenomics? Rex S. Lott, Pharm.D., BCPP Professor, ISU College of Pharmacy Mental Health Clinical Pharmacist, Boise VAMC
ANTIDEPRESSANT UPDATE: What s New? The Cardiac Debate The Efficacy Debate?Pharmacogenomics? Rex S. Lott, Pharm.D., BCPP Professor, ISU College of Pharmacy Mental Health Clinical Pharmacist, Boise VAMC
Core Data Set CYP2D6 Metabolism
 Core Data Set CYP2D6 Metabolism Oxidised metabolites seen in pre-clinical species Inhibitor Target CYP Isoform CLint (µl/min/mg protein) % Inhibition Control 12.5 - Furafylline 1A2 12.9 0 Sulfaphenoxazole
Core Data Set CYP2D6 Metabolism Oxidised metabolites seen in pre-clinical species Inhibitor Target CYP Isoform CLint (µl/min/mg protein) % Inhibition Control 12.5 - Furafylline 1A2 12.9 0 Sulfaphenoxazole
Pharmacokinetics for Physicians. Assoc Prof. Noel E. Cranswick Clinical Pharmacologist Royal Children s Hospital Melbourne
 Pharmacokinetics for Physicians Assoc Prof. Noel E. Cranswick Clinical Pharmacologist Royal Children s Hospital Melbourne The Important Therapeutic Questions What drug? What dose? How long? Drug Dosage
Pharmacokinetics for Physicians Assoc Prof. Noel E. Cranswick Clinical Pharmacologist Royal Children s Hospital Melbourne The Important Therapeutic Questions What drug? What dose? How long? Drug Dosage
Pharmacogenomics of Medications for Pain and Major Depression: Promise and Peril
 Pharmacogenomics of Medications for Pain and Major Depression: Promise and Peril Geoffrey C. Wall, PharmD, FCCP, BCPS, CGP Professor of Clinical Sciences Drake University College of Pharmacy and Health
Pharmacogenomics of Medications for Pain and Major Depression: Promise and Peril Geoffrey C. Wall, PharmD, FCCP, BCPS, CGP Professor of Clinical Sciences Drake University College of Pharmacy and Health
Deriving Rules and Assertions From Pharmacogenomic Knowledge Resources In Support Of Patient Drug Metabolism Efficacy Predictions!
 Deriving Rules and Assertions From Pharmacogenomic Knowledge Resources In Support Of Patient Drug Metabolism Efficacy Predictions! Casey L. Overby 1,2, Beth Devine 1, Peter Tarczy-Hornoch 1, Ira Kalet
Deriving Rules and Assertions From Pharmacogenomic Knowledge Resources In Support Of Patient Drug Metabolism Efficacy Predictions! Casey L. Overby 1,2, Beth Devine 1, Peter Tarczy-Hornoch 1, Ira Kalet
Psychiatric Pharmacogenomics: Introduction and Applications
 Psychiatric Pharmacogenomics: Introduction and Applications Moises Gaviria, MD Distinguished Professor of Psychiatry University of Illinois at Chicago Medical Director The Institute of Neurobehavioral
Psychiatric Pharmacogenomics: Introduction and Applications Moises Gaviria, MD Distinguished Professor of Psychiatry University of Illinois at Chicago Medical Director The Institute of Neurobehavioral
Clozapine Case 1 The Relevance of CYP Jose de Leon, MD
 Clozapine Case 1 The Relevance of CYP 12-18-15 Jose de Leon, MD 1. Clozapine Case 1 J Clin Psychiatry 1996;57:175-176 http://www.ncbi.nlm.nih.gov/pubmed/8601555 Educational Objectives At the conclusion
Clozapine Case 1 The Relevance of CYP 12-18-15 Jose de Leon, MD 1. Clozapine Case 1 J Clin Psychiatry 1996;57:175-176 http://www.ncbi.nlm.nih.gov/pubmed/8601555 Educational Objectives At the conclusion
They deserve personalized treatment
 Your patients are unique They deserve personalized treatment New laboratory service offered by STA 2 R is a panel of genetic tests that gives prescribers answers to the clinical questions below. The test
Your patients are unique They deserve personalized treatment New laboratory service offered by STA 2 R is a panel of genetic tests that gives prescribers answers to the clinical questions below. The test
Mujeeb U. Shad, M.D., M.S.C.S. Associate Professor of Psychiatry Oregon Health & Science University, Portland Oregon Supervising Psychiatrist Oregon
 Mujeeb U. Shad, M.D., M.S.C.S. Associate Professor of Psychiatry Oregon Health & Science University, Portland Oregon Supervising Psychiatrist Oregon State Hospital, Salem Oregon Assurex Health Inc. has
Mujeeb U. Shad, M.D., M.S.C.S. Associate Professor of Psychiatry Oregon Health & Science University, Portland Oregon Supervising Psychiatrist Oregon State Hospital, Salem Oregon Assurex Health Inc. has
3. Describe how variants in the CYP2C19 gene impact Plavix metabolism. 4. Compare molecular genetic technologies for pharmacogenomics testing
 UP! UNDERSTANDING PHARMACOGENOMICS (PGX) Robert Pyatt Ph.D., Director Sanford Medical Genetics and Genomics Laboratories and Associate Professor, Dept of Internal Medicine, University of South Dakota April
UP! UNDERSTANDING PHARMACOGENOMICS (PGX) Robert Pyatt Ph.D., Director Sanford Medical Genetics and Genomics Laboratories and Associate Professor, Dept of Internal Medicine, University of South Dakota April
Ron van Schaik. Pharmacogenetics. London, Oct 8-9, Clinical implementation: a 7 year experience
 Ron van Schaik Associate Professor Pharmacogenetics Eur Clin Chem / Advisor EMA - PGWG London, Oct 8-9, 2012 Pharmacogenetics Clinical implementation: a 7 year experience Pharmacogenetics Core Laboratory*
Ron van Schaik Associate Professor Pharmacogenetics Eur Clin Chem / Advisor EMA - PGWG London, Oct 8-9, 2012 Pharmacogenetics Clinical implementation: a 7 year experience Pharmacogenetics Core Laboratory*
Pharmacogenetics and Pharmacokinetics
 Chapter 2 Pharmacogenetics and Pharmacokinetics Mauro Saivezzo/ShutterStock, Inc. L earning O bjectives Upon completion of this chapter, the student will be able to: 1. Recognize the influence of genetic
Chapter 2 Pharmacogenetics and Pharmacokinetics Mauro Saivezzo/ShutterStock, Inc. L earning O bjectives Upon completion of this chapter, the student will be able to: 1. Recognize the influence of genetic
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
Imipramine therapy (CYP2D6)
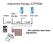 Imipramine therapy (CYP2D6) Intake Start therapy 1st conc 08:00 2nd conc 3e spiegel 4e spiegel 1 week 1 week 1 week 1 week 1 week 22:00 ~ 50% patients need dose adjustment Imipramine therapy (CYP2D6) Intake
Imipramine therapy (CYP2D6) Intake Start therapy 1st conc 08:00 2nd conc 3e spiegel 4e spiegel 1 week 1 week 1 week 1 week 1 week 22:00 ~ 50% patients need dose adjustment Imipramine therapy (CYP2D6) Intake
Quetiapine Case 1 Warfarin Jose de Leon, MD
 Quetiapine Case 1 Warfarin 1-23-16 Jose de Leon, MD 1. Quetiapine Case 1 J Clin Psychopharm 1999;19:382-3 http://www.ncbi.nlm.nih.gov/pubmed/10440472 Educational Objectives At the conclusion of this presentation,
Quetiapine Case 1 Warfarin 1-23-16 Jose de Leon, MD 1. Quetiapine Case 1 J Clin Psychopharm 1999;19:382-3 http://www.ncbi.nlm.nih.gov/pubmed/10440472 Educational Objectives At the conclusion of this presentation,
Application of Pharmacogenetics Supplementary Worksheet
 Application of Pharmacogenetics Supplementary Worksheet Section 1 Health Care Problem Section 2 Drug Metabolism Section 3 Phase I & II metabolism Section 4 Inhibitors and Inducers Section 5 DNA & Drug
Application of Pharmacogenetics Supplementary Worksheet Section 1 Health Care Problem Section 2 Drug Metabolism Section 3 Phase I & II metabolism Section 4 Inhibitors and Inducers Section 5 DNA & Drug
Quetiapine Case 2 Therapeutic Drug Monitoring Jose de Leon, MD
 Quetiapine Case 2 Therapeutic Drug Monitoring 1-27-16 Jose de Leon, MD 2. Quetiapine Case Therapeutic Drug Monitoring (unpublished) Educational Objectives At the conclusion of this presentation, the participant
Quetiapine Case 2 Therapeutic Drug Monitoring 1-27-16 Jose de Leon, MD 2. Quetiapine Case Therapeutic Drug Monitoring (unpublished) Educational Objectives At the conclusion of this presentation, the participant
Article. Reference. Applications of CYP450 Testing in the Clinical Setting. SAMER, Caroline Flora, et al.
 Article Applications of CYP450 Testing in the Clinical Setting SAMER, Caroline Flora, et al. Abstract Interindividual variability in drug response is a major clinical problem. Polymedication and genetic
Article Applications of CYP450 Testing in the Clinical Setting SAMER, Caroline Flora, et al. Abstract Interindividual variability in drug response is a major clinical problem. Polymedication and genetic
CYP2C19-Proton Pump Inhibitors
 CYP2C19-Proton Pump Inhibitors Cameron Thomas, Pharm.D. PGY2 Clinical Pharmacogenetics Resident St. Jude Children s Research Hospital February 1, 2018 Objectives: CYP2C19-PPI Implementation Review the
CYP2C19-Proton Pump Inhibitors Cameron Thomas, Pharm.D. PGY2 Clinical Pharmacogenetics Resident St. Jude Children s Research Hospital February 1, 2018 Objectives: CYP2C19-PPI Implementation Review the
Pharmacogenetics and clinical opioid efficacy. Pål l Klepstad Professor of Intensive Care Medicine St.Olavs University Hospital Trondheim,, Norway
 Pharmacogenetics and clinical opioid efficacy Pål l Klepstad Professor of Intensive Care Medicine St.Olavs University Hospital Trondheim,, Norway Variation in opioid dose Variation in effects from different
Pharmacogenetics and clinical opioid efficacy Pål l Klepstad Professor of Intensive Care Medicine St.Olavs University Hospital Trondheim,, Norway Variation in opioid dose Variation in effects from different
Variation in drug responses & Drug-Drug Interactions
 Variation in drug responses & Drug-Drug Interactions 1 Properties of an Ideal Drug Effective Safety Selective Reversible Action Predictable Freedom from drug interactions Low cost Chemically stable Sources
Variation in drug responses & Drug-Drug Interactions 1 Properties of an Ideal Drug Effective Safety Selective Reversible Action Predictable Freedom from drug interactions Low cost Chemically stable Sources
Risperidone (RIS) is metabolized primarily by 9-hydroxylation
 BRIEF REPORT Effects of CYP2D6 and CYP3A5 Genotypes on the Plasma Concentrations of Risperidone and 9-Hydroxyrisperidone in Korean Schizophrenic Patients Rhee-Hun Kang, MD, PhD,* Sun-Min Jung, MD, PhD,þ
BRIEF REPORT Effects of CYP2D6 and CYP3A5 Genotypes on the Plasma Concentrations of Risperidone and 9-Hydroxyrisperidone in Korean Schizophrenic Patients Rhee-Hun Kang, MD, PhD,* Sun-Min Jung, MD, PhD,þ
Self Assessment Question 1
 Drug Interactions Bruce G. Pollock, M.D., Ph.D. Professor of Psychiatry, Pharmacology and Nursing Chief, Academic Division of Geriatrics and Neuropsychiatry University of Pittsburgh Medical Center 1 Self
Drug Interactions Bruce G. Pollock, M.D., Ph.D. Professor of Psychiatry, Pharmacology and Nursing Chief, Academic Division of Geriatrics and Neuropsychiatry University of Pittsburgh Medical Center 1 Self
Membership Overview: Total Members: 322 Student Members: 160 Resident Members: 8 Fellow Members: 4
 A Closer Look at the Central Nervous System PRN Overview of the PRN The Central Nervous System Practice and Research Network (CNS PRN) provides a forum to encourage networking among pharmacists specializing
A Closer Look at the Central Nervous System PRN Overview of the PRN The Central Nervous System Practice and Research Network (CNS PRN) provides a forum to encourage networking among pharmacists specializing
Effect of Co-medication on the Serum Concentrations of Aripiprazole and the Main Metabolite Dehydroaripiprazole
 Effect of Co-medication on the Serum Concentrations of Aripiprazole and the Main Metabolite Dehydroaripiprazole Thesis submitted for the degree Candidate Pharmaciae at Department of Pharmaceutical Biosciences,
Effect of Co-medication on the Serum Concentrations of Aripiprazole and the Main Metabolite Dehydroaripiprazole Thesis submitted for the degree Candidate Pharmaciae at Department of Pharmaceutical Biosciences,
Supplement to: Clinical Pharmacogenetics Implementation Consortium Guideline (CPIC ) for CYP2D6
 Supplement to: Pharmacogenetics Implementation Consortium Guideline (CPIC ) for CYP2D6 and CYP2C19 Genotypes and Tricyclic Antidepressants: 2016 Update J. Kevin Hicks 1, Katrin Sangkuhl 2, Jesse J. Swen
Supplement to: Pharmacogenetics Implementation Consortium Guideline (CPIC ) for CYP2D6 and CYP2C19 Genotypes and Tricyclic Antidepressants: 2016 Update J. Kevin Hicks 1, Katrin Sangkuhl 2, Jesse J. Swen
Falk Symposium 156: Genetics in Liver Disease. Pharmacogenetics. Gerd Kullak-Ublick
 Falk Symposium 156: Genetics in Liver Disease Pharmacogenetics Gerd Kullak-Ublick Division of Clinical Pharmacology and Toxicology Department of Internal Medicine University Hospital Zurich Freiburg, 8.
Falk Symposium 156: Genetics in Liver Disease Pharmacogenetics Gerd Kullak-Ublick Division of Clinical Pharmacology and Toxicology Department of Internal Medicine University Hospital Zurich Freiburg, 8.
Objectives. Clinical Problem. What if there were a way. Pharmacogenomics in Current Practice MEDICINE 12/1/2017
 Objectives Pharmacogenomics in Current Practice Trinh Pham, PharmD, BCOP Associate Clinical Professor University of Connecticut School of Pharmacy Review the concept of pharmacogenetics and pharmacogenomics
Objectives Pharmacogenomics in Current Practice Trinh Pham, PharmD, BCOP Associate Clinical Professor University of Connecticut School of Pharmacy Review the concept of pharmacogenetics and pharmacogenomics
Pharmacogenomics in Current Practice. Trinh Pham, PharmD, BCOP Associate Clinical Professor University of Connecticut School of Pharmacy.
 Pharmacogenomics in Current Practice Trinh Pham, PharmD, BCOP Associate Clinical Professor University of Connecticut School of Pharmacy Objectives Review the concept of pharmacogenetics and pharmacogenomics
Pharmacogenomics in Current Practice Trinh Pham, PharmD, BCOP Associate Clinical Professor University of Connecticut School of Pharmacy Objectives Review the concept of pharmacogenetics and pharmacogenomics
NIH Public Access Author Manuscript Clin Pharmacol Ther. Author manuscript; available in PMC 2014 March 01.
 NIH Public Access Author Manuscript Published in final edited form as: Clin Pharmacol Ther. 2013 September ; 94(3):. doi:10.1038/clpt.2013.96. META-ANALYSIS OF CYP2D6 METABOLIZER PHENOTYPE AND METOPROLOL
NIH Public Access Author Manuscript Published in final edited form as: Clin Pharmacol Ther. 2013 September ; 94(3):. doi:10.1038/clpt.2013.96. META-ANALYSIS OF CYP2D6 METABOLIZER PHENOTYPE AND METOPROLOL
CORE DME PANEL Highlands Parkway, Suite 100 Smyrna, GA 30082
 CORE DME PANEL Castle's CORE DME panel predicts the activity levels of key - drug metabolizing enzymes in the cytochrome P450 superfamily: CYP2D6, CYP2C9, CYP2C19, CYP2B6, CYP3A4, and - CYP3A5. Apart from
CORE DME PANEL Castle's CORE DME panel predicts the activity levels of key - drug metabolizing enzymes in the cytochrome P450 superfamily: CYP2D6, CYP2C9, CYP2C19, CYP2B6, CYP3A4, and - CYP3A5. Apart from
ETHNICITY AND PSYCHOTROPIC RESPONSE
 Ethnic Differences in Drug Metabolism ETHNICITY AND PSYCHOTROPIC RESPONSE Bridging Cultures: Improving Evaluation & Treatment of Cognitive 8 March 28 Keh-Ming Lin, M.D., M.P.H. Professor Emeritus of Psychiatry,
Ethnic Differences in Drug Metabolism ETHNICITY AND PSYCHOTROPIC RESPONSE Bridging Cultures: Improving Evaluation & Treatment of Cognitive 8 March 28 Keh-Ming Lin, M.D., M.P.H. Professor Emeritus of Psychiatry,
Pharmacogenomics Gene/Drug-Pair Decision Flow Charts 2017
 Pharmacogenomics Gene/Drug-Pair Decision Flow Charts 2017 Atomoxetine Prescription CYP2D6 Genotype (AS = 0) (AS = 0.5 1.0) (As = 2.0) Ultrarapid (AS >2.0) Standard dose. Dose increase probably not necessary;
Pharmacogenomics Gene/Drug-Pair Decision Flow Charts 2017 Atomoxetine Prescription CYP2D6 Genotype (AS = 0) (AS = 0.5 1.0) (As = 2.0) Ultrarapid (AS >2.0) Standard dose. Dose increase probably not necessary;
Caveat: Validation and Limitations of Phenotyping Methods for Drug Metabolizing Enzymes and Transporters
 Caveat: Validation and Limitations of Phenotyping Methods for Drug Metabolizing Enzymes and Transporters Uwe Fuhr, University Hospital Cologne 1 How to Safeguard that Metrics Reflect E/T Activity? in healthy
Caveat: Validation and Limitations of Phenotyping Methods for Drug Metabolizing Enzymes and Transporters Uwe Fuhr, University Hospital Cologne 1 How to Safeguard that Metrics Reflect E/T Activity? in healthy
CYP2D6 Genotype to Phenotype Standardization Project
 CYP2D6 Genotype to Phenotype Standardization Project Project Objectives Determine a strategy for defining CYP2D6 phenotype based on genotype using a modified Delphi method. Standardize phenotype definitions
CYP2D6 Genotype to Phenotype Standardization Project Project Objectives Determine a strategy for defining CYP2D6 phenotype based on genotype using a modified Delphi method. Standardize phenotype definitions
I have NO actual or potential conflicts of interest in relation to this educational presentation.
 Genotyping The Patient Requiring Mental Health Medications: When Does It Make Sense? Charles F. Caley, PharmD, BCPP Clinical Professor UConn School of Pharmacy Storrs, CT I have NO actual or potential
Genotyping The Patient Requiring Mental Health Medications: When Does It Make Sense? Charles F. Caley, PharmD, BCPP Clinical Professor UConn School of Pharmacy Storrs, CT I have NO actual or potential
Personalized Medicine in Real Time
 LABORATORY OF PERSONALIZED HEALTH LPH Personalized Medicine in Real Time DNA-Guided Clopidogrel (Plavix ) Management Pharmacogenetic Foundations and Case Study 1 Clopidogrel (Plavix ) Leading AntiPlatelet
LABORATORY OF PERSONALIZED HEALTH LPH Personalized Medicine in Real Time DNA-Guided Clopidogrel (Plavix ) Management Pharmacogenetic Foundations and Case Study 1 Clopidogrel (Plavix ) Leading AntiPlatelet
Pharmacogenomics and Customized Therapies in Psychiatry
 Pharmacogenomics and Customized Therapies in Psychiatry Toshiyuki Someya,, MD, PhD Department of Psychiatry Niigata University Graduate School of Medical and Dental Sciences The efficacy and side effects
Pharmacogenomics and Customized Therapies in Psychiatry Toshiyuki Someya,, MD, PhD Department of Psychiatry Niigata University Graduate School of Medical and Dental Sciences The efficacy and side effects
JOINT MEETING OF CORONARY REVASCULARIZATION 2014 TIONG LEE LEN SENIOR RESEARCH PHARMACIST CLINICAL RESEARCH CENTER, SARAWAK GENERAL HOSPITAL
 The Effect of Non Steady State and Steady State Clopidogrel Carboxylic Acid Plasma Concentration on Clopidogrel Responsiveness in Patients Planned For Percutaneous Coronary Intervention JOINT MEETING OF
The Effect of Non Steady State and Steady State Clopidogrel Carboxylic Acid Plasma Concentration on Clopidogrel Responsiveness in Patients Planned For Percutaneous Coronary Intervention JOINT MEETING OF
Valproate Case 3: Formulations Jose de Leon, MD
 Valproate Case 3: Formulations 2-12-16 Jose de Leon, MD 3.Valproate Case 3 Described in J Clin Psychiatry 2004;65:724-5 http://www.ncbi.nlm.nih.gov/pubmed/15163266 Pharmacological explanation provided
Valproate Case 3: Formulations 2-12-16 Jose de Leon, MD 3.Valproate Case 3 Described in J Clin Psychiatry 2004;65:724-5 http://www.ncbi.nlm.nih.gov/pubmed/15163266 Pharmacological explanation provided
Review of Pharmacogenetic Testing Today
 Review of Pharmacogenetic Testing Today Gwen McMillin, PhD ARUP Laboratories University of Utah Salt Lake City, Utah A customer says to the pharmacist: "Why does my medication have 40 side effects?" The
Review of Pharmacogenetic Testing Today Gwen McMillin, PhD ARUP Laboratories University of Utah Salt Lake City, Utah A customer says to the pharmacist: "Why does my medication have 40 side effects?" The
SAMPLE REPORT MENTAL HEALTH DNA INSIGHT LABORATORY INFO. Protected Health Information. SSRIs. TCAs. Other Antidepressants
 Test Results Reviewed & Approved by: Laboratory Director, Nilesh Dharajiya,.D. ENTAL HEALTH DNA INSIGHT PERSONAL DETAILS DOB Jan 1, 19XX ETHNICITY Caucasian ORDERING HEALTHCARE PROFESSIONAL Glenn Braunstein.D.
Test Results Reviewed & Approved by: Laboratory Director, Nilesh Dharajiya,.D. ENTAL HEALTH DNA INSIGHT PERSONAL DETAILS DOB Jan 1, 19XX ETHNICITY Caucasian ORDERING HEALTHCARE PROFESSIONAL Glenn Braunstein.D.
CYP2D6: Genotypes, Phenotypes, and Genetic Testing
 CYP2D6: Genotypes, Phenotypes, and Genetic Testing In 1975, several laboratory scientists at St. Mary s Hospital Medical School in London each ingested a 40 mg dose of debrisoquine, an antihypertensive
CYP2D6: Genotypes, Phenotypes, and Genetic Testing In 1975, several laboratory scientists at St. Mary s Hospital Medical School in London each ingested a 40 mg dose of debrisoquine, an antihypertensive
PEER REVIEW HISTORY ARTICLE DETAILS VERSION 1 - REVIEW
 PEER REVIEW HISTORY BMJ Open publishes all reviews undertaken for accepted manuscripts. Reviewers are asked to complete a checklist review form (see an example) and are provided with free text boxes to
PEER REVIEW HISTORY BMJ Open publishes all reviews undertaken for accepted manuscripts. Reviewers are asked to complete a checklist review form (see an example) and are provided with free text boxes to
Supplement to: Clinical Pharmacogenetics Implementation Consortium Guideline for CYP2D6 and CYP2C19 Genotypes and Dosing of Tricyclic Antidepressants
 Supplement to: Pharmacogenetics Implementation Consortium Guideline for CYP2D6 and CYP2C19 Genotypes and Dosing of Tricyclic Antidepressants J. Kevin Hicks 1, Jesse J. Swen 2, Caroline F. Thorn 3, Katrin
Supplement to: Pharmacogenetics Implementation Consortium Guideline for CYP2D6 and CYP2C19 Genotypes and Dosing of Tricyclic Antidepressants J. Kevin Hicks 1, Jesse J. Swen 2, Caroline F. Thorn 3, Katrin
Pharmacogenetics: From Bench to Byte An Update of Guidelines
 nature publishing group Pharmacogenetics: From Bench to Byte An Update of Guidelines JJ Swen 1, M Nijenhuis 2, A de Boer 3, L Grandia 2, AH Maitland-van der Zee 3, H Mulder 3,4, GAPJM Rongen 5,6,7, RHN
nature publishing group Pharmacogenetics: From Bench to Byte An Update of Guidelines JJ Swen 1, M Nijenhuis 2, A de Boer 3, L Grandia 2, AH Maitland-van der Zee 3, H Mulder 3,4, GAPJM Rongen 5,6,7, RHN
This clinical study synopsis is provided in line with Boehringer Ingelheim s Policy on Transparency and Publication of Clinical Study Data.
 abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the
abcd Clinical Study Synopsis for Public Disclosure This clinical study synopsis is provided in line with s Policy on Transparency and Publication of Clinical Study Data. The synopsis which is part of the
Optimal Use of Antidepressants: Focusing on SNRI, NDRI and SSRE
 Optimal Use of Antidepressants: Focusing on SNRI, NDRI and SSRE Chan-Hyung Kim, MD Severance Mental Health Hospital Institute of Behavioral Science in Medicine Diagnostic Criteria Pyramid Etiologic Pathophysiologic
Optimal Use of Antidepressants: Focusing on SNRI, NDRI and SSRE Chan-Hyung Kim, MD Severance Mental Health Hospital Institute of Behavioral Science in Medicine Diagnostic Criteria Pyramid Etiologic Pathophysiologic
Original Policy Date
 MP 2.04.38 Genetic Testing for Helicobacter pylori Treatment Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date Reviewed with literature search/12:2013 Return
MP 2.04.38 Genetic Testing for Helicobacter pylori Treatment Medical Policy Section Medicine Issue 12:2013 Original Policy Date 12:2013 Last Review Status/Date Reviewed with literature search/12:2013 Return
Summary of the risk management plan (RMP) for Wakix (pitolisant)
 EMA/794885/2015 Summary of the risk management plan (RMP) for Wakix (pitolisant) This is a summary of the risk management plan (RMP) for Wakix, which details the measures to be taken in order to ensure
EMA/794885/2015 Summary of the risk management plan (RMP) for Wakix (pitolisant) This is a summary of the risk management plan (RMP) for Wakix, which details the measures to be taken in order to ensure
Genetics and Genomics: Influence on Individualization of Medication Regimes
 Genetics and Genomics: Influence on Individualization of Medication Regimes Joseph S Bertino Jr., Pharm.D., FCCP Schenectady, NY USA Goals and Objectives To discuss pharmacogenetics and pharmacogenomics
Genetics and Genomics: Influence on Individualization of Medication Regimes Joseph S Bertino Jr., Pharm.D., FCCP Schenectady, NY USA Goals and Objectives To discuss pharmacogenetics and pharmacogenomics
Medical Policy An Independent Licensee of the Blue Cross and Blue Shield Association
 Cytochrome p450 Genotyping Page 1 of 24 Medical Policy An Independent Licensee of the Blue Cross and Blue Shield Association Title: See Also: Cytochrome p450 Genotyping Genetic Testing for Helicobacter
Cytochrome p450 Genotyping Page 1 of 24 Medical Policy An Independent Licensee of the Blue Cross and Blue Shield Association Title: See Also: Cytochrome p450 Genotyping Genetic Testing for Helicobacter
Common Antidepressant Medications for Adults
 (and Citalopram (Celexa) Escitalopram (Lexapro) Fluoxetine (Prozac) Fluoxetine Weekly (Prozac Weekly) 20 in AM w/ food (10 mg in elderly or those w/ panic disorder) 20 40 40 (If age >60yo, max 20) 10 10
(and Citalopram (Celexa) Escitalopram (Lexapro) Fluoxetine (Prozac) Fluoxetine Weekly (Prozac Weekly) 20 in AM w/ food (10 mg in elderly or those w/ panic disorder) 20 40 40 (If age >60yo, max 20) 10 10
Intuitive pharmacogenetics: spontaneous risperidone dosage is related to CYP2D6, CYP3A5 and ABCB1 genotypes
 (2010), 1 5 & 2010 Macmillan Publishers Limited. All rights reserved 1470-269X/10 www.nature.com/tpj ORIGINAL ARTICLE Intuitive pharmacogenetics: spontaneous risperidone dosage is related to CYP2D6, CYP3A5
(2010), 1 5 & 2010 Macmillan Publishers Limited. All rights reserved 1470-269X/10 www.nature.com/tpj ORIGINAL ARTICLE Intuitive pharmacogenetics: spontaneous risperidone dosage is related to CYP2D6, CYP3A5
The analgesic drug, tramadol, has a dual mechanism
 The Analgesic Effect of Tramadol After Intravenous Injection in Healthy Volunteers in Relation to CYP2D6 Thomas P. Enggaard, MD*, Lars Poulsen, MD*, Lars Arendt-Nielsen, PhD, Kim Brøsen, MD*, Joachim Ossig,
The Analgesic Effect of Tramadol After Intravenous Injection in Healthy Volunteers in Relation to CYP2D6 Thomas P. Enggaard, MD*, Lars Poulsen, MD*, Lars Arendt-Nielsen, PhD, Kim Brøsen, MD*, Joachim Ossig,
Pharmacogenomics and Pharmacokinetics ^
 Pharmacogenomics and Pharmacokinetics ^ avid F. Kisor, B.S., Pharm.. Profeor of Pharmacokinetics epartment of Pharmaceutical and Biomedical Sciences Raabe College of Pharmacy Ohio Northern University Learning
Pharmacogenomics and Pharmacokinetics ^ avid F. Kisor, B.S., Pharm.. Profeor of Pharmacokinetics epartment of Pharmaceutical and Biomedical Sciences Raabe College of Pharmacy Ohio Northern University Learning
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only.
 The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
The clinical trial information provided in this public disclosure synopsis is supplied for informational purposes only. Please note that the results reported in any single trial may not reflect the overall
Physiologically Based Pharmacokinetic Model of the CYP2D6 Probe Atomoxetine: Extrapolation to Special Populations and Drug-Drug Interactions s
 Supplemental material to this article can be found at: http://dmd.aspetjournals.org/content/suppl/2017/08/31/dmd.117.076455.dc1 1521-009X/45/11/1156 1165$25.00 https://doi.org/10.1124/dmd.117.076455 DRUG
Supplemental material to this article can be found at: http://dmd.aspetjournals.org/content/suppl/2017/08/31/dmd.117.076455.dc1 1521-009X/45/11/1156 1165$25.00 https://doi.org/10.1124/dmd.117.076455 DRUG
Variability Due to Genetic Differences
 1 Variability Due to Genetic Differences Nick Holford Dept Pharmacology & Clinical Pharmacology University of Auckland 2 Objectives Understand how between individual variation may contribute to :» drug
1 Variability Due to Genetic Differences Nick Holford Dept Pharmacology & Clinical Pharmacology University of Auckland 2 Objectives Understand how between individual variation may contribute to :» drug
Strategies for Developing and Validating PBPK Models for Extrapolation to Unstudied Population
 Strategies for Developing and Validating PBPK Models for Extrapolation to Unstudied Population Nina Isoherranen Department of Pharmaceutics University of Washington Processes driving drug disposition are
Strategies for Developing and Validating PBPK Models for Extrapolation to Unstudied Population Nina Isoherranen Department of Pharmaceutics University of Washington Processes driving drug disposition are
Current Approaches and Applications of Phenotyping Methods for Drug Metabolizing Enzymes and Transporters
 Current Approaches and Applications of Phenotyping Methods for Drug Metabolizing Enzymes and Transporters Uwe Fuhr, University Hospital Cologne 1 Definition Phenotyping is quantifying the in vivo activity
Current Approaches and Applications of Phenotyping Methods for Drug Metabolizing Enzymes and Transporters Uwe Fuhr, University Hospital Cologne 1 Definition Phenotyping is quantifying the in vivo activity
Pharmacogenomics In Psychiatry. Wolfgang Sadee OSU Program in Pharmacogenomics The Ohio State University. XGEN Group NIH PGRN
 Pharmacogenomics In Psychiatry Wolfgang Sadee OSU Program in Pharmacogenomics The Ohio State University XGEN Group NIH PGRN Why do we need biomarkers in drug therapy? Limited efficacy Adverse drug effects
Pharmacogenomics In Psychiatry Wolfgang Sadee OSU Program in Pharmacogenomics The Ohio State University XGEN Group NIH PGRN Why do we need biomarkers in drug therapy? Limited efficacy Adverse drug effects
Pharmacogenomics and Clinical Practice: Ready for Prime Time?
 Pharmacogenomics and Clinical Practice: Ready for Prime Time? Manju T. Beier, Pharm D, CGP, FASCP Chief Scientific Officer Natural Molecular Testing Corp. Adjunct Clinical Associate Professor of Pharmacy
Pharmacogenomics and Clinical Practice: Ready for Prime Time? Manju T. Beier, Pharm D, CGP, FASCP Chief Scientific Officer Natural Molecular Testing Corp. Adjunct Clinical Associate Professor of Pharmacy
FEP Medical Policy Manual
 FEP Medical Policy Manual Effective Date: October 15, 2018 Related Policies: 2.04.38 Cytochrome P450 Genotype-Guided Treatment Strategy Genotype-Guided Tamoxifen Treatment Description Tamoxifen is prescribed
FEP Medical Policy Manual Effective Date: October 15, 2018 Related Policies: 2.04.38 Cytochrome P450 Genotype-Guided Treatment Strategy Genotype-Guided Tamoxifen Treatment Description Tamoxifen is prescribed
Authors: Thais Abreu de ALMEIDA 1, Institution:
 Correlation of CYP2D6 s genotype and phenotype with clinicopathological characterization and impact on survival in Brazilian breast cancer population: a prospective study. Authors: Thais Abreu de ALMEIDA
Correlation of CYP2D6 s genotype and phenotype with clinicopathological characterization and impact on survival in Brazilian breast cancer population: a prospective study. Authors: Thais Abreu de ALMEIDA
Pharmacogenomics: Genetic variations in drug metabolism and utilization. Disclosure
 Pharmacogenomics: Genetic variations in drug metabolism and utilization Margaret A. Fitzgerald, DNP, FNP-BC, NP-C, FAANP, CSP, FAAN, DCC, FNAP President, Fitzgerald Health Education Associates, North Andover,
Pharmacogenomics: Genetic variations in drug metabolism and utilization Margaret A. Fitzgerald, DNP, FNP-BC, NP-C, FAANP, CSP, FAAN, DCC, FNAP President, Fitzgerald Health Education Associates, North Andover,
Taking the Pain Out of Pain Management Testing
 Taking the Pain ut of Pain Management Testing Welcome Taking the Pain ut of Pain Management Testing ctober 5, 2010 Your Host: Karen Riba Handout available by clicking on handout icon, in upper right hand
Taking the Pain ut of Pain Management Testing Welcome Taking the Pain ut of Pain Management Testing ctober 5, 2010 Your Host: Karen Riba Handout available by clicking on handout icon, in upper right hand
The role of pharmacogenetics in drug-induced toxicity
 The role of pharmacogenetics in drug-induced toxicity Is there anybody today who does not use any medication? The total turnover of pharmaceutical care in the Netherlands in 2014 was 4.3 billion euros.
The role of pharmacogenetics in drug-induced toxicity Is there anybody today who does not use any medication? The total turnover of pharmaceutical care in the Netherlands in 2014 was 4.3 billion euros.
Pharmacogenomics and Cultural Issues! Y. W. Francis Lam, Pharm.D., FCCP!
 Pharmacogenomics and Cultural Issues in Psychopharmacology Y. W. Francis Lam, Pharm.D., FCCP 1 Pharmacogenomics and Cultural Issues in Psychopharmacology Y. W. Francis Lam, Pharm.D., FCCP Professor of
Pharmacogenomics and Cultural Issues in Psychopharmacology Y. W. Francis Lam, Pharm.D., FCCP 1 Pharmacogenomics and Cultural Issues in Psychopharmacology Y. W. Francis Lam, Pharm.D., FCCP Professor of
Austedo. (deutetrabenazine) New Product Slideshow
 Austedo (deutetrabenazine) New Product Slideshow Introduction Brand name: Austedo Generic name: Deutetrabenazine Pharmacological class: Vesicular monoamine transporter 2 (VMAT2) inhibitor Strength and
Austedo (deutetrabenazine) New Product Slideshow Introduction Brand name: Austedo Generic name: Deutetrabenazine Pharmacological class: Vesicular monoamine transporter 2 (VMAT2) inhibitor Strength and
Clinically Relevant Interactions between Newer Antidepressants and Second-Generation Antipsychotics
 University of Kentucky UKnowledge Psychiatry Faculty Publications Psychiatry 5-2014 Clinically Relevant Interactions between Newer Antidepressants and Second-Generation Antipsychotics Edoardo Spina University
University of Kentucky UKnowledge Psychiatry Faculty Publications Psychiatry 5-2014 Clinically Relevant Interactions between Newer Antidepressants and Second-Generation Antipsychotics Edoardo Spina University
DRUGS THAT ACT IN THE CNS
 DRUGS THAT ACT IN THE CNS Anxiolytic and Hypnotic Drugs Dr Karamallah S. Mahmood PhD Clinical Pharmacology 1 OTHER ANXIOLYTIC AGENTS/ A. Antidepressants Many antidepressants are effective in the treatment
DRUGS THAT ACT IN THE CNS Anxiolytic and Hypnotic Drugs Dr Karamallah S. Mahmood PhD Clinical Pharmacology 1 OTHER ANXIOLYTIC AGENTS/ A. Antidepressants Many antidepressants are effective in the treatment
Cytochrome p450 Genotyping. Description. Section: Medicine Effective Date: July 15, 2015
 2.04.38 Subject: Cytochrome p450 Genotyping Page: 1 of 30 Last Review Status/Date: June 2015 Cytochrome p450 Genotyping Description The cytochrome p450 (CYP450) family is involved in the metabolism of
2.04.38 Subject: Cytochrome p450 Genotyping Page: 1 of 30 Last Review Status/Date: June 2015 Cytochrome p450 Genotyping Description The cytochrome p450 (CYP450) family is involved in the metabolism of
SILOFAST Capsules (Silodosin)
 Published on: 10 Jul 2014 SILOFAST Capsules (Silodosin) Composition SILOFAST-4 Capsules Each hard gelatin capsule contains: Silodosin 4 mg Approved colours used in capsule shell SILOFAST-8 Capsules Each
Published on: 10 Jul 2014 SILOFAST Capsules (Silodosin) Composition SILOFAST-4 Capsules Each hard gelatin capsule contains: Silodosin 4 mg Approved colours used in capsule shell SILOFAST-8 Capsules Each
Cytochrome p450 Genotyping
 Cytochrome p450 Genotyping Policy Number: Original Effective Date: MM.02.004 04/01/2011 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration 12/18/2015 Section: Medicine Place(s) of
Cytochrome p450 Genotyping Policy Number: Original Effective Date: MM.02.004 04/01/2011 Line(s) of Business: Current Effective Date: HMO; PPO; QUEST Integration 12/18/2015 Section: Medicine Place(s) of
Reducing the Anxiety of Pediatric Anxiety Part 2: Treatment
 Reducing the Anxiety of Pediatric Anxiety Part 2: Treatment Lisa Lloyd Giles, MD Medical Director, Behavioral Consultation, Crisis, and Community Services Primary Children s Hospital Associate Professor,
Reducing the Anxiety of Pediatric Anxiety Part 2: Treatment Lisa Lloyd Giles, MD Medical Director, Behavioral Consultation, Crisis, and Community Services Primary Children s Hospital Associate Professor,
AROMASIN 25mg (Tablets)
 APPROVED PACKAGE INSERT AROMASIN SCHEDULING STATUS: S4 PROPRIETARY NAME AND DOSAGE FORM: AROMASIN 25mg (Tablets) COMPOSITION: Each sugar-coated tablet contains 25 mg exemestane. Preservative: methyl p-hydroxybenzoate
APPROVED PACKAGE INSERT AROMASIN SCHEDULING STATUS: S4 PROPRIETARY NAME AND DOSAGE FORM: AROMASIN 25mg (Tablets) COMPOSITION: Each sugar-coated tablet contains 25 mg exemestane. Preservative: methyl p-hydroxybenzoate
SLCO1B1 Pharmacogenetic Competency
 SLCO1B1 Pharmacogenetic Competency Updated on 6/2015 Pre-test Question # 1 Which of the following is not currently a recognized SLCO1B1 phenotype? a) Low function b) Normal function c) Intermediate function
SLCO1B1 Pharmacogenetic Competency Updated on 6/2015 Pre-test Question # 1 Which of the following is not currently a recognized SLCO1B1 phenotype? a) Low function b) Normal function c) Intermediate function
Effect of Multiple-Dose Ketoconazole and the Effect of Multiple-Dose Rifampin on Pharmacokinetics (PK) of the HCV NS3 Protease Inhibitor Asunaprevir
 Effect of Multiple-Dose Ketoconazole and the Effect of Multiple-Dose Rifampin on Pharmacokinetics (PK) of the HCV NS3 Protease Inhibitor Asunaprevir Eley T, 1 He B, 1 Huang S-P, 2 Stonier M, 1 Bedford
Effect of Multiple-Dose Ketoconazole and the Effect of Multiple-Dose Rifampin on Pharmacokinetics (PK) of the HCV NS3 Protease Inhibitor Asunaprevir Eley T, 1 He B, 1 Huang S-P, 2 Stonier M, 1 Bedford
Section 5.2: Pharmacokinetic properties
 Section 5.2: Pharmacokinetic properties SmPC training presentation Note: for full information refer to the European Commission s Guideline on summary of product characteristics (SmPC) SmPC Advisory Group
Section 5.2: Pharmacokinetic properties SmPC training presentation Note: for full information refer to the European Commission s Guideline on summary of product characteristics (SmPC) SmPC Advisory Group
Cytochrome P450 interactions
 Cytochrome P450 interactions Learning objectives After completing this activity, pharmacists should be able to: Explain the mechanism of action of clopidogrel-ppi interaction Assess the risks and benefits
Cytochrome P450 interactions Learning objectives After completing this activity, pharmacists should be able to: Explain the mechanism of action of clopidogrel-ppi interaction Assess the risks and benefits
VESIGARD Tablets (Darifenacin hydrobromide)
 Published on: 10 Jul 2014 VESIGARD Tablets (Darifenacin hydrobromide) Composition VESIGARD 7.5 Extended Release Tablets Each tablet contains: Darifenacin (as a hydrobromide).. 7.5 mg Dosage Form Tablets
Published on: 10 Jul 2014 VESIGARD Tablets (Darifenacin hydrobromide) Composition VESIGARD 7.5 Extended Release Tablets Each tablet contains: Darifenacin (as a hydrobromide).. 7.5 mg Dosage Form Tablets
